Abstract
Background
In non-alcoholic steatohepatitis (NASH), muscle wasting was an aggravating factor for the progression of hepatic steatosis. This study explores the potential benefits of chronic treatment with resveratrol, a strong activator of SIRT1 on the muscle wasting of NASH mice.
Methods
In vivo and in vitro study, we evaluate the SIRT1-dependent mechanisms and effects of resveratrol administration for 6 weeks with high-fat-methionine and choline deficient diet-induced NASH mice and palmitate-pretreated C2C12 myoblast cells.
Results
Resveratrol treatment improved grip strength and muscle mass of limbs, increased running distance and time on exercise wheels in NASH mice. There is a negative correlation between muscular SIRT1 activity and 3-nitrotyrosine levels of NASH and NASH-resv mice. The SIRT1-dependent effect of muscle wasting was associated with the suppression of oxidative stress, upregulation of antioxidants, inhibition of protein degradation, activation of autophagy, suppression of apoptotic activity, upregulation of lipolytic genes and the reduction of fatty infiltration in limb muscles of NASH mice. In vitro, resveratrol alleviated palmitate acid-induced oxidative stress, lipid deposition, autophagy dysfunction, apoptotic signals, and subsequently reduced fusion index and myotube formation of C2C12 cells. The beneficial effects of resveratrol were abolished by EX527.
Conclusions
Our study suggests that chronic resveratrol treatment is a potential strategy for amelioration of hepatic steatosis and muscle wasting in NASH mouse model.
Keywords: apoptosis, nonalcoholic steatohepatitis, oxidative stress, signalling
Summary box.
What is already known about this subject?
SIRT1 is a key player in hepatic carbohydrate and lipid metabolism in animal models of non-alcoholic steatohepatitis (NASH) and non-alcoholic fatty liver disease (NAFLD). We evaluate the extra benefit in skeletal muscle tissues.
What are the new findings?
SIRT1 activation can also have beneficial effects in muscle wasting and muscular fatty infiltration and autophagy dysfunction by the reduction of muscular oxidative stress in NASH mouse model.
How might it impact on clinical practice in the foreseeable future?
Activation of SIRT1 by resveratrol may both protect against liver injury and also prevents muscle wasting in NASH mouse model and it can represent a potential role as therapeutic target for NASH and NAFLD treatment.
Introduction
Muscle wasting in non-alcoholic steatohepatitis (NASH) is characterised by generalised loss of muscle strength, mass, and function. Muscle wasting is associated with progression of NASH and increased risk of advanced hepatic fibrosis independently of obesity and insulin resistance.1–3 The overlap in the pathophysiology of NASH and muscle wasting make it challenging to determine whether muscle wasting is a risk factor for NASH or whether it is a complication of NASH.4–10
Hepatic expression of SIRT1 is downregulated in NASH animals.5 In transgenic mice, activation of SIRT1 protects against high-fat (HF)-induced hepatic oxidative stress and fatty liver.11 Conversely, hepatic SIRT1 deletion significantly increases reactive oxygen species (ROS) levels in the liver as well as in multiple other tissues, consequently leading to severe hepatic oxidative stress and eventually fatty liver diseases.12
Resveratrol is a natural polyphenol with crucial antioxidative property and its beneficial effects have been linked to SIRT1 activation.13 In non-alcoholic fatty liver disease (NAFLD/NASH) status, increased oxidative stress exacerbates steatohepatitis and muscle wasting.6 In NAFLD patients, accumulated muscular oxidative stress promotes protein degradation, induces cell apoptosis, and subsequently develops muscle wasting.7 8 Resveratrol can attenuate hepatic steatosis through the inhibition of oxidative stress by activation of hepatic SIRT1.14 In addition to hepatic steatosis, the liver-specific SIRT1 knockout (KO) mice exhibited elevated oxidative stress in liver and skeletal muscle.12 In diabetic mice, chronic resveratrol treatment significantly improves muscle wasting and function by inhibition of protein degradation and apoptosis.15 In experimental models of muscle wasting, resveratrol treatment reduces oxidative stress in muscle tissues through a SIRT1-dependent manner.16
The antiapoptotic and anticatabolic effects of resveratrol on compression injury in skeletal muscle required the action of SIRT1.17 In addition, metabolic benefits of resveratrol treatment in diabetic mice have been shown in muscle wasting, which was reversed by coadministration of selective SIRT1 inhibitor EX527.18 Autophagy dysfunction plays a crucial role in the pathogenesis of muscle wasting.10 Autophagy promotes cell survival by elimination of damage organelles and is important to maintain muscle mass.9 In diabetic condition, resveratrol improves muscle cell differentiation and myotube hypertrophy by activation of SIRT1-mediated cascades.19 In obese NAFLD patients, ectopic fatty accumulation in the liver and muscle aggravates the NASH and muscle wasting progression.1 2 20 Recent studies have suggested that the muscular fatty infiltration is positively correlated with the severity of NASH.21 22 However, SIRT1-mediated effects of resveratrol on NASH-related myosteatosis have not yet been explored.
Taken together, the molecular mechanisms involved in muscle wasting of NASH is a multifactorial phenomenon that caused by increased oxidative stress, increased protein degradation, autophagy dysfunction, increased apoptosis, and fatty infiltration in muscle tissue.6 7 21 22 Through in vivo and in vitro approaches, this study aims to evaluate the SIRT1-mediated effects of resveratrol on abovementioned pathogenesis in limb muscles of NASH mice with muscle wasting.
Methods
Animals
This study was approved by the Animal Experiments Committee of Yang-Ming University and was performed according to the ‘Guide for the care and use of laboratory animals’ prepared by the National Academy of Science, USA and the ARRIVE guidelines. At the end of the experiments, the mice were euthanised with two to three times the anaesthetic dose of zoletil. All efforts were made to minimise animal numbers necessary to produce reliable results and suffering was reduced by administering anaesthetics (zoletil and xylocaine).
Eight-week-old C57BL/6 mice (Jackson Laboratories, Bar Harbor, ME) were fed with 12 weeks of normal chow (NC, Laboratory Autoclavable Rodent Diet 5010) as control (Ctrl) group (n=5) or as NASH groups (n=9) receiving 12 weeks of HF-methionine and choline deficient (MCD) diet, which composed of 37% calories (Cal) from fat (corn oil), 24.5% Cal from protein (lactalbumin hydrolysate), 38.5% Cal from carbohydrate (dextrose), and vitamins and minerals (Dyets, Bethlehem, PA) as methionine- and choline-deficient diet as recommended.23 24 The NASH-resv (n=9) group were continuously receiving 6-weeks of resveratrol (resv, 30 mg/kg/day) from the 7th to 12th weeks after HF-MCD feeding. NASH-resv+EX527 group (n=9) were NASH mice receiving concomitant treatment with resveratrol and EX527 (1 mg/kg/day) to elucidate the SIRT1-mediated effects on NASH mice.
EX527 is a specific inhibitor of SIRT1. Food and water consumption was estimated by daily observation at the time of feeding, and body weight was recorded every 2 weeks. The mouse was placed in a metabolic cage and had free access to diet and water. To determine muscle strength and coordination, we performed grip strength and rotarod tests to evaluate muscle function.
