Abstract
Multicellularity is defined as the developmental process by which unicellular organisms became pluricellular during the evolution of complex organisms on Earth. This process requires the convergence of genetic, ecological, and environmental factors. In fungi, mycelial and pseudomycelium growth, snowflake phenotype (where daughter cells remain attached to their stem cells after mitosis), and fruiting bodies have been described as models of multicellular structures. Ustilaginomycetes are Basidiomycota fungi, many of which are pathogens of economically important plant species. These fungi usually grow unicellularly as yeasts (sporidia), but also as simple multicellular forms, such as pseudomycelium, multicellular clusters, or mycelium during plant infection and under different environmental conditions: Nitrogen starvation, nutrient starvation, acid culture media, or with fatty acids as a carbon source. Even under specific conditions, Ustilago maydis can form basidiocarps or fruiting bodies that are complex multicellular structures. These fungi conserve an important set of genes and molecular mechanisms involved in their multicellular growth. In this review, we will discuss in-depth the signaling pathways, epigenetic regulation, required polyamines, cell wall synthesis/degradation, polarized cell growth, and other cellular-genetic processes involved in the different types of Ustilaginomycetes multicellular growth. Finally, considering their short life cycle, easy handling in the laboratory and great morphological plasticity, Ustilaginomycetes can be considered as model organisms for studying fungal multicellularity.
Keywords: Ustilaginomycetes, smut fungi, multicellularity, filamentous growth, mycelial growth
1. Introduction
Multicellularity is considered one of the major transitions in the evolution of complex organisms on Earth. It is defined as the developmental process by which unicellular organisms became pluricellular early and repeatedly during the evolution of life, involving genetic, ecological, and environmental factors [1,2,3,4,5]. Multicellularity has different levels of complexity. In microorganisms, such as bacteria and fungi, simple multicellularity has been observed as a mechanism that increases the affinity for substrates and the obtainment of nutrients, defense against predators, tolerance to environmental stress, host colonization, and may ensure offspring [6,7,8,9,10,11,12,13]. Interestingly, two possible hypotheses have been proposed to explain the development of simple multicellularity: (i) Clonal multicellularity, where the cells are held together after mitosis and share the same genotype; and (ii) aggregative multicellularity, when unrelated cells come together to form a chimeric structure [5,14,15]. However, it has been observed that the multicellularity developed in filamentous fungi can be different from that of the previously described fungi. This is because their vegetative mycelium rarely adheres to each other except in fruiting bodies—an example of complex multicellularity, which is defined as a three-dimensional structure with a differentiated organization in time and space and a genetically determined developmental program [16,17]. Comparative genomic studies have shown that both filamentous fungi and yeasts conserve the genetic machinery for multicellular growth [16,18]. Fungal mycelium has been described as a model of a simple multicellular structure, showing a polarized syncytium that expands via tip elongation and somatic cell-cell union [19,20]. Likewise, the snowflake phenotype developed by yeasts and some fungi is considered a model of undifferentiated multicellularity [10,11,13,16].
Some fungi show multicellular growth under specific environmental conditions or development stages, e.g., Neurospora crassa filamentous growth, Candida albicans and Yarrowia lipolytica mycelial growth, Saccharomyces cerevisiae pseudomycelium and multicellular clusters, Coprinus cinereus fruiting bodies, inter alia [20,21,22,23,24,25].
Ustilaginomycetes are a group of approximately 1,185 of Basidiomycetes species, many of which are pathogens of economically important plants [26]. Ustilago maydis is probably the most studied species. Like other Ustilaginomycetes, U. maydis can grow unicellularly as saprophytic yeast-like cells (sporidia), or multicellularly as mycelium during plant infection and colonization. On the maize plant surface, sexually compatible sporidia mate to form a dikaryotic filament that penetrates the plant by forming appressorium. Mating is regulated by the a and b independent alleles. The a allele encodes a pheromone-receptor system and the b allele a bE/bW heterodimer transcription factor, which regulate the development of the infective dikaryotic filament [27,28,29,30,31,32]. In some of these fungi, simple multicellularity has also been observed during their growth under different environmental conditions, such as nitrogen or nutrient starvation, acid culture media, or using fatty acids as the carbon source [33,34,35,36,37]. For example, Sporisorium reilianum shows several types of multicellular growth: (i) As mycelium during the plant infection or under nutrient starvation conditions [29,31,33,38]; (ii) as pseudomycelium by deleting certain genes [39]; or (iii) as multicellular clusters in acidic culture medium [13]. In U. maydis, basidiocarp formation has also been described [40,41].
There are several mechanisms described as important for multicellular growth in Ustilaginomycetes, for example, the Mitogen-Activated Protein Kinase (MAPK) and Protein Kinase A (PKA) signaling pathways [32,42,43,44,45]; the requirement of polyamines [46,47]; epigenetic regulation [48,49,50,51]; and cell wall synthesis and degradation (a cellular structure directly involved in fungal multicellularity) [52,53,54].
Considering that the Ustilaginomycetes can grow unicellularly and multicellularly, and the importance of their morphology during development, in this review we will discuss the signaling pathways, the epigenetic regulation, the polyamine requirements, the polarized cell growth, and the synthesis or degradation of the cell wall during the multicellular growth of this class of fungi, highlighting the experimental advantages of the Ustilaginomycetes as model organisms for studying multicellularity.
2. Signaling Pathways Involved in the Ustilaginomycetes Multicellular Growth
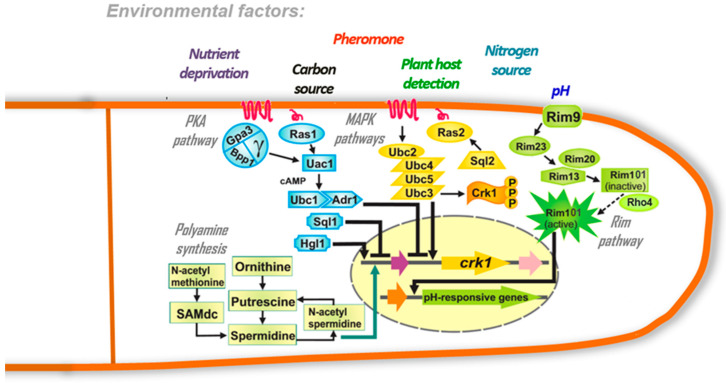
Fungi, like all living organisms, grow and develop under fluctuating environmental conditions. Therefore, they have developed mechanisms to sense, respond, and adapt to conditions that favor their growth, or to deal with adverse conditions (Figure 1).
Figure 1.
Schematic representation of the signal transduction and metabolic pathways involved in the Ustilaginomycetes multicellular growth. Several authors have suggested that the mitogen-activated protein kinase (MAPK) and cyclic AMP (cAMP)/protein kinase A (PKA) pathways are necessary for mycelium development (a simple multicellular structure observed in Ustilaginomycetes) (reviewed by References [32,45,55,56]). Polyamines, especially spermidine, are essential for the expression of the genes involved in the multicellular growth and the response to stress conditions [47,57,58,59]. Although the Rim pathway is not involved in the fungal multicellular growth, it is required to sense stressful environmental conditions such as pH [60,61,62], which is one of the most important inducers of multicellular growth in Ustilaginomycetes [13,35].
Multicellular growth is induced in Ustilaginomycetes: During plant infection and colonization, due to nitrogen or nutrient starvation, in acidic culture media and using fatty acids as carbon sources. U. maydis, for example, can grow multicellularly as basidiocarp in the presence of maize embryogenic calli (Figure 2, Figure 3, and Figure 4B–G,J–L) [13,29,32,33,34,35,36,40].
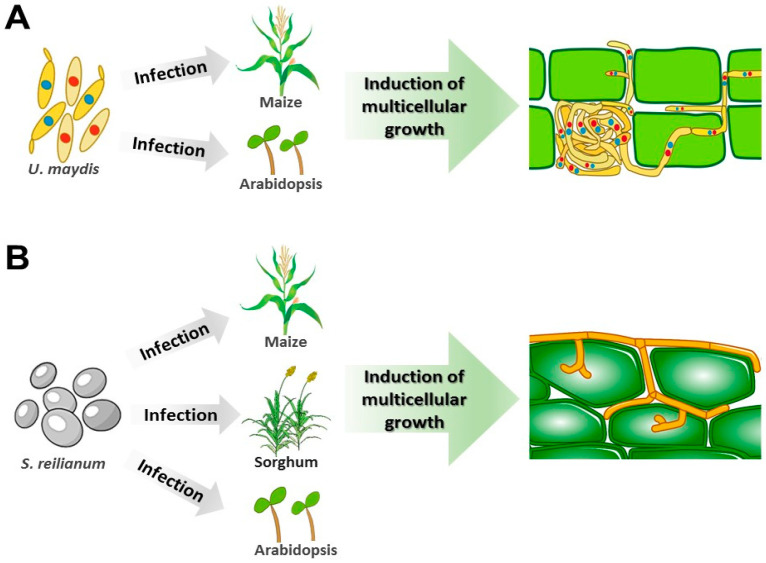
Figure 2.
Multicellular growth of Ustilaginomycetes is induced during infection and colonization of the host plants. (A) Multicellular growth of Ustilago maydis as a filament is induced during the maize and Arabidopsis experimental host infection [27,63,64]. The different colors in sporidia nuclei represent different yeast-like cells that are sexually compatible. (B) Similarly, Sporisorium reilianum multicellular growth is induced during maize, sorghum, and Arabidopsis infection [29,31,38].
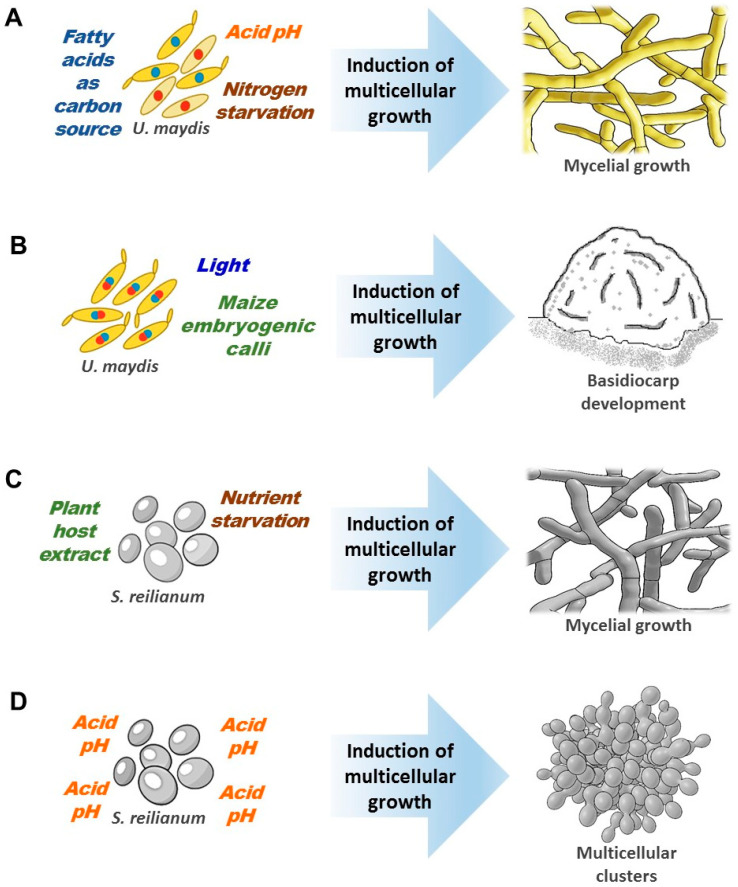
Figure 3.
The environmental conditions induce the multicellular growth of Ustilaginomycetes. (A) Nitrogen starvation [34], acid culture media [35], and fatty acids as a carbon source [36] induce multicellular growth as mycelium in Ustilago maydis. (B) U. maydis grows multicellularly as gastroid-type basidiocarps when it is cultivated in dual cultures with maize embryogenic calli [40]. (C) Host plant extracts or nutrient starvation induce muticellular growth as mycelium in S. reilianum [33]. (D) Acid pH in culture medium induces multicellular cluster growth in S. reilianum [13].
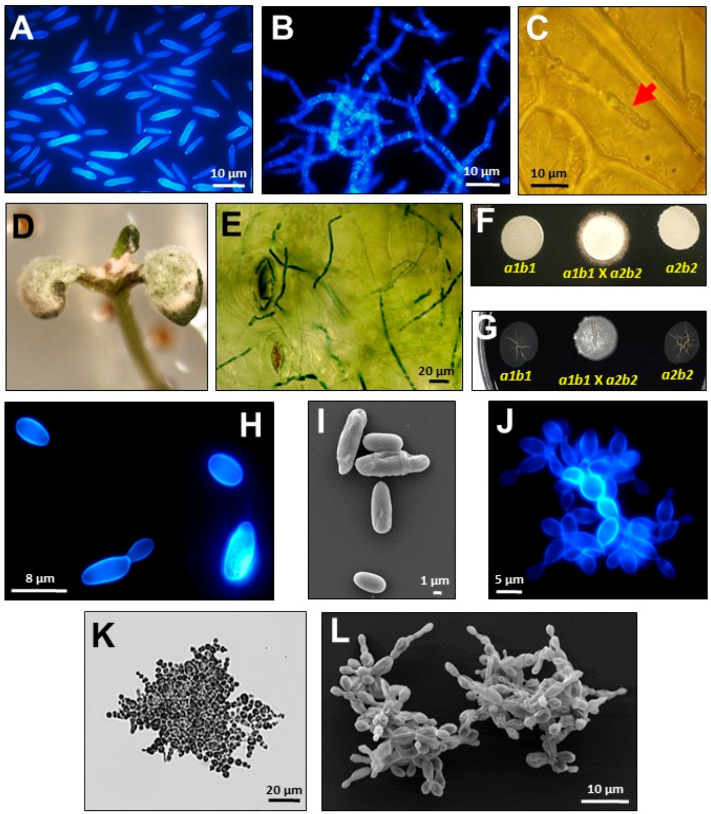
Figure 4.
Multicellular shapes developed by Ustilaginomycetes under different growth or development conditions. (A–F), Images of U. maydis. (G–L) Images of S. reilianum. (A,B) Epifluorescence microphotographs of the fungus showing yeast-like unicellular growth, or multicellular growth as mycelium, in minimal medium pH 7 or pH 3, respectively. (C) Multicellular growth of the fungus as mycelium (red arrow) during the colonization of maize plant cells. (D,E) Multicellular growth of the fungus during Arabidopsis infection. (F,G) Multicellular growth of fungi as white-fuzzy colonies after mating of sexually compatible sporidia on plates with charcoal containing minimal medium. (H,I) Epifluorescence and scanning electron microphotographs of the fungus growing unicellularly like yeasts. (J–L) Epifluorescence, bright field, and scanning electron microphotographs of the multicellular clusters of the fungus. In all the epifluorescence microphotographs, the fungi were stained with calcofluor white (Sigma-aldrich, 18909). In (E,K), the fungi were stained with cotton blue-lactophenol (Sigma-Aldrich, 61335, St. Louis, MO, USA).
Although pH is one of the environmental factors that induce multicellular growth in some Ustilaginomycetes (Figure 3A,D and Figure 4B,J–L), the Pal/Rim pathway, the main mechanism fungi use to sense pH (Figure 1) [61,65], is not involved in U. maydis multicellular growth [60,61,62], and may not be involved in other Ustilaginomycetes growth either (the Pal/Rim pathway is conserved in smut fungi, see Table 1). We will discuss the MAPK and PKA signal transduction pathways involved in Ustilaginomycetes multicellular growth in response to the different conditions or factors previously mentioned.
