Abstract
Our understanding of the hepatic consequences of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection and its resultant coronavirus disease 2019 (COVID-19) has evolved rapidly since the onset of the pandemic. In this Review, we discuss the hepatotropism of SARS-CoV-2, including the differential expression of viral receptors on liver cell types, and we describe the liver histology features present in patients with COVID-19. We also provide an overview of the pattern and relevance of abnormal liver biochemistry during COVID-19 and present the possible underlying direct and indirect mechanisms for liver injury. Furthermore, large international cohorts have been able to characterize the disease course of COVID-19 in patients with pre-existing chronic liver disease. Patients with cirrhosis have particularly high rates of hepatic decompensation and death following SARS-CoV-2 infection and we outline hypotheses to explain these findings, including the possible role of cirrhosis-associated immune dysfunction. This finding contrasts with outcome data in pharmacologically immunosuppressed patients after liver transplantation who seem to have comparatively better outcomes from COVID-19 than those with advanced liver disease. Finally, we discuss the approach to SARS-CoV-2 vaccination in patients with cirrhosis and after liver transplantation and predict how changes in social behaviours and clinical care pathways during the pandemic might lead to increased liver disease incidence and severity.
Subject terms: Liver cirrhosis, Infection, SARS-CoV-2
This Review provides mechanistic and clinical insights into COVID-19 in the context of liver disease, discussing the potential underlying biology and clinical features of SARS-CoV-2 infection in patients with pre-existing liver conditions. The management of these patients is also discussed, including SARS-CoV-2 vaccination strategies.
Key points
Patients with cirrhosis have high rates of hepatic decompensation, acute-on-chronic liver failure and death from respiratory failure following severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection and should be prioritized for coronavirus disease 2019 (COVID-19) vaccination.
The possible pathogenic mechanisms linking cirrhosis with severe COVID-19 lung disease include increased systemic inflammation, cirrhosis-associated immune dysfunction, coagulopathy and intestinal dysbiosis.
Abnormal liver biochemistry values are common in patients with COVID-19; both the prognostic significance of these derangements and whether they are directly attributable to hepatic SARS-CoV-2 infection remain uncertain.
Expression profiles of SARS-CoV-2 entry receptors vary across different in vitro and in vivo liver models; however, evidence of specific viral hepatotropism is limited.
Liver transplant recipients do not appear to have an increased risk of mortality following SARS-CoV-2 infection compared with the matched general population.
The pandemic has been associated with increased alcohol consumption, unhealthy eating habits, and interruptions to hepatology services, which might lead to an upward trend in liver disease incidence and severity.
Introduction
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is an RNA virus first reported in humans in Wuhan, China, in December 2019. The virus has since spread rapidly worldwide causing coronavirus disease 2019 (COVID-19), which continues to have a devastating effect on global health1. The majority of patients with SARS-CoV-2 infection remain asymptomatic or have mild symptoms, including fever, cough, anosmia and headache. However, around 15% develop severe pulmonary disease typically over 10 days, leading to respiratory compromise, which might progress to multi-organ failure, coagulopathy and death2–4. Oxygen supplementation, invasive ventilation and other supportive measures now form part of the standard-of-care in hospitalized patients; however, mortality remains high among those with critical disease. Common risk factors consistently associated with severe COVID-19 are now well established and include advancing age, male sex and a burden of comorbidity, including hypertension, heart disease, diabetes and malignancy5,6.
As the pandemic has evolved, most policy-makers, supported by a growing body of scientific evidence, have sought to protect their populations from infection. Those most at risk of severe COVID-19 have been advised to ‘shield’, effectively avoiding all social contact with others. A detailed understanding of the contribution of specific disease phenotypes to COVID-19 susceptibility and clinical outcomes has been crucial to enable the tailoring of public health advice to specific patient subpopulations.
In parallel, randomized clinical trials continue to evaluate therapeutic strategies against SARS-CoV-2, including direct antiviral and immune-modifying agents7,8. Understanding which patient groups require early or novel therapeutic interventions is a high clinical priority. In addition, SARS-CoV-2 vaccine development has progressed at an unprecedented rate, with the leading candidates all publishing extremely promising safety and efficacy data from phase III trials. There will now be an unprecedented global demand for vaccine deployment and, therefore, establishing which patients are most vulnerable to adverse COVID-19 outcomes is necessary to help inform who is prioritized in immunization programmes.
Since the onset of the pandemic there has been concern that pre-existing chronic liver disease (CLD) might predispose to poor outcomes following SARS-CoV-2 infection, particularly due to the overlapping risk factors for severe COVID-19 and CLD, for example, advancing age, obesity and diabetes. In addition, advanced liver disease is associated with immune dysregulation and coagulopathy, which could contribute to a more severe COVID-19 course9,10. The global burden of CLD is vast, with cirrhosis affecting more than 122 million people worldwide, of whom 10 million have decompensated disease11. Understanding the natural history of COVID-19 in patients with CLD, across different aetiologies and across the spectrum of liver disease severity, is therefore paramount.
This Review focuses on the pathogenesis and consequences of SARS-CoV-2 infection in patients with CLD based on evidence rapidly acquired through large international cohorts throughout 2020. We also explore the utility of liver biochemistry as a prognostic tool during COVID-19, summarize the evidence for direct viral infection of liver cells and explore the likely mechanisms underlying SARS-CoV-2-related liver injury. Finally, we discuss the profound effect of the pandemic on hepatology services and patient behaviours, which could lead to an increase in future liver disease incidence and severity.
Hepatotropism of SARS-CoV-2
The tissue reservoirs for SARS-CoV-2 replication remain to be fully elucidated, partly due to difficulties in accessing biopsy samples from actively infected individuals and the requirement for high level laboratory containment facilities. The virus spike protein binds ACE2 to gain cell entry and transmembrane serine protease 2 (TMPRSS2) and paired basic amino acid cleaving enzyme (FURIN) are also important for infection; therefore, the expression of these receptors provided early clues for putative hepatic permissive cells.
Single-cell RNA sequencing analyses in healthy livers have shown gene expression levels for ACE2 to be highest in cholangiocytes (comparable to alveolar type 2 cells), followed in turn by sinusoidal endothelial cells and hepatocytes12,13. TMPRSS2 and FURIN showed a broad gene expression profile across many liver cell types12. However, in a combined analysis of three single-cell RNA sequencing datasets from liver tissue from healthy individuals, very few hepatocytes co-expressed ACE2 and TMPRSS2 (ref.14). Experimental cellular and organoid models have therefore been important in trying to decipher the permissibility of liver cell types to SARS-CoV-2 infection. Hepatocellular carcinoma-derived cell lines Huh-7 and HepG2 are able to support the complete viral life cycle15; however, replication in primary hepatocytes has not yet been confirmed. This discrepancy between cellular models could be related to the presence of cancer-associated mutations in hepatoma cell lines, such as the tumour suppressor p53, which, under normal conditions, serves to downregulate intracellular SARS-CoV-2 replication16. Zhao et al. generated ACE2-expressing and TMPRSS2-expressing human liver ductal organoids that were able to recapitulate SARS-CoV-2 infection17, suggesting that the bile duct epithelium could support pseudoparticle entry. It is worth noting that the seemingly high SARS-CoV-2 entry receptor expression and viral permissibility of cholangiocytes is at odds with the non-cholestatic pattern of liver biochemistry typically found in COVID-19; the precise reasons for this aspect are currently unknown. However, it is possible that SARS-CoV-2 can undergo low level replication in cholangiocytes in vivo without triggering cell death. This process would be consistent with other reservoirs of long-term viral replication, such as in the small intestine, which can help shape memory B cell responses to the virus over time18. Human pluripotent stem cell-derived liver organoids comprising mostly albumin-expressing hepatocytes have also been shown to express ACE2 and permitted SARS-CoV-2 pseudoparticle entry19.
The influence of liver injury and underlying liver disease on the hepatotropism of SARS-CoV-2 remains unclear and no studies have yet specifically explored the histological changes found in patients with COVID-19 and pre-existing CLD. However, early studies in the era preceding COVID-19 found a greater than 30-fold increase in ACE2 expression in the liver of patients with hepatitis C virus-related cirrhosis compared with healthy individuals20. In addition, liver mRNA expression of ACE2 and TMPRSS2 was upregulated in non-infected patients with obesity and nonalcoholic steatohepatitis but not with steatosis alone21. Similarly, rodent models of liver injury via bile duct ligation have been associated with increased hepatic ACE2 expression and activity in parallel with markers of hypoxia20,22. Liver injury and inflammation could potentiate SARS-CoV-2 hepatotropism by modulating viral receptor expression, with ACE2 being identified as an interferon-inducible gene in human respiratory epithelia23,24. However, this finding must be interpreted with caution, as it might be the truncated isoform of ACE2, termed deltaACE2, rather than the viral receptor molecule itself that is being upregulated25. Whilst the tissue-specific factors controlling SARS-CoV-2 infection are poorly understood, there is an increasing recognition of the role of additional accessory receptors in viral entry. It has been shown that the high-density lipoprotein scavenger receptor B type 1 (SR-B1) helps facilitate ACE2-dependent coronavirus attachment in vitro26, reminiscent of hepatitis C virus infection27. Furthermore, treatments targeting SR-B1 reduced the lipoprotein-mediated enhancement of SARS-CoV-2 infection26. However, the histological assessment of liver tissue in this study confirmed only sparse hepatic ACE2 expression.
To obtain liver tissue and detect viral infection during the short window of active respiratory illness is technically and clinically challenging. However, extrapulmonary infection is recognized, particularly in the gastrointestinal tract, with stool SARS-CoV PCR remaining positive for up to a week after viral clearance from the lungs28. Furthermore, pervasive infection of enterocytes has been well documented29,30, with viral protein and RNA found to persist in intestinal biopsies for many months after clinical infection has resolved18. In post-mortem wedge liver biopsy samples from 48 patients dying from severe COVID‐19 lung disease, SARS-CoV-2 was detected via in situ hybridization in 68% of samples31. Histological assessment also revealed vascular abnormalities characterized by portal venous and sinusoidal microthromboses (100%), microvesicular and macrovesicular steatosis (50%), mild portal inflammation (66%), and portal fibrosis (60%). The latter finding could suggest a degree of underlying liver disease, most likely secondary to nonalcoholic fatty liver disease (NAFLD) given that metabolic risk factors, including hypertension and cardiovascular disease, were enriched in this cohort. Again, the absence of histological evidence of biliary injury is conspicuous given the degree of cholangiocyte viral entry receptor expression. Post-mortem electron microscopy of liver tissue in a case of fatal respiratory COVID-19 has also directly visualized possible coronavirus-like particles in hepatocyte cytoplasm in association with mitochondrial swelling and apoptosis32. This finding would be consistent with large protein interaction maps that have identified connectivity between NSP5, the main protein of SARS-CoV-2, and mitochondrial components33. Conversely, in-depth proteomic assessment of autopsy tissue from 19 patients with COVID-19 revealed little evidence of active viral replication in the liver34. However, hepatic protein signatures did indicate upregulated profibrotic pathways, dysregulated fatty acid oxidation and oxidative phosphorylation, and markers of immune activation. This shifting proteomic landscape was found to be associated with the presence of multi-organ dysfunction, hepatic steatosis and coagulative hepatocyte necrosis34.
Whilst there is variation in viral entry receptor expression profiles across the different in vitro and in vivo liver models (Fig. 1), direct evidence of specific SARS-CoV-2 hepatotropism is lacking. Given the poor outcomes and high rates of hepatic decompensation in patients with cirrhosis and COVID-19, more work is needed to determine the pathogenic role of direct hepatic viral infection and the mechanisms leading to hepatic decompensation in patients with underlying liver disease.
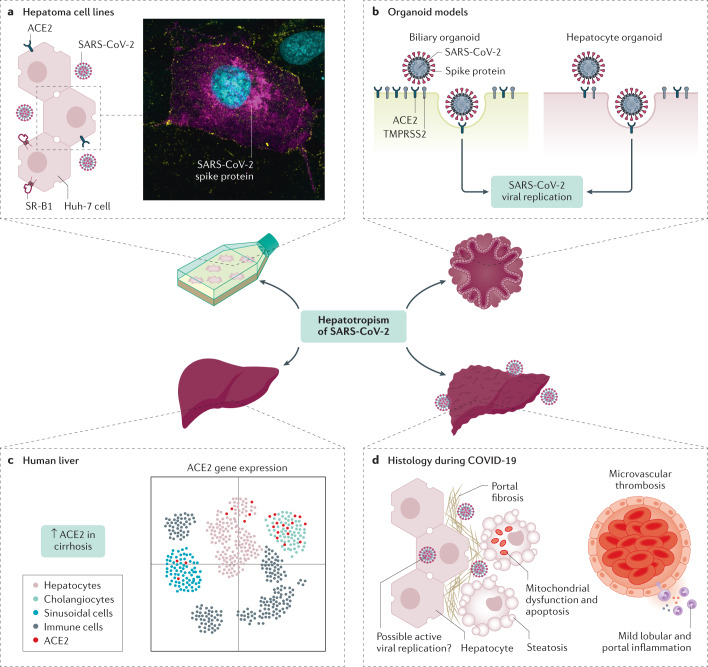
Fig. 1. Hepatotropism of SARS-CoV-2.
Understanding the hepatotropic effects of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has required a combination of experimental and clinical models. Hepatoma cell lines in vitro have been shown to support the entire life cycle of SARS-CoV-2 (part a, which shows a Huh-7 cell with widespread intracellular spike protein staining magenta). In vitro cell models have also demonstrated the role of accessory receptors such as high-density lipoprotein receptor scavenger receptor B type 1 (SR-B1) alongside ACE2 for cell entry. In addition, both biliary and hepatocyte organoid models have been shown to express necessary viral entry receptors and recapitulate SARS-CoV-2 infection (part b). Although there is some variability between gene expression studies regarding the distribution of ACE2 on liver cell types, cholangiocytes seem to have the greatest receptor concentration followed by hepatocytes (part c); there is also ACE2 upregulation in the parenchyma of cirrhotic livers. Lastly, histological examination of livers from patients with fatal respiratory coronavirus disease 2019 (COVID-19) have shown a range of microscopic changes such as widespread vascular abnormalities, steatosis and mitochondrial abnormalities (part d). The evidence for direct hepatocyte infection remains inconclusive. TMPRSS2, transmembrane serine protease 2. Part a microscopy image courtesy of S. Davies, University of Birmingham. Part c adapted with permission from ref.12, Wiley.
