Abstract
Simple Summary
Antibiotic resistance is a growing threat; its indiscriminate use has led to management restrictions in humans and animals. Bacteriocins are powerful antimicrobial peptides that have great potential in the prevention and treatment of diseases in animals. Their antimicrobial activity is rapid, and they show a lower propensity to develop resistance than conventional antibiotics. Currently, their main application is in food preservation systems. However, several studies show their bioactive role as antimicrobials, probiotics, and immunomodulators in animals. Therefore, bacteriocins are an excellent alternative to be applied in several areas of veterinary medicine.
Abstract
In the search for an alternative treatment to reduce antimicrobial resistance, bacteriocins shine a light on reducing this problem in public and animal health. Bacteriocins are peptides synthesized by bacteria that can inhibit the growth of other bacteria and fungi, parasites, and viruses. Lactic acid bacteria (LAB) are a group of bacteria that produce bacteriocins; their mechanism of action can replace antibiotics and prevent bacterial resistance. In veterinary medicine, LAB and bacteriocins have been used as antimicrobials and probiotics. However, another critical role of bacteriocins is their immunomodulatory effect. This review shows the advances in applying bacteriocins in animal production and veterinary medicine, highlighting their biological roles.
Keywords: bacteriocins, antimicrobials, lactic acid bacteria, probiotics, immunomodulation, veterinary medicine
1. Introduction
Bacteriocins are heat-stable, ribosomally synthesized antimicrobial peptides. Both Gram-positive and Gram-negative bacteria, and archaea release antimicrobial peptides extracellularly in the late-exponential to the early-stationary growth phases [1]. An essential attribute of bacteriocins is the antimicrobial activity against different bacteria, fungi, parasites, viruses, and even against natural resistant structures, such as bacterial biofilms [2,3,4,5]. Lactic acid bacteria (LAB) are a heterogeneous group of Gram-positive bacteria. They are classified according to glucose fermentation characteristics, cell morphology, capacity to utilize sugars, and optimum growth temperature range [6]. Thus, this classification system recognized a core group consisting of four genera: Lactobacillus, Pediococcus, Leucononstoc, and Streptococcus [7]. Molecular biological methods have increased the number of genera, including the following: Aerococcus, Alloiococcus, Carnobacterium, Dolosigranulum, Enterococcus, Lactococcus, Lactosphaera, Melissococus, Oenococcus, Tetragenococcus, Vagococcus, and Weissella [6,8]. Various studies have shown that LAB inhibit pathogenic microorganisms growth, degrade mycotoxins, and have a probiotic effect [6]. LAB are found abundantly in nature and symbiotically interact with higher organisms. They have been isolated from several sources, including dairy products, meat, fruits, and vegetables. They can also be found in mucous membranes of the respiratory, intestinal, and other anatomical sites of man and animals, even in plants, wastewater, soil, and manure [9]. Bacteriocins have been used as food preservatives, due to their ability to inhibit microorganisms potentially harmful to human health. They are safe for consumption and do not alter the quality and safety of food [10,11]. Furthermore, bacteriocins from LAB have had a significant development in other fields, such as in the cosmetic industry and human and veterinary medicine [12,13]. In animal production, bacteriocin-producing bacteria have been used as probiotics in the diet or drinking water of pigs, poultry, and fish, which has increased their growth rate [14,15].
LAB are among the important groups of bacteria that provide health benefits for humans and animals. Therefore, in this review, we focus on showing the advances in applying LAB bacteriocins as antimicrobials, probiotics, and immunomodulators in animal production and veterinary medicine.
2. Bacteriocin Overview
Bacteriocins are a diverse group of antimicrobial cationic and hydrophobic peptides composed of 20–60 amino acids. The ribosomal machinery is responsible for the synthesis of bacteriocins. Several genes are implicated in modifying amino acids, the export and regulation of the bacteriocin, and self-immunity proteins [16,17,18]. Bacteriocin encoding genes are organized into operons located in the chromosome, plasmids, or other mobile genetic elements. In general, these operons are inducible and require secretion and extracellular accumulation of bacterial peptides for induction [19,20]. Bacteriocins are extracellularly released and can have bactericidal or bacteriostatic effects on species closely related to the producing strain or affect other genera, phylum, or even domain [21,22].
Furthermore, the influence of environmental factors promotes the secretion of bacteriocins, including bacterial cell density, nutrient availability, the presence of acetic acid, and signaling peptides (competence stimulating peptide molecules) [23]. Interestingly, bacteriocins have been reported to be 103–106 times more potent than various other antimicrobials, including conventional antibiotics [13]. Therefore, bacteriocins-producing bacteria synthesize self-immunity proteins that protect them from their bacteriocins by scavenging bacteriocins or antagonist competition for receptor bacteriocin [17,24,25]. An important advantage of bacteriocins is that they can have activity against pathogenic and opportunistic bacteria, including multidrug-resistant species, without discriminating between antibiotic-resistant and sensitive strains [26]. Several bacteriocins have been shown to act in synergy with conventional antibiotics, reducing concentrations, undesirable side-effects, and the prevalence of resistant strains [27].
Interestingly, the combination of bacteriocins and antibiotics has been proposed as novel therapeutic options for food-producing animals. The possibility of replacing the use of antibiotics is explored to avoid bacterial resistance. Various reports have also established LAB bacteriocins advantages and synergistic actions with other biomolecules, such as nisin and citric acid, against Staphylococcus aureus and Listeria monocytogenes [28]. It has also been documented that bacteria can develop resistance to bacteriocins. However, resistance to bacteriocins is minimal compared to conventional antibiotics. Since the frequency of spontaneous mutations in cells exposed to bacteriocins is low [28]. This resistance is generally through modifications in the cell envelope, such as alterations in the charge and thickness [20,29,30].
3. Classification of Bacteriocins
Numerous bacteriocins have been isolated from LAB and are described in several databases. They have different characteristics, structures, modes of action, biochemical properties, activity spectra, and target cell receptors [31,32]. Bacteriocins produced by Gram-positive bacteria have been classified into three groups according to their biochemical and genetic characteristics or the presence of disulfide or monosulfide bonds, molecular weight, thermal stability, proteolytic enzymatic stability, presence or absence of post-translational modification of amino acids, and antimicrobial action [33] (Table 1). Initially, a fourth class of bacteriocins was described; however, it has been aborted and renamed as bacteriolysins, which comprise large complexes with carbohydrate and lipid residues [34,35].
