Highlights
-
•
COVID-19 can have CNS and PNS neurological manifestations.
-
•
Headache and hyposmia are common neurological symptoms.
-
•
COVID-19 could trigger cytokine storm.
-
•
COVID-19 neurological sequalae may be due to viral neuroinvasion and cytokine storm.
-
•
Cytokine storm-targeting therapies may help manage COVID-19 neurological symptoms.
Keywords: Cytokine storm, COVID-19, Neurological complications, Coronavirus, Hypercytokinemia, Encephalopathy, Interferons, Cerebrovascular disease, Skeletal muscle injury, Guillain-Barré Syndrome
Abstract
The new coronavirus disease 2019 (COVID-19), caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), can trigger a hyperinflammatory state characterized by elevated cytokine levels known as hypercytokinemia or cytokine storm, observed most often in severe patients. Though COVID-19 is known to be a primarily respiratory disease, neurological complications affecting both the central and peripheral nervous systems have also been reported. This review discusses potential routes of SARS-CoV-2 neuroinvasion and pathogenesis, summarizes reported neurological sequelae of COVID-19, and examines how aberrant cytokine levels may precipitate these complications. Clarification of the pathogenic mechanisms of SARS-CoV-2 is needed to encourage prompt diagnosis and optimized care. In particular, identifying the presence of cytokine storm in patients with neurological COVID-19 manifestations will facilitate avenues for treatment. Future investigations into aberrant cytokine levels in COVID-19 patients with neurological symptoms as well as the efficacy of cytokine storm-targeting treatments will be critical in elucidating the pathogenic mechanisms and effective treatments of COVID-19.
1. Introduction
In recent years, respiratory coronaviruses belonging to the genus beta coronavirus have given rise to the Severe Acute Respiratory Syndrome (SARS) and Middle East Respiratory Syndrome (MERS), pandemics that emerged in 2002 and 2012, respectively. In December 2019, COVID-19 emerged in Wuhan, China, caused by the coronavirus SARS-CoV-2. Declared a pandemic by the World Health Organization as of March 11th, 2020, more than 25 million cases have been reported as of September 2nd, 2020 with over 859,000 deaths.
COVID-19 commonly manifests as fever, cough, and shortness of breath. Other complaints include lethargy, myalgia, confusion, headache, sore throat, rhinorrhea, chest pain, and vomiting [1]. Patients typically present with bilateral pneumonia with abnormal chest CT findings of bilateral peripheral ground glass opacities and subsegmental consolidation. Complications include acute respiratory distress syndrome (ARDS), sepsis, cardiac injury, and secondary infections. Although COVID-19 is primarily a respiratory disease, there is mounting evidence of neurological manifestations associated with severe prognosis [2], [3], involving both the central (CNS) and peripheral nervous system (PNS). Neuropathogenesis of SARS-CoV-2 remains unclear, which may be partially attributable to cytokine storm.
2. Pathogenesis of SARS-CoV-2 Infection
-
A.
Viral invasion and cytokine storm
SARS-CoV-2 is mainly transmitted by person to person through respiratory droplets generated by breathing, sneezing, coughing, etc., as well as contact (direct or indirect). The SARS-CoV-2 gets into host cells through the virus’s spike (S) glycoprotein (S protein). S protein binds to the angiotensin-converting enzyme 2 (ACE2) receptors on cell surface to gain entry into the cells. ACE2 receptors have been found in alveolar epithelial cells as well as in cells of the heart, brain, lungs, oral mucosa, intestines, and kidneys [4].
It has been proposed that SARS-CoV-2 colonizes the upper respiratory tract before spreading to the lower respiratory tract [5]. The virus can spread and replicate in tissues, subsequently triggering immune dysfunction and inflammation. Upon viral invasion, infected respiratory mucosal cells secrete type I interferons (IFN) like IFN-α and IFN-β, which induce resistance of uninfected cells to viral replication and activate dendritic cells and macrophages, promoting several pro-inflammatory cytokines, and eventually activate natural killer cells to kill the virus-infected cells via secretion of type II interferon, IFN-γ. Deficient type I IFN production, possibly from causes like Vitamin C insufficiency, may lead to continued pathogenesis of the influenza virus and systemic spread [6]. Similar deficiencies in COVID-19 patients may allow SARS-CoV-2 to spread from the upper respiratory tract to the lower respiratory tract and systemically.
Cytokine storm is a hyperinflammatory, pathological state that results from a sudden increase in certain circulating pro-inflammatory cytokine levels, which leads to overwhelming systemic inflammation, exacerbating viral pathogenesis and causing sepsis, ARDS, and multi-organ failure [7], [8], [9], [10], [11]. The cytokine storm has also been observed in SARS, MERS, H5N1 influenza, and H7N9 influenza, and with other respiratory viruses [12]. SARS-CoV-2 cytokine profile resembles that of secondary hemophagocytic syndrome, or hemophagocytic lymphohistiocytosis (HLH), a hyperinflammatory syndrome characterized by fatal hypercytokinemia with multi-organ failure [13]. Many serum cytokines can be elevated in COVID-19, such as IL-1β, IL-2, IL-4, IL-6, IL-7, IL-8, IL-9, IL-10, IL-18, granulocyte stimulating factor (G-CSF), IP-10, monocyte chemoattractant protein (MCP)-1, MCP-3, macrophage inflammatory protein 1α (MIP-1A), cutaneous T-cell attracting chemokine (CTACK), IFN-γ, and TNF-α [14]. Unlike the cytokine storm associated with SARS, the storm induced by SARS-CoV-2 is associated with increased IL-4 and IL-10, anti-inflammatory cytokines released by T-helper 2 (Th2) cells [14]. However, in severe SARS-CoV-2 infection, the total T cell counts as well as CD4+ and CD8+ T cell counts were all significantly lower than that in more moderate cases [15]. This suggests there is a profound loss of T cells in COVID-19 patients, with progressive T cell exhaustion correlating with severity of the disease [15]
-
B.
The role of interferons
There are three types of IFNs. Type I IFNs includes IFN-α, β and ω. Type II IFN is composed of IFN-γ, and type III IFN is composed of IFN-λ. Both type I and III IFNs share similar expression patterns and signaling cascades, and both up-regulate IFN-stimulated genes (ISGs) to promote an antiviral response. However, the two differ partially by their tissue expression. Type I IFNs are most prominently seen in endothelial cells across various organs, whereas IFN-λ is prominent in epithelial cells of the stomach, intestine, and lungs, but has limited activity in the CNS and spleen [16]. Neutrophils also express high levels of IFN-λ, and IFN-λ appears to be the initial IFN produced to suppress viral spread, followed by type I IFNs to enhance the body’s antiviral response [17].
In severe COVID-19 cases, IFN-α, type I-associated ISGs, and IFN-γ were significantly lower than that in milder cases [1], [18], suggesting that SARS-CoV-2 infection inhibit IFN production, especially in severe cases. Preclinical studies have demonstrated that IFN-λ reduced disease severity and risk of transmission of SARS-CoV-2 and could be a promising candidate for the treatment of COVID-19, even though the underlying mechanism of SARS-CoV-2 immune evasion and suppression of the IFN response remains unclear [19].
-
C.
The pathogenesis of SARS-CoV-2 in the nervous system
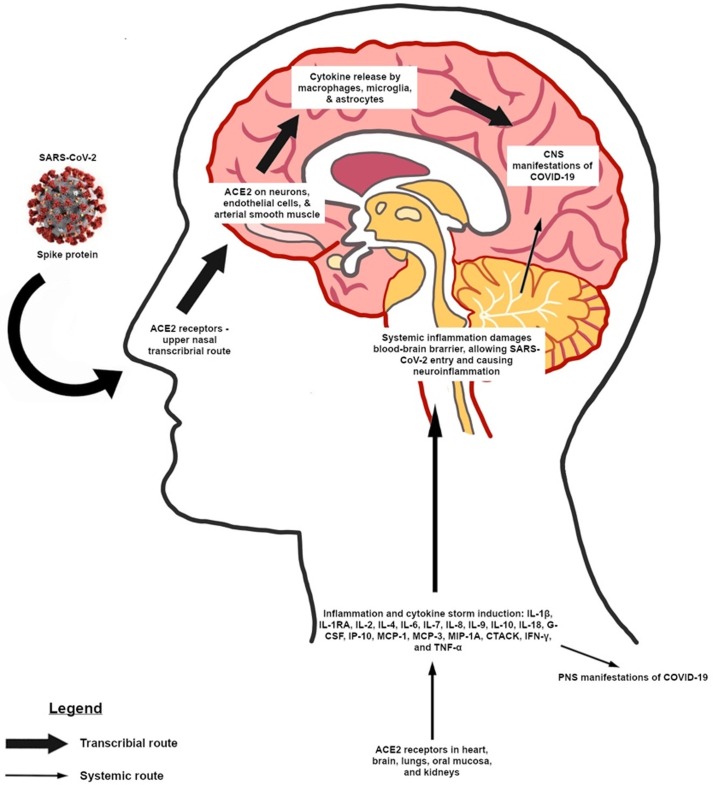
In the brain, the expression of ACE2 receptors has been found on neurons, endothelial cells, and arterial smooth muscle [4]. Similar to SARS and MERS viruses, SARS-CoV-2 may enter the cerebral tissues by crossing the blood-brain barrier via hematogenous or lymphatic spread, or via an upper nasal transcribrial route that includes the olfactory bulb where it binds to ACE2 receptors through its S protein (Fig. 1 ). Viral invasion of the brainstem by SARS-CoV-2 may contribute to brainstem dysfunction and respiratory failure [20]. ACE2 inhibitors are often given to patients with hypertension and diabetes, comorbidities commonly associated with poor prognosis. However, ACE2 inhibitors have been demonstrated to increase the expression of ACE2 receptors [21], increasing the vulnerability of cells to infection with the virus. Endothelial cells in the brain express ACE2 receptors, the expression of which can also be induced in neurons, raising the possibility that SARS-CoV-2 related to the encephalitis and associated stroke, a potential complication [22].
Fig. 1.
Possible pathways of SARS-CoV-2 pathogenesis causing neurological manifestations. SARS-CoV-2 may travel through an upper nasal transcribial route involving the olfactory bulb, binding to ACE2 receptors in the nasopharynx and brain and causing cytokine storm via activation of macrophages, microglia, and astrocytes. Alternatively, systemic infection and inflammation by SARS-CoV-2 can also lead to CNS and PNS manifestations if the blood–brain barrier is compromised.
Coronaviruses can infect macrophages, astrocytes, and microglia, and their infection of induces proinflammatory cytokine release from astrocytes and microglia to facilitate pathogen clearance [23]. Since SARS-CoV is a neurotropic virus, and SARS-CoV-2 likely is as well, CNS infection of these viruses can potentially cause cytokine storm and also chronic inflammation of the brain [20], [24].
3. Neurological manifestations of SARS-CoV-2 infection
Potential impairment of the nervous system was first suggested by reports for the loss of taste and smell in COVID-19 patients [25]. However, these symptoms have been temporary and do not appear to persist more than a month [26]. Other neurological complications including stroke, memory loss, and potentially fatal encephalitis need further investigation [27]. The virus seems to act predominantly on cerebral blood vessels rather than predominantly on neural cells. Because SARS-CoV-2 is not consistently found in cerebrospinal fluid (CSF) of patients with brain damage, the virus is not likely attacking the neural tissues directly. However, the results of some patients’ neuroimaging and biopsies suggest that inflammation was caused by exacerbation of immune response to virus infection [28]. Deficiency in immune regulation is likely the key cause. Follow-up studies are needed to better understand the long-term neurological consequences and characterize the specific drivers of brain inflammation. In the following sections, the CNS and PNS involvement in SARS-CoV-2 infection was discussed.
-
A.
CNS Manifestations
SARS-CoV-2 induced neurological impairment and clinical manifestations are summarized in Table 1 .
-
1.