Grip strength test
Grip strength was assessed using a grip strength metre consisting of horizontal forelimb mesh and an angled hindlimb mesh (Columbus Instruments, Columbus, OH). During grip testing, mice were grasped by the proximal tail and placed on the grip device and then pulled until the grip was lost. Five successful hindlimb and forelimb strength measurements within 2 min were recorded and normalised to body weight.25 The maximum value (in grams) recorded for the respective tests was considered the muscle grip strength and was used in all analyses. All above measurements were undergone in Taiwan Mouse Clinic (national phenotyping and drug testing centre) on week 12 of the feeding regimen for Ctrl, NASH, NASH-resv, and NASH-resv+EX527 groups.
Running distance
The running distance was measured using a treadmill for mice. Briefly, the mice were acclimated to the treadmill running for 5 min at a speed of 10 m/min on a 0% grade. After acclimation, the mice were run on the treadmill with a 10% uphill grade starting at a speed of 10 m/min for 5 min. Every 2 min, the speed was increased by 2 m/min until the mice were exhausted. The running time and speeds were recorded and the running distance was calculated.
Rotarod test
For the rotarod test, the mice were acclimated to the rotarod apparatus for 2 consecutive days prior to data collection. During acclimation, the mice were placed on the rotarod twice a day for 2 min, at a speed of 5 rotations per minute. If the mice fell off the cylinder before the 2 min were up, they were placed back on the cylinder. The mice were then tested twice a day for 3 consecutive days. During each test, the cylinder was set at a speed of 10 rotations per minute for 1 minute. If the mice fell off the cylinder before the minute was up, they were placed back on the cylinder. After 1 minute, the speed accelerated at a rate of 1.2 rotations per second until it reached 40 rotations per minute. Starting at the beginning of the acceleration phase, the length of time (in seconds) that each mouse could stay in the cylinder before falling off was recorded. The maximal running time is 180 s. The final score was determined by averaging the recorded time from all six trials.26
Basal measurements
After the performance of oral glucose tolerance test (OGTT), all animals continued their initial feeding regimen until scarification. Under anaesthesia, 2 days after stabilisation and overnight fasting, heparinised-blood (from the inferior-vena-cava, abdominal-aorta, and heart-chamber) and the forelimb+hindlimb muscles were collected and weighted.
Serum and tissue SIRT1, oxidative stress, and metabolic profiles
Serum SIRT1 levels (ng/mL) were measured by ELISA kits (BD Biosciences (San Jose, CA); hepatic and muscular SIRT1 activities were measured with SIRT1 fluorometric kit (Abcam, relative fluorescence unit, RFU). In bilateral limb muscles, homogenates of mice of different groups, the protein content, and 3-nitrotyrosine (a marker of peroxynitrite) levels were measured in three independent experiments with BCA Protein Assay Kits (EMD Chemical, Darmstadt, Germany) and ELISA kits (Abcam) (Cambridge, MA, UK). Serum free fatty acid and insulin levels were measured using ELISA kits and colorimetric assay kit (Biovision, San Francisco Bay Area, CA, USA). Serum biochemistry data were measured using a standard auto sequential multiple analyzer with computer (SMAC) (Roche Diagnostics GmbH, ANNHEIM, Germany). Additionally, frozen forelimb and hindlimb muscle tissues (30 mg) were homogenised for measurement of triglyceride (TG) content by a TG Colorimetric assay kit (Cayman Chemical Company, Ann Arbor, MI, USA), mRNA and proteins.
Histologic analysis
Non-alcoholic fatty liver disease activity score was measured by H&E-stained liver section. The hepatic hydroxyproline content (μg/mg liver weight) was measured in liver tissue to assess the severity of hepatic fibrosis. In terminal deoxynucleotidyltransferase-mediated dUTP nick end labelling (TUNEL)-stained muscle section (×200), TUNEL-positive area undergoing apoptosis were calculated. At least 10 random microscopic fields per biopsy were used to determine apoptosis, which is expressed as the mean number±SD per microscopic field. In left gastrocnemius, the cross-section area of muscle fibres was evaluated by immunohistochemistry (IHC) staining with α-sarcometric actin (myocyte cytoplasm) antibodies. Additionally, O red oil and Nile red staining were performed to evaluate the severity of muscular fatty infiltration.
Direct effects of resveratrol on palmitate-pretreated C2C12 cells
Mouse C2C12 cells, a well-established model for myogenesis study, were purchased from the Bioresource Collection and Research Center (BCRC, Hsin-Chu, Taiwan). Mouse C2C12 cells were grown, and near confluent cells were induced to differentiate by switching from a confluent Dulbecco's modified eagle medium (DMEM) medium to a differentiation DMEM medium containing 2% horse serum, penicillin/streptomycin antibiotics, and 50 mM 4-(2-Hydroxyethyl)piperazine-1-ethane-sulfonic acid (HEPES) buffer, pH 7.4. Bafilomycin A1 (a blocker of autophage flux) were dissolved in dimethyl sulfoxide (DMSO) at a concentration of 100 µM. Then, palmitate (PA, 100 µM) pretreated C2C12s were incubated with vehicle, resveratrol (resv, 40 µM) or resv+EX527 (100 nM) for 2 hours prior to differentiation. A preliminary dose-finding experiment revealed that among different concentrations, maximal stimulation of SIRT1 activity in cell lysates of C2C12 cells was noted at 40 µM of resveratrol (resv).
Then, myotube formation was assessed by immunofluorescence with antimyogenic differentiation (anti-MyoD1, nuclear)/anti-myosin heavy chain (MHC) (fibre) antibody and visualised with AF488/fluorescein isothiocyanate (FITC)-conjugated secondary antibodies. A muscle cell containing three or more nuclei was considered as a myotube, as defined previously.1 2 20 Total cell nuclei and nuclei within myotubes were counted using the National Institutes of Health ImageJ software. Fusion index for day 3 myotubes was calculated as the number of MyoD1 (+) nuclei in MHC (+) myotubes (cells containing three or more nuclei) to the total number of nuclei in one field for five random microscopic fields. To analyse day 3 myotube diameter, five fields were chosen randomly, and three myotubes were measured per field along the long axis. Then, fusion indices and myotube diameters were compared among different pretreatment groups.27
Protein and mRNA levels in cultured C2C12 cells
Then, various proteins and mRNAs of oxidative stress, autophagic, apoptotic, and lipolytic markers in cell lysates were measured with antibodies purchased from R&D Systems (Minneapolis, USA), Abcam (Cambridge, MA, USA), or Santa Cruz (Biotechnology, CA, USA) and various primers (table 1). TG content in cell lysates was measured by a TG Colorimetric assay kit (Cayman Chemical Company, Ann Arbor, MI, USA).
Table 1.
