Abstract
Comorbid post-traumatic stress disorder with traumatic brain injury (TBI) produce more severe affective and cognitive deficits than PTSD or TBI alone. Both PTSD and TBI produce long-lasting neuroinflammation, which may be a key underlying mechanism of the deficits observed in co-morbid TBI/PTSD. We developed a model of co-morbid TBI/PTSD by combining the closed head (CHI) model of TBI with the chronic variable stress (CVS) model of PTSD and examined multiple behavioral and neuroinflammatory outcomes. Male C57/Bl6 mice received sham treatment, CHI, CVS, CHI then CVS (CHI → CVS) or CVS then CHI (CVS → CHI). The CVS → CHI group had deficits in Barnes maze or active place avoidance not seen in the other groups. The CVS → CHI, CVS and CHI → CVS groups displayed increased basal anxiety level, based on performance on elevated plus maze. The CVS → CHI had impaired performance on Barnes Maze, and Active Place Avoidance. These performance deficits were strongly correlated with increased hippocampal Iba-1 level an indication of activated MP/MG. These data suggest that greater cognitive deficits in the CVS → CHI group were due to increased inflammation. The increased deficits and neuroinflammation in the CVS → CHI group suggest that the order by which a subject experiences TBI and PTSD is a major determinant of the outcome of brain injury in co-morbid TBI/PTSD.
Keywords: behavior, cognition, memory, astrocytes, macrophage/microglia
Introduction
Post-traumatic stress disorder (PTSD) and traumatic brain injury (TBI) are major health concerns in America (Tanev et al., 2014; Motzkin and Koenigs, 2015; Mikolić et al., 2019). Both diseases are common; approximately 25 million Americans have PTSD and there are 2.6 million new cases of TBI annually (http://www.cdc.gov/TraumaticBrainInjury/data/index.html). Multiple studies have shown that TBI increases the probability of developing PTSD, yet the underlying mechanisms are unknown (Bryant, 2011; Yurgil et al., 2014; Spadoni et al., 2018).
TBI results from blunt impact, explosive blast, or an object penetrating the skull (Cernak and Noble-Haeusslein, 2010; Corps et al., 2015). A wide spectrum of deficits arise from TBI that produces can effect motor function, cognition, memory and affect (Cernak and Noble-Haeusslein, 2010; Gould et al., 2011; Silver et al., 2018). Exposure to a psychologically traumatic event can produce PTSD. The Diagnostic and Statistical Manual of Mental Disorders 5th edition, defines PTSD as persistent hypervigilance, avoidance/numbing, intrusive thoughts and negative alterations in cognition and mood. PTSD is a chronic maladaptive fear potentiated response, in response to a traumatic event, lasting more than 30 days (Watkins et al., 2018). PTSD is a proinflammatory condition, involving alterations in neurotransmission and glucocorticoid functioning, and increased output of cytokines including IL-1ß, IL-6, and tumor necrosis factor alpha (TNF α) (Jones and Thomsen, 2013; Mendoza et al., 2016). TBI and PTSD clinical presentation can be difficult to distinguish, for they produce complex and overlapping affective and cognitive deficits. More specifically, there is a great number of common clinical symptoms between postconcussive symptoms (PCS), the long term clinical sequalae post mTBI, and PTSD, which include hyper-arousal, memory and cognitive deficits, as well as emotional dysregulation. The overlapping clinical sequelae of TBI and PTSD can potentially pose a diagnostic challenge for clinicians in care of patients who have experienced both. This is of special relevance in the combat veteran population (Bryant, 2011; Hendrickson et al., 2018; Porter et al., 2018). Hence, the prevalence of comorbid TBI-PTSD is largely unknown (Vasterling et al., 2018).
Neuroinflammation that accompanies TBI or PTSD is thought to be both beneficial and deleterious (Amos et al., 2014; Bergold, 2016). Anti-inflammatory drugs have had varying efficacy with both diseases (Amos et al., 2014; Bergold, 2016). In the case of TBI, both preclinical and clinical studies have reported efficacy of an anti-inflammatory regimen including minocycline, N-acetylcysteine (NAC) and statins (Bergold, 2016). It is important to note that the aforementioned therapeutic regimen have only demonstrated efficacy in the preclinical studies, and have not shown disease modifying effects in the clinical studies (Mohamadpour et al., 2019) Clinical studies using angiotensin converting enzyme inhibitor (ACEI) and angiotensin receptor blockers (ARBs), which modulate neuroinflammatory response, has been shown to be efficacious in treatment of PTSD (Khoury et al., 2012). Inflammation post injury involves a complex cascade of infiltration of immune cells and astrocytes, in turn leading to production of various cytokines and chemokines. These compounds, in turn, lead to recruitment and morphological alteration of native microglia (Lozano et al., 2015). Alteration of neuroinflammatory response in different treatment conditions, however, provides a potential mechanism how TBI and PTSD could interact (Aungst et al., 2014; Simon et al., 2017; Kaplan et al., 2018). An animal model of co-morbid TBI and PTSD could reveal potential relationships between altered neuroinflammation and behavioral deficits (Shultz et al., 2017; Perez-Garcia et al., 2019).
This study introduces a model of co-morbid TBI and PTSD that combines a closed head injury (CHI) model of TBI with the chronic variable stress (CVS) model of PTSD (Ostrander et al., 2006; Grin’kina et al., 2016). Mice receiving both CVS and CHI were compared to mice receiving CHI or CVS alone. An effect of treatment order was also assessed by administering CVS followed by CHI (CVS → CHI) or CHI followed by CVS (CHI → CVS). CHI is a closed head model of TBI which avoids the confounds of injuring the brain through a craniotomy (Grin’kina et al., 2016). The CVS model was used to study PTSD. In the CVS model, mice were randomly exposed daily to two of the following insults; cold water swim, transient restraint, overnight food deprivation, 45-degree cage tilt, and wet cage for 4 days. The CVS model is considered to be a more stressful preclinical model of PTSD models, in turn simulating a more clinically relevant phenotype (Goswami et al., 2013; Whitaker et al., 2014; Deslauriers et al., 2018).
Mouse cognition and memory are tested using Barnes maze and active place avoidance (Rupprecht et al., 2010) following CHI, CVS, or the combination of the two. The intrinsic tendency of mice to leave open spaces is the motivation to escape the brightly lit surface of Barnes maze using distal visual cues (Barnes, 1979; Kim et al., 2017). In APA, mice learn how to avoid a stationary shock zone located on a rotating area (Abdel Baki et al., 2009; Sangobowale et al., 2018). APA requires the functioning of both hippocampi and the axonal tracts connecting them, while Barnes maze can be acquired with only one hippocampus (Cimadevilla et al., 2001; Grin’kina et al., 2013). These data suggest that more brain regions need to function or coordinate properly to acquire APA than Barnes maze.
The affective state of mice was assessed using elevated plus maze and fear-potentiated startle. Elevated plus maze assessed basal anxiety by measuring the intrinsic response of mice to avoid the open arms of a behavioral apparatus (Walf and Frye, 2007; Goodwill et al., 2019). Acoustic Startle Response measured fear-potentiated anxiety by measuring the response of a mouse to differing intensity sounds (Grillon et al., 2017; Lezak et al., 2017).
This study investigated whether a model of comorbid TBI-PTSD using CHI or CVS produced greater cognitive or affective impairments and neuroinflammation than CHI or CVS alone. Mice receiving CVS → CHI had significantly more behavioral impairments and neuroinflammation than the other treatment groups. Hippocampal neuroinflammation correlated to behavioral deficits.
Material and Methods
Male mice (C57Bl/6, 26-28 g, 60-90 days old, Jackson Laboratories, Bar Harbor, Maine) were housed in groups of 3. Mice received LabDiet Brand 5053 Irradiated PicoLab chow (LabDiet St. Louis, MO) and water ad libitum. Mice were randomly divided into 5 groups: sham (n = 12), CHI (n = 6), CVS (n = 10), CHI → CVS (n = 8) and CVS → CHI (n = 7). Mice were transported in group cages from the animal facility to the room containing the behavioral tests. During behavioral tests, all mice actively tested were individually housed in a room adjacent to the testing room. At the completion of testing, mice were transported back to the animal facility in group cages. On the third day of Barnes maze testing, mice received one day of elevated plus maze testing, after the fourth day of Barnes maze testing, mice received two days of APA. Mice were tested on acoustic startle response on the day after APA. Thus, the time interval between tests was standard among all of the groups. All manipulations and behavioral testing were performed between 10 AM and 4PM. This study was carried out in strict accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health. The Institutional Animal Care and Use Committee of SUNY-Downstate Medical Center approved the experimental protocol used in this study (Protocol Number: 15-10480).
