Abstract
Carotenoid cleavage dioxygenase (CCD), a key enzyme in carotenoid metabolism, cleaves carotenoids to form apo-carotenoids, which play a major role in plant growth and stress responses. CCD genes had not previously been systematically characterized in Brassica napus (rapeseed), an important oil crop worldwide. In this study, we identified 30 BnCCD genes and classified them into nine subgroups based on a phylogenetic analysis. We identified the chromosomal locations, gene structures, and cis-promoter elements of each of these genes and performed a selection pressure analysis to identify residues under selection. Furthermore, we determined the subcellular localization, physicochemical properties, and conserved protein motifs of the encoded proteins. All the CCD proteins contained a retinal pigment epithelial membrane protein (RPE65) domain. qRT-PCR analysis of expression of 20 representative BnCCD genes in 16 tissues of the B. napus cultivar Zhong Shuang 11 (‘ZS11’) revealed that members of the BnCCD gene family possess a broad range of expression patterns. This work lays the foundation for functional studies of the BnCCD gene family.
Introduction
“Carotenoids” is the general term for the class of natural pigments widely present in animals, plants, and microorganisms. Carotenoids are lipid-soluble isoprene-like compounds that contain 40 carbon molecules and comprise more than 750 pigments with different structures [1]. These pigments have numerous important biological functions; for example, they are photo-protective and are indispensable components of photosynthesis [2, 3]. They also can scavenge free radicals and have antioxidant properties [4]. Furthermore, carotenoids are components of cell membranes, potential anti-cancer agents, and can interact with proteins [5]. Carotenoids are exploited as coloring agents in flowers and fruits to attract pollinators and agents of seed dispersal [6–8]. In addition, carotenoids are precursors of important phytohormones, such as abscisic acid and strigolactones, which regulate plant development and plant–environment interactions [9–11].
The carotenoid biosynthesis pathway in plants has been fully elucidated; the catalytic oxidative cracking of carotenoids is a key process in this pathway. Multiple conjugated double bonds exist within the carotenoid center chain, which can be specifically cleaved by carotenoid cleavage dioxygenases (CCDs) to form a variety of apo-carotenoids, and some apo-carotenoids can be further degraded into small biologically active molecules [12]. In plants, the addition of two oxygen atoms to the cleavage product means that CCDs possess the characteristics of a dioxygenase [13]. Moreover, CCDs are also a class of non-heme oxygenases, whose catalytic activity requires Fe2+ as a cofactor [14, 15]. CCD proteins contain four highly conserved histidine residues bound to Fe2+ and all contain a retinal pigment epithelial membrane protein (RPE65) domain that is characteristic of enzymes involved in carotenoid cleavage [16, 17].
In plants, CCD proteins are encoded by an ancient gene family. CCD gene family consists of two subfamilies: carotenoid cleavage dioxygenases (CCDs) and 9-cis epoxycarotenoid dioxygenases (NCEDs) [12]. Through analysis of a novel ABA-deficient mutant of maize, the first protein found to specifically cleave carotenoids, viviparous14 (VP14), was identified by Schwartz et al [14]. Vallabhaneni et al. described the characteristics of the CCD gene family in the grass species maize (Zea mays), rice (Oryza sativa), and sorghum (Sorghum bicolor) [18]. During water stress and seed dormancy, TaNCED of Triticum aestivum might play a primary role in regulation of ABA content [19]. Besides, drought stress can induce expression of NCED3 and accumulation of endogenous ABA in Nicotiana tabacum [20]. Wei et al. found at least seven CCD genes in Solanum lycopersicum genome sequence and analyzed their expression patterns [21]. CCD genes have also been identified or functionally expressed in a variety of other plant species, such as Glycine max [22], Gossypium hirsutum, Solanum tuberosum [23], Cucurbita pepo [24], Saccharum officinarum [25], Crocus sativus, Osmanthus fragrans [26], Vitis vinifera [27], Mangifera indica [28] and Amygdalus persica [29].
In Arabidopsis thaliana, the CCD gene family consists of nine members: four CCD genes (AtCCD1, 4, 7, and 8) and five NCED genes (AtNCED2, 3, 5, 6 and 9) [30]. Carotenoid cleavage dioxygenase homologs in other plant species are named according to the system used for Arabidopsis CCD family members.
The CCD1 and CCD4 enzymes catabolize a variety of carotenoids and produce volatile apo-carotenoids, which are important for the biosynthesis of aromas and flavors of flowers and fruits, respectively [31, 32]. The carotenoid content of mature seeds of AtCCD1 mutants was higher than that in the wild type, indicating a role for CCD1 in carotenoid catabolism [33]. CCD4 regulates carotenoid homeostasis and CmCCD4a is specifically expressed in white petals of Chrysanthemum morifolium [34]. RNAi-mediated inhibition of CmCCD4a expression in white-petaled plants results in the production of yellow flowers [35]. In line with this observation, loss or downregulation of CmCCD4a function led to an increase in carotenoid content in petals [12]. CCD7 and CCD8 function in strigolactone biosynthesis, regulate axillary bud growth, and inhibit branching [36, 37]. Using RNAi to reduce Actinidia chinensis CCD8 expression increases in branch development and delays leaf senescence [38].
The reaction catalyzed by 9-cis-epoxycarotenoid dioxygenase (NCED) is a rate-limiting step in ABA biosynthesis, and thus influences plant tolerance to diverse abiotic stresses [39]. AtNCED5, AtNCED6 and AtNCED9 are the dominant contributors to developmentally regulated ABA synthesis in seeds and thereby regulate seed embryo maturation and dormancy [30]. Overexpression of OsNCED3 increased drought resistance in rice and caused an increased ABA level [40]. AtNCED2 and AtNCED3 transcripts are abundant in Arabidopsis roots and their encoded proteins function in abscisic acid biosynthesis and thereby regulate lateral root growth [30]. Furthermore, heterologous expression of Brassica napus NCED3 led to ABA accumulation and NO and ROS generation in transgenic Arabidopsis plants, thereby enhancing abiotic stress tolerance [41].
B. napus, an important oilseed crop globally, is an allopolyploid derived from a natural interspecific cross between Brassica rapa (turnip; 2n = 2x = 20) and Brassica oleracea (kohlrabi; 2n = 2x = 18). The important physiological functions of carotenoid cleavage products in plants, such as abscisic acid and strigolactones, have prompted studies of the lyases involved in carotenoid metabolism. Little is known about the BnCCD gene family. The availability of the B. napus genome sequence would enable the identification and analysis of members of this family [42].
In this study, we identified 30 BnCCD genes. In addition to analyzing their gene evolution and structure, chromosomal localization, conserved motifs, and cis-acting promoter elements, we determined their tissue- and organ-specific expression and examined the physicochemical properties of their encoded proteins. The results form a solid basis for further studies on the biological functions of the BnCCD gene family.
Materials and methods
Plant materials
The B. napus cultivar Zhong Shuang 11 (‘ZS11’) was planted in Chongqing, China (29°45’ N, 106°22’ E). To analyze the expression patterns of BnCCD genes, four different tissues of ‘ZS11’ were harvested when plants were in full flower: mature leaves (Le), sepals (Se), flowers (F), and stems (St). The seeds (S) and silique pericarps (Sp) were harvested at different timepoints following the termination of flowering (25, 30, 35, 40, 45, and 50 days after flowering). Samples were immediately frozen in liquid nitrogen and stored at –80°C for further use.
Identification of CCD genes in Brassica. napus, Brassica. rapa, and Brassica. oleracea
The coding sequences of AtCCD genes were downloaded from TAIR (https://www.arabidopsis.org/) and used as reference sequences. The BnCCD, BrCCD, and BoCCD genes were identified through the BLASTN analysis [43] of AtCCD genes against the Brassica Database (BRAD, http://brassicadb.org/brad/index.php) [44]. The local database was established using Geneious 4.8.5 software (http://www.geneious.com/; Biomatters, Auckland, New Zealand). The genome-wide alignment was verified by the MEGABLAST program [45], and the following screening standards were used: the consistency of aligned sequences with the reference sequence was ≥80% and gene sequences <700 bp were discarded. Because enzymes involved in carotenoid cleavage contain a retinal pigment epithelial membrane protein (RPE65) domain [16, 17], gene sequences from the Pfam database [46] that did not contain this domain were excluded.
Multiple sequence alignment and phylogenetic analysis
CCD amino acid sequences were subjected to multiple sequence alignment using MUSCLE [47] with default parameters. The evolutionary relationships of B. napus CCD proteins with those of A. thaliana, B. oleracea, and B. rapa were analyzed using MEGA7 [45]. To identify the conserved blocks of all predicted sequences, the Gblocks program was used [48]. The substitution saturation was detected using DAMBE [49]. A phylogenetic tree was constructed using the neighbor-joining method implemented in MEGA7. The number of bootstrap replications was 1,000, and paired deletion was performed. The phylogenetic tree was visualized using FigTree v1.4.2 (http://tree.bio.ed.ac.uk/software/figtree/).
