Abstract
The extant literature confirms that an array of polymorphic genes related to- neurotransmitters and second messengers govern the net release of dopamine in the Nucleus Accumbens (NAc) in the mesolimbic region of the brain. They are linked predominantly to motivation, anti-stress, incentive salience (wanting), and wellbeing. Notably, in 2000 the Nobel Prize was awarded to Carlsson, Greengard, and Kandel for their work on the molecular and cellular function of dopaminergic activity at neurons. This historical psychopharmacological work involved neurotransmission of serotonin, endorphins, glutamate, and dopamine, and the seminal work of Blum, Gold, Volkow, Nestler, and others related to neurotransmitter function and related behaviors. Currently, Americans are facing their second and worst opioid epidemic, prescribed opioids, and easy access drive this epidemic of overdoses, and opioid use disorders (OUDs). Presently the clinical consensus is to treat OUD, as if it were an opioid deficiency syndrome, with long-term to life-long opioid substitution therapy. Opioid agonist administration is seen as necessary to replace missing opioids, treat OUD, and prevent overdoses, like insulin is used to treat diabetes. Treatment of OUD and addiction, in general, is similar to the endocrinopathy conceptualization in that it views opioid agonist MATs as an essential core to therapy. Is this approach logical? Other than as harm reduction, is using opioids to treat OUD therapeutic or harmful in the long term? This historical Trieste provides a molecular framework to understand the current underpinnings of endorphinergic/dopaminergic mechanisms related to opioid deficiency syndrome and generalized reward processing depletion.
WC 249.
Keywords: Brain reward Cascade (BRC), Opioid use disorder (OUD), Neurotransmission, Endorphinergic mechanisms, Genetic testing of addiction liability, Dopamine release and homeostasis, Reward deficiency syndrome (RDS), Endorphinergic deficiency syndrome, Dopamine deficiency syndrome, Precision addiction management’
1. Introduction
There are several neurotransmitters involved in the standard processing of reward and punishment. They include pathways involving at least six major neurotransmitters, and many second messengers. These neurotransmitter networks function within mesolimbic and Pre-Frontal Cortex (PFC), where they regulate the final net pathway of “wanting,” causing neuronal dopamine release [1]. In this article, the authors describe the seminal historical findings related to neuro-biological correlates and neurotransmitters associated with addiction. Notably, how various theorems or hypotheses explained these findings that lead to the modern known correlates of neurotransmitter interactions within the brain reward circuitry, see Fig. 1 .
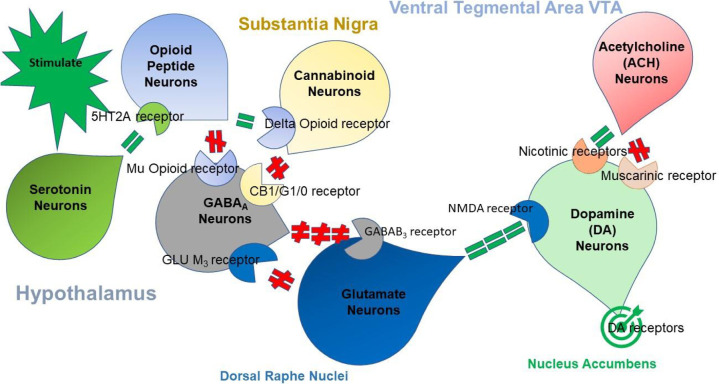
Fig. 1.
The brain reward cascade.
Fig. 1 illustrates the interaction of at least seven major neurotransmitter-pathways involved in the Brain Reward Cascade (BRC). In the hypothalamus environmental stimulation results in the release of serotonin, which in turn via, for example, 5HT-2a receptors activate (green equal sign) the subsequent release of opioid peptides from opioid peptide neurons, also in the hypothalamus. Then, in turn, the opioid peptides have two distinct effects, possibly via two different opioid receptors. One that inhibits (red hash sign) through the mu-opioid receptor (possibly via enkephalin) and projects to the Substania Nigra to GABAA neurons. Another stimulates (green equal sign) cannabinoid neurons (the Anandamide and 2-archydonoglcerol, for example) through Beta –Endorphin linked delta receptors, which in turn inhibit GABAA neurons at the substania nigra. Also, when activated, cannabinoids primarily 2-archydonoglcerol, can indirectly disinhibit (red hash sign) GABAA neurons through activation of G1/0 coupled to CB1 receptors in the Substania Nigra. In the Dorsal Raphe Nuclei (DRN), glutamate neurons can then indirectly disinhibit GABAA neurons in the Substania Nigra through activation of GLU M3 receptors (red hash sign). GABAA neurons, when stimulated, will, in turn, powerfully (red hash signs) inhibit VTA glutaminergic drive via GABAB 3 neurons. It is also possible that stimulation of ACH neurons that at the Nucleus Accumbens ACH can stimulate both muscarinic (red hash) or Nicotinic (green hash). Finally, Glutamate neurons in the VTA will project to dopamine neurons through NMDA receptors (green equal sign)to preferentially release dopamine at the Nucleus Accumbens (NAc) shown as a bullseye indicates a euphoria, or “wanting” response. The result is that when dopamine release is low (endorphin deficiency), unhappiness is felt while general (healthy) happiness depends on the dopamine homeostatic tonic set point (see Fig. 2).
The cascading interaction of these neurotransmitters and second messengers results in the correct release of dopamine within the NAc and across many brain regions. These regions are involved in motivation, cognition (memory), pleasure, stress reduction, drug reinstatement, decision-making, recall, wellbeing, and especially cravings [2]. The result is to provide homo sapiens with a usual happiness set –point (Fig. 2 ) identified as resting-state functional connectivity (rsFC).
Fig. 2.
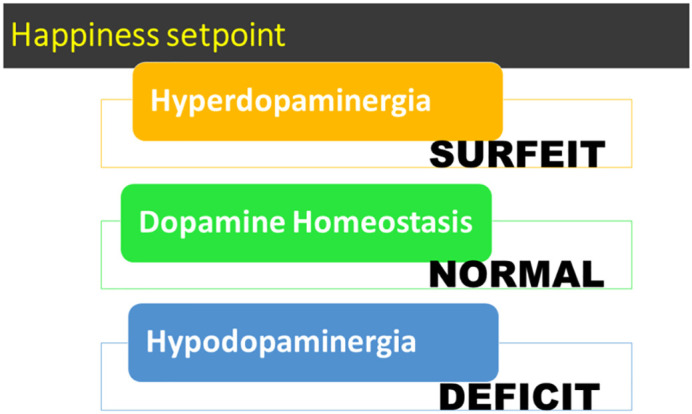
A simple schematic of tonic happiness –set-point based on dopamine release, as explained in Fig. 1.
Over the last 60 years starting in 1968, there has been a remarkable change in understanding the science behind how psychoactive drugs influence behavior through very complex actions on neuronal pathways, especially in the mesolimbic system and the prefrontal cortex –cingulate gyrus of the brain [3]. Ideas about recovery, incorporating the 12-step program and fellowship, and introducing the new “addiction” definition from the American Society of Addiction Medicine (ASAM), had a tremendous impact on our younger generation. The general acceptance of the now well-established phenomena elaborating on the undeniable fact that addiction is indeed, a brain disorder is changing the face of addiction medicine [4].
Early work from several Neuropharmacologists centered on serotonin and brain reward circuitry, revealing that depletion of brain serotonin augments stress-like reactions in electrical brain self-stimulating rodents. They used p-chlorophenyl alanine (PCA), a tryptophan hydroxylase inhibitor that selectively depletes brain serotonin content [5]. This experiment suggested that PCA promotes anxiety, and serotonin is an anti-anxiety brain substance. Along similar lines following the work of Myers and Cicero [3], and knowing at the time that during dark-phase, levels of serotonin in the pineal gland are low, led Geller's group look for evidence in rodents of enhanced darkness-induced ethanol drinking. The first experiment showed that rats placed in a dark closet drank more alcohol than those housed in the light [5]. The hypothesis was that an increase in pineal melatonin caused increased drinking. Subsequently, injections of melatonin in rats exposed under “normal” (nine hours of darkness during a 24-h day) photoperiods revealed this to be accurate since they also displayed augmented ethanol intake; consequently, several experiments validated these results [[6], [7], [8]]. Experiments that found conversion to melatonin via adenylate cyclase stimulation of N- acetyltransferase increased three-fold at night understanding of these theories improved [[9], [10], [11], [12]]. Another theory concerning the neurochemical mechanism(s) of ethanol consumption suggested the role of dopamine condensation products, known as isoquinolines.
In 1970, Davis and Walsh [13] proposed that a product of alcohol, tetrahydropapaverine, a benzyltetrahydroisoquinoline alkaloid derivative of the biogenic amine, dopamine, and acetaldehyde, condenses and can induce ethanol intake in rodents. Simultaneously, others reported a role in alcoholism for biogenic amines (for example, norepinephrine) and later indoleamine (serotonin) aldehyde condensation products salsolinol and carbochol, respectively [14,15]. This work suggested that alcohol and opiate addiction might have common neurochemical mechanisms. Indeed at the time controversy stimulated research by neuroscientists that provided evidence that: ethanol intake augmented the salsolinol metabolite in the rat brain [16,17]; salsolinol induced an increase in ethanol intake [15]; salsolinol acts like an opiate agonist [[18], [19], [20]]; and salsolinol induced alcohol withdrawal tremors blocked by narcotic antagonism [21].
Following these early findings, the 70s promised the world a new era of Sex, Drugs, and Rock N Roll [22]. Then in the 80s, following comments regarding the recreational use of cocaine, Gold's group was the first to propose the “dopamine depletion hypothesis and suggested the use of bromocriptine to reduce cocaine cravings [23]. Finally, in the 90s, researchers began to see attempts to understand the neurogenetic and neurobiological mechanisms related to substance abuse through neuroimaging techniques [24,25].
2. Theorems relevant to opioid peptides and reward processing deficiency
“Theorem” is defined as a proposition that can be deduced from the premises or assumptions of a system. The following information portrays the historical work related to neurotransmitters, their receptors, agonism, and antagonism, and their respective roles in brain reward circuitry. It demonstrates how this body of work and the theorems that evolved from it, now support the modern view of addictive behaviors. The “comments” in Table 1, Table 2, Table 3, Table 4, Table 5 are explanations of the relevance of each study to understanding the premises of “Theorems” or medical hypotheses deduced by an exploration of research topics and serves as the foundational neurobiological basis of addiction in these modern times. It is noteworthy that the neurophysiology of the dopamine depletion theorem is synonymous with hypodopaminergia.
Table 1.
Neurotransmitter deficiency theorem.
| Reference | Topic | Comment |
|---|---|---|
| [27] | Ethanol-induced sleep -time with narcotic antagonism | The first experiment showing Naloxone blocking ethanol action |
| [28] | D-Phenylalanine an inhibitor of enkephalinase in the brain of genetically prone rodents | Enhanced brain endorphins with reduced ethanol intake |
| [29] | Synergistic brain effects of dopamine and ethanol | Dopamine potentiated the soporific action of ethanol |
| [30] | Synergistic brain effects of serotonin and ethanol | Serotonin potentiated the soporific action of ethanol |
| [31] | Enkephalins and drinking behavior | Low enkephalin genetically high drinking |
| [32] | Enkephalinase inhibition of alcohol intake | Inhibition of enkephalinase and precursor amino –acids results in an increase in brain enkephalins and a decrease in alcohol intake |
| [33] | Naloxone blockade of ethanol dependence | Demonstrates common mechanism between alcohol and opiates |
| [34] | Chronic alcohol intake blocks the neuronal synthesis of enkephalins | The first study to show that long-term drinking alcohol disrupts endogenous opioid peptides |
| [35] | Morphine induced attenuation of ethanol withdrawal symptoms | Further support for a common mechanism between alcohol and opiates |
| [36] | Ethanol preference as a function of genotypic levels of whole brain enkephalin in mice was studied | Enkephalin levels in genetically bred mice differ. |
| [37] | Methylated enkephalin reduces alcohol intake in genetically bred alcohol loving mice | Enkephlamide I.C. Brain injections reduced alcohol intake in C57BLK/6 J mice |
| [38] | Association of the dopamine D2 receptor A1 allele with severe alcoholism | The first association of the dopamine D2 receptor A1 allele with severe alcoholism start of Psychiatric Genetics |
| [39] | Binding studies of DRD2 receptor polymorphisms in non-alcoholics | 30–40% reduction of the number of D2 receptors s a function of the D2 polymorphism Taq A1 allele. |
| [40] | The dopamine receptor gene (DRD2) as a determinant of Reward Deficiency Syndrome (RDS) | Coined RDS now featured in SAGE Encyclopedia for Clinical & Abnormal Psychology |
Table 2.
The reward deficiency syndrome theorem.
| Reference | Topic | Comment |
|---|---|---|
| [43] | Neurogenetic evidence for Genetic Addiction Risk Score (GARS) | Developed Genetic Addiction Risk Score(GARS) to identify risk for RDS |
| [44] | Developed DNA directed gene guided therapy with pro-dopamine regulation | Provided first evidence for nutrigenomic coupled precision personalized treatment for obesity |
| [45] | Enhanced resting-state functional connectivity in naive animal models with enkephalinase inhibition | The first study to show activation of mesolimbic dopaminergic activation across brain reward circuitry with Pro-dopamine regulation |
| [46] | Enkephalinase opioid substitution therapy during withdrawal from Bup/Nal | Pro-dopamine agonist therapy blocks opioid withdrawal and maintains opioid-free urines for 432 days |
| [47] | Enkephalinase inhibition attenuation of opioid detoxification | Pro-dopamine regulation as a frontline technology to induce detoxification reducing withdrawal symptoms in opioid dependence |
| [48] | Opioid/dopamine deficiency genotype theorem | Early theory proposing that genetic or epigenetic insult onto endogenous opioid peptides and dopamine will induce drug-seeking |
| [49] | Enkephalinase inhibition attenuation of alcohol binge in drinking in rodents | Significant reduction of binge drinking in an animal model of binging in genetically prone animals with Pro-dopamine regulation |
| [50] | Development of DNA directed precision addiction management | Proposed coupling of GARS and KB220 matched algorithmic polymorphic nutrigenomic anti-RDS |
| [42] | rsfMRI effects of KB220Z™ on neural pathways in the reward circuitry of abstinent genotyped heroin addicts | Enhanced functional connectivity and neuronal dopaminergic recruitment in abstinent heroin addicts |
Table 3.
