Abstract
Hepatocellular carcinoma (HCC) is a leading cause of cancer death worldwide, and the cancer with the fastest increase in mortality in the USA, with more than 39,000 cases and 29,000 deaths in 2018. As with many cancers, survival is significantly improved by early detection. The median survival of patients with early HCC is >60 months but <15 months when detected at an advanced stage. Surveillance of at risk patients improves outcome but fewer than 20% of those at risk for HCC receive surveillance, and current surveillance strategies have limited sensitivity and specificity. Ideally, blood based biomarkers with adequate sensitivity or specificity would be available for early detection of HCC; however, the most commonly used biomarker for HCC, alpha fetoprotein, has inadequate performance characteristics. There are several candidate serum proteomic, glycomic, and genetic markers that have gone through early stages of biomarker validation and have shown promise for the early detection of HCC, but these markers require validation in well curated cohorts. Ongoing prospective cohort studies will permit retrospective longitudinal (phase III biomarker study) validation of biomarkers. In this review, we highlight promising candidate biomarkers and biomarker panels that have completed phase II evaluation but require further validation prior to clinical use.
Introduction
Hepatocellular carcinoma (HCC) is the seventh most common cancer diagnosis worldwide with high associated mortality.(1) HCC is a unique malignancy, as it typically arises in the setting of chronic liver disease in particular cirrhosis, with competing risks of liver failure, contributing to its low 5-year survival rates of 18%-20%.(2) More than half of worldwide HCC deaths occur in Asia, due to endemic hepatitis B (HBV) infection. HCC incidence is rising in many Western countries due to the rising prevalence of nonalcoholic fatty liver disease (NAFLD), alcohol-related liver disease, and hepatitis C (HCV) related complications despite the availability of direct acting anti-viral therapies.(3-5)
Stage of HCC diagnosis is highly predictive of overall mortality. Early stage patients are eligible for curative therapies, including resection, ablative therapies, and liver transplantation, whereas late stage patients are generally only eligible for palliative systemic therapies with suboptimal response rates. As a result, 5-year survival exceeds 70% in patients with early stage HCC while it is less than 5% at advanced stages.(6,7) Unfortunately, due to poor utilization of surveillance, inadequate surveillance methods, and lack of risk-based strategies most patients are diagnosed at late stages.(8)
Surveillance for HCC is recommended in at risk patients, including those with cirrhosis of all etiologies, and certain populations with chronic HBV infection (Table 1).(9) Guidelines recommend HCC surveillance with abdominal ultrasound (US) with or without serum alpha-fetoprotein (AFP) measurement every 6 months.(9,10)
Table 1.
Populations recommended for surveillance for hepatocellular carcinoma
| Population | Annual Incidence |
|---|---|
| Cirrhosis of any etiology | 1%-8% |
| Asian males with chronic hepatitis B ≥ 40 years of age | 0.4%-0.6% |
| Asian females with chronic hepatitis B ≥ 50 years of age | 0.3%-0.6% |
| African patients and North American blacks with chronic hepatitis B ≥ 20 years of age | 0.3%-0.6% |
A recent meta-analysis showed that sensitivity of US based surveillance for HCC early detection is 45%.(11) Notably there was significant heterogeneity in sensitivity of US-based surveillance (21%-89%) across studies included in the meta-analysis, which highlights a limitation of imaging-based surveillance.(11) Other studies have also highlighted the harms of US surveillance due to suboptimal specificity leading to unnecessary further testing that may carry risks of complications.(12,13) Furthermore, the presence of cirrhosis and obesity, both more prevalent in Western patients when compared to Asians, have been shown to decrease sensitivity of abdominal US.(14) The limitations of US-based surveillance have been well documented and highlights the need for more objective and sensitive methods to conduct surveillance for HCC. In addition, US surveillance is intensive and requires a separate patient encounter (i.e. every 6 months in radiology) which can represent significant logistical barriers to attainment for both patients and providers.(15)
Ideally, validated blood based biomarkers with sufficient sensitivity and specificity for the early detection of HCC would be available. There are several candidate biomarkers that have been studied for HCC early detection, and herein we will review the current landscape of these biomarkers.
Biomarker Validation
The definition of a biomarker is broad, and includes any measurable substance, structure, or process that can detect or predict the outcome of a disease. Originally proposed by Pepe et al in conjunction with the Early Detection Research network (EDRN) of the National Cancer Institute (NCI), the discovery and validation of biomarkers require several phases prior to routine clinical usage.(16) (Figure 1) Initial discovery occurs in a preclinical exploratory phase followed by clinical assay validation which involves developing the assay for its measurement (Phase I). Clinical validation begins with conducting retrospective case control studies, comparing, preferably early stage cases and controls without cancer from a relevant screening population (Phase II). Phase III validation involves longitudinal assessment of the biomarker to determine its performance in detecting preclinical disease. Late stage validation studies assess the biomarker performance in clinical practice and determine its impact in reducing the burden of a cancer in a population. Very few biomarkers in HCC have undergone adequate validation due to the lack of large, representative prospective cohorts with adequate duration of follow-up and outcomes (early stage HCC), which is the reason behind their limited clinical use. The NCI EDRN Hepatocellular carcinoma Early Detection Strategy study and ongoing prospective cohort studies will overcome this barrier.(17)
Figure 1.
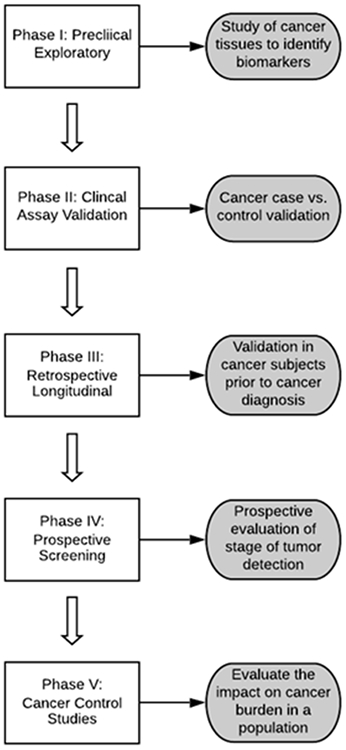
Phases of clinical biomarker validation
Of the biomarkers described below, AFP has been most extensively studied, and despite its limitations have been in use clinically for several decades. By contrast, most of the other biomarkers except for AFP-L3 and des-gamma carboxy prothrombin (DCP) have only been evaluated in few studies and their performance for early HCC detection in clinical settings, particularly among patients with non-viral liver disease, is unknown. (Table 2)
Table 2.
Candidate biomarkers for hepatocellular carcinoma early detection
| Biomarker | Phase of Development |
Early Detection Performance |
AUROC for early detection |
|---|---|---|---|
| Alpha fetoprotein (AFP)(20-25) | 5 | Sensitivity: 39-64% Specificity: 76%-97% |
0.75-0.82 |
| Lens culinaris agglutinin-reactive AFP (AFP-L3)(20,33) | 2/3 | Sensitivity: 49-62% Specificity: 90% |
0.66-0.76 |
| Des-gamma carboxyprothrombin (DCP)(20,33) | 2/3 | Sensitivity: 34-40% Specificity: 81-98% |
0.72 |
| Osteopontin(38,40,41) | 2 | Sensitivity: 49% Specificity: 72% |
0.73 |
| Midikine(44) | 2/3 | Sensitivity: 87% Specificity: 90% |
0.923 |
| Dikkopf-1(45,46) | 2 | Sensitivity: 41%-74% Specificity: 87% |
0.61-0.88 |
| Glypican-3(50-52) | 2 | Sensitivity: 55% Specificity: >95% |
0.793 |
| Alpha-1 fucosidase(53) | 2 | Sensitivity: 56% Specificity: 69% |
0.506 |
| Golgi Protein-73(58,59) | 2 | Sensitivity: 62%-79% Specificity: 62%-88% |
Not available |
| Squamous cell carcinoma antigen (SCCA)(60-63) | 2 | Data for early stage HCC not available | Data for early stage HCC not available |
Biomarker Completed 5 Phases of Validation
Alpha fetoprotein (AFP)
AFP is the most commonly used biomarker in the early detection of HCC and the only biomarker which has been validated for clinical use. AFP alone is not currently included in societal guidelines for HCC surveillance due to concerns about specificity and limited sensitivity in the detection of early stage HCC.(18) However, a recent meta-analysis has shown that AFP can increase the sensitivity of HCC early detection when used in combination with abdominal US (63% vs 45% of US alone.)(11) False positive AFP elevations can occur with elevated serum alanine aminotransferase (ALT) level, seen in chronic hepatitis C and B infections.(19) Up to 40-50% of HCCs do not have elevated levels of AFP, limiting the sensitivity of AFP alone for HCC detection. Published cohort studies, including a large multi-center study funded by the NCI EDRN,(20) show the sensitivity of AFP for detecting early HCC ranges from 39-64% and specificity ranges from 76-97%.(21-25) Cut-off values for serum AFP vary widely across studies, however a value of 20 ng/mL is accepted as a valid threshold in the early detection of HCC. For patients undergoing surveillance, the change in AFP value over time is superior to single AFP values, in the detection of early stage HCC (receiver operating curve 0.81 vs 0.76).(26,27) The change in AFP has also been integrated into the recently validated Hepatocellular Carcinoma Early Detection Screening (HES) algorithm.(28) Thus, while AFP has been through the 5 phases of biomarker development, it’s routine use as a part of the surveillance strategy for HCC early detection is controversial.