Primer of mouse gene used for quantitative real-time PCR analysis
| Gene name | Primers sequences |
| SIRT1 | For:5-GCAACAGCATCTTGCCGAT-3; Rev:5-GTGCTACTGGTCTCACTT-3 |
| CuZnSOD (superoxide dismutase) | For:5-GCGGTGAACCAGTTGTGTTGTC-3; Rev:3-CCTCTGGACCCGTTACACTGAC-5 |
| MnSOD (superoxide dismutase) | For:5-ATGTTACAACTCAGGTCGCTCTTC-3; Rev:3-CCTCTCAACGACCTCCGATAGT-5 |
| Catalase | For:5′-CCGACCAGGGCATCAAAA; Rev:5′-GAGGCCATAATCCGGATCTTC-3′ |
| Glutathione peroxidase (GPx) | For:5′-ATGGGCATAAACTTGGGAGACATTTT-3′; Rev:5′-CCTAATTCTGTTGTACAAACAGGGGTATA-3′ |
| p22phox | For:5-GCGGTGTGGACAGAAGTACC-3; Rev:5-CTTGGGTTTAGGCTCAATGG-3 |
| Nox-4 | For:5-ACAGTCCTGGCTTACCTTCG-3; Rev:5-TTCTGGGATCCTCATTCTGG-3 |
| Adipose triglyceride lipase (ATGL) | For:5′-GAGCCCCGGGTGGAACAAGAT-3′; Rev:5′-AAAAGGTGGTGGGCAGGAGTAAGG-3′ |
| Hormone sensitive lipase (HSL) | For:5′-GCCGGTGACGCTGAAAGTGGT-3′; Rev:5′-CGCGCAGATGGGAGCAAGAGGT-3′ |
| Monoglyceride lipase (MGL) | For:5′-CAGAGAGGCCCACCTACTTTT-3′; Rev:5′-ATGCGCCCCAAGGTCATATTT-3′ |
| PPARα | For:5′-CCGAACATTGGTGTTCGCAG-3′; Rev:5′-AGATACGCCCAAATGCACCA-3′ |
| Myogenic differentiation (MyoD) | For:5′-CCGCCTGAGCAAAGTAAATGA-3′; Rev:5′-GCAACCGCTGGTTTGGATT-3′ |
| Muscle atrophy F-box (MAFbX) | For:5′-AGAAAAGCGGCACCTTCGT-3′; Rev:5′-CTTGGCTGCAACATCGTAGTT-3′ |
| Murf-1 | For:5′-CCTACTTGCTCCTTGTGC3′; Rev:TCCTGCTC CTGC GTGAT-3′ |
| Myosin heavy chain (MCH-II) | For:5′-AAGGTCGGCAATGAGTATGTCA-3′; Rev:5′-CAACCATCCACAGGACACTCTTC-3′ |
| LC3-II | For:5-AATCTAAGGAGTTGCCGTTATAC-3; Rev:5-CCAGTGTCTTCAATCTTGCC-3 |
| Beclin-1 | For:5-AATCTAAGGAGTTGCCGTTATAC-3; Rev:5-CCAGTGTCTTCAATCTTGCC-3 |
| Atg5 | For:5′-AGATGGACAGCTGCACACAC-3′; Rev:5′-GCTGGGGGACAATGCTAATA-3′ |
| Atg 7 | For:5′-TCCGTTGAAGTCTCTGCTT-3′; Rev:5′-CCACTGAGGTTCACCATCCT-3′ |
| Caspase-3 | For:5′-GGTATTGAGACAGACAGTGG-3′; Rev:5′-CATGGGATCTGTTTCTTTGC-3′ |
| Caspase-9 | For:5′-AGTTCCCGGGTGCTGTCTAT-3′; Rev:5′-GCCATGGTCTTTCTGCTCAC-3′ |
| 18S | For:5′-GTAACCCGTTGAACCCCATT-3′; Rev:5′-CCATCCAATCGGTAGTAGCG-3′ |
PPAR, peroxisome proliferator-activated receptor.
Statistical analysis
Results are presented as mean±SD. Data were analysed by analysis of variance and Student-Newman-Keuls tests for multiple comparisons or by Student’s t-test for unpaired data between the two groups. Statistical significance was accepted at the p<0.05 level.
Results
Chronic resveratrol treatment improves metabolic dysregulation of NASH mice
In NASH mice, hepatic steatosis, abnormal liver function test and elevated free fatty acids accompanied by hypertriglyceridemia, hyperglycaemic, hyperinsulinemia, abnormal OGTT, higher homeostasis model assessment-insulin-resistance (HOMA-IR) index and lower metabolic demand demonstrated low respiratory quotient (markers of whole body fat oxidation, average whole body CO2 production/O2 consumption) and low energy expenditure, higher whole body fat mass, more severe hepatic fibrosis, and incremental increases in body weight compared with those in body weight (figures 1A, B and 2B, H, table 2). Notably, all abnormalities were not observed in Ctrl mice. Water consumption was not significantly different among groups (online supplementary figure 1A). In comparison with NASH mice, suppression of hepatic steatosis and adiposity as well as improvement of insulin sensitivity on chronic resveratrol treatment were associated with the decreased serum free fatty acid levels, increased metabolic demands, and a trend of decreased body weight in NASH-resv mice (figure 1A–B, online supplementary figure 1C-H). The effect of resveratrol on NASH mice was abolished by cotreatment with EX527 in NASH-resv+EX527 mice except body weight.
Figure 1.
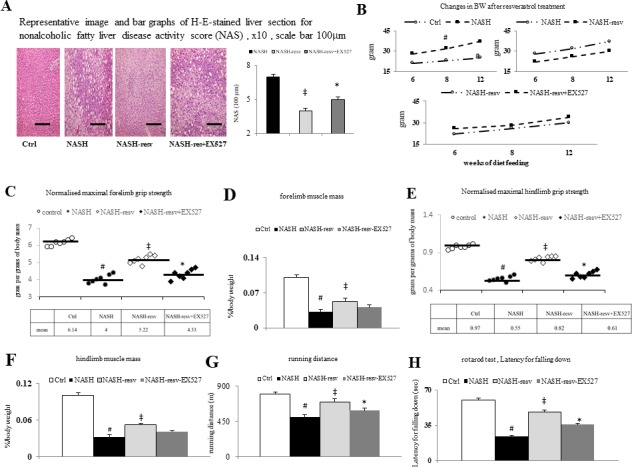
The suppression of hepatic steatosis by chronic resveratrol treatment is associated with significant improvement of muscle wasting in NASH mice: (A) representative image and bar graphs of H&E stain (×400) for non-alcoholic fatty liver disease activity score, x10, scale bar 100 µm; (B) the trend of increased body weight (growth curves); (C) normalised maximal forelimb grip strength (gram per grams of body mass); (D) forelimb muscle mass (%/body weight); (E) normalised maximal hindlimb grip strength (gram per grams of body mass); (F) hindlimb muscle mass (%/body weight); (G) running distance; (H) rota road test for latency for falling down.#p<0.05 vs Ctrl group; ‡p<0.05 vs NASH group; *p<0.05 vs NASH+resv-group. NASH, non-alcoholic steatohepatitis.
Figure 2.
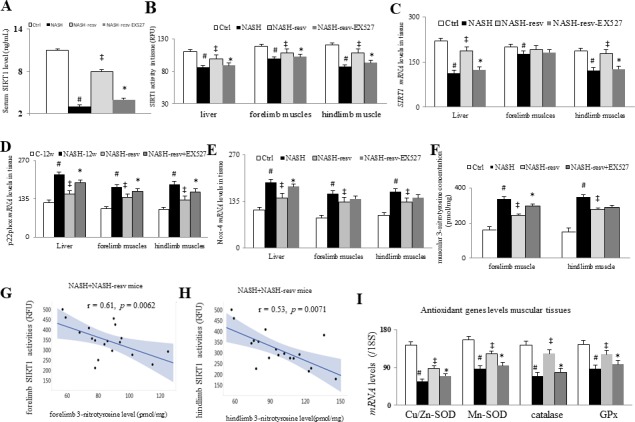
Chronic resveratrol treatment suppress oxidative stress by restoration of muscular SIRT1 levels in NASH mice: (A) serum SIRT1 levels, (B) tissue SIRT1 activity (RFU), (C) tissue SIRT1 mRNA level, (D) p22phox and (E) Nox-4 mRNA levels in tissue, (F) muscular 3-nitrotyrosine concentration (pmol/mg), (G) correlation between forelimb SIRT1 activity and 3-nitrotyrosine level of NASH+NASH resv mice, (H) correlation between hindlimb SIRT1 activity and 3-nitrotyrosine level of NASH+NASH resv mice, (I) antioxidant genes (CuZnSOD, MnSOD, catalase, and GPx) mRNA levels in muscle; #p<0.05 vs Ctrl group; ‡p<0.05 vs NASH group; *p<0.05 vs NASH+resv-group. NASH, non-alcoholic steatohepatitis.
