Abstract
Research and development of the ideal artificial bone-substitute materials to replace autologous and allogeneic bones for repairing bone defects is still a challenge in clinical orthopedics. Recently, poly(lactic-co-glycolic acid) (PLGA)-based artificial bone-substitute materials are attracting increasing attention as the benefit of their suitable biocompatibility, degradability, mechanical properties, and capabilities to promote bone regeneration. In this article, we comprehensively review the artificial bone-substitute materials made from PLGA or the composites of PLGA and other organic and inorganic substances, elaborate on their applications for bone regeneration with or without bioactive factors, and prospect the challenges and opportunities in clinical bone regeneration.
Keywords: Poly(lactic-co-glycolic acid), Composite organic−inorganic biomaterial, Bone-substitute material, Bone tissue engineering, Bone regeneration
Graphical abstract
Highlights
-
•
Poly(lactic-co-glycolic acid) (PLGA) is one of the most promising biodegradable polymers for biomedical applications.
-
•
PLGA-based scaffolds have suitable biocompatibility, degradability, mechanical properties, and osteoinductivity.
-
•
PLGA-based bone-substitute materials combined with cells, growth factors, and drugs promote bone regeneration effectively.
-
•
PLGA-based bone-substitute materials exhibit an excellent clinical prospect for bone repair.
1. Introduction
Trauma [1], bone infection [2], osteonecrosis [3], osteoporosis [4,5], bone tumors [6,7], and iatrogenic injury [8] always lead to primary or secondary bone defects. At present, there are several clinical managements for bone defects. Among them, bone transport is an effective bone repair strategy, but it generally needs to break the bone at one end of the defect and pull it to the other end [9]. The broken bone has self-osteogenic ability [10]. For example, a bone defect of 3 cm can be handled by 1 mm per day and will be regenerated in about one month [11,12]. Bone transport has a long treatment course and shows some disadvantages, such as the pain during the bone regeneration, the susceptibility to be infected of the bone defects area, and the high restrictions on the location and size of bone defects.
The bone graft is a mainstream strategy for the clinical treatment of bone defects. By using different grafts, bone defects would efficiently regenerate under different conditions. The familiar sources of the grafts roughly divided into three categories [[13], [14], [15]]. 1. Bones taken from the autologous healthy areas, such as the ilium or vascularized fibula [16]. Autologous bone graft has excellent compatibility and powerful healing capability. However, patients need to endure the pain in the bone removal area. 2. Allogeneic bones, such as post-processing human bone obtained from the donation of remains. The application of allogeneic bones may avoid the destruction of patients' healthy bone structure. However, it still has limitations, such as insufficient supply, ethical disputes, and immunogenicity [17]. 3. Bone from other species, such as processed products of bovine bones. The source of xenogeneic bones is sufficient, but these grafts will put patients at the risk of immune rejection and virus transmission [18]. For these reasons, it has become a demand to prepare artificial bone-substitute materials.
In order to meet the demands, artificial bone-substitute materials require a variety of properties to maintain bone strength at the area of bone defects and promote bone regeneration, including similar mechanical strength to bone tissue, excellent biocompatibility and biodegradability, as well as a porous network structure for cells to grow in [[19], [20], [21], [22], [23]]. The existing bone-substitute materials are summarized in Table 1.
Table 1.
Bone-substitute materials.
| Material | Strength | Weakness | Reference |
|---|---|---|---|
| HA | Excellent biocompatibility, osteoconductivity | Poor degradability, lack of osteoinductivity, brittle | [24,25] |
| TCP | Excellent biocompatibility, high safety, easy to obtain | Poor processability, poor mechanical strength | [26,27] |
| BCP | Osteogenesis induction, ALP activity, and mineralization ability | Poor degradability, poor processability | [28,29] |
| BG | Special bone-bonding ability | Disordered bone growth, local pH change, brittle | [30] |
| CNTs | Excellent mechanical properties, good biocompatibility, non-toxicity | – | [31,32] |
| Stainless steel | High strength, corrosion resistance | Non-degradability, stress shielding, inflammation and immune response | [35,40] |
| Titanium alloy | High tensile strength, corrosion resistance, shape memory effect, excellent biocompatibility | Non-degradability, stress shielding | [[36], [37], [38]] |
| Cobalt-chromium alloy | Wear resistance, corrosion resistance | Non-degradability | [39] |
| Coral | High porosity, high mechanical strength, bone conductivity | Poor degradability | [41,42] |
| Col | Excellent biocompatibility, suitable pore size, good plasticity, weak antigenicity | Low mechanical strength, difficult to shape, unstable performance, easy to be dissolved by collagenase | [45,46] |
| Chitosan | Excellent biocompatibility, controllable degradability, non-toxicity, inhibition of inflammation | Poor mechanical properties | [47,48] |
| Poly(methyl methacrylate) | Durability, easy to process | Non-degradability | [49,50] |
| PCL | Biodegradability, machinability, high porosity | Low mechanical strength | [51,52] |
| PLA | Biodegradability, strong mechanical properties, excellent biocompatibility, promote bone regeneration | local pH change | [[53], [54], [55]] |
Researchers used to select a series of inorganic substances as bone-substitute materials, such as hydroxyapatite (HA) [24,25], tricalcium phosphate (TCP) [26,27], biphasic calcium phosphate (BCP) [28,29], bioglass (BG) [30], and carbon nanotubes (CNTs) [31,32]. Although these materials showed good biocompatibility, they are too brittle to maintain bone strength [33]. To address this problem, researchers prepared a series of metallic bone substitutes with high strength and stability [[34], [35], [36], [37], [38], [39]]. However, these metallic bone substitutes may lead to stress shielding and non-biocompatibility degradation products [40]. Some animal-derived materials, such as microwave sintered cuttlefish bone and coral, are also used as bone-substitute materials [41,42]. However, these materials lack osteoinductivity and have a slow degradation rate. More recently, collagens (Cols) and fibrin glues showed a wide application in bone defect repair due to the good plasticity [[43], [44], [45], [46]]. However, the therapeutic efficacy is poor owing to its low mechanical strength and readily to be degraded by collagenase. Polymer-based artificial bone substitutes showing great potential in bone repair [[47], [48], [49], [50], [51], [52], [53], [54], [55], [56], [57]]. In comparison with previous materials, synthetic polymers have a range of advantages, such as controllable molecular mass and degradation time, excellent processability, and good mechanical properties.
Poly(lactic-co-glycolic acid) (PLGA) is one of the most widely used biodegradable polymers. PLGA is a linear copolymer of lactic acid and glycolic acid monomers. Lactic acid could be obtained from the fermentation of corn or other grains [58]. Glycolic acid could be obtained by the biochemical enzymatic reaction or by chemical synthesis with chloroacetic acid and sodium hydroxide [59]. The synthesis strategies for PLGA could be divided into two categories. One of them is the polycondensation process of lactic acid and glycolic acid [60,61]. This method usually obtains PLGA with low molecular weight and wide relative molecular weight distribution. The other method is the ring-opening polymerization (ROP) of lactide and glycolide [62,63]. This method could obtain products with a higher molecular weight and more uniform relative molecular weight. The performance of PLGA could also be adjusted by the different ratios of lactide and glycolide.
The controllable degradation time is an essential feature of PLGA. The hydrolysis process of PLGA could be divided into two stages. In the first stage, the molecular weight of PLGA gradually decreases, while the overall weight remains stable. In the second stage, the molecular weight of PLGA drops until the products are soluble in water, and a rapid decrease in the overall weight will appear [64]. By controlling the ratio of lactic acid and glycolic acid, the mechanical properties and degradation performance could be flexibly controlled [65]. When the ratio of lactic acid and glycolic acid monomers is 50/50, the degradation rate is the fastest [66]. Previous studies have shown that PLGA scaffolds with the lactic acid and glycolic acid ratios of 85/15 and 75/25 have degradation periods of approximately 8 and 16 weeks, respectively [67]. Compared with in PBS only, in the condition that simulates the presence of enzymes in the organism, the degradation rate of the porous PLGA scaffold was further accelerated [68].
PLGA is suitable for bone repair for various reasons [69]. First of all, PLGA has excellent biocompatibility. Its metabolites are only lactic acid and glycolic acid, which can be safely eliminated from the metabolic cycle [70]. Secondly, PLGA has excellent processability. Processors could use a variety of processes to prepare PLGA scaffolds [71,72]. Third, by adjusting the ratio of lactic acid and glycolic acid, the degradation time of PLGA can be controlled to be around a few months [67], so that consistent with bone regeneration. With the gradual degradation of PLGA, the stress stimulation to the new bone is gradually strengthened, which can promote bone regeneration and bone structure remodeling. Fourth, PLGA has suitable mechanical strength to support the defect area in the early stage adequately. Fifth, PLGA grafts can be loaded with a variety of bioactive factors, such as stem cells, growth factors, and drugs, to promote regeneration of bone defects [73,74].
On account of the aforementioned advantageous properties, PLGA and composite materials of PLGA and inorganic components have been widely studied (Scheme 1). PLGA-based bone-substitute materials have been summarized in Table 2. This article reviews the recent research and development of single and composite materials based on PLGA in artificial bone tissue materials. The possible prospect and future development are also discussed.
Scheme 1.
PLGA-based bone-substitute materials used in treatment of bone defects.
Table 2.
PLGA-based bone-substitute materials.
