Abstract
In this study, the anti-tumor activity of ilimaquinone (IQ), a sesquiterpene quinone isolated from marine sponge Halichondria sp., in oral squamous cell carcinoma (OSCC) cells, was investigated. IQ suppressed the viability of the OSCC cell lines SCC4 and SCC2095 with IC50 values of 7.5 and 8.5 μM, respectively. Flow cytometric analysis demonstrated that IQ induced caspase-dependent apoptosis in SCC4 cells and modulated the expression of several cell growth-related gene products, including Akt, p38, Mcl-1, and p53. Notably, p53 knockdown caused higher resistance to IQ’s anti-tumor activity. In addition, IQ increased reactive oxygen species generation, which was partially reversed by the addition of antioxidants. Furthermore, it triggered autophagy, as evidenced by acidic organelle formation and LC3B-II and Atg5 expression in SCC4 cells. Pretreatment with the autophagy inhibitor 3-methyladenine or chloroquine partially decreased IQ-induced apoptosis, suggesting that IQ induced protective autophagy. In summary, IQ has potential to be used in OSCC therapy.
Keywords: ilimaquinone, OSCC, autophagy, apoptosis, p53
1. Introduction
The incidence of oral squamous cell carcinoma (OSCC), which has a high mortality rate, has witnessed increases in developing counties, such as India [1,2]. In Asia, betel quid chewing, smoking, and alcohol consumption are the main OSCC risk factors [3]. Several mutated genes, including cyclin D1, p53, and Rb, and the dysregulation of molecular pathways have been associated with its progression [4]. In addition to surgery, current therapies include chemotherapy, radiotherapy, and immunotherapy [5]. However, the 5-year survival rate has remained at 50%, for the past decades [3], indicating the urgent need for new therapies.
For over 34 years, natural products have provided ~40% of developed FDA-approved therapeutic agents [6]. The source of some of these therapeutic agents is the oceans, which contain a rich biodiversity of marine organisms [7]. For example, trabectedin, an alkaloid isolated from a tunicate, has been used to treat advanced soft tissue sarcoma for over 10 years [8]. Additionally, since 2010, eribulin mesylate, originally developed from halichondrin B, a macrocyclic polyether obtained from marine sponges, was approved as metastatic breast cancer therapy [8,9]. Therefore, the ocean is a potential source of anti-tumor agents.
Among marine organisms, marine sponges produce several secondary metabolites, including quinones, steroids, alkaloids, terpenoids, and fatty acids, some of which possess drug-like characteristics [10]. Ilimaquinone (IQ), a sesquiterpene quinone isolated from marine sponges, exhibits a variety of biological activities, including anti-inflammatory [11], antitubercular [12], anti-HIV [13], and anti-tumor [14]. Previous studies have revealed that IQ interferes with protein transport by damaging the Golgi apparatus in cervical cancer cells [15]. Additionally, multiple studies have demonstrated that IQ induces apoptosis in several cancer cell lines, including prostate cancer, colon cancer, and multiple myeloma [16,17,18]. For example, it induces apoptosis through caspase-3 activation, and upregulates the expression of p53 and p21 in colon cancer cells [18]. By activating ROS-ERK/p38 MAPK-CHOP signaling, it also potentiates tumor necrosis factor-related apoptosis-inducing ligand (TRAIL)-induced apoptosis in colon cancer cells [14]. Furthermore, some studies have reported that IQ inhibits cancer cell growth by regulating β-catenin [17], GADD-153 [16], and p53 [18]. However, its anti-tumor activity in OSCC is still unclear. This study therefore investigated its anti-tumor effects in OSCC cells, as well as the underlying mechanism.
2. Materials and Methods
2.1. Reagents, Chemicals, Antibodies
The sponge Halichondria sp. was collected by scuba diving at a depth of 10 m from coral reefs off the Coast of Pingtung County, in 2016. Voucher specimen has been deposited in the National Museum of Marine Biology and Aquarium, Taiwan (specimen No. SP2016-01). The frozen sponge Halichondria sp. material (450 g, wet weight) was freeze-dried, and the resulting material (80 g) was then minced and extracted exhaustively with ethyl acetate (EtOAc) (1 L) six consecutive times with the duration of one week each time. The EtOAc crude extract was evaporated under reduced pressure to afford a residue (3.66 g). Then, the residue was separated over a silica gel by column chromatography to obtain IQ (811 mg). The identity and purity of IQ were verified by a comparison of its 1D and 2D proton nuclear magnetic resonance (NMR) spectral data (Figures S1–S5) with available literature [19]. All agents were dissolved in dimethyl sulfoxide (DMSO), diluted in culture medium, and added to cells at a final DMSO concentration of 0.1%. Other chemicals and reagents were obtained from Sigma-Aldrich unless otherwise noted.
2.2. Cell Culture
SCC4 human oral squamous cell carcinoma cells were purchased from Japanese Collection of Research Bioresources (Tokyo, Japan), and SCC2095 human oral squamous cell carcinoma cells were kindly provided by Professor Susan R. Mallery (The Ohio State University). All cell lines were cultured in dulbecco’s modified eagle medium (DMEM)/F12 medium (Gibco, Grand Island, NY, USA) which containing 5 mg/mL of penicillin, 10% fetal bovine serum (FBS) (Gibco), and 5 mg/mL streptomycin at 37 °C in a humidified incubator containing 5% CO2.
2.3. Cell Viability Analysis
Cell viability of IQ was assessed by using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assays [20]. Briefly, cells (5 × 103) were seeded and incubated in 96-well, flat-bottomed plates in 10% FBS-supplemented medium for 24 h and were exposed to compound at indicated concentrations for different time intervals. After removing the medium, it was replaced by 200 μL of 0.5 mg/mL MTT in 5% FBS-medium, and cells were incubated at 37 °C for 4 h. The medium was removed and the reduced MTT dye was solubilized in 200 μL/well DMSO. Absorbance was determined with a SPECTROstar Nano spectrophotometer (BMG LABTECH, Ortenberg, Germany) at 570 nm.
