Abstract
Aims
Clopidogrel is prescribed for the prevention of atherothrombotic events. While investigations have identified genetic determinants of inter-individual variability in on-treatment platelet inhibition (e.g. CYP2C19*2), evidence that these variants have clinical utility to predict major adverse cardiovascular events (CVEs) remains controversial.
Methods and results
We assessed the impact of 31 candidate gene polymorphisms on adenosine diphosphate (ADP)-stimulated platelet reactivity in 3391 clopidogrel-treated coronary artery disease patients of the International Clopidogrel Pharmacogenomics Consortium (ICPC). The influence of these polymorphisms on CVEs was tested in 2134 ICPC patients (N = 129 events) in whom clinical event data were available. Several variants were associated with on-treatment ADP-stimulated platelet reactivity (CYP2C19*2, P = 8.8 × 10−54; CES1 G143E, P = 1.3 × 10−16; CYP2C19*17, P = 9.5 × 10−10; CYP2B6 1294 + 53 C > T, P = 3.0 × 10−4; CYP2B6 516 G > T, P = 1.0 × 10−3; CYP2C9*2, P = 1.2 × 10−3; and CYP2C9*3, P = 1.5 × 10−3). While no individual variant was associated with CVEs, generation of a pharmacogenomic polygenic response score (PgxRS) revealed that patients who carried a greater number of alleles that associated with increased on-treatment platelet reactivity were more likely to experience CVEs (β = 0.17, SE 0.06, P = 0.01) and cardiovascular-related death (β = 0.43, SE 0.16, P = 0.007). Patients who carried eight or more risk alleles were significantly more likely to experience CVEs [odds ratio (OR) = 1.78, 95% confidence interval (CI) 1.14–2.76, P = 0.01] and cardiovascular death (OR = 4.39, 95% CI 1.35–14.27, P = 0.01) compared to patients who carried six or fewer of these alleles.
Conclusion
Several polymorphisms impact clopidogrel response and PgxRS is a predictor of cardiovascular outcomes. Additional investigations that identify novel determinants of clopidogrel response and validating polygenic models may facilitate future precision medicine strategies.
Keywords: Clopidogrel, Pharmacogenetics, Platelet aggregation
Introduction
Clopidogrel is prescribed for the prevention of atherothrombotic events in patients with coronary artery disease.1 Given its effectiveness and cost, clopidogrel continues to be used frequently compared to newer alternatives such as prasugrel and ticagrelor.2,3 However, substantial inter-individual variability in clopidogrel response exists, and patients who experience sub-optimal therapy have high on-treatment platelet reactivity (HPR) and are at greater risk of experiencing a recurrent event.
Variability in clopidogrel efficacy is caused by multiple factors, including age, sex, smoking, existing comorbidities, and drug–drug interactions.4 However, these factors explain a small proportion of the total phenotypic variation in clopidogrel response. Recent studies indicate that genetic variation substantially impacts clopidogrel efficacy, accounting for ∼75% of the variability in response, as assessed by agonist-induced platelet aggregation.5
Multiple studies have been performed to identify genetic determinants of clopidogrel response. Though several candidate gene polymorphisms, most notably the loss-of-function CYP2C19*2 variant, have been identified, relatively small sample sizes have hampered discovery efforts, and discrepant findings in replication efforts have made it difficult to draw firm conclusions. Here, we assessed the role of 31 candidate single nucleotide polymorphisms (SNPs) implicated in modulating clopidogrel transport (e.g. ABCB1), metabolism (e.g. CYP450 enzymes, PON1, CES1), and mechanism of action (e.g. P2Y12) in a large cohort of 3391 clopidogrel-treated patients of the International Clopidogrel Pharmacogenomics Consortium (ICPC). We also evaluated these polymorphisms on cardiovascular event (CVE) risk. Our results suggest that application of a pharmacogenomic polygenic response score (PgxRS), analogous to polygenic risk score for complex diseases, may be a superior predictive model with potential application to precision antiplatelet medicine.
Methods
Study sites and participants
Participating ICPC sites and patient characteristics have been previously described.6 Briefly, ∼150 investigators who led clopidogrel-related clinical studies of 50 or more subjects and for which DNA samples, on-clopidogrel platelet reactivity data, and clinical outcome information available were invited to participate. In total, 17 centres from 13 countries contributed data. Data deposition into a central database was managed by the Pharmacogenomic Knowledge Base (PharmGKB) and data quality/accuracy was assessed by the Pharmacogenomics Research Network’s Statistical Analysis Resource. This report includes 3391 Caucasian ICPC subjects in whom on-clopidogrel platelet reactivity data, covariate data, and DNA were available.
All protocols were approved by their respective institutional review boards and adhered to the Declaration of Helsinki. Informed Consent was obtained from each participant.
Study outcomes
Platelet reactivity
In ICPC sites, one or more of the following platelet reactivity assays were used to assess clopidogrel response: Vasodilator-Stimulated Phosphoprotein (VASP) assay, Light Transmittance Aggregometry (LTA), Multiplate, and the VerifyNow P2Y12 assay. While all of these assays assess adenosine diphosphate (ADP)-stimulated platelet reactivity, differences in assay biochemistry and units to express each test result required the generation of a standardized platelet reactivity phenotype. The criteria to generate this phenotype were reported previously.6 Briefly, for sites that used more than one platelet test, the following prioritization scheme was predefined and applied: VASP assay > VerifyNow P2Y12 > LTA (higher > lower ADP concentration) > Multiplate. The standardized mean difference of the chosen platelet test within each site was calculated by subtracting mean platelet reactivity from the observed platelet reactivity and dividing by the standard deviation to generate a Z-score, thus harmonizing platelet reactivity data across sites.
Cardiovascular endpoints
Post-discharge CVEs were evaluated in a subset of 2134 patients in whom clinical event data were available and were defined as spontaneous myocardial infarction (troponin value greater than the upper-limits of normal with ischaemic symptoms or electrocardiogram changes), ischaemic stroke, stent thrombosis (probable or definite stent thrombosis according to the Academic Research Consortium criteria7), and cardiovascular-related death. A composite endpoint, consisting of all the aforementioned outcomes, was used for analysis. Individual components of the composite endpoint were also evaluated. Study site physicians locally adjudicated all endpoints through review of source documents obtained from medical records. More information regarding CVE determination and adjudication has been reported previously.6
Genotyping
Thirty-one candidate polymorphisms were chosen based on prior literature and unpublished data by consortium members. Genotyping was performed with the QuantStudio™ 12K Flex OpenArray® System (Thermo Fisher Scientific, Waltham, MA, USA) according to the manufacturer’s instructions. Genotype calls were performed using QuantStudio™ 12K Flex Software v1.2.2. The mean SNP call rate was 98.6% and overall concordance rate in a subset of blind duplicates was 99.3%.
