Abstract
Post-menopausal osteoporosis is characterized by a negative imbalance between bone formation and bone resorption resulting in a net bone loss, increasing the risk of fracture. One of the earliest interventions to protect against this was hormonal replacement therapy (HRT).
Bone strength depends on both the amount and quality of bone, the latter including compositional / material and structural properties. Bone compositional / material properties are greatly dependent on both patient-, and tissue-age.
Raman spectroscopy is an analytical tool ideally suited for the determination of bone compositional / material properties as a function of tissue age as it is capable of analyzing areas ~1 × 1 μm2 in tetracycline labeled bone forming areas. Using such analysis of humeri from an ovariectomized primate animal model, we reported that loss of estrogen results in alteration in the mineralization regulation mechanisms by osteoid organic matrix attributes at actively forming bone surfaces. In the present work, we used Raman microspectroscopic techniques to compare osteoid and youngest mineralized tissue composition, as well as relationships between osteoid organic matrix quality and quality attributes of the earliest mineralized tissue in paired iliac crest biopsies obtained from early postmenopausal women before and after two years of HRT therapy. Significant correlations between osteoid proteoglycans, sulfated proteoglycans, pyridinoline, and earliest mineralized tissue mineral content were observed, suggesting that in addition to changes in bone turnover rates, HRT affects the osteoid composition, mineralization regulation mechanisms, and potentially fibrillogenesis.
Keywords: Early postmenopause, Hormonal replacement therapy, Bone formation, Raman spectroscopy, Osteoid, Organic matrix, Bone quality
Highlights
-
•
Early postmenopausal women exhibit limited correlations between osteoid composition and earliest deposited mineral content.
-
•
HRT administration for two years affects the osteoid composition.
-
•
HRT administration for two years affects mineralization regulation mechanisms.
1. Introduction
Estrogen is one of the most important hormones, pivotal to bone homeostasis (Khosla and Pacifici, 2021). Postmenopausal osteoporosis (PMOP) is characterized by a negative imbalance between bone formation and resorption, resulting in a net bone mineral density loss, which in turn increases the risk that these patients may sustain one or more fragility fractures. Estrogen depletion due to menopause has been shown to increase osteoclast pre-cursor cells (Manolagas, 1999), decrease osteoclast differentiation by modulating RANKL/OPG on mononuclear osteoclast precursors (Shevde et al., 2000), and delaying osteoclast apoptosis (Parfitt et al., 1996; Hughes et al., 1996).
Based on either volumetric BMD (vBMD) values or rates of change in vBMD by QCT, three stages in PMOP were discernible: (1) an early phase of trabecular bone loss in late premenopause, (2) a transient phase commencing with menopause characterized by rapid cortical bone loss and acceleration of preexisting trabecular bone loss, and (3) a subsequent, slower phase of loss involving similar.
amounts of cortical and trabecular bone lasting indefinitely (Khosla and Pacifici, 2021).
Hormone replacement therapy (HRT) prevents the effects on bone of estrogen depletion due to menopause, by reducing the resorptive activity at the BMU level (Eriksen et al., 1999; Stepan et al., 2019), and averting both increased osteoclast recruitment and delayed apoptosis. The HRT preparation used in this study (Trisekvens®) had demonstrated positive effects on BMD and fractures in previous studies (Christiansen, 1986; Mosekilde et al., 2000). Using Fourier transform infrared microscopic imaging (FTIRI) to determine the effects of 2 years HRT administration in postmenopausal osteoporotic patients' bone quality (defined for the purposes of that study as mineral/matrix ratio, mineral crystallinity/maturity, and relative ratio of enzymatic collagen cross-links [pyridinoline/divalent]) at the ultrastructural level, we reported that the mean values of all parameters were increased after treatment, consistent with suppressed osteoclastic activity, thus increased tissue age (Paschalis et al., 2003). In this published study, precise definition of tissue age as rather large (400 × 400 μm2) areas were analyzed with a spatial resolution of ~6.3 μm, yet bone material / compositional properties are very sensitive to patient age as well as tissue age within the same patient (Paschalis et al., 2016a).
In a primate animal model, we showed that estrogen deficiency due to ovariectomy affects bone quality differently on different skeletal envelopes, significantly altering the rate of mineral accumulation and the kinetics of organic matrix modifications (Paschalis et al., 2017a). Moreover, to the best of our knowledge, for the first time we were able to show that it also altered the regulation mechanisms of mineralization, by focusing on events in the osteoid and the freshly deposited mineralized tissue we showed that (Paschalis et al., 2019). Although nonhuman primates (monkeys) are genetically close to humans, have menstrual cycles and a menopause, and their immune system is similar to humans, menopause occurs much later chronologically compared to humans (Bonucci and Ballanti, 2014). Moreover, it should be kept in mind that while ovariectomy results in bone loss and weaker bones in all animal models, none of these models suffer from fragility fractures, a hallmark of osteoporosis. Additionally, subtle yet definite differences between oophorectomy and menopause have been reported in humans (Pansini et al., 1995; Ohta et al., 1992).
In the present work we used Raman microspectroscopic analysis (affording spatial resolution of 0.6–1 μm) to analyze paired iliac crest biopsies obtained from postmenopausal women at baseline and after 2 years treatment with HRT, to achieve two goals: a) to expand on previously published results (Paschalis et al., 2003) and investigate the effects of this treatment on bone material / compositional properties at precisely defined micro-areas and tissue ages. Specifically, we analyzed osteoid, three tissue ages at forming cortical and trabecular surfaces (based on the presence of double fluorescent labels), and interstitial bone. The following parameters were measured: mineral / matrix ratio, mineral maturity / crystallinity, and tissue water, glycosaminoglycan, and pyridinoline content. b) We have previously shown in a nonhuman primate model that estrogen depletion due to ovariectomy alters the correlations between osteoid composition (GAG and Pyd content) and content of earliest deposited mineral. In the present study, we investigated whether these correlations were absent in the untreated postmenopausal women (in line with what was observed with the ovariectomized animals), and whether HRT restored them. Additionally, in the present work we report on the content of osteoid sulfated proteoglycans (sPG), and their correlation with the content of the earliest deposited mineral.
2. Material & Methods
2.1. Patients
Details have been previously published (Eriksen et al., 1999). Briefly, paired iliac crest biopsies from 10 healthy (patient N = 10, total number of biopsies = 20), early postmenopausal (cessation of menstrual bleeding within 6–24 months before inclusion in the study) women, 45–55 years of age, who were not on any medication known to influence calcium metabolism were obtained. The first biopsy was obtained at baseline, and the second after 2 years on a cyclic HRT (estradiol [2 mg]/norethisterone acetate [1 mg]) treatment (Trisequence; NOVO-Nordisk A/S, Copenhagen, Denmark). Histomorphometric analysis showed that progressive osteoclastic hyperactivity characterizes bone remodeling in early postmenopausal women, and this is reduced by cyclic HRT. Moreover, this reduction of resorptive activity at the BMU level after HRT precedes the reduction in activation frequency demonstrated in older postmenopausal women (Eriksen et al., 1999).
All patients provided informed consent, and the present analysis was approved by the Ethics Commission of the city of Vienna (EK 17–096-VK).
Bone tissues were fixed in alcohol and embedded in poly methyl methacrylate (PMMA). All tissues were processed and stored in an identical manner.