Biomarkers with limited Phase III validation data
AFP L3
AFP-L3, or lens culinaris agglutinin-reactive AFP, is a fucosylated glycoform of AFP that has been studied for the detection of early stage HCC.(29) While traditional AFP-L3 assay requires an AFP level above 10 ng/mL for detection, the use of a highly sensitive assay for AFP-L3 (hs-AFP-L3) makes measurements possible in patients with AFP levels as low as 2 ng/mL.(30) Unfortunately, while AFP-L3 has a better specificity for early HCC detection than AFP (~90%), its sensitivity is inferior (49-60%).(20,31,32) A biomarker validation study from Korea, including 42 patients with HCC, showed that AFP-L3 has an area under the receiver operating characteristic curve (AUROC) of 0.73 at the time of HCC diagnosis, compared to 0.77 for AFP. For the 38 Barcelona Clinic Liver Cancer (BCLC) stage 0/A patients (i.e. early stage HCC) in this cohort, the AUROC of AFP was similar at 0.76 which improved to 0.81 when combined with AFP-L3. Additionally, AFP-L3 was significantly higher in patients with HCC 6 months prior to clinical diagnosis compared to controls.(33) In the EDRN HEDS Phase II validation including 131 patients with early stage HCC, the AUROC of AFP-L3 was 0.66 versus 0.80 for AFP alone.(20) One major limitation with AFP-L3 is that AFP itself has low sensitivity, and examination of any isoform will not improve sensitivity. Further phase III validation of AFP-L3 is needed, to confirm whether it has incremental value compared to or in combination with AFP alone.
Des-gamma carboxyprothrombin
Des-gamma carboxyprothrombin (DCP) is an abnormal prothrombin produced because of vitamin K insufficiency caused by dysfunctional intracellular transport mechanisms; defects in gamma-carboxylase enzyme; and cytoskeletal changes that impair vitamin K uptake as hepatocytes undergo malignant transformation.(34) Sensitivity and specificity of DCP in detecting early stage HCC ranges from 34-62% and 81-98%, respectively.(35) In the EDRN phase II study of 131 early HCC patients, DCP had an AUROC of 0.72.(20) Limited Phase III evaluation has demonstrated poor sensitivity in detecting pre-clinical HCC (12.1%).(36) Combining DCP and AFP levels can increase the sensitivity of DCP to 80% for large tumors (>3cm) and 70% for small tumors (2-3 cm). Recent data suggest DCP does not increase discriminatory power when combined with AFP and AFP-L3 for early HCC detection.(33) Despite lack of formal phase III or IV validation, DCP is used in many countries worldwide for HCC early detection. Based on the validation studies thus far, DCP alone does not appear to have sufficient performance characteristics for early stage HCC detection but may still have value as part of a biomarker panel.
Biomarkers with Phase II validation data
All of the below biomarkers have promising results in Phase II evaluation but still require Phase III validation given biomarker performance can be overestimated in Phase II studies.
Osteopontin
Osteopontin is an integrin-binding phosphoprotein that can mediate cell signaling involved in regulating tumor progression.(37-39) Osteopontin for HCC early detection, defined by BCLC stage 0/A, was evaluated in a meta-analysis of 4 studies which showed a sensitivity of 49% (95% CI: 42–56) and specificity of 72% (95% CI: 68–76) both comparable to the performance of AFP alone. (40,41) When osteopontin and AFP were combined the sensitivity improved to 73% (95% CI: 67–79) with little change in the specificity 68% (95% CI: 64–72). One of the studies included in the meta-analysis was a Phase II validation study of 78 patients with early stage HCC and 76 patients with cirrhosis. Osteopontin outperformed AFP alone for early detection (AUROC: 0.73 [95% CI: 0.62-0.85] vs AUROC: 0.68 [95% CI: 0.54-0.82])(38) and performance of osteopontin was further improved when combined with AFP, with AUROC for early stage HCC of 0.81 (95% CI: 0.70-0.91).
Midikine
Midkine (MDK) is a heparin binding growth factor involved in cell growth, invasion and angiogenesis during cancer progression.(42) MDK levels have been shown to be elevated in patients with very early stage HCC and decline following curative surgery. MDK levels rise or remain elevated in patients with incompletely treated or recurrent HCC.(43) A phase II validation study included 119 BCLC 0/A patients, found that the sensitivity of MDK was 87% and specificity 90%, while AFP’s sensitivity was 52% and specificity 35%.(44) The AUROC of MDK in this study was 0.92 for patients with BCLC 0/A HCC compared to patients with cirrhosis. The combination of MDK and AFP further improved the early detection rate of HCC to 96%.(44) In a small phase 3 study of patients with cirrhosis related to nonalcoholic steatohepatitis, MDK was not superior to AFP for the early detection of AFP, but was elevated in roughly half of the patients who did not have an elevation of AFP.(43)
Dikkopf-1
Dikkopf-1 (DKK1) is a glycoprotein that functions as a secretory antagonist of the Wnt/B-catenin signaling pathway. In one study of 1,284 patients (831 in the test cohort and 453 in the validation cohort), DKK1 concentrations were significantly higher in patients with HCC than controls in the test cohort, and values did not differ significantly between the control (cirrhosis patients and healthy controls) groups (p<0.001).(45) The sensitivity for detection of early-stage HCC was 70-72% and specificity was 87-90% in the validation cohort (AUROC: 0.88). Combining DKK1 levels with AFP enhanced the detection rate of early-stage HCC (AUROC: 0.89).(45) In a separate Phase II study of predominantly hepatitis B infected patients in South Korea (n=208), the combination of AFP and DKK1 was similar to AFP alone for the detection of early stage HCC (AUROC: 0.63 vs 0.69).(46) These preliminary data suggest etiology of liver disease may be an important factor in DKK1 performance as an early detection biomarker; thus, its utility remains to be determined.
Glypican-3 (GPC-3)
Glypican 3, (GPC-3) is a cell-surface heparan sulfate proteoglycan that regulates cell proliferation and tumor suppression.(47-49) Meta-analysis of 19 Phase II biomarker studies found the sensitivity of GPC-3 for early detection of HCC is suboptimal when used alone (~55%), and increases to 76% when combined with AFP. However, the available data on early stage HCC detection was limited.(50) The specificity of GPC-3, is >95%,(50-52) suggesting it’s potential utility as a complementary biomarker to increase sensitivity of AFP or other serum biomarkers.
Alpha-1 fucosidase
Alpha-1 fucosidase (AFU) is a lysosomal enzyme that has been shown to be elevated in patients with HCC. In one Phase II biomarker study of 57 patients with early stage HCC, the sensitivity and specificity of AFU in the early detection of HCC was 56% and 69%, respectively. Combining AFU and AFP did not raise the sensitivity or AUROC to an acceptable level as AFP alone outperformed the combination.(53) The specificity of AFU is poor as it is also over-expressed in diabetes, pancreatitis, and hypothyroidism, and varies across patient race/ethnicities.(54) However, a small Phase III study of 27 patients found that AFU activity was elevated in 85% of patients at least 6 months before the detection of HCC indicating additional studies may be warranted.(55)
Golgi protein-73
Golgi protein-73 (GP-73) is a transmembrane protein expressed in epithelial cells and can be elevated in patients with HCC and advanced fibrosis secondary to hepatitis B or hepatitis C infection. It was first identified as a potential biomarker of HCC through glycoproteomics.(56,57) In a Phase II study, GP-73 was found to have a sensitivity and specificity of 69% and 86%, respectively, for distinguishing between HCC and cirrhosis.(58) The sensitivity and specificity for detecting early stage HCC (BCLC 0/A) in this cohort was similar at 62% and 88%, respectively. Combining GP-73 and AFP increased sensitivity and specificity to 98% and 85% for differentiating all stages of HCC from cirrhosis, however performance of the combination in early stage HCC was not reported.(58) In one meta-analysis, the sensitivity of GP-73 was 79%, while the specificity was 62%, similar to the performance of AFP.(59) One major issue with GP-73 is the reliance on western-blot analysis for the accurate measurement of the isoform associated with HCC. ELISA based assays, which are inherently better suited for clinical use, have proven difficult to develop for the specific GP-73 isoforms associated with HCC.