Neurological symptoms
Table 1.
Neurological Manifestations for the CNS in COVID-19 Patients.
| Symptoms | Incidence and Notes | References |
|---|---|---|
| Headache | 8% | Chen et al. [1]; Huang et al. [14] |
| 10% for all patients | Chen et al. [15] | |
| 13.1% (17.0% in severe cases) | Mao et al. [3] | |
| 6.5% (8.3% in severe cases) | Wang et al. [116] | |
| 11% in severe patients | Chen et al. [117] | |
| Dizziness | 16.8% (19.3% in severe cases) | Mao et al. [3] |
| 9.4% (22.2% in severe cases) | Wang et al. [116] | |
| 8% | Chen et al. [15] | |
| Agitation/Delirium | 69% in ICU patients | Helms et al. [2] |
| Impaired consciousness | 14.8% in severe cases | Mao et al. [3] |
| 65% in ICU patients | Helms et al. [2] | |
| Ataxia | 0.5% (1.1% in severe cases) | Mao et al. [3] |
| Acute cerebrovascular disease | 5.7% in severe cases | Mao et al. [3] |
| 5.0% (9.8% in severe cases and 1.6% in non-severe cases) | Li et al. [37] | |
| 3 young (<40y/o) patients (3/3) | Oxley et al. [35] | |
| 23.1% in ICU patients | Helms et al. [2] | |
| 4/4 (4 case study) | Avula et al. [118] | |
| 7.7% | Klok et al. [119] | |
| A case study | Sharifi-Razavi et al. [120] | |
| 20.6% in intubated patients | Dogan et al. [121] | |
| Convulsions/Seizures/Status epilepticus | A case study | Moriguchi et al. [60] |
| 63.6% in EEG of acute ill patients | Galanopoulouet al. [71] | |
| A case study | Vollono et al. [72] | |
| Epilepsy | 0.5% (1.1% in severe cases) | Mao et al. [3] |
| Encephalitis | A case study | Alolama et al. [122]; Duong et al. [123]; McAbee et al. [57]; Pilotto et al. [55]; Ye et al. [106] |
| Rhombencephalitis | A case study | Wong et al. [56] |
| Meningitis | A case study | Moriguchi et al. [60]; Moghimi et al. [61] |
| Encephalopathy | 9% (20% in deceased patients) | Chen et al. [117] |
| A case study | Filatov et al. [44]; Farhadian et al. [124] | |
| A two-case study | Franceschi et al. [125] | |
| Posterior reversible encephalopathy syndrome | A case study | Rogg et al. [126] |
| Acute hemorrhagic necrotizing encephalopathy | A case study | Poyiadji et al. [28] |
| Leukoencephalopathy | A case study | Sachs et al. [127] |
| Demyelinating lesions Neuromyelitis optica |
A case study | Valiuddin et al. [128];Zanin et al. [65] |
| Myelitis | A case study | Chow et al. [63]; Sarma and Bilello [129] |
| Acute disseminated encephalomyelitis | A case study | Parsons et al. [64] |
Headache is one of the most common neurological symptoms in patients with COVID-19. Neurological symptoms were found in 13.9% of 5335 COVID-19 patients, while headache was seen in 11.74% of patients, followed by dizziness and altered consciousness observed in 1.17% and 1.17% of patients respectively [29]. A meta-analysis including over 40,000 patients from 60 studies estimated that the incidence of headache is high as 12% [30]. Headache generally presents between the 7th and 10th days after symptom onset [31].
The occurrence of headache has been associated with cytokine storm. Headache have been suggested as the outcome of proinflammatory cytokine-mediated activation of trigeminal nerve endings, direct SARS-CoV-2 invasion of the nerve endings, or vasculopathy in ACE2-expressing endothelial cells. Cytokines have previously been proposed as mediators of headaches. The CSF IL-1β and TNF-α are elevated in patients with cervicogenic headaches; while IL-1, transforming growth factor-b1 (TGF-β1), and MCP-1 are elevated in the of CSF of patients with episodic tension-type headaches (TTH) and migraines [32]. IL-6 is also significantly increased in episodic and chronic TTH patients [33]. TGN1412, an anti-CD28 monoclonal antibody that directly stimulates T cells triggered a systemic inflammatory response involving headache, myalgias, erythema, vasodilation, and hypotension [34]. The outcome has been associated with TGN1412-induced cytokine storm with elevated TNF-α, IFN-γ, IL-1β, IL-6, IL-8, and IL-10 [34].
Although the direct relationship between cytokine storm and COVID-19 induced headache has not been established, the data discussed above suggest that the cytokine storm induced by SARS-CoV-2 infection may be responsible for their high incidence of headache. However, no specific cytokine profile in cytokine storm induced headache has yet been identified.
-
2.
Acute cerebrovascular disease (CVD)
About 5% of hospitalized COVID-19 patients develop stroke, mostly ischemic [3]. New-onset large-vessel stroke in young COVID-19 patients (<50 years old) were reported [35]. The reason for CVD in younger patients is unclear. Cerebral hemorrhage is a rare complication, though the severe thrombocytopenia associated with severe and critically ill patients increases the risk [36].
Inflammation and a hypercoagulable state have been associated with COVID-19 CVD and severe disease [37], [38]. Proinflammatory cytokines can activate the coagulation system and down-regulate anticoagulant pathways. Of particular interests are cytokines IL-1β, IL-6, and TNF-α, which are increased in the brain after experimental ischemia as well as in the CSF and blood of stroke patients [39], [40]. In addition, IL-6 has been linked to hypercoagulability [41].
Infections with Helicobacter pylori, Chlamydia pneumoniae, Mycoplasma pneumoniae, Haemophilus influenzae, Epstein-Barr virus (EBV), herpes simplex virus (HSV)-1 and HSV-2, and cytomegalovirus increase the risk of stroke via cytokine release [42]. For example, Patients with neisseria meningitidis infection-associated ischemic stroke had significantly elevated IL-6 [43]. A systemic inflammatory response to an infection can lead to vascular endothelial cell injury, predisposing the patient to intracranial hemorrhage. Thus, the cytokine levels may help predict CVD events in COVID-19 patients.
-
3.
Encephalopathy/Encephalitis
-
a.
Acute encephalopathy
COVID-19 associated encephalopathy was first reported by Filatov et al. in a 78-year old male with atrial fibrillation, cardioembolic stroke, Parkinson’s disease, chronic obstructive pulmonary disease, and recent cellulitis [44]. CSF analysis showed no evidence of CNS infection. The pathogenic mechanism of the patient’s encephalopathy is unclear, but cytokine storm seems to be a strong possibility.
Encephalopathy is commonly preceded by influenza, herpesvirus-6 (HHV-6), and RSV [45]. Elevated serum levels of interleukin (IL)-6, IL-10, and soluble tumor necrosis factor (TNF) receptor (sTNFR1) have been associated with poor outcome and neurological sequelae [46].
The causes of encephalopathy can be classified by three mechanisms: metabolic error, excitotoxicity, and cytokine storm. The latter can occur with multiple organ failure that often involves the heart and lungs, disseminated intravascular coagulation (DIC), and HLH [45].
In infection induced encephalopathy, an increase in cytokines has been illustrated. TNF-α are elevated in both serum and CSF of children with influenza virus-associated encephalitis/encephalopathy (IAEE) [47]. Similarly, in adults with IAEE, elevated IL-6 and IL-10 has been reported [48]. HHV-6B associated hemorrhagic shock and encephalopathy syndrome have been linked to elevated IL-1β, IL-6, IL-10, IL-8, CXCL9 (also known as monokine induced by IFN-gamma, MIG), MCP-1, and IP-10 [49]. In the pandemic variant of influenza virus, H1N1, cytokine storm was an early host response, since serum MCP-1, MIP-1β, IFN-γ, TNF-α, IL-6, IL-8, IL-9, IL-12p70, IL-15, IL-17, IP-10, CXCL2, and CCL4 were all elevated, with IL-6, IL-12p70, and IL-15, a hallmark of critical disease [50]. In 3 fatal H1N1 cases, serum IL-6 was increased in all patients, while TNF-α, IL-8, and MCP-1 were increased in 2 of the 3 patients, both of whom had neurological complications, including confusion, headache, dizziness, ataxia, seizures, and encephalopathy [51]. However, COVID-19 induced acute encephalopathy remains to be defined
-
b.
Acute necrotizing encephalopathy
Acute necrotizing encephalopathy (ANE) presents with multiple focal lesions of edematous necrosis, symmetrically distributed in the bilateral thalami and other brain regions like the putamen, cerebral and cerebellar deep white matter, and brainstem tegmentum.
Poyiadji et al. described the first case of COVID-19 associated ANE, a woman in her 50 s with fever, cough, and altered mentation [28]. CSF cultures were negative and SARS-CoV-2 was not tested. MRI showed hemorrhagic lesions in the thalami, medial temporal lobes, and subinsular regions to confirm ANE. She was treated with intravenous immunoglobulin (IVIG).
ANE has been associated with influenza A, herpesviruses, and many other It probably results from cytokine storm with elevated CSF IL-6 and TNF-α levels. The former is neurotoxic at high concentrations and the latter damages the CNS endothelium [52]. High dose steroids and IVIG may help control ANE elevations of IL-6 and TNF-α [52], [53].
Radmanesh et al. described 2 cases of adult-onset HLH with cerebral involvement appearing as ANE [54]. One patient presented with DIC that progressed rapidly to multiple intracerebral hemorrhages with coma. Prior to this report, HLH with associated ANE had only been reported in children [54]. As previously noted, the COVID-19 cytokine storm profile in adults sometimes resembles that of HLH [13]
-
c.
Encephalitis
COVID-19 induced encephalitis was reported in a patient with positive nasopharyngeal swab and negative CSF test for SARS-CoV-2 [55]. His symptoms included altered consciousness, akinetic syndrome with mutism, and moderate nuchal rigidity. MRI was negative but EEG showed generalized theta slowing. CSF analysis showed elevated IL-6, IL-8, and TNF-α during the akinetic mutism. The levels of IL-8 and TNF-α returned to normal after treatment by steroids and with recovery; however, IL-6 remained stable after treatment. A case of adult rhombencephalitis with spinal cord involvement has also been reported, along with a case of in an 11-year-old child who had positive nasopharyngeal and CSF tests for SARS-CoV-2 [56], [57].
Cytokine storms from viral infection by H1N1, lentivirus, and RSV, among others, have been linked to encephalitis with IL-6, TNF-α, and IFN-γ elevated in most cases [58], [59].
-
4.
Meningitis
COVID-19 encephalitis/meningitis was first reported by Moriguchi et al. with SARS-CoV-2 detected in patient’s CSF [60]. His MRI showed hyperintensity along the wall of the right lateral ventricle and hyperintense signal changes in the right mesial temporal lobe and hippocampus. COVID-19 induced meningitis was also reported in a 78-year-old man with pulmonary embolism and acute renal failure [61]. Viral meningitis has been linked to cytokine storm, especially with elevated IL-6 and IFN-γ [62].
-
5.
Myelitis/Demyelination
A 60-year-old man diagnosed with COVID-19 has been reported to present with bilateral lower limb weakness, urinary retention and constipation [63]. Serum C-reactive protein, amyloid, IL-6, and ferritin were all elevated on admission; however, lumbar puncture was not performed due to hospital restrictions. Acute disseminated encephalomyelitis has also been reported in a 51-year female [64].
Brain and spine demyelination was seen in a 54-year-old COVID-19 patient admitted with pneumonia and seizures [65]. MRI showed multiple, non-enhancing demyelinating lesions in the brain and spine. The authors suggested that cytokine storm, especially with elevated IL-1, IL-6, and TNF-α, potentially causes glial cell activation and subsequent demyelination. In cerebellar cultures, activated microglia induced by lipopolysaccharides released proinflammatory cytokines IL-1β, IL-6, and TNF-α, increased inducible nitric oxide synthase expression and reactive oxygen species production. All of this contributed to the resulting demyelination and axonal damage [66]. IL-11 may also regulate autoimmune demyelination as demonstrated in a mouse model of experimental encephalomyelitis [67].