Protein properties, sequence analysis, and duplication time inference
The chromosomal location of BnCCD genes, which was queried from the Brassica napus genome browser, and the genes were mapped onto chromosomal linkage groups by Mapchart software [50]. The Gene Structure Display Server (GSD 2.0) (http://gsds.cbi.pku.edu.cn/index.php) was used to portray the exon–intron structures of the BnCCD genes. The ExPASy proteomics server database (http://expasy.org/) [51]was used to predict the relative molecular weight, theoretical isoelectric point, protein stability, and aliphatic amino acid content of BnCCD proteins. The conserved motifs were identified using the MEME Version 5.0.5 online tool (http://meme-suite.org/tools/meme) [52] and the maximum motif retrieval value was set to 20; other parameters used the default settings. Annotations of the identified motifs were obtained from InterProScan (www.ebi.ac.uk/Tools/InterProScan/) [53].
To determine whether the CCD protein-coding sequences are under selective pressure, the ratios of synonymous substitution rate (ks) and non-synonymous substitution rate (ka) of homologous gene pairs were calculated using TBtools software [54]. The approximate date of duplication events was inferred by substituting Ks values into the formula million years ago (MYA) [55].
Predicted subcellular localization of BnCCD proteins and promoter analysis of BnCCD genes
The subcellular localization of BnCCD proteins were predicted using WoLF PSORT (http://www.genscript.com/tools/wolf-psort) [56]. The SOPMA secondary structure prediction method (https://npsa-prabi.ibcp.fr/cgi-bin/npsa_automat.pl?page=/NPSA/npsa_sopma.html) was used to predict the secondary structure of BnCCD proteins and the presence of transmembrane helices within BnCCD proteins were predicted using TMHMM Server v. 2.0 (http://www.cbs.dtu.dk/services/TMHMM/). The promoter sequences (2.0-kb region immediately upstream of the translation start sites) of the BnCCD genes were obtained from the B. napus genome database (http://www.genoscope.cns.fr/brassicanapus/) [57]. PlantCARE (Error! Hyperlink reference not valid.webtools/plantcare/html/) [58] was used to predict the presence of cis-acting sequences within each promoter.
RNA-seq analysis
To analyze the tissue-specific expression of the BnCCD genes, publically available B.napus RNA-sequencing (RNA-seq) data PRJNA358784 (BioProject) were downloaded from the National Center for Biotechnology Information (NCBI, https://www.ncbi.nlm.nih.gov/) [59]. All BnCCD genes expression levels were quantified in terms of FPKM (fragments per kilobase of exon per million mapped fragments) using Cufflinks with default parameters [60], and then extracted from RNA-seq data according to their B. napus code, including expression data from thirteen different organs (roots, stems, leaves, buds, anthocauli, calyxes, petals, pistils, stamens, anthers, capillaments, seeds and silique pericarps) at different developmental stages in B.napus cultivar ZS11. The heatmap for BnCCD genes was constructed using TBtools software.
RNA extraction and quantitative real-time PCR
Total RNA was extracted using the RNeasy Extraction Kit (Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s instructions. Contaminating genomic DNA was removed with DNase I. Total RNA (1 μg) was used to synthesize cDNA by reverse transcriptase (TaKaRa). The gene-specific primer pairs used to analyze BnCCD genes expression by qRT-PCR were designed using Primer Premier 5 [61] and the ACTIN 7 gene was used as an endogenous reference gene. All the primers were listed in S1 Table.
Quantitative RT-PCR was carried out using TB Green Premix Ex Taq on a Real-Time PCR Detection System (Bio-Rad, Hercules, CA, USA). The reaction mixture included 10 μL TB Green Premix Ex Taq, 0.5 μL forward primer, 0.5 μL reverse primer, 2 μL cDNA template, and 7 μL nuclease-free H2O in a total volume of 20 μL. The PCR cycling conditions were as follows: 95°C for 30s, followed by 40 cycles of 95°C for 5 s and 60°C for 30 s. Following PCR amplification, the dissolution curve was analyzed to ensure the specificity of the amplified products. Three biological replicates and three technical replicates were performed for each reaction. The relative gene expression levels were calculated using the 2−ΔΔCt method [61]. All the results were plotted as the mean ± standard error of mean (SEM) from three independent biological replicates using Graph Pad Prism 5.0 software (GraphPad Software Inc., San Diego, CA, USA).
Results
Identification and characterization of BnCCD genes
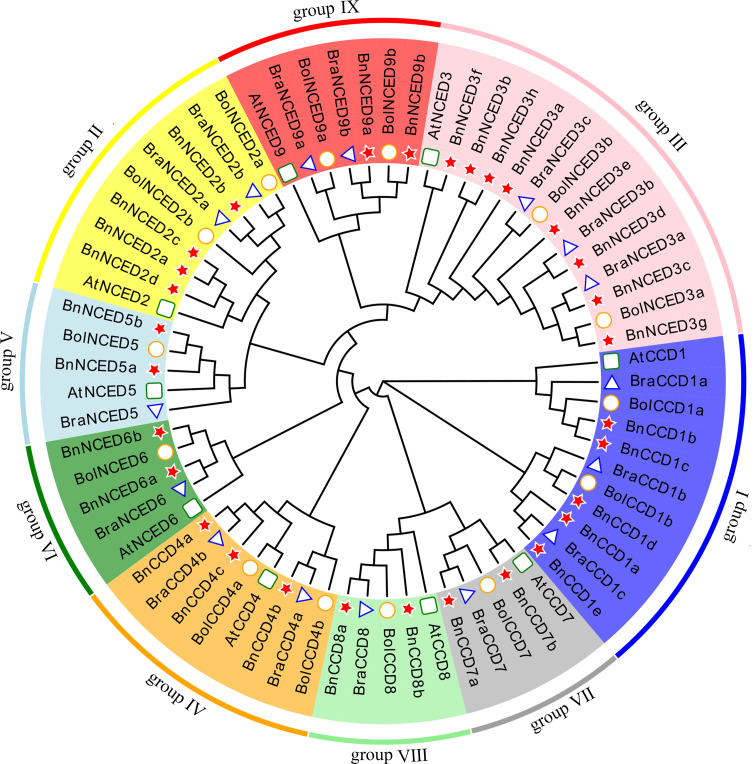
Using the nine AtCCD coding sequences as a BLASTN query, we identified 30 B. napus CCD genes (BnCCDs), 16 originating from B. rapa (BrCCDs) and 14 from B. oleracea (BoCCDs)(S2 Table). All the genes contained sequences encoding the RPE65 domain. RPE65 belongs to a family of carotenoid oxygenases in plant, bacterial, and animal systems that typically oxidatively cleave conjugated double bonds in the polyene backbone of carotenoids [16, 17]. In addition to light related functions, the function and mechanism of RPE65 in plants have not been elucidated. To determine the evolutionary relationships among 60 CCD proteins, we constructed a phylogenetic tree by the neighbor-joining method. Based on the topology of the phylogenetic tree, CCD proteins could be grouped into nine distinct subgroups (I, II, III, IV, V, VI, VII, VIII and IX), each of which contained one of the AtCCD proteins and the corresponding subgroup members of B. rapa, B. oleracea, and B. napus (Fig 1).
Fig 1. Phylogenetic analysis of 60 CCD protein sequences from A. thaliana, B. rapa, B. oleracea, and B. napus.
Sixty CCD protein sequences were used to construct the neighbor-joining tree using Mega 7 with 1,000 bootstrap replicates. The CCD proteins were devided into nine phylogenetic subgroups (I, II, III, IV, V, VI, VII, VIII and IX) and are marked with different background colors.
The physical and chemical properties of the BnCCD proteins are summarized in Table 1. The length of BnCCD proteins ranged from 255 amino acids (aa) (BnNCED3f) to 668 aa (BnCCD7a), with a mean length of 551 aa. The predicted molecular weights varied from 28.65 kDa (BnNCED3f) to 74.79 kDa (BnCCD7a) and the theoretical isoelectric point (PI) ranged from 4.75 (BnNCED3f) to 8.83 (BnNCED3e). Out of the 30 BnCCD proteins, 27 had PI values of less than 7, and the values of the remaining three BnCCD proteins were greater than 7. Based on the hydrophilicity index of amphoteric proteins between –0.5 ~ +0.5 (a negative GRAVY value indicates hydrophilicity and a positive value indicates hydrophobicity), all BnCCD proteins are amphiphilic proteins.