| Material | lactic acid/glycolic acid molar ratio | Preparation method | Bioactive factor | Bone defect model | Defect size | Implantation time | Final BV/TV | Ref. |
|---|---|---|---|---|---|---|---|---|
| PLGA | 75/25 | Unidirectional cooling method | – | Rabbit articular osteochondral defect | Diameter 4 mm, depth 4 mm | 12 weeks | – | [79] |
| PLGA | 85/15 | Compression molding/particulate leaching method | BM-MSCs | Rabbit articular osteochondral defect | Diameter 4 mm, depth 5 mm | 12 weeks | – | [76] |
| PLGA | 85/15 | Compression molding/particulate leaching method | BM-MSCs | Rabbit articular osteochondral defect | Diameter 4 mm, depth 5 mm | 12 weeks | – | [77] |
| PLGA | 75/25 | Compression molding/particulate leaching method | BM-MSCs | Rabbit articular osteochondral defect | Diameter 4 mm, depth 5 mm | 24 weeks | – | [78] |
| PLGA | 85/15 | Compression molding/particulate leaching method | PRP | Rabbit articular osteochondral defect | Diameter 4 mm, depth 4 mm | 6 months | 57 ± 14% | [83] |
| PLGA | 85/15 | Gas foaming/salt leaching method | Human AD-MSCs | Rat calvarial defect | Diameter 7 mm | 16 weeks | 11 ± 2.8% | [86] |
| PLGA | 75/25 | ST and FT 3D bioprinting methods | – | – | – | – | – | [72] |
| PLGA | 75/25 | 3D printing method | – | – | – | – | – | [80] |
| PLGA/Artificial extracellular matrix | 85/15 | Solvent casting/particulate leaching method | – | – | – | – | – | [87,88] |
| PLGA/Col | 75/25 | 3D printing method | BMP2-MP | – | – | – | – | [90] |
| PLGA/HA | 85/15 | Compression molding/particulate leaching method | – | Rabbit radius defect | Length 15 mm | 12 weeks | [94] | |
| PLGA/HA | 50/50 | Melt reaction with transesterification method | – | – | – | – | – | [96] |
| PLGA/HA | 75/25 | Solvent casting/particulate leaching method | – | Rabbit hind femoral condyles defect | Diameter 6 mm, depth 5 mm | 12 months | 23 ± 4% | [97] |
| PLGA/HA | 50/50 | Electrospinning and freeze-drying methods | – | – | – | – | – | [99] |
| PLGA/HA | 85/15 | Gas foaming/salt leaching method | BM-MSCs | Rabbit articular osteochondral defect | Diameter 4 mm, depth 5 mm | 16 weeks | – | [98] |
| PLGA/HA | 85/15 | Electrospinning method | Insulin | – | – | – | – | [105] |
| PLGA/HA | – | Double emulsion solvent evaporation method | BMP-2 and VEGF | – | – | – | – | [106] |
| PLGA/HA | 50/50 | Combining phase separation/particulate leaching method | rhBMP-7 | Mice calvarial defect | Diameter 4 mm | 8 weeks | – | [108] |
| PLGA/HA | 75/25 | 3D printing method | rhBMP-2 | Rabbit mandible defect | Length 13 mm, width 6 mm, height 4 mm | 12 weeks | 33.7% | [107] |
| PLGA/HA-g-PBLG | 75/25 | Phase inversion/particulate leaching method | – | Rabbit radius defect | Length 20 mm | 12 weeks | – | [100] |
| PLGA/HA-g-PBLG | – | Solvent casting/particulate leaching method | – | Rabbit radius defect | Length 20 mm | 12 weeks | 75% | [101] |
| PLGA/CP | – | Compression molding method | Rabbit BM-MSCs | Rabbit radius defect | Length 15 mm | 8 weeks | – | [109] |
| PLGA/TCP | 50/50 | Rapid prototyping method | VEGF | Rabbit radius defect | Length 15 mm | 8 weeks | – | [114] |
| PLGA/TCP | 75/25 | Low-temperature deposition technique | Icariin | – | – | – | – | [116] |
| PLGA/TCP | 75/25 | 3D printing method | Mg | Rabbit distal femora defect | Diameter 3 mm, length 15 mm | 12 weeks | 8% | [115] |
| PLGA/TCP | 75/25 | 3D printing method | Icariin | Rabbit distal femora defect | Diameter 3 mm, length 20 mm | 12 weeks | – | [117] |
| PLGA/TCP | 75/25 | 3D printing method | Icariin | Rabbit distal femora defect | Diameter 3 mm, length 15 mm | 8 weeks | 7% | [118] |
| PLGA/BG/Col | 50/50 | Single emulsion method | – | Rat calvarial defect | Diameter 5 mm | 6 weeks | – | [122] |
| PLGA/CNTs | – | Solvent casting/particulate leaching method | – | – | – | – | – | [125] |
| PLGA/MgOH | 50/50 | Solvent casting method | – | Rat articular osteochondral defect | Diameter 2 mm, depth 2 mm | 8 weeks | – | [126] |
| PLGA/AKR | 75/25 | Solvent casting/particulate leaching method | iPSCs | Mice calvarial defect | Diameter 4 mm | 8 weeks | – | [124] |
2. PLGA-based bone-substitute materials
2.1. PLGA bone-substitute materials
A variety of PLGA-based drug delivery systems have been approved for clinical applications by medical institutions in the United States and Europe. PLGA scaffolds have become an ideal choice for the preparation of artificial bone-substitute materials. The emergence of bone tissue engineering makes the combined application of PLGA-based scaffolds, stem cells, and bioactive agents more promising.
Researchers prepared a series of double-layer PLGA-integrated scaffolds to achieve synchronous repair of osteochondral tissue [[75], [76], [77], [78]]. The researchers made two kinds of scaffolds with different pore sizes and then glued them become an integrated scaffold. They controlled the pore sizes of PLGA scaffolds in different layers to simulate different biomechanical characteristics of bone and cartilage tissue. The mean Young's moduli of the scaffolds were 91.2 MPa. Moreover, they also implanted bone marrow-derived mesenchymal stem cells (BM-MSCs) in the scaffolds. Finally, the scaffolds were implanted into a rabbit model of osteochondral defect on the femoral condyle's articular surface (Fig. 1A). The results showed that the scaffolds with BM-MSCs had better osteogenesis properties than the pure scaffolds within 12 weeks (Fig. 1B and C). Moreover, the remaining scaffolds had become sparse at 24 weeks. It showed that PLGA is an artificial bone scaffold material with well biocompatibility and processability. At the same time, it is proved that the double-layer PLGA scaffold is beneficial for the simultaneous regeneration and repair of different tissues.
Fig. 1.
Effects of PLGA scaffolds with different pore sizes on bone repair [78]. (A) Schematic diagram of double-layer PLGA scaffolds applied to rabbit knee joint osteochondral defect treatment. (B) Specimen pictures and (C) H&E staining after treatment 12 and 24 weeks. The red circle shows the recovery of the defect model. Yellow arrow indicates the interface between new tissue and natural osteochondral tissue. The length of the white vertical line is 1 mm.
Dai et al. prepared another PLGA scaffold with directional holes by radial unidirectional cooling technology [79]. The porosity of the scaffold is higher than 90%. In the dry state, the compression modulus of the scaffold is 4 MPa. Researchers characterized the directionality of the scaffold holes and then repaired the model of rabbit osteochondral defect with the scaffold. At 12 weeks, new tissue was formed, and the scaffold was degraded. The results showed that the migration of cells in the scaffold with directional holes was better, the distribution of the cells was more uniform, and the regeneration of bone and cartilage was more substantial than that in the PLGA scaffold with non-directional holes. The results suggested that the consistency of the hole direction inside the PLGA scaffold has a beneficial effect on bone tissue repair.
The traditional preparation methods of PLGA-based artificial bone scaffolds include particle leaching, phase separation, gas foaming, and fiber bonding methods. Although these methods are mature in technology and rapid in preparation, there are still some deficiencies: 1. It is hard to accurately control the size, shape, and overall porosity, 2. It is weak in preparing stability and has solvent residues in the scaffolds. The rise of 3D printing technology provides more productive methods for the preparation of PLGA scaffolds than the traditional preparation methods.
Liu et al. used 3D printing technology to prepare PLGA scaffolds with different printing angles [80]. They designed a series of different scaffolds by controlling the arrangement of the scaffold, the thickness of each layer, and the porosity (Fig. 2A). The researchers measured Young's modulus and compressive strength of these scaffolds (Fig. 2B and C), and observed the adhesion, proliferation, and differentiation of pre-osteoblasts attached to the scaffolds. The results showed that the scaffolds with a 0°/90° printing angle achieved best cell adhesion, proliferation, and differentiation while possessing suitable mechanical properties and sufficiently long degradation time.
Fig. 2.
Effects of PLGA scaffolds with different printing angles on bone repair [80]. (A) Structure of PLGA scaffolds printed at different angles. (B) Differences in yield strength. (C) Compressive strength between different groups of scaffolds. Data are represented as mean ± SD (standard deviation; n = 5, **P < 0.01).
In another research, Lee et al. compared two manufacturing processes of 3D-printed PLGA scaffolds [72]. They used the manufacturing process of the syringe type (ST) and filament type (FT) distribution modules to print the PLGA scaffold, respectively. After that, researchers compared the scaffolds prepared with the two processes on molecular weight, glass transition temperature, biocompatibility, and degradability. The PLGA scaffold made by the ST method requires high temperature during preparation, so the problems of decreased molecular weight, thermal degradation, and acid by-products have existed. The FT method solves the problem in a step-by-step manner. However, the preparation process is more complicated than the ST method. The above results suggested that researchers should comprehensively evaluate the specific characteristics of materials in 3D printing and chose the appropriate processing technology.
PLGA scaffolds have been used as carriers of bioactive factors [81]. It has been reported that adding vascular endothelial growth factor (VEGF) to gels of the combination of PLGA and methoxy poly(ethylene glycol) (PEG) could achieve the effect of improved local vascularization and promoted bone regeneration on ischemic necrosis of the femoral head [82]. Platelet-rich plasma (PRP) is obtained by centrifugation of autologous whole blood, containing a lot of growth factors and proteins. By adding PRP to the PLGA scaffolds, the bone repairability of the scaffold is significantly improved [83].
Adipose tissue-derived mesenchymal stem cells (AD-MSCs) have the ability to differentiate osteoblasts and chondrocytes, are easy to extract, and have better proliferation ability compared with BM-MSCs [84,85]. Wang et al. prepared a PLGA porous scaffold and seeded AD-MSCs on the surface of the scaffold [86]. The researchers applied this scaffold and used low-power laser irradiation (LPLI) to treat a rat skull bone defect model. The results showed that the combination therapy improved the repair effect of the skull defect. The results enriched the application of PLGA scaffolds.
2.2. Artificial extracellular matrix-covered PLGA bone-substitute materials
The hydrophobicity of PLGA limited the adhesion of cells and proteins. Moreover, it is difficult for the culture medium to enter the high-porosity PLGA scaffold under normal circumstances, which will affect the efficiency of cell seeding [87]. These problems might be effectively solved by covering the surface of the materials with an artificial extracellular matrix [88]. Adding fibronectin, vitronectin, Col, or fibrin to the surface of materials will enhance the adhesion of cells to the polymer scaffolds [89].
The researchers proposed a strategy to cover the surface of the scaffold with an artificial extracellular matrix. This method simulated the composition and structure of natural bones, thereby improving the cytocompatibility of PLGA scaffolds [87]. This artificial extracellular matrix consists of type I collagen (Col I), chondroitin sulfate, and hyaluronic acid sulfate. They compared the ability of this scaffold with other scaffolds that did not encapsulate the extracellular matrix to induce the differentiation of hMSCs. The results showed that the content of alkaline phosphatase (ALP) in the medium increased, and the ability of stem cells to differentiate into osteoblasts was enhanced in the presence of an artificial extracellular matrix. The researchers further explored the influence of the surface microstructures of PLGA scaffolds on the differentiation of hMSCs [88]. They prepared a PLGA scaffold with a pore diameter of several hundreds of micrometers and another PLGA scaffold with two different pore diameters of several micrometers and hundreds of micrometers. Each of the scaffolds was covered with Col I and high-sulfated hyaluronan. They cultured hMSCs on scaffolds and observed their differentiation. The results showed that the expression of Runt-related transcription factor 2 (Runx2), osteopontin, and bone sialoprotein II (BSP II) in the bimodal pore distribution group was higher than that in the single pore distribution one. The results further complement people's understanding of the effect of scaffold's surface microstructure on the differentiation of hMSCs. Moreover, the impact of artificial extracellular matrix-covered PLGA scaffold on hMSCs differentiation was verified again.
