Abstract
Objective
Immune checkpoint inhibitor (ICI) therapy has shown activity against melanoma brain metastases. Recently, promising results have also been reported for ICI combination therapy and ICI combined with radiotherapy. We aimed to evaluate radiologic response and adverse event rates of these therapeutic options by a systematic review and meta-analysis.
Materials and Methods
A systematic literature search of Ovid-MEDLINE and EMBASE was performed up to October 12, 2019 and included studies evaluating the intracranial objective response rates (ORRs) and/or disease control rates (DCRs) of ICI with or without radiotherapy for treating melanoma brain metastases. We also evaluated safety-associated outcomes.
Results
Eleven studies with 14 cohorts (3 with ICI combination therapy; 5 with ICI combined with radiotherapy; 6 with ICI monotherapy) were included. ICI combination therapy {pooled ORR, 53% (95% confidence interval [CI], 44–61%); DCR, 57% (95% CI, 49–66%)} and ICI combined with radiotherapy (pooled ORR, 42% [95% CI, 31–54%]; DCR, 85% [95% CI, 63–95%]) showed higher local efficacy compared to ICI monotherapy (pooled ORR, 15% [95% CI, 11–20%]; DCR, 26% [95% CI, 21–32%]). The grade 3 or 4 adverse event rate was significantly higher with ICI combination therapy (60%; 95% CI, 52–67%) compared to ICI monotherapy (11%; 95% CI, 8–17%) and ICI combined with radiotherapy (4%; 95% CI, 1–19%). Grade 3 or 4 central nervous system (CNS)-related adverse event rates were not different (9% in ICI combination therapy; 8% in ICI combined with radiotherapy; 5% in ICI monotherapy).
Conclusion
ICI combination therapy or ICI combined with radiotherapy showed better local efficacy than ICI monotherapy for treating melanoma brain metastasis. The grade 3 or 4 adverse event rate was highest with ICI combination therapy, and the CNS-related grade 3 or 4 event rate was similar. Prospective trials will be necessary to compare the efficacy of ICI combination therapy and ICI combined with radiotherapy.
Keywords: Immune checkpoint inhibitor, Immunotherapy, Radiation, Radiotherapy, Meta-analysis
INTRODUCTION
Brain metastases from melanoma are common, with the reported incidence ranging from 10% to 75% in autopsy series (1,2,3). Historically, the prognosis of melanoma with brain metastases is dismal with the median survival being only 4 months (4,5), and patients do not respond well to radiotherapy or cytotoxic chemotherapy alone (6,7).
Recently, immune checkpoint inhibitor (ICI) therapy has attracted attention as it has shown a benefit in treating a variety of soft tissue malignancies including melanoma and non-small cell lung cancer (7,8). Several clinical trials have reported ICI activity against advanced melanoma including melanoma brain metastases (9,10,11,12,13,14). ICI combination therapy (i.e., nivolumab and ipilimumab) showed promising results for treating melanoma brain metastases, with a reported intracranial objective response rate (ORR) of 46–55% (15,16,17). Additionally, stereotactic radiosurgery (SRS) is recommended as a local therapy for melanoma brain metastases in the National Comprehensive Cancer Network guidelines (9); several retrospective studies combining ICI and radiotherapy have also reported respectable efficacy (18,19,20,21).
There is a paucity of data from large-scale comparative studies to guide selection among the available therapeutic options. Rulli et al. (22) conducted a meta-analysis evaluating the efficacy of the several therapeutic options in melanoma brain metastases, but only two studies on ICI combined with radiotherapy were included. Furthermore, the safety issue was not covered. Thus, we aimed to investigate ICI monotherapy and combination therapy local efficacy and safety, with or without radiotherapy, for the treatment of melanoma brain metastases.
MATERIALS AND METHODS
This study was conducted according to the guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (23).
Search Strategy and Eligibility Criteria
A literature search of the MEDLINE/PubMed and EMBASE databases was conducted using pertinent MeSH or EMTREE terms with common keywords for relevant articles until October 12, 2019. The search terms were as follows: ((melanoma)) AND ((brain metasta*) OR (intracranial)) AND ((CTLA4) OR (CTLA-4) OR (PD1) OR (PD-1) OR (PDL1) OR (ipilimumab) OR (nivolumab) OR (pembrolizumab) OR (atezolizumab) OR (avelumab) OR (durvalumab)). The search was limited to the English language but not limited to human or animal or by publication date.
After eliminating duplicates, articles were screened based on the title and abstract. Full-text articles were then thoroughly assessed according to the following eligibility criteria: 1) population: malignant melanoma patients with brain metastasis; 2) intervention: ICI with or without radiotherapy; 3) comparator(s)/control: not applicable; 4) outcomes: intracranial objective response or disease control rate (DCR); and 5) study design: observational studies, clinical trials, and conference abstracts reporting the results of clinical trial but not published yet. We excluded studies that met any of the following criteria: 1) review; 2) case reports or case series including fewer than 10 patients; 3) conference abstracts; 4) letters, editorials, and comments; 5) animal studies; 6) studies with a partially overlapping patient cohort (for studies with an overlapping study population, the study with the largest population was selected); 7) phase I trial; and 8) studies with response assessment time not specified.
Data Extraction and Quality Assessment
A standardized extraction form was used to obtain the following information from the selected studies: 1) study characteristics: institution, study location, recruitment period, study design (retrospective vs. prospective vs. clinical trial); 2) demographic and clinical characteristics: number of treated patients/lesions, presence vs. absence of symptoms associated with melanoma brain metastasis; 3) characteristics associated with treatment: treatment arms (ICI monotherapy vs. ICI combined with radiotherapy vs. ICI combination therapy), ICI used (e.g., ipilimumab, pembrolizumab, and nivolumab), type(s) of RT if used (whole brain radiation therapy [WBRT] and/or SRS); and 4) characteristics associated with outcome: response assessment criteria, response assessment time after initiation of therapy. The quality of evidence in the included studies was evaluated using the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) system (24,25). The GRADE system rates the quality of evidence from very low to high based on study design, risk of bias, imprecision, inconsistency, indirectness, magnitude of effect, dose-response relationship, and consideration of all plausible residual confounders.
Data Synthesis and Analysis
The primary study endpoints of this meta-analysis were 1) intracranial ORR (percentage of patients with melanoma brain metastases who were confirmed as achieving complete [CR] or partial response [PR]) and 2) intracranial DCR (percentage of patients with melanoma brain metastases who were confirmed as achieving CR, PR, or stable disease [SD]) assessed using response assessment criteria in the time each study prespecified. These were also pooled separately regarding the treatment arms (ICI monotherapy vs. ICI combined with radiotherapy vs. ICI combination therapy). Of note, intracranial ORR and DCR were pooled excluding symptomatic cohorts, as symptomatic cohorts are believed to show poor response rates (17,26). In addition, intracranial CR rate was evaluated regarding the treatment arms.