Table 1. The genes and encoded protein features of the 30 BnCCDs identified in this study.
| Gene name | Gene ID | Subgroup | Chromosome | Gene length (bp) | Gene position | Number of exons | Protein length (aa) | Molecular wt. (kDa) | pI | GRAVY | Subcellular location | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Start | End | |||||||||||
| BnCCD1a | BnaA09g41150D | I | A09 | 4466 | 28783045 | 28787510 | 13 | 483 | 55.24182 | 6.36 | −0.175 | chlo: 5, cyto: 2, vacu: 2, E.R.: 2, nucl: 1, pero: 1 |
| BnCCD1b | BnaAnng12520D | I | Ann_random | 3193 | 13541287 | 13544479 | 13 | 525 | 59.31523 | 5.70 | −0.256 | cyto: 7, chlo: 3, nucl: 1, mito: 1, plas: 1 |
| BnCCD1c | BnaC04g20610D | I | C04 | 3267 | 21697508 | 21700774 | 14 | 525 | 59.35633 | 5.94 | −0.257 | cyto: 10, chlo: 2, plas: 1 |
| BnCCD1d | BnaC08g33680D | I | C08 | 8770 | 32058916 | 32067685 | 14 | 612 | 69.62388 | 6.95 | −0.367 | chlo: 9, mito: 3, nucl: 1 |
| BnCCD1e | BnaC08g33690D | I | C08 | 7652 | 32077226 | 32084877 | 15 | 611 | 69.52093 | 6.41 | −0.289 | chlo: 14 |
| BnNCED2a | BnaA01g09090D | II | A01 | 1746 | 4447853 | 4449598 | 1 | 581 | 64.52248 | 5.33 | −0.224 | chlo: 9.5, chlo_mito: 5.5, cyto: 1, plas: 1, vacu: 1 |
| BnNCED2b | BnaA03g58230D | II | A03_random | 1752 | 1600131 | 1601882 | 1 | 583 | 64.95409 | 5.49 | −0.237 | chlo: 6, cyto: 5, plas: 1.5, cysk_plas: 1.5 |
| BnNCED2c | BnaC01g10770D | II | C01 | 1746 | 6588592 | 6590337 | 1 | 581 | 64.51950 | 5.31 | −0.214 | chlo: 9, cyto: 4 |
| BnNCED2d | BnaC07g35240D | II | C07 | 1752 | 37711814 | 37713565 | 1 | 583 | 64.82201 | 5.55 | −0.243 | chlo: 7, cyto: 4, plas: 1.5, cysk_plas: 1.5 |
| BnNCED3a | BnaA01g29390D | III | A01 | 1662 | 20329204 | 20331002 | 2 | 553 | 60.99077 | 5.28 | −0.287 | mito: 10, chlo: 4 |
| BnNCED3b | BnaA03g33390D | III | A03 | 1191 | 16162114 | 16163388 | 2 | 396 | 44.24510 | 4.99 | −0.242 | cyto: 6, cysk: 6, mito: 1 |
| BnNCED3c | BnaA05g25030D | III | A05 | 1794 | 18611263 | 18613467 | 1 | 597 | 65.80847 | 5.94 | −0.297 | mito: 8, chlo: 6 |
| BnNCED3d | BnaC01g36910D | III | C01 | 1797 | 36059468 | 36061576 | 1 | 598 | 65.72044 | 5.89 | −0.276 | mito: 8, chlo: 6 |
| BnNCED3e | BnaC05g39180D | III | C05 | 948 | 37757359 | 37758306 | 1 | 315 | 34.06851 | 8.83 | −0.291 | mito: 11, chlo: 3 |
| BnNCED3f | BnaC05g39190D | III | C05 | 768 | 37758388 | 37759155 | 1 | 255 | 28.64751 | 4.75 | −0.217 | cyto: 8, cysk: 4, plas: 1 |
| BnNCED3g | BnaC05g39200D | III | C05 | 1794 | 37769098 | 37770891 | 1 | 597 | 65.84553 | 6.01 | −0.318 | mito: 9, chlo: 5 |
| BnNCED3h | BnaCnng32080D | III | Cnn_random | 1194 | 30423020 | 30424656 | 2 | 396 | 44.29825 | 5.09 | −0.219 | cysk: 8, cyto: 4, mito: 1 |
| BnCCD4a | BnaA01g09900D | IV | A01 | 2020 | 4881104 | 4883125 | 1 | 596 | 65.70485 | 6.33 | −0.236 | chlo: 12, mito: 1.5, cyto_mito: 1.5 |
| BnCCD4b | BnaA08g09110D | IV | A08 | 1782 | 8767981 | 8769762 | 3 | 532 | 58.57082 | 6.16 | −0.129 | chlo: 12, cyto: 1 |
| BnCCD4c | BnaC01g11550D | IV | C01 | 2067 | 7220281 | 7222347 | 1 | 593 | 65.43358 | 6.45 | −0.234 | chlo: 12, mito: 1.5, cyto_mito: 1.5 |
| BnNCED5a | BnaA09g26450D | V | A09 | 1170 | 19605914 | 19607683 | 1 | 589 | 65.33387 | 5.37 | −0.324 | chlo: 11, nucl: 2 |
| BnNCED5b | BnaCnng38510D | V | Cnn_random | 1170 | 37143600 | 37145369 | 1 | 589 | 65.45993 | 5.42 | −0.320 | chlo: 11, nucl: 1, mito: 1 |
| BnNCED6a | BnaA07g06050D | VI | A07 | 1758 | 6379220 | 6380977 | 1 | 585 | 65.00222 | 6.07 | −0.321 | chlo: 7.5, chlo_mito: 6.5, mito: 4.5 |
| BnNCED6b | BnaC07g07580D | VI | C07 | 1758 | 11978406 | 11980163 | 1 | 585 | 65.06919 | 6.00 | −0.329 | pero: 8, chlo: 3, cyto: 2 |
| BnCCD7a | BnaA04g26000D | VII | A04 | 5803 | 18550548 | 18556350 | 10 | 668 | 74.78965 | 5.77 | −0.381 | chlo: 9, plas: 3, mito: 1 |
| BnCCD7b | BnaC04g50070D | VII | C04 | 3912 | 47874505 | 47878416 | 9 | 648 | 72.69485 | 6.19 | −0.303 | chlo: 8, plas: 3, mito: 1, vacu: 1 |
| BnCCD8a | BnaA01g04140D | VIII | A01 | 3287 | 1908477 | 1911763 | 6 | 569 | 63.98902 | 7.13 | −0.341 | chlo: 10, mito: 3 |
| BnCCD8b | BnaC01g05600D | VIII | C01 | 12036 | 2963042 | 2975077 | 7 | 574 | 64.98737 | 8.50 | −0.452 | chlo: 7, cyto: 4, nucl: 2 |
| BnNCED9a | BnaA07g39250D | IX | A07_random | 1809 | 2059180 | 2060988 | 1 | 602 | 66.75458 | 5.90 | −0.299 | chlo: 14 |
| BnNCED9b | BnaC06g38870D | IX | C06 | 1809 | 36264799 | 36266607 | 1 | 602 | 66.79169 | 6.11 | −0.281 | chlo: 14 |
chlo: chloroplast; cyto: cytoplasmic; vacu: vacuolar; E.R: Endoplasmic Reticulum; pero: peroxisomal; mito: mitochondria; nucl: nucleus; plas: plasma membrane.
Chromosomal localization and gene structure
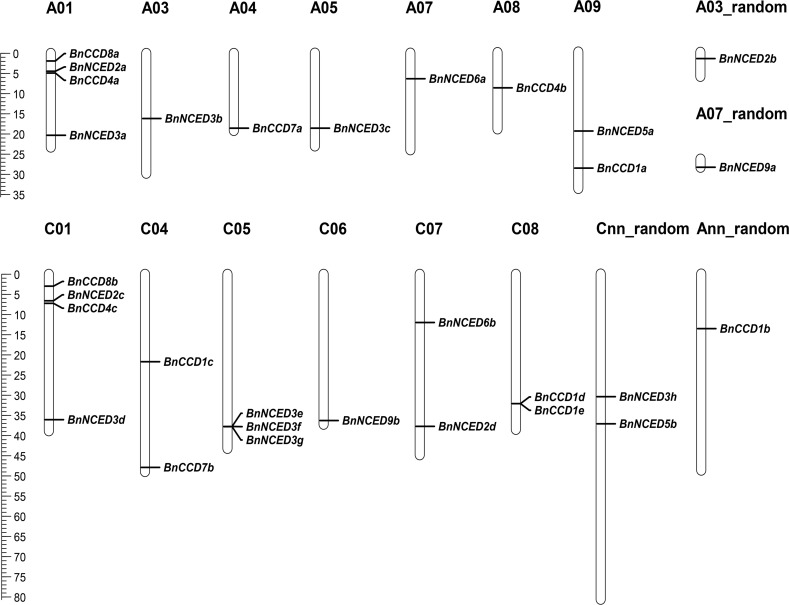
By Mapchart, an analysis of the chromosomal distribution of the BnCCD loci showed that these genes were not evenly distributed across the chromosomes (Fig 2). The 30 BnCCD loci are distributed on 13 chromosomes in B. napus; the 14 BnCCDs are located on the A subgenome and the 16 BnCCDs are located on the C subgenome. Chromosomes A01 and C01 possess the most BnCCD loci, each containing four BnCCDs. Chromosomes A03, A04, A05, A07, A08, C06, A03-random, A07-random, and Ann-random each contain only one BnCCD locus, whereas chromosomes A09, C04, C07, C08, and Cnn-random each contain two BnCCD loci. BnNCED3e, BnNCED3f, and BnNCED3g (subgroup III) are located on chromosome C05.
Fig 2. Genomic distribution of BnCCD genes.
The chromosomal location of each BnCCD locus was mapped to the B. napus genome: the chromosome number is indicated above each chromosome. The scale is in megabases (Mb). Ann and Cnn are pseudo-molecule chromosomes. Random means that the specific location of the gene is unknown.
