Abstract
Objective
Internet-based testing for Chlamydia trachomatis (CT) with self-sampling at home has gradually been implemented in Sweden since 2006 as a free-of-charge service within the public healthcare system. This study evaluated the national diagnostic outcome of this service.
Methods
Requests for data on both self-sampling at home and clinic-based sampling for CT testing were sent to the laboratories in 18 of 21 counties. Four laboratories were also asked to provide data on testing patterns at the individual level for the years 2013–2017.
Results
The proportion of self-sampling increased gradually from 2013, comprising 22.0% of all CT tests in Sweden in 2017. In an analysis of 14 counties (representing 83% of the population), self-sampling increased by 115% between 2013 and 2017 for women, compared with 71% for men, while test volumes for clinic-based sampling were fairly constant for both sexes (1.8% increase for women, 15% increase for men). In 2017 self-sampling accounted for 20.3% of all detected CT cases, and the detection rate was higher than, but similar to, clinic-based testing (5.5% vs 5.1%). The proportion of self-sampling men was also higher, but similar (33.7% vs 30.8%). Analysis of individual testing patterns in four counties over 5 years showed a higher proportion of men using self-sampling only (67%, n=10 533) compared with women (40%, n=8885).
Conclusions
Self-sampling has increased substantially in recent years, especially among women. This service is at least as beneficial as clinic-based screening for detection of CT, and self-sampling reaches men more than clinic-based testing.
Keywords: chlamydia trachomatis, testing, microbiology, diagnostics
Introduction
Chlamydia trachomatis (CT) is the most frequently detected bacterium in STIs and may cause complications such as pelvic inflammatory disease, ectopic pregnancy and infertility.1 CT infection management requires measures at several levels to achieve disease control and reduce complications resulting from lack of treatment. Testing for CT is important in breaking the epidemiological chain of infection, and early diagnosis and prompt treatment reduce the risk of CT-associated complications.2 Testing patients with symptoms is widely performed, and testing for case detection by partner notification is highly recommended.3 4 The two guidelines cited also recommend annual screening of sexually active persons aged <25 years (Centers for Disease Control and Prevention only women), since age <25 is a risk factor for STI. England has implemented CT screening as a structured programme,5 while other countries have a practice of generous testing (Sweden, Denmark, Norway, Estonia), with the aim of reducing transmission and chlamydia-associated complications.
To facilitate CT testing, self-sampling at home with analysis at a hospital laboratory was first tried in Denmark.6 It has subsequently been combined with the use of internet for ordering test kits and provision of results. Non-invasive specimens, first-catch urines for men and vaginal swabs for women, have high sensitivity and specificity with nucleic acid amplification tests, and self-sampling at home gives results as good as sampling by healthcare providers.7–9 Internet-based self-sampling is practical and may diminish barriers such as long waiting times for clinic visits, inconvenient opening hours and perceived stigma.10 11 In randomised controlled trials, the uptake of screening increased when an internet-based self-sampling service was offered,12–14 and such a service reaches persons at risk.15 However, there is considerably less experience of broad, long-term implementation of a service of this kind as part of a healthcare system. In Sweden, internet-based self-sampling tests (denoted ‘self-sampling’ in the text) have gradually been implemented since 2006 as part of routine diagnostics in all 21 counties. This service is provided through the public healthcare system, using one of two platforms in each county (www.1177.se and www.klamydia.se). Persons with detected CT are asked to visit a public healthcare clinic for free treatment and mandatory partner notification, as CT is covered by the provisions of the Swedish Communicable Diseases Act. The objective of this study was to evaluate the national diagnostic outcome of self-sampling CT tests. The specific aims were (1) to describe changes in the use of CT self-sampling and clinic-based sampling over time; (2) to compare the proportions testing positive with the two procedures with respect to demographic characteristics; and (3) to analyse testing patterns for individuals using self-sampling and/or clinic-based testing.
