Abstract
Breast cancer (BC) is the most frequent cause of death among women, representing a global public health problem. Here, we aimed to discuss the correlation between the purinergic system and BC, recognizing therapeutic targets. For this, we analyzed the interaction of extracellular nucleotides and nucleosides with the purinergic receptors P1 and P2, as well as the influence of ectonucleotidase enzymes (CD39 and CD73) on tumor progression. A comprehensive bibliographic search was carried out. The relevant articles for this review were found in the PubMed, Scielo, Lilacs, and ScienceDirect databases. It was observed that among the P1 receptors, the A1, A2A, and A2B receptors are involved in the proliferation and invasion of BC, while the A3 receptor is related to the inhibition of tumor growth. Among the P2 receptors, the P2X7 has a dual function. When activated for a short time, it promotes metastasis, but when activated for long periods, it is related to BC cell death. P2Y2 and P2Y6 receptors are related to BC proliferation and invasiveness. Also, the high expression of CD39 and CD73 in BC is strongly related to a worse prognosis. The receptors and ectonucleotidases involved with BC become possible therapeutic targets. Several purinergic pathways have been found to be involved in BC cell survival and progression. In this review, in addition to analyzing the pathways involved, we reviewed the therapeutic interventions already studied for BC related to the purinergic system, as well as to other possible therapeutic targets.
Keywords: Breast tumors, Purinergic receptors, Ectonucleotidases, Tumor progression, Therapeutic possibilities
Introduction
Considering the relevance of breast cancer (BC) in the health scenario, it becomes even more relevant to understand the pathophysiological pathways involved in its development and control. In this context, the relationship between the purinergic system and the processes of survival, proliferation, invasion, and migration of breast tumor cells emerges. This correlation opens therapeutic possibilities that until then were not used to control BC. Taking this information into consideration, we seek to build a bibliographic review using the databases Pubmed, Scielo, Lilacs, and ScienceDirect, which addressed the main aspects of BC and its correlations with the purinergic system, visualizing new therapeutic possibilities and addressing those that are under development, allowing the discussion of topics that may evolve to improve the prognosis and quality of life of thousands of people affected by this neoplasm.
Epidemiology
The BC is one of the three most common cancer types in the world, after lung and colon cancer [1], and is the most frequent cause of death in women [2, 3]. According to Globocan [4], around 2.09 million women worldwide were diagnosed with this neoplasm in 2018, of which 626 thousand deaths occurred as a result of the disease. Annually, 1.7 million new cases are diagnosed, which means one case every 18 s [5]. In the USA, BC represents 14% of newly diagnosed cancers [6], and the estimate for 2020, for women, is about 276 thousand new cases and 42 thousand deaths [7]. In view of this, it is notable that despite significant advances in the research scenario, BC is still a worldwide public health problem, and thus, it represents a priority in medical research [8].
Risk factors
In women, the etiology of BC is multifactorial, involving environmental (most prevalent) and genetic factors. Among the environmental factors, the female gender stands out; advanced age; the race, that although BC in the USA is more prevalent in white women [9], black women are more likely to have human epidermal growth factor type 2 receptor (HER2) positive cancer [6, 10, 11], be more aggressive and with a worse prognosis [12, 13]; increased weight and body fat (mainly in postmenopause) [14, 15]; high levels of endogenous estrogen in the postmenopause [16, 17]; early menarche (before age 13) and late menopause [18]; nulliparous women with late first pregnancies [19, 20]; alcohol consumption [21–24]; and tobacco use (including secondhand smoke) [25–27]. In addition, protective factors are breastfeeding [28] and physical activity [29]. On the other hand, in relation to genetic factors, family history and gene mutations are involved. As for family history, studies have shown that the risk for BC increases twice as much, if the woman has an affected first-degree relative [30, 31]. Genetic mutations, in turn, happen directly in only 5% of cases, and can occur mainly in the BRCA, p53, STK11, CDH1, PALB2, and PTEN genes and in the mismatch repair genes [9]. Only within the BRCA1 gene, more than 1800 mutations have been identified, a fact that increases the risk of BC for men and women [3].
Pathogenesis
The BC occurs when there is unregulated cell growth within any of the components of the breast (lobes, ducts, adipose tissue, and lymphatic tissue) [32]. About 80% of BC cases start in the mammary duct epithelium [33]. This unregulated growth starts with modifications in the cell cycle, due to changes in genetic information [9]. Normally, it begins with an uncontrolled proliferation, loss of normal cellular characteristics, loss of responses to growth and proliferation-inhibiting factors, and a change in metabolism that favors tumor cell populations [34]. Lastly, it results in a painless breast nodule [3].
After the formation of the breast nodule, BC can advance to other tissues. The metastatic process involves changes in cell adhesions and the extracellular matrix, which is due to changes in the expression of adhesion molecules, such as E-cadherin, and in the activity of matrix metalloproteinases (MMPs) [35]. Following, the regional lymphatic involvement may occur, which is a strong indicator of the metastatic process [36]. To reach secondary sites, the tumor diffuses through platelet emboli and changes in endothelial cell adhesion, allowing tumor cells to pass to other sites. When other tissues are affected, changes in the extracellular matrix occur and the process of angiogenesis and proliferation in metastasis begins [35]. The high expression of the HER2, of the Wnt4 pathway, of the receptor activator of nuclear factor-κβ ligand (RANKL) signals [37], and of the purinergic system signaling, directly influences the metastatic process and tumor survival [38].
BC subtypes
The BC can be classified into invasive and non-invasive (in situ). Most cancers are invasive and extend from the ducts and glands to the lymph nodes and circulating tissues [3]. There are 21 different BC subtypes, varying according to their histological type [39]. Besides that, there are five different intrinsic subtypes, each of them varying in the clinical aspects and treatments required [40]: luminal A, luminal B HER2 positive, luminal B HER2 negative, overexpression of HER2 (HER2+), and triple negative (TNBC) [41]. Knowing which subtype the tumor belongs to, coupled with the knowledge of clinical and pathological markers, is necessary to define the therapeutic conduct of each BC subtype [42].
The luminal A molecular subtype is characteristic of 50% of breast carcinomas [43]; however, it has the best prognosis since the cells grow more slowly [44]. It has positive estrogen receptor (ER) and/or progesterone receptor (PR), but is negative for HER2 overexpression, in addition to being characterized as a low histological grade [40]. In the luminal A subtype, Ki-67, the nuclear marker of cell proliferation, is less than 14% [40, 45, 46]. Chemotherapy is not effective for this subtype, and specific target therapies are anti-estrogens, tamoxifen, and aromatase inhibitors (anastrozole, letrozole, exemestane) [47]. It has the lowest chances of metastasis, and when they occur, the most affected sites are the bones, having, however, a good prognosis [48].
The luminal B subtype has an incidence of 15% [49]. They are tumor cells with high histological grade, besides high level of cellular proliferation [40], making their prognosis worse than that of luminal A [50]. In the luminal B HER2 negative, there are ER and/or PR positive, HER2 negative, and Ki-67 > 14%. In turn, luminal B HER2 positive shows ER and/or PR positive, HER2 positive, and Ki-67 > 14% [40, 41]. This subtype benefits more than luminal A in chemotherapy associated with anti-estrogens [45].
The HER2 overexpression subtype represents 15 to 20% of BC cases [43]. It has a high expression of the HER2 oncoprotein, but does not have hormone receptors [51], and presents an intermediate histological grade [52, 53]. The metastasis, in this case, has a predilection for the brain, bones, liver, and lung [49]. The therapies are based on the use of chemotherapy with the monoclonal antibody trastuzumab [54, 55], and currently pertuzumab and taxane are also used in this combination as the first line for metastatic BC with HER2+ [56].
Finally, the basal subtype or triple negative breast cancer (TNBC) is characterized by lack of expression of hormonal receptors and HER2 [40]. It has a high histological grade and high mitotic rate, being responsible for a large number of deaths due to its aggressiveness [57]. The TNBC is responsible for 10 to 20% of all cases of BC [58]. Here, the first-line therapeutic option is chemotherapy [59, 60], preferably before surgery, and because of this, the outcome of this type of cancer is not favorable [61]. Some studies have also demonstrated the effectiveness of combining chemotherapy with bevacizumab (monoclonal anti-vascular endothelial growth factor antibody) or with atezolizumab (monoclonal anti-programmed death ligand 1 (PD-L1) antibody) for metastatic TNBC. In addition to first-line therapy, there are still other options, such as the use of polyadenosine diphosphate-ribose polymerase (PARP) inhibitors, such as olaparib and talazoparib [62], and through immunotherapy, which includes pembrolizumab (humanized monoclonal antibody (mAb) against programmed cell death protein 1 (PD-1)) [63, 64] and avelumab (human mAb against PD-L1), also for patients with metastatic TNBC [65].
In addition to the molecular classification by the presence of PR, ER, and HER2, tumors present in their center a small group of cells called cancer stem cells (CSC), which divide into other CSCs and also into more differentiated cells. These CSCs are part of the bulk primary tumor, helping to tumor growth and progression [66]. Breast cancer stem cells (BCSC) have markers that are commonly found and characterize this group of cells, such as CD44+/CD24-low [67], aldehyde dehydrogenase 1(ALDH1) [68], and CD133+ [69]. The first is related to a malignant progression phenotype, being highly expressed in the breast cancer lineage MDA-MB-231 (TNBC representative) [70] and in luminal B lineages [71]. In turn, ALDH1 is an intracellular enzyme that is related to the metabolism of retinol and cellular metabolic responses [68], being positively related to the HER-2 overexpression subtypes and TNBC [71]. Finally, CD133+, a transmembrane glycoprotein that maintains the characteristics of stem cells and prevents differentiation [69], is most commonly found in the MDA-MB-468 lineage [70]. Such facts demonstrate the biomolecular complexity of BC and reveal new therapeutic possibilities, since these cell groups are resistant to conventional therapies for the bulk tumor and are important for tumor development and progression [72, 73].
Although there is no consensus, CSCs are believed to originate from stem cells present in adult tissues responsible for tissue regeneration and repair [74], or even from the de-differentiation of cells already transformed. In any case, the tumor microenvironment (TME) in which the CSCs reside is what will regulate their activity [75]. Finally, this discovery of CSCs changed the oncology paradigm. Classically, all cancer cells were functionally identical and had the same tumorigenic properties, and could also initiate and continue cancer growth. However, with the CSCs model, tumors were found to be organized functionally hierarchically and with a population of heterogeneous cells, including cancer cells that have similar properties to CSCs and to most bulk tumor cells. These CSCs are capable of self-renewal and give rise to non-tumorigenic daughter cells, which make up a large part of the tumor [67, 76, 77]. In that sense, only these stem-like cancer cells and bulk tumor cells have intrinsic tumorigenic properties [78, 79]. Given this, the ideal treatment for BC should kill all malignant cells, including CSCs (by specific agents like salinomycin) and bulk tumor cells (by standard chemotherapy with paclitaxel or lipodox) [80].
Purinergic system
The purinergic system is a pathway that regulates immune homeostasis, through the secretion of cytokines, chemokines, removal of intracellular pathogens, and release of antigens and generation of reactive oxygen species (ROS) [81, 82]. In response to cell injuries, some purinergic mediators are released into the extracellular space, such as adenosine triphosphate (ATP) and adenosine (Ado) [83], which act as signals that shape immune responses [84, 85]. The purinergic pathway, therefore, acts by converting extracellular ATP into Ado, by means of a degradation process carried out by ectonucleoside diphosphohydrolases triphosphate (CD39) and ecto-5′-nucleotidase (CD73) ectoenzymes. While the former converts ATP and adenosine diphosphate (ADP) to adenosine monophosphate (AMP), the latter transforms AMP into Ado [86]. It has long been believed that ATP was restricted to the intracellular space, performing its activities as an energy mediator and participating in the synthesis of nucleic acids [87, 88]. However, it was found that ATP performs extracellular activities, acting as a signaling molecule in the purinergic system [89]. This system consists of membrane receptors that can interact with Ado through P1 family receptors, or with purine nucleotides, such as ATP, or pyrimidine, such as UTP, through P2 family receptors [90].
The responses triggered, both in physiological and pathological conditions, vary according to the concentration of the receptors present, with the levels of agonists in the extracellular space, and with the type of cell in which the activation of the receptor will be taking place [91]. In the pathological context, these receptors are involved in neuropathic pain, trauma, Parkinson’s and Alzheimer’s diseases, and other conditions of the nervous system. In addition, they also cover conditions such as inflammation, infections, cardiovascular diseases, and cancer [92].
In cancer, nucleotides, mainly ATP, are released by both tumor cells and host cells, and their actions influence both the activation and migration of immune cells, as well as the growth and proliferation of tumor cells. Nucleotides can cause immunostimulation or immunosuppression, depending on the concentration of the P2 receptors involved [38, 91].
Main purinergic receptors
Purinergic receptors are divided into P1 and P2 families [93]. Several cells present in their membrane receptors for Ado (P1 receptors) and nucleotides as ATP and ADP (P2 receptors) [89].
P1 receptors
The P1 family receptors mediate the actions of Ado [94], comprising four receptors (A1, A2A, A2B, and A3) [93, 95], with the intracellular domain being coupled to G protein, and the extracellular domain to Ado [96]. These receptors are found in all cells of the TME; however, the Ado receptors (ARs) most expressed in tumor cells are A3 and A2B [97]. In BC, A3 promotes a decrease of motility of tumor cells [98], while A2B induces migration and metastasis [99]. The main function of ARs is to regulate tissue homeostasis, and for this, it triggers anti- or pro-inflammatory effects, depending on the cell context [100].
The A1, A2A, and A3 receptors can be stimulated by low levels of Ado, around 30 to 300 nM. The A2B receptor, in turn, needs higher levels of Ado, as seen in hypoxic microenvironments, to be activated [101, 102]. Thus, the A2B receptor is only activated in pathophysiological conditions, such as in inflammatory TME, while in physiological concentrations, signaling occurs via A1, A2A, and A3 [103].
A1 receptor
The A1 receptor is involved in the process of carcinogenesis of several tumors, including BC. This involvement was confirmed by silencing this receptor in cell lines, in which the accumulation of cells in the G2/M phase was observed, demonstrating their proliferative effect (Table 1) [94].
Table 1.
Correlation between purinergic receptors and their effects on BC
| Purinergic receptor | Effects on BC | References |
|---|---|---|
| P1 receptors | ||
| A1 | Activation promotes cell proliferation | [94] |
| Activation promotes cell survival and decreased apoptosis in positive ER cells, worsening prognosis | [104] | |
| A2A | Overexpressed in hormone-dependent lineages | [99] |
| A2B | High expression in the MDA-MB-231 invasive lineage | [105, 106] |
| Activation favors migration and metastasis | [107] | |
| Activation promotes increased resistance to doxorubicin treatment | [108] | |
| A3 | Activation promotes reduction of cell proliferation and growth | [99, 109] |
| P2 receptors | ||
| P2X7 | Activation by high levels of ATP induces metastasis in the MDA-MB-435 and T47D lineages, and in tamoxifen-resistant BC cells | [110–114] |
| ATP activation for long periods promotes growth inhibition | [115] | |
| P2Y2 | ATP activation favors metastasis in the MCF-7 lineage | [38, 88, 110, 116], and [117] |
| Activation by UTP decreased migration in MCF-7 and MDA-MB-231 cells | [118] | |
| Binding of ADP and ATP promotes activation of endothelial cells, allowing migration | [87] | |
| P2Y6 | UTP activation promotes migration and metastasis | [119] |
| P2Y11 | ATP activation favors metastasis in the MCF-7 lineage | [38] |
| P2Y12 | ADP stimulation promotes platelet activation, favoring metastasis | [120] |
In vitro studies have shown that, when an A1 agonist is used, the survival rate of positive ER cells increases, while the levels of apoptosis decrease (Table 1). Also, the expression of p53 (tumor suppressor protein that regulates the cell cycle and apoptosis in tumor cells) and caspases decreases (Fig. 1) [104]. Yet, the use of an A1 antagonist promotes increased expression of p53 and caspases, thus favoring apoptosis in these cells [98]. However, one can observe a beneficial effect of the activation of A1 receptors, since they can guarantee endothelial integrity, thus reducing the invasive process [87].