Closed Head Injury
Closed head injury (CHI) was done as described by Sangobowale, et al (Sangobowale et al., 2018). A baseline weight was obtained prior to sham-CHI or CHI. Deep anesthesia was induced for 3 minutes with isoflurane (3.5% in oxygen (1.0 L/min) and maintained (3% in oxygen (1.0 L/min) until immediately after the impact. The head of the mouse was shaved and closed head injury produced using a 5.0 mm diameter tip controlled by an electromagnetic impactor (Leica Microsystems, Buffalo Grove, IL). The 5 mm diameter impactor tip was placed 2 mm lateral from the midline and 5 mm caudal from the eyes and produced a single 6.3 m/s impact on the scalp to a depth of 3 mm with a 1-s dwell time. Mice receiving the sham-CHI were shaved, anesthetized, and placed into the impactor, but no impact was delivered. Mice received 10 minutes of anesthesia throughout the entire CHI procedure. After the impact, if a mouse did not spontaneously breath within 30 s, cardiopulmonary resuscitation was initiated with the mouse is breathing 100% O2. Slight differences in head motion during CHI can result in a heterogenous brain injury (Grin’kina et al., 2016). Immediately after spontaneous breathing, the mouse was placed on its back and the time to right itself measured. Injury severity was predicted by the time interval between the injury and restoration of righting reflex (Grin’kina et al., 2013). Following assessment of the righting reflex, mice were returned to their home cages. CHI produced transient motor deficits, therefore, mice were allowed to recover for 7 days in their home cages prior to behavioral testing (Grin’kina et al., 2013).
Chronic Variable Stress
PTSD was studied using the chronic variable stress (CVS) model (Ostrander et al., 2006). CVS exposes mice to 5 different aversive stimuli over the span of 4 days. The aversive stimuli included: 3-minute cold water swim at 16–18°C, overnight food deprivation with water provided ad libitum, a 15 minutes restraint in a flat-bottomed cylindrical open-ended restraint chamber (Braintree Scientific Instruments), three hours in a cage with 300 ml of water added to the cage bedding, or a three-hour exposure to a caged tilted at 45 degrees. The mice received a set of two pre-randomized aversive stimuli on a given day in the 4-day period, simulating the unpredictable nature of psychological trauma while minimizing the probability of habituation. Mice receiving the sham-CVS portion of the combined sham CHI and CVS procedure were brought from the animal facility to the laboratory and placed next to the stressor devices but did not receive the aversive stimuli. A single mouse receiving sham CVS portion of the sham procedure was in the room with a single mouse receiving an aversive stimulus.
Barnes Maze
Mice were tested on Barnes maze as described by Sunyer et al. (2007). The apparatus was a circular platform (92 cm diameter) elevated 105 cm above the floor with 20 equally spaced 5 cm diameter holes around the perimeter. One hole is attached to a darkly shaded target box, which is not visible from the surface of the board. The maze was rotated 90° and washed with 70% EtOH after each trial. Even though the position of the holes changed, the location of the target box location beneath the holds remained constant during the testing period. Air flow from a fan located 0.25 meters from the maze exposed the mouse to an additional mild aversive stimulus. Three visual cues were located 60 cm from the Barnes maze: (1) a white square (30 × 30 cm), (2) 5 red ovals (20 × 15 cm) on a silver rectangle (80 × 40 cm), and (3) 5 vertical white stripes (3 cm) placed equidistant on a brown-colored board (30 × 30 cm). The light level of the room containing the Barnes Maze and elevated plus maze was 295 lux. Day 1 comprised of 2 minutes of habituation training with no target box. For four consecutive days, each mouse had four 3-minute trials of spatial acquisition with 15-minute inter-trial interval. Any Maze software (Stolting) analyzed videos of each trail for total distance traveled, speed, the time elapsed to reach the target box (primary latency) and number of entrances into the non-target holes (primary errors).
Active Place Avoidance
APA was done with slight modifications of the method of Burghardt, et al. (Burghardt et al., 2012). APA was conducted on a 40 cm diameter arena apparatus rotating at 1 rpm with prominent visual landmarks on the walls of the room. Mice were tracked by an infrared Firewire camera 1.2 m above the surface of the area and tracker software (BioSignal Group, Brooklyn, NY). During APA, a computer defined a 60° shock zone area. A shock zone entry of at least 500 msec triggered a foot shock produced by a constant 0.2 mA, 60 Hz current delivered through the grid floor. Mice first received a 10-minute habituation/open field session, with the arena rotating and the shock zone turned off. Total distance traveled, speed and linearity were measured. The mice then had 2 days of APA training consisting with a rotating arena and an active shock zone. Each day consisted of four 10-minute sessions with a 50-minute intertrial interval, during which the mice are returned to their home cages. After testing is complete, mice were returned to their home cages. The total distance traveled, speed, number of shock zone entries, number of shocks delivered, and time to first entrance into the shock zone were analyzed using Track analysis software (Bio-Signal Group Corp, Brooklyn, NY). Linearity of the movement of the mouse was also assessed. Linearity was defined as: (Linear Distance)/(Integrated Distance) with linear distance being the length of a line connecting the location of the mouse at the start and end of the 2-second interval and integrated distance was the sum of the distance moved each 33 ms in a 2-second interval. Linearity determined how straight the animal moved, low linearity was suggestive of less activity.
Elevated Plus Maze
Mice were tested on an apparatus for elevated plus maze that has four 10 x 35 cm arms, two were open and two were closed that extended from a 10 x 10 cm platform. The apparatus was elevated 50 cm above the ground. Mice were placed in the center of the maze and video recorded in a one 10-minute trial in which they freely moved between the open or closed arms. Video records for entries and time spent in the open and closed arms were analyzed by AnyMaze software (Stoelting).
Acoustic Startle Response
Acoustic startle response (ASR) was tested using SR-LAB™ startle response system (San Diego Instruments). Mice were first habituated for 5 minutes to the testing apparatus while exposed to 65 dB background ambient noise. The experimental session began with four 120 dB sound bursts lasting 40 msec. The mice were then exposed to alternating trials of 90 and 105 dB. Intertrial intervals vary randomly between 5 to 13 seconds to prevent habituation. Acoustic startle frequently measures the Vmax of the startle response. Previous studies by others showed acoustic startle response was inhibited in a variety of different TBI models (Sackheim et al., 2017; Teutsch et al., 2018; Bodnar et al., 2019). Therefore, the frequency of trials that had a Vmax of zero was assessed.
Immunofluorescent and Immunohistochemical Staining
Mice were transcardially perfused with paraformaldehyde (4% (w/v)) and 20-μm thick coronal sections of the brain prepared (Leica cryostat, Nussloch, Germany) and mounted on slides. Immunofluorescence staining was done concurrently for all groups overnight at 4°C. Immunofluorescence staining was done overnight at 4°C using antibodies against Arginase-I (1:1000, BD Biosciences Cat# 610708, RRID:AB 398031) followed by a Goat anti-Mouse Secondary Antibody, Alexa Fluor 594 (1:1000, Thermo Fisher Scientific Cat# A-21125, RRID:AB 2535767) or anti-Brain-Derived Neurotrophic Factor (BDNF) (1:1000, Santa Cruz Biotechnology Cat# sc-546, RRID:AB_630940) followed by Goat Anti-Rabbit IgG, Alexa Fluor 430. All slides were counterstained with 4′,6-diamidino-2-phenylindole (Southern Biotech). Mean fluorescence intensity was quantified using ImageJ. Total fluorescence particle counts were normalized to the area of images in square millimeters. For immunohistochemistry, sections are incubated in 1% hydrogen peroxide and then incubated with antibodies against ionized calcium binding adaptor molecule 1 (Iba1, 1:1000, Wako, RRID:AB 2811160) or glial fibrillary acidic protein (GFAP, 1:1000, Abcam, RRID:AB 296804) overnight at 4°C, followed by staining with biotinylated secondary antibody. Sections were then washed, and immunocomplexes stained using the Elite ABC (Vectastain) and diaminobenzidine. Sections were then counterstained with hematoxylin, dehydrated in alcohol and xylene washes, coverslipped, and imaged on a Leica SP8x or a Nikon Eclipse E600 microscope at room temperature. Total numbers of diaminobenzidine stained cells were counted per 40x field of view of relevant brain regions. The dorsal hippocampus (DV, −2.0 to −2.3, ML, 1.8 to 2.1) was analyzed in three coronal sections from each mouse located between 1.5 to 1.8 mm from Bregma (Paxinos and Franklin, 2001). The amygdala was analyzed (DV, − 4.4 to 4.6; ML 2.2 to 2.4) in three coronal sections located between −1.3 to −1.5 mm from Bregma. Only the hemisphere ipsilateral to the impact site or sham impact site was analyzed. All histological measurements were done at 14 post- days post-injury.
Statistical Methods
In Barnes maze, primary latency or primary errors for the 4 daily trials were averaged. The values from the 4 days were analyzed by repeated measure 2-way ANOVA. Shock zone entrances and total shocks in APA was also analyzed by repeated measure 2-way ANOVA. Pairwise comparisons were performed among groups in Barnes maze and APA using Bonferroni’s post-hoc test. All other data were assessed using one-way ANOVA followed by pairwise comparisons using Tukey’s post-hoc test. Data was presented as mean ± SEM and analyzed using SPSS v.23 or Graph Pad Prism 7 software with significance set at 0.05.
Results
Mice were divided into 5 groups and received one of five exposures (sham, CHI, CVS, CHI → CVS and CVS → CHI). All groups then were returned to their cages for seven days to minimize the effects of transient motor deficits after CHI (Grin’kina et al., 2016). All groups received the same behavioral assessments.