We also evaluated safety-associated outcomes including treatment-related adverse events of any grade, grade 3 or 4 adverse events, central nervous system (CNS)-related events of any grade, and CNS-related grade 3 or 4 adverse events according to Common Terminology Criteria for Adverse Events v3.0 or v4.0, depending on each included study used. In addition, indirect comparisons were made between the treatment arms.
Meta-analytic pooling was based on the inverse variance method for calculating weights, and pooled estimates with their 95% confidence intervals (CI) were determined using DerSimonian–Laird random-effects modeling. Since grade 3 or 4 adverse events are rare, the pooled incidence rates of overall and CNS-related grade 3 or 4 adverse events were obtained with the binomial-normal model. In this, we calculated the pooled incidence using mixed-effects logistic regression models for dichotomous data, i.e., binomial-normal model, instead of an inverse-variance weighting model, which requires normality assumption (27,28). Heterogeneity across studies was assessed using the Q test and I2 statistic, with I2 > 50% indicating the presence of heterogeneity (29,30,31). Publication bias was evaluated using the funnel plot and Egger's test (32,33). In addition, to test if treatment arms as moderators have statistical effects in the meta-regression, we used a Wald-type chi-square test with multiplicity adjustment and the regression coefficient obtained to estimate the intervention effect and odds ratio (OR) from a reference group (34,35). In addition, we performed sensitivity analysis in the ICI monotherapy group limited to ipilimumab to test whether type of ICI would be the source of heterogeneity and in the ICI combined with radiotherapy group limited to SRS to test whether mode of radiotherapy would be the source of heterogeneity. Statistical analyses were performed using R software (version 3.1.2; R Foundation for Statistical Computing) with the “meta” and the “metafor” packages. In the meta-regression analysis, we used the Knapp and Hartung adjustment, which typically used in the mixed effects meta-regression model, to control the Type 1 error rate of 0.05 for each analysis and reported multiplicity-adjusted p values and 95% CIs.
RESULTS
Literature Search
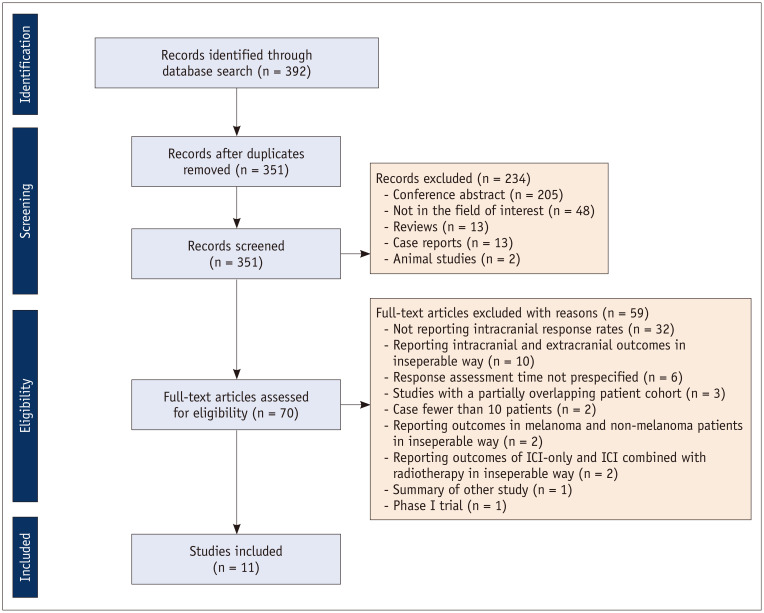
A flow chart of the publication selection process is summarized in Figure 1. Altogether, 392 non-duplicated studies were identified. Of these, 234 articles were excluded on the basis of their titles and abstract because of the following reasons: 1) conference abstract (n = 205) (except Tawbi et al. (17), reporting outcomes of nivolumab and ipilimumab combination therapy in patients with symptomatic melanoma brain metastases); 2) not in the field of interest (n = 48); 3) review (n = 13); 4) case report (n = 13); and 5) animal study (n = 2). Subsequently, 70 potentially eligible articles were assessed according to the eligibility criteria, and a further 59 studies were excluded because of the following reasons: 1) articles not reporting intracranial response rates (n = 32); 2) articles reporting intracranial and extracranial outcomes in an inseparable way (n = 10); 3) response assessment time not prespecified (n = 6); 4) studies with a partially overlapping patient cohort (n = 3); 4) articles including fewer than 10 patients (n = 2); 5) articles reporting outcomes in melanoma and non-melanoma patients in an inseparable way (n = 2); 6) articles reporting outcomes of ICI-only and ICI combined with radiotherapy in an inseparable way (n = 2); 7) summary of other study (n = 1); and 8) phase I trial (n = 1). Consequently, a total of 11 studies including 14 cohorts (divided depending on the treatment arms, what ICI used, and presence vs. absence of symptoms; 6 treated with ICI monotherapy; 5 treated with ICI combined with radiotherapy; 3 treated with ICI combination therapy) met the eligibility criteria and were included in the analysis (15,16,17,18,19,20,21,26,36,37,38).
Fig. 1. Flow chart of the study selection process.
ICI = immune checkpoint inhibitor
Characteristics of the Included Studies
The detailed study characteristics are summarized in Table 1. Five of the 11 studies were phase II clinical trials (15,16,17,26,36), and the remainder were conducted using a retrospective design (18,19,20,21,37,38). Six studies were conducted as multicenter studies (15,16,17,26,37,38). Modified versions of Response Evaluation Criteria in Solid Tumors (RECIST) v1.1, immune-related Response Criteria (irRC), Response Assessment in Neuro-Oncology brain metastases (RANO-BM), modified WHO criteria, and RECIST v1.1 were used for tumor response assessment in four (15,16,17,36), two (18,20), two (21,37), one (38), and one studies (19), respectively. Margolin et al. (26) used both modified WHO and irRC criteria, and we extracted the results based on modified WHO criteria for the meta-analytic pooling. Response assessment time after initiation of therapy varied across the studies, from 4 to 16 weeks. Three studies used nivolumab and ipilimumab combination therapy (15,16). Three studies combined SRS with ICI monotherapy (18,19,20), and one study combined SRS or WBRT with monotherapy (21). Two studies focused on symptomatic melanoma brain metastasis (17,26). Nine of the 11 studies (15,16,19,21,26,36,37,38) conducted per-patient analysis, and the remaining two studies conducted per-lesion analysis (18,20).