2.4. Flow Cytometric Analysis
Cells (2 × 105/3 mL/per well) were plated in a six-well plate and treated with DMSO or the indicated concentration of IQ for 48 h [20]. For apoptosis evaluation, after being washed twice with phosphate-buffered saline (PBS), followed by spinning at 1200 rpm for 5 min, and cells in 500 μL PBS were stained with 3 μL annexin V-fluorescein isothiocyanate (FITC) and 5 μL propidium iodide (PI) at room temperature for 15 min according to the vender’s protocols (BD Pharmingen, San Diego, USA) and analyzed by using an Attune NxT flow cytometer (ThermoFisher Scientific, Waltham, MA, USA). For reactive oxygen species (ROS) production determination, cells were washed with PBS twice and stained with the fluorescence probe 5-(and-6)-carboxy-2′,7’-dichlorodihydrofluoresceindiacetate (carboxy-DCFDA, 5 μL, Invitrogen) at 37 °C for 30 min and analyzed by a flow cytometer.
2.5. Immunoblotting
Protein was collected from the cells after various treatments. For Western blots, a previously described procedure was applied [20]. The immunoblotting was performed with primary antibodies recognizing p473Ser-Akt (#9271), Akt (#9272), PARP (#9542), cleaved caspase-9 (#7237), p180Thr/182Tyr-p38 MAPK (#9215), p38 MAPK (#9212), HIF-1α (#3716), Mcl-1 (#39224), survivin (#2802), LC3B(#2775), p15Ser-p53 (#9284), and p53 (#2527) all from Cell Signaling Technologies (Beverly, MA, USA); procaspase-8 (MAB4708) from Millipore; Bax (ab7977) from Abcam (Cambridge, UK); Bcl-2 (Sc-509) from Santa Cruz Biotechnology (CA, USA); Atg5 (GTX62601) from GeneTex (Irvine, CA, USA); β-actin (A5316) from Sigma-Aldrich (St. Louis, MO, USA). Immunoblotted bands were visualized by an enhanced chemiluminescence reagent (GE Healthcare Bioscience, NJ, USA) and system (FUSION SoLo S, Deutschland, Germany) using secondary antibodies (Santa Cruz Biotechnology, Santa Cruz, CA, USA).
2.6. Detection of Autophagosome Formation with Acridine Orange
To detect the presence of acidic vesicular organelles (AVOs), IQ-treated cells were stained with acridine orange (1 μg/mL) for 15 min and washed with PBS. Then, the cells were examined under a fluorescence microscope [21].
2.7. Transient Transfection
SCC4 cells (2 × 105/3 mL) were seeded in each well of a six-well plate. After a 24 h incubation, cells were transfected with p53 shRNA (TRCN0000003755, the National RNAi Core Facility, Taipei, Taiwan) using Fugene HD reagent (Roche) for another 24 h. Then, the cells were treated with IQ for 48 h and collected the protein for Western blotting.
2.8. Statistical Analysis
The data are presented as the mean of at least three replicates. Statistical significance was performed by Student’s t test, and p-values (* p < 0.05, ** p < 0.01) were considered as significant.
3. Results
3.1. IQ Decreases the Viability of OSCC
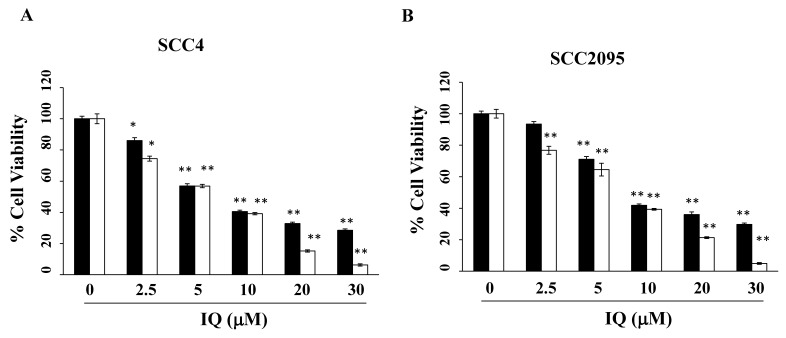
Using MTT assays, the anti-viability effect of IQ was assessed in two OSCC cell lines: SCC4 and SCC2095. As shown in Figure 1A,B, IQ decreased cell viability in a concentration- and time-dependent manner. IQ concentrations of 7.5 and 8.5 μM decreased cell viability by 50% after 48 h in SCC4 and SCC2095 cells, respectively. Given that, after treatment, the IC50 value of IQ in SCC4 cells was lower than that in SCC2095, the former was used in subsequent experiments.
Figure 1.
Effect of ilimaquinone (IQ) on the viability of oral cancer cells (SCC4 and SCC2095). (A) SCC4 and (B) SCC2095. The cells were seeded onto 96-well plates, incubated for 24 (■) and 48 h (□), treated with IQ for 48 h and then cell viability was detected using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assays. Points, means; bars, S.D. (n = 3–6). * p < 0.05 and ** p < 0.01 vs. the control group.