Liver biochemistry in COVID-19
Patterns and frequency of liver biochemistry abnormalities in COVID-19
Although the precise influence of COVID-19 on the liver remains unclear, abnormalities in liver biochemistries are common in patients with COVID-19, occurring in approximately 15–65% of SARS-CoV-2-infected individuals35–42. The wide range in these reported frequencies could be attributable to different definitions of upper limit of normal, varying laboratory values considered as liver enzymes, and geographical variability in the prevalence and type of underlying CLD43. Liver biochemistry abnormalities in COVID-19 are generally characterized by mild (1–2 times the upper limit of normal) elevations of serum alanine aminotransferase (ALT) and aspartate aminotransferase (AST) levels, reported in an estimated 29–39% and 38–63% of patients, respectively36–38,44. Hypoalbuminaemia, a non-specific marker of illness severity, has been reported to be associated with worse COVID-19 outcomes45, but severe liver injury, elevation in serum bilirubin level and liver synthetic dysfunction are all rare in patients infected with SARS-CoV-2 (refs41,42,44). Abnormalities in liver biochemistries are reported in similar frequencies regardless of the presence of pre-existing liver disease46.
Underlying causes of elevated liver enzymes in COVID-19
There are a number of potential contributors to elevated liver enzyme levels in COVID-19. Liver biopsy results in patients with SARS-CoV-2 have been characterized by non-specific findings, including steatosis, mild lobular and/or portal inflammation, and vascular pathology31,47,48. In most cases, abnormal biochemistries are likely multifactorial with potential contributions from immune-mediated inflammatory response, drug-induced liver injury, hepatic congestion and extrahepatic release of transaminases49 as well as possible direct infection of hepatocytes.
Among hospitalized patients with COVID-19, elevations of serum AST levels positively correlate with levels of ALT but not with markers of muscle breakdown (such as creatinine kinase) or systemic inflammation (such as C-reactive protein (CRP) and ferritin)50. These findings imply that elevated liver enzymes in COVID-19 result from direct hepatic injury, although COVID-19-associated rhabdomyolysis is rarely reported51. Lastly, AST is often found to exceed ALT during the course of COVID-19, which would be atypical for a classic hepatocellular pattern of liver injury outside of specific contexts such as alcohol-related liver disease, certain drug-induced liver injuries (for example, lamotrigine), ischaemic hepatitis and cirrhosis50. The mechanisms responsible for an AST-predominant aminotransferase elevation remain incompletely defined but could include COVID-19-related mitochondrial dysfunction33, SARS-CoV-2-induced hepatic steatosis31 and altered hepatic perfusion secondary to microthrombotic disease31,52. A systemic review and meta-analysis of biopsy and autopsy studies reported the prevalence of hepatic vascular thrombosis in COVID-19 to be 29%53. Systemic hypoxia in COVID-19 might also have a contributory role and, interestingly, AST elevations have previously been reported with other viral pneumonias, including influenza A H1N1 infection, where levels increased in parallel with diminishing peripheral oxygen saturations54.
As with many other infections, SARS-CoV-2 is associated with systemic inflammation that could contribute to elevations in liver biochemistries via cytokine release55. Patients with substantial elevations in serum ALT levels often have high levels of CRP (which is synthesized by the liver), D-dimer, ferritin and IL-6 (refs44,45,56,57). IL-6, which is produced by monocytes, macrophages and T cells in response to activation of the innate and adaptive immune system, is the key driver of CRP production and high IL-6 levels are associated with liver injury in COVID-19 (refs44,56). Notably, IL-6 increases during COVID-19 illness, declines as patients recover and correlates with severity of the disease course58.
There are several other potential contributors to abnormal liver biochemistries in COVID-19, including ischaemic hepatitis, hepatic congestion related to cardiomyopathy, and transaminase release due to the breakdown of skeletal and cardiac muscle49. Venous and arterial thromboses are now a well-recognized feature of COVID-19 (refs59–62), including in the liver31,48, which could contribute to elevations in liver biochemistries. Lastly, drug-induced liver injury is likely to contribute to elevated liver enzymes and might have been more common early in the pandemic due to the use of experimental therapies63. However, no study has yet comprehensively mapped the pattern of liver function tests found within studies over the course of the pandemic. Specific COVID-19 treatments implicated in cases of drug-induced liver injury include lopinavir–ritonavir64,65, tocilizumab66,67 and remdesivir. The hepatotoxicity of remdesivir has been subject to debate. Although randomized trials in COVID-19 demonstrate equivalent liver enzyme elevations between treatment and control groups68, screening of the WHO safety reports database still reveals a statistically significant odds ratio for liver injury with the use of remdisivir69. Fortunately, these considerations are likely to become less clinically relevant in light of the SOLIDARITY trial showing no benefit of remdesivir in hospitalized patients with COVID-19 (ref.70).
Prognostic significance of elevated liver enzymes in COVID-19
The prognostic significance of elevated liver enzymes in patients infected with SARS-CoV-2 is currently debated. Some reports have demonstrated that elevations of serum liver enzyme levels are associated with adverse outcomes, including shock, intensive care unit (ICU) admission and mechanical ventilation43,65,71–74. However, these studies could be prone to bias if patients with severe disease received more intensive laboratory monitoring, increasing the likelihood of detecting liver injury. Some have reported that there is no apparent association between liver enzyme level elevations and mortality71,75, whereas others have found that elevated liver enzyme levels, and particularly AST and ALT level elevations greater than five times the upper limit of normal, are associated with an increased risk of death42,44,76,77. It has been proposed that the prognostic significance of elevated liver enzyme levels could be due to a more robust host response and aggressive therapies in those with severe illness78.
SARS-CoV-2 infection and liver disease
Clinical course and outcomes following SARS-CoV-2 infection in pre-existing CLD
Patients with CLD and especially cirrhosis have multiple mechanisms of immune dysfunction that can lead to increased susceptibility to infection and an aberrant inflammatory response during infection — collectively known as cirrhosis-associated immune dysfunction (CAID). This immune dysfunction includes reduced components of the complement system, macrophage activation, impaired lymphocyte and neutrophil function, Toll-like receptor upregulation, and intestinal dysbiosis9,79. Although research attention has mostly focused on mechanisms leading to severe bacterial infection80, CAID has also been shown to predispose to a variety of viral or fungal-related disease81. Whether patients with CLD and cirrhosis are more susceptible to SARS-CoV-2 infection has been the focus of much attention throughout 2020. Data from both large case series of thousands of consecutive patients with COVID-19 and population studies using health records do not suggest that patients with chronic liver disease are over-represented5,37. In fact, electronic health record data from the USA has demonstrated that patients with cirrhosis are actually at lower risk of testing positive for SARS-CoV-2 infection82,83. It remains improbable that cirrhosis is protective against SARS-CoV-2 acquisition and, therefore, this lower rate of positive testing is likely due to a combination of increased patient compliance with preventative measures (for example, social distancing, handwashing) and more frequent routine testing. Patients with cirrhosis who do acquire infections of all types have been shown to have worse clinical outcomes than patients without underlying liver disease84,85 and SARS-CoV-2 infection seems to follow a similar pattern.
The aetiology of liver disease could influence clinical outcome in COVID-19. In the general population, advancing age, obesity and diabetes are risk factors for COVID-19 morbidity and mortality5; however, in these series, such patients were not explicitly diagnosed with NAFLD, in part because hepatic steatosis was not recorded or because alcohol use was not assessed. Indeed, there are wide inconsistencies throughout the literature regarding the influence of NAFLD on the COVID-19 course. This inconsistency could be related to difficulties in separating the effect of NAFLD from other metabolic comorbidities, due to the confounding effect of viral-induced steatosis or because of varying diagnostic criteria. The latter is of particular importance currently as the global hepatology community grapples with the proposed classification changes from NAFLD to metabolic dysfunction-associated liver disease86. One retrospective series of 202 consecutive patients with SARS-CoV-2 infection identified NAFLD as a risk factor for progressive COVID-19, abnormal liver enzyme levels and longer viral shedding time87. Another study in 327 patients showed an intersection between NAFLD and the risk of severe COVID-19 in patients under 60 years of age88. Similarly, MRI data from 287 patients tested for SARS-CoV-2 (79 positive, 208 negative) demonstrated that individuals with obesity and a concomitant liver fat fraction of ≥10% were at threefold increased risk of developing symptomatic laboratory-confirmed COVID-19 (available as a non-peer-reviewed Preprint only)89. However, in a series of 155 consecutive inpatients with COVID-19, the presence of steatosis (in 43%) was not independently associated with mortality90. These findings were confirmed in a larger international cohort of 745 patients with CLD and cirrhosis from 29 countries collected using the SECURE-Cirrhosis and COVID-Hep registries in which the odds ratio for death for patients with NAFLD was 1.01 (95% CI 0.57–1.79)91. In this same study, alcohol-related liver disease was the only liver disease aetiology with a significant odds ratio for death (1.79, 95% CI 1.03–3.13)91. Registry data for 70 patients with autoimmune hepatitis and SARS-CoV-2 infection has also shown equivalent outcomes to patients with CLD of other aetiology and to propensity score-matched controls despite the use of baseline immunosuppression in 86% of cases92. In multiple series, the main cause of death in CLD patients was COVID-19-induced pulmonary disease followed by liver-related mortality91,93.
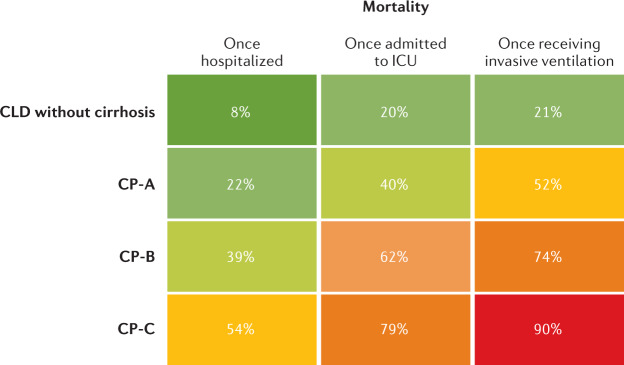
Among patients with prevalent liver disease, once infected with SARS-CoV-2, there is a stepwise increase in morbidity and mortality with increasing severity of cirrhosis as measured by Child–Pugh (CP) class. Although the proportion of patients hospitalized in the SECURE-Cirrhosis and COVID-Hep registries was no different among patients with CLD and CP classes A, B and C, there was a stepwise increase in frequency of ICU admission, renal replacement therapy, invasive ventilation and death. There was also an increase in mortality for all patients as they required more intense medical support, with patients classed as CP-C having only a 10% survival once undergoing mechanical ventilation (Fig. 2). After adjusting for baseline characteristics, COVID-19-related mortality was significantly associated with the severity of pre-existing liver cirrhosis and the odds ratio for death increased across the stages of cirrhosis: CP-A 1.90 (95% CI 1.03–3.52), CP-B 4.14 (95% CI 2.24–7.65) and CP-C 9.32 (95% CI 4.8–18.08)91. Notably, registry data are vulnerable to reporting bias of more severe cases, which likely accounts for the inclusion of predominantly hospitalized patients in these studies. However, it is reassuring that the SECURE-Cirrhosis and COVID-Hep registries included a large proportion of patients without cirrhosis and with non-severe COVID-19 and that mortality was consistent with a range of other smaller cohorts (Table 1). A summary of studies reporting outcomes in patients with SARS-CoV-2 and pre-existing liver disease is presented in Table 1. Although the acute mortality associated with COVID-19 in patients with cirrhosis is high, in those who survive the initial insult, the rates of death and re-admission at 90-days are comparable to those with cirrhosis alone94. Therefore, beyond the acute infective period, SARS-CoV-2 infection does not seem to precipitate liver disease progression over and above the natural history of cirrhosis.
Fig. 2. Mortality following SARS-CoV-2 infection according to baseline liver disease stage and level of medical support.
Rates of mortality in patients with chronic liver disease (CLD) and severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection following hospitalization, admission to intensive care unit (ICU) and invasive ventilation separated by liver disease stage. CP, Child–Pugh. Adapted from ref.91, CC BY 4.0 (https://creativecommons.org/licenses/by/4.0/).
Table 1.