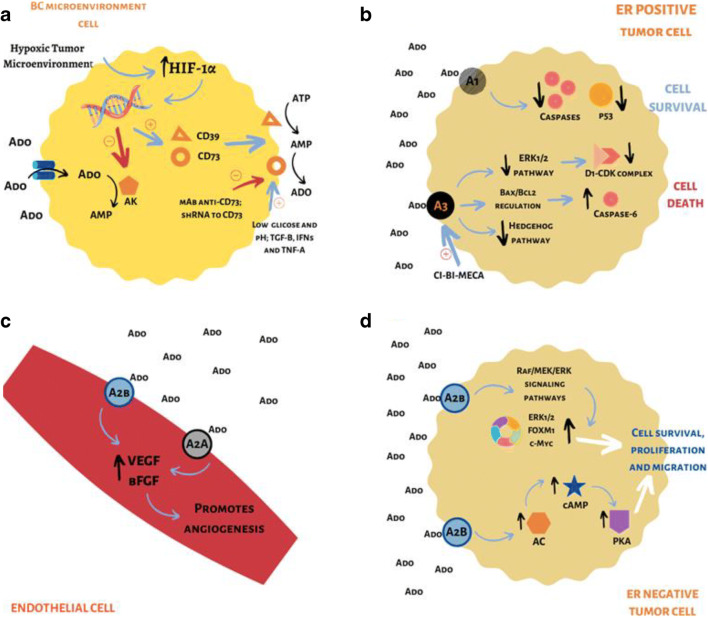
Fig. 1.
a The TME of solid tumors has the common characteristic of hypoxia (1% O2), which is due to a low supply of nutrients and oxygen. Hypoxia induces HIF-1α expression in tumor cells. This, in turn, acts on transcriptional factors, inhibiting the expression of AK, which performs the conversion of Ado into AMP, and favoring the expression of the enzymes CD39 and CD73, which act on cell membrane, converting ATP to AMP, and then to Ado, respectively. The increase of Ado in the extracellular medium by the action of CD73 causes a greater activation of ARs and promotes immunosuppression in the TME, making the control of the CD73 actions a therapeutic target. Low glucose levels and low pH, as well as cytokines, such as TGF-β, IFNs, and TNF-α, positively influence CD73 activity. Anti-CD73 mAb and shRNA for CD73 control and inhibit CD73 activity. b The activation of the A1 receptor in an ER positive BC cell line (MCF-7) promotes reduction in the number of caspases and in the regulatory protein of the cell cycle p53, favoring the survival of these tumor cells. However, in this same cell line, activation of the A3 receptor, which can occur by agonists such as Ado and CI-BI-MECA, promotes (1) the decrease in the activity of the ERK1/2 signaling pathway, decreasing the number of complexes cyclin D1-CDK4, which are related to cell cycle progression; (2) regulation of the Bax/Bcl2 pathway, causing an increase in the level of caspases-6; and (3) decrease in the activity of the Hedgehog signaling pathway. Performance through these three pathways will promote cell death. c The activation of A2A and A2B receptors in the endothelial cells of the TME promotes an increase in VEGF and in the bFGF, favoring angiogenesis. d In an ER negative BC cell line (MDA-MB-231), activation of A2B receptors by Ado regulates the Raf/MEK/ERK pathways, producing an increase in ERK1/2, FOXM1, c-Myc proteins, which are involved in the cell cycle control. Furthermore, stimulation of this receptor leads to an increase in AC, which promotes an increase in intracellular levels of cAMP, which activates PKA. Both A2B pathways will promote the survival, proliferation, and migration of tumor cells
In the studies by Lin et al. [121] and Mohamadi et al. [108], it was found that treatment with 17β estradiol in the MCF-7 lineage (ER positive) promoted an increase in the mRNA of A1 receptors [121] and an increase in activity of A1 receptor, being dose-dependent. Therefore, blocking the actions of estradiol in tumors that have high levels of this receptor appears as a suggestion for the proliferative control of BC [108].
A2 receptors
The A2A receptor is overexpressed in hormone-dependent BC lineages (Table 1), and leads to increased activity of adenylate cyclase (AC) and intracellular Ca2+ levels [99]. The use of 17β estradiol in the MCF-7 lineage also promoted an increase in A2A expression and activity [108].
The MDA-MB-231 (ER negative) lineage has a high expression of A2B receptors (Table 1), being a potential molecular biomarker for diagnosis and prognosis of BC [105, 106]. The A2B receptor in negative ER cell lineage in vitro and in vivo induces migration and metastasis (Table 1) [107], which occurs through intracellular signaling of AC, cyclic adenosine monophosphate (cAMP), and protein kinase A (PKA) (Fig. 1) [101].
In the invasive forms of BC, after the silencing of A2B receptors, a decrease in the expression of ERK1/2, FOXM1, and c-Myc proteins is observed, which are involved in the control of the cell cycle. This diminished expression of these proteins suggests control of the BC metastatic process through changes in the Raf/MEK/ERK signaling pathways (Fig. 1) [108].
Also, the A2B receptor is related to increased resistance to doxorubicin in the treatment of BC (Table 1) [108]. Therefore, its inhibition decreases the growth and metastatic potential of ER negative breast tumors and TNBC, in addition to promoting a better response to chemotherapy treatment [101, 122, 123].
A3 receptor
The A3 receptor is found at higher levels in primary and metastatic breast tumors [124, 125]. The activation of A3 promotes the reduction of cell proliferation and growth (Table 1) [99], through the induction of apoptosis and the deregulation of the Wnt/β-catenin signaling pathway in ER positive and ER negative cells (Fig. 1). In BC, it has been observed that the activation of A3 receptors considerably decreases the motility and migration of BC cells (Table 1) [109].
The A3 receptor induces the death of tumor cells through the signaling of cyclin-dependent kinase (CDK), by Bcl-2 proteins and caspases [126]. In BCSC, stimulation in low concentrations of A3 causes a G1 arrest in the cell cycle, due to the decreased activity of cyclin D1 and CDK-4 [127]. Involved in this process is the ERK1/2 signaling pathway, which is one of the regulators of the transition from the G1 phase to the S phase of the cell cycle, being very important in the stabilization and formation of the D1-CDK4 complexes [128]. The A3 receptor agonists, like the CI-BI-MECA, inhibit the ERK1/2 pathway, which is responsible for phosphorylating BCSC, thus preventing the cell cycle from continuing and decreasing the proliferative rates of these cells. The high stimulation of A3 receptor induces apoptosis by regulating the Bax/Bcl2 pathway that correlates with caspase-6 (Fig. 1). An increase in this caspase was observed both in the MCF-7 lineage and in the MDA-MB-231 lineage [127].
Still, ARs are believed to be inhibitors of the Hedgehog signaling pathway [129], a route of great importance for cell renewal and for the maintenance of breast stem cells [130]. In tumor cells, this signaling occurs through the transcription of genes related to cell proliferation and survival [131]. It was found that this activation of A3 promoted an inhibition of both the ERK1/2 and the Hedgehog pathway (Fig. 1), which are essential for the survival and growth of BCSC [127].
P2 receptors
The P2 receptors have ATP as the main agonist [132]. In general, P2 receptors are molecular structures present in the plasma membrane and activated by purine or pyrimidine nucleotides. They can be divided into P2X (ionotropic) and P2Y (metabotropic) receptors [38]. In all tumor types, there is expression of such receptors [99].
Ion channel receptors (P2X)
So far, the P2X family receptors has seven members (P2X1–7) [92], and these form permeable ion channels for Na+, K+, and Ca2+, with ATP being the only ligand that activates them [87]. The P2X7 receptor is the most expressed in tumors [133], and in addition to its ability to open an ion channel, it is also capable of activating a pathway in the plasma membrane (macropore) that allows the uptake of molecules from high molecular weight (up to 900 Da), an intrinsic characteristic of the stimulation of this receptor [38]. In addition, P2X7 contributes to the spread of metastasis and invasion of cancer cells [87], and therefore, when activated, stimulates inflammatory conditions [134].
P2X7
Among the P2X receptors, the one that stands out the most favoring tumor survival and proliferation is P2X7 [115], and the opening of Ca2+ channels through this receptor is of paramount importance for this activity to happen [38]. In healthy breast tissues, P2X7 expression is not found. However, this receptor is highly expressed in BC, and the high concentration of ATP in the TME favors its activation [135].
The rapid exposure of the P2X7 receptor to ATP promotes the opening of a selective channel for influx of Na+ and Ca2+, and efflux of K+. When stimulation occurs for a long time, the channel loses its selectivity, allowing the passage of several ions and soluble molecules of up to 900 Da [136, 137]. When there is an exaggerated entry of Ca2+ ions, the apoptosis and necrosis pathways can be activated (Fig. 2). In addition, the efflux of many K+ ions can lead the cell to exhaustion, also activating the apoptotic pathway [138]. Therefore, it is clear that in addition to the proliferative activity of the P2X7 receptor, it also has the cytotoxic activity [110]. In these cases, the interaction of P2X7 with ATP will activate the apoptotic cascade, leading tumor cells to death [38].
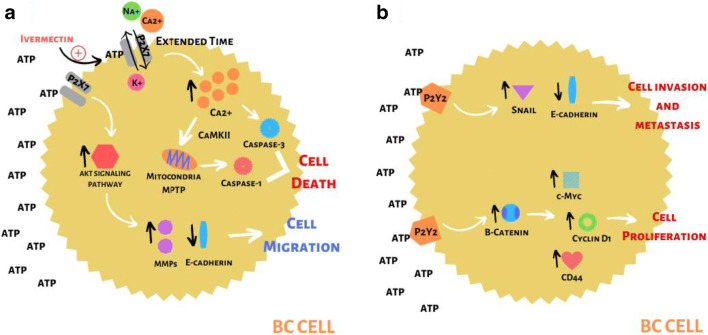
Fig. 2.
a Among P2X receptors, the P2X7 is highlighted within the context of BC. This receptor has a low affinity for ATP, requiring high concentrations of this molecule (such as those found in the TME) for its activation. When the interaction between ATP and P2X7 occurs quickly, there is the formation of the macropore that allows a selective passage of ions, with an influx of Na+ and Ca2+, and an efflux of K+. This passage of ions leads to the activation of signaling pathways such as AKT, which promotes increased levels of MMPs and decreased expression of E-cadherin, both of which are related to increased migration and invasiveness of the tumor. On the other hand, when the interaction between ATP and P2X7 becomes prolonged, there is a loss of channel selectivity, leading to a significant increase in intracellular Ca2+ levels. Under these conditions, there will be the activation of apoptotic and necrotic pathways. Ca2+ will promote the activation of calmodulin-dependent protein kinase II (CaMKII), which in turn will activate the mitochondrial permeability transition pore (MPTP), leading to the release of caspase-1, which is related to the necrotic process. Also, Ca2+ will also lead to the activation of caspase-3, related to the cell’s apoptotic pathway. Both caspases will promote cell death. It should be noted that one of the activities developed by ivermectin is to increase the sensitivity of P2X7 to ATP, causing a prolonged activation of this receptor to occur, thus activating the cell death pathways, favoring the control of tumor growth. b Among the P2Y family, the receptor that has a great correlation with BC is P2Y2. This receptor is found highly expressed in the cells that make up the edges of the tumor, and when activated by ATP, it promotes an increase in the Snail molecule that controls the expression of genes related to invasion and metastasis, and a decrease in E-cadherin. Such conditions favor the invasion and metastasis of tumor cells. Furthermore, the activation of P2Y2 promotes an increase in β-catenin, which in turn leads to an increase in the levels of the molecules c-Myc, cyclin D1, and CD44, all of them related to the control and progression of the cell cycle. Thus, P2Y2, in addition to promoting invasion and metastasis, also promotes the proliferation of BC cells
In the study by Chadet et al. [110], it was found that in the invasive lineage of BC, MDA-MB-435s, stimulation of P2X7 promoted an increase in the extracellular matrix proteolysis, favoring the invasive and metastatic process, while inhibiting this receptor reduced invasiveness in vitro and in vivo (Table 1). However, some in vivo studies have observed that in BC, the presence of P2X7 and high concentrations of ATP are related to the inhibition of tumor cell growth. This is due to an activation of AC, which promotes the increase of cAMP levels and reduces the proliferation and migration of endothelial cells, hindering the metastatic process [115].
In tamoxifen-resistant BC cells, a relationship was observed between P2X7 receptors and the migration and metastasis of these cells (Table 1). This was confirmed by treatment with a selective P2X7 antagonist (KN62), which is also an inhibitor of Ca2+/calmodulin-dependent protein kinase II [139, 140], which promoted a decrease in Zeb-1 levels, an E-cadherin suppressor molecule. Still, the stimulation of these receptors promoted an increase in the expression and activity of MMP-2 and 9, favoring changes in the extracellular matrix [111].
In BC cell line T47D, a high expression of the P2X7 receptor was observed, which when stimulated by ATP or analogues, promotes the invasion and migration of these cells (Table 1). In addition, P2X7 activation is related to the decrease in E-cadherin expression [112] and to the increase in MMP-13 expression and secretion, the latter being very relevant to the bone metastatic process [113, 114]. It was found that P2X7 induces the AKT signaling pathway in T47D cells [112], which is an important pathway for tumor progression [141]. It is believed that it is through the AKT pathway that P2X7 regulates MMP-13 and E-cadherin, favoring the invasive process (Fig. 2) [112].
The P2X7 receptor acts on tumor progression and metastasis and plays an important role in the maintenance of CSCs. This advance of the tumor triggered by the activation of P2X7 occurs because it is capable of inducing phenotypic changes and bringing cancer cells to a state of stemness [142]. CSCs are capable of self-renewing and differentiating into different types of tumor cells, being responsible for tumor relapse and recurrence, even after treatments [143, 144]. Considering the role of P2X7 in maintaining the stemness state of embryonic cells [145], it may be related to the plasticity of CSCs, which includes the balance between differentiated and undifferentiated cancer cells within tumor bulk, as well as its tumorigenic potential, aggressiveness, and resistance to new therapies [142].
Receptors coupled to G protein (P2Y)
The P2Y family receptors have eight members, with P2Y1, P2Y2, P2Y4, and P2Y6 being associated with the Gq protein, while P2Y12, P2Y13, and P2Y14 receptors are associated with the Gi protein. The P2Y11, in turn, can be integrated with Gs and Gq proteins [92]. The ATP is the favorite ligand only for the P2Y11 receptor [87], while the others can be activated preferentially by ADP, UDP, and UTP [146].
The P2Y receptors, in general, tend to promote migration and contribute to the formation of metastatic niches in BC [87]. In the MCF-7 lineage, ATP activation of P2Y2 and P2Y11 promoted a reorganization of the cytoskeleton and plasma membrane of these cells, favoring pseudopoidy, a necessary condition for the metastatic process (Table 1). In the MDA-MB-435 lineage, P2X7 is responsible for promoting pseudopoidy, favoring the invasion of the extracellular matrix [38]. The ADP activation of P2Y12, in turn, stimulates the metastatic process because it is involved in platelet aggregation that aids in displacement in the tumor embolus (Table 1) [120], while P2Y2 is activated by ADP and ATP in endothelial cells, favoring the migration of tumor cells (Table 1) [87].
It was found that P2Y2 activation increases the invasiveness of BC cells by inducing the epithelial mesenchymal transition (EMT) [116], through intracellular signaling triggered by Ca2+ [147]. Still, it was observed that P2Y2 is highly expressed at the edges of the breast tumor, where cells that infiltrate adjacent tissues are found. In these places, a high expression of Snail [116], a factor that directly or indirectly controls the expression of several genes involved in tumor invasion and metastasis [148], and almost no expression of E-cadherin was also noticed. It is believed that P2Y2 controls the migratory process in these cells through the control of Snail and E-cadherin (Fig. 2). It is worth noting that the expression of P2Y2 is inversely correlated with the histological grade, HER2 amplification, and Ki-67 index in breast tumors [116].
In the study by Zhang et al. [117], a high expression of P2Y2 was observed in the MCF-7 and MDA-MB-231 lineages. In this analysis, P2Y2 is related to the migration of these cells through the regulation of β-catenin. The Wnt/β-catenin signaling controls processes such as cell renewal, proliferation, and differentiation, in addition to influencing the control of apoptosis [149]. It was observed that the interaction of ATP with P2Y2 increases the expression of β-catenin as well as the genes regulated by this molecule, such as c-Myc, cyclin D1, and CD44, in the analyzed cell lines (Fig. 2) [117]. Also, the higher metastatic rate in the MDA-MB-231 lineage compared to MCF-7 is related to the greater activity of P2Y2. This receptor induces hypoxia through inducible hypoxia factor (HIF-1α), creating a favorable microenvironment for cell migration [150].
