Abstract
Classical swine fever (CSF) is a highly contagious viral disease of pigs which causes major economic losses worldwide. No specific drug is currently available for the effective treatment of CSFV infection; however, RNA interference (RNAi) has been applied successfully to inhibit the replication of human and other animal viruses. In this study, three effective siRNAs targeting NS3 of CSFV were selected. siNS3-2 targeting NS3 gene was chosen for further experimentation, while siN1 and siN2 targeting Npro gene, and siNS5B targeting NS5B gene describe previously. Single, double and quadruple anti-CSFV siRNA expression plasmids, with loxp sites at each end of the selectable marker genes, were constructed and analyzed using the same promoters or four different promoters, targeting Npro, NS3 and NS5B genes of CSFV. Results indicate that single or multiple siRNA expression plasmids can efficiently inhibit CSFV replication and that inhibition was markedly stronger when multiple siRNAs were expressed targeting different genes of CSFV. Since RNAi applied to anti-CSFV research, this study provides anti-CSFV methods by single and multiple siRNA expression which can target most viral isolates of different subtypes and prevent viral escape. It also provides a basis for development of CSFV-resistant transgenic pigs.
Keywords: CSFV, RNAi, Multiple siRNA expression, Inhibition, Viral escape
1. Introduction
RNA interference (RNAi) is a natural post-transcriptional gene silencing mechanism. It has been applied successfully to inhibit the replication of human and other animal viruses, such as human immunodeficiency virus type 1 (HIV-1) (ter Brake et al., 2008), SARS-CoV (Wu et al., 2005), influenza virus A (Sui et al., 2009), foot-and-mouth disease virus (Chen et al., 2004), porcine reproductive respiratory syndrome virus (Li et al., 2009a) and classical swine fever virus (Xu et al., 2008), both in vitro and in vivo.
Classical swine fever (CSF) is a highly contagious disease of pigs caused by infection with CSFV, and is a notifiable disease of the World Organization for Animal Health (OIE). CSF causes great economic losses in the pig industry worldwide. The disease was first recognized in China in the 1920s and there continue to be major epizootics in this country (Tu et al., 2001, Zhu et al., 2009). CSFV belongs to the genus Pestivirus of the family Flaviviridae. Its genome consists of a single-stranded (+) sense RNA of about 12.5 kb with a single large open reading frame (ORF), encoding a polyprotein of approximately 3400 amino acids. After translation, the polyprotein is cleaved into viral structural and non-structural peptides which are, from N- to C-terminus, Npro-C-Ems-E1-E2-P7-NS2-NS3-NS4A-NS4B-NS5A-NS5B. Npro is essential for evading the cellular antiviral defense system, and protects cells from double-stranded RNA-induced apoptosis (Ruggli et al., 2005). The NS3 serine protease cleaves at downstream cleavage sites (Tautz et al., 1997, Tautz et al., 2000) and additionally possesses helicase and NTPase activities essential for viral RNA replication (Gu et al., 2000, Moulin et al., 2007). NS5B, an RNA-dependent RNA polymerase, is responsible for viral RNA replication (Zhang et al., 2005).
Previous studies of in vitro transcribed siRNA molecules (Xu et al., 2008) and retroviral-mediated RNAi (Li et al., 2010) showed that RNAi is capable of specific and efficient inhibition of CSFV replication. One problem is that siRNA cannot entirely inhibit CSFV replication. This has been suggested to be due to loss of siRNA activity caused by point mutation or deletion of the siRNA target site (Konishi et al., 2006, Westerhout et al., 2005). In this study, single, double, and quadruple anti-CSFV siRNA expression plasmids, with loxp sites at both ends of the selectable marker gene, were constructed and analyzed using the same promoters or four different promoters. Four different siRNA expressing from four different promoters can avoid recombination (ter Brake et al., 2008). Results show that CSFV inhibition was markedly stronger when multiple siRNAs were expressed targeting different genes of CSFV, and that siRNA target region mutation is not a major reason why CSFV replication cannot be completely blocked by RNAi. It also provides a basis for development of CSFV-resistant transgenic pigs, this is the reason why siRNA expression plasmids were constructed with loxp sites at each end of the selectable marker genes which could be excised by Cre-mediated loxP-flanked sequences.
2. Materials and methods
2.1. Cells, virus, and antiserum
Pig kidney cell line PK-15 (80 passages) and virulent CSFV strain Shimen were obtained from the Institute of Veterinary Drug Control, China. MAb WH303 (Kindly provided by Dr. Trevor Drew, Veterinary Laboratories Agency, Weybridge, UK), raised against CSFV strain UK/86/2 (Edwards and Sands, 1990, Paton et al., 1995) and with affinity for the E2 protein, was generated in the ascitic fluid of BALB/c mice.