2.2. Raman microspectroscopic analysis
Raman microspectroscopic analysis utilized a Senterra (Bruker Optik GmbH) instrument. The instrument was operated in a temperature-controlled room (constant temperature of 20o C), to minimize any potential performance variability due to ambient temperature fluctuations. It employs SureCAL™ technology to optimize short- and long-term precision, allowing spectral collection which is independent from typical and unforeseen instrument instabilities (SureCAL, n.d.). A continuous laser beam was focused onto the sample through a Raman fluorescence microscope (Olympus BX51, objective 50×) with an excitation of 785 nm (100 mW) and a lateral resolution of ~0.6 μm. The technical characteristics of the instrument have been published elsewhere (Gamsjaeger et al., 2014a). All Raman spectra were obtained in confocal mode (1 μm below the biopsy surface, FlexFocus, Bruker Optics; www.bruker.com). Confocal measurements restrict the sampling depth to a certain region, improve the rejection of stray light and reduce fluorescence interference. For the Raman measurements, the integration time was 5 s and co-additions were 10 to improve the signal to noise ratio (SNR; minimum SNR for a peak to be considered acceptable was 3 (Mcreery, 2000)). Spectra of pure PMMA from every biopsy block were also obtained to check the consistency of the instrument between the different biopsies. The spectra were acquired from the surface of the bone biopsy, using a thermo-electric–cooled charge-coupled device (CCD) (Bruker Optik GmbH). All data analysis was done with the Opus Ident software package (OPUS 7.2, Bruker Optik GmbH). Raman spectra were cut (350–1800 cm−1) and baseline corrected (5-point rubber band) to account for fluorescence background. No further spectra manipulation was performed. If cosmic spikes were evident in any of the collected spectra, these spectra were rejected from further consideration rather than applying smoothing or spike removal algorithms. The following parameters were calculated:
-
i.
The mineral/matrix ratio (MM) from the integrated areas of the v2PO4 (410–460 cm−1) and the amide III (1215–1300 cm−1) bands, which is independent of tissue organization / orientation (Gamsjaeger et al., 2010), unlike the most commonly used v1PO4/amide I ratio. Different from mineral content measures such as BMD, this ratio corrects mineral content for the amount of organic matrix content in the microvolume analyzed. Spectroscopically determined MM has been validated against ash weight measurements (Boskey et al., 1992a), and is directly proportional to bending stiffness and failure moment, as well as a superior predictor of bone-bending stiffness compared to BMD alone (Donnelly et al., 2010a). The mineral content at the youngest tissue age (between the second fluorescent label and the mineralizing front was also estimated based on the ratio of the integrated area of the v2PO4 (410–460 cm − 1) band to the spectral slice 494–509 cm−1 representative of PMMA (Paschalis et al., 2019).
-
ii.
Tissue water content (TW; nanoporosity), approximated by the ratio of the integrated areas of the spectral slice 494–509 cm−1 (PMMA) to Amide III band (Paschalis et al., 2016b). This metric in embedded bone tissue is a surrogate for tissue water in fresh bone tissue. Bone contains water ~20% by volume, and is a major determinant of mechanical properties of bone, especially strength and toughness (Creecy et al., 2016; Granke et al., 2015; Nyman et al., 2016; Nyman et al., 2019).
-
iii.
The glycosaminoglycan (GAG) content was expressed as the GAG / matrix ratio (the ratio of the integrated areas of the proteoglycan/CH3 [1365–1390 cm−1] band [representative of mucopolysaccharides] to the Amide III [1215–1300 cm−1] band) (Gamsjaeger et al., 2014b), validated against a series of standard proteoglycans, as well as model tissues (Gamsjaeger et al., 2014b). GAGs are part of proteoglycans, present in both cartilage and bone. In bone, they fulfill several roles involving the organic matrix assembly, modulate both organic matrix mineralization and remodeling rates (Gualeni et al., 2013; Bi et al., 2006; Xu et al., 1998; Mochida et al., 2009; Mochida et al., 2003), and preserve the perilacunar matrix around the osteocyte lacunae, and the canaliculi in compact lamellar bone mineral-free (Thompson et al., 2011). They undergo posttranslational modifications, some of which are both age- and tissue-age dependent (Grzesik et al., 2002), including size, sulfation, and charge density, all critical for their specific role (Gualeni et al., 2013). Although it is not possible to identify individual proteoglycans by Raman analysis, the major proteoglycans in bone are biglycan and decorin-chondroitin 4-sulfate, accounting for about 90% of the total GAG content (Smith et al., 1997).
-
iv.
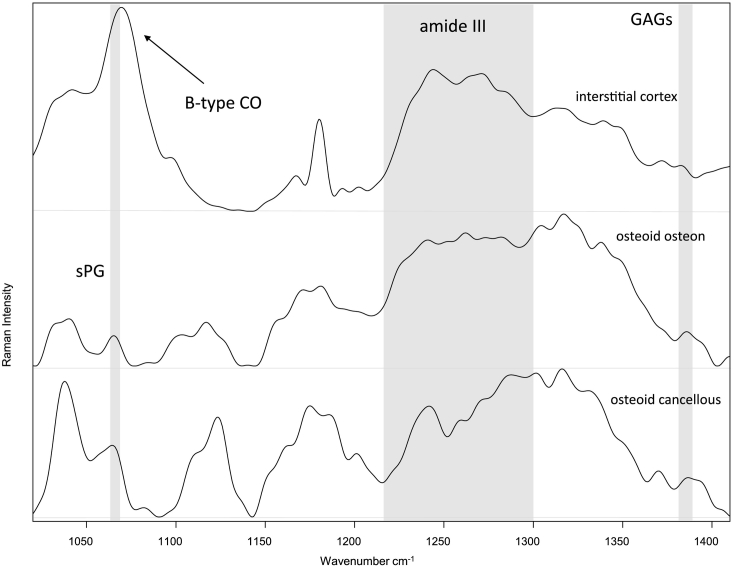
The sulfated proteoglycan content was expressed either as the sPG / organic matrix ratio (the ratio of the absorbance height of proteoglycan at peak ~1062 cm−1 indicative of the OSO3−1 group (Bansil et al., 1978; Ellis et al., 2009) to the Amide III [1215–1300 cm−1] band), or as the sPG / GAG ratio (the ratio of the integrated areas of the proteoglycan peak ~1062 cm−1 indicative of the OSO3−1 group (Bansil et al., 1978; Ellis et al., 2009) to the proteoglycan/CH3 [1365–1390 cm−1] band (representative of mucopolysaccharides). For the sPG calculation, the PMMA was subtracted manually from each Raman spectrum and the peak position was additional verified by second derivative, as collagen has a weak Raman band ~1067 cm−1 (Frushour and Koenig, 1975). Only Raman spectra obtained in the osteoid surface (Fig. 1) were investigated, as in mineralized tissue the peak is overlapped by the strong B- type carbonate peak at ~1070 cm−1 and the v3 phosphate vibrations of apatite (Awonusi et al., 2007; Penel et al., 1998).
-
v.
The pyridinoline (Pyd; enzymatic trivalent collagen cross-link) content was calculated as the absorbance height at 1660 cm−1 / area of the amide I (1620–1700 cm−1) (Gamsjaeger et al., 2017; Paschalis et al., 2001), validated against biochemically characterized collagen cross-linked peptides, as well as biochemically analyzed model tissues (Gamsjaeger et al., 2017). It correlates with mechanical properties at the nano- and whole bone organ levels (McNerny et al., 2015; Paschalis et al., 2011), and is dependent on both age- and tissue-age, as well as health status (Paschalis et al., 2016a). It is the most discriminant bone quality index distinguishing between changes due to healthy aging and postmenopausal osteoporosis (Paschalis et al., 2016a). In cases where bone resistance to fracture cannot be attributed to either BMD or architecture outcomes, or predicted by finite element analysis, this metric positively correlates with fracture incidence rather than predicted fracture risk (Malluche et al., 2013; Misof et al., 2012; Paschalis et al., 2004; Paschalis et al., 2005; Blank et al., 2003; Rokidi et al., 2019).
-
vi.
The maturity/crystallinity (MMC) of the bone mineral apatite crystallites (crystallite chemistry and size) was approximated from the full width at half height (FWHH) of the v1PO4 (930–980 cm−1) band, validated against x-ray diffraction and small angle x-ray spectroscopy (Paschalis et al., 2017b; Kazanci et al., 2006). Size and shape are important mineral crystallite attributes in determining bone strength. Healthy bone has a range of crystallite sizes, which are dependent on both patient- and tissue-age (Paschalis et al., 2016a), while values outside this range associate with fragile bone (Fratzl et al., 2004; Fratzl et al., 1996; Jager and Fratzl, 2000; Gao et al., 2003).