Dopamine depletion hypothesis and similar studies suggesting endorphin deficiency.
| Reference | Topic | Comment |
|---|---|---|
| [55] | A rationale for opiate withdrawal symptomatology | Withdrawal of opiates removes this “tonic” inhibition of the locus coeruleus (LC) and could readily result in a piperoxane-like release from inhibition. |
| [56] | Endorphin locus coeruleus connection mediates opiate action and withdrawal | This hypothesis suggests that chronic opiate administration inhibits LC activity and that opiate abstinence after addiction results in a release from inhibition with resultant LC hyperactivity. |
| [57] | Endorphins, lithium, and Naloxone: their relationship to pathological and drug-induced manic-euphoric states | Opioid peptides play a role in drug-induced manic –euphoric stares |
| [58] | Decreased serum prolactin in opiate withdrawal and dopaminergic hyperactivity is investigated | Dopaminergic hyperactivity is present in opiate withdrawal but may not be related to the signs and symptoms of withdrawal; the culprit is norepinephrine; however, dopamine reduces symptoms. |
| [59] | New concepts in cocaine addiction: the dopamine depletion hypothesis due to hypodopaminergia | Cocaine addiction was a dopamine derangement depletion disease. DA depletion is hypothesized to result from the overstimulation of these neurons and excessive synaptic metabolism of the neurotransmitter. Cocaine was addicting on this basis, and cocaine withdrawal looks like retarded-psychomotor depression. |
| [60] | Bromocriptine treatment for cocaine abuse: the dopamine (DA) depletion hypothesis | Bromocriptine, a DA/antagonist, appears to have efficacy with acute and maintenance trials and may represent a test of the cocaine-dopamine hypothesis and new adjunctive treatment for cocaine abuse. |
| [61] | Anti-endorphin effects of methadone. | Depression is common and may be induced by methadone. Post-detoxification depression and other affective symptoms may be due to hypodopaminergia and contribute to relapse. |
| [62] | A possible opioid receptor dysfunction in some depressive disorders | In some depressive symptoms, the etiology may reside in the dysfunction of the opioid peptide system |
| [63] | Clonidine used in detoxification from opiates | Clonidine hydrochloride in detoxification from opiates has demonstrated that a non-opioid medication can rapidly suppress the symptoms associated with opiate withdrawal. The alpha-2 adrenergic and endorphinergic systems are parallel inhibitors of the nucleus locus coeruleus. |
| [64] | Identification of a subregion within rat neostriatum for the dopaminergic modulation of lateral hypothalamic self-stimulation | Dopaminergic transmission in Ventral Anterior Striatum (VAS), alone among the striatal sites tested, is facilitatory on hypothalamic self-stimulation. |
| [65] | Platelet serotonin transporter in cocaine patients | Chronic cocaine inhibits serotonin transporter increasing synaptic serotonin. This increase has been documented through optogenetics to inhibit GABAergic firing increasing DA signaling |
| [66] | Naltrexone augments the effects of nicotine replacement therapy (NRT) in female smokers | Naltrexone, causes extinction blocks D2 receptors, and combined with NRT and psychosocial therapy, appears to have a positive cessation effect for women and is a potential new treatment for recidivist female nicotine smokers. |
| [67] | Diagnosis of alcohol dependence and family history prevalence in cocaine dependence | The study finds a high prevalence of alcohol (68% and 89%) and cannabis dependence (53% and 46%) in patients with cocaine dependence. These various dependencies support the RDS concept. |
| [68] | Dopaminergic activation in the reward circuitry regulates food and drug craving behavior | The dopaminergic, enkephalinergic, and fos gene expressions are important regulatory genetic pathways for both food and drug craving behaviors as proposed in RDS |
| [69] | Dysfunctional connectivity patterns in chronic heroin users: an fMRI study | This dysfunctional brain connectivity may contribute to decreasing self-control, impaired inhibitory function as well as deficits in stress regulation in chronic heroin users. |
| [70] | Gray matter deficits and resting-state abnormalities in abstinent heroin-dependent individuals was studied | Chronic heroin dependence impairs the right Dorsolateral Prefrontal Cortex (DLPFC) in heroin-dependent individuals, including structural deficits and resting-state functional impairments. |
| [71] | Buprenorphine effect on fentanyl withdrawal in rats | Buprenorphine prevents affective and somatic fentanyl withdrawal signs. Moreover, buprenorphine is rewarding in rats previously exposed to fentanyl, but not in opioid-naïve rats. |
| [72] | The Salted Food Addiction Hypothesis may explain the overeating and obesity epidemic | Salt may increase reinforcement produced by fat-rich and other foods. This combination may be an addictive substance that can stimulate opiate and dopamine receptors in the brain's reward and pleasure center. This effect may explain the salted food preference, urge, craving, and hunger manifestations of opiate withdrawal. |
| [73] | Distinct resting-state brain activities in heroin-dependent individuals | BOLD activation pattern differences of heroin-dependent and healthy subjects included the orbitofrontal cortex (OFC), cingulate gyrus, frontal and paralimbic regions, anterior cingulate cortex (ACC), hippocampal/parahippocampal region, amygdala, caudate, putamen, & posterior insula and thalamus |
| [74] | Body mass index and alcohol use | Obese patients have lower alcohol use rates than found in the general population of women. As BMI increases, rates of alcohol consumption are lower. Overeating may compete with alcohol for brain reward sites, making alcohol ingestion less reinforcing. |
| [75] | Mice with a deficiency of the G protein Go have multiple neurological abnormalities | The G protein Go is highly expressed in neurons and mediates effects of a group of rhodopsin-like receptors that includes the opioid, alpha2-adrenergic, M2 muscarinic, and somatostatin receptors. Go plays a central role in motor control, motor behavior, pain perception and is involved in Ca2+ channel regulation |
| [76] | Neuro-chemical activation of brain reward mesolimbic circuitry is associated with relapse prevention and drug hunger: a hypothesis | Proposing a paradigm shift in residential, non-residential, and aftercare involving the incorporation of genetic testing to identify risk alleles coupled with D2 receptor stimulation using neuroadaptogen amino acid precursor enkephalinase–catecholamine-methyltransferase (COMT) inhibition therapy. A natural but therapeutic nutraceutical formulation potentially induces DA release and cause the induction of D2-directed mRNA and proliferation of D2 receptors in the human. |
| [77] | The relationship between obesity and drug use | Obesity may inhibit drug reinforcement. Neurotransmitter release in the nucleus accumbens is linked to self-administration and learning following drug use. Food intake or sex activates this endogenous reward system. Therefore, hyperphagia following abstinence may be a rebound mechanism to replenish neurotransmitter release in this reward system, which leads to weight gain and a rise in body BMI during recovery from substance abuse. |
| [78] | Na(+) channel blockers for the treatment of pain: context is everything, almost | Voltage-gated Na + channels (VGSCs) remain viable targets for the development of novel analgesics |
| [79] | Addiction conceptualized from an Osteopathic Perspective: Dopamine Homeostasis | The basis of much of osteopathic philosophy is the idea of total body allostasis and homeostasis. The achievement of dopamine homeostasis as part of a comprehensive biopsychosocial treatment strategy in the effective management of addiction is discussed in this article. |
| [80] | The Food and Drug Addiction Epidemic: Targeting Dopamine Homeostasis. | Strategies indicated by this understanding of nutriepigenomics and neurogenetics in the treatment and prevention of metabolic syndrome and obesity include the moderation of mRNA expression by DNA methylation and inhibition of histone deacetylation. |
| [81] | Microglial activation in the brains of human users of Methamphetamine was investigated. | Midbrain, striatum, thalamus, orbitofrontal and insular cortices binding levels correlated inversely with the methamphetamine abstinence duration (p < 0.05). These results suggest that reactive microgliosis can result from chronic self-administration of methamphetamine, but the level of activation appears to subside over longer periods of abstinence. |
| [82] | Methamphetamine (METH)- and trauma-induced brain injuries (TBI): comparative cellular and molecular neurobiological substrates | Functional changes in the brain from METH use disorder (MUD) result in changes similar to those in TBI. A greater benefit to those with MUD might result from rehabilitation and treatment with neuroprotective pharmacological agents such as calpain and caspase inhibitors, used to treat TBI. |
| [83] | Depression in Opiate Addicts | The authors note that methadone programs have a high prevalence of depression and concurrent treatment for depression. They gave the dexamethasone suppression test (DST) to 42 opiate addicts two weeks after detoxification. The DST provided a sensitivity of 80%, and a specificity of 93% for Major Depression diagnosed according to the Research Diagnostic Criteria. |
| [84] | Methadone-induced endorphin dysfunction in MUD. | Similar to alcohol as reported by Blum et al. methadone induces an apparent endorphin dysfunction in polydrug abusers |
Table 4.
Hypodopaminergia receptor theorem.
| Reference | Topic | Comment |
|---|---|---|
| [94] | Biological correlates of mental activity studied with positron emission tomography (PET). | Findings obtained with PET illustrate the highly interactive organization of the brain and can be used to identify the number of dopaminergic and other neurotransmitter receptors |
| [95] | Amphetamine induced decreases in (18F)-N-methylspiroperidol binding in the baboon brain using positron emission tomography (PET) | The uptake and retention, in the rat striatum, were five-fold higher for [18F]-N-methylspiroperidol than for [18F]spiroperidol. These results suggest that [18F]-N-methylspiroperidol is an ideal choice for studies of the dopamine receptor in humans |
| [96] | The metabolism in cocaine dependence and withdrawal changed brain glucose metabolism | Selectivity in changes in glucose metabolism of the basal ganglia and the orbitofrontal cortex suggest that regional metabolic changes during detoxification from CUD are related to changes in brain dopamine activity. |
| [97] | Age-associated decrements in dopamine D2 receptors in the thalamus and temporal insula of human subjects | The decrease in DA D2 receptor availability was 6.6% per decade in caudate, 8.2% in the putamen, 7.6% in the thalamus, and 13% in the temporal insula. This study indicates that D2 losses with age are not limited to the striatum and also involve thalamic as well as temporal cortical regions. This depletion results in well- known increases in substance seeking in older adults. |
| [98] | Cerebral blood flow changes following acute alcohol intoxication were measured with PET | Reduced blood flow to the cerebellum was observed with high and low alcohol doses. The decrease in cerebellum blood flow could account for the muscular incoordination induced by alcohol. While acutely increased alcohol intake causes increased dopamine release, chronic alcohol intake results in reduced dopamine availability. |
| [99] | Blunted response to alcohol intoxication was observed in limbic brain regions of people with CUD. | Decreased sensitivity in reward circuits of people with CUD could be responsible for the blunted limbic and cortical response to alcohol with associated depleted dopamine. |
| [100] | Increases in brain dopamine and D(2) receptor occupancy is associated with the reinforcing effects of psychostimulants in humans | This study is the initial clear demonstration that stimulant-induced high, a mood descriptor that reflects reinforcing effects of drugs in humans, associates with increases in brain dopamine. It also demonstrates a quantitative relationship between the intensity of the high and levels of D(2) receptor occupancy by dopamine. |
| [101] | The serotonergic modulation of dopamine was measured with [11C] raclopride and PET in healthy human subjects. | A decrease in [11C] raclopride binding is consistent with an observed increase in dopamine concentrations and the ability of serotonins' ability to neurobiologically stimulate dopamine activity. |
| [102] | Brain dopamine and obesity | Dopamine modulates motivation and reward. In obese individuals, the availability of dopamine D2 receptors decreased proportionally to BMI. In these individuals, dopamine deficiency may perpetuate compensatory pathological eating to increase dopamine activation. |
| [103] | Dopamine receptor-mediated regulation of striatal cholinergic activity: positron emission tomography studies with norchloro[18F]fluoroepibatidine | This study suggests that under normal physiological conditions, the predominant influence of endogenous dopamine on striatal ACH output is receptor-mediated dopamine D2, not D1. |
| [104] | Initial PET Studies used [18F]Ro41-0960 to map catechol-O-methyltransferase in vivo | Ro41-0960 is a potent, fluorine-containing COMT inhibitor that reported to cross the blood-brain barrier and to inhibit COMT in the brain. This was the first example of a positron emitter labeled radiotracer for COMT and provide initial encouraging evidence that [18F]Ro41-0960 may be used to examine COMT in peripheral organs in vivo. COMT catabolizes synaptic dopamine and serotonin, carriers of the Val variant catabolize excessive amounts at about 4× the standard Met variant |
| [105] | Human striatal dopamine release during aerobic exercise was studied using PET | The subjects exercised vigorously for 30 min; however, no significant changes in synaptic DA concentration were detected. |
| [106] | “Nonheroic” food motivation in humans involves dopamine in the dorsal striatum and methylphenidate amplifies this effect. | The initial evidence that dopamine in the dorsal striatum is involved in food motivation in humans distinct from the role of dopamine in regulating reward through the NAc. This study also demonstrated the ability of methylphenidate to amplify weak dopamine signals. |
| [107] | Naloxone acts as an inverse opiate agonist having mechanistic implications for drug dependence and withdrawal | According to this model, dependence is the overexpression of the active receptors, and withdrawal is a sudden change from the active to the inactive state. |
| [108] | Dopamine D2 receptor availability was measured in opiate-dependent subjects before and after naloxone-precipitated withdrawal. | Although no significant changes in striatal DA concentration are documented in this study during acute withdrawal, decreases in D2 receptors in opiate-dependent subjects are documented. |
| [109] | Bariatric surgery reduced resting connectivity of brain regions involved with self-referential processing in obese patients | Obese individuals exhibit brain alterations of resting-state functional connectivity (RSFC) integrity of resting-state networks (RSNs) related to food intake. Bariatric surgery significantly decreased FCD in regions involved in self-referential processing ventromedial prefrontal cortex (VMPFC), dorsomedial prefrontal cortex (DMPFC), Dorsal anterior cingulate cortex (dACC), precuneus, and interoception (insula), and changes in VMPFC/precuneus were associated with a reduction in BMI suggesting a role in improving control of eating behaviors following surgery. |
| [110] | GABAergic attenuation of cocaine-induced dopamine release and locomotor activity | Gamma-vinyl GABA (GVG) significantly and dose-dependently attenuated cocaine-induced dopamine release in the corpus striatum of freely moving animals. |
| [111] | The dopamine system is effected by Delta (9)-tetrahydrocannabinol. | Acute THC administration causes increased dopamine release and neuron activity, whereas long-term use is associated with blunting of the dopamine system. Future research must examine the long-term and developmental dopaminergic effects of THC. |
| [112] | Misconceptions and mitigation strategies for OUD in chronic pain patients | Review article discussing possible hyperalgesia and other risks |
| [113] | Medication-assisted therapies–tackling the opioid-overdose epidemic | The rates of emergency department visits and substance-abuse treatment admissions related to prescription opioids increased markedly. Insurers paid an estimated $72.5 billion for prescription-opioid abuse in 2007, a substantial increase over previous years |
| [114] | Untangling the complexity of opioid receptor function | Studies reveal multiple layers of complexity of opioid receptor function, including a spatiotemporal specificity in opioid receptor-induced cellular signaling, ligand-directed biased signaling, allosteric modulation of ligand interactions, heterodimerization of different opioid receptors, and the existence of splice variants with different ligand specificity. |
| [115] | Association of genetic ancestry with striatal dopamine D2/D3 receptor availability | Variation in dopaminergic candidate genes did not drive the effects of ethnicity on D2R. Instead, candidate gene associations with striatal D2R diminished when correcting for ancestry. |
| [116] | NOW vs. LATER brain circuits: implications for obesity and addiction. | DA enables shifting from NOW to LATER, while its modulation of the insula, which processes interoceptive information, influences the probability of selecting NOW vs. LATER actions based on an individual's physiological state. |
| [117] | Effect of combined naltrexone and bupropion therapy on the brain's functional connectivity | NB32 (sustained-release Buprenorphine) treatment decreased local and global FCD in superior parietal cortex and increased its connectivity with ACC (involved with saliency attribution), insula (interoception), and decreased local FCD in the medial prefrontal cortex (craving), which might underlie NB32 improved control over eating behaviors. |
| [118] | Impaired periamygdaloid-cortex prodynorphin (PDYN) is characteristic of opiate addiction and depression. | Found reduced PDYN mRNA expression in the postmortem human amygdala nucleus of the periamygdaloid cortex (PAC) in both heroin abusers and MDD subjects, which was confirmed by animal experimentation. |
Table 5.