Table 1.
Classification of bacteriocins.
| Class of Bacteriocin | Subclasses | Molecular Properties | Reference |
|---|---|---|---|
| Class I Lantibiotic |
Ia Lanthipeptides Ib Globular and inflexible bacteriocins Ic Sactipeptides |
Small, heat-stable bacteriocins (<5 kDa), have a post-translational modification, resulting in the formation of atypical amino acids lanthionine and methyllanthionine. | [33,36,37,38,39] |
| Class II Non-lantibiotic |
IIa Pediocin-like IIb Two peptides IIc Leader less IId Non pediocin-like Single-peptide |
Small and flexible bacteriocins (<10 kDa), with an amphiphilic helical structure. These peptides do not contain modified amino acid residues and are pH and heat-resistant. | [13,40,41,42,43,44,45,46] |
| Class III | IIIa Bacteriolysins IIIb Nonlytic |
High molecular weight bacteriocins (>30 kDa), thermolabile and unmodified peptides. | [47,48] |
Class I bacteriocins (lantibiotics) are small peptides (<5 kDa), 19 to 50 amino acids, are heat-stable, and have a post-translational modification, resulting in the formation of atypical amino acids lanthionine and methyllanthionine. These modify and introduce intramolecular cyclic structures, providing rigidity and resistance to the action of proteases [36,37,38]. Further, class I is divided into three subclasses: Class Ia comprises flexible, elongated, positively charged, and hydrophobic peptides associated with a pore formation in bacterial membranes; the most representative bacteriocin of this group is nisin [33]. Class Ib is made up of globular and inflexible bacteriocins that are negatively charged or have no net charge. These peptides can inhibit catalytic enzymes essential for the survival of susceptible bacteria [39]. Class Ic (sactipeptides) are sulfur-to-α-carbon-containing peptides. No bacteriocin of this group from LAB has been characterized [38].
Class II (nonlantibiotic) bacteriocins are small and flexible (<10 kDa), with an amphiphilic helical structure. These peptides do not contain modified amino acid residues and are pH and heat-resistant. Class II bacteriocins are divided into four subclasses based on structure and modifications. Class IIa bacteriocins (pediocin-like) include peptides from 35 to 50 amino acids that contain the YGNGV consensus sequence at the N-terminus. These bacteriocins have potent activity against L. monocytogenes [13,40,41]. Class IIb (two-peptide unmodified bacteriocins) consists of two different complementary peptides. The optimal antimicrobial activity requires both peptides that are members of this group in about equal amounts [42]. Class IIc (circular bacteriocins) contains 35 to 70 amino acids. These bacteriocins are associated with a leader peptide sequence and include one to two cysteine residues in their structure; they are further divided into cystibiotics and thiolbiotics. These peptides are resistant to many proteolytic enzymes [43,44,45]. Class IId comprises linear, non-pediocin-like, single-peptide bacteriocins [46].
Class III bacteriocins have high molecular weight (>30 kDa) and are thermolabile and unmodified peptides. They have a bacteriolytic (class IIIa) or nonlytic mechanism of action (class IIIb). These bacteriocins have been poorly studied [47,48].
4. Mechanism of Action of Bacteriocins
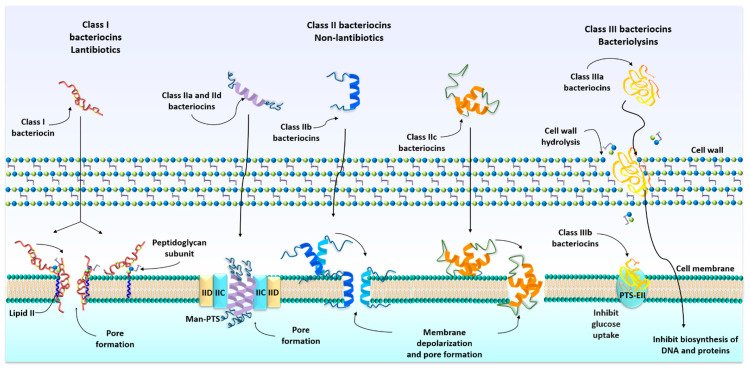
The mechanism of action of bacteriocins depends on their primary structure. Some can exert their activity on the cytoplasmic membrane releasing compounds vital of susceptible bacterial (cell lysis); others can enter the cytoplasm and affect gene expression and protein synthesis (Figure 1) [26].
Figure 1.
Mode action of bacteriocins. Bacteriocins act directly on the membrane or through a specific receptor on the target cell and form pores in the bacterial cell membrane, which leads to cell death.
Lantibiotics (class I) bacteriocins have a dual mode of action. They inhibit bacterial cell wall biosynthesis through binding to lipid II, a hydrophobic carrier of peptidoglycan monomers, from the cytoplasm to the cell wall, compromising cell viability. Additionally, lantibiotics can use lipid II as a docking molecule to initiate a process of membrane insertion and pore formation in the bacterial membrane [49,50,51].
Non-lantibiotics (class II), such as pediocin-like and the one-peptide nonpediocin-like bacteriocins (class IIa and class IId), bind to MptC and MptD subunits of the mannose phosphotransferase permease (Man-PTS). The insertion of these bacteriocins into the target cell membrane leads to an irreversible opening of an intrinsic channel, which leads to the diffusion of ions through the membrane, causing the death of the target cell [41,52,53]. Class IIb, two-peptide unmodified bacteriocins, permeabilize the membrane of sensitive bacteria and form pores. These pores show specificity for monovalent cations, such as Na+, K+, Li+, Cs+, and Rb+ (described in lactococin G) [41,54]. Circular bacteriocins (class IIc) have a positive net charge. These peptides interact directly with the negatively charged bacterial membrane without requiring any receptor molecules. Consequently, pores are formed in the cell membrane, causing ions efflux and the dissipation of the membrane potential, leading to cell death [55,56]. Bacteriolysins (class IIIa bacteriocins) catalyze cell wall hydrolysis, resulting in cell lysis [48,57]. Nonbacteriolytic bacteriocins (class IIIb) exert their action by disturbing the glucose uptake by cells, starving them, and disturbing the membrane potential. Another mechanism is inhibiting the biosynthesis of DNA and proteins of target bacteria [23,58,59].