Summary of COVID-19 outcome studies in patients with CLD and post-liver transplantation
| Study | Design | Country/region and number included | Major findings |
|---|---|---|---|
| Cirrhosis | |||
| Marjot et al.91 (Oct 2020) | Large international registry study | 29 countries; SARS-CoV-2 infection plus cirrhosis (n = 386); SARS-CoV-2 plus CLD without cirrhosis (n = 359); SARS-CoV-2 plus non-CLD (n = 620) | Overall mortality: CP-A (19%), CP-B (35%), CP-C (51%), CLD without cirrhosis (8%); increased risk of death cirrhosis vs CLD without cirrhosis: CP-A (OR 1.9, 95% CI 1.03–3.5), CP-B (OR 4.1, 95% CI 2.4–7.77), CP-C (OR 9.32, 95% CI 4.80–18); increased risk of death compared with propensity score-matched patients without CLD: CP-B (+20%, 8.8–31.3%) and CP-C (+38%, 27.1–49.2%) |
| Iavarone et al.93 (Jun 2020) | Multicentre retrospective cohort study | Italy; SARS-CoV-2 plus cirrhosis (n = 50); SARS-CoV-2 plus no cirrhosis (n = 399); cirrhosis plus bacterial infection (n = 47) | 30-day mortality: SARS-CoV-2 plus cirrhosis vs SARS-CoV-2 plus no cirrhosis (34% vs 18%; P = 0.030); SARS-CoV-2 plus cirrhosis vs cirrhosis plus bacterial infection (34% vs 17%; P = 0.03) |
| Bajaj et al.177 (Jun 2020) | Multicentre retrospective cohort study | North America and Canada; SARS-CoV-2 plus no cirrhosis (n = 108); SARS-CoV-2 plus cirrhosis (n = 37); cirrhosis alone (n = 127) | Overall mortality: cirrhosis plus SARS-CoV-2 higher mortality compared with patients with SARS-CoV-2 alone (30% vs 13%; P = 0.03) but not between patients with cirrhosis plus SARS-CoV-2 and patients with cirrhosis alone (30% vs 20%; P = 0.16) |
| Kim et al.178 (Sep 2020) | Multicentre retrospective cohort study | North America; SARS-CoV-2 plus CLD without cirrhosis (n = 620); SARS-CoV-2 plus cirrhosis (n = 227) | Increased risk of death with decompensated cirrhosis (OR 2.91, 95% CI 1.70–5.00); no increased risk with compensated cirrhosis (OR 0.83, 95% CI 0.46–1.49) |
| Sarin et al.179 (Jun 2020) | Multinational registry study | 13 countries in Asia; SARS-CoV-2 plus CLD without cirrhosis (n = 185); SARS-CoV-2 plus cirrhosis (n = 43) | Overall mortality: SARS-CoV-2 plus CLD without cirrhosis vs SARS-CoV-2 plus cirrhosis (16% vs 3%; P = 0.002) |
| Clift et al.128 (Sep 2020) | Population-based cohort study using electronic health record data | United Kingdom; 6 million adults: 11,865 with cirrhosis, 37 deaths from COVID-19 in patients with cirrhosis and 106 hospitalizations with COVID-19 in patients with cirrhosis | Hazard ratio for COVID-19-related mortality in patients with cirrhosis: women in derivation cohort, 1.8 (95% CI 1.15–2.99); men in derivation cohort, 1.29 (95% CI 0.83–2.02) |
| Ioannou et al.83 (Nov 2020) | Population-based study using electronic health record data | North America; SARS-CoV-2 plus cirrhosis (n = 305); SARS-CoV-2 plus no cirrhosis (n = 9,826); cirrhosis alone (n = 3,301) | Patients with SARS-CoV-2 plus cirrhosis 3.5 times more likely to die than those with SARS-CoV-2 without cirrhosis |
| Liver transplant recipients | |||
| Webb et al.117 (Aug 2020) | Large international registry study | 18 countries; SARS-CoV-2 plus LT recipient (n = 151); SARS-CoV-2 plus non-LT recipients from Oxford University Hospitals (n = 627) | Overall mortality (19%); LT did not significantly increase the risk of death (absolute risk difference 1.4%, 95% CI –7.7 to 10.4); risk factors for mortality within LT recipients: age, renal function and non-HCC cancer |
| Colmenero et al.115 (Aug 2020) | Prospective multicentre cohort study | Spain; SARS-CoV-2 plus LT recipient (n = 111); SARS-CoV-2 plus general population during study period (n = 150,000); LT recipients without SARS-CoV-2 (n = 13,000) |
Mortality in LT recipients (18%) lower than the matched general population; SMR 95.5 (95% CI 94.2–96.8); SIR 191.2 (95% CI 190.3–192.2) Baseline mycophenolate independent risk factor for severe COVID-19 (ICU, IPPV or death) (RR 3.94, 95% CI 1.59–9.74; P = 0.003). |
| Webb et al.129 (Feb 2021) | Combined analysis of Webb et al117 and Colmenero et al.115 | 18 countries including 108 cases from Spain; SARS-CoV-2 plus LT recipient (n = 258) | Age and Charlson Comorbidity Index independently associated with death; no association with type of immunosuppression regime |
| Rabiee et al.127 (Sep 2020) | Multicentre retrospective cohort study | SARS-CoV-2 plus LT recipient (n = 112) | Overall mortality (22%); no independent risk factors for death identified |
| Ravanan et al.116 (Aug 2020) | National cohort study: National Health Service Blood and Transplant registry data | England; SARS-CoV-2 plus LT recipient (n = 64); rates of infection and mortality compared with all SARS-CoV-2-positive general English population | Overall mortality (23%); reduced risk of SARS-CoV-2 infection (OR 0.53, 95% CI 0.40–0.70) |
| Kates et al.180 (Aug 2020) | Multicentre prospective cohort study | USA; SARS-CoV-2 plus LT recipients (n = 73) | 28-day mortality (21%); within whole solid organ transplant cohort (n = 482), LT not independently associated with death |
| Belli et al.181 (Dec 2020) | European registry study | 9 European countries; SARS-CoV-2 plus LT recipient (n = 243) | Overall mortality (20%); risk factors for mortality: age, diabetes, chronic kidney disease; Tacrolimus had positive independent effect of survival (0.55; 95% CI 0.31–0.99) |
| Alcohol-related liver disease | |||
| Marjot et al.91 (Oct 2020) | Large international registry study | 29 countries; SARS-CoV-2 plus CLD (n = 745); SARS = CoV-2 plus ALD (n = 179) | ALD independent risk factor for death (OR 1.79, 95% CI 1.03–3.13) |
| Kim et al.178 (Sep 2020) | Multicentre retrospective cohort study | North America; SARS-CoV-2 plus CLD (n = 867); SARS-CoV-2 plus ALD (n = 94) | ALD independent risk factor for death (HR 2.42, 95% CI 1.29–4.55) |
| Nonalcoholic fatty liver disease | |||
| Marjot et al.91 (Oct 2020) | Large international registry study | 29 countries; SARS-CoV-2 plus CLD (n = 745); SARS-CoV-2 plus NAFLD (n = 322) | No independent association with death controlling for age, sex, ethnicity, liver disease stage, BMI, CVD, T2DM, hypertension, COPD, smoking status |
| Kim et al.178 (Sep 2020) | Multicentre retrospective cohort study | North America; SARS-CoV-2 plus CLD (n = 867); SARS-CoV-2 plus NAFLD (n = 465) | No independent association with death controlling for age, sex, ethnicity, cirrhosis, T2DM, hypertension, CVD, COPD, smoking status |
| Viral hepatitis | |||
| Marjot et al.91 (Oct 2020) | Large international registry study | 29 countries; SARS-CoV-2 plus CLD (n = 745); SARS-CoV-2 plus HBV (n = 96); SARS-CoV-2 plus HCV (n = 92) | No independent association with death |
| Kim et al.178 (Sep 2020) | Multicentre retrospective cohort study | North America; SARS-CoV-2 plus CLD (n = 867); SARS-CoV-2 plus HBV (n = 62); SARS-CoV-2 plus HCV (n = 190) | No independent association with death |
| Butt et al.182 (Feb 2021) | Population-based study using electronic health record data | USA; SARS-CoV-2 plus HCV (n = 975), SARS-CoV-2 no HCV (n = 975) | SARS-CoV-2 plus HCV more likely to be hospitalised but not at increased risk of death. |
| Hepatocellular carcinoma | |||
| Marjot et al.91 (Oct 2020) | Large international registry study | 29 countries; SARS-CoV-2 plus CLD (n = 745); SARS-CoV-2 plus HCC (n = 48) | No independent association with death |
| Kim et al.178 (Sep 2020) | Multicentre retrospective cohort study | North America;SARS-CoV-2 plus CLD (n = 867); SARS-CoV-2 plus HCC (n = 22); locoregional therapy (n = 8), immunotherapy (n = 2) | Independent risk factor for death (HR 3.96, 95% CI 1.74–8.98) |
| Autoimmune liver disease | |||
| Marjot et al.92 (Jan 2021) | Large international registry study | 35 countries; SARS-CoV-2 plus AIH (n = 70); 86% immunosuppression; SARS-CoV-2 plus non-AIH CLD (n = 862); SARS-CoV-2 plus non-CLD (n = 769) | Immunosuppression not an independent risk factor for death in patients with AIH; equivalent rates of mortality for patients with AIH vs non-AIH CLD; higher rates of hospitalization but equivalent rates of mortality for patients with AIH compared to non-CLD |
Summary of studies investigating the effect of SARS-CoV-2 infection on patients with chronic liver disease separated by disease aetiology or post liver transplantation. AIH, autoimmune hepatitis; ALD, alcohol-related liver disease; CLD, chronic liver disease; COPD, chronic obstructive pulmonary disease; COVID-19, coronavirus diseases 2019; CP, Child–Pugh; CVD, cardiovascular disease; HBV, hepatitis B virus; HCC, hepatocellular carcinoma; HCV, hepatitis C virus; ICU, intensive care unit; IPPV, invasive positive pressure ventilation; LT, liver transplant; NAFLD, nonalcoholic fatty liver disease; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; SIR, standardized incidence ratio; SMR, standardized mortality ratio; T2DM, type 2 diabetes mellitus.
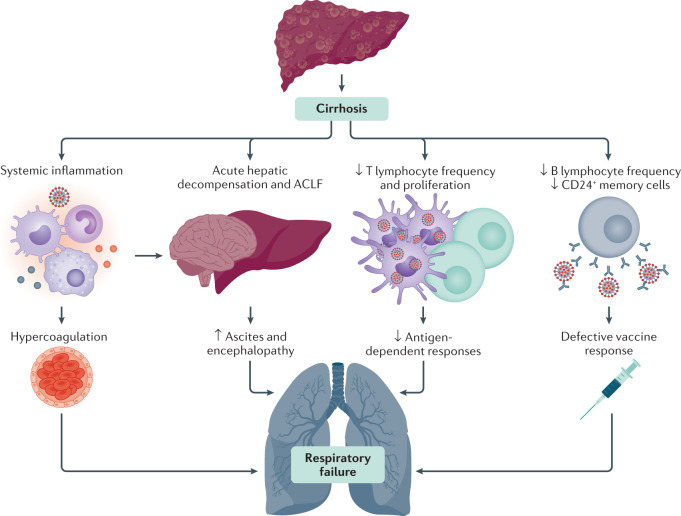
COVID-19 can cause acute-on-chronic liver failure (ACLF). Although traditionally associated with bacterial infections, viral illness can also precipitate ACLF85,95, which is marked by both liver-specific decompensation and increasing severity and frequency of extrahepatic organ system failure96. In the SECURE-Cirrhosis and COVID-Hep registries, hepatic decompensation events were more frequent with increasing severity of liver disease and mortality increased with worsening ACLF as measured by the CLIF-C score91,93,97. CAID likely underlies the intersection between severe COVID-19 pulmonary disease and ACLF. Cirrhosis is known to be associated with an increase in baseline endotoxemia and cytokine production that can lead to an exaggerated inflammatory response in the setting of infection (Fig. 3). This aspect might be particularly severe in patients with alcohol-induced liver disease98, potentially explaining the increased mortality in this group91. It has also been shown that gut microbiota composition plays a role in regulating the magnitude of COVID-19 severity, possibly via modulating host immune responses99. Given that cirrhosis is characterized by changes to gut microbiota composition and function alongside intestinal permeability100, it is possible that alterations in the gut–liver axis might contribute to the severe COVID-19 course observed in this patient group. However, further research into the mechanisms underlying CAID, ACLF and COVID-19 are required.
Fig. 3. Possible mechanisms for adverse COVID-19 outcomes in patients with cirrhosis.
Patients with cirrhosis have a high risk of mortality from respiratory failure following severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection. This risk might occur through multiple converging pathways, including contributions from cirrhosis-associated immune dysfunction, acute hepatic decompensation and a systemic inflammatory response. Cirrhosis-associated immune dysfunction could also lead to defective immune responses following future SARS-CoV-2 vaccination. ACLF, acute-on-chronic liver failure; COVID-19, coronavirus disease 2019.
Lastly, the COVID-19 pandemic has unmasked the long-entrenched associations between race, socioeconomic status and adverse health outcomes. This issue includes patients with CLD for which socioeconomic disparities in American non-Hispanic Black and Hispanic communities have been associated with an increased risk of SARS-CoV-2 infection101. In addition, racial and socioeconomic disparities in internet access have been shown to translate into barriers for the use of telehealth, particularly video technology, in patients with CLD102.
Specific management of patients with COVID-19 and underlying liver disease
The optimal approach to the management of patients with underlying liver disease who acquire SARS-CoV-2 infection is still evolving. However, the characterization of the COVID-19 course through multicentre and international cohorts has helped optimize the treatment strategies for this patient group, as summarized in various consensus guidelines103–105.
Firstly, the recognition that patients with cirrhosis are particularly vulnerable to the severe complications of COVID-19 is paramount. This recognition should encourage a low threshold for SARS-CoV-2 testing and consideration of early admission for those with a positive diagnosis. Acute hepatic decompensation is common, occurring in 47% of patients with cirrhosis and COVID-19, and typically manifests as worsening ascites and encephalopathy91, which should be identified and managed along traditional lines106. Indeed, hepatic decompensation could be the first and only indication of SARS-CoV-2 infection in patients with cirrhosis, with 24% having no concurrent pulmonary symptoms91. Importantly, data have shown that patients with autoimmune hepatitis have similar rates of COVID-19-related mortality to the matched general population92 and that the use of immunosuppression is not an independent risk factor for death. This finding should reassure clinicians and provides a clear rationale not to routinely reduce immunosuppression in these patients during the COVID-19 course.
