What causes absence seizures and are they really benign? Crunelli et al. review the pathophysiology, pharmacotherapy and neuropsychiatric comorbidities of absence seizures, and highlight the key role of cortical, thalamic and basal ganglia mechanisms in driving absence seizure ictogenesis.
Keywords: cortico-thalamo-cortical loop, basal ganglia, limbic system, attention deficits, anti-absence drugs
Abstract
Absence seizures in children and teenagers are generally considered relatively benign because of their non-convulsive nature and the large incidence of remittance in early adulthood. Recent studies, however, show that 30% of children with absence seizures are pharmaco-resistant and 60% are affected by severe neuropsychiatric comorbid conditions, including impairments in attention, cognition, memory and mood. In particular, attention deficits can be detected before the epilepsy diagnosis, may persist even when seizures are pharmacologically controlled and are aggravated by valproic acid monotherapy. New functional MRI-magnetoencephalography and functional MRI-EEG studies provide conclusive evidence that changes in blood oxygenation level-dependent signal amplitude and frequency in children with absence seizures can be detected in specific cortical networks at least 1 min before the start of a seizure, spike-wave discharges are not generalized at seizure onset and abnormal cortical network states remain during interictal periods. From a neurobiological perspective, recent electrical recordings and imaging of large neuronal ensembles with single-cell resolution in non-anaesthetized models show that, in contrast to the predominant opinion, cortical mechanisms, rather than an exclusively thalamic rhythmogenesis, are key in driving seizure ictogenesis and determining spike-wave frequency. Though synchronous ictal firing characterizes cortical and thalamic activity at the population level, individual cortico-thalamic and thalamocortical neurons are sparsely recruited to successive seizures and consecutive paroxysmal cycles within a seizure. New evidence strengthens previous findings on the essential role for basal ganglia networks in absence seizures, in particular the ictal increase in firing of substantia nigra GABAergic neurons. Thus, a key feature of thalamic ictogenesis is the powerful increase in the inhibition of thalamocortical neurons that originates at least from two sources, substantia nigra and thalamic reticular nucleus. This undoubtedly provides a major contribution to the ictal decrease in total firing and the ictal increase of T-type calcium channel-mediated burst firing of thalamocortical neurons, though the latter is not essential for seizure expression. Moreover, in some children and animal models with absence seizures, the ictal increase in thalamic inhibition is enhanced by the loss-of-function of the astrocytic GABA transporter GAT-1 that does not necessarily derive from a mutation in its gene. Together, these novel clinical and experimental findings bring about paradigm-shifting views of our understanding of absence seizures and demand careful choice of initial monotherapy and continuous neuropsychiatric evaluation of affected children. These issues are discussed here to focus future clinical and experimental research and help to identify novel therapeutic targets for treating both absence seizures and their comorbidities.
Introduction
Absence seizures are sudden, relatively brief lapses of consciousness associated with lack of voluntary movements and distinctive electrographic spike-wave discharges (SWD) at 2.5–4 Hz (Fig. 1A) (Panayiotopoulos, 1999, 2008; Crunelli and Leresche, 2002b; Blumenfeld, 2005b; Matricardi et al., 2014). These generalized non-convulsive seizures mostly have a polygenic background and can be present together with other seizure types in various age-dependent and age-independent epilepsies with different rates of remittance and expected outcomes (Crunelli and Leresche, 2002b; Camfield and Camfield, 2005; Gardiner, 2005; Fisher et al., 2017; Scheffer et al., 2017). The personal, familial and societal burden of absence seizures is considerable, being influenced by the immediate symptoms of the seizures and the common psychiatric comorbidities, including attentional, cognitive, memory and mood impairments (Masur et al., 2013; Cnaan et al., 2017). Moreover, the high incidence of pharmaco-resistance (Glauser et al., 2013) and the persistence of comorbidities even after full control of the seizures (Masur et al., 2013; Holmes and Noebels, 2016) highlight the need of an improved and holistic mechanistic understanding to facilitate the development of novel medications and other therapeutic approaches that effectively address the entirety of the disease, i.e. seizures and comorbid conditions. Achieving such mechanistic knowledge has undoubtedly been challenging, given the often unrecognized large spectrum of absence seizure semiology and EEG features, the lack of suitable biomarkers, the heterogeneity of (and lack of drug-free) cohorts used in many studies, the ethical restrictions of invasive clinical studies and the less than optimal experimental conditions used in some studies in animal models. Notwithstanding these difficulties and limitations, advancements with significant clinical implications are being made.
Figure 1.

Spectrum of SWD features and attention levels in children with absence seizures. (A) Characteristic EEG presentation of SWD in CAE. [B(i)] A long SWD with variable cycle waveform (left trace) was associated with lack of consciousness (indicated by the interruption of counting in the period marked by the two arrows) while a short SWD (right trace) in the same CAE child was not. [B(ii)] SWD with double spikes (enlarged on the right). [B(iii)] A long, hyperventilation-induced SWD from another child with CAE. (C) Different absence seizures, characterized by small and large changes in BOLD signal amplitude and 2.5–4 Hz EEG power, are associated with spared and impaired attention, respectively. DMN = default-mode network; TPN = task-positive network; SMT = sensorimotor-thalamic network. A is modified from Cerminara et al. (2012), and C from Guo et al. (2016).
Here, we discuss novel insights on absence seizure initiation, ictogenesis, comorbidity and therapy that have originated both from non-invasive human studies and invasive investigations in animal models. Points of controversy and crucial gaps in knowledge will be highlighted raising questions that should contribute to improve current clinical practice and to focus future clinical and experimental research. The emphasis will mainly be on absence seizure of childhood absence epilepsy (CAE) (where these seizures are the only clinical symptom) and on animal models with an absence seizure-only phenotype, as in these children and model systems the epileptogenesis and ictogenesis of absence seizures are not compromised or modulated by the concomitant presence of other types of seizures. Nevertheless, many of the findings, key issues and conclusions raised and discussed in this review do apply to absence seizures that are present in other epilepsies (Guo et al., 2016; Tangwiriyasakul et al., 2018).
Spectrum of clinical symptoms and electrographic features of absence seizures
Contrary to the classical textbook illustration of regular 2.5–4 Hz SWD (Fig. 1A), the electrographic presentation of absence seizures can be highly variable even in the same individual and may include SWD with different amplitudes and durations, absence of spikes, presence of double spikes and a progressive decrease in frequency towards the end of the paroxysm (Fig. 1B) (Panayiotopoulos, 1999). Moreover, the secondary clinical symptoms of absence seizure (e.g. automatisms, atonic and tonic muscular components, mild clonic eye or mouth jerks etc.) may be highly variable among children and during different absence seizure in the same child. The primary loss of consciousness itself is also variable and a recent study combining functional MRI, EEG and attentional tests has directly demonstrated the broad spectrum of cognitive impairments that is associated with different absence seizures, with some seizures showing a fully spared attention (Fig. 1C) (Guo et al., 2016).
A similarly large spectrum of absence seizure electrographic and behavioural features is also present in various genetic and pharmacological animal models and even in the same mouse or rat during successive absence seizures. However, animal models do show differences with human absence seizures, including a higher SWD frequency at 5–11 Hz and lack of absence seizure remittance with age [for comprehensive summaries of similarities and differences of absence seizures between humans and animal models, see Pitkänen et al. (2017)]. In this context, it is important to mention that two recent studies have questioned the validity of current rodent models of absence seizure because (i) these model rats can modify the duration of their SWD (suggesting that they are not adverse events) (Taylor et al., 2017); and (ii) absence seizure-like events are present in wild-caught rats (implying that they are unlikely to represent a pathological trait) (Taylor et al., 2019). Whereas the results of both studies are welcome as they directly confirm and enlarge anecdotal evidence and previous scientific findings made in inbred and outbred rat strains since the 1980s, their interpretations and conclusions do not consider the variability in clinical symptoms and electrophysiological features that are present in human absence seizures.
The issue raised by the first study (Taylor et al., 2017) has been extensively debated previously (Depaulis et al., 2017) and will thus not be addressed further here except for emphasizing that the broad spectrum of clinical symptoms in affected children (and adults) includes absence seizures that are associated with both impaired and unimpaired attention (Fig. 1C) (Guo et al., 2016). Regarding the issue raised by the second study (Taylor et al., 2019), the majority (65%) of SWD recorded by these authors in wild-caught rats are <1 s long (and none >2 s) and would thus be considered to be subclinical seizures, i.e. absence seizures for which the electrographic but not the cognitive and behavioural components may be evident and measurable. Indeed, such short subclinical SWD rarely have an overt clinical correlate and absence seizures with spared attention tend to have a very short duration (∼3 s) (Guo et al., 2016). Nevertheless, the study of Taylor et al. (2019) strengthens the view that the potential presence of ethosuximide-sensitive SWD in various ‘normal’ (i.e. non-epileptic) inbred and outbred mouse and rat colonies should always be carefully considered when selecting an appropriate control group for comparison with mouse and rat absence seizure model strains. In other words, animals used as the control group should whenever possible be screened for the presence of absence seizures before inclusion in comparative experiments.
Absence seizure initiation
The pioneering non-linear analysis of multi-site local field potentials in cortico-thalamic networks carried out by Meeren et al. (2002) in a genetic model of absence seizures, the Wistar Albino Glaxo from Rijswijk (WAG/Rij) rats (Depaulis and van Luijtelaar, 2006) showed for the first time the key role of a localized cortical region (the peri-oral region of primary somatosensory cortex) in the initiation of absence seizures. Later work in another genetic model, the Genetic Absence Epilepsy Rats from Strasbourg, GAERS (Depaulis and van Luijtelaar, 2006), characterized the barrel field region of primary somatosensory cortex (Studer et al., 2018) and layer 5/6 excitatory pyramidal neurons (Polack et al., 2007) as the originators of the initial paroxysmal activity. Indeed, absence seizures in GAERS rats are abolished by blocking the firing of these neurons (Polack et al., 2009), and are more susceptible to ethosuximide applied in the primary somatosensory cortex than the motor cortex or ventrobasal thalamus (Richards et al., 2003; Manning et al., 2004). Notably, a recent study (Lee et al., 2019) has shown that whereas spontaneous SWD in PLC-β4 knockout mice originate in the somatosensory cortex, the majority of SWD in the pharmacological γ-hydroxybutyric acid (GHB) model (Snead, 1992; Venzi et al., 2015) appear first in the prefrontal cortex, indicating different sites of SWD initiation in different mouse and rat, genetic and pharmacological models as in children with absence seizures (see below).
Studies in humans
Blood oxygenation level-dependent (BOLD) functional MRI studies have been critical in identifying key regions involved in human absence seizures (Aghakhani et al., 2004; Gotman et al., 2005; Hamandi et al., 2006; Moeller et al., 2008). In particular, the lack of generalization at the start of an absence seizure has now been confirmed in many multi-modal investigations of young, adolescent and adult populations with absence seizures, including high-density EEG, MEG and functional MRI studies in which localized changes in precuneus, posterior cingulate cortex, lateral parietal cortex and/or frontal cortex are observed before other brain areas become involved in the paroxysm (Holmes et al., 2004; Westmijse et al., 2009; Carney et al., 2010; Bai et al., 2011; Gupta et al., 2011; Benuzzi et al., 2012; Tenney et al., 2013; Wu et al., 2017). Indeed, the first study on a pure CAE cohort reported increases in BOLD signal amplitude occurring from 14 s before the clinical and electrographic signatures of a seizure could be observed (Bai et al., 2010) (Fig. 2A). Notably, the cortical area(s) where these pre-ictal increases in BOLD amplitude occur may differ from child to child but are mostly consistent between seizures for a given child (Bai et al., 2010, 2011; Moeller et al., 2010). Importantly, from absence seizure onset the same cortical areas show a decreased BOLD signal amplitude that may persist for up to 10 s after the end of the seizure (Bai et al., 2010; Moeller et al., 2010; Benuzzi et al., 2012).
Figure 2.
BOLD signal changes precede absence seizures and persist interictally. (A) Changes in functional MRI BOLD signal amplitude are present in children with absence seizures 14 s before the clinical and electrographic signatures of a seizure are manifested. (B) In individuals with generalized SWD, including a small CAE cohort, an increase in the phase-synchrony of functional MRI BOLD signals is present in a ‘spike-wave cortical network’ from a few seconds before until 20 s after a seizure (top left) (nodes on the far right column). There is also a decreased phase-synchrony in an ‘occipital cortical network’ that starts at least 1 min prior to seizure onset (bottom left). Altered phase-synchrony of BOLD signals in the ‘sensorimotor cortical network’ persists interictally but is not present in healthy controls (middle). Modified from Bai et al. (2010) and Tangwiriyasakul et al. (2018).
Whereas the above functional MRI studies investigated changes in BOLD amplitude, a recent study has analysed the phase-synchrony of BOLD signals in people with generalized SWD, including a small CAE cohort (Tangwiriyasakul et al., 2018). These authors found that there is persistently increased synchrony in a sensorimotor cortical network during non-ictal periods. At least 1 min prior to absence seizure, synchrony in an occipital cortical network begins to decrease (Fig. 2B). Then, from around 10 s prior to SWD onset, the hubs of a SWD-specific cortical network (involving prefrontal regions, precuneus, medial parietal cortex, and medial and lateral prefrontal cortex) begin to show a linear increase in synchrony. Together, this indicates that a persistently abnormal sensorimotor network may represent a ‘pre-ictal’ brain state that can smoothly progress to an ictal SWD network (Fig. 2B). Notably, a high level of synchrony is still apparent in the SWD network 20 s after the end of the electrographic paroxysmal activity, after which the brain reverts to the pre-SWD state (Tangwiriyasakul et al., 2018). In summary, this work demonstrates for the first time the presence of a persistently, i.e. ictally and interictally, altered cortical sensorimotor network in people with absence seizure, a finding of great significance for our understanding of the pathophysiology of these seizures and their comorbidities (see ‘Neuropsychiatric comorbidities of absence seizures’ section).
The existence of altered resting state functional connectivity that may underlie some of the inter-ictal behavioural impairments is supported by the correlation between decreased medial frontal cortex activation during a cognitive task and impaired task performance in CAE (Bai et al., 2011; Killory et al., 2011; Vega et al., 2011; Luo et al., 2014). Furthermore, the variable extent of behavioural impairments during absence seizures has been shown to correlate with the magnitude of ictal BOLD amplitude changes, providing the first indications of where the behavioural component of the seizures might be mediated (Fig. 1B) (Berman et al., 2010; Guo et al., 2016).
Studies in animal models
Attempts to gain additional mechanistic insights by comparing non-invasive imaging in humans with simultaneous non-invasive imaging and invasive recordings in animal models have, however, so far been hampered by the fact that rodent models of absence seizures do not show the widespread cortical ictal and post-ictal decreases in multi-modal haemodynamic measurements (functional MRI, cerebral blood flow and volume) (Tenney et al., 2004; David et al., 2008; Mishra et al., 2011) as observed in humans (Bai et al., 2010; Moeller et al., 2010; Benuzzi et al., 2012). These conflicting results are not due to the frequency difference between rodent and human SWD (5–11 Hz and 2.5–4 Hz, respectively) as anaesthetized ferrets, which exhibit 3 Hz SWD, show cortical functional MRI increases (Youngblood et al., 2015). This clinical-experimental discrepancy must then be explained by either a mechanistic limitation of absence seizure models, differences in higher cortical function between humans and animals, a peculiarity of human versus lower mammal neurovascular coupling or an effect of the anaesthetics hitherto used in the animal imaging studies. A combination of these factors is also possible: ictal cortical BOLD amplitude changes in awake GAERS rats (Kratochvil et al., 2018) are similar to those in individuals with absence seizures, as are those in Sprague Dawley rats administered the pro-absence drug GHB (Venzi et al., 2015) after recovery from ketamine/medetomidine anaesthesia (Tenney et al., 2003), though WAG/Rij rats still express cortical increases in the same ketamine/medetomidine recovery conditions (Tenney et al., 2004). In summary, given these apparent discrepancies between models (GHB versus WAG/Rij) and drug conditions (GAERS drug-free versus WAG/Rij anaesthetized and WAG/Rij post-anaesthesia), a greater understanding of the case-specific influences on neurovascular coupling (Lemieux et al., 2011; Sieu et al., 2015) is required if any insight into the translational relevance or indeed the mechanisms of absence seizure is to be gained from non-invasive imaging in absence seizure animal models.
Significance of new evidence
A number of important implications derive from the findings outlined above.
First, the existence of well-delineated cortical networks or regions where haemodynamic and electrographic changes are first observed before spreading to other areas has raised the question of whether absence seizures should still be considered ‘generalized’ events (van Luijtelaar et al., 2014; Lüttjohann and Van Luijtelaar, 2015). This early pre-ictal cortical activity of absence seizure is a phenomenon very distinct from the structural and functional alterations that characterize the ‘focus’ of focal onset seizures (Fisher et al., 2017; Scheffer et al., 2017), thus emphasizing that the word ‘focus’ should not be used for absence seizures, as has occurred recently (van Luijtelaar and Sitnikova, 2006; Lüttjohann et al., 2011, 2014; Paz and Huguenard, 2015). Rather, in view of the many and far-located areas contributing to the pre-ictal changes of absence seizure, these cortical regions could be accurately referred to as the cortical initiation network (CIN). With this distinction in mind, the current classification of absence seizures as generalized seizures (Fisher et al., 2017; Scheffer et al., 2017) remains clinically appropriate and useful.
Second, larger studies of individuals with absence seizures may disclose different CINs than those so far uncovered (Bai et al., 2010; Tangwiriyasakul et al., 2018) (see also Carney et al., 2012; Masterton et al., 2013). In turn, these diverse networks may be potentially correlated with different genotypes (Crunelli and Leresche, 2002b; Guerrini and Noebels, 2014) and the outcome of monotherapy (Glauser et al., 2013; Shinnar et al., 2017). Indeed, the different location of the pre-ictal changes of cortical BOLD amplitude in various children with absence seizures (Bai et al., 2010, 2011; Moeller et al., 2010) suggests that a diverse genotype may bring about cellular and synaptic alterations preferentially in different cortical networks, a possibility that can be directly tested by large studies of combined functional MRI, EEG and/or MEG of affected individuals with a known genotype.
Third, the existence of a CIN settles the long-standing controversy about cortex or thalamus initiation of absence seizures (Avoli, 2012) in favour of the cortex, but is entirely compatible with the results of many experimental studies since the 1940s showing the emergence of SWD or absence seizures following various means of activation of different thalamic regions in normal (i.e. non-epileptic) animals (Avoli, 2012; Lüttjohann and Van Luijtelaar, 2015; Taylor and Crunelli, 2014; Sorokin et al., 2017). That small localized changes in neuronal excitability limited to a restricted cortical or thalamic region can prompt generalized SWD-like activity throughout cortico-thalamo-cortical networks suggests that these networks are naturally susceptible to such paroxysmal oscillations, as indicated by the expression of absence seizures in normal mice following knockout of a single gene (Cacna1a, coding for P/Q-type Ca2+ channels) selectively in layer 6 pyramidal neurons (Bomben et al., 2016) and the expression of short-duration SWDs in non-epileptic and wild-caught rodents (Taylor et al., 2017, 2019).
Fourth, whereas the above data provide novel insights on absence seizure initiation, there is currently no evidence as to which cortical and thalamic regions (or indeed other brain areas) are crucial for SWD generalization. Many studies in young and adult cohorts with absence seizures indicate the critical involvement of the thalamus in the expression of these seizures (Moeller et al., 2008, 2009, 2010, 2013; Tyvaert et al., 2009; Tenney et al., 2013). Notable in this respect is the observation that in GAERS rats very brief (<1 s), small amplitude SWD that are present in the cortex but not in the thalamus are not accompanied by any overt behavioural sign, i.e. no absence seizure occurs (Polack et al., 2007), suggesting that thalamic recruitment might be a key step for generalization. If so, thalamic higher order and intralaminar nuclei with their wide cortical innervation (Jones, 2003; Giber et al., 2015) might be the critical thalamic region(s). In particular, the rodent thalamic posterior nucleus, roughly equivalent to the anterior pulvinar region in humans (Jones, 2003), may be one of the key hubs for absence seizure generalization in view of (i) its critical involvement in pre- and post-ictal activity (Lüttjohann et al., 2013; Lüttjohann and Van Luijtelaar, 2015; Sysoeva et al., 2016b) and at the onset and offset of SWD (Lüttjohann and Pape, 2019); and (ii) the ability of its stimulation to induce a stronger and longer-lasting excitation of cortical layer 2/3 neurons compared to the activation of other thalamic nuclei (Zhang and Bruno, 2019).
