Abstract
Background
Human granulocytic anaplasmosis (HGA) is a tick-borne infectious disease caused by Anaplasma phagocytophilum. To date, there have been no reported cases of A. phagocytophilum infection found in both the biting tick and the patient following a tick bite.
Case presentation
An 81-year-old woman presented with fever following a tick bite, with the tick still intact on her body. The patient was diagnosed with HGA. The tick was identified as Ixodes nipponensis by morphological and molecular biological detection methods targeting the 16S rRNA gene. The patient’s blood was cultured after inoculation into the human promyelocytic leukemia cell line HL-60. A. phagocytophilum growth was confirmed via culture and isolation. A. phagocytophilum was identified in both the tick and the patient’s blood by Anaplasma-specific groEL- and ankA-based nested polymerase chain reaction followed by sequencing. Moreover, a four-fold elevation in antibodies was observed in the patient’s blood.
Conclusion
We report a case of a patient diagnosed with HGA following admission for fever due to a tick bite. A. phagocytophilum was identified in both the tick and the patient, and A. phagocytophilum was successfully cultured. The present study suggests the need to investigate the possible incrimination of I. nipponensis as a vector for HGA in Korea.
Supplementary information
Supplementary information accompanies this paper at 10.1186/s12879-020-05522-5.
Keywords: Human granulocytic anaplasmosis, Tick bites, Anaplasma phagocytophilum, Ixodes nipponensis
Background
Human granulocytic anaplasmosis (HGA) is a tick-borne infectious disease caused by Anaplasma phagocytophilum, an obligate intracellular bacterium, which grows in membrane-bound vacuoles of humans and animals [1]. The annual incidence of this disease was reported to be 6.3 cases per million person-years between 2008 and 2012 in the United States [2]. Ixodes scapularis, a vector for Lyme disease or babesiosis, is known to be the primary vector of HGA in the United States, whereas Ixodes pacificus (western black-legged tick) and Ixodes ricinus (castor bean tick) are the presumed vectors across the western United States and Europe, respectively [3, 4]. In Korea, following the first report of anaplasmosis in 2013, HGA has been described as an emerging infectious disease [5]. However, to date, no studies have investigated the vectors of HGA in Korea.
We therefore performed molecular detection and isolation of A. phagocytophilum from the blood of a patient who presented with fever after a tick bite, with the tick still attached to the body.
Case presentation
Case
An 81-year-old woman was hospitalized with a chief complaint of fever. She developed fever, headache, and vomiting 5 days prior to admission and was treated conservatively at a local hospital. Three days prior to admission, her fever symptoms recurred and were accompanied by several vomiting episodes, abdominal pain, and shortness of breath.
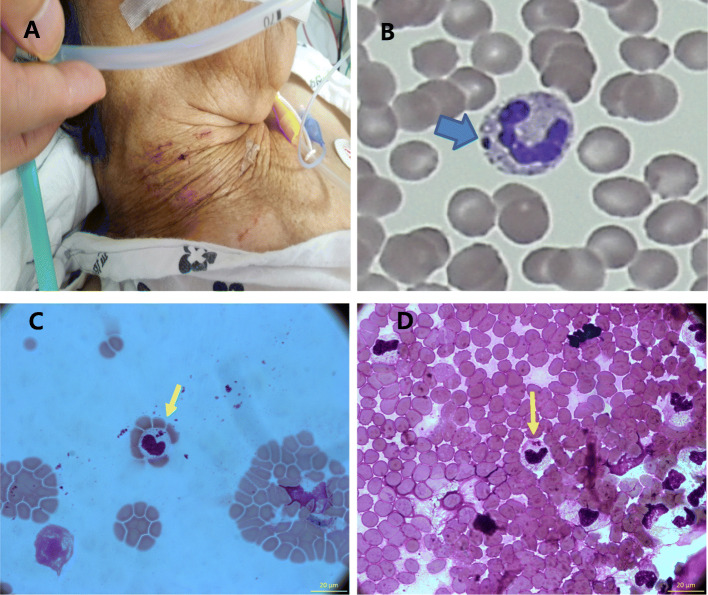
Consequently, the patient visited the emergency room of a local hospital and was misdiagnosed with cholecystitis, based on the results of abdominal computed tomography, and tenderness in her right upper quadrant on examination. She underwent laparoscopic cholecystectomy the next day and received antibiotic therapy; however, her fever persisted. Her guardian identified a mass-like lesion on the right side of her neck where an adhesive patch had been applied and notified the medical staff. Upon confirming the presence of a tick, the tick was removed (May 30). A single dose of 100 mg doxycycline was administered, and the patient (along with the tick) (Fig. 1a) was transferred to the Chosun University Hospital in Gwangju City, Korea (Fig. 2a). On admission (May 30), physical examination indicated that the patient was febrile, with a blood pressure of 90/60 mmHg and body temperature of 37.8 °C. She looked acutely ill on inspection, and her pulse and respiratory rates were 131 beats/min and 22 breaths/min, respectively. Skin examination confirmed a tick bite lesion on the right side of the neck (Fig. 2). Laboratory investigations further revealed the following findings: white blood cell count, 9,260/mm3 (91.6% polymorphonuclear leukocytes); hemoglobin level, 12.6 g/dL; platelet count, 24,000/mm3; aspartate aminotransferase level, 71.1 U/L; alanine aminotransferase level, 38.7 U/L; and creatine phosphokinase level, 255 U/L.
Fig. 1.
Gross findings of the tick removed from the right neck area of the patient. Dorsal view (a) and ventral view (b) of the tick. Identification of Ixodes nipponensis from the phylogenetic tree analysis based on the 16S rRNA gene of the tick of 367-bp amplicons produced from the conventional polymerase chain reaction (c)
Fig. 2.
