Abstract
Objective
To determine the effectiveness of management strategies for uninvestigated dyspepsia.
Design
Systematic review and network meta-analysis.
Data sources
Medline, Embase, Embase Classic, the Cochrane Central Register of Controlled Trials, and clinicaltrials.gov from inception to September 2019, with no language restrictions. Conference proceedings between 2001 and 2019.
Eligibility criteria for selecting studies
Randomised controlled trials that assessed the effectiveness of management strategies for uninvestigated dyspepsia in adult participants (age ≥18 years). Strategies of interest were prompt endoscopy; test for Helicobacter pylori and perform endoscopy in participants who test positive; test for H pylori and eradication treatment in those who test positive (“test and treat”); empirical acid suppression; or symptom based management. Trials reported dichotomous assessment of symptom status at final follow-up (≥12 months).
Results
The review identified 15 eligible randomised controlled trials that comprised 6162 adult participants. Data were pooled using a random effects model. Strategies were ranked according to P score, which is the mean extent of certainty that one management strategy is better than another, averaged over all competing strategies. “Test and treat” ranked first (relative risk of remaining symptomatic 0.89, 95% confidence interval 0.78 to 1.02, P score 0.79) and prompt endoscopy ranked second, but performed similarly (0.90, 0.80 to 1.02, P score 0.71). However, no strategy was significantly less effective than “test and treat.” Participants assigned to “test and treat” were significantly less likely to receive endoscopy (relative risk v prompt endoscopy 0.23, 95% confidence interval 0.17 to 0.31, P score 0.98) than all other strategies, except symptom based management (relative risk v symptom based management 0.60, 0.30 to 1.18). Dissatisfaction with management was significantly lower with prompt endoscopy (P score 0.95) than with “test and treat” (relative risk v “test and treat” 0.67, 0.46 to 0.98), and empirical acid suppression (relative risk v empirical acid suppression 0.58, 0.37 to 0.91). Upper gastrointestinal cancer rates were low in all trials. Results remained stable in sensitivity analyses, with minimal inconsistencies between direct and indirect results. Risk of bias of individual trials was high; blinding was not possible because of the pragmatic trial design.
Conclusions
“Test and treat” was ranked first, although it performed similarly to prompt endoscopy and was not superior to any of the other strategies. “Test and treat” led to fewer endoscopies than all other approaches, except symptom based management. However, participants showed a preference for prompt endoscopy as a management strategy for their symptoms.
Systematic review registration
PROSPERO registration number CRD42019132528.
Introduction
Dyspepsia is a common condition that could involve a variety of upper gastrointestinal symptoms, but the main symptom is upper abdominal pain or discomfort.1 At some point in their lives, one in five adults report epigastric pain, early satiety, postprandial distress, and other associated upper gastrointestinal symptoms, such as heartburn, regurgitation, or nausea. Although dyspepsia is not associated with higher mortality risk,2 3 the condition is chronic in many people4 and follows a fluctuating course.5 6 7 Dyspepsia has a substantial impact on patients’ quality of life,8 and is associated with more time off work and lower productivity at work, and greater medical and prescription drug costs each year.9 10 The financial implications for society as a whole are huge.11
Approximately 40% of people with dyspepsia symptoms will consult a primary care physician.12 The physician has to make a decision about how best to manage the individual patient. Patients with uninvestigated dyspepsia and alarm features, such as dysphagia, weight loss, or anaemia, or those older than a certain age threshold, require urgent endoscopy. However, the management of uninvestigated dyspepsia in the absence of alarm features represents a classic medical decision making problem because several strategies exist. These strategies include prompt endoscopy for all patients; test for Helicobacter pylori and perform endoscopy in those who test positive (“test and scope”); test for H pylori and eradication treatment in those who test positive (“test and treat”); empirical acid suppression for all patients; or symptom based management according to guideline recommendations or the physician’s usual practice.
The effectiveness of these different strategies has been studied in numerous pragmatic randomised controlled trials.13 14 15 16 17 However, there is equipoise among various strategies and uncertainty as to which strategy is best to use first line. Trial based meta-analyses, and even individual patient data meta-analyses, have been unable to resolve this uncertainty completely. Although prompt endoscopy is expensive, it appears to be superior to empirical acid suppression or symptom based management when comparing the effect on symptoms in some patients,15 18 and was superior to “test and treat” in an individual patient data meta-analysis.19 However, it is unlikely to be cost effective,19 and therefore is not recommended as first line treatment in management guidelines for uninvestigated dyspepsia.20 21 Another individual patient data meta-analysis of “test and treat” versus empirical acid suppression showed no difference in either costs or effects between the two strategies.22 As a result, guidelines disagree about which approach should be used for the initial management of uninvestigated dyspepsia (table 1).20 21 23
Table 1.
Recommendations from previous guidelines on various initial management strategies for uninvestigated dyspepsia
| Guideline | When to endoscope | When to use “test and treat” | When to use empirical acid suppression therapy |
|---|---|---|---|
| ACG and CAG 2017 (North America)20 | First line in people aged ≥60 years; strength of recommendation: conditional; level of evidence: very low. First line in those aged ≥60 years with alarm features; strength of recommendation: conditional; level of evidence: moderate | First line in those aged <60 years; likely to be cost effective even with low rates of infection because of reduction in gastric cancer rates in infected individuals; strength of recommendation: strong; level of evidence: high | First line in those aged <60 years if H pylori negative, or in those who remain symptomatic after eradication therapy; use empirical proton pump inhibitor treatment at standard dose; strength of recommendation: strong; level of evidence: high |
| NICE 2014 (England and Wales)21 | First line in people aged ≥55 years with weight loss and dyspepsia; consider when Helicobacter pylori eradication or empirical acid suppression fails; strength of recommendation: “offer”*; level of evidence: high | First line in people with dyspepsia; if this fails use empirical acid suppression with full dose proton pump inhibitor; strength of recommendation: “offer”*; level of evidence: high | First line in people with dyspepsia; use empirical full dose proton pump inhibitor treatment for four weeks; if this fails use “test and treat”; strength of recommendation: “offer”*; level of evidence: high |
| Asia-Pacific Working Party 199823 | First line in people aged 35-55 years (depending on risk of gastric cancer in region) or alarm features (any age); if H pylori eradication or empirical acid suppression fails consider in younger patients; strength of recommendation: not stated; level of evidence: not reported | Consider if empirical acid suppression fails; in areas with high prevalence of H pylori this strategy is unlikely to be beneficial; strength of recommendation: not stated; level of evidence: not reported | First line for young patients with no alarm features; either proton pump inhibitor or histamine 2 receptor antagonists at standard dose for two to four weeks; strength of recommendation: not stated; level of evidence: not reported |
ACG=American College of Gastroenterology; CAG=Canadian Association of Gastroenterology; NICE=National Institute for Health and Care Excellence.
“Offer,” for most patients, an intervention will do more good than harm.
Network meta-analysis might be able to resolve some of this uncertainty because the methods used allow indirect and direct comparisons across different randomised controlled trials, which increases the number of participants’ data available for analysis. Additionally, network meta-analysis allows a credible ranking system to be developed that shows the effectiveness of different management strategies, even in the absence of trials making direct comparisons, which can help to inform clinical decision making. Therefore, we conducted a network meta-analysis of all available randomised controlled trials that have compared five management strategies for uninvestigated dyspepsia.
Methods
Search strategy and study selection
We searched Medline (from 1947 to September 2019), Embase, Embase Classic (from 1947 to September 2019), and the Cochrane Central Register of Controlled Trials to identify potential studies. In addition, we searched national guidelines for the management of dyspepsia, clinicaltrials.gov for unpublished trials, and supplementary data for potentially eligible studies (all up to September 2019). Conference proceedings (Digestive Disease Week, American College of Gastroenterology, United European Gastroenterology Week, and the Asian Pacific Digestive Week) between 2001 and 2019 were hand searched to identify studies published only in abstract form. Finally, we performed a recursive search by using the bibliographies of all obtained articles.
Eligible randomised controlled trials examined the effect of various management strategies for uninvestigated dyspepsia (prompt endoscopy, “test and treat,” “test and scope,” empirical acid suppression, or symptom based management) in adult participants (age ≥18 years). The definition of dyspepsia was broad and included any upper gastrointestinal symptoms referable to the gastroduodenum. We only considered randomised controlled trials to be eligible when they examined the effectiveness of one of the strategies of interest and compared it with at least one of the other strategies. Because dyspepsia is a chronic fluctuating condition,4 a minimum follow-up of 12 months was required. We extracted all endpoints at the final point of follow-up to ensure as much homogeneity as possible among individual trial results, and to avoid overestimating the effectiveness of one management strategy relative to another. Studies had to report a dichotomous assessment of symptom status at the final point of follow-up (box 1). The study protocol was published on the PROSPERO international prospective register of systematic reviews (registration number CRD42019132528).
Box 1. Eligibility criteria.