Table 1.
Signal transduction pathways related to the multicellular growth of Ustilaginomycetes.
| Pathway | Gene | Ustilago maydis a | Ustilago hordei a,b | Ustilago bromivora c | Sporisorium reilianum a,b | Sporisorium graminicola a | Testicularia cyperi a,b,d |
|---|---|---|---|---|---|---|---|
| PKA | RAL2 | UMAG_00884 | UHOR_01334 | UBRO_01334 | sr12177 | EX895_006321 | BCV70DRAFT_173605 |
| GPA3 | UMAG_04474 | UHOR_06981 | UBRO_06981 | sr15360 | EX895_002426 | CE102777_5455 | |
| BPP1 | UMAG_00703 | UHOR_01085 | UBRO_01086 | sr11991 | EX895_000934 | CE92962_8479 | |
| PKA | UMAG_06450 | UHOR_09003 | UBRO_09003 | sr16888 | EX895_004218 | CE31316_43035 | |
| UAC1 | UMAG_05232 | UHOR_03218 | UBRO_03218 | sr13141 | EX895_004629 | BCV70DRAFT_200755 | |
| UBC1 | UMAG_00525 | UHOR_00805 | UBRO_00805 | sr10199.2 | EX895_000753 | BCV70DRAFT_164254 | |
| ADR1 | UMAG_04456 | UHOR_06957 | UBRO_06957 | sr15343 | EX895_002409 | BCV70DRAFT_103006 | |
| SQL1 | UMAG_05501 | UHOR_07888 | UBRO_07888 | sr16182 | EX895_002861 | BCV70DRAFT_212144 | |
| HGL1 | UMAG_11450 | UHOR_00981 | UBRO_00981 | sr11921 | EX895_000866 | BCV70DRAFT_202631 | |
| CRK1 | UMAG_11410 | UHOR_04041 | UBRO_04041 | sr10962 | EX895_005166 | BCV70DRAFT_157158 | |
| MAPK | RAS2 | UMAG_01643 | UHOR_01498 | UBRO_02437 | sr12711 | EX895_004437 | CE98444_23931 |
| SQL2 | UMAG_10803 | UHOR_02247 | UBRO_02247 | sr12585 | EX895_004314 | BCV70DRAFT_231562 | |
| SQL2 | UMAG_11476 | UHOR_04466 | UBRO_04466 | sr13877 | EX895_001846 | BCV70DRAFT_223969 | |
| UBC2 | UMAG_05261 | UHOR_08070 | UBRO_08070 | sr16309 | EX895_003017 | BCV70DRAFT_197656 | |
| MAPKKK | UMAG_04258 | UHOR_06391 | UBRO_06391 | sr15150 | EX895_002006 | BCV70DRAFT_154027 | |
| MAPKK | UMAG_01514 | UHOR_02245 | UBRO_02245 | sr12583 | EX895_004312 | BCV70DRAFT_211523 | |
| MAPKK | UMAG_11453 | UHOR_01494 | UBRO_00992 | sr11928 | EX895_000873 | CE125301_8671 | |
| MAPKK | UMAG_00721 | UHOR_01105 | UBRO_01105 | sr12009 | EX895_000953 | BCV70DRAFT_71012 | |
| MAPK | UMAG_03305 | UHOR_05144 | UBRO_05144 | sr14305 | EX895_005556 | CE107062_12366 | |
| MAPK | UMAG_02331 | UHOR_05144 | UBRO_05144 | sr14305 | EX895_005556 | BCV70DRAFT_107063 | |
| CRK1 | UMAG_11410 | UHOR_04041 | UBRO_04041 | sr10962 | EX895_005166 | BCV70DRAFT_157158 | |
| RIM | RIM101 | UMAG_10426 | UHOR_06410 | UBRO_06410 | sr15166 | EX895_002022 | CE711_11906 |
| RIM9 | UMAG_00581 | UHOR_00899 | UBRO_00899 | sr11859 | EX895_000793 | CE73867_14878 | |
| RIM20 | UMAG_11510 | UHOR_05885 | UBRO_05885 | sr14775 | EX895_001317 | BCV70DRAFT_202631 | |
| RIM13 | UMAG_02075 | UHOR_03447 | UBRO_03447 | sr13297 | EX895_003420 | BCV70DRAFT_198765 | |
| RIM23 | UMAG_04392 | UHOR_06863 | UBRO_06863 | sr15280 | EX895_002350 | BCV70DRAFT_161602 | |
| RHO4 | UMAG_10663 | UHOR_01182 | UBRO_05736 | sr14670 | EX895_001201 | CE23374_4310 |
ID genes according to NCBI (a), ExPASy (b), e!EnsemblFungi (c), and JGI MycoCosm (d), and considering the genomic data published for U. maydis [66], U. hordei [67], U. bromivora [68], S. reilianum [69], S. graminícola [70], and T. cyperi [71]; the six Ustilaginomycetes with the best-annotated genomes. Genes identified based on their homology with U. maydis. The sequences analyzed were deposited in https://github.com/lucilaortiz/Multicellularity_associated_proteins.
2.1. MAPK Pathways
In eukaryotic organisms, including Ustilaginomycetes, the MAPK signaling pathway is one of the most conserved and important mechanisms for the transduction of information from the exterior to the interior of the cell (Table 1). In the MAPK pathway, stimuli are perceived by receptors and proteins anchored in the cell membrane. The receptors can be G-protein-coupled to transmembrane receptors, receptor tyrosine kinases (RTKs) or two-component signal transduction systems (TCS). When the receptors perceive the stimuli, a sequential activation is induced by phosphorylation of serine/threonine residues in the three protein kinases: MAP kinase kinase kinase (MAPKKK), MAP kinase kinase (MAPKK), and MAP kinase (MAPK) (Figure 1). The MAP kinase activates the transcription factors that translocate to the nucleus to regulate the expression of the genes in response to the external stimuli [45,72,73].
In fungi, the MAPK signaling pathway regulates physiological processes, such as the cell cycle, multicellularity, sporulation, mating, cell wall synthesis and integrity, response to stress, and virulence [45,74,75]. The Saccharomyces cerevisiae and Candida albicans MAPK signaling pathways have been extensively studied, so there is more information available. The S. cerevisiae Kss1 MAPK signaling pathway is necessary for multicellular pseudomycelium growth. The C. albicans homologous pathway is known as Cek1, and U. maydis homologous pathway is known as PMM (pathogenesis, mating, multicellularity, or morphogenesis). In U. maydis, the MAPK PMM pathway controls the multicellular growth in vitro [induced by different stress conditions (Figure 3A,B and Figure 4B)], and in vivo [during host plant infection, (Figure 2A and Figure 4C–E)] [32,45]. The deletion of C. albicans and U. maydis MAPK pathway components suppresses the multicellular formation of pseudomycelium and mycelium, respectively [22,32,44,76,77,78,79,80].
In U. maydis, the proteins of the MAPK core are known as: Ubc4/Kpp4 MAPKKK, Fuz7/Ubc5 MAPKK and Ubc3/Kpp2 MAPK (Figure 1). They are important for mating, pathogenicity, and the formation of multicellular mycelium. They show homology with the protein kinases STE11, STE7 and KSS1 of the Saccharomyces Kss1 MAPK pathway [81,82,83,84,85,86].
In the Ustilago esculenta and Sporisorium scitamineum Ustilaginomycetes, the protein kinase Kpp2 has the same function as its ortholog in U. maydis [87,88]. Phylogenetic analysis showed that the Kpp2 sequence is highly conserved among smut fungi [87]. In U. esculenta, the UeKPP2 gene is up-regulated in multicellular filamentous growth, and UeKpp2 interacts with Uefuz7 and UePrf1, the same as in U. maydis [88]. Interestingly, ΔSskpp2 showed a decrease in mating and filamentation, which was partially restored by adding tryptophol, a quorum-sensing (QS) aromatic alcohol secreted by yeasts that stimulates S. cerevisiae and C. albicans mycelium or pseudomycelium growth, respectively. In S. cerevisiae, production of these aromatic alcohols is regulated by extracellular nitrogen and cell density. For example, high concentrations of nitrogen sources repress its synthesis, while low concentrations activate it. However, no multicellularity regulation by QS has been observed in U. maydis or other Ustilaginomycetes [87,89,90].
In Ustilaginomycetes, upstream of the MAPKs PMM core is the Ubc2 adaptor protein (Figure 1), an orthologue of Ste50 from S. cerevisiae. Ubc2 contains the domains: SAM (sterile α motify), RA (Ras association), and in two SH3 C-terminals (Src homology 3). All of them are important for protein-protein interaction. For example, through the SAM domain, Ubc2 interacts with ubc4/kpp4 (MAPKKK) protein kinase [79,91]. UBC2 is necessary for U. maydis multicellular mycelial growth. The ∆ubc2 strains that were affected in the MAPK PMM pathway showed a severe reduction in the formation of dikaryotic filamentous and haploid mycelium, which was induced under in vitro conditions [44,91]. Interestingly, the transcriptomic analysis of Δubc2 showed that 939 genes [≈ 14.0 % of the U. maydis genome (6883 genes)] are directly or indirectly regulated by the MAPK PMM pathway [80]. Among the genes natively regulated by the inactivation of MAPK PMM, there were genes encoding proteins involved in membrane synthesis or cell wall synthesis [chitin synthases, chitin deacetylases, Kre6 (glucan synthase), Rot1 (involved in cell wall function), chitin-binding proteins, exo-1,3-beta-glucanase precursors]; genes encoding proteins involved in multicellular growth (actin cytoskeleton organization, myosins, kinesins); genes encoding protein kinases and serine/threonine kinases, with CRK1 between them; and genes encoding the GTPases, such as RAS and SQL2 [80].
The Ras proteins are small monomeric GTPases that act as molecular switches in the signaling pathways that alternate between the GTP and GDP-bound forms in response to extracellular stimuli. In U. maydis, the SQL2 gene encoding a CDC25-like protein is involved in the activation of Ras2 [92]. The constitutive activation of RAS2 or the overexpression of SQL2 promotes the filamentous multicellular growth of this fungus, even in haploid strains [92,93]. S. cerevisiae Ras2 acts in the same way in the MAPK pathway during pseudomycelial growth [94].
The MAPK pathway is conserved in Ustilaginomycetes (Table 1). As occurs in U. maydis, the MAPK PMM pathway may interact antagonistically with the cAMP/PKA pathway to regulate unicellular (Figure 4A,H,I) or multicellular growth (Figure 4B–G,J–L) [32,45,95] in other Ustilaginomycetes. MAPK PMM is required for multicellular growth as mycelium, and PKA for unicellular growth as yeast. The mutant strains in the MAPK cascade genes, e.g., UBC2, UBC3, UBC4, and UBC5, showed a constitutive yeast growth [81,83,84,85,91]. The PKA and MAPK signal transduction pathways converge with Crk1, a Ser/Thr kinase protein. The Δcrk1 mutant strains, unlike the wild type strains, grow constitutively as unicellular yeasts on acidic culture medium and under nutrient starvation conditions [95]. However, the CRK1 gene is also necessary for the filamentous growth induced by defects in the cAMP/PKA pathway [95]. Likewise, in S. scitamineum, the MAPK and PKA pathways show an antagonistic interaction during multicellular or unicellular growth regulation. The ΔSskpp2 mutant strains show a reduction in filamentation, which is partially restored by adding cAMP [87].
As mentioned above, U. maydis can form large hemi-spheroidal structures with gastroid-type basidiocarp characteristics (a complex multicellular structure) when grown in solid medium supplemented with auxins in dual cultures with maize embryogenic calli (Figure 3B) [40]. In this fungus, the MAPK PMM pathway is involved in the formation of basidiocarps. The ΔFuz7 mutant strains did not develop basidiocaps, and several genes encoding serine/threonine kinases were up-regulated during the early stages of basidiocarp formation, e.g., Kpp2 and Kpp6 were 2.2 and 11.4 times overexpressed, respectively. Similarly, this occurred during the development of Coprinopsis cinérea fruiting body, when the MAPK pathway genes are up-regulated [96]. Finally, during the development of multicellular clusters of Sporisorium reilianum (Figure 3D and Figure 4J–L), the important role of several MAPK proteins and serine/threonine kinases was suggested by transcriptional network analyses [13].
2.2. cAMP/PKA Pathway
In eukaryotic cells, the secondary messenger, cyclic adenosine monophosphate (cAMP) is produced in response to extracellular stimuli. When the receptors perceive a stimulus, a dissociation of the α-subunit of the G-protein is induced in order to activate or repress the cAMP synthesis by the adenylate cyclase enzyme (Uac) (Figure 1). When the levels of cAMP are low, the PKA is an inactive tetramer compound of two catalytic and two regulatory subunits. However, when the levels are high, cAMP binds to a regulatory subunit causing a holoenzyme dissociation in a dimeric regulatory subunit and two active monomeric catalytic subunits which phosphorylate transcription factors and metabolic enzymes [97,98,99].
In fungi, the cAMP/PKA signaling pathway has a function in mating, sporulation, dimorphism, response to stress, and virulence [77,100,101]. In S. cerevisiae and C. albicans, both cAMP/PKA and MAPK pathways participate in the shift from unicellular growth as yeast to multicellular growth in the form pseudomycelium or mycelium, respectively [22,77,78,102]. In contrast, and as described above, cAMP/PKA and MAPK PMM are antagonistic in U. maydis [32,45,55]. In this fungus, the inactivation of the cAMP/PKA pathway by deleting the genes that encode adenylate cyclase (Uac1) or a regulatory subunit of PKA (Adr1) showed a constitutive phenotype of multicellular haploid mycelium. In addition, the unicellular budding growth was restored when exogenous cAMP was added to the culture medium [42,43]. The same phenotype was observed in Δgpa3 and Δbpp1 mutants, that were affected in the α- and β- heterotrimeric GTPase subunits, respectively. The cAMP/PKA pathway in Ustilago hordei acts in the same way as in U. maydis. The deletion in the heterotrimeric GTPase α-subunit (Δfil1) produced constitutive mycelial growth in a solid and liquid medium, and the addition of exogenous cAMP restored the budding growth [103].
Although the cAMP/PKA signaling pathway is highly conserved in Ustilaginomycetes (Table 1), in S. scitamineum, and contrary to U. maydis and U. hordei, the cAMP/PKA pathway is involved in multicellular growth as mycelium. The deletion of different pathway components (Δssgpa3, Δssuac1 and Δssadr1) suppresses the formation of dikaryotic mycelium. The production of cAMP in Δssauc1 was blocked, and severely reduced in ∆ssgpa3 and Δssadr1. The mycelial growth was restored in Δssgpa3 and Δssuac1 by adding exogenous cAMP, but not in Δssadr1. This makes sense because SsGpa3 and SsUac1 act upstream of cAMP production [104]. Moreover, in this smut fungus, the AGC protein kinases (SsAgc1) regulate the signaling pathway involved in mating and dikaryotic filamentous growth; therefore, this protein is crucial for the expression of genes involved in multicellular growth of S. scitamineum [105].
Other cAMP/PKA pathway components and their functions during U. maydis unicellular-multicellular growth have been analyzed. An example of this is the HGL1 gene (hyphal growth locus), which encodes a regulator of the unicellular and multicellular morphologies and is important for mating and teliospore formation during the fungus’ pathogenic process [106]. Moreover, the SQL1 gene encoding a repressor of the U. maydis cAMP/PKA pathway is itself a repressor of unicellular fungus growth [107].
