Abstract
Diabetes mellitus and cancer are diseases of epidemic proportions across the globe. These diseases are influenced by many factors, both genetic and environmental. A possible association between diabetes and cancer risk has long been speculated. Increased incidence of several cancers has been observed in diabetes patients, notably pancreatic, hepatic, colorectal, breast, urinary tract, and endometrial cancers. In contrast, a decreased incidence of prostate cancer is observed in diabetes patients, implying a protective effect. Precise knowledge of the complex associations and interactions between these two conditions is of great importance for their prevention and treatment. Multiple potential mechanisms have been proposed, but they have tended to be site‐specific. Possible common mechanisms for a biological link between diabetes and cancer include hyperinsulinemia, hyperglycemia, and inflammation. Today, 366 million people live with diabetes globally, and this figure is expected to increase. Thus, if diabetes is associated with even a small increase in cancer risk, this may have important consequences at the population level. The aim of this review is to summarize recent epidemiological evidence of an association between diabetes and total cancer and specific sites of cancer, and to consider causal associations between these diseases. (Cancer Sci 2013; 104: 9–14)
Diabetes mellitus is now a common and serious global health problem. It has grown in conjunction with rapid social and cultural changes, such as the aging of populations, urbanization, dietary changes, reduced physical activity, and other unhealthy behaviors, and is associated with various complications that decrease the quality of life and the survival of affected individuals.1 Today, 366 million people have diabetes worldwide, and the number is predicted to reach 552 million by 2030.2 Japan reflects this situation. The National Diabetes Survey in Japan showed that the prevalence of diabetes increased from 8.2% in 1997 to 10.5% in 2007.3 In our Japanese community‐based survey, the prevalence of glucose intolerance increased from 33.9% in 1988 to 43.5% in 2004.4 This trend is presumably attributable to the rapid Westernization of lifestyles in Japan, and is likely shared by the majority of East Asian populations.5
Growing evidence suggests that glucose metabolism abnormalities can represent an independent risk factor for the development of specific cancers and can affect their prognosis.6, 7 Several epidemiological studies have found that diabetes patients have an increased incidence of several types of cancers, such as liver,8 biliary tract,9 pancreas,10 stomach,11 colorectum,12 kidney,13 bladder,14 breast,15 and endometrium.16 In contrast, a reduced incidence of prostate cancer17 has been reported in individuals with diabetes (Table 1). However, the causal mechanisms involved in the association between diabetes and cancer are not clear. It has been reported that some metabolic alterations, such as hyperinsulinemia, insulin resistance, and aberrant production of growth hormone can induce carcinogenesis. Additionally, several potential confounders (e.g. obesity, medications, and diet) may contribute to this association. In the present review, we discuss the epidemiological evidence of an association between diabetes and cancer as well as the mechanisms underlying the association.
Table 1.
Recent meta‐analyses of diabetes mellitus and risk of specific site of cancer
| Cancer site | Author (year) | Studies | No. of cases/subjects,a | Summary RR (95% CI) |
|---|---|---|---|---|
| Liver | Wang et al. (2012)8 | 18 cohort studies | 18 258/3 626 369 | 2.01 (1.61–2.51) |
| Biliary tract | Ren et al. (2011)9 | 8 case–control studies and 13 cohort studies |
1836/165 861a 2595/5 494 975 |
1.43 (1.18–1.72), all studies 1.63 (1.07–2.47), case–control studies 1.49 (1.19–1.86), cohort studies |
| Pancreas | Ben et al. (2011)10 | 35 cohort studies | 20 410/21 616 592 | 1.94 (1.66–2.27) |
| Stomach | Ge et al. (2011)11 | 4 case–control studies and 17 cohort studies |
3211/60 731a 12 759/8 036 686 |
Women,: 1.18 (1.01–1.39), all studies 0.97 (0.64–1.46), case–control studies 1.12 (0.99–1.22), cohort studies Men,: not significant |
| Colorectum | Jiang et al. (2011)12 | 30 cohort studies | 61 690/8 201 654 | 1.27 (1.21–1.34) |
| Kidney | Larsson et al. (2011)13 | 9 cohort studies | 9520/5 769 987 | 1.42 (1.06–1.91) |
| Bladder | Larsson et al. (2006)14 | 7 case–control studies, 3 cohort studies, and 6 cohort of diabetic patients |
4427/12 189a 1619/1 923 472 2965/196 637 |
1.24 (1.08–1.42), all studies 1.37 (1.04–1.80), case–control studies 1.43 (1.18–1.74), cohort studies 1.01 (0.91–1.12), cohort studies with diabetic patients |
| Breast | Larsson SC et al. 200715 | 5 case–control studies and 15 cohort studies |
14 374/14 762a 16 483/1 400 822 |
1.20 (1.12–1.28), all studies 1.18 (1.05–1.32), case–control studies 1.20 (1.11–1.30), cohort studies |
| Endometrium | Friberg et al. (2007)16 | 13 case–control studies and 3 cohort studies |
6912/17 689a 684/71 402 |
2.10 (1.75–2.53), all studies 2.22 (1.80–2.74), case–control studies 1.62 (1.21–2.16), cohort studies |
| Prostate | Kasper et al. (2006)17 | 7 case–control studies and 12 cohort studies |
2481/4726a 16 610/1 054 354 |
0.84 (0.76–0.93), all studies 0.89 (0.72–1.11), case–control studies 0.81 (0.71–0.92), cohort studies |
Number of cases/controls in case–control studies. CI, confidential interval, RR, relative risk.