Randomised controlled trials
Adults (aged ≥18 years)
Uninvestigated dyspepsia, before first investigation
Compared strategy of interest with at least one other strategy: prompt endoscopy, “test and scope,” “test and treat,” empirical acid suppression, or symptom based management
Minimum follow-up duration of 12 months
Dichotomous assessment of dyspeptic symptoms at minimum of 12 months
Two investigators (LHE and ACF) conducted the literature search independently from each other. We report the search strategy in the supplementary materials. There were no language restrictions. Two investigators (LHE and ACF) evaluated all abstracts identified by the search for eligibility, again independently from each other. We obtained all potentially relevant papers and evaluated them in more detail by using predesigned forms to assess eligibility independently, according to the predefined criteria. We translated foreign language papers if required. Disagreements between investigators were resolved by discussion.
Outcome assessment
We assessed the effectiveness of all five management strategies of uninvestigated dyspepsia by comparing the probability of being symptomatic at the final point of follow-up. Additionally, because individual trials reported several other secondary endpoints, we were able to assess the likelihood of participants receiving endoscopy in each treatment arm, and dissatisfaction with management. Finally, we recorded rates of upper gastrointestinal cancer detection.
Data extraction
Two investigators (LHE and ACF) extracted all data independently onto a Microsoft Excel spreadsheet (XP professional edition) as dichotomous outcomes (symptomatic or asymptomatic at final point of follow-up). For all included studies, we also extracted the following data for each trial, when available: country of origin, setting, duration of follow-up, age range of included participants, proportion of female participants, proportion of participants with H pylori infection, and exact management strategy used. Data were extracted as intention to treat analyses, with dropouts assumed to be treatment failures (that is, symptomatic at final point of follow-up), by using the total number of participants randomised to each treatment arm as the denominator, wherever trial reporting allowed. Given the duration of follow-up in individual trials, we also performed a sensitivity analysis by using a per protocol analysis and including all participants with reported evaluable data at the final point of follow-up.
Quality assessment and risk of bias
This assessment was performed at the study level by two investigators (LHE and ACF) independently by using the Cochrane risk of bias tool.24 Disagreements were resolved by discussion. We recorded the methods used to generate the randomisation schedule and conceal treatment allocation. We also noted whether blinding was implemented for participants, personnel, and outcomes assessment, whether there was evidence of incomplete outcomes data, and whether there was evidence of selective reporting of outcomes.
Data synthesis and statistical analysis
We performed a network meta-analysis by using the frequentist model with the statistical package “netmeta” (version 0.9-0, https://cran.r-project.org/web/packages/netmeta/index.html) in R (version 3.4.2). Firstly, we performed a pairwise meta-analysis of the raw data (supplementary figs 1-3) to convert them from contrast based format to long format, and to generate the treatment effect and standard error of the treatment effect for each pairwise treatment comparison. Subsequently, we used these data to conduct a network meta-analysis by using netmeta, which assumes a common τ2 for all pairwise comparisons. The estimate of τ2 is based on the generalised DerSimonian-Laird method.25 Uncertainty is not accounted for fully in this model because the distribution of parameters such as the between study variance is not assumed. In multiarm studies, all pairwise comparisons are considered, not only those with a common comparator, but are downweighted.25 We reported the network meta-analysis according to the PRISMA (preferred reporting items for systematic reviews and meta-analyses) extension statement for network meta-analyses.26 Network meta-analysis results usually give a more precise estimate compared with results from standard, pairwise analyses,27 28 and can rank management strategies to inform clinical decisions.29
We examined the symmetry and geometry of the evidence by producing a network plot with node and connection size corresponding to the number of study participants and number of studies, respectively. We produced comparison adjusted funnel plots to explore publication bias or other small study effects for all available comparisons by using Stata (version 14, Stata, College Station, TX). This is a scatterplot of effect size versus precision, measured through the inverse of the standard error. Symmetry around the effect estimate line indicates the absence of publication bias, or small study effects.30 We produced a pooled relative risk with 95% confidence interval to summarise the effectiveness of each management strategy tested by using a random effects model as a conservative estimate. We used the relative risk of remaining symptomatic at the final point of follow-up; when the relative risk is less than one and the 95% confidence interval does not cross one, there is a substantial benefit of one management strategy over another. Because there were direct comparisons between all of the management strategies, we were able to perform consistency modelling to check the agreement between direct and indirect evidence.31
Many meta-analyses use the I2 statistic to measure heterogeneity, which ranges between 0% and 100%.32 This statistic is easy to interpret and does not vary with the number of studies. However, the I2 value can increase with the number of patients included in the meta-analysis.33 Therefore, we assessed global statistical heterogeneity across all comparisons using the τ2 measure from the netmeta statistical package. Estimates of τ2 of approximately 0.04, 0.16, and 0.36 are considered to represent a low, moderate, and high degree of heterogeneity, respectively.34 We assessed inconsistency in the network analysis by comparing direct and indirect evidence, when available, by producing a network heat plot.31 35 These plots have grey squares, which represent the size of the contribution of the direct estimate in columns, compared with the network estimate in rows.35 The coloured squares around these represent the degree of inconsistency, with red squares indicating “hotspots” of inconsistency. We planned to remove studies that introduced any red “hotspots” and to repeat the analyses to investigate sources of potential inconsistency. We also applied the χ2 test of the Q statistic to test for inconsistency, under the assumption of a full design by treatment interaction random effects model.35 36 Finally, we tested for local inconsistency by splitting the network estimates into the contribution of direct and indirect evidence, and looking for any statistically significant differences.
We ranked management strategies according to their P score, which is between 0 and 1. P scores are based solely on the point estimates and standard errors of the network estimates, and measure the mean extent of certainty that one management strategy is better than another, averaged over all competing strategies.37 Higher scores indicate a greater probability of the strategy being ranked as best,37 but the magnitude of the P score should be considered in addition to the rank. Because the mean P score is always 0.5, individual strategies that cluster around this score are likely to be of similar effectiveness. However, when interpreting the results, it is also important to take into account the relative risk and corresponding 95% confidence interval for each comparison, rather than relying on rankings alone.38 In our primary analysis, we pooled data for the risk of being symptomatic at the final point of follow-up in each study for all included randomised controlled trials by using an intention to treat analysis. We also performed a per protocol analysis, and conducted analyses of the likelihood of receiving endoscopy, dissatisfaction with management among participants, and rates of upper gastrointestinal cancer.
We compared the relative effectiveness of all five management strategies using the “NetMetaXL” tool running in WinBUGS (version 1.4, Imperial College and MRC, London),39 which uses Bayesian methods. We used a random effects model with vague (uninformative) priors to achieve a conservative estimate of relative efficacy. Strategies were ranked according to their surface under the cumulative ranking curve value, which is comparable to the P score used in the frequentist model of our primary analyses.37 There were no differences in rankings among approaches, and therefore, for clarity, we only report the frequentist model in this paper, which is consistent with our approach for reporting previously published network meta-analyses.40 41 42 43 44
Because one of the studies was a cluster randomised trial,45 with patients assigned to treatment strategy by primary care practice, rather than randomised individually, we used the cluster size and the intra cluster correlation coefficient to reduce the size of the trial to its “effective sample size,” which was 440 participants (233 “test and treat” and 207 empirical acid suppression), before any data pooling was carried out.46 If clustering is ignored, a “unit of analysis error” can occur,47 which will overestimate the effect of the intervention in the study, and also mean the study’s weight in the meta-analysis is artificially high.
Patient and public involvement
This was a network meta-analysis of previously published randomised controlled trials. It was not possible for us to involve patients or the public in defining the research question, the design, or the evaluation and discussion of our work. We will disseminate our findings in lay terms through the national charity for people living with digestive diseases, “Guts UK.”
Results
The search strategy generated 8781 citations, 59 of which we retrieved for further assessment because they appeared to be relevant (supplementary fig 4). Of these, 44 were excluded for various reasons, which left 15 eligible randomised controlled trials that comprised 6162 participants. Fourteen trials were fully published,13 14 15 16 17 18 45 48 49 50 51 52 53 54 and data from another trial were available from a previous individual patient data meta-analysis conducted by our group.19 Agreement between investigators for trial eligibility was excellent (κ statistic=0.91). Supplementary table 1 reports risk of bias items for all included trials. Because the trials were all pragmatic, with blinding of participants impossible because of the differences in the strategies used, none was at low risk of bias.
Table 2 presents detailed characteristics of individual randomised controlled trials and the comparisons made. Six randomised controlled trials compared prompt endoscopy with “test and treat”17 19 48 49 50 51; three “test and treat” with empirical acid suppression14 45 52; two prompt endoscopy with empirical acid suppression13 53; one prompt endoscopy with symptom based management15; one “test and scope” with symptom based management16; one prompt endoscopy with empirical acid suppression or symptom based management54; and one prompt endoscopy with “test and scope,” “test and treat,” or empirical acid suppression.18 Direct evidence was therefore available for nine of the 10 possible comparisons. All trials were of 12 months’ duration, with the exception of two randomised controlled trials in which the final point of follow-up was 18 months.15 16
Table 2.
