Abstract
Background
Severe pneumonia is responsible for great mortality and morbidity worldwide, and early-applied effective anti-infective therapy can improve the prognosis of patients. However, identification of infectious agents in severe pneumonia remains a major challenge so far. In this study, the potential utility of transmission electron microscopy (TEM) in detecting nonbacterial pathogens in patients with severe pneumonia was retrospectively evaluated.
Materials and Methods
A total of 106 patients diagnosed with severe pneumonia at our hospital from September 2015 to December 2017 were included, and their baseline clinical characteristics were collected. Nonbacterial infectious agents detected by TEM in bronchoalveolar lavage fluid (BALF) and serological tests were summarized. The detection rates were further compared between TEM and serological tests.
Results
BALF examination under the transmission electron microscope revealed 24 viruses, 16 mycoplasmas, 18 chlamydia, 2 fungi and 74 bacteria in 99 samples, among which 61 samples were mixed infections. The combined use of serological tests and TEM significantly improved the detection rate of nonbacterial infectious agents in patients with severe pneumonia.
Conclusions
Our data support that implementation of TEM could improve the sensitivity for detecting viruses, atypical pathogens and mixed infections in BALF from patient of severe pneumonia. Therefore, TEM may be used as an auxiliary diagnostic method of other microbiological tests in severe pneumonia.
Key Indexing Terms: Severe pneumonia, Transmission electron microscopy, Bronchoalveolar lavage fluid, Pathogens, Etiologic diagnosis
INTRODUCTION
Pneumonia affects approximately 450million people globally and causes nearly 4million deaths each year.1 Severe pneumonia is a common reason for intensive care unit admission and represents a major concern for physicians due to its high mortality rate.2, 3 Without appropriate treatment, it can rapidly progress to respiratory failure, septic shock and even cause death within several days. Early etiologic diagnosis, which would facilitate the prompt initiation of anti-infective therapy, is critical for improving the clinical outcomes of patients with severe pneumonia.
Severe pneumonia may have a bacterial, viral, fungal or other atypical etiology, including Mycoplasma pneumoniae, Chlamydia pneumoniae and Legionella pneumophila. Identification of causative agents in severe nonbacterial pneumonia is very challenging. Microbiological culture of lower respiratory tract specimens, blood and lung tissue is considered as the gold standard for etiologic diagnosis of pneumonia. However, for viruses and atypical pathogens, culture analysis is rarely used due to delay in the results and complicated procedures.4, 5, 6, 7 While serological tests and polymerase chain reaction (PCR) targeting specific pathogens are routinely deployed, their clinical usefulness is limited in identifying a novel or rare pathogen and mixed infections.8, 9 It has been reported that causative agents cannot be confirmed in nearly half of pneumonia episodes despite careful testing.10 Thus, anti-infective therapy is empirically applied based on clinical features and thoracic images in the early stage of pneumonia, which may increase the rate of treatment failure and cause drug-related toxicity. Therefore, there is an urgent need to develop a comprehensive assay system for prompt etiologic diagnosis of severe pneumonia.
Transmission electron microscopy (TEM) has been used in diagnostic microbiology since the 1960s, and had a profound impact on our knowledge and understanding of microorganisms.11 While TEM is well-recognized as a useful tool for rapid morphologic identification of infectious agents in emergent situations and infectious diseases of unknown cause,12, 13 its potential diagnostic value in nonbacterial etiology of severe pneumonia remains unexplored yet.
Bronchoalveolar lavage is a common method to obtain valid samples from the lower respiratory tract. Bronchoalveolar lavage fluid (BALF) has the advantages of improving the detection of etiologic agents in community-acquired pneumonia and being suitable for multiple detection methods, including microscope.14 Therefore, in this study, we retrospectively analyzed the potential diagnostic value of TEM examination of BALF samples in severe pneumonia.
