Abstract
The mechanism of H. pylori-induced atrophy and metaplasia has not been fully understood. Here, we demonstrate the novel role of Apoptosis signal-regulating kinase 1 (ASK1) and downstream MAPKs as a regulator of host immune responses and epithelial maintenance against H. pylori infection. ASK1 gene deficiency resulted in enhanced inflammation with numerous inflammatory cells including Gr-1+CD11b+ myeloid-derived suppressor cells (MDSCs) recruited into the infected stomach. Increase of IL-1β release from apoptotic macrophages and enhancement of TH1-polarized immune responses caused STAT1 and NF-κB activation in epithelial cells in ASK1 knockout mice. Dysregulated immune and epithelial activation in ASK1 knockout mice led to dramatic expansion of gastric progenitor cells and massive metaplasia development. Bone marrow transplantation experiments revealed that ASK1 in inflammatory cells is critical for inducing immune disorder and metaplastic changes in epithelium, while ASK1 in epithelial cells regulates cell proliferation in stem/progenitor zone without changes in inflammation and differentiation. These results suggest that H. pylori-induced immune cells may regulate epithelial homeostasis and cell fate as an inflammatory niche via ASK1 signaling.
Keywords: Helicobacter pylori, ASK1, gastritis, metaplasia, progenitors
1. Introduction
Helicobacter pylori (H. pylori) is associated with various human gastric diseases, including gastric ulcer, chronic gastritis, and gastric neoplasia [1,2]. During H. pylori-induced chronic gastritis, the initial pathological change is oxyntic atrophy or loss of parietal and chief cells [3]. Parietal and chief cell loss is considered to alter the status of proliferation or differentiation in oxyntic glands, and lead to induce metaplastic changes [4,5,6,7]. Recent published papers have identified several stem cell markers in the stomach [8,9,10,11,12,13,14]. However, it has been poorly understood how gastric stem/progenitor cells could differentiate to metaplasia or dysplasia during H. pylori infection.
In humans and mice, the H. pylori-infected gastric mucosa exhibits high levels of proinflammatory cytokines including IL-8, IL-1β, TNF-ɑ, and IL-6 [15,16,17,18,19,20]. Especially in mice, TH1-polarized cellular immune responses are highly involved, resulting in elevated levels of IFN-γ, IL-12, and IL-18 [17,21,22,23]. These numerous cytokines are produced by various kinds of recruited inflammatory cells such as macrophages, neutrophils, lymphocytes, dendritic cells, or fibroblasts, and such inappropriate immune responses in the host may eventually cause the gastric phenotype by H. pylori [23,24,25,26]. Indeed, polymorphisms in proinflammatory cytokine genes have been proved to increase the risk of gastric cancer [27,28].
A series of in vitro reports have shown the importance of cag-PAI in the pathogenesis of H. pylori-induced gastric epithelial changes. CagA that infected into host cells leads to the phosphorylation of CagA by host cell kinases, resulting in activation of SHP-2 tyrosine phosphatase, NF-κB signaling pathways, and mitogen-activated protein kinase (MAPK) signaling pathways [29,30,31,32,33]. Apoptosis signal-regulating kinase 1 (ASK1) is a ubiquitously expressed MAPK kinase kinase (MAP3K), which activates the c-Jun N-terminal kinase (JNK) and p38 signaling pathways and is required for both oxidative stress and cytokine-induced apoptosis [34]. Previously, our studies have shown that ASK1 has important functions in gastric epithelial cells for controlling proliferation and cell cycle both in vivo and in vitro [35,36]. It was also reported that ASK1 and downstream signaling play a critical role in various cancer development, including colon, pancreas, and liver cancers [37,38,39].
It has been also reported that ASK1 is highly involved in immune responses. Lipopolysaccharide (LPS) or tumor necrosis factor-ɑ (TNF-ɑ) induced the formation of a complex of TRAF6 and ASK1, and subsequent activation of the ASK1-p38 pathway in inflammatory cells [40,41]. During colonic inflammation, ASK1 regulates bacterial killing ability in macrophages and controls their cell fate, which finally affects systemic immune responses and carcinogenesis in mice [37]. However, it has not been fully elucidated whether ASK1 plays a role in H. pylori-induced host immune responses and affects epithelial differentiation.
In this study, we demonstrate that lack of ASK1 causes severely enhanced mucosal changes in the H. pylori-infected stomach. In particular, ASK1 in inflammatory cells plays a critical role for preventing gastric atrophy and metaplastic changes through IL-1β release, TH1-polarized immune responses, and the recruitment of Gr-1+CD11b+ myeloid-derived suppressor cells (MDSCs), while ASK1 in epithelial cells regulates stem/progenitor cell proliferation. Moreover, abnormal inflammatory responses that were caused by ASK1 deficiency resulted in altered distribution of gastric facultative progenitor cells in metaplastic lesions. These results highlight the novel role of ASK1 in the interaction between host immune responses and epithelial homeostasis during H. pylori infection.
2. Materials and Methods
2.1. Mice and Helicobacter Infection Model
The generation of ASK1–/– and ASK2–/– mice was described previously [42,43]. ASK1–/– and ASK2−/− mice were back-crossed into the C57BL/6 strain at least 18 times. C57BL/6 WT mice were purchased from Clea Japan (Tokyo, Japan). The Helicobacter strain used in this study was the H. pylori Sydney strain 1 (SS-1) [44] and PMSS-1, which was kindly provided from Dr. Anne Muller [45]. H. pylori was grown as described previously [46]. Six-week-old male WT and ASK1−/− mice were inoculated against H. pylori using three oral gavage dosages per week (108 colony-forming units/0.2 mL). The stomachs were removed and used for histological and immunoblot analyses. For quantitative assessment of H. pylori colonization, one section of each stomach was transferred to a tube containing Brucella broth (Becton, Dickinson and Company, Sparks, MD, USA), and homogenized. Serial dilutions were plated on Brucella agar (Becton, Dickinson and Company) plates to determine bacterial loads. All of the experimental protocols (P19-102) were approved by the Ethics Committee for Animal Experimentation and conducted in accordance with the Guidelines for the Care and Use of Laboratory Animals of the Graduate School of Medicine, the University of Tokyo, and the Institute for Adult Diseases, Asahi Life Foundation, Tokyo, Japan.
2.2. Reagents
Anti-phospho-JNK, anti-JNK, anti-phospho-p38, anti-p38, anti-phospho-MKK3, anti-phospho-MKK4, anti-phospho-ATF2, anti-phospho-MK2, anti-phospho-CREB, anti-phospho-cjun, anti-phospho-IκBɑ, anti-phospho-STAT1, anti-phospho-STAT3, anti-IL-1β, anti-cleaved caspase-3, and anti-phospho-ASK1 antibodies were purchased from Cell Signaling Technology (Danvers, MA, USA). Anti-ASK1 antibody was provided from Dr. Hidenori Ichijo. Anti-F4/80 antibody was obtained from Serotec. Anti-CD4, anti-caspase-1, and anti-Dclk1 antibodies were purchased from Abcam (Cambridge, MA, USA). Anti-TFF2 antibody was kindly provided by Dr. Sachiyo Nomura. Anti-proton pump and anti-Cdx2 antibodies were obtained from Santa Cruz (Santa Cruz, CA, USA). Anti-BrdU antibody was obtained from Dako (Santa Clara, CA, USA). Anti-actin antibody was purchased from Sigma. Anti-cyclin D1(AB3) was acquired from Invitrogen (Waltham, MA, USA).
2.3. Bone Marrow Chimeric Mice Generation
Bone marrow transplantation was performed as described previously [37]. Cell suspensions from female WT or ASK1−/− bone marrow were prepared from femurs and tibias, filtered, and counted. Male recipient mice received a single intravenous injection of 1 × 107 bone marrow cells, after being irradiated with 9.5 Gy x-rays. For 4 weeks following the transplant, drinking water was replaced with water containing neomycin sulfate/polymyxin B sulfate mixture. Transplanted mice were treated with H. pylori 6 weeks after irradiation. Genomic DNA and protein were extracted from spleen, and bone marrow chimerism was determined by PCR and Western blotting.
2.4. Cell Lines
AGS cell line was purchased from ATCC. The cell lines were cultured in Ham’s F-12 medium supplemented with 10% fetal bovine serum.
2.5. Immunostaining
Tissues were fixed in 10% formaldehyde, dehydrated, embedded in paraffin, and sectioned as described previously [47]. The sections were deparaffinized and incubated overnight at 4 °C with indicated antibodies. Binding of the primary antibody was detected with anti-rabbit IgG (Vector Laboratories), followed by visualization with 3,3’-diaminobenzidine (Sigma-Aldrich). To assess cellular proliferation, mice were injected i.p. with 120 mg/kg of bromodeoxyuridine (BrdU; BD-Pharmingen, San Diego, CA, USA) 90 min before sacrifice, and gastric tissues were stained with anti-BrdU antibody. For immunofluorescence, the paraffin-embedded or frozen sections were incubated with primary antibodies, followed by secondary Alexa555 or Alexa488 IgG antibody (Invitrogen). TUNEL was analyzed by using the Apoalert DNA Fragmentation Assay kit (Takara, Shiga, Japan).
2.6. Western Blotting, Immunoprecipitation, and ELISA
Protein lysates were prepared from cells or tissues, separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE), transferred to polyvinylidene difluoride membranes (Millipore, St. Louis, MO, USA). The membrane was probed with primary antibodies, and then incubated with the secondary antibody. Immunocomplexes were detected using the enhanced chemiluminescence system (Amersham Biosciences, Buckinghamshire, UK). For immunoprecipitation, samples were lysed in radioimmunoprecipitation assay buffer and immunoprecipitated with 50 μL of protein A/G Sepharose beads (Santa Cruz Biotechnology) overnight at 4 °C using the indicated antibodies. The beads were washed 3 times with radioimmunoprecipitation buffer and then analyzed by SDS-PAGE. Cytokine levels were measured using ELISAs (R&D Systems, Minneapolis, MN, USA).
2.7. RNA Analysis
Total RNA was extracted from the tissues using the Nucleospin RNA II Kit (Takara). The first-strand complementary DNA was synthesized using the ImProm-II Reverse Transcription System (Promega, Madison, WI, USA). Amplification was performed using the ABI PRISM 7000 Quantitative PCR System (Applied Biosystems). The different mRNAs were quantitated by Real-Time PCR using Gapdh mRNA for normalization. The primer sequences used are available upon request.
2.8. Statistical Analyses
Differences between means were compared using Student’s t-test. P values < 0.05 were considered statistically significant.
