Abstract
Background
Underweight is one form of indicators of under-nutrition, which results from the poor nutrient intake and underlying health problems. Its impact is beyond an individual and extends to a country level. It has been known from the literature that underweight has a negative effect on income and development of a country. In the context of Ethiopia, factors predicting underweight remain unknown and there is a paucity of evidence on geographical distribution of underweight among individuals aged 15–49 years. Therefore, the aim of this study was to examine the geographic distribution of underweight and its associated factors among individuals aged 15–49 years in Ethiopia.
Methods
Secondary data analysis was done on a data set consisting of 28,450 individuals and obtained from the Ethiopian Demography and Health Survey (EDHS) 2016. The spatial distribution of underweight across the country was identified by ArcGIS software. Hotspots analysis was done using Getis-Ord Gi* statistic within ArcGIS. In SaTScan software, the Bernoulli model was fitted by Kulldorff’s methods to identify the purely spatial clusters of underweight. A binary logistic regression was applied to determine factors associated with being underweight.
Result
In Ethiopia, the spatial distribution of underweight was clustered with Global Moran’s I = 0.79 at p-value < 0.0001. The highest underweight clusters were observed in Tigray, Gambella, eastern part of Amhara, and western and central part of Afar regions. Male individuals [AOR = 1.21; 95% CI: (1.15 1.28)], never married [AOR = 1.14; 95% CI: (1.05, 1.24)], rural residents [AOR = 1.32; 95% CI: (1.18, 1.47)], rich [AOR = 0.85; 95% CI: (0.76, 0.94)], cigarette smoking [AOR = 1.25; 95% CI: (1.07, 1.46)], drinking treated water [AOR = 0.91; 95% CI: (0.83, 0.99)] and open filed defecation [AOR = 1.17; 95% CI: (1.08, 1.26)] were found to have a significant association with being underweight.
Conclusions
There was a significant clustering of underweight among individuals aged 15–49 years. Gender, age, marital status, place of residence, wealth index, cigarette smoking, using untreated water and types of toilet were the significant factors of being underweight. Therefore, effective public health interventions like building safe and supportive environments for nutrition, providing socio-economic protection and nutrition-related education for poor and rural resident would be better to mitigate these situations and associated risk factors in hot spot areas. In addition, policymakers should strengthen and promote nutrition sensitive policies and activities in order to alleviate the underlying and basic causes of underweight.
Background
Underweight is a one form of under-nutrition, defined by the World Health Organization (WHO) as body mass index (BMI) less than 18.5 kg/m2 [1]. It also refers to deficiencies, in a person’s intake of energy and/or nutrients that leads to low productivity among adults and it is related to heightened morbidity and mortality [2]. Underweight results from poor nutrient intake and underlying health problems. Patients with such chronic diseases as HIV/AIDS, respiratory infections, malaria and diarrheal disease are prone to being underweight [3].
The prevalence of underweight is low in developed countries, but it remains high in developing countries. Globally, 462 million adults are underweight and more than one third of Low and Middle Income Countries face double burdens of both under and overweight, particularly in sub-Saharan Africa, South Asia, and East Asia and the Pacific [4,5]. According to regional WHO reports, South East Asia and Africa have the highest prevalence of underweight among adults, with 20.3% and 11.1% of adults in the region being underweight, respectively. In Ethiopia, the prevalence of adult underweight is 17.2% among men and 14.5% among women with an overall prevalence of 15.8% [6].
Compared to people of normal-weight, Underweight people have increased risk of mortality [7]. Researchers found that Having BMI less than or equal to 22.5kg/m2 increased the risk of death by 1.48-fold in women and 1.26-fold in men [8–10]. Moreover, chronic malnutrition in women of childbearing age is also linked to poor pregnancy outcomes [11,12]. Malnourished mothers are more likely to give birth to babies with low birth weights who are at a greatly increased risk of death in infancy. Women who are underweight prior to pregnancy and gain little weight during pregnancy are at increased risk of complications and death [13,14].
The effect of underweight extends beyond an individual and it has a negative impact on income and development of a country [15]. It is a particular concern for developing countries as it is a leading cause of death and disability [16]. Lower level of education, low income, inadequate health care coverage, food habits and a sedentary lifestyle are factors that are responsible for high burden of underweight in developing countries [17,18].
In order to manage the negative consequences of underweight and to mitigate the problems associated with it, exploration of underlying socio-economic factors is crucial. Despite its high prevalence, factors influencing underweight remain unclear in Ethiopia. In addition, there is a paucity of evidence on geographical distribution of underweight among individuals aged 15–49 years in the country. Therefore, the current study applies spatial analysis and logistic regression to determine the geographic distribution of underweight and its associated factors among individuals aged 15–49 years in Ethiopia.
Methods
Study setting and data source
Ethiopia is located in the horn of Africa and has nine regions (Afar, Tigray, Amhara, Oromia, Somali, Southern Nations, Nationalities, and People’s Region (SNNPR), Benishangul Gumuz, Gambella and Harari) and two administrative cities (Addis Ababa and Dire Dawa) [15]. The current population of Ethiopia is 114,864,753 as of Monday, June 22, 2020, based on Worldometer elaboration of the latest United Nations data [16].
The data for this study was taken from the demography heath survey (DHS) program official database www.measuredhs.com, after authorization was granted through online request by explaining the goal of our study. The 2016 EDHS was the fourth Demographic and Health Survey conducted in Ethiopia from January 18, 2016, to June 27, 2016 [19].
The 2016 EDHS used a two-stage stratified cluster sampling method. In the first stage, a total of 645 Enumeration Areas (EAs) (202 in urban areas and 443 in rural areas) were selected with probability proportional to EA size and with independent selection in each sampling stratum.
In the second stage of selection, a fixed number of 28 households per cluster were selected with an equal probability systematic selection from the newly created household listing [19]. The 2016 EDHS collected anthropometric data on height and weight for both women and men age 15–49 years. In the present study, total weighted samples of 28,450 individuals were included. Latitude and longitude coordinates had been also taken from selected EAs (clusters). The specific sampling system was presented in the full EDHS report [19].
