Summary
Background
The impact of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) on postoperative recovery needs to be understood to inform clinical decision making during and after the COVID-19 pandemic. This study reports 30-day mortality and pulmonary complication rates in patients with perioperative SARS-CoV-2 infection.
Methods
This international, multicentre, cohort study at 235 hospitals in 24 countries included all patients undergoing surgery who had SARS-CoV-2 infection confirmed within 7 days before or 30 days after surgery. The primary outcome measure was 30-day postoperative mortality and was assessed in all enrolled patients. The main secondary outcome measure was pulmonary complications, defined as pneumonia, acute respiratory distress syndrome, or unexpected postoperative ventilation.
Findings
This analysis includes 1128 patients who had surgery between Jan 1 and March 31, 2020, of whom 835 (74·0%) had emergency surgery and 280 (24·8%) had elective surgery. SARS-CoV-2 infection was confirmed preoperatively in 294 (26·1%) patients. 30-day mortality was 23·8% (268 of 1128). Pulmonary complications occurred in 577 (51·2%) of 1128 patients; 30-day mortality in these patients was 38·0% (219 of 577), accounting for 81·7% (219 of 268) of all deaths. In adjusted analyses, 30-day mortality was associated with male sex (odds ratio 1·75 [95% CI 1·28–2·40], p<0·0001), age 70 years or older versus younger than 70 years (2·30 [1·65–3·22], p<0·0001), American Society of Anesthesiologists grades 3–5 versus grades 1–2 (2·35 [1·57–3·53], p<0·0001), malignant versus benign or obstetric diagnosis (1·55 [1·01–2·39], p=0·046), emergency versus elective surgery (1·67 [1·06–2·63], p=0·026), and major versus minor surgery (1·52 [1·01–2·31], p=0·047).
Interpretation
Postoperative pulmonary complications occur in half of patients with perioperative SARS-CoV-2 infection and are associated with high mortality. Thresholds for surgery during the COVID-19 pandemic should be higher than during normal practice, particularly in men aged 70 years and older. Consideration should be given for postponing non-urgent procedures and promoting non-operative treatment to delay or avoid the need for surgery.
Funding
National Institute for Health Research (NIHR), Association of Coloproctology of Great Britain and Ireland, Bowel and Cancer Research, Bowel Disease Research Foundation, Association of Upper Gastrointestinal Surgeons, British Association of Surgical Oncology, British Gynaecological Cancer Society, European Society of Coloproctology, NIHR Academy, Sarcoma UK, Vascular Society for Great Britain and Ireland, and Yorkshire Cancer Research.
Introduction
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has now spread to most countries, with WHO declaring a COVID-19 pandemic on March 11, 2020.1 The pandemic has tested the resilience of health-care systems, including hospitals, which were largely unprepared for the scale of the pandemic.2 Patients having surgery are a vulnerable group at risk of SARS-CoV-2 exposure in hospital and might be particularly susceptible to subsequent pulmonary complications, due to the pro-inflammatory cytokine and immunosuppressive responses to surgery and mechanical ventilation.3, 4 Evidence of the safety of performing surgery in SARS-CoV-2-exposed hospitals is urgently needed.
Before the SARS-CoV-2 pandemic, high-quality, multinational observational studies established overall baseline rates of postoperative pulmonary complications (up to 10%) and subsequent mortality (up to 3%) after surgery.5, 6, 7 With initiatives such as the UK's National Emergency Laparotomy Audit (NELA), mortality was improving even in high-risk groups.8
Guidelines have been published for the management of surgical patients during the SARS-CoV-2 pandemic,9, 10, 11 but they are based solely on expert opinion. The impact of SARS-CoV-2 on postoperative pulmonary complications and mortality needs to be established in order to enable surgeons and patients to make evidence-based decisions during the pandemic. This study reports the clinical outcomes of patients who had surgery with perioperative SARS-CoV-2 infection, including the impact of pulmonary complications.
Research in context.
Evidence before this study
We searched PubMed and Embase on March 15, 2020, for studies reporting on surgical patients during the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) pandemic. We used the search terms “COVID-19”, “SARS-CoV-2”, “coronavirus”, and “pandemic”, in combination with “surgery”, and applied no language or date restrictions. We identified 13 articles (12 from China and one from Singapore), all of which provided clinical guidance, with none reporting patient-level outcomes.
Added value of this study
This international, observational, cohort study provides cross-specialty, patient-level outcomes data for patients who had surgery and acquired perioperative SARS-CoV-2 infection. 1128 patients were included across 24 countries. Overall 30-day mortality was 23·8% (268 of 1128 patients). Pulmonary complications occurred in 577 (51·2%) patients; these patients accounted for 82·6% (219 of 265) of all deaths. Independent risk factors for mortality were male sex, age 70 years or older, American Society of Anesthesiologists grades 3–5, surgery for malignant disease, emergency surgery, and major surgery.
Implications of all the available evidence
Postoperative pulmonary complications occur in half of patients with perioperative SARS-CoV-2 infection and are associated with high mortality. These pulmonary complication and mortality rates are greater than those reported for even the highest-risk patients before the pandemic. Thresholds for surgery during the SARS-CoV-2 pandemic should be higher than during normal practice; men aged 70 years and older who have emergency or major elective surgery are at particularly high risk of mortality. Consideration should be given for postponing non-critical procedures and promoting non-operative treatment to delay or avoid the need for surgery. When hospitals recommence routine surgery, this will be in hospital environments that remain exposed to SARS-CoV-2, so strategies should be developed to reduce in-hospital SARS-CoV-2 transmission and mitigate the risk of postoperative complications.
Methods
Study design
We did an international, multicentre, observational cohort study in patients with SARS-CoV-2 infection who had surgery at 235 hospitals in 24 countries (appendix p 10). Data release and ethical considerations were discussed with an independent data monitoring and ethics committee. We collected only routine, anonymised data with no change to clinical care pathways. In the UK, the study was registered at each site as either a clinical audit or service evaluation; at the lead centre (University Hospital Birmingham) it was approved as clinical audit, with registration CARMS-15986. In other countries, local principal investigators were responsible for contacting competent research ethics committees to obtain local or national approvals in line with applicable regulations, as well as seeking approvals from data protection officers. In some participating hospitals, informed patient consent was taken, whereas in other countries the requirement for patient consent was waived by local research ethics committees.
Participants
Each participating hospital included all patients undergoing surgery who had SARS-CoV-2 infection diagnosed within 7 days before or 30 days after surgery. Surgery was defined as any procedure done by a surgeon in an operating theatre under general, regional, or local anaesthesia. Patients undergoing surgery for any indication were eligible, including benign disease, cancer, trauma, and obstetrics. The study included children and adults, but individual hospitals had the option to apply local age cutoffs, if appropriate. If patients with SARS-CoV-2 infection had multiple operations, the procedure closest to the time of confirmation of SARS-CoV-2 infection was defined as the index procedure.
Participating hospitals prospectively screened patients for eligibility to ensure that all patients fulfilling eligibility criteria were captured. However, the study was initiated after the SARS-CoV-2 pandemic had peaked in some regions, so retrospective data collection was permitted if collaborators were able to identify and include all eligible patients. The importance of working across surgical specialties to identify all eligible patients was highlighted in site training, because incomplete case ascertainment could introduce bias, if patients with less severe disease were missed. Site investigators were provided with a range of written materials setting out possible strategies to capture consecutive eligible patients. In addition, investigators were invited to join social media groups and teleconferences for the purpose of troubleshooting site-specific recruitment issues and shared learning.
