Highlights
-
•
Virus-like particles -based vaccines have attracted great interest as the next generation of vaccines due to their safety profile, efficacy, shorter production times, and ability to induce both cellular and humoral immunity.
-
•
The production of HBc-based virus-like particles in plants would thus greatly increase the efficiency of vaccine production.
-
•
This review investigates the application of plant-based HBc VLP as a platform for vaccine production.
Keywords: Hepatitis B core, Virus-like particle, VLP-based vaccine, Plant expression system
Abstract
Virus-like particles (VLPs) are a class of structures formed by the self-assembly of viral capsid protein subunits and contain no infective viral genetic material. The Hepatitis B core (HBc) antigen is capable of assembling into VLPs that can elicit strong immune responses and has been licensed as a commercial vaccine against Hepatitis B. The HBc VLPs have also been employed as a platform for the presentation of foreign epitopes to the immune system and have been used to develop vaccines against, for example, influenza A and Foot-and-mouth disease. Plant expression systems are rapid, scalable and safe, and are capable of providing correct post-translational modifications and reducing upstream production costs. The production of HBc-based virus-like particles in plants would thus greatly increase the efficiency of vaccine production. This review investigates the application of plant-based HBc VLP as a platform for vaccine production.
1. Introduction
Due to the establishment of health organizations around the world, the level of health and well-being of people has increased in recent years. The mortality rate of infectious diseases has fallen and average life expectancy has increased. However, infectious diseases are still a major health concern of the World Health Organization (WHO) and other related organizations. In the 21 st century, various epidemics have been responsible for many deaths globally [1]. In recent years, the WHO has faced emerging dangerous viral diseases including SARS in 2002–2003, Influenza in 2009, MERS in 2012, Chikungunya in 2013, Ebola in 2014–2016, Zika in 2015-present, and SARS-CoV-2 (Covid-19) that has been recently (December 2019) identified in China has been declared a pandemic [2,3]. Therefore, in the case of epidemics, it is essential that a vaccine against the target pathogen be rapidly developed in large quantities. Traditionally vaccines have been composed of either live-attenuated or inactivated pathogens. These vaccines are efficacious but carry the risk of reversion to virulence. As an alternative, recombinant subunit vaccines, including virus-like particles (VLPs), are a new generation of vaccines that are not only safe but can also be rapidly produced in heterologous expression systems.
VLPs are essentially composed of structural proteins without incorporation of a genomic component and are thus non-infectious. A number of VLP-based vaccines against, for example, Hepatitis B virus and human papillomavirus are commercially available. The selection of an appropriate expression host is of crucial importance for such VLP-based vaccines [4,5]. Recombinant proteins used as biopharmaceuticals are complex molecules and thus require an appropriate host to attain the desired biological function. Factors that should be considered for the selection of an expression system include the correct folding and appropriate post-translational modifications of the protein, protein expression level and safety, contamination with endogenous agents, scalability, and production and maintenance costs. Recent advances in the development of expression systems have made it easier to select an appropriate system. Bacterial, yeast, insect, mammalian and plant expression systems all have specific advantages, as well as disadvantages, that need to be taken into account when selecting an expression system for a particular type of protein. Although easy to manipulate and scale-up, bacterial cells lack the post-translational machinery required for eukaryotic protein modifications and there is also the possibility of endotoxin and acetate accumulation, which cause detrimental effects on cell culture [6,7]. In insect cells, protein expression levels are often low and sites with potential N-linked glycosylation are often either glycosylated or not glycosylated, differing from glycosylation patterns in mammalian cells [8,9]. Although mammalian cells offer correct post-translational modification and protein folding, are scalable and yield adequate amounts of protein, limitations of this expression system include high costs of production, purification, and maintenance as well as safety issues with the possible contamination with endogenous pathogens. Due to an increased demand for large quantities of high-quality pharmaceuticals and diagnostic proteins in a limited amount of time and at low cost, the plant expression system is a suitable alternative with high potential in the production of recombinant vaccines and antibodies [10,11].
2. VLP-based vaccines
There are many types of biological and chemical agents for the prevention or control of disease, including monoclonal and polyclonal antibodies, peptides, small-molecule drugs, oligonucleotide-based therapeutics, interferons, and vaccines [[12], [13], [14]]. From the 18th century to the present time, great changes have taken place in the process of making vaccines. Vaccination has undoubtedly been one of the most successful and cost-effective health interventions, preventing the deaths of millions of people throughout the world every year. Most commercial vaccines are killed or live attenuated disease agents that induce immunity. However, one of the most critical problems of these vaccines is the possibility of reverting to virulence. New biotechnology and genetic engineering techniques have recently provided a viable, efficacious and, cost-effective alternative to these traditional vaccines. VLPs have been demonstrated to be safe, highly immunogenic and represent a promising new approach to vaccine development [[15], [16], [17], [18], [19]]. VLP vaccines were first developed in the early 1980s with the assembly of HBV VLPs in the yeast expression system [20]. A number of commercially available VLP-based vaccines are listed in Table 1 and include influenza [21] and hepatitis A vaccines [22]. VLP-based vaccines have all the characteristics of traditional vaccines capable of eliciting powerful and rapid cellular and humoral immune response [4] without the ability to replicate and cause disease. This is likely due to the preservation of the symmetrical size, shape, and structures of the infectious virus in the VLPs [23,24].
Table 1.
Commercial VLP-based vaccines and their characterizations.
| Vaccine Name | Pathogen | Antigen | Expression System | Company | Reference |
|---|---|---|---|---|---|
| Gardasil | HPV1 | L1 protein 6,111,618 | Yeast (S. cerevisiae) | Merck | [164] |
| Cervarix | HPV | L1 protein 1618 | Insect/baculovirus | GSK | [165,166] |
| GenHevac B | HBV | PreS1 + 2 and HBsAg | Mammalian (CHO cells) | Pasteur-Merieux Aventis | [167] |
| Bio-Hep-B | HBV | HBsAg | Mammalian (CHO cells) | BTG (SciGen, FDS Pharma) | [168] |
| DTP-Hep B | HBV | HBsAg | Yeast (P. pastoris) | P.T. Bio Farma | [169] |
| Engerix-B | HBV | HBsAg | Yeast (S. cerevisiae) | GSK | [71,108] |
| Euvax B | HBV | HBsAg | Yeast (S. cerevisiae) | LG Life ScienceS | [170] |
| Gene Vac-B | HBV | HBsAg | Yeast (H. polymorpha) | Serum Inst. of India | [171] |
| Heberbiovac HB | HBV | HBsAg | Yeast (P. pastoris) | CIGB-Heber Biotec | [172] |
| Hepavax-Gene | HBV | HBsAg | Yeast (H. polymorpha) | Crucell | [173] |
| Recombivax HB | HBV | HBsAg | Yeast (S. cerevisiae) | MercK | [174,175] |
| Revac-B | HBV | HBsAg | Yeast (P. pastoris) | Bharat Biotech | [176] |
| Shanvac-B | HBsAg | Yeast (P. pastoris) | Shantha | [177] | |
| Epaxal | HAV | Inactivated HAV RG-SB | Cell-free | Crucell | [22] |
| Inflexal V | Influenza | A (H1N1), A (H3N2), B, HA, NA | Cell-free | Crucell | [21] |
| Hecolin | HEV | capsid protein | Escherichia coli (Chinese market) | Xiamen Innovax Biotech Co | [178] |
Pathogen abbreviations.
VLPs are regular nanometer-scale protein units with spontaneous assembly capability [25,26]. They are observed in a variety of viruses due to their size (22–150 nm). The structural proteins of VLPs spontaneously assemble after expression in recombinant systems [27,28]. These particles are often observed in the form of icosahedron or rod-like structures [29] and due to the absence of nucleic acid, they are non-replicating and not infectious [4]. There is thus no possibility of any genetic events occurring such as insertion, recombination, reversion, or re-assortment and therefore VLPs are generally considered safe. VLP vaccines, although viral in origin, have thus far not caused any serious side effects in inoculated individuals bar some classic mild adverse effects, such as local pain at the site of injection. These vaccines also have fewer side effects when compared to some other vaccines on the market. [18,30,31]. Another advantage of VLPs is that they are stable nanoparticles that can be utilized for the delivery of antigens or drugs [4,32,33]. There is also some evidence that the VLPs are less reliant on cold chains for storage and transportation [34,35]. Table 2 details VLP vaccine candidates undergoing preclinical or clinical trials (Table 3).
Table 2.
VLP-based vaccines that were expressed in different expression systems and their research phases.
| Vaccine Name | Antigen | Stage of development | VLP type | Expression System | Sponsor | Reference |
|---|---|---|---|---|---|---|
| Chikungunya Virus | Glycoprotein | Phase I | Chikungunya Virus -VLP | Baculovirus | Jenner Institute, University of Oxford (UK) | [25,179] |
| Ebola virus | VP40, glycoprotein | Ebola virus-VLP | Insect cells | Animal Cell Technology Unit, IBET (Portugal) | [179,180] | |
| Influenza A virus | haemagglutinin and matrix protein | Preclinical trials | Influenza A virus-VLP | Baculovirus | Instituto de TecnologiaQuímica e Biológica/Universidade Nova de Lisboa, (Portugal) | [4,181] |
| Norovirus | NV capsid | Clinical trials | Norovirus-VLP | Escherichia coli | Jenner Institute, University of Oxford (UK) | [25,182] |
| Norwalk virus | Capsid | Phase I | Norwalk virus-VLP | Baculovirus | Animal Cell Technology Unit, IBET (Portugal) | [180,183] |
| Respiratory syncytiavirus (RSV) | G protein | Trials in non-human primates | Alfalfa mosaic virus-VLP | Nicotiana tabacum | Cell Biology Department, The Scripps Research Institute (USA) | [16,184] |
| Rotavirus | SA11 gene | Pre-clinical trials (Animal trials) | Rotavirus-VLP | Baculovirus | Cell Biology Department, The Scripps Research Institute (USA) | [16,185] |
| SARS-CoV-2 | Spike glycoproteins | Phase I | CoVLP | Nicotiana benthamiana | Medicago, Quebec, QC, Canada | [186] |
| Influenza | H1N1 | phase 3 trial | QVLP | Nicotiana benthamiana | Medicago, Quebec, QC, Canada | [187] |
Table 3.