The trajectory of patients with COVID-19 and decompensated cirrhosis at baseline is bleak, with up to 80% mortality in those requiring ICU support91, although this number might decrease with time as the care of patients with COVID-19 continues to improve. Notably, the majority of deaths in patients with cirrhosis and COVID-19 are from respiratory failure, although the precise mechanisms underpinning this observation remain unclear. However, it is plausible that pulmonary venothromboembolic disease, a hallmark of critical COVID-19, has a contributory role given the additional hypercoagulable state associated with cirrhosis. Whilst routine thromboprophylaxis is universally recommended in hospitalized patients with COVID-19, unified risk stratification models and treatment algorithms have yet to emerge, with wide variation in clinical practice and between international guidelines107,108. However, given the coagulopathy associated with both cirrhosis and COVID-19, the coexistence of these conditions might yield a cumulative risk of thrombotic complications103. Although it remains to be determined whether enhanced venothromboembolic prophylaxis will be of benefit in this patient group, studies prior to the COVID-19 pandemic showed a benefit of anticoagulation in reducing portal pressures109 without an excess bleeding risk110. In addition, a multicentre study in northern Italy reassuringly reported no major haemorrhagic complications with the use of thromboprophylaxis in 40 patients with cirrhosis admitted with COVID-19. Unfortunately, patients with advanced cirrhosis are excluded in many (but not all) published and active trials investigating optimal thromboprophylaxis following admission with SARS-CoV-2 infection111,112.
The race to develop specific therapies for COVID-19 continues at pace. However, an international registry study involving 29 countries and 130 different institutions found that patients with cirrhosis were significantly less likely to receive targeted antiviral therapy than those without (33% versus 52%; P < 0.001)91. This finding might reflect prescribing concerns and demonstrates the need for COVID-19 trials to carefully evaluate drug hepatotoxicity to prevent patients with cirrhosis, who are already at high risk of death, from being unnecessarily denied disease-modifying treatment.
Clinical course and outcomes of SARS-CoV-2 infection in liver transplant recipients
In population terms, SARS-CoV-2 infections in liver transplant (LT) recipients are relatively uncommon. However, there is keen interest in the effect of SARS-CoV-2 on this patient cohort, who have frequent exposure to health-care facilities and frequent concurrent comorbidities and among whom the majority have an absolute requirement for chronic immunosuppression113,114.
Assessing the susceptibility of LT recipients to SARS-CoV-2 infection is challenging because of geographical and chronological variability in diagnostic methods, thresholds for testing and individual exposures. Nonetheless, country-wide data from Spain and the UK suggest that diagnoses of SARS-CoV-2 are more frequent among LT recipients than among the general population, although this finding might be partly attributable to more intensive monitoring and a lower threshold for viral testing in the liver transplantation setting115,116. Further data from primary care cohorts will increase our understanding of this area but will require careful correlation with contemporary epidemiology.
It is clear that not all LT recipients diagnosed with SARS-CoV-2 infection develop severe COVID-19. For example, in the Spanish series (n = 111), 6% were described as asymptomatic and, in a multinational series (n = 151), 14% had neither respiratory nor gastrointestinal symptoms115,117. In a German study that used serology to identify LT recipients with likely previous exposure to SARS-CoV-2, 5 of 8 individuals with a detectable IgG response reported no previous symptoms consistent with COVID-19 (ref.118).
In those who develop symptoms, the key respiratory features of COVID-19 appear similar in those with and without liver transplatation, with fever, cough and dyspnoea developing around 1 week following SARS-CoV-2 infection. In patients hospitalized with severe disease, marked elevations in ferritin, D-dimer and IL-6 levels alongside lymphopenia on laboratory assays, and diffuse parenchymal opacities without pleural effusion on chest radiology are well-recognized119,120. One difference among LT recipients seems to be a high frequency of gastrointestinal symptoms, with diarrhoea reported in 31% and 42% of two early cohorts, respectively119,120. In a propensity-matched analysis, gastrointestinal symptoms were also more frequent among patients with LT than in those without117.
Assessing the outcomes associated with liver transplantation among individuals infected with SARS-CoV-2 is complicated by the demography and pattern of comorbidity within the population. LT recipients are more likely to be male121 and more likely to have renal impairment122, type 2 diabetes mellitus123 and obesity124 than the general population. Furthermore, the age of LT recipients is increasing, with a median recipient age in both the USA and the UK of 56 years125. All of these factors are well established as being associated with an increased risk of adverse outcomes following SARS-CoV-2 infection and these risks seem to apply to LT recipients. Within cohort studies of LT recipients, age and burden of comorbidity (for example, Charlson Comorbidity Index) are strongly associated with death or severe disease course115,117. Although only a minority of LT recipients have clinically significant liver dysfunction, if present, this aspect could logically be considered a possible additional risk factor for severe COVID-19 as described elsewhere in this Review126.
When considering the risk conveyed by liver transplantation itself in the context of SARS-CoV-2 infection, adjustment for concurrent comorbidity is required. Clinical data incorporating such adjustments suggest that LT recipients are not at increased risk of severe COVID-19 or death as compared to non-LT recipients115,117. Notably, such data derived from registry work are open to reporting biases, although case fatality rates are broadly consistent across cohort studies from the first wave of the pandemic at around 20%115–117,119,127. Importantly, the observation of there being no increased risk of severe COVID-19 seems to hold true when comparing the national LT and non-LT populations in Spain115. Furthermore, work combining a large cohort of primary care records and hospital episodes in the UK has confirmed an increased risk of death from COVID-19 in patients with CLD but shown no statistically significant difference in mortality in non-renal solid organ recipients128. When considering the relative importance of comorbidity, it is notable that in pooled data from the two large cohorts of SARS-CoV-2 infection in LT recipients, no patient with a Charlson Comorbidity Index of 0 died (points are attributed for age >50 years but not LT itself129.
The current understanding that liver transplantation and the associated immunosuppression itself do not seem to confer an increased risk of poor outcome in SARS-CoV-2 infection is consistent with other observations. First, typical immunosuppression for LT recipients primarily limits the adaptive immune response rather than the innate response, with the latter seeming primary in determining COVID-19 outcome130. The only form of immunosuppression strongly linked with poor outcomes identified to date is of specific deficits in the type I interferon innate response131,132. Second, reactive corticosteroid immunosuppression with high-dose dexamethasone reduces mortality in severe COVID-19 (ref.133). Third, as noted by others, immunosuppression, including for liver transplantation, did not emerge as a risk factor for poor outcome in SARS-CoV-1 or Middle Eastern Respiratory Syndrome infections, whereas comorbidities analogous to those conferring risk in SARS-CoV-2 infection did134. Interestingly, the fact that LT recipients have similar outcomes to the matched general population is in stark contrast to the very high rates of mortality reported in those with advanced liver disease. This finding might suggest that, in the context of COVID-19, immunosuppression secondary to CAID has a more deleterious effect than pharmacological immunosuppression.
It therefore remains the case that major international guidelines currently recommend against the routine cessation or reduction of immunosuppressive therapy in LT recipients before any SARS-CoV-2 infection and that modification following SARS-CoV-2 be considered under special circumstances such as in superadded bacterial infection or worsening respiratory failure103,104,135. As already discussed, disturbances of liver biochemistry are frequent in COVID-19 but seem to be an indirect effect of SARS-CoV-2 infection analogous to that seen in the cytokine storm in other situations55,57,130. Although liver injury — as defined by elevated serum transaminase activities — occurs in LT recipients and is associated with mortality, it seems either no more frequent or less frequent than in comparison groups117,127.
Whilst data about differences on SARS-CoV-2 infection in those with LT is accumulating, numerous issues remain (Box 1). However, to date, it seems clear that the weight of comorbidity is more important in determining outcome than LT status per se and that being an immunosuppressed LT recipient confers less risk from SARS-CoV-2 than having advanced CLD, thereby supporting the continuation of liver transplantation programmes.
Box 1 Key unresolved issues for SARS-CoV-2 infection in LT recipients.
Susceptibility and disease course
What is the role of health-care exposure? Health-care acquisition in liver transplant (LT) recipients has been reported118; it is unclear as to the optimum method of minimizing this exposure.
Is recurrent infection more likely? With early reports of reinfection in non-transplant patients, it remains unknown as to whether LT recipients with impaired adaptive responses will be susceptible to reinfection183.
Is viral shedding prolonged in LT recipients? Case reports suggest prolonged viral shedding in some LT recipients, which might have public health implications184.
Is the post-infection immune response durable? Early reports suggest that the neutralizing IgG response to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is of variable duration185. This aspect is unexplored in LT recipients. It is unclear whether a protective post-infection immune response will persist after transplantation.
Is the apparent increased frequency of gastrointestinal symptoms of clinical importance? Gastrointestinal symptoms seem more frequent in LT recipients; the importance of this finding remains unclear.
Therapeutics and clinical management
Caution with drug–drug interactions with novel therapies: drug interactions are frequent in transplant recipients, especially with calcineurin inhibitors, and must be considered with future novel therapies.
Caution with putative future therapies that act to stimulate aspects of the immune response: putative coronavirus disease 2019 (COVID-19) therapies, such as interferon or checkpoint inhibition, might have severe adverse effects in LT recipients186,187.
The value of additional immunosuppression in severe COVID-19: it is unclear as to whether the benefit of dexamethasone that was demonstrated in those with severe COVID-19 applies to those already immunosuppressed133. Equally, the cytokine response to SARS-CoV-2 in LT recipients is undefined.
Immunization
Response to SARS-CoV-2 vaccination: LT recipients typically have reduced rates of seroconversion and reduced antibody titres in response to vaccination188.
Duration of any response to SARS-CoV-2 vaccines: duration of immune response to vaccination might be reduced in LT recipients.
Immunosuppression
Optimal long-term immunosuppressant regimen: whether interindividual variations in immunosuppression affect disease susceptibility is undefined; some have suggested a negative effect of mycophenolate but this issue has not been confirmed115,180.
The characteristics of infection in the immediate post-operative course are undefined: patients in the immediate post-operative period are typically more substantially immunosuppressed (including potentially with depleting antibodies) and might have a different response to SARS-CoV-2.
Challenges and considerations with SARS-CoV-2 vaccination in patients with CLD
SARS-CoV-2 vaccine development has progressed at an unprecedented rate. The Pfizer/BioNTech BNT162b2 mRNA, Moderna mRNA-1273, AstraZeneca/University of Oxford ChAdOx1-nCoV-19 chimpanzee adenovirus vector vaccine and the heterologous recombinant adenovirus-based Gam-COVID-Vac (Sputnik V) have each reported excellent safety profiles and marked efficacy in preventing symptomatic COVID-19 (62–95%)136–139. Despite the inclusion of over 100,000 participants in phase III trials, data in patients with liver disease are limited, representing less than 0.5% of those enrolled. Therefore, as vaccines accelerate into licensing and deployment, it is now vital to consider the specific implications for individual disease states, including CLD140.
Cirrhosis is associated with both systemic inflammation and innate and adaptive immune dysfunction predisposing to infection-related morbidity and mortality141–143. Similarly, patients with cirrhosis have been shown to have impaired responses to existing licensed vaccinations such as those for pneumococcus and hepatitis B virus144,145 (Fig. 3). Whilst it is biologically plausible that patients with cirrhosis might also respond poorly to SARS-CoV-2 vaccination, current data to support this notion are lacking. It is therefore difficult to speculate as to which vaccine type might be most effective for patients with cirrhosis or whether modifications to the standard vaccine dosing or timing will be required. Similarly, the immunogenicity of SARS-CoV-2 vaccines in LT recipients and immunosuppressed patients with autoimmune liver disease remains to be determined, with these groups also known to have attenuated responses to vaccinations against other diseases146.
Neither the adenoviral vector nor the mRNA vaccine platforms contain live or attenuated virus and it therefore seems unlikely that immunization represents a particular safety concern for liver disease cohorts. Whilst historically there have been anxieties that vaccination in solid organ transplant recipients might lead to the development of alloimmunity and graft rejection, no clinical evidence has ever emerged to support this concern146. In light of the high COVID-19-related mortality in patients with decompensated cirrhosis, it remains of utmost importance to prioritize vaccination in this subgroup. Although LT recipients have comparable rates of COVID-19-related mortality to the matched general population117, they do have higher rates of admission to intensive care and could have been relatively more protected throughout the pandemic due to enhanced social distancing. Thus, this group remain a vulnerable population and should also be prioritized for vaccination, with the benefits far outweighing the potential risks. These principles regarding the prioritization of vaccine delivery are reflected in the American Association for the Study of Liver Diseases (AASLD) consensus statement on COVID-19 vaccination in patients with liver disease, published in January 2021 (ref.147).
As COVID-19 vaccines are disseminated globally, it will be crucial to monitor for post-vaccination infection in patients with CLD. This may be facilitated though existing international registry platforms such as COVID-Hep (supported by the European Association for the Study of the Liver) and SECURE-Liver (formerly known as SECURE-Cirrhosis) (supported by the American Association for the Study of Liver Diseases). This real-world data collection should occur in parallel with detailed mechanistic work assessing T cell and humoral immune responses after vaccination in liver subpopulations. Ideally, this should involve a coordinated response from the hepatology community to account for heterogeneity between vaccine candidates, laboratory immune assays and liver disease phenotype.
COVID-19 pandemic on hepatology care
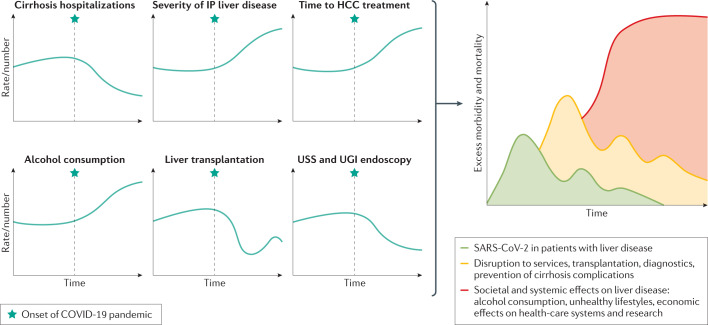
During the early phase of the COVID-19 pandemic, the prevention, control and management of SARS-CoV-2-infected patients rightfully took centre stage and it was therefore reasonable to reduce and postpone services for other non-urgent medical conditions. Nonetheless, such policies will inevitably have collateral downstream effects on patients, including those with CLD. With time, because of the delayed diagnosis and treatment of various liver diseases, there will be escalating morbidity and mortality148 (Fig. 4).
