Abstract
Background. Health care workers continued to contract severe acute respiratory syndrome (SARS), even after barrier precautions were widely implemented.
Methods. We explored the possible contribution of contaminated hospital surfaces to SARS transmission by swabbing surfaces in 2 hospitals and testing the swab samples by reverse-transcriptase polymerase chain reaction (RT-PCR) and viral culture.
Results. Twenty-six of 94 swab samples tested positive for viral RNA. Swab samples of respiratory secretions from each of the 4 patients examined tested positive by RT-PCR, as were 12 of 43 swabs from patient rooms and 10 of 47 swabs from other parts of the hospital, including the computer mouses at 2 nursing stations and the handrail of the public elevator. Specimens from areas with patients with SARS in the most infectious phase of illness (days 5–15 after onset) were more likely to be RNA positive than were swab specimens from elsewhere (24 of 63 samples vs. 2 of 31 samples; P = .001). All cultures showed no growth.
Conclusions. Although the viruses identified may have been noninfectious, health care workers should be aware that SARS coronavirus can contaminate environmental surfaces in the hospital, and fomites should be considered to be a possible mode of transmission of SARS.
During the 2003 outbreak of severe acute respiratory syndrome (SARS), 21% of cases worldwide were in health care workers, varying from 3% in the United States to 43% in Canada [1]. Transmission was believed to be mainly by direct physical contact with ill persons or by large-droplet spread. Several well-described clusters of infection were difficult to explain by these routes, including transmission to 22 persons on an aircraft [2], to 13 guests sharing the same floor of a hotel [3], and to >300 persons in an apartment complex [4]. In addition, many health care workers were infected even after the widespread availability of recommendations on proper barrier-nursing precautions, the availability of personal protective equipment, and the apparent implementation of World Health Organization infection-control guidelines [1, 5, 6]. These observations led some to speculate about the possible role of more-remote transmission, including airborne spread by small droplets or true aerosols and fomite spread after contact with environmental surfaces possibly contaminated with SARS coronavirus (SARS CoV) [2, 6]. Environmental contamination and fomite spread has been occasionally implicated in hospital transmission of other viruses, such as rotavirus, and bacteria, such as Enterobacter cloacae [7, 8]. There is experimental evidence that the SARS CoV can survive for several hours on a dried microscope slide and possibly for longer in stool [1, 9], but the extent to which environmental surfaces were contaminated with SARS CoV during the course of the actual outbreak has not been described.
Methods
Samples were obtained from surfaces in 2 hospitals, one in Bangkok, Thailand (hospital A), and the other in Taipei, Taiwan (hospital B). Information about the SARS CoV infection status of the patients and the clinical stage of illness at the time of swabbing was recorded.
Hospital A was the main designated SARS hospital for the Thai Ministry of Public Health and provided care for several patients with suspected SARS, including one who had SARS CoV infection confirmed by positive culture results, seroconversion, and positive RT-PCR results using multiple primer sets on different samples. This patient died on 29 March 2003 (day 18 after fever onset) after multiple attempts at resuscitation, and environmental swabs were obtained from surfaces in his room and in the nurses' station within 2 h after the death and before any postmortem cleaning of the room or body was performed. At the time of sampling, health care workers in hospital A were consistently using full personal protective equipment, segregating the patient room from other parts of the ward, and consistently applying an effective disinfectant with use of procedures recommended in the most recent Healthcare Infection Control Practices Advisory Committee guidelines [10].
Hospital B, in Taipei, was the origin of the major outbreak of SARS in Taiwan [11]. This 500-bed municipal facility consists of 2 buildings connected by a walkway. On 24 April 2003, the hospital was hastily closed after recognizing an ongoing widespread nosocomial outbreak of SARS among health care workers and patients. All patients, visitors, and health care workers were quarantined, and all patients with recognized cases of SARS were cohorted on the sixth and eighth floors of building 1. On 28 April and 1 May, swab samples were obtained from surfaces on these 2 floors, where more than two-dozen patients with SARS were housed. Swab samples were also obtained from the medical intensive care unit on the fifth floor in building 2, where a single patient in his second week of illness was housed, and from the first floor of building 1, including a public lobby area that served as a SARS-response coordinating center and transfer station and the emergency room department where patients with possible SARS were evaluated and triaged. At the time of swabbing, health care workers in hospital B were using personal protective equipment inconsistently, and ventilation standards and environmental cleaning as recommended by the available guidelines were being established but were not consistently applied [10]. Patients from each of the 5 rooms that were swabbed were confirmed as having SARS CoV infection at some time during the course of their illness, either by RT-PCR, neutralizing antibody testing, or both. The day of illness on which these patients were confirmed as having SARS CoV infection ranged from day 5, for the patient in room 618, to day 12, for the patient in room 5303. Patients remained in the rooms when the surfaces were swabbed.