Statistical analyses
Summary statistics, frequencies, and measures of Hardy–Weinberg equilibrium were calculated using PLINK v1.07 (https://www.cog-genomics.org/plink2). Linkage disequilibrium (LD) metrics (|D’| and r2) were calculated using Haploview v4.2.8 To account for multiple testing, P-values < 0.0016 were considered statistically significant (0.05/31 SNPs) in single-SNP models. All statistical tests were two-sided.
Association analyses with platelet reactivity were performed using linear regression under an additive genetic model while adjusting for age, sex, site, body mass index (BMI), smoking, diabetes, and proton pump inhibitor (PPI) use (PLINK v1.07). For analyses of CVEs, we used logistic regression and adjusted for the same covariates. For variants that were significantly associated with on-clopidogrel platelet reactivity in single-SNP analyses (N = 6), a PgxRS was developed to assess the impact of multiple alleles (min = 0; max = 12) that resulted in increased platelet reactivity on the occurrence of CVEs. For this analysis, alleles that led to increased platelet reactivity were coded as ‘risk’ and logistic regression was used using identical covariates. The impact of carrying a high (eight or above) vs. low (six or fewer) number of alleles that were associated with increased platelet reactivity on CVEs was calculated. Weighted risk score analyses that accounted for genotype effect sizes were also performed. The proportion of variation in on-clopidogrel platelet reactivity explained by the PgxRS was calculated using SAS v9.4 (SAS Institute, Cary, NC, USA).
Power calculations (N = 3391) indicated 80% power at ά = 0.002 to detect SNPs with minor allele frequencies (MAF) ranging from 0.01 to 0.30 with effect sizes ranging from 0.48 to 0.11 standard deviation units, respectively. In CVE analyses, we had 80% power to detect odds ratios (ORs) ranging from 1.70 (MAF = 0.30) to 2.55 (MAF = 0.05) given a sample size of 2134 and 129 recorded events (ά = 0.002). Power calculations were performed using Quanto v1.2.4 (http://biostats.usc.edu/Quanto.html).
Results
Characteristics of the participants are listed in Table 1. Most subjects were male (72.7%), middle-aged (mean age = 60.8 years), overweight (mean BMI = 27.4 kg/m2), and had high prevalence of conventional risk factors [e.g. hypertension (81.6%), hypercholesterolaemia (86.4%), and diabetes (18.3%)]. Most subjects were taking aspirin (94.3%), 64.4% were prescribed statins, and 21.1% were on PPIs.
Table 1.
Characteristics of candidate gene International Clopidogrel Pharmacogenomics Consortium cohort
| Characteristics (units) | Male | Female | All |
|---|---|---|---|
| Number (N) | 2464 | 927 | 3391 |
| Age (years), mean ± SD | 60.8 ± 12.8 | 60.6 ± 15.7 | 60.8 ± 13.6 |
| BMI (kg/m2), mean ± SD | 27.3 ± 4.0 | 27.7 ± 5.1 | 27.4 ± 4.3 |
| Systolic blood pressure (mmHg), mean ± SD | 134.3 ± 23.1 | 127.9 ± 23.0 | 131.9 ± 23.3 |
| Diastolic blood pressure (mmHg), mean ± SD | 77.7 ± 12.2 | 73.5 ± 10.5 | 76.1 ± 11.7 |
| Hypertension,an (%) | 2142 (86.9) | 625 (67.4) | 2767 (81.6) |
| Total cholesterol (mg/dL), mean ± SD | 181.6 ± 49.9 | 205.3 ± 52.9 | 189.0 ± 52.0 |
| LDL-cholesterol (mg/dL), mean ± SD | 115.9 ± 42.9 | 130.3 ± 47.4 | 120.4 ± 44.9 |
| HDL-cholesterol (mg/dL), mean ± SD | 48.2 ± 14.8 | 59.6 ± 16.1 | 51.7 ± 16.1 |
| Triglycerides (mg/dL), mean ± SD | 130.5 ± 112.8 | 101.0 ± 66.8 | 121.3 ± 101.7 |
| Hypercholesterolaemia,bn (%) | 2216 (89.9) | 714 (77.0) | 2930 (86.4) |
| Taking aspirin, n (%) | 2309 (93.7) | 889 (95.9) | 3198 (94.3) |
| Taking statins, n (%) | 1734 (70.4) | 450 (48.5) | 2184 (64.4) |
| Taking PPI, n (%) | 538 (21.8) | 177 (19.1) | 715 (21.1) |
| Self-reported diabetes, n (%) | 449 (18.2) | 171 (18.3) | 620 (18.3) |
| Haematocrit ± SD (%) | 41.6 ± 4.0 | 37.9 ± 3.4 | 40.5 ± 4.2 |
| White blood cell count (n × 1000), mean ± SD | 7.2 ± 3.1 | 6.7 ± 2.5 | 7.1 ± 2.9 |
| Platelet count (n × 100 000), mean ± SD | 2.3 ± 0.7 | 2.5 ± 0.7 | 2.4 ± 0.7 |
| Ever smoker,cn (%) | 1444 (58.6) | 242 (26.2) | 1686 (49.7) |
| Current smoker,cn (%) | 485 (19.7) | 85 (9.2) | 570 (16.8) |
SI conversion factors: to convert HDL-cholesterol, LDL-cholesterol, and total cholesterol values to mmol/L, multiply by 0.0259; triglycerides to mmol/L, multiply by 0.0113.
BMI, body mass index; HDL, high-density lipoprotein; LDL, low-density lipoprotein; PPI, proton pump inhibitor; SD, standard deviation.
Defined as systolic blood pressure >140 mmHg, diastolic blood pressure >90 mmHg, taking prescription blood pressure-lowering medication, and/or source code for hypertension.
Defined as LDL-cholesterol >160 mm⋅mg/dL, taking prescription cholesterol-lowering medication, and/or source code for hypercholesterolaemia.
Self-reported history of smoking.