Outcome and explanatory variable
The outcome variable for this study was BMI, which is categorized as underweight if BMI is less than 18.5 and non-underweight if the BMI is greater than or equal to 18.5. Depending on different literature review; independent variables included in the analysis are described in Table 1.
Table 1. Description of explanatory variables used in the analysis.
| Variables | Description | Category |
|---|---|---|
| Gender | Sex of household member | Female, Male |
| Age (years) | Age of household member | 15–19, 20–24, 25–29, 30–34, 35–39, 40–44, 45–49, 50–54 and 55–59 |
| Marital status | Never married, Currently married & Formerly married (It includes divorced, separated and widowed) | Never married, Currently married & Formerly/ever married |
| Educational level | Highest educational level attained | No education, Primary, Secondary, Higher and Don’t know |
| Number of household members | Number of household members | 1–3, 4–6 and >6 |
| Place of residence | Type of place of residence | Urban and Rural |
| Region | Region | Tigray, Afar, Amhara, Oromia, Somali, Benishangul, SNNPR, Gambella, Harari, Addis Ababa and Dire Dawa |
| Wealth index | It is the percent distribution of the de jure population by wealth quintiles and the Gini coefficient. | Poorest, Poorer, Middle, Richer & Richest |
| Drinking water treatment | Anything done to water to make safe to drink | Yes, No and Don't know |
| Types of toilet | Types of toilet facility | Flush, Pit latrine, Open field and Other* |
| Cigarette smoking | Smoking cigarettes in last 24 hours | Yes and No |
*Other: Composting toilet, bucket toilet, hanging toilet/latrine; SNNPR: Southern Nation and Nationality and Peoples Region.
Data processing
We used STATA 14; ArcGIS 10.1 and SaTScan 9.6 software’s to perform data analysis. Before any statistical analysis, the data was weighted using sampling weight (household sample weight), primary sampling unit, and strata to restore the representativeness of the survey and to tell the STATA to consider the sampling design when calculating standard errors to get reliable statistical estimates. Descriptive statistics and summary statistics were presented using text and tables.
Spatial analysis
Spatial autocorrelation analysis
Global Moran’s index (Moran’s I) was used to identify the presence of spatial autocorrelation. Moran’s I values close to -1 indicated disease/event dispersed, whereas Moran’s I values close to + 1 indicated disease/event clustered, and disease/event distributed randomly if Moran’s I value was zero. A statistically significant Moran’s I (p < 0.05) led to the rejection of the null hypothesis (underweight is randomly distributed) and indicated the presence of spatial autocorrelation. Hotspot analysis was done using as Getis-Ord Gi* statistic.
Spatial scan statistical analysis
Statistically significant Primary (most likely) and secondary clusters of underweight was identified by applying Spatial scan statistics using Kulldorff’s SaTScan software. SaTScan™ works with a moving window and requires fixing of the window size that moves across the study area. The outcome variable has a Bernoulli distribution, so Bernoulli model was used by applying the Kulldorff’s method for purely spatial analysis.
Individuals who were underweight were taken as cases and those who were not underweight were taken as controls to fit the Bernoulli model. The default maximum spatial cluster size of < 50% of the population was used as an upper limit, which allowed both small and large clusters to be detected and ignored clusters that contained more than the maximum limit. Areas with high Log Likelihood Ratio and significant p-value were considered as areas with high underweight compared to areas outside of the window.
Statistical analysis
In EDHS data, individuals within a cluster may be more similar to each other than individuals in other cluster. This violates the assumption independence of observations and equal variance across clusters. This implies that the need to consider the between-cluster variability using advanced models. Since the response variable was dichotomous, binary logistic regression and Generalized Linear Mixed Model were fitted. The Intra-class Correlation Coefficient (ICC) value was 0.036 which indicated us to select a fixed model (binary logistic regression) over the mixed model.
Variables with p-values ≤0.2 in the bi-variable analysis were fitted in the multivariable model to measure the effect of each variable after adjusting for the effect of other variables. Adjusted Odds Ratio (AOR) with a 95% Confidence Interval (CI) and p-value < 0.05 in the multivariable model were declared as determinant factors of underweight. Multi-collinearity and model adequacy were also checked using a variance inflation factor (VIF) and Hosmer-Lemeshow test respectively.
Ethics approval and consent to participate
Permission for data access was obtained from major demographic and health survey through the online request from http://www.dhsprogram.com. The data used for this study were publicly available with no personal identifier. They gave permission to access the data with reference number of 144044.
Results
Characteristics of study population
A total of weighted sample of 27,214 individuals were included in the analysis. Among these study participants, more than half (53.35%) of them were females, more than one-third (36.84%) of them were not educated, more than three-fourth (79.57%) were rural dwellers and one-quarter (25.39%) of them were richest. About 6,052 (22.24%) of the study participants were in the age range of 15–19 years. Regarding the marital status, around 16,189 (59.49%) respondents were married, whereas 9,075 (33.35%) respondents were never married. Nearly two-third (65.18%) of the study participants had pit types of latrine and only 9.16 percent of respondents had done something to the water to make safe to drink (Table 2).
Table 2. Characteristics of respondents in Ethiopia from January 18 to June 27, 2016.
