Abstract
Background
In cluster randomized crossover (CRXO) trials, groups of participants (i.e., clusters) are randomly allocated to receive a sequence of interventions over time (i.e., cluster periods). CRXO trials are becoming more comment when they are feasible, as they require fewer clusters than parallel group cluster randomized trials. However, CRXO trials have not been frequently used in orthopedic fracture trials and represent a novel methodological application within the field. To disseminate the early knowledge gained from our experience initiating two cluster randomized crossover trials, we describe our process for the identification and selection of the orthopedic practices (i.e., clusters) participating in the PREP-IT program and present data to describe their key characteristics.
Methods
The PREP-IT program comprises two ongoing pragmatic cluster randomized crossover trials (Aqueous-PREP and PREPARE) which compare the effect of iodophor versus chlorhexidine solutions on surgical site infection and unplanned fracture-related reoperations in patients undergoing operative fracture management. We describe the process we used to identify and select orthopedic practices (clusters) for the PREP-IT trials, along with their characteristics.
Results
We identified 58 potential orthopedic practices for inclusion in the PREP-IT trials. After screening each practice for eligibility, we selected 30 practices for participation and randomized each to a sequence of interventions (15 for Aqueous-PREP and 20 for PREPARE). The majority of orthopedic practices included in the Aqueous-PREP and PREPARE trials were situated in level I trauma centers (100% and 87%, respectively). Orthopedic practices in the Aqueous-PREP trial operatively treated a median of 149 open fracture patients per year, included a median of 11 orthopedic surgeons, and had access to a median of 5 infection preventionists. Orthopedic practices in the PREPARE trial treated a median of 142 open fracture and 1090 closed fracture patients per year, included a median of 7.5 orthopedic surgeons, and had access to a median of 6 infection preventionists.
Conclusions
The PREP-IT trials provide an example of how to follow the reporting standards for cluster randomized crossover trials by providing a clear definition of the cluster unit, a thorough description of the cluster identification and selection process, and sufficient description of key cluster characteristics.
Trial registration
Both trials are registered at ClinicalTrials.gov (A-PREP: NCT03385304 December 28, 2017, and PREPARE: NCT03523962 May 14, 2018).
Keywords: Cluster, Randomized crossover, Pragmatic, Cluster characteristics, Orthopedic, Surgical site infection
Background
In clinical research, large high-quality randomized controlled trials (RCTs) are considered the highest level of evidence to determine the effectiveness of an intervention [1]. While most RCTs follow a parallel group design and randomize individual participants to one or more intervention and control groups, there are certain situations in which randomization at this level is not feasible or practical [2]. Cluster randomized trials overcome these challenges by randomizing predetermined groups of participants (i.e., clusters) to interventions [3]. In cluster randomized crossover (CRXO) trials, each cluster serves as their own control group by participating in both the treatment and control arm at least once at various periods of time throughout the trial. An advantage to this design [CRXO] over the parallel group cluster randomized trial is that fewer clusters are required to achieve statistical power.
Researchers undertaking CRXO trials must ensure that they take into consideration multiple differences in the conduct and reporting between RCTs and CRXO trials [4, 5]. One of these fundamental differences is the requirement to provide information on the characteristics and flow of clusters throughout the conduct of a CRXO trial (i.e., enrolment, allocation, follow-up, analysis), as described in the CONSORT extension for cluster randomized trials [4]. This is accomplished by including a priori cluster selection criteria (similar to participant eligibility criteria) and a cluster flow diagram that shows the cluster selection and flow over the duration of the trial (similar to the patient flow diagram). This information enables knowledge users to assess the generalizability of the trial and determine if the results are applicable to their setting and practice.
To disseminate the early knowledge gained from our experience initiating two large infection prevention CRXO trials in orthopedic fracture patients, we describe our cluster identification and selection process. Additionally, we present data on the key characteristics of the clusters to illustrate the level of data collection that is required for appropriate reporting in CRXO trials.
Methods
The PREP-IT program
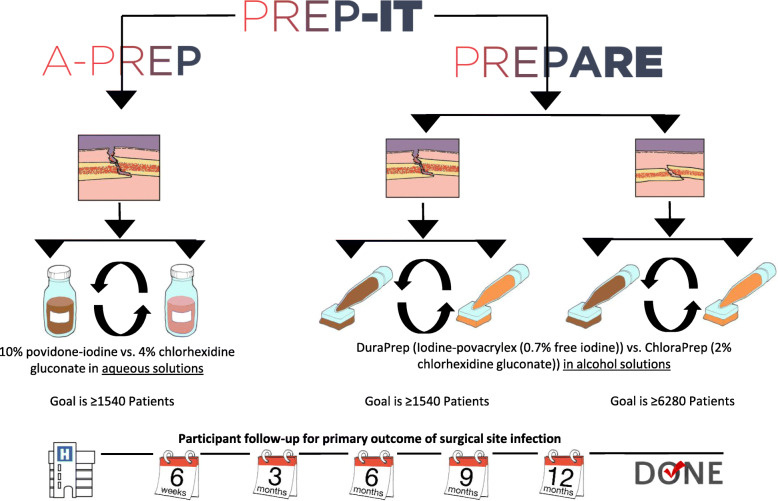
The Program of Randomized trials to Evaluate Pre-operative antiseptic skin solutions in orthopaedic Trauma (PREP-IT) aims to determine the effectiveness of iodophor compared to chlorhexidine solutions at reducing surgical site infection (SSI) and unplanned fracture-related reoperations in fracture surgery patients. The PREP-IT program includes two ongoing multi-center pragmatic CRXO trials that study the effects of four antiseptic solutions in three independent populations of surgically treated fracture patients: the Aqueous-PREP (A Pragmatic Randomized trial Evaluating Pre-operative aqueous antiseptic skin solutions in open fractures) trial and the PREPARE (A Pragmatic Randomized trial Evaluating Pre-operative Alcohol skin solutions in Fractured Extremities) trial (Fig. 1). The Aqueous-PREP trial compares 4% aqueous chlorhexidine versus 10% povidone-iodine in open extremity fracture patients. The PREPARE trial compares 2% chlorhexidine in 70% isopropyl alcohol (ChloraPrep™) versus 0.7% iodine povacrylex in 74% isopropyl alcohol (DuraPrep™) in both open extremity fracture patients and patients with closed lower extremity or pelvic fractures. Both trials follow a single master protocol which has been described in a previous manuscript [6].