Epidemiological Evidence of Increased Risk of Cancer in Patients with Diabetes
Total cancer
Recently, numerous epidemiological studies have shown an association between diabetes and the risk of developing cancer.18, 19, 20, 21, 22, 23 During a 10‐year follow‐up of a cohort of 7148 patients with type 2 diabetes in Italy, a 16% (95% confidence interval [CI], 2–30%) increase in mortality from total cancer was observed in women, but no significant increase was observed in men.18 A cohort study of 64 597 Swedish participants without diabetes showed that women with fasting plasma glucose levels of 6.1–6.9 mmol/L and >6.9 mmol/L had a 1.31‐fold (95%CI, 1.11–1.54) and 1.51‐fold (95%CI, 1.13–1.97) greater incidence of cancer, respectively.19 Obesity has been considered a possible confounding factor for the association between diabetes and cancer risk. However, adjustment for body mass index had no material effect on risk estimates. The Diabetes Epidemiology: Collaborative Analysis of Diagnostic Criteria in Europe (DECODE) Study, which examined data on 44 655 individual participants from 17 European population‐based or occupational cohorts, revealed that the multivariate‐adjusted hazard ratios (RRs) of cancer mortality were 1.12 (95%CI, 1.02–1.23) in subjects with prediabetes, 1.28 (95%CI, 1.08–1.51) in those with newly diagnosed diabetes, and 1.57 (95%CI, 1.29–1.91) in those with known diabetes (Fig. 1).20 There were no substantial differences in the associations between the sexes. The risk of total cancer deaths was higher in individuals with prediabetes or diabetes, irrespective of body mass index. Similar results were found in Asian populations. A 10‐year follow‐up study of 1 298 385 Koreans indicated linear trends of increases in cancer mortality and incidence with higher fasting plasma glucose levels, even in the prediabetes range of 6.1–6.9 mmol/L.21 In our Japanese community‐based cohort study of 2476 individuals with a 14‐year follow‐up, the risk of cancer mortality doubled in individuals with diabetes as compared to those with normal glucose tolerance levels (RR 2.10; 95%CI, 1.41–3.12). Additionally, impaired fasting glucose and impaired glucose tolerance were significantly associated with 1.49‐fold (95%CI, 1.05–2.11) and 1.52‐fold (95%CI, 1.05–2.22) higher mortality, respectively, from any cancer.22 A recent meta‐analysis from individual participant data on 123 205 deaths among 820 900 people in 97 prospective studies, in which our study was also involved, clearly showed that diabetes was associated with a 1.27‐fold (95% CI, 1.20–1.34) higher risk of death from any cancer after adjusting for confounding factors, including body mass index, and cancer mortality increased significantly among individuals whose fasting glucose levels exceeded 5.6 mmol/L.23 Thus, epidemiological studies provide evidence that diabetes and prediabetes are associated with an increased risk of total cancer.
Figure 1.
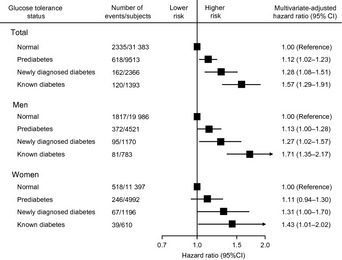
Relationship between glucose tolerance status and the risk of cancer mortality: results of the Diabetes Epidemiology: Collaborative Analysis of Diagnostic Criteria in Europe (DECODE) Study. The risk estimates were adjusted for age, sex, study cohort, systolic blood pressure, cholesterol, body mass index, and smoking status. The results have been adapted from Zhou et al.20 CI, confidence interval.
