Abstract
Nutritional interventions are not commonly a standard of care in rehabilitation interventions. A nutritional approach has the potential to be a low-cost, high-volume strategy that complements the existing standard of care. In this commentary, our aim is to provide an evidence-based, practical guide for athletes with injuries treated surgically or conservatively, along with healing and rehabilitation considerations. Injuries are a normal and expected part of exercise participation. Regardless of severity, an injury typically results in the athlete's short- or long-term removal from participation. Nutritional interventions may augment the recovery process and support optimal healing; therefore, incorporating nutritional strategies is important at each stage of the healing process. Preoperative nutrition and nutritional demands during rehabilitation are key factors to consider. The physiological response to wounds, immobilization, and traumatic brain injuries may be improved by optimizing macronutrient composition, caloric consumption, and nutrient timing and using select dietary supplements. Previous research supports practical nutrition recommendations to reduce surgical complications, minimize deficits after immobilization, and maximize the chance of safe return to play. These recommendations include identifying the individual's caloric requirements to ensure that energy needs are being met. A higher protein intake, with special attention to evenly distributed consumption throughout the day, will help to minimize loss of muscle and strength during immobilization. Dietary-supplement strategies may be useful when navigating the challenges of appropriate caloric intake and timing and a reduced appetite. The rehabilitation process also requires a strong nutritional plan to enhance recovery from injury. Athletic trainers, physical therapists, and other health care professionals should provide basic nutritional recommendations during rehabilitation, discuss the timing of meals with respect to therapy, and refer the patient to a registered dietitian if warranted. Because nutrition plays an essential role in injury recovery and rehabilitation, nutritional interventions should become a component of standard-of-care practice after injury. In this article, we address best practices for implementing nutritional strategies among patients with athletic injuries.
The economic burden of injury is high, with more than $9 billion spent annually on injury and rehabilitation in young adults (17–44 years).1 Injuries are the leading contributor to medical costs in this age group. Beyond monetary costs, athletic injuries also result in significant mental and physical burdens. Health-related rehabilitation interventions are most cost effective when practitioners adopt an interdisciplinary approach that encompasses low-cost, high-volume services.2 Nutritional interventions are not commonly used as standard of care in rehabilitation interventions. However, numerous nutritional approaches are available as potential low-cost, high-volume strategies to complement the existing standard of care, particularly for patients pursuing musculoskeletal rehabilitation.
Injuries are a normal and expected part of exercise participation. Regardless of severity, injuries typically result in short- or long-term removal from participation. The initial healing stage that occurs immediately after trauma involves an inflammatory response and may last from a few hours to several days, depending on the nature of the injury.3,4 This inflammatory response is essential to initiate optimal healing; as a result, nutritional interventions intended to control inflammation in this acute phase may be contraindicated for optimal healing.3 However, minimizing prolonged or excessive inflammation by ingesting dietary supplements may enhance healing and accelerate a safe return to play. Patients with injuries that require immobilization or surgery deserve special nutritional considerations due to the large physiological demand of wound healing (Figure 1). Even athletes with more minor injuries, such as muscular strains, stress fractures, or ankle sprains, warrant special nutritional attention. In a different category, traumatic brain injuries (TBIs)—even mild cases—also need nutritional attention. Nutritional recommendations for patients with TBI constitute a growing field, with new research emerging rapidly. In this article, we will present an evidence-based, practical guide for nutritional considerations of injuries, including wound healing, immobilization, and TBI. Specifically, nutritional considerations for athletes with injuries treated surgically or conservatively who are in the preoperative, postoperative, or healing phase will be addressed.
Figure 1.
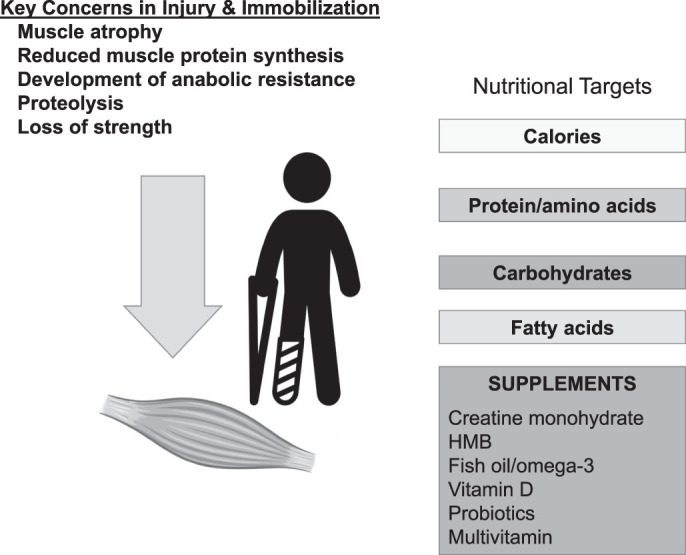
Physiological and metabolic demands after injury can be addressed with key nutritional targets; elevated caloric demand, increased protein, glucose regulation with complex carbohydrates, and essential fatty acids should be considered. In addition, based on the evidence, specific dietary supplements may mitigate the negative effects of surgery and immobilization and support faster healing. Abbreviation: HMB, β-hydroxy-β-methylbutyrate.
PREOPERATIVE NUTRITIONAL CONSIDERATIONS
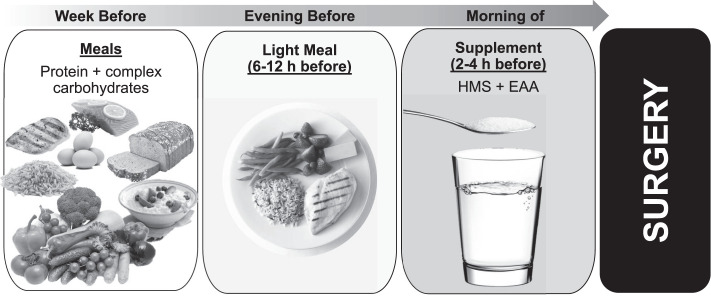
Once an injury is sustained, a cascade of inflammatory, immune, and metabolic responses is activated, resulting in a hypermetabolic state. To support this hypermetabolic state and the injury-healing process, significant macro- and micronutrient support is required. The nutritional needs of a patient before surgery are often omitted from standard presurgical guidelines; preoperative attention to nutrition is typically focused on recommendations to fast from food and fluid beginning at midnight of the day of the operation.5,6 This food and fluid fast ensures an empty stomach at the time of anesthesia and minimizes the risk of pulmonary aspiration.5 More recently, accumulating data have led to suggestions that less strict fasting routines can be adopted without placing patients at risk.6,7 Feeding patients with a high-carbohydrate beverage immediately before surgery was safe, reduced catabolic surgical stress, and might have enhanced postoperative outcomes.6,8 Newer guidelines9 advise that patients fast after a light meal 6 hours before and from clear liquids 2 hours before surgery. With these guidelines in mind, it may be possible to take advantage of this preoperative time to maximize recovery potential.
Preoperative carbohydrates and insulin have been shown to reduce insulin resistance, metabolic stress, and inflammation and prevent hypoglycemia postsurgery.10 Preoperative glycemic control, particularly using carbohydrate feeding, improved surgical outcomes by reducing the catabolic cascade and insulin resistance that occurred with surgical stress.11,12 Specifically, preoperative consumption of 100 g of glucose in an oral solution the night before surgery and 50 g 2 hours before surgery was effective in reducing postoperative insulin resistance.13 Another study14 demonstrated that preoperative consumption of a 100-g carbohydrate-containing drink the evening before the surgery and 50 g 2 to 3 hours before surgery attenuated depletion of muscle mass after surgery. Researchers15 who conducted a perioperative nutritional intervention involving an oral supplement (1.5 kcal + 0.05 g protein/mL) found less postoperative weight loss when the supplement was given perioperatively versus preoperatively or postoperatively; in addition, fewer minor complications occurred in the perioperative- and postoperative-supplement groups than in the control group. Hydrothermally modified waxy maize starch is a low-glycemic, slower digesting complex carbohydrate shown to be effective in stabilizing glucose levels and preventing hypoglycemia over extended periods of time.16 Originally developed to help prevent overnight hypoglycemia in children with glycogen storage disease17 and more recently embraced by the endurance athlete community, modified waxy maize starch is an easy-to-drink clear liquid that could be used as a presurgery supplement to help sustain glucose levels during surgery and possibly prevent postsurgery hypoglycemia (Figure 2).
Figure 2.
Before surgery, increasing the carbohydrates in the diet, particularly from complex sources and proteins, is beneficial. The night before surgery should include a well-rounded meal with slow-releasing carbohydrates and proteins. Two to 4 hours before surgery, a clear liquid consisting of a high molecular weight starch (HMS) and essential amino acids molecular-weight essential amino acids (EAA) is recommended.
