Abstract
The conjunctiva is a goblet cell rich mucosal tissue. Goblet cells are supported by tear growth factors and IL-13 produced by resident immune cells. Goblet cell secretions are essential for maintaining tear stability and ocular surface homeostasis. In addition to producing tear stabilizing mucins, they also produce cytokines and retinoic acid that condition monocyte-derived phagocytic cells in the conjunctiva. Aqueous tear deficiency from lacrimal gland disease and systemic inflammatory conditions results in goblet cell loss that amplifies dry eye severity. Reduced goblet cell density is correlated with more severe conjunctival disease, increased IFN-γ expression and antigen presenting cell maturation. Sterile Alpha Motif (SAM) pointed domain epithelial specific transcription factor (Spdef) gene deficient mice that lack goblet cells have increased infiltration of monocytes and dendritic cells with greater IL-12 expression in the conjunctiva. Similar findings were observed in the conjunctiva of aged mice. Reduced retinoic acid receptor (RXRα) signaling also increases conjunctival monocyte infiltration, IFN-γ expression and goblet cell loss. Evidence suggests that dry eye therapies that suppress IFN-γ expression preserve conjunctival goblet cell number and function and should be considered in aqueous deficiency.
Keywords: conjunctiva, goblet cell, immune response, interferon gamma, immunoregulation, retinoic acid, retinoid receptor
1. Introduction
Dry eye is one of the most prevalent eye conditions, affecting more than 16 million patients in the US.1 It is a multifactorial disease characterized by a persistently unstable tear film that causes discomfort and visual impairment, and is accompanied by varied degrees of ocular surface epithelial disease and inflammation. Dry eye can be classified into aqueous deficient (due to lacrimal hyposecretion) and aqueous sufficient conditions (due to Meibomian gland disease or altered tear spread from conjunctivochalasis). Impression cytology studies have found that aqueous deficiency causes dysfunction and loss of conjunctival goblet cells, while goblet cell number has been reported to remain in the normal range in Meibomian gland disease.2 There are numerous causes for aqueous deficiency, including aging, anticholinergic medications, systemic inflammatory/immune diseases (such as Sjögren syndrome) as well as diseases that affect neural innervation or signaling.
2. Conjunctival Goblet Cell Physiology and Function
The conjunctiva is a goblet cell rich mucosal tissue. The growth factor EGF in tears and the Th2 cytokine IL-13 produced by resident immune cells support the goblet cells by stimulating protein synthesis and proliferation.3–5 Cholinergic neurotransmitters stimulate reflex mucin secretion by the goblet cells.3 The conjunctival goblet cells express the gel-forming mucin genes MUC5AC, MUC5B (in a subpopulation) and MUC2.6–9 Among these, MUC5AC is the major goblet cell secretory mucin that is present in human tears.10,11 Goblet cell mucins function to maintain ocular surface hydration, tear stability and clearance of pathogens and debris.12,13 It remains to be determined if goblet cell secreted mucin adheres to the membrane tethered mucins (MUC1, MUC4, MUC16) produced by the surface epithelium. It is increasingly recognized that in addition to producing mucins, the goblet cells are essential components of the conjunctival mucosal immune system. The conjunctival goblet cells serve as antigen passages from the ocular surface to mononuclear phagocytic cells in the stroma14, and they produce immunoregulatory factors, such as TGF-β215 and retinoic acid (RA)16 that condition these monocyte derived cells by suppressing cytokine production and maturation.
The lacrimal gland takes up vitamin A from the blood and secretes it in the retinol form into the tears.17,18 Studies reported by our group show that conjunctival goblet cells express alcohol (ADH) and aldehyde (ALDH) dehydrogenase enzymes that metabolize retinol into retinoic acid (RA), the biologically active form of vitamin A.16 Lacrimal gland dysfunction can reduce goblet cell RA production because of decreased retinol secretion, altered corneal/conjunctival epithelial differentiation with reduced ALDH expression and conjunctival goblet cell loss.19,20 Evidence from our lab presented below suggests that reduced RA production resulting from goblet cell loss worsens conjunctival inflammation and ocular surface disease. Reduced retinoid signaling in systemic vitamin A deficiency also results in conjunctival squamous metaplasia, goblet cell loss and blinding corneal opacification, vascularization and ulceration.21,22
3. Goblet Cell Loss in Dry Eye Worsens Ocular Surface Disease/Inflammation
Goblet cell loss develops in aqueous deficient dry eye due to hyposecretory lacrimal gland disease/dysfunction and is associated with systemic/ocular surface inflammatory diseases, such as Sjögren syndrome, Stevens-Johnson syndrome and graft vs. host disease (GVHD).23–26 A number of studies have found conjunctival goblet cell loss is correlated with clinical severity and level of ocular surface inflammation in aqueous tear deficiency. Eye irritation severity measured with the OSDI questionnaire was found to be inversely correlated with goblet cell density.27 A significant inverse correlation was found between categorical severity of Sjögren syndrome associated dry eye using the Dry Eye Workshop scale and goblet cell density in the temporal and superior bulbar conjunctiva.28 Goblet cell density in the temporal bulbar conjunctiva was found to inversely correlate with Rose Bengal staining score at that site and with the staining score of the entire exposure zone.25 Goblet cell density was also noted to be inversely correlated with expression of the cytokine interferon gamma (IFN-γ) in the bulbar conjunctiva29 and with the percentage of HLA-DR positive cells obtained in impression cytology.28 Dry eyes due to Stevens-Johnson syndrome and Sjögren syndrome that have significant goblet cell loss are at risk for developing sight-threatening corneal ulceration and opacification that in some cases can occur bilaterally.30–32
The cytokine IFN-γ, produced by T helper 1 (Th1), natural killer (NK) and monocyte-derived cells, is well recognized to cause secretory dysfunction, induction of an unfolded protein response and death of the conjunctival goblet cells.33–35 Expression of IFN-γ and chemokines it induces, such as CXCL-10 have been found to increase in the conjunctiva in aqueous tear deficiency, particularly Sjögren syndrome29,36, and increased tear concentrations of IFN-γ signature cytokines/chemokines have been reported in dry eye, with the highest concentrations in aqueous tear deficiency (Table 1). Increased IFN-γ expression is associated with worse clinical disease and goblet cell loss.29 Experimental murine models have also shown that adoptively transferred IFN-γ producing CXCR3+CD4+ T cells from desiccating stress primed donors cause extensive goblet cell loss in naïve immunodeficient recipients.37 In summary, these studies report increased tear concentrations of IL-12, a cytokine produced by antigen presenting cells that stimulates production of IFN-γ by T cells and monocyte derived cells, IFN-γ and chemokines that are induced by IFN-γ (i.e. CXCL9, CXCL10, CXCL11), and they indicate that increased IFN-γ and goblet cell loss in the conjunctiva create a self-amplifying immune cycle of dry eye 38. On the other end of the immune response spectrum, increased expression of the Th2 cytokine IL-13 in conditions such as atopic keratoconjunctivitis (AKC) can cause goblet cell hyperplasia.39,40
Table 1.
IFN-γ Signature Cytokines/Chemokines in Dry Eye Tears
| Author – Year | Study Group | Findings |
|---|---|---|
| Chen X et al. - 2019105 | Sjögren syndrome (SS) ATD | • Significantly increased IL-12p70, IFN-γ, CXCL10 (IP-10) vs. non-SS ATD and normal control, correlated with conjunctival and corneal staining |
| Enriquez-de-Salamanca et al. - 2010106 | Evaporative dry eye | • CXCL10 was significantly increased compared to normal control |
| Jackson et al. - 2016107 | Dry eye | • Tear IFN-γ level > 3 fold higher in hyperosmolar group (P = 0.03). Tear IFN-γ was significantly correlated with tear osmolarity and total ocular surface dye staining. |
| Shetty et al. - 2016108 | Dry eye | • Significantly higher levels of IFN-γ was observed in the tears of patients with compared with low serum vitamin D concentration vs. controls (P < 0.05) |
| Agrawal et al. - 2016109 | HIV associated dry eye | • CXCL10 increased compared to dry eye without HIV infection. |
| Riemens et al. - 2012110 | Ocular GVHD | • Following allogenic bone marrow stem cell transplant, IFN-γ increased in GVHD vs. no GVHD. Tear IFN-γ inversely correlated with tear break up time. |
| Meadows et al. - 2016111 | 4 subtypes of dry eye | • IFN-γ high in all 4 subtypes of dry eye with highest concentration in ATD that had most severe ocular surface dye staining. |
| Zywalewska-Gorna et al. - 2007112 | SS ATD | • IFN-γ increased vs control and correlated with severity of symptoms and signs. |
| Zhao H et al. - 2018113 | ATD | • IFN-γ higher and IL-12p70 significantly increased (P<0.05) in ATD vs control. |
| Yoon KC et al. - 2010114 | SS ATD | • CXCL9, −10, −11 concentrations significantly increased in tears of SS vs. non-SS dry eye (P < 0.05). CXCL11 significantly correlated (P< 0.05) with keratoepitheliopathy score (positive) and goblet cell density (negative). |
ATD: aqueous tear deficiency; SS: Sjögren syndrome; C-X-C-L: motif chemokine ligand
Consistent with the finding that IFN-γ promotes conjunctival goblet cell loss, preclinical studies in mouse dry eye models have shown that increased IFN-γ expression is associated with conjunctival goblet cell loss and that therapies capable of suppressing dry eye inducing immune mediators, such as IFN-γ, can increase goblet cell density (Table 2).