Col is an excellent carrier of growth factors. The addition of growth factors functionalizes the scaffold covered with Col. Researchers combined BMP-2 mimic peptide (BMP2-MP) with Col and covered it on the surface of PLGA scaffolds [90]. The compressive strength of PLGA/Col scaffold was 16.03 MPa. Besides, researchers evaluated the surface characteristics of scaffolds to affect the proliferation and differentiation of MC3T3-E1 cells. The results showed that the scaffold achieved better cytocompatibility due to the increased hydrophilicity of the surface based on the proper biomechanical properties. MC3T3-E1 cells showed good osteogenic differentiation ability on this scaffold, and the expression of osteogenic genes was significantly increased. The results suggested that the coverage of BMP2-MP and Col is an effective method for improving the osteogenic induction capacity of PLGA scaffolds.
In summary, in order to make PLGA scaffolds have well cell compatibility, suitable biodegradability, excellent mechanical properties, and different functionalization, the researchers conducted multi-layer composite and surface modification to the scaffolds. They adopted 3D printing technology to precisely control the scaffold structure. Among them, 3D printing technology is an essential direction for the future development of PLGA scaffolds prepared.
3. Composite bone-substitute materials of PLGA and inorganic components
HA, CP, and BG are all bone-substitute materials with their advantages. However, due to their inherent shortcomings, their effect of using them alone is not ideal. Composite materials that combine PLGA with these inorganic components would achieve better mechanical and biological properties than using them alone.
3.1. PLGA/HA composite bone-substitute materials
HA is used as an artificial bone-substitute material, owing to its similar mineral composition to human bones [91]. HA is osteoconductive, so that has a promoting effect on the formation of bone tissue [92]. However, due to its low fatigue resistance and poor degradability, it is not suitable to be used as artificial bone tissue material directly. Researchers found that scaffolds made of a mixture of PLGA with HA would achieve better mechanical properties than using two materials alone [93]. Therefore, researchers developed a variety of PLGA/HA composite materials by different manufacturing processes and explored the influence of various indicators of PLGA/HA composite materials on bone repair. In recent years, a variety of bioactive factors is loaded with PLGA/HA scaffolds to promote bone healing.
Researchers found that modifying the surface of the PLGA scaffold with nano-HA may enhance the bone repairability of scaffolds. This function comes from the formation of a biomimetic hierarchical nanostructure on the surface of the scaffolds, which is conducive to the proliferation and differentiation of BM-MSCs [94]. In another work, researchers prepared a variety of PLGA/HA scaffolds by in situ polymerization [95]. They control the formation of composite materials by adjusting the feed ratio to make scaffolds with various PLGA to HA ratios. The mechanical strength of these scaffolds is 54.3–78.8 MPa, 3–4 times higher than that of pure HA. By co-culturing with osteoblasts, the researchers confirmed their excellent biocompatibility. At the same time, the content of ALP in the culture medium proved the induction performance of the scaffolds on osteoblast differentiation. Besides, the other researchers explored the effect of grafting process of PLGA and HA on the performance of the scaffold [96]. They adopted melt grafting and transesterification methods to obtain PLGA/HA composites with higher grafting rates (Fig. 3A). The composite material prepared by this method had two times higher tensile strength (about 55 MPa) than that of the composite material made by the blending method (Fig. 3B−D). In another study, the researchers have explored the influence of the degradation rate of PLGA/HA scaffold on bone repair [97]. They concluded that long degradation time is necessary to obtain good bone repair results.
Fig. 3.
Effect of grafting process on tensile strength of PLGA/HA scaffolds [96]. (A) Schematic diagram of PLGA/HA scaffold preparation process. (B) Schematic diagram of difference in tensile strength of these scaffolds obtained by different preparation methods. Tensile strength (C) and tensile modulus (D) elongation of composite scaffolds with different HA grafting rates. Pre (50) means HA content of 50% in the pre-dispersion process, Pre (70) means HA content of 70% in the pre-dispersion process, Pre (80) means HA content of 80% in the pre-dispersion process, and Pre (90) means HA content of 90% in the pre-dispersion process. Data are represented as mean ± SD (n = 5). Reproduced with permission [96]. Copyright 2019, Elsevier Ltd.
PLGA/HA scaffolds loaded with stem cells have also been studied. The researchers prepared PLGA and PLGA/HA double-layer composite scaffold by molding/particle leaching [98] (Fig. 4A−C). The two parts of this composite scaffold have two Young's moduli, 2.8 and 20.3 MPa, respectively. That successfully simulated the different strengths required for cartilage and bone repair. The researchers applied the hMSC-seeded scaffold to repair the osteochondral defects in rabbit knee joints and obtained an excellent bone repair effect (Fig. 4D). Another researcher prepared the PLGA/HA scaffold through electrospinning technology and seeded the scaffold with stem cell-derived osteoblasts, which derived from human endometrial [99]. The outstanding ability of such scaffold for bone repair was verified by the results of ALP content, osteogenesis-related protein expression, and cell proliferation and differentiation.
Fig. 4.
Bilayered PLGA/HA composite scaffold for osteochondral tissue engineering [98]. (A) Efficacy comparison. (B) and (C) Schematic of composite scaffold in treatment of rabbit bone defects. (D) H&E staining pictures of treated specimens. The yellow arrows indicate the initial defect sites. Reproduced with permission [98]. Copyright 2018, American Chemical Society.
The addition of other polymers further improves the performances of the PLGA/HA scaffolds. Zheng et al. introduced poly(γ-benzyl-l-glutamate) (PBLG) in the preparation of composite scaffold. The compression modulus of the PBLG-added scaffold was 2.5 MPa, which was twice the compression modulus than before. Moreover, the PBLG-added scaffold achieved outstanding bone repair effect [100]. Li et al. prepared a series of PLGA/PBLG-HA scaffolds containing different proportions of PBLG [101]. Firstly, they polymerized PBLG onto HA, and then made PLGA/PBLG-HA scaffold by solvent casting/particle leaching (Fig. 5A). By adjusting the content of PBLG, the scaffold obtained a 1.5–3 MPa compressive strength and achieved different abilities to promote cell proliferation and differentiation. In the rabbit radius defect model, when the PBLG content was 22% and 33%, the composite scaffold could obtain the best new bone formation and mineralization effect (Fig. 5B and C). The above results suggest that a reasonable introduction of other polymers to the PLGA/HA scaffolds could enhance the therapeutic effect of bone repair.
Fig. 5.
Porous scaffolds of PLGA/PBLG-HA for bone repair in vivo [101]. (A) Schematic diagram of PLGA/PBLG-HA scaffold preparation process. (B) Results of 3D-CT of rabbit radial defects treated with scaffolds grafted with different ratios of PBLG. (C) Bone volume fraction statistics. Nothing (blank group) and porous scaffolds of PLGA, MHA/PLGA, MHA-APS/PLGA, and PBLG-g-MHA/PLGA with PBLG graft amounts of 11, 22, 33, and 50 wt%. Data are represented as mean ± SD (n = 4, *P < 0.05). Reproduced with permission [101]. Copyright 2019, American Chemical Society.
PLGA/HA scaffolds carrying bioactive factors can be implanted deep into the lesion area of patients to achieve in situ release for the treatment of local bone defects. Previously, there had been reports of preparing PLGA/HA/vitamin blend particles to make the controlled release of vitamin D3 for the treatment of skull defects [102]. Similarly, local delivery of simvastatin and recombinant human bone morphogenetic protein-2 (rhBMP-2) in PLGA/HA microspheres had also been studied [103,104].
Haider et al. reported a scheme of loading insulin to nanoscale HA and adopting electrospinning technology to prepare the PLGA/HA-I scaffold [105]. The proliferation and differentiation of osteoblasts were enhanced by the effect of PLGA/HA-I scaffold. In the study by Wang et al., MC3T3-E1 cells were cultured on a PLGA/HA composite scaffold loaded with VEGF and BMP-2 [106]. The cells proliferated and differentiated well on factor-loaded scaffolds, and the functional scaffold promoted the maturation of osteoblasts. In another study, chitosan nanoparticle encapsulated with BMP-2 was loaded on PLGA/HA scaffold [107]. After that, the rabbit mandible defect model was treated in 12 weeks, and the BV/TV value of the new bone area reached 33.7%. The excellent results confirmed its effectiveness in promoting bone repair. Another group of researchers grafted rhBMP-7 with polydopamine and then modified it to a PLGA/HA composite scaffold [108]. This scaffold exhibited good osteogenic proliferation and differentiation characteristics in vitro and was validated in the repair of a mouse skull defect model.
3.2. PLGA/CP composite bone-substitute materials
CP is another type of inorganic component in natural bone tissue. Because calcium (Ca) is a favorable factor for bone tissue repair, CP became a widely used inorganic material for constructing composite scaffolds with PLGA. In previous studies, bone cement of CP was combined with PLGA to prepare composite materials for artificial bones [109]. One day after preparation, the material obtained a compressive strength of more than 20 MPa and an elastic modulus of more than 9 MPa. After seven days, these intensity indicators further increased. Rabbit radius defect was repaired in eight weeks, which proved excellent bone repairability of the composite scaffold. The effect of particle size on bone repair has also been studied [110]. Results showed that nanoscale PLGA/CP material had a better bone repair capability than common PLGA/CP material. By adding BMP, VEGF, and other active ingredients to PLGA/CP microsphere, researchers achieved satisfying bone repair results [111].
In the other two studies, PLGA/TCP scaffold carrying rhBMP-2 or bovine BMP (bBMP) was developed [112,113]. The shape of the scaffold disintegrated at 6 weeks and disappeared completely at 24 weeks. In animal models, the excellent bone repair effect of the scaffold was observed. Duan et al. used genetic recombination technology to design BM-MSCs that express VEGF and then mounted the cell onto a PLGA/TCP scaffold [114]. This composite scaffold achieved local expression of VEGF for up to three weeks. This scaffold had shown excellent angiogenic effects in vivo and thus enhanced the bone repair. Similarly, it has been reported that Mg ion was introduced into PLGA/TCP scaffolds to achieve more robust mechanical properties and better angiogenesis capabilities than before, so that achieved satisfying bone repair results (Fig. 6) [115]. The PLGA/TCP scaffold loaded with icariin, a natural plant component, could promote bone repair by promoting local blood supply [[116], [117], [118]]. In another report, a therapeutic strategy for a PLGA/TCP composite scaffold combined with topical aspirin administration was investigated [119]. A low dose of aspirin combined with the PLGA/TCP scaffold would improve the local bone regeneration effect by affecting local angiogenic capacity and inhibiting osteoclast activity.
Fig. 6.
Osteogenic Mg incorporated into PLGA/TCP porous scaffold by 3D printing [115]. (A) Surface structures of PLGA/TCP/Mg scaffold and deposition of Ca and Mg. 3D-CT results of bone growth (B) and angiogenesis (C) at 4 and 8 weeks after treatment with different component scaffolds. PT indicates the PLGA/TCP group. PTM indicates the PLGA/TCP/Mg group. Red rectangle represents the bone tunnel site. Reproduced with permission [115]. Copyright 2019, Elsevier Ltd.