Use of hepatitis B core as a vaccine platform to display epitopes in several expression systems.
| Pathogen | Epitope(s) | Expression system | Site of epitope insertion | Ref. |
|---|---|---|---|---|
| Dengue virus | cEDIII | N. benthamiana | MIR | [68] |
| H1N1 Influenza A virus | matrix protein 2 | E. coli | MIR | [188] |
| H7N9 Influenza | long alpha-helix (LAH) | E. coli | MIR | [189] |
| Mycobacterium Tuberculosis (Tuberculosis) | Culture filtrate protein 10 (CFP 10) | E. coli | MIR | [190] |
| – | Hepatitis B Core Antigen | E. coli | MIR | [86] |
| Nicotiana benthamiana | ||||
| GGS sequence | ||||
| Influenza virus | M2e | E. coli | MIR | [191] |
| Influenza virus | M2e | N. benthamiana | N-terminal | [192] |
| Dengue virus | EDIII-2 | E. coli | MIR | [193] |
| Dengue virus | EDIII | E. coli | MIR | [193] |
| Hepatitis C virus | HCc N-terminus | E. coli | C-terminal | [194] |
| Influenza A | M2e | E. coli | N-terminal | [195] |
| Hepatocellular carcinoma | HBV X protein | E. coli | C-terminal, MIR | [196] |
| Hepatocellular carcinoma | AFP1, AFP2 | E. coli | C-terminal | [197] |
| Hepatitis C virus | HCc T-cell epitope | E. coli | MIR | [198] |
| Foot-and-mouth-disease virus | VP1, VP4 | N. tabacum, E. coli | MIR | [199,200] |
| Hepatitis B virus | Pre-S1 | E. coli | MIR | [201] |
| Hantavirus | Nucleocapsid protein | E. coli | MIR | [202] |
| Hepatitis B virus | Pre-S1 | E. coli | C-terminal | [203] |
| Human Papillomavirus | E7 | E. coli | C-terminal | [204] |
| Theileria annulata | SPAG-1 | E. coli | MIR | [205] |
| Human Immunodeficiency Virus | Gag | E. coli | N-terminal | [206] |
| Human Immunodeficiency Virus | Env | E. coli | C-terminal | [90] |
| Foot-and-mouth-disease virus | VP1 | E. coli | N-terminal | [67] |
3. The immune response to VLPs
VLPs can induce both cellular and humoral immunity without the need for adjuvants [34]. They are able to trigger a potent T cell response [[36], [37], [38], [39], [40], [41], [42], [43]].VLPs are absorbed by a variety of cells, including dendritic cells (DCs), due to their very small size, which is between 20−300 nm. It has been shown that adding sequences such as CpG can promote DC stimulation [44]. The internalization of VLPs by antigen-presenting cells (APCs) and subsequent presentation to CD8 + T cells on the MHC class I prompts cell activation, resulting in the induction of a potent immune response [42,[45], [46], [47]]. In addition, VLPs often display different pathogen-associated molecular patterns (PAMPs) that are recognized by pattern recognition receptors (PRRs) [23,[48], [49], [50], [51]]. PAMPs induce immune responses through interaction with PRRs like Toll-like receptors (TLRs) on sentinel cells [23]. Furthermore, it is possible to insert several types of epitopes into the VLP at the same time using genetic engineering [44,[52], [53], [54]], which causes crosslinking between B cell receptors (BCR) leading to B cell activation [34,55]. Epitopes are also able to trigger a B-cell response [56]. Due to their repetitive and small structures, VLPs can prime the B cell for activation, i.e., multiplication and antibody production [57]. In this case, the antigens bound to MHC class II on the surface of APC interact with T helper cells eliciting IgG production and provide signals required for differentiation of B cells toward memory B cells. VLPs are often delivered without an adjuvant [57], leading to the production of high antibody titers [55,58].
4. Engineering VLPs as a vaccine platform
The regular structured VLPs have made it possible to insert and present heterologous epitopes on the surface of these particulate structures [16]. HPV16 L1 protein, which self-assembled into VLPs in plants, has proven highly immunogenic and efficacious for vaccine production [59]. Also very recently, the use of grapevine fanleaf virus (GFLV) VLPs has been studied as a new carrier for the presentation of the HPV L2 epitope [60]. More than 40 years have passed since VLPs were first obtained from the HBV surface antigen of HBsAg [40,61]. The HBcAg polypeptide is about 21 kDa in size and consists of 183–185 amino acid residues that can self-assemble into 27-nm particles [[62], [63], [64]]. HBc VLPs have been widely studied in the last two decades. They were first reported in 1987 as carriers displaying heterologous epitopes of foot and mouth disease virus. After the primary study by Clarke et al. in 1987 [65], different epitopes and antigens have been introduced into HBc protein for vaccine development (Table 6). The immunogenic epitope of the virus was fused to the N-terminus end of the HBc sequence [[66], [67], [68], [69]]. This VLP has been used to produce HBV vaccine in yeast [70,71] and mammalian cells (CHO) [20]. HBc has been expressed in various prokaryotic and eukaryotic expression systems [20,36,42,43,70,[72], [73], [74], [75], [76], [77], [78], [79], [80], [81], [82], [83], [84], [85]]. X-ray crystallography and cryo-electron microscopy studies on HBc components revealed that these are icosahedral particles [86]. HBc monomers are observed in two different sizes when packaged as a VLP, consisting of 180 or 240 subunits, and their symmetry is obtained in two forms, T = 3 or T = 4 [87]. The linear structure of the protein consists of two parts. Residual amino acids 1–140 comprise the N-terminus region, which consists of a SA domain required for self-assembly. The C-terminus (CTD), is a region (150–183 amino acids) rich in arginine called protamine [88]. Gallina et al. in 1989 showed that the protamine domain is not related to the particle assembly, but it is important for stabilizing the particles with neighboring disulfide bonds [89]. SA domain has a variable area associated with B-cell epitopes, while the second CTD and hinge peptide are highly conserved [78]. The N-terminus in HBcAg is used as an insertion site for external epitopes, which enable the insertion of up to 50 amino acids and induce specific antibody responses. Another place of insertion is the end of the C terminus and in the position of amino acids 144−127. In the case of the C-terminus, it is possible to insert the epitopes at amino acids 144, 149, 153, 163, and 169 [66]. In 1989, Stahl and Murray successfully fused the immunogenic part of HBsAg of HBV and the envelope of Human Immunodeficiency Virus (HIV) to the C-terminus of HBc sequence [90].
Table 6.
VLP-based vaccines produced in plants.
| Application | Antigen | Production Plant | Study Phase | Sponsor | Reference |
|---|---|---|---|---|---|
| Bluetongue | VP3,VP7, VP5 and VP2 protein | N. benthamiana | Pre-clinical | Department of Biological Chemistry, John Innes Centre, Norwich, UK | [207] |
| Foot-and-mouth disease | Structural proteins (VP0, VP1 and VP3) | N. benthamiana | Research | Institute of Infectious Disease and Molecular Medicine (South Africa) | [208] |
| Hepatitis B | HBsAg | Tobacco | Phase I | Biodesign Institute at Arizona State University (USA) | [209] |
| Hepatitis B | HBsAg | Lettuce | Phase I | Institute of Biotechnology and Antibiotics (Poland) | [210,211] |
| Hepatitis B | Glycol protein | Spinach | Phase I | Institute of Biotechnology and Antibiotics (Poland) | [212] |
| HIV | Pr55 gag protein | Tobacco | Research | Institute of Plant Genetics (Italy) | [213] |
| HBV-HIV | env and gag proteins | Tomato | Research | State Research Center of Virology and Biotechnology Vector (Russia) | [214] |
| HPV | 16 L1 | Nicotiana tabacum | Research | Department of Molecular and Cell Biology (South Africa) | [215] |
| Influenza | HA(H5N1) | N. benthamiana | Phase I/II | Medicago (Canada) | [216,217] |
| Influenza | HA(H1N1) | N. benthamiana | Phase I | Medicago (Canada) | [218] |
| Influenza | HA(H5N1) | N. tabacum | Phase II | Infectious Disease Research Institute, Seattle, WA (USA) | [142] |
| Influenza | H5N1 | N. benthamiana | Research | Department of Research & Development, Haukeland University Hospital, Bergen, (Norway) | [219] |
| Malaria | Pfs25-CP | N.Tobacco | Phase I | Center for Molecular Biotechnology, Plymouth, MI(USA) | [220] |
| Noroviruses | NaVCP | N. benthamiana | Research | Center for Infectious Diseases and Vaccinology (USA) | [221] |
| Norwalk | Capsid protein | Potato and tobacco | Phase I | University of Maryland (USA) | [222] |
| Norwalk | Capsid proteins | Tomato | Pre-clinical | Biodesign Institute and School of Life Sciences (USA) | [150] |
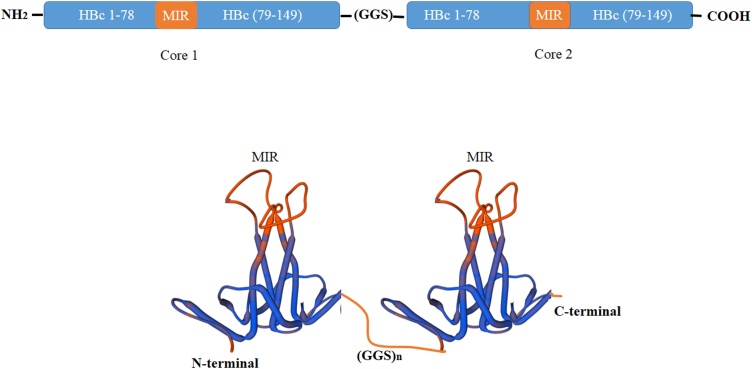
In addition to the N and C termini of the HBc protein, epitopes may also be inserted into the Major immunodominant region (MIR) in the e1 loop of the HBcAg protein [13]. Studies have indicated that such insertions result in a 10-fold stronger immune response and may be the most efficient site for epitope presentation [91,92]. The position of the MIR region is located between the amino acids 82–88 and at the tip of the α-coil [77]. Four cysteines exist in the HBcAg sequence located at 48, 61, 107, and 183 positions. Disulfide bonding between two monomers to create a dimer usually takes place between cys 61 and sometimes cys 48 of adjacent monomers [86]. The icosahedral VLPs are formed by the association of 180 (90 dimers; T = 3) or 240 (120 dimers; T = 4) copies of HBcAg proteins. VLPs with 180 HBcAg copies are about 30 nm, while those with 240 copies are approximately 34 nm in diameter [93]. The dimer consists of four-helix bundles connected by loops that form spikes on the outer surface of VLPs. The amino acid residues 76–82 of each HBcAg monomer known as MIR or c/e1 loop, are placed at the tip of the spike after VLP assembly. Therefore, after inserting an epitope into the MIR or C- or N-termini regions of an HBcAg monomer, HBc VLPs can self-assemble following the expression of the monomeric form [68,69,92]. HBc VLPs was one of the first VLPs produced in the plant system through transient expression technology [76] Recently, Peyret et al. 2015, have introduced a system called the ‘Tandem Core’, which is shown in Fig. 1, and which has advantages over the previous system including the reduction of steric clashes due to the non-random association of subunits, flexibility in inserting a wide range of epitopes, and flexibility to insert larger epitopes. It has been shown that inserting large or hydrophobic sequences in the MIR region can reduce antigenicity and immunogenicity [77]. One way to overcome this problem is to create a mosaic VLP so that HBcAg proteins with large insertions in their MIR area are co-expressed with wild-type HBcAg [94]. Another solution is to use a ‘Split Core’ and insert an antigen into the N- and C-termini regions [86] thus creating a dimer protein. Particles have high inherent antigenicity due to their spiky and repetitive structures that allow the presentation of antigens on their surface [78,95]. Because of its polymeric nature and the presence of a large number of T-cell epitopes, HBc protein has high immunogenicity [95]. HBc protein exists in two forms, T-cell-dependent and T-cell-independent. They can activate macrophages, and high antibody production [[96], [97], [98], [99]]. HBc, as a potent T cell epitope, can stimulate Toll-like receptors [100,101]. These antigens can act as T-cell and B-cell epitopes and induce immunogenicity [[102], [103], [104]]. HBc VLPs are very immunogenic in laboratory models such as mice and no cytotoxic effects have been reported in humans. [69]. Recently HBc was suggested as a new platform against SARS-CoV-2 by exposing immunogenic epitopes [105].