Fig. 1.
Raman spectra obtained in the osteon and trabecular osteoid as well as interstitial bone are shown, with the Raman peak position of the sulfated proteoglycan (sPG) ~ 1062 cm−1, gylcosaminoglycans (GAGs) and the amide III area highlighted. The Raman spectrum from the interstitial bone area of the cortex shows that the sPG area is strongly overlapped by the B-type carbonate Raman band, thus we did not consider sPGs in mineralized bone areas.
2.3. Area selection for analysis
All biopsies were double tetracycline labeled. Raman spectra were acquired at the following microanatomical locations:
-
1.
Osteoid defined as a surface with evident tetracycline labels, 1 μm distance from the mineralizing front, and for which the Raman spectra showed the presence of organic matrix but not mineral, as described elsewhere (Paschalis et al., 2019).
-
2.
TA1: surfaces with evident tetracycline labels, mid-distance between the mineralizing front and the second label.
-
3.
TA2: between (mid-point) two tetracycline labels.
-
4.
TA3: 1 μm behind the 1st label.
-
5.
Interstitial bone (at least 50 μm away from any fluorescent label), and geometrical centers of trabecular bone without any fluorescent label evident at their surfaces, indicative of older tissue age.
2.4. Statistical analysis
The operator was blinded until all the raw spectroscopic data were obtained, at which point the code was broken and the two biopsy groups identified.
Data between the two biopsy groups in the osteoid and interstitial bone were compared with paired t-tests. At the three precisely defined tissue ages at actively forming cortical (osteons) and trabecular surfaces, data were analyzed by 2-way ANOVA.
Correlations between osteoid GAG and Pyd content and TA1 mineral content, and between osteoid GAG and Pyd content were explored. Spearman's or Pearson's r values were calculated (depending on whether the data were normally distributed or not). Additionally, correlations between sulfated proteoglycan content and TA1 mineral content were also investigated, and analyzed by nonlinear regression, followed by extra sum-of-squares F-test.
In all instances, statistical significance was assigned to p < 0.05.
3. Results
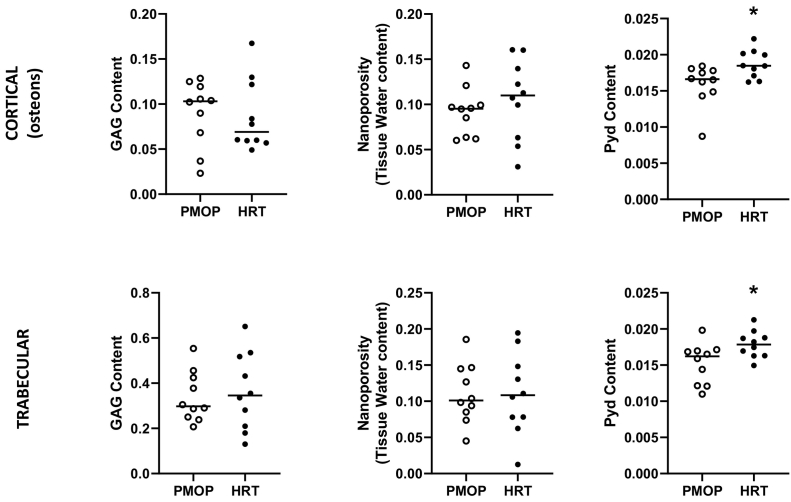
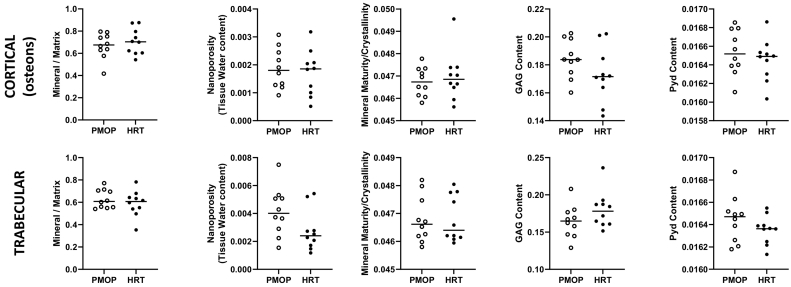
In the osteoid, the Pyd content was significantly elevated after 2 years of HRT therapy (Fig. 2).
Fig. 2.
Osteoid GAG, tissue water, and Pyd content in the osteoid of the forming cortical (top row), and forming trabecular (bottom row) osteoids. Data for the PMOP group are plotted as open, and HRT as solid circles. The only significant difference was the Pyd content (elevated in the HRT group) in both cortical and trabecular osteoid (paired t-test, p < 0.05 in both case).
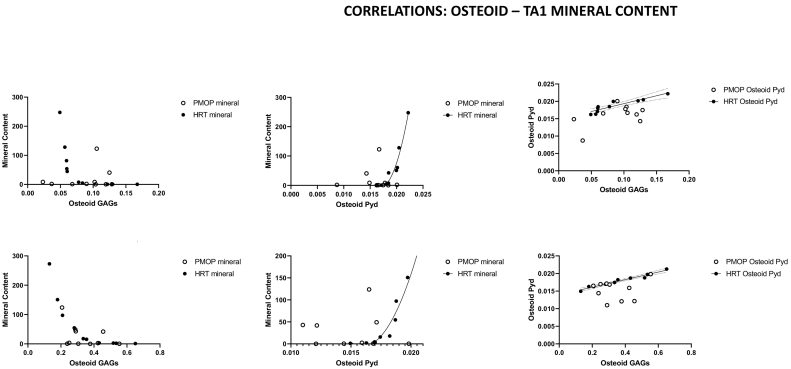
Osteoid GAG and Pyd content significantly correlated with the initial mineral content formed at both cortical (osteons; for PMOP: r = 0.215, p = 0.55 and for HRT: r = −0.632, p = 0.048, respectively) and trabecular actively forming surfaces (for PMOP: r = −0.408, p = 0.242; and for HRT: r = −0.801, p = 0.005, respectively), in the HRT but not in the PMOP group (Fig. 3). Moreover, the GAG and Pyd osteoid content significantly correlated in both the cortical (for cortical PMOP: r = 0.517, p = 0.126; and for HRT: r = 0.948, p < 0.0001) and trabecular (for PMOP: r = 0.134, p = 0.711; for HRT: r = 0.980, p < 0.0001) osteoid in the HRT group only.
Fig. 3.
Correlations between the monitored spectroscopic parameters in the osteoid and the mineral content at TA1 (youngest mineralized tissue formed) for the cortical (top row) and cancellous (bottom row) compartments. Data for the PMOP group are plotted as open, and HRT as solid circles. Osteoid GAG and Pyd content significantly correlated with the mineral content at TA1 at both cortical (osteons; for PMOP: r = 0.215, p = 0.55 and for HRT: r = −0.632, p = 0.048, respectively) and trabecular actively forming surfaces (for PMOP: r = −0.408, p = 0.242; and for HRT: r = −0.801, p = 0.005, respectively), in the HRT but not in the PMOP group. Additionally, the GAG and Pyd osteoid content significantly correlated in both the cortical (for cortical PMOP: r = 0.517, p = 0.126; and for HRT: r = 0.948, p < 0.0001) and trabecular (for PMOP: r = 0.134, p = 0.711; for HRT: r = 0.980, p < 0.0001) osteoid in the HRT group only.