Table 2.
Immune mediated goblet cell loss/prevention in mouse dry eye models
| Author/year | Animal model | Strain | Principal Findings |
|---|---|---|---|
| Strong/2005115 | DS | C57BL/6 | • Topical cyclosporine A significantly inhibited DS induced conjunctival epithelial apoptosis and goblet cell loss |
| Niederkorn/2006116 | DS | C57BL/6 | • CD25 depletion during DS worsens GC loss |
| de Paiva/2007117 | DS | C57BL/6/IFN-γKO | • DS increases GC loss in wild type, but IFN-γKO are resistant to dry eye-induced GC loss • GC loss and expression of cornification marker SPRR2 were increased by subconjunctival IFN- γ injection during DS in wild type and IFN-γKO. |
| Yoon/2007118 | DS | NOD.B10.H2b | • Decreased GC density at 16 weeks of age compared to 4-week-old mice. • Further decrease in GC density under DS |
| Yeh/2009119 | Spontaneous autoimmune dry eye | C57BL/6/AIREKO | • AIREKO strain have decreased GC density compared to wild-type mice |
| de Paiva/20104 | DS | C57BL/6/IL-13KO | • Naïve IL-13KO mice have decreased GC density compared to wild-type • Exogenous administration of IL-13 during DS prevents GC loss • Cyclosporine A administration during DS increases GC density, while decreasing IFN-γ |
| de Paiva/2010120 de Paiva/2015121 |
Spontaneous autoimmune dry eye | CD25KO | • CD25KO has increased concentration of IFN-γ in tears compared to wild type • Decreased GC density in CD25KO compared to wild-type • CD25-IFN-γ double KO have greater GC density compared to parental CD25KO strain |
| Schaumburg/2011122 | DS/Adoptive transfer | C57BL/6 | • Clodronate treatment to deplete APCs during DS improved GC density and decreased pathogenicity of transferred CD4+T cells. |
| de Paiva/2011123 | Spontaneous autoimmune dry eye | DN-TGF-BRII | • Decreased GC density compared to wild-type • Increased IFN-γ mRNA in conjunctiva |
| Zhang/2011124 | DS/Adoptive transfer | C57BL/6 | • IFN-γ neutralization in RAG1KO recipients decreased conjunctival apoptosis after adoptive transfer |
| de Paiva/2012125 | DS | C57BL/6 | • Topical treatment with Resolvin E1 prevents DS-induced goblet cell loss. |
| Li/2012 126 | DS | C57BL/6 | • Topical anti-TNF-α during DS decreases frequency of CD4+CXCR3+ cells in the conjunctiva and improves GC density. |
| Zhang/2013127 | DS | C57BL/6 | • IFN-γ neutralization during DS improves GC density • Adoptive transfer recipients from anti IFN-γ treated have attenuated dry eye phenotype |
| Coursey/2013128 | DS/Adoptive transfer | C57BL/6/CXCR3KO | • CXCR3KO mice are resistant to DS induced GC loss • Decreased production of IFN-γ in the ocular surface • Adoptive transfer of CXCR3KO CD4+T cells primed during DS do not induce GC loss in recipients |
| McClellan/201467 Bian/2018129 |
Aging/Adoptive transfer | C57BL/6 | • Loss of GC starts around 9 months of age • Adoptive transfer of aged CD4+T cells transfer dry eye phenotype, inclusive of GC loss • Aged APCs prime greater frequency of CD4+IFN-γ+ than young APCs • Adoptive transfer of aged CD4+CXCR3+T cells induce greater GC loss than young CD4+CXCR3+ cells. |
| Krauss/2015130 | DS | C57BL/6 | • Topical treatment with a4b1 integrin prevents DS-induced goblet cell loss |
| You/2015131 | Spontaneous autoimmune dry eye | AEC | • Decreased GC density compared to wild-type • Increased IFN-γ mRNA in conjunctiva and lacrimal gland • Increased frequency of CD4+CXCR3+ cells in lacrimal gland |
| Coursey/2016132 | DS | C57BL/6 | • Subconjunctival anti-IFN-γ receptor antibody injection decreases expression of unfolded protein response genes |
| Volpe/201680 | Aging | C57BL/6/IFN-γKO | • IFN-γKO mice are partially protected from age-induced GC loss |
| Choi/2016 133 | DS | C57BL/6 | • Anti-oxidant topical therapy during DS decreased IFN-γ protein levels in CJ, improved GC density and decreased the frequency of CD4+CXCR3+ cells |
| Coursey/2017134 | Aging/adoptive transfer | NOD.B10.H2b | • Aged mice develop GC loss • Increased IFN-γ in conjunctiva, lacrimal gland,serum and splenocytes • Adoptive transfer of aged CD4+T cells transfer dry eye phenotype, inclusive of GC loss |
| Portal/2017135 | BAK | C57BL/6 | • Recombinant IL-13 prevents goblet cell density in BAK treated mice |
| Zhang/2017136 | DS | C57BL/6 | • Topical application of Mizoribine improved GC density |
| Li/2017 137 | DS | C57BL/6 | • Topical adiponectin application during DS decreases IFN-γ protein concentration in conjunctiva, improves GC density and decreases the frequency of CD4+CXCR3+ cells. |
| de Souza/201845 | DS | C57BL/6 | • Topical treatment with Lifitegrast improves GC density while decreasing IFN-γ, CXCL9 and CXCL10 expression in conjunctiva. |
| Wang/2018138 | Germ-free | C57BL/6 | • Spontaneous SS-like KCS, with significant GC loss compared to conventional mice • Increased levels of IL-12+ cells in conjunctiva and greater frequency of CD4+IFN-γ+ cells in the lacrimal gland |
| Zaheer/2018139 | Germ-free/Adoptive transfer | CD25KO | • Early onset and worse dacryoadenitis in germ-free CD25KO mice and greater spontaneous frequency of CD4+IFN-γ+ cells • IL-12 antibody depletion decreases CD4+IFN-γ+ infiltration in lacrimal gland and improves dacryoadenitis |
| Ko/201888 | Naïve | C57BL/6/SPDEFKO | • SPDEF KO mice have no goblet cells • SPDEF KO APCs prime greater frequency of CD4+IFN-γ+ than wild-type • Loss of mucosal tolerance |
| You/2018 140 | DS | C57BL/6 | • Topical treatment with 0.3% and 0.18% hyalorunic acid improved GC density |
| Li/2019 141 | DS | B6/NOD2KO | • NOD2KO mice are resistant to DS-induced changes • Greater GC density and decreased tear IFN-γ protein levels in NOD2KO mice |
Abbreviations: DS=desiccation stress; KO= gene knockout mouse strain; GC= goblet cell; APC= antigen-presenting cells; BAK= benzalkonium chloride; DS= desiccating stress; GC= goblet cell; KO= knock-out; CXCR3: C-X-C motif chemokine receptor 3; SS= Sjogren syndrome; KCS= keratoconjunctivitis sicca
An increase in goblet cell density has also been noted in human clinical trials. In tertiary studies performed for the FDA Phase 3 clinical trials of cyclosporine A (CsA) emulsion for dry eye, a significant increase in goblet cell density was observed in eyes with aqueous deficiency treated with CsA for 6 months (increases of 198% in SS and 234% in non SS ATD) vs. vehicle that had a mean decrease of 95%.41 A Cochrane review of 30 randomized controlled clinical trials of topical CsA therapy of dry eye concluded the effect of CsA on eye discomfort and clinical markers of dry eye, such as tear break-up time, Schirmer test and corneal fluorescein staining was not statistically different from vehicle or artificial tears; however, evidence indicates that CsA may be superior to control in increasing the number of conjunctival goblet cells.42 Trials of a corticosteroid (fluorometholone) or vitamin A (retinal palmitate), both ligands of nuclear receptors that regulate transcription of inflammatory genes were also reported to increase conjunctival goblet cell density.43,44 The LFA-1 antagonist, Lifitigrast was found to significantly increase conjunctival goblet cell density compared to vehicle in a mouse desiccating stress model.45 There are no reported studies evaluating its effects on goblet cells in humans. These findings highlight the suppressive effects of IFN-γ on conjunctival goblet cell number and function.
4. Aging and Conjunctival Goblet cells
Chronological aging has repeatedly been reported as a risk factor for dry eye 46–57, although the mechanisms by which aging predisposes to dry eye have not been fully elucidated. Two large epidemiological studies noted that dry eye prevalence increases in women and men after the age of 50, with higher prevalence in women, compared to men.50,55
Changes in tear meniscus height, lid laxity, increased inflammatory mediators in tears and decreased corneal nerve density are all alterations found in the aging eye. 58–63 Aging is accompanied by an increase in inflammatory markers, including IL-6, TNF-α and IFN-γ in multiple organ systems.64–71 This chronic pro-inflammatory stage has been termed inflamm-aging.72 Among these cytokines, IFN-γ is particularly relevant because it induces apoptosis of the corneal, conjunctival (including goblet cells), and lacrimal gland acinar epithelium.35,73–75 IFN-γ also promotes maturation of APCs that can prime autoreactive T cells.76 Increased frequency of IFN-γ-producing cells has also been noted in aged mice and humans.77,78 There is an increase in CD4+ cells, as well as a decrease in the number of conjunctival goblet cells in the aging mouse conjunctiva (Figure 1).67,79,80 Evidence from animals studies has shown that desiccation-induced goblet cell loss is prevented with topically applied IFN-γ neutralizing antibody and age associated goblet cell loss is significantly lower in IFN-γ deficient mice.75,80 Other factors contributing to the inflammatory microenvironment of the aged conjunctiva include decreased frequency of ALDH+ RA-producing cells19, and increased APC priming of pathogenic Th1 cells that cause greater goblet cell loss when adoptively transferred to naïve immunodeficient recipients.19 These findings suggest that immune-mediated conjunctival goblet cell loss is a component of age induced dry eye.