In the early developmental stages of U. maydis basidiocarps (a complex multicellular structure), the gene encoding the cAMP-dependent protein kinase catalytic subunit was 2.3 times overexpressed [41]. In this regard, in C. cinerea, Schizophyllum commune and Volvariella volvacea, the cAMP content is essential for the formation of the fruiting body [108,109,110,111], and the genes of cAMP/PKA signaling pathways are up-regulated during the development of the C. cinerea fruiting body [96]. Finally, the participation of the kinase A protein and cAMP-dependent protein kinase in the formation of S. reilianum multicellular clusters was predicted by transcriptional network analyses [13].
3. The Role of Polyamines in the Multicellular Growth of Ustilaginomycetes
Polyamines are aliphatic polycations that are present in all living organisms. They are essential for development and growth [112]. The polyamines modulate the enzymatic activities, gene expression, DNA-protein interactions, and protect DNA from enzymic degradation [113,114]. In fungi, they regulate spore germination, appressorium formation, conidiation and multicellular growth in the form of mycelium [46,57,115,116,117,118,119]. The common polyamines in some fungi are putrescine, spermidine, and spermine. However, Ustilaginomycetes, such as U. maydis do not have spermine [59,120,121], and they conserve the same genes for polyamine synthesis (Table 2, Figure 1). This characteristic, and the fact that these fungi can grow as unicellular and multicellular forms, make them excellent model organisms for studying the metabolism and function of polyamines in fungi multicellularity.
Table 2.
Genes involved in the polyamine synthesis pathway of Ustilaginomycetes.
| Gene |
Ustilago
maydis a |
Ustilago
hordei a,b |
Ustilago
bromivora c |
Sporisorium
reilianum a,b |
Sporisorium
graminicola a |
Testicularia
cyperi a,b,d |
|
|---|---|---|---|---|---|---|---|
| Acetylornithine aminotransferase | OAT | UMAG_05671 | UHOR_07330 | UBRO_07330 | sr15996 | EX895_002715 | BCV70DRAFT_201257 |
| Ornithine decarboxylas | ODC | UMAG_01048 | UHOR_01580 | UBRO_01580 | sr12348 | EX895_006099 | BCV70DRAFT_212462 |
| Polyamine oxidase | PAO | UMAG_05850 | UHOR_08347 | UBRO_08347 | sr16473 | EX895_008244 | BCV70DRAFT_173429 |
| Chimeric spermidine synthase/saccharopine reductase | SPE | UMAG_05818 | UHOR_08297 | UBRO_08297 | sr16440 | EX895_003211 | BCV70DRAFT_199161 |
| S-adenosylmethionine decarboxylase | SAMDC | UMAG_10792 | UHOR_01520 | UBRO_01520 | sr12300 | EX895_006201 | BCV70DRAFT_205658 |
| Putative spermidine acetyltrasferase | SSAT | UMAG_00127 | UHOR_00200 | UBRO_00200 | sr11469 | EX895_000152 | BCV70DRAFT_169192 |
ID genes according to NCBI (a), ExPASy (b), e!EnsemblFungi (c), and JGI MycoCosm (d), and considering the genomic data published for U. maydis [66], U. hordei [67], U. bromivora [68], S. reilianum [69], S. graminícola [70], and T. cyperi [71]; the six Ustilaginomycetes with the best-annotated genomes. Genes identified based on their homology with U. maydis. The sequences analyzed were deposited in https://github.com/lucilaortiz/Multicellularity_associated_proteins.
Strains of U. maydis with ODC gene mutated, which encode ornithine decarboxylase (Odc) and is responsible for putrescine synthesis, are unable to grow as mycelia under acidic pH conditions with limiting concentrations of putrescine [46]. In basidiomycota fungi, such as smut fungi, the spermidine synthase (Spe) is encoded by a chimeric bifunctional gene that also encodes saccharopine dehydrogenase (Sdh), an enzyme involved in lysine synthesis [121]. The U. maydis Δspe-Δsdh mutant strains are auxotrophic for lysine and spermidine and require high concentrations of spermidine in order to grow multicellularly as mycelium under acidic conditions in culture medium [57]. Interestingly in Ustilaginomycetes, there is an alternative mechanism for putrescine synthesis, through the polyamine oxidase enzyme involved in the retroconversion of spermidine to putrescine. U. maydis double mutants (Δodc/Δpao) in the genes encoding Odc and Pao grow multicellularly as mycelium with the addition of spermidine only and in complete absence of putrescine [58]. This indicates that spermidine is the most important polyamine for multicellularity in Ustilaginomycetes [58,59,119,122].
4. Epigenetic Regulation of Multicellular Growth in Ustilaginomycetes
In fungi, as in all eukaryotic organisms, DNA is wrapped and highly compacted around histone proteins forming the nucleosomes within the nucleus. This organization can be altered by post-translational modifications of histones that relax or compact the nucleosomes: Acetylation by histone acetyltransferases (HATs), or deacetylation by histone deacetylases (HDACs), respectively. Nucleosome relaxation allows accessibility of DNA to different proteins involved in replication and gene transcription in response to environmental signals.
Although Ustilaginomycetes have homologous genes encoding HATs and HDACs (Table 3), the epigenetic regulation of multicellular growth has only been studied in U. maydis [48,49,50]. In this fungus, the GCN5 HAT was deleted. The mutant strains were slightly more sensitive to different stress conditions than the wild type, but they grew multicellularly as mycelium and fuzz-like colonies (constitutive mycelial growth) under all the growth conditions analyzed. Also, the virulence was dramatically reduced [48]. Through transcriptomic analysis of mutant and wild type strains, it was observed that in the mutant strain (Δgcn5) a total of 1203 were differentially regulated. Of these, 574 were repressed and 629 were overexpressed. Interestingy, 67 genes described as important for yeast growth (unicellularity, Figure 4A) and 66 described as important for mycelial growth (multicellularity, Figure 4B) [123], were down-regulated and up-regulated, respectively in the Δgcn5 mutant [49]. Among the differentially expressed genes, the following phenomena stand out: Genes involved in cell wall biogenesis and genes related with pathogenesis were down-regulated; genes required for mycelium development were overexpressed. For example, the REP1 gene that encodes a repellent protein was 33.2 times up-regulated in the mutant strain [49].
Table 3.
Genes described as related with the multicellular growth of Ustilaginomycetes.
| Gene | Ustilago maydis a | Ustilago hordei a,b | Ustilago bromivora c | Sporisorium reilianum a,b | Sporisorium graminícola a | Testicularia cyperi a,b,d |
|---|---|---|---|---|---|---|
| GCN5—Histone acetyltransferase | UMAG_05168 | UHOR_03120 | UBRO_03120 | sr13072 | EX895_004562 | BCV70DRAFT_198404 |
| Putative histone acetylase | UMAG_10190 | UHOR_03120 | UBRO_03120 | sr13072 | EX895_004562 | BCV70DRAFT_198404 |
| CLR3—Histone deacetylase | UMAG_02102 | UHOR_03487 | UBRO_03487 | sr13325 | EX895_004958 | BCV70DRAFT_166948 |
| HOS2—Histone deacetylase | UMAG_11828 | UHOR_01015 | UBRO_01015 | sr11943 | EX895_000889 | BCV70DRAFT_70918 |
| TEA4—SH3 domain protein | UMAG_01012 | UHOR_01534 | UBRO_01534 | sr12319 | EX895_006176 | BCV70DRAFT_159133 |
| TEA1—Kelch domain protein | UMAG_15019 | UHOR_01124 | UBRO_01124 | sr12022 | EX895_000966 | BCV70DRAFT_69247 |
| REP1—Repellent | UMAG_03924 | UHOR_05948 | UBRO_05948 | sr14829 | EX895_001361 | BCV70DRAFT_80316 |
| REP4—Repellent | UMAG_04517 | UHOR_06499 | UBRO_06499 | sr15402 | EX895_002086 | BCV70DRAFT_202324 |
| HUM2—Hydrophobin 2 | UMAG_11562 | UHOR_07158 | UBRO_07158 | sr15890 | EX895_002555 | BCV70DRAFT_207191 |
| HUM3—Hydrophobin 3 | UMAG_04433 | UHOR_06926 | UBRO_06926 | sr15320 | EX895_002383 | BCV70DRAFT_189664 |
| RRM4—RNA-binding protein | UMAG_03494 | UHOR_05377 | UBRO_05377 | sr14484 | EX895_006466 | BCV70DRAFT_213009 |
| Actin-binding protein | UMAG_05340 | UHOR_08160 | UBRO_08160 | sr16324 | EX895_003081 | BCV70DRAFT_155405 |
| Actin | UMAG_11232 | UHOR_08813 | UBRO_08813 | sr11345 | EX895_003701 | BCV70DRAFT_201455 |
| Actin-interactin gprotein; actin patch component | UMAG_05949 | UHOR_08467 | UBRO_08467 | sr16579 | EX895_005119 | BCV70DRAFT_213703 |
| Actin filament organization | UMAG_04613 | UHOR_06638 | UBRO_06638 | sr15500 | EX895_002183 | BCV70DRAFT_202237 |
| LIS1—Nuclear migration | UMAG_03164 | UHOR_04938 | UBRO_04938 | sr14206 | EX895_005820 | BCV70DRAFT_200264 |
| Actin regulating Ser/Thr kinase | UMAG_03081 | UHOR_04824 | UBRO_04824 | sr14140 | EX895_002483 | - |
| Formin; actin nucleation | UMAG_12254 | UHOR_06202 | UBRO_06202 | sr15020 | EX895_001591 | BCV70DRAFT_205747 |
| Formin; actin nucleation. Functionally redundant with Bni1 | UMAG_01141 | UHOR_01723 | UBRO_01723 | sr12440 | EX895_006065 | BCV70DRAFT_12160 |
| F-actin capping protein alpha subunit | UMAG_00423 | UHOR_00664 | UBRO_00664 | sr11788 | EX895_000652 | BCV70DRAFT_196835 |
| F-actin capping protein beta subunit | UMAG_11177 | UHOR_08001 | UBRO_08001 | sr16266 | EX895_002944 | BCV70DRAFT_201290 |
| Chaperonin role for actin and tubulin | UMAG_01279 | UHOR_01918 | UBRO_01918 | sr10401 | EX895_001741 | BCV70DRAFT_197510 |
| Chaperonin role for actin and tubulin | UMAG_06235 | UHOR_08845 | UBRO_08845 | sr16767 | EX895_003720 | BCV70DRAFT_199599 |
| Chaperonin role for actin and tubulin | UMAG_06067 | UHOR_08612 | UBRO_08612 | sr16694 | EX895_003550 | BCV70DRAFT_202419 |
| Chaperonin role for actin and tubulin | UMAG_02571 | UHOR_04113 | UBRO_04113 | sr17132 | EX895_005225 | BCV70DRAFT_198632 |
| Chaperonin role for actin and tubulin | UMAG_03959 | UHOR_06000 | UBRO_06000 | sr14864 | EX895_001395 | BCV70DRAFT_69984 |
| Chaperonin role for actin and tubulin | UMAG_02350 | UHOR_03838 | UBRO_03838 | sr13553 | EX895_004884 | BCV70DRAFT_198957 |
| Chaperonin role for actin and tubulin | UMAG_00565 | UHOR_00911 | UBRO_00911 | sr11843 | EX895_000809 | BCV70DRAFT_73687 |
| Chaperonin role for actin and tubulin | UMAG_04401 | UHOR_06879 | UBRO_06879 | sr15289 | EX895_002355 | BCV70DRAFT_200184 |
| Actin binding and severing protein | UMAG_04314 | UHOR_06753 | UBRO_06753 | sr15202 | EX895_002269 | BCV70DRAFT_211263 |
| Cortical actin cytoskeleton component | UMAG_04417 | UHOR_06902 | UBRO_06902 | sr15303 | EX895_002369 | BCV70DRAFT_200171 |
| Kinesin—Related to UMAG_ Kin14 | UMAG_11986 | UHOR_06484 | UBRO_06484 | sr15392 | EX895_002076 | BCV70DRAFT_202332 |
| Kinesin—Functionally redundant with Cin8 | UMAG_10678 | UHOR_07574 | UBRO_07574 | sr15603 | EX895_000239 | BCV70DRAFT_203263 |
| Kinesin—Related motor protein (UMAG_ Kin7a and Kin7b) | UMAG_00896 | UHOR_01350 | UBRO_01350 | sr12193 | EX895_006269 | BCV70DRAFT_199338 |
| Kinesin—related motor protein / Related to UMAG_ Kin8 | UMAG_01560 | UHOR_02319 | UBRO_02319 | sr12632 | EX895_004356 | BCV70DRAFT_199992 |
| Interacts with Myo2—related to motor domain of kinesins | UMAG_04218 | UHOR_06328 | UBRO_06328 | sr15103 | EX895_001669 | - |
| Type V myosin | UMAG_04555 | UHOR_06551 | UBRO_06551 | sr15438 | EX895_002121 | BCV70DRAFT_40829 |
| MEP1—Low affinity ammonium transporter | UMAG_04523 | UHOR_06506 | UBRO_06506 | sr15408 | EX895_002092 | BCV70DRAFT_208136 |
| MEP2—High affinity ammonium transporter | UMAG_05889 | UHOR_08388 | UBRO_08388 | sr16507 | EX895_003276 | BCV70DRAFT_199125 |
| Putative arginase—Homology with G6606 of U. esculenta | UMAG_04939 | UHOR_07065 | UBRO_07065 | sr15821 | EX895_005725 | BCV70DRAFT_178133 |
| Homology with SSAGC1 (SPSC_00276) of S. scitamineum) | UMAG_11677 | UHOR_02215 | UBRO_02215 | sr12574 | EX895_004280 | BCV70DRAFT_160776 |
| CDC24—Cell division control protein | UMAG_02422 | UHOR_03963 | UBRO_03963 | sr10836 | EX895_004832 | BCV70DRAFT_192816 |
| CLA4—Serine/threonine-protein kinase | UMAG_10145 | UHOR_03893 | UBRO_03893 | sr10814.2 | EX895_004846 | BCV70DRAFT_158032 |
| RHO1—Ras-like GTP-binding protein | UMAG_01032 | UHOR_01559 | UBRO_01559 | sr12336 | EX895_006088 | BCV70DRAFT_166498 |
| RHO1—Ras-like GTP-binding protein | UMAG_05734 | UHOR_12739 | UBRO_07439 | sr16067 | EX895_002745 | CE53947_104590 |
| FUZ1—MYND domain protein | UMAG_02587 | UHOR_04136 | UBRO_04136 | sr13628 | EX895_005246 | BCV70DRAFT_63386 |
Genes mostly analyzed by its deletion. ID genes according to NCBI (a), ExPASy (b), e!EnsemblFungi (c), and JGI MycoCosm (d), and considering the genomic data published for U. maydis [66], U. hordei [67], U. bromivora [68], S. reilianum [69], S. graminícola [70], and T. cyperi [71]; the six Ustilaginomycetes with the best-annotated genomes. Genes identified based on their homology with U. maydis. The analyzed sequences were deposited in https://github.com/lucilaortiz/Multicellularity_associated_proteins.
Contrary to the deletion of GCN5 HAT, the deletion of HOS2 and CLR3 HDACs affected U. maydis multicellular growth. In the Δhos2 mutant, the formation of the conjugation tube and the mating of compatible single-cell strains were altered. In addition, the multicellular dikaryotic mycelium and virulence were reduced [50].