-
6.
Other disorders
COVID-19 has been linked to convulsions, seizures, and epilepsy [68], [69], [70]. EEG findings showed sporadic epileptiform discharges (EDs) in 40.9% of COVID-19 positive patients with encephalopathy along with frontal sharp waves in most of these patients [71]. Focal status epilepticus (SE) was the presenting symptom for one patient [72].
Several studies have found elevated serum IL-6, IL-1β, and TNF-α levels after acute febrile convulsions. Tonic-clonic seizures have been associated with IL-6 elevations in the serum and CSF [73]. However, the role of cytokines in COVID-19 associated seizures is unclear. Ataxia was reported in one patient without further details [3], and myoclonus and cerebellar ataxia following COVID‐19 has also been illustrated [74]. The role of cytokines was not investigated in these cases.
-
B.PNS Manifestations
-
1.Peripheral nerve and root manifestations (Table 2 )
-
1.
-
a.
Cranial nerve manifestations
Table 2.
Symptoms for PNS in COVID-19 Patients.
| PNS Neurological Symptom | Incidence and Notes | References |
|---|---|---|
| Hypogeusia | 5.1% (3.4% in severe cases) | Mao et al. [3] |
| 19.1% | Bertlich et al. [130] | |
| 10.2% | Giacomelli et al. [78] | |
| 88.0% | Lechien et al. [76] | |
| 71.0% | Yan et al. [77] | |
| 47.2% | Vaira et al. [131] | |
| Hyposmia/Olfactory neuropathy | 5.6% (3.4% in severe cases) | Mao et al. [3] |
| 29.8% | Bagheri et al. [132] | |
| 5.1% | Bertlich et al. [130] | |
| A two-case study | Kirschenbaum et al. [75] | |
| 85.6%% | Lechien et al. [76] | |
| 68.0% | Yan et al. [77] | |
| 86.1% | Vaira et al. [131] | |
| Diplopia | A two-case study | Dinkin et al. [83] |
| Facial nerve palsy | A case study | Goh et al. [81] |
| A case study | Wan et al. [82] | |
| Facial diplegia | A case study | Caamaño et al. [92] |
| Neuralgia | 2.3% (4.5% in severe cases) | Mao et al. [3] |
| A case study | Shors et al. [94] | |
| Guillain-Barré syndrome | A case study | Zhao et al. [84]; Alberti et al. [86]; Ottaviani et al. [87]; Pfefferkorn et al. [88]; Padroni et al. [89]; Chamdessanche et al. [90]; Vaira et al. [131]; Sancho-Saldaña et al. [133]; Sedaghat et al. [134]; Webb et al. [135] |
| A five-case study | Toscanoet al. [136] | |
| Miller Fisher syndrome | A two-case study | Dinkin et al. [83] |
| A two-case study | Gutiérrez-Ortiz et al. [91] | |
| Polyneuritis cranialis | A two-case study | Gutiérrez-Ortiz et al. [91] |
| Myasthenic crisis | A case study | Delly et al. [95] |
| Skeletal muscle injury, rhabdomyolysis | 10.7% (19.3% in severe cases) | Mao et al. [3] |
| A case study | Jin and Tong [96] |
The most reported cranial nerve symptoms are primarily hyposmia and dysgeusia, due to virus contact with gustatory or olfactory ACE2-expressing cells or possibly from peripheral nerve infection [3], [75]. Olfactory nerve fibers of post-mortem showed CD68+ digestion chambers and intraneural CD45+ leukocytes suggesting axonal damage and inflammatory neuropathy. This possibly explains anosmic symptoms seen in COVID-19 patients [75]. In one study, the prevalence of olfactory dysfunction (hyposmia or anosmia) and gustatory dysfunction was reported to be 85.6% 88.8% respectively [76]. In a group of non-intubated patients, reported olfactory and gustatory impairment were 68% and 71% respectively [77]. Yan et al. proposed that inpatient cases may have a more pulmonary-centric viral infection, whereas ambulatory cases may have a more nasal-centric infection, suggesting anosmia and ageusia may have prognostic potential [77]. In an Italian hospital, only 33.9% of 59 patients had reported either olfactory or taste disorder [78].
The primary reports have suggested that cytokines might be the cause of hyposmia symptoms. IL-6 was significantly increased in plasma, saliva, and nasal mucus of patients with hyposmia [79]. In a chronic rhinosinusitis-associated olfactory loss mouse model, TNF-α induction prevented olfactory regeneration [80].
Bell’s palsy can be a manifestation of SARS-CoV-2 infection. A 27-year-old man developed Bell’s palsy 6 days after onset of symptoms [81]. A similar para-infectious case was reported by Wan et al. with a 65-year-old woman who was positive on nasopharyngeal swab for COVID-19 infection [82].
Cranial nerve palsy manifested as diplopia and a right abducens palsy has also been reported in a 71-year-old woman with COVID-19 [83]. Optic nerve sheath and posterior tenon capsules of the involved eye were enhanced on MRI, which could suggest viral leptomeningeal invasion [83].
-
b.
Guillain-Barré Syndrome (GBS)
GBS has been reported as a complication of COVID-19. Though initial reports of GBS [84], [85] could not ascertain a causative relationship between COVID-19 and GBS, subsequent reports support this link [84], [85], [86]. Because GBS is an immune-mediated syndrome, there may be a currently unclear acute dysimmune process triggered by COVID-19-associated systemic inflammatory responses that causes GBS [87]. It should also be noted that most reports of GBS syndrome suggest that GBS in COVID-19 is a para-infectious complication instead of the typical post-infectious GBS manifestation [84], [85], [86], [87], [88]. However, post-infectious GBS has also been reported in some COVID-19 patients [89], [90].
MFS and polyneuritis cranialis, rare GBS variants, have been linked to COVID-19 infection [91]. Both syndromes may have arisen from immune-mediated injury, rather than neurotropic invasion by SARS-CoV-2 since CSF tests for SARS-CoV-2 in the patients were negative along with the positive serum GD1b-IgG antibodies in the MFS patient. Other rare GBS variants are likely still emerging. Facial diplegia in a 61-year-old male has recently been suggested as an atypical GBS variant from COVID-19 infection [92].
GBS is typically associated with various etiologies including preceding infection, most commonly Campylobacter jejuni. Marked increase of cytokines has previously been reported in GBS and its variants as well as in experimental autoimmune neuritis, the animal model of GBS. However, the role of cytokines in GBS associated with COVID-19 infection has not been elucidated.
-
c.
Critical illness polyneuropathy
Critical illness polyneuropathy (CIP) occurs in up to 40% of critically ill patients on mechanical ventilation for over 3 weeks. CIP incidence may be increased and its duration prolonged by COVID-19 [93]. Neuralgia occurred in 2.3% of patients [3]. Severe acute herpetic neuralgia and herpes zoster have also been reported as complications. Despite early anti-viral therapy, one patient developed sensations of skin burning, allodynia, and sinus and tooth pain that was still persistent on follow-up 4 weeks after the initial eruption [94].
-
d.
Neuromuscular junction
A case of myasthenic crisis was reported in a 56-year-old female with a past history of acetylcholine receptor antibody positive myasthenia gravis (MG) for over five years [95]. Both COVID-19 and MG crises are associated with cytokine dysregulation and a proinflammatory state [95].
-
e.
Skeletal muscle injury
Skeletal muscle injury, indicated by elevated creatine kinase levels, was seen in 10.7% of COVID-19 patients [3]. Elevated C-reactive protein, D-dimer, and neutrophil counts were noted in patients neuromuscular dysfunction along with lower lymphocyte counts [3]. This suggests skeletal muscle injury is associated with inflammation and blood coagulation function. Rhabdomyolysis has also been reported in patients with SARS-CoV-2 infection [96]. Studies have found that severe COVID-19 patients were more likely to have skeletal muscle injury [3], [97]. Although cytokines, especially TNF, IL-1, and IL-6, have been reported to play a role in enhancing muscle protein catabolism, their role and mechanism of action remain unclear.
-
C.
Cytokine storm: A common cause of neurological manifestations and poor prognosis of SARS-CoV-2 infection
Elevated cytokine levels in COVID-19 patients can indicate poor prognosis. ICU COVID-19 patients had higher serum levels of G-CSF, IP-10, MCP-1, MIP-1A, and TNF-α compared to COVID-19 patients in the general wards, suggesting that the levels of cytokines are associated with disease severity [14]. Another study found severe cases tended to have more elevated IL-2, IL-6, IL-10, and TNF-α levels compared to moderate [15]. Elevated IP-10, CCL7, and IL-1RA were associated with increased viral load, loss of lung function, and fatal outcome in a cohort of moderate and severe COVID-19 patients [98]. In addition, IL-6 was found to be drastically elevated in critically ill COVID-19 patients, almost 10 times greater than the levels in other patients, and was associated with increased mortality [97], [99]. Moreover, significantly higher levels of inflammatory and hypercoagulability biomarkers, such as D-dimer, C-reactive protein, ferritin, and procalcitonin, have also been associated with severity and mortality [3], [36], [100], [101]. Cytokines may increase the risk of indirect neurological symptoms through widespread organ damage and dysregulation of homeostasis, since neurological symptoms are more commonly seen in severe cases [3], [24]. The ability of serum or CSF cytokines to predict risk of neurological complications and disease severity needs to be further investigated.
-
D.
Cytokine Storm-Targeted Treatments
Since cytokine storm triggered by the SARS-CoV-2 infection is the likely cause of many manifestations, including neurological manifestations, its mitigation along with reduction of viral load becomes an obvious goal for treatment or prophylaxis. Targeting cytokine storm along with reducing viral load in SARS and MERS, especially early in the disease, has been proven to improve prognosis [102]. The same is expected for COVID-19. Though certain promising treatments have been highlighted, many other drugs that are currently being investigated for potential cytokine storm suppression and the anti-inflammatory effects in COVID-19 include immunosuppressants, ulinastatin, stem cell therapy, IL-1 antagonists, and TNF blockers (Table 3 ).
-
1.
Corticosteroids
Table 3.
Proposed and current treatments for cytokine storm in COVID-19 patients.
| Treatment | Status for COVID-19 Treatment | References |
|---|---|---|
| Blood purification | Effective for use with severe/critical patients with cytokine storm and rapid disease progression. Multicenter clinical studies still needed. | [112], [137], [138] |
| Corticosteroids | Various studies have not shown corticosteroids to be effective for mild cases. But it may be effective in severe/critical cases when used early and for a short period, at a low/ moderate dosage or use with cytokine inhibitors like tocilizumab or anakinra. Clinical trials are underway to investigate their efficacy and safety. | [104], [138], [139], [140], [141], [142] |
| IFN-λ | Preclinical studies are promising. IFN-λ has not yet been used in COVID-19 patients, but clinical trials are beginning to investigate its effecicay. | [19], [105] |
| IL-1 inhibitors | Promising preclinical and clinical studies for severe COVID-19 treatment. Clinical trials are still ongoing to confirm the efficacy. | [143], [144], [145], [146] |
| IL-6 inhibitors (tocilizumab, sarilumab, siltuximab) | Tocilizumab effective for severe/critical patients. Approved for use in patients with elevated IL-6 in China. | [108], [147] |
| IVIG | Effective used early and with high dose in severe/critical patients. Must be administered carefully due to numerous adverse effects. | [100], [110], [111] |
| JAK inhibitors (baricitinib, ruxolitinib, fedratinib) | Promising preclinical and clinical studies, but not yet suggested for treatment. Need further confirmation of its efficacy. | [109], [148] |
| Mesenchymal stem cell therapy | Promising preclinical and clinical studies. Shown to be effective in patients, especially in a severe case. Proposed for compassionate use in critically ill patients. | [114], [149], [115] |
| TNF inhibitors (adalimumab) | Promising preclinical studies. Only one clinical trial in China has been registered. More trials are needed. | [144], [150] |
| Ulinastatin | May be a promising therapy at high doses, with clinical trials underway in China. | [151], [152] |
Steroids can be used to broadly suppress immune systems and reduce cytokine storm, but high dose and long-time treatment may be associated with severe and long-term adverse effects as seen with SARS [103]. In a retrospective study of SARS non-ICU patients, early treatment with corticosteroids could aggravate the disease [30]. Because of their immunosuppression effects, corticosteroids may delay the elimination of the virus and increase the risk of secondary infection, especially in the immunocompromised patients; therefore, corticosteroid use in COVID-19 management is not generally recommended [100]. However, when ARDS, shock, or systemic inflammatory response syndrome (SIRS), and/or cardiac failure is present in COVID-19 patients at the severe/hyperinflammation stage, careful corticosteroid use may be justified when used alongside cytokine inhibitors like tocilizumab or anakinra (IL-1 receptor antagonist) [104].
