Abstract
During the development of antimicrobial peptides (AMP) as potential therapeutics, antimicrobial susceptibility testing (AST) stands as an essential part of the process in identification and optimisation of candidate AMP. Standard methods for AST, developed almost 60 years ago for testing conventional antibiotics, are not necessarily fit for purpose when it comes to determining the susceptibility of microorganisms to AMP. Without careful consideration of the parameters comprising AST there is a risk of failing to identify novel antimicrobials at a time when antimicrobial resistance (AMR) is leading the planet toward a post-antibiotic era. More physiologically/clinically relevant AST will allow better determination of the preclinical activity of drug candidates and allow the identification of lead compounds. An important consideration is the efficacy of AMP in biological matrices replicating sites of infection, e.g., blood/plasma/serum, lung bronchiolar lavage fluid/sputum, urine, biofilms, etc., as this will likely be more predictive of clinical efficacy. Additionally, specific AST for different target microorganisms may help to better predict efficacy of AMP in specific infections. In this manuscript, we describe what we believe are the key considerations for AST of AMP and hope that this information can better guide the preclinical development of AMP toward becoming a new generation of urgently needed antimicrobials.
Keywords: antimicrobial peptide (AMP), antimicrobial susceptibility testing, antibiotic, antifungal, host defence peptide (HDP)
Antimicrobial Resistance
Antimicrobial resistance (AMR) is a global health crisis. The over-use and inappropriate prescribing of antibiotics has set us on a path toward a post-antibiotic era where our existing armory of antibiotics will no longer be effective. Increasing numbers of microorganisms are already becoming widely resistant to existing antibiotic classes (O'Neill, 2016; Barlow, 2018; Chatterjee et al., 2018; Van Puyvelde et al., 2018; Roope et al., 2019). The need for new antimicrobial agents is more important now than it has ever been. So too is the need for better antimicrobial stewardship; prudent and appropriate use of antimicrobials.
AMR is responsible for 700,000 deaths per annum globally (Blair et al., 2015; European Commission, 2017) but this is forecast to increase to 10 million deaths annually by 2050 (killing more people than cancer and diabetes) if the measures highlighted above are not urgently implemented successfully to address drug resistant infections (O'Neill, 2016). This assessment was based on scenarios for rising drug resistance and economic growth to 2050 for six major pathogens/infectious diseases; Klebsiella pneumoniae, Escherichia coli, Staphylococcus aureus, malaria, tuberculosis and HIV (O'Neill, 2014). According to the Centers for Disease Control and Prevention (CDC), in the US alone over 2.8 million infections per year are caused by antibiotic-resistant bacteria, causing more than 35,000 deaths in the US per annum and US$55 billion in increased healthcare costs and lost productivity (CDC, 2019). Strikingly, in 2009–2010 in the US almost 20% of pathogens reported from Hospital-acquired infections (HAI) were multidrug-resistant (Sievert et al., 2013).
Most of the antibiotics available today are broad-spectrum molecules derived from agents that have been in use for more than 30 years and as well as “failing” through resistance, oftentimes have unintended side effects, such as toxicity toward beneficial commensal bacteria and mammalian cells, and triggering inflammatory responses (Lepore et al., 2019). Research efforts are ongoing to discover and develop new, more effective and safe antimicrobial agents that can overcome bacterial resistance mechanisms, occasionally even presenting selective activity toward single bacterial species or specific strains of bacteria (de la Fuente-Nunez et al., 2017b).
Antimicrobial Peptides/Host Defence Peptides
Antimicrobial peptides (AMP) have potential as a new therapeutic class of antimicrobials and are one of the most promising scaffolds being explored for the generation of much-needed novel antibacterials and antifungals. The blueprint for many AMP as drugs are endogenous Host Defence Peptides (HDP); relatively small peptides (4–50 amino acid residues) that are generally positively charged and often contain an amphipathic conformation (Jiang et al., 2009; Mercer et al., 2019; Torres et al., 2019). In the context of this manuscript AMP refers to all peptides with antimicrobial properties, whereas HDP are essential innate host defence effector molecules and are amongst the “first responders” in all eukaryotic and some prokaryotic organisms to infectious challenge or an inflammation (Zasloff, 2002; Hassan et al., 2012; Mansour et al., 2014; Kang et al., 2017). As well as having direct antimicrobial activity against bacteria, fungi and parasites, HDP can modulate the host immune response, hence being termed host defence peptides (Hancock and Sahl, 2006; Mansour et al., 2014). HDP are often classified according to the structure they tend to adopt in hydrophilic/hydrophobic interfaces, such as the interface of microbial cell membranes and the extracellular environment, e. g., α-helix, β-sheet, etc. (Wang, 2015). Often, ~50% of their sequence comprises hydrophobic and aliphatic residues that facilitate interactions with and translocation across membranes to form pores or to enter cells (Jenssen et al., 2006; Fjell et al., 2011; Aoki and Ueda, 2013). HDP and the AMP derived from these scaffolds are versatile molecules with a wide diversity of structural and physicochemical properties, and are able to target microorganisms through diverse mechanisms of action, although the most common mechanism of action is membrane perturbation/lysis (Brogden, 2005; Hancock and Sahl, 2006; Le et al., 2017; Pyne et al., 2017; Kumar et al., 2018; Aisenbrey et al., 2019). For the purposes of this manuscript, we will refer to both AMP and HDP as AMP.
Achieving precise control over AMP properties and understanding how peptides behave in different environments are still challenges in the field (Naafs, 2018). The understanding of AMP features and details of their mechanism/s of action are still not clear and have been the target of many studies (Porto et al., 2018; Torres et al., 2018; Torres and de la Fuente-Nunez, 2019; Yount et al., 2019). Some of the most promising approaches to describe the role of structural and physicochemical properties on AMP antimicrobial activity are those involving computer-based strategies combined with high-throughput experiments (Lee et al., 2018; de la Fuente-Nunez, 2019; Torres and de la Fuente-Nunez, 2019). Recent advances in computational biology have allowed the development of new molecular descriptors, which enable the discovery of potent AMP through exploitation of their vast sequence space (Awale et al., 2017; Lin et al., 2018). Genetic and pattern recognition algorithms are examples of successful tools that have been used for the generation of AMP antibiotics that display antimicrobial activity both in vitro and even in animal models (Lipkin and Lazaridis, 2017; Cipcigan et al., 2018; Pane et al., 2018; Pfeil et al., 2018; Porto et al., 2018; Rondon-Villarreal and Pinzon-Reyes, 2018). For example, Guavanin 2, an AMP generated by means of a genetic algorithm through a descriptive function that considered amphipathic distribution, net charge and hydrophobicity, was bactericidal at low concentrations, causing the disruption of Pseudomonas aeruginosa membranes by hyperpolarization of the membrane and displaying anti-infective activity in a mouse model (Porto et al., 2018).
In vivo studies in animals have demonstrated that AMP provide protection against microbial infection and that their absence results in an increased risk of infectious disease (Rivas-Santiago et al., 2009). In some cases, protection against infection is relatively generalised, i.e. effected by a number of AMP, such as the combination of drosomycin and metchnikowin and defence against Candida albicans infection in Drosophila melanogaster (Imler and Bulet, 2005; Hanson et al., 2019), whereas in others interactions are very specific, e.g., diptericin and defence against Providencia rettgeri infection in D. melanogaster (Hanson et al., 2019). Clinical correlations between AMP production and protection against infection exist that extend to humans (Hancock et al., 2016; Mangoni et al., 2016; de la Fuente-Nunez et al., 2017a; Coates et al., 2018), as patients with impaired epithelial AMP production (e.g., atopic dermatitis/eczema) are more susceptible to secondary infection, unlike those with increased AMP production (e.g., psoriasis) (Ong et al., 2002; Yamasaki and Gallo, 2008). Thus, it appears to be clear that AMP function as antimicrobials in vivo.
Despite the promise of AMP as novel antimicrobials, a lack of optimization and standardization of experimental conditions for antimicrobial susceptibility testing (AST), including exposure to different pH, salt solutions, serum half-life, and media/biological matrices used during AST (Mahlapuu et al., 2016; Torres et al., 2019) has been a major block to confirming efficacy potential from the outset in AMP drug development pathways. Standardization of experimental conditions for assessing the antimicrobial properties of AMP, and the difficulties encountered therein, are the subject of this manuscript. It is widely believed that AMP represent a group of molecules with the potential for development into a new generation of antimicrobials and for which “standard” AST protocols can significantly underestimate the AMP efficacy as antimicrobial drug candidates. In this era of increasing levels of AMR worldwide, drug development professionals cannot afford to ignore potential antimicrobial drug candidates simply because they do not perform well using “standard” laboratory test methods. Should AMP be successfully developed as therapeutics, due consideration needs to be given to manufacturing peptides on a large scale and safe and ethical disposal of manufacturing by-products and unused peptides. More than 60 peptide-based drugs have been already approved by the Food and Drug Administration (FDA) and more than 400 are in pre/clinical development (Aoki and Ueda, 2013; da Costa et al., 2015; Ageitos et al., 2016; Mahlapuu et al., 2016; Lee et al., 2019). Of these, at least 70 are AMP, with more than 25 in clinical trials (Koo and Seo, 2019). The peptide therapeutics market was valued at >$23 Bn (US) in 2017 and is predicted to be worth >$43 Bn (US) by 2024 (Zion Market Research, 2018). Additionally, peptide-based antimicrobials have been successfully used in the clinic for a number of years, including the antibacterials colistin, vancomycin, daptomycin and the antifungals of the echinocandin class (Hancock and Chapple, 1999; Mercer and O'Neil, 2013). The peptide components of the complex molecules are, in most cases, cyclic (head-to-tail cyclization) or restricted (side chain-to-side chain or side chain-to-end cyclization) or conjugated with other organic compounds, such as carbohydrates or lipids. Cyclization and/or conjugation confer AMP longer half-life and increased bioavailability, thus improving the probability of achieving a successful treatment (Greber and Dawgul, 2017).
Antimicrobial Susceptibility Testing
Antimicrobial susceptibility testing (AST) determines the concentration of an antimicrobial that inhibits microbial growth, for both microbicidal and microbiostatic agents (Brown et al., 2016; Sanguinetti and Posteraro, 2018; Humphries et al., 2019; van Belkum et al., 2019). The importance of accurate AST in at least guiding antibiotic use in the clinic cannot be underestimated (Doern et al., 1994; Kumar et al., 2009; Weiss et al., 2012; Holmes et al., 2016).
During the development of novel antimicrobials, AST is vitally important; (i) to determine the preclinical activity of drug candidates and allow the identification of lead compounds, (ii) to facilitate the determination of the likelihood of resistance development, (iii) to provide estimates of likely in vivo and critically, clinical efficacy when testing compounds in biological matrices replicating sites of infection, e.g., blood/plasma/serum, lung bronchiolar lavage fluid/sputum, urine, biofilms, etc. (Breteler et al., 2011; Macia et al., 2014; Bottger et al., 2017; Ersoy et al., 2017; Nizet, 2017; Savini et al., 2017; Starr and Wimley, 2017; Haney et al., 2019).
Antimicrobial Susceptibility Testing Methods for Existing Classes of Antimicrobials
Most AST, and its interpretation, is conducted using internationally recognised standards developed by bodies including the International Organization for Standardization (ISO), Clinical and Laboratory Standards Institute (CLSI), the European Committee on Antimicrobial Susceptibility Testing (EUCAST), The United States Committee on Antimicrobial Susceptibility Testing (USCAST) and the US Food and Drug Administration (FDA) Center for Drug Evaluation and Research (CDER) (Table 1) (Magiorakos et al., 2012; Kahlmeter, 2015; Humphries et al., 2019). Susceptibility Test Interpretive Criteria (STIC), also known as “breakpoints,” are used to determine the optimal dose of antimicrobials for treating infection and are based on those published by the CLSI, EUCAST, USCAST and the FDA. In December 2017 the FDA launched the Antimicrobial Susceptibility Test Interpretive Criteria website (https://www.fda.gov/drugs/development-resources/fda-recognised-antimicrobial-susceptibility-test-interpretive-criteria) which includes STIC similar to those published by CLSI and EUCAST. Different documents describe breakpoints for bacteria, yeasts, filamentous fungi (moulds) and other microorganisms (Table 1). Despite many similarities and agreements, there remains some lack of harmonisation between AST methods from different organisations (Pfaller et al., 2011, 2014; Chowdhary et al., 2015; Kahlmeter, 2015; Brown et al., 2016; Sanguinetti and Posteraro, 2018; Simjee et al., 2018; Cusack et al., 2019). Interpretive categories most commonly assigned are susceptible (S), indicative of a high probability of a successful outcome, and resistant (R), indicative of a low probability of a successful outcome, although in less common cases other categories include; non-susceptible, intermediate, susceptible-dose dependent and area of technical uncertainty (See the documents in Table 1 for details about these interpretive categories). An alternative STIC is the Epidemiological Cutoff Value (ECV CLSI, 2018f or ECOFF EUCAST, 2019c). The ECV/ECOFF is defined as the MIC that separates a population into isolates with and those without acquired or mutational resistance based on their phenotypic MIC value. An ECV is not a “breakpoint” as there is no clinical outcome or clinical trial data. Thus, an ECV is not a predictor of clinical success, but allows for prediction of whether an isolate has possible resistance to a given antimicrobial (Turnidge et al., 2006; Lockhart et al., 2017). For conventional antimicrobials with known resistance mechanisms, it is easier to define an ECV/ECOFF than a breakpoint, but for AMP, where resistance mechanisms are not necessarily known, or present, it is much more difficult (if not impossible) to define ECV/ECOFF, let alone a breakpoint. If that is the case, then defining STIC for AMP will require an entirely new definition.
Table 1.
Internationally recognised standards for Antimicrobial Susceptibility Testing (AST) and Susceptibility Testing Interpretive Criteria (STIC)/Breakpoints.
| Organisation | Antimicrobial susceptibility testing document | Interpretive criteria document | References |
|---|---|---|---|
| Bacteria | |||
| CLSI | Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically. M07, ED11. Methods for Antimicrobial Susceptibility Testing of Anaerobic Bacteria. M11, ED9. Susceptibility Testing of Mycobacteria, Nocardia spp., and Other Aerobic Actinomycetes. M24, ED3. Methods for Antimicrobial Susceptibility Testing for Human Mycoplasmas. M43, ED1. Methods for Antimicrobial Dilution and Disk Susceptibility Testing of Infrequently Isolated or Fastidious Bacteria. M45, ED3. |
Performance Standards for Antimicrobial Susceptibility Testing (M100 ED29) Performance Standards for Susceptibility Testing of Mycobacteria, Nocardia spp., and Other Aerobic Actinomycetes. M62, ED1. |
CLSI, 2011, 2015, 2018a,b,c,d, 2019 |
| EUCAST | Antimicrobial susceptibility testing: EUCAST disk diffusion method, Version 7.0. EUCAST uses ISO 20776-1 for other bacterial AST methods |
The European Committee on Antimicrobial Susceptibility Testing. Breakpoint tables for interpretation of MICs and zone diameters. Version 9.0, 2019 | EUCAST, 2019a,b |
| FDA | Antibacterial Susceptibility Test Interpretive Criteria, 2018 | https://www.fda.gov/drugs/development-resources/antibacterial-susceptibility-test-interpretive-criteria | |
| USCAST | 2019 USCAST Interpretive tables | http://www.uscast.org/ | |
| ISO | Clinical laboratory testing and in vitro diagnostic test systems—Susceptibility testing of infectious agents and evaluation of performance of antimicrobial susceptibility test devices - Part 1: Reference method for testing the in vitro activity of antimicrobial agents against rapidly growing aerobic bacteria involved in infectious diseases. ISO20776-1. | ISO, 2019 | |
| Yeasts | |||
| CLSI | Reference Method for Broth Dilution Antifungal Susceptibility Testing of Yeasts. M27, ED4. Method for Antifungal Disk Diffusion Susceptibility Testing of Yeasts. M44, ED3. |
Performance Standards for Antifungal Susceptibility Testing of Yeasts, M60, S1. Epidemiological Cutoff Values for Antifungal Susceptibility Testing, M59, ED2. |
CLSI, 2017a,b, 2018e,f |
| EUCAST | Method for the determination of broth dilution minimum inhibitory concentrations of antifungal agents for yeasts. E.DEF 7.3.1. | The European Committee on Antimicrobial Susceptibility Testing: Breakpoint tables for interpretation of MICs. Version 9.0, 2018 | EUCAST, 2017a, 2018 |
| Filamentous Fungi | |||
| CLSI | Reference Method for Broth Dilution Antifungal Susceptibility Testing of Filamentous Fungi. M38, ED3. Method for Antifungal Disk Diffusion Susceptibility Testing of Nondermatophyte Filamentous Fungi. M51, ED1. |
Performance Standards for Antifungal Susceptibility Testing of Filamentous Fungi. M61, ED1. Epidemiological Cutoff Values for Antifungal Susceptibility Testing, M59, ED2. |
CLSI, 2010, 2017c,d, 2018f |
| EUCAST | Method for the determination of broth dilution minimum inhibitory concentrations of antifungal agents for conidia forming moulds. E.DEF 9.3.1. | The European Committee on Antimicrobial Susceptibility Testing: Breakpoint tables for interpretation of MICs. Version 9.0, 2018 | EUCAST, 2017b, 2018 |
The most commonly used manual AST methods are disk diffusion and broth microdilution, although many large hospital laboratories use automated systems, due to improvements in convenience and flexibility, such as BD Phoenix™, Beckman Coulter MicroScan, bioMerieux Vitek® 2, Accelerate Diagnostics PhenoTest and Thermo Fisher Sensititre™ (Syal et al., 2017). In the case of antifungal susceptibility testing, broth microdilution is almost exclusively used (Pfaller and Diekema, 2012; Ostrosky-Zeichner and Andes, 2017). The time taken for conventional AST varies considerably, depending on the infectious agent, and normally is only performed after the pathogen has been cultured and identified at the species level (van Belkum et al., 2019). For bacteria this can be as quick as 24–48 h, but for fungi, isolation and identification can take days, if not weeks, rather than hours and AST may take 48 h or longer (CLSI, 2012, 2017a,b; EUCAST, 2017a,b). Any delay in appropriate antimicrobial therapy can lead to increased mortality for severe infections (Delaloye and Calandra, 2014; Liu et al., 2017). There is, therefore, an urgent need for more rapid AST (Sanguinetti and Posteraro, 2017; Kim et al., 2018; Cansizoglu et al., 2019; Idelevich and Becker, 2019).
The standards for AST available from ISO, EUCAST and CLSI were first implemented almost 60 years ago (World Health Organization, 1961) and have remained largely unchanged since then in a “one size fits all” approach and are used largely unquestioningly by many users (Nizet, 2017). However, some antimicrobials (and microorganisms) do not work in these standards and require modifications to the testing procedures, either by the use of additives to standard media to generate representative efficacy values or by the use of alternative or modified media for fastidious microorganisms (e.g., Haemophilus Test Medium for Haemophilus influenzae and H. parainfluenzae and the addition of 2.5–5.0% (v/v) lysed horse blood to cation-adjusted Mueller-Hinton (CA-MH) medium when testing streptococci) (CLSI, 2018a,b). Interestingly, EUCAST developed a different medium for use with fastidious bacteria (including streptococci and Haemophilus spp.; a modified version of MH agar, with the addition of 5% mechanically defibrinated horse blood and 20 mg/L β-nicotinamide adenine dinucleotide (β-NAD) (Matuschek et al., 2018). Although most efficacy end-points are 100% growth inhibition, there may be a lesser burden for some pathogen/antimicrobial combinations (e.g., ≥50% growth inhibition for fluconazole, flucytosine and ketoconazole for non-dermatophyte moulds CLSI, 2017c) or the determination of minimum effective concentrations (MEC), rather than MIC (e.g., the MEC of echinocandins vs. filamentous fungi is defined as “the lowest concentration of an antifungal agent that leads to the growth of small, rounded, compact hyphal forms compared with the hyphal growth seen in the control well” CLSI, 2017c). Consideration of other assay parameters, such as those described in Table 2, perhaps require attention when conducting these standard procedures or when they are updated.
Table 2.
Factors influencing antimicrobial activity of AMP.
| In vitro | Ex vivo | In vivo |
|---|---|---|
| pH and ionic strength | Biological matrices (e.g., blood) | Animal models of infection |
| Temperature | Mammalian cells | Pharmacokinetics |
| Medium type/composition | Intracellular pathogens | Pharmacodynamics |
| Nutrient concentrations | Air:Liquid or Solid:liquid interface | Metabolic interactions |
| Buffer | Infection models | Polypharmacy (drug- drug interactions) |
| Bicarbonate | Formulation and delivery | |
| Metal ions | Polymicrobial infections | |
| Salt (NaCl) | ||
| Polysorbate-80 | ||
| Synergy/Antagonism with other antimicrobials | ||
| Inoculum size | ||
| Growth Phase (e.g., biofilms, persisters, spores, small colony variants, and other phenotypic variants) | ||
| Charge effects | ||
| Solubility | ||
| Laboratory materials | ||
| Proteolysis | ||
| Biological macromolecules (e.g., protein, DNA) | ||
| Oxygen (hyper-, norm- and hypoxia) | ||
| Mono/Polymicrobial interactions |
In the context of this manuscript, ex vivo refers to experiment parameters that are not in a living host organism (out of the living), but are simulating in vivo conditions. In vivo refers to experiments conducted in/on a living host organism.
When using the same AST method, e.g., broth microdilution, results can be influenced by factors such as medium age, presence of polysorbate 80 and ion content (Bradford et al., 2005; Fernandez-Mazarrasa et al., 2009; Sader et al., 2012; Sutherland and Nicolau, 2014) as can non-compliance with AST standards (Mouton et al., 2018; Turner and Ashley, 2019). Examples where modifications to existing AST methods have been successfully implemented include the lipopeptide antibiotic daptomycin. Daptomycin requires physiological concentrations of calcium (50 mg/L) in the medium for optimal efficacy or otherwise MIC values can be up to 32-fold higher, clearly affecting whether an isolate could be sensitive or resistant (Eliopoulos et al., 1986; Campeau et al., 2018) and therefore the CA-MH broth or agar is supplemented with additional Ca2+ (CLSI, 2018a,b). The lipoglycopeptides antibiotics (including oritavancin, dalbavancin, and teicoplanin) are subject to binding to laboratory plasticware (Arhin et al., 2008; Ross et al., 2014) and therefore CLSI recommends addition of 0.002% (v/v) polysorbate 80 (Tween 80) to CA-MHB to prevent such binding (CLSI, 2018a). When testing staphylococci for sensitivity to oxacillin (MRSA phenotype) it is recommended to supplement media with 2% (w/v) sodium chloride as this enhances the expression of mecA-mediated oxacillin resistance and reduces the reporting of false negatives that occurs when 5% (w/v) sodium chloride is used (Huang et al., 1993; Brown, 2001). Additionally, CLSI recommends that when testing for oxacillin resistance in staphylococci samples should be incubated at 33–35°C, as testing at temperatures above 35°C may not detect mecA-mediated resistance (CLSI, 2018a). Therefore, antimicrobial substance-specific changes can be made to “standard” AST methods, so there is no reason why this should not be possible for AMP in pre-/clinical development. Additionally, AMP-specific end points do not necessarily have to equate to 100% growth inhibition. Obviously, any and all deviations from “standard” protocols will require rigorous and detailed justification and validation.
AMP represent one such group of molecules for which these “standard” protocols can significantly underestimate their efficacy potential. In this era of increasing levels of AMR worldwide, can drug developers really afford to ignore potential antimicrobial drug candidates simply because they do not perform well using “standard” test methods? AST of AMP is normally performed using the broth microdilution (or macrodilution) procedure, as many AMP are positively charged molecules (and are dependent on that positive charge for activity Jiang et al., 2009; Mercer et al., 2019; Torres et al., 2019). Positively charged AMP interact with negatively charged components in agar and neutralising their activity, meaning that disk diffusion AST methods significantly under-estimate activity or mask it completely (Kunin and Edmondson, 1968; Lehrer et al., 1991), as is the case for the peptide antibiotics daptomycin (Humphries et al., 2013) and colistin (Albur et al., 2014; Poirel et al., 2017; Matuschek et al., 2018).
If AMP are to realise their potential as a future generation of anti-infective therapeutics, AST methods will require approval from regulatory authorities and buy-in from organisations such as EUCAST, CLSI, and ISO (Kahlmeter, 2015; CLSI, 2018g). To do this, any AST method for AMP or modification to an AST method, at least for EUCAST, must be calibrated to the ISO broth microdilution technique, often as part of the formal accreditation process (Kahlmeter, 2015; ISO, 2019). Therefore, any AST method must be accurate, robust, reproducible, have clinical utility and validity and, ultimately, be amenable to automation.
Antimicrobial Susceptibility Testing of AMP
It is clear that current AST methodologies are not “fit-for-purpose” for determining the activity of AMP in vitro. When considering a single antimicrobial and a single species of microorganism, inter-laboratory variation and biological variation (i.e., variation between strains) can still cause a broad distribution of wild-type MIC values (Annis and Craig, 2005; Hombach et al., 2016; Mouton et al., 2019). Therefore, efforts to introduce standardised AST methods for AMP may turn out to be an extremely onerous and difficult task (Jepson et al., 2016). Previous attempts to standardise AST for AMP have met with limited success. The method devised by the Hancock lab (http://cmdr.ubc.ca/bobh/method/modified-mic-method-for-cationic-antimicrobial-peptides/), was an adaptation of the CLSI broth microdilution procedure (CLSI, 2018a), but has not been widely adopted. The main differences between the two methods were careful choice of labware (polypropylene or Sigmacote-coated glass), diluent used (0.01% (v/v) acetic acid containing 0.2% (w/v) bovine serum albumin) and determining an MIC50 (reduction in growth of ≥50%) in the Hancock method. A comparison of the two methods revealed differences in MIC in which the Hancock method generated MIC values that were predominantly lower than the CLSI method (Giacometti et al., 2000). In the two decades since the publication of this method, interest in AMP has grown significantly, so consideration of AST for AMP is perhaps overdue a review and update.
Membrane-active AMP are typically associated with higher MIC values than those of conventional antibiotics, although this is not always the case. Is this simply because the membrane represents a very large target, especially for AMP that kill via the carpet mechanism and even some pore-formers (Figure 1), or is this because currently used test methods are not fit for purpose? For example, Roversi et al. determined that for the cathelicidin peptide PMAP-23 to kill E. coli ATCC25922 required 106-107 bound peptides per cell (1–10 μM; 2.7–27 mg/L) (Roversi et al., 2014). This compares to a typical susceptible MIC distribution of ciprofloxacin vs. E. coli of 0.004–0.064 mg/L (0.012–0.193 μM) (R > 0.5 mg/L; ECOFF = 0.064 mg/L) (http://www.eucast.org) and which specifically targets the enzymes DNA gyrase and topoisomerase IV, thereby preventing DNA replication. PMAP-23 kills bacteria by the “carpet” model (Orioni et al., 2009), so this is likely to be more peptide molecules than required for pore-forming peptides, or those with more specific mechanisms of action (Figure 1).
Figure 1.
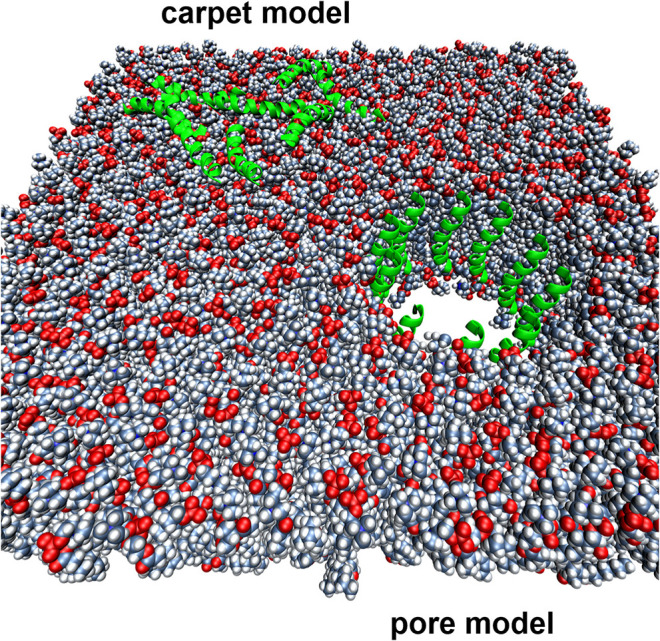
Two different mechanisms of action proposed for membrane-active AMPs. The lipid bilayer is represented by spheres, while the AMPs are represented by green helical cartoons.
A lack of antimicrobial activity of AMP, or attenuation of activity, has been observed for AMP when assayed under conditions in which existing, conventional antibiotic classes are active. This has been ascribed to a variety of features, including inactivation by physiological concentrations of NaCl and divalent metal cations or serum/plasma. However, manipulation of media conditions, including making them more physiologically relevant, could potentially reverse the inhibitory effects of compounds such as NaCl and proteins. For example, addition of sodium bicarbonate was able to reverse the inhibitory effects of physiological NaCl concentration against a number of different AMP, including the human cathelicidin LL-37, human β-defensin 2 (hBD-2) and Cryptidin-4, albeit not dermcidin (Dorschner et al., 2006). Many of the factors for consideration described here have also been investigated for their effects on the activity of conventional antimicrobials with varying outcomes (Hajdu et al., 2010; Erdogan-Yildirim et al., 2011; Ersoy et al., 2017; Oesterreicher et al., 2019).
It has been suggested that the discovery and preclinical development of new antimicrobials should target pathogens as they are found at sites of infection, rather than the potentially different phenotype demonstrated in microbiological growth media (Dorschner et al., 2006; Ersoy et al., 2017). One reason for this is because standard AST does not take into account the potential influence (positive or negative) of the host cell environment on microbial susceptibility and resistance (Sutherland and Nicolau, 2014; Haney et al., 2019). In order to create a standardised AST procedure suitable for AMP there are a number of factors that will need to be considered (Table 2).
Whilst many of the factors described in Table 2 may have an impact on the design of AST for AMP, or the MIC of individual AMP, combinations of changes to these factors must also be considered (Oesterreicher et al., 2019).
Laboratory Materials
A number of publications have demonstrated that the results of AST of many antimicrobials, not just AMP, can be affected by the choice of laboratory plasticware for use with the broth microdilution procedure and even the choice of tubes used for preparing reagents (Singhal et al., 2018; Kavanagh et al., 2019). This also applies to AST of AMP (Otvos and Cudic, 2007; Wiegand et al., 2008; Kristensen et al., 2015).
Some peptide-based antibiotics and AMP need to be prepared in alternative solvents, or with additives included in the media/diluent to prevent binding to the surfaces of tubes and plates. The lipoglycopeptides, including oritavancin and telavancin, must be solubilised in dimethyl sulfoxide (DMSO) or with the addition of a surfactant, 0.002% (v/v) polysorbate 80 (Tween 80), added to the water to prevent binding (Arhin et al., 2008; Ross et al., 2014). When DMSO was used in place of water as solvent for AST of the echinocandins (lipopeptide antifungals) caspofungin and micafungin, MIC values were lower and MIC ranges were narrower (Alastruey-Izquierdo et al., 2012) as was the case when water was supplemented with 50% bovine serum albumin (Arendrup et al., 2011; Garcia-Effron et al., 2011). In contrast, even though colistin (cyclic lipopeptide) binds to plastics (Karvanen et al., 2017), the CLSI and EUCAST recommend broth microdilution for AST of colistin, but without added surfactant (Hindler and Humphries, 2013; CLSI-EUCAST, 2016), as polysorbate 80 can act synergistically with polymyxins and reduce MICs (Brown and Winsley, 1968; Ezadi et al., 2019), presumably due to interactions with lipopolysaccharides in the Gram negative outer membrane (Correa et al., 2017). Additionally, polysorbate has antibacterial properties of its own (Brown et al., 1979; Figura et al., 2012). In the case of AMP, the potential use of polysorbate is more complex as the addition of polysorbate 20 improved antiviral activity of LL-37 and magainin-2B amide, but reduced antibacterial activity (Ulaeto et al., 2016), however the impact of polysorbate on AMP activity has not been extensively studied. The effect of polysorbate for the prevention of binding of AMP to laboratory plastics would need to be carefully investigated in light of possible synergistic activity as observed with the cationic, membrane-active polymyxins. This is clearly an area that requires more detailed investigation before any recommendation specific to AMP susceptibility testing can be made. In the method described by the Hancock laboratory (http://cmdr.ubc.ca/bobh/method/modified-mic-method-for-cationic-antimicrobial-peptides/) for AST of AMP, they recommend the use of a diluent of 0.01% acetic acid containing 0.2% bovine serum albumin (BSA) to reduce peptide binding to plastic surfaces (Wiegand et al., 2008), although it is difficult to ascertain whether this recommendation has been broadly adopted. Even when it comes to the choice of filter for filter-sterilisation of AMP-containing solutions, caution may be required, as cationic AMP may bind to negatively charged membranes, such as cellulose acetate (Wiegand et al., 2008).