5. The Antimicrobial Properties of Bacteriocins in Veterinary Medicine
LAB bacteriocins have great potential in veterinary medicine. Several in vitro and in vivo studies have evaluated their therapeutic use in small animals, livestock, and poultry (Table 2). This evidence demonstrates that bacteriocins can be applied as a substitute for antibiotics and represent an alternative as new antimicrobials that prevent bacterial resistance [22,60].
Table 2.
Bacteriocins produced by lactic acid bacteria inhibit bacteria of interest in veterinary medicine.
| Bacteriocin | LAB Producer of Bacteriocin | Susceptible Bacteria | Reference |
|---|---|---|---|
| Enterocin AS-48 | Enterococcus faecalis UGRA10 | Lactococcus garvieae | [100] |
| Enterocin M | Enterococcus faecium AL41 |
Campylobacter spp. Clostridium spp. |
[96] |
| Enterocin CLE34 | Enterococcus faecium CLE34 | Salmonella pullorum | [114] |
| Enterocin E-760 |
Enterococcus durans
Enterococcus faecium Enterococcus hirae |
Salmonella enterica serovar Enteritidis S. enterica serovar Choleraesuis S. enterica serovar Typhimurium S. enterica serovar Gallinarum Escherichia coli O157:H7 Yersinia enterocolitica Staphylococcus aureus Campylobacter jejuni |
[124] |
| Lacticin 3147 | Lactococcus lactis DPC3147. |
Streptococcus dysgalactiae,
Streptococcus agalactiae Staphylococcus aureus Streptococcus uberis |
[110] |
| Mycobacterium avium subsp. paratuberculosis | [108] | ||
| Macedocin ST91KM | Streptococcus gallolyticus subsp. macedonicus ST91KM |
Streptococcus agalactiae
Streptococcus dysgalactiae Streptococcus uberis Staphylococcus aureus |
[125] |
| Nisin A | Lactococcus lactic subsp. lactis |
Enterococcus faecalis ssp. liquefaciens
Streptococcus equinus Staphylococcus epidermidis Staphylococcus aureus Streptococcus uberis Streptococcus dysgalactiae Streptococcus agalactiae |
[77] |
|
Streptococcus suis
Mycobacterium avium subsp. paratuberculosis |
[85] [108] |
||
| Nisin A Nisin V |
L. lactis NZ9700 L. lactis NZ9800nisA:M21V |
Listeria monocytogenes | [87] |
| Nisin Z | L. lactis NIZO22186 |
Staphylococcus aureus
Streptococcus agalactiae |
[78] |
| Pediocin A | Pediococcus pentosaceus FBB61 | Clostridium perfringens | [119] |
Nisin is an antimicrobial cationic peptide produced by Lactococcus lactis subsp. lactis and Streptococcus species. Nisin was the first antimicrobial agent in reaching the category of food-safe additive in 1969. The use of nisin is, at present, licensed in more than 50 countries [61]. The United States granted the Generally Regarded as Safe (GRAS) designation by the FDA [50]. Researchers have shown that the antimicrobial action of nisin can extend to non-food-related bacteria. Thus, nisin has been included in clinical studies to prevent the formation of dentobacterial plaque and gingivitis in dogs, with an effect comparable to chlorhexidine [62,63]. An advantage of its use is the stability to remain active and express a synergistic action among a series of topical delivery systems: mouthwash, toothpaste, and guar-gum biogel [62,63]. In dogs, diverse oral bacteria species form a polymicrobial biofilm in the tooth surface, developing periodontal disease (PD) [64]. Enterococcus faecalis is the main bacteria in the oral cavity of dogs involved in PD, and it shows resistance to different antibiotics [65]. Further, E. faecalis leads to adherence and aggregation of other pathogen bacteria, all of them forming the biofilm of PD [66]. Pet animals have been considered reservoirs of this species of potentially pathogenic bacteria that is potentially harmful to high-risk individuals [67,68,69]. Nisin shows broad bactericidal action in vitro against reference strains of E. faecalis and other bacteria involved in canine PD. The combination of nisin–biogel inhibits and eradicates canine PD multispecies biofilms tested in a model of co-aggregate bacteria in vitro with E. faecalis, Neisseria zoodegmatis, Corynebacterium canis, Porphyromonas cangingivalis, and Peptostreptococcus canis [66]. Prolonged nisin use in oral canine cleaning does not have the negative effects that chlorhexidine exhibits, such as taste loss or pigmentation of the enamel [70,71,72]. Enterococcus faecium is another bacterium implicated in PD disease; it is considered a zoonotic opportunist, and it is also the most abundantly isolated from the feces of healthy dogs [73]. Paradoxically, some isolates of E. faecium produce bacteriocins like enterocin A, B, and P, which inhibit the growth of L. monocytogenes [74,75].