2.2. Screening for siRNAs targeting NS3 gene
2.2.1. siRNA duplexes
All siRNAs were designed and synthesized by Shanghai GeneChem Co., Ltd. (Shanghai, China) with symmetrical 19-UU overhangs. The target sequences of siNS3-1, siNS3-2 and siNS3-3 were GCU UAU GAG UGG AAU ACA AUU (nucleotides 5713–5731 of CSFV genome, GenBank Accession AF092448), GAU GCC ACA ACC UAA GUU AUU (nucleotides 6046–6064), and CCA CUA UGA UCU ACU GCA AUU (nucleotides 6724–6742), respectively. A negative control siRNA was synthesized with the target sequence UUC UCC GAA CGU GUC ACG UUU, which has no matches either in the viral or the porcine genome.
2.2.2. siRNA transfection and CSFV infection
When cell monolayers were 50–70% confluent, siRNA was introduced using X-tremeGene siRNA Transfection Reagent (Roche, Indianapolis, IN) according to the Manufacturer’s protocol, with a siRNA final concentration of 100 nM. The siRNA-transfected cells were infected with 2000 CCID50 CSFV and cultured for 72 h at 37 °C. Indirect immunofluorescence assay (IFA), detection of the copy number of CSFV genomic RNA and detection of infectious virus production were performed as described previously (Li et al., 2010).
2.3. Plasmid construction
2.3.1. Single and double siRNA expression plasmid construction
Three 21nt candidate sequences, siN1, siN2 and siNS5B, designed and described previously (Xu et al., 2008), were chosen as siRNA for Npro and NS5B genes. siNS3-2, described above, was chosen as siRNA for the NS3 gene. Sequences selected for a negative siRNA control (siC) were the same as in Xu et al., 2008. Oligonucleotides were synthesized for each sequence by Nanjing Genscript Co., Ltd. (Nanjing, China; Table 1 ). These oligonucleotides were annealed and ligated to the BamH1 and HindIII sites of pSilencer 3.1 H1 (Ambion, Austin, TX) to obtain plasmids pH1-(siRNAs), where “siRNAs” represents siN1, siN2, siNS3-2, siNS5B and siC.
Table 1.
List of siRNA sequences in this study.
| Target namea | Sequences of siRNA (5′–3′)b |
|---|---|
| siN1 S | GACGGATCCGGATAGGTAGGGTGACAGGTTCAAGAGACCTGTCACCCTACCTATCCTTTTTTGGAAAAGCTTGCA |
| siN1 AS | TGCAAGCTTTTCCAAAAAAGGATAGGTAGGGTGACAGGTCTCTTGAACCTGTCACCCTACCTATCCGGATCCGTC |
| siN2 S | GACGGATCCGAACCCTGAAGTGGATTAGTTCAAGAGACTAATCCACTTCAGGGTTCTTTTTTGGAAAAGCTTGCA |
| siN2AS | TGCAAGCTTTTCCAAAAAAGAACCCTGAAGTGGATTAGTCTCTTGAACTAATCCACTTCAGGGTTCGGATCCGTC |
| siNS3-2 S | GACGGATCCGATGCCACAACCTAAGTTATTCAAGAGATAACTTAGGTTGTGGCATCTTTTTTGGAAAAGCTTGCA |
| siNS3-2 AS | TGCAAGCTTTTCCAAAAAAGATGCCACAACCTAAGTTATCTCTTGAATAACTTAGGTTGTGGCATCGGATCCGTC |
| siNS5B S | GACGGATCCGAATGAGAAGAGGGACGTCTTCAAGAGAGACGTCCCTCTTCTCATTCTTTTTTGGAAAAGCTTGCA |
| siNS5B AS | TGCAAGCTTTTCCAAAAAAGAATGAGAAGAGGGACGTCTCTCTTGAAGACGTCCCTCTTCTCATTCGGATCCGTC |
| siC S | GGAGGATCCGGACAGTGGGATGGATAGGTTCAAGAGACCTATCCATCCCACTGTCCTTTTTTGGAAAAGCTTCGG |
| siC AS | CCGAAGCTTTTCCAAAAAAGGACAGTGGGATGGATAGGTCTCTTGAACCTATCCATCCCACTGTCCGGATCCTCC |
S means sense strand, AS means antisense strand, C means control shRNA sequence.
For each target gene (sense strand as an example), complementary 75-mer oligonucleotides were designed with 5′ single-stranded overhangs (BamHI) and 3′ single-stranded overhangs (HindIII) for ligation into the pSilencer vectors. Sequences for enzyme sites are underlined. The oligonucleotides should encode 19-mer hairpin sequences specific to the mRNA target, a loop sequence separating the two complementary domains (TTCAAGAGA), and a polythymidine tract to terminate transcription (TTTTTT).