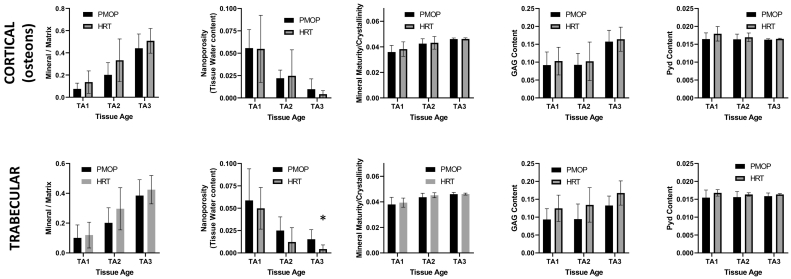
There were no significant differences between the two groups at actively forming surfaces, with the exception of tissue water content which was lower in the HRT group, at the oldest of the three tissue ages considered (Fig. 4).
Fig. 4.
Comparison of spectroscopically determined parameters at actively forming (based on the presence of double fluorescent labels) cortical (top row) and trabecular (bottom row) surfaces, by ANOVA. Data for the PMOP group are plotted as black bars, and HRT as grey bars. Mean and SD values are shown. No significant differences between the two groups was evident, with the exception of tissue water content which was lower in the HRT group, at the oldest of the three tissue ages considered.
No significant differences between the two groups were evident in interstitial bone, in any of the measured parameters (Fig. 5).
Fig. 5.
Comparison of spectroscopically determined parameters at the oldest tissue age considered in the present study for the geometrical centers of cortical (top row) and cancellous compartments. No significant differences (paired t-test) between the two biopsy groups were evident in interstitial bone, in any of the measured parameters.
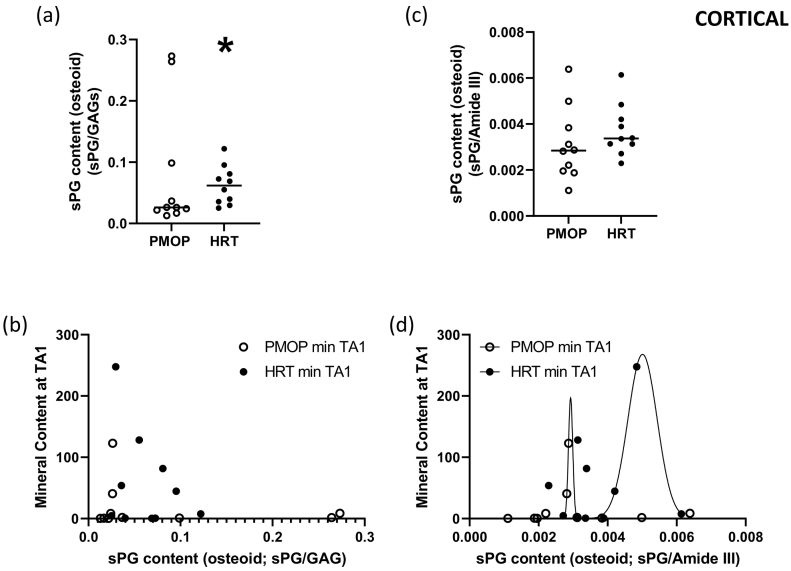
In the cortical compartment, sPG/GAG ratio was significantly elevated in the HRT treated group (p < 0.05), while the ratio of sPG/Amide III although elevated, did not reach significance (Fig. 6a, and c, respectively). Further examination of potential correlations between sPG/GAG ratio and mineral content at TA1 could not be performed, as the two data sets (PMOP and HRT) could not be described by the same non-linear regression model (Fig. 6b). On the other hand, when the sPG / Amide III ratio was considered against the mineral content at TA1, both PMOP and HRT data sets were described by a Gaussian distribution, while the two curves were significantly different (extra sum-of-squares F-test, p = 0.0027; Fig. 6d).
Fig. 6.
In the cortical compartment, sPG/GAG ratio was significantly elevated in the HRT treated group (Wilcoxon matched-pairs signed rank test; p < 0.05), while the ratio of sPG/Amide III although elevated, did not reach significance (Fig. 6a, and c, respectively). The data for the sPG/GAG ratio and mineral content at TA1 could not be fitted by the same non-linear regression model (Fig. 6b). The sPG / Amide III ratio against the mineral content at TA1 for both PMOP and HRT data sets could be fitted by a Gaussian distribution, but the two curves were significantly different (extra sum-of-squares F-test, p = 0.0027; Fig. 6d).
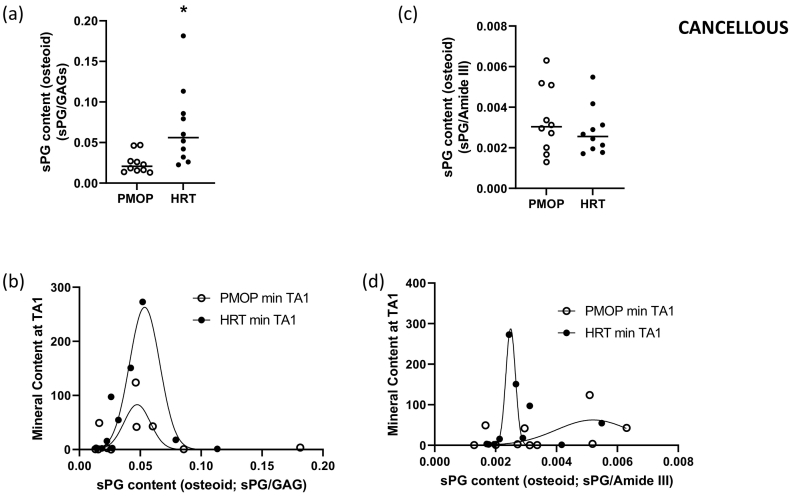
In the cancellous compartment, sPG/GAG ratio was significantly elevated in the HRT treated group (p < 0.05) (Fig. 7a, and c, respectively). Further examination of potential correlations showed that the correlation between either sPG/PG ratio, or sPG/Amide III ratio, and mineral content at TA1 were described by a Gaussian model, while in both instances the curves were significantly different (extra sum-of-squares F-test, p = 0.0009, Fig. 7b; and p = 0.0002, Fig. 7d, respectively).
Fig. 7.
The sPG/GAG ratio in the cancellous compartment was significantly elevated in the HRT treated group (Wilcoxon matched-pairs signed rank test; p < 0.05) (Fig. 7a, and c, respectively). Both the sPG/PG, and sPG/Amide III ratios considered against mineral content at TA1 were described by a Gaussian model, while the curves were significantly different (extra sum-of-squares F-test, p = 0.0009, Fig. 7b; and p = 0.0002, Fig. 7d, respectively).
4. Discussion
HRT is one of the options available to reverse effects of menopausal estrogen depletion on bone quantity and quality.
In a small, randomized, double-blind, prospective clinical trial including 35 early postmenopausal women treated with either HRT (cyclic estradiol/norethisterone acetate) or placebo, Raman microspectroscopic analysis of paired iliac crest biopsies from 10 of them, obtained at baseline and after 2 years of HRT therapy, revealed that HRT administration affected the compositional properties of freshly deposited organic matrix exclusively.
Bone material / compositional properties are important contributors to the determination of bone strength (Fratzl et al., 2004), and are significantly dependent on age, tissue age within the same patient, and health status (Paschalis et al., 2016a; Fratzl et al., 2004; Donnelly et al., 2010b). Moreover, bone is a hierarchically structured composite material consisting of mineral, organic matrix, and water, thus it is expected that the amount and quality of all three components contribute to its mechanical attributes.
There were no significant differences due to 2 years of HRT therapy in the bone quality indices measured in the present study, in either interstitial or actively forming mineralized areas in either cortical or trabecular compartment. We have previously reported using FTIR imaging analysis of large areas from the same iliac crest biopsies analyzed in the present study that HRT therapy has an effect on the mineral maturity / crystallinity and enzymatic collagen cross-links ratio properties of bone tissue (Paschalis et al., 2003). This apparent discrepancy may be explained by the fact that while tissue age was precisely accounted for in the present study (thus changes due to variable bone turnover rates may be excluded), the already published results reflect changes due to changes in bone turnover, thus average tissue age.
