Abstract
STUDY QUESTION
What is the recommended management of ovarian stimulation, based on the best available evidence in the literature?
SUMMARY ANSWER
The guideline development group formulated 84 recommendations answering 18 key questions on ovarian stimulation.
WHAT IS KNOWN ALREADY
Ovarian stimulation for IVF/ICSI has been discussed briefly in the National Institute for Health and Care Excellence guideline on fertility problems, and the Royal Australian and New Zealand College of Obstetricians and Gynaecologist has published a statement on ovarian stimulation in assisted reproduction. There are, to our knowledge, no evidence-based guidelines dedicated to the process of ovarian stimulation.
STUDY DESIGN, SIZE, DURATION
The guideline was developed according to the structured methodology for development of ESHRE guidelines. After formulation of key questions by a group of experts, literature searches and assessments were performed. Papers published up to 8 November 2018 and written in English were included. The critical outcomes for this guideline were efficacy in terms of cumulative live birth rate per started cycle or live birth rate per started cycle, as well as safety in terms of the rate of occurrence of moderate and/or severe ovarian hyperstimulation syndrome (OHSS).
PARTICIPANTS/MATERIALS, SETTING, METHODS
Based on the collected evidence, recommendations were formulated and discussed until consensus was reached within the guideline group. A stakeholder review was organized after finalization of the draft. The final version was approved by the guideline group and the ESHRE Executive Committee.
MAIN RESULTS AND THE ROLE OF CHANCE
The guideline provides 84 recommendations: 7 recommendations on pre-stimulation management, 40 recommendations on LH suppression and gonadotrophin stimulation, 11 recommendations on monitoring during ovarian stimulation, 18 recommendations on triggering of final oocyte maturation and luteal support and 8 recommendations on the prevention of OHSS. These include 61 evidence-based recommendations—of which only 21 were formulated as strong recommendations—and 19 good practice points and 4 research-only recommendations. The guideline includes a strong recommendation for the use of either antral follicle count or anti-Müllerian hormone (instead of other ovarian reserve tests) to predict high and poor response to ovarian stimulation. The guideline also includes a strong recommendation for the use of the GnRH antagonist protocol over the GnRH agonist protocols in the general IVF/ICSI population, based on the comparable efficacy and higher safety. For predicted poor responders, GnRH antagonists and GnRH agonists are equally recommended. With regards to hormone pre-treatment and other adjuvant treatments (metformin, growth hormone (GH), testosterone, dehydroepiandrosterone, aspirin and sildenafil), the guideline group concluded that none are recommended for increasing efficacy or safety.
LIMITATIONS, REASON FOR CAUTION
Several newer interventions are not well studied yet. For most of these interventions, a recommendation against the intervention or a research-only recommendation was formulated based on insufficient evidence. Future studies may require these recommendations to be revised.
WIDER IMPLICATIONS OF THE FINDINGS
The guideline provides clinicians with clear advice on best practice in ovarian stimulation, based on the best evidence available. In addition, a list of research recommendations is provided to promote further studies in ovarian stimulation.
STUDY FUNDING/COMPETING INTEREST(S)
The guideline was developed and funded by ESHRE, covering expenses associated with the guideline meetings, with the literature searches and with the dissemination of the guideline. The guideline group members did not receive payment. F.B. reports research grant from Ferring and consulting fees from Merck, Ferring, Gedeon Richter and speaker’s fees from Merck. N.P. reports research grants from Ferring, MSD, Roche Diagnositics, Theramex and Besins Healthcare; consulting fees from MSD, Ferring and IBSA; and speaker’s fees from Ferring, MSD, Merck Serono, IBSA, Theramex, Besins Healthcare, Gedeon Richter and Roche Diagnostics. A.L.M reports research grants from Ferring, MSD, IBSA, Merck Serono, Gedeon Richter and TEVA and consulting fees from Roche, Beckman-Coulter. G.G. reports consulting fees from MSD, Ferring, Merck Serono, IBSA, Finox, Theramex, Gedeon-Richter, Glycotope, Abbott, Vitrolife, Biosilu, ReprodWissen, Obseva and PregLem and speaker’s fees from MSD, Ferring, Merck Serono, IBSA, Finox, TEVA, Gedeon Richter, Glycotope, Abbott, Vitrolife and Biosilu. E.B. reports research grants from Gedeon Richter; consulting and speaker’s fees from MSD, Ferring, Abbot, Gedeon Richter, Merck Serono, Roche Diagnostics and IBSA; and ownership interest from IVI-RMS Valencia. P.H. reports research grants from Gedeon Richter, Merck, IBSA and Ferring and speaker’s fees from MSD, IBSA, Merck and Gedeon Richter. J.U. reports speaker’s fees from IBSA and Ferring. N.M. reports research grants from MSD, Merck and IBSA; consulting fees from MSD, Merck, IBSA and Ferring and speaker’s fees from MSD, Merck, IBSA, Gedeon Richter and Theramex. M.G. reports speaker’s fees from Merck Serono, Ferring, Gedeon Richter and MSD. S.K.S. reports speaker’s fees from Merck, MSD, Ferring and Pharmasure. E.K. reports speaker’s fees from Merck Serono, Angellini Pharma and MSD. M.K. reports speaker’s fees from Ferring. T.T. reports speaker’s fees from Merck, MSD and MLD. The other authors report no conflicts of interest.
Disclaimer
This guideline represents the views of ESHRE, which were achieved after careful consideration of the scientific evidence available at the time of preparation. In the absence of scientific evidence on certain aspects, a consensus between the relevant ESHRE stakeholders has been obtained.
Adherence to these clinical practice guidelines does not guarantee a successful or specific outcome, nor does it establish a standard of care. Clinical practice guidelines do not replace the need for application of clinical judgment to each individual presentation, nor variations based on locality and facility type.
ESHRE makes no warranty, express or implied, regarding the clinical practice guidelines and specifically excludes any warranties of merchantability and fitness for a particular use or purpose. (Full disclaimer available at www.eshre.eu/guidelines.)
†ESHRE Pages content is not externally peer reviewed. The manuscript has been approved by the Executive Committee of ESHRE.
Keywords: ovarian stimulation, ESHRE, guideline, evidence based, treatment, GRADE, poor responder, high responder, ovarian hyperstimulation syndrome
WHAT DOES THIS MEAN FOR PATIENTS?
Ovarian stimulation is an important first step in many fertility treatments. During ovarian stimulation, doctors prescribe different medications that stimulate the ovaries into producing 5 to 10 mature eggs, instead of one egg in a normal menstrual cycle. These eggs are then removed from the ovaries during egg pickup and later fertilized with sperm in the lab. The resulting fertilized eggs (now embryos) are transferred to the women’s womb resulting in a pregnancy, if all goes well.
There are several options for ovarian stimulation, but they all include a series of different medications taken over several days/weeks, called a stimulation protocol. There is no one treatment scheme that works for each woman undergoing fertility treatment. Some patients may develop only very few eggs (and the stimulation will have to be repeated), while others may over-react with a risk of a serious complication (called ovarian hyperstimulation syndrome).
The current guideline aims to give advice to fertility doctors on which stimulation protocols are safe and effective. The guideline further provides advice on whether clinicians can predict how patients will react and how to adapt the stimulation protocol, for example for patients expected to be at risk of ovarian hyperstimulation syndrome. Finally, there is some advice on ‘add-on’ treatments (growth hormone, aspirin) in ovarian stimulation and whether these are recommended.
A lay version of the guideline is prepared and available on the ESHRE website www.eshre.eu/guidelines.
Introduction
Ovarian stimulation (OS) is defined as pharmacological treatment with the intention of inducing the development of ovarian follicles. It can be used for two purposes: (i) for timed intercourse or insemination and (ii) in assisted reproduction, to obtain multiple oocytes at follicular aspiration (Zegers-Hochschild et al., 2017).
OS for IVF/ICSI has not been addressed by existing evidence-based guidelines. It is discussed briefly in the National Institute for Health and Care Excellence guideline on fertility problems and the Royal Australian, and New Zealand Colleges of Obstetricians and Gynaecologist have published a statement on OS in assisted reproduction. Based on the lack of guidelines, the ESHRE Special Interest Group Reproductive Endocrinology initiated the development of an ESHRE guideline focussing on all aspects of OS for IVF/ICSI.