In summary, the HATs and HDACs play an indispensable, but opposed, role in U. maydis multicellular growth and possibly in other Ustilaginomycetes fungi. While the histone acetylation by Gcn5 allows the expression of genes involved in unicellular growth as yeast (Figure 4A), histone deacetylation by Hos2 and Clr3 allows multicellular growth as mycelium (Figure 4B). Histone deacetylation has been usually associated with gene repression. However, these findings demonstrate that the HDACs also function as transcriptional activators [51]. Considering that these genes are evolutionarily conserved in eukaryotic organisms such as Ustilaginomycetes (Table 3) [124,125,126,127,128,129], and that most eukaryotic organisms are multicellular, it is tempting to speculate about the importance of this histone acetylation or deacetylation switch during the developmental process by which unicellular organisms became multicellular during evolution.
Another epigenetic mechanism suggested in the regulation of the multicellular growth of Ustilaginomycetes is DNA methylation. Different DNA methylation patterns were observed during the unicellular and multicellular growth of U. maydis wild-type strains [130]. Interestingly the mutants affected in the putrescine synthesis (Δodc), which could not grow multicellularly as mycelia (discussed above) [46], showed DNA methylation patterns similar to those of the wild-type yeast [131]. Knowing that polyamines bind to DNA [132], and that DNA methylation is involved in gene silencing [133], it suggests that polyamines, particularly spermidine, prevent DNA methylation, thus allowing the expression of genes involved in U. maydis multicellular growth [134]. Taking into consideration that the genes involved in the polyamine synthesis are conserved in Ustilaginomycetes (Table 2), it is possible that this regulatory mechanism of multicellular growth is present in this class of fungi.
5. The Cell Wall in the Multicellular Growth of Ustilaginomycetes
The cell wall is the rigid outer structure that covers the cells of prokaryotic and eukaryotic organisms. It protects the cells from osmotic pressure differences between the cytoplasm and the external medium and gives shape to the cell. In fungi, the cell wall is a compound of polysaccharide microfibrils (chitin and β-1,3-glucans) immersed in a glycoprotein matrix [135]. This cellular organelle is a dynamic structure whose composition changes during the cell cycle, depending on the environmental conditions. It is also directly involved in the unicellular or multicellular development of fungi. The cell wall components are synthesized in defined quantities, according to time and space to form a coherent and organized structure [136,137].
Chitin is the characteristic component of the fungal cell wall, giving it rigidity. It is synthetized by the chitin synthase proteins (Chs), which are encoded by the genes known as CHS. Fungi have multiple CHS genes in their genomes, and it can be a compensatory mechanism of the cell in response to the loss of a certain proportion of them. S. cerevisie has three CHS genes: CHS1, which has a repair function during cell separation, allowing unicellular growth [138]; CHS2 is involved in cell division, and it is essential for septum formation during multicellular growth [139]; and CHS3, which is responsible for chitin synthesis in the ring at bud emergence, for the chitin synthesized in the cell wall, and for chitosan synthesis in ascospores [140]. In multicellular fungi, such as N. crassa and Aspergillus nidulans, the CHS genes are necessary for normal hyphae development [141,142].
Ustilaginomycetes have approximately eight CHS genes (Table 4). In U. maydis, the Δchs6 mutants showed morphological alterations that were more notorious during the multicellular growth as mycelium [143]. Chs6, Chs5, Chs7 and Chs8 are localized in the growth zone in the hyphal tip, which indicates that these could contribute to polarized growth during multicellularity in the smut fungus [144]. Interestingly, the CHS genes are differentially expressed during the dimorphic transition of several fungi, e.g., Mucor circinelloides and Paracoccidiodes brasiliensis, showing high levels of expression in the mycelium form [145,146]. In U. maydis, its eight CHS genes were expressed in both yeast (unicellular growth, Figure 4A) and mycelium forms (multicellular growth, Figure 4B), and similarly, as in the previously described fungi, almost all the CHS genes showed high levels of expression in the mycelium [52].
Table 4.
Genes involved in synthesis and degradation of the cell wall, and putatively involved in the Ustilaginomycetes multicellular growth.
| Cell wall | Gene | Ustilago maydis a |
Ustilago
hordei a,b |
Ustilago
bromivora c |
Sporisorium
reilianum a,b |
Sporisorium
graminicola a |
Testicularia
cyperi a,b,d |
|---|---|---|---|---|---|---|---|
| Synthesis | 1,3-β-glucan synthase | UMAG_01639 | UHOR_02430 | UBRO_02430 | sr12707 | EX895_004433 | BCV70DRAFT_98536 |
| CHS1 | UMAG_10718 | UHOR_07282 | UBRO_07282 | sr16010 | EX895_002642 | BCV70DRAFT_198128 | |
| CHS2 | UMAG_04290 | UHOR_06435 | UBRO_06435 | sr15181 | EX895_002037 | - | |
| CHS3 | UMAG_10120 | UHOR_00740 | UBRO_00740 | sr10158 | EX895_000703 | BCV70DRAFT_196781 | |
| CHS4 | UMAG_10117 | UHOR_00723 | UBRO_00723 | sr10139 | EX895_000691 | CE2605_17934 | |
| CHS5 | UMAG_10277 | UHOR_04112 | UBRO_04112 | sr11048.2 | EX895_005224 | BCV70DRAFT_156817 | |
| CHS6 | UMAG_10367 | UHOR_04780 | UBRO_04780 | sr14109 | EX895_002515 | BCV70DRAFT_22903 | |
| CHS7 | UMAG_05480 | UHOR_07854 | UBRO_07854 | sr16158 | EX895_002841 | BCV70DRAFT_162870 | |
| CHS8 | UMAG_03204 | UHOR_04988 | UBRO_04988 | sr11106 | EX895_005864 | BCV70DRAFT_105640 | |
| CDA1 d | UMAG_11922 | UHOR_04296 | UBRO_04296 | sr13741 | EX895_005345 | BCV70DRAFT_210413 | |
| CDA2 d | UMAG_00126 | UHOR_00199 | UBRO_00199 | sr11468 | EX895_000151 | BCV70DRAFT_197174 | |
| CDA3 d | UMAG_00638 | - | - | sr11918 | EX895_000863 | - | |
| CDA4 d | UMAG_01143 | UHOR_01725 | UBRO_01725 | sr12442 | EX895_006063 | - | |
| CDA5 d | UMAG_01788 | UHOR_02660 | UBRO_02660 | sr12866 | EX895_003869 | BCV70DRAFT_211320 | |
| CDA6 d | UMAG_02019 | UHOR_03000 | UBRO_03000 | sr12981 | EX895_004113 | BCV70DRAFT_34433 | |
| CDA7 d | UMAG_02381 | UHOR_03898 | UBRO_03898 | sr13587 | EX895_004867 | BCV70DRAFT_163992 | |
| CDA8 d | UMAG_05792 | UHOR_04296 | UBRO_04296 | sr16123 | EX895_002800 | BCV70DRAFT_210413 | |
| Degradation | CTS1 | UMAG_10419 | UHOR_06394 | UBRO_06394 | sr15153 | EX895_002009 | BCV70DRAFT_196730 |
| CTS2 | UMAG_02758 | UHOR_04393 | UBRO_04393 | sr13813 | EX895_005415 | CE60997_75476 | |
| CTS3 | UMAG_06190 | UHOR_08772 | UBRO_06394 | sr11305 | EX895_003673 | BCV70DRAFT_178298 | |
| CTS4 | UMAG_00695 | UHOR_01069 | UBRO_01069 | sr11983 | EX895_000921 | BCV70DRAFT_213542 | |
| CTS5 | UMAG_05290 | UHOR_08119 | UBRO_08119 | sr10043 | EX895_003044 | BCV70DRAFT_197812 | |
| Glucanase 1 | UMAG_04368 | - | UBRO_06828 | sr15253 | - | - | |
| Glucanase 2 | UMAG_05036 | UHOR_07211 | UBRO_07211 | sr15917 | - | CE121843_61034 | |
| Glucanase 3 | UMAG_00876 | UHOR_00370 | UBRO_01319 | sr12165 | EX895_006333 | CE84129_149800 | |
| Glucanase 4 | UMAG_01898 | - | UBRO_02820 | sr10624 | EX895_003984 | - | |
| Glucanase 5 | UMAG_05223 | UHOR_03204 | UBRO_03204 | sr13131 | EX895_004619 | BCV70DRAFT_227049 | |
| Glucanase 6 | UMAG_02134 | - | UBRO_03533 | sr13360 | EX895_004991 | - | |
| Glucanase 7 | UMAG_04357 | UHOR_06809 | UBRO_06809 | sr15243 | EX895_002306 | BCV70DRAFT_189826 | |
| Glucanase 8 | UMAG_05550 | - | UBRO_06823 | sr15250 | EX895_002316 | BCV70DRAFT_200231 | |
| Glucanase 9 | UMAG_10211 | - | UBRO_08206 | sr16359 | EX895_003114 | - |
ID genes according to NCBI (a), ExPASy (b), e!EnsemblFungi (c), and JGI MycoCosm (d), and considering the genomic data published for U. maydis [66], U. hordei [67], U. bromivora [68], S. reilianum [69], S. graminícola [70], and T. cyperi [71]; the six Ustilaginomycetes with the best-annotated genomes. Genes identified based on their homology with U. maydis. CHS, Chitinsynthase; CDA, Chitindeacetylase; and CTS, Chitinase. The sequences analyzed were deposited in https://github.com/lucilaortiz/Multicellularity_associated_proteins.
Chitosan is another component of the fungal cell wall, although its function is less understood than that of chitin. Chitosan is synthetized through the partial deacetylation of chitin by chitin deacetylases (Cda). S. cerevisiae has two CDA genes, CDA1 and CDA2, that are expressed only during sporulation. CDA1 and CDA2 have redundant functions in chitosan synthesis in ascospores, which contributes to their resistance to lytic enzymes [147]. Of the ten Magnaporthe oryzae CDA genes, only CDA1 showed high levels of expression during its multicellular filamentous growth. However, Δcda1 mutat did not show alteration in the chitin content of the wall. Moreover, Cda1 was localized in the cell wall and septa during hyphae multicellular growth, but not in the growth zones. These findings suggest that CDA has an indirect function in hyphae morphology [148]. Ustilaginomycetes have several CDA genes; for example, U. maydis has eight CDA genes (Table 4). CDA1 was 40 times up-regulated in the multicellular mycelial form [53], and the Δcda1 mutants developed aberrant hyphae [149].
Polysaccharide β-1,3-glucans are the major components of the cell wall, which are synthetized by the β-1,3-glucan synthases (Gls). In S. cerevisiae, the genes that encode the catalytic subunit of Gls enzymes are known as FKS, due to their sensitivity to FK506 immunosuppressants, but in other fungi, they are known as GLS. N. crassa has one GLS-1 gene. gls-1-RNAi transformants show a decrease in the β-1,3-glucans synthesis, and abnormalities in the development of the multicellular mycelium; the hyphae were shorter than those of the wild type strain [150]. Gls-1-GFP accumulates at the apex of the hyphae forming a ring structure in the outer region of the Spitzenkörper [151]. In the Ustilaginomycete U. maydis, the GLS gene showed constitutive expression during multicellular growth like mycelium [52].
On the other hand, in the different stages of fungi development and multicellular growth, the cell wall is remodeled through the synthesis and degradation of their compounds. The cell wall degradation by chitinases and glucanases causes plasticity, which allows the introduction of new synthetized components by forming links between polymers and leading to the expansion of the cellular surface through turgor force [136,137].
The chitinases (Cts) remodel chitin in the cell wall. In S. cerevisiae, the endo chitinase Cts1 is necessary for unicellular growth, allowing cellular separation after cytokinesis. The Δcts1 mutants showed abnormal separation during budding growth, forming cellular clusters [152]. During budding (unicellular growth), Cts1 is localized at the mother-daughter neck, contributing to the degradation of the septum, and allowing cell separation [153]. In C. albicans, deletion of the CTS1 and CTS2 chitinase genes increases the development of pseudomycelium or cellular clusters [154]. Interestingly, the N. crassa CTS-1 gene is also important for the development of mycelium, which suggests that it has a function in remodeling the cell wall during fungus multicellular growth. The Δcht-1 mutants showed reduced mycelial growth compared to that of the wild- type strains [155]. In Ustilaginomycetes, there are approximately five putative chitinase genes (Table 4). However, they may have redundant functions. In U. maydis, only the double mutant Δcts1/Δcts2, but not the single mutants, showed alterations in cell separation. Cts1 and Cts2 are active during unicellular growth as yeast, but only Cts1 showed activity during multicellular growth as mycelium [54]. However, CTS2 was 6.7 times up-regulated in the U. maydis mycelium induced by the acidic pH in the culture medium. [53,123]. Likewise, the β-1,3-endo and exo-glucanases have a function in septum separation, and the β-1,3-glucans in elongation and reorganization. ENG1 encodes a β-1,3-endoglucanase in S. cerevisiae. The deletion of ENG1 shows alteration of the separation of septae and induces growth of mutants as multicellular clusters. Eng1 is mainly localized at the bud neck on the daughter side [156]. Ustilaginomycetes have several putative glucanases (Table 4). However, little is known about their function during unicellular or multicellular growth in these fungi.
Finally, the putative importance of genes involved in cell wall synthesis and structure during development of S. reilianum multicellular clusters was determined by transcriptional network analysis [13]. The examples of genes involved include those encoding: 1,3-beta-D-glucan synthase, glucan 1,3-beta-glucosidase, GPI-proteins, UTR2-cell wall proteins, chitin synthase 2 (Chs2), Chs3, exo-1,3-beta-glucanase, Kre6-glucan synthase, chitin deacetylases (Cda), glucan 1,3-beta-glucosidase, and several proteins involved in cell wall biogenesis and architecture. Some of them, such as UTR2-cell wall, CHS3, ECM4, involved in cell wall biogenesis and architecture, and CDA, were down regulated in the multicellular cluster form of the fungus [13]. All these findings demonstrate the direct and critical role of the cell wall in fungi multicellularity. It is tempting to hypothesize that the cell wall is the most important structure involved in this phenomenon in fungi.
6. Polarized Growth during the Multicellularity of Ustilaginomycetes
Mycelia or filamentous growth is one of the best-known forms of simple multicellular structures of fungi. The mycelia are joined and elongated cells in the form of tubes, which extend at the tip and branch out into sub-apical areas involving a coordinated action of the microtubule and actin cytoskeletons [157]. In Ustilaginomycetes, this phenomenon has been studied most in Ustilago maydis, although the genes involved in polarized growth are mostly conserved in this class of fungi (Table 3).
The actin cytoskeleton in U. maydis is formed by three structures: Actin cables, actin patches and an actin ring. During the multicellular growth, the hyphae contain an actin cap at the tip, and the actin cables polarize towards the tip [158]. LIS1 is a gene encoding a microtubule plus-end tracking protein required for microtubule cytoskeleton organization, cell wall integrity, septa positioning, and nuclear migration during the filamentous growth. The Δlis1 mutants showed an aberrant multicellular morphology [159]. Likewise, the TEA4 gene is involved in the polarized growth, the control of position and the number of septa, as well as the nuclear division in the mycelial form [160]. Also, FUZ1, a gene encoding the MYND Zn finger domain protein, is important for the cell wall integrity and dikaryotic filamentous growth after the fusion of compatible sporidia [161].