Although Chadet et al. [110] and Jin et al. [88] demonstrated that the P2Y2 receptor stimulates the proliferation of the MCF-7 lineage, the study by Li et al. [118] demonstrated that the P2Y2 activation by UTP reduced the proliferation of estrogen-dependent breast tumor cells, such as the MCF-7 lineage, and negative ER cells, such as the MDA-MB-231 (Table 1). It is possible that this duplication of results stems from differences between the methodologies used for the development of the cultures carried out in the studies, demonstrating the need for further studies on this subject.
Finally, it has been seen that P2Y6 is highly expressed and mutated in tumor cells and is significantly related to a worse prognosis in BC, since it indirectly represents a potential for malignancy. The relationship between UTP and P2Y6 promotes greater activity of MMPs, through ERK and NF-kB signaling, which stimulates the escape of the primary tumor site and the invasion of other regions (Table 1) [119]. The TNBC subtype has a high P2Y6 expression, contributing to a worse prognosis [151]. Still, some luminal A tumors with a worse prognosis have high levels of P2Y6, which may be indicative of mesenchymal characteristics in these cells, being related to a higher rate of cell migration and metastasis [152].
P0 receptors
There are also the P0 family receptors, which interact with adenine. These receptors are not yet fully understood and several studies have been carried out to verify the reflexes of their activation [92].
Ectonucleotidases
The ATP, under normal conditions, is produced from the gradual phosphorylation of ADP and AMP, originating from a molecule that stores energy and can be used in several metabolic processes [87]. ATP can be released, in addition to central or peripheral neural cells, from all cells through different mechanisms. Therefore, it is formed intracellularly and can be physiologically released [153]; moreover, its release is increased in pathological conditions such as cancer [38]. However, during cellular stress processes, ATP can be released by cells that have suffered damage, and subsequently be degraded by four enzymes from the ectonucleotidase group: ectonucleoside diphosphohydrolases triphosphate (E-NTPDase; CD39), ecto-5′-nucleotidase (E-5′-nucleotidase; CD73), ectonucleotide pyrophosphatase/phosphodiesterase (E-NPPs), and alkaline phosphatases, leading to Ado production [154]. Thus, ectonucleotidases are responsible for regulating ATP and Ado levels in the TME [97, 155].
The enzymatic activity of CD39 and CD73 is important for the regulation of signals involving immune and tumor cells. The CD39 promotes the conversion of ATP and ADP into AMP, while CD73 performs the irreversible catalysis of AMP into Ado [156]. Finally, Ado is removed from the extracellular space from its conversion to inosine (INO) by the action of adenosine deaminase, or by its absorption, back into the cell [157]. The conversion carried out by CD73 is, in theory, irreversible. In order for Ado to regenerate in AMP, it is necessary for membrane transporters to load Ado into the intracellular medium, where the enzyme adenosine kinase (AK) is found. Therefore, the accumulation of Ado in the extracellular medium is influenced both by the action of CD73 and by the activity of Ado membrane transporters. It is worth mentioning that in microenvironments where there is hypoxia, a transcriptional inhibition of AK occurs by the HIF-1α, leading to an increase in extracellular levels of Ado [158]. Furthermore, HIF-1α leads to the expression of the ectoenzymes CD39 and CD73 in tumor cells, which are responsible for maintaining the TME rich in Ado (Fig. 1) [159].
CD73 and CD39
The CD39 and CD73 are the main nucleotide metabolizing enzymes, therefore regulating inflammation and the immune system. Due to the ability of these enzymes to convert ATP to Ado, they are able to move immune cells from a pro-inflammatory state to an anti-inflammatory state [156]. Depending on the pathophysiological context in which they are inserted, these enzymes can shape the meaning of several diseases, including cancer [160, 161], since Ado promotes neoplastic growth and the capacity for tumor metastasis [84]. Studies have shown that deficient CD39 or CD73 mice were susceptible to inflammation, and that these enzymes are biomarkers in tumors, and their high expression is linked to worse prognosis in the case of TNBC [162, 163].
CD39
The first step in purine metabolism is carried out by the CD39 enzyme, which converts ATP to AMP in a Ca2+- and Mg2+-dependent manner [164]. This enzyme is expressed in the thymus, spleen, placenta, and lung [165]. Its expression is regulated by hypoxia, oxidative stress, and pro-inflammatory cytokines [161], through the transcription factors Sp1 [166] and Stat3 [167]. The increased expression of CD39 was found in several solid tumors, which suggests the involvement of this protein in neoplastic progression [168], both in the beginning of angiogenesis and in the progression of neovascularization [169].
CD73
After the action of CD39, the enzyme CD73 dephosphorylates extracellular AMP into Ado [156]. The protein CD73 is encoded by the NT5E gene, and is composed of 523 amino acids, its expression being regulated by various mechanisms [96]. In cancer, in which ATP and AMP are released in the extracellular space, the function of CD73 increases [170]. In this case, CD73 can interact with fibronectin and laminin, which facilitates the motility of cancer cells and results in the process of metastasis in human BC cells [171, 172].
The CD73 is also highly expressed in cells that make up the TME of BC [173, 174], so much so that overexpression of CD73 in cancer cells has been associated with low overall survival in patients with BC [86]. Overexpression of CD73 in the TME is due to hypoxia, which generates angiogenesis and the high rate of cell proliferation, which induces HIF-1α. This, therefore, regulates positively the genes of the purinergic pathway, such as CD73 and CD39 [175]. In addition, several other factors regulate the positive expression of CD73, such as low glucose and low pH, transforming growth factor TGF-β, interferons (INFs) type I, and tumor necrosis factor-α (TNF-α) (Fig. 1). Thus, it is stated that hypoxia and pro-inflammatory cytokines regulate this gene positively [96]. Studies have shown that blocking CD73, by means of monoclonal antibody and interference RNA (RNAi), has resulted in anti-metastatic effects in human BC cells [176, 177].
TNBC and CD73
The TNBC is very heterogeneous [178], and constitutes a very aggressive tumor, with an unfavorable prognosis and few therapeutic options [102, 179], in addition to greater chances of metastasis to vital organs, when compared to other types of BC [180]. The CD73 is inversely related to ER (the largest expression of ER downregulates the expression of CD73) [181], and in the case of TNBC, which does not express ER [180], a high expression of CD73 will be verified [182], which is is correlated with high risk of metastasis [182, 183], worse prognosis, and increased resistance to chemotherapy [184]. Anthracycline-based chemotherapy has been the cornerstone of TNBC treatment for many years, although elevated levels of CD73 are associated with increased resistance to doxorubicin, a widely used anthracycline chemotherapy [182]. Thus, the targeting of CD73 has emerged as a new therapeutic target for TNBC [102].
The TME of BC
In the TME of BC, several cytokines are observed, which favor a condition of chronic inflammation that allows tumor survival and progression [185]. Still, several innate and adaptive immunity cells can be found in the TME of BC [186, 187]. Another common feature found in solid tumors is the presence of hypoxia, caused by a decrease in nutrients and oxygen. Hypoxic TME favors the expression of molecules related to tumor survival and progression [188]. Hypoxia in BC TME is related to the phenotypic change in tumor cells, favoring the invasive and metastatic process [189].
Immune system in the TME of BC
The immune system is fundamental for the progression of cancer [190], and BC can stimulate the immune system [178, 191]. Neoplastic cells interact with immune cells to generate an immunosuppressed environment and favor angiogenesis, so that the development of the neoplasm occurs [84]. To prevent the progression of BC, there are intrinsic (cellular apoptosis) and extrinsic (immune system) mechanisms to contain the growth of cancerous breast cells [192, 193]. The immunosuppressed cells most expressed in breast tumors are regulatory T cells (Treg) and myeloid-derived suppressor cells (MDSCs) [194], in addition to Natural Killer (NK) cells, macrophages, neutrophils, dendritic cells, and T and B lymphocytes, which can also be expressed in the TME of BC [186, 187].
In BC, the interaction between the immune system and the tumor occurs in three stages: elimination, balance, and escape. In the first stage, the acute inflammatory response, initiated by angiogenesis, initiates the recruitment of innate immune cells in the TME, such as macrophages, NK cells, and dendritic cells. As a result, pro-inflammatory cytokines such as interleukin 12 (IL-12) and interferon γ (IFN-γ) are produced, which will activate more innate immune cells and promote the death of neoplastic cells. In addition, dendritic cells mature and move to lymph nodes, where they present antigens and activate TCD4+ and TCD8+ cells, both of which will act in the TME to promote death of the tumor cells [195]. In the second stage, the inflammation becomes chronic, and the tumor grows and evades immune surveillance. One of the factors that promote tumor growth is the change in immune response from TH1 lymphocytes (anti-tumorigenic) to TH2 lymphocytes (pro-tumorigenic) [196, 197]. Finally, in the escape stage, breast tumors grow and metastasize with complete autonomy [195]..
The immune cells that are responsible for eliminating BC cells are NK cells and cytotoxic T lymphocytes (CTLs) [194]. A reduction in NK cells was observed in patients with advanced BC [198]. Studies have shown that NK cells activated by IL-2 and IL-5 increased the effectiveness of the monoclonal antibody cetuximab against TNBC [199], that is, NK cells are an anti-tumor factor in BC treated with chemotherapy, in addition to suppressing the progression of human BC and preventing metastasis [194, 200]. Regarding Treg cells, it is known that they can suppress NK cells, TCD4+, TCD8+, antigen-presenting cells (APC), and B cells [201], thus being negative regulators of the anti-tumor immune response. Thus, one of the ways in which BC cells prevent death is immune evasion, as these neoplastic cells can prevent tumor recognition by NK cells and CTLs [194].
Purinergic signaling and immune system in BC
The accumulation of extracellular Ado by means of the CD73 has an immunosuppressive effect on immune system cells, generating a TME conducive to tumor growth [202, 203], that is, Ado promotes immune escape [86]. In addition to immunosuppression, Ado also decreases the activities of effector T cells by activating A2A receptors [204]. In TNBC, high levels of CD73 and A2B receptors are related to poor clinical results. This is because the CD73 adenosinergic pathway leads to tumor progression through suppression of anti-tumor immunity, one of the ways that leads to the spread of metastasis in the TNBC. Immune evasion in TNBC is also favored by A2A receptors, since they inhibit the cytotoxic function of T and NK cells. In view of these considerations, selectively inhibiting the CD73, A2A/A2B becomes a viable therapeutic option in the treatment of TNBC [102].
BC, hypoxia, and purinergic system
In the TME, hypoxia is a characteristic that favors the EMT, in which cells assume a more invasive phenotype with greater survivability [205]. Hypoxia in the TME is usually related to increased resistance to apoptosis, angiogenesis, and migration [206]. In BC MDA-MB-468 cells, it was observed that EMT, induced by epidermal growth factor (EGF) and hypoxia, alters Ca2+ influx and Ca2+ ATP-dependent signaling, which may be via P2X or P2Y receptors [189].
Hypoxia in MDA-MB-468 cells promotes lower responses to ATP, which is caused by an attenuation of the actions of Ca2+ in the intracellular space and a faster return to baseline levels of free Ca2+ in the cytoplasm [207]. It is believed that this reduction in responses to ATP by regulating intracellular Ca2+ occurs to prevent excessive responses to high levels of ATP in the TME, thus allowing greater migratory and escape capacity of the primary tumor [152].
In cases of hypoxia, an increase in the P2Y6 mRNA was observed in the MDA-MB-468 lineage. This increase was due to the stimulation of growth factors and EMT, since overexpression of P2Y6 is a common characteristic of BC cells that present the mesenchymal phenotype. The main change observed in P2Y6 silencing was the decrease in the vimentin protein, which is one of the markers of EMT in the MDA-MB-468 lineage [152].
In addition, this inhibition promoted a decrease in the migration of MDA-MB-231 cells [152]. As already mentioned, in the TME where there is hypoxia, the transcriptional inhibition of AK [158] and the increased expression of CD39 and CD73 occurs by HIF-1α, favoring an increase in extracellular levels of Ado [159]. The interaction of Ado with ARs can promote various responses in relation to BC, such as proliferation and invasion, and, in some cases, growth inhibition [96].
Therapeutic options
Traditional BC treatment includes mastectomy and/or lumpectomy associated with adjuvant postoperative treatment [33]. Approximately 18 months after surgery is when there is a peak of metastasis, probably because the secondary sites were dormant due to an inhibition of the primary tumor site. When the inhibitory effects of the primary tumor are removed, secondary lesions begin to proliferate [85].
Considering the vast involvement of the purinergic system in the maintenance and metastatic process of BC, several members of this system have become targets for possible therapies to control this neoplasm [38, 115, 208].
Therapeutic options related to P2X receptors
In P2X receptors, the ATP/P2X7 pathway proved to be clinically important, especially for the BC treatment with anthracyclines [115]. The pharmacological silencing or blocking of P2X7 [38] has been shown to inhibit metastatic invasion and dissemination in experimental BC studies [112]. In addition, there was an influence of the drug ivermectin, an anti-parasitic, on TNBC cells, observed in studies with human MDA-MB-231, MCF-7, and SKBR3 lineages, and with 4T1.2 Balb/c triple negative mice BC cells. Ivermectin promoted cell death in mice and humans lineages with TNBC, through purinergic signaling via P2X4/P2X7/Pannexin-1 associated with caspases 1 and 3 (Table 2) [209].
Table 2.
Treatment possibilities for BC targeting the purinergic system, and its clinical outcomes
| Treatment possibility | Analysis phase | Effects on BC control | References |
|---|---|---|---|
| Ivermectin | Preclinical studies with the human MDA-MB-231, MCF-7, and SKBR3 lineages, and in 4T1.2 Balb/c triple negative mice breast cancer cells | Promotes cell death in mice and humans lineages through P2X4/P2X7/Pannexin-1 associated with caspases 1 and 3 | [209] |
| Ticagrelor | Preclinical studies with the MDA-MB-468, MDA-MB-231, and MCF-7 lineages | Promotes P2Y12 blockade, decreased the interaction between human breast tumor cells and platelets, decreasing the metastatic process | [120] |
| Non-selective AR antagonists - aminophylline (AMO) - and selective A2B antagonists (ATL801) | Preclinical studies with metastatic cells 4T1.12B - studies in Balb/c mice. | Growth retardation of 4T1.12B breast cancer cells and their lung metastasis, by blocking AR | [210] |
| A2A antagonist (CPI-444) with anti-PD1 mAb | Preclinical studies with metastatic 4T1.2 cells in Balb/c and C57BL/6 mice | TNBC metastasis reduction | [173] |
| Monoclonal antibody IE9 and 7G2 | In vitro T lymphocytes treatment | Inhibition of cell-bound CD73 enzymatic activity. IE9 is able to induce lymphocyte proliferation when combined with other drugs | [211] |
| Monoclonal antibody 4G4 | Preclinical studies in mice | Performs an inhibition of platelet aggregation onto endothelial cells and a weaker inhibition of CD73 | [212] |
| Monoclonal antibody AD2 | Preclinical studies with MDA-MB-231 lineage | Promotes an internalization of CD73, reducing its activity. | [176] |
| Monoclonal antibody 3F7 | Preclinical studies with human MDA-MB-231 and MDA-MB-468, and in mouse 4T1 cell lines | Endothelial cells migration inhibition, and is able to bind to CD73, decreasing its activity | [213] |
| Suppression of CD73 by shRNA | Preclinical studies with MDA-MB-231 and MCF-7 cell lines | By suppressing CD73, it prevents the proliferation of MDA-MB-231 cells, inducing apoptosis and arrest of the cell cycle | [177] |
| Specific inhibitor of CD73 activity (APCP) | Preclinical studies with MDA-MB-231 and MCF-7 cell lines | By suppressing CD73, it prevents the proliferation of MDA-MB-231 cells | [177] |
Therapeutic options related to P2Y receptors
Among P2Y receptors, the correlation between UTP and P2Y6 appears as an interesting therapeutic possibility. This is because extracellular UTP acts as a regulator of tumor formation, progression, and escape, being an important therapeutic target associated with the control and prevention of the activation of signaling pathways related to metastasis and drug resistance [119]. Still, in vitro studies with MDA-MB-468, MDA-MB-231, and MCF-7 cell lines indicated that ticagrelor, a P2Y12 blocker, decreased the interaction between human breast tumor cells and platelets, influencing the metastatic process (Table 2) [120].