Liver cancer
Some epidemiological evidence has suggested a significant association between diabetes and liver cancer, after taking important confounding factors into account, specifically alcohol consumption and hepatitis B or C virus infection.8, 24, 25 In a population‐based case–control study carried out in the USA consisting of 2061 cases of hepatocellular carcinoma and 6183 controls, diabetes was associated with a 2.87‐fold (95%CI, 2.49–3.30) increase in the risk of liver cancer, even after excluding from the study population individuals with hepatitis B or C virus infections, alcoholic liver diseases, or hemochromatosis.24 Similar results were obtained in a case–control study carried out in Greece on 333 cases of hepatocellular carcinoma and 363 controls. Diabetes patients were at a 1.86‐fold (95%CI, 0.99–3.51) increase in the risk of liver cancer after adjusting for aforementioned risk factors.25 A recent systematic review of 18 cohort studies also indicated a significant association between diabetes and an increased risk of hepatocellular carcinoma; the incidence of hepatocellular carcinoma increased by 101% (95%CI, 61–151%) and that of mortality by 56% (95%CI, 30–87%) in individuals with diabetes compared to those without.8 Subgroup analyses revealed that the increased risk of hepatocellular carcinoma in individuals with diabetes was independent of alcohol consumption, a history of cirrhosis, and hepatitis B or C virus infection. These findings strongly support a positive association between diabetes and the risk of hepatocellular carcinoma.
Diabetes patients may have a greater risk of biliary tract cancer. A meta‐analysis investigated the possible association between diabetes and biliary tract cancers, such as gallbladder cancer and extrahepatic cholangiocarcinoma.9 This meta‐analysis included 8 case–control and 13 cohort studies, and found that diabetes was associated with an increased risk of biliary tract cancer, as compared to no diabetes (RR 1.43; 95%CI, 1.18–1.72). In some studies, although not in all, diabetes was independently associated with an increased risk of biliary stones,26, 27 which are considered major risk factors for biliary tract cancer. Biliary stones may act as an intermediate factor between diabetes and biliary tract cancer.
Pancreatic cancer
Diabetes has been thought to increase the risk of pancreatic cancer. A recent meta‐analysis of 35 cohort studies documented a 94% (95%CI, 66–127%) increase in the risk of pancreatic cancer in individuals with diabetes relative to those without diabetes.10 Although there was significant evidence of heterogeneity in the association across the studies included, subgroup analyses revealed that the increased risk of pancreatic cancer was independent of geographic location, sex, study design, alcohol consumption, body mass index, and smoking status. Interpretation of the causal nature of the association between diabetes and pancreatic cancer is complicated by the fact that abnormal glucose metabolism may be a consequence of pancreatic cancer. However, this meta‐analysis also found that diabetes was associated with a 1.83‐fold (95%CI, 1.38–2.43) increased risk of pancreatic cancer among individuals who have had diabetes for at least 5 years, implying that reverse causality has little influence on the association.
Gastric cancer
Evidence linking diabetes to gastric cancer was reported in a meta‐analysis of 4 case–control and 17 cohort studies, in which diabetes was significantly associated with a 1.18‐fold (95%CI, 1.01–1.39) increased risk of gastric cancer in women, whereas no such association was found in men with diabetes. There was strong evidence of heterogeneity among the studies in this meta‐analysis.11 Similarly, a prospective study of 97 771 Japanese with a follow‐up of 11 years showed that diabetes was significantly associated with an increased incidence (61%) of gastric cancer (95%CI, 2–154%) in women, whereas the association was not significant in men (RR 1.23; 95%CI, 0.98–1.54), after adjusting for potential confounding factors, such as smoking status, body mass index, physical activity, and dietary factors.28 However, the risk estimates used in this prospective study were not adjusted for the status of Helicobacter pylori infection. We also investigated the association between fasting glucose levels and the incidence of gastric cancer in a 9‐year cohort study of 2466 Japanese. As a consequence, the multivariate‐adjusted RR of developing gastric cancer was 2.3 (95%CI, 1.1–5.0) in individuals with modestly elevated fasting plasma glucose levels (5.3–5.8 mmol/L) and 3.1 (95%CI, 1.5–6.4) in individuals with more elevated levels (>5.8 mmol/L), compared to those with values below 5.3 mmol/L.29 This excess risk was documented only in individuals with H. pylori infection, suggesting that hyperglycemia is a possible cofactor increasing the risk posed by H. pylori (Fig. 2). The same was true in an investigation of association between hemoglobin A1c levels and the incidence of gastric cancer.30
Figure 2.
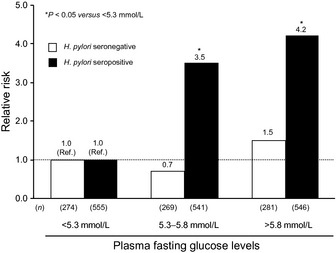
Age‐ and sex‐adjusted relative risks of gastric cancer according to fasting glucose levels under stratification by Helicobacter pylori (H. pylori) status. The results have been adapted from Yamagata et al.29 Ref., Reference.