At the time of the first round of swabbing on 28 April 2003, the entire hospital had been contained for 5 days, a cordon of police officers had quarantined the hospital, and a total of 44 SARS cases had been reported among health care workers. The air conditioner had been turned off, the laundry and garbage removal services were not able to function, and there were early problems with the supply and distribution of personal protective equipment. In this chaotic environment, some hospital workers were observed wearing the same pair of gloves to care for multiple patients, patients with SARS were transported in the main elevators for radiography and other procedures, and family members quarantined inside the hospital continued to provide personal care for some patients with SARS.
Surfaces for swabbing were selected sequentially, beginning with the surfaces believed to be least likely to harbor virus and working towards those surfaces more likely to be contaminated, to minimize the possibility of cross-contamination. Procedures for swabbing were similar though not identical for the 3 sampling events. Personnel doing the swabbing were dressed in full protective clothing, including N-95 respirator, surgical head-and-neck hood, operating gown, clean outer plastic gown, 2 layers of disposable foot covering, and 2 sets of latex gloves. Meticulous care was taken to avoid cross-contamination of swabs. Each set of sterile swabs was opened separately, and viral transport media was closed and the used swab discarded before proceeding to the next surface. A new set of outer gloves was used for each new sample, and outer gowns were changed between rooms. A circular area ∼5 cm in diameter was swabbed with a sterile Dacron or cotton applicator moistened in viral transport media. The applicators were then twirled in a screw top vial, pressed against the side, and discarded.
The vials of viral transport media were brought in a portable cooler on wet ice, removed for the duration of swabbing (∼30 min), placed back in the cooler, and then returned to a -70°C freezer within 90 min after swabbing. The samples were then collected, labeled, packaged, and shipped on dry ice within 7 days after swabbing, according to guidelines issued by the International Air Transport Association.
Swabs were tested by RT-PCR [12] and viral culture [13] in separate laboratories, according to previously published methods. Specimens from Thailand and the samples collected on 28 April in Taiwan were tested in the United States at the Centers for Disease Control and Prevention (CDC; Atlanta, GA), and swabs obtained on 1 May were tested at the Centers for Disease Control in Taipei, Taiwan, using similar methods. In brief, aliquots of viral transport medium were inoculated onto Vero E6 cells and incubated for 2 weeks, with periodic examination for cytopathic effect. Real-time RT-PCR was conducted with use of 3 primer/probe sets complimentary to independent targets on the SARS CoV genome. Each run included a positive control and negative water controls for both RNA extraction and amplification steps. Samples were considered to be PCR-positive if there were positive results for ⩾2 of the 3 RT-PCR targets. To assess for the presence of RT-PCR inhibitors in the samples, all samples were also tested using a primer/probe set complimentary to the 16s ribosomal RNA included in the reaction mixture. Confirmatory analysis of samples that tested positive for SARS CoV was performed in an independent laboratory using 3 different primer/probe sets, and all samples with positive RT-PCR results were retested from new extracts of a previously unopened aliquot from the original sample. PCR products were sequenced and compared with the sequence of the strain of SARS CoV cultured in the laboratory.
Analysis was primarily descriptive. We defined “areas with patients in the most infectious phase” as those areas, including hospital rooms or the associated nursing stations, where there were patients with confirmed SARS CoV infection who had been ill for 5–15 days [9]. χ2 tests were used for comparison of proportions, and a P value of <.05 was considered to be significant.
Results
One of 18 swabs from hospital A in Bangkok tested positive for SARS CoV, as were 16 of 28 swabs collected at hospital B in Taipei on 28 April and 9 of 48 swabs collected at hospital B in Taipei on 1 May (table 1–3). Swab samples of the respiratory secretions of 4 patients were obtained during this exercise, and each patient had ⩾1 swab sample with a positive test result, including 3 swab samples from endotracheal tubes of living patients and 1 swab sample from the nasopharynx of a deceased person obtained <2 h after death. Swab samples with positive test results were obtained from 12 (28%) of 43 surfaces in patient rooms but were also obtained from 10 (21%) of 47 surfaces elsewhere in the hospital (P = .5).
Table 1.
