Abstract
Background and aims
The new coronavirus disease (COVID-19) is a systemic disease. Mounting evidence depict signs and symptoms involving multiple organs, most of which supported by pathological data. A plausible link to these manifestations is vascular and endothelial dysfunction/damage. However, much of the current knowledge relies on opinion and incipient evidence. We aim to objectively appraise current evidence on the association between COVID-19 and vascular disease, specifically endotheliitis and vasculitis.
Methods
Two researchers independently entered the search terms COVID-19 OR SARS-CoV-2 AND vasculitis, endotheliitis OR endothelium in the following online platforms: MedRxiv and LitCovid (PubMed). The search period was set from November 1, 2019 to August 28, 2020. Manuscripts with unavailable full texts, not in English, mainly on pre-clinical data, presenting only study designs or not directly related to the topics of this review were excluded. Retrospective and prospective studies, especially longitudinal ones, were given priority to the purpose of this review. Since there was paucity of prospective controlled evidence, case reports/series were also considered.
Results
A total of 318 manuscripts were initially found. Sixty-seven (21%) were excluded: 59 (18.5%) met exclusion criteria and 8 (2.5%) were duplicates. One hundred and forty-two manuscripts (44,6%) did not provide original data and were also excluded: 35 (11%) were comments, 108 (33.9%) reviews; 1 (0.3%) position paper. One hundred and seven (33.6%) studies were considered for the present scoping review: 81 (25,5%) case reports/series; 18 (5.7%) prospective; 8 (2.5%) retrospective. Viral inclusions in endothelial cells, mononuclear cell infiltrates in the intima of small vessels and markers of endothelial cell apoptosis were demonstrated. Specificities of COVID-19 may lead to diverse vascular manifestations in different levels of the vascular bed.
Conclusions
Evidence indicates that COVID-19 targets vasculature and endothelium. However, high quality data is still lacking and studies with prospective designs and appropriately matched controls are needed.
Keywords: Coronavirus, Endotheliitis, Inflammation, Coagulation, Vascular reactivity
Graphical abstract
1. Introduction
The new coronavirus disease-19 (COVID-19) is a major global issue and despite ubiquitous actions being employed to mitigate its effects, the disease still spreads relentlessly, and recrudescence waves of new cases seem to take place. At this moment, there is clear indication that the disease is systemic by design [1,2] and involves not only the respiratory epithelium but also the cardiovascular endothelium [2]. Endothelial cells, like pneumocytes type II, abundantly express transmembrane angiotensin converting enzyme 2 (ACE-2) and host type 2 transmembrane serine protease (TMPRSS2). These are two fundamental locks through which the virus enters the cells. The disease, in fact, is primarily respiratory but may quickly gain the vascular network and hit gastrointestinal tract, kidneys, heart and nervous system, with associated platelet-endothelial dysfunction and abnormally rapid life-threatening blood clotting and inflammation.
In the lungs, endothelial cells of capillaries comprise up to 50% of the cellular mass of alveoli [3] and approximately one third of the alveolus' septal wall thickness is composed of a layer of endothelium [4]. Thus, it is not unexpected that these vascular cells could also be a target to the virus at least in the lungs. In parallel, the severe acute respiratory syndrome virus-2 (SARS-CoV-2) may also spread and affect endothelial cells in other sites, as it has been demonstrated. Evidence of pathology studies reveal viral inclusions within endothelial cells, inflammatory infiltrates in the intima layer of large and small arteries, and apoptosis - revealed by caspase-3 immunohistochemistry staining - or pyroptosis [5] of these cells in several organs and tissues’ vessels, including lung, heart, kidney, and intestines [2,6,7].
Although pathological findings have been conflicting in demonstrating viral particles within endothelial cells [1], many mechanisms can concur for a vasculitis to happen. First, endothelial cell damage and apoptosis could lead to basal membrane exposure and a thrombotic macro- and/or microangiopathy [8]. Second, endothelial cell dysfunction due to unbalanced angiotensin II upregulation could drive local or diffuse vasoconstrictive responses whether in lungs, limbs, intestines or virtually any affected organ [9,10]. Third, endothelial cells have been frequently recognized as part of the innate immune system and could be responsible for an exacerbated response in the inflammatory phase of the disease [11]. Lastly, perivascular inflammatory infiltrates can be implicated in a constrictive microangiopathy leading to chilblain lesions in extremities, a common symptom seen in COVID-19 mild presenting cases that could be frequently undiagnosed [12].
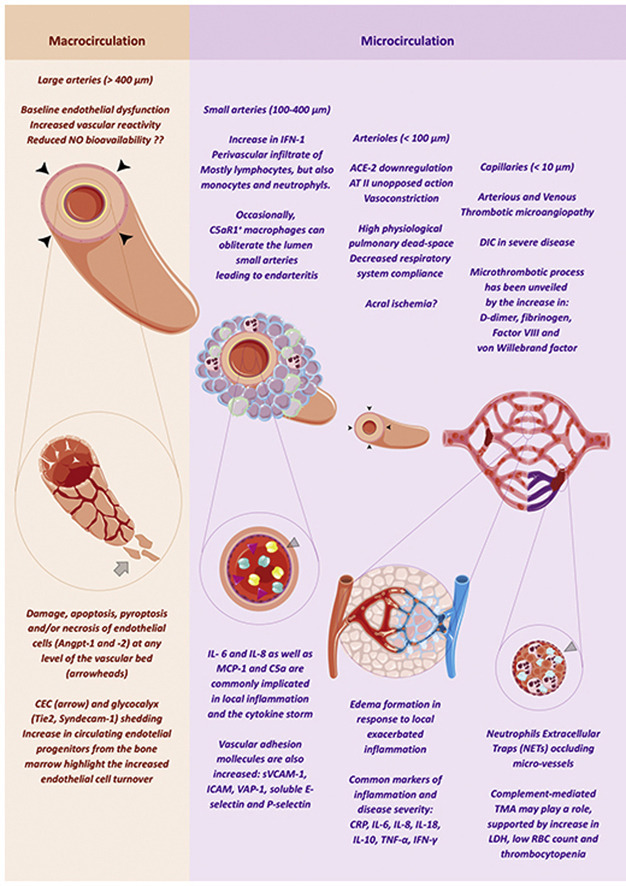
In severe cases, cellular hypoxia may ultimately result from failure at different levels of the vascular bed—macrocirculation and microcirculation—and additionally from mitochondrial dysfunction [13]. At least macro- and microcirculation functions can be affected by vascular damage and/or endothelial dysfunction and may play a role in COVID-19. While macrocirculation failure emerges from impaired vascular tone control, microcirculation failure arises from shunting or maldistributed flow from disrupted perfusion of small arteries and capillaries. In this review, we systematically gather and appraise published data with the focus on vascular and endothelial function/damage and its relationship with clinical manifestations and disease severity.
2. Materials and methods
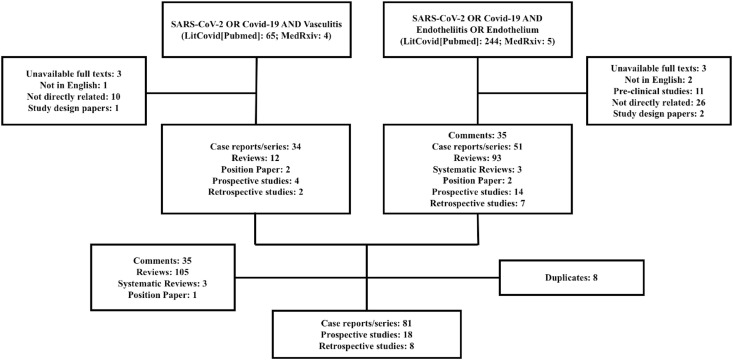
Two researchers (TQ, IB) entered the search terms COVID-19 OR SARS-CoV-2 AND vasculitis, endotheliitis OR endothelium in the following online platforms: MedRxiv and LitCovid (PubMed). The search period was set from November 1, 2019 to August 28, 2020. Manuscripts with unavailable full texts, that were not in English, had mainly pre-clinical data, presented only study designs or were not directly related to the topics of this review were excluded (Fig. 1 ). Retrospective and prospective studies (Table 1 ), especially longitudinal ones, were given priority to the purpose of this review (Fig. 2 ). Since there was paucity of prospective controlled evidence, case reports/series were also considered. The terms COVID-19 OR SARS-CoV-2 AND endothelium OR vasculitis were also entered in the ClinicalTrials.gov platform (endotheliitis did not retrieve any studies in this specific platform), filtered for the same time period and same exclusion criteria, to retrieve ongoing studies investigating these aspects of the disease (Supplementary Table). Additional relevant manuscripts were accessed by a manual search through the references of the initially retrieved studies in order to put data in context. All additional papers were included when there was agreement between researchers.
Fig. 1.
Flow diagram showing main appraised studies after application of inclusion and exclusion criteria.
Table 1.
Prospective and retrospective studies in human subjects indexed in the Pubmed, LitCovid and MedRxiv platforms found with the search terms COVID-19 or SARS-CoV-2 and endotheliitis, endothelium or vasculitis applied alternately.
