Abstract
This chapter includes information about modification of animal products using biotechnology and the importance of different modifications on the natural composition. The species considered for modified products include beef and dairy cattle, sheep, goats, poultry, and a wide variety of fishes. Moreover, the discussion includes the importance of animal food, nongenetically engineered animal modified food products, genetically engineered animal modified food items primarily for meat, milk, or egg and genetically engineered animal food along the transgenic approach for animal welfare. Modern biotechnology can improve productivity, consistency, and quality of alter animal food, fiber, and medical products. The transgenic technology is potentially valuable to alter characters of economic importance in a rapid and precise way. The food safety issue related to genetic engineering is also included in this chapter. The harm of such modified food and transgenic strategy should also be understood by the reader along with its advantages. In this context, transgenic approaches in animal biotechnology are under discussion that ranges from animal food production to their adverse effects.
Keywords: Trans Fatty Acid, Transgenic Animal, Wool Fiber, Transgenic Fish, Transgenic Technology
Introduction
After domestication of animals their products have been used by man for thousands of years. Animal biotechnology has been practiced in one form or another since the beginning of the domestication of animals. Previously, the tools used for modification of desired products in animal breeding, genetics, and nutrition have played an important role in the selection, propagation, and management of desirable and economically important characteristics in livestock. Later, it developed some breeding strategies by phenotypic characters for improvement and selection of the desired traits in animals. However, the desired characters could not have been often achieved by such conventional means of breeding and selection (Madan 2005).
Industrial scale production of proteins can be offered by milk, egg white, blood, urine, and seminal plasma of transgenic animals. In 2000, the first recombinant protein expressed in transgenic animal was marketed. A number of technical problems remain to be addressed before optimization of different systems for the production of various recombinant proteins (Houdebine 2000). However, the productions of transgenic animals became quite easy due to animal cloning technologies and other advancement in transgenic animal techniques, but the cost of production of such animals is still a major limitation. Different studies on transgenic animal production were performed in the last 15 years with limited transgene expression due to inappropriate construction of expression vectors (Houdebine 2000).
Animals were initially considered a good source of biomolecule and drug production due to a number of reasons; (1) The ability of mammary glands for the production of complex molecules, (2) engineered animals can produce high amount of protein, (3) reduced per unit protein cost in animal bioreactors, (4) finally, safer and flexible source for the production of human protein, especially vital human protein and blood products. Recently, animals are considered to develop almost all types of human proteins and proteins of industrial importance (Gottlieb and Wheeler 2011). Previously, assisted reproductive technologies (ART), such as artificial insemination (AI), superovulation (SOV), embryo transfer (ET), and in vitro embryo production (IVEP), contributed to animal breeding programs for faster development of desirable traits in a shorter period of time compared to classical approaches. The use of modern transgenic technologies along with ART to develop livestock with current genomes has played an increasing vital role in the genetic improvement and development of livestock with desired genetic materials. The stem cell technologies inclusion to the genetic “toolbox” has further improved scientists’ capabilities of livestock genome desired modification and physiology (Wheeler et al. 2010).
Modern livestock production has been dependent on biotechnology for development of improved feedstuff, feed ingredients, vaccines, biological, enzymes, high quality genetics, genetic markers, and assisted reproduction. Biotechnology encompasses a variety of modern techniques including genetic engineering, genetic modification, transgenic techniques, recombinant DNA technology, and cloning to develop selected characteristics in animal food products. Some examples of these products are fat and meat content ratio, alteration of milk component, modification in hair or fiber contents, and production of targeted proteins in certain animals (Cowan 2010).
The techniques to produce genetically engineered animals have been existing for more than 20 years, but recently livestock have been produced with desirable characters to improve animal agriculture and human health (Wheeler 2003). Commercial producers are much interested in the application of modern biotechnology to improve productivity, consistency, and quality of altered animal food, fiber, and medical products. The transgenic animal technology is potentially valuable to alter characters of economic importance in a rapid and precise way. However, this technology needs the complete information and knowledge of gene that control specific characters and their regulations. The classical program of breed improvement for desired characters does not need any specific information about genes and gene regulation (Montaldo 2006; Wheeler et al. 2010).
Modification with Modern Transgenic Techniques
Modification in Fat Contents
The animal fat content, as a result of modern transgenic techniques, would be the most important future strategy to improve animal dairy and meat products for safe consumption by humans. These alterations will make such products more valuable and desirable for patients suffering from cancer or heart diseases. Moreover, these products can also reduce risk of lethal diseases such as chronic heart disease and cancer with consumption of animal product lower in bad or higher in good fat contents. Some additional benefits can also be achieved along with safe consumption of fat products of transgenic animals such as increased shelf life, or storage of animal fat to avoid rancidity and oxidation. Future development may make it possible to introduce the transgenes, knockout, and knockin with more precision. Issues related to transgenic products still need attention and further improvement is required, such as large-scale production, cost-effectiveness, GMO regulation, and consumer choice.
The exogenous fats in the human diet serve as raw material for the production of indigenous fats, cholesterol, and phospholipids. Food fats contribute a greater relation to food nutritional calories; about nine calories per gram which is almost twice the energy obtained from carbohydrate and protein. Fats are a group of chemicals that belong to fatty acids. Palmitic acid, stearic acid, and oleic acid are the most common fatty acids found in animal fats. These fats can be synthesized by the human body, but another class of essential fats that cannot be produced by the body include linoleic acid, linolenic acid, and arachidonic acid; these must be from exogenous sources along with the diet (Campbell and Reece 2002).
Naturally occurring fatty acids are of two types: saturated and unsaturated. Saturated fatty acids have no double bond between the carbon atoms, all carbon chains are entirely occupied with hydrogen. Saturated fatty acids are mainly of animal origin (Keys et al. 1965). These fatty acids are considered “bad fats” because they increase both high density lipoprotein and low-density lipoprotein cholesterol level in the body of consumers. Unsaturated fatty acids mainly originate from plants. They are categorized into two major type: monounsaturated fatty acids and polyunsaturated fatty acids. Monounsaturated fatty acids have one double bond in the carbon chain; while polyunsaturated fatty acids have two or more double bonds in the carbon chain. These fatty acid are considered “good fats” because they only increase the level of high density lipoproteins, while they decrease the level of low-density lipoproteins (Keys et al. 1965; Grundy 1986; National Research Council 1988).
Dairy milk has almost 60 % of saturated fats, as a consequence, decrease in the intake of dairy products has been observed worldwide. These saturated fats increase the plasma cholesterol level, as a result, an increased risk of coronary heart diseases (CHD) (Parodi 2004). Milk fat has a number of functional components and bioactive compounds. These functional components play a vital role in development of many diseases. It is now known fact that persons with high risk of heart-related diseases and high risk of prostate, colon and breast cancer are those who consume diet with high quantity of fatty acids, especially saturated fatty acid (Parodi 2004; Jube and Borthakur 2006). The sex, breed, and age of an animal affect the fatty acid composition but nutrition has a direct effect on the fatty acid composition in animal products. Previously, pork meat was produced with increased amounts of omega-3 with the use of 11 % flaxseeds in the diet of pork (Mavromichalis 2001).
The increase in unsaturated fatty acids components in animal products increase the risks of rancidity and reduces the shelf life of such products due to oxidation (Decker and Xu 1998). These types of undesirable sensory and health effects due to lipid oxidation can also be avoided or reduced with vitamin E supplementation in the animal diet. The animal products supplemented with vitamin E enriched diets demonstrate antioxidant activity and reduce rancidity to maintain the natural color of meat. Experimentally, beef and chicken meat and egg compositions have been altered with different feeding strategies (Decker and Xu 1998; Pszczola 1998; Sloan 2000).
Previous investigations suggest that the risk of cancers and CHD can be reduced with the modification of daily diet consumption (Parodi 2004). Health agencies including the American Diabetes Association, the American Dietetic Association, and the American Heart Association suggest that fatty acids should not contribute more than 30 % of the total daily calories consumption (Parodi 2004; Jube and Borthakur 2006). Some other studies also suggest that the risk of these diseases can also be reduced through consumption of plant rich foods. Milk saturated fatty acids are usually considered more harmful for human health. During the past decade, a lot of evidence came into consideration that many animal milk derived fatty acids can offer important human health benefits for chronic heart disease and cancer (Parodi 2004).
The expression of stearoyl-CoA desaturase in goat mammary gland and Caenorhabditis elegans Δ- and Ω-3 desaturase genes in transgenic mice produced milk enriched with linoleic acid contents. These approaches may also help to produce milk from transgenic animals with such value addition to reduce the cardiovascular diseases caused by milk fat. Modified milk of transgenic animals with secretion of Omega-3 lipid is another possibility to reduce the cardiovascular diseases with consumption of dairy milk (Soler et al. 2006).
Dairy milk contains approximately 3–4 % fat, depending on breed, nutrition, and stage of lactation. As previously mentioned, milk fats may increase the risk of cardiovascular disorders in humans (Soler et al. 2006). The change in major enzymes for fat production can modify the production of fat in the milk. The acetyl coenzyme A (CoA) carboxylase is a major enzyme responsible for de novo synthesis of milk fat from acetate conversion inside the mammary gland. Almost 50 % of milk fat is synthesized by this method and modification in this enzyme can lead to alter the milk fat contents. The reduction in the quantity of this enzyme would lead to extreme reduction in the amount of fatty acids in milk. Similarly, it would reduce the requirement of dietary nutrition for the production of fatty acids in milk. However, the alteration in the α-lactalbumin would also be another strategy for modification in milk fat (Yom and Bremel 1993).
The composition of animal fat in red meat can also be managed using animal husbandry practices. The meat or beef fat composition would differ in animals feeding on feedlots and animal feeding on pasture. Fat composition depends on the genetics, nutrition of animals, meat cuts, and fat trimming influences (Scollan et al. 2006). In animals, the fat is deposited and localized intramuscularly (marbling) and subcutaneously (Scollan et al. 2006). Oleic acid contributes the most prominent part of the fatty acids present in pig, sheep, and cattle meat, while palmitic acid is the most predominant saturated fatty acid (Scollan et al. 2006). Poultry rearing in free range systems does not produce meat with high fat value, while the poultry with high energy diet can achieve a significant level of fat contents. Breast meat of free range broiler has high poly unsaturated fatty acids (PUFAs) and mono unsaturated fatty acids (MUFAs) content, while it is low in saturated fatty acids (SFAs) (Ponte et al. 2008). Poultry skin has more than 40 % fat, while dark meat has 10 %, and white meat has 4 % fat. Poultry meat contains 21 % PUFAs, 45 % MUFAs, and 30 % SFAs (Sayed et al. 1999; USDA 2008).
Conjugated linoleic acid is a naturally occurring fatty acid in animal products such as meat and dairy milk. The conjugated linoleic acid in milk has been recorded up to 2–37 mg g−1 , while cis-9 and trans-11 conjugated linoleic acid contents have also been recorded up to 53.7 mg g−1 of fatty acid and 51.5 mg g−1 of total milk fat (Javor et al. 2008). The concentration of conjugated linoleic acid in animal products has been improved with feed and diet such as fresh pasture, total mix ration contain fish oil or sunflower as feed additive. Furthermore, the breed, age, and lactation number also have little influence on the concentration of conjugated linoleic acid. Interestingly, the conjugated linoleic acid concentration improved after cooking or processing (Javor et al. 2008). Most recent significant discovery of animal fats and fatty acids is related to the conjugated linoleic acid (CLA) in association with ruminant milk fats and meat. It has also forced authorities to reconsider the recommendations related to impact of animal fat on public health (Javor et al.2008). Linoleic acid is also a powerful naturally occurring anticarcinogen, antiatherogenic, and immunomodulatory. The National Academy of Science report on carcinogens and anticarcinogens in food declared linoleic acid as the only anticarcinogen fatty acid present in food (Javor et al.2008). In some previous studies, conjugated linoleic acid has successfully suppressed fore-stomach tumors in mice, aberrant cryptic colonic foci in rats, and rat mammary tumors (Javor et al. 2008). Many other saturated fatty acids are neutral inside the human body and do not increase the plasma cholesterol level. Moreover, it has a significant role to alter body fat composition and as a weight reducing agent with reduction in animal fat deposition in body by increasing the rate of lipolysis in adipocytes (National Research Council 1996; Javor et al. 2008).
Moreover, a number of saturated fatty acids present in milk have advantageous effects such as conjugated linoleic acid, vaccenic acid, sphingolipids, butyric acid, 13-methyltetradecanoic acid, and ether lipids which are mostly anticarcinogenic. Conjugated linoleic acid, oleic acid, omega-3 fatty acids are useful to avoid cardiovascular diseases. Conjugated linoleic acid is also very useful to improve the immune response and bone health (National Research Council 1996). However, dairy fats that include lauric, myristic, palmitic acid in human diet increase the blood plasma cholesterol, low and high density lipoprotein cholesterol level (Kris-Etherton and Yu 1997; National Research Council 1996). The transgenic approach to alter such fat components of dairy milk would increase the acceptability of milk along with reduction in the risk of chronic heart diseases (CHD) to consumers.
Normally, unsaturated fats present in diet are of cis-configuration but at the same time they are of trans-configuration. Trans fatty acids are involved in raising the blood cholesterol level. Diets containing trans fatty acids increase the risk of CHD. Fatty acid receives trans double bonds during chemical processing or partial dehydrogenation of vegetable oil. Animal products have trans double bond during rumen biodegradation and formation of intermediates, which is a natural source for generation of trans fatty acids. Unsaturated fatty acids are toxic to rumen microorganisms. The microorganisms carried out massive biodegradation, and as a result, produced 1–8 % trans fatty acids to the total rumen lipids. The major natural sources of trans fatty acids are ruminant meat and dairy products (Lock et al. 2005).
The trans fatty acids-related risk of CHD needs more investigation; but the trans fatty acids of natural or animal origin differ with industrial trans fatty acids with respect to chronic heart diseases. It may be due to the difference in specific amino acids and trans fatty acids isomers such as trans 9 and trans 10 fatty acids that mainly exist in industrial sources. The most important reason of this difference may be due to conversion of vaccenic acid (11-trans octadecenoic acid; VA) a major trans fatty acid in the fat of ruminants to rumenic acid (9-cis, 11-trans octadecenoic acid; RA) in tissues, an isomer of conjugated linoleic acid, by Δ9-desaturase. The human body can also convert this vaccenic acid to rumenic acid (Palmquist et al. 2005).