The epigenetic theorem.
| Reference | Topic | Comment |
|---|---|---|
| [122] | Dual mechanisms of opiate dependence in the locus coeruleus | Together, data from VTA and LC illustrate the complex and important changes in synaptic, cellular, and structural plasticity that mediate the lasting effects of opiate drugs on the brain's catecholamine neurons and other neuronal types in these regions, which in turn influence drug reward and dependence. |
| [123] | The common intracellular actions of chronic morphine and cocaine in dopaminergic brain reward regions | Lesions in the VTA disrupt self-administration of cocaine and opioids due to impairment of dopaminergic signaling |
| [124] | neurotrophic factors influence morphine- and cocaine-induced biochemical changes in the mesolimbic dopamine system | Increase in tyrosine hydroxylase in the VTA following chronic morphine and chronic cocaine treatments has been shown in previous research. Chronic morphine treatment also increases levels of glial fibrillary acidic protein in this brain region. Brain-derived neurotrophic factor, which by itself tended to decrease tyrosine hydroxylase levels in the VTA, prevented the characteristic increase in tyrosine hydroxylase following morphine and cocaine exposure and reversed the increase in rats pretreated with morphine. |
| [125] | Chronic ingestion of ethanol up-regulates NMDAR1 receptor subunit immunoreactivity in rat hippocampus. | Increased NMDAR1 subunit levels in the hippocampus after chronic ethanol exposure may represent an important neurochemical substrate for some of the features associated with ethanol dependence and withdrawal. |
| [126] | A persistent decrease in adenylate cyclase is a novel action of morphine in the rat locus coeruleus | A persistent decrease in adenylate cyclase occurred in a dose-dependent manner mediated through the actions of morphine at opiate receptors. Naltrexone, a long-acting opiate receptor antagonist was administered in vivo concomitantly and prevented the inhibition. |
| [127] | Chronic buprenorphine treatment attenuates cocaine conditioned place preference. | Buprenorphine (BUP), a mixed opioid agonist-antagonist, was shown to suppresses cocaine self-administration in monkeys and reduce cocaine use in humans. This study found that BUP reduces the ability of cocaine to condition a place preference in rats. |
| [128] | The expression of GABAA receptor alpha 1 and alpha 5 subunits in the VTA and hippocampus are regulated by chronic ethanol administration | Based on these data the effects of chronic ethanol administration on the GABAA receptor subunit expression in the hippocampus and VTA may play a role in craving and other detrimental cognitive effects of alcohol. |
| [129] | Chronic cocaine effects regulation of immediate early gene expression and AP-1 binding in the rat nucleus accumbens | Levels of Fos-like c-fos and c-jun mRNA immunoreactivity, which are increased in the NAc by acute cocaine, were reduced to control levels. Meanwhile in animals treated chronically with cocaine, AP-1 binding activity in the NAc remained elevated 18 h after the last chronic injection. |
| [130] | Protein phosphorylation and second messenger mechanisms possibly underly genetic vulnerability to alcoholism | Found that chronic opiates increase levels of the G-protein subunits Gi alpha and Go alpha, adenylate cyclase, cyclic AMP-dependent protein kinase, and some phosphoproteins (including tyrosine hydroxylase) in this brain region |
| [131] | Induction of chronic Fos-related antigens in rat brain by chronic morphine administration | Prolonged withdrawal periods induced Fos-related antigens (chronic Fras) in several brain regions, but not chronic morphine alone. |
| [132] | In the rat mesolimbic dopamine system axoplasmic transport is impaired by chronic morphine | Chronic morphine decreased axonal transport from the VTA to the NAc by 50%. However, consistent with its lack of effect on neurofilaments (NFs) in this brain region chronic morphine did not alter axonal transport from the locus coeruleus to several of its projection areas. |
| [133] | Behavioral, electrophysiological, and biochemical correlates of opiate withdrawal in the locus coeruleus (LC) of the rat | Increased LC neuronal activity is associated with temporally increased levels of G-proteins and an up-regulated cAMP system. These may contribute to the early-withdrawal activation of these neurons in behavioral morphine withdrawal syndrome |
| [134] | Morphine-reward and feeding are regulated by CREB, in the lateral hypothalamus. | The study revealed that opioids and stress induce increased transcription of CREB in the lateral hypothalamus which can effect both drug and food reward |
| [135] | Adenylyl cyclase-5 activity in the NAc regulates anxiety-related behavior | Administration of norbinaltorphimine (a kappa-opioid receptor antagonist) or CCK-8 s (a CCK receptor agonist) reversed the anxiolytic-like behavior exhibited by AC5−/− mutants. These results suggest an essential role of 5 adenylyl cyclase (AC5) in the NAc for maintaining healthy levels of anxiety. |
| [136] | LC behavioral adaptations to chronic emotional stimuli are controlled epigenetically by Histone deacetylase 5 | Exposure to chronic but not acute cocaine or stress decreases HDAC5 function in the NAc in the brain reward region, which allows for increased histone acetylation and transcription of HDAC5 target genes |
| [137] | The induction of Delta FosB in reward-related brain structures after chronic stress was studied. | Chronic restraint-stress induced DeltaFosB expression in several discrete regions of the brain, predominantly in the Frontal Cortex (fCTX), NAc, and basolateral amygdala, with lower levels of induction seen elsewhere. These findings establish that chronic stress induced DeltaFosB induction could contribute to the long-term effects of stress on the brain. |
| [138] | Hcrtr1 and 2 signaling differentially regulate depression-like behaviors. | Significantly reduced behavioral despair in the forced swim test and tail suspension test was displayed by Hcrtr1 null mice. Also, Wild-type mice treated with the hcrtr1 antagonist SB-334867 displayed a similar reduction in behavioral despair. These studies suggest the balance of orexin action at the hcrtr1 or the hcrtr2 receptor produces an anti-depressant or pro-depressant like effect, depending on the receptor subtype activated. |
| [139] | Essential role for RGS9 in opiate action | A family of G protein signaling (RGS) regulators are known to accelerate effector stimulation termination after G protein receptor activation. A brain-specific splice variant of the RGS9 gene, RGS9-2, is highly enriched in the striatum and expressed at much lower levels in the periaqueductal gray and spinal cord structures known to mediate various actions of morphine and other opiates. Mice lacking RGS9 show enhanced behavioral responses to acute and chronic morphine, including a dramatic increase in morphine reward, increased morphine analgesia with delayed tolerance, and exacerbated morphine physical dependence and withdrawal. |
| [140] | Inhibition of cAMP response element-binding protein or dynorphin in the nucleus accumbens produces an antidepressant-like effect. | Together, the results demonstrate that the nucleus accumbens CREB-dynorphin influence behavior in the learned helplessness model and suggest that this signaling cascade may contribute to symptoms of depression. |
| [141] | Epigenetic Basis of Mental Illness | Exposure to environmental insults induces stable changes in gene expression, neural circuit function, and ultimately behavior, and these maladaptations appear distinct between developmental versus adult exposures. Increasing evidence indicates that epigenetic modifications maintain these sustained abnormalities in specific brain regions. Indeed, the transcriptional dysregulation and the aberrant epigenetic regulation that underlies this dysregulation is a unifying theme in psychiatric disorders |
| [142] | The epigenetic mechanisms of chronic pain. | There is evidence that injury-induced changes in chromatin structure drive persistent cellular and molecular adaptations in gene expression and neural function, which may contribute to the development of several symptoms of chronic pain, including allodynia, hyperalgesia, anxiety, and depression. |
| [143] | Epigenetic mechanisms of drug addiction | Three major modes of epigenetic regulation-histone modifications such as acetylation and methylation, DNA methylation, and non-coding RNAs are changes induced within the brain's reward regions by repeated exposure to drugs of abuse |
| [144] | Susceptibility to Social Defeat Stress is controlled by cell-type-specific epigenetic editing at the FosB gene | This demonstrates the first cell- and gene-specific targeted histone modifications, which model naturally occurring transcriptional phenomena that control social defeat stress behavior. This epigenetic-editing approach, which recapitulates physiological changes in gene expression, reveals clear differences in the social defeat phenotype induced by FosB gene manipulation in medium spiny neuron) MSN subtypes. |
| [145] | The mesolimbic dopamine reward circuit in depression | This study focused on the NAc ventral striatum, its dopaminergic input from the VTA and the potential role of the mesolimbic dopamine system in depression. The mesolimbic dopamine system is most often associated with the rewarding effects of food, sex, and drugs of abuse. |
| [146] | The role of DNA methylation within the nucleus accumbens in the incubation of cocaine craving | Withdrawal periods, and cue-induced cocaine seeking, were associated in this study with broad, time-dependent enhanced DNA methylation alterations in the NAc. |
| [147] | Inhibition of Class I, HDAC blocks cocaine-induced plasticity by targeted changes in histone methylation | This study demonstrates that specific and prolonged blockade of HDAC1 in NAc of mice increased global levels of histone acetylation, induced repressive histone methylation and antagonized cocaine-induced changes in behavior. This effect was mediated in-part through a chromatin-mediated suppression of GABAA receptor subunit expression and inhibitory tone on NAc neurons. |
| [148] | The expression of the RhoA network in the NAc to control synaptic structure is augmented by withdrawal from chronic morphine administration | The nucleus NAc is a critical reward region of the brain that mediates the effects of psychoactive substances including morphine and other opiates. An important role for hippocampal RhoA in morphine-facilitated reward behavior. Recent work revealed an important role for RhoA signaling cascades in mediating the effects of long-term morphine withdrawal on NAc MSN dendritic spine elimination. |
| [149] | Histone arginine methylation was observed in cocaine action in the NAc | Src kinase (Srcin1), signaling inhibitor also referred to as (p140Cap) is a key gene target for reduced H3R2me2a binding. These findings suggest that PRMT6 and H3R2me2a down-regulation suppression of Src signaling in NAc D2-MSNs, via, functions is a homeostatic brake to restrain cocaine action. This finding may provide novel candidates for the development of treatments for cocaine addiction-like Pro-dopamine regulator (KB220). |
| [150] | Histone acetylation in drug addiction | Regulation of chromatin structure through post-translational modifications of histones (e.g., acetylation) has emerged as an important mechanism to translate a variety of environmental stimuli, including drugs of abuse, into specific changes in gene expression. |
| [151] | The striatal balancing act in drug addiction: distinct roles of direct and indirect pathway medium spiny neurons | The striatum plays a crucial role in mediating the acute and chronic effects of addictive drugs, with drugs of abuse causing long-lasting molecular and cellular alterations in both dorsal striatum and nucleus accumbens (ventral striatum). We now know there are two MSN subtypes (direct & indirect) in the long-term actions of drugs of abuse. |
| [152] | The epigenetic basis of opiate suppression of Bdnf gene expression in the ventral tegmental area was delineated | Found that morphine suppressed binding of phospho-CREB (cAMP response element-binding protein) to Bdnf promoters in VTA, which resulted from the enrichment of trimethylated H3K27 at the promoters, and that decreased NURR1 (nuclear receptor related-1) expression also contributed to Bdnf repression and associated behavioral plasticity to morphine. |
| [153] | Proenkephalin (Penk) mediates the enduring effects of adolescent cannabis exposure associated with adult opiate vulnerability | Following adolescent Δ(9)-tetrahydrocannabinol (THC) exposure, enhanced heroin self-administration (SA) and dysregulation of the endogenous opioid system in the NAc shell (sh) of adults has been demonstrated in animal studies. A direct association between heroin self-administration and THC-induced NAcsh Penk upregulation indicate that epigenetic dysregulation of Penk underlies the long-term effects of THC were established by this data. |
2.1. Blum's neurotransmitter deficiency theorem
understanding of the common neurochemical mechanisms involved in seemingly diverse chemical agents' alcohol and opiates, followed an array of experimental evidence and provided the material for a book dedicated to this concept, “Alcohol and Opiates: Common Neurochemical Correlates [26]. Along these lines, research from Blum's group focused on the involvement of neurotransmitters in the processing of reward and the neurological underpinnings of addictive behaviors drug and non-drug.
In brief, the following represents studies that dovetail on endorphinergic /dopaminergic mechanisms related to generalized Substance Use Disorder (SUD) with emphasis on AUD and OUD, see Table 1.