ASSESSING POSTOPERATIVE AND REHABILITATIVE NUTRITIONAL NEEDS
Most often, an injury results in immobilization or disuse. Under these circumstances, muscle tissue can be lost after as little as 36 hours of inactivity,18 with global gene expression alterations after 48 hours and a significant loss of muscle tissue within 5 days of inactivity.19 At a fundamental level, the goal of rehabilitative nutrition is to provide enough calories and protein to aid in wound healing and prevent a loss of lean body mass (LBM). Other goals include modulating inflammatory and immune responses, enhancing control of blood glucose levels, and providing macro- and micronutrients to optimize the recovery and healing process.3 During the rehabilitative phase, a stress response initiates an overall increase in energy demand.5 The resulting net catabolic response upregulates the use of amino acids and proteins for healing and concomitantly decreases LBM. When caring for a patient who is recovering from injury, it is important to identify calorie needs, protein needs, and micronutrient requirements (Figure 3).
Figure 3.
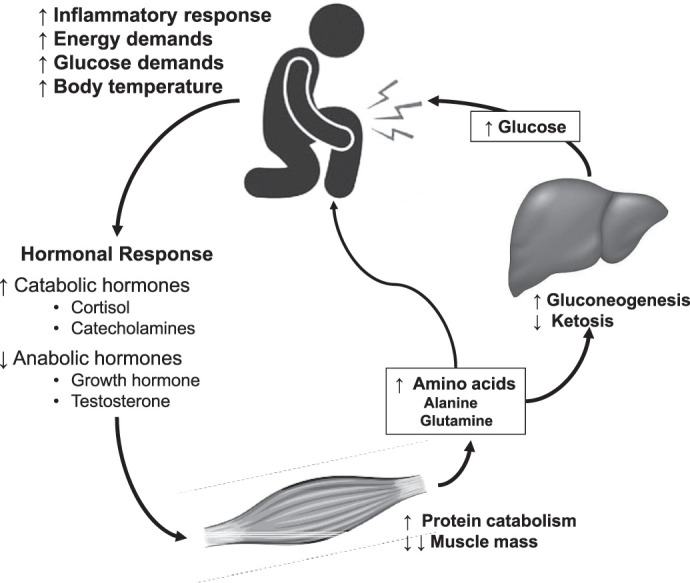
Injury and surgery result in a significant stress response, which highlights the potential for nutritional support. The demand for glucose and amino acids increases, which initiates a hormonal response resulting in a catabolic environment, leading to large losses in lean body mass. Adapted from Demling.23
Calorie Needs
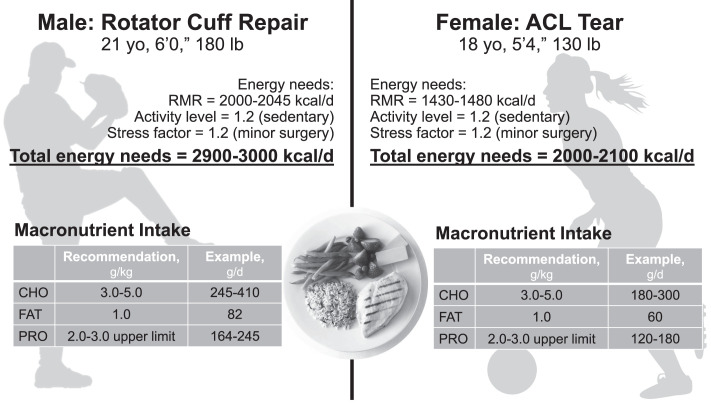
Nutrition is an important aspect of general athlete health and performance. Knowledge of basic energy recommendations may provide additional information regarding an athlete's unique needs before and after injury.20,21 Whether an injury is treated surgically or conservatively, the patient's basal metabolic rate (BMR) increases to match cellular turnover during recovery. To identify the individual's calorie needs, the BMR should be either measured via indirect calorimetry or estimated using predictive equations that reflect the energy required to maintain homeostasis.22 Another estimate of BMR in young adults is 25 kcal/kg ideal body weight.5 Metabolic rate should then include additional calories during the rehabilitative process as estimated using a stress factor (Figure 4). This stress factor can increase metabolic energy demands from about 20% for minor injuries and surgeries to 100% for more severe bodily insults such as burns.23,24 Although musculoskeletal injuries may not increase metabolic rates to the same extent as more severe insults, any injury that activates the stress response may result in increased energy expenditure and protein catabolism. Last, calorie demands from physical activity should be included (Equation 1).
Figure 4.
Identifying energy needs during injury and rehabilitation is a key first step. Metabolic needs increase to account for basic metabolism and the increased energy demands of wound and injury healing. Abbreviations: FFM, fat-free mass; RMR, resting metabolic rate.
(1) Total Daily Energy Requirements = BMR × Stress Factor × Activity Coefficient24
Maintaining the caloric balance and meeting the increased energy demands after an injury is critical. Due to reduced physical activity after surgery (eg, anterior cruciate ligament, rotator cuff, joint replacement, fracture) or an injury, the initial response of many individuals is to reduce caloric intake to avoid weight and fat gain. However, a negative energy balance is likely to exacerbate the problem by slowing wound healing and increasing the loss of muscle.3 In turn, a negative energy balance will likely lead to greater losses in functional strength and prolong the time to return to play, which can significantly affect the athlete's career. Although concerns about weight and fat gain are warranted, the macronutrient composition of the diet, in combination with caloric intake, can help to manage body-composition changes.25 In short, diets emphasizing fewer carbohydrates (ie, approximately 40%) or a 2:1 carbohydrate:protein ratio (eg, 260 g carbs and 130 g protein), have been shown to promote positive changes in body composition.26–28 By consuming a greater proportional increase in protein, in combination with complex carbohydrates, patients can minimize weight and fat gain while still meeting the nutritional needs of recovery. An example of caloric needs and macronutrients is depicted in Figure 5. Of additional concern is alcohol, which should be avoided. Alcohol ingestion impaired muscle protein synthesis and wound healing by reducing the inflammatory response to injury.29,30
Figure 5.
Energy needs and quality of calories should be evaluated postsurgery and during recovery. A diet consisting of complex carbohydrates, high-quality protein, and high-quality fats is advised. For example, create a plate with a 30-g serving of complete proteins paired with a complex whole grain. Then add fresh fruits and vegetables in a variety of colors to help provide antioxidants to enhance recovery, control inflammation, and provide important micronutrients. Abbreviations: ACL, anterior cruciate ligament; CHO, carbohydrates; PRO, protein; RMR, resting metabolic rate.
Protein Needs
Demand for amino acids is also significantly elevated after an injury. Amino acid uptake accelerates to support wound healing, tissue rebuilding, and glycemic control.9,23 Without nutritional support, this hypermetabolic demand for amino acids is met by the catabolism of skeletal muscle.23 Depending on the severity of the injury, degree of immobilization, and length of time between injury and surgery, this could result in substantial loss of skeletal muscle. Loss of LBM is an independent risk factor for increased length of hospital stay, whereas malnutrition, especially of protein, delayed wound healing and increased risk of postsurgical infection.9,23 After a surgical procedure, the amino acid demand would be expected to rise even more to support healing and repair of incisions, tissue grafts, and other secondary trauma. Encouraging increases in protein intake in the days leading up to surgery may be advisable to minimize muscle loss and accelerate recovery.
When the stress response is activated by an injury or wound, the homeostatic mechanisms in place to prevent losses in LBM are interrupted, resulting in increased energy expenditure and protein metabolism. Injury-induced stress elevates protein needs by about 80% above baseline. For active individuals, a range of 1.4 to 2.0 g/kg/d of protein is recommended to maintain a neutral nitrogen protein balance.31 During injury recovery and immobilization, muscle protein breakdown accelerates, thereby increasing protein requirements to maintain protein balance. Nutritional goals should align with more traditional anabolic goals because when catabolic hormones rise, increasing protein intake results in a net protein balance. Thus, during rehabilitation, protein intakes of at least 1.6 g/kg/d and closer to 2.0 to 3.0 g/kg/d are recommended, with an emphasis on consuming about 3 grams of leucine per serving.3 Leucine has been termed “the anabolic trigger” and is the amino acid primarily responsible for stimulating muscle protein synthesis.32 Focusing on foods high in leucine, such as animal proteins (eg, chicken, beef, milk, fish) and whey protein, may aid in a faster recovery from injury.