Figure 1.
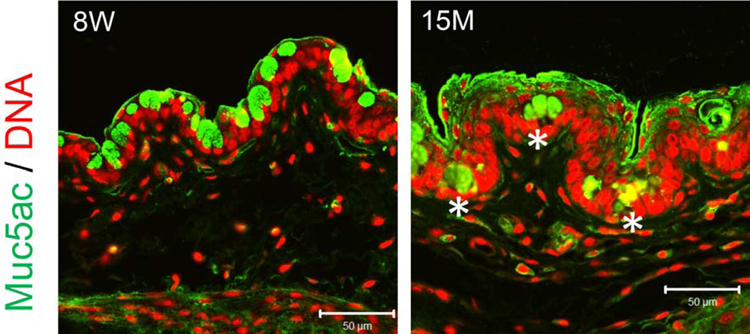
Representative images of palpebral conjunctival cryosections stained for Muc5ac (green) and propidium counterstaining (DNA, in red) of 8-week-old (8W) and 15-months-old (15M) female C57BL/6 mice. Note that some Muc5ac+ cells are buried in the aged conjunctival epithelium (asterisks) and therefore unable to discharge to the ocular surface.
5. Ocular Surface inflammation develops with loss of goblet cells in SPDEF Knockout
Goblet cell loss is associated with higher expression of the cytokine IFN-γ in human dry eye and in mouse dry eye models suggesting that goblet cell secretory products have an immunomodulatory function.20,75 Transcription factor Sterile Alpha Motif (SAM) pointed domain epithelial specific transcription factor (SPDEF) is essential for goblet cell differentiation in the lungs81, intestine82 and conjunctiva.83 Induced expression of SPDEF in the lung or intestinal epithelium promotes goblet cells differentiation and increased mucus production.82,84 Although, exogenous administration of the Th2 cytokine IL-13 increases conjunctival goblet cell number, IL-13−/− mice demonstrate only a 15% reduction in goblet cell number, as compared to total loss in SPDEF−/− strain, which confirms the essential role of SPDEF in goblet cell differentiation and homeostasis.83,85 SPDEF acts as an immunomodulatory factor on the airway epithelium by regulating goblet cell differentiation and mucus production.86 SPDEF overexpression in chronic lung disorders, such asthma or experimental over-expression in the airway epithelium of neonatal mice stimulates a Th2-mediated inflammatory response.87 SPDEF−/− mice lack conjunctival goblet cells and develop cornea epithelial disease with increased uptake of fluorescein dye as an indicator of epithelial barrier disruption.83 SPDEF−/− mice manifest a significantly increased number of CD45+ inflammatory cells in the conjunctiva, as well as APCs consisting of CD11c+ in the superficial conjunctiva and CD11b+ cells in the deep conjunctival epithelium and stroma.83,88 Pro-inflammatory cytokines, including IL-1α, IL-1β and TNF-α were upregulated, while epithelial cell differentiation markers, such as Muc5ac, Foxa3, and Tff1 were down regulated in the conjunctiva of SPDEF−/−.83
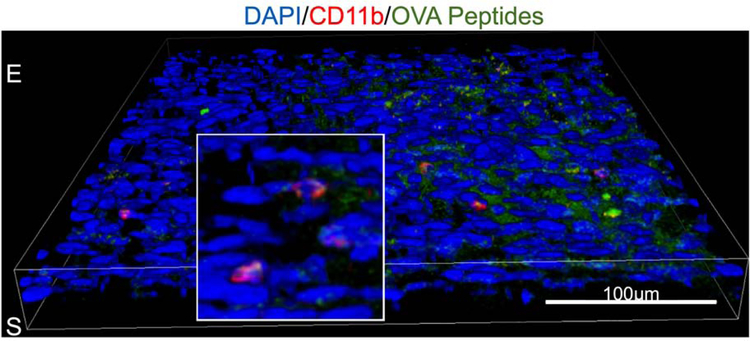
Goblet cells produce immunomodulatory factors, such as TGF-β2, RA and Muc2 which suppress maturation and condition tolerogenic properties in stromal APCs.14,15,89 Goblet cell associated passages (GAPs) serve as conduits for passage of antigens bound to goblet cell mucin into the stroma.14 In SPDEF−/− mice lacking goblet cells, topically applied OVA-antigen was retained within the conjunctival epithelium.14 We found that resident phagocytic CD11b+ cells in the conjunctival stroma sample OVA-antigen when applied topically on the conjunctiva in WT C57BL/6 mice (Figure 2). Topically applied OVA antigen induces T cell immune tolerance90; however, tolerance to OVA-antigen is lost when antigen administration is started after three days of systemic cholinergic blockade and exposure to desiccation stress which inhibits goblet cell secretion, suggesting that the conjunctival goblet cells have a tolerogenic effect on the resident APCs.91 This is consistent with the presense of an increased number of IL-12+ macrophages and dendritic cells in the conjunctiva of the SPDEF−/−.88 Antigen specific CD4+ T cells primed by APCs isolated from SPDEF−/− cervical lymph nodes exhibit greater proliferation with a lower frequency of CD4+Foxp3+ regulatory T cells and increased frequency of CD4+IFN-γ+ and CD4+IL-17+ cells.88 Topical application of conjunctiva conditioned media from wild type mice or RA inhibited LPS stimulated IL-12 expression in the SPDEF−/− conjunctiva, suggesting that RA is an important goblet cell-produced factor that suppresses APC activation in the conjunctiva.88 Indeed, conjunctival goblet cells express aldehyde dehydrogenase ALDH1A3, show aldehyde dehydrogenase activity and produce biologically active RA that was found to significantly inhibit IL-12 production in LPS-treated cultured bone marrow monocyte derived cells.16
Figure 2.
Confocal microscopy of whole mount conjunctiva 2 hours after topical application of fluorescent OVA peptide showing CD11b+ cells (red) beneath conjunctival epithelium that have phagocytosed the OVA peptide (green). Conjunctival epithelium is labeled E and the stroma S. Nuclei are stained blue with DAPI.
6. Goblet cell products modulate antigen presenting cell function
Conjunctival goblet cells, located at the interface of external environment and stromal immune cells, secrete tear mucins that coat the ocular surface and confer protection from adverse environmental conditions, foreign bodies or pathogens.12,14 The goblet cells also release immunomodulatory factors, such as Muc2, retinoic acid (RA), TGF-β1 and -β2 that function in maintaining immunological tolerance on the ocular surface.16,15,89 Muc2 is the major immune regulatory product of goblet cells in the small intestine; however, Muc2 expression also has been detected in human and mouse conjunctival goblet cells, albeit at a lower level than MUC5AC in the human conjunctiva.5,7,89,92 MUC2 was reported to increase production of the anti-inflammatory cytokine IL-10 and suppress production of IL-12 and T cell costimulatory markers CD80 and CD86 in LPS stimulated myeloid dendritic cells via NFκB inhibition.89 Compared to cultured corneal epithelium, conjunctival goblet cell epithelium was also found to have greater expression of alcohol (AD4) and aldehyde (ALdh1a1, ALdh1a3) dehydrogenases, key enzymes that are required to metabolize retinoic acid (RA) from the retinol form of vitamin A that is secreted by the lacrimal gland in the tears.93 RA has been reported to inhibit LPS-stimulated IL-12 production by mouse macrophages in a dose dependent manner.94 Similarly, our group reported that RA in conjunctival goblet cell conditioned media suppressed expression of IFN-γ family cytokines IL-12 and IFN-γ and CD86 in LPS-stimulated cultured myeloid cells (monocytes and macrophages).16 Moreover, goblet cell factors suppressed NFκB p65 activation and expression of NFκB inducible genes, CCR7 and ICAM-1 in these cells.16 Goblet cell conditioned, OVA-loaded APCs suppressed production of IFN-γ and increased production of the Th2 cytokine IL-13 in an OTII co-culture system, an activity attributable to their ability to synthesize RA.16