Studies have investigated the role of milk trans fatty acids on the blood plasma cholesterol. But interestingly, naturally enriched trans fatty acids animal fats have no undesirable effect on the blood plasma cholesterol even at very high intake. Vaccenic acid (VA) to rumenic acid (RA) conversion has potential benefits to control cardiovascular diseases and to reduce the adverse effects of trans fatty acids on human plasma cholesterol level. However, this topic needs more investigation, especially to improve human health and to enhance the use of animal natural fats (Lock 2007). Moreover, the transgenic approaches in animals to improve all adverse effects of fatty acid with substitution of useful fatty acids would be a good strategy to improve the health of consumers and consumption of animal dairy products.
Genetic manipulation in transgenic animals offers to alter the carcass lower in fat and cholesterol. In addition, modern biotechnological techniques make this possible to put useful fatty acids in transgenic animals such as polyunsaturated fatty acids. Polyunsaturated fatty acids (PUAFs) contain 18 or more than 18 carbon atoms; and two or more than two double bonds. PUAFs two major groups are omega-6 (n-6) and omega-3 (n-3), on the basis of double bond near the methyl end of fatty acid. Polyunsaturated fatty acids n-3 and n-6 are important constituents of phospholipids in the tissues throughout the body. PUFAs n-3 is especially enriched in the retina and brain (DeFilippis and Sperling 2006; Simopoulos 2006). Mammals lacking in desaturase require the synthesis of linolenic acid (n-6) and alpha linolenic acid (n-3). Omega-3 fatty acid desaturase is also absent in mammals, as a result, n-3 and n-6 polyunsaturated fatty acids are not interconvertible in mammals. Polyunsaturated fatty acid n-3 and n-6 are essential fatty acids for mammals, which must be taken along with diet. The consumption of n-6 polyunsaturated fatty acid increased during the last one and a half century due to the use of vegetable oils of corn, sunflower seeds, cottonseed, and soybeans origin. The vegetable oil from such sources is much enriched with n-6 polyunsaturated fatty acids, but n-3 polyunsaturated fatty acids are deficient in these sources. Recently, the consumption of n-3 poly unsaturated fatty acids is much lower due to less consumption of fish meat; and consumption of meat and egg produced from commercial feed enriched with n-6 polyunsaturated fatty acids, which is deprived of n-3 polyunsaturated fatty acids (Simopoulos 2006). Previously, many potential useful effects of Omega-3 have been discovered and studied in many populations, large animals, primates, and swine. The major beneficial effects include antiatherosclerotic, reduction in the risk of cardiovascular diseases by reducing the triacylglycerol, cholesterol, and VLDL concentration; reduction in many inflammatory markers such as CRP, IL-6, E-selectin, ICAM-1, VCAM, 1IL-1β, and TNF-α; reduction in platelet accumulation and thrombosis, downregulation of PDGF and triggered mononuclear cells; preventing ventricular arrhythmias (Prather et al. 2008).
In previous studies, omega-3 polyunsaturated fatty acids desaturase gene, also called fat 1, has been successfully introduced in mice from a roundworm called Caenorhabditis elegans (reference). The product of this gene can convert n-6 into n-3 polyunsaturated fatty acids. Some later investigations revealed resistance in these transgenic mice to certain diseases such as colitis, chemically induced hepatitis, prostate cancer, and melanoma (Prather et al. 2008). Human fat 1 gene was introduced into pig with somatic cell nuclear transformation, in a result, threefold increase the level of n-3 polyunsaturated fatty acids along with 25 % decrease in n-6 polyunsaturated fatty acids by the efficient conversion of n-6 into n-3 polyunsaturated fatty acids (Lai et al. 2006; Li et al. 2006). This conversion reduced the n-6/n3 up to five folds in these transgenic pigs compared to control or wild pigs. The reduction of this ration of n-6/n-3 was observed in almost all tissues of transgenic pig in contrast to control animal (Lai et al. 2006; Li et al. 2006). Transgenic pigs were developed with plant gene insert (fatty acid desaturation 2 gene) for 12 fatty acid desaturase from spinach. In transgenic animal linoleic acid (18: 2n-6) in adipocytes was ten times more and white adipose tissue contained 20 % more linoleic acid than wild-type pig (Saeki et al. 2004). Previous findings such as expression of fatty acid desaturase gene insertion in mammals made this possible to alter the fatty acid compositions in domestic animals with modern recombinant biotechnology for production of more useful fats of animal origin for human consumption (Montaldo 2006). Bucher et al. (2002) suggested in 11 randomized meta analysis that the intake of omega-3 reduced mortality due to myocardial infarction and death due to chronic heart disease. The people consumption of omega-3 is 8–10 % of the recommended daily intake, while of omega-6 is 1000–2000 %. This imbalance can be improved with increased intake of omega-3 enrich diet such as fishes, salmons, eel, herring, sardines, omega-3 rich eggs of hens with modified diet. The omega-3 containing transgenic approaches are under consideration of many commercial companies (Lewis et al. 2000; Pulina et al. 2006). Scheeder et al. (2001) produced cow feed rich with omega-3 without any unpleasant effect on meat texture. Furthermore, omega-3 fatty acids gene can be introduced from fish or other sources to livestock to make transgenic animals rich in omega-3. Transgenic animals with omega-3 transgene or the transgene to convert omega-6 to omega-3 would produce valuable diets to balance the omega-6 in daily intake (Wheeler 2012).
Potential other genes can also be targeted from cholesterol and fat biosynthesis pathways to develop transgenic animal for modification in fat contents such as cholesterol 7-alpha hydroxylase, hydroxymethylglutaryl coenzyme A (HMG-CoA) reductase, fatty acid synthase, and lipoprotein lipase. Furthermore, leptin hormone gene and the low-density lipoprotein (LDL) receptor gene can be possible targets in transgenic approaches. The products of these transgenes could decrease fat and cholesterol in dairy and other products of transgenic animals (Wheeler 2012).
Cholesterol 7-alpha hydroxylase (CYP7A1) protein controls the pathway for conversion of cholesterol to bile. Bile acids have a significant role in the digestion of fat soluble vitamins and triglycerides. In addition, bile acids provide a metabolic pathway to reduce excessive cholesterol from the body. Bile acids also have some toxic effects such as farnesoid X receptor (FXR)/small heterodimer partner (SHR) dependent and independent reduction in the expression of cholesterol 7-alpha hydroxylase (CYP7A1) gene (Davis et al. 2002). In a recent study, transgenic C57BL/6 mice produced with CYP7A1 expression in the liver increased fivefold the enzyme activity and twice the bile acid pool size. Instead of toxic effects, the transgenic mice with overexpression of CYP7A1 in the liver clogged the diet-mediated atherosclerosis and gallstone formation. These studies propose that CYP7A1 may be a better therapeutic target in the future to control these diseases (Davis et al. 2002).
Similarly, hydroxymethylglutaryl coenzyme A (HMG-CoA) reductase is microsomal enzyme in the de novo synthesis of cholesterol in liver. More than 50 % of the cholesterol in humans comes from de novo synthesis in liver. The cholesterol level in the body can be modified with alteration in the expression of HMG-CoA gene. Furthermore, fatty acid synthase is a vital dimer protein in fatty acid synthesis. The dimer subunits alter confirmation at two acyl carrier protein center to generate fatty acids. The malonyl-CoA in mammals to palmitate conversion can be catalyzed with this single dimer multifunctional protein, which can be a better target in transgenic approach to modify the fat content in animal products (Smith et al. 2003).
A single lipoprotein lipase (LPL) gene is also a better target for production of transgenic animals with modified fat components. Lipoprotein lipase is also an important protein in mammals for fat metabolism and transportation. Lipoprotein lipase can catalyze the hydrolysis of fatty acids components of circulating chylomicrons and LDL. This catalysis provides nonesterified triglycerides and 2-monoacyl glycerol for tissue consumption. Previous studies reveal that this protein can bind simultaneously to low-density lipoprotein and cell surface receptors such as proteoglycans, which enhances the intracellular uptake of low-density lipoprotein with non-catalytic bridge function. Abnormal expression of LPL and function is directly or indirectly associated with physiological and pathological consequences such as diabetes, Alzheimer’s disease, atherosclerosis, obesity, chylomicronemia, and cachexia (Mead et al. 2002).
Previously, many other studies were conducted and successfully produced transgenic animals with lower in fat contents. Ward et al. (1989) developed transgenic lamb with ovine GH insert; the transgene insert was successfully expressed and controlled by ovine metallothionein IA (oMT-IA) promoter in transgenic animals. Transgenic animal reduced fat components up to five times compared to control animals. Similarly, Pursel et al. (1989) developed transgenic pigs with human GH and bovine GH gene inserts, which successfully produced transgenic animals with reduction in carcass fat but with little effect on growth. The transgene expression in transgenic animals was controlled with mouse metallothionein IA (MT-I) promotor. Wieghart et al. (1990) developed transgenic pig with bovine GH gene insertion and expression was controlled with rat phosphoenolpyruvate carboxykinase (PEPCK) promoter. The back fat depth was reduced by up to 41 % in the transgenic pig.
Modification in Animals for Meat and Carcass
The development of transgenic animals using modern biotechnology has been put great insight into the action of gene regulation to control growth and development. This technology made it possible to manipulate and evaluate the different growth factor receptors, growth factors, and growth modulators (Rexroad et al. 1991; Seidel 1999). Initially, scientists put great emphasis on the production of livestock with simple or transgenic approach to alter carcass composition for meat purpose with direct or foreign gene incorporation.
Previously, metabolic modifiers were also used to increase productivity of animals such as modification of carcass composition (meat-fat ratio), increase in milk yield, and decrease in animal fat. Most common modifiers are hormones of biological origin. The recombinant bovine somatotropin (rBST) with modern biotechnological tool in dairy cows has been successfully used to increase both milk production efficiency and decrease animal fat. The rBST typically increased milk yield by 10–15 %. However, this increase is not significant because of low milk yields and the high cost–benefit ratio. Recently, rBST is used commercially in 19 different countries where the economic returns make its use worthwhile. A porcine somatotropin has been developed and used to increase muscle ratio over fat deposition, which is produced in transgenic pigs at greater market value (Wheeler et al. 2010).
Transgenic mice have been produced in different studies with tremendous increase in growth characteristics. Previously, the transgenic mice have developed successfully for transgene expression for growth factors such as growth hormone (GH), growth hormone releasing factor (GRF), and insulin-like growth factor I (IGF-I) (Palmiter et al. 1982, 1983; Hammer et al. 1885; Mathews et al. 1988). Further successes along the lines of mice and sheep were achieved with independent expression of the foreign genes encoding growth hormone-releasing factor (GRF) or insulin-like growth factor I (IGF-I) (Cameron et al. 1994; Murray et al. 1999; Montaldo 2006). Details of some important studies related to transgenic technology for the improvement in meat and growth are given in Table 4.1.
Table 4.1.
Transgenic technology application to improve meat production and growth rate (Modified from Wall et al. 2009)
| Introduced | Modification | Application species | References |
|---|---|---|---|
| Insulin-like growth factor 1 | Increased meat production | Pig | Pursel et al. (1999) |
| Human and porcine growth hormone releasing factor | Increased meat production | Pig | Draghia-Akli et al. (1999), Pursel et al. (1990) |
| Human growth hormone releasing factor | Increased meat production | Pig | Rexroad et al. (1989) |
| Bovine, human, and porcine growth hormone | Increased meat production | Sheep | Nottle et al. (1999), Pursel et al. (1989, 1990) |
| Ovine growth hormone | Increased meat production | Pig | Adams et al. (2002); Ward and Brown (1998) |
| Inducible myostatin knock out | Increased postnatal muscle growth | Mouse | Grobet et al. (2003) |
| Myostatin disruption | Increased meat production | Mouse | Yang et al. (2001) |
| Sex-specific disruption of myostatin | Efficient cattle production system for dairy cows and superior beef bulls | Mouse | Pirottin et al. (2005) |
| Chicken c-ski oncogene | Primarily involve in hypertrophy of muscles | Mice, sheep, pig and cattle | Palmiter et al. (1982), Pursel et al. (1990), Bowen et al. (1994), Cameron et al. (1994), Murray et al. (1999) |
| Growth hormone releasing factors and insulin-like growth factor | Increase the diameter of muscles | Pig | Neimann (1998) |
| Piscine growth hormone | Shorter time to market | Fish | Devlin et al. (1994), Du et al. (1992) |
In some other transgenic animal trials, growth hormone expression was effectively increased in domestic animals such as pig, sheep, and rabbit (Prather et al. 2008). A number of genes have been used to make transgenic pig for growth and modified carcass products such as growth hormone (GH1), insulin-like growth factor 1 (IGF1), delta 12 fatty acid desaturase, B-cell lymphoma 2 (Bcl-2), omega-3 desaturase (hfat-1), and bovine alpha-lactalbumin (LALBA), phytase (Prather et al. 2008). These modifications improved the body weight and growth rate by increasing the meat quantity, while some modifications only altered the meat composition such as delta 12 fatty acid desaturase and omega-3 desaturase (hfat-1) (Prather et al. 2008). Pig was a suitable candidate for production of modified body composition with increase in carcass quantity. In the first study on transgenic pigs, Pursel et al. (1989) successfully developed transgenic pigs with unsatisfied results. The growth hormone (GH) transgene was inserted in transgenic pig, and little effect was achieved on the growth. The transcription was not efficiently regulated in these transgenic pigs ‘as a result’ the high level of growth hormone expression produced side effects such as lameness, reduction in fertility, and vulnerability to stress (Pursel and Rexroad 1993). Later, “super pigs” were also developed with ovine growth hormone (oGH) transgene into pig. The transgenic pigs clearly gained more muscle content compared to wild or nontransgenic pigs. Unfortunately, super pigs suffered from arthritis and bone thickness (Pursel et al. 1997).