H1-(siRNAs) were amplified by PCR using these plasmids as templates, with the following primers: H1-fw-n, H1-rev-n, H1-fw-c and H1-rev-c (Table 2 ). H1-(siRNAs) were digested and ligated to the appropriate insertion sites of PGKneotpAlox2 (Addgene, Plasmid 13444) to form plasmid pLox-(siRNAs) (see Fig. 2 A–C). The insert sequences were confirmed by Shanghai Sangon Biological Engineering Technology & Service Co. (Shanghai, China).
Table 2.
Primers used in this study.
| Primer name | Sequences (5′–3′) | Enzyme sites |
|---|---|---|
| H1-fw-n | CTTGCGGCCGCGGTTTTCCCAGTCACGAC | NotI |
| H1-rev-n | GCGGCGGCCGCGCACCCCAGGCTTTACACTTT | NotI |
| H1-fw-c | CTTATCGATGGTTTTCCCAGTCACGAC | ClaI |
| H1-rev-c | GCGATCGATGCACCCCAGGCTTTACACTTT | ClaI |
| Npro-fw | GGCCTCGAGATGGAGTTGAATCATTTTG | XhoI |
| Npro-rev | GCCCTGCAGGCAACTGGTAACCCACAATG | PstI |
| ΔNS3-fw | GGCCTCGAGATGAGAGTCTTGGTCTTGATCCC | XhoI |
| ΔNS3-rev | GCCCTGCAGTGCGGTCATGGCTACTACCCG | PstI |
| ΔNS5B-fw | GGCCTCGAGATGGGTGCTGCTGGTTTCTTTGAA | XhoI |
| ΔNS5B-rev | GCCCTGCAGCCATTCCCTCTTTACTTTGTC | PstI |
| S-Npro-fw | ATGGAGTTGAATCATTTTG | |
| S-Npro-rev | GCAACTGGTAACCCACAATG | |
| S-ΔNS3-fw | AGAGTCTTGGTCTTGATCCC | |
| S-ΔNS3-rev | TGCGGTCATGGCTACTACCCG | |
| S-ΔNS5B-fw | GGTGCTGCTGGTTTCTTTGAA | |
| S-ΔNS5B-rev | CCATTCCCTCTTTACTTTGTC |
Fig. 2.
Single or multiple siRNA expression plasmids construction. (A) Four siRNA sites targeting Npro, NS3, NS5B genes of CSFV. (B) Single siRNA expression plasmid construction. (C) Double siRNA expression plasmid construction. (D) Four siRNA expression plasmid construction.
2.3.2. siRNA4 expression plasmid construction
Construction of siRNA4 expression plasmid is based on pGenesil 1.1–1.4 (GeneSil Biotechnology Co., Wuhan, China), which respectively contain human U6, murine U6, human 7SK and human H1 polymerase III promoters (which were renamed phU6, pmU6, ph7SK and phH1). First, siRNA expression plasmids were constructed by inserting annealed oligonucleotides into the appropriate insertion sites, thereby encoding the siRNA transcription unit. Second, the pGenesil-siRNA4 plasmid was constructed from the different siRNA expression plasmids (phU6-siN1, pmU6-siNS3-2, ph7SK-siNS5B and phH1-siN2) by GeneSil Biotechnology Co. Finally, the siRNA cassette was digested and inserted into the SalI/XhoI sites of PGKneotpAlox2, yielding pLox-siRNA4 (see Fig. 2D).
2.3.3. Target site-EGFP fusion reporter plasmid construction
Npro (nucleotides 347–877 of CSFV genome), ΔNS3 (nucleotides 5885–6183) and ΔNS5B (nucleotides 10571–10912) were amplified by PCR using primers Npro-fw, Npro-rev, ΔNS3-fw, ΔNS3-rev, ΔNS5B-fw and ΔNS5B-rev (Table 2). The products were digested and ligated to the XhoI/PstI sites of pEGFP-N1 (Clontech, Mountain View, CA) to form plasmids pEGFP-Npro, pEGFP-ΔNS3 and pEGFP-ΔNS5B.
2.4. Co-transfection of reporters and double siRNA or siRNA4 expression plasmids
PK-15 cells at 50–70% confluence in 96-well plates were co-transfected with 1 μg each of fusion reporter plasmid and double siRNA or siRNA4 expression plasmid, or control plasmid pLox-siC, using FuGENE HD Transfection Reagent (Roche, Indianapolis, IN) according to the manufacturer’s protocol. After 72 h, cells were examined and photographed with a Zeiss Axioskop 40 fluorescence microscope.
2.5. Single or multiple siRNA plasmid transfection and CSFV infection
PK-15 cells at 50–70% confluence in 12-well plates were transfected with 2 μg single or multiple siRNA plasmids using FuGENE HD Transfection Reagent according to the Manufacturer’s protocol, then infected with 1000 CCID50 CSFV and cultured for 48 or 72 h. Indirect immunofluorescence assay (IFA), detection of CSFV genome copy number and detection of infectious virus production were performed as described previously (Li et al., 2010).
