Summary
Organoids (ORGs) are increasingly used as models of cerebral cortical development. Here, we compared transcriptome and cellular phenotypes between telencephalic ORGs and monolayers (MONs) generated in parallel from three biologically distinct induced pluripotent stem cell (iPSC) lines. Multiple readouts revealed increased proliferation in MONs, which was caused by increased integrin signaling. MONs also exhibited altered radial glia (RG) polarity and suppression of Notch signaling, as well as impaired generation of intermediate progenitors, outer RG, and cortical neurons, which were all partially reversed by reaggregation of dissociated cells. Network analyses revealed co-clustering of cell adhesion, Notch-related transcripts and their transcriptional regulators in a module strongly downregulated in MONs. The data suggest that ORGs, with respect to MONs, initiate more efficient Notch signaling in ventricular RG owing to preserved cell adhesion, resulting in subsequent generation of intermediate progenitors and outer RG, in a sequence that recapitulates the cortical ontogenetic process.
Keywords: iPSCs, organoids, RNA-seq, human, proteomics, network analyses, cerebral cortex, Notch signaling
Highlights
-
•
Organoid’s radial glia cells engage efficient Notch signaling
-
•
Monolayer hyperproliferation is due to increased integrin signaling
-
•
Neurogenesis' arrest by cell dissociation is partially reversed with reaggregation
In this research article, Scuderi and colleagues generated organoids and monolayers from three iPSC lines and characterized their transcriptomes, proteomes, and cellular architecture. Disorganized neural stem cell polarity in monolayers impaired Notch signaling and decreased a complex transcription factor cascade involved in the generation of intermediate neuronal progenitors and outer radial glia, disrupting cortical neuron differentiation.
Introduction
Induced pluripotent stem cells (iPSCs), generated by somatic cell reprogramming (Takahashi et al., 2007; Yu et al., 2007), hold considerable promise for fundamental biological studies into early human brain development (Ardhanareeswaran et al., 2017; Arlotta and Pasca, 2019; Sidhaye and Knoblich, 2021). In general, two models have been used to achieve in vitro neuronal differentiation. In the first, iPSCs are used to generate neuroepithelial progenitor cells (NPCs) that are then dissociated into single cells and differentiated into neurons in monolayer (MON) preparations (Chambers et al., 2009; Shi et al., 2012). In the second, NPCs grow and differentiate in the context of organoid (ORG) structures in three-dimensional (3D) conditions (Camp et al., 2015; Eiraku et al., 2008; Lancaster et al., 2013; Mariani et al., 2012, 2015).
MONs are a simpler way to differentiate human neurons, but these cells are not able to segregate in layers and compartments as occurs in normal development, and the underlying mechanism of this failure is not known. In contrast, ORG preparations have a spontaneous self-organizing ability to form a neuroepithelium layer where cell polarity, membrane contacts, and morphogen gradients are maintained. Under appropriate culture conditions, proliferating progenitors within ORGs sequentially generate early- and late-born cortical neurons as well as glial cells. A key difference between MON and ORG preparations is differential contact between cells and the extracellular matrix (ECM), which are processes that are known to affect a variety of cellular behaviors, including cell growth, differentiation, and motility (Engler et al., 2006; Saha et al., 2008). Yet, differences in stem cell differentiation abilities under MON and 3D conditions have not been directly compared. Here, we analyze the effects of cell dissociation into MONs versus continuous growth in 3D conditions upon the processes that lead human iPSCs to differentiate into telencephalic neurons. To avoid confounds created by differences in genetic background and reprogramming methods, three iPSC lines were differentiated into neurons as ORGs or MONs in parallel and exposed to identical culture media and conditions. Immunocytochemical, transcriptome, and proteome analyses revealed enduring, long-term differences in neuron specification and differentiation among the two conditions. Reversal experiments reaggregating the MONs and culturing them under 3D conditions corrected excessive cell proliferation and some, but not all, differences in cell fate and gene expression between MONs and ORGs. By varying the timing of dissociation we also found that, while cell-to-cell contacts exert an enduring influence on regional specification and neuronal differentiation of human stem cell precursors, late dissociation seems to have less deleterious effects upon reaggregation.
Results
Cell Dissociation and MON Culture Impact Cellular Organization and Fate
To understand how cell dissociation and the microenvironment might affect the emergence of cortical identity, we compared the transcriptome, proteome, and cell fate of three iPSC lines derived from adult control males differentiated in parallel under two separate conditions: undissociated ORG and MON cultures (Table S1).
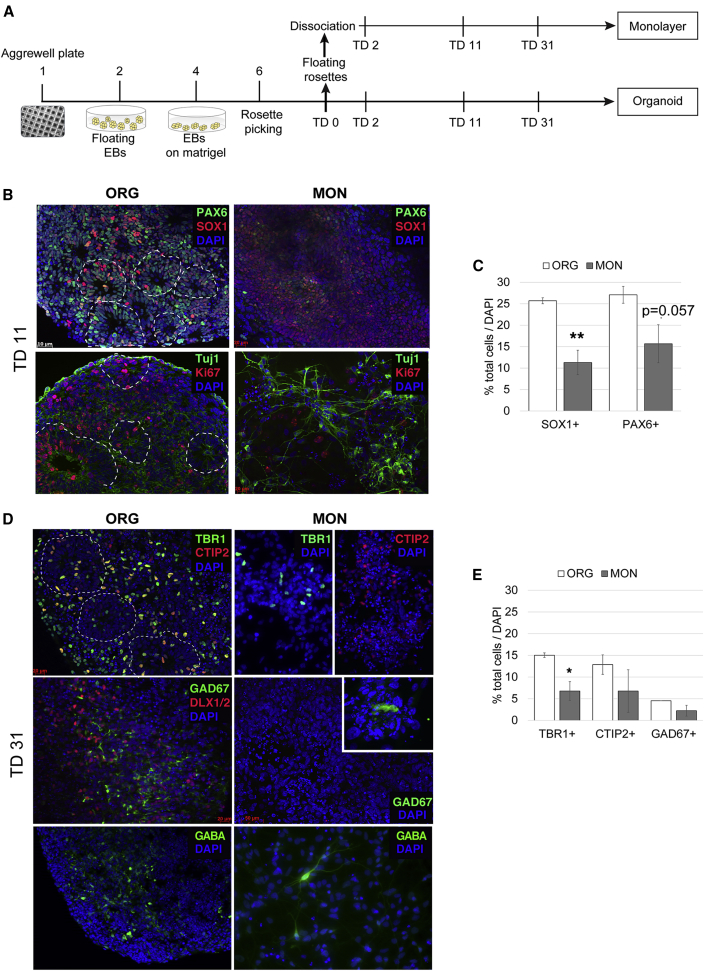
Telencephalic ORGs were prepared (Mariani et al., 2015) using Noggin as neuralizing agent, by aggregating iPSCs into embryoid bodies (EBs), patterning the EBs by BMP, transforming growth factor β, and Wnt inhibition, and culturing the EB-derived neuroepithelium under 3D conditions. The MONs were prepared by dissociating the ORGs at terminal differentiation day 0 (TD0) (which is the day when ORGs are switched into mitogen-free medium) and plating the NPCs on plastic permanox slides coated with poly-L-ornithine-laminin. Both ORG and MON preparations were cultured in parallel in the same medium until analysis (Figure 1A).
Figure 1.
Comparison of 2D versus 3D In Vitro Cell Cultures
(A) Experimental design. TD, terminal differentiation day; EB, embryoid bodies; MON, monolayer; ORG, organoid.
(B) Representative images of immunocytochemical staining with the dorsal telencephalic marker PAX6, the neuroectodermal marker SOX1, the proliferative marker Ki67, and the neuron-specific class III β-tubulin at TD11.
(C) Proportion of SOX1+ and PAX6+ cells by stereological quantification over DAPI+ nuclei.
(D) Immunocytochemical staining of excitatory (TBR1+ and CTIP2+) and inhibitory (GABA+, GAD67+) cortical neurons at TD31.
(E) Proportion of excitatory and inhibitory neurons over DAPI+ nuclei assessed by stereological analysis. Results in (C and E) are the mean ± SEM of n = 3 biologically different iPSC lines per condition (ORG, MON) differentiated in parallel with two technical replicates per cell line.
∗∗p < 0.01, ∗p < 0.05 Student's t test, two tailed. See also Figure S1.
After 11 days in terminal differentiation medium (TD11), the undissociated ORG preparation displayed layers of polarized radial glial (RG) cells expressing the cortical neural progenitor markers SOX1 and PAX6 (Figure 1B). The cell-cycle marker Ki67 was mostly located in the RG cell layer, with cells undergoing mitosis at the apical edge, whereas β3-tubulin+ young neurons were located on the basal side of this layer. In MONs, cells were not polarized or organized into layers. The number of SOX1+ RG cells in MONs was decreased as compared with ORGs (12% ± 3.49% in MONs versus 25% ± 0.69% in ORGs). A comparable trend was observed for PAX6 (Figure 1C). In conclusion, dissociation resulted in disorganized morphology and cellular architecture, with poor expression of RG markers when compared with undissociated ORGs.
On TD31, ORGs displayed a consistent ability to generate TBR1+ (layer 6) and CTIP2+ (layer 5) cortical neurons across the different lines, in agreement with previous results (Mariani et al., 2015). In contrast, the MONs displayed lower and highly variable counts of TBR1+ and CTIP2+ cortical neurons (Figures 1D and 1E). Similarly, we observed a lack of consistent GABAergic neuronal differentiation in MONs, as shown by very low levels of GAD1/GAD67 or GABA immunostaining, as opposed to a reproducible number of GAD67+ inhibitory neurons (about 5% of all cells) in ORGs.
