Abstract
Background
Optimal dosing of the recombinant tissue-type plasminogen activator (rt-PA) is important in treating pulmonary thromboembolism (PTE). The aim of this study was to compare the efficacy and safety of a 50 mg/2 h rt-PA regimen with a 100 mg/2 h rt-PA regimen in patients with acute PTE.
Methods
A prospective, randomized, multicenter trial was conducted in which 118 patients with acute PTE and either hemodynamic instability or massive pulmonary artery obstruction were randomly assigned to receive a treatment regiment of either rt-PA at 50 mg/2 h (n = 65) or 100 mg/2 h (n = 53). The efficacy was determined by observing the improvements of right ventricular dysfunctions (RVDs) on echocardiograms, lung perfusion defects on ventilation perfusion lung scans, and pulmonary artery obstructions on CT angiograms. The adverse events, including death, bleeding, and PTE recurrence, were also evaluated.
Results
Progressive improvements in RVDs, lung perfusion defects, and pulmonary artery obstructions were found to be similarly significant in both treatment groups. This is true for patients with either hemodynamic instability or massive pulmonary artery obstruction. Three (6%) patients in the rt-PA 100 mg/2 h group and one (2%) in the rt-PA 50 mg/2 h group died as the result of either PTE or bleeding. Importantly, the 50 mg/2 h rt-PA regimen resulted in less bleeding tendency than the 100 mg/2 h regimen (3% vs 10%), especially in patients with a body weight < 65 kg (14.8% vs 41.2%, P = .049). No fatal recurrent PTE was found in either group.
Conclusions
Compared with the 100 mg/2 h regimen, the 50 mg/2 h rt-PA regimen exhibits similar efficacy and perhaps better safety in patients with acute PTE. These findings support the notion that optimizing rt-PA dosing is worthwhile when treating patients with PTE.
Trial registration
clinicaltrials.gov; Identifier: NCT00781378
Abbreviations
- CTPA
computed tomographic pulmonary angiography
- ICH
intracranial hemorrhage
- INR
international normalized ratio
- PTE
pulmonary thromboembolism
- rt-PA
recombinant tissue-type plasminogen activator
- RVD
right ventricular dysfunction
- RVED/LVED
right and left ventricular end-diastolic diameter ratio in the parasternal long-axis view
- RVWM
right ventricular wall movements
- SPAP
systolic pulmonary arterial pressure
- V/Q
ventilation perfusion
Recombinant tissue-type plasminogen activator (rt-PA) is currently the most commonly used thrombolytic therapy for pulmonary thromboembolism (PTE). Similar to most thrombolytic agents, rt-PA carries a significant dose-dependent risk of bleeding. Optimal dosing that maximizes benefits and minimizes bleeding complications is important.
The effectiveness of rt-PA in patients with PTE has been evaluated with dosages ranging from 0.6 mg/kg over 15 min to 100 mg/2 h.1, 2 The regimen of rt-PA at 100mg/2 h has been approved by the US Food and Drug Administration and has been recommended in recent guidelines as the standard for PTE thrombolysis.3, 4 Although this dose is effective, increasing evidence suggests that it has a high rate of bleeding complications.5, 6, 7 Thus, there is uncertainty regarding whether the rt-PA 100 mg/2 h regimen is effective for all patients and if it is the optimal rt-PA dosage for PTE thrombolytic therapy.7, 8
Lower dosages of rt-PA, such as a 50 mg/2 h regimen, have been used effectively for treating acute myocardial infarction.9 With potentially fewer bleeding complications, rt-PA at 50 mg/2 h may be a better regimen for PTE treatment as well. However, no direct comparison of rt-PA at 50 mg/2 h and rt-PA at 100 mg/2 h regimens has been conducted in patients with PTE. It is unclear whether the rt-PA at 50 mg/2 h regimen is indeed a better dosage for PTE.
In this study, we compared the efficacy and safety of the rt-PA at 50 mg/2 h with the rt-PA at 100 mg/2 h regimen in patients with acute PTE. Patients with acute PTE and either hemodynamic instability10 or massive pulmonary artery obstruction and right ventricular dysfunction (RVD)3, 10 were enrolled. Efficacy was assessed by the improvements of right ventricular functions on echocardiograms, perfusion defects on ventilation perfusion (V/Q) lung scans, and pulmonary artery obstructions on CT angiograms. The adverse events, including death, bleeding, and PTE recurrence, were also evaluated.
Materials and Methods
Study Organization
A prospective, randomized, open label, multicenter trial was conducted between July 2002 and February 2006 in China. The trial was suspended for nearly 1 year because of the severe acute respiratory syndrome outbreak in 2003. A central steering committee was formed that was in charge of study design, protocol development and standardization, quality control, and data verification and analysis. The study protocols were reviewed and approved by the institutional boards and ethics committees of all participating centers. Patients enrolled in the study were centrally and randomly assigned to a treatment group with a standard computerized randomization program. The clinical information and images of each patient were reviewed independently by the members of the steering committee.
Patient Selection
Patients aged 18 to 75 years with acute PTE and indications for thrombolytic therapy were enrolled. All diagnoses were confirmed either by a high-probability V/Q scan or by the presence of an intraluminal filling defect on spiral computed tomographic pulmonary angiography (CTPA) examination. Only patients with symptoms that occurred within 15 days from the time of enrollment were selected. The inclusion criteria were either hemodynamically massive PTE in patients with hemodynamic instability or cardiogenic shock, or anatomically massive PTE in patients with massive pulmonary artery obstruction (obstructions in more than two lobes on CTPA examination or perfusion defects in more than seven segments on V/Q scan) with combined RVD and pulmonary hypertension on echocardiographic examination.11, 12 Studies have shown that such patients are at high risk of fatal conditions and may benefit from thrombolytic therapy.13, 14, 15 Patients were excluded if they had received parenteral heparin for more than 72 h or had thrombolytic contraindications.10, 16, 17 Written informed consents were obtained from all the patients.
