Abstract
The pathology and neurodegeneration in type 2 diabetes- (T2D-) mediated Alzheimer's disease (AD) have been reported in several studies. Despite the lack of information regarding the basic underlying mechanisms involved in the development of T2D-mediated AD, some common features of the two conditions have been reported, such as brain atrophy, reduced cerebral glucose metabolism, and insulin resistance. T2D phenotypes such as glucose dyshomeostasis, insulin resistance, impaired insulin signaling, and systemic inflammatory cytokines have been shown to be involved in the progression of AD pathology by increasing amyloid-beta accumulation, tau hyperphosphorylation, and overall neuroinflammation. Similarly, oxidative stress, mitochondrial dysfunction, and the generation of advanced glycation end products (AGEs) and their receptor (RAGE) as a result of chronic hyperglycemia may serve as critical links between diabetes and AD. The natural dietary polyflavonoid anthocyanin enhances insulin sensitivity, attenuates insulin resistance at the level of the target tissues, inhibits free fatty acid oxidation, and abrogates the release of peripheral inflammatory cytokines in obese (prediabetic) individuals, which are responsible for insulin resistance, systemic hyperglycemia, systemic inflammation, brain metabolism dyshomeostasis, amyloid-beta accumulation, and neuroinflammatory responses. In this review, we have shown that obesity may induce T2D-mediated AD and assessed the recent therapeutic advances, especially the use of anthocyanin, against T2D-mediated AD pathology. Taken together, the findings of current studies may help elucidate a new approach for the prevention and treatment of T2D-mediated AD by using the polyflavonoid anthocyanin.
1. Introduction
1.1. Obesity and Type 2 Diabetes
Over 40% of the world's population is overweight and obese, which is one of the crucial risk factors for insulin resistance, peripheral inflammation, and type 2 diabetes (T2D) [1]. The incidence of obesity is growing in the adult population in the United States, which is related to unhealthy lifestyle habits such as high calorie intake, inadequate exercise, and sedentary lifestyle [2]. Approximately 69.2% of American citizens are obese or overweight [3]. Alarmingly, the prevalence of obesity among young adults has been increasing rapidly.
Diabetic individuals may be more vulnerable to chronic type 2 diabetes mellitus (T2DM) and cardiovascular disorders (CVDs) [4]. Recent evidence suggests that obesity is an important risk factor for T2D. Obesity and diabetes share the same pathways of activation of inflammatory mediators, which play a role in insulin resistance and the pathogenesis of T2D. The pathogenesis of these disorders is associated with the number and phenotype of adipose tissue macrophages. A previous line of evidence suggested that adipose tissue macrophages release proinflammatory mediators to classically activate the M1 macrophages, which may contribute to insulin resistance and diabetes mellitus. In contrast, the adipose tissue from healthy individuals contains M2 phenotype inflammatory macrophages.
When fat cells in adipose tissue rupture, they usually produce a large quantity of free fatty acids (FFAs). Peripheral adipose tissues are also known to release various kinds of cytokines [5]. In addition to storing FFAs, excessive fat and adipose tissue also contain a large number of other compounds such as angiotensin-2, resistin, and inflammatory cytokines, such as TNF-α, IL-1β, and interleukin-6. The release of an excessive amount of FFAs in the circulation increases the plasma FFA level in obese individuals, and elevated plasma FFA levels prevent the antilipolytic action of insulin, which further increases FFA levels. The presence of a large quantity of FFAs in plasma also leads to peripheral insulin resistance [5–11]. Although FFAs are involved in increasing the mass of pancreatic beta-cells and improving insulin secretion to increase the availability of insulin to absorb the blood glucose derived from foods and via gluconeogenesis (metabolic process to obtain glucose from noncarbohydrate sources), a continuous and chronic increase in FFA levels can disrupt lipid metabolism, causing lipotoxicity, insulin resistance, decreased beta-cell function, and, eventually, T2D [12] (Figure 1).
Figure 1.
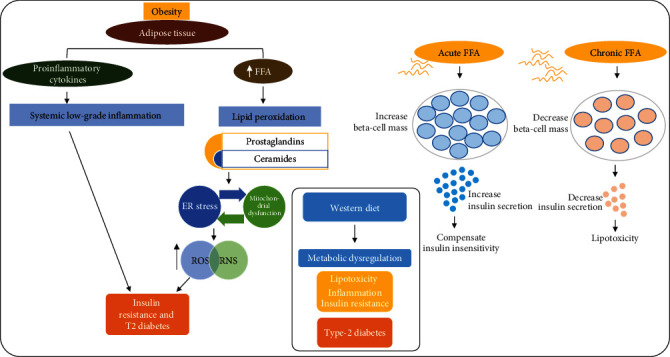
Obesity, free fatty acids, and insulin resistance. A diagrammatic representation showing the role of obesity in metabolic dysregulation via the release of inflammatory cytokines, oxidative stress, and lipotoxicity. Secretion of FFA and proinflammatory cytokines results in ROS/oxidative stress-mediated JNK activation. Inflammatory cytokines cause low-grade inflammation in peripheral organs and induce type 2 diabetes. Similarly, chronic FFA treatment induces insulin resistance and beta-cell dysfunction and causes type 2 diabetes. ROS: reactive oxygen species; RNS: reactive nitrosative stress; FFA: free fatty acid; ER: endoplasmic reticulum.
1.2. Type 2 Diabetes Mellitus and Alzheimer's Disease
Several basic mechanisms are involved in the development of T2D. In one such mechanism, increased secretion of FFAs by adipose tissue in obese individuals induces the production of plasma malondialdehyde (MDA), which is a reactive product of lipid peroxidation. Increased lipid peroxidation subsequently induces the production of the toxic lipid ceramide and oxidative stress. The increased lipid peroxidation induced by FFAs also causes the release of prostaglandins. Moreover, a chronic increase in FFA levels induces insulin resistance and beta-cell dysfunction, mentioned previously. Thus, the reduction of elevated plasma FFA levels might be an important therapeutic target in obesity and T2D [13]. AGEs, which are also called mycotoxins, induce reactive oxygen species (ROS) and trigger the release of proinflammatory cytokines, causing sustained activation of innate immunity, via transcriptional factors such as nuclear factor-kappa B (NF-κB) [14]. These prostaglandins and ceramides then activate the immune cells to secrete IL-1β, TNF-α, and IL-6.
Adipose tissues are also responsible for the release of proinflammatory cytokines and some other hormones. Increased release of proinflammatory cytokines can lead to the development of insulin resistance. Elevated oxidative stress causes lipid peroxidation and activation of several inflammatory signaling pathways [15], such as the c-Jun N-terminal kinase (JNK) and MAP kinase pathways, which leads to diabetes and pancreatic cell death [16–18]. Extensive research studies have shown that diabetes plays a significant role in the execution of Alzheimer's disease (AD) pathology [19]. Both conditions share many pathological features.
1.3. Anthocyanins and Their Effects against Obesity, Diabetes, and Alzheimer's Disease
Anthocyanins (a Greek word; from the flower) are a group of extensively studied natural compounds that belong to the subgroup of flavonoids [20]. They are water-soluble compounds that provide color to many fruits and vegetables; the blue color of various fruits and vegetables is attributable to the presence of anthocyanins in them. The ability of anthocyanins to form flavylium cations distinguishes them from other flavonoids. A total of 27 aglycones and 700 anthocyanins have been classified based on their chemical structures. Anthocyanins naturally occur in cherries, blueberries, strawberries, purple grapes, raspberries, black currants, and red wine [21]. The amount and composition of anthocyanins differ in different foods, varying from 0.1% to 1%. Foods that are rich sources of anthocyanins include blueberries, black soybeans, black plums, grapes, and black rice. Among the various types of fruits, berries are considered a rich source of anthocyanins (Figure 2).
Figure 2.

Rich sources of anthocyanins. Fruits and vegetables rich in the polyflavonoid anthocyanin.
Anthocyanins have recently gained tremendous interest because of their significant therapeutic roles in different diseases, most prominently metabolic and neurodegenerative diseases, since anthocyanin-rich foods have been used in traditional medicines [22]. Although these foods have been extensively used for the management of different diseases, little attention has been paid to their phytochemicals and the related mechanistic approaches. Studies have shown strong antioxidant and anti-inflammatory effects of anthocyanins against different neurodegenerative diseases. Several climatic factors (pH, light, and temperature), oxidation, and enzymolysis can affect the levels of anthocyanins. Recent findings have shown that anthocyanins are stable at low pH, i.e., in acidic conditions. Anthocyanins have been reported to show potent anti-inflammatory, antioxidant, anticancer, and antidiabetic activities [20, 22–33]. The daily consumption of natural dietary anthocyanin has been recently suggested to prevent various diseases and improve immune function [34–37].
2. Methodological Approaches
This review article is aimed at summarizing the findings of studies on the interaction of T2D and AD and the reported preventive and therapeutic effects of anthocyanins. The motivation for preparing this review was based on our previous studies on the role of anthocyanins against different neurodegenerative diseases (APP/PSEN-1 transgenic mice and LPS-treated mice). Here, we searched for potential research articles focusing on obesity, diabetes, and AD. In addition, to identify studies on the role of anthocyanins in obesity, diabetes, and AD, we conducted searches using the keywords “anthocyanins,” obesity,” and “diabetes” in all available and independent databases. For a clear understanding of these studies, the abstracts were fully studied and the main findings were recorded. For comparisons among the different groups, the anthocyanin-treated group was compared with the toxin-treated/model group. The dose and route of administration of anthocyanin, duration of treatment, and toxic compounds used in these studies were not considered. All studies covering animal and cellular models were included.