3.2. IQ Induces Caspase-Dependent Apoptosis in SCC4 Cells
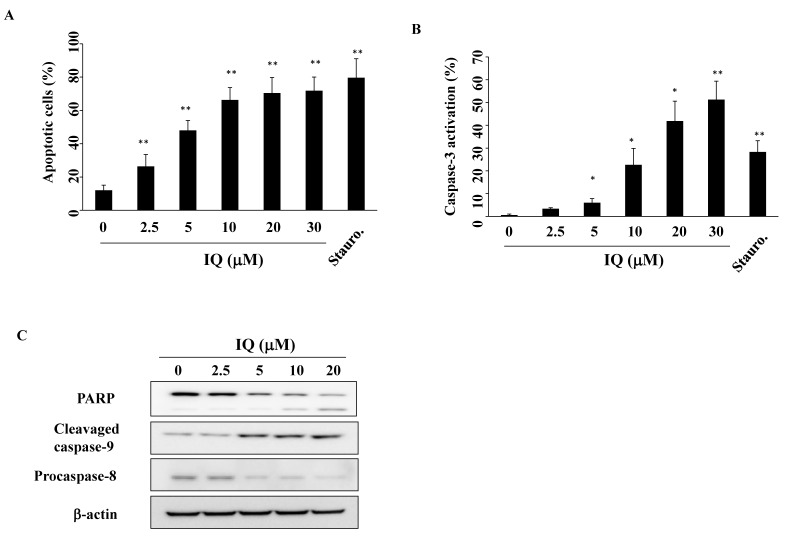
To determine whether apoptosis resulted from IQ-mediated cell death, SCC4 cells were treated with IQ for 48 h, and assessed using propidium iodide (PI)/Annexin V analysis. As shown in Figure 2A, IQ increased the percentage of apoptotic cells in a concentration-dependent manner (staurosporine as a positive control). To confirm the involvement of caspase activation, SCC4 cells were treated with IQ for 48 h, and analyzed for activated caspase-3 using flow cytometry (Figure 2B). This treatment increased activated caspase-3 levels, from 0.6% in the control group to 6.0%, 22.7%, 41.8%, and 51.3% in SCC4 cells treated with 5, 10, 20, and 30 μM of IQ, respectively (staurosporine as a positive control). Compatible with flow cytometric staining findings, IQ treatment led to the activation of caspase-8, caspase-9, and poly (ADP-ribose)polymerase (PARP) cleavage in a concentration-dependent manner (Figure 2C). These results confirm that IQ induces caspase-dependent apoptosis in SCC4 cells.
Figure 2.
Effect of ilimaquinone (IQ) treatment on apoptosis. (A) The percentage of apoptotic cells (Q2 + Q4) after dimethyl sulfoxide (DMSO) vehicle, IQ, or 50 nM staurosporine (Stauro.) treatment for 48 h. SCC4 cells were treated with either DMSO, IQ, or Stauro. in 5% fetal bovine serum (FBS)-supplemented DMEM/F12 medium for 48 h and stained with propidium iodide (PI)/annexin V. Columns, mean; bars, S.D. (n = 4). ** p < 0.01 vs. the control group. (B) Concentration-dependent effect of IQ on caspase-3 activation. Staurosporine (Stauro.; 25 nM) was used as the positive control. Columns, mean; bars, S.D. (n = 3). * p < 0.05 and ** p < 0.01 vs. the control group. (C) Expression of PARP, cleavage caspase-9, and procaspase-8 in SCC4 cells after 48 h treatment in 5% FBS-supplemented DMEM/F12 medium.
3.3. IQ Modulates Akt and p53 Expression in SCC4 Cells
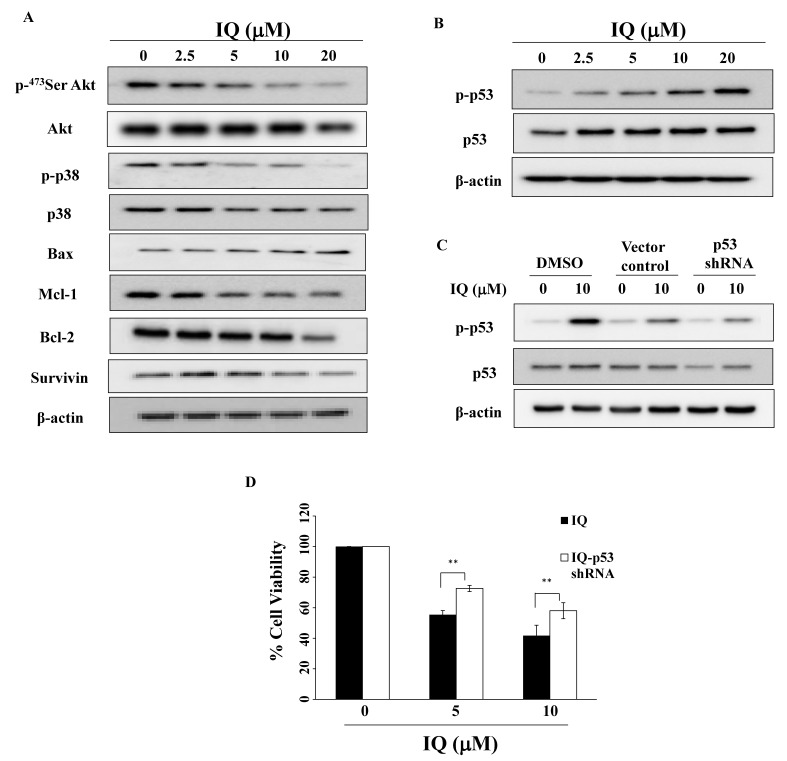
Previous studies demonstrated that Akt and p38 activation mediates OSCC migration and that blocking their signaling increased the sensitivity of OSCC cells to chemotherapeutic agents [22,23,24,25]. This study reveals that, in OSCC cells (Figure 3A), IQ downregulated the expression of p-Akt, p-p38, and the metastasis prognostic marker HIF-1α [26]. Consistent with caspase-dependent apoptosis, IQ increased proapoptotic protein Bax expression in a concentration-dependent manner, accompanied by a parallel decrease of the anti-apoptotic proteins Mcl-1 and Bcl-2, and the apoptotic protein inhibitor survivin [27] (Figure 3A).
Figure 3.