METHODS
Participants
A retrospective review of the medical records was made of all patients who were diagnosed with severe pneumonia and treated at Xiangya Hospital, Central South University (Changsha, Hunan, China) between September 2015 and December 2017. The diagnosis of severe pneumonia was made according to the Infectious Diseases Society of America and the American Society for Microbiology criteria.15 Major criteria include the requirement for mechanical ventilation or need for vasopressors. Minor criteria are listed as follows: (1) BUN >20 mg/dL; (2) confusion or disorientation; (3) hypotension requiring aggressive fluid resuscitation; (4) hypothermia with core temperature <96.8 F; (5) leukopenia with white blood cell count <4000 per mm3; 6) multilobar infiltrates; (7) PaO2 to FiO2 ratio <250; (8) respiratory rate >30 breaths/minute; (9) thrombocytopenia with platelet count <100,000 per mm3. Patients who met at least 1 major criterion and ≥3 minor criteria were diagnosed as severe pneumonia. BALF samples from patients who had failed initial anti-infective treatments were examined by TEM. Patients were excluded if they were newborns who never left the hospital or if TEM examination and serological tests were not performed. A total of 106 patients were included in this study.
Bronchoalveolar Lavage
Bronchoalveolar lavage was performed within 48hours after patient admission by flexible bronchoscopy under local anesthesia with lidocaine in a single-center (Xiangya Hospital) setting using standard methods. Briefly, the sampling area was selected based on the infiltration location on a chest radiograph. Three 20 mL fractions of sterile saline were instilled into the relevant lobe and segment of the lung. BALF was retrieved by gentle syringe suction, put into sterile containers, and immediately submitted to the Department of Pathology for TEM examination.
TEM Examination of BALF
TEM examination of BALF was performed by the Department of Pathology, Xiangya Hospital. BALF was centrifuged at 3000rpm for 10 minutes and the supernatant was removed. Subsequently, the pellet remaining on the bottom of the tube after centrifuge was fixed in 2.5% glutaraldehyde and 1% osmium tetroxide, dehydrated through a graded series of acetone (50%, 70%, 90% and 100%) and embedded in a mixture of epon substitute and araldite. A total of 6 thin sections (50-100 nm) from each sample were stained with 3% uranyl acetate and Reynolds's lead citrate. Imaging was performed at 200kV using a Hitachi H7700 Transmission Electron Microscope (Hitachi, Tokyo, Japan). Digital images of the specimens were acquired using an AMT Advantage XR 12 CCD camera (AMT, Danvers, MA) and analyzed by 2 experienced electron microscopists. In total, 48hours are needed for TEM examination of each batch of BALF samples.
Serological Tests
Serological tests were performed by the Department of Clinical Laboratory, Xiangya Hospital. Serum samples were collected from patients before antibiotic treatment (within 24hours after hospital admission) if feasible, depending on the patients' situations. Serum immunoglobulin (Ig)M antibodies specific to common pathogens in the respiratory tract (including Adenovirus, Respiratory syncytial virus, Influenza B virus, Influenza A virus, Human parainfluenza viruses, M pneumoniae, C pneumoniae, L pneumophila and Coxiella burneti) were detected using indirect immunofluorescence assay.
Statistical Analysis
The McNemar test was performed to compare the detection rates of different diagnostic methods. All statistical analyses were carried out using SPSS Statistics version 24.0 (IBM Corporation, Armonk, NY). A P value < 0.05 was considered to be statistically significant.