Site of the tick bite (right neck area) (a). Suspected morulae in the peripheral blood smear (thick arrow) (b). Diff-Quik stained-peripheral blood smear showing neutrophils with intracellular inclusions (narrow arrows). Original magnification, 1000x (c, d)
The patient indicated that she was a resident of a rural area and lived close to a cattle farm with approximately 100 cows; the farm was separated from her house by only a single wall. The patient had a vegetable garden at her home, where she spent a considerable amount of time working almost every day both in the morning and evening. During the medical history interview on admission, the patient reported that she first noted discomfort in her right side of neck on April 22, 2017, which was prior to the initial appearance of symptoms. When her husband was asked to inspect the area, he mentioned that it appeared to be a wart. At that time, she developed a papular lesion, accompanied by itching and persistent discomfort, for which she began applying adhesive patches on May 6, 2017.
We suspected that the patient was bitten by the tick at least 1 or 2 days prior to showing the site to her husband on April 22. A. phagocytophilum was identified from the patient specimen through polymerase chain reaction (PCR). Subsequently, the patient was treated with 100 mg doxycycline twice daily for 10 days. The patient showed improvement in the C-reactive protein level and pancytopenia, normalization of liver function test results, and symptom improvement with treatment. She was discharged accordingly.
Tick identification
The developmental stage and species of the tick were morphologically identified by microscopy, using standard taxonomic keys (https://shire.science.uq.edu.au/parasites/arachnids/ticks/ticks-identification.php). After morphological identification, the tick was washed in 70% ethanol for 2–5 times, washed again in sterilized distilled water for 3 times, and dried by placing on a sterile filter paper. The tick was then placed in the MK28 hard tissue grinding tube (Bertin Technologies, Rockville, MD, USA), 600 μL of phosphate-buffered saline (10% fetal bovine serum, 5% penicillin/streptomycin) was added, and ground by using FastPrep®-24 Classic Instrument (MP Biomedicals, Solon, OH, USA). The genomic DNA of the tick was then extracted, using a G-spin Total DNA Extraction Kit (iNtRON Biotechnology, Seongnam, Korea), following the manufacturer’s protocol. Total RNA was isolated using the Viral Gene-spin™ Viral DNA/RNA Extraction Kit (iNtRON Biotechnology, Seongnam, Korea). For molecular biological identification of the tick, genomic DNA was subjected to the mitochondrial 16S rRNA gene-targeted PCR assay [6].
PCR detection for vector-borne infectious disease pathogens
PCR was performed on the tick lysate and the patient’s blood to detect for vector-borne infectious pathogens. DNA was extracted from the patient’s initial blood specimen and from the tick, using the QIAamp Blood and Tissue Mini Kit (Qiagen, Hilden, Germany), following the manufacturer’s instructions [7]. Anaplasma-specific ankA- and groEL-based nested PCRs (n-PCRs) were performed; the primers used for groEL were HS1/HS6 (for the first PCR) and HS43/HS45 (for the nPCR), while those for ankA were ANK-F1/ANK-R1 (for the first PCR) and ANK-F2/ANK-R2 (for the nPCR) [8, 9]. To detect Rickettsia spp., Rickettsia-specific ompA fragment was amplified using primers R190.70F/RR190.701R (for the first PCR) and R190.70F /RR190.602R (for the N-PCR) [10, 12]. For the detection of Borrelia spp., pyrG N-PCR was performed by amplifying the CTP synthase genes, using primers pyrG-1F/pyrG-1R (for the first PCR) and pyrG-2F/pyrG-2R (for the N-PCR) [13]. The gene encoding the Orientia tsutsugamushi 56-kDa antigen was amplified using the designed primers 56BO-144F/56BO-1395R (for the first PCR) and 56BO-406F/56BO-1088R (for the N-PCR). To assess for severe fever with thrombocytopenia syndrome virus RNA, we performed reverse transcription PCR, as previously described [11]. The genomic DNAs of A. phagocytophilum KZ_A3, Rickettsia conorii, Borrelia burgdorferi, and the O. tsutsugamushi Karp strain served as positive controls for Anaplasma-specific, Rickettsia-specific, Borrelia-specific and O. tsutsugamushi-specific targets, respectively. In each PCR run, the reaction mixture without template DNA served as the negative control. All primer sequences and PCR cycling conditions are shown in Table 1.
Table 1.