In Ustilaginomycetes, as in all the filamentous fungi, the kinesin proteins are involved in vesicle transport. U. maydis has ten kinesins: Kin1, Kin3, Kin4, Kin6, Kin7a, Kin7b, Kin8, Kin9, and Kin14. Of all these proteins, only Kin1 and Kin3 are important for normal multicellular filamentous growth. Δkin1, Δkin3 and the double mutants Δkin1/Δkin3 show bipolar growth and short hyphae [162]. Kin1 and Kin3, as well as the myosin Myo5 and Tea1, localize and accumulate at the hyphal apex during multicellular growth [162,163,164], although Tea1 is also localized in new sites of the filamentous growth [164]. The mutation of the MYO5 gene (Δmyo5) did not affect the filamentous growth of the fungus. However, it caused irregular growth and irregular chitin deposition [163], thus demonstrating the important relationship between the fungi multicellular filamentous growth and their cell wall.
7. Other Cellular Processes Involved in the Ustilaginomycetes Multicellular Growth
Ustilaginomycetes are model organisms to study the different processes that occur in fungi. Several genes involved in the cellular processes associated with their multicellular growth have been described. Interestingly, these genes are conserved in this class of fungi (Table 3), and it is possible that those genes and the cellular processes work in the same way in Ustilaginomycetes.
In U. maydis, it has been observed that REP1, HUM2, and the HUM3 genes encode repellent proteins and hydrophobins, a group of small cysteine rich proteins required for the development of multicellular aerial hyphae. The Δrep1 and Δhum2 mutants showed a drastic reduction of aerial mycelium [165,166]. Moreover, HUM2 and HUM3 were overexpressed 9.1 and 14.7 times, respectively, during the formation of the hyphal layer in U. maydis basidiocarps [41].
Likewise, the importance of the PEP4 gene encoding the vacuolar acid proteinase PrA during U. maydis mycelial growth under stress conditions and pathogenesis has been reported. The Δpep4 mutants showed a severe reduction of multicellular mycelium growth induced by the acidic culture medium or using fatty acids as a carbon source [167]. The GTPases Cdc4 and Rho1 also played essential roles during the polarized growth and filamentation of this fungus [168,169]. In addition, the RNA-binding protein Rrm4 was crucial for the mycelium polarity during infection, because it is involved in the microtubule-dependent transport of mRNAs during fungus multicellular growth [170].
In S. cerevisiae and other fungi, a low level of nitrogen in the medium induces multicellular growth of pseudomycelium or mycelium [72]. An important mechanism that links the availability of nitrogen and the unicellular-multicellular growth is are the methyl ammonium permeases (Mep). In U. maydis, the MEP1 and MEP2 genes, are important for the filamentous development in a medium with low ammonium. The Δump2 mutants did not grow as mycelia, and the Δump1/Δump2 double mutant showed multicellular aggregates and sediment in liquid media with low ammonia concentrations. Moreover, it was suggested that Mep2 is phosphorylated by the cAMP/PKA signaling pathway in response to low nitrogen conditions [171]. Interestingly, the genes encoding the protein involved in the export of ammonia were up-regulated during U. maydis multicellular growth induced under acidic conditions [123]. Similarly, the SSA2 ammonium permease was up-regulated in Sporisorium scitamineum during unicellular growth induced by mycophenolic acid, which is a fungal mycelium inhibitor [172]. Also, in S. scitamineum, the deletion of the SSATG8 gene encoding the Atg8 protein involved in autophagy caused multicellular growth as pseudomycelium and hypersensitivity to oxidative stress [173].
Finally, in the Ustilaginomycete Ustilago esculenta, a pathogenic fungus of Zizania latifolia, the addition of exogenous arginine or the deletion of the UeArginase gene inhibited its multicellular growth as mycelium and reduced the expression of the UeKPP6, UePKAC and UePRF1 genes, thus suggesting that arginine acts as an inhibitor of the cAMP-PKA signaling pathway [174].
All these findings demonstrate that multicellular development in Ustilaginomycetes is a holistic phenomenon, and that it is necessary to deal with the whole cell to the largest possible extent.
8. Conclusions
Multicellularity is defined as the developmental process by which unicellular organisms have become pluricellular during evolution. The Ustilaginomycetes are eukaryotic microorganisms with the capacity to grow unicellularly in the form of saprophytic yeasts or like undifferentiated and simple multicellular structures: Mycelium, pseudomycelium, and cellular clusters. Complex multicellular structures have also been described in the case of U. maydis through the formation of basidiocarps.
The data discussed in this study show that the molecular mechanisms involved in multicellular growth may be conserved in Ustilaginomycetes. These fungi have an important set of genes and regulatory mechanisms involved in their unicellular and multicellular growth. Examples of these mechanisms are: The MAPK and cAMP/PKA signaling transduction pathways, the mechanisms of epigenetic regulation, the metabolic requirements, the polarized cell growth, and the processes of cell wall synthesis and degradation. However, although these fungi are phylogenetically related, they can respond with different morphologies to. For example, U. maydis grows as mycelium in acidic culture media, while S. reilianum grows in the form of multicellular clusters under the same conditions.
Basically, the MAPK and PKA pathways transduce environmental signals to the cell nucleus by the activation of transcription factors, which bind to DNA for the transcription of target genes involved in the Ustilaginomycetes unicellular or multicellular growth. Putatively, the transcription of target genes requires the binding of polyamines (spermidine) to DNA, avoiding its methylation and allowing its transcription. Undoubtedly, the coordinated acetylation or deacetylation of histones is required to access the target DNA and its transcription.
As described above, Ustilaginomycetes multicellularity can be induced in vitro by different kinds of stress or induced in vivo as a fundamental requirement for host plant infection and colonization. Regarding host plant infection, it is well known that mating of compatible sporidia induces filamentous growth in the fungus. In addition, it is well established that many phytopathogenic Ustilaginomycetes have a limited range of plant hosts. However, little is known about the plant compounds that induce the multicellular filamentous development for host plant colonization; or about the plant compounds sensed by Ustilaginomycetes to recognize potential hosts. It would also be important to increase our knowledge of how cell-cell communication occurs in the different multicellular forms developed by the Ustilaginomycetes. It is possible that the communication between cells is also carried out through the MAPK and cAMP/PKA signaling pathways.
There are many cellular processes involved in fungi multicellularity, and it is important to understand that multicellularity is a holistic phenomenon. Therefore, it is necessary to deal with the whole cell. Finally, taking into consideration the morphological plasticity presented by Ustilaginomycetes, the availability of their sequenced genomes, their short life cycle, and easy handling in the laboratory; in this work, we suggest the use of Ustilaginomycetes as model organisms for studying fungi multicellularity.
Acknowledgments
The authors wish to thank Luis J. Delaye-Arredondo from CINVESTAV, Irapuato Unit, for his assistance with some analyses; and to Bibiana Chávez-Munguía from CINVESTAV, Zacatenco Unit, for her support conducting scanning electron microscopy. We also thank Y. Ithaí Ángeles-López from North Carolina State University, for helping with some scientific drawings. DMS is a postdoctoral researcher with a scholarship granted by UC MEXUS-CONACYT.
Author Contributions
Conceptualization, D.M.-S.; software, L.O.-C.; validation, D.M.-S. and L.O.-C.; formal analysis, D.M.-S. and L.O.-C.; investigation, D.M.-S., L.O.-C., M.R.-B. and C.G.L.-R.; data curation, D.M.-S. and L.O.-C.; writing—original draft preparation, D.M.-S. and M.R.-B.; writing—review and editing, D.M.-S. and L.O.-C.; visualization, D.M.-S., L.O.-C. and C.G.L.-R.; supervision, D.M.-S.; project administration, D.M.-S. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
References
- 1.Maynard-Smith J., Szathmary E. Major Transitions in Evolution. Spektrum; New York, NY, USA: 1995. [Google Scholar]
- 2.Bonner J.T. The origins of multicellularity. Integr. Biol. 1998;1:27–36. doi: 10.1002/(SICI)1520-6602(1998)1:1<27::AID-INBI4>3.0.CO;2-6. [DOI] [Google Scholar]
- 3.Pfeiffer T., Bonhoeffer S. An evolutionary scenario for the transition to undifferentiated multicellularity. Proc. Natl. Acad. Sci. USA. 2003;100:1095–1098. doi: 10.1073/pnas.0335420100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Rokas A. The origins of multicellularity and the early history of the genetic toolkit for animal development. Annu. Rev. Genet. 2008;42:235–251. doi: 10.1146/annurev.genet.42.110807.091513. [DOI] [PubMed] [Google Scholar]
- 5.Rivera-Yoshida N., Arias Del Angel J.A., Benítez M. Microbial multicellular development: Mechanical forces in action. Curr. Opin. Genet. Dev. 2018;51:37–45. doi: 10.1016/j.gde.2018.05.006. [DOI] [PubMed] [Google Scholar]
- 6.Kessin R.H., Gundersen G.G., Zaydfudim V., Grimson M. How cellular slime molds evade nematodes. Proc. Natl. Acad. Sci. USA. 1996;93:4857–4861. doi: 10.1073/pnas.93.10.4857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Boraas M., Seale D., Boxhorn J. Phagotrophy by a flagellate selects for colonial prey: A posible origin of multicellularity. Evol. Ecol. 1998;12:153–164. doi: 10.1023/A:1006527528063. [DOI] [Google Scholar]
- 8.Smukalla S., Caldara M., Pochet N., Beauvais A., Guadagnini S., Yan C., Vinces M.D., Jansen A., Prevost M.C., Latgé J.P., et al. FLO1 is a variable green beard gene that drives biofilm-like cooperation in budding yeast. Cell. 2008;135:726–737. doi: 10.1016/j.cell.2008.09.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Knoll A. The multiple origins of complex multicellularity. Annu. Rev. Earth Planet. Sci. 2011;39:217–239. doi: 10.1146/annurev.earth.031208.100209. [DOI] [Google Scholar]
- 10.Koschwanez J.H., Foster K.R., Murray A. Sucrose utilization in budding yeast as a model for the origin of undifferentiated multicellularity. PLoS Biol. 2011;9:e1001122. doi: 10.1371/annotation/0b9bab0d-1d20-46ad-b318-d2229cde0f6f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Oud B., Guadalupe-Medina V., Nijkamp J.F., de Ridder D., Pronk J.T., van Maris A.J., Daran J.M. Genome duplication and mutations in ACE2 cause multicellular, fast-sedimenting phenotypes in evolved Saccharomyces cerevisiae. Proc. Natl. Acad. Sci. USA. 2013;110:E4223–E4231. doi: 10.1073/pnas.1305949110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Arias Del Angel J.A., Escalante A., Martínez-Castilla L., Benítez M. An evo-devo perspective on multicelular development of Myxobacteria. J. Exp. Zool. Part. B Mol. Dev. Evol. 2017;328:165–178. doi: 10.1002/jez.b.22727. [DOI] [PubMed] [Google Scholar]
- 13.Martínez-Soto D., Velez-Haro J.M., León-Ramírez C.G., Galán-Vásquez E., Chávez-Munguía B., Ruiz-Herrera J. Multicellular growth of the Basidiomycota phytopathogen fungus Sporisorium reilianum induced by acid conditions. Folia Microbiol. (Praha) 2020;65:511–521. doi: 10.1007/s12223-019-00755-7. [DOI] [PubMed] [Google Scholar]
- 14.Rensing S.A. (Why) Does evolution favour embryiogenesis? Trends Plant. Sci. 2016;21:562–573. doi: 10.1016/j.tplants.2016.02.004. [DOI] [PubMed] [Google Scholar]
- 15.Pentz J.T., Marquez-Zacarías P., Yunker P.J., Libby E., Ratcliff W.C. Ecological advantages and evolutionary limitations of aggregative multicellular development. bioRxiv. 2018:255307. doi: 10.1101/255307. [DOI] [PubMed] [Google Scholar]
- 16.Kiss E., Hegedüs B., Virágh M., Varga T., Merényi Z., Kószó T., Bálint B., Prasanna A.N., Krizsán K., Kocsubé S., et al. Comparative genomics reveals the origin of fungal hyphae and multicellularity. Nat. Commun. 2019;10:4080. doi: 10.1038/s41467-019-12085-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Nagy L.G., Kovács G.M., Krizsan K. Complex multicellularity in fungi: Evolutionary convergence, single origin, or both? Biol Rev. Camb Philos Soc. 2018;93:1778–1794. doi: 10.1111/brv.12418. [DOI] [PubMed] [Google Scholar]
- 18.Harris S.D. Cell polarity in filamentous fungi: Shaping the mold. Intl. Rev. Cytol. 2006;251:41–77. doi: 10.1016/S0074-7696(06)51002-2. [DOI] [PubMed] [Google Scholar]
- 19.Fischer M.S., Glass N.L. Communicate and fuse: How filamentous fungi establish and maintain an interconnected mycelial network. Front. Microbiol. 2019;10:619. doi: 10.3389/fmicb.2019.00619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Goncalves A.P., Heller J., Span E.A., Rosenfield G., Do H.P., Palma-Guerrero J., Requena N., Marletta M.A., Glass N.L. Allorecognition upon fungal cell-cell contact determines social cooperation and impacts the acquisition of multicellularity. Curr. Biol. 2019;29:3006–3017. doi: 10.1016/j.cub.2019.07.060. [DOI] [PubMed] [Google Scholar]
