Abstract

We synthesized two dTTP analogues for copper-free “click” chemistry-coupling in the active sites of DNA polymerases. We found that in the presence of both analogues, human immunodeficiency virus (HIV) reverse transcriptase (RT) activity was suppressed by up to 93%. This inhibitory effect was not recovered by an excess amount of primer–template unlike that for a conventional HIV RT inhibitor, azidothymidine. This finding may become the basis for the development of efficient in vivo inhibitors of HIV RT and other DNA polymerases.
Introduction
Acquired immune deficiency syndrome AIDS caused by human immunodeficiency virus (HIV) is the top noncurable infectious disease in the world. Although disease management through antiretroviral therapy has greatly increased patient longevity, antiretrovirals remain largely unchanged since their inception in the 1990s.1 Lack of progress has led to the emergence of multidrug resistant HIV, making many of the current therapies obsolete. Nucleotide reverse transcriptase inhibitors (NRTIs) inhibit HIV reverse transcriptase (RT) by chain termination: once incorporated into a growing DNA strand, they fail to support further elongation due to the absence of the 3′-OH group. Initially, NRTIs efficiently inhibited HIV, but the prolonged use of these drugs along with high viral mutagenicity led to the rise of mutant RT forms.2 There are two major mechanisms of NRTI resistance developed by HIV in patients under therapy: (i) ability of RT mutants to discriminate against the triphosphate derivatives of NRTIs3 and (ii) the ability to excise 3′-terminal chain terminators, through phosphorolysis mediated by a pyrophosphate donor, which is likely to be ATP under physiological conditions.3 Interestingly, the excision reaction can be inhibited by binding of the next complementary nucleotide,4 suggesting a route to generate new RT inhibitors that may address the shortcomings of the current NRTIs. For example, HIV RT inhibitors that block both the primer and dNTP-binding sites would prevent ATP binding and the phosphorolysis reaction. One strategy to achieve this goal is to create a bivalent inhibitor that is synthesized in the active site of HIV RT by cross-linking the terminal nucleotide with incoming dNTP. Here, we synthesized two dNTP analogues that can produce a cross-linking product in the HIV RT active site. The system, named here, two-component inhibitor (TCI), was found to efficiently inhibit HIV RT in vitro.
The formation of a bivalent inhibitor in the active site of RT will have therapeutic significance if achieved using otherwise nontoxic biocompatible reagents. We, therefore, turned our attention to the well-established azide/alkyne-based “click” reaction.5 The reaction has been used in a number of exciting applications including fluorescent probes for tracking biomolecular complexes,6 for attaching antibodies and other ligands to nanoemulsions,7 fluorescent labeling of graphene nanoribbons,8 among a number of other well-recognized applications.5,9 The reaction requires Cu+ as a catalyst, which limits the applicability of click reaction for therapeutic purposes due to poor availability of Cu+ in vivo.5 Bertozzi and colleagues10 developed a Cu-independent “click chemistry” in which azides react with cyclooctynes, for example, the dibenzocyclooctyne (DBCO), in a strain-promoted reaction. The reaction proceeds within minutes in live cells with no apparent toxicity (Scheme 1A).11 Here, we took advantage of the Cu+-free “click” reaction to develop a TCI inhibitor of HIV RT.
Scheme 1. Design of Click-Chemistry-Based Two-Component Inhibitor (TCI) for Inactivation of HIV RT.