Treatment Regimens
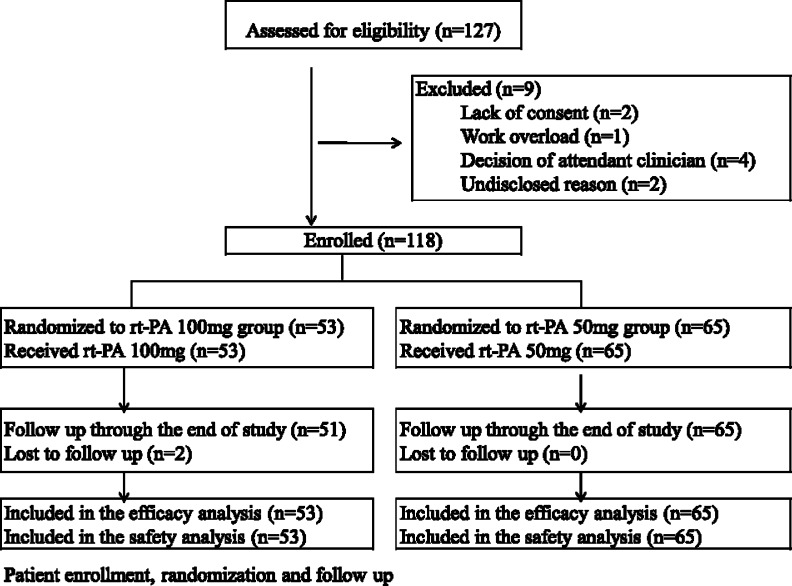
Eligible subjects were randomly assigned to receive either 50 mg rt-PA or 100 mg rt-PA in continuous intravenous infusion for 2 h (Fig 1 ). We used rt-PA (alteplase) from Boehringer Ingelheim Pharma (Shanghai, Republic of China). The activated partial thromboplastin time was determined after the rt-PA infusion. If the value was < 80 s, a subcutaneous injection of low-molecular-weight heparin (nadroparin, 86 antifactor Xa International Units/kg) was given every 12 h. Overlapping oral anticoagulant therapy (warfarin) was started on day 1 to 3 after nadroparin injection to maintain an international normalized ratio (INR) of 2.0 to 3.0. Nadroparin was stopped 4 to 5 days after warfarin was added, and the INR was stabilized at 2.0 to 3.0 for at least 2 days. Warfarin was continuously used for at least 3 to 6 months, and subsequent doses were adjusted to maintain the INR within the 2.0 to 3.0 range, targeting a value of 2.5.
Figure 1.
Flow study diagram. rt-PA = recombinant tissue-type plasminogen activator.
Efficacy
Symptoms and signs of PTE in all patients were monitored. Echocardiograms, V/Q lung scans, and CTPA were evaluated within 48 h before rt-PA infusion and repeated in most patients at 24 h and 14 days after rt-PA treatment. Efficacy was determined by observing the improvements of right ventricular functions on echocardiograms, lung perfusion on lung V/Q scans, and pulmonary artery obstruction on CTPA.
Echocardiograms: Echocardiographic examinations were performed and analyzed by two echocardiographers. Right ventricular function was analyzed by observing improvement of (1) the right and left ventricular end-diastolic diameter ratio in the parasternal long-axis view (RVED/LVED); (2) right ventricular wall movements (RVWM); and (3) estimated systolic pulmonary artery pressure (SPAP), in the absence of left ventricular disease or mitral valve disease as previously reported.14, 15
V/Q Lung Scans: Lung scans were independently reviewed and evaluated by two specialists using the methods described previously.18, 19 Each anatomic segment of the lung was reviewed, and any defect within each segment was scored according to the level of perfusion reduction.
CTPA: The location and severity of thrombus obstruction of the pulmonary vascular bed were reviewed and evaluated using the helical CTPA score system described in previous studies.20, 21 The index is defined as the product of N × D, where N is the value of the proximal clot site (equal to the number of segmental branches arising distally), and D is the degree of obstruction. Partial obstruction is scored as 1, and total obstruction is graded as 2.
Death, Bleeding, and PTE Recurrence
Adverse events were monitored for 14 days after initial treatment with rt-PA. Deaths were classified as being due to PTE, bleeding, or other causes (including myocardial infarction and unknown causes). Major bleeding included cases of fatal bleeding, intracranial hemorrhage (ICH), or a drop in the hemoglobin concentration by at least 20 g/L or a drop that required transfusion of more than 400 mL of RBCs (within 72 h of initiating rt-PA therapy). Minor bleeding included cases of bleeding with a hemoglobin concentration drop < 20 g/L. Recurrence of PTE was confirmed by V/Q scanning or spiral CTPA.
Statistical Analysis
Calculation of the sample size demonstrated that 110 patients were required to show a difference of 10 points between treatment groups in percentage reduction of the score on CTPA at 24 h from the onset of thrombolysis with 80% power and a two-sided level of significance of P < .05, assuming a standard deviation of 10 points. Data were analyzed according to the intention-to-treat principle.
Categorical data were compared using the χ2 test and Fisher exact test (2 × 2 table). Comparison of continuous data between the treatment groups was performed using the t test or Wilcoxon test as appropriate. Changes in the measurements of echocardiograms, V/Q lung scans, and CT pulmonary angiograms over time were analyzed using repeated measures of analysis of variance. Subgroup analysis was also conducted based on the hemodynamic status of the PTEs. All reported P values are two-sided, and P < .05 was considered statistically significant.
Results
Patients and Baseline Characteristics
A total of 127 patients were screened in the multiple centers. Nine (7%) patients were ineligible and were excluded according to the predefined exclusion criteria. Among the 118 patients enrolled in the study, 53 were assigned to receive 100 mg of rt-PA and 65 were assigned to receive 50 mg of rt-PA (see Fig 1). There were no significant differences between the two groups in regard to baseline characteristics (Table 1 ).
Table 1.
