Abstract
Neisseria gonorrhoeae represents an urgent public health threat due to the rapid emergence of resistance to current antibiotics and the limited number of anti-gonococcal agents currently in clinical trials. This study utilized a drug repositioning strategy to investigate FDA-approved gold-containing drugs against N. gonorrhoeae. Auranofin, sodium aurothiomalate and aurothioglucose inhibited 48 clinical isolates of N. gonorrhoeae including multidrug-resistant strains at a concentration as low as 0.03 µg/mL. A time-kill assay revealed that auranofin exhibited rapid bactericidal activity against N. gonorrhoeae. Moreover, both sodium aurothiomalate and aurothioglucose did not inhibit growth of vaginal protective commensal lactobacilli. Auranofin, in combination with azithromycin, ceftriaxone, cefixime or tetracycline showed an additive effect against four N. gonorrhoeae strains, suggesting the possibility of using auranofin in dual therapy. Moreover, auranofin reduced the burden of intracellular N. gonorrhoeae by over 99% outperforming the drug of choice ceftriaxone. Auranofin was found superior to ceftriaxone in reducing the secretion of the pro-inflammatory cytokine IL-8 by endocervical cells infected with N. gonorrhoeae. Furthermore, auranofin exhibited a prolonged post-antibiotic effect over 10 h, as well as inability to generate resistant mutants. Overall, the current study suggests that repurposing gold-containing drugs, like auranofin, for treatment of gonorrhea warrants further investigation.
Subject terms: Drug discovery, Microbiology, Medical research
Introduction
Neisseria gonorrhoeae infects both the human male and female reproductive tracts causing the sexually transmitted infection gonorrhea. Gonorrhea is the second most common notifiable disease in the United States of America, according to the Centers for Disease Control and Prevention (CDC)1,2. The CDC estimates the number of new gonorrhea cases in the U.S. alone will exceed 820,000 cases annually, and this number is expected to increase due to extensive drug resistance1. Globally, the World Health Organization estimates 106 million new cases of gonorrhea will occur each year2. It has been reported that about 80% of N. gonorrhoeae cervical infections are mostly asymptomatic and unnoticed3,4. Without proper treatment, these infections can result in pelvic inflammation-associated damage to the ciliated epithelium5. This damage is permanent and can increase the risk of ectopic pregnancy and lead to tubal-factor infertility6.
With no available vaccines against N. gonorrhoeae, antibiotics are the only effective method to treat gonorrhea. Different countries have established different guidelines to treat gonorrhea, mostly comprised of dual therapy of a 1 g oral dose of azithromycin and an injectable 250 mg dose of intramuscular ceftriaxone for patients afflicted with gonorrhea7–10. However, increasing resistance to this dual therapy options and to the to the last available first-line treatments for gonorrhea11,12, increased the risk of untreatable gonorrhea becoming a widespread public health epidemic12,13. Thus, there is an urgent need for novel therapeutic strategies to treat gonorrhea.
In order to circumvent the cost and time associated with traditional de novo drug discovery, drug repositioning of FDA-approved drugs, particularly those that are off-patent, represents a promising approach to find new antibacterials14. Utilizing this strategy, we investigated auranofin as a potential novel antigonorrheal agent. Auranofin is an FDA-approved, gold-containing, drug used to treat rheumatoid arthritis. Previous studies on auranofin have reported that the drug possesses potent antibacterial activity against clinically-pertinent pathogens including methicillin-resistant Staphylococcus aureus (MRSA), vancomycin-resistant enterococci (VRE), and Clostridioides difficile15–20. However, no reports have investigated the effect of auranofin against N. gonorrhoeae. In this study, we investigated the three FDA approved compounds auranofin, aurothioglucose and sodium aurothiomalate, against a diverse panel of multidrug-resistant N. gonorrhoeae and against isolates of Lactobacillus that limit gonococcal colonization of the genitourinary tract. Additionally, the possibility of using auranofin in combination with antibiotics currently used to treat gonorrhea was explored. Furthermore, the gold compounds’ ability to reduce the burden of intracellular N. gonorrhoeae as well as its immunomodulatory effect were examined in an endocervical cell line infected with N. gonorrhoeae. Finally, we investigated the post-antibiotic effect of auranofin as well as its frequency of spontaneous resistance mutations.
Results
Susceptibility analysis of gold compounds against clinical isolates of N. gonorrhoeae
The anti-gonococcal activity of auranofin, sodium aurothiomalate and aurothioglucose against a panel of N. gonorrhoeae clinical isolates were determined using the broth microdilution assay. In addition, 5 WHO reference strains with well-characterized antibiogram, phenotypic and genetic markers21 were included to validate the results. As presented in Supplementary Table S2 the activity of control antibiotics tested via broth microdilution is almost identical to the values established by the WHO. The gold drugs were equipotent to or superior to azithromycin against the 5 reference strains as well as 48 other N. gonorrhoeae clinical isolates. As presented in Supplementary Table S3, auranofin inhibited growth of all N. gonorrhoeae strains tested at concentrations ranging from 0.007 µg/mL up to 0.125 µg/mL with MIC50 and MIC90 values of 0.06 µg/mL and 0.125 µg/mL, respectively (Table 1). Sodium aurothiomalate and aurothioglucose were slightly less potent compared to auranofin with MIC50 values of 0.25 µg/mL and 0.5 µg/mL respectively, and MIC90 values of 1 µg/mL and 8 µg/mL respectively (Table 1). Azithromycin’s MIC values ranged from 0.25 to 1 µg/mL against N. gonorrhoeae strains sensitive to this antibiotic while against azithromycin-resistant isolates (strains 167, 175, 179, 181, 197), the MIC of azithromycin ranged from 8 to 256 µg/mL(Supplementary Table S2). All three gold drugs maintained their effectiveness against N. gonorrhoeae clinical isolates exhibiting resistance to standard antibiotics used for treatment of gonorrhea such as N. gonorrhoeae strain 181 and N. gonorrhoeae strain 175, which are azithromycin-resistant strains. A group of 5 N. gonorrhoeae strains (167, 175, 179, 181, and 194) has been chosen for its high resistance profile to first line treatment for a confirmatory agar dilution according to the CLSI recommended agar dilution assay. There was no change observed in the MIC values obtained from either method. As depicted (Table 2), the MIC values of gold compounds and control antibiotics remained constant.