Associations with platelet reactivity
We performed single-SNP association tests between all polymorphisms and platelet reactivity (Table 2, Supplementary material online, Figure S1, and Supplementary material online, Table S1). Participants who carried the loss-of-function CYP2C19*2 allele (rs4244285) had significantly higher platelet reactivity compared to non-carriers (β = 0.51, P = 8.8 × 10−54). Participants who carried the loss-of-function CES1 G143E allele (rs71647871) had significantly lower on-treatment platelet reactivity compared to non-carrier patients (β = −0.79, P = 1.3 × 10−16). Similarly, those who carried the putative gain-of-function CYP2C19*17 allele (rs12248560) had better clopidogrel response compared to non-carriers (β = −0.18, P = 9.5 × 10−10). Polymorphisms in CYP2B6 (rs8192719 and rs3745274) and CYP2C9 (rs1057910) were associated with increased platelet reactivity (β = 0.11, P = 3.0 × 10−4, β = 0.10, 1.0 × 10−3, and β = 0.15, P = 0.001, respectively). In contrast, CYP2C9*2 (rs1799853) was associated with lower on-treatment platelet reactivity (β = −0.11, P = 0.001). After correction for multiple testing, no other variant showed association with platelet reactivity (Table 2).
Table 2.
Association of candidate gene single nucleotide polymorphisms with adenosine diphosphate-stimulated platelet aggregation in International Clopidogrel Pharmacogenomics Consortium participants
| Gene | Allele designation | rs number | Chromosome | MAF | HWE P-value | Beta | P-value | CYP2C19*2 adjusted P-value |
|---|---|---|---|---|---|---|---|---|
| CYP2C19 | *2 | rs4244285 | 10q23.33 | 0.163 | 0.084 | 0.506 | 8.80 × 10−54 | NA |
| CES1 | G143E | rs71647871 | 16q12.2 | 0.016 | 0.631 | −0.787 | 1.30 × 10−16 | 4.27 × 10−18 |
| CYP2C19 | *17 | rs12248560 | 10q23.33 | 0.219 | 0.755 | −0.177 | 9.48 × 10−10 | 0.006 |
| CYP2B6 | 1294 + 53C > T | rs8192719 | 19q13.2 | 0.229 | 0.013 | 0.105 | 2.99 × 10−4 | 1.15 × 10−4 |
| CYP2B6 | Q172H | rs3745274 | 19q13.2 | 0.215 | 0.032 | 0.099 | 9.98 × 10−4 | 3.53 × 10−4 |
| CYP2C9 | *2 | rs1799853 | 10q23.33 | 0.151 | 0.022 | −0.113 | 0.001 | 0.489 |
| CYP2C9 | *3 | rs1057910 | 10q23.33 | 0.072 | 0.908 | 0.153 | 0.001 | 2.81 × 10−7 |
| PEAR1 | NA | rs12041331 | 1q23.1 | 0.123 | 0.005 | −0.116 | 0.003 | 4.15 × 10−4 |
| P2RY12 | T744C | rs2046934 | 3q25.1 | 0.175 | 0.003 | 0.088 | 0.004 | 0.003 |
| CES1 | NA | rs2244613 | 16q12.2 | 0.206 | 1.000 | 0.080 | 0.007 | 0.006 |
| MED12L | NA | rs1472122 | 3q25.1 | 0.480 | 0.715 | −0.063 | 0.009 | 0.002 |
| ITGB3 | L59P | rs5918 | 17q21.32 | 0.150 | 0.437 | −0.081 | 0.016 | 0.005 |
| CYP2C19 | *8 | rs41291556 | 10q23.33 | 0.002 | 0.016 | 0.560 | 0.020 | 0.008 |
| CYP2B6 | 485-18C > T | rs4803419 | 19q13.2 | 0.310 | 0.414 | 0.058 | 0.026 | 0.023 |
| CYP2B6 | 14593C > G | rs4803418 | 19q13.2 | 0.306 | 0.444 | 0.056 | 0.038 | 0.033 |
| CYP2B6 | *1B | rs7254579 | 19q13.2 | 0.322 | 0.972 | 0.051 | 0.050 | 0.045 |
| CYP2C19 | *4 | rs28399504 | 10q23.33 | 0.003 | 1.000 | −0.488 | 0.065 | 0.197 |
| CYP2C19 | *3 | rs4986893 | 10q23.33 | 0.001 | 1.000 | 0.709 | 0.107 | 0.071 |
| POR | IVS11 + 20G > A | rs2286823 | 7q11.23 | 0.293 | 0.071 | 0.040 | 0.125 | 0.086 |
| CYP2C9 | *11 | rs28371685 | 10q23.33 | 0.004 | 1.000 | 0.307 | 0.167 | 0.772 |
| CYP2C19 | *6 | rs72552267 | 10q23.33 | 0.002 | 0.024 | 0.335 | 0.175 | 0.198 |
| POR | *28 | rs1057868 | 7q11.23 | 0.288 | 0.249 | −0.032 | 0.234 | 0.240 |
| POR | NA | rs2302429 | 7q11.23 | 0.145 | 6.41 × 10 − 5 | −0.036 | 0.296 | 0.354 |
| CYP2C19 | *7 | rs72558186 | 10q23.33 | 0.002 | 4.03 × 10 − 8 | −0.248 | 0.332 | 0.585 |
| CYP1A2 | *1F | rs762551 | 15q24.1 | 0.317 | 0.648 | −0.024 | 0.343 | 0.521 |
| ABCB1 | C1236T | rs1128503 | 7q21.12 | 0.408 | 0.875 | 0.021 | 0.405 | 0.318 |
| PON1 | L55M | rs854560 | 7q21.3 | 0.371 | 0.359 | −0.019 | 0.456 | 0.617 |
| CYP2C19 | *5 | rs56337013 | 10q23.33 | 2.32 × 10−4 | 1.000 | −0.600 | 0.542 | 0.627 |
| CYP3A5 | *3F | rs28365085 | 7q22.1 | 2.31 × 10−4 | 1.16 × 10−4 | 0.163 | 0.741 | 0.621 |
| PON1 | Q192R | rs662 | 7q21.3 | 0.290 | 0.853 | 0.007 | 0.785 | 0.872 |
| ABCB1 | C3435T | rs1045642 | 7q21.12 | 0.496 | 0.107 | 0.007 | 0.786 | 0.614 |
Association P-values in bold represent those that are statistically significant after correction for multiple testing (P = 0.05/31 = 0.0016). Italicized Hardy–Weinberg P-values denote deviations from expected Hardy–Weinberg proportions (cut-off P-value <1.0 × 10−4).