Baseline Clinical Characteristics
| Characteristics | rt-PA 100 mg (n = 53) | rt-PA 50 mg (n = 65) |
|---|---|---|
| Gender, No., male/female | 32/21 | 37/28 |
| Age, y | 51.9 ± 13.5 | 55.3 ± 14.1 |
| Weight, kg | 71.9 ± 12.6 | 69.3 ± 11.3 |
| BMI, kg/m2 | 25.38 ± 3.52 | 25.09 ± 3.26 |
| Systolic blood pressure, mm Hg | 127.0 ± 21.0 | 121.8 ± 17.0 |
| Diastolic blood pressure, mm Hg | 80.9 ± 12.3 | 74.2 ± 13.9 |
| Respiratory rate, breaths/min | 23.1 ± 4.9 | 23.7 ± 6.4 |
| Heart rate, bpm | 96.4 ± 17.6 | 93.9 ± 17.8 |
| Prior DVT or PE, No. (%) | 6 (11.3) | 11 (16.9) |
| CVD, No. (%) | 8 (15.1) | 9 (13.8) |
| Hypertension, No. (%) | 20 (37.7) | 24 (36.9) |
| Diabetes mellitus, No. (%) | 2 (3.8) | 3 (4.6) |
| COPD, No. (%) | 2 (3.8) | 1 (1.5) |
| Malignancy, No. (%) | 1 (1.9) | 1 (1.5) |
| Hb, g/L | 127.9 ± 22.6 | 129.7 ± 20.6 |
| Hemodynamically massive PTE,a No. (%) | 19 (35.8) | 18 (27.7) |
| Anatomically massive PTE with RVD and PH,b No. (%) | 34 (64.2) | 47 (72.3) |
Data presented as mean ± SD unless otherwise indicated, P > .05 for all comparisons. bpm = beats/min; CVD = cardiovascular disease; PE = pulmonary embolism; PH = pulmonary hypertension; PTE = pulmonary thromboembolism; rt-PA = recombinant tissue-type plasminogen activator; RVD = right ventricular dysfunction.
Arterial hypotension (a systolic arterial pressure < 90 mm Hg or a drop in systolic arterial pressure of at least 40 mm Hg for at least 15 min) or cardiogenic shock.
Pulmonary artery obstruction > 2 lobes on computed tomographic pulmonary angiography or > 7 segments on ventilation perfusion scan combined with RVD and PH, but without hypotension or cardiogenic shock.
Efficacy
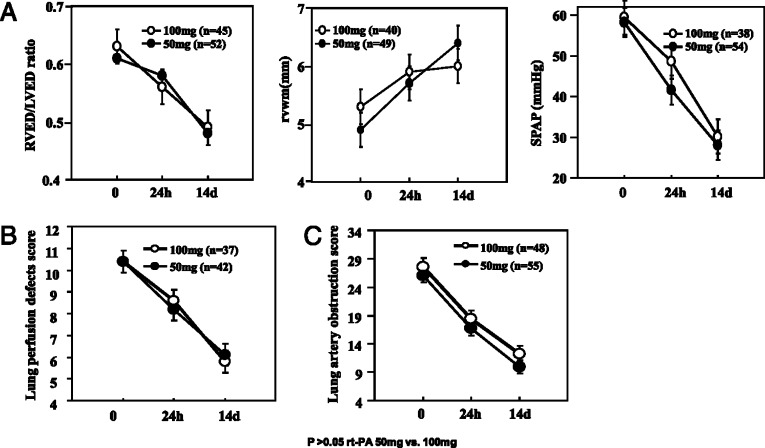
The improvements of pulmonary artery pressure and right ventricular function were evaluated by echocardiography. The baseline, 24-h, and 14-day follow-up evaluations of RVED/LVED, RVWM, and SPAP were performed in 97 (82%) patients, 45 in the 100 mg rt-PA group and 52 in the 50 mg rt-PA group. Both treatment groups showed significantly progressive improvements of right ventricular function as determined by all three measurements. No significant difference was found between the two treatment groups using these measurements at different time points (Fig 2A ).
Figure 2.
Comparison of the right side of the heart functions (A), lung perfusion defect scores (B), and pulmonary artery obstruction scores (C) between the two treatments for PTE. PTE = pulmonary thromboembolism; RVED/LVED = right and left ventricular end-diastolic diameter ratio in the parasternal long-axis view; RVWM = right ventricular wall movements; SPAP = systolic pulmonary artery pressure. See Figure 1 legend for expansion of other abbreviations.
Improvement of lung perfusion was determined by V/Q lung scan. The baseline, 24-h, and 14-day follow-up V/Q lung scans were conducted in 79 (67%) patients, 37 in the high-dose group and 42 in the low-dose group. Both treatments achieved significant improvements in pulmonary vascular perfusion defects, with similar defect scores at 24 h and 14 days after rt-PA administration (Fig 2B).
The improvement of pulmonary artery obstruction was assessed by CTPA. A total of 103 patients (87%) underwent baseline, 24-h, and 14-day follow-up CTPAs; 48 in the high-dose group and 55 in the low-dose group. Quantitative assessment showed that the two treatment groups had substantial reductions in the PTE obstructive index at different time points after treatment, with no significant difference between the groups (Fig 2C). The distributions of significant improvement, slight improvement, no change, and worsening status were similar between the two regimens (Table 2 ).
Table 2.
Comparison of Pulmonary Artery Obstruction Improvement Scales, Comparing Two Treatments for PTE
| 24 h |
14 d |
|||||
|---|---|---|---|---|---|---|
| Qualitative Changea | rt-PA 100 mg (n = 48) | rt-PA 50 mg (n = 55) | P | rt-PA 100 mg (n = 48) | rt-PA 50 mg (n = 55) | P |
| Significant improvement | 30 (62) | 33 (60) | .297 | 29 (60) | 33 (60) | .241 |
| Slight improvement | 10 (21) | 13 (24) | … | 14 (29) | 16 (29) | … |
| No change | 6 (13) | 3 (5) | … | 3 (6) | 5 (9) | … |
| Worse | 2 (4) | 6 (11) | … | 2 (4) | 1 (2) | … |
| Any improvement | 40 (83.3) | 46 (83.6) | .967 | 43 (89.6) | 49 (89.1) | .936 |
Data presented are number (%) of patients. Improvements of pulmonary artery obstruction from baseline between two treatments were compared. See Table 1 for expansion of abbreviations.
Any improvement (%) = (significant + slight)/total, which indicates the percentage of overall improvement. Significant improvement = obstruction decreased by ≥ 75%; slight improvement = obstruction decreased by ≥ 25% but < 75%; no change = obstruction reduced by < 25%; worse = obstruction increased in score index.
Death, Bleeding, and PTE Recurrence
The mortality was low in both groups, with three (6%) patient deaths in the high-dose group (two due to PTE combined with respiratory and congestive heart failure and one due to ICH) and one (2%) patient death in the low-dose group (due to PTE combined with respiratory and congestive heart failure). The total bleeding prevalence was relatively higher in the 100 mg rt-PA group compared with the 50 mg rt-PA group (32% vs 17%, P = .084). Major bleeding occurred more in the 100 mg rt-PA group than in the 50 mg rt-PA group (10% vs 3%, P = .288). One fatal bleeding episode with ICH occurred in the 100 mg rt-PA group (Table 3 ). There were three nonfatal recurrent PTEs during the first 14 days after rt-PA therapy: two (4%) patients in the 100 mg rt-PA group and one (2%) in the 50 mg rt-PA group (Table 3).
