Abstract
Specialized proresolving mediators (SPMs) are endogenous lipid metabolites of long‐chain polyunsaturated fatty acids that are involved in promoting the resolution of inflammation. Many disease conditions characterized by excessive inflammation have impaired or altered SPM biosynthesis, which may lead to chronic, unresolved inflammation. Exogenous administration of SPMs in infectious conditions has been shown to be effective at improving infection clearance and survival in preclinical models. SPMs have also shown tremendous promise in the context of inflammatory lung conditions, such as acute respiratory distress syndrome and chronic obstructive pulmonary disease, mostly in preclinical settings. To date, SPMs have not been studied in the context of the novel Coronavirus, severe acute respiratory syndrome Coronavirus‐2 (SARS‐CoV‐2), however their preclinical efficacy in combatting infections and improving acute respiratory distress suggest they may be a valuable resource in the fight against Coronavirus disease‐19 (COVID‐19). Overall, while the research on SPMs is still evolving, they may offer a novel therapeutic option for inflammatory conditions.
Keywords: acute respiratory distress syndrome, COVID‐19, inflammation resolution, lung injury, specialized proresolving mediators
Abbreviations
- 15‐HETE
15‐hydroxyeicosatetraenoic acid
- 17(S)‐HDHA
17(S)‐hydroxydocosahexaenoic acid
- 18(R)‐HEPE
18(R)‐hydroxyeicosapentaenoic acid
- AA
arachidonic acid
- ALX/FPR2
lipoxin A4 receptor/formyl peptide receptor 2
- ARDS
acute respiratory distress syndrome
- AT
aspirin triggered
- CF
cystic fibrosis
- CLP
cecal ligation and puncture
- COPD
chronic obstructive pulmonary disease
- DHA
docosahexaenoic acid
- DRV1/GPR32
resolvin D1 receptor/G protein‐coupled receptor 32
- EPA
eicosapentaenoic acid
- ERV1/CMKLR1
E series resolvin receptor/chemokine‐like receptor 1
- HSV
herpes simplex virus
- IL
interleukin
- LCPUFA
long‐chain polyunsaturated fatty acids
- LOX
lipoxygenase
- LTB4
leukotriene B4
- LXA4
lipoxin A4
- MaR1
maresin 1
- MERS‐CoV
middle eastern respiratory syndrome Coronavirus
- MyD88
myeloid differentiation primary response 88
- NF‐κB
nuclear factor κB
- NK cells
natural killer cells
- NPD1
neuroprotectin D1
- PD1
protectin D1
- PDX1
protectin DX1
- PLA2G2D
phospholipase A2 group IID
- RSV
respiratory syncytial virus
- RvD1‐RvD6
resolvins D1‐D6
- RvE1
resolvin E1
- RvE2
resolvin E2
- SAA
serum amyloid A
- SARS‐CoV‐2
severe acute respiratory syndrome Coronavirus‐2
- SPM
specialized proresolving mediator
- Th
T helper cells
- TLR4
toll‐like receptor 4
- TNF‐α
tumor necrosis factor α
1. INTRODUCTION
Upon the detection of an invading pathogen or injury, the body initiates the process of acute inflammation as a response. Neutrophils are rapidly recruited to the site of infection to aid in pathogen clearance and host tissue response. This phase of immune response, called “initiation”, is coordinated by many proinflammatory mediators, including leukotrienes, prostaglandins, bradykinin, cytokines, and others. In order to return infected or inflamed tissue to homeostasis, several processes must be completed. These processes include stopping the recruitment of granulocytes to the site; inducing macrophage phagocytosis to clear apoptotic immune cells, pathogens, and other debris; and restoring and repairing any damaged tissue. 1 , 2 For much of history, the second phase of inflammation, called “resolution”, was believed to be a passive process. We are now aware that resolution of inflammation is an active process. A class of proresolving mediators that are biosynthesized during the acute inflammatory response was discovered, called specialized proresolving mediators (SPMs). SPMs are lipid mediators that actively help in the resolution of inflammation. SPMs are involved in all resolution phase processes and are therefore essential to return any tissue to healthy working order. 3 , 4
SPMs help to stop the recruitment of granulocytes, bring in macrophages to clear apoptotic neutrophils and debris, and restore injured tissue homeostasis. 5 Without sufficient SPMs, acute inflammation can become chronic. It is now well established that chronic inflammation is an underlying factor in many diseases, from neurodegeneration to cardiovascular disease. 6 , 7
SPMs are enzymatically derived from long‐chain polyunsaturated fatty acids (LCPUFA) like arachidonic acid (AA), eicosapentaenoic acid (EPA), and docosahexaenoic acid (DHA). SPMs are generally classified as lipoxins (from AA), E‐series resolvins (from EPA), and D‐series resolvins, protectins, and maresins (from DHA) (Figure 1).
1.1. Lipoxins
Lipoxins are synthesized from AA via one of several lipoxygenase (LOX) enzymes, including 5‐, 12‐, and 15‐LOX, derived from leukocytes, platelets, and monocytes, respectively. Some pathways of 5‐LOX, as well as COX‐2, are involved in converting AA to the proinflammatory mediators, leukotrienes, and prostaglandins. 8 The stereochemical structure of the individual types of lipoxins is dependent on the distinct pathway utilized in their biosynthesis.
1.2. E‐series resolvins
The omega‐3 EPA is converted to 18(R)‐hydroxyEPA (18(R)‐HEPE) via CYP450 enzymes. 18(R)‐HEPE is further transformed to resolvins RvE1 and RvE2 via the 5‐LOX enzyme. Additionally, EPA can be converted into the biologically active S enantiomer, 18(S)‐RvE1 by acetylated COX enzymes. 8
1.3. D‐series resolvins, protectins, and maresins
The omega‐3 DHA is converted to D‐series resolvins (RvD1–RvD6), protectins, and maresins via 12‐ and 15‐LOX enzymes. DHA is acted on by 15‐LOX and then peroxidase to form 17(S)‐hydroxyDHA (17(S)‐HDHA), which is further converted by 5‐LOX to form the resolvins. The intermediate between DHA and 17(S)‐HDHA can also be converted to protectin D1 by hydrolase enzymes in the mucosal lining of the respiratory tract. 8 In addition, DHA can be converted to maresins via 12‐LOX action. 5
As mentioned, there are various isomers depending on which of several potential pathways is utilized; for example, the “aspirin‐triggered” D‐series resolvin isomers are synthesized when COX‐2 enzymes are nitrosylated or acetylated. 9
SPMs have distinct and overlapping roles in the immune response and tissue regeneration; the individual roles of the various isomers are outside the scope of this review but are elucidated in other review articles such as the ones cited here. 4 , 10 In general, when resolution of an inflammatory response is required, SPMs act on several different immune and tissue‐resident cells. The different SPMs interact with cognate receptors, such as the lipoxin A4 (LXA4) receptor ALX/FPR2 (a G protein‐coupled receptor) and the RvE1 receptor chemokine‐like receptor 1 (ERV1/CMKLR1), at nanomolar concentrations. 5 ALX/FPR2 is expressed on multiple immune cells, including eosinophils, neutrophils, T cells, monocytes, and others. This receptor can also interact with other SPMs, such as RvD1, 15‐epi‐LXA4, and 17‐epi‐RvD1. 3 The named resolvin D1 receptor, DRV1/GPR32, is also expressed in various immune cells such as neutrophils, monocytes, and lymphocytes. 3 Interestingly, some SPMs also appear to interact with proinflammatory receptors as partial agonists, such as RvE1's interaction with the receptor BLT1 to enhance neutrophil apoptosis. 11 This ability to act as partial agonists may offer an additional mechanism by which SPMs modulate proinflammatory signaling. 3
SPMs act on these various receptors to inhibit neutrophil infiltration and promote their clearance after apoptosis. SPMs augment macrophage functions to promote phagocytosis of apoptotic neutrophils, microorganisms, and debris. Other immune cells, such as eosinophils, natural killer (NK) cells, innate lymphoid cells, and lymphocytes are also affected by SPMs. 5 For example, DHA‐derived SPMs have been shown to be essential for modulating T helper cells (Th1 and Th17) recruitment and response, reducing their number and cytokine production to resolve chronic inflammation. 12 , 13 T helper lymphocytes are critically involved in ongoing inflammatory conditions. 14 In addition, SPMs also help to resolve inflammation by reducing levels of proinflammatory mediators and inducing the production of anti‐inflammatory mediators, as well as reducing levels of reactive oxygen and nitrogen species. 7 , 15 These SPM‐mediated effects help to resolve the inflammation and regenerate any damaged tissue so that the body can return to homeostasis. Under healthy conditions, this process effectively limits inflammatory responses. However, under conditions of severe infection, chronic inflammatory conditions, or other comorbidities, this process can be impaired and leads to chronic, unresolved inflammation.
