Abstract
Feline mammary carcinoma (FMC) is similar to human breast cancer in the late age of onset, incidence, histopathologic features, biological behavior, and pattern of metastasis. Therefore, FMC has been proposed as a relevant model for aggressive human breast cancer. The goals of this study were to develop a nude mouse model of FMC tumor growth and metastasis and to measure the expression of genes responsible for lymphangiogenesis, angiogenesis, tumor progression, and lymph node metastasis in FMC tissues and cell lines. Two primary FMC tissues were injected subcutaneously, and 6 FMC cell lines were injected into 3 sites (subcutaneous, intratibial, and intracardiac) in nude mice. Tumors and metastases were monitored using bioluminescent imaging and characterized by gross necropsy, radiology, and histopathology. Molecular characterization of invasion and metastasis genes in FMC was conducted using quantitative real-time reverse transcription polymerase chain reaction in 6 primary FMC tissues, 2 subcutaneous FMC xenografts, and 6 FMC cell lines. The histologic appearance of the subcutaneous xenografts resembled the primary tumors. No metastasis was evident following subcutaneous injection of tumor tissues and cell lines, whereas lung, brain, liver, kidney, eye, and bone metastases were confirmed following intratibial and intracardiac injection of FMC cell lines. Finally, 15 genes were differentially expressed in the FMC tissues and cell lines. The highly expressed genes in all samples were PDGFA, PDGFB, PDGFC, FGF2, EGFR, ERBB2, ERBB3, VEGFD, VEGFR3, and MYOF. Three genes (PDGFD, ANGPT2, and VEGFC) were confirmed to be of stromal origin. This investigation demonstrated the usefulness of nude mouse models of experimental FMC and identified molecular targets of FMC progression and metastasis.
Keywords: mammary cancer, metastasis, lung, brain, bone, angiogenesis, lymphangiogenesis, cat, animal model
Breast cancer is the most common neoplasm in women, with approximately 230 480 new cases and 39 520 deaths occurring in 2011 in the United States.50 Feline mammary carcinoma (FMC) has been proposed as a useful model for human breast cancer based on age of incidence, risk factors, histopathology, prognostic aspects, metastatic pattern, and response to therapy.20 The annual incidence of feline mammary neoplasia was estimated at 13 to 25 per 100 000 female cats.36 Feline mammary tumors are the most frequent neoplasm causing death in cats25 and the third most commonly diagnosed neoplasm following lymphohematopoietic and skin tumors and account for 12% of all neoplasms and 17% of those in female cats.36
Previous studies revealed a breed-associated risk of mammary cancer in Siamese cats, which had twice the risk of developing FMC compared with other breeds.13,14,21 Mammary neoplasms occur predominantly in middle-aged to older female cats with a mean age of 10 to 12 years.36,61 Feline mammary neoplasms, typically classified according to the World Health Organization criteria, are usually represented by in situ or invasive carcinomas. The invasive carcinomas include tubulopapillary, solid, and cribriform types while less frequent patterns include carcinosarcoma, carcinoma or sarcoma in a benign tumor, mucinous carcinoma, and carcinoma with squamous differentiation.37,62
Unfortunately, more than 80% of feline mammary tumors are malignant and entail rapid progression and metastasis at an early stage.30,36,61 The interval between feline mammary tumor diagnosis and death varies from less than 6 months to about 12 months.38 However, despite efforts made during surgery, cats undergoing mastectomies exhibit a high incidence of surgical failure either in the form of inadequate removal of the primary neoplasms, local recurrence, and/or metastases that suggest the presence of micrometastases at the time of surgery.34,58
Metastasis is the most devastating and life-threatening problem in women with breast cancer and therefore has become an important focus for research.59 FMCs commonly metastasize to lung and regional lymph nodes and sometimes to other organs.27 Malignant tumors spread to lymph nodes through lymphatic vessels due to tumor production of growth factors that directly or indirectly enhance the process of lymphangiogenesis.6 Changes in the expression of genes at the messenger RNA (mRNA) and protein levels have been described in both human breast cancer and FMCs.61 Such genes included the epidermal growth factor receptor-2 (HER2, ERBB2) and RON (tyrosine kinase receptor gene).9,10 Mouse models of mammary cancer are indispensable because they provide an opportunity to investigate the mechanisms of metastasis, genetic cancer progression, and the roles of angiogenesis and lymphangiogenesis.59
Since FMC has a high rate of malignancy, it will be useful to investigate the pathogenesis of metastasis and the expression of tumor-related genes. The goals of this study were to develop a nude mouse model of FMC tumor growth and metastasis and to characterize the molecular features of FMC by the expression of genes responsible for lymphangiogenesis, angiogenesis, tumor progression, and lymph node metastasis in feline mammary tumor tissues and cell lines.
Material and Methods
Feline Mammary Cancer Tissues
Fresh-frozen tumor samples from 6 primary feline mammary tumor tissues (CM, JF, PH, LTC, SB, and MM) were obtained from The Ohio State University (OSU) College of Veterinary Medicine Biospecimen Repository (Dr Holly Borghese, coordinator, and Emily Kuhn). The tumors were derived from cats of different breeds, with case descriptions as follows.
CM was a 14-year-old female spayed Siamese cat that presented to the OSU Veterinary Medical Center (VMC) oncology service for evaluation of multiple masses along the left mammary chain and axilla. CM had previously delivered 3 litters of kittens and then was spayed. The size of the largest mass was 2 × 1.25 cm.
JF was a 14-year-old female spayed Abyssinian cat that presented to the OSU Community Practice for evaluation of weight loss and a mass on her abdomen (5 × 3 cm). Radio-graphs of the lungs showed multiple nodules, which were consistent with pulmonary metastases and confirmed by cytology and postmortem examination. The cat was presented to the OSU Oncology Service for humane euthanasia.
PH was a 13-year-old female intact domestic long-haired cat that presented for complete necropsy following euthanasia. She had a history of swollen tender mammary glands and was diagnosed with mammary carcinoma in 2010. Three months later, the cat had a left cranial mammary tumor of 7 × 4 cm and increased respiratory effort. The lungs, thoracic pleura, pericardial sac, diaphragm, right kidney, and adrenal glands contained numerous 1- to 10-mm metastases.
LTC was an 11-year-old female mixed short-haired cat that presented to the OSU VMC oncology service for excisional biopsy of a 7 × 5 cm mammary mass.
SB was a 3-year-old mixed short-haired female cat that presented with mammary gland masses and pulmonary metastases. The largest tumor was 1.5 × 1 cm.
MM was an 18-year-old mixed medium-haired female cat that presented for excisional biopsy of an 8 × 7 cm mammary mass.
Feline Mammary Cancer Cell Lines
Six FMC cell lines (FMCm, FONp, FONm, FKNp, FYMp, and FNNm) were obtained as a generous donation from Dr Ryohei Nishimura (Laboratory of Veterinary Surgery, Graduate School of Agricultural and Life Sciences, The University of Tokyo, Japan).55 Three of these lines were established from primary neoplasms, and 3 were established from metastases from 5 cats with spontaneous FMC. Capital letters such as FMC indicate identical patients, and lowercase letters such as p and m indicate primary and metastatic neoplasms, respectively. The cat breed, age, sex, and origin of the cell lines are presented in Table 1.55 Cells were cultured in RPMI 1640 (Life Technologies, Grand Island, NY) supplemented with 10% fetal bovine serum (FBS) (Life Technologies), 2 mM L-glutamine, and 0.25 ml gentamicin sulfate solution. Cells were maintained in humidified incubators at 37°C and 5% CO2. Cells were verified to be mycoplasma-free before nude mouse injections using a mycoplasma detection kit (InvivoGen, San Diego, CA). Cells were harvested at 80% to 90% confluence using 0.25% trypsin (Life Technologies) and injected into nude mice by 3 different routes (subcutaneous, intratibial, and left cardiac ventricle). Viable cells (95%–100%) were counted using a Cell-ometer (Nexcelom, Bioscience, Lawrence, MA), resuspended in sterile Dulbecco’s phosphate-buffered saline (PBS) (Life Technologies), and kept on ice until injection.
Table 1.
Origin of Feline Mammary Cancer Cell Lines.
| Cell Line | Breed | Age, y | Sex | Origin |
|---|---|---|---|---|
| FMCm | Japanese domestic | 12 | Female | Lymph node metastasis |
| FYMp | Japanese domestic | 15 | Female | Primary tumor |
| FNNm | Siamese | 12 | Spayed (at 11 years) Female | Pleural effusion |
| FKNp | Persian | 12 | Female | Primary tumor |
| FONp | Siamese | 12 | Female | Primary tumor |
| FONm | Lymph node metastasis |
Lentiviral Luciferase Transduction
The 4 feline mammary tumor cell lines (FKNp, FMCm, FYMp, and FNNm) with the most rapid in vivo tumor growth were grown in 6-well tissue culture plates with RPMI 1640, 10% FBS, 2 mM L-glutamine, and 0.25 ml gentamicin sulfate. When the cells were 95% confluent, the medium was removed and each well was treated with 100 μl of luciferase-containing virus (VC 2192 ConcpLuc (VSV-G)), 1.9 ml of culture medium, and 1.6 μl of polybrene stock (8 μg/ml) (Sigma-Aldrich, St Louis, MO). YFP-Luc lentiviral particles were produced by transient triple transfection of 293 T cells (obtained from Dr Michael Lairmore, The Ohio State University, Columbus, OH) with 10 mg of packaging plasmid pCMVΔR8.2, 2 μg of envelope plasmid pMD.G (both obtained from Dr Kathleen Boris-Lawrie, The Ohio State University), and 10 mg of transfer plasmid pHIVSIN-Luc/YFP using calcium phosphate as previously described.54 Following centrifugation at 2700 rpm for 1 hour at 30°C, the plate was placed in a cell culture incubator at 37°C and 5% CO2 for 24 hours, after which the virus-containing medium was aspirated and replaced with fresh culture medium without virus. After 48 hours, the cells were detached with0.25% trypsin and grown in 25-cm2 flasks.
Subcutaneous Injection of Primary FMC Neoplasms and Cell Lines in Nude Mice
All animal experimental procedures were approved by The Ohio State University Institutional Laboratory Animal Care and Use Committee.
Ten and 15 female nude mice were injected subcutaneously with CM and JF mammary cancer tissue, respectively. Twelve female nude mice were injected subcutaneously with the 6 FMC cell lines. The dorsal thoracic skin was sanitized with 70% alcohol. A suspension of 5 × 106 cancer cells or small tumor pieces (0.5–1.0 mm) in 0.3 ml of PBS was injected subcutaneously between the shoulder blades using 25- or 18-gauge needles, respectively. Tumor volume was measured twice weekly in 3 dimensions using calipers and calculated using the following formula: Length × Width × Height × 1/2. Mice were weighed weekly.
Intratibial and Intracardiac Injection of FMC Cell Lines in Nude Mice
Intratibial.
Twelve female nude mice were anesthetized in an induction chamber with 3% isoflurane and maintained at 2.5% isoflurane. The limb was sanitized with 70% alcohol. The leg was held so that the knee joint was at a 90° angle, and a 27-gauge needle was introduced through the patellar ligament and into the tibial marrow space through the articular cartilage. Using a Hamilton syringe, 10 μl of sterile PBS containing a suspension of 50 000 FKNp Luc, FMCm Luc, FYMp Luc, or FNNm Luc cells (fastest growing of the 6 cell lines) was introduced into the marrow cavity (3 mice for each cell line). Tumor growth was monitored weekly using bioluminescent imaging (BLI).
Intracardiac (left cardiac ventricle).
Eight female and 4 male nude mice were induced at 3% isoflurane gas anesthesia and maintained at 2.5%. The mice were placed in dorsal recumbency, and their front limbs were placed perpendicular to their thorax and restrained to the procedure table with surgical tape. The ventral thorax was sanitized with 70% alcohol. A 1-ml syringe was loaded with 0.1 ml of PBS containing a suspension of 100 000 FKNp Luc, FMCm Luc, FYMp Luc, or FNNm Luc cells for female mice and 100 000 FMCm Luc cells for males. A syringe fitted with a 27-gauge needle was introduced into the left cardiac ventricle through the third intercostal space, approximately 1 mm to the left and lateral from the sternum. Once a pulsatile jet of blood was present in the hub of the needle, the cell suspension was slowly injected into the left ventricle of the heart over 30 seconds. Tumor metastasis was monitored weekly using BLI.
Bioluminescent Imaging (BLI)
Mice were anesthetized in an induction chamber with 3% isoflurane and maintained at 2% isoflurane. A 1-ml syringe was used to inject 0.1 ml of sterile PBS containing 4.5 mg D-Luciferin (Caliper Life Sciences, Hopkinton, MA) into the peritoneal cavity. Bioluminescent in vivo imaging was performed using the IVIS 100 (Caliper Life Sciences), and photon signal intensity was quantified using LivingImage software version 2.50 (Caliper Life Sciences). Imaging was performed every 2 minutes until peak photon signal was achieved (approximately 10 minutes after the injection). The IVIS 100 was set to 1-minute exposures with medium binning. The mice that received intracardiac injections were also imaged immediately after injection to confirm that the tumor cells were successfully placed into the left ventricle, which was demonstrated by generalized light emission from the entire body.
Radiography
Radiography was performed postmortem on the mice that had intratibial and intracardiac injections. Formalin-fixed tissues were placed centrally on a Faxitron laboratory radiography system LX-60 (Faxitron X-ray Corp., Wheeling, IL) imaging platform, and high-resolution digital radiographs were obtained at 26 KV and 5.5-second exposures.
Postmortem and Histopathological Examination
After euthanasia, a complete necropsy was performed and tissues were collected and sectioned to confirm metastases. All neoplasms were weighed and then divided in half for both molecular analysis and histopathologic examination. Half of each tumor was frozen in liquid nitrogen, and the other half was fixed for 48 hours in 10% neutral-buffered formalin, embedded in paraffin, sectioned, and stained with hematoxylin and eosin (HE).
RNA Extraction and Quantitative Real-Time Reverse Transcription Polymerase Chain Reaction
Total RNA was extracted from both FMC neoplasms and cell lines using the QuickGene RNA cultured cell HC kit S (Kurabo, Catalog No. RC-S2, Holliston, MA). Total RNA was reverse-transcribed using the Superscript II First Strand cDNA synthesis kit (Invitrogen, Carlsbad, CA), and quantitative real-time reverse transcription polymerase chain reaction (qRT-PCR) was performed using a LightCycler 480 (Roche, Indianapolis, IN) for the housekeeping genes glyceraldehyde 3-phosphate dehydrogenase (GAPDH) and ubiquitin C (UBC), as well as the following genes related to lymphangiogenesis, angiogenesis, tumor progression, and metastasis: vascular endothelial growth factors C and D (VEGFC and VEGFD), vascular endothelial growth factor receptor-3 (VEGFR-3), platelet-derived growth factors A through D (PDGFA-PDGFD), fibroblast growth factor 2 (FGF2), angiopoietin 1 and 2 (ANGPT1 and ANGPT2), epidermal growth factor receptor family (EGFR, ERBB2, and ERBB3), myoferlin (MYOF), and parathyroid hormone-related protein (PTHrP). The qRT-PCR results were analyzed using the LightCycler 480 Software. The primers were chosen from 3 or 4 different primer pairs designed for each gene, and the primer pair that had the best amplification and qRT-PCR product-melting characteristics for each gene was chosen (Suppl. Table 1). The primer pairs were designed using Primer-BLAST software (http://www.ncbi.nlm.nih.gov/tools/primer-blast). To confirm primer specificity, all qRT-PCR products were verified by electrophoresis on a 2% agarose gel and stained with ethidium bromide to confirm a single amplification product of the expected size. The entire PCR reactions were then purified using the QIAquick PCR Purification Kit (Qiagen, Valencia, CA, Catalog No. 28106) and sequenced at the Plant-Microbe Genomics Facility at The Ohio State University using a 3730 DNA Analyzer (Applied Biosystems, Grand Island, NY) and BigDye Terminator Cycle Sequencing chemistry (Applied Biosystems). Sequences were verified by a BLAST search using the NCBI website.
