ABSTRACT
Introduction: Vaccination remains very effective in stimulating protective immune responses against infections. An important task in antibody and vaccine preparation is to choose an optimal carrier that will ensure a high immune response. Particularly promising in this regard are nanoscale particle carriers. An antigen that is adsorbed or encapsulated by nanoparticles can be used as an adjuvant to optimize the immune response during vaccination. a very popular antigen carrier used for immunization and vaccination is gold nanoparticles, with are being used to make new vaccines against viral, bacterial, and parasitic infections.
Areas covered: This review summarizes what is currently known about the use of gold nanoparticles as an antigen carrier and adjuvant to prepare antibodies in vivo and design vaccines against viral, bacterial, and parasitic infections. The basic principles, recent advances, and current problems in the use of gold nanoparticles are discussed.
Expert opinion: Gold nanoparticles can be used as adjuvants to increase the effectiveness of vaccines by stimulating antigen-presenting cells and ensuring controlled antigen release. Studying the characteristics of the immune response obtained from the use of gold nanoparticles as a carrier and an adjuvant will permit the particles’ potential for vaccine design to be increased.
KEYWORDS: Vaccine, gold nanoparticles, nanocarriers, adjuvant, immunization, antibodies, infections
1. Introduction
Vaccination remains very effective in stimulating protective immune responses against infections. Among the most advanced diagnostic tools is the immunochemical method, based on the use of highly pathogen-specific antibodies. An important task in antibody and vaccine preparation is to choose an optimal carrier (delivery system) that would ensure a high immune response. Such adjuvant carriers can deposit antigens at the injection sites, enhance their presentation to immunocompetent cells, and induce the production of necessary cytokines. Particularly promising in this regard are various nanoscale particle carriers [1–16]. Such nanocarriers can be used as adjuvants in the preparation of antibodies and next-generation vaccines. The term ‘nanovaccinology’ [17] is coming into common scientific use.
Of note, the stimulation of antibody production in response to antigens adsorbed on colloidal particles was discovered as early as the beginning of the twentieth century [18–20]. In the same period, studies began on the effect of colloidal metal particles on the cells of the immune system [21,22]. In recent decades, significant progress has been made in understanding the immunogenicity of nanoparticles, the response of the immune cells, the nature of immunomodulation and immunosuppression, and the cyto- and immunotoxicity of nanoparticles, with account taken of their physical–chemical properties [23–38].
Cells of the immune system constitute the first barrier to the entry of nanoparticles into animal tissues and cells. In this context, of undoubted interest is the study of the interaction of nanoparticles with phagocytic cells, the mechanisms of intracellular penetration, and the response of immune cells to nanoparticles [39]. Depending on their size, nanoparticles penetrate macrophages through receptor-mediated endocytosis (phagocytosis) or pinocytosis and are localized mainly in lysosomes and in perinuclear space. Often, they are wrapped inside fringed vesicles. Some researchers believe that a key part in nanoparticle uptake by macrophages is played by scavenger receptors [40,41]. Nanoparticle penetration into macrophages may enhance the respiratory activity of the cells and, in some cases, the release of inflammatory mediators (cytokines, prostaglandins, etc.) and the activation of the immune response genes [42,43].
Of great interest are studies on the penetration of nanoparticles not only into macrophages but also into other immune system cells, in particular dendritic cells. Dendritic cells treated with gold nanoparticles can affect the activation of CD8+ T cells, which leads to epitope-specific cytotoxic T-lymphocyte responses in vitro [44]. Compared with the use of the native antigen, the internalization of antigen-conjugated nanoparticles into dendritic cells increases the immune response, enhancing lymphocyte proliferation [45]. In addition, nanoparticles can activate other immune system cells – neutrophils [46,47], lymphocytes [48,49], and monocytes [50,51].
Thus, nanoparticle penetration into the immune cells, which causes cytokine induction, stimulation of T cells, activation of the immune response genes, enhanced antigen processing, and antibody secretion by B cells, offers a good possibility of using nanoparticles as carriers and adjuvants in the preparation of antibodies and vaccines against infections [52–55]. In particular, various nanoparticles are being used to make new vaccines against viral [56–60], bacterial [61], and parasitic [62] infections. On the basis of nanocarriers, subunit [63] and peptide [64] vaccines are being developed for the oral [65], intranasal [66], and percutaneous [67] routes of administration.
A very popular antigen carrier used for immunization and vaccination is gold nanoparticles (GNPs) [68–71]. Owing to their unique physicochemical properties, ease of preparation, and low toxicity, GNPs are widely used in various fields of biomedical research [72]. For the first time, GNPs were used as carriers of haptens for antibody preparation in 1986 [73]. Since then, a large number of reports have been published in which this method was used and improved to develop antibodies to a number of haptens and complete antigens [70]. It was found that adjuvant properties are inherent in GNPs themselves [74–76].
This review summarizes what is now known about the use of GNPs to prepare antibodies and vaccines against viral, bacterial, and parasitic infections.
2. Gold nanoparticles in the design of antiviral vaccines
GNPs were first used to develop vaccines by Demenev et al. in 1996 [77], who examined the protective properties of an experimental tick-borne encephalitis vaccine. For tick-borne encephalitis virus and other flaviviruses, a slowly sedimenting virus-specific antigen was described that had complement-binding activity, was not associated with the virion fraction, and did not have infectivity or hemagglutinating activity. This antigen was called a soluble antigen [78]. The vaccine was prepared by conjugating GNPs (average diameter, 15 nm) to the soluble antigen. White laboratory mice were vaccinated intraperitoneally three times, each at 32 μg of antigen per injection. The protective properties of the experimental and commercial vaccines were compared on the basis of the average survival time and the protection coefficients after vaccination of mice challenged with a virus-containing suspension (100,000 and 10,000 LD50). The survival time and the therapeutic and protective efficacy obtained when the experimental vaccine was used were about 1.5 times greater than when the commercial vaccine was used.
Many studies on GNP immunization and vaccination have used such important objects as influenza virus and foot and mouth disease (FMD) virus. Highly specific antibodies were obtained from the immunization of animals with 17-nm GNPs coupled to the pFMDV and pH5N1 antigens of these viruses [79]. BALB/C white mice were vaccinated intraperitoneally. Both antisera obtained had a high titer. The same research team evaluated the effect of GNP size on the immune response to the synthetic FMD peptide pFMDV [80]. GNPs with diameters of 2, 5, 8, 12, 17, 37, and 50 nm were used. BALB/C mice were immunized intraperitoneally six times, each at 1 μg of antigen per injection. The antigens were mixed with complete Freund’s adjuvant (CFA). The antibody titer was maximal for 8-, 12- and 17-nm GNPs. The titer of the antisera obtained by using GNPs was threefold higher than when keyhole limpet hemocyanin (KLH) was used as an adjuvant and did not require further purification from contaminant antibodies (Figure 1). The amount of GNPs that had accumulated in the spleen correlated with the magnitude of the immune response elicited by the GNP/pFMDV conjugate.
Figure 1.
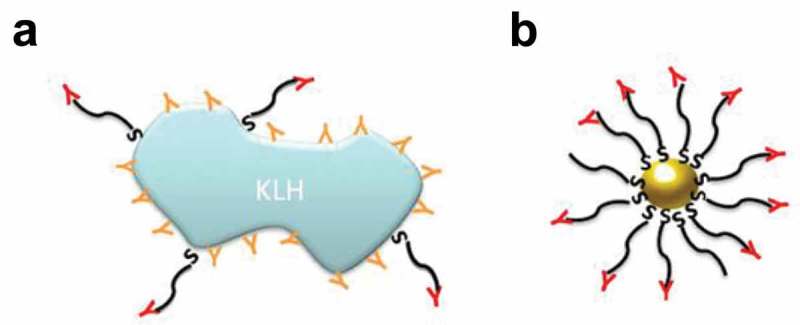
Efficacy of using keyhole limpet hemocyanin (KLH) and GNPs as nanocarriers. (a) Antibodies obtained by using KLH recognize the epitopes of both the antigen and the carrier protein. (b) Antibodies obtained by using GNPs recognize only the antigen’s epitopes [80].
Double intraperitoneal immunization of rats with 15-nm GNPs complexed with the M1 antigen of the influenza virus of the attenuated strain PR8 at a dose of 20 mg/kg produced high-titer antibodies [81]. The production of IFN-γ and interleukins (ILs) 1β and 6 was stimulated, and the respiration of peritoneal macrophages and spleen lymphocytes, was activated. Similar results were produced by double subcutaneous immunization of guinea pigs with 12 μg of 15-nm GNPs conjugated to a synthetic VP1 peptide of the FMD virus capsid [82]. The titer of the resultant antibodies was not lower than that obtained with CFA.
When guinea pigs were injected with FMD viruslike particles complexed with gold nanostars (50 μg, single injection), the titer of specific and neutralizing antibodies and the stimulation of T lymphocyte proliferation were much greater than those in guinea pigs immunized with the antigen in the presence of mineral oil ISA206 as an adjuvant. The proposed nanovaccine more effectively protected virus-infected animals [83].
A prototype of the intranasal influenza a vaccine was first proposed by Gill’s group [84]. The synthetic peptide M2e, a conserved extracellular domain of influenza virus matrix protein 2 conjugated to 12-nm GNPs, was used as an immunogen (Figure 2). As an adjuvant, soluble CpG (cytosine–phosphate–guanosine) oligodeoxynucleotide (CpG ODN) was used. BALB/C mice were immunized twice at an interval of 21 days, each time with 8.2 μg of M2e per animal. The conjugate induced the production of specific IgG (with stimulation of subtypes of both IgG1 and IgG2a), which could recognize M2e and native M2 on influenza a viruses and had a protective effect against a lethal dose of PR8-H1N1 virus. Subsequently, Gill’s group published more detailed results from the use of the proposed prototype vaccine to induce protective immunity against the influenza a virus serotypes H1N1, H3N2, and H5N1 [85,86]. Bimler et al. [87] showed that even at 15 months after being vaccinated with the GNP/M2e+CpG conjugate, the mice retained high titers of M2e-specific antibodies. These mice were protected against lethal H1N1 influenza. In addition, the antibody titers increased after a challenge with influenza a and remained elevated for 3 months. This suggests that old mice retain effective M2e-specific memory B cells.
Figure 2.
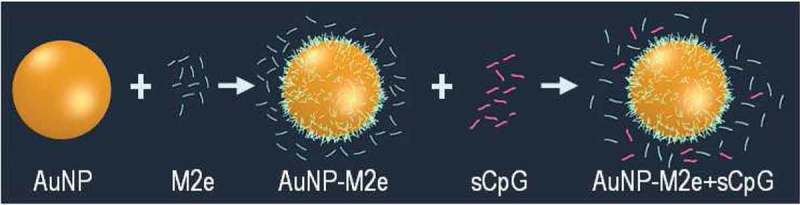
Scheme of vaccine design. M2e is conjugated to GNPs. By keeping M2e in excess in the solution, complete surface-coverage of GNPs with M2e is ensured at all times. Soluble CpG is added to the conjugate [86].