| Authors | Title | Study type | Sample (total) | Control group | Findings |
|---|---|---|---|---|---|
| Diehl et al. [21] | Respiratory mechanics and gas exchanges in the early course of COVID-19 ARDS: a hypothesis-generating study | Prospective (observational) | 22 | No control group | CEC and D-dimer associated with increase of pulmonary physiologic dead-space as calculated by the mechanical respirator in moderate‐to‐severe COVID-19 ARDS patients |
| Karakas et al. [73] | Targeting Endothelial Dysfunction in Eight Extreme-Critically Ill Patients with COVID-19 Using the Anti-Adrenomedullin Antibody Adrecizumab (HAM8101) | Prospective (non-randomized) | 8 | No control group | The PaO2/FiO2 increased and C-reactive protein, procalcitonin, and interleukin-6 decreased with the use of a first-in-class humanized monoclonal anti-Adrenomedullin antibody targeting the sepsis- and inflammation-based vascular and capillary leakage in critically ill patients |
| Viecca et al. [83] | Enhanced platelet inhibition treatment improves hypoxemia in patients with severe COVID-19 and hypercoagulability. A case control, proof of concept study | Prospective (non-randomized) | 10 | COVID-19 patients matched by age, D-dimer and SOFA score (n = 5) | Antiplatelet therapy might be effective in improving the ventilation/perfusion ratio in COVID-19 patients with severe respiratory failure |
| Kang et al. [77] | IL-6 trans-signalling induces plasminogen activator inhibitor-1 from vascular endothelial cells in cytokine release syndrome | Prospective (non-randomized) | 7 | Baseline biomarkers compared with healthy controls (n = 36); patients with sepsis (n = 37), ARDS (n = 19), or burns (n = 35) | Baseline serum levels of IL-6, MCP-1, and IL-10 in the severe COVID-19 patients were higher than in healthy controls but lower than in CRS patients (sepsis, ARDS, and burns). Only PAI-1 levels were similar to those of CRS patients. An IL-6 receptor antagonist use associated with a decrease in PAI-1, IL-10 and C-reactive protein (CRP) levels. Levels of MCP-1 were unaffected and IL-6 increased |
| Stahl et al. [106] | Injury to the Endothelial Glycocalyx in Critically Ill COVID-19 Patients | Prospective (observational) | 39 | Healthy controls (n = 10) | Endothelial markers Angpt-1 and -2 were similar between COVID-19 patients and controls. In contrast, an increase in the soluble form of the Tie2 receptor and in Syndecan-1 indicates shedding of transmembrane proteins of the glycocalyx structure and processing. The endothelial glycocalyx protective molecule Hpa-2 was reduced in COVID-19 patients |
| Perrin et al. [50] | Cytokine release syndrome-associated encephalopathy in patients with COVID-19 | Prospective (observational) | 5 | No control group | Neurologic manifestations occurred simultaneously with the peak in CRS serum markers (CRP, IL-6 and LDH). Serum levels of the astroglial marker, S100B protein, were increased at the time of CRS, reflecting an increased permeability of the blood-brain barrier and returned to their reference range when neurological symptoms and signs of hyperinflammation regressed |
| Ladikou et al. [84] | Von Willebrand factor (vWF): marker of endothelial damage and thrombotic risk in COVID-19? | Prospective (observational) | 24 | No control group | The INR, APTT ratio and platelet count were all within the normal range. Fibrinogen and D-dimer were elevated. Factor VIII and von Willebrand factor antigen were proportionally higher in deceased patients. |
| Carvelli et al. [93] | Association of COVID-19 inflammation with activation of the C5a–C5aR1 axis | Prospective (observational) | 82 | Healthy controls (n = 10) | Increase in plasma complement factor C5a is proportional to COVID-19 severity. High expression of its receptor C5aR1 in blood and pulmonary myeloid cells, as well as, obliterating endarteritis associated with accumulation of C5aR1+ macrophages around the arteries and within thrombus were observed. Findings support a role for the C5a-C5aR1 axis |
| Dupont et al. [28] | Excess soluble fms-like tyrosine kinase 1 correlates with endothelial dysfunction and organ failure in critically ill COVID-19 patients | Prospective (observational) | 56 | No control group | High circulating sFlt-1, a variant of the VEGF-A receptor 1 that impairs endothelial cell homeostasis, is associated with severe COVID-19 and correlates with the endothelial dysfunction biomarker sVCAM-1 and with adverse outcomes during the follow-up of these patients |
| Smadja et al. [29] | Angiopoietin‐2 as a marker of endothelial activation is a good predictor factor for intensive care unit admission of COVID‐19 patients | Prospective (observational) | 40 | Healthy controls (n = 13) | Soluble E-selectin and Angpt-2 were significantly increased in severe COVID-19 patients. Angpt-2 correlated with CRP, creatinine and D-dimers levels and it was a predictor of ICU admission |
| Khider et al. [104] | Curative anticoagulation prevents endothelial lesion in COVID-19 patients | Prospective (observational) | 96 | COVID-19 negative patients (n = 30) | Adding D-dimers to gender and pneumonia at CT scan increased power for COVID-19 diagnosis. CEC at admission were more common in COVID-19 patients but were lower in those previously receiving curative anticoagulation |
| Facchetti et al. [7] | SARS-CoV2 vertical transmission with adverse effects on the newborn revealed through integrated immunohistochemical, electron microscopy and molecular analyses of placenta. | Prospective (observational) | 101 | COVID-19 negative post-partum women (n = 86) | SARS-CoV-2 antigens, RNA and/or particles morphologically consistent with coronavirus were identified in villous syncytiotrophoblast, endothelial cells, fibroblasts, in maternal macrophages, and fetal intravascular mononuclear cells |
| Goshua et al. [85] | Endotheliopathy in COVID-19-associated coagulopathy: evidence from a single-centre, cross-sectional study | Retrospective (cross-sectional) | 81 | Healthy controls (n = 13) | Increased VWF antigen, Factor VII activity, soluble P-selectin, D-dimer and thrombin-antithrombin complex were elevated in ICU patients compared with non-ICU patients and controls. PAI-1 levels were not different between groups. Thrombomodulin increase was associated with reduced rates of hospital discharge |
| Mancuso et al. [23] | Circulating endothelial progenitors are increased in COVID-19 patients and correlate with SARS-CoV-2 RNA in severe cases. | Prospective (randomized) | 34 | Recovered COVID-19 patients (n = 9) Healthy controls (n = 8) |
COVID-19 patients had less apoptotic CEC. However, their precursors, viable circulating endothelial progenitors, were increased in COVID-19 patients compared with controls, although similar between mild and severe cases |
| Leppkes et al. [98] | Vascular occlusion by neutrophil extracellular traps in COVID-19 | Retrospective (observational) | 71 | Healthy controls (n = variable depending on the analysis) | In severe COVID-19, neutrophil granulocytes are strongly activated and adopt a low-density phenotype, prone to spontaneously form NETs. Histopathology of the lungs and other organs showed congestions of numerous micro-vessels by aggregated NETs associated with endothelial damage |
| Li et al. [5] | Clinical and pathological investigation of patients with severe COVID-19 | Retrospective (longitudinal) | 69 | No control group | IL-8, IL-6, TNF-α, IP10, MCP-1, and RANTES, were elevated in patients with severe COVID-19. IL-6 and IL-8 were associated with disease progression. SARS-CoV-2 was demonstrated to infect type II and type I pneumocytes and endothelial cells, leading to severe lung damage through cell pyroptosis and apoptosis |
| Tong et al. [27] | Elevated Serum Endothelial Cell Adhesion Molecules Expression in COVID-19 Patients | Retrospective (longitudinal) | 71 | Uninfected controls (n = 32) | Severity of COVID-19 was associated with serum levels of CRP, IL-18, TNF-α, IFN-γ, FKN, VCAM-1, ICAM-1, and VAP-1. Recovery was associated with reductions in serum CRP, IL-18, TNF-α, FKN, VCAM-1, ICAM-1, and D-dimer levels |
| Hernández-Fernández et al. [10] | Cerebrovascular disease in patients with COVID-19: neuroimaging, histological and clinical description | Retrospective (longitudinal) | 23 | COVID-19 negative patients (n = 66) | An incidence of 1.4% of cerebrovascular disease was seen in patients with COVID-19, mostly ischemic (17 cases). Of these, major etiology was of undetermined origin (9 cases, as in TOAST classification). Age was the only independent predictor of poor prognosis. D-dimer levels were high in most patients |
| John et al. [113] | Characteristics of Large-Vessel Occlusion Associated with COVID-19 and Ischemic Stroke | Retrospective (longitudinal) | 20 | No control group | An incidence of 2.97% of acute ischemic stroke was seen among patients with COVID-19. Large-vessel occlusion occurred in 15 out of 20. Etiology was undetermined in 46% of patients and systemic thrombosis in other sites was not uncommon (26%) |
| Dofferhoff et al. [43] | Reduced vitamin K status as a potentially modifiable risk factor of severe COVID-19 | Retrospective (longitudinal) | 135 | Historical healthy controls (n = 184) | Dp-ucMGP, an inactivated form of MPG, was elevated in COVID-19 patients compared to controls and associated with disease severity. Vitamin K activates MPG, which protects against pulmonary and vascular elastic fiber damage |
| Guervilly et al. [22] | Circulating Endothelial Cells as a Marker of Endothelial Injury in Severe COVID -19 | Retrospective (cross-sectional) | 99 | No control group | CEC count was above the upper normal limit in 55% of COVID-19 patients and it was significantly higher in ICU patients than in non-ICU. CEC count correlated with sVCAM-1 and IP-10 concentrations but not with E-selectin and IL-6 concentrations |
| Herman et al. [82] | Evaluation of Chilblains as a Manifestation of the COVID-19 Pandemic | Prospective (cross-sectional) | 31 | No control group | Histopathologic analysis of skin biopsy (22 patients) confirmed the diagnosis of chilblains and showed occasional lymphocytic or microthrombotic phenomena. Immunofluorescence analyses showed vasculitis of small-diameter vessels in 7 patients. None of the patients tested positive for COVID-19 on nasopharyngeal swabs, nor on blood immunoglobulin (Ig)M or IgG antibodies |
| Ouldali et al. [39] | Emergence of Kawasaki disease related to SARS-CoV-2 infection in an epicentre of the French COVID-19 epidemic: a time-series analysis | Retrospective (longitudinal) | 230 | No control group | An increase of 497% in Kawasaki disease related to SARS-CoV-2 positive testing occurred 2 weeks after the peak of the COVID-19 in April 2020. Another increase of 365% of hospital admissions due to Kawasaki disease occurred in December 2009, concomitant with the influenza A H1N1 pandemic |
| Consiglio et al. [41] | The Immunology of Multisystem Inflammatory Syndrome in Children with COVID-19 | Prospective (cross-sectional) | 73 | Historical control of Kawasaki disease children (n = 28); healthy controls (n = 12) | Differences in T-cell subsets and cytokine mediators place MIS-C between Kawasaki disease and acute COVID-19 immune states. Results suggest a more diffuse endothelial involvement and immunopathology in MIS-C than in Kawasaki disease |
| Chang et al. [40] | Serum responses of children with Kawasaki Disease against SARS-CoV-2 proteins | Retrospective (cross-sectional) | 87 | Historical febrile (n = 36) and Kawasaki disease (n = 39) controls | There is not a non-specific global cross-reactivity from previous coronaviruses contributing to these pediatric COVID-19 related inflammatory disorder cases |
| Shanshal et al. [59] | The Impact of COVID-19 on Medical Practice: A Nationwide Survey of Dermatologists and Healthcare Providers | Prospective (cross-sectional) | 300 | No control group | COVID-19-related cutaneous manifestations were recognized by 20% of dermatologists, including maculo-papular rash (41.67%), urticaria (37.50%), chilblain (25%) and vasculitis (16.67%) |
For the prospective interventional studies, a label of randomized or non-randomized was added to the cell.
Angpt: angiopoietin; APTT: activated partial thromboplastin time; ARDS: acute respiratory distress syndrome; CEC: circulating endothelial cells; CRP: C-reactive protein; CRS: cytokine release syndrome; dp-ucMGP inactive vitamin K-dependent matrix Gla protein, FiO2: fraction of inspired oxygen; FKN: fractalkine; Hpa-2: heparanase-2; ICAM-1: intercellular adhesion molecule-1; ICU: intensive care unit; IFN- γ: interferon- γ; IL-6: interleukin-6; IL-10: interleukin-10; IL-18: interleukin-18; IP-10: interferon-induced protein; INR: international normalized ratio; LDH: lactase dehydrogenase; MCP-1: monocyte chemotactic protein-1; MIS-C: multisystem inflammatory syndrome in children; NETs: neutrophil extracellular traps; PAI-1: plasminogen activator inhibitor-1; PaO2: partial pressure of oxygen; RANTES: regulated on activation, normal T-cell expressed and secreted; sFlt-1: soluble fms-like tyrosine kinase 1; TNF- α: tumor necrosis factor – α; VAP-1: vascular adhesion protein-1; VCAM-1: vascular cell adhesion molecule-1; sVCAM-1: soluble vascular cell adhesion molecule-1.
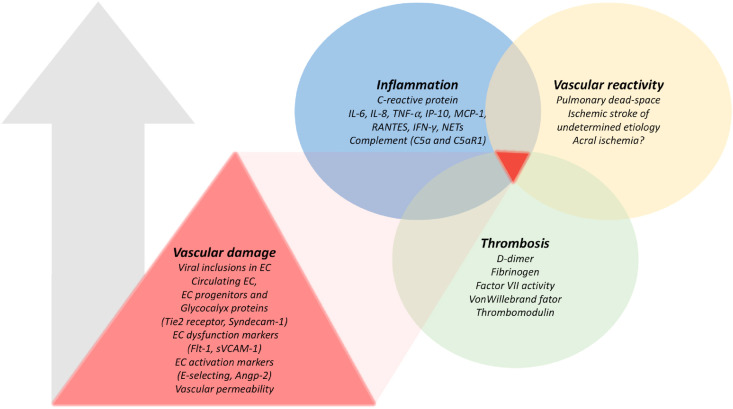
Fig. 2.
Biomarkers and clinical features of vascular damage and associated inflammation, thrombosis and vascular reactivity responses in COVID-19 according to retrospective and prospective studies in human subjects published to date.
Evidence of increase in vascular damage (particularly the endothelium) seems to associate with exacerbated inflammation, thrombosis and vascular reactivity. The arrow indicates a likely positive correlation between these features. These biomarkers and clinical characteristics were extracted from the studies with the best available evidence after the application of inclusion and exclusion criteria of the present review. The studies from which the biomarkers and clinical characteristics were extracted are presented in Table 1 and obtained as outlined in Fig. 1.
3. Results
A total of 318 manuscripts were initially found. Sixty-seven were excluded: 59 (18.5%) met exclusion criteria and 8 (2.5%) were duplicates. One hundred and forty-two manuscripts (44,6%) did not provide original data and were also excluded: 35 (11%) were comments, 108 (33.9%) reviews; 1 (0.3%) position paper. One hundred and seven (33.6%) studies were considered for the present scoping review: 81 (25,5%) case reports/series; 18 (5.7%) prospective; 8 (2.5%) retrospective. None of the studies found in the ClinicalTrials.gov platform had results available. Evidence of viral inclusions in endothelial cells, mononuclear cell infiltrates in the intima of small vessels and markers of endothelial cell apoptosis was demonstrated. Specificities of the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) seem to lead to diverse vascular manifestations and involvement of different levels of the vascular bed mainly driven by pro-inflammatory, pro-thrombotic effects and possibly increased vascular reactivity. We outline below the main findings of the included articles discussing the vascular and endothelial crosstalk with the common features of COVID-19 pathophysiology.
4. Discussion
4.1. Pathophysiology and clinical manifestations of endotheliitis
The SARS-CoV-2 enters the cells through a transmembrane angiotensin-converting enzyme 2 (ACE-2) [14]. Through such mechanism, ACE-2 activity is potentially downregulated. Type II pneumocytes abundantly express the receptor, but also does the endothelium [15]. A host type 2 transmembrane serine protease (TMPRSS2) facilitates entry by cleaving ACE-2 and is also necessary for the process to occur [14]. The affinity of the spike protein of the virus for the transmembrane ACE-2 enzyme is 10- to 20-fold higher than that of the SARS coronavirus-1 (SARS-CoV1), the causal agent of SARS, which is one possible explanation for the virus high infectivity [16]. Thus, SARS-CoV-2 would target the endothelium, and impairment and loss of the vascular barrier integrity could promote arterial reactivity, and a pro-coagulative and pro-inflammatory state, clinically expressed, in the critical disease, as disseminated intravascular coagulation (DIC) and systemic inflammatory response syndrome (SIRS) [[17], [18], [19]].