7. Suppression of monocyte activation in conjunctiva is RXRα mediated
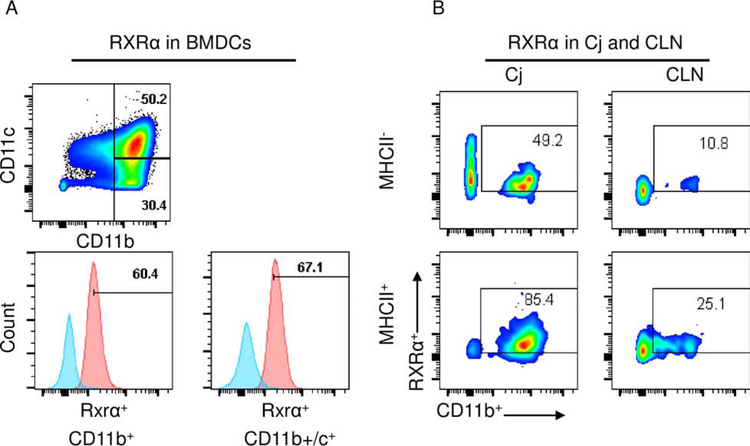
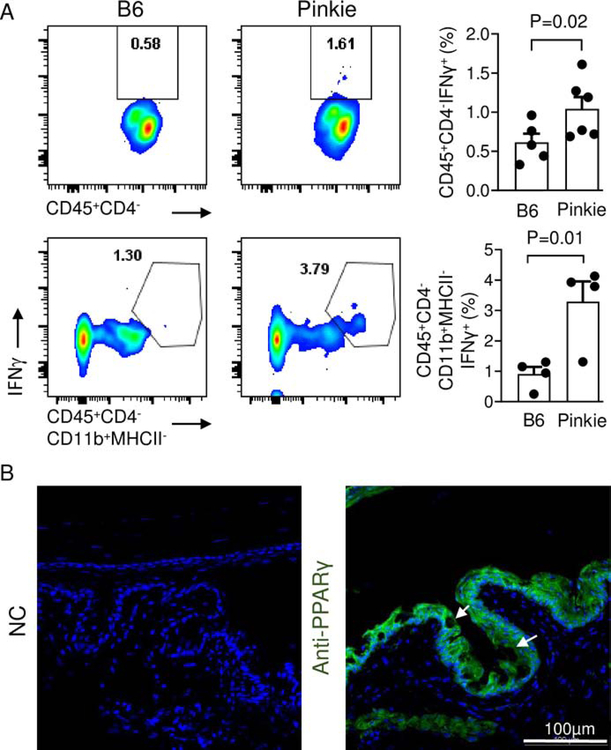
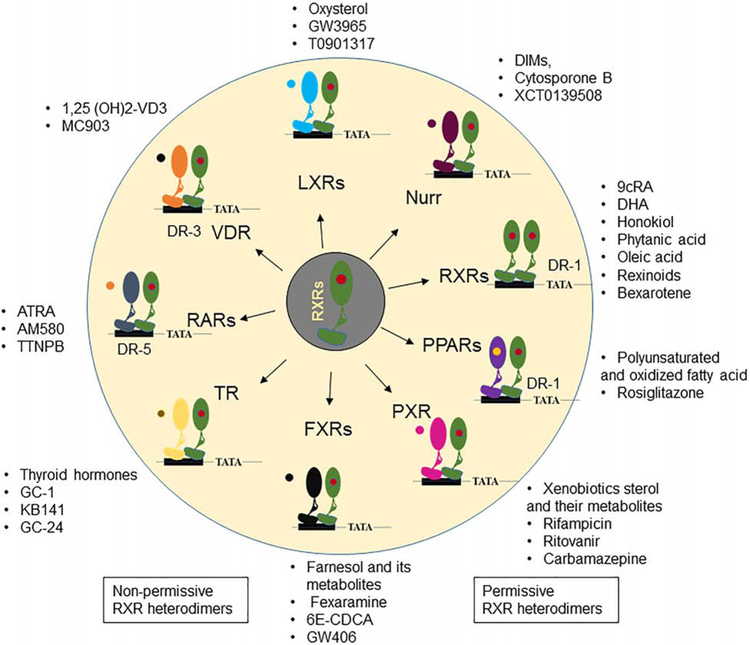
RA exists in vivo as two isomers, 9-cis RA and all-trans RA (ATRA). These isoforms have affinity to heterodimeric nuclear retinoid receptors: 9-cis RA binds to the retinoid receptor X (RXR) and ATRA binds to the retinoic acid receptor (RAR). Once activated, these nuclear receptors regulate transcription of a wide range of genes, including inflammatory and immune response genes. We have discovered that the RXRα nuclear receptors are particularly relevant for suppressing production of dry eye inducing inflammatory mediators by innate immune cells. The RXRα isoform is expressed in the majority of bone marrow derived myeloid cells (Figure 3A) and by > 85% of MHCII+CD11b+ cells in the conjunctiva, while only a quarter of these cells are RXRα positive in the draining cervical lymph nodes (Figure 3B). Compared to wild type C57BL/6, we have found the Pinkie mouse strain with a loss of function RXRα mutation95 has a 39% decrease in conjunctival goblet cell density (P=0.0007). This was accompanied by an increased percentage of IFN-γ positive CD11b+ monocytes which were the predominant IFN-γ producing cell type in the conjunctiva (Figure 4A), suggesting that IFN-γ from these cells contributes to the goblet cell loss.96 RXR dimerizes with a number of partner nuclear receptors (summarized in Figure 5), including those with reported immunoregulatory activity on the ocular surface: vitamin D, peroxisome proliferator-activated gamma (PPARγ) and liver X (LXR) receptors.97–100 RXR heterodimers are classified as permissive when the complex can be activated by either an RXR ligand [e.g. 9-cis RA or docosahexaenoic acid (DHA) in fish oil] or a ligand of the heterodimeric partner (e.g PPAR). Non permissive heterodimers are activated only by the ligands that are specific for the partner nuclear receptors (e.g ATRA, vitamin D or thyroid hormone), with RXR ligands acting as a silent partner.101 Treatment with the RXRα ligand DHA [together with essential fatty acids eicopentaenoic acid (EPA) and gamma linoleic acid (GLA)] was reported to improve dry eye symptoms and prevent an increase in CD11c+ cells in the conjunctiva epithelium during the treatment period.102 PPAR-γ expression has been reported in the meibomian glands103 and we’ve found it is also expressed by the goblet and non-goblet conjunctival epithelium (Figure 4B). Expression of RXRα and the retinol metabolizing enzyme ALdh1a1 were reported to be decreased 4- and 26-fold, respectively, in the conjunctiva of patients with Stevens-Johnson syndrome, a disease characterized by severe or total conjunctival goblet cell loss.104
Figure 3.
A. Flow cytometry was performed on cultured bone marrow derived cells (BMDCs) gated on CD11c and CD11b and the percentage of cells positive for the retinoid X receptor alpha (RXRα) was evaluated. Over 60% of CD11b+ and CD11b+CD11c+ cells were RXRα+; B. The percentage of CD11b+RXRα+ MHCII positive and negative cells in the conjunctiva and draining cervical lymph nodes was evaluated by flow cytometry. The percentage of RXRα+ cells was higher in the conjunctiva than the cervical nodes.
Figure 4.
A. The percentages of CD45+CD4−IFN-γ+ (top) and CD45+CD4-CD11b+ IFN-γ+ (bottom) cell populations in conjunctival tissue obtained from C57BL/6 and Pinkie mouse strains were evaluated by flow cytometry. Both cell populations were significantly higher in the Pinkie strain (bar graphs, right side). B. Mouse conjunctival sections stained for RXRα partner nuclear receptor peroxisome proliferator-activated receptor gamma (PPARγ). Secondary antibody negative control (NC) on the left and PPARγ antibody staining on the right. Arrows indicate goblet cells.
Figure 5.
Retinoid X receptors (RXRs) dimerize with other partner nuclear receptors. Active RXRs regulate gene transcription by forming permissive heterodimers with fernesoid X receptor (FXR), pregnan X receptor (PXR), peroxisome proliferator-activated receptor (PPARs), Nurr1 and Nurr7, and liver X receptors (LXRs) and non-permissive heterodimers with thyroid receptors (TRs), retinoic acid receptor (RAR) and vitamin D receptor (VDR).
Expression of TGF-β2, another immunomodulatory factor produced by goblet cells was noted to increases in response to TLR4 mediated stimuli.15 Goblet cells activate TGF-β2 in a thrombospondin-1 dependent manner which can condition APCs towards a tolerogenic phenotype by down regulating expression of MHC class II and costimulatory molecules CD80 and CD86.15
8. Conclusions
Goblet cells in the conjunctiva play an essential immunomodulatory role by producing factors, such as TGF-β2 and retinoic acid that condition phagocytic cells in the conjunctival stroma, suppressing their maturation and production of dry eye inducing cytokines, such as IFN-γ (Figure 6). Currently there are no widely available clinical tests to evaluate goblet cell number and function, expression of retinoic acid metabolizing enzymes in the conjunctiva or concentration of IFN-γ in tears. Availability of these clinical biomarkers would improve ability of identify patients who might benefit from topical retinoid receptor agonist therapy to preserve or improve goblet cell number and function. These findings also suggest the need for topical RXRα agonists to suppress production of IFN-γ family cytokines by innate immune cells in the conjunctiva.
Figure 6.