The choice of laboratory plasticware is similarly problematic as there are a number of publications that indicate that cationic AMP bind to polypropylene, polystyrene and borosilicate glass, the most commonly used materials used in the manufacture of labware (Chico et al., 2003; Kristensen et al., 2015), although the extent of binding may be similar regardless of the type of material used (Sanchez-Gomez et al., 2008; Kristensen et al., 2015). This situation also applies to some antibiotics (see above) and even antifungals such as the echinocandins (Fothergill et al., 2016; Arendrup et al., 2019). In the study of Kristensen and co-workers, binding to the surface of the tubes (irrespective of material) was saturable and relatively more peptide bound at low concentrations, so a simple solution may be to keep peptide concentrations as high as possible to minimise the percentage lost to protein binding. Irrespective of this, this would lead to an underestimation of AMP efficacy when trying to determine MIC values at lower peptide concentrations. The use of low protein-binding plasticware demonstrated reduced binding of selected AMP when compared to polystyrene, polypropylene and borosilicate glass (Kristensen et al., 2015), but they unfortunately did not carry out AST in this study, so it is not currently possible to assess whether the use of low protein-binding labware would lead to a reduction in MIC for AMP without further studies and, additionally, this is unlikely to provide a low cost solution.
The possibility of using different diluents, additives, or changes to laboratory plasticware clearly warrants further investigation, but perhaps the best recommendations are to be as consistent as possible with your choices of materials and diluents and keep AMP concentrations high where possible to minimise the relative amount of peptide loss by binding.
Media Composition
Most AST using AMP is conducted using broth dilution methods described by EUCAST or CLSI, or close variants thereof. For bacteria, when conducting broth dilution AST, both organisations recommend the use of Mueller-Hinton broth (MHB) containing 10–12.5 mg/L Mg2+ and 20–25 mg/L Ca2+ (CLSI, 2018a; EUCAST, 2019a), whereas for testing of fungi, both recommend RMPI-1640 medium (RPMI), but with different concentrations of glucose; 2.0 g/L for CLSI test methods and 20.0 g/L for EUCAST test methods (CLSI, 2017a,c; EUCAST, 2017a,b). The reasons for the almost universal adoption of MHB for antibacterial susceptibility testing are unclear, but MHB serves as a poor simulation of normal human body fluids (Ersoy et al., 2017; Nizet, 2017). Antifungal susceptibility testing using RPMI-1640 liquid medium (CLSI, 2017a,c; EUCAST, 2017a,b) more closely simulates normal human body fluids, whereas CLSI disk diffusion testing of fungi and yeasts again uses Mueller-Hinton agar (MHA) (CLSI, 2010, 2018e). The effect of media type on AST results has been examined in a number of studies (Schwab et al., 1999; Sajjan et al., 2001; Dorschner et al., 2006; Kumaraswamy et al., 2016; Ersoy et al., 2017; Tucker et al., 2018). The use of MHB for AST of cationic AMP may be an unsuitable medium due to the high content of anionic amino acids in hydrolysed casein. MHB contains 17.5 g/L acid hydrolysate of casein, with only 3 g/L beef extract and 1.5 g/L starch, as well as additional Ca2+ (20–25 mg/L final concentration) and Mg2+ (10–12.5 mg/L final concentration). Casein contains 25% anionic amino acids and these can interfere with cationic AMP activity and cause them to precipitate (Turner et al., 1998). Turner and co-workers compared the activity of LL-37 and protegrin (PG-1) in conventional MHB and MHB that had first been passed through an anion exchange column to deplete the MHB of anionic compounds and demonstrated that MICs against E. coli ML-35p, P. aeruginosa MR3007, Bacillus subtilis and S. aureus 930918-3 were 3 - >20-fold higher in standard MHB compared to MHB passed through an anion exchange column (Turner et al., 1998). The choice of media can make a significant impact on AMP efficacy. Incubation for 2 h with 5 μM of the cationic AMP D4E1 resulted in 100% killing of ~1 × 105 cfu of S. aureus when incubated in RPMI-1640 liquid medium buffered with 30 mM HEPES, whereas the use of MHB, CA-MHB, nutrient broth + 125 mM NaCl or Tryptone soya broth (TSB) resulted in less killing (68.6, 22.2, 56.1, and 5.3%, respectively). Similar trends were observed for P. aeruginosa ATCC27853 and another cationic AMP (D2A21) (Schwab et al., 1999). In the Schwab study, a range of different buffers were also tested (PBS, Tris-NaCl, citrate-phosphate, saline and phosphate), with a trend for less killing in the phosphate buffers. Interestingly, osmolarity did not have any effect on the activity of D2A21 against a range of bacterial pathogens isolated from patients with cystic fibrosis (CF) (Schwab et al., 1999). When determining the efficacy of the histatin derivative P-113, Sajjan and co-workers observed little activity in CA-MHB, but when this was diluted 20-fold the MIC was 3.1 mg/L, indicating that one or more components of CA-MHB were inhibitory to the antimicrobial activity of P-113 against P. aeruginosa. Use of an alternative medium (LM broth) that attained similar growth and growth rates to CA-MHB, but with reduced concentrations of monovalent and divalent salts, retained the efficacy of P-113 to a range of bacterial CF pathogens, but did not affect the efficacy of tobramycin, ceftazidime or imipenem, indicating the detrimental effect of monovalent and/or divalent salts on AMP efficacy (Sajjan et al., 2001).
The activity of azithromycin against different isolates of Stenotrophomonas maltophilia was examined in cation-adjusted MHB (CA-MHB) and RPMI-1640 liquid medium supplemented with 10% Luria-Bertani broth (LB) (Kumaraswamy et al., 2016). In this study MICs in MHB were 32–256 mg/L, whereas in RPMI, MIC values were significantly lower (0.03–0.25 mg/L) and azithromycin also synergised with the AMP LL-37 against MDR P. aeruginosa and MDR Acinetobacter baumannii. When tested in vivo, azithromycin also resulted in clearance of A. baumannii and S. maltophilia, sensitising S. maltophilia to neutrophil killing (Lin et al., 2015; Kumaraswamy et al., 2016). A series of 20 AMP were isolated from a surface-displayed peptide library and tested for activity in MHB via broth microdilution, but only 2 of the peptides, and cecropin P1, demonstrated an MIC, but when the MBC was determined in 10 mM Tris, pH 7.4 + 25 mM NaCl, all AMP were bactericidal (MBC ≤ 128 μM) against at least 1 of 4 Gram negative bacteria (Tucker et al., 2018). In other cases, the use of diluted nutrient media results in improved efficacy of AMP in AST. The MIC of the lactoferricin derivative HLopt2 against selected Candida spp. was >250 mg/L, but when tested in BHI diluted 1:100 the minimum microbicidal concentrations (MMC) were 2–31 mg/L. Similarly, the MIC in CA-MH vs. P. aeruginosa and S. aureus was much higher (125 and 63 mg/L, respectively) than the MMC when tested in 1:100-diluted BHI (4 and 2 mg/L, respectively) (Ptaszynska et al., 2019). In some cases, the choice of media type can have detrimental effects on AMP activity. For example, the semi-synthetic AMP Lin-SB056-1 was bactericidal against 6 P. aeruginosa in 1% TSB (MBC = 1.56–3.12 mg/L), whereas activity was lost in 80% artificial sputum medium, closely resembling the CF lung, except in the presence of additional EDTA (ethylenediaminetetraacetic acid), where bactericidal activity was restored (Maisetta et al., 2017).
The use of a more physiologically representative cell culture medium (RPMI-1640) may be better suited for AST of AMP, and is certainly more representative of conditions in vivo than CA-MHB or other nutrient-rich microbiological growth media and that the presence of higher concentrations of anionic substances adversely affects the efficacy of cationic AMP. In a caveat to this, it has recently been observed that the composition of a number of cell culture media, including RPMI-1640, do not simulate bodily fluids particularly closely (McKee and Komarova, 2017), so perhaps further work is required to develop media that more closely simulates mammalian bodily fluids.
Solubility and Aggregation
Given that most AMP have a net positive charge and hydrophilic regions, aqueous solubility at concentrations required for AST are not problematic, although some AMP aggregate and become insoluble at relatively high concentrations in certain media and in the presence of selected anions. This can result in a loss of activity, stability and/or increased cytotoxicity (Frokjaer and Otzen, 2005; Ratanji et al., 2014; Haney et al., 2017). Hydrophobic regions in peptides are known to self-associate and drive the formation of aggregates (Kim and Hecht, 2006), so modifications to AMP design may reduce/prevent aggregation (Haney et al., 2017). For example, the AMP Temporin L forms aggregates in water and due to an extended hydrophobic region, but substitution of glutamine to lysine at position 3 significantly reduced aggregation in water and improved its antiendotoxin properties (Srivastava and Ghosh, 2013), whereas substitution with arginine at the same position improved activity against P. aeruginosa (and is also likely to attenuate aggregation) (Mangoni et al., 2011). For example, significant aggregation of the immunomodulatory AMP IDR-1018 occurred in the presence of phosphate, benzoate, nitrate, and citrate, but less aggregation was observed in the presence of acetate, chloride, water and, perhaps significantly, bicarbonate (see later). IDR-1018 also exhibited aggregation in 10% RPMI and 1% MEM tissue culture media and also co-precipitated with serum proteins in a concentration-dependent manner, in many cases adversely affected the desired immunomodulatory properties of the peptide and increased cytotoxicity (Hartlieb et al., 2017). Protegrin-4 (PG-4) also aggregates in the presence of phosphate (>2.0 mg/ml PG-4 in 50 mM sodium phosphate buffer, pH 7.4), forming amyloid-like fibrils and retained antimicrobial activity against B. subtilis (Gour et al., 2019). LL-37 exists in equilibrium between monomers and oligomers in solution at low concentrations and oligomerizes in the presence of zwitterionic membranes (Johansson et al., 1998; Oren et al., 1999). Dermaseptin S9 formed aggregates and amyloid-like fibrils and the peptide binds to membranes in an aggregated state (Caillon et al., 2013). Human α-defensin 6 (HD6) is a 32-residue cysteine-rich peptide that lacks the broad-spectrum antimicrobial activity observed for other human α-defensins. Strikingly, HD6 oligomerises to form “nanonets,” due to the disposition of hydrophobic residues in the HD6 primary structure, that entrap microbes and prevent invasive pathogens such as Salmonella enterica serovar Typhimurium and Listeria monocytogenes from entering host cells in the gastrointestinal tract (Chu et al., 2012; Chairatana and Nolan, 2017). An in silico analysis of the aggregative potential of AMP and non-antimicrobial peptides revealed that AMP demonstrate very low in vitro aggregation propensity, but high in vivo aggregation propensity. Non-antimicrobial peptides can be divided in two main groups, presenting either high or low values for both in vivo and in vitro aggregation. These results suggest that most AMP demonstrate minimal aggregation in aqueous solution, but promote aggregation in a more hydrophobic environment (i.e., the bacteria cell membrane) (Torrent et al., 2011), something borne out in many experimental studies. Thus, when conducting AST with AMP, consideration of the solute/s used can be important and, in most cases, it would be advantageous to use aqueous solutions where possible for formulation of drugs for human or animal use, to minimise aggregation and to carefully assess peptide solubility in the media used for AST.
Biological Matrices
When testing the efficacy of any antimicrobial, logic dictates that it would be practical to test efficacy in the biological matrix at the site of infection, e.g., blood, sputum, urine, etc., but this is unlikely to be practical for routine screening. However, prior to the initiation of in vivo efficacy studies it would be sensible to determine the efficacy of AMP in relevant biological matrices. Proteolytic degradation of AMP is often considered a major weakness limiting their potential therapeutic application, as is binding to biological matrices, including serum/plasma proteins (Wang et al., 1998; Sivertsen et al., 2014), nucleic acids (Park et al., 1998; Hsu et al., 2005), ribosomes (Mardirossian et al., 2014) other proteins (Tu et al., 2011) bacterial cell walls (Malanovic and Lohner, 2016) and lipopolysaccharide (Piers et al., 1994; Sun and Shang, 2015). Many AMP can also bind to host cells as part of their innate immune system functions (van der Does et al., 2019) that are present in most biological matrices. For example, LL-37 has direct interactions with ≥16 proteins/receptors, that subsequently interact with >1000 secondary effector proteins and when used to stimulate monocytes >900 gene expression changes were observed (Hancock et al., 2016). It is, therefore, not unreasonable to assume that many AMP will interact with host cells and that the effects of these interactions may not necessarily be desirable. This may not need to be assessed when considering in vitro AST, but would need consideration, for example, at later stages of the drug development process. Significant efforts have been made to improve AMP activity in blood, plasma and/or serum (Hamamoto et al., 2002; Knappe et al., 2010; Nguyen et al., 2010; Chu et al., 2013; Dong et al., 2018; Kumar et al., 2018). When considering AMP activity in blood, serum or plasma the source of the blood and the method of collection must be taken into account. Most blood samples are collected in tubes (vacutainers) containing an anticoagulant to prevent blood clotting and it is known that the presence of an anticoagulant can affect AMP activity. For example, EDTA and citrate are known to enhance the efficacy of some AMP as they can chelate divalent cations which are inhibitory to the activity of a number of AMP (Wei and Bobek, 2005; Walkenhorst et al., 2014; Maisetta et al., 2017; Umerska et al., 2018; Grassi et al., 2019) and EDTA and heparin can inactivate proteases found in blood, including metalloproteases (EDTA), thrombin and Factor Xa (heparin) that may prevent AMP hydrolysis (Bowen and Remaley, 2014; Bottger et al., 2017; Rawlings et al., 2018).
The stability of proline-rich AMP (apidaecin and oncocin derivatives) was examined in murine blood, serum and plasma and, perhaps surprisingly, the general trend was for greatest stability in whole blood, followed by plasma and least stable in serum, albeit only over a 1 h incubation period, and that substitution of L-arginine residues for D-arginine or ornithine improved stability in blood, serum and plasma (Bottger et al., 2017). In another study, pre-incubation of a panel of AMP with red blood cells (RBC) (1 × 109 RBC/ml) before exposure to the pathogen significantly increased the MIC of most AMP, with a similar inhibitory effect caused by serum, even though the affinity of AMP for bacteria was much greater than for RBC. Thus, serum binding and binding to host cells for AMP intended for systemic delivery requires consideration and can be adapted to AST testing in the presence of serum or host cells (Starr et al., 2016). However, when the AMP DNS-PMAP23 or esculentin-1a(1-21)NH2 was added directly to a mixture of RBC and E. coli, no significant inhibition of antibacterial activity took place (Savini et al., 2017), indicating that experimental set-up, and by extension the nature of the infection being potentially treated, is an important consideration. In another study, in the absence of host cells, WLBU2 (12.5 μM) retained activity in the presence of 98% human serum, whereas LL-37 was not active at concentrations up to 100 μM (Deslouches et al., 2005) and therefore the effect of biological matrices on AMP activity may be peptide-specific.
The efficacy of the histatin-derived AMP, P-113, was tested in diluted sputum from CF patients and no activity vs. P. aeruginosa was observed. When the stability of P-113 was determined in sputum, half-lives of 2.8–58.5 min were determined, probably preventing activity in sputum. By switching the composition of P-113 from all L-enantiomer amino acids to all D-enantiomer amino acids (P-113d), the activity against P. aeruginosa was comparable to the L-enantiomer peptide, but P-113d was stable in CF sputum for 7 d. When the efficacy of P-113d was tested in CF sputum, an additional 1 log kill of P. aeruginosa was attained in 1 h and efficacy was enhanced further by pre-treatment of the sputum with recombinant human deoxyribonuclease (rhDNase; Pulmozyme®), a therapeutic used in some CF patients (Sajjan et al., 2001). Interestingly, the activity of an all L-isomer of the AMP temporin lost activity in faeces within 30 min, whereas the all D-isomer version retained activity for 30 min, but activity was lost after 24 h (Oh et al., 2000). Thus, testing of AMP in biological matrices can be factored into AST and the earlier this is conducted will have a bearing on lead selection for AMP intended for specific infections.
pH and Ionic Strength
More than 30 AMP are known to have pH-dependent activity, including LL-37, histatins, psoriasin, and lactoferrin, with greater activity predominantly observed at lower pH values, especially for histidine-containing AMP such as clavanins (Lee et al., 1997; Malik et al., 2016; Alvares et al., 2017). Changes in pH can affect ionic interactions between membranes and AMP by changing the protonation states of functional groups on the membrane and/or AMP, as well as effects on the ionic strength of the solution (Walkenhorst, 2016). Cationic AMP are normally more efficacious at neutral and lower pH due to the loss of net positive charge at alkaline pH (Malik et al., 2016). For example, the efficacy of LL-37 against C. albicans was greater at pH 4.5 (81% death) when compared to pH 5.5 (79% death) and pH 7.2 (40% death) (Lopez-Garcia et al., 2005). Localised pH can significantly impact the interaction of AMP with membranes and their subsequent ability to perturb the membrane (Malik et al., 2016; Alvares et al., 2017). This can reflect their predominant site of action, e.g., skin. The ionic strength of the buffer can also influence efficacy, with reduced efficacy often observed at higher ionic strengths (or an over-estimation of activity at low ionic strengths) (Sanchez-Gomez et al., 2008; Walkenhorst et al., 2013). Additionally, the choice of buffer can affect AMP efficacy, with greater efficacy observed in MOPS compared to phosphate buffer at similar pH and ionic strength (Walkenhorst, 2016). However, there are a number of exceptions to this, that may be peptide-dependent or organism dependent. Walkenhorst and co-workers observed the expected trend of enhanced activity at lower pH values for a family of peptides versus Gram negative bacteria and C. albicans, but the opposite effect was observed against S. aureus, with enhanced efficacy at higher pH values and hypothesized that this was due to changes to teichoic acids in the S. aureus cell wall, making the peptidoglycan layer less negatively charged at neutral and acidic pH (Walkenhorst et al., 2013). Interestingly, a linear form of esculentin 2EM caused greater cell lysis at pH 8.0 compared to pH 6.0 and this correlated with increased α-helicity of the peptide (Malik et al., 2016), indicating that the effect of pH on AMP activity cannot be readily predicted and needs to be determined where necessary.
Despite the effect of pH on AMP activity (which in many cases has not been investigated), AST of AMP is predominantly carried out at neutral pH and the effect of pH modulation is not considered. When considering the relevance of pH on AST of AMP, the main consideration should be of the pH at the site of infection and during delivery to the site of infection. Many assume that this is close to neutral, as an often cited physiological pH is 7.4, although pH can range from 7.0 to 9.0 in blood (Kellum, 2000). However, physiological pH values cover a relatively broad range, including pH 5.0 in the macrophage phagosome a site where intracellular pathogens such as K. pneumoniae, Salmonella typhimurium, E. coli and Mycobacterium tuberculosis can reside (Underhill and Ozinsky, 2002) and between 4 and 7 on the skin surface, with a most frequently determined pH range of 4.0–5.9 (Lambers et al., 2006), although the pH of chronic wounds can be alkaline (pH 7.15–8.9) (Gethin, 2007). Skin infection can cause an elevation in skin pH, as can other conditions, such as diabetes mellitus, that can lead to increased risk of infection. Additionally, wound healing is associated with less acidic pH which can influence microbial colonisation and infection (Rippke et al., 2018). In the urinary tract the pH of normal urine is slightly acidic (pH 6–7.5), but a range as wide as 4.0–8.0 is normal. During infection this can rise to 9.0 (e.g., “urea-splitting” pathogens such as Proteus spp., Klebsiella pneumoniae, or Ureaplasma urealyticum) and is a clear indicator of a UTI (Bono and Reygaert, 2019). Conventional AST is conducted at pH 7.2–7.4 for bacteria and pH 7 for fungi which may be appropriate for many infections, but consideration of the pH (and the buffer used to attain this) for testing should be taken into account when investigating target pathogens or infections at sites with different pH.
Oxygen (Hyper-, Norm-, and Hypoxia)
Antimicrobial susceptibility testing is normally conducted under conditions of normoxia, yet in tissues the amount of oxygen range between <1 and 11% oxygen, whereas in vitro experiments are normally performed in 19.95% oxygen, an artificially high concentration relative to tissue concentrations. For example, in normal air the oxygen partial pressure is 160 mmHg, whereas in alveoli, this is reduced to 110 mmHg, in the brain 23–48 mmHg and in the colon, only 3–4 mmHg (Carreau et al., 2011). Areas of hypoxia are features of sites of bacterial infection, healing wounds and other diseased tissues (Murdoch et al., 2005) and hypoxia can induce the expression of hBD-2 (Nickel et al., 2012). Thus, physiological oxygen concentrations vary widely, yet are largely not considered in the context of antimicrobial efficacy, unless specifically considering activity against anaerobes or microaerophiles. Given the membrane disruption mechanism of action of many AMP, should oxygen be a factor affecting their activity? The plectasin-derived AMP, NZ2114 and MP1102, were bactericidal by membrane lysis vs. Clostridium perfringens under anaerobic conditions (>3 log kill in <60 min) (Zheng et al., 2017), whereas LL-37 and hBD-3 (<5 mg/L) were bactericidal against C. difficile under anaerobic conditions (Nuding et al., 2014). The activity of human defensins against anaerobic bacteria revealed that human α-defensin 5 and hBD-1 were minimally active against a panel of 25 strict anaerobes, hBD-2 demonstrated relatively weak activity against most strict anaerobes, whereas hBD-3 was active against 18 of 25 strict anaerobes tested (Nuding et al., 2009). Interestingly, human α-defensin 6 (HD-6), the second most abundant AMP produced by Paneth cells in the small intestine (Wehkamp et al., 2005), does not demonstrate direct antimicrobial activity under standard aerobic conditions, but demonstrated direct killing of Bifidobacterium adolescentis, Lactobacillus acidophilus, and Bif. breve, Bif. longum and Streptococcus salivarius subsp. thermophilus under reducing conditions (mimicking anaerobiosis) (Schroeder et al., 2015). Oh and co-workers tested 16 CAMEL peptides (cecropin-melittin hybrids) against a selection of anaerobes (Peptostreptococcus spp., C. difficile, Bacteroides fragilis, Prevotella, spp., Fusobacterium nucleatum and Propionibacterium spp.; n = 60) under anaerobic conditions and all were active (MIC90 = 1–32 mg/L) (Oh et al., 2000). Piscidins and ixosin are AMP that contain the copper- and nickel-binding ATCUN motif. Bactericidal activity under aerobic conditions is enhanced when these AMP bind copper, but under anaerobic conditions two piscidins (p1 and p3) and ixosin retain activity, but this does not depend on the presence of copper ions (Libardo et al., 2016; Oludiran et al., 2019). Thus, it would appear that antimicrobial activity of AMP under anaerobic conditions is dependent on the AMP used and for the purposes of AMP vs. anaerobes would need to be assessed on a case-by-case basis.
Proteolysis
Susceptibility to proteolysis is often viewed as one of the most significant limitations when trying to develop peptide drugs, including AMP (Vlieghe et al., 2010; Lecaille et al., 2016; Starr and Wimley, 2017). When conducting AST with AMP, protease production by the pathogen of interest is an obvious potential cause of reduction in/or loss of activity (Stumpe et al., 1998; Schmidtchen et al., 2002; Nesuta et al., 2017; Rapala-Kozik et al., 2018). Additionally, if testing were to be carried out in biological matrices other than growth media (e.g., blood, saliva etc.), then proteolysis by relevant host proteases could adversely affect AMP activity (Knappe et al., 2010; Lecaille et al., 2016; Starr et al., 2016; Bottger et al., 2017; Starr and Wimley, 2017). To determine whether proteolysis could occur during AST, protease inhibitors could be included in the system (Shin et al., 2010). However, this is not necessarily as simple as it appears. A number of protease inhibitors, including EDTA and citrate, can enhance AMP activity by chelation of metal ions, or by other unknown mechanisms, and this activity would need to be determined prior to their use in AST (Wei and Bobek, 2005; Walkenhorst et al., 2014; Maisetta et al., 2017; Umerska et al., 2018; Grassi et al., 2019).
Bicarbonate
Bicarbonate (NaHCO3) is relatively common in mammalian tissues (NaHCO3; 24.90 ± 1.79 mM in human blood Wishart et al., 2018) and the bicarbonate buffer system, sodium bicarbonate in balance with carbonic acid, helps to maintain the physiological pH, including blood, interstitial fluid and the upper gastro-intestinal tract (Boron and Boulpaep, 2005). However, bicarbonate warrants additional consideration beyond its capacity for maintaining pH homeostasis. Despite its importance physiologically, it is not routinely considered when conducting AST or in the maintenance of pH during AST. Even though AST of fungi uses RPMI-1640 liquid medium, a mammalian cell culture medium, this is buffered to pH 7.0 with MOPS (CLSI, 2017a,c), rather than sodium bicarbonate as it would be when culturing mammalian cells and using a CO2 incubator (5% CO2). Sodium bicarbonate has antibacterial, antifungal and antibiofilm properties of its own, but only at supra-physiological concentrations (≥50 mM) (Corral et al., 1988; Xie et al., 2010; Letscher-Bru et al., 2013; Dobay et al., 2018; Farha et al., 2018). Bicarbonate acts as a selective dissipater of the trans-membrane pH gradient, a component of the proton motive force (along with the membrane potential) and can enhance the activity of AMP, including LL-37, α-defensin, indolicidin, protegrin and bactenecin and selected antibiotics, including aminoglycosides, macrolides and selected fluoroquinolones. The activity of AMP was enhanced as both the AMP and bicarbonate perturb bacterial membrane potential and in the case of antibiotics, enhancement of activity was predominantly limited to those whose uptake is driven by the membrane potential (Farha et al., 2018). Interestingly, tobramycin (aminoglycoside) activity against isolates of P. aeruginosa was enhanced in the presence of bicarbonate against planktonic cells, but the combination promoted biofilm growth (Kaushik et al., 2016). The relevance of bicarbonate addition during AST of selected antibiotics against bacteria has been investigated. When CA-MH broth was supplemented with physiological levels of bicarbonate, this improved the predictive value of AST for treatment of in vivo infections for a number of antibiotic and pathogen combinations, potentially due to structural changes to bacteria and changes in gene expression (Ersoy et al., 2017). When analysing the effect of bicarbonate on the sensitivity of isolates of MRSA to anti-staphylococcal β-lactams, two phenotypes became apparent; those that became susceptible on bicarbonate supplementation and those that were unaffected. In the isolates that became susceptible, bicarbonate supplementation caused reduced expression of mecA and sarA, which led to decreased production of penicillin-binding protein 2a and correlated with sensitivity to β-lactams in a rabbit infective endocarditis model comparable to that of MSSA isolates. Additionally, bicarbonate responsive isolates demonstrated lower survival when the β-lactam was combined with LL-37 in vitro and this may have enhanced the efficacy seen in vivo (Ersoy et al., 2019).
Dorschner and colleagues observed inhibition of S. aureus growth by LL-37 was greater in MEM, a cell culture medium, when compared with Tryptone Soy Broth (a nutrient-rich bacterial growth medium) containing the same concentrations of NaCl and FBS (Dorschner et al., 2006). By analysis of individual components of MEM, they determined that it was the presence of physiological concentrations of sodium bicarbonate that enhanced membrane-permeabilising activity of the following AMP: LL-37, mCRAMP (murine cathelicidin-related antimicrobial peptide), PR-39 (a porcine cathelicidin), hBD-2 (human β-defensin 2), but not dermcidin (an anionic AMP from human skin and sweat) and not in a pH-dependent manner. The presence of bicarbonate; may also have ameliorated the inhibitory effect of the 150 mM NaCl in the medium used, as NaCl concentrations of >50 mM can inhibit the activity of many AMP (Goldman et al., 1997; Travis et al., 2000). Culturing E. coli O29 in the presence of NaHCO3 affected the expression of a number of virulence-related genes that could increase susceptibility to AMP (Dorschner et al., 2006). The addition of 25 mM bicarbonate enhanced the activity of the AMP tritrpticin against the protozoan Trichomonas vaginalis (Infante et al., 2011). Conversely, selected S. aureus small-colony variants (SCV) were less susceptible to LL-37 when incubated in the presence of 50 mM NaHCO3; (~2 x physiological concentration) (Zhang et al., 2018), although the effect of physiological bicarbonate concentrations was not examined. Small colony variants (SCV) are slow-growing sub-populations of bacteria with altered metabolism and reduced antibiotic susceptibility which, in the case of S. aureus, can cause persisting and recurrent infections (Baumert et al., 2002). S. aureus SCV are already known to be less susceptible to a number of AMP when compared to wild-type cells (Koo et al., 1996; Sadowska et al., 2002; Samuelsen et al., 2005), although this effect was not observed when a cationic antimicrobial polypeptide was tested against S. aureus SCV (Mercer et al., 2017). It would therefore be relevant to examine the effects of media supplementation with physiological concentrations of bicarbonate (25 mM; 2.1 g/L) when conducting AST with AMP to generate results possibly more predictive of efficacy in vivo. Such results must be viewed with caution, however, in light of the biofilm promoting effects in combination with tobramycin (Kaushik et al., 2016).
Temperature
The effect of temperature on the activity of AMP during AST has not been widely investigated, as most AMP are intended for use against infectious diseases AST is predominantly conducted at body temperature (~37°C) or at temperatures recommended in CLSI or EUCAST guidelines (30–37°C). Thus, it seems most relevant to conduct AST at physiological temperatures. At low temperatures membrane bilayers undergo a reversible change of state from a fluid (disordered, liquid crystalline) to a non-fluid (ordered, gel) array of the fatty acyl chains (de Mendoza, 2014) and this increase in membrane rigidity can lead to reduced AMP efficacy/resistance (Cole and Nizet, 2016; Joo et al., 2016). Interestingly, the P. aeruginosa quorum-sensing molecule 2-n-heptyl-4-hydroxyquinoline N-oxide (HQNO) increases membrane fluidity in S. aureus, so it would be intriguing to determine whether this increases the sensitivity of S. aureus to AMP (Orazi et al., 2019). HQNO can also induce S. aureus to adopt the SCV phenotype (Hoffman et al., 2006) and this may also affect AMP activity. The effect of high and low temperatures on storage or preparation of AMP, as well as activity, has received consideration in a number of studies (Wei et al., 2007; Zhang et al., 2011; Ji et al., 2014; Lee et al., 2014; Jiao et al., 2019). For example, many AMP demonstrate stability at temperatures below 100°C. For example, the thermal stability of 8 AMP (Cap18, Cap11, Cap-11-1-18m2, Cecropin B, Cecropin P1, Indolicidin and Sub5) was assessed by heating them to 70 or 90°C for up to 30 min before conducting AST. All AMP were stable following heating, with only Sub5 demonstrating an MIC increase (4 to 8 mg/L) after being heated to 70°C for 30 min, but not when heated to 90°C (Ebbensgaard et al., 2015). However, this is not the case for all AMP. The AMP epinecidin-1 was not stable at elevated temperatures and demonstrated a 32 - >64-fold increase in MIC against S. aureus following incubation at 60–100°C for 5 min (Huang et al., 2017). As is well-documented, AMP have been isolated from almost all life-forms, including arctic and Antarctic fish. Moronecidin (isolated from the hybrid striped bass) and 2 derivatives were assessed for efficacy against Psychrobacter spp. PAMC25501 at 5–15°C and E. coli DH5α at 15–37°C and no differences in MIC were observed at different temperatures (Shin et al., 2017). The activity of the piscidin-like AMP, chionodracine (isolated from the icefish Chiondraco hamatus) was more active at 25 than 37°C against E. coli (MIC 20 and 5 mg/L, respectively) and B. cereus (MIC 10 and 5 mg/L, respectively) (Buonocore et al., 2012), although this may reflect adaptation to the low temperature environment from which they were isolated.