Characteristics of randomised controlled trials of management strategies for uninvestigated dyspepsia
| Study | Country, setting, and duration of follow-up | Characteristics of included participants | No of participants in each trial arm and management strategies used |
|---|---|---|---|
| Bytzer 199413 | Denmark, primary care, 12 months | 414 participants ≥18 years, mean age 44 years, 238 (57.5%) female | 208 participants prompt endoscopy with medical treatment according to endoscopic findings; 206 participants empirical acid suppression using ranitidine 150 mg twice daily for four weeks |
| Heaney 199948 | Northern Ireland, secondary care,* 12 months | 104 participants ≥18-45 years, mean age 32 years, 45 (43.3%) female, 104 (100%) Helicobacter pylori positive | 52 participants prompt endoscopy with medical treatment according to endoscopic findings; 52 participants “test and treat” by carbon 13 urea breath test, with those testing positive receiving eradication treatment with one week of omeprazole 20 mg twice daily, clarithromycin 250 mg twice daily, and tinidazole 500 mg twice daily |
| Delaney 200015 | England, primary care, 18 months | 442 participants ≥50 years, mean age 65 years, 222 (50.7%) female | 256 participants prompt endoscopy with medical treatment according to endoscopic findings; 186 participants symptom based management according to the primary care physician’s preferred strategy |
| Lassen 200049 | Denmark, secondary care,* 12 months | 500 participants ≥18 years, mean age 46 years, 270 (54.0%) female, 141 (28.2%) H pylori positive | 250 participants prompt endoscopy with medical treatment according to endoscopic findings; 250 participants “test and treat” by carbon 13 urea breath test, with those testing positive receiving eradication treatment with two weeks of lansoprazole 30 mg twice daily, metronidazole 500 mg three times daily, and amoxicillin 1 g twice daily; those testing negative received reassurance and lifestyle advice |
| Delaney 200116 | England, primary care, 18 months | 478 participants ≥18-49 years, mean age 37 years, 204 (42.9%) female, 112 (40.3%) of 278 in “test and scope” arm H pylori positive | 285 participants “test and scope” by serology, with endoscopy for H pylori positive participants and medical treatment according to endoscopic findings; those testing negative received empirical acid suppression; 193 participants symptom based management according to the primary care physician’s preferred strategy |
| Lewin van den Broek 200154 | The Netherlands, primary care, 12 months | 265 participants ≥18 years, mean age 43.5 years, 113 (45.9%) of 246 with data female | 86 participants prompt endoscopy with medical treatment according to endoscopic findings; 89 participants empirical acid suppression using omeprazole 20 mg once daily for up to eight weeks; 90 participants symptom based management according to national primary care guidelines |
| McColl 200217 | Scotland, secondary care,* 12 months | 708 participants ≥18-55 years, mean age 36 years, 331 (46.8%) female, 352 (49.7%) H pylori positive | 352 participants prompt endoscopy; also tested for H pylori by carbon 14 urea breath test, with those testing positive receiving eradication treatment as described below; 356 participants “test and treat” by carbon 14 urea breath test, with those testing positive receiving eradication treatment with one week of omeprazole 20 mg twice daily, clarithromycin 250 mg three times daily, and amoxicillin 500 mg or metronidazole 400 mg three times daily; those testing negative received reassurance |
| Arents 200350 | The Netherlands, primary care, 12 months | 270 participants ≥18 years, mean age 44 years, 141 (52.2%) female, 102 (37.8%) H pylori positive | 129 participants prompt endoscopy with medical treatment according to endoscopic findings; also tested for H pylori by serology, with those testing positive receiving eradication treatment, as described below; 141 participants “test and treat” by serology, with those testing positive receiving eradication treatment with one week of lansoprazole 30 mg twice daily, amoxicillin 1 g twice daily, and metronidazole or clarithromycin 500 mg twice daily; those testing negative received cisapride 20 mg twice daily for four weeks |
| Manes 200352 | Italy, secondary care,* 12 months | 219 participants ≥18-45 years, mean age 38.5 years, 99 (45.2%) female, 67 (60.9%) of 110 in “test and treat” arm H pylori positive | 110 participants “test and treat” by carbon 13 urea breath test, with those testing positive receiving eradication treatment with one week of omeprazole 20 mg twice daily, clarithromycin 500 mg twice daily, and tinidazole 500 mg twice daily; those testing negative received omeprazole 20 mg once daily for four week; 109 participants empirical acid suppression using omeprazole 20 mg once daily for four weeks |
| Jarbol 200645 | Denmark, primary care, 12 months | 472 participants ≥18 years, mean age 45.4 years, 272 (57.6%) female, 60 (24.0%) of 250 in “test and treat” arm H pylori positive | 250 participants “test and treat” by carbon 13 urea breath test, with those testing positive receiving eradication treatment with one week of esomeprazole 20 mg twice daily, clarithromycin 500 mg twice daily, and amoxicillin 1 g twice daily; those testing negative received no treatment; 222 participants empirical acid suppression using esomeprazole 20 mg twice daily for one week |
| Kjeldsen 200753 | Denmark, primary care, 12 months | 368 participants ≥18 years, mean age 48 years, 202 (54.9%) female | 184 participants prompt endoscopy with medical treatment according to endoscopic findings; 184 participants empirical acid suppression using omeprazole 40 mg once daily for two weeks |
| Delaney 200814 | England, primary care, 12 months | 699 participants ≥18-65 years, mean age 41 years, 355 (50.8%) female, 100 (29.2%) of 343 in “test and treat” arm H pylori positive | 343 participants “test and treat” by carbon 13 urea breath test, with those testing positive receiving eradication treatment with one week of omeprazole 20 mg once daily, clarithromycin 250 mg twice daily, and metronidazole 400 mg twice daily; those testing negative received omeprazole 20 mg once daily for four weeks; 356 participants empirical acid suppression using omeprazole 20 mg once daily for four weeks |
| Mahadeva 200851 | Malaysia, secondary care,* 12 months | 432 participants ≥18-45 years, mean age 30.5 years, 234 (54.2%) female, 141 (32.6%) H pylori positive | 210 participants prompt endoscopy with medical treatment according to endoscopic findings; also tested for H pylori by rapid urease test, with those testing positive receiving eradication treatment, as described below; 222 participants “test and treat” by carbon 13 urea breath test, with those testing positive receiving eradication treatment with one week of pantoprazole 40 mg twice daily, clarithromycin 500 mg twice daily, and amoxicillin 1 g twice daily; those testing negative received reassurance and symptom based treatment |
| Duggan 200918 | England, primary care, 12 months | 762 participants ≥18-70 years, mean age 42 years, 351 (46.1%) female, 277 (36.4%) H pylori positive | 187 participants prompt endoscopy with medical treatment according to endoscopic findings; 199 participants “test and scope” by serology, with endoscopy for H pylori positive participants and medical treatment according to endoscopic findings; those testing negative received lansoprazole 30 mg once daily for four weeks; 198 participants “test and treat” by serology, with those testing positive receiving eradication treatment with one week of omeprazole 20 mg twice daily, clarithromycin 250 mg twice daily, and metronidazole 400 mg twice daily; those testing negative received lansoprazole 30 mg once daily for four weeks; 178 participants empirical acid suppression using lansoprazole 30 mg once daily for four weeks |
| Myres (unpublished)† | Wales, primary care, 12 months | 61 participants ≥18-45 years, mean age 34 years, 33 (54.1%) female, 61 (100%) H pylori positive | 28 participants prompt endoscopy with medical treatment according to endoscopic findings; 33 participants “test and treat” by serology, with those testing positive receiving eradication treatment according to the primary care physician’s preferred strategy for treatment of H pylori |
Participants recruited in secondary care at first referral from primary care.
Data available in Ford 2005.19
Effectiveness
Intention to treat analysis
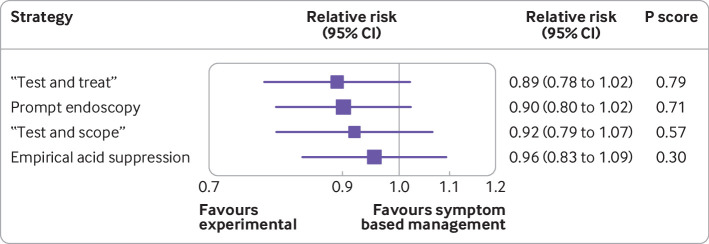
All 15 randomised controlled trials provided dichotomous data for likelihood of remaining symptomatic at the final point of follow-up.13 14 15 16 17 18 19 45 48 49 50 51 52 53 54 In these 15 trials, 1942 participants were randomised to prompt endoscopy, 484 to “test and scope,” 1938 to “test and treat,” 1329 to empirical acid suppression, and 469 to symptom based management. Figure 1 presents the network plot. When data were pooled, there was little observed heterogeneity (τ2=0.007), and no evidence of publication bias or other small study effects (supplementary fig 5). Of the five strategies, “test and treat” was ranked first (relative risk of remaining symptomatic 0.89, 95% confidence interval 0.78 to 1.02, P score 0.79; fig 2). The network heat plot had no red “hotspots” of inconsistency (supplementary fig 6), and there was no evidence of inconsistency under the full design by treatment interaction model after applying the χ2 test of the Q statistic (1.91, P=0.93). The netsplit analysis did not identify any significant differences between the direct and indirect treatment effect estimates for any of the treatment comparisons (supplementary table 2). None of the strategies was significantly less effective than “test and treat,” or more effective than each other, on either direct or indirect comparison (fig 3). Prompt endoscopy was ranked second, but performed similarly to “test and treat” (relative risk of remaining symptomatic 0.90, 95% confidence interval 0.80 to 1.02, P score 0.71). This means that the probability of “test and treat” or prompt endoscopy being the most effective strategy when all five management strategies, including symptom based management, were compared with each other was 79% and 71%, respectively. In contrast, the probability of “test and scope,” empirical acid suppression, or symptom based management being the most effective strategy was 57%, 30%, and 12%, respectively.