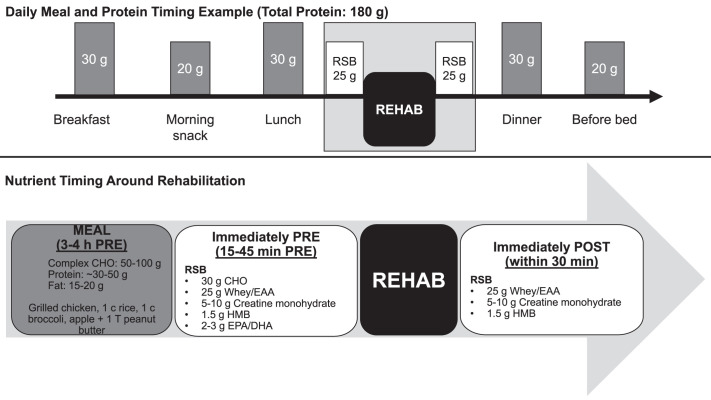
A single 30-g dose of protein was optimal for maximizing protein synthesis in healthy individuals.26 However, the amount of protein per dose needed to stimulate protein synthesis likely increases during immobilization and disuse due to anabolic resistance.33 In addition to greater protein needs, the timing of the protein ingestion is important to maximize absorption and optimize protein synthesis. To attenuate muscle protein degradation, consumption of 20 to 40 g of total dietary protein per sitting is suggested, with leucine-rich foods being eaten throughout the day. Protein consumption should occur within 1 hour of waking, every 3 to 4 hours subsequently, around a rehabilitation session, and before sleep.34,35
Complex Carbohydrates
Carbohydrates are a key source of energy during rehabilitation. Their contribution to recovery is expansive, including roles in immunology, hormonal factors, and enzymatic processes.23 They also have a protein-sparing effect in catabolic states: a high-carbohydrate diet reduced muscle protein breakdown more than a high-fat diet.36 During recovery from an injury, approximately 3 to 5 g/kg or 55% of total calories during rehabilitation or recovery should be delivered as complex carbohydrates,21 including whole grains, fruits, vegetables, and dairy. Carbohydrate needs increase as exercise activity increases. No more than 60% of total calories should be from carbohydrates; greater consumption could lead to hyperglycemia, thereby hindering healing and immune function.23 Simple carbohydrates in the form of processed and refined sugars should be limited. Complex carbohydrates are digested more slowly than simple carbohydrates, resulting in more sustained glucose availability. Complex carbohydrates are also rich in vitamins, minerals, and fiber, which are important during rehabilitation.
Essential Fatty Acids
An inflammatory response is initiated upon injury and is critical for optimal healing.3,37 However, a prolonged inflammatory response, often seen after severe injury or surgery, can be counterproductive to healing. Appropriate postinjury nutrition, specifically essential fat consumption, can help reduce the magnitude of a prolonged inflammatory response.38 Fat is a critical source of energy for healing wounds and increasing cell proliferation. Polyunsaturated and monounsaturated fatty acids are used for cell membrane production, whereas saturated fatty acids are often used for fuel. Polyunsaturated fats contain 2 essential fatty acids, α-linolenic acid (ω-3 fatty acid) and linoleic acid (ω-6 fatty acid), which must be consumed in the diet. During recovery, a diet containing high levels of monounsaturated fatty acids and ω-3 polyunsaturated fatty acids is ideal.39,40 Approximately 20% to 25% of calories should be from fat: 0.8 to 2 g/kg/d (due to the caloric density).23 Recommended daily intakes are 2 g per day of ω-3 fatty acids and 10 g per day of ω-6 fatty acids.41 Common food sources of ω-3 fatty acids are avocado, olive oil, fish, flax, nuts, and seeds. During the recovery process, ω-6 fatty acids, which are found in processed meats, fried and greasy foods, and vegetable oils, should be limited due to their proinflammatory properties.
Nutrient Timing
Considering the ingestion of nutrients throughout the day, rather than an individual's total daily consumption, has been proposed as important for exercise performance and recovery.42,43 Previous researchers demonstrated the value of nutrient timing in stimulating protein synthesis,44 reducing muscle damage,45 enhancing recovery,44 and improving body composition.46 In an injury and rehabilitation setting, the timing of nutrient consumption has the potential for significant effects on changes in LBM, strength and functionality, and return to play. More important is that an athletic trainer or physical therapist can coordinate nutrient ingestion with rehabilitation before and after seeing a patient. The hours and minutes before and after therapy are key for ensuring nutrients are available for the activity. Availability of nutrients, specifically carbohydrates and proteins, during exercise and therapy augmented outcomes (Figure 6). When nutrients were available, greater improvements in strength, functionality, and lean mass occurred.47
Figure 6.
The timing of macronutrient ingestion throughout the day is particularly important, with special attention to nutrition before and after therapy. Abbreviations: CHO, carbohydrates; EAA, essential amino acids; EPA/DHA, eicosapentaenoic acid/docosahexaenoic acid; HMB, beta symbol-hydroxy-beta symbol-methylbutyrate; rehab, rehabilitation; RSB, rehabilitation supplement blend.
DIETARY SUPPLEMENTS
Improving the quality and timing of nutrients while incorporating select dietary supplements may further enhance recovery and reduce the time until return to play. A few dietary supplements have been shown to support recovery and regeneration and mitigate muscle loss when consumed postsurgery or during immobilization. When choosing dietary supplements, purchasing products that have been subjected to third-party testing to verify product contents and rule out contaminants is advised. The most common and recommended certification programs for identifying evidence-based supplements are NSF International, NSF International Contents Certified® and Certified for Sport®, Informed Choice, ConsumerLab.com, and the Banned Substances Control Group (Figure 7). United States Pharmacopeia also provides third-party verification for vitamins. Companies that manufacture and distribute supplements can submit and cover the cost of having their products independently verified and certified by these companies; upon approval, the appropriate logo may be placed on the label to demonstrate that the product passed inspection.
Figure 7.

Third-party testing companies provide a seal or stamp for approved products. Look for these logos on applicable dietary supplements.
Creatine Monohydrate
Creatine is an organic compound that is synthesized in small amounts in the body from the amino acids arginine, methionine, and glycine. Creatine can also be obtained exogenously from foods that are high in protein, such as fish and beef.48 Approximately 95% of all creatine stores are in skeletal muscle; numerous researchers have shown increased intramuscular creatine with supplementation.49 Based on decades of work, creatine monohydrate (CrM) is one of the most effective ergogenic aids available for improving intense exercise performance and enhancing LBM when combined with exercise.49 Additional benefits of creatine included preventing traumatic brain injuries as well as improving bone health and neuromuscular function.50–54 Furthermore, despite reports in the popular press casting doubt on the usefulness of the substance, CrM may act as an agent to improve or maintain hydration and thermoregulation, thereby preventing muscle cramps and dehydration.55 When consumed during immobilization, creatine had positive effects on muscle size and strength. Specifically, when compared with a placebo (PL), 20 g (4 × 5 g) per day of CrM consumed during 2 weeks of arm immobilization better maintained lean mass (CrM: +0.9% versus PL: −3.7%), strength (CrM: −4.1% versus PL: −21.5%), and endurance (CrM: −6.5% versus PL: −35%) in young healthy men.56 However, a similar supplement strategy of 20 g CrM (4 × 5 g) versus PL during 2 weeks of leg immobilization had no effect in maintaining muscle size or strength.57 The immobilization was followed by a 12-week rehabilitation program and a weekly reduced dose of 15 g (weeks 1–3) and 5 g (weeks 4–10); when combined with rehabilitation, CrM enhanced the recovery of muscle mass, resulting in greater hypertrophy of all muscle fiber types than the PL.57 Although the exact mechanisms by which CrM may benefit the immobilized muscle have not been established, some evidence suggests it is the result of an upregulation of myogenic transcription factors (myogenin)57,58 and satellite cell activity.59,60 Additional data61 indicated that CrM may serve as a potential strategy for enhancing bone remodeling by stimulating cell growth, differentiation, and mineralization, which would be beneficial for skeletal injuries.
Creatine monohydrate is the most commonly studied form of creatine and is arguably the most bioavailable and effective.62 More importantly, no detrimental effects of supplementation with CrM have been identified.51,62 To date, the only reported side effect in some individuals was minor weight gain, which subsided after 1 to 2 days of supplementation. Thus, the potential benefits are likely to be significant, with minimal to no risk. After immobilization and injury, a loading dose is recommended: 20-g (4 × 5 g daily) loading dose for 5 days or a daily 5-g dose.63
This is followed by approximately 20 g per day in 4 divided doses for 5 days and then 3 to 5 g per day thereafter to maintain elevated stores.