Slight differences in head acceleration-deceleration during the CHI procedure is significantly correlated with the interval between the head of mouse receiving an impact and recovery of its righting reflex. Time to regain righting reflex correlates with extent of neuronal loss, demyelination and behavioral recovery (Grin’kina et al., 2016). Sham-Injured mice had a righting reflex of 58 ± 24 seconds (n = 12). The groups receiving CHI had did not differ in the time needed to recover righting reflex and suggests that they all received a mild CHI injury ((in min) CHI, 5.1 ± 1.7; CHI → CVS, 6.7 ± 1.8; CVS → CHI, 8.6 ± 1.9: ANOVA, F4,43 = 0.78, p > 0.4). This time to recover of righting reflex is consistent with a mild CHI injury, which was previously termed CHI-1 (Grin’kina et al., 2016).
Barnes Maze
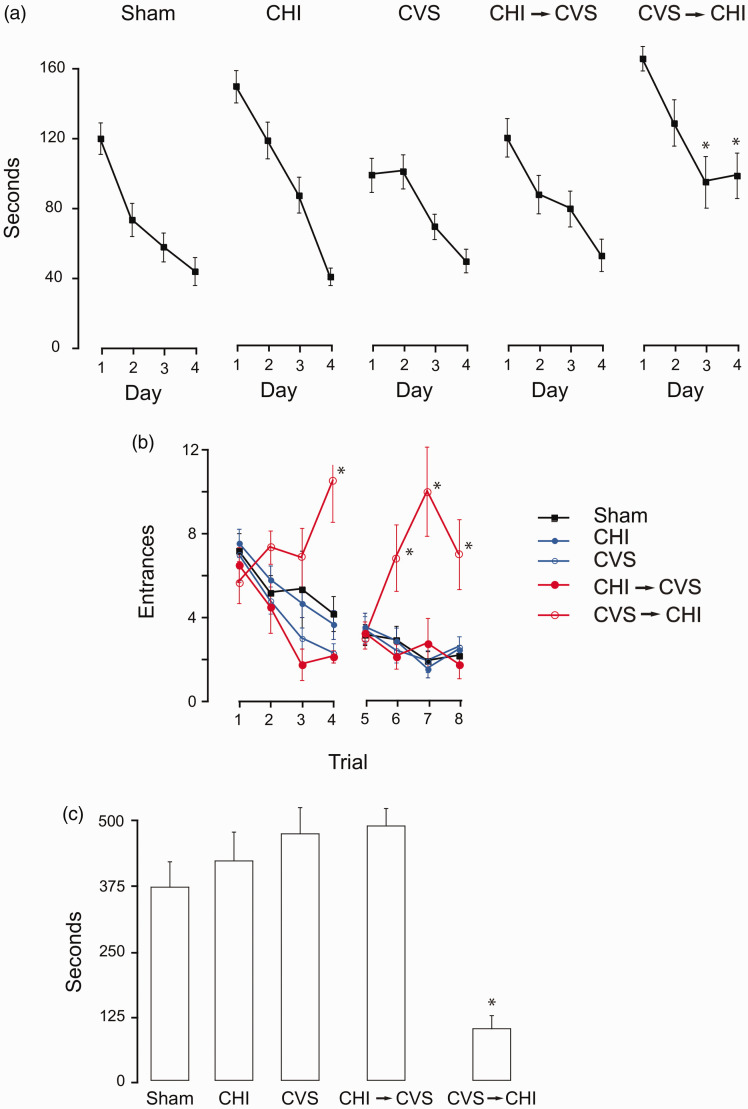
All groups were then tested on Barnes maze. Both the time latency to find the target box and primary errors were significantly affected by the treatment (latency, F4,43 = 30.5, p < 0.005; errors, F4,43 = 15.9, p < 0.005) and day (latency, F3,176 = 19.2, p < 0.001; errors, F3,15 = 27.4, p < 0.001) with no interaction between treatment and day (latency, F6,368 = 380.2, p < 0.05; errors, F6,368 = 27.9, p < 0.02) (Figure 1A, Table 1). On the 3rd and 4th day and final day of testing, the CVS → CHI group had a significantly longer latency than the other groups (p < 0.01) (Figure 1A). On the 4th of Barnes maze testing, the CVS → CHI group also had significantly more errors than the other groups (Table 1). All groups moved a similar distance during testing suggesting that the increased latency and errors of the CVS → CHI group were due to impaired spatial navigation, rather than impaired motor ability (Table 1).
Figure 1.
CVS → CHI Impairs Performance on Barnes Maze and Active Place Avoidance. (a) On the 3rd and 4th day of Barnes testing, the CVS → CHI group had a significantly longer latency to find the escape hole than the other groups (*p < 0.01). (b) On the 4th, 6th, 7th, and 8th trials of active place avoidance testing, the CVS → CHI group CHI had significantly more entrances than the other groups (*p < 0.05). (c) The CVS → CHI group had a significantly shorter time to 1st entrance into the shock zone than the other groups (*p < 0.005). These data suggest that CVS → CHI produces behavioral deficits not seen in the other groups.
Table 1.
Parameters of Mouse Behavior.
| Sham-CHI | CHI | CVS | CHI→ CVS | CVS → CHI | |
|---|---|---|---|---|---|
| Barnes Maze | |||||
| Distance | 45.0 ± 2.1 | 49.5 ± 2.9 | 45.3 ± 2.2 | 46.3 ± 2.6 | 44.14 ± 2.2 |
| Primary errors on Day 4 | 2.9 ± 0.5 | 3.1 ± 0.6 | 4.4 ± 0.9 | 2.5 ± 0.8 | 7.3 ± 1.2* |
| Active Place Avoidance | |||||
| Distance Traveled on Habituation Trial | 45.0 ± 2.1 | 44.0 ± 5.5 | 45.3 ± 2.2 | 46.3 ± 2.6 | 43.7 ± 2.3 |
| Distance Traveled all other Trials | 22.8 ± 1.1 | 25.4 ± 1.3 | 23.6 ± 1.0 | 24.3 ± 1.8 | 27.6 ± 1.4 |
| Speed | 4.2 ± 0.2 | 4.6 ± 0.5 | 3.9 ± 0.1 | 3.9 ± 0.2 | 4.3 ± 0.3 |
| Linearity | 0.50 ± 0.03 | 0.58 ± 0.02 | 0.55 ± 0.02 | 0.48 ± 0.05 | 0.53 ± 0.02 |
| Average Total Shocks | 37.0 ± 3.9 | 34.7 ± 4.0 | 36.1 ± 5.7 | 31.3 ± 4.0 | 67.3 ± 8.9** |
| Average Total Entrances | 32.0 ± 3.4 | 32.1 ± 3.3 | 27.4 ± 3.0 | 24.8 ± 3.9 | 57.2 ± 6.4** |
| Average Shocks per Entrance | 1.23 ± 0.03 | 1.13 ± 0.04 | 1.25 ± 0.07 | 1.22 ± 0.08 | 1.17 ± 0.06 |
| Elevated Plus Maze | |||||
| Distance Traveled | 13.5 ± 2.0 | 16.4 ± 2.2 | 15.0 ± 2.8 | 14.8 ± 2.7 | 24.7 ± 2.2* |
On Barnes maze, distance traveled (in meters) had no treatment effect (F4,43 = 0.7, p > 0.5). On primary errors, there a significant effect of treatment and day with a significant interaction of treatment and day (treatment, F4,43 = 15.9, p < 0.005; day, F3,15 = 27.4, p < 0.001; interaction, errors, F6,368 = 27.9, p < 0.02) On day 4, the CVS → CHI group had significantly more errors than the other groups (*p < 0.05). On active place avoidance, treatment had no significant effect on distance traveled during habituation or training (habituation, F4,43 = 0.09, p < 1.0; training, F4,43 = 1.80, p < 0.9) nor did treatment have a significant effect on speed or linearity (speed, F4,43 = 1.26, p < 0.3; linearity, F4,43 = 2.10, p < 0.1). Average total shocks and shock zone entrances had a significant effect of treatment (shocks, F4,43 = 6.4, p < 0.01 entrances, F4,43 = 8.5, p < 0.001) with the CVS → CHI group having significantly more entrances and shocks than the other groups (**p > 0.005). Despite the CVS → CHI group receiving more shocks and more entrances, treatment showed no significant effect on shocks per entrance (F4,43 = 0.78, p < 0.5). On distance traveled on elevated plus maze, there was a significant effect of treatment (F4,43 = 2.7, p < 0.05) with the CVS → CHI traveling significantly more than the other groups (*p < 0.05).