New alternatives in livestock are being sought to replace antibiotics and diminish bacteria resistant to them. Nisin has been used as a treatment in bovine mastitis caused by complex bacterial: Enterococcus spp., Staphylococcus spp., and Streptococcus spp. [76,77]. The main advantage is that nisin only remained in the milk for 12 h after its application in concentrations that did not generate any risk in consuming the product, aside from avoiding bacterial resistance [78,79]. Due to this, the FDA approved a nisin-based udder disinfectant [33]. In addition, nanoparticles to which nisin was included have shown a bactericidal effect against multidrug-resistant Staphylococcus spp. isolated from bovine mastitis and methicillin-resistant S. aureus [80,81]. Some species of S. aureus produce biofilm, a virulence factor that favors the establishment of infection in the udder and interferes with antibiotics [82]. A study demonstrated the antimicrobial activity of nisin on biofilm-producing S. aureus cultures. Furthermore, it penetrates the biofilm matrix, which leads to the detachment of the biofilm. Likewise, the combination of nisin and lysostaphin resulted in synergy against Staphylococcus spp. biofilm [83]. Nisin alters the biofilm’s architecture and composition, reducing the polysaccharides that constitute the biofilm of S. aureus and Staphylococcus epidermidis without affecting the integrity of the proteins. In addition, nisin reduced the extracellular DNA in S. aureus biofilm, but this effect was not observed in the S. epidermidis biofilm [84]. In pig production, there are opportunistic pathogens, such as Streptococcus and Escherichia coli species. Streptococcus suis serotype 2 is a zoonotic pathogenic that causes great economic losses in the pig industry. The co-administration of nisin with conventional antibiotics for treating swine Streptococcus infection can decrease antibiotic resistance. This bacteriocin elicits the bacterial membrane permeability to facilitate the antibiotic to reach the target [85]. In addition, nisin can cross the capsule of S. suis, degrade biofilm, and kill the bacteria. In mice infected by S. suis, nisin reduced bacterial load and avoided bacterial systemic dissemination [86]. Nisin V, a variant of nisin (bioengineering synthesized), showed enhanced potency against L. monocytogenes in vivo. Nisin V decreased the load of Listeria monocytogenes in the liver and spleen of mice experimentally infected [87]. Avirulent strains of S. suis serotype 2 (isolated from the tonsils of healthy pigs) produce bacteriocins suicin 90–1330, suicin 65, and suicin 3908. These antimicrobial peptides have an antibacterial effect on virulent S. suis, including bacteria resistant to erythromycin and tetracycline. Interestingly, structure amino acid sequences within these suicins have little similarity to each other (<25%) [88,89,90]. A recombinant suicin has shown inhibitory activity against Gram-positive strains [91]. Enterotoxigenic E. coli causes diarrhea in piglets. The use of colistin (polymyxin E) has led to the development of a resistant strain [92,93,94]. The combination of nisin or enterocin DD14 with colistin showed a synergistic effect against colistin-resistant E. coli strains isolated from pigs. This effect is because the loss of membrane stability induced by interacting colistin with LPS allows the entry of bacteriocins to damage the cell wall [95]. In horses, enterocin M in the diet reduced undesirable Gram-negative bacteria: coliforms, Campylobacter, and Clostridium spp. No physiological parameter was altered by the administration of enterocin M in horses [96]. Nisin added to water in weaned rabbits decreases harmful intestinal microbiota: Staphylococcus coagulase-positive, coliforms, Pseudomonas, and Clostridiae. Rabbits with nisin treatment increased their body weight average, and meat quality was not affected [97,98]. A partially purified bacteriocin PPB CCM7420, isolated from E. faecium, showed a significant reduction of parasite Eimeria spp. oocysts in rabbits [98].
Some types of nisin are effective against aquaculture pathogens. Araújo et al. (2015) demonstrated that the Nisin Z (produced by L. lactis subsp. cremoris) prevents lactococcosis in rainbow trout. Pediococcus acidilactici L-14 produces Pediocin PA-1 bacteriocin, which was shown to have antimicrobial activity against fish pathogens, such as Lactococcus garvieae, Streptococcus iniae, Carnobacterium maltaromaticum, and Aeromonas salmonicida [99]. Treatment with enterocin AS-48 in trout infected by L. garvieae, led to an outcome of a survival rate of 60%, compared to untreated fish that did not survive [100]. Other studies suggest that Lactobacillus pentosus HC-2 and E. faecium NRW-2 could be used in the shrimp diet, as they have antibacterial activity against Vibrio harveyi and Vibrio parahaemolyticus (ATCC 17802) [101]. The combination of bacteriocins from LAB and eukaryotic antimicrobial peptides (AMP) showed a synergistic activity and broadened the spectrum range. It has been shown that pediocin PA1, sakacin P, and curvasin A increased the bactericidal activity of pleurocidin and AMP of fish against E. coli. These bacteriocins also have high antimicrobial activity against Listeria ivanovii [102]. The use of nisin and OaBac5mini (ovine cathelicidin) increased the bactericidal activity against methicillin-resistant S. aureus 1056 [103].
The lantibiotic lacticin produced by L. lactis subsp. lactis DPC3147 (GRAS) is active against potential pathogens of veterinary importance, including methicillin-resistant S. aureus, Streptococcus dysgalactiae, Streptococcus uberis, vancomycin-resistant E. faecalis, Clostridium difficile, Mycobacterium avium subsp. paratuberculosis, L. monocytogenes, and others [104,105,106,107,108,109,110]. Some of the most promising research for the use of this lantibiotic is for the treatment of mastitis. Currently, the treatment of choice for bovine mastitis involves the use of commercial therapeutic antibiotic formulations. However, a recent study shows that L. lactis DPC3147 (which produces lacticin 3147) used to treat cows with clinical/subclinical mastitis showed efficacy comparable to that of antibiotic treatment (kanamycin and cephalexin) [109]. Due to the excellent antimicrobial activity exhibited by lacticin 3147, the application of a teat seal that contains a combination of bacteriocin and bismuth to prevent S. dysgalactiae infection in dry cows and S. aureus infection in lactating cows has been proposed [110,111]. The lacticin NK34 is a variant that adds to mastitis control by bacteriocins. It has shown in vitro high antimicrobial activity against S. aureus and coagulase-negative Staphylococcus strains isolated from bovine mastitis. In an experimental infection in mice with S. aureus, antimicrobial activity of lacticin NK34 ensures survival above 80% [112].
The antimicrobial property of bacteriocins has been exploited to control the pathogenic microbiota in poultry. Plantaricin (isolated from Lactobacillus plantarum F1) was proposed as a viable alternative to replacing the use of antibiotics against colibacillosis in broiler chickens [113]. Plantaricin CLP29 and enterocin CLE34, partially purified, have wide antibacterial activity against Salmonella pullorum and E. coli [114]. The discovery of new bacteriocins for application in poultry has been made in the microbiota of the domestic broiler chicken’s gastrointestinal tract. The bacterium Paenibacillus polymyxa NRRL B-30509 was isolated from domestic Russian broiler chickens, producing the bacteriocin paenicidin A, which has activity against Campylobacter jejuni [115]. Another bacteriocin with potential use in poultry is pediocin A (isolated from Pediococcus pentaceus FBB61) [116]. The antimicrobial activity has been reported against Gram-positive bacteria, such as L. monocytogenes and Clostridium perfringens type A [117]. In broilers infected by C. perfringens type A (producer of NetB toxin) involved in necrotic enteritis, Pediocin A was administered in food. The treatment improved the growth performance of the chickens. However, it did not decrease the bacterial load [118]. Microencapsulation of bacteriocin could prevent the inactivation of bacteriocin, due to digestive processes in broilers [119]. The combination of bacteriocins divercin AS7 and nisin as an additive in the diet of broilers has been shown to improve body weight gain [120,121]. Divercin AS7 and nisin showed bactericidal activity similar to salinomycin (ionophore coccidiostat) [121]. Nisin has antimicrobial action on microbiota related to the detriment of productivity in broiler chickens, similar to monensin ionophore. Nisin supplementation positively affected the gut microbiota by reducing potentially pathogenic bacterial populations in the jejunum and ceca, such as Enterobacteriaceae, C. perfringens, Clostridium coccoides–Eubacterium rectale cluster, Bacteroides–Prevotella cluster, Lactobacillus sp./Enterococcus sp., and the Clostridium leptum subgroup [122]. The diminished load of bacteria associated with low productivity in the gastrointestinal tract decreases the competition of nutrients and improves energy use in chickens [123]. These findings highlight the role of bacteriocins as an excellent antibacterial alternative against potentially pathogenic agents for animals and improved growth performance.