HWE, Hardy–Weinberg equilibrium; MAF, minor allele frequency; NA, not applicable.
Given the strong association between CYP2C19*2 and platelet reactivity, we sought to determine if association signals of other variants might be unmasked by adjusting for this variant (Table 2). After adjustment, association between variants in CES1 and PEAR1 and platelet reactivity were strengthened, while associations of CYP2B6 SNPs rs8192719 and rs3745274 with platelet reactivity were essentially unchanged (Table 2). Other variants that were not associated with platelet reactivity in the CYP2C19*2 unadjusted model remained without significant association after adjustment.
The CYP2C locus on chromosome 10q24 includes several variants tested in this investigation and, through LD, could be associated with platelet reactivity because it is physically linked to a causative variant without having an independent effect. Indeed, adjustment for CYP2C19*2 ablated association of CYP2C19*17 with clopidogrel response after correction for multiple testing (P = 0.006). Adjustment for CYP2C19*2 also influenced the magnitude of association between platelet reactivity and the two CYP2C9 SNPs (Table 2).
Since adjustment for CYP2C19*2 substantially altered the association between CYP2C19*17 with platelet reactivity, we assessed LD patterns between these polymorphisms and performed genotype-stratified association analyses (Supplementary material online, Table S2A). The CYP2C19*2 and *17 variants (minor alleles of rs4244285 and rs12248560, respectively) are located ∼20 kbp from each other, and consistent with prior reports,9 the rs12248560 minor allele occurs only on the rs4244285 major allele haplotype (i.e. the so-called *1 allele; |D’|=1.0). Stratified analyses showed that the minor allele at rs4244285 (CYP2C19*2) remained strongly associated with platelet reactivity in patients who were homozygous for the major allele at rs12248560 (N = 2068, P = 7.9 × 10−37), while association of the minor allele of rs12248560 (CYP2C19*17) with platelet aggregation was markedly attenuated in patients who were homozygous for major allele (N = 2386, P = 0.01) or heterozygotes (N = 933, P = 0.09) at rs4244285 (Supplementary material online, Figure S2). These analyses provide further evidence that CYP2C19*17 is not an independent predictor of on-clopidogrel platelet reactivity, but rather dependent on rs4244285 genotype.
Similarly, we assessed LD and performed stratified association analyses between CYP2C19*2 and the 2 CYP2C9 SNPs [*2 (minor allele at rs1799853) and *3 (minor allele at rs1057910)]. LD data and estimation of haplotype frequencies are shown in Supplementary material online, Tables S2B and S2C. While CYP2C19*2 was strongly associated with platelet reactivity in patients who were major allele homozygotes of rs1799853 (N = 2444, P = 6.8 × 10−10), the CYP2C9 minor allele at rs1799853 (CYP2C9*2) was not associated with platelet reactivity in CYP2C19*1 homozygotes (N = 2375, P = 0.69). In contrast, when we evaluated the impact of CYP2C9 minor allele of rs1057910 (CYP2C9*3) in CYP2C19*1 homozygotes (N = 2375), we observed a significant association between this SNP and clopidogrel response (P = 1.7 × 10−4), suggesting that rs1057910 increases platelet reactivity independently of CYP2C19*2.
Associations with clinical outcomes
We evaluated the impact of investigated polymorphisms on CVEs in a subset of 2134 patients for whom event data were available. Clinical characteristics of this subset of participants are shown in Supplementary material online, Table S3. In total, 129 of the 2134 patients experienced an event (event rate = 6.0% over a mean follow-up of ∼13 months). No single variant was associated with CVEs before or after adjustment for CYP2C19*2 (Supplementary material online, Table S4). Similarly, we observed no evidence of association between polymorphisms and the individual components of the composite events phenotype, although the number of events and statistical power for these sub-analyses was considerably diminished (Supplementary material online, Table S5).
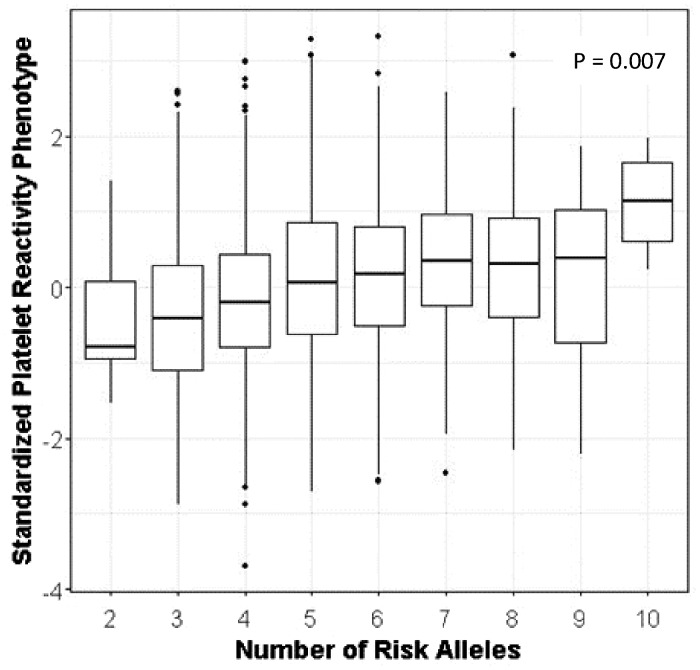
Finally, we developed a PgxRS to assess the impact of carrying multiple alleles that impact platelet reactivity on the occurrence of CVEs. Specifically, we used CYP2C19*2 and the five SNPs that were significantly associated with platelet reactivity after adjustment for CYP2C19*2 and coded each allele that corresponded to increased platelet reactivity as the ‘risk’ allele (Figure 1). Collectively, these SNPs accounted for ∼3.5% of the variation in platelet reactivity. We observed that patients who carry an increasing number of alleles that are associated with high on-clopidogrel platelet reactivity were more likely to experience CVEs and cardiovascular-related death compared to those who carry alleles that lead to better platelet inhibition (β = 0.17, P = 0.01 and β = 0.43, P = 0.007, respectively). Consistent with these findings, when comparing patients by number of risk alleles, patients who carried eight or more alleles associated with increased platelet reactivity were more likely to experience both CVEs [OR = 1.78, 95% confidence interval (CI) 1.14–2.76, P = 0.01] and cardiovascular death (OR = 4.39, 95% CI 1.35–14.27, P = 0.01) compared to patients who carried six or fewer of these alleles. Weighting these results based on genotype effect size did attenuate significant findings (CVE P = 0.07) but provided comparable OR estimates (Table 3).