Table 1. Characteristics of 14 Cohorts from 11 Included Studies.
| Authors (Publication Year) | Nation | Multicenter | Study Design | Recruitment Period | Response Criteria | Response Assessment Time after Initiation of Therapy (Weeks) | ICI Used | Radiotherapy | Symptoms | Analysis | Treated No. |
|---|---|---|---|---|---|---|---|---|---|---|---|
| ICI monotherapy | |||||||||||
| Weber et al. (2011) (38) | USA | Yes | Retrospective | NR | mWHO | 12 | Ipilimumab | - | NR | Per-patient | 12 |
| Margolin et al. (2012) (26) | USA | Yes | Phase II trial | 2008.7–2009.6 | mWHO, irRC | 12 | Ipilimumab | - | Asymptomatic | Per-patient | 51 |
| Margolin et al. (2012) (26) | USA | Yes | Phase II trial | 2008.7–2009.6 | mWHO, irRC | 12 | Ipilimumab | - | Symptomatic | Per-patient | 21 |
| Queirolo et al. (2014) (37) | Italy | Yes | Retrospective | NR | irRC | 12 | Ipilimumab | - | Asymptomatic | Per-patient | 145 |
| Long et al. (2018) (15) | Australia | Yes | Phase II trial | 2014.11–2017.4 | mRECIST 1.1† | 4 | Nivolumab | - | Mixed | Per-patient | 41 |
| Kluger et al. (2019) (36) | USA | No | Phase II trial | 2014.3–2015.8 | mRECIST 1.1* | 8 | Pembrolizumab | - | Asymptomatic | ||
| ICI combined with radiotherapy | |||||||||||
| Silk et al. (2013) (21) | USA | No | Retrospective | 2005–2012 | irRC | 4–16 | Ipilimumab | WBRT/SRS | Mixed | Per-patient | 22 |
| Anderson et al. (2017) (18) | USA | No | Retrospective | 2014.1–2015.12 | RANO-BM | 6–8 | Pembrolizumab | SRS | NR | Per-lesion | 23 |
| Anderson et al. (2017) (18) | USA | No | Retrospective | 2014.1–2015.12 | RANO-BM | 6–8 | Ipilimumab | SRS | NR | Per-lesion | 31 |
| Nardin et al. (2018) (19) | France | No | Retrospective | 2012–2015 | RECIST 1.1 | 4 | Pembrolizumab | SRS | Mixed | Per-patient | 25 |
| Trommer-Nestler et al. (2018) (20) | Germany | No | Retrospective | 2011.8–2016.9 | RANO-BM | 12 | Pembrolizumab/nivolumab | SRS | NR | Per-lesion | 28 |
| ICI combination therapy | |||||||||||
| Long et al. (2018) (15) | Australia | Yes | Phase II trial | 2014.11–2017.4 | mRECIST 1.1* | 4 | Nivolumab + ipilimumab | - | Asymptomatic | Per-patient | 35 |
| Tawbi et al. (2018) (16) | USA | Yes | Phase II trial | 2015.2–2017.6 | mRECIST 1.1‡ | 4 | Nivolumab + ipilimumab | - | Asymptomatic | Per-patient | 94 |
| Tawbi et al. (2019) (17) | USA | Yes | Phase II trial | NR | mRECIST 1.1‡ | 4 | Nivolumab + ipilimumab | - | Symptomatic | Per-patient | 18 |
*Up to five target lesions of 5 mm or greater or at least twice the slice thickness if 2.5 mm or greater, †Up to five target lesions of 5–40 mm in diameter, ‡Up to five target lesions of 5–30 mm in diameter including target lesions measuring 5 to 10 mm in their longest diameter. ICI = immune checkpoint inhibitor, irRC = immune-related Response Criteria, mRECIST = modified RECIST, mWHO = modified World Health Organization, NR = not reported, RANO-BM = Response Assessment in Neuro-Oncology brain metastases, RECIST = Response Evaluation Criteria in Solid Tumors, SRS = stereotactic radiosurgery, WBRT = whole brain radiation therapy
Quality Assessment
The five included clinical trials were initially rated with high certainty rate (15,16,17,26,36), and the six retrospective studies were initially rated with low certainty rate (18,19,20,21,37,38). In the risk of bias domain, two studies were down-rated as they performed per-lesion analysis (18,20). In the imprecision domain, two studies were down-rated because of the widest 95% CI for the local efficacy among studies using the same treatment arms, derived from small sample size (21,38). In addition, the study by Tawbi et al. (17) was down-rated because of the widest 95% CI for the grade 3 or 4 adverse events among the studies using ICI combination therapy. In the inconsistency domain, one study (21) was down-rated because of a large difference of local efficacy compared to other studies using ICI combined with radiotherapy. The study by Queirolo et al. (37) was uprated due to a large effect size (comprising 145 out of 272 patients [53%] among the studies using ICI monotherapy study). Consequently, the quality of evidence was high in three (15,16,26), moderate in two (17,37), low in one (19), and very low in four studies (18,20,21,38).
Efficacy
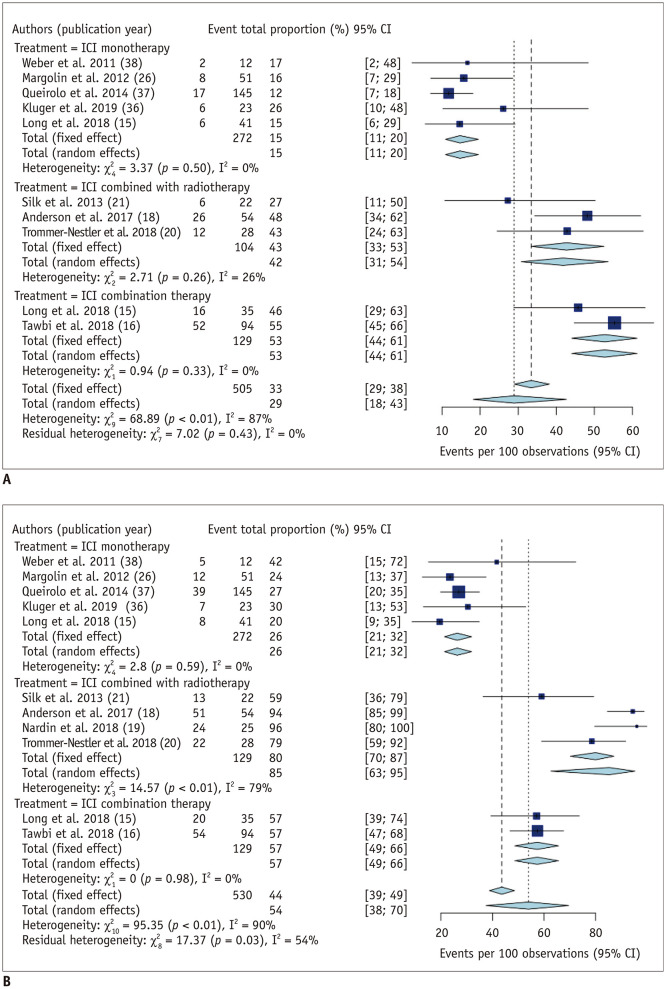
The pooled intracranial ORR and DCR when excluding symptomatic cohorts are summarized in Table 2. Five (15,26,36,37,38), three (18,20,21), and two studies (15,16) reported intracranial ORR when using ICI monotherapy, ICI combined with radiotherapy, and ICI combination therapy, respectively.