Fig. 1.
The PREP-IT research program
Briefly, the order of treatment allocation for each orthopedic practice (i.e., cluster) was randomly assigned using a computer-generated randomization table. Simple, non-stratified randomization was used. Cluster randomization is performed centrally at the Methods Centre by personnel who are unaware of the cluster characteristics. Randomization is performed when a cluster has received local ethics approval and their clinical trial agreement is in place.
Each orthopedic practice starts with the surgical preparation solution to which they were initially allocated and subsequently crosses over to the other solution for their second cluster period. This process of alternating treatments repeats approximately every 2 months as dictated by the initial randomization. This process of alternating treatment periods (crossovers) will continue until the minimum sample size is achieved for each fracture population and the study’s planned recruitment period is completed. Clinical sites must complete at least two treatment periods (one crossover), and the majority of clinical sites will complete 12 treatment periods (11 crossovers), and 24 months of enrollment.
For both trials, the primary outcome is SSI as defined by the Centers for Disease Control and Prevention (CDC) [7] which includes superficial incisional SSI within 30 days and deep incisional or organ/space SSI within 90 days of definitive fracture management surgery. We continue to follow participants for SSI for 12 months to inform our sensitivity analyses. The secondary outcome is unplanned fracture-related reoperations within 12 months to manage infection, wound healing problems, and fracture healing problems. The Aqueous-PREP trial will enroll a minimum of 1540 open fracture patients, and PREPARE will enroll a minimum of 1540 open fracture patients and 6280 closed fracture patients.
Cluster definition
We defined clusters as orthopedic practices within participating hospitals. Each participating hospital has only one participating orthopedic practice. Clustering occurs at the level of the orthopedic practice as only orthopedic surgeons were required to follow the study protocol and use the randomized surgical preparation solution. During the design phase, we considered defining clusters as either the hospitals with which orthopedic practices were affiliated or the operating rooms used by orthopedic practices; however, we decided against these definitions as other specialities associated with the hospital or operating rooms were not required to follow the study protocol.
Cluster eligibility criteria
To be eligible for inclusion in the PREP-IT trials, orthopedic practices were required to meet the predetermined eligibility criteria as specified in the trial protocols. Specifically, the inclusion criteria were as follows: (1) adequate research personnel infrastructure to manage the study, (2) adequate open fracture volume and closed lower extremity and pelvic fracture volume to complete enrollment within the study timeline (i.e., a minimum of 77 open fractures (for Aqueous-PREP and PREPARE) and 314 closed lower extremity fractures per year (for PREPARE only)), (3) commitment from all or most orthopedic surgeons to participate in the trial, and (4) ability to use the two skin preparation solutions. The exclusion criteria were as follows: (1) lack of interest in the trial; (2) anticipated challenges with complying with the protocol; (3) conflicting studies, in the judgment of the Principal Investigators, that would inhibit patient participation; (4) budgeting or contract constraints; and (5) conflicting roles within the trials (e.g., member of the Data and Safety Monitoring Board).
Cluster identification and selection
We identified orthopedic practices through professional contacts with the PREP-IT Principal Investigators. Each orthopedic practice received an email containing information about the PREP-IT trials, as well as an invitation to apply to participate in either one or both trials. For orthopedic practices who declined this invitation, we identified and documented the reason for exclusion using our predetermined eligibility criteria. Orthopedic practices who expressed a desire to participate were required to complete a feasibility questionnaire. This questionnaire consisted of 17 questions and assessed the orthopedic practice’s research experience and infrastructure, fracture volume, current practice patterns, and interest in participating in one or both trials. We used the responses to the feasibility questionnaire to preliminarily screen orthopedic practices for eligibility. Orthopedic practices that were found to be ineligible through this preliminary screen were notified, and the reason for exclusion was documented. Orthopedic practices that were eligible to participate following the preliminary screen were invited to participate in a series of teleconferences with the PREP-IT Principal Investigators and study personnel. The purpose of these calls was to review trial and clinical practice logistics in detail and to confirm whether each orthopedic practice met the eligibility criteria for participation. For any orthopedic practice that was determined to be ineligible through these calls, we documented the reason for exclusion. Orthopedic practices that met all eligibility criteria were selected to participate.