Fig 1.
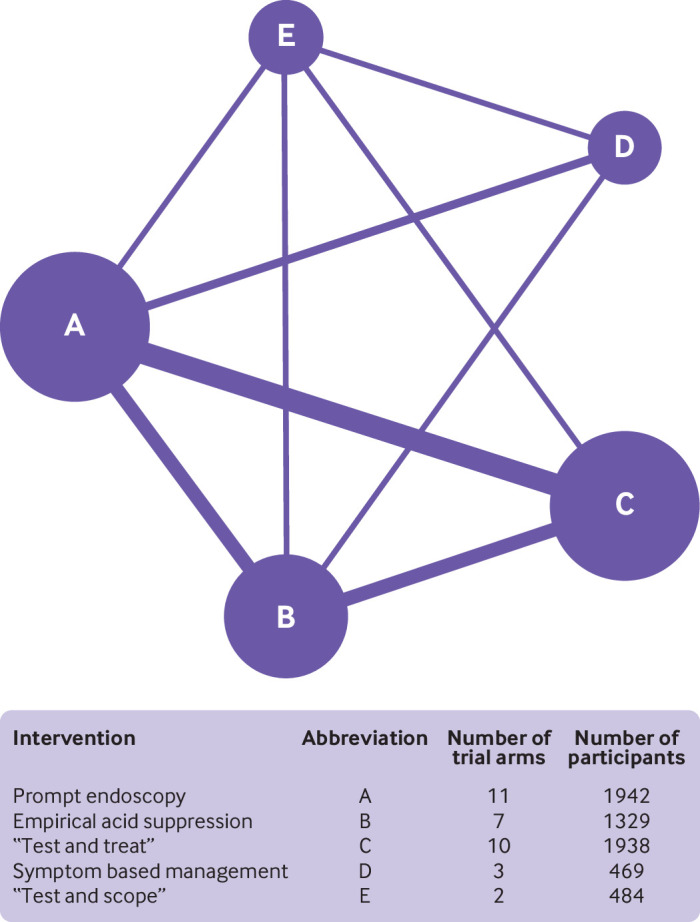
Network plot for likelihood of remaining symptomatic according to intention to treat analysis at final point of follow-up
Fig 2.
Forest plot for likelihood of remaining symptomatic according to intention to treat analysis at final point of follow-up. P score is probability of each treatment being ranked as best in network analysis. Higher score indicates greater probability of being ranked first
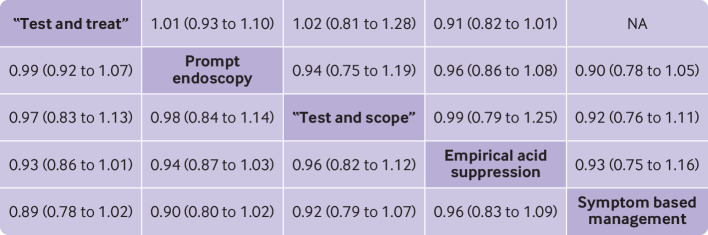
Fig 3.
Summary treatment effects from network meta-analysis for likelihood of remaining symptomatic according to intention to treat analysis at final point of follow-up. Comparisons, column versus row, should be read from left to right, and are ordered relative to overall effectiveness. Treatment in top left position is ranked as best after network meta-analysis of direct and indirect effects. Direct comparisons are provided above strategy labels, and indirect comparisons are below. Values are relative risk (95% confidence interval). NA=not applicable, no randomised controlled trials making direct comparisons
Two of the trials of “test and treat” versus prompt endoscopy recruited only participants with H pylori infection,19 48 and one of the trials of prompt endoscopy versus empirical acid suppression used ranitidine,13 rather than a proton pump inhibitor. Therefore, we excluded these three trials in a retrospective sensitivity analysis so as not to overestimate the effectiveness of “test and treat,” or underestimate the effectiveness of empirical acid suppression. When data were pooled, there was little observed heterogeneity (τ2=0.007). “Test and treat” was ranked first (relative risk 0.89, 95% confidence interval 0.77 to 1.02, P score 0.82) and prompt endoscopy second (0.90, 0.79 to 1.02, P score 0.70). When we excluded the two trials of 18 months’ duration, the overall results were not affected15 16; “test and treat” was still ranked first and prompt endoscopy second.
Per protocol analysis
All 15 randomised controlled trials provided dichotomous data for likelihood of remaining symptomatic at the final point of follow-up according to a per protocol analysis.13 14 15 16 17 18 19 45 48 49 50 51 52 53 54 In this analysis, there were data on 5154 participants, of whom 1667 were randomised to prompt endoscopy, 326 to “test and scope,” 1689 to “test and treat,” 1150 to empirical acid suppression, and 322 to symptom based management. Supplementary fig 7 presents the network plot. Again, when data were pooled, there was little observed heterogeneity (τ2=0.009), and no evidence of publication bias or other small study effects (supplementary fig 8). There were no red “hotspots” of inconsistency on the network heat plot (supplementary fig 9), with no evidence of inconsistency under the full design by treatment interaction model after applying the χ2 test of the Q statistic (1.28, P=0.97). The netsplit analysis did not identify any significant differences between the direct and indirect treatment effect estimates for any of the treatment comparisons (supplementary table 3). Once again, “test and treat” was ranked first (relative risk 0.87, 95% confidence interval 0.74 to 1.03, P score 0.79; supplementary fig 10), but was not superior to any of the other four strategies, and none of the strategies was more effective than any of the others on direct or indirect comparison (supplementary table 4). The P scores for prompt endoscopy, “test and scope,” empirical acid suppression, or symptom based management were 0.69, 0.63, 0.26, and 0.13, respectively. As before, when we excluded the three aforementioned trials in a retrospective sensitivity analysis,13 19 48 there was little observed heterogeneity (τ2=0.007), and “test and treat” was ranked first (relative risk 0.87, 95% confidence interval 0.73 to 1.02, P score 0.81), with prompt endoscopy second (0.88, 0.76 to 1.03, P score 0.68). Again, excluding the two trials of 18 months’ duration did not affect the overall results15 16; “test and treat” was still ranked first and prompt endoscopy second.
Rates of endoscopy
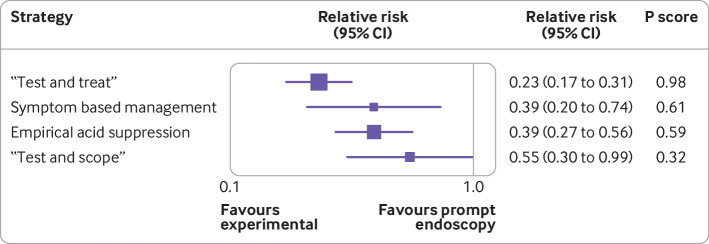
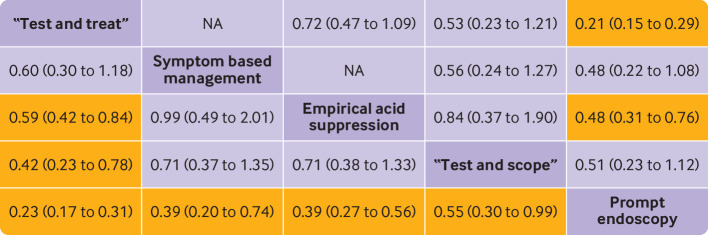
Fourteen randomised controlled trials that comprised 5897 participants, provided data on the number of participants in each arm undergoing endoscopy.13 14 15 16 17 18 19 45 48 49 50 51 52 53 Supplementary fig 11 presents the network plot. When data were pooled, there was a moderate level of statistical heterogeneity (τ2=0.16), but no evidence of publication bias or other small study effects (supplementary fig 12). The network heat plot had no red “hotspots” of inconsistency (supplementary fig 13), and there was no evidence of inconsistency under the full design by treatment interaction model after we applied the χ2 test of the Q statistic (2.56, P=0.63). The netsplit analysis did not identify any significant differences between the direct and indirect treatment effect estimates for any of the treatment comparisons (supplementary table 5). Of the five strategies, “test and treat” was ranked first (relative risk of receiving endoscopy 0.23, 95% confidence interval 0.17 to 0.31, P score 0.98; fig 4). When we performed an indirect comparison we found participants allocated to “test and treat” were significantly less likely to receive endoscopy than those in any of the other management strategies, except symptom based management. Participants assigned to all four other strategies were significantly less likely to receive endoscopy than those randomised to prompt endoscopy (fig 5). When we performed a direct comparison, we found participants randomised to “test and treat” or empirical acid suppression were significantly less likely to receive endoscopy than those assigned to prompt endoscopy.
Fig 4.
Forest plot for likelihood of receiving endoscopy. P score is probability of each treatment being ranked as best in network analysis. Higher score indicates greater probability of being ranked first
Fig 5.