3. Results
3.1. Loss of ASK1 Exacerbated Atrophic and Metaplastic Changes in H. pylori-Infected Stomach
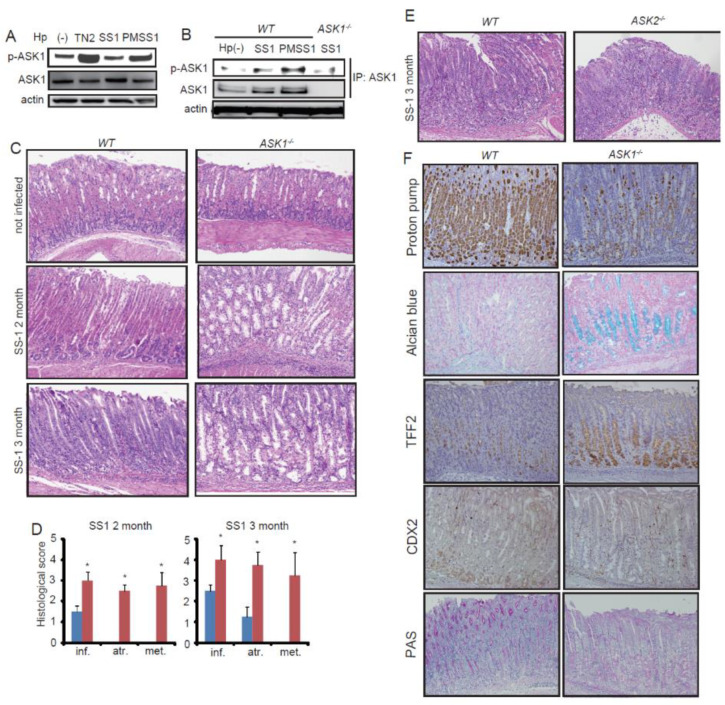
Type IV secretion system (TFSS) has been reported to be important for MAPK pathway activation in gastric epithelial cells [29,30,31,32]. Although H. pylori SS-1 is a well-known strain which can colonize to mouse stomach, functional TFSS is disrupted in this strain [48]. PMSS-1 strain, which was the primarily isolated strain from SS-1-infected patients, has efficient TFSS, and induces more aggressive gastritis in mouse stomach than SS-1 [19,45], suggesting the possibility that efficient TFSS could cause more severe inflammation by activating epithelial MAPK signaling cascade. Thus, first we used both SS-1 and PMSS-1 strains in order to analyze ASK1 activation in vitro and in vivo. In AGS cells, H. pylori TN2 strain and PMSS-1 could increase the phosphorylated level of ASK1, while SS-1 did not, suggesting that TFSS is necessary for ASK1 activation in gastric epithelial cells (Figure 1A). Next, we infected WT and ASK1−/− mice with SS-1 and PMSS-1 for up to 3 months and examined protein expression in the stomachs. Both infected stomachs showed increased level of ASK1 phosphorylation compared to noninfected stomach, and PMSS-1-infected stomach displayed greater level of phosphorylated ASK1 than SS-1-infected stomach (Figure 1B). These results indicate that PMSS-1 activates ASK1 in gastric epithelium via phosphorylation more strongly than SS-1.
Figure 1.
Loss of ASK1 exacerbated atrophic and metaplastic changes in H. pylori-infected stomach. (A) ASK1 phosphorylation in AGS cells that were transfected with ASK1-overexpressing vector. Cells were harvested 24 h after H. pylori infection. (B) ASK1 phosphorylation in mouse stomachs from control (Hp(-)), SS-1-infected, and PMSS-1-infected mice (3 mo post-infection). (C) Representative H&E staining of WT and ASK1−/− mouse stomach with/without H. pylori SS-1 infection. Original magnification, ×100. (D) Histological scoring of SS-1-infected WT and ASK1−/− mouse stomach. Inf., inflammation; atr., atrophy; met., metaplasia; hyp., hyperplasia. Data are shown as mean ± SD. * p <0.05 compared to infected WT mice. (E) H&E staining of SS-1-infected WT and ASK2−/− mouse stomach. Mice were sacrificed 3 mo after SS-1 infection. Original magnification, ×100. (F) Immunohistochemical staining of SS-1-infected WT and ASK1−/− mouse stomachs. Original magnification, ×100.
Noninfected WT and ASK1−/− mice did not show significant differences in the morphology of their stomachs (Figure 1C) as reported previously [35]. However, ASK1−/− mice infected with SS-1 exhibited dramatically increased level of inflammation, gastric atrophy, and metaplasia, compared to WT infected mice after 2 and 3 months (mo) infection (Figure 1C,D). PMSS-1 induced more severe inflammation and atrophic changes in WT mice than SS-1 as reported previously [45]; however, ASK1 deficiency further exacerbated gastric atrophy and metaplasia even in the setting of PMSS-1 infection (Figure S1A,B). There were no significant differences between WT and ASK1−/− mice in SS-1 and PMSS-1 colonization (Figure S1C). These results suggest that systemic ASK1 deficiency enhanced inflammation and metaplastic changes independently from TFSS-mediated signaling and bacterial clearance.
It has been reported that ASK2 is closely related to ASK1 but can activate the JNK and p38 pathways only by forming a heteromeric complex with ASK1, and thus ASK1−/− mice lack ASK2 function [43]. To determine whether the more prominent phenotype in ASK1−/− mice was dependent on ASK1 or ASK2 function, we infected WT and ASK2−/− mice with H. pylori. After 2 mo infection, infected ASK2−/− mice did not show enhanced mucosal changes compared to WT mice (Figure 1E).
Next, we performed immunohistochemistry (IHC) of SS-1-infected WT and ASK1−/− stomach. Alcian blue and proton pump staining confirmed that the development of metaplasia and parietal cell loss was significantly enhanced in ASK1−/− stomach (Figure 1F). It has been reported that there are two types of metaplasia in the stomach: one is the traditional intestinal metaplasia, which is characterized by goblet cell morphology and Cdx2-positive; the other is spasmolytic polypeptide expressing metaplastic lineage (SPEM), a metaplasia in the gastric fundus resembling deep antral gland cells and expressing Trefoil Factor 2 (TFF2) [49,50,51,52]. The metaplastic lesion found in ASK1−/− stomach was TFF2-positive and CDX2-negative, suggesting that these metaplastic changes were SPEM (Figure 1F). Interestingly, although pit-cell hyperplasia is generally accompanied with atrophy and metaplasia during Helicobacter infection, PAS staining showed the decrease of pit-cell hyperplasia in ASK1−/− stomach compared to WT stomach, in contrast to atrophy and metaplasia (Figure 1F). These results indicate that ASK1, not ASK2, is a critical regulator of the development of inflammation and metaplasia in the stomach.
3.2. ASK1 Regulates Stem/Progenitor Cell Proliferation Through Downstream MAPK Activation
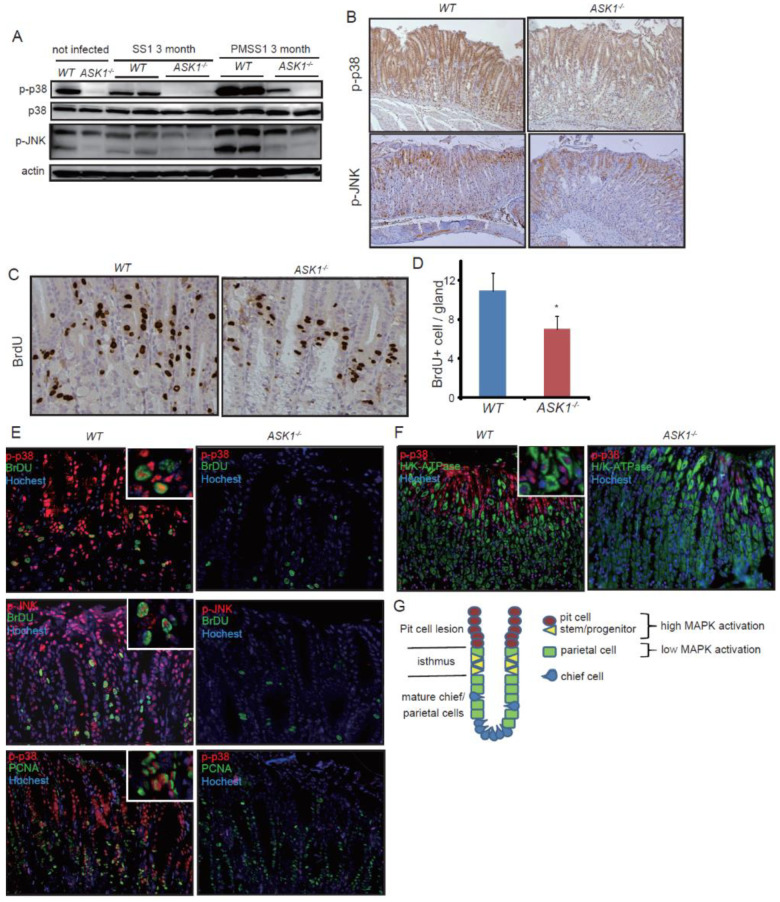
We next tested phosphorylation of JNK and p38, the downstream molecules of ASK1. PMSS-1 enhanced phosphorylation of both JNK and p38 in WT mice, while the effect of SS-1 infection was minimal. Interestingly, ASK1−/− mice showed markedly decreased levels of MAPK activity either with or without Hp infection, compared to WT mice (Figure 2A). In contrast, immunoblotting of ASK2−/− stomach and spleen showed same levels of phosphorylated p38 (p-p38) and phosphorylated JNK (p-JNK) between WT and ASK2−/− mice (Figure S2A). Consistent with the immunoblotting results, immunostaining revealed that ASK1−/− mice showed decreased expression of phopho-p38 and phospho-JNK in the nuclei, which was most evident in the isthmus-surface area of gastric mucosa (Figure 2B). Total p38 expression was preserved in the cytoplasm in WT and ASK1−/− stomach (Figure S2B). These results suggest that epithelial MAPK activation is mostly restricted to the isthmus and pit cell region and largely regulated by upstream ASK1 in the stomach.
Figure 2.
ASK1 regulates stem/progenitor cell proliferation through downstream MAPK activation. (A) Immunoblotting of WT and ASK1−/− mice stomach with/without H. pylori infection with the indicated proteins. (B) Immunohistochemical staining of SS-1-infected WT and ASK1−/− mouse stomachs with the indicated proteins. Original magnification, ×100. (C,D) BrdU staining (C) and the numbers of BrdU+ cells per gland (D) in SS-1-infected WT and ASK1−/− mice. Original magnification, ×200. Data are shown as mean ± SE. * p < 0.05 compared to infected WT mice. (E,F) Immunofluorescence in SS-1-infected WT and ASK1−/− mice. Original magnification, ×200. Colors of the stained proteins are indicated in the panels. (G) Schema of corpus gland structure.