The advent of diffuse endotheliitis could lead to a cytokine storm frequently described in the severe disease [20]. An evidence of endothelial injury has been an elevation of detectable circulating endothelial cells [21,22] and endothelial cells’ progenitors [23]. Systemically, the endothelium and surrounding pericytes (mural cells) act as a barrier preventing immune cell and platelet interactions with tissue factor and collagen of the basement membrane. Thus, endothelial cells operate as a gatekeeper of the innate immune system and have a somewhat similar function to that of macrophages, which would include antigen presentation, cytokine secretion and scavenger activity [11,24]. Reports of cardiac involvement, hypoxemia with apparently healthy lungs, and severe thrombotic complications [25] could be a consequence of diffuse endotheliitis expressed as localized or disseminated vasoconstriction, thrombotic macro- and microangiopathy and a pro-inflammatory systemic response. Clinically, venous and arterial thromboembolic events have been reported in 10–25% of admitted patients [26] (Fig. 2). Integrins seem to be increased, as unveiled by elevated levels of vascular cell adhesion molecule-1 (VCAM-1), intercellular adhesion molecule 1 (ICAM-1), and vascular adhesion protein-1 (VAP-1) [27]. However, an imbalance favouring the loss of vascular endothelial integrity is being indicated. Soluble fms-like tyrosine kinase 1 (sFlt-1) [28] —an inhibitor of the vascular endothelial growth factor pathway and demonstrated factor to promote endothelial dysfunction—is elevated in severe cases. This is also true for angiopoietin-2 [29], also a marker of endothelial damage/dysfunction [30], and could be related to activated angiogenesis associated with extensive vascular damage.
4.1.1. Lungs
In the lung, damaged endothelium could trigger and propagate acute respiratory distress syndrome (ARDS), recognized as the leading cause of mortality in COVID-19 patients [18]. Augmented concentration of angiotensin II, consequent to reduced levels of ACE-2, may lead to vasoconstriction and hypo-perfused lung areas (dead spaces) producing arterial hypoxemia exceeding abnormalities in pulmonary mechanics [31]. Another possible source of hypo-perfusion could derive from micro-thrombosis and congestion of small capillaries intermediated by inflammatory cells, as well as, thrombosis of medium to large vessels. After critical damage of pneumocytes, the pathogenesis of the hypoxemia changes. Alveolar flooding and impaired alveolar-arterial gas exchange give place to a ventilation-perfusion mismatch or intra-pulmonary shunt. Endothelial cells present interleukin-6 (IL-6) and interleukin-2 receptors and stimulation of these induce capillary leakage and vascular fluid loss to interstitium and alveoli. In turn, endothelial cells would intensify IL-6 secretion and perpetuate this response [32,33]. Severe damage and denudation of the pulmonary endothelium could also deflagrate complement system activation which would elicit migration of neutrophils and monocytes further amplifying the inflammatory response [8]. At this point the exacerbated inflammation generates vasodilatation and increased vascular permeability which contributes to the interstitial and progressive alveolar edema. Receptor ACE-2 downregulation can play a role also by releasing the kallikrein-bradykinin pathway which enhances vascular permeability [8]. Pneumonia has been reported to occur in 75% of hospitalized patients and ARDS in 15% [34].
4.1.2. Heart
Another target to SARS-CoV-2 appears to be the heart. Cardiomyocytes present ACE-2 and TMPRSS2 and are susceptible to viral entry. Interstitial infiltration by inflammatory cells and viral particles were identified within the myocardium but viral inclusions were not found in cardiomyocytes or cardiac endothelial cells [35]. Cardiac injury reported as troponin elevation arises in 7–17% in hospitalized patients. Acute heart failure, myocarditis and arrhythmias have been reported as well [36]. In the SARS disease, the SARS-CoV viral RNA was detected in 7 out of 20 (35%) of autopsied human heart samples from a Toronto outbreak. The authors demonstrated that myocardial damage was associated with macrophage infiltration. ACE-2 expression was markedly reduced in the tissue and left ventricular hypertrophy was the most prominent attribute of the cardiac phenotype [37]. SARS-CoV and SARS-CoV-2 share very alike structures (79% similarity in the nucleotide sequence) [38], thus, these findings suggest that the heart is highly susceptible to COVID-19 injury and attention should be paid to signs of cardiac injury. Another piece of evidence indicates that the incidence of Kawasaki disease, a systemic vasculitis of small- and medium-sized arteries that can affect coronary arteries, has been occurring in children and is apparently related to the current coronavirus pandemics [39,40]. Moreover, it has been suggested that the multisystem inflammatory syndrome seen in children can have even more diffuse endothelial involvement than Kawasaki disease [41]. However, this association may not be specific to COVID-19. A similar raise in Kawasaki disease cases has also occurred in a previous influenza pandemic [39]. From a mechanistic standpoint, SARS-CoV-2 could affect the heart through direct cell invasion, induction of atherosclerotic plaque rupture - for instance, via activated macrophage-induced collagen degradation of its fibrous cap-, coronary spasm, microthrombi obstruction of a coronary or cardiomyocyte dysfunction owing to inflammation, or even other still unknown pathways [42].
4.1.3. Kidneys
As for the kidneys, renal epithelial cells, the podocytes, express ACE-2 and TMPRSS2, but, thus far, little expression was demonstrated in human endothelial cells within the kidney [43]. In spite of that, viral particles have been described in endothelial cells of glomerular capillaries [2]. In a case series of deceased patients presenting multiple organ dysfunction, obstruction of capillaries and inclusion of viral particles within tubular epithelium and podocytes was shown without evident vasculitis or overt interstitial inflammation. Acute kidney failure may occur, as well, in consequence of exacerbated inflammation and compromised blood flow. This condition has been reported in around 9% of hospitalized patients. Enhanced coagulation and stagnant flow contribute to systemic hypoxia and can damage oxygen-sensitive organs, such as the kidneys [44]. Mechanistically, kidney injury may occur due to direct invasion of proximal tubular cells or podocytes. Indirect injury may result from reduced blood flow following a dysregulated ACE-2 pathway and thus, acute tubular necrosis, protein leakage in Bowman's capsule, collapsing glomerulopathy and mitochondrial impairment. Exacerbated inflammation and the cytokine storm may also lead to kidney malfunction [45].
4.1.4. Nervous system
High level-expression of ACE-2 has been documented too in brain endothelial cells, neurons and glial cells [46,47]. Particles of the virus were recently identified in brain capillary endothelial cells [46] and in brain tissue of 36% of a COVID-19 series [48]. Infection of the olfactory tract and the consequent anosmia were supported by increased MRI signal of the olfactory cortex [49]. SARS-CoV-2 may reach the central nervous system by retrograde transneuronal routes or hematologic route by crossing a compromised blood-brain barrier due to endothelial injury/endotheliitis [50], direct infection of endothelial cells or through macrophages carrying the virus. Interleukin (IL)-6 and tumor necrosis factor have been previously demonstrated to disrupt the blood-brain barrier [51]. These cytokines are also powerful activators of the hypothalamic-pituitary-adrenocortical axis, a pivotal regulator of the systemic immune activity [52]. Indirectly, the nervous system can also be affected via unbalanced ACE/ACE-2 pathways, which leads to excessive vasoconstriction, ischemia and a possible disturbance of cerebral blood flow regulation [10]. Cytokine storm may contribute to both blood-barrier disruption and neurotoxicity [50] although the mechanisms are not completely understood.
4.1.5. Gastrointestinal system
Intestinal glandular cells present transmembrane ACE-2 and could be a direct target to the virus [53]. The expression ACE-2 appears to be more frequent in ileum and colon mainly in the absorptive enterocytes which could explain the manifestation of diarrhea in some patients [54]. Viral RNA has been isolated in stools [55]. Diffuse endothelial inflammation and mesenteric ischemia in submucosal vessels of small bowel has been described and can be related to local microvascular injury [2].
4.1.6. Cutaneous system
Cutaneous manifestations have been frequently reported in highly probable cases, but with a low positivity of real-time reverse transcriptase polymerase chain reaction (RT-PCR). This may happen because potential mechanisms can relate to immune response to SARS-CoV-2 rather than direct virus infection [56]. For this reason, these cutaneous manifestations may not occur in the early phase of the disease, but patients commonly report previous respiratory symptoms [56]. Evidence to support this theory has come from the observation of superficial perivascular dermatitis evidenced as diffuse and dense lymphoid infiltrates accompanied by endothelial inflammation on biopsies of acrocutaneous lesions [57]. Eventually, small thrombi have been also shown in vessel of the dermis [58]. In a survey, 20% of dermatologists have reported having recognized cutaneous manifestations of COVID-19 and 16% of these lesions to be of a vasculitis nature [59]. Although still largely unknown, the most documented cutaneous lesion has been chilblains. One study has shown a likely association between endothelial infection and this manifestation. Variable degrees of lymphocytic vasculitis, from endothelial swelling to necrosis, and perivascular lymphocytic inflammation were reported [60].
4.2. Endothelium and inflammation
A delayed or halted adaptive immune response has been implicated in severe COVID-19 cases, particularly when a cytokine storm takes place. When uncompensated, unleashing the innate immunity could lead to deleterious exacerbated inflammation and ineffective clearance of the virus. Interferon type I production has been implicated in effective viral debacle in previous coronavirus diseases [61]. However, these viruses seem to present mechanisms to supress IFN-I action [62]. Interferon type I activates the adaptive immune system in mammals through T-cell stimulation. Accordingly, lymphopenia has been linked to worse prognosis particularly due to depletion of circulating CD4+ and CD8+ T-cell [63]. In pathology studies, CD4+ T-cells were more numerous in lungs from patients who died with COVID-19 than in patients with influenza, but the opposite was seen for CD8+ [64]. CD4+ T-cells interact with B-cells to create virus-specific antibody production and also with CD8+ T-cells. The latter, in their turn, recognize peptides in the virus surface and directly attack them through a cytotoxic response. Aside from immune evasion by the virus [62], an early bypass of innate immunity could be due to submicrometer sized virus-laden aerosols reaching deep into alveoli without being undertaken by the defence mechanisms of the upper respiratory tract and the first antigen presenting cells [65]. Another plausible explanation would be a swift replication of SARS-CoV-2, known to be at least 3 times faster than SARS-CoV-1 [66].
However, deleterious inflammation induced by the virus can only occur in conjunction with an inappropriate host response. Only around 20% of patients present severe to critical disease and clinical characteristics in these cases are consistently described as older age, male sex and presence of cardiovascular risk factors [67,68]. These main clinical features are all related to baseline endothelial dysfunction as suggested previously [69]. Which could explain, at least in part, the susceptibility of these subgroups of patients. The endothelium is currently recognized as one of the sentinels of the innate immune system [11]. These cells express IFN-α, a cytokine of paramount importance in regulating innate immune responses against viruses [70] and activating the adaptive response, as mentioned above. An impaired endothelial function could therefore impede an appropriate response in the acute phase of the disease.
Moreover, the endothelium could propagate the inflammatory response when not coordinated as expected. Dysregulated endothelial cells can excessively produce IL-6, a cytokine frequently cited as prognostic in COVID-19 [71] (Fig. 2). IL-6 is one of the major activators of the JAK/STAT signalling pathway, a cascade greatly activated in COVID-19 patients [72,73]. In atherosclerotic disease, chronic elevations of IL-6 have been implicated in the incidence of cardiovascular events [74]. Acute elevations of IL-6 during COVID-19 could somewhat explain the occurrence of the inflammation of the cardiovascular system and microangiopathy frequently seen in these patients [71]. The role of the endothelium in perpetuating a cytokine storm was demonstrated in an animal model of acute influenza virus disease. The S1P1 receptor in respiratory endothelial cells regulates a pivotal signalling loop for initiation of this exacerbated inflammatory response [75]. The causality link is further tested when an S1P1 agonist is shown to blunt cytokines and chemokines related to the pathogenesis of the influenza virus disease and decrease mortality of infected mice [76]. Other inflammatory biomarkers found in severe COVID-19 patients were MCP-1 and IL-10 but they were not different and even presented lower levels than those of other cytokine release syndromes (sepsis, ARDS and burns) [77].