When mice were immunized with the M2e antigen of influenza a virus, the most effective complex immunogens were M2e + GNPs and CpG + GNPs [88]. When they were used, the titer of the obtained antibodies was the highest. In addition, the immunogens activated the respiratory activity of lymphoid cells and the production of proinflammatory cytokines to a greater extent than did other immunogens, including a commercial vaccine. The adjuvant effect of GNPs is probably associated with the more efficient penetration of the conjugate into phagocytic cells, which leads to improved presentation of the antigen to antibody-forming cells.
In general, GNPs as an antigen carrier and adjuvant are often combined with other immune response stimulants, most commonly with CFA and CpG ODNs. It is known that when CpG motifs are unmethylated, they act as immunostimulants [89]. The immunostimulatory activity of synthetic ODNs containing CpG sequences may be similar to the activity of bacterial DNA [90]. According to Wei et al. [91], GNPs coupled to CpG ODNs effectively enhance the intracellular penetration of nanoparticles into macrophages and significantly increase the secretion of proinflammatory cytokines – tumor necrosis factor α (TNF-α) and IL-6. Moreover, the immunostimulatory effect of GNPs conjugated to CpG ODNs is often significantly higher than that of the same concentrations of soluble CpG ODNs [92,93]. Of note, the optimal immunization method is the simultaneous use of two conjugates: GNPs–antigen and CpG ODNs–GNPs [94] (Figure 3). The immune response to an intranasal influenza vaccination can be enhanced with gold nanorods coupled to polyinosinic-polycytidylic acid as an adjuvant, as well as with CpG [95].
Figure 3.
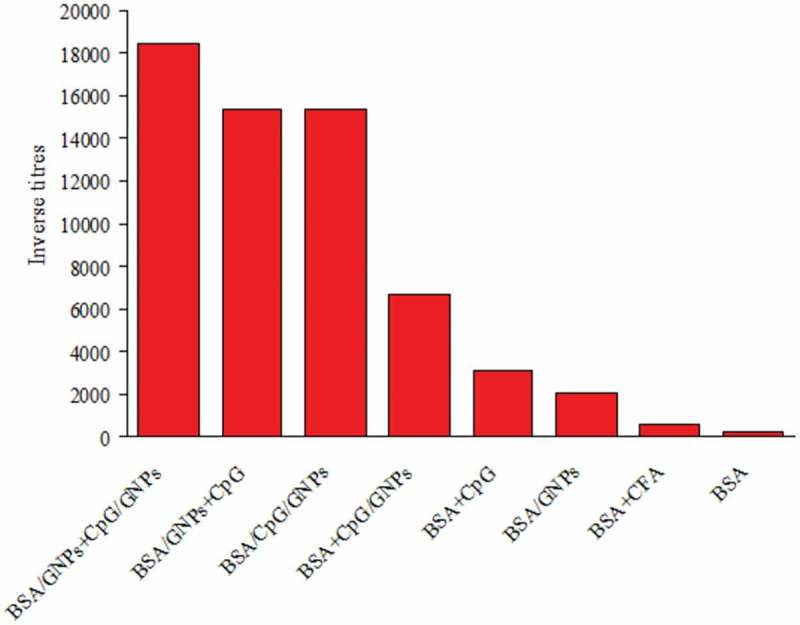
Average antibody titers obtained with various immunostimulant combinations [94].
Wang et al. [96,97] proposed the use of two H3N2 influenza a antigens for vaccination – hemagglutinin and flagellin – in complex with 18-nm GNPs. In a single intranasal immunization, both GNP/hemagglutinin/flagellin and GNP/hemagglutinin complexes induced higher levels of antibody production in mice than did a mixture of soluble hemagglutinin and flagellin. Immunization elicited a strong mucosal and systemic immune response, which protected the hosts from lethal influenza. In addition, it was shown in vitro that the conjugates promote the maturation of dendritic cells for the processing and presentation of antigens, stimulate the production of cytokines, and promote antigen-specific T-cell immunity.
GNPs were used to prepare antibodies to the surface antigens of porcine transmissible gastroenteritis virus [98,99]. Mice were immunized once intraperitoneally with 70 μg of antigen–15-nm GNPs; guinea pigs, twice subcutaneously with 125 μg; and rabbits, three times subcutaneously with 220 μg. The resulting virus-neutralizing antibodies from all animal species had a much higher titer than did antibodies developed against the native virus. Immunization with the GNP/antigen complex significantly enhanced the respiratory activity of peritoneal macrophages and spleen lymphocytes and increased the content of IFN-γ, IL-1β, and IL-6 in the blood plasma of the immunized animals. In addition, there was an increase in the number of follicles in the spleen, indicating the activation of humoral immunity.
A conjugate of 15-nm GNPs with a glycoprotein isolated from fixed rabies virus, strain Moscow 3253, was used to develop highly specific antibodies to the virus [100]. In the first immunization, 25 μg of the antigen was intraperitoneally injected into mice; in the subsequent four immunizations, 50 μg was used. The immunogenicity of the purified glycoprotein was higher than that of whole rabies virus, and the glycoprotein conjugated to GNPs was best able to induce virus-neutralizing antibodies. The resulting antibodies were used to make a diagnostic agent based on solid-phase immunoassay. Antirabies virus antibodies were also prepared by using the GNP-conjugated ribonucleoprotein of an attenuated rabies virus [101].
To prepare antibodies against West Nile fever virus, Niikura et al. [102] used antigen conjugates with GNPs of various sizes and shapes: 20- and 40-nm nanospheres, 40 × 20-nm nanorods, and 40 × 40 × 40-nm nanocubes. Mice were immunized twice intraperitoneally with 100 ng of the conjugate per animal. Forty-nm nanospheres induced the highest concentration of specific antibodies, while the titer of the antibodies obtained with the other GNPs was nearly 2 times lower. The macrophages and dendritic cells absorbed larger numbers of nanorods, which suggests that the production of antibodies does not depend on the efficiency of absorption of various GNPs. Nanorods enhanced the production of IL-1β and IL-18, while nanospheres and nanocubes enhanced that of TNF-α, IL-6, IL-12, and granulocyte–macrophage colony-stimulating factor.
Stone et al. [103] examined the immune response to nanorods coupled to the glycoprotein antigen of respiratory syncytial virus. The conjugate-treated human dendritic cells induced an immune response (proliferation) of primary T cells.
The influence of the particle shape and size on the effectiveness of the immune response was also investigated in [94]. Mice were injected with an antigen coupled to gold nanospheres (diameters, 15 and 50 nm), nanorods (70 × 15 nm), nanoshells (SiO2 core, 140 nm; gold shell, 27 nm), and nanostars (core, 12 nm; rays, 15–25 nm). The antibody titers increased from 1:160 to 1:10240 in the sequence native antigen < nanorods < nanoshells < GNPs-15 nm < GNPs-50 nm. It was concluded that spherical GNPs with diameters of 50 and 15 nm are the optimal antigen carrier and adjuvant for immunization.
The V3β peptide of the HIV-1 gp120 protein in complex with 2-nm gold glyconanoparticles was used as an immunogen to prepare a prototype HIV vaccine [104]. Rabbits were immunized three times intramuscularly with 50 μg of the complex. The resulting antibodies had a high titer and neutralizing activity against HIV-1. The HIV-1 Gag p17 peptide conjugated to 2-nm gold glyconanoparticles increased the proliferation of HIV-specific CD4+ and CD8+ T cells and induced the secretion of the highly functional TNF-α and IL-1β cytokines, as compared to the unconjugated peptide [105]. Also, GNPs conjugated with the gp120 and gp41 viral proteins were used as a platform for the delivery of immunogens in the preparation of an HIV-1 vaccine [106].
Ten-nm GNPs were complexed simultaneously with CpG ODN and with the recombinant hepatitis B virus core antigen in the form of viruslike particles [107]. Mice were immunized four times intraperitoneally with 50 μg of the conjugate. The titer of the resultant antibodies was twofold higher than that of the antibodies prepared without GNPs. The proliferation of the CD4+ and CD8+ T cells, the secretion of IL-4 and IFN-γ, and the stimulation of both Th1 and Th2 immune responses were increased. That is, the conjugates showed a strong cellular and humoral immunostimulatory ability.
The surface antigen of hepatitis B virus (HBsAg) was conjugated to gold nanocages [108]. The immunogenic properties of the conjugate were examined in vitro in RAW 264.7 macrophages. The conjugate was intensely absorbed by the macrophages and contributed to the processing of the antigens and to the secretion of IL-4.
For a prototype hepatitis E vaccine, vaccine monomers were conjugated to gold fluorescent nanoclusters [109]. The intraperitoneally injected conjugate not only enhanced the Th1/Th2 immune response in mice but also reduced the toxicity of the vaccine. In addition, the own fluorescence of the gold clusters made it possible to track the dynamic behavior of the vaccine in vivo. GNPs were also used to prepare antipeptide antibodies to the binding sites of the E2 protein of hepatitis C virus [110].
A prototype dengue virus vaccine was developed by using gold nanorods functionalized with protein E virus (DENVE) [111]. Immunization of mice with GNP/DENVE significant increased IgG synthesis and splenocyte proliferation.
Quach et al. [112] conjugated differently sized GNPs to domain III of envelope glycoprotein derived from serotype 2 of dengue virus (EDIII) as a dengue subunit vaccine. They used GNPs with average particle diameters of 20, 40, and 80 nm. The GNP/EDIII conjugate was used to immunize BALB/C mice three times subcutaneously. The conjugate induced high concentrations of antibodies that provide serotype-specific neutralization of dengue virus. The concentration of the antibodies depended on both size and concentration of the GNPs, and this made it possible to modulate the immune response by adjusting these parameters. In addition, the conjugate activated the complement system, induced proliferative responses in splenocytes and CD4+ and CD8+ T cells, and induced the secretion of IL-4 and IFN-γ. Eighty-nm GNPs performed the best.
Using the surface spike glycoprotein of avian coronavirus, which causes infectious bronchitis in birds, Chen et al. [113] obtained viruslike particles by incubating 100-nm GNPs in a solution containing an optimized concentration of the viral proteins. After free proteins were removed, the particles with the antigen were shown by TEM to be morphologically similar to the natural viral particles. BALB/C mice and specific-pathogen-free chickens were vaccinated intramuscularly with a single dose of 10 μg. Compared to free protein immunization, conjugate vaccination showed improved antigen delivery to lymphoid organs, higher antibody titers, an increased response of spleen T cells, and a reduction in symptoms of coronavirus infection. a comparison with a commercial whole inactivated viral vaccine also showed that the GNP conjugate provided better antiviral protection. GNPs conjugated to the S protein of severe acute respiratory syndrome-related coronavirus induced a strong IgG response upon immunization of mice [114].
