Abstract
The limited number of antifungals and the rising frequency of azole-resistant Candida species are growing challenges to human medicine. Drug repurposing signifies an appealing approach to enhance the activity of current antifungal drugs. Here, we evaluated the ability of Pharmakon 1600 drug library to sensitize an azole-resistant Candida albicans to the effect of fluconazole. The primary screen revealed 44 non-antifungal hits were able to act synergistically with fluconazole against the test strain. Of note, 21 compounds, showed aptness for systemic administration and limited toxic effects, were considered as potential fluconazole adjuvants and thus were termed as “repositionable hits”. A follow-up analysis revealed pitavastatin displaying the most potent fluconazole chemosensitizing activity against the test strain (ΣFICI 0.05) and thus was further evaluated against 18 isolates of C. albicans (n = 9), C. glabrata (n = 4), and C. auris (n = 5). Pitavastatin displayed broad-spectrum synergistic interactions with both fluconazole and voriconazole against ~89% of the tested strains (ΣFICI 0.05–0.5). Additionally, the pitavastatin-fluconazole combination significantly reduced the biofilm-forming abilities of the tested Candida species by up to 73%, and successfully reduced the fungal burdens in a Caenorhabditis elegans infection model by up to 96%. This study presents pitavastatin as a potent azole chemosensitizing agent that warrant further investigation.
Subject terms: Drug discovery, Microbiology
Introduction
Candida species are the most common nosocomial fungal pathogens and are a major cause of healthcare-associated bloodstream infections1–3. In the USA, Candida species are the fourth-leading cause of bloodstream infections4,5. Diseases caused by Candida species can range from self-limited uncomplicated superficial lesions to a deadly form of disseminated invasive infection that is often associated with a high mortality rate (42–65%)6. Available epidemiological data derived from several independent surveillance studies portray C. albicans and C. glabrata as the two major causes of Candida-related infections in North America and Europe7–9. However, the recent emergence of C. auris has become a global health concern, considering its unique multidrug resistance nature, the efficient ability to colonize human tissues and to provoke several global outbreaks10,11. Thus, C. auris was recently categorized by the US Centers for Disease Control and Prevention (CDC) as an urgent health threat12.
Treatment of systemic Candida infections is currently limited to only three major drug classes; azoles, polyenes, and echinocandins13,14. The limited toxicity, oral bioavailability, and broad-spectrum of antifungal activities made azoles the most commonly prescribed drugs for treating and controlling Candida infections14,15. Azoles exert their antifungal activity through the inhibition of lanosterol 14-alpha-demethylase, Erg11, an essential step in the ergosterol biosynthesis pathway. Interference with the ergosterol biosynthesis pathway significantly compromises the functions of fungal cell membranes16. Unfortunately, excessive use of azole antifungal agents has been associated with the emergence of azole-resistant Candida strains17,18.
Given the clinical importance of azole antifungals, there is a pressing need for potent co-drugs that would augment the antifungal effect of azole drugs, particularly against Candida biofilms and azole-resistant strains. Drug repurposing is a promising approach that can be utilized to improve the activity of current antifungal, reduce their toxicity, and even to overcome the rising antifungal resistance. In this study, we explored the fluconazole chemosensitizing activity of ~1600 approved drugs and clinical molecules from the Pharmakon drug library. The primary screen identified 44 non-antifungal hit compounds that were able to sensitize an azole-resistant C. albicans strain to the effect of fluconazole. A follow-up analysis of identified hits revealed pitavastatin as the most potent fluconazole chemosensitizing agent and thus was further investigated in combination with different azole drugs against 18 strains of C. albicans, C. glabrata, and the multidrug-resistant C. auris. The pitavastatin-fluconazole combination was also evaluated for the ability to inhibit Candida biofilm formation and was assessed for the ability to reduce Candida burdens in infected Caenorhabditis elegans. Furthermore, the effect of pitavastatin on the efflux activities of Candida strains with known efflux mechanisms was evaluated.
Results and Discussion
Screening of Pharmakon drug library and identification of fluconazole adjuvants hit compounds
We performed an initial screen of the Pharmakon 1600 drug library, at a 16 µM fixed concentration, to identify potential fluconazole adjuvants, for which we used a standard broth microdilution method following the guidelines of the Clinical and Laboratory Standards Institute (CLSI). The screen was performed twice against the azole-resistant C. albicans NR-29448, in the presence or absence of 8 µg/ml fluconazole. This high fluconazole concentration was opted to maximize the initial pool of positive hits. Positive hits were identified as hit compounds that caused significant growth inhibition (by >50%) of the test strain only in the presence of fluconazole. Positive hits were initially determined by visual inspection then further confirmed spectrophotometrically by measuring the absorbance of Candida culture at OD 490 nM. The primary screen identified a list of 44 positive hits (2.75% initial hit rate) that exhibited synergistic interactions with fluconazole against the azole-resistant strain C. albicans NR-29448. These initial hits were sub-grouped into seven antineoplastic agents, eight antiparasitics, eight topical agents and 21 drugs that were considered potential fluconazole adjuvants for treating systemic infections and thus were termed “repositionable drugs” (Fig. 1). Notably, several hit compounds that were classified as topical agents and antiparasitics (Supplementary Table ST1) could hold promising clinical potential for treating topical Candida infections. For example, bufexamac, a topical anti-inflammatory drug, may worth further investigation as part of a future study to treat mucosal and skin infections, especially those caused by azole-resistant Candida species.
Figure 1.
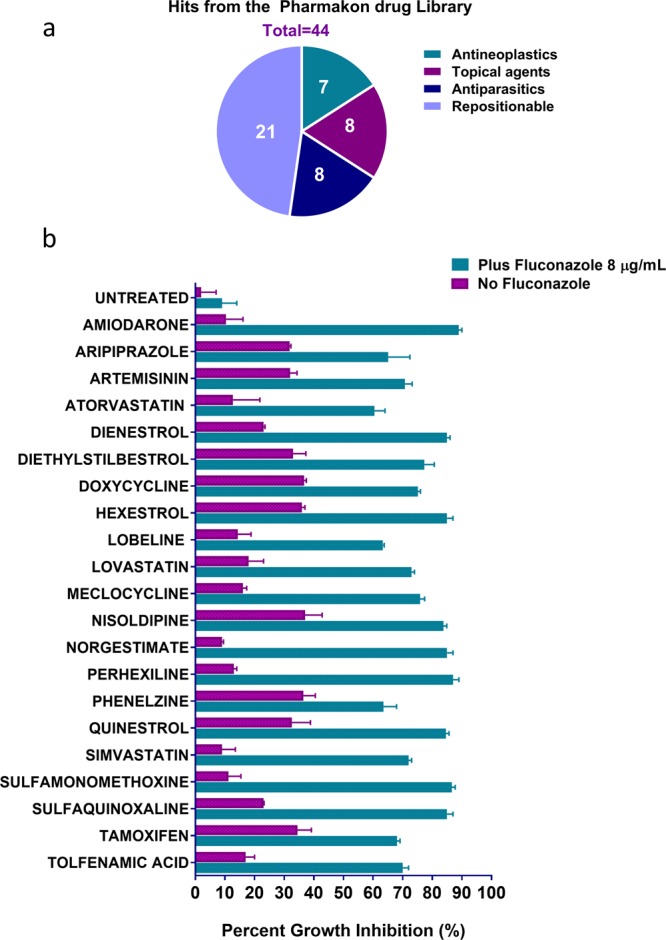
Primary screening of the Pharmakon 1600 drug library. (a) Graphical representation of results of primary screening. Initial hits were classified into four classes: antineoplastic, topical, antiparasitic, and repositionable agents (b). Percent growth inhibition according to OD 490 spectrophotometric readings. Experiments were performed in duplicates and bars indicate standard error.