Statistical Analysis
Data from qRT-PCR were analyzed using Graph Pad Prism 6.0 software (San Diego, CA). All samples were repeated in triplicate. Normal distribution of the data for the different variables was assessed using the Kolmogorov-Smirnov test. The qRT-PCR data from subcutaneous xenografts were statistically compared with the primary FMC tumor data using a paired Student’s t-test for normally distributed data and Wilcoxon signed rank test for skewed data. The qRT-PCR data from the FMC cell lines were statistically compared with the primary tumor group data using Student’s t-test for normally distributed data and Mann-Whitney test for skewed data. A P value <.05 was considered to be statistically significant.
Results
Histopathology of FMC Neoplasms
Primary FMC in the cats.
The FMC neoplasms were graded as described for cat mammary cancer.15,35
CM consisted of a poorly differentiated (grade III) solid mammary carcinoma with multinodular masses and cystic spaces with production of mucoid material (Fig. 1). There was abundant central necrosis within cystic lobules. The neoplastic cells were severely infiltrative and vascular invasion was present.
Figure 1.
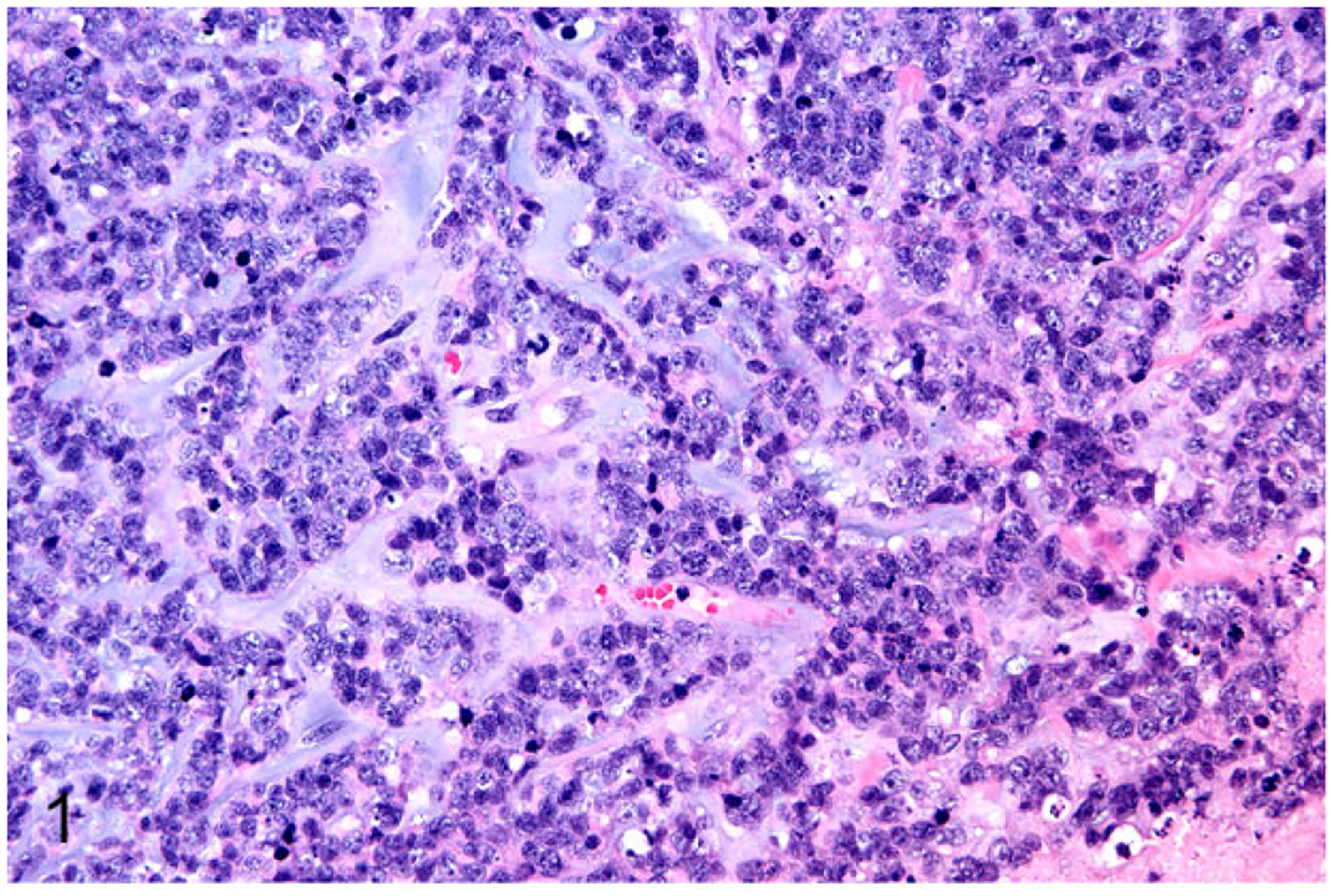
Primary mammary gland carcinoma (patient CM), cat. The neoplasm was a poorly differentiated solid carcinoma with production of mucoid material. Hematoxylin and eosin.
JF was a well-differentiated (grade I) tubulopapillary mammary carcinoma (Fig. 2) with areas of necrosis and papillary structures.
Figure 2.
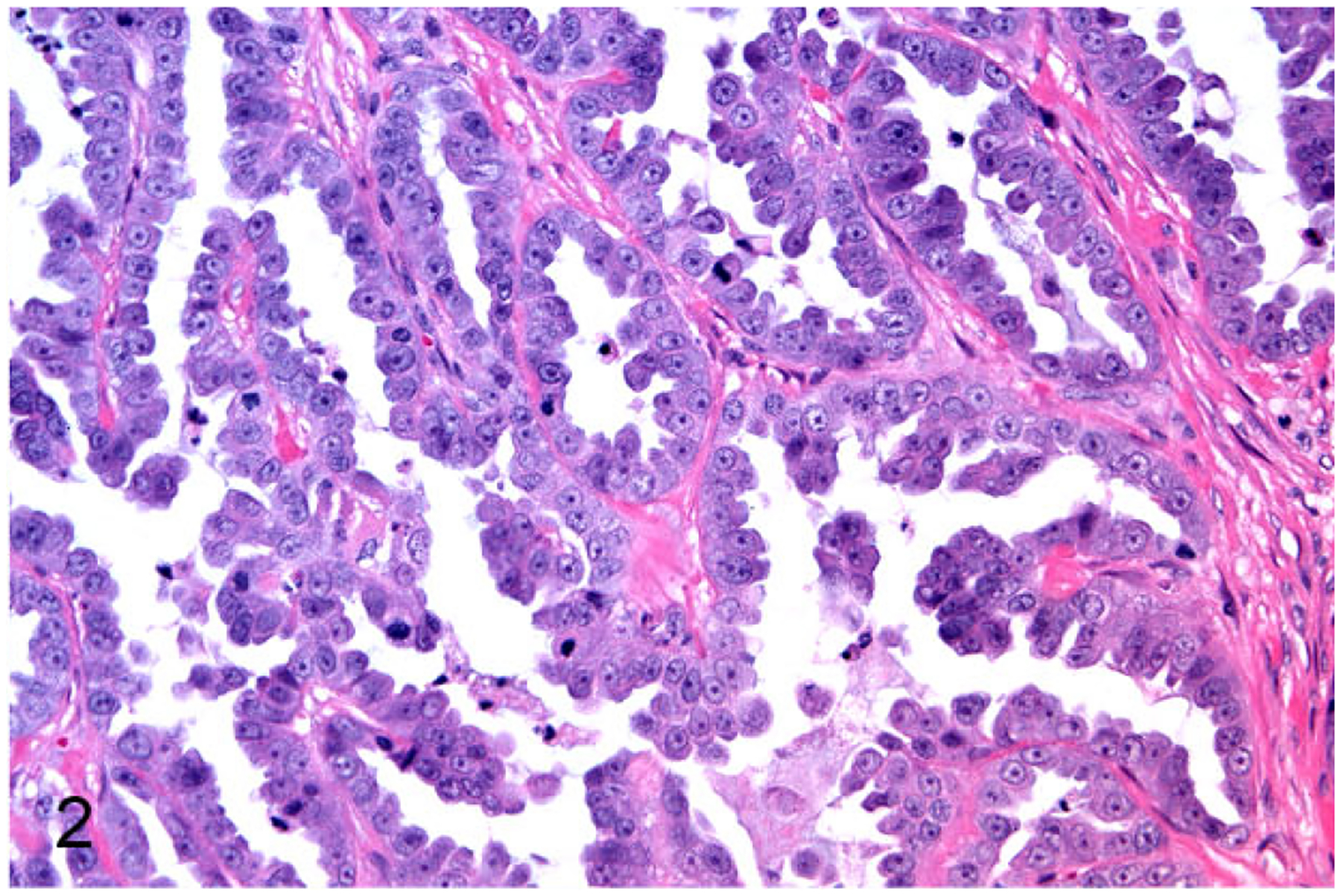
Primary mammary gland carcinoma (patient JF), cat. The neoplasm was a well-differentiated, tubulopapillary carcinoma with papillary projections.
PH was a poorly differentiated (grade III) tubulopapillary mammary carcinoma with extensive necrosis and vascular invasion.
LTC was a moderately differentiated (grade II) tubulopapillary mammary carcinoma with desmoplasia and local tissue invasion with lymphoplasmacytic inflammation.
SB was a well-differentiated (grade I) tubulopapillary mammary carcinoma. There was local invasion into the subcutis and skeletal muscle and vascular invasion with central coagulation necrosis and reactive fibrosis.
MM was a moderately differentiated (grade II) cribriform mammary carcinoma. The tumor had marked tissue invasion with multifocal necrosis and reactive fibrosis.
Nude mouse FMC subcutaneous xenografts.
The CM and JF primary FMC neoplasms were serially passaged (3 times) as subcutaneous neoplasms in nude mice. CM and JF were grossly visible in the transplanted sites of all mice between 2 and 4 months after implantation. Mice were euthanatized starting 8 weeks after implantation. The histologic appearance of the xenografts was similar to that of the primary neoplasms (Figs. 3 and 4). The tumors lines were not invasive and were easily dissectible from the subcutaneous space. CM and JF tumor lines did not metastasize following subcutaneous injection.
Figure 3.
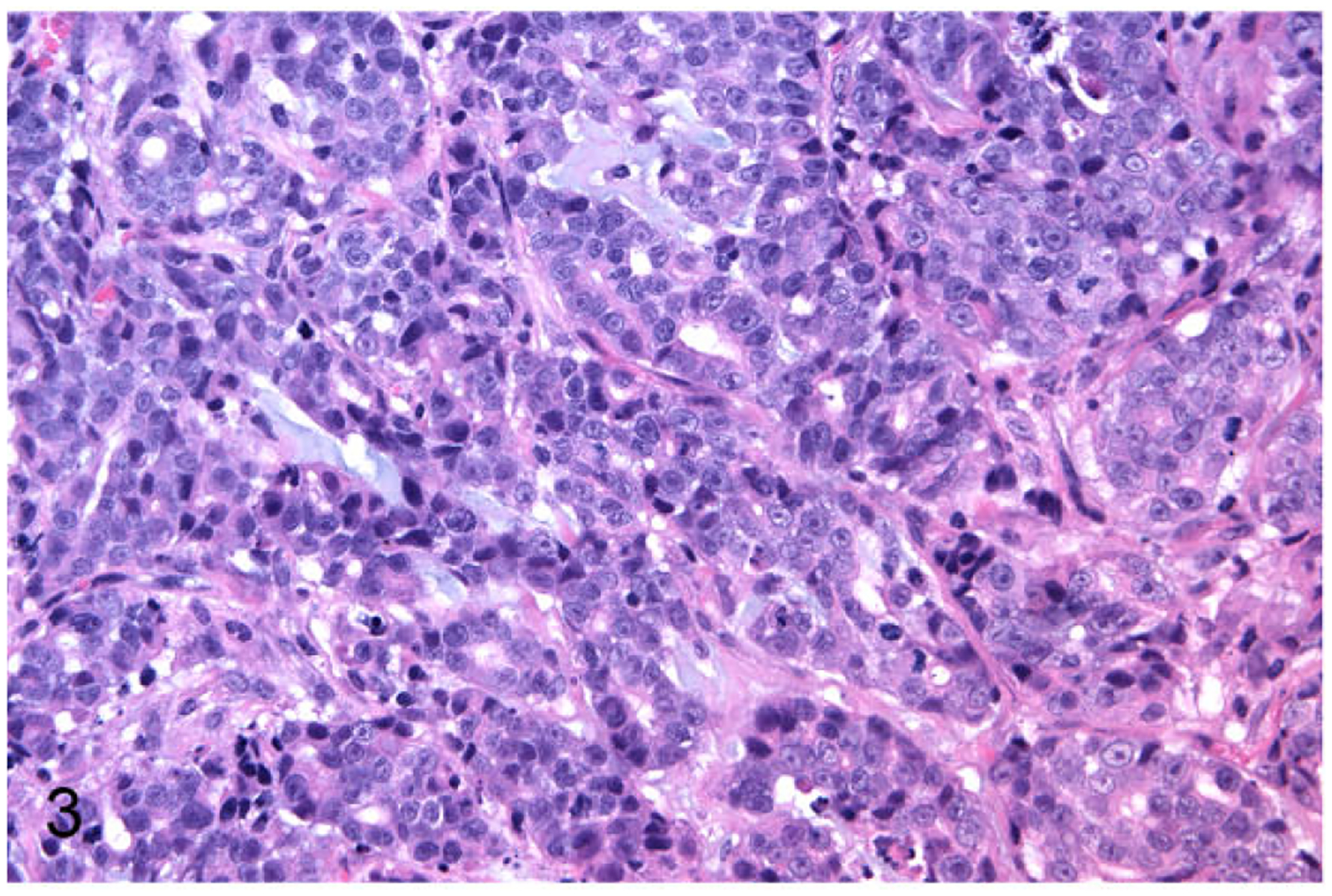
Mammary gland carcinoma (patient CM), nude mouse subcutaneous xenograft. The xenograft was a tubulopapillary carcinoma with mucoid material similar to the primary cancer in the cat.
