Abstract
Background:
Cardiovascular disease (CVD) remains the leading cause of mortality in type 2 diabetes (T2D). Better interventions are needed to mitigate the high lifetime risk for CVD in youth T2D.
Objective:
To compare 30-year risk for cardiovascular disease (CVD) events in 2 cohorts of adolescents with type 2 diabetes (T2D) and severe obesity undergoing medical or surgical treatment of T2D.
Setting:
Longitudinal multicenter studies at University hospitals.
Methods:
A secondary analysis of data collected from the participants with T2D enrolled in the Teen-Longitudinal Assessment of Bariatric Surgery (Teen-LABS, n = 30) and participants of similar age and racial distribution from the Treatment Options of Type 2 Diabetes in Adolescents and Youth (TODAY n = 63) studies was performed. Teen-LABS participants underwent metabolic bariatric surgery (MBS). TODAY participants were randomized to metformin alone or in combination with rosiglitazone or intensive lifestyle intervention, with insulin therapy given for glycemic progression. A 30-year CVD event score developed by the Framingham Heart Study was the primary outcome, assessed at baseline (preoperatively for Teen-LABS), 1 year, and 5 years of follow-up.
Results:
Participants with T2D from Teen-LABS (n = 30; mean ± SD age = 16.9 ± 1.3 yr; 70% female; 60% white; body mass index (BMI) = 54.4 ± 9.5 kg/m2) and TODAY (n = 63; 15.3 ± 1.3 yr; 56% female; 71% white; BMI 40.5 ± 4.9 kg/m2) were compared. The likelihood of CVD events was higher in Teen-LABS versus TODAY at baseline (17.66 [1.59] versus 12.11 [.79]%, adjusted P = .002). One year after MBS, event risk was significantly lower in Teen-LABS versus TODAY (6.79 [1.33] versus 13.64 [0.96]%, adjusted P < .0001), and sustained at 5 years follow-up (adjusted P < .0001).
Conclusion:
Despite higher pretreatment risk for CVD events, treatment with MBS resulted in a reduction in estimated CVD event risks, whereas medical therapy associated with an increase in risk among adolescents with T2D and severe obesity.
Keywords: Type 2 diabetes, Cardiovascular disease, Prediction, Severe obesity, Metabolic bariatric surgery, Medical therapy
Cardiovascular disease (CVD) is the leading cause of mortality in type 2 diabetes (T2D) and is characterized by a long clinically silent period before onset of events including myocardial infarction, congestive heart failure, stroke and coronary death [1-3]. The long latency between risk factor development and disease manifestation makes it difficult to capture events in youth-onset T2D cohorts. Accordingly, surrogate outcomes and risk scores are helpful to predict likelihood of development CVD events and the effect of treatment on risk of events. Compared with adult-onset T2D, youth with T2D have a greater degree of insulin resistance, more rapid β-cell failure, and higher prevalence of diabetic kidney disease (DKD), all causally related to CVD risk [4-8].
The Teen–Longitudinal Assessment of Bariatric Surgery (Teen-LABS) is a multicenter, prospective cohort study evaluating outcomes of metabolic bariatric surgery (MBS) among adolescents with severe obesity. The Treatment Options of Type 2 Diabetes in Adolescents and Youth (TODAY) was a multicenter randomized, controlled trial designed to investigate strategies to achieve durable glycemic control in youth-onset T2D [9]. These unique cohorts provide a rare opportunity to examine differences between MBS and standard medical therapy in adolescents with severe obesity and T2D. In recent analyses by our group, Teen-LABS participants with T2D had a significantly greater baseline cardiovascular risk factor burden than TODAY participants with T2D, with higher systolic and diastolic blood pressure and low-density lipoprotein cholesterol and triglyceride concentrations at baseline. During 2 years of follow-up, mean glycated hemoglobin (HbA1C), body mass index (BMI), fasting glucose, total cholesterol and triglycerides, and systolic and diastolic blood pressure decreased in Teen-LABS but increased in TODAY [10]. Similarly, participants with T2D in TODAY demonstrated increasing rates of DKD over 5 years of follow-up, whereas those undergoing MBS in Teen-LABS experienced regression of DKD over the same time [11]. Further studies are now needed to understand differences in the effect of MBS and medical therapy on other T2D-related co-morbidities including longer-term CVD risk. Long-term prospect of CVD events can be calculated by risk scores. For example, the Framingham Heart Study developed a 30-year CVD event risk score (validated in people between 20–59 years) [12] which was previously applied to youth with severe obesity without T2D in Teen-LABS and demonstrated CVD event risk reduction in response to MBS [13]. However, the effects of MBS versus medical intervention on risk for CVD events has yet to be explored in youth-onset T2D.
Accordingly, the objective of this study was to estimate 30-year risk of CVD events using 5 years of follow-up data from these 2 cohorts (Teen-LABS and TODAY) of adolescents with severe obesity and T2D undergoing medical versus MBS interventions. We hypothesized that youth with T2D undergoing MBS would experience a decrease in likelihood of CVD events, whereas youth with T2D treated medically would experience an increase in likelihood of CVD events over 5 years.
Methods
Study design and participants
Teen-LABS enrolled 242 adolescents (≤19 yr) from March 1, 2007 through December 31, 2011. TODAY enrollment started on May 1, 2004 and ended on December 31, 2009, with a total of 699 randomized participants (10–17 yr). Postintervention follow-up, where participants were provided with standard medical therapy, began immediately after the TODAY clinical trial was completed. Teen-LABS and TODAY have been described in detail elsewhere [9,14,15]. The TODAY and Teen-LABS protocols were approved by the institutional review boards of each participating institution. Participants provided written informed parental consent and child assent. The participants provided consent for identifiers to be maintained at the data coordinating centers for each study. Deidentified data were used for the purposes of this current analysis.