Effect of IQ on various biomarkers in SCC4 cells. (A) Phosphorylation/expression of Akt, p38, Bax, Mcl-1, Bcl-2, and survivin in SCC4 cells. Cells were treated with IQ for 48 h, and cell lysates were immunoblotted as described in Materials and Methods. (B) Expression of p-p53 and p53 of IQ in SCC4 cells. (C) Western blotting of p-p53 and p53 in SCC4 cells transiently transfected with a control vector or p53 shRNA for 24 h. (D) IQ-induced cell viability in p53 knockdown SCC4 cells. After incubation, cells were analyzed using the MTT assays. Columns, mean; bars, S.D. ** p < 0.01.
Previous studies have revealed that Bax and apoptotic protease activating factor (APAF-1) could through the transcriptional activity of p53, a tumor suppressor gene, be implicated in OSCC [28,29]. Western blotting demonstrated that IQ increased p53 phosphorylation in SCC4 cells (Figure 3B). To further investigate the role of p53 in IQ-mediated cytotoxicity, cells were transiently transfected with p53 shRNA or a control vector, and then treated with IQ for 48 h. As shown in Figure 3C,D, p53 knockdown caused higher resistance to IQ’s viability inhibition activity.
3.4. IQ Increases Reactive Oxygen Species (ROS) Generation in SCC4 Cells
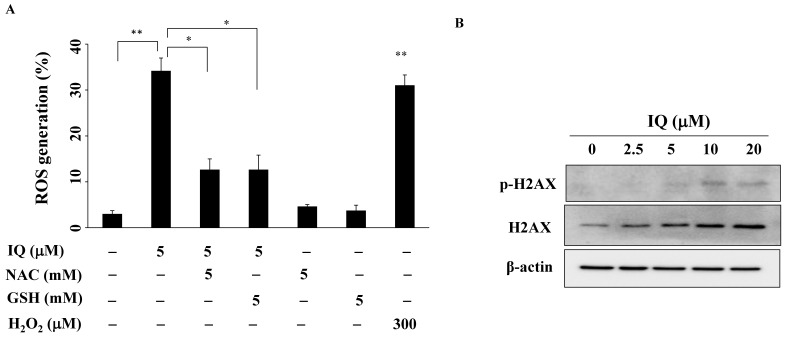
Growing evidence shows that ROS generation in oxidative stress causes DNA damage, which contributes to areca nut-induced OSCC carcinogenesis, and chemotherapy resistance [30,31]. After treatment with IQ, ROS levels in SCC4 cells were examined. Figure 4A shows that ROS generation was significantly higher in the IQ-treated groups, compared with the control group (H2O2 was the positive control), and could be partially reversed by pretreatment with the antioxidant N-acetylcysteine (NAC) or glutathione (GSH) for 15 min. Furthermore, in SCC4 cells, IQ upregulated the expression and phosphorylation of H2AX, a DNA damage response marker (Figure 4B).
Figure 4.
IQ increases reactive oxygen species (ROS) production. (A) Statistical analysis of ROS generation in SCC4 cells after the treatment with only 5 μM of IQ for 3 h or in combination with 5 mM of N-acetylcysteine (NAC) or 5 mM of glutathione (GSH). Cells were stained with carboxy-DCF-DA and analyzed using flow cytometry. Data are expressed as mean ± S.D. (n = 3). * p < 0.05 and ** p < 0.01. (B) IQ-induced H2AX phosphorylation/expression in SCC4 cells.
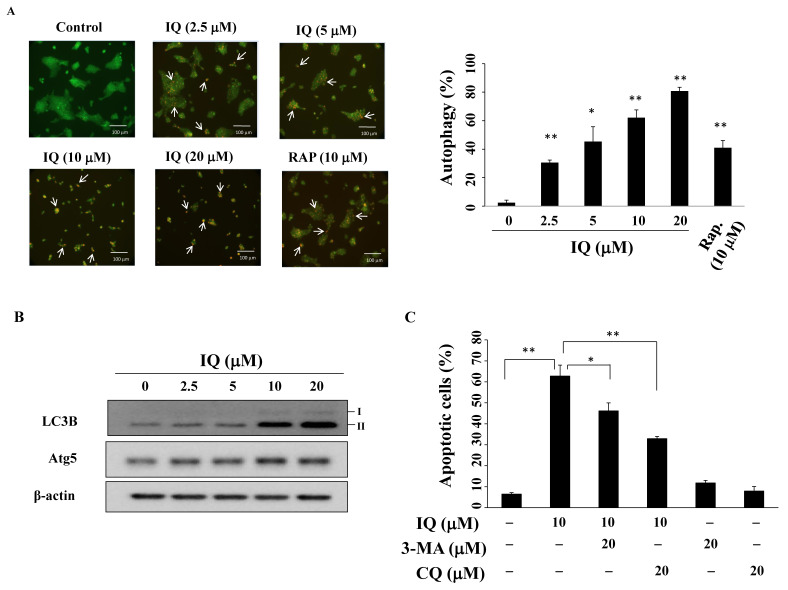
3.5. IQ induces Autophagy in SCC4 Cells
Autophagy, which can promote cell survival or death, plays a dual role in cancer therapy [32]. To investigate autophagy, acidic vesicular organelles (AVOs) were observed under a fluorescence microscope. During autophagy, the volume of the cellular acidic compartment is an autophagy marker that can be visualized using acridine orange staining [33]. After the treatment with IQ, an increase in the number of cytoplasmic AVOs in SCC4 cells was observed (Figure 5A, left panel; rapamycin as the positive control) and the ratio of acridine orange-positive cells increased in a concentration-dependent manner (Figure 5A, right panel). Western blotting demonstrated that IQ increased the expression of the autophagic biomarkers LC3B-II [34] and Atg5 [35] (Figure 5B). The autophagic inhibitor 3-methyladenine (3-MA) or chloroquine (CQ) was used to determine the role of autophagy in IQ-induced cell death in SCC4 cells. As shown in Figure 5C, pretreatment with either 3-MA or CQ decreased the percentage of IQ-induced apoptotic cells, indicating that IQ induced protective autophagy instead of autophagic cell death.