Squamous cell carcinoma antigen
Squamous cell carcinoma antigen (SCCA) is a serine protease inhibitor that is present in squamous epithelium. SCCA is expressed by neoplastic epithelial cells and hepatocytes in which it promotes tumor growth through the inhibition of apoptosis. In addition to SCCA, the SCCA-immune complex (SCCA-IgM) have been investigated for the detection of HCC. SCCA has high sensitivity for HCC (89%), however it suffers from poor specificity (50%) in differentiating HCC from cirrhosis.(60,61) In a meta-analysis of 11 studies, SCCA had an AUROC of 0.80, while SCCA-IgM had an AUROC of 0.77. Unfortunately, performance in the detection of early stage HCC was not separately reported.(62,63)
Glycosylation variants
Fucosylated Glycoproteins:
Based upon the knowledge that changes in biomarkers can occur at the cellular level and not the protein level, others have attempted to identify proteins with glycan changes that could be used as biomarkers of HCC (56,64-72). Increased levels of fucosylated proteins such as hemopexin (66,73-76), fetuin A (74,77,78), alpha1-antitrypsin (65,77,79-83), ceruloplasmin (64,81,84), haptoglobin (80,85-88), serum paraoxonase 1 (89,90), and histidine-rich glycoprotein (78,84) have been observed in the serum of patients with HCC, either by direct glycan sequencing or by lectin based methods. Fucosylation has also been observed directly in the tumor itself (91) and together these results strongly suggest that increased fucosylation, both core and outer arm, occurs on a large number of proteins.
Only a limited number of Phase I and Phase II studies have been conducted for the fucosylated glycoproteins. The most notable is fucosylated kininogen, which has been examined in several independent phase II cohorts (internal and external validation). (71,92,93). On its own, fucosylated kininogen is not an adequate biomarker, but when combined with AFP and other clinical factors appears to have excellent biomarker performance with an AUROC of 0.97 in one Phase II study which included 69 patients with early stage HCC.(94)
Glycosylated Haptoglobin
Another glycoprotein that has been identified as having altered glycosylation in HCC is haptoglobin. Initial work identified alterations in fucosylation and sialyation on this molecule along with other changes.(95) Subsequent work has identified increased levels of branching on this molecule as well.(96-98) Similar to the other glycoprotein markers, individual performance is limited but these markers in combination with AFP and other clinical factors achieve sensitivities close to 80% at 95% specificity for early detection in Phase II studies.(95)
Notably, the analysis of protein glycoforms in plate based assays utilizing lectins are dramatically impacted by the presence of heterophilic antibodies in patients with liver fibrosis.(93,99-102) As most patients with HCC have advanced liver fibrosis, these antibodies have to be accounted for prior to analysis. Direct mass spectrometry based approaches have shown promise but will require some level of refinement and simplification before routine clinical use.(103-105)
Genomic Markers
MicroRNA
The aberrant expression of MicroRNAs (miRNA), which are circulating non-coding RNAs, can contribute to oncogenesis and cancer progression. Due to their inherent stability and their role in tumor proliferation, several studies have evaluated circulating miRNA as a biomarker for the diagnosis of HCC. Two specific miRNA, miRNA-21 and miRNA-199a, have been proposed as potential biomarkers for the early diagnosis of HCC. Serum levels of miRNA-21 have been found to be elevated in patients with HCC, and have shown promise in differentiating between cirrhosis and HCC in small Phase II studies, although the results of these studies can be difficult to interpret due to inadequate control patients and small numbers of early stage HCC patients.(106) There are several additional candidate miRNAs under investigation for the early detection and prognostication of HCC, and many are being studied individually or as components of miRNA panels combined with other biomarkers in Phase I and Phase II studies.(107,108) There are challenges with miRNA analyses due to variable annotation; however, efforts are underway to ensure uniformity in characterization of miRNA molecules.(109,110)
DNA Mutations
Cells derived from HCC tissue harbor genetic mutations and epigenetic modifications that can be involved in the oncogenesis of HCC (111,112), while others may be “passengers” and not of biological consequence, in themselves. In large scale analysis of HCC-specific mutations, deletions or epigenetic modifications occur in at least one of 31 different genes.(111) The most frequent mutation associated with HCC is the TERT promoter, with approximately 60% of all HCC containing these mutations.(113) The next most commonly mutated genes are in p53 and CTNNB1, which are mutated in 25-35% of the HCC tissues.(114) Cell free DNA in plasma and/or urine derived from HCC tissue, containing these abnormalities, has been detected and proposed for use in risk stratification and cancer detection.(115) In one study, detection of TERT promoter mutated DNA in the plasma was 47% sensitive for HCC (all stages), overall, but reached 87% when restricted to males with chronic HCV.(116) Sensitivities and specificities vary greatly with different genes tested and populations studied, complicating implementation of these assays, and practical use awaits further development.
Epigenetic Modifications/DNA Methylation
Methylation of DNA is often involved in the carcinogenesis of HCC and thus studies have been conducted investigating circulating cell free methylated DNA for the early detection of HCC.(117) Several panels of DNA methylation signature exist; however to date, there has been limited validation and adequate comparison to controls for clinical use. One panel of 4 methylated markers, in combination with AFP and AFP-L3 showed a 71% sensitivity for early stage HCC (BCLC 0/A) with a specificity of 90% and an AUROC of 0.91. This panel compared well to AFP alone (sensitivity 21, specificity 98%; AUROC 0.81).(118) While these initial results are promising, these data are awaiting further validation.
Algorithms
HCC tumor biology is heterogeneous, with carcinogenesis involving several genetic alterations even within a single patient.(119) This in part explains the limited performance of any single biomarker. Algorithms/panels comprising multiple biomarkers encompassing heterogeneous pathways in carcinogenesis and tumor biology, and clinical factors associated with risk of HCC such as sex, age and etiology of liver disease have been developed and undergone validation in order to improve the sensitivity and specificity of HCC early detection. (Table 3)
Table 3.
Algorithms that have been evaluated for the detection of hepatocellular carcinoma
| Algorithms | Components | Phase of Development |
Early Detection Performance |
AUROC for Early Detection |
|---|---|---|---|---|
| GALAD score(121) | Gender, age, AFP, AFP-L3 and DCP | 2 | Sensitivity: 68% Specificity: 95% |
0.85-0.95 |
| Doylestown+Fucosylated kininogen(94) | Fucosylated kininogen, log AFP, age, gender, alkaline phosphatase, and alanine aminotransferase | 2 | 7 | 0.97 |
| Hepatocellular Carcinoma Early Detection Screening (HES) Algorithm(124) | Age, AFP, rate of AFP change, alanine aminotransferase, and platelet count | 2/3 | Data for early stage HCC not available. | Data for early stage HCC not available. |
| Methylated DNA Panel(118) | 4 Methylated DNA markers, AFP, AFP-L3 | 2 | Sensitivity: 71% Specificity: 90% |
0.91 |
Gender, Age, AFP-L3, AFP, and Des-carboxy-prothrombin (GALAD) Score
The GALAD score includes gender, age, AFP, AFP-L3 and DCP.(120) The ability of the GALAD score to discern between HCC, cirrhosis, and other hepatobiliary malignancies (e.g. cholangiocarcinoma) has been examined. Derivation of this model was based on data from 833 patients (394 with HCC and 439 with chronic liver disease) in two centers in the United Kingdom, and the model was validated in independent cohorts of 6834 patients in Japan, Germany and Hong Kong (2430 with HCC and 4404 with chronic liver disease). A total of 1038 patients across all centers had early stage HCC, defined as tumor size < 3cm. Overall sensitivity ranged from 80-91%, while the specificity ranged from 81-90% across the cohorts with an AUROC of 0.85-0.95.(121) A Phase 2 validation study in patients with nonalcoholic steatohepatitis with vs. without HCC from 8 centers in Germany had an AUROC of 0.91 for early detection with 68% sensitivity and 95% specificity.(122) There are ongoing studies to provide Phase III validation of this panel in prospective cohorts.
Doylestown Algorithm
The Doylestown algorithm comprised log AFP, age, gender, alkaline phosphatase, and alanine aminotransferase. In a Phase II study of 69 patients with early stage HCC (stage T1 or T2 disease) and 93 cirrhosis controls, the addition of fucosylated kininogen to the algorithm had a higher AUROC than both the Doylestown algorithm and AFP alone (0.97 vs 0.93 and 0.80, respectively). Notably, in 29 patients with early HCC and an AFP<20, the Doylestown algorithm with fucosylated kinnogen had a 89% detection and maintained an AUROC of 0.97.(92,123) This level of performance should be confirmed in larger Phase II studies in addition to Phase 3 validation studies, which are in progress.