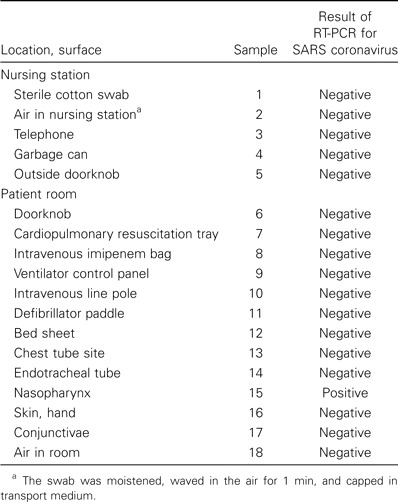
Results of RT-PCR of environmental and patient swab samples obtained from hospital A in Thailand within 2 h after the death of a patient with severe acute respiratory syndrome (SARS).
Table 3.
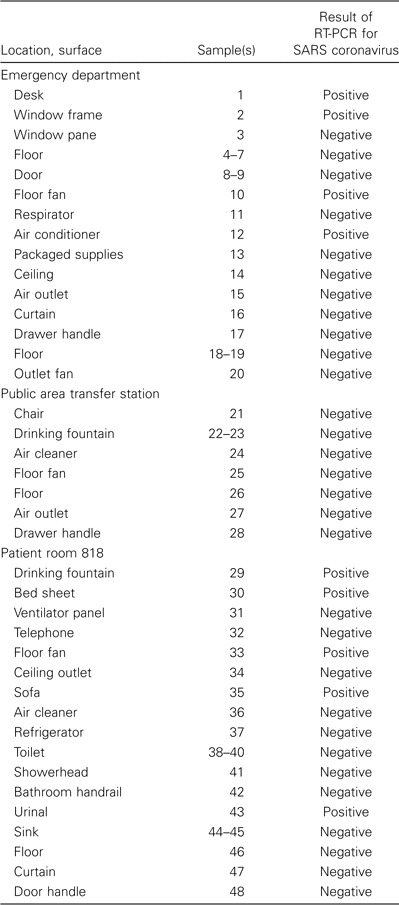
Results of RT-PCR of environmental swab samples obtained on 1 May 2003 from hospital B in Taiwan in which numerous patients with severe acute respiratory syndrome (SARS) were being treated.
Viral genome was detected in 24 of 63 samples obtained from areas with patients in the most infectious phase of SARS, which included the patient rooms, nursing stations, and the emergency department of hospital B in Taiwan. On the other hand, only 2 of 31 samples from the public areas of hospital B and from hospital A in Thailand tested positive (P = .001). Patients with SARS were present only transiently in the public areas of hospital B, and the samples from the patient in Thailand were obtained on day 18 after the onset of fever.
All swabs had cultures that showed no growth. The results of sequencing of the PCR products from Taiwan indicated that these strains were genetically distinct from the strain of SARS CoV routinely used in the CDC laboratory in the United States. All water and swab negative controls had negative RT-PCR results, and all samples had detectable 16S ribosomal RNA, indicating that false-negative results caused by RT-PCR inhibition were unlikely.
Discussion
We found evidence of SARS CoV contaminating a variety of environmental surfaces in some hospital settings but not others. These findings are consistent with observations from previous work with human coronaviruses that these agents can survive on dried inert surfaces and are consistent with proposals that contaminated fomites or hospital surfaces might contribute to spread [14, 15]. Surface contamination with infectious virus could explain transmission to health care workers who used appropriate barrier and airborne precautions when working directly with patients but not when working in other parts of the hospital, and it could explain some transmission to persons without close contact exposures to patients with SARS.
The presence of SARS CoV RNA on surfaces is clearly a concern. These data provide a reminder that surfaces can be easily contaminated, especially under stressful and chaotic situations. Surface contamination may occur by direct landing of droplets expelled during patient coughing or sneezing or as a result of indirect transfer from hands contaminated with excreted virus. The risk of contaminating surfaces with SARS CoV should be emphasized in guidelines for hand washing practices and for the safe removal and disposal of personal protective equipment when leaving areas potentially contaminated with SARS CoV. It also suggests that monitoring and reinforcing strict compliance with infection-control practices should be part of SARS prevention strategies [10]. Additionally, the possibility of environmental contamination following emergency resuscitation procedures and other events where infection-control practices may not have been followed should be addressed.