6. The Probiotic Activity of LAB Bacteriocins
Oral administration of purified or semipurified bacteriocins has been shown to have limitations. Digestive enzymes can degrade bacteriocins, and bacteriocins can adhere to food particles or diffuse through digestion, among others [126]. The protection of bacteriocins in capsules or nanocapsules can be an alternative to prevent enzymatic degradation and avoid various doses and high concentrations of bacteriocins [127]. Thus, the most efficient method for taking advantage of bacteriocins in the digestive tract includes bacteriocin-producing LAB as probiotics. This strategy favors the colonization of bacteria in the gastrointestinal tract, and bacteriocins can be produced in situ [128]. There are multiple benefits of using LAB in place of antibiotics or growth promoters. These include modulation of microbiota, improving the intestinal barrier function and digestion, preventing the colonization of enteric pathogens, and stimulating the immune system [15,129,130,131]. However, studies showed that E. faecium LMG 30881, a producer of enterocin B in the canine diet, caused unfavorable effects, such as runny stools, higher Gram-negative bacterial counts, and lower hemoglobin concentrations [132,133]. The use of bacteriocin-producing LAB probiotic in dogs requires more research, since no further work has been generated in this regard to date. In healthy pigs, it has been shown that some LAB of the gastrointestinal tract prevents villous atrophy of the post-weaning stage, promotes the maturation of gastrointestinal lymphoid tissue, and has immunomodulatory activity. Oral administration of the probiotic Lactobacillus salivarius B1 (isolated from healthy piglets) in newborn piglets showed that the probiotic bacteria colonized the duodenal mucosa and increased the height of the villi, which improved absorption and promoted the integrity of the intestinal barrier. Interestingly, L. salivarius increased the expression of porcine beta-defensin 2 (pBD-2), an antimicrobial peptide produced by host cells. Continuous probiotic administration caused a considerable increase in the production of pBD-2, which could even be detected in the saliva of piglets [134]. In the duodenum and ileum, the number of intraepithelial lymphocytes, plasma cells that produce IgA, and the synthesis of Toll-Like Receptor-2 (TLR-2) increased. In the ileum, interleukin-6 (IL-6), a cytokine that promotes the differentiation and proliferation of B lymphocytes, was increased [130]. These studies suggest that the immunomodulatory effects of L. salivarius B1 are due to bacteriocins [135]. L. salivarius UCC118 (isolated from the human intestinal microbiota) produces the bacteriocin Abp118. This antimicrobial peptide has been shown to have activity against L. monocytogenes [136,137]. L. salivarius UCC 118, as a probiotic added to the diet of pigs after weaning, showed that LAB colonized the ileum and caused a significant decrease in spirochetes (Treponema), considered to be opportunistic pathogens of pigs [138]. Rustic or native animals on farms can be a natural source of bacteriocins-producing probiotics. A study has shown that miniature piglets from Congjiang (a breed of pig native to China) had higher resistance to stress-induced diarrhea during early weaning due to gut microbiota. In the feces of miniature piglets, a higher population of Lactobacillus gasseri LA39 and Lactobacillus frumenti, producers of the bacteriocin gassericin, was found. This study also demonstrated an increase in the signaling pathway involved in protein expression (NHE3, SLC5A1, DRA, and PAT1) associated with intestinal absorption. Gassericin decreased the expression of proteins related to intestinal secretion (NKCC1, CFTR, CaCC1). These bacteria could be transplanted into commercial crossbred piglets before weaning and prevent diarrhea after weaning. This study provides a strategy for the possible prevention of diarrhea in pigs and even other mammals [139]. There are reports of more than 30 LAB as probiotics that inhibit the growth of pathogenic microorganisms in birds; however, their mode of action remain poorly understood [140,141,142]. LAB as probiotics in poultry has been used to control experimental coccidial infection, endemic in the commercial broiler industry. Studies on the inclusion of multispecies probiotics (E. faecium, Bifidobacterium animalis, and L. salivarius) in food or water have shown that the colonization of LAB in the intestine causes a coccidiostatic effect on Eimeria spp. In addition, the probiotic prevented intestinal damage without affecting body weight gain values. On the other hand, there were high probiotic LAB amounts in intestinal microbiota, while coliform and C. perfringens were lower than the control group [143]. The multispecies probiotic for commercial use can be integrated into chicken microbiota early in ovo and for one day of hatching. These bacteria increased protection against Eimeria spp. and commercial vaccine-administered response, at the same time [144,145]. The species of genera Enterococcus spp., as a probiotic for use in the poultry industry, has been shown to produce enterocin A, B, P, and L50 and bacteriocin-like inhibitory substances that are not identified currently. These bacteriocins have demonstrated antimicrobial activity in vitro against pathogenic bacterial C. perfringens, S. aureus, Salmonella Heidelberg, and L. monocytogenes [146,147].