However, since the main focus of this study was to identify potent systemic fluconazole adjuvants, we directed our attention to study the fluconazole chemosensitizing activities of the repositionable drugs for their aptness for systemic administration and for their relatively low toxicity profiles. Next, we determined the minimum fluconazole-chemosensitizing concentrations of these drugs against C. albicans NR-29448, in the presence or absence of fluconazole (at 8 µg/ml). Interestingly, the antihyperlipidemic agent simvastatin demonstrated a significant fluconazole chemosensitizing activity at 8 µM while all other hit compounds showed activities only at16 µM (Table 1).
Table 1.
Minimum inhibitory concentrations (MICs) of repositionable hit compounds in the presence or absence of fluconazole.
| Identified Hit Compounds | MIC (µM) | |
|---|---|---|
| Without fluconazole | Plus fluconazole 8 µg/ml | |
| Amiodarone | >16 | 16 |
| Aripiprazole | >16 | 16 |
| Artemisinin | >16 | 16 |
| Atorvastatin | >16 | 16 |
| Dienestrol | >16 | 16 |
| Diethylstilbestrol | >16 | 16 |
| Doxycycline | >16 | 16 |
| Hexestrol | >16 | 16 |
| Lobeline | >16 | 16 |
| Lovastatin | >16 | 16 |
| Meclocycline | >16 | 16 |
| Nisoldipine | >16 | 16 |
| Norgestimate | >16 | 16 |
| Perhexiline | >16 | 16 |
| Phenelzine | >16 | 16 |
| Quinestrol | >16 | 16 |
| Simvastatin | >16 | 8 |
| Sulfamonomethoxine | >16 | 16 |
| Sulfaquinoxaline | >16 | 16 |
| Tamoxifen | >16 | 16 |
| Tolfenamic acid | >16 | 16 |
To the best of our knowledge, the primary screen revealed novel fluconazole chemosensitizing agents that have never been reported before, such as aripiprazole, perhexiline, phenelzine, quinestrol, dienestrol, hexestrol, norgestimate, meclocycline, tolfenamic acid, and sulfaquinoxaline. In addition and as expected, the primary screen identified several drugs with known fluconazole chemosensitizing activities, such as the cholesterol-lowering agents; simvastatin, atorvastatin, and lovastatin, artemisinin, amiodarone, sulfamethoxazole, doxycycline, and the calcineurin inhibitors nisoldipine and tamoxifen19–25.
Synergistic interactions between fluconazole and the antihyperlipidemic statin drugs against C. albicans NR-29448
The observation that simvastatin demonstrated a significant fluconazole chemosensitizing activity was encouraging to assess the activity of other pharmacologically related antihyperlipidemic statin drugs. Microdilution checkerboard assays were used to assess the interactions between eight statin derivatives and fluconazole against C. albicans NR-29448 strain. Interestingly pitavastatin, whose activity as a fluconazole chemosensitizing agent has not been previously reported, displayed the most potent fluconazole chemosensitizing activity (ΣFICI = 0.05) and was superior to all other tested statin drugs (Table 2). The pitavastatin’s fluconazole chemosensitizing activity was even more superior than the other pharmacologically-related statin drugs, which were reported to have fluconazole-chemosensitizing activities26,27. Pitavastatin, at 0.25 µg/ml, was able to reduce the MIC of fluconazole by 64-fold against C. albicans NR-29448. Except for pravastatin, all other statin drugs demonstrated synergistic interactions with fluconazole against the tested strain (ΣFICI = 0.13–0.26), Table 2. Of note, pitavastatin was shown to reach a peak blood concentration of 0.23 µg/ml following a single oral dose of 4 mg, suggesting that its indication as a fluconazole adjuvant is rationally conceivable28. Due to its potent fluconazole chemosensitizing activity and its potential clinical importance, pitavastatin was selected for subsequent experimental investigation.
Table 2.
Effect of different statin drugs on the antifungal activity of fluconazole against C. albicans NR-29448.
| Test Agent | MIC (µM) | ΣFICIa | Interaction | |||
|---|---|---|---|---|---|---|
| Fluconazole | Test Agent | |||||
| Alone | Combined | Alone | Combined | |||
| Atorvastatin | 256 | 4 | 128 | 16 | 0.14 | SYN |
| Fluvastatin | 256 | 4 | 64 | 8 | 0.14 | SYN |
| Lovastatin | 256 | 2 | 128 | 16 | 0.13 | SYN |
| Mevastatin | 256 | 4 | 256 | 32 | 0.14 | SYN |
| Pitavastatin | 256 | 4 | 8 | 0.25 | 0.05 | SYN |
| Pravastatin | 256 | 2 | 256 | 256 | 1.01 | IND |
| Rosuvastatin | 256 | 2 | 128 | 32 | 0.26 | SYN |
| Simvastatin | 256 | 4 | 64 | 8 | 0.14 | SYN |
aΣFICI (fractional inhibitory concentration index) is used to measure the interaction between the tested combinations. ΣFICI interpretation corresponded to the following definitions: synergism (SYN), ΣFICI ≤ 0.5; additivity (ADD), ΣFICI > 0.5 and ≤1; and indifference (IND), ΣFICI > 1 and ≤4.
Pitavastatin displays a potent, broad-spectrum azole chemosensitizing activity against different Candida species
After identifying pitavastatin as the most potent azole-chemosensitizing agent against C. albicans NR-29448, we examined whether such activity would extend to other strains and species of Candida. As shown in Table 3, pitavastatin exhibited a broad-spectrum synergistic relationship with fluconazole against 16 out of 18 tested Candida strains (~89%), resulting in significant reductions in the fluconazole’s MIC values (4–64 folds). Notably, the pitavastatin-fluconazole combination displayed variable activities against strains displaying different azole resistance mechanisms. Pitavastatin interacted synergistically with fluconazole against C. albicans TWO7243 strain, which is known to exhibit increased mRNA levels of ERG11, CDR1 (an ABC-type transporter) and MDR1 (an MFS-type transporter). Similarly, pitavastatin interacted synergistically with fluconazole against C. albicans SC-TAC1G980E strain, which has a gain of function mutation in TAC1, a positive transcription regulator for the ABC (ATP Binding Cassette) membrane transporters29–31. However, the pitavastatin-fluconazole combination failed to display similar interactions against strain TWO7241, which exhibits increased mRNA levels of both ERG11 and MDR1, and strain SC-MRR1P683S which has a gain of function mutation in MRR1, a positive transcription regulator for the MFS (Major Facilitator Superfamily) membrane transporters29–31. These results indicate that the azole chemosensitizing activity of pitavastatin is dictated by the underlying azole resistance mechanisms and suggest a possible role for the membrane efflux transporters.
Table 3.