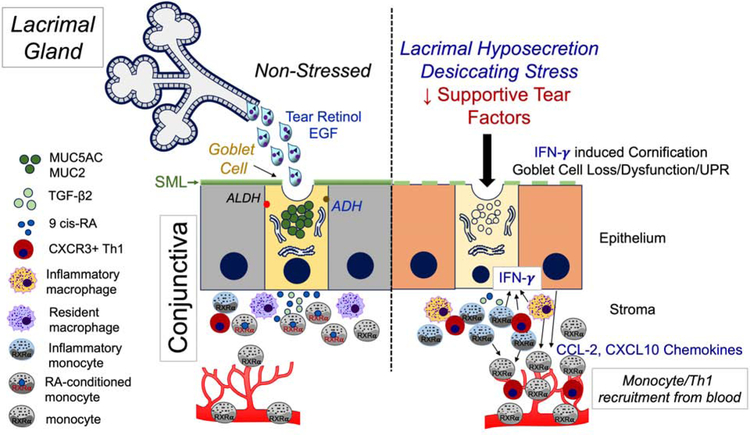
Immune - goblet cell interaction in the conjunctiva. During normal non-stressed conditions (left side), the lacrimal gland secretes tears containing epidermal growth factor (EGF) and vitamin A in the form of retinol. EGF supports goblet cell protein synthesis and proliferation and retinol is taken up by the goblet cells and metabolized by alcohol (ADH) and aldehyde (ALDH) dehyrogenases into the biologically active form retinoic acid (RA) that exists in equilibrium between the all trans- and 9-cis isomers. RA and TGF-β2 condition mononuclear phagocytic cells, including monocytes and macrophages in the conjunctiva. RA signaling through nuclear receptors, including the nuclear receptor RXRα in monocyte-derived cells, suppresses differentiation to inflammatory phenotypes that have higher expression levels of IFN-γ, IL-12 and antigen presenting cell maturation markers such as CD86. When lacrimal gland secretory function is reduced and the ocular surface is exposed to desiccation or other danger signals (right side), goblet cell number and function decreases and there is reduced conditioning of resident and recruited monocyte-derived cells resulting in increased expression of IFN-γ and IL-12 and monocyte and Th1 chemokines, such as CCL-2 and CXCL10, respectively, by the surface epithelium and monocytes. IFN-γ stimulates expression of cornifying genes, inhibits cholinergic signaling and induces an unfolded protein response (UPR) and apoptosis in the conjunctival goblet cells, reducing the secretory mucin layer (SML) and further amplifying ocular surface inflammation and epithelial disease.
Acknowledgements:
This work was supported by NIH Grant EY11915 (SCP), NIH Core Grants-EY002520 & EY020799, Pathology Cell Core P30CA125123, Biology of Inflammation Center Baylor College of Medicine, an unrestricted grant from Research to Prevent Blindness, New York, NY (SCP), the Oshman Foundation, Houston, TX (SCP), the William Stamps Farish Fund, Houston, TX (SCP), Hamill Foundation, Houston, TX (SCP), Sid W. Richardson Foundation, Ft Worth, TX (SCP).
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Disclosure statement: None of the authors have any financial or personal relationships to disclose that would cause a conflict of interest regarding this article.
References
- 1.Farrand KF, Fridman M, Stillman IO, Schaumberg DA. Prevalence of Diagnosed Dry Eye Disease in the United States Among Adults Aged 18 Years and Older. American journal of ophthalmology. 2017;182:90–98. [DOI] [PubMed] [Google Scholar]
- 2.Pflugfelder SC, Gumus K, Feuerman J, Alex A. Tear Volume-based Diagnostic Classification for Tear Dysfunction. International ophthalmology clinics. 2017;57(2):1–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Dartt DA. Regulation of mucin and fluid secretion by conjunctival epithelial cells. Prog Retin Eye Res 2002;21(6):555–576. [DOI] [PubMed] [Google Scholar]
- 4.de Paiva CS, Raince JK, McClellan AJ, et al. Homeostatic control of conjunctival mucosal goblet cells by NKT-derived IL-13. Mucosal Immunol 2011;4(4):397–408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Tukler Henriksson J, Coursey TG, Corry DB, De Paiva CS, Pflugfelder SC. IL-13 Stimulates Proliferation and Expression of Mucin and Immunomodulatory Genes in Cultured Conjunctival Goblet Cells. Investigative ophthalmology & visual science. 2015;56(8):4186–4197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Gipson IK, Inatomi T. Cellular origin of mucins of the ocular surface tear film. Advances in experimental medicine and biology. 1998;438:221–227. [DOI] [PubMed] [Google Scholar]
- 7.McKenzie RW, Jumblatt JE, Jumblatt MM. Quantification of MUC2 and MUC5AC transcripts in human conjunctiva. Investigative ophthalmology & visual science. 2000;41(3):703–708. [PubMed] [Google Scholar]
- 8.Jumblatt MM, McKenzie RW, Steele PS, Emberts CG, Jumblatt JE. MUC7 expression in the human lacrimal gland and conjunctiva. Cornea 2003;22(1):41–45. [DOI] [PubMed] [Google Scholar]
- 9.Marko CK, Tisdale AS, Spurr-Michaud S, Evans C, Gipson IK. The ocular surface phenotype of Muc5ac and Muc5b null mice. Investigative ophthalmology & visual science. 2014;55(1):291–300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Argueso P, Balaram M, Spurr-Michaud S, Keutmann HT, Dana MR, Gipson IK. Decreased levels of the goblet cell mucin MUC5AC in tears of patients with Sjogren syndrome. Investigative ophthalmology & visual science. 2002;43(4):1004–1011. [PubMed] [Google Scholar]
- 11.Spurr-Michaud S, Argueso P, Gipson I. Assay of mucins in human tear fluid. Experimental eye research. 2007;84(5):939–950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Gipson IK. Goblet cells of the conjunctiva: A review of recent findings. Prog Retin Eye Res 2016;54:49–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Baudouin C, Rolando M, Benitez Del Castillo JM, et al. Reconsidering the central role of mucins in dry eye and ocular surface diseases. Prog Retin Eye Res 2018. [DOI] [PubMed]
- 14.Barbosa FL, Xiao Y, Bian F, et al. Goblet Cells Contribute to Ocular Surface Immune Tolerance-Implications for Dry Eye Disease. International journal of molecular sciences. 2017;18(5). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Contreras-Ruiz L, Masli S. Immunomodulatory Cross-Talk between Conjunctival Goblet Cells and Dendritic Cells. PloS one. 2015;10(3):e0120284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Xiao Y, De Paiva CS, Yu Z, Guimaraes de Souza R, Li DQ, Pflugfelder SC. Goblet cell produced retinoic acid suppresses CD86 expression and IL-12 production in bone marrow derived cells. Int Immunol 2018. [DOI] [PMC free article] [PubMed]
- 17.Ubels JL, MacRae SM. Vitamin A is present as retinol in the tears of humans and rabbits. Current eye research. 1984;3(6):815–822. [DOI] [PubMed] [Google Scholar]
- 18.Ubels JL, Foley KM, Rismondo V. Retinol secretion by the lacrimal gland. Investigative ophthalmology & visual science. 1986;27(8):1261–1268. [PubMed] [Google Scholar]
- 19.Bian F, Xiao Y, Barbosa FL, et al. Age-associated antigen-presenting cell alterations promote dry-eye inducing Th1 cells. Mucosal immunology. 2019;12(4):897–908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Pflugfelder SC, De Paiva CS, Moore QL, et al. Aqueous Tear Deficiency Increases Conjunctival Interferon-gamma (IFN-gamma) Expression and Goblet Cell Loss. Investigative ophthalmology & visual science. 2015;56(12):7545–7550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Harris EW, Loewenstein JI, Azar D. Vitamin A deficiency and its effects on the eye. International ophthalmology clinics. 1998;38(1):155–161. [DOI] [PubMed] [Google Scholar]
- 22.Samarawickrama C, Chew S, Watson S. Retinoic acid and the ocular surface. Surv Ophthalmol 2015;60(3):183–195. [DOI] [PubMed] [Google Scholar]
- 23.Ralph RA. Conjunctival goblet cell density in normal subjects and in dry eye syndromes. Investigative ophthalmology. 1975;14(4):299–302. [PubMed] [Google Scholar]
- 24.Nelson JD, Wright JC. Conjunctival goblet cell densities in ocular surface disease. Arch Ophthalmol 1984;102(7):1049–1051. [DOI] [PubMed] [Google Scholar]
- 25.Pflugfelder SC, Tseng SC, Yoshino K, Monroy D, Felix C, Reis BL. Correlation of goblet cell density and mucosal epithelial membrane mucin expression with rose bengal staining in patients with ocular irritation. Ophthalmology. 1997;104(2):223–235. [DOI] [PubMed] [Google Scholar]
- 26.Wang Y, Ogawa Y, Dogru M, et al. Baseline profiles of ocular surface and tear dynamics after allogeneic hematopoietic stem cell transplantation in patients with or without chronic GVHD-related dry eye. Bone marrow transplantation. 2010;45(6):1077–1083. [DOI] [PubMed] [Google Scholar]
- 27.Zuazo F, Lopez-Ponce D, Salinas-Toro D, et al. [Conjunctival impression cytology in patients with normal and impaired OSDI scores]. Arch Soc Esp Oftalmol 2014;89(10):391–396. [DOI] [PubMed] [Google Scholar]
- 28.Pflugfelder SC, Bian F, Gumus K, Farley W, Stern ME, De Paiva CS. Severity of Sjogren’s Syndrome Keratoconjunctivitis Sicca Increases with Increased Percentage of Conjunctival Antigen-Presenting Cells. International journal of molecular sciences. 2018;19(9). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Pflugfelder SC DPC, Moore QL, Volpe EA, Li DQ, Gumus K, Zaheer ML, Corrales RM. Aqueous tear deficiency increases conjunctival interferon-gamma (IFN-γ) expression and goblet cell loss. Investigative ophthalmology & visual science. 2015;56(12):7545–7550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Bagga B, Motukupally SR, Mohamed A. Microbial keratitis in Stevens-Johnson syndrome: Clinical and microbiological profile. The ocular surface. 2018;16(4):454–457. [DOI] [PubMed] [Google Scholar]