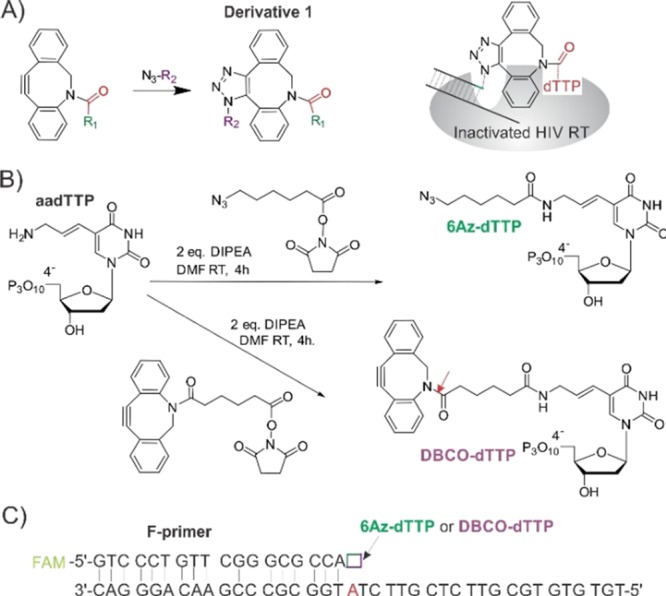
(A) HIV RT-assisted Cu-free “click” reaction and possible HIV RT inhibition. In vivo R1 and R2 could be the residues of NRTI triphosphates or viral RNA/DNA. In this work, R1 and R2 were a dTTP analogue and a DNA primer/template, respectively. (B) Synthetic schemes for 6Az-dTTP and DBCO-dTTP starting from aadTTP. The yields were 31 and 19% for 6Az-dTTP and DBCO-dTTP, respectively. (C) Primer/template (P/T) design for “click” chemistry-based TCI. The primer was fluorescein (FAM) labeled. The protruding 3′ end of the template had only one A (red).
We hypothesized that formation of a cross-linking product between modified incoming NRTI (either R1 or R2 in Scheme 1A) and the primer (either R1 or R2 in Scheme 1A) will form a nonnatural adduct, which will not only terminate primer elongation but also block the dNTP-binding site, thus inhibiting RT more efficiently than conventional chain terminators. In this prove-of-concept study, we synthesized and characterized two dTTP analogues instead of more expensive ddNTP/NRTI analogues. We demonstrated their ability to inhibit HIV RT in an in vitro system.
Results and Discussion
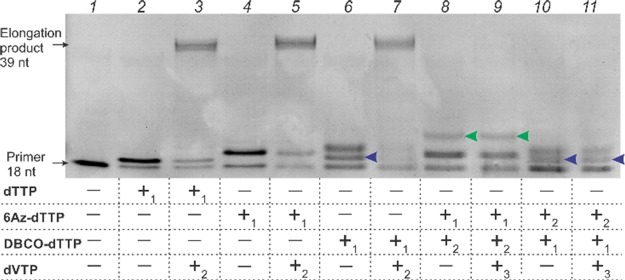
dTTP analogues with an azido group (6Az-dTTP) and with a DBCO group (DBCO-dTTP) were synthesized according to Scheme 1B and procedures described earlier.11,12 Both compounds were characterized by NMR as described in the Supporting Information. We then tested the substrate properties of the dTTP analogues. The primer–template (P/T) substrate was designed to allow the addition of only one dTTP or its analogue to the first position of the growing primer (Scheme 1C). The addition of dVTPs (where V is a mixture of A, C, and G) resulted in the synthesis of a 36-mer fluorescein-labeled DNA, a product of complete primer elongation. Both analogues were efficiently incorporated into the 3′ end of the fluorescein (FAM)-labeled primer (Figure 1, lanes 4 and 6). After incorporation, the modified primers served as substrates for further extension by VTPs (lanes 5 and 7) at a comparable rate to the primer containing natural dTTP (lane 3). This result is in agreement with the early observations that dTTP analogues modified at the fifth position with bulky substituents retain their substrate properties for HIV RT.11−14 Interesting, two elongation products were observed in the presence of DBCO-dTTP (Figure 1, lanes 6, 10, and 11). We hypothesized that the lower band (blue arrows) resulted from DBCO group detachment due to hydrolysis of the amide bond (indicated by the red arrow in Scheme 1B). It is likely that the amide bond is twisted and thus susceptible to hydrolysis as was reported earlier.15,16 Interestingly, a low mobility band was not formed when 6Az-dTTP was added after DBCO-dTTP (lane 10). This finding indicates either the absence of click reaction due to unfavorable steric orientation of the reacting groups or quick degradation of the formed click product when bound to HIV RT.
Figure 1.