We have reported comparisons of Teen-LABS and TODAY in prior publications [10]. Pertinent to this analysis, there were 30 Teen-LABS participants with T2D at the time of MBS. Of these, 24 underwent Roux-en-Y gastric bypass and 6 underwent vertical sleeve gastrectomy procedures. These were pooled in our analyses due to the limited number of participants undergoing vertical sleeve gastrectomy. Participants with T2D in TODAY (irrespective of treatment group assignment) were frequency matched to the 30 Teen-LABS participants with T2D using the following matching criteria: baseline age (13–18 yr), race/ethnicity, sex, and baseline BMI (>35 kg/m2). Through this process, a total of 63 TODAY participants were identified. This secondary analysis included data collected from the 30 MBStreated and 63 medically treated individuals with T2D at the baseline, 1-year, 2-year, 3-year, 4-year, and 5-year study visits.
T2D definition
Standard conventions were followed for the assessment and prevalence of conditions over time. In brief, presence of T2D in Teen-LABS participants was defined as use of medications for diabetes, baseline HbA1C concentration of >6.5%, fasting glucose concentration of >126 mg/dL, or 2-hour glucose value >200 mg/dL during an oral glucose tolerance test in the 6 months before enrollment. T2D in TODAY was defined by standard American Diabetes Association (ADA) glucose and HbA1C criteria [16] except that asymptomatic patients with a normal fasting glucose concentration but elevated 2-hour glucose concentration during an oral glucose tolerance test were also required to have an HbA1C concentration of >6% to limit enrollment of patients with prediabetes [17].
In Teen-LABS, remission of T2D was defined as no use of medication for diabetes, and HbA1C <6.5% (if HbA1C was not available, remission also required fasting glucose concentration <126 mg/dL). In instances where specified laboratory and/or medication use data were unavailable, patient-reported declarations of presence of or absence of diabetes were used, as previously described [18]. Diabetes remission was not collected for the TODAY cohort.
Laboratory assessments
All laboratory assays for the Teen-LABS and TODAY cohorts were performed by the Northwest Lipid Metabolism and Diabetes Research Laboratories, in Seattle, Washington, as previously described [10]. HbA1C (high-performance liquid chromatography), insulin (double-antibody radioimmunoassay) and lipid panel assays were performed as previously described [9,14,15]. Insulin sensitivity was calculated annually as 1/fasting insulin (1/IF [mL/μU]), which correlates strongly with hyperinsulinemic-euglycemic clamp–derived in vivo insulin sensitivity in obese youth with or without T2D [19]. Concentrations of creatinine in serum and urine were determined annually using the Creatinine Plus enzymatic Roche reagent on a Modular P analyzer (Roche Diagnostics, Inc., Indianapolis, IN). The results of this procedure are traceable to the IDMS reference method and allowed for accurate estimated glomerular filtration (eGFR). The reportable range of creatinine in serum/plasma samples is: .03–60.0 mg/dL, and .03–1200.0 mg/dL in urine samples. Concentration of cystatin C in serum was determined at baseline and annually immunochemically by using Siemens reagents (Siemens Healthcare Diagnostics Inc., Newark, DE) on a Siemens nephelometer autoanalyzer (BNII). This method is standardized against the IFCC/ERM DA-471 Reference Material (RT Corp., Laramie, WY).
Elevated UAE and hypertension definition
Due to the expected normal to elevated glomerular filtration rates for age, we calculated eGFR by the Full Age Spectrum (FAS) combined serum creatinine and cystatin C equation, which has been validated in both pediatrics and adults and lends itself well to studies examining the transition from pediatric to early adulthood: [20]
The FAS equation is based on normalized serum creatinine (SCr/Qcrea), where Qcrea is the median serum creatinine (SCr) from healthy populations to account for age and sex, and QcysC is defined as .82 mg/L for <70 years. The coefficient ‘α’ in the denominator is a weighting factor for the normalized renal biomarkers. We used α = .5, which means the denominator is equal to the average of both normalized biomarkers. Urine albumin-to-creatinine ratio (UACR) was measured at baseline and annually thereafter unless a result was abnormal. Spot urine samples were obtained after a 10–14 hr overnight fast. Elevated urine albumin excretion (UAE) (previously known as microalbuminuria) was defined as an UACR of ≥30 μg/mg [14]. Participants who developed elevated UAE in TODAY were promptly treated according to ADA recommendations, which included starting ACE inhibitor. Hypertension was defined per protocol as: use of blood pressure (BP) lowering medications or 1) systolic blood pressure (SBP) ≥95th percentile or diastolic blood pressure (DBP) ≥95th percentile (for age, sex, height) if <18 years or 2) SBP > 140 mmHg or DBP >90 mmHg if ≥18 years. Antihypertensive therapy (antiHTN) was initiated in TODAY according to ADA adult guidelines, with the addition of treatment of BP in the 90th percentile to <95th (dietary) and >95th percentile (dietary and pharmacologic) for those in whom 130/80 mmHg would have been too high a threshold due to age, as previously described [21].
Framingham Heart Study CVD event risk score
The CVD event models from the Framingham Heart Study were developed in, and modeled for, persons 20–59 years. Due to the model constraint and to eliminate the bias, ages <20 were adjusted to 20 for all groups in this study. Full CVD event risks including coronary death, myocardial infarction, and stroke (fatal and nonfatal), angina pectoralis, intermittent claudication, and congestive heart failure. Hard CVD event risks only include coronary death, myocardial infarction, and stroke (fatal and nonfatal).