Of note, reports of patients presenting painful violaceous infiltrated plaques usually on the tip of toes and/or fingers appear to be induced by a common inflammatory reaction as a result of increased production of IFN-I [12,78] (Online Graphical abstract). The lesions resemble chilblains and manifest after weeks of respiratory symptoms onset, so patients may have a negative SARS-CoV-2 RT-PCR, but positive serology [79]. In most cases patients were young and otherwise asymptomatic or just mildly symptomatic. Only few studies described histological findings possibly due to the benign-related course of the disease. Infiltrates of predominantly lymphocytic cells seem to constrain vessel walls like in common interferonopathies. These chilblain lesions of COVID-19 share clinical and biological presentation with systemic lupus erytematosus. COVID-19 as other viral diseases trigger IFN-I release in a normal host response. However, if the production of IFN-I is delayed or blunted an abnormal inflammatory response may develop and could give rise to a cytokine storm [80,81]. Thus, it has been proposed that the presentation of these lesions could be a sign of favorable immune response and good prognosis [12]. Despite the biological plausibility, not all studies have confirmed this association [82]. Moreover, it is important to differentiate these skin manifestations from acral ischemia, which is more related to vasoconstriction and thrombosis and unfold mostly in critically ill patients.
4.3. Endothelium and coagulation
The local and systemic pro-inflammatory state per se promotes an environment prone to coagulation. This is true for influenza and other respiratory coronavirus diseases, in which thrombotic events appear to be increased. However, some particularities pertain to SARS-CoV-2 especially in regard to the vasculature. Pathology studies demonstrate severe endothelial damage associated with intracellular inclusions of the virus and disrupted cell membranes [64]. In a comparison of lung specimens from influenza patients matched for age, sex and disease severity, capillary microthrombi (<10 μm) were 9-fold more common in COVID-19. Although, influenza patients also present thrombi in pulmonary arteries from 1 to 2 mm of diameter and downstream, the greater difference was indeed observed at the capillary level. Also, microvascular structure was significantly distorted due to capillary elongation and angiogenesis (2.7 times more prevalent than in the influenza counterparts). These findings can possibly be consequence of the more common endothelial inflammation and injury and of the capillary thrombosis [64]. The data endorses that the pathogenesis of hypoxemia differs in COVID-19 and influenza disease at least in an initial stage of the disease [83]. In COVID-19 pulmonary dead space—non-perfused but ventilated alveoli—would play more of a role [21], whereas in other viral disease pulmonary shunt—perfused but not ventilated alveoli—would have much of an importance. In accordance with this understanding, the same study shows that lungs from influenza patients were heavier in gross examination than COVID-19 ones indicating a greater amount of fluid accumulation.
Endothelial disruption and apoptosis of endothelial cells expose basement membrane, which in turn, activates thrombotic cascades. Interleukin-1β and Tumor Necrosis Factor stimulate endothelial cells to express P-selectin, von Willebrand factor, factor VIII and fibrinogen [84,85]—ultimately binding substrates to platelets. The endothelial cells release additional cytokines that further increases platelet production, and, in a closed loop, platelets liberate vascular endothelial growth factor (VEGF) upregulating tissue factor expression by the endothelium, a primer to the thrombotic cascade [86]. While under a coronavirus infection, endothelial cells also increase plasminogen activator inhibitor 1 (PAI-1) [77] inhibiting conversion of plasminogen to plasmin, which normally degrades fibrin clots, further contributing to a coagulation state. An antagonizing homeostatic effort generates high levels of fibrin breakdown products (D-dimers) as a result of blood clots dissolution [87]. The ischemic tissue may induce endothelial cells hyperplasia additionally contributing to ischemia or to angiogenesis that could alleviate ischemia, but could also facilitate the spread of inflammatory cells [8].
There has been controversy about the pathophysiology of the thrombotic manifestations in COVID-19 especially in severe cases. Opposition has been made between thrombotic microangiopathy (TMA) and sepsis-induced coagulopathy or DIC as the main pathophysiological process of the microvascular thrombosis. The distinction is very important because of therapeutic implications. TMA is a specific process characterized by microangiopathic hemolytic anemia thrombocytopenia and organ dysfunction and damage. TMA occurs mainly at arterioles and results of increased platelet adhesion to the vascular endothelium in association with platelet aggregation and activation, driving to a consumptive thrombocytopenia [88]. Common laboratorial features in COVID-19 supporting this presentation are increased lactate dehydrogenase (LDH), low red blood cell count (RBC) and thrombocytopenia [89]. However, information on important aspects are still missing, such as the presence of schistocytes in peripheral blood smear to confirm microangiopathic hemolysis. Complement system plays a pivotal role in TMA as part of the innate immune response [90]. A recent study demonstrated that N proteins of SARS-CoV-2 binds to a key serine protease in the lectin pathway of complement activation MASP-2 (Mannan-binding lectin serin protease 2) [91]. Another study corroborates with the activation of the lectin and the alternative pathway of the complement system. The authors found deposits of C5b-9 (the membrane attack complex-MAC), C4d and MASP-2 in the microcirculation of purpuric skin lesions and also in normally-appearing skin, as well as, in the interalveoli septa [92]. Soluble C5a also appears to be increased in a proportionate fashion to disease severity in parallel to high expression of its receptor C5aR1 in blood and pulmonary cells [93]. Together with the fact that renal and cardiac dysfunction are common features of severe to critical COVID-19, it appears that a complement-mediated TMA is a part of COVID-19 pathophysiology.
Deposition of MAC on cell membranes induce calcium influx and activate intracellular signals [94]. Through this mechanism, deposits of MAC can activate platelets and enhance aggregation [95], but also, it can promote von Willebrand factor secretion by endothelial cells [96] possibly associated with prothrombinase activity. Other mechanisms could be increased tissue factor activity on endothelial cells induced by C5a [96] and the cleavage of prothrombin to activated thrombin by MASP-2 [97]. Thus, various mechanisms through which coagulation can be bolstered by the complement system could occur in COVID-19. Another unique, but not specific, characteristic of COVID-19 may be the increased proportion of aggregated neutrophil extracellular traps (NET) in the microcirculation. Lung histopathology revealed diffuse congestion of micro-vessels associated with NETs and endothelial damage [98,99] (Fig. 2). The presence of NETs indicate that thrombosis is immuno-related and may affect many organs, including lung, kidney and heart and is associated with disease severity [99,100]. Also, that viable virus can be responsible for the direct induction of NETs release by healthy neutrophils indicating inhibition of its mechanism may be a potential therapeutic target [101].
On the other end, increased D-dimer levels, thrombocytopenia and prolonged coagulation tests could resemble a DIC pattern often seen in severe sepsis. However, a hallmark of DIC is abnormal thrombin generation and no evidence has been presented towards this direction [102]. Tissue factor is often expressed by mononuclear cells and endothelial cells, which in turn accrues from proinflammatory cytokines’ discharge, particularly IL-1β and IL-6 [102, 103]. DIC is a consumptive coagulopathy often followed by hemorrhagic phenomenon, which is not frequently reported in COVID-19. Although elevation of D-dimer is common (40–60% of cases) in COVID-19, this is possibly due to localized exacerbated coagulation and fibrinolysis within the lung [104]. On top of that, damaged endothelial cells change their properties to a procoagulant mode following disruption of the glycocalyx and its binding protein, antithrombin [105,106] (Fig. 2). Thrombin-antithrombin complex were elevated in ICU patients. Interestingly, thrombomodulin levels—a specific marker of endothelial injury—were associated with reduced hospital discharge. The normally preserved antithrombin, protein C and protein S activities support the idea of a different mechanism than that of DIC [85]. A recent report stated that 8.7% of a series of hospitalized COVID-19 cases met criteria for DIC as established by international guidelines, whereas the prevalence of DIC in sepsis reaches around 30% [17]. As the disease advances in severity other factors come into play and DIC may arise.
4.4. Endothelium, nitric oxide and vascular reactivity
Vascular smooth muscle cells express both ACE-2 receptors and TMPRSS2 proteases as do endothelial cells [107,108]. In normal conditions, ACE-2 cleaves angiotensin II into angiotensin 1-7 which in turn stimulates endothelial cells to produce nitric oxide (Online Graphical Abstract) [109,110]. Vascular tone is physiologically regulated, and anti-inflammatory/anti-thrombotic properties activated by nitric oxide [111]. SARS-CoV-2 occupies and down-regulates ACE-2 possibly unleashing angiotensin II effects, namely vasoconstriction of larger arteries and reduced blood flow, as well as pro-coagulation and inflammatory effects. Direct effect of the virus on smooth muscle cells may have different outcomes depending on the organ involved, but the vasculature may be indirectly and indistinctively affected by COVID-related angiotensin II increase. Also, possible reductions in nitric oxide bioavailability may contribute to these effects. Vasoconstriction and ischemia has been shown to occur in different vascular beds, including ischemia and cyanosis of extremities [9,112] (particularly limbs and fingers), acute cerebrovascular events [10,113] and myocardial infarction with non-obstructive coronary arteries [36] (Fig. 2).
A normal balance of nitric oxide and reactive oxygen species is fundamental for vascular function as are ACE/ACE-2 pathways. Nitric oxide regulates blood flow and certain host defense measures. Reactive oxygen species are important to fight against viral infections [114] but the process should be well regulated by nitric oxide to avoid deleterious consequences [115]. Hypoxia and reoxygenation as ischemia and reperfusion deflagrate inflammation through reactive oxygen species and heparinase-mediated degradation of the glycocalyx and endothelial layer of cells [116]. Progressive endothelial dysfunction can result from an excess release of reactive oxygen species by M1 macrophages in response to high viral loads [117]. M1 macrophages also release IL-2, IL-6, IL-8, interferon-α/β and tumor necrosis factor-α which may contain the SARS-CoV-2 but also strike the vessels. Nitric oxide can regulate macrophages by resetting them back to M2 status withholding the inflammation and initiating a repair process and clearance of debris [118]. Interestingly, S-nitrosylation of viral cysteine proteases and the host serine protease, TMPRSS2, both appear to be nitric oxide sensitive [119] also indicating possible implications of nitric oxide bioavailability.
4.5. Clinical assessment of endothelial function
Endothelial function may be assessed in a variety of ways and many techniques are being applied to assess possible effects and chronic consequences of COVID-19 in the vasculature (Supplementary Table). One of such methods is flow mediated dilation (FMD) evaluated by ultrasound. Its parameters associate with vasomotor endothelial function and may be a proxy to cardiovascular health. Macrocirculation can be assessed by the dilation response of a large artery (diameter > 400 μm), mainly driven by acute nitric oxide production, and microcirculation can be assessed by hyperemic flow increase, due to accumulation of metabolites in arterioles (diameter < 100 μm). Ultrasound-based FMD has been used to assess vasomotor endothelial function in patients with sepsis and septic shock due to primarily respiratory infections and correlates with adverse outcomes and survival [120]. In COVID-19, the correlation could be greater due to the intertwined relationship of the virus cycle and the endothelium/arterial wall. FMD has also been previously validated by magnetic resonance imaging with reduced variability of results [121]. To specifically evaluate microvascular rarefaction nailbed capillaroscopy is being used, a technique that allows direct visualization and count of peripheral capillaries (<10 μm).
4.6. Upcoming clinical trials
Many interventions targeting the vasculature and specifically the endothelium are being tested. Drugs acting on the thrombotic and inflammatory aspects of the endothelium are presented (Supplementary Table). The effects of these drugs can vary from endothelial glycocalyx function restoration with glycosaminoglycans to reduction of vascular permeability with anti VEGF recombinant humanized monoclonal antibody. Actions on the renin-angiotensin-aldosterone system are of particular interest because this seems to be the mainstay of COVID-19 pathophysiology. Angiotensin-converting enzyme (ACE) inhibitors, angiotensin (AT) II receptor blockers and statins are among the most cited. The first trial seem to show no difference in mortality in the first 30 days after discharge [122] for patients who had the treatment discontinued versus those who did not. The effect of ACE inhibitors or AT II receptor blockers is still unknown if initiated in naïve-treated patients admitted for COVID-19. In this specific scenario, these drugs could have an effect on rebalancing the renin-angiotensin pathway and regulating vascular reactivity as well as potentially reducing cell entry by the virus, although the effect in angiotensin II activity is controversial [123]. Recombinant ACE-2 may have a potential benefit, however, because it would directly increase angiotensin II cleavage [124]. Therapy with statins can improve peripheral NO-mediated arterial relaxation and offset the effects of SARS-CoV-2 [125]. This class of drugs may also reduce macrophage chemotaxis and downregulate MCP-1 activity [126]. Other therapies being investigated that could act upon vascular reactivity are inhaled nitric oxide, prostacyclin and phosphodiesterase-5 inhibitors, such as sildenafil.