2.6. Analysis of CSFV escape RNA interference
pLox-siRNAs were linearized by KpnI and SacI and then used to transfect PK-15 cells. G418-resistant (500 μg/mL, GIBCO) cells derived from single cell cultures were screened for stable siRNA expression. Positive cells were examined by IFA for inhibition of CSFV replication after infection with CSFV.
Stable siRNA-expressing cells were used to seed 3 mL cell culture tubes (Nunc, Rochester, NY), incubated with 1000 CCID50 CSFV in MEM for 2 h and then with MEM/2% FBS containing G418 (250 μg/mL) for 72 h. The tubes were then freeze–thawed three times, and the virus-containing supernatant was stored at −80 °C until further use. Using the above procedure, a total of 10 passages were carried out by transferring 1 mL of supernatant from the previous passage. The siRNA target sequences of passage 10 supernatants were amplified by PCR using primers listed in Table 2. Sequences were confirmed by Shanghai Sangon Biological Engineering Technology & Service Co. (Shanghai, China).
2.7. RNA purification
Total RNA was isolated using Trizol reagent (Invitrogen, Carlsbad, CA) or QuickGene RNA cell culture cell HC kit S (FUJIFILM, Japan) according to the Manufacturers’ protocols.
2.8. Statistical analysis
All numerical parameters, including genome copies and viral titers, are expressed as the mean ± 1 standard deviation. Student’s t-test was used for the analysis of numeric parameters (SPSS 13.0 for Windows, SPCC Inc.) and calculation of P values.
3. Results
3.1. Screening for siRNA targeting the NS3 gene
To study the inhibitory effects of the three siRNAs targeting the NS3 gene on CSFV replication, levels of viral antigen, viral genome copy number and production of viable virus in the cells with siRNA transfection and CSFV infection were examined. At 72 h post-CSFV infection, only a few cells displayed green fluorescence compared with the control (Fig. 1 A). The viral genome copy number was significantly decreased in the cells receiving siNS3-1 (P < 0.01), siNS3-2 (P < 0.01) or siNS3-3 (P < 0.05) (Fig. 1B). The production of viable virus was significantly decreased in cells transfected with siNS3-1 (P < 0.05), siNS3-2 (P < 0.01) or siNS3-3 (P < 0.05) (Fig. 1C). These results indicate that the three siRNAs were effective inhibitors of CSFV replication, especially siNS3-2, which was therefore chosen for further experimentation.
Fig. 1.
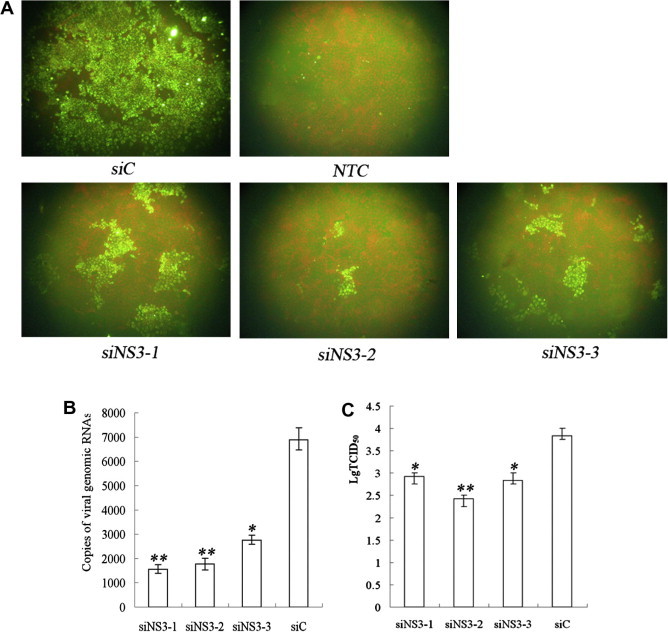
Screening for siRNA targeting NS3 gene. (A) Inhibition of CSFV replication by IFA in siRNA-treated cells (100× magnification). siC (siC transfection), NTC (mock transfection, stained with CSFV-negative serum), siNS3-1 (siNS3-1 transfection), siNS3-2 (siNS3-2 transfection), siNS3-3 (siNS3-3 transfection). (B) Reduction of viral genome replication in siRNA-treated cells. The CSFV genome copy numbers are the mean of three repeat experiments. (C) Inhibition of viable viral production in siRNA-treated cells. CCID50 values are the means of three repeat titrations at the time points indicated. Ninety five percent confidence intervals are shown, ∗P ⩽ 0.05, ∗∗P ⩽ 0.01.