| Variables | Un-weighted frequency | Weighted frequency | Weighted percentage |
|---|---|---|---|
| Gender | |||
| Female | 14,099 | 14,529 | 53.35 |
| Male | 11,285 | 12,695 | 46.65 |
| Age | |||
| 15–19 | 5,707 | 6,052 | 22.24 |
| 20–24 | 4,351 | 4,439 | 16.31 |
| 25–29 | 4,510 | 4,797 | 17.62 |
| 30–34 | 3,464 | 3,903 | 14.34 |
| 35–39 | 3,149 | 3,409 | 12.53 |
| 40–44 | 2,374 | 2,545 | 9.35 |
| 45–49 | 1,829 | 2,069 | 7.60 |
| Marital status | |||
| Never married | 8,829 | 9,075 | 33.35 |
| Currently married | 14,529 | 16,189 | 59.49 |
| Formerly/ever married | 2,026 | 1,950 | 7.16 |
| Educational level | |||
| No education | 8,816 | 10,026 | 36.84 |
| Primary | 9,957 | 11,705 | 43.00 |
| Secondary | 4,120 | 3,624 | 13.32 |
| Higher | 2,444 | 1,814 | 6.67 |
| Don’t know | 47 | 45 | 0.17 |
| Number of household members | |||
| 1–3 | 5,888 | 5,468 | 20.09 |
| 4–6 | 11,629 | 13,028 | 47.87 |
| >6 | 7,867 | 8,718 | 32.04 |
| Place of residence | |||
| Urban | 8,146 | 5,560 | 20.43 |
| Rural | 17,238 | 21,654 | 79.57 |
| Region | |||
| Tigray | 2,787 | 1,917 | 7.05 |
| Afar | 1,660 | 194 | 0.70 |
| Amhara | 3,249 | 7,257 | 26.92 |
| Oromia | 3,243 | 9,780 | 35.70 |
| Somali | 2,010 | 632 | 2.32 |
| Benishangul | 1,849 | 260 | 0.95 |
| SNNPR | 3,105 | 5,541 | 20.41 |
| Gambella | 1,763 | 75 | 0.27 |
| Harari | 1,257 | 51 | 0.19 |
| Addis Ababa | 2,750 | 1,366 | 4.95 |
| Dire Dawa | 1,711 | 141 | 0.51 |
| Wealth index | |||
| Poorest | 6,009 | 4,381 | 16.10 |
| Poorer | 3,482 | 4,918 | 18.07 |
| Middle | 3,407 | 5,336 | 19.61 |
| Richer | 3,614 | 5,669 | 20.83 |
| Richest | 8,872 | 6,910 | 25.39 |
| Drinking water treatment | |||
| Yes | 2,776 | 2,489 | 9.16 |
| No | 22,585 | 24,707 | 90.78 |
| Don't know | 23 | 18 | 0.06 |
| Cigarette smoking | |||
| Yes | 773 | 405 | 1.72 |
| No | 24,506 | 26,721 | 98.28 |
| Types of toilet | |||
| Flush | 1,743 | 844 | 3.10 |
| Pit latrine | 15,152 | 17,738 | 65.18 |
| Open field | 8,129 | 8,235 | 30.26 |
| Other * | 360 | 397 | 1.46 |
| Total | 25,384 | 27,214 | 100% |
*Other: Composting toilet, bucket toilet, hanging toilet/latrine; SNNPR: Southern Nation and Nationality and Peoples Regions.
Spatial analysis of underweight
Spatial distribution of underweight
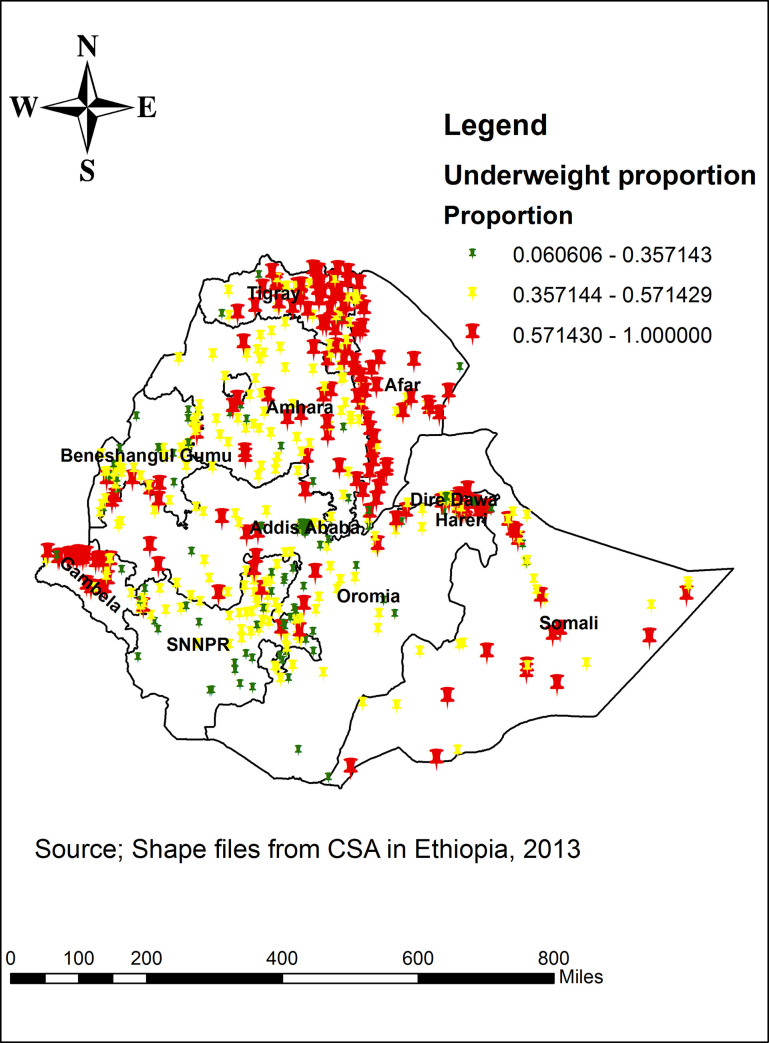
The spatial distribution of underweight among individuals aged 15–49 years in Ethiopia was non-random (clustered) (Global Moran’s I = 0.79, p value < 0.0001) (Fig 1). The highest proportions of underweight was located in the Tigray, Amhara, western and central part of Afar and Gambella regions, while low proportions of underweight was identified in the eastern part of SNNPR, western part of Oromia Harar, Dire Dawa and Addis Ababa (Fig 2).
Fig 1. Spatial autocorrelation analysis of underweight among individuals aged 15–49 years in Ethiopia, 2016.
Fig 2. Spatial distribution of underweight across regions among individuals aged 15–49 years in Ethiopia, 2016.