Effect of the pitavastatin-fluconazole (FLC) combination against different Candida strains.
| Candida Strains | MIC (µg/ml) | ΣFICIa | Interaction | |||
|---|---|---|---|---|---|---|
| FLC | Pitavastatin | |||||
| Alone | Combined | Alone | Combined | |||
| C. albicans SC5314 | 0.125 | 0.0312 | 4 | 1 | 0.50 | SYN |
| C. albicans NR-29448 | 256 | 4 | 8 | 0.25 | 0.05 | SYN |
| C. albicans NR-29437 | 128 | 4 | 8 | 0.5 | 0.09 | SYN |
| C. albicans ATCC 26790 | 128 | 4 | 8 | 0.5 | 0.09 | SYN |
| C. albicans ATCC MYA-573 | 128 | 4 | 16 | 4 | 0.28 | SYN |
| C. albicans TWO7241 | 32 | 16 | 8 | 4 | 1.00 | IND |
| C. albicans TWO7243 | 64 | 16 | 64 | 4 | 0.31 | SYN |
| C. albicans SC-TAC1G980E | 2 | 0.25 | 32 | 4 | 0.25 | SYN |
| C. albicans SC-MRR1P683S | 2 | 1 | 8 | 2 | 0.75 | ADD |
| C. glabrata ATCC 66032 | 4 | 1 | 64 | 16 | 0.50 | SYN |
|
C. glabrata ATCC MYA-2950 |
8 | 1 | 64 | 16 | 0.38 | SYN |
| C. glabrata ATCC 2001 | 4 | 0.5 | 64 | 16 | 0.38 | SYN |
| C. glabrata HM-1123 | 4 | 1 | 64 | 16 | 0.50 | SYN |
| C. auris 385 | 256 | 32 | 128 | 32 | 0.38 | SYN |
| C. auris 386 | 256 | 64 | 128 | 16 | 0.38 | SYN |
| C. auris 388 | 256 | 64 | 64 | 16 | 0.50 | SYN |
| C. auris 389 | 256 | 32 | 64 | 8 | 0.25 | SYN |
| C. auris 390 | 256 | 16 | 64 | 16 | 0.31 | SYN |
aΣFICI (fractional inhibitory concentration index) is used to measure the interaction between the tested combinations. ΣFICI interpretation corresponded to the following definitions: synergism (SYN), ΣFICI ≤ 0.5; additivity (ADD), ΣFICI > 0.5 and ≤1; and indifference (IND), ΣFICI > 1 and ≤4.
Pitavastatin was also evaluated in combination with other azole antifungals including voriconazole and itraconazole. Similar to its effect with fluconazole, pitavastatin possessed broad-spectrum synergistic interactions with voriconazole against 16 strains of C. albicans, C. glabrata, and C. auris (ΣFICI ranged from 0.15 to 0.50, Supplementary Table 2). However, pitavastatin displayed a more narrow-spectrum synergistic relationship with itraconazole, as only 9 out of 18 of the tested Candida strains (50%) responded to the pitavastatin-itraconazole combination (Supplementary Table 3).
Of note, although pitavastatin was able to demonstrate broad-spectrum synergistic interactions with fluconazole, these interactions were not sufficient to restore the antifungal activity of fluconazole in several fluconazole-resistant isolates. Considering the current resistance breakpoints for azole drugs, two C. albicans and four C. auris isolates maintained their resistance profiles to fluconazole32–37. However, combining pitavastatin with either voriconazole or itraconazole displayed better outcomes against isolates displaying a lower susceptibility to either agent, suggesting a potential clinical significance for treating invasive infections caused by voriconazole (or itraconazole) resistant isolates.
The pitavastatin-fluconazole combination significantly reduces the biofilm-forming abilities of Candida species
Candida species are known for their remarkable capabilities of forming robust adherent structures (i.e., biofilms) on surfaces of different abiotic surfaces, such as catheters, and medical implants38–40. Biofilms limit the penetration of antifungal drugs and can contribute to treatment failure and chronic infections41. Fungal cells residing in biofilms have been reported to have increased expression of efflux genes42,43. Biofilms were also reported to trigger the formation of Candida persisters, which can tolerate very high doses of the antifungal agents44. Collectively, these factors contribute significantly to the remarkable ability of Candida’s biofilms to resist the effect of antifungal drugs, especially azoles45,46. Thus, there is a pressing necessity for novel antifungal adjuvants with activity against Candida biofilms. Here, we investigated whether the synergistic relationship between azole drugs and pitavastatin could interfere with the biofilm-forming ability of Candida. Compared to single treatments with either fluconazole or pitavastatin, incubating the tested Candida species with pitavastatin (at 0.5 × MIC) in the presence of a subinhibitory concentration of fluconazole (2 µg/ml) resulted in a significant reduction in the biofilm-forming abilities of C. albicans NR-29448 (by ~92%, Fig. 2a), C. glabrata HM-1123 (by ~70%, Fig. 2b), and C. auris 385 (by ~41%, Fig. 2c). These findings indicate potent inhibitory activities of the pitavastatin-fluconazole combination against different Candida biofilms. However, when tested against preformed biofilms, the pitavastatin-fluconazole combination failed to disrupt mature biofilms suggesting poor penetrating abilities of the tested combination (data not shown).
Figure 2.
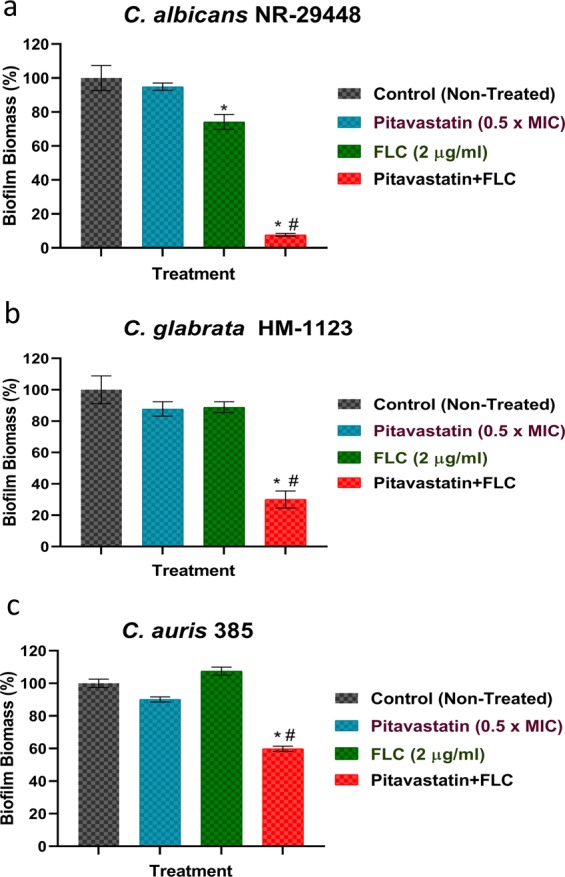
Anti-biofilm activity of the pitavastatin-fluconazole combination. The effect of the pitavastatin-fluconazole (FLC) combination was tested on the biofilm-forming ability of (a) C. albicans NR-29448, (b) C. glabrata HM-1123, and (c) C. auris 385, respectively. Fresh overnight cultures of the tested Candida strains were diluted 1:100 in RPMI 1640 medium. Cells were treated with pitavastatin (at 0.5 × MIC), fluconazole (FLC) at 2 µg/ml, or a combination of the two drugs, at the indicated concentration. Candida strains were incubated at 35 °C for 24 hours before discarding the non-adherent cells and staining the formed biofilms with 0.01% crystal violet. The absorbance of crystal violet-stained biofilms was measured at OD595. *Indicates a significant difference between each treatment compared to the non-treated control. Whereas # indicates a significant difference between the tested pitavastatin-fluconazole combination relative to the single treatment with either fluconazole or pitavastatin. The statistical significance was considered for P < 0.05 as determined by one-way ANOVA with posthoc Dunnet’s test for multiple comparisons.