High level of serum growth hormone was observed in transgenic pigs and sheep with GRF and IGF-I, which incremented with increase in protein diet. However, transgenic animals reduce fat content on body but no serious effect was observed on the growth rate of the animals. Additionally, transgenic animals developed pathological conditions such as severe reduction in reproduction (Murray et al. 1999). In two other studies, transgenic pigs were produced with IGF-I and GRF, which expressed and incremented the IGF-I and growth hormone level. Transgenic pigs developed increased diameter of muscles and reduced fat contents without any serious undesirable or pathological effects (Neimann 1998). Some other genes were also attempted in the development of transgenic pig to change the meat or muscle growth along with metallothionein promoter to control their expressions. A chicken oncogene “c-ski” was introduced into transgenic sheep, mice, pigs, and cattle. This gene is primarily involved in hypertrophy of several muscles, while decreasing the fat deposition on the body. This approach has resulted in partial success although muscular hypertrophy has been obtained in a few pigs and cattle (Palmiter et al. 1982; Cameron et al. 1994; Murray et al. 1999; Wheeler 2012).
The acid meat gene or Rendement Napole gene has been found associated with lower processing output in many Hampshire pig lines and crossbreds. Pork with low pH is produced in these lines and can be distinguished in different qualities such as firmness, processing yield, shearing force, marbling, color, and water holding capacity. Modern biotechnological tools such as “knockout technology” may be a better approach to modify the postmortem pH, glycolytic potential, and pork meat quality (Wheeler 2012). The growth pattern can also be affected by other loci such as ryanodine receptor, myo-D, growth hormone release factor, sheep callipyge, high affinity insulin-like growth factor binding proteins, and myostatin gene (Wheeler 2012). The differentiation factor 8 (DF-8) or myostatin “a member of transforming growth factor β (TGF-β) family” gene has been found to have high suitable locus for the development of transgenic animals for the increase in carcass.
Some cattle breeds such as Belgian Blue and Piedmontese developed double muscle mass as a result of mutation in myostatin gene. The myostatin gene is highly conserved in other species of animals such as human, ovine, rat, murine, zebrafish, chicken, and turkey, which may also suggest to conserve functional role. Myostatin knockout mice has been developed with increased lean muscles mass, which enlarged the hip and shoulder of transgenic mice. Moreover, transgenic mice achieved incremental skeletal muscles mass all over the body without any gross abnormality. The homozygous animals of such knockout animals have achieved 2–3 times more muscle weight and 30 % more body weight compared to normal animals. This increase in the muscular growth was due to hyperplasia of muscles (Mcpherron et al. 1997). The myostatin knockout animals have some adverse effects on reproduction on transgenic animals such as dystocia. The animal welfare concern forced the researchers to overcome this health issue with postnatal inhibition of myostatin gene in transgenic animal (Grobet et al. 2003).
The prodomain of myostatin could bind to mature myostatin, which would inhibit biological activity of myostatin (Yang et al. 2001). The increase in muscle growth and body weight has been observed in experimental mouse with transgene over expression of prodomain segment. The transgenic mouse with overexpression of prodomain segment presented 17–30 % more body weight and 22–44 % total carcass weight compared to wild or control animal. The pMEX-NMCS2 vector with a rat myosin light chain 1 (MLC1) promoter, an SV40 polyadenylation sequence, and an MLC enhancer were used for overexpression of prodomain segment into transgenic animal. In addition, the transgenic animals were not presented any defective phenotype, reproductive, and other undesirable health effects (Yang et al. 2001). The RNAi is a more suitable tool for inhibition of myostatin pathway. The site-specific RNAi molecule could provide more flexible, controlled, and site directed inhibition of this gene expression (Yang et al. 2001). Pirottin et al. (2005) has developed transgenic mice with site-specific Y-chromosome associated expression of such competitive inhibitor molecules, which successfully produced 5–20 % more carcass weight or skeleton muscles in male animals. The females were lacking transgene, transgene expression, and also growth characteristics due to absence of Y-chromosome. The inducible or postnatal expression combine approach would be more suitable to make transgenic bull for meat and elite dairy cow simultaneously. Moreover, the sex-sorting technology would enhance the application of this approach, because only bull contain such transgene and expression (Wall et al. 2009).
There are many important genes associated with growth rate, growth factors, growth receptor, and modulators, which can be considered for the future animal transgenic approach for increased growth rate and feed conversion efficiency. Indirectly, muscle growth or carcass can be achieved with cloning of different genes to make transgenic animals for fat reduction such as hydroxymethylglutaryl Coenzyme A (HMG-CoA) reductase, lipoprotein lipase, lipoprotein synthetase and cholesterol 7-alpha hydroxlyase (Wheeler 2012). Previously, the transgenic pigs were also made with certain transgenes such as Bcl-2 and LALBA to gain indirect carcass benefits. The transgenic pig with Bcl-2 transgene produced more number of viable eggs in females, and transgenic pig with LALBA transgene made possible to massive increase in weaning weight (Prather et al. 2008). Indirectly, more pork could be produced with more number of animals and increased body weight at time of weaning or marketing.
Certainly, many potential target genes can also be used to increase the feed efficiency, feed digestion, and appetite to animals. The increase in feed efficiency and appetite has a profound effect on the animal production for meat purpose. The alteration in digestive enzymes profile can increase the feed efficiency and nutrient up take from the digestive tract. The phosphorous bioavailability can be increased from phytic acid in corn and soybean feed with introduction of digestive enzyme such as xylanase phytase transgene into digestive system of other species such as pig and poultry, which naturally lack this enzyme in their digestive systems (Wheeler 2012).
The enzyme isocitrate lyase glyoxylate cycle can catalyze the conversion of isocitrate to citrate and glyoxylate. The isocitrate lyase and malate synthase are the enzyme of glucose metabolism in bacteria, fungi, and plants, while the placental mammals missing these enzymes. As result of combine effects of both enzymes solicit to evade the two carboxylation process in tricarboxylic acid cycle. The glyoxylate cycle generates intermediates for the glucose synthesis and other biosynthetic precursors. In addition, greater expression of isocitrate lyase in a microorganism such as fungus could increase the pathogenicity of disease in plant, animals, and humans (Dunn et al. 2009).
In higher plants, these enzymes have a most significant role in germination of plants from oil seeds, which provide the acetyl-CoA for glyoxylate cycle to deliver the primary nutrients and other metabolic intermediate prior to photosynthesis (Eastmond and Graham 2001). The organisms could produce the acetyl-CoA derived glucose and other metabolic intermediates from energy sources such as acetate, fatty acids or poly-β-hydroxybutyrate, and ethanol (Dunn et al. 2009). Ward (2000) has been reported sheep with transgene expression of bacterial isocitrate lyase and malate synthase. The expression of both genes in transgenic sheep the successful expression of these transgenes solicited to increase the glucose supply, better feed conversion into energy, and indirectly better growth rate. The transgenic pig has been produced with phytase transgene. The phytase expression was detected in saliva at day 7 of transgenic pig, the salivary phytase made this possible to digest the phytate completely in the diet of transgenic animals. Furthermore, this alteration reduced the need for inorganic phosphorous supplementation and environmental pollution due to animal production (Golovan 2001).
Cellulose is a major component in plant materials and foods. Ruminants have cellulolytic enzyme for degradation and consumption of plant food. Mono stomach or nonruminants are deficient in such enzyme and are incapable to digest plant food. The transgenic nonruminants with transgenes of cellulolytic enzymes and successful expression into digestive system could allow these animals to digest the plant material with cellulose. Fibrous feedstuff could be more frequently used for nonruminants such as poultry and pigs for livestock production. In addition, the competition could be reduced between human and animals for grains and cereals as food. These would be beneficial in the future in the time of more food competition and shortage (Wheeler 2012).
Increased carcass growth and quality would be solicited to overcome the future food shortage challenges. The control of undesirable effects in genetic modification needs more work of researchers for production of meat for safe human consumption. Similarly, commercial producers of meat have great advantage with rapid growth of transgenic animals in rearing to reduce the cost of production. In addition, transgenes with additional properties of animal welfare such as harboring harsh environment would encourage economical and more sustainable farming in different regions.
Genetically Modified Fish for Increased Growth Rate and Other Traits
Fish are considered the healthiest food for human consumption, and they are good candidates to be the first transgenic animal to be approved. The release of transgenic animal in wild condition could be a good option, but a reliable technique for 100 % sterilization of transgenic fish has not yet been established (Jube and Borthakur 2006). However, the transgenic technology solicited to develop large size fish with more than 11 times as control. In addition, transgenic fish has no major side effects and health problems. These “super fish” are more resistant to disease, more adaptable to cold water, more sustainable in the wild condition compared to nontransgenic fish (Devlin et al. 1994; Du et al. 1992).
The additional characteristics along with growth rate can also be achieved such as antifreeze protein expression into plasma of transgenic fish. The ocean pout (Macrozoarces americanus) and winter flounder (Pleuronectes americanus) has been previously developed with such proteins and successfully tolerated the freezing temperature of Atlantic Ocean (Davies and Hew 1990). The antifreezing proteins are characterized into two major types such as antifreeze proteins (AFPs) and antifreeze glycoproteins (AFGs). Antifreezing proteins and antifreezing glycoprotein could lower the freezing temperature of fish serum by attaching to ice surface, to avoid crystal formation. Four type of AFPs (AFP I, AFPII, AFP III, AFP IV) has been discovered, while only one type of AFGs has been identified (Davies and Hew 1990; Jube and Borthakur 2006). The species of fish with aquaculture importance, such as tilapia and Atlantic salmon, do not survive in the freezing temperature especially in Northern Atlantic coast regions. Therefore, the cage forming of such species are not possible in these regions of subzero temperature. The production and use of transgenic fish, especially salmon with antifreezing protein transgenes would expand the fish farming in the regions of subzero temperature along with reduction farming cost and price for consumers (Jube and Borthakur 2006). The flounder AFPs belongs to AFPs type I with two isoforms such as skin type and liver type. Skin type AFPs intracellular protein produced into several peripheral tissues in the form of mature protein, while liver AFPs produce in liver in immature form and need further modification before release (Hew et al. 1986; Jube and Borthakur 2006). Hew et al. (1999) reported transgenic salmons (Salmo salar) to introduce piscine antifreeze protein transgene, which produced liver-specific winter seasonal antifreeze protein. Initially, a single copy of liver type AFPs was injected into fertilized salmon egg, and a stable expression of antifreezing protein was achieved. The consistent expression was observed up to three generation with low concentration of antifreeze protein (250 μg/ml), which was normally present up to 10–20 mg/ml in winter flounders. This expression was insufficient to harbor the freezing temperature in transgenic salmon. Later, the high concentration was achieved with increase in transgenes copy number. This modification made capable to transgenic salmon in successfully harboring the winter freezing temperature. Similar study was performed on goldfish (Carassius auratus), and transgenic goldfish was successfully harboring the freezing water due to inference of piscine antifreeze protein transgene expression (Wang et al. 1995). Previously, the transgenic tilapia fish (Oreochromis niloticus) have been established with several salmonids genes constructs, but better results achieved with the Chinook salmon GH gene under the control expression of ocean pout antifreeze promoter (Rahman et al. 1998). The insertions of transgenes were achieved with fertilized egg cytoplasmic microinjection technique. The researchers achieved a successful genomic integration of transgene into founder (G0) tilapia, as well as in subsequent generations G1 and G2 of transgenic tilapia. Chinook salmon GH transgene expression into transgenic fish presented three times more growth rate compared to wild tilapia and 33 % more feed conversion ratio (Rahman et al. 1998). The farmer would be benefited with more body weight and lower feed consumption. In addition, the expression of this gene would also cause reproductive infertility, which is high desirable trait for the environmental release of transgenic fish (Rahman et al. 1998).
Human glucose transporter 1 protein is encoded by SLC2A1 gene in human and first categorized transporter protein. It belongs to solute carrier protein family 2, which is involved in glucose transport across the cell plasma membrane (Mueckler et al. 1985). The constant glucose supply from blood plasma is required to erythrocyte for energy yielding. The human glucose transporter 1 protein increases the glucose supply to erythrocytes with facilitated diffusion, which is almost 50,000 times higher than translated transmembrane diffusion (Nelson and Cox 2008; Montel-hangen et al. 2008). The expression of human transporter 1 would increase with decrease in glucose level in blood and decrease with increase glucose supply. This transporter protein is very important for the uptake of basal glucose at very low level of glucose for sustainable respiration in all cells (Montel-hangen et al. 2008). In addition, this transported protein is also responsible for the vitamin C transport along with glucose supply in nonvitamin C producing mammals (Montel-hangen et al. 2008).
Hexokinase is an enzyme for the phosphorylation of hexoses and converting into hexoses phosphate. The most commonly glucose is converted into glucose-6-phasphate, and the genes encoded hexokinase are found in different species domains such as bacteria, plants, animals, and human. Hexokinase II and glucose transporter protein 4 are most significant protein isoforms involved in the glucose phosphorylation and transport in glucose sensitive tissues such as heart, skeleton muscles, and adipose tissues (Burcelin et al. 1993). Hexokinase I and II can be associated with mitochondria outside of exterior membrane with specific binding of porin or voltage dependent anion channel. Mitochondrial attachments make this possible to gain ATP directly after synthesis, which is one of the two substrates for hexokinase. The tumor cell have almost 200 times higher or elevated level of hexokinases, compared to normal cells (Bustamante and Pedersen 1977). Krasnov et al. (1999a, b) has been reported the transgenic fishes with transgenes human glucose transporter 1 (GLUT 1) and rat hexokinase II. The successful expression of both transgenes improved glucose consumption in transgenic fish, which indirectly solicited in growth and performance of transgenic fish compared to control or wild animals. Similarly, many other enzymes genes from the glucose metabolism and other pathways may be future strategies to improve productivity of farm animals.
Transgenic approaches in fish with growth-related transgene have been more applicable to increase growth rate of nondomestic fish species, such as salmon, while less applicable to domestic species of fish, such as trout, already selected on base of growth rate. Some hurdles related to transgenic fish massive production and consumption may be included; (1) effect on wild population of fish (2) doubts related to health effects, and (3) whether these will be available soon. The creation and release of animals into environments have two constrains; (1) the issues related to release of such genetically modified organism and (2) controlled production of the transgene expression, which need the production of more complex constructs (Montaldo 2006). Because some studies has not revealed consistent results related to the effect of growth. However, the safe genetic modification in fish such as lacking any undesirable effect during wild culturing on other population would be the next challenge for the researchers. Furthermore, the genetic modification in aquaculture for desirable trait such as medicinal and nutritional benefits would also be under the consideration of researchers.