Substrate properties of 6Az-dTTP and DBCO-dTTP in primer elongation catalyzed by HIV-1 RT. All samples contained an 18 nt FAM-labeled primer/template complex (Scheme 1C) and 50 nM HIV RT in reaction buffer (50 mM Tris pH 8.0, 100 mM KCl, 8 mM MgCl2, 100 mg/mL BSA, and 2 mM DTT). The samples contained 5 mM dTTP, 6Az-dTTP, DBCO-dTTP, and 5 mM dVTP (V is either A, C, or G) added in different orders as indicated (e.g., +1 means first added). All samples were incubated at 37 °C for 20–30 min at each stage of dNTP addition as described in the Supporting Information followed by analysis by electrophoresis in 15% polyacrylamide gel containing 7 M urea. The product of click reaction is indicated by green arrows (lanes 8 and 9). The primer elongation products with detached DBCO groups are indicated by blue arrows in lanes 6, 10, and 11.
Importantly, when 6Az-dTTP and DBCO-dTTP were used together, primer elongation was inhibited. Indeed, when the primer was elongated in the presence of 6Az-dTTP followed by adding DBCO-dTTP, we observed the formation of a lower mobility product (lanes 8 and 9, green arrows), which is likely to be the product of DBCO-dTTP addition to the azido-group-modified primer (Figure S5). Following the addition of dVTP did not lead to further primer elongation (lane 9). These data indicate that (i) binding of DBCO and azido groups can be brought in close proximity at the RT binding site for the “click” reaction to occur, and (ii) the reaction products inhibit HIV RT. The cross-link between DBCO groups containing primer 6Az-dTTPs was confirmed by electrospray ionization mass spectrometry (Figure S5). A similar inhibition of HIV RT activity was observed when DBCO-dTMP was first added to the 3′ end of the primer by HIV RT (lanes 10 and 11).
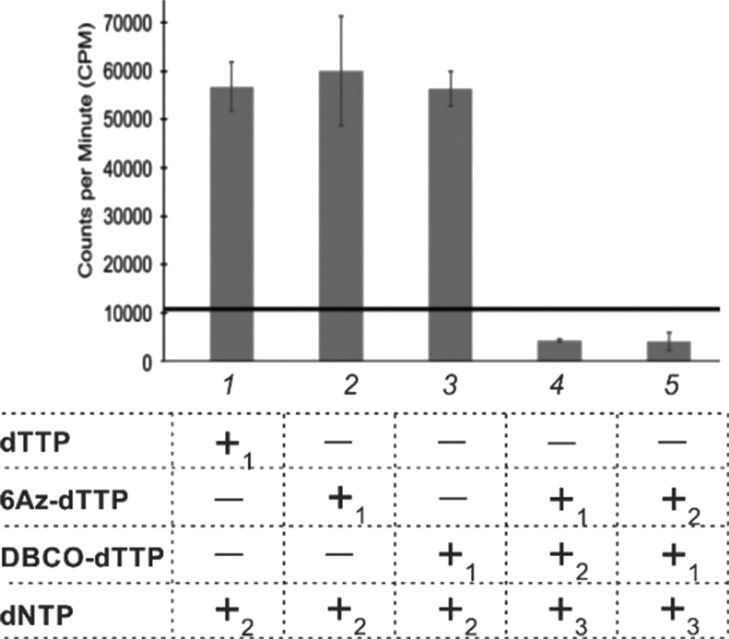
To further confirm HIV RT inactivation, we used a filter-binding assay, a standard method for quantification of DNA polymerase activities.17 Only samples containing the TCI system (Figure 2, bars 4 and 5) inactivated HIV RT efficiently, which correlates with the data shown in Figure 1. Interestingly, TCI efficiently suppressed RT activity regardless of which the dTTP analogue was introduced in the primer first.
Figure 2.
HIV-1 RT activity by TCI and its components measured by the filter binding assay. P/T (1 μM) and HIV RT (50 nM) were incubated with dTTP or first dTTP analogue and incubation for 45 min at 37 °C. Next, second dTTP analogue was added to samples 4 and 5, and all samples were further incubated for 30 min at 37 °C followed by the addition of the dNTP mixture (100 mM dATP, 100 mM dTTP, 100 mM dCTP, and 10 mM [3H]dGTP and incubation for 45 min at 37 °C. All reactions were quenched by the addition of EDTA (0.5 M) and analyzed by filter binding assay as described in the Supporting Information. The data of three independent experiments with single standard deviations are presented.