Active Place Avoidance
At 10 days after the end of sham-treatment or treatments, all groups received APA training. All groups traveled the same distance during habituation (Table 1). In two days of APA testing, the number of shock zone entrances had significant effects of stress/injury, i.e. CVS and CHI, trial with a significant interaction of stress/injury and trial compared to other injury and sham groups (treatment, F 4,43 = 20.0, p < 0.005; trial, F 7,43 = 6.5, p < 0.001, interaction, F 28,352 = 22.1, p < 0.0005) (Figure 1B). On trial 4, the last trial of the 1st day of APA testing, the CVS → CHI group had significantly more shock zone entrances that the other groups (p < 0.05). Despite this difference on the final trial of the 1st day of testing, all groups had similar number of entries on trial 5, the 1st trial of the 2nd day of APA testing (p > 0.5). The similar number of entrances on trial 5 suggests similar retention of the shock zone location of all groups between the 1st and 2nd day of testing. The CVS → CHI group, however, had significantly more entrances than all other groups on the subsequent 6th, 7th and 8th trials (p < 0.05). There was no significant difference among all groups on various parameters of motor function. CVS → CHI group, however, received significantly more shocks (Table 1). The shocks per entrance of the CVS → CHI group, however, were similar to the other groups suggesting that the CVS → CHI group had a similar ability to experience shock and leave the shock zone (Table 1). These data suggest that the impairment of the CVS → CHI group was not due to deficits in sensory or motor function. Time to 1st entrance measures whether a mouse recalls the location of the shock zone from previous trials prior to being shocked (Figure 1C). Time to 1st entrance had a significant group effect (F4,43 = 7.90, p < 0.0001). The time to 1st entrance of the CVS → CHI group was significantly shorter than the other groups (p < 0.005).
Elevated Plus Maze
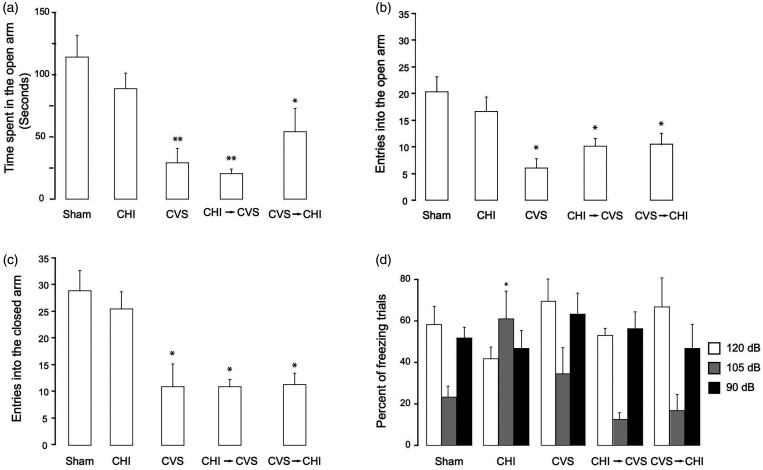
After APA, basal anxiety was examined using elevated plus maze (Figure 2A to C). Time or number of entrances in the open or closed arms had significant stress/injury effects (time, F4,43 = 8.7, p < 0.001; open arm entrances, F4,43 = 6.1, p < 0.001; closed arm entrances, F4,43 = 7.0, p < 0.001). The CVS, CHI→ CVS, and CVS → CHI groups spent significantly less time in the open arms than the sham-injured group (Figure 2A; CVS, CHI→ CVS p < 0.001; CVS → CHI, p < 0.05). The CVS, CHI→ CVS, and CVS → CHI also significantly differed from the sham group with fewer entries into the open or closed arms (Figure 2B and C, p < 0.05). the CVS → CHI group moved significantly more than the other groups despite having a similar number of entrances (Table 1). These data suggest considerably more basal anxiety in the CVS, CHI→ CVS, and CVS → CHI groups than the sham-treated or CHI groups.
Figure 2.
CHI → CVS and CVS → CHI Produces Impairments on Elevated Plus Maze and CHI Impairs Acoustic Startle Response. A: The CVS, CHI → CVS and CVS → CHI groups had spent significantly more time in the open arm compared to the sham-treated group (CVS or CHI → CVS, **p < 0.001; CVS → CHI, *p < 0.05). The CVS, CHI → CVS and CVS → CHI groups also had significantly fewer entrances into open (Panel B) or closed (Panel C) arms (*p < 0.05). D: The CHI group had significantly more trials with behavioral freezing at 105 dB than the other groups (*p < 0.05).
Acoustic Startle Response
A significant effect of treatment was observed during the 105 dB bursts on the acoustic startle response (120 dB, F4,43 = 1.5, p > 0.2; 105 dB, F4,43 = 4.2 p < 0.01, 90 dB, F4,43 = 0.6, p > 0.5) (Figure 2D). The CHI group experienced significantly more freezing sessions than the remaining groups (p < 0.05).
Histological Analysis
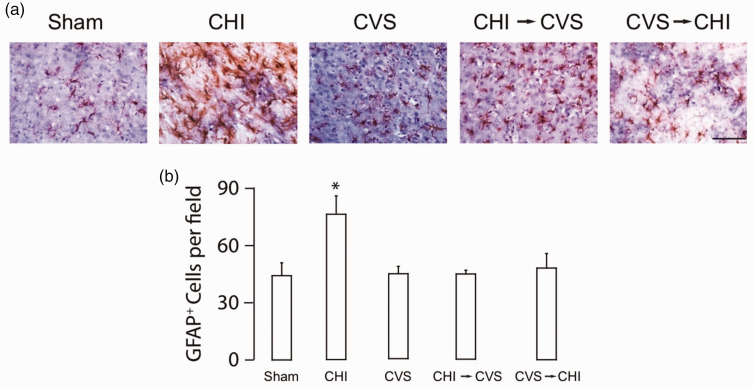
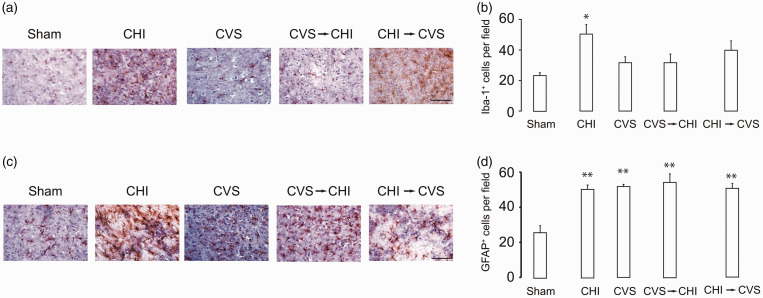
After the completion of acoustic startle testing, histology was done on the same mice to examine neuroinflammation. Astrocyte activation was assessed using GFAP, and Iba-1 levels were used to assess microglial/macrophage (MP/MG) activation (Ito et al., 1998; Rupprecht et al., 2010; Liddelow and Barres, 2017). Inflammation was examined in the hippocampus since hippocampal function was involved in the Barnes maze and APA tests (Sunyer et al., 2007; Lesburguères et al., 2016). GFAP expression was significantly increased in the hippocampal CA3 region (F4,10 = 4.36; p < 0.05) with more GFAP+ cells in the CHI group than the other groups (Figure 3A and B, Supplemental Figure 1). The number of GFAP+ cells in the CVS, CHI → CVS, CVS → CHI groups did not significantly differ from those in the sham treatment group. No significant treatment effect was seen in the dentate gyrus or the CA1 regions of the hippocampus (Dentate, F4,10 = 1.10; p > 0.4; CA1, F4,10 = 0.65, p > 0.3).
Figure 3.
CHI Activates Astrocytes in CA3. (a) Representative images of GFAP immunoreactivity in the CA3 region of the hippocampus. (b) The number of GFAP+ CA3 cells was significantly increased in the CHI group as compared to Sham-group (*p < 0.05). Scale bar, 100 μm.
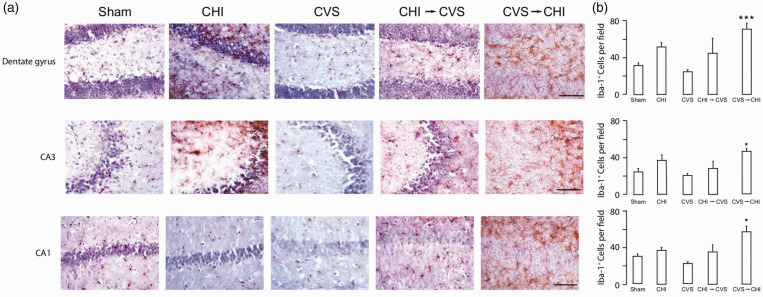
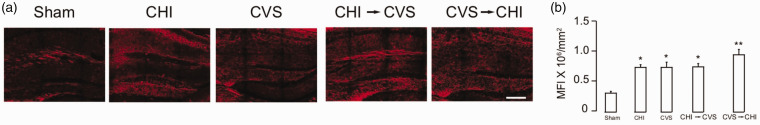
Expression of the Iba-1 was also analyzed. There was a significant increase of Iba-1 immunoreactivity in the dentate gyrus, CA1 and CA3 regions of the hippocampus (Dentate, F4,10 = 17.04; p < 0.005; CA1, F4,10 = 7.13; p < 0.01; CA3, F4,10 = 4.84; p < 0.05) (Figure 4A and B). In all regions, the number of Iba-1+ cells were significantly increased in the CVS → CHI group (Figure 4B).
Figure 4.