Hepatocellular Carcinoma Early Detection Screening (HES) Algorithm
The HES algorithm including age, AFP, rate of AFP change, alanine aminotransferase, and platelet count has been validated in a Phase 2 study using data from the Veterans Administration.(124) In a cohort comprising 4,804 patients with predominantly HCV-related HCC, the HES algorithm outperformed AFP alone in HCC detection in the 6 months prior to clinical diagnosis with a sensitivity of 53% vs 48% at a 10% false positive rate. The validation was limited, however because it did not include the performance in patients with early stage HCC and its performance in other non-viral etiologies of liver disease requires further validation.(124)
Future Directions
While several candidate biomarkers for HCC early detection exist, there has been little translation to clinical practice largely because of lack of well-annotated cohorts for validation studies. There are, however, several prospective studies including the EDRN HEDS study(17) and the Texas Hepatocellular Carcinoma Consortium, that may provide the opportunity to perform large scale Phase III validation studies.(125) These cohorts will allow for validation of individual biomarkers as well as algorithms combining multiple biomarkers and clinical data, in the detection of early HCC for viral as well as non-viral etiologies of liver disease, which is important given the changing epidemiology of HCC in the USA and worldwide.(126) Efforts are underway to better risk stratify patients with regards to their risk of HCC. Patient level factors, such as age and gender, are currently included in many of the existing biomarker algorithms. Further understanding of individual risk based on genetic profile or other biomarkers, may allow for personalized surveillance strategies of patients at risk for HCC(127,128) such that high risk groups may be identified for more intense surveillance while low risk groups may forego surveillance. Finally, studies are underway to develop and validate imaging techniques (e.g. abbreviated MRI) and technologies, such as digital extraction of high dimensional quantitative data from imaging (e.g. radiomics), in order to improve the sensitivity of HCC early detection and for patient risk stratification that may complement serum biomarkers.(129,130)
Conclusions
There are several candidate biomarkers that have the potential to dramatically improve the early detection of HCC. Approaches that combine patient risk stratification and multiple candidate biomarkers will likely yield the best performance characteristics. Support for the establishment and long-term follow-up of well annotated cohorts of diverse race/ethnicity and etiologies of liver disease, and proper collection and storage of biospecimens is crucial for validation of new biomarkers and algorithms in Phase 3 studies, with the goal that some will qualify for progression to Phase 4/5 studies and ultimately contribute to improving outcomes in patients with HCC.
Acknowledgments
Grant Funding:
ASL and NDP are supported in part by NIH U01CA230669
AGS is supported in part by NIH U01CA230694 and R01CA222900
TB is supported by a grant from the Commonwealth of Pennsylvania
Footnotes
Conflict of interest:
NDP is a consultant for Exact Sciences and Wako; receives institutional research funding from Glycotest
ASM reports no conflicts of interest
AGS is a consultant for Exact Sciences, Glycotest, Hoffmann-La Roche, and Wako
TB is a board member of Hepion Inc and a Co-founder/shareholder of Glycotest
JAM is a consultant for Glycotest
ASL is an advisor for Epigenomics
References
- 1.Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2018;68(6):394–424 doi 10.3322/caac.21492. [DOI] [PubMed] [Google Scholar]
- 2.Golabi P, Fazel S, Otgonsuren M, Sayiner M, Locklear CT, Younossi ZM. Mortality assessment of patients with hepatocellular carcinoma according to underlying disease and treatment modalities. Medicine 2017;96(9). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Yang JD, Hainaut P, Gores GJ, Amadou A, Plymoth A, Roberts LR. A global view of hepatocellular carcinoma: trends, risk, prevention and management. Nature Reviews Gastroenterology & Hepatology 2019:1–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Tapper EB, Parikh ND. Mortality due to cirrhosis and liver cancer in the United States, 1999-2016: observational study. bmj 2018;362:k2817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kanwal F, Kramer JR, Asch SM, Cao Y, Li L, El-Serag HB. Long-term risk of hepatocellular carcinoma in HCV patients treated with direct acting antiviral agents. Hepatology 2019. [DOI] [PubMed]
- 6.Heimbach JK, Kulik LM, Finn RS, Sirlin CB, Abecassis MM, Roberts LR, et al. AASLD guidelines for the treatment of hepatocellular carcinoma. Hepatology 2018;67(1):358–80. [DOI] [PubMed] [Google Scholar]
- 7.Galle PR, Forner A, Llovet JM, Mazzaferro V, Piscaglia F, Raoul J-L, et al. EASL clinical practice guidelines: management of hepatocellular carcinoma. Journal of hepatology 2018;69(1):182–236. [DOI] [PubMed] [Google Scholar]
- 8.Choi DT, Kum H-C, Park S, Ohsfeldt RL, Shen Y, Parikh ND, et al. Hepatocellular carcinoma screening is associated with increased survival of patients with cirrhosis. Clinical Gastroenterology and Hepatology 2019;17(5):976–87. e4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Marrero JA, Kulik LM, Sirlin CB, Zhu AX, Finn RS, Abecassis MM, et al. Diagnosis, staging, and management of hepatocellular carcinoma: 2018 practice guidance by the American Association for the Study of Liver Diseases. Hepatology 2018;68(2):723–50. [DOI] [PubMed] [Google Scholar]
- 10.Singal AG, Pillai A, Tiro J. Early detection, curative treatment, and survival rates for hepatocellular carcinoma surveillance in patients with cirrhosis: a meta-analysis. PLoS medicine 2014;11(4):e1001624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Tzartzeva K, Obi J, Rich NE, Parikh ND, Marrero JA, Yopp A, et al. Surveillance imaging and alpha fetoprotein for early detection of hepatocellular carcinoma in patients with cirrhosis: a meta-analysis. Gastroenterology 2018;154(6):1706–18. e1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Konerman MA, Verma A, Zhao B, Singal AG, Lok AS, Parikh ND. Frequency and outcomes of abnormal imaging in patients with cirrhosis enrolled in a hepatocellular carcinoma surveillance program. Liver Transplantation 2019;25(3):369–79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Atiq O, Tiro J, Yopp AC, Muffler A, Marrero JA, Parikh ND, et al. An assessment of benefits and harms of hepatocellular carcinoma surveillance in patients with cirrhosis. Hepatology 2017;65(4):1196–205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Simmons O, Fetzer DT, Yokoo T, Marrero JA, Yopp A, Kono Y, et al. Predictors of adequate ultrasound quality for hepatocellular carcinoma surveillance in patients with cirrhosis. Alimentary pharmacology & therapeutics 2017;45(1):169–77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Farvardin S, Patel J, Khambaty M, Yerokun OA, Mok H, Tiro JA, et al. Patient-reported barriers are associated with lower hepatocellular carcinoma surveillance rates in patients with cirrhosis. Hepatology 2017;65(3):875–84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Pepe MS, Etzioni R, Feng Z, Potter JD, Thompson ML, Thornquist M, et al. Phases of biomarker development for early detection of cancer. J Natl Cancer Inst 2001;93(14):1054–61. [DOI] [PubMed] [Google Scholar]