Methods
Collection of data
Requests for data (see online supplementary table 1) on both self-sampling at home and clinic-based sampling for CT testing during the years 2011–2017 were sent to the laboratories in 18 of 21 Swedish counties. Three counties were excluded due to small population size and recent implementation of the service.
sextrans-2019-054256supp001.pdf (352.1KB, pdf)
After preliminary analysis of the data obtained, further analysis was confined to 14 counties that could deliver data from both self-sampling and clinic-based sampling for the years 2013–2017.
From four counties (Gävleborg, Jönköping, Uppsala and Västerbotten), representing 13% of the Swedish population, extended data were requested: collection date of sample (for self-sampling approximated to the date of arrival at the laboratory), anatomical location (urine, vagina, cervix, rectum, throat, other), age, sex and personal identity number. Exclusion criteria were age <15 years, undefined or unknown sex, and unknown test result (see online supplementary figure 1). Anonymisation of data was secured before analysis by replacing the personal identity number with a unique number at the local laboratory. A second clinic-based CT test collected on the same occasion or within 4 weeks of CT detection was considered part of a single test episode and was excluded to permit comparison with self-sampled tests. This enabled an analysis to be made of individual testing patterns for the years 2013–2017.
Data on national CT testing volumes were obtained from the Public Health Agency of Sweden, to which regional laboratories report CT cases (which are covered by the Swedish Communicable Diseases Act and subject to mandatory reporting) and the number of tests performed. Population figures for counties were obtained from the government agency Statistics Sweden (http://www.scb.se/, accessed 4 May 2018).
Statistics
P values for the binomial distribution of differences in proportions were calculated by χ2 test, and CIs for difference in proportions were calculated using normal approximation. Since the data set was large, statistical hypothesis testing was not performed for all data, as it would have detected differences that were not clinically relevant. The non-parametric Spearman test was used for correlation analysis between the number of tests and the proportion of self-sampled CT tests from individuals in four counties.
Results
Self-sampling in 18 counties
In 2017, 126 544 self-sampled tests were analysed in 18 counties (representing 97% of the Swedish population). These tests comprised 22.0% of all reported CT tests in Sweden. In 7082 (5.6%) of the self-sampled tests CT was detected, ranging from 4.2% to 9.6% between counties. The CT detection rate was 7.2% in men and 4.7% in women. Men accounted for 34.0% and the 15–29 years age group for 72.5% of the self-sampled tests.
The use of self-sampling and clinic-based sampling in 14 counties in 2013–2017
Data from both self-sampling and clinic-based sampling were available from 14 counties (representing 83% of the population) for the years 2013–2017. Analysis of these data showed that the proportion of self-sampled tests increased from 11.1% in 2013 to 18.9% in 2017, compared with reported test numbers. For the 14 counties, the mean number of self-sampled tests per 10 000 inhabitants aged 15–44 was 189 in 2013 (range 95–345), compared with 348 (range 282–417) in 2017. In these counties self-sampling increased by 115% between 2013 (n=33 119) and 2017 (n=71 185) for women, compared with 71% for men (2013: n=21 172; 2017: n=36 184) (figure 1). Test volumes for clinic-based sampling were fairly constant for both sexes (1.8% increase for women; 2013: n=312 769; 2017: n=318 410; 15% increase for men; 2013: n=123 064; 2017: n=141 994). The sampling patterns in each county are presented in online supplementary table 2. An almost twofold variation between counties was seen for the total number of CT tests per 1000 inhabitants aged 15–44 years (range 528–950). Counties with low testing volumes per inhabitant also tended to have high CT detection rates (Västmanland, Jönköping, Blekinge/Kronoberg), but this was not consistent for all counties with low testing. The proportion of tested men differed more between counties for clinic-based testing (range 23%–36%) than for self-collected samples (range 31%–39%). The proportion of tests from the 15–29 years age group varied for both self-collected samples (range 67.8–82.0) and clinic-based testing (range 66.2–76.7).
Figure 1.