Table 2.
Effect of chronic resveratrol (resv) treatment on the metabolic profiles of NASH mice
| Ctrl (n=5) | NASH (n=9) | NASH-resv (n=9) | NASH-resv-EX527 (n=9) | |
| (Triglyceride, TG, mg/dL) | 55±4.8 | 209±28* | 124±17† | 167±9 |
| (Fasting glucose) (mg/dL) | 99±8 | 189±21* | 118±16† | 158±13 |
| (Fasting insulin) (ng/mL) | 1.4±0.09 | 5.8±0.68* | 3.4±0.78† | 4.7±0.42 |
| HOMA-IR index | 2.4±0.1 | 18.8±2.1* | 6.9±1.3† | 12.8±0.8 |
| (Aspartate aminotransferase) (AST, U/L) | 52.3±4.9 | 145.7±5.2* | 98.5±11.4† | 114.8±10.3 |
| (Alanine aminotransferase) (ALT, U/L) | 49.1±7.1 | 120.3±20.1* | 84.4±9.6†† | 114.5±1.6 |
| Hepatic hydroxyproline content (μg/mg liver weight) | 0.19±0.02 | 0.79±0.07* | 0.35±0.02† | 0.64±0.03 |
Ctrl/NASH/NASH-resv: mice receiving 12 week of high-fat diet (HFD)+MCD diet or normal chow (NC) feeding and vehicle or resv (resveratrol, 30 mg/kg/day) treatment for 6 week from the 7th to 12th weeks of HFD+MCD feeding; NASH-resv+EX527: NASH mice are concomitant receiving resveratrol and EX-527 (SIRT1 inhibitor) treatment. HOMA-IR: homeostasis model assessment-insulin-resistance, calculated as ((fasting glucose)×(fasting insulin))/58.32.
*p<0.05 vs Ctrl group.
†p<0.05 vs NASH group.
HOMA-IR, homeostasis model assessment-insulin-resistance; MCD, methionine and choline deficient; NASH, non-alcoholic steatohepatitis.
bmjgast-2020-000381supp001.pdf (244.4KB, pdf)
SIRT1-dependent improvement in metabolic profiles by chronic resveratrol treatment associated with the prevention of muscle wasting in NASH mice
In comparison with control mice, the abovementioned metabolic abnormalities were associated with significant muscle wasting, which is represented as decreased forelimb and hindlimb grip strength, muscle mass, wheel running distance and time in NASH mice (figure 1C–H, table 3). Like metabolic changes, activation of SIRT1 by chronic resveratrol treatment significantly prevents muscle wasting by increasing muscle strength and function in NASH-resv mice (figure 1C–H, table 3). Notably, concomitant administration of a specific SIRT1 inhibitor, EX527, abolished abovementioned resveratrol-related benefits in NASH-resv+EX527 mice.
Table 3.
Muscle wasting parameters
| Ctrl (n=5) | NASH (n=9) | NASH-resv (n=9) | NASH-resv+EX527 (n=9) | |
| Body weight (g) | 24.6±49 | 39.8±3.1* | 32.4±2.4 | 36.1±2.7 |
| Whole forelimb muscle weight (μg) | 173.7±8.9 | 89.1±2.9* | 117.2±4.8† | 104.2±3.1 |
| Whole hindlimb muscle weight (μg) | 246.8±3.2 | 127.7±6.3** | 167.5±3.1† | 148.3±2.6 |
Forelimb muscles are including bilateral quadriceps, deltoid, triceps, and biceps; hindlimb muscles are including bilateral T, gastrocnemius, solues, and plantaris.
†p<0.05 vs NASH-group.
*, **p<0.05, 0.01 vs Ctrl-group.
NASH, non-alcoholic steatohepatitis.
Restoration of muscular SIRT1 by resveratrol is associated with the suppression of muscular oxidative stress and protein degradation in NASH mice
In comparison with Ctrl mice, we observed significantly lower levels of serum, hepatic, forelimb and hindlimb SIRT1 activities and mRNA in NASH mice (figure 2A–C). In NASH mice, lower systemic and muscular SIRT1 levels were associated with increased muscular oxidative stress (increased 3-nitrotyrosine concentration, increased oxidative stress (subunit of NADH oxidase, p22phox, and Nox-4), decreased antioxidants (CuZnSOD, MnSOD, catalase, and GPx)], and decreased muscular protein contents (figures 2D–I and 3A–C). There was a significantly inverse correlation between muscular SIRT1 activity and 3-nitrotyrosine concentration in the forelimb and hindlimb muscle tissues (figure 2G, H). Remarkably, activation of SIRT1 activity by resveratrol and suppression of oxidative stress were associated with normalised muscular protein content in NASH mice (figure 3C). Particularly, concomitant administration of a specific SIRT1 inhibitor, EX527, abolished abovementioned resveratrol-related benefits in the aspects of muscular oxidative stress and protein degradation in NASH-resv+EX527 mice.
Figure 3.
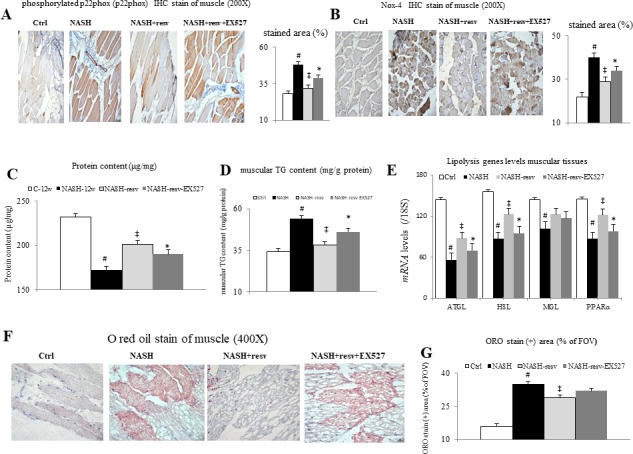
Chronic effects of resveratrol treatment on the muscular oxidative stress and fatty infiltration in mice with NASH-associated muscle wasting. Representative images (200×) and bar graphs for muscular IHC staining of (A) p22phox and (B) Nox-4, (C) muscular protein content (μg/mg), (D) muscular TG content (mg/g protein), (E) lipolytic genes levels in muscle tissue, representative images (400×) (F) and bar graphs (G) for muscle O red oil (ORO) stained positive area (% ofFOV) in muscle; #p<0.05 vs Ctrl group; ‡p<0.05 vs NASH group; *p<0.05 vs NASH+resv-group. FOV, field of view; NASH, non-alcoholic steatohepatitis. TG, triglyceride.