Procedures
Laboratory testing for SARS-CoV-2 infection was based on viral RNA detection by quantitative RT-PCR. Sampling, including nasal swabs or bronchoalveolar lavage, and analyses were done according to individual hospital protocols.
As quantitative RT-PCR testing was not available at all participating hospitals, patients were also included based on either clinical or radiological findings. Clinical diagnosis consistent with SARS-CoV-2 infection was made by a senior physician and based on clinical presentation of symptoms highly indicative of SARS-CoV-2 infection, including cough, fever, and myalgia.12 Radiological diagnosis was based on thorax CT, in keeping with locally implemented protocols. All patients included initially based on clinical or radiological criteria who subsequently had laboratory testing for SARS-CoV-2 infection and returned a negative result were excluded from the study.
Data were collected online using the Research Electronic Data Capture web application. Demographic variables recorded included age, sex, and American Society of Anesthesiologists (ASA) physical status classification. Age was collected as a categorical variable by deciles of age. ASA at the time of surgery was analysed as grades 1–2 versus grades 3–5. The timing of SARS-CoV-2 diagnosis was recorded as either preoperative or postoperative. Clinical symptoms present at the time of hospital admission were recorded for emergency admissions. Physiological variables recorded (respiratory rate, heart rate, and blood pressure) were based on readings taken immediately before surgery. The quick sequential organ failure assessment score13 was calculated on the basis of individual variables recorded immediately before surgery. Operative variables included urgency (elective or emergency surgery), primary procedure completed, and anaesthesia used (local, regional, or general). Emergency surgery was defined as procedures classified by the National Confidential Enquiry into Patient Outcome and Death as immediate, urgent, or expedited.14 Grade of surgery was categorised on the basis of the Bupa schedule of procedures as either minor (minor or intermediate according to the Bupa schedule) or major (major or complex major according to the Bupa schedule). Before locking of the dataset for analysis, the senior local principal investigator for each hospital was asked to confirm data completeness and that all eligible patients had been entered into the database.
Outcomes
The primary outcome was 30-day mortality, with the day of surgery defined as day 0. The key secondary outcome measure was the rate of pulmonary complications, a composite outcome adapted from the Prevention of Respiratory Insufficiency after Surgical Management trial.15, 16 Pulmonary complications were defined as pneumonia, acute respiratory distress syndrome (ARDS), or unexpected postoperative ventilation; these are the most frequent COVID-19-related pulmonary complications in medical patients.12 Unexpected postoperative ventilation was defined as either any episode of non-invasive ventilation, invasive ventilation, or extracorporeal membrane oxygenation after initial extubation after surgery; or patient could not be extubated as planned after surgery. Additional secondary outcomes included pulmonary embolism, intensive care unit admission, reoperation, 7-day mortality, and length of hospital stay.
Statistical analysis
The study was done according to STROBE guidelines for observational studies.17 Continuous data were tested for distribution, with normally distributed data presented as mean and 95% CI, and differences between groups were tested using the unpaired t test. The χ2 and Fisher's exact tests were used for categorical data. Missing data were included in flowcharts and descriptive analyses, allowing denominators to remain consistent in calculations.
Multilevel logistic regression was used to calculate odds ratios (ORs) and 95% CIs. Models included factors that occurred before the outcome of interest. Country was included as a random effect with hospital nested within country, in both the unadjusted and adjusted models. The primary adjusted model included preoperative variables to identify predictors of 30-day mortality. Secondary models identified predictors of 7-day mortality and pulmonary complications. Sensitivity analyses were done, including only patients with laboratory-confirmed SARS-CoV-2 infection; and only patients with preoperatively confirmed SARS-CoV-2 infection. Analyses were done using Stata, version 15.1 for Mac.
Role of the funding source
The funders of the study had no role in study design, data collection, data analysis, data interpretation, or writing of the report. The corresponding author and analysis group had full access to all the data in the study and the corresponding author and the writing committee had final responsibility for the decision to submit for publication.
Results
At the time of analysis (May 2, 2020), 30-day follow-up had been reached for 1128 patients who had surgery between Jan 1 and March 31, 2020. 605 (53·6%) of 1128 patients were men and 523 (46·4%) were women, 214 (19·0%) were younger than 50 years, 353 (31·3%) were aged 50–69 years, and 558 (49·5%) were aged 70 years or older, with age missing for three patients (table 1).
Table 1.
Baseline and demographic characteristics
|
30-day mortality |
Pulmonary complications |
|||||||
|---|---|---|---|---|---|---|---|---|
| No (n=845) | Yes (n=268) | p value | No (n=526) | Yes (n=577) | p value | |||
| Age | .. | .. | <0·0001 | .. | .. | 0·00023 | ||