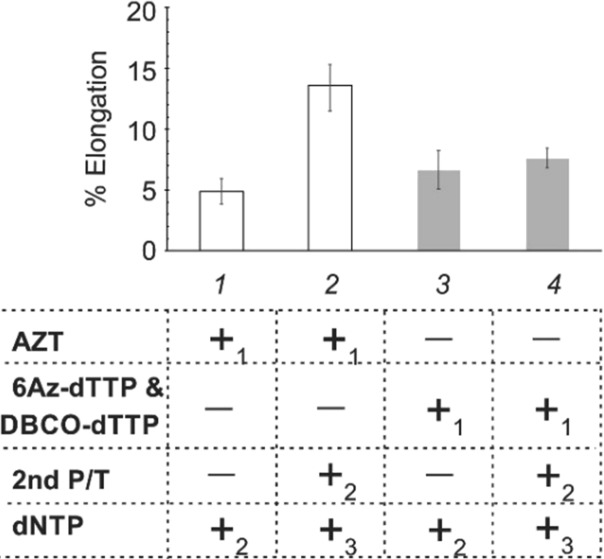
NRTIs used in clinical practice block growing viral DNA but not the RT activity itself. Given that a new primer is available, RT can resume DNA synthesis, thus contributing to the viral survival. This can reduce the efficiency of conventional NRTI and contribute to the development of drug resistant mutants. Data of Figures 1 and 2 suggest the inactivation of HIV RT itself by the TCI system rather than rendering the primer inactive. To further prove this hypothesis, we studied the inhibitory activity of TCI under the condition when the excess amount of DNA substrate is available. We compared the TCI inhibition activity with that of the most common NRTI, azidothymidine (AZT). An almost 3-fold enzyme recovery was observed upon the addition of the excess amount of template to the RT inhibited by AZT (Figure 3, comparing bars 1 and 2). In contrast, the excess amount of P/T did not significantly recover RT inhibited by the TCI system (comparing bars 3 with 4), suggesting that HIV-1 RT binds tightly or irreversibly to the primer-DBCO-dTTP cross-linking product (Scheme 1A), which cannot be displaced by an excess amount of primer.
Figure 3.
TCI inhibitory effect is not significantly recovered by access amount of primer–template (P/T). All reaction mixtures contained P/T and HIV RT in the reaction buffer. In addition, samples 1 and 2 contained azidothymidine (AZT), while samples 3 and 4 contained both 6Az-dTTP and DBCO-dTTP. The samples were incubated for 45 min at 37 °C. Samples 2 and 4 were then treated with 1 μM P/T (to double its concentration), while all samples were treated with the dNTP mixture (100 μM dATP, 100 μM dTTP, 100 μM dCTP, 10 μM [8-H3]-dGTP) followed by incubation for 20 min at 37 °C. All reactions were quenched by the addition of EDTA (0.5 M) and transferred to DE 81 paper for quantification by a filter binding assay (see the Supporting Information for details). The data of three independent experiments with single standard deviations are presented.
Current drug development programs are primarily focused on finding a lead compound that inhibits a protein target by noncovalent binding. Once a lead compound is found, a number of its chemical derivatives are synthesized to achieve high inhibition potency.18 Such inhibition is, however, reversible in most cases and, therefore, inefficient.19 More efficient inhibition of therapeutically significant targets can be achieved by covalent20 or bivalent inhibitors.21−23 For example, apramer-based bivalent inhibitors demonstrated 16.6 times higher inhibition of thrombin than monovalent aprtamers.21
We have been developing a two component (binary) system for highly selective and efficient inactivation of DNA polymerases.24 In this concept, the enzyme is inactivated by interacting with a reagent formed in the active site of the DNA polymerase from two prereactive groups, one of which is located on the 3′ end of the primer, while the other is on incoming dNTP. Earlier, we achieve selective covalent cross-linking and inactivation of DNA polymerases by photoactive TCI.25−28 Importantly, nucleotide-detached prereactive groups did not trigger DNA polymerase inactivation. However, light irradiation is not desirable for therapeutic purposes. In this work, we took advantage of Cu-free click chemistry to produce a two-component inhibitor of HIV RT, which does not depend on light irradiation. We attached the azido and DBCO functional group to dTTP analogues and incorporated one of the groups into the 3′ end of the primer using HIV RT. Following the addition of the second dTTP analogue resulted in formation of the “click” product, which presumably inhibited HIV RT due to tight binding in case when azido-functionality was added first. The inhibition mechanism for the system with the DBCO functional group added first requires further investigations as it cannot be explained by the click product formation, which was not observed. In the presence of both analogues, HIV RT was unable to synthesize DNA even upon the addition of the access amount of DNA substrate, thus indicating an irreversible inhibition mode.