The differences in the extent of surface contamination between the hospitals in Taiwan and Thailand are worth considering. The surfaces in the Taiwan hospital were swabbed at a chaotic time. The hospital had just been quarantined, and most ancillary and support services were not functioning; this is the type of setting in which one would expect that a breakdown in infection control might occur. It is reassuring that no surfaces tested positive in the hospital in Thailand, perhaps implying that, in a nonchaotic setting, attention to standard quarantine procedures minimizes surface contamination. It is, however, also possible that some of the differences in rates of PCR positivity may be due to patient-specific factors and not to differences in infection control practices. The surfaces swabbed in the Thailand hospital were located in an area that had only 1 SARS CoV—positive patient, who had died at day 18 after the onset of illness, a time when viral load would be expected to be low. In contrast, the surfaces swabbed in the Taiwan hospital were located in areas where a number of SARS CoV—infected patients with durations of illness of between 5 and 15 days, when viral shedding is expected to be at its peak [9], had received treatment.
If contamination of hospital surfaces was widespread, and if the viruses were sometimes infectious, these findings might help to explain why health care workers continued to become infected long after the recommendations for strict barrier and airborne precautions were available [1, 5, 14]. It is important to distinguish between the availability of appropriate recommendations for infection-control and their consistent application. In the early days after recognition of the outbreak in the Taiwan hospital, SARS and general infection control guidelines were not consistently applied, and if protective equipment, environmental controls, and appropriate hand hygiene had been consistently applied [10], it is likely that any impact from possible environmental contamination would have been minimized.
We emphasize that none of the specimens was culture positive, indicating the possibility that what we identified was noninfectious viral genome in many, most, or all of the cases. It is also possible that there was viable virus on the surfaces but that it failed to grow after the initial handling, inconsistent refrigeration, or prolonged shipping time to the laboratories in Atlanta and Taipei. Other investigators have demonstrated that SARS CoV can survive dried on microscope slides for several hours and that viable virus can be cultured even after several days in a moist or stable environment, such as in stool samples [1].
Although the number of surfaces contaminated outside of the areas associated with patients with SARS was low, we did detect virus in the public elevator, and there appeared to be more contamination of surfaces in the nursing stations on both the sixth and eighth floors of the hospital in Taiwan. Comparisons of the proportion of swab samples that tested positive for SARS CoV should be interpreted with caution, because the selection of the type and number of surfaces to sample was done nonsystematically and in a chaotic environment and is therefore open to a variety of potential biases. Nevertheless, visitors and staff working in hospitals with patients who have SARS should be aware of the possibility of virus being present outside of the rooms of such patients, despite efforts to contain the spread.
Current dogma holds that SARS is transmitted mainly by direct contact with ill persons and by large-droplet spread [1]. Indeed, much of the epidemiology of the SARS epidemic can be well explained by these modes of transmission [16]. Transmission by more-remote means, such as airborne routes or through contaminated fomites, has been postulated as a way of explaining unusual clusters, such as transmission aboard an aircraft [2], on a hotel floor [3], or throughout an apartment complex [4]. Contamination of environmental surfaces may play a more important role in transmission than is currently appreciated, and the epidemiologic patterns associated with transmission from fomites, if clustered in patient-care areas with a high level of traffic, such as we found here, might be difficult to distinguish from the patterns predicted by person-to-person or large-droplet spread.
Table 2.
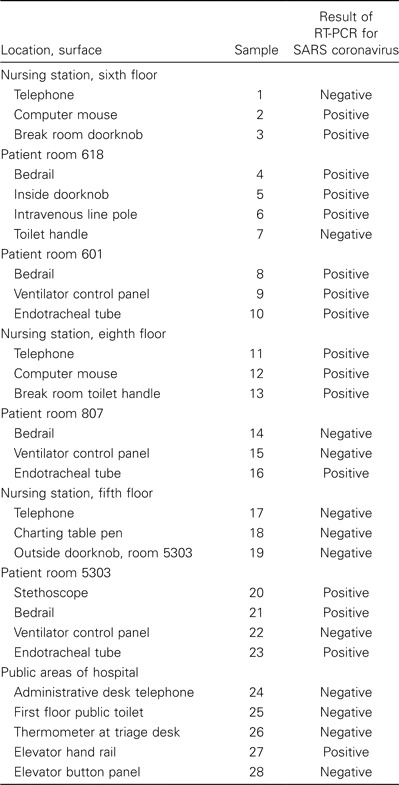
Results of RT-PCR of environmental swab samples obtained on 28 April 2003 from hospital B in Taiwan in which numerous patients with severe acute respiratory syndrome (SARS) were being treated.
Acknowledgments
We acknowledge with gratitude the contributions of James A. Comer, for viral culture; Chwan-Chuen King, Ruey Jen-Su, David Wong, and James Lando, for assistance with the response in Taiwan; and the dedication of the many health care workers in both hospitals, for carrying on their work despite the real threat to their health and lives.