Summary treatment effects from network meta-analysis for likelihood of receiving endoscopy. Comparisons, column versus row, should be read from left to right, and are ordered relative to overall effectiveness. Treatment in top left position is ranked as best after network meta-analysis of direct and indirect effects. Orange boxes indicate significant differences. Direct comparisons are provided above strategy labels, and indirect comparisons are below. Values are relative risk (95% confidence interval). NA=not applicable, no randomised controlled trials making direct comparisons
Participant dissatisfaction with management
Only six trials that comprised 2818 participants reported rates of satisfaction with management according to strategy13 18 45 49 50 51; no randomised controlled trials reported on satisfaction with symptom based management. Supplementary fig 14 presents the network plot. The term “risk” has negative connotations; therefore, in this analysis, we chose to extract data as rates of dissatisfaction with management, such that the best performing strategy has the lowest risk of dissatisfaction (rather than the highest risk of being satisfied). When data were pooled, there was a moderate level of statistical heterogeneity (τ2=0.13), and too few randomised controlled trials to assess for evidence of publication bias, or other small study effects. Of the four strategies, prompt endoscopy was ranked first (relative risk of being dissatisfied 0.58, 95% confidence interval 0.37 to 0.91, P score 0.95; supplementary fig 15). Participants allocated to prompt endoscopy were significantly less likely to be dissatisfied with management compared with participants randomised to “test and treat” or empirical acid suppression, on indirect comparison, and with empirical acid suppression on direct comparison (supplementary table 6). The netsplit analysis did not identify any significant differences between the direct and indirect treatment effect estimates for any of the treatment comparisons (supplementary table 7). However, the network heat plot revealed a red “hotspot” of potential inconsistency (supplementary fig 16), with evidence of inconsistency under the full design by treatment interaction model after applying the χ2 test of the Q statistic (26.07, P<0.001). This was driven by one early study of prompt endoscopy versus empirical acid suppression,13 which showed substantially higher rates of dissatisfaction with empirical acid suppression. Rerunning the network without this trial resolved the inconsistency (Q statistic 1.73, P=0.42) and reduced heterogeneity (τ2=0.002), but did not change the ranking of prompt endoscopy (relative risk 0.85, 95% confidence interval 0.70 to 1.02, P score 0.97).
Rates of upper gastrointestinal cancer detection
Eleven randomised controlled trials reported upper gastrointestinal cancer detection rates among 5028 participants.13 14 15 16 17 18 48 49 50 51 52 In total, 20 (0.40%) cancers were detected: 11 (0.67%) among 1644 participants undergoing prompt endoscopy; four (0.24%) among 1672 participants allocated to “test and treat”; two (0.41%) among 484 participants assigned to “test and scope”; two (0.24%) among 849 participants randomised to empirical acid suppression; and one (0.26%) among 379 participants given symptom based management. Cancer location and type were provided for 16 participants; gastric adenocarcinoma occurred in 12 people, gastric lymphoma in two people, and oesophageal carcinoma in two people. Both participants with oesophageal carcinoma had previously reported dysphagia, and arguably, were recruited inappropriately to the relevant trial.18
Discussion
Principal findings
This systematic review and network meta-analysis has shown that “test and treat” might be the most effective first line strategy for the management of uninvestigated dyspepsia in primary care, in terms of effect on symptoms, although prompt endoscopy performed similarly in this respect. However, no strategy was significantly less effective than “test and treat,” or more effective than another strategy, on either direct or indirect comparison. “Test and treat” was significantly more likely to reduce use of endoscopy compared with all strategies, other than symptom based management. Despite this, rates of dissatisfaction with management were significantly lower among participants allocated to prompt endoscopy compared with “test and treat” or empirical acid suppression on indirect comparison. Finally, detection rates of upper gastrointestinal cancer in these trials were extremely low.
Strengths and limitations of the study
The network meta-analysis allowed us to make indirect comparisons among over 6000 participants in 15 randomised controlled trials. The trials themselves were pragmatic and recruited participants from primary care, or on first referral to secondary care, which meant the results of our study are likely to be generalisable to other patients who present with dyspepsia in this setting. We used the most stringent endpoint for effect on symptoms in all trials, and only classified participants who were completely asymptomatic as having reached the endpoint of interest. We used an intention to treat analysis, with all trial dropouts assumed to be symptomatic. Because of the length of follow-up in individual randomised controlled trials, we performed a sensitivity analysis by using a per protocol analysis. We also excluded two randomised controlled trials that examined effectiveness of prompt endoscopy versus “test and treat” only in participants who were H pylori positive19 48 and one that used empirical ranitidine as an acid suppressant13 in a separate retrospective sensitivity analysis. Our findings remained unchanged in this analysis. Finally, we produced network heat plots and identified inconsistency in one of our analyses, but this was resolved when we excluded one study that reported a large difference in dissatisfaction rates between prompt endoscopy and empirical acid suppression.
Our study had several limitations. We did not have access to individual patient data for the network meta-analysis, which meant that we were unable to study the effects of the various management strategies on other dyspepsia related resource use, or total costs of managing dyspepsia. There were also differences between individual trials in the population studied, study setting, the way the intervention was applied, duration of follow-up, and endpoint used to define symptom response; therefore, it might not be appropriate to combine data from these trials in a meta-analysis. However, we only classed those as entirely asymptomatic as having reached the endpoint of interest, and we performed sensitivity analyses based on some of these study characteristics, and our results were unchanged. These differences could explain the moderate amounts of heterogeneity in some of our analyses.
Additionally, only six randomised controlled trials contributed data to the analysis of dissatisfaction with management,13 18 45 49 50 51 and four of these compared prompt endoscopy with “test and treat,”18 49 50 51 which meant that the findings from this analysis might not be as robust for the other strategies. We were not able to examine the effect of these management strategies on quality of life in the network meta-analysis because the included studies used a variety of instruments, disease specific and generic; they also reported these analyses in a multitude of ways, which precluded pooling of the data. Because most studies were conducted in western populations, with only one randomised controlled trial conducted in Malaysia,51 our findings cannot be extrapolated to the Far East where the incidence of gastric cancer is higher, and prompt endoscopy might therefore be more appropriate. Finally, all of the included randomised controlled trials were at high risk of bias because of their pragmatic design, which meant that blinding of participants was not possible.
Comparison with other studies
We believe this network meta-analysis is an advance over previous meta-analyses in this field for several reasons. Our meta-analysis produces a credible ranking system for each strategy, rather than relying on summary relative risks of comparative effectiveness of one strategy over another. This approach is clinically useful given that in previous individual patient data meta-analyses there was only a small, although statistically significant, difference in the relative risk of remaining symptomatic, which favoured prompt endoscopy over “test and treat.”19 Additionally, no difference was found between “test and treat” and empirical acid suppression.22 Furthermore, more randomised controlled trials have been included in our analysis than in the aforementioned individual patient data meta-analyses,19 22 and a previous trial based meta-analysis.55 Our meta-analysis was also able to make indirect and direct comparisons by using trial data, which led to a change in the strategy that could be the most effective for first line management of uninvestigated dyspepsia. “Test and treat” was ranked above prompt endoscopy, although P scores were similar. Additionally, the equipoise between “test and treat” and empirical acid suppression was no longer seen, with a P score for “test and treat” of 0.79 compared with 0.30 for empirical acid suppression. Although National Institute for Health and Care Excellence guidance states that either “test and treat” or empirical acid suppression can be used first line for uninvestigated dyspepsia,21 the “test and treat” strategy was recommended first line for patients younger than 60 by the more recent American College of Gastroenterology and Canadian Association of Gastroenterology joint practice guideline on dyspepsia.20 The results of our network meta-analysis support the latter recommendation. Finally, a previous trial based meta-analysis combined participants in the empirical acid suppression and symptom based management arms.55 Because those receiving symptom based management did not receive standardised proton pump inhibitor dosing in these randomised controlled trials, we believe this is technically incorrect, and might have led to an underestimate of the effectiveness of empirical acid suppression.
Dyspepsia, however defined, is a frequent reason for consultation with primary care providers and gastroenterologists. Dyspeptic symptoms can cause substantial anxiety for patients who might fear that they have a serious underlying condition to account for their symptoms. This anxiety is despite the fact that upper gastrointestinal malignancy is identified at endoscopy in less than 1% of patients.56 Of note, the rate of upper gastrointestinal malignancy in this meta-analysis was only around 0.4%, which suggests that, among 1000 patients presenting to a primary care provider with uninvestigated dyspepsia, 996 would be cancer free on endoscopy. This makes the strategy of prompt endoscopy for the evaluation of uninvestigated dyspepsia highly questionable, at least in the absence of potentially important alarm features that, arguably, would merit endoscopy in any case. A previous study in the USA estimated that the cost of detecting one upper gastrointestinal cancer in patients aged 50 or older with dyspepsia without alarm features in primary care was over $80 000 (£62 000; €72 300).57 Prompt endoscopy might be justifiable on the basis of providing reassurance to patients about the absence of a sinister underlying cause of their symptoms, but studies suggest this effect is relatively short lived.58 Although “test and treat” was ranked first in terms of effectiveness, and significantly limited the use of endoscopy, we found lower levels of dissatisfaction with management among participants randomised to prompt endoscopy. Given the lack of blinding in all of the included studies, this could relate to patients’ previous expectations of management, which were not met if assigned to a non-invasive management strategy; patients might prefer endoscopy for what they consider to be a more thorough and appropriate evaluation of their symptoms. Furthermore, the impact of negative findings at endoscopy probably also influences patients’ satisfaction with this approach.