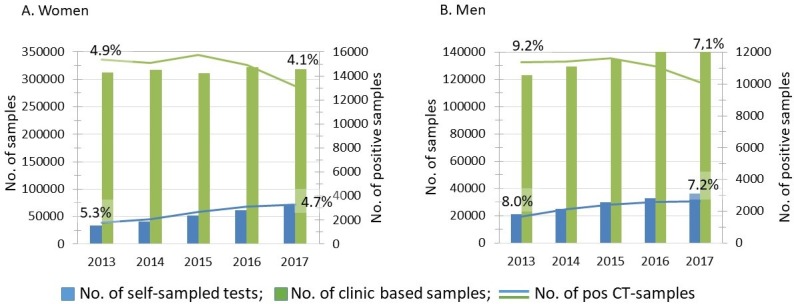
Comparison of Chlamydia trachomatis (CT) self-sampling at home and clinic-based sampling for (A) women and (B) men in 2013–2017 in 14 counties (83% of the population). Percentage figures show the CT detection rates for 2013 and 2017.
On average, self-sampling in men comprised 35.7% of all tests, compared with 27.3% for clinic-based sampling (p<0.001) for this period (online supplementary table 3). For self-sampling, the proportion of men decreased constantly, from 39.0% in 2013 to 33.7% in 2017, while for clinic-based sampling the proportions were 28.2% in 2013 and 30.8% in 2017. The 2.9% difference in the proportion of men between self-sampling and clinic-based testing in 2017 was significant (95% CI 2.6 to 3.2). In 2017 self-sampling accounted for 18.9% of all samples (table 1). In the 20–29 years age group it comprised 30.0%, while in the oldest age group (>44) self-sampling made up a smaller share of the total (13.6%). The youngest age group made sparse use of self-sampling (8.1%).
Table 1.
Analysis of self-sampling at home and clinic-based CT testing in 14 counties in 2017
| Self-sampled tests | Clinic-based tests | P value | All tests | Positive tests, n (%) | Proportion of self-sampled tests of all tests (%) | Proportion of self-sampled positive tests of all positive tests (%) | |||
| Tests, n (%) | Positive tests, n (% positive) | Tests, n (%) | Positive tests, n (%) | Tests (n) | |||||
| Gender | |||||||||
| Male | 36 184 (33.7) | 2615 (7.2) | 141 994 (30.8) | 10 104 (7.1) | 0.463 | 178 178 (31.4) | 12 719 (7.1%) | 20.3 | 20.6 |
| Female | 71 185 (66.3) | 3312 (4.7) | 318 410 (69.2) | 13 160 (4.1) | <0.001 | 389 595 (68.6) | 16 472 (4.2) | 18.3 | 20.1 |
| Total | 107 369 | 5927 (5.5) | 460 404 | 23 264 (5.1) | <0.001 | 567 773 | 29 191 (5.1) | 18.9 | 20.3 |
| Age group | |||||||||
| 15–19 | 5062 (4.7) | 513 (10.1) | 57 566 (12.5) | 4061 (7.1) | <0.001 | 62 628 (11.0) | 4574 (7.3) | 8.1 | 11.2 |
| 20–24 | 37 561 (35.0) | 2463 (6.6) | 94 527 (20.5) | 6629 (7.0) | 0.003 | 132 088 (23.3) | 9092 (6.9) | 28.4 | 27.1 |
| 25–29 | 35 031 (32.6) | 1734 (4.9) | 74 617 (16.2) | 3290 (4.4) | <0.001 | 109 648 (19.3) | 5024 (4.6) | 31.9 | 34.5 |
| 30–34 | 14 484 (13.5) | 602 (4.2) | 48 611 (10.6) | 1572 (3.2) | <0.001 | 63 095 (11.1) | 2174 (3.4) | 23.0 | 27.7 |
| 35–44 | 10 692 (10.0) | 441 (4.1) | 52 215 (11.3) | 1380 (2.6) | <0.001 | 62 907 (11.1) | 1821 (2.9) | 17.0 | 24.2 |
| >44 | 4539 (4.2) | 174 (3.8) | 28 892 (6.3) | 730 (2.5) | <0.001 | 33 431 (5.9) | 904 (2.7) | 13.6 | 19.2 |
| Total | 107 369 (100) | 5927 (5.5) | 460 404 (100) | 23 264 (5.1) | <0.001 | 567 773 (100) | 29 191 (5.1) | 18.9 | 20.3 |
CT, Chlamydia trachomatis.