GNPs were also used to make a conjugate with the antigen of the Newcastle disease virus of birds [115]. Ross chickens were vaccinated once subcutaneously with a mixture of GNPs and the vaccine antigen. High-titer specific antibodies were obtained, and increased secretion of IFN-γ was noted.
The capsid (Cap) protein of a pathogenic porcine circovirus was conjugated to 23-nm GNPs [116]. In vitro studies showed that GNPs contributed to Cap protein phagocytosis. Mice immunized twice subcutaneously with GNP/Cap showed high production of virus-neutralizing antibodies. Similar results were obtained with classical swine fever virus antigen [117].
Soliman et al. [118] investigated the use of gold nanospheres and nanorods as an adjuvant to develop antibodies against the Rift Valley fever virus. They noted that the immune response obtained by a single subcutaneous immunization of rats depended on the shape of the GNPs. Peak antibody concentrations were detected on day 30 after immunization with nanorods and on day 45 with nanospheres. However, the content of IFN-γ and IL-10 was higher with nanospheres than it was with nanorods.
Research is also underway to design GNP-based vaccines against other dangerous pathogens, such as Ebola, Zika, Marburg, Lassa, and Nipah.
Table 1 summarizes the literature data on the viral antigens that have been conjugated with GNP carriers and then used for immunization of animals.
Table 1.
Conjugates of GNPs with viral antigens used for immunization and vaccination of animals.
| Viral antigen | GNP size and shape | Functionalization approach (ligand) | Chemical nature of antigen | Refs. |
|---|---|---|---|---|
| Tick-borne encephalitis virus | Nanospheres, 15 nm | Adsorption | Isolated protein | [77] |
| Foot and mouth disease virus | Nanospheres, 17 nm | Chemosorption (cysteine) | Synthetic peptide | [79] |
| -”- | Nanospheres, 2, 5, 8, 12, 17, 37, and 50 nm | Chemosorption (cysteine) | Synthetic peptide | [80] |
| -”- | Nanospheres, 15 nm | Adsorption | Synthetic peptide | [82] |
| -”- | Nanostars | Adsorption | Virus-like particles | [83] |
| Influenza virus | Nanospheres, 17 nm | Chemosorption (cysteine) | Synthetic peptide | [79] |
| -”- | Nanospheres, 15 nm | Adsorption | Isolated peptide | [81] |
| -”- | Nanospheres, 12 nm | Adsorption | Synthetic peptide | [84] |
| -”- | Nanospheres, 12 nm | Chemosorption (cysteine) | Synthetic peptide | [87] |
| -”- | Nanospheres, 15 nm | Adsorption | Synthetic peptide | [88] |
| -”- | Nanospheres, 18 nm | Chemosorption (N3-PEG-SH, SH-NTA) | Recombinant proteins | [96] |
| Porcine transmissible gastroenteritis virus | Nanospheres, 15 nm | Adsorption | Isolated protein | [99] |
| Rabies virus | Nanospheres, 15 nm | Adsorption | Isolated glycoprotein | [100] |
| -”- | Nanospheres, 15 nm | Adsorption | Isolated ribonucleoprotein | [101] |
| West Nile fever virus | Nanospheres, 20 and 40 nm; Nanorods, 40 × 20 nm; Nanocubes, 40 nm | Chemosorption (PSS-MA) | Isolated protein | [102] |
| Respiratory syncytial virus | Nanorods, 21 × 57 nm | Chemosorption (EDC) | Recombinant glycoprotein | [103] |
| HIV | Glyconanoparticles, 2 nm | Adsorption | Synthetic peptide | [104] |
| Hepatitis B virus | Nanospheres, 10 nm | Adsorption | Recombinant protein | [107] |
| -”- | Nanocages, 50 nm | Adsorption | Recombinant protein | [108] |
| Hepatitis E virus | Nanoclusters | Direct synthesis | Hepatitis E vaccine | [109] |
| Hepatitis C virus | Nanospheres, 15 nm | Adsorption | Synthetic peptide | [110] |
| Dengue virus | Nanospheres, 20, 40, and 80 nm | Adsorption | Recombinant glycoprotein | [112] |
| Avian coronavirus | Nanospheres, 100 nm | Adsorption | Recombinant protein | [113] |
| Severe acute respiratory syndrome-related coronavirus | Nanospheres, 40 nm | Chemosorption (BSPP) | Recombinant protein | [114] |
| Newcastle disease virus | Nanospheres | Mixing | Newcastle disease vaccine | [115] |
| Porcine circovirus | Nanospheres, 23 nm | Adsorption | Recombinant protein | [116] |
| Classical swine fever virus | Nanospheres, 24 nm | Adsorption | Recombinant protein | [117] |
| Rift Valley fever virus | Nanospheres, 20 nm; Nanorods, 40 × 20 nm | Adsorption | Isolated protein | [118] |
3. Gold nanoparticles in the preparation of antibacterial vaccines
GNPs have also been intensely used to develop antibodies and vaccines against bacterial infections. For the first time, 15-nm GNPs were used as an adjuvant to prepare antibodies to the surface antigens of Yersinia pseudotuberculosis [119]. Rabbits received two subcutaneous injections, each with 1 μg of the antigen. The serum titer was as high as when CFA was used, but the amount of antigen injected when GNPs were used as an adjuvant was smaller by two orders of magnitude. GNPs as an antigen carrier activated the phagocytic activity of the lymphoid cells. Similar results were obtained by the same research team in the preparation of antibodies against Y. enterocolitica and Brucella abortus.
The efficacy of 15-nm GNPs coated with Y. pestis F1 antigen in animal immunization was evaluated by Gregory et al. [120]. The conjugate (0.93 μg) was administered to mice once intramuscularly. The animals injected with GNP/F1 generated the greatest IgG antibody response to the F1 antigen, as compared to those injected with unconjugated F1 antigen in phosphate-buffered saline or in Alhydrogel®. Sera obtained against the F1 antigen coupled to GNPs were able to bind to the F1 antigen in an enzyme-linked immunoassay. Similar results were produced by Kireev et al. [121].
GNPs decorated with thiolated glycosides (synthetic analogs of the capsular polysaccharides of Neisseria meningitidis) were proposed as a synthetic antigen for use in immunization [122]. Conjugates of 15-nm GNPs with Salmonella typhimurium surface antigens stimulated macrophage respiratory activity and the activity of the mitochondrial enzymes succinate dehydrogenase and glycerophosphate dehydrogenase [42]. This stimulation may be a significant factor determining the adjuvant properties of GNPs.
GNPs were included in the transmucosal delivery of tetanus toxoid, used for vaccination against tetanus, a deadly disease caused by Clostridium tetani [123]. Subcutaneous injection of tetanus toxoid conjugated to 25-nm GNPs into mice generated a systemic response but did not cause any mucosal response. However, the conjugate generated a significantly higher mucosal response after oral administration. For enhancing the immune response to tetanus toxoid, plant adjuvants (saponins) from Quillaja saponaria [124] and Asparagus racemosus [125] were used together with GNPs. GNPs were also used in the preparation of a vaccine against botulism, a particularly dangerous disease caused by C. botulinum [126].
GNPs (average diameter, ~ 2 nm) were conjugated to a synthetic tetrasaccharide epitope, an analog of the capsular polysaccharide of Streptococcus pneumoniae type 14 [127,128]. The obtained glyconanoparticles conjugated with T-helper peptide generated specific high-titer IgG after a single intradermal immunization of mice with 3 μg of the conjugate. An increase in the content of the cytokines IL-2, IL-4, IL-5, IL-17, and IFN-γ confirmed that the nanoparticles activate T helpers. The antisaccharide antibodies stimulated the phagocytosis of type 14 bacteria by human leukocytes, which indicates the functionality of the antibodies.
A new strategy for preparing antibodies to tuberculin was described [129] that used the adjuvant properties of GNPs. Tuberculin is a mixture of the surface antigens of various types of mycobacteria (Mycobacterium tuberculosis, M. bovis, and M. avium). When injected intradermally, it causes a specific delayed-type hypersensitivity response in infected or vaccinated patients. Polyclonal antituberculin antibodies were raised by injecting 7.5 μg of tuberculin conjugated to 15-nm GNPs into rabbits four times intramuscularly. The obtained antibodies had a high titer, whereas in the control animals, which were given nonconjugated tuberculin, no antibodies were found in the blood serum. The antituberculin antibodies were used to detect mycobacteria by ELISA and by light and electron microscopy.
GNPs were found to partially remove the toxic effect of tuberculin on rat peritoneal macrophages owing to their penetration into the intracellular space. This decrease in toxicity contributed to the better development of the humoral response and to the elaboration of antituberculin antibodies. In addition, GNPs decorated with tuberculin enhanced the adhesion of phagocytic cells to microbial cells. Preliminary vaccination results for GNPs conjugated to guinea pig tuberculin showed a protective effect [129].
For immunoassay of mycobacteria, GNPs were conjugated to the penta- and hexasaccharide fragments of mycobacterial lipoarabinomannan by using aglycon spacers of various lengths [130]. Rabbit polyclonal antibodies to these conjugates were developed. The antisera obtained by hyperimmunization of rabbits detected with a high titer both lipoarabinomannan oligosaccharides and mycobacterial cells.
Gao et al. [131] coated 30-nm GNPs with bacterial vesicles of the outer membrane of Escherichia coli (Figure 4). When injected three times subcutaneously, the conjugate induced rapid activation and maturation of dendritic cells in the lymph nodes of the vaccinated mice. In addition, vaccination with the conjugate gave rise to antibodies that had a higher avidity than those obtained by vaccination with outer membrane vesicles only. The production of IFN-γ and IL-17 increased too, which indicates strong Th1 and Th17 cellular responses to the bacteria.
Figure 4.
Schematic illustration of modulation of antibacterial immunity with bacterial-membrane-coated nanoparticles [130].
Another prototype vaccine against E. coli was demonstrated by Sanchez-Villamil et al. [132]. GNPs were coated with the specific immunogenic LomW and EscC antigens of the enterohemorrhagic strain E. coli O157: H7. Mice were injected three times subcutaneously with GNPs/LomW, GNPs/EscC, or a combination thereof, containing equivalent amounts of both immunogens. As a result, higher-titer IgG were found in sera and higher-titer IgA were found in feces. Serum IgG titers correlated with a significant decrease in the intestinal colonization of E. coli at 3 days after inoculation. The sera from the mice immunized with antigen-coated GNPs reduced the adhesion of E. coli O157: H7 and two other E. coli pathotypes to human intestinal epithelial cells. In addition, the sera had antigen-specific bactericidal properties.