Collection of cluster characteristics
Upon selection, orthopedic practices were required to complete a cluster definition questionnaire. This questionnaire, which is comprised of 44 questions, captures important data on hospital characteristics, current surgeon preferences and practices for pre-operative surgical preparation techniques, and infection co-interventions known to reduce the incidence of SSIs. Each orthopedic practice’s questionnaire data is updated every 4 months over the enrollment and follow-up periods.
Statistical analyses
Our statistical analysis plan was determined a priori. We included data from the orthopedic practices currently enrolling participants into the Aqueous-PREP trial and the PREPARE trial. We used descriptive statistics to summarize all characteristics of the orthopedic practices (frequencies and percentages for categorical variables, and medians and interquartile ranges (IQRs) for continuous variables). We used Microsoft Excel 2016 to conduct all statistical analyses.
Results
Cluster selection
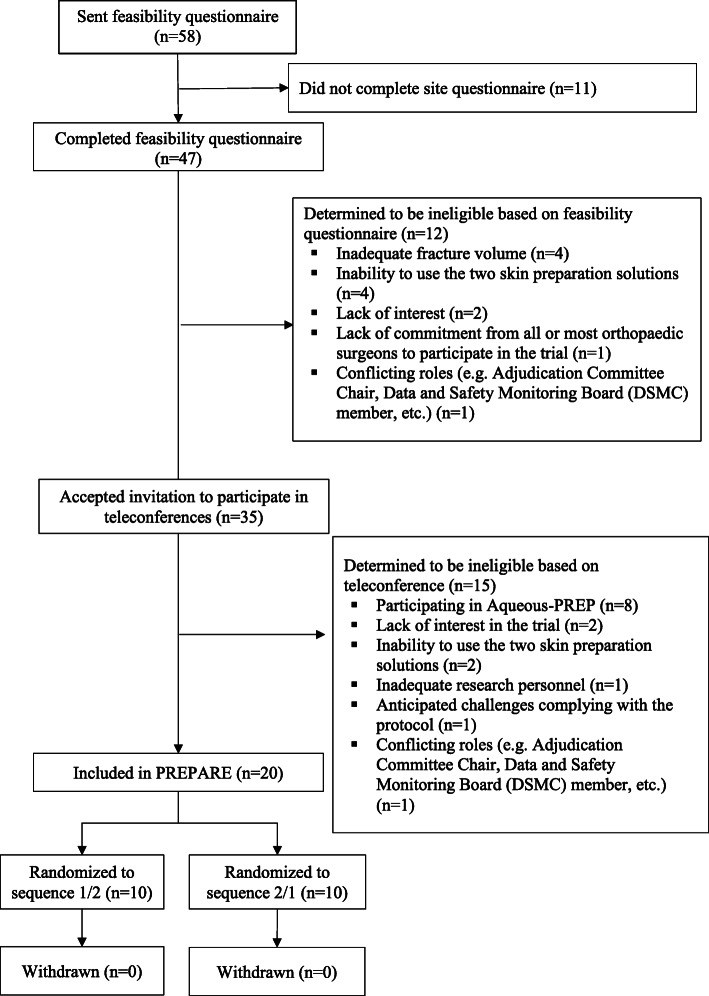
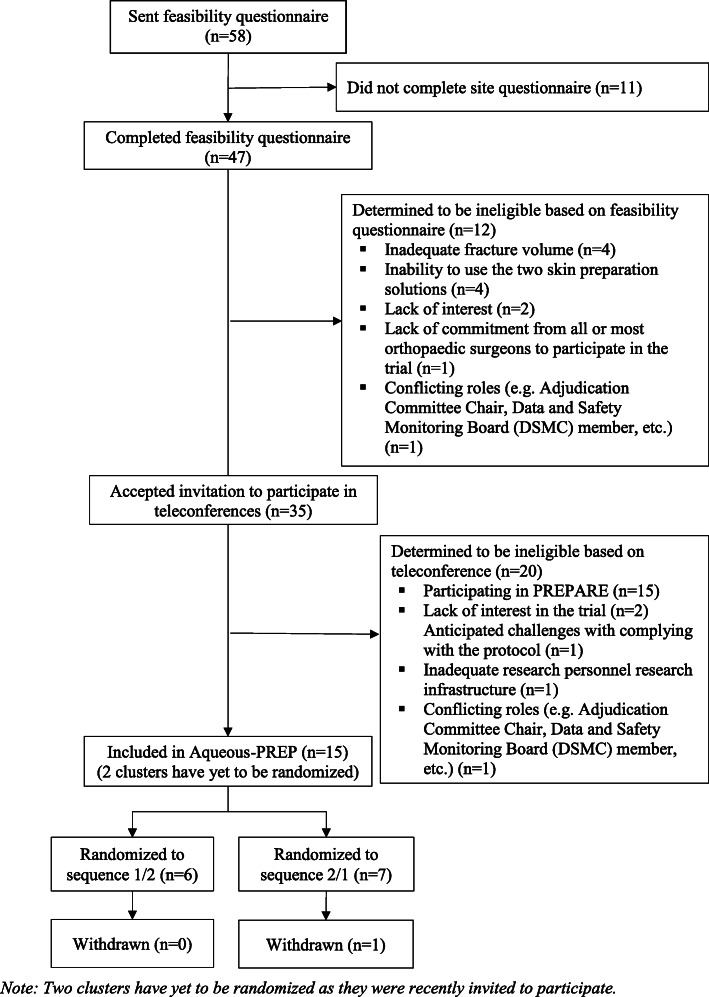
Of the 58 potentially eligible orthopedic practices that we identified, 47 (81%) accepted our invitation to complete a feasibility questionnaire and 11 (19%) declined our invitation and were excluded due to a lack of interest in the trial (Figs. 2 and 3). Of the 47 practices that completed the feasibility questionnaire, 12 were determined to be ineligible and 35 were invited to participate in a teleconference with the Principal Investigators and study team to further assess eligibility. Of the 35 orthopedic practices that participated in the teleconferences, 5 were determined to be ineligible, 5 were selected to participate in both the Aqueous-PREP and PREPARE trials, 10 in the Aqueous-PREP trial only, and 15 in the PREPARE trial only. Specific reasons for orthopedic practice exclusions for each trial are documented in Figs. 2 and 3. One of the practices participating in the Aqueous-PREP trial was discontinued post-randomization due to an inability to follow the trial protocol. Specifically, this cluster did not meet the a priori threshold of treatment compliance (e.g., 90% of patients receive the allocated antiseptic solution) and the clinical site was unable to complete the case report forms, which led to incomplete data submission. The clinical site enrolled 14 participants and was withdrawn within 2 months of initiation. Therefore, there are 14 orthopedic practices in the Aqueous-PREP trial and 20 orthopedic practices in the PREPARE trial.
Fig. 2.
PREPARE CONSORT flowchart for clusters
Fig. 3.
Aqueous-PREP CONSORT flowchart for clusters
Characteristics of participating clusters