In the context of infection, SPMs are involved in resolving the acute response to pathogen invasion. Under conditions where SPM biosynthesis is stunted or insufficient, such as in obese individuals or severe infections, or when there are defects in SPM receptor expression, immune responses can be stunted as well. 5 The SPM shortage and/or other defects in the resolution pathway can contribute to unresolved inflammation as well as impaired antibody production. 16 , 17 It can be hypothesized that exogenous administration of SPMs could help in resolution of unresolved inflammation. Thus far, this hypothesis has been tested mostly in preclinical settings
FIGURE 1.
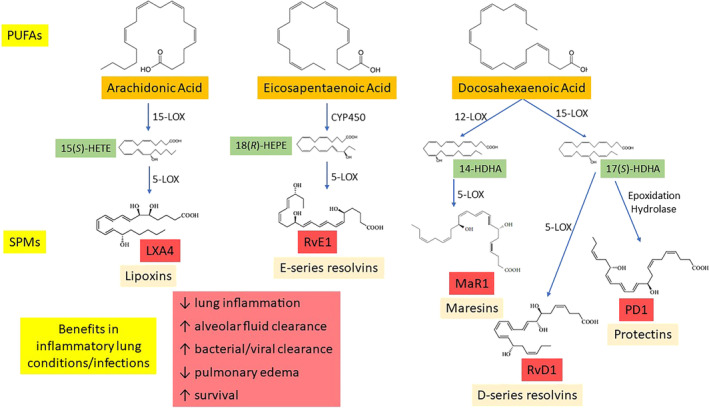
Brief overview of specialized proresolving mediator (SPM) biosynthesis and proresolving actions. SPMs are synthesized from essential polyunsaturated fatty acids via lipoxygenase enzymes and others. In infection and inflammatory lung conditions, SPMs act to resolve inflammation and improve outcomes. 14‐ and 17(S)‐HDHA, 14‐ and 17(S)‐hydroxydocosahexaenoic acid; 15(S)‐HETE, 15(S)‐hydroxyeicosatetraenoic acid; 18(R)‐HEPE, 18(R)‐hydroxyeicosapentaenoic acid; CYP450, cytochrome P450; LOX, lipoxygenase; PUFAs, polyunsaturated fatty acids
The 2019 novel Coronavirus disease (COVID‐19) is a pandemic viral infection that usually causes fever and respiratory symptoms such as coughing and difficulty breathing. Severe cases may progress to acute respiratory distress and other life‐threatening conditions. Patients with COVID‐19 tend to have higher leukocyte numbers and increased levels of proinflammatory cytokines. 18 Additionally, obese individuals are at a much higher risk of severe COVID‐19 infection than nonobese individuals. It has been demonstrated that obese versus nonobese individuals have markedly reduced levels SPMs, and it has been suggested that this deficit may be a reason behind the more severe course of COVID‐19 infection in obesity. 19 SPMs have the potential to aid in addressing common problems of bacterial and viral infections, such as antibiotic resistance, vaccine response, and more. They also have the ability to block leukocyte recruitment and resolve inflammation. SPMs have also been studied for their therapeutic role in lung conditions, like chronic obstructive pulmonary disease (COPD) and acute respiratory distress syndrome (ARDS). As some of the more distressing aspects of COVID‐19 are pneumonia‐like complications, acute respiratory distress, and excessive inflammatory response, 18 it is possible that SPMs may have a potential therapeutic role to play. 20 , 21 , 22 Therefore, this review will discuss the research surrounding SPMs and their role in infection with an emphasis on inflammatory lung conditions.
2. SPMs IN BACTERIAL INFECTIONS
SPMs have been studied in the context of many kinds of infections, including bacterial infections. It is clear that SPMs play a very important role in helping resolve inflammation caused by bacterial infection. The proper balance between proresolution mediators and proinflammatory mediators appears to be important in the body's response to infection or insult. During certain active bacterial infections, coadministration of SPMs has been shown to lower the doses of antibiotics necessary to clear infections, as well as aiding in the healing process even when administered alone. 23 SPMs therefore have great potential to help address the growing issue of antibiotic resistance. It is essential to fully elucidate the roles SPMs play in bacterial infections and understand the delicate balance between proinflammatory and resolving mediators in the context of treating bacterial infections.
2.1. Periodontitis
One area of bacterial infections that has been studied fairly extensively with SPMs is periodontitis. 24 , 25 Exogenous administration of SPMs has been shown in animal studies to dramatically aid in healing periapical lesions and reducing inflammation. 26 , 27 , 28 , 29 Periodontitis is generally caused by bacterial overgrowth, often Porphyromonas gingivalis, leading to biofilm formation and mucosal inflammation. An excessive immune response is believed to drive disease progression; and unresolved periodontitis and its resultant chronic inflammation can increase the risk for several diseases like atherosclerosis and Alzheimer's disease. 30 It is likely that periodontitis' excessive inflammation is the reason SPMs have been so successful in this area.