Figure 4.
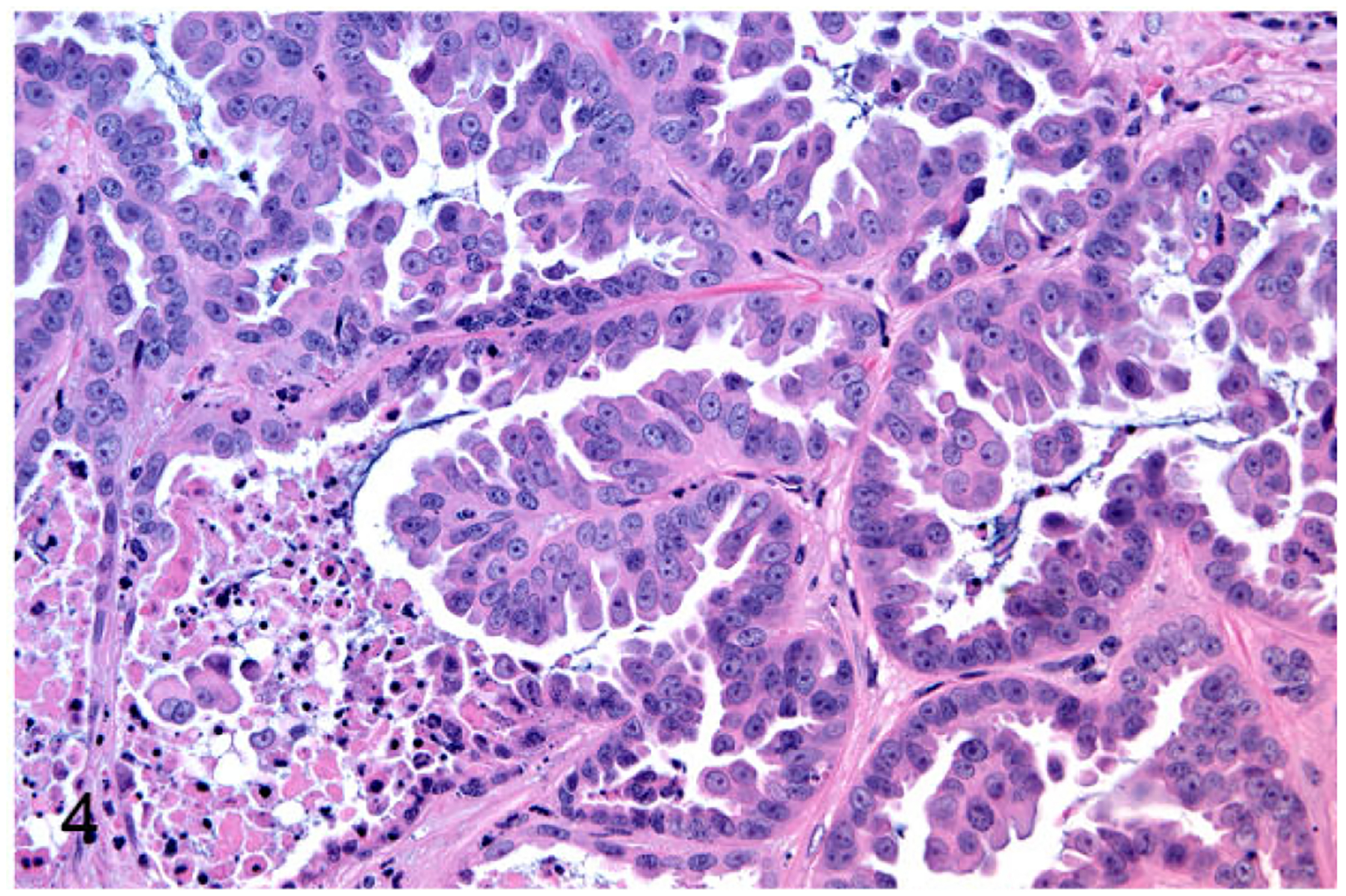
Mammary gland carcinoma (patient JF), nude mouse subcutaneous xenograft. The xenograft was a tubulopapillary carcinoma similar to the primary cancer in the cat.
The FMC cell line subcutaneous xenografts were grossly visible at 2 to 4 weeks after implantation for all the cell lines (FONp and FONm had the slowest growth rate). Mice were euthanatized from 3 to 10 weeks after implantation, and tumors ranged in size from 0.5 to 3.4 cm3. The subcutaneous tumors were not invasive and were easily dissectible. The histopathologic and morphologic characteristics of the FMC cell lines were different depending on the cell line in the subcutaneous tumors as well as in culture (Suppl. Figs. 1–6). No metastases were observed after subcutaneous injection of the 6 FMC cell lines.
FMCm was a poorly differentiated (grade III) tubulopapillary carcinoma with extensive necrosis and mild fibrosis (Fig.5). FYMp was poorly differentiated (grade III) and had 2 populations of neoplastic cells: squamous cell carcinoma and tubulopapillary carcinoma (Fig. 6). FNNm was a poorly differentiated (grade III) solid carcinoma with reactive fibrous stroma (Fig. 7). FKNp was a moderately differentiated (gradeII) basosquamous carcinoma (Fig. 8). FONp was a well-differentiated (grade I) cystic tubulopapillary carcinoma (Fig.9). FONm was a well-differentiated (grade I) basosquamous carcinoma (Fig. 10). Growth curves of the FMC cell line subcutaneous xenografts in nude mice are presented in Fig. 11.
Figure 5.
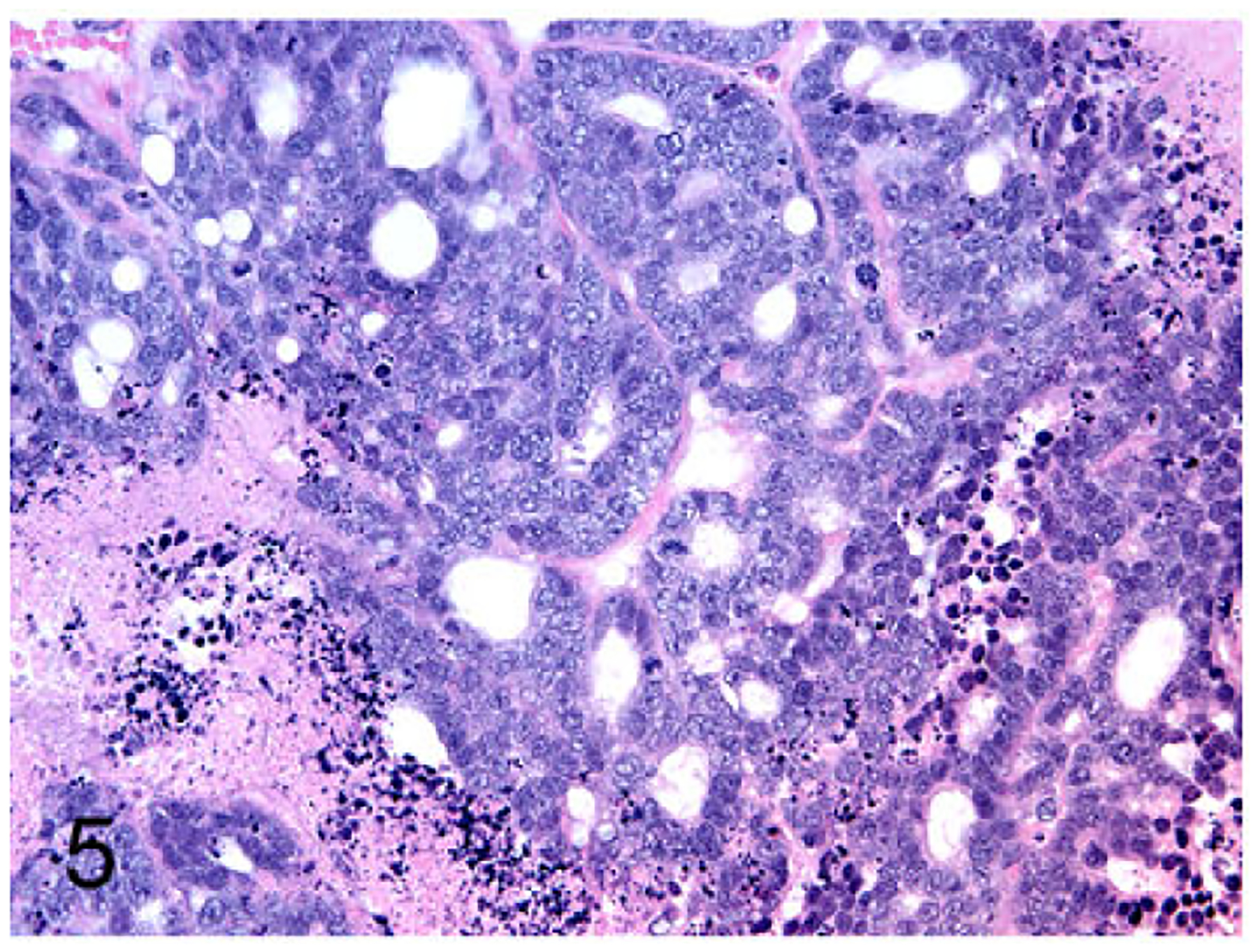
Mammary gland carcinoma (FMCm cell line), nude mouse subcutaneous xenograft. The xenograft was a poorly differentiated tubulopapillary carcinoma with extensive necrosis and mild fibrosis.
Figure 6.
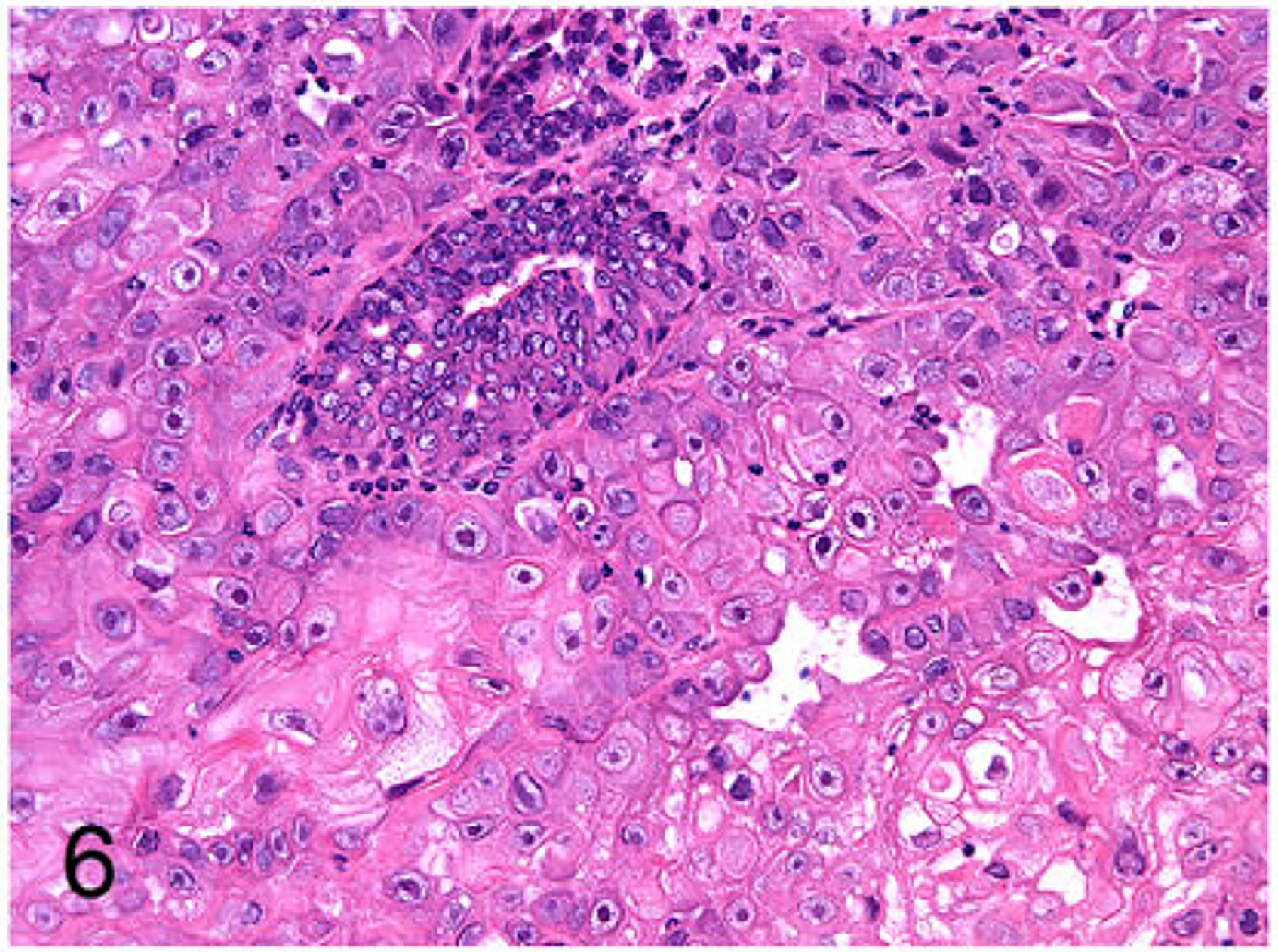
Mammary gland carcinoma (FYMp cell line), nude mouse subcutaneous xenograft. The xenograft consisted of 2 populations of cells: squamous cell carcinoma and tubulopapillary carcinoma.
Figure 7.