To assess differential proliferative activity, we immunostained ORGs and MONs for Ki67, a nuclear protein expressed only during cell division. At TD2, there were 45.65% ± 5.06% Ki67+ in MONs compared with 19.69% ± 1.64% in ORGs; the increase was highly significant (p < 0.001) (Figure S1). At TD11, no significant differences in the number of Ki67+ cells were observed between the conditions. These findings suggest a short-term proliferative effect induced by dissociation in the MON cultures.
Distinct Transcriptional Trajectories of ORG and MON Preparations
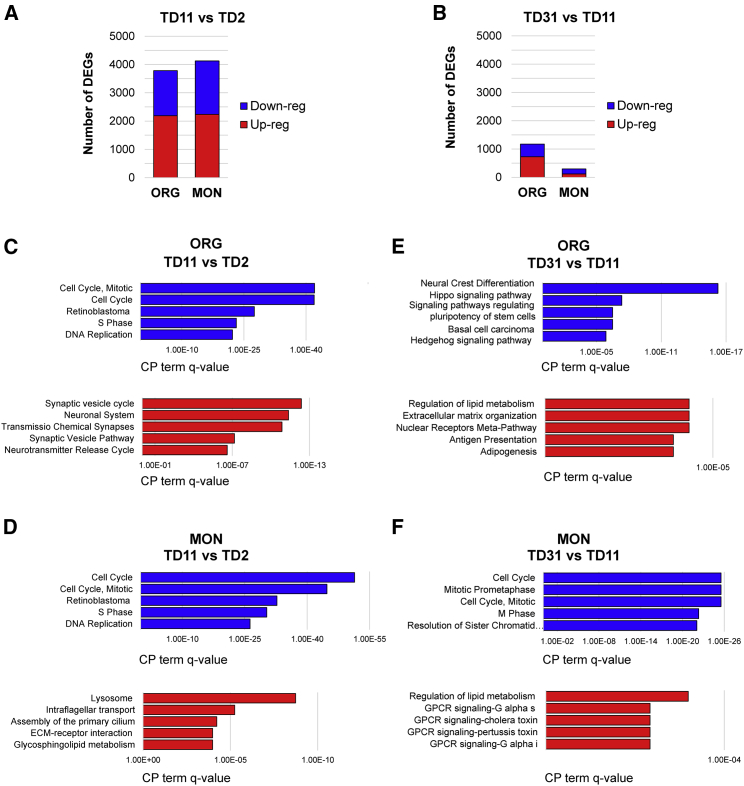
To study each system's individual developmental trajectory, we investigated the temporal evolution of the ORG and MON transcriptome at TD2, TD11, and TD30. For a list of differentially expressed genes (DEGs) across all transitions and their gene ontology (GO) and canonical pathway (CP) annotations (see Table S2).
At TD11 versus TD2, we identified 3,780 and 4,126 DEGs for ORG and MON conditions, respectively (Figure 2A). CP annotations for downregulated genes were mostly related to mitosis and cell cycle (e.g., cyclins CDC2 and CDC25) for both ORGs and MONs (Figures 2C and 2D), reflecting the shared capability of escaping cell division for both systems. Top CP terms in ORGs for upregulated genes were related to synaptic formation and neurotransmitter release, synaptic vesicles, and calcium channel genes. In contrast, the top CP terms for upregulated genes in MONs were related to lysosome, intraflagellar transport, cilium formation, and ECM molecules and receptor interactions (e.g., LAMA2 and COL1A).
Figure 2.
Longitudinal Differential Gene Expression Analysis in ORGs and MONs
(A and B) Number of differentially expressed genes (DEGs) at the first transition (TD11 versus TD2) (A) and at the second transition (TD31 versus TD11) (B) within each model. Blue, downregulated genes; red, upregulated genes.
(C–F) Canonical pathway (CP) term enrichment for genes upregulated (red) and downregulated (blue) per transition as indicated: (C and D) TD11 versus TD2; (E and F) TD31 versus TD11. The x axis indicates FDR-corrected p value in reverse order. For full annotation see Table S2. Results are from between two and three biologically different iPSC lines differentiated in parallel.
At the second transition, TD31 versus TD11, we identified 1,175 and 296 DEGs for ORG and MON conditions, respectively (Figure 2B). Thus, the MON appears to be a relatively static condition, whereas the ORGs keep evolving transcriptionally. Notably, functional analysis of downregulated genes at this transition was still related to cell cycle in the MONs, whereas in ORGs they were related to neural crest differentiation (comprising a number of WNT pathway genes and WNT-targeted transcription factors [TFs]), Hippo signaling, and signaling pathways regulating pluripotency of stem cells (Figure 2E).
Relative expression levels across samples for 34 randomly selected human genes as revealed by RNA sequencing (RNA-seq) were generally concordant with those obtained by qRT-PCR of poly(A) RNA, thus validating the RNA-seq data (Table S3).
Collectively, the data suggest that ORGs downregulate pluripotency genes and WNT signaling to progressively differentiate into telencephalic neurons, with dynamic up- and downregulation of relevant genes, including neurotransmitter- and synapse-related genes and TFs, regulating cell fate, whereas the regional identity of the MON model is much less defined.
Telencephalic Patterning and Neuronal Fate Genes Are Downregulated in MON Cultures
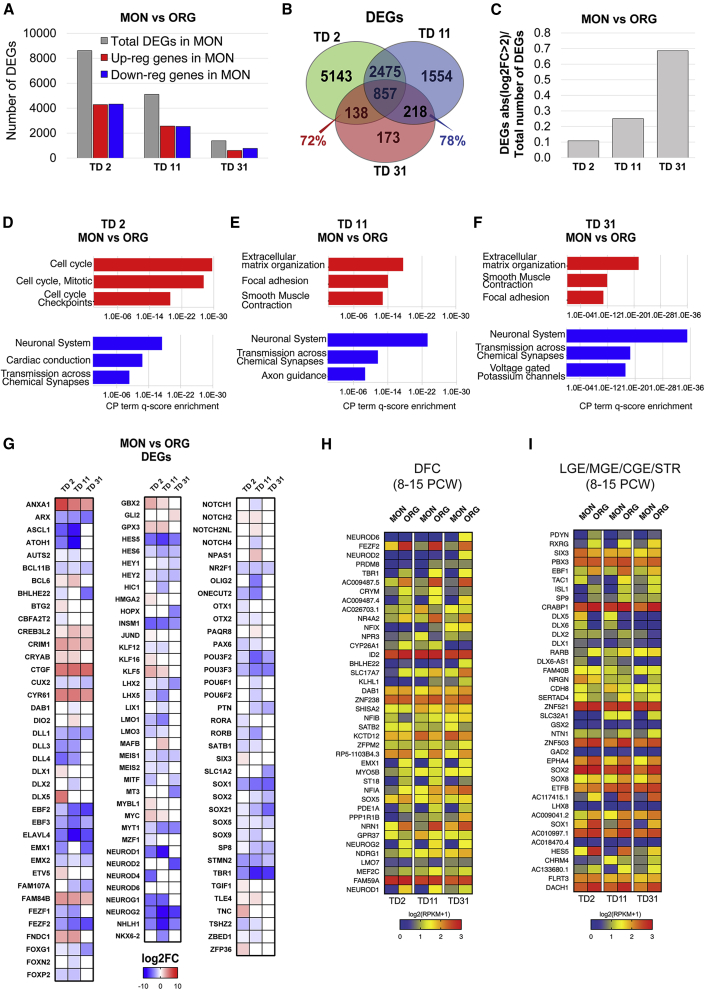
Next, we compared differential gene expression between the isogenic MON and the ORG preparations at each time point, TD2, TD11, and TD31. The DEGs and their GO and CP annotations are listed in Table S3. ORG and the MON DEGs decreased with time (Figure 3A), suggesting a possible convergence of the two systems. More detailed analyses revealed that, whereas the majority of the DEGs present at TD2 were transient and not shared at later time points, 72% and 78% of the DEGs between MONs versus ORGs at TD31 were also present, respectively, at TD2 and TD11 (Figure 3B). In contrast, the relative number of strong transcriptional alterations (i.e., abs (log2 fold change) > 2) increased with time (Figure 3C), suggesting that MONs and ORGs evolve along divergent trajectories.
Figure 3.
Comparison of MON and ORG Transcriptional Profiles at Three Different Stages of Neuronal Differentiation (TD2, TD11, and TD31)
(A) Total number of DEGs (gray bar), downregulated DEGs (blue bar), and upregulated DEGs (red bar) in MONs versus ORGs.
(B) Venn diagram of DEGs in MONs versus ORGs at each time point.
(C) Ratio between the number of highly differentially expressed genes (absolute value [log2 fold change > 2]) and the total number of DEGs in MONs versus ORGs along the time course.
(D–F) CP term enrichment for genes upregulated (red) and downregulated (blue) in MONs versus ORGs at each time point, TD2 (D), TD11 (E), and TD31 (F), with the FDR-corrected p value in reverse order on the x axis. For full annotation see Table S3.
(G) Heatmap displaying the log2 fold change values of transcripts from the Neuronal Cell Fate sublist (Table S4A) differentially expressed in MONs versus ORGs.
(H and I) Expression level (log2 (RPKM +1)) in MONs and ORGs of genes that are highly expressed in human dorsolateral prefrontal cortex (DFC) (H) versus basal telencephalon (lateral ganglionic eminence, medial ganglionic eminence, caudal ganglionic eminence, and striatum) or highly expressed in basal telencephalon versus DFC (I).