Conditions and primers used for PCR, amplified product base pair size, and references used for PCR in this study
| PCR assay | Name of primer seq (5′-3′) | PCR conditions | Product Size (bp) | Reference | |||
|---|---|---|---|---|---|---|---|
| Denaturation | Annealing | Extension | Cycles | ||||
| (°C/sec) | (°C/sec) | (°C/sec) | |||||
| 16 s rRNA PCR for tick identification | 16S - 1-F (CTGCTCAATGAATATTTAAATTGC) | 95/45 | 55/60 | 72/90 | 40 | 450 | (https://shire.science.uq.edu.au/parasites/arachnids/ticks/ticks-identification.php) |
| 16S-1-R (CGGTCTAAACTCAGATCATGTAGG) | |||||||
| ankA N-PCR for Anaplasma phagocytophilum | ANK-F1 (GAAGAAATTACAACTCCTGAAG) | 95/30 | 53/30 | 72/60 | 35 | 705 | [7] |
| ANK-R1 (CAGCCAGATGCAGTAACGTG) | |||||||
| ANK-F2 (TTGACCGCTGAAGCACTAAC) | 95/30 | 52/30 | 72/60 | 5 | 664 | ||
| ANK-R2 (ACCATTTGCTTCTTGAGGAG) | 95/30 | 54/30 | 72/60 | 25 | |||
| groEL N-PCR for Anaplasma phagocytophilum | HS1 TGGGCTGGTA(A/C)TGAAAT | 94/60 | 48/120 | 70/90 | 3 | 1300 | [8] |
| HS6 CCICCIGGIACIA(C/T)ACCTTC | 88/60 | 52/120 | 70/90 | 37 | |||
| HS43 AT(A/T)GC(A/T)AA(G/A)GAAGCATAGTC | 94/60 | 48/120 | 70/90 | 3 | 528 | ||
| HS45 ACTTCACG(C/T)(C/T)TCATAGAC | 88/60 | 55/120 | 70/90 | 37 | |||
| ompA N-PCR for Rickettsia spp. | R190.70F (ATGGCGAATATTTCTCCAAAAA) | 94/30 | 50/30 | 72/60 | 40 | 634 | [9] |
| RR190.701R (GTTCCGTTAATGGCAGCATCT) | |||||||
| R190.70F (ATGGCGAATATTTCTCCAAAAA) | 94/30 | 51/30 | 72/30 | 5 | 535 | ||
| RR190.602R (AGTGCAGCATTCGCTCCCCCT) | 94/30 | 55/30 | 72/30 | 30 | |||
| 56 kDa N-PCR for Orientia tsutsugamushi | 56BO-144F (YGYAGAATCTRCTCGCTTGG) | 94/60 | 60/60 | 72/60 | 35 | 1250 | In this study |
| 56BO-1395R (agctaMccctRcaccaaBac) | |||||||
| 56BO-406F (CCWCCTCARCCTACTAtrTGC) | 94/30 | 61/30 | 72/45 | 30 | 680 | ||
| 56BO-1088R (gcWgctgctRctgcttcttg) | |||||||
| pyrG N-PCR for Borrelia spp. | pyrG-1F (ATTGCAAGTTCTGAGAATA) | 94/20 | 45/30 | 72/30 | 30 | 801 | [10] |
| pyrg-1R (CAAACATTACGAGCAAATTC) | |||||||
| pyrG-2F (GATATGGAAAATATTTTATTTATTG) | 95/30 | 45/30 | 72/30 | 5 | 707 | ||
| pyrg-2R (AAACCAAGACAAATTCCAAG) | 95/30 | 47/30 | 72/30 | 5 | |||
| 95/30 | 49/30 | 72/30 | 25 | ||||
| RT-PCR for SFTSV | MF3 (GATGAGATGGTCCATGCTGATTCT) | 95/20 | 58/40 | 72/30 | 35 | 560 | [11] |
| MR2 (CTCATGGGGTGGAATGTCCTCAC) | |||||||
Positive PCR products from the patient’s blood and the tick were sequenced and aligned to the GenBank database to identify revealed bacterial agents at the species level and to compare our sequences with published sequences using BLAST analysis and ClustalW alignment (www.clustal.org).
Phylogenetic analysis
DNA sequences were identified and analyzed using DNASTAR Lasergene v6 (DNASTAR, Madison, WI, USA). A phylogenetic tree was constructed by the neighbor-joining method using ClustalX version 2.0 (www.clustal.org/), based on sequences of the amplified Anaplasma-specific groEL and ankA gene fragments from the specimen and from GenBank. A bootstrap analysis was performed using 1000 replicates to improve the confidence level of the phylogenetic tree.
Peripheral blood smear
A thick peripheral blood smear was examined for the presence of A. phagocytophilum in the neutrophils. A drop of the patient’s blood was smeared onto a glass slide, air-dried, and stained with Diff-Quik solution (Sysmex Corporation, Kobe, Japan), following the standard procedure. The smear was observed under a microscope at a magnification of 1000x.
A. phagocytophilum culture and isolation
For A. phagocytophilum culture and isolation, the human promyelocytic leukemia cell line HL-60 (KCLB-10240) was maintained in RPMI1640 medium (Gibco, Thermo Fisher Scientific, MA USA), supplemented with 2% fetal bovine serum (Gibco, Thermo Fisher Scientific, MA USA) and 2 mM L-glutamine at 37 °C and under 5% CO2 conditions. The buffy coat or tick lysate solution was inoculated into HL-60 cells. Infected cells were cultured under the same conditions as above, with regular medium changes (cell density of 1–5 × 105 cells/mL). They were then stained with Diff-Quik at 2–3-day intervals and were subsequently cytocentrifuged for microscopic examination.
Indirect immunofluorescence antibody assay
An indirect immunofluorescence assay (IFA) was performed, following the standard procedure and/or manufacturer’s instructions (Fuller Laboratories, Fullerton, CA, USA) [14]. In-house IFA commenced with antigen fixation of the heavily-infected HL-60 cells onto Teflon-coated slides with acetone. An anti-A. phagocytophilum serum (Fuller Laboratories, Fullerton, CA, USA) was then added to the antigen slides, and the slides were incubated in a humidity chamber at 37 °C for 30 min. DyLight 488-labeled goat anti-human IgG or IgM (Fuller Laboratories, Fullerton, CA, USA) was used as a secondary antibody under the same conditions. The antigen slides were counterstained with 0.005% Evans blue and mounted for fluorescence microscopy. Manufacturer’s slides containing infected HL-60 cells (Fuller Laboratories, Fullerton, CA, USA) and the human serum were used as positive and negative controls, respectively. For the serological diagnosis of scrub typhus, murine typhus, and Lyme disease, IgM and IgG antibodies against the standard O. tsutsugamushi antigen (Gilliam, Karp, Kato, and Boryong strains), Rickettsia typhi, and B. burgdorferi were assessed using in-house IFA [15].