The aim of this guideline is to provide clinicians with evidence-based information on the different options for OS for IVF/ICSI, taking into account issues such as the ‘optimal’ ovarian response, live birth rates (LBR), safety, patient compliance and individualization. In this guideline, special attention has also been given to pre- and adjuvant treatments in poor responders and the prevention of ovarian hyperstimulation syndrome (OHSS) in high responders.
Materials and Methods
The guideline was developed according to a well-documented methodology that is universal to ESHRE guidelines (Vermeulen et al., 2017).
In short, 18 key questions were formulated by the Guideline Development Group (GDG) and structured in PICO format (Patient, Intervention, Comparison, Outcome). For each question, databases (PUBMED/MEDLINE and the Cochrane library) were searched from inception to 8 November 2018, with a limitation to studies written in English. From the literature searches, studies were selected based on the PICO questions, assessed for quality and summarized in evidence tables and summary of findings tables. The critical outcomes for this guideline are efficacy in terms of cumulative live birth rate (CLBR) per started cycle and LBR per started cycle, as well as safety in terms of moderate and/or severe OHSS. GDG meetings were organized where the evidence and draft recommendations were presented by the assigned GDG member and discussed until consensus was reached within the group.
Each recommendation was labelled as strong or conditional and a grade was assigned (Andrews et al., 2013) based on the strength of the supporting evidence (High ⊕⊕⊕⊕—Moderate ⊕⊕⊕ ⃝—Low ⊕⊕ ⃝ ⃝—Very low ⊕ ⃝ ⃝ ⃝). In the absence of evidence, the GDG formulated no recommendation or a good practice point (GPP) based on clinical expertise (Table I).
Table I.
Interpretation of strong versus conditional recommendations in the GRADE approach.*
| Implications for | Strong recommendation | Conditional recommendation |
|---|---|---|
| Patients | Most individuals in this situation would want the recommended course of action, and only a small proportion would not. | The majority of individuals in this situation would want the suggested course of action, but many would not. |
| Clinicians | Most individuals should receive the intervention. Adherence to this recommendation according to the guideline could be used as a quality criterion or performance indicator. | Recognize that different choices will be appropriate for individual patients and that you must help each patient arrive at a management decision consistent with his or her values and preferences. |
| Formal decision aids are not likely to be needed to help individuals make decisions consistent with their values and preferences. | Decision aids may be useful in helping individuals to make decisions consistent with their values and preferences. | |
| Policy makers | The recommendation can be adopted as policy in most situations. | Policymaking will require substantial debate and involvement of various stakeholders. |
*(Andrews et al., 2013) GRADE: Grading of Recommendations Assessment, Development and Evaluation
The guideline draft and an invitation to participate in the stakeholder review were published on the ESHRE website. In addition, all relevant stakeholders received a personal invitation to review by e-mail. We received 168 comments from 39 reviewers, representing 21 countries, two national societies (British Fertility Society and working groups from ESHRE). All comments were processed by the GDG, either by adapting the content of the guideline and/or by replying to the reviewer. The review process was summarized in the review report, which is published on the ESHRE website (www.eshre.eu/guidelines).
This guideline will be considered for update 4 years after publication, with an intermediate assessment of the need for updating 2 years after publication.
Results
Key questions and recommendations
The current document summarizes all the key questions and the recommendations from the guideline ‘ovarian stimulation for IVF/ICSI’. Further background information and the supporting evidence for each recommendation can be found in the full version of the guideline available at http://www.eshre.eu/Guidelines-and-Legal/Guidelines. For easy reference, a schematic overview of the guideline is prepared (Fig. 1).
Figure 1.
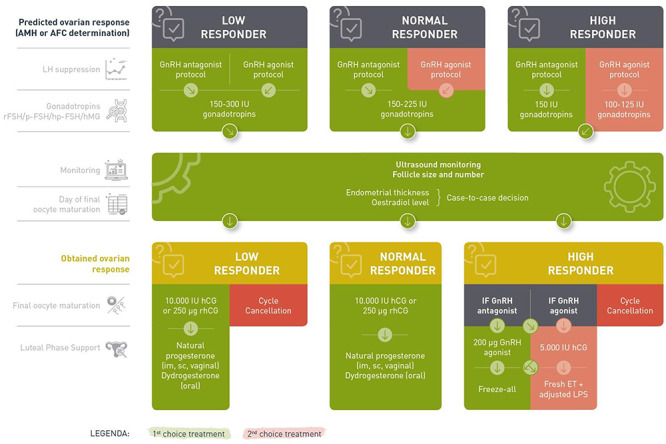
Schematic overview of the guideline ‘ovarian stimulation for IVF/ICSI’. AMH: anti-Müllerian Hormone; AFC: antral follicle count; rFSH: recombinant FSH; p-FSH: purified FSH; hp-FSH: highly purified FSH; LPS: luteal phase support, ET: embryo transfer.
Ovarian response testing
For predicting high and poor response to OS, use of either antral follicle count (AFC) or anti-Müllerian hormone (AMH) is recommended over other ovarian reserve tests. (Broekmans et al., 2006, Broer et al., 2013a, Broer et al., 2013b) Strong ⊕ ⃝ ⃝ ⃝
The clinical implications of these tests regarding change in management with the purpose of improving efficacy and safety have not been evaluated by the GDG.
Assessment of progesterone level on Day 2 of the cycle at the start of OS is probably not recommended. (Panaino et al., 2017) Conditional ⊕ ⃝ ⃝ ⃝
No recommendation can be given in view of the total lack of evidence on the prognostic role of baseline oestradiol in women undergoing OS for IVF/ICSI.
Does hormone pre-treatment improve efficacy and safety of OS?
Pre-treatment with oestrogen before OS using the GnRH antagonist protocol is probably not recommended for improving efficacy and safety. (Farquhar et al., 2017) Conditional ⊕ ⃝ ⃝ ⃝
Pre-treatment with progesterone before OS is probably not recommended for improving efficacy and safety. (Farquhar et al., 2017) Conditional ⊕⊕ ⃝ ⃝
The GDG acknowledges that oestrogen or progesterone is widely used for scheduling purposes. This is probably acceptable given the data on efficacy and safety. GPP
Combined oral contraceptive pill (COCP) pre-treatment (12–28 days) is not recommended in the GnRH antagonist protocol because of reduced efficacy. (Farquhar et al., 2017) Strong ⊕⊕ ⃝ ⃝
GnRH antagonist pre-treatment before OS in a delayed-start gonadotrophin protocol is probably not recommended. (Blockeel et al., 2011a; DiLuigi et al., 2011; Maged et al., 2015; Aflatoonian et al., 2017) Conditional ⊕ ⃝ ⃝ ⃝
LH suppression and OS
According to predicted response-based stratification, which stimulation protocol is most efficient and safe?
High responder
The GnRH antagonist protocol is recommended for women with polycystic ovary syndrome (PCOS), with regards to improved safety and equal efficacy. (Lambalk et al., 2017) Strong ⊕⊕ ⃝ ⃝
The GnRH antagonist protocol is recommended for predicted high responders, with regards to improved safety and equal efficacy. GPP
The addition of clomiphene citrate to gonadotrophins in stimulation protocols is probably not recommended for predicted high responders. (Lin et al., 2007; Saleh et al., 2014; Jiang and Kuang, 2017) Conditional ⊕ ⃝ ⃝ ⃝
There is insufficient evidence to recommend the addition of letrozole to gonadotrophins in stimulation protocols for predicted high responders. (Chen et al., 2018) Conditional ⊕ ⃝ ⃝ ⃝
The GnRH antagonist protocol is recommended for predicted high responders. However, if GnRH agonist protocols are used, a reduced gonadotrophin dose is probably recommended to decrease the risk of OHSS. (Oudshoorn et al., 2017) Conditional ⊕ ⃝ ⃝ ⃝
There is no evidence to justify the use of natural cycle or modified natural cycle for OS in predicted high responders.