CVS → CHI Increases Iba-1+ Cell Number in Multiple Hippocampal Regions. (a) Representative images of Iba-1 immunoreactivity in the dentate gyrus (top), CA3 (middle) or CA1 (bottom). Scale bar 100 μm. (b) Summary of Iba-1+ cell number. In all three hippocampal regions, the CVS → CHI group had significantly more Iba-1+ cells than the other groups (*p < 0.05). Scale bar, 100 μm.
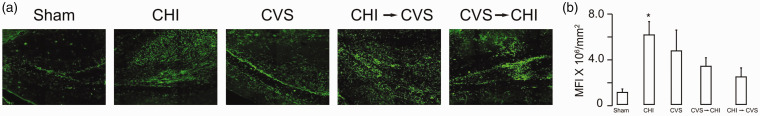
The activated MP/MG, evident by increased Iba-1 expression, may lead to increase in arginase-1 expression via a alteration to an anti-inflammatory subtype (Cherry et al., 2014; Lam et al., 2017). Arginase-1 immunofluorescence showed a significant effect of treatment (Figure 5A and B) (F4,11 = 13.28; p < 0.005). Arginase-1 immunofluorescence also significantly increased in all treatment groups over sham (p < 0.01). The increase in arginase-1 immunofluorescence was similar in all groups exposed to stress/injury (Figure 5B). Thus, it was likely that activated MP/MG increased arginase-1 expression. Alternatively, some arginase-1 expressing cells could be infiltrating granulocytes, or lymphocytes (Rodriguez et al., 2017).
Figure 5.
Increased Hippocampal Arginase-1 Expression Following CHI, CVS, CHI → CVS, CVS → CHI. (a) Representative images of hippocampal arginase-1 immunofluorescence. (b) Summary of the changes in arginase-1 expression. Arginase-1 expression in the CHI, CVS, CHI → CVS, and CVS → CHI groups were significantly greater than the sham-treatment group (*p < 0.005, **p < 0.0005). Scale Bar, 100 μm.
Neuroinflammation was also assessed in the amygdala. Iba-1 trended strongly for an effect of treatment (F4,10 = 4.06; p = 0.07) with significantly more Iba-1+ cells in the CHI-1 group than the sham-treated group (p < 0.05) (Figure 6A and B). GFAP was significantly increased by the treatments (F4,10 = 11.41; p < 0.005), and all treatment groups significantly differed from the sham-treatment group (p < 0.005) (Figure 6C and D).
Figure 6.
Neuroinflammation in the Amygdala. (a) Representative images of Iba-1+ cells. (b) Summary of differences in Iba-1+ cell number. There were significantly more Iba-1+ cells in the CHI group than in the sham-treated group (*p < 0.05). (c) Representative images of GFAP+ cells in the amygdala. (d) There were significantly more GFAP+ cells in all experimental groups as compared to the sham-treated group (stats). Scale bar, 100 μm.
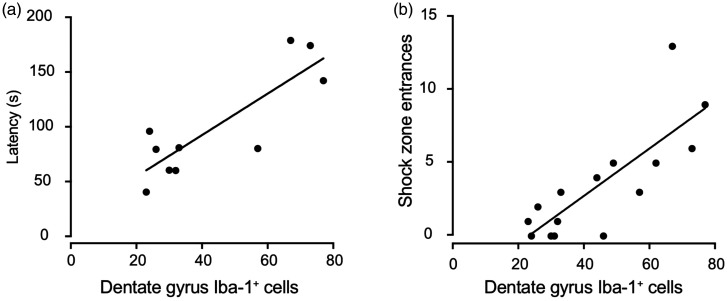
Chronic neuroinflammation has been associated with impaired behavioral outcome. We therefore examined whether inflammation in the hippocampus at 2 weeks post-injury correlated with behavioral impairments on hippocampus-dependent tasks. Performance on Barnes maze significantly correlated with the number of activated MG/MP in the dentate gyrus, CA1 and CA3 (Figure 7A) (Dentate gyrus, r2 = 0.84, p < 0.005; CA1, r2 = 0.78, p < 0.01; CA3, r2 = 0.72, p < 0.05). The number of activated MG/MP cells, measured by Iba-1+ cell number, in the dentate, CA1 and CA3 regions also correlated strongly with performance on the APA task (Figure 7B) (Dentate Gyrus, r2 = 0.80, p < 0.005; CA1, r2 =0.77, P < 0.001; CA3, r2 = 0.71, p < 0.005). These data suggest that long-lasting hippocampal neuroinflammation contributed to impairments in hippocampal-dependent behavioral tasks.
Figure 7.
Inflammation in the Dentate Gyrus Correlates With Impaired Performance on Hippocampal-Dependent Tasks. The number of Iba-1+ cells in the dentate gyrus significantly correlated with average latency of the 4 trials on the first day of Barnes maze training (Panel A) (r2 = 0.77, p < 0.005) and with the number of entrances on trial 6 on active place avoidance (Panel B) (r2 = 0.64, p < 0.005).
Increased expression of brain derived neurotrophic factor (BDNF) increases neuroprotection, promotes connectivity and induces repair (Kaplan et al., 2010). BDNF expression was therefore examined in the hippocampus (Figure 8). BDNF expression showed a significant treatment effect (F4,16 = 4.90; p < 0.01). BDNF expression was significantly increased in the CHI group (p < 0.05), but not in the other tested groups. These data suggest that the combining CHI with CVS did not increase in BDNF expression.
Figure 8.
CHI Increases Hippocampal BDNF Expression. A: Representative images of hippocampal BDNF immunofluorescence in the CA3 regions. B: Summary of differences in BDNF expression. The CHI group had significantly more BDNF expression than the sham-treated group (*p < 0.01).
Discussion
The combination of CHI model of TBI with the CVS model of PTSD was used to examine the behavioral and neuroinflammatory consequences of co-morbid TBI/PTSD. Using multiple outcome measures, combining CHI and CVS produced greater behavioral impairments and neuroinflammation as compared to CVS or CHI alone (Figures 1, 2a, and 4, Table 1). In addition, insult order is important since the CVS → CHI group had greater behavioral deficits and neuroinflammation than the CHI → CVS group (Figures 1, 2a, and 4, Table 1).
The CHI and CHI → CVS groups had a mild CHI as assessed by recovery of righting reflex as well as their ability to acquire Barnes maze and an APA task (Figure 1A and B) (Grin’kina et al., 2016). The CVS → CHI group needed a similar time to regain righting reflex as the CHI and CHI → CVS groups yet was impaired on both Barnes maze and APA (Figure 1A and B). These data suggest that behavioral deficits of the CVS → CHI group did not arise from acute differences in CHI injury. The CVS → CHI group had deficits in Barnes maze and APA that differed from acquisition of these tasks by the groups CHI or CVS (Figures 1 and 2A and B; Table 1). Unexpectedly, the CHI → CVS group lacked deficits on Barnes maze or APA. These data suggest a synergistic interaction between CVS and CHI that depended upon insult order since the CVS → CHI group had behavioral deficits lacking in the CHI → CVS group.
All groups, including the CVS → CHI group, had similar performance on the 1st day of APA and the 1st trial (trial 5) of the 2nd day of APA (Figure 1B). The performance of the CVS → CHI group on trial 5 also suggest a 24 hours retention of the shock zone location between days 1 and 2 of APA testing. The CVS → CHI group, however, had significantly more shock zone entrances than the other groups on trials 6-8 suggesting that the impaired performance of the CVS → CHI group on APA. APA produces stress as measured by elevated corticosteroid levels (Karabeg et al., 2013; Lesburguères et al., 2016). This suggests that the deficits of the CVS → CHI group were not deficits in cognition or memory (Figure 1B). This stress may have been induced by the increased number of shocks received by the CVS → CHI group since basal anxiety of the CVS → CHI group on the elevated plus maze was similar to the CVS and CHI → CVS groups (Figure 2A to C, Table 1). The behavioral deficits of the CVS → CHI on later trials of APA may have resulted from an increased stress response rather than a memory deficit (Harrison et al., 2009; McKim et al., 2016).
Acquisition of APA requires two functional hippocampi and the commissural axonal fibers connecting them (Cimadevilla et al., 2001; Grin’kina, 2013). This suggests that the CHI and CHI → CVS groups retained bilateral hippocampal function (Figure 1B). The CHI and CHI → CVS groups also acquired Barnes maze, which is consistent with prior studies showing that only one functioning hippocampus is required to learn the task (Figure 1A) (Skelton, 1998; Shinohara et al., 2012). In contrast, CVS → CHI group was impaired on both Barnes maze and APA (Figure 1). These data suggest that the CVS → CHI group may have loss of hippocampal function not seen in the CHI or CHI → CVS groups, yet future histological or electrophysiological studies are needed to support this conclusion.
CHI inhibited the acoustic startle response with greater percentage of the mice freezing during trials of 110 dB (Figure 2D). Three disparate experimental models of TBI; controlled cortical impact model, fluid percussion and weight drop showed a similar inhibition of acoustic startle response (Wolf et al., 2017; Teutsch et al., 2018; Bodnar et al., 2019). The underlying cause is unknown why head injury inhibits the acoustic startle response in multiple TBI models and is deserving of further study.