To ensure the quality of any reporting of the results from the present study, the recommended guidelines based on the criteria published by the Standards for Reporting of Diagnostic Accuracy initiative for the accurate reporting of investigations of diagnostic studies were followed.16
RESULTS
Baseline Characteristics of Patients with Severe Pneumonia
Baseline characteristics of 106 patients included in this study were summarized in Table1 . Eighty-one of 106 (76.4%) of these patients were males, and 86 of 106 (81.1%) were older than 40years of age. Forty-eight out of 106 patients (45.3%) did not have a smoking history. The most common clinical manifestations in these patients included fever, cough and dyspnea. Consolidation, ground glass opacity and pleural effusion were most frequently found by computed tomography scan. During the duration of hospital stay, 18 of 106 of these patients (17.0%) progressed into acute respiratory distress syndrome. Patients were usually complicated with chronic renal failure, heart disease and hypertension. A total of 100 patients (94.3%) received antibiotics before admission to our hospital. The average length of hospital stay was 12.9days (ranging from 5 to 30 days), and 86 of 106 (81.1%)patients got significantly relieved and discharged from our hospital, while comprehensive therapies did not work on the remaining 18.9% of patients.
TABLE 1.
Baseline clinical characteristics of patients.
| Clinical characteristics | Patients number (n = 106) |
|---|---|
| Gender (%) | |
| Male | 81 (76.4) |
| Female | 25 (23.6) |
| Age (years) (%) | |
| ≤18 | 8 (7.5) |
| 19-40 | 12 (11.3) |
| 41-60 | 44 (41.5) |
| >60 | 42 (39.6) |
| Smoking history (%) | |
| Nonsmoker | 48 (45.3) |
| Present smoker | 34 (32.1) |
| Former smoker | 24 (22.6) |
| Clinical manifestations (%) | |
| Fever | 83 (78.3) |
| Cough | 78 (73.6) |
| Expectoration | 36 (34.0) |
| Dyspnea | 72 (67.9) |
| CT findings (%) | |
| Consolidation | 56 (52.8) |
| GGO | 38 (35.8) |
| Multiple nodules/masses | 8 (7.5) |
| Pleural effusion | 22 (20.8) |
| ARDS (%) | 18 (17.0) |
| Comorbidities (%) | |
| Hypertension | 29 (27.4) |
| Heart disease | 34 (32.1) |
| Chronic renal failure | 36 (34.0) |
| Diabetes mellitus | 14 (13.2) |
| COPD | 16 (15.1) |
| Cerebrovascular events | 14 (13.2) |
| Malignancy | 10 (9.4) |
| Hypothyroidism | 9 (8.5) |
| Immunosuppression | 2 (1.9) |
| Pre-admission antibiotics (%) | 100 (94.3) |
| Average hospital length of stay (days) | 12.9 |
| Hospital outcome (%) | |
| Treatment failure | 20 (18.9) |
| Relieved and discharged | 86 (81.1) |
