Abstract
The brewing value of hops is mainly affected by the content and composition of humulones (α-acids) and essential oil. Interest in hop plantations is increasing more and more in Italy, in parallel with the rising number of microbreweries and brewpubs, which are strongly oriented towards local production chains. In this context, a selection of 15 international hop varieties were grown, under the same conditions, in an experimental field in the Marche region, Central Italy, with the aim of assessing their suitability for beer production. A multivariate analysis approach to experimental data showed a high content of α- and β-acids and myrcene in the Centennial, Brewer’s Gold, Sterling, Cascade, Nugget, and Columbus varieties; a consistently lower percentages of humulones and a predominance of sesquiterpene hydrocarbons in the cultivars Mount Hood, Northern Brewer, Northdown, Galena, Willamette, and Fuggle; and a desirable high α-acids content and a sesquiterpene-type aroma in cultivars Chinook, Yeoman, and Hallertau. Further studies are needed to assess the environmental adaptability and the yield performance of hop plants in the pedoclimatic conditions of the Central Italy hills.
Keywords: hop variety, Humulus lupulus, bitter substances, humulones, volatile components, SPME
1. Introduction
The unfertilized female inflorescence of hop plant (Humulus lupulus L.) (hops or cones), used in the brewing process in various forms (e.g., whole cones, pellets, and extracts), changes the wort characteristics and provides bitterness, aroma, astringency, and fullness to the finished product. Moreover, hops act as a technological aid (clarifier), contribute to the microbiological stability of beer, and enhance the foam-building ability of beer and foam stability [1]. The young shoots that emerge from the plant in April and May are also used for food preparations, both fresh (salads, risotto, and omelets) and preserved. The vitamin E content of shoots [2] could be higher than other well-known vegetable sources of tocopherols [3] and has been recently highlighted as a nutritional peculiarity of this culinary delicacy.
The brewing value of hops is primarily attributed to a complex mixture of secondary metabolites constituting the yellow resinous powder secreted by the lupulin glands of cones [4]. The chemical composition of hops is strongly affected by variety, ripening stage, environmental factors, agronomic practices, and cultivation area [5]. It is well established that humulones (α-acids) are the most important precursors of bitter substances in finished beer, even if several bitter tasting products of β-acid transformation are generated during wort boiling [1,6]. In fact, beer makers classify hop varieties on the basis of α-acid content into bitter (>7%) and finishing/aromatic ones (<7%) [7]. The hop essential oil is also important to the brewer, as it contributes to the characteristic “hoppy” flavor of the beer, even if the wort boiling and the fermentation process result in the loss or transformation of a large portion of the original hops aroma. However, it is not fully understood how the hop variety, the amount and form (dried cones, pellets, and extracts) of hops, and the hopping method (early, late, or dry) could impact the volatile fraction of beers [8]. The essential oil is primarily made up of terpene hydrocarbons and their oxidized derivatives; the oil content of dry cones ranges from 0.5% to 3% [9]. The volatile profile has been recently proposed for the cultivar characterization [9,10,11].
Wild hop plants normally grow in the whole Italian peninsula. However, winemaking has relegated brewing to second place for a long time and hop plantations in Italy have mostly had an amateur character. Currently, new regulations concerning the production of hops and the management of craft breweries management are increasing the interest of farmers and entrepreneurs in these activities. The increasing number of microbreweries and brewpubs [12] suggests new development opportunities for the beer market. These new entities are increasingly interested in producing beers within a local production chain, from ingredients (hops, malt) to the final product. Italy is still far from meeting the estimated need for hops at 3500 t/year. A recent research project, funded by the Italian Ministry of Agricultural, Food and Forestry Policies and coordinated by the “Consiglio per la ricerca in agricoltura e l’analisi dell’economia agraria” (CREA) [13], showed that the surface invested in hop plantation is just over 50 ha and that the business plantations (size greater than 1000 m2) are mostly concentrated in the Emilia Romagna and Lazio regions. The same study highlighted that Apennine areas of Central Italy are very suitable for cultivating hops for brewing. The excellent potential that the sector could express for the revitalization of agriculture has also been highlighted, especially in the internal marginal areas of the country. However, very little data are currently available on the chemical characteristics of “Italian” hops [14], thus, leaving to empiricism the development of distinctive hop characters in beers by craft brewers.
In this context, a selection of 15 international hop varieties were grown, under the same conditions, in an experimental field in the Marche region of Central Italy. The volatile fingerprint and the α- and β-acid content were compared to the standard characteristics of the varieties, with the aim of assessing the suitability of hops cultivated in Central Italy for beer production.
2. Materials and Methods
2.1. Plant Material
Fifteen international hop cultivars, from USA, UK, and Germany (3 bittering, 4 aroma, and 8 dual purpose) were used for the experiment (Table 1). The trial was carried out at the experimental field of “La Contea” farm (Tavullia, Pesaro Urbino, Italy), located in the Montelabbate (Pesaro Urbino, Italy) municipality (43°48′ N, 12°45′ E, altitude 260 m a.s.l.). Five to seven rhizomes per cultivar were planted, in the spring of 2015, in the typical silt clay soil of the Marche region hills. Hop plants were grown on a standard trellis system 8 m high. The plants were spaced 2.8 m between rows and 1 m apart in the rows. Weed growth was periodically checked, and weeds were manually removed. Pests and pathogens were controlled by preventive spraying of copper products before flowering. A drip irrigation system provided water to the plants whenever necessary.
Table 1.
List of hop varieties used for the experiment, their brewing use, maturity timelines, origin, and chemical characters [15].
| Sample ID | Name | Brewing Use | Seasonal Maturity 1 | Origin | α-Acids% 2 | β-Acids% 2 | Cohumulone% | Myrcene% | Humulene% | Caryophyllene% |
|---|---|---|---|---|---|---|---|---|---|---|
| HAL | Hallertau | Aroma | E to M | Germany | 3.0–3.5 | 3.5–4.5 | 20–26 | 35–44 | 30–55 | 10–15 |
| WIL | Willamette | Aroma | E to M | USA | 4–6 | 3–4 | 30–35 | 30–55 | 20–30 | 7–8 |
| YEO | Yeoman | Dual purpose | E | UK | 12–16 | 4–5 | 25 | 48 | 20 | 10 |
| CEN | Centennial | Dual purpose | M | USA | 9.5–11.5 | 3.5–4.5 | 28–30 | 45–55 | 10–18 | 5–8 |
| FUG | Fuggle | Aroma | E to M | UK | 2.4–6.1 | 2.1–2.8 | 25–29 | 43.4 | 26.6 | 9.1 |
| MHO | Mount Hood | Aroma | E to M | USA | 4–8 | 5–8 | 21–23 | 30–40 | 12–38 | 7–16 |
| NBR | Northern Brewer | Dual purpose | E to M | Germany | 7–10 | 3.5–5 | 27–33 | 25–45 | 35–50 | 10–20 |
| GAL | Galena | Bittering | M | USA | 12 | 7.5 | 39 | 55–60 | 10–15 | 3–6 |
| BRG | Brewer’s Gold | Bittering | L | UK | 7.1–11.3 | 3.3–6.1 | 41 | 66.7 | 11.6 | 6.5 |
| STE | Sterling | Dual purpose | M | USA | 4.5–9 | 4–6 | 21–28 | 44–48 | 19–23 | 5–8 |
| CAS | Cascade | Dual purpose | M | USA | 4.5–8.9 | 3.6–7.5 | 33–40 | 45–60 | 8–16 | 4–6 |
| NUG | Nugget | Bittering | M | USA | 9.5–14 | 4.2–5.8 | 22–30 | 48–59 | 12–22 | 7–10 |
| COL | Columbus | Dual purpose | M to L | USA | 14–18 | 4.5–6 | 28–35 | 25–55 | 9–25 | 6–12 |
| NOR | Northdown | Dual purpose | M | UK | 7–10 | 4–5.5 | 24–32 | 23–29 | 37–45 | 13–17 |
| CHI | Chinook | Dual purpose | M to L | USA | 12–14 | 3–4 | 29–34 | 35–40 | 18–25 | 9–11 |
1 M, medium; L, late; E, early. 2 On dry matter basis.
Cones from the 2018 production year were harvested separately for each variety, freeze-dried and stored at −20 °C, until the analysis. Three samples of each cultivar were used for chemical analyses.
2.2. Determination of α- and β-Acids
The extraction of bitter tasting precursors was carried out according to Stevens et al. [16]. Briefly, 0.5 g of ground dried cones were thoroughly mixed with methanol into a 10 mL volumetric flask. In order to complete the extraction process, the mixture was allowed to stay in the dark for 4 h. The supernatant was filtered using a 0.22 µm PTFE membrane (Thermo Scientific, Waltham, MA, USA) and directly injected in an Agilent (Santa Clara, CA, USA) 1100 Series HPLC system, equipped with a Zorbax Eclipse Plus C18 column (3.0 × 150 mm, 3.5 μm particle size) and a UV detector set at 330 nm. A gradient elution was performed using the following two-solvent system [17]: solvent A = 75% methanol, 24% water, and 1% phosphoric acid; solvent B = methanol and solvent B was increased from 30% to 55% in 10 min. The flow rate was 0.8 mL/min. All solvents were purchased at Sigma-Aldrich (St. Louis, MO, USA).
The International Calibration Extract 4 (ICE-4) (Labor Veritas AG, Zürich, Switzerland) was used to quantify the α- and β-acids. A set of three standard methanolic solutions of ICE-4 (0.4, 1.0, and 2.0 mg/mL) were used to make the calibration curves.