| <29 years | 56 (100%) | 0 | .. | 39 (70·9%) | 16 (29·1%) | .. | ||
| 30–49 years | 146 (94·2%) | 9 (5·8%) | .. | 86 (55·8%) | 68 (44·2%) | .. | ||
| 50–69 years | 277 (79·8%) | 70 (20·2%) | .. | 159 (46·0%) | 187 (54·0%) | .. | ||
| ≥70 years | 364 (65·9%) | 188 (34·1%) | .. | 240 (44·0%) | 305 (56·0%) | .. | ||
| Missing | 2 | 1 | .. | 2 | 1 | .. | ||
| Sex | .. | .. | <0·0001 | .. | .. | 0·0028 | ||
| Male | 424 (71·1%) | 172 (28·9%) | .. | 252 (42·8%) | 337 (57·2%) | .. | ||
| Female | 417 (81·6%) | 94 (18·4%) | .. | 270 (53·1%) | 238 (46·9%) | .. | ||
| Ambiguous | 1 (50·0%) | 1 (50·0%) | .. | 1 (50·0%) | 1 (50·0%) | .. | ||
| Missing | 3 | 1 | .. | 3 | 1 | .. | ||
| American Society of Anesthesiologists grade | .. | .. | <0·0001 | .. | .. | <0·0001 | ||
| 1–2 | 344 (88·4%) | 45 (11·6%) | .. | 235 (60·6%) | 153 (39·4%) | .. | ||
| 3–5 | 475 (68·7%) | 216 (31·3%) | .. | 278 (40·6%) | 407 (59·4%) | .. | ||
| Missing | 26 | 7 | .. | 13 | 17 | .. | ||
| Number of comorbidities | .. | .. | <0·0001 | .. | .. | 0·00017 | ||
| None | 107 (93·0%) | 8 (7·0%) | .. | 73 (63·5%) | 42 (36·5%) | .. | ||
| One | 192 (82·8%) | 40 (17·2%) | .. | 115 (50·7%) | 112 (49·3%) | .. | ||
| Two or more | 527 (70·8%) | 217 (29·2%) | .. | 322 (43·5%) | 418 (56·5%) | .. | ||
| Missing | 19 | 3 | .. | 16 | 5 | .. | ||
| Comorbidities | ||||||||
| Current smoker | 80 (75·5%) | 26 (24·5%) | 0·909 | 42 (40·0%) | 63 (60·0%) | 0·097 | ||
| Asthma | 57 (73·1%) | 21 (26·9%) | 0·542 | 36 (48·0%) | 39 (52·0%) | 0·955 | ||
| Cancer | 146 (77·2%) | 43 (22·8%) | 0·639 | 92 (48·9%) | 96 (51·1%) | 0·707 | ||
| Chronic kidney disease | 109 (66·5%) | 55 (33·5%) | 0·0022 | 64 (39·3%) | 99 (60·7%) | 0·020 | ||
| Chronic obstructive pulmonary disease | 75 (64·7%) | 41 (35·3%) | 0·0027 | 44 (37·9%) | 72 (62·1%) | 0·026 | ||
| Congestive heart failure | 55 (64·7%) | 30 (35·3%) | 0·012 | 29 (34·5%) | 55 (65·5%) | 0·012 | ||
| Dementia | 48 (55·2%) | 39 (44·8%) | <0·0001 | 30 (35·3%) | 55 (64·7%) | 0·017 | ||
| Diabetes | 207 (73·9%) | 73 (26·1%) | 0·367 | 124 (44·1%) | 157 (55·9%) | 0·166 | ||
| Hypertension | 399 (71·0%) | 163 (29·0%) | 0·00010 | 253 (45·3%) | 305 (54·7%) | 0·114 | ||
| Myocardial infarction | 70 (63·1%) | 41 (36·9%) | 0·00084 | 39 (35·4%) | 71 (64·6%) | 0·0068 | ||
| Peripheral vascular disease | 67 (62·0%) | 41 (38·0%) | 0·00038 | 48 (44·4%) | 60 (55·6%) | 0·477 | ||
| Stoke or transient ischaemic attack | 55 (61·1%) | 35 (38·9%) | 0·00061 | 45 (50·0%) | 45 (50·0%) | 0·647 | ||
| Symptoms at admission* | ||||||||
| No symptoms reported | 111 (77·6%) | 32 (22·4%) | 0·281 | 78 (56·5%) | 60 (43·5%) | 0·020 | ||
| Symptoms reported | 499 (73·3%) | 182 (26·7%) | .. | 309 (45·6%) | 368 (54·4%) | .. | ||
| Abdominal pain | 193 (77·5%) | 56 (22·5%) | 0·134 | 122 (49·4%) | 125 (50·6%) | 0·472 | ||
| Dyspnoea | 83 (61·9%) | 51 (38·1%) | 0·00049 | 32 (23·9%) | 102 (76·1%) | <0·0001 | ||
| Cough | 108 (73·0%) | 40 (27·0%) | 0·746 | 55 (37·2%) | 93 (62·8%) | 0·0054 | ||
| Diarrhoea | 18 (69·2%) | 8 (30·8%) | 0·571 | 12 (46·2%) | 14 (53·8%) | 0·890 | ||
| Fatigue | 42 (70·0%) | 18 (30·0%) | 0·460 | 18 (30·0%) | 42 (70·0%) | 0·0048 | ||
| Fever >38°C | 177 (76·6%) | 54 (23·4%) | 0·289 | 94 (40·9%) | 136 (59·1%) | 0·018 | ||
| Haemoptysis | 2 (66·7%) | 1 (33·3%) | 0·771 | 1 (33·3%) | 2 (66·7%) | 0·623 | ||
| Myalgia | 27 (79·4%) | 7 (20·6%) | 0·465 | 9 (26·5%) | 25 (73·5%) | 0·012 | ||
| Nausea or vomiting | 100 (79·4%) | 26 (20·6%) | 0·138 | 62 (49·6%) | 63 (50·4%) | 0·607 | ||
| Sputum | 7 (41·2%) | 10 (58·8%) | 0·0018 | 6 (35·3%) | 11 (64·7%) | 0·309 | ||
| Other | 209 (70·6%) | 87 (29·4%) | 0·094 | 139 (47·3%) | 155 (52·7%) | 0·930 | ||
| Preoperative respiratory support | ||||||||
| None or oxygen only | 805 (76·4%) | 249 (23·6%) | 0·134 | 520 (49·7%) | 526 (50·3%) | <0·0001 | ||
| Non-invasive ventilation | 12 (80·0%) | 3 (20·0%) | 0·710 | 1 (6·7%) | 14 (93·3%) | 0·0014 | ||
| Invasive ventilation | 31 (66·0%) | 16 (34·0%) | 0·103 | 2 (4·3%) | 45 (95·7%) | <0·0001 | ||
| Last available values before surgery | ||||||||
| Systolic blood pressure, mm Hg† | 129·0 (22·6) | 131·7 (26·0) | 0·118 | 129·9 (22·0) | 129·7 (24·6) | 0·896 | ||
| Respiratory rate, rpm† | 18·1 (5·5) | 18·7 (8·8) | 0·211 | 17·5 (5·6) | 18·9 (7·1) | 0·0013 | ||
| Heart rate, bpm† | 85·0 (18·3) | 83·0 (19·0) | 0·130 | 83·4 (16·9) | 85·3 (19·6) | 0·081 | ||
| qSOFA score | .. | .. | 0·011 | .. | .. | <0·0001 | ||
| 0 | 572 (76·3%) | 178 (23·7%) | .. | 382 (51·3%) | 362 (48·7%) | .. | ||
| 1 | 155 (77·5%) | 45 (22·5%) | .. | 73 (36·7%) | 126 (63·3%) | .. | ||
| ≥2 | 37 (59·7%) | 25 (40·3%) | .. | 9 (14·8%) | 52 (82·2%) | .. | ||
| Missing | 81 | 20 | .. | 62 | 37 | .. | ||
Data only presented for patients with 30-day mortality outcome available (n=1113%) and pulmonary complications outcome available (n=1103%). Percentages are presented in rows. bpm=beats per min. qSOFA=quick sequential organ failure assessment. rpm=breaths per minutes.
Data only presented for emergency patients.
Data presented as mean with SD.
SARS-CoV-2 infection was diagnosed preoperatively in 294 (26·1%) of 1128 patients and postoperatively in 806 (71·5%), with timing of diagnosis missing for 28 patients. SARS-CoV-2 diagnosis was confirmed by laboratory testing in 969 (85·9%) patients, radiological findings in 80 (7·1%), and clinical findings in 68 (6·0%), with method of diagnosis missing for 11 patients. Overall, 357 (31·6%) had preoperative thorax CT and the most common radiological finding was ground glass opacity (table 2).
Table 2.