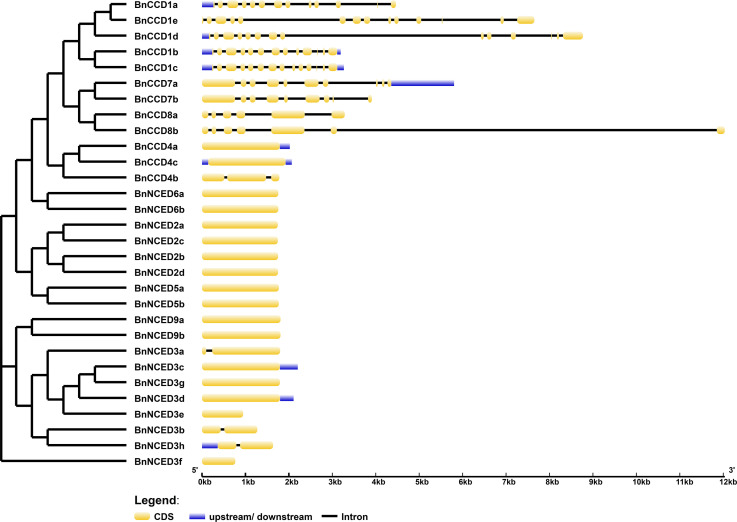
The positions of exons and introns in members of a gene family might have played crucial roles during evolution [62]. Therefore, we next analyzed the gene structure (exon–intron organization) of the 30 BnCCDs (Fig 3). Genes with similar structures were closely related. BnNCED2, BnNCED5, BnNCED6, and BnNCED9 subgroups were intron-less. The BnCCD1 subgroup contained 13–15 exons; two genes contained 13 exons, two genes contained 14 exons, and only BnCCD1e possessed 15 exons. All BnCCD4 subgroup members contained one exon, except for BnCCD4b, which contained three. BnCCD8b contained 6 introns, one more than BnCCD8a.
Fig 3. Exon–intron structures of the BnCCD genes, arranged based on their phylogenetic relationships.
An unrooted phylogenetic tree was constructed based on the full-length sequences of BnCCD proteins using the N-J method in MEGA7. Bootstrap values based on 1,000 replications were calculated. The exon–intron structure of the BnCCD genes was analyzed using the GSDS online tool. The lengths of the exons and introns for each BnCCD gene are drawn to scale.
Conserved motifs and protein profiles of BnCCD proteins
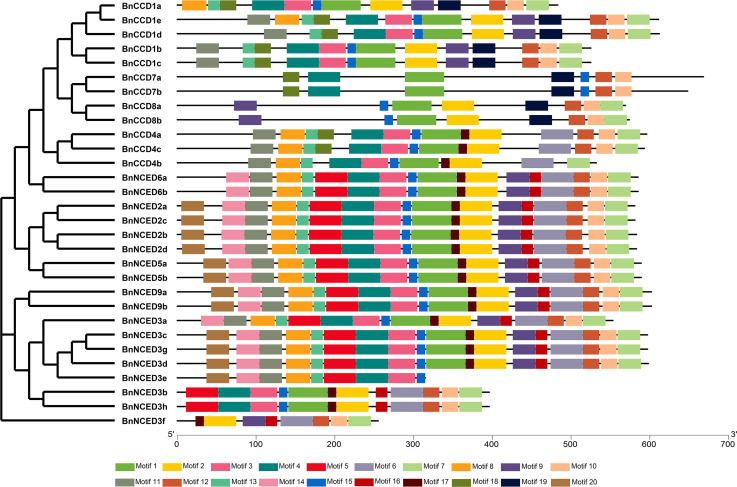
Twenty putative BnCCD protein motifs were predicted by the MEME program (Fig 4). The BnNCED2, NCED5, NCED6, and NCED9 proteins all contain motifs 1–17. Except for BnNCED3e, all other BnNCED3 proteins contain motifs 2, 6, 7, 10, 12, 16, and 17. Motifs 1, 2, 7, and 15 are present in all BnCCD1, BnCCD4, and BnCCD8 proteins and all BnCCD7 members contain motifs 1, 4, 10, 12, 15, 18, and 19. Using the InterProScan program, we searched for annotations for the conserved motifs. Motifs 1–9 and 19 were associated with carotenoid oxygenase (IPR004294) and the remaining motifs had not been annotated.
Fig 4. The conserved motifs of the BnCCD proteins, arranged based on their phylogenetic relationships.
The conserved motifs of the BnCCD proteins were identified by MEME. Gray lines represent non-conserved sequences and each motif is indicated by a colored box (numbered at the bottom). The lengths of the motifs in each protein are drawn to scale.
Prediction of secondary structure using SOPMA showed that the main structure of the BnCCD proteins was a random coil. In addition to BnNCED3e, the alpha helix was more than the extension chain. The alpha helix was equal to the extension chain in BnCCD7a. The secondary structure characteristics of other BnCCD proteins ranged from more to less: random curl, extended strand, alpha helix, beta turn. Analysis using the TMHMM Server v. 2.0 showed that no BnCCD proteins possessed a transmembrane domain.
Nineteen of the 30 BnCCD proteins were predicted to be localized to the chloroplast, five to the cytoplasm, and five to the mitochondrion. BnNCED6b was predicted to be localized to the peroxisome.
Ka and Ks calculation for orthologous CCD genes between Brassica napus and Arabidopsis thaliana
To determine whether the CCD protein-coding genes in B. napus and A. thaliana are under selective pressure, the Ka/Ks ratios for 30 pairs of orthologous genes were calculated using TBtools software (Table 2). The Ka/Ks ratios of all the gene pairs were considerably lower than 1, indicating that the CCD gene family has undergone purifying selection. The Ks values for B. napus relative to A. thaliana ranged from 0.28852 to 0.65433, suggesting that gene duplications occurred approximately 9.62–21.81 million years ago (MYA).
Table 2. The non-synonymous (Ka) and synonymous substitution rate (Ks) for orthologous CCD gene pairs between B. napus and A. thaliana.
| Orthologous gene pairs | Ka | Ks | Ka/Ks | Duplication date (MYA) | |
|---|---|---|---|---|---|
| AtCCD1 | BnCCD1a | 0.08151 | 0.36209 | 0.22510 | 12.07 |
| AtCCD1 | BnCCD1b | 0.04122 | 0.32012 | 0.12875 | 10.67 |
| AtCCD1 | BnCCD1c | 0.03384 | 0.28852 | 0.11730 | 9.62 |
| AtCCD1 | BnCCD1d | 0.05913 | 0.38774 | 0.15250 | 12.92 |
| AtCCD1 | BnCCD1e | 0.07611 | 0.35530 | 0.21422 | 11.84 |
| AtNCED2 | BnNCED2a | 0.06843 | 0.46502 | 0.14716 | 15.50 |
| AtNCED2 | BnNCED2b | 0.04285 | 0.41304 | 0.10374 | 13.77 |
| AtNCED2 | BnNCED2c | 0.06108 | 0.47780 | 0.12784 | 15.93 |
| AtNCED2 | BnNCED2d | 0.04606 | 0.43371 | 0.10620 | 14.46 |
| AtNCED3 | BnNCED3a | 0.04945 | 0.56001 | 0.08829 | 18.67 |
| AtNCED3 | BnNCED3b | 0.05042 | 0.55742 | 0.09045 | 18.58 |
| AtNCED3 | BnNCED3c | 0.05052 | 0.51718 | 0.09768 | 17.24 |
| AtNCED3 | BnNCED3d | 0.04962 | 0.52187 | 0.09509 | 17.40 |
| AtNCED3 | BnNCED3e | 0.05472 | 0.45159 | 0.12116 | 15.05 |
| AtNCED3 | BnNCED3f | 0.05271 | 0.58288 | 0.09043 | 19.43 |
| AtNCED3 | BnNCED3g | 0.04581 | 0.51353 | 0.08921 | 17.12 |
| AtNCED3 | BnNCED3h | 0.05693 | 0.55439 | 0.10268 | 18.48 |
| AtCCD4 | BnCCD4a | 0.07819 | 0.59005 | 0.13252 | 19.67 |
| AtCCD4 | BnCCD4b | 0.06912 | 0.50381 | 0.13720 | 16.79 |
| AtCCD4 | BnCCD4c | 0.07461 | 0.60924 | 0.12246 | 20.31 |
| AtNCED5 | BnNCED5a | 0.04001 | 0.38163 | 0.10484 | 12.72 |
| AtNCED5 | BnNCED5b | 0.03847 | 0.40056 | 0.09605 | 13.35 |
| AtNCED6 | BnNCED6a | 0.08570 | 0.57248 | 0.14970 | 19.08 |
| AtNCED6 | BnNCED6b | 0.09078 | 0.57371 | 0.15824 | 19.12 |
| AtCCD7 | BnCCD7a | 0.07106 | 0.45309 | 0.15683 | 15.10 |
| AtCCD7 | BnCCD7b | 0.07166 | 0.42570 | 0.16834 | 14.19 |
| AtCCD8 | BnCCD8a | 0.05711 | 0.42701 | 0.13374 | 14.23 |
| AtCCD8 | BnCCD8b | 0.09405 | 0.49007 | 0.19191 | 16.34 |
| AtNCED9 | BnNCED9a | 0.07262 | 0.61567 | 0.11796 | 20.52 |
| AtNCED9 | BnNCED9b | 0.06799 | 0.65433 | 0.10391 | 21.81 |
The cis-acting elements predicted to be present in BnCCD promoters
The cis-acting elements present in the promoters are essential for transcriptional gene regulation. In total, 87 types of cis-acting elements were predicted to be present in 30 BnCCD promoters (S3 Table), using the online software PlantCARE (http://bioinformatics.psb.ugent.be/webtools/plantcare/html/). In addition to CAAT and TATA boxes, each promoter contained more than 10 cis-acting elements. Among these, 26 different elements are associated with responses to light, with 206 occurrences across all promoters. The remaining elements are associated with hormonal and stress responses and tissue-specific expression. Hormone-responsive cis-elements account for a large proportion of the total and include ABRE (abscisic acid response element), AuxRR-core and TGA-elements (auxin response), ERE (ethylene-response element), TCA-element (salicylic acid response), P-box and GARE-motif (gibberellin-response), and the CGTCA and TGACG motifs (MeJA-response). Notably, all BnCCDs contain ABRE, CGTCA, and TGACG motifs, suggesting that this gene family functions in hormone response pathways. Additional cis-elements related to stress response were identified; for example, LTR (low-temperature responsiveness) is present in all BnCCD promoters except for BnNCED3f and BnCCD8a. Additional elements include the drought-inducible MBS element, TC-rich repeats, a cis-acting element involved in defense and stress responsiveness, and the WUN-motif, a wound-responsive element.