Ultrastructural analysis of A. phagocytophilum
To investigate A. phagocytophilum infection, the cytoplasmic area of occupied vacuoles was explored by electron microscopy at the ultrastructural level. The cultured cells were stained with Diff-Quik at 2–3-day intervals, cytocentrifuged to confirm the infection level, and then examined, using scanning and transmission electron microscopy (SEM and TEM, respectively).
Results
Tick identification
Morphological analysis confirmed that the tick was a female adult I. nipponensis (Fig. 1a, b). This was further confirmed by molecular identification and phylogenetic tree analysis of the 367-bp 16S rRNA amplicons produced using conventional PCR (C-PCR) followed by sequencing (Fig. 1c).
A. phagocytophilum detection
On initial peripheral blood smear analysis, the Wright–Giemsa-stain showed findings suggestive of morulae (Fig. 2b), whereas Diff-Quik confirmed this by revealing intracytoplasmic inclusion bodies in the neutrophils (Fig. 2c, d). The n-PCR was performed on the patient’s buffy coat and the tick by using the 347-bp amplicons of A. phagocytophilum-specific primers targeting groEL. Sequence alignment indicated 100% homology between the patient and the tick (NCBI accession no. MH492313, MH492314), which in turn shared 99% homology with A. phagocytophilum isolates D-SE-63 (accession no. KU519286) and S-DD-21 (accession no. KU519285), previously identified in dogs and cats, respectively, in Korea. Sequences were cut to a size of 335-bp for the phylogenetic tree analysis (Fig. 3a) and were shown to form clusters with A. phagocytophilum strain D-SE-63 (accession no. KU519286), identified in dogs in Korea, Anaplasma spp. (accession no. JQ622144), identified in a Japanese tick, and A. phagocytophilum strain Kh-Ip144 (accession no. HM366577), identified in a Russian tick (66 bootstraps).
Fig. 3.
Phylogenetic tree analysis, following groEL and ankA gene-targeted PCR of the patient’s buffy coat at the time of admission and of the tick (a, groEL; b, ankA)
In terms of the ankA gene sequence, sequence alignment indicated 100% homology between the patient and the tick (NCBI accession no. MH492315, MH492316), which also shared 100% homology with A. phagocytophilum isolates KZA1 (accession no. KT986059) and gw1 (accession no. KJ677106). Sequences were cut to 837-bp for the phylogenetic tree analysis (Fig. 3b) and further confirmed cluster formation with A. phagocytophilum KZA1 (accession no. KT986059) and gw1 (accession no. KJ677106), originally identified in Korean patients (100 bootstraps).
A. phagocytophilum culture using the Patient’s blood
Cytoplasmic inclusions in the infected cells were observed 32 days post-inoculation of HL-60 cells with A. phagocytophilum isolated from the patient’s buffy coat (Supplementary Figure 1A–D). IFA involving both IgM and IgG antibodies from the antiserum positively reacted with A. phagocytophilum, which have propagated in the HL-60 cells, with fluorescent morulae filled with bacteria observed surrounding the cell’s cytoplasmic membrane (Supplementary Figure 1E, F). The nPCR and direct sequencing of the isolated bacteria provided positive results and confirmed the presence of the same A. phagocytophilum bacterial strain identified in the patient’s blood (named A. phagocytophilum KZ_A3).
A. phagocytophilum ultra-structural studies
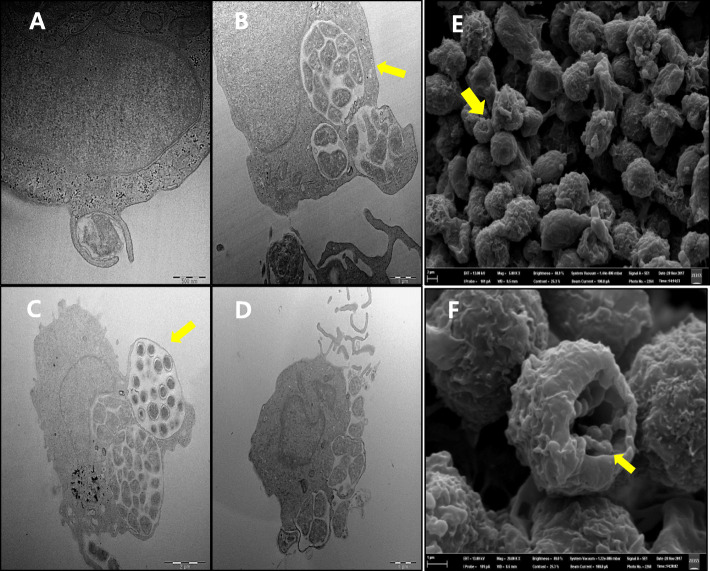
The infection stages adhesion, replication, invasion, and release of isolated A. phagocytophilum KZ_A3 were visualized within HL-60 cells using TEM (Fig. 4a). Reticulate cells were characterized by dispersed nucleoids (Fig. 4b), a smoother outer membrane than that observed with dense-cored cells (Fig. 4c), and pleomorphism (Fig. 4a–d). Infected cells were further examined by SEM at the ultrastructure level (Fig. 4e, f), which showed the replication of A. phagocytophilum KZ_A3 within large vesicles of the cell, with a grape-like cluster appearance. Ruptured vesicles were also observed, revealing a cluster of A. phagocytophilum (Fig. 4f). Infectious dense-cored cells were predominantly coccoid bacteria at the late stages of infection (Fig. 4e, f).
Fig. 4.