Pitavastatin significantly interferes with the ABC-mediated efflux activity of Candida
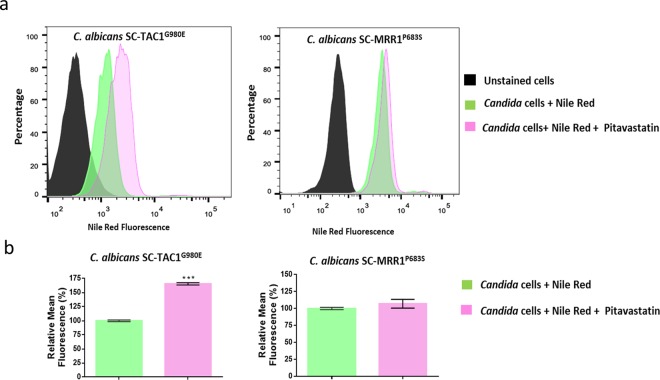
Notably, the azole chemosensitizing activity of statins has been attributed to their ability to interfere with the fungal ergosterol biosynthesis26,47. However, this mechanism does not explain their inconsistent effects against the efflux-activated strains. As shown in Table 3, pitavastatin demonstrated significant fluconazole chemosensitizing activity against strains whose efflux mechanisms involve a significant role of the ABC-type transporters (SC-TAC1G980E and TWO7243) but failed to do so against strains whose efflux mechanisms are solely mediated by the MFS-type transporters (SC-MRR1P683S and TWO7241). Additionally, we noticed a significant reduction in the intrinsic antifungal activity of pitavastatin (by 4–8 fold) against C. albicans strains exhibiting increased mRNA levels of ABC-type efflux transporters (Table 3). Moreover, Candida species that are known for their hyperactive ABC-transporters such as C. glabrata and C. auris displayed significantly reduced susceptibility to pitavastatin as compared to the wild type C. albicans strain48–50. These observations suggest a high affinity of pitavastatin towards the fungal ABC efflux pumps. Therefore, we postulated that pitavastatin may enhance the antifungal activity of fluconazole through a competitive interference with Candida’s ABC-type membrane transporters. To investigate this premise, we first used nile red efflux assay. Nile red is a known substrate for the two major membrane transporters (ABC and MFS) which have been reported as major contributors to azole resistance in Candida51–53. Therefore, nile red can be used efficiently as a non-specific reporter dye to measure drug effects on the efflux activities of C. albicans strains, regardless of their efflux mechanisms. As shown in Fig. 3a, pitavastatin (at 0.25 × MIC) significantly maintained a high level of nile red fluorescence intensity in the ABC efflux-activated strain (SC-TAC1G980E), compared to the non-treated control. However, the nile red fluorescence intensity was greatly diminished in the MFS efflux-activated strain (SC-MRR1P683S), and the signal was comparable to the non-treated control (Fig. 3b). These results suggest a significant ability of pitavastatin to interfere specifically with the ABC efflux-mediated activities in Candida. These effects were confirmed using flow cytometry analysis. C. albicans SC-TAC1G980E exhibited a significant increase in the nile red fluorescence intensity following exposure to pitavastatin at 0.25 × MIC. However, C. albicans SC-MRR1P683S was indifferent to the pitavastatin effect, and the nile red fluorescence intensity was comparable to the non-treated control (Fig. 4a). Pitavastatin treatment resulted in a significant increase (65%) in the mean fluorescent intensity only in the ABC-efflux activated strain (SC-TAC1G980E), as compared to the non-treated control (Fig. 4b), which supports our previous observation.
Figure 3.
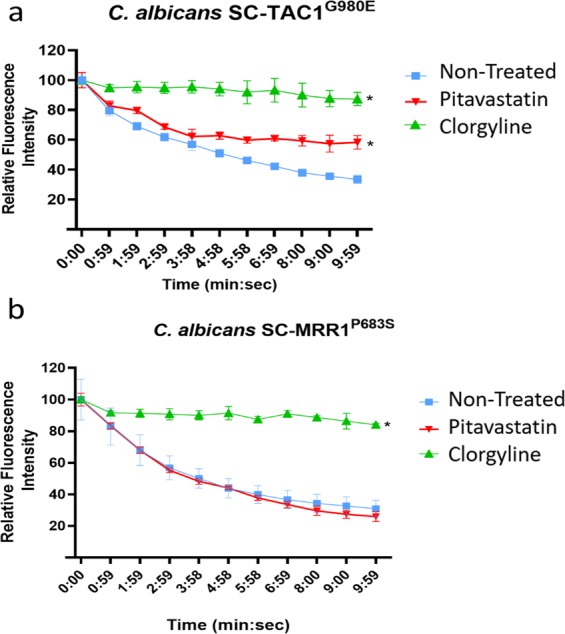
Effect of pitavastatin on Nile red efflux by different efflux hyperactive Candida strains. The effect of pitavastatin on nile red efflux in (a) the ABC efflux-activated strain SC-TAC1G980E, and (b) the MFS efflux-activated strain SC-MRR1P683S. Starved Candida cells were loaded with the fluorescent dye, nile red (7.5 µM). Pitavastatin (0.25 × MIC) and the positive control clorgyline (5 µg/ml) were added to the stained cells. Nile red efflux was initiated by adding glucose (final concentration 10 mM). The nile red fluorescence intensity was monitored over 10 minutes and is expressed as the percentage of change in the fluorescence intensity. *Indicates a statistically significant difference from the non-treated control (P < 0.05, as determined by multiple t-tests using Holm-Sidak statistical method for multiple comparisons).
Figure 4.
Flow cytometry analysis of nile red efflux from two C. albicans strains treated with pitavastatin. (a) Histograms represent overlaid flow cytometry data as a percentage of unstained or nile red-stained C. albicans strains (SC-TAC1G980E and SC-MRR1P683S), following treatment with either PBS or pitavastatin. Starved Candida cells were stained with nile red, treated with pitavastatin (0.25 × MIC), exposed to glucose (10 mM) for 10 minutes to initiate the efflux, and then analyzed with flow cytometry. The shift in the mean fluorescence following pitavastatin treatment indicates increased nile red staining and hence interference with the nile red efflux capacity of the test strain. (b) Graphs of mean fluorescence intensities normalized to the average mean fluorescence intensity of non-treated samples (Candida cells + Nile Red). The means ± S.D. from two independent replicates are shown. Asterisks indicate statistically significant (P < 0.05) pairwise comparisons between the pitavastatin treated and non-treated samples.
These findings were further confirmed using rhodamine 6G efflux assay. Rhodamine 6G has been shown to display a substrate specificity to the ABC membrane transporters54. Similarly, pitavastatin at a subinhibitory concentration (0.25 × MIC) significantly reduced the percentage of effluxed rhodamine in the ABC- efflux activated strain C. albicans SC-TAC1G980E, as compared to the non-treated control (Supplementary Fig. 1). Once again, this result indicates that the azole chemosensitization activities displayed by pitavastatin can be attributed, at least in part, to their ability to interfere with the function of Candida’s ABC transporters.