SIRT1-dependent effects of resveratrol on the muscular fatty infiltration in NASH mice with muscle wasting
In comparison with control mice, the NASH mice were characterised by decreased muscular SIRT1 levels, reduced muscular antioxidants, increased muscular oxidative stress, increased muscular fatty infiltration, and downregulation of muscular lipolytic gene expressions (figures 3 and 4). Significantly, chronic resveratrol treatment upregulated muscular lipolytic (adipose triglyceride lipase (ATGL), hormone sensitive lipase (HSL), and peroxisome proliferator-activated receptor (PPARα)) genes and inhibited muscular fatty infiltration, which was measured by oil red O staining and direct measurement of muscular TG content in NASH-resv mice (figure 3D–G). Nonetheless, the muscular expressions of MGL genes were not modified by chronic treatment in NASH-resv mice. Notably, concomitant administration of a specific SIRT1 inhibitor, EX527, abolished abovementioned resveratrol-related benefits in the aspects of muscular fatty infiltration in NASH-resv+EX527 mice.
Figure 4.
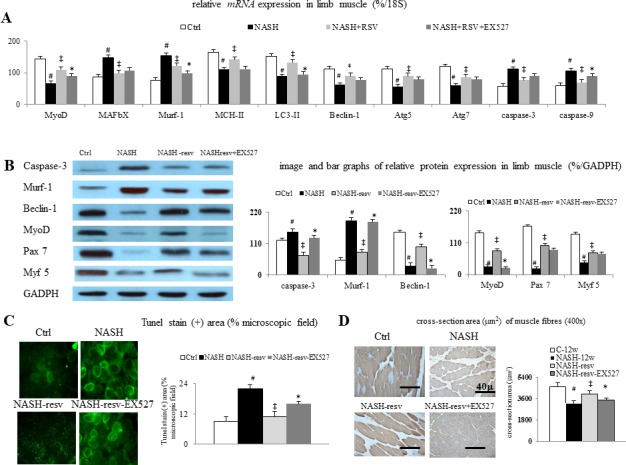
Chronic resveratrol treatment suppresses protein degradation, apoptosis, and autophagy dysfunction in mice with NASH-associated muscle wasting: (A) bar graphs of muscular mRNAs levels and (B) representative images/bar graphs of muscular protein levels (%/GADPH) of myogenic and myotropic, autophagic, and apoptotic markers among groups; (C) representative images (200X)/bar graphs of tunel stain (+) area (% microscopic field) of muscular tunel stain among groups, (D) representative images (400X)/bar graphs of muscular cross-section area among groups. #p<0.05 vs Ctrl group; ‡p<0.05 vs NASH group;*p<0.05 vs NASH+resv-group. GADPH, Glyceraldehyde 3-phosphate dehydrogenase; NASH, non-alcoholic steatohepatitis
Activation of SIRT1 by resveratrol decreases muscular autophagy and apoptosis in NASH mice
In comparison with control mice, the downregulation of expressions of muscular myogenic (MyoD, Pax7, and Myf5) markers and the downregulation of expressions of myotropic (MAFbx and Murf-1) markers were associated with muscular autophagy dysfunction (LC3-II and Beclin-2 expressions), increased muscular apoptosis (increased tunel stained positive area, caspase-3 and caspase-9) and decreased cross-section area of muscle in limb muscles of NASH mice (figure 4).
Significantly, restoration of muscular SIRT1 expression by chronic resveratrol treatment is associated with the correction of autophagy dysfunction, apoptosis by suppression of corresponding pathogenic markers in NASH mice with muscle wasting (figure 4). Notably, the SIRT1-dependent beneficial effects of chronic resveratrol treatment in muscle tissue of NASH-resv mice were reversed by concomitant administration of specific SIRT1 blocker EX527 in NASH-resv+EX527 mice.
In vitro SIRT1-dependent effects of resveratrol in PA-treated C2C12 cells
As presented in figure 5A and B, PA downregulates antioxidative stress (CuZnSOD, MnSOD, catalase, and GPx) genes, upregulates oxidative stress (p22phox and Nox4) genes, downregulates lipolytic (ATGL, HSL, and PPARα) genes, and subsequently increased TG content in cell lysates of plamitate-pretreated C2C12 cells. Meanwhile, plamitate downregulate autophagic (Beclin-1 and LC3-II) genes, upregulate apoptotic (caspase-3 and casapse-9) genes, and increase cell apoptosis (percentage of Annexin V(+)/PI(−) cell measured by flow cytometry) of C2C12 cells (figure 5D and E).
Figure 5.
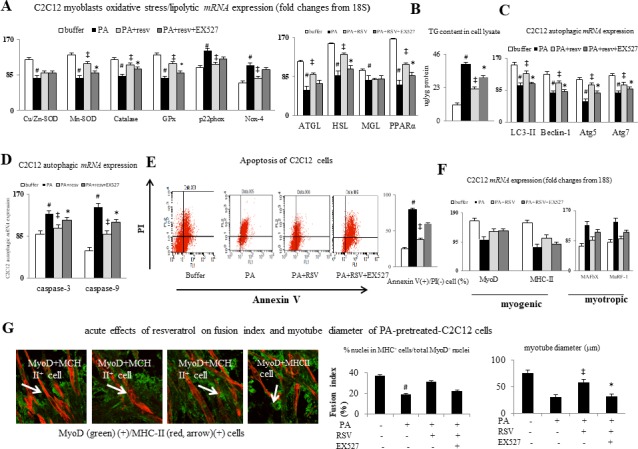
In vivo effects of resveratrol treatment on myogenic profiles. (A and C) mRNA expressions of oxidative stress, antioxidative stress, autophagic, apoptotic, and lipolytic markers, (B) TG content in cell lysates, (D) flow assessed effects of resveratrol on the palmitic acid (PA)-induced apoptosis of C2C12 cell, (E) representative immunoflourensence (IF) image and bar graphs of acute effects of resveratrol on fusion index and myotube diameter of C2C12 cells, relative fusion index, and myotube diameter. PA: palmitic acid (100 uM), RSV (resveratrol, 40 uM), EX527 (100 nM), #p<0.05 vs buffer group; ‡p<0.05 vs PA group; *p<0.05 vs PA+resv-group. ATGL, adipose triglyceride lipase; HSL, hormone sensitive lipase; MGL, monoglyceride lipase; PA, palmitic acid; TG, triglyceride;
Particularly, abovementioned changes in gene expression, increased lipid deposition, and apoptosis were accompanied by suppression of myogenic (MyoD) genes and stimulation of myotropic (MAFbX and MuRF-1) genes in cell lysates of PA-pretreated C2C12 cells as well as inhibition of fusion and tube formation of C2C12 myoblasts, which were all significantly improved by concomitant acute resveratrol incubation (figure 5F, G). Notably, the resveratrol-related effects were blocked by EX527 preincubation.
Discussion
Resveratrol, a well-known antioxidant, is a potent activator of SIRT1.13 14 16 In muscle tissue of obese rats with muscle wasting, increased oxidative stress is parallel to the increased protein degradation and apoptosis.28 In diabetes, the regulation loop between oxidative stress and myotropic gene (MuRF-1) cascades contributes to skeletal muscle atrophy.29 Both in vivo and in vitro experiments in our study revealed that the resveratrol treatment may upregulate the expression of SIRT-1 expression and inhibit oxidative stress leading to downregulation of MuRF-1 expression and improving in muscle strength and function of limb of NASH mice. Resveratrol improves obesity-related muscle wasting and reduces hepatic steatosis through inhibition of oxidative stress.30 31 Our study provided the possible mechanisms about the dual organs’ beneficial effects of resveratrol in NASH mice with attenuating hepatic steatosis and improving muscle wasting by SIRT1-dependent mechanism.