Fig. 1.
Illustration of the Tandem Core technology concept. The image shows two HBcAg genetically fused to one another via a flexible linker. MIR: Major immunodominant region.
5. Expression systems used for VLP-based vaccine production
Despite the commercialization of several VLP-based vaccines, the current bacterial, yeast, insect and mammalian cell expression systems suffer from various limitations [[106], [107], [108]]. As the selection of a suitable expression system results in increased vaccine efficacy, scalability, and performance, and can affect the production costs, it is important that the advantages and disadvantages of each system be weighed for each particular vaccine candidate. A comparison of the differences between the available expression systems is given in Table 4. Bacteria are the most commonly used expression system for the production of recombinant proteins, and 30 % of the VLPs described are produced in the bacteria [109]. Interestingly, despite the high number of VLPs produced in E. coli, no VLP derived from this bacterium has yet been commercialized. The main concerns are the inability of prokaryotes to perform post-translational modifications and the complexity of protein purification owing to the significant differences between the eukaryotes and the prokaryotes [[110], [111], [112]]. Yeast, insect and mammalian cells have also been extensively utilized in the production of VLPs [113,114]. Yeast expression systems are the most utilized in recombinant vaccine production due to the relative ease of genetic manipulation and their rapid growth. Yeast cells are most effective in producing non-enveloped VLPs and also make the study of complex VLPs possible [106,115,116]. However, the glycosylation capacity of yeast cells is limited [117]. Other disadvantages of the yeast expression system include low yield, low plasmid stability, and low secretion capacity [118,119]. In the case of insect cells, post-translational changes can be accompanied by high levels of sugars resulting in hyper glycosylation, which affects the effectiveness of the vaccine and increases the cost [[120], [121], [122], [123]]. Furthermore, protein expression in both bacterial and yeast systems is lower than in plants [124]. There are some disadvantages in using of insect expression system including the possibility of contamination of insect cell cultures, different post-translational modifications and proteolysis cleavage in areas rich in Lysine and Arginine. Mammalian expression systems have advantages such as correct post-translational modifications, correct protein folding and are free of bacterial endotoxins. However, the need for special fermentation devices, time-consuming cell preparation, slow growth, the possibility of contamination with endogenous pathogens, and high production and maintenance costs limit its use in many countries, especially developing countries [125]. Plants are considered a cost-effective, scalable, efficient, and safe alternative to the current mainstream expression systems for the production of VLP-based vaccines.
Table 4.
Comparison of different expression systems for the generation of pharmaceutical proteins.
| Disadvantages | Advantages | Production method |
|---|---|---|
|
|
E. coli |
|
|
Yeast |
|
|
Insect cells |
|
|
Mammalian cells |
|
|
Plants |
6. Plants as expression systems for VLP-based vaccine production
Over the last two decades, plants have been increasingly used as a production host for recombinant proteins. Plant expression systems are characterized by a high yield of proteins, ease of protein purification and faster recombinant protein production [[126], [127], [128], [129]]. A major advantage of the plant expression systems is their ability to produce large amounts of recombinant protein in a fast and cost-effective manner [[130], [131], [132]]. Whole plants can be grown in large quantities in greenhouses without requiring bioreactor-based fermentation methods [133]. In plants, it is only necessary to increase the area under plant cultivation to increase antigen production [124]. Plants have a short growth cycle and mature quickly, thus reducing the production costs compared to other systems. Hiatt et al., in 1989 produced the first monoclonal antibody (mAb) in the transgenic tobacco plant [134]. Examples of VLP-based vaccines produced in plant expression systems include those against Influenza and foot-and-mouth diseases [[135], [136], [137], [138]]. Plant virus nanoparticles have also been used as carrier particles for drug delivery and imaging [139]. Although the glycosylation patterns in plants are slightly different from those of mammalian cells [65,140], plant-expressed H5N1 influenza virus-like particles have demonstrated safety in Phase II clinical trials [141,142] and plant-made pharmaceuticals are aggressively being investigated, most recently against the Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). A phase 1 trial of a VLP-based vaccine against SARS-CoV-2, transiently expressed in plants, was recently conducted by Medicago [143]. In plants, there is also no need to worry about the contamination of toxins and endogenous pathogens, which usually occur in bacterial and mammalian cell-based expression systems [[144], [145], [146]]. Unlike prokaryotic systems, plants have the same secretory pathway as human cells [147]. Another feature of this expression system is the simplicity of storing the recombinant proteins [148,149]. Table 5 shows the list of vaccines produced in plants.
Table 5.
Plant-derived vaccines and their clinical trial phase.
| Target | Interventions? | Plant Host | Study Type | Study Completion Date | Sponsor |
|---|---|---|---|---|---|
| Malaria | Pfs25 VLP- FhCMB | N. benthamiana | Interventional (Clinical Trial: Phase 1) (NCT02013687) | January 2015 | Fraunhofer, Center for Molecular Biotechnology |
| Anthrax | PA83-FhCMB | N. benthamiana | Interventional (Clinical Trial: Phase 1) (NCT02239172) | May 2015 | Fraunhofer, Center for Molecular Biotechnology |
| Influenza A Subtype H5N1 Infection | H5-VLP + GLA-AF; Licensed H5N1 vaccine | N. benthamiana | Interventional (Clinical Trial: Phase 1) (NCT01657929) | January 2014 | IDRI |
| H5N1 Flu | HAI-05 Influenza Vaccine; Saline | N. benthamiana | Interventional (Clinical Trial: Phase 1) (NCT01250795) | July 2011 | Fraunhofer, Center for Molecular Biotechnology |
| 2018−2019 influenza season | Quadrivalent VLP Vaccine | N. benthamiana | Interventional (Clinical Trial: Phase 3) (NCT03739112) | June 14, 2019 | Medicago |
| H1N1 Flu | HAC1 Vaccine | N. benthamiana | Interventional (Clinical Trial: Phase 1) (NCT01177202) | October 2012 | Fraunhofer, Center for Molecular Biotechnology |
| H1N1 Flu | Quadrivalent VLP Vaccine; Placebo | N. benthamiana | Interventional (Clinical Trial: Phase 3) (NCT03301051) | June 2018 | Medicago |
| H1N1 Flu | H1N1 VLP vaccine | N. benthamiana | Interventional (Clinical Trial; phas 1) (NCT01302990) | July 2011 | Medicago |
| H5N1 Flu | H5N1 VLP vaccine | N. benthamiana | Interventional (Clinical Trial; Phase 2) (NCT01991561) | July 2014 | Medicago |
| H7N9 Flu | H7N9 VLP vaccine | N. benthamiana | Interventional (Clinical Trial; Phase 1) (NCT02022163) | September 2014 | Medicago |
| Lymphoma, Follicular | Autologous FL vaccine | N. benthamiana | Interventional (Clinical Trial; Phase 1) (NCT01022255) | October 2013 | Icon Genetics GmbH |
| HIV Infection | P2G12 | N. tabacum | Interventional (Clinical Trial; Phase 1) (NCT02923999) | August 2020 | St George's, University of London |
Although plants such as potatoes, tomatoes, corn, soybeans, rice, and carrots have been used to study immunogenicity, particularly that of oral vaccines that trigger the mucosal immune response [[150], [151], [152]], Nicotiana benthamiana plants are the stalwart of plant expression system and offer a number of advantages, including non-food crop status, high growth rate, growth in greenhouses that reduces the risk of spread of contamination, and accessibility of suitable and efficient vectors for enhanced gene expression [[153], [154], [155], [156]]. As indicated in Table 6, diverse types of VLPs have been expressed in plants. Expression in these plants can be either transient, which enables rapid, high-volume and low-cost production of vaccines [145], or transgenic, which, although stable, typically yields low levels of expression compared to the transient expression [157]. The transient expression makes screening and production of the desired protein possible in a few days which is particularly important for the development of vaccines during epidemics. Protein expression is also limited to the infiltrated tissue preventing the risk of transfer of foreign genes to the gametes of the plant, thus inhibiting cross-pollination and spread within an unintended plant population. Protein production on a large scale can be accomplished by means of vacuum infiltration of all plant leaves [158]. Plant cell culture has emerged as an alternative bio-production system for the production of recombinant pharmaceuticals. The recombinant glucocerebrosidase enzyme (Taliglucerase Alfa) that is produced within carrot cells, has been developed as a plant cell-made pharmaceutical for the treatment of Gaucher’s disease [159].
In transgenic products, the production of vaccines can be directed into the cytoplasm, as it has been done in tobacco and lettuce [160,161]. In a study, the insertion of some epitopes of T. gondii into a truncated HBcΔ particle triggered strong humoral and cellular immune responses [162]. A more immunogenic VLP vaccine containing, in addition to HBcAg, the proteins PreS1 and PreS2, was found to elicit a strong antibody response [163]. Like VLPs produced in other systems, some plant-derived VLPs are able to induce protective humoral and cellular immune responses. Plant-expression systems can produce large quantities of immunogenic HBcAg [24]. Several recombinant VLPs, including HBcAg VLPs displaying M2 epitope of influenza A, yeast transposon Ty VLPs displaying HIV p17/p24 antigens, and HBcAg VLPs displaying malaria epitopes have been evaluated. Also, expression and assembly of HBcAg-HPV16 L2 epitope VLPs in tobacco-induced antigen-specific antibody response in mice [24].
7. Conclusion
Many virus-like particles have been used in the last three decades experimentally or commercially for various purposes such as drug loading and delivery, medical imaging and vaccine production. Hepatitis B virus (HBV) core particles (HBc) have been the VLP of choice for commercial vaccine production, not only against HBV but also against many other viral and non-viral agents. The HBsAg particle was the first VLP-based vaccine and today, due to its compatibility with a diverse array of expression systems, many of these particles are manufactured for commercial purposes. HBc is also one of the most promising VLP presentation platforms due to its high immunogenicity, its enhanced presentation to the immune system, and its flexibility to allow a wide variety of foreign insertions without affecting the protein self-assembly and VLP function. These features will increase the versatility and efficiency of this type of vaccine in the future. The production of HBc VLP-based vaccines in plants is an attractive option in developing countries where the implementation of more sophisticated technologies is often met with difficulties. The use of HBc and other VLP platforms, including those of plant viruses, is a developing field in biopharmaceutical and medical sciences.