Table 1 details the name and location of the hospital associated with each orthopedic practice participating in the Aqueous-PREP and PREPARE trials, as well as the intervention sequence to which they were randomized. The majority of orthopedic practices are affiliated with hospitals that are level I trauma centers and owned by a non-profit, non-religious organization (Table 2). The median population served is 1,625,000 for orthopedic practices participating in the Aqueous-PREP trial and 2,400,000 for the PREPARE trial. The characteristics of the withdrawn cluster are presented in the Appendix.
Table 1.
Clusters and location
| Hospital (cluster) name | Location | Randomization sequence (1/2 vs. 2/1) | Trial |
|---|---|---|---|
| University of Maryland School of Medicine, R Adams Cowley Shock Trauma Center | Baltimore, MD | 2/1 (Aqueous-PREP); 1/2 (PREPARE) | Aqueous-PREP and PREPARE |
| Greenville Health System | Greenville, SC | 2/1 (Aqueous-PREP); 2/1 (PREPARE) | Aqueous-PREP and PREPARE |
| Hamilton Health Sciences – General Site | Hamilton, ON | 1/2 (Aqueous-PREP); 2/1 (PREPARE) | Aqueous-PREP and PREPARE |
| IU Health Methodist Hospital | Indianapolis, IN | 1/2 (Aqueous-PREP); 1/2 (PREPARE) | Aqueous-PREP and PREPARE |
| San Antonio Military Medical Center | San Antonio, TX | 1/2 Aqueous-PREP; 1/2 (PREPARE) | Aqueous-PREP and PREPARE |
| Banner – University Medical Center Tucson | Tucson, AZ | 1/2 | Aqueous-PREP |
| Hospital Universitari Parc Tauli | Barcelona, Spain | To be randomized | Aqueous-PREP |
| McGovern Medical School at UTHealth Houston | Houston, TX | 1/2 | Aqueous-PREP |
| The CORE Institute | Phoenix, AZ | 2/1 | Aqueous-PREP |
| University of California, San Francisco | San Francisco, CA | 1/2 | Aqueous-PREP |
| University of Florida | Gainesville, FL | 2/1 | Aqueous-PREP |
| Vall d’Hebron Hospital | Barcelona, Spain | To be randomized | Aqueous-PREP |
| Vanderbilt Medical Center | Nashville, TN | 2/1 | Aqueous-PREP |
| Wright State University | Dayton, OH | 2/1 | Aqueous-PREP |
| Brigham and Women’s Hospital | Boston, MA | 1/2 | PREPARE |
| Carolina’s Medical Center | Charlotte, NC | 2/1 | PREPARE |
| Dartmouth-Hitchcock Medical Center | Lebanon, NH | 2/1 | PREPARE |
| Duke University Hospital | Durham, NC | 2/1 | PREPARE |
| Inova Fairfax Medical Campus | Fairfax, VA | 2/1 | PREPARE |
| Massachusetts General Hospital | Boston, MA | 1/2 | PREPARE |
| MetroHealth Medical Center | Cleveland, OH | 1/2 | PREPARE |
| Royal Columbian Hospital | New Westminster, BC | 2/1 | PREPARE |
| Regional Medical Center of San Jose | San Jose, CA | 1/2 | PREPARE |
| Sanford Health | Sioux Falls, SD | 1/2 | PREPARE |
| University of Maryland Capital Regional Health | Cheverly, MD | 2/1 | PREPARE |
| University of Mississippi Medical Center | Jackson, MS | 2/1 | PREPARE |
| University of Pennsylvania | Philadelphia, PA | 1/2 | PREPARE |
| University of Utah | Salt Lake City, UT | 2/1 | PREPARE |
| Wake Forest Baptist Hospital | Winston-Salem, NC | 1/2 | PREPARE |
Table 2.