Omega-3 (ω-3) Fatty Acids
The potential benefits of the long-chain ω-3 fatty acids eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) after injury have been examined. Essential nutrients, EPA and DHA can be synthesized from α-linolenic acid (ALA); however, humans are unable to synthesize ALA, making it essential to directly consume EPA and DHA through the diet. Only fish and fish oils contain EPA and DHA, which are incorporated into the wall of each cell in the body. It is important to note that plant-based sources of ω-3 (via ALA) have very low conversion rates to EPA and DHA, rendering them ineffective sources of ω-3. The ω-3 fatty acids are largely known for their anti-inflammatory properties, which have been well documented in many chronic inflammatory diseases. During the acute phase of injury or surgery, the reduction in inflammation should be carefully considered because the innate inflammatory response is necessary for healing. Thus, high doses of ω-3 within hours or a few days of injury or surgery may not be warranted. Nevertheless, if inflammation is excessive or prolonged, consumption of ω-3 may be a beneficial strategy.64 In addition to anti-inflammatory effects, ω-3 fatty acids have demonstrated benefits in immobilized patients by increasing muscle protein synthesis and mitigating muscle loss.3 Daily supplementation with 4000 mg of ω-3 stimulated an upregulation of muscle protein synthesis in response to higher protein and higher insulin levels in healthy young adults.65
The available data indicate that fish oil supplementation may reduce muscle loss during injury and immobilization. However, these effects have not been shown consistently in the literature and consensus is lacking on dosing strategies. Supplementing with 2000 to 4000 mg daily may reduce chronic inflammation and maximize protein synthesis. Side effects of fish oil supplementation include a reduction in platelets, which may increase the bleeding risk. Yet a recent systematic review66 indicated the decrease in platelet activity did not increase the bleeding risk during or after surgery and therefore, supplementation likely need not be discontinued.67 The quality of the fish oil should be evaluated to minimize contamination from high levels of mercury. Fish oil supplementation has the potential for benefit, with moderate side effects.
Essential Amino Acids
Supplemental protein is a simple and effective way to ensure adequate protein intake. Although high-quality, whole-food sources of protein should be emphasized, protein supplements provide a more concentrated and direct form of amino acids, which results in a stronger anabolic effect than dietary protein.68 In determining the quality of a protein source, the percentage of protein as essential amino acids (EAAs) and leucine content are the driving factors related to muscle protein anabolism, along with bioavailability and digestibility. Whey protein is considered the highest-quality protein source, with more than 50% of the amino acid composition coming from EAAs and 2.7 g of leucine in a common dose (25 g of protein).69,70 Whey protein isolate and concentrate are the most common forms of whey protein. The isolate is more highly filtered than the concentrate, resulting in greater protein content per dose and little to no fat and lactose, making it more tolerable for individuals with lactose intolerance. Whey protein is a fast-digesting protein source that rapidly increases EAA availability in the blood. This makes it an ideal source of protein before and after exercise and rehabilitation. In contrast, casein protein (a milk-based protein) is slower to be digested. Research71 suggested that consuming 30 to 40 g of casein before bed enhanced overnight muscle protein synthesis.
Plant-based protein products have recently grown in popularity. The highest-quality and most popular sources of plant protein are soy and pea proteins; blends containing hemp, pumpkin seed, and rice protein are also common. The degree of muscle protein synthesis from plant sources is significantly less than from animal-based products, even when matched for nitrogen and energy content.72,73 This blunted stimulus is the result of a lower EAA and leucine content. Consumption of a greater quantity of plant protein can improve the anabolic potential of plant-based products. For example, plant-based protein supplements often provide about 20 g of protein. To match the leucine content in 25 g of whey protein (2.7 g), an individual would need to consume 38 g of pea protein, 40 g of soy protein, and 54 g of hemp protein.69
Free-form EAA supplements have become available recently that provide EAAs only (ie, no other macronutrients) in a form that can be rapidly absorbed and used directly by muscle,68 resulting in a greater anabolic response than possible from dietary or whey protein supplements.68 Supplementation with 16.5 g of EAA and 30 g of carbohydrate was effective in preserving leg lean mass and reducing strength loss in healthy men after 28 days of bed rest.74 Twice daily ingestion of 20 g of EAA taken for 1 week before and for 2 weeks after total knee replacement decreased quadriceps and hamstrings atrophy at 6 weeks postsurgery more than a PL did.75 Patients in this study who supplemented with EAA also recovered functional mobility earlier than those taking the PL.75 Consumption of EAA immediately before exercise has been shown to stimulate muscle protein synthesis to a greater extent than after exercise, which was attributed to greater amino acid delivery to the muscle76 and indicated that EAA ingestion immediately before surgery or therapy may be particularly beneficial.
Vitamin D
Vitamin D is typically recognized for its importance in calcium and bone regulation, but it also plays a role in innate and acquired immune regulation as well as skeletal muscle function, thereby having potential implications for improving recovery. Vitamin D deficiency is defined as having <75 nmol/L of circulating 25-hydroxyvitamin D (25[OH]D) or <30 nmol/L of vitamin D. An optimal dose of vitamin D has not been clearly identified; the US Institute of Medicine states that greater than 50 nmol/L is sufficient,77 but controversy exists as to whether this value is too conservative. Research2–5 supported the idea that individuals who have insufficient and deficient vitamin D levels may experience impaired muscle repair, regeneration, and hypertrophy. Specifically, low vitamin D has been linked with prolonged recovery from knee surgery.78 If an individual has sufficient vitamin D, supplementation with 25(OH)D is unnecessary and may even be detrimental if used after a bone fracture due to the suppression of certain macrophages.6 After a bout of damaging eccentric exercise, healthy individuals with insufficient concentrations of 25(OH)D displayed improved recovery strength as well as increased serum 25(OH)D at 48 hours and 7 days postexercise with 4000 IU/d vitamin D3 supplementation.4 In addition, at the same dose of vitamin D (4000 IU/d), the immune system was positively affected: proinflammatory cytokines decreased and progression of infections and diseases was prevented.7,8 Vitamin D may also have implications for preventing initial inflammation and oxidative stress.9 Contrary to these positive results, the authors10 of a year-long randomized, double-blind, PL-controlled vitamin D study in older adults did not find decreases in biomarkers of inflammation, but they used a 2000-IU/d dose, which may have been insufficient to produce robust differences. Deficiency of 25(OH)D may actually result from chronic inflammation; supplementing with vitamin D3 during times of immunosuppression, infection, or marked inflammation may prevent greater vitamin D loss.11
Overall, vitamin D levels should be assessed at the time of injury to avoid negative effects from insufficiency or deficiency. The optimal dose may be 4000 IU/d unless the patient has sustained a bone fracture. In ideal circumstances, insufficiency or deficiency would be identified before injury. Should an individual choose not to take a vitamin D supplement, dietary vitamin D is found in regular and fortified foods such as cheese, yogurt, fish, orange juice, and some cereals.17 Both supplemental vitamin D3 and dietary vitamin D are generally absorbed equally efficiently.18 Vitamin D is fat soluble, which means it needs to be consumed with at least 1 serving of fat.
β-Hydroxy-β-Methylbutyrate
A natural metabolite of the EAA leucine, β-hydroxy-β-methylbutyrate (HMB) may play an important role in the prevention of protein breakdown and upregulation of protein synthesis, especially in stressful physiological situations, such as injury or surgery.79 Specifically, HMB may regulate the enzymes responsible for muscle tissue breakdown. During periods of immobilization, HMB may stimulate protein synthesis by activating the mTOR pathway, upregulating the repair rate of muscle and tendon tissue and mitigating myofibrillar degradation, thereby reducing the amount of muscle loss.80,81 Supplementation with 3 g (2 × 1.5 g) HMB daily versus PL in elderly adults during 10 days of bed rest resulted in greater maintenance of total LBM (−0.17 kg versus −2.05 kg [PL]) and leg lean mass (−0.08 kg versus 1.01 kg [PL]).82 In addition, in older adults, consuming 3 g HMB daily for 12 weeks combined with progressive resistance training resulted in greater improvements in LBM and upper body strength.83 Although HMB is commonly found in the diet, typical dietary intakes range from 0.25 to 1 g daily. Supplemental intakes of 3 g daily divided into 1.5-g doses have shown the most effective results for muscle maintenance. Supplemental HMB may preserve LBM during surgery and immobilization, as well as augment LBM when combined with therapy. No side effects have been reported with HMB; thus, the potential benefits are likely to be high and the risk minimal.
Prebiotics and Probiotics
Emerging data have suggested that the gastrointestinal microbiome could positively affect nutritional outcomes. About 70% of the immune system is located in the digestive system, demonstrating the importance of the gut microflora in immune health. Prebiotics are a group of nutrients, usually from dietary fiber, that are degraded by the gut microbiota, producing fuel for the microbiome and short-chain fatty acids, and that also help with the survival of probiotics in the stomach.84 Probiotics, often referred to as “good” bacteria, are live microorganisms that elicit health benefits in the host when administered in adequate and consistent amounts. In the surgical setting, antibiotics often cause a negative shift within the gastrointestinal flora, which may elicit an undesirable change in nutrient metabolism.85 Previous authors86 found a reduction in the rate of postoperative bacterial infections when patients were given probiotics and prebiotics postsurgery. They also demonstrated that the microbiome enhanced mucosal immunity after antibiotic use, thereby decreasing the duration and side effects of antibiotic therapy.86 The health benefits of probiotics are strain specific and dose dependent.87 The strands Lactobacillus acidophilus and Bifidobacterium longum have exhibited positive immune effects. In addition, Bacillus coagulans improved protein absorption,88 which may be important for rehabilitation. The recommended dose consumption of either strand is ≥1010 colony-forming units consumed in fermented foods such as yogurt or as dietary supplements.89 Supplemental probiotics are likely to be more effective if stored in the refrigerator and consumed daily on an empty stomach via high colony-forming units containing at least the strains L. acidophilus and B. longum.