7. Bacteriocins as Immunomodulators
The immunomodulatory effect of bacteriocins has not been fully elucidated. It is known that modulation of the immune system depends on the concentration of bacteriocin used. These activation mechanisms of the immune response by bacteriocins add to the bactericidal effect, thereby increasing host protection, particularly during infections. The lantibiotic nisin is the oldest and most widely used bacteriocin in the food industry [148]. Nisin has shown that its administration in the diet for short periods increases CD4+ and CD8+ T lymphocytes (LT) and reduces the lymphocytes B (LB) levels in the blood. Its consumption in prolonged period results in return to normal LT levels, the maintained decrease in LB, and the count increase of macrophages/monocytes [149]. The high concentration of nisin added to porcine PBMC (Peripheral Blood Mononuclear Cells) in vitro equally increased CD4+ and CD8+ proliferation and cytokine IL-1β and IL-6 production [150]. In rabbit’s vaginal tissue explant culture, nisin showed high biological compatibility with tissue and did not show any immunomodulatory effect. The lacticin did not affect the expression of defensin, TLR3, or TLR9 receptors, nor the expression of cytokines IL-4, IL-6, GM-CSF, IL-8, IL-6, or TNF-α [151,152]. Moreover, in neutrophils, high concentrations of nisin activate extracellular traps (NETs) and increase intracellular superoxide levels [153]. Interestingly, although the antimicrobial activity of nisin in vitro is limited to Gram-positive bacteria, when administered to animals infected by Gram-positive and Gram-negative bacteria, the host’s bacterial load significantly decreases. In these cases, experiments with human PBMC explain that the protection of nisin is due to chemokines (MCP-1, IL-8, and Gro-α) that represses proinflammatory TNF-α production. In this experiment, nisin shows greater potency than human antimicrobial peptide LL-37 [154]. Nisin modulates some nonspecific immune functions in turbot. The treatment with intermediate and lower doses of nisin in turbot head kidney macrophages increased oxygen free radical production and phagocytic function and did not affect nitric oxide production. The lowest doses of nisin injection in turbot augmented lysozyme concentration in serum [155].
The immunomodulatory effect of nisin is expressed even in nonimmune cells. In bovine mammary epithelial cells, nisin increases intracellular lysozyme and even releases it to the extracellular environment [156]. Bacteriocins as immune-modulating agents have shown anti-inflammatory properties in damaged or infected tissue. In LPS-stimulated PBMC, nisin inhibits the synthesis of TNF-α, thereby decreasing the inflammatory response [154]. In porcine PBMC infected by E. coli, nisin decreased the inflammatory response, mainly the production of IL-6 [150]. This anti-inflammatory effect is reproduced in bovine mammary gland epithelial cells by promoting a negative regulation in the production of TNF-α, which benefits the recovery of intramammary tissue. Endometritis represents a frequent health problem in the first three weeks postpartum. In bovines, nisin prevents endometritis induced by experimental infection with S. aureus. Bacteriocins in this infection promote a decrease in the proinflammatory cytokines and increase the anti-inflammatory cytokines [157]. In tissue injuries, such as fractures, the bacteriocins produced by Lactobacillus rhamnosus L34 and L. rhamnosus (ATCC 53103) were shown to reduce postoperative effects, such as inflammation. Additionally, they favor the control of experimental intra-articular infection with S. aureus [158,159]. Bacteriocins from L. rhamnosus caused a decrease in proinflammatory cytokines TNF-α, IL-6, and C-reactive protein in rabbit models of mandible fracture fixation and knee re-placement surgery. In addition, they controlled experimental S. aureus infection by minimizing biofilm formation and promoting tissue repair [158,159]. These results suggest that bacteriocins could be potent agents for preventing postoperative orthopedic infections. A recent study demonstrated that the LAB Pediococcus pentosaceus (SL001) express the bacteriocin coagulin. A supplemented diet with P. pentosaceus enhanced the immunity of grass carp, increasing IgM and C3 (complement 3), whereas IL-8 was downregulated. Moreover, P. pentosaceus contributed to the elimination of pathogens and promoted grass carp growth rate [160].
Interestingly, the bacteriocins produced by pathogens do not have a bactericidal effect. These bacteriocins seriously affect the host’s immune system, favoring disease. Streptococcus iniae is a pathogen that affects fish and humans and produces the bacteriocin Sil. Sil has only shown bacteriostatic activity against Bacillus subtilis and is not cytotoxic in healthy fish tissues. However, Sil bacteriocin administration to fish before S. iniae infection reduced respiratory burst and acid phosphatase activity in turbot head kidney monocytes. Furthermore, the bacterial infection spread to the kidney and spleen [161].
8. Conclusions
Bacteriocins are a powerful weapon that can be exploited in veterinary medicine. The administration of these antimicrobial peptides in domestic animals eliminates potentially pathogenic undesirable microorganisms without causing cytotoxicity on cells or tissues. Interestingly, it does not generate resistance to antibiotics, and resistance to bacteriocins is minimal. Bacteriocins are analogous and synergistic when combined with antiseptics, antibiotics, and ionophores, showing greater potency than antimicrobial peptides from eukaryotic cells. In addition, these combinations can reduce resistance to bacteriocins. The potential use of bacteriocins alone or along with microbicidal agents has potential therapeutic actions in periodontal disease and mastitis in dairy cows, prevents postoperative infections in fractures, and coccidiostats and improves productive parameters in substitution of antibiotics as growth promoters. In animal nutrition, bacteriocins reduce cholesterol and triglycerides, thus improving the quality of the meat as a final product. LAB colonize the intestinal mucosa and produce bacteriocins in situ. LAB and their bacteriocins promote the integrity of the intestinal barrier, eliminate bacteria that interfere with the use of nutrients, and stimulate the expression of proteins associated with the absorption of intestinal fluid. This finding suggests that they can be used as probiotics in poultry and monogastric animals. Furthermore, these bioactive peptides have a role in the immune response as immunomodulators. Bacteriocins modulate the expression of anti-inflammatory cytokines to promote the repair of cell damage. Some of them are potent inducers of antimicrobial peptides in eukaryotic cells, which improve the innate immune response against pathogens. However, the bacteriocins produced by virulent bacteria can be a virulence factor, promoting a proinflammatory cytokine profile, which favors infection in lymphoid cells and organs. Little is known about the immunomodulatory effects of bacteriocins in animals. More studies are required to fully understand the role of bacteriocins on the innate and adaptive response that could contribute to the control or resolution of infections or diseases. LAB bacteriocins are projected as new antimicrobials that could be targeted or stabilized by nanotechnology and prevent enzymatic digestion. In addition, more research is required on modifications that could increase the potency of bacteriocins. The benefits of bacteriocins shown in vitro and in vivo assays provide support for developing and researching clinical trials in the different areas of veterinary medical therapeutics.