Table 2. Pooled Analysis of the Included Studies Evaluating Efficacy (Random-Effects Model).
| Treatment Arm | Intracranial ORR | Intracranial DCR | Intracranial CR | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Proportion | OR (95% CI) | P | Proportion | OR (95% CI) | P | Proportion | OR (95% CI) | P | |
| ICI monotherapy | 15 (11–20) | REF | 26 (21–32) | REF | 6 (2–14) | REF | |||
| ICI combined with radiotherapy | 42 (31–54) | 1.32 (1.17–1.49) | < 0.01 | 85 (63–95) | 1.94 (1.72–2.18)* | < 0.01 | 6 (1–37) | 1.00 (0.99–1.01) | 0.99 |
| ICI combination therapy | 53 (44–61) | 1.48 (1.32–1.65) | < 0.01 | 57 (49–66) | 1.37 (1.16–1.63)* | < 0.01 | 23 (17–32) | 1.26 (1.05–1.50) | 0.02 |
| Total | 29 (18–43) | - | - | 54 (38–70) | - | - | 10 (5–18) | - | - |
Values are expressed as proportion (95% CI). OR was calculated based on indirect comparison. *Intracranial DCR was significantly higher when using ICI combined with radiotherapy compared to ICI combination therapy (OR [95% CI], 1.41 [1.20–1.67]; p < 0.01). CI = confidence interval, CR = complete response, DCR = disease control rate (proportion of the patients who were confirmed as CR, PR, or SD), OR = odds ratio, ORR = objective response rate (proportion of the patients who were confirmed as CR or PR), PR = partial response, REF = reference category, SD = stable disease
Efficacy: ORR
Pooled intracranial ORR based on random-effects modeling was 15% (11–20%; I2 = 0%), 42% (31–54%; I2 = 26%), and 53% (95% CI, 44–61%; I2 = 0%) when using ICI monotherapy, ICI combined with radiotherapy, and ICI combination therapy, respectively. There was no substantial heterogeneity observed in all three treatment arms. The overall intracranial ORR was 29% (95% CI, 18–43%), with a substantial heterogeneity (I2 = 87%; p < 0.01) (Fig. 2). There was no significant publication bias observed in the Deeks funnel plot (p = 0.24). Compared to ICI monotherapy, intracranial ORR was significantly higher when using ICI combined with radiotherapy (OR [95% CI], 1.32 [1.17–1.49]; p < 0.01) and ICI combination therapy (OR [95% CI], 1.48 [1.32–1.65]; p < 0.01). There was no significant difference of intracranial ORR between ICI combined with radiotherapy and ICI combination therapy.
Fig. 2. Forest plot of the intracranial (A) objective response rates and (B) disease control rates excluding symptomatic cohorts.
Intracranial objective response rate was significantly higher when using ICI combined with radiotherapy (42%; 95% CI, 31–54%) and ICI combination therapy (53%; 95% CI, 44–61%) compared to ICI monotherapy (15%; 95% CI, 11–20%). Intracranial disease control rate was also significantly higher when using ICI combined with radiotherapy (85%; 95% CI, 63–95%) and ICI combination therapy (57%; 95% CI, 49–66%) compared to ICI monotherapy (26%; 95% CI, 21–32%). CI = confidence interval
Efficacy: DCR
Five (15,26,36,37,38), four (18,19,20,21), and two studies (15,16) reported intracranial DCR when using ICI monotherapy, ICI combined with radiotherapy, and ICI combination therapy, respectively. Pooled intracranial DCR based on random-effects modeling was 26% (21–32%; I2 = 0%), 85% (63–95%; I2 = 79%), and 57% (95% CI, 49–66%; I2 = 0%) when using ICI monotherapy, ICI combined with radiotherapy, and ICI combination therapy, respectively. There was a substantial heterogeneity observed in ICI combined with radiotherapy (p < 0.01). The overall intracranial DCR was 54% (95% CI, 38–70%), with a substantial heterogeneity (I2 = 90%; p < 0.01) (Fig. 2). There was no significant publication bias observed in the Deeks funnel plot (p = 0.14). Compared to ICI monotherapy, intracranial DCR was significantly higher when using ICI combined with radiotherapy (OR [95% CI], 1.94 [1.72–2.18]; p < 0.01) and ICI combination therapy (OR [95% CI], 1.37 [1.16–1.63]; p < 0.01). In addition, intracranial DCR was significantly higher when using ICI combined with radiotherapy compared to ICI combination therapy (OR [95% CI], 1.41 [1.20–1.67]; p < 0.01).
Efficacy: CR
Five (15,26,36,37,38), three (18,20,21), and two studies (15,16) reported intracranial CR rates when using ICI monotherapy, ICI combined with radiotherapy, and ICI combination therapy, respectively. Pooled intracranial CR rate based on random-effects modeling was 6% (2–14%; I2 = 57%), 6% (1–37%; I2 = 69%), and 23% (95% CI, 17–32%; I2 = 0%) when using ICI monotherapy, ICI combined with radiotherapy, and ICI combination therapy, respectively. There was a substantial heterogeneity observed in ICI monotherapy (p = 0.06) and ICI combined with radiotherapy (p = 0.04). The overall pooled intracranial CR rate was 10% (95% CI, 5–18%), with a substantial heterogeneity (I2 = 74%; p < 0.01) (Supplementary Fig. 1). There was a significant publication bias observed in the Deeks funnel plot (p < 0.01). The intracranial CR rate of ICI combination therapy was significantly higher compared to ICI monotherapy (OR [95% CI], 1.26 [1.05–1.50]; p = 0.02) but not different compared to ICI combined with radiotherapy (OR [95% CI], 1.00 [0.99–1.01]; p = 0.99).
Efficacy in Symptomatic Cohort
Margolin et al. (26) reported the efficacy of ICI monotherapy (ipilimumab) for the symptomatic cohorts, with the ORR, DCR, and CR rate of 5% (1/21), 10% (2/21), and 5% (1/21), respectively. Tawbi et al. (17) reported the efficacy of ICI combination therapy for the symptomatic cohorts, with the ORR, DCR, and CR rate of 17% (3/18), 22% (4/18), and 11% (2/18), respectively.
Safety
The pooled grade 3 or 4 adverse event rates regarding the treatment arms are summarized in Table 3, and the details of adverse events are summarized in Supplementary Table 1. In addition, information on any grade adverse event rates and CNS-related adverse event rates is described in Supplementary Materials and presented as the forest plots in Supplementary Figure 2.