Therapeutic options related to P1 receptors
Studies have been done with P1 receptor antagonists, allowing researchers to recognize which of them could be used in the clinic, favoring the control of BC [98]. In this context, studies carried out on the MCF-7 and MDA-MB-438 strains have found that the use of the adenosine deaminase inhibitor has made adenosine toxic to these lineages, inducing cells to the mitochondrial apoptotic pathway [214]. Furthermore, the exposure of MCF-7 cells to 1,3-dipropyl-8-cyclopentylxanthine (DPCPX), an A1 receptor antagonist, led to an increase in the expression of p53 and caspases [104] and decreased tumor growth by inhibiting ERα [121]. Finally, the depletion of A1 receptors by small interfering RNA (siRNA) in the MDA-MB-468 lineage caused impaired G1 checkpoint, leading to an accumulation of tumor cells in phase G2/M, resulting in a decrease in proliferation and an increase in apoptosis [215].
Regarding A2 receptors, the studies by Cekic et al. [210] found that the exposure of 4T1 tumor cells (ER negative) to N-(5-(1-cyclopropyl-2,6-dioxo-3-propyl-2,3,6,7-tetrahydro-1H-purin-8-yl)pyridin-2-yl)-N-ethyl-6-nicotinamide (ATL801), an A2B receptor antagonist, slowed BC growth in mice, in addition to decreasing lung metastasis by 85%. Therefore, the suppression of A2B promotes the inhibition of invasiveness, migration, and cell growth of ER negative breast cancer lines [107].
Finally, activation of the A3 receptor by exogenous adenosine or by the agonist adenosine-5-Nmethyluronamide (IB-MECA) significantly decreased the migration and motility of the cell line MDA-MB-231 [208]. Going further, the exposure of the MCF-7 and MDA-MB-231 lineages to 2-chloro-N(6)-(3-iodobenzyl)-5′-N-methylcarbamoyl-4′-thioadenosine (LJ-529), a A3 receptor agonist, induced apoptosis and altered the Wnt/β-catenin signaling pathway, leading to decreased proliferation and growth of tumor cells [109].
Therapeutic options related to modulation of CD73 by ARs
Considering the aspects of TNBC, it is possible to infer that it still represents a great challenge in clinical practice, requiring the development of new therapeutic targets [216]. From this, modulation of CD73 and A2A/A2B receptors in the TME becomes a therapeutic option that can inhibit tumor growth and improve anti-tumor immune responses, including in TNBC [86, 173], in order to increase anthracycline activity against tumor cells [182]. Some data showed that targeting the A2A receptor increases the frequency of activation of immune cells and their infiltration into tumors, so much so that 41% of TNBC patients in a study where they were treated with the A2A antagonist CPI-444 had control of the neoplasm (Table 2) [173].
The TNBC metastasis occurs to the lungs by the expression of CD73, through the stimulation of A2B receptors [102, 107, 217, 218]. Although the four ARs are related to an adaptive response to hypoxia, only A2A and A2B receptors are involved in tumor angiogenesis, by the action of vascular endothelial growth factor (VEGF) and basic fibroblast growth factor (bFGF) (Fig. 1) [219]. Still, studies by Cekic et al. [210] demonstrated that the blockade of A2A inhibits angiogenesis in BC and prevents metastasis. In the 4T1.2 mammary tumor cells of mice, A2B activation promoted metastasis in vivo and chemotaxis in vitro [218]. Some studies have shown that the injection of non-selective adenosine-aminophylline (AMO) receptors, and selective A2B antagonists (ATL801), was effective in retarding the growth of 4T1.12B BC cells and their lung metastasis (Table 2) [210].
In addition, studies have evaluated the combination of some Ado receptors with an immune checkpoint inhibitor, such as PD-1 and PD-L1 [173] as an anti-cancer strategy [64]. As a result, A2A receptors with the anti-PD-1 mAb were important in reducing metastasis in 4T1.2 breast carcinomas [220].
Therapeutic options related to anti-CD73 mAb
It is known that CD73 is related to a worse prognosis in TNBC and HER2 positive BC [123]; therefore, studies in mice found that silencing the CD73 gene in BC cells led to a decrease in levels of focal adhesion kinase (FAK), which is related to cells adhesion and migration. The use of anti-CD73 mAb slows the growth of cancer cells and TNBC metastasis [102, 221] to the lung [218], in addition to reducing tumor angiogenesis in mouse models with BC [219].
Four anti-CD73 mAb have already been developed and can influence enzyme activity, promote the internalization of CD73 in metastatic cells, and decrease its levels in lymphocytes [102]. The mAbs are IE9 (capable of inhibiting the enzyme activity of CD73 bound to the cell, and inducing lymphocyte proliferation when combined with other drugs), 7G2 (also capable of inhibiting the enzymatic activity of CD73 bound to the cell) [211], 4G4 (performs a not so potent inhibition of CD73, but performs an inhibition of platelet aggregation in mice endothelial cells) [212], and AD2 (promotes an internalization of CD73, reducing its activity and metastasis potential in the MDA-MB-231 lineage) [176] (Table 2). In addition to reducing tumor growth, these CD73 inhibitors favor immune activity [102]. The mAb 3F7 was developed from studies with human lineages MDA-MB231 and MDA-MB-468, and with the mouse 4T1 cell line. The 3F7 is related to the reduction of tumor growth, through the inhibition of endothelial cell migration and ability to bind to CD73. Moreover, 3F7 has been shown to increase the cytotoxic effects of doxorubicin treatment on TNBC (Table 2) [213].
Therapeutic options related to CD73 suppression
Suppression of CD73 by short hairpin RNA (shRNA) also prevents the proliferation of cancerous breast cells (MDA-MB-231) by inducing apoptosis and interrupting the cell cycle (Table 2) [177]. The same study also demonstrated that it is possible to inhibit the proliferation of breast tumor cells using α, β-methylene adenosine-5′-disphosphate (APCP), a specific inhibitor of CD73 activity (Table 2). Therefore, modulation of CD73 and Ado, derived from the TME, is established as an innovative therapeutic option to limit tumor progression, improve anti-tumor immune responses, and prevent immunological deviations [86].
Conclusions
Considering that BC is the most frequent cause of cancer deaths among women, its study is extremely relevant to the world health scenario. Thus, the understanding of the pathophysiological pathways involved in its development and the relationship with the purinergic system are the basis of future therapeutic options, since purinergic receptors may be involved in the progression and invasion of BC cells, as well as promoting inhibition and control of tumor growth. Thus, several members of the purinergic system have become promising therapeutic options.
From the literature review developed, it was evidenced that the purinergic system is strongly involved with the pathophysiology of BC. Through different intracellular signaling pathways, purinergic receptors promote actions related to the progression and invasion of different BC lineages, which in practice correspond to the intrinsic subtypes of this tumor. In addition, in some cases, its activation may promote the control and inhibition of tumor growth. Also, the ectonucleotidases CD39 and CD73 proved to be closely related to BC, with a greater emphasis on CD73, which participates in pro- and anti-inflammatory control of the TME, in addition to being directly related to the prognosis of TNBC.
In relation specifically to purinergic receptors, it is known that the silencing or pharmacological blocking of P2X7 [38] is a viable option, as it has been shown to inhibit metastatic invasion and dissemination of BC [112]. As for P2Y, the prospects for the correlation between UTP/P2Y6 [119] and UTP/P2Y2 are favorable [118], as well as P2Y12 blockers, such as ticagrelor [120]. As for P1 receptors, A1 inhibition favors apoptosis, A2B antagonists decreased BC metastasis to the lung, and A3 agonists led to decreased growth and proliferation of cancerous breast cells [98], which demonstrates the fundamental role of purinergic receptors in BC control.
In addition, in relation to TNBC, the modulation of CD73 and A2A/A2B Ado receptors in TME is a future bet for the treatment of this type of cancer [86, 173]. This is because the targeting of the A2A receptor increases the infiltration of immune cells in the tumor [173], and its blockade inhibits angiogenesis and prevents metastasis [210], as well as A2B blockade also demonstrated inhibition of lung metastasis in TNBC. The future for the treatment of this type of cancer may also be the combination of Ado targets (such as A2A) with an anti-PD-1 monoclonal antibody to reduce metastasis [220].
Ectonucleotidases are also promising. Silencing CD73 demonstrated to be important to decrease the migration and adhesion of BC cells. The use of anti-CD73 mAb (IE9, 7G2, 4G4, AD2, and 3F7), despite being analyzed for some years, is still a favorable strategy, as it reduces the growth of cancer cells and TNBC metastasis to the lungs [102, 221], also reducing BC angiogenesis [219]. Finally, suppression of CD73 by shRNA and APCP also prevents the proliferation of cancerous breast cells [177]. Therefore, modulation of CD73 and Ado is established as an innovative therapeutic option to limit neoplastic progression, to favor anti-tumor immune responses, and to prevent immunological deviations [86]. Thus, it is stated that the whole purinergic pathway is an extremely important therapeutic intervention option in the current and future treatment of BC.
Therefore, several therapeutic possibilities arise within the purinergic system. Some drugs with applications other than oncology showed actions on cell death involving the purinergic system, such as ivermectin. Going further, there are some drugs that have been developed to block certain pathways or actions of receptors and enzymes of the purinergic system, such as anti-CD73 mAbs. In order to further advance in the knowledge of new therapies and to better understand the applicability of the drugs already developed, it is necessary that even more studies be carried out in animal models and in clinical trials to validate selectivity of action in BC, as well as its effectiveness. Therefore, it is undeniable that the purinergic system is an extremely important intervention pathway for the treatment of BC, and has the perspective of improving the prognosis and quality of life of patients.
Code availability
Not applicable.
Julia Beatrice de Araújo
Academic of the Graduation Course in Medicine at the Federal University of Fronteira Sul - Campus Chapecó. Member of the Research Group “Physical Exercise and Complementary Integrative Practices in Health (PICs) as Tools for Non-Pharmacological Treatments in Chronic Health Conditions”; member of the Extension Group “Production and Dissemination of Educational Materials on Coronavirus and its Impacts on Health”; Vice-Coordinator of the Academic Neuroscience League; Vice-President of the Women’s Health Academic League - 2020.
Authors’ contributions
The suggestion of the theme addressed was made by Débora Tavares de Resende e Silva and Sarah Franco Vieira de Oliveira Maciel. The literature review and development of images and tables was carried out by Julia Beatrice de Araújo and Vanessa Vitória Kerkhoff. The critical review of the article was carried out by Débora Tavares de Resende e Silva and Sarah Franco Vieira de Oliveira Maciel.
Data availability
Not applicable.
Compliance with ethical standards
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
This article does not contain any studies with humans or animal participants performed by any of the authors.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Harbeck N, Gnant M. Breast cancer. Lancet. 2017;389:1134–1150. doi: 10.1016/S0140-6736(16)31891-8. [DOI] [PubMed] [Google Scholar]
- 2.Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65:87–108. doi: 10.3322/caac.21262. [DOI] [PubMed] [Google Scholar]
- 3.Winters S, Martin C, Murphy D, Shokar NK. Breast cancer epidemiology, prevention, and screening. Prog Mol Biol Transl Sci. 2017;151:1–32. doi: 10.1016/bs.pmbts.2017.07.002. [DOI] [PubMed] [Google Scholar]
- 4.Globocan (2019) Cancer today. https://gco.iarc.fr/today/home.