Micronutrients
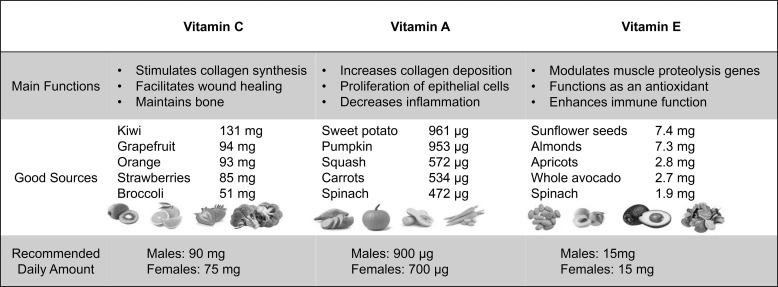
Vitamins and minerals are the most frequently consumed dietary supplements. Unlike other supplements, vitamins and certain minerals are considered essential due to their roles in normal physiological function. Deficiencies in most micronutrients can be problematic and should be avoided, especially during times of high stress. Due to the increased metabolic demands of recovery from injury and surgery, increased emphasis on micronutrient intake may be important. Specifically, vitamins A, C, and E have important roles in immunonutrition for injury recovery and wound healing.90–92 Vitamin A had positive effects on wound healing even in a nondeficient state,91 whereas vitamin C supplementation was largely only beneficial in patients with severe stress or injury.90 Vitamin E supplementation resulted in decreases in oxidative stress, thereby decreasing wound healing time.92 The major functions of these key vitamins and whole food sources are provided in Figure 8. The Institute of Medicine Committee on Nutrient Composition of Rations for Combat Operations recommended that nutrients should be provided as whole foods first, followed by fortified foods and a multivitamin dietary supplement if an individual is having difficulty consuming the recommended daily amount of micronutrients via whole-food sources.93 However, little information is available on the dose and frequency needed for these supplements to be effective. Micronutrient intakes above normal have not been shown to be more effective.3 Despite the lack of direct data, the risk:benefit ratio of multivitamin use is low to moderate, with moderate benefit and few risks.
Figure 8.
An overview of key micronutrients that have been shown to enhance healing and recovery.
TRAUMATIC BRAIN INJURY OR CONCUSSION
Traumatic brain injuries, including repetitive subconcussive injuries, cause a cascade of neurologic dysregulation.94 After a TBI, axonal damage, neuroinflammation, and dysregulation of ions (Na+, K+, Ca2+) occur; the influx of calcium (Ca2+) leads to mitochondrial dysfunction and oxidative stress that contribute to cellular damage.94 Nutritional interventions have the potential to mitigate symptoms of TBI; this area is ever expanding, with novel treatments being reported frequently. Several groups95–97 determined that supplementation of ω-3 fatty acids decreased inflammation, cellular death, and damage to the axons in rodent models. Although studies of ω-3 fatty acid supplementation after TBIs in humans are limited, the recommended dose of 40 mg/kg/d (4 g for a 100-kg individual) immediately after injury was effective in adults.94 In addition, curcumin at a dose of 100 mg/kg containing 95% curcuminoids reduced neuronal apoptosis,97 decreased oxidized proteins,98 and improved cognition after concussive brain injuries.99 However, further human research in healthy and clinical populations is warranted.
In addition to ω-3s, CrM supplementation may improve symptoms after a TBI. Brain creatine is reduced after a TBI100; creatine supplementation may balance adenosine triphosphate stores and reduce the negative effects on energy status. In a recent review101 of healthy and clinical populations, creatine supplementation of varying doses (4–20 g/d) enhanced brain creatine between 3% and 10%. A larger dose of 0.4 g/kg/d (32 g for an 80-kg person) for 6 months post-TBI improved cognitive function and decreased headaches, dizziness, and fatigue in children.102 Creatine monohydrate may improve symptoms of TBI by mitigating nerve damage, mitochondrial dysfunction, oxidative stress, and inflammation101; however, further evaluation of these physiological mechanisms in humans is essential to enable appropriate recommendations after injury. Consumption of creatine monohydrate is low risk, with a high potential for benefits. Thus, a typical 20-g daily dose (4 × 5 g) for 5 days is a safe guideline.
While nutritional interventions after TBI may be beneficial, it is also important to consider nutritional supplements that may be detrimental to recovery. Caffeine should be avoided before and after a concussive injury, given that caffeinated rodent models displayed greater deficits in cognitive and motor tasks as well as greater edema, neuronal degeneration, and more severe hemorrhage.103,104 With high intakes of caffeine in most adults, caffeine reduction in general and elimination after concussion is advised. Additional resources regarding nutrition strategies for mitigating symptoms of TBI are available.105,106
SUMMARY AND APPLICATION
Nutrition plays an essential role in injury recovery and rehabilitation. Rich data support practical nutritional recommendations for reducing surgical complications, minimizing muscle loss during immobilization, and maximizing return to play. First and foremost, the individual's caloric requirements should be identified to ensure that energy needs are being met. Higher protein intakes, with special attention to evenly distributed consumption throughout the day, will minimize loss of muscle mass and strength during times of immobilization. Dietary supplements may be helpful when navigating appropriate caloric intake and timing with low appetite. When supported by a strong nutritional plan, the results of optimal therapy and rehabilitation can be enhanced and potentially accelerated, helping patients to recover faster and safely return to play sooner. It is within the scope of an athletic trainer and physical therapist to incorporate basic nutritional recommendations, such as those we have outlined. At the very minimum, conversations with patients about the timing of their meals with respect to therapy as well as potential referral to a registered dietitian are warranted.
REFERENCES
- 1.Harlan LC, Harlan WR, Parsons PE. The economic impact of injuries: a major source of medical costs. Am J Public Health. 1990;80(4):453–459. doi: 10.2105/ajph.80.4.453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Howard-Wilsher S, Irvine L, Fan H, et al. Systematic overview of economic evaluations of health-related rehabilitation. Disabil Health J. 2016;9(1):11–25. doi: 10.1016/j.dhjo. 2015.08.009. [DOI] [PubMed] [Google Scholar]
- 3.Tipton KD. Nutritional support for exercise-induced injuries. Sports Med. 2015;45(suppl 1):S93–S104. doi: 10.1007/s40279-015-0398-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wall BT, Morton JP, van Loon LJC. Strategies to maintain skeletal muscle mass in the injured athlete: nutritional considerations and exercise mimetics. Eur J Sport Sci. 2015;15(1):53–62. doi: 10.1080/17461391.2014.936326lilin. [DOI] [PubMed] [Google Scholar]
- 5.Bannister WK, Sattilaro AJ. Vomiting and aspiration during anesthesia. Anesthesiology. 1962;23:251–264. doi: 10.1097/00000542-196203000-00012. [DOI] [PubMed] [Google Scholar]
- 6.Ljungqvist O, Soreide E. Preoperative fasting. Br J Surg. 2003;90(4):400–406. doi: 10.1002/bjs.4066. [DOI] [PubMed] [Google Scholar]
- 7.Soreide E, Stromskag KE, Steen PA. Statistical aspects in studies of preoperative fluid intake and gastric content. Acta Anaesthesiol Scand. 1995;39(6):738–743. doi: 10.1111/j.1399-6576.1995.tb04162.x. [DOI] [PubMed] [Google Scholar]
- 8.Nygren J, Thorell A, Ljungqvist O. Preoperative oral carbohydrate nutrition: an update. Curr Opin Clin Nutr Metab Care. 2001;4(4):255–259. doi: 10.1097/00075197-200107000-00002. [DOI] [PubMed] [Google Scholar]
- 9.Evans DC, Martindale RG, Kiraly LN, Jones CM. Nutrition optimization prior to surgery. Nutr Clin Pract. 2014;29(1):10–21. doi: 10.1177/0884533613517006. [DOI] [PubMed] [Google Scholar]
- 10.Gianotti L, Morelli L, Galbiati F, et al. A randomized double-blind trial on perioperative administration of probiotics in colorectal cancer patients. World J Gastroenterol. 2010;16(2):167–175. doi: 10.3748/wjg.v16.i2.167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Schricker T, Lattermann R. Strategies to attenuate the catabolic response to surgery and improve perioperative outcomes. Can J Anaesth. 2007;54(6):414–419. doi: 10.1007/BF03022026. [DOI] [PubMed] [Google Scholar]