- 17.Borges KA, Dai J, Parikh ND, Schwartz M, Nguyen MH, Roberts LR, et al. Rationale and design of the Hepatocellular carcinoma Early Detection Strategy study: A multi-center longitudinal initiative of the National Cancer Institute’s Early Detection Research Network. Contemporary clinical trials 2019;76:49–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bruix J, Sherman M, American Association for the Study of Liver D. Management of hepatocellular carcinoma: an update. Hepatology (Baltimore, Md 2011;53(3):1020–2 doi 10.1002/hep.24199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Yang JD, Dai J, Singal AG, Gopal P, B DA, Nguyen MH, et al. Improved Performance of Serum Alpha-Fetoprotein for Hepatocellular Carcinoma Diagnosis in HCV Cirrhosis with Normal Alanine Transaminase. Cancer Epidemiol Biomarkers Prev 2017. doi 10.1158/1055-9965.EPI-16-0747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Marrero JA, Feng Z, Wang Y, Nguyen MH, Befeler AS, Roberts LR, et al. Alpha-fetoprotein, des-gamma carboxyprothrombin, and lectin-bound alpha-fetoprotein in early hepatocellular carcinoma. Gastroenterology 2009;137(1):110–8 doi 10.1053/j.gastro.2009.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Oka H, Tamori A, Kuroki T, Kobayashi K, Yamamoto S. Prospective study of α-fetoprotein in cirrhotic patients monitored for development of hepatocellular carcinoma. Hepatology 1994;19(1):61–6. [PubMed] [Google Scholar]
- 22.Lok AS, Sterling RK, Everhart JE, Wright EC, Hoefs JC, Di Bisceglie AM, et al. Des-γ-carboxy prothrombin and α-fetoprotein as biomarkers for the early detection of hepatocellular carcinoma. Gastroenterology 2010;138(2):493–502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Chang TS, Wu YC, Tung SY, Wei KL, Hsieh YY, Huang HC, et al. Alpha-Fetoprotein Measurement Benefits Hepatocellular Carcinoma Surveillance in Patients with Cirrhosis. Am J Gastroenterol 2015;110(6):836–44; quiz 45 doi 10.1038/ajg.2015.100. [DOI] [PubMed] [Google Scholar]
- 24.Sherman M, Peltekian KM, Lee C. Screening for hepatocellular carcinoma in chronic carriers of hepatitis B virus: incidence and prevalence of hepatocellular carcinoma in a North American urban population. Hepatology 1995;22(2):432–8. [PubMed] [Google Scholar]
- 25.Chalasani N, Horlander JC, Said A, Hoen H, Kopecky KK, Stockberger SM Jr., et al. Screening for hepatocellular carcinoma in patients with advanced cirrhosis. Am J Gastroenterol 1999;94(10):2988–93 doi 10.1111/j.1572-0241.1999.01448.x. [DOI] [PubMed] [Google Scholar]
- 26.Lee E, Edward S, Singal AG, Lavieri MS, Volk M. Improving screening for hepatocellular carcinoma by incorporating data on levels of alpha-fetoprotein, over time. Clin Gastroenterol Hepatol 2013;11(4):437–40 doi 10.1016/j.cgh.2012.11.029. [DOI] [PubMed] [Google Scholar]
- 27.Tayob N, Stingo F, Do KA, Lok ASF, Feng Z. A Bayesian screening approach for hepatocellular carcinoma using multiple longitudinal biomarkers. Biometrics 2018;74(1):249–59 doi 10.1111/biom.12717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Tayob N, Christie I, Richardson P, Feng Z, White DL, Davila J, et al. Validation of the Hepatocellular Carcinoma Early Detection Screening (HES) Algorithm in a Cohort of Veterans With Cirrhosis. Clin Gastroenterol Hepatol 2019;17(9):1886–93 e5 doi 10.1016/j.cgh.2018.12.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Taketa K, Endo Y, Sekiya C, Tanikawa K, Koji T, Taga H, et al. A collaborative study for the evaluation of lectin-reactive α-fetoproteins in early detection of hepatocellular carcinoma. Cancer Research 1993;53(22):5419–23. [PubMed] [Google Scholar]
- 30.Kobayashi M, Hosaka T, Ikeda K, Seko Y, Kawamura Y, Sezaki H, et al. Highly sensitive AFP-L3% assay is useful for predicting recurrence of hepatocellular carcinoma after curative treatment pre-and postoperatively. Hepatology Research 2011;41(11):1036–45. [DOI] [PubMed] [Google Scholar]
- 31.Li D, Mallory T, Satomura S. AFP-L3: a new generation of tumor marker for hepatocellular carcinoma. Clinica chimica acta 2001;313(1):15–9. [DOI] [PubMed] [Google Scholar]
- 32.Sassa T, Kumada T, Nakano S, Uematsu T. Clinical utility of simultaneous measurement of serum high-sensitivity des-[gamma]-carboxy prothrombin and Lens culinaris agglutinin A-reactive a-fetoprotein in patients with small hepatocellular carcinoma. European journal of gastroenterology & hepatology 1999;11(12):1387–92. [DOI] [PubMed] [Google Scholar]
- 33.Choi J, Kim GA, Han S, Lee W, Chun S, Lim YS. Longitudinal Assessment of Three Serum Biomarkers to Detect Very Early-Stage Hepatocellular Carcinoma. Hepatology 2019;69(5):1983–94 doi 10.1002/hep.30233. [DOI] [PubMed] [Google Scholar]
- 34.Bertino G, Ardiri AM, Calvagno GS, Bertino N, Boemi PM. Prognostic and diagnostic value of des-γ-carboxy prothrombin in liver cancer. Drug news & perspectives 2010;23(8):498–508. [DOI] [PubMed] [Google Scholar]
- 35.Tsai SL, Huang GT, Yang PM, Sheu JC, Sung JL, Chen DS. Plasma Des-γ-carboxyprothrombin in the early stage of hepatocellular carcinoma. Hepatology 1990;11(3):481–7. [DOI] [PubMed] [Google Scholar]
- 36.Kumada T, Toyoda H, Tada T, Kiriyama S, Tanikawa M, Hisanaga Y, et al. High-sensitivity Lens culinaris agglutinin-reactive alpha-fetoprotein assay predicts early detection of hepatocellular carcinoma. J Gastroenterol 2014;49(3):555–63 doi 10.1007/s00535-013-0883-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Fedarko NS, Jain A, Karadag A, Van Eman MR, Fisher LW. Elevated serum bone sialoprotein and osteopontin in colon, breast, prostate, and lung cancer. Clinical Cancer Research 2001;7(12):4060–6. [PubMed] [Google Scholar]
- 38.Shang S, Plymoth A, Ge S, Feng Z, Rosen HR, Sangrajrang S, et al. Identification of osteopontin as a novel marker for early hepatocellular carcinoma. Hepatology 2012;55(2):483–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Zhao J, Dong L, Lu B, Wu G, Xu D, Chen J, et al. Down-regulation of osteopontin suppresses growth and metastasis of hepatocellular carcinoma via induction of apoptosis. Gastroenterology 2008;135(3):956–68. [DOI] [PubMed] [Google Scholar]
- 40.Wan H-G, Xu H, Gu Y-M, Wang H, Xu W, Zu M-H. Comparison osteopontin vs AFP for the diagnosis of HCC: a meta-analysis. Clinics and research in hepatology and gastroenterology 2014;38(6):706–14. [DOI] [PubMed] [Google Scholar]
- 41.Sun T, Tang Y, Sun D, Bu Q, Li P. Osteopontin versus alpha-fetoprotein as a diagnostic marker for hepatocellular carcinoma: a meta-analysis. Onco Targets Ther 2018;11:8925–35 doi 10.2147/OTT.S186230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Koide N, Hada H, Shinji T, Ujike K, Hirasaki S, Yumoto Y, et al. Expression of the midkine gene in human hepatocellular carcinomas. Hepato-gastroenterology 1999;46(30):3189–96. [PubMed] [Google Scholar]
- 43.Vongsuvanh R, van der Poorten D, Iseli T, Strasser SI, McCaughan GW, George J. Midkine increases diagnostic yield in AFP negative and NASH-related hepatocellular carcinoma. PloS one 2016;11(5):e0155800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Zhu W-W, Guo J-J, Guo L, Jia H-L, Zhu M, Zhang J-B, et al. Evaluation of midkine as a diagnostic serum biomarker in hepatocellular carcinoma. Clinical Cancer Research 2013;19(14):3944–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Shen Q, Fan J, Yang X-R, Tan Y, Zhao W, Xu Y, et al. Serum DKK1 as a protein biomarker for the diagnosis of hepatocellular carcinoma: a large-scale, multicentre study. The lancet oncology 2012;13(8):817–26. [DOI] [PubMed] [Google Scholar]
- 46.Jang ES, Jeong SH, Kim JW, Choi YS, Leissner P, Brechot C. Diagnostic Performance of Alpha-Fetoprotein, Protein Induced by Vitamin K Absence, Osteopontin, Dickkopf-1 and Its Combinations for Hepatocellular Carcinoma. PLoS One 2016;11(3):e0151069 doi 10.1371/journal.pone.0151069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Yao M, Yao D-F, Bian Y-Z, Wu W, Yan X-D, Yu D-D, et al. Values of circulating GPC-3 mRNA and alpha-fetoprotein in detecting patients with hepatocellular carcinoma. Hepatobiliary & Pancreatic Diseases International 2013;12(2):171–9. [DOI] [PubMed] [Google Scholar]
- 48.Yao M, Yao D-F, Bian Y-Z, Zhang C-G, Qiu L-W, Wu W, et al. Oncofetal antigen glypican-3 as a promising early diagnostic marker for hepatocellular carcinoma. Hepatobiliary & Pancreatic Diseases International 2011;10(3):289–94. [DOI] [PubMed] [Google Scholar]