2.3. GC-MS Analysis of Volatile Components
Volatiles sampling was carried out by headspace solid phase microextraction (HS-SPME), according to the operative parameters described by Savini et al. [18]. Volatiles were analyzed by gas chromatography-mass spectrometry in a Varian 3900 GC coupled with a Saturn 2100T ion trap mass detector (Varian Analytical Instruments, Walnut Creek, CA, USA) and equipped with a fused silica capillary column ZB-5 (30 m length, 0.25 mm internal diameter, 0.25 μm film thickness; Phenomenex, Torrance, CA, USA). The injector was operating in splitless mode for 0.1 min at a constant temperature of 250 °C; oven temperature was increased from 40 °C to 220 °C at a rate of 6 °C/min, then held at the final temperature for 5 min; carrier gas (He) was set at a constant flow mode (1.0 mL/min); the ion trap and the transfer line were set at 200 °C and 220 °C, respectively. Experiments of both electronic impact fragmentation (EI, 70 eV) and chemical ionization (CI) (reagent gas, methanol) were carried out. Full scan MS data were acquired in the mass range of 31–250 amu.
Volatile compounds were identified by matching mass spectral data with those collected in the NIST/EPA/NIH Mass Spectral Library (Version 2.0a, built 1 July 2002; National Institute of Standards and Technology), and Kovats retention Indices (RIs) with those available in the public access database Pubchem [19]. A C8–C20 normal alkanes mixture (Sigma-Aldrich, St. Louis, MO, USA) was used to calculate RIs in the experimental conditions. An automated spreadsheet [20] was used to simplify the calculation of RIs of the unknown components and speeding up the comparison with the published indices. The CI spectral data (parent and base peaks) were used to confirm the molecular weight of volatile substances.
2.4. Data Analysis
Multivariate data analyses were carried out to explore the structure of the experimental data (principal component analysis, PCA) and to classify varieties (cluster analysis). A Pearson’s correlation analysis was also carried out to assess the relationships among experimental variables. The Tukey–Kramer’s honest significant difference (HSD) test was used to compare the experimental variables among varieties. All statistical analyses were carried out by the software JMP® Version 10 (SAS Institute Inc., Cary, NC, USA).
3. Results and Discussion
3.1. Precursors of Bitter Tasting Compounds of Beers
Table 2 reports the acid contents of hop cones harvested from the 15 varieties. It is well established that most of the perceived pleasant bitterness in beer is provided by isomerization derivatives of α-acids and that isohumulones are more bitter than isocohumulones [1].
Table 2.
Content of α- and β-acids (g/100 g dry matter, mean ± SD) of hop cones harvested from 15 varieties cultivated in the Marche region (Central Italy).
| Sample ID | CoH | AdH + Hum | CoL | AdL + Lup | Total α-Acids | Total β-Acids |
|---|---|---|---|---|---|---|
| HAL | 0.34 ± 0.05 g | 1.24 ± 0.20 efg | 0.76 ± 0.13 efg | 1.23 ± 0.20 cdef | 1.58 ± 0.24 ef | 1.99 ± 0.33 de |
| WIL | 0.31 ± 0.03 g | 0.34 ± 0.00 g | 1.25 ± 0.01 def | 1.09 ± 0.01 def | 0.65 ± 0.03 f | 2.34 ± 0.02 de |
| YEO | 1.86 ± 0.00 cd | 5.94 ± 0.03 b | 1.34 ± 0.01 cde | 1.66 ± 0.00 bcd | 7.81 ± 0.03 b | 3.00 ± 0.01 bcd |
| CEN | 0.72 ± 0.08 fg | 1.80 ± 0.22d ef | 1.08 ± 0.14 defg | 1.30 ± 0.18 cdef | 2.51 ± 0.29 de | 2.37 ± 0.32 de |
| FUG | 0.40 ± 0.03 g | 0.83 ± 0.10f g | 0.46 ± 0.06 fg | 0.42 ± 0.07 f | 1.23 ± 0.13 ef | 0.88 ± 0.13 e |
| MHO | 0.77 ± 0.04 fg | 2.51 ± 0.11 cd | 1.77 ± 0.08 bcd | 2.49 ± 0.09 b | 3.28 ± 0.15 cd | 4.26 ± 0.17 bc |
| NBR | 1.08 ± 0.13 ef | 2.36 ± 0.30 cde | 1.21 ± 0.12 defg | 1.14 ± 0.10 def | 3.44 ± 0.42 cd | 2.35 ± 0.22 de |
| GAL | 0.29 ± 0.11 g | 0.70 ± 0.26 fg | 0.41 ± 0.17 g | 0.53 ± 0.21 ef | 0.99 ± 0.37 ef | 0.95 ± 0.38 e |
| BRG | 0.78 ± 0.06 fg | 1.35 ± 0.11d efg | 1.06 ± 0.10 defg | 0.74 ± 0.07 def | 2.13 ± 0.17 def | 1.79 ± 0.17 de |
| STE | 0.79 ± 0.09 fg | 1.45 ± 0.02d efg | 1.36 ± 0.10 cde | 1.68 ± 0.11 bcd | 2.24 ± 0.11 def | 3.04 ± 0.21 bcd |
| CAS | 1.37 ± 0.24 de | 3.10 ± 0.65 c | 3.14 ± 0.61 a | 4.25 ± 0.81 a | 4.47 ± 0.89 c | 7.39 ± 1.42 a |
| NUG | 2.90 ± 0.05 a | 7.71 ± 0.15 a | 2.23 ± 0.06 b | 2.15 ± 0.06 bc | 10.61 ± 0.20 a | 4.38 ± 0.11 b |
| COL | 2.49 ± 0.35 ab | 4.92 ± 0.70 b | 2.10 ± 0.33 bc | 1.44 ± 0.23 cde | 7.41 ± 1.05 b | 3.54 ± 0.57 bcd |
| NOR | 0.63 ± 0.08 fg | 1.52 ± 0.19 defg | 0.87 ± 0.13 efg | 0.99 ± 0.14 def | 2.15 ± 0.28 def | 1.86 ± 0.26 de |
| CHI | 2.14 ± 0.10 bc | 5.84 ± 0.28 b | 1.29 ± 0.06 de | 1.28 ± 0.06 cdef | 7.98 ± 0.37 b | 2.56 ± 0.11 cde |
CoH, cohumulone; AdH, adhumulone; Hum, humulone; CoL, colupulone; AdL, adlupulone; Lup, lupulone. Values in a column with different letters are significantly different (Tukey test, p < 0.05).
The highest levels of α-acids were observed in Nugget (NUG) (10.61% ± 0.20% DM), Chinook (CHI) (7.98% ± 0.37% DM), and Yeoman (YEO) (7.81% ± 0.03% DM), while Cascade (CAS), NUG, and Mount Hood (MHO) had the highest contents of lupulones (7.39% ± 1.42%, 4.38% ± 0.11%, and 4.26% ± 0.17% DM, respectively). However, the amount of bitter precursors of the cultivars analyzed was generally lower than the typical value of the corresponding commercial varieties (Table 1), with the exception of CAS and NUG. As reported above, a low contribution of cohumulone to the α-acid fraction is also desirable. The cohumulone percentage (of total α-acids) of most of the analyzed samples lay in the range of the reference values in Table 1, while Willamette (WIL) (47.7%), Fuggle (FUG) (32.5%), and Sterling (STE) (35.3%) were characterized by a more unfavorable composition of the α-acid fraction. Cohumulone percentages lower than commercial cultivars were observed in Galena (GAL) (29.3%), Brewer’s Gold (BRG) (36.6%), CAS (30.6%), and CHI (26.8%).
Mongelli et al. [14] reported higher percentages of α- and β-acids for the hop varieties Columbus (COL) (19.6%, 7.2%), FUG (4.5%, 6.5%), and WIL (2.9%, 2.1%) cultivated in Northern Italy, thus, confirming the strong influence of pedoclimatic conditions on hops chemical traits. Pearson et al. [21] reported similar results for COL and CHI varieties cultivated in an open-sided greenhouse in Central Florida, which were characterized by an α-acid content of 4.8% to 6.8% and 9.7% to 10.4% and a β-acid content of 2.7% to 2.8% and 2.1% to 2.5%, respectively. Lafontaine et al. [6] observed in CAS cones collected from 5 to 6 weeks over the years 2014 to 2016 a rough maintenance of the humulones (4.40% to 5.79%) and lupulones (5.81% to 8.50%) concentration throughout the overall harvest period. Conversely, Drexler et al. [22] showed the importance of the harvest time for Hallertauer Mittelfrüh hops; a significant increase of the lupulones concentration from the first to the fourth week or inflorescence development was observed.
3.2. Aroma Components of Cones
A total of 57 volatile substances (19 sesquiterpene hydrocarbons, 15 monoterpene hydrocarbons, 13 esters, four ketones, three oxygenated sesquiterpenes, two n-alkanes, one oxygenated monoterpene) were positively or tentatively identified, on the basis of the criteria described in Section 2.3. (Table 3, Supplementary Figures S1 and S2). Hydrocarbons accounted for 90% to 98% of the total volatile components. Sesquiterpenes (hydrocarbons and oxygenated analogues) were the most abundant chemical class in Hallertau (HAL), WIL, YEO, FUG, MHO, Northern Brewer (NBR), GAL, and Northdown (NOR), while monoterpene hydrocarbons were the most represented aroma components in Centennial (CEN), BRG, STE, CAS, NUG, COL, and CHI.
Table 3.