We next explored the biological identities of the two systems. For downregulated genes in MONs with respect to ORG, GO and CP enrichment analysis revealed similar top annotations at all time points, i.e., neuronal system, axon guidance, neurogenesis, and synaptic transmission. In contrast, top CP annotation terms for upregulated genes were cell cycle at TD2, and ECM organization and focal adhesion at both TD11 and TD31 (Figures 3D–3F).
To better understand these signatures, we constructed a manually curated list of genes which we call Neurodevelopmental Genes and subdivided it into relevant sublists according to specific criteria (see the Experimental Procedures): Neuronal Cell Fate (168 genes), Cell Adhesion (247 genes), Axon Guidance (175), and Extracellular Matrix (86 genes) (Tables S4A–S4D). Overall, 66% of genes in the Neuronal Cell Fate sublist were downregulated in MONs versus ORGs (Figure 3G) at most of the time points considered. This is consistent with the failed upregulation of neuronal determinants in MONs, noted earlier in the longitudinal analysis. Interestingly, genes of the Notch pathway, which regulate neurogenesis from RG cells, were uniformly downregulated in MONs as compared with ORGs, including Notch ligands (DLL1, DLL3, and DLL4), Notch receptors (NOTCH1 and NOTCH4), downstream transcriptional regulators (HES5, HEY1 and HEY2), and downstream basic-helix-loop-helix neurogenic genes, which are the earliest determinants of dorsal and ventral telencephalic fates (ASCL1, NEUROG1, NEUROG2, NEUROD1, NEUROD2, NEUROD4, and NEUROD6). Among the genes significantly downregulated in MONs versus ORGs were a large number of homeodomain genes and other TFs that pattern the telencephalon (LHX2, PAX6, EMX1, EMX2, and FOXG1), specify pyramidal neuron fates (TBR1, CTIP2, and POU3F2) and GABAergic neuron fates (ASCL1, DLX2, and FOXG1) (Figure 3G).
We then examined differential gene expression in the Cell Adhesion sublist of genes, encompassing cell-to-cell adhesion molecules. A large spectrum of genes of the IgCAM family (e.g., N-CAM and NCAM1), cadherin and protocadherin (CDH2, CDH22, CDH7, and PCDH10), neurexins (NRXN1, NRXN2, and NRXN3), and contactin (e.g., CNTN2 and CNTN3) superfamilies, were downregulated in the MONs versus ORGs at all time points, as well as genes in the Axon Guidance sublist, such as EPH/ephrins (Figures S2A, S2B, and S3A). By comparison with lists of genes that are expressed in the dorsal frontal cortex and basal telencephalon of human fetal brains (see the Experimental Procedures), ORGs exhibited a clear dorsal and ventral telencephalic signature, most evident at TD31, and no clear shift in fate was observed in MONs compared with ORGs (Figures 3H and 3I).
The very first cells that express neurogenic determinants in the developing cortex are RG daughter cells that delaminate from the apical layer to form the intermediate precursor cell (IPC) layer and the outer RG cell layer, which are much expanded in the human cortex (Johnson et al., 2015; Pollen et al., 2015). Hence, we further investigated the RG subtype composition in MONs and ORGs by examining the expression of key marker genes for various types of RG (Nowakowski et al., 2017) in our dataset. While we observed increased expression levels of some ventricular RG (vRG) transcripts (e.g., ANXA1, CTGF, and CYR61) and truncated RG (tRG) transcripts (CRYAB and GPX3) in the MONs compared with the ORGs, there was a strong downregulation of outer RG-specific transcripts (FAM107A, HOPX, MT3, and PTN) in MONs, particularly at the later time points (Figure S3C), suggesting failed evolution of neurogenic RG diversity and fate in MONs.
Increased Integrin Signaling in MON Cultures Triggers Cell Proliferation
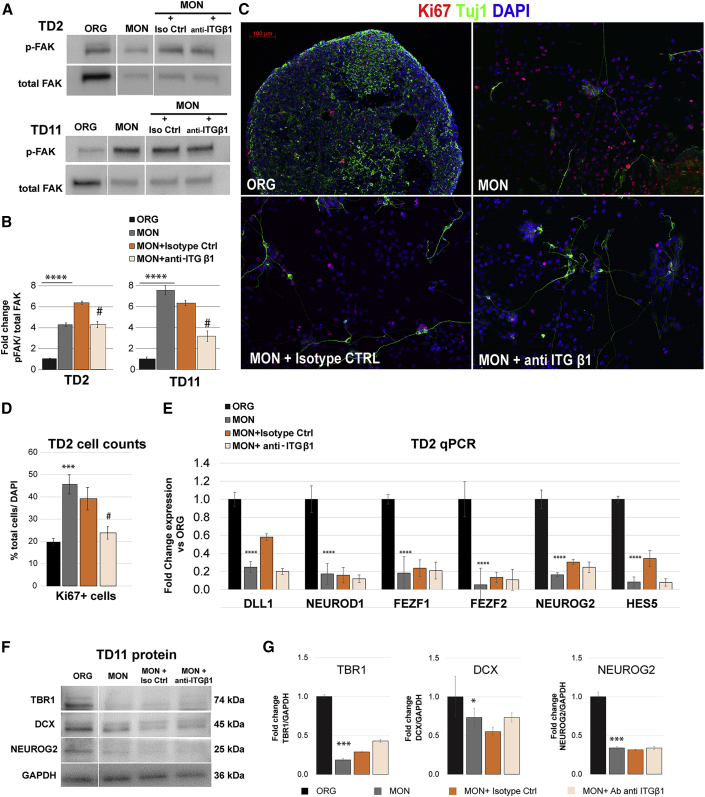
Next, we examined DEGs in the Extracellular Matrix sublist of the curated Neurodevelopmental Genes list (Table S4D). In contrast to cell-to-cell adhesion molecules, there was a strong overexpression of major ECM components in MONs compared with ORGs (Figure S4A), which is concordant with top CP term enrichment for genes upregulated in MONs at both TD11 and TD31 (Figures 3E and 3F). This overexpression was most pronounced for laminins and integrins (e.g., ITGA1, ITGA5, ITGB1, ITGB5, and ITGB6). Integrins are αβ heterodimeric ECM receptors where β1 subunits (ITGβ1) form non-covalent complexes with many α subunits that bind laminin in the CNS (Milner and Campbell, 2002). Consistently, integrin signaling was strongly upregulated at both TD2 and TD11 in MONs, as compared with ORGs, by western blot analyses for phospho-FAK (p-FAK), an adherens junction protein that localizes at sites of integrin receptor accumulation and becomes phosphorylated at different tyrosine residues after engagement of integrin with ECM proteins (Figures 4A and 4B).
Figure 4.
β1 Integrin Signaling Increases MON Cell Proliferation at Early Stages of Differentiation
(A and B) Western blot analysis (A) and quantification of protein expression level (B) of phospho-FAK in ORG, MON and MON treated with either an isotype control antibody (Iso Ctrl) or an anti-β1-integrin antibody (anti- β1ITG) at TD2 and TD11.
(C and D) Representative images (C) and stereological quantification (D) of immunostaining with the proliferative marker Ki67 and the neuron-specific marker TUJ1 at TD2 under the conditions described above.
(E) Relative expression level of a subset of genes from the Neuronal Cell Fate list (Table S4A) at TD2.
(F and G) Western blot analysis (F) and quantification of protein expression level (G) of TBR1, DCX, and NEUROG2 at TD11. GAPDH was used as loading control.
Data are expressed as mean ± SEM of n = 3 preparations per condition (ORGs or MONs with or without each antibody) from one iPSC line. ∗p < 0.05, ∗∗∗p < 0.01, ∗∗∗∗p < 0.001; MONs versus ORGs; #p < 0.05 MONs + anti-ITGβ1 versus MONs + Isotype Ctrl. One-way ANOVA with Tukey multiple comparisons test. See also Figure S4.
Integrin signaling has been involved in the proliferation of neural precursor cells (Long et al., 2016). To test the role of these ECM proteins in the regulation of neuronal stem cell and precursor behavior, we performed an independent experiment where we differentiated one iPSC line into MONs and ORGs with three separate technical replicates each. The MON cultures were treated with a monoclonal β1-integrin-blocking antibody that was previously demonstrated to block integrin function (Iba et al., 2000). In the majority of integrin receptor heterodimers, ITGβ1 is the major mediator of signaling pathways sensing microenvironments rich in laminin. We first confirmed that the p-FAK protein level was decreased upon treatment of MON cultures with the anti-ITGβ1 antibody, both at TD2 and TD11 (Figures 4A and 4B), as compared with an isotype control antibody. We then assessed whether integrin blockade was able to revert the increased proliferation in MONs. As compared with the isotype control antibody condition, in which the number of proliferative cells was similar to that in medium alone (39.20% ± 2.84% and 45.65% ± 5.06%, respectively), blocking ITGβ1 at TD2 significantly reduced the percentage of proliferative cells in the MONs to values close to the ORG preparation (23.38% ± 0.9%) (Figures 4C and 4D).