- 49.Capurro M, Wanless IR, Sherman M, Deboer G, Shi W, Miyoshi E, et al. Glypican-3: a novel serum and histochemical marker for hepatocellular carcinoma. Gastroenterology 2003;125(1):89–97 doi 10.1016/s0016-5085(03)00689-9. [DOI] [PubMed] [Google Scholar]
- 50.Jia X, Liu J, Gao Y, Huang Y, Du Z. Diagnosis accuracy of serum glypican-3 in patients with hepatocellular carcinoma: a systematic review with meta-analysis. Arch Med Res 2014;45(7):580–8 doi 10.1016/j.arcmed.2014.11.002. [DOI] [PubMed] [Google Scholar]
- 51.Shafizadeh N, Ferrell LD, Kakar S. Utility and limitations of glypican-3 expression for the diagnosis of hepatocellular carcinoma at both ends of the differentiation spectrum. Modern Pathology 2008;21(8):1011–8. [DOI] [PubMed] [Google Scholar]
- 52.Qiao S-S, Cui Z, Gong L, Han H, Chen P-C, Guo L-M, et al. Simultaneous measurements of serum AFP, GPC-3 and HCCR for diagnosing hepatocellular carcinoma. Hepato-gastroenterology 2010;58(110-111):1718–24. [DOI] [PubMed] [Google Scholar]
- 53.Xing H, Qiu H, Ding X, Han J, Li Z, Wu H, et al. Clinical performance of alpha-L-fucosidase for early detection of hepatocellular carcinoma. Biomark Med 2019;13(7):545–55 doi 10.2217/bmm-2018-0414. [DOI] [PubMed] [Google Scholar]
- 54.Giardina M, Matarazzo M, Varriale A, Morante R, Napoli A, Martino R. Serum alpha-L-fucosidase. A useful marker in the diagnosis of hepatocellular carcinoma. Cancer 1992;70(5):1044–8. [DOI] [PubMed] [Google Scholar]
- 55.Ishizuka H, Nakayama T, Matsuoka S, Gotoh I, Ogawa M, Suzuki K, et al. Prediction of the development of hepato-cellular-carcinoma in patients with liver cirrhosis by the serial determinations of serum alpha-L-fucosidase activity. Internal medicine 1999;38(12):927–31. [DOI] [PubMed] [Google Scholar]
- 56.Block TM, Comunale MA, Lowman M, Steel LF, Romano PR, Fimmel C, et al. Use of targeted glycoproteomics to identify serum glycoproteins that correlate with liver cancer in woodchucks and humans. Proc Natl Acad Sci U S A 2005;102(3):779–84 doi 0408928102 [pii] 10.1073/pnas.0408928102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Ismail MM, Morsi HK, Abdulateef NA, Noaman MK, Abou El-Ella GA. Evaluation of prothrombin induced by vitamin K absence, macrophage migration inhibitory factor and Golgi protein-73 versus alpha fetoprotein for hepatocellular carcinoma diagnosis and surveillance. Scandinavian Journal of Clinical and Laboratory Investigation 2017;77(3):175–83. [DOI] [PubMed] [Google Scholar]
- 58.Marrero JA, Romano PR, Nikolaeva O, Steel L, Mehta A, Fimmel CJ, et al. GP73, a resident Golgi glycoprotein, is a novel serum marker for hepatocellular carcinoma. Journal of hepatology 2005;43(6):1007–12. [DOI] [PubMed] [Google Scholar]
- 59.Zhou Y, Yin X, Ying J, Zhang B. Golgi protein 73 versus alpha-fetoprotein as a biomarker for hepatocellular carcinoma: a diagnostic meta-analysis. BMC Cancer 2012;12:17 doi 10.1186/1471-2407-12-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Giannelli G, Marinosci F, Trerotoli P, Volpe A, Quaranta M, Dentico P, et al. SCCA antigen combined with alpha-fetoprotein as serologic markers of HCC. International journal of cancer 2005;117(3):506–9. [DOI] [PubMed] [Google Scholar]
- 61.Pozzan C, Cardin R, Piciocchi M, Cazzagon N, Maddalo G, Vanin V, et al. Diagnostic and prognostic role of SCCA-IgM serum levels in hepatocellular carcinoma (HCC). Journal of gastroenterology and hepatology 2014;29(8):1637–44. [DOI] [PubMed] [Google Scholar]
- 62.Witjes CD, van Aalten SM, Steyerberg EW, Borsboom GJ, de Man RA, Verhoef C, et al. Recently introduced biomarkers for screening of hepatocellular carcinoma: a systematic review and meta-analysis. Hepatology international 2013:1–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Liu CH, Gil-Gomez A, Ampuero J, Romero-Gomez M. Diagnostic accuracy of SCCA and SCCA-IgM for hepatocellular carcinoma: A meta-analysis. Liver Int 2018;38(10):1820–31 doi 10.1111/liv.13867. [DOI] [PubMed] [Google Scholar]
- 64.Comunale MA, Lowman M, Long RE, Krakover J, Philip R, Seeholzer S, Evans AA, Hann HWL, Block TM, Mehta AS Proteomic analysis of serum associated fucosylated glycoproteins in the development of primary hepatocellular carcinoma. Journal of Proteome Research 2006;6(5):308–15. [DOI] [PubMed] [Google Scholar]
- 65.Comunale MA, Rodemich-Betesh L, Hafner J, Wang M, Norton P, Di Bisceglie AM, et al. Linkage Specific Fucosylation of Alpha-1-Antitrypsin in Liver Cirrhosis and Cancer Patients: Implications for a Biomarker of Hepatocellular Carcinoma. PLoS ONE 2010;5(8):e12419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Comunale MA, Wang M, Hafner J, Krakover J, Rodemich L, Kopenhaver B, et al. Identification and development of fucosylated glycoproteins as biomarkers of primary hepatocellular carcinoma. J Proteome Res 2009;8(2):595–602 doi 10.1021/pr800752c10.1021/pr800752c [pii]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Comunale MA, Wang M, Rodemich-Betesh L, Hafner J, Lamontagne A, Klein A, et al. Novel changes in glycosylation of serum Apo-J in patients with hepatocellular carcinoma. Cancer epidemiology, biomarkers & prevention : a publication of the American Association for Cancer Research, cosponsored by the American Society of Preventive Oncology 2011;20(6):1222–9 doi 10.1158/1055-9965.EPI-10-1047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Drake RR, Schwegler EE, Malik G, Diaz J, Block T, Mehta A, et al. Lectin capture strategies combined with mass spectrometry for the discovery of serum glycoprotein biomarkers. Mol Cell Proteomics 2006;5(10):1957–67 doi M600176-MCP200 [pii] 10.1074/mcp.M600176-MCP200. [DOI] [PubMed] [Google Scholar]
- 69.Marrero JA, Romano PR, Nikolaeva O, Steel L, Mehta A, Fimmel CJ, et al. GP73, a resident Golgi glycoprotein, is a novel serum marker for hepatocellular carcinoma. Journal of hepatology 2005;43(6):1007–12 doi 10.1016/j.jhep.2005.05.028. [DOI] [PubMed] [Google Scholar]
- 70.Morota K, Nakagawa M, Sekiya R, Hemken PM, Sokoll LJ, Elliott D, et al. A comparative evaluation of Golgi protein-73, fucosylated hemopexin, alpha-fetoprotein, and PIVKA-II in the serum of patients with chronic hepatitis, cirrhosis, and hepatocellular carcinoma. Clin Chem Lab Med 2011. doi 10.1515/CCLM.2011.097. [DOI] [PubMed] [Google Scholar]
- 71.Wang M, Long RE, Comunale MA, Junaidi O, Marrero J, Di Bisceglie AM, et al. Novel fucosylated biomarkers for the early detection of hepatocellular carcinoma. Cancer Epidemiol Biomarkers Prev 2009;18(6):1914–21 doi 10.1158/1055-9965.EPI-08-0980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.White KY, Rodemich L, Nyalwidhe JO, Comunale MA, Clements MA, Lance RS, et al. Glycomic characterization of prostate-specific antigen and prostatic acid phosphatase in prostate cancer and benign disease seminal plasma fluids. J Proteome Res 2009;8(2):620–30 doi 10.1021/pr800754510.1021/pr8007545 [pii]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Debruyne EN, Vanderschaeghe D, Van Vlierberghe H, Vanhecke A, Callewaert N, Delanghe JR. Diagnostic value of the hemopexin N-glycan profile in hepatocellular carcinoma patients. Clin Chem 2010;56(5):823–31 doi clinchem.2009.139295 [pii] 10.1373/clinchem.2009.139295. [DOI] [PubMed] [Google Scholar]
- 74.Matsumoto K, Maeda Y, Kato S, Yuki H. Alteration of asparagine-linked glycosylation in serum transferrin of patients with hepatocellular carcinoma. Clinica chimica acta; international journal of clinical chemistry 1994;224(1):1–8. [DOI] [PubMed] [Google Scholar]
- 75.Morota K, Nakagawa M, Sekiya R, Hemken PM, Sokoll LJ, Elliott D, et al. A comparative evaluation of Golgi protein-73, fucosylated hemopexin, alpha-fetoprotein, and PIVKA-II in the serum of patients with chronic hepatitis, cirrhosis, and hepatocellular carcinoma. Clinical chemistry and laboratory medicine : CCLM / FESCC 2011;49(4):711–8 doi 10.1515/CCLM.2011.097. [DOI] [PubMed] [Google Scholar]