Transmission electron microscopy showing the infection stages of isolated A. phagocytophilum KZ_A3-infected HL-60 cells. Adhesion, replication, invasion, and release stages of the isolated A. phagocytophilum within HL-60 cells (a-d). Representative images of dense-cored cells and reticulate cells; the large arrow indicates a dense nucleoid and a ruffled outer membrane, and the spheroid indicates individual dense-cored cells surrounded by a membranous projection (c). Scanning electron micrograph of a cluster of isolated A. phagocytophilum KZ_A3 within HL-60 cells (e, f) A. phagocytophilum KZ_A3 were observed to replicate in a large vesicle inside the cell. Here, the vesicle has ruptured, revealing A. phagocytophilum (c–d, 20000x)
A. phagocytophilum culture with tick lysates
HL-60 cell culture at 10 days post-inoculation with Anaplasma spp. isolated from the tick lysate showed unexpected microbial growth, which was later observed in other cell culture assays (Supplementary Figure 2). The intracellular morphology was distinct from that of Anaplasma spp., and was similar to that of Rickettsia spp. With Diff-Quik staining, the unexpected bacteria replicated faster than Anaplasma spp.; however, Anaplasma spp. was detected in infected cells using in-house IFA, and fluorescent bacteria were observed following incubation with an A. phagocytophilum antiserum. This prompted a re-evaluation of the infection by this agent (Supplementary Figure 2).
PCR studies for other targets
PCR with Rickettsia genus-specific primers of 435-bp amplicons targeting the outer membrane protein A (ompA) was performed on the tick. Sequence alignment indicated a 100% identity to the R. monacensis strain MT34 (accession no. JX972178) isolated from a Korean tick. We confirmed that the unexpected intracellular bacteria, detected during tick lysate culturing, were R. monacensis. Although Anaplasma spp. was detected during tick lysate culture, its isolation was unsuccessful, possibly due to coinfection. O. tsutsugamushi, R. typhi, and Borrelia spp. were not found in the tick specimen.
Confirmation of infection using IFA studies
A 4-fold or greater rise in titer between acute and convalescent sera represented evidence for infection. IFA results of the patient serum showed that both IgM and IgG antibodies to A. phagocytophilum were negative at the time of admission (5 days after symptom onset). The levels of IgG (1:160) and IgM (1:64) antibodies increased 10 days after symptoms onset; similarly, the levels of antibodies to O. tsutsugamushi and R. typhi were also elevated. However, when the patient’s admission blood specimen was used for ompA-targeted PCR to detect R. typhi and R. monacensis and for PCR to detect O. tsutsugamushi and Borrelia spp., all results were negative (Table 2).
Table 2.
PCR and antibody test results of the patient
| Sample collection day | PCR | IFA | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
ankA nPCR groEL nPCR |
ompA | 56-kDa nPCR |
SFTS C-PCR |
Orientia tsutsugamushi |
Rickettsia typhi |
Anaplasma phagocytophilum | Borrelia burgdorferi | |||||
| IgG | IgM | IgG | IgM | IgG | IgM | IgG | IgM | |||||
| 2017-05-30 | + | – | – | – | 1:2048 | < 1:16 | 1:64 | < 1:16 | < 1:80 | < 1:16 | < 1:16 | < 1:16 |
| 2017-06-02 | 1:1024 | < 1:16 | 1:64 | < 1:16 | ||||||||
| 2017-06-05 | – | – | 1:160 | 1:64 | 1:16 | < 1:16 | ||||||
| 2017-06-07 | 1:512 | < 1:16 | 1:64 | < 1:16 | ||||||||
| 2017-06-20 | – | – | 1:256 | < 1:16 | 1:512 | < 1:16 | 1:640 | 1:64 | 1:32 | < 1:16 | ||
| 2017-06-26 | – | – | 1:512 | < 1:16 | 1:512 | < 1:16 | 1:640 | 1:256 | 1:16 | < 1:16 | ||
| 2017-07-03 | 1:512 | 1:16 | 1:512 | < 1:16 | 1:640 | 1:64 |
1:16 WB+ |
1:16 WB+ |
||||
| 2017-07-20 | 1:512 | < 1:16 | 1:512 | < 1:16 | 1:640 | 1:512 |
1:32 WB+ |
1:16 WB+ |
||||
| 2017-08-04 | 1:512 | < 1:16 | 1:512 | < 1:16 | 1:640 | 1:256 |
1:16 WB+ |
1:16 WB+ |
||||
PCR polymerase chain reaction; IFA indirect immunofluorescence assay; nPCR nested PCR; SFTS severe fever with thrombocytopenia syndrome; C-PCR conventional PCR; WB western blot (Positive means more than 7 antigen bands were detected using the Microgen diagnostic kit)
Discussion and conclusions
A. phagocytophilum is transmitted by I. scapularis in New England and the north-central United States, I. pacificus in the western United States, I. ricinus in Europe, and I. persulcatus in Asia [16]. In Japan, which is in close proximity to Korea, A. phagocytophilum has been identified in I. persulcatus and I. ovatus and was first reported in 2005 [17]. Furthermore, 4.6% of I. persulcatus isolated from northern China was reported to have tested positive for A. phagocytophilum [18]. Kim et al. [19] reported that among 1467 ticks (1463 Haemaphysalis longicornis, 3 I. persulcatus, and 1 I. turdus) collected from 9 Korean provinces, 35 H. longicornis and 1 I. persulcatus were confirmed to be positive for A. phagocytophilum, whereas 1 I. persulcatus tested positive for Ehrlichia chaffeensis.