Multiple studies indicate that SPMs play a significant role in healing periodontitis: in rabbits that overexpressed 15‐lipoxygenase, an enzyme that participates in the synthesis of lipoxins and protectins, bone loss and local inflammation from periodontitis were markedly reduced. External application of the lipoxin LXA4 significantly reduced bone loss and leukocyte infiltration as well. 26 In another rabbit model of periodontitis, the resolvin RvE1 was used alone to completely heal the tissue and bone. In addition to the local effect, RvE1 also exerted systemic reduction in inflammation as shown with reduced C‐reactive protein levels. 27 The resolvin RvD2 also showed dramatic healing effects in rats with periodontitis, inducing calcification around the root and healing bone lesions. 29 In human periodontal ligament cells, LXA4 resolved inflammation through the TLR4/MyD88/NF‐κB signaling pathway. 31
Patients suffering from localized aggressive periodontitis were shown to have increased leukotriene (proinflammatory) levels and significantly lower SPM levels, including LXA4, 15‐hydroxyeicosatetraenoic acid (15‐HETE), and 14‐HDHA, than asymptomatic controls. The macrophages taken from the periodontitis patients also had impaired phagocytosis activity, which RvE1 rescued. These results indicate that the resolution phase is likely impaired in periodontitis patients, which allows the persistence of inflammation and disease progression. 16 In another study, periodontitis patients were shown to have lower LXA4 levels and higher protectin D1 (PD1) and maresin 1 (MaR1) than healthy controls. 32 These results indicate that a particular balance of SPMs may be necessary to overcome periodontal disease. Additionally, LXA4 levels were shown to be correlated with periodontal parameters and metabolic syndrome, suggesting an association between periodontitis and metabolic syndrome, as well as a protective role for LXA4 in these inflammatory conditions. 33
2.2. Sepsis
Sepsis is a global health concern with exceptionally high mortality rates. Unfortunately, the clinical management of sepsis is still less than optimal. Sepsis is caused by an imbalanced host immune response, with excessive inflammation and an inadequate resolution of the inflammation. 34 , 35 SPMs have therefore become an area of interest in the management of sepsis. 36 While anti‐inflammatory therapies could potentially harm septic patients, improving the resolution phase of inflammation appears to allow an adequate immune response and improve the clearance of the bacterial pathogen. 5
LXA4 in combination with antibiotics has been shown to be more effective than antibiotics alone in improving systemic inflammation and survival in mice with sepsis caused by Escherichia coli. 37 Even LXA4 alone was effective at reducing systemic inflammation and bacterial load as well as improving survival in a cecal ligation and puncture (CLP) mouse model of sepsis. The proposed mechanism was that LXA4 reduced the production of proinflammatory mediators via an NF‐κB‐dependent pathway and enhanced macrophage recruitment. 38 MaR1 has also been shown to improve survival in septic mice, improving organ function and reducing serum bacterial burden. The proposed mechanism also partly involved suppressing NF‐κB. 39 Another study on MaR1 in sepsis validated these results, adding that the protective effects of MaR1 could also be attributed to improving the metabolic dysfunction caused by sepsis and ameliorating mitochondrial damage. 40 The resolvin RvD2 and protectin DX (PDX1) were each also shown to improve survival in septic mice by increasing macrophage activity. 41 , 42 RvD1 was shown to protect mice from cardiac injury due to sepsis. 43 Other SPMs have improved survival in animal models as well. 44 In human sepsis patients, SPM levels correlate with survival and may potentially be used as biomarkers to stratify patient risk. 45 , 46
While SPMs have shown great promise in improving sepsis outcomes, one study suggested that a careful, well‐timed balance of proinflammatory and proresolving mediators is necessary to ensure a proper initiation and resolution to the body's immune response to insult. This was demonstrated in a mouse model of pneumosepsis induced with Klebsiella pneumoniae; when the mice were treated with LXA4 in early stages of sepsis, the infection worsened. When the mice were treated with LXA4 in later stages, it reduced the excessive inflammation and improved survival. 47 It is worth noting that one of the earliest measurable changes in an inflammatory response is a sharp increase in LXA4 that quickly wanes. LXA4 is again increased during the resolution phase. 48 The other studies discussed in this section have all shown beneficial effects of administering SPMs in early stages of sepsis and none reported adverse outcomes. It is possible that the different methods of inducing sepsis (CLP vs. K. pneumoniae) play a role in the different outcomes, however further study is needed. It bears emphasizing that the vast majority of sepsis studies have not indicated any cause for concern.
2.3. Tuberculosis
When discussing immune responses to infection, it is crucial to emphasize that both inadequate and excessive inflammation are detrimental. In the case of infections with Mycobacterium tuberculosis, a slow‐growing mycobacterium that can often cause latent infections, the Th1‐mediated immune response (proinflammatory) is essential. The Th2‐mediated response that is responsible for limiting inflammation appears to be almost inconsequential during tuberculosis infection. The Th1 response is significantly impacted by the balance between leukotriene B4 (LTB4, a proinflammatory mediator) and lipoxin A4. 49 If either side of the immune response is faulty or excessive, the host may be adversely impacted. 50
5‐lipoxygenase (5‐LOX), an enzyme involved in lipoxin and leukotriene production, was shown to play a significant role in tuberculosis infections in mice. During the acute response to infection (up to 10 days after being exposed), LXA4 and LTB4 levels were sharply increased. After the initial acute response, only LXA4 continued being synthesized in high amounts (the chronic stage of infection). In mice deficient in 5‐LOX (5‐lo −/− ), survival rates were increased following infection with M. tuberculosis. The deficient mice also had decreased bacterial burdens in their lungs compared to WT mice after 21 and 42 days, indicating that negative regulation of the Th1 response could be detrimental in tuberculosis infections. 49 In humans, variants of the genes that encode for 5‐LOX and LTA4 hydrolase (which can alter levels of lipoxins and leukotrienes) modulate the individual's susceptibility to tuberculosis infection, underscoring the importance of this balance in the tuberculosis immune response. 51 , 52
In patients with newly‐diagnosed active tuberculosis infections, higher levels of D‐series resolvins (RvD1 and RvD2) were detected compared to asymptomatic and presumably uninfected controls, indicating that these resolvins are involved in the acute phase of immune response to recently activated tuberculosis. 53 Levels of SPMs in the cerebrospinal fluids of patients with a more advanced form of the disease, tuberculosis meningitis, were associated with disease severity and mortality—those with severe disease had increased inflammatory eicosanoids and decreased SPMs, while those with higher levels of SPMs tended to survive through the 80 day study. The specific SPMs detected were 5‐LO‐derived, including RvT2 and RvT4, indicating these SPMs may be important for resolving the chronic stage of severe tuberculosis infection. 54 It could be possible that if SPMs were to be exogenously administered in the case of a tuberculosis infection, the timing would be important so as not to affect the initial Th1 immune response. Exogenous administration of several SPMs, including RvD1 and MaR1, reduced tuberculosis‐induced TNF‐α production and tuberculosis growth in infected human macrophages, indicating that they may potentially have therapeutic effects in tuberculosis‐induced inflammation. 55 However, this area of research still requires clarification and the effects of exogenous SPM administration has not been tested in humans or animal models of tuberculosis. Further study is necessary to clarify the role SPMs play in different stages of tuberculosis infection.
2.4. Other bacterial infections
SPMs have also been studied in the context of other bacterial infections, such as bacterial pneumonia (which will be discussed further in the sections below on lung conditions) and others. When comparing susceptible and resistant mouse models of bacterial keratitis induced by Pseudomonas aeruginosa, it was found that the 5/12/15‐LOX pathways were essential for resistance. Imbalances in the lipoxygenase enzymatic pathways contributed to susceptibility, as demonstrated by lower levels of 15‐HETE and 17‐hydroxydocosahexaenoic acid (17‐HDHA). 56 In infant mice with enteropathogenic Citrobacter rodentium infection, which was used as a model for enteropathogenic E. coli in humans, postinfection treatment with the resolvins RvD1 and RvD5 significantly reduced bacterial loads and rescued the infant mice from death. Even more strikingly, the treatment protected the mice from reinfection. 57
3. SPMs IN VIRAL INFECTIONS
As with bacterial infections, SPMs have been studied in the context of viral infections, although perhaps not as extensively. Nevertheless, SPMs have shown promise in improving viral outcomes in several preclinical models.
3.1. Influenza
SPMs may play a vital role in the response to influenza infection. In a study with mice, the highly virulent H5N1 virus was shown to cause dysregulation in the inflammatory response: the virus caused changes in the expression of genes responsible for biogenesis and signaling of lipoxins. 58 Protectins were shown to have protective effects against influenza in mice. PD1 levels were suppressed during severe infection and correlated with the pathogenicity of the H5N1 viruses. PD1 also inhibited viral replication by targeting the export of viral RNA. Prophylactic treatment with PD1 improved survival from severe infection. Importantly, exogenous treatment with PD1 improved lung function even at stages of infection where current antiviral drugs by themselves are no longer effective. Treating mice with an antiviral and PD1 2 days postinfection completely rescued mice from death. 59 , 60 Some researchers have pointed out that there is confusion surrounding PD1 in the literature, and the active lipid mediator may in fact be PDX, an isomer with a different route of biosynthesis. 61 It is possible that either PD1, PDX, or both are involved in blocking influenza.