CT detection rates
In 2017 self-sampling accounted for 29.7% of all detected CT cases in the 20–29 years age group (table 1), while in the oldest age group (>44) it accounted for 19.2% of CT cases. The detection rate was higher for women using self-sampling, but for men there was no difference. Furthermore, for all age groups except 20–24 years, detection rates were significantly higher for self-sampling than for clinic-based testing, and this was especially pronounced in persons older than 30 years.
The detection rate overall decreased from 6.4% to 5.5% for self-sampled tests between 2013 and 2017, while it went from 6.1% to 5.1% for clinic-based testing. The 0.4% difference between self-sampling and clinic-based testing in 2017 was significant (95% CI 0.32 to 0.62). In men, the detection rate decreased for both self-sampling and clinic-based sampling in this period (from 8.0% to 7.2% (p=0.002) vs from 9.2% to 7.1% (p<0.001); see figure 1). Corresponding figures for women decreased from 5.3% to 4.7% (p<0.001) and from 4.9% to 4.1% (p<0.001). The detection rate was on average 6.4% for the 14 counties during the study period.
Unused test kits
Between 23% and 26% of delivered test kits per year were never returned for analysis during 2013–2017. There was no difference in this respect between men and women, and no trend over time.
Extended analysis of testing patterns in individuals
Analysis of individual testing patterns was made possible by data from four counties for the period 2013–2017. Data from both self-sampling at home and clinic-based testing were gathered. The overall CT detection rate was 6.6%, compared with 6.4% for the 14 counties presented above. The sex and age distributions of tests were similar to data from the 14 counties (data not shown).
In the four counties, 11.9% used self-sampling only, while the majority (76.9%) never used self-sampling and 11.2% used both testing methods available (table 2). A higher proportion of men used self-sampling only (19.4%) compared with women (8.2%) (table 2); thus, 67.5% (n=10 533) of men and 40.3% (n=8885) of women using self-sampling had not been tested at any clinic. Individuals who were tested more than twice, in self-sampling or clinic-based testing, were also more likely to have at least one CT-positive test (table 3).
Table 2.
Test patterns in individuals tested for CT in four counties, 2013–2017
| CT-tested men, n (%) (n=54 271) |
Proportion of CT-positive men (%) | CT-tested women, n (%) (n=108 935) |
Proportion of CT-positive women (%) | CT-tested men and women, n (%) (n=163 206) |
Proportion of CT-positive men and women (%) | |
| Only used self-sampling | 10 533 (19.4) | 10.7 | 8885 (8.2) | 6.1 | 19 418 (11.9) | 8.6 |
| Never used self-sampling | 38 673 (71.3) | 13.1 | 86 913 (79.8) | 7.1 | 125 586 (76.9) | 9.0 |
| Used both self-sampling and clinic-based sampling | 5065 (9.3) | 32.8 | 13 137 (12.0) | 25.2 | 18 202 (11.2) | 27.3 |
CT, Chlamydia trachomatis.
Table 3.
Proportion of CT-positive samples correlated to test frequency in persons using self-sampling or clinic-based testing
| Number of samples | Self-sampling | Proportion of CT-positive samples | Clinic-based | Proportion of CT-positive samples | P value | Self-sampling and clinic-based | Proportion of CT-positive samples | P value | |||
| Men | Individuals | Samples | Individuals | Samples | Individuals | Samples | |||||
| 1 | 7733 | 7733 | 7.1 | 29 009 | 29 009 | 8.5 | <0.001 | ||||
| 2 | 1635 | 3270 | 8.7 | 6276 | 12 552 | 11.5 | <0.001 | 1967 | 3934 | 11.7 | <0.001 |
| >2 | 1120 | 4855 | 7.6 | 3771 | 15 183 | 11.1 | <0.001 | 3161 | 15 031 | 11.1 | <0.001 |
| Women | |||||||||||
| 1 | 6019 | 6019 | 3.5 | 55 449 | 55 449 | 2.5 | <0.001 | ||||
| 2 | 1507 | 3014 | 4.1 | 17 293 | 34 586 | 4.2 | 0.62 | 3300 | 6600 | 5.7 | 0.0011 |
| >2 | 1305 | 5953 | 4.0 | 14 666 | 62 485 | 6.8 | <0.001 | 9950 | 55 141 | 6.8 | <0.001 |
P values calculated with the proportion of CT-positive samples for self-collected samples as reference.