In a molecular detection study of Anaplasma spp. infection rates in ticks collected from migratory birds on Hongdo Island, Korea, A. phagocytophilum was detected in only 1 I. nipponensis nymph, among a total of 212 ticks (40 Haemaphysalis flava, 12 H. longicornis, 146 I. turdus, 13 I. nipponensis, and 1 I. ornithophilia) [20]. Thus, it is speculated that H. longicornis, I. persulcatus, and I. nipponensis may act as anaplasmosis vectors in Korea. A. phagocytophilum has previously been identified in three tick species (H. longicornis, I. persulcatus, and I. nipponensis) in Korea, but no direct transmission from ticks to humans has been confirmed.
Since the first report on the serological and molecular detection of HGA in Korea in 2002, numerous studies on A. phagocytophilum vectors have been performed [21] However, there are no reported cases that have characterized the species of the biting tick or that have detected the presence of an identical A. phagocytophilum strain in both the biting tick and the patient, particularly with a tick that was intact on the patient’s body. In the present study, the identical A. phagocytophilum strain was identified in not only the patient who was diagnosed with HGA following admission for fever symptoms after a tick bite but also in the tick itself. Moreover, A. phagocytophilum was successfully cultured from both the patient’s blood and the biting tick,
Follow-up antibody test results of the tick DNA sample showed elevated antibodies not only for A. phagocytophilum but also for O. tsutsugamushi, R. typhi, B. burgdorferi and R. monacensis monacensis. Therefore, the possibility of coinfection or sequential infection could not be dismissed.
In our previous study on 317 patients with scrub typhus, 96.2% showed elevated IgM antibodies (1:10) within 1 month of symptom onset, whereas 67.4% continued to test positive for IgM antibodies at the 6-month follow-up test [22]. A recent infection would have correlated with elevated levels of IgM antibodies; however, this was not observed in the present case. Moreover, all PCR results on the patient’s blood and the tick were negative for O. tsutsugamushi, R. typhi, and B. burgdorferi. Thus, we believed that the probability of a past infection, sequential infection, or antibody cross-reactivity may be higher than that of a coinfection. Similarly, the cross-reactivity between E. canis and A. phagocytophilum has been reported previously in experimental animal [23]. However, additional studies are needed specifically to clarify the cross-reactivity between Anaplasmosis spp. and B. burgdorferi.
The studies showed that the full engorgement of ticks require 7 to12 days of attachment on the host before shedding off to continue with their life cycle. However, it has also been suggested that some ticks may require 30 days or longer [5]. Nonetheless, the literature has shown that the incubation period for human anaplasmosis ranges from 5 to 21 days [24].
A limitation of this study was that the analyzed tick specimens were completely engorged by the blood of the infected woman, and the positivity to A. phagocytophilum observed in the DNA of the tick specimen could have come from the patient’s blood and not from the salivary glands of the tick. Therefore, we could not confirm that I. nipponensis is a vector for A. phagocytophilum. Furthermore, since the patient came from an area with a high risk of tick bites, we could not eliminate the likelihood of undetectable tick bites at other sites of her body.
Conclusion
In conclusion, we presented a case involving a patient diagnosed with HGA following admission for fever due to a tick bite. An identical A. phagocytophilum strain was identified in both the patient and the tick, and A. phagocytophilum was successfully cultured from the patient’s blood. Nevertheless, further studies are warranted to determine whether I. nipponensis can act as a possible vector for anaplasmosis in Korea.
Supplementary information
Additional file 1 Supplement 1. Morulae of Anaplasma phagocytophilum. Light micrograph of A. phagocytophilum cultured in a human promyelocytic cell line (A: dpi32, B: dpi37, C: dpi39, D: Cell passage 2). Diff-Quik staining (A–D). The arrow indicates A. phagocytophilum KZ_A3. Original magnification (A-D; 400x) of in-house immunofluorescence staining of isolated A. phagocytophilum from the patient within the human promyelocytic cell line (dpi 37). Culture preparations stained by IFA using an anti-A. phagocytophilum serum. The arrow indicates intracytoplasmic inclusions filled with numerous bacteria. Fluorescence magnification (E–F; 400x). IFA of A. phagocytophilum KZ_A3 in infected HL-60 cells (37 dpi). The cells were treated in turn, with antiserum and anti-human IgG (E) or IgM (F) conjugate to detect A. phagocytophilum. The yellow arrows indicate intracytoplasmic inclusions filled with numerous bacteria (400x magnification). Supplement 2. Morulae, suspected of A. phagocytophilum. Light micrograph of A. phagocytophilum cultured in a human promyelocytic cell line, using the tick lysate (A: dpi10, B: dpi10). Diff-Quik staining (A, B). The arrow indicates A. phagocytophilum KZ_A3. Original magnification (A; 400x, B; 1000x) of in-house immunofluorescence staining of the A. phagocytophilum infected tick lysate solution within the human promyelocytic cell line (day 10). Culture preparations were stained by IFA using an anti-A. phagocytophilum serum. The arrow indicates intracytoplasmic inclusions filled with numerous bacteria. Fluorescence magnification (C–D; 400x).
Additional file 2. The same description has been provided as a word file, as per the journal requirements.
Acknowledgments
We thank our collaborator Kim Jee-woong, from the Division of Evaluation and Control, for performing the electron microscope analysis.
Abbreviations
- HGA
Human granulocytic anaplasmosis
- PCR
Polymerase chain reaction
- IFA
Indirect immunofluorescent assay
- SEM
Scanning electron microscopy
- TEM
Transmission electron microscopy
Authors’ contributions
NRY and DMK contributed to the management of the patient. DMK was the leader of the clinical team. SHL and NRS conducted the literature review and wrote the manuscript. DMK, DSJ, and SDP revised the article. CMK contributed to molecular identification. SHL, NRS, CMK, and SDP contributed to isolation and identification of the pathogen, the exclusion diagnosis, and confirmation of the final diagnosis. All authors read and approved the final manuscript.