Figure 5.
Ilimaquinone (IQ) induces autophagy. (A) Left panel, SCC4 cells were treated with DMSO vehicle, IQ, or rapamycin (RAP) for 24 h, harvested and stained with acridine orange, and examined under a fluorescence microscope; arrows, acidic vesicular organelles (AVOs). Right panel, statistical analysis of AVOs in IQ-treated SCC4 cells. Columns, mean; bars, S.D. (n = 3). * p < 0.05 and ** p < 0.01 vs. the control group. (B) LC3B-II and Atg5 expression in SCC4 cells after IQ treatment for 48 h. (C) SCC4 cells were pretreated 20 μM of 3-methyladenine (3-MA) or 20 μM of chloroquine (CQ) for 15 min, followed by incubation with 10 μM IQ for 48 h and dual staining with propidium iodide (PI)/annexin V- fluorescein isothiocyanate (FITC). Percentages in the graphs are representative of cell percentage in the respective quadrants (n = 3). Columns, means; bars, S.D. * p < 0.05 and ** p < 0.01.
4. Discussion
Natural marine products have served as a source of potential pharmaceutical agents for centuries [36]. This study demonstrates that IQ, a marine sponge metabolite, can inhibit OSCC growth by mediating Akt downregulation, modulating Bcl-2 family and p53, increasing ROS, and inducing autophagy.
Multiple studies have revealed that some anti-cancer agents exert their effect by activating apoptosis, which is the major pharmacological mechanism in cancer therapy, including OSCC [36,37,38]. During apoptosis, the mitochondria and the activation of death receptors lead to the activation of caspase cascades [38,39]. Previous studies have reported that IQ induces apoptosis via death receptor-mediated extrinsic pathway activation and the downregulation of anti-apoptotic proteins such as Bcl-2 and Bcl-xL, which are related to mitochondria-mediated apoptosis in colon cancer cells [14]. Furthermore, multiple genes are involved in the control of apoptosis. For example, the Bcl-2 family’s oncogenic potential has been demonstrated in oral tumorigenesis [40]. This study reveals that IQ induces PARP cleavage and caspase-dependent apoptosis in SCC4 cells. In addition to the modulation of Mcl-1 and Bcl-2 expression, IQ treatment increased the level of Bax and decreases that of survivin.
The MAPK/p38 and PI3K/Akt signaling are involved in the regulation of the growth, metastasis, and apoptosis of OSCC, and the inhibition of Akt and p38 blocked the migration of OSCC cells and [23,41]. Akt activation has been correlated with the poor prognostic factor with local recurrence and five-year survival rate for OSCC [22]. Neoh et al. reported that flaccidoxide-13-acetate from the soft coral reduces the cell migration of bladder cancer cells through the down-related Akt/mTOR pathway [42]. Our results demonstrate that IQ downregulated the phosphorylation of Akt and p38 in OSCC cells. It has been reported that IQ increased p38 activation to sensitize TRAIL-induced apoptosis in colon cancer cells [14]. It is possible that there are different additional mechanisms regulated by IQ or the difference in cell lines used. Furthermore, our study shows that IQ inhibited the expression of HIF-1α, a key molecule in the regulation of hypoxia, which contributes to the tumor metastasis of OSCC [43].
One of the genes activated in response to several stress stimuli such as oncogene activation and DNA damage is p53 [44]. Immunohistochemical analysis revealed that there are over 60% OSCC patients with higher expression of p53, and the concentration of p53 of OSCC patients in saliva was significantly higher than in healthy participants [45,46]. Previous studies have shown that IQ inhibits cell growth in colon cancer by increasing p-p53 [18]. The results of this study indicate that treatment with IQ did not only increase p53 phosphorylation, but also partially protected against IQ-induced cytotoxicity after p53 knockdown in SCC4 cells.
Increased ROS levels act as an intracellular signal transducer, promoting autophagy, which is the pharmacological mechanism of some anticancer drugs currently in clinical use [47,48]. Do et al. reported that IQ increases ROS generation in TRAIL-induced apoptosis in colon cancer cells [14]. Similarly, we found that the antioxidant NAC or GSH partially rescued IQ-induced ROS production in SCC4 cells.
In tumor cells, autophagy can be activated by cellular stress stimulation, induced by starvation, a hypoxic environment, or cytotoxic agents [49]. In colon cancer and glioblastoma cells, IQ reportedly induced autophagy through p53 activation [18,50]. Similarly, our results show that IQ induces autophagy, as evidenced by the accumulation of autophagosomes in the cytoplasm, and LC3B-II and Atg5 expression in SCC4 cells. Interestingly, in the presence of the autophagic inhibitor 3-MA or CQ, the percentage of IQ-induced apoptotic cells decreased, suggesting that IQ induces protective autophagy in SCC4 cells.
5. Conclusions
In conclusion, IQ exerts anti-tumor effects either by inducing apoptosis and autophagy or by increasing ROS, revealing its translational potential as a new therapeutic strategy in OSCC treatment.
Supplementary Materials
The following are available online at https://www.mdpi.com/2227-9059/8/9/296/s1, Figure S1: 1H NMR spectrum of ilimaquinone in CDCl3 at 400 MHz; Figure S2: 13C NMR spectrum of ilimaquinone in CDCl3 at 100 MHz; Figure S3: HMQC spectrum of ilimaquinone in CDCl3; Figure S4: HMBC spectrum of ilimaquinone in CDCl3; Figure S5: 1H–1H COSY spectrum of ilimaquinone in CDCl3.