- 21.Kües U. Life history and developmental processes in the basidiomycete Coprinus cinereus. Microbiol. Mol. Biol. Rev. 2000;64:316–353. doi: 10.1128/MMBR.64.2.316-353.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Gancedo J.M. Control of pseudohyphae formation in Saccharomyces cerevisiae. FEMS Microbiol. Rev. 2001;25:107–123. doi: 10.1111/j.1574-6976.2001.tb00573.x. [DOI] [PubMed] [Google Scholar]
- 23.Ruiz-Herrera J., Sentandreu R. Different effectors of dimorphism in Yarrowia lipolytica. Arch. Microbiol. 2002;178:477–483. doi: 10.1007/s00203-002-0478-3. [DOI] [PubMed] [Google Scholar]
- 24.Kelly M.T., MacCallum D.M., Clancy S.D., Odds F.C., Brown A.J., Butler G. The Candida albicans CaACE2 gene affects morphogenesis, adherence and virulence. Mol. Microbiol. 2004;53:969–983. doi: 10.1111/j.1365-2958.2004.04185.x. [DOI] [PubMed] [Google Scholar]
- 25.Ratcliff W.C., Frankhauser J.D., Rogers D.W., Greig D., Travisano M. Origins of multicellular evolvability in snowflake yeast. Nat. Commun. 2015;6:6102. doi: 10.1038/ncomms7102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.He M.-Q., Zhao R.-L., Hyde K.D., Begerow D., Kemler M., Yurkov A., McKenzie E.H.C., Raspé O., Kakishima M., Sánchez-Ramírez S., et al. Notes, outline and divergence times of Basidiomycota. Fungal Divers. 2019;99:105–367. doi: 10.1007/s13225-019-00435-4. [DOI] [Google Scholar]
- 27.Snetselaar K.M., Mims C.W. Light and electron microscopy of Ustilago maydis hyphae in maize. Mycol. Res. 1994;98:347–355. doi: 10.1016/S0953-7562(09)80463-2. [DOI] [Google Scholar]
- 28.Martinez-Espinoza A.D., Gerhardt S.A., Sherwood J.E. Morphological and mutational analysis of mating in Ustilago hordei. Exp. Mycol. 1993;17:200–214. doi: 10.1006/emyc.1993.1019. [DOI] [Google Scholar]
- 29.Martinez C., Roux C., Dargent R. Biotrophic development of Sporisorium reilianum f. sp. zeae in vegetative shoot apex of maize. Phytopathology. 1999;89:247–253. doi: 10.1094/PHYTO.1999.89.3.247. [DOI] [PubMed] [Google Scholar]
- 30.Martinez C., Roux C., Jauneau A., Dargent R. The biological cycle of Sporisorium reilianum f. sp. zeae: An overview using microscopy. Mycologia. 2002;94:505–514. doi: 10.1080/15572536.2003.11833215. [DOI] [PubMed] [Google Scholar]
- 31.Martínez-Soto D., Velez-Haro J.M., León-Ramírez C.G., Ruiz-Medrano R., Xoconostle-Cázares B., Ruiz-Herrera J. The cereal phytopathogen Sporisorium reilianum is able to infect the non-natural host Arabidopsis thaliana. Eur. J. Plant. Pathol. 2019;153:417–427. doi: 10.1007/s10658-018-1567-8. [DOI] [Google Scholar]
- 32.Brefort T., Doehlemann G., Mendoza-Mendoza A., Reissmann S., Djamei A., Kahmann R. Ustilago maydis as a pathogen. Annu. Rev. Phytopathol. 2009;47:423–445. doi: 10.1146/annurev-phyto-080508-081923. [DOI] [PubMed] [Google Scholar]
- 33.Bhaskaran S., Smith R.H. Carbohydrates, invertase activity, growth and dimorphism in Sporisorium reilianum. Mycopathologia. 1993;122:35–41. doi: 10.1007/BF01103707. [DOI] [Google Scholar]
- 34.Banuett F., Herskowitz I. Morphological transitions in the life cycle of Ustilago maydis and their genetic control by the a and b loci. Exp. Mycol. 1994;18:247–266. doi: 10.1006/emyc.1994.1024. [DOI] [Google Scholar]
- 35.Ruiz-Herrera J., León-Ramírez C.G., Guevara-Olvera L., Cárabez-Trejo A. Yeast-mycelial dimorphism of haploid and diploid strains of Ustilago maydis. Microbiology. 1995;141:695–703. doi: 10.1099/13500872-141-3-695. [DOI] [Google Scholar]
- 36.Klose J., de Sá M.M., Kronstad J.W. Lipid-induced filamentous growth in Ustilago maydis. Mol. Microbiol. 2004;52:823–835. doi: 10.1111/j.1365-2958.2004.04019.x. [DOI] [PubMed] [Google Scholar]
- 37.Zapata-Morín P.A., Fuetes-Dávila G., Adame-Rodríguez J.M., Aréchiga-Carvajal E.T. Effect of pH and carbon source on the vegetative growth of Ustilago cynodontis (Pass.) Henn. in a solid and liquid culture médium. Rev. Mex. Fitopatol. 2010;28:159–161. [Google Scholar]
- 38.Poloni A., Schirawski J. Host specificity in Sporisorium reilianum is determined by distinct mechanisms in maize and sorghum. Mol. Plant. Pathol. 2016;17:741–754. doi: 10.1111/mpp.12326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Sabbagh S.K., Diagne-Léye G., Naudan M., Roux C.P. Solopathogenic strain formation strongly differs among Ustilaginaceae species. FEMS Microbiol. Lett. 2010;305:121–127. doi: 10.1111/j.1574-6968.2010.01912.x. [DOI] [PubMed] [Google Scholar]
- 40.Cabrera-Ponce J.L., León-Ramírez C.G., Verver-Vargas A., Palma-Tirado L., Ruiz-Herrera J. Metamorphosis of the Basidiomycota Ustilago maydis: Transformation of yeast-like cells into basidiocarps. Fungal Genet. Biol. 2012;49:765–771. doi: 10.1016/j.fgb.2012.07.005. [DOI] [PubMed] [Google Scholar]
- 41.León-Ramírez C.G., Cabrera-Ponce J.L., Martínez-Soto D., Sánchez-Arreguin A., Aréchiga-Carvajal E.T., Ruiz-Herrera J. Transcriptomic analysis of basidiocarp development in Ustilago maydis (DC) Cda. Fungal Genet. Biol. 2017;101:34–45. doi: 10.1016/j.fgb.2017.02.007. [DOI] [PubMed] [Google Scholar]
- 42.Gold S., Duncan G., Barrett K., Kronstad J. cAMP regulates morphogenesis in the fungal pathogen Ustilago maydis. Genes Dev. 1994;8:2805–2816. doi: 10.1101/gad.8.23.2805. [DOI] [PubMed] [Google Scholar]
- 43.Dürrenberger F., Wong K., Kronstad J.W. Identification of a cAMP-dependent protein kinase catalytic subunit required for virulence and morphogenesis in Ustilago maydis. Proc. Natl. Acad. Sci. USA. 1998;95:5684–5689. doi: 10.1073/pnas.95.10.5684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Martínez-Espinoza A.D., Ruiz-Herrera J., León-Ramírez C.G., Gold S.E. MAP kinase and cAMP signaling pathways modulate the pH-induced yeast-to-mycelium dimorphic transition in the corn smut fungus Ustilago maydis. Curr. Microbiol. 2004;49:274–281. doi: 10.1007/s00284-004-4315-6. [DOI] [PubMed] [Google Scholar]
- 45.Martínez-Soto D., Ruiz-Herrera J. Functional analysis of the MAPK pathways in fungi. Rev. Iberoam. Micol. 2017;34:192–202. doi: 10.1016/j.riam.2017.02.006. [DOI] [PubMed] [Google Scholar]
- 46.Guevara-Olvera L., Xoconostle-Cázares B., Ruiz-Herrera J. Cloning and disruption of the ornithine decarboxylase gene of Ustilago maydis: Evidence for a role of polyamines in its dimorphic transition. Microbiology. 1997;143:2237–2245. doi: 10.1099/00221287-143-7-2237. [DOI] [PubMed] [Google Scholar]
- 47.Valdés-Santiago L., Ruiz-Herrera J. Stress and polyamine metabolism in fungi. Front. Chem. 2014;10:42. doi: 10.3389/fchem.2013.00042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.González-Prieto J.M., Rosas-Quijano R., Domínguez A., Ruiz-Herrera J. The UmGcn5 gene encoding histone acetyltransferase from Ustilago maydis is involved in dimorphism and virulence. Fungal Genet. Biol. 2014;71:86–95. doi: 10.1016/j.fgb.2014.09.002. [DOI] [PubMed] [Google Scholar]
- 49.Martínez-Soto D., González-Prieto J.M., Ruiz-Herrera J. Transcriptomic analysis of the GCN5 gene reveals mechanisms of the epigenetic regulation of virulence and morphogenesis in Ustilago maydis. FEMS Yeast Res. 2015;15:fov055. doi: 10.1093/femsyr/fov055. [DOI] [PubMed] [Google Scholar]
- 50.Elías-Villalobos A., Fernández-Álvarez A., Moreno-Sánchez I., Helmlinger D., Ibeas J.I. The Hos2 histone deacetylase controls Ustilago maydis virulence through direct regulation of mating-type genes. PLoS Pathog. 2015;11:e1005134. doi: 10.1371/journal.ppat.1005134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Elías-Villalobos A., Dominique-Helmlinger D., Ibeas J.I. Histone deacetylases: Revealing the molecular base of dimorphism in pathogenic fungi. Microb. Cell. 2015;2:491–493. doi: 10.15698/mic2015.11.240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Robledo-Briones M., Ruiz Herrera J. Transcriptional regulation of the genes encoding chitin and β-1, 3-glucan synthases from Ustilago maydis. Curr. Microbiol. 2012;65:85–90. doi: 10.1007/s00284-012-0129-0. [DOI] [PubMed] [Google Scholar]
- 53.Robledo-Briones M., Ruiz Herrera J. Regulation of genes involved in cell wall synthesis and structure during Ustilago maydis dimorphism. FEMS Yeast Res. 2013;13:74–84. doi: 10.1111/1567-1364.12011. [DOI] [PubMed] [Google Scholar]
- 54.Langner T., Öztürk M., Hartmann S., Cord-Landwehr S., Moerschbacher B., Walton J.D., Göhre V. Chitinases Are essential for cell separation in Ustilago maydis. Eukaryot. Cell. 2015;14:846–857. doi: 10.1128/EC.00022-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Klosterman S.J., Perlin M.H., Garcia-Pedrajas M., Covert S.F., Gold S.E. Genetics of morphoenesis and pathogenic development of Ustilago maydis. Adv. Genet. 2007;57:1–47. doi: 10.1016/S0065-2660(06)57001-4. [DOI] [PubMed] [Google Scholar]
- 56.Vollmeister E., Schipper K., Baumann S., Haag C., Pohlmann T., Janpeter Stock J., Feldbrügge M. Fungal development of the plant pathogen Ustilago maydis. FEMS Microbiol Rev. 2012;36:59–77. doi: 10.1111/j.1574-6976.2011.00296.x. [DOI] [PubMed] [Google Scholar]
- 57.Valdés-Santiago L., Cervantes-Chávez J.A., Ruiz-Herrera J. Ustilago maydis spermidine synthase is encoded by a chimeric gene, required for morphogenesis, and indispensable for survival in the host. FEMS Yeast Res. 2009;9:923–935. doi: 10.1111/j.1567-1364.2009.00539.x. [DOI] [PubMed] [Google Scholar]
- 58.Valdés-Santiago L., Guzmán-de-Peña D., Ruiz-Herrera J. Life without putrescine: Disruption of the gene-encoding polyamine oxidase in Ustilago maydis odc mutants. FEMS Yeast Res. 2010;10:928–940. doi: 10.1111/j.1567-1364.2010.00675.x. [DOI] [PubMed] [Google Scholar]
- 59.Valdés-Santiago L., Cervantes-Chávez J.A., Winkler R., León-Ramírez C.G., Ruiz-Herrera J. Phenotypic comparison of Samdc and Spe mutants reveals complex relationships of polyamine metabolism in Ustilago Maydis. Microbiology. 2012;158:674–684. doi: 10.1099/mic.0.055954-0. [DOI] [PubMed] [Google Scholar]
- 60.Aréchiga-Carvajal E.T., Ruiz-Herrera J. The RIM101/pacC homologue from the Basidiomycete Ustilago maydis is functional in multiple pH-sensitive phenomena. Eukaryot Cell. 2005;4:999–1008. doi: 10.1128/EC.4.6.999-1008.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Cervantes-Chávez J.A., Ortiz-Castellanos L., Tejeda-Sartorius M., Gold S., Ruiz-Herrera J. Functional analysis of the pH responsive pathway Pal/Rim in the phytopathogenic Basidiomycete Ustilago maydis. Fungal Genet. Biol. 2010;47:446–457. doi: 10.1016/j.fgb.2010.02.004. [DOI] [PubMed] [Google Scholar]
- 62.Cervantes-Montelongo J.A., Ruiz-Herrera J. Identification of a novel member of the pH responsive pathway Pal/Rim in Ustilago maydis. J. Basic Microbiol. 2019;59:14–23. doi: 10.1002/jobm.201800180. [DOI] [PubMed] [Google Scholar]
- 63.Méndez-Morán L., Reynaga-Peña C.G., Springer P.S., Ruiz-Herrera J. Ustilago maydis Infection of the nonnatural host Arabidopsis thaliana. Phytopatholoy. 2005;95:480–488. doi: 10.1094/PHYTO-95-0480. [DOI] [PubMed] [Google Scholar]
- 64.Martínez-Soto D., Robledo-Briones A.M., Estrada-Luna A.A., Ruiz-Herrera J. Transcriptomic analysis of Ustilago maydis infecting Arabidopsis reveals important aspects of the fungus pathogenic mechanisms. Plant. Signal. Behav. 2013;8:e25059. doi: 10.4161/psb.25059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Peñalva M.A., Arst H.N., Jr. Recent advances in the characterization of ambient pH regulation of gene expression in filamentous fungi and yeasts. Annu. Rev. Microbiol. 2004;58:425–451. doi: 10.1146/annurev.micro.58.030603.123715. [DOI] [PubMed] [Google Scholar]
- 66.Kämper J., Kahmann R., Bölker M., Ma L.-J., Brefort T., Saville B.J., Banuett F., Kronstad J.W., Gold S.E., Müller O., et al. Insights from the genome of the biotrophic fungal plant pathogen Ustilago maydis. Nature. 2006;444:97–101. doi: 10.1038/nature05248. [DOI] [PubMed] [Google Scholar]
- 67.Laurie J.D., Ali S., Linning R., Mannhaupt G., Wong P., Güldener U., Münsterkötter M., Moore R., Kahmann R., Bakkeren G., et al. Genome comparison of barley and maize smut fungi reveals targeted loss of RNA silencing components and species-specific presence of transposable elements. Plant. Cell. 2012;24:1733–1745. doi: 10.1105/tpc.112.097261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Rabe F., Bosch J., Stirnberg A., Guse T., Bauer L., Seitner D., Rabanal F.A., Czedik-Eysenberg A., Uhse S., Bindics J., et al. A complete toolset for the study of Ustilago bromivora and Brachypodium sp. as a fungal-temperate grass pathosystem. Elife. 2016;5:e20522. doi: 10.7554/eLife.20522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Schirawski J., Mannhaupt G., Münch K., Brefort T., Schipper K., Doehlemann G., Di Stasio M., Rössel N., Mendoza-Mendoza A., Pester D., et al. Pathogenicity determinants in smut fungi revealed by genome comparison. Science. 2010;330:1546–1548. doi: 10.1126/science.1195330. [DOI] [PubMed] [Google Scholar]
- 70.Solano-González S., Darby A.C., Cossar D., Caddick M.X. High-quality draft genome sequence and annotation of the Basidiomycete yeast Sporisorium graminicola CBS10092, a producer of mannosylerythritol lipids. Microbiol Resour Announc. 2019;8:e00479-19. doi: 10.1128/MRA.00479-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Kijpornyongpan T., Mondo S.J., Barry K., Sandor L., Lee J., Lipzen A., Pangilinan J., LaButti K., Hainaut M., Henrissat B., et al. Broad Genomic Sampling Reveals a Smut Pathogenic Ancestry of the Fungal Clade Ustilaginomycotina. Mol. Biol. Evol. 2018;35:1840–1854. doi: 10.1093/molbev/msy072. [DOI] [PubMed] [Google Scholar]