3. Risk Factors for Metabolic Dysfunction Induced by Type 2 Diabetes
3.1. Gut Dysbiosis and Type 2 Diabetes
As previously described, the presence of proinflammatory cytokines in various body tissues, particularly those that participate in metabolic regulation, such as muscles, livers, and adipose tissues, augments insulin resistance and T2D [38]. Growing evidence supports the concept that intestinal microbiomes play a critical role in the development of T2D, and patients with T2D have an altered gut microbiota. These findings have shown the gut microbiota to be one of the important contributors to the development of T2D. Changes in gut microbiota exaggerate systemic low-grade chronic inflammation, alter gut permeability, affect lipid and glucose metabolism, modulate insulin sensitivity, and disturb energy homeostasis. Several important mechanisms have been reported to describe the effects of gut microbiota on insulin resistance and T2D, of which metabolic endotoxemia is one of the most important. Recent studies have demonstrated that most gram-negative bacteria in the gut produce a very potent immunogenic endotoxin known as lipopolysaccharide (LPS). LPS is a component of the cell wall of gram-negative bacteria such as E. coli that causes metabolic endotoxemia. Obesity and increased fat intake induce the abundance of opportunistic gram-negative bacteria in the gut, which later produce LPS [39, 40].
Evidence from multiple studies indicates that opportunistic bacteria secrete LPS and other toxins inside the gut, abnormally increasing gut permeability by disrupting tight junction proteins [41]. This condition is also known as a leaky gut. A leaky gut allows several inflammatory cytokines and toxins to contaminate the blood and subsequently cause systemic inflammation. Furthermore, the presence of LPS in the gut causes epithelial cell inflammation. The secreted LPS further activates immune cells such as microglia and astrocytes. These cells secrete proinflammatory cytokines into the blood and promote their contamination. Once these proinflammatory cytokines enter the peripheral organs such as the liver, pancreas, and adipose tissue via the leaky gut, they cause peripheral inflammation, pancreatic cell dysfunction, insulin resistance, and T2D [41, 42]. Recent evidence suggests that Toll-like receptor 4 (TLR4) is a receptor for LPS present on the microglial surface. Once LPS binds to TLR4, it activates several signaling pathways such as the MAP kinase, NF-κB, and inflammatory pathways [43, 44]. Activation of these pathways leads to the development of glucose intolerance and hepatic insulin resistance [45, 46] (Figure 3).
Figure 3.
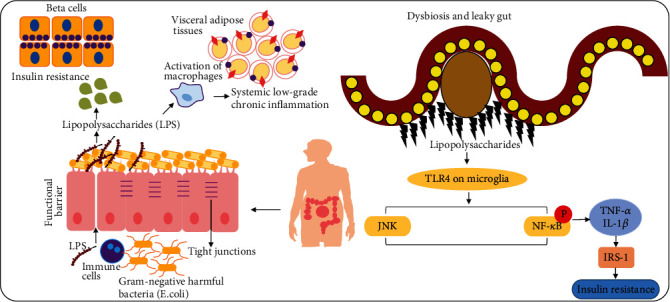
Dysbiosis, leaky gut, inflammatory cytokines, and insulin resistance. A simple illustration showing the release of lipopolysaccharides (LPS) and disruption of tight junctions in the intestinal wall resulting in the release of inflammatory cytokines, which enter the blood and pancreatic beta-cells. In addition, the TLR4 receptors in LPS play a role in the release of microglial cells. The reactive microglial cells secrete inflammatory cytokines and cause insulin resistance. NF-κB: nuclear factor-kappa B; TNF-α: tissue necrosis factor-alpha; TLR4: Toll-like receptor 4.
3.2. Lifestyle, Environmental Factors, and Diabetes
Physical inactivity, sedentary lifestyle, alcohol consumption, and smoking are common factors that play a role in the development of T2D. Treatable obstructive sleep apnea (OSA) and increased body weight are also considered important risk factors for T2D. Obstructive sleep apnea is more common in patients with T2D than in the general population. Similarly, diet and lack of exercise are other risk factors for T2D. Studies have shown that a diet with low fiber and high glycemic content is positively associated with T2D pathophysiology, while dietary fatty acid-rich foods may overcome the incidence of diabetes and insulin resistance [47–55]. Other studies have shown that saturated fatty acid-rich foods, sweets, and refined carbohydrates are potentially associated with an increased risk of diabetes. Furthermore, numerous observational studies have shown that the consumption of foods such as omega-6 industrial seed oils, cereal grains, and fructose in excessive quantities mediates peripheral inflammation and insulin resistance [56–60]. Fructose, also known as fruit sugar, is a simple ketonic monosaccharide found in many plants. There is increasing evidence that the excessive consumption of fructose, e.g., in the form of high-fructose corn syrup (HFCS) in soft drinks, may cause nonalcoholic fatty liver disease, which is strongly associated with diabetes and insulin resistance [61]. Apart from the fructose mentioned previously, some studies have suggested that saturated fatty acids (SFAs) are also hazardous in relation to these disorders. A palmitic acid-rich high-fat diet is associated with reduced sensitivity and enhanced atherosclerotic conditions, while a higher intake of eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) may enhance insulin sensitivity. A detailed discussion may be found elsewhere [62]. Polyunsaturated fat and protein react with fructose and form AGEs. Recent studies provide compelling evidence for the idea that the complications that occur as a result of diabetes, such as neuropathy, nephropathy, cardiomyopathy, and retinopathy, are due to protein glycation and the formation of AGEs [63–67]. Diets with insufficient nutrients may affect the function of beta-cells. For example, a low-protein diet leads to the dysfunction of pancreatic cells. Moreover, several hormones such as stress hormones, such as adrenaline and cortisol, increase blood sugar by releasing glucose from the liver. Therefore, stress can directly increase blood glucose levels and affect body metabolism [68–73].
Viral infections can also directly damage the beta-cells or induce the release of proinflammatory cytokines. Upon exposure to the virus infection, the immune system of the pancreas becomes activated and initiates the release of inflammatory cytokines, which later cause beta-cell damage. The important viruses that are involved in the infection of pancreatic beta-cells are enterovirus, congenital rubella, and cytomegalovirus [74–77].
3.3. Drugs and Diabetes
Drugs such as cyclosporin, diphenylhydantoin, thiazide diuretics, and pentamidine are known to induce hyperglycemia-induced diabetes. Some experimental chemical agents such as streptozotocin and afloxan are also involved in the induction of diabetes. Numerous studies have indicated that the exogenous sources of advanced lipoperoxidation and glycation end products (ALEs and AGEs) are dairy products processed with sugar, roasted foods, and fried foods. Foods processed at high temperatures are a rich source of lipid-associated prooxidants and heat-accelerated proteins [78–84].
3.4. Shared Links between Diabetes and AD Pathology
An increasing body of evidence indicates that metabolic disorders increase the incidence of AD by disturbing glucose metabolism and its bioavailability in the brain [85]. The brain requires high amounts of glucose and oxygen for proper functioning. Therefore, the chances of the release of ROS will be greater in the brain than in other organs. Elevated oxidative stress by different mechanisms causes neuronal cell loss and neurodegeneration [86–88]. Oxidative stress plays a crucial role in the development of T2D-mediated Alzheimer's pathology. Recent studies have shown that chronic hyperglycemia produces oxidative stress in various organs, including the brain. Other studies have reported that hyperglycemia affects the antioxidant system and accelerates the production of free radicals in neuronal cells. Several other studies have demonstrated that persistent hyperglycemia in diabetic animals causes oxidative stress and disturbs the function of the antioxidant system. Diabetic encephalopathy, nowadays termed as “diabetes-associated cognitive decline” (DACD), is a serious brain disorder that is frequently observed in diabetic patients. Cognitive deficits and neuronal cell loss are common features of this disease. Hyperglycemia and oxidative stress are two important contributory factors in the development of diabetic encephalopathy. In the diabetic brain, hyperglycemia induces mitochondrial dysfunction and exaggerates oxidative damage. Oxidative stress produced as a result of excessive production of ROS in the diabetic brain mediates neuronal cell death and cognitive deficits [89–92].
Recent research has provided evidence that oxidative stress disturbs cellular proteins, nucleic acids, and lipids, which eventually interfered with the normal functions of the cell [93]. Oxidative stress activates an important stress kinase pathway known as the JNK pathway [94]. The JNK pathway is an important signaling pathway that plays a central role in the development of several neurodegenerative diseases such as AD and Parkinson's disease [95]. A large number of recent studies provide compelling evidence that JNK activates downstream NF-κB signaling, which further activates the neuroinflammatory cascade and results in neuroinflammation [96]. Furthermore, NF-κB has been shown to increase the activities of the BACE enzyme, which abnormally cleaves Aβ proteins and promotes the formation of amyloid-beta plaques [97, 98] (Figure 4).
Figure 4.
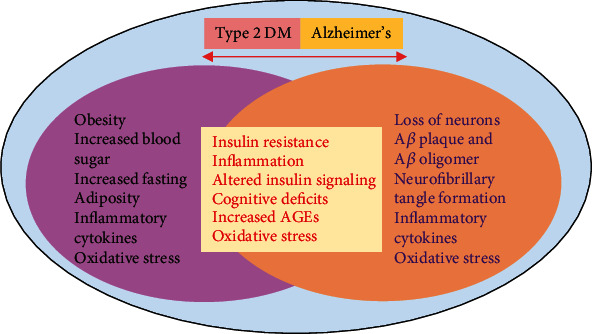
Shared links between Alzheimer's disease and diabetes. A simple illustration showing the shared links between Alzheimer's disease and diabetes.
Recent studies have shown that insulin resistance (IR) plays a crucial role in the progression of T2D pathology. No or very little response of a specific organ to the circulatory insulin is called IR. Loss of insulin binding receptor activities, dysregulation of the insulin receptor, and insulin receptor substrates (IRS-1 and IRS-2) are associated with an increased incidence of T2D pathology. Growing evidence indicates that peripheral IR disturbs brain insulin signaling [85]. Emerging studies have shown that increased circulatory insulin levels decrease the level of endothelial insulin receptors, impair the function of the blood-brain barrier (BBB), and decrease BBB insulin permeability [99].