Metal Ions
Transition metal ions influence the activity of AMP in a variety of ways. In some cases, the activity of AMP are dependent on the presence of metal ions (Paulmann et al., 2012; Melino et al., 2014; Alexander et al., 2018; Jezowska-Bojczuk and Stokowa-Soltys, 2018; Agbale et al., 2019), whereas in others the presence of metal ions can cause reduction, or even complete abrogation, of activity (Friedrich et al., 1999; Deslouches et al., 2005). In most circumstances, the effect of metal ions must be considered on a case-by-case basis as different ions will produce a distinct effect. For example, LL-37 is inactive in the presence of ≥3 μM MgCl2, whereas the de novo designed AMP, WLBU2, remains active at the same concentration. LL-37 is also less potent in the presence of ≥1 μM CaCl2, whereas the MIC of WLBU2 increases by ~4-fold in the presence of ≥6 μM CaCl2, (Deslouches et al., 2005). Activity of the cecropin-melittin hybrid peptide variants was ≥4-fold increased in the presence of 3–5 mM MgCl2 (representing the serum concentration of divalent cations), although one variant is insensitive to the effects of 3 mM MgCl2, but not to 5 mM MgCl2 (Friedrich et al., 1999).
Susceptibility testing of K. pneumoniae against tetracycline in tissue culture medium predicts resistance, whereas testing in MHB and LPM pH 5.5 media predicts susceptibility. Mice infected with K. pneumoniae and treated with tetracycline survive infection mirroring the results obtained in pH 5.5 media, conditions that resemble best the environment in which tetracycline interacts with this pathogen since K. pneumoniae resides within macrophage phagosomes (Ersoy et al., 2017). This example highlights the importance of mimicking the environment of phagosomes when dealing with intracellular pathogens. Besides K. pneumoniae, other important pathogens that thrive in an intracellular environment include Salmonella spp. (de Jong et al., 2012; Helaine et al., 2014), Mycobacterium tuberculosis (Pieters, 2008) and Legionella pneumophila (Escoll et al., 2013). Streptococcus pyogenes is also suspected of sharing this lifestyle (Hertzen et al., 2012).
After phagosomes internalize their cargo, acidification of the interior takes place in a rapid process. For example, the phagocytic compartment of a bone marrow-derived macrophage phagosome following internalization of an immunoglobulin G-coated particle reaches a pH of 5.0 or below within 10–12 min of internalization of an immunoglobulin G (IgG)-coated particle (Yates et al., 2005). The acidic, hydrolytically competent environment of the phagolysosome in combination with other antimicrobial effectors typically lead to the death and digestion of most non-pathogenic microbes. It is in this environment that the intracellular pathogens mentioned above survive and thrive, and more importantly, it is where the interaction between an antimicrobial agent and the pathogen will take place. Besides the low pH, other antimicrobial effectors fill the phagocytic compartment, including metal ions.
Metal ions such as copper and zinc have been observed at high concentrations in the phagocytic milieu as a response to certain types of infections. Wagner et al. using a hard x-ray microprobe with suboptical resolution reported that upon infection by the human pathogens M. tuberculosis and M. avium or with avirulent M. smegmatis the concentration of copper and zinc ions within phagolysosomes of peritoneal macrophages can be as high as 426 ± 393 μM and 459 ± 271 μM, respectively (Table 3) (Wagner et al., 2005). Additional indirect evidence for the presence of copper ions in the intracellular battlefield against pathogens include the observation that in IFN-γ and LPS-activated macrophages the levels of the Ctr1 Cu importer are elevated (White et al., 2009). Moreover, the Cu pump ATP7A is overexpressed and localized to the phagolysosome, suggesting accumulation of Cu within this compartment. Interestingly, macrophage exposure to the Cu chelator tetrathiomolybdate (TTM) results in increased survival of S. typhimurium (Achard et al., 2012). Besides the quantitative determination of Zn by Wagner and co-workers, there is additional evidence for a role of host Zn in the direct overload killing of invading pathogens (Djoko et al., 2015). Using a Zn-responsive fluorescent probe, it was observed that infection of human macrophages with either E. coli or M. tuberculosis leads to an increase in the intracellular levels of Zn (Botella et al., 2011). Consistent with the hypothesis that Zn directly kills phagocytosed pathogens, it was observed that bacterial mutants defective in Zn export (zntA and ctpC in E. coli and M. tuberculosis, respectively) showed decrease survival within these human macrophages. Similar observations of increased Zn concentrations within phagocytic cells have been observed upon infection with Histoplasma capsulatum (Subramanian Vignesh et al., 2013), and Streptococcus pyogenes (Ong et al., 2015). As observed for E. coli and M. tuberculosis, S. pyogenes mutants defective in Zn efflux had a lower survival within the hosts.
Table 3.
Concentration of copper and zinc ions within phagolysosomes of peritoneal macrophages during infection by three Mycobacterium spp.
| Element | Time | M. smegmatis | M. avium | M. tuberculosis |
|---|---|---|---|---|
| Cu | 1 h | 9.9 ± 5.5 μM | 28.3 ± 11.4 μM | 426 ± 393 μM |
| 24 h | 24.8 ± 0.65 μM | 17.3 ± 10.3 μM | 24.7 ± 9.5 μM | |
| Zn | 1 h | 70.5 ± 37.3 μM | 134.6 ± 38.8 μM | 37.8 ± 25.2 μM |
| 24 h | 260 ± 117 μM | 120.8 ± 31.1 μM | 459 ± 271 μM |
Concentrations were determined using hard x-ray microprobe with sub-optical resolution (Wagner et al., 2005).
There is also evidence for removal of Zn ions from phagosomes during infections and excellent reviews on the topic exist. In this work, we wanted to limit ourselves to those environments in which the concentration of Zn ions increased.
Besides the phagocytic environment, there are other sites of microbial infection in which copper and zinc ions are found at concentrations in which they can affect the activity of antimicrobial agents. For instance, during urinary tract infection by the pathogens Proteus mirabilis and K. pneumoniae, copper is found at micromolar concentration as a host response to the infection (Hyre et al., 2017). Additional in vivo studies demonstrated that Cu-deficient mice are more susceptible to uropathogenic E. coli infection, indicating that copper release into urine is an important innate defence mechanism. An additional human fluid that contains metal ions at concentrations high enough to affect the antimicrobial activity of antibiotics is sweat (Troy et al., 2007). Copper concentrations range from 4.6 ± 0.4 μM to 20 ± 10 μM, whereas zinc concentrations can be as high as 630 μM. Copper levels in human saliva also have been reported to range from 1.6 μM to 7.5 μM (Dreizen et al., 1952; Borello, 1976). Human saliva also contains zinc ions with reports indicating a maximum concentration of 6.7 mM (Sejdini et al., 2018). Clearly, copper and zinc ion interactions with antimicrobial agents is plausible outside phagocytic compartments.
Antibiotics, with their richness of functional groups, are poised for metal ion coordination. The result of this interaction can range from antagonism to synergism, although the former is the most common outcome. Back in 1946, Eisner et al. reported the inactivation of penicillin by zinc salts (sulphate, acetate, chloride, and oxide) (Eisner and Porzecanski, 1946). Amoxicillin and ampicillin are also readily degraded by zinc ions (Navarro et al., 2003). Tetracycline and several of its derivatives avidly bind copper and zinc ions to form 2:1 complexes (Doluisio and Martin, 1963; Brion et al., 1985). Indeed, the formation of tetracycline-zinc complexes is suspected to affect the metabolism of the drug and adversely impact its antibiotic activity (Doluisio and Martin, 1963; Brion et al., 1985). Other antibiotics are known to bind metal ions (e.g., quinolones Seedher and Agarwal, 2010; Uivarosi, 2013 and aminoglycosides Lesniak et al., 2003). Interestingly, many chelates of quinolones show equal or enhanced activity compared to that of the parent antibiotic. The reason for the superior activity of the quinolone-metal complexes is not clear. Overall, the impact of metal binding on the activity of many antibiotics is undeniable and deserves attention during susceptibility assays.
The effect on the antimicrobial activity of AMP of the presence of Cu and Zn ions in the assay media is difficult to predict. Typically, compounds that act as simple metal chelators will see a decrease in their antimicrobial activity as the concentration of chelated ions increase. An example of an antimicrobial peptide that acts as a metal chelator is the tick peptide microplusin, which specifically chelates Cu2+ ions (Figure 2). The opposite effect, that is the increase in activity in the presence of these metal ions, also takes place although it has received less attention. In the case of copper ions, several antimicrobial peptides have been recently reported to utilize this ion to enhance its bactericidal activity (Libardo et al., 2014, 2015, 2016, 2017; Hayden et al., 2015; Alexander et al., 2017). One of the earliest examples of the AMP requirement for Zn2+ is that of bacitracin, a mixture of chemically related dodecapeptides isolated from Bacillus subtilis and Bacillus licheniformis. Bacitracin, a drug and food additive, has a multimodal mechanism of action (Storm and Strominger, 1973; Storm, 1974; Karala and Ruddock, 2010; Dickerhof et al., 2011). There is consensus in that the main mode of action involves the inhibition of bacterial cell-wall biosynthesis by targeting extracellular, membrane-associated pyrophosphate groups. Remarkably, recognition of the pyrophosphate group of the lipid target is mediated by a zinc ion which organizes the N-terminal region of the peptide and neutralizes the large charge of the ligand, thus allowing direct antibiotic–lipid interactions (Storm and Strominger, 1973; Economou et al., 2013). Other peptides with enhanced its antimicrobial activity in the presence of Zn2+are the anionic human AMP DCD-1 and its derivative DCD-1L. Both peptides are present in eccrine sweat and are highly active at pH values that range from 5.5 to 7.4 and retain their activities against several microorganisms when the ionic strength of the medium is increased by adding up to 150 mM NaCl (Dennison et al., 2006; Song et al., 2013; Becucci et al., 2014; Libardo and Angeles-Boza, 2014). The Zn2+ ions are required for the formation of ion channels as they promote the formation of a trimer of dimers. One final example that is worth mentioning corresponds to the synthetic *ARVA peptides, which contain no histidines, that were selected from a combinatorial library and shown to be synergistic with copper and zinc ions (Walkenhorst et al., 2014; Walkenhorst, 2016). Thus, there is mounting evidence that the activity of AMP is affected by the presence of copper and zinc ions.
Figure 2.
Structure of microplusin solved by NMR solution spectroscopy (PDB ID: 2KNJ). For illustration purposes the Cu2+ ion has been added, shown as brown sphere, to its putative binding site. The four labelled residues, shown as red liquorice, are highly likely to bind Cu2+ ion due to their proximity to the ion. The other residues, shown as blue liquorice, are also capable of binding Cu ion, as suggested by the NMR experiment (Silva et al., 2009).
An important question that must be asked at this point is whether the presence of other divalent cations would resemble the effect of Cu and Zn ions? The short answer is no, as the effects on the activity of AMP by Ca2+ and Mg2+ ions often differ from those effects that originate from the presence of Cu and Zn ions. For example, the presence of CaCl2 and MgCl2 does not affect the activity of microplusin; however, similar concentrations of CuCl2 have a negative impact in the activity of the AMP (Silva et al., 2009). Similarly, the presence of Ca2+ ions decrease the antimicrobial activity of clavanin A, an AMP found in phagocytic cells, whereas Zn2+ ions increased its activity (Juliano et al., 2017). In short, Ca2+ and Mg2+ ions do not have the same effect as Cu and Zn ions in the antimicrobial activity of AMP.
Excluding the highest and lowest values of the metal concentrations in the set of values shown in Table 3, the average concentrations of Cu and Zn ions are 24 and 146 μM, respectively; therefore, we recommend using Cu and Zn concentrations of 25 and 150 μM, respectively if the phagosomal environment is to be mimicked. [Zn2+] = 150 μM is well above the concentration required to maximize the potency of clavanin A 16-fold at pH 5.5 (Juliano et al., 2017). Likewise, 150 μM is in the upper range of the Zn concentrations that were synergistic with *ARVA peptides (Walkenhorst et al., 2014). Since Ersoy has successfully used LPM (low magnesium medium) media at pH 5.5 to mimic the phagolysosome compartment, it is suggested that using this media in combination with Cu and Zn ions. CuCl2 and ZnCl2 can be used as sources for these ions. If after performing the assay in the presence of copper and zinc ions as suggested above one is suspicious of synergy between any of these metal ions and the AMP, the best assay to follow is the one described by Walkenhorst et al. which allows the determination of FIC values using a 96-well plate. CuCl2 and ZnCl2 were used in liquid test medium (LTM) and the procedure is clearly described in the paper of Walkenhorst and co-workers (Walkenhorst et al., 2014).
Salt (NaCl)
The effect of NaCl on AMP activity needs to be taken into account depending on the site of infection as the concentration can vary significantly, from ~140 mM in blood (Li et al., 2016), 12–52 mM in saliva (Ferguson and Botchway, 1979) and 8–14 mmol/mmol creatinine in urine (Sasaki et al., 1998), whereas in certain infectious disease states this can be much higher (up to 180 mM in CF sputum Grandjean Lapierre et al., 2017 and 300 mM in infected urine Withman et al., 2013). Distinct from other metal ions and salts, some AMP are sensitive to higher salt concentrations. For example, human β-defensin 1 retains activity at low NaCl concentrations (≤50 mM), but this is rapidly lost when the NaCl concentration increases above 75 mM (Goldman et al., 1997), which are physiologically relevant (Wishart et al., 2018). This sensitivity was to the Na+ ion, not the Cl− ion. Similarly, the activity of LL-37 is reduced significantly in the presence of 100 mM NaCl (Turner et al., 1998) and the activities of defensins are similarly reduced (Bals et al., 1998; Garcia et al., 2001). The tryptophan-rich AMP, Pac-525, demonstrated reduced activity in the presence of 100–300 mM NaCl, whereas the Pac-525 derivative, d-Nal-Pac-525 (all d-amino acids and tryptophan residues replaced with d-β-naphthylalanine) was not sensitive to these NaCl concentrations (Wang et al., 2009). The histatin derivative P-113 was also sensitive to high NaCl concentrations (as well as additional Ca2+ and Mg2+ in the media) (Rothstein et al., 2001). Conversely, clavanin retained activity in 100 mM NaCl and Clavanin AK retained activity in 300 mM NaCl (Lee et al., 1997), the de novo designed peptide SHAP1 retained activity in the presence of 200 mM NaCl (Kim et al., 2014), the Streptococcus mutans-specific AMP IMB-2 retained ~85% activity in the presence of 134 mM NaCl (Mai et al., 2011) and the RR12 AMP retained activity in 300 mM NaCl (Mohanram and Bhattacharjya, 2016). Therefore, when considering AST of AMP, the concentration of NaCl at the site of infection is an important factor and the effects of NaCl on AMP activity will need to be assessed on a case-by-case basis. If the tested AMP is salt-sensitive, there are a number of possible strategies available to reduce salt-sensitivity (Harwig et al., 1996; Friedrich et al., 1999; Tam et al., 2000; Park et al., 2004; Yu et al., 2011; Chu et al., 2013).
Inoculum Size
The efficacy of an antimicrobial during AST will necessarily depend on the size of inoculum tested. The inoculum effect is a well-documented laboratory phenomenon that can be described as a significant elevation in antibiotic MIC when the number of inoculated microorganisms is increased (Brook, 1989; Smith and Kirby, 2018; Idelevich and Becker, 2019). Elevated MIC (reduced efficacy) values as a function of increased cell density has been observed for a limited number of AMP, including LL-37 (Snoussi et al., 2018), ARVA (Starr et al., 2016) and DNS-PMAP 23 (Savini et al., 2017). In cultures with a low inoculum density, MICs of AMP and antibiotics can reach a plateau value that is independent of cell density (Udekwu et al., 2009; Savini et al., 2017).
An additional consideration must be the presence of host cells as these can effectively contribute to the inoculum effect. For example, conducting AST in the presence of host cells, e.g., red blood cells, can cause a loss of activity against the target pathogen due to binding of the AMP to these host cells, even if selectivity is lower than for the pathogen (Starr et al., 2016). However, bactericidal concentrations of AMP can be achieved in the presence of host cells (Savini et al., 2017) and this should also be possible clinically. Thus, when conducting AST of AMP the size microbial inoculum (cfu/ml) must be given due consideration and a balance reached between low cell density resulting in efficacy that is unachievable in vivo and high cell density that results in under-estimation of activity and the potential loss of promising therapeutic candidates. As a starting point the cell densities specified in CLSI, ISO and EUCAST documents (Table 1) are not unreasonable starting points, but consideration should also be given to the pathogen and site of infection, as in vivo information may be available to better guide the likely cell density during infection and this would clearly be more relevant to take into consideration.
Growth Phase: Biofilms, Persister Cells Spores and Small Colony Variants (SCV)
When conducting broth dilution AST using CLSI or EUCAST protocols, bacteria are normally prepared when actively growing (exponential phase) or in the stationary phase, depending on whether the inoculum is prepared by the broth culture or colony suspension methods, respectively (CLSI, 2018a; ISO, 2019), but the potential influence of growth phase on AST results is not taken into consideration. Similarly, the growth phase is not factored in when preparing yeast inocula (CLSI, 2017a). Conversely, AST of filamentous fungi starts with an inoculum prepared from a spore suspension (CLSI, 2017c) that can take a number of hours to germinate into actively growing hyphae. In most cases, AST of conventional antibiotics and antifungals requires actively metabolising/growing cells, as their mechanism of action relies on physiological processes, including DNA/RNA replication (fluoroquinolones and flucytosine), protein biosynthesis (tetracyclines and macrolides), cell wall biosynthesis (β-lactams and echinocandins) and membrane biosynthesis (polyenes and azoles) (Silver, 2011; Perfect, 2017). Therefore, it is important that microorganisms are actively growing, or have the capacity to do so, when AST is performed. Membrane-active AMP, on the other hand, are active against metabolically inactive as well as active cells; a major potential therapeutic advantage, e.g., (Mercer et al., 2017). As such, it is not critical that cells be metabolically active when AST is conducted for these AMP, and it is only by following convention than most AST of AMP is done on actively metabolising cells. At sites of infection, microorganisms are rarely found growing in the exponential phase on nutrient-rich media sources; therefore, the importance of microbes existing in other growth phases requires consideration and there is evidence that slower growing pathogens are more virulent than faster growing pathogens (Leggett et al., 2017). Additionally, a number of different parameters influencing growth phase can alter virulence gene expression in pathogens, including toxin and adhesin production in E. coli (Crofts et al., 2018) and hyphal development in Candida spp. (Su et al., 2018).
Given the heterogeneity of microbial populations within an infection site, a range of cell morphologies and environmental conditions will exist and these will have an impact on antimicrobial susceptibility. Given this heterogeneity, it is relevant to test antimicrobial efficacy, including that of AMP, vs. microbes existing in different growth states (Radlinski and Conlon, 2018; Dewachter et al., 2019). Specialised slow or non-growing forms of microbial pathogens include biofilms (Desai et al., 2014; Reichhardt et al., 2016; Koo et al., 2017; Wolfmeier et al., 2018; Orazi and O'Toole, 2019), spores (Setlow, 2014; Gil et al., 2017), persister cells (Fisher et al., 2017; Wuyts et al., 2018; Balaban et al., 2019) and small colony variants (SCV) (Proctor et al., 2006; Johns et al., 2015). In some cases, there are even combinations of these cells, e.g., persister cells and/or SCV within biofilms (Mirani et al., 2015; Waters et al., 2016; Wuyts et al., 2018; Yan and Bassler, 2019). A common feature of microbial growth in these forms is normally reduced susceptibility to antibiotics and antifungals. The efficacy of AMP has been tested against cells demonstrating differing growth modes.
At the simplest level, a microbial biofilm is a surface-associated community of microorganisms surrounded by an extracellular polymeric matrix. It is estimated that more than 80% of microbial infections are caused by microbes growing as biofilms (Romling and Balsalobre, 2012), and therefore their determination of their susceptibility to antimicrobial agents, including AMP, is of paramount importance when developing novel antimicrobial agents. An increasing understanding of biofilm infections has led to the appreciation that many infections are polymicrobial in nature and may contain diverse species of bacteria, fungi and viruses (Peters et al., 2012; Wolcott et al., 2013; Mihai et al., 2015; Todd and Peters, 2019) and a number of models of polymicrobial biofilms have recently been developed (Gabrilska and Rumbaugh, 2015). Polymicrobial biofilms are of relevance in many infections, including chronic wound infections (Clinton and Carter, 2015), CF lung infections (Lopes et al., 2015), bacterial vaginosis (Jung et al., 2017) and medical device-associated infections (Wi and Patel, 2018). Microbes growing in biofilms can be up to 1000-fold more tolerant to antimicrobials than their planktonically growing counterparts (Costerton et al., 1995; Hoiby et al., 2010), so more effective therapeutic options are urgently required. A number of mechanisms are responsible for biofilm antibiotic-tolerance including; (1) reduced diffusion of antibiotics through the biofilm matrix, (2) sequestration of antibiotics by the biofilm matrix; (3) presence of slow-growing and persister cells (see below) refractory to antibiotics targeting bacterial metabolism; and (4) increased exchange of antibiotic resistance genes on mobile genetic elements by cells in close proximity (Stewart and Costerton, 2001; Hall and Mah, 2017; Orazi and O'Toole, 2019). The potential role of AMP as therapeutics to treat biofilm infections has received significantly more attention than activity vs. spores, persister cells or SCV and has been the subject of several review articles in the last few years (Batoni et al., 2016; de la Fuente-Nunez et al., 2016; Pletzer and Hancock, 2016; Delattin et al., 2017; Grassi et al., 2017b; van Dijck et al., 2018; Von Borowski et al., 2018; Yasir et al., 2018; Shahrour et al., 2019). A detailed analysis of the potential role of AMP in the treatment of biofilm infections is beyond the scope of this manuscript, but some key points with respect to AST will be addressed in the following paragraphs. Membrane disruption is the most common mechanism of action of cationic AMP and remains important in some instances for anti-biofilm properties of AMP, but when assessing anti-biofilm properties, other attributes of AMP may be of equal or greater importance (Yasir et al., 2018), including; (1) blocking of/interference with bacterial cell signalling systems, e.g., LL-37 (Overhage et al., 2008); (2) degradation of the biofilm matrix, e.g., piscidin-3 (Libardo et al., 2017); (3) inhibition of the alarmone system to avoid the bacterial stringent response, e.g., peptide 1018 (de la Fuente-Nunez et al., 2014); (4) downregulation of genes responsible for biofilm formation, e.g., hBD-3 (Zhu et al., 2013); and (5) immunomodulatory properties may also confer additional in vivo anti-biofilm properties. Interestingly, some AMP are able to prevent biofilm formation at concentrations significantly below their MIC, or have anti-biofilm properties, but without antibacterial activity (for examples see (Pletzer and Hancock, 2016; Mercer et al., 2017; Haney et al., 2018a; Shahrour et al., 2019), indicating a separate mechanism of action from membrane disruption/direct killing. The membrane disruption properties of AMP have been the main focus of this manuscript and will remain so for the discussion of AST of AMP and biofilms.
Examples of AMP that can kill biofilm microbes by membrane permeabilization include the eosinophil cationic protein-derived AMP, RN3(5-17P22-36), that demonstrated membrane-permeabilising activity (Sytox green membrane permeabilization) against established biofilms of P. aeruginosa, albeit at 2–8-fold higher concentrations than required for the same activity vs. planktonic P. aeruginosa (Pulido et al., 2016). The esculentin-1a-derived AMP, Esc(1-21), also caused permeabilization of the membranes of planktonic cells and biofilms of P. aeruginosa PAO1 (Sytox green membrane permeabilization and β-galactosidase release) (Luca et al., 2013), and LL-37 and selected truncated versions (LL-31, LL7-37, LL13-37, and LL7-31), albeit at relatively high concentrations (20–100 μM) (measured by propidium iodide (PI) uptake) against pre-grown P. aeruginosa PAO1 biofilms (Nagant et al., 2012). The lactoferricin-derived AMP, LF11-215, LF11-324 and a lipopeptide derivative, DI-MB-LF11-324, caused membrane permeabilization of P. aeruginosa PAO1 in biofilms (PI uptake) at 10 × MIC concentrations (Sanchez-Gomez et al., 2015). The related AMP Seg6L and Seg6D both demonstrated antibiofilm properties with Seg6D predominantly causing cell lysis, whereas Seg6L degraded the biofilm by detaching live cells, rather than direct killing, demonstrating that relatively minor changes to AMP composition (substitution of 5 Seg6L amino acids for D-isoform amino acids) can substantially affect antimicrobial properties (Segev-Zarko et al., 2015). Some AMP are also membrane-active against fungal biofilms (van Dijck et al., 2018). Tyrocidines are cationic cyclodecapeptides with broad-spectrum antimicrobial activity and the tyrocidines (TrcA, TrcB, and TrcC) were able to disrupt the membrane of Candida albicans growing as biofilms (PI uptake) at concentrations similar to the planktonic MIC (~12.5 μM), but significantly higher concentrations were required to eradicate biofilms (Troskie et al., 2014). Similarly, the peptidomimetic mPE was able to disrupt the membrane of Candida albicans growing as biofilms (PI uptake) at concentrations 6–12-fold higher (50–100 mg/L) than the MIC vs. planktonic cells (Hua et al., 2010). Studies of the activity of AMP vs. polymicrobial biofilms are less frequently reported, but the Herpes simplex-derived AMP gH625-GCGKKKK was able to prevent formation of mixed species biofilms consisting of Candida tropicalis and S. aureus or C. tropicalis and Serratia marcescens as well as eradicating mixed biofilms containing these microbes (de Alteriis et al., 2018) as did melittin against a polymicrobial biofilm consisting of selected dairy isolates of bacteria (P. aeruginosa, Staphylococcus haemolyticus, K. pneumoniae, and Aeromonas caviae) (Galdiero et al., 2019). The de novo AMP ASP-1 was able to eradicate polymicrobial biofilms containing S, aureus, P. aeruginosa, A. baumannii, and K. pneumoniae when formulated into a polyurethane-based dressing (Bayramov et al., 2018) and the Komodo dragon-derived peptide DRGN-1 demonstrated moderate eradication activity against mixed biofilms containing P. aeruginosa and S. aureus (Chung et al., 2017). Luo and co-workers tested a library of peptoids for antibiofilm activity and demonstrated that 3 peptoids from the library were able to eradicate mixed species biofilms (formed for 8 h) containing C. albicans and S. aureus or C. albicans and E. coli (Luo et al., 2017).
In general, concentrations of AMP required to permeabilise membranes in biofilms are higher than for their planktonic counterparts and this may reflect sequestration (for example, by extracellular DNA or other extracellular polymeric substances), hydrolysis of AMP in the biofilm, increased microbial cell density or may reflect changes to individual microorganisms residing within the biofilm, as bacteria growing in biofilms can have altered membrane permeability (Orazi and O'Toole, 2019), which may affect the activity of membrane-active AMP. Membranes of S. aureus, Listeria monocytogenes, P. aeruginosa, Salmonella Typhimurium growing as biofilms contain significantly higher proportions of saturated fatty acids compared to cells growing planktonically. Increases in saturated fatty acids (in particular long-chain fatty acids are concomitant to decreases in branched-chain fatty acid content, which can lead to increased membrane rigidity and stability (Denich et al., 2003; Dubois-Brissonnet et al., 2016).
Approved standards exist for AST of both bacteria and fungi (Table 1), but these apply only to microbes growing planktonically. Approved AST standards for microorganisms growing as biofilms do not exist and a number of different methods are used (Macia et al., 2014; Magana et al., 2018), making direct comparisons of different studies difficult to undertake. As described above, microorganisms growing as biofilms are more tolerant of antimicrobials than microbes growing planktonically, so the results of conventional AST cannot be used to accurately predict the results of biofilm AST (Macia et al., 2014). As an added level of complexity, the level of biofilm antimicrobial tolerance may also be influenced by the methods used to establish and monitor the biofilms.
At present it is not possible to specify an optimal testing procedure for assessing the activity of AMP against biofilms, given their heterogenous nature and the complexity underlying their development (Haney et al., 2018a; Magana et al., 2018). When reporting activity of AMP vs. biofilms it is vital, initially, to specify whether biofilm prevention or eradication is being described. When describing the anti-biofilm properties of AMP appropriate methodological details must be provided, including device used to grow the biofilm and the parameters used to establish anti-biofilm activity. Most commonly, biofilms are grown in multi-well plates or the Calgary device, which are simpler and cheaper than flow-cell devices and better suited to routine AST of biofilms (Macia et al., 2014). The most commonly used are the minimal biofilm inhibitory concentration (MBIC), defined as the lowest concentration of an antimicrobial at which there is no time-dependent increase in the mean number of biofilm viable cells (Moskowitz et al., 2004), or the minimal biofilm-eradication concentration (MBEC), defined as the lowest concentration of antibiotic required to eradicate the biofilm (Ceri et al., 1999). In the case of membrane-active AMP, evidence of membrane permeabilization must be provided, for example, the use of fluorescence microscopy (standard, confocal laser scanning microscopy or scanning electron microscopy) using membrane-impermeant fluorophores that allow demonstration of membrane permeabilization, including PI (Boulos et al., 1999) or Sytox dyes (Roth et al., 1997) or reporter genes. Recently, Haney and co-workers proposed two quick, easy, reproducible and inexpensive methods; one for assessing biofilm inhibition and the other for assessing biofilm eradication, to determine the activity of AMP against biofilms (Haney et al., 2018b). Both methods were based on the use of microtitre plates for detection and utilised the crystal violet protocol for determining biofilm biomass (O'Toole and Kolter, 1998; O'Toole, 2011) and in the eradication assay they assessed metabolic activity within the biofilm using the tetrazolium chloride dye (triphenyl tetrazolium chloride) (Brown et al., 2013; Sabaeifard et al., 2014). These methods are similar to many reported in the literature and appear suitable for simplified analysis of biofilm prevention and eradication and should also be readily adapted for use in the analysis of fungal biofilms, but do not provide an assessment of the effects of AMP on the bacterial membrane, which would require use of some of the methods outlined above.