Volatile components identified in the headspace of dried hop cones harvested from 15 varieties cultivated in the Marche region (Central Italy).
| Peak ID | RT (min) | RI | CI Parent and Base Ions | Name | CAS Number | Category |
|---|---|---|---|---|---|---|
| V1 | 6.616 | 919 | 145 [M + 1] | isobutyl isobutyrate | 97-85-8 | Ester |
| V2 | 6.773 | 926 | 137 [M + 1] | tricyclene | 508-32-7 | Monoterpene hydrocarbon |
| V3 | 6.947 | 934 | 137 [M + 1] | α-thujene | 2867-05-2 | Monoterpene hydrocarbon |
| V4 | 7.119 | 942 | 137 [M + 1] | α-pinene | 80-56-8 | Monoterpene hydrocarbon |
| V5 | 7.472 | 957 | 137 [M + 1] | camphene | 79-92-5 | Monoterpene hydrocarbon |
| V6 | 8.147 | 983 | 137 [M + 1] | β-pinene | 127-91-3 | Monoterpene hydrocarbon |
| V7 | 8.356 | 991 | 127 [M + 1]. 109 [M + 1 − H2O] | 6-methyl-5-heptene-2-one | 110-93-0 | Ketone |
| V8 | 8.510 | 996 | 137 [M + 1] | β-myrcene | 123-35-3 | Monoterpene hydrocarbon |
| V9 | 8.810 | 1009 | 137 [M + 1] | α-phellandrene | 99-83-2 | Monoterpene hydrocarbon |
| V10 | 8.957 | 1015 | 158 [M] | 2-methylbutyl isobutyrate | 2445-69-4 | Ester |
| V11 | 9.044 | 1019 | 158 [M] | isoamyl isobutyrate | 2050-01-3 | Ester |
| V12 | 9.105 | 1022 | 137 [M + 1] | α-terpinene | 99-86-5 | Monoterpene hydrocarbon |
| V13 | 9.251 | 1028 | 145 [M + 1] | methyl heptanoate | 106-73-0 | Ester |
| V14 | 9.332 | 1032 | 143 [M + 1] | heptenoic acid isomer. methyl ester | Ester | |
| V15 | 9.413 | 1035 | 137 [M + 1] | β-phellandrene | 555-10-2 | Monoterpene hydrocarbon |
| V16 | 9.594 | 1043 | 137 [M + 1] | (Z)-β-ocimene | 3338-55-4 | Monoterpene hydrocarbon |
| V17 | 9.859 | 1054 | 137 [M + 1] | (E)-β-ocimene | 3779-61-1 | Monoterpene hydrocarbon |
| V18 | 10.147 | 1065 | 137 [M + 1] | y-terpinene | 99-85-4 | Monoterpene hydrocarbon |
| V19 | 10.203 | 1067 | 159 [M + 1] | heptanoic acid. 2-methyl. methyl ester | 51209-78-0 | Ester |
| V20 | 10.808 | 1090 | 159 [M + 1] | heptanoic acid. 6-methyl. methyl ester | 2519-37-1 | Ester |
| V21 | 10.883 | 1092 | 137 [M + 1] | terpinolene | 586-62-9 | Monoterpene hydrocarbon |
| V22 | 10.931 | 1094 | 143 [M + 1] | 2-nonanone | 821-55-6 | Ketone |
| V23 | 11.152 | 1102 | 137 [M + 1 − H2O] | linalool | 78-70-6 | Oxygenated monoterpene |
| V24 | 11.726 | 1128 | 159 [M + 1] | methyl octanoate | 111-11-5 | Ester |
| V25 | 11.862 | 1134 | 137 [M + 1] | alloocimene | 7216-56-0 | Monoterpene hydrocarbon |
| V26 | 12.173 | 1147 | 137 [M + 1] | Unidentified | Monoterpene hydrocarbon | |
| V27 | 13.390 | 1195 | 157 [M + 1] | 2-decanone | 693-54-9 | Ketone |
| V28 | 13.549 | 1201 | 157 [M + 1 − CH2] | n-dodecane | 112-40-3 | n-alkane |
| V29 | 13.846 | 1215 | 171 [M + 1]. 139 [M + 1 − CH3OH] | methyl x-nonenoate | Ester | |
| V30 | 14.129 | 1227 | 173 [M + 1] | methyl nonanoate | 1731-84-6 | Ester |
| V31 | 14.638 | 1250 | 187 [M + 1] | heptyl isobutanoate | 2349-13-5 | Ester |
| V32 | 15.749 | 1295 | 171 [M + 1] | 2-undecanone | 112-12-9 | Ketone |
| V33 | 15.874 | 1300 | 171 [M + 1 − CH2] | n-tridecane | 629-50-5 | n-alkane |
| V34 | 16.092 | 1311 | 185 [M + 1]. 153 [M + 1 − CH3OH] | methyl 4-decenoate | 1191-02-2 | Ester |
| V35 | 16.443 | 1328 | 187 [M + 1] | methyl decanoate | 110-42-9 | Ester |
| V36 | 17.100 | 1358 | 205 [M + 1] | α-cubebene | 17699-14-8 | Sesquiterpene hydrocarbon |
| V37 | 17.208 | 1362 | 205 [M + 1] | sativene | 3650-28-0 | Sesquiterpene hydrocarbon |
| V38 | 17.610 | 1380 | 205 [M + 1] | ylangene | 14912-44-8 | Sesquiterpene hydrocarbon |
| V39 | 17.711 | 1384 | 205 [M + 1] | α-copaene | 3856-25-5 | Sesquiterpene hydrocarbon |
| V40 | 18.717 | 1432 | 205 [M + 1] | b-caryophillene | 87-44-5 | Sesquiterpene hydrocarbon |
| V41 | 18.879 | 1440 | 205 [M + 1] | β-cubebene | 13744-15-5 | Sesquiterpene hydrocarbon |
| V42 | 19.463 | 1468 | 205 [M + 1] | humulene | 6753-98-6 | Sesquiterpene hydrocarbon |
| V43 | 19.787 | 1483 | 205 [M + 1] | b-copaene | 18612-33-4 | Sesquiterpene hydrocarbon |
| V44 | 19.844 | 1486 | 205 [M + 1] | (E)-b-Famesene | 18794-84-8 | Sesquiterpene hydrocarbon |
| V45 | 19.916 | 1489 | 205 [M + 1] | α-Farnesene | 502-61-4 | Sesquiterpene hydrocarbon |
| V46 | 20.095 | 1497 | 205 [M + 1] | b-selinene | 17066-67-0 | Sesquiterpene hydrocarbon |
| V47 | 20.212 | 1503 | 205 [M + 1] | α-muurolene | 31983-22-9 | Sesquiterpene hydrocarbon |
| V48 | 20.276 | 1506 | 205 [M + 1] | α-selinene | 473-13-2 | Sesquiterpene hydrocarbon |
| V49 | 20.626 | 1524 | 205 [M + 1] | γ-muurolene | 30021-74-0 | Sesquiterpene hydrocarbon |
| V50 | 20.784 | 1532 | 205 [M + 1] | α-cadinene | 11044-40-9 | Sesquiterpene hydrocarbon |
| V51 | 20.844 | 1535 | 205 [M + 1] | b-cadinene | 523-47-7 | Sesquiterpene hydrocarbon |
| V52 | 20.986 | 1543 | 205 [M + 1] | cadina-1.4-diene | 16728-99-7 | Sesquiterpene hydrocarbon |
| V53 | 21.090 | 1548 | 205 [M + 1] | (E)-calamenene | 40772-39-2 | Sesquiterpene hydrocarbon |
| V54 | 21.210 | 1554 | 205 [M + 1] | α-calacorene | 21391-99-1 | Sesquiterpene hydrocarbon |
| V55 | 22.057 | 1595 | 203 [M + 1 − H2O] | caryophillene oxyde | 1139-30-6 | Oxygenated sesquiterpene |
| V56 | 22.356 | 203 [M + 1 − H2O] | Unidentified | Oxygenated sesquiterpene | ||
| V57 | 22.567 | 203 [M + 1 − H2O] | Unidentified | Oxygenated sesquiterpene |
RT, retention time; RI, Kovats retention index (DB-5 equivalent column); CI, chemical ionization experiment.
Myrcene, humulene, and caryophyllene were the major sesquiterpenoids in all samples. Myrcene is mainly responsible for the characteristic pungent smell of fresh hops, while humulene and caryophyllene are recognized to be precursors of oxygenated aroma-active substances during wort boiling, thus, providing the spicy/herbal hop character to beer [11]. Significant differences were observed between the aroma composition of the experimental samples and the commercial varieties (Table 4). The relative levels of caryophyllene in the experimental hops WIL, YEO, CEN, FUG, MHO, GAL, BRG, and STE where higher than those found in the commercial cultivars. Higher relative percentages of humulene were also observed in the experimental YEO, FUG, GAL, and BRG, together with a lower level of myrcene. In addition, CAS, NUG, COL, NOR, and CHI hops from the Marche region plantation were richer in myrcene than commercial plants. Mongelli et al. [14] reported comparable values for humulene and caryophyllene percentages of COL, FUG, and WIL cultivated in Northern Italy, while the myrcene contents of FUG and WIL were very low (0.91% and 0.83%, respectively).
Table 4.