Preoperative assessment
|
30-day mortality |
Pulmonary complications |
||||||
|---|---|---|---|---|---|---|---|
| No (n=845) | Yes (n=268) | p value | No (n=526) | Yes (n=577) | p value | ||
| Haemoglobin, g/L* | 118·6 (24·7) | 116·1 (24·1) | 0·150 | 118·5 (23·5) | 117·6 (25·4) | 0·537 | |
| Missing | 18 | 4 | .. | 15 | 7 | .. | |
| White blood cell count, ×109 per L* | 10·5 (7·6) | 10·6 (6·8) | 0·859 | 10·1 (5·1) | 10·8 (8·9) | 0·169 | |
| Missing | 19 | 4 | .. | 15 | 8 | .. | |
| Preoperative chest x-ray | .. | .. | 0·0041 | .. | .. | <0·0001 | |
| Not performed | 320 (79·4%) | 83 (20·6%) | .. | 232 (58·0%) | 168 (42·0%) | .. | |
| Yes: normal | 321 (77·4%) | 94 (22·6%) | .. | 205 (49·8%) | 207 (50·2%) | .. | |
| Yes: abnormal | 199 (68·9%) | 90 (31·1%) | .. | 84 (29·4%) | 202 (70·6%) | .. | |
| Missing | 5 | 1 | .. | 5 | 0 | .. | |
| Preoperative thorax CT | |||||||
| Not performed | 598 (78·1%) | 168 (21·9%) | 0·013 | 376 (49·5%) | 384 (50·5%) | 0·077 | |
| Performed: normal | 96 (75·0%) | 32 (25·0%) | 0·796 | 60 (47·6%) | 66 (52·4%) | 0·987 | |
| Performed: consolidation | 44 (75·9%) | 14 (24·14%) | 0·991 | 23 (39·7%) | 35 (60·3%) | 0·208 | |
| Performed: ground glass opacity | 57 (71·3%) | 23 (28·7%) | 0·310 | 31 (39·2%) | 48 (60·8%) | 0·119 | |
| Performed: pulmonary infiltration | 27 (67·5%) | 13 (32·5%) | 0·205 | 13 (33·3%) | 26 (66·7%) | 0·068 | |
| Performed: other abnormality | 50 (61·0%) | 32 (39·0%) | 0·0010 | 30 (37·0%) | 51 (63·0%) | 0·046 | |
| SARS-CoV-2 diagnosis | .. | .. | 0·719 | .. | .. | 0·085 | |
| Laboratory confirmed | 727 (76·0%) | 230 (24·0%) | .. | 454 (47·9%) | 493 (52·1%) | .. | |
| Radiological (CT thorax) | 58 (72·5%) | 22 (27·5%) | .. | 29 (36·3%) | 51 (63·7%) | .. | |
| Clinical | 53 (77·9%) | 15 (22·1%) | .. | 36 (52·9%) | 32 (47·1%) | .. | |
| Missing | 7 | 1 | .. | 7 | 1 | .. | |
| Timing of SARS-CoV-2 diagnosis | .. | .. | 0·128 | .. | .. | 0·155 | |
| Preoperative | 231 (78·8%) | 62 (21·2%) | .. | 148 (51·0%) | 142 (49·0%) | .. | |
| Postoperative | 595 (74·4%) | 205 (25·6%) | .. | 367 (46·2%) | 428 (53·8%) | .. | |
| Missing | 19 | 1 | .. | 11 | 7 | .. | |
Data only presented for patients with 30-day mortality outcome available (n=1113) and pulmonary complications outcome available (n=1103). Percentages are presented in rows. SARS-CoV-2=severe acute respiratory syndrome coronavirus 2.
Last available blood test results from before surgery, presented as mean with SD.
Emergency surgery was done in 835 (74·0%) of 1128 patients and elective surgery in 280 (24·8%; table 3), with urgency missing for 13 patients. Indications for surgery were benign disease in 615 (54·5%), cancer in 278 (24·6%), and trauma in 227 (20·1%), with indication missing for eight patients. 251 (22·3%) procedures were categorised as minor and 841 (74·6%) as major, with grade of surgery missing for 36 patients. Procedures included gastrointestinal and general (373 [33·1%]), orthopaedic (302 [26·8%]), cardiothoracic (86 [7·6%]), hepatobiliary (62 [5·5%]), obstetric (51 [4·5%]), vascular (45 [4·0%]), head and neck (40 [3·5%]), neurosurgery (39 [3·5%]), urological (37 [3·3%]), and other (58 [5·1%]) surgeries. Procedure type was missing for 36 patients. A full breakdown of procedures is in the appendix (pp 11–14).
Table 3.
Operative details
|
30-day mortality |
Pulmonary complications |
||||||
|---|---|---|---|---|---|---|---|
| No (n=845) | Yes (n=268) | p value | No (n=526) | Yes (n=577) | p value | ||
| Urgency of surgery | .. | .. | 0·020 | .. | .. | 0·873 | |
| Elective | 225 (80·9%) | 53 (19·1%) | .. | 130 (46·9%) | 147 (53·1%) | .. | |
| Emergency | 610 (74·0%) | 214 (26·0%) | .. | 387 (47·5%) | 428 (52·5%) | .. | |
| Missing | 10 | 1 | .. | 9 | 2 | .. | |
| Anaesthesia | .. | .. | 0·383 | .. | .. | 0·488 | |
| Local | 34 (69·4%) | 15 (30·6%) | .. | 24 (49·0%) | 25 (51·0%) | .. | |
| Regional | 119 (78·8%) | 32 (21·2%) | .. | 78 (51·7%) | 73 (48·3%) | .. | |
| General | 658 (75·2%) | 217 (24·8%) | .. | 403 (46·5%) | 464 (53·5%) | .. | |
| Missing | 34 | 4 | .. | 21 | 15 | .. | |
| Surgical diagnosis | .. | .. | 0·030 | .. | .. | 0·502 | |
| Benign or obstetric case | 480 (78·3%) | 133 (21·7%) | .. | 281 (46·3%) | 326 (53·7%) | .. | |
| Cancer | 183 (72·9%) | 68 (27·1%) | .. | 114 (45·6%) | 136 (54·4%) | .. | |
| Trauma | 157 (70·1%) | 67 (29·9%) | .. | 112 (50·5%) | 110 (49·6%) | .. | |
| Missing | 25 | 0 | .. | 19 | 5 | .. | |
| Grade of surgery | .. | .. | 0·00055 | .. | .. | 0·022 | |
| Minor | 209 (83·6%) | 41 (16·4%) | .. | 132 (53·2%) | 116 (46·8%) | .. | |
| Major | 607 (72·9%) | 226 (27·1%) | .. | 372 (45·0%) | 455 (55·0%) | .. | |
| Missing | 29 | 1 | .. | 22 | 6 | .. | |
| Specialty | .. | .. | <0·0001 | .. | .. | <0·0001 | |
| Breast | 3 (100·0%) | 0 (0%) | .. | 2 (66·6%) | 1 (33·3%) | .. | |
| Cardiac | 33 (66·0%) | 17 (34·0%) | .. | 3 (5·9%) | 48 (94·1%) | .. | |
| Gastrointestinal and general | 286 (76·9%) | 86 (23·1%) | .. | 172 (46·4%) | 199 (53·6%) | .. | |
| Gynaecology | 20 (95·2%) | 1 (4·8%) | .. | 16 (76·2%) | 5 (23·8%) | .. | |
| Head and neck | 32 (80·0%) | 8 (20·0%) | .. | 10 (25·6%) | 29 (74·4%) | .. | |
| Hepatobiliary | 50 (84·8%) | 9 (15·2%) | .. | 29 (50·9%) | 28 (49·1%) | .. | |
| Neurosurgery | 31 (81·6%) | 7 (18·4%) | .. | 19 (50·0%) | 19 (50·0%) | .. | |
| Obstetrics | 50 (98·0%) | 1 (2·0%) | .. | 26 (51·0%) | 25 (49·0%) | .. | |
| Ophthalmology | 4 (100·0%) | 0 (0%) | .. | 3 (75·0%) | 1 (25·0%) | .. | |
| Orthopaedics | 213 (71·2%) | 86 (28·8%) | .. | 165 (55·7%) | 131 (44·3%) | .. | |
| Other | 19 (73·1%) | 7 (26·9%) | .. | 11 (42·3%) | 15 (57·7%) | .. | |
| Plastic and reconstructive | 3 (100·0%) | 0 (0%) | .. | 1 (33·3%) | 2 (66·7%) | .. | |
| Thoracic | 20 (57·1%) | 15 (42·9%) | .. | 12 (34·3%) | 23 (65·7%) | .. | |
| Urology | 25 (67·6%) | 12 (32·4%) | .. | 15 (42·3%) | 20 (57·1%) | .. | |
| Vascular | 27 (60·0%) | 18 (40·0%) | .. | 20 (44·4%) | 25 (55·6%) | .. | |
| Missing | 29 (96·7%) | 1 (3·3%) | .. | 22 (78·6%) | 6 (21·4%) | .. | |
Data only presented for patients with 30-day mortality outcome available (n=1113) and pulmonary complications outcome available (n=1103). Percentages are presented in rows.