Declaration of Competing Interest
The authors declare that they have no conflict of interest. The research reported here did not involve experimentation with human participants or animals.
Contributor Information
Abozar Ghorbani, Email: Abozar.ghorbani@shirazu.ac.ir.
Mohammad Hadi Eskandari, Email: eskandar@shirazu.ac.ir.
References
- 1.Bloom D.E., Cadarette D. Infectious disease threats in the twenty-first century: strengthening the global response. Front. Immunol. 2019;10:549. doi: 10.3389/fimmu.2019.00549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Huang C. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395(10223):497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hemmati F. Mysterious virus: a review on behavior and treatment approaches of the novel coronavirus, 2019-nCoV. Arch. Med. Res. 2020 doi: 10.1016/j.arcmed.2020.04.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Roldão A. Virus-like particles in vaccine development. Expert Rev. Vaccines. 2010;9(10):1149–1176. doi: 10.1586/erv.10.115. [DOI] [PubMed] [Google Scholar]
- 5.Crisci E., Bárcena J., Montoya M. Virus-like particle-based vaccines for animal viral infections. Inmunologia. 2013;32(3):102–116. doi: 10.1016/j.inmuno.2012.08.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Martínez J.L. Pharmaceutical protein production by yeast: towards production of human blood proteins by microbial fermentation. Curr. Opin. Biotechnol. 2012;23(6):965–971. doi: 10.1016/j.copbio.2012.03.011. [DOI] [PubMed] [Google Scholar]
- 7.Sahdev S., Khattar S.K., Saini K.S. Production of active eukaryotic proteins through bacterial expression systems: a review of the existing biotechnology strategies. Mol. Cell. Biochem. 2008;307(1-2):249–264. doi: 10.1007/s11010-007-9603-6. [DOI] [PubMed] [Google Scholar]
- 8.Demain A.L., Vaishnav P. Production of recombinant proteins by microbes and higher organisms. Biotechnol. Adv. 2009;27(3):297–306. doi: 10.1016/j.biotechadv.2009.01.008. [DOI] [PubMed] [Google Scholar]
- 9.Houdebine L.-M. Production of pharmaceutical proteins by transgenic animals. Comp. Immunol. Microbiol. Infect. Dis. 2009;32(2):107–121. doi: 10.1016/j.cimid.2007.11.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Schillberg S. Critical analysis of the commercial potential of plants for the production of recombinant proteins. Front. Plant Sci. 2019;10:720. doi: 10.3389/fpls.2019.00720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Fischer R., Buyel J.F. Molecular farming - the slope of enlightenment. Biotechnol. Adv. 2020;40:107519. doi: 10.1016/j.biotechadv.2020.107519. [DOI] [PubMed] [Google Scholar]
- 12.Li G., De Clercq E. Therapeutic options for the 2019 novel coronavirus (2019-nCoV) Nat. Rev. Drug Discov. 2020;19(3):149–150. doi: 10.1038/d41573-020-00016-0. [DOI] [PubMed] [Google Scholar]
- 13.Andre F.E. Vaccination greatly reduces disease, disability, death and inequity worldwide. Bull. World Health Organ. 2008;86(2):140–146. doi: 10.2471/BLT.07.040089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Balke I., Zeltins A. Use of plant viruses and virus-like particles for the creation of novel vaccines. Adv. Drug Deliv. Rev. 2019;145:119–129. doi: 10.1016/j.addr.2018.08.007. [DOI] [PubMed] [Google Scholar]
- 15.Liu F. Virus-like particles: potential veterinary vaccine immunogens. Res. Vet. Sci. 2012;93(2):553–559. doi: 10.1016/j.rvsc.2011.10.018. [DOI] [PubMed] [Google Scholar]
- 16.Plummer E.M., Manchester M. Viral nanoparticles and virus‐like particles: platforms for contemporary vaccine design. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2011;3(2):174–196. doi: 10.1002/wnan.119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Haynes J.R. Influenza virus-like particle vaccines. Expert Rev. Vaccines. 2009;8(4):435–445. doi: 10.1586/erv.09.8. [DOI] [PubMed] [Google Scholar]
- 18.Noad R., Roy P. Virus-like particles as immunogens. Trends Microbiol. 2003;11(9):438–444. doi: 10.1016/s0966-842x(03)00208-7. [DOI] [PubMed] [Google Scholar]
- 19.Roy P., Noad R. Virus-like particles as a vaccine delivery system: myths and facts. Hum. Vaccin. 2008;4(1):5–12. doi: 10.4161/hv.4.1.5559. [DOI] [PubMed] [Google Scholar]
- 20.Valenzuela P. Synthesis and assembly of hepatitis B virus surface antigen particles in yeast. Nature. 1982;298(5872):347–350. doi: 10.1038/298347a0. [DOI] [PubMed] [Google Scholar]
- 21.Herzog C. Eleven years of Inflexal V-a virosomal adjuvanted influenza vaccine. Vaccine. 2009;27(33):4381–4387. doi: 10.1016/j.vaccine.2009.05.029. [DOI] [PubMed] [Google Scholar]
- 22.Bovier P.A. Recent advances with a virosomal hepatitis A vaccine. Expert Opin. Biol. Ther. 2008;8(8):1177–1185. doi: 10.1517/14712598.8.8.1177. [DOI] [PubMed] [Google Scholar]
- 23.Bachmann M.F., Jennings G.T. Vaccine delivery: a matter of size, geometry, kinetics and molecular patterns. Nat. Rev. Immunol. 2010;10(11):787–796. doi: 10.1038/nri2868. [DOI] [PubMed] [Google Scholar]
- 24.Chen Q., Lai H. Plant-derived virus-like particles as vaccines. Hum. Vaccin. Immunother. 2013;9(1):26–49. doi: 10.4161/hv.22218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Mohsen M.O. Major findings and recent advances in virus-like particle (VLP)-based vaccines. Semin. Immunol. 2017;34:123–132. doi: 10.1016/j.smim.2017.08.014. [DOI] [PubMed] [Google Scholar]
- 26.Gren E., Pumpen P. Recombinant viral capsids as a new age of immunogenic proteins and vaccines. Zh. Vsesoyuzn. Mendeleevsk. Obshch. Soc. 1988;33:531–536. [Google Scholar]
- 27.Manolova V. Nanoparticles target distinct dendritic cell populations according to their size. Eur. J. Immunol. 2008;38(5):1404–1413. doi: 10.1002/eji.200737984. [DOI] [PubMed] [Google Scholar]
- 28.Johnson J.E., Chiu W. Structures of virus and virus-like particles. Curr. Opin. Struct. Biol. 2000;10(2):229–235. doi: 10.1016/s0959-440x(00)00073-7. [DOI] [PubMed] [Google Scholar]
- 29.Chroboczek J., Szurgot I., Szolajska E. Virus-like particles as vaccine. Acta Biochim. Pol. 2014;61(3):531–539. [PubMed] [Google Scholar]
- 30.Bachmann M.F., Zinkernagel R.M., Oxenius A. Immune responses in the absence of costimulation: viruses know the trick. J. Immunol. 1998;161(11):5791–5794. [PubMed] [Google Scholar]
- 31.Zepeda-Cervantes J., Ramírez-Jarquín J.O., Vaca L. Interaction between virus-like particles (VLPs) and pattern recognition receptors (PRRs) from dendritic cells (DCs): toward better engineering of VLPs. Front. Immunol. 2020;11(1100) doi: 10.3389/fimmu.2020.01100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Pumpens P. Medicinal Protein Engineering. CRC Press Taylor & Francis Group; Boca Raton, FL: 2008. Construction of novel vaccines on the basis of the virus-like particles: hepatitis B virus proteins as vaccine carriers; pp. 205–248. [Google Scholar]
- 33.Zeltins A. Construction and characterization of virus-like particles: a review. Mol. Biotechnol. 2013;53(1):92–107. doi: 10.1007/s12033-012-9598-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Chackerian B. Virus-like particles: flexible platforms for vaccine development. Expert Rev. Vaccines. 2007;6(3):381–390. doi: 10.1586/14760584.6.3.381. [DOI] [PubMed] [Google Scholar]
- 35.Chen Q. Subunit vaccines produced using plant biotechnology. New generation vaccines. 2009;4 [Google Scholar]
- 36.Braun M. Virus-like particles induce robust human T-helper cell responses. Eur. J. Immunol. 2012;42(2):330–340. doi: 10.1002/eji.201142064. [DOI] [PubMed] [Google Scholar]
- 37.Martins G., Calame K. Regulation and functions of Blimp-1 in t and B lymphocytes. Annu. Rev. Immunol. 2008;26:133–169. doi: 10.1146/annurev.immunol.26.021607.090241. [DOI] [PubMed] [Google Scholar]
- 38.Gomes A.C. Adjusted Particle Size Eliminates the Need of Linkage of Antigen and Adjuvants for Appropriated T Cell Responses in Virus-Like Particle-Based Vaccines. Front. Immunol. 2017;8:226. doi: 10.3389/fimmu.2017.00226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Pomwised R. Coupling peptide antigens to virus-like particles or to protein carriers influences the Th1/Th2 polarity of the resulting immune response. Vaccines (Basel) 2016;4(2) doi: 10.3390/vaccines4020015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Sailaja G. Human immunodeficiency virus-like particles activate multiple types of immune cells. Virology. 2007;362(2):331–341. doi: 10.1016/j.virol.2006.12.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Bachmann M.F. The influence of antigen organization on B cell responsiveness. Science. 1993;262(5138):1448–1451. doi: 10.1126/science.8248784. [DOI] [PubMed] [Google Scholar]
- 42.Bachmann M.F. Dendritic cells process exogenous viral proteins and virus-like particles for class I presentation to CD8+ cytotoxic T lymphocytes. Eur. J. Immunol. 1996;26(11):2595–2600. doi: 10.1002/eji.1830261109. [DOI] [PubMed] [Google Scholar]
- 43.Schirmbeck R., Böhm W., Reimann J. Virus-like particles induce MHC class I-restricted T-cell responses. Lessons learned from the hepatitis B small surface antigen. Intervirology. 1996;39(1-2):111–119. doi: 10.1159/000150482. [DOI] [PubMed] [Google Scholar]
- 44.Storni T. Nonmethylated CG motifs packaged into virus-like particles induce protective cytotoxic T cell responses in the absence of systemic side effects. J. Immunol. 2004;172(3):1777–1785. doi: 10.4049/jimmunol.172.3.1777. [DOI] [PubMed] [Google Scholar]