Colorectal cancer
Lifestyle factors, such as a lack of exercise and obesity, are thought to be convincing risk factors for colorectal cancer.31 The association between diabetes and colorectal cancer has been elucidated in numerous epidemiological studies, and meta‐analyses have been published.12 A recent meta‐analysis of 30 cohort studies documented that diabetes was associated with an increased incidence of colorectal cancer (RR 1.27; 95%CI, 1.21–1.34) and with mortality from colorectal cancer (RR 1.20; 95%CI, 1.03–1.40) (Fig. 3). Although there was strong evidence of heterogeneity among the studies in that meta‐analysis, subgroup analysis and meta‐regression analysis by controlling for confounders showed that the increased risk of colorectal cancer in individuals with diabetes was independent of geographic location (Europe versus Asia), sex, family history of colorectal cancer, smoking, physical activity, and body mass index. This association may be attributable to slower bowel transit times in individuals with diabetes, which could contribute to the increased exposure of colonic mucosa to potential carcinogens (e.g. bile acid).32, 33
Figure 3.
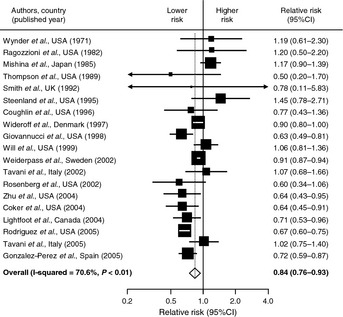
Meta‐analysis of 19 studies that examined the association between diabetes mellitus and prostate cancer. The pooled risk estimate was computed using a random‐effects model because there was significant heterogeneity in the association among studies included. The results are adapted from Kasper & Giovannucci17. CI, confidence interval.
Urinary organ cancer
A meta‐analysis of nine cohort studies reported a significant association between diabetes and kidney cancer (RR 1.42; 95%CI, 1.06–1.91).13 However, this association was weaker and not statistically significant when the studies were restricted to those that were controlled for body mass index or obesity. This finding suggests that the observed association is attributable in part to obesity as a confounding factor.
The results of epidemiological studies that investigated the association between diabetes and bladder cancer were inconsistent. In a meta‐analysis of 16 studies (7 case–control studies, 3 cohort studies, and 6 cohort studies of diabetes patients), individuals with diabetes had a modest (24%) increase in the risk of bladder cancer (95%CI, 8–42%).14 However, there was strong evidence of between‐study heterogeneity, and a funnel plot showed some asymmetry. Thus, the possibility that the association is due to confounding or bias cannot be ruled out.
Sex‐specific cancers
Breast cancer is the most common malignancy affecting women worldwide. Its incidence rises with age and is associated with hormonal factors, benign breast disease, family history of breast cancer, and genetic factors.34 A meta‐analysis of 20 studies (5 case–control and 15 cohort studies) showed a 20% (95%CI, 12–28%) increased risk of breast cancer in women with diabetes, with the vast majority of the cases being type 2 diabetes.15 The same was true among studies that were controlled for body mass index. Increased exposure to estrogen in diabetes is thought to be one of the biological mechanisms between diabetes and breast cancer.35 However, the data on the influence of menopausal status on the association between diabetes and breast cancer are inconclusive.
One meta‐analysis based on 16 studies (13 case–control and 3 cohort studies) has shown an increased risk of endometrial cancer in women with diabetes.16 The site‐specific biological mechanism underlying the development of endometrial cancer in women with diabetes is hyperinsulinemia, which may increase levels of bioactive estrogen by decreasing concentrations of circulating sex hormone‐binding globulin.36 Estrogen has been shown to increase cancer risk by stimulating the proliferation of endometrial cells37 when unopposed by progesterone.38
The prostate differs from other sites of cancer in its association with diabetes. A meta‐analysis of 19 studies (9 case–control and 10 cohort studies) has shown that men with diabetes were at a significantly decreased risk of developing prostate cancer (Fig. 3).17 The suggested mechanism underlying this inverse association is thought to be reduced testosterone level, which is commonly observed in men with diabetes or with obesity secondary to low levels of sex hormone‐binding globulin.39 Furthermore, several studies have determined that variants of the HNF1B gene at 17q12, which predispose haplotype‐carrying subjects to diabetes, were associated with a decreased risk of prostate cancer.40 However, it is unknown whether or not HNF1B alters levels of various metabolic and hormonal factors that may influence prostate cancer risk in men with diabetes.
Type 1 and Type 2 Diabetes and Cancer Risk
Diabetes is divided into two subtypes, type 1 and type 2, which differ in metabolic and hormonal characteristics. In type 1 diabetes patients (5–10% of all diabetics), hyperglycemia is associated with an absolute deficiency of endogenous insulin secretion, whereas hyperglycemia and hyperinsulinemia coexist for a long time in type 2 diabetes, because of insulin resistance in peripheral tissues. Despite these considerable pathogenetic and clinical differences, most studies on the association between diabetes and cancer have been carried out without distinction between the two subtypes of diabetes. Because type 1 diabetes is less frequent than type 2 diabetes, especially in adult populations, it is reasonable to suppose that the large majority of the epidemiological data on cancer incidence and mortality has been obtained in type 2 diabetes patients. Therefore, it is questionable whether these data can be automatically extended to type 1 diabetes patients.