Table 3.
Comparison of Adverse Events During the First 14 d After Treatment, Comparing Two Treatments for PTE
| Adverse Events | rt-PA 100 mg (n = 48) | rt-PA 50 mg (n = 55) | P |
|---|---|---|---|
| Death | 3 (6) | 1 (2) | .472 |
| Due to PTE | 2 (4) | 1 (2) | … |
| Due to bleeding | 1 (2) | 0 (0) | … |
| Bleeding complications | 17 (32) | 11 (17) | .054 |
| Major bleeding | 5 (10) | 2 (3) | .288 |
| Fatal bleeding | 1 (2) | 0 (0) | … |
| Others | 4 (8) | 2 (3) | … |
| Minor bleeding | 12 (22) | 9 (14) | .214 |
| Recurrent PTE | 2 (4) | 1 (2) | .858 |
| Fatal | 0 (0) | 0 (0) | … |
| Nonfatal | 2 (4) | 1 (2) | … |
Data presented are number (%) of patients. Others = other major bleeding without death. See Table 1 for expansion of abbreviations.
Efficacy and Safety in Patients with Hemodynamic Instability or Massive Pulmonary Artery Obstruction
Further subgroup comparisons were conducted separately in patients with hemodynamic instability and in patients with massive pulmonary artery obstruction. In patients with hemodynamic instability, 50 mg rt-PA and 100 mg rt-PA produced similar progressive improvements in pulmonary artery obstructions. Additionally, no difference was found between these two treatments in terms of death, bleeding, and PTE recurrence in these patients (Table 4 ) Similarly, these two treatments showed similar efficacy and safety in patients with massive pulmonary vascular obstruction.
Table 4.
Comparison of Efficacy and Adverse Events Between Two Treatments in Subgroups of Patients With Hemodynamically and Anatomically Massive PTE
| Hemodynamically Massive PTE |
Anatomically Massive PTE |
|||||
|---|---|---|---|---|---|---|
| rt-PA 100 mg | rt-PA 50 mg | P | rt-P 100 mg | rt-PA 50 mg | P | |
| Efficacy | ||||||
| Pulmonary artery obstruction score by CTPA | (n = 16) | (n = 16) | (n = 32) | (n = 39) | … | |
| Baseline | 26.5 ± 13.0 | 24.6 ± 10.9 | .652 | 28.2 ± 12.5 | 26.5 ± 12.3 | .572 |
| 24-hour | 15.9 ± 10.6 | 16.9 ± 8.7 | .787 | 19.6 ± 8.9 | 16.6 ± 8.7 | .157 |
| 14-day | 13.1 ± 11.8 | 11.6 ± 10.4 | .706 | 11.8 ± 8.8 | 9.2 ± 7.2 | .179 |
| Adverse events | (n = 19) | (n = 18) | … | (n = 34) | (n = 47) | … |
| Death | 1 (5) | 1 (6) | 1.000 | 2 (6) | 0 (0) | .417 |
| Due to PTE | 1 (5) | 0 (0) | … | 1 (3) | 0 (0) | … |
| Due to bleeding | 0 (0) | 1 (6) | … | 1 (3) | 0 (0) | … |
| Recurrent PTE | 1 (5) | 1 (6) | 1.000 | 1 (0) | 0 (0) | .418 |
| Bleeding complications | 7 (37) | 5 (28) | .728 | 10 (29) | 6 (13) | .090 |
| Major bleeding | … | … | 1.000 | 3 (9) | 0 (0) | .070 |
| Fatal bleeding | 0 (0) | 0 (0) | … | 1 (3) | 0 (0) | … |
| Others | 2 (11) | 2 (11) | … | 2 (6) | 0 (0) | … |
| Minor bleeding | 5 (26) | 3 (17) | .693 | 7 (21) | 6 (13) | .373 |
Data presented represent mean ± SD or number (%) of patients. CTPA = computed tomographic pulmonary angiography; Others = other major bleeding without death. See Table 1 for expansion of other abbreviations.
Efficacy and Safety in Patients with Different Body Weights
We further compared the efficacy and safety of the two dose regimens in patients with different body weights. Patients were divided into groups either by the tertiles of body weight or by the tertiles of BMI. As shown in Table 5 , the CTPA scores did not differ significantly between the two treatments in the high-, medium-, or low-body-weight groups. However, the total number of bleeding episodes was significantly lower in the 50 mg rt-PA group than in the 100 mg rt-PA group, especially in patients with body weights < 65 kg (14.8% vs 41.2%, P = .049; age-sex adjusted risk ratio, 0.19, 95% CI 0.04–0.92) or BMI < 24 kg/m2 (8.7% vs 42.9%, P = .014; age-sex adjusted risk ratio, 0.05, 95% CI 0.01–0.57). Similar findings were found when patients were grouped according to BMI < 25 kg/m2 and BMI ≥ 25 kg/m2 (Fig 3 ).
Table 5.