- 5.Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, Parkin DM, Forman D, Bray F. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2015;136:E359–E386. doi: 10.1002/ijc.29210. [DOI] [PubMed] [Google Scholar]
- 6.DeSantis CE, Fedewa SA, Goding Sauer A, Kramer JL, Smith RA, Jemal A. Breast cancer statistics, 2015: convergence of incidence rates between black and white women. CA Cancer J Clin. 2016;66:31–42. doi: 10.3322/caac.21320. [DOI] [PubMed] [Google Scholar]
- 7.Siegel RL, Miller KD, Jemal A. Cancer statistics, 2020. CA Cancer J Clin. 2019;70:7–30. doi: 10.3322/caac.21590. [DOI] [PubMed] [Google Scholar]
- 8.Anastasiadi Z, Lianos GD, Ignatiadou E, Harissis HV, Mitsis M. Breast cancer in young women: an overview. Updat Surg. 2017;69:313–317. doi: 10.1007/s13304-017-0424-1. [DOI] [PubMed] [Google Scholar]
- 9.Chen WY (2020) Factors that modify breast cancer risk in women. UpToDate. https://www.uptodate.com/contents/factors-that-modify-breast-cancer-risk-in-women. Accessed 18 May 2020
- 10.Iqbal J, Ginsburg O, Rochon PA, Sun P, Narod SA. Differences in breast cancer stage at diagnosis and cancer-specific survival by race and ethnicity in the United States. JAMA. 2015;313:165–173. doi: 10.1001/jama.2014.17322. [DOI] [PubMed] [Google Scholar]
- 11.Tao L, Gomez SL, Keegan TH, Kurian AW, Clarke CA. Breast cancer mortality in African-American and non-Hispanic white women by molecular subtype and stage at diagnosis: a population-based study. Cancer Epidemiol Prev. 2015;24:1039–1045. doi: 10.1158/1055-9965.EPI-15-0243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Blows FM, Driver KE, Schmidt MK, Broeks A, van Leeuwen FE, Wesseling J, Cheang MC, Gelmon K, Nielsen TO, Blomqvist C, Heikkilä P, Heikkinen T, Nevanlinna H, Akslen LA, Bégin LR, Foulkes WD, Couch FJ, Wang X, Cafourek V, Olson JE, Baglietto L, Giles GG, Severi G, McLean CA, Southey MC, Rakha E, Green AR, Ellis IO, Sherman ME, Lissowska J, Anderson WF, Cox A, Cross SS, Reed MWR, Provenzano E, Dawson SJ, Dunning AM, Humphreys M, Easton DF, García-Closas M, Caldas C, Pharoah PD, Huntsman D. Subtyping of breast cancer by immunohistochemistry to investigate a relationship between subtype and short and long term survival: a collaborative analysis of data for 10,159 cases from 12 studies. PLoS Med. 2010;7(5):e1000279. doi: 10.1371/journal.pmed.1000279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Perou CM, Børresen-Dale AL. Systems biology and genomics of breast cancer. Cold Spring Harb Perspect Biol. 2011;3(2):a003293. doi: 10.1101/cshperspect.a003293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lahmann PH, Hoffmann K, Allen N, van Gils CH, Khaw KT, Tehard B, Berrino F, Tjønneland A, Bigaard J, Olsen A, Overvad K, Clavel-Chapelon F, Nagel G, Boeing H, Trichopoulos D, Economou G, Bellos G, Palli D, Tumino R, Panico S, Sacerdote C, Krogh V, Peeters PHM, Bueno-de-Mesquita HB, Lund E, Ardanaz E, Amiano P, Pera G, Quirós JR, Martínez C, Tormo MJ, Wirfält E, Berglund G, Hallmans G, Key TJ, Reeves G, Bingham S, Norat T, Biessy C, Kaaks R, Riboli E. Body size and breast cancer risk: findings from the European Prospective Investigation into cancer And Nutrition (EPIC) Int J Cancer. 2004;111:762–771. doi: 10.1002/ijc.20315. [DOI] [PubMed] [Google Scholar]
- 15.Lauby-Secretan B, Scoccianti C, Loomis D, Grosse Y, Bianchini F, Straif K. Body fatness and cancer—viewpoint of the IARC working group. N Engl J Med. 2016;375:794–798. doi: 10.1056/NEJMsr1606602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Key TJ, Appleby PN, Reeves GK, Travis RC, Brinton LA, Helzlsouer KJ, Dorgan JF, Gapstur SM, Gaudet MM, Kaaks R, Riboli E, Rinaldi S, Manjer J, Hallmans G, Giles GG, le Marchand L, Kolonel LN, Henderson BE, Tworoger SS, Hankinson SE, Zeleniuch-Jacquotte A, Koenig K, Krogh V, Sieri S, Muti P, Ziegler RG, Schairer C, Fuhrman BJ, Barrett-Connor E, Laughlin GA, Grant EJ, Cologne J, Ohishi W, Hida A, Cauley JA, Fourkala EO, Menon U, Rohan TE, Strickler HD, Gunter MJ, Endogenous Hormones and Breast Cancer Collaborative Group Steroid hormone measurements from different types of assays in relation to body mass index and breast cancer risk in postmenopausal women: reanalysis of eighteen prospective studies. Steroids. 2015;99:49–55. doi: 10.1016/j.steroids.2014.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Sieri S, Krogh V, Bolelli G, Abagnato CA, Grioni S, Pala V, Evangelista A, Allemani C, Micheli A, Tagliabue G, Schunemann HJ, Menard S, Berrino F, Muti P. Sex hormone levels, breast cancer risk, and cancer receptor status in postmenopausal women: the ORDET cohort. Cancer Epidemiol Prev. 2009;18:169–176. doi: 10.1158/1055-9965.EPI-08-0808. [DOI] [PubMed] [Google Scholar]
- 18.Ritte R, Lukanova A, Tjønneland A, Olsen A, Overvad K, Mesrine S, Fagherazzi G, Dossus L, Teucher B, Steindorf K, Boeing H, Aleksandrova K, Trichopoulou A, Lagiou P, Trichopoulos D, Palli D, Grioni S, Mattiello A, Tumino R, Sacerdote C, Quirós JR, Buckland G, Molina-Montes E, Chirlaque MD, Ardanaz E, Amiano P, Bueno-de-Mesquita B, van Duijnhoven F, van Gils CH, Peeters PHM, Wareham N, Khaw KT, Key TJ, Travis RC, Krum-Hansen S, Gram IT, Lund E, Sund M, Andersson A, Romieu I, Rinaldi S, McCormack V, Riboli E, Kaaks R. Height, age at menarche and risk of hormone receptor-positive and-negative breast cancer: a cohort study. Int J Cancer. 2013;132:2619–2629. doi: 10.1002/ijc.27913. [DOI] [PubMed] [Google Scholar]
- 19.Colditz GA, Rosner B. Cumulative risk of breast cancer to age 70 years according to risk factor status: data from the Nurses’ Health Study. Am J Epidemiol. 2000;152:950–964. doi: 10.1093/aje/152.10.950. [DOI] [PubMed] [Google Scholar]
- 20.Kelsey JL, Gammon MD, John EM. Reproductive factors and breast cancer. Epidemiol Rev. 1993;15:36–47. doi: 10.1093/oxfordjournals.epirev.a036115. [DOI] [PubMed] [Google Scholar]
- 21.Bagnardi V, Rota M, Botteri E, Tramacere I, Islami F, Fedirko V, Scotti L, Jenab M, Turati F, Pasquali E, Pelucchi C, Galeone C, Bellocco R, Negri E, Corrao G, Boffetta P, la Vecchia C. Alcohol consumption and site-specific cancer risk: a comprehensive dose–response meta-analysis. Br J Cancer. 2015;112:580–593. doi: 10.1038/bjc.2014.579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Cao Y, Willett WC, Rimm EB, Stampfer MJ, Giovannucci EL. Light to moderate intake of alcohol, drinking patterns, and risk of cancer: results from two prospective US cohort studies. BMJ. 2015;351:h4238. doi: 10.1136/bmj.h4238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Chen WY, Rosner B, Hankinson SE, Colditz GA, Willett WC. Moderate alcohol consumption during adult life, drinking patterns, and breast cancer risk. JAMA. 2011;306:1884–1890. doi: 10.1001/jama.2011.1590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.White AJ, DeRoo LA, Weinberg CR, Sandler DP. Lifetime alcohol intake, binge drinking behaviors, and breast cancer risk. Am J Epidemiol. 2017;186:541–549. doi: 10.1093/aje/kwx118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Gaudet MM, Carter BD, Brinton LA, Falk RT, Gram IT, Luo J, Milne RL, Nyante SJ, Weiderpass E, Beane Freeman LE, Sandler DP, Robien K, Anderson KE, Giles GG, Chen WY, Feskanich D, Braaten T, Isaacs C, Butler LM, Koh WP, Wolk A, Adami HO, White E, Margolis KL, Thun MJ, Gapstur SM. Pooled analysis of active cigarette smoking and invasive breast cancer risk in 14 cohort studies. Int J Epidemiol. 2017;46:881–893. doi: 10.1093/ije/dyw288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Gram IT, Park SY, Kolonel LN, Maskarinec G, Wilkens LR, Henderson BE, Le Marchand L. Smoking and risk of breast cancer in a racially/ethnically diverse population of mainly women who do not drink alcohol: the MEC study. Am J Epidemiol. 2015;182:917–925. doi: 10.1093/aje/kwv092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Johnson KC, Miller AB, Collishaw NE, Palmer JR, Hammond SK, Salmon AG, Cantor KP, Miller MD, Boyd NF, Millar J, Turcotte F. Active smoking and secondhand smoke increase breast cancer risk: the report of the Canadian Expert Panel on Tobacco Smoke and Breast Cancer Risk (2009) Tob Control. 2011;20(1):e2. doi: 10.1136/tc.2010.035931. [DOI] [PubMed] [Google Scholar]
- 28.Stuebe AM, Willett WC, Xue F, Michels KB. Lactation and incidence of premenopausal breast cancer: a longitudinal study. Arch Intern Med. 2009;169:1364–1371. doi: 10.1001/archinternmed.2009.231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hildebrand JS, Gapstur SM, Campbell PT, Gaudet MM, Patel AV. Recreational physical activity and leisure-time sitting in relation to postmenopausal breast cancer risk. Cancer Epidemiol Biomark Prev. 2013;22:1906–1912. doi: 10.1158/1055-9965.EPI-13-0407. [DOI] [PubMed] [Google Scholar]
- 30.Braithwaite D, Miglioretti DL, Zhu W, Demb J, Trentham-Dietz A, Sprague B, Tice JA, Onega T, Henderson LM, Buist DSM, Ziv E, Walter LC, Kerlikowske K, for the Breast Cancer Surveillance Consortium Family history and breast cancer risk among older women in the breast cancer surveillance consortium cohort. JAMA Intern Med. 2018;178:494–501. doi: 10.1001/jamainternmed.2017.8642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Reiner AS, Sisti J, John EM, Lynch CF, Brooks JD, Mellemkjær L, Boice JD, Knight JA, Concannon P, Capanu M, Tischkowitz M, Robson M, Liang X, Woods M, Conti DV, Duggan D, Shore R, Stram DO, Thomas DC, Malone KE, Bernstein L, The WECARE Study Collaborative Group. Bernstein JL. Breast cancer family history and contralateral breast cancer risk in young women: an update from the women’s environmental cancer and radiation epidemiology study. J Clin Oncol. 2018;36:1513–1520. doi: 10.1200/JCO.2017.77.3424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Bhat V, Allan AL, Raouf A. Role of the microenvironment in regulating normal and cancer stem cell activity: implications for breast cancer progression and therapy response. Cancers. 2019;11:1240. doi: 10.3390/cancers11091240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Brasil (2020) Ministério da Saúde. Instituto Nacional de Câncer. https://www.inca.gov.br/assuntos/ministerio-da-saude. Accessed 21 Mar 2020
- 34.Hirschey MD, DeBerardinis RJ, Diehl A, Drew JE, Frezza C, Green MF, Jones LW, Ko YH, le A, Lea MA, Locasale JW, Longo VD, Lyssiotis CA, McDonnell E, Mehrmohamadi M, Michelotti G, Muralidhar V, Murphy MP, Pedersen PL, Poore B, Raffaghello L, Rathmell JC, Sivanand S, Vander Heiden MG, Wellen KE, Target Validation Team Dysregulated metabolism contributes to oncogenesis. Semin Cancer Biol. 2015;35:129–150. doi: 10.1016/j.semcancer.2015.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Scully OJ, Bay BH, Yip G, Yu Y. Breast cancer metastasis. Cancer Genomics Proteomics. 2012;9:311–320. [PubMed] [Google Scholar]
- 36.Giuliano AE, Connolly JL, Edge SB, Mittendorf EA, Rugo HS, Solin LJ, Weaver DL, Winchester DJ, Hortobagyi GN. Breast cancer—major changes in the American Joint Committee on Cancer eighth edition cancer staging manual. CA J Clin. 2017;67:290–303. doi: 10.3322/caac.21393. [DOI] [PubMed] [Google Scholar]
- 37.Schwartz RS, Erban JK. Timing of metastasis in breast cancer. N Engl J Med. 2017;376:2486–2488. doi: 10.1056/NEJMcibr1701388. [DOI] [PubMed] [Google Scholar]
- 38.Di Virgilio F, Sarti AC, Falzoni S, De Marchi E, Adinolfi E. Extracellular ATP and P2 purinergic signalling in the tumour microenvironment. Nat Rev Cancer. 2018;18:601–618. doi: 10.1038/s41568-018-0037-0. [DOI] [PubMed] [Google Scholar]
- 39.Dieci MV, Orvieto E, Dominici M, Conte P, Guarneri V. Rare breast cancer subtypes: histological, molecular, and clinical peculiarities. Oncologist. 2014;19:805–813. doi: 10.1634/theoncologist.2014-0108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ortiz AG, Sanchez-Muñoz A, Chica Parrado MR, Álvarez M, Ribelles N, Rueda Dominguez A, Alba E. Deciphering HER2 breast cancer disease: biological and clinical implications. Front Oncol. 2019;9:1124. doi: 10.3389/fonc.2019.01124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Serra KP, Ramalho S, Torresan R, Vassallo J, Sarian LO, Silva GR, Derchain S. The new classification of breast cancers: finding the luminal A. Rev Bras Ginecol Obstet. 2014;36:575–580. doi: 10.1590/so100-720320140005158. [DOI] [PubMed] [Google Scholar]
- 42.Melichar B. Biomarkers in the treatment of cancer: opportunities and pitfalls. Clin Chem Lab Med. 2013;51:1329–1333. doi: 10.1515/cclm-2013-0323. [DOI] [PubMed] [Google Scholar]
- 43.Trop I, LeBlanc SM, David J, Lalonde L, Tran-Thanh D, Labelle M, El Khoury MM. Molecular classification of infiltrating breast cancer: toward personalized therapy. Radiographics. 2014;34:1178–1195. doi: 10.1148/rg.345130049. [DOI] [PubMed] [Google Scholar]
- 44.Sørlie T, Perou CM, Tibshirani R, et al. Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc Natl Acad Sci U S A. 2001;98:10869–10874. doi: 10.1073/pnas.191367098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Kennecke H, Yerushalmi R, Woods R, Cheang MCU, Voduc D, Speers CH, Nielsen TO, Gelmon K. Metastatic behavior of breast cancer subtypes. J Clin Oncol. 2010;28:3271–3277. doi: 10.1200/JCO.2009.25.9820. [DOI] [PubMed] [Google Scholar]
- 46.Voduc KD, Cheang MC, Tyldesley S, Gelmon K, Nielsen TO. Kennecke H. Breast cancer subtypes and the risk of local and regional relapse. J Clin Oncol. 2010;28:1684–1691. doi: 10.1200/JCO.2009.24.9284. [DOI] [PubMed] [Google Scholar]
- 47.Weigel MT, Dowsett M. Current and emerging biomarkers in breast cancer: prognosis and prediction. Endocr Relat Cancer. 2010;17(4):R245–R262. doi: 10.1677/ERC-10-0136. [DOI] [PubMed] [Google Scholar]
- 48.Kast K, Link T, Friedrich K, Petzold A, Niedostatek A, Schoffer O, Werner C, Klug SJ, Werner A, Gatzweiler A, Richter B, Baretton G, Wimberger P. Impact of breast cancer subtypes and patterns of metastasis on outcome. Breast Cancer Res Treat. 2015;150:621–629. doi: 10.1007/s10549-015-3341-3. [DOI] [PubMed] [Google Scholar]
- 49.Bonilla JM, Tabanera MT, Mendoza LR. Breast cancer in the 21st century: from early detection to new therapies. Radiología. 2017;59:368–379. doi: 10.1016/j.rx.2017.06.003. [DOI] [PubMed] [Google Scholar]
- 50.Sørlie T, Tibshirani R, Parker J, Hastie T, Marron JS, Nobel A, Deng S, Johnsen H, Pesich R, Geisler S, Demeter J, Perou CM, Lønning PE, Brown PO, Børresen-Dale AL, Botstein D. Repeated observation of breast tumor subtypes in independent gene expression data sets. Proc Natl Acad Sci U S A. 2003;100:8418–8423. doi: 10.1073/pnas.0932692100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Cianfrocca M, Goldstein LJ. Prognostic and predictive factors in early-stage breast cancer. Oncologist. 2004;9:606–616. doi: 10.1634/theoncologist.9-6-606. [DOI] [PubMed] [Google Scholar]
- 52.Elias SG, Adams A, Wisner DJ, Esserman LJ, van’t Veer LJ, Mali WPTM, Gilhuijs KGA, Hylton NM. Imaging features of HER2 overexpression in breast cancer: a systematic review and meta-analysis. Cancer Epidemiol Biomark Prev. 2014;23:1464–1483. doi: 10.1158/1055-9965.EPI-13-1170. [DOI] [PubMed] [Google Scholar]