Long-lasting neuroinflammation is a prominent feature of TBI and animal models of TBI (Faden et al., 2016; Morganti-Kossmann et al., 2019). PTSD may also produce a chronic pro-inflammatory state as seen with increased plasma cytokine levels (Daskalakis et al., 2016; Mellon et al., 2018). Increased inflammation has been described in animal PTSD models as well. The CVS → CHI group had greater hippocampal MP/MG activation APA place avoidance positively correlated with increased Iba-1 expression in the dentate gyrus (Figure 7A and B). These data suggest that increased neuroinflammation in the CVS → CHI group may contribute to impairments on APA and Barnes maze. The finding that the CHI → CVS group did not have deficits on Barnes Maze or APA highlights the importance of the order stress/injury exposure. This finding supports a synergistic and yet asymmetrical relationship between CHI and CVS. TBI has been reported to impair extinction of contextual fear memory via preferentially impacting the infralimbic area (IL) of the medial pre-frontal (mPFC) cortical area (Zhao et al., 2018). Hence exposure to CVS prior the CHI injury, would potentially lead to a more hyper-arousable state, as evident by the CVS → CHI group performance on APA and ASR. The resultant hyper-arousable state would result in further activation of the HPA axis, and a subsequent pro-inflammatory state (Lamers et al., 2013). The state of MG/MP activation is not known at the time of CHI injury in mice receiving CVS → CHI, since the immunohistological studies were not done at this time point. The possibility remains that CHI may occur at an increased pro-inflammatory state in CVS → CHI mice. Arginase-1 recognizes some, but not all, forms of anti-inflammatory microglia. Additional studies are needed to fully characterize the gamut of microglial phenotypes produces by CHI, CVS and its combination (Wolf et al., 2017). Increased GFAP expression was also observed in the amygdala of the CVS, CHI and CHI → CVS and CVS → CHI groups (Figure 6C and D). GFAP expression was also elevated in the basal lateral amygdala after midline fluid percussion injury in rats (Hoffman et al., 2017).
The finding of interconnected pathophysiology of TBI and PTSD has potential clinical implications. This study suggests that individuals with PTSD may be more vulnerable to neurocognitive sequelae of TBI. The importance of the order of TBI and PTSD experiences is unexpected. Moreover, the order of stress and injury, as well as symptoms attributable to TBI or PTSD individually, are challenging to determine outside the laboratory. This is in part due the fact that the clinical studies examining the relationship of TBI and PTSD, heavily rely on individual self-reports subject to recall bias, as opposed to more objective clinical assessments at different time points (Hendrickson et al., 2018) . Others have suggested an interaction of pre-existent psychiatric illnesses, such as PTSD with TBI.
According to a study involving 102 adults, 74.5% of participants with a pre-injury psychiatric history experienced a post-injury psychiatric disorder, while 45.8% of the subjects without a pre-injury psychiatric history developed a novel psychiatric disorder (Gould et al., 2011). An implication of this study would be to exercise more vigilance in treatment of individuals with PTSD with subsequent exposure to TBI.
There are important caveats to this study. Comorbid TBI/PTSD likely produces chronic effects, and additional studies are needed to see if behavioral deficits in this study are similarly long-lasting. The interval between the two insults was only one day and any of the reported effects may change as the time intervals between the two insults increases. Finally, this study only studied male mice. There is little evidence supporting a role of sex in the outcomes of TBI, in terms of chronic neurocognitive and neuropsychiatric deficits, as well as recovery outcomes (Breslau et al., 1997; Ratcliff et al., 2007; Bazarian et al., 2010; Cancelliere et al., 2016). There is, however, sexual dimorphisms of the neuroinflammatory response to experimental TBI (Villapol et al., 2017; Doran et al., 2019). In addition, women have a higher incidence of PTSD than men, and mouse models of PTSD show an increased vulnerability of females (Breslau et al., 1997; Whitaker et al., 2014). The comparison of male and female mice should be included in future studies of comorbid TBI/PTSD.
This study reports a greater degree of deficit in the CVS → CHI group, an animal model for PTSD proceeding TBI, based on increased neurocognitive deficits as well as heightened neuroinflammatory response. This result has important clinical correlates, as it highlights the importance of the order of TBI vs PTSD, and their synergistic as well as asymmetrical relationship. This in turn implies a great degree of care of individuals with premorbid PTSD diagnosis, in settings of high risk for brain injury including combat settings and sports arenas.
Summary
Increased behavioral deficits and inflammation was seen in a novel model of co-morbid TBI/PTSD. Insult order influenced the deficits severity and the amount of neuroinflammation.
Supplemental Material
Supplemental material, sj-jpg-1-asn-10.1177_1759091420979567 for Increased Behavioral Deficits and Inflammation in a Mouse Model of Co-Morbid Traumatic Brain Injury and Post-Traumatic Stress Disorder by Arman Fesharaki-Zadeh, Jeremy T. Miyauchi, Karrah St. Laurent-Arriot, Stella E. Tsirka and Peter J. Bergold in ASN Neuro
Supplemental material, sj-pdf-2-asn-10.1177_1759091420979567 for Increased Behavioral Deficits and Inflammation in a Mouse Model of Co-Morbid Traumatic Brain Injury and Post-Traumatic Stress Disorder by Arman Fesharaki-Zadeh, Jeremy T. Miyauchi, Karrah St. Laurent-Arriot, Stella E. Tsirka and Peter J. Bergold in ASN Neuro
Footnotes
Author Contributions: A. F.-Z., S. E. T., and P. J. B. designed the experiments; A. F.-Z., J. T. M., and K. St. L.-A. performed the experiments. A. F.-Z., S. E. T., and P. J. B. prepared the manuscript.
Declaration of Conflicting Interests: The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant to P. J. B. and S. E. T. from the State University of New York Brain Network of Excellence and National Institutes of Health F30CA196110 to J. T. M.
ORCID iD: Arman Fesharaki-Zadeh https://orcid.org/0000-0002-8294-7163
Stella E. Tsirka https://orcid.org/0000-0003-0022-1770
Supplemental material: Supplemental material for this article is available online.
References
- Abdel Baki S. G., Kao H.-Y., Kelemen E., Fenton A. A., Bergold P. J. (2009). A hierarchy of neurobehavioral tasks discriminates between mild and moderate brain injury in rats. Brain Research, 1280, 98–106. [DOI] [PubMed] [Google Scholar]
- Amos T., Stein D. J., Ipser J. C. (2014). Pharmacological interventions for preventing post‐traumatic stress disorder (PTSD). Cochrane Database of Systematic Reviews, 7, CD006239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aungst S. L., K S., Thompson S. M., Stoica B. M., Faden A. I. (2014). Repeated mild traumatic brain injury causes chronic neuroinflammation, changes in hippocampal synaptic plasticity, and associated cognitive deficits. Journal of Cerebral Blood Flow and Metabolism: Official Journal of the International Society of Cerebral Blood Flow and Metabolism, 34(7), 1223–1232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barnes C. A. (1979). Memory deficits associated with senescence: A neurophysiological and behavioral study in the rat. Journal of Comparative and Physiological Psychology, 93(1), 74–104. [DOI] [PubMed] [Google Scholar]
- Bazarian J. J., Blyth B., Mookerjee S., He H., McDermott M. P. (2010). Sex differences in outcome after mild traumatic brain injury. Journal of Neurotrauma, 27(3), 527–539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bergold P. J. (2016). Treatment of traumatic brain injury with anti-inflammatory drugs. Experimental Neurology, 275 (Pt 3), 367–380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bodnar C. N., Roberts K. N., Higgins E. K., Bachstetter A. D. (2019). A systematic review of closed head injury models of mild traumatic brain injury in mice and rats. Journal of Neurotrauma, 36(11), 1683–1706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Breslau N., Davis G. C., Andreski P., Peterson E. L., Schultz L. R. (1997). Sex differences in posttraumatic stress disorder. Archives of General Psychiatry, 54(11), 1044–1048. [DOI] [PubMed] [Google Scholar]
- Bryant R. (2011). Post-traumatic stress disorder vs traumatic brain injury. Dialogues in Clinical Neuroscience, 13(3), 251–262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burghardt N. S., Park E. H., Hen R., Fenton A. A. (2012). Adult-Born hippocampal neurons promote cognitive flexibility in mice. Hippocampus, 22(9), 1795–1808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cancelliere C., Donovan J., Cassidy J. D. (2016). Is sex an indicator of prognosis after mild traumatic brain injury: A systematic analysis of the findings of the world health organization collaborating centre task force on mild traumatic brain injury and the international collaboration on mild traumatic brain injury prognosis. Archives of Physical Medicine and Rehabilitation, 97(2 Suppl), S5–S18. [DOI] [PubMed] [Google Scholar]