Cluster characteristics
| Characteristic | Aqueous-PREP, N = 14* | PREPARE, N = 20 |
|---|---|---|
| Level of trauma care provided, n (%) | ||
| Level I | 14 (100) | 17 (85) |
| Level II | 0 (0) | 3 (15) |
| Ownership of hospital, n (%) | ||
| Non-profit, not religious order affiliated | 8 (57) | 15 (75) |
| Government | 4 (29) | 4 (20) |
| Private | 1 (7) | 1 (5) |
| Private administration with public funding | 1 (7) | |
| Hospital affiliation, n (%) | ||
| Independent, free-standing | 2 (14) | 1 (5) |
| Multi-facility organization (chain) | 5 (36) | 5 (25) |
| Hospital system, attached | 4 (29) | 7 (35) |
| Hospital system, free-standing | 3 (21) | 7 (35) |
| Population size served, median (IQR) | 1,625,000 (3,735,699) | 2,400,000 (2,875,319) |
| Number of inpatient beds, median (IQR) | 679 (287.3) | 616 (388.5) |
| Number of operating rooms, median (IQR) | 21 (18) | 31 (22.5) |
| Number of beds in intensive care unit, median (IQR) | 48.5 (44.8) | 74 (81.5) |
| Hospital is a primary teaching hospital for a medical school, n (%) | 14 (100) | 16 (80) |
IQR interquartile range
*The cluster that was withdrawn is not included
Orthopedic practices participating in the Aqueous-PREP trial treat a median of 149 open fractures annually and include a median of 11 orthopedic surgeons who treat fractures and 8.5 who take trauma calls (Table 3). Orthopedic practices participating in the PREPARE trial treat a median of 142 open fractures annually and 1090 closed fractures annually. These orthopedic practices include a median of 7.5 orthopedic surgeons who treat fracture patients and 9.5 who take trauma calls.
Table 3.
Orthopedic characteristics
| Characteristic | Aqueous-PREP, N = 14* | PREPARE, N = 20 |
|---|---|---|
| Number of orthopedic surgeons who treat fracture patients, median (IQR) | 11 (11.3) | 7.5 (7) |
| Number of orthopedic surgeons who take trauma calls, median (IQR) | 8.5 (13) | 9.5 (9.5) |
| Orthopedic training programs, n (%)** | ||
| Orthopedic fellowship | 13 (93) | 17 (85) |
| Orthopedic residency | 14 (100) | 18 (90) |
| Orthopedic clerkship | 14 (100) | 17 (85) |
| None of above | 0 (0) | 1 (5) |
| Annual number of operatively managed open fractures, median (IQR) | 149 (125) | 142 (185) |
| Annual number of operatively managed closed fractures, median (IQR) | 1200 (828.8) | 1090 (630.5) |
| Annual number of operatively managed closed lower extremity and pelvic fractures, median (IQR) | – | 725 (435) |
| Number of operating rooms used to treat fractures, median (IQR) | 4 (2) | 4 (5.5) |
IQR interquartile range
*The cluster that was withdrawn is not included
**Percentages do not sum to 100 as categories are not mutually exclusive
Participating orthopedic practices employ a variety of surgical infection prevention measures (Table 4). Specifically, practices participating in the Aqueous-PREP trial have a median of 2.2 infection preventionists per 250 hospital beds. Sixty-four percent of hospitals (n = 9) perform active surveillance for methicillin-resistant Staphylococcus aureus (MRSA) in open fracture patients. None of the clinical sites perform active surveillance for vancomycin-resistant Enterococcus (VRE) in open fracture patients. Sixty-four percent of practices (n = 9) have a decolonization protocol for MRSA. All practices have some infection control parameters in place in their operating rooms (e.g., high-efficiency particulate air (HEPA) filters, positive pressure operating rooms, air changes per hour, temperature maintained between 20 and 24 °C, and humidity between 20 and 60%).
Table 4.