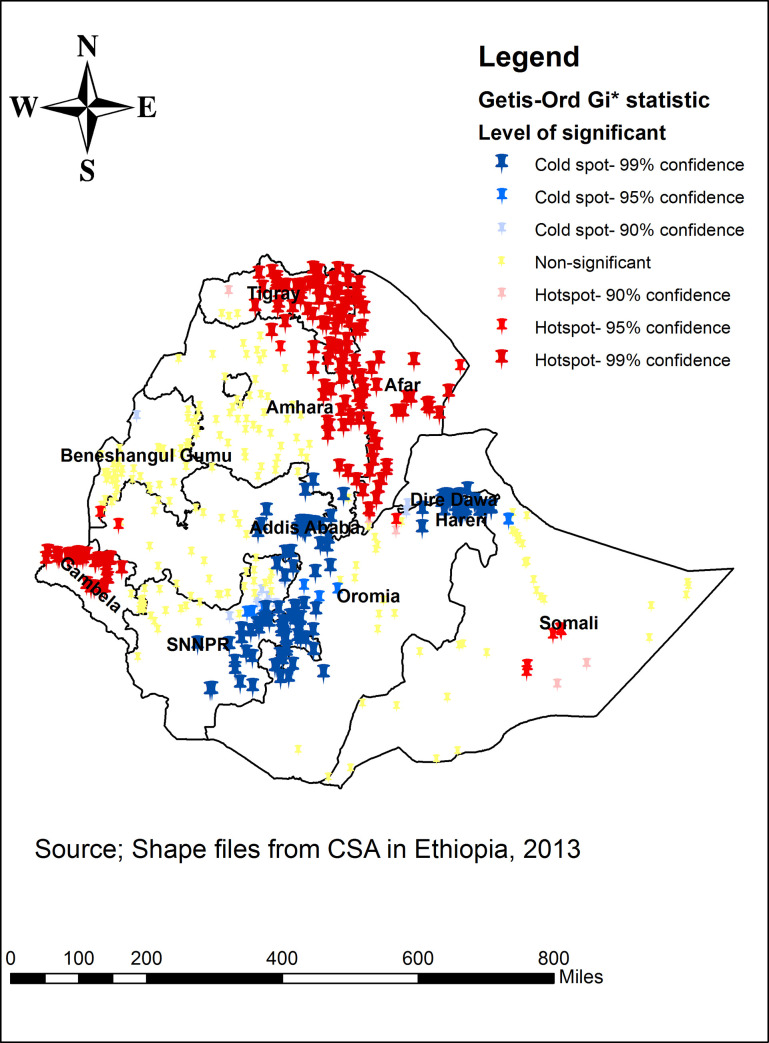
Getis-Ord Gi* statistical analysis of underweight
The Gettis-Ord Gi* statistical analysis indicated the hotspot and cold spot of underweight among individuals aged 15–49 years in Ethiopia. The red colors show the significant hotspot areas (higher cluster of underweight), which was located in Tigray, Gambella, eastern part of Amhara, western and central part of Afar regions. In contrast, the blue color indicates significant cold spot (lower cluster of underweight), identified in eastern part of SNNPR, western part of Oromia, Harar, Dire Dawa and Addis Ababa (Fig 3).
Fig 3. Hotspot and Cold spot underweight across regions among individuals aged 15–49 years in Ethiopia, 2016.
Spatial SaTScan analysis of underweight (Bernoulli based model)
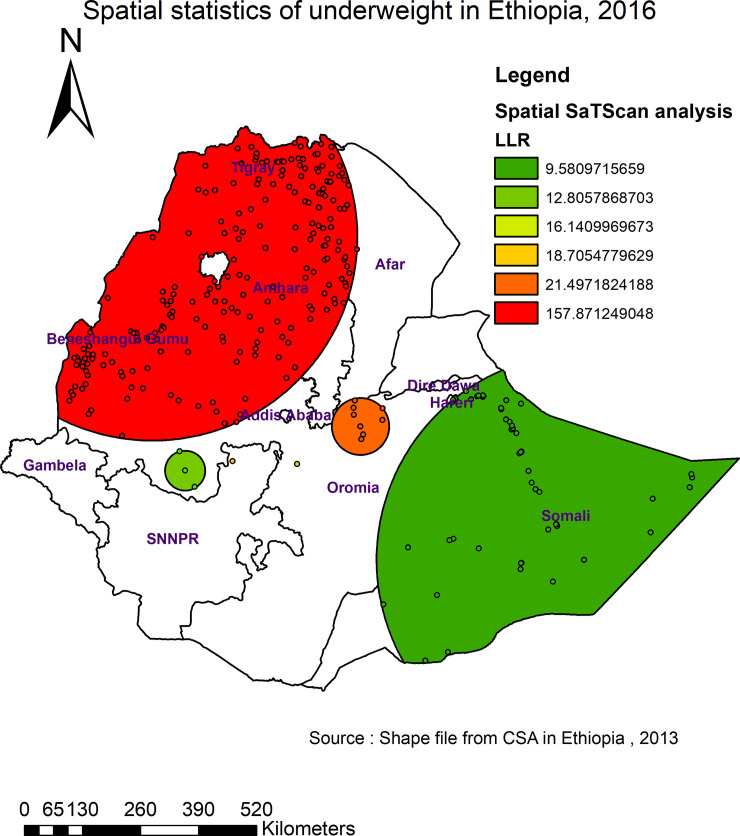
In the SaTScan analysis, a total of 279 significant clusters of underweight among individuals age 15–49 years in Ethiopia were identified (Fig 4). Among these, 216 were primary clusters. The primary clusters were found in Tigray, Amhara, Benishangul and western part of Afar at 12.579086 N, 36.033274 E with 457.42 km radius, a Relative Risk (RR) of 1.27, and Log-Likelihood Ratio (LRR) of 157.87, at p-value < 0.001. Individuals within the spatial window had 1.27 times higher chance of being underweight as compared to those individuals outside the spatial window (Table 3). The secondary clusters were located in the Gambella region.
Fig 4. Primary and secondary clusters of underweight among individuals age 15–49 years in Ethiopia, 2016.
