Abstract
Background
Congenital heart disease (CHD) is common and is associated with impaired early brain development and neurodevelopmental outcomes, yet the exact mechanisms underlying these associations are unclear.
Purpose
To utilize MRI data from a cohort of fetuses with CHD as well as typically developing fetuses to test the hypothesis that expected cerebral substrate delivery is associated with total and regional fetal brain volumes.
Study Type
Retrospective case−control study.
Population
Three hundred eighty fetuses (188 male), comprising 45 healthy controls and 335 with isolated CHD, scanned between 29 and 37 weeks gestation. Fetuses with CHD were assigned into one of four groups based on expected cerebral substrate delivery.
Field Strength/Sequence
T2-weighted single-shot fast-spin-echo sequences and a balanced steady-state free precession gradient echo sequence were obtained on a 1.5 T scanner.
Assessment
Images were motion-corrected and reconstructed using an automated slice-to-volume registration reconstruction technique, before undergoing segmentation using an automated pipeline and convolutional neural network that had undergone semi-supervised training. Differences in total, regional brain (cortical gray matter, white matter, deep gray matter, cerebellum, and brainstem) and brain:body volumes were compared between groups.
Statistical Tests
ANOVA was used to test for differences in brain volumes between groups, after accounting for sex and gestational age at scan. PFDR-values <0.05 were considered statistically significant.
Results
Total and regional brain volumes were smaller in fetuses where cerebral substrate delivery is reduced. No significant differences were observed in total or regional brain volumes between control fetuses and fetuses with CHD but normal cerebral substrate delivery (all PFDR > 0.12). Severely reduced cerebral substrate delivery is associated with lower brain:body volume ratios.
Data Conclusion
Total and regional brain volumes are smaller in fetuses with CHD where there is a reduction in cerebral substrate delivery, but not in those where cerebral substrate delivery is expected to be normal.
Evidence Level
3 Technical Efficacy: Stage: 3
Congenital heart disease (CHD) is the most common group of congenital malformations, affecting ∼1% of live births.1 CHD is also associated with developmental impairments across multiple domains including speech and language, motor coordination and cognition.2
The mechanisms which underlie these impairments remain unclear, but are likely to be multifactorial, with periods of postnatal hypoxia, the surgery and associated pro-longed hospital stays all implicated.3,4 Notably, MRI studies have shown that brain development in infants with CHD is abnormal prior to surgery, including an increased risk of brain injury and reduced global and regional volumes.5–8
There is mounting evidence that CHD is associated with impaired brain development in-utero, with multiple studies identifying smaller total9–11 and regional12,13 brain volumes in different types of CHD.14–17 Underlying cardiac physiology and subsequent cerebrovascular hemodynamics play an important role, particularly in the presence of reduced cerebral oxygenation.18–20
In the normal fetus, inter-atrial and systemic arterial shunts are present, with both the right and left sides of the heart supporting the fetal circulation in parallel. A notable feature of the normal fetal circulation is the preferential streaming of oxygen and nutrient rich placental blood to the fetal brain. The umbilical vein enters the fetus via the umbilicus, and after a portion then drains to the portal circulation, the remainder is shunted to the right heart via the ductus venosus. This blood is then preferentially streamed across the patent foramen ovale by the Eustachian valve into the left atrium, left ventricle and ultimately to the developing fetal brain.21
The effects of CHD on the substrate content, including oxygen, glucose, and other nutrients, of blood reaching the fetal brain have been categorized in various ways, based on both directly measured MRI data, and the theoretical impacts based on underlying anatomy.18,22,23 Different types of CHD will impact this streaming pattern in different ways, with potentially important implications for the delivery of substrate-rich blood to the developing brain.11,19,24 For example, in some conditions, such as a right aortic arch or a double aortic arch, there is no theoretical disruption either to intracardiac streaming or the vascular pathway of streamed blood from heart to the brain.25 In fetuses with suspected coarctation of the aorta (CoA), a cardinal sign is flow reversal at the aortic isthmus,26 which, in the absence of other major lesions will result in a variable amount of fetal systemic venous blood being redirected back to the cerebral circulation. In lesions where there is only one functional outlet to the heart, such as truncus arteriosus, pulmonary atresia or hypoplastic left heart syndrome, placental and fetal systemic venous blood are necessarily mixed before leaving the heart, and the substrate availability of blood reaching the brain is further reduced.19 In transposition of the great arteries (TGA), the two main arteries leaving the heart—the pulmonary artery and the aorta—arise from the left and right ventricles respectively, leading to a complete reversal of normal fetal streaming patterns, with systemic venous return comprising the majority of cerebral blood flow.19
Imaging the fetal brain remains challenging. Fetal and maternal motion can cause image artifacts. The fetal brain is small, and some structures such as the cortex, are only a few millimeters thick. As such, high-resolution imaging is required to delineate brain structures posing challenges for signal-to-noise ratio (SNR). Recent advances in MRI acquisition and reconstruction techniques enable sub-millimeter resolution imaging of the fetal brain that is resistant to fetal motion.27 This allows characterization of early brain volumetric development in both typically developing fetuses and those with pathology affecting brain growth, including CHD.
Previous studies have focused on individual cardiac diagnoses, such as TGA or HLHS, and sometimes treat all types of CHD as a heterogeneous group.9,10,14,28 Additionally, previous work has focused largely on CHD lesions defined as critical or severe, requiring early postnatal intervention, with other less severe forms of CHD less well described.10,14,29–31 When considering brain development in CHD, a physiological rather than diagnostic approach may be beneficial, particularly with the potential benefits this affords when it comes to understanding the biological mechanisms behind why brain growth may be impaired in an individual fetus with CHD.
The aim of this study was to utilize advanced fetal neuroimaging data from a large cohort of fetuses with suspected or confirmed CHD as well as typically developing fetuses to test the hypothesis that expected cerebral substrate delivery in-utero is associated with total and regional volumes in the fetal brain.
Materials and Methods
Ethical Approval
The National Research Ethics Service West London committee provided ethical approval (07/H0707/105; 14/LO/1806; 17/LO/0292). Informed written consent was obtained before fetal MRI.
Subjects
For this retrospective study, all imaging data was acquired between June 2014 and June 2022. For the CHD cohort, 529 women (age at scan = 31.52 [±5.73] years) carrying a fetus with known or suspected CHD who consented for research prior to clinical fetal cardiac MRI scans, or from research MRI scans where appropriate consents were already in place for analysis of these data. For the control cohort, pregnant women undergoing fetal MRI beyond 29 weeks gestation who consented to the images being used for research were included. For both cohorts, maternal exclusion criteria included multiple pregnancy, weight over 125 kg, severe claustrophobia, inability to give informed consent, or age under 18 years at the time of referral. Fetal exclusion criteria included: Confirmed genetic abnormalities on antenatal or postnatal genetic testing (either from amniocentesis, comparative genomic hybridization array [CGH-array], or clinical genetic testing); major fetal extracardiac anomalies, such as congenital diaphragmatic hernia or duodenal atresia; or structural brain abnormalities as reported on fetal MRI, including bilateral ventriculomegaly, cerebellar hypoplasia or absence of the corpus callosum.