Absence seizure ictogenesis
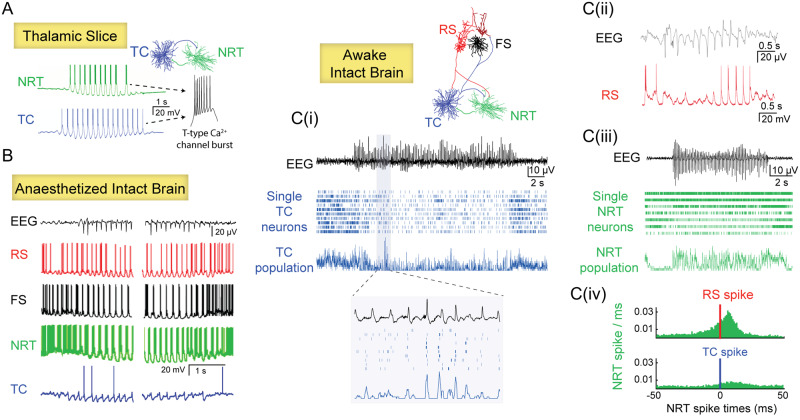
Our understanding of absence seizure ictogenesis has been dominated for 25 years by the results of brain slice studies, which suggested that hyperexcitability of the glutamatergic thalamocortical and the GABAergic reticular thalamic (NRT) neurons through their bursts of action potentials mediated by T-type Ca2+ channels (T-channels) at each paroxysmal cycle of a SWD form the rhythmogenic network mechanism of absence seizures (von Krosigk et al., 1993; Bal et al., 1995a, b; McCormick and Contreras, 2001) (Fig. 3A). Strong firing at each paroxysmal cycle is also present in NRT neurons and excitatory cortico-thalamic and inhibitory neurons of the CIN in animal models under anaesthesia (with either urethane, ketamine, fentanyl or fentanyl+antipsychotic) (Slaght et al., 2002; Polack et al., 2007; Chipaux et al., 2011; Williams et al., 2016), though under this experimental condition many thalamocortical neurons do not express T-channel-mediated bursts of action potentials and could, in fact, be electrically silent during many paroxysmal cycles within a SWD (Steriade and Contreras, 1995; Pinault et al., 1998, 2006) (Fig. 3B).
Figure 3.
Absence seizure ictogenesis in cortico-thalamo-cortical networks. (A) In thalamic slices, glutamatergic thalamocortical (TC) neurons and GABAergic reticular thalamic (NRT) neurons elicit a T-type Ca2+ channel-mediated burst of action potentials at each cycle of the putative paroxysmal activity. (B) In anaesthetized absence seizure models, except TC neurons that fire rarely, all other constituent neurons of cortico-thalamo-cortical networks—cortical regular-spiking excitatory pyramidal neurons (RS), cortical fast-spiking inhibitory neurons (FS) and NRT neurons—show strong (mostly burst) firing at each cycle of a SWD. (C) In sharp contrast, in awake animal models, RS neurons [C(ii)] show a marked ictal decrease of firing (bottom trace is an intracellular recording). The vast majority of TC neurons decrease their firing rate during seizures [raster plot in C(i)] though there is a consistent output at each cycle of the SWD when the firing of these neurons is grouped together [see enlarged population activity in C(i)]. Two groups of NRT neurons can be distinguished on the basis of their ictal firing: one group that increases their firing ictally (mostly consisting of T-type Ca2+ channel-mediated bursts of action potentials) [top three cells in the raster plot in C (iii)] and another group that shows a decreased activity [bottom three cells in the raster plot in C(iii)]. [C(iv)] Histograms of the activity of NRT neurons recorded simultaneously with an RS (top) or and a TC neuron (bottom) in a freely moving absence seizure model. Note how, in contrast to results in thalamic slices (A), the ictal NRT neuron firing (green bars) is mainly driven by the cortical RS neuron spikes rather than the TC neuron spikes. Time zero marks the firing of the RS and TC neurons (red and blue line, respectively). Modified from Bal et al. (1995b); Pinault et al. (1998); Polack et al. (2007); Chipaux et al. (2011); McCafferty et al. (2018b) and Meyer et al. (2018).
Recent evidence
Applying electrical recordings and calcium imaging of large neuronal ensembles to freely moving and head-restrained non-anaesthetized animals, respectively, two recent studies (McCafferty et al., 2018b; Meyer et al., 2018) have for the first time characterized with single-cell resolution the ictogenic network activity that takes place in cortex and thalamus during absence seizures in three models: a polygenic model (the GAERS rat), a monogenic model (the stargazer mouse) (Noebels et al., 1990) and the pharmacological GHB model in rats.
The first common discovery of both studies is that the vast majority of cortico-thalamic and thalamocortical neurons do not increase their total firing rate during a seizure but exhibit either a decrease or no change, with single neurons of both neuronal populations showing electrical silence at many SWD cycles [Fig. 3C(i, ii, iv) and 4A] (McCafferty et al., 2018b; Meyer et al., 2018). When they do fire, they elicit either a single spike or a burst of action potentials in synchrony with the SWD spike.
Figure 4.
Pre-ictal and ictal temporal firing dynamics of absence seizures. (A) Average fluorescence changes indicate a decrease in firing in unidentified cortical layer 5 and 6 (but not layer 4) neurons of the visual cortex of non-anaesthetized, head-restrained stargazer mice that begins more than 2 s before and ends almost 2 s after the electrographic seizure. A similar decrease in total firing rate that start 2 s before seizure onset and terminates at seizure offset also occurs in identified fast-spiking (FS) interneurons of the cortical initiation network (CIN) and in ventrobasal thalamocortical (TC) neurons (blue trace) of freely moving GAERS rats, whereas the total firing GABAergic thalamic reticular neurons (green trace) decreases from 3 s before seizure onset but then show a persistent ictal increase until seizure offset. In contrast, there is an increase in T-channel mediated burst firing in ventrobasal thalamocortical (blue) and thalamic reticular neurons (green) neurons that begins ∼1 s prior to seizure onset and continues until seizure offset. (B) Timing of action potential output with respect to the EEG-spike of different neurons in cortico-thalamo-cortical and basal ganglia pathways are superimposed on a schematic spike-and-wave (light black line). Time zero indicates the peak of the EEG spike and individual brain regions are shown on the right. Data for cortico-thalamic CIN layer 5/6, substantia nigra, ventrobasal thalamocortical and reticular nucleus are from freely moving GAERS rats (Deransart et al., 2003; McCafferty et al., 2018b), whereas data from striatum, subthalamic nucleus, and globus pallidus are from GAERS rats under neurolept anaesthesia (Slaght et al., 2004; Paz et al., 2005, 2007). As the firing time of cortico-thalamic layer 5/6 neurons in the CIN of freely moving GAERS rats occurs about 10 ms earlier than that of the same GAERS neurons recorded under neurolept anaesthesia, the timing of cortico-subthalamic, cortico-striatal, cortico-thalamic CIN layer 2/3 and layer 4 neurons taken from GAERS under neurolept anaesthesia were modified accordingly. FS-INT = fast-spiking GABAergic interneurons; MSN = striatal medium spiny neurons. Open and filled symbols indicate inhibitory GABAergic and excitatory glutamatergic neurons, respectively. (A) Data for CIN FS interneurons are unpublished observations, the others were modified from McCafferty et al. (2018b) and Meyer et al. (2018).
The second key and common finding is that cortico-thalamic, thalamocortical and NRT neurons are not hard-wired to either prevalently express electrical silence, bursts, or single action potential firing but can switch from one pattern to the others in successive paroxysmal cycles and seizures, with some neurons being mostly silent during one seizure but very active in subsequent ones (see Supplementary Fig. 5 in McCafferty et al., 2018b) (Meyer et al., 2018). Nevertheless, both cortex and thalamus consistently receive a robust EEG-spike-correlated barrage of action potentials from the other region at each and every SWD cycle see enlarged single cells and population activity in Fig. 3C(i)] (McCafferty et al., 2018b), indicating that ictal cortical and thalamic outputs are a population activity, as originally suggested by Buzsáki (1991) for thalamocortical neurons. In this respect, an important finding that has often been overlooked or misinterpreted is that, while the firing in the somatosensory cortex leads that of the somatosensory thalamus in the first 500 ms of a seizure (i.e. in the first three to four paroxysmal cycles in rodents), the activity of thalamic neurons may precede that of cortical neurons or vice versa during subsequent cycles (Meeren et al., 2002): i.e. once a seizure is fully generalized, either of these two regions may, in different paroxysmal cycles within a seizure, take a temporally leading role in reinforcing firing in the other. Thus, the average timing of the ictal firing of cortico-thalamic and thalamocortical neurons (of different thalamic nuclei) within a paroxysmal cycle greatly overlaps, as does that of NRT neurons (Fig. 4B).
The third common and unexpected result is that cortical, thalamocortical and NRT neurons markedly decrease their total firing ∼2–3 s before a SWD can be detected in the EEG (Fig. 4A) (McCafferty et al., 2018b; Meyer et al., 2018). In contrast, T-channel-mediated burst firing shows a steep increase that begins 0.5–1.0 s before seizure onset, peaks just few tens of milliseconds and 2.5 s after seizure onset in ventrobasal thalamocortical neurons and NRT neurons, respectively, and is maintained until seizure termination (Fig. 4A) (McCafferty et al., 2018b). While at present we do not know the mechanism underlying this pre-ictal reduction in cortical firing, the pre-ictal decrease in thalamocortical and NRT neuron firing might result from the reduced activity of layer 5/6 cortico-thalamic neurons (Fig. 4A). An alternative cause may the increased inhibitory input from a common source, such as the GABAergic neurons of the substantia nigra pars reticulata (SNr), which are known to innervate both NRT neurons and thalamocortical neurons of many first and higher-order thalamic nuclei (Ray and Price, 1992; Miyamoto and Jinnai, 1994; Sakai et al., 1998; François et al., 2002; Cebrián et al., 2005). However, while SNr neurons do markedly increase their firing during absence seizure in freely moving rats (Deransart et al., 2003) (see ‘Essential control of absence seizure by the basal ganglia’ section), it remains to be demonstrated whether such enhancement occurs before seizure onset.
In contrast to cortico-thalamic and thalamocortical neurons, NRT neurons in the intact brain of non-anaesthetized models fire at almost each SWD cycle [Fig. 3C(iii)] and show an increase in their ictal total firing compared to interictal periods (Fig. 4A) (McCafferty et al., 2018b). Moreover, in contrast to the results in vitro (von Krosigk et al., 1993; Bal et al., 1995a, b; Huntsman et al., 1999; McCormick and Contreras, 2001; Sohal et al., 2003) and under anaesthesia (Slaght et al., 2002), two populations of NRT neurons can be detected in freely moving models: one that preferentially fires T-channel bursts in successive cycles, while the other fires mostly tonic spikes relatively asynchronously [see top three and bottom four neurons, respectively, in Fig. 3C(iii)], with the former group showing a higher coherence with SWD in the EEG (McCafferty et al., 2018b). Whether these two groups may be related to the rostral and caudal sectors of the NRT that have different contributions to absence seizure in genetic models (Aker et al., 2006; Meeren et al., 2009) or to the chemically-distinct populations of NRT neurons that show different bursting activity in normal non-epileptic animals (Halassa et al., 2014) remains to be elucidated. Moreover, in contrast to the prevailing view (von Krosigk et al., 1993; Bal et al., 1995a,b; Huntsman et al., 1999; McCormick and Contreras, 2001), the ictal firing of NRT neurons in freely moving animals is driven by cortical and not thalamocortical neuron activity [Fig. 3C(iv)]: i.e. the NRT-mediated ictal inhibition of thalamocortical neurons is of a feed-forward and not a feed-back nature.
T-type Ca2+calcium channels and absence seizures
Another key finding of McCafferty et al. (2018b) is that T-channel bursts of cortical and NRT neurons are critically involved in the ictal synaptic interactions with thalamocortical neurons, whereas T-channel bursts of thalamocortical neurons do not play a role in the ictal interactions of these neurons with both cortical and NRT neurons. Indeed, localized injection of TTA-P2, a potent and selective pan-T-channel antagonist (Shipe et al., 2008; Dreyfus et al., 2010), by McCafferty et al. (2018b) shows that the block of T-channels in the NRT and CIN of GAERS rats drastically decreases absence seizures (see also Avanzini et al., 1992), whereas blocking T-channels in thalamocortical neurons of the ventrobasal thalamus, one of the somatotopic thalamic nuclei of the GAERS CIN, has no effect on absence seizures (Fig. 5), the rhythmicity of the thalamic output and its synchrony with the SWD spike. Thus, the use of TTA-P2 to provide a fast block of T-channel function without compensatory activities shows that T-channels of ventrobasal thalamocortical neurons (i.e. Cav3.1) are not essential for spontaneous absence seizures in GAERS rats whereas those of the CIN (i.e. Cav3.1, Cav3.2 and Cav3.3) and NRT (i.e Cav3.2 and Cav3.3) are (Fig. 5).
Figure 5.
T-type Ca2+ channels in cortex and nucleus reticularis thalami (NRT) are necessary for absence seizures. Schematic representation of injection sites with their complement of T-channel subtypes (left) and original data (right) showing the ability of the potent and selective pan T-type Ca2+ channel antagonist, TTA-P2, to block absence seizures in GAERS rats when bilaterally injected by microdialysis in the CIN (top row) or the NRT (middle row). Similar administration in the ventrobasal (VB) thalamic nucleus (one of the somatotopic thalamic nuclei of the CIN in this animal model) that contains only thalamocortical neurons, has no effect on absence seizures (bottom row).
This critical role of T-channels of cortical and NRT, but not thalamocortical neurons in experimental absence seizures is compatible with the gain-of-function mutations of the Cav3.2 subtype of T-channels reported in some children with absence seizures (Chen et al., 2003; Khosravani et al., 2004) and in GAERS rats (Talley et al., 2000; Powell et al., 2009; Cain et al., 2018), as this subtype of T-channels is expressed in cortical and NRT neurons but not in thalamocortical neurons (Fig. 5) (Talley et al., 1999). Indeed, selective expression of human Cav3.2 (C456S) mutant channels in the barrel cortex of normal mice does elicit absence seizures (Wang et al., 2015), and in vivo knockdown of Cav3.2 in GAERS NRT neurons markedly decreases absence seizures (Cain et al., 2018). Moreover, the lack of an essential role for Cav3.1 subtype of T-channels of ventrobasal thalamocortical neurons in absence seizures of GAERS rats (McCafferty et al., 2018b) is compatible with the expression of absence seizure in PLCβ4 knockout mice with increased L-type and Cav3.1 channel function (Cheong et al., 2009). This prior study, in fact, did not distinguish between L-type and Cav3.1 channels as it used mibefradil, a non-specific blocker of Na+, ATP-sensitive K+, L-type Ca2+ and T-type Ca2+ channels (Mishra and Hermsmeyer, 1994; Bezprozvanny and Tsien, 1995; Nilius et al., 1997; Viana et al., 1997; Gomora et al., 1999; Liu et al., 1999).
Significance of new evidence
A number of important issues derive from the novel findings outlined above. First, the emerging picture from the investigations conducted in the absence of anaesthesia suggests that, rather than an exclusively thalamic rhythmogenesis, cortical mechanisms are key in driving absence seizures and determining SWD frequency. In particular, the synchronous output of thalamocortical neurons is not determined by the dynamics of their T-channel-mediated burst firing but rather is driven by top-down cortical excitation and sharpened by cortically driven, i.e. feed-forward, NRT inhibition, with both the cortically-driven excitation and the NRT inhibition critically depending on T-channel bursts (McCafferty et al., 2018b). Notably, the key role of a cortical drive in absence seizure ictogenesis had been predicted by studies in thalamic slices with an artificially-generated cortical feed-back (Bal et al., 2000; Blumenfeld and McCormick, 2000).
Second, as during absence seizure all thalamic neurons are strongly driven by the excitatory cortical afferent activity, why do thalamocortical neurons show a decrease in ictal firing? The most likely explanation is that the stronger cortico-NRT neuron synapses (compared to cortico-thalamocortical neuron synapses) (Contreras et al., 1993; Golshani et al., 2001) and the robust NMDA-mediated drive of NRT neuron bursts (Lacey et al., 2012; Wang et al., 2015) assure a strong rhythmic inhibition of thalamocortical neurons that overcomes the cortically driven ictal excitation and thus restrict the output of thalamocortical neurons within a narrow temporal window around the EEG spike (Fig. 4B) (McCafferty et al., 2018b). Notably, the NRT-mediated inhibition of thalamocortical neurons is strengthened by the increased ictal firing of the GABAergic SNr neurons that is also phase-locked to the SWD-spike (Fig. 6) (see ‘Essential control of absence seizure by the basal ganglia’ section).
Figure 6.
Ictogenic firing of basal ganglia neurons. Schematic diagram of the ictal activity of the component neurons of the direct and indirect basal ganglia pathways recorded under neurolept anaesthesia (except the substantia nigra neurons) are illustrated close to the morphological representation of each neuronal type. Electrical activity of fast-spiking striatal interneurons and substantia nigra and subthalamic neurons shows extracellular recordings, the others are intracellular recordings. The excitatory and inhibitory nature of the synaptic connections is indicated by encircled plus and minus symbols. The relative size of the morphological reconstructions has been altered for graphical purposes. See text for additional details. Modified from Deransart et al. (2003); Slaght et al. (2004) and Paz et al. (2005, 2007).
Third, the ictal decrease in the correlation of neuronal activity among visual cortex neurons compared to interictal periods in stargazer mice (Meyer et al., 2018) is, on the one hand, surprising, as it leaves unexplained the origin of the EEG spikes that are present in the same cortical region. On the other hand, this decreased correlation may underlie the decreased ictal BOLD signal synchrony observed in the ‘occipital cortical network’ of individuals with generalized SWD and absence seizures (Tangwiriyasakul et al., 2018) (Fig. 2B) and the deficits in visual tasks often reported as comorbid condition of absence seizure (see ‘absence seizure neuropsychiatric comorbidity’ section).
Fourth, the relative paucity of the firing of cortical and thalamic neurons and their variable recruitment between subsequent cycles within a seizure and between seizures in non-anaesthetized animal models is in sharp contrast with the results of in vitro studies in cortical and thalamic slices and in vivo investigations under anaesthesia (Fig. 3). This conclusively demonstrates that absence seizure ictogenesis is not fully recapitulated either by anaesthetized models (without behaviour, with altered arousal and excitability, and often under the influence of a neuromuscular blocking drug) and by cortical or thalamic slices (in which reciprocal cortico-thalamic and thalamo-cortical connectivity are absent and the membrane potential deviates from that in the awake intact brain). Thus, isolated cortical and thalamic slices are of limited use when investigating the paroxysmal firing dynamics of SWD and care should be taken in the interpretation of firing patterns and temporal dynamics obtained under anaesthetized conditions. Nevertheless, isolated brain slices (from cortex, thalamus or other brain regions) still offer the opportunity to characterize the constitutive biophysical abnormalities of the voltage- and transmitter-gated channels of genetic absence seizure models or transfected human mutant channels.
Essential control of absence seizures by the basal ganglia
Since the original observation of the anti-absence effect of blocking SNr neuron activity by local injection of muscimol, a GABA-A receptor agonist, in freely moving GAERS rats (Depaulis et al., 1988a, b), many investigations using localized and selective pharmacological manipulations in this brain region, the striatum and the subthalamic nucleus have demonstrated that both the direct and indirect basal ganglia pathways exert a tight control over absence seizures in different freely behaving genetic and pharmacological models (Depaulis et al., 1989; Deransart et al., 1996, 2000, 2001; Vercueil et al., 1998). Notably, the involvement of the basal ganglia in absence seizures is supported by the consistent finding of ictal changes in functional MRI BOLD signals in the basal ganglia of CAE and adult cohorts with absence seizure (Li et al., 2009; Bai et al., 2010; Berman et al., 2010; Luo et al., 2011).