2.2. Reward deficiency syndrome
In 1996 following the association of the A1 allele of the DRD2 gene with severe alcoholism [38], Blum et al., found that a host of addictive, impulsive and compulsive behaviors had also associated with this same DRD2 allele that caused reduced numbers of DRD2 receptors and hypodopaminergia [39]. He called these behaviors collectively “Reward Deficiency Syndrome” [40] now included in the SAGE Encyclopedia for Clinical and Abnormal Psychology [41]. Based on the reward circuitry, genetic polymorphisms that also contributed to this deficiency, were identified from the available and continually expanding field of genetic research (see Table 2), and were foundational in the development of pro dopamine regulation [42] and the Genetic Addiction Risk Score (GARS) test [43].
2.3. Gold's dopamine depletion hypothesis
Cocaine escalation for many years was considered to occur when dopamine increased in the reward circuitry of the brain. Indeed, the basis for all psychoactive substance abuse was the induction of neuronal dopamine release [51]. The conceptual framework behind the current list of FDA approved drugs for both SUD has been the idea of blocking increased dopaminergic function in the brain [52].
However, we must consider the comparison of dopamine surfeit to dopamine deficit theories. We have argued in favor of a dopamine deficit for the known escalation of, for example, cocaine [53]. Neuroimaging work favors dopamine agonist modalities rather than dopamine antagonist therapy for all RDS behaviors, including alcohol, opioids, and psychostimulants. The surfeit theory of stress also reduces dopamine in the reward circuitry [54].
In brief, the following studies by Gold's group reflect upon the neurological correlates of clinical aspects of drug-seeking, drug reinstatement, withdrawal symptomatology, relapse; psychostimulant dopaminergic agonist therapy; and prophylaxis, see Table 3.
2.4. Volkow's hypodopaminergia receptor theorem
Hypodopaminergia or dopamine depletion is a complicated but essential aspect of the genetic predisposition to reward-seeking behavior induced by chronic ethanol abuse [85]. Thousands of reports in the literature looked for SNPs of reward genes based on low-dopamine function. There are many notable examples of progress in the understanding of the neurochemical mechanisms involved in the addiction process, including withdrawal symptomatology [86]. Also understanding the physiological basis of neurotransmission [87]; the neurochemical mechanisms for synaptic function [88]; the role of long-term potentiation in drug self- administration and sensitization [89]; the mechanisms of pre and postsynaptic neurobiological storage, release, and catabolism of neurotransmitters [90]; the role of the” Brain Reward Cascade” in craving behavior and relapse [91]; and the role of neurogenetics and epigenetics in all aspects of drug-seeking and process addictions [92]. One study, in particular, shows that childhood maltreatment alters brain gray matter, which may induce relapse to psychoactive drugs later in life [93].
In brief, the following studies represent the work of Volkow's group primarily related to the utilization of positron emission tomography (PET) to map neurotransmitter receptors and investigate the neuro-mechanisms related to both drugs of abuse, obesity and reward circuitry, see Table 4.
Recently, through the work of a select few neuroscientists notably, Eric Nestler and his team, the newer understanding of how the environment may change phenotypic expression without actually altering DNA over two or more generations have been characterized.
2.5. Nestler's epigenetic theorem
Epigenetics is the study of heritable phenotypical changes that do not involve alterations in the DNA sequence [119]. The epigenetics features of RNA are situated above and in addition to the traditional genetic basis for inheritance DNA [120]. Epigenetics changes that effect gene activity and expression. These effects on physiological cellular phenotypic traits may result from external or environmental factors and be part of healthy development. Importantly, the standard definition of epigenetics requires that these alterations be heritable [121] either in the progeny of cells or organisms.
In brief, the following studies represent the work of Nestler's group, especially as it relates to the development of epigenetics linked to brain reward and addiction, see Table 5.
The “comments” in Table 1, Table 2, Table 3, Table 4, Table 5 are explanations of the relevance of each study to understanding the premises of “Theorems” or medical hypotheses deduced by an exploration of research topics and serves as the foundational neurobiological basis of addiction in these modern times.
3. Understanding endorphin deficiency syndrome and opioid deficiency syndrome
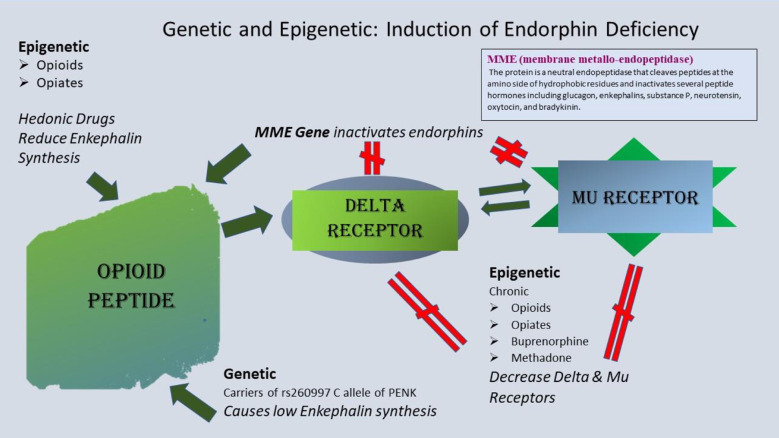
Induction of an Opioid Peptide Deficiency Syndrome may involve both genetic and epigenetic insults to the brain reward circuitry specific to opioid peptides (i.e., endorphins, enkephalins, and dynorphins). Genetic deficits may include polymorphisms in the Membrane metal- endopeptidase (MME), a carboxypeptidase enzyme, responsible for the inactivation of endogenous opioid peptides. Comings et al. [154] identified a dinucleotide polymorphism in the 5′ region of the MME gene that provides a high activity of this enzyme and subsequently reduced endogenous opioid peptides by substantial inactivation. The resultant general “hypo-opioidergia” or a specific “hypo-endorphinergia” function at both the delta or mu receptors and a ‘hypo-dynophinergia” at kappa receptors (not shown in Fig. 3). Also, at birth, an individual may carry the of the rs260997 C allele of Proenkephalin (PENK), which causes, for example, a significantly reduced enkephalin synthesis across the BRC [155]. Moreover, Niikura et al. [156] reported that there are epigenetic insults that reduce mRNA expression involving downregulation of mu receptor numbers or availability by chronic use/misuse of opiate analgesics like heroin or potent synthetic opioids like methadone, buprenorphine. Furthermore, Nylander et al. [157] showed a reduced leu-enkephalin in the VTA of Sprague Dawley rats with chronic morphine treatment.
Fig. 3.
Schematic of genetic and epigenetic induction of Opioid Peptide Deficiency Syndrome.
Induction of an Opioid Peptide Deficiency Syndrome (OPDS) may involve both genetic and epigenetic insults to the brain reward circuitry specific to opioid peptides (i.e., endorphins, enkephalins, and dynorphins). Genetic deficits may include polymorphisms in the Membrane metal- endopeptidase (MME), a carboxypeptidase enzyme, responsible for the inactivation of endogenous opioid peptides. Comings et al. identified a dinucleotide polymorphism in the 5′ region of the MME gene that provides a high activity of this enzyme and subsequent reduced endogenous opioid peptides by strong inactivation. Consequently, a general “hypoopioidergia” or a specific “hypoendorphinergia” function at both the delta or mu receptors and a ‘hypodynophinergia” at kappa receptors (not shown in Fig. 3) [red hatch sign]. Also, at birth, an individual may carry the of the rs260997 C allele of Proenkephalin (PENK) which causes, significantly reduced enkephalin synthesis across the BRC [red hatch sign]. Moreover, Niikura et al. reported that there are epigenetic insults that reduce mRNA expression involving downregulation of mu receptor numbers or availability by chronic use/misuse of opiate analgesics, like heroin, or powerful synthetic opioids like methadone, buprenorphine, [red hatch sign]. Furthermore, Nylander et al. showed a reduced leu-enkephalin in the VTA of Sprague Dawley rats with chronic morphine treatment [red hatch sign] The dark green arrows represent the normal flow.
Various human activities trigger the production of endorphins or enkephalins. Laughter is known to increased resistance to pain, stimulate endorphin production [158], and vigorous aerobic exercise releases β-endorphin [159,160]. Endorphins are released by pleasurable behaviors like eating chocolate, which contains TIQs, sex, orgasm, eating food, yoga, meditation, and listening to music [161,162].
Enkephalins are in high concentrations in the brain, especially in the hippocampus and prefrontal cortex. Stressors impact neuropeptides and their action in metabotropic enkephalin G-protein-coupled receptors, including delta-opioid and mu receptors [163,164]. Other endogenous opioid ligands such as dynorphins bind to kappa receptors, while endorphins per se bind to mu receptors [165,166].
Within the mesolimbic reward process, enkephalins inhibit the release of the neurotransmitter GABA when they bind to the μ-receptor to increase the production and release of dopamine [167]. Endorphin or opioid deficiency will result in a lowered inhibitory control over GABAergic inhibition, and thus through raphe nucleus interaction at the substania nigra –VTA junction reduces dopamine release at the NAc. The resultant hypodopaminergia will set people up for addiction liability.
It is well known now that the interaction of at least seven neurotransmitter systems (serotonin, opioid peptides, cannabinoids, GABA, Glutamate, acetylcholine, and dopamine) and associated second messengers play a role in reward processing in the brain of Homo sapiens. The Brain Reward Cascade (BRC) provides dopamine release illustrated in Fig. 1 is the basis of pleasures from everyday natural rewards. The suggestion is that balanced neurotransmission will animate feelings of wellbeing and reduced stress, while impairments in the BRC can result in a hypodopaminergia. For example, if an individual carries a polymorphism in the OPM1 (rs1799971 - A118G) and as such reduced function of this mu-opioid receptor. This functional reduction will result in an over-expression of GABAergic signaling. In turn, this excess GABA signaling will significantly reduce the net preferential-neuronal release of Dopamine at the NAc. Also, there could be epigenetically induced methylation that can cause a reduced mRNA expression for at least two generations [168]. Having an antecedent opioid/endorphin deficiency configures the individual with the potential to have uncontrolled dependence once exposed to a powerful opioid like Oxycontin. This risk reflected the challenges the entire scientific community has in the face of the worst opioid epidemic ever seen in America.
4. Summary
Medical health professionals have learned that opioid exposure as part of a pain treatment regimen for a wisdom tooth or a fracture can lead to a lifelong opioid use disorder. Genetic researchers wonder whether some people might have genetically fragile opioid systems that may implode when exposed to exogenous opioids. The genetic addiction risk score (GARS) is a predictor of vulnerability to opioid dependence [169]. These theorems argue that Endorphinergic Deficiency Syndrome, a subset of RDS [170], rather than generalized opioid deficiency syndrome, is the process underlying the opioid epidemic. Gold's work on concurrent inhibition of the nucleus locus coeruleus by the alpha-2 adrenergic systems provides for novel pharmacological therapies like clonidine and lofexidine [171], rapid and slow nonopioid detoxification and possible new treatments for craving and anxiety.
The current emphasis on opioid replacement in opioid use disorders (OUD) is a treatment based on a disease model that has not been proven. Oddly, the most efficacious treatments for OUDs are the treatments that stimulate compliance [172], are so closely related to opioid drugs of abuse, that the patient becomes addicted to the treatment, so discontinuation becomes unlikely [173]. The brain has not carved out an opiate (like codeine) or opioids (like buprenorphine) deficiency as a phenotypic target per se. However, methadone and buprenorphine treatments are the first options for most, if not all, patients with OUDs [174]. Like in the early days of methadone maintenance, opioid replacement is seen as insulin, might be, for a diabetic but provided in dose equivalents that are not related to endogenous opioids but rather saturation or blocking exogenous opioid reinforcement doses [175]. Dose escalation of the exogenous opioids provided as treatments for OUDs may further compromise the patients' ability to ever recover from an underlying Endorphinergic Deficiency Syndrome. Relatively opioid naïve OUD patients may acquire an iatrogenic endorphin deficiency. Exogenous opioids produce Dopamine Deficiency Syndrome (DDS), also a subset of RDS, depression, and anhedonia and may also be a cause of exogenous opioid seeking and abuse [176,177]. Most guidelines around the world recommend psychosocially assisted agonist maintenance therapy, and not medication alone as a treatment for OUD. This assistance has been difficult or impossible for many patients during the COVID 19 crisis. Many have dropped out of treatment, and others have tried telemedicine and technologically assisting behavioral therapies while continuing their MATs.
While escalating the dosing of prescribed opioids reduces further opioid seeking behavior in general, the fact of the matter is that subsequent urine analysis, even in a controlled environment, for example, residential and out-patient treatment centers reveal the opposite [173]. Two independent studies from our group have shown that urine drug testing revealed very high illicit drug abuse up to 49% even during treatment with maintenance drugs like methadone [178] and buprenorphine/naloxone [173]. Although MATs are safe and effective, early dropouts and discontinuations are common [172].
Opioid antagonist treatment currently is the least popular of the FDA approved treatments for OUD treatment. However, patients treated with naltrexone and long-acting injectable naltrexone may have advantages over those treated with agonists [179]. Long-term outcomes include evaluation of a full return to pre-OUD social, psychological, and intellectual function. Physician addicts [180], but not others with OUD, are preferentially treated with antagonists. Physicians with OUDs have the best five-year outcomes reported, including a return to the demanding work of physician practice without agonist MATs [181]. Business executives, union members, and others with active case management or employee assistance program may also have physician-like outcomes with antagonist treatment [182]. Treatment of OUDs with opioid agonists used for pain, cause complaints of attentional, cognitive, memory and affective impairments and are increasingly used with caution in these and other populations, except those with OUDs. Cognitive changes and performance are highlighted by Polles and her colleagues from the State of Florida's impaired physician program in this special volume. Transitioning from agonist to antagonist has been an obstacle for some but can be managed [183]. Physicians with OUD can achieve abstinence, long-term sobriety, return to pre-morbid function, life, work and retain free will to discontinue treatment. Craving and provoked craving may also be preferentially treated with antagonists. Also, Hill et al. showed in long-term SUBX patients a significantly flat affect (p < 0.01), and they had less self-awareness of being happy, sad, and anxious compared to both the GP and AA groups [184]. Depression is a central problem whether patients are chronically treated with high dose opioids for pain or OUDs.
We believe while opioid agonists are safe, effective, significantly reduce overdose, and are positive for harm reduction for many, the negatives are ignored because we have few alternatives and no non-addicting substitutes to help with both tertiary treatment and or prophylaxis [79].