- 53.Youk JH, Son EJ, Chung J, Kim JA, Kim EK. Triple-negative invasive breast cancer on dynamic contrast-enhanced and diffusion-weighted MR imaging: comparison with other breast cancer subtypes. Eur Radiol. 2012;22:1724–1734. doi: 10.1007/s00330-012-2425-2. [DOI] [PubMed] [Google Scholar]
- 54.Constantinidou A, Smith I. Is there a case for anti-HER2 therapy without chemotherapy in early breast cancer? Breast. 2011;20:158–161. doi: 10.1016/S0960-9776(11)70316-2. [DOI] [PubMed] [Google Scholar]
- 55.Rakha EA, Reis-Filho JS, Ellis IO. Combinatorial biomarker expression in breast cancer. Breast Cancer Res Treat. 2010;120:293–308. doi: 10.1007/s10549-010-0746-x. [DOI] [PubMed] [Google Scholar]
- 56.Kreutzfeldt J, Rozeboom B, Dey N, De P. The trastuzumab era: current and upcoming targeted HER2+ breast cancer therapies. Am J Cancer Res. 2020;10(4):1045–1067. [PMC free article] [PubMed] [Google Scholar]
- 57.Podo F, Buydens LM, Degani H, Hilhorst R, Klipp E, Gribbestad IS, van Huffel S, van Laarhoven H, Luts J, Monleon D, Postma GJ, Schneiderhan-Marra N, Santoro F, Wouters H, Russnes HG, Sørlie T, Tagliabue E, Børresen-Dale AL, FEMME Consortium Triple-negative breast cancer: present challenges and new perspectives. Mol Oncol. 2010;4:209–229. doi: 10.1016/j.molonc.2010.04.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Goldhirsch A, Winer EP, Coates AS, Gelber RD, Piccart-Gebhart M, Thürlimann B, Senn HJ, Panel members Personalizing the treatment of women with early breast cancer: highlights of the St Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2013. Ann Oncol. 2013;24(9):2206–2223. doi: 10.1093/annonc/mdt303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Cardoso F, Senkus E, Costa A, Papadopoulos E, Aapro M, André F, Harbeck N, Aguilar Lopez B, Barrios CH, Bergh J, Biganzoli L, Boers-Doets CB, Cardoso MJ, Carey LA, Cortés J, Curigliano G, Diéras V, el Saghir NS, Eniu A, Fallowfield L, Francis PA, Gelmon K, Johnston SRD, Kaufman B, Koppikar S, Krop IE, Mayer M, Nakigudde G, Offersen BV, Ohno S, Pagani O, Paluch-Shimon S, Penault-Llorca F, Prat A, Rugo HS, Sledge GW, Spence D, Thomssen C, Vorobiof DA, Xu B, Norton L, Winer EP. 4th ESO-ESMO International Consensus Guidelines for Advanced Breast Cancer (ABC 4)†. Ann Oncol. 2018;29:1634–1657. doi: 10.1093/annonc/mdy192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Irvin WJ, Jr, Carey LA. What is triple-negative breast cancer? Eur J Cancer. 2008;44:2799–2805. doi: 10.1016/j.ejca.2008.09.034. [DOI] [PubMed] [Google Scholar]
- 61.Le Du F, Perrin C, Brunot A, et al. Therapeutic innovations in breast cancer. Presse Med. 2019;48:1131–1137. doi: 10.1016/j.lpm.2019.04.005. [DOI] [PubMed] [Google Scholar]
- 62.Khosravi-Shahi P, Cabezón-Gutiérrez L, Aparicio Salcedo MI. State of art of advanced triple negative breast cancer. Breast J. 2019;25:967–970. doi: 10.1111/tbj.13369. [DOI] [PubMed] [Google Scholar]
- 63.Adams S, Loi S, Toppmeyer D, Cescon DW, de Laurentiis M, Nanda R, Winer EP, Mukai H, Tamura K, Armstrong A, Liu MC, Iwata H, Ryvo L, Wimberger P, Rugo HS, Tan AR, Jia L, Ding Y, Karantza V, Schmid P. Pembrolizumab monotherapy for previously untreated, PD-L1-positive, metastatic triple-negative breast cancer: cohort B of the phase II KEYNOTE-086 study. Ann Oncol. 2019;30:405–411. doi: 10.1093/annonc/mdy518. [DOI] [PubMed] [Google Scholar]
- 64.Nanda R, Chow LQ, Dees EC et al (2016) Pembrolizumab in patients with advanced triple-negative breast cancer: phase Ib KEYNOTE-012 study. J Clin Oncol 34:2460–2467. https://doi.org/doi:10.1200/JCO.2015.64.8931. 10.1200/JCO.2015.64.8931 [DOI] [PMC free article] [PubMed]
- 65.Dirix LY, Takacs I, Nikolinakos P, et al. Abstract S1-04: Avelumab (MSB0010718C), an anti-PD-L1 antibody, in patients with locally advanced or metastatic breast cancer: a phase Ib JAVELIN solid tumor trial. Breast Cancer Res Treat. 2018;167:671–686. doi: 10.1007/s10549-017-4537-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Koren S, Bentires-Alj M. Breast tumor heterogeneity: source of fitness, hurdle for therapy. Mol Cell. 2015;60:537–546. doi: 10.1016/j.molcel.2015.10.031. [DOI] [PubMed] [Google Scholar]
- 67.Al-Hajj M, Wicha MS, Benito-Hernandez A, Morrison SJ, Clarke MF. Prospective identification of tumorigenic breast cancer cells. Proc Natl Acad Sci U S A. 2003;100:3983–3988. doi: 10.1073/pnas.0530291100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Ginestier C, Hur MH, Charafe-Jauffret E, Monville F, Dutcher J, Brown M, Jacquemier J, Viens P, Kleer CG, Liu S, Schott A, Hayes D, Birnbaum D, Wicha MS, Dontu G. ALDH1 is a marker of normal and malignant human mammary stem cells and a predictor of poor clinical outcome. Cell Stem Cell. 2007;1:555–567. doi: 10.1016/j.stem.2007.08.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Wright MH, Calcagno AM, Salcido CD, Carlson MD, Ambudkar SV, Varticovski L. Brca1 breast tumors contain distinct CD44+/CD24- and CD133+ cells with cancer stem cell characteristics. Breast Cancer Res. 2008;10:R10. doi: 10.1186/bcr1855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Hwang-Verslues WW, Kuo WH, Chang PH. Multiple lineages of human breast cancer stem/progenitor cells identified by profiling with stem cell markers. PLoS One. 2009;4:e8377. doi: 10.1371/journal.pone.0008377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Tsang JY, Huang YH, Luo MH. Cancer stem cell markers are associated with adverse biomarker profiles and molecular subtypes of breast cancer. Breast Cancer Res Treat. 2012;136:407–417. doi: 10.1007/s10549-012-2271-6. [DOI] [PubMed] [Google Scholar]
- 72.Phillips TM, McBride WH, Pajonk F. The response of CD24(-/low)/CD44+ breast cancer-initiating cells to radiation. J Natl Cancer Inst. 2006;98:1777–1785. doi: 10.1093/jnci/djj495. [DOI] [PubMed] [Google Scholar]
- 73.Samanta D, Gilkes DM, Chaturvedi P, Xiang L, Semenza GL. Hypoxia-inducible factors are required for chemotherapy resistance of breast cancer stem cells. Proc Natl Acad Sci U S A. 2014;111:E5429–E5438. doi: 10.1073/pnas.1421438111. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 74.Ratajczak MZ, Shin DM, Liu R, Marlicz W, Tarnowski M, Ratajczak J, Kucia M. Epiblast/germ line hypothesis of cancer development revisited: lesson from the presence of Oct-4+ cells in adult tissues. Stem Cell Rev Rep. 2010;6:307–316. doi: 10.1007/s12015-010-9143-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Albini A, Bruno A, Gallo C, Pajardi G, Noonan DM, Dallaglio K. Cancer stem cells and the tumor microenvironment: interplay in tumor heterogeneity. Connect Tissue Res. 2015;56:414–425. doi: 10.3109/03008207.2015.1066780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Al-Hajj M, Clarke MF. Self-renewal and solid tumor stem cells. Oncogene. 2004;23:7274–7282. doi: 10.1038/sj.onc.1207947. [DOI] [PubMed] [Google Scholar]
- 77.Mcdermott SP, Wicha MS. Targeting breast cancer stem cells. Mol Oncol. 2010;4:404–419. doi: 10.1016/j.molonc.2010.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Yu F, Yao H, Zhu P, Zhang X, Pan Q, Gong C, Huang Y, Hu X, Su F, Lieberman J, Song E. Let-7 regulates self renewal and tumorigenicity of breast cancer cells. Cell. 2007;131:1109–1123. doi: 10.1016/j.cell.2007.10.054. [DOI] [PubMed] [Google Scholar]
- 79.Creighton CJ, Li X, Landis M, Dixon JM, Neumeister VM, Sjolund A, Rimm DL, Wong H, Rodriguez A, Herschkowitz JI, Fan C, Zhang X, He X, Pavlick A, Gutierrez MC, Renshaw L, Larionov AA, Faratian D, Hilsenbeck SG, Perou CM, Lewis MT, Rosen JM, Chang JC. Residual breast cancers after conventional therapy display mesenchymal as well as tumor-initiating features. Proc Natl Acad Sci U S A. 2009;106:13820–13825. doi: 10.1073/pnas.0905718106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Wang T, Narayanaswamy R, Ren H, Torchilin VP. Combination therapy targeting both cancer stem-like cells and bulk tumor cells for improved efficacy of breast cancer treatment. Cancer Biol Ther. 2016;17:698–707. doi: 10.1080/15384047.2016.1190488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Junger WG. Immune cell regulation by autocrine purinergic signalling. Nat Rev Immunol. 2011;11:201–212. doi: 10.1038/nri2938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Haskó G, Linden J, Cronstein B, Pacher P. Adenosine receptors: therapeutic aspects for inflammatory and immune diseases. Nat Rev Drug Discov. 2008;7:759–770. doi: 10.1038/nrd2638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Sperlágh B, Vizi ES. The role of extracellular adenosine in chemical neurotransmission in the hippocampus and basal ganglia: pharmacological and clinical aspects. Curr Top Med Chem. 2011;11:1034–1046. doi: 10.2174/156802611795347564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Antonioli L, Blandizzi C, Pacher P, Haskó G. Immunity, inflammation and cancer: a leading role for adenosine. Nat Rev Cancer. 2013;13:842–857. doi: 10.1038/nrc3613. [DOI] [PubMed] [Google Scholar]
- 85.Buxton IL, Yokdang N, Matz RM. Purinergic mechanisms in breast cancer support intravasation, extravasation and angiogenesis. Cancer Lett. 2010;291:131–141. doi: 10.1016/j.canlet.2009.09.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.De Leve S, Wirsdörfer F, Jendrossek V. Targeting the immunomodulatory CD73/adenosine system to improve the therapeutic gain of radiotherapy. Front Immunol. 2019;10:698. doi: 10.3389/fimmu.2019.00698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Di Virgilio F, Adinolfi E. Extracellular purines, purinergic receptors and tumor growth. Oncogene. 2017;36:293–303. doi: 10.1038/onc.2016.206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Jin H, Eun SY, Lee JS, Park SW, Lee JH, Chang KC, Kim HJ. P2Y2 receptor activation by nucleotides released from highly metastatic breast cancer cells increases tumor growth and invasion via crosstalk with endothelial cells. Breast Cancer Res. 2014;16:77. doi: 10.1186/bcr3694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Burnstock G. Purine and purinergic receptors. Brain Neurosci Adv. 2018;2:2398212818817494. doi: 10.1177/2398212818817494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Müller CE, Baqi Y, Namasivayam V. Agonists and antagonists for purinergic receptors. Methods Mol Biol. 2041;2020:45–64. doi: 10.1007/978-1-4939-9717-6_3. [DOI] [PubMed] [Google Scholar]
- 91.Burnstock G. Introduction to purinergic signalling in the brain. Adv Exp Med Biol. 2013;986:1–12. doi: 10.1007/978-94-007-4719-7_1. [DOI] [PubMed] [Google Scholar]
- 92.Giuliani AL, Sarti AC, Di Virgilio F. Extracellular nucleotides and nucleosides as signalling molecules. Immunol Lett. 2019;205:16–24. doi: 10.1016/j.imlet.2018.11.006. [DOI] [PubMed] [Google Scholar]
- 93.Borea PA, Gessi S, Merighi S, Vincenzi F, Varani K. Pharmacology of adenosine receptors: the state of the art. Physiol Rev. 2018;98:1591–1625. doi: 10.1152/physrev.00049.2017. [DOI] [PubMed] [Google Scholar]
- 94.Gessi S, Merighi S, Sacchetto V, Simioni C, Borea PA. Adenosine receptors and cancer. Biochim Biophys Acta. 2011;1808:1400–1412. doi: 10.1016/j.bbamem.2010.09.020. [DOI] [PubMed] [Google Scholar]
- 95.Sek K, Mølck C, Stewart GD, Kats L, Darcy PK, Beavis PA. Targeting adenosine receptor signaling in cancer immunotherapy. Int J Mol Sci. 2018;19:3837. doi: 10.3390/ijms19123837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Soleimani A, Taghizadeh E, Shahsavari S, Amini Y, Rashidpour H, Azadian E, Jafari A, Parizadeh MR, Mashayekhi K, Soukhtanloo M, Jaafari MR. CD73; a key ectonucleotidase in the development of breast cancer: recent advances and perspectives. J Cell Physiol. 2019;234:14622–14632. doi: 10.1002/jcp.28187. [DOI] [PubMed] [Google Scholar]
- 97.Allard B, Longhi MS, Robson SC, Stagg J. The ectonucleotidases CD39 and CD73: novel checkpoint inhibitor targets. Immunol Rev. 2017;276:121–144. doi: 10.1111/imr.12528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Bahreyni A, Samani SS, Rahmani F, Behnam-Rassouli R, Khazaei M, Ryzhikov M, Parizadeh MR, Avan A, Hassanian SM. Role of adenosine signaling in the pathogenesis of breast cancer. Cell Physiol. 2018;233:1836–1843. doi: 10.1002/jcp.25944. [DOI] [PubMed] [Google Scholar]
- 99.Burnstock G, Di Virgilio F. Purinergic signalling and cancer. Purinergic Signal. 2013;9:491–540. doi: 10.1007/s11302-013-9372-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Ferrari D, Gambari R, Idzko M, Müller T, Albanesi C, Pastore S, la Manna G, Robson SC, Cronstein B. Purinergic signaling in scarring. FASEB J. 2016;30:3–12. doi: 10.1096/fj.15-274563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Fernandez-Gallardo M, González-Ramírez R, Sandoval A, Felix R, Monjaraz E. Adenosine stimulate proliferation and migration in triple negative breast cancer cells. PLoS One. 2016;11(12):e0167445. doi: 10.1371/journal.pone.0167445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Allard B, Turcotte M, Stagg J. Targeting CD73 and downstream adenosine receptor signaling in triple-negative breast cancer. Expert Opin Ther Targets. 2014;18:863–881. doi: 10.1517/14728222.2014.915315. [DOI] [PubMed] [Google Scholar]
- 103.Da Rocha LF, de Oliveira APL, Accetturi BG, et al. Anti-inflammatory effects of inosine in allergic lung inflammation in mice: evidence for the participation of adenosine A 2A and A 3 receptors. Purinergic Signal. 2013;9:325–336. doi: 10.1007/s11302-013-9351-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Dastjerdi MN, Rarani MZ, Valiani A, Mahmoudieh M. The effect of adenosine A1 receptor agonist and antagonist on p53 and caspase 3, 8, and 9 expression and apoptosis rate in MCF-7 breast cancer cell line. Res Pharm Sci. 2016;11:303–310. doi: 10.4103/1735-5362.189301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Bieber D, Lorenz K, Yadav R, Klotz KN. A2B adenosine receptors mediate an inhibition of ERK1/2 phosphorylation in the breast cancer cell line MDA-MB-231. Naunyn Schmiedeberg's Arch Pharmacol. 2008;377:1–98. [Google Scholar]
- 106.Panjehpour M, Castro M, Klotz KN. Human breast cancer cell line MDA-MB-231 expresses endogenous A2B adenosine receptors mediating a Ca2+ signal. Br J Pharmacol. 2005;145:211–218. doi: 10.1038/sj.bjp.0706180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Desmet CJ, Gallenne T, Prieur A, Reyal F, Visser NL, Wittner BS, Smit MA, Geiger TR, Laoukili J, Iskit S, Rodenko B, Zwart W, Evers B, Horlings H, Ajouaou A, Zevenhoven J, van Vliet M, Ramaswamy S, Wessels LFA, Peeper DS. Identification of a pharmacologically tractable Fra-1/ADORA2B axis promoting breast cancer metastasis. Proc Natl Acad Sci U S A. 2013;110:5139–5144. doi: 10.1073/pnas.1222085110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Mohamadi A, Aghaei M, Panjehpour M. Estrogen stimulates adenosine receptor expression subtypes in human breast cancer MCF-7 cell line. Res Pharm Sci. 2018;13:57–64. doi: 10.4103/1735-5362.220968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Varani K, Vincenzi F, Targa M, Paradiso B, Parrilli A, Fini M, Lanza G, Borea PA. The stimulation of A(3) adenosine receptors reduces bone-residing breast cancer in a rat preclinical model. Eur J Cancer. 2013;49:482–491. doi: 10.1016/j.ejca.2012.06.005. [DOI] [PubMed] [Google Scholar]