Mr Image Acquisition
Fetal MR images were acquired on a Philips Ingenia 1.5 T scanner, with 28-channel dStream anterior and posterior in-built coils. Fetal brain images were acquired using a 2D T2-weighted fast-spin-echo sequence (Repetition Time [TR] = 15,000 msec, Echo Time [TE] = 80 msec, voxel size = 1.25 × 1.25, slice thickness 2.5 mm, slice spacing 1.25 mm, field-of-view = 420 × 420 mm, matrix = 336 × 336, SENSE factor = 2, flip angle 90°, bandwidth = 413 Hz), optimized for fetal imaging.32 To image the fetal body, sagittal whole-uterus images were acquired using a balanced steady-state free precession (bSSFP) sequence (TR = 5 msec, TE = 1.98 msec, voxel size = 1.25 × 1.25, slice thickness 5 mm, field-of-view = 420 × 420 mm, matrix = 288 × 288, SENSE factor = 2, flip angle = 90R0, bandwidth = 689 Hz). No sedation or intravenous contrast was used.
Fetal Brain Image Reconstruction
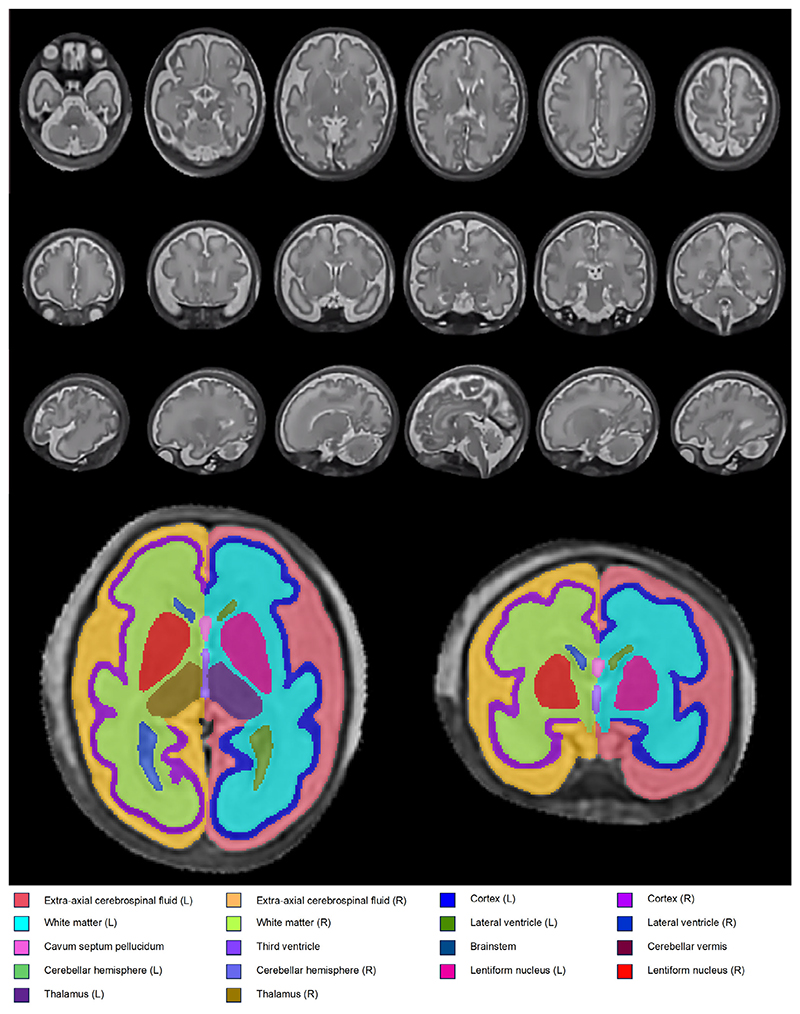
Images were motion corrected and reconstructed using an automated version of the classical 3D slice-to-volume registration (SVR) reconstruction technique,33,34 (auto brain reconstruction SVRTK docker—https://github.com/SVRTK/SVRTK; https://hub.docker.com/r/fetalsvrtk/svrtk tag auto-2.20), before being up sampled to 0.5 mm3 isotropic resolution (Fig. 1).
Figure 1. Example 3D slice-to-volume reconstructions of T2-weighted fetal brain MR images in coronal (first row), axial (second row), and sagittal (third row) views.
These images were acquired at a gestational age of 34+0 weeks. Regional segmentations, based on the developing Human Connectome Project atlas segmentation protocol with 19 label regions of interest, generated by BOUNTI automated fetal brain MRI segmentation tool are shown in axial (bottom left) and coronal (bottom right) slices.
Image Review
MR images were reported by perinatal neurora-diologists (MR, AE), both with over 10 years experience. The presence of any structural abnormalities was recorded. The quality of the fetal brain reconstructions, taking into account the definition of anatomical features, signal to noise ratio, contrast and alignment, was independently assessed by two clinicians (DC, PC) trained in the assessment of fetal and neonatal brain MRI (DC = 5 years experience, PC = 4 years experience), with each image being graded as good (4/4), acceptable (3/4), poor (2/4) or failed (1/4), as previously described.35 Only datasets where both reviewers gave a rating of acceptable or good were included. An example of a good quality reconstruction is shown in Fig. 1.
Fetal Brain Segmentation
Each brain SVR was segmented using an automated Brain vOlumetry and aUtomated parcellatioN for 3D feTal MRI (BOUNTI) pipeline36 (SVRTK segmentation docker: https://hub.docker.com/r/fetalsvrtk/segmentation tag bounti_brain_tissue_1.00) based on the developing Human Connectome Project atlas segmentation protocol with 19 label regions of interest (https://gin.g-node.org/kcl_cdb/fetal_brain_mri_atlas) and a combination of 3D UNet and Attention UNet37 convolutional neural networks that had undergone semi-supervised training in the Medical Open Network for Artificial Intelligence (MONAI). All resulting segmentations were reviewed and failed or inaccurate segmentations were excluded. Those confirmed to be acceptable for volumetry were kept. No manual editing was performed on segmentations.