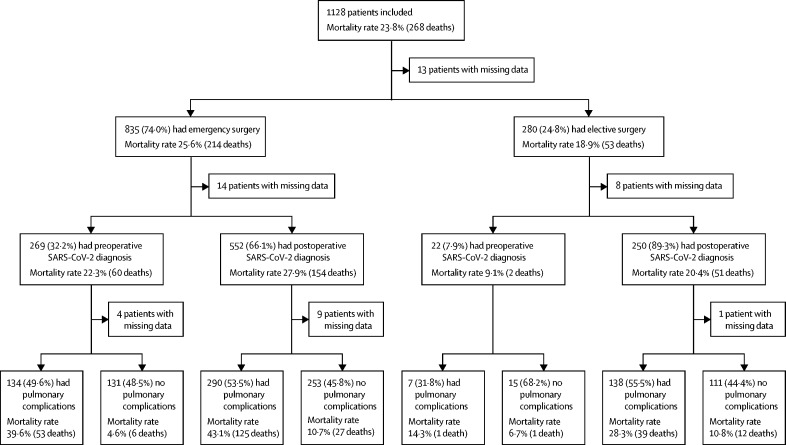
30-day mortality was 23·8% (268 of 1128). Men had higher 30-day mortality than women (28·4% [172 of 605] vs 18·2% [94 of 517], p<0·0001). Patients aged 70 years or older had higher mortality than patients younger than 70 years (33·7% [188 of 558] vs 13·9% [79 of 567], p<0·0001). Mortality was higher after emergency surgery (25·6% [214 of 835]) than elective surgery (18·9% [53 of 280]; p=0·023; figure 1). Men had higher mortality rates than women, and men and women aged 70 years or older had higher rates than those younger than 70 years (figure 2).
Figure 1.
30-day mortality rates by timing of surgery and development of pulmonary complications
Patients with missing data are included in denominators (appendix p 21). Pulmonary complications are pneumonia, acute respiratory distress syndrome, or unexpected postoperative ventilation. SARS-CoV-2=severe acute respiratory syndrome coronavirus 2.
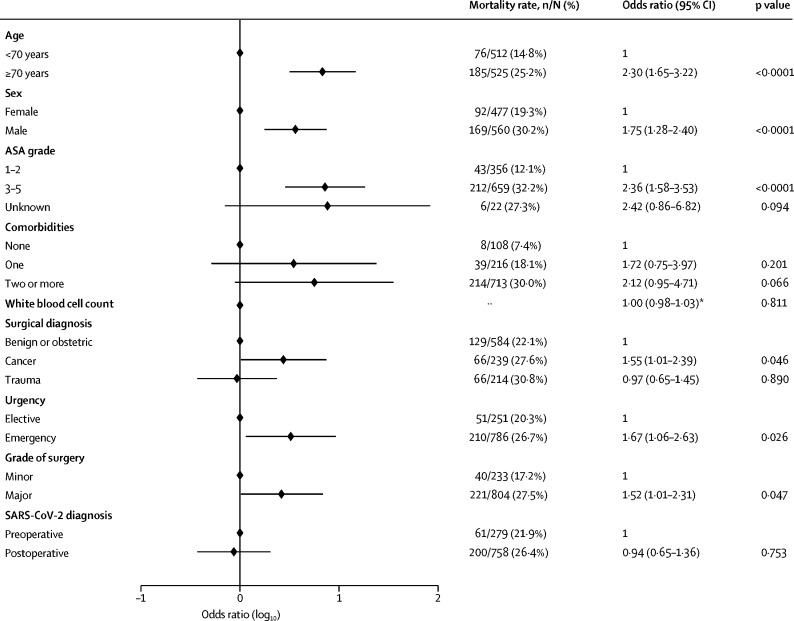
Figure 2.
30-day mortality rates by patient subgroup
Grade of surgery was classified based on the Bupa Schedule as either minor (minor or intermediate in Bupa Schedule) or major (major or complex major in Bupa Schedule).
In adjusted analyses (figure 3; appendix p 15), predictors of 30-day mortality were male sex (OR 1·75 [95% CI 1·28–2·40], p<0·0001), age 70 years or older versus younger than 70 years (2·30 [1·65–3·22], p<0·0001), ASA grades 3–5 versus grades 1–2 (2·35 [1·57–3·53], p<0·0001), malignant versus benign or obstetric diagnosis (1·55 [1·01–2·39], p=0·046), emergency versus elective surgery (1·67 [1·06–2·63], p=0·026), and major versus minor surgery (1·52, [1·01–2·31], p=0·047).
Figure 3.
Adjusted model of predictors for 30-day mortality
1037 patients with complete data were included in the adjusted model. Of the patients excluded because of missing data, seven had died and 84 patients had not died at 30 days. ASA=American Society of Anesthesiologists. SARS-CoV-2=severe acute respiratory syndrome coronavirus 2. *Adjusted odds ratio reported per unit increase in white blood cell count (×109).
7-day mortality was 5·2% (59 of 1128; table 4). In adjusted analyses (appendix p 16), having ASA grades 3–5 versus grades 1–2 was associated with increased odds of 7-day mortality (OR 2·52 [95% CI 1·10–5·77], p<0·029), whereas postoperative diagnosis was associated with decreased risk (0·25 [0·13–0·46], p<0·0001).
Table 4.