Modification in Milk Contents
Milk is almost considered a perfect food in many societies due to its balanced protein, fat, carbohydrates, mineral and ash contents. Milk has been used for the nourishment of suckling animals for a long time due to complete and hygienic source of food. The ruminant milk and dairy products have been accepted as significant human foods from as early as 4000 B.C. The dairy industry has made tremendous progress over the years and develops a variety of new dairy products for human use (Lock 2007; National Research Council 2002). Moreover, dairy industry helps to meet the dietary requirements of human beings for energy, great quality of desire proteins, minerals and vitamins. In the last quarter-century, the nutritional quality of food has become increasingly important as a result of emerging buyer awareness related to strong relation of food and health. Many dairy foods have been identified or developed that contain specific components with potential benefit to human being along with traditional nourishment (Karatzas 2003; Lock 2007).
Cow milk constitutes 88 % water, 4.7 % carbohydrate, 3 % fat, 3.3 % protein, and 0.7 % ash, which contains mineral and vitamin contents. Recent research has illustrated that milk comprises a variety of bioactive constituents such as protein, peptides, and fatty acids. The growth and quality of health in newborns can be improved by increasing the level of any nutrient major nutrient such as protein, fat, or lactose. Moreover, fermented milk products with desirable traits have the potential to elicit addition health benefits (Lock 2007) (Fig. 4.1).
Fig. 4.1.
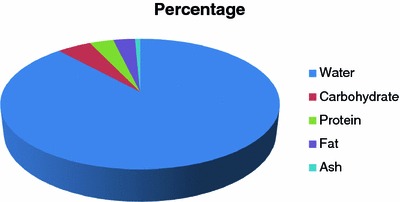
Composition of cow milk (Lock 2007)
Bovine caseins can bind to bivalent cations due to phosphorylation; so this milk is a good source of dietary calcium. Caseins can bind other bivalent cations such as magnesium, iron, and zinc more in bovine milk than caseins in human milk. Casein in bovine milk binds almost 65 % of calcium, while human milk binds only 5 % calcium (Karatzas and Jeffrey 1997).
Milk Modification and Value Additions for Human Consumption
Large scale sequencing and genome mapping techniques improved the knowledge of animal genetics. A number of gene sequences or gene elements information is available for desire effects (Karatzas and Jeffrey 1997). The conventional crossbreeding of livestock, improvement through nutritional managements, and other methods of quantitative genetics have very slow change in milk yield improvement and now change in protein composition of milk. Milk with alter composition from transgenic animals are now possible with modern technology. The modified milk can be produced with high amount of proteins and all other growth factors naturally deficient in milk (Bremel 1989). The modification in the composition of milk is not limited to milk proteins; but can be extended to the manipulation in the lactose, metabolic enzymes, milk fat, and the minerals in milk (Yom and Bremel 1993).
Modification of milk composition through genetic engineering can also improve the health of animals and supplementation of other components along with milk such as growth hormones, bioactive reagents, and other growth factors. In addition to human, these components can also help to improve the immune status, growth, development of gut and endocrine system of neonates (Wall et al. 1991). The genetically modified animals can produce; (1) high quantity of milk, (2) greater quantity of nutrient in milk, (3) milk with beneficial value additions such as nutraceutical proteins in the milk (Hartmann 1984) (4) milk without the undesired components (e.g., lactose) (5) modified milk with the composition to make more suitable for human consumption especially in neonates (Karatzas and Jeffrey 1997). The number of such proteins like epidermal growth factor (EGF), insulin-like growth factor 1 (IGF-I), transforming growth factor beta (TGF-β) and lactoferrin in the newborns milk can increase the growth, health, and mental status (Konakci et al. 2005; Malcarney et al. 2005; Zhang et al. 2008).
Previously, only pharmaceutical proteins were produced from transgenic dairy animals (reference). Now, milk designed with special compositions or milk with potential human benefits may be competed for next a few years to capture the global marketing worth almost $400 billion annually (Karatzas 2003). In addition, the modified milk for nutrients and therapeutic value can be very beneficial for the health of newborns. However, the concept has been postulated for many years that milk protein contents can be incremented to the natural milk composition with the help of modification in casein gene quantity (Table 4.2).
Table 4.2.
Potential modifications of milk composition by gene addition, with expected functional outcome (Karatzas 2003)
| S.No. | Modification functional consequence | Modification functional consequence |
|---|---|---|
| 1. | Introduction of casein genes | |
| Increase ratio of қ-casein to β-casein or concomitant increase of all caseins by transferring casein locus | Increase in protein and calcium content. Reduction in micelle size, enhancement of heat stability | |
| 2. | Modification of casein genes | |
| Add phosphorylation sites | Increase in calcium content, micelle size, and stability of milk. Enhanced amphiphilicity of β-casein increases its emulsifying and foaming properties | |
| 3. | Introduction of protease (chymosin) cleavage sites | Increase in rate of cheese ripening |
| 4. | Deletion of protease (plasmin) site from β-casein | Increase in emulsifying properties. Elimination of bitter flavor in cheese |
| 5. | Introduction of other functional proteins | |
| Add lysozyme, lactoferrin, or lysostaphin | Milk with antimicrobial activity | |
| 6. | Add reversibly inactive lactase that is activated in gastrointestinal tract upon ingestion of milk | Elimination of sweet taste of lactose hydrolyzed milk and alleviation of lactose intolerance symptoms |
Milk composition can be altered by changing the specific and unique copy of different milk protein genes. Milk composition modifications in transgenic animals have different applications. Modified milk composition can make more suitable milk for infants to drink. In composition human milk is deficient with β-lactoglobulin, which has higher relationship of serum protein to caseins. Human milk has more lactoferrin and lysozyme compared to bovine milk. These are involved in transportation of iron and inhibition of bacterial growth in milk (Van-Berkel et al. 2002). Previously, transgenic cow was successfully developed with human lactoferrin gene and bovine milk with lactoferrin expression for human consumption (Reference). In addition, human lipase can also be expressed in the milk of the transgenic animals. Milk with human lipase is capable to digest the lipid content of milk more efficiently, especially in neonates. Lipase can stimulate the bile salt to increase the conversion of milk triglycerides into fatty acid and glycerol (Houdebine 1995).
Instead of value addition to the components in bovine milk, the removal of undesirable components such as β-lactoglobulin is also interesting subject among to major bovine milk allergens (Wheeler 2003). The bovine milk is directly dependent on the lactose production; lactose is prepared inside the Golgi complex of the mammary secretory epithelial cells with lactose synthase complex. This complex comprises on the α-lactalbumin and the enzyme β1,4 galactosyltransferase and a catalyst of reaction lactose synthase. Lactose is released from Golgi complex in secretory vesicles and transported to apical of epithelial cell and secreted into the lumen. First lactose attracts water inside the vesicle by osmosis, because it cannot diffuse out from vesicles without this process. The lactose synthase have vital role in lactose synthesis, influx of water into secretory vesicles and finally lactose release into the lumen. The α-lactalbumine have vital role in osmoregulation of milk (Wheeler 2003).
A milk modifications have been made this possible to consume the milk by patients suffering with metabolic disorders such as phenylketonuria. The patient suffering with phenylketonuria lacks the enzyme that metabolizes phenylalanine and needs diet with lower phenylalanine contents. Otherwise, the modification in milk proteins such as α-lactalbumine without disturbing the structure of protein can be used in diet of the patient suffering with phenylketonuria as supplementary food without undesired components (Colman et al. 1995). The reduction of lactose in the milk is also among the major desirable characters related to the consumption of bovine milk in people with lactose intolerance. According to an estimate, 70 % of people worldwide are suffering with lactose intolerance due to deficient level of lactase in their intestine. The lactose can be reduced into milk by expressing β-glactosidase into bovine milk or with removal of α-lactalbumin components from milk with modern biotechnological techniques (Montaldo 2006). Stinnakre et al. (1994) were reported α-lactalbumine gene inactivation from mouse. Transgenic mouse has been produced milk without lactose with a serious disadvantage that milk of such animal was very viscous in consistency and unable to secrete from mammary glands of mouse, because lactose is an important protein in osmoregulation of milk.
One glass of milk comprises almost 8 g of protein with 78–80 % caseins contents of this amount. Casein along with milk fat gives white chalky color to milk. In cow milk, four types of casein proteins such as αS1- and β-casein (10 g/l each), αS2-casein (3.7 g/l), and κ-casein (3.5 g/l). The sequestering and binding of calcium and magnesium with in a spherical particle (micelle) is one of the major functions of the caseins. The outer surface of micelle is made up of κ-casein, which can easily destabilize by chymosin enzyme to the form the curd and finally cheese from milk. The modification in chymosin cleavage sites of aS1-CN to peptide linkages for more efficient hydrolysis can also reduce curd formation time of cheese. Moreover, casein has little sulfur containing amino acids; the alteration in milk by increasing Met contents can improve the nutritional value of such milk. So, any alteration in casein in milk can directly affect the cheese yield from milk (Bawden 1994; Karatzas 2003). Therefore, modification in casein gene is also very helpful to alter and increase nutritive value, cheese yield from milk and other components of milk processing (Table 4.2). Previously, efforts were made to increase the copies of the κ-casein gene into transgenic animals to decrease the size of micelles and to make more susceptible modified κ-casein (References). These studies successfully demonstrated that extra copies of casein gene in mouse model successfully encoding two type of casein: bovine β-casein and κ-casein. Modified milk collected from cloned transgenic animals with extra copies of casein genes were enriched with β- and κ-casein, which increased the 30 % total milk casein and 13 % milk protein (Gutiérrez-Adan et al. 1996; Brophy et al. 2003) with such alterations change the physical properties of protein (Brophy et al. 2003). In another study, the transgenic cow was developed with modification in κ-casein and an overexpression of this gene decrease the size of micelles in milk, which favor the cheese industry; while this milk was slightly more yellow in color and lower β-casein contents. Economic value of this milk still needs to be proven (Soler et al. 2006).
In addition, casein solubility could increase only with increasing in glycosylation, which could reduce the time for coagulation of rennet and expulsion of whey. This process would yield the firm curds from milk and would improve in cheese making. Alteration in other physical properties could improve the quality of food produce from milk, such as cheese with low fat and better taste. The production of milk along with processing and transportation cost of milk can be reduced with genetic engineering techniques. These techniques made this possible to use more milk and milk products with better value addition. These methods offered a better opportunity for the production of protein milk to improve animal agriculture and creation of new medicines in milk (Gottlieb and Wheeler 2011).
Mastitis is a major factor on the milk quality deterioration. It is most devastating disease of dairy industry with reduction or termination of milk. This disease leads to undesirable consequence of remaining milk such inclusion of bacteria and inflammatory cells in the milk, unpleasant taste and undesirable effects. Mastitis causes of 53 billion dollars loss per annum worldwide (Ratafia 1987; Miller 1993). Some specific antibodies can be produced in the mammary gland in genetically engineered animals which can protect animals from specific problems such mastitis. The antibodies secreted through the milk can protect our domestic livestock and humans from diseases (Zang et al. 2008). Mastitis resistant animal is possible by inclusion of certain genes in animals which are expressing antibacterial substances into milk such as nutriceuticals, proteases, lysozyme, and transferrin (Houdebine 2000; Kerr and Wellnitz 2003; Felmer 2004). Cow’s milk with some antibacterial proteins such as lysostaphin has great antibacterial activity. These animals have been recognized very resistant to Staphylococcus aureus mammary infection. The milk of these animals was also resistant to spontaneous bacterial infections arised from environmental contamination. However, the acceptability of lysostaphin containing milk for humans was unexplored, whether this milk would alter the composition of human intestinal microflora (Soler et al. 2006).
The bacterial systems cannot glycosylate the mammalian protein, while the fungal system although have glycosylation ability with addition of many undesirable groups on the proteins. So, the functional properties of such proteins are strongly affected by this alteration. Other techniques of protein production such as baculovirus system and mammalian cell culture techniques are suitable for the production of proteins along with specific problems. Instead of all, the genetically engineered animals are very suitable alternative for the production of complex mammalian proteins (Rudolph 1999).
The 5′ flanking end of the genes for milk proteins, which normally have regulatory role for their expression, can help to express foreign genes in mammary epithelia cells of genetically engineered animals. Previously, transgenic animals were developed for the expression of different proteins along with these regulator genes; almost have similar observations of expression as in the case of transgenic mouse (Wheeler 2003). Interestingly in the case of complex process of milk production, the expression of one protein is indigenously counterbalanced by the expression of other protein. So, it is quite difficult to alter the milk production with single gene transgenic approach. It is very necessary to understand the biological control for the production of milk before the remarkable investment to make transgenic animals of dairy importance (Wheeler 2003).
With a biotechnological approach, many proteins of human therapeutic importance were expressed in the milk of transgenic animals. Even human collagen protein has been produced into the milk of transgenic animals, which is a very large and complex protein human protein. Modern technologies such as bacterial fermentation and cell culture can produce human proteins e.g., human plasminogen activator (htPA) more economically into transgenic animal milk. In other proteins of human industrial interest were successfully expressed and isolated from the milk of transgenic domestic animals include antitrypsin and human antithrombin III. Industrial research is focused on the production of the human industrial proteins with greater value and low volume human beneficial protein in milk of the transgenic domestic animals (Karatzas and Jeffrey 1997).
The previous studies clearly revealed the importance of modern biotechnological tools for synthesis of modified milk with value addition human. Considerable time and resources are required for the production of dairy animals with new genetics and commercial values. Such modifications in the milk would be made animals of low value to animals of higher commercial importance. Other policies related to consumption of genetically novel milk of modified dairy animals will be overcome soon in future by the drug and food administrative authorities of developing countries. Modified animals with desire characters will be soon in the breeding programs of developing nations to improve the livestock.
However, the production of modified milk with wanted character or component is an excellent source for the production of protein at a large quantity. Milk transgenic technology can also exploit the economical productivity of mammary glands. In major problems include the accurate production of transgenic animals along with regulatory issues. Additionally, the issue to address is the large scale economical production of milk and to overcome the public concern.