We demonstrated the feasibility of inhibiting HIV RT using a bivalent inhibitor composed of azido and DBCO components attached to nonchain terminating dTTP analogues. We hypothesize that the approach can be used as new in vivo HIV RT inhibitors. To adapt this approach of in vivo inhibition of HIV RT, it is easy to envision synthesis of chain-terminating forms of DBCO and azido analogues. Incorporation of the first NRTI-click chemistry component will terminate DNA synthesis followed by binding of the second component, which will couple with the primer, thus locking HIV RT in tightly bound unproductive complex in which both the RT and viral primer–template are inactivated. This inactivation scheme may become more efficient than currently used NRTIs.
Conclusions
We synthesized two dTTP analogues, one of each is conjugated in an azido component and another with an alkyne component. When added together (a two-component system), the two analogues strongly inhibited HIV RT DNA polymerase activity. Unlike the state-of-the-art inhibitor AZT, the inhibition effect was not recovered by the access amount of primer–template, which suggests a stronger inhibitory effect. The designed and tested two-component inhibitor promises to become a more efficient inhibitor of HIV RT than conventional NRTIs and should be tested in vivo.
Experimental Section
Analogues of deoxythymidine-5′-triphosphate (dTTP) were synthesized from dUTP (Sigma-Aldrich, Missouri) and characterized as described below. Primer: 5′-GTC CCT GTT CGG GCG CCA and template: 5′–TGT GTG TGC GTT CTC GTT CTA TGG CGC CCG AAC AGG GAC were custom-made by Integrated DNA Technologies, Inc. (Coralville, IA). T7 RNA polymerase was purchased from New England Biolabs (Ipswich, MA). HIV-1 RT was from Worthington Biochemical Corporation (Lakewood, NJ). Electrospray ionization (ES) mass spectrometry (MS) was conducted by Florida University facilities. 6Az-dTTP and DBCO-dTTP based on the procedures described previously.11,12,29
Acknowledgments
The authors are grateful to Dr. Yulia V. Gerasimova for helpful discussion and corrections. This work was supported and funded by the start-up funds from College of Science University of Central Florida.
Supporting Information Available
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acsomega.9b03942.
6Az-dTTP and DBCO-dTTP synthesis and characterization (PDF)
Author Present Address
† Current address: Indiana University School of Medicine, 980 W. Walnut Street, Indianapolis, Indiana 46202, United States (E.M.C.).
The authors declare no competing financial interest.