Abbreviations: ARDS, acute respiratory distress syndrome; COPD, chronic obstructive pulmonary disease; CT, computer tomography; GGO, ground glass opacity.
Pathogens Detected by TEM in BALF
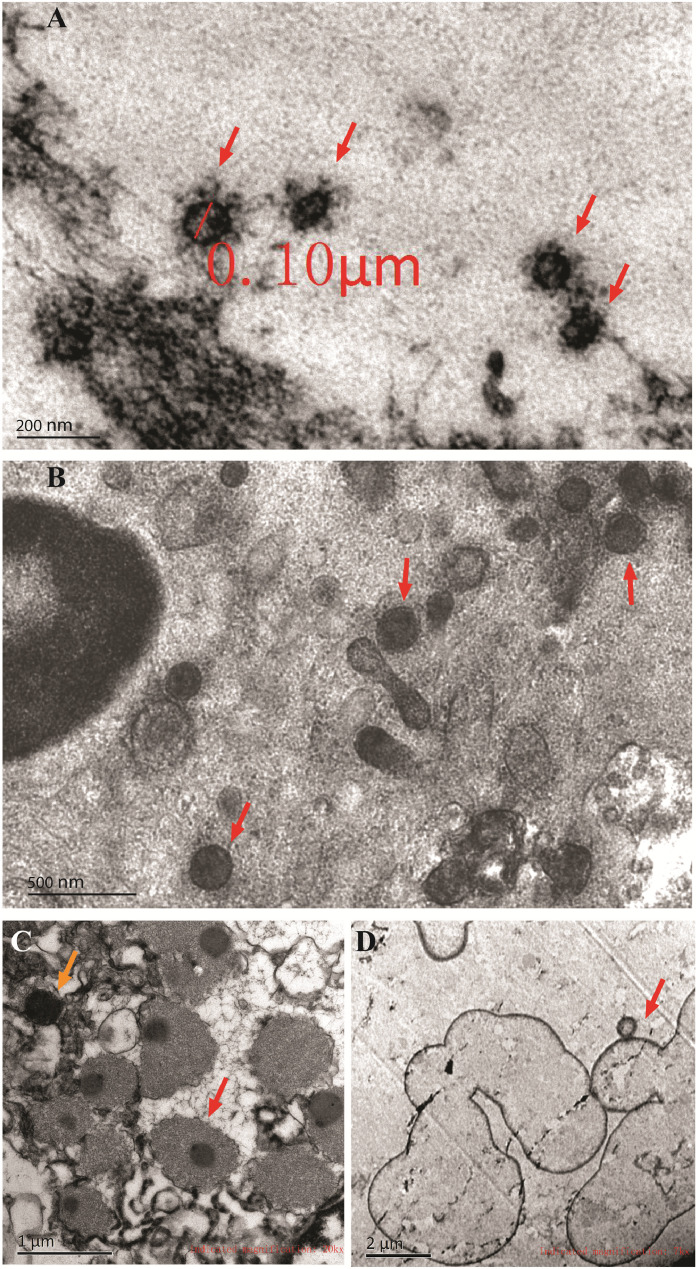
Representative transmission electron micrographs images of pathogens in BALF were shown in Figure1 and Figure S1, including virus, chlamydia, mycoplasma, fungus, coccus and bacillus. Generally, categories of pathogens were determined according to their sizes, shapes and characteristic structures. For example, most viruses have a diameter between 20 and 300nm. There are 4 main morphological virus types, including helical, icosahedral, enveloped and complex symmetric viruses. Mycoplasma is a mollicute genus of bacteria that lack a cell wall around their cell membranes and have a diameter of 100-300nm, while chlamydia is a genus of pathogenic bacteria that are obligate intracellular parasites, which may be found in the form of an elementary body or a reticulate body. Besides, fungi usually grow as hyphae, which are cylindrical, thread-like structures 2-10 µm in diameter and up to several centimeters in length. Fungi also disperse spores or spore-containing propagules for reproduction purpose.
Figure1.
Representative transmission electron micrographs of nonbacterial pathogens detected in BALF from patients with severe pneumonia. A, Coronavirus (red arrows), showing a diameter of about 100nm and crown-like projections on the surfaces; B, mycoplasma, showing nonhelical spherical cell morphology with a diameter of 200-300nm and the absence of cell wall; C, chlamydia, showing intracellular irregular spherical cell morphology with a diameter of 0.3-0.8μm. Both forms of chlamydia were seen, including the elementary body (smaller, orange arrow) and the reticulate body (larger, red arrow); D, fungus, showing spheroid fungal cells with a diameter of 2-5μm, and some were dispersing spores (red arrow). Scale bars were marked in each panel.
In total, TEM examination of BALF revealed 24 virus-positive samples, 16 mycoplasma-positive samples, 18 chlamydia-positive samples, 2 fungus-positive samples and 74 bacterium-positive samples, while no pathogens in the remaining 7 samples were detected. A total of 61 samples were found to be mixed infections by TEM (Table2 ). A total of 42 bacteria in 33 samples were identified by bacterial culture analyses of lower respiratory tract specimens, among which 9 samples were mixed infections (Table S1). Even though TEM showed a higher detection rate of bacteria than cultures, it could not differentiate bacterial strains and provide any information of antibiotic sensitivity, thus it had little diagnostic value in detecting bacteria in BALF.
TABLE 2.