Statistical analysis
Continuous variables were presented by means and standard deviations, except those with highly skewed distributions, which were summarized by medians and interquartile ranges (IQRs). Categorical variables were presented by numbers and percentages. Baseline variable comparisons between the Teen-LABS and TODAY groups were accomplished by an F test or χ2 test. For categorical measures with limited number of observations, Fisher’s exact test was used. For continuous variables without normal distribution, Wilcoxon rank-sum test was used. Due to the issue of missing data, multiple imputation with fully conditional specification (MI-FCS) [22] was used for variables with high missing or drop-out rate. Fifty imputations were generated. Variables imputed by MI-FCS included: SBP, total cholesterol, high-density lipoprotein cholesterol (HDL-C), BMI, antiHTN treatment, smoking status, diabetes status, and baseline eGFR, UACR, and insulin sensitivity. Smoking and diabetes status for the TODAY cohort were first imputed by the Last Observation Carry Forward (LOCF) method, and then combined with the Teen-LABS cohort for multiple imputation. The data were assumed to be missing at random [23]. Full and hard CVD event risks were calculated using 2 models provided by [12]: 1) the first model included BMI but excluded lipids (total cholesterol and HDL-C); 2) the second model excluded BMI but included lipids (total cholesterol and HDL-C). Sex, age, SBP, antiHTN treatment, smoking status, and diabetes status are used for both risk models. Linear mixed models (LMM) were used for analyzing the relationship between cohort and CVD event risk over time. Compound symmetry covariance structure was used for modeling within-patient variations. LMM for analyzing group effects at baseline were adjusted for baseline eGFR, UACR, and insulin sensitivity only. LMM for analyzing group effects at 1 year and 5 years were further adjusted for baseline risk. Analyses for individual variables of CVD event risk scores were based on observed data. Analyses for CVD event risk scores were based on imputed data. Descriptive statistics (Fig. 3, Supplemental Fig. 1 and 2) and group effect estimates (Fig. 4) were summarized with 50 imputed data analyses using Rubin’s rule [24]. Where appropriate we present the observed data because they are not subject to the assumptions of the imputation techniques. However, omission of incomplete cases in data analysis can result in bias, and therefore our model results use imputed data in an attempt to correct for that bias. All statistical analyses were performed with SAS version 9.4 (SAS Institute Inc.).
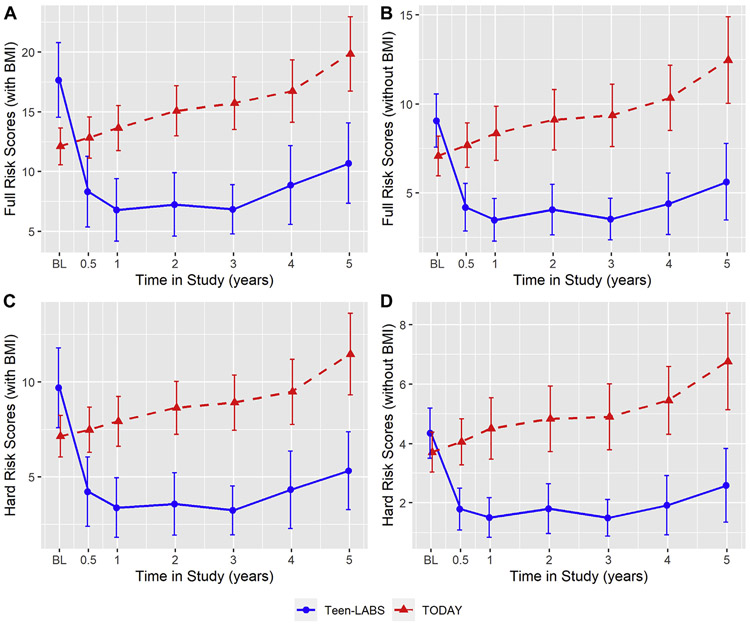
Fig. 3.
Risk scores in TODAY and Teen-LABS over 5 years. Line charts for full CVD risk (with BMI, Fig. 1a), full CVD risk (without BMI, Fig. 1b), hard CVD risk (with BMI, Fig. 1c) and hard CVD risk (without BMI, Fig. 1d). Risk scores were calculated from model using imputed data. Risk score means and standard errors used in the figure were pooled across imputed data analyses using Rubin’s rule. Error bars indicate 95% confidence intervals of the pooled means. Means and error bars were jittered to avoid overlapping.
Fig. 4.
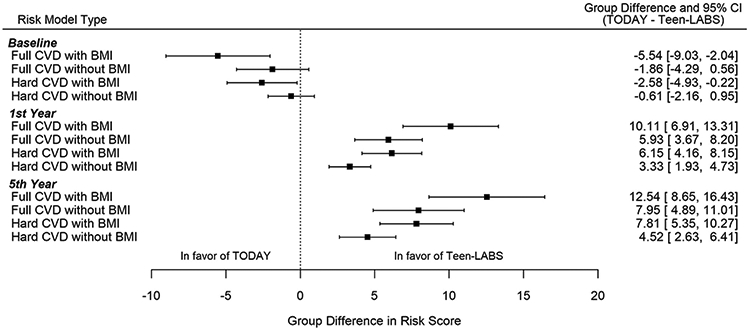
Group differences in risk scores between TODAY and Teen-LABS participants. Group difference indicates the parameter estimate of group effect (Teen-LABS as reference) in mixed model. Mixed model for baseline risks adjusted for baseline estimated glomerular filtration (eGFR), urine albumin-to-creatinine ratio (UACR), and insulin sensitivity. Mixed models for first and fifth year risks adjusted for baseline CVD risk score, eGFR, UACR, and insulin sensitivity.
Results
Baseline comparison between youth with T2D in TODAY versus Teen-LABS
As previously published, participants in Teen-LABS were older and had a higher mean BMI, SBP, DBP, total cholesterol and triglycerides at baseline compared with those in TODAY (Table 1). There was also a high prevalence of antihypertensive use in Teen-LABS versus TODAY (Table 1).
Table 1.