To understand the potential effect of ITGβ1 signaling on neuronal differentiation, we then assessed levels of several cell fate- and neurogenesis-related transcripts by qPCR, whose expression levels were lower in MONs as opposed to the ORGs in the RNA-seq experiments. We confirmed that MONs had a several-fold decrease in the expression of FEZF2, FEZF1, NEUROG2, NEUROD1, DLL1, and HES5 transcripts with respect to ORGs. However, the expression level of these transcripts did not change in the MONs as a consequence of blocking ITGβ1, either at the RNA or the protein level (Figures 4E–4G). Together, these data suggest that the increase in proliferation in MONs is triggered by integrin signaling, whereas the neurogenesis defect in MONs is not. Furthermore, the increased proliferation and decreased production of cortical neurons in the MON condition are independent from each other as they can be uncoupled by manipulating integrin signaling.
Differences in Neurogenic Cell Fate Are Partially Reversed by Reaggregation
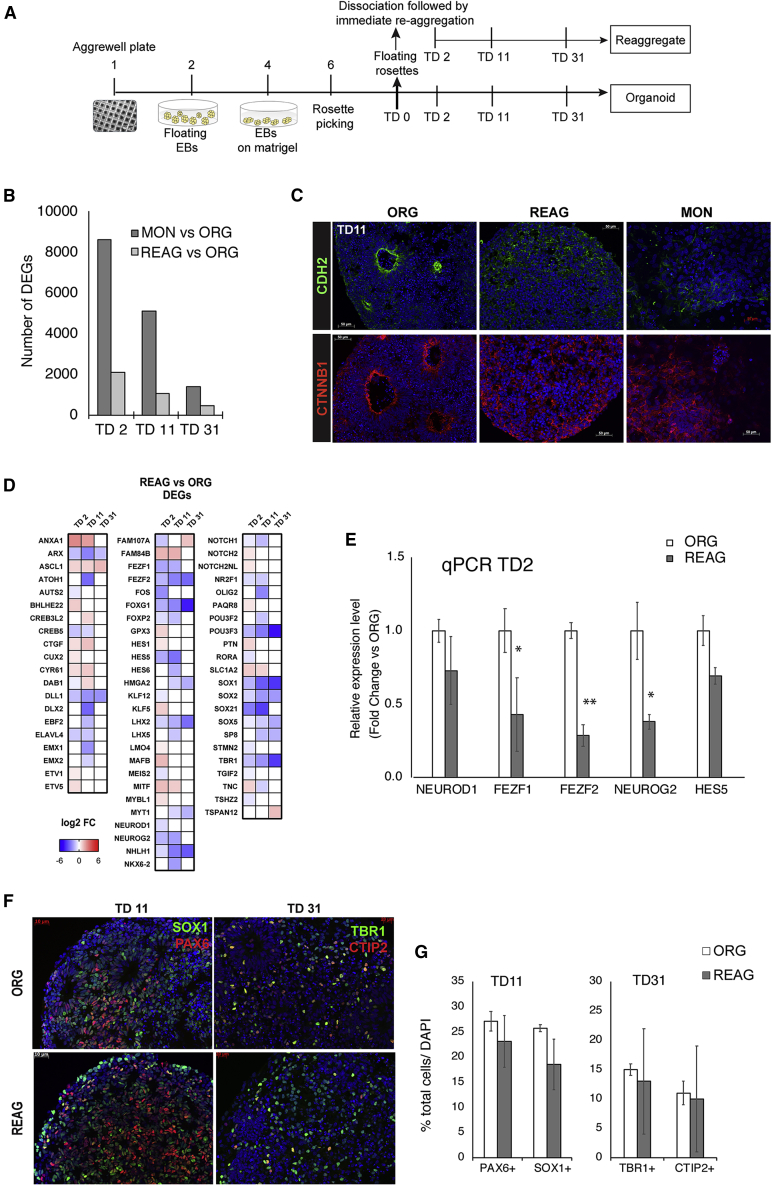
We reasoned that decreased cortical neurogenesis in MONs could be attributable to decreased cell-to-cell contact, rather than increased cell-to-substrate adhesion. To assess whether cell-to-cell interactions per se were responsible for the failure of MONs to properly differentiate, we performed an experiment in which a portion of the dissociated NPCs were immediately re-aggregated and cultured in 3D in parallel to the MON and ORG preparations, using the same TD medium. We call this preparation a re-aggregated culture (REAG) (Figure 5A).
Figure 5.
Dissociation followed by Immediate Reaggregation
(A) Experimental design.
(B) Number of DEGs, at each time point, for the comparisons REAGs versus ORGs and MONs versus ORGs.
(C) Representative images of immunocytochemical staining for N-cadherin (CDH2, green) and β-catenin (CTNNB1, red) at TD11 in ORG, REAGs, and MON preparations (DAPI+ nuclei in blue).
(D and E) Heatmap showing the log2 fold change (D) and bar graph of mRNA expression level by qPCR (E) of DEGs from the Neuronal Cell Fate gene sublist (Table S4A).
(F and G) Representative images of immunocytochemical staining with neuronal progenitor markers (PAX6, SOX1), and the excitatory cortical neuron markers TBR1 and CTIP2 (F) with proportion of different cell types assessed by stereological analysis (G). Results of RNA-seq analysis are from n = 2 biologically different iPSC lines per condition (ORG, REAG) differentiated in parallel.
Immunocytochemical data are expressed as mean ± SEM of n = 3 biologically different iPSC lines per condition (ORG, REAG). Two technical replicates per cell line were analyzed. ∗∗p < 0.01, ∗p < 0.05, MONs versus ORGs analyzed by t test, two tailed. See also Table S5.
We then compared the transcriptomes of REAGs and ORGs (the lists of DEGs, and their GO and CP annotations, are shown in Table S5). In comparison to MONs versus ORGs, the number of DEGs at TD2 and TD11 was much less pronounced in REAGs versus ORGs (Figure 5B). However, the number of DEGs at TD30 were comparable with those in MON versus ORG preparations, and functional enrichment analysis of the DEGs in REAGs versus ORGs still revealed similar top GO and CP annotations as for the DEGs in MONs versus ORGs, i.e., neurogenesis and neuronal differentiation. Interestingly, in the Extracellular Matrix sublist, 66% of the genes were differentially expressed in MONs versus ORGs and only 24% in REAGs versus ORGs (Figures S4A and S4B; Table S4D), suggesting that the overexpression of ECM transcripts noted in MONs were partly compensated by reaggregation. Correspondingly, no changes in FAK phosphorylation were noted in the REAG versus ORG comparison (Figures S4C–S4F). Confirming the causal relationship between integrin signaling and cell proliferation, the increase in cell proliferation previously noted in MONs versus ORGs was no longer evident when REAG cultures were compared with ORGs (19.69% ± 1.64% in ORGs and 20.24% ± 4.31% in REAGs at TD2, Figure S1). Hence, the prompt reaggregation after dissociation prevents the upregulation in ITG signaling as well as other dissociation-induced short-term transcriptional alterations.
IgCAM family members (cadherin and protocadherin) were also partially restored under the REAG conditions. In the Cell Adhesion sublist, 51% of the genes were differentially expressed in MONs versus ORGs and only 20% in REAGs versus ORGs (Figures S2A–S2D). However, immunocytochemical analyses showed mislocalization of N-cadherin (CDH2) protein to a similar extent in both MONs and REAGs, as compared with ORGs where CDH2 is strictly localized in the apical RG cell feet (Figure 5C). This mislocalization was concomitant with a partial disruption in cell polarity of the neuroepithelium and a lack of segregation between layers of dividing progenitors and postmitotic neurons. The apical-basal polarity loss after dissociation was confirmed by absence of β-catenin at the apical endfeet of RG in both REAGs and MONs (Figure 5C). Progenitor cells did express SOX1 and PAX6, but the RG layer was thinner as compared with the ORG preparation, with TBR1+, CTIP2+, and β3-tubulin+ neurons dispersed throughout rather than being excluded from the RG progenitor layer (Figures 5F and S1). There appeared to be some improvement in cortical cell fate with respect to MONs, as the number of DEGs in the Neuronal Cell Fate sublist was 66% in MONs versus ORGs and only 35% in REAGs versus ORGs (Figure 5D, Table S4A). In addition there was a recovery in expression of axon guidance transcripts (Figures S3A and S3B) and outer RG gene markers in REAGs (Figures S3C and S3D). However, the expression of some Notch-related transcripts (NOTCH1, DLL1, and HES5), telencephalic neurogenic genes (NEUROG2 and NEUROD1), and key telencephalic cell fate genes, such as FOXG1, LHX2, FEZF1/2, EMX1, TBR1, and EOMES, were still downregulated in REAGs versus ORGs, both by RNA-seq and qPCR (Figures 5D and 5E). Immunocytochemical characterization of the REAG cultures revealed persistent variability in cortical cell fate, as determined by counts of cortical neuron precursors expressing the layer-specific TFs TBR1 and CTIP2 (Figures 5F and 5G). Although without ultrastructural data we cannot ensure total restoration of membrane contacts, the data suggest that not cell adhesion per se, but proper orientation and polarity of cell-to-cell contacts, which was not entirely restored in the REAGs, must be responsible for persistent neurogenic defects in REAG preparations.