- 76.Kobayashi S, Nouso K, Kinugasa H, Takeuchi Y, Tomoda T, Miyahara K, et al. Clinical utility of serum fucosylated hemopexin in Japanese patients with hepatocellular carcinoma. Hepatol Res 2012;42(12):1187–95 doi 10.1111/j.1872-034X.2012.01044.x. [DOI] [PubMed] [Google Scholar]
- 77.Ahn YH, Shin PM, Oh NR, Park GW, Kim H, Yoo JS. A lectin-coupled, targeted proteomic mass spectrometry (MRM MS) platform for identification of multiple liver cancer biomarkers in human plasma. J Proteomics 2012. doi 10.1016/j.jprot.2012.06.027. [DOI] [PubMed] [Google Scholar]
- 78.Comunale MA, Wang M, Hafner J, Krakover J, Rodemich L, Kopenhaver B, et al. Identification and development of fucosylated glycoproteins as biomarkers of primary hepatocellular carcinoma. Journal of Proteome Research 2009;8(2):595–602 doi 10.1021/pr800752c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Naitoh A, Aoyagi Y, Asakura H. Highly enhanced fucosylation of serum glycoproteins in patients with hepatocellular carcinoma. Journal of gastroenterology and hepatology 1999;14(5):436–45. [DOI] [PubMed] [Google Scholar]
- 80.Ahn YH, Shin PM, Ji ES, Kim H, Yoo JS. A lectin-coupled, multiple reaction monitoring based quantitative analysis of human plasma glycoproteins by mass spectrometry. Anal Bioanal Chem 2012;402(6):2101–12 doi 10.1007/s00216-011-5646-3. [DOI] [PubMed] [Google Scholar]
- 81.Block TM, Comunale MA, Lowman M, Steel LF, Romano PR, Fimmel C, et al. Use of targeted glycoproteomics to identify serum glycoproteins that correlate with liver cancer in woodchucks and humans. Proceedings of the National Academy of Sciences of the United States of America 2005;102(3):779–84 doi 10.1073/pnas.0408928102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Comunale MA, Lowman M, Long RE, Krakover J, Philip R, Seeholzer S, et al. Proteomic analysis of serum associated fucosylated glycoproteins in the development of primary hepatocellular carcinoma. J Proteome Res 2006;5(2):308–15 doi 10.1021/pr050328x. [DOI] [PubMed] [Google Scholar]
- 83.Chen R, Wang F, Tan Y, Sun Z, Song C, Ye M, et al. Development of a combined chemical and enzymatic approach for the mass spectrometric identification and quantification of aberrant N-glycosylation. J Proteomics 2012;75(5):1666–74 doi 10.1016/j.jprot.2011.12.015. [DOI] [PubMed] [Google Scholar]
- 84.Liu Y, He J, Li C, Benitez R, Fu S, Marrero J, et al. Identification and confirmation of biomarkers using an integrated platform for quantitative analysis of glycoproteins and their glycosylations. J Proteome Res 2010;9(2):798–805 doi 10.1021/pr900715p. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Chandler KB, Pompach P, Goldman R, Edwards N. Exploring site-specific N-glycosylation microheterogeneity of haptoglobin using glycopeptide CID tandem mass spectra and glycan database search. Journal of Proteome Research 2013;12(8):3652–66 doi 10.1021/pr400196s. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Pompach P, Brnakova Z, Sanda M, Wu J, Edwards N, Goldman R. Site-specific glycoforms of haptoglobin in liver cirrhosis and hepatocellular carcinoma. Molecular & cellular proteomics : MCP 2013;12(5):1281–93 doi 10.1074/mcp.M112.023259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Sanda M, Pompach P, Brnakova Z, Wu J, Makambi K, Goldman R. Quantitative liquid chromatography-mass spectrometry-multiple reaction monitoring (LC-MS-MRM) analysis of site-specific glycoforms of haptoglobin in liver disease. Molecular & cellular proteomics : MCP 2013;12(5):1294–305 doi 10.1074/mcp.M112.023325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Zhu J, Lin Z, Wu J, Yin H, Dai J, Feng Z, et al. Analysis of serum haptoglobin fucosylation in hepatocellular carcinoma and liver cirrhosis of different etiologies. Journal of Proteome Research 2014;13(6):2986–97 doi 10.1021/pr500128t. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Ahn JM, Sung HJ, Yoon YH, Kim BG, Yang WS, Lee C, et al. Integrated glycoproteomics demonstrates fucosylated serum paraoxonase 1 alterations in small cell lung cancer. Molecular & cellular proteomics : MCP 2014;13(1):30–48 doi 10.1074/mcp.M113.028621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Sun C, Chen P, Chen Q, Sun L, Kang X, Qin X, et al. Serum paraoxonase 1 heteroplasmon, a fucosylated, and sialylated glycoprotein in distinguishing early hepatocellular carcinoma from liver cirrhosis patients. Acta Biochim Biophys Sin (Shanghai) 2012;44(9):765–73 doi 10.1093/abbs/gms055. [DOI] [PubMed] [Google Scholar]
- 91.West CA, Wang M, Herrera H, Liang H, Black A, Angel PM, et al. N-Linked Glycan Branching and Fucosylation Are Increased Directly in Hcc Tissue As Determined through in Situ Glycan Imaging. J Proteome Res 2018;17(10):3454–62 doi 10.1021/acs.jproteome.8b00323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Wang M, Sanda M, Comunale MA, Herrera H, Swindell C, Kono Y, et al. Changes in the Glycosylation of Kininogen and the Development of a Kininogen-Based Algorithm for the Early Detection of HCC. Cancer Epidemiol Biomarkers Prev 2017;26(5):795–803 doi 10.1158/1055-9965.EPI-16-0974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Wang M, Shen J, Herrera H, Singal A, Swindell C, Renquan L, et al. Biomarker analysis of fucosylated kininogen through depletion of lectin reactive heterophilic antibodies in hepatocellular carcinoma. J Immunol Methods 2018;462:59–64 doi 10.1016/j.jim.2018.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Wang M, Sanda M, Comunale MA, Herrera H, Swindell C, Kono Y, et al. Changes in the Glycosylation of Kininogen and the Development of a Kininogen-Based Algorithm for the Early Detection of HCC. Cancer Epidemiology and Prevention Biomarkers 2017;26(5):795–803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Ang IL, Poon TC, Lai PB, Chan AT, Ngai SM, Hui AY, et al. Study of serum haptoglobin and its glycoforms in the diagnosis of hepatocellular carcinoma: a glycoproteomic approach. J Proteome Res 2006;5(10):2691–700 doi 10.1021/pr060109r. [DOI] [PubMed] [Google Scholar]
- 96.Darebna P, Novak P, Kucera R, Topolcan O, Sanda M, Goldman R, et al. Changes in the expression of N- and O-glycopeptides in patients with colorectal cancer and hepatocellular carcinoma quantified by full-MS scan FT-ICR and multiple reaction monitoring. J Proteomics 2017;153:44–52 doi 10.1016/j.jprot.2016.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Pompach P, Ashline DJ, Brnakova Z, Benicky J, Sanda M, Goldman R. Protein and site specificity of fucosylation in liver-secreted glycoproteins. J Proteome Res 2014;13(12):5561–9 doi 10.1021/pr5005482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Pompach P, Brnakova Z, Sanda M, Wu J, Edwards N, Goldman R. Site-specific glycoforms of haptoglobin in liver cirrhosis and hepatocellular carcinoma. Mol Cell Proteomics 2013;12(5):1281–93 doi 10.1074/mcp.M112.023259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Lamontagne A, Long RE, Comunale MA, Hafner J, Rodemich-Betesh L, Wang M, et al. Altered functionality of anti-bacterial antibodies in patients with chronic hepatitis C virus infection. PLoS One 2013;8(6):e64992 doi 10.1371/journal.pone.0064992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Mehta AS, Long RE, Comunale MA, Wang M, Rodemich L, Krakover J, et al. Increased levels of galactose-deficient anti-Gal immunoglobulin G in the sera of hepatitis C virus-infected individuals with fibrosis and cirrhosis. Journal of virology 2008;82(3):1259–70 doi 10.1128/JVI.01600-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Wang M, Long RE, Comunale MA, Junaidi O, Marrero J, Di Bisceglie AM, et al. Novel fucosylated biomarkers for the early detection of hepatocellular carcinoma. Cancer epidemiology, biomarkers & prevention : a publication of the American Association for Cancer Research, cosponsored by the American Society of Preventive Oncology 2009;18(6):1914–21 doi 10.1158/1055-9965.EPI-08-0980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Wang M, Comunale MA, Herrera H, Betesh L, Kono Y, Mehta A. Identification of IgM as a contaminant in lectin-FLISA assays for HCC detection. Biochem Biophys Res Commun 2016;476(3):140–5 doi 10.1016/j.bbrc.2016.05.