Table 1.
The minimum inhibitory concentration 50 and 90 (MIC in µg/mL) of gold drugs (auranofin, sodium aurothiomalate and aurothioglucose) and control antibiotics azithromycin, and ceftriaxone against 48 clinical isolates of Neisseria gonorrhoeae. MIC50 and MIC90 are the minimum inhibitory concentrations needed to inhibit 50% and 90% of the strains.
| Strain Name | Auranofin | Aurothiomalate | Aurothioglucose | Azithromycin | Ceftriaxone |
|---|---|---|---|---|---|
| MIC50 | 0.06 | 0.25 | 0.5 | 1 | 0.03 |
| MIC90 | 0.125 | 1 | 8 | 4 | 0.06 |
Table 2.
The minimum inhibitory concentration (MIC in µg/mL) of gold drugs (auranofin, sodium aurothiomalate and aurothioglucose) and control antibiotic azithromycin against four strains of N. gonorrhoeae (167, 175, 179, 181, and 197) via agar dilution.
| Strain | Auranofin | Aurothiomalate | Aurothioglucose | Azithromycin |
|---|---|---|---|---|
| N. gonorrhoeae 167 | 0.125 | 0.5 | 8 | 16 |
| N. gonorrhoeae 175 | 0.125 | 1 | 2 | 0.5 |
| N. gonorrhoeae 179 | 0.06 | 0.125 | 0.25 | 4 |
| N. gonorrhoeae 181 | 0.125 | 1 | 0.5 | 265 |
| N. gonorrhoeae 197 | 0.06 | 0.5 | 0.5 | 8 |
Evaluation of the gold drugs’ antibacterial activity against N. gonorrhoeae via a time-kill assay
After confirming all three gold-containing drugs possessed potent antibacterial activity against N. gonorrhoeae, we examined whether the three agents exhibit bacteriostatic or bactericidal activity. Auranofin, sodium aurothiomalate, aurothioglucose and azithromycin (all tested at 3 × MIC) were evaluated against N. gonorrhoeae strain 194 via a standard time-kill assay. As presented in Fig. 1, auranofin exhibited rapid bactericidal activity and completely eradicated the high bacterial inoculum within four hours. Azithromycin required eight hours to achieve the same effect. No reduction in bacterial inoculum was observed in the presence of both sodium aurothiomalate and aurothioglucose, at 3 × MIC, over 24 hours indicating these agents are bacteriostatic in vitro against N. gonorrhoeae.
Figure 1.
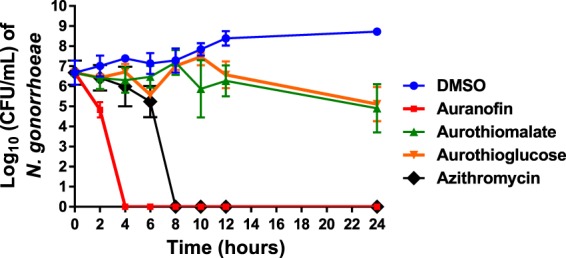
Time-kill analysis of gold compounds (at 3 × MIC) against N. gonorrhoeae strain 194 over a 24 hour incubation period at 37 °C. DMSO served as a negative control. Error bars represent standard deviation values. Lowest limit of detection is 100 CFU/mL.
Combination testing of auranofin with control antibiotics against N. gonorrhoeae
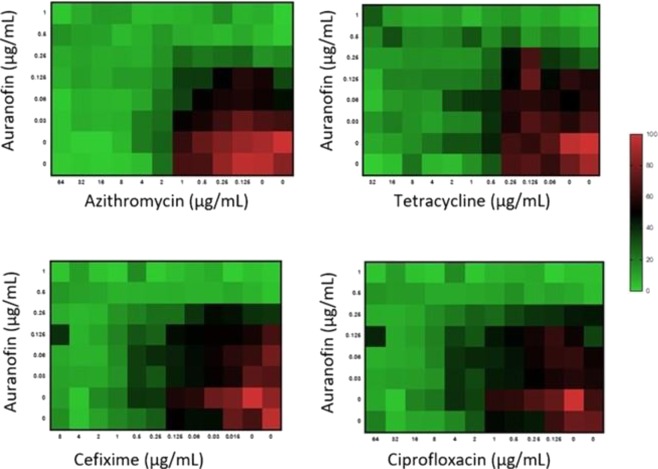
Antimicrobial resistance to gonorrhea treatment is continuously increasing; this necessitates new antibacterials and drug combinations be identified22,23. The current CDC guidelines for treatment of N. gonorrhoeae infections recommends dual therapy with two different antibiotics. The recommended treatment consists of a single dose of 250 mg of intramuscular ceftriaxone and 1 g of oral azithromycin in order to curb resistance forming to either agent. Given that auranofin exhibited the most potent in vitro antibacterial activity against N. gonorrhoeae, we investigated auranofin in combination with first-line antibiotics (azithromycin, ceftriaxone and cefixime) used to treat gonorrhea as well as tetracycline. Auranofin exhibited an additive relationship with all four drugs (fractional inhibitory concentration (FIC) index ranged from 1.2 to 2) against N. gonorrhoeae strain 194 (Fig. 2). This suggests that dual therapy using auranofin in combination with azithromycin, cefixime, ceftriaxone or tetracycline may be possible but requires further investigation.
Figure 2.
Checkerboard analysis of auranofin tested in combination with four different antibiotics against N. gonorrhoeae strain 194. Data presented as a heat map indicating additive relationship between auranofin, azithromycin, cefixime, ciprofloxacin or tetracycline (fractional inhibitory concentration index ranged from 1.25–2).
Selectivity of gold compounds to N. gonorrhoeae over commensal strains
N. gonorrhoeae pathogenesis greatly depends on colonization of the reproductive tract. One of the natural barriers of such colonization is an intact healthy microbiome. Species of lactobacilli present in the genitourinary tract play a significant role in hindering colonization by Neisseria. Thus, we explored the antibacterial activity of the gold compounds against eight commensal vaginal lactobacilli strains. As presented in Table 3, both sodium aurothiomalate and aurothioglucose were inactive against all eight strains of lactobacilli (MIC > 256 μg /mL), suggesting that they could be used against gonococci without disrupting the normal microbiota. Conversely, azithromycin, the drug of choice, inhibited the tested normal microbiota strains (MIC < 1 µg/mL). This was similar to the result obtained for auranofin (MIC values <1 µg/mL).