- 45.Bosio C.M. Ebola and Marburg virus-like particles activate human myeloid dendritic cells. Virology. 2004;326(2):280–287. doi: 10.1016/j.virol.2004.05.025. [DOI] [PubMed] [Google Scholar]
- 46.Mohsen M.O. Delivering adjuvants and antigens in separate nanoparticles eliminates the need of physical linkage for effective vaccination. J. Control. Release. 2017;251:92–100. doi: 10.1016/j.jconrel.2017.02.031. [DOI] [PubMed] [Google Scholar]
- 47.Kouskoff V., Lacaud G., Nemazee D. T cell-independent rescue of B lymphocytes from peripheral immune tolerance. Science. 2000;287(5462):2501–2503. doi: 10.1126/science.287.5462.2501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Kolumam G.A. Type I interferons act directly on CD8 T cells to allow clonal expansion and memory formation in response to viral infection. J. Exp. Med. 2005;202(5):637–650. doi: 10.1084/jem.20050821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Rynda-Apple A., Patterson D.P., Douglas T. Virus-like particles as antigenic nanomaterials for inducing protective immune responses in the lung. Nanomedicine (Lond) 2014;9(12):1857–1868. doi: 10.2217/nnm.14.107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Le Bon A. Cross-priming of CD8+ T cells stimulated by virus-induced type I interferon. Nat. Immunol. 2003;4(10):1009–1015. doi: 10.1038/ni978. [DOI] [PubMed] [Google Scholar]
- 51.López-Macías C. Virus-like particle (VLP)-based vaccines for pandemic influenza: performance of a VLP vaccine during the 2009 influenza pandemic. Hum. Vaccin. Immunother. 2012;8(3):411–414. doi: 10.4161/hv.18757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Win S.J. Cross-presentation of epitopes on virus-like particles via the MHC I receptor recycling pathway. Immunol. Cell Biol. 2011;89(6):681–688. doi: 10.1038/icb.2010.161. [DOI] [PubMed] [Google Scholar]
- 53.Rostovtsev V.V. A stepwise huisgen cycloaddition process: copper (I)‐catalyzed regioselective “ligation” of azides and terminal alkynes. Angew. Chemie Int. Ed. 2002;41(14):2596–2599. doi: 10.1002/1521-3773(20020715)41:14<2596::AID-ANIE2596>3.0.CO;2-4. [DOI] [PubMed] [Google Scholar]
- 54.Fiedler J.D. Engineered mutations change the structure and stability of a virus-like particle. Biomacromolecules. 2012;13(8):2339–2348. doi: 10.1021/bm300590x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Grgacic E.V., Anderson D.A. Virus-like particles: passport to immune recognition. Methods. 2006;40(1):60–65. doi: 10.1016/j.ymeth.2006.07.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Jennings G.T., Bachmann M.F. The coming of age of virus-like particle vaccines. Biol. Chem. 2008;389(5):521–536. doi: 10.1515/bc.2008.064. [DOI] [PubMed] [Google Scholar]
- 57.Xiang S.D. Pathogen recognition and development of particulate vaccines: does size matter? Methods. 2006;40(1):1–9. doi: 10.1016/j.ymeth.2006.05.016. [DOI] [PubMed] [Google Scholar]
- 58.Deml L. Recombinant HIV-1 Pr55gag virus-like particles: potent stimulators of innate and acquired immune responses. Mol. Immunol. 2005;42(2):259–277. doi: 10.1016/j.molimm.2004.06.028. [DOI] [PubMed] [Google Scholar]
- 59.Zahin M. Scalable production of HPV16 L1 protein and VLPs from tobacco leaves. PLoS One. 2016;11(8):e0160995. doi: 10.1371/journal.pone.0160995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Yazdani R. Production and characterization of virus-like particles of grapevine fanleaf virus presenting L2 epitope of human papillomavirus minor capsid protein. BMC Biotechnol. 2019;19(1):81. doi: 10.1186/s12896-019-0566-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Bayer M.E., Blumberg B.S., Werner B. Particles associated with Australia antigen in the sera of patients with leukaemia, Down’s Syndrome and hepatitis. Nature. 1968;218(5146):1057–1059. doi: 10.1038/2181057a0. [DOI] [PubMed] [Google Scholar]
- 62.Cohen B.J., Richmond J.E. Electron microscopy of hepatitis B core antigen synthesized in E. coli. Nature. 1982;296(5858):677–679. doi: 10.1038/296677a0. [DOI] [PubMed] [Google Scholar]
- 63.Stahl S. Hepatitis B virus core antigen: synthesis in Escherichia coli and application in diagnosis. Proc. Natl. Acad. Sci. U. S. A. 1982;79(5):1606–1610. doi: 10.1073/pnas.79.5.1606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Zhou S., Standring D.N. Hepatitis B virus capsid particles are assembled from core-protein dimer precursors. Proc Natl Acad Sci U S A. 1992;89(21):10046–10050. doi: 10.1073/pnas.89.21.10046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Strasser R. Generation of glyco‐engineered Nicotiana benthamiana for the production of monoclonal antibodies with a homogeneous human‐like N‐glycan structure. Plant Biotechnol. J. 2008;6(4):392–402. doi: 10.1111/j.1467-7652.2008.00330.x. [DOI] [PubMed] [Google Scholar]
- 66.Borisova G. Recombinant capsid structures for exposure of protein antigenic epitopes. Mol. Gen.(Life Sci. Adv.) 1987;6:169–174. [Google Scholar]
- 67.Clarke B. Improved immunogenicity of a peptide epitope after fusion to hepatitis B core protein. Nature. 1987;330(6146):381–384. doi: 10.1038/330381a0. [DOI] [PubMed] [Google Scholar]
- 68.Pang E.L. Epitope presentation of dengue viral envelope glycoprotein domain III on hepatitis B core protein virus-like particles produced in Nicotiana benthamiana. Front. Plant Sci. 2019;10:455. doi: 10.3389/fpls.2019.00455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Whitacre D.C., Lee B.O., Milich D.R. Use of hepadnavirus core proteins as vaccine platforms. Expert Rev. Vaccines. 2009;8(11):1565–1573. doi: 10.1586/erv.09.121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Michel M.L. Synthesis in animal cells of hepatitis B surface antigen particles carrying a receptor for polymerized human serum albumin. Proc. Natl. Acad. Sci. U. S. A. 1984;81(24):7708–7712. doi: 10.1073/pnas.81.24.7708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Harford N. Expression of hepatitis B surface antigen in yeast. Dev. Biol. Stand. 1983;54:125–130. [PubMed] [Google Scholar]
- 72.Hirschman S.Z., Garfinkel E., Sugrue S. Expression of hepatitis B viral antigens in animal cells transfected with viral DNA. Trans. Assoc. Am. Physicians. 1982;95:53–62. [PubMed] [Google Scholar]
- 73.Wingfield P.T. Hepatitis core antigen produced in Escherichia coli: subunit composition, conformational analysis, and in vitro capsid assembly. Biochemistry. 1995;34(15):4919–4932. doi: 10.1021/bi00015a003. [DOI] [PubMed] [Google Scholar]
- 74.Burrell C.J. Expression in Escherichia coli of hepatitis B virus DNA sequences cloned in plasmid pBR322. Nature. 1979;279(5708):43–47. doi: 10.1038/279043a0. [DOI] [PubMed] [Google Scholar]
- 75.Clarke B.E. Improved immunogenicity of a peptide epitope after fusion to hepatitis B core protein. Nature. 1987;330(6146):381–384. doi: 10.1038/330381a0. [DOI] [PubMed] [Google Scholar]
- 76.Mechtcheriakova I. The use of viral vectors to produce hepatitis B virus core particles in plants. J. Virol. Methods. 2006;131(1):10–15. doi: 10.1016/j.jviromet.2005.06.020. [DOI] [PubMed] [Google Scholar]
- 77.Pumpens P., Grens E. HBV core particles as a carrier for B cell/T cell epitopes. Intervirology. 2001;44(2-3):98–114. doi: 10.1159/000050037. [DOI] [PubMed] [Google Scholar]
- 78.Walker A., Skamel C., Nassal M. SplitCore: an exceptionally versatile viral nanoparticle for native whole protein display regardless of 3D structure. Sci. Rep. 2011;1:5. doi: 10.1038/srep00005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Kniskem P.J. Unusually high-level expression of a foreign gene (hepatitis B virus core antigen) in Saccharomyces cerevisiae. Gene. 1986;46(1):135–141. doi: 10.1016/0378-1119(86)90177-0. [DOI] [PubMed] [Google Scholar]
- 80.Rolland D. Purification of recombinant HBc antigen expressed in Escherichia coli and Pichia pastoris: comparison of size-exclusion chromatography and ultracentrifugation. J. Chromatogr. B Biomed. Sci. Appl. 2001;753(1):51–65. doi: 10.1016/s0378-4347(00)00538-7. [DOI] [PubMed] [Google Scholar]
- 81.Freivalds J. Highly efficient production of phosphorylated hepatitis B core particles in yeast Pichia pastoris. Protein Expr. Purif. 2011;75(2):218–224. doi: 10.1016/j.pep.2010.09.010. [DOI] [PubMed] [Google Scholar]
- 82.Miyanohara A. Expression of hepatitis B virus core antigen gene in Saccharomyces cerevisiae: synthesis of two polypeptides translated from different initiation codons. J. Virol. 1986;59(1):176–180. doi: 10.1128/jvi.59.1.176-180.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Hilditch C.M., Rogers L.J., Bishop D.H. Physicochemical analysis of the hepatitis B virus core antigen produced by a baculovirus expression vector. J. Gen. Virol. 1990;71(Pt 11):2755–2759. doi: 10.1099/0022-1317-71-11-2755. [DOI] [PubMed] [Google Scholar]
- 84.Tsuda S. Application of the human hepatitis B virus core antigen from transgenic tobacco plants for serological diagnosis. Vox Sang. 1998;74(3):148–155. [PubMed] [Google Scholar]
- 85.Kushnir N., Streatfield S.J., Yusibov V. Virus-like particles as a highly efficient vaccine platform: diversity of targets and production systems and advances in clinical development. Vaccine. 2012;31(1):58–83. doi: 10.1016/j.vaccine.2012.10.083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Peyret H. Tandem fusion of hepatitis B core antigen allows assembly of virus-like particles in bacteria and plants with enhanced capacity to accommodate foreign proteins. PLoS One. 2015;10(4) doi: 10.1371/journal.pone.0120751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Wynne S.A., Crowther R.A., Leslie A.G. The crystal structure of the human hepatitis B virus capsid. Mol. Cell. 1999;3(6):771–780. doi: 10.1016/s1097-2765(01)80009-5. [DOI] [PubMed] [Google Scholar]