Normal responder
The GnRH antagonist protocol is recommended for predicted normal responder women, with regards to improved safety. (Lambalk et al., 2017) Strong ⊕⊕ ⃝ ⃝
The addition of letrozole to gonadotrophins in stimulation protocols is probably not recommended for predicted normal responders. (Verpoest et al., 2006; Mukherjee et al., 2012) Conditional ⊕ ⃝ ⃝ ⃝
A reduced gonadotrophin dose is probably not recommended over a conventional gonadotrophin dose for predicted normal responders. (Hohmann et al., 2003; Baart et al., 2007, Blockeel et al., 2011b; Sterrenburg et al., 2011) Conditional ⊕⊕ ⃝ ⃝
There is no evidence to recommend the use of clomiphene citrate in stimulation protocols for predicted normal responders.
Poor responder
GnRH antagonists and GnRH agonists are equally recommended for predicted poor responders. (Xiao et al., 2013; Lambalk et al., 2017) Conditional ⊕⊕ ⃝ ⃝
Clomiphene citrate alone or in combination with gonadotrophins and gonadotrophin stimulation alone is equally recommended for predicted poor responders. (Bechtejew et al., 2017) Strong ⊕⊕ ⃝ ⃝
The addition of letrozole to gonadotrophins in stimulation protocols is probably not recommended for predicted poor responders. (Bechtejew et al., 2017) Conditional ⊕⊕ ⃝ ⃝
It is unclear whether a higher gonadotrophin dose is recommended over 150 IU for predicted poor responders. (Lensen et al., 2017) Conditional ⊕ ⃝ ⃝ ⃝
A gonadotrophin dose higher than 300 IU is not recommended for predicted poor responders. (Lensen et al., 2017) Strong ⊕ ⃝ ⃝ ⃝
The use of modified natural cycle is probably not recommended over conventional OS for predicted poor responders. (Morgia et al., 2004) Conditional ⊕ ⃝ ⃝ ⃝
No studies were found comparing a reduced FSH dose (<150 IU/day) to conventional FSH stimulation in poor responders.
Which LH suppression regimen is preferable?
If GnRH agonists are used, the long GnRH agonist protocol is probably recommended over the short or ultrashort GnRH agonist protocol. (Siristatidis et al., 2015) Conditional ⊕⊕ ⃝ ⃝
The GnRH antagonist protocol is recommended over the GnRH agonist protocols given the comparable efficacy and higher safety in the general IVF/ICSI population. (Al-Inany et al., 2016) Strong ⊕⊕⊕ ⃝
The use of progestin for LH peak suppression is probably not recommended. If applied, progestin can only be used in the context of non-transfer cycles. (Kuang et al., 2015; Chen et al., 2017; Hamdi et al., 2018) Conditional ⊕ ⃝ ⃝ ⃝
Is the type of stimulation drug associated with efficacy and safety?
The use of recombinant FSH (rFSH) and hMG for OS is equally recommended. (van Wely et al., 2011) Strong ⊕⊕⊕ ⃝
The use of rFSH and purified FSH (p-FSH) for OS in GnRH agonist protocol is equally recommended. (van Wely et al., 2011) Strong ⊕⊕ ⃝ ⃝
The use of either rFSH and highly purified FSH (hp-FSH) for OS in GnRH agonist protocol is equally recommended. (van Wely et al., 2011) Strong ⊕⊕ ⃝ ⃝
The use of hp-FSH and hMG for OS in GnRH agonist protocols is equally recommended. (Duijkers et al., 1993; Westergaard et al., 1996; Parsanezhad et al., 2017) Conditional ⊕⊕ ⃝ ⃝
The use of recombinant LH (rLH) + rFSH for OS is probably not recommended over hMG in GnRH agonist protocols with regards to safety. (Pacchiarotti et al., 2010) Conditional ⊕ ⃝ ⃝ ⃝
Letrozole is probably not recommended as a substitute for gonadotrophins in poor responders. (Verpoest et al., 2006; Yasa et al., 2013; Ebrahimi et al., 2017) Conditional ⊕ ⃝ ⃝ ⃝
The use of long-acting and daily rFSH is equally recommended in GnRH antagonist cycles for normal responders. (Griesinger et al., 2016a) Strong ⊕⊕⊕ ⃝
There is no evidence available to recommend the substitution of FSH by clomiphene citrate in OS.
Is adjustment of the gonadotrophin dosage during the stimulation phase meaningful in terms of efficacy and safety?
Adjustment (increase or decrease) of the gonado-trophin dose in the mid-stimulation phase during OS is probably not recommended. (van Hooff et al., 1993; Aboulghar et al., 2000; Cedrin-Durnerin et al., 2000; Aboulghar et al., 2004; Martin et al., 2006) Conditional ⊕ ⃝ ⃝ ⃝
Is the addition of adjuvants in OS meaningful in terms of efficacy and safety?
Routine use of adjuvant metformin before and/or during OS is not recommended with the GnRH antagonist protocol for women with PCOS. (Tso et al., 2014; Jacob et al., 2016) Strong ⊕⊕ ⃝ ⃝
Use of adjuvant GH before and/or during OS is probably not recommended for poor responders. (Duffy et al., 2010; Li et al., 2017) Conditional ⊕⊕ ⃝ ⃝
Use of testosterone before OS is probably not recommended for poor responders. (Nagels et al., 2015) Conditional ⊕⊕⊕ ⃝
Use of dehydroepiandrosterone before and/or during OS is probably not recommended for poor responders. (Nagels et al., 2015) Conditional ⊕⊕⊕ ⃝
Use of aspirin before and/or during OS is not recommended in the general IVF/ICSI population and for poor responders. (Siristatidis et al., 2016) Strong ⊕⊕⊕ ⃝
Use of sildenafil before and/or during OS is not recommended for poor responders. (Ataalla et al., 2017) Strong ⊕ ⃝ ⃝ ⃝
There is no evidence, i.e. controlled studies or randomised controlled studies (RCTs), addressing the efficacy and safety of adjuvant indomethacin use, to support a recommendation on the use of indomethacin during OS.
What is the safety and efficacy of non-conventional start stimulation compared to standard early follicular phase stimulation?
Random-start OS is probably not recommended for the general IVF/ICSI population. (Pereira et al., 2017) Conditional ⊕ ⃝ ⃝ ⃝
Late luteal phase start of gonadotrophins is probably not recommended for poor responders. (Kuang et al., 2014; Zhang et al., 2016; Liu et al., 2017; Wu et al., 2017; Vaiarelli et al., 2018; Zhang et al., 2018) Conditional ⊕ ⃝ ⃝ ⃝
Early luteal phase start of gonadotrophins is probably not recommended for normal and poor responders. (Zhang et al., 2018) Conditional ⊕ ⃝ ⃝ ⃝
Luteal phase stimulation could be used in non-transfer cycles. GPP
Double stimulation in poor responders should only be used in the context of clinical research. Research only
Double stimulation can be considered for urgent fertility preservation cycles. GPP
What is the preferred stimulation protocol for fertility preservation and freezing for social reasons?
For OS in women seeking fertility preservation for medical reasons, the GnRH antagonist protocol is probably recommended. (Boots et al., 2016; Rodgers et al., 2017) Conditional ⊕ ⃝ ⃝ ⃝
In urgent (oncology) fertility preservation cycles, random-start OS is an important option. (Boots et al., 2016) Conditional ⊕ ⊕ ⃝ ⃝
In OS for fertility preservation in oestrogen-sensitive diseases, the concomitant use of anti-oestrogen therapy, such as letrozole or tamoxifen, can be considered. GPP
Monitoring Is the addition of hormonal assessment (oestradiol/progesterone/LH) to ultrasound monitoring improving efficacy and safety?
The addition of oestradiol measurements to ultra-sound monitoring is probably not recommended. (Kwan et al., 2014) Conditional ⊕⊕ ⃝ ⃝
The addition of a hormonal panel consisting of a combination of oestradiol, progesterone and LH measurements to ultrasound monitoring is probably not recommended. (Golan et al., 1994; Wiser et al., 2012) Conditional ⊕ ⃝ ⃝ ⃝
Does monitoring of endometrial thickness affect the efficacy and safety?
Routine monitoring of endometrial thickness during OS is probably not recommended. (Kasius et al., 2014) Conditional ⊕ ⃝ ⃝ ⃝
The guideline group suggests performing a single measurement of the endometrium during ultrasound assessment on the day of triggering or oocyte retrieval to counsel patients on potential lower pregnancy chance. GPP
Is the outcome of OS dependent on the criteria for triggering?