Under conditions of obesity, immune responses may be blunted. In humans as in mice, obesity was associated with impaired B cell secretion and stimulation. When the obese mice were provided DHA in their diet, they had improved antibody titers in response to influenza infection. Importantly, the DHA did not directly target B cells; rather, DHA administration led to an increase in the SPMs 14‐HDHA, 17‐HDHA, and PDX. These SPMs helped boost B cell activity and increased antibody titers. 62 A mouse model of influenza vaccination demonstrated that coadministration of the SPM 17‐HDHA with the vaccine led to better outcomes: the mice given concurrent 17‐HDHA had increased antibody titers and enhanced protection against live H1N1 infection, indicating that SPMs may serve as potential flu vaccine adjuvants. 63 An in vitro study provided further evidence for SPMs' potential role as flu vaccine adjuvants; LXB4 was shown to boost memory B cell activation, which could help provide enhanced antibody‐mediated immune response. 64
3.2. Coronaviruses
To date, only one study has directly examined the relationship between proresolving mediators and Coronaviruses, specifically severe acute respiratory syndrome‐Coronavirus (SARS‐CoV). In the study, the age‐related increase in disease severity was explored in mice and determined to be at least partly dependent on the increased expression of phospholipase A2 (PLA2) group IID (PLA2G2D) in the aged mouse lungs. As mice age, the enzyme PLA2G2D expression increases. PLA2G2D is involved in the biosynthesis of SPMs, as it hydrolyzes phospholipids to release the LCPUFA that then undergo further metabolism to form SPMs. Under normal aging conditions, this helps address the increased inflammation due to aging. However, in this study, the increased PLA2G2D levels (and the corresponding increase in free AA, EPA, and DHA) in the lungs led to worse outcomes from SARS‐CoV infection. In middle‐aged mice that lacked the expression of PLA2G2D (Pla2g2d −/− ), survival was increased to more than 80%, while all the WT mice died. The authors conclude that higher basal levels of proresolving mediators in the lungs might be detrimental in the context of a lung infection, as it may dampen the initial immune response. 65 This appears to contradict to the numerous studies that have shown SPMs have many beneficial effects in infections, and particularly lung diseases, and to not be immunosuppressive. The higher levels of AA measured in the mouse lungs could also have led to higher leukotriene LTB4 levels, which may help explain the results. Unfortunately, the effect of exogenous administration of SPMs in SARS and other Coronavirus infections has not been tested yet and thus no firm conclusion can be reached.
Some authors posit that LCPUFA and their SPM derivatives may be effective against Coronaviruses like SARS‐CoV, MERS‐CoV, and SARS‐CoV‐2 (the novel Coronavirus that causes COVID‐19), as LCPUFA have shown inhibitory effects against enveloped viruses in vitro. 66 , 67 In addition, obese individuals have been shown to have SPM deficits, which may be at least partly responsible for their more severe course of COVID‐19 illness due to dysfunctional resolution of inflammation. 19 Further study is necessary before any conclusions can be drawn in this regard.
3.3. Herpes simplex virus
SPMs have been studied with other viral infections as well. In mice with herpes simplex virus (HSV)‐induced keratitis, topical administration of a prodrug form of neuroprotectin D1 (NPD1) helped reduce the severity of the keratitis by decreasing levels of proinflammatory mediators such as IL‐6 and several matrix metalloproteinases (MMPs), as well as dampening the recruitment of neutrophils to the cornea. 68 Similar results were seen in another study with mice with HSV‐induced keratitis; treatment with RvE1 controlled corneal lesion severity. RvE1 appeared to work similarly to NPD1, lowering neutrophil and Th1 recruitment to the cornea, increasing anti‐inflammatory IL‐10 levels, and inhibiting the production of proinflammatory mediators like IL‐6. 12
3.4. Respiratory syncytial virus
Respiratory syncytial virus (RSV) primarily targets airway epithelial cells and lung macrophages, inducing a strong inflammatory response. In 5‐lo −/− mice (mice lacking the 5‐LOX enzyme), RSV infection was worsened, and they had increased lung pathology from elevated COX‐2. Although this example is with a viral infection, it is interesting to note that this is the opposite effect that was seen in the tuberculosis mouse model, where 5‐lo −/− mice had improved outcomes. Treating macrophages lacking 5‐LOX with LXA4 and RvE1 partially counteracted the COX‐2 pathology, and treating RSV‐infected mice with a COX‐2 inhibitor enhanced lung 5‐LOX expression, suggesting that there was improved resolution of inflammation. 69
3.5. Other viruses
While SPMs have not been studied extensively in other viral conditions, there are preliminary data that indicate they do play a role in regulating the immune response to other viral infections. 70 Kaposi's sarcoma (KS) is a cancer that forms in the linings of blood vessels and is caused by Kaposi's sarcoma‐associated herpesvirus (KSHV). In a KS cell line, treatment with LXA4 or its epimer reduced levels of proinflammatory COX‐2, PGE2, LTB4, and IL‐6 and IL‐8 cytokines. Treatment also increased levels of the anti‐inflammatory IL‐10 as well as reducing angiogenic factors. 71 These results suggest that lipoxins may serve as a valuable therapeutic for KS. 72
In vitro studies indicate that SPMs appear to be involved in direct response to infection with other viruses, such as pneumonia virus of mouse and mouse adenovirus, 73 as well as HIV, 74 as viral infection increased the production of lipoxins, however these results have not been replicated or expounded upon in further studies to the best of our knowledge.
While SPMs clearly play an important role in resolving viral infections, the research is still in early phases and needs to be further elucidated.
4. SPMS IN INFLAMMATORY LUNG AND AIRWAY CONDITIONS AND INJURY
The role of SPMs in lung and airway diseases has been extensively studied in preclinical models. Conditions like ARDS, COPD, cystic fibrosis (CF), asthma, as well as respiratory illnesses due to infections as were touched on in previous sections, have all been explored. 3 SPMs have shown tremendous promise in many of these conditions, which is important as the standard of care in many cases is insufficient to improve disease outcomes. 75 , 76 , 77
4.1. Acute respiratory distress syndrome
ARDS is characterized by excessive inflammation in the lungs that causes life‐threatening respiratory failure. There are limited treatment options. ARDS can be caused by sterile injuries like burns, or by pathogens including bacteria and viruses. During ARDS, there is increased permeability of the alveolar endothelial and epithelial barriers, leading to edema and fluid buildup in the alveoli and impaired gas exchange. At times, excessive neutrophils penetrate the lungs and cause tissue injury. 3 SPMs have been shown to be important mediators in resolving preclinical models of lung injury.
One mechanism by which SPMs may aid in the resolution of ARDS is by regulating alveolar fluid clearance. 78 Under healthy conditions, ion transport across alveolar epithelial cells is responsible for creating the osmotic gradient necessary for fluid clearance and maintaining homeostasis. Under ARDS, the damaged barriers do not allow for effective fluid clearance. Additionally, excessive proinflammatory mediators like TNF‐α, and IL‐1β are present in pulmonary edema fluids and if left unchecked can promote alveolar injury. SPMs appear to be closely involved in keeping the excessive inflammation in check and allow for alveolar fluid clearance. In a rat model of acute lung injury, treatment with LXA4 stimulated alveolar fluid clearance and resolved pulmonary edema via activation of alveolar epithelial sodium channels. 79 In another rat model of acute lung injury, LXA4 helped to heal lung tissue by inhibiting IL‐6 and TNF‐α as well as improving alveolar fluid clearance. 80 RvD1 and MaR1 have also both been shown to improve fluid clearance in animal models of acute lung injury, at least partly via improving alveolar epithelial sodium channels as well as the PI3k signaling pathway that leads to Na,K‐ATPase activation. 81 , 82 In mice with lipopolysaccharide (LPS)‐induced acute lung injury, RvD1 treatment helped to protect the mice and improve lung pathology. The treated mice also had reduced TNF‐α and neutrophil infiltration into the lungs. 83 Additionally, PDX improved lung histopathology, reduced lung inflammation, and mitigated pulmonary edema in a mouse model of acute lung injury. 84
While further studies, and particularly clinical studies, are necessary, the preclinical data suggest that SPMs could offer a new therapeutic strategy to decrease the severity and duration of ARDS by improving alveolar fluid clearance and decreasing excessive inflammation.