The patterns and temporal dynamics of the ictal firing of morphologically and electrophysiologically identified neurons of all basal ganglia nuclei have been thoroughly investigated (Slaght et al., 2004; Paz et al., 2005, 2007) and are illustrated in Figs 4B and 6. The ictal activity of cortico-striatal neurons consists of rhythmic depolarizations with mostly single action potential that are phase-locked to the SWD spike. In the striatum, the fast-spiking GABAergic interneurons show short (two to four spikes) high frequency (300–400 Hz) bursts during absence seizures, and thus the striatal output neurons, the GABAergic medium spiny neurons, do not fire ictally but only exhibit large rhythmic subthreshold depolarizations (Slaght et al., 2004). In contrast to cortico-striatal and cortico-thalamic neurons, the cortico-subthalamic neurons show robust firing in two distinct periods, one well before and one around the EEG spike (Paz et al., 2005). This double wave of excitation leads the excitatory subthalamic neurons to have a characteristic temporal profile consisting of a single action potential (well before the SWD spike), which is followed by a short period of silence and then a high frequency burst of action potentials around the SWD spike (Paz et al., 2005). Thus, the output of the basal ganglia to the thalamus, i.e. the firing of the GABAergic SNr neurons, is increased during absence seizures (Deransart et al., 2003) as a result of the combined effect of (i) the ictal electrical silence of the GABAergic striatal medium spiny neurons (Slaght et al., 2004); and (ii) the increased ictal firing of the glutamatergic subthalamic neurons (Fig. 6) (Paz et al., 2005). Notably, all these studies were carried out in GAERS rats and, except the one in the SNr (Deransart et al., 2003), performed under neurolept anaesthesia. Thus, since the firing of both cortico-thalamic, thalamocortical and NRT neurons show subtle but key differences when recorded under this anaesthetic regime and in freely moving animals (see previous section and Fig. 3), it would be important to investigate the activity of the various neuronal populations of the basal ganglia under the latter experimental condition and in other animal models.
A recent study on STXBP1, a gene encoding the presynaptic protein Munc18-1, shows that injection of muscimol in the striatum of Stxbp1−/− mice blocks their short (0.5–2 s) ethosuximide-sensitive absence seizures (Miyamoto et al., 2019). Moreover, striatal application of NASPM, a selective blocker of calcium-permeable AMPA receptors, which are more abundant in the fast-spiking striatal interneurons than in the striatal medium spiny neurons (Deng et al., 2007), elicits absence seizures in wild-type mice and (continuous) activation of Designer Receptor Exclusively Activated by Designer Drugs (DREADDS)-transfected fast-spiking striatal interneurons markedly reduces ASs in Stxbp+/− mice (Miyamoto et al., 2019). These pharmacological and pharmaco-genetic results directly demonstrate a key role for the striatum in the control of absence seizures but are not supported by the lack of changes in the ictal firing of putative striatal fast-spiking and medium spiny neurons. Indeed, the latter result and other findings of Miyamoto et al. (2019) are in contrast with previous observations in morphological identified striatal neurons of GAERS rats (see above) (Fig. 6) (Slaght et al., 2004). It could be that different populations of striatal neurons have a differential sensitivity to these drugs because in monkeys there is evidence of a diverse activation of encephalin-, parvalbumin- and substance P-containing striatal neurons of the direct and indirect basal ganglia pathways following different stimulation paradigms of cortico-striatal neurons in the sensorimotor cortex (Parthasarathy and Graybiel, 1997). Thus, further studies using selective manipulations for either the direct or the indirect basal ganglia pathways are required to fully clarify the pro- or anti-absence contribution that the DRD1- and DRD2-containing striatal medium spiny neurons and various striatal interneuron populations may make to absence seizure.
Enhanced inhibition of thalamocortical neurons: a key ictogenic feature of absence seizures
The essential control of absence seizures by the SNr (Depaulis et al., 1988a, b, 1989; Deransart et al., 1996, 2001) implies a causal role for the increased firing of its output neurons in absence seizure generation and thus for an enhanced ictal GABAergic inhibition of thalamocortical neurons of the ventromedial, ventrolateral and ventrobasal nuclei as well as the many other thalamic regions, including the limbic thalamus and intralaminar thalamic nuclei, that are innervated by the SNr (Ray and Price, 1992; Miyamoto and Jinnai, 1994; Sakai et al., 1998; François et al., 2002; Cebrián et al., 2005). Notably, recent findings have shown a direct excitatory connection from the substantia nigra to the NRT (Antal et al., 2014), which will undoubtedly add to the ictal cortical excitation of these thalamic GABAergic neurons whose axons are known to cover almost the entire thalamus (Pinault, 2004). Thus, two separate sources of increased ictal GABAergic inhibition, one from the SNr and one from the NRT, engulf thalamocortical neurons of the majority of thalamic nuclei during absence seizure, providing (i) a tight temporal control of the ictal firing of these neurons via activation of synaptic GABA-A receptors, i.e. phasic GABA-A inhibition (Fig. 4B) (McCafferty et al., 2018b); and (ii) a persistent decrease in excitability, via activation of extra-synaptic GABA-A receptors, i.e. tonic GABA-A inhibition (Cope et al., 2005). This ictal increase in the inhibition of thalamocortical neurons markedly contributes to the overall decrease in total firing, the paucity of single action potential firing and the increased T-channel-mediated burst firing during absence seizures (Figs 3 and 4A). Within these similarities, however, thalamocortical neurons in the ventrobasal, ventromedial and ventrolateral nuclei do show slightly different ictal firing features (Figs 3 and 6) (Pinault et al., 1998; Paz et al., 2007; McCafferty et al., 2018b). Thus, as different classes of layer 5/6 output neurons, i.e. cortico-thalamic, cortico-striatal and cortico-subthalamic neurons, show different ictal firing patterns (Figs 3 and 6), so do thalamocortical neurons in different thalamic nuclei. Though the precise reasons for these differences still remain to be fully elucidated, three possibilities might be considered. First, in contrast to the accepted view the posterior thalamus, but not the ventrobasal nucleus, receives not only inhibitory but also direct excitatory connections from the substantia nigra (Antal et al., 2014). Thus, it may be possible that the balance between SNr-derived inhibition and excitation plays a role in the different ictal firing observed in thalamocortical neurons of the posterior nucleus and other thalamic nuclei. Second, there could be a differential modulation by DRD1 and DRD2 receptors of the tonic GABA-A inhibition in thalamocortical neurons of different thalamic nuclei (Connelly et al., 2013a, b; Yague et al., 2013). Third, the involvement of GABA-B receptors, including the generation of GABA-B receptor-mediated synaptic potentials (Crunelli et al., 1988; Crunelli and Leresche, 1991; Charpier et al., 1999), their direct modulation of extra-synaptic GABA-A receptors (Connelly et al., 2013a, b), the regulation of transmitter release via presynaptic GABA-B receptors (Gervasi et al., 2003; Bessaih et al., 2006) and the direct activation of GABA-B receptors of thalamic astrocytes (Gould et al., 2014), may be different in various thalamic regions.
Notably, in some mouse and rat genetic models of absence seizure, including the GAERS rats and the stargazer mice, the combined NRT- and SNr-derived ictal increase in GABAergic inhibition of thalamocortical neurons may be enhanced by the loss-of-function of the astrocytic GABA transporter GAT-1 (but not GAT-3) (Cope et al., 2009; Pirttimaki et al., 2013), providing additional restraints to the thalamocortical neuron firing. Notably, since there is only a silent mutation in SLC6A1 (the gene encoding GAT-1) in these two absence seizure models (Cope et al., 2009), their GAT-1 loss-of-function might be due to its aberrant intracellular transport, misplaced location around the synaptic cleft, alteration in phosphorylation states or inefficient modulation by various endogenous signalling molecules (Beckman et al., 1999; Venderova et al., 2005; Vaz et al., 2011). Both in GAERS rats and stargazer mice, this GAT-1 loss of function leads to constitutively higher GABA levels in the thalamus, but not somatosensory cortex (Richards et al., 1995) and an increased basal tonic GABA-A inhibition in thalamocortical neurons (Cope et al., 2009; Errington et al., 2011a). Indeed, enhanced tonic inhibition in ventrobasal thalamocortical neurons is sufficient to elicit absence seizure in normal mice and rats and is necessary for the expression of absence seizure in the abovementioned models as well as in the GHB and other models (Cope et al., 2009; Errington et al., 2011b). These results in animal models are corroborated by the following findings in humans: (i) higher GABA levels were observed in ipsilateral thalamus, but not cortex, of a child with atypical unilateral SWD (Leal et al., 2016); (ii) anti-epileptic drugs that increase GABA levels, such as tiagabine and vigabatrin, aggravate or induce absence seizures and are contraindicated in individuals with absence seizures (Perucca et al., 1998; Ettinger et al., 1999; Panayiotopoulos, 1999); (iii) the vast majority of currently known human mutations in SLC6A1 lead to a GAT-1 loss-of-function (Mattison et al., 2018); and (iv) absence seizures are by far the most common epileptic phenotype in children with SLC6A1 mutations (Dikow et al., 2014; Johannesen et al., 2018).
In summary, solid experimental and clinical evidence strongly suggest that, in contrast to the classical view that absence seizures are driven by increased excitation and/or decreased inhibition in all brain regions involved in the paroxysmal activity, the ictogenic thalamic mechanism of absence seizure is uniquely shaped by the enhanced GABAergic inhibition of thalamocortical neurons that derives both from intra-thalamic (NRT), and extra-thalamic (SNr) sources. Whether other sources of thalamic GABAergic inhibition (i.e. zona incerta, anterior pretectal nucleus) (Giber et al., 2008) participate in the ictal increase of thalamocortical neuron inhibition is currently unknown. In some absence seizure models and children with absence seizure, this increased ictal inhibition of thalamocortical neurons is enhanced by the loss-of-function of GAT-1, which does not always result from a genetic mutation of this transporter gene. Whether a similarly increased inhibition and GAT-1 loss-of-function also shape the ictogenic mechanisms of excitatory and inhibitory neurons within cortical and basal ganglia networks remains to be established.
Pharmaco-resistance of absence seizure: recognizing the problem
For many years now, the pharmacological therapy of absence seizures of different epilepsies, and in particular of CAE, juvenile absence epilepsy and juvenile myoclonic epilepsy, has mostly relied on ethosuximide, valproic acid, lamotrigine or any of their combinations (Penovich and Willmore, 2009). In 2013, Glauser et al. published the results of the first large (446 newly diagnosed) double-blind randomized controlled trial in a pure CAE cohort that compared the efficacy and tolerability of the above medications. This study shows that at 12 months the freedom-from-failure rate for monotherapy with either ethosuximide or valproic acid is similar (∼45%) and much higher than that for lamotrigine (21%) (Fig. 7). Surprisingly, children who had longer seizures before the start of the pharmacological therapy were more likely to achieve seizure-freedom, a result that did not depend on the type of anti-absence drug used (Dlugos et al., 2013). Notably, children receiving valproic acid performed substantially worse in tests for attention than those receiving ethosuximide or lamotrigine (see below), and 42% of all children who discontinued the treatment because of adverse events were receiving valproic acid. Later, the same authors used their well-characterized large cohort to examine the clinical response to a second monotherapy in children experiencing failure of the initial treatment (Cnaan et al., 2017). After 1 year, 49% of children were seizure-free with a higher proportion receiving ethosuximide or valproic acid (57% or 49%, respectively) than lamotrigine (36%) (Fig. 7). The superior efficacy of ethosuximide over lamotrigine as initial monotherapy and the presence of fewer cognitive and behavioural adverse effects than valproic acid have been confirmed by another recent study (Shinnar et al., 2017) and by analysis of a prospective observational cohort that found better long-term (≥5 and ≥10 years) outcomes (i.e. seizure- and medication-free) in children initially treated with ethosuximide (76% and 76% complete remission, respectively) than with valproic acid (39% and 44%, respectively) (Berg et al., 2014).
Figure 7.
Pharmaco-resistance of absence seizures. The first 12-month-long monotherapy with either ethosuximide (ETX) or valproic acid (VPA) is more effective than that with lamotrigine (LTG) in controlling absence seizures (left). Similar results are obtained when children unresponsive to the initial monotherapy undergo a second 12-month-long monotherapy with a different drug (middle). The combined results of the first and second monotherapy (right) show that about 30% of children with childhood absence epilepsy have pharmaco-resistant absence seizures. Modified from Glauser et al. (2013) and Cnaan et al. (2017).
In summary, the serious implications of these studies are that (i) in contrast to prevailing views ‘appropriately chosen and used antiepileptic drugs’ (the ILAE criterion for drug-resistant epilepsy) (Kwan et al., 2009) leave ∼30% of children with uncontrolled absence seizures; and (ii) there are strong neuropsychiatric adverse effects associated with valproic acid treatment, a critical factor to be considered when selecting the first monotherapy in paediatric and juvenile populations with absence seizures.
Neuropsychiatric comorbidities of absence seizures
Studies in humans
It is only relatively recently that the full extent of the psychiatric comorbid conditions that accompany absence seizures has been investigated, though clear warnings were available since the early 1960s from studies of mixed-age mixed-epilepsy populations with generalized SWD, mostly under pharmacological therapy (Hertoft, 1963; Ounsted et al., 1963; Loiseau et al., 1983; Wirrell et al., 1997; Aldenkamp et al., 2001; Ott et al., 2001; Henkin et al., 2005; Vanasse et al., 2005). The unpromising picture that emerges from recent investigations shows that ∼60% of children with CAE have neuropsychiatric comorbidities, with attention deficits as the most common comorbidity (35–40%), followed by mood disorders (Caplan et al., 2008; Glauser et al., 2010; Masur et al., 2013; Gencpinar et al., 2016; Lee et al., 2018) (Fig. 8), in sharp contrast to the high prevalence of depression associated with convulsive seizures (Noebels, 2015b). Notably, in the only study of a large (446 children) drug-naïve CAE cohort, the impairments in attention were not of an ADHD type as they were ‘associated more with an inattentive than hyperactive form of attention deficit, i.e. loss of focus on a task more than impulsivity of response’ (Masur et al., 2013), a result similar to that reported previously in a smaller cohort of children with absence seizures and other idiopathic epilepsies (Hermann et al., 2007). Moreover, though not always assessed in drug-naïve populations, reductions of visual-related functions, including visual memory and visuo-spatial tasks, have also been consistently reported (Fedio and Mirsky, 1969; Jambaqué et al., 1993; Pavone et al., 2001; Nolan et al., 2004; Cheng et al., 2017), a finding that may be linked to the recently reported decrease of interictal BOLD synchrony in the occipital cortex of a CAE cohort (Tangwiriyasakul et al., 2018) and the lack of ictal firing synchrony among visual cortical neurons in an absence seizure model (Meyer et al., 2018).
Figure 8.
Neuropsychiatric comorbidities of absence seizures. Attention deficits are the most common psychiatric comorbidity of childhood absence epilepsy; they can be present in pre-symptomatic children and persist even when seizures are pharmacologically controlled. Other cognitive impairments and mood disorders (e.g. anxiety, depression) may also be present. It is likely that the aberrant cortico-thalamo-cortical networks underlying absence seizure combine with the abnormal basal ganglia-limbic-monoamine networks underlying cognitive impairments and mood disorders to generate the overall neurological and neuropsychiatric phenotype of epilepsies with absence seizures (left column). Interactions between these abnormal networks might contribute to a lower seizure threshold and an increased risk of comorbidity. Moreover, absence seizures and anti-absence drugs may induce, or aggravate existing, comorbid conditions (right column). Solid and dashed blue lines represent established and putative links, respectively.
The presence of neuropsychiatric deficits is complemented by structural imaging data in cohorts with absence seizures showing (i) smaller grey matter volume of brain regions involved in attention (including orbital frontal gyrus and temporal lobes) (Caplan et al., 2009); (ii) smaller volume of the amygdala (a region linked to both attention and anxiety/depression) (Schreibman Cohen et al., 2009); (iii) altered age-related changes in cortical thickness and sulcal depth (Tosun et al., 2011); and (iv) diffusion tensor imaging abnormalities in default mode network nodes (Qiu et al., 2016). Functional imaging evidence of abnormalities in the default mode, dorsal attention and salience networks have also been reported (Berman et al., 2010; Killory et al., 2011; Luo et al., 2014; Li et al., 2015). Moreover, BOLD signal synchrony in frontal and precuneus regions is altered ictally while that in precentral areas and cingulum is affected both ictally and interictally (Tangwiriyasakul et al., 2018) (Fig. 2B). However, more than 90% of the children and adults enrolled in all these structural and functional imaging studies were on anti-absence medication for variable lengths of time and no analysis of the drug effects as a covariate was carried out: related confounds cannot, therefore, be discounted since the effect of anti-absence drugs on both structural and functional brain imaging features both in humans and animal models is unknown.
Psychiatric comorbid conditions, in particular attention deficits, may precede the first absence seizure and the epilepsy diagnosis (Hermann et al., 2007; Jones et al., 2007) (Fig. 8). Whereas these results may suggest that the pathophysiological abnormalities underlying absence seizure comorbidities are at least partially independent from seizure occurrence, it is highly likely that the epileptogenesis existing in presymptomatic children may also underlie (and/or contribute to) their comorbid conditions (Fig. 8). Moreover, attention deficits do persist even when seizures are pharmacologically controlled (Williams et al., 2002; Glauser et al., 2013; Masur et al., 2013), and notably more children show this comorbidity after valproic acid (49%) than ethosuximide (32%) and lamotrigine (24%) monotherapy (Glauser et al., 2013). However, it remains unclear whether the comorbidity observed after anti-absence drug treatment is pre-existing, drug-induced and/or due to lack of seizure control (Fig. 8, right). The mutual causal interaction between epilepsy and comorbidity is a very complex one in particular for epilepsies with a genetic aetiology (Noebels, 2015b; Keezer et al., 2016): answering the above question, therefore, is essential and will require knowledge of the psychiatric effects of anti-absence drugs in healthy individuals (and normal animals, see below), which unfortunately are currently unknown (Loring et al., 2007; Verrotti et al., 2018).
Studies in animal models
Absence seizure models do show psychiatric comorbidities, providing additional validity to these models and a means for future mechanistic studies into these conditions. Subtle alterations in attention, that may be task-specific, have been reported in GAERS (Marques-Carneiro et al., 2016; Marks et al., 2016a, b) and WAG/Rij rats (Karson et al., 2012; Jafarian et al., 2015), but the majority of the animal studies has focused on anxiety‐ and depression-like behaviours that have been consistently observed in GAERS (Jones et al., 2008; Bouilleret et al., 2009, Powell et al., 2009, 2014a, b; Tringham et al., 2012; Dezsi et al., 2013; Marques-Carneiro et al., 2014) and WAG/Rij rats (Sarkisova et al., 2010; Russo et al., 2011; van Luijtelaar et al., 2013; Moyanova et al., 2018). Notably, anxiety and absence seizures in GAERS rats can be reduced by environmental enrichment and these anxiolytic and anti-epileptogenic effects are heritable into the next generation (Dezsi et al., 2016), prompting future investigations on potential epigenetic contributions to human absence seizures and their comorbidity. Moreover, rearing off-spring of WAG/Rij rats by non-epileptic Wistar mothers from the second postnatal day results in a marked decrease of absence seizures in early adulthood (Sitnikova et al., 2016), suggesting that an increased maternal care and/or a reduction in stressful environment early in development may contribute to alleviate genetically determined absence seizures.