Aroma composition (chromatographic area %, mean ± SD) of hop cones harvested from 15 varieties cultivated in the Marche region (Central Italy).
| Peak ID | HAL | WIL | YEO | CEN | FUG | MHO | NBR | GAL | BRG | STE | CAS | NUG | COL | NOR | CHI |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| V1 | 0.03 ± 0.00 b | 0.03 ± 0.00 b | 0.28 ± 0.18 b | 0.08 ± 0.06 b | 0.05 ± 0.04 b | 0.05 ± 0.02 b | 0.09 ± 0.03 b | 0.08 ± 0.08 b | 0.35 ± 0.10 b | 0.04 ± 0.00 b | 0.11 ± 0.04 b | 0.44 ± 0.09 a | 0.90 ± 0.42 a | 0.07 ± 0.03 b | 0.30 ± 0.02 b |
| V2 | 0.01 ± 0.00 | 0.00 ± 0.00 | 0.00 ± 0.00 | 0.01 ± 0.01 | 0.02 ± 0.01 | 0.01 ± 0.00 | 0.01 ± 0.00 | 0.00 ± 0.00 | 0.01 ± 0.00 | 0.00 ± 0.00 | 0.01 ± 0.01 | 0.00 ± 0.00 | 0.07 ± 0.09 | 0.01 ± 0.00 | 0.01 ± 0.00 |
| V3 | 0.02 ± 0.00 ab | 0.00 ± 0.00 b | 0.00 ± 0.00 b | 0.04 ± 0.03 ab | 0.14 ± 0.12 a | 0.01 ± 0.00 ab | 0.01 ± 0.00 ab | 0.00 ± 0.00 b | 0.02 ± 0.01 ab | 0.02 ± 0.00 ab | 0.03 ± 0.01 ab | 0.04 ± 0.02 ab | 0.05 ± 0.02 ab | 0.01 ± 0.00 b | 0.01 ± 0.00 ab |
| V4 | 0.08 ± 0.03 | 0.07 ± 0.03 | 0.15 ± 0.06 | 0.35 ± 0.35 | 0.09 ± 0.06 | 0.12 ± 0.06 | 0.18 ± 0.03 | 0.06 ± 0.04 | 0.22 ± 0.04 | 0.24 ± 0.07 | 0.25 ± 0.14 | 0.24 ± 0.04 | 0.31 ± 0.10 | 0.19 ± 0.12 | 0.22 ± 0.00 |
| V5 | 0.00 ± 0.00 c | 0.00 ± 0.00 bc | 0.02 ± 0.01 abc | 0.02 ± 0.02 abc | 0.01 ± 0.00 bc | 0.00 ± 0.00 c | 0.01 ± 0.00 bc | 0.01 ± 0.00 bc | 0.02 ± 0.01 abc | 0.01 ± 0.00 abc | 0.04 ± 0.03 abc | 0.06 ± 0.01 a | 0.06 ± 0.02 ab | 0.02 ± 0.01 abc | 0.02 ± 0.01 abc |
| V6 | 0.85 ± 0.21 | 0.44 ± 0.17 | 0.57 ± 0.24 | 2.49 ± 2.19 | 0.50 ± 0.35 | 1.03 ± 0.42 | 0.92 ± 0.17 | 0.49 ± 0.25 | 1.81 ± 0.20 | 1.67 ± 0.18 | 1.93 ± 0.86 | 2.00 ± 0.33 | 2.26 ± 0.74 | 1.01 ± 0.48 | 1.60 ± 0.11 |
| V7 | 0.01 ± 0.01 ab | 0.02 ± 0.01 ab | 0.11 ± 0.09 ab | 0.00 ± 0.00 b | 0.06 ± 0.03 ab | 0.01 ± 0.00 ab | 0.05 ± 0.03 ab | 0.04 ± 0.00 ab | 0.01 ± 0.01 ab | 0.02 ± 0.00 ab | 0.01 ± 0.01 ab | 0.04 ± 0.01 ab | 0.12 ± 0.02 a | 0.04 ± 0.01 ab | 0.00 ± 0.00 b |
| V8 | 41.44 ± 1.94 bcd | 14.14 ± 0.04 e | 14.95 ± 6.07 e | 49.45 ± 4.05 abc | 14.08 ± 2.47 e | 40.82 ± 7.23 bcd | 34.01 ± 2.05 cd | 26.33 ± 3.53 b | 54.05 ± 5.40 | 53.69 ± 5.85 ab | 67.10 ± 5.66 a | 64.45 ± 1.34 a | 45.20 ± 4.84 bc | 33.72 ± 4.43 cd | 45.35 ± 5.24 bc |
| V9 | 0.01 ± 0.00 b | 0.01 ± 0.00 b | 0.06 ± 0.05 ab | 0.01 ± 0.01 b | 0.05 ± 0.04 b | 0.02 ± 0.01 b | 0.03 ± 0.00 b | 0.03 ± 0.02 b | 0.04 ± 0.01 b | 0.02 ± 0.00 b | 0.01 ± 0.01 b | 0.03 ± 0.00 b | 0.12 ± 0.01 a | 0.04 ± 0.01 b | 0.04 ± 0.00 b |
| V10 | 0.01 ± 0.00 | 0.01 ± 0.01 | 0.37 ± 0.22 | 0.05 ± 0.04 | 0.07 ± 0.05 | 0.02 ± 0.01 | 0.10 ± 0.03 | 0.12 ± 0.12 | 0.04 ± 0.03 | 0.01 ± 0.00 | 0.08 ± 0.08 | 0.21 ± 0.26 | 0.10 ± 0.12 | 0.09 ± 0.04 | 0.33 ± 0.02 |
| V11 | 0.03 ± 0.03 d | 0.15 ± 0.04 d | 1.16 ± 0.62 bcd | 0.28 ± 0.12 cd | 0.18 ± 0.18 d | 0.11 ± 0.03 d | 0.28 ± 0.11 cd | 0.60 ± 0.84 bcd | 2.16 ± 0.52 bc | 0.03 ± 0.03 d | 0.20 ± 0.14 d | 1.85 ± 0.01 bcd | 4.32 ± 1.41 a | 0.30 ± 0.08 cd | 2.36 ± 0.07 b |
| V12 | 0.01 ± 0.00 | 0.01 ± 0.01 | 0.23 ± 0.26 | 0.04 ± 0.04 | 0.47 ± 0.50 | 0.02 ± 0.02 | 0.04 ± 0.01 | 0.01 ± 0.01 | 0.06 ± 0.00 | 0.03 ± 0.00 | 0.20 ± 0.23 | 0.02 ± 0.01 | 0.06 ± 0.01 | 0.03 ± 0.01 | 0.06 ± 0.01 |
| V13 | 0.14 ± 0.05 bc | 0.01 ± 0.01 c | 0.13 ± 0.06 bc | 0.12 ± 0.05 bc | 0.07 ± 0.04 bc | 0.23 ± 0.07 abc | 0.23 ± 0.19 abc | 0.13 ± 0.14 bc | 0.35 ± 0.09 ab | 0.13 ± 0.00 bc | 0.17 ± 0.02 bc | 0.50 ± 0.06 a | 0.23 ± 0.05 abc | 0.09 ± 0.05 bc | 0.27 ± 0.06 abc |
| V14 | 0.62 ± 0.07 | 0.07 ± 0.00 | 0.06 ± 0.03 | 0.20 ± 0.12 | 0.15 ± 0.05 | 0.20 ± 0.05 | 0.29 ± 0.22 | 0.16 ± 0.12 | 0.06 ± 0.07 | 0.16 ± 0.02 | 0.11 ± 0.03 | 0.25 ± 0.02 | 0.44 ± 0.46 | 0.19 ± 0.05 | 0.11 ± 0.02 |
| V15 | 0.68 ± 0.05 ab | 0.30 ± 0.02 b | 0.44 ± 0.18 ab | 1.46 ± 0.79 a | 0.41 ± 0.18 ab | 0.73 ± 0.26 ab | 0.87 ± 0.16 ab | 0.42 ± 0.10 ab | 1.22 ± 0.32 ab | 0.99 ± 0.01 ab | 1.08 ± 0.03 ab | 0.95 ± 0.02 ab | 1.16 ± 0.39 ab | 0.60 ± 0.19 bc | 0.92 ± 0.04 ab |
| V16 | 0.02 ± 0.00 b | 0.02 ± 0.00 b | 0.05 ± 0.03 b | 0.05 ± 0.05 b | 0.03 ± 0.00 b | 0.02 ± 0.01 b | 0.13 ± 0.02 a | 0.01 ± 0.01 b | 0.04 ± 0.01 b | 0.04 ± 0.01 b | 0.03 ± 0.01 b | 0.04 ± 0.00 b | 0.01 ± 0.01 b | 0.05 ± 0.01 b | 0.02 ± 0.00 b |
| V17 | 0.03 ± 0.00 b | 0.13 ± 0.05 b | 0.09 ± 0.06 b | 0.14 ± 0.10 b | 0.09 ± 0.03 b | 0.06 ± 0.02 b | 1.39 ± 0.21 a | 0.10 ± 0.08 b | 1.17 ± 0.41 a | 0.07 ± 0.01 b | 0.23 ± 0.02 b | 1.01 ± 0.09 a | 1.09 ± 0.27 a | 0.26 ± 0.01 b | 0.06 ± 0.01 b |
| V18 | 0.02 ± 0.01 | 0.02 ± 0.00 | 0.02 ± 0.00 | 0.06 ± 0.02 | 0.04 ± 0.04 | 0.03 ± 0.01 | 0.14 ± 0.16 | 0.01 ± 0.00 | 0.04 ± 0.01 | 0.02 ± 0.00 | 0.04 ± 0.00 | 0.04 ± 0.01 | 0.10 ± 0.01 | 0.02 ± 0.01 | 0.04 ± 0.01 |
| V19 | 0.15 ± 0.20 | 0.12 ± 0.04 | 0.01 ± 0.01 | 0.02 ± 0.01 | 0.11 ± 0.06 | 0.01 ± 0.01 | 0.08 ± 0.07 | 0.04 ± 0.00 | 0.03 ± 0.02 | 0.11 ± 0.01 | 0.01 ± 0.01 | 0.02 ± 0.01 | 0.03 ± 0.02 | 0.10 ± 0.03 | 0.01 ± 0.00 |