- 12.Soop M, Nygren J, Thorell A, et al. Preoperative oral carbohydrate treatment attenuates endogenous glucose release 3 days after surgery. Clin Nutr. 2004;23(4):733–741. doi: 10.1016/j.clnu. 2003.12.007. [DOI] [PubMed] [Google Scholar]
- 13.Nygren J, Soop M, Thorell A, Efendic S, Nair KS, Ljungqvist O. Preoperative oral carbohydrate administration reduces postoperative insulin resistance. Clin Nutr. 1998;17(2):65–71. doi: 10.1152/ajpendo.2001.280.4.E576. [DOI] [PubMed] [Google Scholar]
- 14.Yuill KA, Richardson RA, Davidson HI, Garden OJ, Parks RW. The administration of an oral carbohydrate-containing fluid prior to major elective upper-gastrointestinal surgery preserves skeletal muscle mass postoperatively—a randomised clinical trial. Clin Nutr. 2005;24(1):32–37. doi: 10.1016/j.clnu.2004.06.009. [DOI] [PubMed] [Google Scholar]
- 15.Smedley F, Bowling T, James M, et al. Randomized clinical trial of the effects of preoperative and postoperative oral nutritional supplements on clinical course and cost of care. Br J Surg. 2004;91(8):983–990. doi: 10.1002/bjs.4578. [DOI] [PubMed] [Google Scholar]
- 16.Roberts MD, Lockwood C, Dalbo VJ, Volek J, Kerksick CM. Ingestion of a high-molecular-weight hydrothermally modified waxy maize starch alters metabolic responses to prolonged exercise in trained cyclists. Nutrition. 2011;27(6):659–665. doi: 10.1016/j.nut.2010.07.008. [DOI] [PubMed] [Google Scholar]
- 17.Correia CE, Bhattacharya K, Lee PJ, et al. Use of modified cornstarch therapy to extend fasting in glycogen storage disease types Ia and Ib. Am J Clin Nutr. 2008;88(5):1272–1276. doi: 10.3945/ajcn.2008.26352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Tipton KD, Borsheim E, Wolf SE, Sanford AP, Wolfe RR. Acute response of net muscle protein balance reflects 24-h balance after exercise and amino acid ingestion. Am J Physiol Endocrinol Metab. 2003;284(1):E76–E89. doi: 10.1152/ajpendo.00234.2002. [DOI] [PubMed] [Google Scholar]
- 19.Reich KA, Chen YW, Thompson PD, Hoffman EP, Clarkson PM. Forty-eight hours of unloading and 24 h of reloading lead to changes in global gene expression patterns related to ubiquitination and oxidative stress in humans. J Appl Physiol (1985) 2010;109(5):1404–1415. doi: 10.1152/japplphysiol.00444.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Burke LM, Castell LM, Casa DJ, et al. International Association of Athletics Federations consensus statement 2019: nutrition for athletics. Int J Sport Nutr Exerc Metab. 2019;29(2):73–84. doi: 10.1123/ijsnem.2019-0065. [DOI] [PubMed] [Google Scholar]
- 21.Thomas DT, Erdman KA, Burke LM. American College of Sports Medicine joint position statement. nutrition and athletic performance. Med Sci Sports Exerc. 2016;48(3):543–568. doi: 10.1249/MSS.0000000000000852. [DOI] [PubMed] [Google Scholar]
- 22.Harris J, Benedict F. A biometric study of basal metabolism in man. Proc Natl Acad Sci U S A. 1918;4(12):370–373. doi: 10.1073/pnas.4.12.370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Demling RH. Nutrition, anabolism, and the wound healing process: an overview. Eplasty. 2009;9:e9. [PMC free article] [PubMed] [Google Scholar]
- 24.Wolfe RR, Goodenough RD, Burke JF, Wolfe MH. Response of protein and urea kinetics in burn patients to different levels of protein intake. Ann Surg. 1983;197(2):163–171. doi: 10.1097/00000658-198302000-0000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Aragon AA, Schoenfeld BJ, Wildman R, et al. International Society of Sports Nutrition position stand: diets and body composition. J Int Soc Sports Nutr. 2017;14:16. doi: 10.1186/s12970-017-0174-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Layman DK. Dietary guidelines should reflect new understandings about adult protein needs. Nutr Metab (Lond) 2009;6:12. doi: 10.1186/1743-7075-6-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Layman DK, Boileau RA, Erickson DJ, et al. A reduced ratio of dietary carbohydrate to protein improves body composition and blood lipid profiles during weight loss in adult women. J Nutr. 2003;133(2):411–417. doi: 10.1093/jn/133.2.411. [DOI] [PubMed] [Google Scholar]
- 28.Weigle DS, Breen PA, Matthys CC, et al. A high-protein diet induces sustained reductions in appetite, ad libitum caloric intake, and body weight despite compensatory changes in diurnal plasma leptin and ghrelin concentrations. Am J Clin Nutr. 2005;82(1):41–48. doi: 10.1093/ajcn.82.1.41. [DOI] [PubMed] [Google Scholar]
- 29.Parr EB, Camera DM, Areta JL, et al. Alcohol ingestion impairs maximal post-exercise rates of myofibrillar protein synthesis following a single bout of concurrent training. PLoS One. 2014;9(2):e88384. doi: 10.1371/journal.pone.0088384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Jung MK, Callaci JJ, Lauing KL, et al. Alcohol exposure and mechanisms of tissue injury and repair. Alcohol Clin Exp Res. 2011;35(3):392–399. doi: 10.1111/j.1530-0277.2010.01356.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Jager R, Kerksick CM, Campbell BI, et al. International Society of Sports Nutrition position stand: protein and exercise. J Int Soc Sports Nutr. 2017;14:20. doi: 10.1186/s12970-017-0177-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Nicastro H, Artioli GG, Costa Ados S, et al. An overview of the therapeutic effects of leucine supplementation on skeletal muscle under atrophic conditions. Amino Acids. 2011;40(2):287–300. doi: 10.1007/s00726-010-0636-x. [DOI] [PubMed] [Google Scholar]
- 33.Wall BT, Snijders T, Senden JM, et al. Disuse impairs the muscle protein synthetic response to protein ingestion in healthy men. J Clin Endocrinol Metab. 2013;98(12):4872–4881. doi: 10.1210/jc.2013-2098. [DOI] [PubMed] [Google Scholar]
- 34.Areta JL, Burke LM, Ross ML, et al. Timing and distribution of protein ingestion during prolonged recovery from resistance exercise alters myofibrillar protein synthesis. J Physiol. 2013;591(9):2319–2331. doi: 10.1113/jphysiol.2012.244897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Yang Y, Breen L, Burd NA, et al. Resistance exercise enhances myofibrillar protein synthesis with graded intakes of whey protein in older men. Br J Nutr. 2012;108(10):1780–1788. doi: 10.1017/S0007114511007422. [DOI] [PubMed] [Google Scholar]
- 36.Hart DW, Wolf SE, Zhang XJ, et al. Efficacy of a high-carbohydrate diet in catabolic illness. Crit Care Med. 2001;29(7):1318–1324. doi: 10.1097/00003246-200107000-00004. [DOI] [PubMed] [Google Scholar]
- 37.Stechmiller JK. Understanding the role of nutrition and wound healing. Nutr Clin Pract. 2010;25(1):61–68. doi: 10.1177/0884533609358997. [DOI] [PubMed] [Google Scholar]
- 38.Lin E, Kotani JG, Lowry SF. Nutritional modulation of immunity and the inflammatory response. Nutrition. 1998;14(6):545–550. doi: 10.1016/s0899-9007(98)00046-x. [DOI] [PubMed] [Google Scholar]
- 39.Calder PC. n-3 fatty acids, inflammation and immunity: new mechanisms to explain old actions. Proc Nutr Soc. 2013;72(3):326–336. doi: 10.1017/S0029665113001031. [DOI] [PubMed] [Google Scholar]
- 40.Galland L. Diet and inflammation. Nutr Clin Pract. 2010;25(6):634–640. doi: 10.1177/0884533610385703. [DOI] [PubMed] [Google Scholar]
- 41.EFSA Panel on Dietetic Products, Nutrition and Allergies. Scientific opinion on the tolerable upper intake level of eicosapentaenoic acid (EPA), docosahexaenoic acid (DHA) and docosapentaenoic acid (DPA) Eur Food Safe Author J. 2012;10(7):2815. doi: 10.2903/j.efsa.2012.2815. [DOI] [Google Scholar]
- 42.Aragon AA, Schoenfeld BJ. Nutrient timing revisited: is there a post-exercise anabolic window? J Int Soc Sports Nutr. 2013;10(1):5. doi: 10.1186/1550-2783-10-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Cribb PJ, Hayes A. Effects of supplement timing and resistance exercise on skeletal muscle hypertrophy. Med Sci Sports Exerc. 2006 Nov;38(11):1918–1925. doi: 10.1249/01.mss.0000233790.08788.3e. [DOI] [PubMed] [Google Scholar]