The association of follicle size as a triggering criterion with outcome has not been sufficiently studied. Physicians may choose the follicle size upon which final oocyte maturation is triggered on a case to case basis. (Chen et al., 2014) Conditional ⊕⊕ ⃝ ⃝
The decision on timing of triggering in relation to follicle size is multi-factorial, taking into account the size of the growing follicle cohort, the hormonal data on the day of pursued trigger, duration of stimulation, patient burden, financial costs, experience of previous cycles and organizational factors for the centre. Most often, final oocyte maturation is triggered at sizes of several of the leading follicles between 16 and 22 mm. GPP
The GDG does not recommend to base timing of final oocyte maturation triggering on oestradiol levels alone. GPP
The GDG does not recommended to base timing of final oocyte maturation on oestradiol/follicle ratio alone. GPP
Which criteria for cycle cancellation are meaningful regarding predicted low/high oocyte yield?
A poor response to OS alone is not a reason to cancel a cycle. (Oudendijk et al., 2012) Strong ⊕ ⃝ ⃝ ⃝
The physician should counsel the individual poor responder regarding pregnancy prospects and decide individually whether to continue this and/or further cycles. GPP
In GnRH agonist cycles with an ovarian response of ≥18 follicles, there is an increased risk of OHSS and preventative measures are recommended, which could include cycle cancellation. (Griesinger et al., 2016b, Mathur et al., 2000; Papanikolaou et al., 2006; Steward et al., 2014) Strong ⊕ ⃝ ⃝ ⃝
Triggering ovulation and luteal support What is the preferred drug for triggering of final oocyte maturation in terms of efficacy and safety in the overall IVF/ICSI population?
The use of recombinant hCG and urinary hCG is equally recommended for triggering final oocyte maturation during OS protocols. (Youssef et al., 2016) Strong ⊕⊕ ⃝ ⃝
A reduced-dose of 5000 IU urinary hCG for final oocyte maturation is probably recommended over a 10 000 IU dose in GnRH agonist protocols, as it may improve safety. (Shaltout et al., 2006; Kolibianakis et al., 2007; Madani et al., 2013) Conditional ⊕ ⃝ ⃝ ⃝
It is not recommended to administer rLH for triggering final oocyte maturation. (Youssef et al., 2016) Strong ⊕ ⃝ ⃝ ⃝
The use of GnRH agonist for final oocyte maturation with conventional luteal support and fresh transfer is not recommended in the general IVF/ICSI population. (Griesinger et al., 2006) Strong ⊕⊕ ⃝ ⃝
The use of GnRH agonist for final oocyte maturation, luteal support with LH-activity and fresh transfer is probably not recommended for the predicted normal responder. (Humaidan et al., 2006; Humaidan et al., 2010; Papanikolaou et al., 2011; Humaidan et al., 2013) Conditional ⊕ ⃝ ⃝ ⃝
If the GnRH agonist trigger with triptorelin is applied, dosages ranging from 0.1 to 0.4 mg can be chosen. (Vuong et al., 2016) GPP
The addition of a GnRH agonist to hCG as a dual trigger for final oocyte maturation is probably not recommended for predicted normal responders. (Ding et al., 2017) Conditional ⊕⊕ ⃝ ⃝
There are no studies investigating the direct comparison of hCG with different dosages of GnRH agonist trigger with buserelin or leuprolide. No controlled studies or RCT could be found comparing different dosages of buserelin or leuprolide for final oocyte maturation. Therefore, no recommendation can be formulated regarding optimal dosage.
What is the efficacy and safety of luteal support protocols?
Progesterone is recommended for luteal phase support after IVF/ICSI. (van der Linden et al., 2015) Strong ⊕ ⃝ ⃝ ⃝
Any of the previously mentioned administration routes (non-oral) for natural progesterone as luteal phase support can be used. GPP
The dosing of natural progesterone has evolved empirically, usually dosages used include:50 mg once daily for i.m. progesterone25 mg once daily for s.c. progesterone 90 mg once daily for vaginal progesterone gel 200 mg three times daily for micronized vaginal progesterone in-oil capsules 100 mg two or three times daily for micronized vaginal progesterone in starch suppositories 400 mg two times daily for vaginal pessary. GPP
Starting of progesterone for luteal phase support should be in the window between the evening of the day of oocyte retrieval and Day 3 post oocyte retrieval. GPP
Progesterone for luteal phase support should be administered at least until the day of the pregnancy test. GPP
Dydrogesterone is probably recommended for luteal phase support. (Barbosa et al., 2018) Conditional ⊕⊕⊕ ⃝
The addition of oestradiol to progesterone for luteal phase support is probably not recommended. (van der Linden et al., 2015) Conditional ⊕⊕ ⃝ ⃝
In hCG triggered OS cycles, hCG as luteal phase support in standard dosages of 1500 IU is probably not recommended. (van der Linden et al., 2015) Conditional ⊕⊕ ⃝ ⃝
A GnRH agonist bolus, in addition to progesterone for luteal phase support in hCG triggered cycles, can only be used in the context of a clinical trial. Research only
Repeated GnRH agonist injections, alone or in addition to progesterone for luteal phase support in hCG triggered cycles, can only be used in the context of a clinical trial. Research only
Addition of LH to progesterone for luteal phase support can only be used in the context of a clinical trial. Research only
Prevention of OHSS Which GnRH agonist medication as a method of triggering will add to the prevention of OHSS also with regards to overall efficacy
A GnRH agonist trigger is recommended for final oocyte maturation in women at risk of OHSS (Babayof et al., 2006; Engmann et al., 2008; Humaidan et al., 2013; Youssef et al., 2014) Strong ⊕ ⃝ ⃝ ⃝
A freeze-all strategy is recommended to eliminate the risk of late-onset OHSS and is applicable in both GnRH agonist and GnRH antagonist protocols. GPP
If a GnRH agonist trigger with freeze-all strategy is not used in patients at risk of OHSS, it is not clear whether the use of a 5000 IU hCG trigger or GnRH agonist trigger is preferred. The GnRH agonist trigger should be followed by luteal phase support with LH-activity (Humaidan et al., 2013) Conditional ⊕ ⃝ ⃝ ⃝
In patients at risk of OHSS, the use of a GnRH agonist for final oocyte maturation is probably recommended over hCG in cases where no fresh transfer is performed (Borges et al., 2016; Tannus et al., 2017) Conditional ⊕ ⃝ ⃝ ⃝
A GnRH agonist trigger for final oocyte maturation with or without a freeze-all strategy is preferred over a coasting strategy in patients at risk of OHSS. GPP
Cabergoline or albumin as additional preventive measures for OHSS is not recommended when GnRH agonist is used for triggering final oocyte maturation. GPP
Is the freeze-all protocol meaningful in the prevention of OHSS also with regard to efficacy?
A freeze-all strategy is recommended to fully eliminate the risk of late-onset OHSS (Wong et al., 2017) Strong ⊕⊕⊕ ⃝
Prior to start of OS, a risk assessment for high response is advised. GPP
Discussion
This ESHRE guideline on OS for IVF/ICSI aims to supply healthcare providers with the best available evidence for approaches in the various steps and phases of OS for IVF/ICSI.
All recommendations in the guideline were formulated after an assessment of the best available evidence in the literature and discussion within the GDG, taking into account the balance of benefits versus harms, patient preferences, clinicians’ expertise and resource use. The guideline includes 84 recommendations, including 61 evidence-based recommendations—of which 21 were formulated as strong recommendations and 40 as conditional—and 19 GPPs and four research only recommendations. The evidence supporting OS was often limited and of low quality. Of the evidence-based recommendations, only eight (13.1%) were supported by moderate quality evidence. The remaining recommendations were supported by low- (22 recommendations: 36.1%) or very low-quality evidence (31 recommendations: 50.8%). There were no recommendations based on high-quality evidence.
One of the difficulties the guideline group encountered when collecting and interpreting the available evidence was the lack of uniform definitions of a high and poor response. Despite the definitions of poor response provided by the Bologna consensus paper (Ferraretti et al., 2011) and the ICMART glossary (Zegers-Hochschild et al., 2017), numerous publications still use a slightly different definition, complicating the interpretation and comparison of data between publications. Similarly, despite several key publications demonstrating the connection between a high number of retrieved oocytes and the risk of OHSS, definitions of high response differ greatly between publications and are often ill-defined within the publications.