The emerging evidence concerning the utility of the best practice to potentially prevent, treat, and reduce relapse involves the concept of precision addiction/behavioral management and concomitant induction of “dopamine homeostasis” [185]. The importance of opioid-free abstinence and return to pre-morbid function as outcomes are being debated by clinicians treating OUDs. Meanwhile, future access to personalized medicine, genetic testing and real-time imaging may help us understand for whom opioid exposure may produce a life-long opioid dysfunction—the relative benefits of agonist vs. antagonist OUD therapy for the particular neurobiology of the patient. The importance of understanding parallel systems in the neurobiology of signs and symptoms [55] as well as management of opioid-induced withdrawal and interventions as espoused by Gold and associates [23,71,171,[186], [187], [188]] provided the present “standard of care” in the addiction medicine space. The seminal work of Volkow's group utilizing brain PET scanning to provide a mirror to the actual function of neurotransmitters and associated receptors, especially in humans, has advanced our understanding of the underpinnings of addictive behaviors [94]. Finally, Nestler's group persuasively demonstrated how the environment through epigenetics impacts the genetic expression of many reward-based genes and acts over at least two generations to influence homeostatic mechanisms [149].
In terms of future research, we are encouraged that scientists will continue to discover more than one OUD, subtypes based on genetics and neurobiological system function. We will continue to explore these historical and timely concepts. This translational research will provide a novel pathway whereby each individual will be able to enjoy the inborn legacy of reward and pleasure without needing addictive substances or behaviors, without having to pay the cruel price of addiction and pain [189].
This historical review and framework emphasized the similarities in the theories that resulted from the work of four independent major scientific research groups carried out from the ‘70s until the present day. These researches having used various and many basic and clinical measures, remarkably support essentially the same view; that psychoactive substances misuse and non-substance addictive behaviors have a common mechanism, a deficiency in healthy neurotransmission of reward, dopamine depletion, mapped neurotransmission depletions, and dysfunction that have been identified as both genetic and epigenetic hypodopaminergia.
References
- 1.Berridge K.C., Robinson T.E. Liking, wanting, and the incentive-sensitization theory of addiction. Am. Psychol. 2016;71(8):670–679. doi: 10.1037/amp0000059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Kleykamp B.A., Weiss R.D., Strain E.C. Time to Reconsider the Role of Craving in Opioid Use Disorder. JAMA Psychiatry. 2019 Jul 24 doi: 10.1001/jamapsychiatry.2019.1839. [published online ahead of print] [DOI] [PubMed] [Google Scholar]
- 3.Myers R.D., Cicero T.J. Effects of serotonin depletion on the volitional alcohol intake of rats during a condition of psychological stress. Psychopharmacologia. 1969;15(5):373–381. doi: 10.1007/BF00403712. [DOI] [PubMed] [Google Scholar]
- 4.Smith D.E. The process addictions and the new ASAM definition of addiction. J. Psychoactive Drugs. 2012;44(1):1–4. doi: 10.1080/02791072.2012.662105. [DOI] [PubMed] [Google Scholar]
- 5.Geller I., Blum K. The effects of 5-HTP on Para-Chlorophenylalanine (p-CPA) attenuation of "conflict" behavior. Eur. J. Pharmacol. 1970;9(3):319–324. doi: 10.1016/0014-2999(70)90229-3. [DOI] [PubMed] [Google Scholar]
- 6.Reiter R.J., Blum K., Wallace J.E., Merritt J.H. Effect of the pineal gland on alcohol consumption by congenitally blind male rats. Q. J. Stud. Alcohol. 1973;34(3):937–939. [PubMed] [Google Scholar]
- 7.Geller I. Ethanol preference in the rat as a function of photoperiod. Science. 1971;173(3995):456–459. doi: 10.1126/science.173.3995.456. [DOI] [PubMed] [Google Scholar]
- 8.Blum K., Merritt J.H., Reiter R.J., Wallace J.E. A possible relationship between the pineal gland and ethanol preference in the rat. Curr. Ther. Res. Clin. Exp. 1973;15(1):25–30. [PubMed] [Google Scholar]
- 9.Reiter R.J., Blum K., Wallace J.E., Merritt J.H. Pineal gland: evidence for an influence on ethanol preference in male Syrian hamsters. Comp. Biochem. Physiol. A Comp. Physiol. 1974;47(1):11–16. doi: 10.1016/0300-9629(74)90045-0. [DOI] [PubMed] [Google Scholar]
- 10.Raiewski E.E., Elliott J.A., Evans J.A., Glickman G.L., Gorman M.R. Twice daily melatonin peaks in Siberian but not Syrian hamsters under 24 h light:dark:light:dark cycles. Chronobiol. Int. 2012;29(9):1206–1215. doi: 10.3109/07420528.2012.719965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Namboodiri M.A., Sugden D., Klein D.C., Tamarkin L., Mefford I.N. Serum melatonin and pineal indoleamine metabolism in a species with a small day/night N-acetyltransferase rhythm. Comp. Biochem. Physiol. B. 1985;80(4):731–736. doi: 10.1016/0305-0491(85)90453-5. [DOI] [PubMed] [Google Scholar]
- 12.Coon S.L., Munson P.J., Cherukuri P.F., Sugden D., Rath M.F., Moller M., et al. Circadian changes in long noncoding RNAs in the pineal gland. Proc. Natl. Acad. Sci. U. S. A. 2012;109(33):13319–13324. doi: 10.1073/pnas.1207748109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Davis V.E., Walsh M.J. Alcohol, amines, and alkaloids: a possible biochemical basis for alcohol addiction. Science. 1970;167(3920):1005–1007. doi: 10.1126/science.167.3920.1005. [DOI] [PubMed] [Google Scholar]
- 14.Cohen G., Collins M. Alkaloids from catecholamines in adrenal tissue: possible role in alcoholism. Science. 1970;167(3926):1749–1751. doi: 10.1126/science.167.3926.1749. [DOI] [PubMed] [Google Scholar]
- 15.Myers R.D. Isoquinolines, beta-carbolines and alcohol drinking: involvement of opioid and dopaminergic mechanisms. Experientia. 1989;45(5):436–443. doi: 10.1007/BF01952025. [DOI] [PubMed] [Google Scholar]
- 16.Hamilton M.G., Blum K., Hirst M. Identification of an isoquinoline alkaloid after chronic exposure to ethanol. Alcohol. Clin. Exp. Res. 1978;2(2):133–137. doi: 10.1111/j.1530-0277.1978.tb04713.x. [DOI] [PubMed] [Google Scholar]
- 17.Blum K., Hamilton M.G., Meyer E.K., Hirst M., Marshall A. Isoquinoline alkaloids as possible regulators of alcohol addiction. Lancet. 1977;1(8015):799–800. doi: 10.1016/s0140-6736(77)92981-6. [DOI] [PubMed] [Google Scholar]
- 18.Marshall A., Hirst M., Blum K. Analgesic effects of 3-carboxysalsolinol alone and in combination with morphine. Experientia. 1977;33(6):754–755. doi: 10.1007/BF01944172. [DOI] [PubMed] [Google Scholar]
- 19.Hamilton M.G., Hirst M., Blum K. Opiate-like activity of salsolinol on the electrically stimulated Guinea pig ileum. Life Sci. 1979;25(26):2205–2210. doi: 10.1016/0024-3205(79)90093-6. [DOI] [PubMed] [Google Scholar]
- 20.Blum K., DeLallo L., Briggs A.H., Hamilton M.G. Opioid responses of isoquinoline alkaloids (TIQs) Prog. Clin. Biol. Res. 1982;90:387–398. [PubMed] [Google Scholar]
- 21.Blum K. Narcotic antagonism of seizures induced by a dopamine-derived tetrahydroisoquinoline alkaloid. Experientia. 1988;44(9):751–753. doi: 10.1007/BF01959150. [DOI] [PubMed] [Google Scholar]
- 22.Blum K., Werner T., Carnes S., Carnes P., Bowirrat A., Giordano J., et al. Sex, drugs, and rock 'n' roll: hypothesizing common mesolimbic activation as a function of reward gene polymorphisms. J. Psychoactive Drugs. 2012;44(1):38–55. doi: 10.1080/02791072.2012.662112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Dackis C.A., Gold M.S. Pharmacological approaches to cocaine addiction. J. Subst. Abus. Treat. 1985;2(3):139–145. doi: 10.1016/0740-5472(85)90043-1. [DOI] [PubMed] [Google Scholar]
- 24.Wang G.J., Volkow N.D., Fowler J.S., Wolf A.P., MacGregor R.R., Shea C.E., et al. Comparison of two PET radioligands for imaging extrastriatal dopamine receptors in the human brain. Synapse (New York, NY) 1993;15(3):246–249. doi: 10.1002/syn.890150311. [DOI] [PubMed] [Google Scholar]
- 25.Boundy V.A., Gold S.J., Messer C.J., Chen J., Son J.H., Joh T.H., et al. Regulation of tyrosine hydroxylase promoter activity by chronic morphine in TH9.0-LacZ transgenic mice. J. Neurosci. 1998;18(23):9989–9995. doi: 10.1523/JNEUROSCI.18-23-09989.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Blum K., Hamilton M.G., JE W. In: Alcohol and Opiates: Neurochemical and Behavioral Mechanisms. Blum K., editor. Academic Press; New York, London: 1977. Alcohol and opiates: A review of common neurochemical and behavioral mechnisms. [Google Scholar]
- 27.Blum K., Wallace J.E., Eubanks J.D. Effects of naloxone on ethanol withdrawal: Preference and narcosis. Pharmacologist. 1975;17(197) [Google Scholar]
- 28.Blum K., Briggs A.H., Trachtenberg M.C., Delallo L., Wallace J.E. Enkephalinase inhibition: regulation of ethanol intake in genetically predisposed mice. Alcohol. 1987;4(6):449–456. doi: 10.1016/0741-8329(87)90084-x. [DOI] [PubMed] [Google Scholar]
- 29.Blum K., Calhoun W., Wallace J.E., Merritt J.H., Geller I. Soporific action of ethanol in mice: possible role of biogenic amines. Pharmacol. Biochem. Behav. 1973;1(3):271–276. doi: 10.1016/0091-3057(73)90116-0. [DOI] [PubMed] [Google Scholar]
- 30.Blum K., Wallace J.E., Calhoun W., Tabor R.G., Eubanks J.D. Ethanol narcosis in mice: serotonergic involvement. Experientia. 1974;30(9):1053–1054. doi: 10.1007/BF01939005. [DOI] [PubMed] [Google Scholar]
- 31.Blum K., Elston S.F., DeLallo L., Briggs A.H., Wallace J.E. Ethanol acceptance as a function of genotype amounts of brain [Met]enkephalin. Proc. Natl. Acad. Sci. U. S. A. 1983;80(21):6510–6512. doi: 10.1073/pnas.80.21.6510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Blum K., Trachtenberg M.C., Elliott C.E., Dingler M.L., Sexton R.L., Samuels A.I., et al. Enkephalinase inhibition and precursor amino acid loading improves inpatient treatment of alcohol and polydrug abusers: double-blind placebo-controlled study of the nutritional adjunct SAAVE. Alcohol. 1988;5(6):481–493. doi: 10.1016/0741-8329(88)90087-0. [DOI] [PubMed] [Google Scholar]
- 33.Blum K., Futterman S., Wallace J.E., Schwertner H.A. Naloxone-induced inhibition of ethanol dependence in mice. Nature. 1977;265(5589):49–51. doi: 10.1038/265049a0. [DOI] [PubMed] [Google Scholar]
- 34.Blum K., Briggs A.H., Elston S.F., DeLallo L., Sheridan P.J., Sar M. Reduced leucine-enkephalin--like immunoreactive substance in hamster basal ganglia after long-term ethanol exposure. Science. 1982;216(4553):1425–1427. doi: 10.1126/science.7089531. [DOI] [PubMed] [Google Scholar]
- 35.Blum K., Wallace J.E., Schwerter H.A., Eubanks J.D. Morphine suppression of ethanol withdrawal in mice. Experientia. 1976;32(1):79–82. doi: 10.1007/BF01932634. [DOI] [PubMed] [Google Scholar]
- 36.Blum K., Briggs A.H., Elston S.F., DeLallo L. Ethanol preference as a function of genotypic levels of whole brain enkephalin in mice. Toxicol. Eur. Res. 1981;3(5):261–262. [PubMed] [Google Scholar]
- 37.Trachtenberg M.C., Blum K. Alcohol and opioid peptides: neuropharmacological rationale for physical craving of alcohol. Am. J. Drug Alcohol Abuse. 1987;13(3):365–372. doi: 10.3109/00952998709001520. [DOI] [PubMed] [Google Scholar]
- 38.Blum K., Noble E.P., Sheridan P.J., Montgomery A., Ritchie T., Jagadeeswaran P., et al. Allelic association of human dopamine D2 receptor gene in alcoholism. Jama. 1990;263(15):2055–2060. [PubMed] [Google Scholar]
- 39.Noble E.P., Blum K., Ritchie T., Montgomery A., Sheridan P.J. Allelic association of the D2 dopamine receptor gene with receptor-binding characteristics in alcoholism. Arch. Gen. Psychiatry. 1991;48(7):648–654. doi: 10.1001/archpsyc.1991.01810310066012. [DOI] [PubMed] [Google Scholar]
- 40.Blum K., Sheridan P.J., Wood R.C., Braverman E.R., Chen T.J., Cull J.G., et al. The D2 dopamine receptor gene as a determinant of reward deficiency syndrome. J. R. Soc. Med. 1996;89(7):396–400. doi: 10.1177/014107689608900711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Blum K. In: The Sage Encyclopedia of Abnormal Clinical Psychology. Wenzel A., editor. Sage Publications; Pensylvania: 2017. Reward deficiency syndrome. [Google Scholar]