4.2. Chronic obstructive pulmonary disease
COPD is a condition that arises from prolonged exposure to smoke or environmental toxins. Patients with COPD often have chronic lung inflammation and are susceptible to infection. 85 The standard treatment, inhaled corticosteroids with bronchodilators, is not particularly effective in many cases. COPD patients are specifically susceptible to persistent infection because of impaired macrophage ability to clear pathogens. During infection, COPD patients may exhibit an excessive neutrophil response that does more harm than good. Additionally, COPD patients exhibit altered fatty acid metabolism from airway epithelial remodeling, leading to lower levels of some SPMs. 86 , 87 SPMs have therefore been proposed as treatment options to dampen the excessive neutrophil response as well as address the imbalance between proinflammatory and proresolution mediators in COPD. 85
Serum amyloid A (SAA) is an apolipoprotein that is sharply increased during severe COPD infections. It is involved in recruiting neutrophils to the lungs, which under COPD becomes excessive. SAA and the proresolving mediator LXA4 both compete to bind to the G‐protein coupled receptor ALX/FPR2, and so they oppose each other's actions. 88 LXA4 blocks the SAA‐mediated recruitment of neutrophils to the lungs to help resolve the associated damage. 89
In studies with patient lung tissue samples, proresolving and metabolic pathways were disrupted. This indicates that exogenous administration of SPMs may help prevent the chronic inflammation and emphysema that arises from COPD. 90 Furthermore, in mice exposed to long‐term cigarette smoke, RvD1 treatment reduced the development of emphysema and inflammation. 90 RvD1 treatment in mice exposed to cigarette smoke also reduced inflammation in the lungs caused by neutrophil infiltration as well as increasing levels of the anti‐inflammatory IL‐10. 91 Similar results were seen with the aspirin triggered form of RvD1 (AT‐RvD1) in mice with cigarette‐induced emphysema. 92 In mice exposed to inhaled organic dust, MaR1 reduced neutrophil infiltration, IL‐6, and TNF‐α. 93
4.3. Pathogen‐mediated lung infections
Respiratory tract infections are caused by a multitude of viral and bacterial pathogens, such as Coronaviruses, hantaviruses, influenza, Staphylococcus aureus, and Streptococcus pneumoniae. Several infections also cause the host to become susceptible to a secondary coinfection. As mentioned in the sections on bacterial and viral infections, SPMs play an important role in resolving the inflammation caused by infection.
A mouse model of pneumonia triggered by bacterial infection demonstrated that RvE1, when administered before the injury, decreased neutrophil accumulation by more than 50% and helped clear the bacteria. Additionally, levels of proinflammatory cytokines were reduced and survival improved. 94 RvE1 was also shown to resolve acute lung inflammation in mice caused by bacterial infection by improving the phagocytical clearance of apoptotic neutrophils. 95 Other SPMs such as 15‐epi‐LXA4 and AT‐RvD1 have also shown effectiveness at resolving acute lung inflammation from bacteria and improving survival. 96 , 97 Importantly, AT‐RvD1 was shown to be effective at reducing the severity of pneumonia in a coinfection mouse model of S. pneumoniae and influenza A. The coinfected mice had increased circulating and lung levels of SAA as well as neutrophilic inflammation. The mice were then treated with AT‐RvD1 at the acute phase of infection and subsequently experienced a reduction in pneumococcal load and improvement in the severity of pneumonia. 98
4.4. Other lung conditions
SPMs have also been studied in other conditions that affect the lungs, such as CF, asthma, and allergies. 99 , 100 , 101 In CF, the lungs generally have persistent mucus secretions that can obstruct airways and cause susceptibility to infection. Chronic lung inflammation is common. 102 LXA4 levels were found to be suppressed in CF patient sputum, while those that had detectable levels of RvE1 also tended to have better lung function than those without detectable levels. 103 There is preclinical evidence that SPM treatment may help improve lung inflammation in CF and lead to less severe disease. 99 , 104 In a mouse model of CF, RvD1 treatment reduced P. aeruginosa burden in the lungs and improved disease scores. 101 Some clinical evidence indicates that boosting SPMs in CF patients may have some benefit as well; use of Acebilustat, a LTA4 hydrolase inhibitor that can limit leukotriene production and increase lipoxins, reduced neutrophil concentrations in patient sputum. 105 , 106
Asthma in humans is associated with reduced levels of SPMs, specifically LXA4 and PD1, when compared to healthy controls. 3 , 107 , 108 The first human trial with SPMs was conducted in asthmatic patients, where LXA4 inhalation was shown to shift the response curve to a bronchial inhalation challenger, LTC4. 109 Many other preclinical studies have demonstrated beneficial effects of SPMs on asthma and allergic inflammation. 3 , 5
Taken as a whole, the existing data on SPMs in lung conditions indicate that they are a highly promising category of new therapeutics that may help to limit excessive inflammation and improve disease pathology.
5. CONCLUSION
SPMs have become an increasingly popular area of research because of their widespread potential therapeutic value. As we have become aware of the dangers of unresolved inflammation in many disease states, the best ways to counter inflammation have been thoroughly explored. From anti‐inflammatory therapies to SPMs, quenching chronic inflammation has become an area of public health concern. The worry, however, that quenching inflammation could dampen the body's (helpful) immune response has been raised. SPMs are attractive options, as they are widely considered to not be immunosuppressive like many anti‐inflammatories and may expedite microbial clearance and activate the resolution of inflammation. 10
The overwhelming majority of preclinical models have shown treatment of inflammatory conditions with exogenous SPMs effectively promotes resolution of inflammation, thereby improving disease pathology, and enhancing survival. A few preclinical studies suggest that successful treatment with SPMs may depend on timing of SPM administration or basal SPM levels. It is possible that under certain conditions, allowing the body to launch an immune response before administering SPMs to resolve it would be advisable, although further research is necessary. In conditions that are characterized by established chronic inflammation and/or faulty resolution pathways, such as infections, ARDS, and COPD, SPM administration has been shown to be helpful in preclinical models.
While SPMs have not yet been directly studied in the context of COVID‐19, it is reasonable to suggest that they could be a valuable therapeutic option. One of the strongest risk factors of severe COVID‐19 illness, obesity, is linked to decreased SPM levels. In addition, many of the distressing aspects of COVID‐19, including the pneumonia‐like presentation, ARDS, and excessive inflammation and leukocyte number, have been shown to be improved in preclinical studies with SPMs. SPMs may be tested as adjuvants to a new vaccine or coadministered with antiviral drugs to hopefully improve patient outcome.
Taken as a whole, the research on SPMs as therapeutics is still fairly new. Preclinical models indicate that there is reason to be optimistic that SPMs can become useful tools to improve outcomes in pathogenic infections, lung diseases, and other inflammatory conditions.
CONFLICT OF INTEREST
Shayna Sandhaus and Andrew G. Swick are paid employees of Life Extension Inc.
ACKNOWLEDGMENTS
The authors would like to acknowledge Dr. Gerard Bannenberg and Dr. Jana Barlic‐Dicen for their critical review and suggestions, and Dr. Cristina Matthewman for helping design the graphical abstract.
Sandhaus S, Swick AG. Specialized proresolving mediators in infection and lung injury. BioFactors. 2021;47:6–18. 10.1002/biof.1691
DATA AVAILABILITY STATEMENT
Data sharing is not applicable to this article as no new data were created or analyzed in this study.