More specific interventions, such as monoclonal antibodies against certain cytokines may be a better choice than steroids, but the large number of cytokine types elevated in COVID-19 patients makes it difficult to decide which and how many antibodies should be used. Nevertheless, antibodies against key proinflammatory cytokines, such as IL-1β, IFN-γ, and TNF-α, are worth trying to use for mitigating the severity and neurological consequences of COVID-19. Since cytokine storm is often followed by reactive oxygen species/reactive nitrogen species (ROS/RNS) surge and directly damage tissues/organs, ROS/RNS mitigating agents may also be a treatment.
-
2.
IFN-λ
IFN-λ, a type III IFN, is a promising therapy for antiviral resistance by activating epithelial cells and inhibiting neutrophil recruitment to the site of inflammation [105]. It has no known proinflammatory effects in the lungs and has demonstrated the potency against various coronaviruses including SARS-CoV and MERS-CoV [105]. Administration of interferons, including IFN-λ, as prophylactic or early-stage viral disease treatment may reduce viral load and improve clinical symptoms; however, it does not appear to reduce the mortality [106].
More recently, interferon has been reported to play distinct roles in the lung depending on the tissue environment. IFN-λ is not found in the upper airways of COVID-19 patients but is abundantly produced in the lower airways. The authors demonstrated that the IFN-λ produced by lung dendritic cells of the lower airways in response to virus can increase susceptibility to bacterial superinfections and exacerbate COVID-19 disease [107]. These findings demonstrate new avenues for the role of IFN-λ in COVID-19 and the potential use of IFN-λ antagonists.
-
3.
IL-6 and Janus kinase (JAK) inhibitors
IL-6 plays a key role in cytokine storm by inducing B cell proliferation, promoting CD4+ T cell response, and inhibiting induction of regulatory T cells, among other effects [108], [109]. Tocilizumab, which binds IL-6 receptors, has been approved for treatment of rheumatoid arthritis and is known to be effective in treatment of B-cell chronic lymphocytic leukemia patients with cytokine storm [108]. Currently, a multicenter, randomized controlled trial of tocilizumab for COVID-19 patients with pneumonia and elevated IL-6 levels in China (ChiCTR2000029765), a single arm open multicenter study of tocilizumab (ChiCTR2000030796), and studies of tocilizumab combined with other treatments (ChiCTR2000030442, ChiCTR2000030894) are all ongoing. However, it should be noted that in animal models of COVID-19, tocilizumab showed limited CNS penetration (brain: plasma ratio of 0.1%). Instead, baricitinib showed high CNS penetration (brain: plasma ratio of 20%) [109]. Baricitinib, a JAK inhibitor with anti-inflammatory and anti-viral activity, has been shown to reduce viral titers, reduce fever and cough symptoms, and decrease IL-6 in COVID-19 patients for 10–12 days [109]. Studies of baricitinib and another JAK inhibitor, ruxolitinib, are currently underway (e.g., NCT04321993, NCT04348071).
-
4.
Intravenous immunoglobulin (IVIG)
IVIG may improve the body’s immune defenses, in part by limiting inflammatory cytokine production and blocking Th1 and Th17 responses, decreasing inflammatory injury in COVID-19 patients [110]. In a retrospective study by Xie and Cao, IVIG treatment in COVID-19 patients with severe pneumonia within 48 h of admission reduced ventilator use and shortened hospital stay while improving 28-day mortality [110]. In a multicenter retrospective cohort study, Shao et al. found that patients hospitalize ≤7 days given a high dose of >15 g/d IVIG significantly reduced 60-day mortality, although 28- and 60-day mortality were not improved with IVIG in the overall cohort [111]. This suggests that patient selection as well as the timing and dosage of IVIG administration are important.
-
5.
Blood purification
Artificial-liver blood purification systems are highly promising for targeting cytokine storm in COVID-19 patients. These systems consist of blood-purification modules, such as plasma exchange and absorption and/or hemo/plasma filtration, which can remove inflammatory factors and block the cytokine storm [112]. When used to treat severe H7N9 influenza patients, the system reduced levels of 17 cytokines and chemokines, including G-CSF, IL-1β, Il-2, IL-4, IL-8, IL-9, IFN-γ, and TNF-α [113]. Since these cytokines are also elevated in COVID-19 patients, it is likely to be effective in reducing cytokine storm in severe and critical cases. Artificial-liver blood-purification systems have already been recommended in multiple expert guidelines for COVID-19 treatment [112]. To demonstrate the ability of this blood purification system to reduce cytokine levels, large, multicenter treatment studies in COVID-19 patients are essential.
-
6.
Mesenchymal stem cells (MSCs)
MSCs are powerful immunomodulators that may help reverse cytokine storm and improve patient outcomes. Treatment of seven COVID-19 pneumonia patients with MSCs significantly improved functional outcomes of all patients including curing 1 severe and 2 common patients with no side effects [114]. Notably in these patients after treatment TNF-α was significantly decreased while anti-inflammatory IL-10 was increased along with decreased cytokine-secreting natural killer and T cells. Other studies have also supported the use of MSCs to treat severe or critically ill COVID-19 cases [115].
4. Conclusion
COVID-19 is an infectious disease caused by the SARS-CoV-2 virus that typically manifests as respiratory symptoms; however, it can cause severe CNS and PNS complications, possibly due to viral neuroinvasion and cytokine storm. Understanding the COVID-19 cytokine storm in context of neurological manifestations will improve clinical management via cytokine storm-targeted treatment. Moving forward, analyzing the presence of SARS-CoV-2 in CSF and serum and cytokine storm will be essential, as will be monitoring recovered COVID-19 patients for post-infection neurological sequelae like GBS, myositis, and Parkinsonism, seen in past coronavirus epidemics.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- 1.Chen N., Zhou M., Dong X., Qu J., Gong F., Han Y., Qiu Y., Wang J., Liu Y., Wei Y., Xia J., Yu T., Zhang X., Zhang L. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020;395(10223):507–513. doi: 10.1016/S0140-6736(20)30211-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Helms J., Kremer S., Merdji H., Clere-Jehl R., Schenck M., Kummerlen C., Collange O., Boulay C., Fafi-Kremer S., Ohana M., Anheim M., Meziani F. Neurologic features in severe SARS-CoV-2 infection. N. Engl. J. Med. 2020;382(23):2268–2270. doi: 10.1056/NEJMc2008597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.L. Mao, H. Jin, M. Wang, Y. Hu, S. Chen, Q. He, J. Chang, C. Hong, Y. Zhou, D. Wang, X. Miao, Y. Li, B. Hu, Neurologic manifestations of hospitalized patients with Coronavirus Disease 2019 in Wuhan, China, JAMA Neurol (2020). [DOI] [PMC free article] [PubMed]
- 4.Xia H., Lazartigues E. Angiotensin-converting enzyme 2 in the brain: properties and future directions. J. Neurochem. 2008;107(6):1482–1494. doi: 10.1111/j.1471-4159.2008.05723.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Jin Y., Yang H., Ji W., Wu W., Chen S., Zhang W., Duan G. Virology, epidemiology, pathogenesis, and control of COVID-19. Viruses. 2020;12(4) doi: 10.3390/v12040372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kim Y., Kim H., Bae S., Choi J., Lim S.Y., Lee N., Kong J.M., Hwang Y.I., Kang J.S., Lee W.J. Vitamin C Is an essential factor on the anti-viral immune responses through the production of interferon-alpha/beta at the initial stage of Influenza A Virus (H3N2) infection. Immune Netw. 2013;13(2):70–74. doi: 10.4110/in.2013.13.2.70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Chousterman B.G., Swirski F.K., Weber G.F. Cytokine storm and sepsis disease pathogenesis. Semin. Immunopathol. 2017;39(5):517–528. doi: 10.1007/s00281-017-0639-8. [DOI] [PubMed] [Google Scholar]
- 8.Cloutier M., Nandi M., Ihsan A.U., Chamard H.A., Ilangumaran S., Ramanathan S. ADE and hyperinflammation in SARS-CoV2 infection- comparison with dengue hemorrhagic fever and feline infectious peritonitis. Cytokine. 2020;136:155256. doi: 10.1016/j.cyto.2020.155256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.A. Mahmud-Al-Rafat, M. Muzammal Haque Asim, A.W. Taylor-Robinson, A. Majumder, A. Muktadir, H. Muktadir, M. Karim, I. Khan, M. Mainul Ahasan, M. Morsaline Billah, A combinational approach to restore cytokine balance and to inhibit virus growth may promote patient recovery in severe COVID-19 cases, Cytokine 136 (2020) 155228. [DOI] [PMC free article] [PubMed]
- 10.Mahmudpour M., Roozbeh J., Keshavarz M., Farrokhi S., Nabipour I. COVID-19 cytokine storm: the anger of inflammation. Cytokine. 2020;133:155151. doi: 10.1016/j.cyto.2020.155151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Noroozi R., Branicki W., Pyrc K., Labaj P.P., Pospiech E., Taheri M., Ghafouri-Fard S. Altered cytokine levels and immune responses in patients with SARS-CoV-2 infection and related conditions. Cytokine. 2020;133:155143. doi: 10.1016/j.cyto.2020.155143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.M.D. de Jong, C.P. Simmons, T.T. Thanh, V.M. Hien, G.J. Smith, T.N. Chau, D.M. Hoang, N.V. Chau, T.H. Khanh, V.C. Dong, P.T. Qui, B.V. Cam, Q. Ha do, Y. Guan, J.S. Peiris, N.T. Chinh, T.T. Hien, J. Farrar, Fatal outcome of human influenza A (H5N1) is associated with high viral load and hypercytokinemia, Nat Med 12(10) (2006) 1203-7. [DOI] [PMC free article] [PubMed]
- 13.P. Mehta, D.F. McAuley, M. Brown, E. Sanchez, R.S. Tattersall, J.J. Manson, U.K. Hlh Across Speciality Collaboration, COVID-19: consider cytokine storm syndromes and immunosuppression, Lancet 395(10229) (2020) 1033-1034. [DOI] [PMC free article] [PubMed]