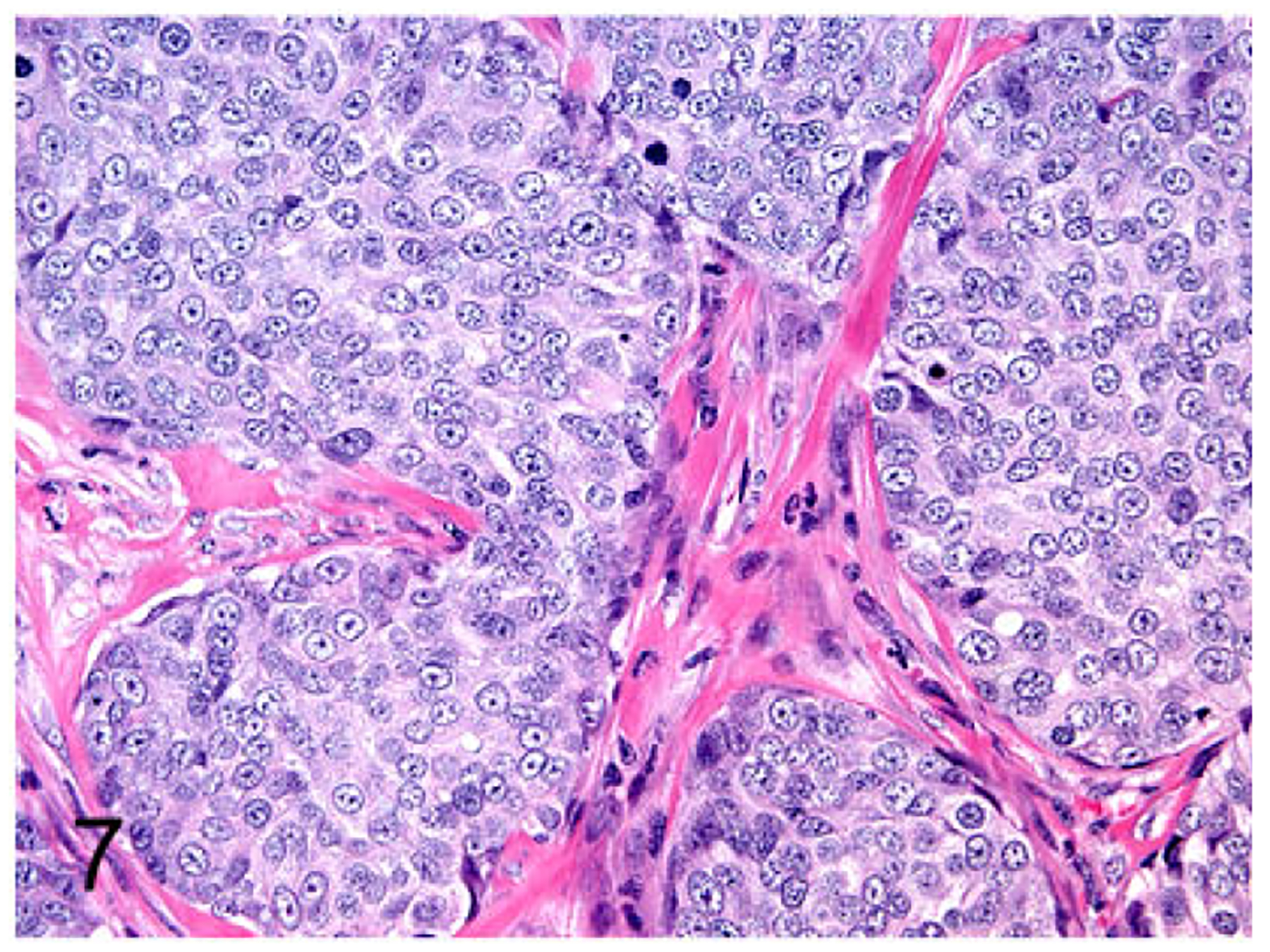
Mammary gland carcinoma (FNNm cell line), nude mouse subcutaneous xenograft. The xenograft was a poorly differentiated solid carcinoma with reactive fibrous tissue.
Figure 8.
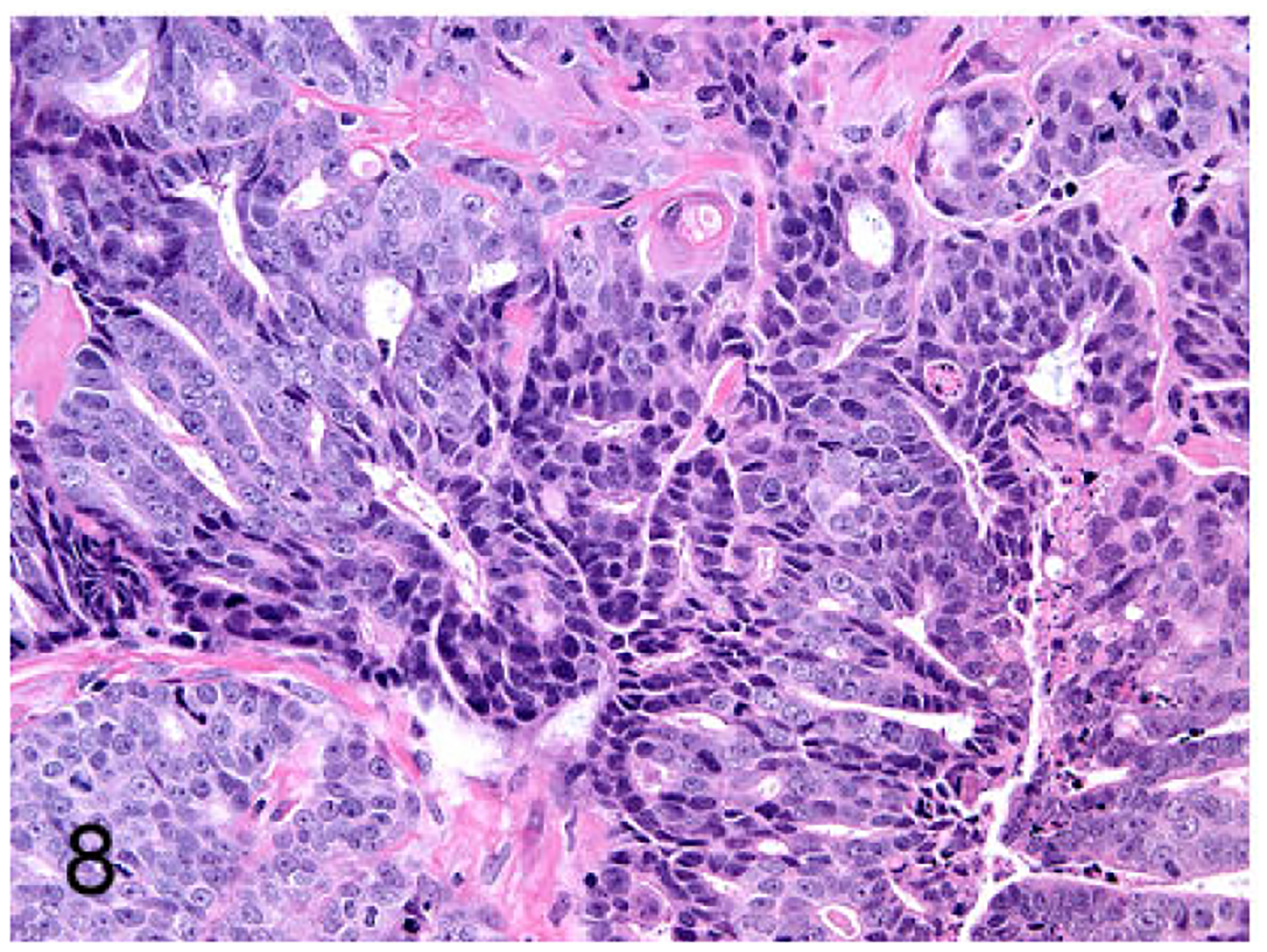
Mammary gland carcinoma (FKNp cell line), nude mouse subcutaneous xenograft. The xenograft was a moderately differentiated basosquamous carcinoma.
Figure 9.
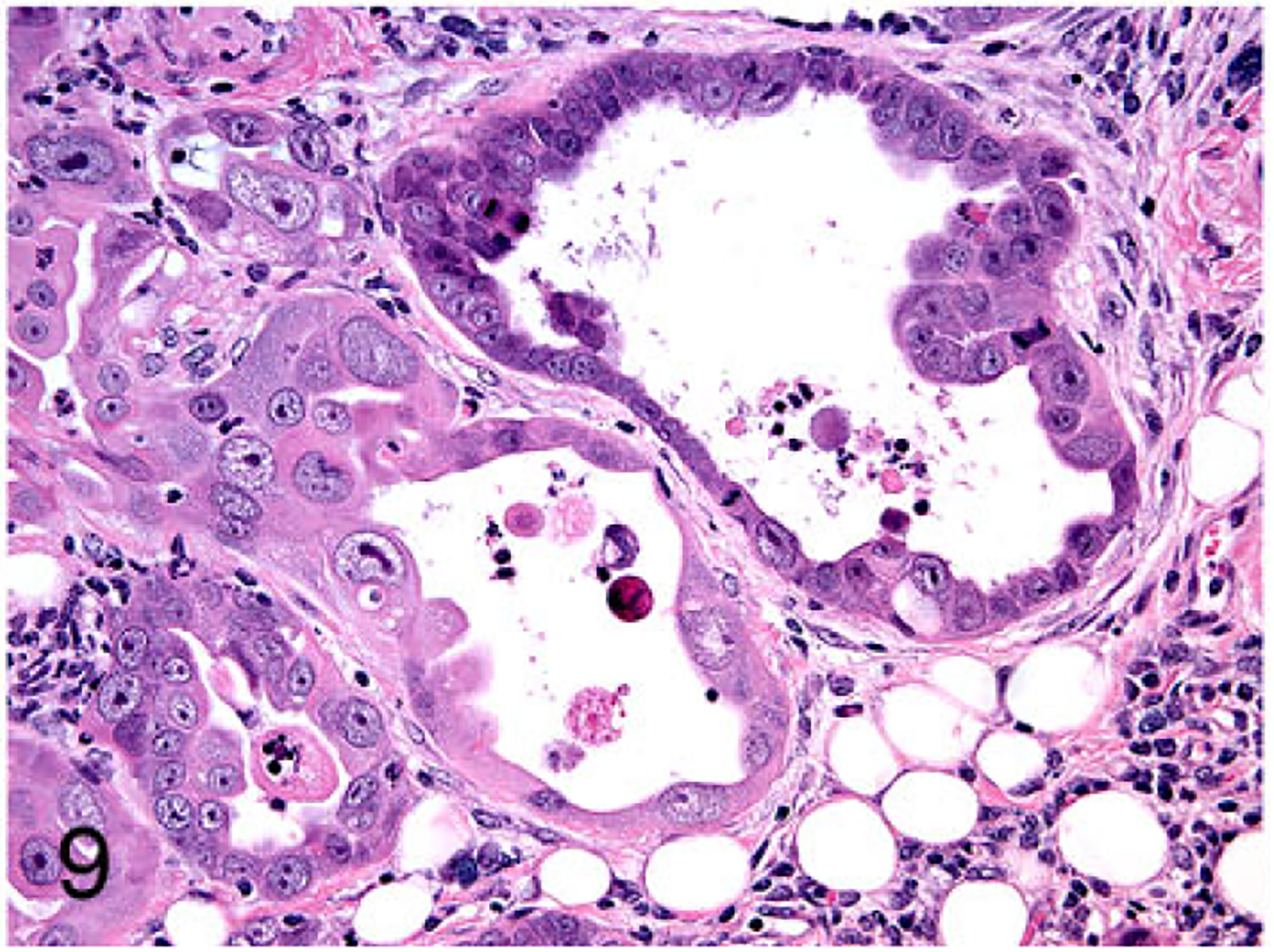
Mammary gland carcinoma (FONp cell line), nude mouse subcutaneous xenograft. The xenograft was a well-differentiated tubulopapillary carcinoma.
Figure 10.
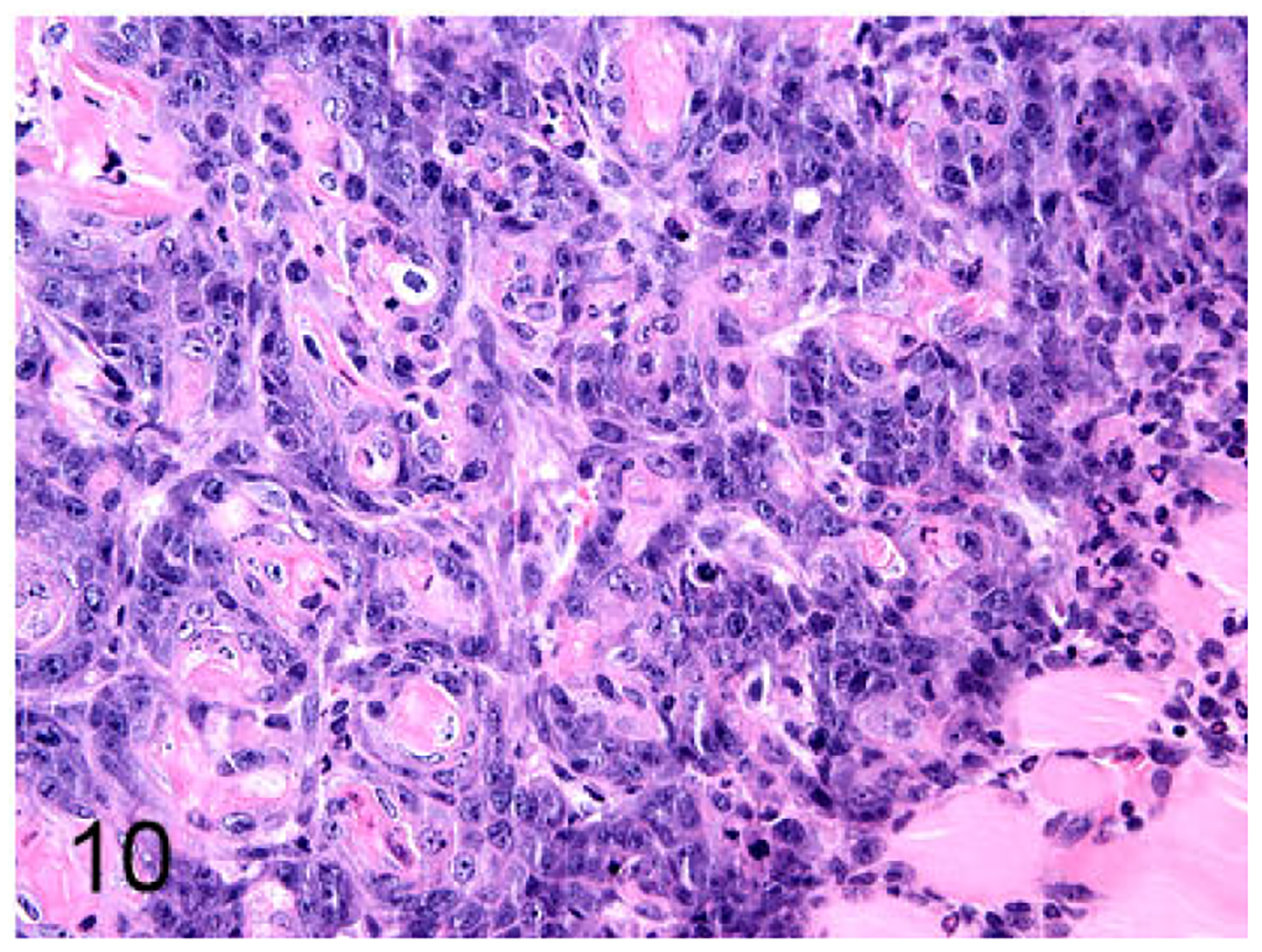
Mammary gland carcinoma (FONm cell line), nude mouse subcutaneous xenograft. The xenograft was a well-differentiated basosquamous carcinoma.
Figure 11.