3.2. Effect of multiple siRNA expression
PK-15 cell cultures were co-transfected with the multiple siRNA expression plasmids and EGFP reporters containing CSFV target sequences described above. EGFP expression levels were reduced only when the siRNA matched the reporter (data not shown). This confirmed that the observed inhibition of CSFV replication occurred through a sequence-specific mechanism. Similar results were obtained for multiple siRNA expression plasmids and matching reporters (Fig. 3 ). These data showed that each siRNA was still functional when two or four different siRNAs were expressed within a single vector.
Fig. 3.
Co-transfection of PK-15 cells with EGFP reporters and siRNA2 or siRNA4 expression plasmids (40× magnification).
3.3. Examination of the single or multiple siRNA effect by IFA
Using IFA and MAb WH303, levels of viral antigen were determined following CSFV infection of PK-15 cells transfected with single or multiple siRNA expression plasmids. At 72 h post-CSFV infection, 85% PK-15 cells expressing siC exhibited bright green fluorescence in the cytoplasm, indicating that most cells in the control were producing virus (Fig. 4 I). By contrast, only a small proportion of cells (<5%) receiving single or multiple siRNA expression plasmids displayed green fluorescence, indicating that most cells were effectively protected by the siRNAs (Fig. 4A–H). Additionally, the level of protection was enhanced by multiple siRNA expression, especially that of siRNA4 targeting different genes of CSFV.
Fig. 4.
Expression of viral antigen in PK-15 cells transfected with single or multiple siRNA expression plasmids at 72 h post-CSFV infection (40× magnification). (A) siN1 transfection, (B) siN2 transfection, (C) siNS3-2 transfection, (D) siNS5B transfection, (E) siN1N2 transfection, (F) siN1NS3-2 transfection, (G) siN1NS5B transfection, (H) siRNA4 transfection, (I) siC transfection, (J) NTC (mock transfection, stained with CSFV-negative serum).
3.4. Single or multiple siRNA effects by real-time RT-PCR
Viral genome copy number was determined by real-time PCR (Mx3000P, Stratagene), using serially diluted plasmid pT-5′UTR as a standard. The R 2 value of the standard curve was 0.987 and the average amplification efficiency E was 0.994. Sequencing of products showed that they were the expected part of the 5′UTR of the gene, thereby demonstrating the specificity and reliability of the analysis. These data indicate that real-time PCR was highly reliable in the assay. The copy number of the viral genome was significantly decreased (77.2–96.8%) in cells expressing single or multiple siRNAs compared with siC cells (P < 0.01) (Fig. 5 ). The viral genome copy number was also significantly decreased in cells expressing siRNA4 compared with those expressing single siRNA plasmids (P < 0.05), indicating that inhibition efficiency was enhanced by multiple siRNAs targeting different genes of CSFV.
Fig. 5.
Reduction of viral genome copy number in PK-15 cells transfected with single or multiple siRNA expression plasmids at 48 or 72 h post-CSFV infection. Genome copy numbers are the means of three experiments. The viral genome copy numbers per nanogram of total RNA from siC-transfected cells, calculated from the standard curve, were 1.55 × 105/ng and 2.11 × 105/ng at 48 and 72 h post-CSFV infection, respectively. Ninety five percent confidence intervals are shown, ∗P ⩽ 0.05, ∗∗P ⩽ 0.01.
3.5. Single or multiple siRNA effects by infectious virus assay
As measured by CCID50 assay, production of viable virus was decreased 2.9- to 30.6-fold in cells expressing single or multiple siRNAs compared with siC cells (Fig. 6 ). Production of viable virus was also significantly decreased in the siRNA4 cells expressing single siRNAs (P < 0.05), showing that multiple siRNA expression targeting different genes of CSFV provides more effective inhibition.
Fig. 6.
Inhibition of viable viral production in PK-15 cells transfected with single or multiple siRNA expression plasmids at 48 or 72 h post-CSFV infection. CCID50 values were calculated by the Kärber method. CCID50 are the means of three repeat titrations at the time points indicated. The viable viral production from siC-transfected cells were 103.92 CCID50/mL and 104.33 CCID50/mL at 48 and 72 h post-CSFV infection, respectively. Ninety five percent confidence intervals are shown, ∗P ⩽ 0.05, ∗∗P ⩽ 0.01.
3.6. Genetic stability of CSFV exposed to RNA interference
To investigate if CSFV could develop resistance to RNAi, growth studies were carried out in stable siRNA-expressing PK-15 cells. Ten lines of cells were infected with CSFV and passaged 10 times. Viral genomes from each line at the 10th passage were sequenced in the siRNA target region and compared with the corresponding region of the parental virus genome. No mutations were detected (data not shown).