Some cis-acting elements in BnCCD promoters might determine the tissue-specific expression pattern, such as the CAT-box, which is associated with meristematic expression; GCN4_motif, involved in endosperm expression; HD-Zip 1, involved in differentiation of the palisade mesophyll cells; and RY-element, which confers seed-specific expression.
RNA-seq analysis
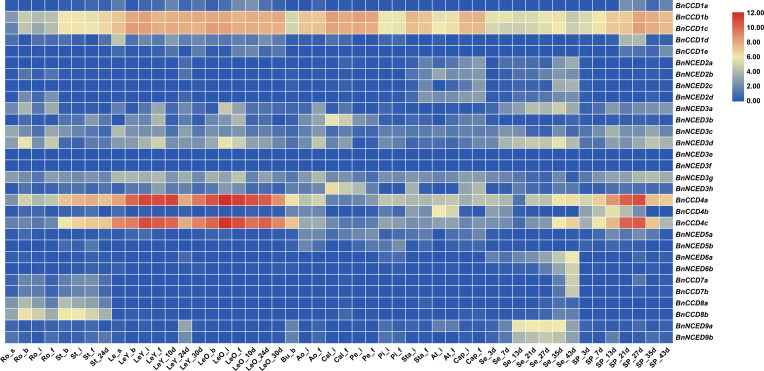
We analyzed the expression of 30 BnCCDs at different developmental stages and in various tissues from the B. napus cultivar ZS11 and constructed an expression heat map (Fig 5). Whereas no BnNCED3f and BnNCED3g transcripts were detected in any tissue, the remaining 28 BnCCDs showed tissue- and development-specific expression levels. BnCCD1b and BnCCD1c were highly expressed in leaves, stems, buds, flowers, seeds, and silique pericarp, but expression was relatively low in roots. BnNCED9a and BnNCED9b had similar expression patterns and were highly expressed in seeds. Genes belonging to the same subgroup had a wide range of expression patterns: BnCCD4a and BnCCD4c were highly expressed in leaves, stems, and pericarps; however, BnCCD4b, which belongs to the BnCCD4 subgroup, had a low expression level in leaves, stems, and pericarps but was highly expressed in anthers, suggesting that it might function in stamen growth and development.
Fig 5. Heatmap of the expression patterns of selected BnCCD genes in different tissues and at different developmental stages.
The expression data were obtained from publically available RNA-sequening (RNA-seq) data. Ro: Roots; St: Stems; Le: Leaves; LeY: Young leaves; LeO: Old leaves; Bu: Buds; Ao: Anthocauli; Cal: Calyxes; Pe: Petals; Pi: Pistils; Sta: Stamens; At: Anthers; Cap: Capillaments; Se: Seeds; Sp: Silique pericarps; _s: At seedling stage; _b: In the bud stage; _i: At the initial flowering stage; _f: At the flourishing flowering stage; _3d: 3 days after flowering; _7,10,13,21,24,27,30,35,43d: 7,10,13, 21,24,27,30,35,43days after flowering; Ro_s: Roots at seedling stage.
Expression profiling of BnCCDs in different organs
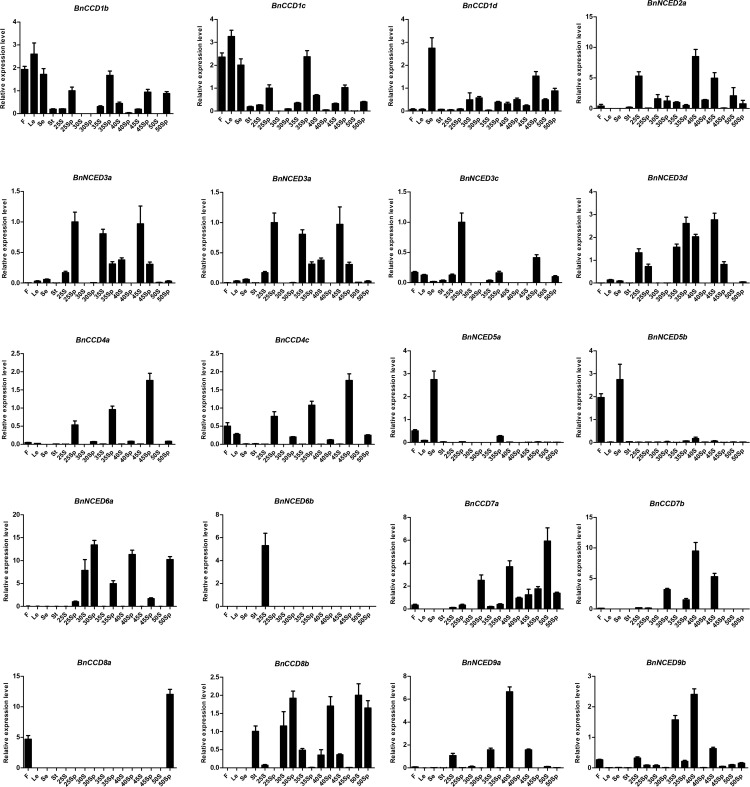
To gain insight into the biological functions of BnCCDs, we analyzed their expression patterns in flowers, leaves, sepals, stems, seeds, and silique pericarps, and for seeds and silique pericarps also at different developmental stages (25, 30, 35, 40, 45, and 50 days after flowering) by qRT-PCR. Several BnCCDs, including BnCCD1b, BnCCD1c, BnCCD1d, BnNCED2a, BnNCED2c, BnNCED3a, BnNCED3c, BnNCED3d, BnCCD4a, BnCCD4c, BnNCED5a, BnNCED5b, BnNCED6a, BnNCED6b, BnCCD7a, BnCCD7b, BnCCD8a, BnCCD8b, BnNCED9a, and BnNCED9b, had diverse expression patterns in different tissues (Fig 6). Except for BnCCD8b, the expression level of CCD genes was low in the stem. BnNCED5b and BnCCD8a transcripts were abundant in flowers, BnNCED5a and BnNCED5b transcripts were abundant in sepals, and BnNCED9a and BnNCED9b transcripts were abundant in seeds. However, the expression patterns of some genes within the same family differed: the transcript levels of BnCCD1b and BnCCD1c in leaves were higher than those in other tissues, whereas BnCCD1d was most highly expressed in sepals. In addition, BnNCED2a and BnNCED2c transcript levels were higher in seeds at 40–50 days after flowering (40S, 45S, and 50S) than at 25–35 days after flowering (25S, 30S and 35S). BnCCD4a and BnCCD4c transcript levels were abundant in silique pericarps collected at the 25 days after flowering (25Sp), 35Sp, and 45Sp stages, but were present at low levels at the 30Sp, 40Sp, 45Sp and 50Sp stages.
Fig 6. The expression patterns of selected BnCCD genes in different tissues and at different developmental stages.
The expression patterns of 20 BnCCD genes in B.napus cultivar Zhong Shuang 11 (ZS11) were analyzed by qRT-PCR. F: flowers; Le: leaves; Se: sepals; St: stems; 25S, 30S, 35S, 40S, 45S, 50S: seeds collected at 25, 30, 35, 40, 45and 50 days after flowering, respectively. 25Sp, 30Sp, 35Sp, 40Sp, 45Sp, 50Sp: silique pericarps collected at 25, 30, 35, 40, 45 and 50 days after flowering, respectively. Values represented the mean ± standard error of mean (SEM) of three biological replicates with three technical replicates at each developmental stage. Error bars indicated the SEM among three experiments.