- 14.Huang C., Wang Y., Li X., Ren L., Zhao J., Hu Y., Zhang L., Fan G., Xu J., Gu X., Cheng Z., Yu T., Xia J., Wei Y., Wu W., Xie X., Yin W., Li H., Liu M., Xiao Y., Gao H., Guo L., Xie J., Wang G., Jiang R., Gao Z., Jin Q., Wang J., Cao B. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395(10223):497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Chen G., Wu D., Guo W., Cao Y., Huang D., Wang H., Wang T., Zhang X., Chen H., Yu H., Zhang X., Zhang M., Wu S., Song J., Chen T., Han M., Li S., Luo X., Zhao J., Ning Q. Clinical and immunological features of severe and moderate coronavirus disease 2019. J. Clin. Invest. 2020;130(5):2620–2629. doi: 10.1172/JCI137244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Sommereyns C., Paul S., Staeheli P., Michiels T. IFN-lambda (IFN-lambda) is expressed in a tissue-dependent fashion and primarily acts on epithelial cells in vivo. PLoS Pathog. 2008;4(3):e1000017. doi: 10.1371/journal.ppat.1000017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Galani I.E., Triantafyllia V., Eleminiadou E.E., Koltsida O., Stavropoulos A., Manioudaki M., Thanos D., Doyle S.E., Kotenko S.V., Thanopoulou K., Andreakos E. Interferon-lambda mediates non-redundant front-line antiviral protection against influenza virus infection without compromising host fitness. Immunity. 2017;46(5):875–890 e6. doi: 10.1016/j.immuni.2017.04.025. [DOI] [PubMed] [Google Scholar]
- 18.Hadjadj J., Yatim N., Barnabei L., Corneau A., Boussier J., Smith N., Pere H., Charbit B., Bondet V., Chenevier-Gobeaux C., Breillat P., Carlier N., Gauzit R., Morbieu C., Pene F., Marin N., Roche N., Szwebel T.A., Merkling S.H., Treluyer J.M., Veyer D., Mouthon L., Blanc C., Tharaux P.L., Rozenberg F., Fischer A., Duffy D., Rieux-Laucat F., Kerneis S., Terrier B. Impaired type I interferon activity and inflammatory responses in severe COVID-19 patients. Science. 2020;369(6504):718–724. doi: 10.1126/science.abc6027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.O'Brien T.R., Thomas D.L., Jackson S.S., Prokunina-Olsson L., Donnelly R.P., Hartmann R. Weak induction of interferon expression by SARS-CoV-2 supports clinical trials of interferon lambda to treat early COVID-19. Clin. Infect. Dis. 2020 doi: 10.1093/cid/ciaa453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Li Y.C., Bai W.Z., Hashikawa T. The neuroinvasive potential of SARS-CoV2 may play a role in the respiratory failure of COVID-19 patients. J. Med. Virol. 2020;92(6):552–555. doi: 10.1002/jmv.25728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sriram K., Insel P.A. Risks of ACE inhibitor and ARB usage in COVID-19: evaluating the evidence. Clin. Pharmacol. Ther. 2020;108(2):236–241. doi: 10.1002/cpt.1863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Nath A. Neurologic complications of coronavirus infections. Neurology. 2020;94(19):809–810. doi: 10.1212/WNL.0000000000009455. [DOI] [PubMed] [Google Scholar]
- 23.Li Y., Fu L., Gonzales D.M., Lavi E. Coronavirus neurovirulence correlates with the ability of the virus to induce proinflammatory cytokine signals from astrocytes and microglia. J. Virol. 2004;78(7):3398–3406. doi: 10.1128/JVI.78.7.3398-3406.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Baig A.M., Khaleeq A., Ali U., Syeda H. Evidence of the COVID-19 virus targeting the CNS: tissue distribution, host-virus interaction, and proposed neurotropic mechanisms. ACS Chem. Neurosci. 2020;11(7):995–998. doi: 10.1021/acschemneuro.0c00122. [DOI] [PubMed] [Google Scholar]
- 25.Baig A.M. Updates on wWhat ACS reported: emerging evidences of COVID-19 with nervous system involvement. ACS Chem. Neurosci. 2020;11(9):1204–1205. doi: 10.1021/acschemneuro.0c00181. [DOI] [PubMed] [Google Scholar]
- 26.Li Z., Liu T., Yang N., Han D., Mi X., Li Y., Liu K., Vuylsteke A., Xiang H., Guo X. Neurological manifestations of patients with COVID-19: potential routes of SARS-CoV-2 neuroinvasion from the periphery to the brain. Front. Med. 2020 doi: 10.1007/s11684-020-0786-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hess D.C., Eldahshan W., Rutkowski E. COVID-19-related stroke. Transl. Stroke Res. 2020;11(3):322–325. doi: 10.1007/s12975-020-00818-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Poyiadji N., Shahin G., Noujaim D., Stone M., Patel S., Griffith B. COVID-19-associated acute hemorrhagic necrotizing encephalopathy: imaging features. Radiology. 2020;296(2):E119–E120. doi: 10.1148/radiol.2020201187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Daou B.J., Koduri S., Palmateer G., Thompson B.G., Chaudhary N., Gemmete J.J., Pandey A.S. Letter: neurological implications of COVID-19 and lessons learned from prior epidemics and pandemics. Neurosurgery. 2020;87(2):E234–E238. doi: 10.1093/neuros/nyaa186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Auyeung T.W., Lee J.S., Lai W.K., Choi C.H., Lee H.K., Lee J.S., Li P.C., Lok K.H., Ng Y.Y., Wong W.M., Yeung Y.M. The use of corticosteroid as treatment in SARS was associated with adverse outcomes: a retrospective cohort study. J. Infect. 2005;51(2):98–102. doi: 10.1016/j.jinf.2004.09.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Belvis R. Headaches during COVID-19: my clinical case and review of the literature. Headache. 2020 doi: 10.1111/head.13841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Bo S.H., Davidsen E.M., Gulbrandsen P., Dietrichs E., Bovim G., Stovner L.J., White L.R. Cerebrospinal fluid cytokine levels in migraine, tension-type headache and cervicogenic headache. Cephalalgia. 2009;29(3):365–372. doi: 10.1111/j.1468-2982.2008.01727.x. [DOI] [PubMed] [Google Scholar]
- 33.Koçer A., Koçer E., Memisogullari R., Domaç F.M., Yüksel H. Interleukin-6 levels in tension headache patients. Clin. J. Pain. 2010;26(8):690–693. doi: 10.1097/AJP.0b013e3181e8d9b6. [DOI] [PubMed] [Google Scholar]
- 34.Suntharalingam G., Perry M.R., Ward S., Brett S.J., Castello-Cortes A., Brunner M.D., Panoskaltsis N. Cytokine storm in a phase 1 trial of the anti-CD28 monoclonal antibody TGN1412. N. Engl. J. Med. 2006;355(10):1018–1028. doi: 10.1056/NEJMoa063842. [DOI] [PubMed] [Google Scholar]
- 35.Oxley T.J., Mocco J., Majidi S., Kellner C.P., Shoirah H., Singh I.P., De Leacy R.A., Shigematsu T., Ladner T.R., Yaeger K.A., Skliut M., Weinberger J., Dangayach N.S., Bederson J.B., Tuhrim S., Fifi J.T. Large-vessel stroke as a presenting feature of Covid-19 in the Young. N. Engl. J. Med. 2020;382(20):e60. doi: 10.1056/NEJMc2009787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.G. Lippi, M. Plebani, B.M. Henry, Thrombocytopenia is associated with severe coronavirus disease 2019 (COVID-19) infections: A meta-analysis, Clin Chim Acta 506 (2020) 145-148. [DOI] [PMC free article] [PubMed]
- 37.Li Y., Li M., Wang M., Zhou Y., Chang J., Xian Y., Wang D., Mao L., Jin H., Hu B. Acute cerebrovascular disease following COVID-19: a single center, retrospective, observational study. Stroke Vasc. Neurol. 2020 doi: 10.1136/svn-2020-000431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Tan C.W., Low J.G.H., Wong W.H., Chua Y.Y., Goh S.L., Ng H.J. Critically ill COVID-19 infected patients exhibit increased clot waveform analysis parameters consistent with hypercoagulability. Am. J. Hematol. 2020;95(7):E156–E158. doi: 10.1002/ajh.25822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ferrarese C., Mascarucci P., Zoia C., Cavarretta R., Frigo M., Begni B., Sarinella F., Frattola L., De Simoni M.G. Increased cytokine release from peripheral blood cells after acute stroke. J. Cereb. Blood Flow Metab. 1999;19(9):1004–1009. doi: 10.1097/00004647-199909000-00008. [DOI] [PubMed] [Google Scholar]
- 40.Intiso D., Zarrelli M.M., Lagioia G., Di Rienzo F., Checchia De Ambrosio C., Simone P., Tonali P., Cioffi Dagger R.P. Tumor necrosis factor alpha serum levels and inflammatory response in acute ischemic stroke patients. Neurol. Sci. 2004;24(6):390–396. doi: 10.1007/s10072-003-0194-z. [DOI] [PubMed] [Google Scholar]
- 41.Stouthard J.M., Levi M., Hack C.E., Veenhof C.H., Romijn H.A., Sauerwein H.P., van der Poll T. Interleukin-6 stimulates coagulation, not fibrinolysis, in humans. Thromb. Haemost. 1996;76(5):738–742. [PubMed] [Google Scholar]
- 42.Grau A.J., Buggle F., Becher H., Zimmermann E., Spiel M., Fent T., Maiwald M., Werle E., Zorn M., Hengel H., Hacke W. Recent bacterial and viral infection is a risk factor for cerebrovascular ischemia: clinical and biochemical studies. Neurology. 1998;50(1):196–203. doi: 10.1212/wnl.50.1.196. [DOI] [PubMed] [Google Scholar]
- 43.de Souza A.L., de Oliveira A.C., Romano C.C., Sztajnbok J., Duarte A.J., Seguro A.C. Interleukin-6 activation in ischemic stroke caused by Neisseria meningitidis serogroup C. Int. J. Cardiol. 2008;127(3):e160–e163. doi: 10.1016/j.ijcard.2007.04.122. [DOI] [PubMed] [Google Scholar]
- 44.Filatov A., Sharma P., Hindi F., Espinosa P.S. Neurological complications of Coronavirus Disease (COVID-19): Encephalopathy. Cureus. 2020;12(3):e7352. doi: 10.7759/cureus.7352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Nara A., Nagai H., Yamaguchi R., Yoshida K., Iwase H., Mizuguchi M. An unusual autopsy case of cytokine storm-derived influenza-associated encephalopathy without typical histopathological findings: autopsy case report. Am. J. Forensic Med. Pathol. 2015;36(1):3–5. doi: 10.1097/PAF.0000000000000129. [DOI] [PubMed] [Google Scholar]
- 46.Hasegawa S., Matsushige T., Inoue H., Shirabe K., Fukano R., Ichiyama T. Serum and cerebrospinal fluid cytokine profile of patients with 2009 pandemic H1N1 influenza virus-associated encephalopathy. Cytokine. 2011;54(2):167–172. doi: 10.1016/j.cyto.2011.01.006. [DOI] [PubMed] [Google Scholar]
- 47.Ichiyama T., Endo S., Kaneko M., Isumi H., Matsubara T., Furukawa S. Serum cytokine concentrations of influenza-associated acute necrotizing encephalopathy. Pediatr. Int. 2003;45(6):734–736. doi: 10.1111/j.1442-200x.2003.01822.x. [DOI] [PubMed] [Google Scholar]
- 48.Kimura E., Okamoto S., Uchida Y., Hirahara T., Ikeda T., Hirano T., Uchino M. A reversible lesion of the corpus callosum splenium with adult influenza-associated encephalitis/encephalopathy: a case report. J. Med. Case Rep. 2008;2:220. doi: 10.1186/1752-1947-2-220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Kawamura Y., Nakai H., Sugata K., Asano Y., Yoshikawa T. Serum biomarker kinetics with three different courses of HHV-6B encephalitis. Brain Dev. 2013;35(6):590–595. doi: 10.1016/j.braindev.2012.08.005. [DOI] [PubMed] [Google Scholar]