- 31.Ormerod LD, Fong LP, Foster CS. Corneal infection in mucosal scarring disorders and Sjogren’s syndrome. American journal of ophthalmology. 1988;105(5):512–518. [DOI] [PubMed] [Google Scholar]
- 32.Pflugfelder SC, Wilhelmus KR, Osato MS, Matoba AY, Font RL. The autoimmune nature of aqueous tear deficiency. Ophthalmology. 1986;93(12):1513–1517. [DOI] [PubMed] [Google Scholar]
- 33.De Paiva CS, Villarreal AL, Corrales RM, et al. Dry eye-induced conjunctival epithelial squamous metaplasia is modulated by interferon-gamma. Investigative ophthalmology & visual science. 2007;48(6):2553–2560. [DOI] [PubMed] [Google Scholar]
- 34.Garcia-Posadas L, Hodges RR, Li D, et al. Interaction of IFN-gamma with cholinergic agonists to modulate rat and human goblet cell function. Mucosal immunology. 2016;9(1):206–217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Coursey TG, Tukler Henriksson J, Barbosa FL, de Paiva CS, Pflugfelder SC. Interferon-gamma-Induced Unfolded Protein Response in Conjunctival Goblet Cells as a Cause of Mucin Deficiency in Sjogren Syndrome. The American journal of pathology. 2016;186(6):1547–1558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Yoon KC, Park CS, You IC, et al. Expression of CXCL9, −10, −11, and CXCR3 in the tear film and ocular surface of patients with dry eye syndrome. Invest OphthalmolVisSci 2010;51(2):643–650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Coursey TG, Gandhi NB, Volpe EA, Pflugfelder SC, de Paiva CS. Chemokine receptors CCR6 and CXCR3 are necessary for CD4(+) T cell mediated ocular surface disease in experimental dry eye disease. PloS one. 2013;8(11):e78508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Pflugfelder SC, de Paiva CS. The Pathophysiology of Dry Eye Disease: What We Know and Future Directions for Research. Ophthalmology. 2017;124(11s):S4–s13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Foster CS, Rice BA, Dutt JE. Immunopathology of atopic keratoconjunctivitis. Ophthalmology. 1991;98(8):1190–1196. [DOI] [PubMed] [Google Scholar]
- 40.Roat MI, Ohji M, Hunt LE, Thoft RA. Conjunctival epithelial cell hypermitosis and goblet cell hyperplasia in atopic keratoconjunctivitis. American journal of ophthalmology. 1993;116(4):456–463. [DOI] [PubMed] [Google Scholar]
- 41.Kunert KS, Tisdale AS, Gipson IK. Goblet cell numbers and epithelial proliferation in the conjunctiva of patients with dry eye syndrome treated with cyclosporine. Arch Ophthalmol 2002;120(3):330–337. [DOI] [PubMed] [Google Scholar]
- 42.de Paiva CS, Pflugfelder SC, Ng SM, Akpek EK. Topical cyclosporine A therapy for dry eye syndrome. The Cochrane database of systematic reviews. 2019;9:Cd010051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Avunduk AM, Avunduk MC, Varnell ED, Kaufman HE. The comparison of efficacies of topical corticosteroids and nonsteroidal anti-inflammatory drops on dry eye patients: a clinical and immunocytochemical study. American journal of ophthalmology. 2003;136(4):593–602. [DOI] [PubMed] [Google Scholar]
- 44.Kim EC, Choi JS, Joo CK. A comparison of vitamin a and cyclosporine a 0.05% eye drops for treatment of dry eye syndrome. American journal of ophthalmology. 2009;147(2):206–213 e203. [DOI] [PubMed] [Google Scholar]
- 45.Guimaraes de Souza R, Yu Z, Stern ME, Pflugfelder SC, de Paiva CS. Suppression of Th1-Mediated Keratoconjunctivitis Sicca by Lifitegrast. Journal of ocular pharmacology and therapeutics : the official journal of the Association for Ocular Pharmacology and Therapeutics. 2018;34(7):543–549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Schein OD, Munoz B, Tielsch JM, Bandeen-Roche K, West S. Prevalence of dry eye among the elderly. Am J Ophthalmol 1997;124(6):723–728. [DOI] [PubMed] [Google Scholar]
- 47.Bandeen-Roche K, Munoz B, Tielsch JM, West SK, Schein OD. Self-reported assessment of dry eye in a population-based setting. Invest OphthalmolVisSci 1997;38(12):2469–2475. [PubMed] [Google Scholar]
- 48.McCarty CA, Bansal AK, Livingston PM, Stanislavsky YL, Taylor HR. The epidemiology of dry eye in Melbourne, Australia. Ophthalmology. 1998;105(6):1114–1119. [DOI] [PubMed] [Google Scholar]
- 49.Moss SE, Klein R, Klein BE. Prevalence of and risk factors for dry eye syndrome. ArchOphthalmol 2000;118(9):1264–1268. [DOI] [PubMed] [Google Scholar]
- 50.Schaumberg DA, Sullivan DA, Buring JE, Dana MR. Prevalence of dry eye syndrome among US women. AmJOphthalmol 2003;136(2):318–326. [DOI] [PubMed] [Google Scholar]
- 51.Lin PY, Tsai SY, Cheng CY, Liu JH, Chou P, Hsu WM. Prevalence of dry eye among an elderly Chinese population in Taiwan: the Shihpai Eye Study. Ophthalmology. 2003;110(6):1096–1101. [DOI] [PubMed] [Google Scholar]
- 52.Chia EM, Mitchell P, Rochtchina E, Lee AJ, Maroun R, Wang JJ. Prevalence and associations of dry eye syndrome in an older population: the Blue Mountains Eye Study. Clin Exp Ophthalmol 2003;31(3):229–232. [DOI] [PubMed] [Google Scholar]
- 53.Smith JA, Vitale S, Reed GF, et al. Dry eye signs and symptoms in women with premature ovarian failure. ArchOphthalmol 2004;122(2):151–156. [DOI] [PubMed] [Google Scholar]
- 54.Moss SE, Klein R, Klein BE. Incidence of dry eye in an older population. Arch Ophthalmol 2004;122(3):369–373. [DOI] [PubMed] [Google Scholar]
- 55.Schaumberg DA, Dana R, Buring JE, Sullivan DA. Prevalence of dry eye disease among US men: estimates from the Physicians’ Health Studies. Arch Ophthalmol 2009;127(6):763–768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Cui L, Shen M, Wang J, et al. Age-related changes in tear menisci imaged by optical coherence tomography. OptomVisSci 2011;88(10):1214–1219. [DOI] [PubMed] [Google Scholar]
- 57.Shimmura S, Shimazaki J, Tsubota K. Results of a population-based questionnaire on the symptoms and lifestyles associated with dry eye. Cornea 1999;18(4):408–411. [DOI] [PubMed] [Google Scholar]
- 58.Stepp MA, Pal-Ghosh S, Tadvalkar G, Williams A, Pflugfelder SC, de Paiva CS. Reduced intraepithelial corneal nerve density and sensitivity accompany desiccating stress and aging in C57BL/6 mice. Experimental eye research. 2018;169:91–98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Giebel J, Woenckhaus C, Fabian M, Tost F. Age-related differential expression of apoptosis-related genes in conjunctival epithelial cells. Acta ophthalmologica Scandinavica 2005;83(4):471–476. [DOI] [PubMed] [Google Scholar]
- 60.Zhu W, Hong J, Zheng T, Le Q, Xu J, Sun X. Age-related changes of human conjunctiva on in vivo confocal microscopy. The British journal of ophthalmology. 2010;94(11):1448–1453. [DOI] [PubMed] [Google Scholar]
- 61.Di Zazzo A, Micera A, Coassin M, et al. InflammAging at Ocular Surface: Clinical and Biomolecular Analyses in Healthy Volunteers. Invest Ophthalmol Vis Sci 2019;60(5):1769–1775. [DOI] [PubMed] [Google Scholar]
- 62.Micera A, Di Zazzo A, Esposito G, et al. Age-Related Changes to Human Tear Composition. Investigative Ophthalmology & Visual Science. 2018;59(5):2024–2031. [DOI] [PubMed] [Google Scholar]
- 63.De Silva MEH, Hill LJ, Downie LE, Chinnery HR. The Effects of Aging on Corneal and Ocular Surface Homeostasis in Mice. Invest Ophthalmol Vis Sci 2019;60(7):2705–2715. [DOI] [PubMed] [Google Scholar]
- 64.Cayetanot F, Nygard M, Perret M, Kristensson K, Aujard F. Plasma levels of interferon-gamma correlate with age-related disturbances of circadian rhythms and survival in a non-human primate. Chronobiology international. 2009;26(8):1587–1601. [DOI] [PubMed] [Google Scholar]
- 65.Kim KS, Kang KW, Seu YB, Baek SH, Kim JR. Interferon-gamma induces cellular senescence through p53-dependent DNA damage signaling in human endothelial cells. Mechanisms of ageing and development. 2009;130(3):179–188. [DOI] [PubMed] [Google Scholar]
- 66.Liang EC, Rossetti M, Sidwell T, et al. Differences in Proinflammatory Cytokines and Monocyte Subtypes in Older as Compared With Younger Kidney Transplant Recipients. Transplant Direct. 2018;4(3):e348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.McClellan AJ, Volpe EA, Zhang X, et al. Ocular Surface Disease and Dacryoadenitis in Aging C57BL/6 Mice. Am J Pathol 2014;184(3):631–643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Ogawa T, Boylan SA, Oltjen SL, Hjelmeland LM. Changes in the spatial expression of genes with aging in the mouse RPE/choroid. Molecular vision. 2005;11:380–386. [PubMed] [Google Scholar]
- 69.Wendt W, Lubbert H, Stichel CC. Upregulation of cathepsin S in the aging and pathological nervous system of mice. Brain research. 2008;1232:7–20. [DOI] [PubMed] [Google Scholar]