Pathogens identified by TEM examination of BALF.
| Pathogens | Number of positive samples |
|---|---|
| Viruses | 24 |
| Mycoplasma | 16 |
| Chlamydia | 18 |
| Fungi | 2 |
| Bacteria | 74 |
| Cocci | 45 |
| Bacilli | 39 |
| Mixed infections | 61 |
Abbreviations: BALF, bronchoalveolar lavage fluid; TEM, transmission electron microscopy.
Comparison of Pathogen-positive Samples Detected by TEM and Serological Tests
Nonbacterial pathogens such as viruses, M pneumoniae, C pneumoniae and L pneumophila cannot be identified by routine culture method. Specific isolation and culture tests, which are labor-intensive and time-consuming, are rarely used in the hospital for etiologic diagnosis. Instead, pathogen-specific IgM antibody in serum is considered as significantly indicative and routinely measured when there is a clinical suspicion of infection with these pathogens. As shown in Table3 , TEM examination of BALF successfully confirmed most pathogen-positive samples indicated by serological tests, including 14 of 16 (87.5%) virus, 4 of 6 (66.7%) mycoplasma and 2 of 2 (100%) chlamydia. Strikingly, TEM also detected infectious agents in a number of samples from patients reported as negative results in serological tests (Table3).
TABLE 3.
Comparison of viruses, mycoplasma or chlamydia positive samples detetcted by serological tests and TEM.
| TEM | ||||
|---|---|---|---|---|
| Pathogens | Serum IgM | Positive | Negative | Total |
| Viruses | Positive | 14 | 2 | 16 |
| Negative | 10 | 80 | 90 | |
| Total | 24 | 82 | 106 | |
| Mycoplasma | Positive | 4 | 2 | 6 |
| Negative | 12 | 88 | 100 | |
| Total | 16 | 90 | 106 | |
| Positive | 2 | 0 | 2 | |
| Chlamydia | Negative | 16 | 88 | 104 |
| Total | 18 | 88 | 106 | |
Abbreviations: IgM, immunoglobulin M; TEM, transmission electron microscopy.
Improved Detection Rate of Infectious Agents by Combining TEM with Serological Tests
To investigate whether the application of TEM could facilitate etiologic diagnosis of severe pneumonia, we further compared the pathogen detection rate of serological tests and TEM. It revealed that the detection rate of TEM was significantly higher than serological tests (Table4 ). Furthermore, the number of samples confirmed as pathogen-positive markedly increased when combining serological tests and TEM (Table4).
TABLE 4.
Detection rates of infectious agents by combining TEM with serological test.
| Methods | Viruses | Mycoplasma | Chlamydia |
|---|---|---|---|
| IgM assay | 16/106 | 6/106 | 2/106 |
| TEM | 24/106a | 16/106a | 18/106a |
| IgM assay + TEM | 30/106a | 18/106a | 18/106a |
Abbreviations: IgM, immunoglobulin M; TEM, transmission electron microscopy
P < 0.05vs. culture (or IgM).
DISCUSSION
Identification of infectious agents is challenging in patients with severe pneumonia. In the present study, we found that TEM had a good detectability of nonbacterial pathogens in BALF obtained from patients with severe pneumonia, and the implementation of TEM could significantly improve the diagnostic sensitivity of nonbacterial etiology. To the best of our knowledge, this is the first retrospective study reporting the usefulness of TEM in the etiologic diagnosis of severe pneumonia.