One of the most important consequences of the limited evidence is the absence of evidence for interventions aimed at improving OS in poor responders. For most of these interventions, such as adjuvant therapies, there are limited and often very low-quality data. Despite this lack of evidence, several of these adjuvant therapies are regularly administered to women experiencing poor ovarian response. Similarly, there is very limited evidence regarding gonadotrophin dosages in poor responders, yet, high dosages are commonly used without evidence-based motivation. Until large RCTs have been conducted on these interventions, the GDG formulated recommendations against these interventions or dosing levels.
Another consequence of the limited evidence is the number of recommendations specifying (newer) interventions to be applied in a research context rather than routine clinical practice. The current guideline contains four recommendations on interventions to be applied in a research context only. A controversial example of a research-only recommendation is the use of double stimulation, specifically for poor responders.
The current guideline clearly exposes areas where more research is necessary and a research agenda has been developed, with the aim of stimulating research on OS and more specifically on the questions in urgent need of an answer (Fig. 2). While awaiting evidence and evidence-based recommendations, GPPs are provided to support clinicians in routine practice.
Figure 2.

Recommendations for research in ovarian stimulation for IVF/ICSI.
Acknowledgements
The Guideline Development Group acknowledges the help of many clinicians and patient organizations who refereed the content of the Guideline and submitted helpful comments to the draft version.
Authors’ roles
F.B. chaired the guideline development group and hence fulfilled a leading role in collecting the evidence, writing the manuscript and dealing with reviewer comments. N.L.C., as methodological expert, performed all literature searches for the guideline, provided methodological support and coordinated the guideline development. All other authors, listed in alphabetical order, as guideline group members, contributed equally to the manuscript, by drafting key questions, synthesizing evidence, writing the different parts of the guideline and discussing recommendations until consensus within the group was reached. All authors approved of the final version of the guideline.
Funding
The study has no external funding; all costs for meetings were covered by ESHRE.
Conflict of interest
F.B. reports research grant from Ferring and consulting fees from Merck, Ferring and Gedeon Richter and speaker’s fees from Merck. N.P. reports research grants from Ferring, MSD, Roche Diagnostics, Theramex and Besins Healthcare; consulting fees from MSD, Ferring and IBSA and speaker’s fees from Ferring, MSD, Merck Serono, IBSA, Theramex, Besins Healthcare, Gedeon Richter and Roche Diagnostics. A.L.M. reports research grants from Ferring, MSD, IBSA, Merck Serono, Gedeon-Richter and TEVA and consulting fees from Roche, Beckman-Coulter. G.G. reports consulting fees from MSD, Ferring, Merck Serono, IBSA, Finox, Theramex, Gedeon-Richter, Glycotope, Abbott, Vitrolife, Biosilu, ReprodWissen, Obseva and PregLem and speaker’s fees from MSD, Ferring, Merck Serono, IBSA, Finox, TEVA, Gedeon Richter, Glycotope, Abbott, Vitrolife and Biosilu. E.B. reports research grants from Gedeon Richter, consulting and speaker’s fees from MSD, Ferring, Abbot, Gedeon Richter, Merck Serono, Roche Diagnostics and IBSA and ownership interest from IVI-RMS Valencia. P.H. reports research grants from Gedeon Richter, Merck, IBSA and Ferring and speaker’s fees from MSD, IBSA, Merck and Gedeon Richter. J.U. reports speaker’s fees from IBSA, Ferring. N.M. reports research grants from MSD, Merck and IBSA; consulting fees from MSD, Merck, IBSA and Ferring and speaker’s fees from MSD, Merck, IBSA, Gedeon Richter and Theramex. M.G. reports speaker’s fees from Merck Serono, Ferring, Gedeon Richter and MSD. S.K.S. reports speaker’s fees from Merck, MSD, Ferring and Pharmasure. E.K. reports speaker’s fees from Merck Serono, Angellini Pharma and MSD. M.K. reports speaker’s fees from Ferring. T.T. reports speaker’s fees from Merck, MSD and MLD. The other authors report no conflicts of interest.
References
- Aboulghar MA, Mansour RT, Serour GI, Al-Inany HG, Amin YM, Aboulghar MM. Increasing the dose of human menopausal gonadotrophins on day of GnRH antagonist administration: randomized controlled trial. Reprod Biomed Online 2004;8:524–527. [DOI] [PubMed] [Google Scholar]
- Aboulghar MA, Mansour RT, Serour GI, Rhodes CA, Amin YM. Reduction of human menopausal gonadotropin dose before coasting prevents severe ovarian hyperstimulation syndrome with minimal cycle cancellation. J Assist Reprod Genet 2000;17:298–301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aflatoonian A, Hosseinisadat A, Baradaran R, Farid MM. Pregnancy outcome of “delayed start” GnRH antagonist protocol versus GnRH antagonist protocol in poor responders: a clinical trial study. Int J Reprod Biomed 2017;15:231–238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Al-Inany HG, Youssef MA, Ayeleke RO, Brown J, Lam WS, Broekmans FJ. Gonadotrophin-releasing hormone antagonists for assisted reproductive technology. Cochrane Database Syst Rev 2016;4:Cd001750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andrews J, Guyatt G, Oxman AD, Alderson P, Dahm P, Falck-Ytter Y, Nasser M, Meerpohl J, Post PN, Kunz R et al. GRADE guidelines: 14. Going from evidence to recommendations: the significance and presentation of recommendations. J Clin Epidemiol 2013;66:719–725. [DOI] [PubMed] [Google Scholar]
- Ataalla W, Elhamid T, Elhalwagy A. Adjuvant sildenafil therapy in poor responders undergoing in vitro fertilization: a prospective, randomized, double-blind, placebo-controlled trial. Middle East Fertil Soc J 2017;21:175–179. [Google Scholar]
- Baart EB, Martini E, Eijkemans MJ, Van Opstal D, Beckers NG, Verhoeff A, Macklon NS, Fauser BC. Milder ovarian stimulation for in-vitro fertilization reduces aneuploidy in the human preimplantation embryo: a randomized controlled trial. Hum Reprod 2007;22:980–988. [DOI] [PubMed] [Google Scholar]
- Babayof R, Margalioth EJ, Huleihel M, Amash A, Zylber-Haran E, Gal M, Brooks B, Mimoni T, Eldar-Geva T. Serum inhibin A, VEGF and TNFalpha levels after triggering oocyte maturation with GnRH agonist compared with HCG in women with polycystic ovaries undergoing IVF treatment: a prospective randomized trial. Hum Reprod 2006;21:1260–1265. [DOI] [PubMed] [Google Scholar]
- Barbosa MWP, Valadares NPB, Barbosa ACP, Amaral AS, Iglesias JR, Nastri CO, Martins WP, Nakagawa HM. Oral dydrogesterone vs. vaginal progesterone capsules for luteal-phase support in women undergoing embryo transfer: a systematic review and meta-analysis. JBRA Assist Reprod 2018;22:148–156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bechtejew TN, Nadai MN, Nastri CO, Martins WP. Clomiphene citrate and letrozole to reduce follicle-stimulating hormone consumption during ovarian stimulation: systematic review and meta-analysis. Ultrasound Obstet Gynecol 2017;50:315–323. [DOI] [PubMed] [Google Scholar]
- Blockeel C, Riva A, De Vos M, Haentjens P, Devroey P. Administration of a gonadotropin-releasing hormone antagonist during the 3 days before the initiation of the in vitro fertilization/intracytoplasmic sperm injection treatment cycle: impact on ovarian stimulation. A pilot study. Fertil Steril 2011a;95:1714–1719e1711-1712. [DOI] [PubMed] [Google Scholar]