- 70.Singh P, Goode T, Dean A, Awad SS, Darlington GJ. Elevated interferon gamma signaling contributes to impaired regeneration in the aged liver. The journals of gerontology Series A, Biological sciences and medical sciences. 2011;66(9):944–956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Takayama E, Seki S, Ohkawa T, et al. Mouse CD8+ CD122+ T cells with intermediate TCR increasing with age provide a source of early IFN-gamma production. Journal of immunology. 2000;164(11):5652–5658. [DOI] [PubMed] [Google Scholar]
- 72.Franceschi C, BonaFe M, Valensin S, et al. Inflamm-aging. An evolutionary perspective on immunosenescence. AnnNYAcadSci 2000;908:244–254. [DOI] [PubMed] [Google Scholar]
- 73.Zhang X, Chen W, De Paiva CS, et al. Interferon-gamma exacerbates dry eye-induced apoptosis in conjunctiva through dual apoptotic pathways. Investigative ophthalmology & visual science. 2011;52(9):6279–6285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Zhang X, Chen W, De Paiva CS, et al. Desiccating stress induces CD4+ T-cell-mediated Sjogren’s syndrome-like corneal epithelial apoptosis via activation of the extrinsic apoptotic pathway by interferon-gamma. The American journal of pathology. 2011;179(4):1807–1814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Zhang X, De Paiva CS, Su Z, Volpe EA, Li DQ, Pflugfelder SC. Topical interferon-gamma neutralization prevents conjunctival goblet cell loss in experimental murine dry eye. Experimental eye research. 2014;118:117–124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Gottenberg JE, Chiocchia G. Dendritic cells and interferon-mediated autoimmunity. Biochimie 2007;89(6–7):856–871. [DOI] [PubMed] [Google Scholar]
- 77.McClellan AJ, Volpe EA, Zhang X, et al. Ocular surface disease and dacryoadenitis in aging C57BL/6 mice. The American journal of pathology. 2014;184(3):631–643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Oxenkrug GF. Interferon-gamma-inducible kynurenines/pteridines inflammation cascade: implications for aging and aging-associated psychiatric and medical disorders. J Neural Transm (Vienna). 2011;118(1):75–85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Williams GP, Denniston AK, Oswal KS, et al. The dominant human conjunctival epithelial CD8alphabeta+ T cell population is maintained with age but the number of CD4+ T cells increases. Age (Dordr). 2012;34:1517–1528. Epub 2011 Sep 1527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Volpe EA, Henriksson JT, Wang C, et al. Interferon-gamma deficiency protects against aging-related goblet cell loss. Oncotarget 2016;7(40):64605–66461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Park KS, Korfhagen TR, Bruno MD, et al. SPDEF regulates goblet cell hyperplasia in the airway epithelium. J Clin Invest 2007;117(4):978–988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Noah TK, Kazanjian A, Whitsett J, Shroyer NF. SAM pointed domain ETS factor (SPDEF) regulates terminal differentiation and maturation of intestinal goblet cells. Exp Cell Res 2010;316(3):452–465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Marko CK, Menon BB, Chen G, Whitsett JA, Clevers H, Gipson IK. Spdef null mice lack conjunctival goblet cells and provide a model of dry eye. The American journal of pathology. 2013;183(1):35–48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Chen G, Korfhagen TR, Xu Y, et al. SPDEF is required for mouse pulmonary goblet cell differentiation and regulates a network of genes associated with mucus production. J Clin Invest 2009;119(10):2914–2924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.De Paiva CS, Raince JK, McClellan AJ, et al. Homeostatic control of conjunctival mucosal goblet cells by NKT-derived IL-13. Mucosal Immunol 2011;4(4):397–408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Whitsett JA. Airway Epithelial Differentiation and Mucociliary Clearance. Ann Am Thorac Soc 2018;15(Supplement_3):S143–S148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Rajavelu P, Chen G, Xu Y, Kitzmiller JA, Korfhagen TR, Whitsett JA. Airway epithelial SPDEF integrates goblet cell differentiation and pulmonary Th2 inflammation. J Clin Invest 2015;125(5):2021–2031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Ko BY, Xiao Y, Barbosa FL, de Paiva CS, Pflugfelder SC. Goblet cell loss abrogates ocular surface immune tolerance. JCI insight. 2018;3(3). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Shan M, Gentile M, Yeiser JR, et al. Mucus enhances gut homeostasis and oral tolerance by delivering immunoregulatory signals. Science. 2013;342(6157):447–453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Egan RM, Yorkey C, Black R, et al. In vivo behavior of peptide-specific T cells during mucosal tolerance induction: antigen introduced through the mucosa of the conjunctiva elicits prolonged antigen-specific T cell priming followed by anergy. Journal of immunology (Baltimore, Md : 1950). 2000;164(9):4543–4550. [DOI] [PubMed] [Google Scholar]
- 91.Guzman M, Keitelman I, Sabbione F, Trevani AS, Giordano MN, Galletti JG. Desiccating stress-induced disruption of ocular surface immune tolerance drives dry eye disease. Clinical and experimental immunology. 2016;184(2):248–256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Corrales RM, Narayanan S, Fernandez I, et al. Ocular mucin gene expression levels as biomarkers for the diagnosis of dry eye syndrome. Investigative ophthalmology & visual science. 2011;52(11):8363–8369. [DOI] [PubMed] [Google Scholar]
- 93.Koppaka V, Thompson DC, Chen Y, et al. Aldehyde dehydrogenase inhibitors: a comprehensive review of the pharmacology, mechanism of action, substrate specificity, and clinical application. Pharmacol Rev 2012;64(3):520–539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Na SY, Kang BY, Chung SW, et al. Retinoids inhibit interleukin-12 production in macrophages through physical associations of retinoid X receptor and NFkappaB. The Journal of biological chemistry. 1999;274(12):7674–7680. [DOI] [PubMed] [Google Scholar]
- 95.Du X, Tabeta K, Mann N, Crozat K, Mudd S, Beutler B. An essential role for Rxr alpha in the development of Th2 responses. Eur J Immunol 2005;35(12):3414–3423. [DOI] [PubMed] [Google Scholar]
- 96.Pflugfelder SC DSR, Alam J, Yu Z, De Paiva CS. Increased conjunctival monocyte/macrophage antigen presenting cells in Pinkie RXRa deficient mice with accelerated dry eye. Investigative ophthalmology & visual science. 2019;60(9). [Google Scholar]
- 97.Reins RY, Baidouri H, McDermott AM. Vitamin D Activation and Function in Human Corneal Epithelial Cells During TLR-Induced Inflammation. Investigative ophthalmology & visual science. 2015;56(13):7715–7727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Chen Y, Zhang X, Yang L, et al. Decreased PPAR-gamma expression in the conjunctiva and increased expression of TNF-alpha and IL-1beta in the conjunctiva and tear fluid of dry eye mice. Molecular medicine reports. 2014;9(5):2015–2023. [DOI] [PubMed] [Google Scholar]
- 99.Nien CJ, Massei S, Lin G, et al. Effects of age and dysfunction on human meibomian glands. Arch Ophthalmol 2011;129(4):462–469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Mukwaya A, Lennikov A, Xeroudaki M, et al. Time-dependent LXR/RXR pathway modulation characterizes capillary remodeling in inflammatory corneal neovascularization. Angiogenesis. 2018;21(2):395–413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Roszer T, Menendez-Gutierrez MP, Cedenilla M, Ricote M. Retinoid X receptors in macrophage biology. Trends in endocrinology and metabolism: TEM. 2013;24(9):460–468. [DOI] [PubMed] [Google Scholar]
- 102.Sheppard JD Jr., Singh R, McClellan AJ, et al. Long-term Supplementation With n-6 and n-3 PUFAs Improves Moderate-to-Severe Keratoconjunctivitis Sicca: A Randomized Double-Blind Clinical Trial. Cornea 2013. [DOI] [PubMed]
- 103.Jester JV, Potma E, Brown DJ. PPARgamma Regulates Mouse Meibocyte Differentiation and Lipid Synthesis. The ocular surface. 2016;14(4):484–494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Srividya G, Angayarkanni N, Iyer G, Srinivasan B, Agarwal S. Altered retinoid metabolism gene expression in chronic Stevens-Johnson syndrome. The British journal of ophthalmology. 2019;103(8):1015–1023. [DOI] [PubMed] [Google Scholar]
- 105.Chen X, Aqrawi LA, Utheim TP, et al. Elevated cytokine levels in tears and saliva of patients with primary Sjogren’s syndrome correlate with clinical ocular and oral manifestations. Scientific reports. 2019;9(1):7319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Enriquez-de-Salamanca A, Castellanos E, Stern ME, et al. Tear cytokine and chemokine analysis and clinical correlations in evaporative-type dry eye disease. Molecular vision. 2010;16:862–873. [PMC free article] [PubMed] [Google Scholar]