4.7. Limitations
This study has limitations which are mostly associated with the nature of the manuscripts used for this review. COVID-19 is a newly discovered disease and although much progress was done towards building the knowledge on its different aspects, high quality evidence is still lacking in some specific aspects of the disease. The vast amount of data derives from case reports and case series. Only a few retrospective and prospective studies were published with the specific focus of understanding the association with COVID-19 and endotheliitis and/or vasculitis. Moreover, the majority of them did not have any or had control groups of healthy subjects which helps shed light on the pathophysiology of the disease but may introduce some bias in understanding some of its features. Having appropriate controls, for instance other viral pneumonias or viral diseases, would allow identifying specific biomarkers of COVID-19. At this moment, there is no certainty of the specific features of COVID-19. We also did not exclude case report or series from the present analysis due to the paucity of prospective controlled evidence. Another limitation of the present study is that a meta-analysis to assess the incidence of vascular events was hindered by the heterogeneity of control groups across studies and this greatly limits more precise conclusions about the topic.
4.8. Conclusion
Knowledge on COVID-19 pathophysiology expands in accelerated fashion. Escalating evidence indicate that the disease is systemic by design and a possible link between organs and systems is vascular damage and dysfunction, particularly of the endothelium. Vasculitis may assume different presentations at different levels of the vascular bed. A possible decline in nitric oxide bioavailability may target both macro- and microcirculation affecting blood flow, vascular tone regulation and exchange at capillary level which culminates with a worsened cellular hypoxia. Other factors may contribute to the pathogenesis such as a thrombotic microangiopathy often described in pathology studies and a hyper-inflammatory state that could be consequence to impaired endothelial cell immune properties. Of note, some manifestations may result from normal immune response as seen in constrained vessel walls by lymphocytic infiltrates. Thus far, the corollary of the inflammation related severity of the disease has been laid by the positive trials showing beneficial effect of anti-inflammatory drugs. However, other inflammatory manifestations of the disease, such as endarteritis of medium to small vessels and NETs still need to be addressed. Furthermore, the specific aspects of induced-thrombosis also need further understanding. Whether complement-related or sepsis-like the management of thrombosis can be quite different. Lastly, the proposed vascular reactivity is still mostly in the hypothesis field. Clinical trials targeting this specific aspect of the disease will clarify how important this is to its pathophysiology. Further larger studies with prospective designs are needed to better delineate the disease pathophysiology and potential targets for treatments.
Author contributions
Drs. Quinaglia, Silber, Lima and Sposito designed the study. Drs. Quinaglia, Shabani and Breder strategized and extracted the data. Drs. Quinaglia and Breder drafted the manuscript.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgements
Dr. Thiago Quinaglia reveived a grant from the Department of Cardiovascular Imaging of the Brazilian Society of Cardiology. All other authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.atherosclerosis.2021.02.009.
Appendix A. Supplementary data
The following are the Supplementary data to this article:
References
- 1.Bradley B.T., Maioli H., Johnston R., Chaudhry I., Fink S.L., Xu H., Najafian B., Deutsch G., Lacy J.M., Williams T., Yarid N., Marshall D.A. Histopathology and ultrastructural findings of fatal COVID-19 infections in Washington State: a case series. Lancet. 2020;396:320–332. doi: 10.1016/S0140-6736(20)31305-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Varga Z., Flammer A.J., Steiger P., Haberecker M., Andermatt R., Zinkernagel A.S., Mehra M.R., Schuepbach R.A., Ruschitzka F., Moch H. Endothelial cell infection and endotheliitis in COVID-19. Lancet. 2020 2-8 May;395:1417–1418. doi: 10.1016/S0140-6736(20)30937-5. 10234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Townsley M.I. Structure and composition of pulmonary arteries, capillaries, and veins. Comp. Physiol. 2012;2:675–709. doi: 10.1002/cphy.c100081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Crapo J.D., Barry B.E., Gehr P., Bachofen M., Weibel E.R. Cell number and cell characteristics of the normal human lung. Am. Rev. Respir. Dis. 1982;125:740–745. doi: 10.1164/arrd.1982.125.6.740. [DOI] [PubMed] [Google Scholar]
- 5.Li S., Jiang L., Li X., Lin F., Wang Y., Li B., Jiang T., An W., Liu S., Liu H., Xu P., Zhao L., Zhang L., Mu J., Wang H., Kang J., Li Y., Huang L., Zhu C., Zhao S., Lu J., Ji J., Zhao J. Clinical and pathological investigation of patients with severe COVID-19. JCI Insight. 2020;5 doi: 10.1172/jci.insight.138070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Inciardi R.M., Lupi L., Zaccone G., Italia L., Raffo M., Tomasoni D., Cani D.S., Cerini M., Farina D., Gavazzi E., Maroldi R., Adamo M., Ammirati E., Sinagra G., Lombardi C.M., Metra M. Cardiac involvement in a patient with coronavirus disease 2019 (COVID-19) JAMA Cardiol. 2020;5(7):819–824. doi: 10.1001/jamacardio.2020.1096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Facchetti F., Bugatti M., Drera E., Tripodo C., Sartori E., Cancila V., Papaccio M., Castellani R., Casola S., Boniotti M.B., Cavadini P., Lavazza A. SARS-CoV2 vertical transmission with adverse effects on the newborn revealed through integrated immunohistochemical, electron microscopy and molecular analyses of Placenta. EBioMedicine. 2020;59:102951. doi: 10.1016/j.ebiom.2020.102951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Pober J.S., Sessa W.C. Evolving functions of endothelial cells in inflammation. Nat. Rev. Immunol. 2007;7:803–815. doi: 10.1038/nri2171. [DOI] [PubMed] [Google Scholar]
- 9.Kaur P., Posimreddy S., Singh B., Qaqa F., Habib H.A., Maroules M., Shamoon F. COVID-19 presenting as acute limb Ischaemia. Eur J Case Rep Intern Med. 2020;7 doi: 10.12890/2020_001724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hernandez-Fernandez F., Valencia H.S., Barbella-Aponte R.A., Collado-Jimenez R., Ayo-Martin O., Barrena C., Molina-Nuevo J.D., Garcia-Garcia J., Lozano-Setien E., Alcahut-Rodriguez C., Martinez-Martin A., Sanchez-Lopez A., Segura T. Cerebrovascular disease in patients with COVID-19: neuroimaging, histological and clinical description. Brain J. Neurol. 30 Sep 2020;143(10):3089–3103. doi: 10.1093/brain/awaa239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Shao Y., Saredy J., Yang W.Y., Sun Y., Lu Y., Saaoud F., Drummer Ct, Johnson C., Xu K., Jiang X., Wang H., Yang X. Vascular endothelial cells and innate immunity. Arterioscler. Thromb. Vasc. Biol. 2020;40:e138–e152. doi: 10.1161/ATVBAHA.120.314330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kolivras A., Dehavay F., Delplace D., Feoli F., Meiers I., Milone L., Olemans C., Sass U., Theunis A., Thompson C.T., Van De Borne L., Richert B. Coronavirus (COVID-19) infection-induced chilblains: a case report with histopathologic findings. JAAD Case Rep. 2020 Jun;6(6):489–492. doi: 10.1016/j.jdcr.2020.04.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Nencioni A., Trzeciak S., Shapiro N.I. The microcirculation as a diagnostic and therapeutic target in sepsis. Intern Emerg Med. 2009;4:413–418. doi: 10.1007/s11739-009-0297-5. [DOI] [PubMed] [Google Scholar]
- 14.Hoffmann M., Kleine-Weber H., Schroeder S., Kruger N., Herrler T., Erichsen S., Schiergens T.S., Herrler G., Wu N.H., Nitsche A., Muller M.A., Drosten C., Pohlmann S. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181:271–280 e8. doi: 10.1016/j.cell.2020.02.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zou X., Chen K., Zou J., Han P., Hao J., Han Z. Single-cell RNA-seq data analysis on the receptor ACE2 expression reveals the potential risk of different human organs vulnerable to 2019-nCoV infection. Front. Med. 2020 Mar;12:1–8. doi: 10.1007/s11684-020-0754-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Wrapp D., Wang N., Corbett K.S., Goldsmith J.A., Hsieh C.L., Abiona O., Graham B.S., McLellan J.S. Cryo-EM structure of the 2019-nCoV spike in the prefusion conformation. Science. 2020;367:1260–1263. doi: 10.1126/science.abb2507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Tang N., Li D., Wang X., Sun Z. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J. Thromb. Haemostasis. 2020;18:844–847. doi: 10.1111/jth.14768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wu Z., McGoogan J.M. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: summary of a report of 72314 cases from the Chinese center for disease control and prevention. JAMA. 2020;323(13):1239–1242. doi: 10.1001/jama.2020.2648. [DOI] [PubMed] [Google Scholar]
- 19.Fan B.E., Chong V.C.L., Chan S.S.W., Lim G.H., Lim K.G.E., Tan G.B., Mucheli S.S., Kuperan P., Ong K.H. Hematologic parameters in patients with COVID-19 infection. Am. J. Hematol. 2020;95:E131–E134. doi: 10.1002/ajh.25774. [DOI] [PubMed] [Google Scholar]
- 20.Mehta P., McAuley D.F., Brown M., Sanchez E., Tattersall R.S., Manson J.J., Hlh Across Speciality Collaboration UK COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet. 2020;395:1033–1034. doi: 10.1016/S0140-6736(20)30628-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Diehl J.L., Peron N., Chocron R., Debuc B., Guerot E., Hauw-Berlemont C., Hermann B., Augy J.L., Younan R., Novara A., Langlais J., Khider L., Gendron N., Goudot G., Fagon J.F., Mirault T., Smadja D.M. Respiratory mechanics and gas exchanges in the early course of COVID-19 ARDS: a hypothesis-generating study. Ann. Intensive Care. 2020;10:95. doi: 10.1186/s13613-020-00716-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Guervilly C., Burtey S., Sabatier F., Cauchois R., Lano G., Abdili E., Daviet F., Arnaud L., Brunet P., Hraiech S., Jourde-Chiche N., Koubi M., Lacroix R., Pietri L., Berda Y., Robert T., Degioanni C., Velier M., Papazian L., Kaplanski G., Dignat-George F. Circulating endothelial cells as a marker of endothelial injury in severe COVID -19. J. Infect. Dis. 2020;222:1789–1793. doi: 10.1093/infdis/jiaa528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Mancuso P., Gidaro A., Gregato G., Raveane A., Cremonesi P., Quarna J., Caccia S., Gusso L., Rusconi S., Giacomelli A., Cogliati C., Bertolini F. Circulating endothelial progenitors are increased in COVID-19 patients and correlate with SARS-CoV-2 RNA in severe cases. J. Thromb. Haemostasis. 30 Aug 2020;18(10):2744–2750. doi: 10.1111/jth.15044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Goveia J., Rohlenova K., Taverna F., Treps L., Conradi L.C., Pircher A., Geldhof V., de Rooij L., Kalucka J., Sokol L., Garcia-Caballero M., Zheng Y., Qian J., Teuwen L.A., Khan S., Boeckx B., Wauters E., Decaluwe H., De Leyn P., Vansteenkiste J., Weynand B., Sagaert X., Verbeken E., Wolthuis A., Topal B., Everaerts W., Bohnenberger H., Emmert A., Panovska D., De Smet F., Staal F.J.T., McLaughlin R.J., Impens F., Lagani V., Vinckier S., Mazzone M., Schoonjans L., Dewerchin M., Eelen G., Karakach T.K., Yang H., Wang J., Bolund L., Lin L., Thienpont B., Li X., Lambrechts D., Luo Y., Carmeliet P. An integrated gene expression landscape profiling approach to identify lung tumor endothelial cell heterogeneity and angiogenic candidates. Canc. Cell. 2020;37:421. doi: 10.1016/j.ccell.2020.03.002. [DOI] [PubMed] [Google Scholar]