Severe pneumonia is life-threatening. Without any clear indications of causative agents, antibiotic treatment is usually determined empirically, which may cause severe drug resistance, higher cost and increased mortality risk. Therefore, microbiological tests are strongly recommended for patients with severe pneumonia, in whom the probability of changing the empirical treatment is high, to reduce treatment failure and prevent antibiotic overuse.10, 17 , 18 As the gold standard of etiologic diagnosis, microbiological culture is not efficient in severe pneumonia due to its time-consuming procedures and high incidence of false negative results.19 More importantly, culture analysis is rarely used to detect viruses and atypical pathogens due to delay in the results and complicated procedures. PCR and serological tests are popular due to the high-speed and capacity of processing large quantities of samples at the same time; however, they are not suitable for detecting novel/rare pathogens or mixed infections. Therefore, additional techniques that could facilitate etiologic diagnosis of severe pneumonia are urgently needed, especially when initial empirical anti-infective therapy has failed. In the present study, our data demonstrated that TEM had a good sensitivity in detecting nonbacterial infectious agents in BALF. The combined use of serological tests with TEM improved the detection rate of nonbacterial pathogens. Furthermore, it has been reported that mixed infections, defined as at least 2 types of pathogens contained in 1 sample, occur in more than half of cases of community-acquired pneumonia and are associated with more severe inflammatory disorders.20, 21 Interestingly, based on our data, TEM appeared to be far more sensitive in detecting mixed infections when compared with serological tests. In spite of these combined detection modalities, the overall rate of treatment failure was 20 of 106 (18.9%) in this study, which we thought was caused by severe systemic complications and antibiotic resistance.
TEM has been widely used in diagnostic microbiology for decades. Even though the clinical usefulness of TEM has been gradually declined as the popularization of molecular and immunological techniques, it offers significant advantages in the detectability of viruses and atypical pathogens and the capacity of detecting multiple pathogens present within a sample by a single test.13 Moreover, TEM examination of BALF samples only takes 48hours, which means that clinical physicians could get fast information of what kind of infectious agents are existing in the lungs of patients. Unlike serological test and PCR, TEM does not require any organism-specific reagents for detecting pathogens.9 However, there are several drawbacks to the TEM technique. First of all, samples must be processed individually for TEM examination, leading to a low throughput. The field of view under TEM is also relatively small, increasing the possibility that the region analyzed may not be representative of the whole sample, which could cause false negative results. In addition, the accuracy of TEM examination is dependent on the skill and experience of microscopists. Thus, in this study, 2 experienced electron microscopists were involved in imaging analysis of each sample to avoid misdiagnosis. Furthermore, the diagnostic value of TEM in bacterial pneumonia is limited, because routine TEM cannot differentiate bacterial strains and provide any information of antibiotic sensitivity, thus, had few benefits to subsequent antibiotic therapies. So far, advanced electron microscopy (EM) techniques have been applied to microbiology in basic and clinical studies, including cryogenic electron tomography, immuno-EM,22 3-dimensional EM,23 correlated light microscopy and TEM.24 When TEM is integrated with immunohistochemistry or other molecular labeling techniques, it would become practical for electron microscopists and clinical physicians to distinguish various pathogens on the strain level under TEM.25 Recently, rapid methods of sample preparation and transportable EM equipment, which makes EM an easier and faster tool to use, have also been reported to improve the identification of infectious agents.26, 27, 28 These studies and our data collectively support that in future, TEM may be used along with other diagnostic tools, as one part of a comprehensive assay system for microbiological diagnosis of severe pneumonia.
Several imitations should be noted in the present study. First of all, no BALF samples from healthy volunteer subjects were obtained, which made the diagnostic specificity of TEM unevaluable and the interpretation of TEM examination difficult, which we plan to address in future studies. In addition, the major disadvantage of TEM used in this study was that it did not identify a pathogen on the strain level. Hopefully, the application of more advanced techniques and combined use with other microbial tests would enhance the diagnostic accuracy of TEM, thus further improving its diagnostic value. Moreover, even though TEM examination of BALF appears to have a potential utility in improving diagnostic sensitivity in severe pneumonia caused by nonbacterial pathogens, its cost-effectiveness is still unknown. Therefore, clinical trials with large patient numbers must be conducted to determine whether the implementation of TEM could benefit patients with severe pneumonia through promoting the prompt initiation of anti-infective treatments.