An effective vaccine against the human pathogen Listeria monocytogenes was made by conjugating the listeriolysin O peptide LLO91-99 to gold glyconanoparticles [133]. a single intraperitoneal or intravenous immunization of mice with the conjugate induced a specific T-cell response and protected the animals against Listeria infection. The protection correlated with an increase in the number of splenic CD4+ and CD8+ T cells, NK cells, and CD8α+ dendritic cells, and it also correlated with an increase in the production of the cytokines IL-12, IFN-γ, TNF-α, and MCP-1 after infection. Subsequently, it was shown that vaccination of pregnant mice with gold glyconanoparticles carrying L. monocytogenes peptides protects newborns from listeriosis [134]. Newborn mice born to vaccinated females were free of bacteria and healthy, whereas nonvaccinated mice had obvious lesions of the brain and skin.
An interesting vaccination schedule against glanders, a disease caused by Burkholderia mallei, was proposed by Gregory et al. [135]. GNPs (average diameter, 15 nm) were first covalently bound to recombinant protein carriers – the Hc fragment of tetanus toxin, the hemolysin coregulated protein produced by both B. mallei and B. pseudomallei, and the flagellin produced by B. pseudomallei. The conjugates so prepared were functionalized with purified LPS from a nonvirulent B. thailandensis strain. Mice were immunized three times intranasally, and the dose used was 0.93 μg. The GNP/protein/LPS conjugates generated significantly higher antibody titers than did native LPS. In addition, they improved protection against a lethal inhalation challenge of B. mallei in the murine model of infection. The proposed scheme in the form of aerosol immunization was successfully tested on rhesus monkeys [136]. a similar approach was developed for vaccination against B. pseudomallei myeloidosis [137]. Mice immunized three times subcutaneously with GNPs/protein/LPS generated high-titer antibodies. Importantly, the immunized animals survived nearly 100% and their lungs were less contaminated with bacteria after a lethal infection with B. pseudomallei.
Dakterzada et al. [138] evaluated the efficacy of GNPs conjugated with the N-terminal domains of Pseudomonas aeruginosa flagellin as an immunogen in the vaccination of mice. Nanoparticles were complexed with a recombinant flagellin (1–161). Flagellin, GNP/flagellin, and flagellin emulsified with CFA were administered twice subcutaneously to BALB/C mice at a dose of 10 μg. The mice given GNPs/flagellin elaborated more antiflagellin antibodies than did those that either were not immunized at all or were given adjuvant-free flagellin. In a whole-cell ELISA, these antibodies efficiently recognized native flagellin on the bacterial cells. An assay of opsonophagocytosis demonstrated the functional activity and specificity of the antiflagellin antibodies prepared by using the GNP/flagellin conjugate against the homologous strain.
The acidic serine protease AprV2 secreted by virulent Dichelobacter nodosus, the causative agent of infectious pododermatitis, was conjugated to 20-nm GNPs [139]. The nanovaccine induced higher IgG responses than did native AprV2 or the vaccine adjuvanted with monophosphoryl lipid A.
Conjugates of 15-nm GNPs with two isolated Francisella tularensis antigens, a protective antigenic complex and a glycosylated protein complex, were used to obtain antitularemia sera and vaccinate animals [140]. Subcutaneous immunization of mice with GNPs decorated with a glycosylated protein complex was more effective than was immunization with an unconjugated antigen. This greater effectiveness was manifested as an increase in the protection ability and titers of antibodies. The conjugation of GNPs to both antigens in the immunization of rabbits afforded sera with a high titer of specific antibodies in a relatively short period and with minimal consumption of the antigen. The use in ELISA of the immunoglobulins isolated from the sera allowed detection of F. tularensis cells of different subspecies, enabling their further use in the manufacture of diagnostic agents for tularemia.
Recombinant immunodominant antigens of Coxiella burnetii, the causative agent of Q fever, were conjugated to GNPs [141]. Immunization of mice and guinea pigs gave rise to specific antibodies and activated the response of the CD4+ T cells. The protective effect of the GNP-associated antigens was comparable to that of the commercial vaccine used.
Conjugates of 15-nm GNPs with seven Vibrio cholerae antigens were synthesized in [142]. Rabbits were immunized five times subcutaneously, and the resultant polyclonal antibodies to the test antigens showed a high specific activity. By using white laboratory mice, the protective activity of the conjugates was evaluated during infection of vaccinated animals, with a commercial vaccine as a control. The cholera vaccine prototypes made with GNPs as a carrier and an adjuvant corresponded to the commercial chemical vaccine in terms of protection efficiency.
Table 2 summarizes the literature data on the bacterial antigens that have been conjugated with GNP carriers and then used for immunization of animals.
Table 2.
Conjugates of GNPs with bacterial antigens used for immunization and vaccination of animals.
| Bacterial antigen | GNP size and shape | Functionalization approach (ligand) | Chemical nature of antigen | Refs. |
|---|---|---|---|---|
| Yersinia pseudotuberculosis | Nanospheres, 15 nm | Adsorption | Isolated protein | [119] |
| Yersinia pestis | Nanospheres, 15 nm | Chemosorption (NHS, EDC) | Recombinant protein | [120] |
| -”- | Nanospheres, 15 nm | Adsorption | Isolated protein | [121] |
| Neisseria meningitides* | Nanospheres, 5 nm | Chemosorption (thiols) | Synthetic carbohydrate | [122] |
| Salmonella typhimurium | Nanospheres, 15 nm | Adsorption | Isolated protein | [42] |
| Clostridium tetani* | Glyconanoparticles, 25 nm | Adsorption | Isolated protein | [123] |
| Streptococcus pneumoniae* | Glyconanoparticles, 2 nm | Direct synthesis | Synthetic carbohydrate | [127] |
| Mycobacterium tuberculosis | Nanospheres, 15 nm | Adsorption | Isolated peptide | [129] |
| -”-* | Nanospheres, 15 nm | Adsorption | Synthetic carbohydrate | [130] |
| Escherichia coli | Nanospheres, 30 nm | Adsorption | Isolated bacterial membrane | [131] |
| -”- | Nanospheres, 15 nm | Chemosorption (MHDA) | Recombinant proteins | [132] |
| Listeria monocytogenes* | Glyconanoparticles, 1.5 nm | Direct synthesis | Synthetic peptides | [133] |
| Burkholderia mallei* | Nanospheres, 15 nm | Chemosorption (NHS, EDC) | Isolated LPS | [135] |
| Burkholderia pseudomallei | Nanospheres, 15 nm | Chemosorption (NHS, EDC) | Isolated LPS | [137] |
| Pseudomonas aeruginosa | Nanospheres, 15 nm | Adsorption | Recombinant protein | [138] |
| Dichelobacter nodosus | Nanospheres, 20 nm | Adsorption | Recombinant protein | [139] |
| Francisella tularensis | Nanospheres, 15 nm | Adsorption | Isolated proteins | [140] |
| Coxiella burnetii | Nanospheres, 15 nm | Adsorption | Recombinant proteins | [141] |
| Vibrio cholerae | Nanospheres, 15 nm | Adsorption | Isolated proteins and LPS | [142] |
*Glyconanovaccine
4. Gold nanoparticles in the preparation of antiparasitic vaccines
Besides being used as an adjuvant in immunization against viral and bacterial diseases, GNPs are also used to develop antibodies and vaccines against parasitic infections. Parween et al. [143] reported that 17-nm GNPs in complex with recombinant peptides of the C-terminal 19kDa fragment of merozoite surface protein 1 from the malaria pathogen Plasmodium falciparum had a pronounced immunogenic activity when combined with the adjuvant Alhydrogel®. Mice were immunized three times subcutaneously at a dose of 25 μg. Antibodies against the weakly immunogenic peptides conjugated to GNPs recognized the native protein on the parasite’s surface and inhibited the invasion of P. falciparum in an in vitro assay.
Another immunogen was proposed to make an antimalarial vaccine – the P. falciparum surface protein Pfs25, expressed in zygotes and ookinetes [144]. The recombinant protein Pfs25 was attached to various GNPs, including nanospheres, nanostars, nanocages, and nanoprisms. Mice were immunized with the resulting conjugates three times intramuscularly at a dose of 10 μg. This yielded high-titer antibodies, the highest titers being obtained with gold nanospheres and nanostars. In membrane feeding assays, the antibodies blocked the transmission of parasites to mosquitoes.
GNPs were also used to prepare anti-idiotypic antibodies [145]. These antibodies were used for an indirect and a competitive ELISA of the opisthorchiasis pathogen Opisthorchis felineus. Work is underway to develop a vaccine against Babesia canis, the causative agent of canine piroplasmosis, by using the adjuvant properties of GNPs.
For vaccination against schistosomiasis, a parasitic disease caused by Schistosoma mansoni trematodes, gold nanorods were complexed with the recombinant protein rSm2, located on the surface of larvae and adult worms [146]. Mice were immunized three times intraperitoneally with 2 μg of the conjugate. After immunization, a Th1 immunological response was observed with higher production of IFN-γ, mainly by CD4+ and CD8+ T cells. In addition, the conjugate activated dendritic cells in vitro, enhancing the expression of MHCII and MHCI and the production of IL-1β. After vaccination with the conjugate, followed by a challenge with S. mansoni cercariae, the number of worms in the liver’s portal vein and the number of eggs in the liver tissues decreased, as compared with the control group.
Table 3 summarizes the literature data on the parasite antigens that have been conjugated with GNP carriers and then used for immunization of animals.
Table 3.
Conjugates of GNPs with parasite antigens used for immunization and vaccination of animals.
| Parasite antigen | GNP size and shape | Functionalization approach (ligand) | Chemical nature of antigen | Refs. |
|---|---|---|---|---|
| Plasmodium falciparum | Nanospheres, 17 nm | Adsorption | Recombinant peptides | [143] |
| -”- | Nanospheres, 30 nm; nanostars, 50 nm; nanocages, 60 nm; nanoprisms, 40 nm | Chemosorption (4-ATP) | Recombinant protein | [144] |
| Babesia canis | Nanospheres, 15 nm | Adsorption | Isolated proteins | [unpublished data] |
| Schistosoma mansoni | Nanorods | Adsorption | Recombinant protein | [146] |
5. Expert opinion
GNPs have been used to prepare antibodies and vaccines against more than 45 pathogens of viral, bacterial, and parasitic infections. The sizes of the GNPs used for this purpose have ranged from 2 to 100 nm. Moreover, in most studies, particle diameters ranging from 15 to 50 were recognized as optimal. Variously shaped GNPs have been used, including nanospheres, nanorods, nanocages, nanostars, nanocubes, nanoshells, nanoprisms, and nanoclusters. Most researchers have indicated that the best effect is achieved with gold nanospheres as antigen carriers.