Figure 1.
Change in standardized adenosine diphosphate-induced platelet reactivity based on increasing number of alleles used in the pharmacogenomic polygenic response score. For each box plot, the horizontal line within each box indicates the median; the top and bottom borders of each box indicate the interquartile range. The whiskers extending from each box indicate the 95% confidence interval and the points beyond the whiskers indicate outliers beyond ±2.5% CI. Platelet reactivity is expressed as Z-scores as described in the Methods section.
Table 3.
Polygenic response score of alleles associated with platelet reactivity on the occurrence of cardiovascular endpoints
| Raw analysis |
Weighted analysis |
|||||
|---|---|---|---|---|---|---|
| Trait | OR | 95% CI | P-value | OR | 95% CI | P-value |
| Composite CVE phenotype | 1.78 | 1.14–2.76 | 0.01 | 1.55 | 0.97–2.48 | 0.07 |
| Myocardial infarction | 1.70 | 0.99–2.91 | 0.05 | 1.55 | 0.87–2.76 | 0.13 |
| Cardiovascular death | 4.39 | 1.35–14.27 | 0.01 | 2.49 | 0.81–7.56 | 0.11 |
| Stent thrombosis | 1.42 | 0.62–3.27 | 0.41 | 1.17 | 0.48–2.85 | 0.73 |
| Stroke | 1.23 | 0.57–2.66 | 0.90 | 1.04 | 0.45–2.39 | 0.93 |
Polygenic response score was derived from variants that were associated with ADP-based platelet reactivity in single-SNP analyses (CYP2C19*2, CES1 G143E, CYP2B6 1294 + 53 C > T, CYP2B6 Q172H, CYP2C9*3, and PEAR1 rs12041331) and alleles that led to increased ADP-based platelet reactivity were coded as ‘risk’. Odds ratios were calculated comparing patients who carried eight or more alleles associated with increased platelet reactivity with patients who carried six or fewer of these alleles. Analyses were conducted in the 2134 ICPC participants in which clinical event data were available.
CI, confidence interval; CVE, cardiovascular event; OR, odds ratio.
Discussion
We assessed 31 candidate gene polymorphisms on platelet reactivity and CVEs in clopidogrel-treated Caucasian participants of the ICPC. Consistent with prior reports, we observed that the CYP2C19*2 allele was a strong determinant of on-clopidogrel platelet reactivity.5,10,11 We also observed that CES1 G143E strongly influenced clopidogrel response with carriers of the 143E-allele having decreased on-treatment platelet reactivity. CES1 is responsible for converting clopidogrel into an inactive carboxylic acid derivative.12 In fact, ∼85% of administered clopidogrel is metabolized into inactive derivatives by CES1.13 The 143E-allele results in an approximate 83% reduction in CES1 activity compared to the wild type 143G-allele, and is among the strongest determinant of clopidogrel response using physiology-directed pharmacokinetic/pharmacodynamic modelling.14,15 These results suggest that comprehensive characterization of CES1 G143E in clopidogrel-treated patients with regard to potential for pathological bleeding is needed.
We also assessed the well-described putative gain-of-function CYP2C19*17 variant on platelet reactivity. CYP2C19*17 is believed to lead to better inhibition of platelet aggregation by increasing transcription of the CYP2C19 gene.16 We initially observed that CYP2C19*17 resulted in reduced platelet reactivity. However, in conditional analyses adjusting for CYP2C19*2, no association was evident. These results are consistent with prior data showing that CYP2C19*17 association results are highly influenced by CYP2C19*2 through LD.9 In stratified analyses, markedly attenuated association between CYP2C19*17 and platelet reactivity was observed in patients who did not carry the CYP2C19*2 allele. These results suggest that CYP2C19*17 has little to no independent effect on clopidogrel response. This is particularly noteworthy given that multiple investigations have reported CYP2C19*17 association results without taking into account the effects of CYP2C19*2 and the fact that current pharmacogenetic prescribing algorithms may be incorrectly classifying patients who carry the CYP2C19*17 allele as ultra-rapid clopidogrel metabolizers.17
Association between platelet reactivity and SNPs in CYP2C9 was significantly influenced by CYP2C19*2 adjustment. Rs1799853, which encodes CYP2C9*2, was no longer associated with clopidogrel response while rs1057910, which encodes CYP2C9*3, was more strongly associated with platelet reactivity. Evaluation of LD between CYP2C19*2 and CYP2C9 SNPs showed that while CYP2C9 rs1799853 does not independently influence clopidogrel efficacy, rs1057910 increases platelet reactivity independently of CYP2C19*2.
Two SNPs in CYP2B6 (rs8192719 and rs3745274) and one SNP in PEAR1 (rs12041331) were associated with platelet reactivity after adjustment for CYP2C19*2. Previous investigations of CYP2B6 on clopidogrel response have shown mixed results.18,19 Of note, many of these investigations were conducted in a relatively small sample and likely had limited statistical power. PEAR1 is a transmembrane receptor that is important in platelet aggregation-induced secondary signalling.20 Rs12041331, which is an intronic variant proposed to modify PEAR1 expression through differences in allele-specific DNA methylation,21 has been implicated in baseline22 and on-treatment aspirin,23 ticagrelor,24 and dual antiplatelet therapy with aspirin and clopidogrel25 platelet aggregation and myocardial infarction in stable coronary artery disease patients.25 Additional studies in large, well-powered cohorts will be needed to further define the role of CYP2B6 and PEAR1 in clopidogrel response.