- 72.Banuett F. Signalling in the yeasts: An informational cascade with links to thefilamentous fungi. Microbiol. Mol. Biol. Rev. 1998;62:249–274. doi: 10.1128/MMBR.62.2.249-274.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Dohlman H.G. Thematic minireview series: Complexities of cellular signaling revealed by simple model organisms. J. Biol. Chem. 2016;291:7786–7787. doi: 10.1074/jbc.R116.722934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Correia I., Román E., Prieto D., Eisman B., Pla J. Complementary roles of the cek1 and cek2 map kinases in Candida albicans cell-wall biogénesis. Future Microbiol. 2016;11:51–67. doi: 10.2217/fmb.15.127. [DOI] [PubMed] [Google Scholar]
- 75.Davidson R.C., Nichols C.B., Cox G.M., Perfect J.R., Heitman J. A MAP kinase cascade composed of cell type specific and nonspecific elements controls mating and differentiation of the fungal pathogen Cryptococcus neoformans. Mol. Microbiol. 2003;49:469–485. doi: 10.1046/j.1365-2958.2003.03563.x. [DOI] [PubMed] [Google Scholar]
- 76.Leberer E., Harcus D., Broadbent I.D., Clark K.L., Dignard D., Ziegelbauer K., Schmidt A., Gow N.A., Brown A.J., Thomas D.Y. Signal transduction through homologs of the Ste20p and Ste7p protein kinases can trigger hyphal formation in the pathogenic fungus Candida albicans. Proc. Natl. Acad. Sci. USA. 1996;93:13217–13222. doi: 10.1073/pnas.93.23.13217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Pan X., Harashima T., Heitman J. Signal transduction cascades regulating pseudohyphal differentiation of Saccharomyces cerevisiae. Curr. Opin. Microbiol. 2000;3:567–572. doi: 10.1016/S1369-5274(00)00142-9. [DOI] [PubMed] [Google Scholar]
- 78.Biswas S., Van Dijck P., Datta A. Environmental sensing and signal transduction pathways regulating morphopathogenic determinants of Candida albicans. Microbiol. Mol. Biol. Rev. 2007;71:348–376. doi: 10.1128/MMBR.00009-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Klosterman S.J., Martinez-Espinoza A.D., Andrews D.L., Seay J.R., Gold S.E. UBC2, an ortholog of the yeast Ste50p adaptor, possesses a Basidiomycete-specific carboxy terminal extension essential for pathogenicity independent of pheromone response. Mol. Plant. Microbe Interact. 2008;21:110–121. doi: 10.1094/MPMI-21-1-0110. [DOI] [PubMed] [Google Scholar]
- 80.Martínez-Soto D., Ruiz-Herrera J. Regulation of the expression of the whole genome of Ustilago maydis by a MAPK pathway. Arch. Microbiol. 2015;197:575–588. doi: 10.1007/s00203-015-1087-2. [DOI] [PubMed] [Google Scholar]
- 81.Banuett F., Herskowitz I. Identification of fuz7, a Ustilago maydis MEK/MAPKK homolog required for a-locus-dependent and -independent steps in the fungal life cycle. Genes Devev. 1994b;8:1367–1378. doi: 10.1101/gad.8.12.1367. [DOI] [PubMed] [Google Scholar]
- 82.Mayorga M.E., Gold S.E. Characterization and molecular genetic complementation of mutants affecting dimorphism in the fungus Ustilago maydis. Fungal Genet. Biol. 1998;24:364–376. doi: 10.1006/fgbi.1998.1078. [DOI] [PubMed] [Google Scholar]
- 83.Mayorga M.E., Gold S.E. A MAP kinase encoded by the ubc3 gene of Ustilago maydis is required for filamentous growth and full virulence. Mol. Microbiol. 1999;34:485–497. doi: 10.1046/j.1365-2958.1999.01610.x. [DOI] [PubMed] [Google Scholar]
- 84.Andrews D.L., Egan J.D., Mayorga M.E., Gold S.E. The Ustilago maydis ubc4 and ubc5 genes encode members of a MAP Kinase cascade required for filamentous growth. Mol. Plant. Microbe Interact. 2000;13:781–786. doi: 10.1094/MPMI.2000.13.7.781. [DOI] [PubMed] [Google Scholar]
- 85.Müller P., Aichinger C., Feldbrügge M., Kahmann R. The MAP Kinase kpp2 regulates mating and pathogenic development in Ustilago maydis. Mol. Microbiol. 1999;34:1007–1017. doi: 10.1046/j.1365-2958.1999.01661.x. [DOI] [PubMed] [Google Scholar]
- 86.Müller P., Weinzierl G., Brachmann A., Feldbrügge M., Kahmann R. Mating and pathogenic development of the smut fungus Ustilago maydis are regulated by one mitogen-activated protein kinase cascade. Eukaryot. Cell. 2003;2:1187–1199. doi: 10.1128/EC.2.6.1187-1199.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Deng Y.Z., Zhang B., Chang C., Wang Y., Lu S., Sun S., Zhang X., Chen B., Jiang Z. The MAP Kinase SsKpp2 is required for mating/filamentation in Sporisorium scitamineum. Front. Microbiol. 2018;9:2555. doi: 10.3389/fmicb.2018.02555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Zhang Y., Ge Q., Cao Q., Cui H., Hu P., Yu X., Ye Z. Cloning and characterization of two mapk genes Uekpp2 and Uekpp6 in Ustilago esculenta. Curr. Microbiol. 2018;75:1016–1024. doi: 10.1007/s00284-018-1483-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Chen H., Fink G.R. Feedback control of morphogenesis in fungi by aromatic alcohols. Genes Dev. 2006;20:1150–1161. doi: 10.1101/gad.1411806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Wongsuk T., Pumeesat P., Luplertlop N. Fungal quorum sensingmolecules: Role in fungal morphogenesis and pathogenicity. J. Basic. Microbiol. 2016;56:440–447. doi: 10.1002/jobm.201500759. [DOI] [PubMed] [Google Scholar]
- 91.Mayorga M.E., Gold S.E. The ubc2 gene of Ustilago maydis encodes a putative novel adaptor protein required for filamentous growth, pheromone response and virulence. Mol. Microbiol. 2001;41:1365–1379. doi: 10.1046/j.1365-2958.2001.02606.x. [DOI] [PubMed] [Google Scholar]
- 92.Müller P., Katzenberger J.D., Loubradou G., Kahmann R. Guanyl nucleotide exchange factor Sql2 and Ras2 regulate filamentous growth in Ustilago maydis. Eukaryot. Cell. 2003;2:609–617. doi: 10.1128/EC.2.3.609-617.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Lee N., Kronstad J.W. Ras2 controls morphogenesis, pheromone response, and pathogenicity in the fungal pathogen Ustilago maydis. Eukaryot. Cell. 2002;1:954–966. doi: 10.1128/EC.1.6.954-966.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Mösch H.U., Roberts R.L., Fink G.R. Ras2 signals via the cdc42/ste20/mitogen-activated protein kinase module to induce filamentous growth in Saccharomyces cerevisiae. Proc. Natl. Acad. Sci. USA. 1996;93:5352–5356. doi: 10.1073/pnas.93.11.5352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Garrido E., Pérez-Martín J. The crk1 gene encodes an Ime2-related protein that is required for morphogenesis in the plant pathogen Ustilago maydis. Mol. Microbiol. 2003;47:729–743. doi: 10.1046/j.1365-2958.2003.03323.x. [DOI] [PubMed] [Google Scholar]
- 96.Cheng C.K., Au C.H., Wilke S.K., Stajich J.E., Miriam E., Zolan M.E., Pukkila P.J., Kwan H.S. 5′-Serial analysis of gene expression studies reveal a transcriptomic switch during fruiting body development in Coprinopsis Cinerea. BMC Genomics. 2013;14:195. doi: 10.1186/1471-2164-14-195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Gilman A.G. G proteins and dual control of adenylate cyclase. Cell. 1984;36:577–579. doi: 10.1016/0092-8674(84)90336-2. [DOI] [PubMed] [Google Scholar]
- 98.Taylor S.S., Juechler J.A., Yonemoto W. cAMP-dependent protein kinase: Framework for a diverse family of regulatory enzymes. Annu. Rev. Biochem. 1990;59:971–1005. doi: 10.1146/annurev.bi.59.070190.004543. [DOI] [PubMed] [Google Scholar]
- 99.Levitzki A., Bar-Sinai A. The regulation of Adenylyl cyclase by receptor-perated G proteins. Pharmacol. Ther. 1991;50:271–283. doi: 10.1016/0163-7258(91)90045-N. [DOI] [PubMed] [Google Scholar]
- 100.Thevelein J.M., Cauwenberg L., Colombo S., De Winde J.H., Donation M., Dumortier F., Kraakman L., Lemaire K., Ma P., Nauwelaers D., et al. Nutrient-induced signal transduction through the protein kinase a pathway and its role in the control of metabolism, stress resistance, and growth in yeast. Enzyme Microb. Technol. 2000;26:819–825. doi: 10.1016/S0141-0229(00)00177-0. [DOI] [PubMed] [Google Scholar]
- 101.Caza M., Kronstad J.W. The cAMP/protein kinase a pathway regulates virulence and adaptation to host conditions in Cryptococcus neoformans. Front. Cell Infect. Microbiol. 2019;9:212. doi: 10.3389/fcimb.2019.00212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Sabie F.T., Gadd G.M. Effect of Nucleosides and Nucleotides and the relationship between cellular adenosine 3′:5′-cyclic monophosphate (Cyclic AMP) and germ tube formation in Candida albicans. Mycopathologia. 1992;119:147–156. doi: 10.1007/BF00448812. [DOI] [PubMed] [Google Scholar]
- 103.Lichter A., Mills D. Fil1, a G-protein alpha-subunit that acts upstream of camp and is essential for dimorphic switching in haploid cells of Ustilago hordei. Mol. Gen. Genet. 1997;256:26–35. doi: 10.1007/s004380050586. [DOI] [PubMed] [Google Scholar]
- 104.Chang C., Enping Cai E., Deng Y.Z., Mei D., Qiu S., Chen B., Zhang L.H., Jiang Z. cAMP/PKA signalling pathway regulates redox homeostasis essential for Sporisorium scitamineum mating/filamentation and virulence. Environ. Microbiol. 2019;21:959–971. doi: 10.1111/1462-2920.14496. [DOI] [PubMed] [Google Scholar]
- 105.Wang Y., Deng Y.Z., Cui G., Huang C., Zhang B., Chang C., Jiang Z., Zhang L.H. The AGC Kinase SsAgc1 regulates Sporisorium scitamineum mating/filamentation and pathogenicity. mSphere. 2019;4:e00259-19. doi: 10.1128/mSphere.00259-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Dürrenberger F., Laidlaw R.D., Kronstad J.W. The hgl1 gene is required for dimorphism and teliospore formation in the fungal pathogen Ustilago maydis. Mol. Microbiol. 2001;41:337–348. doi: 10.1046/j.1365-2958.2001.02528.x. [DOI] [PubMed] [Google Scholar]
- 107.Loubradou G., Brachmann A., Feldbrugge M., Kahmann R. A homologue of the transcriptional repressor Ssn6p antagonizes cAMP signalling in Ustilago maydis. Mol. Microbiol. 2001;40:719–730. doi: 10.1046/j.1365-2958.2001.02424.x. [DOI] [PubMed] [Google Scholar]
- 108.Schwalb M.N. Regulation of fruiting. In: Schwalb M.N., Miles P.G., editors. Genetics and Morphogenesis in the Basidiomicetes. Academic Press; New York, NY, USA: 1978. pp. 135–165. [Google Scholar]
- 109.Swamy S., Uno I., Ishikawa T. Regulation of cyclic AMP-dependent phosphorylation of cellular proteins by the incompatibility factors in Coprinus cinereus. J. Gen. Appl. Microbiol. 1985;31:339–346. doi: 10.2323/jgam.31.339. [DOI] [Google Scholar]
- 110.Swamy S., Uno I., Ishikawa T. Regulation of cyclic AMP metabolism by the incompatibility factors in Coprinus cinereus. J. Gen. Microbiol. 1985;131:3211–3217. doi: 10.1099/00221287-131-12-3211. [DOI] [Google Scholar]
- 111.Lu Y., Lian L., Guo L., Xie B., Wang W., Chen B., van Peer A.F., Li S., Wu T., Xie B. The accordant trend of both parameters (rgs expression and cAMP content) follows the pattern of development of fruiting body in Volvariella volvácea. Curr. Microbiol. 2015;71:579–784. doi: 10.1007/s00284-015-0885-8. [DOI] [PubMed] [Google Scholar]
- 112.Tabor C.W., Tabor H. Polyamines. Annu. Rev. Biochem. 1984;53:749–790. doi: 10.1146/annurev.bi.53.070184.003533. [DOI] [PubMed] [Google Scholar]
- 113.D’Agostino L., Di Pietro M., Di Luccia A. Nuclear aggregates of polyamines. IUBMB Life. 2006;58:75–82. doi: 10.1080/15216540600662525. [DOI] [PubMed] [Google Scholar]
- 114.Igarashi K., Kashiwagi K. Modulation of cellular function by polyamines. Int. J. Biochem. Cell B. 2010;42:39–51. doi: 10.1016/j.biocel.2009.07.009. [DOI] [PubMed] [Google Scholar]
- 115.Rajam M.V., Galston A.W. The effects of some polyamine biosynthetic inhibitors on growth and morphology of phytopathogenic fungi. Plant. Cell Physiol. 1985;26:683–692. doi: 10.1093/oxfordjournals.pcp.a076958. [DOI] [PubMed] [Google Scholar]
- 116.Ruiz-Herrera J. Polyamines, DNA methylation, and fungal differentiation. Crit. Rev. Microbiol. 1994;20:143–150. doi: 10.3109/10408419409113553. [DOI] [PubMed] [Google Scholar]
- 117.Khurana N., Saxena R.K., Gupta R., Rajam M.V. Polyamines as modulators of microcycle conidiation in Aspergillus flavus. Microbiology. 1996;142:517–523. doi: 10.1099/13500872-142-3-517. [DOI] [PubMed] [Google Scholar]
- 118.Mueller E., Bailey A., Corran A., Michael A.J., Bowyer P. Ornithine decarboxylase knockout in Tapesia yallundae abolishes infection plaque formation in vitro but does not reduce virulence toward wheat. Mol. Plant. Microbe Interact. 2001;14:1303–1311. doi: 10.1094/MPMI.2001.14.11.1303. [DOI] [PubMed] [Google Scholar]
- 119.Pal M., Janda T. Role of polyamine metabolism in plant pathogen interactions. J. Plant. Sci. Phytopathol. 2017;1:95–100. doi: 10.29328/journal.jpsp.1001012. [DOI] [Google Scholar]
- 120.Nickerson K.W., Dunkle L.D., Van Etten J.L. Absence of spermine in filamentous fungi. J. Bacteriol. 1977;129:173–176. doi: 10.1128/JB.129.1.173-176.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.León-Ramírez C.G., Valdés-Santiago L., Campos-Góngora E., Ortiz-Castellanos L., Aréchiga-Carvajal E.T., Ruiz-Herrera J. A molecular probe for Basidiomycota: The spermidine synthase-saccharopine dehydrogenase chimeric gene. FEMS Microbiol. Lett. 2010;312:77–83. doi: 10.1111/j.1574-6968.2010.02099.x. [DOI] [PubMed] [Google Scholar]
- 122.Rocha R.O., Wilson R.A. Essential, deadly, enigmatic: Polyamine metabolism and roles in fungal cells. Fungal. Biol. Rev. 2019;33:47–57. doi: 10.1016/j.fbr.2018.07.003. [DOI] [Google Scholar]
- 123.Martínez-Soto D., Ruiz-Herrera J. Transcriptomic analysis of the dimorphic transition of Ustilago maydis induced in vitro by a change in pH. Fungal Genet. Biol. 2013;58–59:116–125. doi: 10.1016/j.fgb.2013.08.011. [DOI] [PubMed] [Google Scholar]
- 124.Kou M.H., Allis C.D. Roles of histone acetyltransferases and deacetylases in gene regulation. BioEssays. 1998;20:615–626. doi: 10.1002/(SICI)1521-1878(199808)20:83.0.CO;2-H. [DOI] [PubMed] [Google Scholar]