| V20 | 0.19 ± 0.09 b | 0.04 ± 0.02 b | 0.03 ± 0.03 b | 0.12 ± 0.05 b | 0.06 ± 0.01 b | 0.10 ± 0.02 b | 0.09 ± 0.05 b | 0.25 ± 0.31 b | 0.15 ± 0.21 b | 0.04 ± 0.00 b | 0.16 ± 0.00 b | 0.35 ± 0.04 b | 0.45 ± 0.20 ab | 0.14 ± 0.04 b | 0.87 ± 0.03 a |
| V21 | 0.18 ± 0.18 | 0.02 ± 0.01 | 0.04 ± 0.03 | 0.07 ± 0.03 | 0.06 ± 0.06 | 0.03 ± 0.01 | 0.05 ± 0.01 | 0.03 ± 0.02 | 0.16 ± 0.12 | 0.04 ± 0.01 | 0.06 ± 0.00 | 0.05 ± 0.00 | 0.07 ± 0.05 | 0.05 ± 0.00 | 0.00 ± 0.00 |
| V22 | 0.28 ± 0.19 | 0.01 ± 0.00 | 0.34 ± 0.02 | 0.18 ± 0.06 | 0.21 ± 0.08 | 0.30 ± 0.09 | 0.17 ± 0.04 | 0.22 ± 0.17 | 0.10 ± 0.02 | 0.15 ± 0.01 | 0.16 ± 0.01 | 0.12 ± 0.03 | 0.31 ± 0.18 | 0.15 ± 0.07 | 0.02 ± 0.01 |
| V23 | 0.94 ± 0.12 a | 0.21 ± 0.10 d | 0.17 ± 0.07 d | 0.50 ± 0.20 abcd | 0.40 ± 0.07 bcd | 0.33 ± 0.33 cd | 0.40 ± 0.02 bcd | 0.42 ± 0.07 bcd | 0.45 ± 0.02 abcd | 0.83 ± 0.14 ab | 0.35 ± 0.08 bcd | 0.72 ± 0.06 abc | 0.47 ± 0.04 abcd | 0.63 ± 0.11 abcd | 0.30 ± 0.01 cd |
| V24 | 0.18 ± 0.14 bcde | 0.02 ± 0.01 e | 0.06 ± 0.03 de | 0.12 ± 0.05 cde | 0.03 ± 0.02 e | 0.05 ± 0.00 de | 0.06 ± 0.04 de | 0.10 ± 0.08 cde | 0.34 ± 0.01 abcd | 0.02 ± 0.00 e | 0.14 ± 0.01 cde | 0.48 ± 0.02 ab | 0.50 ± 0.02 a | 0.08 ± 0.03 cde | 0.37 ± 0.02 abc |
| V25 | 0.01 ± 0.00 c | 0.00 ± 0.00 c | 0.01 ± 0.01 bc | 0.04 ± 0.01 abc | 0.05 ± 0.03 a | 0.01 ± 0.00 bc | 0.05 ± 0.00 ab | 0.01 ± 0.01 bc | 0.02 ± 0.00 abc | 0.01 ± 0.01 bc | 0.05 ± 0.01 ab | 0.02 ± 0.01 abc | 0.01 ± 0.00 bc | 0.02 ± 0.01 bc | 0.01 ± 0.00 bc |
| V26 | 0.02 ± 0.01 bc | 0.01 ± 0.01 c | 0.01 ± 0.01 bc | 0.04 ± 0.02 bc | 0.04 ± 0.03 bc | 0.01 ± 0.00 bc | 0.12 ± 0.02 a | 0.01 ± 0.01 bc | 0.03 ± 0.01 bc | 0.02 ± 0.01 bc | 0.03 ± 0.00 bc | 0.04 ± 0.02 bc | 0.06 ± 0.01 b | 0.05 ± 0.00 bc | 0.01 ± 0.01 c |
| V27 | 0.35 ± 0.08 ab | 0.07 ± 0.05 b | 0.28 ± 0.01 ab | 0.07 ± 0.05 b | 0.42 ± 0.13 | 0.25 ± 0.06 ab | 0.36 ± 0.05 ab | 0.61 ± 0.33 a | 0.07 ± 0.00 b | 0.29 ± 0.01 ab | 0.06 ± 0.01 b | 0.09 ± 0.01 b | 0.07 ± 0.10 ab | 0.26 ± 0.03 ab | 0.57 ± 0.05 a |
| V28 | 0.03 ± 0.03 ab | 0.00 ± 0.00 b | 0.01 ± 0.01 ab | 0.01 ± 0.01 ab | 0.06 ± 0.01 ab | 0.01 ± 0.01 ab | 0.04 ± 0.03 ab | 0.08 ± 0.01 a | 0.00 ± 0.00 ab | 0.01 ± 0.01 ab | 0.02 ± 0.02 ab | 0.00 ± 0.00 b | 0.04 ± 0.05 ab | 0.04 ± 0.02 ab | 0.00 ± 0.01 ab |
| V29 | 0.06 ± 0.02 ab | 0.01 ± 0.00 b | 0.04 ± 0.03 ab | 0.03 ± 0.03 ab | 0.06 ± 0.01 ab | 0.03 ± 0.00 ab | 0.07 ± 0.04 ab | 0.14 ± 0.14 ab | 0.04 ± 0.02 ab | 0.08 ± 0.01 ab | 0.08 ± 0.01 ab | 0.19 ± 0.04 a | 0.06 ± 0.00 ab | 0.07 ± 0.06 ab | 0.08 ± 0.02 ab |
| V30 | 0.07 ± 0.03 bcd | 0.02 ± 0.01 d | 0.02 ± 0.01 d | 0.03 ± 0.03 d | 0.06 ± 0.00 bcd | 0.05 ± 0.01 bcd | 0.07 ± 0.04 bcd | 0.18 ± 0.22 abcd | 0.24 ± 0.02 abcd | 0.03 ± 0.00 cd | 0.11 ± 0.01 bcd | 0.38 ± 0.02 a | 0.29 ± 0.06 ab | 0.10 ± 0.07 bcd | 0.29 ± 0.03 abc |
| V31 | 0.01 ± 0.01 bc | 0.01 ± 0.00 c | 0.05 ± 0.02 bc | 0.01 ± 0.00 bc | 0.11 ± 0.08 ab | 0.02 ± 0.01 bc | 0.08 ± 0.03 abc | 0.05 ± 0.02 bc | 0.04 ± 0.01 bc | 0.01 ± 0.00 bc | 0.02 ± 0.02 bc | 0.10 ± 0.01 abc | 0.17 ± 0.01 a | 0.10 ± 0.03 abc | 0.03 ± 0.01 bc |
| V32 | 0.81 ± 0.01 ab | 0.05 ± 0.05 b | 0.44 ± 0.04 ab | 0.15 ± 0.07 b | 0.35 ± 0.08 b | 0.32 ± 0.20 b | 0.37 ± 0.03 b | 1.22 ± 0.73 a | 0.01 ± 0.001 b | 0.44 ± 0.01 ab | 0.09 ± 0.05 b | 0.11 ± 0.02 b | 0.11 ± 0.05 b | 0.62 ± 0.28 ab | 0.12 ± 0.02 b |
| V33 | 0.01 ± 0.00 | 0.01 ± 0.01 | 0.04 ± 0.05 | 0.04 ± 0.00 | 0.12 ± 0.03 | 0.03 ± 0.03 | 0.20 ± 0.00 | 0.14 ± 0.16 | 0.00 ± 0.00 | 0.01 ± 0.00 | 0.01 ± 0.01 | 0.06 ± 0.07 | 0.20 ± 0.27 | 0.07 ± 0.05 | 0.00 ± 0.00 |
| V34 | 0.64 ± 0.39 | 0.07 ± 0.05 | 0.16 ± 0.03 | 0.35 ± 0.41 | 0.18 ± 0.03 | 0.21 ± 0.10 | 0.20 ± 0.04 | 0.55 ± 0.68 | 0.76 ± 0.07 | 0.19 ± 0.04 | 0.31 ± 0.06 | 1.00 ± 0.07 | 0.95 ± 0.18 | 0.54 ± 0.03 | 0.88 ± 0.09 |
| V35 | 0.13 ± 0.07 | 0.09 ± 0.00 | 0.10 ± 0.06 | 0.96 ± 0.72 | 0.18 ± 0.09 | 0.11 ± 0.00 | 0.90 ± 1.00 | 0.31 ± 0.19 | 1.22 ± 0.30 | 0.06 ± 0.04 | 0.45 ± 0.15 | 0.28 ± 0.04 | 0.81 ± 0.08 | 0.15 ± 0.02 | 0.76 ± 0.04 |
| V36 | 0.18 ± 0.02 cd | 0.12 ± 0.03 def | 0.03 ± 0.00 f | 0.17 ± 0.02 cd | 0.07 ± 0.01 def | 0.28 ± 0.02 bc | 0.04 ± 0.00 ef | 0.15 ± 0.02 de | 0.12 ± 0.05 def | 0.08 ± 0.00 def | 0.10 ± 0.01 def | 0.11 ± 0.00 def | 0.31 ± 0.08 | 0.04 ± 0.01 ef | 0.44 ± 0.04 a |
| V37 | 0.01 ± 0.00 e | 0.05 ± 0.00 bc | 0.08 ± 0.01 a | 0.03 ± 0.01 cde | 0.04 ± 0.00 bcd | 0.04 ± 0.01 bcde | 0.04 ± 0.00 bcde | 0.03 ± 0.01 cde | 0.02 ± 0.00 e | 0.02 ± 0.00 de | 0.02 ± 0.00 de | 0.02 ± 0.00 e | 0.05 ± 0.01 ab | 0.04 ± 0.00 bcd | 0.03 ± 0.00 bcde |
| V38 | 0.36 ± 0.06 bcd | 0.47 ± 0.03 ab | 0.57 ± 0.05 a | 0.18 ± 0.01 fg | 0.46 ± 0.03 ab | 0.27 ± 0.05 cdef | 0.42 ± 0.00 abc | 0.24 ± 0.06 defg | 0.15 ± 0.05 fg | 0.20 ± 0.04 efg | 0.12 ± 0.01 fg | 0.10 ± 0.01 g | 0.25 ± 0.01 defg | 0.39 ± 0.06 bcd | 0.35 ± 0.04 bcde |