- 42.Blum K., Liu Y., Wang W., Wang Y., Zhang Y., Oscar-Berman M., et al. rsfMRI effects of KB220Z on neural pathways in reward circuitry of abstinent genotyped heroin addicts. Postgrad. Med. 2015;127(2):232–241. doi: 10.1080/00325481.2015.994879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Blum K., Oscar-Berman M., Demetrovics Z., Barh D., Gold M.S. Genetic addiction risk score (GARS): molecular neurogenetic evidence for predisposition to reward deficiency syndrome (RDS) Mol. Neurobiol. 2014;50(3):765–796. doi: 10.1007/s12035-014-8726-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Blum K., Downs B.W., Dushaj K., Li M., Braverman E.R., Fried L., et al. The benefits of customized DNA directed nutrition to balance the brain reward circuitry and reduce addictive behaviors. Precision Med. 2016;1(1):18–33. [PMC free article] [PubMed] [Google Scholar]
- 45.Febo M., Blum K., Badgaiyan R.D., Perez P.D., Colon-Perez L.M., Thanos P.K., et al. Enhanced functional connectivity and volume between cognitive and reward centers of naive rodent brain produced by pro-dopaminergic agent KB220Z. PLoS One. 2017;12(4) doi: 10.1371/journal.pone.0174774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Blum K., Oscar-Berman M., Femino J., Waite R.L., Benya L., Giordano J., et al. Withdrawal from buprenorphine/naloxone and maintenance with a natural dopaminergic agonist: a cautionary note. J. Addict. Res. Ther. 2013;4(2) doi: 10.4172/2155-6105.1000146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Blum K., Whitney D., Fried L., Febo M., Waite R.L., Braverman E.R., et al. Hypothesizing that a pro-dopaminergic regulator (KB220z™ Liquid Variant) can induce "dopamine homeostasis" and provide adjunctive detoxification benefits in opiate/opioid dependence. Clin. Med. Rev. Case Rep. 2016;3(8) doi: 10.23937/2378-3656/1410125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Blum K., Topel H. Opioid peptides and alcoholism: genetic deficiency and chemical management. Funct. Neurol. 1986;1(1):71–83. [PubMed] [Google Scholar]
- 49.Solanki N., Abijo T., Galvao C., Darius P., Blum K., Gondre-Lewis M.C. Administration of a putative pro-dopamine regulator, a neuronutrient, mitigates alcohol intake in alcohol-preferring rats. Behav. Brain Res. 2020;385:112563. doi: 10.1016/j.bbr.2020.112563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Blum K., Gondre-Lewis M.C., Baron D., Thanos P.K., Braverman E.R., Neary J., et al. Introducing precision addiction management of reward deficiency syndrome, the construct that underpins all addictive Behaviors. Front. Psychiatry. 2018;9:548. doi: 10.3389/fpsyt.2018.00548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Di Chiara G., Imperato A. Drugs abused by humans preferentially increase synaptic dopamine concentrations in the mesolimbic system of freely moving rats. Proc. Natl. Acad. Sci. U. S. A. 1988;85(14):5274–5278. doi: 10.1073/pnas.85.14.5274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Medication Assisted Treatment for Opioid Use Disorders . 2016. Final rule. Federal register; pp. 44711–44739. [PubMed] [Google Scholar]
- 53.Willuhn I., Burgeno L.M., Groblewski P.A., Phillips P.E. Excessive cocaine use results from decreased phasic dopamine signaling in the striatum. Nat. Neurosci. 2014;17(5):704–709. doi: 10.1038/nn.3694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Wise R.A., Koob G.F. The development and maintenance of drug addiction. Neuropsychopharmacol. 2014;39(2):254–262. doi: 10.1038/npp.2013.261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Gold M.S., Kleber H.D. A rationale for opiate withdrawal symptomatology. Drug Alcohol Depend. 1979;4(5):419–424. doi: 10.1016/0376-8716(79)90074-7. [DOI] [PubMed] [Google Scholar]
- 56.Gold M.S., Byck R., Sweeney D.R., Kleber H.D. Endorphin-locus coeruleus connection mediates opiate action and withdrawal. Biomedicine. 1979;30(1):1–4. [PubMed] [Google Scholar]
- 57.Gold M.S., Byck R. Endorphins, lithium, and naloxone: their relationship to pathological and drug-induced manic-euphoric states. NIDA Res. Monogr. 1978;19:192–209. [PubMed] [Google Scholar]
- 58.Gold M.S., Sweeney D.R., Pottash A.L., Kleber H.D. Decreased serum prolactin in opiate withdrawal and dopaminergic hyperactivity. Am. J. Psychiatry. 1979;136(6):849–850. doi: 10.1176/ajp.136.6.849. [DOI] [PubMed] [Google Scholar]
- 59.Dackis C.A., Gold M.S. New concepts in cocaine addiction: the dopamine depletion hypothesis. Neurosci. Biobehav. Rev. 1985;9(3):469–477. doi: 10.1016/0149-7634(85)90022-3. [DOI] [PubMed] [Google Scholar]
- 60.Dackis C.A., Gold M.S., Davies R.K., Sweeney D.R. Bromocriptine treatment for cocaine abuse: the dopamine depletion hypothesis. Int. J. Psychiatry Med. 1985;15(2):125–135. doi: 10.2190/gxpa-98ak-4rr4-hd9b. [DOI] [PubMed] [Google Scholar]
- 61.Gold M.S., Pottash A.L., Extein I., Kleber H.D. Anti-endorphin effects of methadone. Lancet. 1980;2(8201):972–973. doi: 10.1016/s0140-6736(80)92125-x. [DOI] [PubMed] [Google Scholar]
- 62.Extein I., Pottash A.L., Gold M.S. A possible opioid receptor dysfunction in some depressive disorders. Ann. N. Y. Acad. Sci. 1982;398:113–119. doi: 10.1111/j.1749-6632.1982.tb39484.x. [DOI] [PubMed] [Google Scholar]
- 63.Kleber H.D., Gold M.S., Riordan C.E. The use of clonidine in detoxification from opiates. Bull. Narc. 1980;32(2):1–10. [PubMed] [Google Scholar]
- 64.Neill D.B., Peay L.A., Gold M.S. Identification of a subregion within rat neostriatum for the dopaminergic modulation of lateral hypothalamic self-stimulation. Brain Res. 1978;153(3):515–528. doi: 10.1016/0006-8993(78)90336-0. [DOI] [PubMed] [Google Scholar]
- 65.Dackis C.A., Dackis M.A., Martin D., Pottash A.L., Gold M.S. Platelet serotonin transporter in cocaine patients. NIDA Res. Monogr. 1984;55:164–169. [PubMed] [Google Scholar]
- 66.Byars J.A., Frost-Pineda K., Jacobs W.S., Gold M.S. Naltrexone augments the effects of nicotine replacement therapy in female smokers. J. Addict. Dis. 2005;24(2):49–60. doi: 10.1300/J069v24n02_05. [DOI] [PubMed] [Google Scholar]
- 67.Miller N.S., Gold M.S., Belkin B.M., Klahr A.L. Family history and diagnosis of alcohol dependence in cocaine dependence. Psychiatry Res. 1989;29(2):113–121. doi: 10.1016/0165-1781(89)90026-7. [DOI] [PubMed] [Google Scholar]
- 68.Blum K., Liu Y., Shriner R., Gold M.S. Reward circuitry dopaminergic activation regulates food and drug craving behavior. Curr. Pharm. Des. 2011;17(12):1158–1167. doi: 10.2174/138161211795656819. [DOI] [PubMed] [Google Scholar]
- 69.Liu J., Liang J., Qin W., Tian J., Yuan K., Bai L., et al. Dysfunctional connectivity patterns in chronic heroin users: an fMRI study. Neurosci. Lett. 2009;460(1):72–77. doi: 10.1016/j.neulet.2009.05.038. [DOI] [PubMed] [Google Scholar]
- 70.Yuan K., Qin W., Dong M., Liu J., Sun J., Liu P., et al. Gray matter deficits and resting-state abnormalities in abstinent heroin-dependent individuals. Neurosci. Lett. 2010;482(2):101–105. doi: 10.1016/j.neulet.2010.07.005. [DOI] [PubMed] [Google Scholar]
- 71.Bruijnzeel A.W., Marcinkiewcz C., Isaac S., Booth M.M., Dennis D.M., Gold M.S. The effects of buprenorphine on fentanyl withdrawal in rats. Psychopharmacology. 2007;191(4):931–941. doi: 10.1007/s00213-006-0670-2. [DOI] [PubMed] [Google Scholar]
- 72.Cocores J.A., Gold M.S. The salted food addiction hypothesis may explain overeating and the obesity epidemic. Med. Hypotheses. 2009;73(6):892–899. doi: 10.1016/j.mehy.2009.06.049. [DOI] [PubMed] [Google Scholar]
- 73.Zhang Y., Tian J., Yuan K., Liu P., Zhuo L., Qin W., et al. Distinct resting-state brain activities in heroin-dependent individuals. Brain Res. 2011;1402:46–53. doi: 10.1016/j.brainres.2011.05.054. [DOI] [PubMed] [Google Scholar]
- 74.Kleiner K.D., Gold M.S., Frost-Pineda K., Lenz-Brunsman B., Perri M.G., Jacobs W.S. Body mass index and alcohol use. J. Addict. Dis. 2004;23(3):105–118. doi: 10.1300/J069v23n03_08. [DOI] [PubMed] [Google Scholar]
- 75.Jiang M., Gold M.S., Boulay G., Spicher K., Peyton M., Brabet P., et al. Multiple neurological abnormalities in mice deficient in the G protein go. Proc. Natl. Acad. Sci. U. S. A. 1998;95(6):3269–3274. doi: 10.1073/pnas.95.6.3269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Blum K., Gold M.S. Neuro-chemical activation of brain reward meso-limbic circuitry is associated with relapse prevention and drug hunger: a hypothesis. Med. Hypotheses. 2011;76(4):576–584. doi: 10.1016/j.mehy.2011.01.005. [DOI] [PubMed] [Google Scholar]
- 77.Warren M.W., Gold M.S. The relationship between obesity and drug use. Am. J. Psychiatry. 2007;164(8):1268. doi: 10.1176/appi.ajp.2007.07030388. author reply −9. [DOI] [PubMed] [Google Scholar]
- 78.Gold M.S. Na(+) channel blockers for the treatment of pain: context is everything, almost. Exp. Neurol. 2008;210(1):1–6. doi: 10.1016/j.expneurol.2007.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Baron D., Blum K., Chen A., Gold M., Badgaiyan R.D. Conceptualizing addiction from an osteopathic perspective: dopamine homeostasis. J. Am. Osteopathic Assoc. 2018;118(2):115–118. doi: 10.7556/jaoa.2018.026. [DOI] [PubMed] [Google Scholar]
- 80.Blum K., Thanos P.K., Wang G.J., Febo M., Demetrovics Z., Modestino E.J., et al. The food and drug addiction epidemic: targeting dopamine homeostasis. Curr. Pharm. Des. 2018;23(39):6050–6061. doi: 10.2174/1381612823666170823101713. [DOI] [PubMed] [Google Scholar]
- 81.Sekine Y., Ouchi Y., Sugihara G., Takei N., Yoshikawa E., Nakamura K., et al. Methamphetamine causes microglial activation in the brains of human abusers. J. Neurosci. 2008;28(22):5756–5761. doi: 10.1523/JNEUROSCI.1179-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Gold M.S., Kobeissy F.H., Wang K.K., Merlo L.J., Bruijnzeel A.W., Krasnova I.N., et al. Methamphetamine- and trauma-induced brain injuries: comparative cellular and molecular neurobiological substrates. Biol. Psychiatry. 2009;66(2):118–127. doi: 10.1016/j.biopsych.2009.02.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Dackis C.A., Gold M.S. In: Substance Abuse and Psychopathology. Mirin S.M., editor. American Psychiatric Press; Arlington, VA: 1984. Depression in opiate addicts; pp. 20–39. [Google Scholar]
- 84.Gold M.S., Pottash A.C., Extein I., Martin D., Kleber H.D. Methadone-induced endorphin dysfunction in addicts. NIDA Res. Monogr. 1982;41:476–482. [PubMed] [Google Scholar]
- 85.Siciliano C.A., Calipari E.S., Yorgason J.T., Lovinger D.M., Mateo Y., Jimenez V.A., et al. Increased presynaptic regulation of dopamine neurotransmission in the nucleus accumbens core following chronic ethanol self-administration in female macaques. Psychopharmacology. 2016;233(8):1435–1443. doi: 10.1007/s00213-016-4239-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Zandy S.L., Matthews D.B., Tokunaga S., Miller A., Blaha C.D., Mittleman G. Reduced dopamine release in the nucleus accumbens core of adult rats following adolescent binge alcohol exposure: age and dose-dependent analysis. Psychopharmacology. 2015;232(4):777–784. doi: 10.1007/s00213-014-3712-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Williams C.L., Buchta W.C., Riegel A.C. CRF-R2 and the heterosynaptic regulation of VTA glutamate during reinstatement of cocaine seeking. J. Neurosci. 2014;34(31):10402–10414. doi: 10.1523/JNEUROSCI.0911-13.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Talani G., Licheri V., Masala N., Follesa P., Mostallino M.C., Biggio G., et al. Increased voluntary ethanol consumption and changes in hippocampal synaptic plasticity in isolated C57BL/6J mice. Neurochem. Res. 2014;39(6):997–1004. doi: 10.1007/s11064-013-1216-8. [DOI] [PubMed] [Google Scholar]
- 89.Polter A.M., Bishop R.A., Briand L.A., Graziane N.M., Pierce R.C., Kauer J.A. Poststress block of kappa opioid receptors rescues long-term potentiation of inhibitory synapses and prevents reinstatement of cocaine seeking. Biol. Psychiatry. 2014;76(10):785–793. doi: 10.1016/j.biopsych.2014.04.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Palm S., Nylander I. Dopamine release dynamics change during adolescence and after voluntary alcohol intake. PLoS One. 2014;9(5) doi: 10.1371/journal.pone.0096337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Chen K.C., Lin Y.C., Chao W.C., Chung H.K., Chi S.S., Liu W.S., et al. Association of genetic polymorphisms of glutamate decarboxylase 2 and the dopamine D2 receptor with obesity in Taiwanese subjects. Ann. Saudi Med. 2012;32(2):121–126. doi: 10.5144/0256-4947.2012.121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Starkman B.G., Sakharkar A.J., Pandey S.C. Epigenetics-beyond the genome in alcoholism. Alcohol Res. 2012;34(3):293–305. doi: 10.35946/arcr.v34.3.04. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Van Dam N.T., Rando K., Potenza M.N., Tuit K., Sinha R. Childhood maltreatment, altered limbic neurobiology, and substance use relapse severity via trauma-specific reductions in limbic gray matter volume. JAMA Psychiatry. 2014;71(8):917–925. doi: 10.1001/jamapsychiatry.2014.680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Volkow N.D., Tancredi L.R. Biological correlates of mental activity studied with PET. Am. J. Psychiatry. 1991;148(4):439–443. doi: 10.1176/ajp.148.4.439. [DOI] [PubMed] [Google Scholar]