Table 3. Significant spatial clusters with high rate underweight among individuals age 15–49 years in Ethiopia, 2016.
| Cluster | Enumeration area (cluster) identified | Coordinate (radius) | Population | Case | RR | LLR | P-value |
|---|---|---|---|---|---|---|---|
| 1 | 52, 279, 292, 259, 415, 602, 386, 504, 541, 296, 548, 638, 640, 256, 169, 361, 431, 515, 612, 516, 615, 73, 498, 312, 163, 158, 327, 253, 322, 382, 152, 533, 246, 109, 559, 512, 36, 167, 35, 258, 137, 364, 183, 80, 132, 150, 429, 3, 244, 403, 456, 628, 199, 24, 457, 184, 583, 188, 425, 494, 340, 375, 268, 120, 324, 474, 569, 98, 627, 285, 255, 320, 551, 409, 38, 65, 209, 181, 528, 78, 407, 88, 563, 584, 335, 595, 124, 66, 581, 545, 621, 542, 156, 206, 433, 508, 203, 531, 317, 597, 579, 636, 400, 6, 575, 482, 165, 416, 590, 176, 218, 81, 70, 229, 349, 17, 538, 350, 401, 300, 591, 84, 424, 161, 392, 355, 136, 374, 10, 481, 304, 478, 460, 45, 143, 294, 604, 430, 462, 160, 395, 461, 479, 449, 97, 354, 267, 237, 94, 550, 351, 200, 89, 510, 442, 616, 605, 275, 455, 280, 399, 220, 384, 193, 617, 226, 129, 79, 128, 410, 496, 598, 623, 643, 341, 421, 175, 404, 99, 249, 298, 488, 234, 517, 248, 18, 511, 196, 611, 332, 413, 572, 189, 344, 130, 345, 423, 117, 127, 241, 192, 172, 571, 485, 191, 362, 389, 235, 263, 103, 118, 254, 558, 585, 23, 544 |
(12.579086 N, 36.033274 E) / 457.42 km | 11149 | 5692 | 1.27 | 157.87 | < 0.001 |
| 2 | 122, 245, 529, 71, 49, 476, 51, 506 | (8.757437 N, 40.299443 E) / 64.13 km | 827 | 462 | 1.26 | 21.50 | < 0.001 |
LLR: Likelihood ratio; RR: Relative risk.
Factors associated with underweight
According to multivariable logistic regression analysis, gender, age, marital status, number of household members, place of residence, region, wealth index, drinking treated water, types of toilet and cigarette smoking were significantly associated with underweight among individuals aged 15–49 years in Ethiopia at p-value 0.05 (Table 4).
Table 4. Bi-variable and multivariable logistic regression analysis of underweight among individuals age 15–49 years in Ethiopia, 2016.
| Variables | Underweight | Odds Ratio (95% CI) | P-value | ||
|---|---|---|---|---|---|
| Yes | No | COR | AOR | ||
| Gender | |||||
| Female | 6,086 | 8,433 | 1 | 1 | - |
| Male | 6,125 | 6,570 | 1.25 (1.19, 1.31) | 1.21 (1.15, 1.28) | <0.001** |
| Age | |||||
| 15–19 | 3,071 | 2,981 | 1 | 1 | |
| 20–24 | 2,029 | 2,410 | 0.79 (0.73, 0.86) | 0.87 (0.80, 0.96) | 0.003* |
| 25–29 | 1,957 | 2,840 | 0.67 (0.62, 0.73) | 0.75 (0.68, 0.83) | <0.001** |
| 30–34 | 1,548 | 2,355 | 0.63 (0.58, 0.68) | 0.67 (0.60, 0.76) | <0.001** |
| 35–39 | 1,485 | 1,924 | 0.71 (0.65, 0.78) | 0.75 (0 .67, 0.85) | <0.001** |
| 40–44 | 1,156 | 1,389 | 0.77 (0.70, 0.85) | 0.78 (0.69, 0.88) | <0.001** |
| 45–49 | 965 | 1,104 | 0.81 (0.73, 0.90) | 0.82 (0.72, 0.94) | 0.004* |
| Marital status | |||||
| Currently married | 7,024 | 9,165 | 1 | 1 | |
| Never married | 4,327 | 4,748 | 1.06 (1.01, 1.12) | 1.14 (1.05, 1.24) | 0.002* |
| Formerly/ever married | 860 | 1,090 | 0.93 (0.84, 1.02) | 1.02 (0.92, 1.13) | 0.069 |
| Educational level | |||||
| No education | 4,608 | 5,418 | 1 | 1 | |
| Primary | 5,549 | 6,156 | 0.90 (0 .85, 0.95) | 1.01 (0.94, 1.08) | 0.797 |
| Secondary | 1,436 | 2,188 | 0.66 (0.62, 0.72) | 0.97 (0 .89, 1.07) | 0.574 |
| Higher | 600 | 1,214 | 0.46 (0.42, 0.51) | 0.91 (0 .81, 1.02) | 0.116 |
| Don’t know | 18 | 27 | 0.57 (0 .29, 0.95) | 0.65 (0.34, 1.26) | 0.204 |
| Number of household members | |||||
| 1–3 | 2,270 | 3,198 | 1 | 1 | |
| 4–6 | 5,964 | 7,064 | 1.20 (1.13, 1.28) | 1.07 (1.01, 1.15) | 0.049* |
| >6 | 3,977 | 4,741 | 1.36 (1.27, 1.45) | 1.06 (0.98, 1.14) | 0.138 |
| Place of residence | |||||
| Urban | 1,779 | 3,781 | 1 | 1 | |
| Rural | 10,432 | 11,222 | 2.43 (2.30, 2.57) | 1.32(1.18, 1.47) | <0.001** |
| Region | |||||
| Oromia | 1,118 | 799 | 1 | 1 | |
| Tigray | 113 | 81 | 1.63(1.47, 1.81) | 1.74 (1.56, 1.94) | <0.001** |
| Afar | 3,522 | 3,735 | 1.94 (1.72, 2.19) | 1.89 (1.65, 2.16) | <0.001** |
| Amhara | 4,487 | 5,293 | 1.10 (1.00, 1.21) | 1.08 (0.97, 1.19) | 0.145 |
| Somali | 337 | 295 | 1.28 (1.14, 1.43) | 1.22 (1.08, 1.38) | <0.001** |
| Benishangul | 113 | 147 | 0.86 (0.77, 0.97) | 0.84 (0.74, 0.94) | 0.004* |
| SNNPR | 2,075 | 3,466 | 0.68 (0.61, 0.75) | 0.67 (0.64, 0.74) | <0.001** |
| Gambella | 37 | 38 | 1.42 (1.26, 1.59) | 1.62 (1.43, 1.83) | <0.001** |
| Harari | 20 | 31 | 0.72 (0.63, 0.82) | 1.04 (0 .90, 1.20) | 0.590 |
| Addis Ababa | 334 | 1,032 | 0.37 (0.33, 0.41) | 0.81 (0 .71, 0.93) | 0.003* |
| Dire Dawa | 55 | 86 | 0.70 (0.62, 0.79) | 1.06 (0.94, 1.21) | 0.278 |
| Wealth index | |||||
| Poorest | 2,220 | 2,161 | 1 | 1 | |
| Poorer | 2,478 | 2,440 | 0.76 (0.70, 0.83) | 0.97 (0 .88, 1.07) | 0.572 |
| Middle | 2,570 | 2,766 | 0.70 (0.64, 0.76) | 0.93 (0.84, 1.03) | 0.182 |
| Richer | 2,659 | 3,010 | 0.61 (0.56, 0.66) | 0.85 (0.76, 0.94) | 0.002* |
| Richest | 2,284 | 4,626 | 0.31 (0.29, 0.33) | 0.55 (0 .48, 0.63) | <0.001** |
| Drinking water treatment | |||||
| No | 1,032 | 1,457 | 1 | 1 | |
| Yes | 11,170 | 13,537 | 0.77 (0.71, 0.83) | 0.91 (0.83, 0.99) | 0.031* |
| Don't know | 9 | 9 | 0.90 (0.39, 2.05) | 0.70 (0.30, 1.65) | 0.415 |
| Types of toilet | |||||