On the other hand, significant differences due to HRT therapy were observed in the osteoid, as well as the relationship between osteoid components and the content of the youngest mineral deposited. Specifically, Pyd content was elevated after 2 years of HRT therapy. The reason for this is not clear, but it may not be attributed to the suppression of bone turnover rates previously reported (Eriksen et al., 1999), as the comparison between the two groups here was between newly formed osteoid, thus changes in the average tissue age may be excluded.
Significant negative correlations were observed between osteoid GAG content and mineral content at the youngest mineralized surfaces, in the HRT but not in the PMOP group. Additionally, significant positive correlations were evident between osteoid Pyd content and mineral content at the youngest mineralized surfaces, in the HRT but not in the PMOP group. GAGs are part of the proteoglycans which fulfill multiple roles in bone, one of which is negative modulation of mineralization (Mochida et al., 2009; Boskey et al., 1997a; Chen and Boskey, 1985; Boskey et al., 1992b; Boskey et al., 1991; Boskey et al., 1997b), while collagen cross-link content has been reported to modulate mineralization (Wassen et al., 2000), amongst other roles in bone homeostasis and determination of bone strength. Thus, the correlation between osteoid GAGs (negative) and Pyd (positive) content, and earliest deposited mineral content would be in agreement with these publications. We have previously shown in an ovariectomized monkey animal model, that while similar correlations exist in the SHAM-operated animals, they are lost in the ovariectomized ones (Paschalis et al., 2019). The lack of any correlations in the PMOP group would be in agreement with what was observed in the ovariectomized animals (Paschalis et al., 2019), while their manifestation in the HRT group suggests that this therapy restores the mineralization regulation mechanisms.
Proteoglycans fulfill a multitude of roles in mineralizing tissues, and although they have been shown to negatively modulate mineralization, one sub-category of them, namely sulfated proteoglycans, have been reported to be mineralization promoters (Lormée et al., 1996; Arias et al., 2004; Slater et al., 1994). In both cortical and cancellous compartments, the ratio of sPG/GAG in the osteoid was significantly higher in the HRT group, unlike the sPG/Amide III ratio. This may be attributed to the fact that while GAGs are present in PGs, Amide III encompasses all organic matrix moieties present in the osteoid. Further exploring the relationship between sPGs and mineral content at TA1, we found that this relationship is different between the PMOP and HRT groups, a finding that suggests that the mineralization regulation mechanism is altered between the two groups, in general agreement with what we previously reported in a primate ovariectomized animal model (Paschalis et al., 2019). Moreover, it has been shown that in murine bladder, the level of estrogen does not affect the initiation of the polysaccharide chain, but instead affects the elongation and sulfation (Anand et al., 2012).
Early on, it was postulated that the acidic glycosaminoglycan component of proteoglycans most likely controls the size and organization of collagen fibers (Borcherding et al., 1975). Nowadays, it is well established that collagen intimately interacts with proteoglycans (Mochida et al., 2003; Chen et al., 2020; Robins, 2006; Robins, 2007; Kalamajski and Oldberg, 2010); it's striated appearance, as well as the diameter of the fibrils (which is tissue-specific and unique for each collagen type), is greatly influenced by its association with proteoglycans (Junqueira and Montes, 1983). Glycosaminoglycans (GAGs), part of proteoglycans, are, generally composed of repeating disaccharides possessing sulfated sugars with the exception of hyaluronic acid (Junqueira and Montes, 1983). In aqueous environments, the polysaccharide chains of GAGs electrostatically repel each other, filling any space available (Junqueira and Montes, 1983). The acid groups of GAGs bind to the basic groups of collagen (the amino groups present in the side chains of collagen lysyl and arginyl residues), thus held in the extracellular matrix through their interaction with collagen (Junqueira and Montes, 1983). They are also reported to be involved in the non-enzymatic conversion of immature, divalent collagen cross-links into mature trivalent ones (Pyd being of the latter). This collagen cross-link conversion is thought to be controlled by the microenvironment (presence of mineral), the glycosylation state of the immature cross-links, and the presence of proteoglycans around the collagen cross-linking sites (Terajima et al., 2014). Collagen fibrillogenesis is a multistep process concerning assembly of molecules into fibrils and bundles of fibrils. In in vitro experiments, dermatan sulphate and chondroitin sulphate do not change the diameter of fibrils formed during the early stages of fibrillogenesis (nucleation), whereas proteoglycans added during the final stages of collagen fiber formation cause changes in ultimate tensile strength. Chondroitin sulphate proteoglycans have been reported to result in efficient stress transfer between collagen fibrils, modulating the ultimate tensile strength. (Garg et al., 1989). When we explored potential correlations between GAGs and Pyd content in the osteoid, the data indicated that significant correlations in both cortical and trabecular bone compartments were evident in the HRT group only, possibly suggesting that HRT therapy influences fibrillogenesis. This suggestion would agree with published data showing that both estrogen and estrogen receptors are involved in fibrillogenesis (Markiewicz et al., 2013; Magruder et al., 2014). These correlations were not evident in PMOP.
Immediately following menopause, there is a significant increase in bone turnover rates which eventually levels off (Khosla and Pacifici, 2021). The major effect of this on bone compositional/material properties is a reduction of average tissue age. In the present study we compared properties at precisely defined tissue ages, thus the confounding factor of altered bone turnover rates is lifted.
A limitation of the present study is that no iliac crest biopsies were available from placebo-treated patients, and the N is small. One the other hand, the analyzed biopsies were paired.
In summary, the results of the present study offer indications that in addition to changes in bone turnover rates, HRT affects the osteoid composition, mineralization regulation mechanisms, and possibly fibrillogenesis.
Credit author statement
All authors participated in the study conceptualization, experiment performance, data analysis and interpretation, and drafting of the manuscript.
Declaration of competing interest
None of the authors has any conflict of interest.
Acknowledgements
The present study was supported by the Allgemeine Unfallversicherungsanstalt (AUVA), research funds of the Austrian workers compensation board, and the Österreichische Gesundheitskasse (ÖGK).