- 88.Porterfield J.Z. Full-length hepatitis B virus core protein packages viral and heterologous RNA with similarly high levels of cooperativity. J. Virol. 2010;84(14):7174–7184. doi: 10.1128/JVI.00586-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Gallina A. A recombinant hepatitis B core antigen polypeptide with the protamine-like domain deleted self-assembles into capsid particles but fails to bind nucleic acids. J. Virol. 1989;63(11):4645–4652. doi: 10.1128/jvi.63.11.4645-4652.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Stahl S.J., Murray K. Immunogenicity of peptide fusions to hepatitis B virus core antigen. Proc. Natl. Acad. Sci. 1989;86(16):6283–6287. doi: 10.1073/pnas.86.16.6283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Brown A. Foreign epitopes in immunodominant regions of hepatitis B core particles are highly immunogenic and conformationally restricted. Vaccine. 1991;9(8):595–601. doi: 10.1016/0264-410x(91)90248-5. [DOI] [PubMed] [Google Scholar]
- 92.Schödel F. The position of heterologous epitopes inserted in hepatitis B virus core particles determines their immunogenicity. J. Virol. 1992;66(1):106–114. doi: 10.1128/jvi.66.1.106-114.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Crowther R. Three-dimensional structure of hepatitis B virus core particles determined by electron cryomicroscopy. Cell. 1994;77(6):943–950. doi: 10.1016/0092-8674(94)90142-2. [DOI] [PubMed] [Google Scholar]
- 94.Vogel M. In vitro assembly of mosaic hepatitis B virus capsid-like particles (CLPs): rescue into CLPs of assembly-deficient core protein fusions and FRET-suited CLPs. FEBS Lett. 2005;579(23):5211–5216. doi: 10.1016/j.febslet.2005.08.044. [DOI] [PubMed] [Google Scholar]
- 95.Chisari F.V., Ferrari C. Hepatitis B virus immunopathogenesis. Annu. Rev. Immunol. 1995;13:29–60. doi: 10.1146/annurev.iy.13.040195.000333. [DOI] [PubMed] [Google Scholar]
- 96.Francis M.J. Immunological properties of hepatitis B core antigen fusion proteins. Proc. Natl. Acad. Sci. U. S. A. 1990;87(7):2545–2549. doi: 10.1073/pnas.87.7.2545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Cooper A. Cytokine induction by the hepatitis B virus capsid in macrophages is facilitated by membrane heparan sulfate and involves TLR2. J. Immunol. 2005;175(5):3165–3176. doi: 10.4049/jimmunol.175.5.3165. [DOI] [PubMed] [Google Scholar]
- 98.Milich D.R., McLachlan A. The nucleocapsid of hepatitis B virus is both a T-cell-independent and a T-cell-dependent antigen. Science. 1986;234(4782):1398–1401. doi: 10.1126/science.3491425. [DOI] [PubMed] [Google Scholar]
- 99.Mason H.S., Lam D.M., Arntzen C.J. Expression of hepatitis B surface antigen in transgenic plants. Proc. Natl. Acad. Sci. U. S. A. 1992;89(24):11745–11749. doi: 10.1073/pnas.89.24.11745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Riedl P. Priming Th1 immunity to viral core particles is facilitated by trace amounts of RNA bound to its arginine-rich domain. J. Immunol. 2002;168(10):4951–4959. doi: 10.4049/jimmunol.168.10.4951. [DOI] [PubMed] [Google Scholar]
- 101.Lee B.O. Interaction of the hepatitis B core antigen and the innate immune system. J. Immunol. 2009;182(11):6670–6681. doi: 10.4049/jimmunol.0803683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Milich D.R. Antibody production to the nucleocapsid and envelope of the hepatitis B virus primed by a single synthetic T cell site. Nature. 1987;329(6139):547–549. doi: 10.1038/329547a0. [DOI] [PubMed] [Google Scholar]
- 103.Pumpens P. Hepatitis B virus core particles as epitope carriers. Intervirology. 1995;38(1-2):63–74. doi: 10.1159/000150415. [DOI] [PubMed] [Google Scholar]
- 104.Schödel F. Hybrid hepatitis B virus core antigen as a vaccine carrier moiety: I. Presentation of foreign epitopes. J. Biotechnol. 1996;44(1-3):91–96. doi: 10.1016/0168-1656(95)00118-2. [DOI] [PubMed] [Google Scholar]
- 105.Ghorbani A. Development of a novel platform of virus-like particle (VLP) based vaccine against coronavirus 2019 (SARS-CoV-2) by exposing of epitopes: an immunoinformatics approach. New Microbes New Infect. 2020:100786. doi: 10.1016/j.nmni.2020.100786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Li H.-Y. Virus-like particles for enterovirus 71 produced from Saccharomyces cerevisiae potently elicits protective immune responses in mice. Vaccine. 2013;31(32):3281–3287. doi: 10.1016/j.vaccine.2013.05.019. [DOI] [PubMed] [Google Scholar]
- 107.Lua L.H. Bioengineering virus-like particles as vaccines. Biotechnol. Bioeng. 2014;111(3):425–440. doi: 10.1002/bit.25159. [DOI] [PubMed] [Google Scholar]
- 108.Stephenne J. Production in yeast versus mammalian cells of the first recombinant DNA human vaccine and its proved safety, efficacy, and economy: hepatitis B vaccine. Adv. Biotechnol. Processes. 1990;14:279. [PubMed] [Google Scholar]
- 109.Hwang D.J., Roberts I.M., Wilson T.M. Expression of tobacco mosaic virus coat protein and assembly of pseudovirus particles in Escherichia coli. Proc. Natl. Acad. Sci. U. S. A. 1994;91(19):9067–9071. doi: 10.1073/pnas.91.19.9067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Zhao Q. Disassembly and reassembly improves morphology and thermal stability of human papillomavirus type 16 virus-like particles. Nanomedicine. 2012;8(7):1182–1189. doi: 10.1016/j.nano.2012.01.007. [DOI] [PubMed] [Google Scholar]
- 111.Edman J.C. Synthesis of hepatitis B surface and core antigens in E. coli. Nature. 1981;291(5815):503–506. doi: 10.1038/291503a0. [DOI] [PubMed] [Google Scholar]
- 112.Michel M.L., Tiollais P. Hepatitis B vaccines: protective efficacy and therapeutic potential. Pathol Biol (Paris) 2010;58(4):288–295. doi: 10.1016/j.patbio.2010.01.006. [DOI] [PubMed] [Google Scholar]
- 113.Rodríguez-Limas W.A. Molecular and process design for rotavirus-like particle production in Saccharomyces cerevisiae. Microb. Cell Fact. 2011;10:33. doi: 10.1186/1475-2859-10-33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Y, K, P. P . CRC Press, Taylor and Francis Group; USA: 2016. Viral Nanotechnology; p. 2016. [Google Scholar]
- 115.Lua L.H. Bioengineering virus‐like particles as vaccines. Biotechnol. Bioeng. 2014;111(3):425–440. doi: 10.1002/bit.25159. [DOI] [PubMed] [Google Scholar]
- 116.Sakuragi S. HIV type 1 Gag virus-like particle budding from spheroplasts of Saccharomyces cerevisiae. Proc. Natl. Acad. Sci. U. S. A. 2002;99(12):7956–7961. doi: 10.1073/pnas.082281199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Ma J.K.C. Molecular farming for new drugs and vaccines. EMBO Rep. 2005;6(7):593–599. doi: 10.1038/sj.embor.7400470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Loh H.-S., Green B.J., Yusibov V. Using transgenic plants and modified plant viruses for the development of treatments for human diseases. Curr. Opin. Virol. 2017;26:81–89. doi: 10.1016/j.coviro.2017.07.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Wildt S., Gerngross T.U. The humanization of N-glycosylation pathways in yeast. Nat. Rev. Microbiol. 2005;3(2):119–128. doi: 10.1038/nrmicro1087. [DOI] [PubMed] [Google Scholar]
- 120.Fernandes F. Insect cells as a production platform of complex virus-like particles. Expert Rev. Vaccines. 2013;12(2):225–236. doi: 10.1586/erv.12.153. [DOI] [PubMed] [Google Scholar]
- 121.Palomares L.A., Ramírez O.T. Challenges for the production of virus-like particles in insect cells: the case of rotavirus-like particles. Biochem. Eng. J. 2009;45(3):158–167. [Google Scholar]
- 122.Pushko P. Influenza virus-like particles comprised of the HA, NA, and M1 proteins of H9N2 influenza virus induce protective immune responses in BALB/c mice. Vaccine. 2005;23(50):5751–5759. doi: 10.1016/j.vaccine.2005.07.098. [DOI] [PubMed] [Google Scholar]
- 123.Buonaguro L. Baculovirus-derived human immunodeficiency virus type 1 virus-like particles activate dendritic cells and induce ex vivo T-cell responses. J. Virol. 2006;80(18):9134–9143. doi: 10.1128/JVI.00050-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Streatfield S.J. Plant-based vaccines for animal health. Rev. Sci. Tech. 2005;24(1):189–199. [PubMed] [Google Scholar]
- 125.Koya V. Plant-based vaccine: mice immunized with chloroplast-derived anthrax protective antigen survive anthrax lethal toxin challenge. Infect. Immun. 2005;73(12):8266–8274. doi: 10.1128/IAI.73.12.8266-8274.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Fischer R., Twyman R.M., Schillberg S. Production of antibodies in plants and their use for global health. Vaccine. 2003;21(7-8):820–825. doi: 10.1016/s0264-410x(02)00607-2. [DOI] [PubMed] [Google Scholar]
- 127.Whitelam G.C., Cockburn W., Owen M.R. Antibody production in transgenic plants. Biochem. Soc. Trans. 1994;22(4):940–944. doi: 10.1042/bst0220940. [DOI] [PubMed] [Google Scholar]
- 128.Daniell H., Streatfield S.J., Wycoff K. Medical molecular farming: production of antibodies, biopharmaceuticals and edible vaccines in plants. Trends Plant Sci. 2001;6(5):219–226. doi: 10.1016/S1360-1385(01)01922-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Chen Q. Expression and purification of pharmaceutical proteins in plants. Biol. Eng. Trans. 2008;1(4):291–321. [Google Scholar]