Fig. 4. Trends in liver disease risk factors and management and possible future effect on liver disease incidence and severity.
Trends over time in liver disease risk factors and hepatology care provision in relation to the onset of the coronavirus disease 2019 (COVID-19) pandemic and the cumulative short-term, medium-term and long-term effects this pandemic might have on liver health. HCC, hepatocellular carcinoma; IP, inpatient; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; UGI, upper gastrointestinal; USS, ultrasound scan.
Cirrhosis
In patients with cirrhosis, it is important to treat the underlying liver disease, screen for hepatocellular carcinoma (HCC) and varices, and promptly detect and treat complications of cirrhosis 106,149. All these strategies might be affected during the pandemic. For instance, the delayed initiation of antiviral therapy in patients with chronic viral hepatitis and the relapse of problem drinking could lead to disease progression and decompensating events150. Postponing routine therapeutic paracentesis for tense ascites might convert an elective procedure into one requiring emergency hospitalization. Acute variceal haemorrhage could also develop in patients without timely endoscopic surveillance.
Hepatocellular carcinoma
Currently, both the AASLD and the European Association for the Study of the Liver (EASL) support the resumption of HCC surveillance in high-risk patients (for example, advanced cirrhosis, chronic hepatitis B virus infection) during the COVID-19 pandemic but recognize potential feasibility issues due to the strain on imaging and radiology resources103,104. The AASLD, therefore, recognises that an arbitrary delay of 2 months might be reasonable following the discussion of risks and benefits with the patient104. If HCC surveillance is deferred indefinitely, it is inevitable that the proportion of patients presenting with HCC not amenable to curative treatments will increase151. The proper management of HCC requires input from hepatologists, surgeons, intervention radiologists, oncologists and allied health workers, and therefore the maintenance of multidisciplinary care via telemedicine should be actively pursued. However, a multicentre study within the metropolitan area of Paris, France, during a period of high SARS-CoV-2 infection prevalence demonstrated a statistically significant reduction in the number of HCC diagnoses and double the rate of HCC treatment delay compared with the same period the previous year152. Nevertheless, the HCC treatment modality, including liver surgery, remained unchanged between the two time periods.
Liver transplantation
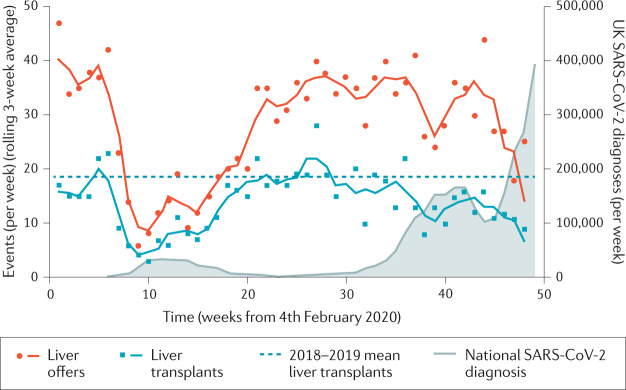
Liver transplantation programmes have been affected by the pandemic in a multitude of ways153,154. First, the recommendation against using organs from deceased donors with SARS-CoV-2 infection is consistent across all major guidelines155. Similarly, to avoid the risk of SARS-CoV-2 exposure and transmission, live donor transplantation has often been reduced or suspended103,104. During times of peak SARS-CoV-2 infection the strain on hospital resources, and particularly ICU availability, has meant that, for certain periods, liver transplantation has been reserved only for super-urgent and highly urgent cases156. Accordingly, data from both the American United Network for Organ Sharing–Organ Procurement Transplant Network and the UK National Health Service Blood and Transplant service show a clear inverse correlation between transplant activity and national rates of SARS-CoV-2 infection, with living donor liver transplantations most affected (Fig. 5). In order to maintain some transplant service provision, the pathways for organ procurement have had to remain flexible, with donors being sent to recipient centres or local organ retrieval and/or surgical teams from recipient centres travelling to donor hospitals.
Fig. 5. UK Liver transplant activity before and during COVID-19 pandemic.
United Kingdom National Health Service Blood and Transplant service data on liver transplant activity before and during the coronavirus disease 2019 (COVID-19) pandemic. The data for Fig. 5 that support the plots within this paper are available from the NHS Blood and Transplant Organ Donation and Transplantation Reports and the Coronavirus (COVID-19) in the UK website. SARS-CoV-2, severe acute respiratory syndrome coronavirus 2.
Elimination of viral hepatitis
In 2017, the WHO published the first Global Hepatitis Report and set the aim to eliminate viral hepatitis as a major public health threat by 2030 (ref.157). Since then, healthcare providers, policy-makers and patients have joined forces to scale-up screening, diagnosis, assessment and treatment of viral hepatitis. During the COVID-19 pandemic, although different countries have adopted measures, such as telemedicine and automatic drug refill, to ensure the continuation of current antiviral therapies, there has been a major impact on new case identification and treatment initiation. In a modelling study for 110 countries/territories, a 1-year delay in the progress of the hepatitis elimination programme is estimated to increase the number of HCC and liver-related deaths by 44,800 and 72,300, respectively158. However, despite these understandable concerns, there could be some positive ways to harness the widespread practical changes forced by the pandemic. For example, the decentralization of elimination efforts through postal blood-spot testing and medication delivery might have sustainable long-term benefits. Furthermore, political and health-care systems are now far more familiar with mass testing strategies, contact tracing and vaccination delivery. There might now be an opportunity to harness this infrastructure to help combat chronic viral hepatitis at a population level159. This progress will also go hand-in-hand with other adaptations to health-care delivery such as telemedicine, remote monitoring and greater interaction with caregivers at home. These all represent promising opportunities to help deliver good hepatology care not only for viral hepatitis but across the spectrum of liver disease type and severity160.
Liver research
COVID-19 has had a major effect on liver research, especially clinical trials, due to uncertainties surrounding infection control and difficulties adhering to the study protocols161. The follow-up, assessment and distribution of investigational products could all be hampered by quarantine measures and participant illness. For these reasons, many sponsors have stopped new case recruitment into clinical trials, which is likely to delay important drug development. It is therefore currently important to consider flexible solutions such as replacing less critical visits with phone or video calls, remote monitoring using local laboratories, and direct shipment of investigational products to the patients. There are also concerns about research funding due to economic downturn in the next few years162. The negative impact of the pandemic on existing non-COVID-19 basic and translational research must also not be underestimated. Institutions across the globe have endured major research set-backs, including laboratory closures, diversion of resources towards COVID-19 studies, and the loss of cell lines and animal models during national lockdowns163. Furthermore, the cancellation or postponement of national and international conferences has disrupted the valuable exchange of scientific information. In recovering from the COVID-19 pandemic it is therefore vital that policy and funding is structured in a way that helps rejuvenate basic science research activity.
COVID-19 pandemic and patient behaviours
The COVID-19 pandemic has understandably modified human behaviour beyond recognition. However, some of these behaviours could negatively influence liver health, particularly with respect to alcohol use, diet and exercise, and patient interactions with medical services.
An increased alcohol consumption is now a well-recognized feature of the pandemic, particularly during periods of social isolation. In the weeks leading up the first national ‘lockdown’ in the UK, an additional £160 million were spent stockpiling alcohol compared with the same timeframe the previous year164,165. Furthermore, nationwide consumer data from the UK have shown a pervasive elevation in alcohol purchasing amongst all but the very poorest in society166,167. Data on alcohol sales have shown similar trends in the USA168. This change in behaviour has translated into harmful drinking habits, with national and international surveys consistently demonstrating an increase in regular alcohol use during the first wave of COVID-19 (refs169–171) and an overall escalation in alcohol consumption reported in up to 48% of respondents170. In addition, 17% of abstinent individuals with a history of alcohol use disorder were found to relapse to drinking under lockdown conditions172. This finding contrasts with a decline in smoking, mostly due to a reduction in the number of lighter smokers169. Single-centre observations in the UK have also shown that referrals for alcoholic liver disease (ALD) and the proportion of critically unwell inpatients with ALD (but without COVID-19) more than doubled in June 2020 compared with June 2019 (ref.173). The upsurge in harmful drinking, alongside barriers to cessation services and the association between ALD and COVID-19 mortality (discussed earlier) is of major concern and suggests that alcohol advice should be central to the health messages delivered to patients and the general public during the pandemic.
The pandemic has also propagated unhealthy lifestyles predisposing to NAFLD. A study in northern Italy found that patients with obesity gained an average of 1.5 kg in weight during the national quarantine, in parallel with reduced exercise, excess calory intake, and increased self-reported anxiety and depression174. Similarly, a US survey of over 4,000 patients with diabetes found that one-third reported a less healthy diet and half reported performing less exercise during the pandemic175. Despite these observations, no study has yet directly evaluated the incidence of NAFLD in the COVID-19 era.
Lastly, the interaction of patients with secondary care services has changed. National US data from the Veterans Health Administration showed that that hospitalizations with cirrhosis decreased by a third following the onset of the pandemic compared with the previous year, with a significant increase in the severity of liver disease in those who were admitted176. This might reflect patient concerns about potential SARS-CoV-2 exposure leading to delayed presentations with more advanced complications of cirrhosis.
Conclusions
The hepatic consequences of SARS-CoV-2 infection are now recognized as an important component of COVID-19. This aspect is most clinically relevant in patients with pre-existing cirrhosis who are at remarkably high risk of severe COVID-19 and death. Whilst further work is required to understand the pathogenic mechanisms that drive this clinical deterioration, there are likely to be contributions from systemic inflammation, disordered coagulation and immune dysfunction. Although a range of in vitro and in vivo models have been used to help decipher the specific hepatotropism of SARS-CoV-2, the clinical impact of direct viral infection of liver cell types remains to be determined. The grave prognosis with COVID-19 in patients with cirrhosis contrasts with the LT population who have comparably better outcomes. It therefore seems that cirrhosis-associated immune dysfunction has a far more detrimental effect on the COVID-19 course than pharmacological immunosuppression. With efficacious SARS-CoV-2 vaccines now available, patients with cirrhosis should be seen as a priority for immunization and the hepatology community should prepare to carefully monitor the immune response in this subpopulation. Lastly, we must all be aware of the profound negative effect of the pandemic on liver services and unhealthy patient behaviours, which might culminate in an increase in the global burden of liver disease in the coming months and years.
Acknowledgements
T.M., G.J.W. and E.B. have received registry grant funding from the European Association for Study of the Liver (EASL) (grant number 2020RG03). T.M. is funded via a Wellcome Trust Clinical Research Fellowship (grant number 102176/B/13/Z). A.M.M. has received grant funding from the National Institutes of Health (grant number T32 DK007634). V.W.W. has received a grant funding from Gilead Sciences for fatty liver research. Z.S. is supported by the Medical Research Foundation (grant number MRF-169-0001-F-STAM-C0826) E.B. is supported by the Oxford National Institute for Health Research (NIHR) Biomedical Research Centre and is an NIHR Senior Investigator. The views expressed in this article are those of the authors and not necessarily those of the NHS, the NIHR or the UK Department of Health.
Author contributions
T.M., G.J.W. and E.B. were responsible for the review’s concept and design. All authors contributed equally to the writing and critical revision of the manuscript.
Data availability
The UK Liver transplant and coronavirus data that support the plots within this paper are available from the NHS Blood and Transplant Organ Donation and Transplantation Reports website at https://www.odt.nhs.uk/statistics-and-reports/ and the Coronavirus (COVID-19) in the UK website at https://coronavirus.data.gov.uk/.
Competing interests
A.S.B. has served as a consultant to Target RWE, Genfit and Intercept Pharmaceuticals. V.W.W. has served as a consultant or advisory board member for 3V-BIO, AbbVie, Allergan, Boehringer Ingelheim, Center for Outcomes Research in Liver Diseases, Echosens, Gilead Sciences, Hanmi Pharmaceutical, Intercept, Merck, Novartis, Novo Nordisk, Perspectum Diagnostics, Pfizer, ProSciento, Sagimet Biosciences, Taerget RWE, and Terns and as a speaker for AbbVie, Bristol-Myers Squibb, Echosens and Gilead Sciences. The remaining authors declare no competing interests.
Footnotes
Peer review information
Nature Reviews Gastroenterology & Hepatology thanks M. Cornberg, M. Schilsky and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Coronavirus (COVID-19) in the UK: https://coronavirus.data.gov.uk/
COVID-Hep Registry: https://www.covid-hep.net/
NHS Blood and Transplant Organ Donation and Transplantation Reports: https://www.odt.nhs.uk/statistics-and-reports/
SECURE-Liver (formerly SECURE-Cirrhosis) Registry: https://covidcirrhosis.web.unc.edu/
Contributor Information
Thomas Marjot, Email: thomas.marjot@ndm.ox.ac.uk.
Eleanor Barnes, Email: ellie.barnes@ndm.ox.ac.uk.
References
- 1.COVID-19 Dashboard by the Center for Systems Science and Engineering (CSSE) at Johns Hopkins University (JHU) https://coronavirus.jhu.edu/map.html (2021).
- 2.Berlin DA, Gulick RM, Martinez FJ. Severe Covid-19. N. Engl. J. Med. 2020;383:2451–2460. doi: 10.1056/NEJMcp2009575. [DOI] [PubMed] [Google Scholar]
- 3.Tay MZ, Poh CM, Renia L, MacAry PA, Ng LFP. The trinity of COVID-19: immunity, inflammation and intervention. Nat. Rev. Immunol. 2020;20:363–374. doi: 10.1038/s41577-020-0311-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.World Health Organization. Clinical Management of COVID-19: Interim Guidance (2020).