Table 3.
The minimum inhibitory concentration (MIC in µg/mL) of gold drugs (auranofin, sodium aurothiomalate and aurothioglucose) and control antibiotic azithromycin against eight different commensal Lactobacillus strains from the female reproductive tract.
| Strain | Auranofin | Aurothiomalate | Aurothioglucose | Azithromycin |
|---|---|---|---|---|
| L. crispatus HM638 | <1 | >128 | >128 | <1 |
| L. jensenii HM105 | <1 | >128 | >128 | <1 |
| L. gasseri HM-403 | <1 | >128 | >128 | <1 |
| L. johnsonii HM643 | <1 | >128 | >128 | <1 |
| L. gasseri HM642 | <1 | >128 | >128 | <1 |
| L. jensenii HM639 | <1 | >128 | >128 | <1 |
| L. jensenii HM640 | <1 | >128 | >128 | <1 |
| L. gasseri HM644 | 1 | >128 | >128 | <1 |
Intracellular infection clearance assay
N. gonorrhoeae has the ability to invade mucosal epithelia and survive intracellularly. An intracellular clearance assay was performed to determine if auranofin can clear intracellular N. gonorrhoeae. As depicted in Fig. 3 auranofin (at 6 × MIC) could successfully reduce the burden of intracellular N. gonorrhoeae infection of endocervical (END1/E6E7) by over 99% after 24 hours relative to the negative control. The control drug ceftriaxone could only achieve a non-significant reduction in intracellular bacterial counts. These results indicate that auranofin is able to effectively curb intracellular N. gonorrhoeae from infected endocervical cells.
Figure 3.
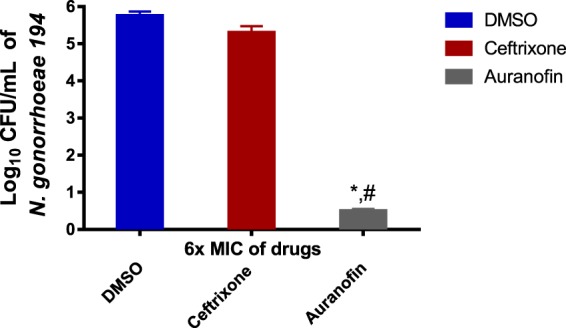
Effectiveness of auranofin and ceftriaxone (both at 6 × MIC) against intracellular N. gonorrhoeae in infected human endocervical cells (End1/E6E7). End1/E6E7 cells were infected with N. gonorrhea strain 194 for six hours and then treated with either auranofin or ceftriaxone for 24 hours. End1/E6E7 cells were subsequently lysed and intracellular bacterial CFU was determined. Error bars represent standard deviation values from triplicate samples used for each test agent. Auranofin was compared to both untreated cells (*) and cells treated with ceftriaxone (#). (P < 0.01 analyzed via unpaired t-test).
Auranofin reduces IL-8 production by endocervical cells infected with N. gonorrhoeae
Pro-inflammatory cytokines production is one of the hallmarks of N. gonorrhoeae infection. IL-8 is produced at the site of gonococcal infection and is elevated after the onset of symptoms and returns to baseline values after antibiotic therapy. Therefore, IL-8 is one of the inflammatory cytokines that mediates N. gonorrhoeae infection24. Auranofin is known to possess potent anti-inflammatory activity but its effect on IL-8 in endocervical cells infected with N. gonorrhoeae has not been previously reported. Thus we determined the amount of IL-8 produced from N. gonorrhoeae 194 infected End1/E6E7 cells, in the presence or absence of gold compounds and the control antibiotic ceftriaxone (at ½ × MIC). DMSO was used as a negative control to determine the baseline amount of IL-8 produced by infected endocervical cells. Auranofin was able to significantly reduce the level of IL-8 produced by infected endocervical cells by 28% relative to DMSO (Fig. 4). This was superior to ceftriaxone, which only achieved a 4.1% reduction. Though both sodium aurothiomalate and aurothioglucose reduced IL-8 production from infected endocervical cells by more than 10%, it was not statistically different compared to ceftriaxone treatment.
Figure 4.
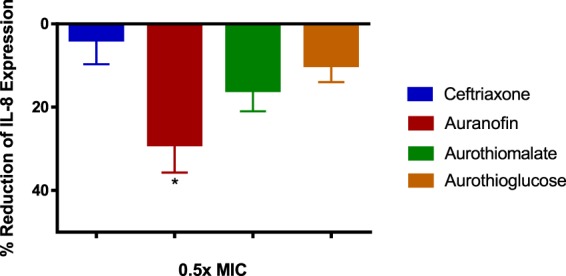
Evaluation of anti-inflammatory activity of gold drugs and ceftriaxone (all at 0.5 × MIC) in endocervical cells infected with N. gonorrhoeae strain 194. Auranofin and ceftriaxone reduced the level of IL-8 produced by End1/E6E7 cells 6 h post-infection. Error bars represent mean IL-8 measurement ± SD in three technical replicate wells from the same representative experiment. Significance indicated by (*) denotes a significant difference between cells treated with auranofin compared to ceftriaxone (P < 0.01 analyzed via One-way ANOVA).
Frequency of spontaneous mutation
Given the promising in vitro results of auranofin described above we next sought to investigate the likelihood that N. gonorrhoeae will develop resistance to auranofin using the single step resistance assay method19 and the results are reported in Supplementary Table S4. No resistant mutants were isolated at a concentration of 10 × MIC in the presence of a high inoculum, equal to 2.4 × 1010 CFU/mL, of N. gonorrhoeae strains 197, 202 or 206; indicating a resistance frequency < 2.4 × 10−10 for all tested strains which was lower than the drug of choice azithromycin. The positive control, rifmapicin’s frequency of spontaneous mutation was 1.8 ×10−6 which is equal to the value previously reported in other bacterial strains25,26.