Acknowledgments
The authors kindly acknowledge the financial support of the PRODEP and CONACyT.
Author Contributions
J.C.H.-G. and N.S.C.-J. are responsible for the conceptualization, investigation, analysis, writing, and editing of this review. A.M.-T. and G.L.-H. participated in the research of published literature associated with veterinary concepts. B.E.G.-P. participated in the editing and analysis of this work. The manuscript was edited through the contribution of all authors. All authors have read and agreed to the published version of the manuscript.
Funding
The authors kindly acknowledge the financial support of the PRODEP (Programa para el Desarrollo Profesional Docente en Educación Superior, Convenio 511-6/19-8268; No., de Proyecto:2019-0046), México. Universidad Autónoma del Estado de Hidalgo and Consejo Nacional de Ciencia y Tecnología (CONACyT).
Conflicts of Interest
The authors declare no conflict of interest.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Zheng J., Gänzle M.G., Lin X.B., Ruan L., Sun M. Diversity and Dynamics of Bacteriocins from Human Microbiome. Environ. Microbiol. 2014;17:2133–2143. doi: 10.1111/1462-2920.12662. [DOI] [PubMed] [Google Scholar]
- 2.Torres N.I., Noll K.S., Xu S., Li J., Huang Q., Sinko P.J., Wachsman M.B., Chikindas M.L. Safety, Formulation and In Vitro Antiviral Activity of the Antimicrobial Peptide Subtilosin Against Herpes Simplex Virus Type 1. Probiotics Antimicrob. Proteins. 2013;5:26–35. doi: 10.1007/s12602-012-9123-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Eyang S.-C., Elin C.-H., Sung C.T., Efang J.-Y. Antibacterial Activities of Bacteriocins: Application in Foods and Pharmaceuticals. Front. Microbiol. 2014;5:241. doi: 10.3389/fmicb.2014.00241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Graham C.E., Cruz M.R., Garsin D.A., Lorenz M.C. Enterococcus Faecalisbacteriocin EntV Inhibits Hyphal Morphogenesis, Biofilm Formation, and Virulence of Candida Albicans. Proc. Natl. Acad. Sci. USA. 2017;114:4507–4512. doi: 10.1073/pnas.1620432114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Martín-Escolano R., Cebrián R., Martín-Escolano J., Rosales M.J., Maqueda M., Sánchez-Moreno M., Marín C. Insights into Chagas Treatment Based on the Potential of Bacteriocin AS-48. Int. J. Parasitol. Drugs Drug Resist. 2019;10:1–8. doi: 10.1016/j.ijpddr.2019.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Mokoena M.P. Lactic Acid Bacteria and Their Bacteriocins: Classification, Biosynthesis and Applications against Uropathogens: A Mini-Review. Molecules. 2017;22:1255. doi: 10.3390/molecules22081255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lahtinen S., Ouwehand A.C., Salminen S., Wright A. Von Lactic Acid Bacteria: Microbiological and Functional Aspects. 4th ed. CRC Press; Boca Raton, FL, USA: 2011. [Google Scholar]
- 8.Agriopoulou S., Stamatelopoulou E., Sachadyn-Król M., Varzakas T. Lactic Acid Bacteria as Antibacterial Agents to Extend the Shelf Life of Fresh and Minimally Processed Fruits and Vegetables: Quality and Safety Aspects. Microorganisms. 2020;8:952. doi: 10.3390/microorganisms8060952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Liu W., Pang H., Zhang H., Cai Y. Biodiversity of Lactic Acid Bacteria. In: J.B. Metzler, editor. Lactic Acid Bacteria. Springer; Berlin/Heidelberg, Germany: 2014. pp. 103–203. [Google Scholar]
- 10.Kheadr E., Zihler A., Dabour N., Lacroix C., Le Blay G., Fliss I. Study of the Physicochemical and Biological Stability of Pediocin PA-1 in the Upper Gastrointestinal Tract Conditions Using a Dynamicin Vitromodel. J. Appl. Microbiol. 2010;109:54–64. doi: 10.1111/j.1365-2672.2009.04644.x. [DOI] [PubMed] [Google Scholar]
- 11.Silva C.C.G., Silva S.P.M., Ribeiro S.C. Application of Bacteriocins and Protective Cultures in Dairy Food Preservation. Front. Microbiol. 2018;9:594. doi: 10.3389/fmicb.2018.00594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Maurício E., Rosado C., Duarte M.P., Verissimo J., Bom S., Vasconcelos L. Efficiency of Nisin as Preservative in Cosmetics and Topical Products. Cosmetics. 2017;4:41. doi: 10.3390/cosmetics4040041. [DOI] [Google Scholar]
- 13.Bédard F., Biron E. Recent Progress in the Chemical Synthesis of Class II and S-Glycosylated Bacteriocins. Front. Microbiol. 2018;9:1048. doi: 10.3389/fmicb.2018.01048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Dowarah R., Verma A., Agarwal N. The Use of Lactobacillus as an Alternative of Antibiotic Growth Promoters in Pigs: A Review. Anim. Nutr. 2017;3:1–6. doi: 10.1016/j.aninu.2016.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Vieco-Saiz N., Belguesmia Y., Raspoet R., Auclair E., Gancel F., Kempf I., Drider D. Benefits and Inputs from Lactic Acid Bacteria and Their Bacteriocins as Alternatives to Antibiotic Growth Promoters During Food-Animal Production. Front. Microbiol. 2019;10:57. doi: 10.3389/fmicb.2019.00057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Riley M.A., Wertz J.E. Bacteriocins: Evolution, Ecology, and Application. Annu. Rev. Microbiol. 2002;56:117–137. doi: 10.1146/annurev.micro.56.012302.161024. [DOI] [PubMed] [Google Scholar]