Efficacy of the pitavastatin-fluconazole combination in Caenorhabditis elegans infection model
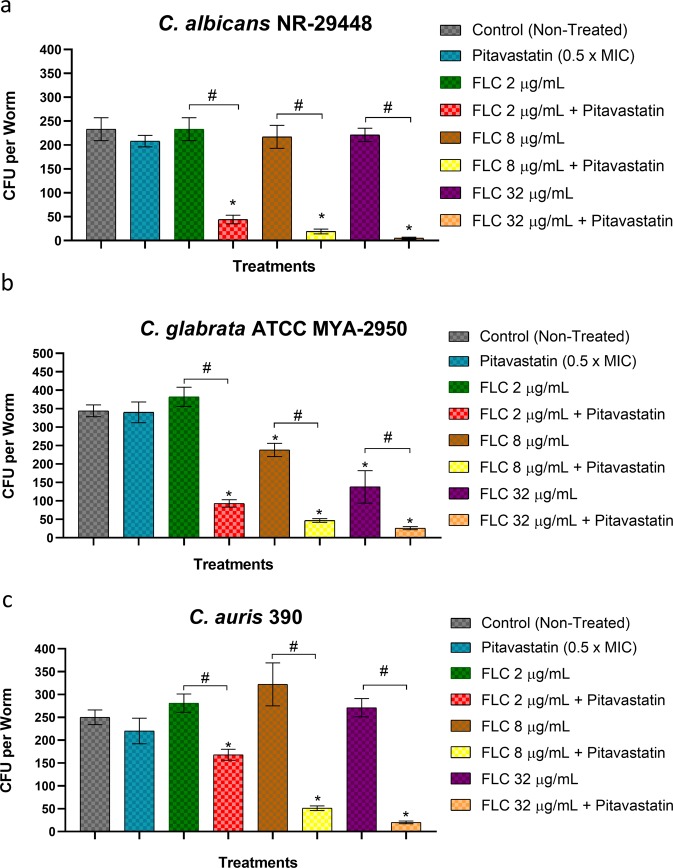
In the field of antimicrobial drug discovery, it is quite frequent to notice that several promising antimicrobial compounds fail when assessed in vivo in animal models, despite potent in vitro activities. Given the in-vitro promising activity of the pitavastatin-fluconazole combination, together with its potent antibiofilm activity against different Candida species, it was necessary to assess its activity in vivo. C. elegans is a satisfactory animal model for the initial assessment of promising antimicrobial agents prior to their evaluation in mammalian models. In order to validate our in vitro results, C. elegans was utilized as an animal model to investigate the fluconazole chemosensitizing activity of pitavastatin. As shown in Fig. 5, treating C. elegans infected nematodes with pitavastatin (at 0.5 x MIC) combined with three different concentrations of fluconazole (2, 8, and 32 µg/ml) displayed variable outcomes depending on the fluconazole concentration and the infectious strain. Compared to the untreated control which accumulated 233 ± 21 CFU/worm, pitavastatin-fluconazole combinations significantly reduced the mean fungal CFU burdens of C. albicans NR-29448 in the infected nematodes by ~82–96% (Fig. 5a). Likewise, pitavastatin-fluconazole combinations reduced the fungal burdens of C. glabrata ATCC MYA-2950 by ~84–93% compared to the untreated control which accumulated 344 ± 19 CFU/worm (Fig. 5b). Against C. auris 390, pitavastatin-fluconazole combinations reduced the CFU burdens in the infected nematodes by 14–92% compared to the untreated control which accumulated 250 ± 25 CFU/ml (Fig. 5c). As expected, single fluconazole treatments failed to reduce the CFU burdens in nematodes infected with the fluconazole-resistant isolates (C. albicans NR-29448 or C. auris 390). However, single treatments with fluconazole at 8 or 32 µg/ml were able to reduce CFU burdens of C. glabrata ATCC MYA-2950 by only 26 and 57% respectively, though more potent activities were attained with combination treatments as shown earlier. Altogether, these results are encouraging for future evaluation of the pitavastatin-fluconazole combination in higher animal models.
Figure 5.
Efficacy of the pitavastatin-fluconazole combination in a Caenorhabditis elegans infection model. C. elegans strain AU37 genotype [glp-4(bn2) I; sek-1(km4) X], was co-incubated with cell suspensions of (a) C. albicans NR-29448, (b) C. glabrata ATCC MYA-2950, or (c) C. auris 390 using an inoculum size of ~5 × 107 CFU/ml for 3 hours at room temperature. Infected nematodes were washed with PBS and then treated with the pitavastatin-fluconazole combination at the respective concentration. Treatment with PBS, pitavastatin alone, or fluconazole alone served as controls. After 24 hours of treatment, worms were lysed to determine the fungal burden (CFU/worm) after treatment. *Indicates a significant difference between each treatment compared to the non-treated control. Whereas # indicates significant differences between the tested pitavastatin-fluconazole combinations relative to the single treatment with the respective fluconazole concentration. The statistical significance was considered for P < 0.05 as determined by one-way ANOVA with posthoc Dunnet’s test for multiple comparisons.
Conclusion
The present study characterized pitavastatin as a promising agent for sensitizing azole-resistant Candida species to the antifungal effect of azoles. Pitavastatin, exhibited broad-spectrum synergistic interactions with fluconazole against a variety of clinically-relevant Candida species, including emerging multi-drug resistant C. auris isolates. Moreover, the pitavastatin-fluconazole combination significantly interfered with Candida’s biofilm-forming abilities. Additionally, the pitavastatin-fluconazole combination significantly reduced Candida’s CFU burdens in infected C. elegans, suggesting potential clinical importance. Finally, the mechanism of synergy displayed by pitavastatin and fluconazole embroils, at least in part, significant interference with Candida’s efflux machinery. Further in vivo studies in higher animals are required to assess the potential of pitavastatin to be repurposed as a promising fluconazole adjuvant for controlling invasive Candida infections in humans.
Materials and Methods
Fungal strains and culture reagents
Fungal strains used in this study are listed in Supplementary Table 4. C. albicans clinical isolates TWO7241 and TWO7243 were obtained from professor Theodor White (UMKC). SC5314 mutant derivatives SC-MRR1P683S and SC-TAC1G980E, containing gain-of-function alleles (MRR1P683S and TAC1G980E), were obtained from professor David Rogers (University of Tennessee Health Science Center). RPMI 1640 powder with glutamine, but without NaHCO3, was purchased from Thermo Fisher Scientific (Waltham, MA). 3-(N-Morpholino) propanesulfonic acid (MOPS) was obtained from Sigma Aldrich (St. Louis, MO). YPD broth medium and YPD agar were obtained from Becton, Dickinson Company (Franklin Lakes, NJ).
Chemicals and drugs
The Pharmakon 1600 drug library was purchased from MicroSource Discovery Systems, Inc. (Gaylordsville, CT). Compounds were delivered in microplates (10 mM, dissolved in DMSO) and stored at −80 °C until use. Nile red, voriconazole, and itraconazole were obtained from TCI America (Portland, OR). Pitavastatin was obtained from Ark Pharm (Arlington Heights, IL). Fluconazole was obtained from Fisher Scientific (Pittsburgh, PA). Gentamicin sulfate was purchased from Chem-Impex International INC. (Wood Dale, IL).
Screening of Pharmakon library and structurally-related compounds
The Pharmakon 1600 drug library was screened against C. albicans NR-29448, a strain that displayed high-level resistance to several azole antifungal drugs. Briefly, C. albicans NR-29448 was diluted to approximately 0.5–2.5 × 103 cells/ml in RPMI 1640 medium buffered with 0.165 M MOPS reagent. An aliquot (100 µl) of the fungal suspension was transferred to the wells of a round-bottomed 96-well microtitre plate containing 16 µM of each drug. The plates were then incubated for 24 hours at 35 °C. Drugs that only inhibited the growth of C. albicans in the presence of fluconazole were identified as “positive hits”.
Microdilution checkerboard assays
The interactions between the identified hits and different azole antifungal drugs were assessed using broth microdilution checkerboard assays, as previously reported55–57. ΣFICI (fractional inhibitory concentration index) is used to assess the potential interactions between the tested drug combinations. ΣFICI interpretation corresponded to the following definitions: synergism (SYN), ΣFICI ≤ 0.5; additivity (ADD), ΣFICI > 0.5 and ≤ 1; and indifference, ΣFICI > 1 and ≤ 458.
Biofilm inhibition assay
Three Candida species, C. albicans NR-29448, C. glabrata HM-1123, and C. auris 385 demonstrated a prominent ability to form robust adherent biofilms. As such, these strains were used to study the antibiofilm activity of the pitavastatin-fluconazole combination. The microtiter biofilm formation assay using crystal violet was used, as previously described4,21. Briefly, overnight cultures of the tested Candida strains, grown in YPD broth, were diluted in RPMI 1640 medium to approximately 1 × 105 CFU/ml. Then 100 µl aliquots of each suspension were transferred to wells of tissue-culture treated polystyrene 96-well plates. Pitavastatin (at 0.5 × MIC) was added either individually or in combination with fluconazole (2 µg/ml) and the plates were then incubated for 24 h at 35 °C. Following incubations, adherent biofilms were then rinsed twice with phosphate-buffered saline (PBS) and left to dry at room temperature. Air-dried biofilms were stained with crystal violet (0.01%). Stained biofilms were rinsed thrice with PBS and then air-dried. The resultant biofilm biomasses were quantified by dissolving the crystal violet-stained biofilms in absolute ethanol before recording absorbance values (OD595).