3.3. PLGA/BG composite bone-substitute materials
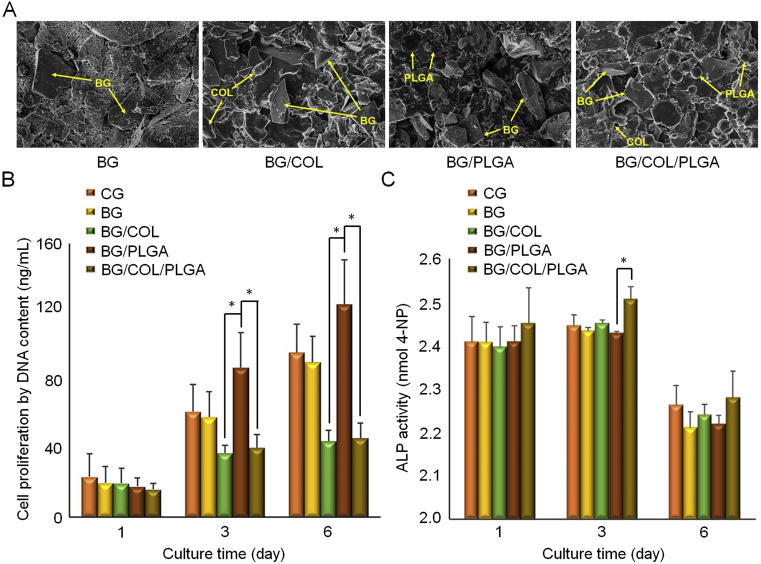
As a class of synthetic bioactive materials based on silicon (Si), BG is used to repair bone defects because of its excellent bone tissue binding ability and osteogenic properties [120,121]. However, due to its poor chemical stability and low mechanical strength, it is not an ideal artificial bone material. Solutions for applying BG to the preparation of synthetic bone-substitute materials were explored. Magri et al. prepared a series of BG-containing composite scaffolds by mixing BG with PLGA or Col or mixing them three [122] (Fig. 7A). In terms of the effect on the proliferation, the proliferation ability of osteoblasts implanted on the PLGA/BG scaffold is better than that of other groups (Fig. 7B). However, in the ALP activity assay of MC3T3-E1 cells, osteoblasts implanted on the PLGA/BG/Col scaffold achieved the highest score (Fig. 7C). In the rat skull defect model, PLGA/BG/Col composite scaffold was observed to degrade gradually at 2 and 6 weeks. The degradation of products maintained a local physiological pH. Furthermore, the PLGA/BG/Col group showed the best bone repair effect. This suggested that the PLGA/BG/Col composite bone-substitute materials have a good application prospect in the field of bone repair.
Fig. 7.
BG-based materials with hierarchical porosity [122]. (A) Microstructure of composite scaffolds with different components. Statistics of cell proliferation activity (B) and ALP activity (C) when different scaffolds are used for osteoblast culture. Data are represented as mean ± SD (n = 4, *P < 0.05). Reproduced with permission [122]. Copyright 2019, Elsevier Ltd.
3.4. PLGA-based bone-substitute materials compounded with other inorganic substances
In addition to HA, CP, and BG mentioned above, other inorganic materials had been used in combination with PLGA to constitute composite artificial bone-substitute materials. Previous studies had found that the Si released from the Si-based bioceramics akermanite (AKR) was able to induce differentiation of human-induced pluripotent stem cells (iPSCs) [123]. Therefore, researchers developed PLGA/AKR composite scaffolds with different levels of AKR [124]. These scaffolds were used to induce the active expression of angiogenic and osteogenic genes in iPSCs. The composite scaffold with 10% AKR content was used to repair the skull defect in rats. The degradation of the scaffold maintained a physiological pH value in the local microenvironment and achieved optimal bone repair effect in 8 weeks.
CNTs are lightweight nanomaterials with a hexagonal structure. It has many unique mechanical, electrical, and chemical properties. In a study, nanocomposites of carboxylated multi-walled CNTs (MWCNTs) and PLGA were prepared [125]. This material has excellent histocompatibility and osteogenesis promotion effect. Molecular biology studies had shown that this composite material would affect the phosphorylation of extracellular signal-regulated kinase (ERK1/2), thereby affecting the DNA synthesis, proliferation, and differentiation of osteoblasts.
Some researchers believe that although the systemic toxic and side effects of the PLGA decomposition products are slight, the local acidic microenvironment after PLGA scaffold hydrolysis is negative to bone tissue regeneration. In response to this drawback, PLGA/magnesium hydroxide (PLGA/MgOH) scaffolds were prepared [126]. By neutralizing the acidic decomposition products with MgOH, the scaffold reduced the lysis of interleukin-1 converting enzyme-mediated death of BM-MSCs and promoted the differentiation effect of cells. This effect makes the PLGA-based artificial bone scaffold closer to clinical application.
Scaffolds prepared by compounding various inorganic components with PLGA have excellent mechanical properties, reasonable degradation time, and osteoconductivity. Furthermore, the combined use of stem cells, growth factors, and drugs with scaffolds had achieved excellent bone regeneration effect in different bone defect models. For the development of bone tissue engineering, the exploration of suitable inorganic materials and organic materials is the focus of this field. The ingenious chemical connection between them instead of pure mixing may have a surprising repair effect.
4. Conclusion and perspectives
In the past few decades, significant progress has been achieved in the development of artificial bone-substitute materials. Many artificial bone-substitute materials have entered clinical trials. Clinical trials are an essential stage for synthetic bone-substitute materials from the laboratory to the clinical application. The representative clinical trial projects of artificial bone-substitute materials are summarized in Table 3.
Table 3.
Representative clinical trial projects of artificial bone-substitute materials.
| ClinicalTrials.gov identifier | Material | Start year | Condition or disease | Number of patient | Country/Region | Status |
|---|---|---|---|---|---|---|
| NCT03608280 | 3D-printed titanium implant | 2020 | Bone defect in the orbit | 92 | France | Not yet recruiting |
| NCT03302429 | BCP | 2020 | Alveolar ridge abnormality | 18 | Egypt | Not yet recruiting |
| NCT04449211 | 3D-printed titanium implant | 2020 | Long bone defect | 10 | Vietnam | Not yet recruiting |
| NCT04069923 | Mg-based bone void filler | 2020 | Bone voids or defects | 6 | United States | Recruiting |
| NCT03804086 | HA | 2019 | Periodontitis | 28 | Egypt | Not yet recruiting |
| NCT03941028 | 3D-printed titanium implant | 2019 | Large segmental bone defect | 20 | China | Recruiting |
| NCT04480073 | Titanium mesh | 2018 | Bone resorption | 24 | Italy | Recruiting |
| NCT03588507 | HA | 2018 | Intrabony periodontal defect | 12 | Egypt | Unknown |
| NCT02803177 | TCP | 2016 | Humerus fracture displaced proximal | 55 | Germany | Completed |
| NCT02575352 | CP | 2015 | Bone defect | 500 | Multiple countries | Recruiting |
| NCT03185286 | 3D-printed titanium implant | 2015 | Ankle bone defects | 40 | China | Unknown |
| NCT02795546 | TCP | 2015 | Intrabony periodontal defect | 32 | – | Completed |
| NCT02456194 | CaSO4 | 2015 | Tibial fracture,intra-articular fracture, fracture fixation | 30 | Canada | Active, not recruiting |
| NCT02982681 | BG | 2015 | Bone resorption | 10 | – | Completed |
| NCT02748343 | TCP scaffold | 2014 | Bone defect, bone nonunion | 20 | China | Recruiting |
| NCT02000297 | CP/CaSO4 | 2013 | Knee osteoarthritis | 60 | Korea | Unknown |
| NCT02474498 | TCP/HA | 2013 | Periodontal disease | 41 | Brazil | Completed |
| NCT02015390 | Titanium cage | 2013 | Segmental long bone defect | 30 | United States | Unknown |
| NCT01899807 | Titanium/CP | 2013 | Defect of skull ossification | 11 | Sweden | Unknown |
| NCT01725698 | HA-CaSO4 | 2012 | Bone defect | 5 | Indonesia | Unknown |
| NCT01159899 | HA | 2010 | Osteonecrosis | 50 | France | Unknown |
| NCT02218515 | Enamel matrix derivatives | 2010 | Intrabony periodontal defect | 30 | Turkey | Completed |
| NCT00841152 | BG/TCP | 2009 | Bone neoplasm | 120 | Finland | Unknown |
| NCT00496847 | TCP | 2007 | Intrabony periodontal defect | 60 | India | Completed |
| NCT02827006 | CP | 2007 | Dorsal malunion of the distal radius | 20 | Norway | Completed |
| NCT01304121 | Bioactive glass | 2006 | Bone tumor | 48 | Finland | Completed |
| NCT00206791 | CP | 2006 | Bone defect | 12 | France | Unknown |
| NCT00835432 | BCP | 2005 | Alveolar bone defect | 40 | Germany | Completed |
PLGA-based artificial bone-substitute materials have obtained appealing results in bone repair, owing to their suitable properties, such as biocompatibility, degradability, processability, mechanical properties, and the ability to promote bone regeneration. First of all, PLGA has excellent processability, facilitating the fabrication of PLGA scaffolds with different pore sizes. These scaffolds can be used in combination to repair various tissues. The application of 3D printing technology has refined the processing of PLGA scaffolds. By changing the parameters of 3D printing, various PLGA scaffolds could be fabricated with different morphologies and properties. Researchers also explored printing methods suitable for different raw materials by comparing ST and FT 3D printing methods. Another fascinating property of PLGA is well biocompatibility, a fundamental requirement for the proliferation of loaded stem cells in bone defect treatment. In addition, the proliferation ability of stem cells is strictly related to the properties of PLGA. For instance, increased porosity of the PLGA scaffold may achieve good proliferation activity of BM-MSCs. The use of the unidirectional cooling technology to control the direction of the pore in the scaffold is also beneficial to the proliferation of cells attached to it. Besides, the decoration of artificial extracellular matrix on the surface of the PLGA scaffolds also achieved benefits to increase the activity of stem cells.
More recently, a variety of PLGA/inorganic composite materials have shown encouraging bone repair effects. Compared with the PLGA scaffold, composite scaffold consisting of PLGA and natural bone component HA has better mechanical strength and bone conductivity. By adjusting the feed ratio of PLGA and HA and introducing the PBLG, researchers obtained a composite scaffold with the balanced mechanical and biological properties. PLGA is also combining with other inorganic materials, such as CP and BG, thereby promoting the mechanical properties and increasing the bone tissue binding capacity to support local bone repair. AKR, CNTs, and MgOH are also combined with PLGA to improve bone repair effects by locally releasing silicon ions, promoting the osteogenic differentiation process and adjusting the local acidic environment. On this basis, the PLGA composite scaffold implanted with stem cells and drugs obtains an enhancing bone repair effect.