We must weigh these potential benefits of endoscopy against its substantial costs and the risk of adverse events, albeit small. Because endoscopy is the principal driver of overall costs in the management of dyspepsia, it cannot be supported based on cost effectiveness. This consideration is underlined by a previous individual patient data meta-analysis of prompt endoscopy versus “test and treat,”19 which estimated that prompt endoscopy was only cost effective if the willingness to pay per patient cured of their dyspepsia was $180 000. Endoscopy can identify H pylori infection, although it is not essential for that purpose because there are widely available, cheaper, non-invasive tests for active infection with excellent performance characteristics. However, the American Gastroenterological Association has recommended routine collection of gastric biopsies at endoscopy when performed in patients with dyspepsia to document the presence or absence of H pylori infection59; however, treatment of the infection leads to sustained symptom improvement in only a minority of patients.60
Conclusions and policy implications
The strategy of “test and treat” has proved popular in many countries. H pylori infection is usually asymptomatic, but it can lead to dyspepsia even in the absence of peptic ulcer disease.61 The non-invasive detection of H pylori infection with a reliable test, such as the urea breath test or faecal antigen test, should lead automatically to treatment for the infection. The “test and treat” strategy would detect most patients with dyspepsia and underlying peptic ulcer disease, although it would not identify them individually; they would certainly benefit from eradication of the infection. However, most patients with H pylori infection would not have peptic ulcer disease and many would fulfil diagnostic criteria for functional dyspepsia. Eradication of the infection would produce sustained improvement in only a minority of these patients, but it would remove a potentially serious cause of disease in the remainder. Population screening and treatment for H pylori appears to reduce future dyspepsia related costs in the West,62 and also reduces incidence of gastric cancer in high risk populations,63 so there are probably other benefits from more widespread use of “test and treat.” That said, many of the trials included in the network meta-analysis were conducted more than 15 years ago, and the prevalence of H pylori infection might have declined in Western populations during this time. A simulation model of the cost effectiveness of management strategies for uninvestigated dyspepsia in the USA suggested that “test and treat” was unlikely to remain cost effective below a prevalence of infection of 20%, although the confidence intervals were wide.64
Symptom based management was ranked the lowest of all the strategies when considering effectiveness. Management of dyspepsia with drug treatments is unsatisfactory and often lacks an adequate evidence base because the underlying causes of symptoms are poorly understood. This makes targeted drug interventions empirical at best. Patients with dyspepsia might be treated with a variety of drugs, depending on local availability and approval, physicians’ personal experience, and to some extent, on assessment of an individual patient’s symptom profile. Recent guidelines recommend the use of empirical proton pump inhibitor treatment for patients younger than 60 in whom “test and treat” is unsuccessful and in those without H pylori infection.20 Although acid suppression with a proton pump inhibitor might be effective for some patients,65 their long term efficacy is unclear and the optimal duration of treatment is not defined. In patients whose dyspeptic symptoms do not respond to a proton pump inhibitor, there is no value in continuing with this treatment. Furthermore, recent concerns about the long term safety of these drugs, although often based on weak evidence,66 could have altered perceptions of their appropriateness for the long term management of dyspepsia. Additional drug interventions that could be used for the management of dyspepsia include drugs with presumed prokinetic effects67 and neuromodulators, including tricyclic antidepressants.68 The role of prokinetic agents is limited because of their lack of availability in many countries. Neuromodulators have an important role in the management of dyspepsia and other functional gastrointestinal disorders.68 69 70 However, the decision to use any of these drugs, and the order in which they might be tried, is based on choices made by individual physicians and patients, and to some extent is influenced by the factors listed here. Therefore, it is perhaps unsurprising that this largely empirical strategy was the least effective.
In summary, dyspepsia continues to be a highly prevalent condition that can influence quality of life profoundly and accounts for major healthcare expenditures. Many different management strategies have been studied in individual randomised controlled trials. This network meta-analysis provides additional support for the so called “test and treat” approach in management. This strategy, recently recommended in national guidelines,20 was consistently associated with the lowest chance of remaining symptomatic and with the lowest use of endoscopy. Therefore, it is probably of benefit in reducing overall costs, at least in some healthcare delivery models. However, despite the low diagnostic yield of endoscopy in detecting upper gastrointestinal tract malignancy, it might be the strategy most preferred by patients. Management of patients with dyspepsia should continue to be based on best evidence, but should also take into account the nuances of the individual patient within the confines of the healthcare setting.
What is already known on this topic
Dyspepsia is a highly prevalent and costly condition
Many management approaches have been compared in pragmatic randomised controlled trials, and summarised in individual patient data meta-analyses, but there is equipoise between strategies
Guidelines disagree about which approach should be used for the initial management of uninvestigated dyspepsia
What this study adds
This network meta-analysis found “test and treat” was ranked first, although it performed similarly to prompt endoscopy and was not superior to any of the other strategies
“Test and treat” led to fewer endoscopies than all other strategies except symptom based management
Participants showed a preference for prompt endoscopy as a management strategy for their symptoms
Wider application of a “test and treat” strategy for dyspepsia at the primary care level, which is recommended in recent national guidelines, should be encouraged
Web extra.
Extra material supplied by authors
Web appendix: Supplementary material
Contributors: LHE, CJB, CWH, and ACF conceived and drafted the study. LHE, CJB, CWH, and ACF analysed and interpreted the data. ACF drafted the manuscript. LHE and CJB contributed equally to the manuscript and are joint first authors. All authors have approved the final draft of the manuscript. ACF is guarantor. ACF accepts full responsibility for the work and the conduct of the study, had access to the data, and controlled the decision to publish. The corresponding author attests that all listed authors meet authorship criteria and that no others meeting the criteria have been omitted.
Funding: No funding given.
Competing interests: All authors have completed the ICMJE uniform disclosure form at www.icmje.org/coi_disclosure.pdf and declare: no support from any organisation for the submitted work; no financial relationships with any organisations that might have an interest in the submitted work in the previous three years; no other relationships or activities that could appear to have influenced the submitted work.
Ethical approval: Ethical approval for this evidence synthesis was not required.
Data sharing: No additional data available.
The lead author (ACF) affirms that the manuscript is an honest, accurate, and transparent account of the study being reported; that no important aspects of the study have been omitted; and that any discrepancies from the study as originally planned (and, if relevant, registered) have been explained.