REFERENCES
- 1. Nathan C, Ding A. Nonresolving inflammation. Cell. 2010;140:871–882. [DOI] [PubMed] [Google Scholar]
- 2. Chen L, Deng H, Cui H, et al. Inflammatory responses and inflammation‐associated diseases in organs. Oncotarget. 2018;9:7204–7218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Krishnamoorthy N, Abdulnour R‐EE, Walker KH, Engstrom BD, Levy BD. Specialized proresolving mediators in innate and adaptive immune responses in airway diseases. Physiol Rev. 2018;98:1335–1370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Serhan CN, Chiang N, Dalli J. New pro‐resolving n‐3 mediators bridge resolution of infectious inflammation to tissue regeneration. Mol Aspects Med. 2018;64:1–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Basil MC, Levy BD. Specialized pro‐resolving mediators: Endogenous regulators of infection and inflammation. Nat Rev Immunol. 2016;16:51–67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Serhan CN. Pro‐resolving lipid mediators are leads for resolution physiology. Nature. 2014;510:92–101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Chiurchiù V, Leuti A, Maccarrone M. Bioactive lipids and chronic inflammation: Managing the fire within. Front Immunol. 2018;9:38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Serhan CN, Petasis NA. Resolvins and protectins in inflammation resolution. Chem Rev. 2011;111:5922–5943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Serhan CN, Chiang N. Endogenous pro‐resolving and anti‐inflammatory lipid mediators: A new pharmacologic genus. Br J Pharmacol. 2008;153(1):S200–S215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Dalli J, Serhan CN. Identification and structure elucidation of the pro‐resolving mediators provides novel leads for resolution pharmacology. Br J Pharmacol. 2019;176:1024–1037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Arita M, Ohira T, Sun YP, Elangovan S, Chiang N, Serhan CN. Resolvin E1 selectively interacts with leukotriene B4 receptor BLT1 and ChemR23 to regulate inflammation. J Immunol. 2007;178:3912–3917. [DOI] [PubMed] [Google Scholar]
- 12. Rajasagi NK, Reddy PBJ, Suryawanshi A, Mulik S, Gjorstrup P, Rouse BT. Controlling herpes simplex virus‐induced ocular inflammatory lesions with the lipid‐derived mediator resolvin E1. J Immunol. 2011;186:1735–1746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Chiurchiù V, Leuti A, Dalli J, et al. Proresolving lipid mediators resolvin D1, resolvin D2, and maresin 1 are critical in modulating T cell responses. Sci Transl Med. 2016;8:353ra111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Chiurchiù V, Leuti A, Saracini S, et al. Resolution of inflammation is altered in chronic heart failure and entails a dysfunctional responsiveness of T lymphocytes. FASEB. 2019;33:909–916. [DOI] [PubMed] [Google Scholar]
- 15. Leuti A, Maccarrone M, Chiurchiu V. Proresolving lipid mediators: Endogenous modulators of oxidative stress. Oxid Med Cell Longev. 2019;2019:8107265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Fredman G, Oh SF, Ayilavarapu S, Hasturk H, Serhan CN, Van Dyke TE. Impaired phagocytosis in localized aggressive periodontitis: Rescue by resolvin E1. PLoS One. 2011;6:e24422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Crouch M, Al‐Shaer A, Shaikh SR. Hormonal dysregulation and unbalanced specialized pro‐resolving mediator biosynthesis contribute toward impaired B cell outcomes in obesity. Mol Nutr Food Res. 2020;e1900924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Rothan HA, Byrareddy SN. The epidemiology and pathogenesis of Coronavirus disease (COVID‐19) outbreak. J Autoimmun. 2020;109:102433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Pal A, Gowdy KM, Oestreich KJ, Beck M, Shaikh SR. Obesity‐driven deficiencies of specialized pro‐resolving mediators may drive adverse outcomes during SARS‐CoV‐2 infection. Front Immunol. 2020;11:1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Hammock BD, Wang W, Gilligan MM, Panigrahy D. Eicosanoids: The overlooked storm in COVID‐19? Am J Pathol. 2020;190:1782–1788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Panigrahy D, Gilligan MM, Huang S, et al. Inflammation resolution: A dual‐pronged approach to averting cytokine storms in COVID‐19? Cancer Metastasis Rev. 2020;39:337–340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Sousa LP, Pinho V, Teixeira MM. Harnessing inflammation resolving‐based therapeutic agents to treat pulmonary viral infections: What can the future offer to COVID‐19? Br J Pharmacol. 2020;177:3898–3904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Chiang N, Fredman G, Bäckhed F, et al. Infection regulates pro‐resolving mediators that lower antibiotic requirements. Nature. 2012;484:524–528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Van Dyke TE. Shifting the paradigm from inhibitors of inflammation to resolvers of inflammation in periodontitis. J Periodontol. 2020;91:S19–S25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Van Dyke TE, Sima C. Understanding resolution of inflammation in periodontal diseases: Is chronic inflammatory periodontitis a failure to resolve? Periodontol. 2000;2020(82):205–213. [DOI] [PubMed] [Google Scholar]
- 26. Serhan CN, Jain A, Marleau S, et al. Reduced inflammation and tissue damage in transgenic rabbits overexpressing 15‐lipoxygenase and endogenous anti‐inflammatory lipid mediators. J Immunol. 2003;171:6856–6865. [DOI] [PubMed] [Google Scholar]
- 27. Hasturk H, Kantarci A, Goguet‐Surmenian E, et al. Resolvin E1 regulates inflammation at the cellular and tissue level and restores tissue homeostasis in vivo. J Immunol. 2007;179:7021–7029. [DOI] [PubMed] [Google Scholar]
- 28. Osorio Parra MM, Elangovan S, Lee CT. Specialized pro‐resolving lipid mediators in experimental periodontitis: A systematic review. Oral Dis. 2019;25:1265–1276. [DOI] [PubMed] [Google Scholar]
- 29. Siddiqui YD, Omori K, Ito T, et al. Resolvin D2 induces resolution of periapical inflammation and promotes healing of periapical lesions in rat periapical periodontitis. Front Immunol. 2019;10:307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Meghil MM, Cutler CW. Oral microbes and mucosal dendritic cells, "spark and flame" of local and distant inflammatory diseases. Int J Mol Sci. 2020;21:1643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Ali M, Yang F, Jansen JA, Walboomers XF. Lipoxin suppresses inflammation via the TLR4/MyD88/NF‐κB pathway in periodontal ligament cells. Oral Dis. 2020;26:429–438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Tobón‐Arroyave SI, Isaza‐Guzmán DM, Gómez‐Ortega J, Flórez‐Alzate AA. Salivary levels of specialized pro‐resolving lipid mediators as indicators of periodontal health/disease status. J Clin Periodontol. 2019;46:978–990. [DOI] [PubMed] [Google Scholar]
- 33. Doğan ESK, Doğan B, Fentoğlu Ö, Kırzıoğlu FY. The role of serum lipoxin A4 levels in the association between periodontal disease and metabolic syndrome. J Periodontal Implant Sci. 2019;49:105–113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Shukla P, Rao GM, Pandey G, et al. Therapeutic interventions in sepsis: Current and anticipated pharmacological agents. Br J Pharmacol. 2014;171:5011–5031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Boeddha NP, Bycroft T, Nadel S, Hazelzet JA. The inflammatory and hemostatic response in sepsis and meningococcemia. Crit Care Clin. 2020;36:391–399. [DOI] [PubMed] [Google Scholar]
- 36. Buechler C, Pohl R, Aslanidis C. Pro‐resolving molecules‐new approaches to treat sepsis? Int J Mol Sci. 2017;18:476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Ueda T, Fukunaga K, Seki H, et al. Combination therapy of 15‐epi‐lipoxin A4 with antibiotics protects mice from Escherichia coli‐induced sepsis*. Crit Care Med. 2014;42:288–295. [DOI] [PubMed] [Google Scholar]
- 38. Walker J, Dichter E, Lacorte G, et al. Lipoxin a4 increases survival by decreasing systemic inflammation and bacterial load in sepsis. Shock. 2011;36:410–416. [DOI] [PubMed] [Google Scholar]