A few studies have elucidated the association between type 1 diabetes and the risk of cancer. A cohort of 29 187 Swedish patients who were hospitalized for type 1 diabetes showed a 20% increase in overall cancer incidence (standardized incidence ratio [SIR] 1.2; 95% CI, 1.0–1.3), when data for the entire Swedish population were used as a reference.41 In this study, patients with type 1 diabetes had excess risks of cancers of the stomach (SIR 2.3; 95% CI, 1.1–4.1), cervix (SIR 1.6; 95%CI, 1.1–2.2), and endometrium (SIR 2.7; 95%CI, 1.4–4.7). In addition, a meta‐analysis including three cohort studies and six case–control studies found that the overall RR for pancreatic cancer in young‐onset or type 1 diabetes was 2.00 (95%CI, 1.37–3.01) as compared with no diabetes, without significant heterogeneity between cohort studies and case–control studies.42 However, the relationship between type 1 diabetes and excess cancer risk remains inconclusive because the method of diagnosing diabetes subtypes in these studies is not reliable.
Interpretation of Epidemiological Evidence between Diabetes and Cancer Risk
An accumulation of epidemiological evidence based on observational studies has indicated a statistically significant association between diabetes and cancer risk. However, the evidence does not directly prove biological causality, and we cannot fully exclude the possibility of bias or a confounding of the association.
A case–control study is generally dependent on the collection of retrospective data and thus is inherently more sensitive to recall and selection bias than a prospective study design. Additionally, there is a consensus that a systematic review can provide a more reliable estimation of published research than can traditional narrative reviews. However, in a systematic literature review, publication bias is an inevitable problem, because negative or inconclusive results tend to be unpublished and to be unavailable for analysis. Thus, the risk estimates from a systematic review of published studies may be overestimated. Surveillance bias may also occur in the association between diabetes and cancer risk: diabetes patients visit physicians more frequently than do non‐diabetes patients, and consequently, cancer may be detected more in diabetes patients than in those without diabetes.
Confounding is likely to happen in the association between diabetes and cancer risk, because diabetes and cancer share several risk factors, including obesity and low physical activity. Thus, the increased risk of cancer in diabetes patients may in part reflect confounding by these risk factors. Even though the risk estimates were adjusted for these possible confounding factors in most epidemiological studies, residual confounding cannot be excluded completely. Furthermore, reverse causality may also exist. Cancer causes insulin resistance through the production of inflammatory cytokine (e.g. tumor necrosis factor‐α) and subsequent hyperglycemia and diabetes.43, 44
Accordingly, several possible biases or confounding factors might exist in the relationship between diabetes and cancer risk. Nevertheless, it is evident that diabetes increases the risk of cancer from an epidemiological viewpoint. To confirm a causal association, it would be necessary to identify the biological mechanisms involved through experimental studies, and to ascertain whether or not intervention against key factors of this mechanism reduces cancer risk in humans.
Possible Biological Mechanisms Underlying the Association between Diabetes and Cancer
Although biological mechanisms underlying the association between diabetes and cancer have been inconclusive, several possible mechanisms have been proposed, such as hyperinsulinemia, hyperglycemia, and inflammation.
Type 2 diabetes is characterized by insulin resistance and secondary hyperinsulinemia. The role of insulin in promoting cancer growth was first recognized in studies with experimental animals. When insulin deficiency was induced by alloxan in rats, insulin given to these animals appeared to display intrinsic growth‐stimulating effects on the mammary tumor tissue induced by 7,12‐dimethylbenz(a)anthracene dissolved in sesame oil.45 Most cancer cells express insulin receptors, thus hyperinsulinemia has been thought to promote carcinogenesis by directly stimulating the proliferating pathway after insulin receptors.46 Elevated insulin levels may occur early in the natural history of diabetes and decline as diabetes advances. If hyperinsulinemia has a role in promoting cancer initiation and/or progression, these aspects should be considered when estimating the risk of an individual diabetes patient developing cancer. Intriguingly, this hypothesis may be supported by evidence that treatment with metformin, which increases insulin sensitivity and decreases hyperinsulinemia, is associated with a lower incidence of cancer in diabetes patients than in those being treated with insulin or sulfonylurea.47, 48
Apart from the direct effects of insulin on cancer cells, it is possible that hyperinsulinemia promotes carcinogenesis indirectly through the effect of insulin‐like growth factor (IGF)‐1. Insulin reduces the production of IGF binding protein‐1 and consequently increases the bioactive IGF‐1. Insulin‐like growth factor‐1 has more potent mitogenic and anti‐apoptotic activities than insulin and could act as a stimulus for growing preneoplastic and neoplastic cells.49, 50, 51
Hyperglycemia itself has also been reported to promote tumor cell proliferation and metastasis in type 2 diabetes.52 A clinical study of diabetes patients and healthy volunteers demonstrated that diabetes is associated with increased production of reactive oxygen species and greater oxidative damage to DNA, which may lead to mutational changes in oncogenes and tumor suppressor genes, and thereby to the development of cancer.53 However, the aforementioned study using a rat model with alloxan showed reduced tumor growth, possibly implying that hyperglycemia itself does not lead to induce neoplastic growth.45 Thus, the effect of hyperglycemia itself on carcinogenesis should be further elucidated.