- 95.Dewey S.L., Logan J., Wolf A.P., Brodie J.D., Angrist B., Fowler J.S., et al. Amphetamine induced decreases in (18F)-N-methylspiroperidol binding in the baboon brain using positron emission tomography (PET) Synapse (New York, NY) 1991;7(4):324–327. doi: 10.1002/syn.890070409. [DOI] [PubMed] [Google Scholar]
- 96.Volkow N.D., Fowler J.S., Wolf A.P., Hitzemann R., Dewey S., Bendriem B., et al. Changes in brain glucose metabolism in cocaine dependence and withdrawal. Am. J. Psychiatry. 1991;148(5):621–626. doi: 10.1176/ajp.148.5.621. [DOI] [PubMed] [Google Scholar]
- 97.Wang G.J., Volkow N.D., Fowler J.S., Logan J., Gur R., Netusil N., et al. Age associated decrements in dopamine D2 receptors in thalamus and in temporal insula of human subjects. Life Sci. 1996;59(1):Pl31–5. doi: 10.1016/0024-3205(96)00262-7. [DOI] [PubMed] [Google Scholar]
- 98.Volkow N.D., Mullani N., Gould L., Adler S.S., Guynn R.W., Overall J.E., et al. Effects of acute alcohol intoxication on cerebral blood flow measured with PET. Psychiatry Res. 1988;24(2):201–209. doi: 10.1016/0165-1781(88)90063-7. [DOI] [PubMed] [Google Scholar]
- 99.Volkow N.D., Wang G.J., Fowler J.S., Franceschi D., Thanos P.K., Wong C., et al. Cocaine abusers show a blunted response to alcohol intoxication in limbic brain regions. Life Sci. 2000;66(12):Pl161–7. doi: 10.1016/s0024-3205(00)00421-5. [DOI] [PubMed] [Google Scholar]
- 100.Volkow N.D., Wang G.J., Fowler J.S., Logan J., Gatley S.J., Wong C., et al. Reinforcing effects of psychostimulants in humans are associated with increases in brain dopamine and occupancy of D(2) receptors. J. Pharmacol. Exp. Ther. 1999;291(1):409–415. [PubMed] [Google Scholar]
- 101.Smith G.S., Dewey S.L., Brodie J.D., Logan J., Vitkun S.A., Simkowitz P., et al. Serotonergic modulation of dopamine measured with [11C]raclopride and PET in normal human subjects. Am. J. Psychiatry. 1997;154(4):490–496. doi: 10.1176/ajp.154.4.490. [DOI] [PubMed] [Google Scholar]
- 102.Wang G.J., Volkow N.D., Logan J., Pappas N.R., Wong C.T., Zhu W., et al. Brain dopamine and obesity. Lancet. 2001;357(9253):354–357. doi: 10.1016/s0140-6736(00)03643-6. [DOI] [PubMed] [Google Scholar]
- 103.Ding Y.S., Logan J., Bermel R., Garza V., Rice O., Fowler J.S., et al. Dopamine receptor-mediated regulation of striatal cholinergic activity: positron emission tomography studies with norchloro[18F]fluoroepibatidine. J. Neurochem. 2000;74(4):1514–1521. doi: 10.1046/j.1471-4159.2000.0741514.x. [DOI] [PubMed] [Google Scholar]
- 104.Ding Y.S., Gatley S.J., Fowler J.S., Chen R., Volkow N.D., Logan J., et al. Mapping catechol-O-methyltransferase in vivo: initial studies with [18F]Ro41-0960. Life Sci. 1996;58(3):195–208. doi: 10.1016/0024-3205(95)02277-5. [DOI] [PubMed] [Google Scholar]
- 105.Wang G.J., Volkow N.D., Fowler J.S., Franceschi D., Logan J., Pappas N.R., et al. PET studies of the effects of aerobic exercise on human striatal dopamine release. J. Nucl. Med. 2000;41(8):1352–1356. [PubMed] [Google Scholar]
- 106.Volkow N.D., Wang G.J., Fowler J.S., Logan J., Jayne M., Franceschi D., et al. "Nonhedonic" food motivation in humans involves dopamine in the dorsal striatum and methylphenidate amplifies this effect. Synapse (New York, NY) 2002;44(3):175–180. doi: 10.1002/syn.10075. [DOI] [PubMed] [Google Scholar]
- 107.Cruz S.L., Villarreal J.E., Volkow N.D. Further evidence that naloxone acts as an inverse opiate agonist: implications for drug dependence and withdrawal. Life Sci. 1996;58(26):Pl381–9. doi: 10.1016/0024-3205(96)00250-0. [DOI] [PubMed] [Google Scholar]
- 108.Wang G.J., Volkow N.D., Fowler J.S., Logan J., Abumrad N.N., Hitzemann R.J., et al. Dopamine D2 receptor availability in opiate-dependent subjects before and after naloxone-precipitated withdrawal. Neuropsychopharmacol. 1997;16(2):174–182. doi: 10.1016/S0893-133X(96)00184-4. [DOI] [PubMed] [Google Scholar]
- 109.Li G., Ji G., Hu Y., Xu M., Jin Q., Liu L., et al. Bariatric surgery in obese patients reduced resting connectivity of brain regions involved with self-referential processing. Hum. Brain Mapp. 2018;39(12):4755–4765. doi: 10.1002/hbm.24320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Dewey S.L., Chaurasia C.S., Chen C.E., Volkow N.D., Clarkson F.A., Porter S.P., et al. GABAergic attenuation of cocaine-induced dopamine release and locomotor activity. Synapse (New York, NY) 1997;25(4):393–398. doi: 10.1002/(SICI)1098-2396(199704)25:4<393::AID-SYN11>3.0.CO;2-W. [DOI] [PubMed] [Google Scholar]
- 111.Bloomfield M.A., Ashok A.H., Volkow N.D., Howes O.D. The effects of Delta(9)-tetrahydrocannabinol on the dopamine system. Nature. 2016;539(7629):369–377. doi: 10.1038/nature20153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Volkow N.D., McLellan A.T. Opioid abuse in chronic pain--misconceptions and mitigation strategies. N. Engl. J. Med. 2016;374(13):1253–1263. doi: 10.1056/NEJMra1507771. [DOI] [PubMed] [Google Scholar]
- 113.Volkow N.D., Frieden T.R., Hyde P.S., Cha S.S. Medication-assisted therapies--tackling the opioid-overdose epidemic. N. Engl. J. Med. 2014;370(22):2063–2066. doi: 10.1056/NEJMp1402780. [DOI] [PubMed] [Google Scholar]
- 114.Valentino R.J., Volkow N.D. Untangling the complexity of opioid receptor function. Neuropsychopharmacol. 2018;43(13):2514–2520. doi: 10.1038/s41386-018-0225-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Wiers C.E., Towb P.C., Hodgkinson C.A., Shen P.H., Freeman C., Miller G., et al. Association of genetic ancestry with striatal dopamine D2/D3 receptor availability. Mol. Psychiatry. 2018;23(8):1711–1716. doi: 10.1038/mp.2017.208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Volkow N.D., Baler R.D. NOW vs LATER brain circuits: implications for obesity and addiction. Trends Neurosci. 2015;38(6):345–352. doi: 10.1016/j.tins.2015.04.002. [DOI] [PubMed] [Google Scholar]
- 117.Wang G.J., Zhao J., Tomasi D., Kojori E.S., Wang R., Wiers C.E., et al. Effect of combined naltrexone and bupropion therapy on the brain's functional connectivity. Int. J. Obes. 2005;42(11):1890–1899. doi: 10.1038/s41366-018-0040-2. 2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Anderson S.A., Michaelides M., Zarnegar P., Ren Y., Fagergren P., Thanos P.K., et al. Impaired periamygdaloid-cortex prodynorphin is characteristic of opiate addiction and depression. J. Clin. Invest. 2013;123(12):5334–5341. doi: 10.1172/JCI70395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Dupont C., Armant D.R., Brenner C.A. Epigenetics: definition, mechanisms and clinical perspective. Semin. Reprod. Med. 2009;27(5):351–357. doi: 10.1055/s-0029-1237423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Ledford H. Language: disputed definitions. Nature. 2008;455(7216):1023–1028. doi: 10.1038/4551023a. [DOI] [PubMed] [Google Scholar]
- 121.Berger S.L., Kouzarides T., Shiekhattar R., Shilatifard A. An operational definition of epigenetics. Genes Dev. 2009;23(7):781–783. doi: 10.1101/gad.1787609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Aghajanian G.K., Alreja M., Nestler E.J., Kogan J.H. Dual mechanisms of opiate dependence in the locus coeruleus. Clin. Neuropharmacol. 1992;15(Suppl. 1) doi: 10.1097/00002826-199201001-00077. Pt A:143a-4a. [DOI] [PubMed] [Google Scholar]
- 123.Beitner-Johnson D., Guitart X., Nestler E.J. Common intracellular actions of chronic morphine and cocaine in dopaminergic brain reward regions. Ann. N. Y. Acad. Sci. 1992;654:70–87. doi: 10.1111/j.1749-6632.1992.tb25957.x. [DOI] [PubMed] [Google Scholar]
- 124.Berhow M.T., Russell D.S., Terwilliger R.Z., Beitner-Johnson D., Self D.W., Lindsay R.M., et al. Influence of neurotrophic factors on morphine- and cocaine-induced biochemical changes in the mesolimbic dopamine system. Neuroscience. 1995;68(4):969–979. doi: 10.1016/0306-4522(95)00207-y. [DOI] [PubMed] [Google Scholar]
- 125.Trevisan L., Fitzgerald L.W., Brose N., Gasic G.P., Heinemann S.F., Duman R.S., et al. Chronic ingestion of ethanol up-regulates NMDAR1 receptor subunit immunoreactivity in rat hippocampus. J. Neurochem. 1994;62(4):1635–1638. doi: 10.1046/j.1471-4159.1994.62041635.x. [DOI] [PubMed] [Google Scholar]
- 126.Beitner D.B., Duman R.S., Nestler E.J. A novel action of morphine in the rat locus coeruleus: persistent decrease in adenylate cyclase. Mol. Pharmacol. 1989;35(5):559–564. [PubMed] [Google Scholar]
- 127.Kosten T.A., Marby D.W., Nestler E.J. Cocaine conditioned place preference is attenuated by chronic buprenorphine treatment. Life Sci. 1991;49(24):Pl201–6. doi: 10.1016/0024-3205(91)90490-3. [DOI] [PubMed] [Google Scholar]
- 128.Charlton M.E., Sweetnam P.M., Fitzgerald L.W., Terwilliger R.Z., Nestler E.J., Duman R.S. Chronic ethanol administration regulates the expression of GABAA receptor alpha 1 and alpha 5 subunits in the ventral tegmental area and hippocampus. J. Neurochem. 1997;68(1):121–127. doi: 10.1046/j.1471-4159.1997.68010121.x. [DOI] [PubMed] [Google Scholar]
- 129.Hope B., Kosofsky B., Hyman S.E., Nestler E.J. Regulation of immediate early gene expression and AP-1 binding in the rat nucleus accumbens by chronic cocaine. Proc. Natl. Acad. Sci. U. S. A. 1992;89(13):5764–5768. doi: 10.1073/pnas.89.13.5764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Nestler E.J., Guitart X., Ortiz J., Trevisan L. Second messenger and protein phosphorylation mechanisms underlying possible genetic vulnerability to alcoholism. Ann. N. Y. Acad. Sci. 1994;708:108–118. doi: 10.1111/j.1749-6632.1994.tb24703.x. [DOI] [PubMed] [Google Scholar]
- 131.Nye H.E., Nestler E.J. Induction of chronic Fos-related antigens in rat brain by chronic morphine administration. Mol. Pharmacol. 1996;49(4):636–645. [PubMed] [Google Scholar]
- 132.Beitner-Johnson D., Nestler E.J. Chronic morphine impairs axoplasmic transport in the rat mesolimbic dopamine system. Neuroreport. 1993;5(1):57–60. doi: 10.1097/00001756-199310000-00014. [DOI] [PubMed] [Google Scholar]
- 133.Rasmussen K., Beitner-Johnson D.B., Krystal J.H., Aghajanian G.K., Nestler E.J. Opiate withdrawal and the rat locus coeruleus: behavioral, electrophysiological, and biochemical correlates. J. Neurosci. 1990;10(7):2308–2317. doi: 10.1523/JNEUROSCI.10-07-02308.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Olson V.G., Green T.A., Neve R.L., Nestler E.J. Regulation of morphine reward and feeding by CREB in the lateral hypothalamus. Synapse (New York, NY). 2007;61(2):110–113. doi: 10.1002/syn.20344. [DOI] [PubMed] [Google Scholar]
- 135.Kim K.S., Lee K.W., Baek I.S., Lim C.M., Krishnan V., Lee J.K., et al. Adenylyl cyclase-5 activity in the nucleus accumbens regulates anxiety-related behavior. J. Neurochem. 2008;107(1):105–115. doi: 10.1111/j.1471-4159.2008.05592.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Renthal W., Maze I., Krishnan V., Covington H.E., 3rd, Xiao G., Kumar A., et al. Histone deacetylase 5 epigenetically controls behavioral adaptations to chronic emotional stimuli. Neuron. 2007;56(3):517–529. doi: 10.1016/j.neuron.2007.09.032. [DOI] [PubMed] [Google Scholar]