Autophagy can reduce sources of stress and injury-related cell apoptosis. Many proteins activate autophagy by binding to Beclin-1. Both LC3-II and Beclin-1 are autophagic markers. Beclin-1 links apoptosis to autophagy. Interaction between Beclin-1 and caspases maintains the homeostatic between autophagy and apoptosis. The presence of caspases inhibits the activity of Beclin-1. In our study, the increased caspase-3 and casapase-9 expression was associated with downregulation of the Beclin-1 in the muscle of NASH mice and PA-pretreated C2C12 cells.
It had been reported that muscular protein synthesis (myogenesis) is suppressed in NAFLD and obese patients.1 2 20 21 In line with previous studies,15–19 our current study revealed that chronic resveratrol normalised the expression of muscular MyoD and MHC-II in NASH mice. The skeletal satellite cells (myoblasts) can self-renew and differentiation. Pax7 and Myf5 are markers for the differentiation and proliferation of myoblasts, and muscle protein synthesis.32 Resveratrol can increase myoblasts survival and enhance differentiation through SIRT1 activation.19 33 In current study, the downregulation of muscular Pax7 and Myf5 in NASH mice was normalised by chronic resveratrol treatment. These results suggested that the beneficial effect of resveratrol treatment in NASH-resv mice may partially be contributed by the improvement of myoblast function.
It had been reported that the inhibition of SIRT1 was shown to exacerbate oxidative stress and suppress autophagy in stem cells.34 Autophagy dysfunction results in muscle wasting by increasing protein degradation and apoptosis.35 In diabetic condition, resveratrol activated autophagy and inhibited apoptosis.36 In consistent with previous reports,9 33 34 our results suggest that acute and chronic resveratrol treatment significantly suppresses oxidative stress, activates autophagy function, decreases protein degradation, reduces apoptosis, and ameliorates muscle wasting in NASH mice.
Upregulation of ATGL in human primary myotubes results in reduced lipid deposition, increased fatty acid release and oxidation.37 In addition to ATGL, hormone‐sensitive lipase (HSL) facilitates lipolysis in adipose tissue infiltrating skeletal muscle by elevating rate of β-oxidation.38 Both ATGL and HSL‐KO mice are characterised by increased circulating free fatty acid levels and fatty infiltration in skeletal muscle.39 40 PPARα gene expression and fatty acid oxidation are enhanced by the activation of SIRT in the liver.41 ATGL increases the activity of the nuclear receptor PPAR-α to promote fatty acid oxidation.42 In our study, increased serum free fatty acid levels and decreased expression of muscular lipolytic (HSL, ATGL, and PPARα) markers were observed in NASH mice with muscular fatty infiltration and muscle wasting.
In our study, PA pretreated C2C12 cell systems were used to test the cellular mechanism of resveratrol-related effects on muscle tissue of NASH mice. In cultured cardiomyocytes, resveratrol protects the heart from hypertrophy and metabolic dysregulation through SIRT1-dependent activation of PPARα.43 In our study, resveratrol treatment inhibits the PA-induced upregulation of lipolytic markers as well as decreases TG content in cell lysates, which were abolished by concomitant administration of EX527.
In muscle, upregulation of ATGL prevents oxidative stress-related myotropic response.44 In addition to lipid metabolism genes, PA can induce expression of apoptotic genes in C2C12 cells.45 In our current in vivo study, decreased ATGL expression was paralleled to the increased muscular oxidative stress, increased apoptotic activity, reduction of myogenic, and increased myotropic markers. Resveratrol can suppress oxidative stress-related genes in PA-treated C2C12 cells.46 Muscular fatty infiltration increases oxidative stress and accumulates lipotoxic by-products, and subsequent loss of muscle strength and reduction of muscle function.47 48
Increasing the skeletal muscle mass has been reported a therapeutic option for treating NAFLD.49 50 The current study revealed that chronic administration of resveratrol, through the SIRT1-dependent mechanisms (summarised in online supplementary figure 2), significantly improved muscle wasting and hepatic steatosis of NASH mice by suppression of parallel pathogenic changes, including increased oxidative stress, increased protein degradation, autophagy dysfunction, increased apoptosis, and fatty infiltration in muscle tissue of NASH mice.
bmjgast-2020-000381supp002.pdf (65.3KB, pdf)
There are some limitations of our study. We only evaluated the effects of resveratrol administration for 6 weeks in NASH mice after high-fat diet–MCD diet feeding. The study design was according to previous reports about muscle wasting and we supposed that muscle wasting is a chronic process that developing at the late stage of NASH fibrosis. Longer duration (6 weeks) of treatment was given to reverse this chronic process. However, the effects of 2 and 4 weeks of resveratrol treatment and washout effects after withdrawal of resveratrol for weeks are needed to evaluate in the future study. In terms of the potential therapeutic utility of resveratrol for fatty liver, the proposed therapeutic doses for humans are not fully evaluated with animal-to-human dosage conversions. Some clinical intervention trials have shown that resveratrol supplementation is safe but gastrointestinal side-effects have been observed at higher doses.51 52 In vivo studies, biological effects of resveratrol are limited by its low bioavailability and there are many human clinical trials using a wide range of dosage protocols and durations of treatment.53 More future clinical researches were needed to explore the therapeutic application of resveratrol supplementation in NASH patients.
Conclusion
Our study suggests that chronic resveratrol treatment is a feasible strategy for amelioration of hepatic steatosis, metabolic abnormalities, and muscle wasting in NASH mouse model. In addition to well-known metabolic benefits, resveratrol may be a potential therapeutic agent to simultaneously suppress hepatic steatosis and muscle wasting in NASH mice.
Acknowledgments
The authors thank the Chi-Yi Peng, Ching-Han Huang, Lin-Yu Chen, Ya-Wei Liu, and Yi-Tsau Huang are appreciated for their excellent technical assistances.
Footnotes
Contributors: C-WL, C-CH, H-CL, and C-FH drafted manuscript; H-CT and Y-LT prepared figures; M-WL, T-HL, and S-FH analysed data; C-WL, Y-YY, and C-CH interpreted results of experiments; C-YT, Y-CH, M-CH, and Y-HH approved final version of manuscript; C-WL, T-YL, S-FH, and Y-YY performed experiments; H-CL and Y-YY conceived and designed research.
Funding: This work was supported by Ministry of Science and Technology (grant no: MOST-106-2511-S-010–001 -MY3 and MOST-108-2314-B-075-050-MY3) from the National Science Council and Taipei Veterans General Hospital, Taipei, Taiwan (grant no: V106C-007).
Competing interests: None declared.
Patient consent for publication: Not required.
Ethics approval: This study was approved by the Animal Experiments Committee of Yang-Ming University and was performed according to the 'Guide for the care and use of laboratory animals' prepared by the National Academy of Science, USA and the ARRIVE guidelines.
Provenance and peer review: Not commissioned; externally peer reviewed.
Data availability statement: All data relevant to the study are included in the article or uploaded as supplementary information. All data relevant to the study are included in the article.