Baseline participant characteristics stratified by study*
| Teen-LABS (n = 30) | TODAY (n = 63) | P value | |
|---|---|---|---|
| Age (yr) | 16.9 ± 1.3 | 15.4 ± 1.3 | <.0001 |
| Sex (female, %) | 70.0 | 55.6 | .18 |
| Race/ethnicity (%) | |||
| Black nonHispanic | 30.0 | 28.6 | .89 |
| Hispanic | 3.3 | 0 | .32 |
| White nonHispanic | 60.0 | 71.4 | .27 |
| Other | 6.7 | 0 | .10 |
| BMI (kg/m2) | 54.4 ± 9.5 | 40.5 ± 4.9 | <.0001 |
| Blood pressure (mmHg) | |||
| Systolic | 129 ± 12 | 119 ± 11.4 | .0002 |
| Diastolic | 76 ± 12 | 70 ± 9 | .02 |
| Hypertension (%) | 66.7 | 25.4 | .0001 |
| Antihypertensive use (%) | 60.0 | 12.7 | <.0001 |
| ACE inhibitor/ARB use (%) | 30.0 | 12.7 | .04 |
| Lipid-lowering medication use (%) | 10.0 | 0 | .03 |
| UACR (μg/mg) | 11 (5–32) | 10 (5–22) | .66 |
| Elevated UAE (≥30 mg/g) (%) | 26.7 | 21.3 | .57 |
| Serum cystatin C (mg/L) | .80 ± .18 | .79 ± .14 | .94 |
| Serum creatinine (mg/dL) | .66 ± .15 | .67 ± .14 | .88 |
| eGFR (mL/min/1.73 m2) | 118 ± 22 | 115 ± 15 | .53 |
| Hyperfiltration (%) | 20.7 | 7.1 | .15 |
| HbA1C (%) | 6.8 ± 1.9 | 6.2 ± .7 | .53 |
| Insulin sensitivity (1/IF, mL/μU) | .04 ± .04 | .04 ± .04 | .14 |
| Total cholesterol (mg/dL) | 172 ± 30 | 147 ± 27 | .0001 |
| HDL-C (mg/dL) | 40 ± 10 | 39 ± 9 | .50 |
| Triglycerides (mg/dL) | 153 ± 68 | 132 ± 100 | .03 |
| Smoking status (%) | 0 | 28.6 | .0005 |
BMI = body mass index, ACE = angiotensin-converting enzyme; ARB = angiotensin II receptor blocker; UACR = urine albumin creatinine ratio; UAE = urinary albumin excretion; eGFR = estimated glomerular filtration; HbA1C = glycated hemoglobin; HDL-C = high density lipoprotein cholesterol.
Data are presented as mean ± standard deviation, median, interquartile range, or percent.
Medical versus surgical intervention on individual variables of CVD event risk scores
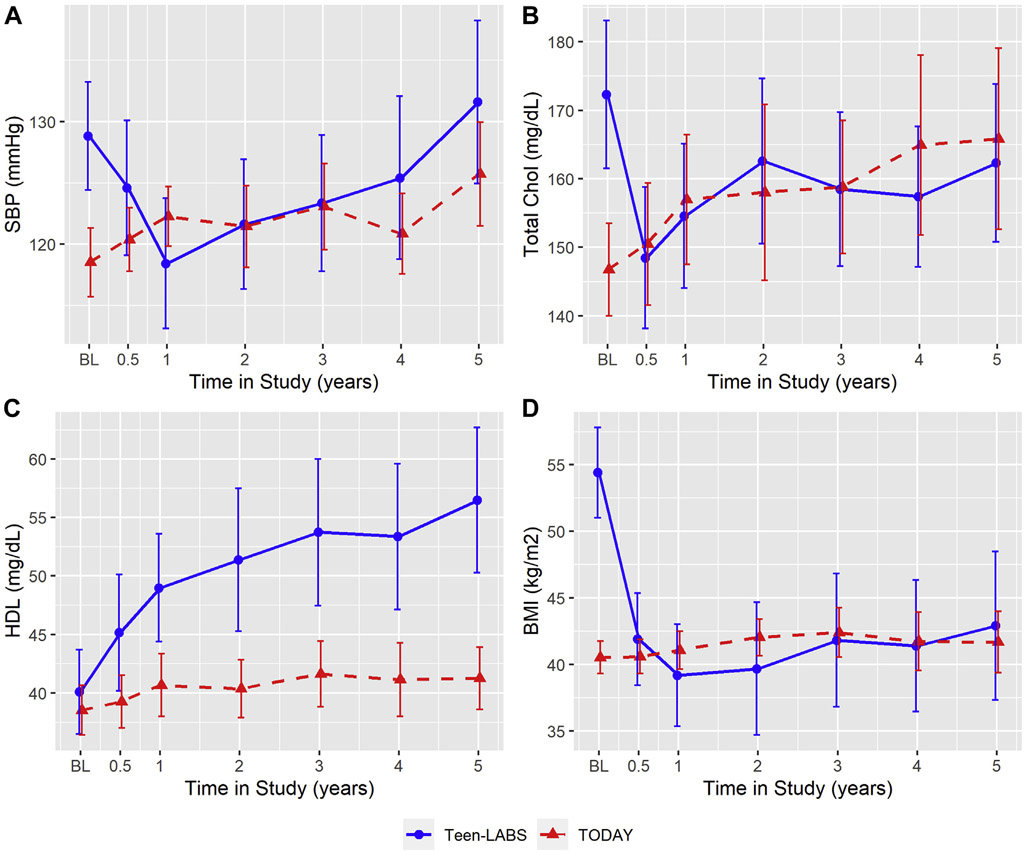
SBP, total cholesterol, HDL and BMI (Fig. 1). SBP (adjusted for medications use) increased from 129 mmHg at baseline to 132 mmHg at year 5 in Teen-LABS participants (P = .23), whereas TODAY participants experienced an increase SBP from 119 mmHg at baseline to 126 mmHg at year 5 (P = .002). Total cholesterol (adjusted for medications use) decreased from 172 mg/dL at baseline to 162 mg/dL at year 5 in Teen-LABS (P = .37). TODAY participants experienced a progressive increase in total cholesterol from baseline 147 mg/dL to 166 mg/dL at year 5 (P < .0001). HDL-C increased from 40 mg/dL at baseline to 57 mg/dL at year 5 in Teen-LABS (P < .0001). Participants in TODAY also experienced a modest increase in HDL-C from 39 mg/dL at baseline to 41 mg/dL at year 5 of follow-up (P = .09). Among Teen-LABS participants, BMI decreased from 54.4 kg/m2 at baseline to 42.9 kg/m2 at year 5 (P < .0001). In TODAY, mean BMI increased from 40.5 kg/m2 at baseline to 41.7 kg/m2 at year 5 (P = .11). The above means and P values are reported from observed data. To account for missing data, these variables were imputed in the CVD risk score calculations, and the imputed data are shown in Supplemental Fig. 1.