CT, Chlamydia trachomatis.
The number of tests performed per person increased over time for both sexes and was significantly higher for self-collected samples than for clinic-based testing (data not shown, but similar to data for 14 counties in figure 1). The proportion of self-collected samples was higher among individuals with a large number of performed tests (correlation 0.988, p<0.001; figure 2). Thus, self-collected samples comprised 18.2% of samples for persons tested twice, compared with 44.7% for those tested 10 times during the study period. Of 335 persons using only self-collected samples >5 times between 2013 and 2017, 73% were never CT-positive. The corresponding figure for persons tested only at clinics (n=1633) was 55%, and for those who used both test routes (n=2444) it was 56%.
Figure 2.
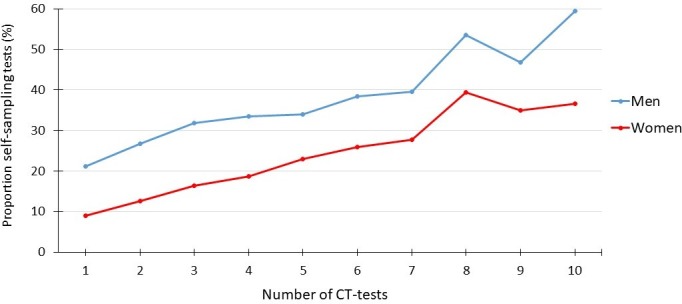
Correlation between the proportion of self-collected samples and the overall number of Chlamydia trachomatis (CT) tests from 163 206 individuals: 108 935 women and 54 271 men.
Discussion
Internet-based self-sampling at home for CT testing is now implemented in all Swedish counties as part of the public healthcare system. The study shows a substantial increase in self-sampling tests in recent years, with about a fifth of all chlamydia tests in Sweden now based on this user-friendly testing service. The detection rate of this service was slightly higher than that of clinic-based sampling, making self-sampling at least as beneficial as clinic-based testing for case detection. Women accounted for two-thirds of self-sampled tests, but the service nevertheless reached men to a higher degree than clinic-based testing, and almost 20% of men used self-sampling only. Self-sampling has thus become an important tool in CT testing.
The strength of our study is that the concept evaluated has been implemented for several years in public healthcare. The study describes what has been achieved in real life, with all its shortcomings, rather than in a temporary, controlled study of a concept. The high coverage of tests taken in the country is another strength. Analysis of the years 2013–2017 is based on data from 14 counties, representing 83% of the Swedish population, while the analysis of individual test patterns for chlamydia uses data from four counties, representing 13% of the population. The difference in chlamydia detection rate was 0.2%, and no statistical difference in sex and age group distribution was seen when data from the 14 and 4 counties were compared; that is, the analysis of 4 counties is likely to be representative of the whole country. This study is representative, although it does not include commercial CT tests available on the internet. Since CT testing is free of charge, commercial testing is marginal in Sweden, and commercial quality-assured nucleic acid amplification tests accounted for only 2.3% of all CT tests in 2017 (J Ovemyr, Dynamic Code, personal communication). CT treatment may differ between test routes, causing bias in the study results. However, the treatment coverage of detected cases is >98% for both self-sampling and clinic-based testing (data from three local communicable disease authorities).
There are also limitations. We initially asked for data for the years 2011–2017, but for some counties data were not available for all years due to technical shortcomings in laboratory information systems, and in some counties self-sampling was not established before 2013. Furthermore, analysis of test patterns for individuals could have been performed on data from more than the four selected counties. Other limitations are that this study does not include a cost analysis and does not encompass the perspective of test users. These two topics will be covered in ongoing work.