Surgical infection prevention information
| Characteristic | Aqueous-PREP, N = 14* | PREPARE, N = 20 |
|---|---|---|
| Number of infection preventionists per 250 hospital beds, median (IQR) | 2.2 (1.7) | 2.6 (1.9) |
| Infection preventionists certified in infection control, median (IQR) | 3 (2.8) | 4 (3) |
| Perform active surveillance cultures for MRSA in open fracture patients, n (%) | 9 (64) | 4 (20) |
| Perform active surveillance cultures for VRE in open fracture patients, n (%) | 0 (0) | 4 (20) |
| Perform active surveillance cultures for other organisms in patients with open fractures, n (%) | 2 (14) | 3 (15) |
| Perform active surveillance cultures for MRSA in closed fracture patients, n (%) | – | 3 (15) |
| Perform active surveillance cultures for VRE in closed fracture patients, n (%) | – | 3 (15) |
| Perform active surveillance cultures for other organisms in patients with closed fractures, n (%) | – | 1 (5) |
| Decolonization protocol for MRSA, n (%) | 9 (64) | 12 (60) |
| Policies and/or guidelines on maintaining normothermia during perioperative period, n (%) | 14 (100) | 16 (80) |
| Airflow system(s) in operating room, n (%)** | ||
| None | 0 (0) | 1 (5) |
| Vertical laminar flow | 9 (64) | 14 (70) |
| Conventional ventilation | 6 (43) | 7 (35) |
| Other | 0 (0) | |
| Parameters in place in hospital operating rooms, n (%)** | ||
| High-efficiency particulate air (HEPA) filters | 12 (86) | 15 (75) |
| Operating rooms are positive pressure | 14 (100) | 16 (80) |
| Air changes per hour | 13 (93) | 17 (85) |
| Temperature maintained between 20 and 24 °C and humidity between 20 and 60% | 13 (93) | 18 (90) |
*The cluster that was withdrawn is not included
**Percentages do not sum to 100 as categories are not mutually exclusive
Orthopedic practices participating in the PREPARE trial employ a median of 2.6 infection preventionists per 250 hospital beds. Twenty percent of orthopedic practices (n = 4) perform active surveillance for MRSA in open fracture patients, and 15% of orthopedic practices (n = 3) perform active surveillance in closed fracture patients. No clusters perform active surveillance for VRE in either open or closed fracture patients. Sixty percent (n = 12) of orthopedic practices have a decolonization protocol for MRSA. Additionally, all practices had some infection control parameters in place in their operating rooms.
Discussion
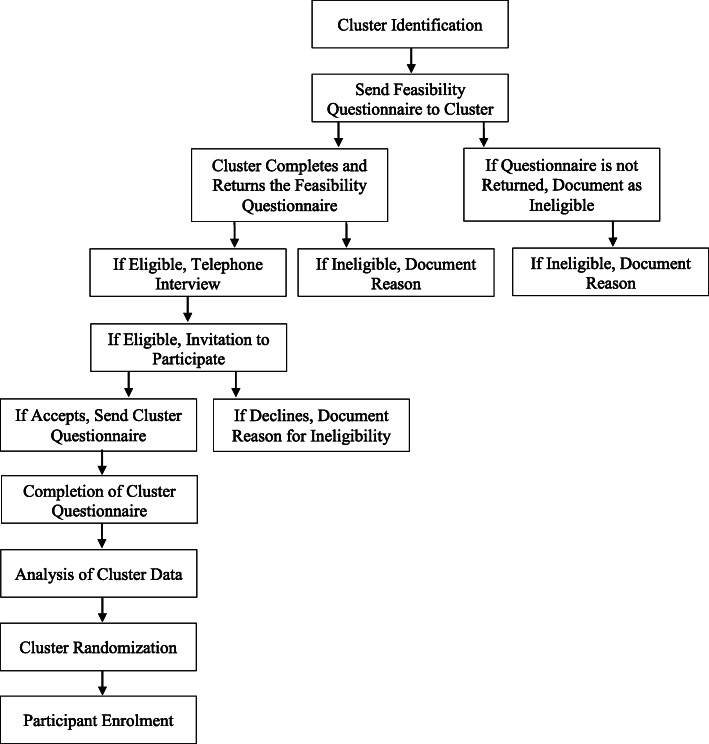
The Aqueous-PREP and PREPARE trials represent two ongoing CRXO trials within the field of orthopedics. The successes we have experienced with cluster identification and selection provide support for the feasibility of using this trial design in orthopedic trauma trials. As part of the PREP-IT program’s commitment to transparency and upholding rigorous reporting standards, this paper clearly defines the cluster unit, provides detailed information about the cluster identification and selection process, and describes key characteristics of the included clusters (Fig. 4). These are some of the key reporting requirements described in the CONSORT extension for CRTs [4].
Fig. 4.
Overview of the PREP-IT cluster identification, selection, and randomization processes
Given that the Aqueous-PREP and PREPARE trials use a pragmatic design, when selecting orthopedic practices (i.e., clusters), we aimed to include a sample that was representative of the orthopedic practices that treat the patient population in “real world” usual care settings. This is an essential component of pragmatic trials which aim to determine how well interventions work in the “real world” as opposed to explanatory trials which aim to determine how well interventions can work in ideal settings [8]. To accomplish this goal, we designed our cluster eligibility criteria to be as broad as possible while still ensuring that participating orthopedic practices would have the infrastructure and patient volume to successfully follow the protocol and meet study timelines. Through our cluster selection process, we identified 58 potentially eligible orthopedic practices and ultimately selected 5 sites to participate in both the Aqueous-PREP and PREPARE trials, 10 in the Aqueous-PREP trial only, and 15 in the PREPARE trial only. This represents an inclusion rate of 26% and 34%, respectively. Using the Pragmatic-Explanatory Continuum Indicator Summary (PRECIS-2) toolkit, a tool designed to measure where a trial falls on the pragmatic versus explanatory continuum on several domains, the setting of the Aqueous-PREP and PREPARE trials received a score of 4 out of a possible 5 indicating that the trials are “mostly pragmatic.” This score is reflective of the fact that most of our included orthopedic practices are affiliated with teaching hospitals that are level I trauma centers and regional referral centers versus a mix of trauma centers and local community hospitals. This is unsurprising as large academic hospitals tend to have both the infrastructure and patient volume to make participation in the PREP-IT trials feasible, whereas smaller community hospitals may have insufficient research infrastructure.