| Pit latrine | 247 | 597 | 1 | 1 | |
| Flush | 7,526 | 10,212 | 0.58 (0.52, 0.65) | 0.78 (0.69, 0.88) | <0.001** |
| Open field | 4,262 | 3,973 | 2.03 (1.93, 2.15) | 1.17 (1.08, 1.26) | <0.001** |
| Other | 176 | 221 | 1.11 (0.90, 1.37) | 1.05 (0.84, 1.31) | 0.677 |
| Cigarette smoking | |||||
| No | 190 | 215 | 1 | 1 | |
| Yes | 11,970 | 14,751 | 1.42 (1.23, 1.64) | 1.25 (1.07, 1.46) | 0.005* |
| Hosmer-Lemeshow chi2 (10) | 9.33(0.3150) | ||||
| Mean VIF (Max) | 1.88(5.78) | ||||
*: P ≤ 0.05
**: P ≤ 0.001; Other: Composting toilet, bucket toilet, hanging toilet/latrine; AOR: Adjusted Odd ratio; COR: Crude Odd Ratio; CI: Confidence interval; SNNPR: Southern Nation and Nationality and Peoples Regions; VIF: variance inflation factor.
Male individuals were 1.21 times more likely to be underweight than female individuals. The odds of being underweight were increased among individuals aged 20–59 years than individuals aged 15–19 years. Never married individuals were 1.14 times more likely to be underweight than married individuals. Persons who have lived with 4–6 household members were 1.07 times more likely to be underweight than persons who were living with 1–3 household members.
Regions were an important variable that showed significant association with underweight among individuals aged 15–49 years in Ethiopia. Persons residing in Tigray, Afar, Somali and Gambella were 1.74, 1.89, 1.22 and 1.62 times more likely to be underweight than a person residing in Oromia respectively, whereas the odds of underweight were decreased by 16 percent, 18 percent and 33 percent among individuals residing in Benishangul, Addis Ababa and SNNPR as compared with adult residing in Oromia respectively. Rural residents were 1.32 times more likely to be underweight than their counterpart.
Regarding wealth index, the probabilities of underweight were decreased by 15 percent and 45 percent among richer and richest individuals as compared with poorest individuals. Persons who smoke cigarette were 1.25 times more likely to be underweight than their counterpart. The odds of underweight were decreased by 9 percent among individuals who treat drinking water before use when compared with their counterpart.
Type of toilet was also another factor, which has a determinate effect on underweight in Ethiopia. The likelihoods of being underweight were decreased by 22 percent among person who use flush type of latrine than those who use a pit type of latrine. In contrast, persons who defecate in the open field were 1.17 times more likely to be underweight while compared with individuals who use a pit type of latrine.
Discussion
According to the current study, the spatial distribution of underweight among individuals aged 15–49 years in Ethiopia was non-random. The significant hotspot areas with high rates of underweight were found in Tigray, Gambella, and eastern part of Amhara, western and central part of Afar region. In contrast, the significant cold spots with high rates of underweight were observed in the eastern part of SNNPR, western part of Oromia Harar, Dire Dawa and Addis Ababa. The possible explanation for this variation might be due to different socio-economical compositions of the study participants. Specifically, the majority of residents in Addis Ababa and Dire Dawa were rich in their wealth index, whereas majority dwellers of Afar and Gambella were poorer. Availability of natural resources may also be the reasons for this variation. These natural resources include water, land along with all vegetation and animal life. Regions like SNNPR and Oromia, Benishangul and Amhara have lands which are suitable for agriculture.
This study revealed that male individuals were more likely to be underweight than females. This finding is in line with a study conducted in Yemen [20]. The result shows that boys have less access to nutritious food and fall sick more frequently as compared to the girls. This similar finding might be explained by the fact that boys are more likely to be addicted to alcohol and cigarettes [21]. In line with studies conducted in Nepal [22], Indonesia [23] and India [24] the present study reported that the odds of being underweight were increased among adult age 15–19 years than adult age 20–59 years. Explanations for the increased odds of underweight during early adulthood may be related to food insecurity, fear of being fat, high physiologic need and eating disorder [25].
Marital status is a social factor which is of significance in health and mortality. According to the present study, the likelihoods of being underweight were decreased among married individuals than never married ones. A similar result has been found in Nepal [22]. This is because married people, especially men, are more likely to be healthier than unmarried men [26,27]. The current study also documented that a significant association between family size and underweight. Persons who have lived with 4–6 household members were more likely to be underweight than persons who were living with 1–3 household members. This finding may not be surprising because as the family size increases, keeping food security becomes a big issue, especially in poor families [28].