Self-sampling at home was introduced in Sweden as a study project in 2004.16 Its gradual implementation throughout the country has resulted in a mature service within the public healthcare system that has now existed for several years for the vast majority of the Swedish population. In 2017 self-collected sampling for CT testing accounted for over 20% of tests in the 15–24 years age group, which is in contrast to the reported 10% from these ages in England the same year,17 and in the highly sexually active age group 25–29 years an even higher proportion was seen. The detection rate in our study was also lower than in most randomised controlled trials,12 indicating that the testing service in Sweden is widely used. However, in almost all age groups the detection rate was significantly higher for self-sampling than for clinic-based testing. This is in contrast to the review of randomised controlled trials for CT screening where the detection rate was significantly lower for self-sampling at home compared with clinic-based specimen collection.12 In Sweden, approximately 40%–50% of all CT cases are detected by partner notification,18 and opportunistic screening based on sampling in clinics thus results in substantially lower detection rates compared with self-sampling. Our study shows that women tested only once in clinics were rarely CT-positive. In local data from Uppsala county, the detection rate for samples from gynaecology clinics (hospital-based and private) is about 2.5%. This indicates excessive testing, and changes in testing practice are likely to be beneficial. Thus, the efficiency of the widespread chlamydia testing in Sweden, with about 600 000 annual samples in a population of 10 million,19 may be questioned, and more selective testing for case detection is desirable. This also indicates that self-sampling is more efficient for CT case detection than opportunistic screening as it is performed in Sweden.
One reason for introducing this easily accessible service was to reach men to a greater extent. This goal has been achieved, but only to a limited degree, given the proportion of tests from men. However, our analysis of testing patterns at the individual level showed that a considerable proportion, 67%, of men using self-sampling were never tested in any clinic, and the detection rate in this group was almost the same as for men tested in clinics. The self-sampling at home service thus appears to reach men at risk who would not otherwise have been tested.
The study showed significantly lower use of self-sampling among teenagers compared with the 20+ age group. This is not surprising, considering that most teenagers live with parents and may feel uncomfortable about test kits arriving through the post. They also have access to ‘youth clinics’, providing testing and counselling for STIs.
Frequent use of self-sampling may be due to a need for control after a high-risk behaviour, or represent unnecessary testing explained by anxiousness. In our study it seems that both testing patterns were seen. Individuals who were tested repeatedly were also more likely to be CT-positive. But there was also a group of individuals who were tested at least five times without chlamydia being detected. Since the test is free of charge and about a quarter of requested test kits are not returned to the laboratories, overconsumption is likely. A separate study of the experience of test users is investigating this issue.
To our knowledge there is no other country than Sweden with such widely implemented internet-based CT self-sampling at home in its public health service. Regional use of self-sampling is established in England,20 the USA21 and Australia.22 In the Netherlands, such a service was provided in one region,23 but was considered too expensive and therefore discontinued. Online ordering of CT tests is also commercially available in many countries. Some of these are quality-assured nucleic acid amplification tests, while others are inferior antigen-based tests with unacceptable performance.24 The study also raises questions about chlamydia testing in general. What is sufficient testing? Can testing reach more people at risk? Do the benefits justify the costs? Testing activity also needs to be related to the risk of complications, which are not the same today as when chlamydia infections were initially described.25 26 A disadvantage with self-sampling at home is the missed opportunity to meet healthcare providers who can offer counselling on sexual health and additional medical care. Further evaluation of chlamydia testing activities in Sweden and elsewhere may be warranted to optimise the use of resources for STI prevention and treatment.
In summary, this study shows how internet-based self-sampling at home has been broadly implemented in the public healthcare system in Sweden and is at least as beneficial in detecting chlamydia infections as clinic-based opportunistic screening. This user-friendly concept is well suited to an expansion of chlamydia testing to unreached risk groups, but its use could be improved.
Key messages.
The public health service of free self-sampling at home for Chlamydia trachomatis (CT) testing covers the entire Swedish population.
Self-sampling has increased significantly in recent years, especially among women.
Self-sampling reaches men more than clinic-based testing.