Funding
This study was supported by research grants from the Korea Centers for Disease Control and Prevention (KCDC) (2017-NI52002–00) and Korean Health Technology Research & Development Project, Ministry of Health & Welfare, Republic of Korea (HI16C2118).
Availability of data and materials
The datasets analyzed during the current study are available at National Center for Biotechnology Information (NCBI) repository. (Accession numbers; MH492313, MH492314, MH492315, MH492316, KU519285, KU519286, JQ622144, HM366577, KT986059, KJ677106, KT986059, KJ677106, JX972178).
Ethics approval and consent to participate
The study was approved by the Ethics in Human Research Committee of Chosun University Hospital (IRB No. 2013–10–001-018). The patient provided written informed consent to participate in the study.
Consent for publication
Written informed consent was obtained from the patient for publication of this case report and any accompanying images. A copy of the written consent is available for review by the Editor of this journal upon request.
Competing interests
The authors do not have any commercial interests or other associations, which might pose a conflict of interest.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Sungdo Park, Email: sdpark09@korea.kr.
Dong-Min Kim, Email: drongkim@chosun.ac.kr.
References
- 1.Kim CM, Kim SW, Kim DM, Yoon NR, Jha P, Jang SJ, et al. Case report: polymerase chain reaction testing of tick bite site samples for the diagnosis of human granulocytic anaplasmosis. Am J Trop Med Hyg. 2017;97:403–406. doi: 10.4269/ajtmh.16-0570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Dahlgren FS, Heitman KN, Drexler NA, Massung RF, Behravesh CB. Human granulocytic anaplasmosis in the United States from 2008 to 2012: a summary of national surveillance data. Am J Trop Med Hyg. 2015;93:66–72. doi: 10.4269/ajtmh.15-0122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bakken JS, Dumler JS. Human granulocytic ehrlichiosis. Clin Infect Dis. 2000;31:554–560. doi: 10.1086/313948. [DOI] [PubMed] [Google Scholar]
- 4.Sormunen JJ, Penttinen R, Klemola T, Vesterinen EJ, Hanninen J. Anaplasma phagocytophilum in questing Ixodes ricinus ticks in southwestern Finland. Exp Appl Acarol. 2016;70:491–500. doi: 10.1007/s10493-016-0093-7. [DOI] [PubMed] [Google Scholar]
- 5.Kim KH, Yi J, Oh WS, Kim NH, Choi SJ, Choe PG, et al. Human granulocytic anaplasmosis, South Korea, 2013. Emerg Infect Dis. 2014;20:1708–1711. doi: 10.3201/eid2010.131680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Tian Z, Liu G, Xie J, Yin H, Luo J, Zhang L, et al. Discrimination between Haemaphysalis longicornis and H. qinghaiensis based on the partial 16S rDNA and the second internal transcribed spacer (ITS-2) Exp Appl Acarol. 2011;54:165–172. doi: 10.1007/s10493-010-9423-3. [DOI] [PubMed] [Google Scholar]
- 7.Kim DM, Kim HL, Park CY, Yang TY, Lee JH, Yang JT, et al. Clinical usefulness of eschar polymerase chain reaction for the diagnosis of scrub typhus: a prospective study. Clin Infect Dis. 2006;43:1296–1300. doi: 10.1086/508464. [DOI] [PubMed] [Google Scholar]
- 8.Massung RF, Levin ML, Munderloh UG, Silverman DJ, Lynch MJ, Gaywee JK, et al. Isolation and propagation of the Ap-variant 1 strain of Anaplasma phagocytophilum in a tick cell line. J Clin Microbiol. 2007;45:2138–2143. doi: 10.1128/JCM.00478-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Sumner JW, Nicholson WL, Massung RF. PCR amplification and comparison of nucleotide sequences from the groESL heat shock operon of Ehrlichia species. J Clin Microbiol. 1997;35:2087–2092. doi: 10.1128/JCM.35.8.2087-2092.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Regnery RL, Spruill CL, Plikaytis BD. Genotypic identification of rickettsiae and estimation of intraspecies sequence divergence for portions of two rickettsial genes. J Bacteriol. 1991;173:1576–1589. doi: 10.1128/JB.173.5.1576-1589.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Yun SM, Lee WG, Ryou J, Yang SC, Park SW, Roh JY, et al. Severe fever with thrombocytopenia syndrome virus in ticks collected from humans, South Korea, 2013. Emerg Infect Dis. 2014;20:1358–1361. doi: 10.3201/eid2008.131857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Roux V, Fournier PE, Raoult D. Differentiation of spotted fever group rickettsiae by sequencing and analysis of restriction fragment length polymorphism of PCR-amplified DNA of the gene encoding the protein rOmpA. J Clin Microbiol. 1996;34:2058–2065. doi: 10.1128/JCM.34.9.2058-2065.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sapi E, Pabbati N, Datar A, Davies EM, Rattelle A, Kuo BA. Improved culture conditions for the growth and detection of Borrelia from human serum. Int J Med Sci. 2013;10:362–376. doi: 10.7150/ijms.5698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Eremeeva ME, Balayeva NM, Raoult D. Serological response of patients suffering from primary and recrudescent typhus: comparison of complement fixation reaction, Weil-Felix test, microimmunofluorescence, and immunoblotting. Clin Diagn Lab Immunol. 1994;1:318–324. doi: 10.1128/CDLI.1.3.318-324.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Robinson DM, Brown G, Gan E, Huxsoll DL. Adaptation of a microimmunofluorescence test to the study of human Rickettsia tsutsugamuskh antibody. Am J Trop Med Hyg. 1976;25:900–905. doi: 10.4269/ajtmh.1976.25.900. [DOI] [PubMed] [Google Scholar]