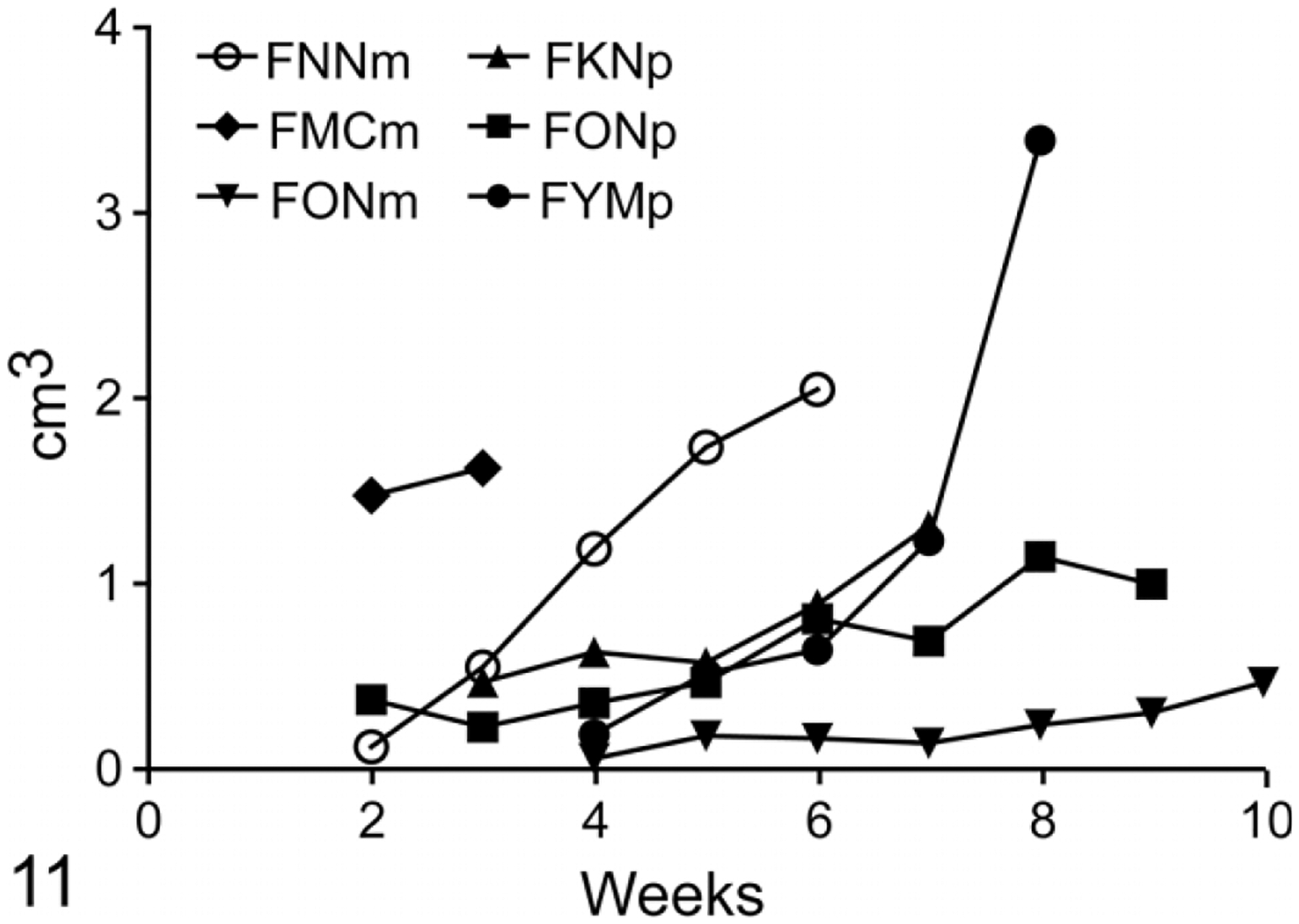
Growth curves of feline mammary cancer (FMC) cell lines as subcutaneous xenografts in nude mice. FMCm, FNNm, FKNp, and FYMp had a high proliferation rate, while FONp and FONm had a low proliferation rate.
Intratibial Injections and Bioluminescence of FMC Cell Lines in Nude Mice
Four of 12 (33%) mice developed intratibial neoplasms: 2 with FMCm Luc, 1 with FKNP Luc, and 1 with FYMp Luc. Periosteal tumor growth was present in 1 mouse with the FMCm Luc cells likely due to injection of tumor cells outside of the tibia. BLI was effective at monitoring the presence and growth of intratibial neoplasms, and the FMCm Luc neoplasms had the most intense signals with a minimum of 1.2 × 107 and a maximum of 1.3 × 108 photons per square centimeter. Faxitron high-resolution radiographs demonstrated either osteosclerotic bone metastases (Fig. 12) or mixed osteolytic-osteoblastic bone metastases (Fig. 13) in the tibias. Histopathology of the osteosclerotic neoplasms demonstrated new woven bone formation in the medullary cavity with hypertrophied osteoblasts in close association with the FMC cells (Figs. 14 and 15). The intratibial neoplasms formed solid sheets of cells within the medullary cavity of the metaphysis or diaphysis. Two of 3 mice injected intratibially with FMCm Luc cells developed lung metastases (ranging from 14 to 36 metastases per section and 70 to 560 μm in diameter).
Figure 12.
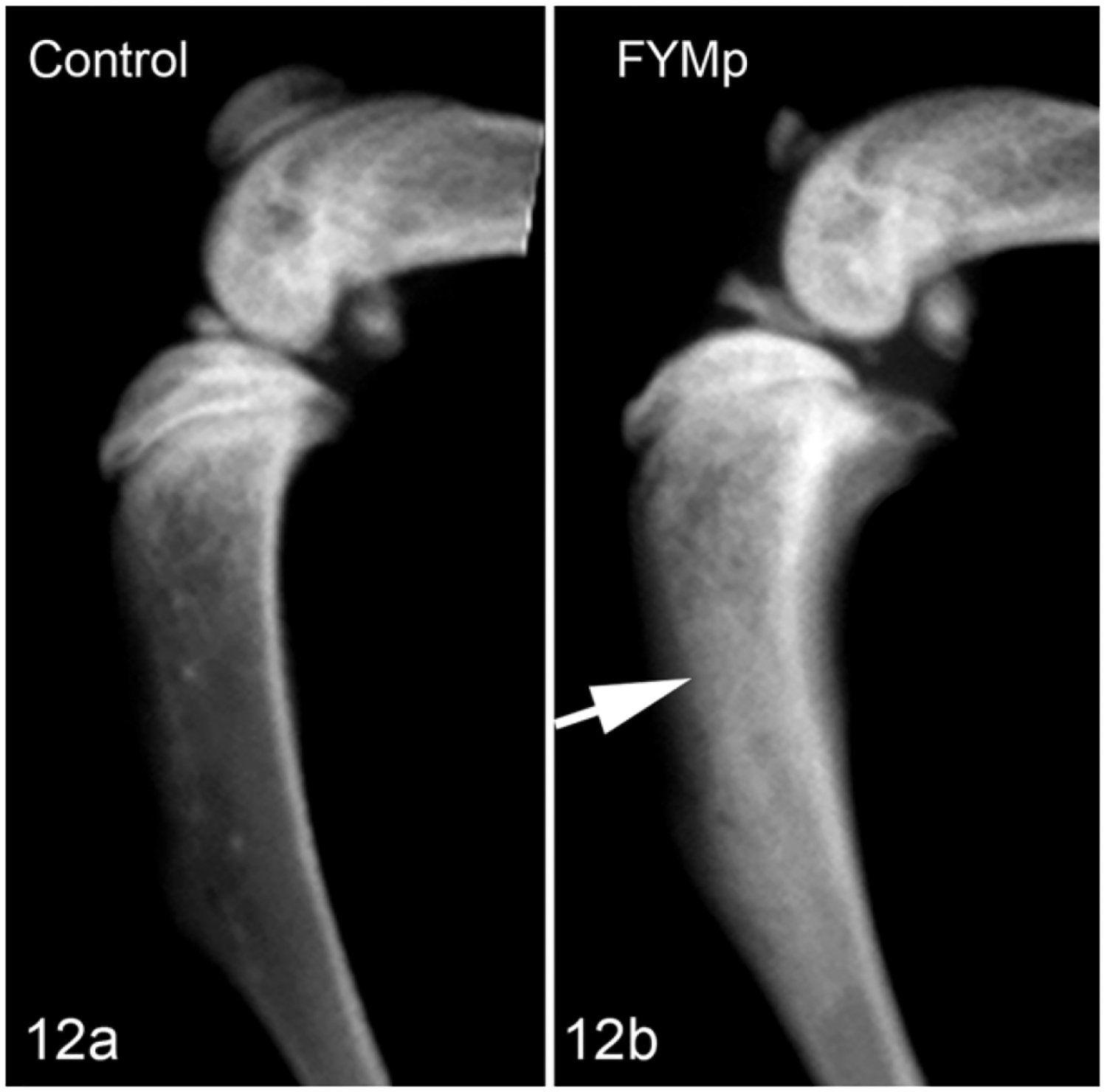
Tibia, Nude mouse. Radiograph. (a) Tibia without injection used as control. (b) The FYMp Luc cells in the tibia induced abundant new woven bone formation in the metaphysis and diaphysis (“osteosclerotic” metastasis) (arrow).
Figure 13.
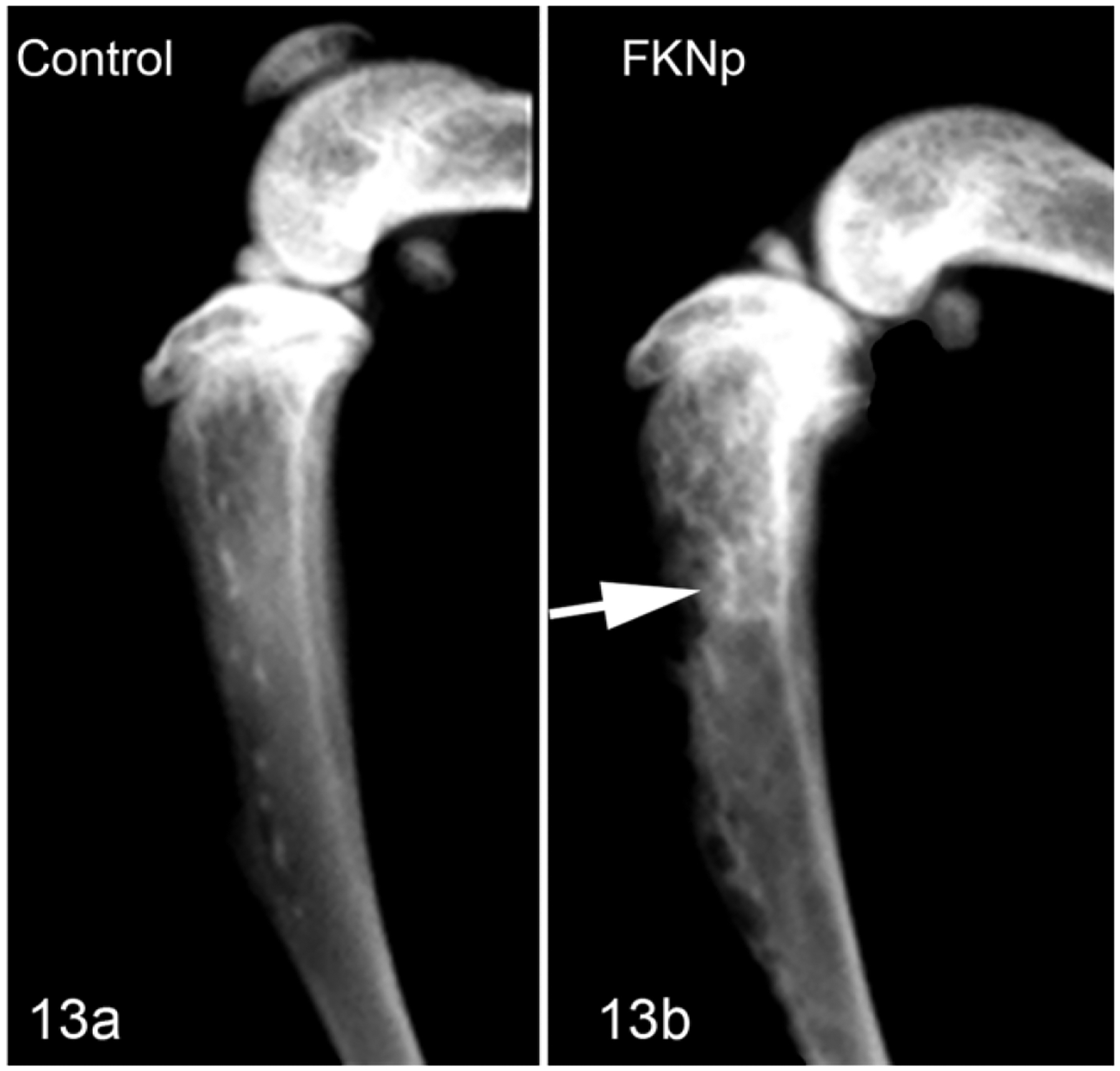
Tibia, Nude mouse. Radiograph. (a) Tibia without injection used as control. (b) The FKNp Luc cells in the tibia marrow space induced mixed osteolytic/osteoblastic bone metastases in the metaphysis and diaphysis (arrow).
Figure 14.

Tibia, Nude mouse. FYMp Luc cell line. New woven bone formation was induced by the mammary cancer within the medullary cavity of the diaphysis and along the endosteal surfaces. Hematoxylin and eosin.
Figure 15.
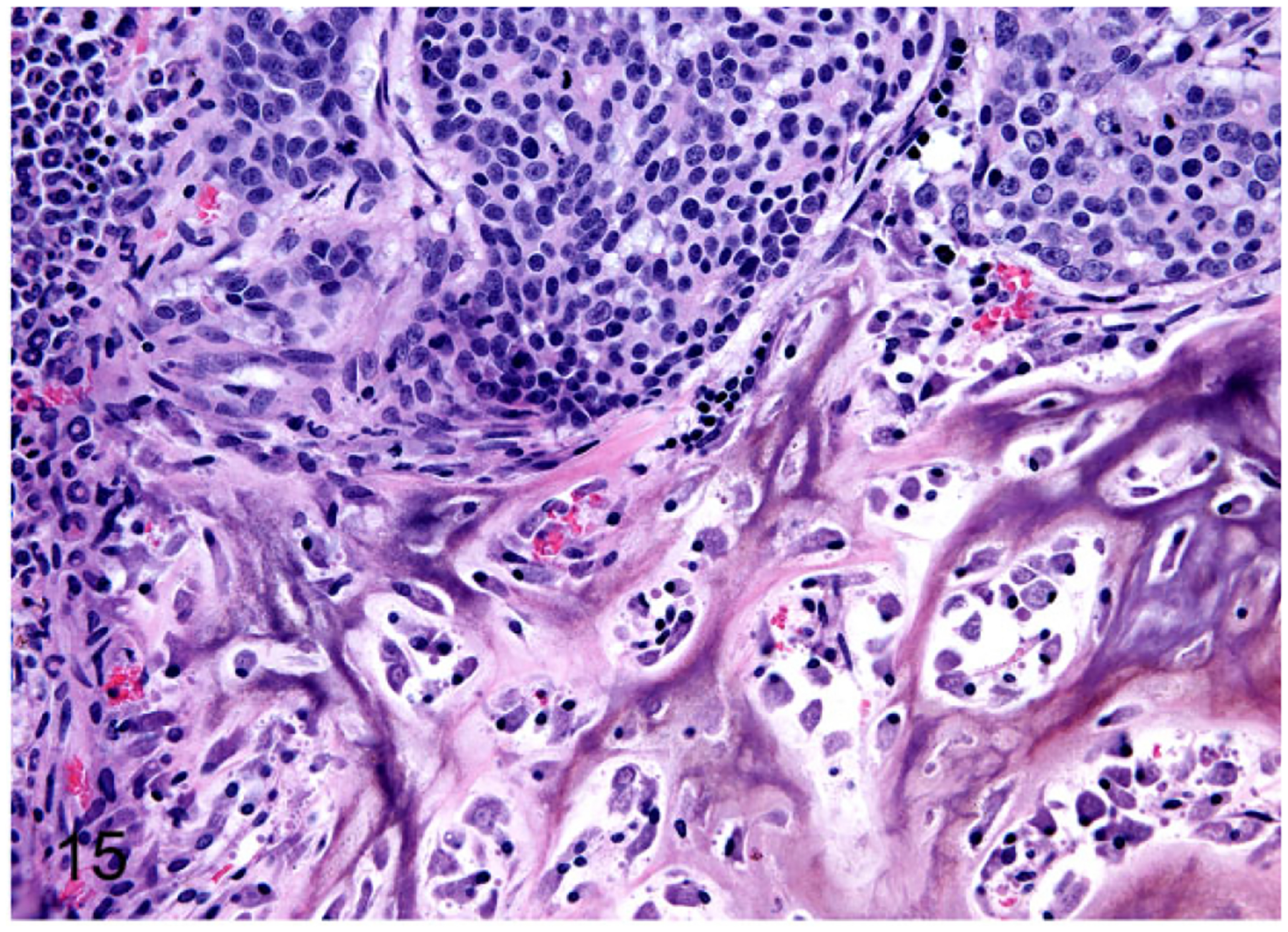
Tibia, Nude mouse. FMCm Luc cell line. New woven bone formation was induced by the mammary cancer (top of image) within the medullary cavity.