Despite our rigorous cluster selection process, we withdrew one cluster as they were unable to comply with the Aqueous-PREP protocol [6]. Specifically, this cluster was unable to meet the a priori threshold 90% of patients receiving the allocated antiseptic preparation solution. Additionally, the clinical site was unable to complete the case report forms, which led to incomplete data submission. These issues were the result of changes in research infrastructure from the time of site selection to site initiation. The Executive Committee, with support from the Methodology Core, made the decision to withdraw this site. As the criteria for cluster withdrawal were established prior to the initiation of the trial and a small proportion of data are affected, bias should not be introduced as a result of the decision to withdraw this cluster. Additionally, withdrawing this cluster will improve the overall integrity of the trial, as allowing them to continue in Aqueous-PREP would have led to increased protocol violations, treatment contaminations, and missing data.
As the primary outcome for both the Aqueous-PREP and PREPARE trials is SSI, we documented infection control processes. To remain pragmatic with data collection, we documented the infection control processes at three different levels: cluster level, surgeon level, and patient level. The most important SSI prevention strategies and infection strategies that are likely to vary from patient-to-patient, including antibiotic prophylaxis and blood sugar control, are documented at the patient level on the case report forms. Other infection control details (e.g., hair removal methods, type of surgical gown) are collected at the surgeon level, as these typically vary by surgeon but remain consistent across patients. Infection control measures that are implemented at the hospital level, including normothermia and decolonization protocols, are documented at the cluster level.
We aimed to select orthopedic practices that follow infection prevention protocols that are representative of the usual care that fracture patients would receive in the real world. We have found some variation in the infection control processes across the clusters in both the Aqueous-PREP trial and the PREPARE trial. Specifically, we found that 64% of all orthopedic practices in the Aqueous-PPREP trial perform active surveillance cultures for MRSA (64%). In the PREPARE trial, we found that only 20% of orthopedic practices perform active surveillance for MRSA in open fracture patients and 15% for closed fracture patients. In both trials, there was substantial variation in the use of decolonization protocols for MRSA (protocols in use at 64% of Aqueous-PREP practices and 60% of PREPARE practices).
Providing the knowledge user with a high level of detail regarding the hospital characteristics, orthopedic surgery details, and infection prevention protocols and procedures allows the knowledge user to determine the generalizability of the trial and the applicability to their hospital and surgical practice.
Conclusions
When undertaking CRXO trials, it is imperative to follow rigorous reporting standards, as outlined in the CONSORT extensions for CRTs and CRXO trials, including providing knowledge users with a clear definition of the cluster unit, a thorough description of the cluster identification and selection process, and sufficient description of key cluster characteristics. The PREP-IT trials illustrate how this can be accomplished using two large infection prevention CRXO trials in orthopedic fracture patients as an example.
Supplementary information
Additional file 1. THE PREP-IT Investigators.
Acknowledgements
We acknowledge and appreciate the many patients, nurses, national organizations, and others who have contributed to the success of this trial. Additional file 1 details the names, affiliations, and roles of the PREP-IT team; the Executive Committee is responsible for the overall conduct of the trial and is comprised of the Principal Investigators and a patient partner. The Executive Committee is advised by a Steering Committee; multiple clinical, research, and stakeholder specialty cores; and experts in patient engagement (University of Maryland PATIENTS Program). An Adjudication Committee reviews participant eligibility and reported study events. The Methods Centre is responsible for the day-to-day management of the PREP-IT trials, which includes clinical site management, data management, and data analysis. The Administrative Centre is responsible for piloting each trial protocol, contracting with each clinical site, and overseeing the Central Institutional Review Board activities.
Abbreviations
- PREP-IT Program
The Program of Randomized trials to Evaluate Pre-operative antiseptic skin solutions in orthopaedic Trauma
- RCTs
Randomized controlled trials
- CRXO
Cluster randomized crossover
- SSI
Surgical site infection
- Aqueous-PREP
A Pragmatic Randomized trial Evaluating Pre-operative aqueous antiseptic skin solutions in open fractures
- PREPARE
A Pragmatic Randomized trial Evaluating Pre-operative Alcohol skin solutions in Fractured Extremities
- CDC
Center for Disease Control and Prevention
- IQR
Interquartile range
- MRSA
Methicillin-resistant Staphylococcus aureus
- VRE
Vancomycin-resistant Enterococcus
- HEPA
High-efficiency particulate air
- PRECIS-2
Pragmatic-Explanatory Continuum Indicator Summary
Appendix
Characteristics of the withdrawn cluster
Table 5.