A serine/threonine-specific protein kinase known as Akt is downregulated in IR. Akt facilitates glucose metabolism and inhibits glycogen synthase kinase 3 beta (GSK3β). GSK3β is an important kinase that phosphorylates tau protein. The overactivation of GSK3β in IR mediates the increased phosphorylation of tau protein, which later initiates the formation of neurofibrillary tangles. Moreover, another important enzyme called insulin-degrading enzyme (IDE) is dysregulated during IR. This enzyme usually prevents the accumulation of Aβ peptide in the brain via the degradation process [100, 101]. Several other pathways that have been observed in patients with AD and diabetes are inflammatory, oxidative stress, and protein misfolding signaling pathways. Numerous studies have reported that AGEs accumulate in the brain of a diabetic person. Recent evidence has demonstrated that dyslipidemia and hyperglycemia result in glucolipotoxicity, which plays a key role in the development of diabetes complications. Hyperglycemia also induces the production and release of AGEs, which act as an important link between diabetes and AD. The two known AGEs, pentosidine and glyceraldehyde-derived pyridinium (GLAP), have been identified to induce cognitive dysfunction and increase BACE1 activity. BACE1 is an enzyme that triggers the generation of Aβ by the activation of NF-κB. The receptor for AGEs is RAGE, and these receptors also function for Aβ. Increased expression of RAGE has been observed in Aβ-treated cultured microglia. Other cells of the brain, such as neurons, astrocytes, and microvascular cells, also show an elevated level of RAGE during the Aβ burden and AD pathology. The levels of both RAGE and AGEs increased during the progression of AD pathology [19, 102–115]. A previous study showed that diabetes induced via streptozotocin elevates the level of RAGE, increases senile plaque formation, and enhances AD pathology in transgenic mice. Elevated levels of microglial RAGE have been found in AD transgenic mice, and this causes an increase in the production of proinflammatory cytokines. The elevated levels of proinflammatory cytokines in turn lead to an increase in Aβ plaque formation, tau hyperphosphorylation, and cognitive impairments [116–118] (Figure 5).
Figure 5.
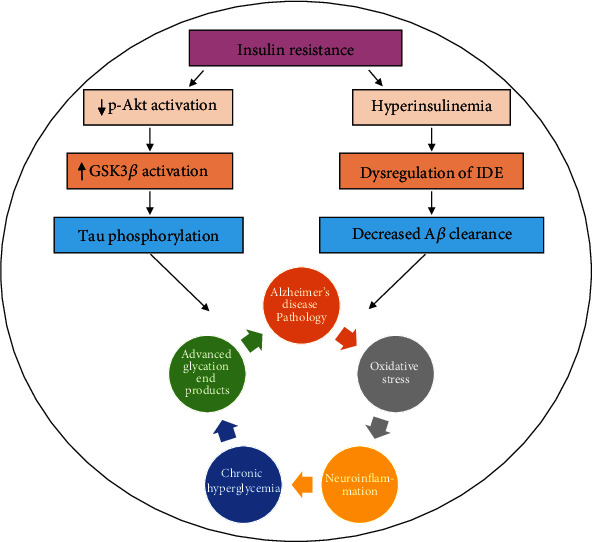
Insulin resistance, oxidative stress, and Aβ pathology. Downregulation of Akt and perturbation of the activities of the IDE lead to aggregation of amyloid-beta plaques and hyperphosphorylation of tau protein. On the other hand, chronic hyperglycemia directly promotes oxidative stress and neuroinflammation and causes neurodegeneration. Aβ = amyloid-beta; IDE = insulin-degrading enzyme; GSK3β = glycogen synthase kinase 3 beta.
4. Beneficial Role of Anthocyanins against T2D-Mediated AD Pathology (Figure 6)
Figure 6.
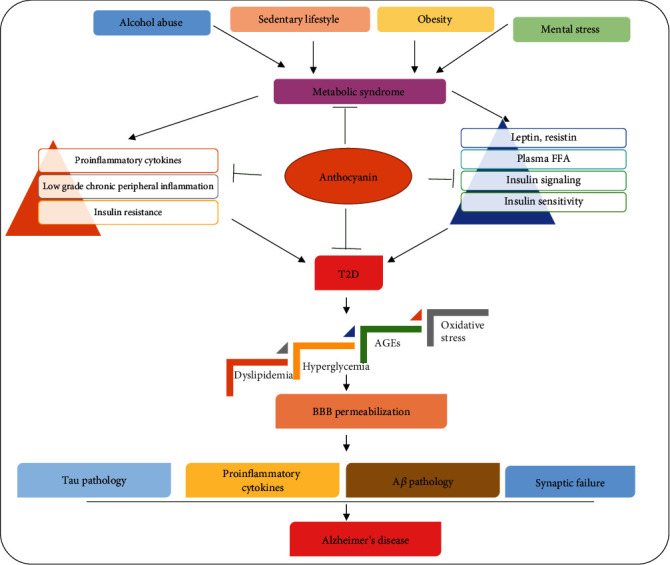
The effects of sedentary lifestyle, obesity, alcohol consumption, and mental distress on insulin sensitivity, induction of inflammation, release of free fatty acids, oxidative stress, hyperglycemia, neuronal insulin resistance, and AD pathology. T2DM = type 2 diabetes mellitus; Aβ = amyloid-beta; FFA = free fatty acid; BBB = blood-brain barrier.
Adipose tissue hormones such as resistin, adiponectin, and leptin have regulatory effects on body metabolism. Several recent studies have reported that dysregulation or abnormal levels of these hormones (also known as adipokines) can cause metabolic disorders such as T2D. Increased levels of leptin and resistin in obese individuals have been shown to be responsible for the development of IR. Several studies have suggested that insulin may reduce T2D by different mechanisms. Hyperglycemia-induced ROS generation has been suggested to be a critical feature in the progression and pathogenesis of DM, since oxidative stress is an important factor responsible for the progression of DM [119]. An imbalance in the production of ROS and dysregulation in the antioxidant defense system are major factors in the pathophysiology of DM-associated disorders. Therefore, regulation of oxidative stress either by boosting the endogenous antioxidant mechanism [15] or by inhibiting oxidative stress may provide benefits against DM [120] and other neurodegenerative diseases [96, 121]. Previous studies have suggested that treatment of pancreatic β-cells with anthocyanins obtained from wild Chinese blueberries markedly reduced the ROS and upregulated the endogenous antioxidant defense mechanism, thereby regulating the hyperglycemia-induced glucolipotoxicity [122]. Another study conducted on streptozotocin-induced DM suggested that anthocyanins can potentially rescue DM-associated oxidative stress, since anthocyanins reduce the levels of oxidative stress-related markers such as malondialdehyde and restore superoxide dismutase and catalase activity in diabetic rats [123]. Studies conducted on rats showed that grape-bilberry juice rich in anthocyanins could reduce the levels of resistin and leptin. Additionally, grape-bilberry juice was shown to decrease the levels of SFAs and increase the proportion of polyunsaturated fatty acids in the rat plasma. In conclusion, the authors suggested that anthocyanins could largely prevent metabolic diseases such as T2D and IR [124, 125].
Increasing the level of beneficial bacteria and regulating gut dysbiosis is another approach that may tackle DM-associated complications, since beneficial bacteria in the gut not only participate in the synthesis of several vitamins and amino acids but also ferment complex carbohydrates to produce short-chain fatty acids (SCFAs). Several studies have indicated that anthocyanins could increase the levels of beneficial bacterial species such as Bifidobacterium and Lactobacillus-Enterococcus [126–128]. A wealth of emerging studies in recent years indicated that abnormal glucose metabolism produces oxidative stress in body cells, which plays a pivotal role in the development of T2D. Several vitamins such as vitamin B complex, vitamin C, and vitamin K have antioxidant properties that abrogate oxidative stress and regulate glucose metabolism. Furthermore, these vitamins also reduce the risk of T2D in prediabetic individuals [129–133].
Accumulating recent evidence has highlighted that hyperglycemia and IR are two important features of T2D and that hyperglycemia mediates neuroinflammation and induces oxidative stress. Increased oxidative stress activates several neuronal apoptotic pathways and causes neurodegeneration. Furthermore, increased oxidative stress and neuroinflammation are involved in the pathogenesis of AD. Anthocyanins not only stimulate glucose metabolism but also reduce hyperglycemia and thus prevent diabetes-induced neuroinflammation and AD pathology [134–144].
Insulin resistance causes hyperinsulinemia in the body, which in turn leads to dysregulation of IDEs, which are important enzymes involved in the clearance of Aβ proteins. Dysregulation of IDEs causes an Aβ burden in the brain, and excessive accumulation of Aβ proteins induces neurodegeneration and accelerates AD pathology. Emerging evidence suggests that the daily consumption of the natural dietary polyflavonoid anthocyanin increases insulin sensitivity, regulates insulin signaling, and prevents insulin resistance-mediated AD pathology. Moreover, anthocyanin enhances the function of insulin-secreting pancreatic β-cells and reduces the risk of diabetes-induced AD pathology [122, 145–150]. The LPS secreted by gram-negative bacteria during gut dysbiosis has been shown to cause systemic low-grade inflammation, pancreatic cell dysfunction, IR, and high levels of cytokines in the blood. Circulatory cytokines and LPS affect the function of peripheral organs and enter the brain via the BBB, causing neuroinflammation, neuronal cell death, and Aβ pathology. A recent report has shown that anthocyanins have strong antioxidant and anti-inflammatory properties. Daily intake of anthocyanins improves the growth of beneficial bacteria in the gut and prevents the induction of peripheral as well as central nervous system (CNS) inflammation. Additionally, anthocyanins can also reduce the prevalence of T2D and its related complications. Several recent studies have suggested that anthocyanins could prevent diabetic neuropathy, brain IR, and AD pathology [151–164].