In agreement with studies conducted in Nepal [22] and Indonesia [23], the current study documented that rural residents were more likely to be underweight than urban dwellers. The possible explanations for such results might be rural residents are less likely to work in sedentary employment, use motorized transportation, eat diets high in processed grains and sugars [29,30]. Furthermore, because socioeconomic statuses are more likely to be lower among rural residents and this is associated with a lower BMI [31,32]. According to the present study, the odds of underweight were decreased among rich individuals than poor individuals, this is of course in congruent with another report [22]. This finding might be due to the fact that poverty intensifies the hazard of, and risks from, malnutrition. People who are poor are more likely to be suffering from distinct forms of malnutrition. Additionally, malnutrition increases health care costs, reduces productivity, and slows financial growth, which can perpetuate a cycle of poverty and ill-health [33].
The current study documented that persons who smoke cigarette were more likely to be underweight than their counterparts. This finding is consistent with other previous studies [34–36]. Such finding might be related to reasons like decrement in appetite and calorie intake, enhanced metabolism, and reduced fat accumulation [34]. This may be due to the effects of nicotine on brain’s regulation of appetite and energy expenditure [37,38].
The present study also reported that the significant association between types of latrine and being underweight. The odds of underweight were decreased among adult who treats drinking water before use when compared with their counterparts. Persons who defecate in the open field were more likely to be underweight while compared with persons who use a pit type of latrine. We included these two factors in the analysis because of their association with Feco-oral diseases. Especially those individuals who used untreated water and defecated in an open field are high risk of contracting fecal-oral diseases. Furthermore, these factors are signs of lower socio-economic status, which can determine the nutritional status of individuals.
The main strength of the present study was the use of nationally representative data. As a result, the findings of this study are generalizable to the target population of Ethiopia. Moreover, the simultaneous use of both ArcGIS and Sat Scan statistical tests enabled to find similar and statistically significant area with a high cluster of underweight (hot spot area).
This study has limitations that should be considered when interpreting the results. One of the limitations of the current study was, the location of data values was shifted up to 2 kilometers for urban and up to 5 kilometers for rural areas to ensure respondent confidentiality. Thus, this was the challenge to know the exact cases’ location. Since the current study used secondary data, some important variable like dietary intake, physical activity and sedentary lifestyle were not included in the analysis.
Conclusion
There was a significant clustering of underweight among individuals age 15–49 years in Tigray, Gambella, and eastern part of Amhara, western and central part of Afar regions. The odds of being underweight were higher among males, individuals aged 15–19 years, never married, rural residents, poorest and cigarette smoker. Persons, who drank untreated water, defecate on open field and households with large family size were more likely to be underweight. Therefore, effective public health interventions like building safe and supportive environments for nutrition, providing socio-economic protection and nutrition-related education for poor and rural resident would be better to mitigate these situations and associated risk factors in hot spot areas. In addition, policymakers should strengthen and promote nutrition sensitive policies and activities in order to alleviate the underlying and basic causes of underweight.
Supporting information
(XLS)
Acknowledgments
We would like to thank the Ethiopian Central Statistics Agency for providing us with all the relevant secondary data used in this study.
Abbreviations
- AOR
Adjusted Odds Ratio
- BMI
Body mass index
- CI
Confidence Interval
- CSA
Central Statistics Agency
- DHS
Demography heath survey
- EAs
Enumeration Areas
- EDHS
Ethiopian Demographic and Health Survey
- SNNPR
Southern Nation and Nationality and Peoples Regions
- VIF
variance inflation factor
- WHO
World Health Organization
Data Availability
All relevant data are within the manuscript and its Supporting Information files.
Funding Statement
The author(s) received no specific funding for this work.
References
- 1.WHO. The use and interpretation of anthropometry: report of a WHO expert committee 1995. [312–409]. [PubMed] [Google Scholar]
- 2.Maleta KJMmjtjoMAoM. Undernutrition. 2006;18(4):189. [PMC free article] [PubMed] [Google Scholar]
- 3.Fryar CD, Ogden CLJAJ. Prevalence of underweight among adults aged 20 years and over: United States, 2007–2008. 2010;1. [Google Scholar]
- 4.Popkin BM, Slining M. New dynamics in global obesity facing low‐and middle‐income countries. Obesity reviews. 2013;14:11–20. 10.1111/obr.12102 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.WHO. More than one in three low- and middle-income countries face both extremes of malnutrition 2019 [updated 16 December].
- 6.WHO. Prevalence of underweight among adults, BMI < 18.5, age-standardized Estimates by country 2017 [updated 22, September].