- 39. Li R, Wang Y, Ma Z, et al. Maresin 1 mitigates inflammatory response and protects mice from sepsis. Mediators Inflamm. 2016;2016:3798465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Hao Y, Zheng H, Wang RH, et al. Maresin1 alleviates metabolic dysfunction in septic mice: A (1)H NMR‐based metabolomics analysis. Mediators Inflamm. 2019;2019:2309175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Spite M, Norling LV, Summers L, et al. Resolvin D2 is a potent regulator of leukocytes and controls microbial sepsis. Nature. 2009;461:1287–1291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Xia H, Chen L, Liu H, et al. Protectin DX increases survival in a mouse model of sepsis by ameliorating inflammation and modulating macrophage phenotype. Sci Rep. 2017;7:99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Wang M, Liu M, Zhang J, et al. Resolvin D1 protects against sepsis‐induced cardiac injury in mice. BioFactors. 2020;46: 766–776. [DOI] [PubMed] [Google Scholar]
- 44. Lee S, Nakahira K, Dalli J, et al. NLRP3 Inflammasome deficiency protects against microbial sepsis via increased lipoxin B(4) synthesis. Am J Respir Crit Care Med. 2017;196:713–726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Dalli J, Colas RA, Quintana C, et al. Human sepsis eicosanoid and proresolving lipid mediator temporal profiles: Correlations with survival and clinical outcomes. Crit Care Med. 2017;45:58–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Padovan MG, Norling LV. Pro‐resolving lipid mediators in sepsis and critical illness. Curr Opin Clin Nutr Metab Care. 2020;23:76–81. [DOI] [PubMed] [Google Scholar]
- 47. Sordi R, Menezes‐de‐Lima O Jr, Horewicz V, Scheschowitsch K, Santos LF, Assreuy J. Dual role of lipoxin A4 in pneumosepsis pathogenesis. Int Immunopharmacol. 2013;17:283–292. [DOI] [PubMed] [Google Scholar]
- 48. Bannenberg GL, Chiang N, Ariel A, et al. Molecular circuits of resolution: Formation and actions of resolvins and protectins. J Immunol. 2005;174:4345–4355. [DOI] [PubMed] [Google Scholar]
- 49. Bafica A, Scanga CA, Serhan C, et al. Host control of Mycobacterium tuberculosis is regulated by 5‐lipoxygenase‐dependent lipoxin production. J Clin Invest. 2005;115:1601–1606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Tobin DM, Roca FJ, Oh SF, et al. Host genotype‐specific therapies can optimize the inflammatory response to mycobacterial infections. Cell. 2012;148:434–446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Herb F, Thye T, Niemann S, et al. ALOX5 variants associated with susceptibility to human pulmonary tuberculosis. Hum Mol Genet. 2008;17:1052–1060. [DOI] [PubMed] [Google Scholar]
- 52. Tobin DM, Vary JC Jr, Ray JP, et al. The lta4h locus modulates susceptibility to mycobacterial infection in zebrafish and humans. Cell. 2010;140:717–730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Frediani JK, Jones DP, Tukvadze N, et al. Plasma metabolomics in human pulmonary tuberculosis disease: A pilot study. PLoS One. 2014;9:e108854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Colas RA, Nhat LTH, Thuong NTT, et al. Proresolving mediator profiles in cerebrospinal fluid are linked with disease severity and outcome in adults with tuberculous meningitis. FASEB. 2019;33:13028–13039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Ruiz A, Sarabia C, Torres M, Juárez E. Resolvin D1 (RvD1) and maresin 1 (Mar1) contribute to human macrophage control of M. tuberculosis infection while resolving inflammation. Int Immunopharmacol. 2019;74:105694. [DOI] [PubMed] [Google Scholar]
- 56. Carion TW, Greenwood M, Ebrahim AS, et al. Immunoregulatory role of 15‐lipoxygenase in the pathogenesis of bacterial keratitis. FASEB. 2018;32:5026–5038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Diaz LA, Altman NH, Khan WN, Serhan CN, Adkins B. Specialized proresolving mediators rescue infant mice from lethal Citrobacter rodentium infection and promote immunity against reinfection. Infect Immun. 2017;85:e00464–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Cilloniz C, Pantin‐Jackwood MJ, Ni C, et al. Lethal dissemination of H5N1 influenza virus is associated with dysregulation of inflammation and lipoxin signaling in a mouse model of infection. J Virol. 2010;84:7613–7624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Morita M, Kuba K, Ichikawa A, et al. The lipid mediator protectin D1 inhibits influenza virus replication and improves severe influenza. Cell. 2013;153:112–125. [DOI] [PubMed] [Google Scholar]
- 60. Imai Y. Role of omega‐3 PUFA‐derived mediators, the protectins, in influenza virus infection. Biochim Biophys Acta. 2015;1851:496–502. [DOI] [PubMed] [Google Scholar]
- 61. Balas L, Guichardant M, Durand T, Lagarde M. Confusion between protectin D1 (PD1) and its isomer protectin DX (PDX). An overview on the dihydroxy‐docosatrienes described to date. Biochimie. 2014;99:1–7. [DOI] [PubMed] [Google Scholar]
- 62. Kosaraju R, Guesdon W, Crouch MJ, et al. Cell activity is impaired in human and mouse obesity and is responsive to an essential fatty acid upon murine influenza infection. J Immunol. 2017;198:4738–4752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Ramon S, Baker SF, Sahler JM, et al. The specialized proresolving mediator 17‐HDHA enhances the antibody‐mediated immune response against influenza virus: a new class of adjuvant? J Immunol. 2014;193:6031–6040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Kim N, Lannan KL, Thatcher TH, Pollock SJ, Woeller CF, Phipps RP. Lipoxin B(4) enhances human memory B cell antibody production via upregulating cyclooxygenase‐2 expression. J Immunol. 2018;201:3343–3351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Vijay R, Hua X, Meyerholz DK, et al. Critical role of phospholipase A2 group IID in age‐related susceptibility to severe acute respiratory syndrome‐CoV infection. J Exp Med. 2015;212:1851–1868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Das UN. Arachidonic acid and other unsaturated fatty acids and some of their metabolites function as endogenous antimicrobial molecules: A review. J Adv Res. 2018;11:57–66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Das UN. Can bioactive lipids inactivate Coronavirus (COVID‐19)? Arch Med Res. 2020;51:282–286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Rajasagi NK, Reddy PBJ, Mulik S, Gjorstrup P, Rouse BT. Neuroprotectin D1 reduces the severity of herpes simplex virus‐induced corneal immunopathology. Invest Ophthalmol Vis Sci. 2013;54:6269–6279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Shirey KA, Lai W, Pletneva LM, et al. Role of the lipoxygenase pathway in RSV‐induced alternatively activated macrophages leading to resolution of lung pathology. Mucosal Immunol. 2014;7:549–557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Russell CD, Schwarze J. The role of pro‐resolution lipid mediators in infectious disease. Immunology. 2014;141:166–173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Marginean A, Sharma‐Walia N. Lipoxins exert antiangiogenic and anti‐inflammatory effects on Kaposi's sarcoma cells. Transl Res. 2015;166:111–133. [DOI] [PubMed] [Google Scholar]
- 72. Chandrasekharan JA, Sharma‐Walia N. Arachidonic acid derived lipid mediators influence Kaposi's sarcoma‐associated herpesvirus infection and pathogenesis. Front Microbiol. 2019;10:358–358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Kim SJ. Elevated formation of lipoxins in viral antibody‐positive rat alveolar macrophages. Am J Respir Cell Mol Biol. 1990;3:113–118. [DOI] [PubMed] [Google Scholar]
- 74. Genis P, Jett M, Bernton EW, et al. Cytokines and arachidonic metabolites produced during human immunodeficiency virus (HIV)‐infected macrophage‐astroglia interactions: Implications for the neuropathogenesis of HIV disease. J Exp Med. 1992;176:1703–1718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Roberts CM, Lowe D, Bucknall CE, Ryland I, Kelly Y, Pearson MG. Clinical audit indicators of outcome following admission to hospital with acute exacerbation of chronic obstructive pulmonary disease. Thorax. 2002;57:137–141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Chung KF, Wenzel SE, Brozek JL, et al. International ERS/ATS guidelines on definition, evaluation and treatment of severe asthma. Eur Respir J. 2014;43:343–373. [DOI] [PubMed] [Google Scholar]