Modification of Hairs or Fibers and Wool Production
The biotechnological modifications of wool and fiber contents were relatively slow compared to other characteristics. The skin biology and development in cellular level has been understood since 1950s. However, most of the studies were focused on the wool follicles and growth of wool fibers from wool follicles; while other higher organization of mechanisms in this process such as trio groups of primary and secondary follicles received little attention of scientific investigations (Fraser and Short 1960; Purvis and Franklin 2005). The development of wool follicles start at early stage of fetus development and continue through the lifespan of the animals (Fraser and Short 1960; Purvis and Franklin 2005).
Animal hairs, fibers and wool are used in fabric and yarn production. Transgenic manipulation in livestock for modification in fabric and yarn production has been recognized another application area of modern animal biotechnology. The genetic manipulation has been focused for the modification in hairs, fibers and wool for color, quality, yield, harvesting ease (Wheeler 2010). The different transgenic method has also been solicited to examine the wool and hair fibers refinement, excellence and crimp. The modification in the keratin and keratin associated protein (KAP) genes of sheep have been improved the processing and wearing quality of wool fibers. The cortical-specific high expression of intermediate wool filament type II keratin gene (K2.10) produced the modified microstructures and macrostructures of wool filaments with higher luster and lower crimps. Several modifications in genome of transgenic animals have been demonstrated with more elastic and strong fibers, which would reduce the shrinkage quality of garments (Bawden et al. 1998; Wheeler 2010).
The efforts were made to increase the growth of wool yield in sheep. The insulin growth factor-1 (IGF-1) gene expression in the follicle was used to increase the wool production. The expression of IGF-1 gene in transgenic sheep produced 6 % increase growth of wool at year 1 compared to wild type sheep. Moreover, the wool was poor in quality wool with lower stapled strength and coarse in texture. In addition, the transgenic sheep produced higher wool content only at first year and no significant improvement was observed in next year (Damak et al. 1996; Su et al. 1998).
The wool production was boosted with over expression of ovine growth hormone, while the higher plasma concentration produced some harmful effects (Ward and Brown 1998). The transgenic merino sheep was produced with improve construct, resulted in 12 % improvement in fleece production. In Poll Dorset cross-breed sheep, opposite results were observed such as reduction in wool contents in transgenic sheep contain ovine GH gene construct. The results explained a significant association of breed type with transgene expression and response (Adam et al. 2002; Wall et al. 2009). The wool produce from transgenic animal with ovine GH transgene expression were poor in quality such as larger but less desirable diameter of fibers. The ovine intermediate filament keratin transgene in wool follicle has also been expressed to improve the quality less desirable fibers (Adam et al. 2002; Wall et al. 2009). In sheep, harvesting at specific time is another problem for the farmers and labor intensive. The modern methods of transgenic technologies made this possible that sheep shed their wool fiber at specific time period (Hollis et al. 1983). The mouse epidermal growth factor (EGF) has been introduced in transgenic sheep under the influence of inducible promoter (Hollis et al. 1983). The infusion of Mouse epidermal growth factor (mEGF) induced follicular regression and fleece harvesting more rapid than previous depilatory agents without any anomaly, apart from sustained epidermal thickening. A spot in the wool fibers can be produced with successful expression of such a transgene and endorsed fleece to remove with hand pressure. This modification reduced the cost of metal shearers or clippers during wool harvesting. This cost effectiveness is really an advantageous for wool producers or farmers (Hollis et al. 1983; Wheeler 2012). This modification can also be applied to other wool producers such as camels, mohair goat, alpacas, and other wool producers (Wheeler 2012).
The single transgene expression in transgenic animals to improve the wool and fiber content was forced to utilize some novel transgenic strategies such as bacterial derived biosynthetic pathway genes for synthesis of amino acids such as cysteine. Cysteine is a wool growth regulating element in livestock rear for wool or fiber production. The approach was successfully demonstrated with two different bacterial genes in transgenic mice, while this approach was unsuccessful in sheep due to low level of unsustainable biosynthetic enzyme expression (Ward et al. 1994; Bawden et al. 1995; Ward 2000).
Animal wool production and properties of fibers can be altered with transgenic technology. Sheep wool contains limited cysteine fibers contents, with increase in this amino acid may increase the production and properties of wool in transgenic animals. Some previous studies have been performed to introduce the cysteine biosynthesis genes from bacteria to the genome of sheep. This strategy did not allow to produce and efficient expression of these enzymes in the rumen of transgenic sheep (Murray et al. 1999; Montaldo 2006).
Transgenic approach has been used for the expression of very valuable fiber in the milk of transgenic goat (Karatzas et al. 1999). The seven different types of silk are used by the spider in the synthesis of orb webs. The unique mechanical properties of each fiber differentiate these fibers from other natural and synthetic fibers (Karatzas et al. 1999). The dragline silk is the most robust variety of silk, which has properties of 35 % elongation from original material and high tensile strength. In addition, the capability of energy absorption before cracking is more than steel (Wheeler 2010). Breed Early Lactate Early (BELE®) goat system has been used for expression of protein monomers gather to develop the spider silk fiber in the milk of transgenic goat. The fibers assemble from protein monomers of transgenic goat were obtained similar quality of spider silk. These fibers have different applications such as medical devices, aircrafts, sutures, automotive composite, ballistic protection, and special clothing (Wheeler 2012).
Further investigations are required in addition to rate limiting amino acid modification such as cysteine, protein modification like wool intermediate filament keratin, wool-related amino acid enrichment of keratin and Insulin growth factor 1 overexpression in wool follicle; which can increase the wool fiber growth or harvesting ease. The consideration of wool and fiber growth and quality-related QTLs could also be included in future animal breeding strategies for the production of more suitable breed with better growth and quality of wool. The human future demand for better cloth may be emphasized researcher to produce the better wool and fiber contents from transgenic animals with different and unique transgenic approaches. The transgenic animal biotechnology can play a vital role for the production of fine fiber and wool to meet the incremental future demands of garments with better processing and wearing quality.
Modification for Infectious Disease Resistance
Genetic engineering related to animal biotechnology should primarily apply on the animal welfare and health. The interesting application of modern animal biotechnology related to animal welfare is to make the animal resistance to different diseases. The genetic information has been made this possible to make transgenic resistant animals to various diseases. The disease resistance in animals is polygenic trait and a few loci are identified related to disease resistance against particular disease. The transgenic approaches related to animal disease resistance include the transfer of major histocompatibility complex, immunoglobulin genes, T cell receptor gene, gene affect lymphokines/specific disease resistance. The genes involve in the histocompatibility complex (MHC) during immune response can be used as major instrument for the animal disease resistance and transgenic animal with better disease resistance (Wheeler 2012).
Many genetic aspects for the disease resistance in livestock have been determined, while the role of specific genes in immune response and disease development still need keen intentions of researchers (Ebert and Selgrath 1991). Previous study results showed that, the genetic manipulation for disease resistance revealed that the embryonic cells are very useful to modify such genes. The germ line transfer has been possible up to >100 kb with pronuclear injection, while >400 kb has been transformed into transgenic animals with yeast artificial chromosome vector. Therefore, the disease resistant livestock could easily be produced with embryonic stem cell transgenic approach (Wheeler 2012).
Haller et al. (1981) has been produced newborn transgenic swine containing the Mx1 protein gene and high expression has been made resistance to lethal infection of influenza virus. The MxcDNA transformation and expression in 3T3 cell lines produce resistance against the lethal influenza virus infection (Staeheli et al. 1986). The transgenic piglets have been developed with cDNA construct encoding Murine Mx1 protein. The transgenic piglets were successfully developed resistance against lethal influenza virus infection (Muller et al. 1992). The transgenic piglet with over expression of Mx1 protein might have much detrimental effect on the piglets. Furthermore, the expression of Mx1 protein was eliminated with rearrangement of transgene insert in transgenic piglets (Muller et al. 1992). Mastitis is udder inflammatory disease of cattle ‘as a result’ animal loss productivity and milk yield. Previously, the transgenic cattle resistant to Staphylococcus aureus infection has been produced with transgene expression of lysostaphin, an antimicrobial peptide secreted in milk. Lysostaphin killed the bacteria with in a dose dependent manner (Donovan et al. 2005). Moreover, the mammary gland associated expression of lysozyme in goat inhibited the growth and development of bacteria involve in mastitis and cold spoilage of milk (Maga et al. 2006).
The transgenic technology has been proved a vital tool to produce transgenic animals resistant to different diseases in above mentioned studies. Furthermore, the transgenic technology made this possible to enhance the useful allele inside transgenic animals and to knockout the harmful genes from these animals. Previously, the knockout of Escherichia coli K88 receptor inside transgenic experimental swine has been ascertained completely resistant to E. coli positive with K88 compared to wild animal (Edfors-Lilia et al. 1986). Moreover, the similar investigations should be applied on other animal health issues including parasitic infections such as trypanosomes and nematodes, viral infections such as foot and mouth disease, bovine leukemia virus, pseudorabies virus, bacterial infection such as clostridium infection, streptococcus infection and deficiency disorder such as deficiency of UMP synthase, mule foot and bovine leukocyte adhesion deficiency (Wheeler 2012).
The transgenic pig, sheep and mice were successfully developed with the mouse immunoglobulin A (IgA) transgene construct and expression (Lo et al. 1991). The transgene of murine IgA was successfully introduced in two transgenic pig lines with only light chain expression of IgA molecules (Lo et al. 1991), while a transgenic pig has been developed with successful expression of murine monoclonal antibodies with great binding ability to specific antigen (Weidle et al. 1991). The resistance against the ovine Visna virus has been developed in transgenic sheep. Transgenic sheep was developed with Visna virus envelop transgene and expression without any harmful or pathological effects (Clements et al. 1994). The transgene of viral envelop was not expressed in monocytes, while antibodies were developed after experimental infection to transgenic sheep (Clements et al. 1994). The virus neutralizing antibodies were produced in the transgenic mice model milk against a coronavirus responsible for economically important transmissible gastroenteritis (TGEV) disease (Castilla et al. 1998). The transgene expression and engineered passive immunity was achieved throughout the lactation. In near future, this work would also be demonstrated and verified in transgenic pig (Castilla et al. 1998).
The self-immunization against pathogenic organisms is also a great application of this exciting technology. The construction and transformation of transgenes under the influence of physiological or specific stimulus to express could be produced antigen at specific time or with specific response to stimulus with this tremendous technology. This expression would immunize the transgenic animals against particular disease in response of specific stimulus or physiological condition (Wheeler 2012). The transgenic animals will be developed with transgene construct under the control of stimuli zinc in feeding or specific antibiotic to express the antigen at specific time period to increase the specific antibiotic level in serum (Wheeler 2012).
Scrapie and bovine spongiform encephalopathy free or naturally resistant animals may be produced in future with technology. The transgenic technology is a vital tool to develop animal model in epidemiological investigation of such disease and eradication from farm animals. The experimental production of either human or bovine prion proteins in transgenic mice is a great example of such application (Bishop et al. 2006). In addition, knockout is only a successful of approach to avoid the infection and transmission of spongiform encephalopathies like scrapie orBSE. Denning et al. (2001) has first time targeted the ovine prion (PrP) gene locus, while the clone lamb died short after birth. The transgenic cattle cloned with knockout of prion locus have also been developed (Cyranoski 2003).
Similarly, the resistance against brucellosis has been successfully established in fetus of transgenic bull develop from somatic cell line nuclear transfer, which is highly contagious disease of zoonotic importance, and caused high fever, muscular pain, and reproductive disorder in females (Shin et al. 1999). It is still a very short and selective discussion related to application of transgenic technology on the animal and human welfare. It can be applied on a number of organisms related to animal health and productivity. Healthy animals would be more productive and useful for farmer and less effort would be required in rearing. In future, the use of animal biotechnology especially transgenic technology will be increased in future to improve the animal health and production. Some ethical issue related to transgenic animals and very high cost associated to animal transgenic technology are still major constraints in application of this technology for animal welfare.
Harmful Aftermaths of Different Animal Modifications
The transgenic animal approach is very useful to understand the function of different proteins and secondary gene products related to these proteins. The transgenic research related to allied fields of nutrition is benefiting to understand the normal and alter metabolism effects (Prieto et al. 1999). The food ingredients, food nutrients and food products from transgenic animals have not yet extended to consumers reach. This technology is maturing and producing thrilling results in model and experimental farm animals. The advent transgenic food products emphasized the need of guidelines and legislation, which has been developed in many countries to anticipate such products in market for consumer (Prieto et al. 1999).
Practically, the transgene expression is not always useful for the animal health. The transgene unusually expressed in the nonspecific tissue and biological fluid in which they not normally expressed. This nonspecific expression was caused some adverse and undesirable effects; for example, the transgenic mice with GH have elevated level of GH compared to wild-type littermates. This high expression of GH elevated the level of insulin, as result, the transgenic animals died due to liver and kidney impairment (Chen et al. 1997). The products of transgenic plants such as some fruits and vegetables are already available in the market, while animal functional foods or modified foods are not yet reached to consumer market. The animal modified food derived from predictable or nonpredictable animal modification methods and food safety consequences, which may be a major reason for animal derived food unavailability in the market (Prieto et al. 1999).
The undesirable effects observed in transgenic animals emphasize the need for further research related to genes/genes expression control and physiological consequences due to chronic expression in case of certain proteins. Furthermore, it is clearly demonstrated from previous studies that the fetus is unable to develop, when transgenes used the systemic regulatory elements during gene expression. In this regard, the expressions of glycosyltransferases and enzymes’ general effect on glycosylation are most significant (Metzler et al. 1994; Prieto et al. 1999), while the early development is arrested due to a sialic acid-specific esterase under the metallothionein transcription regulatory element (Varki et al. 1991). Similarly, the development of mammary gland morphogenesis of transgenic mice was impaired and inhibited due to overexpression of cell surface β1,4-galactosyltransferase, under the influence of metallothione in transcription regulatory element (Hathaway and Shur 1996).