- 110.Chadet S, Jelassi B, Wannous R, Angoulvant D, Chevalier S, Besson P, Roger S. The activation of P2Y2 receptors increases MCF-7 breast cancer cells migration through the MEK-ERK1/2 signalling pathway. Carcinogenesis. 2014;35:1238–1247. doi: 10.1093/carcin/bgt493. [DOI] [PubMed] [Google Scholar]
- 111.Park M, Kim J, Phuong N, Park JG, Park JH, Kim YC, Baek MC, Lim SC, Kang KW. Involvement of the P2X7 receptor in the migration and metastasis of tamoxifen-resistant breast cancer: effects on small extracellular vesicles production. Sci Rep. 2019;9:11587. doi: 10.1038/s41598-019-47734-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Xia J, Yu X, Tang L, Li G, He T. P2X7 receptor stimulates breast cancer cell invasion and migration via the AKT pathway. Oncol Rep. 2015;34:103–110. doi: 10.3892/or.2015.3979. [DOI] [PubMed] [Google Scholar]
- 113.Morrison C, Mancini S, Cipollone J, Kappelhoff R, Roskelley C, Overall C. Microarray and proteomic analysis of breast cancer cell and osteoblast co-cultures: role of osteoblast matrix metalloproteinase (MMP)-13 in bone metastasis. J Biol Chem. 2011;286:34271–34285. doi: 10.1074/jbc.M111.222513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Pivetta E, Scapolan M, Pecolo M, Wassermann B, Abu-Rumeileh I, Balestreri L, Borsatti E, Tripodo C, Colombatti A, Spessotto P. MMP-13 stimulates osteoclast differentiation and activation in tumour breast bone metastases. Breast Cancer Res. 2011;13:105. doi: 10.1186/bcr3047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Avanzato D, Genova T, Pla AF, et al. Activation of P2X7 and P2Y11 purinergic receptors inhibits migration and normalizes tumor-derived endothelial cells via cAMP signaling. Sci Rep. 2016;6:32602. doi: 10.1038/srep32602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Qiu Y, Liu Y, Li WH, Zhang HQ, Tian XX, Fang WG. P2Y2 receptor promotes the migration and invasion of breast cancer cells via EMT-related genes Snail and E-cadherin. Oncol Rep. 2018;39:138–150. doi: 10.3892/or.2017.6081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Zhang JL, Liu Y, Yang H, Zhang HQ, Tian XX, Fang WG. ATP-P2Y2-β-catenin axis promotes cell invasion in breast cancer cells. Cancer Sci. 2017;108:1318–1327. doi: 10.1111/cas.13273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Li HJ, Wang LY, Qu HN, Yu L, Burnstock G, Ni X, Xu M, Ma B (2011) P2Y2 receptor-mediated modulation of estrogen-induced proliferation of breast cancer cells. Mol Cell Endocrinol 338:28–37. doi:10.1016/j.mce.2011.02.014 [DOI] [PubMed]
- 119.Ma X, Pan X, Wei Y, Tan B, Yang L, Ren H, Qian M, Du B. Chemotherapy-induced uridine diphosphate release promotes breast cancer metastasis through P2Y6 activation. Oncotarget. 2016;7:29036. doi: 10.18632/oncotarget.8664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Gareau AJ, Brien C, Gebremeskel S, Liwski RS, Johnston B, Bezuhly M. Ticagrelor inhibits platelet–tumor cell interactions and metastasis in human and murine breast cancer. Clin Exp Metastasis. 2018;35:25–35. doi: 10.1007/s10585-018-9874-1. [DOI] [PubMed] [Google Scholar]
- 121.Lin Z, Yin P, Reierstad S, O'Halloran H, Coon VJS, Pearson EK, Mutlu GM, Bulun SE. Adenosine A1 receptor, a target and regulator of estrogen receptorα action, mediates the proliferative effects of estradiol in breast cancer. Oncogene. 2010;29:1114–1122. doi: 10.1038/onc.2009.409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Mittal D, Sinha D, Barkauskas D, Young A, Kalimutho M, Stannard K, Caramia F, Haibe-Kains B, Stagg J, Khanna KK, Loi S, Smyth MJ. Adenosine 2B receptor expression on cancer cells promotes metastasis. Cancer Res. 2016;76:4372–4382. doi: 10.1158/0008-5472.CAN-16-0544. [DOI] [PubMed] [Google Scholar]
- 123.Huang Y, Gu Z, Fan Y, Guangxi Z, Xiaogang Z, Qifeng S, Yanbin S, Guimei L. Inhibition of the adenosinergic pathway: the indispensable part of oncological therapy in the future. Purinergic Signal. 2019;15:53–67. doi: 10.1007/s11302-018-9641-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Gessi S, Cattabriga E, Avitabile A, Gafa' R, Lanza G, Cavazzini L, Bianchi N, Gambari R, Feo C, Liboni A, Gullini S, Leung E, Mac-Lennan S, Borea PA. Elevated expression of A3 adenosine receptors in human colorectal cancer is reflected in peripheral blood cells. Clin Cancer Res. 2004;10:5895–5901. doi: 10.1158/1078-0432.CCR-1134-03. [DOI] [PubMed] [Google Scholar]
- 125.Madi L, Ochaion A, Rath-Wolfson L, Bar-Yehuda S, Erlanger A, Ohana G, Harish A, Merimski O, Barer F, Fishman P. The A3 adenosine receptor is highly expressed in tumor versus normal cells: potential target for tumor growth inhibition. Clin Cancer Res. 2004;10:4472–4479. doi: 10.1158/1078-0432.CCR-03-0651. [DOI] [PubMed] [Google Scholar]
- 126.Abedi H, Aghaei M, Panjehpour M, Hajiahmadi S. Mitochondrial and caspase pathways are involved in the induction of apoptosis by IB-MECA in ovarian cancer cell lines. Tumour Biol. 2014;35:11027–11039. doi: 10.1007/s13277-014-2396-9. [DOI] [PubMed] [Google Scholar]
- 127.Jafari SM, Panjehpour M, Aghaei M, Joshaghani HR, Enderami SE. A3 adenosine receptor agonist inhibited survival of breast cancer stem cells via GLI-1 and ERK1/2 pathway. J Cell Biochem. 2017;118:2909–2920. doi: 10.1002/jcb.25945. [DOI] [PubMed] [Google Scholar]
- 128.Meloche S, Pouysségur J. The ERK1/2 mitogen-activated protein kinase pathway as a master regulator of the G1-to S-phase transition. Oncogene. 2007;26:3227–3239. doi: 10.1038/sj.onc.1210414. [DOI] [PubMed] [Google Scholar]
- 129.Wolff F, Loipetzberger A, Gruber W, Esterbauer H, Aberger F, Frischauf AM. Imiquimod directly inhibits Hedgehog signalling by stimulating adenosine receptor/protein kinase A-mediated GLI phosphorylation. Oncogene. 2013;32:5574–5581. doi: 10.1038/onc.2013.343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Cochrane CR, Szczepny A, Watkins DN, Cain JE. Hedgehog signaling in the maintenance of cancer stem cells. Cancers (Basel) 2015;7:1554–1585. doi: 10.3390/cancers7030851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Hanna A, Shevde LA. Hedgehog signaling: modulation of cancer properies and tumor mircroenvironment. Mol Cancer. 2016;15:24. doi: 10.1186/s12943-016-0509-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Ferrari D, Malavasi F, Antonioli L. A purinergic trail for metastases. Trends Pharmacol Sci. 2017;38:277–290. doi: 10.1016/j.tips.2016.11.010. [DOI] [PubMed] [Google Scholar]
- 133.Adinolfi E, Raffaghello L, Giuliani AL, Cavazzini L, Capece M, Chiozzi P, Bianchi G, Kroemer G, Pistoia V, di Virgilio F. Expression of P2X7 receptor increases in vivo tumor growth. Cancer Res. 2012;72:2957–2969. doi: 10.1158/0008-5472.CAN-11-1947. [DOI] [PubMed] [Google Scholar]
- 134.Savio L, de Andrade MP, da Silva CG, Coutinho-Silva R. The P2X7 receptor in inflammatory diseases: angel or demon? Front Pharmacol. 2018;9:52. doi: 10.3389/fphar.2018.00052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Tan C, Han LI, Zou L, Luo C, Liu A, Sheng X, Xi D. Expression of P2X7R in breast cancer tissue and the induction of apoptosis by the gene-specific shRNA in MCF-7 cells. Exp Ther Med. 2015;10:1472–1478. doi: 10.3892/etm.2015.2705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Ferrari D, Pizzirani C, Adinolfi E, Lemoli RM, Curti A, Idzko M, Panther E, di Virgilio F. The P2X7 receptor: a key player in IL-1 processing and release. J Immunol. 2006;176:3877–3883. doi: 10.4049/jimmunol.176.7.3877. [DOI] [PubMed] [Google Scholar]
- 137.Gilbert SM, Oliphant CJ, Hassan S, Peille AL, Bronsert P, Falzoni S, di Virgilio F, McNulty S, Lara R. ATP in the tumour microenvironment drives expression of nfP2X 7, a key mediator of cancer cell survival. Oncogene. 2019;38:194–208. doi: 10.1038/s41388-018-0426-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Qu Y, Misaghi S, Newton K, Gilmour LL, Louie S, Cupp JE, Dubyak GR, Hackos D, Dixit VM. Pannexin-1 is required for ATP release during apoptosis but not for inflammasome activation. J Immunol. 2011;186:6553–6561. doi: 10.4049/jimmunol.1100478. [DOI] [PubMed] [Google Scholar]
- 139.Tokumitsu H, Chijiwa T, Hagiwara M, Mizutani A, Terasawa M, Hidaka H. KN-62, 1-[N,O-bis(5-isoquinolinesulfonyl)-N-methyl-L-tyrosyl]-4-phenylpiperazi ne, a specific inhibitor of Ca2+/calmodulin-dependent protein kinase II. J Biol Chem. 1990;265:4315–4320. doi: 10.1016/S0021-9258(19)39565-1. [DOI] [PubMed] [Google Scholar]
- 140.Minami H, Inoue S, Hidaka H. The effect of KN-62, Ca2+/calmodulin dependent protein kinase II inhibitor on cell cycle. Biochem Biophys Res Commun. 1994;199:241–248. doi: 10.1006/bbrc.1994.1220. [DOI] [PubMed] [Google Scholar]
- 141.Lauring J, Park BH, Wolff AC. The phosphoinositide-3-kinase-Akt-mTOR pathway as a therapeutic target in breast cancer. J Natl Compr Cancer Netw. 2013;11:670–678. doi: 10.6004/jnccn.2013.0086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Arnaud-Sampaio VF, Rabelo ILA, Ulrich H, Lameu C. The P2X7 receptor in the maintenance of cancer stem cells, chemoresistance and metastasis. Stem Cell Rev Rep. 2020;16:288–300. doi: 10.1007/s12015-019-09936-w. [DOI] [PubMed] [Google Scholar]
- 143.Rycaj K, Tang DG. Cell-of-origin of cancer versus cancer stem cells: assays and interpretations. Cancer Res. 2015;75:4003–4011. doi: 10.1158/0008-5472.CAN-15-0798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Craig BT, Rellinger EJ, Alvarez AL, Dusek HL, Qiao J, Chung DH. Induced differentiation inhibits sphere formation in neuroblastoma. Biochem Biophys Res Commun. 2016;477:255–259. doi: 10.1016/j.bbrc.2016.06.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Glaser T, de Oliveira SL, Cheffer A, et al. Modulation of mouse embryonic stem cell proliferation and neural differentiation by the P2X7 receptor. PLoS One. 2014;9(5):e96281. doi: 10.1371/journal.pone.0096281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Jacobson KA, Paoletta S, Katritch V, Wu B, Gao ZG, Zhao Q, Stevens RC, Kiselev E. Nucleotides acting at P2Y receptors: connecting structure and function. Mol Pharmacol. 2015;88:220–230. doi: 10.1124/mol.114.095711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Bilbao PS, Boland R, de Boland AR, Santillán G. ATP modulation of mitogen activated protein kinases and intracellular Ca2+ in breast cancer (MCF-7) cells. Arch Biochem Biophys. 2007;466:15–23. doi: 10.1016/j.abb.2007.07.012. [DOI] [PubMed] [Google Scholar]
- 148.Lee JM, Dedhar S, Kalluri R, Thompson EW. The epithelial–mesenchymal transition: new insights in signaling, development, and disease. J Cell Biol. 2006;172:973–981. doi: 10.1083/jcb.200601018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.De Sousa e Melo F. Vermeulen L. Wnt signaling in cancer stem cell biology. Cancers. 2016;8(7):60. doi: 10.3390/cancers8070060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Eun SY, Ko YS, Park SW, Chang KC, Kim HJ. P2Y2 nucleotide receptor-mediated extracellular signal-regulated kinases and protein kinase C activation induces the invasion of highly metastatic breast cancer cells. Oncol Rep. 2015;34:195–202. doi: 10.3892/or.2015.3972. [DOI] [PubMed] [Google Scholar]
- 151.Sarrió D, Rodriguez-Pinilla SM, Hardisson D, Cano A, Moreno-Bueno G, Palacios J. Epithelial-mesenchymal transition in breast cancer relates to the basal-like phenotype. Cancer Res. 2008;68:989–997. doi: 10.1158/0008-5472.CAN-07-2017. [DOI] [PubMed] [Google Scholar]
- 152.Azimi I, Beilby H, Davis FM, Marcial DL, Kenny PA, Thompson EW, Roberts-Thomson SJ, Monteith GR. Altered purinergic receptor-Ca2+ signaling associated with hypoxia-induced epithelial-mesenchymal transition in breast cancer cells. Mol Oncol. 2016;10:166–178. doi: 10.1016/j.molonc.2015.09.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Bodin P, Burnstock G. Purinergic signalling: ATP release. Neurochem Res. 2001;26:959–969. doi: 10.1023/a:1012388618693. [DOI] [PubMed] [Google Scholar]
- 154.Tang Z, Ye W, Chen H, Kuang X, Guo J, Xiang M, Peng C, Chen X, Liu H. Role of purines in regulation of metabolic reprogramming. Purinergic signall. 2019;15:423–438. doi: 10.1007/s11302-019-09676-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Ghalamfarsa G, Rastegari A, Atyabi F, Hassannia H, Hojjat-Farsangi M, Ghanbari A, Anvari E, Mohammadi J, Azizi G, Masjedi A, Yousefi M, Yousefi B, Hadjati J, Jadidi-Niaragh F. Anti-angiogenic effects of CD73-specific siRNA-loaded nanoparticles in breast cancer-bearing mice. J Cell Physiol. 2018;233:7165–7177. doi: 10.1002/jcp.26743. [DOI] [PubMed] [Google Scholar]
- 156.Antonioli L, Pacher P, Vizi ES, Haskó G. CD39 and CD73 in immunity and inflammation. Trends Mol Med. 2013;19:355–367. doi: 10.1016/j.molmed.2013.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Stockwell J, Jakova E, Cayabyab FS. Adenosine A1 and A2A receptors in the brain: current research and their role in neurodegeneration. Molecules. 2017;22:676. doi: 10.3390/molecules22040676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Sitkovsky MV. Damage control by hypoxia-inhibited AK. Blood. 2008;111:5424–5425. doi: 10.1182/blood-2008-03-143990. [DOI] [PubMed] [Google Scholar]
- 159.Wang J, Matosevic S. Adenosinergic signaling as a target for natural killer cell immunotherapy. J Mol Med (Berl) 2018;96:903–913. doi: 10.1007/s00109-018-1679-9. [DOI] [PubMed] [Google Scholar]
- 160.Zhang B. CD73 promotes tumor growth and metastasis. Oncoimmunology. 2012;1:67–70. doi: 10.4161/onci.1.1.18068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Deaglio S, Robson SC. Ectonucleotidases as regulators of purinergic signaling in thrombosis, inflammation, and immunity. Adv Pharmacol. 2011;61:301–332. doi: 10.1016/B978-0-12-385526-8.00010-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Tsukamoto H, Chernogorova P, Ayata K, Gerlach UV, Rughani A, Ritchey JW, Ganesan J, Follo M, Zeiser R, Thompson LF, Idzko M. Deficiency of CD73/ecto-5′-nucleotidase in mice enhances acute graft-versus-host disease. Blood. 2012;119:4554–4564. doi: 10.1182/blood-2011-09-375899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Stagg J, Divisekera U, Duret H, Sparwasser T, Teng MWL, Darcy PK, Smyth MJ. CD73-deficient mice have increased antitumor immunity and are resistant to experimental metastasis. Cancer Res. 2011;71:2892–2900. doi: 10.1158/0008-5472.CAN-10-4246. [DOI] [PubMed] [Google Scholar]
- 164.Heine P, Braun N, Sévigny J, Robson SC, Servos J, Zimmermann H. The C-terminal cysteine-rich region dictates specific catalytic properties in chimeras of the ectonucleotidases NTPDase1 and NTPDase2. Eur J Biochem. 2001;268:364–373. doi: 10.1046/j.1432-1033.2001.01896.x. [DOI] [PubMed] [Google Scholar]