- 125.Marmorstein R., Roth S.Y. Histone acetyltransferases: Function, structure, and catalysis. Curr. Opin. Genet. Dev. 2001;11:155–161. doi: 10.1016/S0959-437X(00)00173-8. [DOI] [PubMed] [Google Scholar]
- 126.Legube G., Trouche D. Regulating histone acetyltransferases and deacetylases. EMBO Rep. 2003;4:944–947. doi: 10.1038/sj.embor.embor941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Thiagalingam S., Cheng K.H., Lee H.J., Mineva N., Thiagalingam A., Ponte J.F. Histone deacetylases: Unique players in shaping the epigenetic histone code. Ann. N. Y. Acad. Sci. 2003;983:84–100. doi: 10.1111/j.1749-6632.2003.tb05964.x. [DOI] [PubMed] [Google Scholar]
- 128.Lee K.K., Workman J.L. Histone acetyltransferase complexes: One size doesn’t fit all. Nat. Rev. Mol. Cell Biol. 2007;8:284–295. doi: 10.1038/nrm2145. [DOI] [PubMed] [Google Scholar]
- 129.Liu X., Yang S., Zhao M., Lou M., Yu C.M., Chen C.Y., Tai R., Wu K. Transcriptional repression by histone deacetylases in plants. Mol. Plant. 2014;7:764–772. doi: 10.1093/mp/ssu033. [DOI] [PubMed] [Google Scholar]
- 130.Reyna-López G.E., Simpson J., Ruiz-Herrera J. Differences in DNA methylation patterns are detectable during the dimorphic transition of fungi by amplification of restriction polymorphisms. Mol. Gen. Genet. 1997;253:703–710. doi: 10.1007/s004380050374. [DOI] [PubMed] [Google Scholar]
- 131.Reyna-López G.E., Ruiz-Herrera J. Specificity of DNA methylation changes during fungal dimorphism and its relationship to polyamines. Curr. Microbiol. 2004;48:118–123. doi: 10.1007/s00284-003-4142-1. [DOI] [PubMed] [Google Scholar]
- 132.Kusano T., Berberich T., Tateda C., Takahashi Y. Polyamines: Essential factors for growth and survival. Planta. 2008;228:367–381. doi: 10.1007/s00425-008-0772-7. [DOI] [PubMed] [Google Scholar]
- 133.He C., Zhang Z., Li B., Tian S. The pattern and function of DNA methylation in fungal plant pathogens. Microorganisms. 2020;8:227. doi: 10.3390/microorganisms8020227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Ruiz-Herrera J., Pérez-Rodríguez F., Velez-Haro J. The signaling mechanisms involved in the dimorphic phenomenon of the Basidiomycota fungus Ustilago maydis. Int. Microbiol. 2020;23:121–126. doi: 10.1007/s10123-019-00100-5. [DOI] [PubMed] [Google Scholar]
- 135.Ruiz-Herrera J. Fungal Cell Wall Structure, Synthesis, and Assembly. 2nd ed. CRC Press; Boca Raton, FL, USA: 2012. p. 168. [Google Scholar]
- 136.Ruiz-Herrera J., Elorza M.V., Álvarez P.E., Sentandreu R. Biosynthesis of the fungal cell wall. In: San-Blas G., Calderone R.A., editors. Pathogenic Fungi, Structural, Biology and Taxonomy. Caister Academic Press; Wymondham, UK: 2004. pp. 41–99. [Google Scholar]
- 137.Sentandreu R., Elorza M.V., Valentín E., Ruiz-Herrera J. The structure and composition of fungal cell wall. In: San-Blas G., Calderone R.A., editors. Pathogenic Fungi, Structural, Biology and Taxonomy. Caister Academic Press; Wymondham, UK: 2004. pp. 3–27. [Google Scholar]
- 138.Cabib E., Sburlati A., Bowers B., Silverman S.J. Chitin synthase 1, an auxiliary enzyme for chitin synthesis in Saccharomyces cerevisiae. J. Cell Biol. 1989;108:1665–1672. doi: 10.1083/jcb.108.5.1665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Silverman S.J., Sburlati A., Slater M.L., Cabib E. Chitin synthase 2 is essential for septum formation and cell division in Saccharomyces cerevisiae. Proc. Natl. Acad. Sci. USA. 1988;85:4735–4739. doi: 10.1073/pnas.85.13.4735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Shaw J.A., Mol P.C., Bowers B., Silverman S.J., Valdivieso M.H., Durán A., Cabib E. The function of chitin synthases 2 and 3 in the Saccharomyces cerevisiae cell cycle. J. Cell Biol. 1991;114:111–123. doi: 10.1083/jcb.114.1.111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Yarden O., Yanofsky C. chitin synthase 1 plays a major role in cell wall biogenesis in Neurospora crassa. Genes Dev. 1991;5:2420–2430. doi: 10.1101/gad.5.12b.2420. [DOI] [PubMed] [Google Scholar]
- 142.Borgia P.-T., Iartchouk N., Riggle P.J., Winter K.R., Koltin Y., Bulawa C.E. The chsB gene of Aspergillus nidulans is necessary for normal hyphal growth and development. Fungal Genet. Biol. 1996;20:193–203. doi: 10.1006/fgbi.1996.0035. [DOI] [PubMed] [Google Scholar]
- 143.Garcerá-Teruel A., Xoconoxtle-Cázares B., Rosas-Quijano R., Ortíz L., León-Ramírez C., Specht C.A., Sentandreu R., Ruiz Herrera J. Loss of virulence in Ustilago maydis by Umchs6 gene disruption. Res. Microbiol. 2004;155:87–97. doi: 10.1016/j.resmic.2003.11.005. [DOI] [PubMed] [Google Scholar]
- 144.Weber I., Assmann D., Thines E., Steinberg G. Polar localizing class V myosin chitin synthases are essential during early plant infection in the plant pathogenic fungus Ustilago maydis. Plant. Cell. 2006;18:225–242. doi: 10.1105/tpc.105.037341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.López-Matas M.A., Eslava A.P., Díaz-Minguez J.M. Mcchs1, a member of a chitin synthase gene family in Mucor circinelloides, is differentially expressed during dimorphism. Curr. Microbiol. 2000;40:169–175. doi: 10.1007/s002849910034. [DOI] [PubMed] [Google Scholar]
- 146.Niño-Vega G.A., Munro C.A., San-Blas G., Gooday G.W., Gow N.A. Differential expression of chitin synthase genes during temperature-induced dimorphic transitions in Paracoccidioides Brasiliensis. Med. Mycol. 2000;38:31–39. doi: 10.1080/mmy.38.1.31.39. [DOI] [PubMed] [Google Scholar]
- 147.Christodoulidou A., Bouriotis V., Thireos G. Two sporulation-specific chitin deacetylase-encoding genes are required for the ascospore wall rigidity of Saccharomyces cerevisiae. J. Biol. Chem. 1996;271:31420–31425. doi: 10.1074/jbc.271.49.31420. [DOI] [PubMed] [Google Scholar]
- 148.Geoghegan I.A., Gurr S.J. Investigating chitin deacetylation and chitosan hydrolysis during vegetative growth in Magnaporthe oryzae. Cell Microbiol. 2017;19:e12743. doi: 10.1111/cmi.12743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Ruiz-Herrera J., Robledo-Briones M., Martínez-Soto D. Experimental pathosystems as a tool for the identification of virulence factors in pathogenic fungi. In: Deshpande M.V., Ruiz-Herrera J., editors. Biotechnology: Beyond Borders. CSIR-National Chemical Laboratory; Pune, India: 2013. pp. 30–38. [Google Scholar]
- 150.Tentler S., Palas J., Enderlin C., Campbell J., Taft C., Miller T.K., Wood R.L., Selitrennikoff C.P. Inhibition of Neurospora crassa growth by a glucan synthase-1 antisense construct. Curr. Microbiol. 1997;34:303–308. doi: 10.1007/s002849900186. [DOI] [PubMed] [Google Scholar]
- 151.Verdín J., Bartnicki-Garcia S., Riquelme M. Functional stratification of the Spitzenkörper of Neurospora crassa. Mol. Microbiol. 2009;74:1044–1053. doi: 10.1111/j.1365-2958.2009.06917.x. [DOI] [PubMed] [Google Scholar]
- 152.Kuranda M.J., Robbins P.W. Chitinase is required for cell separation during growth of Saccharomyces cerevisiae. J. Biol. Chem. 1991;266:19758–19767. [PubMed] [Google Scholar]
- 153.Colman-Lerner A., Chin T.E., Brent R. Yeast Cbk1 and Mob2 activate daughter-specific genetic programs to induce asymmetric cell fates. Cell. 2001;107:739–750. doi: 10.1016/S0092-8674(01)00596-7. [DOI] [PubMed] [Google Scholar]
- 154.Dünkler A., Walther A., Specht C.A., Wendland J. Candida albicans Cht3 encodes the functional homolog of the cts1 chitinase of Saccharomyces cerevisiae. Fungal Genet. Biol. 2005;42:935–947. doi: 10.1016/j.fgb.2005.08.001. [DOI] [PubMed] [Google Scholar]
- 155.Tzelepis G.D., Melin P., Jensen D.F., Stenlid J., Magnus Karlsson M. Functional analysis of glycoside hydrolase family 18 and 20 genes in Neurospora crassa. Fungal Genet. Biol. 2012;49:717–730. doi: 10.1016/j.fgb.2012.06.013. [DOI] [PubMed] [Google Scholar]
- 156.Baladrón V., Ufano S., Dueñas E., Martín-Cuadrado A.B., del Rey F., Vázquez de Aldana C.R. Eng1p, an endo-1,3-beta-glucanase localized at the daughter side of the septum, is involved in cell separation in Saccharomyces cerevisiae. Eukaryot. Cell. 2002;1:774–786. doi: 10.1128/EC.1.5.774-786.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Riquelme M., Aguirre J., Bartnicki-García S., Braus G.H., Feldbrügge M., Fleig U., Hansberg W., Herrera-Estrella A., Kämper J., Kück U., et al. Fungal morphogenesis, from the polarized growth of hyphae to complex reproduction and infection structures. Microbiol. Mol. Biol. Rev. 2018;82:e00068-17. doi: 10.1128/MMBR.00068-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Banuett F., Herskowitz I. Bud morphogenesis and the actin and microtubule cytoskeletons during budding in the corn smut fungus, Ustilago maydis. Fungal Genet. Biol. 2002;37:149–170. doi: 10.1016/S1087-1845(02)00548-0. [DOI] [PubMed] [Google Scholar]
- 159.Valinluck M., Ahlgren S., Sawada M., Locken K., Banuett F. Role of the nuclear migration protein Lis1 in cell morphogenesis in Ustilago maydis. Mycologia. 2010;102:493–512. doi: 10.3852/09-193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Valinluck M., Woraratanadharm T., Lu C.Y., Quintanilla R.H., Jr., Bannuett F. The cell end marker Tea4 regulates morphogenesis and pathogenicity in the basidiomycete fungus Ustilago maydis. Fungal Genet. Biol. 2014;66:54–68. doi: 10.1016/j.fgb.2014.02.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Chew E., Aweiss Y., Lu C.Y., Banuett F. Fuz1, a MYND domain protein, is required for cell morphogenesis in Ustilago maydis. Mycologia. 2008;100:31–46. doi: 10.1080/15572536.2008.11832497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Schuchardt I., Assmann D., Thines E., Schuberth C., Steinberg G. Myosin-V, Kinesin-1, and Kinesin-3 cooperate in hyphal growth of the fungus Ustilago maydis. Mol. Biol. Cell. 2005;16:5191–5201. doi: 10.1091/mbc.e05-04-0272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Weber I., Gruber C., Steinberg G. A class V myosin required for mating, hyphal growth, and pathogenicity in the dimorphic plant pathogen Ustilago maydis. Plant. Cell. 2003;15:2826–2842. doi: 10.1105/tpc.016246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Woraratanadharm T., Kmosek S., Banuett F. UmTea1, a Kelch and BAR domain-containing protein, acts at the cell cortex to regulate cell morphogenesis in the dimorphic fungus Ustilago maydis. Fungal Genet. Biol. 2018;121:10–28. doi: 10.1016/j.fgb.2018.09.002. [DOI] [PubMed] [Google Scholar]
- 165.Wösten H.A., Bohlmann R., Eckerskorn C., Lottspeich F., Bölker M., Kahmann R. A novel class of small amphipathic peptides affect aerial hyphal growth and surface hydrophobicity in Ustilago maydis. EMBO J. 1996;15:4272–4281. doi: 10.1002/j.1460-2075.1996.tb00802.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Teertstra W.R., Deelstra H.J., Vranes M., Bohlmann R., Kahmann R., Kämper J., Wösten H.A.B. Repellents have functionally replaced hydrophobins in mediating attachment to a hydrophobic surface and in formation of hydrophobic aerial hyphae in Ustilago maydis. Microbiology. 2006;152:3607–3612. doi: 10.1099/mic.0.29034-0. [DOI] [PubMed] [Google Scholar]
- 167.Soberanes-Gutiérrez C.V., Juárez-Montiel M., Olguín-Rodríguez O., Hernández-Rodríguez C., Ruiz-Herrera J., Villa-Tanaca L. The Pep4 gene encoding proteinase A is involved in dimorphism and pathogenesis of Ustilago maydis. Mol. Plant. Pathol. 2015;16:837–846. doi: 10.1111/mpp.12240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Pham C.D., Yu Z., Sandrock B., Bölker M., Gold S.E., Perlin M.H. Ustilago maydis Rho1 and 14-3-3 homologues participate in pathways controlling cell separation and cell polarity. Eukaryot. Cell. 2009;8:977–989. doi: 10.1128/EC.00009-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Frieser S.H., Hlubek A., Sandrock B., Bölker M. Cla4 kinase triggers destruction of the Rac1-GEF Cdc24 during polarized growth in Ustilago maydis. Mol. Biol. Cell. 2011;22:3253–3262. doi: 10.1091/mbc.e11-04-0314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.König J., Baumann S., Koepke J., Pohlmann T., Zarnack K., Feldbrügge M. The fungal RNA-binding protein Rrm4 mediates long-distance transport of ubi1 and rho3 mRNAs. EMBO J. 2009;28:1855–1866. doi: 10.1038/emboj.2009.145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Smith D.G., Garcia-Pedrajas M.D., Gold S.E., Perlin M.H. Isolation and characterization from pathogenic fungi of genes encoding ammonium permeases and their roles in dimorphism. Mol. Microbiol. 2003;50:259–275. doi: 10.1046/j.1365-2958.2003.03680.x. [DOI] [PubMed] [Google Scholar]
- 172.Zhong Y., Yan M., Jiang J., Zhang Z., Huang J., Zhang L., Deng Y., Xiaofan Zhou X., He F. Mycophenolic acid as a promising fungal dimorphism inhibitor to control sugar cane disease caused by Sporisorium scitamineum. J. Agric. Food Chem. 2019;67:112–119. doi: 10.1021/acs.jafc.8b04893. [DOI] [PubMed] [Google Scholar]
- 173.Zhang B., Cui G.B., Chang C.Q., Wang Y.X., Zhang H.Y., Chen B.S., Deng Y.Z., Jiang Z.D. The autophagy gene ATG8 affects morphogenesis and oxidative stress tolerance in Sporisorium scitamineum. J. Integr. Agric. 2019;18:1024–1034. doi: 10.1016/S2095-3119(18)62109-4. [DOI] [Google Scholar]
- 174.Zhang Y., Wu M., Ge Q., Yang M., Xia W., Cui H., Yu X., Zhang S., Ye Z. Cloning and disruption of the UeArginase in Ustilago esculenta: Evidence for a role of arginine in its dimorphic transition. BMC Microbiol. 2019;19:208. doi: 10.1186/s12866-019-1588-2. [DOI] [PMC free article] [PubMed] [Google Scholar]