- 130.Nandi S. MAbs. Taylor & Francis; 2016. Techno-economic analysis of a transient plant-based platform for monoclonal antibody production. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Tusé D., Tu T., McDonald K.A. Manufacturing economics of plant-made biologics: case studies in therapeutic and industrial enzymes. Biomed Res. Int. 2014;2014:256135. doi: 10.1155/2014/256135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Shanmugaraj B., Ramalingam S. Plant expression platform for the production of recombinant pharmaceutical proteins. Austin J. Biotechnol. Bioeng. 2014;1(6):4. [Google Scholar]
- 133.Chen Q., Davis K.R. The potential of plants as a system for the development and production of human biologics. F1000Res. 2016;5 doi: 10.12688/f1000research.8010.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Hiatt A., Cafferkey R., Bowdish K. Production of antibodies in transgenic plants. Nature. 1989;342(6245):76–78. doi: 10.1038/342076a0. [DOI] [PubMed] [Google Scholar]
- 135.Scotti N., Rybicki E.P. Virus-like particles produced in plants as potential vaccines. Expert Rev. Vaccines. 2013;12(2):211–224. doi: 10.1586/erv.12.147. [DOI] [PubMed] [Google Scholar]
- 136.Joung Y.H. The Last Ten Years of Advancements in Plant-Derived Recombinant Vaccines against Hepatitis B. Int. J. Mol. Sci. 2016;17(10) doi: 10.3390/ijms17101715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.D’Aoust M.A. Influenza virus‐like particles produced by transient expression in Nicotiana benthamiana induce a protective immune response against a lethal viral challenge in mice. Plant Biotechnol. J. 2008;6(9):930–940. doi: 10.1111/j.1467-7652.2008.00384.x. [DOI] [PubMed] [Google Scholar]
- 138.Huang Y. Immunogenicity of the epitope of the foot-and-mouth disease virus fused with a hepatitis B core protein as expressed in transgenic tobacco. Viral Immunol. 2005;18(4):668–677. doi: 10.1089/vim.2005.18.668. [DOI] [PubMed] [Google Scholar]
- 139.Alemzadeh E. Plant virus nanoparticles: novel and robust nanocarriers for drug delivery and imaging. Colloids Surf. B Biointerfaces. 2018;167:20–27. doi: 10.1016/j.colsurfb.2018.03.026. [DOI] [PubMed] [Google Scholar]
- 140.Le Mauff F. Biochemical composition of haemagglutinin‐based influenza virus‐like particle vaccine produced by transient expression in tobacco plants. Plant Biotechnol. J. 2015;13(5):717–725. doi: 10.1111/pbi.12301. [DOI] [PubMed] [Google Scholar]
- 141.Landry N. Influenza virus-like particle vaccines made in Nicotiana benthamiana elicit durable, poly-functional and cross-reactive T cell responses to influenza HA antigens. Clin. Immunol. 2014;154(2):164–177. doi: 10.1016/j.clim.2014.08.003. [DOI] [PubMed] [Google Scholar]
- 142.Pillet S. Humoral and cell-mediated immune responses to H5N1 plant-made virus-like particle vaccine are differentially impacted by alum and GLA-SE adjuvants in a Phase 2 clinical trial. NPJ Vaccines. 2018;3(1):1–9. doi: 10.1038/s41541-017-0043-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Capell T. Potential applications of plant biotechnology against SARS-CoV-2. Trends Plant Sci. 2020 doi: 10.1016/j.tplants.2020.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Ma J.K., Drake P.M., Christou P. The production of recombinant pharmaceutical proteins in plants. Nat. Rev. Genet. 2003;4(10):794–805. doi: 10.1038/nrg1177. [DOI] [PubMed] [Google Scholar]
- 145.Twyman R.M. Molecular farming in plants: host systems and expression technology. Trends Biotechnol. 2003;21(12):570–578. doi: 10.1016/j.tibtech.2003.10.002. [DOI] [PubMed] [Google Scholar]
- 146.Lico C., Chen Q., Santi L. Viral vectors for production of recombinant proteins in plants. J. Cell. Physiol. 2008;216(2):366–377. doi: 10.1002/jcp.21423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.P, J Plantibody: an overview. Asian J. Pharm. Sci. 2011;1(1):87–94. [Google Scholar]
- 148.Yao J. Plants as factories for human pharmaceuticals: applications and challenges. Int. J. Mol. Sci. 2015;16(12):28549–28565. doi: 10.3390/ijms161226122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Marsian J., Lomonossoff G.P. Molecular pharming - VLPs made in plants. Curr. Opin. Biotechnol. 2016;37:201–206. doi: 10.1016/j.copbio.2015.12.007. [DOI] [PubMed] [Google Scholar]
- 150.Huang Z. Virus-like particle expression and assembly in plants: hepatitis B and Norwalk viruses. Vaccine. 2005;23(15):1851–1858. doi: 10.1016/j.vaccine.2004.11.017. [DOI] [PubMed] [Google Scholar]
- 151.Guerrero-Andrade O. Expression of the Newcastle disease virus fusion protein in transgenic maize and immunological studies. Transgenic Res. 2006;15(4):455–463. doi: 10.1007/s11248-006-0017-0. [DOI] [PubMed] [Google Scholar]
- 152.Jiang X.L. Cholera toxin B protein in transgenic tomato fruit induces systemic immune response in mice. Transgenic Res. 2007;16(2):169–175. doi: 10.1007/s11248-006-9023-5. [DOI] [PubMed] [Google Scholar]
- 153.Goodin M.M. Nicotiana benthamiana: its history and future as a model for plant-pathogen interactions. Molecular Plant-Microbe Interactions: MPMI. 2015;2015(1):28–39. doi: 10.1094/MPMI-00-00-1015-REV.testissue. [DOI] [PubMed] [Google Scholar]
- 154.Moffat A.S. Exploring transgenic plants as a new vaccine source. Science. 1995;268(5211):658. doi: 10.1126/science.7732373. 660. [DOI] [PubMed] [Google Scholar]
- 155.Holler C., Zhang C. Purification of an acidic recombinant protein from transgenic tobacco. Biotechnol. Bioeng. 2008;99(4):902–909. doi: 10.1002/bit.21638. [DOI] [PubMed] [Google Scholar]
- 156.Mason H.S., Arntzen C.J. Transgenic plants as vaccine production systems. Trends Biotechnol. 1995;13(9):388–392. doi: 10.1016/S0167-7799(00)88986-6. [DOI] [PubMed] [Google Scholar]
- 157.Oluwayelu D.O., Adebiyi A.I. Plantibodies in human and animal health: a review. Afr. Health Sci. 2016;16(2):640–645. doi: 10.4314/ahs.v16i2.35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Smith M.D., Glick B.R. Elsevier; 2000. The Production of Antibodies in Plants: an Idea Whose Time Has Come? [DOI] [PubMed] [Google Scholar]
- 159.Pastores G.M. A Phase 3, multicenter, open-label, switchover trial to assess the safety and efficacy of taliglucerase alfa, a plant cell-expressed recombinant human glucocerebrosidase, in adult and pediatric patients with Gaucher disease previously treated with imiglucerase. Blood Cells Mol. Dis. 2014;53(4):253–260. doi: 10.1016/j.bcmd.2014.05.004. [DOI] [PubMed] [Google Scholar]
- 160.Oey M. Exhaustion of the chloroplast protein synthesis capacity by massive expression of a highly stable protein antibiotic. Plant J. 2009;57(3):436–445. doi: 10.1111/j.1365-313X.2008.03702.x. [DOI] [PubMed] [Google Scholar]
- 161.Job D. Plant biotechnology in agriculture. Biochimie. 2002;84(11):1105–1110. doi: 10.1016/s0300-9084(02)00013-5. [DOI] [PubMed] [Google Scholar]
- 162.Guo J. Immunogenicity of a virus-like-Particle vaccine containing multiple antigenic epitopes of toxoplasma gondii against acute and chronic toxoplasmosis in mice. Front. Immunol. 2019;10:592. doi: 10.3389/fimmu.2019.00592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Bárcena J., Blanco E. Design of novel vaccines based on virus-like particles or chimeric virions. Subcell. Biochem. 2013;68:631–665. doi: 10.1007/978-94-007-6552-8_21. [DOI] [PubMed] [Google Scholar]
- 164.Monie A. Cervarix: a vaccine for the prevention of HPV 16, 18-associated cervical cancer. Biologics. 2008;2(1):97–105. [PMC free article] [PubMed] [Google Scholar]
- 165.Kash N. Safety and efficacy data on vaccines and immunization to human papillomavirus. J. Clin. Med. 2015;4(4):614–633. doi: 10.3390/jcm4040614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.McKeage K., Romanowski B. AS04-Adjuvanted Human Papillomavirus (HPV) types 16 and 18 vaccine (Cervarix®) Drugs. 2011;71(4):465–488. doi: 10.2165/11206820-000000000-00000. [DOI] [PubMed] [Google Scholar]
- 167.Soulié J.C. Immunogenicity and safety in newborns of a new recombinant hepatitis B vaccine containing the S and pre-S2 antigens. Vaccine. 1991;9(8):545–548. doi: 10.1016/0264-410x(91)90240-7. [DOI] [PubMed] [Google Scholar]
- 168.Ltd, S., Sci-B-VacTM. www.scigenltd.com/productsscibvacs.htm. 2012.
- 169.Farma, B., Company Profile 2011. http://biofarma.co.id/tlfiles/data/compro.pdf [accessed 12 April, 2012]. 2011.
- 170.Hauser P. Immunological properties of recombinant HBsAg produced in yeast. Postgrad. Med. J. 1987;63(Suppl 2):83–91. [PubMed] [Google Scholar]
- 171.B®, E., Sanofi Pasteur Ltd. Euvax B®. Summary of product characteristics. http://drug.fda.moph.go.th/zonesearch/files/EUVAX%20B1C%2022341.pdf [accessed 12 April, 2012]. 2012.
- 172.Vac-B®, G., Serum Institute of India Ltd. Gene Vac-B®. http://www.seruminstitute.com/content/products/productgenevac.htm [accessed 12 April, 2012]. 2012.
- 173.Hepavax-Gene®, Diethelm Ltd./Berna Biotech Korea Corp. Hepavax-Gene®. Summary of product characteristics.http://drug.fda.moph.go.th/zonesearch/files/Hepavax1C%202249.pdf[accessed 12 April, 2012]. 2012.
- 174.HB® R. Vol. 2011. Merck; 2011. (Prescribing Information). July. [Google Scholar]
- 175.Ellis R.W., Gerety R.J. Plasma-derived and yeast-derived hepatitis B vaccines. Am. J. Infect. Control. 1989;17(3):181–189. doi: 10.1016/0196-6553(89)90214-9. [DOI] [PubMed] [Google Scholar]
- 176.Revac-B+TM, Bharat Biotech. Revac-B+TM. http://www.bharatbiotech.com/prevac-b.htm. 2008.
- 177.Shanvac®-B, Shantha Biotechnics Ltd. Shanvac®-B. http://www.shanthabiotech.com/files/Shanvac%20B%20Domestic%20Pack%20insert.pdf [accessed 12 April, 2012]. 2012.