- 165.Kapojos JJ, Van Den Berg A, Borghuis T, Banas B, Huitema S, Poelstra K, Bakker WW. Enhanced ecto-apyrase activity of stimulated endothelial or mesangial cells is downregulated by glucocorticoids in vitro. Eur J Pharmacol. 2004;501:191–198. doi: 10.1016/j.ejphar.2004.08.008. [DOI] [PubMed] [Google Scholar]
- 166.Eltzschig HK, Köhler D, Eckle T, Kong T, Robson SC, Colgan SP. Central role of Sp1-regulated CD39 in hypoxia/ischemia protection. Blood. 2009;113:224–232. doi: 10.1182/blood-2008-06-165746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Chalmin F, Mignot G, Bruchard M, Chevriaux A, Végran F, Hichami A, Ladoire S, Derangère V, Vincent J, Masson D, Robson SC, Eberl G, Pallandre JR, Borg C, Ryffel B, Apetoh L, Rébé C, Ghiringhelli F. Stat3 and Gfi-1 transcription factors control Th17 cell immunosuppressive activity via the regulation of ectonucleotidase expression. Immunity. 2012;36:362–373. doi: 10.1016/j.immuni.2011.12.019. [DOI] [PubMed] [Google Scholar]
- 168.Bastid J, Cottalorda-Regairaz A, Alberici G, Bonnefoy N, Eliaou JF, Bensussan A. ENTPD1/CD39 is a promising therapeutic target in oncology. Oncogene. 2013;32:1743–1751. doi: 10.1038/onc.2012.269. [DOI] [PubMed] [Google Scholar]
- 169.Goepfert C, Sundberg C, Sévigny J, Enjyoji K, Hoshi T, Csizmadia E, Robson S. Disordered cellular migration and angiogenesis in cd39-null mice. Circulation. 2001;104:3109–3115. doi: 10.1161/hc5001.100663. [DOI] [PubMed] [Google Scholar]
- 170.Allard D, Turcotte M, Stagg J. Targeting A2 adenosine receptors in cancer. Immunol Cell Biol. 2017;95:333–339. doi: 10.1038/icb.2017.8. [DOI] [PubMed] [Google Scholar]
- 171.Antonioli L, Novitskiy SV, Sachsenmeier KF, Fornai M, Blandizzi C, Haskó G. Switching off CD73: a way to boost the activity of conventional and targeted antineoplastic therapies. Drug Discov Today. 2017;22:1686–1696. doi: 10.1016/j.drudis.2017.06.005. [DOI] [PubMed] [Google Scholar]
- 172.Wang L, Zhou X, Zhou T, Ma D, Chen S, Zhi X, Yin L, Shao Z, Ou Z, Zhou P. Ecto-5′-nucleotidase promotes invasion, migration and adhesion of human breast cancer cells. J Cancer Res Clin Oncol. 2008;134:365–372. doi: 10.1007/s00432-007-0292-z. [DOI] [PubMed] [Google Scholar]
- 173.Buisseret L, Pommey S, Allard B, Garaud S, Bergeron M, Cousineau I, Ameye L, Bareche Y, Paesmans M, Crown JPA, di Leo A, Loi S, Piccart-Gebhart M, Willard-Gallo K, Sotiriou C, Stagg J. Clinical significance of CD73 in triple-negative breast cancer: multiplex analysis of a phase III clinical trial. Ann Oncol. 2018;29:1056–1062. doi: 10.1093/annonc/mdx730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Beavis PA, Stagg J, Darcy PK, Smyth MJ. CD73: a potent suppressor of antitumor immune responses. Trends Immunol. 2012;33:231–237. doi: 10.1016/j.it.2012.02.009. [DOI] [PubMed] [Google Scholar]
- 175.Sitkovsky MV, Hatfield S, Abbott R, Belikoff B, Lukashev D, Ohta A. Hostile, hypoxia-A2-adenosinergic tumor biology as the next barrier to overcome for tumor immunologists. Cancer Immunol Res. 2014;2:598–605. doi: 10.1158/2326-6066.CIR-14-0075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Terp MG, Olesen KA, Arnspang EC, Lund RR, Lagerholm BC, Ditzel HJ, Leth-Larsen R. Anti-human CD73 monoclonal antibody inhibits metastasis formation in human breast cancer by inducing clustering and internalization of CD73 expressed on the surface of cancer cells. J Immunol. 2013;191:4165–4173. doi: 10.4049/jimmunol.1301274. [DOI] [PubMed] [Google Scholar]
- 177.Zhi X, Wang Y, Zhou X, Yu J, Jian R, Tang S, Yin L, Zhou P. RNAi-mediated CD73 suppression induces apoptosis and cell-cycle arrest in human breast cancer cells. Cancer Sci. 2010;101:2561–2569. doi: 10.1111/j.1349-7006.2010.01733.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.García-Teijido P, Cabal ML, Fernández IP, Pérez YF. Tumor-infiltrating lymphocytes in triple negative breast cancer: the future of immune targeting. Clin Med Insights Oncol. 2016;10:31–39. doi: 10.4137/CMO.S34540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Lawrence RT, Perez EM, Hernández D, Miller CP, Haas KM, Irie HY, Lee SI, Blau CA, Villén J. The proteomic landscape of triple-negative breast cancer. Cell Rep. 2015;11:630–644. doi: 10.1016/j.celrep.2015.03.050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Foulkes WD, Smith IE, Reis-Filho JS. Triple-negative breast cancer. N Engl J Med. 2010;363:1938–1948. doi: 10.1056/NEJMra1001389. [DOI] [PubMed] [Google Scholar]
- 181.Gao ZW, Dong K, Zhang HZ. The roles of CD73 in cancer. Biomed Res Int. 2014;2014:460654–460659. doi: 10.1155/2014/460654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Loi S, Pommey S, Haibe-Kains B, Beavis PA, Darcy PK, Smyth MJ, Stagg J. CD73 promotes anthracycline resistance and poor prognosis in triple negative breast cancer. Proc Natl Acad Sci U S A. 2013;110:11091–11096. doi: 10.1073/pnas.1222251110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Leth-Larsen R, Lund R, Hansen HV, Laenkholm AV, Tarin D, Jensen ON, Ditzel HJ. Metastasis-related plasma membrane proteins of human breast cancer cells identified by comparative quantitative mass spectrometry. Mol Cell Proteomics. 2009;8:1436–1449. doi: 10.1074/mcp.M800061-MCP200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.O'Toole SA, Beith JM, Millar EK, West R, McLean A, Cazet A, Swarbrick A, Oakes SR. Therapeutic targets in triple negative breast cancer. J Clin Pathol. 2013;66:530–542. doi: 10.1136/jclinpath-2012-201361. [DOI] [PubMed] [Google Scholar]
- 185.Ribatti D. The concept of immune surveillance against tumors. The first theories. Oncotarget. 2017;8:7175–7180. doi: 10.18632/oncotarget.12739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Chen Y, Tan W, Wang C. Tumor-associated macrophage-derived cytokines enhance cancer stem-like characteristics through epithelial–mesenchymal transition. Onco Targets Ther. 2018;11:3817–3826. doi: 10.2147/OTT.S168317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Korkaya H, Liu S, Wicha MS. Breast cancer stem cells, cytokine networks, and the tumor microenvironment. J Clin Invest. 2011;121:3804–3809. doi: 10.1172/JCI57099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Gilkes DM, Semenza GL, Wirtz D. Hypoxia and the extracellular matrix: drivers of tumour metastasis. Nat Rev Cancer. 2014;14:430–439. doi: 10.1038/nrc3726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Davis FM, Peters AA, Grice DM, Cabot PJ, Parat MO, Roberts-Thomson SJ, Monteith GR. Non-stimulated, agonist-stimulated and store-operated Ca2+ influx in MDA-MB-468 breast cancer cells and the effect of EGF-induced EMT on calcium entry. PLoS One. 2012;7(5):e36923. doi: 10.1371/journal.pone.0036923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Zitvogel L, Apetoh L, Ghiringhelli F, André F, Tesniere A, Kroemer G. The anticancer immune response: indispensable for therapeutic success? J Clin Invest. 2008;118:1991–2001. doi: 10.1172/JCI35180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Disis ML, Stanton SE. Triple-negative breast cancer: immune modulation as the new treatment paradigm. Am Soc Clin Oncol Educ Book. 2015;35:25–30. doi: 10.14694/EdBook_AM.2015.35.e25. [DOI] [PubMed] [Google Scholar]
- 192.Schreiber RD, Old LJ, Smyth MJ. Cancer immunoediting: integrating immunity’s roles in cancer suppression and promotion. Science. 2011;331:1565–1570. doi: 10.1126/science.1203486. [DOI] [PubMed] [Google Scholar]
- 193.Vesely MD, Kershaw MH, Schreiber RD, Smyth MJ. Natural innate and adaptive immunity to cancer. Ann Rev Immunol. 2011;29:235–271. doi: 10.1146/annurev-immunol-031210-101324. [DOI] [PubMed] [Google Scholar]
- 194.Jiang X, Shapiro DJ. The immune system and inflammation in breast cancer. Mol Cell Endocrinol. 2014;382:673–682. doi: 10.1016/j.mce.2013.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Cimino-Mathews A, Foote JB, Emens LA. Immune targeting in breast cancer. Oncology (Williston Park) 2015;29:375–385. [PubMed] [Google Scholar]
- 196.Emens LA. Breast cancer immunobiology driving immunotherapy: vaccines and immune checkpoint blockade. Expert Rev Anticancer Ther. 2012;12:1597–1611. doi: 10.1586/era.12.147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Coussens LM, Pollard JW. Leukocytes in mammary development and cancer. Cold Spring Harb Perspect Biol. 2011;3(3):003285. doi: 10.1101/cshperspect.a003285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Mamessier E, Sylvain A, Thibult ML, Houvenaeghel G, Jacquemier J, Castellano R, Gonçalves A, André P, Romagné F, Thibault G, Viens P, Birnbaum D, Bertucci F, Moretta A, Olive D. Human breast cancer cells enhance self tolerance by promoting evasion from NK cell antitumor immunity. J Clin Invest. 2011;121:3609–3622. doi: 10.1172/JCI45816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Roberti MP, Rocca YS, Amat M, Pampena MB, Loza J, Coló F, Fabiano V, Loza CM, Arriaga JM, Bianchini M, Barrio MM, Bravo AI, Domenichini E, Chacón R, Mordoh J, Levy EM. IL-2- or IL-15-activated NK cells enhance cetuximab-mediated activity against triple-negative breast cancer in xenografts and in breast cancer patients. Breast Cancer Res Treat. 2012;136:659–671. doi: 10.1007/s10549-012-2287-y. [DOI] [PubMed] [Google Scholar]
- 200.Olkhanud PB, Baatar D, Bodogai M, Hakim F, Gress R, Anderson RL, Deng J, Xu M, Briest S, Biragyn A. Breast cancer lung metastasis requires expression of chemokine receptor CCR4 and regulatory T cells. Cancer Res. 2009;69:5996–6004. doi: 10.1158/0008-5472.CAN-08-4619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Sakaguchi S, Miyara M, Costantino CM, Hafler DA. FOXP3+ regulatory T cells in the human immune system. Nat Rev Immunol. 2010;10:490–500. doi: 10.1038/nri2785. [DOI] [PubMed] [Google Scholar]
- 202.Leone RD, Emens LA. Targeting adenosine for cancer immunotherapy. J Immunother Cancer. 2018;6:57. doi: 10.1186/s40425-018-0360-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Vijayan D, Young A, Teng M, Smyth MJ. Targeting immunosuppressive adenosine in cancer. Nat Rev Cancer. 2017;17:709–724. doi: 10.1038/nrc.2017.86. [DOI] [PubMed] [Google Scholar]
- 204.Antonioli L, Blandizzi C, Malavasi F, Ferrari D, Haskó G. Anti-CD73 immunotherapy: a viable way to reprogram the tumor microenvironment. Oncoimmunology. 2016;5(9):e1216292. doi: 10.1080/2162402X.2016.1216292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Polyak K, Weinberg R. Transitions between epithelial and mesenchymal states: acquisition of malignant and stem cell traits. Nat Rev Cancer. 2009;9:265–273. doi: 10.1038/nrc2620. [DOI] [PubMed] [Google Scholar]
- 206.Ruan K, Song G, Ouyang G. Role of hypoxia in the hallmarks of human cancer. J Cell Biochem. 2009;107:1053–1062. doi: 10.1002/jcb.22214. [DOI] [PubMed] [Google Scholar]
- 207.Davis FM, Kenny PA, Soo ET, et al. Remodeling of purinergic receptor-mediated Ca2+ signaling as a consequence of EGF-induced epithelial-mesenchymal transition in breast cancer cells. PLoS One. 2011;6(8):e23464. doi: 10.1371/journal.pone.0023464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Ledderose C, Hefti MM, Chen Y, Bao Y, Seier T, Li L, Woehrle T, Zhang J, Junger WG. Adenosine arrests breast cancer cell motility by A3 receptor stimulation. Purinergic signal. 1Lee JM, Dedhar S, Kalluri R, Thompson EW (2006) The epithelial–mesenchymal transition: new insights in signaling, development, and disease. J Cell Biol. 2016;172:973–981. doi: 10.1083/jcb.200601018. [DOI] [Google Scholar]
- 209.Draganov D, Gopalakrishna-Pillai S, Chen YR, Zuckerman N, Moeller S, Wang C, Ann D, Lee PP. Modulation of P2X4/P2X7/Pannexin-1 sensitivity to extracellular ATP via Ivermectin induces a non-apoptotic and inflammatory form of cancer cell death. Sci Rep. 2015;5:16222. doi: 10.1038/srep16222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Cekic C, Sag D, Li Y, Theodorescu D, Strieter RM, Linden J. Adenosine A2B receptor blockade slows growth of bladder and breast tumors. J Immunol. 2012;188:198–205. doi: 10.4049/jimmunol.1101845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Thompson LF, Ruedi JM, Glass A, Low MG, Lucas AH. Antibodies to 5′-nucleotidase (CD73), a glycosyl-phosphatidylinositol-anchored protein, cause human peripheral blood T cells to proliferate. J Immunol. 1989;143:1815–1821. [PubMed] [Google Scholar]
- 212.Airas L, Salmi M, Jalkanen S. Lymphocyte-vascular adhesion protein-2 is a novel 70-kDa molecule involved in lymphocyte adhesion to vascular endothelium. J Immunol. 1993;151(8):4228–4238. [PubMed] [Google Scholar]
- 213.Qiao Z, Li X, Kang N, Yang Y, Chen C, Wu T, Zhao M, Liu Y, Ji X. A novel specific anti-CD73 antibody inhibits triple-negative breast cancer cell motility by regulating autophagy. Int J Mol Sci. 2019;20:1057. doi: 10.3390/ijms20051057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Hashemi M, Karami-Tehrani F, Ghavami S, Maddika S, Los M (2005) Adenosine and deoxyadenosine induces apoptosis in oestrogen receptor-positive and -negative human breast cancer cells via the intrinsic pathway. Cell Prolif 38:269–285. 10.1111/j.1365-2184.2005.00349.x. 10.1111/j.1365-2184.2005.00349.x [DOI] [PMC free article] [PubMed]
- 215.Mirza A, Basso A, Black S, Malkowski M, Kwee L, Patcher JA, Lachowicz JE, Wang Y, Liu S (2005) RNA interference targeting of A1 receptor-overexpressing breast carcinoma cells leads to diminished rates of cell proliferation and induction of apoptosis. Cancer Biol Ther 4:1355–1360. 10.4161/cbt.4.12.219610.4161/cbt.4.12.2196 [DOI] [PubMed]
- 216.Irshad S, Ellis P, Tutt A. Molecular heterogeneity of triple-negative breast cancer and its clinical implications. Curr Opin Oncol. 2011;23:566–577. doi: 10.1097/CCO.0b013e32834bf8ae. [DOI] [PubMed] [Google Scholar]
- 217.Ntantie E, Gonyo P, Lorimer EL, Hauser AD, Schuld N, McAllister D, Kalyanaraman B, Dwinell MB, Auchampach JA, Williams CL. An adenosine-mediated signaling pathway suppresses prenylation of the GTPase Rap1B and promotes cell scattering. Sci Signal. 2013;6(277):39. doi: 10.1126/scisignal.2003374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Stagg J, Divisekera U, McLaughlin N, Sharkey J, Pommey S, Denoyer D, Dwyer KM, Smyth MJ. Anti-CD73 antibody therapy inhibits breast tumor growth and metastasis. Proc Natl Acad Sci U S A. 2010;107:1547–1552. doi: 10.1073/pnas.0908801107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Allard B, Turcotte M, Spring K, Pommey S, Royal I, Stagg J. Anti-CD73 therapy impairs tumor angiogenesis. Int J Cancer. 2014;134:1466–1473. doi: 10.1002/ijc.28456. [DOI] [PubMed] [Google Scholar]
- 220.Mittal D, Young A, Stannard K, Yong M, Teng MWL, Allard B, Stagg J, Smyth MJ. Antimetastatic effects of blocking PD-1 and the adenosine A2A receptor. Cancer Res. 2014;74:3652–3658. doi: 10.1158/0008-5472.CAN-14-0957. [DOI] [PubMed] [Google Scholar]
- 221.Chen S, Wainwright DA, Wu JD, Wan Y, Matei DE, Zhang Y, Zhang B. CD73: an emerging checkpoint for cancer immunotherapy. Immunotherapy. 2019;11:983–997. doi: 10.2217/imt-2018-0200. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.