Intracardiac Injections and Bioluminescence of FMC Cell Lines in Nude Mice
Four of 7 (57%) female mice (1 mouse died on the same day of injection) were successfully injected into the left ventricle of the heart. Successful injections were characterized by bioluminescence diffusely distributed throughout the body at 5 minutes after injection of FMC cells. At 2 weeks, the mice injected with FMCm Luc cells had bioluminescence within the mandible, and at week 3 these mice had metastases in the brain and the hind limbs. One mouse injected with FKNp Luc cells had bioluminescence within the mandible. The 2 mice injected with FMCm Luc cells developed lung metastases (ranging from 1 to 2 metastases per section and 160 to 470 μm in diameter) (Fig. 16) and brain metastases (ranging from 1 to 5 metastases per section and 90 to 470 mm in diameter) (Fig. 17). The intensity of the BLI increased from week 3 to 4, indicating progressive tumor growth. At the end of week 4, metastases were present in the lungs, brain, mandible, eyes, and long bones of the hind limbs of the 2 mice injected with FMCm Luc cells.
Figure 16.
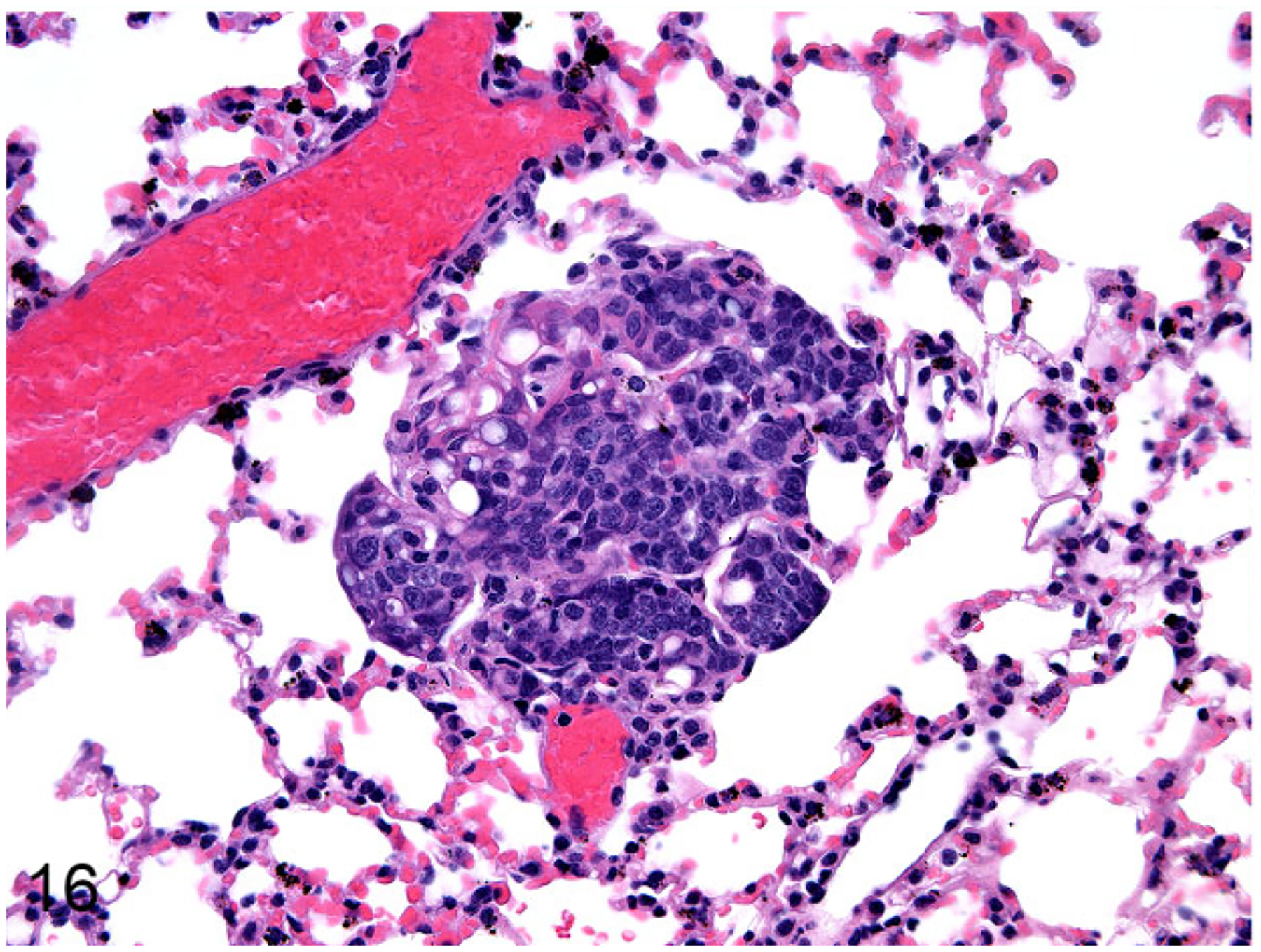
Lung, Nude mouse. FMCm Luc lung metastasis. The lung metastasis had an epithelial phenotype.
Figure 17.
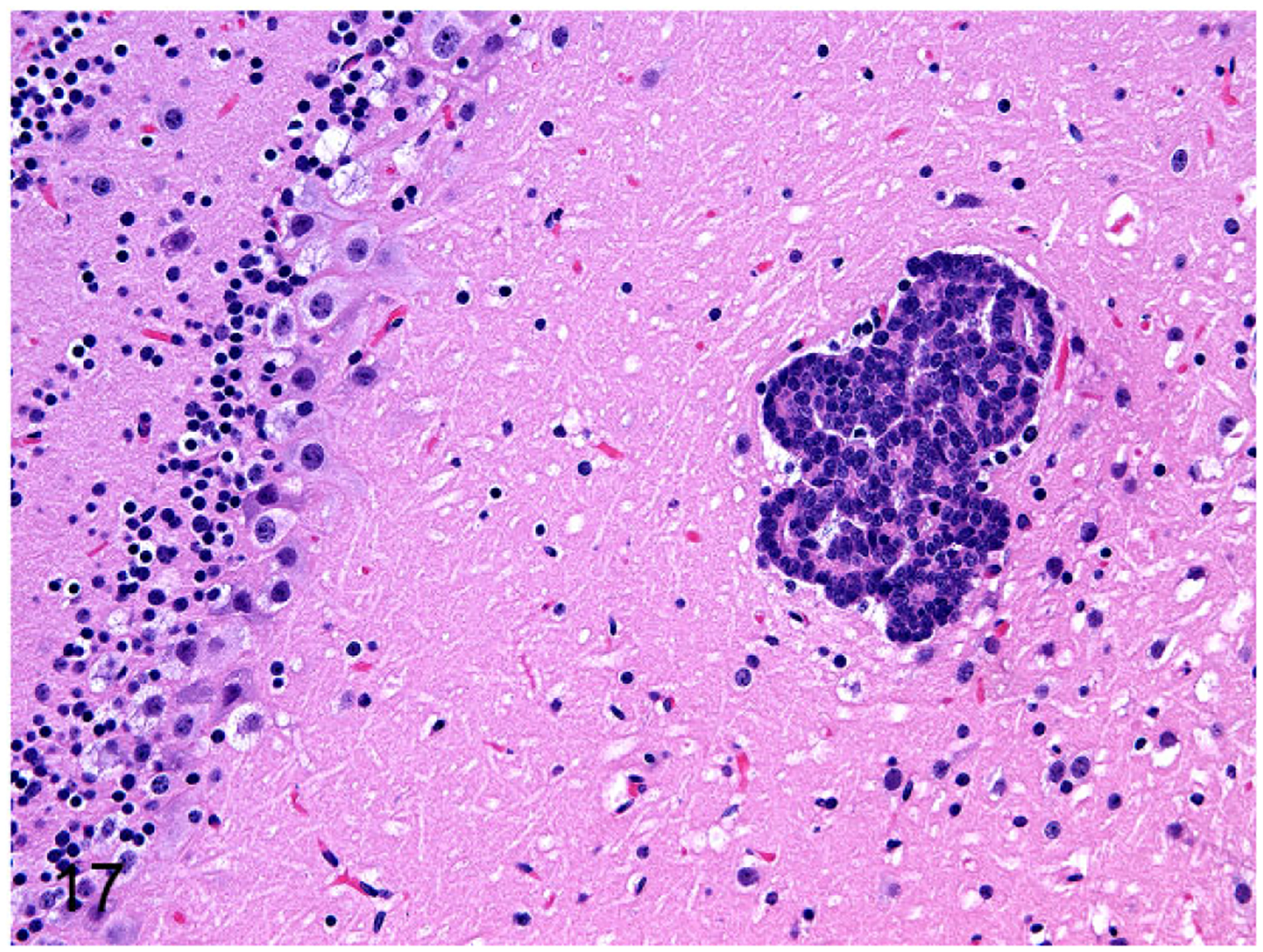
Brain, Nude mouse. FMCm Luc brain metastasis.
Four of 4 male mice were successfully injected with FMCm Luc cells into the left ventricle of the heart. Successful injections were characterized by bioluminescence diffusely distributed throughout the body at 5 minutes after injection. At 2 weeks, the mice had bioluminescence within the mandible, lungs, and hind limbs. The intensity of the BLI increased from week 3 to 5, indicating increased tumor burden. At the end of week 5, metastases were present in mandible, eyes, brain, lungs, kidneys, liver, lumbar vertebrae, and hind limbs. In vivo tumor growth and metastases are summarized in Table 2.
Table 2.
In Vivo Growth and Metastasis of Feline Mammary Cancer Cell Lines After Injection Into the Subcutis, Tibia, or Left Ventricle of the Heart (Intracardiac) of Nude Mice.
| Cell Line | Subcutis | Intratibial | Intracardiac |
|---|---|---|---|
| FMCm | 2/2 | 2/3 Tibia with lung metastases | Female mice: |
| 2/2 Lung, brain, long bones, and mandible | |||
| 1/2 Eyes | |||
| Male mice: | |||
| 4/4 Lungs and kidneys | |||
| 3/4 Brain, eyes, long bones, and mandible | |||
| 2/4 Spinal cord and liver | |||
| FYMp | 2/2 | 1/3 Tibia | 0/2 |
| FNNm | 2/2 | 0/3 | 0/2 |
| FKNp | 2/2 | 2/3 Tibia | 1/2 Mandible |
| FONp | 2/2 | n/d | n/d |
| FONm | 1/2 | n/d | n/d |
Abbreviation: n/d, not done.
qRT-PCR of Primary FMC, Xenografts, and Cell Lines in Nude Mice
This study examined the expression of 15 mRNAs (PDGFA, PDGFB, PDGFC, PDGFD, FGF2, EGFR, ERBB2, ERBB3, VEGFC, VEGFD, VEGFR3, ANGPT1, ANGPT2, PTHrP, and MYOF) in all feline mammary neoplasms, mouse xenografts, and cell lines. The FMC cell lines expressed higher levels of PDGFA (threshold cycle [Ct] = 22) (Fig. 18), PDGFB (Ct = 23) (Fig. 19), EGFR (Ct = 23) (Fig. 20), and VEGFR-3 (Ct = 24) (Fig. 21) compared with the primary FMC neoplasms and subcutaneous xenografts that had the following expression levels: PDGFA (Ct = 25), PDGFB (Ct = 27), EGFR (Ct = 27), and VEGFR3 (Ct = 27.5–29). All the samples had high expression of PDGFC (Ct = 22–24) (Fig. 22), ERBB3 (Ct = 20–22) (Fig. 23), ERBB2 (Ct = 23.7–25) (Fig. 24), and MYOF (Ct = 22–26.8) (Fig. 25) and moderate expression of FGF2 (Ct = 27–29) (Fig. 26) and VEGFD (Ct = 27–28) (Fig. 27). All the samples had low expression of ANGPT1 (Ct = 29–33) (Fig. 28) and PTHrP (Ct 30.6–31) (Fig. 29). There was no expression of PDGFD, ANGPT2, and VEGFC in either the subcutaneous xenografts or the cell lines, in contrast to the primary FMC neoplasms, which expressed high levels of these mRNAs (Ct = 26 for each). Therefore, we designed mouse-specific primers for PDGFD, ANGPT2, and VEGFC in order to determine whether the mouse stroma in the xenografts could express these genes. Interestingly, there was moderate expression of mouse PDGFD (Ct = 30), ANGPT2 (Ct = 29), and VEGFC (Ct = 29) in the subcutaneous xenografts, indicating that mouse stroma was expressing these genes within the tumor, rather than expression by the tumor cells themselves (Figs. 30–32). This may also be the case in the primary FMC neoplasms, but this could not be confirmed in this investigation since the primary neoplasms contain feline stroma and tumor cells.
Figures 18–29.