- 137.Perrotti L.I., Hadeishi Y., Ulery P.G., Barrot M., Monteggia L., Duman R.S., et al. Induction of deltaFosB in reward-related brain structures after chronic stress. J. Neurosci. 2004;24(47):10594–10602. doi: 10.1523/JNEUROSCI.2542-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Scott M.M., Marcus J.N., Pettersen A., Birnbaum S.G., Mochizuki T., Scammell T.E., et al. Hcrtr1 and 2 signaling differentially regulates depression-like behaviors. Behav. Brain Res. 2011;222(2):289–294. doi: 10.1016/j.bbr.2011.02.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Zachariou V., Georgescu D., Sanchez N., Rahman Z., DiLeone R., Berton O., et al. Essential role for RGS9 in opiate action. Proc. Natl. Acad. Sci. U. S. A. 2003;100(23):13656–13661. doi: 10.1073/pnas.2232594100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Newton S.S., Thome J., Wallace T.L., Shirayama Y., Schlesinger L., Sakai N., et al. Inhibition of cAMP response element-binding protein or dynorphin in the nucleus accumbens produces an antidepressant-like effect. J. Neurosci. 2002;22(24):10883–10890. doi: 10.1523/JNEUROSCI.22-24-10883.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Nestler E.J., Pena C.J., Kundakovic M., Mitchell A., Akbarian S. Epigenetic basis of mental illness. Neuroscientist. 2016;22(5):447–463. doi: 10.1177/1073858415608147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Descalzi G., Ikegami D., Ushijima T., Nestler E.J., Zachariou V., Narita M. Epigenetic mechanisms of chronic pain. Trends Neurosci. 2015;38(4):237–246. doi: 10.1016/j.tins.2015.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Nestler E.J. Epigenetic mechanisms of drug addiction. Neuropharmacology. 2014;76(Pt B):259–268. doi: 10.1016/j.neuropharm.2013.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Hamilton P.J., Burek D.J., Lombroso S.I., Neve R.L., Robison A.J., Nestler E.J., et al. Cell-type-specific epigenetic editing at the Fosb gene controls susceptibility to social defeat stress. Neuropsychopharmacol. 2018;43(2):272–284. doi: 10.1038/npp.2017.88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Nestler E.J., Carlezon W.A., Jr. The mesolimbic dopamine reward circuit in depression. Biol. Psychiatry. 2006;59(12):1151–1159. doi: 10.1016/j.biopsych.2005.09.018. [DOI] [PubMed] [Google Scholar]
- 146.Massart R., Barnea R., Dikshtein Y., Suderman M., Meir O., Hallett M., et al. Role of DNA methylation in the nucleus accumbens in incubation of cocaine craving. J. Neurosci. 2015;35(21):8042–8058. doi: 10.1523/JNEUROSCI.3053-14.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Kennedy P.J., Feng J., Robison A.J., Maze I., Badimon A., Mouzon E., et al. Class I HDAC inhibition blocks cocaine-induced plasticity by targeted changes in histone methylation. Nat. Neurosci. 2013;16(4):434–440. doi: 10.1038/nn.3354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Cahill M.E., Browne C.J., Wang J., Hamilton P.J., Dong Y., Nestler E.J. Withdrawal from repeated morphine administration augments expression of the RhoA network in the nucleus accumbens to control synaptic structure. J. Neurochem. 2018;147(1):84–98. doi: 10.1111/jnc.14563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Damez-Werno D.M., Sun H., Scobie K.N., Shao N., Rabkin J., Dias C., et al. Histone arginine methylation in cocaine action in the nucleus accumbens. Proc. Natl. Acad. Sci. U. S. A. 2016;113(34):9623–9628. doi: 10.1073/pnas.1605045113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Renthal W., Nestler E.J. Histone acetylation in drug addiction. Semin. Cell Dev. Biol. 2009;20(4):387–394. doi: 10.1016/j.semcdb.2009.01.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Lobo M.K., Nestler E.J. The striatal balancing act in drug addiction: distinct roles of direct and indirect pathway medium spiny neurons. Front. Neuroanat. 2011;5:41. doi: 10.3389/fnana.2011.00041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Koo J.W., Mazei-Robison M.S., LaPlant Q., Egervari G., Braunscheidel K.M., Adank D.N., et al. Epigenetic basis of opiate suppression of Bdnf gene expression in the ventral tegmental area. Nat. Neurosci. 2015;18(3):415–422. doi: 10.1038/nn.3932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Tomasiewicz H.C., Jacobs M.M., Wilkinson M.B., Wilson S.P., Nestler E.J., Hurd Y.L. Proenkephalin mediates the enduring effects of adolescent cannabis exposure associated with adult opiate vulnerability. Biol. Psychiatry. 2012;72(10):803–810. doi: 10.1016/j.biopsych.2012.04.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Comings D.E., Dietz G., Gade-Andavolu R., Blake H., Muhleman D., Huss M., et al. Association of the neutral endopeptidase (MME) gene with anxiety. Psychiatr. Genet. 2000;10(2):91–94. doi: 10.1097/00041444-200010020-00007. [DOI] [PubMed] [Google Scholar]
- 155.Comings D.E., Blake H., Dietz G., Gade-Andavolu R., Legro R.S., Saucier G., et al. The proenkephalin gene (PENK) and opioid dependence. Neuroreport. 1999;10(5):1133–1135. doi: 10.1097/00001756-199904060-00042. [DOI] [PubMed] [Google Scholar]
- 156.Niikura K., Narita M., Butelman E.R., Kreek M.J., Suzuki T. Neuropathic and chronic pain stimuli downregulate central mu-opioid and dopaminergic transmission. Trends Pharmacol. Sci. 2010;31(7):299–305. doi: 10.1016/j.tips.2010.04.003. [DOI] [PubMed] [Google Scholar]
- 157.Nylander I., Vlaskovska M., Terenius L. The effects of morphine treatment and morphine withdrawal on the dynorphin and enkephalin systems in Sprague-Dawley rats. Psychopharmacology. 1995;118(4):391–400. doi: 10.1007/BF02245939. [DOI] [PubMed] [Google Scholar]
- 158.Manninen S., Tuominen L., Dunbar R.I., Karjalainen T., Hirvonen J., Arponen E., et al. Social laughter triggers endogenous opioid release in humans. J. Neurosci. 2017;37(25):6125–6131. doi: 10.1523/JNEUROSCI.0688-16.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Kami K., Tajima F., Senba E. Exercise-induced hypoalgesia: potential mechanisms in animal models of neuropathic pain. Anat. Sci. Int. 2017;92(1):79–90. doi: 10.1007/s12565-016-0360-z. [DOI] [PubMed] [Google Scholar]
- 160.Hicks S.D., Jacob P., Perez O., Baffuto M., Gagnon Z., Middleton F.A. The transcriptional signature of a Runner's high. Med. Sci. Sports Exerc. 2019;51(5):970–978. doi: 10.1249/MSS.0000000000001865. [DOI] [PubMed] [Google Scholar]
- 161.Tuenter E., Foubert K., Pieters L. Mood components in cocoa and chocolate: the mood pyramid. Planta Med. 2018;84(12−13):839–844. doi: 10.1055/a-0588-5534. [DOI] [PubMed] [Google Scholar]
- 162.Blum K., Simpatico T., Febo M., Rodriquez C., Dushaj K., Li M., et al. Hypothesizing music intervention enhances brain functional connectivity involving dopaminergic recruitment: common neuro-correlates to abusable drugs. Mol. Neurobiol. 2017;54(5):3753–3758. doi: 10.1007/s12035-016-9934-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Henry M.S., Gendron L., Tremblay M.E., Drolet G. Enkephalins: endogenous analgesics with an emerging role in stress resilience. Neural. Plast. 2017;2017:1546125. doi: 10.1155/2017/1546125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Mao L., Wang J.Q. Selective activation of group I metabotropic glutamate receptors upregulates preprodynorphin, substance P, and preproenkephalin mRNA expression in rat dorsal striatum. Synapse (New York, NY). 2001;39(1):82–94. doi: 10.1002/1098-2396(20010101)39:1<82::AID-SYN11>3.0.CO;2-B. [DOI] [PubMed] [Google Scholar]
- 165.Opioid peptides: molecular pharmacology, biosynthesis, and analysisNIDA Res. Monogr. 1986;70:1–414. [PubMed] [Google Scholar]
- 166.Wagner J.J., Evans C.J., Chavkin C. Focal stimulation of the mossy fibers releases endogenous dynorphins that bind kappa 1-opioid receptors in Guinea pig hippocampus. J. Neurochem. 1991;57(1):333–343. doi: 10.1111/j.1471-4159.1991.tb02132.x. [DOI] [PubMed] [Google Scholar]
- 167.Watanabe Y., Aono Y., Komiya M., Waddington J.L., Saigusa T. Stimulation of accumbal GABA(B) receptors inhibits delta1- and delta2-opioid receptor-mediated dopamine efflux in the nucleus accumbens of freely moving rats. Eur. J. Pharmacol. 2018;837:88–95. doi: 10.1016/j.ejphar.2018.08.003. [DOI] [PubMed] [Google Scholar]
- 168.Szutorisz H., Hurd Y.L. Epigenetic effects of cannabis exposure. Biol. Psychiatry. 2016;79(7):586–594. doi: 10.1016/j.biopsych.2015.09.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Blum K., Chen A.L.C., Thanos P.K., Febo M., Demetrovics Z., Dushaj K., et al. Genetic addiction risk score (GARS) ™, a predictor of vulnerability to opioid dependence. Front. Biosci. (Elite edition). 2018;10:175–196. doi: 10.2741/e816. [DOI] [PubMed] [Google Scholar]
- 170.Borsook D., Linnman C., Faria V., Strassman A.M., Becerra L., Elman I. Reward deficiency and anti-reward in pain chronification. Neurosci. Biobehav. Rev. 2016;68:282–297. doi: 10.1016/j.neubiorev.2016.05.033. [DOI] [PubMed] [Google Scholar]
- 171.Gold M.S., Pottash A.C., Sweeney D.R., Extein I., Annitto W.J. Opiate detoxification with lofexidine. Drug Alcohol Depend. 1981;8(4):307–315. doi: 10.1016/0376-8716(81)90040-5. [DOI] [PubMed] [Google Scholar]
- 172.Oesterle T.S., Thusius N.J., Rummans T.A., Gold M.S. Medication-assisted treatment for opioid-use disorder. Mayo Clin. Proc. 2019;94(10):2072–2086. doi: 10.1016/j.mayocp.2019.03.029. [DOI] [PubMed] [Google Scholar]
- 173.Blum K., Han D., Modestino E.J., Saunders S., Roy A.K., 3rd, Jacobs W., et al. A systematic, intensive statistical investigation of data from the comprehensive analysis of reported drugs (CARD) for compliance and illicit opioid abstinence in substance addiction treatment with buprenorphine/naloxone. Subst. Use Misuse. 2018;53(2):220–229. doi: 10.1080/10826084.2017.1400064. [DOI] [PubMed] [Google Scholar]
- 174.Srivastava A.B., Gold M.S. Beyond supply: how we must tackle the opioid epidemic. Mayo Clin. Proc. 2018;93(3):269–272. doi: 10.1016/j.mayocp.2018.01.018. [DOI] [PubMed] [Google Scholar]
- 175.Dunn J.P., Kessler R.M., Feurer I.D., Volkow N.D., Patterson B.W., Ansari M.S., et al. Relationship of dopamine type 2 receptor binding potential with fasting neuroendocrine hormones and insulin sensitivity in human obesity. Diabetes Care. 2012;35(5):1105–1111. doi: 10.2337/dc11-2250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Donahue R.J., Muschamp J.W., Russo S.J., Nestler E.J., Carlezon W.A., Jr. Effects of striatal ΔFosB overexpression and ketamine on social defeat stress-induced anhedonia in mice. Biol. Psychiatry. 2014;76(7):550–558. doi: 10.1016/j.biopsych.2013.12.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Kleber H.D., Gold M.S. Use of psychotropic drugs in treatment of methadone maintained narcotic addicts. Ann. N. Y. Acad. Sci. 1978;311:81–98. doi: 10.1111/j.1749-6632.1978.tb16767.x. [DOI] [PubMed] [Google Scholar]
- 178.Blum K., Han D., Femino J., Smith D.E., Saunders S., Simpatico T., et al. Systematic evaluation of "compliance" to prescribed treatment medications and "abstinence" from psychoactive drug abuse in chemical dependence programs: data from the comprehensive analysis of reported drugs. PLoS One. 2014;9(9) doi: 10.1371/journal.pone.0104275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Srivastava A.B., Gold M.S. Naltrexone: a history and future directions. Cerebrum. 2018;2018 [PMC free article] [PubMed] [Google Scholar]
- 180.Merlo L.J., Gold M.S. Prescription opioid abuse and dependence among physicians: hypotheses and treatment. Harvard Rev. Psychiatry. 2008;16(3):181–194. doi: 10.1080/10673220802160316. [DOI] [PubMed] [Google Scholar]
- 181.DuPont R.L., McLellan A.T., White W.L., Merlo L.J., Gold M.S. Setting the standard for recovery: Physicians' health programs. J. Subst. Abus. Treat. 2009;36(2):159–171. doi: 10.1016/j.jsat.2008.01.004. [DOI] [PubMed] [Google Scholar]
- 182.Washton A.M., Pottash A.C., Gold M.S. Naltrexone in addicted business executives and physicians. J. Clin. Psychiatry. 1984;45(9 Pt 2):39–41. [PubMed] [Google Scholar]
- 183.Gold M.S., Dackis C.A., Washton A.M. The sequential use of clonidine and naltrexone in the treatment of opiate addicts. Adv. Alcohol Subst. Abuse. 1984;3(3):19–39. doi: 10.1300/J251v03n03_03. [DOI] [PubMed] [Google Scholar]
- 184.Hill E., Han D., Dumouchel P., Dehak N., Quatieri T., Moehs C., et al. Long term Suboxone™ emotional reactivity as measured by automatic detection in speech. PLoS One. 2013;8(7) doi: 10.1371/journal.pone.0069043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Fried L., Modestino E.J., Siwicki D., Lott L., Thanos P.K., Baron D., et al. Hypodopaminergia and "precision Behavioral management" (PBM): it is a generational family affair. Curr. Pharm. Biotechnol. 2020;21(6):528–541. doi: 10.2174/1389201021666191210112108. [DOI] [PubMed] [Google Scholar]
- 186.Gold M.S., Redmond D.E., Jr., Kleber H.D. Clonidine blocks acute opiate-withdrawal symptoms. Lancet. 1978;2(8090):599–602. doi: 10.1016/s0140-6736(78)92823-4. [DOI] [PubMed] [Google Scholar]
- 187.Orsini C.A., Ginton G., Shimp K.G., Avena N.M., Gold M.S., Setlow B. Food consumption and weight gain after cessation of chronic amphetamine administration. Appetite. 2014;78:76–80. doi: 10.1016/j.appet.2014.03.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Edge P.J., Gold M.S. Drug withdrawal and hyperphagia: lessons from tobacco and other drugs. Curr. Pharm. Des. 2011;17(12):1173–1179. doi: 10.2174/138161211795656738. [DOI] [PubMed] [Google Scholar]
- 189.New Blum K., Yok N. The Free Press; NYC: 1991. Alcohol and the Addictive Brain. [Google Scholar]