- Blockeel C, Sterrenburg MD, Broekmans FJ, Eijkemans MJ, Smitz J, Devroey P, Fauser BC. Follicular phase endocrine characteristics during ovarian stimulation and GnRH antagonist cotreatment for IVF: RCT comparing recFSH initiated on cycle day 2 or 5. J Clin Endocrinol Metab 2011b;96:1122–1128. [DOI] [PubMed] [Google Scholar]
- Boots CE, Meister M, Cooper AR, Hardi A, Jungheim ES. Ovarian stimulation in the luteal phase: systematic review and meta-analysis. J Assist Reprod Genet 2016;33:971–980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Borges E Jr, Braga DP, Setti AS, Vingris LS, Figueira RC, Iaconelli A Jr. Strategies for the management of OHSS: results from freezing-all cycles. JBRA Assist Reprod 2016;20:8–12. [DOI] [PubMed] [Google Scholar]
- Broekmans FJ, Kwee J, Hendriks DJ, Mol BW, Lambalk CB. A systematic review of tests predicting ovarian reserve and IVF outcome. Hum Reprod Update 2006;12:685–718. [DOI] [PubMed] [Google Scholar]
- Broer SL, Dolleman M, Disseldorp J, Broeze KA, Opmeer BC, Bossuyt PM, Eijkemans MJ, Mol BW, Broekmans FJ. Prediction of an excessive response in in vitro fertilization from patient characteristics and ovarian reserve tests and comparison in subgroups: an individual patient data meta-analysis. Fertil Steril 2013a;100:420–429e427. [DOI] [PubMed] [Google Scholar]
- Broer SL, Disseldorp J, Broeze KA, Dolleman M, Opmeer BC, Bossuyt P, Eijkemans MJ, Mol BW, Broekmans FJ. Added value of ovarian reserve testing on patient characteristics in the prediction of ovarian response and ongoing pregnancy: an individual patient data approach. Hum Reprod Update 2013b;19:26–36. [DOI] [PubMed] [Google Scholar]
- Cedrin-Durnerin I, Bstandig B, Herve F, Wolf J, Uzan M, Hugues J. A comparative study of high fixed-dose and decremental-dose regimens of gonadotropins in a minidose gonadotropin-releasing hormone agonist flare protocol for poor responders. Fertil Steril 2000;73:1055–1056. [DOI] [PubMed] [Google Scholar]
- Chen Q, Wang Y, Sun L, Zhang S, Chai W, Hong Q, Long H, Wang L, Lyu Q, Kuang Y. Controlled ovulation of the dominant follicle using progestin in minimal stimulation in poor responders. Reprod Biol Endocrinol 2017;15:71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen Y, Yang T, Hao C, Zhao J. A retrospective study of letrozole treatment prior to human chorionic gonadotropin in women with polycystic ovary syndrome undergoing in vitro fertilization at risk of ovarian hyperstimulation syndrome. Med Sci Mon Int Med J Exp Clin Res 2018;24:4248–4253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen Y, Zhang Y, Hu M, Liu X, Qi H. Timing of human chorionic gonadotropin (hCG) hormone administration in IVF/ICSI protocols using GnRH agonist or antagonists: a systematic review and meta-analysis. Gynecol Endocrinol 2014;30:431–437. [DOI] [PubMed] [Google Scholar]
- DiLuigi A, Engmann L, Schmidt D, Benadiva C, Nulsen J. A randomized trial of microdose leuprolide acetate protocol versus luteal phase ganirelix protocol in predicted poor responders. Fertil Steril 2011;95:2531–2533. [DOI] [PubMed] [Google Scholar]
- Ding N, Liu X, Jian Q, Liang Z, Wang F. Dual trigger of final oocyte maturation with a combination of GnRH agonist and hCG versus a hCG alone trigger in GnRH antagonist cycle for in vitro fertilization: a systematic review and meta-analysis. Eur J Obstet Gynecol Reprod Biol 2017;218:92–98. [DOI] [PubMed] [Google Scholar]
- Duffy JM, Ahmad G, Mohiyiddeen L, Nardo LG, Watson A. Growth hormone for in vitro fertilization. Cochrane Database Syst Rev 2010;Cd000099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duijkers IJ, Vemer HM, Hollanders JM, Willemsen WN, Bastiaans LA, Hamilton CJ, Thomas CM, Borm GF. Different follicle stimulating hormone/luteinizing hormone ratios for ovarian stimulation. Hum Reprod 1993;8:1387–1391. [DOI] [PubMed] [Google Scholar]
- Ebrahimi M, Akbari-Asbagh F, Ghalandar-Attar M. Letrozole+ GnRH antagonist stimulation protocol in poor ovarian responders undergoing intracytoplasmic sperm injection cycles: an RCT. Int J Reprod Biomed 2017;15:101–108. [PMC free article] [PubMed] [Google Scholar]
- Engmann L, DiLuigi A, Schmidt D, Nulsen J, Maier D, Benadiva C. The use of gonadotropin-releasing hormone (GnRH) agonist to induce oocyte maturation after cotreatment with GnRH antagonist in high-risk patients undergoing in vitro fertilization prevents the risk of ovarian hyperstimulation syndrome: a prospective randomized controlled study. Fertil Steril 2008;89:84–91. [DOI] [PubMed] [Google Scholar]
- Farquhar C, Rombauts L, Kremer JA, Lethaby A, Ayeleke RO. Oral contraceptive pill, progestogen or oestrogen pretreatment for ovarian stimulation protocols for women undergoing assisted reproductive techniques. Cochrane Database Syst Rev 2017;5:Cd006109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferraretti AP, La Marca A, Fauser BC, Tarlatzis B, Nargund G, Gianaroli L. ESHRE consensus on the definition of ‘poor response’ to ovarian stimulation for in vitro fertilization: the Bologna criteria. Hum Reprod 2011;26:1616–1624. [DOI] [PubMed] [Google Scholar]
- Golan A, Herman A, Soffer Y, Bukovsky I, Ron-El R. Ultrasonic control without hormone determination for ovulation induction in in-vitro fertilization/embryo transfer with gonadotrophin-releasing hormone analogue and human menopausal gonadotrophin. Hum Reprod 1994;9:1631–1633. [DOI] [PubMed] [Google Scholar]
- Griesinger G, Boostanfar R, Gordon K, Gates D, McCrary Sisk C, Stegmann BJ. Corifollitropin alfa versus recombinant follicle-stimulating hormone: an individual patient data meta-analysis. Reprod Biomed Online 2016a;33:56–60. [DOI] [PubMed] [Google Scholar]
- Griesinger G, Diedrich K, Devroey P, Kolibianakis EM. GnRH agonist for triggering final oocyte maturation in the GnRH antagonist ovarian hyperstimulation protocol: a systematic review and meta-analysis. Hum Reprod Update 2006;12:159–168. [DOI] [PubMed] [Google Scholar]
- Griesinger G, Verweij PJ, Gates D, Devroey P, Gordon K, Stegmann BJ, Tarlatzis BC. Prediction of ovarian hyperstimulation syndrome in patients treated with corifollitropin alfa or rFSH in a GnRH antagonist protocol. PloS one 2016b;11:e0149615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hamdi K, Farzadi L, Ghasemzadeh A, Navali N, Atashkhoei S, Pia H, Shahnazi V, Fattahi A, Bahrami-Asl Z, Sepasi F et al. Comparison of medroxyprogesterone acetate with cetrotide for prevention of premature luteinizing hormone surges in women undergoing in vitro fertilization. Int J Women Health Reprod Sci 2018;6:187–191. [Google Scholar]
- Hohmann F, Macklon N, Fauser B. A randomized comparison of two ovarian stimulation protocols with gonadotropin-releasing hormone (GnRH) antagonist cotreatment for in vitro fertilization commencing recombinant follicle-stimulating hormone on cycle day 2 or 5 with the standard long GnRH agonist protocol. J Clin Endocrinol Metab 2003;88:166–173. [DOI] [PubMed] [Google Scholar]
- Humaidan P, Bungum L, Bungum M, Yding AC. Rescue of corpus luteum function with peri-ovulatory HCG supplementation in IVF/ICSI GnRH antagonist cycles in which ovulation was triggered with a GnRH agonist: a pilot study. Reprod Biomed Online 2006;13:173–178. [DOI] [PubMed] [Google Scholar]