Post-antibiotic effect
The ability of auranofin to exhibit a prolonged inhibitory effect against N. gonorrhoeae following a brief exposure period was investigated through analyzing its post-antibiotic effect (PAE). Following a 2-hour exposure to 10 × MIC of either auranofin or azithromycin, four strains of N. gonorrhoeae were allowed to grow for 12 hours, sampled and plated every 2 hours. The results of the experiment were analyzed and reported in Supplementary Table S5. Auranofin exhibited a long PAE against all tested strains as it inhibited their growth for 10 hours post exposure. This was superior to the drug of choice azithromycin which showed only PAE = 8 hours.
Discussion
The timeline of antibiotic development against N. gonorrhoeae has always been paralleled by rapid resistance acquisition by the bacterium. Since the development of sulfonamides in the 1930s, N. gonorrhoeae has acquired resistance to every first-line antibiotic class introduced27. More recently, the problem has worsened with the emergence of new bacterial strains exhibiting multidrug-resistant and extensively drug-resistant phenotypes. The emergence of untreatable strains of N. gonorrhoeae combined with the limited pipeline of novel antigonococcal agents underscores the critical need to discover new antibacterial agents28,29. Revisiting FDA-approved drugs for an alternative indication to treat gonorrhea can be a fast and effective strategy to provide new therapeutic options or identify molecules with weak antibacterial activity that can be modified to enhance their potency. Using a drug-repositioning approach, we identified auranofin and two gold thiol drugs, aurothioglucose and aurothiomalate, as potential candidates for investigation to treat drug-resistant gonorrhea.
The antibacterial activity of auranofin and its analogues was explored using broth microdilution method, which was herein validated using five N. gonorrhoeae strains from WHO used for quality assurance and quality control of gonococcal AMR testing. A panel of 48 N. gonorrhoeae clinical isolates was tested and to further confirm the method 5 strains with different resistance profile to control antibiotics were selected for a run via agar dilution yielded the same results. Auranofin and its two analogues successfully inhibited the growth of all the tested isolates of N. gonorrhoeae. Auranofin was the most potent drug against N. gonorrhoeae with MIC values ranging from 0.03 to 0.25 µg/mL. Aurothiomalate’s MIC values ranged from 0.06 to 32 µg/mL while aurothioglucose’s MIC values ranged from 0.125 to 16 µg/mL. In a time-kill assay, auranofin (at 3 × MIC) showed rapid bactericidal activity against N. gonorrhoeae, successfully reducing the bacterial count below the limit of detection (250 CFU/mL) after only four hours. Being the most potent of the tested gold compounds, auranofin was chosen for further analysis. Moreover, to address the problem of rapid emergence of resistance of N. gonorrhoeae to auranofin we examined the possibility of isolating auranofin resistant mutants after carrying out a single step resistance assay. At a high inoculum size (~1010 CFU/mL), no mutants were obtained indicating that spontaneous resistance mutations of N. gonorrhoeae against auranofin did not occur at a high inoculum, on the other hand resistance was developed against rifampicin which is in agreement with a previous report in S. aureus19. However, other long term resistance studies should be performed to further investigate the potential of resistance development against auranofin.
Pharmacodynamic parameters such as the post antibiotic effect are increasingly applied to help shape a dosing regimen (size and frequency of deses given to a patient) for antibiotics. The post-antibiotic effect is defined as the period through which bacterial growth will be suppressed after a brief exposure to an antibiotic30. The post-antibiotic effect analysis performed provides valuable evidence that auranofin would need fewer doses if used to treat N. gonorrhoeae infections. As auranofin exhibited a long PAE (=10 hours) against N. gonorrhoeae, this indicates a very slow bacterial recovery after exposure and hence patients would need to be subjected to fewer doses, which is advantageous in terms of limited toxicity to host tissues, reduced costs, and more patient compliance to the prescribed regimen31.
The presence of a healthy microbiome in the female reproductive tract can aid in reducing susceptibility to sexually transmitted diseases32. In particular, vaginal species of lactobacilli in women can reduce the risk of contracting infections from G. vaginalis and N. gonorrhoeae33. In addition, women whose vaginal microbiota are most dominated by lactobacilli species have a lower incidence of N. gonorrhoeae infection34,35. Previous studies have shown that dysbiosis contributes to gonococcal infections. However, first-line gonorrhoeae treatments unfortunately inhibit growth of important bacterial species that comprise the vaginal microbiome and hinder colonization by gonococci. Finding alternative therapeutics that selectively target gonococci without inhibiting important commensal species such as lactobacilli would be desirable. We thus investigated the antibacterial activity of the gold-containing drugs against important lactobacilli species that comprise the vaginal microbiome. Aurothioglucose and sodium aurothiomalate exhibited high selectivity towards N. gonorrhoeae while not inhibiting growth of eight different commensal lactobacilli strains. In contrast, auranofin exhibited similar potency against species of lactobacilli and N. gonorrhoeae, which was similar to azithromycin.
N. gonorrhoeae can invade the female genital tract epithelial cells and cross the epithelial barrier into the sub-epithelial space36. N. gonorrhoeae has been previously shown to be able to survive inside host cells as well as to pass epithelial cell layers, which is a key step in disseminated infections. Auranofin was able to clear N. gonorrhoeae infection in vitro, thus we investigated its ability to clear intracellular bacteria present in infected endocervical cells. After 24 hours, at 6 × MIC, auranofin was superior to the drug of choice ceftriaxone in reducing the burden of intracellular bacteria inside infected END1/E6E7 endocervical cells. Auranofin was able to achieve a 5.0 −log10 reduction in N. gonorrhoeae inside infected endocervical cells while ceftriaxone, on the contrary, was not significantly effective. To rule out the possibility of cell death leading to a false positive result, auranofin was assayed for its in vitro cytotoxicity against End1/E6E7 endocervical cells for 24 hours via MTS cytotoxicity assay37. As depicted in Supplementary Fig. S6, cells viability was around 78% and 85% at 8× and 4× MIC respectively. Collectively, these results indicate that auranofin can significantly clear intracellular N. gonorrhoeae at a rate outperforming the drug of choice ceftriaxone.