References
- 1.Koo BK, Kim D, Joo SK, et al. . Sarcopenia is an independent risk factor for non-alcoholic steatohepatitis and significant fibrosis. J Hepatol 2017;66:123–31. 10.1016/j.jhep.2016.08.019 [DOI] [PubMed] [Google Scholar]
- 2.Lee Y-ho, Kim SU, Song K, et al. . Sarcopenia is associated with significant liver fibrosis independently of obesity and insulin resistance in nonalcoholic fatty liver disease: nationwide surveys (KNHANES 2008-2011). Hepatology 2016;63:776–86. 10.1002/hep.28376 [DOI] [PubMed] [Google Scholar]
- 3.Bhanji RA, Narayanan P, Allen AM, et al. . Sarcopenia in hiding: the risk and consequence of underestimating muscle dysfunction in nonalcoholic steatohepatitis. Hepatology 2017;66:2055–65. 10.1002/hep.29420 [DOI] [PubMed] [Google Scholar]
- 4.Bruce KD, Szczepankiewicz D, Sihota KK, et al. . Altered cellular redox status, sirtuin abundance and clock gene expression in a mouse model of developmentally primed NASH. Biochim Biophys Acta 1861;2016:584–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wu T, Liu YH, YC F, et al. . Direct evidence of sirtuin down-regulation in the liver of non-alcoholic fatty liver disease patients. Ann Clin Lab Sci 2014;44:410–8. [PubMed] [Google Scholar]
- 6.Kalyani RR, Corriere M, Ferrucci L. Age-Related and disease-related muscle loss: the effect of diabetes, obesity, and other diseases. Lancet Diabetes Endocrinol 2014;2:819–29. 10.1016/S2213-8587(14)70034-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Khambu B, Yan S, Huda N, et al. . Autophagy in non-alcoholic fatty liver disease and alcoholic liver disease. Liver Res 2018;2:112–9. 10.1016/j.livres.2018.09.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Czaja MJ. Function of autophagy in nonalcoholic fatty liver disease. Dig Dis Sci 2016;61:1304–13. 10.1007/s10620-015-4025-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Masiero E, Agatea L, Mammucari C, et al. . Autophagy is required to maintain muscle mass. Cell Metab 2009;10:507–15. 10.1016/j.cmet.2009.10.008 [DOI] [PubMed] [Google Scholar]
- 10.McClung JM, Judge AR, Powers SK, et al. . P38 MAPK links oxidative stress to autophagy-related gene expression in cachectic muscle wasting. Am J Physiol Cell Physiol 2010;298:C542–9. 10.1152/ajpcell.00192.2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Pfluger PT, Herranz D, Velasco-Miguel S, et al. . Sirt1 protects against high-fat diet-induced metabolic damage. Proc Natl Acad Sci U S A 2008;105:9793–8. 10.1073/pnas.0802917105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wang R-H, Kim H-S, Xiao C, et al. . Hepatic SIRT1 deficiency in mice impairs mTORC2/Akt signaling and results in hyperglycemia, oxidative damage, and insulin resistance. J Clin Invest 2011;121:4477–90. 10.1172/JCI46243 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Yun J-M, Chien A, Jialal I, et al. . Resveratrol up-regulates SIRT1 and inhibits cellular oxidative stress in the diabetic milieu: mechanistic insights. J Nutr Biochem 2012;23:699–705. 10.1016/j.jnutbio.2011.03.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zhu W, Chen S, Li Z, et al. . Effects and mechanisms of resveratrol on the amelioration of oxidative stress and hepatic steatosis in KKAy mice. Nutr Metab 2014;11:35 10.1186/1743-7075-11-35 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wang D, Sun H, Song G, et al. . Resveratrol improves muscle atrophy by modulating mitochondrial quality control in STZ-induced diabetic mice. Mol Nutr Food Res 2018;62:e1700941. 10.1002/mnfr.201700941 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hori YS, Kuno A, Hosoda R, et al. . Resveratrol Ameliorates Muscular Pathology in the Dystrophic mdx Mouse, a Model for Duchenne Muscular Dystrophy. J Pharmacol Exp Ther 2011;338:784–94. 10.1124/jpet.111.183210 [DOI] [PubMed] [Google Scholar]
- 17.Sin TK, Yung BY, Yip SP, et al. . Sirt1-Dependent myoprotective effects of resveratrol on muscle injury induced by compression. Front Physiol 2015;6:293 10.3389/fphys.2015.00293 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Jeong J. Sirt1-Independent rescue of muscle regeneration by resveratrol in type I diabetes. J Diabetes Metab 2013;4:1000289. [Google Scholar]
- 19.Dugdale HF, Hughes DC, Allan R, et al. . The role of resveratrol on skeletal muscle cell differentiation and myotube hypertrophy during glucose restriction. Mol Cell Biochem 2018;444:109–23. 10.1007/s11010-017-3236-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kitajima Y, Eguchi Y, Ishibashi E, et al. . Age-Related fat deposition in multifidus muscle could be a marker for nonalcoholic fatty liver disease. J Gastroenterol 2010;45:218–24. 10.1007/s00535-009-0147-2 [DOI] [PubMed] [Google Scholar]
- 21.Kitajima Y, Hyogo H, Sumida Y, et al. . Severity of non-alcoholic steatohepatitis is associated with substitution of adipose tissue in skeletal muscle. J Gastroenterol Hepatol 2013;28:1507–14. 10.1111/jgh.12227 [DOI] [PubMed] [Google Scholar]
- 22.Wohlgemuth SE, Seo AY, Marzetti E, et al. . Skeletal muscle autophagy and apoptosis during aging: effects of calorie restriction and life-long exercise. Exp Gerontol 2010;45:138–48. 10.1016/j.exger.2009.11.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Rinella ME, Elias MS, Smolak RR, et al. . Mechanisms of hepatic steatosis in mice fed a lipogenic methionine choline-deficient diet. J Lipid Res 2008;49:1068–76. 10.1194/jlr.M800042-JLR200 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Yang Y-Y, Tsai T-H, Huang Y-T, et al. . Hepatic endothelin-1 and endocannabinoids-dependent effects of hyperleptinemia in nonalcoholic steatohepatitis-cirrhotic rats. Hepatology 2012;55:1540–50. 10.1002/hep.25534 [DOI] [PubMed] [Google Scholar]
- 25.Spurney CF, Gordish-Dressman H, Guerron AD, et al. . Preclinical drug trials in the mdx mouse: Assessment of reliable and sensitive outcome measures. Muscle Nerve 2009;39:591–602. 10.1002/mus.21211 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Pandey SN, Cabotage J, Shi R, et al. . Conditional over-expression of PITX1 causes skeletal muscle dystrophy in mice. Biol Open 2012;1:629–39. 10.1242/bio.20121305 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Li T-H, Lee P-C, Lee K-C, et al. . Down-regulation of common NFκB-iNOS pathway by chronic Thalidomide treatment improves Hepatopulmonary Syndrome and Muscle Wasting in rats with Biliary Cirrhosis. Sci Rep 2016;6:39405. 10.1038/srep39405 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Abrigo J, Rivera JC, Aravena J, et al. . High fat diet-induced skeletal muscle wasting is decreased by mesenchymal stem cells administration: implications on oxidative stress, ubiquitin proteasome pathway activation, and myonuclear apoptosis. Oxid Med Cell Longev 2016;2016:1–13. 10.1155/2016/9047821 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Pedersen M, Bruunsgaard H, Weis N, et al. . Circulating levels of TNF-alpha and IL-6-relation to truncal fat mass and muscle mass in healthy elderly individuals and in patients with type-2 diabetes. Mech Ageing Dev 2003;124:495–502. 10.1016/S0047-6374(03)00027-7 [DOI] [PubMed] [Google Scholar]