Considering the advantages of PLGA artificial bone-substitute materials mentioned above, they have great potential in clinical application. However, several obstacles require to be conquered in future studies. In terms of scientific research, firstly, the process of bone formation is affected by the balance of osteoblast and osteoclast activity. But existing studies only focus on the businesses of osteoblasts, lacking studies focuses on the activities of osteoclasts. Additionally, the degradation of PLGA will cause a local acidic microenvironment, which is harmful to cell proliferation and bone repair. Since PLGA with different ratios of lactic acid and glycolic acid have different degradation rates, the faster the degradation rate, the more significant the impact. It is possible to solve this problem by introducing alkaline components into the composite materials. For example, add Mg or Mg alloy to the scaffolds, or mix alkaline inorganic salt components in the materials. It will further improve the bone repairability of the scaffold. Moreover, some of the bioactive factors currently used in combination with scaffolds are derived from animals and have limited efficacy in the clinic. Hopefully, the problem may be solved through the combined application of autologous PRP transfusion and autologous stem cell transfusion technology.
In terms of clinical translation, firstly, there have been several studies on the influence of PLGA-based scaffolds porosity, mechanical properties, and other factors on bone defect repair, but no consensus concerning the optimal values, which is very critical for future clinical applications. Besides, the conditions of clinical bone defects are diverse. At present, there is no studies on the indications and contraindications of PLGA-based scaffolds applied in the treatment of bone defects. This is a critical step in the transition of PLGA-based scaffolds from laboratory to the clinic. In addition, personalized therapy becomes a research hotspot. The areas and shapes of bone defects in each patient are different in clinical practice, but the design of current PLGA-based artificial bone materials hardly meets the needs of each patient. Fortunately, relying on the computer design and the advanced 3D printing technology, scaffolds adapted to different positions, shapes, and mechanical requirements may be expected to solve this problem. Finally, the current production technology of PLGA-based artificial bone materials is still facing many limitations, such as small production scale and low efficiency. These problems limited the clinical application of PLGA-based artificial bone materials and increasing the economic burden on patients. Therefore, it is necessary to simplify the manufacturing process and improve the yield and quality of scaffolds.
In summary, more studies are needed for the development of PLGA-based artificial bone-substitute materials to become ideal artificial bone sources for clinical application.
CRediT authorship contribution statement
Duoyi Zhao: Methodology, Validation, Formal analysis, Resources, Writing - original draft, Visualization. Tongtong Zhu: Validation, Resources, Writing - review & editing. Jie Li: Writing - review & editing. Liguo Cui: Conceptualization, Writing - review & editing, Supervision. Zhiyu Zhang: Writing - review & editing, Supervision. Xiuli Zhuang: Writing - review & editing, Supervision. Jianxun Ding: Conceptualization, Writing - review & editing, Supervision, Funding acquisition.
Declaration of competing interest
The authors declare no competing financial interests.
Acknowledgments
This study was financially supported by the National Natural Science Foundation of China (Grant Nos. 51973216, 51873207, 51803006, and 51833010), the Science and Technology Development Program of Jilin Province (Grant No. 20200404182YY), the Youth Innovation Promotion Association of the Chinese Academy of Sciences (Grant No. 2019005), and the State Key Laboratory of Advanced Technology for Materials Synthesis and Processing (Wuhan University of Technology) (Grant No. 2020-KF-5).
Footnotes
Peer review under responsibility of KeAi Communications Co., Ltd.
Contributor Information
Liguo Cui, Email: cuilg@ciac.ac.cn.
Jianxun Ding, Email: jxding@ciac.ac.cn.
References
- 1.Wascher D.C., Bulthuis L. Extremity trauma: Field management of sports injuries. Curr. Rev. Musculoskelet. Med. 2014;7(4):387–393. doi: 10.1007/s12178-014-9242-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Saridis A., Panagiotopoulos E., Tyllianakis M., Matzaroglou C., Vandoros N., Lambiris E. The use of the Ilizarov method as a salvage procedure in infected nonunion of the distal femur with bone loss. J. Bone Joint Surg. Br. 2006;88(2):232–237. doi: 10.1302/0301-620X.88B2.16976. [DOI] [PubMed] [Google Scholar]
- 3.Assouline-Dayan Y., Chang C., Greenspan A., Shoenfeld Y., Gershwin M.E. Pathogenesis and natural history of osteonecrosis. Semin. Arthritis Rheum. 2002;32(2):94–124. [PubMed] [Google Scholar]
- 4.Sözen T., Özışık L., Başaran N.Ç. An overview and management of osteoporosis. Eur. J. Rheumatol. 2017;4(1):46–56. doi: 10.5152/eurjrheum.2016.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lems W.F., Raterman H.G. Critical issues and current challenges in osteoporosis and fracture prevention. An overview of unmet needs. Ther. Adv. Musculoskelet. Dis. 2017;9(12):299–316. doi: 10.1177/1759720X17732562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Guise T.A. Bone loss and fracture risk associated with cancer therapy. Oncologist. 2006;11(10):1121–1131. doi: 10.1634/theoncologist.11-10-1121. [DOI] [PubMed] [Google Scholar]
- 7.Ibrahim T., Mercatali L., Amadori D. Bone and cancer: The osteoncology. Clin. Cases Miner. Bone Metab. 2013;10(2):121–123. [PMC free article] [PubMed] [Google Scholar]
- 8.Alazraki N., Moitoza J., Heaphy J., Taylor A. The effect of iatrogenic trauma on the bone scintigram: An animal study: Concise communication. J. Nucl. Med. 1984;25(9):978–981. [PubMed] [Google Scholar]
- 9.Ilizarov G.A., Deviatov A.A., Trokhova V.G. Surgical lengthening of the shortened lower extremities, Vestn. Khir. Im. I. I. Grek. 1972;108(2):100–103. [PubMed] [Google Scholar]
- 10.Nandi S.K., Roy S., Mukherjee P., Kundu B., De D.K., Basu D. Orthopaedic applications of bone graft & graft substitutes: A review. Indian J. Med. Res. 2010;132(1):15–30. [PubMed] [Google Scholar]
- 11.Ilizarov G.A. The tension stress effect on the genesis and growth of tissues. 2. The influence of the rate and frequency of distraction. Clin. Orthop. Relat. Res. 1989;239:263–285. [PubMed] [Google Scholar]
- 12.Ilizarov G.A. Clinical-application of the tension-stress effect for limb lengthening. Clin. Orthop. Relat. Res. 1990;250:8–26. [PubMed] [Google Scholar]
- 13.Oryan A., Alidadi S., Moshiri A., Maffulli N. Bone regenerative medicine: Classic options, novel strategies, and future directions. J. Orthop. Surg. Res. 2014;9(1):18. doi: 10.1186/1749-799X-9-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Schwartz A.M., Schenker M.L., Ahn J., Willett N. Building better bone: The weaving of biologic and engineering strategies for managing bone loss. J. Orthop. Res. 2017;35(9):1855–1864. doi: 10.1002/jor.23592. [DOI] [PubMed] [Google Scholar]
- 15.Allison D.C., McIntyre J.A., Ferro A., Brien E., Menendez L. Bone grafting alternatives for cavitary defects in children. Curr. Orthop. Pract. 2013;24(3):267–279. [Google Scholar]
- 16.Ehrler D.M., Vaccaro A.R. The use of allograft bone in lumbar spine surgery. Clin. Orthop. Relat. Res. 2000;371:38–45. doi: 10.1097/00003086-200002000-00005. [DOI] [PubMed] [Google Scholar]
- 17.Brydone A.S., Meek D., Maclaine S. Bone grafting, orthopaedic biomaterials, and the clinical need for bone engineering. Proc. Inst. Mech. Eng. H. 2010;224(12):1329–1343. doi: 10.1243/09544119JEIM770. [DOI] [PubMed] [Google Scholar]
- 18.Dimitriou R., Jones E., McGonagle D., Giannoudis P.V. Bone regeneration: Current concepts and future directions. BMC Med. 2011;9(1):66. doi: 10.1186/1741-7015-9-66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Babaie E., Bhaduri S.B. Fabrication aspects of porous biomaterials in orthopedic applications: A review. ACS Biomater. Sci. Eng. 2018;4(1):1–39. doi: 10.1021/acsbiomaterials.7b00615. [DOI] [PubMed] [Google Scholar]
- 20.Chen X., Fan H., Deng X., Wu L., Yi T., Gu L., Zhou C., Fan Y., Zhang X. Scaffold structural microenvironmental cues to guide tissue regeneration in bone tissue applications. Nanomaterials. 2018;8(11):960. doi: 10.3390/nano8110960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Bružauskaitė I., Bironaitė D., Bagdonas E., Bernotienė E. Scaffolds and cells for tissue regeneration: Different scaffold pore sizes-different cell effects. Cytotechnology. 2016;68(3):355–369. doi: 10.1007/s10616-015-9895-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Weisgerber D.W., Erning K., Flanagan C.L., Hollister S.J., Harley B.A.C. Evaluation of multi-scale mineralized collagen–polycaprolactone composites for bone tissue engineering. J. Mech. Behav. Biomed. Mater. 2016;61:318–327. doi: 10.1016/j.jmbbm.2016.03.032. [DOI] [PubMed] [Google Scholar]
- 23.Zhang J., Wang L., Zhang W., Zhang M., Luo Z.P. Synchronization of calcium sulphate cement degradation and new bone formation is improved by external mechanical regulation. J. Orthop. Res. 2015;33(5):685–691. doi: 10.1002/jor.22839. [DOI] [PubMed] [Google Scholar]