To investigate the effect of ASK1 on gastric epithelium more precisely, we next performed BrdU labeling assay. In the glandular stomach, BrdU-positive cells were seen in the isthmus region where stem/progenitor cells are considered to reside (Figure 2C). BrdU staining showed the significantly decreased numbers of BrdU+ cells in ASK1−/− stomach compared to WT stomach (Figure 2D). Double immunofluorescence staining revealed that the phosphorylation of p38 and JNK was found to be strongest in the isthmus and surface pit region, and that approximately 50% of BrDU-positive actively cycling stem/progenitor cells was positive for phospho-p38 and phospho-JNK (Figure 2E). In contrast, ASK1−/− stomach showed quite weak or no JNK/p38 activation in the isthmus and pit cells (Figure 2E). Proliferating cell nuclear antigen (PCNA) staining demonstrated similar results to BrdU staining (Figure 2E). We observed weak p38 phosphorylation in mature parietal cells in WT mice, but p38 phosphorylation in ASK1−/− parietal cells was almost absent (Figure 2F). These suggest that p38 and JNK are highly activated in the isthmus stem/progenitor cells and the surface pit cells, and activation of p38 and JNK may promote isthmus proliferation and pit-cell differentiation (Figure 2G).
3.3. ASK1 Deficiency Enhanced NF-κB and STAT1 Activation in the Hp Infected Stomach
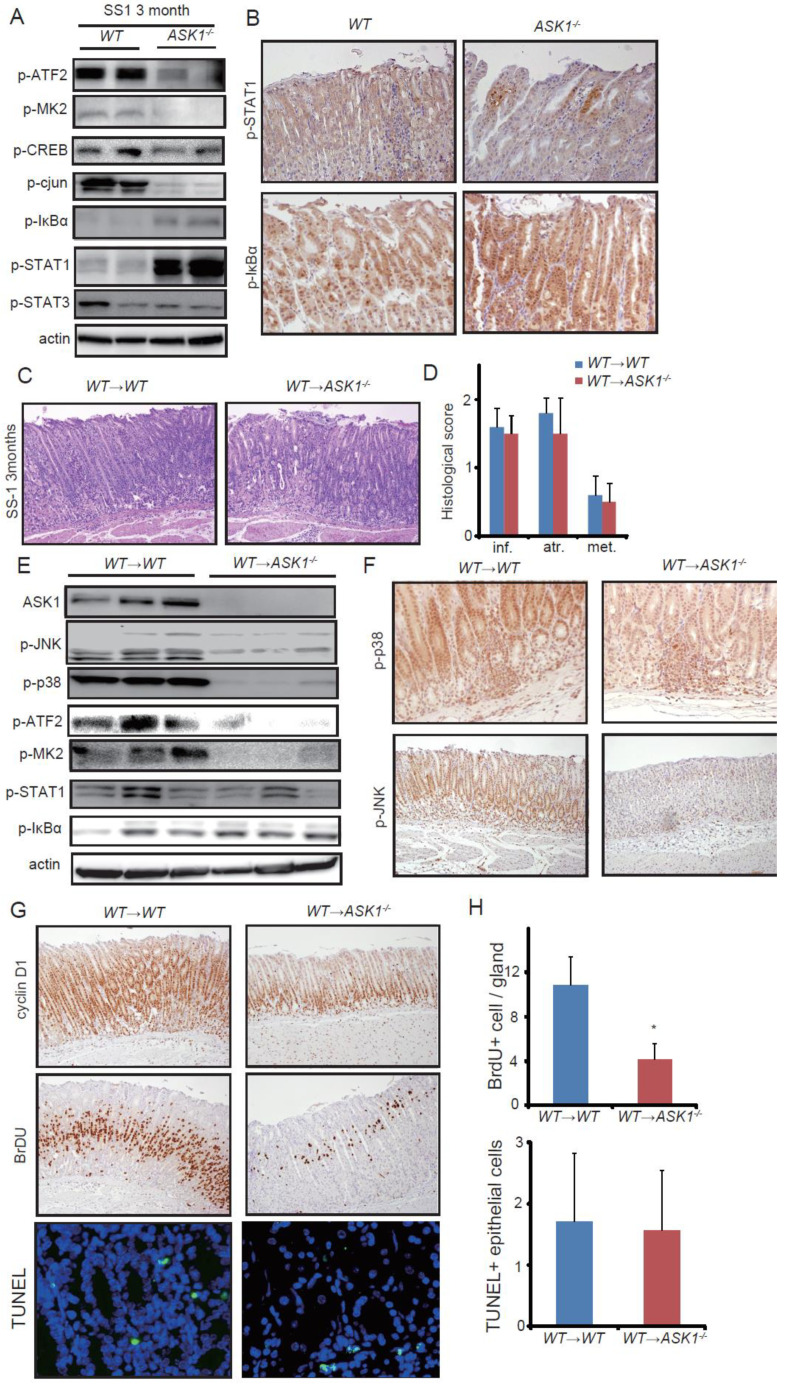
To further address downstream transcriptional factors that are affected by ASK1 in Hp-infected epithelium, we performed additional immunoblotting. It is known that JNK activates c-jun by phosphorylation and that p38 regulates the activation of several transcriptional factors, including activating transcription factor (ATF), MAPK-activated protein kinase 2 (MK2), and cAMP response element binding protein (CREB) [53,54,55]. Immunoblotting of SS-1 and PMSS-1 infected stomachs revealed the decrease levels of phopho-ATF, phospho-MK2, and phospho-c-jun, but did not show the deference in phospho-CREB (Figure 3A and Figure S3). MAPK pathways have various interactions with other signaling pathways such as NF-κB and signal transducer and activator of transcription family (STAT) signaling, and these signaling pathways are also important for gastric inflammation and carcinogenesis [56,57,58,59]. Interestingly, infected ASK1−/− stomach showed enhanced activation of NF-κB and STAT1, but not STAT3 (Figure 3A and Figure S3). In immunohistochemistry, we observed stronger activation of NF-κB and STAT1 in epithelial cells in infected ASK1−/− mice compared to infected WT mice (Figure 3B). These results suggest that downregulation of p38 and JNK in ASK1−/− stomach might induce compensatory activation of NF-κB and STAT1 pathways, which potentially lead to gastric atrophy and metaplasia.
Figure 3.
Epithelial ASK1 does not contribute to inflammation and metaplasia development, but mediates proliferation. (A) Immunoblotting of the indicated proteins in SS-1-infected WT and ASK1−/− mouse stomachs. (B) Immunohistochemical staining of SS-1-infected WT and ASK1−/− mouse stomachs with the indicated proteins. (C) H&E staining of WT (WT→WT) and ASK1−/− (WT→ASK1−/−) mice transplanted with WT bone marrow. Mice were sacrificed 3 mo after SS-1 infection. Original magnification, ×100. (D,E) Histological scoring (D), and immunoblotting (E) of SS-1-infected WT→WT and WT→ASK1−/− mice. (F–H) Immunohistochemistry of the indicated proteins (F,G) and the numbers of BrdU and TUNEL positive cells per gland (H) in SS-1-infected WT→WT and WT→ASK1−/− mice. Original magnification: TUNEL, ×400; p-p38, ×200; p-JNK, cyclin D1, and BrdU, ×100. Data are shown as mean ± SE. * p < 0.05 compared to infected WT→WT mice.
3.4. Epithelial ASK1 Did Not Affect Inflammatory and Metaplastic Changes After H. pylori Infection
In order to explore the precise function of ASK1 in these phenotypes, we next transplanted bone marrow cells derived from WT mice into WT and ASK1−/− mice. After 2 mo infection with SS-1, ASK1−/− mice transplanted with WT bone marrow (WT→ASK1−/−) exhibited similar levels of inflammation, atrophy, and metaplasia to control WT transplanted mice (WT→WT) (Figure 3C,D). Immunoblotting of the stomach tissue from these mice showed decreased activity of JNK, p38, and downstream ATF and MK2, while similar levels of STAT1 and NF-κB activation (Figure 3E). IHC with phospho-p38 and phospho-JNK revealed that activation of these molecules was reduced in the epithelial cells, but not in the myeloid cells (Figure 3F). In the WT→ASK1−/− mouse stomach, the number of cyclin D1 and BrDU positive proliferating cells were decreased compared to WT→WT mice (Figure 3G,H). To analyze epithelial apoptosis after H. pylori infection, we performed TUNEL staining. In the oxyntic gland, we found rare TUNEL-positive cells, but no difference was seen between WT→WT and WT→ASK1−/− mice (Figure 3G,H). These results indicate that ASK1 in the epithelial cells is not associated with the enhanced gastric inflammation and metaplastic changes seen in systemic ASK1−/− mice, but has a promoting effect on epithelial cell proliferation.
3.5. ASK1 Deficiency Promotes TH1-Dependent Immune Response and Recruits Immature Gr-1+CD11b+ Cells
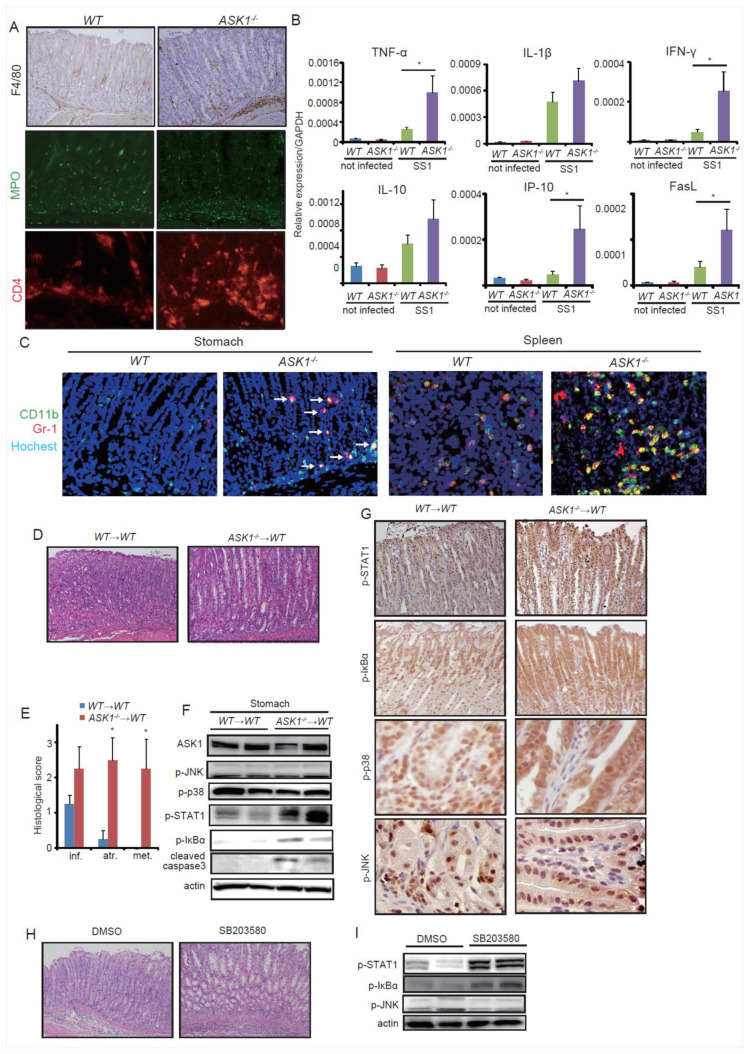
To further elucidate the direct cause of dramatic metaplasic changes in ASK1−/− stomach, we assessed the inflammatory profile after H. pylori infection in both genotypes. To investigate which types of inflammatory cells were recruited in H. pylori-infected WT and ASK1−/− stomach, we stained with immune cell surface markers. F4/80, myeloperoxidase (MPO), and CD4 staining indicated a marked increase in the recruitment of macrophages, neutrophils, and T lymphocytes in ASK1−/− stomach (Figure 4A). When we analyzed the expression of inflammatory cytokines by real-time PCR, Th1-polarized factors TNF-ɑ, IFN-γ, and IP-10 were significantly upregulated in H. pylori-infected ASK1−/− stomach compared to infected WT stomach, while the induction of IL-1β and IL-10, which were reported as important for gastric inflammation [18,60], were not significantly different between these genotypes (Figure 4B). It has been reported that IFN-γ could induce FasL production and that FasL may play a role in the development of gastric atrophy [61,62,63]. Indeed, our data showed the significant increase of FasL expression in ASK1−/− stomach compared to WT stomach (Figure 4B). Moreover, ASK1−/− stomach showed the increase of Gr-1+Cd11b+ immature myeloid cells (Figure 4C), which have been reported as a critical regulator of inflammation-associated cancer [18,64,65]. Gr-1+Cd11b+ MDSCs were also increased in ASK1−/− spleen (Figure 4C), suggesting that myeloid maturation was inhibited in ASK1−/− mice and that immature MDSCs were recruited to the stomach from the spleen in response to exaggerated inflammation. These results suggest that in ASK1−/− stomach, Th1-dependent immune responses were more activated than WT mice, and that the recruitment of immature myeloid cells and the increased production of cytokines and apoptotic ligands may lead to the development of gastric atrophy and metaplasia.