- 25.Fox S.E., Akmatbekov A., Harbert J.L., Li G., Quincy Brown J., Vander Heide R.S. Pulmonary and cardiac pathology in African American patients with COVID-19: an autopsy series from New Orleans. Lancet Respir Med. 2020;8:681–686. doi: 10.1016/S2213-2600(20)30243-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Middeldorp S., Coppens M., van Haaps T.F., Foppen M., Vlaar A.P., Muller M.C.A., Bouman C.C.S., Beenen L.F.M., Kootte R.S., Heijmans J., Smits L.P., Bonta P.I., van Es N. Incidence of venous thromboembolism in hospitalized patients with COVID-19. J. Thromb. Haemostasis. 2020 Aug;18(8):1995–2002. doi: 10.1111/jth.14888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Tong M., Jiang Y., Xia D., Xiong Y., Zheng Q., Chen F., Zou L., Xiao W., Zhu Y. Elevated expression of serum endothelial cell adhesion molecules in COVID-19 patients. J. Infect. Dis. 2020;222:894–898. doi: 10.1093/infdis/jiaa349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Dupont V., Kanagaratnam L., Goury A., Poitevin G., Bard M., Julien G., Bonnivard M., Champenois V., Noel V., Mourvillier B., Nguyen P. Excess soluble fms-like tyrosine kinase 1 correlates with endothelial dysfunction and organ failure in critically ill COVID-19 patients. Clin. Infect. Dis. 2020 July:1–4. doi: 10.1093/cid/ciaa1007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Smadja D.M., Guerin C.L., Chocron R., Yatim N., Boussier J., Gendron N., Khider L., Hadjadj J., Goudot G., Debuc B., Juvin P., Hauw-Berlemont C., Augy J.L., Peron N., Messas E., Planquette B., Sanchez O., Charbit B., Gaussem P., Duffy D., Terrier B., Mirault T., Diehl J.L. Angiopoietin-2 as a marker of endothelial activation is a good predictor factor for intensive care unit admission of COVID-19 Patients. Angiogenesis. 2020 Nov;23(4):611–620. doi: 10.1007/s10456-020-09730-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Lim H.S., Blann A.D., Chong A.Y., Freestone B., Lip G.Y. Plasma vascular endothelial growth factor, angiopoietin-1, and angiopoietin-2 in diabetes: implications for cardiovascular risk and effects of multifactorial intervention. Diabetes Care. 2004;27:2918–2924. doi: 10.2337/diacare.27.12.2918. [DOI] [PubMed] [Google Scholar]
- 31.Tobin M.J. Basing respiratory management of coronavirus on physiological principles. Am. J. Respir. Crit. Care Med. 2020 Jun 1;201(11):1319–1336. doi: 10.1164/rccm.202004-1076ED. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Desai T.R., Leeper N.J., Hynes K.L., Gewertz B.L. Interleukin-6 causes endothelial barrier dysfunction via the protein kinase C pathway. J. Surg. Res. 2002;104:118–123. doi: 10.1006/jsre.2002.6415. [DOI] [PubMed] [Google Scholar]
- 33.Krieg C., Letourneau S., Pantaleo G., Boyman O. Improved IL-2 immunotherapy by selective stimulation of IL-2 receptors on lymphocytes and endothelial cells. Proc. Natl. Acad. Sci. U. S. A. 2010;107:11906–11911. doi: 10.1073/pnas.1002569107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Rodriguez-Morales A.J., Cardona-Ospina J.A., Gutierrez-Ocampo E., Villamizar-Pena R., Holguin-Rivera Y., Escalera-Antezana J.P., Alvarado-Arnez L.E., Bonilla-Aldana D.K., Franco-Paredes C., Henao-Martinez A.F., Paniz-Mondolfi A., Lagos-Grisales G.J., Ramirez-Vallejo E., Suarez J.A., Zambrano L.I., Villamil-Gomez W.E., Balbin-Ramon G.J., Rabaan A.A., Harapan H., Dhama K., Nishiura H., Kataoka H., Ahmad T., Sah R. Latin American Network of Coronavirus Disease C-REahwlo. Clinical, laboratory and imaging features of COVID-19: a systematic review and meta-analysis. Trav. Med. Infect. Dis. 2020;34 doi: 10.1016/j.tmaid.2020.101623. 101623. Pubmed Exact. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Tavazzi G., Pellegrini C., Maurelli M., Belliato M., Sciutti F., Bottazzi A., Sepe P.A., Resasco T., Camporotondo R., Bruno R., Baldanti F., Paolucci S., Pelenghi S., Iotti G.A., Mojoli F., Arbustini E. Myocardial localization of coronavirus in COVID-19 cardiogenic shock. Eur. J. Heart Fail. 2020;22:911–915. doi: 10.1002/ejhf.1828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Long B., Brady W.J., Koyfman A., Gottlieb M. Cardiovascular complications in COVID-19. Am. J. Emerg. Med. 2020;38:1504–1507. doi: 10.1016/j.ajem.2020.04.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Oudit G.Y., Kassiri Z., Jiang C., Liu P.P., Poutanen S.M., Penninger J.M., Butany J. SARS-coronavirus modulation of myocardial ACE2 expression and inflammation in patients with SARS. Eur. J. Clin. Invest. 2009;39:618–625. doi: 10.1111/j.1365-2362.2009.02153.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Lu R., Zhao X., Li J., Niu P., Yang B., Wu H., Wang W., Song H., Huang B., Zhu N., Bi Y., Ma X., Zhan F., Wang L., Hu T., Zhou H., Hu Z., Zhou W., Zhao L., Chen J., Meng Y., Wang J., Lin Y., Yuan J., Xie Z., Ma J., Liu W.J., Wang D., Xu W., Holmes E.C., Gao G.F., Wu G., Chen W., Shi W., Tan W. Genomic characterisation and epidemiology of 2019 novel coronavirus: implications for virus origins and receptor binding. Lancet. 2020;395:565–574. doi: 10.1016/S0140-6736(20)30251-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ouldali N., Pouletty M., Mariani P., Beyler C., Blachier A., Bonacorsi S., Danis K., Chomton M., Maurice L., Le Bourgeois F., Caseris M., Gaschignard J., Poline J., Cohen R., Titomanlio L., Faye A., Melki I., Meinzer U. Emergence of Kawasaki disease related to SARS-CoV-2 infection in an epicentre of the French COVID-19 epidemic: a time-series analysis. Lancet Child Adolesc Health. 2020;4:662–668. doi: 10.1016/S2352-4642(20)30175-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Chang A., Croix M., Kenney P., Baron S., Hicar M. Serum responses of children with Kawasaki Disease against SARS-CoV-2 proteins. medRxiv. 2020:2020. doi: 10.1097/INF.0000000000002863. 05.24.20111732. [DOI] [PubMed] [Google Scholar]
- 41.Consiglio C.R., Cotugno N., Sardh F., Pou C., Amodio D., Rodriguez L., Tan Z., Zicari S., Ruggiero A., Pascucci G.R., Santilli V., Campbell T., Bryceson Y., Eriksson D., Wang J., Marchesi A., Lakshmikanth T., Campana A., Villani A., Rossi P., Team C.S., Landegren N., Palma P., Brodin P. The Immunology of multisystem inflammatory syndrome in children with COVID-19. Cell. 12 Nov 2020;183(4):968–981. doi: 10.1016/j.cell.2020.09.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Nishiga M., Wang D.W., Han Y., Lewis D.B., Wu J.C. COVID-19 and cardiovascular disease: from basic mechanisms to clinical perspectives. Nat. Rev. Cardiol. 2020;17:543–558. doi: 10.1038/s41569-020-0413-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Dofferhoff A.S.M., Piscaer I., Schurgers L.J., Visser M.P.J., van den Ouweland J.M.W., de Jong P.A., Gosens R., Hackeng T.M., van Daal H., Lux P., Maassen C., Karssemeijer E.G.A., Vermeer C., Wouters E.F.M., Kistemaker L.E.M., Walk J., Janssen R. Reduced vitamin K status as a potentially modifiable risk factor of severe COVID-19. Clin. Infect. Dis. 2020:1–8. doi: 10.1093/cid/ciaa1258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Cheng Y., Luo R., Wang K., Zhang M., Wang Z., Dong L., Li J., Yao Y., Ge S., Xu G. Kidney disease is associated with in-hospital death of patients with COVID-19. Kidney Int. 2020;97:829–838. doi: 10.1016/j.kint.2020.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Ahmadian E., Hosseiniyan Khatibi S.M., Razi Soofiyani S., Abediazar S., Shoja M.M., Ardalan M., Zununi Vahed S. Covid-19 and kidney injury: pathophysiology and molecular mechanisms. Rev. Med. Virol. 2020 doi: 10.1002/rmv.2176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Paniz-Mondolfi A., Bryce C., Grimes Z., Gordon R.E., Reidy J., Lednicky J., Sordillo E.M., Fowkes M. Central nervous system involvement by severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) J. Med. Virol. 2020;92:699–702. doi: 10.1002/jmv.25915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Hamming I., Timens W., Bulthuis M.L., Lely A.T., Navis G., van Goor H. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J. Pathol. 2004;203:631–637. doi: 10.1002/path.1570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Puelles V.G., Lütgehetmann M., Lindenmeyer M.T., Sperhake J.P., Wong M.N., Allweiss L., Chilla S., Heinemann A., Wanner N., Liu S., Braun F., Lu S., Pfefferle S., Schröder A.S., Edler C., Gross O., Glatzel M., Wichmann D., Wiech T., Kluge S., Pueschel K., Aepfelbacher M., Huber T.B. Multiorgan and renal tropism of SARS-CoV-2. N. Engl. J. Med. 2020;385:590–592. doi: 10.1056/NEJMc2011400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Politi L.S., Salsano E., Grimaldi M. Magnetic resonance imaging alteration of the brain in a patient with coronavirus disease 2019 (COVID-19) and anosmia. JAMA Neurol. 2020;77:1028–1029. doi: 10.1001/jamaneurol.2020.2125. [DOI] [PubMed] [Google Scholar]
- 50.Perrin P., Collongues N., Baloglu S., Bedo D., Bassand X., Lavaux T., Gautier-Vargas G., Keller N., Kremer S., Fafi-Kremer S., Moulin B., Benotmane I., Caillard S. Cytokine release syndrome-associated encephalopathy in patients with COVID-19. Eur. J. Neurol. 2021 Jan;28(1):248–258. doi: 10.1111/ene.14491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Erickson M.A., Banks W.A. Neuroimmune axes of the blood-brain barriers and blood-brain Interfaces: bases for physiological regulation, disease states, and pharmacological interventions. Pharmacol. Rev. 2018;70:278–314. doi: 10.1124/pr.117.014647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Dantzer R. Neuroimmune interactions: from the brain to the immune system and vice versa. Physiol. Rev. 2018;98:477–504. doi: 10.1152/physrev.00039.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Lamers M.M., Beumer J., van der Vaart J., Knoops K., Puschhof J., Breugem T.I., Ravelli R.B.G., Paul van Schayck J., Mykytyn A.Z., Duimel H.Q., van Donselaar E., Riesebosch S., Kuijpers H.J.H., Schipper D., van de Wetering W.J., de Graaf M., Koopmans M., Cuppen E., Peters P.J., Haagmans B.L., Clevers H. SARS-CoV-2 productively infects human gut enterocytes. Science. 2020;369:50–54. doi: 10.1126/science.abc1669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Zhang H., Kang Z., Gong H., Xu D., Wang J., Li Z., Cui X., Xiao J., Meng T., Zhou W., Liu J., Xu H. The digestive system is a potential route of 2019-nCov infection: a bioinformatics analysis based on single-cell transcriptomes. Gut. 2020;69(6):1010–1018. doi: 10.1136/gutjnl-2020-320953. [DOI] [Google Scholar]
- 55.Mao R., Qiu Y., He J.S., Tan J.Y., Li X.H., Liang J., Shen J., Zhu L.R., Chen Y., Iacucci M., Ng S.C., Ghosh S., Chen M.H. Manifestations and prognosis of gastrointestinal and liver involvement in patients with COVID-19: a systematic review and meta-analysis. Lancet Gastroenterol Hepatol. 2020;5:667–678. doi: 10.1016/S2468-1253(20)30126-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Recalcati S. Cutaneous manifestations in COVID-19: a first perspective. J. Eur. Acad. Dermatol. Venereol. 2020;34:e212–e213. doi: 10.1111/jdv.16387. [DOI] [PubMed] [Google Scholar]