- Humaidan P, Ejdrup Bredkjaer H, Westergaard LG, Yding AC. 1,500 IU human chorionic gonadotropin administered at oocyte retrieval rescues the luteal phase when gonadotropin-releasing hormone agonist is used for ovulation induction: a prospective, randomized, controlled study. Fertil Steril 2010;93:847–854. [DOI] [PubMed] [Google Scholar]
- Humaidan P, Polyzos NP, Alsbjerg B, Erb K, Mikkelsen AL, Elbaek HO, Papanikolaou EG, Andersen CY. GnRHa trigger and individualized luteal phase hCG support according to ovarian response to stimulation: two prospective randomized controlled multi-centre studies in IVF patients. Hum Reprod 2013;28:2511–2521. [DOI] [PubMed] [Google Scholar]
- Jacob SL, Brewer C, Tang T, Picton HM, Barth JH, Balen AH. A short course of metformin does not reduce OHSS in a GnRH antagonist cycle for women with PCOS undergoing IVF: a randomised placebo-controlled trial. Hum Reprod 2016;31:2756–2764. [DOI] [PubMed] [Google Scholar]
- Jiang S, Kuang Y. Clomiphene citrate is associated with favorable cycle characteristics but impaired outcomes of obese women with polycystic ovarian syndrome undergoing ovarian stimulation for in vitro fertilization. Medicine 2017;96:e7540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kasius A, Smit JG, Torrance HL, Eijkemans MJ, Mol BW, Opmeer BC, Broekmans FJ. Endometrial thickness and pregnancy rates after IVF: a systematic review and meta-analysis. Hum Reprod Update 2014;20:530–541. [DOI] [PubMed] [Google Scholar]
- Kolibianakis EM, Papanikolaou EG, Tournaye H, Camus M, Van Steirteghem AC, Devroey P. Triggering final oocyte maturation using different doses of human chorionic gonadotropin: a randomized pilot study in patients with polycystic ovary syndrome treated with gonadotropin-releasing hormone antagonists and recombinant follicle-stimulating hormone. Fertil Steril 2007;88:1382–1388. [DOI] [PubMed] [Google Scholar]
- Kuang Y, Chen Q, Fu Y, Wang Y, Hong Q, Lyu Q, Ai A, Shoham Z. Medroxyprogesterone acetate is an effective oral alternative for preventing premature luteinizing hormone surges in women undergoing controlled ovarian hyperstimulation for in vitro fertilization. Fertil Steril 2015;104:62–70e63. [DOI] [PubMed] [Google Scholar]
- Kuang Y, Chen Q, Hong Q, Lyu Q, Ai A, Fu Y, Shoham Z. Double stimulations during the follicular and luteal phases of poor responders in IVF/ICSI programmes (Shanghai protocol). Reprod Biomed Online 2014;29:684–691. [DOI] [PubMed] [Google Scholar]
- Kwan I, Bhattacharya S, Kang A, Woolner A. Monitoring of stimulated cycles in assisted reproduction (IVF and ICSI). Cochrane Database Syst Rev 2014;Cd005289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lambalk CB, Banga FR, Huirne JA, Toftager M, Pinborg A, Homburg R, Veen F, Wely M. GnRH antagonist versus long agonist protocols in IVF: a systematic review and meta-analysis accounting for patient type. Hum Reprod Update 2017;23:560–579. [DOI] [PubMed] [Google Scholar]
- Lensen SF, Wilkinson J, Mol BWJ, La MA, Torrance H, Broekmans FJ. Individualised gonadotropin dose selection using markers of ovarian reserve for women undergoing IVF/ICSI In: Cochrane Database of Systematic Reviews. John Wiley & Sons, Ltd, 2017. [Google Scholar]
- Li XL, Wang L, Lv F, Huang XM, Wang LP, Pan Y, Zhang XM. The influence of different growth hormone addition protocols to poor ovarian responders on clinical outcomes in controlled ovary stimulation cycles: a systematic review and meta-analysis. Medicine 2017;96:e6443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin YH, Seow KM, Hsieh BC, Huang LW, Chen HJ, Huang SC, Chen CY, Chen PH, Hwang JL, Tzeng CR. Application of GnRH antagonist in combination with clomiphene citrate and hMG for patients with exaggerated ovarian response in previous IVF/ICSI cycles. J Assist Reprod Genet 2007;24:331–336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu C, Jiang H, Zhang W, Yin H. Double ovarian stimulation during the follicular and luteal phase in women >/=38 years: a retrospective case-control study. Reprod Biomed Online 2017;35:678–684. [DOI] [PubMed] [Google Scholar]
- Madani T, Mohammadi Yeganeh L, Ezabadi Z, Hasani F, Chehrazi M. Comparing the efficacy of urinary and recombinant hCG on oocyte/follicle ratio to trigger ovulation in women undergoing intracytoplasmic sperm injection cycles: a randomized controlled trial. J Assist Reprod Genet 2013;30:239–245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maged A, Nada A, Abohamila F, Hashem A, Mostafa W, Elzayat A. Delayed start versus conventional GnRH antagonist protocol in poor responders pretreated with estradiol in luteal phase: a randomized controlled trial. Reprod Sci 2015;22:1627–1631. [DOI] [PubMed] [Google Scholar]
- Martin JR, Mahutte NG, Arici A, Sakkas D. Impact of duration and dose of gonadotrophins on IVF outcomes. Reprod Biomed Online 2006;13:645–650. [DOI] [PubMed] [Google Scholar]
- Mathur RS, Akande AV, Keay SD, Hunt LP, Jenkins JM. Distinction between early and late ovarian hyperstimulation syndrome. Fertil Steril 2000;73:901–907. [DOI] [PubMed] [Google Scholar]
- Morgia F, Sbracia M, Schimberni M, Giallonardo A, Piscitelli C, Giannini P, Aragona C. A controlled trial of natural cycle versus microdose gonadotropin-releasing hormone analog flare cycles in poor responders undergoing in vitro fertilization. Fertil Steril 2004;81:1542–1547. [DOI] [PubMed] [Google Scholar]
- Mukherjee S, Sharma S, Chakravarty BN. Letrozole in a low-cost in vitro fertilization protocol in intracytoplasmic sperm injection cycles for male factor infertility: a randomized controlled trial. J Hum Reprod Sci 2012;5:170–174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagels HE, Rishworth JR, Siristatidis CS, Kroon B. Androgens (dehydroepiandrosterone or testosterone) for women undergoing assisted reproduction. Cochrane Database Syst Rev 2015;Cd009749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oudendijk JF, Yarde F, Eijkemans MJ, Broekmans FJ, Broer SL. The poor responder in IVF: is the prognosis always poor?: a systematic review. Hum Reprod Update 2012;18:1–11. [DOI] [PubMed] [Google Scholar]
- Oudshoorn SC, Tilborg TC, Eijkemans MJC, Oosterhuis GJE, Friederich J, Hooff MHA, Santbrink EJP, Brinkhuis EA, Smeenk JMJ, Kwee J et al. Individualized versus standard FSH dosing in women starting IVF/ICSI: an RCT. Part 2: the predicted hyper responder. Hum Reprod 2017;32:2506–2514. [DOI] [PubMed] [Google Scholar]
- Pacchiarotti A, Sbracia M, Frega A, Selman H, Rinaldi L, Pacchiarotti A. Urinary hMG (Meropur) versus recombinant FSH plus recombinant LH (Pergoveris) in IVF: a multicenter, prospective, randomized controlled trial. Fertil Steril 2010;94:2467–2469. [DOI] [PubMed] [Google Scholar]
- Panaino TR, Silva JB, Lima MA, Lira P, Areas PC, Mancebo AC, Souza MM, Antunes RA, Souza MD. High progesterone levels in the beginning of ICSI antagonist cycles and clinical pregnancy: still a concern? JBRA Assist Reprod 2017;21:11–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Papanikolaou EG, Pozzobon C, Kolibianakis EM, Camus M, Tournaye H, Fatemi HM, Van Steirteghem A, Devroey P. Incidence and prediction of ovarian hyperstimulation syndrome in women undergoing gonadotropin-releasing hormone antagonist in vitro fertilization cycles. Fertil Steril 2006;85:112–120. [DOI] [PubMed] [Google Scholar]
- Papanikolaou EG, Verpoest W, Fatemi H, Tarlatzis B, Devroey P, Tournaye H. A novel method of luteal supplementation with recombinant luteinizing hormone when a gonadotropin-releasing hormone agonist is used instead of human chorionic gonadotropin for ovulation triggering: a randomized prospective proof of concept study. Fertil Steril 2011;95:1174–1177. [DOI] [PubMed] [Google Scholar]