Nile Red efflux assay and flow cytometry
Nile red efflux assay was performed following a previously reported protocol59–61. Briefly, exponential phase Candida cells were harvested by centrifugation (3,000 × g, 5 minutes), washed thrice with PBS, and incubated for an additional 2 hours at 35 °C with shaking (200 rpm). Cells were incubated overnight on ice, then resuspended at a concentration of ~1 × 107 cells per ml in HEPES-NaOH (50 mM; pH 7.0) containing 7.5 mM nile red and incubated at 35 °C for 30 minutes. Stained cells were washed three times with cold HEPES-NaOH (50 mM; pH 7.0). Cell suspensions were transferred onto opaque 96-well plates containing two-fold serial dilutions of the test agents. Glucose at final concentration 10 mM was used to initiate the nile red efflux. Detection of nile red fluorescence intensity was commenced about 15 seconds after glucose addition (T0) and then in one-minute intervals for 10 minutes. Nile red fluorescence intensity was measured at an excitation wavelength of 485/9 and an emission wavelength of 528/15 using the SpectraMax i3x microplate reader (Molecular Devices, CA, USA). For flow cytometric analysis, pitavastatin (at 0.25 × MIC) was added to nile red-loaded cells as previously described, then glucose (at final concentration 10 mM) was used to initiate the nile red efflux. After 10 minutes of adding glucose, cells were fixed in 2% paraformaldehyde and were examined in a Canto II flow cytometer (BD Bioscience, San Jose, CA), following a previously reported protocol62. Data were analyzed using FlowJo software v10 (Tree Star, Ashland, OR).
Rhodamine Rh6G efflux assay
Rhodamine 6G efflux assay was conducted following a previously reported protocol63. Briefly, exponential growth phase Candida cells (SC-TAC1G980E) were harvested as described earlier, washed thrice with PBS, and incubated for additional 2 hours at 35 °C with shaking (200 rpm) to induce starvation. The cells were then resuspended at a concentration of ~1 × 107 cells per ml in HEPES-NaOH (50 mM; pH 7.0) buffer containing rhodamine 6G (10 mM) and 2-deoxyglucose (5 mM). Cells were incubated with shaking for 90 minutes at 30 °C, to permit rhodamine accumulation under energy-depleting conditions. Rhodamine-stained cells were harvested and washed at least five times with HEPES-NaOH to remove extracellular rhodamine. Pitavastatin at 0.25 × MIC, clorgyline (5 µg/ml), or the vehicle (1% DMSO) was added to the cells and incubated for 5 minutes at 30 °C. Rhodamine efflux was induced by glucose addition at a final concentration of 10 mM. After 10 minutes of adding glucose, cells were harvested by centrifugation, and 100 µl aliquots of cell supernatants were transferred to 96-well plates for detecting the amount of effluxed rhodamine. The rhodamine fluorescence intensity was measured by SpectraMax i3x microplate reader (Molecular Devices, CA, USA), using 529 nm and 553 nm as excitation and emission wavelengths, respectively.
Caenorhabditis elegans fungal infection model
To examine the in vivo efficacy of pitavastatin in enhancing the activity of fluconazole against azole-resistant C. albicans, we used the C. elegans animal model following previously reported guidelines13,21 C. albicans NR-29448, C. glabrata ATCC MYA-2950, and C. auris 390 displayed enhanced susceptibility to the effect of the pitavastatin-fluconazole combination and were selected for this experiment. Briefly, L4 stage worms [strain AU37 genotype glp-4(bn2) I; sek-1(km4) X] were infected by co-incubating them with approximately 5 × 107 CFU/ml of Candida suspensions for 3 h at room temperature. After infection, C. elegans nematodes were washed five times with M9 buffer and transferred into microcentrifuge tubes (20 worms per tube). Infected nematodes were treated with combinations of pitavastatin (at 0.5 × MIC) plus fluconazole at three different concentrations (2, 8, or 32 µg/ml), and incubated for 24 hours at 25 °C. Treatment with either PBS, pitavastatin or fluconazole at the same concentrations served as controls. Posttreatment, worms were examined microscopically to evaluate morphological changes and ensure viability. Worms were washed with M9 buffer five times and then disrupted by vigorous vortexing with silicon carbide particles. The resulting Candida suspensions were serially diluted and transferred to YPD agar plates containing gentamicin (100 g/ml). Plates were incubated for 48 hours at 35 °C before the viable CFU per worm was determined.
Statistical analyses
All experiments were performed in triplicates and repeated at least three times. Statistical analyses were performed using GraphPad Prism 6.0 (Graph Pad Software, La Jolla, CA, USA). P-values were calculated using multiple t-tests and one-way ANOVA, and P-values < 0.05 were considered significant. Data are presented as means ± standard deviation.
Supplementary information
Acknowledgements
The fungal isolates used in this study were generously provided by BEI Resources and the US Centers for Disease Control and Prevention (CDC). The authors would like to thank Professor Theodor White (University of Missouri-Kansas City) for kindly providing C. albicans clinical isolates TWO7241 and TWO7243. Also, the authors would like to thank Professor David Rogers (University of Tennessee Health Science Center), for graciously providing C. albicans SC5314 mutant constructs (SC-TAC1G980E and SC-MRR1P683S) used in the efflux experiments. Finally, the authors would like to thank Dr. Haroon Mohammad (Purdue University) for language editing and proofreading the manuscript.
Author contributions
H.E., E.S. and X.L. performed the experiments. H.E. and M.S. conceived and designed the study. H.E, T.H., A.M., and M.S. analyzed the data and interpreted the results. H.E. and M.S. wrote the manuscript. All authors reviewed and discussed the results.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Abdelrahman S. Mayhoub, Email: amayhoub@azhar.edu.eg
Mohamed N. Seleem, Email: mseleem@purdue.edu
Supplementary information
is available for this paper at 10.1038/s41598-020-64571-7.