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Zhu J, Warner E, Parikh ND, Lubman DM. Glycoproteomic markers of hepatocellular carcinoma-mass spectrometry based approaches. Mass Spectrom Rev 2019;38(3):265–90 doi 10.1002/mas.21583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Black AP, Angel PM, Drake RR, Mehta AS. Antibody Panel Based N-Glycan Imaging for N-Glycoprotein Biomarker Discovery. Curr Protoc Protein Sci 2019;98(1):e99 doi 10.1002/cpps.99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Black AP, Liang H, West CA, Wang M, Herrera HP, Haab BB, et al. A Novel Mass Spectrometry Platform for Multiplexed N-Glycoprotein Biomarker Discovery from Patient Biofluids by Antibody Panel Based N-Glycan Imaging. Anal Chem 2019;91(13):8429–35 doi 10.1021/acs.analchem.9b01445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Zhang Y, Li T, Qiu Y, Zhang T, Guo P, Ma X, et al. Serum microRNA panel for early diagnosis of the onset of hepatocellular carcinoma. Medicine 2017;96(2). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Zekri AN, Youssef AS, El-Desouky ED, Ahmed OS, Lotfy MM, Nassar AA, et al. Serum microRNA panels as potential biomarkers for early detection of hepatocellular carcinoma on top of HCV infection. Tumour Biol 2016;37(9):12273–86 doi 10.1007/s13277-016-5097-8. [DOI] [PubMed] [Google Scholar]
- 108.Shi KQ, Lin Z, Chen XJ, Song M, Wang YQ, Cai YJ, et al. Hepatocellular carcinoma associated microRNA expression signature: integrated bioinformatics analysis, experimental validation and clinical significance. Oncotarget 2015;6(28):25093–108 doi 10.18632/oncotarget.4437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Ambros V, Bartel B, Bartel DP, Burge CB, Carrington JC, Chen X, et al. A uniform system for microRNA annotation. RNA 2003;9(3):277–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Zou Q, Mao Y, Hu L, Wu Y, Ji Z. miRClassify: an advanced web server for miRNA family classification and annotation. Comput Biol Med 2014;45:157–60 doi 10.1016/j.compbiomed.2013.12.007. [DOI] [PubMed] [Google Scholar]
- 111.Ally A, Balasundaram M, Carlsen R, Chuah E, Clarke A, Dhalla N, et al. Comprehensive and integrative genomic characterization of hepatocellular carcinoma. Cell 2017;169(7):1327–41. e23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Wheeler DA, Roberts LR, Network CGAR. Comprehensive and integrative genomic characterization of hepatocellular carcinoma. Cell 2017;169(7):1327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Nault JC, Mallet M, Pilati C, Calderaro J, Bioulac-Sage P, Laurent C, et al. High frequency of telomerase reverse-transcriptase promoter somatic mutations in hepatocellular carcinoma and preneoplastic lesions. Nature communications 2013;4:2218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Llovet JM, Zucman-Rossi J, Pikarsky E, Sangro B, Schwartz M, Sherman M, et al. Hepatocellular carcinoma. Nature reviews Disease primers 2016;2:16018. [DOI] [PubMed] [Google Scholar]
- 115.Su Y-H, Lin SY, Song W, Jain S. DNA markers in molecular diagnostics for hepatocellular carcinoma. Expert review of molecular diagnostics 2014;14(7):803–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Jiao J, Watt GP, Stevenson HL, Calderone TL, Fisher-Hoch SP, Ye Y, et al. Telomerase reverse transcriptase mutations in plasma DNA in patients with hepatocellular carcinoma or cirrhosis: prevalence and risk factors. Hepatology communications 2018;2(6):718–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Xu RH, Wei W, Krawczyk M, Wang W, Luo H, Flagg K, et al. Circulating tumour DNA methylation markers for diagnosis and prognosis of hepatocellular carcinoma. Nat Mater 2017;16(11):1155–61 doi 10.1038/nmat4997. [DOI] [PubMed] [Google Scholar]
- 118.Chalasani NP, Ramasubramanian T, Bruinsma JJ, Allawi HT, Olson M, Roberts LR, et al. COMBINED METHYLATED DNA AND PROTEIN MARKERS: AN ACCURATE BLOOD-BASED TEST FOR EARLY-STAGE DETECTION OF HEPATOCELLULAR CARCINOMA. 2019. WILEY; 111 RIVER ST, HOBOKEN 07030-5774, NJ USA: p 72A–3A. [Google Scholar]
- 119.Craig AJ, von Felden J, Garcia-Lezana T, Sarcognato S, Villanueva A. Tumour evolution in hepatocellular carcinoma. Nat Rev Gastroenterol Hepatol 2019. doi 10.1038/s41575-019-0229-4. [DOI] [PubMed] [Google Scholar]
- 120.Johnson PJ, Pirrie SJ, Cox TF, Berhane S, Teng M, Palmer D, et al. The detection of hepatocellular carcinoma using a prospectively developed and validated model based on serological biomarkers. Cancer Epidemiol Biomarkers Prev 2014;23(1):144–53 doi 10.1158/1055-9965.EPI-13-0870. [DOI] [PubMed] [Google Scholar]
- 121.Berhane S, Toyoda H, Tada T, Kumada T, Kagebayashi C, Satomura S, et al. Role of the GALAD and BALAD-2 serologic models in diagnosis of hepatocellular carcinoma and prediction of survival in patients. Clinical Gastroenterology and Hepatology 2016;14(6):875–86. e6. [DOI] [PubMed] [Google Scholar]
- 122.Best J, Bechmann LP, Sowa JP, Sydor S, Dechene A, Pflanz K, et al. GALAD Score Detects Early Hepatocellular Carcinoma in an International Cohort of Patients With Nonalcoholic Steatohepatitis. Clin Gastroenterol Hepatol 2019. doi 10.1016/j.cgh.2019.11.012. [DOI] [PubMed] [Google Scholar]
- 123.Wang M, Devarajan K, Singal AG, Marrero JA, Dai J, Feng Z, et al. The Doylestown Algorithm: A Test to Improve the Performance of AFP in the Detection of Hepatocellular Carcinoma. Cancer Prev Res (Phila) 2016;9(2):172–9 doi 10.1158/1940-6207.CAPR-15-0186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Tayob N, Christie I, Richardson P, Feng Z, White DL, Davila J, et al. Validation of the hepatocellular carcinoma early detection screening (HES) algorithm in a cohort of veterans with cirrhosis. Clinical Gastroenterology and Hepatology 2019;17(9):1886–93. e5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Feng Z, Marrero JA, Khaderi S, Singal AG, Kanwal F, Loo N, et al. Design of the Texas Hepatocellular Carcinoma Consortium Cohort Study. Am J Gastroenterol 2019;114(3):530–2 doi 10.14309/ajg.0000000000000068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Younossi Z, Stepanova M, Ong JP, Jacobson IM, Bugianesi E, Duseja A, et al. Nonalcoholic Steatohepatitis Is the Fastest Growing Cause of Hepatocellular Carcinoma in Liver Transplant Candidates. Clin Gastroenterol Hepatol 2019;17(4):748–55 e3 doi 10.1016/j.cgh.2018.05.057. [DOI] [PubMed] [Google Scholar]
- 127.Hoshida Y, Villanueva A, Kobayashi M, Peix J, Chiang DY, Camargo A, et al. Gene expression in fixed tissues and outcome in hepatocellular carcinoma. New England Journal of Medicine 2008;359(19):1995–2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Goossens N, Singal AG, King LY, Andersson KL, Fuchs BC, Besa C, et al. Cost-Effectiveness of Risk Score–Stratified Hepatocellular Carcinoma Screening in Patients with Cirrhosis. Clinical and translational gastroenterology 2017;8(6):e101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Besa C, Lewis S, Pandharipande PV, Chhatwal J, Kamath A, Cooper N, et al. Hepatocellular carcinoma detection: diagnostic performance of a simulated abbreviated MRI protocol combining diffusion-weighted and T1-weighted imaging at the delayed phase post gadoxetic acid. Abdominal Radiology 2017;42(1):179–90. [DOI] [PubMed] [Google Scholar]
- 130.Mokrane F-Z, Lu L, Vavasseur A, Otal P, Peron J-M, Luk L, et al. Radiomics machine-learning signature for diagnosis of hepatocellular carcinoma in cirrhotic patients with indeterminate liver nodules. European radiology 2020;30(1):558–70. [DOI] [PubMed] [Google Scholar]