- 24.Liao C.Z., Li K., Wong H.M., Tong W.Y., Yeung K.W.K., Tjong S.C. Novel polypropylene biocomposites reinforced with carbon nanotubes and hydroxyapatite nanorods for bone replacements. Mater. Sci. Eng. C Mater. Biol. Appl. 2013;33(3):1380–1388. doi: 10.1016/j.msec.2012.12.039. [DOI] [PubMed] [Google Scholar]
- 25.Pascu E.I., Stokes J., McGuinness G.B. Electrospun composites of PHBV, silk fibroin and nano-hydroxyapatite for bone tissue engineering. Mater. Sci. Eng. C Mater. Biol. Appl. 2013;33(8):4905–4916. doi: 10.1016/j.msec.2013.08.012. [DOI] [PubMed] [Google Scholar]
- 26.Ghanaati S., Barbeck M., Orth C., Willershausen I., Thimm B.W., Hoffmann C., Rasic A., Sader R.A., Unger R.E., Peters F., Kirkpatrick C.J. Influence of β-tricalcium phosphate granule size and morphology on tissue reaction in vivo. Acta Biomater. 2010;6(12):4476–4487. doi: 10.1016/j.actbio.2010.07.006. [DOI] [PubMed] [Google Scholar]
- 27.Wang L., Fan H.B., Zhang Z.Y., Lou A.J., Pei G.X., Jiang S., Mu T.W., Qin J.J., Chen S.Y., Jin D. Osteogenesis and angiogenesis of tissue-engineered bone constructed by prevascularized β-tricalcium phosphate scaffold and mesenchymal stem cells. Biomaterials. 2010;31(36):9452–9461. doi: 10.1016/j.biomaterials.2010.08.036. [DOI] [PubMed] [Google Scholar]
- 28.Lobo S.E., Arinzeh T.L. Biphasic calcium phosphate ceramics for bone regeneration and tissue engineering applications. Materials. 2010;3(2):815–826. [Google Scholar]
- 29.Nihouannen D.L., Duval L., Lecomte A., Julien M., Guicheux J., Daculsi G., Layrolle P. Interactions of total bone marrow cells with increasing quantities of macroporous calcium phosphate ceramic granules. J. Mater. Sci. Mater. Med. 2007;18(10):1983–1990. doi: 10.1007/s10856-007-3098-2. [DOI] [PubMed] [Google Scholar]
- 30.Fan J.P., Kalia P., Silvio L.D., Huang J. In vitro response of human osteoblasts to multi-step sol−gel derived bioactive glass nanoparticles for bone tissue engineering. Mater. Sci. Eng. C Mater. Biol. App. 2014;36:206–214. doi: 10.1016/j.msec.2013.12.009. [DOI] [PubMed] [Google Scholar]
- 31.Liu Y., Dang Z., Wang Y., Huang J., Li H. Hydroxyapatite/graphene-nanosheet composite coatings deposited by vacuum cold spraying for biomedical applications: Inherited nanostructures and enhanced properties. Carbon. 2014;67:250–259. [Google Scholar]
- 32.Chowdhury S.M., Lalwani G., Zhang K.V., Yang J.Y., Neville K., Sitharaman B. Cell specific cytotoxicity and uptake of graphene nanoribbons. Biomaterials. 2013;34(1):283–293. doi: 10.1016/j.biomaterials.2012.09.057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Pilia M., Guda T., Appleford M. Development of composite scaffolds for load-bearing segmental bone defects. BioMed Res. Int. 2013 doi: 10.1155/2013/458253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ostermann P.A.W., Haase N., Rübberdt A., Wich M., Ekkernkamp A. Management of a long segmental defect at the proximal meta-diaphyseal junction of the tibia using a cylindrical titanium mesh cage. J. Orthop. Trauma. 2002;16(8):597–601. doi: 10.1097/00005131-200209000-00010. [DOI] [PubMed] [Google Scholar]
- 35.Alvarez K., Hyun S.K., Nakano T., Umakoshi Y., Nakajima H. In vivo osteocompatibility of lotus-type porous nickel-free stainless steel in rats. Mater. Sci. Eng. C Mater. Biol. Appl. 2009;29(4):1182–1190. [Google Scholar]
- 36.Kapanen A., Ryhanen J., Danilov A., Tuukkanen J. Effect of nickel-titanium shape memory metal alloy on bone formation. Biomaterials. 2001;22(18):2475–2480. doi: 10.1016/s0142-9612(00)00435-x. [DOI] [PubMed] [Google Scholar]
- 37.Long M., Rack H.J. Titanium alloys in total joint replacement—A materials science perspective. Biomaterials. 1998;19(18):1621–1639. doi: 10.1016/s0142-9612(97)00146-4. [DOI] [PubMed] [Google Scholar]
- 38.Alvarez K., Nakajima H. Metallic scaffolds for bone regeneration. Materials. 2009;2(3):790–832. [Google Scholar]
- 39.Goodman S.B., Fornasier V.L., Lee J., Kei J. The effects of bulk versus particulate titanium and cobalt chrome alloy implanted into the rabbit tibia. J. Biomed. Mater. Res. 1990;24(11):1539–1549. doi: 10.1002/jbm.820241109. [DOI] [PubMed] [Google Scholar]
- 40.Avval P.T., Klika V., Bougherara H. Predicting bone remodeling in response to total hip arthroplasty: Computational study using mechanobiochemical model. J. Biomech. Eng. 2014;136(5) doi: 10.1115/1.4026642. [DOI] [PubMed] [Google Scholar]
- 41.Matta C., Szucs-Somogyi C., Kon E., Robinson D., Neufeld T., Altschuler N., Berta A., Hangody L., Vereb Z., Zakany R. Osteogenic differentiation of human bone marrow-derived mesenchymal stem cells is enhanced by an aragonite scaffold. Differentiation. 2019;107:24–34. doi: 10.1016/j.diff.2019.05.002. [DOI] [PubMed] [Google Scholar]
- 42.Zhang H., Zhou Y., Yu N., Ma H., Wang K., Liu J., Zhang W., Cai Z., He Y. Construction of vascularized tissue-engineered bone with polylysine-modified coral hydroxyapatite and a double cell-sheet complex to repair a large radius bone defect in rabbits. Acta Biomater. 2019;91:82–98. doi: 10.1016/j.actbio.2019.04.024. [DOI] [PubMed] [Google Scholar]
- 43.Berruto M., Delcogliano M., de Caro F., Carimati G., Uboldi F., Ferrua P., Ziveri G., De Biase C.F. Treatment of large knee osteochondral lesions with a biomimetic scaffold: Results of a multicenter study of 49 patients at 2-year follow-up. Am. J. Sports Med. 2014;42(7):1607–1617. doi: 10.1177/0363546514530292. [DOI] [PubMed] [Google Scholar]
- 44.Pei M., He F., Boyce B.M., Kish V.L. Repair of full-thickness femoral condyle cartilage defects using allogeneic synovial cell-engineered tissue constructs. Osteoarthritis Cartilage. 2009;17(6):714–722. doi: 10.1016/j.joca.2008.11.017. [DOI] [PubMed] [Google Scholar]
- 45.Tsai K.S., Kao S.Y., Wang C.Y., Wang Y.J., Wang J.P., Hung S.C. Type I collagen promotes proliferation and osteogenesis of human mesenchymal stem cells via activation of ERK and Akt pathways. J. Biomed. Mater. Res. 2010;94(3):673–682. doi: 10.1002/jbm.a.32693. [DOI] [PubMed] [Google Scholar]
- 46.Kruger T.E., Miller A.H., Wang J. Collagen scaffolds in bone sialoprotein-mediated bone regeneration. Sci. World J. 2013 doi: 10.1155/2013/812718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Duarte A.R.C., Mano J.F., Reis R.L. Novel 3D scaffolds of chitosan-PLLA blends for tissue engineering applications: Preparation and characterization. J. Supercrit. Fluids. 2010;54(3):282–289. [Google Scholar]
- 48.Kavya K.C., Jayakumar R., Nair S., Chennazhi K.P. Fabrication and characterization of chitosan/gelatin/nSiO2 composite scaffold for bone tissue engineering. Int. J. Biol. Macromol. 2013;59:255–263. doi: 10.1016/j.ijbiomac.2013.04.023. [DOI] [PubMed] [Google Scholar]
- 49.Liu X., Cheng C., Peng X., Xiao H., Guo C.R., Wang X., Li L., Yu X. A promising material for bone repair: PMMA bone cement modified by dopamine-coated strontium-doped calcium polyphosphate particles. R. Soc. Open Sci. 2019;6(10) doi: 10.1098/rsos.191028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Russo T., De Santis R., Gloria A., Barbaro K., Altigeri A., Fadeeva I.V., Rau J.V. Modification of PMMA cements for cranioplasty with bioactive glass and copper doped tricalcium phosphate particles. Polymers. 2020;12(1):37. doi: 10.3390/polym12010037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Rodrigues A.I., Gomes M.E., Leonor I.B., Reis R.L. Bioactive starch-based scaffolds and human adipose stem cells are a good combination for bone tissue engineering. Acta Biomater. 2012;8(10):3765–3776. doi: 10.1016/j.actbio.2012.05.025. [DOI] [PubMed] [Google Scholar]
- 52.Correia S.I., Pereira H., Silva-Correia J., Van Dijk C.N., Espregueira-Mendes J., Oliveira J.M., Reis R.L. Current concepts: Tissue engineering and regenerative medicine applications in the ankle joint. J. R. Soc. Interface. 2014;11(92):20130784. doi: 10.1098/rsif.2013.0784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Xu H., Han D., Dong J.S., Shen G.X., Chai G., Yu Z.Y., Lang W.J., Ai S.T. Rapid prototyped PGA/PLA scaffolds in the reconstruction of mandibular condyle bone defects. Int. J. Med. Robot. 2010;6(1):66–72. doi: 10.1002/rcs.290. [DOI] [PubMed] [Google Scholar]
- 54.Rainer A., Spadaccio C., Sedati P., De Marco F., Carotti S., Lusini M., Vadala G., Di Martino A., Morini S., Chello M., Covino E., Denaro V., Trombetta M. Electrospun hydroxyapatite-functionalized PLLA scaffold: Potential applications in sternal bone healing. Ann. Biomed. Eng. 2011;39(7):1882–1890. doi: 10.1007/s10439-011-0289-2. [DOI] [PubMed] [Google Scholar]
- 55.Jaiswal A.K., Kadam S.S., Soni V.P., Bellare J.R. Improved functionalization of electrospun PLLA/gelatin scaffold by alternate soaking method for bone tissue engineering. Appl. Surf. Sci. 2013;268:477–488. [Google Scholar]
- 56.Zhang Y., Liu X., Zeng L., Zhang J., Zuo J., Zou J., Ding J., Chen X. Polymer fiber scaffolds for bone and cartilage tissue engineering. Adv. Funct. Mater. 2019;29(36) [Google Scholar]
- 57.Zhu T., Cui Y., Zhang M., Zhao D., Liu G., Ding J. Engineered three-dimensional scaffolds for enhanced bone regeneration in osteonecrosis. Bioact. Mater. 2020;5(3):584–601. doi: 10.1016/j.bioactmat.2020.04.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Gupta A.P., Kumar V. New emerging trends in synthetic biodegradable polymers – Polylactide: A critique. Eur. Polym. J. 2007;43(10):4053–4074. [Google Scholar]
- 59.Mir M., Ahmed N., Rehman A.U. Recent applications of PLGA based nanostructures in drug delivery. Colloids Surf. B Biointerfaces. 2017;159:217–231. doi: 10.1016/j.colsurfb.2017.07.038. [DOI] [PubMed] [Google Scholar]