Discussion
Arabidopsis contains nine CCD genes. Brassicaceae, including Arabidopsis, underwent an ancient α, β and γ whole-genome duplication polyploidization events, and also experienced an additional recent genome-wide tripling (WGT) event [63]. Because it is an allotetraploid formed by interspecific hybridization between B. rapa and B. oleracea, B. napus would be expected to contain six copies of each Arabidopsis thaliana gene (i.e., 54) [64]. Because of genome shrinkage and gene loss, the number of BnCCDs identified in this study was a little lower (i.e., 30), with only 2–6 copies of each Arabidopsis gene present. The B. rapa and B. oleracea genomes contain 16 BrCCD and 14 BoCCD genes, respectively, and the number of BnCCDs is the sum of these. However, only two members of the BnNCED9 subgroup and three of the BnCCD4 subgroup were identified in this study, whereas two members were found in the corresponding subgroups of both B. rapa and B. oleracea, indicating that gene loss might have occurred among subgroups BnCCD4 and BnNCED9.
The molecular characteristics of all BnCCD protein members were found to differ, but proteins in the same subgroup had similar molecular weights and isoelectric points. The position of exon–intron boundaries reflects the evolution of these genes [62]. Subcellular localization predictions suggested that BnCCD proteins are localized to the chloroplast, mitochondrion, cytoplasm, and peroxisome. BnCCD1b, BnCCD1c, BnNCED3b, BnNCED3f, and BnNCED3h were predicted to be cytoplasmic, suggesting that BnCCD1 and BnNCED3 might interact in the cytoplasm. It also showed that these genes may not participate in chlorophyll photosynthesis, which was consistent with the research by Zhang et al. [28]. BnNCED2 subgroup is located in the chloroplast. Studies have shown that histidine residues in NCED amino acids can bind Fe 2+ to make NCED proteins function [65], and there is a chloroplast transit peptide structure in them [66]. Wang et al. [67] found that NCED2 protein in Camellia sinensis is localized in chroloplast and has N-terminal chloroplast targeting signal peptide sequences, which further proves that CsNCED2 has the biological activity of cleaving epoxy carotenoids to generate ABA precursors in plastids. Besides, in both sugarcane and rice, CCD8 are proteins located in chloroplast [25, 68]. Through online analysis we found that BnCCD8 is also localized in the chloroplast.
Seven cis-acting elements associated with various stress responses were predicted among the promoters of the 30 BnCCDs; LTR, MBS, TC-rich repeats, and nine cis-acting elements were associated with responses to abscisic acid, MeJA, salicylic acid, and auxin. CCD1, 4, and 8 and NCED2 and 9 are transcriptionally upregulated following treatment of B. rapa with the phytohormones ABA and SL [16]. Furthermore, Nicotiana tabacum plants that heterologously expressed NCED1 of Stylosanthes guianensis had increased tolerance to light, oxidative, drought, salt, and cold stress, indicating that CCD might regulate plant tolerance against these abiotic stresses [69]. The G-Box, GT1, and ACE motifs, which are transcriptionally responsive to light, are present in the BnCCD gene promoters. In addition, BnCCD promoters contain circadian response elements, RY-elements, and MSA-like and MBSI elements, indicating that most CCD genes function in plant growth and development and stress responses.
Based on RNA-seq data (S4 Table), we constructed an expression heatmap, which demonstrated that the 30 BnCCDs were differentially expressed in different B. napus tissues. Members of the same gene subgroup occasionally exhibited different expression patterns, such as BnCCD1 and BnCCD4, potentially reflecting new functionalization among the gene family during evolution. Genes of the CCD1 and CCD4 subgroups function in the formation of a variety of apo-carotenoids, which confer unique colors, tastes, and aromas [70–72]. A CCD1 loss-of-function mutant showed a decreased level of β-ionone in tomato fruit (Solanum lycopersicum) [31] and petunia flowers (Petunia hybrida) [73]. Carotenoid homeostasis is regulated by CCD4 in different tissues, such as Arabidopsis seeds [74] and potato tubers [23]. BnCCD4b was mainly expressed in stamens, suggesting that it might function in stamen growth and development. Species such as Solanumly copersicum, Prunus persica, and Crocus, were also reported to preferentially express CCD4 in floral organs, indicating that the evolution of CCD4 genes might have been adaptive and have enhanced specific physiological traits unique to flowering plants [21, 75, 76]. CCD4 with normal function or lack of function will change the color of fruit and flower organs. Previous studies have suggested CCD4 gene can fade the yellow petals of Rhododendron japonicum and Eustoma grandiflorum [77, 78], because its ability of cleaving carotenoids. Inactivation of CCD4 will change the color of B.napus and Chrysanthemum morifolium petals from white to yellow [79, 80]. Controlling the expression of CCD gene family provides a way to change the color of plant petals.
Conclusion
In conclusion, we performed a comprehensive study of CCD gene family in B.napus. We identified 30 putative BnCCDs that were classed into nine subgroups (I-IX) on the basis of their phylogenetic relationships. The length of the BnCCD proteins ranged from 255 to 668 aa, and the Ks values for B. napus relative to A. thaliana ranged from 0.28852 to 0.65433. In addition, RNA-seq data and qRT-PCR analysis revealed that BnCCDs were differentially expressed in different tissues and organs, and had tissue/organ specificity and expression preference, suggesting that BnCCDs had clear function differentiation. Our results will help lay the foundation for the functional characterization of the CCD gene family and better understand the structural and functional relationships among these family members.
Supporting information
(XLSX)
(XLSX)
(XLSX)
(XLSX)
Data Availability
All relevant data are within the manuscript and its Supporting Information files.
Funding Statement
JNL was funded to undertake research by Ministry of Science and Technology of the People´s Republic of China (http://www.most.gov.cn/) (The National Key Research and Development Plan, 2018YFD0100504-05, 2016YFD0101305) (The National Natural Science Foundation of China, 31830067, U1302266). JNL was funded to undertake research by Ministry of Education of the People's Republic of China (http://www.moe.gov.cn/) (The 111 Project, B12006). XTZ was funded to do research by Ministry of Education of the People's Republic of China (National Undergraduate Training Program for Innovation and Entrepreneurship, 201810635080). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Johnson JD. Do carotenoids serve as transmembrane radical channels? Free Radical Biology & Medicine. 2009;47(3):321–3. [DOI] [PubMed] [Google Scholar]
- 2.Walter MH, Strack D. Carotenoids and their cleavage products: biosynthesis and functions. Natural product reports. 2011;28(4):663–92. 10.1039/c0np00036a [DOI] [PubMed] [Google Scholar]
- 3.Xuewu S, Kiser PD, Johannes Von L, Krzysztof P. Structural basis of carotenoid cleavage: From bacteria to mammals. Archives of Biochemistry & Biophysics. 2013;539(2):203–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Paiva SA, Russell RM. Beta-carotene and other carotenoids as antioxidants. Journal of the American College of Nutrition. 1999;18(5):426–33. 10.1080/07315724.1999.10718880 [DOI] [PubMed] [Google Scholar]
- 5.Britton G. Structure and properties of carotenoids in relation to function. The FASEB Journal. 1995;9(15):1551–8. [PubMed] [Google Scholar]
- 6.Navot G, Gil R, Zehava K, Dani Z, Joseph H. A chromoplast-specific carotenoid biosynthesis pathway is revealed by cloning of the tomato white-flower locus. Plant Cell. 2006;18(8):1947–60. 10.1105/tpc.105.039966 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lewinsohn E, Sitrit Y, Bar E, Azulay Y, Ibdah M, Meir A, et al. Not just colors—carotenoid degradation as a link between pigmentation and aroma in tomato and watermelon fruit. Trends in Food Science & Technology. 2005;16(9):0–415. [Google Scholar]
- 8.Suzuki S, Nishihara M, Nakatsuka T, Misawa N, Ogiwara I, Yamamura S. Flower color alteration in Lotus japonicus by modification of the carotenoid biosynthetic pathway. Plant Cell Reports. 2007;26(7):951–9. 10.1007/s00299-006-0302-7 [DOI] [PubMed] [Google Scholar]