Various methods have been used to functionalize GNPs with target antigens. Conjugation has been carried out both by physical adsorption and by chemisorption. Passive adsorption of an antigen on the particle surface occurs because of the electrostatic and hydrophobic interactions. Coulomb interactions between the H2 N groups of lysine and citrate ions on the surface of gold nanoparticles have also been reported. Recently, information has appeared about an important role of the SH groups of cysteine molecules in protein binding to the surface of gold particles. It is known that between sulfur and gold atoms, there arises a donor–acceptor (semipolar) bond (dative bound). This fact has led to the use of alkanethiol linkers (chemisorption method) for more stable attachment of biomolecules to GNPs. In addition, GNPs have often been conjugated to protein or polysaccharide linkers before being functionalized with an antigen. Most often, gold glyconanoparticles have been used as immunomodulators.
What other adjuvants can be used with GNPs remains an open question. Specific antibodies against target antigens have been obtained by using GNPs as the sole adjuvant in combination with immune response enhancers such as CFA, CpG-ODNs, MontanideTM, AdvaxTM, Alhydrogel®, Imiquimod, monophosphoryl lipid A, ISA206, and saponins. Moreover, both natural (native) antigens and genetically engineered (recombinant) ones have been used as immunogens.
Various animals have been used for immunization and vaccination, including mice, rats, guinea pigs, rabbits, chickens, and monkeys. The methods used to administer immunogens have included intraperitoneal, subcutaneous, intradermal, intramuscular, intravenous, intranasal, and oral. Moreover, the immunization doses of the antigens varied within a very wide range – for example, from 100 ng to 50 μg for mice or from 1 μg to 220 μg for rabbits. The frequency of administration also varied – from single to sixfold.
The antibody elaboration during immunization with GNP–antigen conjugates enhanced the secretion of cytokines – most often, IFN-γ, TNF-α, IL-1β, IL-4, IL-6, IL-10, and IL-12, and less often, IL-2, IL-5, IL-17, IL-18, MCP-1, and GM-CSF. Several researchers have noted the stimulation of the respiratory activity of peritoneal macrophages and spleen lymphocytes and the activation of the macrophage mitochondrial enzymes succinate dehydrogenase and glycerophosphate dehydrogenase. Immunization with GNP–antigen conjugates enhanced the proliferation of the CD4+ and CD8+ T cells and NK cells and stimulated both Th1 and Th2 immune responses. Other effects noted were the activation of dendritic cell maturation for antigen processing and presentation, increased expression of MHCII and MHCI, and activation of NLRP3 inflammasomes. The active uptake of GNPs by macrophages contributed to the processing of antigens. Splenocytes of the animals immunized with GNPs had higher proliferation rates than the cells immunized with the native antigen alone, which shows the ability of GNP-based carriers to induce enhanced cellular responses to the associated antigens. Often, GNPs decorated with bacterial antigens enhanced the phagocytic adhesion of microbial cells.
Antibodies obtained by using GNPs have been successfully applied to the immunochemical identification of pathogens with various solid-phase and microscopic methods and with biosensor devices. Vaccination of animals with GNP–antigen conjugates decreased the symptoms of infections and protected the animals challenged with virulent pathogens (up to lethal doses). In some cases, the vaccinated animals survived 100%. In most studies, the low toxicity of the gold carrier itself was noted [147].
Currently, the immunostimulatory activity of nanomaterials, including GNPs, as adjuvants is explained by the following mechanisms (Figure 5). Antigen–nanocarrier conjugates are more effectively delivered to the lymph nodes and induce a depot effect, which contributes to the steady and gradual release of the antigens. The use of nanoparticles as adjuvants helps antigens to concentrate on the membrane of dendritic cells and activates their maturation and the expression of MHCI and MHCII. The spatial organization (multivalence) of antigens on the surface of the particles enhances the proliferation of T cells and the activation of B cells. Both events are accompanied by the release of soluble mediators such as cytokines, chemokines, and immunomodulating molecules, which regulate the immune response.
Figure 5.
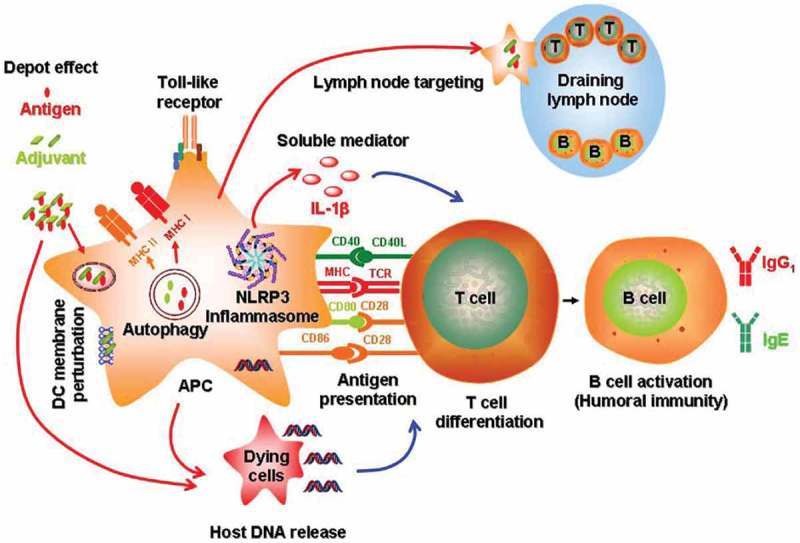
Mechanisms of immune system activation by engineered nanomaterials [11].
Antigen-presenting cells are crucial for the response to vaccines. Among the antigen-presenting cells, dendritic cells are especially important for the primary immune response, because they control the activation of the CD4+ and CD8+ T cells, which assist in the induction of antibodies and exhibit direct cytotoxic activity. Therefore, the key application of nanoparticles in immunology is the modulation of the functions of antigen-presenting cells. By ‘programming’ the activation state of dendritic or other antigen-presenting cells, nanoparticles directly affect the induction of cellular and humoral immunity [148].
It should be emphasized that GNPs are not biodegradable. Therefore, the biodistribution and excretion kinetics have to be studied comprehensively for different animal models. As the excretion of accumulated particles from the liver and spleen can take up to 3–4 months, the question as to the injected doses and possible inflammation processes is still of great importance. Bioaccumulated GNPs can interfere with different diagnostic techniques, or accumulated GNPs can exhibit catalytic properties. All these concerns, together with potential toxicity, are big limitations of GNPs on a successfully clinical translation. Nowadays, despite the huge numbers of studies regarding the synthesis and functionalization of GNPs (different shapes, coatings, sizes, charges, etc.), there are very few nanomaterial-based pharmaceuticals on the market.
The synthetic and natural polymeric biodegradable nanomaterials also can serve as antigen carriers. The advantages of nanoparticles of this type are that they are utilized well in the organism, the target substance is highly efficiently involved, there is a higher capability to overcome different physiological barriers, and there are less systematic side effects. The action mechanisms of biodegradable nanoparticles and GNPs as carriers of antigens in the immune system are probably similar. These two types of nanoobjects can compete in the development of a new generation of vaccines, taking into account data on bioinertness, low toxicity, and the good excretion of gold nanoparticles from the organism with the involvement of a hepatobiliary system [147].
Thus, GNPs, which have adjuvant properties, can be an excellent tool in the design of effective vaccines against infectious diseases.
Funding Statement
This work was supported by the Russian Science Foundation under grant no. 19-14-00077.
Article Highlights
Gold nanoparticles have been used to prepare antibodies and vaccines against more than 45 pathogens of viral, bacterial, and parasitic infections.
The antibody elaboration during immunization with antigens conjugated to gold nanoparticles enhances the secretion of cytokines.
Vaccination of animals with antigens conjugated to gold nanoparticles decreases the symptoms of infections and protects the animals challenged with virulent pathogens (up to lethal doses).
By ‘programming’ the activation state of dendritic or other antigen-presenting cells, nanoparticles directly affect the induction of cellular and humoral immunity.
Studying the characteristics of the immune response obtained from the use of gold nanoparticles as a carrier and an adjuvant will permit the particles’ potential for vaccine design to be increased.
Acknowledgments
I thank Mr DN Tychinin for his help in preparation of the manuscript.
Declaration of interest
The author has no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
Reviewer Disclosures
Peer reviewers on this manuscript have no relevant financial or other relationships to disclose.
References
Papers of special note have been highlighted as either of interest (•) or of considerable interest (••) to readers.