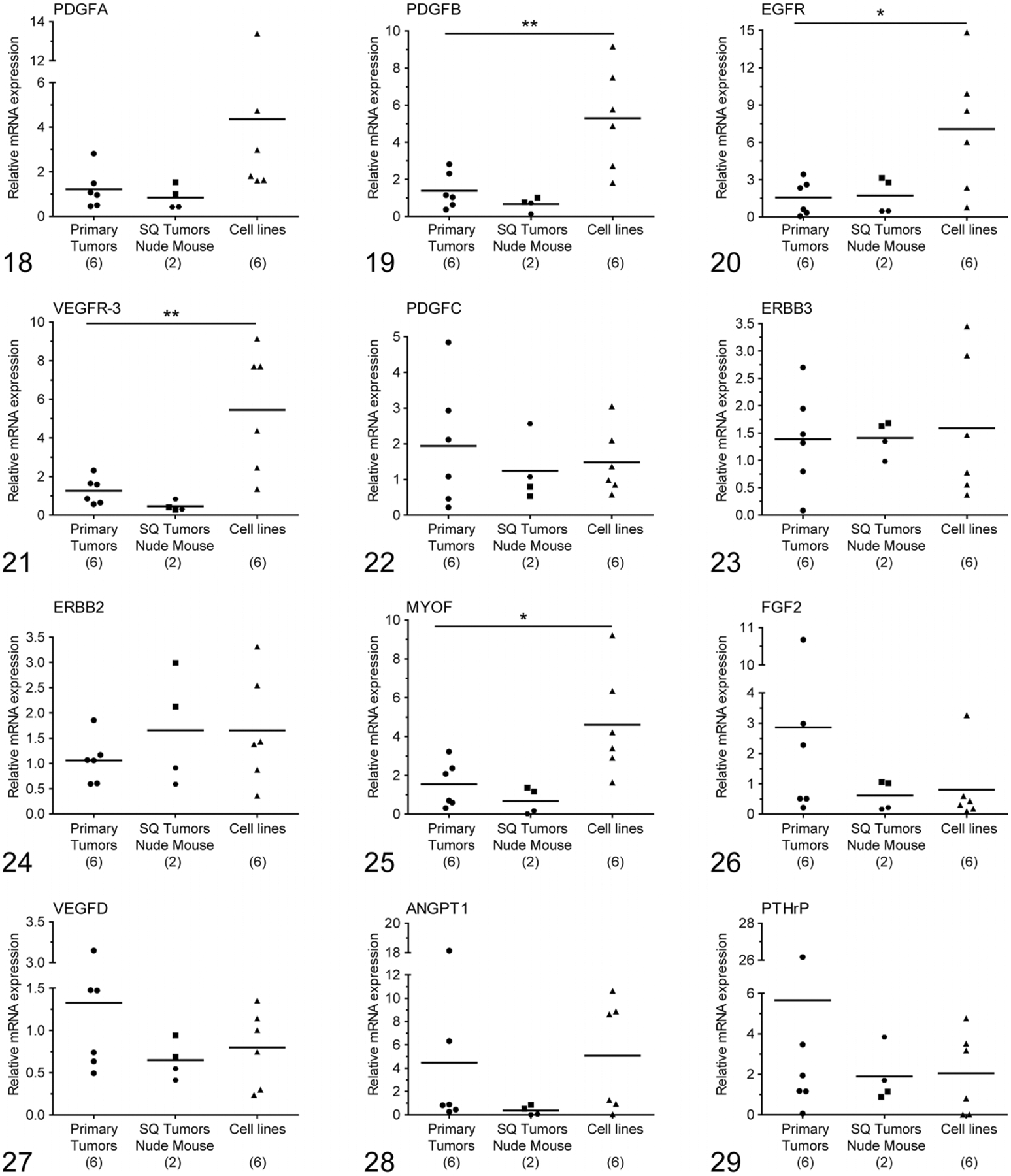
The relative mRNA expression of platelet-derived growth factor A (PDGFA), platelet-derived growth factor B (PDGFB), epidermal growth factor receptor (EGFR), vascular endothelial growth factor receptor-3 (VEGFR-3), platelet-derived growth factor C (PDGFC), epidermal growth factor receptors (ERBB3 and ERBB2), myoferlin (MYOF), fibroblast growth factor 2 (FGF2), vascular endothelial growth factor D (VEGFD), angiopoietin 1 (ANGPT1) and parathyroid hormone-related protein (PTHrP), in feline primary mammary cancers (n = 6), nude mouse subcutaneous xenografts (n = 2 at 2 different passages), and mammary cancer cell lines (n = 6). *P < .05, **P < .001. Significant differences were identified for PDGFB, EGFR, VEGFR-3, and MYOF but not for PDGFA, PDGFC, ERBB3, ERBB2, FGF2, VEGFD, ANGPT1, and PTHrP. The mean is presented by the horizontal bars. Figure 18. PDGFA. Figure 19. PDGFB. Figure 20. EGFR. Figure 21. VEGFR-3. Figure 22. PDGFC. Figure 23. ERBB3. Figure 24. ERBB2. Figure 25. MYOF. Figure 26. FGF2. Figure 27. VEGFD. Figure 28. ANGPT1. Figure 29. PTHrP.
Figures 30–32.
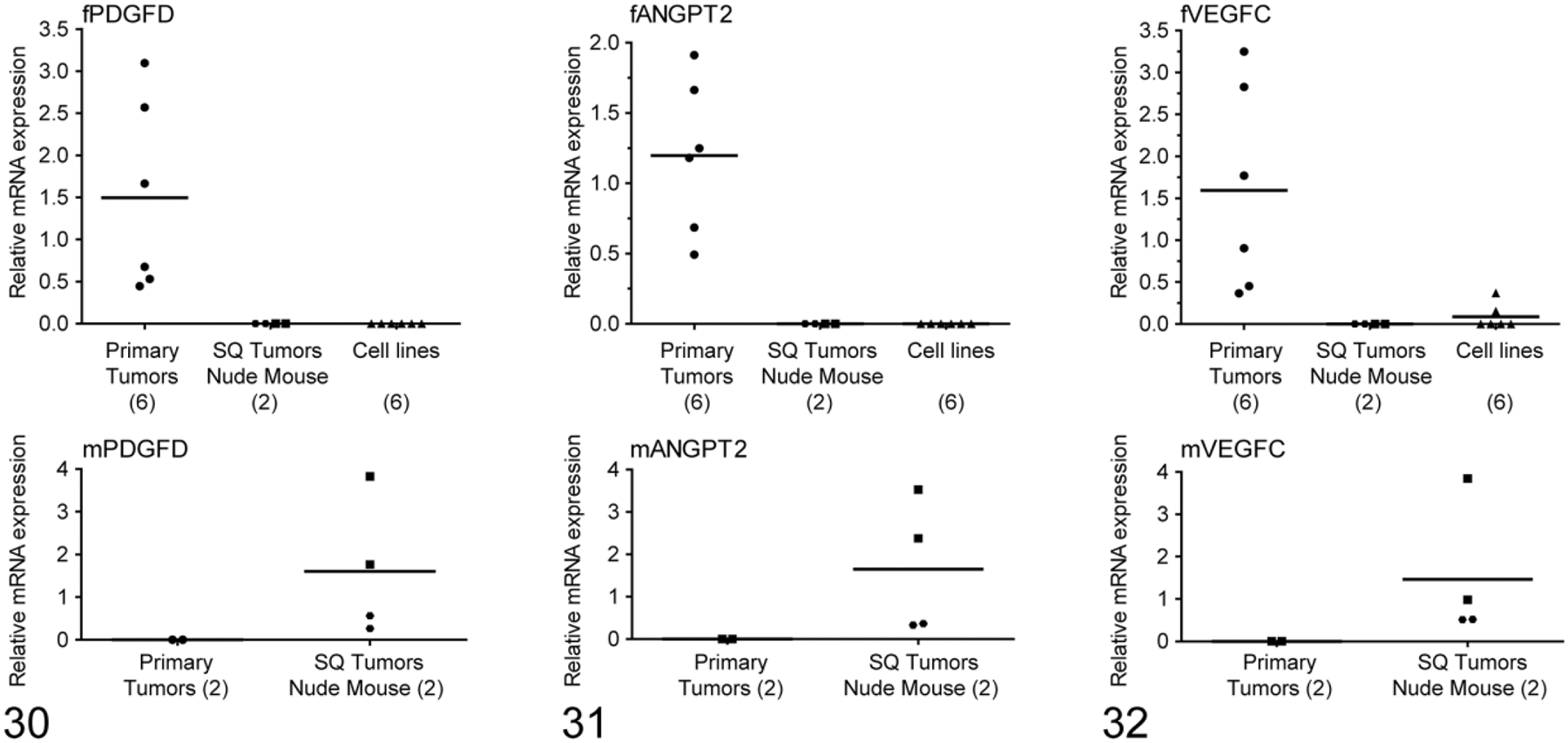
Relative mRNA expression of platelet-derived growth factor D (PDGFD), angiopoietin 2 (ANGPT2), and vascular endothelial growth factor C (VEGFC) mRNA based on qRT-PCR using cat-specific and mouse-specific primers. Upper panels: the feline mRNAs were expressed in feline primary mammary carcinomas but not in subcutaneous xenografts or cell lines. Lower panels: in contrast, murine PDGFD, ANGPT2, and VEGFC mRNAs were expressed in the subcutaneous xenografts, which suggests that these genes were expressed only in the tumor stroma and not the epithelial cells.
Discussion
Breast cancer is the most common cancer and the second leading cause of cancer-related death in women over the age of 35 years.42 Feline mammary cancer is the leading cause of cancer-related death in middle-aged to older cats.25,36,61 Women with breast cancer and metastases have decreased 5-year survival rate from 85% to 23%. There is no effective treatment for metastatic breast cancer, and relevant in vivo models are necessary to study the pathogenesis and treatment of metastasis.11,23 Human breast cancer metastasizes via the lymphatics to regional lymph nodes and also to bones, brain, adrenal gland, liver, and lung.27
Bone metastases are classified as osteolytic, osteosclerotic, or mixed lesions based on the predominant phenotype. Osteolytic metastases appear radiographically as radiolucent areas, whereas histologically, tumor cells are present in the medullary cavity and induce osteoclastic bone resorption. Osteoblastic metastases appear radiographically as radio-opaque areas, whereas histologically, tumor cells are present in the medullary cavity and induce new woven bone formation.19,45 Bone metastases in human breast cancer usually induce osteolytic bone metastases with local bone destruction due to increased osteoclast activity; however, a subset of patients develop osteosclerotic metastases.5,7,12,18,22,48,51
Animal models are essential to investigate tumor progression and metastasis in vivo.46 The ideal animal model of metastatic breast cancer would demonstrate all stages of the metastatic cascade with similar pathogenesis and patterns as in women.43 This is not possible in mice, but mouse models can be used to investigate specific stages of metastatic progression.59 We successfully developed a mouse model of feline mammary cancer by establishing 2 serial transplantable tumor lines from primary feline cancers that retained features of the original cancers including histopathology and gene expression profile. The mouse model of FMC will be useful for translational research relevant to women with metastatic breast cancer.
Eight FMC cell lines were developed from 5 of 13 cats, indicating a relatively high rate of success.55 Six of these 8 cell lines were used in this study. Although the morphological characteristics of these cell lines have been reported to be similar,55 they were distinguishable in our studies, both in tissue culture and in histopathology of xenografts. Four of 6 FMC cell lines (FMCm, FKNp, FYMp, and FNNm) grew rapidly as xenografts in nude mice after subcutaneous transplantation. FONp and FONm cells grew more slowly. In contrast to findings of a previous study, FONm cells did not grow in vivo after subcutaneous transplantation.55 No metastases were observed after 10 weeks of subcutaneous transplantation. Distant metastases were reported up to 1 year after subcutaneous transplantation of the FMC cell lines.55 However, lung, liver, kidney, eye, brain, and bone metastases were seen following intracardiac injection of FMCm Luc cells in nude mice, and pulmonary metastases occurred in mice with intratibial injection of FMCm Luc cells. It is possible that intratibial injection of FMC cells acted as an intravenous injection with subsequent spread of the cells to the lungs.
It was interesting that the bone metastases formed osteosclerotic lesions with formation of intramedullary new woven bone. Most bone metastases in women are osteolytic, but a subset of bone metastases can be osteosclerotic. Tail vein injections should also be used to characterize the metastatic ability of the FMC cell lines in the future.44 The FMCm Luc cells were considered to be the most useful cell line for future investigations.
In this study, we focused on 15 genes that are related to lymphangiogenesis, angiogenesis, tumor growth, and metastasis in feline mammary cancer. The EGFR family members are important genes that regulate several important pathways in metastasis.40,41,47 The expression of EGFR and ERBB2, a member of EGFR family, has been correlated with poor prognosis in breast cancer patients.39 The feline EGFR tyrosine kinase (TK) amino acid sequence has a high degree of homology with the human EGFR TK sequence.4 The high expression of ERBB2 and ERBB3 and high to moderate expression of EGFR in the primary cancers, xenografts, and cell lines, as well as homology between feline and human EGFR TK, support their use as a potentially important therapeutic target in FMC.
Angiogenesis and lymphangiogenesis are required for tumor progression and metastasis and are induced by stromal or tumor growth factors.17 Based on previous studies, platelet-derived growth factors (PDGFs), fibroblast growth factor (FGF), and ANGPT1 have been associated with angiogenesis,16 and VEGFC,26 VEGFD,1 and VEGFR-332 have been associated with lymphangiogenesis. We measured the expression of ANGPT1 and ANGPT2. ANGPT1 is the primary ligand for TIE2,8,53 which stabilizes nascent vasculature by recruiting mural cells. In contrast, ANGPT2 is an antagonist or partial agonist ligand31,56 that destabilizes blood vessels by interfering with ANGPT1-TIE2-induced endothelial mural cells interactions.60 ANGPT2 expression has been correlated with poor prognosis in breast cancers.49 The increased expression levels of ANGPT2 demonstrated in the primary feline cancers suggest that it may be associated with cancer progression and metastasis.
VEGFC, VEGFD, and their receptor VEGFR-3 are important regulators of tumor metastasis through the lymphatic system.32 Furthermore, VEGFC plays a role in lymphangiogenesis by activating VEGFR-3 on lymphatic endothelial cells2,24,29 and facilitates tumor spread to the regional lymph nodes in mouse models of breast cancer.28,33,52 Both VEGFD and VEGFR-3 were highly to moderately expressed in all FMC primary cancers, xenografts, and cell lines investigated in this study, which is consistent with the tendency for FMC to metastasize through the lymphatic system. VEGFR-3 may be used as a potential target for treatment of FMC.
PDGFD, ANGPT2, and VEGFC were likely produced by the tumor stromal cells and not the cancer cells. This was discovered because only the in vivo tumors produced these mRNAs and not the FMC cell lines. In addition, species-specific RT-PCR primers were necessary to amplify the mRNAs in both feline primary tumors and murine xenografts.
Cell migration is an important function for invasion and metastasis of cancer, and epithelial to mesenchymal transition (EMT) stimulates cells to migrate from primary tumors.57 A newly identified invasion gene is MYOF, which has been shown to be overexpressed in breast cancer.3,57 Its depletion is associated with mesenchymal to epithelial transition (MET) and reduced invasion through the extracellular matrix.57 We measured MYOF gene expression, and all FMC neoplasms, xenografts, and cell lines expressed higher levels of MYOF. This suggests that FMC will be a suitable model for evaluating therapeutics that target MYOF.
In conclusion, mouse models of feline mammary cancer were developed in nude mice and resulted in metastasis to bones, lung, liver, kidney, and brain, which are common sites of metastasis in human breast cancer patients. Gene expression of important invasion and metastasis genes was identified in primary cancers, xenografts, and cell lines of FMC. Feline and mouse models of feline mammary cancer will be useful to investigate the pathogenesis and treatment of metastasis and translate research findings from the laboratory to human relevance.