While several polymorphisms in this investigation influenced platelet reactivity, none were individually associated with CVEs. This was relatively surprising, particularly for CYP2C19*2, as there is an increasing amount of evidence that suggests this variant increases risk of cardiovascular outcomes, particularly in acute coronary syndrome patients undergoing percutaneous coronary intervention. Indeed, several high-profile investigations including the TRITON-TIMI 38 trial,26 FAST-MI,27 PLATO Genetic Substudy,28 and IGNITE network29 have shown that clopidogrel-treated patients who carry this allele have worse clinical outcomes compared to those who do not. Interestingly, however, when we combined the impact of multiple SNPs through generation of a PgxRS, it revealed that patients who carried a greater number of alleles that lead to increased platelet reactivity were more likely to experience CVEs. While significance was diminished in weighted analyses, OR point estimates do suggest enhanced risk of CVEs in these patients. These findings may have important implications. Specifically, for complex phenotypes such as CVEs, multiple genetic and non-genetic factors are critical in the development (or prevention) of the clinical outcome. Given this complexity, it is often difficult to reproducibly identify any one factor of small to moderate effect, and clinical utility of any single factor to predict risk and act to prevent adverse clinical outcomes is poor.30,31 Our findings underscore the importance of considering multiple sources of genetic variability. Therefore, we highly encourage strategies such as those used in the current investigation to construct robust genetic response scores of platelet reactivity to evaluate potential impact on clinical outcomes. As shown here, these response scores can be built-in cohorts that utilize several platelet function tests through use of statistical approaches that implement Z-scores as well as cohorts that have limited genetic and platelet function data. The use of such strategies by the scientific community will be required to determine whether a clopidogrel PgxRS has high enough predictive value to identify those at high risk of recurrent CVEs in whom an alternative anti-platelet agent might be indicated.
This investigation has limitations. All participants evaluated were of European descent. Therefore, caution should be used in extrapolating these findings to populations of different ethnic or racial origins. This may be particularly important for polymorphisms that vary considerably in frequency across populations. For example, CYP2C19*3, which is more common in Asian populations, and other reported loss-of-function variants in CYP2C19 (i.e. CYP2C19*4–8) were extremely rare in this population, consistent with other European-derived populations; therefore, caution should be used when interpreting the negative association results reported here as statistical power of analyses related to these variants is low. In addition, the prognostic value of the HPR phenotype is not established in patients at low cardiovascular risk even if they previously had a myocardial infarction.32 While CYP2C19*2 and the PgxRS used are strong determinants of platelet reactivity in these patients, the association with clinical outcomes will require further investigation. Finally, this was a retrospective study. Prospective studies will be required to determine whether alternative antiplatelet therapy can ameliorate increased risk of CVEs in high PgxRS patients.
In summary, we have performed one of the largest studies of clopidogrel pharmacogenetics conducted to date. Our findings suggest that several polymorphisms independently influence on-clopidogrel platelet reactivity and that accumulation of these alleles may increase CVE risk. The availability of alternative treatment options for patients with clinical indications for clopidogrel underscores the need to better understand the genetic architecture of variable drug response which is critical for optimizing anti-platelet pharmacotherapy, reducing therapeutic failure and adverse side effects, and ultimately improving patient outcomes.
Supplementary material
Supplementary material is available at European Heart Journal – Cardiovascular Pharmacotherapy online.
Funding
This work was supported by the National Institutes of Health [U01 GM074518, U01 HL105198, U19 HL065962, R01 HL137922, and K23 GM102678].
Conflict of interest: J.P.L. reports NIH grant support to study the pharmacogenetics of antiplatelet therapy. W.H. reports consulting and lecture fees from AstraZeneca, Boehringer Ingelheim, Daiichi Sankyo, and The Medicines Company. M.D.R. reports grants from the NIH. D.A. reports personal fees from DSI/Lilly and AstraZeneca. M.S., E.S., and S.W. are supported by the DFG Germany [grant number SCHW858/1–2] and in part by the EU Horizon 2020 UPGx grant [668353] and the Robert Bosch Stiftung Stuttgart, Germany. D.T. reports personal fees from Amgen, AstraZeneca, Bayer, Berlin Chemie, Boehringer Ingelheim, Bristol-Myers Squibb, Daiichi Sankyo, Pfizer, and Sanofi. T.G. reports grants or personal fees from Spartan Bioscience, Astra Zeneca, Bayer Healthcare, Bristol Myers Squibb, Pfizer, Daiichy Sankyo, and Novartis. M.V. reports grants or personal fees from Abbott, Chiesi, Bayer, Daiichi Sankyo, Amgen, Terumo, Alvimedica, Medicure, AstraZeneca, Biosensors, and Idorsia. J.M.S.-M. reports personal fees from AstraZeneca, Daiichi Sankyo, Eli Lilly, Roche, and Bayer. P.F. reports non-financial support from NovoNordisk, Bayer, and Sobi. D.A. reports receiving advisory board fees from AstraZeneca, Bayer, Boehringer Ingelheim, and the Medicines Company, and lecture fees from AstraZeneca. P.A.G. reports grants from Amgen, DCRI, Haemonetics, Idorsia, Ionis, Janssen, Merck, NIH, disclosures from Boehringer, Merck, Janssen, and Bayer as well as a patent for platelet function testing. R.B.A. reports disclosures from Personalis, Youscript, and 23andMe. T.O.B. received a personal grant from the St. Antonius Research Fund. J.D.B. is an employee of Regeneron Pharmaceuticals, Inc. A.R.S. reports grants from the National Institutes of Health that funded this work and is an employee of Regeneron Pharmaceuticals, Inc.