- 1.Gregory AE, Titball R, Williamson D.. Vaccine delivery using nanoparticles. Front Cell Infect Microbiol. 2013;3:13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Zaman M, Good MF, Toth I. Nanovaccines and their mode of action. Methods. 2013;60(3):226–231. [DOI] [PubMed] [Google Scholar]
- 3.Prashant CK, Kumar M, Dinda AK. Nanoparticle based tailoring of adjuvant function: the role in vaccine development. J Biomed Nanotechnol. 2014;10(9):2317–2331. [DOI] [PubMed] [Google Scholar]
- 4.Zhao L, Seth a, Wibowo N, et al. Nanoparticle vaccines. Vaccine. 2014;32(3):327–337. [DOI] [PubMed] [Google Scholar]
- 5.Sahdev P, Ochyl LJ, Moon JJ. Biomaterials for nanoparticle vaccine delivery systems. Pharm Res. 2014;31(10):2563–2582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Zhu M, Wang R, Nie G. Applications of nanomaterials as vaccine adjuvants. Hum Vaccin Immunother. 2014;10(9):2761–2774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Liu Y, Xu Y, Tian Y, et al. Functional nanomaterials can optimize the efficacy of vaccines. Small. 2014;10(22):4505–4520. . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Irvine DJ, Hanson MC, Rakhra K, et al. Synthetic nanoparticles for vaccines and immunotherapy. Chem Rev. 2015;115(19):11109–11146. . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Maughan CN, Preston SG, Williams GR. Particulate inorganic adjuvants: recent developments and future outlook. J Pharm Pharmacol. 2015;67(3):426–449. [DOI] [PubMed] [Google Scholar]
- 10.Gupta a, Das S, Schanen B, et al. Adjuvants in micro- to nanoscale: current state and future direction. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2016;8(1):61–84. [DOI] [PubMed] [Google Scholar]
- 11.Sun B, Xia T. Nanomaterial-based vaccine adjuvants. J Mater Chem B. 2016;4(33):5496–5509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Yang L, Li W, Kirberger M, et al. Design of nanomaterial based systems for novel vaccine development. Biomater Sci. 2016;4(5):785–802. . [DOI] [PubMed] [Google Scholar]
- 13.Andrade LM, Cox L, Versiani AF, et al. a growing world of small things: a brief review on the nanostructured vaccines. Future Virol. 2017;12(12):767–779. . [Google Scholar]
- 14.Shen Y, Hao T, Ou S, et al. Applications and perspectives of nanomaterials in novel vaccine development. MedChemComm. 2017;9(2):226–238. . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Li X, Wang X, Ito a. Tailoring inorganic nanoadjuvants towards next-generation vaccines. Chem Soc Rev. 2018;47(13):4954–4980. [DOI] [PubMed] [Google Scholar]
- 16.Hess KL, Medintz IL, Jewell CM. Designing inorganic nanomaterials for vaccines and immunotherapies. Nano Today. 2019;27:73–98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Facciolà a, Visalli G, Laganà P, et al. The new era of vaccines: the “nanovaccinology”. Eur Rev Med Pharmacol Sci. 2019;23(16):7163–7182. . [DOI] [PubMed] [Google Scholar]
- 18.Steabben DB. Studies on the physiological action of colloids. The action of colloidal substances on blob-elements and antibody content. Br J Exp Path. 1925;6:1–13. [Google Scholar]
- 19.Pacheco G. Studies on the action of metallic colloids on immunization. Mem Inst Oswaldo Cruz. 1925;18:119–149. [Google Scholar]
- 20.Zozaya J. Carbohydrates adsorbed on colloids as antigens. J Exp Med. 1932;55(3):325–351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Gros O, O’Connor JM. Einige Beobachtungen bei colloidalen Metallen mit Rucksicht auf ihre physikalisch-chemischen Eigenschaften und deren pharmakologische Wirkungen. Archiv F Exp Path U Pharmakol. 1911;64(5–6):456–467. [Google Scholar]
- 22.Zilber LA, Friese WW. Über die antigene Eigenschaften kolloidaler Metalle. Zh Eksp Biol. 1929;11:128–135. [Google Scholar]
- 23.Syed S, Zubair a, Frieri M. Immune response to nanomaterials: implications for medicine and literature review. Curr Allergy Asthma Rep. 2013;13(1):50–57. [DOI] [PubMed] [Google Scholar]
- 24.Farrera C, Fadeel B. It takes two to tango: understanding the interactions between engineered nanomaterials and the immune system. Eur J Pharm Biopharm. 2015;95:3–12. [DOI] [PubMed] [Google Scholar]
- 25.Petrarca C, Clemente E, Amato V, et al. Engineered metal based nanoparticles and innate immunity. Clin Mol Allergy. 2015;13(1):13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kononenko V, Narat M, Drobne D. Nanoparticle interaction with the immune system. Arh Hig Rada Toksikol. 2015;66(2):97–108. [DOI] [PubMed] [Google Scholar]
- 27.Luo Y-H, Chang LW, Lin P. Metal-based nanoparticles and the immune system: activation, inflammation, and potential applications. Biomed Res Int. 2015;2015:143720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Seth a, Oh D-B, Lim YT. Nanomaterials for enhanced immunity as an innovative paradigm in nanomedicine. Nanomedicine (Lond). 2015;10(6):959–975. [DOI] [PubMed] [Google Scholar]
- 29.Bracho-Sanchez E, Xia CQ, Clare-Salzler MJ, et al. Micro and nano material carriers for immunomodulation. Am J Transplant. 2016;16(12):3362–3370. . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Ilinskaya AN, Dobrovolskaia MA. Understanding the immunogenicity and antigenicity of nanomaterials: past, present and future. Toxicol Appl Pharmacol. 2016;299:70–77. [DOI] [PMC free article] [PubMed] [Google Scholar]; • This review extensively explores the current understanding of the immunogenicity and antigenicity of nanomaterials.
- 31.Fang RH, Zhang L. Nanoparticle-based modulation of the immune system. Annu Rev Chem Biomol Eng. 2016;7(1):13.1–13.22. [DOI] [PubMed] [Google Scholar]
- 32.David CAW, Owen a, Liptrott NJ. Determining the relationship between nanoparticle characteristics and immunotoxicity: key challenges and approaches. Nanomedicine (Lond). 2016;11(11):1447–1464. [DOI] [PubMed] [Google Scholar]
- 33.Moyano DF, Liu Y, Peer D, et al. Modulation of immune response using engineered nanoparticle surfaces. Small. 2016;12(1):76–82. . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Najafi-Hajivar S, Zakeri-Milani P, Mohammadi H, et al. Overview on experimental models of interactions between nanoparticles and the immune system. Biomed Pharmacother. 2016;83:1365–1378. [DOI] [PubMed] [Google Scholar]
- 35.Chattopadhyay S, Chen J-Y, Chen H-W, et al. Nanoparticle vaccines adopting virus-like features for enhanced immune potentiation. Nanotheranostics. 2017;1(3):244–260. . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Frey M, Bobbala S, Karabin N, et al. Influences of nanocarrier morphology on therapeutic immunomodulation. Nanomedicine (Lond). 2018;13(14):1795–1811. . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Kelly HG, Kent SJ, Wheatley AK. Immunological basis for enhanced immunity of nanoparticle vaccines. Expert Rev Vaccines. 2019;18(3):269–280. [DOI] [PubMed] [Google Scholar]
- 38.Fadeel B. Hide and seek: nanomaterial interactions with the immune system. Front Immunol. 2019;10:133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Dykman LA, Khlebtsov NG. Uptake of engineered gold nanoparticles into mammalian cells. Chem Rev. 2014;114(2):1258–1288. [DOI] [PubMed] [Google Scholar]
- 40.Patel PC, Giljohann DA, Daniel WL, et al. Scavenger receptors mediate cellular uptake of polyvalent oligonucleotide-functionalized gold nanoparticles. Bioconjug Chem. 2010;21(12):2250–2256. . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Franca a, Aggarwal P, Barsov EV, et al. Macrophage scavenger receptor a mediates the uptake of gold colloids by macrophages in vitro. Nanomedicine (Lond). 2011;6(7):1175–1188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Staroverov SA, Aksinenko NM, Gabalov KP, et al. Effect of gold nanoparticles on the respiratory activity of peritoneal macrophages. Gold Bull. 2009;42(2):153–156. [Google Scholar]
- 43.Yen H-J, Hsu S-H, Tsai C-L. Cytotoxicity and immunological response of gold and silver nanoparticles of different sizes. Small. 2009;5(13):1553–1561. [DOI] [PubMed] [Google Scholar]
- 44.Cheung W-H, Chan VS-F, Pang H-W, et al. Conjugation of latent membrane protein (LMP)-2 epitope to gold nanoparticles as highly immunogenic multiple antigenic peptides for induction of Epstein−Barr virus-specific cytotoxic T-Lymphocyte responses in vitro. Bioconjug Chem. 2009;20(1):24–31. . [DOI] [PubMed] [Google Scholar]
- 45.Cruz LJ, Rueda F, Cordobilla B, et al. Targeting nanosystems to human DCs via Fc receptor as an effective strategy to deliver antigen for immunotherapy. Mol Pharm. 2011;8(1):104–116. . [DOI] [PubMed] [Google Scholar]
- 46.Bartneck M, Keul HA, Singh S, et al. Rapid uptake of gold nanorods by primary human blood phagocytes and immunomodulatory effects of surface chemistry. ACS Nano. 2010;4(6):3073–3086. . [DOI] [PubMed] [Google Scholar]
- 47.Chekanov AV, Baranova OA, Levin AD, et al. Influence of gold nanoparticles on activation of human blood neutrophils. Biophysics. 2013;58:385–388. [PubMed] [Google Scholar]
- 48.Le Guevel X, Palomares F, Torres MJ, et al. Nanoparticle size influences the proliferative responses of lymphocyte subpopulations. RSC Adv. 2015;5(104):85305–85309. . [Google Scholar]
- 49.Liptrott NJ, Kendall E, Nieves DJ, et al. Partial mitigation of gold nanoparticle interactions with human lymphocytes by surface functionalization with a ‘mixed matrix’. Nanomedicine (Lond). 2014;9(16):2467–2479. . [DOI] [PubMed] [Google Scholar]
- 50.Li Y, Italiani P, Casals E, et al. Assessing the immunosafety of engineered nanoparticles with a novel in vitro model based on human primary monocytes. ACS Appl Mater Interfaces. 2016;8(42):28437–28447. . [DOI] [PubMed] [Google Scholar]
- 51.Srijampa S, Buddhisa S, Ngernpimai S, et al. Effects of gold nanoparticles with different surface charges on cellular internalization and cytokine responses in monocytes. BioNanoSci. 2019;9(3):580–586. . [Google Scholar]
- 52.Blecher K, Nasir a, Friedman a. The growing role of nanotechnology in combating infectious disease. Virulence. 2011;2(5):395–401. [DOI] [PubMed] [Google Scholar]
- 53.Fan Y, Moon JJ. Particulate delivery systems for vaccination against bioterrorism agents and emerging infectious pathogens. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2017;9(1):e1403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Neto LMM, Kipnis a, Junqueira-Kipnis AP. Role of metallic nanoparticles in vaccinology: implications for infectious disease vaccine development. Front Immunol. 2017;8:239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Pati R, Shevtsov M, Sonawane a. Nanoparticle vaccines against infectious diseases. Front Immunol. 2018;9:2224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Sokolova V, Westendorf AM, Buer J, et al. The potential of nanoparticles for the immunization against viral infections. J Mater Chem B. 2015;3(24):4767–4779. . [DOI] [PubMed] [Google Scholar]
- 57.Glass JJ, Kent SJ, De Rose R. Enhancing dendritic cell activation and HIV vaccine effectiveness through nanoparticle vaccination. Expert Rev Vaccines. 2016;15(6):719–729. [DOI] [PubMed] [Google Scholar]
- 58.Liu Y, Chen C. Role of nanotechnology in HIV/AIDS vaccine development. Adv Drug Deliv Rev. 2016;103:76–89. [DOI] [PubMed] [Google Scholar]
- 59.Deng L, Wang B-Z. a perspective on nanoparticle universal influenza vaccines. ACS Infect Dis. 2018;4(12):1656–1665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Al-Halifa S, Gauthier L, Arpin D, et al. Nanoparticle-based vaccines against respiratory viruses. Front Immunol. 2019;10:22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Lin LC-W, Chattopadhyay S, Lin J-C, et al. Advances and opportunities in nanoparticle- and nanomaterial-based vaccines against bacterial infections. Adv Healthc Mater. 2018;7(13):e1701395. [DOI] [PubMed] [Google Scholar]
- 62.Huang W-C, Sia ZR, Lovell JF. Adjuvant and antigen systems for malaria transmission-blocking vaccines. Adv Biosys. 2018;2(10):e1800011. [Google Scholar]
- 63.Vartak a, Sucheck SJ. Recent advances in subunit vaccine carriers. Vaccines (Basel). 2016;4(2):12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Fujita Y, Taguchi H. Current status of multiple antigen-presenting peptide vaccine systems: application of organic and inorganic nanoparticles. Chem Cent J. 2011;5(1):48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Marasini N, Skwarczynski M, Toth I. Oral delivery of nanoparticle-based vaccines. Expert Rev Vaccines. 2014;13(11):1361–1376. [DOI] [PubMed] [Google Scholar]
- 66.Wang S, Liu H, Zhang X, et al. Intranasal and oral vaccination with protein-based antigens: advantages, challenges and formulation strategies. Protein Cell. 2015;6(7):480–503. . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Kaurav M, Minz S, Sahu K, et al. Nanoparticulate mediated transcutaneous immunization: myth or reality. Nanomedicine. 2016;12(4):1063–1081. . [DOI] [PubMed] [Google Scholar]
- 68.Comber JD, Bamezai a. Gold nanoparticles (AuNPs): a new frontier in vaccine delivery. J Nanomedine Biotherapeutic Discov. 2015;5(4):4. [Google Scholar]
- 69.Salazar-González JA, González-Ortega O, Rosales-Mendoza S. Gold nanoparticles and vaccine development. Expert Rev Vaccines. 2015;14(9):1197–1211. [DOI] [PubMed] [Google Scholar]
- 70.Dykman LA, Khlebtsov NG. Immunological properties of gold nanoparticles. Chem Sci. 2017;8(3):1719–1735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Carabineiro SAC. Applications of gold nanoparticles in nanomedicine: recent advances in vaccines. Molecules. 2017;22:857. [DOI] [PMC free article] [PubMed] [Google Scholar]; •• This review explores the important role of gold nanoparicles in vaccine design and their use as an adjuvant and a carrier.