- Parsanezhad M, Jahromi B, Rezaee S, Kooshesh L, Alaee S. The effect of four different gonadotropin protocols on oocyte and embryo quality and pregnancy outcomes in IVF/ICSI cycles; a randomized controlled trial. Iran J Med Sci 2017;42:57–65. [PMC free article] [PubMed] [Google Scholar]
- Pereira N, Voskuilen-Gonzalez A, Hancock K, Lekovich JP, Schattman GL, Rosenwaks Z. Random-start ovarian stimulation in women desiring elective cryopreservation of oocytes. Reprod Biomed Online 2017;35:400–406. [DOI] [PubMed] [Google Scholar]
- Rodgers RJ, Reid GD, Koch J, Deans R, Ledger WL, Friedlander M, Gilchrist RB, Walters KA, Abbott JA. The safety and efficacy of controlled ovarian hyperstimulation for fertility preservation in women with early breast cancer: a systematic review. Hum Reprod 2017;32:1033–1045. [DOI] [PubMed] [Google Scholar]
- Saleh S, Ismail M, Elshmaa N. The efficacy of converting high response - ovulation induction cycles to in vitro fertilization in patients with PCOS. Middle East Fertil Soc J 2014;19:51–56. [Google Scholar]
- Shaltout A, Eid M, Shohayeb A. Does triggering ovulation by 5000 IU of uhCG affect ICSI outcome? Middle East Fertil Soc J 2006;11:99–103. [Google Scholar]
- Siristatidis CS, Basios G, Pergialiotis V, Vogiatzi P. Aspirin for in vitro fertilisation. Cochrane Database Syst Rev 2016;11:Cd004832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siristatidis CS, Gibreel A, Basios G, Maheshwari A, Bhattacharya S. Gonadotrophin-releasing hormone agonist protocols for pituitary suppression in assisted reproduction. Cochrane Database Syst Rev 2015;Cd006919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sterrenburg MD, Veltman-Verhulst SM, Eijkemans MJ, Hughes EG, Macklon NS, Broekmans FJ, Fauser BC. Clinical outcomes in relation to the daily dose of recombinant follicle-stimulating hormone for ovarian stimulation in in vitro fertilization in presumed normal responders younger than 39 years: a meta-analysis. Hum Reprod Update 2011;17:184–196. [DOI] [PubMed] [Google Scholar]
- Steward RG, Lan L, Shah AA, Yeh JS, Price TM, Goldfarb JM, Muasher SJ. Oocyte number as a predictor for ovarian hyperstimulation syndrome and live birth: an analysis of 256,381 in vitro fertilization cycles. Fertil Steril 2014;101:967–973. [DOI] [PubMed] [Google Scholar]
- Tannus S, Turki R, Cohen Y, Son WY, Shavit T, Dahan MH. Reproductive outcomes after a single dose of gonadotropin-releasing hormone agonist compared with human chorionic gonadotropin for the induction of final oocyte maturation in hyper-responder women aged 35-40 years. Fertil Steril 2017;107:1323–1328e1322. [DOI] [PubMed] [Google Scholar]
- Tso LO, Costello MF, Albuquerque LE, Andriolo RB, Macedo CR. Metformin treatment before and during IVF or ICSI in women with polycystic ovary syndrome. Cochrane Database Syst Rev 2014;Cd006105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vaiarelli A, Cimadomo D, Trabucco E, Vallefuoco R, Buffo L, Dusi L, Fiorini F, Barnocchi N, Bulletti FM, Rienzi L et al. Double stimulation in the same ovarian cycle (DuoStim) to maximize the number of oocytes retrieved from poor prognosis patients: a multicenter experience and SWOT analysis. Front Endocrinol 2018;9:317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Linden M, Buckingham K, Farquhar C, Kremer JA, Metwally M. Luteal phase support for assisted reproduction cycles. Cochrane Database Syst Rev 2015;Cd009154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hooff MH, Alberda AT, Huisman GJ, Zeilmaker GH, Leerentveld RA. Doubling the human menopausal gonadotrophin dose in the course of an in-vitro fertilization treatment cycle in low responders: a randomized study. Hum Reprod 1993;8:369–373. [DOI] [PubMed] [Google Scholar]
- Wely M, Kwan I, Burt AL, Thomas J, Vail A, Van der Veen F, Al-Inany HG. Recombinant versus urinary gonadotrophin for ovarian stimulation in assisted reproductive technology cycles. Cochrane Database Syst Rev 2011;Cd005354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vermeulen N, Le Clef N, D'Angelo A, Tilleman K, Veleva Z, Nelen WL. Manual for ESHRE Guideline Development, 2017;www.eshre.eu/guidelines.
- Verpoest W, Kolibianakis E, Papanikolaou E, Smitz J, Steirteghem A, Devroey P. Aromatase inhibitors in ovarian stimulation for IVF/ICSI: a pilot study. Reprod Biomed Online 2006;13:166–172. [DOI] [PubMed] [Google Scholar]
- Vuong TN, Ho MT, Ha TD, Phung HT, Huynh GB, Humaidan P. Gonadotropin-releasing hormone agonist trigger in oocyte donors co-treated with a gonadotropin-releasing hormone antagonist: a dose-finding study. Fertil Steril 2016;105:356–363. [DOI] [PubMed] [Google Scholar]
- Westergaard LG, Erb K, Laursen S, Rasmussen PE, Rex S. The effect of human menopausal gonadotrophin and highly purified, urine-derived follicle stimulating hormone on the outcome of in-vitro fertilization in down-regulated normogonadotrophic women. Hum Reprod 1996;11:1209–1213. [DOI] [PubMed] [Google Scholar]
- Wiser A, Gonen O, Ghetler Y, Shavit T, Berkovitz A, Shulman A. Monitoring stimulated cycles during in vitro fertilization treatment with ultrasound only--preliminary results. Gynecol Endocrinol 2012;28:429–431. [DOI] [PubMed] [Google Scholar]
- Wong KM, Wely M, Mol F, Repping S, Mastenbroek S. Fresh versus frozen embryo transfers in assisted reproduction. Cochrane Database Syst Rev 2017;3:Cd011184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu Y, Zhao FC, Sun Y, Liu PS. Luteal-phase protocol in poor ovarian response: a comparative study with an antagonist protocol. J Int Med Res 2017;45:1731–1738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xiao J, Chang S, Chen S. The effectiveness of gonadotropin-releasing hormone antagonist in poor ovarian responders undergoing in vitro fertilization: a systematic review and meta-analysis. Fertil Steril 2013;100:1594–1601e1591-1599. [DOI] [PubMed] [Google Scholar]
- Yasa C, Bastu E, Dural O, Celik E, Ergun B. Evaluation of low-dose letrozole addition to ovulation induction in IVF. Clin Exp Obstet Gynecol 2013;40:98–100. [PubMed] [Google Scholar]
- Youssef MA, Abou-Setta AM, Lam WS. Recombinant versus urinary human chorionic gonadotrophin for final oocyte maturation triggering in IVF and ICSI cycles. Cochrane Database Syst Rev 2016;4:Cd003719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Youssef MA, Van der Veen F, Al-Inany HG, Mochtar MH, Griesinger G, Nagi Mohesen M, Aboulfoutouh I, Wely M. Gonadotropin-releasing hormone agonist versus HCG for oocyte triggering in antagonist-assisted reproductive technology. Cochrane Database Syst Rev 2014;Cd008046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zegers-Hochschild F, Adamson GD, Dyer S, Racowsky C, Mouzon J, Sokol R, Rienzi L, Sunde A, Schmidt L, Cooke ID et al. The International Glossary on Infertility and Fertility Care, 2017. Hum Reprod 2017;32:1786–1801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Q, Guo XM, Li Y. Implantation rates subsequent to the transfer of embryos produced at different phases during double stimulation of poor ovarian responders. Reprod Fertil Dev 2016;29:1178–1183. [DOI] [PubMed] [Google Scholar]
- Zhang W, Wang M, Wang S, Bao H, Qu Q, Zhang N, Hao C. Luteal phase ovarian stimulation for poor ovarian responders. JBRA Assist Reprod 2018;22:193–198. [DOI] [PMC free article] [PubMed] [Google Scholar]