The efficacy of AMP vs. bacterial and fungal spores has been the subject of a relatively limited number of studies. Bacterial endospores with relevance to human infectious diseases are mainly produced by Clostridiodes spp. (Clostridium spp.) and Bacillus spp. Bacterial endospores are metabolically dormant and environmentally resistant and are capable of surviving antibiotic exposure, extremes of temperature, desiccation and ionizing radiation (Higgins and Dworkin, 2012). The cathelicidin family AMP PG-1, BMAP28, and LL-37 demonstrated sporicidal activity against B. anthracis spores in the low mg/L range, whereas SMAP-29, CAP-18 were not effective and NA-CATH and mCRAMP demonstrated activity only at high concentrations (1,000 mg/L) (Blower et al., 2018). The AMP chrysophin-3 also demonstrated sporicidal activity vs. B. anthracis (Pinzon-Arango et al., 2013) and activity of PG-1 vs. spores of B. anthracis had been previously reported (Lisanby et al., 2008). The AMP TC19, TC84, BP2 and the lantibiotic nisin A were tested for efficacy against spores of Bacillus subtilis and were bactericidal against germinated spores by perturbing the inner membrane, thus preventing outgrowth to vegetative cells, but were not able to prevent germination (Omardien et al., 2018). Presumably, this is because the inner membrane of endospores is immobile and becomes fluid only during spore germination (Setlow, 2006). Other than this, the only peptides with activity vs. spores are the lantibiotics nisin (Gut et al., 2008) and subtilin (Liu and Hansen, 1993), both of which were only active against germinated spores, as above. Assessing the efficacy of AMP vs. bacterial spores for AST is relatively simple, by substituting a spore suspension for the normal inoculum, but in order to fully understand the precise effect of AMP on spores requires more specialised techniques, such as those described by Omardien et al. (2018). As spores are not normally the cause of the disease, rather this is vegetatively growing cells, the importance of AMP activity against spores is of more relevance to disease prevention, rather than cure.
Activity specifically against fungal spores (conidia) has received limited attention compared to activity against vegetatively growing fungi or yeast. Fungal spores are often the infectious propagule that initiates disease, e.g., inhalation of Aspergillus spp. spores into the lung, but the disease is caused once the spores germinate and the fungi begin vegetative growth (Kosmidis and Denning, 2015). As with efficacy vs. bacterial endospores, sporicidal activity is of more relevance to disease prevention than cure. Nevertheless, activity against vegetatively growing cells and spores is a desirable property for any antifungal AMP. The thaumatin-like protein osmotin demonstrated antifungal activity against fungal hyphae of predominantly plant-pathogenic fungi and caused lysis of spores of Fusarium moniliforme, F. oxysporum, Trichoderma longibrachatum, and Verticillium dahlaie (Abad et al., 1996) as did the synthetic undecapeptides Pep3, BP15, BP20, BP33, and BP76 vs. F. oxysporum conidia (Badosa et al., 2009). The antifungal peptide drosomycin, originally isolated from Drosophila spp., inhibited fungal spore germination (Zhang and Zhu, 2009) as did bombinins H2 and H4 which were active against spores of Phytophthora nicotianae (Matejuk et al., 2010). Peptide KK14 and derivatives inhibited spore germination of F. culmorum, Penicillium expansum, and A. niger, albeit with reduced activity vs. germinated conidia (Thery et al., 2019) as did the AMP O3TR and derivatives (Thery et al., 2018). Rabbit neutrophil cationic peptides, however, were not active against ungerminated spores of Aspergillus fumigatus or Rhizopus oryzae (Levitz et al., 1986). When carrying out broth dilution AST using filamentous fungi, the inoculum used is a spore (conidial) suspension, but no attempt is made to determine whether the antifungal used prevents spore germination or is solely active against germinated spores/hyphae. The measure of inhibition is optical density or a visual (i.e., not using microscopy) determination of growth/no growth (CLSI, 2017c; EUCAST, 2017b), except in the case of echinocandins using EUCAST methodology in which the Minimum Effective Concentration (MEC) endpoint is determined (EUCAST, 2017b). Thus, it is clear that echinocandins do not prevent spore germination (i.e., not sporicidal), but this cannot be determined for other antifungals, including AMP, without additional analyses, such as microscopic analysis over time (Abad et al., 1996). Alternatively, spores could be exposed to AMP for only limited time periods in which spore germination could not have taken place (i.e., killing activity can have affected only spores) followed by plating on non-selective media to determination sporicidal activity (Badosa et al., 2009).
Small colony variants (SCV) constitute a slow-growing sub-population of bacteria that are characterized by impaired growth, atypical colony morphology, and an ability to persist in mammalian cells. SCV are characterized by down-regulation of genes for metabolism and virulence, while genes important for adhesion, persistence and biofilm formation are often up-regulated (Proctor et al., 2006; Johns et al., 2015; Kahl et al., 2016). SCV are frequently deficient in electron transport (menadione and haemin auxotrophs) or thiamine biosynthesis (thymidine auxotrophs) and these phenotypes can be reversed by supplementation with menadione, haemin or thymidine, respectively (Proctor et al., 2006). Additionally, SCV are less susceptible to various antibiotics including aminoglycosides, such as tobramycin, trimethoprim-sulfamethoxazole, fluorquinolones, fusidic acid, and even to antiseptics like triclosan (Kahl, 2014; Evans, 2015; Bui et al., 2017). SCV are relatively commonly isolated from infections and are also responsible for latent or recurrent infections, often once antibiotic selection is removed, and are often present in chronic infections (Proctor et al., 2006; Johns et al., 2015). In a number of cases, intracellular SCV have been isolated from sites of infection, including fibroblasts, osteoblasts, macrophages and endothelial cells (Kahl et al., 2016) and intracellular presence can induce SCV formation (Vesga et al., 1996). SCV normally constitute a minor sub-population of cells at the site of infection and therefore their isolation in sufficient numbers for the purposes of AST can be challenging (Johns et al., 2015; Kahl et al., 2016). SCV have been identified in diverse range of bacterial genera and isolated from clinical specimens, including S. aureus, S. epidermidis, S. capitis, P. aeruginosa, Salmonella serovars, Burkholderia spp., Vibrio cholerae, Shigella spp., Brucella melitensis, E. coli, Lactobacillus acidophilus, Serratia marcescens, and Neisseria gonorrhoeae (Proctor et al., 2006; Johns et al., 2015; Kahl et al., 2016). At present, there are no approved methods for AST of SCV, and S. aureus SCV do not grow well in CA-MH broth, making it difficult to generate meaningful results from broth-dilution AST (Precit et al., 2016). Precit and co-workers developed a disk diffusion method suitable for AST of S. aureus SCV (Precit et al., 2016), but this method is unlikely to work well with AMP due to the know interactions of cationic AMP with the negatively charged sulphate and sugar components of the agaropectin in agar (Lehrer et al., 1991). Another difficulty when working with SCV is their reversion to a normal colony phenotype (Proctor et al., 2006; Johns et al., 2015; Kahl et al., 2016). Other studies have described AST of SCV (see Table S1 in Precit et al., 2016 for a complete list), including direct use of the CLSI broth dilution procedure (Gao et al., 2010; Singh et al., 2010) and modifications of this, including prolonged incubation times (von Eiff et al., 1997; Samuelsen et al., 2005; Mercer et al., 2017) and media changes (e.g., Brain Heart Infusion (BHI) Kahl et al., 1998; Yagci et al., 2013). Naturally, AST of intracellular SCV adds a further level of complexity and the authors are not aware of any studies on the activity of AMP against intracellular SCV. For experimental purposes, SCV of S. aureus (with the characteristics of clinical SCV) can be selected for in vitro by incubation in the presence of the P. aeruginosa quorum sensing molecule 4-hydroxy-2-heptylquinoline-N-oxide (HQNO) (Hoffman et al., 2006), although the authors are not aware whether this works with other staphylococci or other bacterial genera. It was reported that SCV of E. coli could be generated following exposure to 2-methyl-1,4-napthoquinone (Colwell, 1946) or copper ions (Weed and Longfellow, 1954) and that mutants in a number of bacterial genes can generate SCV (Santos and Hirshfield, 2016; Tikhomirova et al., 2018; Al Ahmar et al., 2019; Vidovic et al., 2019). The importance of SCV in the context of infection means that AMP activity vs. SCV could represent an important method for the treatment of chronic, recurrent and antibiotic resistant infections, but has received only limited attention in the context of AMP. Glaser and co-workers demonstrated that SCV of S. aureus were less susceptible to the skin-derived AMP RNase7, hBD-2, hBD-3, and LL-37 than wild-type S. aureus as was a hemB mutant when compared to the complemented mutant, with the exception of LL-37. SCV were also less susceptible to the killing activity of human stratum corneum (Glaser et al., 2014). Similarly, SCV of P. aeruginosa were also less susceptible to LL-37 than the wild-type (Pestrak et al., 2018). S. aureus SCV were less susceptible to the AMP lactoferricin B and thrombin-induced microbicidal protein (tPMP) when compared to the wild-type and in the case of lactoferricin B, this was irrespective of the underlying auxotrophy (Koo et al., 1996; Samuelsen et al., 2005). Conversely, an antimicrobial polypeptide (NP108) retained comparable efficacy vs. S. aureus SCV when compared to wild-type S. aureus (Mercer et al., 2017). In another study, S. aureus SCV were less susceptible to the AMP protamine, but equally susceptible to magainin and HNP-1, whereas SCV were more susceptible to dermaseptin than the wild-type S. aureus. Interestingly, exposure to sub-MIC concentrations of protamine selected for SCV in vitro (Sadowska et al., 2002). Zhang and co-workers reported that wild-type and SCV of S. aureus were equally susceptible to LL-37, but that SCV became less susceptible in the presence of bicarbonate, adding a further level of complexity of AST of SCV (Zhang et al., 2018). The above results appear to indicate that susceptibility of SCV of S. aureus to AMP needs to be determined on a case-by-case basis and potentially that he method of AST adopted is also important.
Persister cells are dormant or slow-growing variants of normal wild-type cells, forming a sub-population that are highly tolerant to antimicrobials (despite no genetic basis for resistance), but that can revert back to wild-type growth and sensitivity. Persister cells can be responsible for recalcitrance of infections and relapse following treatment and the emergence of antibiotic resistance (Fisher et al., 2017; Balaban et al., 2019). Most work to-date has been carried out on bacterial persister cells, however, fungal persister cells have been isolated from biofilms and that share many of the characteristics of bacterial persisters, including tolerance to high doses of antifungals (Bojsen et al., 2017; Wuyts et al., 2018). Given the importance of persister cells in infection, it is important to develop antimicrobial agents capable of eradicating persister cells and the efficacy of AMP against bacterial persister cells has been examined in a limited number of studies. The dendrimeric AMP, 2D-24, was active against persister cells of P. aeruginosa PAO1 and a mucoid mutant PDO300, as well as planktonic and biofilm cells (Bahar et al., 2015). Trp/Arg-containing AMP successfully killed planktonic and biofilm persister cells of E. coli (Chen et al., 2011), and the aryl-alkyl-lysine NCK-10 against persister cells, planktonic cells and biofilms of S. aureus (Ghosh et al., 2015) and the LL-37 derivative SAAP-148 (de Breij et al., 2018). Additionally, the temporin analogue, TB_L1FK, the β-defensin derivative, C5, and the dendrimeric AMP, Den-SB056, were equally effective against in vitro generated persister and wild-type cells of P. aeruginosa and S. aureus (Grassi et al., 2017b). Persister cells of some bacterial species can be generated in the laboratory by antibiotic exposure (Dorr et al., 2009; Sulaiman et al., 2018) or treatment with specific chemicals, including E. coli, P. aeruginosa, and S. aureus using the uncoupling agent carbonyl cyanide m-chlorophenyl hydrazone (CCCP) (Kwan et al., 2013; Grassi et al., 2017a) and E. coli using salicylate (Wang et al., 2017), facilitating AST of persisters with AMP.
Interactions of AMP With Antibiotics/Antifungals
A large number of publications have described in vitro synergy of selected AMP with other AMP (Zerweck et al., 2017; Hanson et al., 2019), antibiotics (for example Cassone and Otvos, 2010; Sakoulas et al., 2014; Soren et al., 2015; Pollini et al., 2017; Pizzolato-Cezar et al., 2019) or antifungals (Duggineni et al., 2007; Singh et al., 2017), although this is not always the case (He et al., 2015) and is something that is no doubt under-reported. Conjugates of AMP and antibiotics, organometallic compounds, gold nanoparticles or to create AMP polymers to increase efficacy, to reduce toxicity and/or to improve formulation have become of increasing interest over recent years (for examples, see Reinhardt and Neundorf, 2016; Rajchakit and Sarojini, 2017; David et al., 2018; Sun et al., 2018) and will undoubtedly require additional consideration when it comes to AST. In vitro synergy is often species- or even strain-specific and is often specific to certain antimicrobials, so there are no general rules with respect to predicting synergy. Whilst in vitro synergy between antibiotics or antifungals demonstrates potential, there is limited evidence that synergy translates into the clinic, beyond β-lactam/β-lactamase combinations such as piperacillin/tazobactam and ceftazidime/avibactam, so perhaps it might be set to temper this enthusiasm until detailed standardisation of testing methods and clinical trials have been carried out (Doern, 2014). When conducting AST with combinations of AMP with conventional antimicrobials care needs to be taken analysing antimicrobials where solvent other than water are used (e.g., DMSO), or where conventional media requires supplements, including blood, NaCl and polysorbate (see above for the potential effects of these compounds on AMP), as they may impact upon AMP activity. Dimethyl sulfoxide (DMSO) can function as a membrane permeabiliser and can depolarize membranes (Yu and Quinn, 1998) and clearly, this may enhance the apparent activity of membrane permeabilising AMP.
AMP in Clinical Development
A limited number of antibiotics comprising a peptide element are in clinical use (Table 4), but as yet, no AMP have been approved as therapies although a number are in clinical trials (Table 5). Koo and Seo, in a comprehensive review of AMP drug development, state that approximately half of AMP currently in development are at the preclinical stage, while a third have moved forward to clinical trials, most of those are currently in phase II. Approximately 15% of the AMP failed during one of these stages (Koo and Seo, 2019). These numbers are much more promising than the ones presented by Lau and Dunn for previous years, where the number of discontinued AMP trials was higher than 50% (Lau and Dunn, 2018). To increase the clinical pipeline of AMP drug candidates and more importantly, approval of these potentially AMR game-changing antimicrobials, better and more appropriate AST (and in vivo models, although this not a topic for this manuscript) is critical.
Table 4.
Clinically approved peptide antimicrobials.
| Antimicrobial | Drug name | Company | Class | Source | Mol Wt | Application | MOA | Dosing | References |
|---|---|---|---|---|---|---|---|---|---|
| Colistin (Polymyxin E) | Coly-Mycin | Generic | Lipopeptide | Bacillus colistinus | 1155.4 | Antibacterial; Gram - | Membrane disruption | IV, IM and Inhalation | Karaiskos et al., 2017 |
| Dalbavancina | Dalvance | Durata Therapeutics | Lipoglycopeptide | Semi-synthetic | 1816.7 | Antibacterial; Gram + | Cell wall biosynthesis | IV | Bassetti et al., 2018 |
| Daptomycin | Cubicin | Merck (Cubist) | Lipopeptide | Streptomyces roseosporus | 1620.6 | Antibacterial; Gram + | Membrane disruption | IV | Gonzalez-Ruiz et al., 2016 |
| Gramicidin D | NA | Generic | Linear peptides | Bacillus brevis | 1882.2 | Antibacterial; Gram + | Membrane disruption | Topical | Burkhart et al., 1999 |
| Gramicidin S | NA | Generic | Cyclic peptide | Bacillus brevis | 1141.4 | Antibacterial; Gram + and - | Membrane disruption | Topical | Mogi and Kita, 2009 |
| Oritavancinb | Orbactiv | Melinta Therapeutics | Lipoglycopeptide | Semi-synthetic | 1793.1 | Antibacterial; Gram + | Cell wall biosynthesis | IV | Saravolatz and Stein, 2015 |
| Polymyxin B | NA | Generic | Lipopeptide | Bacillus polymyxa | 1203.5 | Antibacterial; Gram - | Membrane disruption | IV and IM | Rigatto et al., 2019 |
| Teicoplanin | Targocid | NPS Pharma | Glycopeptide | Actinoplanes teichomyceticus | 1879.6 | Antibacterial; Gram + | Cell wall biosynthesis | IV and IM | Campoli-Richards et al., 1990 |
| Telavancinb | Vibativ | Theravance | Lipoglycopeptide | Semi-synthetic | 1755.7 | Antibacterial; Gram + | Cell wall biosynthesis | IV | Higgins et al., 2005 |
| Vancomycin | NA | Generic | Glycopeptide | Streptomyces orientalis | 1449.3 | Antibacterial; Gram + | Cell wall biosynthesis | Oral and IV | Alvarez et al., 2016 |
| Anidulafungin | Eraxis | Pfizer | Echinocandin (lipopeptide) | Semi-synthetic | 1140.2 | Antifungal | Cell wall biosynthesis | IV | Mayr et al., 2011 |
| Caspofungin | Cancidas | Merck | Echinocandin (lipopeptide) | Semi-synthetic | 1093.3 | Antifungal | Cell wall biosynthesis | IV | Song and Stevens, 2016 |
| Micafungin | Mycamine | Astellas | Echinocandin (lipopeptide) | Semi-synthetic | 1270.3 | Antifungal | Cell wall biosynthesis | IV | Scott, 2012 |
IV, Intravenous; IM, Intramuscular; NA, Not applicable;
Dalbavancin is a semi-synthetic derivative of Teicoplanin;
Oritavancin and Telavancin are semi-synthetic derivatives of Vancomycin.
Table 5.
AMP in clinical development (adapted from Koo and Seo, 2019).
| AMP | Source | Targetc | Phase | Company | Administrationd |
|---|---|---|---|---|---|
| EA-230 | hCG derivative | Sepsis and renal failure protection | II | Exponential biotherapies | Iv |
| CZEN-002 | α-MSH derivative | Anti-fungal | IIb | Zengen | Top |
| D2A21 | Synthetic | Burn wound infections | III | Demegen | Top |
| XMP-629 | BPI derivative | Impetigo and acne rosacea | IIIb | Xoma Ltd. | Top |
| Neuprex(rBPI21) | BPR derivative | Peadiatric meningococcemia | IIIb | Xoma Ltd. | iv |
| Delmitide(RDP58) | HLA class I derivative | Inflammatory bowel disease | IIa | Genzyme | Top |
| Ghrelin | Endogenous HDP | Chronic respiratory failure | IIa | University of Miyazaki; Papworth Hospital | iv |
| hLF1-11 | Lactoferricin derivative | MRSA, K. pneumoniae, L. monocytogenes | I/II | AM-Pharma | iv |
| C16G2 | Synthetic | Tooth decay by Streptococcus mutans | II | C3 Jian Inc. | Mouthwash |
| SGX942(Dusquetide) | Synthetic | Oral mucositis | III | Soligenix | Oral rinse |
| DPK-060 | Kininogen derivative | Acute external otitis | II | ProMore pharma | Ear drops |
| PXL01 | Lactoferrin analogue | Postsurgical adhesions | III | ProMore pharma | Top |
| PAC113 | Histatin 5 analogue | Oral candidiasis | IIa | Pacgen biopharmaceuticals | Mouth rinse |
| POL7080 | Protegrin analogue | P. aeruginosa, K. pneumoniae | III | Polyphor Ltd. | iv |
| LTX-109 (Lytixar) | Synthetic | G+, MRSA skin infection; impetigo | IIa | Lytix biopharma | Top |
| OP-145 | LL-37 derivative | Chronic middle ear infection | IIa | Dr. Reddy's research | Ear drops |
| LL-37 | Human cathelicidin | Leg ulcer | IIb | ProMore pharma | Top |
| Novexatin (NP213) | Cyclic cationic peptide | Fungal nail infection | IIa | NovaBiotics Ltd. | Top |
| p2TA (AB103) | Synthetic | Necrotizing soft tissue infections | III | Atox Bio Ltd. | iv |
| Iseganan (IB-367) | Protegrin analogue | Pneumonia, stomatitis | IIIb | IntraBiotics pharmaceuticals | Top |
| Pexiganan (MSI-78) | Magainin analogue | Diabetic foot ulcers | IIIb | Dipexium pharmaceuticals | Top |
| Omiganan (CLS001) | Indolicidin derivative | Rosacea | III | Cutanea life sciences | Top |
Clinical trial completed.
Clinical trial discontinued.
Target microorgansim: G+ - Gram positive; MRSA – methicillin-resistant S. aureus.
Route of administration: top - topical; iv – intravenous.
Conclusions
Antimicrobial resistance is a global health problem that will require the survey of a large number of molecules with a variety of chemistries and new functional families. AMP are clearly a group of molecules with significant potential as a new class of therapy to address the urgent need for better and AMR-mitigating antibacterial and antifungal therapies. Among other advantages highlighted in this and other reviews, AMP offer great versatility in terms of chemical functionality. Unfortunately, “standard” AST protocols can significantly underestimate the efficacy of AMP and improved, more predictive methods are required to identify leads and facilitate faster progress of AMP into pre-/clinical development. These changes to “standard” protocols will require rigorous and detailed justification and a degree of validation. New AST of AMP will have to take into account that many AMP depend on their positive charge for activity as well as the many other factors we have enumerated in this review (see Table 2). Despite the manifold factors that influence AMP activity, we are confident that research toward developing more appropriate AST for AMP is headed in the right direction. New protocols will, in the near future, accelerate the discovery process of novel AMP therapeutics.
Author Contributions
DM, SD, MT, EL, LS, MK-B, CF-N, DO'N, and AA-B contributed to the writing of this manuscript. DM, MT, CF-N, DO'N, and AA-B contributed to the editing of this manuscript. All authors contributed to the article and approved the submitted version.
Conflict of Interest
DM, EL, LS and DO'N are employed by NovaBiotics Ltd. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Footnotes
Funding. This work was partially supported by the National Science Foundation (MCB1715494 to AA-B). Cesar de la Fuente-Nunez holds a Presidential Professorship at the University of Pennsylvania, is a recipient of the Langer Prize by the AIChE Foundation, and acknowledges funding from the Institute for Diabetes, Obesity, and Metabolism and the Penn Mental Health AIDS Research Center of the University of Pennsylvania.
References
- Abad L. R., D'Urzo M. P., Liu D., Narasimhan M. L., Reuveni M., Zhu J. K., et al. (1996). Antifungal activity of tobacco osmotin has specificity and involves plasma membrane permeabilization. Plant Sci. 118, 11–23. 10.1016/0168-9452(96)04420-2 [DOI] [Google Scholar]
- Achard M. E., Stafford S. L., Bokil N. J., Chartres J., Bernhardt P. V., Schembri M. A., et al. (2012). Copper redistribution in murine macrophages in response to Salmonella infection. Biochem. J. 444, 51–57. 10.1042/BJ20112180 [DOI] [PubMed] [Google Scholar]
- Agbale C. M., Sarfo J. K., Galyuon I. K., Juliano S. A., Silva G. G. O., Buccini D. F., et al. (2019). Antimicrobial and antibiofilm activities of helical antimicrobial peptide sequences incorporating metal-binding motifs. Biochemistry 58, 3802–3812. 10.1021/acs.biochem.9b00440 [DOI] [PubMed] [Google Scholar]
- Ageitos J. M., Sanchez-Perez A., Calo-Mata P., Villa T. G. (2016). Antimicrobial peptides (AMPs): Ancient compounds that represent novel weapons in the fight against bacteria. Biochem. Pharmacol. 133, 117–138. 10.1016/j.bcp.2016.09.018 [DOI] [PubMed] [Google Scholar]
- Aisenbrey C., Marquette A., Bechinger B. (2019). The mechanisms of action of cationic antimicrobial peptides refined by novel concepts from biophysical investigations. Adv. Exp. Med. Biol. 1117, 33–64. 10.1007/978-981-13-3588-4_4 [DOI] [PubMed] [Google Scholar]
- Al Ahmar R., Kirby B. D., Yu H. D. (2019). Pyrimidine biosynthesis regulates the small-colony variant and mucoidy in Pseudomonas aeruginosa through sigma factor competition. J. Bacteriol. 201:e00575–18. 10.1128/JB.00575-18 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alastruey-Izquierdo A., Gomez-Lopez A., Arendrup M. C., Lass-Florl C., Hope W. W., Perlin D. S., et al. (2012). Comparison of dimethyl sulfoxide and water as solvents for echinocandin susceptibility testing by the EUCAST methodology. J. Clin. Microbiol. 50, 2509–2512. 10.1128/JCM.00791-12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Albur M., Noel A., Bowker K., Macgowan A. (2014). Colistin susceptibility testing: time for a review. J. Antimicrob. Chemother. 69, 1432–1434. 10.1093/jac/dkt503 [DOI] [PubMed] [Google Scholar]
- Alexander J. L., Thompson Z., Cowan J. A. (2018). Antimicrobial metallopeptides. ACS Chem. Biol. 13, 844–853. 10.1021/acschembio.7b00989 [DOI] [PubMed] [Google Scholar]
- Alexander J. L., Yu Z., Cowan J. A. (2017). Amino terminal copper and nickel binding motif derivatives of ovispirin-3 display increased antimicrobial activity via lipid oxidation. J. Med. Chem. 60, 10047–10055. 10.1021/acs.jmedchem.7b01117 [DOI] [PubMed] [Google Scholar]
- Alvares D. S., Viegas T. G., Ruggiero Neto J. (2017). Lipid-packing perturbation of model membranes by pH-responsive antimicrobial peptides. Biophys. Rev. 9, 669–682. 10.1007/s12551-017-0296-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alvarez R., Lopez Cortes L. E., Molina J., Cisneros J. M., Pachon J. (2016). Optimizing the clinical use of vancomycin. Antimicrob. Agents Chemother. 60, 2601–2609. 10.1128/AAC.03147-14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Annis D. H., Craig B. A. (2005). The effect of interlaboratory variability on antimicrobial susceptibility determination. Diagn. Microbiol. Infect. Dis. 53, 61–64. 10.1016/j.diagmicrobio.2005.03.012 [DOI] [PubMed] [Google Scholar]
- Aoki W., Ueda M. (2013). Characterization of antimicrobial peptides toward the development of novel antibiotics. Pharmaceuticals 6, 1055–1081. 10.3390/ph6081055 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arendrup M. C., Jorgensen K. M., Hare R. K., Cuenca-Estrella M., Zaragoza O. (2019). EUCAST reference testing of rezafungin susceptibility: impact of choice of plastic plates. Antimicrob. Agents Chemother. 63, e00659–e00619. 10.1128/AAC.00659-19 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arendrup M. C., Rodriguez-Tudela J. L., Park S., Garcia-Effron G., Delmas G., Cuenca-Estrella M., et al. (2011). Echinocandin susceptibility testing of Candida spp. Using EUCAST EDef 7.1 and CLSI M27-A3 standard procedures: analysis of the influence of bovine serum albumin supplementation, storage time, and drug lots. Antimicrob. Agents Chemother. 55, 1580–1587. 10.1128/AAC.01364-10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arhin F. F., Sarmiento I., Belley A., McKay G. A., Draghi D. C., Grover P., et al. (2008). Effect of polysorbate 80 on oritavancin binding to plastic surfaces: implications for susceptibility testing. Antimicrob. Agents Chemother. 52, 1597–1603. 10.1128/AAC.01513-07 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Awale M., Visini R., Probst D., Arus-Pous J., Reymond J. L. (2017). Chemical space: big data challenge for molecular diversity. Chimia 71, 661–666. 10.2533/chimia.2017.661 [DOI] [PubMed] [Google Scholar]
- Badosa E., Ferre R., Frances J., Bardaji E., Feliu L., Planas M., et al. (2009). Sporicidal activity of synthetic antifungal undecapeptides and control of Penicillium rot of apples. Appl. Environ. Microbiol. 75, 5563–5569. 10.1128/AEM.00711-09 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bahar A. A., Liu Z., Totsingan F., Buitrago C., Kallenbach N., Ren D. (2015). Synthetic dendrimeric peptide active against biofilm and persister cells of Pseudomonas aeruginosa. Appl. Microbiol. Biotechnol. 99, 8125–8135. 10.1007/s00253-015-6645-7 [DOI] [PubMed] [Google Scholar]