Successful colonization by N. gonorrhea of the female urogenital tract is accompanied by a severe inflammatory response that results in production of numerous pro-inflammatory cytokines38. Alleviating such inflammation is a potential approach to reduce the infection’s severity. It has been previously reported that gold drugs such as auranofin have potent anti-inflammatory activity and can reduce the expression of pro-inflammatory cytokines such as tumor necrosis factor-α, interleukin-6, interleukin-1 β, and monocyte chemoattractant protein-118. Based upon this finding, we hypothesized the gold compounds investigated in this report would reduce the production of pro-inflammatory cytokines by endocervical cells infected with N. gonorrhoeae. In this regard, auranofin was able to significantly reduce IL-8 expression of pre-infected human endocervical cells. This effect was superior to the effect of the drug of choice ceftriaxone. The anti-inflammatory effect of auranofin could be advantageous to curbing damage of the epithelial tissue present in the urogenital tract in patients afflicted with gonorrhea.
It is important to mention that the data of the clinical trial of auranofin for treatment of rheumatoid arthritis which included over 5000 patients administered (6 mg/day) revealed no signs of accumulated toxicity and good tolerability39. In a recent clinical trial of auranofin, through a maximal daily dose of 21 mg over 14 days regimen, trough plasma concentrations of auranofin ranging between 0.8–1.5 mg/L after the 14th day were measured with a half-life time of 35 days40. These pharmacokinetic properties will assure a higher concentration of auranofin than the MICs covered in this study. Moreover, the treatment course of rheumatoid arthritis can extend to over 5 years. Such treatment plan is much longer than the course of treatment usually prescribed for antibiotics which ranges from one to two weeks29. The toxicity of auranofin over HaCaT cells was evaluated to be 6.38 ± 0.29 μg/mL18. This value is 100 times greater than the MIC50 value of auranofin against N. gonorrhoeae.
To conclude, we report that three FDA-approved gold drugs, auranofin, aurothiomalate and aurothioglucose have potent in vitro antibacterial activity against N. gonorrhoeae. Aurothiomalate and aurothioglucose possess an advantage over current antibiotics used to treat gonorrhea in that both gold-containing drugs do not inhibit growth of commensal Lactobacillus spp. that provide a protective barrier to gonococcal colonization. Auranofin was found to possess advantageous properties than drugs of choice such as having an additive relationship with antibiotics used to treat gonorrhea, which may open the door for additional dual therapy treatment options for gonorrhea, a very low frequency of spontaneous mutation which suggests a low probability to develop resistance and a highly prolonged post-antibiotic effect that suggests smaller and less frequent doses of the drug if used to treat gonorrhea. Finally, auranofin was found to reduce the burden of intracellular N. gonorrhoeae infecting endocervical cells as well as modulate the production of the pro-inflammatory cytokine IL-8, which can be advantageous in reducing inflammation, one of the hallmarks of gonococcal infection. A future aim is to validate the in vitro results presented in this study in vivo via a rodent model of N. gonorrhoeae infection.
Materials and Methods
Bacterial strains, chemicals and media
The N. gonorrhoeae reference strains were obtained from the WHO reference strain panel for global quality assurance and quality control of gonococcal AMR testing21,41. The rest of N. gonorrhoeae strains used were clinical isolates obtained from the CDC (Supplementary Table 1). The commensal vaginal bacterial strains were obtained from the Biodefense and Emerging Infections Research Resources Repository (BEI Resources) (Supplementary Table 2). All chemical compounds used in this study were obtained commercially from chemical vendors: auranofin (Chem-Impex International, Wood Dale, IL), sodium aurothiomalate, tetracycline and ciprofloxacin (Sigma-Aldrich, St. Louis, MO), aurothioglucose (USP reference standard Rockville, MD), ceftriaxone and cefixime (Acros Organics, NJ), and azithromycin (TCI America, Portland, OR).
Difco lactobacilli MRS broth, Brucella base broth and chocolate II agar (GC II Agar with hemoglobin and IsoVitaleX) were purchased from Becton, Dickinson and Company (Cockeysville, MD). Yeast extract and dextrose (Fisher Bioreagents, Fairlawn, NJ), proteose-peptone, nicotinamide adenine dinucleotide (NAD) and agarose (Sigma-Aldrich, St. Louis, MO), hematin solution, Tween 80, and pyridoxal (Chem-Impex International, Wood Dale, IL) were purchased from chemical vendors.
Antibacterial susceptibility analysis for gold drugs and control antibiotics
The minimum inhibitory concentration (MIC) of gold drugs (auranofin, sodium aurothiomalate, and aurothioglucose) and azithromycin was determined using the broth microdilution assay, as previously described42. Briefly, a 1.0 McFarland standard was prepared and diluted in Brucella Supplemented broth (BSB) (Brucella broth supplemented with yeast extract, dextrose, agarose, proteose-peptone, NAD, pyrodixal and hematin) to reach a bacterial count of 5 × 105 CFU/mL. Drugs were added and serially diluted in broth. Plates were then incubated for 24 hours at 37 °C in the presence of 5% CO2. In order to confirm the MIC values obtained by broth microdilution a confirmatory test was carried out on five N. gonorrhoeae strains (167, 175, 179, 181, 197), which have different resistance profile to azithromycin, ciprofloxacin and ceftriaxone, according to the CLSI recommended agar dilution as described43.
Time-kill kinetics of gold compounds and azithromycin against N. gonorrhoeae
The killing kinetics of the gold drugs was investigated following the method described in a previous study44. In brief, an overnight culture of N. gonorrhoeae strain 194 was diluted in fresh BSB and incubated until the inoculum was ~5 × 106 CFU/mL. The bacterial solution was subsequently exposed to 3 × MIC of the tested drugs (in triplicates). DMSO (Solvent for drugs) served as a negative control and azithromycin was used as a positive control. Aliquots were collected after 0, 2, 4, 6, 8, 10, 12, and 24 hours and plated onto GC II agar supplemented with hemoglobin and 1% IsoVitaleX. Plates were incubated at 37 °C with 5% CO2 for 24 hours prior to counting colonies present on the plate. The experiment was repeated 3 times with lowest limit of detection was 250 CFU/mL.
Vaginal microbiome sensitivity to gold drugs
The selectivity of the three gold-containing drugs to pathogenic gonococci over bacterial species that comprise the vaginal commensal flora of the microbiome was assessed by determining the drugs’ MIC against Lactobacillus gasseri, L. jensenii, L. rhamnosus, and L. crispatus. Lactobacilli were streaked on de Man, Rogosa, Sharpe (MRS) agar and incubated for 48 hours at 37 °C with 5% CO2. The broth microdilution assay method was utilized to determine the MIC of the gold drugs and azithromycin following the guidelines outlined by the Clinical and Laboratory Standards Institute43,45.