References
- Anand M., Wang C., French J., Isaacson-Schmid M., Wall L.L., Mysorekar I.U. Estrogen affects the glycosaminoglycan layer of the murine bladder. Female Pelvic Med. Reconstr. Surg. 2012;18(3):148–152. doi: 10.1097/SPV.0b013e31824b76bd. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arias J.L., Neira-Carrillo A., Arias J.I., Escobar C., Bodero M., David M., Fernández M.S. Sulfated polymers in biological mineralization: a plausible source for bio-inspired engineering. J. Mater. Chem. 2004;14(14):2154–2160. [Google Scholar]
- Awonusi A., Morris M.D., Tecklenburg M.M. Carbonate assignment and calibration in the Raman spectrum of apatite. Calcif. Tissue Int. 2007;81(1):46–52. doi: 10.1007/s00223-007-9034-0. [DOI] [PubMed] [Google Scholar]
- Bansil R., Yannas I.V., Stanley H.E. Raman spectroscopy: a structural probe of glycosaminoglycans. Biochim. Biophys. Acta. 1978;541(4):535–542. doi: 10.1016/0304-4165(78)90163-0. [DOI] [PubMed] [Google Scholar]
- Bi Y., Nielsen K.L., Kilts T.M., Yoon A., M A.K., Wimer H.F., Greenfield E.M., Heegaard A.M., Young M.F. Biglycan deficiency increases osteoclast differentiation and activity due to defective osteoblasts. Bone. 2006;38(6):778–786. doi: 10.1016/j.bone.2005.11.005. [DOI] [PubMed] [Google Scholar]
- Blank R.D., Baldini T.H., Kaufman M., Bailey S., Gupta R., Yershov Y., Boskey A.L., Coppersmith S.N., Demant P., Paschalis E.P. Spectroscopically determined collagen Pyr/deH-DHLNL cross-link ratio and crystallinity indices differ markedly in recombinant congenic mice with divergent calculated bone tissue strength. Connect. Tissue Res. 2003;44(3–4):134–142. doi: 10.1080/03008200390223918. [DOI] [PubMed] [Google Scholar]
- Bonucci E., Ballanti P. Osteoporosis-bone remodeling and animal models. Toxicol. Pathol. 2014;42(6):957–969. doi: 10.1177/0192623313512428. [DOI] [PubMed] [Google Scholar]
- Borcherding M.S., Blacik L.J., Sittig R.A., Bizzell J.W., Breen M., Weinstein H.G. Proteoglycans and collagen fibre organization in human corneoscleral tissue. Exp. Eye Res. 1975;21(1):59–70. doi: 10.1016/0014-4835(75)90057-3. [DOI] [PubMed] [Google Scholar]
- Boskey A.L., Maresca M., Wikstrom B., Hjerpe A. Hydroxyapatite formation in the presence of proteoglycans of reduced sulfate content: studies in the brachymorphic mouse. Calcif. Tissue Int. 1991;49(6):389–393. doi: 10.1007/BF02555848. [DOI] [PubMed] [Google Scholar]
- Boskey A., Pleshko N., Doty S., Mendelsohn R. Applications of Fourier transform infrared (FT-IR) microscopy to the study of mineralization in bone and cartilage. Cell Mater. 1992;2:209–220. [Google Scholar]
- Boskey A.L., Maresca M., Armstrong A.L., Ehrlich M.G. Treatment of proteoglycan aggregates with physeal enzymes reduces their ability to inhibit hydroxyapatite proliferation in a gelatin gel. J. Orthop. Res. 1992;10(3):313–319. doi: 10.1002/jor.1100100302. [DOI] [PubMed] [Google Scholar]
- Boskey A.L., Stiner D., Binderman I., Doty S.B. Effects of proteoglycan modification on mineral formation in a differentiating chick limb-bud mesenchymal cell culture system. J. Cell. Biochem. 1997;64(4):632–643. doi: 10.1002/(sici)1097-4644(19970315)64:4<632::aid-jcb11>3.0.co;2-e. [DOI] [PubMed] [Google Scholar]
- Boskey A.L., Spevak L., Doty S.B., Rosenberg L. Effects of bone CS-proteoglycans, DS-decorin, and DS-biglycan on hydroxyapatite formation in a gelatin gel. Calcif. Tissue Int. 1997;61(4):298–305. doi: 10.1007/s002239900339. [DOI] [PubMed] [Google Scholar]
- Chen C.C., Boskey A.L. Mechanisms of proteoglycan inhibition of hydroxyapatite growth. Calcif. Tissue Int. 1985;37(4):395–400. doi: 10.1007/BF02553709. [DOI] [PubMed] [Google Scholar]
- Chen D., Smith L.R., Khandekar G., Patel P., Yu C.K., Zhang K., Chen C.S., Han L., Wells R.G. Distinct effects of different matrix proteoglycans on collagen fibrillogenesis and cell-mediated collagen reorganization. Sci. Rep. 2020;10(1):19065. doi: 10.1038/s41598-020-76107-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Christiansen C. Estrogen/progestogen as a prophylactic treatment of postmenopausal osteoporosis. Acta Pharmacol. Toxicol. (Copenh) 1986;59(Suppl. 7):305–310. doi: 10.1111/j.1600-0773.1986.tb02769.x. [DOI] [PubMed] [Google Scholar]
- Creecy A., Uppuganti S., Merkel A.R., O’Neal D., Makowski A.J., Granke M., Voziyan P., Nyman J.S. Changes in the fracture resistance of bone with the progression of type 2 diabetes in the ZDSD rat. Calcif. Tissue Int. 2016;99(3):289–301. doi: 10.1007/s00223-016-0149-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Donnelly E., Chen D.X., Boskey A.L., Baker S.P., van der Meulen M.C. Contribution of mineral to bone structural behavior and tissue mechanical properties. Calcif. Tissue Int. 2010;87(5):450–460. doi: 10.1007/s00223-010-9404-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Donnelly E., Boskey A.L., Baker S.P., van der Meulen M.C. Effects of tissue age on bone tissue material composition and nanomechanical properties in the rat cortex. J. Biomed. Mater. Res. A. 2010;92(3):1048–1056. doi: 10.1002/jbm.a.32442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ellis R., Green E., Winlove C.P. Structural analysis of glycosaminoglycans and proteoglycans by means of Raman microspectrometry. Connect. Tissue Res. 2009;50(1):29–36. doi: 10.1080/03008200802398422. [DOI] [PubMed] [Google Scholar]
- Eriksen E.F., Langdahl B., Vesterby A., Rungby J., Kassem M. Hormone replacement therapy prevents osteoclastic hyperactivity: a histomorphometric study in early postmenopausal women. J. Bone Miner. Res. 1999;14(7):1217–1221. doi: 10.1359/jbmr.1999.14.7.1217. [DOI] [PubMed] [Google Scholar]
- Fratzl P., Schreiber S., Roschger P., Lafage M.H., Rodan G., Klaushofer K. Effects of sodium fluoride and alendronate on the bone mineral in minipigs: a small-angle X-ray scattering and backscattered electron imaging study. J. Bone Miner. Res. 1996;11(2):248–253. doi: 10.1002/jbmr.5650110214. [DOI] [PubMed] [Google Scholar]
- Fratzl P., Gupta H., Paschalis E., Roschger P. Structure and mechanical quality of the collagen–mineral nano-composite in bone. J. Mater. Chem. 2004;14:2115–2123. [Google Scholar]
- Frushour B.G., Koenig J.L. Raman scattering of collagen, gelatin, and elastin. Biopolymers. 1975;14(2):379–391. doi: 10.1002/bip.1975.360140211. [DOI] [PubMed] [Google Scholar]
- Gamsjaeger S., Masic A., Roschger P., Kazanci M., Dunlop J.W., Klaushofer K., Paschalis E.P., Fratzl P. Cortical bone composition and orientation as a function of animal and tissue age in mice by Raman spectroscopy. Bone. 2010;47:392–399. doi: 10.1016/j.bone.2010.04.608. [DOI] [PubMed] [Google Scholar]
- Gamsjaeger S., Hofstetter B., Fratzl-Zelman N., Roschger P., Roschger A., Fratzl P., Brozek W., Masic A., Misof B.M., Glorieux F.H., Klaushofer K., Rauch F., Paschalis E.P. Pediatric reference Raman data for material characteristics of iliac trabecular bone. Bone. 2014;69:89–97. doi: 10.1016/j.bone.2014.09.012. [DOI] [PubMed] [Google Scholar]
- Gamsjaeger S., Klaushofer K., Paschalis E. Raman analysis of proteoglycans simultaneously in bone and cartilage. J. Raman Spectrosc. 2014;45(9):794–800. [Google Scholar]