- Cernak I., Noble-Haeusslein L. J. (2010). Traumatic brain injury: An overview of pathobiology with emphasis on military population. Journal of Cerebral Blood Flow and Metabolism: Official Journal of the International Society of Cerebral Blood Flow and Metabolism, 30(2), 255–266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cherry J. D., Olschowka J. A., O'Banion M. K. (2014). Neuroinflammation and M2 microglia: The good, the bad, and the inflamed. Journal of Neuroinflammation, 11, 98–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cimadevilla J. M., Wesierska M., Fenton A. A., Bures J. (2001). Inactivating one hippocampus impairs avoidance of a stable room-defined place during dissociation of arena cues from room cues by rotation of the arena. Proceedings of the National Academy of Sciences of the United States of America, 98(6), 3531–3536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Corps K. N., Roth T. L., McGavern D. B. (2015). Inflammation and neuroprotection in traumatic brain injury. JAMA Neurology, 72(3), 355–362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Daskalakis N. P., Cohen H., Nievergelt C. M., Baker D. G., Buxbaum J. D., Russo S. J., Yehuda R. (2016). New translational perspectives for blood-based biomarkers of PTSD: From glucocorticoid to immune mediators of stress susceptibility. Experimental Neurology, 284(Pt B), 133–140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deslauriers J., Toth M., Der-Avakian A., Risbrough V. B. (2018). Current status of animal models of posttraumatic stress disorder: Behavioral and biological phenotypes, and future challenges in improving translation. Biological Psychiatry, 83(10), 895–907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doran S. J., Ritzel R. M., Glaser E. P., Henry R. J., Faden A. I., Loane D. J. (2019). Sex differences in acute neuroinflammation after experimental traumatic brain injury are mediated by infiltrating myeloid cells. Journal of Neurotrauma, 36(7), 1040–1053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Faden A. I., Wu J., Stoica B. A., Loane D. J. (2016). Progressive inflammation-mediated neurodegeneration after traumatic brain or spinal cord injury. British Journal of Pharmacology, 173(4), 681–691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goodwill H. L., Manzano-Nieves G., Gallo M., Lee H.-I., Oyerinde E., Serre T., Bath K. G. (2019). Early life stress leads to sex differences in development of depressive-like outcomes in a mouse model. Neuropsychopharmacology: Official Publication of the American College of Neuropsychopharmacology, 44(4), 711–720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goswami S., R S. O., Cascardi M., Pare D. (2013). Animal models of post-traumatic stress disorder: Face validity. Frontiers in Neuroscience, 7(89). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gould K. R., Ponsford J. L., Johnston L., Schönberger M. (2011). The nature, frequency and course of psychiatric disorder in the first year after traumatic brain injury: A prospective study. Psychological Medicine, 41(10), 2099–2109. [DOI] [PubMed] [Google Scholar]
- Grillon C., Robinson O. J., O'Connell K., Davis A., Alvarez G., Pine D. S., Ernst M. (2017). Clinical anxiety promotes excessive response inhibition. Psychological Medicine, 47(3), 484–494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grin’kina N. M., Li Y., Haber M., Sangobowale M., Nikulina E., Le’Pre C., El Sehamy A. M., Dugue R., Ho J. S., Bergold P. J. (2016). Righting reflex predicts long-term histological and behavioral outcomes in a closed head model of traumatic brain injury. PLoS One, 11(9), e0161053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grin'kina N. M., Abdel-Baki S. G., Bergold P. J. (2013). Reversible behavioral deficits in rats during a cycle of demyelination-remyelination of the fimbria. PLoS One, 8(1), e53775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harrison F. E., Hosseini A. H., McDonald M. P. (2009). Endogenous anxiety and stress response in water maze and barnes maze spatial memory tasks. Behavioural Brain Research, 198(1), 247–251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hendrickson R. C., Schindler A. G., Pagulayan K. F. (2018). Untangling PTSD and TBI: Challenges and strategies in clinical care and research. Current Neurology and Neuroscience Reports, 18(12), 106. [DOI] [PubMed] [Google Scholar]
- Hoffman A. N., Paode P. R., May H. G., Ortiz J. B., Kemmou S., Lifshitz J., Conrad C. D., Currier Thomas T. (2017). Early and persistent dendritic hypertrophy in the basolateral amygdala following experimental diffuse traumatic brain injury. Journal of Neurotrauma, 34(1), 213–219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ito D., Imai Y., Ohsawa K., Nakajima K., Fukuuchi Y., Kohsaka S. (1998). Microglia-specific localisation of a novel calcium binding protein, Iba1. Brain Research. Molecular Brain Research, 57(1), 1–9. [DOI] [PubMed] [Google Scholar]
- Jones K. A., Thomsen C. (2013). The role of the innate immune system in psychiatric disorders. Molecular and Cellular Neurosciences, 53, 52–62. [DOI] [PubMed] [Google Scholar]
- Kaplan G. B., Leite-Morris K. A., Wang L., Rumbika K. K., Heinrichs S. C., Zeng X., Wu L., Arena D. T., Teng Y. D. (2018). Pathophysiological bases of comorbidity: Traumatic brain injury and post-traumatic stress disorder. Journal of Neurotrauma, 35(2), 210–225. [DOI] [PubMed] [Google Scholar]
- Kaplan G. B., Vasterling J. J., Vedak P. C. (2010). Brain-derived neurotrophic factor in traumatic brain injury, post-traumatic stress disorder, and their comorbid conditions: Role in pathogenesis and treatment. Behavioural Pharmacology, 21(5–6), 427–437. [DOI] [PubMed] [Google Scholar]
- Karabeg M. M., Grauthoff S., Kollert S. Y., Weidner M., Heiming R. S., Jansen F., Popp S., Kaiser S., Lesch K.-P., Sachser N., Schmitt A. G., Lewejohann L. (2013). 5-HTT deficiency affects neuroplasticity and increases stress sensitivity resulting in altered spatial learning performance in the Morris water maze but not in the barnes maze. PLoS One, 8(10), e78238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khoury N. M., Marvar P. J., Gillespie C. F., Wingo A., Schwartz A., Bradley B., Kramer M., Ressler K. J. (2012). The renin-angiotensin pathway in posttraumatic stress disorder: Angiotensin-converting enzyme inhibitors and angiotensin receptor blockers are associated with fewer traumatic stress symptoms. The Journal of Clinical Psychiatry, 73(6), 849–855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim D. J., St Louis N., Molaro R. A., Hudson G. T., Chorley R. C., Anderson B. J. (2017). Repeated unpredictable threats without harm impair spatial working memory in the barnes maze. Neurobiology of Learning and Memory, 137, 92–100. [DOI] [PubMed] [Google Scholar]
- Lam D., Lively S., Schlichter L. C. (2017). Responses of rat and mouse primary microglia to pro-and anti-inflammatory stimuli: Molecular profiles, K+ channels and migration. Journal of Neuroinflammation, 14(1), 166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lamers F., Vogelzangs N., Merikangas K. R., de Jonge P., Beekman A. T. F., Penninx B. W. J. H. (2013). Evidence for a differential role of HPA-axis function, inflammation and metabolic syndrome in melancholic versus atypical depression. Molecular Psychiatry, 18(6), 692–699. [DOI] [PubMed] [Google Scholar]
- Lesburguères E., Sparks F. T., O'Reilly K. C., Fenton A. A. (2016). Active place avoidance is no more stressful than unreinforced exploration of a familiar environment. Hippocampus, 26(12), 1481–1485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lezak K. R., Missig G., Carlezon W. A., Jr, (2017). Behavioral methods to study anxiety in rodents. Dialogues in Clinical Neuroscience, 19(2), 181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liddelow S. A., Barres B. A. (2017). Reactive astrocytes: Production, function, and therapeutic potential. Immunity, 46(6), 957–967. [DOI] [PubMed] [Google Scholar]
- Lozano D., Gonzales-Portillo G. S., Acosta S., de la Pena I., Tajiri N., Kaneko Y., Borlongan C. V. (2015). Neuroinflammatory responses to traumatic brain injury: Etiology, clinical consequences, and therapeutic opportunities. Neuropsychiatric Disease and Treatment, 11, 97–106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McKim D. B., Niraula A., Tarr A. J., Wohleb E. S., Sheridan J. F., Godbout J. P. (2016). Neuroinflammatory dynamics underlie memory impairments after repeated social defeat. The Journal of Neuroscience : The Official Journal of the Society for Neuroscience, 36(9), 2590–2604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mellon S. H., Gautam A., Hammamieh R., Jett M., Wolkowitz O. M. (2018). Metabolism, metabolomics, and inflammation in posttraumatic stress disorder. Biological Psychiatry, 83(10), 866–875. [DOI] [PubMed] [Google Scholar]