Gene Network Analyses Suggest Relationships between Cell Adhesion and Cell Fate
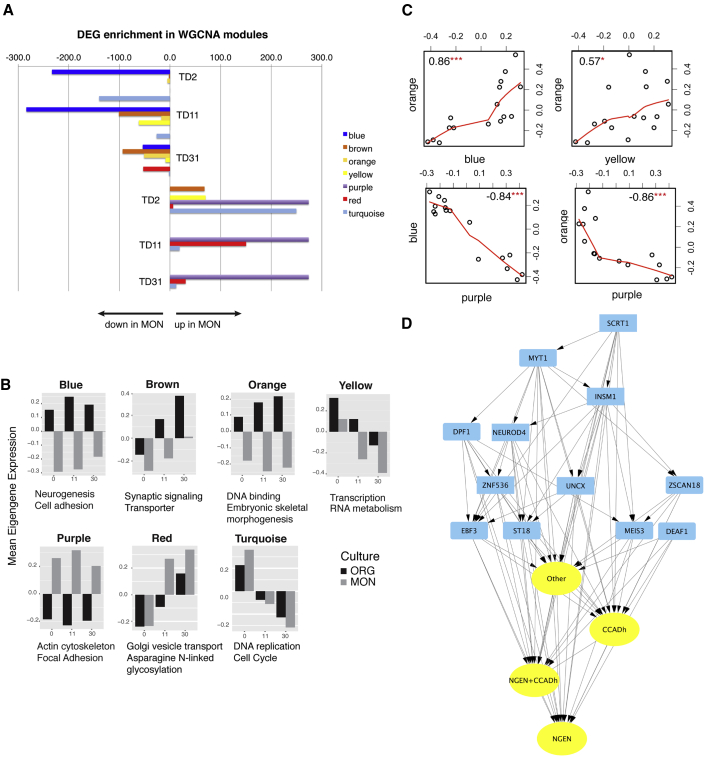
To obtain additional insights into the proposed interplay between cell-to-cell adhesion molecules and disruption of neural cell fate, we applied weighted gene co-expression network analysis (WGCNA) (Langfelder and Horvath, 2008) to the full set of MON and ORG samples. We identified 42 co-expression modules (Table S5A), all of which survived robustness testing. We found that 35 of the 42 modules are enriched in DEGs for MONs versus ORGs at one or more time points (Table S6B), and seven modules (blue, brown, orange, purple, red, turquoise, and yellow) are strongly enriched in DEGs at all three time points (more than 20 genes and false discovery rate [FDR] < 0.05). Among those, the blue, brown, orange, and yellow are enriched in DEGs mostly downregulated in MONs, whereas the purple, red, and turquoise are enriched in DEGs mostly upregulated in MONs (Figure 6A). Most of the modules downregulated in MONs were annotated by neuronal functions. The blue module was enriched in neurogenesis and cell adhesion GO terms, with most differentially expressed proneural, neurogenic genes, cadherins, and protocadherins clustered in this module, suggesting a relationship between cell adhesion and neurogenesis (Tables S6E and S6F). The brown module was enriched in synaptic genes; while the orange and yellow modules were enriched in DNA binding, RNA metabolism, and transcriptional regulation. Notch ligands, receptors, and downstream molecules clustered in both the blue (RFNG, RBPJ, MAML3, DLL3, DLL4, MFNG, NOTCH4, and DTX1) and yellow (MAML1, HEYL, DLL1, NOTCH1, HES5, HES6, DTX4, and NEURL1B) modules. In contrast, the three modules upregulated in MONs displayed non-neuronal annotations: the purple module was enriched in actin, focal adhesion, and ECM, with most of the differentially expressed ITG receptors clustered into this module; the turquoise module was enriched in cell-cycle and DNA replication terms, and the red module was enriched in peptide and vesicle transport (Figure S5A; Tables S6C and S6D). Module eigengene analysis can provide a simplified global picture of the system. Consistently with enrichment in DEGs, the blue, brown, orange, and yellow modules showed higher eigengene expression in ORGs, and the purple, red, and turquoise showed higher expression in MONs (Figure 6B). Correlation analysis between module eigengenes showed positive correlation between the orange, blue, and yellow modules and a negative correlation between the purple and those in the blue and orange modules (Figure 6C). To explore the mechanism of these relationships, we performed TF analysis using the Enrichr online database (see the Experimental Procedures) for the genes in each module and identified a set of putative TF-target gene pairs. At the network level, we find that TFs upstream of genes in the blue module belong to the blue, orange, and yellow modules, indicating that the strong positive correlations among these modules may represent, at least in silico, TF-target regulatory relationships. Focusing on the blue module and its putative TFs, we filtered out upstream TFs whose targets were not in the list of DEGs at any time points, resulting in a set of 60 TFs regulating the blue module (Table S7A). Among these, 41 TFs targeted both cell adhesion- and neurogenesis-related genes (Table S7B), and, among those, 12 (MYT1, INSM1, ZSCAN18, SCRT1, EBF3, DPF1, MEIS3, UNCX, NEUROD4, ZNF536, ST18, and DEAF1) were co-expressed with their targets in the blue module (Figure 6D; Table S7C). Interestingly, 11 out of these 12 TFs are downregulated in MONs versus ORGs at least at one time point. The co-expression of these TFs and their targets within the same module support a regulatory relationship, suggesting that this set of TFs is at least in part responsible for downregulating cell adhesion and neurogenic genes in MONs.
Figure 6.
Characterization of the Seven Transcript Modules Differentially Expressed between MONs and ORGs
(A) Modules' overlap q values (as –log10 [q-value]) with DEGs in MONs versus ORGs at each time point.
(B) Barplots of modules' eigengenes versus time in MONs and ORGs. Reported also are the top scoring functional annotation for each module.
(C) Module to module correlation plots. Represented are the eigengenes as dots and the corresponding correlation coefficients.
(D) Blue module subnetwork, focusing on inferred TFs and associated target genes, as described in Table S7C, after filtering out any edge with an absolute value of the correlation coefficient <0.5. Yellow ovals, cell adhesion (CCADh)-related genes and neurogenesis (NGEN)-related genes, differentially expressed between MONs and ORGs that are TF targets. Blue, upstream TF; arrows, direction of TF-target relationship.
Dissociation-Induced Transcriptional Alterations Are Independent of Neural Induction Protocol
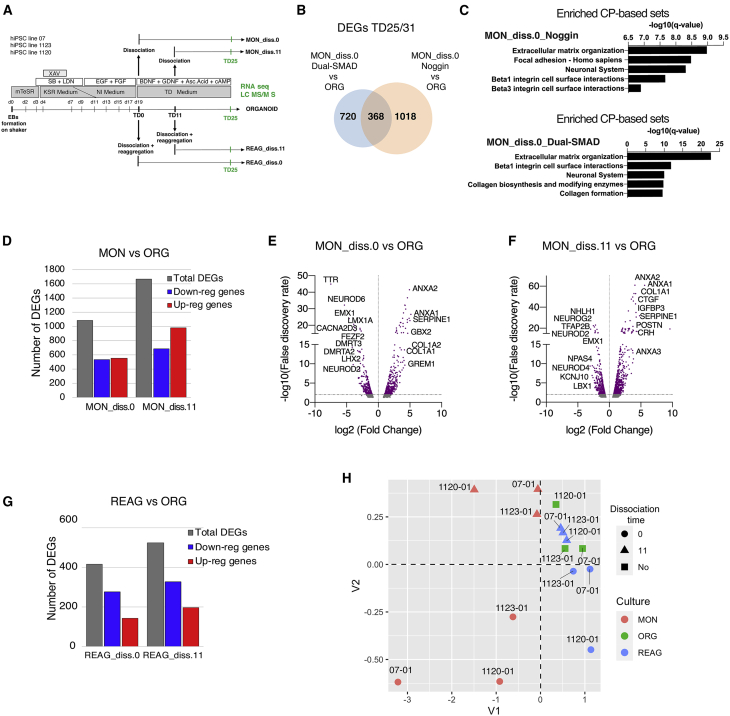
To assess the reproducibility of the observed dissociation-induced transcriptional alterations, we repeated the experiments using the same iPSC lines with a different neuronal induction protocol that uses the Dual-SMAD inhibition (Figure 7A). Similarly to our previous experiment, we generated ORGs, as well as parallel MONs and REAGs, after early dissociation at TD0, and used RNA-seq to infer transcriptional alterations in MONs and REAGs, compared with the intact ORGs, our reference preparation. The samples generated in this replication experiment, the DEGs between MONs and REAGs compared with ORGs, and their GO and CP annotations are listed in Tables S8A–S4D. We identified 1,088 DEGs in MONs versus ORGs, which we tested for overlap with the 1,386 previously identified DEGs under the Noggin protocol. We observed a significant overlap between the two lists of DEGs (368 genes, p < 10−165) (Figure 7B), strong correlation in log2 fold change in gene expression (correlation coefficient ~0.76; not shown), and virtually identical top CP and GO terms for the sets of 1,088 and 1,386 DEGs comparing MONs versus ORGs under Noggin and Dual-SMAD protocols (Figure 7C), suggesting that dissociation-induced key transcriptional alterations are not specific to the Noggin protocol.
Figure 7.
Consistency of Transcriptomic Changes between MONs and ORGs across Protocols and Times of Dissociation
(A) Experimental design. Cells derived from three iPSC lines were processed for transcriptomic or proteomic analysis. Abbreviations as in the text.
(B) Venn diagram showing overlap between early dissociated MON versus ORG DEGs under Noggin (n = 2 biological different iPSC lines per condition) or Dual-SMAD neuronal induction protocol (n = 3 biologically different iPSC lines per condition).
(C) Top CP-based annotations for the sets of DEGs between early dissociated MONs versus ORGs under Noggin and Dual-SMAD protocols, respectively.
(D) Total number of DEGs (gray bar), downregulated DEGs (blue bar), and upregulated DEGs (red bar) in early and late dissociated MONs versus ORGs at TD25.
(E and F) Volcano plots of MON_diss.0 versus ORG (E) and MON_diss.11 versus ORG (F) DEGs after early and late dissociation. Dots above the horizontal line are statistically significant (FDR < 0.01).