Self-sampling is at least as efficient as clinic-based opportunistic screening for CT detection.
Footnotes
Handling editor: Claudia S Estcourt
Collaborators: The Chlamydia Study Group represents the other 14 participating county laboratories in Sweden: Ann-Kristine Bergström, Marianne Bäckman, Karin Elfving, Helena Enroth, Lena Ericson, Hans Fredlund, Sultan Golbob, Marit Hansen, Håkan Janson, Malin Karlsson, Eva-Kristina Kinnunen, Helena Larsson, Marita Lindqvist, Anna Lindqvist Angervall, Annika Ljung, Lena Pizum, Helena Riise, Martin Sundqvist and Magnus Unemo.
Contributors: BH designed and coordinated the study. Each participating laboratory represented in the study group contributed data. JS performed all data management and analyses. BH wrote the first draft of the manuscript, with support from JS. All authors, including the members of the study group, were involved in finalising the manuscript.
Funding: This work was supported by grants from the Public Health Agency of Sweden.
Competing interests: None declared.
Patient consent for publication: Not required.
Ethics approval: The study was approved by the Regional Ethical Review Board in Uppsala, Sweden (ref 2018/250).
Provenance and peer review: Not commissioned; externally peer reviewed.
Data availability statement: All data relevant to the study are included in the article or uploaded as supplementary information.
References
- 1. Carey AJ, Beagley KW. Chlamydia trachomatis, a hidden epidemic: effects on female reproduction and options for treatment. Am J Reprod Immunol 2010;63:576–86. 10.1111/j.1600-0897.2010.00819.x [DOI] [PubMed] [Google Scholar]
- 2. Low N, Broutet N, Adu-Sarkodie Y, et al. Global control of sexually transmitted infections. The Lancet 2006;368:2001–16. 10.1016/S0140-6736(06)69482-8 [DOI] [PubMed] [Google Scholar]
- 3. Lanjouw E, Ouburg S, de Vries HJ, et al. 2015 European guideline on the management of Chlamydia trachomatis infections. Int J STD AIDS 2016;27:333–48. 10.1177/0956462415618837 [DOI] [PubMed] [Google Scholar]
- 4. Centers for Disease Control and Prevention Sexually transmitted diseases treatment guidelines: diagnosis and management of uncomplicated Chlamydia trachomatis infections in adolescents and adults, 2015. Available: https://www.cdc.gov/std/tg2015/chlamydia.htm [DOI] [PubMed]
- 5. National Chlamydia Screening Programme (NCSP) Information, data, guidance and resources about the NCSP. Available: www.gov.uk/government/collections/national-chlamydia-screening-programme-ncsp [Accessed 7 Jun 2019].
- 6. Ostergaard L, Møller JK, Andersen B, et al. Diagnosis of urogenital Chlamydia trachomatis infection in women based on mailed samples obtained at home: multipractice comparative study. BMJ 1996;313:1186–9. 10.1136/bmj.313.7066.1186 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Hobbs MM, van der Pol B, Totten P, et al. From the NIH: proceedings of a workshop on the importance of self-obtained vaginal specimens for detection of sexually transmitted infections. Sex Transm Dis 2008;35:8–13. 10.1097/OLQ.0b013e31815d968d [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Masek BJ, Arora N, Quinn N, et al. Performance of three nucleic acid amplification tests for detection of Chlamydia trachomatis and Neisseria gonorrhoeae by use of self-collected vaginal swabs obtained via an internet-based screening program. J Clin Microbiol 2009;47:1663–7. 10.1128/JCM.02387-08 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Shih S, Graseck A, Secura G, et al. Screening for STIs at home or in the clinic? Curr Opin Infect Dis 2011;24:78–84. 10.1097/QCO.0b013e32834204a8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Lorimer K, McDaid L. Young men’s views toward the barriers and facilitators of internet-based Chlamydia trachomatis screening: qualitative study. J Med Internet Res 2013;15:e265 10.2196/jmir.2628 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Aicken CRH, Fuller SS, Sutcliffe LJ, et al. Young people’s perceptions of smartphone-enabled self-testing and online care for sexually transmitted infections: qualitative interview study. BMC Public Health 2016;16:1–11. 