Table 3. Pooled Analysis of the Included Studies Evaluating Safety (Random-Effects Model).
| Treatment Arm | Grade 3/4 AE | CNS-Related Grade 3/4 AE | ||||
|---|---|---|---|---|---|---|
| Proportion | OR (95% CI) | P | Proportion | OR (95% CI) | P | |
| ICI monotherapy | 11 (8–17) | REF | 5 (3–8) | REF | ||
| ICI combined with radiotherapy | 4 (1–19) | 0.24 (0.01–4.29)* | 0.24 | 8 (3–20) | 1.70 (0.43–6.73) | 0.40 |
| ICI combination therapy | 60 (52–67) | 11.72 (5.29–25.95)* | < 0.01 | 9 (5–15) | 2.08 (0.79–5.50) | 0.12 |
| Total | 26 (10–52) | - | - | 7 (5–9) | - | - |
Values are expressed as proportion (95% CI). OR was calculated based on indirect comparison. *Grade 3/4 AE rate was significantly higher when using ICI combination therapy compared to ICI combined with radiotherapy (OR [95% CI], 49.22 [2.83–856.62]; p = 0.02). AE = adverse event, CNS = central nervous system
Safety: Grade 3 or 4 Adverse Event
Two (15,37), two (18,20), and three studies (15,16,17) reported grade 3 or 4 adverse event rates when using ICI monotherapy, ICI combined with radiotherapy, and ICI combination therapy, respectively. The pooled grade 3 or 4 adverse event rates were 11% (8–17%; I2 = 0%), 4% (1–19%; I2 = 0%), and 60% (95% CI, 52–67%; I2 = 0%) when using ICI monotherapy, ICI combined with radiotherapy, and ICI combination therapy, respectively. The overall grade 3 or 4 adverse event rate was 26% (95% CI, 10–52%) with a substantial heterogeneity (I2 = 93%; p < 0.01) (Supplementary Fig. 3). There was no significant publication bias observed in the Deeks funnel plot (p = 0.40). The grade 3 or 4 adverse event rate was significantly higher when using ICI combination therapy compared to ICI monotherapy (OR [95% CI], 11.72 [5.29–25.95]; p < 0.01) and ICI combined with radiotherapy (OR [95% CI], 49.22 [2.83–856.62]; p = 0.02).
Safety: Grade 3 or 4 CNS-Related Adverse Event
Five (15,26,36,37,38), three (18,19,20), and three studies (15,16,17) reported grade 3 or 4 CNS-related adverse event rates when using ICI monotherapy, ICI combined with radiotherapy, and ICI combination therapy, respectively. The pooled grade 3 or 4 CNS-related adverse event rates were 5% (3–8%; I2 = 8%), 8% (3–20%; I2 = 0%), and 9% (95% CI, 5–15%; I2 = 0%) when using ICI monotherapy, ICI combined with radiotherapy, and ICI combination therapy, respectively. The overall grade 3 or 4 CNS-related adverse event rate was 7% (95% CI, 5–9%), without heterogeneity (I2 = 3%; p = 0.41) (Supplementary Fig. 3). There was no significant publication bias observed in the Deeks funnel plot (p = 0.88). Grade 3 or 4 CNS-related adverse event rates were not significantly different between the three arms.
Sensitivity Analysis
To test whether types of ICI (anti-cytotoxic T-lymphocyte antigen 4 [anti-CTLA-4; ipilimumab] vs. anti-programmed death 1 [anti-PD 1; nivolumab and pembrolizumab]) were the source of heterogeneity, we performed sensitivity analysis in the ICI monotherapy group. Three cohorts used anti-CTLA-4 drugs (26,37,38), and the other two cohorts used anti-PD-1 drugs (15,36). The pooled estimates were robust against types of ICI except intracranial CR; compared to anti-CTLA-4, anti-PD-1 drugs showed higher intracranial CR (12% [95% CI,5–26%; I2 = 0%] vs. 3% [95% CI, 1–6%; I2 = 31%]; p < 0.01) (Supplementary Table 2).
To test whether mode of radiotherapy was the source of heterogeneity, we performed sensitivity analysis in the ICI combined with radiotherapy group. In a study by Silk et al. (21), WBRT and SRS was used in 48.5% and 51.5% of the patients, respectively. The other three cohorts only used SRS (18,19,20). Since Silk et al. (21) did not report the data regarding adverse events, sensitivity analysis was possible only in response rates. Intracranial ORR (42% [95% CI, 31–54%] to 46% [95% CI, 36–57%]; p = 0.11), DCR (85% [95% CI, 63–95%] to 91% [95% CI, 74–97%]; p = 0.10), and CR (6% [95% CI, 1–37%] to 9% [95% CI, 1–59%]; p = 0.56) slightly increased when excluding the Silk et al. (21) study but did not show statistically significant difference (Supplementary Table 3).
DISCUSSION
This meta-analysis showed that intracranial ORR and DCR were significantly higher when using ICI combined with radiotherapy (pooled ORR, 42%; DCR, 85%) or ICI combination therapy (pooled ORR, 53%; DCR, 57%) compared to ICI monotherapy (pooled ORR, 15%; DCR, 26%). Intracranial DCR was highest when using ICI combined radiotherapy, and intracranial CR rate was highest when using ICI combination therapy (23%). In terms of safety, the grade 3 or 4 adverse event rate was significantly higher with ICI combination therapy (60%) compared to ICI monotherapy (11%) and ICI combined with radiotherapy (4%), but grade 3 or 4 CNS-related adverse event rates were not significantly different across the treatment arms (5% in ICI monotherapy, 8% in ICI combined with radiotherapy, 9% in ICI combination therapy). Except for differences in intracranial CR depending on types of ICI used, response rates were not statistically different depending on types of ICI or mode of radiotherapy.
In several studies, ipilimumab (anti-CTLA 4) combined with nivolumab (anti-PD 1) has shown superior efficacy compared to ipilimumab alone for metastatic melanoma (39,40). Similarly, for melanoma brain metastases, a study by Long et al. (15) showed better intracranial response when using nivolumab combined with ipilimumab compared to nivolumab alone. The largest single-arm trial, conducted by Tawbi et al. (16), showed intracranial ORR and DCR of nivolumab and ipilimumab combination therapy to be 55% and 57%, respectively. Our study reaffirmed the superior local efficacy of ICI combination therapy compared to ICI monotherapy. Meanwhile, the pooled grade 3 or 4 adverse event was also significantly higher with ICI combination therapy. Most but not all reported grade 3 or 4 adverse events resolved after appropriate management following safety guideline. The pooled CNS-related grade 3 or 4 events with ICI combination therapy were similar to other treatment arms.