Fig. 1.
Individual continuous determinants of full and hard cardiovascular disease (CVD) risk scores in Treatment Options of Type 2 Diabetes in Adolescents and Youth (TODAY) and Teen-Longitudinal Assessment of Bariatric Surgery (Teen-LABS) over 5 years based on observed data line charts for systolic blood pressure (SBP) (mmHg, Fig. 1a), total cholesterol (mg/dL, Fig. 1b), high-density lipoprotein cholesterol (HDL-C) (mg/dL, Fig. 1c) and body mass index (BMI) (kg/m2, Fig. 1d). All figures are based on observed data. Error bars indicate 95% confidence intervals of the means. Means and error bars were jittered to avoid overlapping.
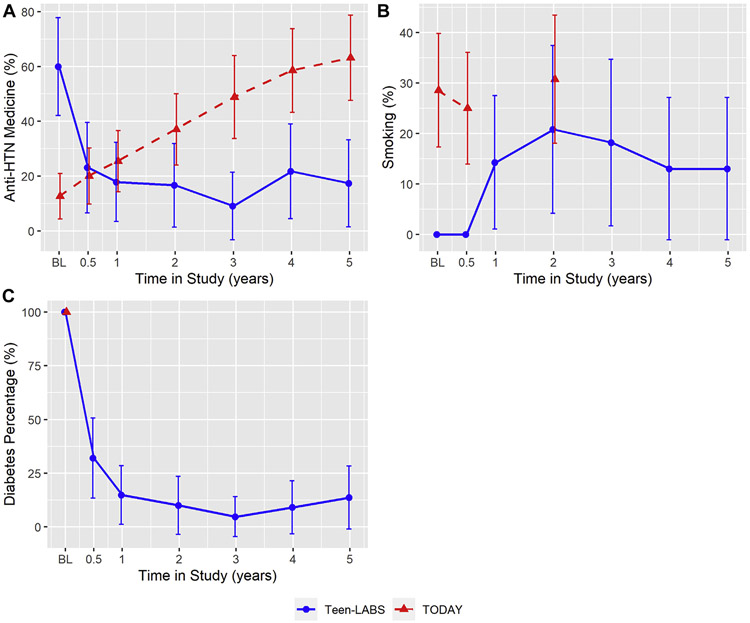
AntiHTN use, smoking and diabetes remission (Fig. 2). The prevalence of antiHTN use decreased by 42.6% (60.0 to 17.4%) from baseline to year 5 in Teen-LABS. In TODAY, prevalence of antiHTN use progressively increased from baseline at 12.7% to 63.2% at year 5. Smoking prevalence increased in Teen-LABS from 0% at baseline to 13.0% at year 5. In Teen-LABS, 85.4% of participants experienced diabetes remission at year 5 with mean HbA1C decreasing from 6.8% at baseline to 5.9% at year 5 of follow-up. In TODAY, glycemic control continued to worsen (P < .0001) (Supplemental Fig. 3). The above frequencies and P values are reported from observed data. Due to missing data, these variables were imputed in the CVD risk score calculations (Supplemental Fig. 2).
Fig. 2.
Individual categorical determinants of full and hard CVD risk scores in TODAY and Teen-LABS over 5 years based on observed data line charts for antihypertensive therapy (antiHTN) medicine (%, Fig. 1a), smoking status (%, Fig. 1b), diabetes status (%, Fig. 1c). All figures are based on observed data. Smoking status at year 1, 3, 4, and 5 were not collected. Diabetes remission was not collected for TODAY cohort (see supplemental Fig. 3 for glycated hemoglobin [HbA1C] trends). Error bars indicate 95% confidence intervals of the mean percentages. Means and error bars were jittered to avoid overlapping.
Medical versus surgical intervention on CVD event risk scores. Fig. 3 shows the risk scores over 5 years in Teen-LABS and TODAY. Fig. 4 shows Forest plots for risk score differences between Teen-LABS and TODAY (Teen-LABS as reference) of full and hard CVD events at baseline, year 1, and year 5. For models with BMI, at baseline the likelihood of CVD events was higher in Teen-LABS versus TODAY (mean [standard error], full CVD events: 17.66 [1.59] versus 12.11 [0.79]%; hard CVD events: 9.70 [1.07] versus 7.15 [.56]%). At baseline, after adjusting for baseline eGFR, UACR and insulin sensitivity, the likelihood of CVD events was higher in Teen-LABS versus TODAY (full CVD events, P = .002; hard CVD events, P = .03). At 1 year follow-up, after multivariable adjustments including baseline risk scores, risk scores in Teen-LABS dropped significantly compared to an increase in TODAY (full CVD events: 6.79 [1.33] versus 13.64 [0.96]%, P < .0001; hard CVD events: 3.39 [.80] versus 7.93 [.67]%, P < .0001). At 5 years follow-up, after multivariable adjustments including baseline risk scores, risk scores in Teen-LABS remained lower than in TODAY (full CVD events: 10.70 [1.71] versus 19.84 [1.59]%, P < .0001; hard CVD events: 5.34 [1.05] versus 11.46 [1.09]%, P < .0001). The differences in risk scores between Teen-LABS and TODAY were attenuated for full CVD without BMI and hard CVD without BMI in multivariable models, but remained statistically significantly different at years 1 and 5 (Fig. 4). The above means and SEs for CVD risk are unadjusted, while P values indicate the significance of group difference after multivariable adjustments.
Missing data.
Missing data are shown in Supplemental Table 1. Notably, data on smoking were only available at baseline, 6 months, and 24 months in TODAY, whereas smoking status was captured at all visits in Teen-LABS. To account for the missing data, all models presented in this manuscript included multiple imputations under missing at random assumption.