Supplementary Material
References
- Zhan P.; Pannecouque C.; De Clercq E.; Liu X. Anti-HIV Drug Discovery and Development: Current Innovations and Future Trends. J. Med. Chem. 2016, 59, 2849–2878. 10.1021/acs.jmedchem.5b00497. [DOI] [PubMed] [Google Scholar]
- Wainberg M. A.; Sawyer J. P.; Montaner J. S.; Murphy R. L.; Kuritzkes D. R.; Raffi F. Challenges for the clinical development of new nucleoside reverse transcriptase inhibitors for HIV infection. Antiviral Ther. 2005, 10, 13–28. [PubMed] [Google Scholar]
- Menéndez-Arias L. Molecular basis of human immunodeficiency virus type 1 drug resistance: overview and recent developments. Antiviral Res. 2013, 98, 93–120. 10.1016/j.antiviral.2013.01.007. [DOI] [PubMed] [Google Scholar]
- Meyer P. R.; Matsuura S. E.; Schinazi R. F.; So A. G.; Scott W. A. Differential removal of thymidine nucleotide analogues from blocked DNA chains by human immunodeficiency virus reverse transcriptase in the presence of physiological concentrations of 2′-deoxynucleoside triphosphates. Antimicrob. Agents Chemother. 2000, 44, 3465–3472. 10.1128/AAC.44.12.3465-3472.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thirumurugan P.; Matosiuk D.; Jozwiak K. Click chemistry for drug development and diverse chemical-biology applications. Chem. Rev. 2013, 113, 4905–4979. 10.1021/cr200409f. [DOI] [PubMed] [Google Scholar]
- Jun J. V.; Haney C. M.; Karpowicz R. J. Jr.; Giannakoulias S.; Lee V. M.-Y.; Petersson E. J.; Chenoweth D. M. A ″Clickable″ Photoconvertible Small Fluorescent Molecule as a Minimalist Probe for Tracking Individual Biomolecule Complexes. J. Am. Chem. Soc. 2019, 141, 1893–1897. 10.1021/jacs.8b13094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amirshaghaghi A.; Altun B.; Nwe K.; Yan L.; Stein J. M.; Cheng Z.; Tsourkas A. Site-Specific Labeling of Cyanine and Porphyrin Dye-Stabilized Nanoemulsions with Affibodies for Cellular Targeting. J. Am. Chem. Soc. 2018, 140, 13550–13553. 10.1021/jacs.8b07866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joshi D.; Hauser M.; Veber G.; Berl A.; Xu K.; Fischer F. R. Super-Resolution Imaging of Clickable Graphene Nanoribbons Decorated with Fluorescent Dyes. J. Am. Chem. Soc. 2018, 140, 9574–9580. 10.1021/jacs.8b04679. [DOI] [PubMed] [Google Scholar]
- Kacprzak K.; Skiera I.; Piasecka M.; Paryzek Z. Alkaloids and Isoprenoids Modification by Copper(I)-Catalyzed Huisgen 1,3-Dipolar Cycloaddition (Click Chemistry): Toward New Functions and Molecular Architectures. Chem. Rev. 2016, 116, 5689–5743. 10.1021/acs.chemrev.5b00302. [DOI] [PubMed] [Google Scholar]
- Baskin J. M.; Prescher J. A.; Laughlin S. T.; Agard N. J.; Chang P. V.; Miller I. A.; Lo A.; Codelli J. A.; Bertozzi C. R. Copper-free click chemistry for dynamic in vivo imaging. Proc. Natl. Acad. Sci. 2007, 104, 16793–16797. 10.1073/pnas.0707090104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Godovikova T. S.; Kolpashchikov D. M.; Orlova T. N.; Richter V. A.; Ivanova T. M.; Grochovsky S. L.; Nasedkina T. V.; Victorova L. S.; Poletaev A. I. 5-[3-(E)-(4-azido-2,3,5,6-tetrafluorobenzamido)propenyl-1]-2′-deoxy-uridine-5′-triphosphate substitutes for thymidine-5′-triphosphate in the polymerase chain reaction. Bioconjugate Chem. 1999, 10, 529–537. 10.1021/bc980144r. [DOI] [PubMed] [Google Scholar]
- Kolpashchikov D. M.; Ivanova T. M.; Boghachev V. S.; Nasheuer H.-P.; Weisshart K.; Favre A.; Pestryakov P. E.; Lavrik O. I. Synthesis of base-substituted dUTP analogues carrying a photoreactive group and their application to study human replication protein A. Bioconjugate Chem. 2000, 11, 445–451. 10.1021/bc990102i. [DOI] [PubMed] [Google Scholar]