Radiotherapy has been used to potentially enhance efficacy of ICI monotherapy for melanoma brain metastases, and our study demonstrated better local efficacy of ICI combined with radiotherapy. In addition, ICI combined with radiotherapy showed better intracranial DCR compared to ICI combination therapy, which indicates a higher proportion of tumors maintain a stable state when using ICI combined with radiotherapy. The synergistic effect of combining radiotherapy may be explained by the fact that radiation increases permeability of the blood-brain barrier (41), induces mitotic cell death, and releases tumor cell antigens, which can stimulate a cytotoxic immune response (42), and activates immune cells to attack tumor cells outside of irradiated zone, i.e., the abscopal effect (43). Furthermore, our study showed that the pooled grade 3 or 4 adverse events when using ICI combined with radiotherapy were not significantly different compared with ICI monotherapy. One of the specific concerns of using of radiotherapy is radionecrosis. Although still controversial, increased incidence of radionecrosis when using SRS combined with ICI compared to SRS alone cannot be excluded, considering previous results (44,45,46,47). The reported incidence of radionecrosis in treating melanoma brain metastases has been 0–38% (18,19,44,45,46,47,48) with the meta-analytic pooled incidence of 5.3% (49). Further studies are required to clarify this issue.
The included studies used various response assessment criteria. In 2015, the RANO working group announced RANO-BM criteria, which has recently gained wide acceptance. In contrast to WHO or RECIST criteria, mainly used for solid tumors of the body, RANO-BM was developed solely for evaluating treatment response of brain metastasis. Of note, only RANO-BM criteria consider pseudoprogression when evaluating the therapeutic response of ICI and SRS. Pseudoprogression can be considered when an image mimics local progression after initiation of the treatment but decreases rapidly on subsequent imaging. Regarding extracranial melanoma, the reported incidence of pseudoprogression ranged from 5% to 10% (50,51,52). In our included studies, incidence of pseudoprogression was 4% after pembrolizumab monotherapy (36) and 8–14% after ICI combined radiotherapy (19,20).
There are several limitations of note. First, this meta-analysis was conducted using study-level data without detailed patient-level data. Second, all included studies on ICI combined with radiotherapy were conducted using a retrospective design, decreasing the comparability with the other treatment arms. Furthermore, because not all studies on ICI combined with radiotherapy reported both CNS-related grade 3 or 4 and grade 3 or 4 adverse event rates, comparison of these two pooled adverse event rates was limited. For example, only the grade 3 or 4 adverse event rate (not CNS-related grade 3 or 4 adverse event rate) was available in the study by Nardin et al. (19), which created a discrepancy between two pooled rates. Therefore, large prospective trials investigating the outcomes of ICI combined with radiotherapy, especially focusing on the comparison with ICI combination therapy, seem to be necessary. Third, thorough meta-regression analysis considering clinically important factors, i.e., metastatic burden, associated symptoms, and response criteria, to adjust response criteria effect was not feasible due to insufficient data. Regardless, this study-level meta-analysis provides important information for future practice and research.
In conclusion, ICI combination therapy and ICI combined with radiotherapy showed better local efficacy than ICI monotherapy for treating melanoma brain metastases. Overall, grade 3 or 4 adverse events were more frequent when using ICI combination therapy while CNS-related grade 3 or 4 adverse events were not statistically different across the three arms. However, since our analyses were based on indirect comparison and thorough meta-regression analysis was not available, prospective trials will be necessary to compare the efficacy of ICI combination therapy and ICI combined with radiotherapy.
Footnotes
Conflicts of Interest: The authors have no potential conflicts of interest to disclose.
Supplementary Materials
The Data Supplement is available with this article at https://doi.org/10.3348/kjr.2020.0728.
Forest plot of the intracranial complete response rates excluding symptomatic cohorts.
Summary of Grade 3 or 4 AE
Forest plot of the (A) any grade adverse event rates and (B) any grade CNS-related adverse event rates.
Forest plot of the (A) grade 3 or 4 adverse event rates and (B) grade 3 or 4 CNS-related adverse event rates.
Summary of Sensitivity Analysis Regarding Types of ICI
Summary of Sensitivity Analysis Regarding Mode of Radiotherapy
References
- 1.Sampson JH, Carter JH, Jr, Friedman AH, Seigler HF. Demographics, prognosis, and therapy in 702 patients with brain metastases from malignant melanoma. J Neurosurg. 1998;88:11–20. doi: 10.3171/jns.1998.88.1.0011. [DOI] [PubMed] [Google Scholar]
- 2.Amer MH, Al-Sarraf M, Vaitkevicius VK. Clinical presentation, natural history and prognostic factors in advanced malignant melanoma. Surg Gynecol Obstet. 1979;149:687–692. [PubMed] [Google Scholar]
- 3.Budman DR, Camacho E, Wittes RE. The current causes of death in patients with malignant melanoma. Eur J Cancer. 1978;14:327–330. doi: 10.1016/0014-2964(78)90201-3. [DOI] [PubMed] [Google Scholar]
- 4.Davies MA, Liu P, McIntyre S, Kim KB, Papadopoulos N, Hwu WJ, et al. Prognostic factors for survival in melanoma patients with brain metastases. Cancer. 2011;117:1687–1696. doi: 10.1002/cncr.25634. [DOI] [PubMed] [Google Scholar]
- 5.Fife KM, Colman MH, Stevens GN, Firth IC, Moon D, Shannon KF, et al. Determinants of outcome in melanoma patients with cerebral metastases. J Clin Oncol. 2004;22:1293–1300. doi: 10.1200/JCO.2004.08.140. [DOI] [PubMed] [Google Scholar]