- 178.Zhang X. Robust manufacturing and comprehensive characterization of recombinant hepatitis E virus-like particles in Hecolin(®) Vaccine. 2014;32(32):4039–4050. doi: 10.1016/j.vaccine.2014.05.064. [DOI] [PubMed] [Google Scholar]
- 179.Sun Y. Protection against lethal challenge by Ebola virus-like particles produced in insect cells. Virology. 2009;383(1):12–21. doi: 10.1016/j.virol.2008.09.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Vicente T. Large-scale production and purification of VLP-based vaccines. J. Invertebr. Pathol. 2011;107(Suppl):S42–8. doi: 10.1016/j.jip.2011.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Krammer F. Trichoplusia ni cells (High Five) are highly efficient for the production of influenza A virus-like particles: a comparison of two insect cell lines as production platforms for influenza vaccines. Mol. Biotechnol. 2010;45(3):226–234. doi: 10.1007/s12033-010-9268-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Estes M.K. Norwalk virus vaccines: challenges and progress. J. Infect. Dis. 2000;181(Suppl 2):S367–73. doi: 10.1086/315579. [DOI] [PubMed] [Google Scholar]
- 183.Jiang X. Expression, self-assembly, and antigenicity of the Norwalk virus capsid protein. J. Virol. 1992;66(11):6527–6532. doi: 10.1128/jvi.66.11.6527-6532.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Yusibov V. Peptide-based candidate vaccine against respiratory syncytial virus. Vaccine. 2005;23(17-18):2261–2265. doi: 10.1016/j.vaccine.2005.01.039. [DOI] [PubMed] [Google Scholar]
- 185.Bertolotti-Ciarlet A. Immunogenicity and protective efficacy of rotavirus 2/6-virus-like particles produced by a dual baculovirus expression vector and administered intramuscularly, intranasally, or orally to mice. Vaccine. 2003;21(25-26):3885–3900. doi: 10.1016/s0264-410x(03)00308-6. [DOI] [PubMed] [Google Scholar]
- 186.Ward B. 2020. Phase 1 Trial of a Candidate Recombinant Virus-like Particle Vaccine for Covid-19 Disease Produced in Plants. [Google Scholar]
- 187.Ward B.J. Efficacy, immunogenicity, and safety of a plant-derived, quadrivalent, virus-like particle influenza vaccine in adults (18–64 years) and older adults (≥65 years): two multicentre, randomised phase 3 trials. Lancet. 2020;396(10261):1491–1503. doi: 10.1016/S0140-6736(20)32014-6. [DOI] [PubMed] [Google Scholar]
- 188.Ramirez A. A virus-like particle vaccine candidate for influenza A virus based on multiple conserved antigens presented on hepatitis B tandem core particles. Vaccine. 2018;36(6):873–880. doi: 10.1016/j.vaccine.2017.12.053. [DOI] [PubMed] [Google Scholar]
- 189.Zheng D. Influenza H7N9 LAH-HBc virus-like particle vaccine with adjuvant protects mice against homologous and heterologous influenza viruses. Vaccine. 2016;34(51):6464–6471. doi: 10.1016/j.vaccine.2016.11.026. [DOI] [PubMed] [Google Scholar]
- 190.Dhanasooraj D., Kumar R.A., Mundayoor S. Vaccine Design. Springer; 2016. Subunit protein vaccine delivery system for tuberculosis based on hepatitis B virus core VLP (HBc-VLP) particles; pp. 377–392. [DOI] [PubMed] [Google Scholar]
- 191.Blokhina E.A. A molecular assembly system for presentation of antigens on the surface of HBc virus-like particles. Virology. 2013;435(2):293–300. doi: 10.1016/j.virol.2012.09.014. [DOI] [PubMed] [Google Scholar]
- 192.Ravin N. Plant-produced recombinant influenza vaccine based on virus-like HBc particles carrying an extracellular domain of M2 protein. Biochemistry (Moscow) 2012;77(1):33–40. doi: 10.1134/S000629791201004X. [DOI] [PubMed] [Google Scholar]
- 193.Arora U. Chimeric Hepatitis B core antigen virus-like particles displaying the envelope domain III of dengue virus type 2. J. Nanobiotechnology. 2012;10(1):30. doi: 10.1186/1477-3155-10-30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Sominskaya I. Construction and immunological evaluation of multivalent hepatitis B virus (HBV) core virus-like particles carrying HBV and HCV epitopes. Clin. Vaccine Immunol. 2010;17(6):1027–1033. doi: 10.1128/CVI.00468-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.De Filette M. Universal influenza A M2e-HBc vaccine protects against disease even in the presence of pre-existing anti-HBc antibodies. Vaccine. 2008;26(51):6503–6507. doi: 10.1016/j.vaccine.2008.09.038. [DOI] [PubMed] [Google Scholar]
- 196.Ding F.X. Multiepitope peptide‐loaded virus‐like particles as a vaccine against hepatitis B virus–related hepatocellular carcinoma. Hepatology. 2009;49(5):1492–1502. doi: 10.1002/hep.22816. [DOI] [PubMed] [Google Scholar]
- 197.Zhang Y. Generation of chimeric HBc proteins with epitopes in E. coli: formation of virus-like particles and a potent inducer of antigen-specific cytotoxic immune response and anti-tumor effect in vivo. Cell. Immunol. 2007;247(1):18–27. doi: 10.1016/j.cellimm.2007.07.003. [DOI] [PubMed] [Google Scholar]
- 198.Chen J.-Y., Li F. Development of hepatitis C virus vaccine using hepatitis B core antigen as immuno-carrier. World J. Gastroenterol. WJG. 2006;12(48):7774. doi: 10.3748/wjg.v12.i48.7774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Huang Y. Immunogenicity of the epitope of the foot-and-mouth disease virus fused with a hepatitis B core protein as expressed in transgenic tobacco. Viral Immunol. 2005;18(4):668–677. doi: 10.1089/vim.2005.18.668. [DOI] [PubMed] [Google Scholar]
- 200.Zhang Y.L. Enhanced immunogenicity of modified hepatitis B virus core particle fused with multiepitopes of foot‐and‐mouth disease virus. Scand. J. Immunol. 2007;65(4):320–328. doi: 10.1111/j.1365-3083.2007.01900.x. [DOI] [PubMed] [Google Scholar]
- 201.Yang H.-J. Expression and immunoactivity of chimeric particulate antigens of receptor binding site-core antigen of hepatitis B virus. World J. Gastroenterol. WJG. 2005;11(4):492. doi: 10.3748/wjg.v11.i4.492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Geldmacher A. A hantavirus nucleocapsid protein segment exposed on hepatitis B virus core particles is highly immunogenic in mice when applied without adjuvants or in the presence of pre-existing anti-core antibodies. Vaccine. 2005;23(30):3973–3983. doi: 10.1016/j.vaccine.2005.02.025. [DOI] [PubMed] [Google Scholar]
- 203.Chen X. Recombinant hepatitis B core antigen carrying preS1 epitopes induce immune response against chronic HBV infection. Vaccine. 2004;22(3-4):439–446. doi: 10.1016/j.vaccine.2003.07.014. [DOI] [PubMed] [Google Scholar]
- 204.Pumpens P. Evaluation of HBs, HBc, and frCP virus-like particles for expression of human papillomavirus 16 E7 oncoprotein epitopes. Intervirology. 2002;45(1):24–32. doi: 10.1159/000050084. [DOI] [PubMed] [Google Scholar]
- 205.Boulter N.R. Theileria annulata sporozoite antigen fused to hepatitis B core antigen used in a vaccination trial. Vaccine. 1995;13(13):1152–1160. doi: 10.1016/0264-410x(95)00026-w. [DOI] [PubMed] [Google Scholar]
- 206.Ulrich R. Immunogenicity of recombinant core particles of hepatitis B virus containing epitopes of human immunodeficiency virus 1 core antigen. Arch. Virol. 1992;126(1-4):321–328. doi: 10.1007/BF01309705. [DOI] [PubMed] [Google Scholar]
- 207.Thuenemann E.C. A method for rapid production of heteromultimeric protein complexes in plants: assembly of protective bluetongue virus-like particles. Plant Biotechnol. J. 2013;11(7):839–846. doi: 10.1111/pbi.12076. [DOI] [PubMed] [Google Scholar]
- 208.Veerapen V.P. Novel expression of immunogenic foot-and-mouth disease virus-like particles in Nicotiana benthamiana. Virus Res. 2018;244:213–217. doi: 10.1016/j.virusres.2017.11.027. [DOI] [PubMed] [Google Scholar]
- 209.Thanavala Y. Immunogenicity in humans of an edible vaccine for hepatitis B. Proc. Natl. Acad. Sci. U. S. A. 2005;102(9):3378–3382. doi: 10.1073/pnas.0409899102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Kapusta J. Oral immunization of human with transgenic lettuce expressing hepatitis B surface antigen. Adv. Exp. Med. Biol. 2001;495:299–303. doi: 10.1007/978-1-4615-0685-0_41. [DOI] [PubMed] [Google Scholar]
- 211.Kapusta J. A plant-derived edible vaccine against hepatitis B virus. FASEB J. 1999;13(13):1796–1799. doi: 10.1096/fasebj.13.13.1796. [DOI] [PubMed] [Google Scholar]
- 212.Page, U.S.N.I.o.H.C.T.H., Available online: https://clinicaltrials.gov.
- 213.Scotti N. High-level expression of the HIV-1 Pr55gag polyprotein in transgenic tobacco chloroplasts. Planta. 2009;229(5):1109–1122. doi: 10.1007/s00425-009-0898-2. [DOI] [PubMed] [Google Scholar]
- 214.Shchelkunov S.N. Immunogenicity of a novel, bivalent, plant-based oral vaccine against hepatitis B and human immunodeficiency viruses. Biotechnol. Lett. 2006;28(13):959–967. doi: 10.1007/s10529-006-9028-4. [DOI] [PubMed] [Google Scholar]
- 215.Varsani A. Expression of Human papillomavirus type 16 major capsid protein in transgenic Nicotiana tabacum cv. Xanthi. Arch. Virol. 2003;148(9):1771–1786. doi: 10.1007/s00705-003-0119-4. [DOI] [PubMed] [Google Scholar]
- 216.Potera C. Vaccine manufacturing gets boost from tobacco plants: canada-based medicago opens US Facility to exploit its influenza vaccine production method. Genet. Eng. Biotechnol. News. 2012;32(6):8–10. [Google Scholar]
- 217.Medicago Clinical trials update: avian flu vaccine. Genet. Eng. Biotechnol. News. 2011;31(61):2011. [Google Scholar]
- 218.Medicago, Medicago reports positive U.S. clinical trial results for its H1N1 / seasonal influenza vaccine http://wwwmedicagocom/English/news/News-ReleasesNews-ReleaseDetails/2011/Medicago-reports-positive-US-clinical-trial-results-for-its-H1N1–seasonal-influenza-vaccine1125593/defaultaspx 2011 ; (accessed on July 04 2012) Prnewswire, 2011.
- 219.Major D. Intranasal vaccination with a plant-derived H5HA vaccine protects mice and ferrets against highly pathogenic avian influenza virus challenge. Hum. Vaccin. Immunother. 2015;11(5):1235–1243. doi: 10.4161/21645515.2014.988554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220.Jones R.M. A plant-produced Pfs25 VLP malaria vaccine candidate induces persistent transmission blocking antibodies against Plasmodium falciparum in immunized mice. PLoS One. 2013;8 doi: 10.1371/journal.pone.0079538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Mathew L.G., Herbst-Kralovetz M.M., Mason H.S. Norovirus Narita 104 virus-like particles expressed in Nicotiana benthamiana induce serum and mucosal immune responses. Biomed Res. Int. 2014;2014:807539. doi: 10.1155/2014/807539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Tacket C.O. Human immune responses to a novel norwalk virus vaccine delivered in transgenic potatoes. J. Infect. Dis. 2000;182(1):302–305. doi: 10.1086/315653. [DOI] [PubMed] [Google Scholar]