- 5.Williamson EJ, et al. Factors associated with COVID-19-related death using OpenSAFELY. Nature. 2020;584:430–436. doi: 10.1038/s41586-020-2521-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ioannou GN, et al. Risk factors for hospitalization, mechanical ventilation, or death among 10131 US veterans with SARS-CoV-2 infection. JAMA Netw. Open. 2020;3:e2022310. doi: 10.1001/jamanetworkopen.2020.22310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Group RC, et al. Dexamethasone in hospitalized patients with Covid-19 - preliminary report. N. Engl. J. Med. 2020 doi: 10.1056/NEJMoa2021436. [DOI] [Google Scholar]
- 8.Wiersinga WJ, Rhodes A, Cheng AC, Peacock SJ, Prescott HC. Pathophysiology, transmission, diagnosis, and treatment of coronavirus disease 2019 (COVID-19): a review. JAMA. 2020;324:782–793. doi: 10.1001/jama.2020.12839. [DOI] [PubMed] [Google Scholar]
- 9.Albillos A, Lario M, Álvarez-Mon M. Cirrhosis-associated immune dysfunction: distinctive features and clinical relevance. J. Hepatol. 2014;61:1385–1396. doi: 10.1016/j.jhep.2014.08.010. [DOI] [PubMed] [Google Scholar]
- 10.Tapper EB, Robson SC, Malik R. Coagulopathy in cirrhosis - the role of the platelet in hemostasis. J. Hepatol. 2013;59:889–890. doi: 10.1016/j.jhep.2013.03.040. [DOI] [PubMed] [Google Scholar]
- 11.Collaborators GBDC. The global, regional, and national burden of cirrhosis by cause in 195 countries and territories, 1990-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet Gastroenterol. Hepatol. 2020;5:245–266. doi: 10.1016/S2468-1253(19)30349-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Pirola CJ, Sookoian S. SARS-CoV-2 virus and liver expression of host receptors: putative mechanisms of liver involvement in COVID-19. Liver Int. 2020;40:2038–2040. doi: 10.1111/liv.14500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Qi F, Qian S, Zhang S, Zhang Z. Single cell RNA sequencing of 13 human tissues identify cell types and receptors of human coronaviruses. Biochem. Biophys. Res. Commun. 2020;526:135–140. doi: 10.1016/j.bbrc.2020.03.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.De Smet V, Verhulst S, van Grunsven LA. Single cell RNA sequencing analysis did not predict hepatocyte infection by SARS-CoV-2. J. Hepatol. 2020;73:993–995. doi: 10.1016/j.jhep.2020.05.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Chu H, et al. Comparative tropism, replication kinetics, and cell damage profiling of SARS-CoV-2 and SARS-CoV with implications for clinical manifestations, transmissibility, and laboratory studies of COVID-19: an observational study. Lancet Microbe. 2020;1:e14–e23. doi: 10.1016/S2666-5247(20)30004-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ma-Lauer Y, et al. p53 down-regulates SARS coronavirus replication and is targeted by the SARS-unique domain and PLpro via E3 ubiquitin ligase RCHY1. Proc. Natl Acad. Sci. USA. 2016;113:E5192–E5201. doi: 10.1073/pnas.1603435113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zhao B, et al. Recapitulation of SARS-CoV-2 infection and cholangiocyte damage with human liver ductal organoids. Protein Cell. 2020;11:771–775. doi: 10.1007/s13238-020-00718-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Gaebler C, et al. Evolution of antibody immunity to SARS-CoV-2. Nature. 2021 doi: 10.1038/s41586-021-03207-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Yang L, et al. A human pluripotent stem cell-based platform to study SARS-CoV-2 tropism and model virus infection in human cells and organoids. Cell Stem Cell. 2020;27:125–136.e7. doi: 10.1016/j.stem.2020.06.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Paizis G, et al. Chronic liver injury in rats and humans upregulates the novel enzyme angiotensin converting enzyme 2. Gut. 2005;54:1790–1796. doi: 10.1136/gut.2004.062398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Fondevila MF, et al. Obese patients with NASH have increased hepatic expression of SARS-CoV-2 critical entry points. J. Hepatol. 2021;74:469–471. doi: 10.1016/j.jhep.2020.09.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Herath CB, et al. Upregulation of hepatic angiotensin-converting enzyme 2 (ACE2) and angiotensin-(1-7) levels in experimental biliary fibrosis. J. Hepatol. 2007;47:387–395. doi: 10.1016/j.jhep.2007.03.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Chua RL, et al. COVID-19 severity correlates with airway epithelium-immune cell interactions identified by single-cell analysis. Nat. Biotechnol. 2020;38:970–979. doi: 10.1038/s41587-020-0602-4. [DOI] [PubMed] [Google Scholar]
- 24.Ziegler CGK, et al. SARS-CoV-2 receptor ACE2 is an interferon-stimulated gene in human airway epithelial cells and is detected in specific cell subsets across tissues. Cell. 2020;181:1016–1035.e19. doi: 10.1016/j.cell.2020.04.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Onabajo OO, et al. Interferons and viruses induce a novel truncated ACE2 isoform and not the full-length SARS-CoV-2 receptor. Nat. Genet. 2020;52:1283–1293. doi: 10.1038/s41588-020-00731-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wei C, et al. HDL-scavenger receptor B type 1 facilitates SARS-CoV-2 entry. Nat. Metab. 2020;2:1391–1400. doi: 10.1038/s42255-020-00324-0. [DOI] [PubMed] [Google Scholar]
- 27.Grove J, et al. Scavenger receptor BI and BII expression levels modulate hepatitis C virus infectivity. J. Virol. 2007;81:3162–3169. doi: 10.1128/JVI.02356-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Zuo T, et al. Depicting SARS-CoV-2 faecal viral activity in association with gut microbiota composition in patients with COVID-19. Gut. 2021;70:276–284. doi: 10.1136/gutjnl-2020-322294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lamers MM, et al. SARS-CoV-2 productively infects human gut enterocytes. Science. 2020;369:50–54. doi: 10.1126/science.abc1669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Qian Q, et al. Direct evidence of active SARS-CoV-2 replication in the intestine. Clin. Infect. Dis. 2020 doi: 10.1093/cid/ciaa925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Sonzogni A, et al. Liver histopathology in severe COVID 19 respiratory failure is suggestive of vascular alterations. Liver Int. 2020;40:2110–2116. doi: 10.1111/liv.14601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Wang Y, et al. SARS-CoV-2 infection of the liver directly contributes to hepatic impairment in patients with COVID-19. J. Hepatol. 2020;73:807–816. doi: 10.1016/j.jhep.2020.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Gordon DE, et al. A SARS-CoV-2 protein interaction map reveals targets for drug repurposing. Nature. 2020;583:459–468. doi: 10.1038/s41586-020-2286-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Nie X, et al. Multi-organ proteomic landscape of COVID-19 autopsies. Cell. 2021;184:775–791.e14. doi: 10.1016/j.cell.2021.01.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Guan WJ, et al. Clinical characteristics of coronavirus disease 2019 in China. N. Engl. J. Med. 2020;382:1708–1720. doi: 10.1056/NEJMoa2002032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Sultan S, et al. AGA institute rapid review of the gastrointestinal and liver manifestations of COVID-19, meta-analysis of international data, and recommendations for the consultative management of patients with COVID-19. Gastroenterology. 2020;159:320–334.e27. doi: 10.1053/j.gastro.2020.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Richardson S, et al. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York city area. JAMA. 2020;323:2052–2059. doi: 10.1001/jama.2020.6775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Goyal P, et al. Clinical characteristics of Covid-19 in New York city. N. Engl. J. Med. 2020;382:2372–2374. doi: 10.1056/NEJMc2010419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Youssef M, et al. COVID-19 and liver dysfunction: a systematic review and meta-analysis of retrospective studies. J. Med. Virol. 2020;92:1825–1833. doi: 10.1002/jmv.26055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Hundt MA, Deng Y, Ciarleglio MM, Nathanson MH, Lim JK. Abnormal liver tests in COVID-19: a retrospective observational cohort study of 1827 patients in a major US Hospital Network. Hepatology. 2020;72:1169–1176. doi: 10.1002/hep.31487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Elmunzer BJ, et al. Digestive manifestations in patients hospitalized with COVID-19. Clin. Gastroenterol. Hepatol. 2020 doi: 10.1016/j.cgh.2020.09.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Fu Y, et al. Clinical features of COVID-19-infected patients with elevated liver biochemistries: a multicenter, retrospective study. Hepatology. 2020 doi: 10.1002/hep.31446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Mao R, et al. Manifestations and prognosis of gastrointestinal and liver involvement in patients with COVID-19: a systematic review and meta-analysis. Lancet Gastroenterol. Hepatol. 2020;5:667–678. doi: 10.1016/S2468-1253(20)30126-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Phipps MM, et al. Acute liver injury in COVID-19: prevalence and association with clinical outcomes in a large US cohort. Hepatology. 2020;72:807–817. doi: 10.1002/hep.31404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Zhou F, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395:1054–1062. doi: 10.1016/S0140-6736(20)30566-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Singh S, Khan A. Clinical characteristics and outcomes of coronavirus disease 2019 among patients with preexisting liver disease in the united states: a multicenter research network study. Gastroenterology. 2020;159:768–771.e3. doi: 10.1053/j.gastro.2020.04.064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Xu Z, et al. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir. Med. 2020;8:420–422. doi: 10.1016/S2213-2600(20)30076-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Lagana SM, et al. Hepatic pathology in patients dying of COVID-19: a series of 40 cases including clinical, histologic, and virologic data. Mod. Pathol. 2020;33:2147–2155. doi: 10.1038/s41379-020-00649-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Jothimani D, Venugopal R, Abedin MF, Kaliamoorthy I, Rela M. COVID-19 and the liver. J. Hepatol. 2020;73:1231–1240. doi: 10.1016/j.jhep.2020.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Bloom PP, et al. Liver biochemistries in hospitalized patients with COVID-19. Hepatology. 2020 doi: 10.1002/hep.31326. [DOI] [PubMed] [Google Scholar]
- 51.Buckholz AP, Kaplan A, Rosenblatt RE, Wan D. Clinical characteristics, diagnosis, and outcomes of 6 patients with COVID-19 infection and rhabdomyolysis. Mayo Clin. Proc. 2020;95:2557–2559. doi: 10.1016/j.mayocp.2020.09.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Zhang Y, et al. Coagulopathy and antiphospholipid antibodies in patients with Covid-19. N. Engl. J. Med. 2020;382:e38. doi: 10.1056/NEJMc2007575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Díaz LA, et al. High prevalence of hepatic steatosis and vascular thrombosis in COVID-19: a systematic review and meta-analysis of autopsy data. World J. Gastroenterol. 2020;26:7693–7706. doi: 10.3748/wjg.v26.i48.7693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Papic N, et al. Liver involvement during influenza infection: perspective on the 2009 influenza pandemic. Influenza Other Respir. Viruses. 2012;6:e2–e5. doi: 10.1111/j.1750-2659.2011.00287.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Mehta P, et al. COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet. 2020;395:1033–1034. doi: 10.1016/S0140-6736(20)30628-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Da BL, et al. Liver injury in hospitalized patients with COVID-19 correlates with hyper inflammatory response and elevated IL-6. Hepatol. Commun. 2020;5:177–188. doi: 10.1002/hep4.1631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Liu J, et al. Longitudinal characteristics of lymphocyte responses and cytokine profiles in the peripheral blood of SARS-CoV-2 infected patients. EBioMedicine. 2020;55:102763. doi: 10.1016/j.ebiom.2020.102763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Diao B, et al. Reduction and functional exhaustion of T cells in patients with coronavirus disease 2019 (COVID-19) Front. Immunol. 2020;11:827. doi: 10.3389/fimmu.2020.00827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Al-Samkari H, et al. COVID-19 and coagulation: bleeding and thrombotic manifestations of SARS-CoV-2 infection. Blood. 2020;136:489–500. doi: 10.1182/blood.2020006520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Klok FA, et al. Confirmation of the high cumulative incidence of thrombotic complications in critically ill ICU patients with COVID-19: an updated analysis. Thromb. Res. 2020;191:148–150. doi: 10.1016/j.thromres.2020.04.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Middeldorp S, et al. Incidence of venous thromboembolism in hospitalized patients with COVID-19. J. Thromb. Haemost. 2020;18:1995–2002. doi: 10.1111/jth.14888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Poissy J, et al. Pulmonary embolism in patients with COVID-19: awareness of an increased prevalence. Circulation. 2020;142:184–186. doi: 10.1161/CIRCULATIONAHA.120.047430. [DOI] [PubMed] [Google Scholar]