- 77. Bellani G, Laffey JG, Pham T, et al. Epidemiology, patterns of care, and mortality for patients with acute respiratory distress syndrome in intensive care units in 50 countries. JAMA. 2016;315:788–800. [DOI] [PubMed] [Google Scholar]
- 78. Wang Q, Yan S‐F, Hao Y, Jin S‐W. Specialized pro‐resolving mediators regulate alveolar fluid clearance during acute respiratory distress syndrome. Chin Med J. 2018;131:982–989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79. Wang Q, Lian QQ, Li R, et al. Lipoxin A(4) activates alveolar epithelial sodium channel, Na,K‐ATPase, and increases alveolar fluid clearance. Am J Respir Cell Mol Biol. 2013;48:610–618. [DOI] [PubMed] [Google Scholar]
- 80. Yang Y, Cheng Y, Lian QQ, et al. Contribution of CFTR to alveolar fluid clearance by lipoxin A4 via PI3K/Akt pathway in LPS‐induced acute lung injury. Mediators Inflamm. 2013;2013:862628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81. Wang Q, Zheng X, Cheng Y, et al. Resolvin D1 stimulates alveolar fluid clearance through alveolar epithelial sodium channel, Na,K‐ATPase via ALX/cAMP/PI3K pathway in lipopolysaccharide‐induced acute lung injury. J Immunol. 2014;192:3765–3777. [DOI] [PubMed] [Google Scholar]
- 82. Zhang JL, Zhuo XJ, Lin J, et al. Maresin1 stimulates alveolar fluid clearance through the alveolar epithelial sodium channel Na,K‐ATPase via the ALX/PI3K/Nedd4‐2 pathway. Laboratory investigation. J Tech Methods Pathol. 2017;97:543–554. [DOI] [PubMed] [Google Scholar]
- 83. Zhang HW, Wang Q, Mei HX, et al. RvD1 ameliorates LPS‐induced acute lung injury via the suppression of neutrophil infiltration by reducing CXCL2 expression and release from resident alveolar macrophages. Int Immunopharmacol. 2019;76:105877. [DOI] [PubMed] [Google Scholar]
- 84. Tan W, Chen L, Wang YX, et al. Protectin DX exhibits protective effects in mouse model of lipopolysaccharide‐induced acute lung injury. Chin Med J. 2018;131:1167–1173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85. Wang H, Anthony D, Selemidis S, Vlahos R, Bozinovski S. Resolving viral‐induced secondary bacterial infection in COPD: A concise review. Front Immunol. 2018;9:2345–2345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86. Thatcher TH, Woeller CF, McCarthy CE, Sime PJ. Quenching the fires: Pro‐resolving mediators, air pollution, and smoking. Pharmacol Ther. 2019;197:212–224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87. van der Does AM, Heijink M, Mayboroda OA, et al. Dynamic differences in dietary polyunsaturated fatty acid metabolism in sputum of COPD patients and controls. Biochim Biophys Acta Mol Cell Biol Lipids. 2019;1864:224–233. [DOI] [PubMed] [Google Scholar]
- 88. Bozinovski S, Anthony D, Anderson GP, Irving LB, Levy BD, Vlahos R. Treating neutrophilic inflammation in COPD by targeting ALX/FPR2 resolution pathways. Pharmacol Ther. 2013;140:280–289. [DOI] [PubMed] [Google Scholar]
- 89. Bozinovski S, Uddin M, Vlahos R, et al. Serum amyloid a opposes lipoxin a₄ to mediate glucocorticoid refractory lung inflammation in chronic obstructive pulmonary disease. Proc Natl Acad Sci USA. 2012;109:935–940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90. Hsiao HM, Thatcher TH, Colas RA, Serhan CN, Phipps RP, Sime PJ. Resolvin D1 reduces emphysema and chronic inflammation. Am J Pathol. 2015;185:3189–3201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91. Hsiao HM, Sapinoro RE, Thatcher TH, et al. A novel anti‐inflammatory and pro‐resolving role for resolvin D1 in acute cigarette smoke‐induced lung inflammation. PLoS One. 2013;8:e58258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92. Posso SV, Quesnot N, Moraes JA, et al. AT‐RVD1 repairs mouse lung after cigarette smoke‐induced emphysema via downregulation of oxidative stress by NRF2/KEAP1 pathway. Int Immunopharmacol. 2018;56:330–338. [DOI] [PubMed] [Google Scholar]
- 93. Nordgren TM, Bauer CD, Heires AJ, et al. Maresin‐1 reduces airway inflammation associated with acute and repetitive exposures to organic dust. Transl Res. 2015;166:57–69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94. Seki H, Fukunaga K, Arita M, et al. The anti‐inflammatory and proresolving mediator resolvin E1 protects mice from bacterial pneumonia and acute lung injury. J Immunol. 2010;184:836–843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95. El Kebir D, Gjorstrup P, Filep JG. Resolvin E1 promotes phagocytosis‐induced neutrophil apoptosis and accelerates resolution of pulmonary inflammation. Proc Natl Acad Sci USA. 2012;109:14983–14988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96. El Kebir D, József L, Pan W, et al. 15‐epi‐lipoxin A4 inhibits myeloperoxidase signaling and enhances resolution of acute lung injury. Am J Respir Crit Care Med. 2009;180:311–319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97. Sekheri M, El Kebir D, Edner N, Filep JG. 15‐epi‐LXA(4) and 17‐epi‐RvD1 restore TLR9‐mediated impaired neutrophil phagocytosis and accelerate resolution of lung inflammation. Proc Natl Acad Sci USA. 2020;117:7971–7980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98. Wang H, Anthony D, Yatmaz S, et al. Aspirin‐triggered resolvin D1 reduces pneumococcal lung infection and inflammation in a viral and bacterial coinfection pneumonia model. Clin Sci. 2017;131:2347–2362. [DOI] [PubMed] [Google Scholar]
- 99. Duvall MG, Bruggemann TR, Levy BD. Bronchoprotective mechanisms for specialized pro‐resolving mediators in the resolution of lung inflammation. Mol Aspects Med. 2017;58:44–56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100. Philippe R, Urbach V. Specialized pro‐resolving lipid mediators in cystic fibrosis. Int J Mol Sci. 2018;19:2865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101. Isopi E, Mattoscio D, Codagnone M, et al. Resolvin D1 reduces lung infection and inflammation activating resolution in cystic fibrosis. Front Immunol. 2020;11:581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102. Teopompi E, Risé P, Pisi R, et al. Arachidonic acid and docosahexaenoic acid metabolites in the airways of adults with cystic fibrosis: Effect of docosahexaenoic acid supplementation. Front Pharmacol. 2019;10:938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103. Yang J, Eiserich JP, Cross CE, Morrissey BM, Hammock BD. Metabolomic profiling of regulatory lipid mediators in sputum from adult cystic fibrosis patients. Free Radic Biol Med. 2012;53:160–171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104. Karp CL, Flick LM, Park KW, et al. Defective lipoxin‐mediated anti‐inflammatory activity in the cystic fibrosis airway. Nat Immunol. 2004;5:388–392. [DOI] [PubMed] [Google Scholar]
- 105. Elborn JS, Horsley A, MacGregor G, et al. Phase I studies of Acebilustat: Biomarker response and safety in patients with cystic fibrosis. Clin Transl Sci. 2017;10:28–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106. Recchiuti A, Mattoscio D, Isopi E. Roles, actions, and therapeutic potential of specialized pro‐resolving lipid mediators for the treatment of inflammation in cystic fibrosis. Front Pharmacol. 2019;10:252–252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107. Levy BD, Kohli P, Gotlinger K, et al. Protectin D1 is generated in asthma and dampens airway inflammation and hyperresponsiveness. J Immunol. 2007;178:496–502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108. Kazani S, Planaguma A, Ono E, et al. Exhaled breath condensate eicosanoid levels associate with asthma and its severity. J Allergy Clin Immunol. 2013;132:547–553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109. Christie PE, Spur BW, Lee TH. The effects of lipoxin A4 on airway responses in asthmatic subjects. Am Rev Respir Dis. 1992;145:1281–1284. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data sharing is not applicable to this article as no new data were created or analyzed in this study.