Postoperative outcomes
|
Urgency |
Grade of surgery |
|||||||
|---|---|---|---|---|---|---|---|---|
| Elective (n=280) | Emergency (n=835) | p value | Minor (n=251) | Major (n=841) | p value | |||
| Mortality | ||||||||
| 7-day | 7 (2·5%) | 52 (6·2%) | 0·015 | 8 (3·2%) | 51 (6·1%) | 0·074 | ||
| 30-day | 53 (18·9%) | 214 (25·6%) | 0·020 | 41 (16·3%) | 226 (26·9%) | 0·00055 | ||
| Missing | 2 (0·7%) | 11 (1·3%) | .. | 1 (0·4%) | 8 (1·0%) | .. | ||
| Pulmonary complications | ||||||||
| Composite of pulmonary complications | 147 (52·5%) | 428 (51·3%) | 0·873 | 116 (46·2%) | 455 (54·1%) | 0·022 | ||
| Pneumonia | 118 (42·1%) | 334 (40·0%) | 0·527 | 94 (37·5%) | 355 (42·2%) | 0·178 | ||
| Acute respiratory distress syndrome | 41 (14·6%) | 119 (14·3%) | 0·872 | 33 (13·2%) | 127 (15·1%) | 0·442 | ||
| Unexpected postoperative ventilation | .. | .. | 0·262 | .. | .. | 0·160 | ||
| Non-invasive ventilation | 23 | 31 | .. | 12 | 41 | .. | ||
| Invasive ventilation | 40 | 156 | .. | 41 | 153 | .. | ||
| Missing | 3 | 21 | .. | 4 | 14 | .. | ||
| Duration of invasive ventilation | .. | .. | 0·049 | .. | .. | 0·023 | ||
| 1–23 h | 16 | 32 | .. | 7 | 41 | .. | ||
| 24–47 h | 5 | 27 | .. | 3 | 28 | .. | ||
| 48–71 h | 2 | 21 | .. | 3 | 20 | .. | ||
| ≥72 h | 17 | 79 | .. | 29 | 66 | .. | ||
| Missing | 240 | 676 | .. | 209 | 686 | .. | ||
| Pulmonary embolism | ||||||||
| 30-day | 4 (1·4%) | 18 (2·2%) | 0·449 | 8 (3·2%) | 14 (1·7%) | 0·132 | ||
| Missing | 3 | 21 | .. | 4 | 14 | .. | ||
| Postoperative intensive care unit admission | .. | .. | 0·0034 | .. | .. | 0·177 | ||
| None | 158 (56·4%) | 570 (68·3%) | .. | 177 (70·5%) | 538 (64·0%) | .. | ||
| Planned | 64 (22·9%) | 189 (22·6%) | .. | 46 (18·3%) | 203 (24·1%) | .. | ||
| Unplanned from theatre | 16 (5·7%) | 25 (3·0%) | .. | 10 (4·0%) | 31 (3·7%) | .. | ||
| Unplanned from ward | 23 (8·2%) | 38 (4·6%) | .. | 17 (6·8%) | 43 (5·1%) | .. | ||
| Missing | 19 (6·8%) | 13 (1·6%) | .. | 1 (0·4%) | 26 (3·1%) | .. | ||
| Reoperation | .. | .. | 0·0015 | .. | .. | 0·487 | ||
| Reoperated | 53 (18·9%) | 101 (12·1%) | .. | 39 (15·5%) | 115 (13·7%) | .. | ||
| Not reoperated | 209 (74·6%) | 717 (85·9%) | .. | 207 (82·5%) | 702 (83·5%) | .. | ||
| Missing | 18 (6·4%) | 17 (2·0%) | .. | 5 (2·0%) | 24 (2·9%) | .. | ||
| Length of stay | ||||||||
| Median (IQR), days | 13 (5–28) | 16 (7–28) | 0·012 | 10 (3–27) | 17 (8–29) | <0·0001 | ||
| >30 days | 64 (22·9%) | 168 (20·1%) | 0·352 | 52 (20·7%) | 176 (20·9%) | 0·911 | ||
| Missing | 2 (0·7%) | 11 (1·3%) | .. | 1 (0·4%) | 8 (1·0%) | .. | ||
Urgency data missing for 13 patients and grade of surgery data missing for 36 patients. Percentages shown are based on the denominator of patients in the subgroup.
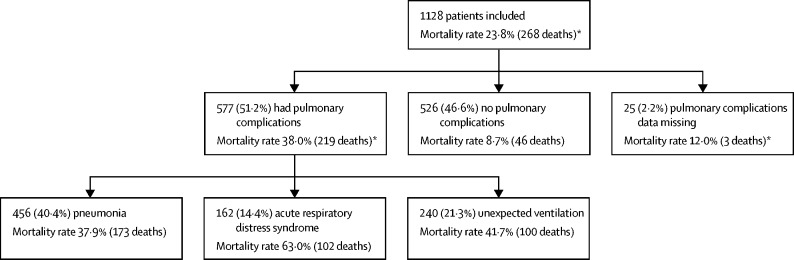
577 (51·2%) of 1128 patients had at least one pulmonary complication (figure 4): 456 (40·4%) had pneumonia, 240 (21·3%) had unexpected ventilation, and 162 (14·4%) had ARDS. Patients who developed pulmonary complications had a higher 30-day mortality than those who did not (38·0% [219 of 577] versus 8·7% [46 of 526], p<0·0001). Pulmonary complications had occurred in 219 (81·7%) of 268 patients who died. Among patients who developed pulmonary complications, 30-day mortality was highest in those who developed ARDS (102 [63·0%] of 162). Pulmonary complications were associated with high 30-day mortality rates across elective patients with a postoperative SARS-CoV-2 diagnosis (39 [28·3%] of 138), emergency patients with a preoperative SARS-CoV-2 diagnosis (53 [39·6%] of 134), and emergency patients with a postoperative SARS-CoV-2 diagnosis (125 [43·1%] of 290; figure 1). Pulmonary complication rates were similar in patients with laboratory-confirmed and clinically diagnosed SARS-CoV-2 infection (493 [50·9%] of 969 vs 32 [47·1%] of 68, p=0·543).
Figure 4.
30-day mortality rates associated with components of pulmonary complications
Relationships between the pulmonary complications are in the appendix (p 20). *Mortality data were missing for 15 patients; pulmonary complications data were also missing for 14 of these patients, the other one patient had a pulmonary complication (unexpected ventilation).
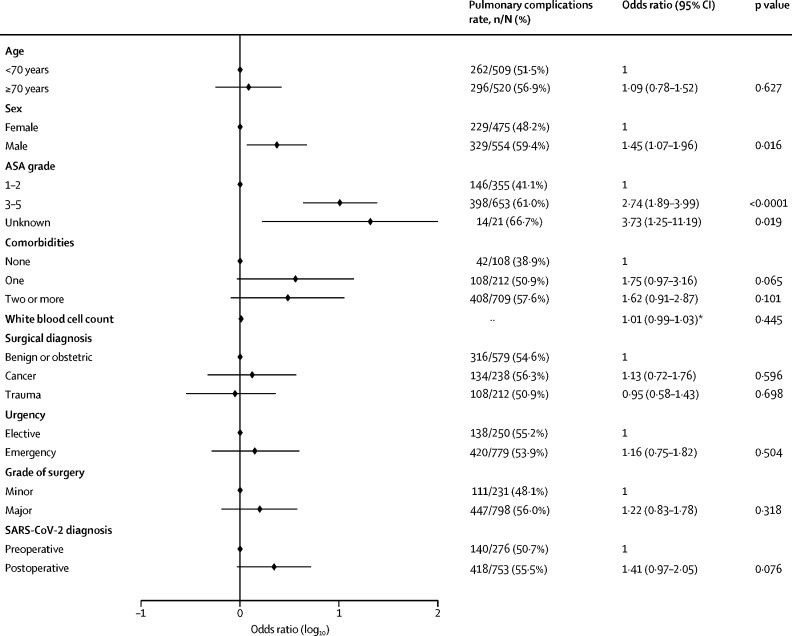
In adjusted analyses (figure 5; appendix p 17) pulmonary complications were independently associated with ASA grades 3–5 versus grades 1–2 (2·74 [95% CI 1·89–3·99], p<0·0001).
Figure 5.
Adjusted model of predictors for pulmonary complications
1029 patients with complete data are included in the adjusted model. Of the patients excluded because of missing data, 19 developed pulmonary complications and 80 patients did not. ASA=American Society of Anesthesiologists. SARS-CoV-2=severe acute respiratory syndrome coronavirus 2.
At 30 days, pulmonary embolism had occurred in 22 (2·0%) of 1128 patients. The 30-day mortality rate in patients with pulmonary embolism was similar to that in patients who did not have pulmonary embolism (five [22·7%] of 22 vs 263 [23·8%] of 1106, p=0·909).
In a sensitivity analysis including only patients with laboratory-confirmed SARS-CoV-2, the overall 30-day mortality rate was 23·7% (230 of 969), and pulmonary complications occurred in 493 (50·9%) of 969 patients. In adjusted analyses (appendix p 18), predictors of 30-day mortality were consistent with the main analysis: male sex, age 70 years or older, ASA grades 3–5, cancer surgery, and emergency surgery. The only independent predictor for 30-day pulmonary complications was ASA grades 3–5.