Chronic (19 weeks) and acute administration of ethosuximide mitigate the anxiety-like phenotype in GAERS (Dezsi et al., 2013), and anxiety and depression in SWD-expressing Long-Evans rats (Shaw et al., 2009). In WAG/Rij rats, chronic ethosuximide ameliorates depressive-like behaviour (Sarkisova et al., 2010; van Luijtelaar et al., 2013) (but see Russo et al., 2011). Notably, however, the abnormal phase-amplitude coupling of EEG beta-gamma waves observed in stargazer mice is unaffected by ethosuximide (Maheshwari et al., 2017), indicating that aberrant cortical circuit-level dynamics persist even after seizure control. The pan-T-channel blocker Z944 abolishes absence seizures, reduces aggressive behaviour, rescues both recognition memory deficits (Marks et al., 2016a) and sociability impairment (Henbid et al., 2017), but increases anxiety-like behaviour in GAERS rats (Marks et al., 2019). Thus, this potent and selective T-channel blocker controls absence seizures, but in contrast to the weak and non-selective T-channels blocker ethosuximide (Crunelli and Leresche, 2002a), it worsens anxiety-like behaviour, at least in GAERS rats. Finally, chronic lamotrigine, which is the least effective absence seizure monotherapy in humans (Glauser et al., 2013; Cnaan et al., 2017), blocks absence seizures and ameliorates comorbid anxiety- and depression-like phenotypes in Long-Evans rats (Huang et al., 2012). Notably, no study has investigated whether anti-absence drugs induce psychiatric deficits in normal non-epileptic (either young or adult) animals, except for one investigation reporting negative effects of ethosuximide on fear memory but not spatial learning (Ponnusamy and Pradhan, 2006).
However, the interpretation of many of these studies on comorbidity in absence seizure models is compromised by several issues. First, the majority of these investigations did not record the EEG during the behavioural tests, making it impossible to ascertain how the observed changes in behaviour are linked to ictal and interictal periods. Second, in studies using inbred absence seizure models, a comparison with normal animals as well as inbred non-epileptic control strains would be essential to control for potential effects of long-term inbreeding of the non-epileptic strain (Marques-Carneiro et al., 2014; Bombardi et al., 2018). Third, all but two studies (Marks et al., 2016a; Henbid et al., 2017) have used only male animals. Fourth, fear-reaction tests are not the ideal procedure to study attention and memory in absence seizure models that have inherent anxiety- and depression-like comorbid conditions and should be replaced by touch-screen technology (Markham et al., 1996).
Significance of new evidence
In summary, attention deficits are the most common psychiatric comorbidity of absence seizures when present as the only seizure type: they may precede the first absence seizure (i.e. are present in pre-symptomatic children), persist even after seizures are pharmacologically controlled (Fig. 8) and are much more frequent following monotherapy with valproic acid than ethosuximide or lamotrigine. Other cognitive impairments and mood disorders may also be present (Fig. 8), but systematic analyses of their incidence, life-progression and causal link with treatment are not yet available. It is likely that the aberrant function of cortico-thalamo-cortical networks (underlying the generation of SWD and absence seizures) (Crunelli and Leresche, 2002a; Blumenfeld, 2005a) combines with the abnormal activity of basal ganglia-limbic-monoamine networks (underlying attention deficits, cognitive impairments and mood disorders) (De Deurwaerdère et al., 2017; Miskowiak and Petersen, 2019; Park et al., 2019) to generate the overall neuropsychiatric comorbid phenotype of absence seizure (Fig. 8, left). Notably, anterior and mediodorsal thalamic nuclei, which show consistent ictal BOLD changes in individuals with absence seizure (Tyvaert et al., 2009) and are crucial for attentional control (Schmitt et al., 2017), provide a structural and functional bridge between limbic and cortico-thalamo-cortical networks (Jones, 2003). Moreover, alterations in cognition and memory may be significantly influenced by the characteristic biophysical properties of thalamocortical neurons (Connelly et al., 2015, 2016, 2017) and by the T-channel-mediated low-threshold spike and bursts that occur during absence seizures, as long-term changes in the plasticity of thalamic synapses preferentially occur when thalamic neurons fire T-channel bursts of action potentials and are rarely observed when these neurons fire tonic single action potentials (Sieber et al., 2013; Pigeat et al., 2015; Leresche and Lambert, 2017; Crunelli et al., 2018). Unravelling the true nature of the link between absence seizure epileptogenesis/ictogenesis and comorbidities, and understanding whether and to what extent psychiatric comorbid conditions are drug-induced and/or drug-aggravated (Fig. 8, right) are hindered by the lack of data on the effects of currently available anti-absence medicines on attention, learning and mood in healthy humans and in normal (young and adult) animals.
New treatments for absence seizures and their psychiatric comorbid conditions
The pharmaco-resistance of 30% of children with absence seizure, the persistence of neuropsychiatric comorbidities in seizure-free cohorts and the adverse effects of current medications (particularly in polytherapy) demand urgent action on novel treatments. Naturally, the best therapy for any epilepsy is one that would block epileptogenesis, i.e. has a disease-modifying effect. As far as current anti-absence drugs are concerned, some limited evidence in children suggests that early, effective treatment with ethosuximide has positive long-term (≥10 years) outcomes, i.e. seizure- and medication-free (Berg et al., 2014). This potential disease-modifying effect of ethosuximide is strongly supported by results in WAG/Rij and GAERS rats showing that initiating treatment with ethosuximide prior to seizure onset and continuing it for 3–4 months not only fully blocks absence seizures even 3 months after the end of treatment and markedly reduces comorbid anxiety (Blumenfeld et al., 2008; Dezsi et al., 2013), but also increases the mRNA levels of DNA methyltransferases, a key component of epigenetic pathways, in the CIN of GAERS rats (Dezsi et al., 2013) and abolishes the abnormal expression of Nav1.1, Nav1.6 and HCN1 in WAG/Rij rats (Blumenfeld et al., 2008). In contrast, a shorter treatment (3–5 weeks) initiated even prior to seizure onset does not protect GAERS rats after the end of the treatment (Dedeurwaerdere et al., 2005; Jarre et al., 2017). Indeed, when the start and the duration of the ethosuximide treatment were directly compared in the same study on WAG/Rij rats, only the 4-month treatment (but not the 2-month treatment starting either at the second or fourth month of life) was effective up to 2 months post-treatment (van Luijtelaar et al., 2013). This suggests that either epileptogenic mechanisms are still operant even when SWD appear to be fully developed or that an effective prophylactic treatment needs to affect both epileptogenic and early epileptic processes. Obviously, any potential future translation of an early treatment to humans would require identification of solid genetic (Noebels, 2015a; Holmes and Noebels, 2016) or phenotypic biomarkers before the epileptogenic processes have fully established themselves.
As far as novel pharmacological therapies are concerned, there is only one new medication (Epidiolex®), an oral form of cannabidiol, a phytocannabinoid devoid of psychotropic actions (Stockings et al., 2018), that is currently in phase 2 trials in children with pharmaco-resistant absence seizure (ClinicalTrials.gov #NCT03336242; ClinicalTrials.gov #NCT03355300). This is surprising and of concern as this drug was shown to have no effect on non-convulsive absence seizures in previous trials in two severe childhood epilepsies, i.e. Lennox-Gastaux and Dravet syndromes (Devinsky et al., 2019; Thiele et al., 2018). Moreover, to the best of our knowledge there are no publicly available data on the anti-absence effect of cannabidiol in mice and rats with absence seizures and no study has investigated its potential adverse effects on attention, cognition, learning and memory both in normal non-epileptic young animals and in absence seizure models.
As indicated previously, T-type Ca2+ channels of cortical and NRT neurons are key to absence seizure expression (McCafferty et al., 2018b) (Fig. 5). This suggests that potent and selective T-channels blockers, in contrast to the weak and non-selective actions of ethosuximide and valproic acid on these channels (Leresche et al., 1998; Crunelli and Leresche, 2002a, b), may offer concrete hope for pharmaco-resistant absence seizures. Regrettably, the first generation of potent and selective T-channel blockers (Dreyfus et al., 2010) elicited major changes in the vigilance states of absence seizure models (Shipe et al., 2008). However, a member of a new generation of state-dependent T-channel antagonists, CX-8998, is currently in a phase 2 trial in adolescent and adult populations with generalized epilepsy and absence seizures that do not respond to standard of care treatment (ClinicalTrials.gov #NCT03406702, 2018). Moreover, Z944 (now called PRAX-944), another pan-T-channel antagonist that blocks experimental absence seizure and ameliorates some aspects of their comorbidity, is in a phase 2 trial in an adult population with pharmaco-resistant absence seizure (ANZCTR.). It is hoped that CX-8998 and PRAX-944 may, in the near future, be tested for their efficacy and safety in paediatric and juvenile cohorts with absence seizure.
As mentioned earlier, a loss-of-function of GAT-1 in some rat and mouse genetic models leads to increased thalamic GABA levels and to experimental absence seizures via activation of extrasynaptic δ subunit-containing GABA-A receptors (Cope et al., 2009). Thus, drugs that increase the activity of GAT-1 and antagonists of δ-containing GABA-A receptors may represent new targets for treating absence seizure (Errington et al., 2011a). Proof-of-concept for this potential therapy is provided by the rescue of the absence phenotype (and lack of adverse effects) in the off-springs of stargazer mice crossed with GABA-A δ-subunit knockout mice (McCafferty et al., 2018a). Another interesting target could be the 5-HT2C receptor (Di Giovanni and De Deurwaerdère, 2016), as selective agonists at these receptors have been shown to dose-dependently block experimental absence seizure (Venzi et al., 2016). Finally, positive allosteric modulators of metabotropic glutamate receptors 1 and 5, which are key to cortico-thalamic transmission (Baude et al., 1993; Romano et al., 1995; Errington et al., 2011c), block experimental absence seizures (D’Amore et al., 2015; Celli et al., 2019), as does the selective thalamic block of hyperpolarization-activated, cyclic nucleotide-gated channels (David et al., 2018) (see also Hammelmann et al., 2019; Zobeiri et al., 2019), opening up the possibility of selectively targeting one of these receptor/channel subtypes for treating CAE and other epilepsies with absence seizure.
Moving away from pharmaco-therapy, optogenetics has been suggested as a potential future alternative to neurosurgery for adults with pharmaco-resistant epilepsies (Wykes et al., 2012, 2016; Bui et al., 2017; Christenson Wick and Krook-Magnuson, 2018). However, the inherent invasiveness of optogenetic techniques and the substantial incidence of absence seizure remittance in CAE (Camfield and Camfield, 2005) do not lead at present to a favourable risk-benefit analysis for a standard optogenetic therapy. Notably though, the brief reversible and safe opening of the blood–brain barrier induced by non-invasive focused ultrasound in a small brain region (Munoz et al., 2018) allows locally restricted diffusion of an intravenously injected (virally-driven) opsin in mice that can then be activated by transcranial light stimulation (Pouliopoulos et al., 2018). In principle, therefore, following the identification of the CIN in an individual with absence seizures this technique does provide the possibility of delivering and activating light-sensitive proteins without surgical intervention. The safety and reliability of focused ultrasound have already been tested in healthy adults and people with other neurological diseases (Carpentier et al., 2016; Lipsman et al., 2018) but has not been investigated yet in younger populations.
Finally, the block of experimental absence seizures by non-invasive transcranial electrical stimulation (Berenyi et al., 2012; Liu et al., 2018) offers at present a more realistic potential than optogenetics and may have the additional advantage of improving attention as shown in some studies of adult populations (Reteig et al., 2017). However, the implementation of both therapeutic approaches will require improvements of existing algorithms for absence seizure prediction (Maksimenko et al., 2017). Moreover, as transcranial electrical stimulation has been shown to affect brain oscillations and functional connectivity (Krause et al., 2017, 2019) its application to children and teenagers with absence seizures demands knowledge of the long-term effects of repetitive electrical stimulation on brain development before it might be considered a reliable and safe therapeutic avenue.
Future priorities
Mechanism of pre-ictal and abnormal interictal activity
A key future challenge will be to uncover the electrical correlates of the changes in BOLD signal amplitude and synchrony that precede by tens of seconds and even up to 1 min, respectively, the start of absence seizure in humans (Holmes et al., 2004; Bai et al., 2010, 2011; Tangwiriyasakul et al., 2018). Detecting such early pre-ictal changes in EEG spike-wave morphology, frequency, synchrony and/or coupling strength will be fundamental for the development of efficient prediction algorithms (Gadhoumi et al., 2016; Freestone et al., 2017), which in turn are necessary for the potential implementation of transcranial electrical, optogenetic (or other therapeutic/prophylactic) stimulation protocols. So far, no study has systematically analysed the EEG at such early pre-ictal periods in children with absence seizure, and the possibility cannot be discarded that the changes in electrical activity underlying these early pre-ictal functional MRI alterations in humans are preferentially confined to deep cortical layers (Polack et al., 2007) and thus undetectable by standard EEG electrodes.
From an experimental perspective, unambiguous changes in cortical and thalamic single-neuron activity (Fig 4A) (Pinault, 2003; McCafferty et al., 2018b; Meyer et al., 2018), in the power of EEG theta, delta and beta frequency bands and in inter-cortical area coupling strengths (Sysoeva et al., 2014, 2016a, b; Sorokin et al., 2016) have been reported in mouse and rat absence seizure models but they all occur only around 2–4 s prior to seizure onset. As changes in functional MRI BOLD signal are evident up to 1 min before absence seizure onset (Tangwiriyasakul et al., 2018), does this large time difference in early pre-ictal changes represent another key difference between human and animal models because of their potentially different neurovascular coupling? More generally, does it derive from our lack of fully understanding the intricate relationship between electrical and BOLD signals (Goense and Logothetis, 2008), and/or is it a reflection of the fact that BOLD changes in humans with epilepsy and in animal models may not necessarily correlate with, or even relate to, abnormal neuronal activity (Lemieux et al., 2011)?
The early pre-ictal changes of absence seizures are likely to be tightly linked to the presence of a persistently, i.e. interictally, altered cortical sensorimotor network in humans with absence seizures compared to healthy controls (Tangwiriyasakul et al., 2018). Future clinical studies should confirm this finding in larger drug-free cohorts of CAE, whereas the existence and location of similarly abnormal networks during the interictal periods should be investigated in absence seizure models with electrical and/or non-invasive imaging approaches.
Mechanism of absence seizure initiation
Whereas the ictogenesis and the temporal dynamics of single-cell interactions underlying cortico-thalamo-cortical network activity in the cycle-by-cycle maintenance of SWDs have started to be unravelled in non-anaesthetized animal models (McCafferty et al., 2018b; Meyer et al., 2018), we still lack knowledge of the cellular, synaptic and network mechanisms that lead to absence seizure initiation in the CIN. Because of what has been discussed in the previous point, it is of paramount importance that future studies in animal models must strictly take into account the fact that the dynamic state of single neurons and neuronal networks of the CIN before the start of an electrographic and behavioural absence seizure is not identical to that of the same neurons and networks in a control (i.e. non-epileptic) animal. Moreover, investigations of absence seizure initiation should carefully consider the different location of the CIN among animal models (Meeren et al., 2002; Studer et al., 2018; Lee et al., 2019) and between models and children with absence seizures (Bai et al., 2010; Guo et al., 2016; Tangwiriyasakul et al., 2018).
Understanding the absence seizure initiation process will undoubtedly be a massive and technically highly demanding task that will require an intelligent combination of recording the firing dynamics of identified (or identifiable) neurons from CIN neuronal ensembles in non-anaesthetized animal models coupled with a systematic investigation of subthreshold oscillations and passive and active membrane properties of the various neuronal populations in CIN slices. Some patchy experimental evidence does exist but was obtained in cortical areas other than the CIN or under anaesthetic conditions (D’Arcangelo et al., 2002; Polack et al., 2007; Chipaux et al., 2011). Moreover, while we know that layer 5/6 pyramidal neurons in the CIN of absence models have different intrinsic membrane properties (Kole et al., 2007; Polack et al., 2007) and morphological features (van Luijtelaar and Sitnikova, 2016) than non-epileptic animals, similar information is not available for the different classes of CIN interneurons. Furthermore, there are no data on potential abnormalities of synaptic innervation, strength and plasticity among excitatory and inhibitory neurons in the CIN of any absence seizure model. Indeed, some of the key differences that exist in normal animals between parvalbumin- and somatostatin-positive cortical interneurons (Ascoli et al., 2008) may be altered in the CIN of absence seizure models. Notably, selective inhibition by DREADDS of cortical parvalbumin-positive interneurons has been shown to elicit absence-like seizures in normal mice (Panthi and Leitch, 2019) and genetic ablation of Cav2.1 channels in both parvalbumin- and somatostatin-positive cortical interneurons elicit severe generalized seizures and absence seizures (Rossignol et al., 2013). However, whether and how somatostatin-positive interneurons, the main mediators of the dendritic inhibition-dependent regulation of input-output transformations in pyramidal neurons (Lovett-Barron et al., 2012; Wilson et al., 2012; Lee et al., 2013), may contribute to absence seizure generation remains to be established. Moreover, somatostatin-positive interneurons in the somatosensory cortex are active during quiet wakefulness, the brain state where most absence seizures are expressed (Halász, 2015) and their silencing increases burst firing in cortical pyramidal neurons (Gentet et al., 2012).
Mechanism of absence seizure termination
The mechanism underlying the termination of absence seizures has not been directly investigated. Changes in Granger causality values among the activity of cortical and subcortical regions (Lüttjohann et al., 2014) as well as changes in the firing of cortical, thalamic and basal ganglia neuronal populations just around SWD offset have been proposed as potential contributors to absence seizure termination (Paz et al., 2005, 2007; McCafferty et al., 2018b) but no causal evidence is available.
From a biophysical perspective, as elegantly summarized by Barthó et al. (2014), the length of a SWD (as any other spontaneous transient oscillation) could either be predetermined by the state of the generating network at the start of the SWD or depend on the arrival of a signal (intrinsic or extrinsic to the generating network) at any time after the SWD has started. In the former case, the activity of each SWD follows a fixed trajectory and thus the time of its termination is set by the initial state of the network, whereas in the latter case there is no correlation between the initial and the end state of the SWD-generating network and thus the length (and time of termination) of a SWD. Understanding which of these two scenarios applies to SWD is of paramount importance as it will help to guide more detailed research towards either the key elements that set the conditions of the SWD-generating network at the start of a SWD or the intrinsic and extrinsic perturbations that bring to a stop the activity of the SWD-generating network. Notably, the correlation between EEG-functional MRI data and cognitive impairments during absence seizure in children with CAE has been shown to be already set at the very start of a seizure (Guo et al., 2016).
Because of the hypothesis linking natural sleep spindles (a spontaneous transient oscillation) and SWD of absence seizure (Avoli and Gloor, 1981; Kostopoulos et al., 1981; but see Leresche et al., 2012), currently proposed mechanisms of sleep spindle termination might apply to SWD, though spindles are driven by a thalamic pacemaker with critical cortical modulation (Steriade et al., 1987; Crunelli et al., 2018; Adamantidis et al., 2019; Fernandez and Luthi, 2020) whereas absence seizures require the contribution of many different neuronal populations in cortico-thalamo-cortical and basal ganglia networks. Thus, increased intracellular Ca2+ leading to thalamocortical neuron depolarization (Lüthi and McCormick, 1998; Lüthi et al., 1998), desynchronization of the cortical output to the thalamus (Bonjean et al., 2011) and progressive hyperpolarization of NRT neurons (Bal et al., 1995b; Kim and McCormick, 1998) coupled with a marked drop in their firing just a few cycles before spindle termination (Barthó et al., 2014), that have been suggested as potential mechanisms for spindle termination, might also be involved in controlling the duration of SWD. Notably, the latter mechanism has been shown to be predetermined by the state of the network at the start of the spindle oscillation in normal (i.e. non-epileptic) rats (Barthó et al., 2014).
Finally, the slowing of the rhythmic activity in both excitatory and inhibitory neurons towards the end of partial (focal) seizures has been suggested to involve a progressive increase in synchrony among participating neurons and to play a role in seizure termination (de Curtis and Avoli, 2015). As a slowing of the SWD frequency is observed within many absence seizure (Fig. 1A) (Panayiotopoulos, 1999; Blumenfeld, 2005b; Matricardi et al., 2014) it could be that an increased synchronization of cortical excitatory and inhibitory networks in the CIN may also be critical for absence seizure termination.