The metabolic abnormalities that characterize diabetes, especially under conditions of poor metabolic control, cause a permanent pro‐inflammatory condition. Inflammatory cytokines produced by adipose tissues, such as interleukin‐6, monocyte chemoattractant protein, and plasminogen activator inhibitor‐1, may play important roles in the carcinogenic process, cancer progression, or poor prognosis. Additionally, increased leptin and decreased adiponectin levels may also contribute to the increased risk of cancer among individuals with obesity and type 2 diabetes.54 This chronic pro‐inflammatory state reduces intracellular antioxidant capacity, predisposing susceptible cells to malignant transformation. In fact, high concentrations of diverse free radicals and oxidants generate potent reactive oxygen species that can damage cell DNA by direct oxidation or by interfering with the mechanisms of DNA repair.
Conclusions
Epidemiologic evidence suggests that individuals with diabetes are at significantly higher risk for many forms of cancer except prostate cancer. The excess risk of cancer conferred by diabetes is moderate. Nevertheless, the cancer risk among diabetes patients may become a clinical, social, and economic burden in the future because of the growing worldwide frequency of diabetes. Further investigation is needed to develop a more rational approach to cancer prevention and treatment among individuals with diabetes.
Disclosure Statement
The authors have no conflict of interest.
(Cancer Sci, doi: 10.1111/cas.12043, 2012)
References
- 1. Zimmet P, Alberti KG, Shaw J. Global and societal implications of the diabetes epidemic. Nature 2001; 414: 782–7. [DOI] [PubMed] [Google Scholar]
- 2. The IDF DIABETES ATLAS, 5th edn [Cited 15 August 2012.] Available from: http://www.idf.org/diabetesatlas/5e/diabetes. [Google Scholar]
- 3. Ministry of Health, Labour and Welafare, Japan (in Japanese). [Cited 15 August 2012.] Available from: http://www.mhlw.go.jp/houdou/2008/12/dl/h1225-5d.pdf.
- 4. Kubo M, Hata J, Doi Y, Tanizaki Y, Iida M, Kiyohara Y. Secular trends in the incidence of and risk factors for ischemic stroke and its subtypes in Japanese population. Circulation 2008; 118: 2672–8. [DOI] [PubMed] [Google Scholar]
- 5. Chan JC, Malik V, Jia W et al Diabetes in Asia: epidemiology, risk factors, and pathophysiology. JAMA 2009; 301: 2129–40. [DOI] [PubMed] [Google Scholar]
- 6. Lam EK, Batty GD, Huxley RR et al Associations of diabetes mellitus with site‐specific cancer mortality in the Asia‐Pacific region. Ann Oncol 2011; 22: 730–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Vigneri P, Frasca F, Sciacca L, Pandini G, Vigneri R. Diabetes and cancer. Endocr Relat Cancer 2009; 16: 1103–23. [DOI] [PubMed] [Google Scholar]
- 8. Wang C, Wang X, Gong G et al Increased risk of hepatocellular carcinoma in patients with diabetes mellitus: a systematic review and meta‐analysis of cohort studies. Int J Cancer 2012; 130: 1639–48. [DOI] [PubMed] [Google Scholar]
- 9. Ren HB, Yu T, Liu C, Li YQ. Diabetes mellitus and increased risk of biliary tract cancer: systematic review and meta‐analysis. Cancer Causes Control 2011; 22: 837–47. [DOI] [PubMed] [Google Scholar]
- 10. Ben Q, Xu M, Ning X, Wang Y, Li Y. Diabetes mellitus and risk of pancreatic cancer: a meta‐analysis of cohort studies. Eur J Cancer 2011; 47: 1928–37. [DOI] [PubMed] [Google Scholar]
- 11. Ge Z, Ben Q, Qian J, Wang Y, Li Y. Diabetes mellitus and risk of gastric cancer: a systematic review and meta‐analysis of observational studies. Eur J Gastroenterol Hepatol 2011; 23: 1127–35. [DOI] [PubMed] [Google Scholar]
- 12. Jiang Y, Ben Q, Shen H, Lu W, Zhang Y, Zhu J. Diabetes mellitus and incidence and mortality of colorectal cancer: a systematic review and meta‐analysis of cohort studies. Eur J Epidemiol 2011; 26: 863–76. [DOI] [PubMed] [Google Scholar]
- 13. Larsson SC, Wolk A. Diabetes mellitus and incidence of kidney cancer: a meta‐analysis of cohort studies. Diabetologia 2011; 54: 1013–8. [DOI] [PubMed] [Google Scholar]