Synergistic activity of auranofin with azithromycin, cefixime, ciprofloxacin and tetracycline
The ability of auranofin to work in combination with conventional antibiotics used in the treatment of gonorrhea was evaluated as previously described46,47. In brief, a bacterial suspension of N. gonorrhoeae strain 194 equivalent to 1.0 McFarland standard was prepared and diluted in BSB to achieve a bacterial inoculum of 5 × 105 CFU/mL. Subsequently, auranofin and control drugs were added at different concentrations along with bacteria containing media. The plates were incubated for 24 hours at 37 °C in the presence of 5% CO2. Next, the optical density was measured at 600 nm, using Spectra Max i3 (Molecular Devices, LLC, San Jose, CA) spectrophotometer, to calculate the percentage growth of bacteria. Results were plotted as a heat map and fractional inhibitory concentrations indices (FIC) were calculated. In this analysis, interactions with calculated FIC that were ≤0.5 were categorized as synergistic. An FICI of >0.5 but ≤1.25 was categorized as additive. An FICI > 1.25 but ≤4 was considered as indifference, while an FICI > 4 was categorized as antagonistic16,17.
Intracellular clearance assay
In order to investigate the ability of auranofin to enter human endocervical cells and reduce the burden of intracellular N. gonorrhoeae an intracellular bacterial clearance assay was utilized as previously described42. Briefly, a cell density of ~1 ×105 per well of human endocervical cells (ATCC CRL-2615, End1/E6E7) were seeded in 96-well tissue culture and then infected with N. gonorrhoeae strain194 at a multiplicity of infection of 100:1 for six hours at 37 °C and 5% CO2. Cells were then washed with 320 μg/mL gentamicin containing PBS to remove and wash-off non-phagocytized bacteria and then incubated with 32 μg/mL gentamicin containing PBS for 4 hours. Either auranofin or the control drug ceftriaxone were added subsequently to the cells at 6 × MIC and incubated for 24 hours at 37 °C with 5% CO2. The cells were lysed the following day and the plated onto Chocolate II Agar plates. Experiments were performed using triplicate samples for each treatment group and the experiment was repeated at least twice. Data were analyzed using an unpaired t-test using GraphPad Prism 6.00 (GraphPad Software, La Jolla, CA).
Anti-inflammatory activity of gold-containing drugs on IL-8 expression by infected endocervical cells
To investigate the anti-inflammatory activity of the gold compounds, IL-8, a pro-inflammatory cytokine, was detected in the supernatants of N. gonorrhoeae infected End1/E6E7 – human endocervix cells (ATCC CRL-2615) exposed to either the gold-containing drugs or ceftriaxone, as previously described42,48. In brief, N. gonorrhoeae strain 194 was allowed to infect the endocervical cells for two hours at 37°C with 5% CO2 before initiating treatment with 0.5 × MIC of ceftriaxone, auranofin, sodium aurothiomalate, or aurothioglucose (tested in triplicates) and left for four hours. DMSO served as a negative control. Supernatants of cells were collected and tested for IL-8 concentration using the Human IL-8 ELISA Kit (Human IL-8 PicoKine ELISA Kit, Boster Biological Technology, Pleasanton, CA) following the manufacturer’s protocol. Data were analyzed using a one-way ANOVA with post-hoc Dunnett’s multiple comparisons test using GraphPad Prism 6.0 (GraphPad Software, La Jolla, CA).
N. gonorrhoeae frequency of spontaneous mutation
Comprising a wide range of antibacterial susceptibility to control antibiotics, N. gonorrhoeae strains 197, 202 and 206 were chosen to be further investigated to determine the frequency of mutation. The bacteria were tested against both auranofin and rifampicin at 10× the MIC determined via agar dilution assay following the procedure that was previously described in19. Rifampicin was used as a positive control as it is known to be susceptible to spontaneous mutations49,50. Briefly, to prepare the media containing the drugs, GC agar base was autoclaved for 30 mins then human blood was added after cooling down to a final concentration of 5%. Either auranofin, or rifampin were then added to achieve a final concentration of 10 × MIC inside the media which was then poured in plates and left to dry out. A high inoculum ~1.0 × 1010CFU/mL of each N. gonorrhoeae strain tested was plated and incubated for 48 hours. Plates were checked after 48 hours to count grown colonies.
Post-antibiotic effect of auranofin
For PAE testing the assay was carried out on four N. gonorrhoeae strains with various resistance profiles (strains 181,186,194,198) following the procedure previously described51. Briefly, 10 × MIC of either auranofin or control antibiotic azithromycin were added to tubes containing 2 mL of BSB inoculated with 1.0 × 106 CFU/mL from each strain. Tubes were incubated in shaking incubator at 37 °C for one-hour exposure period after which cultures were diluted 1:1000 with pre-warmed BSB to remove the antibiotic. The tubes were then returned back to same incubation conditions. The tubes were routinely sampled and plated on chocolate agar plates before exposure and immediately after dilution every 2 hours for 12 hours. The PAE was calculated as follows: PAE = T − C, where T is the time taken by the viable counts of an antibiotic exposed culture to increase by 1 log10 unit above the count observed right after dilution, while C is the corresponding time for the growth of control.
Supplementary information
Acknowledgements
We would like to thank Marwa Alhashimi and Young Jin Seong for their kind help in this work. We are also gratefully for Dr. Haroon Mohammad for editing the manuscript. This work was supported by the National Institutes of Health (Grant No. R01AI130186).
Author contributions
M.N.S. and A.E. designed the study. A.E. performed experiments. A.E. and M.N.S. analyzed data and wrote the manuscript. All authors discussed the results and commented on the manuscript.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
is available for this paper at 10.1038/s41598-020-62696-3.
References
- 1.Control, C. f. D. & Prevention. Antibiotic resistance threats in the United States, 2013. (Centres for Disease Control and Prevention, US Department of Health and …, 2013).
- 2.World Health Organization. Emergence of multi-drug resistant Neisseria gonorrhoeae: Threat of global rise in untreatable sexually transmitted infections, 2011).