References
- 1.Nett, J. E. Special Issue: Candida and Candidiasis. J Fungi(Basel)4 (2018). [DOI] [PMC free article] [PubMed]
- 2.Sterkel, A., Bateman, A., Valley, A. & Warshauer, D. Viability of Candida auris and Other Candida Species after Various Matrix-Assisted Laser Desorption Ionization-Time of Flight (MALDI-TOF) Mass Spectrometry-Based Extraction Protocols. J Clin Microbiol56 (2018). [DOI] [PMC free article] [PubMed]
- 3.Gow NAR, Yadav B. Microbe Profile: Candida albicans: a shape-changing, opportunistic pathogenic fungus of humans. Microbiology. 2017;163:1145–1147. doi: 10.1099/mic.0.000499. [DOI] [PubMed] [Google Scholar]
- 4.Mohammad H, Eldesouky HE, Hazbun T, Mayhoub AS, Seleem MN. Identification of a Phenylthiazole Small Molecule with Dual Antifungal and Antibiofilm Activity Against Candida albicans and Candida auris. Sci Rep. 2019;9:18941. doi: 10.1038/s41598-019-55379-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lamoth F, Lockhart SR, Berkow EL, Calandra T. Changes in the epidemiological landscape of invasive candidiasis. J Antimicrob Chemother. 2018;73:i4–i13. doi: 10.1093/jac/dkx444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Jabra-Rizk MA, et al. Candida albicans Pathogenesis: Fitting within the Host-Microbe Damage Response Framework. Infection and Immunity. 2016;84:2724–2739. doi: 10.1128/IAI.00469-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Cleveland AA, et al. Declining incidence of candidemia and the shifting epidemiology of Candida resistance in two US metropolitan areas, 2008-2013: results from population-based surveillance. PLoS One. 2015;10:e0120452. doi: 10.1371/journal.pone.0120452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Pfaller MA, Jones RN, Castanheira M. Regional data analysis of Candida non-albicans strains collected in United States medical sites over a 6-year period, 2006-2011. Mycoses. 2014;57:602–11. doi: 10.1111/myc.12206. [DOI] [PubMed] [Google Scholar]
- 9.Guinea J. Global trends in the distribution of Candida species causing candidemia. Clin Microbiol Infect. 2014;20(Suppl 6):5–10. doi: 10.1111/1469-0691.12539. [DOI] [PubMed] [Google Scholar]
- 10.Nett JE. Candida auris: An emerging pathogen “incognito”? PLoS Pathog. 2019;15:e1007638. doi: 10.1371/journal.ppat.1007638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lone SA, Ahmad A. Candida auris-the growing menace to global health. Mycoses. 2019;62:620–637. doi: 10.1111/myc.12904. [DOI] [PubMed] [Google Scholar]
- 12.Centers for Disease Control and Prevention (CDC). Department of Health and Human Services, Antibiotic Resistance Threats in the United States, Atlanta, GA: U.S (2019).
- 13.Mohammad H, et al. Discovery of a Novel Dibromoquinoline Compound Exhibiting Potent Antifungal and Antivirulence Activity That Targets Metal Ion Homeostasis. ACS Infect Dis. 2018;4:403–414. doi: 10.1021/acsinfecdis.7b00215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Whaley SG, et al. Azole Antifungal Resistance in Candida albicans and Emerging Non-albicans Candida Species. Front Microbiol. 2016;7:2173. doi: 10.3389/fmicb.2016.02173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Won EJ, et al. Antifungal susceptibilities of bloodstream isolates of Candida species from nine hospitals in Korea: application of new antifungal breakpoints and relationship to antifungal usage. PLoS One. 2015;10:e0118770. doi: 10.1371/journal.pone.0118770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Song JL, Harry JB, Eastman RT, Oliver BG, White TC. The Candida albicans lanosterol 14-alpha-demethylase (ERG11) gene promoter is maximally induced after prolonged growth with antifungal drugs. Antimicrobial Agents and Chemotherapy. 2004;48:1136–1144. doi: 10.1128/AAC.48.4.1136-1144.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ghannoum MA, Rice LB. Antifungal agents: Mode of action, mechanisms of resistance, and correlation of these mechanisms with bacterial resistance. Clinical Microbiology Reviews. 1999;12:501–+. doi: 10.1128/CMR.12.4.501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Pfaller MA, et al. Trends in species distribution and susceptibility to fluconazole among blood stream isolates of Candida species in the United States. Diagnostic Microbiology and Infectious Disease. 1999;33:217–222. doi: 10.1016/S0732-8893(98)00160-6. [DOI] [PubMed] [Google Scholar]
- 19.Liu S, et al. Synergistic Effect of Fluconazole and Calcium Channel Blockers against Resistant Candida albicans. PLoS One. 2016;11:e0150859. doi: 10.1371/journal.pone.0150859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Spitzer M, et al. Cross-species discovery of syncretic drug combinations that potentiate the antifungal fluconazole. Mol Syst Biol. 2011;7:499. doi: 10.1038/msb.2011.31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Eldesouky, H. E., Mayhoub, A., Hazbun, T. R. & Seleem, M. N. Reversal of Azole Resistance in Candida albicans by Sulfa Antibacterial Drugs. Antimicrob Agents Chemother62 (2018). [DOI] [PMC free article] [PubMed]
- 22.Fiori A, Van Dijck P. Potent synergistic effect of doxycycline with fluconazole against Candida albicans is mediated by interference with iron homeostasis. Antimicrob Agents Chemother. 2012;56:3785–96. doi: 10.1128/AAC.06017-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.De Cremer K, et al. Artemisinins, new miconazole potentiators resulting in increased activity against Candida albicans biofilms. Antimicrob Agents Chemother. 2015;59:421–6. doi: 10.1128/AAC.04229-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Gamarra S, et al. Mechanism of the synergistic effect of amiodarone and fluconazole in Candida albicans. Antimicrob Agents Chemother. 2010;54:1753–61. doi: 10.1128/AAC.01728-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Cabral ME, Figueroa LIC, Farina JI. Synergistic antifungal activity of statin-azole associations as witnessed by Saccharomyces cerevisiae- and Candida utilis-bioassays and ergosterol quantification. Revista Iberoamericana De Micologia. 2013;30:31–38. doi: 10.1016/j.riam.2012.09.006. [DOI] [PubMed] [Google Scholar]
- 26.Song JL, Lyons CN, Holleman S, Oliver BG, White TC. Antifungal activity of fluconazole in combination with lovastatin and their effects on gene expression in the ergosterol and prenylation pathways in Candida albicans. Med Mycol. 2003;41:417–25. doi: 10.1080/1369378031000137233. [DOI] [PubMed] [Google Scholar]
- 27.Cabral ME, Figueroa LI, Farina JI. Synergistic antifungal activity of statin-azole associations as witnessed by Saccharomyces cerevisiae- and Candida utilis-bioassays and ergosterol quantification. Rev Iberoam Micol. 2013;30:31–8. doi: 10.1016/j.riam.2012.09.006. [DOI] [PubMed] [Google Scholar]
- 28.Luo Z, Zhang Y, Gu J, Feng P, Wang Y. Pharmacokinetic Properties of Single- and Multiple-Dose Pitavastatin Calcium Tablets in Healthy Chinese Volunteers. Curr Ther Res Clin Exp. 2015;77:52–7. doi: 10.1016/j.curtheres.2015.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.White TC. Increased mRNA levels of ERG16, CDR, and MDR1 correlate with increases in azole resistance in Candida albicans isolates from a patient infected with human immunodeficiency virus. Antimicrob Agents Chemother. 1997;41:1482–7. doi: 10.1128/AAC.41.7.1482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Vasicek EM, Berkow EL, Flowers SA, Barker KS, Rogers PD. UPC2 is universally essential for azole antifungal resistance in Candida albicans. Eukaryot Cell. 2014;13:933–46. doi: 10.1128/EC.00221-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Koselny K, et al. The Celecoxib Derivative AR-12 Has Broad-Spectrum Antifungal Activity In Vitro and Improves the Activity of Fluconazole in a Murine Model of Cryptococcosis. Antimicrob Agents Chemother. 2016;60:7115–7127. doi: 10.1128/AAC.01061-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Khan Z, et al. Increasing prevalence, molecular characterization and antifungal drug susceptibility of serial Candida auris isolates in Kuwait. PLoS One. 2018;13:e0195743. doi: 10.1371/journal.pone.0195743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Centers for Disease Control and Prevention (CDC), Candida auris: Antifungal Susceptibility Testing and Interpretation, https://www.cdc.gov/fungal/candida-auris/c-auris-antifungal.html (2019).