Figure 4.
Impaired ASK1-p38 pathway in myeloid lineage promotes atrophic and metaplastic changes through NF-κB and STAT1 activation. (A) Immunostaining of immune cell markers in SS-1-infected WT and ASK1−/− mouse stomachs. Original magnification: CD4, ×200; others, ×100. (B) Relative mRNA expression per GAPDH in WT and ASK1−/− mouse stomachs with or without SS-1 infection. Data are shown as mean ± SE. * p < 0.05 compared to infected WT mice. (C) Immunofluorescence of CD11b (green) and Gr-1 (red) in SS-1-infected WT and ASK1−/− mouse stomachs and spleens. Original magnification, ×200. (D) H&E staining of WT mice transplanted with WT (WT→WT) or ASK1−/− (ASK1−/−→WT) bone marrow. Mice were sacrificed 3 mo after SS-1 infection. Original magnification, ×100. (E–G) Histological scoring (E), immunoblotting (F), and immunohistochemical staining (G) of SS-1-infected WT→WT and WT→ASK1−/− mice. Original magnification: p-p38 and p-JNK, ×400; others, ×200. Data are shown as mean ± SE. * p < 0.05 compared to infected WT→WT mice. (H,I) H&E staining (H) and immunoblotting (I) of H. pylori-infected mouse stomach treated with or without SB203580. Mice were sacrificed 3 mo after SS-1 infection. Original magnification, ×100.
3.6. Lack of ASK1 and p38 Activation in Myeloid Lineage Induced Atrophic and Metaplastic Changes through NF-κB and STAT1 Activation
To investigate the role of ASK1 in myeloid cells, we next transplanted WT and ASK1−/− bone marrow into WT mice (WT→WT and ASK1−/−→WT). Interestingly, ASK1−/−→WT mice infected with SS-1 for 2 months exhibited more severe gastric atrophy and metaplasia than WT→WT mice (Figure 4D,E). In the stomach, STAT1 and NF-κB activation were enhanced in ASK1−/−→WT mice, while the levels of p38 and JNK activation did not differ from WT→WT mice (Figure 4F). IHC with these molecules confirmed that STAT1 and NF-κB were activated in epithelial cells, and that p38 and JNK activation were decreased only in myeloid cells of ASK1−/−→WT mice (Figure 4G). These results suggest that decreased activity of p38 and JNK in ASK1−/− myeloid cells causes STAT1 and NF-κB activation in epithelial cells, followed by atrophic and metaplastic changes.
To address whether p38 or JNK is essential for the protection of H. pylori-induced mucosal changes, pharmacological inhibition of these molecules was performed. We found that treatment with p38 inhibitor SB203580 worsened H. pylori-induced atrophy and metaplasia (Figure 4H), while JNK inhibitor SP600125 treatment did not (not shown). Treatment with p38 inhibitor induced STAT1 and NF-κB activation in the stomach, as seen in ASK1−/− or ASK1−/−→WT mice (Figure 4I), suggesting that ASK1-p38 pathway in myeloid cells is a critical regulator of H. pylori-induced gastritis and metaplasia.
3.7. ASK1 and p38 Suppressed Macrophage Cell Death and IL-1β Secretion
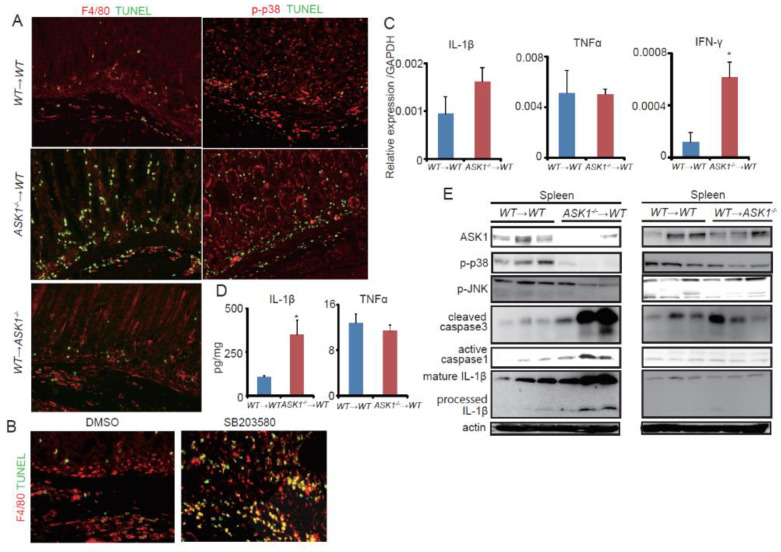
As we previously reported that ASK1 protects macrophage apoptosis in the models of colitis [37], we examined macrophage apoptosis in H. pylori-infected gastric tissues by TUNEL staining (Figure 5A). In H. pylori-infected WT→WT mouse stomach, rare TUNEL-positive cells were seen in both stromal and epithelial cells. However, in ASK1−/−→WT mice, a greater number of TUNEL-positive cells were found in the submucosa than control groups, while WT→ASK1−/− mouse stomach appears similar to control group. Increased apoptosis in the stomach was confirmed by immunoblotting with cleaved caspase-3 (Figure 4F). Double staining with F4/80 showed that the majority of these apoptotic cells were F4/80-positive macrophages (Figure 5A). Indeed, CD4 or MPO-positive cells were rarely positive for TUNEL staining in both genotypes (Figure S4). Thus, ASK1−/− macrophage were more susceptible to cell death in H. pylori-infected stomach, as seen in the previous colitis models [37]. Although most of inflammatory cells were phospho-p38-positive in WT→WT mice, inflammatory cells in ASK1−/−→WT mouse stomach were mostly phospho-p38-negative (Figure 5A). These phospho-p38-negative immune cells were likely to positive for TUNEL staining (Figure 5A), and treatment with p38 inhibitor SB203580 increased apoptosis in F4/80 positive macrophages (Figure 5B). Thus, ASK1-p38 pathway likely protects macrophage cell death.
Figure 5.
ASK1 and p38 suppressed macrophage cell death and IL-1β secretion in the stomach and spleen. (A) Immunofluorescent staining of SS-1-infected stomach isolated from WT→WT, ASK1−/−→WT, and WT→ASK1−/− mice. Red, F4/80 and p-p38; Green, TUNEL. Original magnification, ×200. (B) Immunofluorescent staining of SS-1-infected stomach treated with DMSO or SB203580. Red, F4/80; Green, TUNEL. Original magnification, ×200. (C) Relative mRNA expression in WT→WT and ASK1−/−→WT mouse spleens with SS-1 infection. Data are shown as mean ± SE. * p < 0.05 compared to infected WT→WT mice. (D) Protein levels of the indicated cytokines in WT→WT and ASK1−/−→WT mouse spleens measured by ELISA. Data are shown as mean ± SE. * p < 0.05 compared to infected WT→WT mice. (E) Immunoblotting of WT→WT, ASK1−/−→WT, and WT→ASK1−/− mouse spleens with SS-1 infection. Data are shown as mean ± SE. * p < 0.05.
Previous papers reported that processed IL-1β was released from dead macrophage through caspase-1 dependent pathway [66]. Thus, we investigated the expression of IL-1β and other cytokines in H. pylori-infected mouse spleen. We found no significant differences in mRNA expression of IL-1β or TNF-ɑ between WT→WT and ASK1−/−→WT mouse spleens (Figure 5C). On the other hand, protein level of IL-1β was found by ELISA to be increased in ASK1−/−→WT spleen, but TNF-ɑ protein was not upregulated (Figure 5D). Immunoblotting of these spleens revealed that ASK1−/−→WT spleen exhibited lower levels of phospho-p38 and phospho-JNK with higher levels of cleaved caspase-3, active form of caspase-1, and processed or matured form of IL-1β (Figure 5E). In contrast, WT→ASK1−/− spleen showed similar levels of these molecules to WT→WT spleen (Figure 5E). We also found upregulation of IFN-γ in ASK1−/−→WT spleen as seen in ASK1−/− stomach (Figure 5C), indicating that T cells were activated in ASK1−/− spleen and stomach. Taken together, lack of ASK1 and p38 induced macrophage apoptosis in Hp-infected stomach and spleen, and overload of processed IL-1β and IFN-γ production appears to be a critical inducer of epithelial changes.
3.8. ASK1 Deficiency Induced the Expansion of Facultative Gastric Progenitor Cells
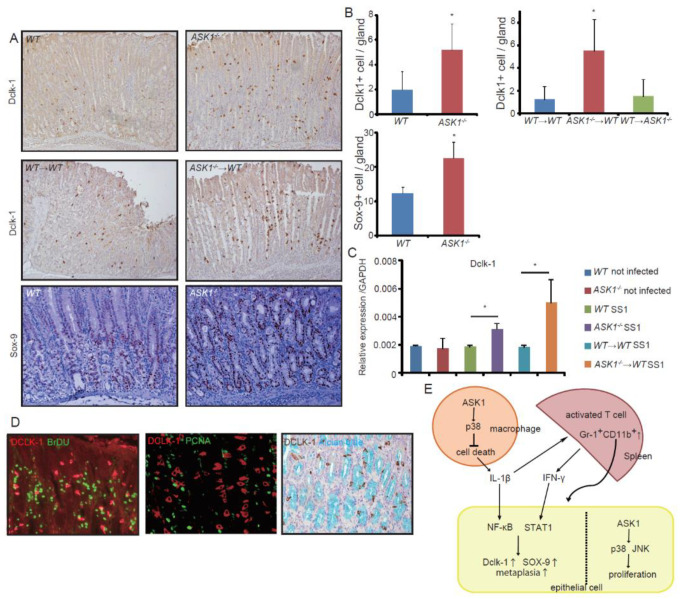
Previous studies have reported that stem/progenitor cells play a critical role in maintaining gastric mucosal homeostasis and carcinogenesis [8,10,13]. In the gastrointestine, Dclk1+ tuft cells are thought to be facultative progenitors that act as an origin of cancers in a specific condition [25,67,68,69]. Thus, we examined the distribution of Dclk1-positive cells in WT and ASK1−/− stomach. In ASK1−/− and ASK1−/−→WT stomach, the numbers of Dclk1-positive cells were significantly increased compared to WT and WT→WT stomach (Figure 6A,B). The expression level of Dclk1 mRNA was also upregulated in ASK1−/− and ASK1−/−→WT stomach (Figure 6C). The number of cells expressing another stem/progenitor cell marker Sox9 [19] was also increased in ASK1−/− stomach compared to WT mice (Figure 6A,B).