- 9.Xie XN, Yoneyama K, Yoneyama K. The strigolactone story. Annual Review of Phytopathology. 2010;48(1):93–117. [DOI] [PubMed] [Google Scholar]
- 10.Norman JMV, Sieburth LE. Dissecting the biosynthetic pathway for the bypass1 root-derived signal. Plant Journal. 2010;49(4):619–28. [DOI] [PubMed] [Google Scholar]
- 11.Ruiz-Sola MÁ, Manuel R-C. Carotenoid Biosynthesis in Arabidopsis: A Colorful Pathway. The Arabidopsis Book: BIOONE; 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ohmiya A. Carotenoid cleavage dioxygenases and their apocarotenoid products in plants. Plant Tissue Culture Letters. 2009;26(4):351–8. [Google Scholar]
- 13.Lange BM, Ghassemian M. Genome organization in Arabidopsis thaliana: a survey for genes involved in isoprenoid and chlorophyll metabolism. Plant Molecular Biology. 2003;51(6):925 10.1023/a:1023005504702 [DOI] [PubMed] [Google Scholar]
- 14.Schwartz SH, Tan BC, Gage DA, Zeevaart JA, McCarty DR. Specific oxidative cleavage of carotenoids by VP14 of maize. Science. 1997;276(5320):1872–4. 10.1126/science.276.5320.1872 [DOI] [PubMed] [Google Scholar]
- 15.Kiefer C, Hessel S, Lampert JM, Vogt K, Lederer MO, Breithaupt DE, et al. Identification and characterization of a mammalian enzyme catalyzing the asymmetric oxidative cleavage of provitamin A. Journal of Biological Chemistry. 2001;276(17):14110–6. 10.1074/jbc.M011510200 [DOI] [PubMed] [Google Scholar]
- 16.Kim Y, Hwang I, Jung HJ, Park JI, Kang JG, Nou IS. Genome-Wide Classification and Abiotic Stress-Responsive Expression Profiling of Carotenoid Oxygenase Genes in Brassica rapa and Brassica oleracea. Journal of Plant Growth Regulation. 2016;35(1):202–14. [Google Scholar]
- 17.Hamel CP, Tsilou E, Pfeffer BA, Hooks JJ, Detrick B, Redmond TM. Molecular cloning and expression of RPE65, a novel retinal pigment epithelium-specific microsomal protein that is post-transcriptionally regulated in vitro. Journal of Biological Chemistry. 1993;268(21):15751–7. [PubMed] [Google Scholar]
- 18.Vallabhaneni R, Bradbury LMT, Wurtzel ET. The carotenoid dioxygenase gene family in maize, sorghum, and rice. Archives of Biochemistry & Biophysics. 2010;504(1):104–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Tong SM, Xi HX, Ai KJ, Hou HS. Overexpression of wheat TaNCED gene in Arabidopsis enhances tolerance to drought stress and delays seed germination. Biologia Plantarum. 2016;61(1):1–9. [Google Scholar]
- 20.Niu Z, Liu G, Shi T, Zhang S, Jia H, Zhang H, et al. Cloning of NCED3 gene in Nicotiana tabacum and analysis of its drought stress-induced expression. Acta Tabacaria Sinica. 2015;21(3):100–6. [Google Scholar]
- 21.Wei Y, Wan H, Wu Z, Wang R, Ruan M, Ye Q, et al. A Comprehensive Analysis of Carotenoid Cleavage Dioxygenases Genes in Solanum Lycopersicum. Plant Molecular Biology Reporter. 2016;34(2):512–23. [Google Scholar]
- 22.Qiong-Qiong LI, Zhang J, Deng Y, Wei-Jun YU, Gao HT, Jin J, et al. Cloning and expression analysis of GmNCED1 from Glycine max. Chinese Journal of Oil Crop Sciences. 2014;36(4). [Google Scholar]
- 23.Campbell R, Ducreux LJM, Morris WL, Morris JA, Suttle JC, Ramsay G, et al. The metabolic and developmental roles of carotenoid cleavage dioxygenase4 from potato. Plant Physiology. 2010;154(2):656–64. 10.1104/pp.110.158733 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Gonzálezverdejo CI, Ángeles O, Román B, Gómez P. Expression Profile of Carotenoid Cleavage Dioxygenase Genes in Summer Squash (Cucurbita pepo L.). Plant Foods for Human Nutrition. 2015;70(2):200–6. 10.1007/s11130-015-0482-9 [DOI] [PubMed] [Google Scholar]
- 25.Zhuan-Di WU, Liu XL, Liu JY, Zan FG, Zhao PF, Lin XQ, et al. Cloning and Expression Analysis of Strigolactones BiosynthesisRelated Gene ScCCD8 in Sugarcane. Scientia Agricultura Sinica. 2016. [Google Scholar]
- 26.Huang FC, Molnclr P, Schwab W. Cloning and functional characterization of carotenoid cleavage dioxygenase 4 genes. Journal of Experimental Botany. 2009;60(11):3011–22. 10.1093/jxb/erp137 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lashbrooke JG, Young PR, Dockrall SJ, Vasanth K, Vivier MA. Functional characterisation of three members of the Vitis viniferaL. carotenoid cleavage dioxygenase gene family. Bmc Plant Biology. 2013;13(1):156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Zhang M, Gao A, Zhao Z, Zhou K, Huang J, Dang Z, et al. Cloning and Sequence Analysis of Carotenoid Cleavage of Dioxygenase 1 (CCD1) Gene from Mango(Mangifera indica). Molecular Plant Breeding. 2018. [Google Scholar]
- 29.Mei Z, Ping L, Guanglian Z, Xiangxin L. Cloning and functional analysis of 9-cis-epoxycarotenoid dioxygenase (NCED) genes encoding a key enzyme during abscisic acid biosynthesis from peach and grape fruits. Journal of Plant Physiology. 2009;166(12):1241–52. 10.1016/j.jplph.2009.01.013 [DOI] [PubMed] [Google Scholar]
- 30.Tan B-C, Joseph LM, Deng W-T, Liu L, Li Q-B, Cline K, et al. Molecular characterization of the Arabidopsis 9-cis epoxycarotenoid dioxygenase gene family. The Plant Journal. 2003;35(1%@ 0960–7412):44–56. 10.1046/j.1365-313x.2003.01786.x [DOI] [PubMed] [Google Scholar]
- 31.Simkin AJ, Schwartz SH, Michele A, Taylor MG, Klee HJ. The tomato carotenoid cleavage dioxygenase 1 genes contribute to the formation of the flavor volatiles beta-ionone, pseudoionone, and geranylacetone. Plant Journal. 2010;40(6):882–92. [DOI] [PubMed] [Google Scholar]
- 32.Ilg A, Yu Q, Schaub P, Beyer P, Al-Babili S. Overexpression of the rice carotenoid cleavage dioxygenase 1 gene in Golden Rice endosperm suggests apocarotenoids as substrates in planta. Planta. 2010;232(3):691–9. 10.1007/s00425-010-1205-y [DOI] [PubMed] [Google Scholar]
- 33.Auldridge M, Block A, Vogel J, Dabney-Smith C, Mila I, Bouzayen MLM, et al. Characterization of three members of the Arabidopsis carotenoid cleavage dioxygenase family demonstrates the divergent roles of this multifunctional enzyme family. Plant Journal. 2013;2(12):G8–G10. [DOI] [PubMed] [Google Scholar]
- 34.Akemi O. Carotenoid cleavage dioxygenase (CmCCD4a) contributes to white color formation in chrysanthemum petals. Plant physiology. 2006;3(142). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Yoshioka S, Aida R, Yamamizo C, Shibata M, Ohmiya A. The carotenoid cleavage dioxygenase 4 (CmCCD4a) gene family encodes a key regulator of petal color mutation in chrysanthemum. Euphytica. 2012;184(3):377–87. [Google Scholar]
- 36.Rameau C. Strigolactones, a novel class of plant hormone controlling shoot branching. Comptes rendus—Biologies. 2010;333(4):344–9. 10.1016/j.crvi.2010.01.012 [DOI] [PubMed] [Google Scholar]
- 37.Adrian A. The path from β-carotene to carlactone, a strigolactone-like plant hormone. Science (New York, NY). 2012;6074(335). [DOI] [PubMed] [Google Scholar]
- 38.Ledger SE, Janssen BJ, Sakuntala K, Tianchi W, Snowden KC. Modified CAROTENOID CLEAVAGE DIOXYGENASE8 expression correlates with altered branching in kiwifruit (Actinidia chinensis). New Phytologist. 2010;188(3):803–13. 10.1111/j.1469-8137.2010.03394.x [DOI] [PubMed] [Google Scholar]
- 39.Chernys JT, Zeevaart JA. Characterization of the 9-cis-epoxycarotenoid dioxygenase gene family and the regulation of abscisic acid biosynthesis in avocado. Plant Physiology. 2000;124(1):343–53. 10.1104/pp.124.1.343 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Xue-Zhong XU, Wang T, Wan W, Si-Hui LI, Zhu GH. ABA Biosynthesis Gene OsNCED3 Confers Drought Stress Tolerance in Rice. Acta Agronomica Sinica. 2018. [Google Scholar]
- 41.Xu P, Cai W. Functional characterization of the BnNCED3 gene in Brassica napus. Plant Science. 2017;256:16–24. 10.1016/j.plantsci.2016.11.012 [DOI] [PubMed] [Google Scholar]