- 14. Larsson SC, Orsini N, Brismar K, Wolk A. Diabetes mellitus and risk of bladder cancer: a meta‐analysis. Diabetologia 2006; 49: 2819–23. [DOI] [PubMed] [Google Scholar]
- 15. Larsson SC, Mantzoros CS, Wolk A. Diabetes mellitus and risk of breast cancer: a meta‐analysis. Int J Cancer 2007; 121: 856–62. [DOI] [PubMed] [Google Scholar]
- 16. Friberg E, Orsini N, Mantzoros CS, Wolk A. Diabetes mellitus and risk of endometrial cancer: a meta‐analysis. Diabetologia 2007; 50: 1365–74. [DOI] [PubMed] [Google Scholar]
- 17. Kasper JS, Giovannucci E. A meta‐analysis of diabetes mellitus and the risk of prostate cancer. Cancer Epidemiol Biomarkers Prev 2006; 15: 2056–62. [DOI] [PubMed] [Google Scholar]
- 18. Verlato G, Zoppini G, Bonora E, Muggeo M. Mortality from site‐specific malignancies in type 2 diabetic patients from Verona. Diabetes Care 2003; 26: 1047–51. [DOI] [PubMed] [Google Scholar]
- 19. Stattin P, Bjor O, Ferrari P et al Prospective study of hyperglycemia and cancer risk. Diabetes Care 2007; 30: 561–7. [DOI] [PubMed] [Google Scholar]
- 20. Zhou XH, Qiao Q, Zethelius B et al Diabetes, prediabetes and cancer mortality. Diabetologia 2010; 53: 1867–76. [DOI] [PubMed] [Google Scholar]
- 21. Jee SH, Ohrr H, Sull JW, Yun JE, Ji M, Samet JM. Fasting serum glucose level and cancer risk in Korean men and women. JAMA 2005; 293: 194–202. [DOI] [PubMed] [Google Scholar]
- 22. Hirakawa Y, Ninomiya T, Mukai N et al Association between glucose levels and cancer deth in a general Japanese population: the Hisayama Study. Am J Epidemiol 2012; 176: 856–64. [DOI] [PubMed] [Google Scholar]
- 23. The Emerging Risk Factors Collaboration . Diabetes mellitus, fasting glucose, and risk of cause‐specific death. N Engl J Med 2011; 364: 829–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Davila JA, Morgan RO, Shaib Y, McGlynn KA, El‐Serag HB. Diabetes increases the risk of hepatocellular carcinoma in the United States: a population based case control study. Gut 2005; 54: 533–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Lagiou P, Kuper H, Stuver SO, Tzonou A, Trichopoulos D, Adami HO. Role of diabetes mellitus in the etiology of hepatocellular carcinoma. J Natl Cancer Inst 2000; 92: 1096–9. [DOI] [PubMed] [Google Scholar]
- 26. Shebl FM, Andreotti G, Rashid A et al Diabetes in relation to biliary tract cancer and stones: a population‐based study in Shanghai, China. Br J Cancer 2010; 103: 115–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Festi D, Dormi A, Capodicasa S et al Incidence of gallstone disease in Italy: results from a multicenter, population‐based Italian study (the MICOL project). World J Gastroenterol 2008; 14: 5282–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Inoue M, Iwasaki M, Otani T, Sasazuki S, Noda M, Tsugane S. Diabetes mellitus and the risk of cancer: results from a large‐scale population‐based cohort study in Japan. Arch Intern Med 2006; 166: 1871–7. [DOI] [PubMed] [Google Scholar]
- 29. Yamagata H, Kiyohara Y, Aoyagi K et al Impact of Helicobacter pylori infection on gastric cancer incidence in a general Japanese population: the Hisayama study. Arch Intern Med 2000; 160: 1962–8. [DOI] [PubMed] [Google Scholar]
- 30. Ikeda F, Doi Y, Yonemoto K et al Hyperglycemia increases risk of gastric cancer posed by Helicobacter pylori infection: a population‐based cohort study. Gastroenterology 2009; 136: 1234–41. [DOI] [PubMed] [Google Scholar]
- 31. American Institute for Cancer Research . World Cancer Research Fund and American Institute for Cancer Research: Food, nutrition, physical activity, and the prevention of cancer: a global prespective 2007. Washington DC: American Institute for Cancer Research, 2007. [Google Scholar]
- 32. Will JC, Galuska DA, Vinicor F, Calle EE. Colorectal cancer: another complication of diabetes mellitus? Am J Epidemiol 1998; 147: 816–25. [DOI] [PubMed] [Google Scholar]
- 33. Yang R, Arem R, Chan L. Gastrointestinal tract complications of diabetes mellitus. Pathophysiology and management. Arch Intern Med 1984; 144: 1251–6. [PubMed] [Google Scholar]