- 34.Arendrup, M.C., Prakash, A., Meletiadis, J., Sharma, C. & Chowdhary, A. Comparison of EUCAST and CLSI Reference Microdilution MICs of Eight Antifungal Compounds for Candida auris and Associated Tentative Epidemiological Cutoff Values. Antimicrob Agents Chemother61 (2017). [DOI] [PMC free article] [PubMed]
- 35.Sears D, Schwartz BS. Candida auris: An emerging multidrug-resistant pathogen. Int J Infect Dis. 2017;63:95–98. doi: 10.1016/j.ijid.2017.08.017. [DOI] [PubMed] [Google Scholar]
- 36.Fothergill AW, Sutton DA, McCarthy DI, Wiederhold NP. Impact of new antifungal breakpoints on antifungal resistance in Candida species. J Clin Microbiol. 2014;52:994–7. doi: 10.1128/JCM.03044-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Yenisehirli G, Bulut N, Yenisehirli A, Bulut Y. In Vitro Susceptibilities of Candida albicans Isolates to Antifungal Agents in Tokat, Turkey. Jundishapur J Microbiol. 2015;8:e28057. doi: 10.5812/jjm.28057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Kojic EM, Darouiche RO. Candida infections of medical devices. Clin Microbiol Rev. 2004;17:255–67. doi: 10.1128/CMR.17.2.255-267.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Nobile CJ, Johnson AD. Candida albicans Biofilms and Human Disease. Annu Rev Microbiol. 2015;69:71–92. doi: 10.1146/annurev-micro-091014-104330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ramage G, Martinez JP, Lopez-Ribot JL. Candida biofilms on implanted biomaterials: a clinically significant problem. FEMS Yeast Res. 2006;6:979–86. doi: 10.1111/j.1567-1364.2006.00117.x. [DOI] [PubMed] [Google Scholar]
- 41.Cavalheiro M, Teixeira MC. Candida Biofilms: Threats, Challenges, and Promising Strategies. Front Med (Lausanne) 2018;5:28. doi: 10.3389/fmed.2018.00028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Kean, R. et al. Transcriptome Assembly and Profiling of Candida auris Reveals Novel Insights into Biofilm-Mediated Resistance. Msphere3 (2018). [DOI] [PMC free article] [PubMed]
- 43.Taff HT, Mitchell KF, Edward JA, Andes DR. Mechanisms of Candida biofilm drug resistance. Future Microbiology. 2013;8:1325–1337. doi: 10.2217/fmb.13.101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Wuyts, J., Van Dijck, P. & Holtappels, M. Fungal persister cells: The basis for recalcitrant infections? Plos Pathogens14 (2018). [DOI] [PMC free article] [PubMed]
- 45.Sardi JCO, Scorzoni L, Bernardi T, Fusco-Almeida AM, Giannini MJSM. Candida species: current epidemiology, pathogenicity, biofilm formation, natural antifungal products and new therapeutic options. Journal of Medical Microbiology. 2013;62:10–24. doi: 10.1099/jmm.0.045054-0. [DOI] [PubMed] [Google Scholar]
- 46.Wu SC, Wang Y, Liu N, Dong GQ, Sheng CQ. Tackling Fungal Resistance by Biofilm Inhibitors. Journal of Medicinal Chemistry. 2017;60:2193–2211. doi: 10.1021/acs.jmedchem.6b01203. [DOI] [PubMed] [Google Scholar]
- 47.Tashiro M, et al. Pravastatin inhibits farnesol production in Candida albicans and improves survival in a mouse model of systemic candidiasis. Med Mycol. 2012;50:353–60. doi: 10.3109/13693786.2011.610037. [DOI] [PubMed] [Google Scholar]
- 48.Rybak, J.M. et al. Abrogation of Triazole Resistance upon Deletion of CDR1 in a Clinical Isolate of Candida auris. Antimicrobial Agents and Chemotherapy63 (2019). [DOI] [PMC free article] [PubMed]
- 49.Sanglard D, Ischer F, Bille J. Role of ATP-binding-cassette transporter genes in high-frequency acquisition of resistance to azole antifungals in Candida glabrata. Antimicrobial Agents and Chemotherapy. 2001;45:1174–1183. doi: 10.1128/AAC.45.4.1174-1183.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Izumikawa K, Kakeya H, Tsai HF, Grimberg B, Bennett JE. Function of Candida glabrata ABC transporter gene, PDH1. Yeast. 2003;20:249–261. doi: 10.1002/yea.962. [DOI] [PubMed] [Google Scholar]
- 51.Prasad R, Rawal MK. Efflux pump proteins in antifungal resistance. Front Pharmacol. 2014;5:202. doi: 10.3389/fphar.2014.00202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Coste AT, Karababa M, Ischer F, Bille J, Sanglard D. TAC1, transcriptional activator of CDR genes, is a new transcription factor involved in the regulation of Candida albicans ABC transporters CDR1 and CDR2. Eukaryot Cell. 2004;3:1639–52. doi: 10.1128/EC.3.6.1639-1652.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Morschhauser J, et al. The transcription factor Mrr1p controls expression of the MDR1 efflux pump and mediates multidrug resistance in Candida albicans. PLoS Pathog. 2007;3:e164. doi: 10.1371/journal.ppat.0030164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Maesaki S, Marichal P, Vanden Bossche H, Sanglard D, Kohno S. Rhodamine 6G efflux for the detection of CDR1-overexpressing azole-resistant Candida albicans strains. Journal of Antimicrobial Chemotherapy. 1999;44:27–31. doi: 10.1093/jac/44.1.27. [DOI] [PubMed] [Google Scholar]
- 55.Gu WR, Guo DM, Zhang LP, Xu DM, Sun SJ. The Synergistic Effect of Azoles and Fluoxetine against Resistant Candida albicans Strains Is Attributed to Attenuating Fungal Virulence. Antimicrobial Agents and Chemotherapy. 2016;60:6179–6188. doi: 10.1128/AAC.03046-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Sun LM, Liao K, Liang S, Yu PH, Wang DY. Synergistic activity of magnolol with azoles and its possible antifungal mechanism against Candida albicans. J Appl Microbiol. 2015;118:826–38. doi: 10.1111/jam.12737. [DOI] [PubMed] [Google Scholar]
- 57.Chen YL, Lehman VN, Averette AF, Perfect JR, Heitman J. Posaconazole exhibits in vitro and in vivo synergistic antifungal activity with caspofungin or FK506 against Candida albicans. PLoS One. 2013;8:e57672. doi: 10.1371/journal.pone.0057672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Silva LV, et al. Milbemycins: more than efflux inhibitors for fungal pathogens. Antimicrob Agents Chemother. 2013;57:873–86. doi: 10.1128/AAC.02040-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Keniya, M. V., Fleischer, E., Klinger, A., Cannon, R. D. & Monk, B. C. Inhibitors of the Candida albicans Major Facilitator Superfamily Transporter Mdr1p Responsible for Fluconazole Resistance. Plos One10 (2015). [DOI] [PMC free article] [PubMed]
- 60.Ivnitski-Steele I, et al. Identification of Nile red as a fluorescent substrate of the Candida albicans ATP-binding cassette transporters Cdr1p and Cdr2p and the major facilitator superfamily transporter Mdr1p. Analytical Biochemistry. 2009;394:87–91. doi: 10.1016/j.ab.2009.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Eldesouky HE, Li X, Abutaleb NS, Mohammad H, Seleem MN. Synergistic interactions of sulfamethoxazole and azole antifungal drugs against emerging multidrug-resistant Candida auris. Int J Antimicrob Agents. 2018;52:754–761. doi: 10.1016/j.ijantimicag.2018.08.016. [DOI] [PubMed] [Google Scholar]
- 62.Greenspan P, Mayer EP, Fowler SD. Nile red: a selective fluorescent stain for intracellular lipid droplets. J Cell Biol. 1985;100:965–73. doi: 10.1083/jcb.100.3.965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Nakamura K, et al. Functional expression of Candida albicans drug efflux pump Cdr1p in a Saccharomyces cerevisiae strain deficient in membrane transporters. Antimicrob Agents Chemother. 2001;45:3366–74. doi: 10.1128/AAC.45.12.3366-3374.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.