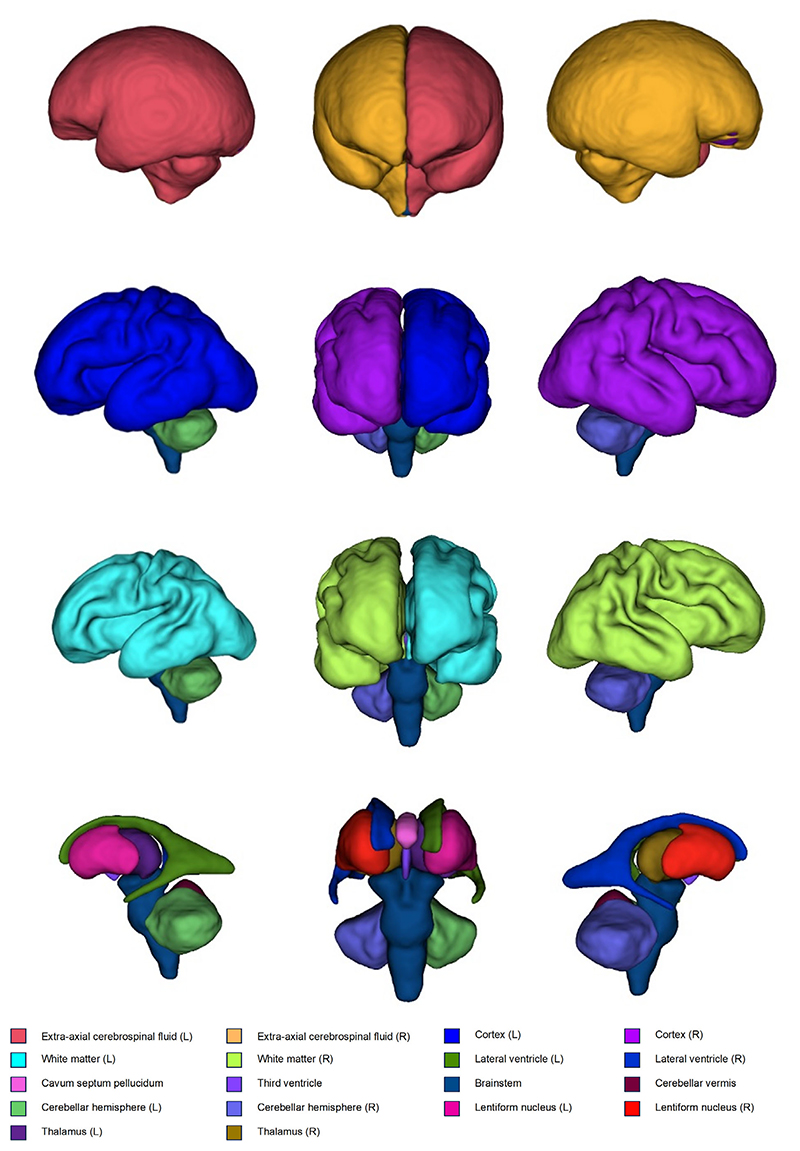
Volumes were generated for five brain tissue regions: 1) cortical gray matter (cGM); 2) white matter (WM); 3) deep gray matter (dGM) (thalamus + lentiform nucleus); 4) cerebellum (cerebellar hemispheres + cerebellar vermis); and 5) brainstem. Volumes were also calculated for total cerebrospinal fluid (CSF) by summing the volume of extra-axial CSF, lateral ventricles, cavum septum pellucidum, and the third fourth ventricles. Total brain tissue volume was calculated by summing cGM, WM, dGM, cerebellum, and brainstem. These segmentations and their volumetric representations can be seen in Figs. 1 and 2.
Figure 2. 3D representations of fetal brain MRI segmentations, for a fetus imaged at 34+0 weeks, from which total and regional volumes were generated.
Fetal Body Segmentation
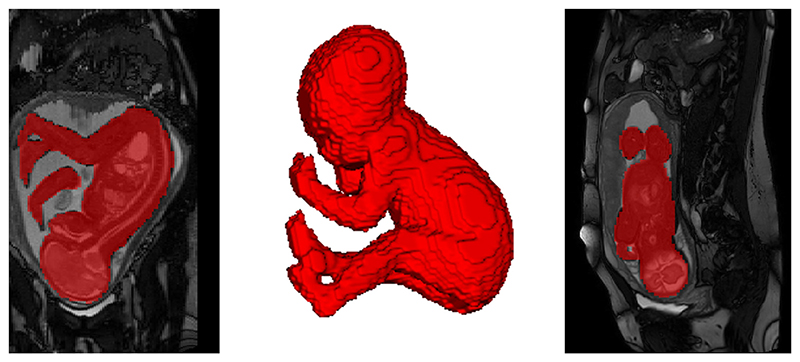
A separate 3D UNet37 convolutional neural network that had undergone semi-supervised training in MONAI was used to generate fetal whole-body segmentations, using the sagittal whole-uterus T2-weighted bSSFP images (SVRTK segmentation docker: https://hub.docker.com/r/fetalsvrtk/segmentation tag btfe_whole_body). The whole-uterus images and resulting segmentations were independently assessed for motion artifacts and whether there was any cropping of the fetal body (by DC, 5 years experience & MVP, 6 years experience). Datasets with minimal motion artifact, no cropping of the region of interest, and fetal body segmentations considered good quality were included (Fig. 3). Estimated fetal body volumes were derived from these whole-body segmentations using ITK-snap.38
Figure 3. Automatically generated whole-fetus segmentations, allowing estimation of total fetal body volumes, from T2-weighted balanced steady-state free precession (bSSFP) sequences.
Coronal bSSFP + segmentation (left), 3D fetal body rendering (middle), Sagittal bSSFP + segmentation (right).
Fetal CHD Categorization
In the absence of direct measurements of cerebral substrate delivery in this cohort, cases were classified according to the expected effect of the underlying cardiac defect, using MRI derived fetal blood flows where available, as described in Lloyd et al,26 on the oxygen and nutritional content of blood delivered to the carotid arteries, and therefore the brain (referred to onwards for brevity as “cerebral substrate content”). Fetal echocardiogram data were acquired as per routine clinical care and where information about the direction of blood flow at the aortic isthmus was important for CHD categorization, and this information was not otherwise available from MRI derived fetal blood flows, it was extracted from the clinical fetal echocardiogram report. Other speculative effects on streaming, abnormal flow patterns or determination of specific substrates were not included. This led to the development of four distinct categories:
Substrate content of cerebral blood is expected to be normal;
Substrate content of cerebral blood is expected to be mildly reduced (i.e., some mixing of placental and fetal systemic venous blood);
Substrate content of cerebral blood is expected to be moderately reduced (i.e., complete mixing of placental and fetal systemic venous blood);
Substrate content of cerebral blood is expected to be severely reduced (i.e., complete reversal of normal placental streaming).
In cases where a diagnosis could potentially fit into more than one category, depending on severity or underlying hemodynamics, a combination of phase contrast (with metric optimized gating) fetal flow measurements (described in Refs.26,39) and/or contemporaneously acquired echocardiographic data were used to assign cases individually, following assessment of the data by a fetal cardiologist.
A summary of all diagnoses, including the rationale for inclusion in the categories above, is given in Table S1 in the Supplemental Material.
Statistical Analyses
All statistical analyses were performed using statsmodels (v0.13.2) and Jupyter Notebook, python3. A one-way ANOVA was used to test for differences in volumes between groups, after accounting for relevant confounders. We first performed an analysis comparing total and regional brain volumes in the “control” group of typically developing fetuses (Group 0) and fetuses with CHD where cerebral substrate delivery is expected to be normal (Group 1), after accounting for GA at scan and fetal sex. We then performed an analysis comparing total and regional brain volumes between groups for all fetuses in the study. For this analysis, both typically developing fetuses and fetuses with CHD where cerebral substrate delivery is expected to be normal were assigned to the same group, with the label “normal cerebral substrate delivery.” We also ran an additional post-hoc analysis to explore whether there were any differences in fetal brain:body ratios between each of the groups described above, after accounting for GA at scan and fetal sex. Benjamini and Hochberg false discovery rate (FDR) was applied to correct for multiple comparisons (reported as PFDR). PFDR-values <0.05 were considered statistically significant.
Results
Demographics
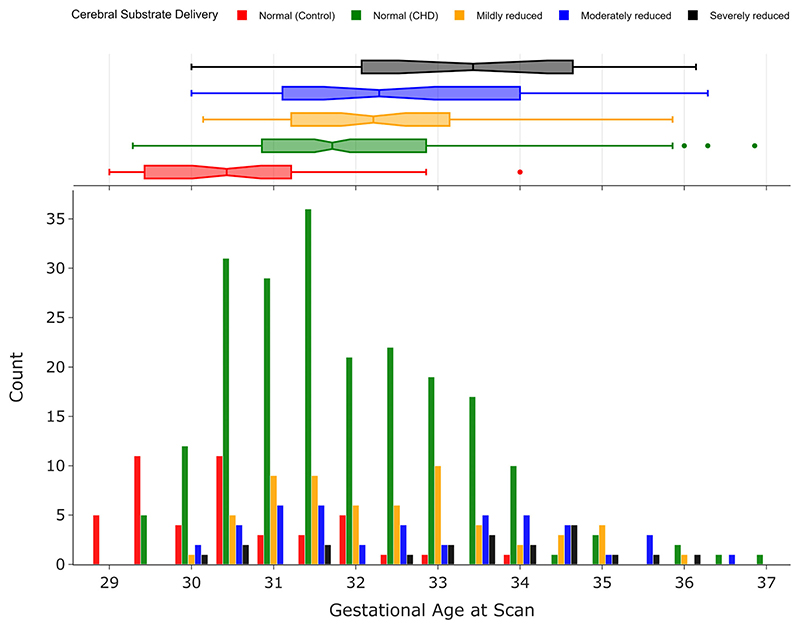
Data from 380 fetuses me the inclusion criteria: 45 healthy controls and 335 with CHD. A breakdown of the number of fetuses allocated to each of the different groups based on expected cerebral oxygenation and the median (IQR) age at scan for each group is shown in Table 1. There were 192 female fetuses and 188 males. There was a significant difference in GA at scan (), but not fetal sex (F = 2.01, P = 0.28) between groups. Histograms showing the distribution of GA at scan for each group are shown in Fig. 4.
Table 1. Classification of Congenital Heart Disease (CHD) According to Expected Cerebral Substrate Delivery in Study Population of 380 Fetuses.
| Group | N (M:F) | Median (IQR) GA at Scan (Weeks) |
|---|---|---|
| (0) Control | 45 (22:23) | 30.43 (29.43−31.14) |
| (1) Substrate content of cerebral blood is expected to be normal | 210 (103:107) | 31.71 (30.86−32.86) |
| (2) Substrate content of cerebral blood is expected to be mildly reduced | 60 (33:27) | 32.21 (31.25−33.14) |
| (3) Substrate content of cerebral blood is expected to be moderately reduced | 45 (20:25) | 32.29 (31.14−34.0) |
| (4) Substrate content of cerebral blood is expected to be severely reduced | 20 (10:10) | 33.43 (32.32−34.61) |
| TOTAL | 380 (188:192) | 31.71 (30.86−33.00) |
Figure 4. Histograms showing counts for gestational age at the time of MRI scan for all 380 fetuses (192 female, 188 male) included in this study, classified according to their expected cerebral substrate delivery.
CHD With Expected Normal Cerebral Substrate Delivery Compared to Controls
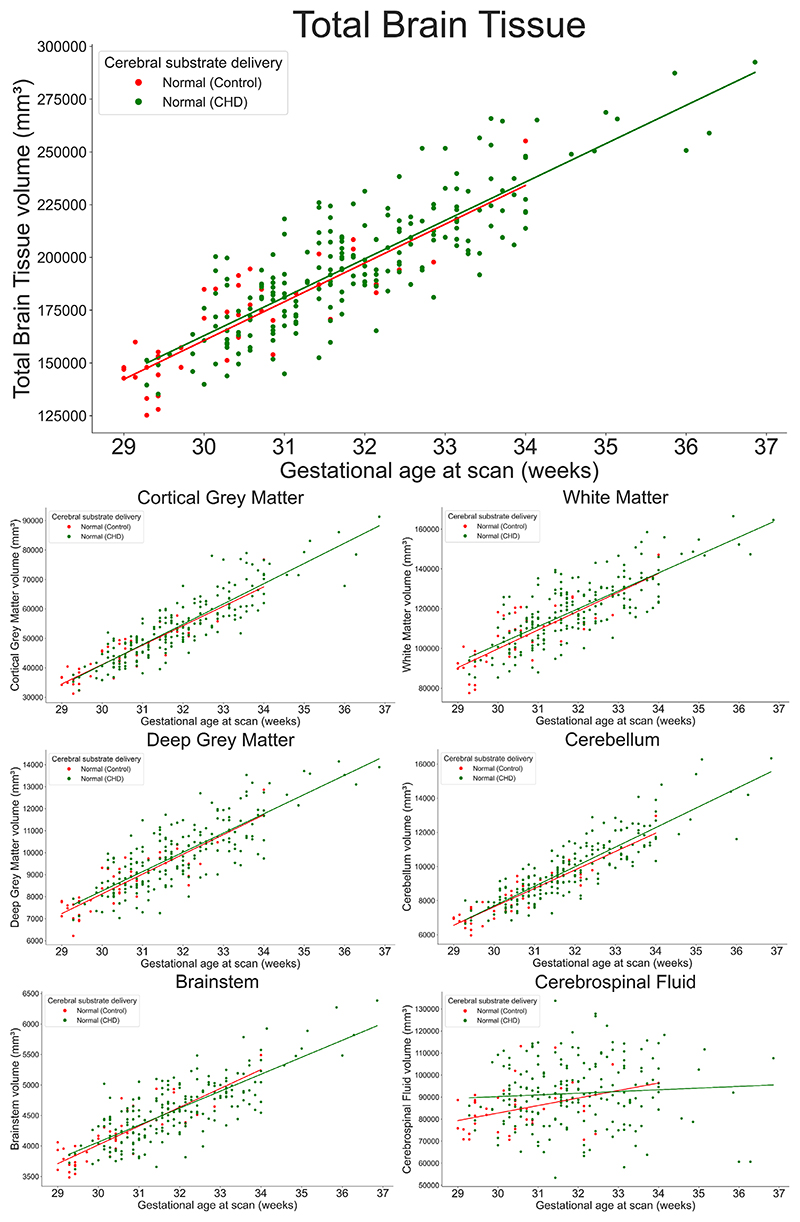
There was a significant difference in GA at scan, but not fetal sex (F = 0.27, P = 0.60) between the control group of typically developing fetuses (Group 0) and fetuses with CHD where cerebral substrate delivery is expected to be normal (Group 1). After accounting for sex and GA at scan, no significant difference was seen in total brain tissue, or any regional brain tissue or CSF volumes between these two groups (All PFDR > 0.12) (Fig. 5).
Figure 5. Scatter plots showing total brain tissue (top panel) and regional brain volumes across gestational age for 45 typically developing fetuses and 210 fetuses with CHD where cerebral substrate delivery is expected to be normal.
After accounting for sex and gestational age at scan, no significant group difference was seen in any brain tissue regions or CSF volumes (All PFDR > 0.12) between these two groups.
Differences in Total and Regional Brain Volumes Between Groups
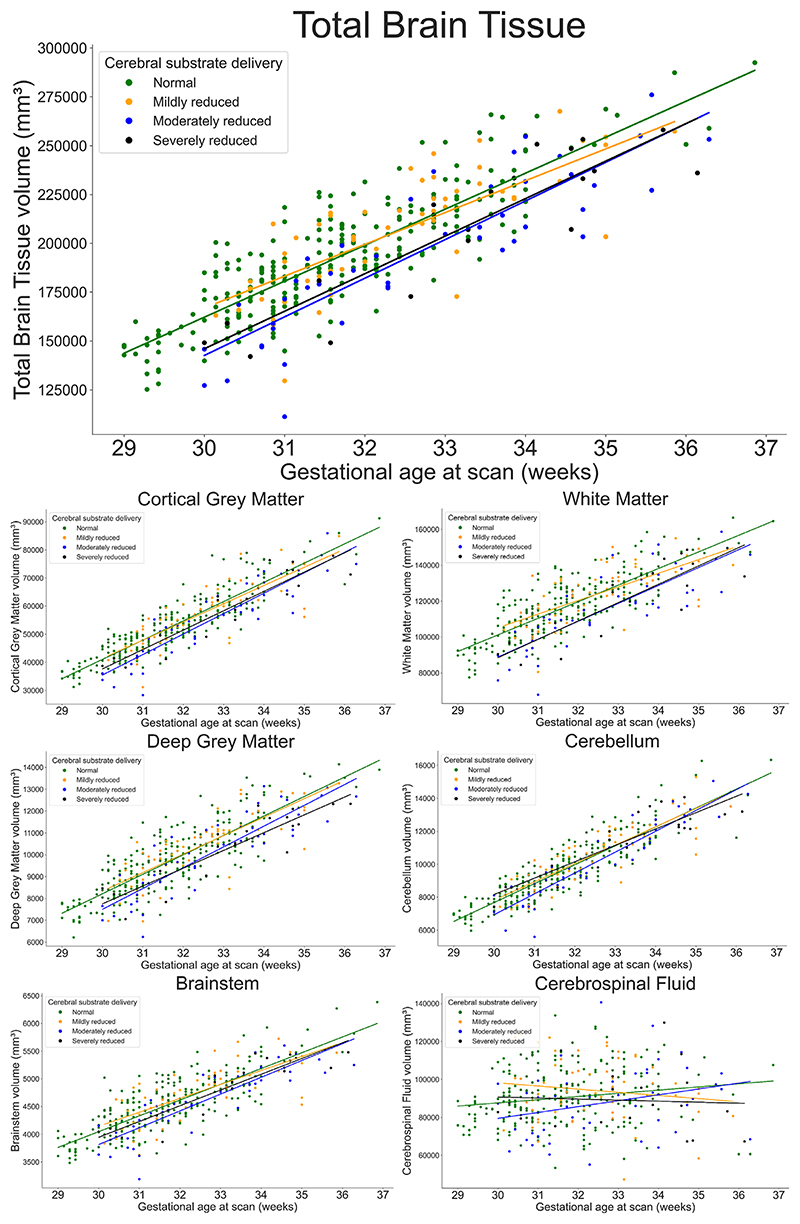
After accounting for sex and gestational age at scan, there was a significant difference in total brain tissue volume and cGM, WM, dGM, cerebellum and brainstem volumes (between all four groups, but not in CSF volumes (PFDR = 0.13) (Fig. 6). These reductions in brain volumes are evident in-utero from 30 weeks gestation and persist until at least 37 weeks gestation.
Figure 6. Scatter plots showing total brain tissue (top panel) and regional brain volumes across gestational age for fetuses grouped according to expected cerebral substrate delivery for 380 fetuses.
There is a significant difference in total brain tissue (PFDR = 4.8 × 10−8), cortical gray matter (top left; PFDR = 8.3 × 10−6), white matter (top right; PFDR = 1.19 × 10−8), deep gray matter (middle left; PFDR = 3.7 × 10−5), cerebellum (middle right; PFDR = 3.44 × 10−2), and brainstem (bottom left; PFDR = 4.0 × 10−3) volumes between groups after accounting for sex and gestational age at scan, but not cerebrospinal fluid volumes (bottom right; PFDR = 0.13).
Post-hoc analyses showed that, after accounting for sex and gestational age at scan, there was a significant difference in total brain tissue volumes between fetuses with normal cerebral substrate delivery and both moderately reduced cerebral substrate delivery () and severely reduced cerebral sub-strate delivery (), but not between fetuses with normal cerebral substrate delivery and mildly reduced cerebral sub-strate delivery (F = 0.43, P = 0.47) or between fetuses with moderately and severely reduced cerebral substrate delivery (F = 0.11, P = 0.53).
Fetal Brain:Body Volume Ratios
Paired fetal brain and body segmentations were available for 256 of the 380 fetuses described above (134 female, 122 male. Substrate content of cerebral blood is expected to be normal [Group 1], N = 183; Substrate content of cerebral blood is expected to be mildly reduced [Group 2], N = 39; Substrate content of cerebral blood is expected to be moderately reduced [Group 3], N = 20; Substrate content of cerebral blood is expected to be severely reduced [Group 4], N = 14). There remained a significant difference in gestational age at scan () but not fetal sex (P = 0.16) between groups.
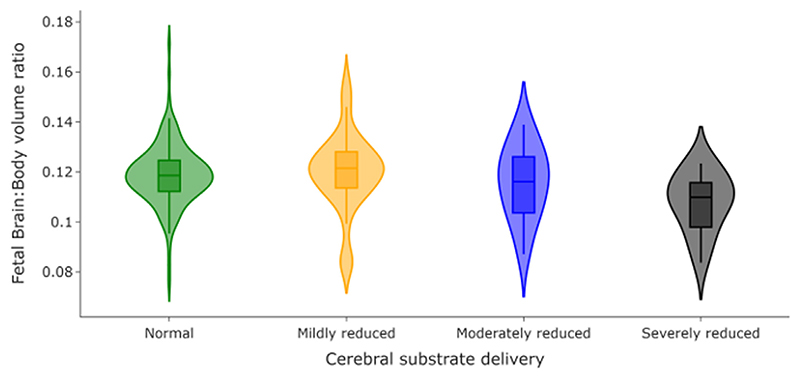
After accounting for sex and gestational age at scan, there was no significant difference in total body volume between groups (F = 1.18, P = 0.32), but there was a significant difference in fetal brain:body volume ratios (). A further subsequent post-hoc analysis showed that there was no significant difference in fetal brain:body ratios between fetuses with normal cerebral substrate delivery and either fetuses with mildly reduced cerebral substrate delivery (F = 0.23, PFDR = 0.47) or with moderately reduced cerebral substrate delivery (F = 0.95, PFDR = 0.392) but there was a significant difference in fetal brain:body ratios between fetuses with normal cerebral substrate delivery and fetuses with severely reduced cerebral substrate delivery () (Fig. 7).
Figure 7. Violin plots comparing brain:body volume ratios for 256 fetuses, grouped according to expected cerebral substrate delivery.
Fetuses with severely reduced cerebral substrate delivery have significantly lower brain:body ratios than those with normal cerebral substrate delivery (PFDR = 0.004).
Discussion
This study utilized automated MR image reconstruction and segmentation techniques to generate sub-millimeter resolution, motion-tolerant fetal brain images with corresponding segmentations of multiple brain tissue regions to show that fetal cardiac physiology resulting in a presumed reduction in expected cerebral substrate delivery is associated with significantly reduced total and regional brain volumes in cortical gray matter, white matter, deep gray matter, the cerebellum and the brainstem.
This work aligns with recent research showing that reduced cerebral oxygenation, as measured using T2-relaxometry, is associated with smaller brain volumes in fetuses with CHD.20 It also has important implications for understanding the impaired neurodevelopmental outcomes seen in individuals with CHD, given the associations identified between fetal brain volumes and subsequent neurodevelopmental outcomes in this population.40
The approach to categorizing CHD differs from previously published studies in that CHD groups were classified according solely to the expected effect of the underlying cardiac defect on the streaming of substrate-rich placental blood to the cerebral circulation, supported by fetal phase-contrast MRI flow data and/or contemporaneous echocardiography where available. It is important to note disordered streaming is one of several proposed mechanisms affecting substrate delivery and/or brain development in fetuses with CHD. For example, as described in Rudolph,41 “in aortic atresia blood flow to the brain is derived from the right ventricle, passing circuitously through the pulmonary artery, ductus arteriosus, and aortic arch. In addition, CoA frequently imposes some obstruction to flow into the aortic arch. In pulmonary atresia, the left ventricle ejects a large stroke volume directly into the ascending aorta.” Therefore, even when the oxygenation of blood reaching the brain is the same in different diagnoses, blood flow patterns, pulsatility and pressures, and the availability of other substrates besides oxygen (i.e., glucose availability) are all likely to play a role.41,42 Recent MRI advances that have allowed in-utero quantification of arterial and venous oxygen saturations in fetal blood vessels,19,23,43 as well as visualization of 4D flow patterns in the heart and major vessels,22 may in future allow for a more detailed analysis of the relationship between the fetoplacental circulation and prenatal brain development in CHD.
We included fetuses with suspected coarctation prenatally who were not shown to have this condition in the neonatal period (“false positive” or CoA (–)). It has been shown previously that this group differs significantly from a healthy control population in terms of the distribution of the fetal circulation, for reasons that remain unclear.26 Arguably they represent a unique group in this cohort as they do not have postnatally confirmed CHD, however, as all cases were referred prenatally, this outcome was not known at the time of fetal imaging, and MRI flow and/or ultrasound data were available to make informed per case estimates regarding cerebral blood oxygen content. As the primary hypothesis of this work is that reduction in cerebral substrate delivery negatively impacts cerebral growth, and data were available to stratify this for the CoA (–) group, this group was retained for the analysis.
These results support existing literature on altered volumetric brain development in fetuses with CHD. They show that total brain volumes are reduced in CHD associated with an expected reduction in cerebral substrate.9,10,44 In line with previous work looking at specific individual brain regions, the results also show that growth is impaired in the cerebellum and brainstem,13 cortical gray matter and white matter.14 Imaging was all performed in the third trimester of pregnancy and thus complements existing work exploring associations between reduced cerebral substrate delivery and brain volumes in earlier gestation.11,17
After accounting for fetal sex and gestational age at scan, there was no significant difference in total brain tissue volumes between fetuses where cerebral substrate delivery is expected to be moderately and severely reduced, as highlighted. While this work suggests that the substrate content of blood reaching the fetal brain is an important factor in determining brain growth, this particular result highlights that there are other important variables that impact cerebral substrate delivery in-utero and should be considered, such as measures of placental function or additional environmental factors.45
For the group with normal cerebral substrate delivery, representing the largest individual group in this study, no significant difference in total or regional brain volumes between these fetuses and a cohort of typically developing fetuses was identified (), supporting the hypothesis that cerebral substrate delivery is a key mediator of fetal brain growth in CHD. Knowing that volumetric brain development is less likely to be affected in specific subtypes of CHD may be helpful for clinicians to identify appropriate groups for neurodevelopmental follow-up and support in postnatal life, as well as focusing future research efforts in this area.
Recent work has also highlighted that reduced fetal brain volumes in CHD are associated with an increased risk of subsequent neonatal ischemic injury13 and neonatal white matter injury.46 Linking reduced fetal brain volumes with expected cerebral substrate delivery in CHD allows speculation as to the underlying biological mechanisms involved in these processes,41,47 and provides further incentives to study fetal brain volumes in fetuses with CHD.
The results also indicate that fetal brain:body volumes are significantly reduced in fetuses with reduced cerebral substrate delivery in-utero, suggesting that these fetuses not only have smaller absolute brain volumes, but smaller brain volumes than would be expected when considering the size of their body. Fetal brain development is a complex, tightly regulated process, but reduced oxygen levels, however they occur, can disrupt these processes, leading to overall diminished brain growth.20,48,49 Potential “brain-sparing” compensatory mechanisms such as cerebrovascular dilatation, increased cardiac output or hematological adaptations can mitigate some of these effects.50 However, these compensatory mechanisms are likely to rely at least in part on normal streaming of oxygen-rich blood from the placenta. It is interesting to note therefore that when this fetal streaming is entirely reversed in-utero, resulting in a severe reduction in the substrate content of cerebral blood, as seen in fetuses with TGA, there is a greater-than-expected reduction in brain growth, whereas body growth remains relatively unaffected. This finding agrees with previous studies showing disproportion in head circumference and birth weight in fetuses and newborns with TGA.10,51 This brain:body volume relationship in TGA fetuses may be an important consideration in interpreting both indexed flow measurements and normal growth trajectories in utero.
Limitations
This study was performed in a single center, using a single magnet, vendor and field strength (1.5 T), which may limit how widely the results can be interpreted.
This work involves making some assumptions about expected cerebral oxygen and substrate delivery based on the underlying cardiac physiology, supported by additional fetal imaging such as fetal echocardiography or MRI-derived cardiac flow data. While there is work that quantifies actual arterial and venous oxygen saturations in major blood vessels in fetuses with and without CHD,22,23,43 it is not possible to know if the assumptions made are true for each individual fetus. In addition, other factors that might be relevant in the altered cerebrovascular hemodynamics that is seen in the fetus with CHD, such as whether the blood reaching the brain is arriving under normal pressures, whether the blood from the brain drains normally or what the likely impact on the altered cardiac physiology is on cerebrovascular resistance were not explored. These considerations, coupled with the innovative methods described above, allowing in-utero quantification of oxygen saturations in fetal blood vessels, represent important directions for future research in this area.
This work focuses on fetuses with suspected or confirmed CHD imaged between 29- and 37-weeks gestational age. This reflects current clinical practice, mainly related to the optimal timing of fetal cardiac MR imaging following initial diagnosis, taking into account referral time, the gestational age at diagnosis and the potential impact of new information on fetal counseling and management that may be required. Linear growth for total and regional brain volumes during these gestational ages was assumed, in-line with other literature.11 Future advanced imaging studies with longitudinal data in pregnancy may help to understand both how and when these changes in brain growth occur.
Finally, all fetuses with some degree of flow reversal in the aortic arch were classified into Group 2 (oxygen content of cerebral blood is expected to be mildly reduced). This group represents a range of conditions where blood flow into the carotid arteries up to the fetal brain cannot be safely assumed to be normal. However, this may be a relatively minor effect in some cases, which may in part explain why no significant difference in total brain tissue volumes between fetuses in Group 1 and Group 2 were identified. Future studies leveraging non-invasive MRI methods as above to quantify cerebral oxygen delivery more accurately, may help to better define the thresholds beyond which fetal brain growth is affected.
Conclusion
Brain development is a complex, multi-faceted process and this study explored just one aspect that is involved in fetal brain growth. A significant association between disruptions to the normal streaming of oxygen and nutrient-rich placental blood to the brain and reduced total and regional brain volumes in-utero in fetuses with CHD was identified. It was also observed that, when the fetal CHD results in a complete reversal in the normal streaming of fetal blood patterns, brain:body volume ratios are also reduced. In CHD subtypes where cerebral substrate delivery is expected to be normal, no significant volume differences were observed when compared to healthy control fetuses, highlighting the role of cerebral substrate delivery in fetal brain growth.
Supplementary Material
Acknowledgments
We would like to thank the families who participated in this research. We also thank our research radiologists, our research radiographers, and our fetal scanning team. In addition, we thank the staff from the Evelina London Children’s Hospital Fetal and Paediatric Cardiology Departments and the Centre for the Developing Brain at King’s College London. This research was funded by the Medical Research Council UK (MR/L011530/1 and MR/V002465/1) and a Wellcome Trust IEH Award (102431). This research was supported by core funding from the Wellcome/EPSRC Centre for Medical Engineering (WT203148/Z/16/Z), and by the National Institute for Health Research (NIHR) Biomedical Research Centre based at Guy’s and St Thomas’ NHS Foundation Trust and King’s College London. The views expressed are those of the authors and not necessarily those of the NHS, the NIHR or the Department of Health.
References
- 1.EUROCAT. European platform on rare disease registration. 2020 [Google Scholar]
- 2.Latal B. Neurodevelopmental outcomes of the child with congenital heart disease. Clin Perinatol. 2016;43:173–185. doi: 10.1016/j.clp.2015.11.012. [DOI] [PubMed] [Google Scholar]
- 3.von Rhein M, Buchmann A, Hagmann C, et al. Brain volumes predict neurodevelopment in adolescents after surgery for congenital heart disease. Brain J Neurol. 2014;137(Pt 1):268–276. doi: 10.1093/brain/awt322. [DOI] [PubMed] [Google Scholar]
- 4.Cromb D, Bonthrone AF, Maggioni A, et al. Individual assessment of perioperative brain growth trajectories in infants with congenital heart disease: Correlation with clinical and surgical risk factors. J Am Heart Assoc. 2023;12:e8599. doi: 10.1161/JAHA.122.028565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.von Rhein M, Buchmann A, Hagmann C, et al. Severe congenital heart defects are associated with global reduction of neonatal brain volumes. J Pediatr. 2015;167:1259–1263.:e1. doi: 10.1016/j.jpeds.2015.07.006. [DOI] [PubMed] [Google Scholar]
- 6.Kelly CJ, Arulkumaran S, Tristão Pereira C, et al. Neuroimaging findings in newborns with congenital heart disease prior to surgery: An observational study. Arch Dis Child. 2019;104:1042–1048. doi: 10.1136/archdischild-2018-314822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ng IHX, Bonthrone AF, Kelly CJ, et al. Investigating altered brain development in infants with congenital heart disease using tensor-based morphometry. Sci Rep. 2020;10:14909. doi: 10.1038/s41598-020-72009-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bonthrone AF, Dimitrova R, Chew A, et al. Individualized brain development and cognitive outcome in infants with congenital heart disease. Brain Commun. 2021;3:3. doi: 10.1093/braincomms/fcab046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Limperopoulos C, Tworetzky W, McElhinney DB, et al. Brain volume and metabolism in fetuses with congenital heart disease. Circulation. 2010;121:26–33. doi: 10.1161/CIRCULATIONAHA.109.865568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Jørgensen DES, Tabor A, Rode L, et al. Longitudinal brain and body growth in fetuses with and without transposition of the great arteries. Circulation. 2018;138:1368–1370. doi: 10.1161/CIRCULATIONAHA.118.034467. [DOI] [PubMed] [Google Scholar]
- 11.Rollins CK, Ortinau CM, Stopp C, et al. Regional brain growth trajectories in fetuses with congenital heart disease. Ann Neurol. 2021;89:143–157. doi: 10.1002/ana.25940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wu Y, Lu Y-C, Kapse K, et al. In utero MRI identifies impaired second trimester subplate growth in fetuses with congenital heart disease. Cereb Cortex. 2021;32:2858–2867. doi: 10.1093/cercor/bhab386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Dovjak GO, Hausmaninger G, Zalewski T, et al. Brainstem and cerebellar volumes at magnetic resonance imaging are smaller in fetuses with congenital heart disease. Am J Obstet Gynecol. 2022;227:282e1–282e15. doi: 10.1016/j.ajog.2022.03.030. [DOI] [PubMed] [Google Scholar]
- 14.Clouchoux C, du Plessis AJ, Bouyssi-Kobar M, et al. Delayed cortical development in fetuses with complex congenital heart disease. Cereb Cortex. 2013;23:2932–2943. doi: 10.1093/cercor/bhs281. [DOI] [PubMed] [Google Scholar]
- 15.Lee F-T, Seed M, Sun L, Marini D. Fetal brain issues in congenital heart disease. Transl Pediatr. 2021;10:2182–2196. doi: 10.21037/tp-20-224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Paladini D, Finarelli A, Donarini G, et al. Frontal lobe growth is impaired in fetuses with congenital heart disease. Ultrasound Obstet Gynecol. 2021;57:776–782. doi: 10.1002/uog.22127. [DOI] [PubMed] [Google Scholar]
- 17.Ren J-Y, Zhu M, Dong S-Z. Three-dimensional volumetric magnetic resonance imaging detects early alterations of the brain growth in fetuses with congenital heart disease. J Magn Reson Imaging. 2021;54:263–272. doi: 10.1002/jmri.27526. [DOI] [PubMed] [Google Scholar]
- 18.Sun L, Macgowan CK, Sled JG, et al. Reduced fetal cerebral oxygen consumption is associated with smaller brain size in fetuses with congenital heart disease. Circulation. 2015;131:1313–1323. doi: 10.1161/CIRCULATIONAHA.114.013051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Sun L, van Amerom JFP, Marini D, et al. MRI characterization of hemodynamic patterns of human fetuses with cyanotic congenital heart disease. Ultrasound Obstet Gynecol. 2021;58:824–836. doi: 10.1002/uog.23707. [DOI] [PubMed] [Google Scholar]
- 20.Peyvandi S, Xu D, Wang Y, et al. Fetal cerebral oxygenation is impaired in congenital heart disease and shows variable response to maternal Hyperoxia. J Am Heart Assoc. 2021;10:e018777. doi: 10.1161/JAHA.120.018777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kiserud T. Physiology of the fetal circulation. Semin Fetal Neonatal Med. 2005;10:493–503. doi: 10.1016/j.siny.2005.08.007. [DOI] [PubMed] [Google Scholar]
- 22.Roberts TA, van Amerom JFP, Uus A, et al. Fetal whole heart blood flow imaging using 4D cine MRI. Nat Commun. 2020;11:4992. doi: 10.1038/s41467-020-18790-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Saini BS, Darby JRT, Portnoy S, et al. Normal human and sheep fetal vessel oxygen saturations by T2 magnetic resonance imaging. J Physiol. 2020;598:3259–3281. doi: 10.1113/JP279725. [DOI] [PubMed] [Google Scholar]
- 24.Rudolph AM. Congenital cardiovascular malformations and the fetal circulation. Arch Dis Child Fetal Neonatal Ed. 2010;95:F132–F136. doi: 10.1136/adc.2007.128777. [DOI] [PubMed] [Google Scholar]
- 25.Vigneswaran T, Zidere V. In: Fetal cardiology a practical approach to diagnosis and management. Simpson J, Zidere V, Miller OI, editors. Cham: Springer International Publishing; 2018. Aortic arch abnormalities; pp. 139–151. [Google Scholar]
- 26.Lloyd DFA, van Poppel MPM, Pushparajah K, et al. Analysis of 3-dimensional arch anatomy, vascular flow, and postnatal outcome in cases of suspected coarctation of the aorta using fetal cardiac magnetic resonance imaging. Circ Cardiovasc Imaging. 2021;14:e012411. doi: 10.1161/CIRCIMAGING.121.012411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Uus A, Zhang T, Jackson LH, et al. Deformable slice-to-volume registration for motion correction of fetal body and placenta MRI. IEEE Trans Med Imaging. 2020;39:2750–2759. doi: 10.1109/TMI.2020.2974844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Ortinau CM, Rollins CK, Gholipour A, et al. Early-emerging sulcal patterns are atypical in fetuses with congenital heart disease. Cereb Cortex. 2019;29:3605–3616. doi: 10.1093/cercor/bhy235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Donofrio MT, Moon-Grady AJ, Hornberger LK, et al. Diagnosis and treatment of fetal cardiac disease: A scientific statement from the American Heart Association. Circulation. 2014;129:2183–2242. doi: 10.1161/01.cir.0000437597.44550.5d. [DOI] [PubMed] [Google Scholar]
- 30.Ewer AK, Middleton LJ, Furmston AT, et al. Pulse oximetry screening for congenital heart defects in newborn infants (PulseOx): A test accuracy study. Lancet Lond Engl. 2011;378:785–794. doi: 10.1016/S0140-6736(11)60753-8. [DOI] [PubMed] [Google Scholar]
- 31.Schellen C, Ernst S, Gruber GM, et al. Fetal MRI detects early alterations of brain development in tetralogy of Fallot. Am J Obstet Gynecol. 2015;213:392e1–392e7. doi: 10.1016/j.ajog.2015.05.046. [DOI] [PubMed] [Google Scholar]
- 32.Uus AU, Grigorescu I, van Poppel MPM, et al. Automated 3D reconstruction of the fetal thorax in the standard atlas space from motion-corrupted MRI stacks for 21-36 weeks GA range. Med Image Anal. 2022;80:102484. doi: 10.1016/j.media.2022.102484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kuklisova-Murgasova M, Quaghebeur G, Rutherford MA, Hajnal JV, Schnabel JA. Reconstruction of fetal brain MRI with intensity matching and complete outlier removal. Med Image Anal. 2012;16:1550–1564. doi: 10.1016/j.media.2012.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Uus A, Grigorescu I, van Poppel M, et al. 3D UNet with GAN discriminator for robust localisation of the fetal brain and trunk in MRI with partial coverage of the fetal body. Bioengineering. 2021 doi: 10.1101/2021.06.23.449574. [DOI] [Google Scholar]
- 35.Uus AU, Egloff Collado A, Roberts TA, Hajnal JV, Rutherford MA, Deprez M. Retrospective motion correction in foetal MRI for clinical applications: Existing methods, applications and integration into clinical practice. Br J Radiol. 2022;96:20220071. doi: 10.1259/bjr.20220071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Uus AU, Kyriakopoulou V, Makropoulos A, et al. BOUNTI: Brain vOlumetry and aUtomated parcellatioN for 3D feTal MRI. bioRxiv. 2023 doi: 10.1101/2023.04.18.537347. [DOI] [Google Scholar]
- 37.Çiçek Ö, Abdulkadir A, Lienkamp SS, Brox T, Ronneberger O. In: Medical Image Computing and Computer-Assisted Intervention − MICCAI 2016. Ourselin S, Joskowicz L, Sabuncu MR, Unal G, Wells W, editors. Cham: Springer International Publishing; 2016. 3D U-net: Learning dense volumetric segmentation from sparse annotation; pp. 424–432. [Google Scholar]
- 38.Yushkevich PA, Piven J, Hazlett HC, et al. User-guided 3D active contour segmentation of anatomical structures: Significantly improved efficiency and reliability. Neuroimage. 2006;31:1116–1128. doi: 10.1016/j.neuroimage.2006.01.015. [DOI] [PubMed] [Google Scholar]
- 39.Jansz MS, Seed M, van Amerom JFP, et al. Metric optimized gating for fetal cardiac MRI. Magn Reson Med. 2010;64:1304–1314. doi: 10.1002/mrm.22542. [DOI] [PubMed] [Google Scholar]
- 40.Sadhwani A, Wypij D, Rofeberg V, et al. Fetal brain volume predicts neurodevelopment in congenital heart disease. Circulation. 2022;145:1108–1119. doi: 10.1161/CIRCULATIONAHA.121.056305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Rudolph AM. Impaired cerebral development in fetuses with congenital cardiovascular malformations: Is it the result of inadequate glucose supply? Pediatr Res. 2016;80:172–177. doi: 10.1038/pr.2016.65. [DOI] [PubMed] [Google Scholar]
- 42.Yamamoto Y, Khoo NS, Brooks PA, Savard W, Hirose A, Hornberger LK. Severe left heart obstruction with retrograde arch flow influences fetal cerebral and placental blood flow. Ultrasound Obstet Gynecol. 2013;42:294–299. doi: 10.1002/uog.12448. [DOI] [PubMed] [Google Scholar]
- 43.Peyvandi S, Donofrio MT. Circulatory changes and cerebral blood flow and oxygenation during transition in newborns with congenital heart disease. Semin Pediatr Neurol. 2018;28:38–47. doi: 10.1016/j.spen.2018.05.005. [DOI] [PubMed] [Google Scholar]
- 44.Hansen T, Henriksen TB, Bach CC, Matthiesen NB. Congenital heart defects and measures of prenatal brain growth: A systematic review. Pediatr Neurol. 2017;72:7–18.:e1. doi: 10.1016/j.pediatrneurol.2017.03.014. [DOI] [PubMed] [Google Scholar]
- 45.Peyvandi S, Rollins C. Fetal brain development in congenital heart disease. Can J Cardiol. 2023;39:115–122. doi: 10.1016/j.cjca.2022.09.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Peyvandi S, Lim JM, Marini D, et al. Fetal brain growth and risk of postnatal white matter injury in critical congenital heart disease. J Thorac Cardiovasc Surg. 2021;162:1007–1014. doi: 10.1016/j.jtcvs.2020.09.096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Vannucci RC, Vannucci SJ. Glucose metabolism in the developing brain. Semin Perinatol. 2000;24:107–115. doi: 10.1053/sp.2000.6361. [DOI] [PubMed] [Google Scholar]
- 48.Seed M, Limperopoulos C. In utero brain growth matters for fetuses with congenital heart disease. Circulation. 2022;145:1120–1122. doi: 10.1161/CIRCULATIONAHA.122.058683. [DOI] [PubMed] [Google Scholar]
- 49.Ortinau CM, Shimony JS. The congenital heart disease brain: Prenatal considerations for perioperative Neurocritical care. Pediatr Neurol. 2020;108:23–30. doi: 10.1016/j.pediatrneurol.2020.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Ramirez Zegarra R, Dall’Asta A, Ghi T. Mechanisms of fetal adaptation to chronic hypoxia following placental insufficiency: A review. Fetal Diagn Ther. 2022;49:279–292. doi: 10.1159/000525717. [DOI] [PubMed] [Google Scholar]
- 51.Matthiesen NB, Henriksen TB, Gaynor JW, et al. Congenital heart defects and indices of fetal cerebral growth in a Nationwide Cohort of 924 422 Liveborn Infants. Circulation. 2016;133:566–575. doi: 10.1161/CIRCULATIONAHA.115.019089. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.