In a sensitivity analysis including only patients with preoperatively diagnosed SARS-CoV-2, the overall 30-day mortality rate was 21·1% (62 of 294), and pulmonary complications occurred in 142 (48·3%) of 294 patients. In adjusted analyses (appendix p 19), predictors of 30-day mortality were male sex and ASA grades 3–5. The only independent predictor for 30-day pulmonary complications was ASA grades 3–5.
Discussion
This study identified that postoperative pulmonary complications occur in half of patients with perioperative SARS-CoV-2 infection and are associated with high mortality. This has direct implications for clinical practice around the world. The increased risks associated with SARS-CoV-2 infection should be balanced against the risks of delaying surgery in individual patients; this study identified men, people aged 70 years or older, those with comorbidities (ASA grades 3–5), those having cancer surgery, and those needing emergency or major surgery as being most vulnerable to adverse outcomes.
Thresholds for surgery during the SARS-CoV-2 pandemic should be higher than during normal practice. Men aged 70 years and over who have emergency or major elective surgery are at particularly high risk of mortality, although minor elective surgery is also associated with higher-than-usual mortality. During SARS-CoV-2 outbreaks, consideration should be given for postponing non-critical procedures and promoting non-operative treatment to delay or avoid the need for surgery.18
Postoperative outcomes in SARS-CoV-2-infected patients are substantially worse than pre-pandemic baseline rates of pulmonary complications and mortality. The overall 30-day mortality in this study was 23·8%, and was high across all patient subgroups; all-cause mortality rates were 18·9% in elective patients, 25·6% in emergency patients, 16·3% in patients who had minor surgery, and 26·9% in patients who had major surgery. SARS-CoV-2-infected patients had greater mortality than even the highest-risk subgroups of the UK's NELA. The 2019 NELA report presented 30-day mortality rates of 16·9% in patients with a high preoperative risk of death, 16·8% in patients with an unexpected critical care admission, and 23·4% in frail patients older than 70 years.19 The mortality rates identified in this study are also higher than those previously reported across international settings; a study across 58 countries, including low-income and middle-income countries, reported a 30-day mortality of 14·9% in the high-risk subgroup who had emergency midline laparotomy.20 Postoperative mortality rates in SARS-CoV-2-infected patients with postoperative pulmonary complications approach those of the sickest patients with community-acquired COVID-19 who are admitted to intensive care.21
Mortality in patients with SARS-CoV-2 was mainly in those who had postoperative pulmonary complications, which was about 50% of patients. This rate is far higher than the pre-pandemic baseline; in the POPULAR multicentre, prospective, observational study of 211 hospitals from 28 European countries in 2014–15, the pulmonary complication rate was 8%.5 In our study, ARDS had the highest mortality rate of the different complications (mortality 63·0%) and occurred much more frequently (20%) than reported in the pre-pandemic African Surgical Outcomes Study (0·05%).22 In another study of high-risk ASA grade 3 patients undergoing non-cardiac surgery in seven US centres, 0·2% developed ARDS, with an overall mortality related to postoperative pulmonary complications of 2·3%.23 Even considering differences in the case-mix, the incidence of and mortality associated with pulmonary complications in SARS-CoV-2-infected patients is disproportionately high.
This study has limitations. Protocols for laboratory testing and radiological interpretation were not standardised across participating centres. We describe outcomes in the early phases of the pandemic when routine testing was not available across all sites; setting study inclusion criteria requiring laboratory-confirmed SARS-CoV-2 would have excluded some infected patients. Therefore, patients who did not have a laboratory test or CT scan were eligible for inclusion on the basis of clinical diagnosis. Only a minority of patients (6·0%) were included on the basis of a clinical diagnosis and these patients had similar clinical outcomes to patients with laboratory-confirmed SARS-CoV-2. The limitations of laboratory testing mean that some infected patients were excluded from the study based on false negative laboratory test results. Future studies need to make recommendations on the role of preoperative testing in patient selection for surgery.
The study included patients having any type of surgery and although this has produced generalisable results, it is possible that in large hospitals investigators might have not identified all patients. To mitigate this, the importance of identifying and enrolling all eligible patients was highlighted in training packages for local site investigators and strategies to support comprehensive patient identification were shared regularly with all sites. Final case ascertainment and data completeness were confirmed with local principal investigators, creating as robust a dataset as possible. As far as we are aware, this is the first international study assessing mortality rates after surgery in patients with SARS-CoV-2 infection, and the first that reaches across all surgical specialties.24, 25, 26, 27 It was not feasible for all participating hospitals, many of which were experiencing significant stress, to collect data on all patients who had surgery during the pandemic period. Consequently, this study's findings should be interpreted with caution because they have been benchmarked against pulmonary complication and mortality rates from high-quality pre-pandemic studies, rather than against contemporaneous non-SARS-CoV-2-infected comparators.
Data were collected in hospitals with ongoing SARS-CoV-2 infection outbreaks, which were predominantly in Europe and North America at the time of this study. As the pandemic continues, the evidence this study provides will be relevant to countries where large-scale outbreaks might take place in the future. To facilitate rapid study approvals, this study has focused on key outcomes (mortality and pulmonary complications) that can be collected using routine data. To support decision making by patients and surgeons, future studies should collect longer-term and patient-centred outcomes.
When hospitals resume routine surgery, it is likely to be in environments that remain exposed to SARS-CoV-2. In the future, routine preoperative screening for SARS-CoV-2 might be possible with rapid tests that have low false positive rates, but hospital-acquired infection would remain a challenge.12, 28 Strategies are urgently required to minimise in-hospital SARS-CoV-2 transmission and mitigate the risk of postoperative pulmonary complications in SARS-CoV-2-infected patients whose surgery cannot be delayed.
This online publication has been corrected. The corrected version first appeared at thelancet.com on June 9, 2020
Data sharing
Data sharing requests will be considered by the management group upon written request to the corresponding author. Deidentified participant data or other prespecified data will be available subject to a written proposal and a signed data sharing agreement.
Acknowledgments
Acknowledgments
This study was funded by a National Institute for Health Research (NIHR) Global Health Research Unit Grant (NIHR 16.136.79), using UK aid from the UK Government to support global health research; the Association of Coloproctology of Great Britain and Ireland, Bowel and Cancer Research, Bowel Disease Research Foundation; Association of Upper Gastrointestinal Surgeons; British Association of Surgical Oncology; British Gynaecological Cancer Society; European Society of Coloproctology; NIHR Academy; Sarcoma UK; Vascular Society for Great Britain and Ireland; and Yorkshire Cancer Research. The views expressed are those of the authors and not necessarily those of the National Health Service, the NIHR, or the UK Department of Health and Social Care.
Contributors
The writing group (appendix p 1) contributed to study conception, protocol development, data collection, data interpretation, and critical revision of the manuscript. AB is the guarantor.
Declaration of interests
We declare no competing interests.