10.1186/s12889-016-3648-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Fajardo-Bernal L, Aponte-Gonzalez J, Vigil P, et al. Home-based versus clinic-based specimen collection in the management of Chlamydia trachomatis and Neisseria gonorrhoeae infections. Cochrane Database Syst Rev 2015;316 10.1002/14651858.CD011317.pub2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Wilson E, Free C, Morris TP, et al. Internet-accessed sexually transmitted infection (e-STI) testing and results service: a randomised, single-blind, controlled trial. PLoS Med 2017;14:e1002479–20. 10.1371/journal.pmed.1002479 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Kersaudy-Rahib D, Lydié N, Leroy C, et al. Chlamyweb Study II: a randomised controlled trial (RCT) of an online offer of home-based Chlamydia trachomatis sampling in France. Sex Transm Infect 2017;93:188–95. 10.1136/sextrans-2015-052510 [DOI] [PubMed] [Google Scholar]
- 15. Novak M, Novak D. Risk factors for Chlamydia trachomatis infection among users of an Internet-based testing service in Sweden. Sex Reprod Healthc 2013;4:23–7. 10.1016/j.srhc.2012.11.004 [DOI] [PubMed] [Google Scholar]
- 16. Novak DP, Karlsson RB. Simplifying chlamydia testing: an innovative Chlamydia trachomatis testing approach using the internet and a home sampling strategy: population based study. Sex Transm Infect 2006;82:142–7. 10.1136/sti.2005.016832 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Public Health England Sexually transmitted infections and screening for Chlamydia in England, 2017. Health Protection Report, 2018. [Google Scholar]
- 18. Löfdahl M, Rydevik G, Blaxhult A, et al. Klamydia HOS kvinnor I Sverige: smittspårning och rutiner för rapportering måste förbättras. Läkartidningen 2008;105:3116–20. [PubMed] [Google Scholar]
- 19. Public health agency Sweden. Available: https://www.folkhalsomyndigheten.se/folkhalsorapportering-statistik/statistikdatabaser-och-visualisering/sjukdomsstatistik/klamydiainfektion/ [Accessed 7 Jun 2019].
- 20. National Chlamydia Screening Programme (NCSP) GOV.UK. Available: https://www.gov.uk/government/collections/national-chlamydia-screening-programme-ncsp [Accessed 7 Jun 2019].
- 21. I want the kit. Available: https://www.iwantthekit.org [Accessed 7 Jun 2019].
- 22. Queensland Government home Sexual health Available: https://www.qld.gov.au/health/staying-healthy/sexual-health/chlamydia-test [Accessed 7 June 2019].
- 23. van Rooijen MS, Koekenbier RH, Hendriks A, et al. Young low-risk heterosexual clients prefer a Chlamydia home collection test to a sexually transmitted infection clinic visit in Amsterdam, the Netherlands, a cross-sectional study. Sex Transm Dis 2016;43:710–6. 10.1097/OLQ.0000000000000517 [DOI] [PubMed] [Google Scholar]
- 24. Horner P. Home based testing: Unintended consequences and implications for antimicrobial stewardship – should we be concerned? : Lewis D, Blanchard J, Cameron C, Abstracts for the STI & HIV World Congress (Joint Meeting of the 23rd ISSTDR and 20th IUSTI. Vancouver, Canada: Sexually Transmitted Infections, BMJ Publishing Group Ltd 2019., 2019. [Google Scholar]
- 25. Price MJ, Ades AE, Soldan K, et al. The natural history of Chlamydia trachomatis infection in women: a multi-parameter evidence synthesis. Health Technol Assess 2016;20:1–250. 10.3310/hta20220 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Bender N, Herrmann B, Andersen B, et al. Chlamydia infection, pelvic inflammatory disease, ectopic pregnancy and infertility: cross-national study. Sex Transm Infect 2011;87:601–8. 10.1136/sextrans-2011-050205 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
sextrans-2019-054256supp001.pdf (352.1KB, pdf)