Discussion
Among participants in the Teen-LABS and TODAY studies, treatment with MBS resulted in a significant reduction in estimated CVD event risk compared with medical therapy at 1 year and was sustained out to 5 years after treatment. The risk reduction due to MBS was attributed to improved glycemic control, weight loss, changes in blood pressure over time, and an increase in HDL-C. Although MBS induced significant weight loss, when BMI reductions were removed from the prediction models, the robust differences in CVD event risks remained. Despite standard medical treatment (TODAY), a linear increase in estimated 30-year risk CVD events was observed with a doubling in risk over 5 years. These data provide further support for MBS as potential primary treatment option for adolescents with T2D and severe obesity to mitigate CVD and related mortality.
The incidence of youth-onset T2D is increasing in the US [6,25,26], translating to premature mortality from CVD and other chronic diseases such as DKD. There are limited therapeutic options available for adolescents with T2D with the currently FDA approved medications including metformin, insulin, and glucagon-like peptide 1 (GLP-1) receptor agonists. Further compounding the issue, pharmacotherapy may have different effects in youth-onset versus adult-onset T2D as illustrated in the recently completed Restoring Insulin Secretion (RISE) study [7,27,28]. Moreover, data suggest that rates of vascular complications are higher, and insulin resistance and β-cell dysfunction markedly worse in youth with T2D in comparison with adults, calling for novel and targeted approaches to youth-onset T2D [7,27].
MBS is currently the only treatment available in youth with severe obesity and T2D that results in considerable and durable weight loss and improvement in glycemic control in the majority of patients [10,29]. Interestingly, comparing rates of T2D remission in youth versus adults over 5 years found greater rates of T2D resolution in youth compared with adults undergoing MBS [18]. These data suggest that early MBS may be an important intervention to treat youth-onset T2D [18]. Despite this, MBS faces many barriers such as cost, acceptability, insurance coverage, and side effects like excess skin which limit its scalability and wide-spread application [30-33]. Another major consideration for stake-holders is cost-effectiveness of any treatment. Despite the initial cost incurred by MBS in adolescents, it has been found to be cost-effective versus a nonsurgical option over a 5-year period [34].
Most data in youth-onset T2D are limited to Roux-en-Y gastric bypass and it is yet unknown whether similar benefits are afforded by vertical sleeve gastrectomy [18]. There may also be cardioprotective mechanisms or disease duration considerations that are independent of weight and glycemic control which are also influenced by the type of MBS, arguing for prospective, longitudinal, controlled studies to better define the underlying mechanisms of MBS in youth-onset T2D. Among adolescents with and without T2D, we have previously found that the likelihood of CVD events was higher among adolescents undergoing MBS compared with adolescents with severe obesity not referred for MBS, as well as to adolescents with moderate obesity, overweight, and normal weight [13]. The overall CVD risk in this analysis of adolescents with T2D and severe obesity in TODAY and Teen-LABS was high, and the reduction in CVD events was significant, by nearly half as a result of MBS and sustained for 5 years. These data are supported by randomized controlled trials in adults with obesity and T2D comparing MBS to conventional medical therapy [35]. Finally, the high CVD risk observed in TODAY participants, despite their lower baseline BMI, underscores the inadequacy of standard medical therapy in mitigating risk of CVD events, and calls for more aggressive therapy in this at-risk population. The mitigation of CVD risk is likely explained by several factors including attenuation of key risk factors, DKD, and glycemic control, which are prevalent in these cohorts [11,14,18]. Yet the cardioprotective mechanisms of MBS are still incompletely defined in youth with T2D. It remains unclear whether similar benefits are afforded by vertical sleeve gastrectomy as with gastric bypass, and the overall effect of weight loss versus metabolic changes on the cardiovascular benefit needs to be comprehensively detailed. In a prior analysis comparing metabolic outcomes over 5 years of follow-up in adolescents with severe obesity and T2D from the TODAY and Teen-LABS studies, we found worsening of glycemic control, increased BMI, triglycerides, insulin sensitivity, and rates of DKD in participants receiving medical therapy compared with the participants who had MBS [10].
The present study has several strengths including 2 wellcharacterized cohorts of adolescents with T2D with longitudinal follow-up over 5 years and the use of clinically relevant outcomes and measures. However, there are several limitations inherent in the present analysis. The algorithm used to calculate risk reduction makes certain assumptions that deserve mention. The risk scores were originally developed for persons 20–50 years old, therefore, our models used an age of 20 for calculations despite primary data from individuals with mean ages of about 17 years. While this assumption represents a constraint and a study limitation, there is little reason to believe that there are substantive differences in CVD event risk between the mean age of 17 and the 20 years used to conduct our analysis, therefore, this assumption likely does not alter the risk assessment. The sociodemographic distribution of the Framingham cohort (e.g., mainly white and educated) may have led to bias in calculation of risk. Although the risk scores were developed in the Framingham Heart study whose benefits and limitations have been well documented [36,37], the predicted risk of events does not always equate to actual event occurrence and may be an over or underestimated risk as presented in this analysis. In addition, the Teen-LABS and TODAY were not designed to be directly compared, and thus baseline BMI, age, and CVD risk profile were higher in the Teen-LABS cohort versus the TODAY cohort, which could introduce bias regarding response to MBS. The higher baseline BMI in Teen-LABS versus TODAY also likely reflect reluctance of patients, parents, and physicians to consider MBS in adolescents with T2D and obesity. Smoking status was assessed annually in Teen-LABS, but only at baseline, 6 months, and 2 years in TODAY. Moreover, factors we were not able to compare in both Teen-LABS and TODAY such as sleep apnea, nonalcoholic fatty liver disease, and changes in sex steroids in response to MBS versus medical therapy may also influence CVD risk. Finally, adherence to medications, supplements, and lifestyle interventions was a challenge of both cohorts and could have confounded our findings.