Comparison of Improvement of Pulmonary Artery Obstructions Between Two Treatments for PTE in Patients With Different Body Weights and BMI Subgroups
| Subgroups | rt-PA 100 mg | rt-PA 50 mg | P |
|---|---|---|---|
| Body weight tertiles | |||
| < 65 kg | (n = 15) | (n = 22) | |
| baseline | 28.7 ± 13.6 | 23.8 ± 12.3 | .263 |
| 14 d | 9.5 ± 7.4 | 8.2 ± 6.5 | .592 |
| 65-74 kg | (n = 13) | (n = 13) | … |
| baseline | 27.4 ± 12.2 | 28.2 ± 13.0 | .878 |
| 14 d | 16.1 ± 13.1 | 11.7 ± 11.0 | .377 |
| ≥ 75 kg | (n = 20) | (n = 19) | … |
| baseline | 27.1 ± 12.6 | 27.3 ± 10.9 | .944 |
| 14 d | 11.8 ± 8.5 | 10.1 ± 7.2 | .504 |
| BMI tertiles | |||
| < 24 kg/m2 | (n = 13) | (n = 18) | |
| baseline | 24.8 ± 12.4 | 23.2 ± 11.8 | .726 |
| 14 d | 10.9 ± 7.9 | 8.5 ± 6.7 | .366 |
| 24-25.9 kg/m2 | (n = 17) | (n = 14) | … |
| baseline | 28.5 ± 13.5 | 24.2 ± 12.7 | .371 |
| 14 d | 13.4 ± 12.6 | 10.5 ± 10.3 | .501 |
| ≥ 26 kg/m2 | (n = 15) | (n = 20) | … |
| baseline | 31.2 ± 9.7 | 29.9 ± 11.6 | .667 |
| 14 d | 13.3 ± 8.3 | 10.7 ± 8.2 | .356 |
| BMI groups | |||
| < 25 kg/m2 | (n = 21) | (n = 30) | … |
| baseline | 27.1 ± 11.8 | 22.6 ± 11.6 | .179 |
| 14 d | 13.1 ± 11.3 | 9.5 ± 8.4 | .192 |
| ≥ 25 kg/m2 | (n = 24) | (n = 22) | … |
| baseline | 29.4 ± 12.4 | 30.5 ± 11.2 | .749 |
| 14 d | 12.2 ± 8.7 | 10.4 ± 8.2 | .457 |
Data presented are mean ± SD unless otherwise indicated. See Table 1 for expansion of abbreviations.
Figure 3.
Comparisons of total bleeding complications between two treatments for PTE in patients with different body weights and BMI subgroups. See Figure 1, Figure 2 legends for expansion of other abbreviations.
Discussion
The 50 mg rt-PA regimen exhibited the same efficacy as the 100 mg rt-PA regimen in PTE thrombolytic therapy. The two regimens showed similar progressive improvements in RVDs, lung perfusion defects, and pulmonary artery obstructions in patients with either hemodynamic instability or massive pulmonary artery obstruction. Although the overall prevalence of death and PTE recurrence was similar between the two treatments, the 50 mg rt-PA group presented with less bleeding, especially in low-body-weight patients.
Thrombolytic Efficacy
Although rt-PA has been shown to be effective in treating acute PTE, the optimal regimens are less defined.22, 23, 24, 25 Several studies have indicated that lower-dose rt-PA infusion may be similarly effective in improving clinical presentation and lung perfusion defects in PTE compared with the conventional rt-PA 100 mg regimen.8, 26, 27 Consistent with these studies, our study clearly showed that the rt-PA 50 mg/2 h regimen produced similar improvements in right ventricular functions, lung perfusion defects, and pulmonary artery obstructions as compared with the rt-PA 100 mg/2 h regimen.
In our practice, we use thrombolytic therapy instead of anticoagulation therapy alone to treat patients with extensive pulmonary artery obstruction (defined as an obstruction exceeding 50% of the pulmonary vasculature or the occlusion of two or more lobar arteries with RVD).28, 29, 30, 31 Even when presented with normal systemic arterial pressure, these patients are at risk for deteriorating conditions and have worse prognoses.21, 32 Studies have shown that these patients may benefit from thrombolytic therapy.13, 33 In the current study, therefore, we included these patients. Although this inclusion may make our efficacy comparison of the two regimens less conclusive, subgroup analysis showed that the 50 mg rt-PA regimen was effective not only for patients with massive pulmonary artery obstruction but also for patients with hemodynamic instability. Nevertheless, future studies with anticoagulation control in patients with massive pulmonary artery obstruction will be valuable in determining whether thrombolytic therapy is indeed beneficial in these patients.
Further comparison of the two dose regimens in patients with different body weights showed that the two treatments produced similar efficacy in high-, medium-, or low-body-weight groups. This observation indicates that both doses may have reached therapeutic threshold and are similarly effective in all body-weight subgroups in our patients. However, additional analysis on patients with body weights greater than 100 kg or BMIs greater than 30 kg/m2 was not conducted in our study because of the small sample size. Further studies are desirable to examine the efficacy of the 50 mg regimen in this patient population.
Adverse Events
Thrombolysis is associated with the risk of bleeding complications.34, 35, 36 ICH is one of the most serious complications of thrombolytic therapy.37 Although the difference was not statistically significant, previous studies demonstrated that the prevalence of ICH was relatively higher in patients receiving 100 mg rt-PA.2, 26, 37 In our study, we observed that the overall bleeding rate was significantly lower in the 50 mg rt-PA group than that in the 100 mg ft-PA group. This difference was even more obvious in patients with a body weight < 65 kg or BMI < 24 kg/m2. A similar trend was also found in major bleeding. The rate of major bleeding in our study (3%–10%) is comparable to previously reported trials.5, 26, 27, 34, 35 Our findings suggest that an rt-PA dose of 50 mg may be safer, especially in patients with lower body weights. We recommend that this dose can be safely used for patients with body weight < 65 kg. Further studies with larger patient populations would be useful to validate these findings.
One major criterion in evaluating thrombolytic therapy for PTE is the recurrence of embolisms, which occur mostly during the first week of follow-up after initial treatment and can result in a high mortality rate. The PTE recurrence was low in the two therapy groups: one patient (2%) in the 50 mg rt-PA group and two (4%) in the 100 mg rt-PA group, which is consistent with previous studies.27, 38
Although the difference is far from significant (P = .42) with our sample size, our study showed that the mortality was lower in the 50 mg rt-PA group (2%) than in the 100 mg rt-PA group (6%). This seems the result of both less fatal bleeding and less recurrent PTE in the 50 mg rt-PA group. The mortality rates in our study (2%–6%) are comparable to those in previous clinical trials,5, 26, 27, 34, 35 but significantly lower than those in the International Cooperative Pulmonary Embolism Registry study (23%).38 The high mortality rate in the retrospective registry study was probably the result of the enrollment of more seriously ill patients.
Study Limitations
There are a few limitations in our study. First, the small sample size, a common limitation for PTE thrombolytic studies, prevented us from achieving more powerful efficacy and safety analyses. This is particularly true for the efficacy analysis in patients with hemodynamic instability and in patients with higher body weight, as well as for the mortality and PTE recurrence evaluations in all patients. Future studies with larger sample sizes are desirable to examine the efficacy and safety of the 50 mg regimen in these patient populations. Second, although physicians evaluating the imaging studies were blinded to the treatment assignments, physicians treating patients were not. This open-label study may potentially introduce bias to a certain degree. Last, a control group using heparin alone was not included for patients with extensive pulmonary artery obstruction and right ventricular dysfunction. Future studies using anticoagulation control will be valuable to determine whether thrombolytic therapy is indeed beneficial in these patients.