- 60.Ajioka M., Suizu H., Higuchi C., Kashima T. Aliphatic polyesters and their copolymers synthesized through direct condensation polymerization. Polym. Degrad. Stabil. 1998;59(1–3):137–143. [Google Scholar]
- 61.Zhou S.B., Deng X.M., Li X.H., Jia W.X., Liu L. Synthesis and characterization of biodegradable low molecular weight aliphatic polyesters and their use in protein-delivery systems. J. Appl. Polym. Sci. 2004;91(3):1848–1856. [Google Scholar]
- 62.Duval C., Nouvel C., Six J. Is bismuth subsalicylate an effective nontoxic catalyst for PLGA synthesis? J. Polym. Sci. Polym. Chem. 2014;52(8):1130–1138. [Google Scholar]
- 63.Varma I.K., Albertsson A.C., Rajkhowa R., Srivastava R.K. Enzyme catalyzed synthesis of polyesters. Prog. Polym. Sci. 2005;30(10):949–981. [Google Scholar]
- 64.Holy C.E., Dang S.M., Davies J.E., Shoichet M.S. In vitro degradation of a novel poly(lactide-co-glycolide) 75/25 foam. Biomaterials. 1999;20(13):1177–1185. doi: 10.1016/s0142-9612(98)00256-7. [DOI] [PubMed] [Google Scholar]
- 65.Yoon J.J., Park T.G. Degradation behaviors of biodegradable macroporous scaffolds prepared by gas foaming of effervescent salts. J. Biomed. Mater. Res. 2001;55(3):401–408. doi: 10.1002/1097-4636(20010605)55:3<401::aid-jbm1029>3.0.co;2-h. [DOI] [PubMed] [Google Scholar]
- 66.Gentile P., Chiono V., Carmagnola I., Hatton P.V. An overview of poly(lactic-co-glycolic) acid (PLGA)-based biomaterials for bone tissue engineering. Int. J. Mol. Sci. 2014;15(3):3640–3659. doi: 10.3390/ijms15033640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Wu L., Ding J. In vitro degradation of three-dimensional porous poly(D,L-lactide-co-glycolide) scaffolds for tissue engineering. Biomaterials. 2004;25(27):5821–5830. doi: 10.1016/j.biomaterials.2004.01.038. [DOI] [PubMed] [Google Scholar]
- 68.Cai Q., Shi G., Bei J., Wang S. Enzymatic degradation behavior and mechanism of poly(lactide-co-glycolide) foams by trypsin. Biomaterials. 2003;24(4):629–638. doi: 10.1016/s0142-9612(02)00377-0. [DOI] [PubMed] [Google Scholar]
- 69.Danhier F., Ansorena E., Silva J.M., Coco R., Le Breton A., Préat V. PLGA-based nanoparticles: An overview of biomedical applications. J. Contr. Release. 2012;161(2):505–522. doi: 10.1016/j.jconrel.2012.01.043. [DOI] [PubMed] [Google Scholar]
- 70.Kumari A., Yadav S.K., C Yadav S. Biodegradable polymeric nanoparticles based drug delivery systems. Colloids Surf. B Biointerfaces. 2010;75(1):1–18. doi: 10.1016/j.colsurfb.2009.09.001. [DOI] [PubMed] [Google Scholar]
- 71.Liang X., Qi Y., Pan Z., He Y., Liu X., Cui S., Ding J. Design and preparation of quasi-spherical salt particles as water-soluble porogens to fabricate hydrophobic porous scaffolds for tissue engineering and tissue regeneration. Mater. Chem. Front. 2018;2(8):1539–1553. [Google Scholar]
- 72.Lee H., Yoo J.J., Kang H.W., Cho D.W. Investigation of thermal degradation with extrusion-based dispensing modules for 3D bioprinting technology. Biofabrication. 2016;8(1) doi: 10.1088/1758-5090/8/1/015011. [DOI] [PubMed] [Google Scholar]
- 73.Yu Y., Sun B., Yi C., Mo X. Stem cell homing-based tissue engineering using bioactive materials. Front. Mater. Sci. 2017;11(2):93–105. [Google Scholar]
- 74.Han Y., Li X., Zhang Y., Han Y., Chang F., Ding J. Mesenchymal stem cells for regenerative medicine. Cells. 2019;8(8):886. doi: 10.3390/cells8080886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Pan Z., Ding J. Poly(lactide-co-glycolide) porous scaffolds for tissue engineering and regenerative medicine. Interface Focus. 2012;2(3):366–377. doi: 10.1098/rsfs.2011.0123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Duan P., Pan Z., Cao L., He Y., Wang H., Qu Z., Dong J., Ding J. The effects of pore size in bilayered poly(lactide-co-glycolide) scaffolds on restoring osteochondral defects in rabbits. J. Biomed. Mater. Res. 2014;102(1):180–192. doi: 10.1002/jbm.a.34683. [DOI] [PubMed] [Google Scholar]
- 77.Pan Z., Duan P., Liu X., Wang H., Cao L., He Y., Dong J., Ding J. Effect of porosities of bilayered porous scaffolds on spontaneous osteochondral repair in cartilage tissue engineering. Regen. Biomater. 2015;2(1):9–19. doi: 10.1093/rb/rbv001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Duan P., Pan Z., Cao L., Gao J., Yao H., Liu X., Guo R., Liang X., Dong J., Ding J. Restoration of osteochondral defects by implanting bilayered poly(lactide-co-glycolide) porous scaffolds in rabbit joints for 12 and 24 weeks. J. Orthop. Translat. 2019;19:68–80. doi: 10.1016/j.jot.2019.04.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Dai Y., Shen T., Ma L., Wang D., Gao C. Regeneration of osteochondral defects in vivo by a cell-free cylindrical poly(lactide-co-glycolide) scaffold with a radially oriented microstructure. J. Tissue Eng. Regen. Med. 2018;12(3):e1647–e1661. doi: 10.1002/term.2592. [DOI] [PubMed] [Google Scholar]
- 80.Liu C.G., Zeng Y.T., Kankala R.K., Zhang S.S., Chen A.Z., Wang S.B. Characterization and preliminary biological evaluation of 3D-printed porous scaffolds for engineering bone tissues. Materials. 2018;11(10):1832. doi: 10.3390/ma11101832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Cui L., Zhang J., Zou J., Yang X., Guo H., Tian H., Zhang P., Wang Y., Zhang N., Zhuang X., Li Z., Ding J., Chen X. Electroactive composite scaffold with locally expressed osteoinductive factor for synergistic bone repair upon electrical stimulation. Biomaterials. 2020;230:119617. doi: 10.1016/j.biomaterials.2019.119617. [DOI] [PubMed] [Google Scholar]
- 82.Chen D., Zhang C., Huo H., Ji C., Sun M., Nie L. Injectable temperature-sensitive hydrogel with VEGF loaded microspheres for vascularization and bone regeneration of femoral head necrosis. Mater. Lett. 2018;229:138–141. [Google Scholar]
- 83.Zhang Y.T., Pan Z., Xia L.H., Liu X.N., Guo X.L., He Y., Zhou J., Qu Z.H., Mei G., Jin D., Ding J.D. Bilayered poly(lactide-co-glycolide) scaffold with platelet-rich plasma and mesenchymal stem cells improves restoration of osteochondral defects. J. Biomater. Tissue Eng. 2015;5(10):757–765. [Google Scholar]
- 84.Beane O.S., Fonseca V.C., Cooper L.L., Koren G., Darling E.M. Impact of aging on the regenerative properties of bone marrow-, muscle-, and adipose-derived mesenchymal stem/stromal cells. PloS One. 2014;9(12) doi: 10.1371/journal.pone.0115963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Chen H.T., Lee M.J., Chen C.H., Chuang S.C., Chang L.F., Ho M.L., Hung S.H., Fu Y.C., Wang Y.H., Wang H.I. Proliferation and differentiation potential of human adipose-derived mesenchymal stem cells isolated from elderly patients with osteoporotic fractures. J. Cell Mol. Med. 2012;16(3):582–592. doi: 10.1111/j.1582-4934.2011.01335.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Wang Y.H., Wu J.Y., Kong S.C., Chiang M.H., Ho M.L., Yeh M.L., Chen C.H. Low power laser irradiation and human adipose-derived stem cell treatments promote bone regeneration in critical-sized calvarial defects in rats. PloS One. 2018;13(4) doi: 10.1371/journal.pone.0195337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Wojak-Cwik I., Hintze V., Schnabelrauch M., Moeller S., Dobrzynski P., Pamula E., Scharnweber D. Poly(L-lactide-co-glycolide) scaffolds coated with collagen and glycosaminoglycans: Impact on proliferation and osteogenic differentiation of human mesenchymal stem cells. J. Biomed. Mater. Res. 2013;101(11):3109–3122. doi: 10.1002/jbm.a.34620. [DOI] [PubMed] [Google Scholar]
- 88.Wojak-Cwik I.M., Rumian L., Krok-Borkowicz M., Hess R., Bernhardt R., Dobrzynski P., Moller S., Schnabelrauch M., Hintze V., Scharnweber D., Pamula E. Synergistic effect of bimodal pore distribution and artificial extracellular matrices in polymeric scaffolds on osteogenic differentiation of human mesenchymal stem cells. Mater. Sci. Eng. C Mater. Biol. Appl. 2019;97:12–22. doi: 10.1016/j.msec.2018.12.012. [DOI] [PubMed] [Google Scholar]
- 89.Rentsch B., Bernhardt R., Scharnweber D., Schneiders W., Rammelt S., Rentsch C. Embroidered and surface coated poly(caprolactone-co-lactide) scaffolds: A potential graft for bone tissue engineering. Biomatter. 2012;2(3):158–165. doi: 10.4161/biom.21931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Liu G., Lu T., Ji X., Jia W., Li Y., Zhao Y., Luo Y. Preparation and osteogenic induction activity of CBD-BMP2-MP/PLGA 3D printed composite scaffolds. Chem. Res. Chin. Univ. 2019;40(7):1552–1560. [Google Scholar]
- 91.Suchanek W., Yoshimura M. Processing and properties of hydroxyapatite-based biomaterials for use as hard tissue replacement implants. J. Mater. Res. 1998;13(1):94–117. [Google Scholar]
- 92.Zhao X., Han Y., Zhu T., Feng N., Sun Y., Song Z., Li S., Liu J., Ding J. Electrospun polylactide-nano-hydroxyapatite vancomycin composite scaffolds for advanced osteomyelitis therapy. J. Biomed. Nanotechnol. 2019;15(6):1213–1222. doi: 10.1166/jbn.2019.2773. [DOI] [PubMed] [Google Scholar]
- 93.Fisher P.D., Venugopal G., Milbrandt T.A., Hilt J.Z., Puleo D.A. Hydroxyapatite-reinforced in situ forming PLGA systems for intraosseous injection. J. Biomed. Mater. Res. 2015;103(7):2365–2373. doi: 10.1002/jbm.a.35375. [DOI] [PubMed] [Google Scholar]
- 94.Wang D.X., He Y., Bi L., Qu Z.H., Zou J.W., Pan Z., Fan J.J., Chen L., Dong X., Liu X.N., Pei G.X., Ding J.D. Enhancing the bioactivity of poly(lactic-co-glycolic acid) scaffold with a nano-hydroxyapatite coating for the treatment of segmental bone defect in a rabbit model. Int. J. Nanomed. 2013;8:1855–1865. doi: 10.2147/IJN.S43706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Takeoka Y., Hayashi M., Sugiyama N., Yoshizawa-Fujita M., Aizawa M., Rikukawa M. In situ preparation of poly(L-lactic acid-co-glycolic acid)/hydroxyapatite composites as artificial bone materials. Polym. J. 2015;47(2):164–170. [Google Scholar]