5. Conclusions and Future Perspectives
Type 2 diabetes is one of the major health-related challenges worldwide. Excessive consumption of fast foods and sedentary lifestyles are the most common causes of T2D. Lack of physical activity influences the hormone levels and induces obesity, which may further aggravate the progression of DM and its associated pathological consequences via several mechanisms. On the other hand, contagious diseases and contaminated foods increase the usage of broad-spectrum antibiotics, and prolonged usage of these drugs induces gut dysbiosis and reduces the abundance of beneficial bacteria, which are largely involved in the synthesis of multivitamins in the body. These multivitamins protect the body from oxidative stress-mediated metabolic diseases such as diabetes, abnormal weight gain, impaired insulin signaling, and AD-like pathological consequences. Various phytonutrients have drawn much attention in the management of DM-associated disorders such as neurodegeneration and neuroinflammation [96, 165]. One such phytonutrient is anthocyanin, a polyflavonoid that shows protective effects via different mechanisms, especially by reducing oxidative stress and boosting the endogenous antioxidant system [22]. Patients with diabetes are at a high risk of developing AD and dementia. This comprehensive study suggests that the daily consumption of natural dietary anthocyanins obtained from fruits, vegetables, and beans protects our bodies from various metabolic and neurological disorders. We believe that continuous research and detailed analyses will further elucidate the “dark side,” i.e., the basic molecular mechanisms underlying the effects of anthocyanins in diabetes and metabolic disease-mediated AD pathology, and diabetes patients will soon “wind down with red wine, pop in the purple potatoes, grab the grapes, and revel with radishes.”
Acknowledgments
This research was supported by the Neurological Disorder Research Program of the National Research Foundation (NRF) funded by the Korea Government (MSIT) (2020M3E5D9080660).
Abbreviations
- GLAP:
Glyceraldehyde-derived pyridinium
- AD:
Alzheimer's disease
- CNS:
Central nervous system
- IL-1β:
Interleukin-1β
- LPS:
Lipopolysaccharide
- MDA:
Malondialdehyde
- p-JNK:
Phospho-c-Jun N-terminal kinase 1
- T2D:
Type 2 diabetes
- AGEs:
Advanced glycation end products
- FFA:
Free fatty acid
- TNF-α:
Tumor necrosis factor-α
- ROS:
Reactive oxygen species
- E. coli:
Escherichia coli
- TLR4:
Toll-like receptor 4
- NF-κB:
Nuclear factor kappa-light-chain-enhancer of activated B cells
- GIT:
Gastrointestinal tract
- OSA:
Obstructive sleep apnea
- DACD:
Diabetes-associated cognitive decline
- IR:
Insulin resistance
- IRS:
Insulin receptor substrates
- BBB:
Blood-brain barrier
- SCFAs:
Short-chain fatty acids.
Data Availability
The datasets used and/or analyzed during the current study are available from the corresponding author upon request.
Conflicts of Interest
The authors declared no competing financial interests.
Authors' Contributions
Muhammad Sohail Khan, Muhammad Ikram, and Tae Ju Park designed, managed, and wrote the manuscript. Myeong Ok Kim is a corresponding author who provided all the technical support, reviewed the manuscript, and holds all the responsibilities related to this manuscript.
References
- 1.Fang J. Y., Lin C.-H., Huang T.-H., Chuang S.-Y. In vivo rodent models of type 2 diabetes and their usefulness for evaluating flavonoid bioactivity. Nutrients. 2019;11(3):p. 530. doi: 10.3390/nu11030530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Flegal K. M., Carroll M. D., Ogden C. L., Curtin L. R. Prevalence and trends in obesity among US adults, 1999-2008. JAMA. 2010;303(3):235–241. doi: 10.1001/jama.2009.2014. [DOI] [PubMed] [Google Scholar]
- 3.Forrest K. Y. Z., Stuhldreher W. L. Prevalence and correlates of vitamin D deficiency in US adults. Nutrition Research. 2011;31(1):48–54. doi: 10.1016/j.nutres.2010.12.001. [DOI] [PubMed] [Google Scholar]
- 4.Devereux R. B., Alderman M. H. Role of preclinical cardiovascular disease in the evolution from risk factor exposure to development of morbid events. Circulation. 1993;88(4):1444–1455. doi: 10.1161/01.CIR.88.4.1444. [DOI] [PubMed] [Google Scholar]
- 5.Boden G. Obesity and free fatty acids. Endocrinology and Metabolism Clinics of North America. 2008;37(3):635–646. doi: 10.1016/j.ecl.2008.06.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Santomauro A. T., Boden G., Silva M. E., et al. Overnight lowering of free fatty acids with acipimox improves insulin resistance and glucose tolerance in obese diabetic and nondiabetic subjects. Diabetes. 1999;48(9):1836–1841. doi: 10.2337/diabetes.48.9.1836. [DOI] [PubMed] [Google Scholar]
- 7.Reaven G. M., Hollenbeck C., Jeng C. Y., Wu M. S., Chen Y. D. I. Measurement of plasma glucose, free fatty acid, lactate, and insulin for 24 h in patients with NIDDM. Diabetes. 1988;37(8):1020–1024. doi: 10.2337/diab.37.8.1020. [DOI] [PubMed] [Google Scholar]
- 8.Kershaw E. E., Flier J. S. Adipose tissue as an endocrine organ. The Journal of Clinical Endocrinology and Metabolism. 2004;89(6):2548–2556. doi: 10.1210/jc.2004-0395. [DOI] [PubMed] [Google Scholar]
- 9.Jensen M. D., Haymond M. W., Rizza R. A., Cryer P. E., Miles J. M. Influence of body fat distribution on free fatty acid metabolism in obesity. The Journal of Clinical Investigation. 1989;83(4):1168–1173. doi: 10.1172/JCI113997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Boden G., Chen X., Ruiz J., White J. V., Rossetti L. Mechanisms of fatty acid-induced inhibition of glucose uptake. The Journal of Clinical Investigation. 1994;93(6):2438–2446. doi: 10.1172/JCI117252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bjorntorp P., Bergman H., Varnauskas E. Plasma free fatty acid turnover rate in obesity. Acta Medica Scandinavica. 1969;185(4):351–356. doi: 10.1111/j.0954-6820.1969.tb07347.x. [DOI] [PubMed] [Google Scholar]
- 12.Sharma R. B., Alonso L. C. Lipotoxicity in the pancreatic beta cell: not just survival and function, but proliferation as well? Current Diabetes Reports. 2014;14(6):p. 492. doi: 10.1007/s11892-014-0492-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Oh Y. S., Bae G. D., Baek D. J., Park E.-Y., Jun H.-S. Fatty acid-induced lipotoxicity in pancreatic beta-cells during development of type 2 diabetes. Frontiers in Endocrinology. 2018;9:p. 384. doi: 10.3389/fendo.2018.00384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Uribarri J., Cai W., Peppa M., et al. Circulating glycotoxins and dietary advanced glycation endproducts: two links to inflammatory response, oxidative stress, and aging. The Journals of Gerontology Series A: Biological Sciences and Medical Sciences. 2007;62(4):427–433. doi: 10.1093/gerona/62.4.427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Badshah H., Ikram M., Ali W., Ahmad S., Hahm J. R., Kim M. O. Caffeine may abrogate LPS-induced oxidative stress and neuroinflammation by regulating Nrf2/TLR4 in adult mouse brains. Biomolecules. 2019;9(11):p. 719. doi: 10.3390/biom9110719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Banerjee M., Saxena M. Genetic polymorphisms of cytokine genes in type 2 diabetes mellitus. World Journal of Diabetes. 2014;5(4):493–504. doi: 10.4239/wjd.v5.i4.493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ikram M., Saeed K., Khan A., et al. Natural dietary supplementation of curcumin protects mice brains against ethanol-induced oxidative stress-mediated neurodegeneration and memory impairment via Nrf2/TLR4/RAGE signaling. Nutrients. 2019;11(5, article 1082) doi: 10.3390/nu11051082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Eckardt K., Görgens S. W., Raschke S., Eckel J. Myokines in insulin resistance and type 2 diabetes. Diabetologia. 2014;57(6):1087–1099. doi: 10.1007/s00125-014-3224-x. [DOI] [PubMed] [Google Scholar]
- 19.Akter K., Lanza E. A., Martin S. A., Myronyuk N., Rua M., Raffa R. B. Diabetes mellitus and Alzheimer’s disease: shared pathology and treatment? British Journal of Clinical Pharmacology. 2011;71(3):365–376. doi: 10.1111/j.1365-2125.2010.03830.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Smeriglio A., Barreca D., Bellocco E., Trombetta D. Chemistry, pharmacology and health benefits of anthocyanins. Phytotherapy Research. 2016;30(8):1265–1286. doi: 10.1002/ptr.5642. [DOI] [PubMed] [Google Scholar]
- 21.Mazza G. J. Anthocyanins and heart health. Annali dell’Istituto Superiore di Sanità. 2007;43(4):369–374. [PubMed] [Google Scholar]
- 22.Ali T., Kim T., Rehman S. U., et al. Natural dietary supplementation of anthocyanins via PI3K/Akt/Nrf2/HO-1 pathways mitigate oxidative stress, neurodegeneration, and memory impairment in a mouse model of Alzheimer’s disease. Molecular Neurobiology. 2018;55(7):6076–6093. doi: 10.1007/s12035-017-0798-6. [DOI] [PubMed] [Google Scholar]
- 23.Pan P., Oshima K., Huang Y. W., et al. Gut bacteria are required for the benefits of black raspberries in ApcMin/+ mice. Journal of Berry Research. 2018;8(4):239–249. doi: 10.3233/JBR-180337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Woodward G., Kroon P., Cassidy A., Kay C. Anthocyanin stability and recovery: implications for the analysis of clinical and experimental samples. Journal of Agricultural and Food Chemistry. 2009;57(12):5271–5278. doi: 10.1021/jf900602b. [DOI] [PubMed] [Google Scholar]