Conclusions
This study showed that 50 mg/2 h and 100 mg/2 h rt-PA regimens exhibited similar efficacy in patients with PTE and either hemodynamic instability or with massive pulmonary artery obstruction. Although the death rates and PTE recurrence rates were similar between the two treatments, the 50 mg/2 h rt-PA regimen had less bleeding tendency, especially in patients with lower body weights. Thus, compared with the rt-PA at 100 mg/2 h regimen, the rt-PA at 50 mg/2 h regimen is as effective and perhaps safer in treating lower-body-weight patients with PTE. These findings support the notion that optimizing rt-PA dosing is valuable when treating patients with acute PTE.
Acknowledgments
Author contributions: All authors made substantial contributions to conception and design, or acquisition of data, or analysis and interpretation of data; reviewed and approved the final manuscript; and contributed significantly to this study. Drs Wang, Zhai, and Yang contributed equally to the work.
Dr Wang: the principal investigator, takes full responsibility for the integrity of the submission and publication, and was involved in the study design as part of the steering committee.
Dr Zhai: had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis, and was responsible for the data verification, analysis, and draft of the manuscript.
Dr Yang: had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Dr Wu: was responsible for the patient enrollment and data collection.
Dr Z. Cheng: was responsible for the patient enrollment and data collection.
Dr Liang: had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Dr Dai: was responsible for the patient enrollment and data collection.
Dr Huang: was responsible for the patient enrollment and data collection.
Dr Lu: was involved in study design as part of the steering committee.
Dr Zhang: was involved in study design as part of the steering committee.
Dr X. Cheng: was involved in study design as part of the steering committee.
Dr Shen: was responsible for the data verification, analysis, and draft of the manuscript.
Financial/nonfinancial disclosures: The authors have reported to CHEST that no potential conflicts of interest exist with any companies/organizations whose products or services may be discussed in this article.
Other contributions: We are grateful to Drs Charles A. Hales, Jason X. J. Yuan, and Lan Zhao for their valuable suggestions on this manuscript.
Footnotes
Funding/Support: This study was supported by the China Key Research Projects of the 10th National Five-Year Development Plan (2004BA703B07 to Dr Wang).
Reproduction of this article is prohibited without written permission from the American College of Chest Physicians (www.chestjournal.org/site/misc/reprints.xhtml).
Appendix
All of the investigators in the China Venous Thromboembolism (VTE) Study Group contributed to this study. The members of the study group were as follows: Steering Committee: Chen Wang, Xiansheng Cheng, Xinzhi Weng, Weixuan Lu, Quanying He, Yiming Zhao, Binrong Ma, and Nanshan Zhong; Independent Central Adjudication Committee: Xiansheng Cheng, Ruping Dai, Youmin Guo, Tie Wang, Weixuan Lu, Quanying He, Yiming Zhao, and Chen Wang; Data and Coordinating Center: Yuanhua Yang, Zhenguo Zhai, and Lirong Liang. The following investigators participated in this study: Beijing Chao-Yang Hospital, Capital Medical University: Chen Wang, Yuanhua Yang, Zhenguo Zhai, Baosen Pang, Yafeng Wu, Xiaojuan Wang, Tie Wang, Lei Zhang, Xin Deng, Kewu Huang, Huaping Dai, and Zhaohui Tong; Tianjin Medical University General Hospital: Qi Wu, Yanling Yin, and Wei Zhou; The Affiliated Hospital of Medical College Qingdao: Zhaozhong Cheng and Handong Jiang; The First Affiliated Hospital of Wenzhou Medical College: Shaoxian Chen and Yupeng Xie; The General Hospital of Shenyang Military Command: Zhuang Ma, Ping Chen, and Lei Liu; The Affiliated Hospital of Ningxia Medical University: Jin Zhang and Xiwei Zheng; Shenzhen People's Hospital: Shengwen Chen and Chen Qiu; The First Affiliated Hospital of Shanxi Medical University: Yongcheng Du, Jianying Xu, and Xiaoyun Hu; The Second Affiliated Hospital of Hebei Medical University: Yadong Yuan and Baofa Wang; The First Hospital of China Medical University: Jian Kang and Lili Tian; Beijing Friendship Hospital, Capital Medical University: Zhengyi He; Peking University Third Hospital: Yongchang Sun and Wanzhen Yao; Beijing Fu Xing Hospital, Capital Medical University: Yuxiang Liu; The Second Affiliated Hospital of Shanxi Medical University: Zhuola Liu and Xu Wang; Peking Union Medical College Hospital, Chinese Academy of Medical Sciences: Weixuan Lu, Chunping Liu, and Yongjian Liu; Guangzhou Institute of Respiratory Disease, Guangzhou Medical University: Nanshan Zhong, Rongchang Chen, and Hua Wu; The Third Affiliated Hospital, Sun Yat-Sen University: Tiantuo Zhang; Sir Run Run Shaw Hospital, affiliated with Zhejiang University: Kejing Ying and Liying Chen; Wuhan Union Hospital: Ming Bai; Peking University First Hospital: Guangfa Wang and Chunhua Chi; The Affiliated Hospital of Shenyang Medical University: Shi Wang and Shuyue Xia; Peking University People's Hospital: Quanying He and Xingyu Tan; Tangshan Worker's Hospital, Hebei Medical University: Huilin Liu and Yingqi Zhang; The First Affiliated Hospital, Sun Yat-Sen University: Canmao Xie and Mian Zeng; The First Affiliated Hospital of Guangxi Medical University: Yiqiang Chen, Shenglan Guo, and Tangwei Liu; The First Affiliated Hospital of Zhengzhou University: Peizong Sun and Jie Chen; The Affiliated Hospital of Hubei Coal University: Xinrong Liu and Hongyang Wang; Beijing Naval General Hospital: Yunyou Duan and Zhoushan Nie; Beijing No. 6 Hospital: Xiaoping Xiang and Chun Zhang; Beijing Hospital: Baomin Fang and Tieying Sun.