CONCLUSIONS
In summary, our data supported that TEM had a good sensitivity in detecting nonbacterial pathogens in BALF, including viruses, atypical pathogens and mixed infections. The combined use of serological tests and TEM could improve the detection rate of nonbacterial infectious agents in patients with severe pneumonia. Considering the severity and rapid progression of severe pneumonia, a single diagnostic method is not recommended in such a case. Implementation of TEM would be a judicious strategy for the etiologic diagnosis of severe pneumonia caused by nonbacterial pathogens in the future.
Footnotes
Funding: This research was supported in part by grants from the National Natural Science Foundation of China (81600025, 81873406 and 81502699), and the National Key Research and Development Program of China (SQ2016YFSF110276).
Disclosures: The authors declare that they have nothing to disclose.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.1016/j.amjms.2018.11.012.
Appendix. Supplementary materials
REFERENCES
- 1.Ruuskanen O., Lahti E., Jennings L.C. Viral pneumonia. Lancet. 2011;377(9773):1264–1275. doi: 10.1016/S0140-6736(10)61459-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Restrepo M.I., Mortensen E.M., Velez J.A. A comparative study of community-acquired pneumonia patients admitted to the ward and the ICU. Chest. 2008;133(3):610–617. doi: 10.1378/chest.07-1456. [DOI] [PubMed] [Google Scholar]
- 3.Woodhead M., Welch C.A., Harrison D.A. Community-acquired pneumonia on the intensive care unit: secondary analysis of 17,869 cases in the ICNARC Case Mix Programme Database. Crit Care. 2006;10(suppl 2)):S1. doi: 10.1186/cc4927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Baron E.J., Miller J.M., Weinstein M.P. A guide to utilization of the microbiology laboratory for diagnosis of infectious diseases: 2013 recommendations by the Infectious Diseases Society of America (IDSA) and the American Society for Microbiology (ASM)(a) Clin Infect Dis. 2013;57(4):e22–e121. doi: 10.1093/cid/cit278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Waites K.B., Talkington D.F. Mycoplasma pneumoniae and its role as a human pathogen. Clin Microbiol Rev. 2004;17(4):697–728. doi: 10.1128/CMR.17.4.697-728.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kumar S., Hammerschlag M.R. Acute respiratory infection due to Chlamydia pneumoniae: current status of diagnostic methods. Clin Infect Dis. 2007;44(4):568–576. doi: 10.1086/511076. [DOI] [PubMed] [Google Scholar]
- 7.Mercante J.W., Winchell J.M. Current and emerging Legionella diagnostics for laboratory and outbreak investigations. Clin Microbiol Rev. 2015;28(1):95–133. doi: 10.1128/CMR.00029-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Jain S., Williams D.J., Arnold S.R. Community-acquired pneumonia requiring hospitalization among U.S. children. N Engl J Med. 2015;372(9):835–845. doi: 10.1056/NEJMoa1405870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Goldsmith C.S., Miller S.E. Modern uses of electron microscopy for detection of viruses. Clin Microbiol Rev. 2009;22(4):552–563. doi: 10.1128/Cmr.00027-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Mandell L.A., Wunderink R.G., Anzueto A. Infectious Diseases Society of America/American Thoracic Society consensus guidelines on the management of community-acquired pneumonia in adults. Clin Infect Dis. 2007;44(suppl 2):S27–S72. doi: 10.1086/511159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Curry A., Appleton H., Dowsett B. Application of transmission electron microscopy to the clinical study of viral and bacterial infections: present and future. Micron. 2006;37(2):91–106. doi: 10.1016/j.micron.2005.