- 25.Welch C. R., Wu Q., Simon J. E. Recent advances in anthocyanin analysis and characterization. Current Analytical Chemistry. 2008;4(2):75–101. doi: 10.2174/157341108784587795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wallace T. C., Giusti M. M. Anthocyanins. Advances in Nutrition. 2015;6(5):620–622. doi: 10.3945/an.115.009233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Tedesco I., Carbone V., Spagnuolo C., Minasi P., Russo G. L. Identification and quantification of flavonoids from two Southern Italian cultivars of Allium cepa L., Tropea (red onion) and Montoro (copper onion), and their capacity to protect human erythrocytes from oxidative stress. Journal of Agricultural and Food Chemistry. 2015;63(21):5229–5238. doi: 10.1021/acs.jafc.5b01206. [DOI] [PubMed] [Google Scholar]
- 28.Samadi A. K., Bilsland A., Georgakilas A. G., et al. A multi-targeted approach to suppress tumor-promoting inflammation. Seminars in Cancer Biology. 2015;35:S151–S184. doi: 10.1016/j.semcancer.2015.03.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Morais C. A., de Rosso V. V., Estadella D., Pisani L. P. Anthocyanins as inflammatory modulators and the role of the gut microbiota. The Journal of Nutritional Biochemistry. 2016;33:1–7. doi: 10.1016/j.jnutbio.2015.11.008. [DOI] [PubMed] [Google Scholar]
- 30.McGhie T. K., Walton M. C. The bioavailability and absorption of anthocyanins: towards a better understanding. Molecular Nutrition & Food Research. 2007;51(6):702–713. doi: 10.1002/mnfr.200700092. [DOI] [PubMed] [Google Scholar]
- 31.Khoo H. E., Azlan A., Tang S. T., Lim S. M. Anthocyanidins and anthocyanins: colored pigments as food, pharmaceutical ingredients, and the potential health benefits. Food & Nutrition Research. 2017;61(1):p. 1361779. doi: 10.1080/16546628.2017.1361779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Junker L. V., Ensminger I. Relationship between leaf optical properties, chlorophyll fluorescence and pigment changes in senescing Acer saccharum leaves. Tree Physiology. 2016;36(6):694–711. doi: 10.1093/treephys/tpv148. [DOI] [PubMed] [Google Scholar]
- 33.He J., Giusti M. M. Anthocyanins: natural colorants with health-promoting properties. Annual Review of Food Science and Technology. 2010;1(1):163–187. doi: 10.1146/annurev.food.080708.100754. [DOI] [PubMed] [Google Scholar]
- 34.Ding S., Jiang H., Fang J. Regulation of immune function by polyphenols. Journal of Immunology Research. 2018;2018:8. doi: 10.1155/2018/1264074.1264074 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Lan X., Wang W., Li Q., Wang J. The natural flavonoid pinocembrin: molecular targets and potential therapeutic applications. Molecular Neurobiology. 2016;53(3):1794–1801. doi: 10.1007/s12035-015-9125-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Shih P. H., Yeh C. T., Yen G. C. Anthocyanins induce the activation of phase II enzymes through the antioxidant response element pathway against oxidative stress-induced apoptosis. Journal of Agricultural and Food Chemistry. 2007;55(23):9427–9435. doi: 10.1021/jf071933i. [DOI] [PubMed] [Google Scholar]
- 37.Panche A. N., Diwan A. D., Chandra S. R. Flavonoids: an overview. Canadian Journal of Biotechnology. 2016;5, article e47 doi: 10.1017/jns.2016.41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Chait A., den Hartigh L. J. Adipose tissue distribution, inflammation and its metabolic consequences, including diabetes and cardiovascular disease. Frontiers in Cardiovascular Medicine. 2020;7:p. 22. doi: 10.3389/fcvm.2020.00022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Creely S. J., PG M. T., Kusminski C. M., et al. Lipopolysaccharide activates an innate immune system response in human adipose tissue in obesity and type 2 diabetes. American Journal of Physiology-Endocrinology and Metabolism. 2007;292(3):E740–E747. doi: 10.1152/ajpendo.00302.2006. [DOI] [PubMed] [Google Scholar]
- 40.Erridge C., Attina T., Spickett C. M., Webb D. J. A high-fat meal induces low-grade endotoxemia: evidence of a novel mechanism of postprandial inflammation. The American Journal of Clinical Nutrition. 2007;86(5):1286–1292. doi: 10.1093/ajcn/86.5.1286. [DOI] [PubMed] [Google Scholar]
- 41.Khan M. S., Ikram M., Park J. S., Park T. J., Kim M. O. Gut microbiota, its role in induction of Alzheimer’s disease pathology, and possible therapeutic interventions: special focus on anthocyanins. Cells. 2020;9(4):p. 853. doi: 10.3390/cells9040853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Naseer M., Bibi F., Alqahtani M., et al. Role of gut microbiota in obesity, type 2 diabetes and Alzheimer’s disease. CNS & Neurological Disorders Drug Targets. 2014;13(2):305–311. doi: 10.2174/18715273113126660147. [DOI] [PubMed] [Google Scholar]
- 43.Palsson-McDermott E. M., O'Neill L. A. Signal transduction by the lipopolysaccharide receptor, Toll-like receptor-4. Immunology. 2004;113(2):153–162. doi: 10.1111/j.1365-2567.2004.01976.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Ali W., Ikram M., Park H. Y., et al. Oral administration of alpha linoleic acid rescues Aβ-induced glia-mediated neuroinflammation and cognitive dysfunction in C57BL/6N mice. Cells. 2020;9(3):p. 667. doi: 10.3390/cells9030667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Cheng J., Xue F., Zhang M., et al. TRIM31 deficiency is associated with impaired glucose metabolism and disrupted gut microbiota in mice. Frontiers in Physiology. 2018;9:p. 24. doi: 10.3389/fphys.2018.00024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Cani P. D., Amar J., Iglesias M. A., et al. Metabolic endotoxemia initiates obesity and insulin resistance. Diabetes. 2007;56(7):1761–1772. doi: 10.2337/db06-1491. [DOI] [PubMed] [Google Scholar]
- 47.Hu F. B., Manson J. A. E., Stampfer M. J., et al. Diet, lifestyle, and the risk of type 2 diabetes mellitus in women. The New England Journal of Medicine. 2001;345(11):790–797. doi: 10.1056/NEJMoa010492. [DOI] [PubMed] [Google Scholar]
- 48.Manson J. E., Ajani U. A., Liu S., Nathan D. M., Hennekens C. H. A prospective study of cigarette smoking and the incidence of diabetes mellitus among US male physicians. The American Journal of Medicine. 2000;109(7):538–542. doi: 10.1016/S0002-9343(00)00568-4. [DOI] [PubMed] [Google Scholar]
- 49.Cullmann M., Hilding A., Ostenson C. G. Alcohol consumption and risk of pre-diabetes and type 2 diabetes development in a Swedish population. Diabetic Medicine. 2012;29(4):441–452. doi: 10.1111/j.1464-5491.2011.03450.x. [DOI] [PubMed] [Google Scholar]
- 50.Belkina A. C., Denis G. V. Obesity genes and insulin resistance. Current Opinion in Endocrinology, Diabetes, and Obesity. 2010;17(5):472–477. doi: 10.1097/MED.0b013e32833c5c48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Pamidi S., Tasali E. Obstructive sleep apnea and type 2 diabetes: is there a link? Frontiers in Neurology. 2012;3:p. 126. doi: 10.3389/fneur.2012.00126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Ioja S., Weir I. D., Rennert N. J. Relationship between sleep disorders and the risk for developing type 2 diabetes mellitus. Postgraduate Medicine. 2015;124(4):119–129. doi: 10.3810/pgm.2012.07.2573. [DOI] [PubMed] [Google Scholar]
- 53.Lindberg E., Theorell-Haglöw J., Svensson M., Gislason T., Berne C., Janson C. Sleep apnea and glucose metabolism: a long-term follow-up in a community-based sample. Chest. 2012;142(4):935–942. doi: 10.1378/chest.11-1844. [DOI] [PubMed] [Google Scholar]
- 54.Einhorn D., Stewart D. A., Erman M. K., Gordon N., Philis-Tsimikas A., Casal E. Prevalence of sleep apnea in a population of adults with type 2 diabetes mellitus. Endocrine Practice. 2007;13(4):355–362. doi: 10.4158/EP.13.4.355. [DOI] [PubMed] [Google Scholar]
- 55.Schober A. K., Neurath M. F., Harsch I. A. Prevalence of sleep apnoea in diabetic patients. The Clinical Respiratory Journal. 2011;5(3):165–172. doi: 10.1111/j.1752-699X.2010.00216.x. [DOI] [PubMed] [Google Scholar]
- 56.Simopoulos A. P. An increase in the omega-6/omega-3 fatty acid ratio increases the risk for obesity. Nutrients. 2016;8(3):p. 128. doi: 10.3390/nu8030128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Macdonald I. A. A review of recent evidence relating to sugars, insulin resistance and diabetes. European Journal of Nutrition. 2016;55(Supplement 2):17–23. doi: 10.1007/s00394-016-1340-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Maki K. C., Phillips A. K. Dietary substitutions for refined carbohydrate that show promise for reducing risk of type 2 diabetes in men and women. The Journal of Nutrition. 2015;145(1):159S–163S. doi: 10.3945/jn.114.195149. [DOI] [PubMed] [Google Scholar]
- 59.Fritsche K. L. Linoleic acid, vegetable oils & inflammation. Missouri Medicine. 2014;111(1):41–43. [PMC free article] [PubMed] [Google Scholar]
- 60.Bordoni A., Danesi F., Dardevet D., et al. Dairy products and inflammation: a review of the clinical evidence. Critical Reviews in Food Science and Nutrition. 2017;57(12):2497–2525. doi: 10.1080/10408398.2014.967385. [DOI] [PubMed] [Google Scholar]