- 63.Olry A, et al. Drug-induced liver injury and COVID-19 infection: the rules remain the same. Drug Saf. 2020;43:615–617. doi: 10.1007/s40264-020-00954-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Cao B, et al. A trial of lopinavir-ritonavir in adults hospitalized with severe COVID-19. N. Engl. J. Med. 2020;382:1787–1799. doi: 10.1056/NEJMoa2001282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Cai Q, et al. COVID-19: abnormal liver function tests. J. Hepatol. 2020;73:566–574. doi: 10.1016/j.jhep.2020.04.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Muhovic D, et al. First case of drug-induced liver injury associated with the use of tocilizumab in a patient with COVID-19. Liver Int. 2020;40:1901–1905. doi: 10.1111/liv.14516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Mahamid M, Mader R, Safadi R. Hepatotoxicity of tocilizumab and anakinra in rheumatoid arthritis: management decisions. Clin. Pharmacol. 2011;3:39–43. doi: 10.2147/CPAA.S24004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Beigel JH, et al. Remdesivir for the treatment of Covid-19 - final report. N. Engl. J. Med. 2020;383:1813–1826. doi: 10.1056/NEJMoa2007764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Montastruc F, Thuriot S, Durrieu G. Hepatic disorders with the use of remdesivir for coronavirus 2019. Clin. Gastroenterol. Hepatol. 2020;18:2835–2836. doi: 10.1016/j.cgh.2020.07.050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.WHO Solidarity Trial Consortium, et al. Repurposed antiviral drugs for Covid-19 - Interim WHO Solidarity Trial Results. N. Engl. J. Med. 2021;384:497–511. doi: 10.1056/NEJMoa2023184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Ponziani FR, et al. Liver involvement is not associated with mortality: results from a large cohort of SARS-CoV-2 positive patients. Aliment. Pharmacol. Ther. 2020;52:1060–1068. doi: 10.1111/apt.15996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Yip TCF, et al. Liver injury is independently associated with adverse clinical outcomes in patients with COVID-19. Gut. 2020 doi: 10.1136/gutjnl-2020-321726. [DOI] [PubMed] [Google Scholar]
- 73.Weber S, et al. Liver function test abnormalities at hospital admission are associated with severe course of SARS-CoV-2 infection: a prospective cohort study. Gut. 2021 doi: 10.1136/gutjnl-2020-323800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Ding ZY, et al. Association of liver abnormalities with in-hospital mortality in patients with COVID-19. J. Hepatol. 2020 doi: 10.1016/j.jhep.2020.12.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Zhang Y, et al. Liver impairment in COVID-19 patients: a retrospective analysis of 115 cases from a single centre in Wuhan city, China. Liver Int. 2020;40:2095–2103. doi: 10.1111/liv.14455. [DOI] [PubMed] [Google Scholar]
- 76.Yadav DK, et al. Involvement of liver in COVID-19: systematic review and meta-analysis. Gut. 2020 doi: 10.1136/gutjnl-2020-322072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Lei F, et al. Longitudinal association between markers of liver injury and mortality in COVID-19 in China. Hepatology. 2020;72:389–398. doi: 10.1002/hep.31301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Bangash MN, et al. SARS-CoV-2: is the liver merely a bystander to severe disease? J. Hepatol. 2020;73:995–996. doi: 10.1016/j.jhep.2020.05.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Noor MT, Manoria P. Immune dysfunction in cirrhosis. J. Clin. Transl. Hepatol. 2017;5:50–58. doi: 10.14218/JCTH.2016.00056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Pose E, et al. PD-L1 is overexpressed in liver macrophages in chronic liver diseases and its blockade improves the antibacterial activity against infections. Hepatology. 2020 doi: 10.1002/hep.31644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Piano S, Brocca A, Mareso S, Angeli P. Infections complicating cirrhosis. Liver Int. 2018;38(Suppl. 1):126–133. doi: 10.1111/liv.13645. [DOI] [PubMed] [Google Scholar]
- 82.Fan VS, et al. Risk factors for testing positive for SARS-CoV-2 in a national US healthcare system. Clin. Infect. Dis. 2020 doi: 10.1093/cid/ciaa1624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Ioannou GN, et al. Cirrhosis and SARS-CoV-2 infection in US veterans: risk of infection, hospitalization, ventilation and mortality. Hepatology. 2020 doi: 10.1002/hep.31649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Fine MJ, et al. A prediction rule to identify low-risk patients with community-acquired pneumonia. N. Engl. J. Med. 1997;336:243–250. doi: 10.1056/NEJM199701233360402. [DOI] [PubMed] [Google Scholar]
- 85.Schütte A, Ciesek S, Wedemeyer H, Lange CM. Influenza virus infection as precipitating event of acute-on-chronic liver failure. J. Hepatol. 2019;70:797–799. doi: 10.1016/j.jhep.2018.11.015. [DOI] [PubMed] [Google Scholar]
- 86.Eslam M, et al. A new definition for metabolic dysfunction-associated fatty liver disease: an international expert consensus statement. J. Hepatol. 2020;73:202–209. doi: 10.1016/j.jhep.2020.03.039. [DOI] [PubMed] [Google Scholar]
- 87.Ji D, et al. Non-alcoholic fatty liver diseases in patients with COVID-19: a retrospective study. J. Hepatol. 2020;73:451–453. doi: 10.1016/j.jhep.2020.03.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Zhou YJ, et al. Younger patients with MAFLD are at increased risk of severe COVID-19 illness: a multicenter preliminary analysis. J. Hepatol. 2020;73:719–721. doi: 10.1016/j.jhep.2020.04.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Roca-Fernandez A, et al. High liver fat associates with higher risk of developing symptomatic covid-19 infection - initial UK biobank observations. medRxiv. 2020 doi: 10.1101/2020.06.04.20122457. [DOI] [Google Scholar]
- 90.Lopez-Mendez I, et al. Association of liver steatosis and fibrosis with clinical outcomes in patients with SARS-CoV-2 infection (COVID-19) Ann. Hepatol. 2021;20:100271. doi: 10.1016/j.aohep.2020.09.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Marjot T, et al. Outcomes following SARS-CoV-2 infection in patients with chronic liver disease: an international registry study. J. Hepatol. 2020;74:567–577. doi: 10.1016/j.jhep.2020.09.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Marjot T, et al. SARS-CoV-2 infection in patients with autoimmune hepatitis. J. Hepatol. 2021 doi: 10.1016/j.jhep.2021.01.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Iavarone M, et al. High rates of 30-day mortality in patients with cirrhosis and COVID-19. J. Hepatol. 2020;73:1063–1071. doi: 10.1016/j.jhep.2020.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Bajaj JS, et al. Cirrhosis is associated with high mortality and readmissions over 90 days regardless of COVID-19: a multi-center cohort. Liver Transpl. 2021 doi: 10.1002/lt.25981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Wu T, et al. Development of diagnostic criteria and a prognostic score for hepatitis B virus-related acute-on-chronic liver failure. Gut. 2018;67:2181–2191. doi: 10.1136/gutjnl-2017-314641. [DOI] [PubMed] [Google Scholar]
- 96.Moreau R, et al. Acute-on-chronic liver failure is a distinct syndrome that develops in patients with acute decompensation of cirrhosis. Gastroenterology. 2013;144:1426–1437.e9. doi: 10.1053/j.gastro.2013.02.042. [DOI] [PubMed] [Google Scholar]
- 97.Horwitz LI, et al. Trends in COVID-19 risk-adjusted mortality rates. J. Hosp. Med. 2021;16:90–92. doi: 10.12788/jhm.3552. [DOI] [PubMed] [Google Scholar]
- 98.Duddempudi AT. Immunology in alcoholic liver disease. Clin. Liver Dis. 2012;16:687–698. doi: 10.1016/j.cld.2012.08.003. [DOI] [PubMed] [Google Scholar]
- 99.Yeoh YK, et al. Gut microbiota composition reflects disease severity and dysfunctional immune responses in patients with COVID-19. Gut. 2021 doi: 10.1136/gutjnl-2020-323020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Bajaj JS. Altered microbiota in cirrhosis and its relationship to the development of infection. Clin. Liver Dis. 2019;14:107–111. doi: 10.1002/cld.827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Adeniji N, et al. Socioeconomic factors contribute to the higher risk of COVID-19 in racial and ethnic minorities with chronic liver diseases (CLD) Gastroenterology. 2020 doi: 10.1053/j.gastro.2020.11.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Wegermann K, et al. Racial and socioeconomic disparities in utilization of telehealth in patients with liver disease during COVID-19. Dig. Dis. Sci. 2021 doi: 10.1007/s10620-021-06842-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Boettler T, et al. Impact of COVID-19 on the care of patients with liver disease: EASL-ESCMID position paper after 6 months of the pandemic. JHEP Rep. 2020;2:100169. doi: 10.1016/j.jhepr.2020.100169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Fix OK, et al. Clinical best practice advice for hepatology and liver transplant providers during the COVID-19 pandemic: AASLD expert panel consensus statement. Hepatology. 2020;72:287–304. doi: 10.1002/hep.31281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.APASL Covid-19 Task Force. Lau G, Sharma M. Clinical practice guidance for hepatology and liver transplant providers during the COVID-19 pandemic: APASL expert panel consensus recommendations. Hepatol. Int. 2020;14:415–428. doi: 10.1007/s12072-020-10054-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Angeli P, et al. EASL clinical practice guidelines for the management of patients with decompensated cirrhosis. J. Hepatol. 2018;69:406–460. doi: 10.1016/j.jhep.2018.03.024. [DOI] [PubMed] [Google Scholar]
- 107.Manolis AS, Manolis TA, Manolis AA, Papatheou D, Melita H. COVID-19 infection: viral macro- and micro-vascular coagulopathy and thromboembolism/prophylactic and therapeutic management. J. Cardiovasc. Pharmacol. Ther. 2021;26:12–24. doi: 10.1177/1074248420958973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Bikdeli B, et al. COVID-19 and thrombotic or thromboembolic disease: implications for prevention, antithrombotic therapy, and follow-up: JACC state-of-the-art review. J. Am. Coll. Cardiol. 2020;75:2950–2973. doi: 10.1016/j.jacc.2020.04.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Turco L, de Raucourt E, Valla DC, Villa E. Anticoagulation in the cirrhotic patient. JHEP Reports. 2019;1:227–239. doi: 10.1016/j.jhepr.2019.02.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Loffredo L, Pastori D, Farcomeni A, Violi F. Effects of anticoagulants in patients with cirrhosis and portal vein thrombosis: a systematic review and meta-analysis. Gastroenterology. 2017;153:480–487.e1. doi: 10.1053/j.gastro.2017.04.042. [DOI] [PubMed] [Google Scholar]
- 111.Tritschler T, et al. Anticoagulant interventions in hospitalized patients with COVID-19: a scoping review of randomized controlled trials and call for international collaboration. J. Thromb. Haemost. 2020;18:2958–2967. doi: 10.1111/jth.15094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Lemos ACB, et al. Therapeutic versus prophylactic anticoagulation for severe COVID-19: a randomized phase II clinical trial (HESACOVID) Thromb. Res. 2020;196:359–366. doi: 10.1016/j.thromres.2020.09.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Watt KD. Keys to long-term care of the liver transplant recipient. Nat. Rev. Gastroenterol. Hepatol. 2015;12:639–648. doi: 10.1038/nrgastro.2015.172. [DOI] [PubMed] [Google Scholar]
- 114.Benitez C, et al. Prospective multicenter clinical trial of immunosuppressive drug withdrawal in stable adult liver transplant recipients. Hepatology. 2013;58:1824–1835. doi: 10.1002/hep.26426. [DOI] [PubMed] [Google Scholar]
- 115.Colmenero J, et al. Epidemiological pattern, incidence and outcomes of COVID-19 in liver transplant patients. J. Hepatol. 2021;74:148–155. doi: 10.1016/j.jhep.2020.07.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Ravanan R, et al. SARS-CoV-2 infection and early mortality of wait-listed and solid organ transplant recipients in England: a national cohort study. Am. J. Transplant. 2020;20:3008–3018. doi: 10.1111/ajt.16247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Webb GJ, et al. Outcomes following SARS-CoV-2 infection in liver transplant recipients: an international registry study. Lancet Gastroenterol. Hepatol. 2020;5:1008–1016. doi: 10.1016/S2468-1253(20)30271-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Rauber C, et al. SARS-CoV-2 seroprevalence and clinical features of COVID-19 in a German liver transplant recipient cohort: a prospective serosurvey study. Transplant. Proc. 2020 doi: 10.1016/j.transproceed.2020.11.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Lee BT, et al. COVID-19 in liver transplant recipients: an initial experience from the US epicenter. Gastroenterology. 2020;159:1176–1178.e2. doi: 10.1053/j.gastro.2020.05.050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Pereira MR, et al. COVID-19 in solid organ transplant recipients: initial report from the US epicenter. Am. J. Transplant. 2020;20:1800–1808. doi: 10.1111/ajt.15941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Mathur AK, Schaubel DE, Gong Q, Guidinger MK, Merion RM. Sex-based disparities in liver transplant rates in the United States. Am. J. Transplant. 2011;11:1435–1443. doi: 10.1111/j.1600-6143.2011.03498.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Sharma P, et al. Impact of MELD-based allocation on end-stage renal disease after liver transplantation. Am. J. Transplant. 2011;11:2372–2378. doi: 10.1111/j.1600-6143.2011.03703.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Lieber SR, et al. The impact of post-transplant diabetes mellitus on liver transplant outcomes. Clin. Transplant. 2019;33:e13554. doi: 10.1111/ctr.13554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Everhart JE, et al. Weight change and obesity after liver transplantation: incidence and risk factors. Liver Transpl. Surg. 1998;4:285–296. doi: 10.1002/lt.500040402. [DOI] [PubMed] [Google Scholar]
- 125.Su F, et al. Aging of liver transplant registrants and recipients: trends and impact on waitlist outcomes, post-transplantation outcomes, and transplant-related survival benefit. Gastroenterology. 2016;150:441–453.e6. doi: 10.1053/j.gastro.2015.10.043. [DOI] [PubMed] [Google Scholar]
- 126.Seyam M, Neuberger JM, Gunson BK, Hubscher SG. Cirrhosis after orthotopic liver transplantation in the absence of primary disease recurrence. Liver Transpl. 2007;13:966–974. doi: 10.1002/lt.21060. [DOI] [PubMed] [Google Scholar]