Contributor Information
COVIDSurg Collaborative:
James E Archer, Abdulrahman Odeh, Simon Ereidge, Hosni Khairy Salem, Gareth P Jones, Anne Gardner, Shiva S Tripathi, Anne Gregg, Reuben Jeganathan, Muhammad Harris Siddique, Celestino P Lombardi, Benjamin Martin, Cosimo Alex Leo, Debashis Dass, Gregorio Di Franco, Long R Jiao, Giulio Maria Mari, Luis-Cristobal Capitan-Morales, Tara M Connelly, Ammar Alanbuki, Armando De Virgilio, Clare Schilling, Carlos San Miguel Mendez, Gauri Kulkarni, Kulsoom Nizami, Stewart Walsh, Harry Dean, Miguel Ruiz-Marin, Rory Houston, Mario Trompetto, David Chrastek, Vasileios Kouritas, Maria Cannoletta, Francesco Rosato, Manish V Kaushal, Paulo Matos Costa, Hannah H Elkadi, Jack R Johnstone, Esmee Irvine, Maria R Alvarez, Carlo Corbellini, Gowtham S Venkatesan, Olga Mateo-Sierra, Carolina Martínez-Pérez, Javier Serrano González, Miguel Õngel Hernández Bartolomé, David Díaz Pérez, María Gutiérrez Samaniego, Pablo Galindo Jara, and Neil Sharma
Supplementary Material
References
- 1.WHO WHO announces COVID-19 outbreak a pandemic. March 12, 2020. http://www.euro.who.int/en/health-topics/health-emergencies/coronavirus-covid-19/news/news/2020/3/who-announces-covid-19-outbreak-a-pandemic
- 2.Horton R. Offline: COVID-19 and the NHS-“a national scandal”. Lancet. 2020;395 doi: 10.1016/S0140-6736(20)30727-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Besnier E, Tuech JJ, Schwarz L. We asked the experts: Covid-19 outbreak: is there still a place for scheduled surgery? “Reflection from pathophysiological data”. World J Surg. 2020;44:1695–1698. doi: 10.1007/s00268-020-05501-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Huang C, Wang Y, Li X. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kirmeier E, Eriksson LI, Lewald H. Post-anaesthesia pulmonary complications after use of muscle relaxants (POPULAR): a multicentre, prospective observational study. Lancet Respir Med. 2019;7:129–140. doi: 10.1016/S2213-2600(18)30294-7. [DOI] [PubMed] [Google Scholar]
- 6.Neto AS, da Costa LGV, Hemmes SNT. The LAS VEGAS risk score for prediction of postoperative pulmonary complications: an observational study. Eur J Anaesthesiol. 2018;35:691–701. doi: 10.1097/EJA.0000000000000845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Tu RH, Lin JX, Li P. Prognostic significance of postoperative pneumonia after curative resection for patients with gastric cancer. Cancer Med. 2017;6:2757–2765. doi: 10.1002/cam4.1163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.NELA Project Team Fourth patient report of the National Emergency Laparotomy Audit. October, 2018. https://www.hqip.org.uk/wp-content/uploads/2018/11/The-Fourth-Patient-Report-of-the-National-Emergency-Laparotomy-Audit-October-2018.pdf
- 9.Coccolini F, Perrone G, Chiarugi M. Surgery in COVID-19 patients: operational directives. World J Emerg Surg. 2020;15:25. doi: 10.1186/s13017-020-00307-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.COVIDSurg Collaborative Global guidance for surgical care during the COVID-19 pandemic. Br J Surg. 2020 doi: 10.1002/bjs.11646. published online April 15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Tao KX, Zhang BX, Zhang P. Recommendations for general surgery clinical practice in novel coronavirus pneumonia situation. Zhonghua Wai Ke Za Zhi. 2020;58:E001. doi: 10.3760/cma.j.issn.0529-5815.2020.0001. (in Chinese). [DOI] [PubMed] [Google Scholar]
- 12.Zhou F, Yu T, Du R. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395:1054–1062. doi: 10.1016/S0140-6736(20)30566-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Seymour CW, Liu VX, Iwashyna TJ. Assessment of Clinical Criteria for Sepsis: For the Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3) JAMA. 2016;315:762–774. doi: 10.1001/jama.2016.0288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.National Confidential Enquiry into Perioperative Deaths . National Confidential Enquiry into Perioperative Deaths; London: 2003. Who Operates When? II. A report by the National Confidential Enquiry into Perioperative Deaths 2001/02. [Google Scholar]
- 15.Abbott TEF, Fowler AJ, Pelosi P. A systematic review and consensus definitions for standardised end-points in perioperative medicine: pulmonary complications. Br J Anaesth. 2018;120:1066–1079. doi: 10.1016/j.bja.2018.02.007. [DOI] [PubMed] [Google Scholar]
- 16.Pearse RM, Abbott TE, Haslop R. The Prevention of Respiratory Insufficiency after Surgical Management (PRISM) Trial. Report of the protocol for a pragmatic randomized controlled trial of CPAP to prevent respiratory complications and improve survival following major abdominal surgery. Minerva Anestesiol. 2017;83:175–182. doi: 10.23736/S0375-9393.16.11502-0. [DOI] [PubMed] [Google Scholar]
- 17.von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. Lancet. 2007;370:1453–1457. doi: 10.1016/S0140-6736(07)61602-X. [DOI] [PubMed] [Google Scholar]
- 18.American College of Surgeons COVID-19: guidance for triage of non-emergent surgical procedures. March 17, 2020. https://www.facs.org/covid-19/clinical-guidance/triage
- 19.NELA Project Team Fifth patient report of the National Emergency Laparotomy Audit. December, 2019. https://www.nela.org.uk/downloads/EMBARGO%20-%20The%20Fifth%20Patient%20Report%20of%20the%20NELA%202019%20-%2027-11-19.pdf
- 20.GlobalSurg Collaborative Mortality of emergency abdominal surgery in high-, middle- and low-income countries. Br J Surg. 2016;103:971–988. doi: 10.1002/bjs.10151. [DOI] [PubMed] [Google Scholar]
- 21.Intensive Care National Audit & Research Centre ICNARC report on COVID-19 in critical care. March 27, 2020. https://www.icnarc.org/DataServices/Attachments/Download/b5f59585-5870-ea11-9124-00505601089b
- 22.Biccard BM, Madiba TE, Kluyts HL. Perioperative patient outcomes in the African Surgical Outcomes Study: a 7-day prospective observational cohort study. Lancet. 2018;391:1589–1598. doi: 10.1016/S0140-6736(18)30001-1. [DOI] [PubMed] [Google Scholar]
- 23.Fernandez-Bustamante A, Frendl G, Sprung J. Postoperative pulmonary complications, early mortality, and hospital stay following noncardiothoracic surgery: a multicenter study by the perioperative research network investigators. JAMA Surg. 2017;152:157–166. doi: 10.1001/jamasurg.2016.4065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kaafarani HMA, El Moheb M, Hwabejire JO. Gastrointestinal complications in critically ill patients with COVID-19. Ann Surg. 2020 doi: 10.1097/SLA.0000000000004004. published online May 1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Aminian A, Safari S, Razeghian-Jahromi A, Ghorbani M, Delaney CP. COVID-19 outbreak and surgical practice: unexpected fatality in perioperative period. Ann Surg. 2020 doi: 10.1097/SLA.0000000000003925. published online March 26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Cai M, Wang G, Zhang L. Performing abdominal surgery during the COVID-19 epidemic in Wuhan, China: a single-centred, retrospective, observational study. Br J Surg. 2020 doi: 10.1002/bjs.11643. published online April 27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lei S, Jiang F, Su W. Clinical characteristics and outcomes of patients undergoing surgeries during the incubation period of COVID-19 infection. EClinicalMedicine. 2020;21 doi: 10.1016/j.eclinm.2020.100331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Wang D, Hu B, Hu C. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus–infected pneumonia in Wuhan, China. JAMA. 2020;323:1061–1069. doi: 10.1001/jama.2020.1585. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data sharing requests will be considered by the management group upon written request to the corresponding author. Deidentified participant data or other prespecified data will be available subject to a written proposal and a signed data sharing agreement.