References
- 1. Ford AC, Marwaha A, Sood R, Moayyedi P. Global prevalence of, and risk factors for, uninvestigated dyspepsia: a meta-analysis. Gut 2015;64:1049-57. 10.1136/gutjnl-2014-307843 [DOI] [PubMed] [Google Scholar]
- 2. Ford AC, Forman D, Bailey AG, Axon AT, Moayyedi P. Effect of dyspepsia on survival: a longitudinal 10-year follow-up study. Am J Gastroenterol 2012;107:912-21. 10.1038/ajg.2012.69 [DOI] [PubMed] [Google Scholar]
- 3. Chang JY, Locke GR, 3rd, McNally MA, et al. Impact of functional gastrointestinal disorders on survival in the community. Am J Gastroenterol 2010;105:822-32. 10.1038/ajg.2010.40 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Ford AC, Forman D, Bailey AG, Axon AT, Moayyedi P. Initial poor quality of life and new onset of dyspepsia: results from a longitudinal 10-year follow-up study. Gut 2007;56:321-7. 10.1136/gut.2006.099846 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Agréus L, Svärdsudd K, Talley NJ, Jones MP, Tibblin G. Natural history of gastroesophageal reflux disease and functional abdominal disorders: a population-based study. Am J Gastroenterol 2001;96:2905-14. 10.1111/j.1572-0241.2001.04680.x [DOI] [PubMed] [Google Scholar]
- 6. Halder SLS, Locke GR, 3rd, Schleck CD, Zinsmeister AR, Melton LJ, 3rd, Talley NJ. Natural history of functional gastrointestinal disorders: a 12-year longitudinal population-based study. Gastroenterology 2007;133:799-807. 10.1053/j.gastro.2007.06.010 [DOI] [PubMed] [Google Scholar]
- 7. Ford AC, Forman D, Bailey AG, Axon AT, Moayyedi P. Fluctuation of gastrointestinal symptoms in the community: a 10-year longitudinal follow-up study. Aliment Pharmacol Ther 2008;28:1013-20. 10.1111/j.1365-2036.2008.03813.x [DOI] [PubMed] [Google Scholar]
- 8. Enck P, Dubois D, Marquis P. Quality of life in patients with upper gastrointestinal symptoms: results from the Domestic/International Gastroenterology Surveillance Study (DIGEST). Scand J Gastroenterol Suppl 1999;231(suppl 231):48-54. 10.1080/003655299750025264 [DOI] [PubMed] [Google Scholar]
- 9. Camilleri M, Dubois D, Coulie B, et al. Prevalence and socioeconomic impact of upper gastrointestinal disorders in the United States: results of the US Upper Gastrointestinal Study. Clin Gastroenterol Hepatol 2005;3:543-52. 10.1016/S1542-3565(05)00153-9 [DOI] [PubMed] [Google Scholar]
- 10. Brook RA, Kleinman NL, Choung RS, Melkonian AK, Smeeding JE, Talley NJ. Functional dyspepsia impacts absenteeism and direct and indirect costs. Clin Gastroenterol Hepatol 2010;8:498-503. 10.1016/j.cgh.2010.03.003 [DOI] [PubMed] [Google Scholar]
- 11. Lacy BE, Weiser KT, Kennedy AT, Crowell MD, Talley NJ. Functional dyspepsia: the economic impact to patients. Aliment Pharmacol Ther 2013;38:170-7. 10.1111/apt.12355 [DOI] [PubMed] [Google Scholar]
- 12. Ford AC, Forman D, Bailey AG, Cook MB, Axon AT, Moayyedi P. Who consults with dyspepsia? Results from a longitudinal 10-yr follow-up study. Am J Gastroenterol 2007;102:957-65. 10.1111/j.1572-0241.2007.01080.x [DOI] [PubMed] [Google Scholar]
- 13. Bytzer P, Hansen JM, Schaffalitzky de Muckadell OB. Empirical H2-blocker therapy or prompt endoscopy in management of dyspepsia. Lancet 1994;343:811-6. 10.1016/S0140-6736(94)92023-0 [DOI] [PubMed] [Google Scholar]
- 14. Delaney BC, Qume M, Moayyedi P, et al. Helicobacter pylori test and treat versus proton pump inhibitor in initial management of dyspepsia in primary care: multicentre randomised controlled trial (MRC-CUBE trial). BMJ 2008;336:651-4. 10.1136/bmj.39479.640486.AE [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Delaney BC, Wilson S, Roalfe A, et al. Cost effectiveness of initial endoscopy for dyspepsia in patients over age 50 years: a randomised controlled trial in primary care. Lancet 2000;356:1965-9. 10.1016/S0140-6736(00)03308-0 [DOI] [PubMed] [Google Scholar]
- 16. Delaney BC, Wilson S, Roalfe A, et al. Randomised controlled trial of Helicobacter pylori testing and endoscopy for dyspepsia in primary care. BMJ 2001;322:898-901. 10.1136/bmj.322.7291.898 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. McColl KEL, Murray LS, Gillen D, et al. Randomised trial of endoscopy with testing for Helicobacter pylori compared with non-invasive H pylori testing alone in the management of dyspepsia. BMJ 2002;324:999-1002. 10.1136/bmj.324.7344.999 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Duggan AE, Elliott CA, Miller P, Hawkey CJ, Logan RF. Clinical trial: a randomized trial of early endoscopy, Helicobacter pylori testing and empirical therapy for the management of dyspepsia in primary care. Aliment Pharmacol Ther 2009;29:55-68. 10.1111/j.1365-2036.2008.03852.x [DOI] [PubMed] [Google Scholar]
- 19. Ford AC, Qume M, Moayyedi P, et al. Helicobacter pylori “test and treat” or endoscopy for managing dyspepsia: an individual patient data meta-analysis. Gastroenterology 2005;128:1838-44. 10.1053/j.gastro.2005.03.004 [DOI] [PubMed] [Google Scholar]
- 20. Moayyedi P, Lacy BE, Andrews CN, Enns RA, Howden CW, Vakil N. ACG and CAG clinical guideline: management of dyspepsia. Am J Gastroenterol 2017;112:988-1013. 10.1038/ajg.2017.154 [DOI] [PubMed] [Google Scholar]
- 21.Dyspepsia and gastro-oesophageal reflux disease: Investigation and management of dyspepsia, symptoms suggestive of gastro-oesophageal reflux disease, or both. https://www.nice.org.uk/guidance/cg184 2014. [PubMed]
- 22. Ford AC, Moayyedi P, Jarbol DE, Logan RF, Delaney BC. Meta-analysis: Helicobacter pylori ‘test and treat’ compared with empirical acid suppression for managing dyspepsia. Aliment Pharmacol Ther 2008;28:534-44. 10.1111/j.1365-2036.2008.03784.x [DOI] [PubMed] [Google Scholar]
- 23. Talley NJ, Lam SK, Goh KL, Fock KM. Management guidelines for uninvestigated and functional dyspepsia in the Asia-Pacific region: First Asian Pacific Working Party on Functional Dyspepsia. J Gastroenterol Hepatol 1998;13:335-53. 10.1111/j.1440-1746.1998.tb00644.x [DOI] [PubMed] [Google Scholar]
- 24.Boutron I, Page MJ, Higgins JPT, et al. Chapter 7: Considering bias and conflicts of interest among the included studies. In: Higgins JPT, Thomas J, Chandler J, et al (eds), Cochrane Handbook for Systematic Reviews of Interventions version 6.0 (updated July 2019). Cochrane, 2019. http://www.training.cochrane.org/handbook
- 25. Rücker G, Schwarzer G. Reduce dimension or reduce weights? Comparing two approaches to multi-arm studies in network meta-analysis. Stat Med 2014;33:4353-69. 10.1002/sim.6236 [DOI] [PubMed] [Google Scholar]
- 26. Hutton B, Salanti G, Caldwell DM, et al. The PRISMA extension statement for reporting of systematic reviews incorporating network meta-analyses of health care interventions: checklist and explanations. Ann Intern Med 2015;162:777-84. 10.7326/M14-2385 [DOI] [PubMed] [Google Scholar]
- 27. Salanti G, Higgins JP, Ades AE, Ioannidis JP. Evaluation of networks of randomized trials. Stat Methods Med Res 2008;17:279-301. 10.1177/0962280207080643 [DOI] [PubMed] [Google Scholar]
- 28. Salanti G. Indirect and mixed-treatment comparison, network, or multiple-treatments meta-analysis: many names, many benefits, many concerns for the next generation evidence synthesis tool. Res Synth Methods 2012;3:80-97. 10.1002/jrsm.1037 [DOI] [PubMed] [Google Scholar]
- 29. Salanti G, Ades AE, Ioannidis JP. Graphical methods and numerical summaries for presenting results from multiple-treatment meta-analysis: an overview and tutorial. J Clin Epidemiol 2011;64:163-71. 10.1016/j.jclinepi.2010.03.016 [DOI] [PubMed] [Google Scholar]
- 30. Chaimani A, Higgins JP, Mavridis D, Spyridonos P, Salanti G. Graphical tools for network meta-analysis in STATA. PLoS One 2013;8:e76654. 10.1371/journal.pone.0076654 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Higgins JP, Jackson D, Barrett JK, Lu G, Ades AE, White IR. Consistency and inconsistency in network meta-analysis: concepts and models for multi-arm studies. Res Synth Methods 2012;3:98-110. 10.1002/jrsm.1044 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Higgins JPT, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ 2003;327:557-60. 10.1136/bmj.327.7414.557 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Rücker G, Schwarzer G, Carpenter JR, Schumacher M. Undue reliance on I(2) in assessing heterogeneity may mislead. BMC Med Res Methodol 2008;8:79. 10.1186/1471-2288-8-79 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. da Costa BR, Juni P. Systematic reviews and meta-analyses of randomized trials: principles and pitfalls. Eur Heart J 2014;35:3336-45. 10.1093/eurheartj/ehu424 [DOI] [PubMed] [Google Scholar]
- 35. Krahn U, Binder H, König J. A graphical tool for locating inconsistency in network meta-analyses. BMC Med Res Methodol 2013;13:35. 10.1186/1471-2288-13-35 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Rücker G. Network meta-analysis, electrical networks and graph theory. Res Synth Methods 2012;3:312-24. 10.1002/jrsm.1058 [DOI] [PubMed] [Google Scholar]