Furthermore, the genetically engineered mice has been developed with transgene expression of human α1-3/4-FT glycosyltransferase for carbohydrate biosynthesis pathway under the control of rat intestinal fatty acid binding protein transcription regulatory element or the whey acidic protein promoter without any harmful effect. The secondary gene product and tissue-specific expression of transgene was localized into tissue, but the outcome of the expression may be specific to species (Bry et al. 1996; Prieto et al. 1999). The transgenic rabbits with transgene expression of fusion protein were demonstrated harmful effect to milk production and lactose free milk was produced from transgenic animals. The transgenic animals were studied up to many generations to understand the harmful of enzyme effect on the lactation. These studies demonstrated many factors effect on final result including the capability of transgenic animal species, even when the same gene construct or primary gene product is expressed (Prieto et al. 1999). In other factors included the limitation of tissues for the expression of specific protein such as lactating mammary gland (Bleck et al. 1995).
The transgenic rabbits developed from human erythropoietin gene under the influence of whey acidic protein gene promoter, and the ectopic expression of transgene produced some deleterious effects (Massoud et al. 1996). Moreover, the transgenic pig was developed with transgenic extemporaneous expression of whey acidic protein. The mammary glands development was impaired in transgenic pig (Shammay et al. 1992). These studies are remarkable related to protein function, development and morphogenesis along with species-specific response. Wall et al. (1996) demonstrated that the transgenic sheep expressed the mouse whey acidic protein in all tissue except into lactating mammary glands. Some of the harmful effects are mentioned in Table 4.3.
Table 4.3.
The undesirable and unpredictable effects from transgenic animals (Modified from Prieto et al. 1999)
| S.No. | Transgenic animal | Trasngene | Effect | References |
|---|---|---|---|---|
| 1. | Pig | MTh-bovine GH | Gastric ulcer, cardiomegaly, arthritis | Pursel et al. (1989) |
| 2. | Pig | mWAP | Failure to lactate (agalactia; characteristic mammary gland phenotype) | Shamay et al. (1992) |
| 3. | Mice | baLac-bbcasein | Short lactation | Bleck et al. (1995) |
| 4. | Rabbit | rWAP-EPO | Infertility, agalactia, premature death | Massoud et al. (1996) |
| 5. | Mice | MTh-b-Gal-transferase | Impaired mammary gland development | Hathaway and Shur (1996) |
| 6. | Rabbit | mWAP-a1-2FUT I | Lactose free milk (changes in milk protein quality and content) | Prieto, unpublished results |
MTh mouse metallothionein promoter. mWAP mouse whey acidic protein promoter. rWAP rabbit whey acidic protein promoter. EPO human erythropoietin. baLac-bbcasein bovine alactalbumin promoter-bovine b-casein. α1-2FUT human α1-2 fucosyltransferase “H”
Future Constrains and Prospective
The green revolution has brought about reasonable prosperity in the life of farmers with land, but the majority of landless farmers remain poor. Poverty, malnutrition, disease, poor hygiene, and unemployment is widespread in the developing world and biotechnologies can play a vital role in this context. Some modified animal food products are developed with modern biotechnological tools. Such animal food items are economical and more useful in respect to health. The modern biotechnology implementation to alter animal products for fat and other characteristics is constrained by a number of factors. One of the major constraints on applying biotechnologies is the economic and wealth status of animal farmers. The major constrains to adopt modified food products with new technology in developing nations include the absence of an accurate database on livestock, biodiversity with animals, disease resistance, and nutrient utilization characteristics.
Moreover, in other challenges to adopt such products in developing nations include the lack of trained manpower both in the government and in the private sectors for the development of modified animal food products along with the absence of an interface between industry, universities and institutions, the high cost of technological inputs such as materials, biological and equipment, the failure to address issues of biosafety and to conduct risk analyses of new modified animal food products, the negligible investment in animal biotechnology. Criticisms of such technologies and applications involve issues ranging from food safety and social resistance to potential negative impacts on animal safety and on ecosystems along with the current regulatory structure to assess and manage risks created by these technologies.
This technology has massive expectations in the future for production of transgenic livestock with desired modification for food production. Furthermore, this technology still needs to improve related to modified animal food composition and production capability such as growth rate, disease resistance, and reproductive efficiency. These modifications need improvement in the desirable traits quantity, identification of markers linked to certain genes with specific characters or traits, selection of better animals, propagation of superior animals, practical and economically viable methods. The four things to be considered for animals in the transgenic approach for desirable traits are time period for commercial development, regulatory concerns and related issues, economic feasibility, and adoptability of these production methods (Prieto et al. 1999). Transgenic animal technology is a powerful tool to improve food production and to study nutrition effects in health and disease development.
Contributor Information
Muhammad Abubakar, Email: hayee42@yahoo.com.
Ali Saeed, Email: alisaeed@bzu.edu.pk.
Oguz Kul, Phone: +903183573304, Email: oguzkul72@yahoo.com.
Ali Saeed, Email: alisaeednibge@gmail.com.
References
- Adams NR, Briegel JR, Ward KA. The impact of a transgene for ovine growth hormone on the performance of two breeds of sheep. J Ani Sci. 2002;80(9):2325–2333. doi: 10.2527/2002.8092325x. [DOI] [PubMed] [Google Scholar]
- Bawden WS, Passey RJ, MacKinlay AG. The genes encoding the major milk-specific proteins and their use in transgenic studies and protein engineering. Biotechnol Genet Eng Rev. 1994;12:89. doi: 10.1080/02648725.1994.10647910. [DOI] [PubMed] [Google Scholar]
- Bawden CS, Powell, BC Walker SK, Rogers GE (1998) Expression of a wool intermediate filament keratin transgene in sheep fiber alters structure. Transgenic Res 7:273–287 [DOI] [PubMed]
- Bawden CS, Sivaprasad AV, Verma PJ, Walker SK, Rogers GE. Expression of bacterial cysteine biosynthesis genes in transgenic mice and sheep: toward a new in vivo amino acid biosynthesis pathway and improved wool growth. Transgenic Res. 1995;4:87–104. doi: 10.1007/BF01969411. [DOI] [PubMed] [Google Scholar]
- Bishop MT, Hart P, Aitchison L, Baybutt HN, Plinston C, Thomson V, Tuzi NL, Head MW, Ironside JW, Will RG, Manson JC. Predicting susceptibility and incubation time of human-to-human transmission of vCJD. Lancet Neurol. 2006;5:393–398. doi: 10.1016/S1474-4422(06)70413-6. [DOI] [PubMed] [Google Scholar]
- Bleck G, Jimenez-Flores R, Bremel R. Abnormal properties of milk from transgenic mice expressing bovine β-casein under control of the bovine α-lactalbumin 59 flanking region. Int Dairy J. 1995;5:619–632. [Google Scholar]
- Bowen RA, Reed M, Schnieke A, Seidel GE, Stacey A, Thomas WK, Kaijkawa O. Transgenic cattle resulting from biopsied embryos: expression of c-ski in a transgenic calf. Biol Reprod. 1994;50:664–668. doi: 10.1095/biolreprod50.3.664. [DOI] [PubMed] [Google Scholar]
- Bremel RD, Yom HC, Bleck GT. Alteration of milk composition using molecular genetics. J Dairy Sci. 1989;72:2826–2833. doi: 10.3168/jds.S0022-0302(89)79428-5. [DOI] [PubMed] [Google Scholar]
- Brophy B, Smolenski G, Wheeler T, Wells D, L’huillier P, Laible G. Cloned transgenic cattle produce milk with higher levels of beta-casein and kappa-casein. Nat Biotechnol. 2003;2(21):157–162. doi: 10.1038/nbt783. [DOI] [PubMed] [Google Scholar]
- Bry L, Falk PG, Gordon JL. Genetic engineering of carbohydrate biosynthetic pathways in transgenic mice demonstrates cell cycle-associated regulation of glycoconjugate production in small intestinal epithelial cells. Proc Natl Acad Sci USA. 1996;93:1161–1166. doi: 10.1073/pnas.93.3.1161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bucher HD, Hengstler P, Schindler C, Meiter G. N-3 PUFA in coronary heart disease: a meta-analysis of randomized controlled trials. Am J Med. 2002;112:298–304. doi: 10.1016/s0002-9343(01)01114-7. [DOI] [PubMed] [Google Scholar]
- Burcelin R, Printz RL, Kande J, Assan R, Granner DK, Girard J. Regulation of glucose transporter and hexokinase II expression in tissues of diabetic rats. Am J Physiol. 1993;265(3 Pt 1):E392–E401. doi: 10.1152/ajpendo.1993.265.3.E392. [DOI] [PubMed] [Google Scholar]
- Bustamante E, Pedersen P. High aerobic glycolysis of rat hepatoma cells in culture: role of mitochondrial hexokinase. Proc Natl Acad Sci USA. 1977;74(9):3735–3739. doi: 10.1073/pnas.74.9.3735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campbell NA, Reece JB. Biology. 6. San Francisco: Pearson Education; 2002. pp. 69–70. [Google Scholar]
- Cameron ER, Harvey MJ, Onions GE. Transgenic science. Br Vet J. 1994;150(1):9–24. doi: 10.1016/S0007-1935(05)80093-4. [DOI] [PubMed] [Google Scholar]
- Castilla J, Pintado B, Sola I, Sánchez-Morgado JM, Enjuanes L. Engineering passive immunity in transgenic mice secreting virus-neutralizing antibodies in milk. Nat Biotechnol. 1998;16(4):349–354. doi: 10.1038/nbt0498-349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen N, Chen WY, Striker LJ, Striker GE, Kopchick JJ. Co-expression of bovine growth hormone (GH) and human GH antagonist genes in transgenic mice. Endocrinology. 1997;138:851–854. doi: 10.1210/endo.138.2.5036. [DOI] [PubMed] [Google Scholar]
- Clements JE, Wall RJ, Narayan O, Hauer D, Schoborg R, Sheffer D, Powell A, Carruth LM, Zink MC, Rexroad CE. Development of transgenic sheep that express the Visna virus envelope gene. Virology. 1994;200(2):370–380. doi: 10.1006/viro.1994.1201. [DOI] [PubMed] [Google Scholar]
- Colman A, Wright G, Sawyer L, Rigden DJ, inventors (1995) Modified alpha-lactalbumin Pharmaceutical Proteins Ltd., assignee. WO 95/02692
- Cowan T (2010) Biotechnology in animal agriculture: status and current issues. CRS report prepared for members and committees of congress. Congressional research service, pp 1–22
- Cyranoski D. Koreans rustle up madness-resistant cows. Nature. 2003;426(6968):743. doi: 10.1038/426743a. [DOI] [PubMed] [Google Scholar]
- Damak S, Su H, Jay NP, Bullock DW. Improved wool production in transgenic sheep expressing insulin-like growth factor 1. Biotechnol (NY) 1996;14:185–188. doi: 10.1038/nbt0296-185. [DOI] [PubMed] [Google Scholar]
- Davies PL, Hew CL. Biochemistry of fish antifreeze proteins. FASEB J. 1990;4:2460–2468. doi: 10.1096/fasebj.4.8.2185972. [DOI] [PubMed] [Google Scholar]
- Davis RA, Miyake JH, Hui TY, Spann NJ. Regulation of cholesterol-7-hydroxylase: BAREly missing a SHP. J Lipid Resear. 2002;43:533–543. [PubMed] [Google Scholar]
- Decker EA, Xu Z. Minimizing rancidity in muscle foods. Food Technol. 1998;52(10):54–59. [Google Scholar]
- DeFilippis AP, Sperling LS. Understanding omega-3’s. Am Heart J. 2006;151:564–570. doi: 10.1016/j.ahj.2005.03.051. [DOI] [PubMed] [Google Scholar]
- Denning C, Burl S, Ainslie A, Bracken J, Dinnyes A, Fletcher J, King T, Ritchie M, Ritchie WA, Rollo M, de Sousa P, Travers A, Wilmut I, Clark AJ. Deletion of the alpha(1,3) galactosyltransferase (GGTA1) and the prion protein (PrP) gene in sheep. Nat Biotechnol. 2001;19(6):559–562. doi: 10.1038/89313. [DOI] [PubMed] [Google Scholar]
- Devlin RH, Yesaki YT, Biagi CA, Donaldson EM, Swanson P, Chan WK. Extraordinary salmon growth. Nature. 1994;371:209–210. [Google Scholar]
- Donovan DM, Kerr DE, Wall RJ. Engineering disease resistant cattle. Transgenic Res. 2005;14:563–567. doi: 10.1007/s11248-005-0670-8. [DOI] [PubMed] [Google Scholar]
- Draghia-Akli R, Fiorotto ML, Hill LA, Malone PB, Deaver DR, Schwartz RJ. Myogenic expression of an injectable protease-resistant growth hormone-releasing hormone augments long-term growth in pigs. Nat Biotechnol. 1999;17:1179–1183. doi: 10.1038/70718. [DOI] [PubMed] [Google Scholar]