Mechanism of absence seizure pharmaco-resistance
If we want to prevent the adverse effects of polytherapy, we need to understand the mechanisms underlying pharmaco-resistant absence seizures. As these seizures are genetically determined, a key goal would be to increase efforts in investigating differences in genetic abnormalities between responders and non-responders to currently available anti-absence medications in large cohorts. For experimental research, the priority would be to develop animal models that are insensitive to ethosuximide and valproate. Such a development may be guided by the findings in humans that (i) a more negative treatment outcome, independently of the initial treatment (with either ethosuximide, valproic acid or lamotrigine), occurs for absence seizure without face/limb motor automatisms (Kessler et al., 2017); (ii) ethosuximide responders have decreased parietal cortex connectivity for ictal low frequency EEG components whereas non-responders have increased frontal cortex connectivity for high frequencies (Tenney et al., 2018); and (iii) a CACNA1H polymorphism (P640L) is more commonly expressed in children non-responding to ethosuximide monotherapy (Glauser et al., 2017).
Mechanism and treatment of absence seizure comorbidities
A major future research task relates to the inability of current therapies to effectively control absence seizure comorbidities since we have no indication as to whether absence seizures and comorbidities share initial common genetic and mechanistic pathways that once fully developed become independent from each other (Sharp et al., 2008; Fejgin et al., 2014; Noebels, 2015b; Nilsson et al., 2016). Clinically, the persistence of interictally altered cortical networks (Tangwiriyasakul et al., 2018) calls for routine pre-symptomatic functional MRI and neuropsychiatric analysis for early detection of abnormal cortical network functions in children and teenagers at risk of developing absence seizures. Experimentally, the neuropsychiatric features of absence seizure models should be systematically investigated to understand the underlying neurobiological mechanisms and assess potential treatments. At present, the only drug that has been shown to abolish absence seizures and ameliorate memory deficits in animal models is the pan-T-channel blocker Z944 (Marks et al., 2016a; Henbid et al., 2017): selective antagonists for one T-channel subtype may prove more effective than pan T-channel blockers in controlling both absence seizure and associated comorbidity (Powell et al., 2014a).
Valproic acid-induced comorbidity and continuous neuropsychiatric evaluation
The large incidence of neuropsychiatric comorbidities and the marked increase in attention deficits observed with valproic acid treatment in CAE (Masur et al., 2013) advise against the use of this drug as first monotherapy in children and teenagers with absence seizures. Moreover, the presence of widespread psychiatric comorbidity in individuals with absence seizures makes it of paramount importance to establish closer interactions between neurologists and psychiatrists as part of the standard clinical practice in the initial assessment and continuous follow-up of children and teenagers with CAE, juvenile absence epilepsy and juvenile myoclonic epilepsy. The major efforts that are currently being made towards a better understanding and treatment of attention, cognition and learning deficits and mood disorders (Duman et al., 2016; Postorino et al., 2017; Feldman et al., 2018) will undoubtedly also prove beneficial to children, teenagers and adults with absence seizures.
Acknowledgements
We wish to thank Drs Hal Blumenfeld, Marco de Curtis and Khalid Hamandi for their critical comments and suggestions on an early draft of the manuscript. We are indebted to the children with absence seizures, the University Hospital of Wales in Cardiff, and Drs K. Hamandi and M. Venzi for the EEG recordings shown in Fig. 1A. We also wish to thank the three anonymous referees for their comments and suggestions that greatly helped us improve this review.
Funding
Work on absence seizures in our labs has been or is supported by the Wellcome Trust (grant 91882 to V.C.), the MRC (grant G0900671 to V.C.), the EU (grant ITN Marie Sklodowska-Curie 722053 to V.C.; grant COST Action CM1103 to G.D.), the CNRS (grant LIA 528 to N.L., R.C.L. and V.C.), the Malta Council for Science and Technology MCST R&I (grant R&I-2013-014 to G.D. and V.C.), the Hungarian Scientific Research Fund (grant NN125601 to M.L.L.) and the Hungarian Brain Research Program (grant KTIA_NAP_13-2-2014-0014 to M.L.L.).
Competing interests
The authors report no competing interests.
Glossary
- BOLD =
blood oxygenation level-dependent
- CAE =
childhood absence epilepsy
- CIN =
cortical initiation network
- NRT =
nucleus reticularis thalami
- SNr =
substantia nigra pars reticulata
- SWD =
spike-wave discharges
References
- Adamantidis AR, Gutierrez Herrera C, Gent TC.. Oscillating circuitries in the sleeping brain. Nat Rev Neurosci 2019; 20: 746–62. [DOI] [PubMed] [Google Scholar]
- Aghakhani Y, Bagshaw AP, Bénar CG, Hawco C, Andermann F, Dubeau F, et al. fMRI activation during spike and wave discharges in idiopathic generalized epilepsy. Brain 2004; 127: 1127–44. [DOI] [PubMed] [Google Scholar]
- Aker RG, Yananli HR, Gurbanova AA, Ozkaynakci AE, Ates N, Luijtelaar G, et al. Amygdala kindling in the WAG/Rij rat model of absence epilepsy. Epilepsia 2006; 47: 33–40. [DOI] [PubMed] [Google Scholar]
- Aldenkamp AP, Arends J, Overweg-Plandsoen TC, van Bronswijk KC, Schyns-Soeterboek A, Linden I, et al. Acute cognitive effects of nonconvulsive difficult-to-detect epileptic seizures and epileptiform electroencephalographic discharges. J Child Neurol 2001; 16: 119–23. [DOI] [PubMed] [Google Scholar]
- Antal M, Beneduce BM, Regehr WG.. The substantia nigra conveys target-dependent excitatory and inhibitory outputs from the basal ganglia to the thalamus. J. Neurosci 2014; 34: 8032–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- ANZCTR.org.au #ACTRN12618000961246. Study of PRAX-944 to assess the safety, tTolerability, PK, and efficacy of escalating doses in adults with absence seizures. https://www.anzctr.org.au/Trial/Registration/TrialReview.aspx?id=375174&isReview=true (21 March 2020, date last accessed).
- Ascoli GA, Alonso-Nanclares L, Anderson SA, Barrionuevo G, Benavides-Piccione R, Burkhalter A, et al. Petilla terminology: nomenclature of features of GABAergic interneurons of the cerebral cortex. Nat Rev Neurosci 2008; 9: 557–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Avanzini G, de Curtis M, Marescaux C, Panzica F, Spreafico R, Vergnes M.. Role of the thalamic reticular nucleus in the generation of rhythmic thalamo-cortical activities subserving spike and waves. J Neural Transm Suppl 1992; 35: 85–95. [DOI] [PubMed] [Google Scholar]
- Avoli M. A brief history on the oscillating roles of thalamus and cortex in absence seizures. Epilepsia 2012; 53: 779–89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Avoli M, Gloor P.. The effects of transient functional depression of the thalamus on spindles and on bilateral synchronous epileptic discharges of feline generalized penicillin epilepsy. Epilepsia 1981; 22: 443–52. [DOI] [PubMed] [Google Scholar]
- Bai X, Guo J, Killory B, Vestal M, Berman R, Negishi M, et al. Resting functional connectivity between the hemispheres in childhood absence epilepsy. Neurology 2011; 76: 1960–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bai X, Vestal M, Berman R, Negishi M, Spann M, Vega C, et al. Dynamic time course of typical childhood absence seizures: EEG, behavior, and functional magnetic resonance imaging. J Neurosci 2010; 30: 5884–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bal T, Debay D, Destexhe A.. Cortical feedback controls the frequency and synchrony of oscillations in the visual thalamus. J Neurosci 2000; 20: 7478–88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bal T, von Krosigk M, McCormick DA.. Synaptic and membrane mechanisms underlying synchronized oscillations in the ferret lateral geniculate nucleus in vitro. J Physiol 1995a; 483: 641–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bal T, von Krosigk M, McCormick DA.. Role of the ferret perigeniculate nucleus in the generation of synchronized oscillations in vitro. J Physiol 1995b; 483: 665–85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barthó P, Slézia A, Mátyás F, Faradzs-Zade L, Ulbert I, Harris KD, et al. Ongoing network state controls the length of sleep spindles via inhibitory activity. Neuron 2014; 82: 1367–79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baude A, Nusser Z, Roberts JD, Mulvihill E, McIlhinney RA, Somogyi P.. The metabotropic glutamate receptor (mGluR1 alpha) is concentrated at perisynaptic membrane of neuronal subpopulations as detected by immunogold reaction. Neuron 1993; 11: 771–87. [DOI] [PubMed] [Google Scholar]
- Beckman ML, Bernstein EM, Quick MW.. Multiple G protein-coupled receptors initiate protein kinase C redistribution of GABA transporters in hippocampal neurons. J Neurosci 1999; 19: RC9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benuzzi F, Mirandola L, Pugnaghi M, Farinelli V, Tassinari CA, Capovilla G, et al. Increased cortical BOLD signal anticipates generalized spike and wave discharges in adolescents and adults with idiopathic generalized epilepsies. Epilepsia 2012; 53: 622–30. [DOI] [PubMed] [Google Scholar]
- Berenyi A, Belluscio M, Mao D, Buzsaki G.. Closed-loop control of epilepsy by transcranial electrical stimulation. Science 2012; 337: 735–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berg AT, Levy SR, Testa FM, Blumenfeld H.. Long-term seizure remission in childhood absence epilepsy: might initial treatment matter? Epilepsia 2014; 55: 551–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berman R, Negishi M, Vestal M, Spann M, Chung MH, Bai X, et al. Simultaneous EEG, fMRI, and behavior in typical childhood absence seizures. Epilepsia 2010; 51: 2011–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bessaïh T, Bourgeais L, Badiu CI, Carter DA, Toth TI, Ruano D, et al. Nucleus-specific abnormalities of GABAergic synaptic transmission in a genetic model of absence seizures. J Neurophysiol 2006; 96: 3074–81. [DOI] [PubMed] [Google Scholar]
- Bezprozvanny I, Tsien RW.. Voltage-dependent blockade of diverse types of voltage-gated Ca2+ channels expressed in Xenopus oocytes by the Ca2+ channel antagonist mibefradil (Ro 40-5967). Mol Pharmacol 1995; 48: 540–9. [PubMed] [Google Scholar]
- Blumenfeld H. Consciousness and epilepsy: why are patients with absence seizures absent? Prog. Prog Brain Res 2005a; 150: 271–86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blumenfeld H. Cellular and network mechanisms of spike-wave seizures. Epilepsia 2005b; 46: 21–33. [DOI] [PubMed] [Google Scholar]
- Blumenfeld H, Klein JP, Schridde U, Vestal M, Rice T, Khera DS, et al. Early treatment suppresses the development of spike-wave epilepsy in a rat model. Epilepsia 2008; 49: 400–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blumenfeld H, McCormick D. A.. Corticothalamic inputs control the pattern of activity generated in thalamocortical networks. J Neurosci 2000; 20: 5153–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bombardi C, Venzi M, Crunelli V, Di Giovanni G.. Developmental changes of GABA immunoreactivity in cortico-thalamic networks of an absence seizure model. Neuropharmacology 2018; 136: 56–67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bomben VC, Aiba I, Qian J, Mark MD, Herlitze S, Noebels JL.. Isolated P/Q calcium channel deletion in layer VI corticothalamic neurons generates absence epilepsy. J Neurosci 2016; 36: 405–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bonjean M, Baker T, Lemieux M, Timofeev I, Sejnowski T, Bazhenov M.. Corticothalamic feedback controls sleep spindle duration in vivo. J Neurosci 2011; 31: 9124–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bouilleret V, Hogan RE, Velakoulis D, Salzberg MR, Wang L, Egan GF, et al. Morphometric abnormalities and hyperanxiety in genetically epileptic rats: a model of psychiatric comorbidity? Neuroimage 2009; 45: 267–74. [DOI] [PubMed] [Google Scholar]
- Bui AD, Alexander A, Soltesz I.. Seizing control. Neuroscientist 2017; 23: 68–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buzsáki G. The thalamic clock: emergent network properties. Neuroscience 1991; 41: 351–64. [DOI] [PubMed] [Google Scholar]
- Cain SM, Tyson JR, Choi H-B, Ko R, Lin PJC, LeDue JM, et al. CaV3.2 drives sustained burst-firing, which is critical for absence seizure propagation in reticular thalamic neurons. Epilepsia 2018; 59: 778–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Camfield C, Camfield P.. Management guidelines for children with idiopathic generalized epilepsy. Epilepsia 2005; 46: 112–6. [DOI] [PubMed] [Google Scholar]
- Caplan R, Levitt J, Siddarth P, Wu KN, Gurbani S, Sankar R, et al. Frontal and temporal volumes in childhood absence epilepsy. Epilepsia 2009; 50: 2466–72. [DOI] [PubMed] [Google Scholar]
- Caplan R, Siddarth P, Stahl L, Lanphier E, Vona P, Gurbani S, et al. Childhood absence epilepsy: behavioral, cognitive, and linguistic comorbidities. Epilepsia 2008; 49: 1838–46. [DOI] [PubMed] [Google Scholar]
- Carney PW, Masterton RA, Flanagan D, Berkovic SF, Jackson GD.. The frontal lobe in absence epilepsy: EEG-fMRI findings. Neurology 2012; 78: 1157–65. [DOI] [PubMed] [Google Scholar]
- Carney PW, Masterton RAJ, Harvey AS, Scheffer IE, Berkovic SF, Jackson GD.. The core network in absence epilepsy: differences in cortical and thalamic BOLD response. Neurology 2010; 75: 904–11. [DOI] [PubMed] [Google Scholar]
- Carpentier A, Canney M, Vignot A, Reina V, Beccaria K, Horodyckid C, et al. Clinical trial of blood-brain barrier disruption by pulsed ultrasound. Sci Transl Med 2016; 8: 343re2. [DOI] [PubMed] [Google Scholar]
- Cebrián C, Parent A, Prensa L.. Patterns of axonal branching of neurons of the substantia nigra pars reticulata and pars lateralis in the rat. J Comp Neurol 2005; 492: 349–69. [DOI] [PubMed] [Google Scholar]
- Celli R, Santolini I, Van Luijtelaar G, Ngomba RT, Bruno V, Nicoletti F.. Targeting metabotropic glutamate receptors in the treatment of epilepsy: rationale and current status. Expert Opin Ther Targets 2019; 23: 341–51. [DOI] [PubMed] [Google Scholar]
- Cerminara C, Coniglio A, El-Malhany N, Casarelli L, Curatolo P.. Two epileptic syndromes, onebrain: childhood absence epilepsy and benign childhood epilepsy with centrotemporal spikes. Seizure 2012; 21: 70–4. [DOI] [PubMed] [Google Scholar]
- Charpier S, Leresche N, Deniau J-M, Mahon S, Hugues SW, Crunelli V.. On the putative contribution of GABAB receptors to the electrical events occuring during spontaneous spike and wave discharges. Neuropharmacology 1999; 38: 1699–706. [DOI] [PubMed] [Google Scholar]
- Chen Y, Lu J, Pan H, Zhang Y, Wu H, Xu K, et al. Association between genetic variation of CACNA1H and childhood absence epilepsy. Ann Neurol 2003; 54: 239–43. [DOI] [PubMed] [Google Scholar]
- Cheng D, Yan X, Gao Z, Xu K, Zhou X, Chen Q.. Neurocognitive profiles in childhood absence epilepsy. J Child Neurol 2017; 32: 46–52. [DOI] [PubMed] [Google Scholar]
- Cheong E, Zheng Y, Lee K, Lee J, Kim S, Sanati M, et al. Deletion of phospholipase C B4 in thalamocortical relay nucleus leads to absence seizures. Proc Natl Acad Sci USA 2009; 106: 21912–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chipaux M, Charpier S, Polack PO.. Chloride-mediated inhibition of the ictogenic neurones initiating genetically-determined absence seizures. Neuroscience 2011; 192: 642–51. [DOI] [PubMed] [Google Scholar]
- Christenson Wick Z, Krook-Magnuson E.. Specificity, versatility, and continual development: the power of optogenetics for epilepsy research. Front Cell Neurosci 2018; 12: 151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- ClinicalTrials.gov #NCT03336242. Cannabidiol oral solution in pediatric participants with treatment-resistant childhood absence seizures. https://clinicaltrials.gov/ct2/show/NCT03336242 (18 March 2020, date last accessed).
- ClinicalTrials.gov #NCT03355300. Long-term safety of pharmaceutical grade synthetic cannabidiol oral solution in pediatric participants with treatment-resistant childhood absence seizures. https://clinicaltrials.gov/ct2/show/NCT03355300 (21 March 2020, date last accessed).
- ClinicalTrials.gov #NCT03406702. A Phase 2 study of CX-8998 in adolescents and adults with idiopathic generalized epilepsy with absence seizures (T-WAVE). https://clinicaltrials.gov/ct2/show/NCT03406702 (21 March 2020, date last accessed).