- 50.Bermejo-Martin J.F., Ortiz de Lejarazu R., Pumarola T., Rello J., Almansa R., Ramirez P., Martin-Loeches I., Varillas D., Gallegos M.C., Seron C., Micheloud D., Gomez J.M., Tenorio-Abreu A., Ramos M.J., Molina M.L., Huidobro S., Sanchez E., Gordon M., Fernandez V., Del Castillo A., Marcos M.A., Villanueva B., Lopez C.J., Rodriguez-Dominguez M., Galan J.C., Canton R., Lietor A., Rojo S., Eiros J.M., Hinojosa C., Gonzalez I., Torner N., Banner D., Leon A., Cuesta P., Rowe T., Kelvin D.J. Th1 and Th17 hypercytokinemia as early host response signature in severe pandemic influenza. Crit. Care. 2009;13(6):R201. doi: 10.1186/cc8208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Baltagi S.A., Shoykhet M., Felmet K., Kochanek P.M., Bell M.J. Neurological sequelae of 2009 influenza A (H1N1) in children: a case series observed during a pandemic. Pediatr. Crit. Care Med. 2010;11(2):179–184. doi: 10.1097/PCC.0b013e3181cf4652. [DOI] [PubMed] [Google Scholar]
- 52.Kansagra S.M., Gallentine W.B. Cytokine storm of acute necrotizing encephalopathy. Pediatr. Neurol. 2011;45(6):400–402. doi: 10.1016/j.pediatrneurol.2011.09.007. [DOI] [PubMed] [Google Scholar]
- 53.Huang S.M., Chen C.C., Chiu P.C., Cheng M.F., Lai P.H., Hsieh K.S. Acute necrotizing encephalopathy of childhood associated with influenza type B virus infection in a 3-year-old girl. J. Child Neurol. 2004;19(1):64–67. doi: 10.1177/08830738040190010709. [DOI] [PubMed] [Google Scholar]
- 54.Radmanesh F., Rodriguez-Pla A., Pincus M.D., Burns J.D. Severe cerebral involvement in adult-onset hemophagocytic lymphohistiocytosis. J. Clin. Neurosci. 2020;76:236–237. doi: 10.1016/j.jocn.2020.04.054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Pilotto A., Odolini S., Masciocchi S., Comelli A., Volonghi I., Gazzina S., Nocivelli S., Pezzini A., Foca E., Caruso A., Leonardi M., Pasolini M.P., Gasparotti R., Castelli F., Ashton N.J., Blennow K., Zetterberg H., Padovani A. Steroid-Responsive Encephalitis in Coronavirus Disease 2019. Ann. Neurol. 2020 doi: 10.1002/ana.25783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Wong P.F., Craik S., Newman P., Makan A., Srinivasan K., Crawford E., Dev D., Moudgil H., Ahmad N. Lessons of the month 1: A case of rhombencephalitis as a rare complication of acute COVID-19 infection. Clin. Med. (Lond) 2020 doi: 10.7861/clinmed.2020-0182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.McAbee G.N., Brosgol Y., Pavlakis S., Agha R., Gaffoor M. Encephalitis Associated with COVID-19 Infection in an 11-Year-Old Child. Pediatr. Neurol. 2020;109:94. doi: 10.1016/j.pediatrneurol.2020.04.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Sy M., Kitazawa M., Medeiros R., Whitman L., Cheng D., Lane T.E., Laferla F.M. Inflammation induced by infection potentiates tau pathological features in transgenic mice. Am. J. Pathol. 2011;178(6):2811–2822. doi: 10.1016/j.ajpath.2011.02.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Bastos M.S., Coelho-Dos-Reis J.G., Zauli D.A., Naveca F.G., Monte R.L., Pimentel J.P., Macario V.M., da Silva N.L., Peruhype-Magalhaes V., Pascoal-Xavier M.A., Guimaraes A., Carvalho A.T., Malheiro A., Martins-Filho O.A., Mourao M.P. Divergent cerebrospinal fluid cytokine network induced by non-viral and different viral infections on the central nervous system. BMC Infect. Dis. 2015;15:345. doi: 10.1186/s12879-015-1035-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Moriguchi T., Harii N., Goto J., Harada D., Sugawara H., Takamino J., Ueno M., Sakata H., Kondo K., Myose N., Nakao A., Takeda M., Haro H., Inoue O., Suzuki-Inoue K., Kubokawa K., Ogihara S., Sasaki T., Kinouchi H., Kojin H., Ito M., Onishi H., Shimizu T., Sasaki Y., Enomoto N., Ishihara H., Furuya S., Yamamoto T., Shimada S. A first case of meningitis/encephalitis associated with SARS-Coronavirus-2. Int. J. Infect. Dis. 2020;94:55–58. doi: 10.1016/j.ijid.2020.03.062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Moghimi M., Ghodrati S., Abbaspourrad Z., Mojhdehi A.M., Jafari S., Mansouri R., Khodadadi K., Muhammmadi M.J. Case Report of 78 –year-old man with meningitis, Pulmonary Thromboembolism & SARS-Coronavirus-2 infection. Research Square Version. 2020;1 [Google Scholar]
- 62.Nagafuchi M., Nagafuchi Y., Sato R., Imaizumi T., Ayabe M., Shoji H., Ichiyama T. Adult meningism and viral meningitis, 1997–2004: clinical data and cerebrospinal fluid cytokines. Intern. Med. 2006;45(21):1209–1212. doi: 10.2169/internalmedicine.45.1769. [DOI] [PubMed] [Google Scholar]
- 63.Chow C.C.N., Magnussen J., Ip J., Su Y. Acute transverse myelitis in COVID-19 infection. BMJ Case Rep. 2020;13(8) doi: 10.1136/bcr-2020-236720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Parsons T., Banks S., Bae C., Gelber J., Alahmadi H., Tichauer M. COVID-19-associated acute disseminated encephalomyelitis (ADEM) J. Neurol. 2020 doi: 10.1007/s00415-020-09951-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Zanin L., Saraceno G., Panciani P.P., Renisi G., Signorini L., Migliorati K., Fontanella M.M. SARS-CoV-2 can induce brain and spine demyelinating lesions. Acta Neurochir (Wien) 2020;162(7):1491–1494. doi: 10.1007/s00701-020-04374-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.di Penta A., Moreno B., Reix S., Fernandez-Diez B., Villanueva M., Errea O., Escala N., Vandenbroeck K., Comella J.X., Villoslada P. Oxidative stress and proinflammatory cytokines contribute to demyelination and axonal damage in a cerebellar culture model of neuroinflammation. PLoS ONE. 2013;8(2):e54722. doi: 10.1371/journal.pone.0054722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Gurfein B.T., Zhang Y., Lopez C.B., Argaw A.T., Zameer A., Moran T.M., John G.R. IL-11 regulates autoimmune demyelination. J. Immunol. 2009;183(7):4229–4240. doi: 10.4049/jimmunol.0900622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Abdulsalam M.A., Abdulsalam A.J., Shehab D. Generalized status epilepticus as a possible manifestation of COVID-19. Acta Neurol. Scand. 2020 doi: 10.1111/ane.13321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Kuroda N. Epilepsy and COVID-19: Associations and important considerations. Epilepsy Behav. 2020;108:107122. doi: 10.1016/j.yebeh.2020.107122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Yasri S., Wiwanikit V. COVID-19 and Epilepsy. Ann. Indian Acad. Neurol. 2020;23(Suppl. 1):S43. doi: 10.4103/aian.AIAN_254_20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Galanopoulou A.S., Ferastraoaru V., Correa D.J., Cherian K., Duberstein S., Gursky J., Hanumanthu R., Hung C., Molinero I., Khodakivska O., Legatt A.D., Patel P., Rosengard J., Rubens E., Sugrue W., Yozawitz E., Mehler M.F., Ballaban-Gil K., Haut S.R., Moshe S.L., Boro A. EEG findings in acutely ill patients investigated for SARS-CoV-2/COVID-19: a small case series preliminary report. Epilepsia Open. 2020;5(2):314–324. doi: 10.1002/epi4.12399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Vollono C., Rollo E., Romozzi M., Frisullo G., Servidei S., Borghetti A., Calabresi P. Focal status epilepticus as unique clinical feature of COVID-19: a case report. Seizure. 2020;78:109–112. doi: 10.1016/j.seizure.2020.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Lehtimaki K.A., Keranen T., Palmio J., Rainesalo S., Saransaari P., Peltola J. Regulation of cerebrospinal fluid levels of cytokines after seizures: the role of IL-6 and glutamic acid. Eur. J. Neurol. 2009;16(4) doi: 10.1111/j.1468-1331.2009.02557.x. [DOI] [PubMed] [Google Scholar]
- 74.Dijkstra F., Van den Bossche T., Willekens B., Cras P., Crosiers D. Myoclonus and cerebellar ataxia following Coronavirus Disease 2019 (COVID-19) Mov. Disord. Clin. Pract. 2020 doi: 10.1002/mdc3.13049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Kirschenbaum D., Imbach L.L., Ulrich S., Rushing E.J., Keller E., Reimann R.R., Frauenknecht K.B.M., Lichtblau M., Witt M., Hummel T., Steiger P., Aguzzi A., Frontzek K. Inflammatory olfactory neuropathy in two patients with COVID-19. Lancet. 2020;396(10245):166. doi: 10.1016/S0140-6736(20)31525-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Lechien J.R., Chiesa-Estomba C.M., De Siati D.R., Horoi M., Le Bon S.D., Rodriguez A., Dequanter D., Blecic S., El Afia F., Distinguin L., Chekkoury-Idrissi Y., Hans S., Delgado I.L., Calvo-Henriquez C., Lavigne P., Falanga C., Barillari M.R., Cammaroto G., Khalife M., Leich P., Souchay C., Rossi C., Journe F., Hsieh J., Edjlali M., Carlier R., Ris L., Lovato A., De Filippis C., Coppee F., Fakhry N., Ayad T., Saussez S. Olfactory and gustatory dysfunctions as a clinical presentation of mild-to-moderate forms of the coronavirus disease (COVID-19): a multicenter European study. Eur. Arch. Otorhinolaryngol. 2020;277(8):2251–2261. doi: 10.1007/s00405-020-05965-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Yan C.H., Faraji F., Prajapati D.P., Boone C.E., DeConde A.S. Association of chemosensory dysfunction and COVID-19 in patients presenting with influenza-like symptoms. Int. Forum Allergy Rhinol. 2020;10(7):806–813. doi: 10.1002/alr.22579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Giacomelli A., Pezzati L., Conti F., Bernacchia D., Siano M., Oreni L., Rusconi S., Gervasoni C., Ridolfo A.L., Rizzardini G., Antinori S., Galli M. Self-reported olfactory and taste disorders in patients with severe acute respiratory coronavirus 2 infection: a cross-sectional study. Clin. Infect. Dis. 2020;71(15):889–890. doi: 10.1093/cid/ciaa330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Henkin R.I., Schmidt L., Velicu I. Interleukin 6 in hyposmia. JAMA Otolaryngol. Head Neck Surg. 2013;139(7):728–734. doi: 10.1001/jamaoto.2013.3392. [DOI] [PubMed] [Google Scholar]
- 80.Turner J.H., Liang K.L., May L., Lane A.P. Tumor necrosis factor alpha inhibits olfactory regeneration in a transgenic model of chronic rhinosinusitis-associated olfactory loss. Am. J. Rhinol. Allergy. 2010;24(5):336–340. doi: 10.2500/ajra.2010.24.3498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Goh Y., Beh D.L.L., Makmur A., Somani J., Chan A.C.Y. Pearls & Oy-sters: Facial nerve palsy in COVID-19 infection. Neurology. 2020;95(8):364–367. doi: 10.1212/WNL.0000000000009863. [DOI] [PubMed] [Google Scholar]
- 82.Wan Y., Cao S., Fang Q., Wang M., Huang Y. Coronavirus disease 2019 complicated with Bell’s palsy: a case report. Research Square. 2020 [Google Scholar]
- 83.Dinkin M., Gao V., Kahan J., Bobker S., Simonetto M., Wechsler P., Harpe J., Greer C., Mints G., Salama G., Tsiouris A.J., Leifer D. COVID-19 presenting with ophthalmoparesis from cranial nerve palsy. Neurology. 2020;95(5):221–223. doi: 10.1212/WNL.0000000000009700. [DOI] [PubMed] [Google Scholar]