Cluster characteristics
| Characteristic | Withdrawn cluster |
|---|---|
| Level of trauma care provided | Level I |
| Ownership of hospital | Non-profit, not religious order affiliated |
| Hospital affiliation | Hospital system, free-standing |
| Population size served | 6,000,000 |
| Number of inpatient beds | 640 |
| Number of operating rooms | 17 |
| Number of beds in intensive care unit | 102 |
| Hospital is a primary teaching hospital for a medical school | Yes |
Table 6.
Surgical infection prevention information
| Characteristic | Withdrawn cluster |
|---|---|
| Number of infection preventionists per 250 hospital beds | 5 |
| Infection preventionists certified in infection control | 1 |
| Perform active surveillance cultures for MRSA in open fracture patients | No |
| Perform active surveillance cultures for VRE in open fracture patients | No |
| Perform active surveillance cultures for other organisms in patients with open fractures | No |
| Decolonization protocol for MRSA | Yes |
| Policies and/or guidelines on maintaining normothermia during perioperative period | Yes |
| Airflow system(s) in operating room | Conventional ventilation |
| Parameters in place in hospital operating rooms | High-efficiency particulate air (HEPA) filters |
| Operating rooms are positive pressure | |
| Air changes per hour | |
| Temperature maintained between 20 and 24 °C and humidity between 2 and 60% |
Authors’ contributions
SS, MB, and GPS conceived the study. SS, TS, and LT analyzed and interpreted the data. SS, TS, SD, DP, and PM drafted the manuscript. All authors critically revised the manuscript and gave final approval of the version to be published.
Funding
The PREPARE trial is funded by the Patient-Centered Outcomes Research Institute (PCORI) (PCS-1609-36512) and the Canadian Institutes of Health Research (CIHR) (Foundation Grant); the Aqueous-PREP trial is funded by the US Department of Defense (W81XWH-17-1-070) and the CIHR (Foundation Grant). McMaster University Surgical Associates funded start-up activities at the Methods Centre and The Physician Services Incorporated provided funding to the Methods Centre and Hamilton Health Sciences for the Aqueous-PREP trial. The views in this publication are solely the responsibility of the authors and do not necessarily represent the views of the PCORI, its board of governors, or methodology committee.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethics approval and consent to participate
Ethics approval for PREPARE and A-PREP was obtained through the Hamilton Integrated Research Ethics Board (Project #4913 and #4336) and Advarra (Pro00028360 and Pro00023709). Informed consent was obtained for all study participants enrolled in the PREP-IT trials.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary information accompanies this paper at 10.1186/s13063-020-04611-9.
References
- 1.Sackett D. Rules of evidence and clinical recommendations on the use of antithrombotic agents. Chest. 1989;95(2 Suppl):2S–4S. doi: 10.1378/chest.95.2_Supplement.2S. [DOI] [PubMed] [Google Scholar]
- 2.Arnup SJ, Forbes AB, Kahan BC, Morgan KE, McDonald S, McKenzie JE. The use of the cluster randomized crossover design in clinical trials: protocol for a systematic review. Syst Rev. 2014. 10.1186/2046-4053-3-86. [DOI] [PMC free article] [PubMed]
- 3.Arnup SJ, McKenzie JE, Hemming K, Pilcher D, Forbes AB. Understanding the cluster randomised crossover design: a graphical illustraton of the components of variation and a sample size tutorial. Trials. 2017. 10.1186/s13063-017-2113-2. [DOI] [PMC free article] [PubMed]
- 4.Campbell MK, Piaggio G, Elbourne DR, Altman DG. CONSORT 2010 statement: extension to cluster randomised trials. BMJ. 2012;345(sep04 1):e5661. doi: 10.1136/bmj.e5661. [DOI] [PubMed] [Google Scholar]
- 5.Arnup SJ, Forbes AB, Kahan BC, Morgan KE, McKenzie JE. The quality of reporting in cluster randomised crossover trials: proposal for reporting items and an assessment of reporting quality. Trials. 2016. 10.1186/s13063-016-1685-6. [DOI] [PMC free article] [PubMed]
- 6.Program of Randomized Trials to Evaluate Pre-operative Antiseptic Skin Solutions in Orthopaedic Trauma (PREP-IT) Investigators, Slobogean GP, Sprague S, Wells J, Bhandari M, Rojas A, Garibaldi A, Wood A, Howe A, Harris AD, Petrisor BA, Mullins DC, Pogorzelski D, Marvel D, Heels-Ansdell D, Mossuto F, Grissom F, Del Fabbro G, Guyatt GH, Della Rocca GJ, Nguyen U, et al. Effectiveness of iodophor vs chlorhexidine solutions for surgical site infections and unplanned reoperations for patients who underwent fracture repair: the PREP-IT master protocol. JAMA Network Open. 2020;3(4):e202215. 10.1001/jamanetworkopen.2020.2215. [DOI] [PMC free article] [PubMed]
- 7.Centers for Disease Control and Prevention (CDC) Surgical site infection (SSI) event. 2017. [Google Scholar]
- 8.Ford I, Norrie J. Pragmatic trials. N Engl J Med. 2016. 10.1056/NEJMra1510059. [DOI] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1. THE PREP-IT Investigators.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.