- 44.Schoenfeld BJ, Aragon AA, Krieger JW. The effect of protein timing on muscle strength and hypertrophy: a meta-analysis. J Int Soc Sports Nutr. 2013;10(1):53. doi: 10.1186/1550-2783-10-53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Kerksick C, Harvey T, Stout J, et al. International Society of Sports Nutrition position stand: nutrient timing. J Int Soc Sports Nutr. 2008;5:17. doi: 10.1186/1550-2783-5-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Josse AR, Tang JE, Tarnopolsky MA, Phillips SM. Body composition and strength changes in women with milk and resistance exercise. Med Sci Sports Exerc. 2010;42(6):1122–1130. doi: 10.1249/MSS.0b013e3181c854f6. [DOI] [PubMed] [Google Scholar]
- 47.Kerksick CM, Arent S, Schoenfeld BJ, et al. International Society of Sports Nutrition position stand: nutrient timing. J Int Soc Sports Nutr. 2017;14:33. doi: 10.1186/s12970-017-0189-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Branch JD. Effect of creatine supplementation on body composition and performance: a meta-analysis. Int J Sport Nutr Exerc Metab. 2003;13(2):198–226. doi: 10.1123/ijsnem.13.2.198. [DOI] [PubMed] [Google Scholar]
- 49.Buford TW, Kreider RB, Stout JR, et al. International Society of Sports Nutrition position stand: creatine supplementation and exercise. J Int Soc Sports Nutr. 2007;4:6. doi: 10.1186/1550-2783-4-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Candow DG, Chilibeck PD. Potential of creatine supplementation for improving aging bone health. J Nutr Health Aging. 2010;14(2):149–153. doi: 10.1007/s12603-009-0224-5. [DOI] [PubMed] [Google Scholar]
- 51.Havenetidis K. The use of creatine supplements in the military. J R Army Med Corps. 2016;162(4):242–248. doi: 10.1136/jramc-2014-000400. [DOI] [PubMed] [Google Scholar]
- 52.Rae C, Digney AL, McEwan SR, Bates TC. Oral creatine monohydrate supplementation improves brain performance: a double-blind, placebo-controlled, cross-over trial. Proc Biol Sci. 2003;270(1529):2147–2150. doi: 10.1098/rspb.2003.2492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Smith-Ryan AE, Ryan ED, Fukuda DH, Costa PB, Cramer JT, Stout JR. The effect of creatine loading on neuromuscular fatigue in women. Med Sci Sports Exerc. 2014;46(5):990–997. doi: 10.1249/MSS.0000000000000194. [DOI] [PubMed] [Google Scholar]
- 54.Sullivan PG, Geiger JD, Mattson MP, Scheff SW. Dietary supplement creatine protects against traumatic brain injury. Ann Neurol. 2000 Nov;48(5):723–729. [PubMed] [Google Scholar]
- 55.Sobolewski EJ, Thompson BJ, Smith AE, Ryan ED. The physiological effects of creatine supplementation on hydration: a review. Am J Lifestyle Med. 2011;5(4):320–327. [Google Scholar]
- 56.Johnston APW, Burke DG, MacNeil LG, Candow DG. Effect of creatine supplementation during cast-induced immobilization on the preservation of muscle mass, strength, and endurance. J Strength Cond Res. 2009;23(1):116–120. doi: 10.1519/jsc.0b013e31818efbcc. [DOI] [PubMed] [Google Scholar]
- 57.Hespel P, Op't Eijnde B, Van Leemputte M, et al. Oral creatine supplementation facilitates the rehabilitation of disuse atrophy and alters the expression of muscle myogenic factors in humans. J Physiol. 2001;536(pt 2):625–633. doi: 10.1111/j.1469-7793.2001.0625c.xd. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Parise G, Mihic S, MacLennan D, Yarasheski KE, Tarnopolsky MA. Effects of acute creatine monohydrate supplementation on leucine kinetics and mixed-muscle protein synthesis. J Appl Physiol (1985) 2001;91(3):1041–1047. doi: 10.1152/jappl.2001.91.3.1041. [DOI] [PubMed] [Google Scholar]
- 59.Olsen S, Aagaard P, Kadi F, et al. Creatine supplementation augments the increase in satellite cell and myonuclei number in human skeletal muscle induced by strength training. J Physiol. 2006;573(pt 2):525–534. doi: 10.1113/jphysiol.2006.107359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Dangott B, Schultz E, Mozdziak PE. Dietary creatine monohydrate supplementation increases satellite cell mitotic activity during compensatory hypertrophy. Int J Sports Med. 2000;21(1):13–16. doi: 10.1055/s-2000-8848. [DOI] [PubMed] [Google Scholar]
- 61.Gerber I, Gwynn I, Alini M, Wallimann T. Stimulatory effects of creatine on metabolic activity, differentiation and mineralization of primary osteoblast-like cells in monolayer and micromass cell cultures. Eur Cell Mater. 2005;10:8–22. doi: 10.22203/ecm.v010a02. [DOI] [PubMed] [Google Scholar]
- 62.Jager R, Purpura M, Shao A, Inoue T, Kreider RB. Analysis of the efficacy, safety, and regulatory status of novel forms of creatine. Amino Acids. 2011;40(5):1369–1383. doi: 10.1007/s00726-011-0874-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Kreider RB, Kalman DS, Antonio J, et al. International Society of Sports Nutrition position stand: safety and efficacy of creatine supplementation in exercise, sport, and medicine. J Int Soc Sports Nutr. 2017;14:18. doi: 10.1186/s12970-017-0173-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Calder PC, Albers R, Antoine JM, et al. Inflammatory disease processes and interactions with nutrition. Br J Nutr. 2009;101(suppl 1):S1–S45. doi: 10.1017/S0007114509377867. [DOI] [PubMed] [Google Scholar]
- 65.Smith GI, Atherton P, Reeds DN, et al. Omega-3 polyunsaturated fatty acids augment the muscle protein anabolic response to hyperinsulinaemia-hyperaminoacidaemia in healthy young and middle-aged men and women. Clin Sci (Lond) 2011;121(6):267–278. doi: 10.1042/CS20100597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Begtrup KM, Krag AE, Hvas AM. No impact of fish oil supplements on bleeding risk: a systematic review. Dan Med J. 2017;64(5):A5366. [PubMed] [Google Scholar]
- 67.Akintoye E, Sethi P, Harris WS, et al. Fish oil and perioperative bleeding. Circ Cardiovasc Qual Outcomes. 2018;11(11):e004584. doi: 10.1161/CIRCOUTCOMES.118.004584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Paddon-Jones D, Sheffield-Moore M, Aarsland A, Wolfe RR, Ferrando AA. Exogenous amino acids stimulate human muscle anabolism without interfering with the response to mixed meal ingestion. Am J Physiol Endocrinol Metab. 2005;288(4):E761–E767. doi: 10.1152/ajpendo.00291.2004. [DOI] [PubMed] [Google Scholar]
- 69.Gorissen SHM, Crombag JJR, Senden JMG, et al. Protein content and amino acid composition of commercially available plant-based protein isolates. Amino Acids. 2018;50(12):1685–1695. doi: 10.1007/s00726-018-2640-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.van Vliet S, Burd NA, van Loon LJ. The skeletal muscle anabolic response to plant- versus animal-based protein consumption. J Nutr. 2015;145(9):1981–1991. doi: 10.3945/jn.114.204305. [DOI] [PubMed] [Google Scholar]
- 71.Res PT, Groen B, Pennings B, et al. Protein ingestion before sleep improves postexercise overnight recovery. Med Sci Sports Exerc. 2012;44(8):1560–1569. doi: 10.1249/MSS.0b013e31824cc363. [DOI] [PubMed] [Google Scholar]
- 72.Phillips SM. Nutrient-rich meat proteins in offsetting age-related muscle loss. Meat Sci. 2012;92(3):174–178. doi: 10.1016/j.meatsci.2012.04.027. [DOI] [PubMed] [Google Scholar]
- 73.Wilkinson SB, Tarnopolsky MA, Macdonald MJ, Macdonald JR, Armstrong D, Phillips SM. Consumption of fluid skim milk promotes greater muscle protein accretion after resistance exercise than does consumption of an isonitrogenous and isoenergetic soy-protein beverage. Am J Clin Nutr. 2007;85(4):1031–1040. doi: 10.1093/ajcn/85.4.1031. [DOI] [PubMed] [Google Scholar]
- 74.Paddon-Jones D, Sheffield-Moore M, Urban RJ, et al. Essential amino acid and carbohydrate supplementation ameliorates muscle protein loss in humans during 28 days bedrest. J Clin Endocrinol Metab. 2004;89(9):4351–4358. doi: 10.1210/jc.2003-032159. [DOI] [PubMed] [Google Scholar]