- 7.Zheng W, McLerran DF, Rolland B, Zhang X, Inoue M, Matsuo K, et al. Association between body-mass index and risk of death in more than 1 million Asians. New England Journal of Medicine. 2011;364(8):719–29. 10.1056/NEJMoa1010679 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Pereira IFdS, Spyrides MHC, Andrade LdMB. Nutritional status of elderly Brazilians: a multilevel approach. Cadernos de Saúde Pública. 2016;32(5). 10.1590/0102-311X00178814 [DOI] [PubMed] [Google Scholar]
- 9.Flegal KM, Graubard BI, Williamson DF, Gail MH. Excess deaths associated with underweight, overweight, and obesity. Jama. 2005;293(15):1861–7. 10.1001/jama.293.15.1861 [DOI] [PubMed] [Google Scholar]
- 10.Padwal R, Majumdar SR, Leslie WD. Relationship among body fat percentage, body mass index, and all-cause mortality. Annals of internal medicine. 2016;165(8):604. [DOI] [PubMed] [Google Scholar]
- 11.Black RE, Victora CG, Walker SP, Bhutta ZA, Christian P, De Onis M, et al. Maternal and child undernutrition and overweight in low-income and middle-income countries. The lancet. 2013;382(9890):427–51. 10.1016/S0140-6736(13)60937-X [DOI] [PubMed] [Google Scholar]
- 12.Han Z, Mulla S, Beyene J, Liao G, McDonald SD. Maternal underweight and the risk of preterm birth and low birth weight: a systematic review and meta-analyses. International journal of epidemiology. 2011;40(1):65–101. 10.1093/ije/dyq195 [DOI] [PubMed] [Google Scholar]
- 13.Organization A. The double burden of malnutrition: Case studies from six developing countries: Food & Agriculture Org.; 2006. [PubMed] [Google Scholar]
- 14.Black R, Bhutta Z, Bryce J, Morris S, Victora C. The Lancet’s Series on Maternal and Child Undernutrition. The Lancet. 2013;2013:1–12. [Google Scholar]
- 15.Mendez MA, Monteiro CA, Popkin BM. Overweight exceeds underweight among women in most developing countries. The American journal of clinical nutrition. 2005;81(3):714–21. 10.1093/ajcn/81.3.714 [DOI] [PubMed] [Google Scholar]
- 16.Woodward M, Reid MAJTMjoA. Cardiovascular disease in the Asia–Pacific region: challenges for health research and policy. 2003;179(2):71–2. [DOI] [PubMed] [Google Scholar]
- 17.Doku DT, Neupane SJBph. Double burden of malnutrition: increasing overweight and obesity and stall underweight trends among Ghanaian women. 2015;15(1):670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Pryer JA, Rogers S, Rahman AJSb. Factors affecting nutritional status in female adults in Dhaka slums, Bangladesh. 2003;50(3–4):259–69. [DOI] [PubMed] [Google Scholar]
- 19.Agency CS, ICF. Ethiopia Demographic and Health Survey 2016: Key Indicators Report. Addis Ababa, Ethiopia, and Rockville, Maryland, USA: CSA and ICF; 2016. [Google Scholar]
- 20.Gunaid A. Obesity, overweight and underweight among adults in an urban community in Yemen. EMHJ-Eastern Mediterranean Health Journal, 18 (12), 1187–1193, 2012. 2012. [DOI] [PubMed] [Google Scholar]
- 21.Becker JB, McClellan ML, Reed BG. Sex differences, gender and addiction. J Neurosci Res. 2017;95(1–2):136–47. 10.1002/jnr.23963 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Rawal LB, Kanda K, Mahumud RA, Joshi D, Mehata S, Shrestha N, et al. Prevalence of underweight, overweight and obesity and their associated risk factors in Nepalese adults: data from a Nationwide Survey, 2016. PloS one. 2018;13(11):e0205912 10.1371/journal.pone.0205912 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Pengpid S, Peltzer K. The Prevalence of Underweight, Overweight/Obesity and Their Related Lifestyle Factors in Indonesia, 2014–2015. AIMS Public Health [Internet]. 2017 2017; 4(6):[633–49 pp.]. 10.3934/publichealth.2017.6.633 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Siddiqui MZ, Donato R. Undernutrition among adults in India: the significance of individual-level and contextual factors impacting on the likelihood of underweight across sub-populations. Public health nutrition. 2017;20(1):130–41. 10.1017/S1368980016001968 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Patton GC, Selzer R, Coffey C, Carlin JB, Wolfe RJB. Onset of adolescent eating disorders: population based cohort study over 3 years. 1999;318(7186):765–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hu Y, Goldman N. Mortality differentials by marital status: an international comparison. Demography. 1990;27(2):233–50. [PubMed] [Google Scholar]
- 27.Trovato F, Lauris G. Marital status and mortality in Canada: 1951–1981. Journal of Marriage and the Family. 1989:907–22. 12343800 [Google Scholar]
- 28.Ajao K, Ojofeitimi E, Adebayo A, Fatusi A, Afolabi O. Influence of family size, household food security status, and child care practices on the nutritional status of under-five children in Ile-Ife, Nigeria. African journal of reproductive health. 2010;14(4). [PubMed] [Google Scholar]
- 29.Monteiro CA, Conde WL, Popkin BM. The burden of disease from undernutrition and overnutrition in countries undergoing rapid nutrition transition: a view from Brazil. American journal of public health. 2004;94(3):433–4. 10.2105/ajph.94.3.433 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Popkin BM. The nutrition transition and obesity in the developing world. The Journal of nutrition. 2001;131(3):871S–3S. 10.1093/jn/131.3.871S [DOI] [PubMed] [Google Scholar]
- 31.Neuman M, Finlay JE, Davey Smith G, Subramanian S. The poor stay thinner: stable socioeconomic gradients in BMI among women in lower-and middle-income countries. The American journal of clinical nutrition. 2011;94(5):1348–57. 10.3945/ajcn.111.018127 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Subramanian S, Perkins JM, Özaltin E, Davey Smith G. Weight of nations: a socioeconomic analysis of women in low-to middle-income countries. The American journal of clinical nutrition. 2011;93(2):413–21. 10.3945/ajcn.110.004820 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.WHO. Malnutrition. 2020. 1 April. [Google Scholar]
- 34.Wang Q. Smoking and body weight: evidence from China health and nutrition survey. BMC Public Health. 2015;15(1):1238 10.1186/s12889-015-2549-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Jitnarin N, Kosulwat V, Rojroongwasinkul N, Boonpraderm A, Haddock CK, Poston WS. The relationship between smoking, body weight, body mass index, and dietary intake among Thai adults: results of the national Thai food consumption survey. Asia Pacific Journal of Public Health. 2014;26(5):481–93. 10.1177/1010539511426473 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ward KD, Ahn S, Mzayek F, Al Ali R, Rastam S, Asfar T, et al. The relationship between waterpipe smoking and body weight: population-based findings from Syria. Nicotine & Tobacco Research. 2015;17(1):34–40. 10.1093/ntr/ntu121 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Stadler M, Tomann L, Storka A, Wolzt M, Peric S, Bieglmayer C, et al. Effects of smoking cessation on b-cell function, insulin sensitivity, body weight, and appetite. Eur J Endocrinol. 2014;170(2):219–27. 10.1530/EJE-13-0590 [DOI] [PubMed] [Google Scholar]
- 38.Chen H, Hansen MJ, Jones JE, Vlahos R, Anderson GP, Morris MJ. Long-term cigarette smoke exposure increases uncoupling protein expression but reduces energy intake. Brain research. 2008;1228:81–8. 10.1016/j.brainres.2008.06.067 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(XLS)
Data Availability Statement
All relevant data are within the manuscript and its Supporting Information files.