- Balaban N. Q., Helaine S., Lewis K., Ackermann M., Aldridge B., Andersson D. I., et al. (2019). Definitions and guidelines for research on antibiotic persistence. Nat. Rev. Microbiol. 17, 441–448. 10.1038/s41579-019-0196-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bals R., Wang X., Wu Z., Freeman T., Bafna V., Zasloff M., et al. (1998). Human beta-defensin 2 is a salt-sensitive peptide antibiotic expressed in human lung. J. Clin. Invest. 102, 874–880. 10.1172/JCI2410 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barlow G. (2018). Clinical challenges in antimicrobial resistance. Nat. Microbiol. 3, 258–260. 10.1038/s41564-018-0121-y [DOI] [PubMed] [Google Scholar]
- Bassetti M., Peghin M., Carnelutti A., Righi E. (2018). The role of dalbavancin in skin and soft tissue infections. Curr. Opin. Infect. Dis. 31, 141–147. 10.1097/QCO.0000000000000430 [DOI] [PubMed] [Google Scholar]
- Batoni G., Maisetta G., Esin S. (2016). Antimicrobial peptides and their interaction with biofilms of medically relevant bacteria. Biochim. Biophys. Acta 1858, 1044–1060. 10.1016/j.bbamem.2015.10.013 [DOI] [PubMed] [Google Scholar]
- Baumert N., von Eiff C., Schaaff F., Peters G., Proctor R. A., Sahl H. G. (2002). Physiology and antibiotic susceptibility of Staphylococcus aureus small colony variants. Microb. Drug Resist. 8, 253–260. 10.1089/10766290260469507 [DOI] [PubMed] [Google Scholar]
- Bayramov D., Li Z., Patel E., Izadjoo M., Kim H., Neff J. (2018). A novel peptide-based antimicrobial wound treatment is effective against biofilms of multi-drug resistant wound pathogens. Mil. Med. 183(Suppl_1), 481–486. 10.1093/milmed/usx135 [DOI] [PubMed] [Google Scholar]
- Becucci L., Valensin D., Innocenti M., Guidelli R. (2014). Dermcidin, an anionic antimicrobial peptide: influence of lipid charge, pH and Zn2+ on its interaction with a biomimetic membrane. Soft Matter 10, 616–626. 10.1039/C3SM52400K [DOI] [PubMed] [Google Scholar]
- Blair J. M., Webber M. A., Baylay A. J., Ogbolu D. O., Piddock L. J. (2015). Molecular mechanisms of antibiotic resistance. Nat. Rev. Microbiol. 13, 42–51. 10.1038/nrmicro3380 [DOI] [PubMed] [Google Scholar]
- Blower R. J., Popov S. G., van Hoek M. L. (2018). Cathelicidin peptide rescues G. mellonella infected with B. anthracis. Virulence 9, 287–293. 10.1080/21505594.2017.1293227 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bojsen R., Regenberg B., Folkesson A. (2017). Persistence and drug tolerance in pathogenic yeast. Curr. Genet. 63, 19–22. 10.1007/s00294-016-0613-3 [DOI] [PubMed] [Google Scholar]
- Bono M. J., Reygaert W. C. (2019). Urinary tract infection, in StatPearls (Treasure Island, FL: StatPearls Publishing. [Google Scholar]
- Borello J. S. (1976). The effect of copper on taste sensitivity and caries activity (Ph.D. thesis). Loyola University Chicago, Chicago, IL, United States. [Google Scholar]
- Boron W. F., Boulpaep E. L. (2005). Medical Physiology: A Cellular and Molecular Approach. Philadelphia, PA: Elsevier Saunders. [Google Scholar]
- Botella H., Peyron P., Levillain F., Poincloux R., Poquet Y., Brandli I., et al. (2011). Mycobacterial p(1)-type ATPases mediate resistance to zinc poisoning in human macrophages. Cell Host Microbe 10, 248–259. 10.1016/j.chom.2011.08.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bottger R., Hoffmann R., Knappe D. (2017). Differential stability of therapeutic peptides with different proteolytic cleavage sites in blood, plasma and serum. PLoS ONE 12:e0178943. 10.1371/journal.pone.0178943 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boulos L., Prevost M., Barbeau B., Coallier J., Desjardins R. (1999). LIVE/DEAD BacLight : application of a new rapid staining method for direct enumeration of viable and total bacteria in drinking water. J. Microbiol. Methods 37, 77–86. 10.1016/S0167-7012(99)00048-2 [DOI] [PubMed] [Google Scholar]
- Bowen R. A., Remaley A. T. (2014). Interferences from blood collection tube components on clinical chemistry assays. Biochem. Med. 24, 31–44. 10.11613/BM.2014.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradford P. A., Petersen P. J., Young M., Jones C. H., Tischler M., O'Connell J. (2005). Tigecycline MIC testing by broth dilution requires use of fresh medium or addition of the biocatalytic oxygen-reducing reagent oxyrase to standardize the test method. Antimicrob. Agents Chemother. 49, 3903–3909. 10.1128/AAC.49.9.3903-3909.2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Breteler K. B., Rentenaar R. J., Verkaart G., Sturm P. D. (2011). Performance and clinical significance of direct antimicrobial susceptibility testing on urine from hospitalized patients. Scand. J. Infect. Dis. 43, 771–776. 10.3109/00365548.2011.588609 [DOI] [PubMed] [Google Scholar]
- Brion M., Lambs L., Berthon G. (1985). Metal ion-tetracycline interactions in biological fluids. Part 5. Formation of zinc complexes with tetracycline and some of its derivatives and assessment of their biological significance. Agents Actions 17, 229–242. 10.1007/BF01966597 [DOI] [PubMed] [Google Scholar]
- Brogden K. A. (2005). Antimicrobial peptides: pore formers or metabolic inhibitors in bacteria? Nat. Rev. Microbiol. 3, 238–250. 10.1038/nrmicro1098 [DOI] [PubMed] [Google Scholar]
- Brook I. (1989). Inoculum effect. Rev. Infect. Dis. 11, 361–368. 10.1093/clinids/11.3.361 [DOI] [PubMed] [Google Scholar]
- Brown D. F. (2001). Detection of methicillin/oxacillin resistance in Staphylococci. J. Antimicrob. Chemother. 48(Suppl. 1), 65–70. 10.1093/jac/48.suppl_1.65 [DOI] [PubMed] [Google Scholar]
- Brown D. F., Wootton M., Howe R. A. (2016). Antimicrobial susceptibility testing breakpoints and methods from BSAC to EUCAST. J. Antimicrob. Chemother. 71, 3–5. 10.1093/jac/dkv287 [DOI] [PubMed] [Google Scholar]
- Brown H. L., van Vliet A. H., Betts R. P., Reuter M. (2013). Tetrazolium reduction allows assessment of biofilm formation by Campylobacter jejuni in a food matrix model. J. Appl. Microbiol. 115, 1212–1221. 10.1111/jam.12316 [DOI] [PubMed] [Google Scholar]
- Brown M. R., Geaton E. M., Gilbert P. (1979). Additivity of action between polysorbate 80 and polymyxin B towards spheroplasts of Pseudomonas aeruginosa NCTC 6750. J. Pharm. Pharmacol. 31, 168–170. 10.1111/j.2042-7158.1979.tb13463.x [DOI] [PubMed] [Google Scholar]
- Brown M. R., Winsley B. E. (1968). Synergistic action of polysorbate 80 and polymyxin B sulphate on Pseudomonas aeruginosa. J. Gen. Microbiol. 50:Suppl:ix. [PubMed] [Google Scholar]
- Bui L. M., Conlon B. P., Kidd S. P. (2017). Antibiotic tolerance and the alternative lifestyles of Staphylococcus aureus. Essays Biochem. 61, 71–79. 10.1042/EBC20160061 [DOI] [PubMed] [Google Scholar]
- Buonocore F., Randelli E., Casani D., Picchietti S., Belardinelli M. C., de Pascale D., et al. (2012). A piscidin-like antimicrobial peptide from the icefish Chionodraco hamatus (Perciformes: Channichthyidae): molecular characterization, localization and bactericidal activity. Fish Shellfish Immunol. 33, 1183–1191. 10.1016/j.fsi.2012.09.005 [DOI] [PubMed] [Google Scholar]
- Burkhart B. M., Gassman R. M., Langs D. A., Pangborn W. A., Duax W. L., Pletnev V. (1999). Gramicidin D conformation, dynamics and membrane ion transport. Biopolymers 51, 129–144. [DOI] [PubMed] [Google Scholar]
- Caillon L., Killian J. A., Lequin O., Khemtemourian L. (2013). Biophysical investigation of the membrane-disrupting mechanism of the antimicrobial and amyloid-like peptide dermaseptin S9. PLoS ONE 8:e75528. 10.1371/journal.pone.0075528 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campeau S. A., Schuetz A. N., Kohner P., Arias C. A., Hemarajata P., Bard J. D., et al. (2018). Variability of daptomycin MIC values for enterococcus faecium when measured by reference broth microdilution and gradient diffusion tests. Antimicrob. Agents Chemother. 62:e00745–18. 10.1128/AAC.00745-18 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campoli-Richards D. M., Brogden R. N., Faulds D. (1990). Teicoplanin. A review of its antibacterial activity, pharmacokinetic properties and therapeutic potential. Drugs 40, 449–486. 10.2165/00003495-199040030-00007 [DOI] [PubMed] [Google Scholar]
- Cansizoglu M. F., Tamer Y. T., Farid M., Koh A. Y., Toprak E. (2019). Rapid ultrasensitive detection platform for antimicrobial susceptibility testing. PLoS Biol. 17:e3000291. 10.1371/journal.pbio.3000291 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carreau A., El Hafny-Rahbi B., Matejuk A., Grillon C., Kieda C. (2011). Why is the partial oxygen pressure of human tissues a crucial parameter? Small molecules and hypoxia. J. Cell Mol. Med. 15, 1239–1253. 10.1111/j.1582-4934.2011.01258.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cassone M., Otvos L., Jr. (2010). Synergy among antibacterial peptides and between peptides and small-molecule antibiotics. Expert Rev. Anti Infect. Ther. 8, 703–716. 10.1586/eri.10.38 [DOI] [PubMed] [Google Scholar]
- CDC (2019). Antibiotic Resistance Threats in the United States, 2019. Atlanta, GA: US Department of Health and Human Services, CDC. [Google Scholar]
- Ceri H., Olson M. E., Stremick C., Read R. R., Morck D., Buret A. (1999). The Calgary Biofilm Device: new technology for rapid determination of antibiotic susceptibilities of bacterial biofilms. J. Clin. Microbiol. 37, 1771–1776. 10.1128/JCM.37.6.1771-1776.1999 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chairatana P., Nolan E. M. (2017). Human alpha-defensin 6: a small peptide that self-assembles and protects the host by entangling microbes. Acc. Chem. Res. 50, 960–967. 10.1021/acs.accounts.6b00653 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chatterjee A., Modarai M., Naylor N. R., Boyd S. E., Atun R., Barlow J., et al. (2018). Quantifying drivers of antibiotic resistance in humans: a systematic review. Lancet Infect. Dis. 18, e368–78. 10.1016/S1473-3099(18)30296-2 [DOI] [PubMed] [Google Scholar]
- Chen X., Zhang M., Zhou C., Kallenbach N. R., Ren D. (2011). Control of bacterial persister cells by Trp/Arg-containing antimicrobial peptides. Appl. Environ. Microbiol 77, 4878–4885. 10.1128/AEM.02440-10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chico D. E., Given R. L., Miller B. T. (2003). Binding of cationic cell-permeable peptides to plastic and glass. Peptides 24, 3–9. 10.1016/S0196-9781(02)00270-X [DOI] [PubMed] [Google Scholar]
- Chowdhary A., Singh P. K., Kathuria S., Hagen F., Meis J. F. (2015). Comparison of the EUCAST and CLSI broth microdilution methods for testing isavuconazole, posaconazole, and amphotericin b against molecularly identified mucorales species. Antimicrob. Agents Chemother. 59, 7882–7887. 10.1128/AAC.02107-15 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chu H., Pazgier M., Jung G., Nuccio S. P., Castillo P. A., de Jong M. F., et al. (2012). Human alpha-defensin 6 promotes mucosal innate immunity through self-assembled peptide nanonets. Science 337, 477–481. 10.1126/science.1218831 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chu H. L., Yu H. Y., Yip B. S., Chih Y. H., Liang C. W., Cheng H. T., et al. (2013). Boosting salt resistance of short antimicrobial peptides. Antimicrob. Agents Chemother. 57, 4050–4052. 10.1128/AAC.00252-13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chung E. M. C., Dean S. N., Propst C. N., Bishop B. M., van Hoek M. L. (2017). Komodo dragon-inspired synthetic peptide DRGN-1 promotes wound-healing of a mixed-biofilm infected wound. NPJ Biofilms Microb. 3:9. 10.1038/s41522-017-0017-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cipcigan F., Carrieri A. P., Pyzer-Knapp E. O., Krishna R., Hsiao Y. W., Winn M., et al. (2018). Accelerating molecular discovery through data and physical sciences: Applications to peptide-membrane interactions. J. Chem. Phys. 148, 241744. 10.1063/1.5027261 [DOI] [PubMed] [Google Scholar]
- Clinton A., Carter T. (2015). Chronic wound biofilms: pathogenesis and potential therapies. Lab. Med. 46, 277–284. 10.1309/LMBNSWKUI4JPN7SO [DOI] [PubMed] [Google Scholar]
- CLSI (2010). Method for Antifungal Disk Diffusion Susceptibility Testing of Nondermatophyte Filamentous Fungi. M51, ED1. Wayne, PA, USA: Clinical and Laboratory Standards Institute. [Google Scholar]
- CLSI (2011). Methods for Antimicrobial Susceptibility Testing for Human Mycoplasmas. M43, ED1. Wayne, PA: Clinical and Laboratory Standards Institute. [PubMed] [Google Scholar]
- CLSI (2012). Principles and Procedures for Detection of Fungi in Clinical Specimens—Direct Examination and Culture, M54, ED1. Wayne, PA: Clinical and Laboratory Standards Institute. [Google Scholar]
- CLSI (2015). Methods for Antimicrobial Dilution and Disk Susceptibility Testing of Infrequently Isolated or Fastidious Bacteria. M45, ED3. Wayne, PA: Clinical and Laboratory Standards Institute. [DOI] [PubMed] [Google Scholar]
- CLSI (2017b). Reference method for Broth Dilution Antifungal Susceptibility Testing of Filamentous Fungi; 3rd Edn; M38-Ed3. Wayne, PA: Clinical and Laboratory Standards Institute. [Google Scholar]
- CLSI (2017c). Reference Method for Broth Dilution Antifungal Susceptibility Testing of Filamentous Fungi; 3rd Edn; M38-Ed3. Wayne, PA: Clinical and Laboratory Standards Institute. [Google Scholar]
- CLSI (2017d). Performance Standards for Antifungal Susceptibility Testing of Filamentous Fungi. M61, ED1. Wayne, PA: Clinical and Laboratory Standards Institute. [Google Scholar]
- CLSI (2018a). Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria that Grow Aerobically; Approved Standard – 11th Edn; M07-ed10. Wayne, PA: Clinical and Laboratory Standards Institute. [Google Scholar]
- CLSI (2018b). Methods for Antimicrobial Susceptibility Testing of Anaerobic Bacteria. M11, ED9. Wayne, PA: Clinical and Laboratory Standards Institute. [PubMed] [Google Scholar]
- CLSI (2018c). Susceptibility Testing of Mycobacteria, Nocardia spp., and Other Aerobic Actinomycetes. M24, ED3. Wayne, PA: Clinical and Laboratory Standards Institute. [PubMed] [Google Scholar]
- CLSI (2018d). Performance Standards for Susceptibility Testing of Mycobacteria, Nocardia spp., and Other Aerobic Actinomycetes. M62, ED1. Wayne, PA: Clinical and Laboratory Standards Institute. [PubMed] [Google Scholar]
- CLSI (2018e). Method for Antifungal Disk Diffusion Susceptibility Testing of Yeasts. M44, ED3. Wayne, PA: Clinical and Laboratory Standards Institute. [Google Scholar]
- CLSI (2018f). Epidemiological Cutoff Values for Antifungal Susceptibility Testing, M59, ED2. Wayne, PA: Clinical and Laboratory Standards Institute. [Google Scholar]
- CLSI (2018g). Development of In vitro Susceptibility Testing Criteria and Quality Control Parameters, M23, ED5. Wayne, PA: Clinical and Laboratory Standards Institute. [Google Scholar]
- CLSI (2019). Performance Standards for Antimicrobial Susceptibility Testing. M100, ED29. Wayne, PA: Clinical and Laboratory Standards Institute. [DOI] [PMC free article] [PubMed] [Google Scholar]
- CLSI (2017a). Reference Method for Broth Dilution Antifungal Susceptibility Testing of Yeasts, 4th Edn; M27-Ed4. Wayne, PA: Clinical and Laboratory Standards Institute. [Google Scholar]
- CLSI-EUCAST (2016). Recommendations for MIC Determination of Colistin (polymyxin E). Available online at: http://www.eucast.org/ast_of_bacteria/guidance_documents/
- Coates M., Blanchard S., MacLeod A. S. (2018). Innate antimicrobial immunity in the skin: a protective barrier against bacteria, viruses, and fungi. PLoS Pathog. 14:e1007353. 10.1371/journal.ppat.1007353 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cole J. N., Nizet V. (2016). Bacterial evasion of host antimicrobial peptide defenses. Microbiol. Spectr. 4. 10.1128/microbiolspec.VMBF-0006-2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Colwell C. A. (1946). Small colony variants of Escherichia coli. J. Bacteriol. 52, 417–422. 10.1128/JB.52.4.417-422.1946 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Corral L. G., Post L. S., Montville T. J. (1988). Antimicrobial activity of sodium bicarbonate. J. Food Sci. 53, 981–982. 10.1111/j.1365-2621.1988.tb09005.x [DOI] [Google Scholar]
- Correa W., Brandenburg K., Zahringer U., Ravuri K., Khan T., von Wintzingerode F. (2017). Biophysical analysis of lipopolysaccharide formulations for an understanding of the Low Endotoxin Recovery (LER) Phenomenon. Int. J. Mol. Sci. 18:2737. 10.3390/ijms18122737 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Costerton J. W., Lewandowski Z., Caldwell D. E., Korber D. R., Lappin-Scott H. M. (1995). Microbial biofilms. Annu. Rev. Microbiol. 49, 711–745. 10.1146/annurev.mi.49.100195.003431 [DOI] [PubMed] [Google Scholar]
- Crofts A. A., Giovanetti S. M., Rubin E. J., Poly F. M., Gutierrez R. L., Talaat K. R., et al. (2018). Enterotoxigenic E. coli virulence gene expression in human infections. Proc. Natl. Acad. Sci. U.S.A. 115, E8968–E8976. 10.1073/pnas.1808982115 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cusack T. P., Ashley E. A., Ling C. L., Rattanavong S., Roberts T., Turner P., et al. (2019). Impact of CLSI and EUCAST breakpoint discrepancies on reporting of antimicrobial susceptibility and AMR surveillance. Clin. Microbiol. Infect. 25, 910–911. 10.1016/j.cmi.2019.03.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- da Costa J. P., Cova M., Ferreira R., Vitorino R. (2015). Antimicrobial peptides: an alternative for innovative medicines? Appl. Microbiol. Biotechnol. 99, 2023–2040. 10.1007/s00253-015-6375-x [DOI] [PubMed] [Google Scholar]
- David A. A., Park S. E., Parang K., Tiwari R. K. (2018). Antibiotics-peptide conjugates against multidrug-resistant bacterial pathogens. Curr. Top. Med. Chem. 18, 1926–1936. 10.2174/1568026619666181129141524 [DOI] [PubMed] [Google Scholar]
- de Alteriis E., Lombardi L., Falanga A., Napolano M., Galdiero S., Siciliano A., et al. (2018). Polymicrobial antibiofilm activity of the membranotropic peptide gH625 and its analogue. Microb. Pathog. 125, 189–195. 10.1016/j.micpath.2018.09.027 [DOI] [PubMed] [Google Scholar]
- de Breij A., Riool M., Cordfunke R. A., Malanovic N., de Boer L., Koning R. I., et al. (2018). The antimicrobial peptide SAAP-148 combats drug-resistant bacteria and biofilms. Sci. Transl. Med. 10:aan4044. 10.1126/scitranslmed.aan4044 [DOI] [PubMed] [Google Scholar]
- de Jong H. K., Parry C. M., van der Poll T., Wiersinga W. J. (2012). Host-pathogen interaction in invasive Salmonellosis. PLoS Pathog. 8:e1002933. 10.1371/journal.ppat.1002933 [DOI] [PMC free article] [PubMed] [Google Scholar]
- de la Fuente-Nunez C. (2019). Toward autonomous antibiotic discovery. mSystems 4:e00151–19. 10.1128/mSystems.00151-19 [DOI] [PMC free article] [PubMed] [Google Scholar]
- de la Fuente-Nunez C., Cardoso M. H., de Souza Candido E., Franco O. L., Hancock R. E. (2016). Synthetic antibiofilm peptides. Biochim. Biophys. Acta 1858, 1061–1069. 10.1016/j.bbamem.2015.12.015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- de la Fuente-Nunez C., Reffuveille F., Haney E. F., Straus S. K., Hancock R. E. (2014). Broad-spectrum anti-biofilm peptide that targets a cellular stress response. PLoS Pathog. 10:e1004152. 10.1371/journal.ppat.1004152 [DOI] [PMC free article] [PubMed] [Google Scholar]
- de la Fuente-Nunez C., Silva O. N., Lu T. K., Franco O. L. (2017a). Antimicrobial peptides: role in human disease and potential as immunotherapies. Pharmacol. Ther. 178, 132–140. 10.1016/j.pharmthera.2017.04.002 [DOI] [PubMed] [Google Scholar]
- de la Fuente-Nunez C., Torres M. D., Mojica F. J., Lu T. K. (2017b). Next-generation precision antimicrobials: towards personalized treatment of infectious diseases. Curr. Opin. Microbiol. 37, 95–102. 10.1016/j.mib.2017.05.014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Mendoza D. (2014). Temperature sensing by membranes. Annu. Rev. Microbiol. 68, 101–116. 10.1146/annurev-micro-091313-103612 [DOI] [PubMed] [Google Scholar]
- Delaloye J., Calandra T. (2014). Invasive candidiasis as a cause of sepsis in the critically ill patient. Virulence 5, 161–169. 10.4161/viru.26187 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Delattin N., Brucker K., Cremer K., Cammue B. P., Thevissen K. (2017). Antimicrobial peptides as a strategy to combat fungal biofilms. Curr. Top. Med. Chem. 17, 604–612. 10.2174/1568026616666160713142228 [DOI] [PubMed] [Google Scholar]
- Denich T. J., Beaudette L. A., Lee H., Trevors J. T. (2003). Effect of selected environmental and physico-chemical factors on bacterial cytoplasmic membranes. J. Microbiol. Methods 52, 149–182. 10.1016/S0167-7012(02)00155-0 [DOI] [PubMed] [Google Scholar]
- Dennison S. R., Howe J., Morton L. H., Brandenburg K., Harris F., Phoenix D. A. (2006). Interactions of an anionic antimicrobial peptide with Staphylococcus aureus membranes. Biochem. Biophys. Res. Commun. 347, 1006–1010. 10.1016/j.bbrc.2006.06.181 [DOI] [PubMed] [Google Scholar]
- Desai J. V., Mitchell A. P., Andes D. R. (2014). Fungal biofilms, drug resistance, and recurrent infection. Cold Spring Harb. Perspect. Med 4:a019729. 10.1101/cshperspect.a019729 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deslouches B., Islam K., Craigo J. K., Paranjape S. M., Montelaro R. C., Mietzner T. A. (2005). Activity of the de novo engineered antimicrobial peptide WLBU2 against Pseudomonas aeruginosa in human serum and whole blood: implications for systemic applications. Antimicrob. Agents Chemother. 49, 3208–3216. 10.1128/AAC.49.8.3208-3216.2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dewachter L., Fauvart M., Michiels J. (2019). Bacterial heterogeneity and antibiotic survival: understanding and combatting persistence and heteroresistance. Mol. Cell 76, 255–267. 10.1016/j.molcel.2019.09.028 [DOI] [PubMed] [Google Scholar]
- Dickerhof N., Kleffmann T., Jack R., McCormick S. (2011). Bacitracin inhibits the reductive activity of protein disulfide isomerase by disulfide bond formation with free cysteines in the substrate-binding domain. FEBS J. 278, 2034–2043. 10.1111/j.1742-4658.2011.08119.x [DOI] [PubMed] [Google Scholar]
- Djoko K. Y., Ong C. L., Walker M. J., McEwan A. G. (2015). The role of copper and zinc toxicity in innate immune defense against bacterial pathogens. J. Biol. Chem 290, 18954–18961. 10.1074/jbc.R115.647099 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dobay O., Laub K., Stercz B., Keri A., Balazs B., Tothpal A., et al. (2018). Bicarbonate inhibits bacterial growth and biofilm formation of prevalent cystic fibrosis pathogens. Front. Microbiol. 9:2245. 10.3389/fmicb.2018.02245 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doern C. D. (2014). When does 2 plus 2 equal 5? a review of antimicrobial synergy testing. J. Clin. Microbiol. 52, 4124–4128. 10.1128/JCM.01121-14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doern G. V., Vautour R., Gaudet M., Levy B. (1994). Clinical impact of rapid in vitro susceptibility testing and bacterial identification. J. Clin. Microbiol. 32, 1757–1762. 10.1128/JCM.32.7.1757-1762.1994 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doluisio J. T., Martin A. N. (1963). Metal complexation of the tetracycline hydrochlorides. J. Med. Chem 6, 16–20. 10.1021/jm00337a003 [DOI] [PubMed] [Google Scholar]
- Dong N., Chou S., Li J., Xue C., Li X., Cheng B., et al. (2018). Short symmetric-end antimicrobial peptides centered on beta-turn amino acids unit improve selectivity and stability. Front. Microbiol 9:2832. 10.3389/fmicb.2018.02832 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dorr T., Lewis K., Vulic M. (2009). SOS response induces persistence to fluoroquinolones in Escherichia coli. PLoS Genet. 5:e1000760. 10.1371/journal.pgen.1000760 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dorschner R. A., Lopez-Garcia B., Peschel A., Kraus D., Morikawa K., Nizet V., et al. (2006). The mammalian ionic environment dictates microbial susceptibility to antimicrobial defense peptides. FASEB J. 20, 35–42. 10.1096/fj.05-4406com [DOI] [PubMed] [Google Scholar]
- Dreizen S., Spies H. A., Jr., Spies T. D. (1952). The copper and cobalt levels of human saliva and dental caries activity. J. Dent. Res. 31, 137–142. 10.1177/00220345520310011001 [DOI] [PubMed] [Google Scholar]
- Dubois-Brissonnet F., Trotier E., Briandet R. (2016). The biofilm lifestyle involves an increase in bacterial membrane saturated fatty acids. Front. Microbiol 7:1673. 10.3389/fmicb.2016.01673 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duggineni S., Srivastava G., Kundu B., Kumar M., Chaturvedi A. K., Shukla P. K. (2007). A novel dodecapeptide from a combinatorial synthetic library exhibits potent antifungal activity and synergy with standard antimycotic agents. Int. J. Antimicrob. Agents 29, 73–78. 10.1016/j.ijantimicag.2006.08.038 [DOI] [PubMed] [Google Scholar]
- Ebbensgaard A., Mordhorst H., Overgaard M. T., Nielsen C. G., Aarestrup F. M., Hansen E. B. (2015). Comparative evaluation of the antimicrobial activity of different antimicrobial peptides against a range of pathogenic bacteria. PLoS ONE 10:e0144611. 10.1371/journal.pone.0144611 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Economou N. J., Cocklin S., Loll P. J. (2013). High-resolution crystal structure reveals molecular details of target recognition by bacitracin. Proc. Natl. Acad. Sci. U.S.A. 110, 14207–14212. 10.1073/pnas.1308268110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eisner H., Porzecanski B. (1946). Inactivation of penicillin by zinc salts. Science 103:629 10.1126/science.103.2681.629 [DOI] [PubMed] [Google Scholar]
- Eliopoulos G. M., Willey S., Reiszner E., Spitzer P. G., Caputo G., Moellering R. C., Jr. (1986). In vitro and in vivo activity of LY 146032, a new cyclic lipopeptide antibiotic. Antimicrob. Agents Chemother. 30, 532–535. 10.1128/AAC.30.4.532 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Erdogan-Yildirim Z., Burian A., Manafi M., Zeitlinger M. (2011). Impact of pH on bacterial growth and activity of recent fluoroquinolones in pooled urine. Res. Microbiol 162, 249–252. 10.1016/j.resmic.2011.01.004 [DOI] [PubMed] [Google Scholar]
- Ersoy S. C., Abdelhady W., Li L., Chambers H. F., Xiong Y. Q., Bayer A. S. (2019). Bicarbonate resensitization of methicillin-resistant Staphylococcus aureus to beta-lactam antibiotics. Antimicrob. Agents Chemother. 63:e00496–19. 10.1128/AAC.00496-19 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ersoy S. C., Heithoff D. M., Barnes L. t, Tripp G. K., House J. K., Marth J. D., et al. (2017). Correcting a fundamental flaw in the paradigm for antimicrobial susceptibility testing. EBioMedicine 20, 173–181. 10.1016/j.ebiom.2017.05.026 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Escoll P., Rolando M., BGomez-Valero L., Buchrieser C. (2013). From amoeba to macrophages: exploring the molecular mechanisms of Legionella pneumophila infection in both hosts, in Molecular Mechanisms in Legionella Pathogenesis, ed Hilbi H. (Berlin; Heidelberg: Springer; ), 1–34. [DOI] [PubMed] [Google Scholar]
- EUCAST (2017a). Method for the Determination of Broth Dilution Minimum Inhibitory Concentrations of Antifungal Agents for Yeasts. E.DEF 7.3.1. Available online at: http://www.eucast.org/ (accessed June, 2020).
- EUCAST (2017b). Method for the Determination of Broth Dilution Minimum Inhibitory Concentrations of Antifungal Agents for Conidia Forming Moulds. E.DEF 9.3.1. Available online at: http://www.eucast.org/ (accessed June, 2020). [DOI] [PubMed]
- EUCAST (2018). The European Committee on Antimicrobial Susceptibility Testing: Breakpoint tables for interpretation of MICs. Version 9.0, 2018. Available online at: http://www.eucast.org/ (accessed June, 2020).
- EUCAST (2019a). Antimicrobial Susceptibility Testing: EUCAST Disk Diffusion Method, Version 7.0. Available online at: http://www.eucast.org/
- EUCAST (2019b). The European Committee on Antimicrobial Susceptibility Testing. Breakpoint tables for interpretation of MICs and zone diameters. Version 9.0. Available online at: http://www.eucast.org (accessed June, 2020).
- EUCAST (2019c). MIC Distributions and the Setting of Epidemiological Cut-off (ECOFF) Values, EUCAST SOP 10.1. European Committee on Antimicrobial Susceptibility Testing; ). [Google Scholar]
- European Commission (2017). A European One Health Action Plan against Antimicrobial Resistance (AMR). Available online at: https://ec.europa.eu/health/sites/health/files/antimicrobial_resistance/docs/amr_2017_action-plan.pdf (accessed June, 2020).