Author Contributions
C.-W.L. and L.-Y.B. wrote the manuscript; J.-H.S. performed some of the experiments; C.-F.C. and W.-Y.L. contributed to the data analysis; W.-T.H., M.-C.S., and Y.-T.H. isolated and characterized ilimaquinone; J.-L.H. performed part of the experiments; J.-R.W. designed and supervised the study, and checked the final manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by grants from the Ministry of Science and Technology (MOST 106-2320-B-110-003-MY3, MOST 109-2321-8-006-011-), the Ministry of Health and Welfare, China Medical University Hospital Cancer Research Center of Excellence (MOHW108-TDU-B-212-112015, MOHW109-TDU-B-212-010001, MOHW109-TDU-B-212-134026), the National Health Research Institutes (NHRI-109A1-CACO-13202002), and Kaohsiung Armed Forces General Hospital, Taiwan.
Conflicts of Interest
The authors declare no competing financial interests.
References
- 1.Warnakulasuriya S. Global epidemiology of oral and oropharyngeal cancer. Oral Oncol. 2009;45:309–316. doi: 10.1016/j.oraloncology.2008.06.002. [DOI] [PubMed] [Google Scholar]
- 2.Ghantous Y., Abu Elnaaj I. Global incidence and risk factors of oral cancer. Harefuah. 2017;156:645–649. [PubMed] [Google Scholar]
- 3.Kumar M., Nanavati R., Modi T.G., Dobariya C. Oral cancer: Etiology and risk factors: A review. J. Cancer Res. Ther. 2016;12:458–463. doi: 10.4103/0973-1482.186696. [DOI] [PubMed] [Google Scholar]
- 4.Lakshminarayana S., Augustine D., Rao R.S., Patil S., Awan K.H., Venkatesiah S.S., Haragannavar V.C., Nambiar S., Prasad K. Molecular pathways of oral cancer that predict prognosis and survival: A systematic review. J. Carcinog. 2018;17:7. doi: 10.4103/jcar.JCar_17_18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chatzistefanou I., Lubek J., Markou K., Ord R.A. The role of perineural invasion in treatment decisions for oral cancer patients: A review of the literature. J. Cranio-Maxillo-Facial Surg. 2017;45:821–825. doi: 10.1016/j.jcms.2017.02.022. [DOI] [PubMed] [Google Scholar]
- 6.Newman D.J., Cragg G.M. Natural products as sources of new drugs from 1981 to 2014. J. Nat. Prod. 2016;79:629–661. doi: 10.1021/acs.jnatprod.5b01055. [DOI] [PubMed] [Google Scholar]
- 7.Ngo D.H., Vo T.S., Ngo D.N., Wijesekara I., Kim S.K. Biological activities and potential health benefits of bioactive peptides derived from marine organisms. Int. J. Biol. Macromol. 2012;51:378–383. doi: 10.1016/j.ijbiomac.2012.06.001. [DOI] [PubMed] [Google Scholar]
- 8.Rangel M., Falkenberg M. An overview of the marine natural products in clinical trials and on the market. J. Coast. Life Med. 2015;3:471–478. [Google Scholar]
- 9.Aftimos P., Awada A. Survival benefit of eribulin mesylate in heavily pretreated metastatic breast cancer: What next? Adv. Ther. 2011;28:973–985. doi: 10.1007/s12325-011-0070-9. [DOI] [PubMed] [Google Scholar]
- 10.Mehbub M.F., Lei J., Franco C., Zhang W. Marine sponge derived natural products between 2001 and 2010: Trends and opportunities for discovery of bioactives. Mar. Drugs. 2014;12:4539–4577. doi: 10.3390/md12084539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kiem P.V., Huyen L.T., Hang D.T., Nhiem N.X., Tai B.H., Anh H.L., Cuong P.V., Quang T.H., Minh C.V., Dau N.V., et al. Sesquiterpene derivatives from marine sponge smenospongia cerebriformis and their anti-inflammatory activity. Bioorganic Med. Chem. Lett. 2017;27:1525–1529. doi: 10.1016/j.bmcl.2017.02.040. [DOI] [PubMed] [Google Scholar]
- 12.Simithy J., Fuanta N.R., Hobrath J.V., Kochanowska-Karamyan A., Hamann M.T., Goodwin D.C., Calderon A.I. Mechanism of irreversible inhibition of mycobacterium tuberculosis shikimate kinase by ilimaquinone. Biochim. Et. Biophys. Acta. Proteins Proteom. 2018;1866:731–739. doi: 10.1016/j.bbapap.2018.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Loya S., Hizi A. The interaction of illimaquinone, a selective inhibitor of the rnase h activity, with the reverse transcriptases of human immunodeficiency and murine leukemia retroviruses. J. Biol. Chem. 1993;268:9323–9328. [PubMed] [Google Scholar]
- 14.Do M.T., Na M., Kim H.G., Khanal T., Choi J.H., Jin S.W., Oh S.H., Hwang I.H., Chung Y.C., Kim H.S., et al. Ilimaquinone induces death receptor expression and sensitizes human colon cancer cells to trail-induced apoptosis through activation of ros-erk/p38 mapk-chop signaling pathways. Food Chem. Toxicol. 2014;71:51–59. doi: 10.1016/j.fct.2014.06.001. [DOI] [PubMed] [Google Scholar]
- 15.Sonoda H., Okada T., Jahangeer S., Nakamura S. Requirement of phospholipase d for ilimaquinone-induced golgi membrane fragmentation. J. Biol. Chem. 2007;282:34085–34092. doi: 10.1074/jbc.M705593200. [DOI] [PubMed] [Google Scholar]
- 16.Lu P.H., Chueh S.C., Kung F.L., Pan S.L., Shen Y.C., Guh J.H. Ilimaquinone, a marine sponge metabolite, displays anticancer activity via gadd153-mediated pathway. Eur. J. Pharmacol. 2007;556:45–54. doi: 10.1016/j.ejphar.2006.10.061. [DOI] [PubMed] [Google Scholar]