- 72.Dykman LA, Khlebtsov NG. Gold Nanoparticles in Biomedical Applications. Boca Raton: CRC Press; 2017. [Google Scholar]
- 73.Shiosaka S, Kiyama H, Wanaka a, et al. a new method for producing a specific and high titre antibody against glutamate using colloidal gold as a carrier. Brain Res. 1986;382(2):399–403. . [DOI] [PubMed] [Google Scholar]
- 74.Dykman LA, Sumaroka MV, Staroverov SA, et al. Immunogenic properties of colloidal gold. Biol Bull Russ Acad Sci. 2004;31(1):75–79. . [PubMed] [Google Scholar]
- 75.Dykman LA, Staroverov SA, Bogatyrev VA, et al. Adjuvant properties of gold nanoparticles. Nanotechnol Russ. 2010;5(11–12):748–761. . [Google Scholar]
- 76.Dykman LA, Staroverov SA, Bogatyrev VA, et al. Gold nanoparticles as an antigen carrier and an adjuvant. Hauppauge: Nova Science Publishers; 2010. [Google Scholar]
- 77.Demenev VA, Shchinova MA, Ivanov LI, et al. Perfection of methodical approaches to designing vaccines against tick-borne encephalitis. Vopr Virusol. 1996;41(3):107–110. [PubMed] [Google Scholar]
- 78.Gaidamovich SY, Demenev VA, Obukhova VR. Differentiation in the complement fixation test of the viruses of tick-borne encephalitis complex by means of a type-specific soluble antigen. Acta Virol. 1985;29(2):143–149. [PubMed] [Google Scholar]
- 79.Chen Y-S, Hung Y-C, Liau I, et al. Assessment of the in vivo toxicity of gold nanoparticles. Nanoscale Res Lett. 2009;4(8):858–864. . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Chen Y-S, Hung Y-C, Liau I, et al, Assessment of gold nanoparticles as a size-dependent vaccine carrier for enhancing the antibody response against synthetic foot-and-mouth disease virus peptide. Nanotechnology. 2010;21(19): 195101. . [DOI] [PubMed] [Google Scholar]; • This research article extensively explores the use of gold nanoparticles as a vaccine carrier for enhancing the antibody response against synthetic foot-and-mouth disease virus peptide.
- 81.Mezhenny PV, Staroverov SA, Volkov AA, et al. Construction of conjugates of colloidal selenium and colloidal gold with the protein of influenza virus and the study of their immunogenic properties. Bull Saratov State Agrarian Univ. 2013;2:29–32. [Google Scholar]
- 82.Dykman LA, Staroverov SA, Mezhenny PV, et al. Use of a synthetic foot-and-mouth disease virus peptide conjugated to gold nanoparticles for enhancing immunological response. Gold Bull. 2015;48(1–2):93–101. . [Google Scholar]
- 83.Teng Z, Sun S, Chen H, et al. Golden-star nanoparticles as adjuvant effectively promotes immune response to foot-and-mouth disease virus-like particles vaccine. Vaccine. 2018;36(45):6752–6760. . [DOI] [PubMed] [Google Scholar]
- 84.Tao W, Ziemer KS, Gill HS. Gold nanoparticle–M2e conjugate coformulated with CpG induces protective immunity against influenza a virus. Nanomedicine (Lond). 2014;9(2):237–251. . [DOI] [PMC free article] [PubMed] [Google Scholar]; •• This research article extensively explores the mechanisms of protective immunity against influenza a virus, with gold nanoparticle conjugates used as an adjuvant.
- 85.Tao W, Gill HS. M2e-immobilized gold nanoparticles as influenza a vaccine: role of soluble M2e and longevity of protection. Vaccine. 2015;33(20):2307–2315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Tao W, Hurst B, Shakya AK, et al. Consensus M2e peptide conjugated to gold nanoparticles confers protection against H1N1, H3N2 and H5N1 influenza a viruses. Antiviral Res. 2017;141:62–72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Bimler L, Song AY, Le DT, et al. AuNP-M2e + sCpG vaccination of juvenile mice generates lifelong protective immunity to influenza a virus infection. Immun Ageing. 2019;16(1):23. . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Dykman LA, Staroverov SA, Fomin AS. Effect of M2e peptide–gold nanoparticle conjugates on development of anti-influenza antibodies. Gold Bull. 2018;51(4):197–203. [Google Scholar]
- 89.Weiner GJ, Liu HM, Wooldridge JE, et al. Immunostimulatory oligodeoxynucleotides containing the CpG motif are effective as immune adjuvants in tumor antigen immunization. Proc Natl Acad Sci USA. 1997;94(20):10833–10837. . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Rothenfusser S, Tuma E, Wagner M, et al. Recent advances in immunostimulatory CpG oligonucleotides. Curr Opin Mol Ther. 2003;5(2):98–106. [PubMed] [Google Scholar]
- 91.Wei M, Chen N, Li J, et al. Polyvalent immunostimulatory nanoagents with self-assembled CpG oligonucleotide-conjugated gold nanoparticles. Angew Chem Int Ed Engl. 2012;51(5):1202–1206. . [DOI] [PubMed] [Google Scholar]
- 92.Chen N, Wei M, Sun Y, et al. Self-assembly of poly-adenine-tailed CpG oligonucleotide-gold nanoparticle nanoconjugates with immunostimulatory activity. Small. 2014;10(2):368–375. . [DOI] [PubMed] [Google Scholar]
- 93.Zhang H. Gao X-D Nanodelivery systems for enhancing the immunostimulatory effect of CpG oligodeoxynucleotides. Mater Sci Eng C. 2017;70:935–946. [DOI] [PubMed] [Google Scholar]
- 94.Dykman LA, Staroverov SA, Fomin AS, et al. Gold nanoparticles as an adjuvant: influence of size, shape, and technique of combination with CpG on antibody production. Int Immunopharmacol. 2018;54:163–168. [DOI] [PubMed] [Google Scholar]
- 95.Tazaki T, Tabata K, Ainai a, et al. Shape-dependent adjuvanticity of nanoparticle-conjugated RNA adjuvants for intranasal inactivated influenza vaccines. RSC Adv. 2018;8(30):16527–16536. . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Wang C, Zhu W, Wang B-Z. Dual-linker gold nanoparticles as adjuvanting carriers for multivalent display of recombinant influenza hemagglutinin trimers and flagellin improve the immunological responses in vivo and in vitro. Int J Nanomedicine. 2017;12:4747–4762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Wang C, Zhu W, Luo Y, et al. Gold nanoparticles conjugating recombinant influenza hemagglutinin trimers and flagellin enhanced mucosal cellular immunity. Nanomedicine. 2018;14(4):1349–1360. . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Staroverov SA, Vidyasheva IV, Gabalov KP, et al. Immunostimulatory effect of gold nanoparticles conjugated with transmissible gastroenteritis virus. Bull Exp Biol Med. 2011;151(4):436–439. . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Staroverov SA, Volkov AA, Mezhennyy PV, et al. Prospects for the use of spherical gold nanoparticles in immunization. Appl Microbiol Biotechnol. 2019;103(1):437–447. . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Sharapova NA, Kireev MN, Abramova EG, et al. Isolation of glicoproteid from the fixed rabies virus, strain ‘Moscow 3253ʹ, and constructing of dot-immunoassay diagnosticum on its basis. Acta Biomedica Scientifica. 2012;5:347–350. [Google Scholar]
- 101.Gavrilova YK, Generalov SV, Kireev MN, et al. Development of the scheme of obtaining antibodies to the ribonucleoprotein of attenuated rabies virus. Zh Microbiol Epidemiol Immunobiol. 2019;5:3–8. [Google Scholar]
- 102.Niikura K, Matsunaga T, Suzuki T, et al. Gold nanoparticles as a vaccine platform: influence of size and shape on immunological responses in vitro and in vivo. ACS Nano. 2013;7(5):3926–3938. . [DOI] [PubMed] [Google Scholar]
- 103.Stone JW, Thornburg NJ, Blum DL, et al. Gold nanorod vaccine for respiratory syncytial virus. Nanotechnology. 2013;24(29):295102. . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Di Gianvincenzo P, Calvo J, Perez S, et al. Negatively charged glyconanoparticles modulate and stabilize the secondary structures of a gp120 V3 loop peptide: toward fully synthetic HIV vaccine candidates. Bioconjug Chem. 2015;26(4):755–765. . [DOI] [PubMed] [Google Scholar]
- 105.Climent N, García I, Marradi M, et al. Loading dendritic cells with gold nanoparticles (GNPs) bearing HIV-peptides and mannosides enhance HIV-specific T cell responses. Nanomedicine. 2018;14(2):339–351. . [DOI] [PubMed] [Google Scholar]
- 106.Lin F. Development of gold nanoparticle-based antigen delivery platform for vaccines against HIV-1 [Graduate Theses and Dissertations]. Iowa State University; 2015. [Google Scholar]
- 107.Wang Y, Wang Y, Kang N, et al. Construction and immunological evaluation of CpG-Au@HBc virus-like nanoparticles as a potential vaccine. Nanoscale Res Lett. 2016;11(1):338. . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Yavuz E, Bagriacik EU. Gold-based nano-adjuvants. Proc. IEEE 7-th International Conference on Nanomaterials: Applications and Properties, Odesa, Ukraine 2017;04NB06-1–04NB06-4. [Google Scholar]
- 109.Wang H, Ding Y, Su S, et al. Assembly of hepatitis E vaccine by ‘in situ’ growth of gold clusters as nano-adjuvants: an efficient way to enhance the immune responses of vaccination. Nanoscale Horiz. 2016;1(5):394–398. . [DOI] [PubMed] [Google Scholar]
- 110.Olenina LV, Kolesanova EF, Gervaziev YV, et al. Preparation of anti-peptide antibodies to the protein binding sites E2 of hepatitis C virus with CD81. Med Immunol. 2001;3:231. [Google Scholar]