4. Discussion
CSFV genome is a single-stranded RNA that functions as both messenger RNA and a replication template, making it an attractive target for the study of RNA interference. Successful RNA interference with inhibition of CSFV replication has already been demonstrated by synthetic siRNA (Xu et al., 2008) and retroviral vector-mediated siRNA targeting Npro, NS4A and NS5B genes (Li et al., 2010). To our knowledge, no siRNA targeting NS3 gene of CSFV has been reported. NS3 possesses serine protease, helicase and NTPase activities that are all essential for viral RNA replication. Korf et al. (2007) reported inhibition of the HCV NS3 gene by siRNA. In the present study, three siRNAs targeting the NS3 gene of CSFV effectively inhibited CSFV replication, and siNS3-2 was most effective with a reduction in titer of up to 95.1%. These provided the candidate targeting sites to explore a new approach for preventing CSF.
For the development of an effective RNAi-based gene silencing against CSFV, a simple strategy would be to express multiple siRNA from a single or multiple promoters in one vector (Liu et al., 2008, Saayman et al., 2008, ter Brake et al., 2006, ter Brake et al., 2008). Previous studies have performed an analysis of a multiple siRNA expression strategy in which three or four siRNAs were expressed from the same promoters and found that this caused frequent recombinations at repeat sequences of the expression cassette, resulting in deletion of one or two cassettes (ter Brake et al., 2008). To prevent recombination, therefore, we selected four different promoters lacking sequence similarity for the four siRNA expression strategy. Human polymerase III promoters H1, U6 and 7SK and the murine polymerase III promoter U6 were selected because they have well defined transcription start and termination sites. Cloning the target sequences upstream of a reporter gene is a simple and convenient method for examining whether siRNA transcription occurs from multiple expression plasmids. Fig. 3 shows that each siRNA expression cassette was active, confirming that multiple siRNA expression from these promoters is a valid approach.
With vectors expressing siRNAs in PK-15 cells, multiple siRNA expression inhibited CSFV replication much more effectively than a single siRNA. This observation is consistent with a previous report that multiple siRNAs targeting the same mRNA results in enhanced gene silencing (Ji et al., 2003). Reduction in viral genome copy number and viable virus production at 48 and 72 h post-CSFV infection from multiple siRNA expression indicates that the effect of inhibition of CSFV replication is additive and maintained. This cumulative effect may restrict optimal shRNA processing by limiting the cellular components of the RNAi machinery (Castanotto et al., 2007), which might explain why each siRNA expression level from the siRNA4 was reduced compared to the single siRNA expression vector, as measured by Northern blot (ter Brake et al., 2008).
Viral replication is not completely blocked by the combined expression of multiple siRNAs. This is not surprising since RNAi does not target the incoming genome. Virus replication may still occur, therefore, albeit at a very low level, perhaps slowly accumulating until a certain threshold is reached.
Virus escape as a result of long-term exposure to antiviral RNAi has been observed in several studies (Das et al., 2004, Gitlin et al., 2002, Konishi et al., 2006). In the present study, however, no mutations of the target sites were detected even by the tenth virus passage in siRNA-expressing cells. These results indicate that target site mutation is not the major reason why CSFV replication cannot be completely blocked by RNAi.
The Cre/LoxP DNA recombinase system has been developed as a powerful tool for manipulating DNA both in vitro and in vivo (Guo et al., 1997, Li et al., 2009b, Schmidt et al., 2000), with many transgenic mouse models having been established using Cre/Loxp technology (Chen et al., 2008, Lin et al., 2008). Li et al. (2009b) created an EGFP reporter transgenic pig by applying the Cre/Loxp recombinase system. If the anti-CSFV activity of RNAi were to be combined with the technology of producing a transgenic pig, the result could be a pig resistant to CSFV infection. This is the reason why we constructed single and multiple anti-CSFV siRNA expression plasmids with loxp sites at each end of the selectable marker genes which could be excised by Cre-mediated loxP-flanked sequences. Work to make this concept a reality is currently in progress.
Acknowledgements
This work was supported by National New Breed of Transgenic Livestock Program (Grant No. 2008ZX08006-001). The authors would like to thank Professor James B. Campbell, Department of Medical Genetics and Microbiology, University of Toronto, Canada, for comments and help with the English language.