| V39 | 0.87 ± 0.02 efg | 1.50 ± 0.0 ab | 1.71 ± 0.14 a | 0.73 ± 0.03 fgh | 1.43 ± 0.02 ab | 0.94 ± 0.09 cdef | 1.36 ± 0.01 abc | 0.86 ± 0.16 efg | 0.51 ± 0.13 gh | 0.63 ± 0.12 fgh | 0.69 ± 0.12 fgh | 0.36 ± 0.07 | 0.90 ± 0.09 defg | 1.21 ± 0.13 bcde | 1.28 ± 0.14 bcd |
| V40 | 12.08 ± 0.22 efg | 25.54 ± 2.35 a | 20.84 ± 2.10 abc | 12.71 ± 1.78 efg | 23.14 ± 1.34 ab | 18.17 ± 0.38 bcd | 19.57 ± 0.05 bcd | 16.66 ± 0.24 cde | 10.44 ± 1.54 fg | 12.20 ± 1.78 efg | 7.44 ± 1.24 g | 7.64 ± 0.13 g | 14.20 ± 1.58 def | 18.94 ± 1.40 bcd | 9.75 ± 0.98 fg |
| V41 | 0.79 ± 0.10 bcd | 1.21 ± 0.02 b | 0.72 ± 0.05 bcd | 0.75 ± 0.15 bcd | 1.02 ± 0.02 bc | 1.00 ± 0.13 bc | 0.80 ± 0.10 bcd | 1.00 ± 0.23 bc | 0.63 ± 0.20 cd | 0.54 ± 0.07 cd | 0.44 ± 0.07 d | 0.49 ± 0.11 g | 0.87 ± 0.08 bcd | 0.77 ± 0.13 bcd | 1.91 ± 0.29 a |
| V42 | 21.72 ± 0.66 defg | 46.10 ± 0.16 a | 33.84 ± 2.81 bc | 20.85 ± 5.05 defg | 45.79 ± 2.37 a | 25.63 ± 3.83 cdef | 28.79 ± 2.07 bcde | 38.53 ± 1.28 ab | 17.81 ± 3.80 efg | 22.35 ± 3.13 defg | 12.01 ± 3.45 g | 11.58 ± 1.07 g | 15.72 ± 1.17 fg | 29.11 ± 4.09 bcd | 17.21 ± 1.71 fg |
| V43 | 0.35 ± 0.13 ab | 0.10 ± 0.13 abc | 0.16 ± 0.08 abc | 0.02 ± 0.00 c | 0.17 ± 0.02 abc | 0.24 ± 0.07 abc | 0.21 ± 0.04 abc | 0.11 ± 0.12 abc | 0.17 ± 0.04 abc | 0.08 ± 0.11 bc | 0.01 ± 0.01 c | 0.09 ± 0.00 abc | 0.26 ± 0.00 abc | 0.23 ± 0.05 abc | 0.38 ± 0.08 a |
| V44 | 1.55 ± 0.19 bcd | 1.60 ± 0.33 bcd | 2.92 ± 0.56 a | 1.11 ± 0.26 bcd | 1.32 ± 0.14 bcd | 1.69 ± 0.66 bc | 1.13 ± 0.23 bcd | 1.34 ± 0.18 bcd | 0.77 ± 0.10 cd | 0.75 ± 0.01 cd | 0.82 ± 0.13 cd | 0.47 ± 0.06 d | 0.99 ± 0.14 bcd | 1.39 ± 0.11 bcd | 2.03 ± 0.38 ab |
| V45 | 0.06 ± 0.03 bc | 0.21 ± 0.02 ab | 0.25 ± 0.07 a | 0.09 ± 0.00 abc | 0.13 ± 0.04 abc | 0.07 ± 0.09 bc | 0.16 ± 0.02 abc | 0.12 ± 0.08 abc | 0.08 ± 0.02 bc | 0.10 ± 0.02 abc | 0.07 ± 0.00 bc | 0.04 ± 0.00 c | 0.15 ± 0.01 abc | 0.18 ± 0.03 abc | 0.22 ± 0.04 ab |
| V46 | 3.18 ± 0.51 b | 0.85 ± 0.26 c | 5.69 ± 0.58 a | 1.17 ± 0.50 c | 1.00 ± 0.18 c | 0.56 ± 0.18 c | 0.47 ± 0.24 c | 0.93 ± 0.32 c | 0.51 ± 0.03 c | 0.36 ± 0.02 c | 1.05 ± 0.30 c | 0.78 ± 0.09 c | 0.76 ± 0.03 c | 1.29 ± 0.31 c | 1.09 ± 0.20 c |
| V47 | 0.00 ± 0.00 c | 0.31 ± 0.05 ab | 0.16 ± 0.18 abc | 0.21 ± 0.06 abc | 0.31 ± 0.03 ab | 0.38 ± 0.19 ab | 0.34 ± 0.01 ab | 0.35 ± 0.02 ab | 0.14 ± 0.01 bc | 0.17 ± 0.04 abc | 0.11 ± 0.08 bc | 0.10 ± 0.00 bc | 0.27 ± 0.04 abc | 0.25 ± 0.05 abc | 0.46 ± 0.33 a |
| V48 | 5.19 ± 1.18 b | 1.57 ± 0.36 c | 8.41 ± 1.02 a | 1.69 ± 0.68 c | 1.53 ± 0.18 c | 1.05 ± 0.02 c | 1.31 ± 0.39 c | 1.76 ± 0.66 c | 1.06 ± 0.03 c | 0.74 ± 0.05 c | 1.55 ± 0.41 c | 0.98 ± 0.21 c | 1.38 ± 0.00 c | 2.56 ± 0.55 c | 2.28 ± 0.37 c |
| V49 | 0.61 ± 0.02 bc | 1.07 ± 0.28 abc | 0.96 ± 0.16 abc | 0.72 ± 0.26 abc | 1.04 ± 0.13 abc | 1.20 ± 0.63 abc | 0.85 ± 0.23 abc | 1.28 ± 0.25 ab | 0.59 ± 0.06 bc | 0.61 ± 0.05 bc | 0.46 ± 0.15 bc | 0.31 ± 0.06 c | 0.78 ± 0.04 abc | 0.91 ± 0.16 abc | 1.55 ± 0.26 a |
| V50 | 1.81 ± 0.25 ab | 2.09 ± 0.60 ab | 2.24 ± 0.32 ab | 1.36 ± 0.49 ab | 2.15 ± 0.50 ab | 2.39 ± 1.41 ab | 1.62 ± 0.41 ab | 2.13 ± 0.41 ab | 1.10 ± 0.07 ab | 1.06 ± 0.15 ab | 0.89 ±0.36 b | 0.56 ± 0.07 b | 1.50 ± 0.02 ab | 1.81 ± 0.30 ab | 3.03 ± 0.39 a |
| V51 | 0.04 ± 0.00 c | 0.26 ± 0.08 abc | 0.38 ± 0.03 a | 0.16 ± 0.00 abc | 0.18 ± 0.16 abc | 0.09 ± 0.09 bc | 0.31 ± 0.02 ab | 0.17 ± 0.01 abc | 0.14 ± 0.00 abc | 0.16 ± 0.03 abc | 0.10 ± 0.00 bc | 0.05 ± 0.02 c | 0.05 ± 0.07 c | 0.31 ± 0.01 ab | 0.11 ± 0.11 bc |
| V52 | 0.07 ± 0.01 b | 0.14 ± 0.04 ab | 0.16 ± 0.03 ab | 0.09 ± 0.04 ab | 0.13 ± 0.02 ab | 0.17 ± 0.09 ab | 0.13 ± 0.04 ab | 0.13 ± 0.03 ab | 0.09 ± 0.00 ab | 0.06 ± 0.00 b | 0.05 ± 0.03 b | 0.04 ± 0.00 b | 0.10 ± 0.01 ab | 0.13 ± 0.02 ab | 0.19 ± 0.01 a |
| V53 | 1.45 ± 0.38 a | 0.20 ± 0.10 c | 0.19 ± 0.03 c | 0.12 ± 0.06 c | 0.17 ± 0.03 c | 0.20 ± 0.13 c | 0.14 ± 0.04 c | 0.24 ± 0.09 c | 0.11 ± 0.01 c | 0.09 ± 0.01 c | 0.08 ± 0.03 c | 0.06 ± 0.01 c | 0.15 ± 0.01 c | 0.16 ± 0.02 c | 0.90 ± 0.13 b |
| V54 | 1.30 ± 0.37 a | 0.08 ± 0.05 c | 0.09 ± 0.02 c | 0.03 ± 0.02 c | 0.06 ± 0.02 c | 0.06 ± 0.04 c | 0.05 ± 0.02 c | 0.09 ± 0.05 c | 0.03 ± 0.00 c | 0.03 ± 0.00 c | 0.03 ± 0.01 c | 0.01 ± 0.00 c | 0.01 ± 0.01 c | 0.07 ± 0.01 c | 0.66 ± 0.12 b |
| V55 | 0.10 ± 0.03 | 0.09 ± 0.07 | 0.03 ± 0.00 | 0.05 ± 0.02 | 0.49 ± 0.61 | 0.06 ± 0.06 | 0.05 ± 0.03 | 0.10 ± 0.12 | 0.04 ± 0.02 | 0.04 ± 0.02 | 0.06 ± 0.04 | 0.01 ± 0.00 | 0.04 ± 0.01 | 0.07 ± 0.03 | 0.03 ± 0.00 |
| V56 | 0.03 ± 0.00 | 0.05 ± 0.03 | 0.01 ± 0.00 | 0.02 ± 0.01 | 0.05 ± 0.00 | 0.02 ± 0.02 | 0.02 ± 0.02 | 0.55 ± 0.76 | 0.02 ± 0.01 | 0.02 ± 0.00 | 0.03 ± 0.02 | 0.00 ± 0.00 | 0.01 ± 0.00 | 0.04 ± 0.02 | 0.01 ± 0.00 |
| V57 | 0.20 ± 0.02 | 0.19 ± 0.18 | 0.04 ± 0.01 | 0.14 ± 0.10 | 0.24 ± 0.00 | 0.16 ± 0.16 | 0.12 ± 0.08 | 0.31 ± 0.42 | 0.11 ± 0.06 | 0.15 ± 0.01 | 0.15 ± 0.10 | 0.02 ± 0.01 | 0.04 ± 0.01 | 0.20 ± 0.08 | 0.04 ± 0.00 |
Values in a row with different letters are significantly different (Tukey test, p < 0.05).