- 37. Rücker G, Schwarzer G. Ranking treatments in frequentist network meta-analysis works without resampling methods. BMC Med Res Methodol 2015;15:58. 10.1186/s12874-015-0060-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Morton SC, Murad MH, O’Connor E, et al. AHRQ methods for effective health care. Quantitative synthesis-an update. Methods Guide for Effectiveness and Comparative Effectiveness Reviews. Rockville (MD). Agency for Healthcare Research and Quality, 2018. [Google Scholar]
- 39. Brown S, Hutton B, Clifford T, et al. A Microsoft-Excel-based tool for running and critically appraising network meta-analyses--an overview and application of NetMetaXL. Syst Rev 2014;3:110. 10.1186/2046-4053-3-110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Black CJ, Burr NE, Camilleri M, et al. Efficacy of pharmacological therapies in patients with IBS with diarrhoea or mixed stool pattern: systematic review and network meta-analysis. Gut 2019;•••:gutjnl-2018-318160. 10.1136/gutjnl-2018-318160. [DOI] [PubMed] [Google Scholar]
- 41. Black CJ, Burr NE, Quigley EMM, Moayyedi P, Houghton LA, Ford AC. Efficacy of secretagogues in patients with irritable bowel syndrome with constipation: Systematic review and network meta-analysis. Gastroenterology 2018;155:1753-63. 10.1053/j.gastro.2018.08.021 [DOI] [PubMed] [Google Scholar]
- 42. Luthra P, Burr NE, Brenner DM, Ford AC. Efficacy of pharmacological therapies for the treatment of opioid-induced constipation: systematic review and network meta-analysis. Gut 2018;68:434-44. 10.1136/gutjnl-2018-316001 [DOI] [PubMed] [Google Scholar]
- 43. Luthra P, Camilleri M, Burr NE, Quigley EMM, Black CJ, Ford AC. Efficacy of drugs in chronic idiopathic constipation: a systematic review and network meta-analysis. Lancet Gastroenterol Hepatol 2019;4:831-44. 10.1016/S2468-1253(19)30246-8. [DOI] [PubMed] [Google Scholar]
- 44. Black CJ, Burr NE, Ford AC. Relative efficacy of tegaserod in a systematic review and network meta-analysis of licensed therapies for irritable bowel syndrome with constipation. Clin Gastroenterol Hepatol 2019;S1542-3565(19)30740-2. 10.1016/j.cgh.2019.07.007. [DOI] [PubMed] [Google Scholar]
- 45. Jarbol DE, Kragstrup J, Stovring H, Havelund T, Schaffalitzky de Muckadell OB. Proton pump inhibitor or testing for Helicobacter pylori as the first step for patients presenting with dyspepsia? A cluster-randomized trial. Am J Gastroenterol 2006;101:1200-8. 10.1038/ajg2006227 [DOI] [PubMed] [Google Scholar]
- 46. Rao JNK, Scott AJ. A simple method for the analysis of clustered binary data. Biometrics 1992;48:577-85. 10.2307/2532311 [DOI] [PubMed] [Google Scholar]
- 47. Whiting-O’Keefe QE, Henke C, Simborg DW. Choosing the correct unit of analysis in Medical Care experiments. Med Care 1984;22:1101-14. 10.1097/00005650-198412000-00005 [DOI] [PubMed] [Google Scholar]
- 48. Heaney A, Collins JSA, Watson RGP, McFarland RJ, Bamford KB, Tham TC. A prospective randomised trial of a “test and treat” policy versus endoscopy based management in young Helicobacter pylori positive patients with ulcer-like dyspepsia, referred to a hospital clinic. Gut 1999;45:186-90. 10.1136/gut.45.2.186 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Lassen AT, Pedersen FM, Bytzer P, Schaffalitzky de Muckadell OB. Helicobacter pylori test-and-eradicate versus prompt endoscopy for management of dyspeptic patients: a randomised trial. Lancet 2000;356:455-60. 10.1016/S0140-6736(00)02553-8 [DOI] [PubMed] [Google Scholar]
- 50. Arents NLA, Thijs JC, van Zwet AA, et al. Approach to treatment of dyspepsia in primary care: a randomized trial comparing “test-and-treat” with prompt endoscopy. Arch Intern Med 2003;163:1606-12. 10.1001/archinte.163.13.1606 [DOI] [PubMed] [Google Scholar]
- 51. Mahadeva S, Chia YC, Vinothini A, Mohazmi M, Goh KL. Cost-effectiveness of and satisfaction with a Helicobacter pylori “test and treat” strategy compared with prompt endoscopy in young Asians with dyspepsia. Gut 2008;57:1214-20. 10.1136/gut.2007.147728 [DOI] [PubMed] [Google Scholar]
- 52. Manes G, Menchise A, de Nucci C, Balzano A. Empirical prescribing for dyspepsia: randomised controlled trial of test and treat versus omeprazole treatment. BMJ 2003;326:1118. 10.1136/bmj.326.7399.1118 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Kjeldsen HC, Bech M, Christensen B. Cost-effectiveness analysis of two management strategies for dyspepsia. Int J Technol Assess Health Care 2007;23:376-84. 10.1017/S0266462307070420 [DOI] [PubMed] [Google Scholar]
- 54. Lewin van den Broek NT, Numans ME, Buskens E, Verheij TJ, de Wit NJ, Smout AJ. A randomised controlled trial of four management strategies for dyspepsia: relationships between symptom subgroups and strategy outcome. Br J Gen Pract 2001;51:619-24. [PMC free article] [PubMed] [Google Scholar]
- 55. Delaney BC, Ford AC, Forman D, et al. Initial management strategies for dyspepsia. Cochrane Library, Issue 2. John Wiley & Sons, 2005. [Google Scholar]
- 56. Ford AC, Marwaha A, Lim A, Moayyedi P. What is the prevalence of clinically significant endoscopic findings in subjects with dyspepsia? Systematic review and meta-analysis. Clin Gastroenterol Hepatol 2010;8:830-7, 837.e1-2. 10.1016/j.cgh.2010.05.031 [DOI] [PubMed] [Google Scholar]
- 57. Vakil N, Talley N, van Zanten SV, et al. STARS I Study Group Cost of detecting malignant lesions by endoscopy in 2741 primary care dyspeptic patients without alarm symptoms. Clin Gastroenterol Hepatol 2009;7:756-61. 10.1016/j.cgh.2009.03.031 [DOI] [PubMed] [Google Scholar]
- 58. Lucock MP, Morley S, White C, Peake MD. Responses of consecutive patients to reassurance after gastroscopy: results of self administered questionnaire survey. BMJ 1997;315:572-5. 10.1136/bmj.315.7108.572 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Yang YX, Brill J, Krishnan P, Leontiadis G, American Gastroenterological Association Clinical Practice Guidelines Committee American Gastroenterological Association Institute guideline on the role of upper gastrointestinal biopsy to evaluate dyspepsia in the adult patient in the absence of visible mucosal lesions. Gastroenterology 2015;149:1082-7. 10.1053/j.gastro.2015.07.039 [DOI] [PubMed] [Google Scholar]
- 60. Moayyedi P, Soo S, Deeks J, et al. Dyspepsia Review Group Systematic review and economic evaluation of Helicobacter pylori eradication treatment for non-ulcer dyspepsia. BMJ 2000;321:659-64. 10.1136/bmj.321.7262.659 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Sugano K, Tack J, Kuipers EJ, et al. faculty members of Kyoto Global Consensus Conference Kyoto global consensus report on Helicobacter pylori gastritis. Gut 2015;64:1353-67. 10.1136/gutjnl-2015-309252 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Ford AC, Forman D, Bailey AG, Axon AT, Moayyedi P. A community screening program for Helicobacter pylori saves money: 10-year follow-up of a randomized controlled trial. Gastroenterology 2005;129:1910-7. 10.1053/j.gastro.2005.09.016 [DOI] [PubMed] [Google Scholar]
- 63. Ford AC, Forman D, Hunt RH, Yuan Y, Moayyedi P. Helicobacter pylori eradication therapy to prevent gastric cancer in healthy asymptomatic infected individuals: systematic review and meta-analysis of randomised controlled trials. BMJ 2014;348:g3174. 10.1136/bmj.g3174 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Barton PM, Moayyedi P, Talley NJ, Vakil NB, Delaney BC. A second-order simulation model of the cost-effectiveness of managing dyspepsia in the United States. Med Decis Making 2008;28:44-55. 10.1177/0272989X07309644 [DOI] [PubMed] [Google Scholar]
- 65. Moayyedi P, Delaney BC, Vakil N, Forman D, Talley NJ. The efficacy of proton pump inhibitors in nonulcer dyspepsia: a systematic review and economic analysis. Gastroenterology 2004;127:1329-37. 10.1053/j.gastro.2004.08.026 [DOI] [PubMed] [Google Scholar]
- 66. Vaezi MF, Yang YX, Howden CW. Complications of proton pump inhibitor therapy. Gastroenterology 2017;153:35-48. 10.1053/j.gastro.2017.04.047 [DOI] [PubMed] [Google Scholar]
- 67. Pittayanon R, Yuan Y, Bollegala NP, et al. Prokinetics for functional dyspepsia: a systematic review and meta-analysis of randomized control trials. Am J Gastroenterol 2019;114:233-43. 10.1038/s41395-018-0258-6 [DOI] [PubMed] [Google Scholar]
- 68. Ford AC, Luthra P, Tack J, Boeckxstaens GE, Moayyedi P, Talley NJ. Efficacy of psychotropic drugs in functional dyspepsia: systematic review and meta-analysis. Gut 2017;66:411-20. 10.1136/gutjnl-2015-310721 [DOI] [PubMed] [Google Scholar]
- 69. Ford AC, Lacy BE, Harris LA, Quigley EMM, Moayyedi P. Effect of antidepressants and psychological therapies in irritable bowel syndrome: an updated systematic review and meta-analysis. Am J Gastroenterol 2019;114:21-39. 10.1038/s41395-018-0222-5 [DOI] [PubMed] [Google Scholar]
- 70. Drossman DA, Tack J, Ford AC, Szigethy E, Törnblom H, Van Oudenhove L. Neuromodulators for functional gastrointestinal disorders (disorders of gut-brain interaction): a Rome Foundation working team report. Gastroenterology 2018;154:1140-1171.e1. 10.1053/j.gastro.2017.11.279 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Web appendix: Supplementary material