References
- Castanotto D., Sakurai K., Lingeman R., Li H., Shively L., Aagaard L., Soifer H., Gatignol A., Riggs A., Rossi J.J. Combinatorial delivery of small interfering RNAs reduces RNAi efficacy by selective incorporation into RISC. Nucleic Acids Res. 2007;35:5154–5164. doi: 10.1093/nar/gkm543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen J., Futami K., Petillo D., Peng J., Wang P., Knol J., Li Y., Khoo S.K., Huang D., Qian C.N., Zhao P., Dykema K., Zhang R., Cao B., Yang X.J., Furge K., Williams B.O., Teh B.T. Deficiency of FLCN in mouse kidney led to development of polycystic kidneys and renal neoplasia. PLoS One. 2008;3:e3581. doi: 10.1371/journal.pone.0003581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen W., Yan W., Du Q., Fei L., Liu M., Ni Z., Sheng Z., Zheng Z. RNA interference targeting VP1 inhibits foot-and-mouth disease virus replication in BHK-21 cells and suckling mice. J. Virol. 2004;78:6900–6907. doi: 10.1128/JVI.78.13.6900-6907.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Das A.T., Brummelkamp T.R., Westerhout E.M., Vink M., Madiredjo M., Bernards R., Berkhout B. Human immunodeficiency virus type 1 escapes from RNA interference-mediated inhibition. J. Virol. 2004;78:2601–2605. doi: 10.1128/JVI.78.5.2601-2605.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Edwards S., Sands J.J. Antigenic comparisons of hog cholera virus isolates from Europe, America and Asia using monoclonal antibodies. Dtsch. Tierarztl. Wochenschr. 1990;97:79–81. [PubMed] [Google Scholar]
- Gitlin L., Karelsky S., Andino R. Short interfering RNA confers intracellular antiviral immunity in human cells. Nature. 2002;418:430–434. doi: 10.1038/nature00873. [DOI] [PubMed] [Google Scholar]
- Gu B., Liu C., Lin-Goerke J., Maley D.R., Gutshall L.L., Feltenberger C.A., Del Vecchio A.M. The RNA helicase and nucleotide triphosphatase activities of the bovine viral diarrhea virus NS3 protein are essential for viral replication. J. Virol. 2000;74:1794–1800. doi: 10.1128/jvi.74.4.1794-1800.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guo F., Gopaul D.N., van Duyne G.D. Structure of Cre recombinase complexed with DNA in a site-specific recombination synapse. Nature. 1997;389:40–46. doi: 10.1038/37925. [DOI] [PubMed] [Google Scholar]
- Ji J., Wernli M., Klimkait T., Erb P. Enhanced gene silencing by the application of multiple specific small interfering RNAs. FEBS Lett. 2003;552:247–252. doi: 10.1016/s0014-5793(03)00893-7. [DOI] [PubMed] [Google Scholar]
- Konishi M., Wu C.H., Kaito M., Hayashi K., Watanabe S., Adachi Y., Wu G.Y. SiRNA-resistance in treated HCV replicon cells is correlated with the development of specific HCV mutations. J. Viral Hepat. 2006;13:756–761. doi: 10.1111/j.1365-2893.2006.00752.x. [DOI] [PubMed] [Google Scholar]
- Korf M., Meyer A., Jarczak D., Beger C., Manns M.P., Krüger M. Inhibition of HCV subgenomic replicons by siRNAs derived from plasmids with opposing U6 and H1 promoters. J. Viral Hepat. 2007;14:122–132. doi: 10.1111/j.1365-2893.2006.00793.x. [DOI] [PubMed] [Google Scholar]
- Li G., Jiang P., Li Y., Wang X., Huang J., Bai J., Cao J., Wu B., Chen N., Zeshan B. Inhibition of porcine reproductive and respiratory syndrome virus replication by adenovirus-mediated RNA interference both in porcine alveolar macrophages and swine. Antiviral Res. 2009;82:157–165. doi: 10.1016/j.antiviral.2009.02.202. [DOI] [PubMed] [Google Scholar]
- Li J., Guo H., Shi Z., Tu C. In vitro inhibition of CSFV replication by retroviral vector-mediated RNA interference. J. Virol. Methods. 2010;169:316–321. doi: 10.1016/j.jviromet.2010.07.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li L., Pang D., Wang T., Li Z., Chen L., Zhang M., Song N., Nie D., Chen Z., Lai L., Ouyang H. Production of a reporter transgenic pig for monitoring Cre recombinase activity. Biochem. Biophys. Res. Commun. 2009;382:232–235. doi: 10.1016/j.bbrc.2009.02.146. [DOI] [PubMed] [Google Scholar]