References
- 1.Goldhaber SZ. Tissue plasminogen activator in acute pulmonary embolism. Chest. 1989;95(5 Suppl):282S–289S. doi: 10.1378/chest.95.5_supplement.282s. [DOI] [PubMed] [Google Scholar]
- 2.Goldhaber SZ, Feldstein ML, Sors H. Two trials of reduced bolus alteplase in the treatment of pulmonary embolism. An overview. Chest. 1994;106(3):725–726. doi: 10.1378/chest.106.3.725. [DOI] [PubMed] [Google Scholar]
- 3.Kearon C, Kahn SR, Agnelli G, Goldhaber S, Raskob GE, Comerota AJ, American College of Chest Physicians Antithrombotic therapy for venous thromboembolic disease: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th Edition) Chest. 2008;133(6 Suppl):454S–545S. doi: 10.1378/chest.08-0658. [DOI] [PubMed] [Google Scholar]
- 4.Torbicki A, Perrier A, Konstantinides S, Task Force for the Diagnosis and Management of Acute Pulmonary Embolism of the European Society of Cardiology Guidelines on the diagnosis and management of acute pulmonary embolism: the Task Force for the Diagnosis and Management of Acute Pulmonary Embolism of the European Society of Cardiology (ESC) Eur Heart J. 2008;29(18):2276–2315. doi: 10.1093/eurheartj/ehn310. [DOI] [PubMed] [Google Scholar]
- 5.Goldhaber SZ, Kessler CM, Heit J. Randomised controlled trial of recombinant tissue plasminogen activator versus urokinase in the treatment of acute pulmonary embolism. Lancet. 1988;2(8606):293–298. doi: 10.1016/s0140-6736(88)92354-9. [DOI] [PubMed] [Google Scholar]
- 6.Goldhaber SZ, Vaughan DE, Markis JE. Acute pulmonary embolism treated with tissue plasminogen activator. Lancet. 1986;2(8512):886–889. doi: 10.1016/s0140-6736(86)90411-3. [DOI] [PubMed] [Google Scholar]
- 7.Braithwaite BD, Birch PA, Poskitt KR, Heather BP, Earnshaw JJ. Accelerated thrombolysis with high dose bolus t-PA extends the role of peripheral thrombolysis but may increase the risks. Clin Radiol. 1995;50(11):747–750. doi: 10.1016/s0009-9260(05)83212-x. [DOI] [PubMed] [Google Scholar]
- 8.Levine MN. Bolus, front-loaded, and accelerated thrombolytic infusions for myocardial infarction and pulmonary embolism. Chest. 1991;99(4 Suppl):128S–134S. doi: 10.1378/chest.99.4.128s. [DOI] [PubMed] [Google Scholar]
- 9.Ross AM, Gao R, Coyne KS, TUCC Investigators A randomized trial confirming the efficacy of reduced dose recombinant tissue plasminogen activator in a Chinese myocardial infarction population and demonstrating superiority to usual dose urokinase: the TUCC trial. Am Heart J. 2001;142(2):244–247. doi: 10.1067/mhj.2001.116963. [DOI] [PubMed] [Google Scholar]
- 10.Tebbe U, Graf A, Kamke W. Hemodynamic effects of double bolus reteplase versus alteplase infusion in massive pulmonary embolism. Am Heart J. 1999;138(1 Pt 1):39–44. doi: 10.1016/s0002-8703(99)70243-7. [DOI] [PubMed] [Google Scholar]
- 11.Goldhaber SZ. Evolving concepts in thrombolytic therapy for pulmonary embolism. Chest. 1992;101(4 Suppl):183S–185S. doi: 10.1378/chest.101.4_supplement.183s. [DOI] [PubMed] [Google Scholar]
- 12.Daniels LB, Parker JA, Patel SR, Grodstein F, Goldhaber SZ. Relation of duration of symptoms with response to thrombolytic therapy in pulmonary embolism. Am J Cardiol. 1997;80(2):184–188. doi: 10.1016/s0002-9149(97)00315-9. [DOI] [PubMed] [Google Scholar]
- 13.Ashton RW, Daniels CE, Ryu JH. Thrombolytic therapy in patients with submassive pulmonary embolism. N Engl J Med. 2003;348(4):357–359. doi: 10.1056/NEJM200301233480416. [DOI] [PubMed] [Google Scholar]
- 14.Kreit JW. The impact of right ventricular dysfunction on the prognosis and therapy of normotensive patients with pulmonary embolism. Chest. 2004;125(4):1539–1545. doi: 10.1378/chest.125.4.1539. [DOI] [PubMed] [Google Scholar]
- 15.Zhu L, Yang YH, Wu YF, Zhai ZG, Wang C, National Project of the Diagnosis and Treatment Strategies for Pulmonary Thromboembolism investigators Value of transthoracic echocardiography combined with cardiac troponin I in risk stratification in acute pulmonary thromboembolism. Chin Med J (Engl) 2007;120(1):17–21. [PubMed] [Google Scholar]
- 16.Arcasoy SM, Kreit JW. Thrombolytic therapy of pulmonary embolism: a comprehensive review of current evidence. Chest. 1999;115(6):1695–1707. doi: 10.1378/chest.115.6.1695. [DOI] [PubMed] [Google Scholar]
- 17.Goldhaber SZ. Thrombolysis in venous thromboembolism. An international perspective. Chest. 1990;97(4 Suppl):176S–181S. [PubMed] [Google Scholar]
- 18.Stein PD, Coleman RE, Gottschalk A, Saltzman HA, Terrin ML, Weg JG. Diagnostic utility of ventilation/perfusion lung scans in acute pulmonary embolism is not diminished by pre-existing cardiac or pulmonary disease. Chest. 1991;100(3):604–606. doi: 10.1378/chest.100.3.604. [DOI] [PubMed] [Google Scholar]
- 19.Parker JA, Markis JE, Palla A. Pulmonary perfusion after rt-PA therapy for acute embolism: early improvement assessed with segmental perfusion scanning. Radiology. 1988;166(2):441–445. doi: 10.1148/radiology.166.2.3122266. [DOI] [PubMed] [Google Scholar]
- 20.Qanadli SD, El Hajjam M, Vieillard-Baron A. New CT index to quantify arterial obstruction in pulmonary embolism: comparison with angiographic index and echocardiography. AJR Am J Roentgenol. 2001;176(6):1415–1420. doi: 10.2214/ajr.176.6.1761415. [DOI] [PubMed] [Google Scholar]