- Evans T. J. (2015). Small colony variants of Pseudomonas aeruginosa in chronic bacterial infection of the lung in cystic fibrosis. Future Microbiol. 10, 231–239. 10.2217/fmb.14.107 [DOI] [PubMed] [Google Scholar]
- Ezadi F., Ardebili A., Mirnejad R. (2019). Antimicrobial susceptibility testing for polymyxins: challenges, issues, and recommendations. J. Clin. Microbiol. 57:e01390–18. 10.1128/JCM.01390-18 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Farha M. A., French S., Stokes J. M., Brown E. D. (2018). Bicarbonate alters bacterial susceptibility to antibiotics by targeting the proton motive force. ACS Infect Dis. 4, 382–390. 10.1021/acsinfecdis.7b00194 [DOI] [PubMed] [Google Scholar]
- Ferguson D. B., Botchway C. A. (1979). Circadian variations in the flow rate and composition of whole saliva stimulated by mastication. Arch. Oral Biol. 24, 877–881. 10.1016/0003-9969(79)90212-7 [DOI] [PubMed] [Google Scholar]
- Fernandez-Mazarrasa C., Mazarrasa O., Calvo J., del Arco A., Martinez-Martinez L. (2009). High concentrations of manganese in Mueller-Hinton agar increase MICs of tigecycline determined by Etest. J. Clin. Microbiol. 47, 827–829. 10.1128/JCM.02464-08 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Figura N., Marcolongo R., Cavallo G., Santucci A., Collodel G., Spreafico A., et al. (2012). Polysorbate 80 and Helicobacter pylori: a microbiological and ultrastructural study. BMC Microbiol. 12:217. 10.1186/1471-2180-12-217 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fisher R. A., Gollan B., Helaine S. (2017). Persistent bacterial infections and persister cells. Nat. Rev. Microbiol. 15, 453–464. 10.1038/nrmicro.2017.42 [DOI] [PubMed] [Google Scholar]
- Fjell C. D., Hiss J. A., Hancock R. E., Schneider G. (2011). Designing antimicrobial peptides: form follows function. Nat. Rev. Drug Discov. 11, 37–51. 10.1038/nrd3591 [DOI] [PubMed] [Google Scholar]
- Fothergill A. W., McCarthy D. I., Albataineh M. T., Sanders C., McElmeel M., Wiederhold N. P. (2016). Effects of treated versus untreated polystyrene on caspofungin in vitro activity against candida species. J. Clin. Microbiol. 54, 734–738. 10.1128/JCM.02659-15 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Friedrich C., Scott M. G., Karunaratne N., Yan H., Hancock R. E. (1999). Salt-resistant alpha-helical cationic antimicrobial peptides. Antimicrob. Agents Chemother. 43, 1542–1548. 10.1128/AAC.43.7.1542 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frokjaer S., Otzen D. E. (2005). Protein drug stability: a formulation challenge. Nat. Rev. Drug Discov. 4, 298–306. 10.1038/nrd1695 [DOI] [PubMed] [Google Scholar]
- Gabrilska R. A., Rumbaugh K. P. (2015). Biofilm models of polymicrobial infection. Future Microbiol. 10, 1997–2015. 10.2217/fmb.15.109 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Galdiero E., Siciliano A., Gesuele R., Di Onofrio V., Falanga A., Maione A., et al. (2019). Melittin inhibition and eradication activity for resistant polymicrobial biofilm isolated from a dairy industry after disinfection. Int. J. Microbiol. 2019:4012394. 10.1155/2019/4012394 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gao W., Chua K., Davies J. K., Newton H. J., Seemann T., Harrison P. F., et al. (2010). Two novel point mutations in clinical Staphylococcus aureus reduce linezolid susceptibility and switch on the stringent response to promote persistent infection. PLoS Pathog. 6:e1000944. 10.1371/journal.ppat.1000944 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garcia J. R., Krause A., Schulz S., Rodriguez-Jimenez F. J., Kluver E., Adermann K., et al. (2001). Human beta-defensin 4: a novel inducible peptide with a specific salt-sensitive spectrum of antimicrobial activity. FASEB J. 15, 1819–1821. 10.1096/fj.00-0865fje [DOI] [PubMed] [Google Scholar]
- Garcia-Effron G., Park S., Perlin D. S. (2011). Improved detection of Candida sp. fks hot spot mutants by using the method of the CLSI M27-A3 document with the addition of bovine serum albumin. Antimicrob. Agents Chemother. 55, 2245–2255. 10.1128/AAC.01350-10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gethin G. (2007). The significance of surface pH in chronic wounds. Wounds 3, 52–56. [Google Scholar]
- Ghosh C., Manjunath G. B., Konai M. M., Uppu D. S., Hoque J., Paramanandham K., et al. (2015). Aryl-alkyl-lysines: agents that kill planktonic cells, persister cells, biofilms of MRSA and protect mice from skin-infection. PLoS ONE 10:e0144094. 10.1371/journal.pone.0144094 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giacometti A., Cirioni O., Barchiesi F., Del Prete M. S., Fortuna M., Caselli F., et al. (2000). In vitro susceptibility tests for cationic peptides: comparison of broth microdilution methods for bacteria that grow aerobically. Antimicrob. Agents Chemother. 44, 1694–1696. 10.1128/AAC.44.6.1694-1696.2000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gil F., Lagos-Moraga S., Calderon-Romero P., Pizarro-Guajardo M., Paredes-Sabja D. (2017). Updates on Clostridium difficile spore biology. Anaerobe 45, 3–9. 10.1016/j.anaerobe.2017.02.018 [DOI] [PubMed] [Google Scholar]
- Glaser R., Becker K., von Eiff C., Meyer-Hoffert U., Harder J. (2014). Decreased susceptibility of Staphylococcus aureus small-colony variants toward human antimicrobial peptides. J. Invest. Dermatol. 134, 2347–2350. 10.1038/jid.2014.176 [DOI] [PubMed] [Google Scholar]
- Goldman M. J., Anderson G. M., Stolzenberg E. D., Kari U. P., Zasloff M., Wilson J. M. (1997). Human beta-defensin-1 is a salt-sensitive antibiotic in lung that is inactivated in cystic fibrosis. Cell 88, 553–560. 10.1016/S0092-8674(00)81895-4 [DOI] [PubMed] [Google Scholar]
- Gonzalez-Ruiz A., Seaton R. A., Hamed K. (2016). Daptomycin: an evidence-based review of its role in the treatment of Gram-positive infections. Infect. Drug Resist. 9, 47–58. 10.2147/IDR.S99046 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gour S., Kumar V., Singh A., Gadhave K., Goyal P., Pandey J., et al. (2019). Mammalian antimicrobial peptide protegrin-4 self assembles and forms amyloid-like aggregates: assessment of its functional relevance. J. Pept. Sci. 25:e3151. 10.1002/psc.3151 [DOI] [PubMed] [Google Scholar]
- Grandjean Lapierre S., Phelippeau M., Hakimi C., Didier Q., Reynaud-Gaubert M., Dubus J. C., et al. (2017). Cystic fibrosis respiratory tract salt concentration: an exploratory cohort study. Medicine 96:e8423. 10.1097/MD.0000000000008423 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grassi L., Batoni G., Ostyn L., Rigole P., Van den Bossche S., Rinaldi A. C., et al. (2019). The antimicrobial peptide lin-SB056-1 and its dendrimeric derivative prevent Pseudomonas aeruginosa biofilm formation in physiologically relevant models of chronic infections. Front. Microbiol. 10:198. 10.3389/fmicb.2019.00198 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grassi L., Di Luca M., Maisetta G., Rinaldi A. C., Esin S., Trampuz A., et al. (2017b). Generation of persister cells of Pseudomonas aeruginosa and Staphylococcus aureus by chemical treatment and evaluation of their susceptibility to membrane-targeting agents. Front. Microbiol. 8:1917. 10.3389/fmicb.2017.01917 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grassi L., Maisetta G., Esin S., Batoni G. (2017a). Combination strategies to enhance the efficacy of antimicrobial peptides against bacterial biofilms. Front. Microbiol. 8:2409. 10.3389/fmicb.2017.02409 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greber K. E., Dawgul M. (2017). Antimicrobial peptides under clinical trials. Curr. Top. Med. Chem. 17, 620–628. 10.2174/1568026616666160713143331 [DOI] [PubMed] [Google Scholar]
- Gut I. M., Prouty A. M., Ballard J. D., van der Donk W. A., Blanke S. R. (2008). Inhibition of Bacillus anthracis spore outgrowth by nisin. Antimicrob. Agents Chemother. 52, 4281–4288. 10.1128/AAC.00625-08 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hajdu S., Holinka J., Reichmann S., Hirschl A. M., Graninger W., Presterl E. (2010). Increased temperature enhances the antimicrobial effects of daptomycin, vancomycin, tigecycline, fosfomycin, and cefamandole on staphylococcal biofilms. Antimicrob. Agents Chemother. 54, 4078–4084. 10.1128/AAC.00275-10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hall C. W., Mah T. F. (2017). Molecular mechanisms of biofilm-based antibiotic resistance and tolerance in pathogenic bacteria. FEMS Microbiol. Rev. 41, 276–301. 10.1093/femsre/fux010 [DOI] [PubMed] [Google Scholar]
- Hamamoto K., Kida Y., Zhang Y., Shimizu T., Kuwano K. (2002). Antimicrobial activity and stability to proteolysis of small linear cationic peptides with D-amino acid substitutions. Microbiol. Immunol. 46, 741–749. 10.1111/j.1348-0421.2002.tb02759.x [DOI] [PubMed] [Google Scholar]
- Hancock R. E., Chapple D. S. (1999). Peptide antibiotics. Antimicrob. Agents Chemother. 43, 1317–1323. 10.1128/AAC.43.6.1317 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hancock R. E., Haney E. F., Gill E. E. (2016). The immunology of host defence peptides: beyond antimicrobial activity. Nat. Rev. Immunol. 16, 321–334. 10.1038/nri.2016.29 [DOI] [PubMed] [Google Scholar]
- Hancock R. E., Sahl H. G. (2006). Antimicrobial and host-defense peptides as new anti-infective therapeutic strategies. Nat. Biotechnol. 24, 1551–1557. 10.1038/nbt1267 [DOI] [PubMed] [Google Scholar]
- Haney E. F., Brito-Sanchez Y., Trimble M. J., Mansour S. C., Cherkasov A., Hancock R. E. W. (2018a). Computer-aided discovery of peptides that specifically attack bacterial biofilms. Sci. Rep. 8:1871. 10.1038/s41598-018-19669-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haney E. F., Straus S. K., Hancock R. E. W. (2019). Reassessing the host defense peptide landscape. Front. Chem. 7:43. 10.3389/fchem.2019.00043 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haney E. F., Trimble M. J., Cheng J. T., Valle Q., Hancock R. E. W. (2018b). Critical assessment of methods to quantify biofilm growth and evaluate antibiofilm activity of host defence peptides. Biomolecules 8:29. 10.3390/biom8020029 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haney E. F., Wu B. C., Lee K., Hilchie A. L., Hancock R. E. W. (2017). Aggregation and its influence on the immunomodulatory activity of synthetic innate defense regulator peptides. Cell Chem. Biol. 24, 969–980.e964. 10.1016/j.chembiol.2017.07.010 [DOI] [PubMed] [Google Scholar]
- Hanson M. A., Dostalova A., Ceroni C., Poidevin M., Kondo S., Lemaitre B. (2019). Synergy and remarkable specificity of antimicrobial peptides in vivo using a systematic knockout approach. Elife 8:20 10.7554/eLife.44341.020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hartlieb M., Williams E. G. L., Kuroki A., Perrier S., Locock K. E. S. (2017). Antimicrobial polymers: mimicking amino acid functionali ty, sequence control and three-dimensional structure of host-defen se peptides. Curr. Med. Chem 24, 2115–2140. 10.2174/0929867324666170116122322 [DOI] [PubMed] [Google Scholar]
- Harwig S. S., Waring A., Yang H. J., Cho Y., Tan L., Lehrer R. I. (1996). Intramolecular disulfide bonds enhance the antimicrobial and lytic activities of protegrins at physiological sodium chloride concentrations. Eur. J. Biochem. 240, 352–357. 10.1111/j.1432-1033.1996.0352h.x [DOI] [PubMed] [Google Scholar]
- Hassan M., Kjos M., Nes I. F., Diep D. B., Lotfipour F. (2012). Natural antimicrobial peptides from bacteria: characteristics and potential applications to fight against antibiotic resistance. J. Appl. Microbiol. 113, 723–736. 10.1111/j.1365-2672.2012.05338.x [DOI] [PubMed] [Google Scholar]
- Hayden R. M., Goldberg G. K., Ferguson B. M., Schoeneck M. W., Libardo M. D., Mayeux S. E., et al. (2015). Complementary effects of host defense peptides piscidin 1 and piscidin 3 on DNA and lipid membranes: biophysical insights into contrasting biological activities. J. Phys. Chem. B 119, 15235–15246. 10.1021/acs.jpcb.5b09685 [DOI] [PubMed] [Google Scholar]
- He J., Starr C. G., Wimley W. C. (2015). A lack of synergy between membrane-permeabilizing cationic antimicrobial peptides and conventional antibiotics. Biochim. Biophys. Acta 1848(1 Pt A), 8–15. 10.1016/j.bbamem.2014.09.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Helaine S., Cheverton A. M., Watson K. G., Faure L. M., Matthews S. A., Holden D. W. (2014). Internalization of Salmonella by macrophages induces formation of nonreplicating persisters. Science 343, 204–208. 10.1126/science.1244705 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hertzen E., Johansson L., Kansal R., Hecht A., Dahesh S., Janos M., et al. (2012). Intracellular Streptococcus pyogenes in human macrophages display an altered gene expression profile. PLoS ONE 7:e35218. 10.1371/journal.pone.0035218 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Higgins D., Dworkin J. (2012). Recent progress in Bacillus subtilis sporulation. FEMS Microbiol. Rev. 36, 131–148. 10.1111/j.1574-6976.2011.00310.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Higgins D. L., Chang R., Debabov D. V., Leung J., Wu T., Krause K. M., et al. (2005). Telavancin, a multifunctional lipoglycopeptide, disrupts both cell wall synthesis and cell membrane integrity in methicillin-resistant Staphylococcus aureus. Antimicrob. Agents Chemother. 49, 1127–1134. 10.1128/AAC.49.3.1127-1134.2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hindler J. A., Humphries R. M. (2013). Colistin MIC variability by method for contemporary clinical isolates of multidrug-resistant gram-negative bacilli. J. Clin. Microbiol. 51, 1678–1684. 10.1128/JCM.03385-12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoffman L. R., Deziel E., D'Argenio D. A., Lepine F., Emerson J., McNamara S., et al. (2006). Selection for Staphylococcus aureus small-colony variants due to growth in the presence of Pseudomonas aeruginosa. Proc. Natl. Acad. Sci. U.S.A. 103, 19890–19895. 10.1073/pnas.0606756104 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoiby N., Bjarnsholt T., Givskov M., Molin S., Ciofu O. (2010). Antibiotic resistance of bacterial biofilms. Int. J. Antimicrob. Agents 35, 322–332. 10.1016/j.ijantimicag.2009.12.011 [DOI] [PubMed] [Google Scholar]
- Holmes A. H., Moore L. S., Sundsfjord A., Steinbakk M., Regmi S., Karkey A., et al. (2016). Understanding the mechanisms and drivers of antimicrobial resistance. Lancet 387, 176–187. 10.1016/S0140-6736(15)00473-0 [DOI] [PubMed] [Google Scholar]
- Hombach M., Ochoa C., Maurer F. P., Pfiffner T., Bottger E. C., Furrer R. (2016). Relative contribution of biological variation and technical variables to zone diameter variations of disc diffusion susceptibility testing. J. Antimicrob. Chemother. 71, 141–151. 10.1093/jac/dkv309 [DOI] [PubMed] [Google Scholar]
- Hsu C. H., Chen C., Jou M. L., Lee A. Y., Lin Y. C., Yu Y. P., et al. (2005). Structural and DNA-binding studies on the bovine antimicrobial peptide, indolicidin: evidence for multiple conformations involved in binding to membranes and DNA. Nucleic Acids Res. 33, 4053–4064. 10.1093/nar/gki725 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hua J., Yamarthy R., Felsenstein S., Scott R. W., Markowitz K., Diamond G. (2010). Activity of antimicrobial peptide mimetics in the oral cavity: I. activity against biofilms of Candida albicans. Mol. Oral Microbiol. 25, 418–425. 10.1111/j.2041-1014.2010.00590.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang H. N., Wu C. J., Chen J. Y. (2017). The effects of gamma irradiation sterilization, temperature and pH on the antimicrobial activity of Epinecidin-1. J. Mar. Sci. Technol. 25, 352–357. 10.6119/JMST-016-1229-2 [DOI] [Google Scholar]
- Huang M. B., Gay T. E., Baker C. N., Banerjee S. N., Tenover F. C. (1993). Two percent sodium chloride is required for susceptibility testing of staphylococci with oxacillin when using agar-based dilution methods. J. Clin. Microbiol. 31, 2683–2688. 10.1128/JCM.31.10.2683-2688.1993 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Humphries R. M., Abbott A. N., Hindler J. A. (2019). Understanding and addressing CLSI breakpoint revisions: a primer for clinical laboratories. J. Clin. Microbiol. 57:e00203–19. 10.1128/JCM.00203-19 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Humphries R. M., Pollett S., Sakoulas G. (2013). A current perspective on daptomycin for the clinical microbiologist. Clin. Microbiol. Rev. 26, 759–780. 10.1128/CMR.00030-13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hyre A. N., Kavanagh K., Kock N. D., Donati G. L., Subashchandrabose S. (2017). Copper is a host effector mobilized to urine during urinary tract infection to impair bacterial colonization. Infect. Immun. 85, e01041–16. 10.1128/IAI.01041-16 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Idelevich E. A., Becker K. (2019). How to accelerate antimicrobial susceptibility testing. Clin. Microbiol. Infect. 25, 1347–1355. 10.1016/j.cmi.2019.04.025 [DOI] [PubMed] [Google Scholar]
- Imler J. L., Bulet P. (2005). Antimicrobial peptides in Drosophila: structures, activities and gene regulation. Chem. Immunol. Allergy 86, 1–21. 10.1159/000086648 [DOI] [PubMed] [Google Scholar]
- Infante V. V., Miranda-Olvera A. D., De Leon-Rodriguez L. M., Anaya-Velazquez F., Rodriguez M. C., Avila E. E. (2011). Effect of the antimicrobial peptide tritrpticin on the in vitro viability and growth of Trichomonas vaginalis. Curr. Microbiol. 62, 301–306. 10.1007/s00284-010-9709-z [DOI] [PubMed] [Google Scholar]
- ISO (2019). Susceptibility Testing of Infectious Agents and Evaluation of Performance of Antimicrobial Susceptibility Test Devices - Part 1: Broth Micro-Dilution Reference Method for Testing the in vitro Activity of Antimicrobial Agents Against Rapidly Growing Aerobic Bacteria Involved in Infectious Diseases. ISO 20776-1:2019. Geneva: International Organization for Standardization. [Google Scholar]
- Jenssen H., Hamill P., Hancock R. E. (2006). Peptide antimicrobial agents. Clin. Microbiol. Rev. 19, 491–511. 10.1128/CMR.00056-05 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jepson A. K., Schwarz-Linek J., Ryan L., Ryadnov M. G., Poon W. C. (2016). What Is the 'Minimum Inhibitory Concentration' (MIC) of pexiganan acting on Escherichia coli?-a cautionary case study. Adv. Exp. Med. Biol. 915, 33–48. 10.1007/978-3-319-32189-9_4 [DOI] [PubMed] [Google Scholar]
- Jezowska-Bojczuk M., Stokowa-Soltys K. (2018). Peptides having antimicrobial activity and their complexes with transition metal ions. Eur. J. Med. Chem. 143, 997–1009. 10.1016/j.ejmech.2017.11.086 [DOI] [PubMed] [Google Scholar]
- Ji S., Li W., Zhang L., Zhang Y., Cao B. (2014). Cecropin A-melittin mutant with improved proteolytic stability and enhanced antimicrobial activity against bacteria and fungi associated with gastroenteritis in vitro. Biochem. Biophys. Res. Commun. 451, 650–655. 10.1016/j.bbrc.2014.08.044 [DOI] [PubMed] [Google Scholar]
- Jiang Z., Vasil A. I., Hale J., Hancock R. E., Vasil M. L., Hodges R. S. (2009). Effects of net charge and the number of positively charged residues on the biological activity of amphipathic alpha-helical cationic antimicrobial peptides. Adv. Exp. Med. Biol. 611, 561–562. 10.1007/978-0-387-73657-0_246 [DOI] [PubMed] [Google Scholar]
- Jiao K., Gao J., Zhou T., Yu J., Song H., Wei Y., et al. (2019). Isolation and purification of a novel antimicrobial peptide from Porphyra yezoensis. J. Food Biochem. 43:e12864. 10.1111/jfbc.12864 [DOI] [PubMed] [Google Scholar]
- Johansson J., Gudmundsson G. H., Rottenberg M. E., Berndt K. D., Agerberth B. (1998). Conformation-dependent antibacterial activity of the naturally occurring human peptide LL-37. J. Biol. Chem. 273, 3718–3724. 10.1074/jbc.273.6.3718 [DOI] [PubMed] [Google Scholar]
- Johns B. E., Purdy K. J., Tucker N. P., Maddocks S. E. (2015). Phenotypic and genotypic characteristics of small colony variants and their role in chronic infection. Microbiol. Insights 8, 15–23. 10.4137/MBI.S25800 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joo H. S., Fu C. I., Otto M. (2016). Bacterial strategies of resistance to antimicrobial peptides. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 371:20150292. 10.1098/rstb.2015.0292 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Juliano S. A., Pierce S., deMayo J. A., Balunas M. J., Angeles-Boza A. M. (2017). Exploration of the innate immune system of styela clava: Zn(2+) binding enhances the antimicrobial activity of the tunicate peptide clavanin A. Biochemistry 56, 1403–1414. 10.1021/acs.biochem.6b01046 [DOI] [PubMed] [Google Scholar]
- Jung H. S., Ehlers M. M., Lombaard H., Redelinghuys M. J., Kock M. M. (2017). Etiology of bacterial vaginosis and polymicrobial biofilm formation. Crit. Rev. Microbiol. 43, 651–667. 10.1080/1040841X.2017.1291579 [DOI] [PubMed] [Google Scholar]
- Kahl B., Herrmann M., Everding A. S., Koch H. G., Becker K., Harms E., et al. (1998). Persistent infection with small colony variant strains of Staphylococcus aureus in patients with cystic fibrosis. J. Infect. Dis 177, 1023–1029. 10.1086/515238 [DOI] [PubMed] [Google Scholar]
- Kahl B. C. (2014). Small colony variants (SCVs) of Staphylococcus aureus–a bacterial survival strategy. Infect. Genet. Evol. 21, 515–522. 10.1016/j.meegid.2013.05.016 [DOI] [PubMed] [Google Scholar]
- Kahl B. C., Becker K., Loffler B. (2016). Clinical significance and pathogenesis of staphylococcal small colony variants in persistent infections. Clin. Microbiol. Rev. 29, 401–427. 10.1128/CMR.00069-15 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kahlmeter G. (2015). The 2014 Garrod Lecture: EUCAST - are we heading towards international agreement? J. Antimicrob. Chemother. 70, 2427–2439. 10.1093/jac/dkv145 [DOI] [PubMed] [Google Scholar]
- Kang H. K., Kim C., Seo C. H., Park Y. (2017). The therapeutic applications of antimicrobial peptides (AMPs): a patent review. J. Microbiol. 55, 1–12. 10.1007/s12275-017-6452-1 [DOI] [PubMed] [Google Scholar]
- Karaiskos I., Souli M., Galani I., Giamarellou H. (2017). Colistin: still a lifesaver for the 21st century? Expert Opin. Drug Metab. Toxicol. 13, 59–71. 10.1080/17425255.2017.1230200 [DOI] [PubMed] [Google Scholar]
- Karala A. R., Ruddock L. W. (2010). Bacitracin is not a specific inhibitor of protein disulfide isomerase. FEBS J. 277, 2454–2462. 10.1111/j.1742-4658.2010.07660.x [DOI] [PubMed] [Google Scholar]
- Karvanen M., Malmberg C., Lagerback P., Friberg L. E., Cars O. (2017). Colistin is extensively lost during standard in vitro experimental conditions. Antimicrob. Agents Chemother. 61:e00857–17. 10.1128/AAC.00857-17 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaushik K. S., Stolhandske J., Shindell O., Smyth H. D., Gordon V. D. (2016). Tobramycin and bicarbonate synergise to kill planktonic Pseudomonas aeruginosa, but antagonise to promote biofilm survival. Biofilms Microbiomes 2:16006. 10.1038/npjbiofilms.2016.6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kavanagh A., Ramu S., Gong Y., Cooper M. A., Blaskovich M. A. T. (2019). Effects of microplate type and broth additives on microdilution MIC susceptibility assays. Antimicrob. Agents Chemother. 63:e01760–18. 10.1128/AAC.01760-18 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kellum J. A. (2000). Determinants of blood pH in health and disease. Crit Care. 4, 6–14. 10.1186/cc644 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim D. J., Lee Y. W., Park M. K., Shin J. R., Lim K. J., Cho J. H., et al. (2014). Efficacy of the designer antimicrobial peptide SHAP1 in wound healing and wound infection. Amino Acids 46, 2333–2343. 10.1007/s00726-014-1804-1 [DOI] [PubMed] [Google Scholar]
- Kim J. H., Kim T. S., Song S. H., Choi J., Han S., Kim D. Y., et al. (2018). Direct rapid antibiotic susceptibility test (dRAST) for blood culture and its potential usefulness in clinical practice. J. Med. Microbiol. 67, 325–331. 10.1099/jmm.0.000678 [DOI] [PubMed] [Google Scholar]
- Kim W., Hecht M. H. (2006). Generic hydrophobic residues are sufficient to promote aggregation of the Alzheimer's Abeta42 peptide. Proc. Natl. Acad. Sci. U.S.A. 103, 15824–15829. 10.1073/pnas.0605629103 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knappe D., Henklein P., Hoffmann R., Hilpert K. (2010). Easy strategy to protect antimicrobial peptides from fast degradation in serum. Antimicrob. Agents Chemother. 54, 4003–4005. 10.1128/AAC.00300-10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koo H., Allan R. N., Howlin R. P., Stoodley P., Hall-Stoodley L. (2017). Targeting microbial biofilms: current and prospective therapeutic strategies. Nat. Rev. Microbiol. 15, 740–755. 10.1038/nrmicro.2017.99 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koo H. B., Seo J. (2019). Antimicrobial peptides under clinical investigation. Peptide Sci. 111:e24122 10.1002/pep2.24122 [DOI] [Google Scholar]
- Koo S. P., Bayer A. S., Sahl H. G., Proctor R. A., Yeaman M. R. (1996). Staphylocidal action of thrombin-induced platelet microbicidal protein is not solely dependent on transmembrane potential. Infect. Immun. 64, 1070–1074. 10.1128/IAI.64.3.1070-1074.1996 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kosmidis C., Denning D. W. (2015). The clinical spectrum of Pulmonary aspergillosis. Thorax 70, 270–277. 10.1136/thoraxjnl-2014-206291 [DOI] [PubMed] [Google Scholar]
- Kristensen K., Henriksen J. R., Andresen T. L. (2015). Adsorption of cationic peptides to solid surfaces of glass and plastic. PLoS ONE 10:e0122419. 10.1371/journal.pone.0122419 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumar A., Ellis P., Arabi Y., Roberts D., Light B., Parrillo J. E., et al. (2009). Initiation of inappropriate antimicrobial therapy results in a fivefold reduction of survival in human septic shock. Chest 136, 1237–1248. 10.1378/chest.09-0087 [DOI] [PubMed] [Google Scholar]
- Kumar P., Kizhakkedathu J. N., Straus S. K. (2018). Antimicrobial peptides: diversity, mechanism of action and strategies to improve the activity and biocompatibility in vivo. Biomolecules 8:4. 10.3390/biom8010004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumaraswamy M., Lin L., Olson J., Sun C. F., Nonejuie P., Corriden R., et al. (2016). Standard susceptibility testing overlooks potent azithromycin activity and cationic peptide synergy against MDR Stenotrophomonas maltophilia. J. Antimicrob. Chemother. 71, 1264–1269. 10.1093/jac/dkv487 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kunin C. M., Edmondson W. P. (1968). Inhibitor of antibiotics in bacteriologic agar. Proc. Soc. Exp. Biol. Med. 129, 118–122. 10.3181/00379727-129-33264 [DOI] [PubMed] [Google Scholar]
- Kwan B. W., Valenta J. A., Benedik M. J., Wood T. K. (2013). Arrested protein synthesis increases persister-like cell formation. Antimicrob. Agents Chemother. 57, 1468–1473. 10.1128/AAC.02135-12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lambers H., Piessens S., Bloem A., Pronk H., Finkel P. (2006). Natural skin surface pH is on average below 5, which is beneficial for its resident flora. Int. J. Cosmet. Sci. 28, 359–370. 10.1111/j.1467-2494.2006.00344.x [DOI] [PubMed] [Google Scholar]
- Lau J. L., Dunn M. K. (2018). Therapeutic peptides: Historical perspectives, current development trends, and future directions. Bioorg. Med. Chem. 26, 2700–2707. 10.1016/j.bmc.2017.06.052 [DOI] [PubMed] [Google Scholar]
- Le C. F., Fang C. M., Sekaran S. D. (2017). Intracellular targeting mechanisms by antimicrobial peptides. Antimicrob. Agents Chemother 61:e02340–16. 10.1128/AAC.02340-16 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lecaille F., Lalmanach G., Andrault P. M. (2016). Antimicrobial proteins and peptides in human lung diseases: a friend and foe partnership with host proteases. Biochimie 122, 151–168. 10.1016/j.biochi.2015.08.014 [DOI] [PubMed] [Google Scholar]
- Lee A. C., Harris J. L., Khanna K. K., Hong J. H. (2019). A comprehensive review on current advances in peptide drug development and design. Int. J. Mol. Sci. 20:2383. 10.3390/ijms20102383 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee C. S., Tung W. C., Lin Y. H. (2014). Deletion of the carboxyl-terminal residue disrupts the amino-terminal folding, self-association, and thermal stability of an amphipathic antimicrobial peptide. J. Pept. Sci. 20, 438–445. 10.1002/psc.2635 [DOI] [PubMed] [Google Scholar]
- Lee E. Y., Wong G. C. L., Ferguson A. L. (2018). Machine learning-enabled discovery and design of membrane-active peptides. Bioorg. Med. Chem. 26, 2708–2718. 10.1016/j.bmc.2017.07.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee I. H., Cho Y., Lehrer R. I. (1997). Effects of pH and salinity on the antimicrobial properties of clavanins. Infect. Immun. 65, 2898–2903. 10.1128/IAI.65.7.2898-2903.1997 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leggett H. C., Cornwallis C. K., Buckling A., West S. A. (2017). Growth rate, transmission mode and virulence in human pathogens. Philos. Trans. R. Soc. Lond. B Biol. Sci. 372:94. 10.1098/rstb.2016.0094 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lehrer R. I., Rosenman M., Harwig S. S., Jackson R., Eisenhauer P. (1991). Ultrasensitive assays for endogenous antimicrobial polypeptides. J. Immunol. Methods 137, 167–173. 10.1016/0022-1759(91)90021-7 [DOI] [PubMed] [Google Scholar]
- Lepore C., Silver L., Theuretzbacher U., Thomas J., Visi D. (2019). The small-molecule antibiotics pipeline: 2014-2018. Nat. Rev. Drug Discov. 18:739. 10.1038/d41573-019-00130-8 [DOI] [PubMed] [Google Scholar]
- Lesniak W., Harris W. R., Kravitz J. Y., Schacht J., Pecoraro V. L. (2003). Solution chemistry of copper(II)-gentamicin complexes: relevance to metal-related aminoglycoside toxicity. Inorg. Chem. 42, 1420–1429. 10.1021/ic025965t [DOI] [PubMed] [Google Scholar]
- Letscher-Bru V., Obszynski C. M., Samsoen M., Sabou M., Waller J., Candolfi E. (2013). Antifungal activity of sodium bicarbonate against fungal agents causing superficial infections. Mycopathologia 175, 153–158. 10.1007/s11046-012-9583-2 [DOI] [PubMed] [Google Scholar]
- Levitz S. M., Selsted M. E., Ganz T., Lehrer R. I., Diamond R. D. (1986). In vitro killing of spores and hyphae of Aspergillus fumigatus and Rhizopus oryzae by rabbit neutrophil cationic peptides and bronchoalveolar macrophages. J. Infect. Dis. 154, 483–489. 10.1093/infdis/154.3.483 [DOI] [PubMed] [Google Scholar]
- Li H., Sun S. R., Yap J. Q., Chen J. H., Qian Q. (2016). 0.9% saline is neither normal nor physiological. J. Zhejiang Univ. Sci. B 17, 181–187. 10.1631/jzus.B1500201 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Libardo M. D., Cervantes J. L., Salazar J. C., Angeles-Boza A. M. (2014). Improved bioactivity of antimicrobial peptides by addition of amino-terminal copper and nickel (ATCUN) binding motifs. ChemMedChem 9, 1892–1901. 10.1002/cmdc.201402033 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Libardo M. D., Gorbatyuk V. Y., Angeles-Boza A. M. (2016). Central role of the copper-binding motif in the complex mechanism of action of ixosin: enhancing oxidative damage and promoting synergy with ixosin B. ACS Infect. Dis. 2, 71–81. 10.1021/acsinfecdis.5b00140 [DOI] [PubMed] [Google Scholar]