- 42.Cheng F, Liu S, Wu J, Fang L, Sun S, Liu B, et al. BRAD, the genetics and genomics database for Brassica plants. Bmc Plant Biology. 2011;11(1):1–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Altschul SFM T.L.; Schäffer A.A.; Zhang J.; Zhang Z.; Miller W.; Lipman D.J. Gapped BLAST and PSI-BLAST: a new generation of protein detabase search programs. Nucleic Acids Research. 1997;25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Cheng F, Shengyi L, Jian W, Lu F, Silong S, Bo L, et al. BRAD, the genetics and genomics database for Brassica plants. 2011;11(1):136–0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Sudhir K, Glen S, Koichiro T. MEGA7: Molecular Evolutionary Genetics Analysis Version 7.0 for Bigger Datasets. Molecular Biology & Evolution. 2016(7):7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Finn RD, Mistry J, Tate J, Coggill P, Heger A, Pollington Joanne E, et al. The Pfam protein families database. Nucleic Acids Research. 2009(suppl_1):suppl_1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Edgar RC. MUSCLE: a multiple sequence alignment method with reduced time and space complexity. 2004;5(1):113–0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Capella-Gutierrez S, Silla-Martinez JM, Gabaldon T. trimAl: a tool for automated alignment trimming in large-scale phylogenetic analyses. Bioinformatics. 2009;25(15):1972–3. 10.1093/bioinformatics/btp348 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Xia X. DAMBE5: A Comprehensive Software Package for Data Analysis in Molecular Biology and Evolution. Molecular Biology & Evolution. 2013(7):7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Voorrips RE. MapChart: Software for the graphical presentation of linkage maps and QTLs. The Journal of Heredity. 2002;1(93). [DOI] [PubMed] [Google Scholar]
- 51.Elisabeth G, Alexandre G, Christine H, Ivan I, Appel RD, Amos B. ExPASy: the proteomics server for in-depth protein knowledge and analysis. Nucleic Acids Research. 2003(13):13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Bailey TL, Nadya W, Chris M, Li WW. MEME: discovering and analyzing DNA and protein sequence motifs. Nucleic Acids Research. 2006(suppl_2):suppl_2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Zdobnov EM, Rolf A. InterProScan–an integration platform for the\n signature-recognition methods in InterPro\n. Bioinformatics. 2001(9):9. [DOI] [PubMed] [Google Scholar]
- 54.Chen C, Xia R, Chen H, He Y. TBtools, a Toolkit for Biologists integrating various HTS-data handling tools with a user-friendly interface. bioRxiv. 2018:289660. [Google Scholar]
- 55.Koch MA, Haubold B, Mitchell-Olds T. Comparative evolutionary analysis of chalcone synthase and alcohol dehydrogenase loci in Arabidopsis, Arabis, and related genera (Brassicaceae). Molecular Biology & Evolution. 2000;17(10):1483. [DOI] [PubMed] [Google Scholar]
- 56.Paul H, Keun-Joon P, Takeshi O, Naoya F, Hajime H, Adams-Collier CJ, et al. WoLF PSORT: protein localization predictor. Nucleic Acids Research. 2007(suppl_2):suppl_2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Chalhoub B, Denoeud F, Liu S, Parkin IAP, Tang H, Wang X, et al. Early allopolyploid evolution in the post-Neolithic Brassica napus oilseed genome. Science. 2014;345(6199):950–3. 10.1126/science.1253435 [DOI] [PubMed] [Google Scholar]
- 58.Lescot M. PlantCARE, a database of plant cis-acting regulatory elements and a portal to tools for in silico analysis of promoter sequences. Nucleic Acids Research. 2002;30(1):325–7. 10.1093/nar/30.1.325 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Lu K, Wei L, Li X, Wang Y, Wu J, Liu M, et al. Whole-genome resequencing reveals Brassica napus origin and genetic loci involved in its improvement. 2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Trapnell C, Roberts A, Goff L, Pertea G, Kim D, Kelley DR, et al. Differential gene and transcript expression analysis of RNA-seq experiments with TopHat and Cufflinks. NATURE PROTOCOLS. 2012;7(3):562–78. 10.1038/nprot.2012.016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Kj L, Td S. Analysis of Relative Gene Expression Data Using Real-Time Quantitative PCR and the 2-△△Ct Method. Methods. 2000;25(4). [DOI] [PubMed] [Google Scholar]
- 62.Guo C, Guo R, Xu X, Gao M, Li X, Song J, et al. Evolution and expression analysis of the grape (Vitis vinifera L.) WRKY gene family. Journal of Experimental Botany. 2014;65(6):1513–28. 10.1093/jxb/eru007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Boulos C, France D, Shengyi L, Parkin IAP, Haibao T, Xiyin W, et al. Plant genetics. Early allopolyploid evolution in the post-Neolithic Brassica napus oilseed genome. Science. 2014;345(6199):950–3. 10.1126/science.1253435 [DOI] [PubMed] [Google Scholar]
- 64.Cheng F, Wu J, Wang X. Genome triplication drove the diversification of Brassica plants. Horticulture Research. 2014;1:14024 10.1038/hortres.2014.24 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Son SH, Chitnis VR, Liu A, Gao F, Nguyen TN, Ayele BT. Abscisic acid metabolic genes of wheat (Triticuma estivum L.): identification and insights into their functionality in seed dormancy and dehydration tolerance. Planta. 2016;244(2):429–47. 10.1007/s00425-016-2518-2 [DOI] [PubMed] [Google Scholar]
- 66.Tian X, Ji J, Wang G, Jin C, Guan C, Wu D, et al. Cloning and Expression Analysis of 9-cis-Epoxycarotenoid Dioxygenase Gene 1 Involved in Fruit Maturation and Abiotic Stress Response inLycium chinense. Journal of Plant Growth Regulation. 2015;34(3):465–74. [Google Scholar]
- 67.Wang Z, Dan C, Yue C, Cao H, Guo Y. Cloning and Expression Analysis of CsNCED2 Gene in Tea Plant(Camellia sinensis). Acta Botanica Boreali-Occidentalia Sinica. 2018. [Google Scholar]
- 68.Lin H, Wang R, Qian Q, Yan M, Meng X, Fu Z, et al. DWARF27, an Iron-Containing Protein Required for the Biosynthesis of Strigolactones, Regulates Rice Tiller Bud Outgrowth. Plant Cell. 2009;21(5):1512–25. 10.1105/tpc.109.065987 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Zhang Y, Yang J, Lu S, Cai J, Guo Z. Overexpressing SgNCED1 in Tobacco Increases ABA Level, Antioxidant Enzyme Activities, and Stress Tolerance. Journal of Plant Growth Regulation. 2008;27(2):151–8. [Google Scholar]
- 70.Schwartz S H. Characterization of a Novel Carotenoid Cleavage Dioxygenase from Plants. Journal of Biological Chemistry. 2001;276(27):25208–11. 10.1074/jbc.M102146200 [DOI] [PubMed] [Google Scholar]
- 71.Schmidt H, Kurtzer R, Eisenreich W, Schwab W. The Carotenase AtCCD1 from Arabidopsis thaliana Is a Dioxygenase. Journal of Biological Chemistry. 2006;281(15):9845–51. 10.1074/jbc.M511668200 [DOI] [PubMed] [Google Scholar]
- 72.Garci?A-Limones C, Schna?bele K, Blanco-Portales R, Luz Bellido M, Caballero JL, Schwab W, et al. Functional Characterization of FaCCD1: A Carotenoid Cleavage Dioxygenase from Strawberry Involved in Lutein Degradation during Fruit Ripening. J Agric Food Chem. 2008;56(19):9277–85. 10.1021/jf801096t [DOI] [PubMed] [Google Scholar]
- 73.Simkin AJ, Underwood BA, Auldridge M, Loucas HM, Shibuya K, Schmelz E, et al. Circadian Regulation of the PhCCD1 Carotenoid Cleavage Dioxygenase Controls Emission of β-Ionone, a Fragrance Volatile of Petunia Flowers. Plant Physiology. 2004;136(3):3504–14. 10.1104/pp.104.049718 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Sabrina GJ, Sun-Hwa H, Maria ML, Laura Ullrich G, Ailing Z, Lipka AE, et al. Carotenoid cleavage dioxygenase4 is a negative regulator of β-carotene content in Arabidopsis seeds. Plant Cell. 2013;25(12):4812–26. 10.1105/tpc.113.119677 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Shu H, Guo S, Gong Y, Lu J, Zhu J, Ni W. Advances in Crocin Biosynthesis and CCD Gene Family. Chinese Agricultural Science Bulletin. 2016. [Google Scholar]
- 76.Rachele F, Elisa V, Laura Z, Simone S, Guido C, Ignazio V, et al. Three distinct mutational mechanisms acting on a single gene underpin the origin of yellow flesh in peach. Plant Journal for Cell & Molecular Biology. 2013;76(2):175–87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Ureshino K, Nakayama M, Miyajima I. Contribution made by the carotenoid cleavage dioxygenase 4 gene to yellow colour fade in azalea petals. Euphytica. 2016;207(2):401–17. [Google Scholar]
- 78.Liu H, Kishimoto S, Yamamizo C, Fukuta N, Ohmiya A, Debener T. Carotenoid accumulations and carotenogenic gene expressions in the petals of\\r Eustoma grandiflorum. Plant Breeding. 2013;132(4):417–22. [Google Scholar]
- 79.Zhang B, Liu C, Wang Y, Yao X, Liu K. Disruption of a CAROTENOID CLEAVAGE DIOXYGENASE 4 gene converts flower colour from white to yellow in Brassica species. New Phytologist. 2015;206(4):1513–26. 10.1111/nph.13335 [DOI] [PubMed] [Google Scholar]
- 80.Jo YD, Kim Y-S, Ryu J, Choi H-I, Kim SW, Kang HS, et al. Deletion of carotenoid cleavage dioxygenase 4a (CmCCD4a) and global up-regulation of plastid protein-coding genes in a mutant chrysanthemum cultivar producing yellow petals. Scientia Horticulturae. 2016;212:49–59. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(XLSX)
(XLSX)
(XLSX)
(XLSX)
Data Availability Statement
All relevant data are within the manuscript and its Supporting Information files.