- Du SJ, Gong Z, Fletcher GL, Shears MA, King MJ, Idler DR, Hew CL. Growth enhancement in transgenic Atlantic salmon by the use of an “all fish” chimeric growth hormone gene construct. Biotechnol. 1992;10:176–181. doi: 10.1038/nbt0292-176. [DOI] [PubMed] [Google Scholar]
- Dunn MF, Ramirez-Trujill JA, Hernandez-Lucas I (2009). Major roles of isocitrate lyase and malate synthase in bacterial and fungal pathogenesis. Microbiology 155(10):3166–3175 [DOI] [PubMed]
- Eastmond PJ, Graham IA. Re-examining the role of the glyoxylate cycle in oilseeds. Trends Plant Sci. 2001;6(2):72–78. doi: 10.1016/s1360-1385(00)01835-5. [DOI] [PubMed] [Google Scholar]
- Ebert KM, Selgrath JP (1991) Changes in domestic livestock through genetic engineering. In: Pedersen RA, McLaren A, First NL (ed) Current communication in cell and molecular biology: animal applications of research in mammalian development, pp 233–266
- Edfors-Lilia I, Petersson H, Gahne B. Performance of pigs with or without the intestinal receptor for Escherichia Coli K88. Anim Prod. 1986;42:381–387. [Google Scholar]
- Felmer R. Animales transgénicos: pasado, presente y futuro. Arch Med Vet. 2004;2(36):105–117. [Google Scholar]
- Fraser AS, Short BF (1960) The biology of the fleece. In: Animal research laboratories technical paper no 3, CSIRO Melbourne
- Golovan SP, Meidinger RG, Ajakaiye A, Cottrill M, Wiederkehr MZ, Barney DJ, Plante C, Pollard JW, Fan MZ, Hayes MA, Laursen J, Hjorth JP, Hacker RR, Phillips JP, Forsberg CW. Pigs expressing salivary phytase produce low-phosphorus manure. Nat Biotechnol. 2001;19:741–745. doi: 10.1038/90788. [DOI] [PubMed] [Google Scholar]
- Grobet L, Pirottin D, Farnir F, Poncelet D, Royo LJ, Brouwers B, Christians E, Desmecht D, Coignoul F, Kahn R, Georges M. Modulating skeletal muscle mass by postnatal, muscle-specific inactivation of the myostatin gene. Genesis. 2003;35:227–238. doi: 10.1002/gene.10188. [DOI] [PubMed] [Google Scholar]
- Gottlieb S, Wheeler MB (2011) Genetically engineered animals and public health: compelling benefits for health care, nutrition, the environment, and animal welfare. Bio, biotechnology-industry organization (revised edn), Washington DC, pp 1–34
- Grundy SM. Comparison of monounsaturated fatty acids and carbohydrates for lowering plasma cholesterol. N Engl J Med. 1986;314:745–748. doi: 10.1056/NEJM198603203141204. [DOI] [PubMed] [Google Scholar]
- Gutiérrez-adán A, Maga EA, Meade HM, Shoemaker CF, Medrano JF, Anderson GB, Murray JD. Alterations of the physical characteristics of milk from transgenic mice producing bovine κ-Casein. J Dairy Sci. 1996;5(79):791–799. doi: 10.3168/jds.S0022-0302(96)76427-5. [DOI] [PubMed] [Google Scholar]
- Haller O, Arnheiter H, Gresser I, Lindenmann J. Virus-specific interferon action/protection of newborn Mx carries against lethal infection with influenza virus. J Exp Med. 1981;154:199–203. doi: 10.1084/jem.154.1.199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hammer RE, Brinster RL, Rosenfeld MG, Evans RM, Mayo KE. Expression of human growth hormone-releasing factor in transgenic mice results in increased somatic growth. Nature. 1985;315:413–416. doi: 10.1038/315413a0. [DOI] [PubMed] [Google Scholar]
- Hartmann PE, McCauley I, Gooneratne AD, Whitely LJ. Inadequacies of sow lactation: survival of the fittest. Symp Zool Soc Lond. 1984;51:301–326. [Google Scholar]
- Hathaway H, Shur B. Mammary gland morphogenesis is inhibited in transgenic mice that overexpress cell surface β1, 4-galactosyltransferase. Development. 1996;122:2859–2872. doi: 10.1242/dev.122.9.2859. [DOI] [PubMed] [Google Scholar]
- Hew C, Poon P, Xiong F, Gauthier S, Shears M, King M, Davies P, Fletcher G. Liver-specific and seasonal expression of transgenic Atlantic salmon harboring the winter flounder antifreeze protein gene. Transgenic Res. 1999;8:405–414. doi: 10.1023/a:1008900812864. [DOI] [PubMed] [Google Scholar]
- Hew CL, Scott GK, Davies PL (1986) Molecular biology of antifreeze. Living in the cold: physiological and biochemical adaptation. Elsevier Press, New York, pp 117–123
- Hollis DE, Chapman RE, Panaretto BA, Moore GP. Morphological changes in the skin and wool fibers of Merino sheep infused with mouse epidermal growth factor. Aust J Biol Sci. 1983;36(4):419–434. doi: 10.1071/bi9830419. [DOI] [PubMed] [Google Scholar]
- Houdebine LM. The production of pharmaceutical proteins from the milk of transgenic animals. Reprod Nutr Dev. 1995;35:609. doi: 10.1051/rnd:19950601. [DOI] [PubMed] [Google Scholar]
- Houdebine LM. Transgenic animal bioreactors. Transgenic Res. 2000;9(305–320):2000. doi: 10.1023/A:1008934912555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Javor A, Czeglédi A, Madai H, Vass N (2008) New trends in functional food of animal origin. Analele Universitatii Din Oradea Fascicula: Ecotoxicologie, Zootehnie Si Tehnologii De Industrie Alimentara vol VII:267–271
- Jube S, and Borthakur D (2006) Recent advances in food biotechnology research. In: Hui YH, Nip W-K, Nollet LML, Paliyath G, Sahlstrøm S, Simpson BK (eds) Food biochemistry and food processing. Blackwell Publishing, Oxford, pp 35–70
- Karatzas CN. Designer milk from transgenic clones. Nat Biotechnol. 2003;21:138–139. doi: 10.1038/nbt0203-138. [DOI] [PubMed] [Google Scholar]
- Karatzas CN, Turner JD (1997) Toward altering milk composition by genetic manipulation: current status and challenges. J Dairy Sci 80:2225–2232 [DOI] [PubMed]
- Karatzas CN, Zhou JF, Huang Y, Duguay F, Chretien N, Bhatia B, Bilodeau A, Keyston R, Tao T, Keefer CL, Wang B, Baldassare H, Lazaris A. Production of recombinant spider silk (BiosteelTM) in the milk of transgenic animals. Transgenic Res. 1999;8:476–477. [Google Scholar]
- Krasnov A, Pitkänen TI, Mölsä H (1999a) Gene transfer for targeted modification of salmonid fish metabolism. Genet Anal: Biomol Eng 15:115–119 [DOI] [PubMed]
- Krasnov A, Pitkänen I, Reinisalo M, Mölsä H. Expression of human glucose transporter type 1 and rat hexokinase type II complementary DNAs in rainbow trout embryos: effects on glucose metabolism. Marine, Biotechnol. 1999;1:25–32. doi: 10.1007/pl00011748. [DOI] [PubMed] [Google Scholar]
- Kerr DE, Wellnitz O. Mammary expression of new genes to combat mastitis. J Anim Sci. 2003;3(81):38–47. doi: 10.2527/2003.81suppl_338x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keys A, Anderson T, Grande F. Serum cholesterol response to changes in diet. IV. Particular saturated fatty acids in the diet. Metabolism. 1965;14:776. doi: 10.1016/0026-0495(65)90004-1. [DOI] [PubMed] [Google Scholar]
- Konakci KZ, Bohle B, Blumer R, Hoetzenecker W, Roth G, Moser B, Boltz-Nitulescu G, Gorlitzer M, Klepetko W, Wolner E, Ankersmit HJ. Alpha-gal on bioprostheses: xenograft immune response in cardiac surgery. Eur J of Clin Invest. 2005;35:17–33. doi: 10.1111/j.1365-2362.2005.01441.x. [DOI] [PubMed] [Google Scholar]
- Kris-Etherton PM, Yu S. Individual fatty acid effects on plasma lipids and lipoproteins: human studies. Am J Clin Nutr. 1997;65:1628–1644. doi: 10.1093/ajcn/65.5.1628S. [DOI] [PubMed] [Google Scholar]
- Lai L, Kang JX, Li R, Wang J, Witt WT, Yong HY, Hao Y, Wax DM, Murp hy CN, Rieke A,Samuel M, Linville ML, Korte SW, Evans RW, Starzl TE, Prather RS, Dai Y (2006) Generation of cloned transgenic pigs rich in omega-3 fatty acids. Nat Biotechnol 24:435–6 [DOI] [PMC free article] [PubMed]
- Lewis NM, Seburg S, Flanagan NL (2000) Enriched Eggs as a Source of N-3 Polyunsaturated Fatty Acids for Humans. Poultry Science 79:971–974 [DOI] [PubMed]
- Li R, Lai L, Wax D, Hao Y, Murphy CN, Rieke A, Samuel M, Linville ML, Korte SW, Evans RW et al (2006) Cloned transgenic swine via in vitro production and cryopreservation. Biol Reprod 75:226–30 [DOI] [PubMed]
- Lock AL, Parodi PW, Bauman DE. The biology of trans fatty acids: implications for human health and the dairy industry. Aust J Dairy Technol. 2005;60:134–142. [Google Scholar]
- Lock AL (2007) Nutritional importance of dairy fats In: Dairy nutrition and health [Edited by Bryans J et al]. The Dairy Council Issue, London, vol 1, pp 1–10
- Lo D, Pursel V, Linton PJ, Sandgren E, Behringer R, Rexroad C, Palmiter RD, Brinster RL. Expression of mouse IgA by transgenic mice, pigs and sheep. Eur J Immunol. 1991;21(4):1001–1006. doi: 10.1002/eji.1830210421. [DOI] [PubMed] [Google Scholar]
- Madan ML. Animal biotechnology: applications and economic implications in developing countries. Rev Sci Tech Off Int Epiz. 2005;24(1):127–139. [PubMed] [Google Scholar]
- Maga EA, Cullor JS, Smith W, Anderson GB, Murray JD. Human lysozyme expressed in the mammary gland of transgenic dairy goats can inhibit the growth of bacteria that cause mastitis and the cold-spoilage of milk. Foodborne Path Dis. 2006;3:384–392. doi: 10.1089/fpd.2006.3.384. [DOI] [PubMed] [Google Scholar]
- Malcarney HL, Bonar F, Murrell GA. Early inflammatory reactions after rotator cuff repair with porcine small intestine submucosal implant: a report of 4 cases. Am J Sports Med. 2005;33:907. doi: 10.1177/0363546504271500. [DOI] [PubMed] [Google Scholar]
- Massoud M, Attal J, Thepot D, Pointu H, Stinnakre M, Theron M, Lopez C, Houdebine L. The deleterious effects of human erythropoietin gene driven by the rabbit whey acidic protein gene promoter in transgenic rabbits. Reprod Nutr Devel. 1996;36:555–563. doi: 10.1051/rnd:19960511. [DOI] [PubMed] [Google Scholar]
- Mathews LS, Hammer RE, Behringer RR, D’Ercole AJ, Bell GI, Brinster RL, Palmiter RD. Growth enhancement of transgenic mice expressing human insulin-like growth factor I. Endocrinology. 1988;123:2827–2833. doi: 10.1210/endo-123-6-2827. [DOI] [PubMed] [Google Scholar]
- Mavromichalis A. Research into practice. Pig Prog. 2001;17(4):30–31. [Google Scholar]
- Mcpherron AC, Lawler AM, Le S-J. Regulation of skeletal muscle mass in mice by a new TGF-beta superfamily member. Nature. 1997;387:83–90. doi: 10.1038/387083a0. [DOI] [PubMed] [Google Scholar]
- Mead JR, Irvine SA, Ramji DP. Lipoprotein lipase: structure, function, regulation, and role in diseaseJ. Mol Med. 2002;80:753–769. doi: 10.1007/s00109-002-0384-9. [DOI] [PubMed] [Google Scholar]
- Metzler M, Gertz A, Sarkar M, Schachter H, Schrader JW, Marth JD. Complex asparagine-linked oligosaccharides are required for morphogenic events during post-implantation development. Embo J. 1994;13:2056–2065. doi: 10.1002/j.1460-2075.1994.tb06480.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller GY, Barlet PC, Lance SE, Anderson J, Heider LE. Cost of clinical masititis and mastitis prevention in dairy herds. J Amer Vet Med Assoc. 1993;202:1230–1236. [PubMed] [Google Scholar]
- Smith S, Witkowski A, Joshi AK. Structural and functional organization of the animal fatty acid synthase. Prog Lipid Resear. 2003;42:289–317. doi: 10.1016/s0163-7827(02)00067-x. [DOI] [PubMed] [Google Scholar]
- Montaldo HH. Genetic engineering applications in animal breeding. Electron J Biotech. 2006;9(2):157–170. [Google Scholar]
- Montel-hagen A, Kinet S, Manel N, Mongellaz C, Prohaska R, Battini J-L, Delaunay J, Sitbon M, Taylor N. Erythrocyte Glut1 triggers dehydroascorbic acid uptake in mammals unable to synthesize vitamin C. Cell. 2008;132(6):1039–1048. doi: 10.1016/j.cell.2008.01.042. [DOI] [PubMed] [Google Scholar]
- Mueckler M, Caruso C, Baldwin SA, Panico M, Blench I, Morris HR, Allard WJ, Lienhard GE, Lodish HF. Sequence and structure of a human glucose transporter. Science. 1985;229(4717):941–945. doi: 10.1126/science.3839598. [DOI] [PubMed] [Google Scholar]
- Muller M, Brenig B, Winnacker EL, Brem G. Transgenic pigs carrying cDNA copies encoding the murine Mx1 protein which confers resistance to influenza virus infection. Gene. 1992;121:263–270. doi: 10.1016/0378-1119(92)90130-h. [DOI] [PubMed] [Google Scholar]
- Murray Jd, Oberbauer AM, Mcgloughlin MM. Transgenic animals in agriculture. Davis: CABI Publishing; 1999. [Google Scholar]
- National Research Council (NRC) (1988) Designing foods: animal product options in the marketplace. National Academy Press, Washington DC [PubMed]
- National Research Council . Carcinogens and anticarcinogens in the human diet. Washington DC: National Academy Press; 1996. [PubMed] [Google Scholar]
- National Research Council (2002) 4 Food safety concerns. Animal biotechnology: science based concerns. National Academies Press 1 Print, Washington DC [PubMed]
- Nelson DL, Cox MM (2008) Lehninger, principles of biochemistry. W. H. Freeman and Company, New York. ISBN 978-0-7167-7108-1