Figure 6.
ASK1 deficiency induced the expansion of gastric facultative progenitor cells. (A,B) Dclk-1 and Sox9 staining (A) and the numbers of positive cells per gland in the indicated mouse stomachs. Original magnification, ×100. (C) Relative Dclk1 mRNA expression in the indicated mouse stomachs. (D) Immunohistochemical staining of the indicated markers in ASK1−/− mouse stomach infected with SS-1. Original magnification, ×200. (E) Schematic model of the roles of ASK1 in myeloid cells, spleen, and epithelium.
Even in the inflamed stomach, Dclk1-positive cells remain BrDU-negative, i.e., quiescent, however, those were accumulated very close to BrDU-positive or PCNA-positive isthmus stem/progenitor cells (Figure 6D). In addition, plenty of Dclk1-positive cells were observed in Alcian blue-positive, BrDU-negative metaplastic gland (Figure 6D). Therefore, largely quiescent Dclk1-positive tuft cells expand within the inflamed mucosa of ASK1−/− mice, and may support the isthmus proliferation and development of metaplasia as a niche cell.
In summary, we observed that the lack of ASK1 in myeloid cells dramatically affected H. pylori-induced epithelial changes, including atrophy, metaplasia, and the expansion of gastric progenitors through disrupted immune responses (Figure 6E). In contrast, epithelial ASK1 is important for stem/progenitor cell proliferation. ASK1 and downstream molecules have multiple functions on gastric inflammation and homeostasis.
4. Discussion
In this report, we showed that ASK1 deficiency promoted gastric inflammation, atrophy, and metaplasia after H. pylori infection. On the other hand, we demonstrated that ASK1 and downstream MAPKs are activated in gastric stem/progenitor cells and important for their proliferation. In infected ASK1−/− mice, a variety of inflammatory cells were involved and TH1-immune responses were greatly enhanced. Further, Gr1+Cd11b+ MDSCs were recruited to ASK1−/− stomach and spleen. We found that ASK1-p38 pathway in myeloid cells was important for macrophage cell death and following IL-1β secretion. These exaggerated immune responses activated NF-κB and STAT1 signaling in the epithelium, and caused dramatic expansion of facultative gastric progenitor cells.
Our bone marrow transplantation results found that epithelial ASK1 was important for isthmus progenitor proliferation, but dispensable for SPEM formation. We showed that H. pylori activates ASK1 and MAPK pathway in part through production of reactive oxygen species (ROS) in gastric epithelial cells [70]. Since ROS production by H. pylori appears to be CagA-dependent as shown in previous studies [71,72], CagA translocation may contribute to ASK1 activation in isthmus progenitor cells. Nevertheless, it remains undetermined how and where metaplastic cells come from after H. pylori infection. In mouse models, several reports have shown that SPEM may be transdifferentiated from mature chief cells [5,73], while we and others reported that metaplasia arises from isthmus stem/progenitor cells [7,10,52,74,75,76]. Our current data suggest that while the strong MAPK activation in the isthmus and pit cells regulates their proliferation, development of SPEM, in which MAPK activity is absent, may not be directly affected by MAPK activation, but depends more on the degree of inflammation.
For stem cell maintenance, Wnt signaling activation is thought to be most critical. In the mouse corpus, Wnt5a-dependent noncanonical Wnt signaling appears to be involved in stem cell function [10,14]. While other pathways such as gastrin, acetylcholine, R-spondin, BMP signaling likely contribute to gastric stem cell niche [11,77,78,79,80,81,82,83], accumulating evidence highlighted the importance of MAPK pathway in gut stem cells. For example, it has been reported that JNK is one of Wnt activators in the intestine and thus promotes epithelial proliferation and carcinogenesis [84]. Gastrointestinal organoid culture often requires pharmacological inhibition of p38MAPK [85], and p38MAPK activation is reported to be involved in gastrointestinal stem cell aging [86]. Interaction between ASK1 and Notch signaling [87], which is known to expand gastric stem cell differentiation [88], was also reported. Thus, ASK1 and other MAPK pathways could be functionally important in gastric stem/progenitor cells. Further analysis would be required for exploring the detailed mechanism in which ASK1-MAPK pathway regulates stem cell functions.
We previously reported that ASK1–JNK pathway plays a critical role on gastric carcinogenesis by controlling epithelial cell proliferation [35,89]. In these studies, we used MNU-induced chemically induced gastric tumor model. Given that most of MNU-induced tumors arise from the mouse antrum or pylorus, the mechanism of MNU-tumor development must be different from the way of corpus metaplasia or tumor. Indeed, corpus glands are in many aspects distinct from antral glands, with distinguished gland structure containing different stem/progenitor cells as well as different responses to external stimulants including carcinogens and gastrin hormones [12,13,33,50,74,90,91]. Nevertheless, MAPK activation can be found both in the antrum and corpus, in particular in the area between the isthmus and the pit cells. Thus, MAPK activation in pit cells and isthmus progenitors is important for cell proliferation in both regions, and that MNU-induced tumor, whose development is suppressed by ASK1 deficiency, would be most likely derived from these cell zone.
Our present study revealed that ASK1 deficiency in myeloid lineage caused macrophage apoptosis and IL-1β secretion, which is consistent with our previous study of colitis model [37]. IL-1 signaling has been reported to play an important role on gastric carcinogenesis [18,19,20,27,58]. While it remains possible that IL-1β can directly activate epithelial cell population, our current data and past studies suggest that IL-1β is a key mediator for broad immune reaction. Indeed, we demonstrated that innate immune disorder induced by IL-1β release triggered adaptive immune responses including IFN-γ and FasL production, and also initiated the recruitment of Gr-1+CD11b+ MDSCs. Several previous papers have shown that MDSCs could be engaged by abnormal cytokine production such as IL-1β, TNF-ɑ, and IFN-γ [18,92,93,94]. However, it remains unclear whether MDSCs are induced by higher levels of IL-1β or IFN-γ in ASK1−/− mice, or ASK1 deficiency causes the impairment of myeloid cell maturation. Future investigation would be needed concerning the relationship between ASK1 and myeloid cell maturation.
Stem cell niche contributes to stem cell maintenance as a source of growth factors and cytokines. Abnormal immune cells and excessive levels of cytokines around gastric stem cells could change their survival and functions. Thus, we here raise the conclusion that disrupted immune responses in ASK1−/− mice including innate immunity, adaptive immunity, and MDSCs, alter the gastric stem cell niche, which eventually cause progenitor cell expansion and metaplastic changes.
Acknowledgments
We thank Hidenori Ichijo and Kosuke Takeda for providing ASK1−/− and ASK2−/− mice.
Supplementary Materials
The following are available online at https://www.mdpi.com/2076-2607/8/12/1995/s1, Supplementary Figure S1. PMSS-1 infection experiments. (A) Representative H&E staining of WT and ASK1−/− mouse stomach with H. pylori PMSS-1 infection. Original magnification, ×100. (B) Histological scoring of PMSS-1-infected WT and ASK1−/− mouse stomach. Inf., inflammation; atr., atrophy; met., metaplasia; hyp., hyperplasia. Data are shown as mean ± SD. * p < 0.05 compared to infected WT mice. (C) Colony-forming units of H. pylori isolated from the infected WT and ASK1−/− mouse stomach. Supplementary Figure S2. ASK1, not ASK2, is responsible for downstream MAPK activation in the stomach. (A) Immunoblotting of WT and ASK2−/− mouse stomach and spleen infected with SS-1. (B) Total p38 staining in SS-1-infected WT and ASK1−/− stomach. Original magnification, ×200. Supplementary Figure S3. ASK1 deficiency induced NF-κB and STAT1 activation in the PMSS-1 infected stomach. Immunoblotting of PMSS-1-infected WT and ASK1−/− mouse stomachs. Supplementary Figure S4. ASK1 deficiency did not promote apoptosis in T cells and neutrophils. Immunofluorescent staining combined with TUNEL assay in SS-1-infected WT and ASK1−/− stomach and spleen. Original magnification, ×200.
Author Contributions
Conceptualization, Y.H. (Yoku Hayakawa), Y.H. (Yoshihiro Hirata), and S.M.; investigation, Y.H. (Yoku Hayakawa), Y.H. (Yoshihiro Hirata), M.H., M.T., Y.O., K.K. (Ken Kurokawa), S.A., J.A., N.S., H.N., and H.F.; resources, H.F.; writing—original draft preparation, Y.H. (Yoku Hayakawa) and Y.H. (Yoshihiro Hirata); supervision, K.T., S.M., and K.K. (Kazuhiko Koike); funding acquisition, Y.H. (Yoku Hayakawa) and Y.H. (Yoshihiro Hirata). All authors have read and agreed to the published version of the manuscript.
Funding
Y.H. (Yoku Hayakawa) is supported by the KAKENHI Grant-in-Aid for Scientific Research, 20H03656, 20K08323, 20K08375, P-CREATE from AMED, the Inoue Science Research Award, the Takeda Science Foundation Visionary Research Grant, the Uehara Memorial Foundation, the Naito Foundation, and the Advanced Research and Development Programs for Medical Innovation (PRIME). Y.H. (Yoshihiro Hirata) is supported by the KAKENHI Grant-in Aid for Scientific Research, 18KT0067 and 20K08377, and a grant from Manpei Suzuki Diabetes Foundation.