- Mendoza C., Barreto G. E., Ávila-Rodriguez M., Echeverria V. (2016). Role of neuroinflammation and sex hormones in war-related PTSD. Molecular and Cellular Endocrinology, 434, 266–277. [DOI] [PubMed] [Google Scholar]
- Mikolić A., Polinder S., Retel Helmrich I. R. A., Haagsma J. A., Cnossen M. C. (2019). Treatment for posttraumatic stress disorder in patients with a history of traumatic brain injury: A systematic review. Clinical Psychology Review, 73, 101776. [DOI] [PubMed] [Google Scholar]
- Mohamadpour M., Whitney K., Bergold P. J. (2019). The importance of therapeutic time window in the treatment of traumatic brain injury. Frontiers in Neuroscience, 13, 7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morganti-Kossmann M. C., Semple B. D., Hellewell S. C., Bye N., Ziebell J. M. (2019). The complexity of neuroinflammation consequent to traumatic brain injury: From research evidence to potential treatments. Acta Neuropathologica, 137(5), 731–755. [DOI] [PubMed] [Google Scholar]
- Motzkin J. C., Koenigs M. R. Post-traumatic stress disorder and traumatic brain injury, in Handbook of clinical neurology. 2015, Elsevier; p. 633–648. [DOI] [PubMed] [Google Scholar]
- Ostrander M. M., Ulrich-Lai Y. M., Choi D. C., Richtand N. M., Herman J. P. (2006). Hypoactivity of the hypothalamo-pituitary-adrenocortical axis during recovery from chronic variable stress. Endocrinology, 147(4), 2008–2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paxinos G., Franklin K. (2001). The mouse brain in stereotaxic coordinates: (deluxe). Academic Press. [Google Scholar]
- Perez-Garcia G., Gama Sosa M. A., De Gasperi R., Tschiffely A. E., McCarron R. M., Hof P. R., Gandy S., Ahlers S. T., Elder G. A. (2019). Blast-induced “PTSD”: Evidence from an animal model. Neuropharmacology, 145(Pt B), 220–229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ponsford J., Alway Y., Gould K. R. (2018). Epidemiology and natural history of psychiatric disorders after TBI. The Journal of Neuropsychiatry and Clinical Neurosciences, 30(4), 262–270. [DOI] [PubMed] [Google Scholar]
- Porter K. E., Stein M. B., Martis B., Avallone K. M., McSweeney L. B., Smith E. R., Simon N. M., Gargan S., Liberzon I., Hoge C. W., Rauch S. A. M. (2018). Postconcussive symptoms (PCS) following combat-related traumatic brain injury (TBI) in veterans with posttraumatic stress disorder (PTSD): Influence of TBI, PTSD, and depression on symptoms measured by the neurobehavioral symptom inventory (NSI). Journal of Psychiatric Research, 102, 8–13. [DOI] [PubMed] [Google Scholar]
- Ratcliff J. J., Greenspan A. I., Goldstein F. C., Stringer A. Y., Bushnik T., Hammond F. M., Novack T. A., Whyte J., Wright D. W. (2007). Gender and traumatic brain injury: Do the sexes fare differently? Brain Injury, 21(10), 1023–1030. [DOI] [PubMed] [Google Scholar]
- Rodriguez P. C., Ochoa A. C., Al-Khami A. A. (2017). Arginine metabolism in myeloid cells shapes innate and adaptive immunity. Frontiers in Immunology, 8, 93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rupprecht R., Papadopoulos V., Rammes G., Baghai T. C., Fan J., Akula N., Groyer G., Adams D., Schumacher M. (2010). Translocator protein (18 kDa)(TSPO) as a therapeutic target for neurological and psychiatric disorders. Nature Reviews. Drug Discovery, 9(12), 971–988. [DOI] [PubMed] [Google Scholar]
- Sackheim A. M., Stockwell D., Villalba N., Haines L., Scott C. L., Russell S., Hammack S. E., Freeman K. (2017). Traumatic brain injury impairs sensorimotor function in mice. The Journal of Surgical Research, 213, 100–109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sangobowale M. A., Grin'kina N. M., Whitney K., Nikulina E., St Laurent-Ariot K., Ho J. S., Bayzan N., Bergold P. J. (2018). Minocycline plus N-acetylcysteine reduce behavioral deficits and improve histology with a clinically useful time window. Journal of Neurotrauma, 35(7), 907–917. [DOI] [PubMed] [Google Scholar]
- Sangobowale M. A., N Gk., Whitney K., Nikulina E., St Laurent-Ariot K., Ho J. S., Bayzan N., Bergold P. J. (2018). Minocycline plus N-Acetylcysteine reduce behavioral deficits and improve histology with a clinically useful time window. J Neurotrauma, 35, 1–11. [DOI] [PubMed] [Google Scholar]
- Shinohara Y., Hosoya A., Yamasaki N., Ahmed H., Hattori S., Eguchi M., Yamaguchi S., Miyakawa T., Hirase H., Shigemoto R. (2012). Right-hemispheric dominance of spatial memory in split-brain mice. Hippocampus, 22(2), 117–121. [DOI] [PubMed] [Google Scholar]
- Shultz S. R., McDonald S. J., Vonder Haar C., Meconi A., Vink R., van Donkelaar P., Taneja C., Iverson G. L., Christie B. R. (2017). The potential for animal models to provide insight into mild traumatic brain injury: Translational challenges and strategies. Neuroscience and Biobehavioral Reviews, 76(Pt B), 396–414. [DOI] [PubMed] [Google Scholar]
- Silver J. M., McAllister T. W., Arciniegas D. B. (2018). Textbook of traumatic brain injury. American Psychiatric Pub. [Google Scholar]
- Simon D. W., McGeachy M. J., Bayır H., Clark R. S. B., Loane D. J., Kochanek P. M. (2017). The far-reaching scope of neuroinflammation after traumatic brain injury. Nature Reviews. Neurology, 13(3), 171–191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Skelton R. W. (1998). Modelling recovery of cognitive function after traumatic brain injury: Spatial navigation in the morris water maze after complete or partial transections of the perforant path in rats. Behavioural Brain Research, 96(1–2), 13–35. [DOI] [PubMed] [Google Scholar]
- Spadoni A. D., Huang M., Simmons A. N. (2018). Emerging approaches to neurocircuits in PTSD and TBI: Imaging the interplay of neural and emotional trauma In Behavioral neurobiology of PTSD (pp. 163–192). Springer. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sunyer B., P. S., Hoger H., Lubec G. (2007). Barnes maze, a useful task to assess spatial reference memory in mice. Nature Protocols, 198, 58–68. [Google Scholar]
- Tanev K. S., Pentel K. Z., Kredlow M. A., Charney M. E. (2014). PTSD and TBI co-morbidity: Scope, clinical presentation and treatment options. Brain Injury, 28(3), 261–270. [DOI] [PubMed] [Google Scholar]
- Teutsch P., Jones C. E., Kaiser M. E., Avalon Gardner N., Lim M. M. (2018). Gait and conditioned fear impairments in a mouse model of comorbid TBI and PTSD. Behavioural Neurology, 2018, 1–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vasterling J. J., Jacob S. N., Rasmusson A. (2018). Traumatic brain injury and posttraumatic stress disorder: Conceptual, diagnostic, and therapeutic considerations in the context of co-occurrence. The Journal of Neuropsychiatry and Clinical Neurosciences, 30(2), 91–100. [DOI] [PubMed] [Google Scholar]
- Villapol S., Loane D. J., Burns M. P. (2017). Sexual dimorphism in the inflammatory response to traumatic brain injury. Glia, 65(9), 1423–1438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walf A. A., Frye C. A. (2007). The use of the elevated plus maze as an assay of anxiety-related behavior in rodents. Nature Protocols, 2(2), 322–328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Watkins L. E., Sprang K. R., Rothbaum B. O. (2018). Treating PTSD: A review of evidence-based psychotherapy interventions. Frontiers in Behavioral Neuroscience, 12, 258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Whitaker A. M., Gilpin N. W., Edwards S. (2014). Animal models of post-traumatic stress disorder and recent neurobiological insights. Behavioural Pharmacology, 25(5–6), 398–409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wolf S. A., Boddeke H., Kettenmann H. (2017). Microglia in physiology and disease. Annual Review of Physiology, 79, 619–643. [DOI] [PubMed] [Google Scholar]
- Yurgil K. A., Barkauskas D. A., Vasterling J. J., Nievergelt C. M., Larson G. E., Schork N. J., Litz B. T., Nash W. P., Baker D. G. (2014). Association between traumatic brain injury and risk of posttraumatic stress disorder in active-duty marines. JAMA Psychiatry, 71(2), 149–157. [DOI] [PubMed] [Google Scholar]
- Zhao J., Huynh J., Hylin M. J., O'Malley J. J., Perez A., Moore A. N., Dash P. K. (2018). Mild traumatic brain injury reduces spine density of projection neurons in the medial prefrontal cortex and impairs extinction of contextual fear memory. Journal of Neurotrauma, 35(1), 149–156. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplemental material, sj-jpg-1-asn-10.1177_1759091420979567 for Increased Behavioral Deficits and Inflammation in a Mouse Model of Co-Morbid Traumatic Brain Injury and Post-Traumatic Stress Disorder by Arman Fesharaki-Zadeh, Jeremy T. Miyauchi, Karrah St. Laurent-Arriot, Stella E. Tsirka and Peter J. Bergold in ASN Neuro
Supplemental material, sj-pdf-2-asn-10.1177_1759091420979567 for Increased Behavioral Deficits and Inflammation in a Mouse Model of Co-Morbid Traumatic Brain Injury and Post-Traumatic Stress Disorder by Arman Fesharaki-Zadeh, Jeremy T. Miyauchi, Karrah St. Laurent-Arriot, Stella E. Tsirka and Peter J. Bergold in ASN Neuro