- 75.Dreyer HC, Strycker LA, Senesac HA, et al. Essential amino acid supplementation in patients following total knee arthroplasty. J Clin Invest. 2013;123(11):4654–4566. doi: 10.1172/JCI70160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Tipton KD, Rasmussen BB, Miller SL, et al. Timing of amino acid-carbohydrate ingestion alters anabolic response of muscle to resistance exercise. Am J Physiol Endocrinol Metab. 2001;281(2):E197–E206. doi: 10.1152/ajpendo.2001.281.2.E197. [DOI] [PubMed] [Google Scholar]
- 77.Institute of Medicine, Food and Nutrition Board. Dietary Reference Intakes for Calcium and Vitamin D. Washington, DC: National Academy Press;; 2010. [Google Scholar]
- 78.Barker T, Martins TB, Hill HR, et al. Low vitamin D impairs strength recovery after anterior cruciate ligament surgery. J Evid Based Complementary Altern Med. 2011;16(3):201–209. doi: 10.1177/2156587211413768. [DOI] [Google Scholar]
- 79.Nissen SL, Sharp RL. Effect of dietary supplements on lean mass and strength gains with resistance exercise: a meta-analysis. J Appl Physiol (1985) 2003;94(2):651–659. doi: 10.1152/japplphysiol.00755.2002. [DOI] [PubMed] [Google Scholar]
- 80.Molfino A, Gioia G, Rossi Fanelli F, Muscaritoli M. Beta-hydroxy-beta-methylbutyrate supplementation in health and disease: a systematic review of randomized trials. Amino Acids. 2013;45(6):1273–1292. doi: 10.1007/s00726-013-1592-z. [DOI] [PubMed] [Google Scholar]
- 81.Zanchi NE, Gerlinger-Romero F, Guimaraes-Ferreira L, et al. HMB supplementation: clinical and athletic performance-related effects and mechanisms of action. Amino Acids. 2011;40(4):1015–1025. doi: 10.1007/s00726-010-0678-0. [DOI] [PubMed] [Google Scholar]
- 82.Deutz NE, Pereira SL, Hays NP, et al. Effect of β-hydroxy-β-methylbutyrate (HMB) on lean body mass during 10 days of bed rest in older adults. Clin Nutr. 2013;32(5):704–712. doi: 10.1016/j.clnu.2013.02.011. [DOI] [PubMed] [Google Scholar]
- 83.Stout JR, Smith-Ryan AE, Fukuda DH, et al. Effect of calcium β-hydroxy-β-methylbutyrate (CaHMB) with and without resistance training in men and women 65+ yrs: a randomized, double-blind pilot trial. Exp Gerontol. 2013;48(11):1303–1310. doi: 10.1016/j.exger.2013.08.007. [DOI] [PubMed] [Google Scholar]
- 84.Davani-Davari D, Negahdaripour M, Karimzadeh I, et al. Prebiotics: definition, types, sources, mechanisms, and clinical applications. Foods. 2019;8(3):92. doi: 10.3390/foods8030092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.D'Souza AL, Rajkumar C, Cooke J, Bulpitt CJ. Probiotics in prevention of antibiotic associated diarrhoea: meta-analysis. BMJ. 2002;324(7350):1361. doi: 10.1136/bmj.324.7350.1361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Rayes N, Seehofer D, Theruvath T, et al. Supply of pre- and probiotics reduces bacterial infection rates after liver transplantation—a randomized, double-blind trial. Am J Transplant. 2005;5(1):125–130. doi: 10.1111/j.1600-6143.2004.00649.x. [DOI] [PubMed] [Google Scholar]
- 87.Gleeson M, Bishop NC, Oliveira M, McCauley T, Tauler P, Lawrence C. Effects of a Lactobacillus salivarius probiotic intervention on infection, cold symptom duration and severity, and mucosal immunity in endurance athletes. Int J Sport Nutr Exerc Metab. 2012;22(4):235–242. doi: 10.1123/ijsnem.22.4.235. [DOI] [PubMed] [Google Scholar]
- 88.Jager R, Purpura M, Farmer S, Cash HA, Keller D. Probiotic Bacillus coagulans GBI-30, 6086 improves protein absorption and utilization. Probiotics Antimicrob Proteins. 2018;10(4):611–615. doi: 10.1007/s12602-017-9354-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Maughan RJ, Burke LM, Dvorak J, et al. IOC consensus statement: dietary supplements and the high-performance athlete. Br J Sports Med. 2018;52(7):439–455. doi: 10.1136/bjsports-2018-099027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Carr AC, Maggini S. Vitamin C and immune function. Nutrients. 2017;9(11):1211. doi: 10.3390/nu9111211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Raverdeau M, Mills KH. Modulation of T cell and innate immune responses by retinoic acid. J Immunol. 2014;192(7):2953–2958. doi: 10.4049/jimmunol.1303245. [DOI] [PubMed] [Google Scholar]
- 92.Jiang Q. Natural forms of vitamin E: metabolism, antioxidant, and anti-inflammatory activities and their role in disease prevention and therapy. Free Radic Biol Med. 2014;72:76–90. doi: 10.1016/j.freeradbiomed.2014.03.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Institute of Medicine. Nutrient Composition of Rations for ShortTerm HighIntensity Combat Operations. Washington, DC: National Academies Press;; 2005. [Google Scholar]
- 94.Barrett EC, McBurney MI, Ciappio ED. ω-3 fatty acid supplementation as a potential therapeutic aid for the recovery from mild traumatic brain injury/concussion. Adv Nutr. 2014;5(3):268–277. doi: 10.3945/an.113.005280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Wu A, Ying Z, Gomez-Pinilla F. Dietary strategy to repair plasma membrane after brain trauma: implications for plasticity and cognition. Neurorehabil Neural Repair. 2014;28(1):75–84. doi: 10.1177/1545968313498650. [DOI] [PubMed] [Google Scholar]
- 96.Wang T, Van KC, Gavitt BJ, et al. Effect of fish oil supplementation in a rat model of multiple mild traumatic brain injuries. Restor Neurol Neurosci. 2013;31(5):647–659. doi: 10.3233/RNN-130316. [DOI] [PubMed] [Google Scholar]
- 97.Mills JD, Hadley K, Bailes JE. Dietary supplementation with the omega-3 fatty acid docosahexaenoic acid in traumatic brain injury. Neurosurgery. 2011;68(2):474–481. doi: 10.1227/NEU.0b013e3181ff692b. [DOI] [PubMed] [Google Scholar]
- 98.Zhu HT, Bian C, Yuan JC, et al. Curcumin attenuates acute inflammatory injury by inhibiting the TLR4/MyD88/NF-κB signaling pathway in experimental traumatic brain injury. J Neuroinflammation. 2014;11:59. doi: 10.1186/1742-2094-11-59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Wu A, Ying Z, Gomez-Pinilla F. Dietary curcumin counteracts the outcome of traumatic brain injury on oxidative stress, synaptic plasticity, and cognition. Exp Neurol. 2006;197(2):309–317. doi: 10.1016/j.expneurol.2005.09.004. [DOI] [PubMed] [Google Scholar]
- 100.Vagnozzi R, Signoretti S, Floris R, et al. Decrease in N-acetylaspartate following concussion may be coupled to decrease in creatine. J Head Trauma Rehabil. 2013;28(4):284–292. doi: 10.1097/HTR.0b013e3182795045. [DOI] [PubMed] [Google Scholar]
- 101.Dolan E, Gualano B, Rawson ES. Beyond muscle: the effects of creatine supplementation on brain creatine, cognitive processing, and traumatic brain injury. Eur J Sport Sci. 2019;19(1):1–14. doi: 10.1080/17461391.2018.1500644. [DOI] [PubMed] [Google Scholar]
- 102.Ainsley Dean PJ, Arikan G, Opitz B, Sterr A. Potential for use of creatine supplementation following mild traumatic brain injury. Concussion. 2017 doi: 10.2217/cnc-2016-0016. 2(2):CNC34. [DOI] [PMC free article] [PubMed]
- 103.Christensen J, Yamakawa GR, Salberg S, Wang M, Kolb B, Mychasiuk R. Caffeine consumption during development alters spine density and recovery from repetitive mild traumatic brain injury in young adult rats. Synapse. doi: 10.1002/syn.22142. 2020:74(4):e22142. [DOI] [PubMed]
- 104.Al Moutaery K, Al Deeb S, Ahmad Khan H, Tariq M. Caffeine impairs short-term neurological outcome after concussive head injury in rats. Neurosurgery. 2003;53(3):704–712. doi: 10.1227/01.neu.0000079487.66013.6f. [DOI] [PubMed] [Google Scholar]
- 105.Lucke-Wold BP, Logsdon AF, Nguyen L, et al. Supplements, nutrition, and alternative therapies for the treatment of traumatic brain injury. Nutr Neurosci. 2018;21(2):79–91. doi: 10.1080/1028415X.2016.1236174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Scrimgeour AG, Condlin ML. Nutritional treatment for traumatic brain injury. J Neurotrauma. 2014;31(11):989–999. doi: 10.1089/neu.2013.3234. [DOI] [PubMed] [Google Scholar]