Conflicts of Interest
The authors declare no conflict of interest.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Parsonnet J., Friedman G.D., Vandersteen D.P., Chang Y., Vogelman J.H., Orentreich N., Sibley R.K. Helicobacter pylori infection and the risk of gastric carcinoma. N. Engl. J. Med. 1991;325:1127–1131. doi: 10.1056/NEJM199110173251603. [DOI] [PubMed] [Google Scholar]
- 2.Marshall B.J., Warren J.R. Unidentified curved bacilli in the stomach of patients with gastritis and peptic ulceration. Lancet. 1984;1:1311–1315. doi: 10.1016/S0140-6736(84)91816-6. [DOI] [PubMed] [Google Scholar]
- 3.Correa P. Human gastric carcinogenesis: A multistep and multifactorial process--first american cancer society award lecture on cancer epidemiology and prevention. Cancer Res. 1992;52:6735–6740. [PubMed] [Google Scholar]
- 4.Canfield V., West A.B., Goldenring J.R., Levenson R. Genetic ablation of parietal cells in transgenic mice: A new model for analyzing cell lineage relationships in the gastric mucosa. Proc. Natl. Acad. Sci. USA. 1996;93:2431–2435. doi: 10.1073/pnas.93.6.2431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Nam K.T., Lee H.J., Sousa J.F., Weis V.G., O’Neal R.L., Finke P.E., Romero-Gallo J., Shi G., Mills J.C., Peek R.M., et al. Mature chief cells are cryptic progenitors for metaplasia in the stomach. Gastroenterology. 2010;139:2028–2037.e2029. doi: 10.1053/j.gastro.2010.09.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Han S., Fink J., Jorg D.J., Lee E., Yum M.K., Chatzeli L., Merker S.R., Josserand M., Trendafilova T., Andersson-Rolf A., et al. Defining the identity and dynamics of adult gastric isthmus stem cells. Cell Stem Cell. 2019;25:342–356. doi: 10.1016/j.stem.2019.07.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hata M., Kinoshita H., Hayakawa Y., Konishi M., Tsuboi M., Oya Y., Kurokawa K., Hayata Y., Nakagawa H., Tateishi K., et al. Gpr30-expressing gastric chief cells do not dedifferentiate but are eliminated via pdk-dependent cell competition during development of metaplasia. Gastroenterology. 2020;158:1650–1666.e1615. doi: 10.1053/j.gastro.2020.01.046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Barker N., Huch M., Kujala P., van de Wetering M., Snippert H.J., van Es J.H., Sato T., Stange D.E., Begthel H., van den Born M., et al. Lgr5(+ve) stem cells drive self-renewal in the stomach and build long-lived gastric units in vitro. Cell Stem Cell. 2010;6:25–36. doi: 10.1016/j.stem.2009.11.013. [DOI] [PubMed] [Google Scholar]
- 9.Arnold K., Sarkar A., Yram M.A., Polo J.M., Bronson R., Sengupta S., Seandel M., Geijsen N., Hochedlinger K. Sox2(+) adult stem and progenitor cells are important for tissue regeneration and survival of mice. Cell Stem Cell. 2011;9:317–329. doi: 10.1016/j.stem.2011.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hayakawa Y., Ariyama H., Stancikova J., Sakitani K., Asfaha S., Renz B.W., Dubeykovskaya Z.A., Shibata W., Wang H., Westphalen C.B., et al. Mist1 expressing gastric stem cells maintain the normal and neoplastic gastric epithelium and are supported by a perivascular stem cell niche. Cancer Cell. 2015;28:800–814. doi: 10.1016/j.ccell.2015.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Sheng W., Malagola E., Nienhuser H., Zhang Z., Kim W., Zamechek L., Sepulveda A., Hata M., Hayakawa Y., Zhao C.M., et al. Hypergastrinemia expands gastric ecl cells through cck2r+ progenitor cells via erk activation. Cell. Mol. Gastroenterol. Hepatol. 2020;10:434–449.e1. doi: 10.1016/j.jcmgh.2020.04.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lee Y., Urbanska A.M., Hayakawa Y., Wang H., Au A.S., Luna A.M., Chang W., Jin G., Bhagat G., Abrams J.A., et al. Gastrin stimulates a cholecystokinin-2-receptor-expressing cardia progenitor cell and promotes progression of barrett’s-like esophagus. Oncotarget. 2016;8:203. doi: 10.18632/oncotarget.10667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Chang W., Wang H., Kim W., Liu Y., Deng H., Liu H., Jiang Z., Niu Z., Sheng W., Napoles O.C., et al. Hormonal suppression of stem cells inhibits symmetric cell division and gastric tumorigenesis. Cell Stem Cell. 2020;26:739–754.e738. doi: 10.1016/j.stem.2020.01.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Nienhuser H., Kim W., Malagola E., Ruan T., Valenti G., Middelhoff M., Bass A., Der C.J., Hayakawa Y., Wang T.C. Mist1+ gastric isthmus stem cells are regulated by wnt5a and expand in response to injury and inflammation in mice. Gut. 2020 doi: 10.1136/gutjnl-2020-320742. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Crabtree J.E., Shallcross T.M., Heatley R.V., Wyatt J.I. Mucosal tumour necrosis factor alpha and interleukin-6 in patients with helicobacter pylori associated gastritis. Gut. 1991;32:1473–1477. doi: 10.1136/gut.32.12.1473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Peek R.M., Jr., Miller G.G., Tham K.T., Perez-Perez G.I., Zhao X., Atherton J.C., Blaser M.J. Heightened inflammatory response and cytokine expression in vivo to caga+ helicobacter pylori strains. Lab. Invest. 1995;73:760–770. [PubMed] [Google Scholar]
- 17.Fox J.G., Sheppard B.J., Dangler C.A., Whary M.T., Ihrig M., Wang T.C. Germ-line p53-targeted disruption inhibits helicobacter-induced premalignant lesions and invasive gastric carcinoma through down-regulation of th1 proinflammatory responses. Cancer Res. 2002;62:696–702. [PubMed] [Google Scholar]
- 18.Tu S., Bhagat G., Cui G., Takaishi S., Kurt-Jones E.A., Rickman B., Betz K.S., Penz-Oesterreicher M., Bjorkdahl O., Fox J.G., et al. Overexpression of interleukin-1beta induces gastric inflammation and cancer and mobilizes myeloid-derived suppressor cells in mice. Cancer Cell. 2008;14:408–419. doi: 10.1016/j.ccr.2008.10.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Serizawa T., Hirata Y., Hayakawa Y., Suzuki N., Sakitani K., Hikiba Y., Ihara S., Kinoshita H., Nakagawa H., Tateishi K., et al. Gastric metaplasia induced by helicobacter pylori is associated with enhanced sox9 expression via interleukin-1 signaling. Infect. Immun. 2015;84:562–572. doi: 10.1128/IAI.01437-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kinoshita H., Hirata Y., Nakagawa H., Sakamoto K., Hayakawa Y., Takahashi R., Nakata W., Sakitani K., Serizawa T., Hikiba Y., et al. Interleukin-6 mediates epithelial-stromal interactions and promotes gastric tumorigenesis. PLoS ONE. 2013;8:e60914. doi: 10.1371/journal.pone.0060914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Eaton K.A., Mefford M., Thevenot T. The role of t cell subsets and cytokines in the pathogenesis of helicobacter pylori gastritis in mice. J. Immunol. 2001;166:7456–7461. doi: 10.4049/jimmunol.166.12.7456. [DOI] [PubMed] [Google Scholar]
- 22.Akhiani A.A., Pappo J., Kabok Z., Schon K., Gao W., Franzen L.E., Lycke N. Protection against helicobacter pylori infection following immunization is il-12-dependent and mediated by th1 cells. J. Immunol. 2002;169:6977–6984. doi: 10.4049/jimmunol.169.12.6977. [DOI] [PubMed] [Google Scholar]
- 23.Sayi A., Kohler E., Hitzler I., Arnold I., Schwendener R., Rehrauer H., Müller A. The cd4+ t cell-mediated ifn-gamma response to helicobacter infection is essential for clearance and determines gastric cancer risk. J. Immunol. 2009;182:7085–7101. doi: 10.4049/jimmunol.0803293. [DOI] [PubMed] [Google Scholar]
- 24.Kurt-Jones E.A., Cao L., Sandor F., Rogers A.B., Whary M.T., Nambiar P.R., Cerny A., Bowen G., Yan J., Takaishi S., et al. Trefoil family factor 2 is expressed in murine gastric and immune cells and controls both gastrointestinal inflammation and systemic immune responses. Infect. Immun. 2007;75:471–480. doi: 10.1128/IAI.02039-05. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Quante M., Tu S.P., Tomita H., Gonda T., Wang S.S., Takashi S., Baik G.H., Shibata W., Diprete B., Betz K.S., et al. Bone marrow-derived myofibroblasts contribute to the mesenchymal stem cell niche and promote tumor growth. Cancer Cell. 2011;19:257–272. doi: 10.1016/j.ccr.2011.01.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hitzler I., Sayi A., Kohler E., Engler D.B., Koch K.N., Hardt W.D., Muller A. Caspase-1 has both proinflammatory and regulatory properties in helicobacter infections, which are differentially mediated by its substrates il-1beta and il-18. J. Immunol. 2012;188:3594–3602. doi: 10.4049/jimmunol.1103212. [DOI] [PubMed] [Google Scholar]
- 27.El-Omar E.M., Carrington M., Chow W.H., McColl K.E., Bream J.H., Young H.A., Herrera J., Lissowska J., Yuan C.C., Rothman N., et al. The role of interleukin-1 polymorphisms in the pathogenesis of gastric cancer. Nature. 2001;412:99. doi: 10.1038/35083631. [DOI] [PubMed] [Google Scholar]
- 28.El-Omar E.M., Rabkin C.S., Gammon M.D., Vaughan T.L., Risch H.A., Schoenberg J.B., Stanford J.L., Mayne S.T., Goedert J., Blot W.J., et al. Increased risk of noncardia gastric cancer associated with proinflammatory cytokine gene polymorphisms. Gastroenterology. 2003;124:1193–1201. doi: 10.1016/S0016-5085(03)00157-4. [DOI] [PubMed] [Google Scholar]
- 29.Maeda S., Yoshida H., Ogura K., Mitsuno Y., Hirata Y., Yamaji Y., Akanuma M., Shiratori Y., Omata M.H. Pylori activates nf-kappab through a signaling pathway involving ikappab kinases, nf-kappab-inducing kinase, traf2, and traf6 in gastric cancer cells. Gastroenterology. 2000;119:97–108. doi: 10.1053/gast.2000.8540. [DOI] [PubMed] [Google Scholar]