- 127.Rabiee A, et al. Liver injury in liver transplant recipients with coronavirus disease 2019 (COVID-19): US multicenter experience. Hepatology. 2020 doi: 10.1002/hep.31574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Clift AK, et al. Living risk prediction algorithm (QCOVID) for risk of hospital admission and mortality from coronavirus 19 in adults: national derivation and validation cohort study. BMJ. 2020;371:m3731. doi: 10.1136/bmj.m3731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Webb GJ, Moon AM, Barnes E, Barritt AS, Marjot T. Age and comorbidity are central to the risk of death from COVID-19 in liver transplant recipients. J. Hepatol. 2021 doi: 10.1016/j.jhep.2021.01.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Del Valle DM, et al. An inflammatory cytokine signature predicts COVID-19 severity and survival. Nat. Med. 2020;26:1636–1643. doi: 10.1038/s41591-020-1051-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Zhang Q, et al. Inborn errors of type I IFN immunity in patients with life-threatening COVID-19. Science. 2020 doi: 10.1126/science.abd4570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Bastard P, et al. Autoantibodies against type I IFNs in patients with life-threatening COVID-19. Science. 2020 doi: 10.1126/science.abd4585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.RECOVERY Collaborative Group et al. Dexamethasone in hospitalized patients with Covid-19 - preliminary report. N. Engl. J. Med. 2020 doi: 10.1056/NEJMoa2021436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.D’Antiga L. Coronaviruses and immunosuppressed patients: the facts during the third epidemic. Liver Transpl. 2020;26:832–834. doi: 10.1002/lt.25756. [DOI] [PubMed] [Google Scholar]
- 135.Boettler T, et al. Care of patients with liver disease during the COVID-19 pandemic: EASL-ESCMID position paper. JHEP Rep. 2020;2:100113. doi: 10.1016/j.jhepr.2020.100113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Polack FP, et al. Safety and efficacy of the BNT162b2 mRNA Covid-19 vaccine. N. Engl. J. Med. 2020;383:2603–2615. doi: 10.1056/NEJMoa2034577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Voysey M, et al. Safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: an interim analysis of four randomised controlled trials in Brazil, South Africa, and the UK. Lancet. 2021;397:99–111. doi: 10.1016/S0140-6736(20)32661-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Baden LR, et al. Efficacy and safety of the mRNA-1273 SARS-CoV-2 vaccine. N. Engl. J. Med. 2021;384:403–416. doi: 10.1056/NEJMoa2035389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Logunov DY. Safety and efficacy of an rAd26 and rAd5 vector-based heterologous prime-boost COVID-19 vaccine: an interim analysis of a randomised controlled phase 3 trial in Russia. Lancet. 2021 doi: 10.1016/S0140-6736(21)00234-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Marjot, T. et al. SARS-CoV-2 vaccination in patients with liver disease: responding to the next big question. Lancet Gastroenterol. Hepatol. (2021). Summary of SARS-CoV-2 vaccination safety and efficacy data from phase III trials and discussion of key clinical and research considerations for patients with liver disease and transplantation. [DOI] [PMC free article] [PubMed]
- 141.Lebosse F, et al. CD8+ T cells from patients with cirrhosis display a phenotype that may contribute to cirrhosis-associated immune dysfunction. EBioMedicine. 2019;49:258–268. doi: 10.1016/j.ebiom.2019.10.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Liaskou E, Hirschfield GM. Cirrhosis-associated immune dysfunction: novel insights in impaired adaptive immunity. EBioMedicine. 2019;50:3–4. doi: 10.1016/j.ebiom.2019.10.056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Arvaniti V, et al. Infections in patients with cirrhosis increase mortality four-fold and should be used in determining prognosis. Gastroenterology. 2010;139:1246–1256. doi: 10.1053/j.gastro.2010.06.019. [DOI] [PubMed] [Google Scholar]
- 144.McCashland TM, Preheim LC, Gentry MJ. Pneumococcal vaccine response in cirrhosis and liver transplantation. J. Infect. Dis. 2000;181:757–760. doi: 10.1086/315245. [DOI] [PubMed] [Google Scholar]
- 145.Aggeletopoulou, I., Davoulou, P., Konstantakis, C., Thomopoulos, K. & Triantos, C. Response to hepatitis B vaccination in patients with liver cirrhosis. Rev. Med. Virol. 27, 10.1002/rmv.1942 (2017). [DOI] [PubMed]
- 146.Chong PP, Avery RK. A comprehensive review of immunization practices in solid organ transplant and hematopoietic stem cell transplant recipients. Clin. Ther. 2017;39:1581–1598. doi: 10.1016/j.clinthera.2017.07.005. [DOI] [PubMed] [Google Scholar]
- 147.Fix, O. K. AASLD expert panel consensus statement: vaccines to prevent COVID-19 infection in patients with liver disease. (AASLD, 2021). AASLD consensus statements on the approach to SARS-CoV-2 vaccination in patients with liver disease and after transplantation. [DOI] [PMC free article] [PubMed]
- 148.Tapper EB, Asrani SK. The COVID-19 pandemic will have a long-lasting impact on the quality of cirrhosis care. J. Hepatol. 2020;73:441–445. doi: 10.1016/j.jhep.2020.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Garcia-Tsao G, Abraldes JG, Berzigotti A, Bosch J. Portal hypertensive bleeding in cirrhosis: risk stratification, diagnosis, and management: 2016 practice guidance by the American Association for the study of liver diseases. Hepatology. 2017;65:310–335. doi: 10.1002/hep.28906. [DOI] [PubMed] [Google Scholar]
- 150.Erman A, Wong WWL, Feld JJ, Grootendorst P, Krahn MD. The health impact of delaying direct-acting antiviral treatment for chronic hepatitis C: a decision-analytic approach. Liver Int. 2020;40:51–59. doi: 10.1111/liv.14247. [DOI] [PubMed] [Google Scholar]
- 151.Wu CY, et al. Association between ultrasonography screening and mortality in patients with hepatocellular carcinoma: a nationwide cohort study. Gut. 2016;65:693–701. doi: 10.1136/gutjnl-2014-308786. [DOI] [PubMed] [Google Scholar]
- 152.Amaddeo G, et al. Impact of COVID-19 on the management of hepatocellular carcinoma in a high-prevalence area. JHEP Rep. 2021;3:100199. doi: 10.1016/j.jhepr.2020.100199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Di Maira T, Berenguer M. COVID-19 and liver transplantation. Nat. Rev. Gastroenterol. Hepatol. 2020;17:526–528. doi: 10.1038/s41575-020-0347-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Merola J, Schilsky ML, Mulligan DC. The impact of COVID-19 on organ donation, procurement and liver transplantation in the United States. Hepatol. Commun. 2020;5:5–11. doi: 10.1002/hep4.1620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Ritschl PV, et al. Solid organ transplantation programs facing lack of empiric evidence in the COVID-19 pandemic: a by-proxy society recommendation consensus approach. Am. J. Transplant. 2020;20:1826–1836. doi: 10.1111/ajt.15933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Thorburn D, et al. Resuming liver transplantation amid the COVID-19 pandemic. Lancet Gastroenterol. Hepatol. 2021;6:12–13. doi: 10.1016/S2468-1253(20)30360-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.World Health Organization. Global Hepatitis Report, 2017. https://www.who.int/hepatitis/publications/global-hepatitis-report2017/en/ (2017).
- 158.Blach S, et al. Impact of COVID-19 on global hepatitis C elimination efforts. J. Hepatol. 2021;74:31–36. doi: 10.1016/j.jhep.2020.07.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.The Lancet Gastroenterology Hepatology. Eliminating viral hepatitis in the COVID-19 era: weighing challenge and opportunity. Lancet Gastroenterol. Hepatol. 2020;5:789. doi: 10.1016/S2468-1253(20)30237-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Serper M, et al. A local response to COVID-19 for advanced liver disease: current model of care, challenges and opportunities. J. Hepatol. 2020;73:708–709. doi: 10.1016/j.jhep.2020.05.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Verna EC, et al. Clinical research in hepatology in the COVID-19 pandemic and post-pandemic era: challenges and the need for innovation. Hepatology. 2020;72:1819–1837. doi: 10.1002/hep.31491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Nicola M, et al. The socio-economic implications of the coronavirus pandemic (COVID-19): a review. Int. J. Surg. 2020;78:185–193. doi: 10.1016/j.ijsu.2020.04.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Nassisi M, et al. Impact of the COVID-19 lockdown on basic science research in ophthalmology: the experience of a highly specialized research facility in France. Eye. 2020;34:1187–1188. doi: 10.1038/s41433-020-0944-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.The Grocer. Coronavirus: crisis drives £160 m additional spend on supermarket booze. (The Grocer, 2020).
- 165.Institute of Alcohol Studies. Alcohol consumption during the COVID-19 lockdown in the UK ias.orghttps://www.ias.org.uk/wp-content/uploads/2020/06/sb28062020.pdf (2020).
- 166.Public Health England. Wider impacts of COVID-19 on health monitoring tool. gov.ukhttps://www.gov.uk/government/publications/wider-impacts-of-covid-19-on-health-monitoring-tool (2020).
- 167.Institute of Alcohol Studies. Alcohol consumption during the COVID-19 pandemic in the UK – Second IAS briefing ias.orghttps://www.ias.org.uk/wp-content/uploads/2020/10/sb29102020.pdf (2020).
- 168.Da BL, Im GY, Schiano TD. COVID-19 hangover: a rising tide of alcohol use disorder and alcohol-associated liver disease. Hepatology. 2020;72:1102–1108. doi: 10.1002/hep.31307. [DOI] [PubMed] [Google Scholar]
- 169.Niedzwiedz CL, et al. Mental health and health behaviours before and during the initial phase of the COVID-19 lockdown: longitudinal analyses of the UK Household Longitudinal Study. J. Epidemiol. Community Health. 2021;75:224–231. doi: 10.1136/jech-2020-215060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Winstock, A. R. et al. GDS COVID-19 Special Edition Key Findings Report https://www.globaldrugsurvey.com/gds-covid-19-special-edition-key-findings-report/ (2020).
- 171.Alcohol Toolkit Study. Alcohol Consumption in England ucl.ac.ukhttps://www.ucl.ac.uk/pals/research/clinical-educational-and-health-psychology/research-groups/health-psychology-research-45 (2020).
- 172.Kim JU, et al. Effect of COVID-19 lockdown on alcohol consumption in patients with pre-existing alcohol use disorder. Lancet Gastroenterol. Hepatol. 2020;5:886–887. doi: 10.1016/S2468-1253(20)30251-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Cargill Z, et al. Severe alcohol-related liver disease admissions post-COVID-19 lockdown: canary in the coal mine? Frontline Gastroenterol. 2020 doi: 10.1136/flgastro-2020-101693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Pellegrini M, et al. Changes in weight and nutritional habits in adults with obesity during the “Lockdown” period caused by the COVID-19 virus emergency. Nutrients. 2020;12:2016. doi: 10.3390/nu12072016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.dQ&A. Impact of COVID-19 on the diabetes community in the United States https://d-qa.com/our-work/ (2020).
- 176.Mahmud N, Hubbard RA, Kaplan DE, Serper M. Declining cirrhosis hospitalizations in the wake of the COVID-19 pandemic: a National Cohort Study. Gastroenterology. 2020;159:1134–1136.e3. doi: 10.1053/j.gastro.2020.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Bajaj JS, et al. Comparison of mortality risk in patients with cirrhosis and COVID-19 compared with patients with cirrhosis alone and COVID-19 alone: multicentre matched cohort. Gut. 2021;70:531–536. doi: 10.1136/gutjnl-2020-322118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Kim D, et al. Predictors of outcomes of COVID-19 in patients with chronic liver disease: US multi-center study. Clin. Gastroenterol. Hepatol. 2020 doi: 10.1016/j.cgh.2020.09.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Sarin SK, et al. Pre-existing liver disease is associated with poor outcome in patients with SARS CoV2 infection; the APCOLIS study (APASL COVID-19 Liver Injury Spectrum Study) Hepatol. Int. 2020;14:690–700. doi: 10.1007/s12072-020-10072-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Kates OS, et al. COVID-19 in solid organ transplant: a multi-center cohort study. Clin. Infect. Dis. 2020 doi: 10.1093/cid/ciaa1097. [DOI] [Google Scholar]
- 181.Belli LS, et al. Protective role of tacrolimus, deleterious role of age and comorbidities in liver transplant recipients with Covid-19: results from the ELITA/ELTR multi-center European study. Gastroenterology. 2020 doi: 10.1053/j.gastro.2020.11.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Butt AA, Yan P, Chotani RA, Shaikh OS. Mortality is not increased in SARS-CoV-2 infected persons with hepatitis C virus infection. Liver Int. 2021 doi: 10.1111/liv.14804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Tillett RL, et al. Genomic evidence for reinfection with SARS-CoV-2: a case study. Lancet Infect. Dis. 2021;21:52–58. doi: 10.1016/S1473-3099(20)30764-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Qin J, et al. Perioperative presentation of COVID-19 disease in a liver transplant recipient. Hepatology. 2020;72:1491–1493. doi: 10.1002/hep.31257. [DOI] [PubMed] [Google Scholar]
- 185.Long QX, et al. Clinical and immunological assessment of asymptomatic SARS-CoV-2 infections. Nat. Med. 2020;26:1200–1204. doi: 10.1038/s41591-020-0965-6. [DOI] [PubMed] [Google Scholar]
- 186.Levitsky J, et al. Risk for immune-mediated graft dysfunction in liver transplant recipients with recurrent HCV infection treated with pegylated interferon. Gastroenterology. 2012;142:1132–1139.e1. doi: 10.1053/j.gastro.2012.01.030. [DOI] [PubMed] [Google Scholar]
- 187.Friend BD, et al. Fatal orthotopic liver transplant organ rejection induced by a checkpoint inhibitor in two patients with refractory, metastatic hepatocellular carcinoma. Pediatr. Blood Cancer. 2017;64:e26682. doi: 10.1002/pbc.26682. [DOI] [PubMed] [Google Scholar]
- 188.Duchini A, Hendry RM, Nyberg LM, Viernes ME, Pockros PJ. Immune response to influenza vaccine in adult liver transplant recipients. Liver Transpl. 2001;7:311–313. doi: 10.1053/jlts.2001.23010. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The UK Liver transplant and coronavirus data that support the plots within this paper are available from the NHS Blood and Transplant Organ Donation and Transplantation Reports website at https://www.odt.nhs.uk/statistics-and-reports/ and the Coronavirus (COVID-19) in the UK website at https://coronavirus.data.gov.uk/.