Other terpenic hydrocarbons were distinctive of some hop cultivars. WIL was previously reported to be a high-farnesene hop cultivar [23]; however, YEO and CHI cultivated in the Marche hills showed higher percentages of farnesene isomers. A group of experimental cultivars of American origin (CEN, STE, CAS, NUG, and COL), together with BRG (UK), showed the highest β-pinene percentages (1.67% to 2.26%). The interest concerning this monoterpene lies in its role in the initiation of autoxidation of α-acids [24]. Amounts of β-phellandrene higher than 1% were only found in CEN, BRG, CAS, and COL. (E)-β-ocimene contents were much lower than 1% except in NBR, BRG, NUG, and COL (1.01% to 1.39%). (E)-calamenene and α-calacorene characterized the aroma of HAL (1.45% and 1.30%, respectively) and CHI (0.90% and 0.66%, respectively), while selinene isomers characterized YEO volatile fraction (7.93%). The percentages of α- and β-selinene observed in COL, FUG, and WIL were similar to those reported by Mongelli et al. [14] for the same varieties cultivated in Northern Italy.
Esters and ketones were relatively minor groups, ranging from 0.66% (WIL) to 9.24% (COL) and from 0.15% (WIL) to 2.09% (GAL) of the total volatiles, respectively. Esters are known for their fruity notes [11]. According to Yan et al. [10], the most abundant ketone was 2-undecanone (ranging from 0.01% in BRG to 1.22% in GAL), while isoamyl isobutyrate was the most abundant ester. COL was the richest cultivar (4.32%); in YEO, BRG, NUG, and CHI varieties the isoamyl isobutyrate contents were in the range 1.16% to 2.36%; in all the other varieties the quantitative contribution of this ketone to aroma was much lower than 1%.
Among the hop oil components, linalool is widely accepted as being flavor-active in beer and it is associated with floral impressions in both hops and beer [25]. In fact, terpenic alcohols (linalool and geraniol) were detected as predominant compounds in the pitching wort and in late and dry hopped beers, followed by monoterpene (β-myrcene) and sesquiterpene hydrocarbons (humulene and β-caryophyllene). Differences in polarity, volatility, and solubility were used to explain the more selective retention of alcohols than hydrocarbons [8]. The linalool content of the experimental cultivars in this study ranged from 0.17% (YEO) to 0.94% (HAL).
3.3. Multivariate Analysis
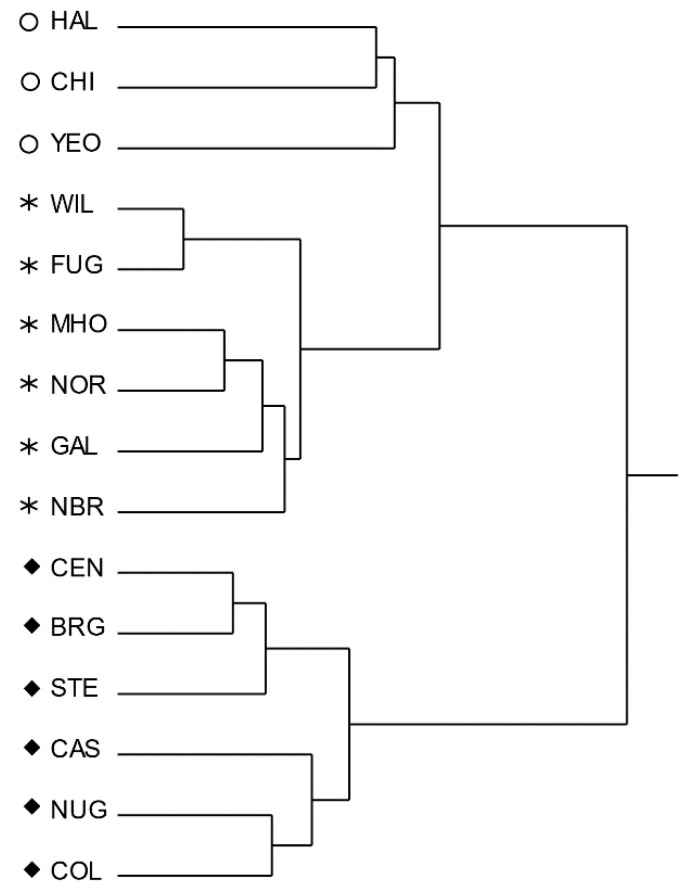
Hierarchical clustering split samples into three groups, on the basis of their volatile profiles (18 variables with at least one value greater than 1%) and the vinylogous type of organic acids (four variables) (Figure 1).
Figure 1.
Dendrogram of the hierarchical cluster analysis among commercial varieties of H. lupulus L. cultivated in the Marche region, as determined by chemical similarity (Ward, distance scale).
The Group A cultivars (CEN, BRG, STE, CAS, NUG, COL) had both the highest levels of humulones (2.13% to 10.61%) and lupulones (1.79% to 7.39%) and the highest ratio between mono- (myrcene) and sesquiterpene (humulene + caryophyllene) hydrocarbons. The Group B varieties (WIL, FUG, MHO, NOR, GAL, and NBR) had the lowest contents of α-acids (0.65% to 3.44%) and their aroma showed consistently higher percentages of the sesquiterpenes humulene (25.63% to 46.10%) and caryophyllene (18.17% to 25.54%). Group C (HAL, CHI, YEO) showed intermediate characteristics. The grouping did not fully reflect the general characteristics of the commercial varieties in Table 1, according to their brewing use, maturity timelines, origin, and chemical parameters. However, most of aroma hops (WIL, FUG, and MHO) belonged to Group B and two out of three bittering hops belonged to Group A. The dual purpose hops were mainly included in Group A as well. Rossini et al. [26] reported good yield performance for the cultivars CAS and YEO under the climatic conditions of Central Italy, and a more complex appreciated profile for beers flavored with local cones than those hopped with commercial products. Our experimental data also showed interesting properties for those two varieties, i.e., a content of α-acid comparable to commercial variety, a favorable composition of the humulones fraction for CAS, and a high level of α-acids and sesquiterpene hydrocarbons for YEO.
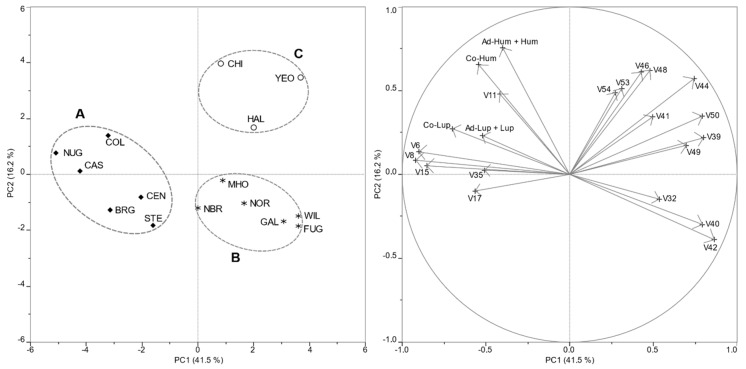
The PCA reflected these relationships (Figure 2). The first two principal components explained 57.7% of the total variance. PC1 was mainly affected by a cluster of monoterpene hydrocarbons (myrcene, β-pinene, and β-phellandrene) with negative loadings, and a cluster of sesquiterpene hydrocarbons (humulene, caryophyllene, copaene, muurolene, farnesene, and selinene) with positive loadings. Sesquiterpene hydrocarbons (humulene, caryophyllene) had negative loadings on PC2, whereas humulones only affected PC2. Positive linear correlations were found between β-pinene and β-phellandrene (r = 0.9471) and between the pairs of sesquiterpenes caryophyllene vs. humulene (r = 0.9149), β-selinene vs. α-selinene (r = 0.9916), γ-muurolene vs. α-cadinene (r = 0.9421, and (E)-calamenene vs. α-calacorene (r = 0.0.9945). Humulones were also positively correlated (r = 0.9619), as well as lupulones (r = 0.9022), while no correlations (variables positioned in approximately orthogonal directions between them) were found between the aroma components and the precursors of bitter tasting compounds. The strongest inverse correlations (variables positioned in opposite direction respect to the axes origin and far from the plot origin) were found between myrcene and the sesquiterpenes caryophyllene (r = −0.9276) and humulene (r = −0.9318).
Figure 2.
(Left) PCA scores plot of chemical data of hop cones from 15 commercial varieties of H. lupulus L. cultivated in the Marche region. Cultivar identifiers are as in Table 1; (Right) PCA loadings plot of variables (volatiles and acids). Identifiers of chemical substances are as in Table 1 and Table 2.
PC1 was able to differentiate the cultivars that fell into Group A (highest myrcene percentages and lowest humulene and caryophyllene relative amounts) from the others. Lower contents of humulones and higher relative amounts of sesquiterpenes (humulene and caryophyllene) drove the differentiation between cultivars that fell into Groups B and C toward the lower right and the upper right quadrant, respectively.
4. Conclusions
The hop plant can adapt to different environments and pedoclimatic conditions, because of its rustic habit, high degree of intraspecific genetic variability, and easiness of cultivation. However, difficulties in plantation management (high trellis systems, long periods of time before plants are ready for data collection, manual harvesting of cones) have hindered systematic studies on environmental adaptability and yield performance of hop cultivars in different countries.
The present study aimed to identify, among a selection of 15 commercial hop varieties of different origin and traditional use, those potentially suited to the Central Italy hilly environment in terms of brewing quality. Multivariate analysis of experimental data concerning the volatiles and acid profiles identified a group of cultivars (CEN, BRG, STE, CAS, NUG, and COL) characterized by high contents of α- and β-acids and a prevalence of monoterpenes (namely myrcene) in their aroma. Precursors of the spicy/herbal hop character of beer (the sesquiterpenes humulene and caryophyllene) predominated in all the other varieties. Particularly, the cultivars MHO, NBR, NOR, GAL, WIL, and FUG showed consistently lower percentages of humulones, while the varieties CHI, YEO, and HAL had a desirable high α-acids content and a sesquiterpene-type aroma. CHI and YEO appeared to be the most promising varieties, due to the highest level of α-acids (CHI and YEO), the most favorable composition of the humulone fraction (CHI), and the highest percentage of volatile precursors of the hoppy character of beer (YEO).