- 30.Mitsuno Y., Yoshida H., Maeda S., Ogura K., Hirata Y., Kawabe T., Shiratori Y., Omata M. Helicobacter pylori induced transactivation of sre and ap-1 through the erk signalling pathway in gastric cancer cells. Gut. 2001;49:18–22. doi: 10.1136/gut.49.1.18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Higashi H., Tsutsumi R., Muto S., Sugiyama T., Azuma T., Asaka M., Hatakeyama M. Shp-2 tyrosine phosphatase as an intracellular target of helicobacter pylori caga protein. Science. 2002;295:683–686. doi: 10.1126/science.1067147. [DOI] [PubMed] [Google Scholar]
- 32.Hirata Y., Ohmae T., Shibata W., Maeda S., Ogura K., Yoshida H., Kawabe T., Omata M. Myd88 and tnf receptor-associated factor 6 are critical signal transducers in helicobacter pylori-infected human epithelial cells. J. Immunol. 2006;176:3796–3803. doi: 10.4049/jimmunol.176.6.3796. [DOI] [PubMed] [Google Scholar]
- 33.Kinoshita H., Hayakawa Y., Koike K. Metaplasia in the stomach-precursor of gastric cancer? Int. J. Mol. Sci. 2017;18:2063. doi: 10.3390/ijms18102063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ichijo H., Nishida E., Irie K., ten Dijke P., Saitoh M., Moriguchi T., Takagi M., Matsumoto K., Miyazono K., Gotoh Y. Induction of apoptosis by ask1, a mammalian mapkkk that activates sapk/jnk and p38 signaling pathways. Science. 1997;275:90–94. doi: 10.1126/science.275.5296.90. [DOI] [PubMed] [Google Scholar]
- 35.Hayakawa Y., Hirata Y., Nakagawa H., Sakamoto K., Hikiba Y., Kinoshita H., Nakata W., Takahashi R., Tateishi K., Tada M., et al. Apoptosis signal-regulating kinase 1 and cyclin d1 compose a positive feedback loop contributing to tumor growth in gastric cancer. Proc. Natl. Acad. Sci. USA. 2011;108:780–785. doi: 10.1073/pnas.1011418108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Hayakawa Y., Hirata Y., Sakitani K., Nakagawa H., Nakata W., Kinoshita H., Takahashi R., Takeda K., Ichijo H., Maeda S., et al. Apoptosis signal-regulating kinase-1 inhibitor as a potent therapeutic drug for the treatment of gastric cancer. Cancer Sci. 2012;103:2181–2185. doi: 10.1111/cas.12024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Hayakawa Y., Hirata Y., Nakagawa H., Sakamoto K., Hikiba Y., Otsuka M., Ijichi H., Ikenoue T., Tateishi K., Akanuma M., et al. Apoptosis signal-regulating kinase 1 regulates colitis and colitis-associated tumorigenesis by the innate immune responses. Gastroenterology. 2010;138:1055–1067. doi: 10.1053/j.gastro.2009.11.015. [DOI] [PubMed] [Google Scholar]
- 38.Takahashi R., Hirata Y., Sakitani K., Nakata W., Kinoshita H., Hayakawa Y., Nakagawa H., Sakamoto K., Hikiba Y., Ijichi H., et al. Therapeutic effect of c-jun n-terminal kinase inhibition on pancreatic cancer. Cancer Sci. 2013;104:337–344. doi: 10.1111/cas.12080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Nakagawa H., Hirata Y., Takeda K., Hayakawa Y., Sato T., Kinoshita H., Sakamoto K., Nakata W., Hikiba Y., Omata M., et al. Apoptosis signal-regulating kinase 1 inhibits hepatocarcinogenesis by controlling the tumor-suppressing function of stress-activated mitogen-activated protein kinase. Hepatology. 2011;54:185–195. doi: 10.1002/hep.24357. [DOI] [PubMed] [Google Scholar]
- 40.Matsuzawa A., Saegusa K., Noguchi T., Sadamitsu C., Nishitoh H., Nagai S., Koyasu S., Matsumoto K., Takeda K., Ichijo H. Ros-dependent activation of the traf6-ask1-p38 pathway is selectively required for tlr4-mediated innate immunity. Nat. Immunol. 2005;6:587–592. doi: 10.1038/ni1200. [DOI] [PubMed] [Google Scholar]
- 41.Kundu M., Pathak S.K., Kumawat K., Basu S., Chatterjee G., Pathak S., Noguchi T., Takeda K., Ichijo H., Thien C.B., et al. A tnf- and c-cbl-dependent flip(s)-degradation pathway and its function in mycobacterium tuberculosis-induced macrophage apoptosis. Nat. Immunol. 2009;10:918–926. doi: 10.1038/ni.1754. [DOI] [PubMed] [Google Scholar]
- 42.Tobiume K., Matsuzawa A., Takahashi T., Nishitoh H., Morita K., Takeda K., Minowa O., Miyazono K., Noda T., Ichijo H. Ask1 is required for sustained activations of jnk/p38 map kinases and apoptosis. EMBO Rep. 2001;2:222–228. doi: 10.1093/embo-reports/kve046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Takeda K., Shimozono R., Noguchi T., Umeda T., Morimoto Y., Naguro I., Tobiume K., Saitoh M., Matsuzawa A., Ichijo H. Apoptosis signal-regulating kinase (ask) 2 functions as a mitogen-activated protein kinase kinase kinase in a heteromeric complex with ask1. J. Biol. Chem. 2007;282:7522–7531. doi: 10.1074/jbc.M607177200. [DOI] [PubMed] [Google Scholar]
- 44.Lee A., O’Rourke J., De Ungria M.C., Robertson B., Daskalopoulos G., Dixon M.F. A standardized mouse model of helicobacter pylori infection: Introducing the sydney strain. Gastroenterology. 1997;112:1386–1397. doi: 10.1016/S0016-5085(97)70155-0. [DOI] [PubMed] [Google Scholar]
- 45.Arnold I.C., Lee J.Y., Amieva M.R., Roers A., Flavell R.A., Sparwasser T., Muller A. Tolerance rather than immunity protects from helicobacter pylori-induced gastric preneoplasia. Gastroenterology. 2011;140:199–209. doi: 10.1053/j.gastro.2010.06.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Ogura K., Maeda S., Nakao M., Watanabe T., Tada M., Kyutoku T., Yoshida H., Shiratori Y., Omata M. Virulence factors of helicobacter pylori responsible for gastric diseases in mongolian gerbil. J. Exp. Med. 2000;192:1601–1610. doi: 10.1084/jem.192.11.1601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Hayakawa Y., Maeda S., Nakagawa H., Hikiba Y., Shibata W., Sakamoto K., Yanai A., Hirata Y., Ogura K., Muto S., et al. Effectiveness of ikappab kinase inhibitors in murine colitis-associated tumorigenesis. J. Gastroenterol. 2009;44:935–943. doi: 10.1007/s00535-009-0098-7. [DOI] [PubMed] [Google Scholar]
- 48.Crabtree J.E., Ferrero R.L., Kusters J.G. The mouse colonizing helicobacter pylori strain ss1 may lack a functional cag pathogenicity island. Helicobacter. 2002;7:139–140. doi: 10.1046/j.1083-4389.2002.00071.x. [DOI] [PubMed] [Google Scholar]
- 49.Goldenring J.R., Nomura S. Differentiation of the gastric mucosa iii. Animal models of oxyntic atrophy and metaplasia. Am. J. Physiol. Gastrointest. Liver Physiol. 2006;291:G999–G1004. doi: 10.1152/ajpgi.00187.2006. [DOI] [PubMed] [Google Scholar]
- 50.Hayakawa Y., Fox J., Gonda T., Worthley D., Muthupalani S., Wang T. Mouse models of gastric cancer. Cancers. 2013;5:92–130. doi: 10.3390/cancers5010092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Hayakawa Y., Fox J.G., Wang T.C. The origins of gastric cancer from gastric stem cells: Lessons from mouse models. Cell. Mol. Gastroenterol. Hepatol. 2017;3:331–338. doi: 10.1016/j.jcmgh.2017.01.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Hayakawa Y., Fox J.G., Wang T.C. Isthmus stem cells are the origins of metaplasia in the gastric corpus. Cell. Mol. Gastroenterol. Hepatol. 2017;4:89–94. doi: 10.1016/j.jcmgh.2017.02.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Derijard B., Hibi M., Wu I.H., Barrett T., Su B., Deng T., Karin M., Davis R.J. Jnk1: A protein kinase stimulated by uv light and ha-ras that binds and phosphorylates the c-jun activation domain. Cell. 1994;76:1025–1037. doi: 10.1016/0092-8674(94)90380-8. [DOI] [PubMed] [Google Scholar]
- 54.Park J.M., Greten F.R., Wong A., Westrick R.J., Arthur J.S., Otsu K., Hoffmann A., Montminy M., Karin M. Signaling pathways and genes that inhibit pathogen-induced macrophage apoptosis--creb and nf-kappab as key regulators. Immunity. 2005;23:319–329. doi: 10.1016/j.immuni.2005.08.010. [DOI] [PubMed] [Google Scholar]
- 55.Ashwell J.D. The many paths to p38 mitogen-activated protein kinase activation in the immune system. Nat. Rev. Immunol. 2006;6:532–540. doi: 10.1038/nri1865. [DOI] [PubMed] [Google Scholar]
- 56.Maeda S., Kamata H., Luo J.L., Leffert H., Karin M. Ikkbeta couples hepatocyte death to cytokine-driven compensatory proliferation that promotes chemical hepatocarcinogenesis. Cell. 2005;121:977–990. doi: 10.1016/j.cell.2005.04.014. [DOI] [PubMed] [Google Scholar]
- 57.Sengupta T.K., Talbot E.S., Scherle P.A., Ivashkiv L.B. Rapid inhibition of interleukin-6 signaling and stat3 activation mediated by mitogen-activated protein kinases. Proc. Natl. Acad. Sci. USA. 1998;95:11107–11112. doi: 10.1073/pnas.95.19.11107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Sakamoto K., Hikiba Y., Nakagawa H., Hayakawa Y., Yanai A., Akanuma M., Ogura K., Hirata Y., Kaestner K.H., Omata M., et al. Inhibitor of kappab kinase beta regulates gastric carcinogenesis via interleukin-1alpha expression. Gastroenterology. 2010;139:226–238.e226. doi: 10.1053/j.gastro.2010.03.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Nakata W., Hayakawa Y., Nakagawa H., Sakamoto K., Kinoshita H., Takahashi R., Hirata Y., Maeda S., Koike K. Anti-tumor activity of the proteasome inhibitor bortezomib in gastric cancer. Int. J. Oncol. 2011;39:1529–1536. doi: 10.1016/S0016-5085(11)62794-7. [DOI] [PubMed] [Google Scholar]
- 60.Berg D.J., Lynch N.A., Lynch R.G., Lauricella D.M. Rapid development of severe hyperplastic gastritis with gastric epithelial dedifferentiation in helicobacter felis-infected il-10(-/-) mice. Am. J. Pathol. 1998;152:1377–1386. [PMC free article] [PubMed] [Google Scholar]
- 61.Boselli D., Losana G., Bernabei P., Bosisio D., Drysdale P., Kiessling R., Gaston J.S., Lammas D., Casanova J.L., Kumararatne D.S., et al. Ifn-gamma regulates fas ligand expression in human cd4+ t lymphocytes and controls their anti-mycobacterial cytotoxic functions. Eur. J. Immunol. 2007;37:2196–2204. doi: 10.1002/eji.200636541. [DOI] [PubMed] [Google Scholar]
- 62.Cai X., Stoicov C., Li H., Carlson J., Whary M., Fox J.G., Houghton J. Overcoming fas-mediated apoptosis accelerates helicobacter-induced gastric cancer in mice. Cancer Res. 2005;65:10912–10920. doi: 10.1158/0008-5472.CAN-05-1802. [DOI] [PubMed] [Google Scholar]