- 6.Goyal S, Silk AW, Tian S, Mehnert J, Danish S, Ranjan S, et al. Clinical management of multiple melanoma brain metastases: a systematic review. JAMA Oncol. 2015;1:668–676. doi: 10.1001/jamaoncol.2015.1206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Braschi-Amirfarzan M, Tirumani SH, Hodi FS, Jr, Nishino M. Immune-checkpoint inhibitors in the era of precision medicine: what radiologists should know. Korean J Radiol. 2017;18:42–53. doi: 10.3348/kjr.2017.18.1.42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Darvin P, Toor SM, Sasidharan Nair V, Elkord E. Immune checkpoint inhibitors: recent progress and potential biomarkers. Exp Mol Med. 2018;50:1–11. doi: 10.1038/s12276-018-0191-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Nabors LB, Portnow J, Ammirati M, Baehring J, Brem H, Butowski N, et al. NCCN guidelines insights: central nervous system cancers, version 1.2017. J Natl Compr Canc Netw. 2017;15:1331–1345. doi: 10.6004/jnccn.2017.0166. [DOI] [PubMed] [Google Scholar]
- 10.Robert C, Ribas A, Schachter J, Arance A, Grob JJ, Mortier L, et al. Pembrolizumab versus ipilimumab in advanced melanoma (KEYNOTE-006): post-hoc 5-year results from an open-label, multicentre, randomised, controlled, phase 3 study. Lancet Oncol. 2019;20:1239–1251. doi: 10.1016/S1470-2045(19)30388-2. [DOI] [PubMed] [Google Scholar]
- 11.Robert C, Schachter J, Long GV, Arance A, Grob JJ, Mortier L, et al. Pembrolizumab versus ipilimumab in advanced melanoma. N Engl J Med. 2015;372:2521–2532. doi: 10.1056/NEJMoa1503093. [DOI] [PubMed] [Google Scholar]
- 12.Weber JS, D'Angelo SP, Minor D, Hodi FS, Gutzmer R, Neyns B, et al. Nivolumab versus chemotherapy in patients with advanced melanoma who progressed after anti-CTLA-4 treatment (CheckMate 037): a randomised, controlled, open-label, phase 3 trial. Lancet Oncol. 2015;16:375–384. doi: 10.1016/S1470-2045(15)70076-8. [DOI] [PubMed] [Google Scholar]
- 13.Hodi FS, O'Day SJ, McDermott DF, Weber RW, Sosman JA, Haanen JB, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363:711–723. doi: 10.1056/NEJMoa1003466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ribas A, Kefford R, Marshall MA, Punt CJ, Haanen JB, Marmol M, et al. Phase III randomized clinical trial comparing tremelimumab with standard-of-care chemotherapy in patients with advanced melanoma. J Clin Oncol. 2013;31:616–622. doi: 10.1200/JCO.2012.44.6112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Long GV, Atkinson V, Lo S, Sandhu S, Guminski AD, Brown MP, et al. Combination nivolumab and ipilimumab or nivolumab alone in melanoma brain metastases: a multicentre randomised phase 2 study. Lancet Oncol. 2018;19:672–681. doi: 10.1016/S1470-2045(18)30139-6. [DOI] [PubMed] [Google Scholar]
- 16.Tawbi HA, Forsyth PA, Algazi A, Hamid O, Hodi FS, Moschos SJ, et al. Combined nivolumab and ipilimumab in melanoma metastatic to the brain. N Engl J Med. 2018;379:722–730. doi: 10.1056/NEJMoa1805453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Tawbi HAH, Forsyth PA, Hodi FS, Lao CD, Moschos SJ, Hamid O, et al. Efficacy and safety of the combination of nivolumab (NIVO) plus ipilimumab (IPI) in patients with symptomatic melanoma brain metastases (CheckMate 204) J Clin Oncol. 2019;37:9501 [Google Scholar]
- 18.Anderson ES, Postow MA, Wolchok JD, Young RJ, Ballangrud Å, Chan TA, et al. Melanoma brain metastases treated with stereotactic radiosurgery and concurrent pembrolizumab display marked regression; efficacy and safety of combined treatment. J Immunother Cancer. 2017;5:76. doi: 10.1186/s40425-017-0282-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Nardin C, Mateus C, Texier M, Lanoy E, Hibat-Allah S, Ammari S, et al. Tolerance and outcomes of stereotactic radiosurgery combined with anti-programmed cell death-1 (pembrolizumab) for melanoma brain metastases. Melanoma Res. 2018;28:111–119. doi: 10.1097/CMR.0000000000000413. [DOI] [PubMed] [Google Scholar]
- 20.Trommer-Nestler M, Marnitz S, Kocher M, Rueß D, Schlaak M, Theurich S, et al. Robotic stereotactic radiosurgery in melanoma patients with brain metastases under simultaneous anti-PD-1 treatment. Int J Mol Sci. 2018;19:2653. doi: 10.3390/ijms19092653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Silk AW, Bassetti MF, West BT, Tsien CI, Lao CD. Ipilimumab and radiation therapy for melanoma brain metastases. Cancer Med. 2013;2:899–906. doi: 10.1002/cam4.140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Rulli E, Legramandi L, Salvati L, Mandala M. The impact of targeted therapies and immunotherapy in melanoma brain metastases: a systematic review and meta-analysis. Cancer. 2019;125:3776–3789. doi: 10.1002/cncr.32375. [DOI] [PubMed] [Google Scholar]
- 23.Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gøtzsche PC, Ioannidis JP, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. Ann Intern Med. 2009;151:W65–W94. doi: 10.7326/0003-4819-151-4-200908180-00136. [DOI] [PubMed] [Google Scholar]
- 24.Atkins D, Eccles M, Flottorp S, Guyatt GH, Henry D, Hill S, et al. Systems for grading the quality of evidence and the strength of recommendations I: critical appraisal of existing approaches The GRADE Working Group. BMC Health Serv Res. 2004;4:38. doi: 10.1186/1472-6963-4-38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Guyatt G, Oxman AD, Akl EA, Kunz R, Vist G, Brozek J, et al. GRADE guidelines: 1. introduction-GRADE evidence profiles and summary of findings tables. J Clin Epidemiol. 2011;64:383–394. doi: 10.1016/j.jclinepi.2010.04.026. [DOI] [PubMed] [Google Scholar]
- 26.Margolin K, Ernstoff MS, Hamid O, Lawrence D, McDermott D, Puzanov I, et al. Ipilimumab in patients with melanoma and brain metastases: an open-label, phase 2 trial. Lancet Oncol. 2012;13:459–465. doi: 10.1016/S1470-2045(12)70090-6. [DOI] [PubMed] [Google Scholar]
- 27.Hamza TH, van Houwelingen HC, Stijnen T. The binomial distribution of meta-analysis was preferred to model within-study variability. J Clin Epidemiol. 2008;61:41–51. doi: 10.1016/j.jclinepi.2007.03.016. [DOI] [PubMed] [Google Scholar]
- 28.Stijnen T, Hamza TH, Ozdemir P. Random effects meta-analysis of event outcome in the framework of the generalized linear mixed model with applications in sparse data. Stat Med. 2010;29:3046–3067. doi: 10.1002/sim.4040. [DOI] [PubMed] [Google Scholar]