- 96.Park J.W., Hwang J.U., Back J.H., Jang S.W., Kim H.J., Kim P.S., Shin S., Kim T. High strength PLGA/hydroxyapatite composites with tunable surface structure using PLGA direct grafting method for orthopedic implants. Compos. B Eng. 2019;178 [Google Scholar]
- 97.Huang J., Xia X., Zou Q., Ma J., Jin S., Li J., Zuo Y., Li Y. The long-term behaviors and differences in bone reconstruction of three polymer-based scaffolds with different degradability. J. Mater. Chem. B. 2019;7(48):7690–7703. doi: 10.1039/c9tb02072a. [DOI] [PubMed] [Google Scholar]
- 98.Liang X., Duan P., Gao J., Guo R., Qu Z., Li X., He Y., Yao H., Ding J. Bilayered PLGA/PLGA-HAp composite scaffold for osteochondral tissue engineering and tissue regeneration. ACS Biomater. Sci. Eng. 2018;4(10):3506–3521. doi: 10.1021/acsbiomaterials.8b00552. [DOI] [PubMed] [Google Scholar]
- 99.Namini M.S., Bayat N., Tajerian R., Ebrahimi-Barough S., Azami M., Irani S., Jangjoo S., Shirian S., Ai J. A comparison study on the behavior of human endometrial stem cell-derived osteoblast cells on PLGA/HA nanocomposite scaffolds fabricated by electrospinning and freeze-drying methods. J. Orthop. Surg. Res. 2018;13:63. doi: 10.1186/s13018-018-0754-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Zheng S., Guan Y., Yu H., Huang G., Zheng C. Poly(L-lysine)-coated PLGA/poly(amino acid)-modified hydroxyapatite porous scaffolds as efficient tissue engineering scaffolds for cell adhesion, proliferation, and differentiation. New J. Chem. 2019;43(25):9989–10002. [Google Scholar]
- 101.Li L., Shi X., Wang Z., Guo M., Wang Y., Jiao Z., Zhang P. Porous scaffolds of poly(lactic-co-glycolic acid) and mesoporous hydroxyapatite surface modified by poly(γ-benzyl-L-glutamate) (PBLG) for in vivo bone repair. ACS Biomater. Sci. Eng. 2019;5(5):2466–2481. doi: 10.1021/acsbiomaterials.8b01614. [DOI] [PubMed] [Google Scholar]
- 102.Ignjatovic N., Uskokovic V., Ajdukovic Z., Uskokovic D. Multifunctional hydroxyapatite and poly(D,L-lactide-co-glycolide) nanoparticles for the local delivery of cholecalciferol. Mater. Sci. Eng. C Mater. Biol. Appl. 2013;33(2):943–950. doi: 10.1016/j.msec.2012.11.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Tai I.C., Fu Y.C., Wang C.K., Chang J.K., Ho M.L. Local delivery of controlled-release simvastatin/PLGA/HAp microspheres enhances bone repair. Int. J. Nanomed. 2013;8:3895–3904. doi: 10.2147/IJN.S48694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Wang C.K., Ho M.L., Wang G.J., Chang J.K., Chen C.H., Fu Y.C., Fu H.H. Controlled-release of rhBMP-2 carriers in the regeneration of osteonecrotic bone. Biomaterials. 2009;30(25):4178–4186. doi: 10.1016/j.biomaterials.2009.04.029. [DOI] [PubMed] [Google Scholar]
- 105.Haider A., Gupta K.C., Kang I.K. PLGA/nHA hybrid nanofiber scaffold as a nanocargo carrier of insulin for accelerating bone tissue regeneration. Nanoscale Res. Lett. 2014;9:314. doi: 10.1186/1556-276X-9-314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Wang T., Guo S., Zhang H. Synergistic effects of controlled-released BMP-2 and VEGF from nHAC/PLGAs scaffold on osteogenesis. BioMed Res. Int. 2018 doi: 10.1155/2018/3516463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Deng N., Sun J., Li Y., Chen L., Chen C., Wu Y., Wang Z., Li L. Experimental study of rhBMP-2 chitosan nano-sustained release carrier-loaded PLGA/nHA scaffolds to construct mandibular tissue-engineered bone. Arch. Oral Biol. 2019;102:16–25. doi: 10.1016/j.archoralbio.2019.03.023. [DOI] [PubMed] [Google Scholar]
- 108.Xu Q., Li Y., Zhu Y., Zhao K., Gu R., Zhu Q. Recombinant human BMP-7 grafted poly(lactide-co-glycolide)/hydroxyapatite scaffolds via polydopamine for enhanced calvarial repair. RSC Adv. 2018;8(48):27191–27200. doi: 10.1039/c8ra05606d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Zheng Z., Wang J.L., Mi L., Liu Y., Hou G.H., Chen D., Huang B.Y. Preparation of new tissue engineering bone-CPC/PLGA composite and its application in animal bone defects. J. Cent. South. Univ. 2010;17(2):202–210. [Google Scholar]
- 110.Ignjatovic N.L., Ajdukovic Z.R., Savic V.P., Uskokovic D.P. Size effect of calcium phosphate coated with poly(DL-lactide-co-glycolide) on healing processes in bone reconstruction. J. Biomed. Mater. Res. B Appl. Biomater. 2010;94(1):108–117. doi: 10.1002/jbm.b.31630. [DOI] [PubMed] [Google Scholar]
- 111.Zhang H.X., Zhang X.P., Xiao G.Y., Hou Y., Cheng L., Si M., Wang S.S., Li Y.H., Nie L. In vitro and in vivo evaluation of calcium phosphate composite scaffolds containing BMP-VEGF loaded PLGA microspheres for the treatment of avascular necrosis of the femoral head. Mater. Sci. Eng. C Mater. Biol. Appl. 2016;60:298–307. doi: 10.1016/j.msec.2015.11.055. [DOI] [PubMed] [Google Scholar]
- 112.Yu D., Li Q., Mu X., Chang T., Xiong Z. Bone regeneration of critical calvarial defect in goat model by PLGA/TCP/rhBMP-2 scaffolds prepared by low-temperature rapid-prototyping technology. Int. J. Oral Maxillofac. Surg. 2008;37(10):929–934. doi: 10.1016/j.ijom.2008.07.012. [DOI] [PubMed] [Google Scholar]
- 113.Ma X., Wu X., Hu Y., Xiong Z., Lv R., Wang J., Li D., Yan Y. Intervertebral spinal fusion using a RP-based PLGA/TCP/bBMP biomimetic grafting material. J. Bioact. Compat Polym. 2009;24:146–157. [Google Scholar]
- 114.Duan C., Liu J., Yuan Z., Meng G., Yang X., Jia S., Zhang J., Chen S. Adenovirus-mediated transfer of VEGF into marrow stromal cells combined with PLGA/TCP scaffold increases vascularization and promotes bone repair in vivo. Arch. Med. Sci. 2014;10(1):174–181. doi: 10.5114/aoms.2012.30950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Lai Y., Li Y., Cao H., Long J., Wang X., Li L., Li C., Jia Q., Teng B., Tang T., Peng J., Eglin D., Alini M., Grijpma D.W., Richards G., Qin L. Osteogenic magnesium incorporated into PLGA/TCP porous scaffold by 3D printing for repairing challenging bone defect. Biomaterials. 2019;197:207–219. doi: 10.1016/j.biomaterials.2019.01.013. [DOI] [PubMed] [Google Scholar]
- 116.Xie X.H., Wang X.L., Zhang G., He Y.X., Leng Y., Tang T.T., Pan X., Qin L. Biofabrication of a PLGA–TCP-based porous bioactive bone substitute with sustained release of icaritin. J. Tissue Eng. Regen. Med. 2015;9(8):961–972. doi: 10.1002/term.1679. [DOI] [PubMed] [Google Scholar]
- 117.Qin L., Yao D., Zheng L., Liu W.C., Liu Z., Lei M., Huang L., Xie X., Wang X., Chen Y. Phytomolecule icaritin incorporated PLGA/TCP scaffold for steroid-associated osteonecrosis: proof-of-concept for prevention of hip joint collapse in bipedal emus and mechanistic study in quadrupedal rabbits. Biomaterials. 2015;59:125–143. doi: 10.1016/j.biomaterials.2015.04.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Lai Y., Cao H., Wang X., Chen S., Zhang M., Wang N., Yao Z., Dai Y., Xie X., Zhang P. Porous composite scaffold incorporating osteogenic phytomolecule icariin for promoting skeletal regeneration in challenging osteonecrotic bone in rabbits. Biomaterials. 2018;153:1–13. doi: 10.1016/j.biomaterials.2017.10.025. [DOI] [PubMed] [Google Scholar]
- 119.Tao Z.S., Wu X.J., Zhou W.S., Wu X.J., Liao W., Yang M., Xu H.G., Yang L. Local administration of aspirin with β-tricalcium phosphate/poly(lactic-co-glycolic acid) (β-TCP/PLGA) could enhance osteoporotic bone regeneration. J. Bone Miner. Metabol. 2019;37(6):1026–1035. doi: 10.1007/s00774-019-01008-w. [DOI] [PubMed] [Google Scholar]
- 120.Välimäki V.V., Aro H.T. Molecular basis for action of bioactive glasses as bone graft substitute. Scand. J. Surg. 2006;95(2):95–102. doi: 10.1177/145749690609500204. [DOI] [PubMed] [Google Scholar]
- 121.Baino F., Fiorilli S., Vitale-Brovarone C. Bioactive glass-based materials with hierarchical porosity for medical applications: Review of recent advances. Acta Biomater. 2016;42:18–32. doi: 10.1016/j.actbio.2016.06.033. [DOI] [PubMed] [Google Scholar]
- 122.Magri A.M.P., Fernandes K.R., Assis L., Kido H.W., Avanzi I.R., Medeiros M.D., Granito R.N., Braga F.J.C., Renno A.C.M. Incorporation of collagen and PLGA in bioactive glass: In vivo biological evaluation. Int. J. Biol. Macromol. 2019;134:869–881. doi: 10.1016/j.ijbiomac.2019.05.090. [DOI] [PubMed] [Google Scholar]
- 123.Dong X., Li H., Zhou Y., Ou L., Cao J., Chang J. The stimulation of osteogenic differentiation of embryoid bodies from human induced pluripotent stem cells by akermanite bioceramics. J. Mater. Chem. B. 2016;4(13):2369–2376. doi: 10.1039/c6tb00398b. [DOI] [PubMed] [Google Scholar]
- 124.Dong X., Li H., Lingling E., Cao J., Guo B. Bioceramic akermanite enhanced vascularization and osteogenic differentiation of human induced pluripotent stem cells in 3D scaffolds in vitro and vivo. RSC Adv. 2019;9(44):25462–25470. doi: 10.1039/c9ra02026h. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Jiang Y.Q., Ouyang Z., Zhang H.G., Wang Y.F., Kang N., Lin C.L., Hou Z.Q., Ye S.F., Ren L. A Molecular basis for enhanced biocompatibility of osteoblasts on poly(lactic-co-glycolic acid)-multi-wall carbon nanotubes nanocomposite. Curr. Nanosci. 2012;8(4):587–597. [Google Scholar]
- 126.Park K.S., Kim B.J., Lih E., Park W., Lee S.H., Joung Y.K., Han D.K. Versatile effects of magnesium hydroxide nanoparticles in PLGA scaffold-mediated chondrogenesis. Acta Biomater. 2018;73:204–216. doi: 10.1016/j.actbio.2018.04.022. [DOI] [PubMed] [Google Scholar]