- 16.Ismail N, Bloch KC, McBride JW. Human ehrlichiosis and anaplasmosis. Clin Lab Med. 2010;30:261–292. doi: 10.1016/j.cll.2009.10.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ohashi N, Inayoshi M, Kitamura K, Kawamori F, Kawaguchi D, Nishimura Y, et al. Anaplasma phagocytophilum-infected ticks, Japan. Emerg Infect Dis. 2005;11:1780–1783. doi: 10.3201/eid1111.050407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Cao WC, Zhao QM, Zhang PH, Yang H, Wu XM, Wen BH, et al. Prevalence of Anaplasma phagocytophila and Borrelia burgdorferi in Ixodes persulcatus ticks from northeastern China. Am J Trop Med Hyg. 2003;68:547–550. doi: 10.4269/ajtmh.2003.68.547. [DOI] [PubMed] [Google Scholar]
- 19.Kim CM, Kim MS, Park MS, Park JH, Chae JS. Identification of Ehrlichia chaffeensis, Anaplasma phagocytophilum, and A. bovis in Haemaphysalis longicornis and Ixodes persulcatus ticks from Korea. Vector Borne Zoonotic Dis. 2003;3:17–26. doi: 10.1089/153036603765627424. [DOI] [PubMed] [Google Scholar]
- 20.Kang JG, Kim HC, Choi CY, Nam HY, Chae HY, Chong ST, et al. Molecular detection of Anaplasma, Bartonella, and Borrelia species in ticks collected from migratory birds from Hong-Do Island, Republic of Korea. Vector Borne Zoonotic Dis. 2013;13:215–225. doi: 10.1089/vbz.2012.1149. [DOI] [PubMed] [Google Scholar]
- 21.Heo EJ, Park JH, Koo JR, Park MS, Park MY, Dumler JS, et al. Serologic and molecular detection of Ehrlichia chaffeensis and Anaplasma phagocytophila (human granulocytic ehrlichiosis agent) in Korean patients. J Clin Microbiol. 2002;40:3082–3085. doi: 10.1128/JCM.40.8.3082-3085.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kim DM, Lee YM, Back JH, Yang TY, Lee JH, Song HJ, et al. Park MY. A serosurvey of Orientia tsutsugamushi from patients with scrub typhus. Clin Microbiol Infect. 2010;16:447–451. doi: 10.1111/j.1469-0691.2009.02865.x. [DOI] [PubMed] [Google Scholar]
- 23.Krämer F, Schaper R, Schunack B, Połozowski A, Piekarska J, Szwedko A, Jodies R, Kowalska D, Schüpbach D, Pantchev N. Serological detection of Anaplasma phagocytophilum, Borrelia burgdorferi sensu lato and Ehrlichia canis antibodies and Dirofilaria immitis antigen in a countrywide survey in dogs in Poland. Parasitol Res. 2014;113(9):3229–3239. doi: 10.1007/s00436-014-3985-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Brouqui P, Bacellar F, Baranton G, Birtles RJ, Bjoersdorff A, Blanco JR, Caruso G, Cinco M, Fournier PE, Francavilla E, Jensenius M. Guidelines for the diagnosis of tick-borne bacterial diseases in Europe. Clin Microbiol Infect. 2004;10(12):1108–1132. doi: 10.1111/j.1469-0691.2004.01019.x. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1 Supplement 1. Morulae of Anaplasma phagocytophilum. Light micrograph of A. phagocytophilum cultured in a human promyelocytic cell line (A: dpi32, B: dpi37, C: dpi39, D: Cell passage 2). Diff-Quik staining (A–D). The arrow indicates A. phagocytophilum KZ_A3. Original magnification (A-D; 400x) of in-house immunofluorescence staining of isolated A. phagocytophilum from the patient within the human promyelocytic cell line (dpi 37). Culture preparations stained by IFA using an anti-A. phagocytophilum serum. The arrow indicates intracytoplasmic inclusions filled with numerous bacteria. Fluorescence magnification (E–F; 400x). IFA of A. phagocytophilum KZ_A3 in infected HL-60 cells (37 dpi). The cells were treated in turn, with antiserum and anti-human IgG (E) or IgM (F) conjugate to detect A. phagocytophilum. The yellow arrows indicate intracytoplasmic inclusions filled with numerous bacteria (400x magnification). Supplement 2. Morulae, suspected of A. phagocytophilum. Light micrograph of A. phagocytophilum cultured in a human promyelocytic cell line, using the tick lysate (A: dpi10, B: dpi10). Diff-Quik staining (A, B). The arrow indicates A. phagocytophilum KZ_A3. Original magnification (A; 400x, B; 1000x) of in-house immunofluorescence staining of the A. phagocytophilum infected tick lysate solution within the human promyelocytic cell line (day 10). Culture preparations were stained by IFA using an anti-A. phagocytophilum serum. The arrow indicates intracytoplasmic inclusions filled with numerous bacteria. Fluorescence magnification (C–D; 400x).
Additional file 2. The same description has been provided as a word file, as per the journal requirements.
Data Availability Statement
The datasets analyzed during the current study are available at National Center for Biotechnology Information (NCBI) repository. (Accession numbers; MH492313, MH492314, MH492315, MH492316, KU519285, KU519286, JQ622144, HM366577, KT986059, KJ677106, KT986059, KJ677106, JX972178).