- Libardo M. D., Paul T. J., Prabhakar R., Angeles-Boza A. M. (2015). Hybrid peptide ATCUN-sh-Buforin: influence of the ATCUN charge and stereochemistry on antimicrobial activity. Biochimie 113, 143–155. 10.1016/j.biochi.2015.04.008 [DOI] [PubMed] [Google Scholar]
- Libardo M. D. J., Angeles-Boza A. M. (2014). Bioinorganic chemistry of antimicrobial and host-defense peptides. Comments Inorg. Chem. 34, 42–58. 10.1080/02603594.2014.960923 [DOI] [Google Scholar]
- Libardo M. D. J., Bahar A. A., Ma B., Fu R., McCormick L. E., Zhao J., et al. (2017). Nuclease activity gives an edge to host-defense peptide piscidin 3 over piscidin 1, rendering it more effective against persisters and biofilms. FEBS J. 284, 3662–3683. 10.1111/febs.14263 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin A., Horvath D., Afonina V., Marcou G., Reymond J. L., Varnek A. (2018). Mapping of the available chemical space versus the chemical universe of lead-like compounds. ChemMedChem 13, 540–554. 10.1002/cmdc.201700561 [DOI] [PubMed] [Google Scholar]
- Lin L., Nonejuie P., Munguia J., Hollands A., Olson J., Dam Q., et al. (2015). Azithromycin synergizes with cationic antimicrobial peptides to exert bactericidal and therapeutic activity against highly multidrug-resistant Gram-negative bacterial pathogens. EBioMedicine 2, 690–698. 10.1016/j.ebiom.2015.05.021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lipkin R., Lazaridis T. (2017). Computational studies of peptide-induced membrane pore formation. Philos. Trans. R Soc. Lond. B. Biol. Sci. 372:0219. 10.1098/rstb.2016.0219 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lisanby M. W., Swiecki M. K., Dizon B. L., Pflughoeft K. J., Koehler T. M., Kearney J. F. (2008). Cathelicidin administration protects mice from Bacillus anthracis spore challenge. J. Immunol. 181, 4989–5000. 10.4049/jimmunol.181.7.4989 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu V. X., Fielding-Singh V., Greene J. D., Baker J. M., Iwashyna T. J., Bhattacharya J., et al. (2017). The timing of early antibiotics and hospital mortality in sepsis. Am. J. Respir. Crit. Care Med. 196, 856–863. 10.1164/rccm.201609-1848OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu W., Hansen J. N. (1993). The antimicrobial effect of a structural variant of subtilin against outgrowing Bacillus cereus T spores and vegetative cells occurs by different mechanisms. Appl. Environ. Microbiol. 59, 648–651. 10.1128/AEM.59.2.648-651.1993 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lockhart S. R., Ghannoum M. A., Alexander B. D. (2017). Establishment and use of epidemiological cutoff values for molds and yeasts by use of the clinical and laboratory standards institute M57 standard. J. Clin. Microbiol. 55, 1262–1268. 10.1128/JCM.02416-16 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lopes S. P., Azevedo N. F., Pereira M. O. (2015). Microbiome in cystic fibrosis: shaping polymicrobial interactions for advances in antibiotic therapy. Crit. Rev. Microbiol. 41, 353–365. 10.3109/1040841X.2013.847898 [DOI] [PubMed] [Google Scholar]
- Lopez-Garcia B., Lee P. H., Yamasaki K., Gallo R. L. (2005). Anti-fungal activity of cathelicidins and their potential role in Candida albicans skin infection. J. Invest. Dermatol. 125, 108–115. 10.1111/j.0022-202X.2005.23713.x [DOI] [PubMed] [Google Scholar]
- Luca V., Stringaro A., Colone M., Pini A., Mangoni M. L. (2013). Esculentin(1-21), an amphibian skin membrane-active peptide with potent activity on both planktonic and biofilm cells of the bacterial pathogen Pseudomonas aeruginosa. Cell. Mol. Life Sci. 70, 2773–2786. 10.1007/s00018-013-1291-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luo Y., Bolt H. L., Eggimann G. A., McAuley D. F., McMullan R., Curran T., et al. (2017). Peptoid efficacy against polymicrobial biofilms determined by using propidium monoazide-modified quantitative PCR. Chembiochem 18, 111–118. 10.1002/cbic.201600381 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Macia M. D., Rojo-Molinero E., Oliver A. (2014). Antimicrobial susceptibility testing in biofilm-growing bacteria. Clin. Microbiol. Infect. 20, 981–990. 10.1111/1469-0691.12651 [DOI] [PubMed] [Google Scholar]
- Magana M., Sereti C., Ioannidis A., Mitchell C. A., Ball A. R., Magiorkinis E., et al. (2018). Options and limitations in clinical investigation of bacterial biofilms. Clin. Microbiol. Rev. 31:e00084–16. 10.1128/CMR.00084-16 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Magiorakos A. P., Srinivasan A., Carey R. B., Carmeli Y., Falagas M. E., Giske C. G., et al. (2012). Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: an international expert proposal for interim standard definitions for acquired resistance. Clin. Microbiol. Infect. 18, 268–281. 10.1111/j.1469-0691.2011.03570.x [DOI] [PubMed] [Google Scholar]
- Mahlapuu M., Hakansson J., Ringstad L., Bjorn C. (2016). Antimicrobial peptides: an emerging category of therapeutic agents. Front. Cell. Infect. Microbiol. 6:194. 10.3389/fcimb.2016.00194 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mai J., Tian X. L., Gallant J. W., Merkley N., Biswas Z., Syvitski R., et al. (2011). A novel target-specific, salt-resistant antimicrobial peptide against the cariogenic pathogen Streptococcus mutans. Antimicrob. Agents Chemother. 55, 5205–5213. 10.1128/AAC.05175-11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maisetta G., Grassi L., Esin S., Serra I., Scorciapino M. A., Rinaldi A. C., et al. (2017). The semi-synthetic peptide lin-SB056-1 in combination with EDTA exerts strong antimicrobial and antibiofilm activity against Pseudomonas aeruginosa in conditions mimicking cystic fibrosis sputum. Int. J. Mol. Sci. 18:1994. 10.3390/ijms18091994 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malanovic N., Lohner K. (2016). Gram-positive bacterial cell envelopes: the impact on the activity of antimicrobial peptides. Biochim. Biophys. Acta 1858, 936–946. 10.1016/j.bbamem.2015.11.004 [DOI] [PubMed] [Google Scholar]
- Malik E., Dennison S. R., Harris F., Phoenix D. A. (2016). pH dependent antimicrobial peptides and proteins, their mechanisms of action and potential as therapeutic agents. Pharmaceuticals 9:67. 10.3390/ph9040067 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mangoni M. L., Carotenuto A., Auriemma L., Saviello M. R., Campiglia P., Gomez-Monterrey I., et al. (2011). Structure-activity relationship, conformational and biological studies of temporin L analogues. J. Med. Chem. 54, 1298–1307. 10.1021/jm1012853 [DOI] [PubMed] [Google Scholar]
- Mangoni M. L., McDermott A. M., Zasloff M. (2016). Antimicrobial peptides and wound healing: biological and therapeutic considerations. Exp. Dermatol 25, 167–173. 10.1111/exd.12929 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mansour S. C., Pena O. M., Hancock R. E. (2014). Host defense peptides: front-line immunomodulators. Trends Immunol. 35, 443–450. 10.1016/j.it.2014.07.004 [DOI] [PubMed] [Google Scholar]
- Mardirossian M., Grzela R., Giglione C., Meinnel T., Gennaro R., Mergaert P., et al. (2014). The host antimicrobial peptide Bac71-35 binds to bacterial ribosomal proteins and inhibits protein synthesis. Chem. Biol. 21, 1639–1647. 10.1016/j.chembiol.2014.10.009 [DOI] [PubMed] [Google Scholar]
- Matejuk A., Leng Q., Begum M. D., Woodle M. C., Scaria P., Chou S. T., et al. (2010). Peptide-based antifungal therapies against emerging infections. Drugs Future 35:197. 10.1358/dof.2010.035.03.1452077 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matuschek E., Ahman J., Webster C., Kahlmeter G. (2018). Antimicrobial susceptibility testing of colistin - evaluation of seven commercial MIC products against standard broth microdilution for Escherichia coli, Klebsiella pneumoniae, Pseudomonas aeruginosa, and Acinetobacter spp. Clin. Microbiol. Infect. 24, 865–870. 10.1016/j.cmi.2017.11.020 [DOI] [PubMed] [Google Scholar]
- Mayr A., Aigner M., Lass-Florl C. (2011). Anidulafungin for the treatment of invasive candidiasis. Clin. Microbiol. Infect. 17 (Suppl. 1), 1–12. 10.1111/j.1469-0691.2010.03448.x [DOI] [PubMed] [Google Scholar]
- McKee T. J., Komarova S. V. (2017). Is it time to reinvent basic cell culture medium? Am. J. Physiol. Cell Physiol. 312, C624–C626. 10.1152/ajpcell.00336.2016 [DOI] [PubMed] [Google Scholar]
- Melino S., Santone C., Di Nardo P., Sarkar B. (2014). Histatins: salivary peptides with copper(II)- and zinc(II)-binding motifs: perspectives for biomedical applications. FEBS J. 281, 657–672. 10.1111/febs.12612 [DOI] [PubMed] [Google Scholar]
- Mercer D. K., Katvars L. K., Hewitt F., Smith D. W., Robertson J., O'Neil D. A. (2017). NP108, an antimicrobial polymer with activity against methicillin- and mupirocin-resistant Staphylococcus aureus. Antimicrob. Agents Chemother 61:e00502–17. 10.1128/AAC.00502-17 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mercer D. K., O'Neil D. A. (2013). Peptides as the next generation of anti-infectives. Future Med. Chem. 5, 315–337. 10.4155/fmc.12.213 [DOI] [PubMed] [Google Scholar]
- Mercer D. K., Stewart C. S., Miller L., Robertson J., Duncan V. M. S., O'Neil D. A. (2019). Improved methods for assessing therapeutic potential of antifungal agents against dermatophytes and their application in the development of NP213, a novel onychomycosis therapy candidate. Antimicrob. Agents Chemother. 63:e02117–18. 10.1128/AAC.02117-18 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mihai M. M., Holban A. M., Giurcaneanu C., Popa L. G., Oanea R. M., Lazar V., et al. (2015). Microbial biofilms: impact on the pathogenesis of periodontitis, cystic fibrosis, chronic wounds and medical device-related infections. Curr. Top. Med. Chem. 15, 1552–1576. 10.2174/1568026615666150414123800 [DOI] [PubMed] [Google Scholar]
- Mirani Z. A., Aziz M., Khan S. I. (2015). Small colony variants have a major role in stability and persistence of Staphylococcus aureus biofilms. J. Antibiot. 68, 98–105. 10.1038/ja.2014.115 [DOI] [PubMed] [Google Scholar]
- Mogi T., Kita K. (2009). Gramicidin S and polymyxins: the revival of cationic cyclic peptide antibiotics. Cell. Mol. Life Sci. 66, 3821–3826. 10.1007/s00018-009-0129-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mohanram H., Bhattacharjya S. (2016). Salt-resistant short antimicrobial peptides. Biopolymers 106, 345–356. 10.1002/bip.22819 [DOI] [PubMed] [Google Scholar]
- Moskowitz S. M., Foster J. M., Emerson J., Burns J. L. (2004). Clinically feasible biofilm susceptibility assay for isolates of Pseudomonas aeruginosa from patients with cystic fibrosis. J. Clin. Microbiol. 42, 1915–1922. 10.1128/JCM.42.5.1915-1922.2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mouton J. W., Meletiadis J., Voss A., Turnidge J. (2019). Variation of MIC measurements: the contribution of strain and laboratory variability to measurement precision-authors' response. J. Antimicrob. Chemother. 74, 1761–1762. 10.1093/jac/dkz142 [DOI] [PubMed] [Google Scholar]
- Mouton J. W., Muller A. E., Canton R., Giske C. G., Kahlmeter G., Turnidge J. (2018). MIC-based dose adjustment: facts and fables. J. Antimicrob. Chemother. 73, 564–568. 10.1093/jac/dkx427 [DOI] [PubMed] [Google Scholar]
- Murdoch C., Muthana M., Lewis C. E. (2005). Hypoxia regulates macrophage functions in inflammation. J. Immunol. 175, 6257–6263. 10.4049/jimmunol.175.10.6257 [DOI] [PubMed] [Google Scholar]
- Naafs M. A. B. (2018). The antimicrobial peptides: ready for clinical trials? Biomed. J. Sci. Tech. Res. 7, 6038–6042. 10.26717/BJSTR.2018.07.001536 [DOI] [Google Scholar]
- Nagant C., Pitts B., Kamran N., Vandenbranden M., Bolscher J. G., Stewart P. S., et al. (2012). Identification of peptides derived from the human antimicrobial peptide LL-37 active against the biofilms formed by Pseudomonas aeruginosa using a library of truncated fragments. Antimicrob. Agents Chemother. 56, 5678–5708. 10.1128/AAC.00918-12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Navarro P. G., Blazquez I. H., Osso B. Q., Martinez de las Parras P. J., Puentedura M. I., Garcia A. A. (2003). Penicillin degradation catalysed by Zn(II) ions in methanol. Int. J. Biol. Macromol. 33, 159–166. 10.1016/S0141-8130(03)00081-3 [DOI] [PubMed] [Google Scholar]
- Nesuta O., Budesinsky M., Hadravova R., Monincova L., Humpolickova J., Cerovsky V. (2017). How proteases from Enterococcus faecalis contribute to its resistance to short alpha-helical antimicrobial peptides. Pathog. Dis 75:ftx091. 10.1093/femspd/ftx091 [DOI] [PubMed] [Google Scholar]
- Nguyen L. T., Chau J. K., Perry N. A., de Boer L., Zaat S. A., Vogel H. J. (2010). Serum stabilities of short tryptophan- and arginine-rich antimicrobial peptide analogs. PLoS ONE 5:e12684. 10.1371/journal.pone.0012684 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nickel D., Busch M., Mayer D., Hagemann B., Knoll V., Stenger S. (2012). Hypoxia triggers the expression of human beta defensin 2 and antimicrobial activity against Mycobacterium tuberculosis in human macrophages. J. Immunol. 188, 4001–4007. 10.4049/jimmunol.1100976 [DOI] [PubMed] [Google Scholar]
- Nizet V. (2017). The accidental orthodoxy of Drs. Mueller and Hinton. EBioMedicine 22, 26–27. 10.1016/j.ebiom.2017.07.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nuding S., Frasch T., Schaller M., Stange E. F., Zabel L. T. (2014). Synergistic effects of antimicrobial peptides and antibiotics against Clostridium difficile. Antimicrob. Agents Chemother. 58, 5719–5725. 10.1128/AAC.02542-14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nuding S., Zabel L. T., Enders C., Porter E., Fellermann K., Wehkamp J., et al. (2009). Antibacterial activity of human defensins on anaerobic intestinal bacterial species: a major role of HBD-3. Microbes Infect. 11, 384–393. 10.1016/j.micinf.2009.01.001 [DOI] [PubMed] [Google Scholar]
- Oesterreicher Z., Eberl S., Nussbaumer-Proell A., Peilensteiner T., Zeitlinger M. (2019). Impact of different pathophysiological conditions on antimicrobial activity of glycopeptides in vitro. Clin. Microbiol. Infect. 25, 759 e751–759.e757. 10.1016/j.cmi.2018.09.004 [DOI] [PubMed] [Google Scholar]
- Oh H., Hedberg M., Wade D., Edlund C. (2000). Activities of synthetic hybrid peptides against anaerobic bacteria: aspects of methodology and stability. Antimicrob. Agents Chemother. 44, 68–72. 10.1128/AAC.44.1.68-72.2000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oludiran A., Courson D. S., Stuart M. D., Radwan A. R., Poutsma J. C., Cotten M. L., et al. (2019). How oxygen availability affects the antimicrobial efficacy of host defense peptides: lessons learned from studying the copper-binding peptides piscidins 1 and 3. Int. J. Mol. Sci. 20:5289. 10.3390/ijms20215289 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Omardien S., Drijfhout J. W., Zaat S. A., Brul S. (2018). Cationic amphipathic antimicrobial peptides perturb the inner membrane of germinated spores thus inhibiting their outgrowth. Front. Microbiol. 9:2277. 10.3389/fmicb.2018.02277 [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Neill J. (2014). Antimicrobial resistance: tackling a crisis for the health and wealth of nations, in The Review on Antimicrobial Resistance. London: HM Government, 1–20. [Google Scholar]
- O'Neill J. (2016). Tackling drug-resistant infections globally: final report and recommendations, in The Review on Antimicrobial Resistance. London: HM Government, 1–81. [Google Scholar]
- Ong C.-L., Walker M. J., McEwan A. G. (2015). Zinc disrupts central carbon metabolism and capsule biosynthesis in Streptococcus pyogenes. Sci. Rep. 5:10799. 10.1038/srep10799 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ong P. Y., Ohtake T., Brandt C., Strickland I., Boguniewicz M., Ganz T., et al. (2002). Endogenous antimicrobial peptides and skin infections in atopic dermatitis. N. Engl. J. Med. 347, 1151–1160. 10.1056/NEJMoa021481 [DOI] [PubMed] [Google Scholar]
- Orazi G., O'Toole G. A. (2019). 'It takes a village': mechanisms underlying antimicrobial recalcitrance of polymicrobial biofilms. J. Bacteriol. 202:e00530–19. 10.1128/JB.00530-19 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Orazi G., Ruoff K. L., O'Toole G. A. (2019). Pseudomonas aeruginosa increases the sensitivity of biofilm-grown Staphylococcus aureus to membrane-targeting antiseptics and antibiotics. MBio 10:e01501–19. 10.1128/mBio.01501-19 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oren Z., Lerman J. C., Gudmundsson G. H., Agerberth B., Shai Y. (1999). Structure and organization of the human antimicrobial peptide LL-37 in phospholipid membranes: relevance to the molecular basis for its non-cell-selective activity. Biochem. J. 341(Pt 3), 501–513. 10.1042/bj3410501 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Orioni B., Bocchinfuso G., Kim J. Y., Palleschi A., Grande G., Bobone S., et al. (2009). Membrane perturbation by the antimicrobial peptide PMAP-23: a fluorescence and molecular dynamics study. Biochim. Biophys. Acta 1788, 1523–1533. 10.1016/j.bbamem.2009.04.013 [DOI] [PubMed] [Google Scholar]
- Ostrosky-Zeichner L., Andes D. (2017). The role of in vitro susceptibility testing in the management of candida and aspergillus. J. Infect. Dis. 216(Suppl. 3), S452–S457. 10.1093/infdis/jix239 [DOI] [PubMed] [Google Scholar]
- O'Toole G. A. (2011). Microtiter dish biofilm formation assay. J. Vis. Exp. 30:2437 10.3791/2437 [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Toole G. A., Kolter R. (1998). Initiation of biofilm formation in Pseudomonas fluorescens WCS365 proceeds via multiple, convergent signalling pathways: a genetic analysis. Mol. Microbiol. 28, 449–461. 10.1046/j.1365-2958.1998.00797.x [DOI] [PubMed] [Google Scholar]
- Otvos L., Cudic M. (2007). Broth microdilution antibacterial assay of peptides. Methods Mol. Biol. 386, 309–320. 10.1007/978-1-59745-430-8_12 [DOI] [PubMed] [Google Scholar]
- Overhage J., Campisano A., Bains M., Torfs E. C., Rehm B. H., Hancock R. E. (2008). Human host defense peptide LL-37 prevents bacterial biofilm formation. Infect. Immun. 76, 4176–4182. 10.1128/IAI.00318-08 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pane K., Cafaro V., Avitabile A., Torres M. T., Vollaro A., De Gregorio E., et al. (2018). Identification of novel cryptic multifunctional antimicrobial peptides from the human stomach enabled by a computational-experimental platform. ACS Synth. Biol. 7, 2105–2115. 10.1021/acssynbio.8b00084 [DOI] [PubMed] [Google Scholar]
- Park C. B., Kim H. S., Kim S. C. (1998). Mechanism of action of the antimicrobial peptide buforin II: buforin II kills microorganisms by penetrating the cell membrane and inhibiting cellular functions. Biochem. Biophys. Res. Commun. 244, 253–257. 10.1006/bbrc.1998.8159 [DOI] [PubMed] [Google Scholar]
- Park I. Y., Cho J. H., Kim K. S., Kim Y. B., Kim M. S., Kim S. C. (2004). Helix stability confers salt resistance upon helical antimicrobial peptides. J. Biol. Chem. 279, 13896–13901. 10.1074/jbc.M311418200 [DOI] [PubMed] [Google Scholar]
- Paulmann M., Arnold T., Linke D., Ozdirekcan S., Kopp A., Gutsmann T., et al. (2012). Structure-activity analysis of the dermcidin-derived peptide DCD-1L, an anionic antimicrobial peptide present in human sweat. J. Biol. Chem. 287, 8434–8443. 10.1074/jbc.M111.332270 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perfect J. R. (2017). The antifungal pipeline: a reality check. Nat. Rev. Drug Discov. 16, 603–616. 10.1038/nrd.2017.46 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pestrak M. J., Chaney S. B., Eggleston H. C., Dellos-Nolan S., Dixit S., Mathew-Steiner S. S., et al. (2018). Pseudomonas aeruginosa rugose small-colony variants evade host clearance, are hyper-inflammatory, and persist in multiple host environments. PLoS Pathog. 14:e1006842. 10.1371/journal.ppat.1006842 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peters B. M., Jabra-Rizk M. A., O'May G. A., Costerton J. W., Shirtliff M. E. (2012). Polymicrobial interactions: impact on pathogenesis and human disease. Clin. Microbiol. Rev. 25, 193–213. 10.1128/CMR.00013-11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pfaller M. A., Castanheira M., Messer S. A., Rhomberg P. R., Jones R. N. (2014). Comparison of EUCAST and CLSI broth microdilution methods for the susceptibility testing of 10 systemically active antifungal agents when tested against Candida spp. Diagn. Microbiol. Infect. Dis. 79, 198–204. 10.1016/j.diagmicrobio.2014.03.004 [DOI] [PubMed] [Google Scholar]
- Pfaller M. A., Diekema D. J. (2012). Progress in antifungal susceptibility testing of Candida spp. by use of Clinical and Laboratory Standards Institute broth microdilution methods, 2010 to 2012. J. Clin. Microbiol. 50, 2846–2856. 10.1128/JCM.00937-12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pfaller M. A., Espinel-Ingroff A., Boyken L., Hollis R. J., Kroeger J., Messer S. A., et al. (2011). Comparison of the broth microdilution (BMD) method of the European Committee on antimicrobial susceptibility testing with the 24-hour CLSI BMD method for testing susceptibility of Candida species to fluconazole, posaconazole, and voriconazole by use of epidemiological cutoff values. J. Clin. Microbiol. 49, 845–850. 10.1128/JCM.02441-10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pfeil M. P., Pyne A. L. B., Losasso V., Ravi J., Lamarre B., Faruqui N., et al. (2018). Tuneable poration: host defense peptides as sequence probes for antimicrobial mechanisms. Sci. Rep. 8:14926 10.1038/s41598-018-33289-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- Piers K. L., Brown M. H., Hancock R. E. (1994). Improvement of outer membrane-permeabilizing and lipopolysaccharide-binding activities of an antimicrobial cationic peptide by C-terminal modification. Antimicrob. Agents Chemother. 38, 2311–2316. 10.1128/AAC.38.10.2311 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pieters J. (2008). Mycobacterium tuberculosis and the macrophage: maintaining a balance. Cell Host Microbe 3, 399–407. 10.1016/j.chom.2008.05.006 [DOI] [PubMed] [Google Scholar]
- Pinzon-Arango P. A., Nagarajan R., Camesano T. A. (2013). Interactions of antimicrobial peptide chrysophsin-3 with Bacillus anthracis in sporulated, germinated, and vegetative states. J. Phys. Chem. B 117, 6364–6372. 10.1021/jp400489u [DOI] [PubMed] [Google Scholar]
- Pizzolato-Cezar L. R., Okuda-Shinagawa N. M., Machini M. T. (2019). Combinatory therapy antimicrobial peptide-antibiotic to minimize the ongoing rise of resistance. Front. Microbiol. 10:1703. 10.3389/fmicb.2019.01703 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pletzer D., Hancock R. E. (2016). Antibiofilm peptides: potential as broad-spectrum agents. J. Bacteriol. 198, 2572–2578. 10.1128/JB.00017-16 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poirel L., Jayol A., Nordmann P. (2017). Polymyxins: antibacterial activity, susceptibility testing, and resistance mechanisms encoded by plasmids or chromosomes. Clin. Microbiol. Rev. 30, 557–596. 10.1128/CMR.00064-16 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pollini S., Brunetti J., Sennati S., Rossolini G. M., Bracci L., Pini A., et al. (2017). Synergistic activity profile of an antimicrobial peptide against multidrug-resistant and extensively drug-resistant strains of gram-negative bacterial pathogens. J. Pept. Sci. 23, 329–333. 10.1002/psc.2978 [DOI] [PubMed] [Google Scholar]
- Porto W. F., Irazazabal L., Alves E. S. F., Ribeiro S. M., Matos C. O., Pires A. S., et al. (2018). In silico optimization of a guava antimicrobial peptide enables combinatorial exploration for peptide design. Nat. Commun. 9:1490. 10.1038/s41467-018-03746-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Precit M. R., Wolter D. J., Griffith A., Emerson J., Burns J. L., Hoffman L. R. (2016). Optimized in vitro antibiotic susceptibility testing method for small-colony variant Staphylococcus aureus. Antimicrob. Agents Chemother. 60, 1725–1735. 10.1128/AAC.02330-15 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Proctor R. A., von Eiff C., Kahl B. C., Becker K., McNamara P., Herrmann M., et al. (2006). Small colony variants: a pathogenic form of bacteria that facilitates persistent and recurrent infections. Nat. Rev. Microbiol. 4, 295–305. 10.1038/nrmicro1384 [DOI] [PubMed] [Google Scholar]
- Ptaszynska N., Gucwa K., Olkiewicz K., Legowska A., Okonska J., Ruczynski J., et al. (2019). Antibiotic-based conjugates containing antimicrobial HLopt2 peptide: design, synthesis, antimicrobial and cytotoxic activities. ACS Chem. Biol. 14, 2233–2242. 10.1021/acschembio.9b00538 [DOI] [PubMed] [Google Scholar]
- Pulido D., Prats-Ejarque G., Villalba C., Albacar M., Gonzalez-Lopez J. J., Torrent M., et al. (2016). A novel RNase 3/ECP peptide for Pseudomonas aeruginosa biofilm eradication that combines antimicrobial, lipopolysaccharide binding, and cell-agglutinating activities. Antimicrob. Agents Chemother. 60, 6313–6325. 10.1128/AAC.00830-16 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pyne A., Pfeil M. P., Bennett I., Ravi J., Iavicoli P., Lamarre B., et al. (2017). Engineering monolayer poration for rapid exfoliation of microbial membranes. Chem. Sci. 8, 1105–1115. 10.1039/C6SC02925F [DOI] [PMC free article] [PubMed] [Google Scholar]
- Radlinski L., Conlon B. P. (2018). Antibiotic efficacy in the complex infection environment. Curr. Opin. Microbiol. 42, 19–24. 10.1016/j.mib.2017.09.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rajchakit U., Sarojini V. (2017). Recent developments in antimicrobial-peptide-conjugated gold nanoparticles. Bioconjug. Chem. 28, 2673–2686. 10.1021/acs.bioconjchem.7b00368 [DOI] [PubMed] [Google Scholar]
- Rapala-Kozik M., Bochenska O., Zajac D., Karkowska-Kuleta J., Gogol M., Zawrotniak M., et al. (2018). Extracellular proteinases of Candida species pathogenic yeasts. Mol. Oral. Microbiol. 33, 113–124. 10.1111/omi.12206 [DOI] [PubMed] [Google Scholar]
- Ratanji K. D., Derrick J. P., Dearman R. J., Kimber I. (2014). Immunogenicity of therapeutic proteins: influence of aggregation. J. Immunotoxicol. 11, 99–109. 10.3109/1547691X.2013.821564 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rawlings N. D., Barrett A. J., Thomas P. D., Huang X., Bateman A., Finn R. D. (2018). The MEROPS database of proteolytic enzymes, their substrates and inhibitors in 2017 and a comparison with peptidases in the PANTHER database. Nucleic Acids Res. 46, D624–D632. 10.1093/nar/gkx1134 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reichhardt C., Stevens D. A., Cegelski L. (2016). Fungal biofilm composition and opportunities in drug discovery. Future Med. Chem. 8, 1455–1468. 10.4155/fmc-2016-0049 [DOI] [PubMed] [Google Scholar]
- Reinhardt A., Neundorf I. (2016). Design and application of antimicrobial peptide conjugates. Int. J. Mol. Sci. 17:701. 10.3390/ijms17050701 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rigatto M. H., Falci D. R., Zavascki A. P. (2019). Clinical use of polymyxin B. Adv. Exp. Med. Biol. 1145, 197–218. 10.1007/978-3-030-16373-0_14 [DOI] [PubMed] [Google Scholar]
- Rippke F., Berardesca E., Weber T. M. (2018). pH and microbial infections. Curr. Probl. Dermatol. 54, 87–94. 10.1159/000489522 [DOI] [PubMed] [Google Scholar]
- Rivas-Santiago B., Serrano C. J., Enciso-Moreno J. A. (2009). Susceptibility to infectious diseases based on antimicrobial peptide production. Infect. Immun. 77, 4690–4695. 10.1128/IAI.01515-08 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Romling U., Balsalobre C. (2012). Biofilm infections, their resilience to therapy and innovative treatment strategies. J. Intern. Med. 272, 541–561. 10.1111/joim.12004 [DOI] [PubMed] [Google Scholar]
- Rondon-Villarreal P., Pinzon-Reyes E. (2018). Computer aided design of non-toxic antibacterial peptides. Curr. Top. Med. Chem. 18, 1044–1052. 10.2174/1568026618666180719163251 [DOI] [PubMed] [Google Scholar]
- Roope L. S. J., Smith R. D., Pouwels K. B., Buchanan J., Abel L., Eibich P., et al. (2019). The challenge of antimicrobial resistance: what economics can contribute. Science 364:eaau4679. 10.1126/science.aau4679 [DOI] [PubMed] [Google Scholar]
- Ross J. E., Mendes R. E., Jones R. N. (2014). Quality control MIC ranges used for telavancin with application of a revised CLSI reference broth microdilution method. J. Clin. Microbiol. 52, 3399–3401. 10.1128/JCM.01210-14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roth B. L., Poot M., Yue S. T., Millard P. J. (1997). Bacterial viability and antibiotic susceptibility testing with SYTOX green nucleic acid stain. Appl. Environ. Microbiol. 63, 2421–2431. 10.1128/AEM.63.6.2421-2431.1997 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rothstein D. M., Spacciapoli P., Tran L. T., Xu T., Roberts F. D., Dalla Serra M., et al. (2001). Anticandida activity is retained in P-113, a 12-amino-acid fragment of histatin 5. Antimicrob. Agents Chemother. 45, 1367–1373. 10.1128/AAC.45.5.1367-1373.2001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roversi D., Luca V., Aureli S., Park Y., Mangoni M. L., Stella L. (2014). How many antimicrobial peptide molecules kill a bacterium? the case of PMAP-23. ACS Chem. Biol. 9, 2003–2007. 10.1021/cb500426r [DOI] [PubMed] [Google Scholar]
- Sabaeifard P., Abdi-Ali A., Soudi M. R., Dinarvand R. (2014). Optimization of tetrazolium salt assay for Pseudomonas aeruginosa biofilm using microtiter plate method. J. Microbiol. Methods 105, 134–140. 10.1016/j.mimet.2014.07.024 [DOI] [PubMed] [Google Scholar]
- Sader H. S., Rhomberg P. R., Flamm R. K., Jones R. N. (2012). Use of a surfactant (polysorbate 80) to improve MIC susceptibility testing results for polymyxin B and colistin. Diagn. Microbiol. Infect. Dis. 74, 412–414. 10.1016/j.diagmicrobio.2012.08.025 [DOI] [PubMed] [Google Scholar]
- Sadowska B., Bonar A., von Eiff C., Proctor R. A., Chmiela M., Rudnicka W., et al. (2002). Characteristics of Staphylococcus aureus, isolated from airways of cystic fibrosis patients, and their small colony variants. FEMS Immunol. Med. Microbiol. 32, 191–197. 10.1111/j.1574-695X.2002.tb00553.x [DOI] [PubMed] [Google Scholar]
- Sajjan U. S., Tran L. T., Sole N., Rovaldi C., Akiyama A., Friden P. M., et al. (2001). P-113D, an antimicrobial peptide active against Pseudomonas aeruginosa, retains activity in the presence of sputum from cystic fibrosis patients. Antimicrob. Agents Chemother. 45, 3437–3444. 10.1128/AAC.45.12.3437-3444.2001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sakoulas G., Okumura C. Y., Thienphrapa W., Olson J., Nonejuie P., Dam Q., et al. (2014). Nafcillin enhances innate immune-mediated killing of methicillin-resistant Staphylococcus aureus. J. Mol. Med. 92, 139–149. 10.1007/s00109-013-1100-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Samuelsen O., Haukland H. H., Kahl B. C., von Eiff C., Proctor R. A., Ulvatne H., et al. (2005). Staphylococcus aureus small colony variants are resistant to the antimicrobial peptide lactoferricin B. J. Antimicrob. Chemother. 56, 1126–1129. 10.1093/jac/dki385 [DOI] [PubMed] [Google Scholar]