Supplementary Material
References
- 1. Amsterdam EA, Wenger NK, Brindis RG, Casey DE Jr, Ganiats TG, Holmes DR Jr, Jaffe AS, Jneid H, Kelly RF, Kontos MC, Levine GN, Liebson PR, Mukherjee D, Peterson ED, Sabatine MS, Smalling RW, Zieman SJ.. 2014 AHA/ACC Guideline for the management of patients with non-ST-elevation acute coronary syndromes: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol 2014;64:e139–e228. [DOI] [PubMed] [Google Scholar]
- 2. Wiviott SD, Braunwald E, McCabe CH, Montalescot G, Ruzyllo W, Gottlieb S, Neumann FJ, Ardissino D, De Servi S, Murphy SA, Riesmeyer J, Weerakkody G, Gibson CM, Antman EM; TRITON–TIMI 38 Investigators. Prasugrel versus clopidogrel in patients with acute coronary syndromes. N Engl J Med 2007;357:2001–2015. [DOI] [PubMed] [Google Scholar]
- 3. Wallentin L, Becker RC, Budaj A, Cannon CP, Emanuelsson H, Held C, Horrow J, Husted S, James S, Katus H, Mahaffey KW, Scirica BM, Skene A, Steg PG, Storey RF, Harrington RA, Investigators P, Freij A, Thorsen M.. Ticagrelor versus clopidogrel in patients with acute coronary syndromes. N Engl J Med 2009;361:1045–1057. [DOI] [PubMed] [Google Scholar]
- 4. Siller-Matula JM, Trenk D, Krahenbuhl S, Michelson AD, Delle-Karth G.. Clinical implications of drug-drug interactions with P2Y12 receptor inhibitors. J Thromb Haemost 2014;12:2–13. [DOI] [PubMed] [Google Scholar]
- 5. Shuldiner AR, O'Connell JR, Bliden KP, Gandhi A, Ryan K, Horenstein RB, Damcott CM, Pakyz R, Tantry US, Gibson Q, Pollin TI, Post W, Parsa A, Mitchell BD, Faraday N, Herzog W, Gurbel PA.. Association of cytochrome P450 2C19 genotype with the antiplatelet effect and clinical efficacy of clopidogrel therapy. JAMA 2009;302:849–857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Bergmeijer TO, Reny JL, Pakyz RE, Gong L, Lewis JP, Kim EY, Aradi D, Fernandez-Cadenas I, Horenstein RB, Lee MTM, Whaley RM, Montaner J, Gensini GF, Cleator JH, Chang K, Holmvang L, Hochholzer W, Roden DM, Winter S, Altman RB, Alexopoulos D, Kim HS, Dery JP, Gawaz M, Bliden K, Valgimigli M, Marcucci R, Campo G, Schaeffeler E, Dridi NP, Wen MS, Shin JG, Simon T, Fontana P, Giusti B, Geisler T, Kubo M, Trenk D, Siller-Matula JM, Ten Berg JM, Gurbel PA, Hulot JS, Mitchell BD, Schwab M, Ritchie MD, Klein TE, Shuldiner AR; ICPC Investigators. Genome-wide and candidate gene approaches of clopidogrel efficacy using pharmacodynamic and clinical end points-Rationale and design of the International Clopidogrel Pharmacogenomics Consortium (ICPC). Am Heart J 2018;198:152–159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Cutlip DE, Windecker S, Mehran R, Boam A, Cohen DJ, van Es GA, Steg PG, Morel MA, Mauri L, Vranckx P, McFadden E, Lansky A, Hamon M, Krucoff MW, Serruys PW; Academic Research Consortium. Clinical end points in coronary stent trials: a case for standardized definitions. Circulation 2007;115:2344–2351. [DOI] [PubMed] [Google Scholar]
- 8. Barrett JC, Fry B, Maller J, Daly MJ.. Haploview: analysis and visualization of LD and haplotype maps. Bioinformatics 2005;21:263–265. [DOI] [PubMed] [Google Scholar]
- 9. Lewis JP, Stephens SH, Horenstein RB, O'Connell JR, Ryan K, Peer CJ, Figg WD, Spencer SD, Pacanowski MA, Mitchell BD, Shuldiner AR.. The CYP2C19*17 variant is not independently associated with clopidogrel response. J Thromb Haemost 2013;11:1640–1646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Hulot JS, Bura A, Villard E, Azizi M, Remones V, Goyenvalle C, Aiach M, Lechat P, Gaussem P.. Cytochrome P450 2C19 loss-of-function polymorphism is a major determinant of clopidogrel responsiveness in healthy subjects. Blood 2006;108:2244–2247. [DOI] [PubMed] [Google Scholar]
- 11. Backman JD, O’Connell JR, Tanner K, Peer CJ, Figg WD, Spencer SD, Mitchell BD, Shuldiner AR, Yerges-Armstrong LM, Horenstein RB, Lewis JP.. Genome-wide analysis of clopidogrel active metabolite levels identifies novel variants that influence antiplatelet response. Pharmacogenet Genomics 2017;27:159–163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Zhu HJ, Wang X, Gawronski BE, Brinda BJ, Angiolillo DJ, Markowitz JS.. Carboxylesterase 1 as a determinant of clopidogrel metabolism and activation. J Pharmacol Exp Ther 2013;344:665–672. [DOI] [PubMed] [Google Scholar]
- 13. Lins R, Broekhuysen J, Necciari J, Deroubaix X.. Pharmacokinetic profile of 14C-labeled clopidogrel. Semin Thromb Hemost 1999;25 Suppl 2:29–33. [PubMed] [Google Scholar]
- 14. Jiang XL, Samant S, Lewis JP, Horenstein RB, Shuldiner AR, Yerges-Armstrong LM, Peletier LA, Lesko LJ, Schmidt S.. Development of a physiology-directed population pharmacokinetic and pharmacodynamic model for characterizing the impact of genetic and demographic factors on clopidogrel response in healthy adults. Eur J Pharm Sci 2016;82:64–78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Lewis JP, Horenstein RB, Ryan K, O’Connell JR, Gibson Q, Mitchell BD, Tanner K, Chai S, Bliden KP, Tantry US, Peer CJ, Figg WD, Spencer SD, Pacanowski MA, Gurbel PA, Shuldiner AR.. The functional G143E variant of carboxylesterase 1 is associated with increased clopidogrel active metabolite levels and greater clopidogrel response. Pharmacogenet Genomics 2013;23:1–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Sim SC, Risinger C, Dahl ML, Aklillu E, Christensen M, Bertilsson L, Ingelman-Sundberg M.. A common novel CYP2C19 gene variant causes ultrarapid drug metabolism relevant for the drug response to proton pump inhibitors and antidepressants. Clin Pharmacol Ther 2006;79:103–113. [DOI] [PubMed] [Google Scholar]