(G) Total number of DEGs (gray bar), downregulated DEGs (blue bar), and upregulated DEGs (red bar) in early and late REAGs versus ORGs at TD25.
(H) Multidimensional scaling plot of RNA-seq data at TD25 for all conditions.
Time of Dissociation Has Marginal Effects on Transcriptional Alterations
To investigate whether there is a critical period when cell-to-cell contact must be maintained for proper forebrain neurogenesis, we generated MON and REAG cultures after a later dissociation time point at TD11 (respectively, MON_diss.11 and REAG_diss.11) along with those described previously after dissociation at TD0, using the Dual-SMAD inhibition protocol described above. We then compared DEGs between the early dissociated MONs versus ORGs (1,088) with the DEGs between the late dissociated MONs versus ORGs (1,667) (Figure 7D). We found nearly 500 DEGs in common, which also displayed good correlation in direction of change (correlation coefficient r = 0.795) (Figures S6A and S6B). Early and late dissociation DEGs sets showed overlap of CP and GO terms, specifically Extracellular Matrix-related terms for genes upregulated in MONs and Nervous System Development and Neurogenesis for genes upregulated in ORGs (Figures S6C–S6F; Tables S8A–S8D). Top upregulated genes in both early and late dissociated MON preparations included several collagen types (COL1A1, COL1A2) and gene products involved in ECM organization (SERPINE1, CTGF, GREM1, ANXA1, and ANXA2), and top downregulated genes included many neurogenic TFs (NEUROD2, EMX1/2) and deep and upper cortical layer neuronal markers (BCL11B, FEZF2, and CUX2) (Figures 7E, 7F, and S6G).
When gene expression in the REAG conditions was compared with the ORGs, early (REAG_diss.0) and late (REAG_diss.11) dissociation revealed only 417 and 523 DEGs (Figure 7G; Table S8A), respectively, confirming a weaker transcriptional perturbation compared with the MON condition, as described previously with the Noggin protocol (Figure 5B).
Indeed, multidimensional scaling showed a clear separation along the V1 axis between MON conditions and everything else (REAGs and ORGs), while along the V2 axis we observed a separation associated with the early versus late dissociation, more pronounced in MONs than in REAGs. Also, there was a wider spread in early dissociation versus late dissociation, more pronounced in REAGs, perhaps suggesting that late dissociation results in lower line-to-line variability, possibly because of a less drastic perturbation on the phenotype. In particular, the late dissociation REAGs seem closer to the ORGs, further supporting the idea that a later dissociation has less disruptive effect on neural development (Figure 7H).
Next, we used mass spectrometry-based label-free quantitative proteomics in two cell lines to interrogate proteome alterations between early dissociated MON and ORG conditions. We quantified 4,783 peptides across all conditions, with approximately 90% overlap with the list of expressed genes (Figure S6H). We then tested proteins for differential expression in MON_diss.0 versus ORG and identified 199 differentially expressed proteins (DEPs) (FDR < 0.05) with 107 upregulated (i.e., up in MONs) and 92 downregulated proteins (i.e., up in ORGs) (Figure S6I). The list of DEPs and their GO and CP annotations are presented in Tables S8E and S8F.
While comparison of the DEPs with the DEGs (Figure S6J), under identical conditions, resulted in modest overlap (about 30 DEPs also found as DEGs), this is consistent with previous work (Freiberg et al., 2016; Ghazalpour et al., 2011; Pacheco et al., 2017), and is likely related to the generally poor correlation between protein and transcript abundance (Haider and Pal, 2013; Vogel and Marcotte, 2012). Nevertheless, among the top GO terms associated to the upregulated proteins in MONs we find terms all related to focal adhesion and the cell-substrate adherens junction (Figure S6K). Among the top DEPs, also in overlap with the cell-substrate adhesion annotations, were gene products, such as FLNA/B, VCL, TLN1/2, ACTN1/4, PLEC, and ILK/Integrin linked kinase, linking the plasma cell membrane with the cortical actin cytoskeleton and interacting with integrins and other transmembrane receptors. Therefore, there is a consistent functional annotation between gene and proteins upregulated in MONs, both supporting a role of cell adhesion and ECM in disrupting cellular organization and fate. Interestingly, the DEPs offer a different, complementary picture with respect to the differential gene expression, providing insights into potential additional mechanisms that are impacted by variations in cell-to-cell contacts, e.g., the cell cytoskeleton, including contractile proteins, such as actin, myosin, and associated molecules, that influence cell shape as well as the symmetry of cell divisions, processes which may be highly disrupted by culture on a 2D substrate.
Discussion
In this work, we compare the ability to generate cortical neural progenitor cells, and subsequently neurons, from three human iPSC lines differentiated under 3D ORG versus 2D MON conditions, using two different directed differentiation protocols. Longitudinal transcriptome analyses revealed that dissociation introduced a wave of transient transcriptional alterations dissipating over time, as the number of DEGs decrease from about a total of 8,500 to 8,000 at TD2 to 1,300 to 1,000 at TD31. However, the proportion of strong transcriptional alterations increased with time, delineating a picture of divergent systems with a more and more defined identity. Important signaling molecules and cell fate determinants were altered in the MONs at all time points analyzed, including a downregulation of cell-to-cell adhesion molecules, Notch pathway genes, and cortical cell fate genes, with a concomitant upregulation of cell-to-substrate adhesion molecules, including collagens, integrins, and laminins. Thus, contrasting MON with ORG transcriptomes over time and both preparations with fetal human brain (Figures 3H and 3I) does not support a maturational delay, i.e., a closer similarity of MONs to ORGs or to fetal brain at the later time point.
This is consistent with previous findings of divergent changes in cell membrane and ECM molecules when comparing 3D versus 2D differentiations (Simao et al., 2018). The potentially permanent result of this dynamic process may have led MONs onto a different developmental trajectory, at least within the observed temporal window.
Inhibiting ITGβ1 receptor signaling in MONs decreased cell-to-matrix adhesion and normalized cell division to levels comparable with ORGs, suggesting that increased ITG-laminin interactions are causing excessive proliferation. This is consistent with previous works showing that laminin promotes human primary neurosphere growth (Hall et al., 2008). However, suppressing ITGβ1 signaling in MONs did not change the downregulated expression of neurogenic and telencephalic cell fate genes. Thus, excessive ECM signaling causes abnormalities in cell proliferation but is not related to aberrant cell fate in this model.
A likely reason for the impaired differentiation of MONs is that loss of cell-to-cell contacts decreases intercellular signaling, while at the same time enhancing relatively unspecific cell-ECM contacts with laminin. In support of this, the prompt reaggregation of the dissociated neuronal progenitor cells partially restored aberrant cell fate. However, REAG preparations still displayed a disorganized architecture, with decreased RG cell polarity, smaller rosette structures, and loss of clear separation between RG and the postmitotic cell layer due to intermixing of newly born neurons with progenitor cells. Dissociation at a later time point did not substantially improve the differences in gene expression between MONs and ORGs, although transcriptomes of late dissociated REAGs were closer to those of ORGs. Overall, the data suggest that even a temporary disruption of cell-to-cell contacts, especially at early time points, has profound and possibly permanent effects.
Crucial components of layer- and compartment-specific cell adhesion are proto-cadherins and cadherin surface molecules. Cadherin-mediated junctions are located between the apical endfeet of RG cells and regulate mitotic spindle orientation, cortical architecture, and cell fate (Chilov et al., 2011; Gloerich et al., 2017; Tuncay and Ebnet, 2016). We show that, along with decreased CDH2 protein expression along their apical endfeet, the polarized organization of RG cells is almost completely lost in MONs and REAGs. Focused cell-to-cell Notch signaling and translocation of the Notch intracellular domain to RG nuclei promotes RG self-renewal (Hatakeyama et al., 2014). We indeed observed a global suppression in Notch signaling in MONs, including downstream proneural and neurogenic genes. This was likely the result of disruption in CDH2-containing apical complexes, as shown by the partial reversal of Notch and neurogenic gene expression upon reaggregation.
However, rather than premature differentiation, as often seen after disruption of apical complexes in animal models (Bultje et al., 2009; Cappello et al., 2006; Kim et al., 2010; Rousso et al., 2012; Zhang et al., 2010), we observed defective activation of proneural genes (i.e., NEUROG1/2 and NEUROD1/4/6) and outer RG genes (e.g., FAM107A, MT3, and HOPX) in MONs compared with ORGs, suggesting an impaired capability of RG cells to give rise not only to other RG cells, but also to intermediate progenitors, outer RG progenitors, and early neurons. This could perhaps be attributed to a role of cell adhesion in controlling the spread and stability of Notch-dependent HES1 and DLL/NEUROG2 transcriptional oscillations between adjacent RG cells, oscillations which precede sustained NEUROG2 expression, which is required for neuronal differentiation (Imayoshi et al., 2013; Shimojo et al., 2011). NEUROG2+ progenitors, in turn, upregulate the transcriptional repressors FOXP2/4, MYT1, INSM1, and SCRT1, which downregulate CDH2 transcription and promote detachment of TBR2+ IPCs and outer RG from the ventricular layer (Itoh et al., 2013; Rousso et al., 2012; Tavano et al., 2018; Vasconcelos et al., 2016). This reciprocal regulation between RG cells and nascent neurons is maintained in the ORGs, as corroborated by co-expression of Notch-related genes, cadherins, and the above-mentioned IPC transcriptional repressors MYT1, INSM1, and SCRT1 within the blue module, but fails to be implemented in the MONs where all these genes are downregulated. Owing to a lack of such cell-to-cell contacts, MON and to a certain extent REAG preparations fail to reproducibly implement and maintain IPC generation and cortical neurogenesis.