- 17.Park S., Yun E., Hwang I.H., Yoon S., Kim D.E., Kim J.S., Na M., Song G.Y., Oh S. Ilimaquinone and ethylsmenoquinone, marine sponge metabolites, suppress the proliferation of multiple myeloma cells by down-regulating the level of beta-catenin. Mar. Drugs. 2014;12:3231–3244. doi: 10.3390/md12063231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lee H.Y., Chung K.J., Hwang I.H., Gwak J., Park S., Ju B.G., Yun E., Kim D.E., Chung Y.H., Na M., et al. Activation of p53 with ilimaquinone and ethylsmenoquinone, marine sponge metabolites, induces apoptosis and autophagy in colon cancer cells. Mar. Drugs. 2015;13:543–557. doi: 10.3390/md13010543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ribeiro M., Teixeira S., Azevedo M.N., Fraga A.C., Jr., Gontijo A.P., Vêncio E.F. Ilimaquinone, a sesquiterpenoid quinone, induces apoptosis and autophagy. Tetrahedron. 1979;35:609–612. [Google Scholar]
- 20.Weng J.R., Bai L.Y., Chiu S.J., Chiu C.F., Lin W.Y., Hu J.L., Shieh T.M. Divaricoside exerts antitumor effects, in part, by modulating mcl-1 in human oral squamous cell carcinoma cells. Comput. Struct. Biotechnol. J. 2019;17:151–159. doi: 10.1016/j.csbj.2019.01.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Bai L.Y., Chiu C.F., Chiu S.J., Chu P.C., Weng J.R. Fty720 induces autophagy-associated apoptosis in human oral squamous carcinoma cells, in part, through a reactive oxygen species/mcl-1-dependent mechanism. Sci. Rep. 2017;7:5600. doi: 10.1038/s41598-017-06047-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lim J., Kim J.H., Paeng J.Y., Kim M.J., Hong S.D., Lee J.I., Hong S.P. Prognostic value of activated akt expression in oral squamous cell carcinoma. J. Clin. Pathol. 2005;58:1199–1205. doi: 10.1136/jcp.2004.024786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Brusevold I.J., Aasrum M., Bryne M., Christoffersen T. Migration induced by epidermal and hepatocyte growth factors in oral squamous carcinoma cells in vitro: Role of mek/erk, p38 and pi-3 kinase/akt. J. Oral Pathol. Med. 2012;41:547–558. doi: 10.1111/j.1600-0714.2012.01139.x. [DOI] [PubMed] [Google Scholar]
- 24.Chen S.F., Nieh S., Jao S.W., Liu C.L., Wu C.H., Chang Y.C., Yang C.Y., Lin Y.S. Quercetin suppresses drug-resistant spheres via the p38 mapk-hsp27 apoptotic pathway in oral cancer cells. PLoS ONE. 2012;7:e49275. doi: 10.1371/journal.pone.0049275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Smolensky D., Rathore K., Bourn J., Cekanova M. Inhibition of the pi3k/akt pathway sensitizes oral squamous cell carcinoma cells to anthracycline-based chemotherapy in vitro. J. Cell. Biochem. 2017;118:2615–2624. doi: 10.1002/jcb.25747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Qian J., Wenguang X., Zhiyong W., Yuntao Z., Wei H. Hypoxia inducible factor: A potential prognostic biomarker in oral squamous cell carcinoma. Tumour Biol. 2016;37:10815. doi: 10.1007/s13277-016-4976-3. [DOI] [PubMed] [Google Scholar]
- 27.Poomsawat S., Punyasingh J., Vejchapipat P. Overexpression of survivin and caspase 3 in oral carcinogenesis. Appl. Immunohistochem. Mol. Morphol. 2014;22:65–71. doi: 10.1097/PAI.0b013e31828a0d0c. [DOI] [PubMed] [Google Scholar]
- 28.Selvakumaran M., Lin H.K., Miyashita T., Wang H.G., Krajewski S., Reed J.C., Hoffman B., Liebermann D. Immediate early up-regulation of bax expression by p53 but not tgf beta 1: A paradigm for distinct apoptotic pathways. Oncogene. 1994;9:1791–1798. [PubMed] [Google Scholar]
- 29.Sinevici N., O’Sullivan J. Oral cancer: Deregulated molecular events and their use as biomarkers. Oral Oncol. 2016;61:12–18. doi: 10.1016/j.oraloncology.2016.07.013. [DOI] [PubMed] [Google Scholar]
- 30.Ji W.T., Yang S.R., Chen J.Y., Cheng Y.P., Lee Y.R., Chiang M.K., Chen H.R. Arecoline downregulates levels of p21 and p27 through the reactive oxygen species/mtor complex 1 pathway and may contribute to oral squamous cell carcinoma. Cancer Sci. 2012;103:1221–1229. doi: 10.1111/j.1349-7006.2012.02294.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Xu Y., Gao W., Zhang Y., Wu S., Liu Y., Deng X., Xie L., Yang J., Yu H., Su J., et al. Abt737 reverses cisplatin resistance by targeting glucose metabolism of human ovarian cancer cells. Int. J. Oncol. 2018;53:1055–1068. doi: 10.3892/ijo.2018.4476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Salazar M., Carracedo A., Salanueva I.J., Hernandez-Tiedra S., Lorente M., Egia A., Vazquez P., Blazquez C., Torres S., Garcia S., et al. Cannabinoid action induces autophagy-mediated cell death through stimulation of er stress in human glioma cells. J. Clin. Investig. 2009;119:1359–1372. doi: 10.1172/JCI37948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Paglin S., Hollister T., Delohery T., Hackett N., McMahill M., Sphicas E., Domingo D., Yahalom J. A novel response of cancer cells to radiation involves autophagy and formation of acidic vesicles. Cancer Res. 2001;61:439–444. [PubMed] [Google Scholar]