- Gamsjaeger S., Robins S.P., Tatakis D.N., Klaushofer K., Paschalis E.P. Identification of Pyridinoline trivalent collagen cross-links by Raman microspectroscopy. Calcif. Tissue Int. 2017;100(6):565–574. doi: 10.1007/s00223-016-0232-5. [DOI] [PubMed] [Google Scholar]
- Gao H., Ji B., Jager I.L., Arzt E., Fratzl P. Materials become insensitive to flaws at nanoscale: lessons from nature. Proc. Natl. Acad. Sci. U. S. A. 2003;100(10):5597–5600. doi: 10.1073/pnas.0631609100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garg A.K., Berg R.A., Silver F.H., Garg H.G. Effect of proteoglycans on type I collagen fibre formation. Biomaterials. 1989;10(6):413–419. doi: 10.1016/0142-9612(89)90133-6. [DOI] [PubMed] [Google Scholar]
- Granke M., Does M.D., Nyman J.S. The role of water compartments in the material properties of cortical bone. Calcif. Tissue Int. 2015;97(3):292–307. doi: 10.1007/s00223-015-9977-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grzesik W.J., Frazier C.R., Shapiro J.R., Sponseller P.D., Robey P.G., Fedarko N.S. Age-related changes in human bone proteoglycan structure. Impact of osteogenesis imperfecta. J. Biol. Chem. 2002;277(46):43638–43647. doi: 10.1074/jbc.M202124200. [DOI] [PubMed] [Google Scholar]
- Gualeni B., de Vernejoul M.C., Marty-Morieux C., De Leonardis F., Franchi M., Monti L., Forlino A., Houillier P., Rossi A., Geoffroy V. Alteration of proteoglycan sulfation affects bone growth and remodeling. Bone. 2013;54(1):83–91. doi: 10.1016/j.bone.2013.01.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hughes D.E., Dai A., Tiffee J.C., Li H.H., Mundy G.R., Boyce B.F. Estrogen promotes apoptosis of murine osteoclasts mediated by TGF-beta. Nat. Med. 1996;2(10):1132–1136. doi: 10.1038/nm1096-1132. [DOI] [PubMed] [Google Scholar]
- Jager I., Fratzl P. Mineralized collagen fibrils: a mechanical model with a staggered arrangement of mineral particles. Biophys. J. 2000;79(4):1737–1746. doi: 10.1016/S0006-3495(00)76426-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Junqueira L.C., Montes G.S. Biology of collagen-proteoglycan interaction. Arch. Histol. Jpn. 1983;46(5):589–629. doi: 10.1679/aohc.46.589. [DOI] [PubMed] [Google Scholar]
- Kalamajski S., Oldberg A. The role of small leucine-rich proteoglycans in collagen fibrillogenesis. Matrix Biol. 2010;29(4):248–253. doi: 10.1016/j.matbio.2010.01.001. [DOI] [PubMed] [Google Scholar]
- Kazanci M., Fratzl P., Klaushofer K., Paschalis E.P. Complementary information on in vitro conversion of amorphous (precursor) calcium phosphate to hydroxyapatite from Raman microspectroscopy and wide-angle X-ray scattering. Calcif. Tissue Int. 2006;79(5):354–359. doi: 10.1007/s00223-006-0011-9. [DOI] [PubMed] [Google Scholar]
- Khosla S., Pacifici R. Estrogen deficiency and the pathogenesis of osteoporosis. In: Dempster D., Cauley J., Bouxsein M., Cosman F., editors. Marcus and Feldman's Osteoporosis. Academic Press; London, United Kingdom: 2021. pp. 773–797. [Google Scholar]
- Lormée P., Septier D., Lécolle S., Baudoin C., Goldberg M. Dual incorporation of (35S)sulfate into dentin proteoglycans acting as mineralization promotors in rat molars and predentin proteoglycans. Calcif. Tissue Int. 1996;58(5):368–375. doi: 10.1007/BF02509387. [DOI] [PubMed] [Google Scholar]
- Magruder H.T., Quinn J.A., Schwartzbauer J.E., Reichner J., Huang A., Filardo E.J. The G protein-coupled estrogen receptor-1, GPER-1, promotes fibrillogenesis via a Shc-dependent pathway resulting in anchorage-independent growth. Horm Cancer. 2014;5(6):390–404. doi: 10.1007/s12672-014-0195-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malluche H.H., Porter D.S., Mawad H., Monier-Faugere M.C., Pienkowski D. Low-energy fractures without low T-scores characteristic of osteoporosis: a possible bone matrix disorder. J. Bone Joint Surg. Am. 2013;95(19):e1391–e1396. doi: 10.2106/JBJS.L.01281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manolagas S.C. Cell number versus cell vigor--what really matters to a regenerating skeleton? Endocrinology. 1999;140(10):4377–4381. doi: 10.1210/endo.140.10.7129. [DOI] [PubMed] [Google Scholar]
- Markiewicz M., Znoyko S., Stawski L., Ghatnekar A., Gilkeson G., Trojanowska M. A role for estrogen receptor-alpha and estrogen receptor-beta in collagen biosynthesis in mouse skin. J. Invest. Dermatol. 2013;133(1):120–127. doi: 10.1038/jid.2012.264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McNerny E.M., Gong B., Morris M.D., Kohn D.H. Bone fracture toughness and strength correlate with collagen cross-link maturity in a dose-controlled lathyrism mouse model. J. Bone Miner. Res. 2015;30(3):455–464. doi: 10.1002/jbmr.2356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mcreery R. Signal-to-noise in Raman spectroscopy. In: Winefordner J., McCreery R., editors. Raman Spectroscopy for Chemical Analysis. 2000. pp. 49–71. [Google Scholar]
- Misof B.M., Gamsjaeger S., Cohen A., Hofstetter B., Roschger P., Stein E., Nickolas T.L., Rogers H.F., Dempster D., Zhou H., Recker R., Lappe J., McMahon D., Paschalis E.P., Fratzl P., Shane E., Klaushofer K. Bone material properties in premenopausal women with idiopathic osteoporosis. J. Bone Miner. Res. 2012;27(12):2551–2561. doi: 10.1002/jbmr.1699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mochida Y., Duarte W.R., Tanzawa H., Paschalis E.P., Yamauchi M. Decorin modulates matrix mineralization in vitro. Biochem. Biophys. Res. Commun. 2003;305(1):6–9. doi: 10.1016/s0006-291x(03)00693-4. [DOI] [PubMed] [Google Scholar]
- Mochida Y., Parisuthiman D., Pornprasertsuk-Damrongsri S., Atsawasuwan P., Sricholpech M., Boskey A.L., Yamauchi M. Decorin modulates collagen matrix assembly and mineralization. Matrix Biol. 2009;28(1):44–52. doi: 10.1016/j.matbio.2008.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mosekilde L., Beck-Nielsen H., Sorensen O.H., Nielsen S.P., Charles P., Vestergaard P., Hermann A.P., Gram J., Hansen T.B., Abrahamsen B., Ebbesen E.N., Stilgren L., Jensen L.B., Brot C., Hansen B., Tofteng C.L., Eiken P., Kolthoff N. Hormonal replacement therapy reduces forearm fracture incidence in recent postmenopausal women - results of the Danish osteoporosis prevention study. Maturitas. 2000;36(3):181–193. doi: 10.1016/s0378-5122(00)00158-4. [DOI] [PubMed] [Google Scholar]
- Nyman J.S., Granke M., Singleton R.C., Pharr G.M. Tissue-level mechanical properties of bone contributing to fracture risk. Curr. Osteoporos. Rep. 2016;14(4):138–150. doi: 10.1007/s11914-016-0314-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nyman J.S., Uppuganti S., Unal M., Leverant C.J., Adabala S., Granke M., Voziyan P., Does M.D. Manipulating the amount and structure of the organic matrix affects the water compartments of human cortical bone. JBMR Plus. 2019;3(6) doi: 10.1002/jbm4.10135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ohta H., Masuzawa T., Ikeda T., Suda Y., Makita K., Nozawa S. Which is more osteoporosis-inducing, menopause or oophorectomy? Bone Miner. 1992;19(3):273–285. doi: 10.1016/0169-6009(92)90876-f. [DOI] [PubMed] [Google Scholar]