- 107.Jackson DC, Zeng W, Wong CY, et al. Tear Interferon-Gamma as a Biomarker for Evaporative Dry Eye Disease. Investigative ophthalmology & visual science. 2016;57(11):4824–4830. [DOI] [PubMed] [Google Scholar]
- 108.Shetty R, Sethu S, Chevour P, et al. Lower Vitamin D Level and Distinct Tear Cytokine Profile Were Observed in Patients with Mild Dry Eye Signs but Exaggerated Symptoms. Translational vision science & technology. 2016;5(6):16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Agrawal R, Balne PK, Veerappan A, et al. A distinct cytokines profile in tear film of dry eye disease (DED) patients with HIV infection. Cytokine 2016;88:77–84. [DOI] [PubMed] [Google Scholar]
- 110.Riemens A, Stoyanova E, Rothova A, Kuiper J. Cytokines in tear fluid of patients with ocular graft-versus-host disease after allogeneic stem cell transplantation. Molecular vision. 2012;18:797–802. [PMC free article] [PubMed] [Google Scholar]
- 111.Meadows JF, Dionne K, Nichols KK. Differential Profiling of T-Cell Cytokines as Measured by Protein Microarray Across Dry Eye Subgroups. Cornea 2016;35(3):329–335. [DOI] [PubMed] [Google Scholar]
- 112.Zywalewska-Gorna N, Mrugacz M, Bakunowicz-Lazarczyk A. [The evaluation of chosen cytokines in induction of ocular changes in Sjogren’s syndrome of dry eye]. Klin Oczna 2007;109(10–12):435–437. [PubMed] [Google Scholar]
- 113.Zhao H, Li Q, Ye M, Yu J. Tear Luminex Analysis in Dry Eye Patients. Medical science monitor : international medical journal of experimental and clinical research. 2018;24:7595–7602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Yoon KC, Park CS, You IC, et al. Expression of CXCL9, −10, −11, and CXCR3 in the tear film and ocular surface of patients with dry eye syndrome. Investigative ophthalmology & visual science. 2010;51(2):643–650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Strong B, Farley W, Stern ME, Pflugfelder SC. Topical cyclosporine inhibits conjunctival epithelial apoptosis in experimental murine keratoconjunctivitis sicca. Cornea 2005;24(1):80–85. [DOI] [PubMed] [Google Scholar]
- 116.Niederkorn JY, Stern ME, Pflugfelder SC, et al. Desiccating Stress Induces T Cell-Mediated Sjogren’s Syndrome-Like Lacrimal Keratoconjunctivitis. J Immunol 2006;176(7):3950–3957. [DOI] [PubMed] [Google Scholar]
- 117.de Paiva CS, Villarreal AL, Corrales RM, et al. Dry Eye-Induced Conjunctival Epithelial Squamous Metaplasia Is Modulated by Interferon-{gamma}. Invest Ophthalmol Vis Sci 2007;48(6):2553–2560. [DOI] [PubMed] [Google Scholar]
- 118.Yoon KC, de Paiva CS, Qi H, et al. Expression of th-1 chemokines and chemokine receptors on the ocular surface of C57BL/6 mice: effects of desiccating stress. Invest OphthalmolVisSci 2007;48(6):2561–2569. [DOI] [PubMed] [Google Scholar]
- 119.Yeh S, de Paiva CS, Hwang CS, et al. Spontaneous T cell mediated keratoconjunctivitis in Aire-deficient mice. The British journal of ophthalmology. 2009;93(9):1260–1264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.de Paiva CS, Hwang CS, Pitcher JD III, et al. Age-related T-cell cytokine profile parallels corneal disease severity in Sjogren’s syndrome-like keratoconjunctivitis sicca in CD25KO mice. Rheumatology. 2010;49(2):246–258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.de Paiva CS, Rocha EM. Sjogren syndrome: what and where are we looking for? Current opinion in ophthalmology. 2015;26(6):517–525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Schaumburg CS, Siemasko KF, de Paiva CS, et al. Ocular surface APCs are necessary for autoreactive T cell-mediated experimental autoimmune lacrimal keratoconjunctivitis. J Immunol 2011;187(7):3653–3662. [DOI] [PubMed] [Google Scholar]
- 123.de Paiva CS, Volpe EA, Gandhi NB, et al. Disruption of TGF-beta Signaling Improves Ocular Surface Epithelial Disease in Experimental Autoimmune Keratoconjunctivitis Sicca. PLoS One. 2011;6(12):e29017 Epub 22011 Dec 29014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Zhang X, Chen W, de Paiva CS, et al. Interferon-gamma exacerbates dry eye-induced apoptosis in conjunctiva through dual apoptotic pathways. Invest Ophthalmol Vis Sci 2011;52(9):6279–6285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.de Paiva CS, Schwartz CE, Gjorstrup P, Pflugfelder SC. Resolvin E1 (RX-10001) reduces corneal epithelial barrier disruption and protects against goblet cell loss in a murine model of dry eye. Cornea 2012;31(11):1299–1303. [DOI] [PubMed] [Google Scholar]
- 126.Li Z, Choi W, Oh HJ, Yoon KC. Effectiveness of topical infliximab in a mouse model of experimental dry eye. Cornea 2012;31 Suppl 1:S25–31. [DOI] [PubMed] [Google Scholar]
- 127.Zhang X, de Paiva CS, Su Z, Volpe EA, Li DQ, Pflugfelder SC. Topical interferon-gamma neutralization prevents conjunctival goblet cell loss in experimental murine dry eye. Exp Eye Res 2014;118:117–124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Coursey TG, Gandhi NB, Volpe EA, Pflugfelder SC, de Paiva CS. Chemokine receptors CCR6 and CXCR3 are necessary for CD4(+) T cell mediated ocular surface disease in experimental dry eye disease. PLoSOne 2013;8(11):e78508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Bian FX, Y; Barbosa FL; de Souza RG; Hernandez H; Yu Z; Pflugfelder SC; de Paiva CS. Age-associated Antigen-Presenting Cell Alterations Promote Dry-Eye Inducing Th1 cells. Mucosal immunology. 2018;in press. [DOI] [PMC free article] [PubMed]
- 130.Krauss AH, Corrales RM, Pelegrino FS, Tukler-Henriksson J, Pflugfelder SC, de Paiva CS. Improvement of Outcome Measures of Dry Eye by a Novel Integrin Antagonist in the Murine Desiccating Stress Model. Investigative ophthalmology & visual science. 2015;56(10):5888–5895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.You IC, Bian F, Volpe EA, de Paiva CS, Pflugfelder SC. Age-related conjunctival disease in the C57BL/6.NOD-Aec1Aec2 Mouse Model of Sjogren Syndrome develops independent of lacrimal dysfunction. Invest Ophthalmol Vis Sci 2015;2015 Mar 10. pii: IOVS-14–15668. [DOI] [PMC free article] [PubMed]
- 132.Coursey TG, Henriksson JT, Barbosa FL, de Paiva CS, Pflugfelder SC. Interferon-gamma-Induced Unfolded Protein Response in Conjunctival Goblet Cells as a Cause of Mucin Deficiency in Sjogren Syndrome. The American journal of pathology. 2016;S0002–9440((16)):30005–30000. [DOI] [PMC free article] [PubMed]
- 133.Choi W, Lee JB, Cui L, et al. Therapeutic Efficacy of Topically Applied Antioxidant Medicinal Plant Extracts in a Mouse Model of Experimental Dry Eye. Oxidative medicine and cellular longevity. 2016;2016:4727415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Coursey TG, Bian F, Zaheer M, Pflugfelder SC, Volpe EA, de Paiva CS. Age-related spontaneous lacrimal keratoconjunctivitis is accompanied by dysfunctional T regulatory cells. Mucosal immunology. 2017;10(3):743–456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Portal C, Gouyer V, Gottrand F, Desseyn JL. Preclinical mouse model to monitor live Muc5b-producing conjunctival goblet cell density under pharmacological treatments. PloS one. 2017;12(3):e0174764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Zhang X, Lin X, Liu Z, et al. Topical Application of Mizoribine Suppresses CD4+ T-cell-Mediated Pathogenesis in Murine Dry Eye. Invest Ophthalmol Vis Sci 2017;58(14):6056–6064. [DOI] [PubMed] [Google Scholar]
- 137.Li Z, Woo JM, Chung SW, et al. Therapeutic effect of topical adiponectin in a mouse model of desiccating stress-induced dry eye. Investigative ophthalmology & visual science. 2013;54(1):155–162. [DOI] [PubMed] [Google Scholar]
- 138.Wang C, Zaheer M, Bian F, et al. Sjogren-Like Lacrimal Keratoconjunctivitis in Germ-Free Mice. International journal of molecular sciences. 2018;19(2):pii: E565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Zaheer M, Wang C, Bian F, et al. Protective role of commensal bacteria in Sjogren Syndrome. Journal of autoimmunity. 2018;pii: S0896–8411(18):30179–30173. [DOI] [PMC free article] [PubMed]
- 140.You IC, Li Y, Jin R, Ahn M, Choi W, Yoon KC. Comparison of 0.1%, 0.18%, and 0.3% Hyaluronic Acid Eye Drops in the Treatment of Experimental Dry Eye. Journal of ocular pharmacology and therapeutics : the official journal of the Association for Ocular Pharmacology and Therapeutics. 2018;34(8):557–564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Li Y, Jin R, Li L, et al. Expression and Role of Nucleotide-Binding Oligomerization Domain 2 (NOD2) in the Ocular Surface of Murine Dry Eye. Investigative ophthalmology & visual science. 2019;60(7):2641–2649. [DOI] [PubMed] [Google Scholar]