- 21.van der Meer RW, Pattynama PM, van Strijen MJ. Right ventricular dysfunction and pulmonary obstruction index at helical CT: prediction of clinical outcome during 3-month follow-up in patients with acute pulmonary embolism. Radiology. 2005;235(3):798–803. doi: 10.1148/radiol.2353040593. [DOI] [PubMed] [Google Scholar]
- 22.Capstick T, Henry MT. Efficacy of thrombolytic agents in the treatment of pulmonary embolism. Eur Respir J. 2005;26(5):864–874. doi: 10.1183/09031936.05.00002505. [DOI] [PubMed] [Google Scholar]
- 23.Baruah DB, Dash RN, Chaudhari MR, Kadam SS. Plasminogen activators: a comparison. Vascul Pharmacol. 2006;44(1):1–9. doi: 10.1016/j.vph.2005.09.003. [DOI] [PubMed] [Google Scholar]
- 24.Agnelli G, Parise P. Bolus thrombolysis in venous thromboembolism. Chest. 1992;101(4 Suppl):172S–182S. doi: 10.1378/chest.101.4_supplement.172s. [DOI] [PubMed] [Google Scholar]
- 25.Meyer G, Collignon MA, Guinet F, Jeffrey AA, Barritault L, Sors H. Comparison of perfusion lung scanning and angiography in the estimation of vascular obstruction in acute pulmonary embolism. Eur J Nucl Med. 1990;17(6–8):315–319. doi: 10.1007/BF01268022. [DOI] [PubMed] [Google Scholar]
- 26.Goldhaber SZ, Agnelli G, Levine MN, The Bolus Alteplase Pulmonary Embolism Group Reduced dose bolus alteplase vs conventional alteplase infusion for pulmonary embolism thrombolysis. An international multicenter randomized trial. Chest. 1994;106(3):718–724. doi: 10.1378/chest.106.3.718. [DOI] [PubMed] [Google Scholar]
- 27.Sors H, Pacouret G, Azarian R, Meyer G, Charbonnier B, Simonneau G. Hemodynamic effects of bolus vs 2-h infusion of alteplase in acute massive pulmonary embolism. A randomized controlled multicenter trial. Chest. 1994;106(3):712–717. doi: 10.1378/chest.106.3.712. [DOI] [PubMed] [Google Scholar]
- 28.Meneveau N, Schiele F, Metz D. Comparative efficacy of a two-hour regimen of streptokinase versus alteplase in acute massive pulmonary embolism: immediate clinical and hemodynamic outcome and one-year follow-up. J Am Coll Cardiol. 1998;31(5):1057–1063. doi: 10.1016/s0735-1097(98)00068-0. [DOI] [PubMed] [Google Scholar]
- 29.Miller GA, Sutton GC. Acute massive pulmonary embolism. Clinical and haemodynamic findings in 23 patients studied by cardiac catheterization and pulmonary arteriography. Br Heart J. 1970;32(4):518–523. doi: 10.1136/hrt.32.4.518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Tow DE, Simon AL. Comparison of lung scanning and pulmonary angiography in the detection and follow-up of pulmonary embolism: the Urokinase-Pulmonary Embolism Trial experience. Prog Cardiovasc Dis. 1975;17(4):239–245. doi: 10.1016/s0033-0620(75)80015-6. [DOI] [PubMed] [Google Scholar]
- 31.Morgenthaler TI, Ryu JH. Clinical characteristics of fatal pulmonary embolism in a referral hospital. Mayo Clin Proc. 1995;70(5):417–424. doi: 10.4065/70.5.417. [DOI] [PubMed] [Google Scholar]
- 32.Lim KE, Chan CY, Chu PH, Hsu YY, Hsu WC. Right ventricular dysfunction secondary to acute massive pulmonary embolism detected by helical computed tomography pulmonary angiography. Clin Imaging. 2005;29(1):16–21. doi: 10.1016/j.clinimag.2004.04.023. [DOI] [PubMed] [Google Scholar]
- 33.Konstantinides S, Geibel A, Heusel G, Heinrich F, Kasper W, Management Strategies and Prognosis of Pulmonary Embolism-3 Trial Investigators Heparin plus alteplase compared with heparin alone in patients with submassive pulmonary embolism. N Engl J Med. 2002;347(15):1143–1150. doi: 10.1056/NEJMoa021274. [DOI] [PubMed] [Google Scholar]
- 34.Meyer G, Sors H, Charbonnier B, The European Cooperative Study Group for Pulmonary Embolism Effects of intravenous urokinase versus alteplase on total pulmonary resistance in acute massive pulmonary embolism: a European multicenter double-blind trial. J Am Coll Cardiol. 1992;19(2):239–245. doi: 10.1016/0735-1097(92)90472-y. [DOI] [PubMed] [Google Scholar]
- 35.Goldhaber SZ, Kessler CM, Heit JA. Recombinant tissue-type plasminogen activator versus a novel dosing regimen of urokinase in acute pulmonary embolism: a randomized controlled multicenter trial. J Am Coll Cardiol. 1992;20(1):24–30. doi: 10.1016/0735-1097(92)90132-7. [DOI] [PubMed] [Google Scholar]
- 36.Meneveau N, Schiele F, Vuillemenot A. Streptokinase vs alteplase in massive pulmonary embolism. A randomized trial assessing right heart haemodynamics and pulmonary vascular obstruction. Eur Heart J. 1997;18(7):1141–1148. doi: 10.1093/oxfordjournals.eurheartj.a015410. [DOI] [PubMed] [Google Scholar]
- 37.Kanter DS, Mikkola KM, Patel SR, Parker JA, Goldhaber SZ. Thrombolytic therapy for pulmonary embolism. Frequency of intracranial hemorrhage and associated risk factors. Chest. 1997;111(5):1241–1245. doi: 10.1378/chest.111.5.1241. [DOI] [PubMed] [Google Scholar]
- 38.Goldhaber SZ, Visani L, De Rosa M. Acute pulmonary embolism: clinical outcomes in the International Cooperative Pulmonary Embolism Registry (ICOPER) Lancet. 1999;353(9162):1386–1389. doi: 10.1016/s0140-6736(98)07534-5. [DOI] [PubMed] [Google Scholar]