- Sanchez-Gomez S., Ferrer-Espada R., Stewart P. S., Pitts B., Lohner K., Martinez de Tejada G. (2015). Antimicrobial activity of synthetic cationic peptides and lipopeptides derived from human lactoferricin against Pseudomonas aeruginosa planktonic cultures and biofilms. BMC Microbiol. 15:137. 10.1186/s12866-015-0473-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanchez-Gomez S., Lamata M., Leiva J., Blondelle S. E., Jerala R., Andra J., et al. (2008). Comparative analysis of selected methods for the assessment of antimicrobial and membrane-permeabilizing activity: a case study for lactoferricin derived peptides. BMC Microbiol. 8:196. 10.1186/1471-2180-8-196 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanguinetti M., Posteraro B. (2017). New approaches for antifungal susceptibility testing. Clin. Microbiol. Infect. 23, 931–934. 10.1016/j.cmi.2017.03.025 [DOI] [PubMed] [Google Scholar]
- Sanguinetti M., Posteraro B. (2018). Susceptibility testing of fungi to antifungal drugs. J. Fungi 4:110. 10.3390/jof4030110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Santos V., Hirshfield I. (2016). The physiological and molecular characterization of a small colony variant of Escherichia coli and its phenotypic rescue. PLoS ONE 11:e0157578. 10.1371/journal.pone.0157578 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saravolatz L. D., Stein G. E. (2015). Oritavancin: a long-half-life lipoglycopeptide. Clin. Infect. Dis. 61, 627–632. 10.1093/cid/civ311 [DOI] [PubMed] [Google Scholar]
- Sasaki S., Yanagibori R., Amano K. (1998). Validity of a self-administered diet history questionnaire of sodium and potassium: comparison with single 24-hour urinary excretion. JPN. Circ. J. 62, 431–435. 10.1253/jcj.62.431 [DOI] [PubMed] [Google Scholar]
- Savini F., Luca V., Bocedi A., Massoud R., Park Y., Mangoni M. L., et al. (2017). Cell-density dependence of host-defense peptide activity and selectivity in the presence of host cells. ACS Chem. Biol. 12, 52–56. 10.1021/acschembio.6b00910 [DOI] [PubMed] [Google Scholar]
- Schmidtchen A., Frick I. M., Andersson E., Tapper H., Bjorck L. (2002). Proteinases of common pathogenic bacteria degrade and inactivate the antibacterial peptide LL-37. Mol. Microbiol. 46, 157–168. 10.1046/j.1365-2958.2002.03146.x [DOI] [PubMed] [Google Scholar]
- Schroeder B. O., Ehmann D., Precht J. C., Castillo P. A., Kuchler R., Berger J., et al. (2015). Paneth cell alpha-defensin 6 (HD-6) is an antimicrobial peptide. Mucosal. Immunol. 8, 661–671. 10.1038/mi.2014.100 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwab U., Gilligan P., Jaynes J., Henke D. (1999). In vitro activities of designed antimicrobial peptides against multidrug-resistant cystic fibrosis pathogens. Antimicrob. Agents Chemother. 43, 1435–1440. 10.1128/AAC.43.6.1435 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scott L. J. (2012). Micafungin: a review of its use in the prophylaxis and treatment of invasive Candida infections. Drugs 72, 2141–2165. 10.2165/11209970-000000000-00000 [DOI] [PubMed] [Google Scholar]
- Seedher N., Agarwal P. (2010). Effect of metal ions on some pharmacologically relevant interactions involving fluoroquinolone antibiotics. Drug Metabol. Drug Interact. 25, 17–24. 10.1515/DMDI.2010.003 [DOI] [PubMed] [Google Scholar]
- Segev-Zarko L., Saar-Dover R., Brumfeld V., Mangoni M. L., Shai Y. (2015). Mechanisms of biofilm inhibition and degradation by antimicrobial peptides. Biochem. J. 468, 259–270. 10.1042/BJ20141251 [DOI] [PubMed] [Google Scholar]
- Sejdini M., Begzati A., Salihu S., Krasniqi S., Berisha N., Aliu N. (2018). The role and impact of salivary Zn levels on dental caries. Int. J. Dent. 2018:8137915. 10.1155/2018/8137915 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Setlow P. (2006). Spores of Bacillus subtilis: their resistance to and killing by radiation, heat and chemicals. J. Appl. Microbiol. 101, 514–525. 10.1111/j.1365-2672.2005.02736.x [DOI] [PubMed] [Google Scholar]
- Setlow P. (2014). Spore resistance properties. Microbiol. Spectr. 2 10.1128/microbiolspec.TBS-0003-2012 [DOI] [PubMed] [Google Scholar]
- Shahrour H., Ferrer-Espada R., Dandache I., Barcena-Varela S., Sanchez-Gomez S., Chokr A., et al. (2019). AMPs as anti-biofilm agents for human therapy and prophylaxis. Adv. Exp. Med. Biol. 1117, 257–279. 10.1007/978-981-13-3588-4_14 [DOI] [PubMed] [Google Scholar]
- Shin S. C., Ahn I. H., Ahn D. H., Lee Y. M., Lee J., Lee J. H., et al. (2017). Characterization of two antimicrobial peptides from antarctic fishes (Notothenia coriiceps and Parachaenichthys charcoti). PLoS ONE 12:e0170821. 10.1371/journal.pone.0170821 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shin Y. P., Park H. J., Shin S. H., Lee Y. S., Park S., Jo S., et al. (2010). Antimicrobial activity of a halocidin-derived peptide resistant to attacks by proteases. Antimicrob. Agents Chemother. 54, 2855–2866. 10.1128/AAC.01790-09 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sievert D. M., Ricks P., Edwards J. R., Schneider A., Patel J., Srinivasan A., et al. (2013). Antimicrobial-resistant pathogens associated with healthcare-associated infections: summary of data reported to the National Healthcare Safety Network at the Centers for Disease Control and Prevention, 2009-2010. Infect. Control Hosp. Epidemiol. 34, 1–14. 10.1086/668770 [DOI] [PubMed] [Google Scholar]
- Silva F. D., Rezende C. A., Rossi D. C., Esteves E., Dyszy F. H., Schreier S., et al. (2009). Structure and mode of action of microplusin, a copper II-chelating antimicrobial peptide from the cattle tick Rhipicephalus (Boophilus) microplus. J. Biol. Chem. 284, 34735–34746. 10.1074/jbc.M109.016410 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Silver L. L. (2011). Challenges of antibacterial discovery. Clin. Microbiol. Rev. 24, 71–109. 10.1128/CMR.00030-10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simjee S., McDermott P., Trott D. J., Chuanchuen R. (2018). Present and future surveillance of antimicrobial resistance in animals: principles and practices. Microbiol. Spectr. 6. 10.1128/microbiolspec.ARBA-0028-2017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singh K., Shekhar S., Yadav Y., Xess I., Dey S. (2017). DS6: anticandidal, antibiofilm peptide against Candida tropicalis and exhibit synergy with commercial drug. J. Pept. Sci. 23, 228–235. 10.1002/psc.2973 [DOI] [PubMed] [Google Scholar]
- Singh R., Ray P., Das A., Sharma M. (2010). Enhanced production of exopolysaccharide matrix and biofilm by a menadione-auxotrophic Staphylococcus aureus small-colony variant. J. Med. Microbiol. 59 (Pt 5), 521–527. 10.1099/jmm.0.017046-0 [DOI] [PubMed] [Google Scholar]
- Singhal L., Sharma M., Verma S., Kaur R., Britto X. B., Kumar S. M., et al. (2018). Comparative evaluation of broth microdilution with polystyrene and glass-coated plates, agar dilution, e-test, vitek, and disk diffusion for susceptibility testing of colistin and polymyxin b on carbapenem-resistant clinical isolates of acinetobacter baumannii. Microb. Drug Resist. 24, 1082–1088. 10.1089/mdr.2017.0251 [DOI] [PubMed] [Google Scholar]
- Sivertsen A., Isaksson J., Leiros H. K., Svenson J., Svendsen J. S., Brandsdal B. O. (2014). Synthetic cationic antimicrobial peptides bind with their hydrophobic parts to drug site II of human serum albumin. BMC Struct. Biol. 14:4. 10.1186/1472-6807-14-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith K. P., Kirby J. E. (2018). The inoculum effect in the era of multidrug resistance: Minor differences in inoculum have dramatic effect on MIC determination. Antimicrob. Agents Chemother. 62:e00433–18. 10.1128/AAC.00433-18 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Snoussi M., Talledo J. P., Del Rosario N. A., Mohammadi S., Ha B. Y., Kosmrlj A., et al. (2018). Heterogenous absorption of antimicrobial peptide LL37 in Escherichia coli cells enhances population survivability. Elife 7:38174. 10.7554/eLife.38174.023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Song C., Weichbrodt C., E S. S., Dynowski M., B O. F., Bechinger B., et al. (2013). Crystal structure and functional mechanism of a human antimicrobial membrane channel. Proc. Natl. Acad. Sci. U.S.A. 110, 4586–4591. 10.1073/pnas.1214739110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Song J. C., Stevens D. A. (2016). Caspofungin: pharmacodynamics, pharmacokinetics, clinical uses and treatment outcomes. Crit. Rev. Microbiol. 42, 813–846. 10.3109/1040841X.2015.1068271 [DOI] [PubMed] [Google Scholar]
- Soren O., Brinch K. S., Patel D., Liu Y., Liu A., Coates A., et al. (2015). Antimicrobial peptide novicidin synergizes with rifampin, ceftriaxone, and ceftazidime against antibiotic-resistant enterobacteriaceae in vitro. Antimicrob. Agents Chemother. 59, 6233–6240. 10.1128/AAC.01245-15 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Srivastava S., Ghosh J. K. (2013). Introduction of a lysine residue promotes aggregation of temporin L in lipopolysaccharides and augmentation of its antiendotoxin property. Antimicrob. Agents Chemother. 57, 2457–2466. 10.1128/AAC.00169-13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Starr C. G., He J., Wimley W. C. (2016). Host cell interactions are a significant barrier to the clinical utility of peptide antibiotics. ACS Chem. Biol. 11, 3391–3399. 10.1021/acschembio.6b00843 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Starr C. G., Wimley W. C. (2017). Antimicrobial peptides are degraded by the cytosolic proteases of human erythrocytes. Biochim. Biophys. Acta Biomembr. 1859, 2319–2326. 10.1016/j.bbamem.2017.09.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stewart P. S., Costerton J. W. (2001). Antibiotic resistance of bacteria in biofilms. Lancet 358, 135–138. 10.1016/S0140-6736(01)05321-1 [DOI] [PubMed] [Google Scholar]
- Storm D. R. (1974). Mechanism of bacitracin action: a specific lipid-peptide interaction. Ann. N. Y. Acad. Sci. 235, 387–398. 10.1111/j.1749-6632.1974.tb43278.x [DOI] [PubMed] [Google Scholar]
- Storm D. R., Strominger J. L. (1973). Complex formation between bacitracin peptides and isoprenyl pyrophosphates. The specificity of lipid-peptide interactions. J. Biol. Chem. 248, 3940–3945. [PubMed] [Google Scholar]
- Stumpe S., Schmid R., Stephens D. L., Georgiou G., Bakker E. P. (1998). Identification of OmpT as the protease that hydrolyzes the antimicrobial peptide protamine before it enters growing cells of Escherichia coli. J. Bacteriol. 180, 4002–4006. 10.1128/JB.180.15.4002-4006.1998 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Su C., Yu J., Lu Y. (2018). Hyphal development in Candida albicans from different cell states. Curr. Genet. 64, 1239–1243. 10.1007/s00294-018-0845-5 [DOI] [PubMed] [Google Scholar]
- Subramanian Vignesh K., Landero Figueroa J. A., Porollo A., Caruso J. A., Deepe G. S., Jr. (2013). Granulocyte macrophage-colony stimulating factor induced Zn sequestration enhances macrophage superoxide and limits intracellular pathogen survival. Immunity 39, 697–710. 10.1016/j.immuni.2013.09.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sulaiman J. E., Hao C., Lam H. (2018). Specific enrichment and proteomics analysis of Escherichia coli persisters from rifampin pretreatment. J. Proteome Res. 17, 3984–3996. 10.1021/acs.jproteome.8b00625 [DOI] [PubMed] [Google Scholar]
- Sun H., Hong Y., Xi Y., Zou Y., Gao J., Du J. (2018). Synthesis, self-assembly, and biomedical applications of antimicrobial peptide-polymer conjugates. Biomacromolecules 19, 1701–1720. 10.1021/acs.biomac.8b00208 [DOI] [PubMed] [Google Scholar]
- Sun Y., Shang D. (2015). Inhibitory effects of antimicrobial peptides on lipopolysaccharide-induced inflammation. Mediators Inflamm. 2015:167572. 10.1155/2015/167572 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sutherland C. A., Nicolau D. P. (2014). To add or not to add polysorbate-80: impact on colistin MIC values for clinical strains of enterobacteriaceae, P. aeruginosa and quality controls. J. Clin. Microbiol. 52:3810. 10.1128/JCM.01454-14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Syal K., Mo M., Yu H., Iriya R., Jing W., Guodong S., et al. (2017). Current and emerging techniques for antibiotic susceptibility tests. Theranostics 7, 1795–1805. 10.7150/thno.19217 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tam J. P., Lu Y. A., Yang J. L. (2000). Marked increase in membranolytic selectivity of novel cyclic tachyplesins constrained with an antiparallel two-beta strand cystine knot framework. Biochem. Biophys. Res. Commun. 267, 783–790. 10.1006/bbrc.1999.2035 [DOI] [PubMed] [Google Scholar]
- Thery T., O'Callaghan Y., O'Brien N., Arendt E. K. (2018). Optimisation of the antifungal potency of the amidated peptide H-Orn-Orn-Trp-Trp-NH2 against food contaminants. Int. J. Food Microbiol. 265, 40–48. 10.1016/j.ijfoodmicro.2017.10.024 [DOI] [PubMed] [Google Scholar]
- Thery T., Shwaiki L. N., O'Callaghan Y. C., O'Brien N. M., Arendt E. K. (2019). Antifungal activity of a de novo synthetic peptide and derivatives against fungal food contaminants. J. Pept. Sci. 25:e3137. 10.1002/psc.3137 [DOI] [PubMed] [Google Scholar]
- Tikhomirova A., Trappetti C., Standish A. J., Zhou Y., Breen J., Pederson S., et al. (2018). Specific growth conditions induce a Streptococcus pneumoniae non-mucoidal, small colony variant and determine the outcome of its co-culture with Haemophilus influenzae. Pathog. Dis. 76:fty074. 10.1093/femspd/fty074 [DOI] [PubMed] [Google Scholar]
- Todd O. A., Peters B. M. (2019). Candida albicans and Staphylococcus aureus pathogenicity and polymicrobial interactions: lessons beyond koch's postulates. J. Fungi 5:81. 10.3390/jof5030081 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Torrent M., Andreu D., Nogues V. M., Boix E. (2011). Connecting Peptide physicochemical and antimicrobial properties by a rational prediction model. PLoS ONE 6:e16968. 10.1371/journal.pone.0016968 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Torres M. D. T., Pedron C. N., Higashikuni Y., Kramer R. M., Cardoso M. H., Oshiro K. G. N., et al. (2018). Structure-function-guided exploration of the antimicrobial peptide polybia-CP identifies activity determinants and generates synthetic therapeutic candidates. Commun. Biol. 1:221. 10.1038/s42003-018-0224-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Torres M. D. T., Sothiselvam S., Lu T. K., de la Fuente-Nunez C. (2019). Peptide design principles for antimicrobial applications. J. Mol. Biol. 431, 3547–3567. 10.1016/j.jmb.2018.12.015 [DOI] [PubMed] [Google Scholar]
- Torres M. T., de la Fuente-Nunez C. (2019). Toward computer-made artificial antibiotics. Curr. Opin. Microbiol. 51, 30–38. 10.1016/j.mib.2019.03.004 [DOI] [PubMed] [Google Scholar]
- Travis S. M., Anderson N. N., Forsyth W. R., Espiritu C., Conway B. D., Greenberg E. P., et al. (2000). Bactericidal activity of mammalian cathelicidin-derived peptides. Infect. Immun. 68, 2748–2755. 10.1128/IAI.68.5.2748-2755.2000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Troskie A. M., Rautenbach M., Delattin N., Vosloo J. A., Dathe M., Cammue B. P., et al. (2014). Synergistic activity of the tyrocidines, antimicrobial cyclodecapeptides from Bacillus aneurinolyticus, with amphotericin B and caspofungin against Candida albicans biofilms. Antimicrob. Agents Chemother. 58, 3697–3707. 10.1128/AAC.02381-14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Troy D. C., James P. M., Samuel M. C. (2007). Trace mineral losses in sweat. Curr. Nutr. Food Sci 3, 236–241. 10.2174/157340107781369215 [DOI] [Google Scholar]
- Tu Y. H., Ho Y. H., Chuang Y. C., Chen P. C., Chen C. S. (2011). Identification of lactoferricin B intracellular targets using an Escherichia coli proteome chip. PLoS ONE 6:e28197. 10.1371/journal.pone.0028197 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tucker A. T., Leonard S. P., DuBois C. D., Knauf G. A., Cunningham A. L., Wilke C. O., et al. (2018). Discovery of next-generation antimicrobials through bacterial self-screening of surface-displayed peptide libraries. Cell 172, 618–628.e613. 10.1016/j.cell.2017.12.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Turner J., Cho Y., Dinh N. N., Waring A. J., Lehrer R. I. (1998). Activities of LL-37, a cathelin-associated antimicrobial peptide of human neutrophils. Antimicrob. Agents Chemother. 42, 2206–2214. 10.1128/AAC.42.9.2206 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Turner P., Ashley E. A. (2019). Standardising the reporting of microbiology and antimicrobial susceptibility data. Lancet Infect. Dis. 19, 1163–1164. 10.1016/S1473-3099(19)30561-4 [DOI] [PubMed] [Google Scholar]
- Turnidge J., Kahlmeter G., Kronvall G. (2006). Statistical characterisation of bacterial wild-type MIC value distributions and the determination of epidemiological cut-off values. Clin. Microbiol. Infect. 12, 418–425. 10.1111/j.1469-0691.2006.01377.x [DOI] [PubMed] [Google Scholar]
- Udekwu K. I., Parrish N., Ankomah P., Baquero F., Levin B. R. (2009). Functional relationship between bacterial cell density and the efficacy of antibiotics. J. Antimicrob. Chemother. 63, 745–757. 10.1093/jac/dkn554 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uivarosi V. (2013). Metal complexes of quinolone antibiotics and their applications: an update. Molecules 18, 11153–11197. 10.3390/molecules180911153 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ulaeto D. O., Morris C. J., Fox M. A., Gumbleton M., Beck K. (2016). Destabilization of alpha-helical structure in solution improves bactericidal activity of antimicrobial peptides: opposite effects on bacterial and viral targets. Antimicrob. Agents Chemother. 60, 1984–1991. 10.1128/AAC.02146-15 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Umerska A., Strandh M., Cassisa V., Matougui N., Eveillard M., Saulnier P. (2018). Synergistic effect of combinations containing EDTA and the antimicrobial peptide AA230, an Arenicin-3 derivative, on gram-negative bacteria. Biomolecules 8:122. 10.3390/biom8040122 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Underhill D. M., Ozinsky A. (2002). Phagocytosis of microbes: complexity in action. Annu. Rev. Immunol. 20, 825–852. 10.1146/annurev.immunol.20.103001.114744 [DOI] [PubMed] [Google Scholar]
- van Belkum A., Bachmann T. T., Ludke G., Lisby J. G., Kahlmeter G., Mohess A., et al. (2019). Developmental roadmap for antimicrobial susceptibility testing systems. Nat. Rev. Microbiol. 17, 51–62. 10.1038/s41579-018-0098-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- van der Does A. M., Hiemstra P. S., Mookherjee N. (2019). Antimicrobial host defence peptides: immunomodulatory functions and translational prospects. Adv. Exp. Med. Biol. 1117, 149–171. 10.1007/978-981-13-3588-4_10 [DOI] [PubMed] [Google Scholar]
- van Dijck P., Sjollema J., Cammue B. P., Lagrou K., Berman J., d'Enfert C., et al. (2018). Methodologies for in vitro and in vivo evaluation of efficacy of antifungal and antibiofilm agents and surface coatings against fungal biofilms. Microb. Cell 5, 300–326. 10.15698/mic2018.07.638 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Puyvelde S., Deborggraeve S., Jacobs J. (2018). Why the antibiotic resistance crisis requires a one health approach. Lancet Infect. Dis. 18, 132–134. 10.1016/S1473-3099(17)30704-1 [DOI] [PubMed] [Google Scholar]
- Vesga O., Groeschel M. C., Otten M. F., Brar D. W., Vann J. M., Proctor R. A. (1996). Staphylococcus aureus small colony variants are induced by the endothelial cell intracellular milieu. J. Infect. Dis. 173, 739–742. 10.1093/infdis/173.3.739 [DOI] [PubMed] [Google Scholar]
- Vidovic S., An R., Rendahl A. (2019). Molecular and physiological characterization of fluoroquinolone-highly resistant salmonella enteritidis strains. Front. Microbiol. 10:729. 10.3389/fmicb.2019.00729 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vlieghe P., Lisowski V., Martinez J., Khrestchatisky M. (2010). Synthetic therapeutic peptides: science and market. Drug Discov. Today 15, 40–56. 10.1016/j.drudis.2009.10.009 [DOI] [PubMed] [Google Scholar]
- Von Borowski R. G., Macedo A. J., Gnoatto S. C. B. (2018). Peptides as a strategy against biofilm-forming microorganisms: Structure-activity relationship perspectives. Eur. J. Pharm. Sci. 114, 114–137. 10.1016/j.ejps.2017.11.008 [DOI] [PubMed] [Google Scholar]
- von Eiff C., Heilmann C., Proctor R. A., Woltz C., Peters G., Gotz F. (1997). A site-directed Staphylococcus aureus hemB mutant is a small-colony variant which persists intracellularly. J. Bacteriol. 179, 4706–4712. 10.1128/JB.179.15.4706-4712.1997 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wagner D., Maser J., Lai B., Cai Z., Barry C. E., 3rd, Honer Zu, Bentrup K., et al. (2005). Elemental analysis of Mycobacterium avium-, Mycobacterium tuberculosis-, and Mycobacterium smegmatis-containing phagosomes indicates pathogen-induced microenvironments within the host cell's endosomal system. J. Immunol. 174, 1491–1500. 10.4049/jimmunol.174.3.1491 [DOI] [PubMed] [Google Scholar]
- Walkenhorst W. F. (2016). Using adjuvants and environmental factors to modulate the activity of antimicrobial peptides. Biochim. Biophys. Acta 1858, 926–935. 10.1016/j.bbamem.2015.12.034 [DOI] [PubMed] [Google Scholar]
- Walkenhorst W. F., Klein J. W., Vo P., Wimley W. C. (2013). pH Dependence of microbe sterilization by cationic antimicrobial peptides. Antimicrob. Agents Chemother. 57, 3312–3320. 10.1128/AAC.00063-13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walkenhorst W. F., Sundrud J. N., Laviolette J. M. (2014). Additivity and synergy between an antimicrobial peptide and inhibitory ions. Biochim. Biophys. Acta 1838, 2234–2242. 10.1016/j.bbamem.2014.05.005 [DOI] [PubMed] [Google Scholar]
- Wang C. W., Yip B. S., Cheng H. T., Wang A. H., Chen H. L., Cheng J. W., et al. (2009). Increased potency of a novel D-beta-naphthylalanine-substituted antimicrobial peptide against fluconazole-resistant fungal pathogens. FEMS Yeast Res. 9, 967–970. 10.1111/j.1567-1364.2009.00531.x [DOI] [PubMed] [Google Scholar]
- Wang G. (2015). Improved methods for classification, prediction and design of antimicrobial peptides. Methods Mol. Biol 1268, 43–66. 10.1007/978-1-4939-2285-7_3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang T., El Meouche I., Dunlop M. J. (2017). Bacterial persistence induced by salicylate via reactive oxygen species. Sci. Rep. 7:43839. 10.1038/srep43839 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Y., Agerberth B., Lothgren A., Almstedt A., Johansson J. (1998). Apolipoprotein A-I binds and inhibits the human antibacterial/cytotoxic peptide LL-37. J. Biol. Chem. 273, 33115–33118. 10.1074/jbc.273.50.33115 [DOI] [PubMed] [Google Scholar]
- Waters E. M., Rowe S. E., O'Gara J. P., Conlon B. P. (2016). Convergence of Staphylococcus aureus persister and biofilm research: can biofilms be defined as communities of adherent persister cells? PLoS Pathog. 12:e1006012. 10.1371/journal.ppat.1006012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weed L. L., Longfellow D. (1954). Morphological and biochemical changes induced by copper in a population of Escherichia coli. J. Bacteriol. 67, 27–33. 10.1128/JB.67.1.27-33.1954 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wehkamp J., Salzman N. H., Porter E., Nuding S., Weichenthal M., Petras R. E., et al. (2005). Reduced Paneth cell alpha-defensins in ileal Crohn's disease. Proc. Natl. Acad. Sci. U.S.A. 102, 18129–18134. 10.1073/pnas.0505256102 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wei G. X., Bobek L. A. (2005). Human salivary mucin MUC7 12-mer-L and 12-mer-D peptides: antifungal activity in saliva, enhancement of activity with protease inhibitor cocktail or EDTA, and cytotoxicity to human cells. Antimicrob. Agents Chemother. 49, 2336–2342. 10.1128/AAC.49.6.2336-2342.2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wei G. X., Campagna A. N., Bobek L. A. (2007). Factors affecting antimicrobial activity of MUC7 12-mer, a human salivary mucin-derived peptide. Ann. Clin. Microbiol. Antimicrob. 6:14. 10.1186/1476-0711-6-14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weiss C. H., Persell S. D., Wunderink R. G., Baker D. W. (2012). Empiric antibiotic, mechanical ventilation, and central venous catheter duration as potential factors mediating the effect of a checklist prompting intervention on mortality: an exploratory analysis. BMC Health Serv. Res. 12:198. 10.1186/1472-6963-12-198 [DOI] [PMC free article] [PubMed] [Google Scholar]
- White C., Lee J., Kambe T., Fritsche K., Petris M. J. (2009). A role for the ATP7A copper-transporting ATPase in macrophage bactericidal activity. J. Biol. Chem. 284, 33949–33956. 10.1074/jbc.M109.070201 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wi Y. M., Patel R. (2018). Understanding biofilms and novel approaches to the diagnosis, prevention, and treatment of medical device-associated infections. Infect. Dis. Clin. North Am. 32, 915–929. 10.1016/j.idc.2018.06.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wiegand I., Hilpert K., Hancock R. E. (2008). Agar and broth dilution methods to determine the minimal inhibitory concentration (MIC) of antimicrobial substances. Nat. Protoc. 3, 163–175. 10.1038/nprot.2007.521 [DOI] [PubMed] [Google Scholar]
- Wishart D. S., Feunang Y. D., Marcu A., Guo A. C., Liang K., Vazquez-Fresno R., et al. (2018). HMDB 4.0: the human metabolome database for 2018. Nucleic Acids Res. 46, D608–D617. 10.1093/nar/gkx1089 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Withman B., Gunasekera T. S., Beesetty P., Agans R., Paliy O. (2013). Transcriptional responses of uropathogenic Escherichia coli to increased environmental osmolality caused by salt or urea. Infect. Immun. 81, 80–89. 10.1128/IAI.01049-12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wolcott R., Costerton J. W., Raoult D., Cutler S. J. (2013). The polymicrobial nature of biofilm infection. Clin. Microbiol. Infect. 19, 107–112. 10.1111/j.1469-0691.2012.04001.x [DOI] [PubMed] [Google Scholar]
- Wolfmeier H., Pletzer D., Mansour S. C., Hancock R. E. W. (2018). New perspectives in biofilm eradication. ACS Infect. Dis. 4, 93–106. 10.1021/acsinfecdis.7b00170 [DOI] [PubMed] [Google Scholar]
- World Health Organization (1961). Standardization of Methods for Conducting Microbic Sensitivity Tests : Second Report of the Expert Committee on Antibiotics “[meeting held in Geneva from 11 to 16 July 1960]”. World Health Organization. Expert Committee on Antibiotics & World Health Organization. [Google Scholar]
- Wuyts J., Van Dijck P., Holtappels M. (2018). Fungal persister cells: the basis for recalcitrant infections? PLoS Pathog. 14:e1007301. 10.1371/journal.ppat.1007301 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xie C., Tang X., Xu W., Diao R., Cai Z., Chan H. C. (2010). A host defense mechanism involving CFTR-mediated bicarbonate secretion in bacterial prostatitis. PLoS ONE 5:e15255. 10.1371/journal.pone.0015255 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yagci S., Hascelik G., Dogru D., Ozcelik U., Sener B. (2013). Prevalence and genetic diversity of Staphylococcus aureus small-colony variants in cystic fibrosis patients. Clin. Microbiol. Infect. 19, 77–84. 10.1111/j.1469-0691.2011.03742.x [DOI] [PubMed] [Google Scholar]
- Yamasaki K., Gallo R. L. (2008). Antimicrobial peptides in human skin disease. Eur. J. Dermatol. 18, 11–21. 10.1684/ejd.2008.0304 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yan J., Bassler B. L. (2019). Surviving as a community: antibiotic tolerance and persistence in bacterial biofilms. Cell Host Microbe 26, 15–21. 10.1016/j.chom.2019.06.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yasir M., Willcox M. D. P., Dutta D. (2018). Action of antimicrobial peptides against bacterial biofilms. Materials 11:2468. 10.3390/ma11122468 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yates R. M., Hermetter A., Russell D. G. (2005). The kinetics of phagosome maturation as a function of phagosome/lysosome fusion and acquisition of hydrolytic activity. Traffic 6, 413–420. 10.1111/j.1600-0854.2005.00284.x [DOI] [PubMed] [Google Scholar]
- Yount N. Y., Weaver D. C., Lee E. Y., Lee M. W., Wang H., Chan L. C., et al. (2019). Unifying structural signature of eukaryotic alpha-helical host defense peptides. Proc. Natl. Acad. Sci. U.S.A. 116, 6944–6953. 10.1073/pnas.1819250116 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu H. Y., Tu C. H., Yip B. S., Chen H. L., Cheng H. T., Huang K. C., et al. (2011). Easy strategy to increase salt resistance of antimicrobial peptides. Antimicrob. Agents Chemother. 55, 4918–4921. 10.1128/AAC.00202-11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu Z. W., Quinn P. J. (1998). The modulation of membrane structure and stability by dimethyl sulphoxide (review). Mol. Membr. Biol. 15, 59–68. 10.3109/09687689809027519 [DOI] [PubMed] [Google Scholar]
- Zasloff M. (2002). Antimicrobial peptides of multicellular organisms. Nature 415, 389–395. 10.1038/415389a [DOI] [PubMed] [Google Scholar]
- Zerweck J., Strandberg E., Kukharenko O., Reichert J., Burck J., Wadhwani P., et al. (2017). Molecular mechanism of synergy between the antimicrobial peptides PGLa and magainin 2. Sci. Rep. 7:13153. 10.1038/s41598-017-12599-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang J., Yang Y., Teng D., Tian Z., Wang S., Wang J. (2011). Expression of plectasin in Pichia pastoris and its characterization as a new antimicrobial peptide against Staphyloccocus and Streptococcus. Protein Expr. Purif. 78, 189–196. 10.1016/j.pep.2011.04.014 [DOI] [PubMed] [Google Scholar]
- Zhang P., Wright J. A., Tymon A., Nair S. P. (2018). Bicarbonate induces high-level resistance to the human antimicrobial peptide LL-37 in Staphylococcus aureus small colony variants. J. Antimicrob. Chemother. 73, 615–619. 10.1093/jac/dkx433 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Z. T., Zhu S. Y. (2009). Drosomycin, an essential component of antifungal defence in Drosophila. Insect Mol. Biol. 18, 549–556. 10.1111/j.1365-2583.2009.00907.x [DOI] [PubMed] [Google Scholar]
- Zheng X., Wang X., Teng D., Mao R., Hao Y., Yang N., et al. (2017). Mode of action of plectasin-derived peptides against gas gangrene-associated Clostridium perfringens type A. PLoS ONE 12:e0185215. 10.1371/journal.pone.0185215 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu C., Tan H., Cheng T., Shen H., Shao J., Guo Y., et al. (2013). Human beta-defensin 3 inhibits antibiotic-resistant Staphylococcus biofilm formation. J. Surg. Res. 183, 204–213. 10.1016/j.jss.2012.11.048 [DOI] [PubMed] [Google Scholar]
- Zion Market Research (2018). Global Peptide Therapeutics Market. Zion Market Research. Available online at: https://www.zionmarketresearch.com/news/peptide-therapeutics-market. (accessed April 21, 2020).