- 30.Huang Y, Zhu X, Chen K, et al. . Resveratrol prevents sarcopenic obesity by reversing mitochondrial dysfunction and oxidative stress via the PKA/LKB1/AMPK pathway. Aging 2019;11:2217–40. 10.18632/aging.101910 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Cheng K, Song Z, Zhang H, et al. . The therapeutic effects of resveratrol on hepatic steatosis in high-fat diet-induced obese mice by improving oxidative stress, inflammation and lipid-related gene transcriptional expression. Med Mol Morphol 2019:1–11. [DOI] [PubMed] [Google Scholar]
- 32.Zammit PS. Function of the myogenic regulatory factors myf5, MyoD, myogenin and MRF4 in skeletal muscle, satellite cells and regenerative myogenesis. Semin Cell Dev Biol 2017;72:19–32. 10.1016/j.semcdb.2017.11.011 [DOI] [PubMed] [Google Scholar]
- 33.Saini A, Al-Shanti N, Sharples AP, et al. . Sirtuin 1 regulates skeletal myoblast survival and enhances differentiation in the presence of resveratrol. Exp Physiol 2012;97:400–18. 10.1113/expphysiol.2011.061028 [DOI] [PubMed] [Google Scholar]
- 34.Ou X, Lee MR, Huang X, et al. . Sirt1 positively regulates autophagy and mitochondria function in embryonic stem cells under oxidative stress. Stem Cells 2014;32:1183–94. 10.1002/stem.1641 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Sandri M. Protein breakdown in muscle wasting: role of autophagy-lysosome and ubiquitin-proteasome. Int J Biochem Cell Biol 2013;45:2121–9. 10.1016/j.biocel.2013.04.023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Xu K, Liu X-F, Ke Z-Q, et al. . Resveratrol modulates apoptosis and autophagy induced by high glucose and palmitate in cardiac cells. Cell Physiol Biochem 2018;46:2031–40. 10.1159/000489442 [DOI] [PubMed] [Google Scholar]
- 37.Badin P-M, Louche K, Mairal A, et al. . Altered skeletal muscle lipase expression and activity contribute to insulin resistance in humans. Diabetes 2011;60:1734–42. 10.2337/db10-1364 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Jocken JWE, Smit E, Goossens GH, et al. . Adipose triglyceride lipase (ATGL) expression in human skeletal muscle is type I (oxidative) fiber specific. Histochem Cell Biol 2008;129:535–8. 10.1007/s00418-008-0386-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Haemmerle G, Lass A, Zimmermann R, et al. . Defective lipolysis and altered energy metabolism in mice lacking adipose triglyceride lipase. Science 2006;312:734–7. 10.1126/science.1123965 [DOI] [PubMed] [Google Scholar]
- 40.Chen H-H, Sue Y-M, Chen C-H, et al. . Peroxisome proliferator-activated receptor alpha plays a crucial role in L-carnitine anti-apoptosis effect in renal tubular cells. Nephrol Dial Transplant 2009;24:3042–9. 10.1093/ndt/gfp258 [DOI] [PubMed] [Google Scholar]
- 41.Purushotham A, Schug TT, Xu Q, et al. . Hepatocyte-Specific deletion of SIRT1 alters fatty acid metabolism and results in hepatic steatosis and inflammation. Cell Metab 2009;9:327–38. 10.1016/j.cmet.2009.02.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Khan SA, Sathyanarayan A, Mashek MT, et al. . ATGL-catalyzed lipolysis regulates SIRT1 to control PGC-1α/PPAR-α signaling. Diabetes 2015;64:418–26. 10.2337/db14-0325 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Planavila A, Iglesias R, Giralt M, et al. . Sirt1 acts in association with PPARα to protect the heart from hypertrophy, metabolic dysregulation, and inflammation. Cardiovasc Res 2011;90:276–84. 10.1093/cvr/cvq376 [DOI] [PubMed] [Google Scholar]
- 44.Aquilano K, Baldelli S, La Barbera L, et al. . Adipose triglyceride lipase decrement affects skeletal muscle homeostasis during aging through FAs-PPARα-PGC-1α antioxidant response. Oncotarget 2016;7:23019–32. 10.18632/oncotarget.8552 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Li L-J, Ma J, Li S-B, et al. . Vascular endothelial growth factor B inhibits lipid accumulation in C2C12 myotubes incubated with fatty acids. Growth Factors 2019;37:76–84. 10.1080/08977194.2019.1626851 [DOI] [PubMed] [Google Scholar]
- 46.Sadeghi A, Seyyed Ebrahimi SS, Golestani A, et al. . Resveratrol ameliorates palmitate-induced inflammation in skeletal muscle cells by attenuating oxidative stress and JNK/NF-κB pathway in a SirT1-independent mechanism. J Cell Biochem 2017;118:2654–63. 10.1002/jcb.25868 [DOI] [PubMed] [Google Scholar]
- 47.Brioche T, Pagano AF, Py G, et al. . Muscle wasting and aging: experimental models, fatty infiltrations, and prevention. Mol Aspects Med 2016;50:56–87. 10.1016/j.mam.2016.04.006 [DOI] [PubMed] [Google Scholar]
- 48.Brøns C, Grunnet LG. Mechanisms in endocrinology: skeletal muscle lipotoxicity in insulin resistance and type 2 diabetes: a causal mechanism or an innocent bystander? Eur J Endocrinol 2017;176:R67–78. 10.1530/EJE-16-0488 [DOI] [PubMed] [Google Scholar]
- 49.Hallsworth K, Fattakhova G, Hollingsworth KG, et al. . Resistance exercise reduces liver fat and its mediators in non-alcoholic fatty liver disease independent of weight loss. Gut 2011;60:1278–83. 10.1136/gut.2011.242073 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Bacchi E, Negri C, Targher G, et al. . Both resistance training and aerobic training reduce hepatic fat content in type 2 diabetic subjects with nonalcoholic fatty liver disease (the RAED2 randomized trial). Hepatology 2013;58:1287–95. 10.1002/hep.26393 [DOI] [PubMed] [Google Scholar]
- 51.Yiu EM, Tai G, Peverill RE, et al. . An open-label trial in Friedreich ataxia suggests clinical benefit with high-dose resveratrol, without effect on frataxin levels. J Neurol 2015;262:1344–53. 10.1007/s00415-015-7719-2 [DOI] [PubMed] [Google Scholar]
- 52.la Porte C, Voduc N, Zhang G, et al. . Steady-State Pharmacokinetics and Tolerability of Trans-Resveratrol 2000 mg Twice Daily with Food, Quercetin and Alcohol (Ethanol) in Healthy Human Subjects. Clin Pharmacokinet 2010;49:449–54. 10.2165/11531820-000000000-00000 [DOI] [PubMed] [Google Scholar]
- 53.de Vries K, Strydom M, Steenkamp V. Bioavailability of resveratrol: possibilities for enhancement. J Herb Med 2018;11:71–7. 10.1016/j.hermed.2017.09.002 [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
bmjgast-2020-000381supp001.pdf (244.4KB, pdf)
bmjgast-2020-000381supp002.pdf (65.3KB, pdf)