- Lin P.P., Pandey M.K., Jin F., Xiong S., Deavers M., Parant J.M., Lozano G. EWS-FLI1 induces developmental abnormalities and accelerates sarcoma formation in a transgenic mouse model. Cancer Res. 2008;68:8968–8975. doi: 10.1158/0008-5472.CAN-08-0573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu Y.P., Haasnoot J., ter Brake O., Berkhout B., Konstantinova P. Inhibition of HIV-1 by multiple siRNAs expressed from a single microRNA polycistron. Nucleic Acids Res. 2008;36:2811–2824. doi: 10.1093/nar/gkn109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moulin H.R., Seuberlich T., Bauhofer O., Bennett L.C., Tratschin J.D., Hofmann M.A., Ruggli N. Nonstructural proteins NS2–3 and NS4A of classical swine fever virus: essential features for infectious particle formation. Virology. 2007;365:376–389. doi: 10.1016/j.virol.2007.03.056. [DOI] [PubMed] [Google Scholar]
- Paton D.J., Sands J.J., Lowings J.P., Smith J.E., Ibata G., Edwards S. A proposed division of the pestivirus genus using monoclonal antibodies, supported by cross-neutralisation assays and genetic sequencing. Vet. Res. 1995;26:92–109. [PubMed] [Google Scholar]
- Ruggli N., Bird B.H., Liu L., Bauhofer O., Tratschin J.D., Hofmann M.A. N(pro) of classical swine fever virus is an antagonist of double-stranded RNA-mediated apoptosis and IFN-alpha/beta induction. Virology. 2005;340:265–276. doi: 10.1016/j.virol.2005.06.033. [DOI] [PubMed] [Google Scholar]
- Saayman S., Barichievy S., Capovilla A., Morris K.V., Arbuthnot P., Weinberg M.S. The efficacy of generating three independent anti-HIV-1 siRNAs from a single U6 RNA Pol III-expressed long hairpin RNA. PLoS One. 2008;3:e2602. doi: 10.1371/journal.pone.0002602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmidt E.E., Taylor D.S., Prigge J.R., Barnett S., Capecchi M.R. Illegitimate Cre-dependent chromosome rearrangements in transgenic mouse spermatids. Proc. Natl. Acad. Sci. USA. 2000;97:13702–13707. doi: 10.1073/pnas.240471297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sui H.Y., Zhao G.Y., Huang J.D., Jin D.Y., Yuen K.Y., Zheng B.J. Small interfering RNA targeting m2 gene induces effective and long term inhibition of influenza A virus replication. PLoS One. 2009;4:e5671. doi: 10.1371/journal.pone.0005671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tautz N., Elbers K., Stoll D., Meyers G., Thiel H.J. Serine protease of pestiviruses: determination of cleavage sites. J. Virol. 1997;71:5415–5422. doi: 10.1128/jvi.71.7.5415-5422.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tautz N., Kaiser A., Thiel H.J. NS3 serine protease of bovine viral diarrhea virus: characterization of active site residues, NS4A cofactor domain, and protease-cofactor interactions. Virology. 2000;273:351–363. doi: 10.1006/viro.2000.0425. [DOI] [PubMed] [Google Scholar]
- ter Brake O., Konstantinova P., Ceylan M., Berkhout B. Silencing of HIV-1 with RNA interference: a multiple shRNA approach. Mol. Ther. 2006;14:883–892. doi: 10.1016/j.ymthe.2006.07.007. [DOI] [PubMed] [Google Scholar]
- ter Brake O., t Hooft K., Liu Y.P., Centlivre M., von Eije K.J., Berkhout B. Lentiviral vector design for multiple shRNA expression and durable HIV-1 inhibition. Mol. Ther. 2008;16:557–564. doi: 10.1038/sj.mt.6300382. [DOI] [PubMed] [Google Scholar]
- Tu C.C., Lu Z.J., Li H.W., Yu X.L., Liu X.T., Li Y.H., Zhang H.Y., Yin Z. Phylogenetic comparison of classical swine fever virus in China. Virus Res. 2001;81:29–37. doi: 10.1016/s0168-1702(01)00366-5. [DOI] [PubMed] [Google Scholar]
- Westerhout E.M., Ooms M., Vink M., Das A.T., Berkhout B. HIV-1 can escape from RNA interference by evolving an alternative structure in its RNA genome. Nucleic Acids Res. 2005;33:796–804. doi: 10.1093/nar/gki220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu C.J., Huang H.W., Liu C.Y., Hong C.F., Chan Y.L. Inhibition of SARS-CoV replication by siRNA. Antiviral Res. 2005;65:45–48. doi: 10.1016/j.antiviral.2004.09.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu X., Guo H., Xiao C., Zha Y., Shi Z., Xia X., Tu C. In vitro inhibition of classical swine fever virus replication by siRNAs targeting Npro and NS5B genes. Antiviral Res. 2008;78:188–193. doi: 10.1016/j.antiviral.2007.12.012. [DOI] [PubMed] [Google Scholar]
- Zhang P., Xie J., Yi G., Zhang C., Zhou R. De novo RNA synthesis and homology modeling of the classical swine fever virus RNA polymerase. Virus Res. 2005;112:9–23. doi: 10.1016/j.virusres.2005.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu Y., Shi Z.X., Drew T.W., Wang Q., Qiu H.J., Guo H.C., Tu C.C. Antigenic differentiation of classical swine fever viruses in China by monoclonal antibodies. Virus Res. 2009;142:169–174. doi: 10.1016/j.virusres.2009.02.011. [DOI] [PubMed] [Google Scholar]