- 34. Key TJ, Verkasalo PK, Banks E. Epidemiology of breast cancer. Lancet Oncol 2001; 2: 133–40. [DOI] [PubMed] [Google Scholar]
- 35. Kaaks R, Rinaldi S, Key TJ et al Postmenopausal serum androgens, oestrogens and breast cancer risk: the European prospective investigation into cancer and nutrition. Endocr Relat Cancer 2005; 12: 1071–82. [DOI] [PubMed] [Google Scholar]
- 36. Kaaks R. Nutrition, hormones, and breast cancer: is insulin the missing link? Cancer Causes Control 1996; 7: 605–25. [DOI] [PubMed] [Google Scholar]
- 37. Graham JD, Clarke CL. Physiological action of progesterone in target tissues. Endocr Rev 1997; 18: 502–19. [DOI] [PubMed] [Google Scholar]
- 38. Key TJ, Pike MC. The dose‐effect relationship between ‘unopposed’ oestrogens and endometrial mitotic rate: its central role in explaining and predicting endometrial cancer risk. Br J Cancer 1988; 57: 205–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Bonovas S, Filioussi K, Tsantes A. Diabetes mellitus and risk of prostate cancer: a meta‐analysis. Diabetologia 2004; 47: 1071–8. [DOI] [PubMed] [Google Scholar]
- 40. Sun J, Zheng SL, Wiklund F et al Evidence for two independent prostate cancer risk‐associated loci in the HNF1B gene at 17q12. Nat Genet 2008; 40: 1153–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Zendehdel K, Nyren O, Ostenson CG, Adami HO, Ekbom A, Ye W. Cancer incidence in patients with type 1 diabetes mellitus: a population‐based cohort study in Sweden. J Natl Cancer Inst 2003; 95: 1797–800. [DOI] [PubMed] [Google Scholar]
- 42. Stevens RJ, Roddam AW, Beral V. Pancreatic cancer in type 1 and young‐onset diabetes: systematic review and meta‐analysis. Br J Cancer 2007; 96: 507–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. McCall JL, Tuckey JA, Parry BR. Serum tumour necrosis factor alpha and insulin resistance in gastrointestinal cancer. Br J Surg 1992; 79: 1361–3. [DOI] [PubMed] [Google Scholar]
- 44. Noguchi Y, Yoshikawa T, Marat D et al Insulin resistance in cancer patients is associated with enhanced tumor necrosis factor‐alpha expression in skeletal muscle. Biochem Biophys Res Commun 1998; 253: 887–92. [DOI] [PubMed] [Google Scholar]
- 45. Heuson JC, Legros N, Heimann R. Influence of insulin administration on growth of the 7,12‐dimethylbenz(a)anthracene‐induced mammary carcinoma in intact, oophorectomized, and hypophysectomized rats. Cancer Res 1972; 32: 233–8. [PubMed] [Google Scholar]
- 46. Giovannucci E, Harlan DM, Archer MC et al Diabetes and cancer: a consensus report. Diabetes Care 2010; 33: 1674–85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Currie CJ, Poole CD, Gale EA. The influence of glucose‐lowering therapies on cancer risk in type 2 diabetes. Diabetologia 2009; 52: 1766–77. [DOI] [PubMed] [Google Scholar]
- 48. Libby G, Donnelly LA, Donnan PT, Alessi DR, Morris AD, Evans JM. New users of metformin are at low risk of incident cancer: a cohort study among people with type 2 diabetes. Diabetes Care 2009; 32: 1620–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Bruning PF, Bonfrer JM, van Noord PA, Hart AA, de Jong‐Bakker M, Nooijen WJ. Insulin resistance and breast‐cancer risk. Int J Cancer 1992; 52: 511–6. [DOI] [PubMed] [Google Scholar]
- 50. Giovannucci E. Insulin and colon cancer. Cancer Causes Control 1995; 6: 164–79. [DOI] [PubMed] [Google Scholar]
- 51. Hu FB, Manson JE, Liu S et al Prospective study of adult onset diabetes mellitus (type 2) and risk of colorectal cancer in women. J Natl Cancer Inst 1999; 91: 542–7. [DOI] [PubMed] [Google Scholar]
- 52. Richardson LC, Pollack LA. Therapy insight: influence of type 2 diabetes on the development, treatment and outcomes of cancer. Nat Clin Pract Oncol 2005; 2: 48–53. [DOI] [PubMed] [Google Scholar]
- 53. Lorenzi M, Montisano DF, Toledo S, Barrieux A. High glucose induces DNA damage in cultured human endothelial cells. J Clin Invest 1986; 77: 322–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Vona‐Davis L, Howard‐McNatt M, Rose DP. Adiposity, type 2 diabetes and the metabolic syndrome in breast cancer. Obes Rev 2007; 8: 395–408. [DOI] [PubMed] [Google Scholar]