Supplementary Material
Acknowledgements
We thank Dr Holly Borghese and Ms Emily Kuhn for invaluable assistance in collecting FMC tissues. We thank Alan Flechtner and Anne Saulsbery for tissue processing and preparation of slides and Tim Vojt for assistance with figures.
Funding
The author(s) received the following financial support for the research, authorship, and/or publication of this article: The Ohio State College of Veterinary Medicine Biospecimen Repository was supported by the following grants: UL1TR001070 from the National Center for Advancing Translational Sciences and P30CA016058 from the National Cancer Institute to The Ohio State University. The research was supported by the Ministry of Higher Education and Scientific Research, Egypt.
Footnotes
Supplemental material for this article is available on the Veterinary Pathology website at http://journals.sagepub.com/home/vet/doi/suppl/10.1177/0300985816650243.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
References
- 1.Achen MG, Jeltsch M, Kukk E, et al. Vascular endothelial growth factor D (VEGF-D) is a ligand for the tyrosine kinases VEGF receptor 2 (Flk1) and VEGF receptor 3 (Flt4). Proc Natl Acad Sci U S A. 1998;95(2):548–553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Alitalo K, Carmeliet P. Molecular mechanisms of lymphangiogenesis in health and disease. Cancer Cell. 2002;1(3):219–227. [DOI] [PubMed] [Google Scholar]
- 3.Amatschek S, Koenig U, Auer H, et al. Tissue-wide expression profiling using cDNA subtraction and microarrays to identify tumor-specific genes. Cancer Res. 2004;64(3):844–856. [DOI] [PubMed] [Google Scholar]
- 4.Bergkvist GT, Argyle DJ, Pang LY, et al. Studies on the inhibition of feline EGFR in squamous cell carcinoma: enhancement of radiosensitivity and rescue of resistance to small molecule inhibitors. Cancer Biol Ther. 2011;11(11): 927–937. [DOI] [PubMed] [Google Scholar]
- 5.Boyde A, Maconnachie E, Reid SA, et al. Scanning electron microscopy in bone pathology: review of methods, potential and applications. Scan Electron Microsc. 1986(pt 4):1537–1554. [PubMed] [Google Scholar]
- 6.Cao Y. Opinion: emerging mechanisms of tumour lymphangiogenesis and lymphatic metastasis. Nat Rev Cancer. 2005;5(9):735–743. [DOI] [PubMed] [Google Scholar]
- 7.Coleman RE, Rubens RD. The clinical course of bone metastases from breast cancer. Br J Cancer. 1987;55(1):61–66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Davis S, Aldrich TH, Jones PF, et al. Isolation of angiopoietin-1, a ligand for the TIE2 receptor, by secretion-trap expression cloning. Cell. 1996;87(7): 1161–1169. [DOI] [PubMed] [Google Scholar]
- 9.De Maria R, Maggiora P, Biolatti B, et al. Feline STK gene expression in mammary carcinomas. Oncogene. 2002;21(11):1785–1790. [DOI] [PubMed] [Google Scholar]
- 10.De Maria R, Olivero M, Iussich S, et al. Spontaneous feline mammary carcinoma is a model of HER2 overexpressing poor prognosis human breast cancer. Cancer Res. 2005;65(3):907–912. [PubMed] [Google Scholar]
- 11.DeRose YS, Wang G, Lin YC, et al. Tumor grafts derived from women with breast cancer authentically reflect tumor pathology, growth, metastasis and disease outcomes. Nat Med. 2011;17(11):1514–1520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Diel IJ, Solomayer EF, Costa SD, et al. Reduction in new metastases in breast cancer with adjuvant clodronate treatment. N Engl J Med. 1998;339(6):357–363. [DOI] [PubMed] [Google Scholar]
- 13.Egenvall A, Bonnett BN, Häggström J, et al. Morbidity of insured Swedish cats during 1999–2006 by age, breed, sex, and diagnosis. J Feline Med Surg. 2010; 12(12):948–959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Egenvall A, Nødtvedt A, Häggström J, et al. Mortality of life-insured Swedish cats during 1999–2006: age, breed, sex, and diagnosis. J Vet Intern Med. 2009; 23(6):1175–1183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Elston CW, Ellis IO. Pathological prognostic factors in breast cancer, I: the value of histological grade in breast cancer: experience from a large study with long-term follow-up. Histopathology. 1991;19(5):403–410. [DOI] [PubMed] [Google Scholar]
- 16.Ferrara N, Kerbel RS. Angiogenesis as a therapeutic target. Nature. 2005; 438(7070):967–974. [DOI] [PubMed] [Google Scholar]
- 17.Folkman J. Tumor angiogenesis: therapeutic implications. N Engl J Med. 1971; 285(21):1182–1186. [DOI] [PubMed] [Google Scholar]
- 18.Goldstein RH, Weinberg RA, Rosenblatt M. Of mice and (wo)men: mouse models of breast cancer metastasis to bone. J Bone Miner Res. 2010;25(3): 431–436. [DOI] [PubMed] [Google Scholar]
- 19.Guise TA, Mohammad KS, Clines G, et al. Basic mechanisms responsible for osteolytic and osteoblastic bone metastases. Clin Cancer Res. 2006;12(20 pt 2): 6213s–6216s. [DOI] [PubMed] [Google Scholar]
- 20.Hahn KA, Bravo L, Avenell JS. Feline breast carcinoma as a pathologic and therapeutic model for human breast cancer. In Vivo. 1994;8(5):825–828. [PubMed] [Google Scholar]
- 21.Hayes HM, Milne KL, Mandell CP. Epidemiological features of feline mammary carcinoma. Vet Rec. 1981;108(22):476–479. [DOI] [PubMed] [Google Scholar]
- 22.Hortobagyi GN, Theriault RL, Porter L, et al. Efficacy of pamidronate in reducing skeletal complications in patients with breast cancer and lytic bone metastases. Protocol 19 Aredia Breast Cancer Study Group. N Engl J Med. 1996; 335(24):1785–1791. [DOI] [PubMed] [Google Scholar]
- 23.Iorns E, Drews-Elger K, Ward TM, et al. A new mouse model for the study of human breast cancer metastasis. PLoS One. 2012;7(10):e47995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Jeltsch M, Kaipainen A, Joukov V, et al. Hyperplasia of lymphatic vessels in VEGF-C transgenic mice. Science. 1997;276(5317):1423–1425. [DOI] [PubMed] [Google Scholar]
- 25.Jemal A, Murray T, Samuels A, et al. Cancer statistics, 2003. CA Cancer J Clin. 2003;53(1):5–26. [DOI] [PubMed] [Google Scholar]
- 26.Joukov V, Pajusola K, Kaipainen A, et al. A novel vascular endothelial growth factor, VEGF-C, is a ligand for the Flt4 (VEGFR-3) and KDR (VEGFR-2) receptor tyrosine kinases. EMBO J. 1996;15(7):1751. [PMC free article] [PubMed] [Google Scholar]
- 27.Kim JB, O’Hare MJ, Stein R. Models of breast cancer: is merging human and animal models the future? Breast Cancer Res. 2004;6(1):22–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Krishnan J, Kirkin V, Steffen A, et al. Differential in vivo and in vitro expression of vascular endothelial growth factor (VEGF)-C and VEGF-D in tumors and its relationship to lymphatic metastasis in immunocompetent rats. Cancer Res. 2003;63(3):713–722. [PubMed] [Google Scholar]
- 29.Lee J, Gray A, Yuan J, et al. Vascular endothelial growth factor-related protein: a ligand and specific activator of the tyrosine kinase receptor Flt4. Proc Natl Acad Sci U S A. 1996;93(5):1988–1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.MacEwen EG. Spontaneous tumors in dogs and cats: models for the study of cancer biology and treatment. Cancer Metastasis Rev. 1990;9(2):125–136. [DOI] [PubMed] [Google Scholar]
- 31.Maisonpierre PC, Suri C, Jones PF, et al. Angiopoietin-2, a natural antagonist for Tie2 that disrupts in vivo angiogenesis. Science. 1997;277(5322):55–60. [DOI] [PubMed] [Google Scholar]
- 32.Matsumoto M, Roufail S, Inder R, et al. Signaling for lymphangiogenesis via VEGFR-3 is required for the early events of metastasis. Clin Exp Metastasis. 2013;30(6):819–832. [DOI] [PubMed] [Google Scholar]
- 33.Mattila MM, Ruohola JK, Karpanen T, et al. VEGF-C induced lymphangiogenesis is associated with lymph node metastasis in orthotopic MCF-7 tumors. Int J Cancer. 2002;98(6):946–951. [DOI] [PubMed] [Google Scholar]
- 34.Mauldin GN, Matus RE, Patnaik AK, et al. Efficacy and toxicity of doxorubicin and cyclophosphamide used in the treatment of selected malignant tumors in 23 cats. J Vet Intern Med. 1988;2(2):60–65. [DOI] [PubMed] [Google Scholar]
- 35.Mills SW, Musil KM, Davies JL, et al. Prognostic value of histologic grading for feline mammary carcinoma: a retrospective survival analysis. Vet Pathol. 2015;52(2):238–249. [DOI] [PubMed] [Google Scholar]
- 36.Misdorp W. Tumors of the mammary gland In: Meuten DJ, ed. Tumors in Domestic Animals. Ames, Iowa: Iowa State Press; 2008:575–606. [Google Scholar]
- 37.Misdorp W, Else RW, HellménLipscomb TP. Histological Classification of Mammary Tumors of the Dog and the Cat. 2nd series Vol 38 Washington, DC: Armed Forces Institute of Pathology in cooperation with the American Registry of Pathology and the World Health Organization Collaborating Center for Worldwide Reference on Comparative Oncology; 1999. [Google Scholar]
- 38.Moulton JE. Tumors in Domestic Animals. Berkeley, CA: University of California Press; 1990. [Google Scholar]
- 39.Newby JC, Johnston SR, Smith IE, et al. Expression of epidermal growth factor receptor and c-ERBB2 during the development of tamoxifen resistance in human breast cancer. Clin Cancer Res. 1997;3(9):1643–1651. [PubMed] [Google Scholar]
- 40.Normanno N, Bianco C, De Luca A, et al. Target-based agents against ErbB receptors and their ligands: a novel approach to cancer treatment. Endocr Relat Cancer. 2003;10(1):1–21. [DOI] [PubMed] [Google Scholar]
- 41.Normanno N, Bianco C, Strizzi L, et al. The ErbB receptors and their ligands in cancer: an overview. Curr Drug Targets. 2005;6(3):243–257. [DOI] [PubMed] [Google Scholar]
- 42.Peto R, Boreham J, Clarke M, et al. UK and USA breast cancer deaths down 25% in year 2000 at ages 20–69 years. Lancet. 2000;355(9217):1822. [DOI] [PubMed] [Google Scholar]
- 43.Poste G, Fidler IJ. The pathogenesis of cancer metastasis. Nature. 1980; 283(5743):139–146. [DOI] [PubMed] [Google Scholar]
- 44.Rashid OM, Nagahashi M, Ramachandran S, et al. Is tail vein injection a relevant breast cancer lung metastasis model? J Thorac Dis. 2013;5(4):385–392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Roodman GD. Mechanisms of bone metastasis. N Engl J Med. 2004;350(16): 1655–1664. [DOI] [PubMed] [Google Scholar]
- 46.Rosol TJ, Tannehill-Gregg SH, LeRoy BE, et al. Animal models of bone metastasis. Cancer. 2003;97(3 suppl):748–757. [DOI] [PubMed] [Google Scholar]
- 47.Salomon DS, Brandt R, Ciardiello F, et al. Epidermal growth factor-related peptides and their receptors in human malignancies. Crit Rev Oncol Hematol. 1995;19(3):183–232. [DOI] [PubMed] [Google Scholar]
- 48.Sasaki A, Boyce BF, Story B, et al. Bisphosphonate risedronate reduces metastatic human breast cancer burden in bone in nude mice. Cancer Res. 1995; 55(16):3551–3557. [PubMed] [Google Scholar]
- 49.Sfiligoi C, de Luca A, Cascone I, et al. Angiopoietin-2 expression in breast cancer correlates with lymph node invasion and short survival. Int J Cancer. 2003;103(4):466–474. [DOI] [PubMed] [Google Scholar]
- 50.Siegel R, Ward E, Brawley O, et al. Cancer statistics, 2011: the impact of eliminating socioeconomic and racial disparities on premature cancer deaths. CA Cancer J Clin. 2011;61(4):212–236. [DOI] [PubMed] [Google Scholar]
- 51.Simmons JK, Hildreth BE, Supsavhad W, et al. Animal models of bone metastasis. Vet Pathol. 2015;52(5):827–841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Skobe M, Hawighorst T, Jackson DG, et al. Induction of tumor lymphangiogenesis by VEGF-C promotes breast cancer metastasis. Nat Med. 2001;7(2): 192–198. [DOI] [PubMed] [Google Scholar]
- 53.Suri C, Jones PF, Patan S, et al. Requisite role of angiopoietin-1, a ligand for the TIE2 receptor, during embryonic angiogenesis. Cell. 1996;87(7): 1171–1180. [DOI] [PubMed] [Google Scholar]
- 54.Tannehill-Gregg SH, Levine AL, Nadella MV, et al. The effect of zoledronic acid and osteoprotegerin on growth of human lung cancer in the tibias of nude mice. Clin Exp Metastasis. 2006;23(1):19–31. [DOI] [PubMed] [Google Scholar]
- 55.Uyama R, Hong SH, Nakagawa T, et al. Establishment and characterization of eight feline mammary adenocarcinoma cell lines. J Vet Med Sci. 2005;67(12): 1273–1276. [DOI] [PubMed] [Google Scholar]
- 56.Valenzuela DM, Griffiths JA, Rojas J, et al. Angiopoietins 3 and 4: diverging gene counterparts in mice and humans. Proc Natl Acad Sci U S A. 1999;96(5): 1904–1909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Volakis LI, Li R, Ackerman WE, et al. Loss of myoferlin redirects breast cancer cell motility towards collective migration. PLoS One. 2014;9(2):e86110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Weijer K, Head KW, Misdorp W, et al. Feline malignant mammary tumors, I: morphology and biology: some comparisons with human and canine mammary carcinomas. J Natl Cancer Inst. 1972;49(6):1697–1704. [DOI] [PubMed] [Google Scholar]
- 59.Werbeck JL, Thudi NK, Martin CK, et al. Tumor microenvironment regulates metastasis and metastasis genes of mouse MMTV-PymT mammary cancer cells in vivo. Vet Pathol. 2014;51(4):868–881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Yancopoulos GD, Davis S, Gale NW, et al. Vascular-specific growth factors and blood vessel formation. Nature. 2000;407(6801):242–248. [DOI] [PubMed] [Google Scholar]
- 61.Zappulli V, De Zan G, Cardazzo B, et al. Feline mammary tumours in comparative oncology. J Dairy Res. 2005;72(S1):98–106. [DOI] [PubMed] [Google Scholar]
- 62.Zappulli V, Rasotto R, Caliari D, et al. Prognostic evaluation of feline mammary carcinomas: a review of the literature. Vet Pathol. 2015;52(1):46–60. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