The comparison, with very little available data concerning chemical and agronomical traits of hop varieties cultivated in different Italian regions, highlighted the strong influence of pedoclimatic conditions on the overall brewing quality of cones, and thus limited the applicability of experimental data to different environments. These results suggest the need to further investigate the environmental adaptability and the agronomic performance of hop varieties in the pedoclimatic conditions of the hills of Central Italy. Experimental data should be collected for more years, as most of the Italian hop plantations are relatively young, and therefore still not qualitatively stable. Additionally, the extreme variability of plantation management and post-harvesting processing (drying and storing) of cones add difficulty to data comparison.
Acknowledgments
Authors would like to thank Sofia Paccapelo (La Contea” farm, Tavullia, Pesaro Urbino, Italy) for supplying the hop samples and for the technical support, as coordinator of the project “Highops”.
Supplementary Materials
The following are available online at https://www.mdpi.com/2304-8158/9/5/541/s1, Figure S1: Headspace SPME-GC-MS profile (TIC, total ion current) of the of the Northern Brewer cultivar, Figure S2: Comparison among the headspace SPME-GC-MS profiles (TIC, total ion current) of fifteen hop varieties cultivated in the Marche region, Italy.
Author Contributions
Conceptualization, M.M., R.F., and C.M.; methodology, M.M.; validation, R.F. and C.M.; formal analysis, M.M., R.F., and C.M.; investigation, R.F. and C.M.; resources, M.M.; data curation, R.F. and C.M.; writing—original draft preparation, M.M., R.F., and C.M.; writing—review and editing, M.M., R.F., and C.M.; visualization, M.M., R.F., and C.M.; supervision, M.M.; project administration, M.M.; funding acquisition, M.M. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by REGIONE MARCHE (Programma di Sviluppo Rurale 2014/2020–M16.1.A.2–Project “Highops”), grant number 434.
Conflicts of Interest
The authors declare no conflict of interest.
References
- 1.Almaguer C., Schönberger C., Gastl M., Arendt E.K., Becker T. Humulus lupulus—A story that begs to be told. J. Inst. Brew. 2014;120:289–314. doi: 10.1002/jib.160. [DOI] [Google Scholar]
- 2.Morales P., Carvalho A.M., Sánchez-Mata M.C., Cámara M., Molina M., Ferreira I.C.F.R. Tocopherol composition and antioxidant activity of Spanish wild vegetables. Genet. Resour. Crop Evol. 2012;59:851–863. doi: 10.1007/s10722-011-9726-1. [DOI] [Google Scholar]
- 3.Mozzon M., Pacetti D., Frega N.G., Lucci P. Crude palm oil from interspecific hybrid Elaeis oleifera × E. guineensis: Alcoholic constituents of unsaponifiable matter. JAOCS. 2015;92:717–724. doi: 10.1007/s11746-015-2628-1. [DOI] [Google Scholar]
- 4.Van Cleemput M., Cattoor K., De Bosscher K., Haegeman G., De Keukeleire D., Heyerick A. Hop (Humulus lupulus)-derived bitter acids as multipotent bioactive compounds. J. Nat. Prod. 2009;72:1220–1230. doi: 10.1021/np800740m. [DOI] [PubMed] [Google Scholar]
- 5.Inui T., Okumura K., Matsui H., Hosoya T., Kumazawa S. Effect of harvest time on some in vitro functional properties of hop polyphenols. Food Chem. 2017;225:69–76. doi: 10.1016/j.foodchem.2017.01.002. [DOI] [PubMed] [Google Scholar]
- 6.Lafontaine S., Varnum S., Roland A., Delpech S., Dagan L., Vollmer D., Kishimoto T., Shellhammer T. Impact of harvest maturity on the aroma characteristics and chemistry of Cascade hops used for dry-hopping. Food Chem. 2019;278:228–239. doi: 10.1016/j.foodchem.2018.10.148. [DOI] [PubMed] [Google Scholar]
- 7.Kao T.H., Wu G.Y. Simultaneous determination of prenylflavonoid and hop bitter acid in beer lee by HPLC-DAD-MS. Food Chem. 2013;14:1218–1226. doi: 10.1016/j.foodchem.2013.04.032. [DOI] [PubMed] [Google Scholar]
- 8.Dresel M., Praet T., Van Opstaele F., Van Holle A., Naudts D., De Keukeleire D., De Cooman L., Aerts G. Comparison of the analytical profiles of volatiles in single-hopped worts and beers as a function of the hop variety. Brew. Sci. 2015;68:8–28. [Google Scholar]
- 9.Kovačevič M., Kač M. Determination and verification of hop varieties by analysis of essential oils. Food Chem. 2002;77:489–494. doi: 10.1016/S0308-8146(02)00114-0. [DOI] [Google Scholar]
- 10.Yan D.D., Wong Y.F., Shellie R.A., Marriott P.J., Whittock S.P., Koutoulis A. Assessment of the phytochemical profiles of novel hop (Humulus lupulus L.) cultivars: A potential route to beer crafting. Food Chem. 2019;275:15–23. doi: 10.1016/j.foodchem.2018.09.082. [DOI] [PubMed] [Google Scholar]
- 11.Van Opstaele F., De Causmaecker B., Aerts G., De Cooman L. Characterization of novel varietal floral hop aromas by headspace solid phase microextraction and gas chromatography-mass spectrometry/olfactometry. J. Agric. Food Chem. 2012;60:12270–12281. doi: 10.1021/jf304421d. [DOI] [PubMed] [Google Scholar]
- 12.Assobirra [(accessed on 21 February 2020)];2018 Available online: https://www.assobirra.it/
- 13.LUPPOLO.IT. [(accessed on 21 February 2020)];2016 Available online: http://luppolo.crea.gov.it/
- 14.Mongelli A., Rodolfi M., Ganino T., Marieschi M., Dall’Asta C., Bruni R. Italian hop germplasm: Characterization of wild Humulus lupulus L. genotypes from Northern Italy by means of phytochemical, morphological traits and multivariate data analysis. Ind. Crop. Prod. 2015;70:16–27. doi: 10.1016/j.indcrop.2015.02.036. [DOI] [Google Scholar]
- 15.Hopslist [(accessed on 14 January 2020)];2018 Available online: http://www.hopslist.com/hops/
- 16.Stevens J.F., Taylor A.W., Deinzer M.L. Quantitative analysis of xanthohumol and related prenylflavonoids in hops and beer by liquid chromatography—Tandem mass spectrometry. J. Chromatogr. A. 1999;832:97–107. doi: 10.1016/S0021-9673(98)01001-2. [DOI] [PubMed] [Google Scholar]
- 17.Labor Veritas A.G. A User’s Guide to the New International Calibration Standards for HPLC Analysis of Isomerized & Reduced Isomerized α-Acids. [(accessed on 10 January 2020)];2018 Available online: https://laborveritas.ch/de/produkte/
- 18.Savini S., Loizzo M.R., Tundis R., Mozzon M., Foligni R., Longo E., Morozova K., Scampicchio M., Martin-Vertedor D., Boselli E. Fresh refrigerated Tuber melanosporum truffle: Effect of the storage conditions on the antioxidant profile, antioxidant activity and volatile profile. Eur. Food Res. Technol. 2017;243:2255–2263. doi: 10.1007/s00217-017-2927-x. [DOI] [Google Scholar]
- 19.PubChem. [(accessed on 5 March 2020)]; Available online: https://pubchem.ncbi.nlm.nih.gov/
- 20.Lucero M., Estell R., Tellez M., Fredrickson E. A retention index calculator simplifies identification of plant volatile organic compounds. Phytochem. Anal. 2009;20:378–384. doi: 10.1002/pca.1137. [DOI] [PubMed] [Google Scholar]
- 21.Pearson B.J., Smith R.M., Chen J. Growth, strobile yield, and quality of four Humulus lupulus varieties cultivated in a protected open-sided greenhouse structure. HortScience. 2016;51:838–842. doi: 10.21273/HORTSCI.51.7.838. [DOI] [Google Scholar]
- 22.Drexler G., Bailey B., Schönberger C., Gahr A., Newmann R., Pöschl M., Eberhard G. The influence of hop harvest date on flavor stability in dry-hopped beers. Tech. Q. Master Brew. Assoc. Am. 2010;47:1–4. doi: 10.1094/TQ-47-1-0219-01. [DOI] [Google Scholar]
- 23.Eri S., Khoo B.K., Lech J., Hartman T.G. Direct thermal desorption–gas chromatography and gas chromatography–mass spectrometry profiling of hop (Humulus lupulus L.) essential oils in support of varietal characterization. J. Agric. Food Chem. 2000;48:1140–1149. doi: 10.1021/jf9911850. [DOI] [PubMed] [Google Scholar]
- 24.Menary R.C., Williams E.A., Nickerson G.B. Effect of myrtenol on the rate of oxidation of alpha- and beta-acids in hops. Acta Hortic. 1986;188:149–156. doi: 10.17660/ActaHortic.1986.188.17. [DOI] [Google Scholar]
- 25.Peacock V.E. The value of linalool in modeling hop aroma in beer. Tech. Q. Master Brew. Assoc. Am. 2010;47:29–32. doi: 10.1094/TQ-47-4-1102-01. [DOI] [Google Scholar]
- 26.Rossini F., Loreti P., Provenzano M.E., De Santis D., Ruggeri R. Agronomic performance and beer quality assessment of twenty hop cultivars grown in Central Italy. Ital. J. Agron. 2016;11:180–187. doi: 10.4081/ija.2016.746. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.