- Le Grice S. F. J. Human immunodeficiency virus reverse transcriptase: 25 years of research, drug discovery, and promise. J. Biol. Chem. 2012, 287, 40850–40857. 10.1074/jbc.R112.389056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zakharenko A. L.; Kolpashchikov D. M.; Khodyreva S. N.; Lavrik O. I.; Menéndez-Arias L. Investigation of the dNTP-binding site of HIV-1 reverse transcriptase using photoreactive analogs of dNTP. Biochemistry (Moscow) 2001, 66, 999–1007. 10.1023/A:1012373626717. [DOI] [PubMed] [Google Scholar]
- Aubé J. A new twist on amide solvolysis. Angew. Chem., Int. Ed. 2012, 51, 3063–3065. 10.1002/anie.201108173. [DOI] [PubMed] [Google Scholar]
- Hutchby M.; Houlden C. E.; Haddow M. F.; Tyler S. N. G.; Lloyd-Jones G. C.; Booker-Milburn K. I. Switching pathways: room-temperature neutral solvolysis and substitution of amides. Angew. Chem., Int. Ed. 2012, 51, 548–551. 10.1002/anie.201107117. [DOI] [PubMed] [Google Scholar]
- Mescalchin A.; Wünsche W.; Laufer S. D.; Grohmann D.; Restle T.; Sczakiel G. Specific binding of a hexanucleotide to HIV-1 reverse transcriptase: a novel class of bioactive molecules. Nucleic Acids Res. 2006, 34, 5631–5637. 10.1093/nar/gkl533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mayr L. M.; Bojanic D. Novel trends in high-throughput screening. Curr. Opin. Pharmacol. 2009, 9, 580–588. 10.1016/j.coph.2009.08.004. [DOI] [PubMed] [Google Scholar]
- Singh J.; Petter R. C.; Baillie T. A.; Whitty A. The resurgence of covalent drugs. Nat. Rev. Drug Discovery 2011, 10, 307–317. 10.1038/nrd3410. [DOI] [PubMed] [Google Scholar]
- Baillie T. A. Targeted Covalent Inhibitors for Drug Design. Angew. Chem., Int. Ed. 2016, 55, 13408–13421. 10.1002/anie.201601091. [DOI] [PubMed] [Google Scholar]
- Kim Y.; Cao Z.; Tan W. Molecular assembly for high-performance bivalent nucleic acid inhibitor. Proc. Natl. Acad. Sci. U. S. A. 2008, 105, 5664–5669. 10.1073/pnas.0711803105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang J.; Zang W.; Liu J.; Zheng W. Bivalent SIRT1 inhibitors. Bioorg. Med. Chem. Lett. 2017, 27, 180–186. 10.1016/j.bmcl.2016.11.082. [DOI] [PubMed] [Google Scholar]
- Wong M. L.; Murphy J.; Harrington E.; Gower C. M.; Jain R. K.; Schirle M.; Thomas J. R. Examining the influence of specificity ligands and ATP-competitive ligands on the overall effectiveness of bivalent kinase inhibitors. Proteome Sci. 2016, 15, 17. 10.1186/s12953-017-0125-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kolpashchikov D. M. Superselective labelling of proteins: approaches and techniques. J. Biomol. Struct. DynJ. Biomol Struct Dyn. 2003, 21, 55–64. 10.1080/07391102.2003.10506905. [DOI] [PubMed] [Google Scholar]
- Kolpashchikov D. M.; Rechkunova N. I.; Dobrikov M. I.; Khodyreva S. N.; Lebedeva N. A.; Lavrik O. I. Sensitized photomodification of mammalian DNA polymerase β. A new approach for highly selective affinity labeling of polymerases. FEBS Lett. 1999, 448, 141–144. 10.1016/S0014-5793(99)00354-3. [DOI] [PubMed] [Google Scholar]
- Lavrik O. I.; Kolpashchikov D. M.; Prasad R.; Sobol R. W.; Wilson S. H. Binary system for selective photoaffinity labeling of base excision repair DNA polymerases. Nucleic Acids Res. 2002, 30, e73 10.1093/nar/gnf073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cornett E. M.; Gerasimova Y. V.; Kolpashchikov D. M. Two-component covalent inhibitor. Bioorg. Med. Chem. 2013, 21, 1988–1991. 10.1016/j.bmc.2013.01.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lebedeva N. A.; Kolpashchikov D. M.; Rechkunova N. I.; Khodyreva S. N.; Lavrik O. I. A binary system of photoreagents for high-efficiency labeling of DNA polymerases. Biochem. Biophys. Res. Commun. 2001, 287, 530–535. 10.1006/bbrc.2001.5623. [DOI] [PubMed] [Google Scholar]
- Godovikova T. S.; Kolpashchikov D. M.; Orlova T. N.; Richter V. A. New photoreactive deoxynucleoside-5′-triphosphates substitutes for thymidine-5′-triphosphate in the polymerase chain reaction. FASEB J. 1997, 11, A1367. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.