- 84.Zhao H., Shen D., Zhou H., Liu J., Chen S. Guillain-Barre syndrome associated with SARS-CoV-2 infection: causality or coincidence? Lancet Neurol. 2020;19(5):383–384. doi: 10.1016/S1474-4422(20)30109-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Virani A., Rabold E., Hanson T., Haag A., Elrufay R., Cheema T., Balaan M., Bhanot N. Guillain-Barre Syndrome associated with SARS-CoV-2 infection. IDCases. 2020;20:e00771. doi: 10.1016/j.idcr.2020.e00771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Alberti P., Beretta S., Piatti M., Karantzoulis A., Piatti M.L., Santoro P., Vigano M., Giovannelli G., Pirro F., Montisano D.A., Appollonio I., Ferrarese C. Guillain-Barre syndrome related to COVID-19 infection. Neurol Neuroimmunol. Neuroinflamm. 2020;7(4) doi: 10.1212/NXI.0000000000000741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Ottaviani D., Boso F., Tranquillini E., Gapeni I., Pedrotti G., Cozzio S., Guarrera G.M., Giometto B. Early Guillain-Barre syndrome in coronavirus disease 2019 (COVID-19): a case report from an Italian COVID-hospital. Neurol. Sci. 2020;41(6):1351–1354. doi: 10.1007/s10072-020-04449-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Pfefferkorn T., Dabitz R., von Wernitz-Keibel T., Aufenanger J., Nowak-Machen M., Janssen H. Acute polyradiculoneuritis with locked-in syndrome in a patient with Covid-19. J. Neurol. 2020;267(7):1883–1884. doi: 10.1007/s00415-020-09897-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Padroni M., Mastrangelo V., Asioli G.M., Pavolucci L., Abu-Rumeileh S., Piscaglia M.G., Querzani P., Callegarini C., Foschi M. Guillain-Barre syndrome following COVID-19: new infection, old complication? J. Neurol. 2020;267(7):1877–1879. doi: 10.1007/s00415-020-09849-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Camdessanche J.P., Morel J., Pozzetto B., Paul S., Tholance Y., Botelho-Nevers E. COVID-19 may induce Guillain-Barre syndrome. Rev. Neurol. (Paris) 2020;176(6):516–518. doi: 10.1016/j.neurol.2020.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Gutierrez-Ortiz C., Mendez-Guerrero A., Rodrigo-Rey S., San Pedro-Murillo E., Bermejo-Guerrero L., Gordo-Manas R., de Aragon-Gomez F., Benito-Leon J. Miller Fisher syndrome and polyneuritis cranialis in COVID-19. Neurology. 2020;95(5):e601–e605. doi: 10.1212/WNL.0000000000009619. [DOI] [PubMed] [Google Scholar]
- 92.Juliao Caamano D.S., Alonso Beato R. Facial diplegia, a possible atypical variant of Guillain-Barre Syndrome as a rare neurological complication of SARS-CoV-2. J. Clin. Neurosci. 2020;77:230–232. doi: 10.1016/j.jocn.2020.05.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Ghiasvand F., Ghadimi M., Ghadimi F., Safarpour S., Hosseinzadeh R., SeyedAlinaghi S. Symmetrical polyneuropathy in coronavirus disease 2019 (COVID-19) IDCases. 2020;21:e00815. doi: 10.1016/j.idcr.2020.e00815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Shors A.R. Herpes zoster and severe acute herpetic neuralgia as a complication of COVID-19 infection. JAAD Case Rep. 2020;6(7):656–657. doi: 10.1016/j.jdcr.2020.05.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Delly F., Syed M.J., Lisak R.P., Zutshi D. Myasthenic crisis in COVID-19. J. Neurol. Sci. 2020;414:116888. doi: 10.1016/j.jns.2020.116888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Jin M., Tong Q. Rhabdomyolysis as potential late complication associated with COVID-19. Emerg. Infect. Dis. 2020;26(7):1618–1620. doi: 10.3201/eid2607.200445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Chen X., Zhao B., Qu Y., Chen Y., Xiong J., Feng Y., Men D., Huang Q., Liu Y., Yang B., Ding J., Li F. Detectable serum SARS-CoV-2 viral load (RNAaemia) is closely correlated with drastically elevated interleukin 6 (IL-6) level in critically ill COVID-19 patients. Clin. Infect. Dis. 2020 doi: 10.1093/cid/ciaa449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Y. Yang, C. Shen, J. Li, J. Yuan, J. Wei, F. Huang, F. Wang, G. Li, Y. Li, L. Xing, L. Peng, M. Yang, M. Cao, H. Zheng, W. Wu, R. Zou, D. Li, Z. Xu, H. Wang, M. Zhang, Z. Zhang, G.F. Gao, C. Jiang, L. Liu, Y. Liu, Plasma IP-10 and MCP-3 levels are highly associated with disease severity and predict the progression of COVID-19, J Allergy Clin Immunol 146(1) (2020) 119-127 e4. [DOI] [PMC free article] [PubMed]
- 99.Ruan Q., Yang K., Wang W., Jiang L., Song J. Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China. Intensive Care Med. 2020;46(5):846–848. doi: 10.1007/s00134-020-05991-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Zhang J., Xie B., Hashimoto K. Current status of potential therapeutic candidates for the COVID-19 crisis. Brain Behav. Immun. 2020;87:59–73. doi: 10.1016/j.bbi.2020.04.046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.T. Bai, S. Tu, Y. Wei, L. Xiao, Y. Jin, L. Zhang, J. Song, W. Liu, Q. Zhu, L. Yang, H. Chen, X. Hou, Clinical and laboratory factors predicting the prognosis of patients with COVID-19: an analysis of 127 patients in Wuhan, China, SSRN Electronic Journal (2020).
- 102.Arabi Y.M., Shalhoub S., Mandourah Y., Al-Hameed F., Al-Omari A., Al Qasim E., Jose J., Alraddadi B., Almotairi A., Al Khatib K., Abdulmomen A., Qushmaq I., Sindi A.A., Mady A., Solaiman O., Al-Raddadi R., Maghrabi K., Ragab A., Al Mekhlafi G.A., Balkhy H.H., Al Harthy A., Kharaba A., Gramish J.A., Al-Aithan A.M., Al-Dawood A., Merson L., Hayden F.G., Fowler R. Ribavirin and interferon therapy for critically ill patients with middle east respiratory syndrome: a multicenter observational study. Clin. Infect. Dis. 2020;70(9):1837–1844. doi: 10.1093/cid/ciz544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Chan M.H., Chan P.K., Griffith J.F., Chan I.H., Lit L.C., Wong C.K., Antonio G.E., Liu E.Y., Hui D.S., Suen M.W., Ahuja A.T., Sung J.J., Lam C.W. Steroid-induced osteonecrosis in severe acute respiratory syndrome: a retrospective analysis of biochemical markers of bone metabolism and corticosteroid therapy. Pathology. 2006;38(3):229–235. doi: 10.1080/00313020600696231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Siddiqi H.K., Mehra M.R. COVID-19 illness in native and immunosuppressed states: a clinical-therapeutic staging proposal. J. Heart Lung Transplant. 2020;39(5):405–407. doi: 10.1016/j.healun.2020.03.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Prokunina-Olsson L., Alphonse N., Dickenson R.E., Durbin J.E., Glenn J.S., Hartmann R., Kotenko S.V., Lazear H.M., O'Brien T.R., Odendall C., Onabajo O.O., Piontkivska H., Santer D.M., Reich N.C., Wack A., Zanoni I. COVID-19 and emerging viral infections: the case for interferon lambda. J. Exp. Med. 2020;217(5) doi: 10.1084/jem.20200653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Ye Q., Wang B., Mao J. The pathogenesis and treatment of the ‘Cytokine Storm' in COVID-19. J. Infect. 2020;80(6):607–613. doi: 10.1016/j.jinf.2020.03.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Broggi A., Ghosh S., Sposito B., Spreafico R., Balzarini F., Lo Cascio A., Clementi N., De Santis M., Mancini N., Granucci F., Zanoni I. Type III interferons disrupt the lung epithelial barrier upon viral recognition. Science. 2020;369(6504):706–712. doi: 10.1126/science.abc3545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Zhang C., Wu Z., Li J.W., Zhao H., Wang G.Q. Cytokine release syndrome in severe COVID-19: interleukin-6 receptor antagonist tocilizumab may be the key to reduce mortality. Int. J. Antimicrob. Agents. 2020;55(5):105954. doi: 10.1016/j.ijantimicag.2020.105954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Richardson P.J., Ottaviani S., Prelle A., Stebbing J., Casalini G., Corbellino M. CNS penetration of potential anti-COVID-19 drugs. J. Neurol. 2020;267(7):1880–1882. doi: 10.1007/s00415-020-09866-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Xie Y., Cao S., Dong H., Li Q., Chen E., Zhang W., Yang L., Fu S., Wang R. Effect of regular intravenous immunoglobulin therapy on prognosis of severe pneumonia in patients with COVID-19. J. Infect. 2020;81(2):318–356. doi: 10.1016/j.jinf.2020.03.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Shao Z., Feng Y., Zhong L., Xie Q., Lei M., Liu Z., Wang C., Ji J., Li W., Liu H., Gu Z., Hu Z., Su L., Wu M., Liu Z. Clinical efficacy of intravenous immunoglobulin therapy in critical patients with COVID-19: a multicenter retrospective cohort study SSRN. Electronic J. 2020 doi: 10.1002/cti2.1192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Y. Zhang, L. Yu, L. Tang, M. Zhu, Y. Jin, Z. Wang, L. Li, A Promising Anti-Cytokine-Storm Targeted Therapy for COVID-19: The Artificial-Liver Blood-Purification System, Engineering, Beijing, 2020. [DOI] [PMC free article] [PubMed]
- 113.Liu X., Zhang Y., Xu X., Du W., Su K., Zhu C., Chen Y., Lei S., Zheng S., Jiang J., Yang S., Guo J., Shao L., Yang Q., Chen J., Li L. Evaluation of plasma exchange and continuous veno-venous hemofiltration for the treatment of severe avian influenza A (H7N9): a cohort study. Ther. Apher. Dial. 2015;19(2):178–184. doi: 10.1111/1744-9987.12240. [DOI] [PubMed] [Google Scholar]
- 114.Leng Z., Zhu R., Hou W., Feng Y., Yang Y., Han Q., Shan G., Meng F., Du D., Wang S., Fan J., Wang W., Deng L., Shi H., Li H., Hu Z., Zhang F., Gao J., Liu H., Li X., Zhao Y., Yin K., He X., Gao Z., Wang Y., Yang B., Jin R., Stambler I., Lim L.W., Su H., Moskalev A., Cano A., Chakrabarti S., Min K.J., Ellison-Hughes G., Caruso C., Jin K., Zhao R.C. Transplantation of ACE2(-) mesenchymal stem cells improves the outcome of patients with COVID-19 Pneumonia. Aging Dis. 2020;11(2):216–228. doi: 10.14336/AD.2020.0228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Liang B., Chen J., Li T., Wu H., Yang W., Li Y., Li J., Yu C., Nie F., Ma Z., Yang M., Xiao M., Nie P., Gao Y., Qian C., Hu M. Clinical remission of a critically ill COVID-19 patient treated by human umbilical cord mesenchymal stem cells: a case report. Medicine (Baltimore) 2020;99(31):e21429. doi: 10.1097/MD.0000000000021429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.D. Wang, B. Hu, C. Hu, F. Zhu, X. Liu, J. Zhang, B. Wang, H. Xiang, Z. Cheng, Y. Xiong, Y. Zhao, Y. Li, X. Wang, Z. Peng, Clinical Characteristics of 138 Hospitalized Patients With 2019 Novel Coronavirus-Infected Pneumonia in Wuhan, China, JAMA (2020). [DOI] [PMC free article] [PubMed]