- 34.Liu Y., Shi Q.F., Ye Y.C., Tashiro S., Onodera S., Ikejima T. Activated o2(*-) and h2o2 mediated cell survival in su11274-treated non-small-cell lung cancer a549 cells via c-met-pi3k-akt and c-met-grb2/sos-ras-p38 pathways. J. Pharmacol. Sci. 2012;119:150–159. doi: 10.1254/jphs.12048FP. [DOI] [PubMed] [Google Scholar]
- 35.Miller B.C., Zhao Z., Stephenson L.M., Cadwell K., Pua H.H., Lee H.K., Mizushima N.N., Iwasaki A., He Y.W., Swat W., et al. The autophagy gene atg5 plays an essential role in b lymphocyte development. Autophagy. 2008;4:309–314. doi: 10.4161/auto.5474. [DOI] [PubMed] [Google Scholar]
- 36.Mayer A.M., Rodriguez A.D., Berlinck R.G., Hamann M.T. Marine pharmacology in 2005-6: Marine compounds with anthelmintic, antibacterial, anticoagulant, antifungal, anti-inflammatory, antimalarial, antiprotozoal, antituberculosis, and antiviral activities; affecting the cardiovascular, immune and nervous systems, and other miscellaneous mechanisms of action. Biochim. Et. Biophys. Acta. 2009;1790:283–308. doi: 10.1016/j.bbagen.2009.03.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Kuo Y.Y., Jim W.T., Su L.C., Chung C.J., Lin C.Y., Huo C., Tseng J.C., Huang S.H., Lai C.J., Chen B.C., et al. Caffeic acid phenethyl ester is a potential therapeutic agent for oral cancer. Int. J. Mol. Sci. 2015;16:10748–10766. doi: 10.3390/ijms160510748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Wu H., Li L., Ai Z., Yin J., Chen L. Pristimerin induces apoptosis of oral squamous cell carcinoma cells via g1 phase arrest and mapk/erk1/2 and akt signaling inhibition. Oncol. Lett. 2019;17:3017–3025. doi: 10.3892/ol.2019.9903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Herr I., Debatin K.M. Cellular stress response and apoptosis in cancer therapy. Blood. 2001;98:2603–2614. doi: 10.1182/blood.V98.9.2603. [DOI] [PubMed] [Google Scholar]
- 40.Popovic B., Jekic B., Novakovic I., Lukovic L.J., Tepavcevic Z., Jurisic V., Vukadinovic M., Milasin J. Bcl-2 expression in oral squamous cell carcinoma. Ann. New York Acad. Sci. 2007;1095:19–25. doi: 10.1196/annals.1397.003. [DOI] [PubMed] [Google Scholar]
- 41.Kashyap T., Pramanik K.K., Nath N., Mishra P., Singh A.K., Nagini S., Rana A., Mishra R. Crosstalk between raf-mek-erk and pi3k-akt-gsk3beta signaling networks promotes chemoresistance, invasion/migration and stemness via expression of cd44 variants (v4 and v6) in oral cancer. Oral Oncol. 2018;86:234–243. doi: 10.1016/j.oraloncology.2018.09.028. [DOI] [PubMed] [Google Scholar]
- 42.Neoh C.A., Wu W.T., Dai G.F., Su J.H., Liu C.I., Su T.R., Wu Y.J. Flaccidoxide-13-acetate extracted from the soft coral cladiella kashmani reduces human bladder cancer cell migration and invasion through reducing activation of the fak/pi3k/akt/mtor signaling pathway. Molecules. 2017;23:58. doi: 10.3390/molecules23010058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Zhou J., Huang S., Wang L., Yuan X., Dong Q., Zhang D., Wang X. Clinical and prognostic significance of hif-1alpha overexpression in oral squamous cell carcinoma: A meta-analysis. World J. Surg. Oncol. 2017;15:1–8. doi: 10.1186/s12957-017-1163-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Aubrey B.J., Kelly G.L., Janic A., Herold M.J., Strasser A. How does p53 induce apoptosis and how does this relate to p53-mediated tumour suppression? Cell Death Differ. 2018;25:104–113. doi: 10.1038/cdd.2017.169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Verma R., Singh A., Jaiswal R., Chandra A., Verma R., Tak J. Association of ki-67 antigen and p53 protein at invasive tumor front of oral squamous cell carcinoma. Indian J. Pathol. Microbiol. 2014;57:553–557. doi: 10.4103/0377-4929.142660. [DOI] [PubMed] [Google Scholar]
- 46.Agha-Hosseini F., Mirzaii-Dizgah I., Miri-Zarandi N. Unstimulated salivary p53 in patients with oral lichen planus and squamous cell carcinoma. Acta Med. Iran. 2015;53:439–443. [PubMed] [Google Scholar]
- 47.Zou Z., Chang H., Li H., Wang S. Induction of reactive oxygen species: An emerging approach for cancer therapy. Apoptosis. 2017;22:1321–1335. doi: 10.1007/s10495-017-1424-9. [DOI] [PubMed] [Google Scholar]
- 48.Gwangwa M.V., Joubert A.M., Visagie M.H. Crosstalk between the warburg effect, redox regulation and autophagy induction in tumourigenesis. Cell. Mol. Biol. Lett. 2018;23:20. doi: 10.1186/s11658-018-0088-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Adhauliya N., Kalappanavar A.N., Ali I.M., Annigeri R.G. Autophagy: A boon or bane in oral cancer. Oral Oncol. 2016;61:120–126. doi: 10.1016/j.oraloncology.2016.09.001. [DOI] [PubMed] [Google Scholar]
- 50.Ratovitski E.A. Tumor protein (tp)-p53 members as regulators of autophagy in tumor cells upon marine drug exposure. Mar. Drugs. 2016;14:154. doi: 10.3390/md14080154. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.