- 61.Basaranoglu M., Basaranoglu G., Bugianesi E. Carbohydrate intake and nonalcoholic fatty liver disease: fructose as a weapon of mass destruction. Hepatobiliary Surg Nutr. 2015;4(2):109–116. doi: 10.3978/j.issn.2304-3881.2014.11.05. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Silva Figueiredo P., Carla Inada A., Marcelino G., et al. Fatty acids consumption: the role metabolic aspects involved in obesity and its associated disorders. Nutrients. 2017;9(10):p. 1158. doi: 10.3390/nu9101158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Vlassara H., Uribarri J. Advanced glycation end products (AGE) and diabetes: cause, effect, or both? Current Diabetes Reports. 2014;14(1):p. 453. doi: 10.1007/s11892-013-0453-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Johnson R. J., Nakagawa T., Sanchez-Lozada L. G., et al. Sugar, uric acid, and the etiology of diabetes and obesity. Diabetes. 2013;62(10):3307–3315. doi: 10.2337/db12-1814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Lustig R. H. Fructose and nonalcoholic fatty liver disease. Journal of the California Dental Association. 2016;44(10):613–617. [PubMed] [Google Scholar]
- 66.Kizaki Z., Sawada T. Fructose. Nihon Rinsho. 1995;53, Su Part 1:536–539. [PubMed] [Google Scholar]
- 67.Singh V. P., Bali A., Singh N., Jaggi A. S. Advanced glycation end products and diabetic complications. The Korean Journal of Physiology & Pharmacology. 2014;18(1):1–14. doi: 10.4196/kjpp.2014.18.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Liu W., Mao Y., Schoenborn J., Wang Z., Tang G., Tang X. Whole blueberry protects pancreatic beta-cells in diet-induced obese mouse. Nutrition & Metabolism. 2019;16(1):p. 34. doi: 10.1186/s12986-019-0363-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Iglesias-Barreira V., Ahn M. T., Reusens B., Dahri S., Hoet J. J., Remacle C. Pre- and postnatal low protein diet affect pancreatic islet blood flow and insulin release in adult rats. Endocrinology. 1996;137(9):3797–3801. doi: 10.1210/endo.137.9.8756549. [DOI] [PubMed] [Google Scholar]
- 70.Theys N., Clippe A., Bouckenooghe T., Reusens B., Remacle C. Early low protein diet aggravates unbalance between antioxidant enzymes leading to islet dysfunction. PLoS One. 2009;4(7, article e6110) doi: 10.1371/journal.pone.0006110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Verberne A. J. M., Korim W. S., Sabetghadam A., Llewellyn‐Smith I. J. Adrenaline: insights into its metabolic roles in hypoglycaemia and diabetes. British Journal of Pharmacology. 2016;173(9):1425–1437. doi: 10.1111/bph.13458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Seematter G., Binnert C., Tappy L. Stress and metabolism. Metabolic Syndrome and Related Disorders. 2005;3(1):8–13. doi: 10.1089/met.2005.3.8. [DOI] [PubMed] [Google Scholar]
- 73.Dinan T. G. Stress and the genesis of diabetes mellitus in schizophrenia. The British Journal of Psychiatry Supplement. 2004;47:S72–S75. doi: 10.1192/bjp.184.47.s72. [DOI] [PubMed] [Google Scholar]
- 74.Yeung W. C. G., Rawlinson W. D., Craig M. E. Enterovirus infection and type 1 diabetes mellitus: systematic review and meta-analysis of observational molecular studies. BMJ. 2011;342(1, article d35) doi: 10.1136/bmj.d35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Shaheen Z. R., Stafford J. D., Voss M. G., Oleson B. J., Stancill J. S., Corbett J. A. The location of sensing determines the pancreatic β-cell response to the viral mimetic dsRNA. The Journal of Biological Chemistry. 2020;295(8):2385–2397. doi: 10.1074/jbc.RA119.010267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Yoon J. W. The role of viruses and environmental factors in the induction of diabetes. Current Topics in Microbiology and Immunology. 1990;164:95–123. doi: 10.1007/978-3-642-75741-9_6. [DOI] [PubMed] [Google Scholar]
- 77.Horwitz M. S., Ilic A., Fine C., Rodriguez E., Sarvetnick N. Presented antigen from damaged pancreatic β cells activates autoreactive T cells in virus-mediated autoimmune diabetes. The Journal of Clinical Investigation. 2002;109(1):79–87. doi: 10.1172/JCI0211198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Ottum M. S., Mistry A. M. Advanced glycation end-products: modifiable environmental factors profoundly mediate insulin resistance. Journal of Clinical Biochemistry and Nutrition. 2015;57(1):1–12. doi: 10.3164/jcbn.15-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Tosur M., Viau-Colindres J., Astudillo M., Redondo M. J., Lyons S. K. Medication-induced hyperglycemia: pediatric perspective. BMJ Open Diabetes Research & Care. 2020;8(1, article e000801) doi: 10.1136/bmjdrc-2019-000801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Goyal S. N., Reddy N. M., Patil K. R., et al. Challenges and issues with streptozotocin-induced diabetes - a clinically relevant animal model to understand the diabetes pathogenesis and evaluate therapeutics. Chemico-Biological Interactions. 2016;244:49–63. doi: 10.1016/j.cbi.2015.11.032. [DOI] [PubMed] [Google Scholar]
- 81.Qian X., Lin L., Zong Y., et al. Shifts in renin-angiotensin system components, angiogenesis, and oxidative stress-related protein expression in the lamina cribrosa region of streptozotocin-induced diabetic mice. Graefe’s Archive for Clinical and Experimental Ophthalmology. 2018;256(3):525–534. doi: 10.1007/s00417-017-3866-8. [DOI] [PubMed] [Google Scholar]
- 82.Uribarri J., Woodruff S., Goodman S., et al. Advanced glycation end products in foods and a practical guide to their reduction in the diet. Journal of the American Dietetic Association. 2010;110(6):911–916.e12. doi: 10.1016/j.jada.2010.03.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Bettiga A., Fiorio F., Di Marco F., et al. The modern western diet rich in advanced glycation end-products (AGEs): an overview of its impact on obesity and early progression of renal pathology. Nutrients. 2019;11(8, article 1748) doi: 10.3390/nu11081748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Vistoli G., de Maddis D., Cipak A., Zarkovic N., Carini M., Aldini G. Advanced glycoxidation and lipoxidation end products (AGEs and ALEs): an overview of their mechanisms of formation. Free Radical Research. 2013;47(Supplement 1):3–27. doi: 10.3109/10715762.2013.815348. [DOI] [PubMed] [Google Scholar]
- 85.Shah S. A., Yoon G. H., Chung S. S., et al. Novel osmotin inhibits SREBP2 via the AdipoR1/AMPK/SIRT1 pathway to improve Alzheimer’s disease neuropathological deficits. Molecular Psychiatry. 2017;22(3):407–416. doi: 10.1038/mp.2016.23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Kubis-Kubiak A., Dyba A., Piwowar A. The interplay between diabetes and Alzheimer’s disease-in the hunt for biomarkers. International Journal of Molecular Sciences. 2020;21(8, article 2744) doi: 10.3390/ijms21082744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Neth B. J., Craft S. Insulin resistance and Alzheimer’s disease: bioenergetic linkages. Frontiers in Aging Neuroscience. 2017;9:p. 345. doi: 10.3389/fnagi.2017.00345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Massaad C. A., Klann E. Reactive oxygen species in the regulation of synaptic plasticity and memory. Antioxidants & Redox Signaling. 2011;14(10):2013–2054. doi: 10.1089/ars.2010.3208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Kamboj S. S., Sandhir R. Protective effect of N-acetylcysteine supplementation on mitochondrial oxidative stress and mitochondrial enzymes in cerebral cortex of streptozotocin-treated diabetic rats. Mitochondrion. 2011;11(1):214–222. doi: 10.1016/j.mito.2010.09.014. [DOI] [PubMed] [Google Scholar]
- 90.Hong J. H., Kim M. J., Park M. R., et al. Effects of vitamin E on oxidative stress and membrane fluidity in brain of streptozotocin-induced diabetic rats. Clinica Chimica Acta. 2004;340(1-2):107–115. doi: 10.1016/j.cccn.2003.10.003. [DOI] [PubMed] [Google Scholar]
- 91.Volpe C. M. O., Villar-Delfino P. H., Dos Anjos P. M., Nogueira-Machado J. A. Cellular death, reactive oxygen species (ROS) and diabetic complications. Cell Death & Disease. 2018;9(2):p. 119. doi: 10.1038/s41419-017-0135-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Bonnefont-Rousselot D. Glucose and reactive oxygen species. Current Opinion in Clinical Nutrition and Metabolic Care. 2002;5(5):561–568. doi: 10.1097/00075197-200209000-00016. [DOI] [PubMed] [Google Scholar]
- 93.Ikram M., Park T. J., Ali T., Kim M. O. Antioxidant and neuroprotective effects of caffeine against Alzheimer’s and Parkinson’s disease: insight into the role of Nrf-2 and A2AR signaling. Antioxidants. 2020;9(9):p. 902. doi: 10.3390/antiox9090902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Khan M. S., Muhammad T., Ikram M., Kim M. O. Dietary supplementation of the antioxidant curcumin halts systemic LPS-induced neuroinflammation-associated neurodegeneration and memory/synaptic impairment via the JNK/NF-κB/Akt signaling pathway in adult rats. Oxidative Medicine and Cellular Longevity. 2019;2019:23. doi: 10.1155/2019/7860650.7860650 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Ullah R., Jo M. H., Riaz M., et al. Glycine, the smallest amino acid, confers neuroprotection against D-galactose-induced neurodegeneration and memory impairment by regulating c-Jun N-terminal kinase in the mouse brain. Journal of Neuroinflammation. 2020;17(1):1–21. doi: 10.1186/s12974-020-01989-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Ikram M., Muhammad T., Rehman S. U., et al. Hesperetin confers neuroprotection by regulating Nrf2/TLR4/NF-κB signaling in an Aβ mouse model. Molecular Neurobiology. 2019;56(9):6293–6309. doi: 10.1007/s12035-019-1512-7. [DOI] [PubMed] [Google Scholar]
- 97.Chen C. H., Zhou W., Liu S., et al. Increased NF-κB signalling up-regulates BACE1 expression and its therapeutic potential in Alzheimer’s disease. The International Journal of Neuropsychopharmacology. 2012;15(1):77–90. doi: 10.1017/S1461145711000149. [DOI] [PubMed] [Google Scholar]