- 17. Scott SA, Sangkuhl K, Stein CM, Hulot JS, Mega JL, Roden DM, Klein TE, Sabatine MS, Johnson JA, Shuldiner AR; Clinical Pharmacogenetics Implementation Consortium. Clinical Pharmacogenetics Implementation Consortium guidelines for CYP2C19 genotype and clopidogrel therapy: 2013 update. Clin Pharmacol Ther 2013;94:317–323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Kassimis G, Davlouros P, Xanthopoulou I, Stavrou EF, Athanassiadou A, Alexopoulos D.. CYP2C19*2 and other genetic variants affecting platelet response to clopidogrel in patients undergoing percutaneous coronary intervention. Thromb Res 2012;129:441–446. [DOI] [PubMed] [Google Scholar]
- 19. Park JJ, Park KW, Kang J, Jeon KH, Kang SH, Ahn HS, Han JK, Koh JS, Lee SE, Yang HM, Lee HY, Kang HJ, Koo BK, Oh BH, Park YB, Kim HS.. Genetic determinants of clopidogrel responsiveness in Koreans treated with drug-eluting stents. Int J Cardiol 2013;163:79–86. [DOI] [PubMed] [Google Scholar]
- 20. Nanda N, Bao M, Lin H, Clauser K, Komuves L, Quertermous T, Conley PB, Phillips DR, Hart MJ.. Platelet endothelial aggregation receptor 1 (PEAR1), a novel epidermal growth factor repeat-containing transmembrane receptor, participates in platelet contact-induced activation. J Biol Chem 2005;280:24680–24689. [DOI] [PubMed] [Google Scholar]
- 21. Izzi B, Pistoni M, Cludts K, Akkor P, Lambrechts D, Verfaillie C, Verhamme P, Freson K, Hoylaerts MF.. Allele-specific DNA methylation reinforces PEAR1 enhancer activity. Blood 2016;128:1003–1012. [DOI] [PubMed] [Google Scholar]
- 22. Johnson AD, Yanek LR, Chen MH, Faraday N, Larson MG, Tofler G, Lin SJ, Kraja AT, Province MA, Yang Q, Becker DM, O'Donnell CJ, Becker LC.. Genome-wide meta-analyses identifies seven loci associated with platelet aggregation in response to agonists. Nat Genet 2010;42:608–613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Faraday N, Yanek LR, Yang XP, Mathias R, Herrera-Galeano JE, Suktitipat B, Qayyum R, Johnson AD, Chen MH, Tofler GH, Ruczinski I, Friedman AD, Gylfason A, Thorsteinsdottir U, Bray PF, O'Donnell CJ, Becker DM, Becker LC.. Identification of a specific intronic PEAR1 gene variant associated with greater platelet aggregability and protein expression. Blood 2011;118:3367–3375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Li M, Hu Y, Wen Z, Li H, Hu X, Zhang Y, Zhang Z, Xiao J, Tang J, Chen X.. Association of PEAR1 rs12041331 polymorphism and pharmacodynamics of ticagrelor in healthy Chinese volunteers. Xenobiotica 2017;1–9. [DOI] [PubMed] [Google Scholar]
- 25. Lewis JP, Ryan K, O’Connell JR, Horenstein RB, Damcott CM, Gibson Q, Pollin TI, Mitchell BD, Beitelshees AL, Pakzy R, Tanner K, Parsa A, Tantry US, Bliden KP, Post WS, Faraday N, Herzog W, Gong Y, Pepine CJ, Johnson JA, Gurbel PA, Shuldiner AR.. Genetic variation in PEAR1 is associated with platelet aggregation and cardiovascular outcomes. Circ Cardiovasc Genet 2013;6:184–192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Mega JL, Close SL, Wiviott SD, Shen L, Hockett RD, Brandt JT, Walker JR, Antman EM, Macias W, Braunwald E, Sabatine MS.. Cytochrome p-450 polymorphisms and response to clopidogrel. N Engl J Med 2009;360:354–362. [DOI] [PubMed] [Google Scholar]
- 27. Simon T, Verstuyft C, Mary-Krause M, Quteineh L, Drouet E, Meneveau N, Steg PG, Ferrieres J, Danchin N, Becquemont L; French Registry of Acute ST-Elevation and Non-ST-Elevation Myocardial Infarction (FAST-MI) Investigators. Genetic determinants of response to clopidogrel and cardiovascular events. N Engl J Med 2009;360:363–375. [DOI] [PubMed] [Google Scholar]
- 28. Wallentin L, James S, Storey RF, Armstrong M, Barratt BJ, Horrow J, Husted S, Katus H, Steg PG, Shah SH, Becker RC; PLATO investigators. Effect of CYP2C19 and ABCB1 single nucleotide polymorphisms on outcomes of treatment with ticagrelor versus clopidogrel for acute coronary syndromes: a genetic substudy of the PLATO trial. Lancet 2010;376:1320–1328. [DOI] [PubMed] [Google Scholar]
- 29. Cavallari LH, Lee CR, Beitelshees AL, Cooper-DeHoff RM, Duarte JD, Voora D, Kimmel SE, McDonough CW, Gong Y, Dave CV, Pratt VM, Alestock TD, Anderson RD, Alsip J, Ardati AK, Brott BC, Brown L, Chumnumwat S, Clare-Salzler MJ, Coons JC, Denny JC, Dillon C, Elsey AR, Hamadeh IS, Harada S, Hillegass WB, Hines L, Horenstein RB, Howell LA, Jeng LJB, Kelemen MD, Lee YM, Magvanjav O, Montasser M, Nelson DR, Nutescu EA, Nwaba DC, Pakyz RE, Palmer K, Peterson JF, Pollin TI, Quinn AH, Robinson SW, Schub J, Skaar TC, Smith DM, Sriramoju VB, Starostik P, Stys TP, Stevenson JM, Varunok N, Vesely MR, Wake DT, Weck KE, Weitzel KW, Wilke RA, Willig J, Zhao RY, Kreutz RP, Stouffer GA, Empey PE, Limdi NA, Shuldiner AR, Winterstein AG, Johnson JA; IGNITE Network. Multisite investigation of outcomes with implementation of CYP2C19 genotype-guided antiplatelet therapy after percutaneous coronary intervention. JACC Cardiovasc Interv 2018;11:181–191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Johnson JA, Roden DM, Lesko LJ, Ashley E, Klein TE, Shuldiner AR.. Clopidogrel: a case for indication-specific pharmacogenetics. Clin Pharmacol Ther 2012;91:774–776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Siller-Matula JM, Trenk D, Schror K, Gawaz M, Kristensen SD, Storey RF, Huber K; European Platelet Academy. How to improve the concept of individualised antiplatelet therapy with P2Y12 receptor inhibitors—is an algorithm the answer? Thromb Haemost 2015;113:37–52. [DOI] [PubMed] [Google Scholar]
- 32. Reny JL, Fontana P, Hochholzer W, Neumann FJ, Ten Berg J, Janssen PW, Geisler T, Gawaz M, Marcucci R, Gori AM, Cuisset T, Alessi MC, Berdague P, Gurbel PA, Yong G, Angiolillo DJ, Aradi D, Beigel R, Campo G, Combescure C.. Vascular risk levels affect the predictive value of platelet reactivity for the occurrence of MACE in patients on clopidogrel. Systematic review and meta-analysis of individual patient data. Thromb Haemost 2016;115:844–855. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.