- 3.Edwards JL, Apicella MA. The molecular mechanisms used by Neisseria gonorrhoeae to initiate infection differ between men and women. Clinical microbiology reviews. 2004;17:965–981. doi: 10.1128/CMR.17.4.965-981.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Walker CK, Sweet RL. Gonorrhea infection in women: prevalence, effects, screening, and management. Int j. Womens Health. 2011;3:197. doi: 10.2147/IJWH.S13427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Mitchell C, Prabhu M. Pelvic inflammatory disease: current concepts in pathogenesis, diagnosis and treatment. Infectious Disease Clinics. 2013;27:793–809. doi: 10.1016/j.idc.2013.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Little JW. Gonorrhea: update. Oral Surgery, Oral Medicine, Oral Pathology, Oral Radiology, and Endodontology. 2006;101:137–143. doi: 10.1016/j.tripleo.2005.05.077. [DOI] [PubMed] [Google Scholar]
- 7.Bignell C, Unemo M, Board ESGE. 2012 European guideline on the diagnosis and treatment of gonorrhoea in adults. International journal of STD & AIDS. 2013;24:85–92. doi: 10.1177/0956462412472837. [DOI] [PubMed] [Google Scholar]
- 8.Bignell C, FitzGerald M. UK national guideline for the management of gonorrhoea in adults, 2011. International journal of STD & AIDS. 2011;22:541–547. doi: 10.1258/ijsa.2011.011267. [DOI] [PubMed] [Google Scholar]
- 9.(WHO), W. H. O. Guidelines for the Treatment of Neisseria gonorrhoeae, http://www.who.int/reproductivehealth/publications/rtis/gonorrhoea-treatment-guidelines/en/. [PubMed]
- 10.Alliance, A. S. H. Australian STI Management Guidelines for Use in Primary Care., http://www.sti.guidelines.org.au/sexually-transmissible-infections/gonorrhoea.
- 11.England, P. H. UK case of Neisseria gonorrhoeae with high-level resistance to azithromycin and resistance to ceftriaxone acquired abroad. Health Protection Report12 (2018).
- 12.Fifer H, et al. Failure of dual antimicrobial therapy in treatment of gonorrhea. New England Journal of Medicine. 2016;374:2504–2506. doi: 10.1056/NEJMc1512757. [DOI] [PubMed] [Google Scholar]
- 13.Bolan GA, Sparling PF, Wasserheit JN. The emerging threat of untreatable gonococcal infection. The New England journal of medicine. 2012;366:485. doi: 10.1056/NEJMp1112456. [DOI] [PubMed] [Google Scholar]
- 14.Schäberle TF, Hack IM. Overcoming the current deadlock in antibiotic research. Trends in microbiology. 2014;22:165–167. doi: 10.1016/j.tim.2013.12.007. [DOI] [PubMed] [Google Scholar]
- 15.AbdelKhalek A, Abutaleb NS, Elmagarmid KA, Seleem MN. Repurposing auranofin as an intestinal decolonizing agent for vancomycin-resistant enterococci. Scientific reports. 2018;8:8353. doi: 10.1038/s41598-018-26674-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Thangamani S, et al. Repurposing approach identifies auranofin with broad spectrum antifungal activity that targets Mia40-Erv1 pathway. Frontiers in cellular and infection microbiology. 2017;7:4. doi: 10.3389/fcimb.2017.00004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Parsonage D, et al. X-ray structures of thioredoxin and thioredoxin reductase from Entamoeba histolytica and prevailing hypothesis of the mechanism of Auranofin action. Journal of structural biology. 2016;194:180–190. doi: 10.1016/j.jsb.2016.02.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Thangamani S, Mohammad H, Abushahba MF, Sobreira TJ, Seleem MN. Repurposing auranofin for the treatment of cutaneous staphylococcal infections. International journal of antimicrobial agents. 2016;47:195–201. doi: 10.1016/j.ijantimicag.2015.12.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Thangamani S, et al. Antibacterial activity and mechanism of action of auranofin against multi-drug resistant bacterial pathogens. Sci. Rep. 2016;6:22571. doi: 10.1038/srep22571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.AbdelKhalek A, Abutaleb NS, Mohammad H, Seleem MN. Antibacterial and antivirulence activities of auranofin against Clostridium difficile. International journal of antimicrobial agents. 2019;53:54–62. doi: 10.1016/j.ijantimicag.2018.09.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Unemo M, et al. The novel 2016 WHO Neisseria gonorrhoeae reference strains for global quality assurance of laboratory investigations: phenotypic, genetic and reference genome characterization. Journal of Antimicrobial Chemotherapy. 2016;71:3096–3108. doi: 10.1093/jac/dkw288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Workowski KA, Bolan GA. Sexually transmitted diseases treatment guidelines. 2015. MMWR. Recommendations and reports: Morbidity and mortality weekly report. Recommendations and reports. 2015;64:1. [PMC free article] [PubMed] [Google Scholar]
- 23.Workowski KA. Centers for Disease Control and Prevention sexually transmitted diseases treatment guidelines. Clinical Infectious Diseases. 2015;61:S759–S762. doi: 10.1093/cid/civ771. [DOI] [PubMed] [Google Scholar]
- 24.Ramsey KH, et al. Inflammatory Cytokines Produced in Response to Experimental Human Gonorrhea. J. Infect. Dis. 1995;172:186–191. doi: 10.1093/infdis/172.1.186. [DOI] [PubMed] [Google Scholar]
- 25.Björkholm, B. et al. Mutation frequency and biological cost of antibiotic resistance in Helicobacter pylori. 98, 14607–14612 (2001). [DOI] [PMC free article] [PubMed]
- 26.Binet, R., Maurelli, A. T. J. A. A. & chemotherapy. Frequency of development and associated physiological cost of azithromycin resistance in Chlamydia psittaci 6BC and C. trachomatis L2. 51, 4267–4275 (2007). [DOI] [PMC free article] [PubMed]
- 27.Quillin SJ, Seifert HS. Neisseria gonorrhoeae host adaptation and pathogenesis. Nature Reviews Microbiology. 2018;16:226. doi: 10.1038/nrmicro.2017.169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Singh S, Sivakumar R. Challenges and new discoveries in the treatment of leishmaniasis. Journal of infection and chemotherapy. 2004;10:307–315. doi: 10.1007/s10156-004-0348-9. [DOI] [PubMed] [Google Scholar]