- 98.Wu J., Mei C., Vlassara H., Striker G. E., Zheng F. Oxidative stress-induced JNK activation contributes to proinflammatory phenotype of aging diabetic mesangial cells. American Journal of Physiology-Renal Physiology. 2009;297(6):F1622–F1631. doi: 10.1152/ajprenal.00078.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Maciejczyk M., Zebrowska E., Chabowski A. Insulin resistance and oxidative stress in the brain: what’s new? International Journal of Molecular Sciences. 2019;20(4):p. 874. doi: 10.3390/ijms20040874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Qiu W. Q., Folstein M. F. Insulin, insulin-degrading enzyme and amyloid-β peptide in Alzheimer’s disease: review and hypothesis. Neurobiology of Aging. 2006;27(2):190–198. doi: 10.1016/j.neurobiolaging.2005.01.004. [DOI] [PubMed] [Google Scholar]
- 101.Haque R., Nazir A. Insulin-degrading enzyme: a link between Alzheimer’s and type 2 diabetes mellitus. CNS & Neurological Disorders Drug Targets. 2014;13(2):259–264. doi: 10.2174/18715273113126660139. [DOI] [PubMed] [Google Scholar]
- 102.Beeri M. S., Moshier E., Schmeidler J., et al. Serum concentration of an inflammatory glycotoxin, methylglyoxal, is associated with increased cognitive decline in elderly individuals. Mechanisms of Ageing and Development. 2011;132(11-12):583–587. doi: 10.1016/j.mad.2011.10.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Byun K., Bayarsaikhan E., Kim D., et al. Induction of neuronal death by microglial AGE-albumin: implications for Alzheimer’s disease. PLoS One. 2012;7(5, article e37917) doi: 10.1371/journal.pone.0037917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Cai W., Uribarri J., Zhu L., et al. Oral glycotoxins are a modifiable cause of dementia and the metabolic syndrome in mice and humans. Proceedings of the National Academy of Sciences of the United States of America. 2014;111(13):4940–4945. doi: 10.1073/pnas.1316013111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Choi B. R., Cho W. H., Kim J., et al. Increased expression of the receptor for advanced glycation end products in neurons and astrocytes in a triple transgenic mouse model of Alzheimer's disease. Experimental & Molecular Medicine. 2014;46(2, article e75) doi: 10.1038/emm.2013.147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Goh S. Y., Cooper M. E. Clinical review: the role of advanced glycation end products in progression and complications of diabetes. The Journal of Clinical Endocrinology and Metabolism. 2008;93(4):1143–1152. doi: 10.1210/jc.2007-1817. [DOI] [PubMed] [Google Scholar]
- 107.Guglielmotto M., Aragno M., Tamagno E., et al. AGEs/RAGE complex upregulates BACE1 via NF-κB pathway activation. Neurobiology Aging. 2012;33(1):196.e13–196.e27. doi: 10.1016/j.neurobiolaging.2010.05.026. [DOI] [PubMed] [Google Scholar]
- 108.Kojro E., Postina R. Regulated proteolysis of RAGE and AbetaPP as possible link between type 2 diabetes mellitus and Alzheimer’s disease. Journal of Alzheimer’s Disease. 2009;16(4):865–878. doi: 10.3233/JAD-2009-0998. [DOI] [PubMed] [Google Scholar]
- 109.Kroner Z. The relationship between Alzheimer’s disease and diabetes: type 3 diabetes? Alternative Medicine Review. 2009;14(4):373–379. [PubMed] [Google Scholar]
- 110.Liu L. P., Hong H., Liao J. M., et al. Upregulation of RAGE at the blood-brain barrier in streptozotocin-induced diabetic mice. Synapse. 2009;63(8):636–642. doi: 10.1002/syn.20644. [DOI] [PubMed] [Google Scholar]
- 111.Miller M. C., Tavares R., Johanson C. E., et al. Hippocampal RAGE immunoreactivity in early and advanced Alzheimer's disease. Brain Research. 2008;1230:273–280. doi: 10.1016/j.brainres.2008.06.124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Sasaki N., Toki S., Chowei H., et al. Immunohistochemical distribution of the receptor for advanced glycation end products in neurons and astrocytes in Alzheimer’s disease. Brain Research. 2001;888(2):256–262. doi: 10.1016/S0006-8993(00)03075-4. [DOI] [PubMed] [Google Scholar]
- 113.Srikanth V., Maczurek A., Phan T., et al. Advanced glycation endproducts and their receptor RAGE in Alzheimer’s disease. Neurobiology of Aging. 2011;32(5):763–777. doi: 10.1016/j.neurobiolaging.2009.04.016. [DOI] [PubMed] [Google Scholar]
- 114.Toth C., Martinez J., Zochodne D. W. RAGE, diabetes, and the nervous system. Current Molecular Medicine. 2007;7(8):766–776. doi: 10.2174/156652407783220705. [DOI] [PubMed] [Google Scholar]
- 115.Yan S. D., Chen X., Fu J., et al. RAGE and amyloid-β peptide neurotoxicity in Alzheimer's disease. Nature. 1996;382(6593):685–691. doi: 10.1038/382685a0. [DOI] [PubMed] [Google Scholar]
- 116.Wang X., Yu S., Hu J. P., et al. Streptozotocin-induced diabetes increases amyloid plaque deposition in AD transgenic mice through modulating AGEs/RAGE/NF-kappaB pathway. The International Journal of Neuroscience. 2014;124(8):601–608. doi: 10.3109/00207454.2013.866110. [DOI] [PubMed] [Google Scholar]
- 117.Fang F., Lue L. F., Yan S., et al. RAGE-dependent signaling in microglia contributes to neuroinflammation, Abeta accumulation, and impaired learning/memory in a mouse model of Alzheimer’s disease. The FASEB Journal. 2009;24(4):1043–1055. doi: 10.1096/fj.09-139634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Guzman-Martinez L., Maccioni R. B., Andrade V., Navarrete L. P., Pastor M. G., Ramos-Escobar N. Neuroinflammation as a common feature of neurodegenerative disorders. Frontiers in Pharmacology. 2019;10, article 1008 doi: 10.3389/fphar.2019.01008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Baynes J. W., Thorpe S. R. Role of oxidative stress in diabetic complications: a new perspective on an old paradigm. Diabetes. 1999;48(1):1–9. doi: 10.2337/diabetes.48.1.1. [DOI] [PubMed] [Google Scholar]
- 120.Spinola V., Llorent-Martinez E. J., Castilho P. C. Polyphenols of _Myrica faya_ inhibit key enzymes linked to type II diabetes and obesity and formation of advanced glycation end-products (in vitro): potential role in the prevention of diabetic complications. Food Research International. 2019;116:1229–1238. doi: 10.1016/j.foodres.2018.10.010. [DOI] [PubMed] [Google Scholar]
- 121.Ikram M., Ullah R., Khan A., Kim M. O. Ongoing research on the role of gintonin in the management of neurodegenerative disorders. Cells. 2020;9(6, article 1464) doi: 10.3390/cells9061464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Liu J., Gao F., Ji B., et al. Anthocyanins-rich extract of wild Chinese blueberry protects glucolipotoxicity-induced INS832/13 β-cell against dysfunction and death. Journal of Food Science and Technology. 2015;52(5):3022–3029. doi: 10.1007/s13197-014-1379-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Nizamutdinova I. T., Jin Y. C., Chung J. I., et al. The anti-diabetic effect of anthocyanins in streptozotocin-induced diabetic rats through glucose transporter 4 regulation and prevention of insulin resistance and pancreatic apoptosis. Molecular Nutrition & Food Research. 2009;53(11):1419–1429. doi: 10.1002/mnfr.200800526. [DOI] [PubMed] [Google Scholar]
- 124.Ouchi N., Parker J. L., Lugus J. J., Walsh K. Adipokines in inflammation and metabolic disease. Nature Reviews Immunology. 2011;11(2):85–97. doi: 10.1038/nri2921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Graf D., Seifert S., Jaudszus A., Bub A., Watzl B. Anthocyanin-rich juice lowers serum cholesterol, leptin, and resistin and improves plasma fatty acid composition in Fischer rats. PLoS One. 2013;8(6, article e66690) doi: 10.1371/journal.pone.0066690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Hidalgo M., Oruna-Concha M. J., Kolida S., et al. Metabolism of anthocyanins by human gut microflora and their influence on gut bacterial growth. Journal of Agricultural and Food Chemistry. 2012;60(15):3882–3890. doi: 10.1021/jf3002153. [DOI] [PubMed] [Google Scholar]
- 127.Marques C., Fernandes I., Meireles M., et al. Gut microbiota modulation accounts for the neuroprotective properties of anthocyanins. Scientific Reports. 2018;8(1, article 11341) doi: 10.1038/s41598-018-29744-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Pan P., Lam V., Salzman N., et al. Black raspberries and their anthocyanin and fiber fractions alter the composition and diversity of gut microbiota in F-344 rats. Nutrition and Cancer. 2017;69(6):943–951. doi: 10.1080/01635581.2017.1340491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Valdes-Ramos R., Laura G. L., Elina M. C., Donaji B. A. Vitamins and type 2 diabetes mellitus. Endocrine, Metabolic & Immune Disorders Drug Targets. 2015;15(1):54–63. doi: 10.2174/1871530314666141111103217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Sanlier N., Gokcen B. B., Sezgin A. C. Health benefits of fermented foods. Critical Reviews in Food Science and Nutrition. 2019;59(3):506–527. doi: 10.1080/10408398.2017.1383355. [DOI] [PubMed] [Google Scholar]
- 131.Higashi-Okai K., Nagino H., Yamada K., Okai Y. Antioxidant and prooxidant activities of B group vitamins in lipid peroxidation. Journal of UOEH. 2006;28(4):359–368. doi: 10.7888/juoeh.28.359. [DOI] [PubMed] [Google Scholar]
- 132.Morowitz M. J., Carlisle E. M., Alverdy J. C. Contributions of intestinal bacteria to nutrition and metabolism in the critically ill. The Surgical Clinics of North America. 2011;91(4):771–785. doi: 10.1016/j.suc.2011.05.001. viii. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Vervoort L. M., Ronden J. E., Thijssen H. H. The potent antioxidant activity of the vitamin K cycle in microsomal lipid peroxidation. Biochemical Pharmacology. 1997;54(8):871–876. doi: 10.1016/S0006-2952(97)00254-2. [DOI] [PubMed] [Google Scholar]