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Goldsmith C.S., Ksiazek T.G., Rollin P.E. Cell culture and electron microscopy for identifying viruses in diseases of unknown cause. Emerg Infect Dis. 2013;19(6):886–891. doi: 10.3201/eid1906.130173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hazelton P.R., Gelderblom H.R. Electron microscopy for rapid diagnosis of infectious agents in emergent situations. Emerg Infect Dis. 2003;9(3):294–303. doi: 10.3201/eid0903.020327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.De Schutter I., De Wachter E., Crokaert F. Microbiology of bronchoalveolar lavage fluid in children with acute nonresponding or recurrent community-acquired pneumonia: identification of nontypeable Haemophilus influenzae as a major pathogen. Clin Infect Dis. 2011;52(12):1437–1444. doi: 10.1093/cid/cir235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Salih W., Schembri S., Chalmers J.D. Simplification of the IDSA/ATS criteria for severe CAP using meta-analysis and observational data. Eur Respir J. 2014;43(3):842–851. doi: 10.1183/09031936.00089513. [DOI] [PubMed] [Google Scholar]
- 16.Bossuyt P.M., Reitsma J.B., Bruns D.E. STARD 2015: an updated list of essential items for reporting diagnostic accuracy studies. BMJ. 2015;351:h5527. doi: 10.1136/bmj.h5527. [Epub October 28, 2015] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lim W.S., Baudouin S.V., George R.C. BTS guidelines for the management of community acquired pneumonia in adults: update 2009. Thorax. 2009;64(suppl 3):iii1–55. doi: 10.1136/thx.2009.121434. [DOI] [PubMed] [Google Scholar]
- 18.Woodhead M., Blasi F., Ewig S. Guidelines for the management of adult lower respiratory tract infections—full version. Clin Microbiol Infect. 2011;17(Suppl 6):E1–59. doi: 10.1111/j.1469-0691.2011.03672.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wang X., Li Z.Y., Zeng L. Neutrophil CD64 expression as a diagnostic marker for sepsis in adult patients: a meta-analysis. Crit Care. 2015;19:245. doi: 10.1186/s13054-015-0972-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Hoffmann J., Machado D., Terrier O. Viral and bacterial co-infection in severe pneumonia triggers innate immune responses and specifically enhances IP-10: a translational study. Sci Rep. 2016;6:38532. doi: 10.1038/srep38532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Cawcutt K.A., Kalil A.C. Viral and bacterial co-infection in pneumonia: do we know enough to improve clinical care? Crit Care. 2017;21(1):19. doi: 10.1186/s13054-016-1592-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lovejoy T., Rez P., Dellby N. Ondrej Krivanek: A pioneering visionary in electron microscopy. Ultramicroscopy. 2017 doi: 10.1016/j.ultramic.2017.02.003. [DOI] [PubMed] [Google Scholar]
- 23.Cyrklaff M., Frischknecht F., Kudryashev M. Functional insights into pathogen biology from 3D electron microscopy. FEMS Microbiol Rev. 2017 doi: 10.1093/femsre/fux041. [DOI] [PubMed] [Google Scholar]
- 24.de Boer P., Hoogenboom J.P., Giepmans B.N. Correlated light and electron microscopy: ultrastructure lights up! Nat Methods. 2015;12(6):503–513. doi: 10.1038/nmeth.3400. [DOI] [PubMed] [Google Scholar]
- 25.Erokhina T.N., Lazareva E.A., Richert-Poggeler K.R. Subcellular localization and detection of tobacco mosaic virus ORF6 protein by immunoelectron microscopy. Biochemistry. 2017;82(1):60–66. doi: 10.1134/S0006297917010060. [DOI] [PubMed] [Google Scholar]
- 26.Beniac D.R., Siemens C.G., Wright C.J. A filtration based technique for simultaneous SEM and TEM sample preparation for the rapid detection of pathogens. Viruses. 2014;6(9):3458–3471. doi: 10.3390/v6093458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Beniac D.R., Hiebert S.L., Siemens C.G. A mobile biosafety microanalysis system for infectious agents. Sci Rep. 2015;5:9505. doi: 10.1038/srep09505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Golding C.G., Lamboo L.L., Beniac D.R. The scanning electron microscope in microbiology and diagnosis of infectious disease. Sci Rep. 2016:6. doi: 10.1038/srep26516. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.