- 111.Versiani AF, Andrade LM, Martins EMN, et al. Gold nanoparticles and their applications in biomedicine. Future Virol. 2016;11(4):293–309. . [Google Scholar]
- 112.Quach QH, Ang SK, Chu J-HJ, et al. Size-dependent neutralizing activity of gold nanoparticle-based subunit vaccine against dengue virus. Acta Biomater. 2018;78:224–235. [DOI] [PubMed] [Google Scholar]
- 113.Chen H-W, Huang C-Y, Lin S-Y, et al. Synthetic virus-like particles prepared via protein corona formation enable effective vaccination in an avian model of coronavirus infection. Biomaterials. 2016;106:111–118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Sekimukai H, Iwata-Yoshikawa N, Fukushi S, et al. Gold nanoparticle-adjuvanted S protein induces a strong antigen-specific IgG response against severe acute respiratory syndrome-related coronavirus infection, but fails to induce protective antibodies and limit eosinophilic infiltration in lungs. Microbiol Immunol. 2020;64(1):33–51. . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Kareem S, Altimimi MB, Jarullah BA. Improvement of Newcastle disease virus vaccine by using gold nanoparticles and some natural food additives. J Thi-Qar Sci. 2017;6:59–64. [Google Scholar]
- 116.Ding P, Zhang T, Li Y, et al. Nanoparticle orientationally displayed antigen epitopes improve neutralizing antibody level in a model of porcine circovirus type 2. Int J Nanomedicine. 2017;12:5239–5254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Li Y, Jin Q, Ding P, et al. Gold nanoparticles enhance immune responses in mice against recombinant classical swine fever virus E2 protein. Biotechnol Lett. 2020. doi: 10.1007/s10529-020-02853-w. [DOI] [PubMed] [Google Scholar]
- 118.Soliman MG, Mohamed AF, El Sayed RA, et al. Immune enhancing potential of sphere and rod gold nanoparticles to Rift Valley fever vaccine relative to time: in vitro study. Eur J Biomed Pharm Sci. 2017;4:529–536. [Google Scholar]
- 119.Staroverov SA, Ermilov DN, Shcherbakov AA, et al. Generation of antibodies to Yersinia pseudotuberculosis antigens using the colloid gold particles as an adjuvant. Zh Mikrobiol Epidemiol Immunobiol. 2003;3:54–57. [PubMed] [Google Scholar]
- 120.Gregory AE, Williamson ED, Prior JL, et al. Conjugation of Y. pestis F1-antigen to gold nanoparticles improves immunogenicity. Vaccine. 2012;30(48):6777–6782. . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Kireev MN, Polunina TA, Guseva NP, et al. Studying the immunogenic properties of plague microbe capsule antigen F1 conjugated with nanoparticles of colloid gold and silver. Prob Particularly Dangerous Infect. 2008;2(2(96)):43–45. . [Google Scholar]
- 122.Manea F, Bindoli C, Fallarini S, et al. Multivalent, saccharide-functionalized gold nanoparticles as fully synthetic analogs of type a Neisseria meningitidis Antigens. Adv Mater. 2008;20(22):4348–4352. . [Google Scholar]
- 123.Pokharkar V, Bhumkar D, Suresh K, et al. Gold nanoparticles as a potential carrier for transmucosal vaccine delivery. J Biomed Nanotechnol. 2011;7(1):57–59. . [DOI] [PubMed] [Google Scholar]
- 124.Barhate G, Gautam M, Gairola S, et al. Quillaja saponaria extract as mucosal adjuvant with chitosan functionalized gold nanoparticles for mucosal vaccine delivery: stability and immunoefficiency studies. Int J Pharm. 2013;441(1–2):636–642. . [DOI] [PubMed] [Google Scholar]
- 125.Barhate G, Gautam M, Gairola S, et al. Enhanced mucosal immune responses against tetanus toxoid using novel delivery system comprised of chitosan-functionalized gold nanoparticles and botanical adjuvant: characterization, immunogenicity, and stability assessment. J Pharm Sci. 2014;103(11):3448–3456. [DOI] [PubMed] [Google Scholar]
- 126.Rabiee Rudsari a, Ebrahimi F, Arefpour Torabi MA. Study of adjuvant capability of the gold nanoparticles on the immunity of botulinum neurotoxin serotype E in mouse. Sci J Adv Defence Sci Technol. 2013;4:87–92. [Google Scholar]
- 127.Safari D, Marradi M, Chiodo F, et al. Gold nanoparticles as carriers for a synthetic Streptococcus pneumoniae type 14 conjugate vaccine. Nanomedicine (Lond). 2012;5(5):651–662. . [DOI] [PubMed] [Google Scholar]
- 128.Vetro M, Safari D, Fallarini S, et al. Preparation and immunogenicity of gold glyco-nanoparticles as antipneumococcal vaccine model. Nanomedicine (Lond). 2017;12(1):13–23. . [DOI] [PubMed] [Google Scholar]
- 129.Staroverov SA, Dykman LA. Use of gold nanoparticles for the preparation of antibodies to tuberculin, the immunoassay of mycobacteria, and animal vaccination. Nanotechnol Russia. 2013;8(11–12):816–822. [Google Scholar]
- 130.Burygin GL, Abronina PI, Podvalnyy NM, et al. Preparation and in vivo evaluation of glyco-gold nanoparticles carrying synthetic mycobacterial hexaarabinofuranoside. Beilstein J Nanotechnol. 2020;11:480–493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Gao W, Fang RH, Thamphiwatana S, et al. Modulating antibacterial immunity via bacterial membrane-coated nanoparticles. Nano Lett. 2015;15(2):1403–1409. . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Sanchez-Villamil JI, Tapia D, Torres AG. Development of a gold nanoparticle vaccine against enterohemorrhagic Escherichia coli O157:H7. mBio. 2019;10(4):e01869–19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Rodriguez-Del Rio E, Marradi M, Calderon-Gonzalez R, et al. a gold glyco-nanoparticle carrying a listeriolysin O peptide and formulated with AdvaxTM delta inulin adjuvant induces robust T-cell protection against listeria infection. Vaccine. 2015;33(12):1465–1473. . [DOI] [PubMed] [Google Scholar]
- 134.Calderón-Gonzalez R, Terán-Navarro H, Frande-Cabanes E, et al. Pregnancy vaccination with gold glyco-nanoparticles carrying Listeria monocytogenes peptides protects against listeriosis and brain- and cutaneous-associated morbidities. Nanomaterials. 2016;6(8):151. . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Gregory AE, Judy BM, Qazi O, et al. a gold nanoparticle-linked glycoconjugate vaccine against Burkholderia mallei. Nanomedicine. 2015;11(2):447–456. . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Torres AG, Gregory AE, Hatcher CL, et al. Protection of non-human primates against glanders with a gold nanoparticle glycoconjugate vaccine. Vaccine. 2015;33(5):686–692. . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Muruato LA, Tapia D, Hatcher CL, et al. Use of reverse vaccinology in the design and construction of nanoglycoconjugate vaccines against Burkholderia pseudomallei. Clin Vaccine Immunol. 2017;24(11):e00206–17. . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Dakterzada F, Mohabati Mobarez a, Habibi Roudkenar M, et al. Induction of humoral immune response against Pseudomonas aeruginosa flagellin(1-161) using gold nanoparticles as an adjuvant. Vaccine. 2016;34(12):1472–1479. . [DOI] [PubMed] [Google Scholar]
- 139.Srivastva P. Evaluation of a candidate GNP-APRV2 footrot nanovaccine in mice. CSK-Himachal Pradesh Agricultural University [Graduate Theses and Dissertations] 2017. [Google Scholar]
- 140.Dykman LA, Volokh OA, Kuznetsova EM, et al. Immunogenicity of conjugates of protective antigen complexes of tularemia microbe with gold nanoparticles. Nanotechnol Russ. 2018;13(7–8):384–392. . [Google Scholar]
- 141.Van Schaik EJ, Gregory AE, Audette GF, et al. Nanovaccines against intracellular pathogens using Coxiella burnetii as a model organism. Immune aspects of biopharmaceuticals and nanomedicines. Singapore: Pan Stanford Publishing; 2018. [Google Scholar]
- 142.Dykman LA, Volokh OA, Gromova OV, et al. Obtaining and characteristic of antibodies to Vibrio cholerae protective antigens conjugated with gold nanoparticles. Dokl Biochem Biophys. 2020;490:19-12. [DOI] [PubMed] [Google Scholar]
- 143.Parween S, Gupta PK, Chauhan VS. Induction of humoral immune response against PfMSP-119 and PvMSP-119 using gold nanoparticles along with alum. Vaccine. 2011;29(13):2451–2460. [DOI] [PubMed] [Google Scholar]
- 144.Kumar R, Ray PC, Datta D, et al. Nanovaccines for malaria using Plasmodium falciparum antigen Pfs25 attached gold nanoparticles. Vaccine. 2015;33(39):5064–5071. . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Bulashev AK, Borovikov SN, Serikova SS, et al. Development of an ELISA using anti-idiotypic antibody for diagnosis of opisthorchiasis. Folia Parasitol. 2016;63:025. [DOI] [PubMed] [Google Scholar]
- 146.Assis NRG, Caires AJ, Figueiredo BC, et al. The use of gold nanorods as a new vaccine platform against schistosomiasis. J Control Release. 2018;275:40–52. [DOI] [PubMed] [Google Scholar]
- 147.Khlebtsov NG, Dykman LA. Biodistribution and toxicity of engineered gold nanoparticles: a review of in vitro and in vivo studies. Chem Soc Rev. 2011;40(3):1647–1671. [DOI] [PubMed] [Google Scholar]
- 148.Vijayan V, Mohapatra a, Uthaman S, et al. Recent advances in nanovaccines using biomimetic immunomodulatory materials. Pharmaceutics. 2019;11:534. [DOI] [PMC free article] [PubMed] [Google Scholar]; • This review summarizes the recent advances in the design of nanovaccines using biomimetic immunomodulatory materials.