- 29.Ventola CL. The antibiotic resistance crisis: part 1: causes and threats. Pharmacy and therapeutics. 2015;40:277. [PMC free article] [PubMed] [Google Scholar]
- 30.Mohammad H, et al. Antibacterial characterization of novel synthetic thiazole compounds against methicillin-resistant Staphylococcus pseudintermedius. PloS one. 2015;10:e0130385. doi: 10.1371/journal.pone.0130385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Levison ME. Pharmacodynamics of antimicrobial drugs. Infectious Disease. Clinics. 2004;18:451–465. doi: 10.1016/j.idc.2004.04.012. [DOI] [PubMed] [Google Scholar]
- 32.Ravel J, et al. Vaginal microbiome of reproductive-age women. Proceedings of the National Academy of Sciences. 2011;108:4680–4687. doi: 10.1073/pnas.1002611107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Breshears LM, Edwards VL, Ravel J, Peterson ML. Lactobacillus crispatus inhibits growth of Gardnerella vaginalis and Neisseria gonorrhoeae on a porcine vaginal mucosa model. BMC microbiology. 2015;15:276. doi: 10.1186/s12866-015-0608-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Amant DCS, Valentin-Bon IE, Jerse AE. Inhibition of Neisseria gonorrhoeae by Lactobacillus species that are commonly isolated from the female genital tract. Infection and immunity. 2002;70:7169–7171. doi: 10.1128/IAI.70.12.7169-7171.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Saigh JH, Sanders CC, Sanders W. Inhibition of Neisseria gonorrhoeae by aerobic and facultatively anaerobic components of the endocervical flora: evidence for a protective effect against infection. Infection and immunity. 1978;19:704–710. doi: 10.1128/IAI.19.2.704-710.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Lu P, et al. A subpopulation of intracellular Neisseria gonorrhoeae escapes autophagy-mediated killing inside epithelial cells. The Journal of infectious diseases. 2018;219:133–144. doi: 10.1093/infdis/jiy237. [DOI] [PubMed] [Google Scholar]
- 37.Elsebaei MM, et al. Alkynyl-containing phenylthiazoles: Systemically active antibacterial agents effective against methicillin-resistant Staphylococcus aureus (MRSA) European journal of medicinal chemistry. 2018;148:195–209. doi: 10.1016/j.ejmech.2018.02.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Spurbeck RR, Arvidson CG. Lactobacilli at the front line of defense against vaginally acquired infections. Future microbiology. 2011;6:567–582. doi: 10.2217/fmb.11.36. [DOI] [PubMed] [Google Scholar]
- 39.Blodgett JR, Pietrusko R. Long-term efficacy and safety of auranofin: a review of clinical experience. Scandinavian journal of rheumatology. Supplement. 1986;63:67–78. [PubMed] [Google Scholar]
- 40.Capparelli EV, Bricker-Ford R, Rogers MJ, McKerrow JH, Reed SL. Phase I clinical trial results of auranofin, a novel antiparasitic agent. Antimicrobial agents and chemotherapy. 2017;61:e01947–01916. doi: 10.1128/AAC.01947-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Unemo M, Fasth O, Fredlund H, Limnios A, Tapsall J. Phenotypic and genetic characterization of the 2008 WHO Neisseria gonorrhoeae reference strain panel intended for global quality assurance and quality control of gonococcal antimicrobial resistance surveillance for public health purposes. Journal of Antimicrobial Chemotherapy. 2009;63:1142–1151. doi: 10.1093/jac/dkp098. [DOI] [PubMed] [Google Scholar]
- 42.Alhashimi, M., Mayhoub, A. & Seleem, M. N. Repurposing salicylamide for combating multidrug-resistant Neisseria gonorrhoeae. Antimicrobial Agents and Chemotherapy, AAC. 01225–01219 (2019). [DOI] [PMC free article] [PubMed]
- 43.CLSI. (9th ed. M07-A9, 2012).
- 44.Takei M, Yamaguchi Y, Fukuda H, Yasuda M, Deguchi T. Cultivation of Neisseria gonorrhoeae in liquid media and determination of its in vitro susceptibilities to quinolones. Journal of clinical microbiology. 2005;43:4321–4327. doi: 10.1128/JCM.43.9.4321-4327.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Kushiro A, et al. Antimicrobial susceptibility testing of lactic acid bacteria and bifidobacteria by broth microdilution method and Etest. International journal of food microbiology. 2009;132:54–58. doi: 10.1016/j.ijfoodmicro.2009.03.012. [DOI] [PubMed] [Google Scholar]
- 46.Meletiadis J, Pournaras S, Roilides E, Walsh TJ. Defining fractional inhibitory concentration index cutoffs for additive interactions based on self-drug additive combinations, Monte Carlo simulation analysis, and in vitro-in vivo correlation data for antifungal drug combinations against Aspergillus fumigatus. Antimicrobial agents and chemotherapy. 2010;54:602–609. doi: 10.1128/AAC.00999-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.King AM, et al. Aspergillomarasmine A overcomes metallo-β-lactamase antibiotic resistance. Nature. 2014;510:503. doi: 10.1038/nature13445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Lenz JD, Shirk KA, Jolicoeur A, Dillard JP. Selective inhibition of Neisseria gonorrhoeae by a dithiazoline in mixed infections with Lactobacillus gasseri. Antimicrobial agents and chemotherapy. 2018;62:e00826–00818. doi: 10.1128/AAC.00826-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Telenti A, et al. Detection of rifampicin-resistance mutations in Mycobacterium tuberculosis. The Lancet. 1993;341:647–651. doi: 10.1016/0140-6736(93)90417-F. [DOI] [PubMed] [Google Scholar]
- 50.Baltz RH. Spontaneous and induced mutations to rifampicin, streptomycin and spectinomycin resistances in actinomycetes: mutagenic mechanisms and applications for strain improvement. The Journal of antibiotics. 2014;67:619–624. doi: 10.1038/ja.2014.105. [DOI] [PubMed] [Google Scholar]
- 51.Pankuch G, Jacobs M, Appelbaum P. Postantibiotic effects of garenoxacin (BMS-284756) against 12 gram-positive or-negative organisms. Antimicrobial agents and chemotherapy. 2003;47:1140–1142. doi: 10.1128/AAC.47.3.1140-1142.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.