Conclusion
In summary, we found that MBS in adolescents with severe obesity and T2D conferred lower calculated risk of CVD events compared with standard medical therapy over 5 years of follow-up. Longer-term outcome studies for adolescents and young adults with T2D undergoing MBS are, however, needed to evaluate whether the risk score predictions hold true. Additionally, studies directly comparing the cardioprotective effects of MBS with newer antidiabetic drugs including sodium glucose co-transporter 2 (SGLT2) inhibitors and GLP-1 receptor and glucose-dependent insulinotropic polypeptide (GIP) agonists, as well as studies combining MBS with these adjunctive therapies to further mitigate residual CVD risk are critical. Finally, mechanistic studies interrogating novel pathways activated by MBS are needed to identify nonsurgical interventions for those adolescents who are deemed unfit for surgery.
Supplementary Material
Acknowledgments
The research protocols of TODAY and Teen-LABS were approved by the relevant institutional review boards and all human participants gave written informed consent. The TODAY Study Group thanks the following companies for donations in support of the study’s efforts: Becton, Dickinson and Company; Bristol-Myers Squibb; Eli Lilly and Company; GlaxoSmithKline; LifeScan, Inc.; Pfizer; Sanofi Aventis. We also gratefully acknowledge the participation and guidance of the American Indian partners associated with the clinical center located at the University of Oklahoma Health Sciences Center, including members of the Absentee Shawnee Tribe, Cherokee Nation, Chickasaw Nation, Choctaw Nation of Oklahoma, and Oklahoma City Area Indian Health Service. The opinions expressed in this paper are those of the authors and do not necessarily reflect the views of the respective Tribes or the Indian Health Service.
Materials developed and used for the TODAY standard diabetes education program and the intensive lifestyle intervention program are available to the public at https://today.bsc.gwu.edu/. A complete list of the members of the TODAY Study Group can be found in the Supplementary Data online. Materials developed and used for the Teen-LABS are available to the public at https://teen-labs.org/.
Drs. Xie and Xu are the guarantors of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Support
The TODAY study was funded by the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK/NIH). This work was completed with funding from NIDDK/NIH grant numbers K23-DK116720, U01-DK61212, U01-DK61230, U01-DK61239, U01-DK61242, and U01-DK61254; from the National Center for Research Resources General Clinical Research Centers Program grant numbers M01-RR00036 (Washington University School of Medicine), M01-RR00043-45 (Children’s Hospital Los Angeles), M01-RR00069 (University of Colorado Denver), M01-RR00084 (Children’s Hospital of Pittsburgh), M01-RR01066 (Massachusetts General Hospital), M01-RR00125 (Yale University), and M01-RR14467 (University of Oklahoma Health Sciences Center); and from the NCRR Clinical and Translational Science Awards grant numbers UL1-RR024134 (Children’s Hospital of Philadelphia), UL1-RR024139 (Yale University), UL1-RR024153 (Children’s Hospital of Pittsburgh), UL1-RR024989 (Case Western Reserve University), UL1-RR024992 (Washington University in St Louis), UL1-RR025758 (Massachusetts General Hospital), and UL1-RR025780 (University of Colorado Denver). NIDDK had no role in study design; collection, analysis, interpretation of data or in writing the report.
The Teen-LABS consortium is funded by cooperative agreements with the NIDDK, through grants: UM1 DK072493 (PI, Dr. Thomas Inge, University of Colorado, Denver), and (PI, Dr. Changchun Xie, University of Cincinnati).
Footnotes
Disclosures
P.B. has acted as a consultant for Bayer, Bristol-Myers Squibb, Boehringer Ingelheim, Sanofi, Novo Nordisk, AstraZeneca, and Horizon Pharma. P.B. serves on the advisory board of XORTX and Boehringer Ingelheim. All support was outside the submitted work. Dr Ryder receives support from Boehringer Ingelheim in the form of drug/placebo.
Supplementary materials
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.1016/j.soard.2020.09.002.
References
- [1].Tancredi M, Rosengren A, Svensson A-M, et al. Excess mortality among persons with type 2 diabetes. N Engl J Med 2015;373(18): 1720–32. [DOI] [PubMed] [Google Scholar]
- [2].Desouza CV, Bolli GB, Fonseca V. Hypoglycemia, diabetes, and cardiovascular events. Diabetes Care 2010;33(6):1389–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [3].Haffner SM, Lehto S, Rönnemaa T, Pyörälä K, Laakso M. Mortality from coronary heart disease in subjects with type 2 diabetes and in nondiabetic subjects with and without prior myocardial infarction. N Engl J Med 1998;339(4):229–34. [DOI] [PubMed] [Google Scholar]
- [4].Dabelea D, Stafford JM, Mayer-Davis EJ, et al. Association of type 1 diabetes vs type 2 diabetes diagnosed during childhood and adolescence with complications during teenage years and young adulthood. JAMA 2017;317(8):825–35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [5].TODAY Study Group. Rapid rise in hypertension and nephropathy in youth with type 2 diabetes: the TODAY clinical trial. Diabetes Care 2013;36(6): 1735–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [6].Al-Saeed AH, Constantino MI, Molyneaux L, et al. An inverse relationship between age of type 2 diabetes onset and complication risk and mortality: the impact of youth-onset type 2 diabetes. Diabetes Care 2016;39(5):823–9. [DOI] [PubMed] [Google Scholar]
- [7].RISE Consortium. Metabolic contrasts between youth and adults with impaired glucose tolerance or recently diagnosed type 2 diabetes: I. observations using the hyperglycemic clamp. Diabetes Care 2018;41 (8): 1696–706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [8].Weiner DE, Tighiouart H, Amin MG, et al. Chronic kidney disease as a risk factor for cardiovascular disease and all-cause mortality: a pooled analysis of community-based studies. J Am Soc Nephrol 2004;15(5): 1307–15. [DOI] [PubMed] [Google Scholar]