- 57.Recalcati S., Barbagallo T., Frasin L.A., Prestinari F., Cogliardi A., Provero M.C., Dainese E., Vanzati A., Fantini F. Acral cutaneous lesions in the time of COVID-19. J. Eur. Acad. Dermatol. Venereol. 2020;34:e346–e347. doi: 10.1111/jdv.16533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Gianotti R., Veraldi S., Recalcati S., Cusini M., Ghislanzoni M., Boggio F., Fox L.P. Cutaneous clinico-pathological findings in three COVID-19-positive patients observed in the metropolitan area of milan, Italy. Acta Derm. Venereol. 2020;100 doi: 10.2340/00015555-3490. adv00124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Shanshal M., Ahmed H.S., Asfoor H., Salih R.I., Ali S.A., Aldabouni Yk. Impact of COVID-19 on medical practice: A nationwide survey of dermatologists and health care providers in Iraq. Clinics in Dermatology. 2020 doi: 10.1016/j.clindermatol.2020.11.010. 2020.07.26.20156380, In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Colmenero I., Santonja C., Alonso-Riano M., Noguera-Morel L., Hernandez-Martin A., Andina D., Wiesner T., Rodriguez-Peralto J.L., Requena L., Torrelo A. SARS-CoV-2 endothelial infection causes COVID-19 chilblains: histopathological, immunohistochemical and ultrastructural study of seven paediatric cases. Br. J. Dermatol. 2020;183:729–737. doi: 10.1111/bjd.19327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Scheuplein V.A., Seifried J., Malczyk A.H., Miller L., Hocker L., Vergara-Alert J., Dolnik O., Zielecki F., Becker B., Spreitzer I., Konig R., Becker S., Waibler Z., Muhlebach M.D. High secretion of interferons by human plasmacytoid dendritic cells upon recognition of Middle East respiratory syndrome coronavirus. J. Virol. 2015;89:3859–3869. doi: 10.1128/JVI.03607-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.de Wit E., van Doremalen N., Falzarano D., Munster V.J. SARS and MERS: recent insights into emerging coronaviruses. Nat. Rev. Microbiol. 2016;14:523–534. doi: 10.1038/nrmicro.2016.81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Tan L., Wang Q., Zhang D., Ding J., Huang Q., Tang Y.Q., Wang Q., Miao H. Lymphopenia predicts disease severity of COVID-19: a descriptive and predictive study. Signal Transduct Target Ther. 2020;5:33. doi: 10.1038/s41392-020-0148-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Ackermann M., Verleden S.E., Kuehnel M., Haverich A., Welte T., Laenger F., Vanstapel A., Werlein C., Stark H., Tzankov A., Li W.W., Li V.W., Mentzer S.J., Jonigk D. Pulmonary vascular endothelialitis, thrombosis, and angiogenesis in covid-19. N. Engl. J. Med. 2020;383:120–128. doi: 10.1056/NEJMoa2015432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Tellier R., Li Y., Cowling B.J., Tang J.W. Recognition of aerosol transmission of infectious agents: a commentary. BMC Infect. Dis. 2019;19:101. doi: 10.1186/s12879-019-3707-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Chu H., Chan J.F., Wang Y., Yuen T.T., Chai Y., Hou Y., Shuai H., Yang D., Hu B., Huang X., Zhang X., Cai J.P., Zhou J., Yuan S., Kok K.H., To K.K., Chan I.H., Zhang A.J., Sit K.Y., Au W.K., Yuen K.Y. Comparative replication and immune activation profiles of SARS-CoV-2 and SARS-CoV in human lungs: an ex vivo study with implications for the pathogenesis of COVID-19. Clin. Infect. Dis. 15 Sept 2020;71(6):1400–1409. doi: 10.1093/cid/ciaa410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Epidemiology Working Group for Ncip Epidemic Response CCfDC and Prevention The epidemiological characteristics of an outbreak of 2019 novel coronavirus diseases (COVID-19) in China. Zhonghua Liuxingbingxue Zazhi. 2020;41:145–151. doi: 10.3760/cma.j.issn.0254-6450.2020.02.003. [DOI] [PubMed] [Google Scholar]
- 68.Richardson S., Hirsch J.S., Narasimhan M., Crawford J.M., McGinn T., Davidson K.W., the Northwell C.-R.C., Barnaby D.P., Becker L.B., Chelico J.D., Cohen S.L., Cookingham J., Coppa K., Diefenbach M.A., Dominello A.J., Duer-Hefele J., Falzon L., Gitlin J., Hajizadeh N., Harvin T.G., Hirschwerk D.A., Kim E.J., Kozel Z.M., Marrast L.M., Mogavero J.N., Osorio G.A., Qiu M., Zanos T.P. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York city area. J. Am. Med. Assoc. 2020 May 26;323(20):2052–2059. doi: 10.1001/jama.2020.6775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Flammer A.J., Anderson T., Celermajer D.S., Creager M.A., Deanfield J., Ganz P., Hamburg N.M., Luscher T.F., Shechter M., Taddei S., Vita J.A., Lerman A. The assessment of endothelial function: from research into clinical practice. Circulation. 2012;126:753–767. doi: 10.1161/CIRCULATIONAHA.112.093245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Tissari J., Siren J., Meri S., Julkunen I., Matikainen S. IFN-alpha enhances TLR3-mediated antiviral cytokine expression in human endothelial and epithelial cells by up-regulating TLR3 expression. J. Immunol. 2005;174:4289–4294. doi: 10.4049/jimmunol.174.7.4289. [DOI] [PubMed] [Google Scholar]
- 71.Catanzaro M., Fagiani F., Racchi M., Corsini E., Govoni S., Lanni C. Immune response in COVID-19: addressing a pharmacological challenge by targeting pathways triggered by SARS-CoV-2. Signal Transduct Target Ther. 2020;5:84. doi: 10.1038/s41392-020-0191-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Qin C., Zhou L., Hu Z., Zhang S., Yang S., Tao Y., Xie C., Ma K., Shang K., Wang W., Tian D.S. Dysregulation of immune response in patients with COVID-19 in Wuhan, China. Clin. Infect. Dis. 2020 Jul 28;71(15):762–768. doi: 10.1093/cid/ciaa248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Karakas M., Jarczak D., Becker M., Roedl K., Addo M.M., Hein F., Bergmann A., Zimmermann J., Simon T.P., Marx G., Lutgehetmann M., Nierhaus A., Kluge S. Targeting endothelial dysfunction in eight extreme-critically ill patients with COVID-19 using the anti-adrenomedullin antibody adrecizumab (HAM8101) Biomolecules. 2020;10 doi: 10.3390/biom10081171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Qu D., Liu J., Lau C.W., Huang Y. IL-6 in diabetes and cardiovascular complications. Br. J. Pharmacol. 2014;171:3595–3603. doi: 10.1111/bph.12713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Teijaro J.R., Walsh K.B., Cahalan S., Fremgen D.M., Roberts E., Scott F., Martinborough E., Peach R., Oldstone M.B., Rosen H. Endothelial cells are central orchestrators of cytokine amplification during influenza virus infection. Cell. 2011;146:980–991. doi: 10.1016/j.cell.2011.08.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Teijaro J.R., Walsh K.B., Rice S., Rosen H., Oldstone M.B. Mapping the innate signaling cascade essential for cytokine storm during influenza virus infection. Proc. Natl. Acad. Sci. U. S. A. 2014;111:3799–3804. doi: 10.1073/pnas.1400593111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Kang S., Tanaka T., Inoue H., Ono C., Hashimoto S., Kioi Y., Matsumoto H., Matsuura H., Matsubara T., Shimizu K., Ogura H., Matsuura Y., Kishimoto T. IL-6 trans-signaling induces plasminogen activator inhibitor-1 from vascular endothelial cells in cytokine release syndrome. Proc. Natl. Acad. Sci. U. S. A. 2020;117:22351–22356. doi: 10.1073/pnas.2010229117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Alramthan A., Aldaraji W. Two cases of COVID-19 presenting with a clinical picture resembling chilblains: first report from the Middle East. Clin. Exp. Dermatol. 2020;45:746–748. doi: 10.1111/ced.14243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.El Hachem M., Diociaiuti A., Concato C., Carsetti R., Carnevale C., Ciofi Degli Atti M., Giovannelli L., Latella E., Porzio O., Rossi S., Stracuzzi A., Zaffina S., Onetti Muda A., Zambruno G., Alaggio R. A clinical, histopathological and laboratory study of 19 consecutive Italian paediatric patients with chilblain-like lesions: lights and shadows on the relationship with COVID-19 infection. J. Eur. Acad. Dermatol. Venereol. 2020 May 31 doi: 10.1111/jdv.16682. published ahead of print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Blanco-Melo D., Nilsson-Payant B.E., Liu W.-C., Møller R., Panis M., Sachs D., Albrecht R.A., tenOever B.R. SARS-CoV-2 launches a unique transcriptional signature from in vitro, ex vivo, and in vivo systems. bioRxiv. 2020 2020.03.24.004655. [Google Scholar]
- 81.Zhou Z., Ren L., Zhang L., Zhong J., Xiao Y., Jia Z., Guo L., Yang J., Wang C., Jiang S., Yang D., Zhang G., Li H., Chen F., Xu Y., Chen M., Gao Z., Yang J., Dong J., Liu B., Zhang X., Wang W., He K., Jin Q., Li M., Wang J. Heightened innate immune responses in the respiratory tract of COVID-19 patients. Cell Host Microbe. 2020;27:883–890 e2. doi: 10.1016/j.chom.2020.04.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Herman A., Peeters C., Verroken A., Tromme I., Tennstedt D., Marot L., Dachelet C., Gruson D., Hermans C., Baeck M. Evaluation of chilblains as a manifestation of the COVID-19 pandemic. JAMA Dermatol. 2020;156(9):998–1003. doi: 10.1001/jamadermatol.2020.2368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Viecca M., Radovanovic D., Forleo G.B., Santus P. Enhanced platelet inhibition treatment improves hypoxemia in patients with severe Covid-19 and hypercoagulability. A case control, proof of concept study. Pharmacol. Res. 2020;158:104950. doi: 10.1016/j.phrs.2020.104950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Ladikou E.E., Sivaloganathan H., Milne K.M., Arter W.E., Ramasamy R., Saad R., Stoneham S.M., Philips B., Eziefula A.C., Chevassut T. Von Willebrand factor (vWF): marker of endothelial damage and thrombotic risk in COVID-19? Clin. Med. 2020;20:e178–e182. doi: 10.7861/clinmed.2020-0346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Goshua G., Pine A.B., Meizlish M.L., Chang C.H., Zhang H., Bahel P., Baluha A., Bar N., Bona R.D., Burns A.J., Dela Cruz C.S., Dumont A., Halene S., Hwa J., Koff J., Menninger H., Neparidze N., Price C., Siner J.M., Tormey C., Rinder H.M., Chun H.J., Lee A.I. Endotheliopathy in COVID-19-associated coagulopathy: evidence from a single-centre, cross-sectional study. Lancet Haematol. 2020;7:e575–e582. doi: 10.1016/S2352-3026(20)30216-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Nachman R.L., Rafii S. Platelets, petechiae, and preservation of the vascular wall. N. Engl. J. Med. 2008;359:1261–1270. doi: 10.1056/NEJMra0800887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Collen D., Lijnen H.R. Basic and clinical aspects of fibrinolysis and thrombolysis. Blood. 1991;78:3114–3124. [PubMed] [Google Scholar]
- 88.George J.N., Nester C.M. Syndromes of thrombotic microangiopathy. N. Engl. J. Med. 2014;371:654–666. doi: 10.1056/NEJMra1312353. [DOI] [PubMed] [Google Scholar]
- 89.Levi M., Thachil J., Iba T., Levy J.H. Coagulation abnormalities and thrombosis in patients with COVID-19. Lancet Haematol. 2020;7:e438–e440. doi: 10.1016/S2352-3026(20)30145-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Gavriilaki E., Anagnostopoulos A., Mastellos D.C. Complement in thrombotic microangiopathies: unraveling ariadne's thread into the labyrinth of complement therapeutics. Front. Immunol. 2019;10:337. doi: 10.3389/fimmu.2019.00337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Gao T., Hu M., Zhang X., Li H., Zhu L., Liu H., Dong Q., Zhang Z., Wang Z., Hu Y., Fu Y., Jin Y., Li K., Zhao S., Xiao Y., Luo S., Li L., Zhao L., Liu J., Zhao H., Liu Y., Yang W., Peng J., Chen X., Li P., Liu Y., Xie Y., Song J., Zhang L., Ma Q., Bian X., Chen W., Liu X., Mao Q., Cao C. Highly pathogenic coronavirus N protein aggravates lung injury by MASP-2-mediated complement over-activation. medRxiv. 2020:2020. 03.29.20041962. [Google Scholar]