- 63.Rudi J., Kuck D., Strand S., von Herbay A., Mariani S.M., Krammer P.H., Galle P.R., Stremmel W. Involvement of the cd95 (apo-1/fas) receptor and ligand system in helicobacter pylori-induced gastric epithelial apoptosis. J. Clin. Invest. 1998;102:1506–1514. doi: 10.1172/JCI2808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Yang X.D., Ai W., Asfaha S., Bhagat G., Friedman R.A., Jin G., Park H., Shykind B., Diacovo T.G., Falus A., et al. Histamine deficiency promotes inflammation-associated carcinogenesis through reduced myeloid maturation and accumulation of cd11b+ly6g+ immature myeloid cells. Nat. Med. 2011;17:87–95. doi: 10.1038/nm.2278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Dubeykovskaya Z., Si Y., Chen X., Worthley D.L., Renz B.W., Urbanska A.M., Hayakawa Y., Xu T., Westphalen C.B., Dubeykovskiy A., et al. Neural innervation stimulates splenic tff2 to arrest myeloid cell expansion and cancer. Nat. Commun. 2016;7:10517. doi: 10.1038/ncomms10517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Greten F.R., Arkan M.C., Bollrath J., Hsu L.C., Goode J., Miething C., Goktuna S.I., Neuenhahn M., Fierer J., Paxian S., et al. Nf-kappab is a negative regulator of il-1beta secretion as revealed by genetic and pharmacological inhibition of ikkbeta. Cell. 2007;130:918–931. doi: 10.1016/j.cell.2007.07.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Okumura T., Ericksen R.E., Takaishi S., Wang S.S., Dubeykovskiy Z., Shibata W., Betz K.S., Muthupalani S., Rogers A.B., Fox J.G., et al. K-ras mutation targeted to gastric tissue progenitor cells results in chronic inflammation, an altered microenvironment, and progression to intraepithelial neoplasia. Cancer Res. 2010;70:8435–8445. doi: 10.1158/0008-5472.CAN-10-1506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Nakanishi Y., Seno H., Fukuoka A., Ueo T., Yamaga Y., Maruno T., Nakanishi N., Kanda K., Komekado H., Kawada M., et al. Dclk1 distinguishes between tumor and normal stem cells in the intestine. Nat. Genet. 2013;45:98–103. doi: 10.1038/ng.2481. [DOI] [PubMed] [Google Scholar]
- 69.Westphalen C.B., Asfaha S., Hayakawa Y., Takemoto Y., Lukin D.J., Nuber A.H., Brandtner A., Setlik W., Remotti H., Muley A., et al. Long-lived intestinal tuft cells serve as colon cancer-initiating cells. J. Clin. Investig. 2014;124:1283–1295. doi: 10.1172/JCI73434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Hayakawa Y., Hirata Y., Kinoshita H., Sakitani K., Nakagawa H., Nakata W., Takahashi R., Sakamoto K., Maeda S., Koike K. Differential roles of ask1 and tak1 in helicobacter pylori-induced cellular responses. Infect. Immun. 2013;81:4551–4560. doi: 10.1128/IAI.00914-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Chaturvedi R., Asim M., Romero-Gallo J., Barry D.P., Hoge S., de Sablet T., Delgado A.G., Wroblewski L.E., Piazuelo M.B., Yan F., et al. Spermine oxidase mediates the gastric cancer risk associated with helicobacter pylori caga. Gastroenterology. 2011;141:1696–1708. doi: 10.1053/j.gastro.2011.07.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Hanada K., Uchida T., Tsukamoto Y., Watada M., Yamaguchi N., Yamamoto K., Shiota S., Moriyama M., Graham D.Y., Yamaoka Y. Helicobacter pylori infection introduces DNA double-strand breaks in host cells. Infect. Immun. 2014;82:4182–4189. doi: 10.1128/IAI.02368-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Nam K.T., O’Neal R.L., Coffey R.J., Finke P.E., Barker N., Goldenring J.R. Spasmolytic polypeptide-expressing metaplasia (spem) in the gastric oxyntic mucosa does not arise from lgr5-expressing cells. Gut. 2012;61:1678–1685. doi: 10.1136/gutjnl-2011-301193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Hata M., Hayakawa Y., Koike K. Gastric stem cell and cellular origin of cancer. Biomedicines. 2018;6:100. doi: 10.3390/biomedicines6040100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Kinoshita H., Hayakawa Y., Niu Z., Konishi M., Hata M., Tsuboi M., Hayata Y., Hikiba Y., Ihara S., Nakagawa H., et al. Mature gastric chief cells are not required for the development of metaplasia. Am. J. Physiol. Gastrointest. Liver Physiol. 2018;314:G583–G596. doi: 10.1152/ajpgi.00351.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Yoshioka T., Fukuda A., Araki O., Ogawa S., Hanyu Y., Matsumoto Y., Yamaga Y., Nakanishi Y., Kawada K., Sakai Y., et al. Bmi1 marks gastric stem cells located in the isthmus in mice. J. Pathol. 2019;248:179–190. doi: 10.1002/path.5244. [DOI] [PubMed] [Google Scholar]
- 77.Worthley D.L., Churchill M., Compton J.T., Tailor Y., Rao M., Si Y., Levin D., Schwartz M.G., Uygur A., Hayakawa Y., et al. Gremlin 1 identifies a skeletal stem cell with bone, cartilage, and reticular stromal potential. Cell. 2015;160:269–284. doi: 10.1016/j.cell.2014.11.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Hayakawa Y., Chang W., Jin G., Wang T.C. Gastrin and upper gi cancers. Curr. Opin. Pharmacol. 2016;31:31–37. doi: 10.1016/j.coph.2016.08.013. [DOI] [PubMed] [Google Scholar]
- 79.Sigal M., Logan C.Y., Kapalczynska M., Mollenkopf H.J., Berger H., Wiedenmann B., Nusse R., Amieva M.R., Meyer T.F. Stromal r-spondin orchestrates gastric epithelial stem cells and gland homeostasis. Nature. 2017;548:451–455. doi: 10.1038/nature23642. [DOI] [PubMed] [Google Scholar]
- 80.Zhao C.M., Hayakawa Y., Kodama Y., Muthupalani S., Westphalen C.B., Andersen G.T., Flatberg A., Johannessen H., Friedman R.A., Renz B.W., et al. Denervation suppresses gastric tumorigenesis. Sci. Transl. Med. 2014;6:250ra115. doi: 10.1126/scitranslmed.3009569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Rabben H.L., Zhao C.M., Hayakawa Y., Wang T.C., Chen D. Vagotomy and gastric tumorigenesis. Curr. Neuropharmacol. 2016;14:967–972. doi: 10.2174/1570159X14666160121114854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Hayakawa Y., Sakitani K., Konishi M., Asfaha S., Niikura R., Tomita H., Renz B.W., Tailor Y., Macchini M., Middelhoff M., et al. Nerve growth factor promotes gastric tumorigenesis through aberrant cholinergic signaling. Cancer Cell. 2017;31:21–34. doi: 10.1016/j.ccell.2016.11.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Konishi M., Hayakawa Y., Koike K. Role of muscarinic acetylcholine signaling in gastrointestinal cancers. Biomedicines. 2019;7:58. doi: 10.3390/biomedicines7030058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Sancho R., Nateri A.S., de Vinuesa A.G., Aguilera C., Nye E., Spencer-Dene B., Behrens A. Jnk signalling modulates intestinal homeostasis and tumourigenesis in mice. EMBO J. 2009;28:1843–1854. doi: 10.1038/emboj.2009.153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Sato T., Stange D.E., Ferrante M., Vries R.G., Van Es J.H., Van den Brink S., Van Houdt W.J., Pronk A., Van Gorp J., Siersema P.D., et al. Long-term expansion of epithelial organoids from human colon, adenoma, adenocarcinoma, and barrett’s epithelium. Gastroenterology. 2011;141:1762–1772. doi: 10.1053/j.gastro.2011.07.050. [DOI] [PubMed] [Google Scholar]
- 86.He D., Wu H., Xiang J., Ruan X., Peng P., Ruan Y., Chen Y.G., Wang Y., Yu Q., Zhang H., et al. Gut stem cell aging is driven by mtorc1 via a p38 mapk-p53 pathway. Nat. Commun. 2020;11:37. doi: 10.1038/s41467-019-13911-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Mo J.S., Yoon J.H., Ann E.J., Ahn J.S., Baek H.J., Lee H.J., Kim S.H., Kim Y.D., Kim M.Y., Park H.S. Notch1 modulates oxidative stress induced cell death through suppression of apoptosis signal-regulating kinase 1. Proc. Natl. Acad. Sci. USA. 2013;110:6865–6870. doi: 10.1073/pnas.1209078110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Kim T.H., Shivdasani R.A. Notch signaling in stomach epithelial stem cell homeostasis. J. Exp. Med. 2011;208:677–688. doi: 10.1084/jem.20101737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Shibata W., Maeda S., Hikiba Y., Yanai A., Sakamoto K., Nakagawa H., Ogura K., Karin M., Omata M. C-jun nh2-terminal kinase 1 is a critical regulator for the development of gastric cancer in mice. Cancer Res. 2008;68:5031–5039. doi: 10.1158/0008-5472.CAN-07-6332. [DOI] [PubMed] [Google Scholar]
- 90.Sakitani K., Hayakawa Y., Deng H., Ariyama H., Kinoshita H., Konishi M., Ono S., Suzuki N., Ihara S., Niu Z., et al. Cxcr4-expressing mist1(+) progenitors in the gastric antrum contribute to gastric cancer development. Oncotarget. 2017;8:111012–111025. doi: 10.18632/oncotarget.22451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Hayakawa Y., Jin G., Wang H., Chen X., Westphalen C.B., Asfaha S., Renz B.W., Ariyama H., Dubeykovskaya Z.A., Takemoto Y., et al. Cck2r identifies and regulates gastric antral stem cell states and carcinogenesis. Gut. 2015;64:544–553. doi: 10.1136/gutjnl-2014-307190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Sade-Feldman M., Kanterman J., Ish-Shalom E., Elnekave M., Horwitz E., Baniyash M. Tumor necrosis factor-alpha blocks differentiation and enhances suppressive activity of immature myeloid cells during chronic inflammation. Immunity. 2013;38:541–554. doi: 10.1016/j.immuni.2013.02.007. [DOI] [PubMed] [Google Scholar]
- 93.Gallina G., Dolcetti L., Serafini P., De Santo C., Marigo I., Colombo M.P., Basso G., Brombacher F., Borrello I., Zanovello P., et al. Tumors induce a subset of inflammatory monocytes with immunosuppressive activity on cd8+ t cells. J. Clin. Investig. 2006;116:2777–2790. doi: 10.1172/JCI28828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Oya Y., Hayakawa Y., Koike K. Tumor microenvironment in gastric cancers. Cancer Sci. 2020;111:2696. doi: 10.1111/cas.14521. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.