- 29.Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327:557–560. doi: 10.1136/bmj.327.7414.557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kim KW, Lee J, Choi SH, Huh J, Park SH. Systematic review and meta-analysis of studies evaluating diagnostic test accuracy: a practical review for clinical researchers-part I. general guidance and tips. Korean J Radiol. 2015;16:1175–1187. doi: 10.3348/kjr.2015.16.6.1175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Lee J, Kim KW, Choi SH, Huh J, Park SH. Systematic review and meta-analysis of studies evaluating diagnostic test accuracy: a practical review for clinical researchers-part II. Statistical methods of meta-analysis. Korean J Radiol. 2015;16:1188–1196. doi: 10.3348/kjr.2015.16.6.1188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315:629–634. doi: 10.1136/bmj.315.7109.629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Sterne JA, Egger M, Smith GD. Systematic reviews in health care: investigating and dealing with publication and other biases in meta-analysis. BMJ. 2001;323:101–105. doi: 10.1136/bmj.323.7304.101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Higgins JP, Thompson SG. Controlling the risk of spurious findings from meta-regression. Stat Med. 2004;23:1663–1682. doi: 10.1002/sim.1752. [DOI] [PubMed] [Google Scholar]
- 35.Knapp G, Hartung J. Improved tests for a random effects meta-regression with a single covariate. Stat Med. 2003;22:2693–2710. doi: 10.1002/sim.1482. [DOI] [PubMed] [Google Scholar]
- 36.Kluger HM, Chiang V, Mahajan A, Zito CR, Sznol M, Tran T, et al. Long-term survival of patients with melanoma with active brain metastases treated with pembrolizumab on a phase II trial. J Clin Oncol. 2019;37:52–60. doi: 10.1200/JCO.18.00204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Queirolo P, Spagnolo F, Ascierto PA, Simeone E, Marchetti P, Scoppola A, et al. Efficacy and safety of ipilimumab in patients with advanced melanoma and brain metastases. J Neurooncol. 2014;118:109–116. doi: 10.1007/s11060-014-1400-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Weber JS, Amin A, Minor D, Siegel J, Berman D, O'Day SJ. Safety and clinical activity of ipilimumab in melanoma patients with brain metastases: retrospective analysis of data from a phase 2 trial. Melanoma Res. 2011;21:530–534. doi: 10.1097/CMR.0b013e32834d3d88. [DOI] [PubMed] [Google Scholar]
- 39.Larkin J, Chiarion-Sileni V, Gonzalez R, Grob JJ, Cowey CL, Lao CD, et al. Combined nivolumab and ipilimumab or monotherapy in untreated melanoma. N Engl J Med. 2015;373:23–34. doi: 10.1056/NEJMoa1504030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Postow MA, Chesney J, Pavlick AC, Robert C, Grossmann K, McDermott D, et al. Nivolumab and ipilimumab versus ipilimumab in untreated melanoma. N Engl J Med. 2015;372:2006–2017. doi: 10.1056/NEJMoa1414428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Cao Y, Tsien CI, Shen Z, Tatro DS, Ten Haken R, Kessler ML, et al. Use of magnetic resonance imaging to assess blood-brain/blood-glioma barrier opening during conformal radiotherapy. J Clin Oncol. 2005;23:4127–4136. doi: 10.1200/JCO.2005.07.144. [DOI] [PubMed] [Google Scholar]
- 42.Quail DF, Joyce JA. The microenvironmental landscape of brain tumors. Cancer Cell. 2017;31:326–341. doi: 10.1016/j.ccell.2017.02.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Walle T, Martinez Monge R, Cerwenka A, Ajona D, Melero I, Lecanda F. Radiation effects on antitumor immune responses: current perspectives and challenges. Ther Adv Med Oncol. 2018 Jan; doi: 10.1177/1758834017742575. [Epub] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Colaco RJ, Martin P, Kluger HM, Yu JB, Chiang VL. Does immunotherapy increase the rate of radiation necrosis after radiosurgical treatment of brain metastases? J Neurosurg. 2016;125:17–23. doi: 10.3171/2015.6.JNS142763. [DOI] [PubMed] [Google Scholar]
- 45.Patel KR, Shoukat S, Oliver DE, Chowdhary M, Rizzo M, Lawson DH, et al. Ipilimumab and stereotactic radiosurgery versus stereotactic radiosurgery alone for newly diagnosed melanoma brain metastases. Am J Clin Oncol. 2017;40:444–450. doi: 10.1097/COC.0000000000000199. [DOI] [PubMed] [Google Scholar]
- 46.Kaidar-Person O, Zagar TM, Deal A, Moschos SJ, Ewend MG, Sasaki-Adams D, et al. The incidence of radiation necrosis following stereotactic radiotherapy for melanoma brain metastases: the potential impact of immunotherapy. Anticancer Drugs. 2017;28:669–675. doi: 10.1097/CAD.0000000000000497. [DOI] [PubMed] [Google Scholar]
- 47.Diao K, Bian SX, Routman DM, Yu C, Ye JC, Wagle NA, et al. Stereotactic radiosurgery and ipilimumab for patients with melanoma brain metastases: clinical outcomes and toxicity. J Neurooncol. 2018;139:421–429. doi: 10.1007/s11060-018-2880-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Skrepnik T, Sundararajan S, Cui H, Stea B. Improved time to disease progression in the brain in patients with melanoma brain metastases treated with concurrent delivery of radiosurgery and ipilimumab. Oncoimmunology. 2017;6:e1283461. doi: 10.1080/2162402X.2017.1283461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Lehrer EJ, Peterson J, Brown PD, Sheehan JP, Quiñones-Hinojosa A, Zaorsky NG, et al. Treatment of brain metastases with stereotactic radiosurgery and immune checkpoint inhibitors: an international meta-analysis of individual patient data. Radiother Oncol. 2019;130:104–112. doi: 10.1016/j.radonc.2018.08.025. [DOI] [PubMed] [Google Scholar]
- 50.Wolchok JD, Hoos A, O'Day S, Weber JS, Hamid O, Lebbé C, et al. Guidelines for the evaluation of immune therapy activity in solid tumors: immune-related response criteria. Clin Cancer Res. 2009;15:7412–7420. doi: 10.1158/1078-0432.CCR-09-1624. [DOI] [PubMed] [Google Scholar]
- 51.Nishino M, Giobbie-Hurder A, Manos MP, Bailey N, Buchbinder EI, Ott PA, et al. Immune-related tumor response dynamics in melanoma patients treated with pembrolizumab: identifying markers for clinical outcome and treatment decisions. Clin Cancer Res. 2017;23:4671–4679. doi: 10.1158/1078-0432.CCR-17-0114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Hodi FS, Hwu WJ, Kefford R, Weber JS, Daud A, Hamid O, et al. Evaluation of immune-related response criteria and RECIST v1.1 in patients with advanced melanoma treated with pembrolizumab. J Clin Oncol. 2016;34:1510–1517. doi: 10.1200/JCO.2015.64.0391. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Forest plot of the intracranial complete response rates excluding symptomatic cohorts.
Summary of Grade 3 or 4 AE
Forest plot of the (A) any grade adverse event rates and (B) any grade CNS-related adverse event rates.
Forest plot of the (A) grade 3 or 4 adverse event rates and (B) grade 3 or 4 CNS-related adverse event rates.
Summary of Sensitivity Analysis Regarding Types of ICI
Summary of Sensitivity Analysis Regarding Mode of Radiotherapy