- 134.Agostinho P., Cunha R. A., Oliveira C. Neuroinflammation, oxidative stress and the pathogenesis of Alzheimer’s disease. Current Pharmaceutical Design. 2010;16(25):2766–2778. doi: 10.2174/138161210793176572. [DOI] [PubMed] [Google Scholar]
- 135.Bahniwal M., Little J. P., Klegeris A. High glucose enhances neurotoxicity and inflammatory cytokine secretion by stimulated human astrocytes. Current Alzheimer Research. 2017;14(7):731–741. doi: 10.2174/1567205014666170117104053. [DOI] [PubMed] [Google Scholar]
- 136.Castro-Acosta M. L., Smith L., Miller R. J., DI M. C., Farrimond J. A., Hall W. L. Drinks containing anthocyanin-rich blackcurrant extract decrease postprandial blood glucose, insulin and incretin concentrations. The Journal of Nutritional Biochemistry. 2016;38:154–161. doi: 10.1016/j.jnutbio.2016.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Dludla P. V., Joubert E., Muller C. J., Louw J., Johnson R. Hyperglycemia-induced oxidative stress and heart disease-cardioprotective effects of rooibos flavonoids and phenylpyruvic acid-2-O-β-D-glucoside. Nutrition & Metabolism. 2017;14(1):p. 45. doi: 10.1186/s12986-017-0200-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.King G. L., Loeken M. R. Hyperglycemia-induced oxidative stress in diabetic complications. Histochemistry and Cell Biology. 2004;122(4):333–338. doi: 10.1007/s00418-004-0678-9. [DOI] [PubMed] [Google Scholar]
- 139.Matsui T., Ebuchi S., Kobayashi M., et al. Anti-hyperglycemic effect of diacylated anthocyanin derived from Ipomoea batatas cultivar Ayamurasaki can be achieved through the alpha-glucosidase inhibitory action. Journal of Agricultural and Food Chemistry. 2002;50(25):7244–7248. doi: 10.1021/jf025913m. [DOI] [PubMed] [Google Scholar]
- 140.Muhammad T., Ali T., Ikram M., Khan A., Alam S. I., Kim M. O. Melatonin rescue oxidative stress-mediated neuroinflammation/neurodegeneration and memory impairment in scopolamine-induced amnesia mice model. Journal of Neuroimmune Pharmacology. 2019;14(2):278–294. doi: 10.1007/s11481-018-9824-3. [DOI] [PubMed] [Google Scholar]
- 141.Rom S., Zuluaga-Ramirez V., Gajghate S., et al. Hyperglycemia-driven neuroinflammation compromises BBB leading to memory loss in both diabetes mellitus (DM) type 1 and type 2 mouse models. Molecular Neurobiology. 2019;56(3):1883–1896. doi: 10.1007/s12035-018-1195-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Solleiro-Villavicencio H., Rivas-Arancibia S. Effect of chronic oxidative stress on neuroinflammatory response mediated by CD4(+)T cells in neurodegenerative diseases. Frontiers in Cellular Neuroscience. 2018;12:p. 114. doi: 10.3389/fncel.2018.00114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Takahashi A., Okazaki Y., Nakamoto A., et al. Dietary anthocyanin-rich haskap phytochemicals inhibit postprandial hyperlipidemia and hyperglycemia in rats. Journal of Oleo Science. 2014;63(3):201–209. doi: 10.5650/jos.ess13196. [DOI] [PubMed] [Google Scholar]
- 144.Zhao B., Ren B., Guo R., et al. Supplementation of lycopene attenuates oxidative stress induced neuroinflammation and cognitive impairment via Nrf2/NF-κB transcriptional pathway. Food and Chemical Toxicology. 2017;109, Part 1:505–516. doi: 10.1016/j.fct.2017.09.050. [DOI] [PubMed] [Google Scholar]
- 145.Belwal T., Nabavi S. F., Nabavi S. M., Habtemariam S. Dietary anthocyanins and insulin resistance: when food becomes a medicine. Nutrients. 2017;9(10):p. 1111. doi: 10.3390/nu9101111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Hong S. H., Heo J. I., Kim J. H., et al. Antidiabetic and beta cell-protection activities of purple corn anthocyanins. Biomolecules & Therapeutics. 2013;21(4):284–289. doi: 10.4062/biomolther.2013.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Li R., Zhang Y., Rasool S., Geetha T., Babu J. R. Effects and underlying mechanisms of bioactive compounds on type 2 diabetes mellitus and Alzheimer’s disease. Oxidative Medicine and Cellular Longevity. 2019;2019:25. doi: 10.1155/2019/8165707.8165707 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Rugina D., Diaconeasa Z., Coman C., Bunea A., Socaciu C., Pintea A. Chokeberry Anthocyanin Extract as Pancreatic β-Cell Protectors in Two Models of Induced Oxidative Stress. Oxidative Medicine and Cellular Longevity. 2015;2015:10. doi: 10.1155/2015/429075.429075 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Tsuda T. Regulation of adipocyte function by anthocyanins; possibility of preventing the metabolic syndrome. Journal of Agricultural and Food Chemistry. 2008;56(3):642–646. doi: 10.1021/jf073113b. [DOI] [PubMed] [Google Scholar]
- 150.Pugazhenthi S., Qin L., Reddy P. H. Common neurodegenerative pathways in obesity, diabetes, and Alzheimer’s disease. Biochimica et Biophysica Acta - Molecular Basis of Disease. 2017;1863(5):1037–1045. doi: 10.1016/j.bbadis.2016.04.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Alasmari F., Alshammari M. A., Alasmari A. F., Alanazi W. A., Alhazzani K. Neuroinflammatory cytokines induce amyloid beta neurotoxicity through modulating amyloid precursor protein levels/metabolism. BioMed Research International. 2018;2018:8. doi: 10.1155/2018/3087475.3087475 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Bergland A. K., Soennesyn H., Dalen I., et al. Effects of anthocyanin supplementation on serum lipids, glucose, markers of inflammation and cognition in adults with increased risk of dementia - a pilot study. Frontiers in Genetics. 2019;10:p. 536. doi: 10.3389/fgene.2019.00536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Burton-Freeman B., Brzeziński M., Park E., Sandhu A., Xiao D., Edirisinghe I. A selective role of dietary anthocyanins and flavan-3-ols in reducing the risk of type 2 diabetes mellitus: a review of recent evidence. Nutrients. 2019;11(4):p. 841. doi: 10.3390/nu11040841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Hsieh P. S., Chan J. Y. H., Shyu J. F., Chen Y. T., Loh C. H. Mild portal endotoxaemia induces subacute hepatic inflammation and pancreatic beta-cell dysfunction in rats. European Journal of Clinical Investigation. 2008;38(9):640–648. doi: 10.1111/j.1365-2362.2008.01991.x. [DOI] [PubMed] [Google Scholar]
- 155.Jayarathne S., Stull A. J., Park O. H., Kim J. H., Thompson L., Moustaid‐Moussa N. Protective effects of anthocyanins in obesity-associated inflammation and changes in gut microbiome. Molecular Nutrition & Food Research. 2019;63(20, article e1900149) doi: 10.1002/mnfr.201900149. [DOI] [PubMed] [Google Scholar]
- 156.Khan M. S., Ali T., Kim M. W., Jo M. H., Chung J. I., Kim M. O. Anthocyanins improve hippocampus-dependent memory function and prevent neurodegeneration via JNK/Akt/GSK3β signaling in LPS-treated adult mice. Molecular Neurobiology. 2019;56(1):671–687. doi: 10.1007/s12035-018-1101-1. [DOI] [PubMed] [Google Scholar]
- 157.Kowalski K., Mulak A. Brain-gut-microbiota axis in Alzheimer’s disease. Journal of Neurogastroenterology and Motility. 2019;25(1):48–60. doi: 10.5056/jnm18087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Liang H., Hussey S. E., Sanchez-Avila A., Tantiwong P., Musi N. Effect of lipopolysaccharide on inflammation and insulin action in human muscle. PLoS One. 2013;8(5, article e63983) doi: 10.1371/journal.pone.0063983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Lin L., Zheng L. J., Zhang L. J. Neuroinflammation, gut microbiome, and Alzheimer’s disease. Molecular Neurobiology. 2018;55(11):8243–8250. doi: 10.1007/s12035-018-0983-2. [DOI] [PubMed] [Google Scholar]
- 160.Lukiw W. J. Bacteroides fragilis lipopolysaccharide and inflammatory signaling in Alzheimer’s disease. Frontiers in Microbiology. 2016;7, article 1544 doi: 10.3389/fmicb.2016.01544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Putta S., Yarla N. S., Kumar K. E., et al. Preventive and therapeutic potentials of anthocyanins in diabetes and associated complications. Current Medicinal Chemistry. 2018;25(39):5347–5371. doi: 10.2174/0929867325666171206101945. [DOI] [PubMed] [Google Scholar]
- 162.Wang W. Y., Tan M. S., Yu J. T., Tan L. Role of pro-inflammatory cytokines released from microglia in Alzheimer’s disease. Ann Transl Med. 2015;3(10):p. 136. doi: 10.3978/j.issn.2305-5839.2015.03.49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Zhan X., Stamova B., Jin L. W., DeCarli C., Phinney B., Sharp F. R. Gram-negative bacterial molecules associate with Alzheimer disease pathology. Neurology. 2016;87(22):2324–2332. doi: 10.1212/WNL.0000000000003391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Zhao Y., Jaber V., Lukiw W. J. Secretory products of the human GI tract microbiome and their potential impact on Alzheimer’s disease (AD): detection of lipopolysaccharide (LPS) in AD hippocampus. Frontiers in Cellular and Infection Microbiology. 2017;7:p. 318. doi: 10.3389/fcimb.2017.00318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Muhammad T., Ikram M., Ullah R., Rehman S. U., Kim M. O. Hesperetin, a citrus flavonoid, attenuates LPS-induced neuroinflammation, apoptosis and memory impairments by modulating TLR4/NF-κB signaling. Nutrients. 2019;11(3):p. 648. doi: 10.3390/nu11030648. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author upon request.