- Cnaan A, Shinnar S, Arya R, Adamson PC, Clark PO, Dlugos D, et al. Second monotherapy in childhood absence epilepsy. Neurology 2017; 88: 182–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Connelly WM, Errington AC, Giovanni Di, Crunelli G. V. Metabotropicregulation of extrasynaptic GABAA receptors. Front Neural Circuits 2013a; 7: 171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Connelly WM, Fyson SJ, Errington AC, McCafferty CP, Cope DW, Di Giovanni G, et al. GABAB receptors regulate extra synaptic GABAA receptors. J Neurosci 2013b; 33: 3780–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Connelly WM, Crunelli V, Errington AC.. The global spike: conserved dendritic properties enable unique Ca2+ spike generation in low- threshold spiking neurons. J Neurosci 2015; 35: 15505–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Connelly WM, Crunelli V, Errington AC.. Passive synaptic normalization and input synchrony-dependent amplification of cortical feedback in thalamocortical neuron dendrites. J Neurosci 2016; 36: 3735–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Connelly WM, Crunelli V, Errington AC.. Variable action potential back propagation during tonic firing and low-threshold spike bursts in thalamocortical but not thalamic reticular nucleus neurons. J Neurosci 2017; 37: 5319–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Contreras D, Curró Dossi R, Steriade M.. Electrophysiological properties of cat reticular thalamic neurones in vivo. J Physiol 1993; 470: 273–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cope DW, Di Giovanni G, Fyson SJ, Orbán G, Errington AC, Lorincz ML, et al. Enhanced tonic GABAA inhibition in typical absence epilepsy. Nat Med 2009; 15: 1392–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cope DW, Hughes SW, Crunelli V.. GABAA receptor-mediated tonic inhibition in thalamic neurons. J Neurosci 2005; 25: 11553–563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crunelli V, Haby M, Jassik-Gerschenfeld D, Leresche N, Pirchio M.. Cl- and K+ dependent inhibitory synaptic potentials evoked by interneurones of the rat lateral geniculate nucleus. J Physiol 1988; 399: 153–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crunelli V, Leresche N.. A role for GABAB receptors in excitation and inhibition of thalamocortical cells. Trends Neurosci 1991; 14: 16–21. [DOI] [PubMed] [Google Scholar]
- Crunelli V, Leresche N.. Block of thalamic T-type Ca2+ channels by ethosuximide is not the whole story. Epilepsy Curr 2002a; 2: 53–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crunelli V, Leresche N.. Childhood absence epilepsy: genes, channels, neurons and networks. Nat Rev Neurosci 2002b; 3: 371–82. [DOI] [PubMed] [Google Scholar]
- Crunelli V, Lörincz M, Connelly W, David F, Hughes S, Lambert RC, et al. Dual function of thalamic low-vigilance state oscillations: rhythm-regulation and plasticity. Nat Rev Neurosci 2018; 19: 107–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Curtis M, Avoli M.. Initiation, propagation, and termination of partial (focal) seizures. Cold Spring Harb Perspect Med 2015; 5: a022368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- D’Amore V, Von Randow C, Nicoletti F, Ngomba RT, Van Luijtelaar G.. Anti-absence activity of mGlu1 and mGlu5 receptor enhancers and their interaction with a GABA reuptake inhibitor: effect of local infusions in the somatosensory cortex and thalamus. Epilepsia 2015; 56: 1141–51. [DOI] [PubMed] [Google Scholar]
- D’Arcangelo G, D’Antuono M, Biagini G, Warren R, Tancredi V, Avoli M.. Thalamocortical oscillations in a genetic model of absence seizures. Eur J Neurosci 2002; 16: 2383–93. [DOI] [PubMed] [Google Scholar]
- David O., Guillemain I., Saillet S., Reyt S., Deransart C., Segebarth C, et al. Identifying neural drivers with functional MRI: an electrophysiological validation. PLoS Biol 2008; 6: 2683–97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- David F, Çarçak N, Furdan S, Onat F, Gould T, Mészáros Á, et al. Suppression of hyperpolarization-activated cyclic nucleotide-gated channel function in thalamocortical neurons prevents genetically determined and pharmacologically induced absence seizures. J Neurosci 2018; 38: 6615–27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dedeurwaerdere S, Boon P, De Smedt T, Claeys P, Raedt R, Bosman T, et al. Chronic levetiracetam treatment early in life decreases epileptiform events in young GAERS, but does not prevent the expression of spike and wave discharges during adulthood. Seizure 2005; 14: 403–11. [DOI] [PubMed] [Google Scholar]
- Deng YP, Xie JP, Wang HB, Lei WL, Chen Q, Reiner A.. Differential localization of the GluR1 and GluR2 subunits of the AMPA-type glutamate receptor among striatal neuron types in rats. J Chem Neuroanat 2007; 33: 167–92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Depaulis A, Bourguignon JJ, Marescaux C, Vergnes M, Schmitt M, Micheletti G, et al. Effects of gamma-hydroxybutyrate and gamma-butyrolactone derivatives on spontaneous generalized non-convulsive seizures in the rat. Neuropharmacology 1988a; 27: 683–9. [DOI] [PubMed] [Google Scholar]
- Depaulis A, Blumenfeld H, Charpier S, Coulter D, Crunelli V, Frankel W, et al. et al. Loss of consciousness during absence seizures. J Neurosci 2017; 37: 5861–9. https://www.jneurosci.org/content/37/24/5861/tab-e-letters.28522734 [Google Scholar]
- Depaulis A, van Luijtelaar G.. Characteristics of genetic absence seizures in the rat In: Pitkanen A, Schwartzkroin PA, Moshe S, editors. Models of seizure and epilepsy. International: Elsevier Academic Press; 2006. p. 233–48. [Google Scholar]
- Depaulis A, Snead OC, Marescaux C, Vergnes M.. Suppressive effects of intranigral injection of muscimol in three models of generalized non-convulsive epilepsy induced by chemical agents. Brain Res 1989; 498: 64–72. [DOI] [PubMed] [Google Scholar]
- Depaulis A, Vergnes M, Marescaux C, Lannes B, Warter JM.. Evidence that activation of GABA receptors in the substantia nigra suppresses spontaneous spike-and-wave discharges in the rat. Brain Res 1988b; 448: 20–29. [DOI] [PubMed] [Google Scholar]
- Deransart C, Hellwig B, Heupel-Reuter M, Léger J-F, Heck D, Lücking CH.. Single-unit analysis of substantia nigra pars reticulata neurons in freely behaving rats with genetic absence epilepsy. Epilepsia 2003; 44: 1513–20. [DOI] [PubMed] [Google Scholar]
- Deransart C, Lê-Pham BT, Hirsch E, Marescaux C, Depaulis A.. Inhibition of the substantia nigra suppresses absences and clonic seizures in audiogenic rats, but not tonic seizures: evidence for seizure specificity of the nigral control. Neuroscience 2001; 105: 203–11. [DOI] [PubMed] [Google Scholar]
- Deransart C, Marescaux C, Depaulis A.. Involvement of nigral glutamatergic inputs in the control of seizures in a genetic model of absence epilepsy in the rat. Neuroscience 1996; 71: 721–8. [DOI] [PubMed] [Google Scholar]
- Deransart C, Riban V, Lê BT, Marescaux C, Depaulis A.. Dopamine in the striatum modulates seizures in a genetic model of absence epilepsy in the rat. Neuroscience 2000; 100: 335–44. [DOI] [PubMed] [Google Scholar]
- De Deurwaerdère P, Ramsay RR, Di Giovanni G.. Neurobiology and neuropharmacology of monoaminergic systems. Prog Neurobiol 2017; 151: 1–3. [DOI] [PubMed] [Google Scholar]
- Devinsky O, Nabbout R, Miller I, Laux L, Zolnowska M, Wright S, et al. Long-termcannabidiol treatment in patients with Dravet syndrome: An open-label extension trial. Epilepsia 2019; 60: 294–302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dezsi G, Ozturk E, Salzberg MR, Morris M, O’Brien TJ, Jones NC.. Environmental enrichment imparts disease-modifying and transgenerational effects on genetically-determined epilepsy and anxiety. Neurobiol Dis 2016; 93: 129–136. [DOI] [PubMed] [Google Scholar]
- Dezsi G, Ozturk E, Stanic D, Powell KL, Blumenfeld H, O’Brien TJ, et al. Ethosuximide reduces epileptogenesis and behavioral comorbidity in the GAERS model of genetic generalized epilepsy. Epilepsia 2013; 54: 635–643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Di Giovanni G,, De Deurwaerdère P.. New therapeutic opportunities for 5-HT2C receptor ligands in neuropsychiatric disorders. Pharmacol Ther 2016; 157: 125–62. [DOI] [PubMed] [Google Scholar]
- Dikow N, Maas B, Karch S, Granzow M, Janssen JWG, Jauch A, et al. 3p25.3 microdeletion of GABA transporters SLC6A1 and SLC6A11 results in intellectual disability, epilepsy and stereotypic behavior. Am J Med Genet 2014; 164: 3061–8. [DOI] [PubMed] [Google Scholar]
- Dlugos D, Shinnar S, Cnaan A, Hu F, Moshe S, Mizrahi E, et al. Pretreatment EEG in childhood absence epilepsy: associations with attention and treatment outcome. Neurology 2013; 81: 150–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dreyfus FM, Tscherter A, Errington AC, Renger JJ, Shin H-S, Uebele VN, et al. Selective T-type calcium channel block in thalamic neurons reveals channel redundancy and physiological impact of I(T)window. J Neurosci 2010; 30: 99–109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duman RS, Aghajanian GK, Sanacora G, Krystal JH.. Synaptic plasticity and depression: new insights from stress and rapid-acting antidepressants. Nat Med 2016; 22: 238–49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Errington AC, Cope DW, Crunelli V.. Augmentation of tonic GABA A inhibition in absence epilepsy: therapeutic value of inverse agonists at extrasynaptic GABA A receptors. Adv Pharmacol Sci 2011a; 2011: 790590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Errington AC, Gibson KM, Crunelli V, Cope DW.. Aberrant GABAA receptor-mediated inhibition in cortico-thalamic networks of succinic semialdehyde dehydrogenase deficient mice. PLoS One 2011b; 6: e19021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Errington AC, Di Giovanni G, Crunelli V, Cope DW.. mGluR control of interneuron output regulates feed forward tonic GABAA inhibition in the visual thalamus. J Neurosci 2011c; 31: 8669–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ettinger AB, Bernal OG, Andriola MR, Bagchi S, Flores P, Just C, et al. Two cases of nonconvulsive status epilepticus in association with tiagabine therapy. Epilepsia 1999; 40: 1159–62. [DOI] [PubMed] [Google Scholar]
- Fedio P, Mirsky AF.. Selective intellectual deficits in children with temporal lobe or centrencephalic epilepsy. Neuropsychologia 1969; 7: 287–300. [Google Scholar]
- Fejgin K, Nielsen J, Birknow MR, Bastlund JF, Nielsen V, Lauridsen JB, et al. A mouse model that recapitulates cardinal features of the 15q13.3 microdeletion syndrome including schizophrenia- and epilepsy-related alterations. Biol Psychiatry 2014; 76: 128–37. [DOI] [PubMed] [Google Scholar]
- Feldman L, Lapin B, Busch RM, Bautista JF.. Evaluating subjective cognitive impairment in the adult epilepsy clinic: effects of depression, number of antiepileptic medications, and seizure frequency. Epilepsy Behav 2018; 81: 18–24. [DOI] [PubMed] [Google Scholar]
- Fernandez LMJ, Luthi A.. Sleep spindles: mechanisms and functions. Physiol Rev 2020;100: 805–68. [DOI] [PubMed] [Google Scholar]
- Fisher RS, Cross JH, French JA, Higurashi N, Hirsch E, Jansen FE, et al. Operational classification of seizure types by the international league against epilepsy: position paper of the ILAE commission for classification and terminology. Epilepsia 2017; 58: 522–30. [DOI] [PubMed] [Google Scholar]
- François C, Tande D, Yelnik J, Hirsch EC.. Distribution and morphology of nigral axons projecting to the thalamus in primates. J Comp Neurol 2002; 447: 249–60. [DOI] [PubMed] [Google Scholar]
- Freestone DR, Karoly PJ, Cook MJ.. A forward-looking review of seizure prediction. Curr Opin Neurol 2017; 30: 167–73. [DOI] [PubMed] [Google Scholar]
- Gadhoumi K, Lina J-M, Mormann F, Gotman J.. Seizure prediction for therapeutic devices: a review. J Neurosci Methods 2016; 260: 270–82. [DOI] [PubMed] [Google Scholar]
- Gardiner M. Genetics of Idiopathic Generalized Epilepsies. Epilepsia 2005; 46: 15–20. [DOI] [PubMed] [Google Scholar]
- Gencpinar P, Kalay Z, Turgut S, Bozkurt Ö, Duman Ö, Ozel D, et al. Evaluation of executive functions in patients with childhood absence epilepsy. J Child Neurol 2016; 31: 824–30. [DOI] [PubMed] [Google Scholar]
- Gentet LJ, Kremer Y, Taniguchi H, Huang ZJ, Staiger JF, Petersen C.. Unique functional properties of somatostatin-expressing GABAergic neurons in mouse barrel cortex. Nat Neurosci 2012; 15: 607–12. [DOI] [PubMed] [Google Scholar]
- Gervasi N, Monnier Z, Vincent P, Paupardin-Tritsch D, Hugues SW, Crunelli V, et al. Pathways-specific action of γ-hydroxybutyricacid in sensory thalamus and its relevance to absence seizures. J Neurosc 2003; 23: 11469–78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giber K, Diana MA, Plattner VM, Dugué GP, Bokor H, Rousseau CV, et al. A subcortical inhibitory signal for behavioral arrest in the thalamus. Nat Neurosci 2015; 18: 562–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giber K, Slézia A, Bokor H, Bodor AL, Ludányi A, Katona I, et al. Heterogeneous output pathways link the anterior pretectal nucleus with the zona incerta and the thalamus in rat. J Comp Neurol 2008; 506: 122–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Glauser T, Cnaan A, Shinnar S, Hirtz DG, Dlugos D, Masur D, et al. Ethosuximide, valproic acid, and lamotrigine in childhood absence epilepsy. N Engl J Med 2010; 362: 790–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Glauser TA, Cnaan A, Shinnar S, Hirtz DG, Dlugos D, Masur D, et al. Ethosuximide, valproic acid, and lamotrigine in childhood absence epilepsy: initial monotherapy outcomes at 12 months. Epilepsia 2013; 54: 141–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Glauser TA, Holland K, O’Brien VP, Keddache M, Martin LJ, Clark PO, et al. Pharmacogenetics of antiepileptic drug efficacy in childhood absence epilepsy. Ann Neurol 2017; 81: 444–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goense JBM, Logothetis NK.. Neurophysiology of the BOLD fMRI signal in awake monkeys. Curr Biol 2008; 18: 631–40. [DOI] [PubMed] [Google Scholar]
- Golshani P, Liu XB, Jones EG.. Differences in quantal amplitude reflect GluR4- subunit number at corticothalamic synapses on two populations of thalamic neurons. Proc Natl Acad Sci USA 2001; 98: 4172–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gomora JC, Enyeart JA, Enyeart JJ.. Mibefradil potently blocks ATP-activated K+ channels in adrenal cells. Mol Pharmacol 1999; 56: 1192–7. [DOI] [PubMed] [Google Scholar]
- Gotman J, Grova C, Bagshaw A, Kobayashi E, Aghakhani Y, Dubeau F.. Generalized epileptic discharges show thalamocortical activation and suspension of the default state of the brain. Proc Natl Acad Sci USA 2005; 102: 15236–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gould T, Chen L, Emri Z, Pirttimaki T, Errington AC, Crunelli V, et al. GABA(B) receptor-mediated activation of astrocytes by gamma-hydroxybutyricacid. Philos Trans R Soc Lond B Biol Sci 2014; 369: 20130607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guerrini R, Noebels J.. How can advances in epilepsy genetics lead to better treatments and cures? Adv Exp Med Biol 2014; 813: 309–17. [DOI] [PubMed] [Google Scholar]
- Guo JN, Kim R, Chen Y, Negishi M, Jhun S, Weiss S, et al. Impaired consciousness in patients with absence seizures investigated by functional MRI, EEG, and behavioural measures: a cross-sectional study. Lancet Neurol 2016; 15: 1336–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gupta D, Ossenblok P, van Luijtelaar G.. Space-time network connectivity and cortical activations preceding spike wave discharges in human absence epilepsy: a MEG study. Med Biol Eng Comput 2011; 49: 555–65. [DOI] [PubMed] [Google Scholar]
- Halassa MM, Chen Z, Wimmer RD, Brunetti PM, Zhao S, Zikopoulos B, et al. State-dependent architecture of thalamic reticular subnetworks. Cell 2014; 158: 808–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Halász P. Are absence epilepsy and nocturnal frontal lobe epilepsy system epilepsies of the sleep/wake system? Behav Neurol 2015; 2015: 231676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hamandi K, Salek-Haddadi A, Laufs H, Liston A, Friston K, Fish DR, et al. EEG–fMRI of idiopathic and secondarily generalized epilepsies. Neuroimage 2006; 31: 1700–10. [DOI] [PubMed] [Google Scholar]
- Hammelmann V, Stieglitz MS, Hülle H, Le Meur K, Kass J, Brümmer M, et al. Abolishing cAMP sensitivity in HCN2 pacemaker channels induces generalized seizures. JCI Insight 2019; 4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henbid MT, Marks WN, Collins MJ, Cain SM, Snutch TP, Howland JG.. Sociability impairments in genetic absence epilepsy rats from Strasbourg: reversal by the T-type calcium channel antagonist Z944. Exp Neurol 2017; 296: 16–22. [DOI] [PubMed] [Google Scholar]
- Henkin Y, Sadeh M, Kivity S, Shabtai E, Kishon-Rabin L, Gadoth N.. Cognitive function in idiopathic generalized epilepsy of childhood. Dev Med Child Neurol 2005; 47: 126–32. [DOI] [PubMed] [Google Scholar]
- Hermann B, Jones J, Dabbs K, Allen CA, Sheth R, Fine J, et al. The frequency, complications and aetiology of ADHD in new onset paediatric epilepsy. Brain 2007; 130: 3135–48. [DOI] [PubMed] [Google Scholar]
- Hertoft P. The clinical, electroencephalographic and social prognosis in petit mal epilepsy. Epilepsia 1963; 4: 298–314. [DOI] [PubMed] [Google Scholar]
- Holmes GL, Noebels JL.. The epilepsy spectrum: targeting future research challenges. Cold Spring Harb Perspect Med 2016; 6: a028043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holmes MD, Brown M, Tucker DM.. Are ‘generalized’ seizures truly generalized? Evidence of localized mesial frontal and frontopolar discharges in absence. Epilepsia 2004; 45: 1568–79. [DOI] [PubMed] [Google Scholar]
- Huang H-Y, Lee H-W, Chen S-D, Shaw F-Z.. Lamotrigine ameliorates seizures and psychiatric comorbidity in a rat model of spontaneous absence epilepsy. Epilepsia 2012; 53: 2005–14. [DOI] [PubMed] [Google Scholar]
- Huntsman MM, Porcello D, Homanics GE, DeLorey TM, Huguenard JR.. Reciprocal inhibitory connections and network synchrony in the mammalian thalamus. Science 1999; 283: 541–3. [DOI] [PubMed] [Google Scholar]
- Jafarian M, Karimzadeh F, Alipour F, Attari F, Lotfinia AA, Speckmann EJ, et al. Cognitive impairments and neuronal injury in different brain regions of a genetic rat model of absence epilepsy. Neuroscience 2015; 298: 161–70. [DOI] [PubMed] [Google Scholar]
- Jambaqué I, Dellatolas G, Dulac O, Ponsot G, Signoret JL.. Verbal and visual memory impairment in children with epilepsy. Neuropsychologia 1993; 31: 1321–37. [DOI] [PubMed] [Google Scholar]
- Jarre G, Altwegg-Boussac T, Williams MS, Studer F, Chipaux M, David O, et al. Building up absence seizures in the somatosensory cortex: from network to cellular epileptogenic processes. Cereb Cortex 2017; 27: 4607–23. [DOI] [PubMed] [Google Scholar]
- Johannesen KM, Gardella E, Linnankivi T, Courage C, de Saint Martin A, Lehesjoki AE, et al. Defining the phenotypic spectrum of SLC6A1 mutations. Epilepsia 2018; 59: 389–402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones EG. The thalamus, 1st edn New York: Plenum; 2003. [Google Scholar]
- Jones JE, Watson R, Sheth R, Caplan R, Koehn M, Seidenberg M, et al. Psychiatric comorbidity in children with new onset epilepsy. Dev Med Child Neurol 2007; 49: 493–7. [DOI] [PubMed] [Google Scholar]
- Jones NC, Salzberg MR, Kumar G, Couper A, Morris MJ, O’Brien TJ.. Elevated anxiety and depressive-like behavior in a rat model of genetic generalized epilepsy suggesting common causation. Exp Neurol 2008; 209: 254–60. [DOI] [PubMed] [Google Scholar]
- Karson A, Utkan T, Balcı F, Arıcıoğlu F, Ateş N.. Age-dependent decline in learning and memory performances of WAG/Rij rat model of absence epilepsy. Behav Brain Funct 2012; 8: 51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keezer MR, Sisodiya SM, Sander JW.. Comorbidities of epilepsy: current concepts and future perspectives. Lancet Neurol 2016; 15: 106–15. [DOI] [PubMed] [Google Scholar]
- Kessler SK, Shinnar S, Cnaan A, Dlugos D, Conry J, Hirtz DG, et al. Pretreatment seizure semiology in childhood absence epilepsy. Neurology 2017; 89: 673–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khosravani H, Altier C, Simms B, Hamming KS, Snutchll TP, Mezeyova J, et al. Gating effects of mutations in the Cav3.2 T-type calcium channel associated with childhood absence epilepsy. J Biol Chem 2004; 279: 9681–4. [DOI] [PubMed] [Google Scholar]
- Killory BD, Bai X, Negishi M, Vega C, Spann MN, Vestal M, et al. Impaired attention and network connectivity in childhood absence epilepsy. Neuroimage 2011; 56: 2209–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim U, McCormick DA.. Functional and ionic properties of a slow afterhyperpolarization in ferret perigeniculate neurons in vitro. J Neurophysiol 1998; 80: 1222–35. [DOI] [PubMed] [Google Scholar]
- Kole MHP, Bräuer AU, Stuart GJ.. Inherited cortical HCN1 channel loss amplifies dendritic calcium electrogenesis and burst firing in a rat absence epilepsy model. J Physiol 2007; 578: 507–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kostopoulos G, Gloor P, Pellegrini A, Gotman J.. A study of the transition from spindles to spike and wave discharge in feline generalized penicillin epilepsy: microphysiological features. Exp Neurol 1981; 73: 55–77. [DOI] [PubMed] [Google Scholar]
- Kratochvil Z, Mccafferty C, Herman P, Ryu J, Sanganahalli B, Gruenbaum B, et al. Awake fMRI in a rat model of absence epilepsy. Society for Neuroscience meeting, San Diego (CA), 2018, Abstract 561.12. Available at: https://www.abstractsonline.com/pp8/#!/4649/presentation/25847.