- [9].TODAY Study Group, Zeitler P, Hirst K, et al. A clinical trial to maintain glycemic control in youth with type 2 diabetes. N Engl J Med 2012;366(24):2247–56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [10].Inge TH, Laffel LM, Jenkins TM, et al. Comparison of surgical and medical therapy for type 2 diabetes in severely obese adolescents. JAMA pediatrics 2018;172(5):452–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [11].Bjornstad P, Hughan K, Kelsey MM, et al. Effect of surgical versus medical therapy on diabetic kidney disease over 5 years in severely obese adolescents with type 2 diabetes. Diabetes Care 2020;43(1):187–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [12].Pencina MJ, D’Agostino RB Sr, Larson MG, Massaro JM, Vasan RS. Predicting the 30-year risk of cardiovascular disease: the framingham heart study. Circulation 2009;119(24):3078–84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [13].Ryder JR, Xu PX, Inge TH, et al. Thirty-year risk of cardiovascular disease events in adolescents with severe obesity. Obesity 2020;28(3):616–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [14].Bjornstad P, Nehus E, El Ghormli L, et al. Insulin sensitivity and diabetic kidney disease in children and adolescents with type 2 diabetes: an observational analysis of data from the TODAY clinical trial. Am J Kidney Dis 2018;71(1):65–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [15].Inge TH, Courcoulas AP, Jenkins TM, et al. Weight loss and health status 3 years after bariatric surgery in adolescents. N Engl J Med 2016;374(2): 113–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [16].American Diabetes Association. 2. Classification and diagnosis of diabetes: standards of medical care in diabetes-2019. Diabetes Care 2019;42(Suppl 1):S13–28. [DOI] [PubMed] [Google Scholar]
- [17].TODAY Study Group, Zeitler P, Epstein L, et al. Treatment options for type 2 diabetes in adolescents and youth: a study of the comparative efficacy of metformin alone or in combination with rosiglitazone or lifestyle intervention in adolescents with type 2 diabetes. Pediatr Diabetes 2007; 8(2): 74–87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [18].Inge TH, Courcoulas AP, Jenkins TM, et al. Five-year outcomes of gastric bypass in adolescents as compared with adults. N Engl J Med 2019;380(22):2136–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [19].George L, Bacha F, Lee S, Tfayli H, Andreatta E, Arslanian S. Surrogate estimates of insulin sensitivity in obese youth along the spectrum of glucose tolerance from normal to prediabetes to diabetes. J Clin Endocrinol Metab 2011;96(7):2136–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [20].Pottel H, Delanaye P, Schaeffner E, et al. Estimating glomerular filtration rate for the full age spectrum from serum creatinine and cystatin C. Nephrol Dial Transplant 2017;32(3):497–507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [21].Baker-Smith CM, Flinn SK, Flynn JT, et al. Diagnosis, evaluation, and management of high blood pressure in children and adolescents. Pediatrics 2018;142(3). [DOI] [PubMed] [Google Scholar]
- [22].Van Buuren S, Brand JPL, Groothuis-Oudshoorn CGM, Rubin DB. Fully conditional specification in multivariate imputation. J Stat Comput and Simul 2006;76(12):1049–64. [Google Scholar]
- [23].Rubin DB. Inference and missing data. Biometrika 1976;63(3):581–92. [Google Scholar]
- [24].Rubin’s rule. 1987.
- [25].Writing Group for the SfDiYSG, Dabelea D, Bell RA, et al. Incidence of diabetes in youth in the United States. JAMA 2007;297(24):2716–24. [DOI] [PubMed] [Google Scholar]
- [26].Dabelea D, Mayer-Davis EJ, Saydah S, et al. Prevalence of type 1 and type 2 diabetes among children and adolescents from 2001 to 2009. JAMA 2014;311(17):1778–86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [27].RISE Consortium. Impact of insulin and metformin versus metformin alone on beta-cell function in youth with impaired glucose tolerance or recently diagnosed type 2 diabetes. Diabetes Care 2018;41(8):1717–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [28].RISE Consortium. Lack of durable improvements in beta-cell function following withdrawal of pharmacological interventions in adults with impaired glucose tolerance or recently diagnosed type 2 diabetes. Diabetes Care 2019;42(9):1742–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [29].Olbers T, Beamish AJ, Gronowitz E, et al. Laparoscopic Roux-en-Y gastric bypass in adolescents with severe obesity (AMOS): a prospective, 5-year, Swedish nationwide study. Lancet Diabetes Endocrinol 2017;5(3):174–83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [30].Pratt JSA, Browne A, Browne NT, et al. ASMBS pediatric metabolic and bariatric surgery guidelines, 2018. Surg Obes Relat Dis 2018;14(7):882–901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [31].Derderian SC, Dewberry LC, Patten L, et al. Excess skin problems among adolescents after bariatric surgery. Surg Obes Relat Dis 2020;16(8):993–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [32].Armstrong SC, Bolling CF, Michalsky MP, Reichard KW. Pediatric metabolic and bariatric surgery: evidence, barriers, and best practices. Pediatrics 2019;144(6):e20193223. [DOI] [PubMed] [Google Scholar]
- [33].Xanthakos SA, Khoury JC, Inge TH, et al. Nutritional risks in adolescents after bariatric surgery. Clin Gastroenterol Hepatol 2020;18(5):1070–1081.e5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [34].Klebanoff MJ, Chhatwal J, Nudel JD, Corey KE, Kaplan LM, Hur C. Cost-effectiveness of bariatric surgery in adolescents with obesity. JAMA Surg 2017;152(2):136–41. [DOI] [PubMed] [Google Scholar]
- [35].Mingrone G, Panunzi S, De Gaetano A, et al. Bariatric-metabolic surgery versus conventional medical treatment in obese patients with type 2 diabetes: 5 year follow-up of an open-label, single-centre, randomised controlled trial. Lancet 2015;386(9997):964–73. [DOI] [PubMed] [Google Scholar]
- [36].Schlendorf KH, Nasir K, Blumenthal RS. Limitations of the framingham risk score are now much clearer. Prev Med 2009;48(2):115–6. [DOI] [PubMed] [Google Scholar]
- [37].Hemann BA, Bimson WF, Taylor AJ. The framingham risk score: an appraisal of its benefits and limitations. AHHJ 2007;5(2):91–6. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.