Overall, the evidence supports a fundamental role of contacts between precursor cells in triggering and maintaining dynamic intercellular signaling systems, including cadherins and Notch, and undoubtedly others that govern the regional and cellular fate commitment of neuronal progenitors.
Experimental Procedures
Neuronal Differentiation of iPSC Lines
We used three previously generated human male control iPSC lines (Mariani et al., 2015). Telencephalic ORGs were differentiated as described previously (Mariani et al., 2015). To generate MONs, neuronal rosettes were dissociated into single cells at TD0 and plated onto poly-L-ornithine- and laminin-coated wells, and, to generate REAGs, cell suspensions were quickly re-aggregated in a 96-well plate. The experiment was repeated using the same lines differentiated with a Dual-SMAD inhibition protocol adapted from Rigamonti et al. (2016) with SB431542 and LDN-193189 in place of Noggin (see the Supplemental Experimental Procedures and Table S1). Immunostaining, stereological analysis, qRT-PCR, and western blot analysis were performed as described in the Supplemental Experimental Procedures.
Transcriptomic and Proteomic Analysis
Samples (see Table S1) were processed for RNA-seq and label-free liquid chromatography-tandem mass spectrometry (LC-MS/MS) analysis as described in the Supplemental Experimental Procedures. RNA-seq reads were mapped to the human genome (hg19) and the GencodeV7 (Harrow et al., 2012) transcriptome annotation. DEGs were inferred using the edgeR pipeline, and an FDR cut-off of 0.05 was used for all the tests. We used the WGCNA package (Langfelder and Horvath, 2008) for co-expression network analysis using gene expression estimates (as log2 (RPKM + 1)) from all the iPSC-derived ORGs (three cell lines and three time points). We inferred TFs potentially upstream of genes within the blue module using the Enrichr web application (Kuleshov et al., 2016) and filtered out non-significant TFs (see the Supplemental Experimental Procedures). The LC-MS/MS data were processed using Proteome Discoverer (v.2.1; Thermo Fisher Scientific) and protein identification was carried out using the Mascot search algorithm (Matrix Science), and analyzed using Scaffold (Proteome Software).
Data and Code Availability
The source data described in this manuscript are available via the PsychENCODE Knowledge Portal (https://psychencode.synapse.org/). The PsychENCODE Knowledge Portal is a platform for accessing data, analyses, and tools generated through grants funded by the National Institute of Mental Health (NIMH) PsychENCODE program. The accession number for RNA-seq and proteomic data reported in this paper is https://doi.org/10.7303/syn23593358. Data are available for general research use after complying with requirements for data access and data attribution.
Author Contributions
F.M.V. and G.C. conceived the study. F.M.V. supervised the project. G.G.A., S.S., and G.C. designed, performed, and analyzed experiments. G.C. analyzed RNA-seq and LC-MS/MS data. F.M.V., S.S., and G.C. prepared display items. G.G.A., S.S., G.C., and F.M.V. wrote the manuscript. All authors participated in discussion of results and manuscript editing.
Acknowledgments
We thank Junhyun Park for his help in stereological analyses, Kaya Bilguvar and Christopher Castaldi of the Yale Center for Genomic Analysis for help with RNA sequencing and Rashaun Wilson and TuKiet T. Lam of the W.M. Keck Foundation Biotechnology Resource Laboratory at Yale University for help with the LC-MS/MS. We thank Jessica Mariani, Anahita Amiri, and Alexandre Jourdon for help with the neuronal Dual-SMAD protocol, and Jeremy Schreiner for proofreading and editing the manuscript.
This work was funded by NIH grants U01 MH103365 and R01 MH109648, and by the Harris Family Professorship fund. We are grateful to the Sage Bionetworks Data Coordination Center for help with data access.
Published: January 28, 2021
Footnotes
Supplemental Information can be found online at https://doi.org/10.1016/j.stemcr.2020.12.019.
Contributor Information
Gianfilippo Coppola, Email: gianfilippo.coppola@yale.edu.
Flora M. Vaccarino, Email: flora.vaccarino@yale.edu.
Supplemental Information
s
. Primer sequences used for qPCR validation of RNA-seq data analysis
. Proteomic analysis by LC-MS/MS in early dissociated MONs and ORGs at TD25.
References
- Ardhanareeswaran K., Mariani J., Coppola G., Abyzov A., Vaccarino F.M. Human induced pluripotent stem cells for modelling neurodevelopmental disorders. Nat. Rev. Neurol. 2017;13:265–278. doi: 10.1038/nrneurol.2017.45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arlotta P., Pasca S.P. Cell diversity in the human cerebral cortex: from the embryo to brain organoids. Curr. Opin. Neurobiol. 2019;56:194–198. doi: 10.1016/j.conb.2019.03.001. [DOI] [PubMed] [Google Scholar]
- Bultje R.S., Castaneda-Castellanos D.R., Jan L.Y., Jan Y.N., Kriegstein A.R., Shi S.H. Mammalian Par3 regulates progenitor cell asymmetric division via notch signaling in the developing neocortex. Neuron. 2009;63:189–202. doi: 10.1016/j.neuron.2009.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Camp J.G., Badsha F., Florio M., Kanton S., Gerber T., Wilsch-Brauninger M., Lewitus E., Sykes A., Hevers W., Lancaster M. Human cerebral organoids recapitulate gene expression programs of fetal neocortex development. Proc. Natl. Acad. Sci. U S A. 2015;112:15672–15677. doi: 10.1073/pnas.1520760112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cappello S., Attardo A., Wu X., Iwasato T., Itohara S., Wilsch-Brauninger M., Eilken H.M., Rieger M.A., Schroeder T.T., Huttner W.B. The Rho-GTPase cdc42 regulates neural progenitor fate at the apical surface. Nat. Neurosci. 2006;9:1099–1107. doi: 10.1038/nn1744. [DOI] [PubMed] [Google Scholar]
- Chambers S.M., Fasano C.A., Papapetrou E.P., Tomishima M., Sadelain M., Studer L. Highly efficient neural conversion of human ES and iPS cells by dual inhibition of SMAD signaling. Nat. Biotechnol. 2009;27:275–280. doi: 10.1038/nbt.1529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chilov D., Sinjushina N., Rita H., Taketo M.M., Makela T.P., Partanen J. Phosphorylated beta-catenin localizes to centrosomes of neuronal progenitors and is required for cell polarity and neurogenesis in developing midbrain. Dev. Biol. 2011;357:259–268. doi: 10.1016/j.ydbio.2011.06.029. [DOI] [PubMed] [Google Scholar]
- Eiraku M., Watanabe K., Matsuo-Takasaki M., Kawada M., Yonemura S., Matsumura M., Wataya T., Nishiyama A., Muguruma K., Sasai Y. Self-organized formation of polarized cortical tissues from ESCs and its active manipulation by extrinsic signals. Cell Stem Cell. 2008;3:519–532. doi: 10.1016/j.stem.2008.09.002. [DOI] [PubMed] [Google Scholar]
- Engler A.J., Sen S., Sweeney H.L., Discher D.E. Matrix elasticity directs stem cell lineage specification. Cell. 2006;126:677–689. doi: 10.1016/j.cell.2006.06.044. [DOI] [PubMed] [Google Scholar]
- Freiberg J.A., Le Breton Y., Tran B.Q., Scott A.J., Harro J.M., Ernst R.K., Goo Y.A., Mongodin E.F., Goodlett D.R., McIver K.S. Global analysis and comparison of the transcriptomes and proteomes of group A Streptococcus biofilms. mSystems. 2016;1:e00149-16. doi: 10.1128/mSystems.00149-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ghazalpour A., Bennett B., Petyuk V.A., Orozco L., Hagopian R., Mungrue I.N., Farber C.R., Sinsheimer J., Kang H.M., Furlotte N. Comparative analysis of proteome and transcriptome variation in mouse. PLoS Genet. 2011;7:e1001393. doi: 10.1371/journal.pgen.1001393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gloerich M., Bianchini J.M., Siemers K.A., Cohen D.J., Nelson W.J. Cell division orientation is coupled to cell-cell adhesion by the E-cadherin/LGN complex. Nat. Commun. 2017;8:13996. doi: 10.1038/ncomms13996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haider S., Pal R. Integrated analysis of transcriptomic and proteomic data. Curr. Genomics. 2013;14:91–110. doi: 10.2174/1389202911314020003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hall P.E., Lathia J.D., Caldwell M.A., Ffrench-Constant C. Laminin enhances the growth of human neural stem cells in defined culture media. BMC Neurosci. 2008;9:71. doi: 10.1186/1471-2202-9-71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harrow J., Frankish A., Gonzalez J.M., Tapanari E., Diekhans M., Kokocinski F., Aken B.L., Barrell D., Zadissa A., Searle S. GENCODE: the reference human genome annotation for the ENCODE Project. Genome Res. 2012;22:1760–1774. doi: 10.1101/gr.135350.111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hatakeyama J., Wakamatsu Y., Nagafuchi A., Kageyama R., Shigemoto R., Shimamura K. Cadherin-based adhesions in the apical endfoot are required for active Notch signaling to control neurogenesis in vertebrates. Development. 2014;141:1671–1682. doi: 10.1242/dev.102988. [DOI] [PubMed] [Google Scholar]