- Krause MR, Vieira PG, Csorba BA, Pilly PK, Pack CC.. Transcranial alternating current stimulation entrains single-neuron activity in the primate brain. Proc Natl Acad Sci USA 2019; 116: 5747–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krause MR, Zanos TP, Csorba BA, Pilly PK, Choe J, Phillips ME, et al. Transcranial direct current stimulation facilitates associative learning and alters functional connectivity in the primate brain. Curr Biol 2017; 27: 3086–96.e3. [DOI] [PubMed] [Google Scholar]
- von Krosigk M, Bal T, McCormick DA.. Cellular mechanisms of a synchronized oscillation in the thalamus. Science 1993; 261: 361–4. [DOI] [PubMed] [Google Scholar]
- Kwan P, Arzimanoglou A, Berg AT, Brodie MJ, Allen Hauser W, Mathern G, et al. Definition of drug resistant epilepsy: consensus proposal by the ad hoc task force of the ILAE commission on therapeutic strategies. Epilepsia 2009; 51: 1069–77. [DOI] [PubMed] [Google Scholar]
- Lacey CJ, Bryant A, Brill J, Huguenard JR.. Enhanced NMDA receptor-dependent thalamic excitation and network oscillations in stargazer mice. J Neurosci 2012; 32: 11067–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leal A, Vieira JP, Lopes R, Nunes RG, Gonçalves SI, Lopes da Silva F, et al. Dynamics of epileptic activity in a peculiar case of childhood absence epilepsy and correlation with thalamic levels of GABA. Epilepsy Behav Case Rep 2016; 5: 57–65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee H-J, Kim E-H, Yum M-S, Ko T-S, Kim H-W.. Attention profiles in childhood absence epilepsy compared with attention-deficit/hyperactivity disorder. Brain Dev 2018; 40: 94–9. [DOI] [PubMed] [Google Scholar]
- Lee S, Hwang E, Lee M, Choi JH.. Distinct topographical patterns of spike-wave discharge in transgenic and pharmacologically induced absence seizure models. Exp Neurobiol 2019; 28: 474–84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee S, Kruglikov I, Huang ZJ, Fishell G, Rudy B.. A disinhibitory circuit mediates motor integration in the somatosensory cortex. Nat Neurosci 2013; 16: 1662–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lemieux L, Daunizeau J, Walker MC.. Concepts of connectivity and human epileptic activity. Front Syst Neurosci 2011; 5: 12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leresche N, Lambert RC, Errington AC, Crunelli V.. From sleep spindles of natural sleep to spike and wave discharges of typical absence seizures: is the hypothesis still valid? Pflugers Arch 2012; 463: 201–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leresche N, Lambert RC.. T-type calcium channels in synaptic plasticity. Channels 2017; 11: 121–39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leresche N, Parri HR, Erdemli G, Guyon A, Turner JP, Williams SR, et al. On the action of the anti-absence drug ethosuximide in the rat and cat thalamus. J Neurosci 1998; 18: 4842–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Q, Cao W, Liao X, Chen Z, Yang T, Gong Q, et al. Altered resting state functional network connectivity in children absence epilepsy. J Neurol Sci 2015; 354: 79–85. [DOI] [PubMed] [Google Scholar]
- Li Q, Luo C, Yang T, Yao Z, He L, Liu L, et al. EEG-fMRI study on the interictal and ictal generalized spike-wave discharges in patients with childhood absence epilepsy. Epilepsy Res 2009; 87: 160–8. [DOI] [PubMed] [Google Scholar]
- Lipsman N, Meng Y, Bethune AJ, Huang Y, Lam B, Masellis M, et al. Blood–brain barrier opening in Alzheimer’s disease using MR-guided focused ultrasound. Nat Commun 2018; 9: 2336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu A, Vöröslakos M, Kronberg G, Henin S, Krause MR, Huang Y, et al. Immediate neurophysiological effects of transcranial electrical stimulation. Nat Commun 2018; 9: 5092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu JH, Bijlenga P, Occhiodoro T, Fischer-Lougheed J, Bader CR, Bernheim L.. Mibefradil (Ro 40-5967) inhibits several Ca2+ and K+ currents in human fusion-competent myoblasts. Br J Pharmacol 1999; 126: 245–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Loiseau P, Dartigues JF, Pestre M.. Prognosis of partial epileptic seizures in the adolescent. Epilepsia 1983; 24: 472–81. [DOI] [PubMed] [Google Scholar]
- Loring DW, Marino S, Meador KJ.. Neuropsychological and behavioral effects of antiepilepsy drugs. Neuropsychol Rev 2007; 17: 413–25. [DOI] [PubMed] [Google Scholar]
- Lovett-Barron M, Turi GF, Kaifosh P, Lee PH, Bolze F, Sun X-H, et al. Regulation of neuronal input transformations by tunable dendritic inhibition. Nat Neurosci 2012; 15: 423–30. [DOI] [PubMed] [Google Scholar]
- van Luijtelaar G, Behr C, Avoli M.. Is there such a thing as “generalized” epilepsy? Adv Exp Med Biol 2014; 813: 81–91. [DOI] [PubMed] [Google Scholar]
- van Luijtelaar G, Mishra AM, Edelbroek P, Coman D, Frankenmolen N, Schaapsmeerders P, et al. Anti-epileptogenesis: electrophysiology, diffusion tensor imaging and behavior in a genetic absence model. Neurobiol Dis 2013; 60: 126–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Luijtelaar G, Sitnikova E.. Global and focal aspects of absence epilepsy: the contribution of genetic models. Neurosci Biobehav Rev 2006; 30: 983–1003. [DOI] [PubMed] [Google Scholar]
- Luo C, Xia Y, Li Q, Xue K, Lai Y, Gong Q, et al. Diffusion and volumetry abnormalities in subcortical nuclei of patients with absence seizures. Epilepsia 2011; 52: 1092–9. [DOI] [PubMed] [Google Scholar]
- Luo C, Yang T, Tu S, Deng J, Liu D, Li Q, et al. Altered intrinsic functional connectivity of the salience network in childhood absence epilepsy. J Neurol Sci 2014; 339: 189–95. [DOI] [PubMed] [Google Scholar]
- Lüthi A, Bal T, McCormick DA.. Periodicity of thalamic spindle waves is abolished by ZD7288, a blocker of Ih. J Neurophysiol 1998; 79: 3284–9. [DOI] [PubMed] [Google Scholar]
- Lüthi A, McCormick DA.. Periodicity of thalamic synchronized oscillations: the role of Ca2+-mediated upregulation of Ih. Neuron 1998; 20: 553–63. [DOI] [PubMed] [Google Scholar]
- Lüttjohann A, Van Luijtelaar G.. Dynamics of networks during absence seizure’s on- and offset in rodents and man. Front Physiol 2015; 6: 16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lüttjohann A, Pape H-C.. Regional specificity of cortico-thalamic coupling strength and directionality during waxing and waning of spike and wave discharges. Sci Rep 2019; 9: 2100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lüttjohann A, Schoffelen J-M, van Luijtelaar G.. Peri-ictal network dynamics of spike-wave discharges: phase and spectral characteristics. Exp Neurol 2013; 239: 235–47. [DOI] [PubMed] [Google Scholar]
- Lüttjohann A, Schoffelen J-M, van Luijtelaar G.. Termination of ongoing spike-wave discharges investigated by cortico-thalamic network analyses. Neurobiol Dis 2014; 70: 127–37. [DOI] [PubMed] [Google Scholar]
- Lüttjohann A, Zhang S, de Peijper R, van Luijtelaar G.. Electrical stimulation of the epileptic focus in absence epileptic WAG/Rij rats: assessment of local and network excitability. Neuroscience 2011; 188: 125–34. [DOI] [PubMed] [Google Scholar]
- Maheshwari A, Akbar A, Wang M, Marks RL, Yu K, Park S, et al. Persistent aberrant cortical phase-amplitude coupling following seizure treatment in absence epilepsy models. J Physiol 2017; 595: 7249–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maksimenko VA, van Heukelum S, Makarov VV, Kelderhuis J, Lüttjohann A, Koronovskii AA, et al. Absence seizure control by a brain computer interface. Sci Rep 2017; 7: 2487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manning J, Richards DA, Leresche N, Crunelli V, Bowery NG.. Cortical-area specific block of genetically determined absence seizures by ethosuximide. Neuroscience 2004; 123: 5–9. [DOI] [PubMed] [Google Scholar]
- Markham MR, Butt AE, Dougher MJ.. A computer touch-screen apparatus for training visual discriminations in rats. J Exp Anal Behav 1996; 65: 173–82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marks WN, Cain SM, Snutch TP, Howland JG.. The T-type calcium channel antagonist Z944 rescues impairments in crossmodal and visual recognition memory in genetic absence epilepsy rats from Strasbourg. Neurobiol Dis 2016a; 94: 106–15. [DOI] [PubMed] [Google Scholar]
- Marks WN, Cavanagh ME, Greba Q, Cain SM, Snutch TP, Howland JG.. The genetic absence epilepsy rats from Strasbourg model of absence epilepsy exhibits alterations in fear conditioning and latent inhibition consistent with psychiatric comorbidities in humans. Eur J Neurosci 2016b; 43: 25–40. [DOI] [PubMed] [Google Scholar]
- Marks WN, Zabder NK, Greba Q, Cain SM, Snutch TP, Howland JG.. The T‐type calcium channel blocker Z944 reduces conditioned fear in Genetic Absence Epilepsy Rats from Strasbourg and the non‐epileptic control strain. Eur J Neurosci 2019; 50: 3046–59. [DOI] [PubMed] [Google Scholar]
- Marques-Carneiro JE, Faure J-B, Barbelivien A, Nehlig A, Cassel J-C.. Subtle alterations in memory systems and normal visual attention in the GAERS model of absence epilepsy. Neuroscience 2016; 316: 389–401. [DOI] [PubMed] [Google Scholar]
- Marques-Carneiro JE, Faure JB, Cosquer B, Koning E, Ferrandon A, De Vasconcelos AP, et al. Anxiety and locomotion in Genetic Absence Epilepsy Rats from Strasbourg (GAERS): inclusion of Wistar rats as a second control. Epilepsia 2014; 55: 1460–68. [DOI] [PubMed] [Google Scholar]
- Masterton RA., Carney PW., Abbott DF., Jackson GD Absence epilepsy subnetworks revealed by event-related independent components analysis of functional magnetic resonance imaging. Epilepsia2013; 54: 801–8. [DOI] [PubMed] [Google Scholar]
- Masur D, Shinnar S, Cnaan A, Shinnar RC, Clark P, Wang J, et al. Pretreatment cognitive deficits and treatment effects on attention in childhood absence epilepsy. Neurology 2013; 81: 1572–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matricardi S, Verrotti A, Chiarelli F, Cerminara C, Curatolo P.. Current advances in childhood absence epilepsy. Pediatr Neurol 2014; 50: 205–12. [DOI] [PubMed] [Google Scholar]
- Mattison KA, Butler KM, Inglis GAS, Dayan O, Boussidan H, Bhambhani V, et al. SLC6A1 variants identified in epilepsy patients reduce γ-aminobutyric acid transport. Epilepsia 2018; 59: e135–41. [DOI] [PubMed] [Google Scholar]
- McCafferty C, Connelly WM, Celli R, Ngomba RT, Nicoletti F, Crunelli V.. Genetic rescue of absence seizures. CNS Neurosci Ther 2018a; 24: 745–58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCafferty CP, David F, Venzi M, Lorincz ML, Delicata F, Atherton Z, et al. Cortical drive and thalamic feed-forward inhibition control thalamic output synchrony during absence seizures. Nat. Neurosci 2018b; 21: 744–56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCormick DA, Contreras D.. On the cellular and network bases of epileptic seizures. Annu Rev Physiol 2001; 63: 815–46. [DOI] [PubMed] [Google Scholar]
- Meeren HKM, Pijn JPM, Van Luijtelaar E, Coenen AML, Lopes da Silva FH.. Cortical focus drives widespread corticothalamic networks during spontaneous absence seizures in rats. J Neurosci 2002; 22: 1480–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meeren HKM, Veening JG, Möderscheim TAE, Coenen AML, van Luijtelaar G.. Thalamic lesions in a genetic rat model of absence epilepsy: dissociation between spike-wave discharges and sleep spindles. Exp Neurol 2009; 217: 25–37. [DOI] [PubMed] [Google Scholar]
- Meyer J, Maheshwari A, Noebels J, Smirnakis S.. Asynchronous suppression of visual cortex during absence seizures in stargazer mice. Nat Commun 2018; 9:1938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mishra AM, Ellens DJ, Schridde U, Motelow JE, Purcaro MJ, DeSalvo MN, et al. Where fMRI and electrophysiology agree to disagree: corticothalamic and striatal activity patterns in the WAG/Rij rat. J Neurosci 2011; 31: 15053–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mishra SK, Hermsmeyer K.. Selective inhibition of T-type Ca2+ channels by Ro 40-5967. Circ Res 1994; 75: 144–8. [DOI] [PubMed] [Google Scholar]
- Miskowiak KW, Petersen CS.. Neuronal underpinnings of cognitive impairment and - improvement in mood disorders. CNS Spectr 2019; 24: 30–53. [DOI] [PubMed] [Google Scholar]
- Miyamoto H, Tatsukawa T, Shimohata A, Yamagata T, Suzuki T, Amano K, et al. Impaired cortico-striatal excitatory transmission triggers epilepsy. Nat Commun 2019; 10: 1917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miyamoto Y, Jinnai K.. The inhibitory input from the substantia nigra to the mediodorsal nucleus neurons projecting to the prefrontal cortex in the cat. Brain Res 1994; 649: 313–8. [DOI] [PubMed] [Google Scholar]
- Moeller F, Levan P, Muhle H, Stephani U, Dubeau F, Siniatchkin M, et al. Absence seizures: Individual patterns revealed by EEG-fMRI. Epilepsia 2010; 51: 2000–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moeller F, Muthuraman M, Stephani U, Deuschl G, Raethjen J, Siniatchkin M.. Representation and propagation of epileptic activity in absences and generalized photoparoxysmal responses. Hum Brain Mapp 2013; 34: 1896–909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moeller F, Siebner HR, Wolff S, Muhle H, Granert O, Jansen O, et al. Simultaneous EEG-fMRI in drug-naive children with newly diagnosed absence epilepsy. Epilepsia 2008; 49: 1510–9. [DOI] [PubMed] [Google Scholar]
- Moeller F, Tyvaert L, Nguyen DK, LeVan P, Bouthillier A, Kobayashi E, et al. EEG-fMRI: adding to standard evaluations of patients with nonlesional frontal lobe epilepsy. Neurology 2009; 73: 2023–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moyanova S, De Fusco A, Santolini I, Celli R, Bucci D, Mastroiacovo F, et al. Abnormal hippocampal melatoninergic system: a potential link between absence epilepsy and depression-like behavior in WAG/Rij rats? Int J Mol Sci 2018; 19: 1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Munoz F, Aurup C, Konofagou EE, Ferrera VP.. Modulation of brain function and behavior by focused ultrasound. Curr Behav Neurosci Rep 2018; 5: 153–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nilius B, Prenen J, Kamouchi M, Viana F, Voets T, Droogmans G.. Inhibition by mibefradil, a novel calcium channel antagonist, of Ca2+- and volume-activated Cl- channels in macrovascular endothelial cells. Br J Pharmacol 1997; 121: 547–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nilsson SRO, Celada P, Fejgin K, Thelin J, Nielsen J, Santana N, et al. A mouse model of the 15q13.3 microdeletion syndrome shows prefrontal neurophysiological dysfunctions and attentional impairment. Psychopharmacology (Berl) 2016; 233: 2151–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Noebels J. Pathway-driven discovery of epilepsy genes. Nat Neurosci 2015a; 18: 344–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Noebels JL. Single-gene determinants of epilepsy comorbidity. Cold Spring Harb Perspect Med 2015b; 5: a022756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Noebels JL, Qiao X, Bronson RT, Spencer C, Davisson MT.. Stargazer: a new neurological mutant on chromosome 15 in the mouse with prolonged cortical seizures. Epilepsy Res 1990; 7: 129–35. [DOI] [PubMed] [Google Scholar]
- Nolan MA, Redoblado MA, Lah S, Sabaz M, Lawson JA, Cunningham AM, et al. Memory function in childhood epilepsy syndromes. J Paediatr Child Health 2004; 40: 20–7. [DOI] [PubMed] [Google Scholar]
- Ott D, Caplan R, Guthrie D, Siddarth P, Komo S, Shields WD, et al. Measures of psychopathology in children with complex partial seizures and primary generalized epilepsy with absence. J Am Acad Child Adolesc Psychiatry 2001; 40: 907–14. [DOI] [PubMed] [Google Scholar]
- Ounsted C, Hutt SJ, Lee D.. The retrograde amnesia of petit mal. Lancet (London, England) 1963; 1: 671. [DOI] [PubMed] [Google Scholar]
- Panayiotopoulos CP. Typical absence seizures and their treatment. Arch Dis Child 1999; 81: 351–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Panayiotopoulos CP. Typical absence seizures and related epileptic syndromes: assessment of current state and directions for future research. Epilepsia 2008; 49: 2131–9. [DOI] [PubMed] [Google Scholar]
- Panthi S, Leitch B.. The impact of silencing feed-forward parvalbumin-expressing inhibitory interneurons in the cortico-thalamocortical network on seizure generation and behaviour. Neurobiol. Dis 2019; 132: 104610. [DOI] [PubMed] [Google Scholar]
- Park C, Rosenblat JD, Lee Y, Pan Z, Cao B, Iacobucci M, et al. The neural systems of emotion regulation and abnormalities in major depressive disorder. Behav Brain Res 2019; 367: 181–8. [DOI] [PubMed] [Google Scholar]
- Parthasarathy HB, Graybiel AM.. Cortically driven immediate-early gene expression reflects modular influence of sensorimotor cortex on identified striatal neurons in the squirrel monkey. J Neurosci 1997; 17: 2477–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pavone P, Bianchini R, Trifiletti RR, Incorpora G, Pavone A, Parano E.. Neuropsychological assessment in children with absence epilepsy. Neurology 2001; 56: 1047–51. [DOI] [PubMed] [Google Scholar]
- Paz JT, Chavez M, Saillet S, Deniau J-M, Charpier S.. Activity of ventral medial thalamic neurons during absence seizures and modulation of cortical paroxysms by the nigrothalamic pathway. J Neurosci 2007; 27: 929–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paz JT, Deniau JM, Charpier S.. Rhythmic bursting in the cortico-subthalamo-pallidal network during spontaneous genetically determined spike and wave discharges. J Neurosci 2005; 25: 2092–101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paz JT, Huguenard JR.. Microcircuits and their interactions in epilepsy: is the focus out of focus? Nat Neurosci 2015; 18: 351–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Penovich PE, Willmore L.. Use of a new antiepileptic drug or an old one as first drug for treatment of absence epilepsy. Epilepsia 2009; 50: 37–41. [DOI] [PubMed] [Google Scholar]
- Perucca E, Gram L, Avanzini G, Dulac O.. Antiepileptic drugs as a cause of worsening seizures. Epilepsia 1998; 39: 5–17. [DOI] [PubMed] [Google Scholar]
- Pigeat R, Chausson P, Dreyfus FM, Leresche N, Lambert RC.. Sleep slow wave-related homo and heterosynaptic LTD of intrathalamic GABAAergic synapses: involvement of T-type Ca2+ channels and metabotropic glutamate receptors. J. Neurosci 2015; 35: 64–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pinault D. Cellular interactions in the rat somatosensory thalamocortical system during normal and epileptic 5-9 Hz oscillations. J Physiol 2003; 552: 881–905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pinault D. The thalamic reticular nucleus: Structure, function and concept. Brain Res Brain Res Rev 2004; 46: 1–31. [DOI] [PubMed] [Google Scholar]