- 17.Ben Lagha A., Haas B., Gottschalk M., Grenier D. Antimicrobial Potential of Bacteriocins in Poultry and Swine Production. Veter. Res. 2017;48:1–12. doi: 10.1186/s13567-017-0425-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Noda M., Miyauchi R., Danshiitsoodol N., Matoba Y., Kumagai T., Sugiyama M. Expression of Genes Involved in Bacteriocin Production and Self-Resistance inLactobacillus brevis174A Is Mediated by Two Regulatory Proteins. Appl. Environ. Microbiol. 2018;84:e02707-17. doi: 10.1128/AEM.02707-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Zimina M., Babich O., Prosekov A., Sukhikh S., Ivanova S., Shevchenko M., Noskova S. Overview of Global Trends in Classification, Methods of Preparation and Application of Bacteriocins. Antibiotics. 2020;9:553. doi: 10.3390/antibiotics9090553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kumariya R., Garsa A.K., Rajput Y., Sood S., Akhtar N., Patel S. Bacteriocins: Classification, Synthesis, Mechanism of Action and Resistance Development in Food Spoilage Causing Bacteria. Microb. Pathog. 2019;128:171–177. doi: 10.1016/j.micpath.2019.01.002. [DOI] [PubMed] [Google Scholar]
- 21.Bédard F., Hammami R., Zirah S., Rebuffat S., Fliss I., Biron E. Synthesis, Antimicrobial Activity and Conformational Analysis of the Class IIa Bacteriocin Pediocin PA-1 and Analogs Thereof. Sci. Rep. 2018;8:1–13. doi: 10.1038/s41598-018-27225-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Schofs L., Sparo M.D., Bruni S.F.S. Gram-Positive Bacteriocins: Usage as Antimicrobial Agents in Veterinary Medicine. Veter. Res. Commun. 2020;44:1–12. doi: 10.1007/s11259-020-09776-x. [DOI] [PubMed] [Google Scholar]
- 23.Meade E., Slattery M.A., Garvey M. Bacteriocins, Potent Antimicrobial Peptides and the Fight against Multi Drug Resistant Species: Resistance Is Futile? Antibiotics. 2020;9:32. doi: 10.3390/antibiotics9010032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Siegers K., Entian K.D. Genes Involved in Immunity to the Lantibiotic Nisin Produced by Lactococcus Lactis 6F3. Appl. Environ. Microbiol. 1995;61:1082–1089. doi: 10.1128/AEM.61.3.1082-1089.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Alkhatib Z., Abts A., Mavaro A., Schmitt L., Smits S.H. Lantibiotics: How Do Producers Become Self-Protected? J. Biotechnol. 2012;159:145–154. doi: 10.1016/j.jbiotec.2012.01.032. [DOI] [PubMed] [Google Scholar]
- 26.Cotter P.D., Ross R.P., Hill C. Bacteriocins—A Viable Alternative to Antibiotics? Nat. Rev. Genet. 2013;11:95–105. doi: 10.1038/nrmicro2937. [DOI] [PubMed] [Google Scholar]
- 27.Cavera V.L., Arthur T.D., Kashtanov D., Chikindas M.L. Bacteriocins and Their Position in the Next Wave of Conventional Antibiotics. Int. J. Antimicrob. Agents. 2015;46:494–501. doi: 10.1016/j.ijantimicag.2015.07.011. [DOI] [PubMed] [Google Scholar]
- 28.Bacteriocins as a New Generation of Antimicrobials: Toxicity Aspects and Regulations|FEMS Microbiology Reviews, Oxford Academic. [(accessed on 18 March 2021)]; doi: 10.1093/femsre/fuaa039. Available online: https://academic.oup.com/femsre/article/45/1/fuaa039/5900525. [DOI] [PMC free article] [PubMed]
- 29.Sakayori Y., Muramatsu M., Hanada S., Kamagata Y., Kawamoto S., Shima J. Characterization of Enterococcus Faecium Mutants Resistant to Mundticin KS, a Class IIa Bacteriocin. Microbiology. 2003;149:2901–2908. doi: 10.1099/mic.0.26435-0. [DOI] [PubMed] [Google Scholar]
- 30.McBride S.M., Sonenshein A.L. The dlt Operon Confers Resistance to Cationic Antimicrobial Peptides in Clostridium Difficile. Microbiology. 2011;157:1457–1465. doi: 10.1099/mic.0.045997-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hammami R., Zouhir A., Ben Hamida J., Fliss I. Bactibase: A New Web-Accessible Database for Bacteriocin Characterization. BMC Microbiol. 2007;7:89. doi: 10.1186/1471-2180-7-89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Al Kassaa I., Rafei R., Moukhtar M., Zaylaa M., Gharsallaoui A., Asehraou A., El Omari K., Shahin A., Hamze M., Chihib N.-E. LA Biocin Database: A New Database Designed Specifically for Lactic Acid Bacteria Bacteriocins. Int. J. Antimicrob. Agents. 2019;54:771–779. doi: 10.1016/j.ijantimicag.2019.07.012. [DOI] [PubMed] [Google Scholar]
- 33.Ahmad V., Khan M.S., Jamal Q.M.S., Alzohairy M.A., Al Karaawi M.A., Siddiqui M.U. Antimicrobial Potential of Bacteriocins: In Therapy, Agriculture and Food Preservation. Int. J. Antimicrob. Agents. 2017;49:1–11. doi: 10.1016/j.ijantimicag.2016.08.016. [DOI] [PubMed] [Google Scholar]
- 34.Klaenhammer T.R. Genetics of Bacteriocins Produced by Lactic Acid Bacteria. FEMS Microbiol. Rev. 1993;12:39–85. doi: 10.1016/0168-6445(93)90057-G. [DOI] [PubMed] [Google Scholar]
- 35.Güllüce M., Karadayı M., Bariş Ö. Bacteriocins: Promising Natural Antimicrobials. In: Mendes-Vilas A., editor. Science, Technology and Education. Formatex; Madrid, Spain: 2013. pp. 1016–1027. [Google Scholar]
- 36.Guder A., Wiedemann I., Sahl H.G. Posttranslationally Modified Bacteriocins—The Lantibiotics. Biopolymers. 2000;55:62–73. doi: 10.1002/1097-0282(2000)55:1<62::AID-BIP60>3.0.CO;2-Y. [DOI] [PubMed] [Google Scholar]
- 37.Bierbaum G., Götz F., Peschel A., Kupke T., Van De Kamp M., Sahl H.-G. The Biosynthesis of the Lantibiotics Epidermin, Gallidermin, Pep5 and Epilancin K7. Antonie Leeuwenhoek. 1996;69:119–127. doi: 10.1007/BF00399417. [DOI] [PubMed] [Google Scholar]
- 38.Alvarez-Sieiro P., Montalbán-López M., Mu D., