Abstract
The outbreak of the deadly virus (novel coronavirus or Severe Acute Respiratory Syndrome Coronavirus-2) that emerged in December 2019, remained a controversial subject of intense speculations regarding its origin, became a worldwide health problem resulting in serious coronavirus disease 2019 (acronym COVID-19). The concern regarding this new viral strain “Severe Acute Respiratory Syndrome Coronavirus-2” (acronym SARS-CoV-2) and diseases it causes (COVID-19) is well deserved at all levels. The incidence of COVID-19 infection and infectious patients are increasing at a high rate. Coronaviruses (CoVs), enclosed positive-sense RNA viruses, are distinguished by club-like spikes extending from their surface, an exceptionally large genome of RNA, and a special mechanism for replication. Coronaviruses are associated with a broad variety of human and other animal diseases spanning from enteritis in cattle and pigs and upper chicken respiratory disease to extremely lethal human respiratory infections. With World Health Organization (WHO) declaring COVID-19 as pandemic, we deemed it necessary to provide a detailed review of coronaviruses discussing their history, current situation, coronavirus classification, pathogenesis, structure, mode of action, diagnosis and treatment, the effect of environmental factors, risk reduction and guidelines to understand the virus and develop ways to control it.
Keywords: Coronaviruses, RNA viruses, SARS-CoV-2, COVID-19, Biocidal agents
1. 2019 novel coronavirus
A novel coronavirus (termed 2019-nCoV) was reported in December 2019 from genomic screening of clinical samples from patients with viral pneumonia in Wuhan, China. The primary viral pneumonia patients were found to be epidemiologically linked to the Huanan seafood market in Wuhan City, Hubei Province, China, where other non-aquatic animals, such as bats, pangolins and rabbits, were on sale before the outbreak [[1], [2], [3]]. Through the use of next-generation sequencing, a new, human-infecting coronavirus, provisionally called 2019 novel coronavirus (2019-nCoV), was identified. Subsequently, on February 11, 2020, outbreak or disease previously known as “novel coronavirus” or 2019-nCoV was officially renamed as C-O-V-I-D-19 or COVID-19 and causal virus was named as “Severe acute respiratory syndrome-related coronavirus 2” or SARS-CoV-2 [4].
2. Historical perspective
The importance of the class of coronaviruses in both medical and economic terms has become much more palpable since the First International Congress was organized in Germany in 1980. The incorporation of modern molecular biology and immunology has aided in providing fresh acumens into explaining the nature of the viruses and the pathogenesis of the diseases they cause [5]. Since the very earliest stages of Coronavirology, the virus invited the interests of veterinary, medical, as well as basic scientists and it revolved around the clear virulence that these viruses had for the gastrointestinal tract, respiratory system, and nervous system. Baudette and Hudson probably made the first scientific annotations in 1933, when they described chickens “gasping disease” and then communicated the disease to embryos [6]. Gasping disease has been seen as an appallingly deadly respiratory illness. This virus was subsequently recognized as an infectious bronchitis virus (IBV) that became the prototype of this family of viruses [7]. Isolation of the mouse hepatitis virus by Gledhill and Andrewes managed to draw the field for development [8]. They stated in their paper that the infectious agent they named as mouse hepatitis virus (MHV) would not have produced such extensive devastation in the event of gasping death. And thereafter important concerns were posed with the discovery of MHV. For example, combined infection with an otherwise harmless murine protozoan, Eperythrozoon coccids, lead to a lethal hepatitis. This resulted in the introduction of the concept that activation of the latent virus by different forms of stress is critical in disease pathogenesis [8,9]. The discovery that the human respiratory viruses, mouse hepatitis viruses as well as the prototype infectious chicken bronchitis virus had analogous manifestations resulted in a classic 1968 publication classifying such viruses as coronavirus [8]. The paper published by Almedia, Berry, Cunningham, Hamre, Hofstad, Mallucci, McIntosh and Tyrrell used the characterization of Tyrrell in that the viruses exhibited “a characteristic fringe of broad, distinctive, petal-shaped spikes resembling a crown like the corona spinarum in religious art” [9]. In 1975, the International Committee on Virus Taxonomy named a new family with one genus coronavirus, Coronaviridae. Additional viruses including canine coronavirus, feline peritonitis virus, human enteric coronavirus, and transmissible pig gastroenteritis virus (TGEV), neonatal calf diarrhoea coronavirus (BCV) and rat coronavirus (RCV) have been identified [10]. Mahy has been the author of a review of the First International Congress on Coronaviruses in 1980, “Coronavirus come of age” [11]. This symposium takes the field of animal virology one step further and brings the state of understanding of the coronavirus molecular biology to standards that will be followed by those researching the pathogenesis of both human and animal virus diseases [5].
3. Current situation
According to WHO situation report-59 (April 17th, 2020), COVID-19 is affecting 167 countries around the globe and 1 international conveyance [12]. The total number of confirmed cases of SARS-CoV-2, had gone up to more than 6,057,853 worldwide with 371,166 deaths. There are currently 3,087,529 active cases out of total confirmed cases worldwide with 3,034,131 (98%) in mild condition and 2% (53,398 cases) in critical condition. Out of the total closed cases, 2,924,078 (89%) have recovered/discharged with 377,888 (18%) deaths so far [13,14]. There have also been records of infections in medical workers and family clusters, and evidence of human-to-human transmission [4]. A few of the patients diagnosed had high fever and others had dyspnoea, with radiographs of the chest showing intrusive lesions in both lungs [[14], [15], [16]]. The average evolutionary rate for coronaviruses as a typical RNA virus is roughly 10–4 nucleotide substitutions per site per year [17], with mutations occurring during each replication cycle. The average evolutionary rate for SARS-CoV-2 as a typical RNA virus is roughly 8 × 10−4 nucleotide substitutions per site per year, with one mutation occurring in two weeks [18,19]. This occurrence is similar to that of two viruses of the same family, SARS and MERS [20].
The symptoms or signs of COVID-19 at the time of onset and throughout disease include: fever or chills, coughing, breathlessness, fatigue, muscle ache, headache, nausea or vomiting, sore throat, congestion or running nose, diarrhoea, loss of smell (anosmia) and loss of taste (ageusia) [21,22]. The severity of COVID-19 symptoms ranges from mild to critical: Mild (mild symptoms to mild pneumonia in 81%), severe (dyspnoea, hypoxia and 50% lung involvement in 14%) and critical (multi-organ failure, respiratory failure or shock in 5%). The fatality rate among critical cases was around 49% [23]. The fatality rate in COVID-19 was below and around 2% relative to the previous two epidemics (SARS and MERS), although only fewer than 15% of patients were requiring hospital services. Nevertheless, the SARS and MERS fatality levels were 10% and 34% respectively [24]. The overall case fatality rate for COVID-19 [25] was reported to be 2.3% from a study conducted in China and some studies showed that case fatality rate in Beijing, China was 0.9% [26]. In another study, Jung and colleagues have reported that COVID-19 has a 5.3% to 8.4% chance for fatality [27]. The case fatality rate in China was highest in age group of ≥80 (14.8%), 70–79 years (8.0%), 60–69 years (3.6%), 50–59 years (1.3%), 40–49 years (0.4%) and <40 years (0.2%) [23,28]. According to U.S Epidemiological data, the case fatality rate was highest in age group of ≥85 years (10%–27%), 65–84 years (3%–11%), 55–64 years (1%–3%) and <55 years (<1%) [29]. The median time of dyspnoea from onset of diseases or symptoms was between 5 and 8 days, the average time for acute respiratory distress (ARDS) from disease onset was 8 to 12 days and the mean time the patients received ICU from the onset of disease or the symptoms ranged between 10 and 12 days [30,31]. In China, the average case fatality of patients without reported underlying medical problems was 0.9%. The fatality was greater for comorbidity: 10.5% for cardiovascular diseases, 7.3% for diabetes, and about 6% for chronic respiratory disorder or cancer [22,31]. In Chinese mainland, the median time from onset of symptoms to recovery rate in case of mild or severe patients ranges from 2 and 3–6 weeks. Moreover, the time from the onset of symptoms and developing severe hypoxia is one week. The studies conducted outside Chinese mainland, the time from the onset of symptoms to recovery was 22.2 days at 95% confidence interval 18–83. Furthermore, the time from the onset of symptoms to death varies from 20.2 days (95% confidence interval 15.1–29.9) to 22.3 days (95% confidence interval 18–82) [27].
One effective tactics in the battle against COVID-19 is to keep the public informed about the progress through media, maps and graphics. Gao et al. [32] used cartograms for visualizing COVID-19 expansion and spread. Their study included 31 Chinese provinces (except Hong Kong, Macau, and Taiwan) for the same source of data, namely provincial health commissions. After employing an area cartogram, they identified three main categories: contiguous, non-contiguous and circular. After testing all the three major types, circular was found to be the most appropriate in their case. Circular cartograms were coloured using proper scientific scheme with some minor constraints: (a) adjoining circles do not have the same color, and (b) each province's color remained constant between different cartograms. Each cartogram has two important characteristics: a circle in each cartograms represents the province and its size is directly proportional to the number of COVID-19 cases reported from that province. Gao et al. [32] concluded from their study that the COVID-19 probably originated in Hubei and then spread to the other provinces. Another important study to map the changing internet attention to the spread of COVID-19 in China was carried out by Zhang et al. [33]. The investigators used cartograms to visualize the spread of COVID-19 and people's internet attention in China based on diffusion method [34], effective in spatial data representation [35]. Zhang et al. [33] concluded from their study that the number of searches and COVID-19 cases showed a different pattern, probably because people not only refer to internet but use different social media platforms to share the disease information. Authors further argued that the number of searches decreased drastically in most provinces leading to reduced anxiety, provinces paid much attention towards the COVID-19 after declared public health emergency of international concern (PHEIC) indicating WHO as a vital functionary in fight against COVID-19 and number of searches from Hubei province wasn't outstanding from other provinces.
4. Coronavirus classification
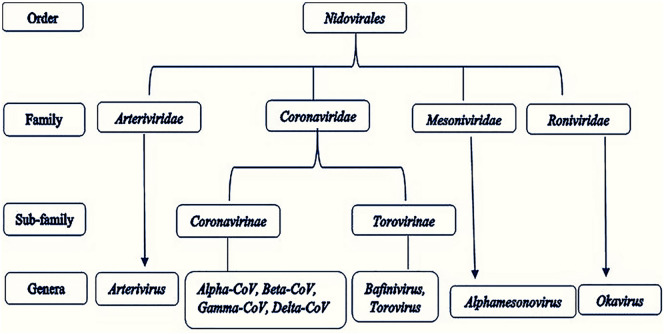
Coronaviruses (CoVs) are the Nidovirales order's largest group of viruses, including Coronaviridae, Arteriviridae, Roniviridae, and Mesoniviridae. The Coronavirinae forms one of two subfamilies in the family Coronaviridae, the other one being the Torovirinae. Coronavirinae are divided into four categories: Alpha, Beta, Gamma, and Delta coronaviruses [36] (Fig. 1 ). Initially, the viruses were divided into those groups based on serology, but are now distinguished by phylogenetic clustering. Both viruses are classified, in the class Nidovirales, non-segmented positive-sense RNA viruses. They all contain significant genomes for RNA viruses, with Coronavirinae having the largest known genomes for RNA, containing approximately 30 kilobase (kb) genomes. The main differences among the Nidovirus families are the number, shape, and scale of the structural proteins. Such differences cause significant changes in the structure and morphology of nucleocapsids and virions [[37], [38], [39], [40]].
Fig. 1.
The taxonomy of order Nidovirales.
5. Pathogenesis
Until the SARS-CoV outbreak, coronaviruses were only thought to cause mild, self-limiting respiratory infections in humans. Four of the known human coronaviruses are α-coronaviruses (HCoV-229E and HCoV-NL63), and the four others are β-coronaviruses (HCoV-OC43 and HCoV-HKU1). One important feature of these viruses is the variability in susceptibility to genetic variations. HCoV-229E isolates have only marginal sequence divergence [41] whereas HCoV-OC43 isolates from the same region but are isolated in different years display significant genetic variability [42]. That explains the failure of HCoV-229E to cross the species boundary to infect mice while HCoV-OC43 and the strongly related bovine coronavirus (BCoV) can infect mice and other ruminants. SARS-CoV, a 2b β-coronavirus species, was identified as the causative agent for the 2002–2003 outbreak of Severe Acute Respiratory Syndrome (SARS) in China's Guangdong Province. It is generally accepted that SARS-CoV originated in bats, because a large number of Chinese horseshoe bats exhibit sequences of SARS-related CoVs and provide serological evidence of an earlier CoV infection [43,44] SARS-CoV primarily infects the epithelial cells within the lung. The virus can invade macrophages and dendritic cells but only progresses to an abortive infection [45,46]. Nonetheless, infection of these cell types may be necessary for the activation of pro-inflammatory cytokines that can lead to disease [47]. The exact mechanism of lung damage and the cause of serious illness remains undetermined in humans. Globally, based on hospitalization data, the incubation period for SARS-CoV-2 ranges from 5.1 to 14 days, and about 80% of patients with mild or asymptomatic, 15% severe (needed oxygen) and 5% critical (needed ventilation) [14,48]. Cough, fever and fatigue are among the most common symptoms [14]. The S protein and the SARS-CoV-2 N protein during infection are the two most immunogenic and predominantly expressed proteins [49].
6. Structure
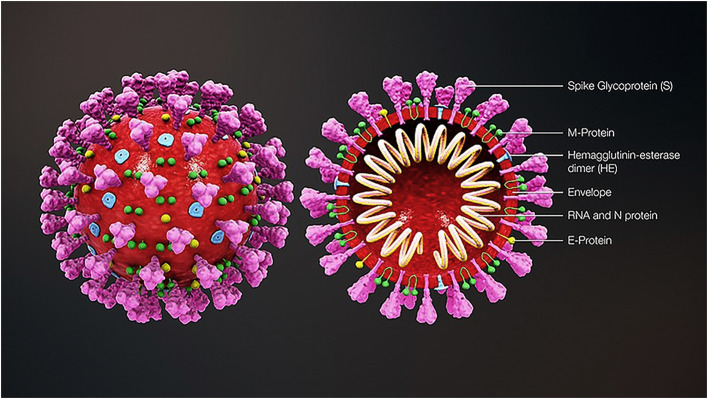
Coronavirus virions are spherical with diameters of roughly 125 nm, as described by cryo-electron tomography and cryo-electron microscopy studies [50,51]. The very well-known feature of coronaviruses is spike club-shaped projections emerging from the surface of the virions. These spikes are a defining characteristic of the virion and give them the look of a solar corona by inspiring the name coronaviruses. Nucleocapsid lies inside the virion envelope. Coronaviruses possess helically symmetric nucleocapsids, which are uncommon in positive-sense RNA viruses, but are much more common in negative-sense RNA viruses. Coronavirus virus particles contain four primary structural proteins. These are the spike (S), membrane (M), envelope (E), and nucleocapsid (N) proteins all coded inside the viral genome's 3′ end. The S protein (available at 150 kDa), controls the signal sequence of the N-terminals to reach the ER. The M protein is by far the most abundant structural protein present in the virion. It is an integral part of its shape and is a tiny protein with 3 transmembrane domains (25 to 30 kDa) [52]. Recent studies suggest that M protein exists as a dimer in the virion, and may adopt two different configurations to promote membrane curvature as well as nucleocapsid binding [53]. In small amounts within the virion, the E protein (first 8 to 12 kDa) is contained. E coronavirus protein is widely divergent but usually have common structural design [54]. E protein's membrane topology is not completely known but most evidence says it's a transmembrane protein. This allows the virus to be installed and published but has other benefits as well. For example, E protein is not required for viral replication in the operation of ion channels in SARS-CoV but is required for pathogenesis [55]. The single protein contained in nucleocapsid is the N protein. It consists of two separate domains, an N-terminal domain (NTD) and a C-terminal domain (CTD), both capable of binding RNA in vitro but each domain uses multiple modes to bind RNA. A fifth structural protein, hemagglutinin-esterase (HE), is found in a subset of β-coronaviruses. The protein acts as a hemagglutinin that binds sialic acids to surface glycoproteins, and has an acetyl-esterase activity [56]. These behaviours are suggested to enhance the entry of S protein-mediated cells and the spread of the virus through the mucosa [57].
A most recent research to examine SARS-CoV-2's structural basis showed that its viral 3-chymotrypsin-like cysteine protease (3CLpro) is retained in SARS-CoV-2 (Fig. 2 ) [58]. Furthermore, the investigators find that the SARS-CoV-2 is quite similar to bat-SARS such as coronavirus 3CLpro sharing 99.02% of sequence identity, with key differences from other beta-coronaviruses [59]. SARS-CoV-2 3CLpro also holds a certain sequence identity with SARS-CoV (96.08%), MERS-CoV (87.00%), Human-CoV (90.00%), and Bovine-CoV (90.00%). SARS-CoV-2 3CLpro’s physico-chemical characteristics showed that it contains 306 amino acid long polypeptides with a molecular weight of 33,796.64 Da and a Gravy score of −0.019 [[58], [59], [60], [61]]. Protein has also been described as stable, hydrophilic and able to form hydrogen bonds based on physico-chemical analysis [58]. BLAST analysis found 12 point mutations between SARS-CoV and SARS-CoV-2 3CLpro enzymes, except for Leu being replaced by Ala in position 286. Such mutations are likely to affect 3CLpro’s structure and function. SARS-CoV-2 3CLpro’s 3D various morphological analyses showed that the structural design is compatible with the SARS-CoV crystal structure, with root mean square deviation between homology model and prototype approximately 0.629 Å. The binding site for substrates is situated in a cleft between domain I and domain II in SARS-CoV-2 3CLpro. A residue loop 184 to 199 crosses the domains N-terminal and Domain III which is also 135 called the domain C-terminal and includes a five-helix anti-parallel cluster [58].
Fig. 2.
3D medical animation of coronavirus structure. Source: https://commons.wikimedia.org/wiki/File:3D_medical_animation_coronavirus_structure.jpg
7. Mode of action
The virions link to the host cell is triggered by interactions between the S protein and its receiver. Spots of receptor binding domains (RBD) inside the S1 region of a coronavirus S protein depend largely on the virus, with several having the RBD at the N-terminus of S1 (MHV) whereas others (SARS-CoV) have the RBD at the C-terminus of S1 [62,63]. The S-protein / receptor interaction is the key determinant for a coronavirus to invade a host species and also controls the tissue tropism of the virus. Some coronaviruses use peptidases as their cell receptor. Most α-coronaviruses use aminopeptidase N (APN) as their receptor, SARS-CoV and HCoV-NL63 use angiotensin-converting enzyme 2 (ACE2) as the receptor, MHV comes in through CEACAM1, and the newly mentioned MERS-CoV binds to dipeptidyl-peptidase 4 (DPP4) to infect human cells [64]. Upon binding the receptor, the virus must have access to the host cell's cytosol. Acid-dependent proteolytic cleavage of the S protein is usually achieved by a cathepsin, TMPRRS2 or other proteases, accompanied by viral and cellular membrane fusion. S protein cleavage occurs at two sites inside the S2 fragment of the protein, with the very first important cleavage for separating the RBD and fusion domains of the S protein [65] and the second important for revealing the fusion peptide (cleavage at S2′). Fusion usually occurs within acidified endosomes, however some coronaviruses, including MHV, can fuse in at the plasma membrane. Cleavage at S2′ shows a fusion peptide that incorporates into the membrane, followed by two heptad repeats in S2 that produce an antiparallel array of six helixes [10]. The composition of this kit allows for the mixing of viral and cellular membranes, leading in a fusion and ultimately release into the viral genome cytoplasm. The next step in the coronavirus lifecycle is the gene replicase translation from of the genomic virion RNA. The replicase gene encodes two large ORFS, rep1a and rep1b, encoding two co-terminals of polyproteins, pp1a and pp1ab. The virus uses a slippery sequence (5′-UUUAAAC-3′) and an RNA pseudoknot to produce both polyproteins, allowing ribosomal frameshifting from the rep1a read frame into the rep1b ORF. The nsps 1–11 and 1–16 contain pp1a and pp1ab polyproteins, respectively. In pp1ab, nsp11 from pp1a becomes nsp12 following extension of pp1a into pp1b. There is no comparable nsp1 in π-coronaviruses however. Then these polyproteins are clogged into the individual nsps [66]. The nsps integrate into the Replicase-Transcriptase Complex (RTC) to create an environment that is conducive to RNA synthesis, and are liable for the replication and transcription of subgenomic RNAs. Synthesis of Viral RNA requires the transcription and assembly of complexes connected to viral replicas. The Viral RNA synthesis generates both genomic and subgenomic RNAs. Subgenomic RNAs serve as mRNAs for the structural and accessory genes residing downstream of replicas of polyproteins. Both genomic and subgenomic RNAs are produced in the negative-strand using intermediates. Many cis-acting sequences are essential for a replication of viral RNA. Within the genome's 5′ UTR, there are seven stem-loop structures which can stretch into the replicase 1a gene [[67], [68], [69], [70]].
The 3′ UTR comprises a bulged stem-loop, a pseudoknot, and hypervariable area [[71], [72], [73], [74]]. Ironically, at the 3′ end, the stem-loop and the pseudoknot overlap, and therefore cannot develop at the same time [[72], [73], [74], [75]]. Thus, these structures are suggested to handle alternate stages of RNA synthesis, although it is still unknown exactly what stages are regulated and their exact mechanisms involved. The novelistic feature of coronavirus replication is how the leader and body TRS segments fused during subgenomic RNA evolution. This was originally estimated to result during positive-strand synthesis and is now reported to cause during discontinuous extension of the negative-strand RNA [76]. The current model suggests that the RdRp pauses at each of the body's TRS sequences (TRS-B); after this pause the RdRp either continues to extend to the next TRS or moves to reinforce the leader sequence at the 5′ end of the genome guided by the complementarity of the TRS-B with the leader TRS (TRS-L). Several pieces of research currently support this theory, including the appearance on the 3′ end of subgenomic negative-strand RNAs of the anti‑leader sequence [77]. However, there are several queries left to completely explain the pattern. After the RNA replication and subgenomic synthesis, the viral structural proteins, S, E, and M are encoded and incorporated into the endoplasmic reticulum (ER). Such proteins pass through the secretory pathway into the ERGIC (Endoplasmic Reticulum-Golgi Intermediate Compartment) [78,79]. There, viral genomes encapsidated by the N protein bud forming mature virions in ERGIC membranes that contain viral structural proteins [80]. In several coronaviruses, S protein which is not assembled into virions transits to the cell surface where it facilitates fusion between infected cells and adjacent, uninfected cells. This contributes to the creation of large, multi-nucleated cells which allow the virus to spread within an infected organism without the detection or neutralization of virus-specific antibodies.
8. Diagnosis and treatment
Also, diagnosis is important in areas where there is a severe CoV epidemic, as is currently the case in the United States of America, India and Brazil. Cases recognizing will guide the effectiveness of new health strategies for disease prevention. RT-PCR has become the preferred method for human CoV diagnosis, because multiplex RT-PCR assays have been identified in real time, can detect all four respiratory HCoVs and could even be further adapted to new CoVs [81,82]. RT-PCR is used for testing of COVID-19 in real time. To present, there are no anti-viral therapies directly targeting human coronaviruses, so treatments are supportive only. The interferons (IFNs) are selective only slightly against in vitro coronaviruses [83]. IFNs infusion with ribavirin may well have improved in vitro activity against those coronaviruses compared to IFNs alone; however, the viability of this in vivo combination needs further evaluation [84].
As there is no proven treatment yet for the virus and pneumonia it causes, there are more than 70 drugs or combinations potentially worth trying [4]. An injectable drug named Remdesivir (Virus blocker by Gilead Sciences), a broad-spectrum antiviral drug is highly optimistic. It worked well in mice and monkeys infected with Middle East Respiratory Syndrome (MERS) but didn't work well when administered to Ebola victims in the Congo basin 2008 [85]. According to the Centers for Disease Control and Prevention (CDC), treatments are mainly based on the kind of treatment given for influenza or flu and other severe respiratory illnesses called supportive care. This supportive care treatment especially treats symptoms like cough, fever and shortness of breath. In mild cases, acetaminophen (Tylenol) medication is used to reduce fever. Sometimes, COVID-19 patients are given antiviral drug Oseltamivir or Tamiflu, which suppresses virus reproduction in some cases. However, in the case of pneumonia treatment involves ventilation through a mask or tube injected directly into the windpipe [86]. Table 1 shows the potential drugs and mechanism for the treatment of COVID-19. Most information for the endemic human coronavirus strain (HCoV-) 229E suggest that it may remain infectious on different types of materials for 2 h to 9 days. Extremely pathogenic MERS-CoV (Middle East Respiratory Syndrome-Coronavirus), TGEV (Transmissible Gastroenteritis Virus) and MHV (Mouse Hepatitis Virus) survival times have been shortened by higher temperatures such as 30 °C or 40 °C. Nonetheless, the lifespan of TGEV and MHV can be increased to about 28 days at 4 °C. Few comparative data obtained with SARS-CoV (Severe Acute Respiratory Syndrome-Coronavirus) indicate a prolonged persistence with higher inocula [[87], [88], [89], [90], [91], [92]]. At room temperature, it was also shown that HCoV-229E survives better at 50% compared to 30% RH [93]. In order to avoid inanimate surface transmission and contamination, suspended biocidal agents such as ethanol (78%–95%), 2-propanol (70%–100%), a mixture of 45% 2-propanol with 30% 1-propanol, glutardialdehyde (0.5%–2.5%), formaldehyde (0.7%–1%) and povidone‑iodine (0.23%–7.5%) were found to be effective in inactivating coronavirus infection. Sodium hypochlorite required a minimum concentration of at least 0.21% [[94], [95], [96]] to be more effective. However, biocidal agents in carrier tests such as Ethanol at concentrations between 62% and 71% showed 2.0–4.0 log infectivity reduction of the coronavirus on inanimate surfaces within 1 min of exposure time. Concentrations of 0.1%–0.5% sodium hypochlorite and 2% glutardialdehyde have also been quite effective with 3.0 log reduction in viral titre. Conversely, 0.04% of benzalkonium chloride, 0.06% of sodium hypochlorite and 0.55% of orthophtaldehyde were less successful. Surface disinfection with 0.1% sodium hypochlorite or 62%–71% ethanol seems to be promising in substantially decreasing surface contamination with coronavirus within 1 min of exposure. Kampf et al. [97] are highly optimistic that biocidal agents will show similar effects on SARS-CoV-2.
Table 1.
List of potential drugs currently being applied for the treatment of COVID-19.
| Potential drug | Mechanism | Study |
|---|---|---|
| Hydroxychloroquine | HCQ a safe and effective treatment against COVID-19 | [[110], [111], [112]] |
| Chloroquine | Increasing endosomal pH, immunomodulating, autophagy inhibitors | [113,114] |
| Azithromycin | Significant reduction in viral load in COVID-19 patients | [110,115] |
| Human immunoglobulin | Contain natural antibodies and proteins representing the first line of defence against pathogens | [[116], [117], [118]] |
| Remdesivir | Blocks the replication of coronaviruses | [119,120] |
| Arbidol (umifenovir) | It blocks the entry of the virus into the host cell | [121,122] |
| Oseltamivir | Inhibitor of viral neuraminidase and consequently blocks the release of viral particles from the host cell | [120,121,123,124] |
| Lopinavir-ritonavir | Inhibiting HIV-1 protease for protein cleavage, resulting in non-infectious, immature viral particles | [125,126] |
| Darunavir-cobicistat combination | HIV protease inhibitor and Pharmacokinetic and Pharmacodynamics booster | [[127], [128], [129]] |
| Traditional Chinese Medicine (TCM) combination with lopinavir-ritonavir, α-interferon via aerosol | Under trial | [[130], [131], [132], [133]] |
| Recombinant human interferon α2β | Inhibits MERS-CoV and SARS-CoV | [82,126,132] |
| Danoprevir-ritonavir and interferoninhalation or lopinavir-ritonavir or TCM plus interferon inhalation | Under trial | [134,135] |
| Xiyanping | Significant antiviral and antibacterial effects | [[136], [137], [138]] |
| Combinations of Oseltamivir, favipiravir, and chloroquine | Under trial | [121] |
| Thalidomide | Degrades messenger RNA in blood cells and reduces tumor necrosis factor-α (TNFα) | [[139], [140], [141]] |
| Vitamin C | Antioxidant and reduces oxidative stress, inflammation, improves vasopressor synthesis and immune cell function | [142,143] |
| Methylprednisolone | Prolongs the survival time of the clinical cases | [144,145] |
| Bromhexine hydrochloride | Transmembrane protease serine inhibitor, responsible for activation of S-glycoprotein of MERS-CoV and SARS-CoV | [146,147] |
| Bevacizumab | Suppresses the edema in COVID-19 patients by reducing the levels of vascular endothelial growth factor (VEGF) | [148,149] |
| Fingolimod | Immunology modulator used in multiple sclerosis | [150] |
| Baricitinib | Binding to AP2-associated protein kinase 1 (AAK1) | [151] |
| Lithium | Probably by reducing apoptosis and inhibition of glycogen synthase kinase 3 beta (GSK-3β) | [152] |
| Angiotensin-converting enzyme inhibitors and Angiotensin1 receptor inhibitors | Rebalancing Renin-Angiotensin-Aldosterone System (RAAS) (might reduce the pulmonary inflammatory response and mortality) | [153] |
| Cepharanthine, selamectin and mefloquine | Complete inhibition of cytopathic effects in cell culture by all three drugs |
[154] |
| Qingfei paidu decoction | Controlling of COVID-19 symptoms |
[155] |
| Pirfenidone | Anti-inflammatory and anti-oxidant by inhibiting IL-1β and IL-4 | [156] |
9. Effect of environmental factors on viruses
Various studies have identified the potential relationship between environment and respiratory infections, such as respiratory syncytial infection (RSV), which causes mild, cold-like side effects in adults and healthier children, SARS, and flu [[98], [99], [100]]. There is also some evidence that latitude may play a role in viral transmission, and there is considerable incongruity in the results of the study [100,101]. At least three factors have been attributed to viral transmission due to change in seasons.1. Infection tolerance to the host may be due to seasonal melatonin fluctuations [[102], [103]] 2. Metabolites of diet D or a deficiency in vitamin D can also weaken human immunity [104] 3. A difference in ambient temperature and relative humidity (RH) impact virus survival [105]. Spending more time indoors is postulated to improve the transmissibility of influenza. Viral vulnerability to RH tends to be an independent characteristic of a virus. For instance, in the next few studies the incidence of RSV will increase with increasing temperature but with lowering temperatures in others and peaks in different parts of the world at both low and high temperatures [106]. RSV is correlated with peaks of high and low temperature activity and 45%–65% RH, while one exclusive virus survives best at high RH and two others thrive best at low RH [107]. No link between survival and temperature has been found in a Swedish investigation [108]. RSV pastime was constant throughout the year in continuously warm and high humidity regions, as well as in areas where temperatures stayed cold throughout the year, but RSV operation was highest in temperate climates during the winter, associated with lower temperatures [98]. There is no definite proof that any single factor, be it a specific temperature, RH or geographic location, can be applied universally to a wide range of infectious viruses to reduce airborne or touch transmission, even though there is widespread evidence in the literature that perhaps the survival of viruses and various infectious agents depends to some extent on the RH levels [109].
10. Conclusion and perspective
The COVID-19 caused by SARS-CoV-2, that emerged in December 2019, remained a controversial subject of intense speculations regarding its origin, rapidly escalated into a global health emergency, posing a challenge to the existing health care system and scientific technology. Researchers have made significant progress in characterizing the SARS-CoV-2 and are working extensively on prospective vaccines and virus therapies. In this backdrop, integrated biological and behavioural surveillance will be of prime importance in identifying the potential hotspots of COVID-19 [100]. Due to the absence of successful therapeutics or vaccinations, the best approaches for managing human coronaviruses are to build a steady system of public health surveillance combined with rapid diagnostic testing and quarantine when appropriate. We also believe that policies must be reviewed regarding the utilisation of wild birds and animals as a source of food. As the COVID-19 pandemic is rapidly spreading throughout the world. The Case rates and Case fatality rates are continuously fluctuating. Thus it is important to understand the dynamics of the virus holistically to develop ways to control it. Furthermore, cooperation of government agencies, public health authorities, and healthcare professionals throughout the world is critical for managing the COVID-19 pandemic.
Conflict of interest statement
The authors declare that there are no conflicts of interest.
Author Contributors
Mohd Sharjeel Sofi: Conceptualization, Writing - original draft. Aadil Hamid: Conceptualization, Writing - original draft. Sami Ullah Bhat: Supervision, Writing - review & editing.
References
- 1.Dietz L., Patrick H.F., David C., Mark F., Eisen J.A., Kevin V.D.W. 2019 novel coronavirus (COVID-19) pandemic: built environment considerations to reduce transmission. M. Systems. 2020;5 doi: 10.1128/mSystems.00245-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Tan W., Zhao X., Ma X. A novel coronavirus genome identified in a cluster of pneumonia cases—Wuhan, China 2019–2020. China CDC Weekly. 2020;2:61–62. doi: 10.46234/ccdcw2020.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Zhu N., Zhang D., Wang W. A novel coronavirus from patients with pneumonia in China, 2019. N. Engl. J. Med. 2020;382(8):727–733. doi: 10.1056/NEJMoa2001017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.World Health Organization (WHO) Novel Coronavirus(2019-nCoV) Situation Report – 22. 2020. https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200211-sitrep-22-ncov.pdf (accessed 13 April 2020)
- 5.Weiner L.P. In: Coronaviruses. Lai M.M.C., Stohlman S.A., editors. Advances in Experimental Medicine and Biology; Springer Boston, MA: 1987. Coronaviruses: A historical perspective; pp. 1–5. [DOI] [PubMed] [Google Scholar]
- 6.Baudette F.R., Hudson C.B. New recognized poultry disease. N. Am. Vet. 1933;14:50–54. [Google Scholar]
- 7.Baudette F.R., Hudson C.B. Cultivation of the virus of infectious bronchitis. JAVMA. 1937;90:51–60. [Google Scholar]
- 8.Gledhill A.W., Dick G.W.A., Andrewes C.H. Production of hepatitis in mice by the combined action of two filterable agents. Lancet. 1952;2:509–511. doi: 10.1016/s0140-6736(52)90292-4. [DOI] [PubMed] [Google Scholar]
- 9.Gledhill A.W., Niven J.S.F. Latent virus as exemplified by mouse hepatitis virus (MHV) Vet. Rev. Annotations. 1955;1:82–90. [Google Scholar]
- 10.Sturman L.S., Holmes K.V. The molecular biology of coronaviruses. Adv. Virus Res. 1983;28:35–112. doi: 10.1016/s0065-3527(08)60721-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Mahy B.W.J. Coronavirus come of age. Nature. 1980;288:536–538. doi: 10.1038/288536a0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.WHO Coronavirus Disease 2019 (COVID-19). Situation Report – 88. 2020. https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200417-sitrep-88-covid-191b6cccd94f8b4f219377bff55719a6ed.pdf?sfvrsn=ebe78315_6 (accessed 19 April 2020)
- 13.World Health Organization (WHO) Coronavirus disease 2019 (COVID-19). Situation Report-133. 2020. https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200601-covid-19-sitrep-133.pdf?sfvrsn=9a56f2ac_4 (accessed 2 June 2020)
- 14.Worldometer. 2020. http://www.worldometersinfo/coronavirus/#countries (accessed 2 June 2020)
- 15.Chan J.F.W., Yuan S.K.H., Kok A. A familial cluster of pneumonia associated with the 2019 novel coronavirus indicating person-to-person transmission: a study of a family cluster. Lancet. 2020;395:514–523. doi: 10.1016/S0140-6736(20)30154-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Huang C., Wang Y., Li X. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Su S., Wong G., Shi W. Epidemiology, genetic recombination, and pathogenesis of coronaviruses. Trends Microbiol. 2016;24:490–502. doi: 10.1016/j.tim.2016.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Louis D. Temporal Signal of nCoV-2019 Based on 30 Genomes. 2020. https://virological.org/t/phylodynamic-analysis-of-ncov-2019-genomes-29-jan-2020/353/7 (accessed 13 February 2020)
- 19.Rambaut A. Phylodynamic Analysis | 176 Genomes. 2020. https://virological.org/t/phylodynamic-analysis-176-genomes-6-mar-2020/356 (accessed 13 February 2020)
- 20.MacLean O.A., Orton R.J., Singer J.B., Robertson D.L. No evidence for distinct types in the evolution of SARS-CoV-2. Virus Evol. 2020;6(1) doi: 10.1093/ve/veaa034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Chen N., Zhou M., Dong X., et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020;395:507–513. doi: 10.1016/S0140-6736(20)30211-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Guan W.J., Ni Z.Y., Hu Y., et al. Clinical characteristics of coronavirus disease 2019 in China. NEJM. 2020;382:1708–1720. doi: 10.1056/NEJMoa2002032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.K.K. To, Tsang O.T., Leung W.S., et al. Temporal profiles of viral load in posterior oropharyngeal saliva samples and serum antibody responses during infection by SARS-CoV-2: an observational cohort study. Lancet Infect. Dis. 2020;20:565–574. doi: 10.1016/S1473-3099(20)30196-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Swerdlow D.L., Finelli L. Preparation for possible sustained transmission of 2019 novel coronavirus: lessons from previous epidemics. JAMA. 2020;323(12):1129–1130. doi: 10.1001/jama.2020.1960. [DOI] [PubMed] [Google Scholar]
- 25.Liu Z., Bing X., Zhi X.Z. Epidemiology working group for NCIP epidemic response. China CDC Weekly. 2020;41(2):145–151. doi: 10.3760/cma.j.issn.0254-6450.2020.02.003. [DOI] [Google Scholar]
- 26.Tian S., Hu N., Lou J., Chen K., Kang X., Xiang Z., Chen H., Wang D., Liu N., Liu D., Chen G., Zhang Y., Li D., Li J., Lian H., Niu S., Zhang L., Zhang J. Characteristics of COVID-19 infection in Beijing. J. Inf. Secur. 2020;80(4):401–406. doi: 10.1016/j.jinf.2020.02.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Jung S.-M., Akhmetzhanov A.R., Hayashi K., Linton N.M., Yang Y., Yuan B., et al. Real-Time estimation of the risk of death from novel coronavirus (COVID-19) infection: inference using exported cases. MedRxiv. 2020;9(2):523. doi: 10.1101/2020.01.29.20019547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Livingston E., Bucher K. Coronavirus disease 2019 (COVID-19) in Italy. JAMA. 2020;323(14):1335. doi: 10.1001/jama.2020.4344. [DOI] [PubMed] [Google Scholar]
- 29.Li R., Pei S., Chen B., et al. Substantial undocumented infection facilitates the rapid dissemination of novel coronavirus (SARS-CoV-2) J. Sci. 2020;368:489–493. doi: 10.1126/science.abb3221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Huang C., Wang Y., Li X., et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Wang D., Hu B., Hu C., et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA. 2020;323:1061–1069. doi: 10.1001/jama.2020.1585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Gao P., Zhang H., Wu Z., Wang J. Visualizing the expansion and spread of coronavirus disease 2019 by cartograms. Environ. Plan. A. 2020;52:698–701. doi: 10.1177/0308518X20910162. [DOI] [Google Scholar]
- 33.Zhang H., Chen Y., Gao P., Wu Z. Mapping the changing internet attention to the spread of coronavirus disease 2019 in China. Environ. Plan. A. 2020;52(4):691–694. doi: 10.1177/0308518X20922238. [DOI] [Google Scholar]
- 34.Gastner M.T., Newman M.E. Diffusion-based method for producing density-equalizing maps. PNAS USA. 2004;101(20):7499–7504. doi: 10.1073/pnas.0400280101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Sun H., Li Z.L. Effectiveness of cartogram for the representation of spatial data. Cartogr. J. 2010;47(1):12–21. doi: 10.1179/000870409X12525737905169. [DOI] [Google Scholar]
- 36.Gorbalenya E., Enjuanes L., Ziebuhr J., Snijder E.J. Nidovirales: evolving the largest RNA virus genome. Virus Res. 2006;117:17–37. doi: 10.1016/j.virusres.2006.01.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.De Vries A.A.F., Horzinek M.C., Rottier P.J.M., De Groot R.J. The genome organization of the nidovirales: similarities and differences between Arteri-, Toro-, and coronaviruses. Semin. Virol. 1997;8(1):33–47. doi: 10.1006/smvy.1997.0104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Cong Y., Zarlenga D.S., Richt J.A., Wang X., Wang Y., Suo S., Wang J., Ren Y., Ren X. Evolution and homologous recombination of the hemagglutinin-esterase gene sequences from porcine torovirus. Virus Genes. 2013;47:66–74. doi: 10.1007/s11262-013-0926-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Vasilakis N., Guzman H., Firth C., Forrester N.L., Widen S.G., Wood T.G., Rossi S.L., Ghedin E., Popov V., Blasdell K.R. Mesoniviruses are mosquito-specific viruses with extensive geographic distribution and host range. Virol. J. 2014;11:97. doi: 10.1186/1743-422X-11-97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Fehr R.A., Stanle P. Coronaviruses: an overview of their replication and pathogenesis. Coronaviruses. 2015;1282:1–23. doi: 10.1007/978-1-4939-2438-7_1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Chibo D., Birch C. Analysis of human coronavirus 229E spike and nucleoprotein genes demonstrates genetic drift between chronologically distinct strains. J. Gen. Virol. 2006;87:1203–1208. doi: 10.1099/vir.0.81662-0. [DOI] [PubMed] [Google Scholar]
- 42.Vijgen L., Keyaerts E., Lemey P. Circulation of genetically distinct contemporary human coronavirus OC43 strains. Virol. 2005;337:85–92. doi: 10.1016/j.virol.2005.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Lau S.K., Woo P.C., Li K.S. Severe acute respiratory syndrome coronavirus-like virus in Chinese horseshoe bats. PNAS USA. 2005;102:14040–14045. doi: 10.1073/pnas.0506735102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Li W., Shi Z., Yu M., Ren W., Smith C., Epstein J.H., Wang H., Crameri G., et al. Bats are natural reservoirs of SARS-like coronaviruses. Science. 2005;310:676–679. doi: 10.1126/science.1118391. [DOI] [PubMed] [Google Scholar]
- 45.Peiris J.S., Chu C.M., Cheng V.C., et al. Clinical progression and viral load in a community outbreak of coronavirus-associated SARS pneumonia: a prospective study. Lancet. 2003;361:1767–1772. doi: 10.1016/s0140-6736(03)13412-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Spiegel M., Schneider K., Weber F., et al. Interaction of severe acute respiratory syndrome- associated coronavirus with dendritic cells. J. Gen. Virol. 2006;87:1953–1960. doi: 10.1099/vir.0.81624-0. [DOI] [PubMed] [Google Scholar]
- 47.Law H.K., Cheung C.Y., Ng H.Y., et al. Chemokine upregulation in SARS coronavirus infected human monocyte derived dendritic cells. Blood. 2005;106:2366–2376. doi: 10.1182/blood-2004-10-4166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Lauer S., Grantz K., Bi Q., Jones F., et al. The incubation period of coronavirus disease 2019 (COVID-19) from publicly reported confirmed cases: estimation and application. Ann. Intern. Med. 2020;173:577–582. doi: 10.7326/M20-0504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Baruah V., Bose S. Immuno informatics-aided identification of T cell and B cell epitopes in the surface glycoprotein of 2019-nCoV. J. Med. Virol. 2020;92(5):495–500. doi: 10.1002/jmv.25698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Barcena M., Oostergetel G.T., Bartelink W., Faas F.G., Verkleij A., Rottier P.J., Koster A.J., Bosch B.J. Cryo-electron tomography of mouse hepatitis virus: insights into the structure of the coronavirion. PNAS USA. 2009;106(2):582–587. doi: 10.1073/pnas.0805270106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Neuman B.W., Adair B.D., Yoshioka C., Quispe J.D., Orca G., Kuhn P., Milligan R.A., Yeager M., Buchmeier M.J. Supramolecular architecture of severe acute respiratory syndrome coronavirus revealed by electron cryomicroscopy. J. Virol. 2006;80(16):7918–7928. doi: 10.1128/JVI.00645-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Armstrong J., Niemann H., Smeekens S., Rottier P., Warren G. Sequence and topology of a model intracellular membrane protein, E1 glycoprotein, from a coronavirus. Nature. 1984;308(5961):751–752. doi: 10.1038/308751a0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Neuman B.W., Kiss G., Kunding A.H., Bhella D., Baksh M.F., Connelly S., Droese B.J.P. Klaus, Makino S., Sawicki S.G., Siddell S.G., Stamou D.G., Wilson I.A., Kuhn P., Buchmeier M.J. A structural analysis of M protein in coronavirus assembly and morphology. J. Struct. Biol. 2011;174(1):11–22. doi: 10.1016/j.jsb.2010.11.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Godet M., L’Haridon R., Vautherot J.F., Laude H. TGEV corona virus ORF4 encodes a membrane protein that is incorporated into virions. Virol. 1992;188(2):666–675. doi: 10.1016/0042-6822(92)90521-P. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Nieto-Torres J.L., Dediego M.L., Verdia-Baguena C., Jimenez-Guardeno J.M., Regla-Nava J.A., Fernandez-Delgado R., Castano-Rodriguez C., Alcaraz A., Torres J., Aguilella V.M., Enjuanes L. Severe acute respiratory syndrome coronavirus envelope protein ion channel activity promotes virus fitness and pathogenesis. PLoS Pathog. 2014;10(5) doi: 10.1371/journal.ppat.1004077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Klausegger A., Strobl B., Regl G., Kaser A., Luytjes W., Vlasak R. Identification of a coronavirus hemagglutinin-esterase with a substrate specificity different from those of influenza C virus and bovine coronavirus. J. Virol. 1999;73(5):3737–3743. doi: 10.1128/JVI.73.5.3737-3743.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Cornelissen L.A., Wierda C.M., van der Meer F.J., Herrewegh A.A., Horzinek M.C., Egberink H.F., de Groot R.J. Hemagglutinin-esterase, a novel structural protein of torovirus. J. Virol. 1997;71(7):5277–5286. doi: 10.1128/jvi.71.7.5277-5286.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Qamar T. Ul, Alqahtani S.M., Alamri M.A., Chen L.L. Structural basis of SARS-CoV-2 3CLpro and anti-COVID-19 drug discovery from medicinal plants. J. Pharm. Anal. 2020;10:313–319. doi: 10.1016/j.jpha.2020.03.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Xu X., Chen P., Wang J., et al. Evolution of the novel coronavirus from the ongoing Wuhan 256 outbreak and modeling of its spike protein for risk of human transmission. Sci. China Life Sci. 2020;63:457–460. doi: 10.1007/s11427-020-1637-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Zhou P., Yang X.L., Wang X.G., et al. A pneumonia outbreak associated with a new 327 coronavirus of probable bat origin. Nature. 2020;579:270–273. doi: 10.1038/s41586-020-2012-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Zhu N., Zhang D., Wang W., et al. A novel coronavirus from patients with pneumonia in 261 China, 2019. NEJM. 2020;382:727–733. doi: 10.1056/NEJMoa2001017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Kubo H., Yamada Y.K., Taguchi F. Localization of neutralizing epitopes and the receptor-binding site within the amino-terminal 330 amino acids of the murine coronavirus spike protein. J. Virol. 1994;68:5403–5410. doi: 10.3109/00365517109080235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Cheng P.K., Wong D.A., Tong L.K., Ming Ip Sin, Lo A.C.T., Lau C.S., Yeung E.Y., Lim W.W. Viral shedding patterns of coronavirus in patients with probable severe acute respiratory syndrome. Lancet. 2004;363(9422):1699–1700. doi: 10.1016/S0140-6736(04)16255-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Guan B.J., Wu H.Y., Brian D.A. An optimal cis-replication stem-loop IV in the 5′ untranslated region of the mouse coronavirus genome extends 16 nucleotides into open reading frame 1. J. Virol. 2011;85(11):5593–5605. doi: 10.1128/JVI.00263-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Belouzard S., Chu V.C., Whittaker G.R. Activation of the SARS coronavirus spike protein via sequential proteolytic cleavage at two distinct sites. PNAS USA. 2009;106(14):5871–5876. doi: 10.1073/pnas.0809524106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Ziebuhr J., Snijder E.J., Gorbalenya A.E. Virus-encoded proteinases and proteolytic processing in the Nidovirales. J. Virol. 2000;81(4):853–879. doi: 10.1099/0022-1317-81-4-853. [DOI] [PubMed] [Google Scholar]
- 67.Brown C.G., Nixon K.S., Senanayake S.D., Brian D.A. An RNA stem-loop within the bovine coronavirus nsp1 coding region is a cis-acting element in defective interfering RNA replication. J. Virol. 2007;81(14):7716–7724. doi: 10.1128/JVI.00549-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Emery S.L., Erdman D.D., Bowen M.D. Real-time reverse transcription polymerase chain reaction assay for SARS associated coronavirus. Emerg. Infect. Dis. 2004;10:311–316. doi: 10.3201/eid1002.030759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Liu P., Li L., Keane S.C., Yang D., Leibowitz J.L., Giedroc D.P. Mouse hepatitis virus stem-loop 2 adopts a uYNMG(U)a-like tetraloop structure that is highly functionally tolerant of base substitutions. J. Virol. 2009;83(23):12084–12093. doi: 10.1128/JVI.00915-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Raman S., Bouma P., Williams G.D., Brian D.A. Stem-loop III in the 5′ untranslated region is a cisacting element in bovine coronavirus defective interfering RNA replication. J. Virol. 2003;77(12):6720–6730. doi: 10.1128/jvi.77.12.6720-6730.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Liu Q., Johnson R.F., Leibowitz J.L. Secondary structural elements within the 3′ untranslated region of mouse hepatitis virus strain JHM genomic RNA. J. Virol. 2001;75(24):12105–12113. doi: 10.1128/JVI.75.24.12105-12113.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Goebel S.J., Miller T.B., Bennett C.J., Bernard K.A., Masters P.S. A hypervariable region within the 3′ cis-acting element of the murine coronavirus genome is nonessential for RNA synthesis but affects pathogenesis. J. Virol. 2007;81(3):1274–1287. doi: 10.1128/JVI.00803-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Williams G.D., Chang R.Y., Brian D.A. A phylogenetically conserved hairpin-type 3′ untranslated region pseudoknot functions in coronavirus RNA replication. J. Virol. 1999;73(10):8349–8355. doi: 10.1128/JVI.73.10.8349-8355.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Hsue B., Masters P.S. A bulged stem-loop structure in the 3′ untranslated region of the genome of the coronavirus mouse hepatitis virus is essential for replication. J. Virol. 1997;71(10):7567–7578. doi: 10.1097/DMP.0b013e3181493708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Hsue B., Hartshorne T., Masters P.S. Characterization of an essential RNA secondary structure in the 3′ untranslated region of the murine coronavirus genome. J. Virol. 2000;74(15):6911–6921. doi: 10.1128/JVI.74.15.6911-6921.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Sawicki S.G., Sawicki D.L., Siddell S.G. A contemporary view of coronavirus transcription. J. Virol. 2007;81(1):20–29. doi: 10.1128/jvi.01358-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Sethna P.B., Hofmann M.A., Brian D.A. Minus-strand copies of replicating coronavirus mRNAs contain antileaders. J. Virol. 1991;65(1):320–325. doi: 10.1128/JVI.65.1.320-325.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Krijnse-Locker J., Ericsson M., Rottier P.J.M., Griffiths G. Characterization of the budding compartment of mouse hepatitis virus: evidence that transport from the RER to the golgi complex requires only one vesicular transport step. J. Cell Biol. 1994;124:55–70. doi: 10.1083/jcb.124.1.55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Tooze J., Tooze S., Warren G. Replication of coronavirus MHV-A59 in sac- cells: determination of the first site of budding of progeny virions. Eur. J. Cell Biol. 1984;33(2):281–293. doi: 10.1016/S0140-6736(02)41295-0. [DOI] [PubMed] [Google Scholar]
- 80.de Haan C.A., Rottier P.J. Molecular interactions in the assembly of coronaviruses. Adv. Virus Res. 2005;64:165–230. doi: 10.1016/S0065-3527(05)64006-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Templeton K.E., Scheltinga S.A., Beersma M.F.C., Kroes A.C.M., Claas E.C.J. Rapid and sensitive method using multiplex real-time PCR for diagnosis of infections by influenza A and influenza B viruses, respiratory syncytial virus, and parainfluenza viruses 1, 2, 3, and 4. J. Clin. Microbiol. 2004;42:1564–1569. doi: 10.1128/JCM.42.4.1564-1569.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Gaunt E.R., Hardie A., Claas E.C. Epidemiology and clinical presentations of the four human coronaviruses 229E, HKU1, NL63, and OC43 detected over 3 years using a novel multiplex real-time PCR method. J. Clin. Microbiol. 2010;48:2940–2947. doi: 10.1128/JCM.00636-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Cinatl J., Morgenstern B., Bauer G. Treatment of SARS with human interferons. Lancet. 2003;362:293–294. doi: 10.1016/S0140-6736(03)13973-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Stockman L.J., Bellamy R., Garner P. SARS: systematic review of treatment effects. PLoS Med. 2006;3 doi: 10.1371/journal.pmed.0030343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Regalado A. What are the Best Coronavirus Treatments? MIT Technology Review. 2020. www.tecgnologyreview.com (accessed 5 March 2020)
- 86.Pappas S. Is There a Cure for the New Coronavirus? Live Science. 2020. www.livescience.com (accessed 5 March 2020)
- 87.Sizun J., Yu M.W., Talbot P.J. Survival of human coronaviruses 229E and OC43 in suspension and after drying on surfaces: a possible source of hospital-acquired infections. J. Hosp. Infect. 2000;46:55–60. doi: 10.1053/jhin.2000.0795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Duan S.M., Zhao X.S., Wen R.F., Huang J.J., Pi G.H., Zhang S.X., et al. Stability of SARS coronavirus in human specimens and environment and its sensitivity to heating and UV irradiation. Biomed. Environ. Sci. 2003;16:246–255. https://doi.org/CNKI:SUN:SWYX.0.2003-03-006. [PubMed] [Google Scholar]
- 89.Lai M.Y., Cheng P.K., Lim W.W. Survival of severe acute respiratory syndrome coronavirus. Clin. Infect. Dis. 2005;41:e67–e71. doi: 10.1086/433186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.van Doremalan N., Bushmaker T., Munster V.J. Stability of Middle East respiratory syndrome coronavirus (MERS-CoV) under different environmental conditions. Eur. Surveill. 2013;18:20590. doi: 10.2807/1560-7917.es2013.18.38.20590. [DOI] [PubMed] [Google Scholar]
- 91.Casanova L.M., Jeon S., Rutala W.A., Weber D.J., Sobsey M.D. Effects of air temperature and relative humidity on coronavirus survival on surfaces. Appl. Environ. Microbiol. Appl. Environ. Microbiol. 2010;76:2712–2717. doi: 10.1128/AEM.02291-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Warnes S.L., Little Z.R., Keevil C.W. Human coronavirus 229E remains infectious on common touch surface materials. MBio. 2015;6 doi: 10.1128/mBio.01697-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Ijaz M.K., Brunner A.H., Sattar S.A., Nair R.C., Johnson-Lussenburg C.M. Survival characteristics of airborne human coronavirus 229E. J. Gen. Virol. 1985;66:2743–2748. doi: 10.1099/0022-1317-66-12-2743. [DOI] [PubMed] [Google Scholar]
- 94.Rabenau H.F., Kampf G., Cinatl J., Doerr H.W. Efficacy of various disinfectants against SARS coronavirus. J. Inf. Secur. 2005;61:107–111. doi: 10.1016/j.jhin.2004.12.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Saknimit M., InatsukiI I., Sugiyama Y., Yagami K., et al. Jikken Dobutsu. 1988;37:341–345. doi: 10.1538/expanim1978.37.3_341. [DOI] [PubMed] [Google Scholar]
- 96.Siddharta A., Pfaender S., Vielle N.J., Dijkman R., Friesland M., Becker B., et al. Virucidal activity of World Health Organization- recommended formulations against enveloped viruses, including Zika, Ebola, and emerging coronaviruses. J. Infect. Dis. 2017;215:902–906. doi: 10.1093/infdis/jix046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Kampf G., Todt D., Pfaender S., Steinmann E. Persistence of coronaviruses on inanimate surfaces and their inactivation with biocidal agents. J. Hosp. Infect. 2020;104:246–251. doi: 10.1016/j.jhin.2020.01.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Thompson W.W., Shay D.K., Weintraub E. Mortality associated with influenza and respiratory syncytial virus in the United States. JAMA. 2003;289:179–186. doi: 10.1001/jama.289.2.179. [DOI] [PubMed] [Google Scholar]
- 99.Thompson W.W., Comanor L., Shay D.K. Epidemiology of seasonal influenza: use of surveillance data and statistical models to estimate the burden of disease. J. Infect. Dis. 2006;194:S82–S91. doi: 10.1086/507558. [DOI] [PubMed] [Google Scholar]
- 100.Tang J.W. The effect of environmental parameters on the survival of airborne infectious agents. J. R. Soc. Interface. 2009;6:S737–S746. doi: 10.1098/rsif.2009.0227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Cecile V., Wladimir J.A., Lone S. Influenza in tropical regions. PLoS Med. 2006;3 doi: 10.1371/journal.pmed.0030089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Tang J.W., Lai F.Y., Wong F., Hon K.L. Incidence of common respiratory viral infections related to climate factors in hospitalized children in Hong Kong. Epidemiol. Infect. 2010;138:226–235. doi: 10.1017/S0950268809990410. [DOI] [PubMed] [Google Scholar]
- 103.Dowell S.F. Seasonal variation in host susceptibility and cycles of certain infectious diseases. Emerg. Infect. Dis. 2001;7(3):369–374. doi: 10.3201/eid0703.010301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Cannell J.J., Vieth R., Umhau J.C., Holick M.F., Grant W.B., Madronich S., Garland C.F., Giovannucci E. Epidemic influenza and vitamin D. Epidemiol. Infect. 2006;134(6):1129–1140. doi: 10.1017/S0950268806007175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Shaman J., Kohn M. Absolute humidity modulates influenza survival, transmission, and seasonality. Proc. Natl. Acad. Sci. U. S. A. 2009;106:3243–3248. doi: 10.1073/pnas.0806852106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Songer J.R. Influence of RH on the survival of some airborne viruses. Appl. Microbiol. 1967;15:35–42. doi: 10.1128/am.15.1.35-42.1967. https://aem.asm.org/content/aem/15/1/35.full.pdf [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Welliver R.C., Sr. Temperature, humidity, and ultraviolet B radiation predict community respiratory syncytial virus activity. Pediatr. Infect. Dis. 2007;26(11):S29–S35. doi: 10.1097/INF.0b013e318157da59. [DOI] [PubMed] [Google Scholar]
- 108.Linde A., Rotzén-Östlund M., Zweygberg-Wirgart B., Rubinova S., Brytting M. Does viral interference affect spread of influenza? Eur. Surveill. 2009;14(40) doi: 10.1186/1475-2875-8-226. pii=19354, [DOI] [PubMed] [Google Scholar]
- 109.Farhad M. Literature review of the effect of temperature and humidity on viruses. ASHRAE Trans. 2011;117(2) [Google Scholar]
- 110.Gautret P., Lagier J.C., Parola P., Hoang V.T., Medde L., Mailhe M., Doudier B., Courjon J., Giordanengo V., Vieira V.E., Dupont H.T., Honoré S., Colson P., Chabrière E., Scola B.L., Rolain J.M., Brouqui P., Raoulta D. Hydroxychloroquine and azithromycin as a treatment of COVID-19: results of an open-label non-randomized clinical trial. Int. J. Antimicrob. 2020;56(1):105949. doi: 10.1016/j.ijantimicag.2020.105949. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 111.Liu J., Cao R., Xu M., et al. Hydroxychloroquine, a less toxic derivative of chloroquine, is effective in inhibiting SARS-CoV-2 infection in vitro. Cell Discov. 2020;6:16. doi: 10.1038/s41421-020-0156-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). 2020 Feb 18 – Identifier NCT01067417, Evaluation of the Efficacy of Hydroxychloroquine in Decreasing Immune Activation in Asymptomatic HIV-Infected Patients (HCQ01) 2020. https://clinicaltrials.gov/ct2/show/NCT01067417 (accessed 20 March 2020)
- 113.Rolain J.M., Colson P., Raoult D. Recycling of chloroquine and its hydroxyl analogue to face bacterial, fungal and viral infections in the 21st century. Int. J. Antimicrob. 2007;30(4):297–308. doi: 10.1016/j.ijantimicag.2007.05.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). 2020 Mar 12 – Identifier NCT04303507, Chloroquine Prevention of Coronavirus Disease (COVID-19) in the Healthcare Setting (COPCOV) 2020. https://clinicaltrials.gov/ct2/show/NCT04291729?term=NCT04291729&draw=2&rank=1 (accessed 15 April 2020)
- 115.ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). 2020 Jan 22 – Identifier Treatment and Prevention of Traditional Chinese Medicines (TCMs) on COVID-19 Infection. 2020. https://clinicaltrials.gov/ct2/show/NCT04251871 (accessed 15 March 2020)
- 116.Cherin P., Marie I., Michallet M., Pelus E., Dantal J., Crave J.C., et al. Management of adverse events in the treatment of patients with immunoglobulin therapy: a review of evidence. Autoimmun. Rev. 2016;15(1):71–81. doi: 10.1016/j.autrev.2015.09.002. [DOI] [PubMed] [Google Scholar]
- 117.Varadarajan R., Srinivasan S., Maity S., Ghosh M. Broadly neutralizing antibodies for therapy of viral infections. Antib. Technol. J. 2016;6:1–15. doi: 10.2147/ANTI.S92190. [DOI] [Google Scholar]
- 118.ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). 2020 Mar 12 – Identifier NCT04261426, The Efficacy of Intravenous Immunoglobulin Therapy for Severe 2019-nCoV infected Pneumonia. 2020. https://clinicaltrials.gov/ct2/show/NCT04261426 (accessed 20 April 2020)
- 119.Dyer O. Two Ebola treatments halve deaths in trial in DRC outbreak. BMJ. 2019;366:15140. doi: 10.1136/bmj.l5140. [DOI] [PubMed] [Google Scholar]
- 120.Wang M., Cao R., Zhang L., Yang X., Liu J., Xu M., et al. Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro. Cell Res. 2020;30:269–271. doi: 10.1038/s41422-020-0282-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Blaising J. SJ. Polyak, EI. Pecheur, Arbidol as a broad-spectrum antiviral: an update. Antivir. Res. 2014;107(1):84–94. doi: 10.1016/j.antiviral.2014.04.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). 2020 Mar 12 – Identifier NCT04260594, Clinical Study of Arbidol Hydrochloride Tablets in the Treatment of Pneumonia Caused by Novel Coronavirus. 2020. https://clinicaltrials.gov/ct2/show/NCT04260594 (accessed 21 April 2020)
- 123.ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). 2020 Mar 12 – Identifier NCT04303299, Various combination of Protease Inhibitors, Oseltamivir, Favipiravir, and Chloroquine for Treatment of COVID-19: A Randomized Control Trial (THDMS-COVID19) 2020. https://clinicaltrials.gov/ct2/show/NCT04291729?term=NCT04291729&draw=2&rank=1 (accessed 15 April 2020)
- 124.Uyeki T.M. Oseltamivir treatment of influenza in children. Clin. Infect. Dis. 2018;66(10):1501–1503. doi: 10.1093/cid/cix1150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Okubo K., Isono M., Asano T., Sato A. Lopinavir-ritonavir combination induces endoplasmic reticulum stress and kills urological cancer cells. Anticancer Res. 2019;39(11):5891–5901. doi: 10.21873/anticanres.13793. [DOI] [PubMed] [Google Scholar]
- 126.Arabi Y.M., Alothman A., Balkhy H.H., Al-Dawood A., AlJohani S., Al Harbi S., et al. Treatment of Middle East Respiratory Syndrome with a combination of lopinavir-ritonavir and interferon-β1b (MIRACLE trial): Study protocol for a randomized controlled trial. Trials. 2018;19(1):1–13. doi: 10.1186/s13063-019-3846-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Santos J.R., Curran A., Navarro-Mercade J., Ampuero M.F., Pelaez P., Pérez-Alvarez N., et al. Simplification of antiretroviral treatment from darunavir/ritonavir monotherapy to darunavir/cobicistat monotherapy: effectiveness and safety in routine clinical practice. AIDS Res. Hum. Retrovir. 2019;35(6):513–518. doi: 10.1089/AID.2018.0178. [DOI] [PubMed] [Google Scholar]
- 128.Mathias A.A., German P., Murray B.P., Wei L., Jain A., West S., Warren D., Hui J., Kearney B.P. Pharmacokinetics and pharmacodynamics of cobicistat GS-9350: A novel pharmacokinetic enhancer without anti-HIV activity. Clin. Pharmacol. Ther. 2010;87(3):322–329. doi: 10.1038/clpt.2009.228. [DOI] [PubMed] [Google Scholar]
- 129.ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). 2020 Mar 12 – Identifier NCT04252274. Efficacy and Safety of Darunavir and Cobicistat for Treatment of Pneumonia Caused by 2019-nCoV (DACO-nCoV) 2020. https://clinicaltrials.gov/ct2/show/NCT04252274 (accessed 12 April 2020)
- 130.ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). 2020 Mar 12 – Identifier NCT04251871, Treatment and Prevention of Traditional Chinese Medicines (TCMs) on2019-nCoV Infection. 2020. https://clinicaltrials.gov/ct2/show/NCT04251871 (accessed 12 April 2020)
- 131.Luo Y., Wang C.Z., Hesse-Fong J., Lin J.G., Yuan C.S. Application of Chinese medicine in acute and critical medical conditions. Am. J. Chin. Med. 2019;47(6):1223–1235. doi: 10.1142/S0192415X19500629. [DOI] [PubMed] [Google Scholar]
- 132.Schneider W.M., Chevillotte M.D., Rice C.M. Vol. 32. 2015. Interferon-stimulated Genes: A complex web of host defences NIH public access; pp. 513–545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Cao B., et al. A trial of Lopinavir–Ritonavir in adults hospitalized with severe Covid-19. N. Engl. J. Med. 2020;382:1787–1799. doi: 10.1056/NEJMoa2001282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). 2020 Mar 12 – Identifier NCT04291729, Evaluation of Ganovo (Danoprevir) combined with Ritonavir in the treatment of the novel coronavirus infection. 2020. https://clinicaltrials.gov/ct2/show/NCT04291729?term=NCT04291729&draw=2&rank=1 (accessed 20 April 2020)
- 135.Markham A., Keam S.J. Vol. 78. 2018. Danoprevir: First Global Approval. Drugs; pp. 1271–1276. [DOI] [PubMed] [Google Scholar]
- 136.Tang T. Application of Xiyanping in treatment of infantile rotavirus diarrhea. J. Hainan Med. Univ. 2016;22(13):113–115. http://www.hnykdxxb.com/PDF/201613/30.pdf [Google Scholar]
- 137.ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). 2020 Mar 12 – Identifier NCT04275388, Xiyanping injection for the treatment of new coronavirus infected pneumonia. 2020. https://clinicaltrials.gov/ct2/show/NCT04275388 (accessed 20 April 2020)
- 138.ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). 2020 Mar 12 – Identifier NCT04295551, Multicentre clinical study on the efficacy and safety of xiyanping injection in the treatment of the new coronavirus infection pneumonia (general and severe) 2020. https://clinicaltrials.gov/ct2/show/NCT04295551?term=NCT04295551&draw=2&rank=1 (accessed 20 April 2020)
- 139.Newfield C. New medical indications for thalidomide and its derivatives new medical indications for thalidomide and its derivatives. Sci. J. Lander Coll. Arts Sci. 2018;12(1):11–19. [Google Scholar]
- 140.ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). 2020 Mar 12 – Identifier NCT04273581, The efficacy and safety of thalidomide combined with low-dose hormones in the treatment of severe covid-19. 2020. https://clinicaltrials.gov/ct2/show/NCT04273581?term=NCT04273581&draw=2&rank=1 (accessed 20 April 2020)
- 141.ClinicalTrials.gov [Internet]. Bethesda (MD): National library of medicine (us). 2020 mar 12 – identifier nct04273529, the efficacy and safety of thalidomide in the adjuvant treatment of moderate new coronavirus (covid-19) pneumonia. 2020. https://clinicaltrials.gov/ct2/show/NCT04273529?term=NCT04273529&draw=2&rank=1 (accessed 20 April 2020)
- 142.Kashiouris M.G., Lheureux M., Cable C.A., Fisher B.J., Leichtle S.W., Fowler A.A. The emerging role of vitamin C as a treatment for sepsis. Nutrients. 2020;12(2):1–16. doi: 10.3390/nu12020292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). 2020 Mar 12 – Identifier NCT04264533, vitamin c infusion for the treatment of severe 2019-ncov infected pneumonia. 2020. https://clinicaltrials.gov/ct2/show/NCT04264533?term=NCT04264533&draw=2&rank=1 (accessed 20 April 2020)
- 144.Long Y., Xu Y., Wang B., Zhang L., Jia D., Xue F., et al. Clinical recommendations from an observational study on MERS: glucocorticoids was benefit in treating SARS patients. Int. J. Clin. Exp. Med. 2016;9(5):8865–8873. [Google Scholar]
- 145.ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). 2020 Mar 12 – Identifier NCT04244591. Glucocorticoid therapy for novel coronavirus critically ill patients with severe acute respiratory failure (steroids-sari) 2020. https://clinicaltrials.gov/ct2/show/NCT04244591 (accessed 20 April 2020)
- 146.ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). 2020 Mar 12 – Identifier NCT04273763, Evaluating the efficacy and safety of bromhexine hydrochloride tablets combined with standard treatment/standard treatment in patients with suspected and mild coronavirus pneumonia (covid-19) 2020. https://clinicaltrials.gov/ct2/show/NCT04273763?term=NCT04273763&draw=2&rank=1 (accessed 21 April 2020)
- 147.Li C.C., Wang X.J., Wang H.C.R. Repurposing host-based therapeutics to control coronavirus and influenza virus. Drug Discov. Today. 2019;24(3):726–736. doi: 10.1016/j.drudis.2019.01.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Wang Y., Fei D., Vanderlaan M., Song A. Biological activity of bevacizumab, a humanized anti-VEGF antibody in vitro. Angiogenesis. 2004;7(4):335–345. doi: 10.1007/s10456-004-8272-2. [DOI] [PubMed] [Google Scholar]
- 149.ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). 2020 Mar 12 – Identifier NCT04275414, Bevacizumab in severe or critical patients with Covid-19 pneumonia (BEST-CP) 2020. https://clinicaltrials.gov/ct2/show/NCT04275414?term=NCT04275414&draw=2&rank=1 (accessed 25 April 2020)
- 150.ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). 2020 Mar 12 – Identifier NCT04280588, Fingolimod in COVID-19. 2020. https://clinicaltrials.gov/ct2/show/NCT04280588?term=NCT04280588&draw=2&rank=1 (accessed 25 April 2020)
- 151.Richardson P., Griffin I., Tucker C., Smith D., Oechsle O., Phelan A., et al. Baricitinib as potential treatment for 2019-nCoV acute respiratory disease. Lancet. 2020;395(10223) doi: 10.1016/S0140-6736(20)30304-4. e30-e1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Nowak J.K., Walkowiak J. Is lithium a potential treatment for the novel Wuhan (2019-nCoV) coronavirus? A scoping review. F1000research. 2020;9(93):93. doi: 10.12688/f1000research.22299.1. [DOI] [Google Scholar]
- 153.Sun M., Yang J., Sun Y., Su G. Inhibitors of RAS might be a good choice for the therapy of COVID-19 pneumonia. Chin. J. Tuberc. Respir. Dis. 2020;16(43):E014. doi: 10.3760/cma.j.issn.1001-0939.2020.0014. [DOI] [PubMed] [Google Scholar]
- 154.Fan H.H., Wang L.-Q., Liu W.-L., An X.-P., Liu Z.-D., He X.-Q., Song L.-H., Tong Y.-G. Repurposing of clinically approved drugs for treatment of coronavirus disease 2019 in a 2019-novel coronavirus (2019-nCoV) related coronavirus model. Chin. Med. J. 2020;133:1053–1056. doi: 10.1097/CM9.0000000000000797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Ren J.L., Zhang A.-H., Wang X.-J. Traditional Chinese medicine for COVID-19 treatment. Pharmacol. Res. 2020;155:104743. doi: 10.1016/j.phrs.2020.104743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). 2020 Mar 12 – Identifier NCT04282902, A Study to Evaluate the Efficacy and Safety of Pirfenidone with Novel Coronavirus Infection. 2020. https://clinicaltrials.gov/ct2/show/NCT04282902?term=NCT04282902&draw=2&rank=1 (accessed 20 April 2020)