- Niemann H. Transgenic farm animals get off the ground. Transgenic Res. 1998;7(1):73–75. [PubMed] [Google Scholar]
- Nottle MB, Nagashima H, Verma PJ, Du ZT, Grupen CG, McIlfatrick SM, Ashman RJ, Harding MP, Giannakis C, Wigley PL, Lyons IG, Harrison DT, Luxford BG, Campbell RG, Crawford RJ, Robins AJ (1999) Production and analysis of transgenic pigs containing a metallothionein porcine growth hormone gene construct. In: Murray JD, Anderson GB, Oberbauer AM, McGloughlin MM (eds) Transgenic animals in agriculture. CABI Publishing, Oxon, pp 145–156, 304 pp
- Palmiter RD, Brinster RL, Hammer RE, Trumbauer ME, Rosenfeld MG, Birnberg NC, Evans RM. Dramatic growth of mice that develop from eggs microinjected with metallothionein-growth hormone fusion genes. Nature. 1982;300:611–615. doi: 10.1038/300611a0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Palmiter RD, Norstedt G, Gelinas RE, Hammer RE, Brinster RL. Metallothionein-human GH fusion genes stimulate growth of mice. Science. 1983;222:809–814. doi: 10.1126/science.6356363. [DOI] [PubMed] [Google Scholar]
- Palmquist DL, Lock AL, Shingfield KJ, Bauman DE (2005) Biosynthesis of conjugated linoleic acid in ruminants and humans. In: Taylor SL (ed) Advances in food and nutrition research vol 50. Elsevier Inc., San Diego, pp 179–217 [DOI] [PubMed]
- Parodi PW. Milk fat in human nutrition. Aust J Dairy Tech. 2004;59:3–59. [Google Scholar]
- Pirottin D, Grobet L, Adamantidis A, Farnir F, Herens C, Daa Schroder H, Georges M. Transgenic engineering of male-specific muscular hypertrophy. Proc Natl Acad Sci USA. 2005;102:6413–6418. doi: 10.1073/pnas.0502426102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ponte PIP, Alves SP, Bessa RJB, Ferreira MA, Gama LT, Brás JLA, Fontes CMGA, Prates JAM. Influence of pasture intake on the fatty acid composition, and cholesterol, tocopherols, and tocotrienols content in meat from freerange broilers. Poult Sci. 2008;87(1):80–88. doi: 10.3382/ps.2007-00148. [DOI] [PubMed] [Google Scholar]
- Prather RS, Shen M, Dai Y. Genetically modified pigs for medicine and agriculture. Biotechnol Genet Eng Rev. 2008;25:245–266. doi: 10.7313/upo9781904761679.011. [DOI] [PubMed] [Google Scholar]
- Prieto PA, Kopchick JJ, Kelder B. Transgenic animals and nutrition research. J Nutr Biochem. 1999;10:682–695. doi: 10.1016/s0955-2863(99)00063-7. [DOI] [PubMed] [Google Scholar]
- Pszczola DE. Addressing functional problems in fortified foods. Food Technol. 1998;52(7):38–46. [Google Scholar]
- Pulina G, Nudda A, Battacone G, Cannas A. Effects of nutrition on the contents of fat, protein, somatic cells, aromatic compounds, and undesirable substances in sheep milk. Anim Feed Sci Tech. 2006;131(3–4):255–291. [Google Scholar]
- Pursel VG, Pinkert C, Miller K, Bolt D, Campbell R, Palmiter R, Brinster R, Hammer R. Genetic engineering of livestock. Science. 1989;244:1281–1288. doi: 10.1126/science.2499927. [DOI] [PubMed] [Google Scholar]
- Pursel VG, Hammer RE, Bolt DJ, Palmiter RD, Brinster RL. Integration, expression and germ-line transmission of growth-related genes in pigs. J Reprod Fertil Suppl. 1990;41:77–87. [PubMed] [Google Scholar]
- Pursel VG, Rexroad CE., Jr Recent progress in the transgenic modification of swine and sheep. Mol Reprod Dev. 1993;36:251–254. doi: 10.1002/mrd.1080360223. [DOI] [PubMed] [Google Scholar]
- Pursel VG, Wall RJ, Solomon MB, Bolt DJ, Murray JD, Ward KA. Transfer of ovine metallothionein-ovine growth hormone fusion gene into swine. J Anim Sci. 1997;75:2208–2214. doi: 10.2527/1997.7582208x. [DOI] [PubMed] [Google Scholar]
- Pursel VG, Wall RJ, Mitchell AD, Elasser TH, Solomon MB, Coleman ME, DeMayo F, Schwartz RJ (1999) Expression of insulin-like growth factor-1 in skeletal muscle of transgenic swine. In: Murray JD, Anderson GB, Oberbauer AM, McGloughlin MM (eds) Transgenic animals in agriculture. CABI Publishing, Oxon, pp 131–144, 304 pp
- Purvis IW, Franklin IR. Major genes and QTL influencing wool production and quality: a review. Genet Sel Evol. 2005;37(Suppl. 1):S97–S107. doi: 10.1186/1297-9686-37-S1-S97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rahman MA, Mak R, Ayad H, Smith A, Maclean N. Expression of a novel piscine growth hormone gene results in growth enhancement in transgenic tilapia (Oreochromis niloticus) Transgenic Res. 1998;7:357–369. doi: 10.1023/a:1008837105299. [DOI] [PubMed] [Google Scholar]
- Ratafia M. Worldwide opportunities in genetically engineered vaccines. Biotechnol. 1987;5:1154. [Google Scholar]
- Rexroad CE, Jr, Mayo K, Bolt DJ, Elsasser TH, Miller KF, Behringer RR, Palmiter RD, Brinster RL. Transferrin- and albumin-directed expression of growth-related peptides in transgenic sheep. J Anim Sci. 1991;69:2995–3004. doi: 10.2527/1991.6972995x. [DOI] [PubMed] [Google Scholar]
- Rexroad CE, Jr, Hammer RE, Bolt DJ, Mayo KE, Frohman LA, Palm-iter RD, Brinster RL. Production of transgenic sheep with growth-regulating genes. Mol Reprod Dev. 1989;1:164–169. doi: 10.1002/mrd.1080010304. [DOI] [PubMed] [Google Scholar]
- Rudolph NS. Biopharmaceutical production in transgenic livestock. Tibitech. 1999;17:367–374. doi: 10.1016/s0167-7799(99)01341-4. [DOI] [PubMed] [Google Scholar]
- Saeki K, Matsumoto K, Kinoshita M, Suzuki I, Tasaka Y, Kano K, Tagushi Y, Mikami K, Hirabayashi M, Kashiwazaki N, Hosoi Y, Murata N, Iritani A. Functional expression of a Δ12 fatty acid desaturase gene from spinach in transgenic pigs. Proc Nat Acad Sci USA. 2004;101(17):6361–6366. doi: 10.1073/pnas.0308111101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scheeder MRL, Casutt MM, Roulin M, Escher F, Dufey PA, Kreuzer M (2001) Fatty acid composition, cooking loss and texture of beef patties from meat of bulls fed different fats. Meat Sci 58 (3):321–328 [DOI] [PubMed]
- Scollan N, Hocquette J-F, Nuernberg K, Dannenberger D, Richardson I, Moloney A. Innovations in beef production systems that enhance the nutritional and health value of beef lipids and their relationship with meat quality. Meat Sci. 2006;74(1):17–33. doi: 10.1016/j.meatsci.2006.05.002. [DOI] [PubMed] [Google Scholar]
- Seidel GE (1999) The future of transgenic farm animals. In: Murray JD, Anderson GB, Oberbauer AM, McGloughlin MM (eds) Transgenic animals in agriculture. CABI Publishing, New York, pp 269–283
- Shamay A, Pursel V, Wilkinson E, Wall R, Hennighausen L. Expression of the whey acidic protein in transgenic pigs impairs mammary development. Transgenic Res. 1992;1:124–132. doi: 10.1007/BF02528777. [DOI] [PubMed] [Google Scholar]
- Shin T, Adams LG, Templeton JW, Westhusin ME (1999) Nuclear transfer using somatic cell line derived from a bull genetically resistance to brucellosis. Transgenic Res 8:488 (abstr)
- Simopoulos AP. Evolutionary aspects of diet, the omega-6/omega-3 ratio and genetic variation: nutritional implications for chronic diseases. Biomed Pharmacother. 2006;60:502–507. doi: 10.1016/j.biopha.2006.07.080. [DOI] [PubMed] [Google Scholar]
- Sloan AE. The top ten functional food trends. Food Technol. 2000;54(4):33–62. [Google Scholar]
- Soler E, Thepot D, Rival-Gervier S, Jolivet G, Houdebine L-M. Preparation of recombinant proteins in milkto improve human and animal health. Reprod Nutr Dev. 2006;46:579–588. doi: 10.1051/rnd:2006029. [DOI] [PubMed] [Google Scholar]
- Staeheli P, Haller O, Boll W, Lindemann NJ, Weismann C. Mx protein: constitutive expression in 3T3 cells transformed with cloned MxcDNA confers selective resistance to influenza virus. Cell. 1986;44:147–158. doi: 10.1016/0092-8674(86)90493-9. [DOI] [PubMed] [Google Scholar]
- Stinnakre MG, Vilotte JL, Soulier S, Mercier JC. Creation and phenotypic analysis of alphalactalbumin-deficient mice. Proc Nat Acad Sci USA. 1994;91(14):6544–6548. doi: 10.1073/pnas.91.14.6544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sayed N, Frans Y, Schönfeldt HC (1999) Composition of South African foods: milk and milk products, eggs, meat and meat products. Supplement to the MRC food composition tables 1991. South African Medical Research Council, Parow Valley, Cape Town
- Su HY, Jay NP, Gourley TS, Kay GW, Damak S. Wool production in transgenic sheep: Results from first-generation adults and second-generation lambs. Anim Biotechnol. 1998;9:135–147. doi: 10.1080/10495399809525900. [DOI] [PubMed] [Google Scholar]
- USDA (2008) Agricultural research service. USDA national nutrient database for standard reference, release 21. Nutrient data laboratory home page, www.ars.usda.gov/nutrientdata
- van-Berkel PHC, Welling MM, Geerts M, van-Veen HA, Ravensbergen B, Salaheddine M, Pauwels EKJ, Pieper F, Nuijens JH, Nibbering PH (2002) Large scale production of recombinant human lactoferrin in the milk of transgenic cows. Nat Biotechnol 20(5):484–487 [DOI] [PubMed]
- Varki A, Hooshmand F, Diaz S, Varki NM, Hedrick SM. Developmental abnormalities in transgenic mice expressing a sialic acid-specific 9-O-acetylesterase. Cell. 1991;65:65–74. doi: 10.1016/0092-8674(91)90408-q. [DOI] [PubMed] [Google Scholar]
- Wall R, Laible G, Maga E, Jr. Seidel G, Whitelaw B (2009) Council for agricultural science and technology (CAST). Animal productivity and genetic diversity: cloned and transgenic animals. Issue paper 43. CAST, Ames, Iowa
- Wall RJ, Pursel VG, Shamay A, McKnight RA, Pittius CW, Henninhausen L. High-level synthesis of a heterologousmilk protein in the mammary glands of transgenic swine. Proc Natl Acad Sci USA. 1991;88:1696–1700. doi: 10.1073/pnas.88.5.1696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wall RJ, Rexroad CE, Jr, Powell A, Shamay A, McKnight R, Hennighausen L. Synthesis and secretion of the mouse whey acidic protein in transgenic sheep. Transgenic Res. 1996;5:67–72. doi: 10.1007/BF01979923. [DOI] [PubMed] [Google Scholar]
- Wang R, Zhang P, Gong Z, Hew CL. Expression of the antifreeze protein gene in transgenic goldfish (Carassius auratus) and its implication in cold adaptation. Mol Mar Biol Biotechnol. 1995;4:20–26. [PubMed] [Google Scholar]
- Ward KA. Transgene-mediated modifications to animal biochemistry. Trends Biotechnol. 2000;18:99–102. doi: 10.1016/s0167-7799(99)01417-1. [DOI] [PubMed] [Google Scholar]
- Ward KA, Brown BW. The production of transgenic domestic livestock: successes, failures and the need for nuclear transfer. Reprod Fertil Dev. 1998;10:659–665. doi: 10.1071/rd98074. [DOI] [PubMed] [Google Scholar]
- Ward KA, Leish Z, Bonsing J, Nishimura N, Cam GR, Brownlee AG, Nancarrow CD. Preventing hair loss in mice. Nature. 1994;371:563–564. doi: 10.1038/371563b0. [DOI] [PubMed] [Google Scholar]
- Ward K, Nancarrow C, Murray J, Wynn P, Speck P, Hales J (1989). The physiological consequences of growth hormone fusion gene expression in transgenic sheep. J Cell Biochem B:164
- Weidle UH, Lenz H, Brem G. Genes encoding a mouse monoclonal antibody are expressed in transgenic mice, rabbits and pigs. Gene. 1991;98(2):185–191. doi: 10.1016/0378-1119(91)90172-8. [DOI] [PubMed] [Google Scholar]
- Wheeler MB. Production of transgenic livestock: promise fulfilled. J Anim Sci. 2003;81(Suppl. 3):32–37. doi: 10.2527/2003.81suppl_332x. [DOI] [PubMed] [Google Scholar]
- Wheeler MB, Monaco E, Bionaz M, Tanaka T (2010) The Role of existing and emerging biotechnologies for livestock production: toward holism. Acta Sci Vet 38(Supl 2):463–484
- Wheeler, MB (2012) Cloning and transgenic animals for agriculture. Biotecnologia da reprodução em bovinos (2o simpósio internacional de reprodução animal aplicada), pp181–194
- Wieghart M, Hoover J, McGrane M, Hanson R, Rottmen R, Holtzman S, Wagner T, Pinkert C. Production of swine harboring a rat phosphoenolpyruvate carboxykinase-bovine growth hormone fusion gene. J Reprod Fertil. 1990;41:89–96. [PubMed] [Google Scholar]
- Yang J, Ratovitski T, Brady JP, Solomon MB, Wells KD, Wall RJ. Expression of myostatin pro domain results in muscular transgenic mice. Mol Reprod Devel. 2001;60:351–361. doi: 10.1002/mrd.1097. [DOI] [PubMed] [Google Scholar]
- Yom H-C, Bremel RD. Genetic engineering of milk composition: modification of milk components in lactating transgenic animals. Am J Clin Nutr. 1993;58(suppl):299S–306S. doi: 10.1093/ajcn/58.2.299S. [DOI] [PubMed] [Google Scholar]
- Zhang J, Li L, Cai Y, Xu X, Chen J, Wu Y, Yu H, Yu G, Liu S, Zhang A, Chen J, Cheng G. Expression of active recombinant human lactoferrin in the milk of transgenic goats. Protein Expr Purif. 2008;57:127–135. doi: 10.1016/j.pep.2007.10.015. [DOI] [PubMed] [Google Scholar]