Financial support. The study was conducted during an acute outbreak and had no dedicated funding, but investigators were supported by the government of Taiwan and by the US Centers for Disease Control and Prevention.
Conflict of interest. All authors: No conflict.
References
- 1.World Health Organization. Consensus document on the epidemiology of severe acute respiratory syndrome (SARS) Geneva: World Health Organization; 2003. [Google Scholar]
- 2.Olsen S, Chang H, Cheung T, et al. Transmission of the severe acute respiratory syndrome on aircraft. N Engl J Med. 2003;349:2416–2422. doi: 10.1056/NEJMoa031349. [DOI] [PubMed] [Google Scholar]
- 3.Centers for Disease Control Prevention. Update: outbreak of severe acute respiratory syndrome—worldwide, 2003. MMWR Morb Mortal Wkly Rep. 2003;52:241–6. 248. [PubMed] [Google Scholar]
- 4.Lee SH. The SARS epidemic in Hong Kong. J Epidemiol Community Health. 2003;57:652–4. doi: 10.1136/jech.57.9.652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Centers for Disease Control Prevention. Cluster of severe acute respiratory syndrome cases among protected health care workers—Toronto, Canada, April 2003. MMWR Morb Mortal Wkly Rep. 2003;52:433–6. [PubMed] [Google Scholar]
- 6.Scales DC. Illness in intensive care staff after brief exposure to severe acute respiratory syndrome. Emerg Infect Dis. 2003;9:1205–10. doi: 10.3201/eid0910.030525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Widdowson MA, van Doornum GJ, van der Poel WH, et al. An outbreak of diarrhea in a neonatal medium care unit caused by a novel strain of rotavirus: investigation using both epidemiologic and microbiological methods. Infect Control Hosp Epidemiol. 2002;23:665–70. doi: 10.1086/501991. [DOI] [PubMed] [Google Scholar]
- 8.Yu WL, Cheng HS, Lin HC, Peng CT, Tsai CH. Outbreak investigation of nosocomial enterobacter cloacae bacteraemia in a neonatal intensive care unit. Scand J Infect Dis. 2000;32:293–8. doi: 10.1080/00365540050165947. [DOI] [PubMed] [Google Scholar]
- 9.Peiris JS, Chu CM, Cheng VC, et al. Clinical progression and viral load in a community outbreak of coronavirus-associated SARS pneumonia: a prospective study. Lancet. 2003;361:1767–72. doi: 10.1016/S0140-6736(03)13412-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Sehulster L, Chinn RY. Guidelines for environmental infection control in health-care facilities: recommendations of CDC and the Healthcare Infection Control Practices Advisory Committee (HICPAC) MMWR Recomm Rep. 2003;52:1–42. [PubMed] [Google Scholar]
- 11.Centers for Disease Control Prevention. Severe acute respiratory syndrome—Taiwan, 2003. MMWR Morb Mortal Wkly Rep. 2003;52:461–6. [PubMed] [Google Scholar]
- 12.Emery S, Erdman D, Meyer R, et al. Real-time reverse-transcriptase polymerase chain reaction assay for the SARS-associated Coronavirus. Emerg Infect Dis. 2004;10:311–6. doi: 10.3201/eid1002.030759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ksiazek TG, Erdman D, Goldsmith CS, et al. A novel coronavirus associated with severe acute respiratory syndrome. N Engl J Med. 2003;348:1953–66. doi: 10.1056/NEJMoa030781. [DOI] [PubMed] [Google Scholar]
- 14.Sizun J, Yu MW, Talbot PJ. Survival of human coronaviruses 229E and OC43 in suspension and after drying onsurfaces: a possible source of hospital-acquired infections. J Hosp Infect. 2000;46:55–60. doi: 10.1053/jhin.2000.0795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ijaz MK, Brunner AH, Sattar SA, Nair RC, Johnson-Lussenburg CM. Survival characteristics of airborne human coronavirus 229E. J Gen Virol. 1985;66:2743–8. doi: 10.1099/0022-1317-66-12-2743. [DOI] [PubMed] [Google Scholar]
- 16.Seto WH, Tsang D, Yung RW, et al. Effectiveness of precautions against droplets and contact in prevention of nosocomial transmission of severe acute respiratory syndrome (SARS) Lancet. 2003;361:1519–20. doi: 10.1016/S0140-6736(03)13168-6. [DOI] [PMC free article] [PubMed] [Google Scholar]