- Pansini F., Bagni B., Bonaccorsi G., Albertazzi P., Zanotti L., Farina A., Campobasso C., Orlandi R., Mollica G. Oophorectomy and spine bone density: evidence of a higher rate of bone loss in surgical compared with spontaneous menopause. Menopause. 1995;2(2):109–116. [Google Scholar]
- Parfitt A.M., Mundy G.R., Roodman G.D., Hughes D.E., Boyce B.F. A new model for the regulation of bone resorption, with particular reference to the effects of bisphosphonates. J. Bone Miner. Res. 1996;11(2):150–159. doi: 10.1002/jbmr.5650110203. [DOI] [PubMed] [Google Scholar]
- Paschalis E.P., Verdelis K., Doty S.B., Boskey A.L., Mendelsohn R., Yamauchi M. Spectroscopic characterization of collagen cross-links in bone. J. Bone Miner. Res. 2001;16(10):1821–1828. doi: 10.1359/jbmr.2001.16.10.1821. [DOI] [PubMed] [Google Scholar]
- Paschalis E.P., Boskey A.L., Kassem M., Eriksen E.F. Effect of hormone replacement therapy on bone quality in early postmenopausal women. J. Bone Miner. Res. 2003;18(6):955–959. doi: 10.1359/jbmr.2003.18.6.955. [DOI] [PubMed] [Google Scholar]
- Paschalis E.P., Shane E., Lyritis G., Skarantavos G., Mendelsohn R., Boskey A.L. Bone fragility and collagen cross-links. J. Bone Miner. Res. 2004;19(12):2000–2004. doi: 10.1359/JBMR.040820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paschalis E.P., Glass E.V., Donley D.W., Eriksen E.F. Bone mineral and collagen quality in iliac crest biopsies of patients given teriparatide: new results from the fracture prevention trial. J. Clin. Endocrinol. Metab. 2005;90(8):4644–4649. doi: 10.1210/jc.2004-2489. [DOI] [PubMed] [Google Scholar]
- Paschalis E.P., Tatakis D.N., Robins S., Fratzl P., Manjubala I., Zoehrer R., Gamsjaeger S., Buchinger B., Roschger A., Phipps R., Boskey A.L., Dall’ara E., Varga P., Zysset P., Klaushofer K., Roschger P. Lathyrism-induced alterations in collagen cross-links influence the mechanical properties of bone material without affecting the mineral. Bone. 2011;49:1232–1241. doi: 10.1016/j.bone.2011.08.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paschalis E.P., Fratzl P., Gamsjaeger S., Hassler N., Brozek W., Eriksen E.F., Rauch F., Glorieux F.H., Shane E., Dempster D., Cohen A., Recker R., Klaushofer K. Aging versus postmenopausal osteoporosis: bone composition and maturation kinetics at actively-forming trabecular surfaces of female subjects aged 1 to 84 years. J. Bone Miner. Res. 2016;31(2):347–357. doi: 10.1002/jbmr.2696. [DOI] [PubMed] [Google Scholar]
- Paschalis E.P., Gamsjaeger S., Fratzl-Zelman N., Roschger P., Masic A., Brozek W., Hassler N., Glorieux F.H., Rauch F., Klaushofer K., Fratzl P. Evidence for a role for Nanoporosity and Pyridinoline content in human mild Osteogenesis Imperfecta. J. Bone Miner. Res. 2016;31(5):1050–1059. doi: 10.1002/jbmr.2780. [DOI] [PubMed] [Google Scholar]
- Paschalis E.P., Gamsjaeger S., Hassler N., Klaushofer K., Burr D. Ovarian hormone depletion affects cortical bone quality differently on different skeletal envelopes. Bone. 2017;95:55–64. doi: 10.1016/j.bone.2016.10.029. [DOI] [PubMed] [Google Scholar]
- Paschalis E., Gamsjaeger S., Klaushofer K. Vibrational spectroscopic techniques to assess bone quality. Osteoporos. Int. 2017;28(8):2275–2291. doi: 10.1007/s00198-017-4019-y. [DOI] [PubMed] [Google Scholar]
- Paschalis E.P., Gamsjaeger S., Condon K., Klaushofer K., Burr D. Estrogen depletion alters mineralization regulation mechanisms in an ovariectomized monkey animal model. Bone. 2019;120:279–284. doi: 10.1016/j.bone.2018.11.004. [DOI] [PubMed] [Google Scholar]
- Penel G., Leroy G., Rey C., Bres E. MicroRaman spectral study of the PO4 and CO3 vibrational modes in synthetic and biological apatites. Calcif. Tissue Int. 1998;63(6):475–481. doi: 10.1007/s002239900561. [DOI] [PubMed] [Google Scholar]
- Robins S. Fibrillogenesis and maturation of collagens. In: Seibel M., Robins S., Bilezikian J., editors. Dynamics of Bone and Cartilage Metabolism. Academic Press; New York: 2006. pp. 41–53. [Google Scholar]
- Robins S.P. Biochemistry and functional significance of collagen cross-linking. Biochem. Soc. Trans. 2007;35(Pt 5):849–852. doi: 10.1042/BST0350849. [DOI] [PubMed] [Google Scholar]
- Rokidi S., Paschalis E.P., Klaushofer K., Vennin S., Desyatova A., Turner J.A., Watson P., Lappe J., Akhter M.P., Recker R.R. Organic matrix quality discriminates between age- and BMD-matched fracturing versus non-fracturing post-menopausal women: a pilot study. Bone. 2019;127:207–214. doi: 10.1016/j.bone.2019.06.017. [DOI] [PubMed] [Google Scholar]
- Shevde N.K., Bendixen A.C., Dienger K.M., Pike J.W. Estrogens suppress RANK ligand-induced osteoclast differentiation via a stromal cell independent mechanism involving c-Jun repression. Proc. Natl. Acad. Sci. U. S. A. 2000;97(14):7829–7834. doi: 10.1073/pnas.130200197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Slater M., Patava J., Mason R.S. Role of chondroitin sulfate glycosaminoglycans in mineralizing osteoblast-like cells: effects of hormonal manipulation. J. Bone Miner. Res. 1994;9(2):161–169. doi: 10.1002/jbmr.5650090205. [DOI] [PubMed] [Google Scholar]
- Smith A.J., Singhrao S.K., Newman G.R., Waddington R.J., Embery G. A biochemical and immuno-electron microscopical analysis of chondroitin sulphate-rich proteoglycans in human alveolar bone. Histochem. J. 1997;29(1):1–9. doi: 10.1023/a:1026406932452. [DOI] [PubMed] [Google Scholar]
- Stepan J.J., Hruskova H., Kverka M. Update on menopausal hormone therapy for fracture prevention. Curr. Osteoporos. Rep. 2019;17(6):465–473. doi: 10.1007/s11914-019-00549-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- SureCAL Automated Calibration for Raman Microscopy. https://www.bruker.com/products/infrared-near-infrared-and-raman-spectroscopy/raman/surecal.html. (Accessed on 1-Dec-2020).
- Terajima M., Perdivara I., Sricholpech M., Deguchi Y., Pleshko N., Tomer K.B., Yamauchi M. Glycosylation and cross-linking in bone type I collagen. J. Biol. Chem. 2014;289(33):22636–22647. doi: 10.1074/jbc.M113.528513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thompson W.R., Modla S., Grindel B.J., Czymmek K.J., Kirn-Safran C.B., Wang L., Duncan R.L., Farach-Carson M.C. Perlecan/Hspg2 deficiency alters the pericellular space of the lacunocanalicular system surrounding osteocytic processes in cortical bone. J. Bone Miner. Res. 2011;26(3):618–629. doi: 10.1002/jbmr.236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wassen M.H., Lammens J., Tekoppele J.M., Sakkers R.J., Liu Z., Verbout A.J., R.A. Bank Collagen structure regulates fibril mineralization in osteogenesis as revealed by cross-link patterns in calcifying callus. J. Bone Miner. Res. 2000;15(9):1776–1785. doi: 10.1359/jbmr.2000.15.9.1776. [DOI] [PubMed] [Google Scholar]
- Xu T., Bianco P., Fisher L.W., Longenecker G., Smith E., Goldstein S., Bonadio J., Boskey A., Heegaard A.M., Sommer B., Satomura K., Dominguez P., Zhao C., Kulkarni A.B., Robey P.G., Young M.F. Targeted disruption of the biglycan gene leads to an osteoporosis-like phenotype in mice. Nat. Genet. 1998;20(1):78–82. doi: 10.1038/1746. [DOI] [PubMed] [Google Scholar]