- Pinault D, Leresche N, Charpier S, Deniau JM, Marescaux C, Vergnes M, et al. Intracellular recordings in thalamic neurones during spontaneous spike and wave discharges in rats with absence epilepsy. J Physiol 1998; 509: 449–56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pinault D, Slézia A, Acsády L.. Corticothalamic 5-9 Hz oscillations are more pro-epileptogenic than sleep spindles in rats. J Physiol 2006; 574: 209–27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- PitkäNen A, Buckmaster PS, Galanopoulou AS, Moshé SL.. Models of seizures and epilepsy. Cambridge: Academic Press; 2017. [Google Scholar]
- Pirttimaki T, Parri HR, Crunelli V.. Astrocytic GABA transporter GAT-1 dysfunction in experimental absence seizures. J Physiol 2013; 591: 823–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Polack PO, Mahon S, Chavez M, Charpier S.. Inactivation of the somatosensory cortex prevents paroxysmal oscillations in cortical and related thalamic neurons in a genetic model of absence epilepsy. Cereb Cortex 2009; 19: 2078–91. [DOI] [PubMed] [Google Scholar]
- Polack P-O, Guillemain I, Hu E, Deransart C, Depaulis A, Charpier S.. Deep layer somatosensory cortical neurons initiate spike-and-wave discharges in a genetic model of absence seizures. J. Neurosci 2007; 27: 6590–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ponnusamy R, Pradhan N.. The effects of chronic administration of ethosuximide on learning and memory: a behavioral and biochemical study on nonepileptic rats. Behav Pharmacol 2006; 17: 573–80. [DOI] [PubMed] [Google Scholar]
- Postorino V, Kerns CM, Vivanti G, Bradshaw J, Siracusano M, Mazzone L.. Anxiety disorders and obsessive-compulsive disorder in individuals with autism spectrum disorder. Curr Psychiatry Rep 2017; 19: 92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pouliopoulos A, Kwon N, Hussani S, Konofagou A.. Non-invasive optogenetic excitation using focused-ultrasound-mediated delivery of virus-encoded Chrimson and transcranial red-light exposure. Society for Neuroscience meeting San Diego (CA)2018; Abstract 255.19. Available at: https://www.abstractsonline.com/pp8/#!/4649/presentation/3386. [Google Scholar]
- Powell KL, Cain SM, Ng C, Sirdesai S, David LS, Kyi M, et al. A Cav3.2 T-type calcium channel point mutation has splice-variant-specific effects on function and segregates with seizure expression in a polygenic rat model of absence epilepsy. J Neurosci 2009; 29: 371–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Powell KL, Cain SM, Snutch TP, O’Brien TJ.. Low threshold T-type calcium channels as targets for novel epilepsy treatments. Br J Clin Pharmacol 2014a; 77: 729–39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Powell KL, Tang H, Ng C, Guillemain I, Dieuset G, Dezsi G, et al. Seizure expression, behavior, and brain morphology differences in colonies of genetic absence epilepsy rats from Strasbourg. Epilepsia 2014b; 55: 1959–68. [DOI] [PubMed] [Google Scholar]
- Qiu W, Gao Y, Yu C, Miao A, Tang L, Huang S, et al. Structural abnormalities in childhood absence epilepsy: voxel-based analysis using diffusion tensor imaging. Front Hum Neurosci 2016; 10: 483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ray JP, Price JL.. The organization of the thalamocortical connections of the mediodorsal thalamic nucleus in the rat, related to the ventral forebrain–prefrontal cortex topography. J Comp Neurol 1992; 323: 167–97. [DOI] [PubMed] [Google Scholar]
- Reteig LC, Talsma LJ, van Schouwenburg MR, Slagter HA.. Transcranial electrical stimulation as a tool to enhance attention. J Cogn Enhanc 2017; 1: 10–25. [Google Scholar]
- Richards D. A, Lemos T, Whitton PS, Bowery NG.. Extracellular GABA in the ventrolateral thalamus of rats exhibiting spontaneous absence epilepsy: a microdialysis study. J Neurochem 1995; 65: 1674–80. [DOI] [PubMed] [Google Scholar]
- Richards DA, Manning JPA, Barnes D, Rombola L, Bowery NG, Caccia S, et al. Targeting thalamic nuclei is not sufficient for the full anti-absence action of ethosuximide in a rat model of absence epilepsy. Epilepsy Res 2003; 54: 97–107. [DOI] [PubMed] [Google Scholar]
- Romano C, Sesma MA, McDonald CT, O’Malley K, Van den Pol AN, Olney JW.. Distribution of metabotropic glutamate receptor mGluR5 immunoreactivity in rat brain. J Comp Neurol 1995; 355: 455–69. [DOI] [PubMed] [Google Scholar]
- Rossignol E, Kruglikov I, van den Maagdenberg AM, Rudy B, Fishell G.. CaV 2.1 ablation in cortical interneurons selectively impairs fast-spiking basket cells and causes generalizedseizures. Ann Neurol 2013; 74: 209–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Russo E, Citraro R, Scicchitano F, De Fazio S, Perrota I, Di Paola ED, et al. Effects of early long-term treatment with antiepileptic drugs on development of seizures and depressive-like behavior in a rat genetic absence epilepsy model. Epilepsia 2011; 52: 1341–50. [DOI] [PubMed] [Google Scholar]
- Sakai ST, Grofova I, Bruce K.. Nigrothalamic projections and nigrothalamocortical pathway to the medial agranular cortex in the rat: single- and double-labeling light and electron microscopic studies. J Comp Neurol 1998; 391: 506–25. [DOI] [PubMed] [Google Scholar]
- Sarkisova KY, Kuznetsova GD, Kulikov MA, van Luijtelaar G.. Spike-wave discharges are necessary for the expression of behavioral depression-like symptoms. Epilepsia 2010; 51: 146–60. [DOI] [PubMed] [Google Scholar]
- Scheffer IE, Berkovic S, Capovilla G, Connolly MB, French J, Guilhoto L, et al. ILAE classification of the epilepsies: position paper of the ILAE commission for classification and terminology. Epilepsia 2017; 58: 512–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmitt LI, Wimmer RD, Nakajima M, Happ M, Mofakham S, Halassa MM.. Thalamic amplification of cortical connectivity sustains attentional control. Nature 2017; 545: 219–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schreibman Cohen A, Daley M, Siddarth P, Levitt J, Loesch IK, Altshuler L, et al. Amygdala volumes in childhood absence epilepsy. Epilepsy Behav 2009; 16: 436–41. [DOI] [PubMed] [Google Scholar]
- Sharp AJ, Mefford HC, Li K, Baker C, Skinner C, Stevenson RE, et al. A recurrent 15q13.3 microdeletion syndrome associated with mental retardation and seizures. Nat Genet 2008; 40: 322–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shaw F-Z, Chuang S-H, Shieh K-R, Wang Y-J.. Depression- and anxiety-like behaviors of a rat model with absence epileptic discharges. Neuroscience 2009; 160: 382–93. [DOI] [PubMed] [Google Scholar]
- Shinnar RC, Shinnar S, Cnaan A, Clark P, Dlugos D, Hirtz DG, et al. Pretreatment behavior and subsequent medication effects in childhood absence epilepsy. Neurology 2017; 89: 1698–706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shipe WD, Barrow JC, Yang Z-Q, Lindsley CW, Yang FV, Schlegel K-A, et al. Design, synthesis, and evaluation of a novel 4-aminomethyl-4-fluoropiperidine as a T-type Ca2+ channel antagonist. J Med Chem 2008; 51: 3692–5. [DOI] [PubMed] [Google Scholar]
- Sieber AR, Min R, Nevian T.. Non-Hebbian long-term potentiation of inhibitory synapses in the thalamus. J Neurosci 2013; 33: 15675–85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sieu L-A, Bergel A, Tiran E, Deffieux T, Pernot M, Gennisson J-L, et al. EEG and functional ultrasound imaging in mobile rats. Nat Methods 2015; 12: 831–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sitnikova E, Rutskova EM, Raevsky VV.. Maternal care affects EEG properties of spike-wave seizures (including pre- and post ictal periods) in adult WAG/Rij rats with genetic predisposition to absence epilepsy. Brain Res Bull 2016; 127: 84–91. [DOI] [PubMed] [Google Scholar]
- Slaght SJ, Leresche N, Deniau J-M, Crunelli V, Charpier S.. Activity of thalamic reticular neurons during spontaneous genetically determined spike and wave discharges. J Neurosci 2002; 22: 2323–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Slaght SJ, Paz T, Chavez M, Deniau J-M, Mahon S, Charpier S.. On the activity of the corticostriatal networks during spike-and-wave discharges in a genetic model of absence epilepsy. J Neurosci 2004; 24: 6816–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Snead OC. Pharmacological models of generalized absence seizures in rodents In: Pitkanen A, Schwartzkroin PA, Moshe S, editors. Journal of neural transmission. Supplementum. Cambridge: Elsevier Academic Press; 1992. p. 7–19. [DOI] [PubMed] [Google Scholar]
- Sohal VS, Keist R, Rudolph U, Huguenard JR.. Dynamic GABA(A) receptor subtype-specific modulation of the synchrony and duration of thalamic oscillations. J Neurosci 2003; 23: 3649–57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sorokin JM, Davidson TJ, Frechette E, Abramian AM, Deisseroth K, Huguenard JR, et al. Bidirectional control of generalized epilepsy networks via rapid real-time switching of firing mode. Neuron 2017; 93: 194–210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sorokin JM, Paz JT, Huguenard JR.. Absence seizure susceptibility correlates with pre-ictal β oscillations. J Physiol Paris 2016; 110: 372–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steriade M, Contreras D.. Relations between cortical and thalamic cellular events during transition from sleep patterns to paroxysmal activity. J Neurosci 1995; 15: 623–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steriade M, Domich L, Oakson G, Deschenes M.. The deafferented reticular thalamic nucleus generates spindle rhythmicity. J Neurophysiol 1987; 57: 260–73. [DOI] [PubMed] [Google Scholar]
- Stockings E, Zagic D, Campbell G, Weier M, Hall WD, Nielsen S, et al. Evidence for cannabis and cannabinoids for epilepsy: a systematic review of controlled and observational evidence. J Neurol Neurosurg Psychiatry 2018; 89: 741–53. [DOI] [PubMed] [Google Scholar]
- Studer F, Laghouati E, Jarre G, David O, Pouyatos B, Depaulis A.. Sensory coding is impaired in rat absence epilepsy. J Physiol 2018; 597: JP277297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sysoeva MV, Sitnikova E, Sysoev IV, Bezruchko BP, van Luijtelaar G.. Application of adaptive nonlinear Granger causality: disclosing network changes before and after absence seizure onset in a genetic rat model. J Neurosci Methods 2014; 226: 33–41. [DOI] [PubMed] [Google Scholar]
- Sysoeva MV, Vinogradova LV, Kuznetsova GD, Sysoev IV, van Rijn CM.. Changes in corticocortical and corticohippocampal network during absence seizures in WAG/Rij rats revealed with time varying Granger causality. Epilepsy Behav 2016a; 64: 44–50. [DOI] [PubMed] [Google Scholar]
- Sysoeva MV, Lüttjohann A, van Luijtelaar G, Sysoev IV.. Dynamics of directional coupling underlying spike-wave discharges. Neuroscience 2016b; 314: 75–89. [DOI] [PubMed] [Google Scholar]
- Talley EM, Cribbs LL, Lee JH, Daud A, Perez-Reyes E, Bayliss DA.. Differential distribution of three members of a gene family encoding low voltage-activated (T-type) calcium channels. J Neurosci 1999; 19: 1895–911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Talley EM, Solórzano G, Depaulis A, Perez-Reyes E, Bayliss D. A.. Low-voltage-activated calcium channel subunit expression in a genetic model of absence epilepsy in the rat. Brain Res Mol Brain Res 2000; 75: 159–65. [DOI] [PubMed] [Google Scholar]
- Tangwiriyasakul C, Perani S, Centeno M, Yaakub SN, Abela E, Carmichael DW, et al. Dynamic brain network states in human generalized spike-wave discharges. Brain 2018; 141: 2981–994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taylor H, Crunelli V. Optogenetic drive of thalamocortical neurons can block and induce experimental absence seizures in freely moving animals. Society for Neuroscience meeting, Washington (DC) 2014. Abstract 314.07. Available at: https://www.abstractsonline.com/Plan/ViewSession.aspx?sKey=1cc3cd25-a707-4503-a8fb-1ef65cf06c89&mKey=54c85d94-6d69-4b09-afaa-502c0e680ca7.
- Taylor JA, Reuter JD, Kubiak RA, Mufford TT, Booth CJ, Dudek FE, et al. Spontaneous recurrent absence seizure-like events in wild-caught Rats. J Neurosci 2019; 39: 4829–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taylor JA, Rodgers KM, Bercum FM, Booth CJ, Dudek FE, Barth DS.. Voluntary control of epileptiform spike–wave discharges in awake rats. J Neurosci 2017; 37: 5861–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tenney JR, Duong TQ, King JA, Ferris CF.. fMRI of brain activation in a genetic rat model of absence seizures. Epilepsia 2004; 45: 576–82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tenney JR, Duong TQ, King JA, Ludwig R, Ferris CF.. Corticothalamic modulation during absence seizures in rats: a functional MRI assessment. Epilepsia 2003; 44: 1133–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tenney JR, Fujiwara H, Horn PS, Jacobson SE, Glauser TA, Rose DF.. Focal corticothalamic sources during generalized absence seizures: a MEG study. Epilepsy Res 2013; 106: 113–22. [DOI] [PubMed] [Google Scholar]
- Tenney JR, Kadis DS, Agler W, Rozhkov L, Altaye M, Xiang J, et al. Ictal connectivity in childhood absence epilepsy: associations with outcome. Epilepsia 2018; 59: 971–81. [DOI] [PubMed] [Google Scholar]
- Thiele EA, Marsh ED, French JA, Mazurkiewicz-Beldzinska M, Benbadis SR, Joshi C, et al. Cannabidiol in patients with seizures associated with Lennox-Gastaut syndrome (GWPCARE4): arandomised, double-blind, placebo-controlled phase 3 trial. Lancet 2018; 391: 1085–96. [DOI] [PubMed] [Google Scholar]
- Tosun D, Siddarth P, Toga AW, Hermann B, Caplan R.. Effects of childhood absence epilepsy on associations between regional cortical morphometry and aging and cognitive abilities. Hum Brain Mapp 2011; 32: 580–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tringham E, Powell KL, Cain SM, Kuplast K, Mezeyova J, Weerapura M, et al. T-type calcium channel blockers that attenuate thalamic burst firing and suppress absence seizures. Sci Transl Med 2012; 4: 121ra19. [DOI] [PubMed] [Google Scholar]
- Tyvaert L, Chassagnon S, Sadikot A, Levan P, Dubeau F, Gotman J.. Thalamic nuclei activity in idiopathic generalized epilepsy: an EEG-fMRI study. Neurology 2009; 73: 2018–22. [DOI] [PubMed] [Google Scholar]
- Vanasse CM, Béland R, Carmant L, Lassonde M.. Impact of childhood epilepsy on reading and phonological processing abilities. Epilepsy Behav 2005; 7: 288–96. [DOI] [PubMed] [Google Scholar]
- Vaz SH, Jørgensen TN, Cristóvão-Ferreira S, Duflot S, Ribeiro JA, Gether U, et al. Brain-derived neurotrophic factor (BDNF) enhances gaba transport by modulating the trafficking of GABA transporter-1 (GAT-1) from the plasma membrane of rat cortical astrocytes. J Biol Chem 2011; 286: 40464–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vega C, Guo J, Killory B, Danielson N, Vestal M, Berman R, et al. Symptoms of anxiety and depression in childhood absence epilepsy. Epilepsia 2011; 52: e70–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Venderova K, Brown TM, Brotchie JM.. Differential effects of endocannabinoids on [3H]-GABA uptake in the rat globus pallidus. Exp Neurol 2005; 194: 284–7. [DOI] [PubMed] [Google Scholar]
- Venzi M, David F, Bellet J, Cavaccini A, Bombardi C, Crunelli V, et al. Role for serotonin2A (5-HT2A) and 2C (5-HT2C) receptors in experimental absence seizures. Neuropharmacology 2016; 108: 292–304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Venzi M, Giovanni Crunelli DG.. V. A critical evaluation of the gamma-hydroxybutyrate (GHB) model of absence seizures. CNS Neurosci Ther 2015; 21: 123–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vercueil L, Benazzouz A, Deransart C, Bressand K, Marescaux C, Depaulis A, et al. High-frequency stimulation of the subthalamic nucleus suppresses absence seizures in the rat: comparison with neurotoxic lesions. Epilepsy Res 1998; 31: 39–46. [DOI] [PubMed] [Google Scholar]
- Verrotti A, Moavero R, Panzarino G, Di Paolantonio C, Rizzo R, Curatolo P.. The challenge of pharmacotherapy in children and adolescents with epilepsy-ADHD comorbidity. Clin Drug Investig 2018; 38: 1–8. [DOI] [PubMed] [Google Scholar]
- Viana F, Van den Bosch L, Missiaen L, Vandenberghe W, Droogmans G, Nilius B, et al. Mibefradil (Ro 40-5967) blocks multiple types of voltage-gated calcium channels in cultured rat spinal motoneurones. Cell Calcium 1997; 22: 299–311. [DOI] [PubMed] [Google Scholar]
- Wang G, Bochorishvili G, Chen Y, Salvati KA, Zhang P, Dubel SJ, et al. CaV3.2 calcium channels control NMDA receptor-mediated transmission: a new mechanism for absence epilepsy. Genes Dev 2015; 29: 1535–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Westmijse I, Ossenblok P, Gunning B, van Luijtelaar G.. Onset and propagation of spike and slow wave discharges in human absence epilepsy: a MEG study. Epilepsia 2009; 50: 2538–48. [DOI] [PubMed] [Google Scholar]
- Williams J, Sharp GB, DelosReyes E, Bates S, Phillips T, Lange B, et al. Symptom differences in children with absence seizures versus inattention. Epilepsy Behav 2002; 3: 245–8. [DOI] [PubMed] [Google Scholar]
- Williams MS, Altwegg-Boussac T, Chavez M, Lecas S, Mahon S, Charpier S.. Integrative properties and transfer function of cortical neurons initiating absence seizures in a rat genetic model. J Physiol 2016; 594: 6733–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilson NR, Runyan CA, Wang FL, Sur M.. Division and subtraction by distinct cortical inhibitory networks in vivo. Nature 2012; 488: 343–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wirrell EC, Camfield CS, Camfield PR, Dooley JM, Gordon KE, Smith B.. Long-term psychosocial outcome in typical absence epilepsy. Arch Pediatr Adolesc Med 1997; 151: 152–8. [DOI] [PubMed] [Google Scholar]
- Wu C, Xiang J, Sun J, Huang S, Tang L, Miao A, et al. Quantify neuromagnetic network changes from pre-ictal to ictal activities in absence seizures. Neuroscience 2017; 357: 134–44. [DOI] [PubMed] [Google Scholar]
- Wykes RC, Heeroma JH, Mantoan L, Zheng K, MacDonald DC, Deisseroth K, et al. Epilepsy: optogenetic and potassium channel gene therapy in a rodent model of focal neocortical epilepsy. Sci Transl Med 2012; 4: 152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wykes RC, Kullmann DM, Pavlov I, Magloire V.. Optogenetic approaches to treat epilepsy. J Neurosci Methods 2016; 260: 215–20. [DOI] [PubMed] [Google Scholar]
- Yagüe JG, Cavaccini A, Errington AC, Crunelli V, Di Giovanni G.. Dopaminergic modulation of tonic but not phasic GABAA-receptor-mediatedcurrent in the ventrobasal thalamus of Wistar and GAERS rats. Exp Neurol 2013; 247: 1–7. [DOI] [PubMed] [Google Scholar]
- Youngblood MW, Chen WC, Mishra AM, Enamandram S, Sanganahalli BG, Motelow JE, et al. Rhythmic 3-4Hz discharge is insufficient to produce cortical BOLD fMRI decreases in generalized seizures. Neuroimage 2015; 109: 368–77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang W, Bruno RM.. High-order thalamic inputs to primary somatosensory cortex are stronger and longer lasting than cortical inputs. Elife 2019; 8; e44158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zobeiri M, Chaudhary R, Blaich A, Rottmann M, Herrmann S, Meuth P, et al. The hyperpolarization-activated HCN4 channel is important for proper maintenance of oscillatory activity in the thalamocortical system. Cereb Cortex 2019; 29: 2291–304. [DOI] [PMC free article] [PubMed] [Google Scholar]