- 117.Chen T., Wu D., Chen H., Yan W., Yang D., Chen G., Ma K., Xu D., Yu H., Wang H., Wang T., Guo W., Chen J., Ding C., Zhang X., Huang J., Han M., Li S., Luo X., Zhao J., Ning Q. Clinical characteristics of 113 deceased patients with coronavirus disease 2019: retrospective study. BMJ. 2020;368:m1091. doi: 10.1136/bmj.m1091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Avula A., Nalleballe K., Narula N., Sapozhnikov S., Dandu V., Toom S., Glaser A., Elsayegh D. COVID-19 presenting as stroke. Brain Behav. Immun. 2020;87:115–119. doi: 10.1016/j.bbi.2020.04.077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Klok F.A., Kruip M., van der Meer N.J.M., Arbous M.S., Gommers D., Kant K.M., Kaptein F.H.J., van Paassen J., Stals M.A.M., Huisman M.V., Endeman H. Confirmation of the high cumulative incidence of thrombotic complications in critically ill ICU patients with COVID-19: an updated analysis. Thromb. Res. 2020;191:148–150. doi: 10.1016/j.thromres.2020.04.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Sharifi-Razavi A., Karimi N., Rouhani N. COVID-19 and intracerebral haemorrhage: causative or coincidental? New Microbes New Infect. 2020;35:100669. doi: 10.1016/j.nmni.2020.100669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Dogan L., Kaya D., Sarikaya T., Zengin R., Dincer A., Akinci I.O., Afsar N. Plasmapheresis treatment in COVID-19-related autoimmune meningoencephalitis: case series. Brain Behav. Immun. 2020;87:155–158. doi: 10.1016/j.bbi.2020.05.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Al-Olama M., Rashid A., Garozzo D. COVID-19-associated meningoencephalitis complicated with intracranial hemorrhage: a case report. Acta Neurochir (Wien) 2020;162(7):1495–1499. doi: 10.1007/s00701-020-04402-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Duong L., Xu P., Liu A. Meningoencephalitis without respiratory failure in a young female patient with COVID-19 infection in Downtown Los Angeles, early April 2020. Brain Behav. Immun. 2020;87:33. doi: 10.1016/j.bbi.2020.04.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Farhadian S., Glick L.R., Vogels C.B.F., Thomas J., Chiarella J., Casanovas-Massana A., Zhou J., Odio C., Vijayakumar P., Geng B., Fournier J., Bermejo S., Fauver J.R., Alpert T., Wyllie A.L., Turcotte C., Steinle M., Paczkowski P., Cruz C.D., Wilen C., Ko A.I., MacKay S., Grubaugh N.D., Spudich S., Aoun Barakat L. Acute encephalopathy with elevated CSF inflammatory markers as the initial presentation of COVID-19. Res. Sq. 2020 doi: 10.1186/s12883-020-01812-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Franceschi A.M., Ahmed O., Giliberto L., Castillo M. Hemorrhagic posterior reversible encephalopathy syndrome as a manifestation of COVID-19 infection. AJNR Am. J. Neuroradiol. 2020;41(7):1173–1176. doi: 10.3174/ajnr.A6595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Rogg J., Baker A., Tung G. Posterior reversible encephalopathy syndrome (PRES): another imaging manifestation of COVID-19. Interdiscip. Neurosurg. 2020;22:100808. doi: 10.1016/j.inat.2020.100808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Sachs J.R., Gibbs K.W., Swor D.E., Sweeney A.P., Williams D.W., Burdette J.H., West T.G., Geer C.P. COVID-19-associated Leukoencephalopathy. Radiology. 2020;296(3):E184–E185. doi: 10.1148/radiol.2020201753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Valiuddin H., Skwirsk B., Paz-Arabo P. Acute transverse myelitis associated with SARS-CoV-2: a case-report. Brain Behav. Immun. Health. 2020;5:100091. doi: 10.1016/j.bbih.2020.100091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Sarma D., Bilello L.A. A case report of acute transverse myelitis following novel coronavirus infection. Clin. Pract. Cases Emerg. Med. 2020;4(3):321–323. doi: 10.5811/cpcem.2020.5.47937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Bertlich M., Stihl C., Weiss B.G., Canis M., Haubner F., Ihler F. Characteristics of impaired chemosensory function in hospitalized COVID-19 patients. SSRN Electronic J. 2020 [Google Scholar]
- 131.Vaira L.A., Deiana G., Fois A.G., Pirina P., Madeddu G., De Vito A., Babudieri S., Petrocelli M., Serra A., Bussu F., Ligas E., Salzano G., De Riu G. Objective evaluation of anosmia and ageusia in COVID-19 patients: single-center experience on 72 cases. Head Neck. 2020;42(6):1252–1258. doi: 10.1002/hed.26204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.S.H.R. Bagheri, A.M. Asghari, M. Farhadi, A.R. Shamshiri, A. Kabir, S.K. Kamrava, M. alessi, A. Mohebbi, R. Alizadeh, A.A. Honarmand, B. Ghalehbaghi, A. Salimi, Coincidence of COVID-19 epidemic and olfactory dysfunction outbreak, medRxiv (2020). [DOI] [PMC free article] [PubMed]
- 133.Sancho-Saldana A., Lambea-Gil A., Liesa J.L.C., Caballo M.R.B., Garay M.H., Celada D.R., Serrano-Ponz M. Guillain-Barre syndrome associated with leptomeningeal enhancement following SARS-CoV-2 infection. Clin. Med. (Lond) 2020;20(4):e93–e94. doi: 10.7861/clinmed.2020-0213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Sedaghat Z., Karimi N. Guillain Barre syndrome associated with COVID-19 infection: a case report. J. Clin. Neurosci. 2020;76:233–235. doi: 10.1016/j.jocn.2020.04.062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Webb S., Wallace V.C., Martin-Lopez D., Yogarajah M. Guillain-Barré syndrome following COVID-19: a newly emerging post-infectious complication. BMJ Case Rep. 2020;13:e236182. doi: 10.1136/bcr-2020-236182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Toscano G., Palmerini F., Ravaglia S., Ruiz L., Invernizzi P., Cuzzoni M.G., Franciotta D., Baldanti F., Daturi R., Postorino P., Cavallini A., Micieli G. Guillain-barre syndrome associated with SARS-CoV-2. N. Engl. J. Med. 2020;382(26):2574–2576. doi: 10.1056/NEJMc2009191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Ma J., Xia P., Zhou Y., Liu Z., Zhou X., Wang J., Li T., Yan X., Chen L., Zhang S., Qin Y., Li X. Potential effect of blood purification therapy in reducing cytokine storm as a late complication of critically ill COVID-19. Clin Immunol. 2020;214:108408. doi: 10.1016/j.clim.2020.108408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Xu K., Cai H., Shen Y., Ni Q., Chen Y., Hu S., Li J., Wang H., Yu L., Huang H., Qiu Y., Wei G., Fang Q., Zhou J., Sheng J., Liang T., Li L. Management of corona virus disease-19 (COVID-19): the Zhejiang experience. Zhejiang Da Xue Xue Bao Yi Xue Ban. 2020;49(1) doi: 10.3785/j.issn.1008-9292.2020.02.02. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Fang X., Mei Q., Yang T., Li L., Wang Y., Tong F., Geng S., Pan A. Low-dose corticosteroid therapy does not delay viral clearance in patients with COVID-19. J. Infect. 2020;81(1):147–178. doi: 10.1016/j.jinf.2020.03.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Lu X., Chen T., Wang Y., Wang J., Yan F. Adjuvant corticosteroid therapy for critically ill patients with COVID-19. Crit. Care. 2020;24(1):241. doi: 10.1186/s13054-020-02964-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Zhou Z., Xie S., Zhang J., Zheng F., Jiang D., Li K., Zuo Q., Yan Y., Liu J., Xie Y., Peng H., Zhang L. Short-term moderate-dose corticosteroid plus immunoglobulin effectively reverses COVID-19 patients who have failed low-dose therapy. Preprints. 2020:2020030065. [Google Scholar]
- 142.Zha L., Li S., Pan L., Tefsen B., Li Y., French N., Chen L., Yang G., Villanueva E.V. Corticosteroid treatment of patients with coronavirus disease 2019 (COVID-19) Med. J. Aust. 2020;212(9):416–420. doi: 10.5694/mja2.50577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Cavalli G., De Luca G., Campochiaro C., Della-Torre E., Ripa M., Canetti D., Oltolini C., Castiglioni B., Tassan Din C., Boffini N., Tomelleri A., Farina N., Ruggeri A., Rovere-Querini P., Di Lucca G., Martinenghi S., Scotti R., Tresoldi M., Ciceri F., Landoni G., Zangrillo A., Scarpellini P., Dagna L. Interleukin-1 blockade with high-dose anakinra in patients with COVID-19, acute respiratory distress syndrome, and hyperinflammation: a retrospective cohort study. Lancet Rheumatol. 2020;2(6):e325–e331. doi: 10.1016/S2665-9913(20)30127-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Russell B., Moss C., George G., Santaolalla A., Cope A., Papa S., Van Hemelrijck M. Associations between immune-suppressive and stimulating drugs and novel COVID-19-a systematic review of current evidence. Ecancermedicalscience. 2020;14:1022. doi: 10.3332/ecancer.2020.1022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Huet T., Beaussier H., Voisin O., Jouveshomme S., Dauriat G., Lazareth I., Sacco E., Naccache J.M., Bezie Y., Laplanche S., Le Berre A., Le Pavec J., Salmeron S., Emmerich J., Mourad J.J., Chatellier G., Hayem G. Anakinra for severe forms of COVID-19: a cohort study. Lancet Rheumatol. 2020;2(7):e393–e400. doi: 10.1016/S2665-9913(20)30164-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Aouba A., Baldolli A., Geffray L., Verdon R., Bergot E., Martin-Silva N., Justet A. Targeting the inflammatory cascade with anakinra in moderate to severe COVID-19 pneumonia: case series. Ann. Rheum. Dis. 2020 doi: 10.1136/annrheumdis-2020-217706. [DOI] [PubMed] [Google Scholar]
- 147.Xu X., Han M., Li T., Sun W., Wang D., Fu B., Zhou Y., Zheng X., Yang Y., Li X., Zhang X., Pan A., Wei H. Effective treatment of severe COVID-19 patients with tocilizumab. Proc. Natl. Acad. Sci. USA. 2020;117(20):10970–10975. doi: 10.1073/pnas.2005615117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Wu D., Yang X.O. TH17 responses in cytokine storm of COVID-19: an emerging target of JAK2 inhibitor Fedratinib. J. Microbiol. Immunol. Infect. 2020;53(3):368–370. doi: 10.1016/j.jmii.2020.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Atluri S., Manchikanti L., Hirsch J.A. Expanded Umbilical Cord Mesenchymal Stem Cells (UC-MSCs) as a therapeutic strategy in managing critically ill COVID-19 patients: the case for compassionate use. Pain Physician. 2020;23(2):E71–E83. [PubMed] [Google Scholar]
- 150.Duret P.M., Sebbag E., Mallick A., Gravier S., Spielmann L., Messer L. Recovery from COVID-19 in a patient with spondyloarthritis treated with TNF-alpha inhibitor etanercept. Ann. Rheum. Dis. 2020;79(9):1251–1252. doi: 10.1136/annrheumdis-2020-217362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.R. Han, Y. Wang, M. Dabbous, S. Liang, T. Qiu, M. Toumi, Chinese Clinical Studies for Pharmacological Treatments of Coronavirus Disease 2019 (COVID-19), Preprints (2020) 2020040279.
- 152.Huang H., Hu P.F., Sun L.L., Guo Y.B., Wang Q., Liu Z.M., Yin J.Z., Shi P.M., Yuan Z.L., Tan Y., Zhou C., Liu Y.L., Chen C., Song H.H., Xie W. Treatment of Covid-19 patients with high dose of ulinastatin. Research Squar. 2020 [Google Scholar]