- 92.Magro C., Mulvey J.J., Berlin D., Nuovo G., Salvatore S., Harp J., Baxter-Stoltzfus A., Laurence J. Complement associated microvascular injury and thrombosis in the pathogenesis of severe COVID-19 infection: a report of five cases. Transl. Res. 2020;220:1–13. doi: 10.1016/j.trsl.2020.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Carvelli J., Demaria O., Vely F., Batista L., Benmansour N.C., Fares J., Carpentier S., Thibult M.L., Morel A., Remark R., Andre P., Represa A., Piperoglou C., Explore C-IpHg, Explore C-Mig, Cordier P.Y., Le Dault E., Guervilly C., Simeone P., Gainnier M., Morel Y., Ebbo M., Schleinitz N., Vivier E. Association of COVID-19 inflammation with activation of the C5a-C5aR1 axis. Nature. 2020;588:146–150. doi: 10.1038/s41586-020-2600-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Merle N.S., Noe R., Halbwachs-Mecarelli L., Fremeaux-Bacchi V., Roumenina L.T. Complement system Part II: role in immunity. Front. Immunol. 2015;6:257. doi: 10.3389/fimmu.2015.00257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Polley M.J., Nachman R.L. Human complement in thrombin-mediated platelet function: uptake of the C5b-9 complex. J. Exp. Med. 1979;150:633–645. doi: 10.1084/jem.150.3.633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Hattori R., Hamilton K.K., McEver R.P., Sims P.J. Complement proteins C5b-9 induce secretion of high molecular weight multimers of endothelial von Willebrand factor and translocation of granule membrane protein GMP-140 to the cell surface. J. Biol. Chem. 1989;264:9053–9060. [PubMed] [Google Scholar]
- 97.Krarup A., Wallis R., Presanis J.S., Gal P., Sim R.B. Simultaneous activation of complement and coagulation by MBL-associated serine protease 2. PloS One. 2007;2:e623. doi: 10.1371/journal.pone.0000623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Leppkes M., Knopf J., Naschberger E., Lindemann A., Singh J., Herrmann I., Sturzl M., Staats L., Mahajan A., Schauer C., Kremer A.N., Volkl S., Amann K., Evert K., Falkeis C., Wehrfritz A., Rieker R.J., Hartmann A., Kremer A.E., Neurath M.F., Munoz L.E., Schett G., Herrmann M. Vascular occlusion by neutrophil extracellular traps in COVID-19. EBioMedicine. 2020;58:102925. doi: 10.1016/j.ebiom.2020.102925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Schurink B., Roos E., Radonic T., Barbe E., Bouman C.S.C., de Boer H.H., de Bree G.J., Bulle E.B., Aronica E.M., Florquin S., Fronczek J., Heunks L.M.A., de Jong M.D., Guo L., du Long R., Lutter R., Molenaar P.C.G., Neefjes-Borst E.A., Niessen H.W.M., van Noesel C.J.M., Roelofs J., Snijder E.J., Soer E.C., Verheij J., Vlaar A.P.J., Vos W., van der Wel N.N., van der Wal A.C., van der Valk P., Bugiani M. Viral presence and immunopathology in patients with lethal COVID-19: a prospective autopsy cohort study. Lancet Microbe. 2020;1:e290–e299. doi: 10.1016/S2666-5247(20)30144-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Nicolai L., Leunig A., Brambs S., Kaiser R., Weinberger T., Weigand M., Muenchhoff M., Hellmuth J.C., Ledderose S., Schulz H., Scherer C., Rudelius M., Zoller M., Hochter D., Keppler O., Teupser D., Zwissler B., von Bergwelt-Baildon M., Kaab S., Massberg S., Pekayvaz K., Stark K. Immunothrombotic dysregulation in COVID-19 pneumonia is associated with respiratory failure and coagulopathy. Circulation. 2020;142:1176–1189. doi: 10.1161/CIRCULATIONAHA.120.048488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Veras F.P., Pontelli M.C., Silva C.M., Toller-Kawahisa J.E., de Lima M., Nascimento D.C., Schneider A.H., Caetite D., Tavares L.A., Paiva I.M., Rosales R., Colon D., Martins R., Castro I.A., Almeida G.M., Lopes M.I.F., Benatti M.N., Bonjorno L.P., Giannini M.C., Luppino-Assad R., Almeida S.L., Vilar F., Santana R., Bollela V.R., Auxiliadora-Martins M., Borges M., Miranda C.H., Pazin-Filho A., da Silva L.L.P., Cunha L.D., Zamboni D.S., Dal-Pizzol F., Leiria L.O., Siyuan L., Batah S., Fabro A., Mauad T., Dolhnikoff M., Duarte-Neto A., Saldiva P., Cunha T.M., Alves-Filho J.C., Arruda E., Louzada-Junior P., Oliveira R.D., Cunha F.Q. SARS-CoV-2-triggered neutrophil extracellular traps mediate COVID-19 pathology. J. Exp. Med. 2020:217. doi: 10.1084/jem.20201129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Levi M., Thachil J. Coronavirus disease 2019 coagulopathy: disseminated intravascular coagulation and thrombotic microangiopathy-either, neither, or both. Semin. Thromb. Hemost. 2020 Oct;46(7):781–784. doi: 10.1055/s-0040-1712156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Levi M., van der Poll T., Buller H.R. Bidirectional relation between inflammation and coagulation. Circulation. 2004;109:2698–2704. doi: 10.1161/01.CIR.0000131660.51520.9A. [DOI] [PubMed] [Google Scholar]
- 104.Khider L., Gendron N., Goudot G., Chocron R., Hauw-Berlemont C., Cheng C., Rivet N., Pere H., Roffe A., Clerc S., Lebeaux D., Debuc B., Veyer D., Rance B., Gaussem P., Bertil S., Badoual C., Juvin P., Planquette B., Messas E., Sanchez O., Hulot J.S., Diehl J.L., Mirault T., Smadja D.M. Curative anticoagulation prevents endothelial lesion in COVID-19 patients. J. Thromb. Haemostasis. 2020 Jul 29;18(9):2391–2399. doi: 10.1111/jth.14968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Iba T., Levy J.H., Connors J.M., Warkentin T.E., Thachil J., Levi M. The unique characteristics of COVID-19 coagulopathy. Crit. Care. 2020;24:360. doi: 10.1186/s13054-020-03077-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Stahl K., Gronski P.A., Kiyan Y., Seeliger B., Bertram A., Pape T., Welte T., Hoeper M.M., Haller H., David S. Injury to the endothelial glycocalyx in critically ill COVID-19 patients. Am. J. Respir. Crit. Care Med. 2020 Oct 15;202(8):1178–1181. doi: 10.1164/rccm.202007-2676LE. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Bertram S., Heurich A., Lavender H., Gierer S., Danisch S., Perin P., Lucas J.M., Nelson P.S., Pohlmann S., Soilleux E.J. Influenza and SARS-coronavirus activating proteases TMPRSS2 and HAT are expressed at multiple sites in human respiratory and gastrointestinal tracts. PloS One. 2012;7 doi: 10.1371/journal.pone.0035876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Rice G.I., Thomas D.A., Grant P.J., Turner A.J., Hooper N.M. Evaluation of angiotensin-converting enzyme (ACE), its homologue ACE2 and neprilysin in angiotensin peptide metabolism. Biochem. J. 2004;383:45–51. doi: 10.1042/BJ20040634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Vickers C., Hales P., Kaushik V., Dick L., Gavin J., Tang J., Godbout K., Parsons T., Baronas E., Hsieh F., Acton S., Patane M., Nichols A., Tummino P. Hydrolysis of biological peptides by human angiotensin-converting enzyme-related carboxypeptidase. J. Biol. Chem. 2002;277:14838–14843. doi: 10.1074/jbc.M200581200. [DOI] [PubMed] [Google Scholar]
- 110.Sampaio W.O., Souza dos Santos R.A., Faria-Silva R., da Mata Machado L.T., Schiffrin E.L., Touyz R.M. Angiotensin-(1-7) through receptor Mas mediates endothelial nitric oxide synthase activation via Akt-dependent pathways. Hypertension. 2007;49:185–192. doi: 10.1161/01.HYP.0000251865.35728.2f. [DOI] [PubMed] [Google Scholar]
- 111.Fernandes D., Assreuy J. Nitric oxide and vascular reactivity in sepsis. Shock. 2008;30(Suppl 1):10–13. doi: 10.1097/SHK.0b013e3181818518. [DOI] [PubMed] [Google Scholar]
- 112.Qian S.Z., Pan J.Y. COVID-19 with limb ischemic necrosis. J. Cardiothorac. Vasc. Anesth. 2020 Oct;34(10):2846–2847. doi: 10.1053/j.jvca.2020.03.063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.John S., Kesav P., Mifsud V.A., Piechowski-Jozwiak B., Dibu J., Bayrlee A., Elkambergy H., Roser F., Elhammady M.S., Zahra K., Hussain S.I. Characteristics of large-vessel occlusion associated with COVID-19 and ischemic stroke. AJNR Am J Neuroradiol. 2020 Dec;41(12):2263–2268. doi: 10.3174/ajnr.A6799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Akaike T., Maeda H. Nitric oxide and virus infection. Immunology. 2000;101:300–308. doi: 10.1046/j.1365-2567.2000.00142.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Napoli C., Ignarro L.J. Nitric oxide and pathogenic mechanisms involved in the development of vascular diseases. Arch Pharm. Res. (Seoul) 2009;32:1103–1108. doi: 10.1007/s12272-009-1801-1. [DOI] [PubMed] [Google Scholar]
- 116.Uchimido R., Schmidt E.P., Shapiro N.I. The glycocalyx: a novel diagnostic and therapeutic target in sepsis. Crit. Care. 2019;23:16. doi: 10.1186/s13054-018-2292-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Tejero J., Shiva S., Gladwin M.T. Sources of vascular nitric oxide and reactive oxygen species and their regulation. Physiol. Rev. 2019;99:311–379. doi: 10.1152/physrev.00036.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Cheung C.Y., Poon L.L., Ng I.H., Luk W., Sia S.F., Wu M.H., Chan K.H., Yuen K.Y., Gordon S., Guan Y., Peiris J.S. Cytokine responses in severe acute respiratory syndrome coronavirus-infected macrophages in vitro: possible relevance to pathogenesis. J. Virol. 2005;79:7819–7826. doi: 10.1128/JVI.79.12.7819-7826.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Akerstrom S., Gunalan V., Keng C.T., Tan Y.J., Mirazimi A. Dual effect of nitric oxide on SARS-CoV replication: viral RNA production and palmitoylation of the S protein are affected. Virology. 2009;395:1–9. doi: 10.1016/j.virol.2009.09.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Becker L., Prado K., Foppa M., Martinelli N., Aguiar C., Furian T., Clausell N., Rohde L.E. Endothelial dysfunction assessed by brachial artery ultrasound in severe sepsis and septic shock. J. Crit. Care. 2012;27:316. doi: 10.1016/j.jcrc.2011.08.002. e9-14. [DOI] [PubMed] [Google Scholar]
- 121.Silber H.A., Bluemke D.A., Ouyang P., Du Y.P., Post W.S., Lima J.A. The relationship between vascular wall shear stress and flow-mediated dilation: endothelial function assessed by phase-contrast magnetic resonance angiography. J. Am. Coll. Cardiol. 2001;38:1859–1865. doi: 10.1016/s0735-1097(01)01649-7. [DOI] [PubMed] [Google Scholar]
- 122.Lopes R.D., Macedo A.V.S., de Barros E.S.P.G.M., Moll-Bernardes R.J., Dos Santos T.M., Mazza L., Feldman A., D'Andrea Saba Arruda G., de Albuquerque D.C., Camiletti A.S., de Sousa A.S., de Paula T.C., Giusti K.G.D., Domiciano R.A.M., Noya-Rabelo M.M., Hamilton A.M., Loures V.A., Dionisio R.M., Furquim T.A.B., De Luca F.A., Dos Santos Sousa I.B., Bandeira B.S., Zukowski C.N., de Oliveira R.G.G., Ribeiro N.B., de Moraes J.L., Petriz J.L.F., Pimentel A.M., Miranda J.S., de Jesus Abufaiad B.E., Gibson C.M., Granger C.B., Alexander J.H., de Souza OF, Investigators B.C. Effect of discontinuing vs continuing angiotensin-converting enzyme inhibitors and angiotensin II receptor blockers on days alive and out of the hospital in patients admitted with COVID-19: a randomized clinical trial. J. Am. Med. Assoc. 2021;325:254–264. doi: 10.1001/jama.2020.25864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Vaduganathan M., Vardeny O., Michel T., McMurray J.J.V., Pfeffer M.A., Solomon S.D. Renin-angiotensin-aldosterone system inhibitors in patients with covid-19. N. Engl. J. Med. 2020;382:1653–1659. doi: 10.1056/NEJMsr2005760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Zoufaly A., Poglitsch M., Aberle J.H., Hoepler W., Seitz T., Traugott M., Grieb A., Pawelka E., Laferl H., Wenisch C., Neuhold S., Haider D., Stiasny K., Bergthaler A., Puchhammer-Stoeckl E., Mirazimi A., Montserrat N., Zhang H., Slutsky A.S., Penninger J.M. Human recombinant soluble ACE2 in severe COVID-19. Lancet Respir Med. 2020;8:1154–1158. doi: 10.1016/S2213-2600(20)30418-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.O'Driscoll G., Green D., Taylor R.R. Simvastatin, an HMG-coenzyme A reductase inhibitor, improves endothelial function within 1 month. Circulation. 1997;95:1126–1131. doi: 10.1161/01.cir.95.5.1126. [DOI] [PubMed] [Google Scholar]
- 126.Bustos C., Hernandez-Presa M.A., Ortego M., Tunon J., Ortega L., Perez F., Diaz C., Hernandez G., Egido J. HMG-CoA reductase inhibition by atorvastatin reduces neointimal inflammation in a rabbit model of atherosclerosis. J. Am. Coll. Cardiol. 1998;32:2057–2064. doi: 10.1016/s0735-1097(98)00487-2. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.