- Iba K., Albrechtsen R., Gilpin B., Frohlich C., Loechel F., Zolkiewska A., Ishiguro K., Kojima T., Liu W., Langford J.K. The cysteine-rich domain of human ADAM 12 supports cell adhesion through syndecans and triggers signaling events that lead to beta1 integrin-dependent cell spreading. J. Cell Biol. 2000;149:1143–1156. doi: 10.1083/jcb.149.5.1143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Imayoshi I., Isomura A., Harima Y., Kawaguchi K., Kori H., Miyachi H., Fujiwara T., Ishidate F., Kageyama R. Oscillatory control of factors determining multipotency and fate in mouse neural progenitors. Science. 2013;342:1203–1208. doi: 10.1126/science.1242366. [DOI] [PubMed] [Google Scholar]
- Itoh Y., Moriyama Y., Hasegawa T., Endo T.A., Toyoda T., Gotoh Y. Scratch regulates neuronal migration onset via an epithelial-mesenchymal transition-like mechanism. Nat. Neurosci. 2013;16:416–425. doi: 10.1038/nn.3336. [DOI] [PubMed] [Google Scholar]
- Johnson M.B., Wang P.P., Atabay K.D., Murphy E.A., Doan R.N., Hecht J.L., Walsh C.A. Single-cell analysis reveals transcriptional heterogeneity of neural progenitors in human cortex. Nat. Neurosci. 2015;18:637–646. doi: 10.1038/nn.3980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim S., Lehtinen M.K., Sessa A., Zappaterra M.W., Cho S.H., Gonzalez D., Boggan B., Austin C.A., Wijnholds J., Gambello M.J. The apical complex couples cell fate and cell survival to cerebral cortical development. Neuron. 2010;66:69–84. doi: 10.1016/j.neuron.2010.03.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuleshov M.V., Jones M.R., Rouillard A.D., Fernandez N.F., Duan Q., Wang Z., Koplev S., Jenkins S.L., Jagodnik K.M., Lachmann A. Enrichr: a comprehensive gene set enrichment analysis web server 2016 update. Nucleic Acids Res. 2016;44:W90–W97. doi: 10.1093/nar/gkw377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lancaster M.A., Renner M., Martin C.A., Wenzel D., Bicknell L.S., Hurles M.E., Homfray T., Penninger J.M., Jackson A.P., Knoblich J.A. Cerebral organoids model human brain development and microcephaly. Nature. 2013;501:373–379. doi: 10.1038/nature12517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Langfelder P., Horvath S. WGCNA: an R package for weighted correlation network analysis. BMC Bioinformatics. 2008;9:559. doi: 10.1186/1471-2105-9-559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Long K., Moss L., Laursen L., Boulter L., ffrench-Constant C. Integrin signalling regulates the expansion of neuroepithelial progenitors and neurogenesis via Wnt7a and Decorin. Nat. Commun. 2016;7:10354. doi: 10.1038/ncomms10354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mariani J., Coppola G., Zhang P., Abyzov A., Provini L., Tomasini L., Amenduni M., Szekely A., Palejev D., Wilson M. FOXG1-dependent dysregulation of GABA/glutamate neuron differentiation in autism spectrum disorders. Cell. 2015;162:375–390. doi: 10.1016/j.cell.2015.06.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mariani J., Simonini M.V., Palejev D., Tomasini L., Coppola G., Szekely A.M., Horvath T.L., Vaccarino F.M. Modeling human cortical development in vitro using induced pluripotent stem cells. Proc. Natl. Acad. Sci. U S A. 2012;109:12770–12775. doi: 10.1073/pnas.1202944109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Milner R., Campbell I.L. The integrin family of cell adhesion molecules has multiple functions within the CNS. J. Neurosci. Res. 2002;69:286–291. doi: 10.1002/jnr.10321. [DOI] [PubMed] [Google Scholar]
- Nowakowski T.J., Bhaduri A., Pollen A.A., Alvarado B., Mostajo-Radji M.A., Di Lullo E., Haeussler M., Sandoval-Espinosa C., Liu S.J., Velmeshev D. Spatiotemporal gene expression trajectories reveal developmental hierarchies of the human cortex. Science. 2017;358:1318–1323. doi: 10.1126/science.aap8809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pacheco N.L., Heaven M.R., Holt L.M., Crossman D.K., Boggio K.J., Shaffer S.A., Flint D.L., Olsen M.L. RNA sequencing and proteomics approaches reveal novel deficits in the cortex of Mecp2-deficient mice, a model for Rett syndrome. Mol. Autism. 2017;8:56. doi: 10.1186/s13229-017-0174-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pollen A.A., Nowakowski T.J., Chen J., Retallack H., Sandoval-Espinosa C., Nicholas C.R., Shuga J., Liu S.J., Oldham M.C., Diaz A. Molecular identity of human outer radial glia during cortical development. Cell. 2015;163:55–67. doi: 10.1016/j.cell.2015.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rigamonti A., Repetti G.G., Sun C., Price F.D., Reny D.C., Rapino F., Weisinger K., Benkler C., Peterson Q.P., Davidow L.S. Large-scale production of mature neurons from human pluripotent stem cells in a three-dimensional suspension culture system. Stem Cell Reports. 2016;6:993–1008. doi: 10.1016/j.stemcr.2016.05.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rousso D.L., Pearson C.A., Gaber Z.B., Miquelajauregui A., Li S., Portera-Cailliau C., Morrisey E.E., Novitch B.G. Foxp-mediated suppression of N-cadherin regulates neuroepithelial character and progenitor maintenance in the CNS. Neuron. 2012;74:314–330. doi: 10.1016/j.neuron.2012.02.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saha K., Keung A.J., Irwin E.F., Li Y., Little L., Schaffer D.V., Healy K.E. Substrate modulus directs neural stem cell behavior. Biophys. J. 2008;95:4426–4438. doi: 10.1529/biophysj.108.132217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi Y., Kirwan P., Smith J., Robinson H.P., Livesey F.J. Human cerebral cortex development from pluripotent stem cells to functional excitatory synapses. Nat. Neurosci. 2012;15:477–486. doi: 10.1038/nn.3041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shimojo H., Ohtsuka T., Kageyama R. Dynamic expression of notch signaling genes in neural stem/progenitor cells. Front. Neurosci. 2011;5:78. doi: 10.3389/fnins.2011.00078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sidhaye J., Knoblich J.A. Brain organoids: an ensemble of bioassays to investigate human neurodevelopment and disease. Cell Death Differ. 2021;28:52–67. doi: 10.1038/s41418-020-0566-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simao D., Silva M.M., Terrasso A.P., Arez F., Sousa M.F.Q., Mehrjardi N.Z., Saric T., Gomes-Alves P., Raimundo N., Alves P.M. Recapitulation of human neural microenvironment signatures in iPSC-derived NPC 3D differentiation. Stem Cell Reports. 2018;11:552–564. doi: 10.1016/j.stemcr.2018.06.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takahashi K., Tanabe K., Ohnuki M., Narita M., Ichisaka T., Tomoda K., Yamanaka S. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell. 2007;131:861–872. doi: 10.1016/j.cell.2007.11.019. [DOI] [PubMed] [Google Scholar]
- Tavano S., Taverna E., Kalebic N., Haffner C., Namba T., Dahl A., Wilsch-Brauninger M., Paridaen J., Huttner W.B. Insm1 induces neural progenitor delamination in developing neocortex via downregulation of the adherens junction belt-specific protein Plekha7. Neuron. 2018;97:1299–1314.e8. doi: 10.1016/j.neuron.2018.01.052. [DOI] [PubMed] [Google Scholar]
- Tuncay H., Ebnet K. Cell adhesion molecule control of planar spindle orientation. Cell. Mol. Life Sci. 2016;73:1195–1207. doi: 10.1007/s00018-015-2116-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vasconcelos F.F., Sessa A., Laranjeira C., Raposo A., Teixeira V., Hagey D.W., Tomaz D.M., Muhr J., Broccoli V., Castro D.S. MyT1 counteracts the neural progenitor program to promote vertebrate neurogenesis. Cell Rep. 2016;17:469–483. doi: 10.1016/j.celrep.2016.09.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vogel C., Marcotte E.M. Insights into the regulation of protein abundance from proteomic and transcriptomic analyses. Nat. Rev. Genet. 2012;13:227–232. doi: 10.1038/nrg3185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu J., Vodyanik M.A., Smuga-Otto K., Antosiewicz-Bourget J., Frane J.L., Tian S., Nie J., Jonsdottir G.A., Ruotti V., Stewart R. Induced pluripotent stem cell lines derived from human somatic cells. Science. 2007;318:1917–1920. doi: 10.1126/science.1151526. [DOI] [PubMed] [Google Scholar]
- Zhang J., Woodhead G.J., Swaminathan S.K., Noles S.R., McQuinn E.R., Pisarek A.J., Stocker A.M., Mutch C.A., Funatsu N., Chenn A. Cortical neural precursors inhibit their own differentiation via N-cadherin maintenance of beta-catenin signaling. Dev. Cell. 2010;18:472–479. doi: 10.1016/j.devcel.2009.12.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
s
. Primer sequences used for qPCR validation of RNA-seq data analysis
. Proteomic analysis by LC-MS/MS in early dissociated MONs and ORGs at TD25.
Data Availability Statement
The source data described in this manuscript are available via the PsychENCODE Knowledge Portal (https://psychencode.synapse.org/). The PsychENCODE Knowledge Portal is a platform for accessing data, analyses, and tools generated through grants funded by the National Institute of Mental Health (NIMH) PsychENCODE program. The accession number for RNA-seq and proteomic data reported in this paper is https://doi.org/10.7303/syn23593358. Data are available for general research use after complying with requirements for data access and data attribution.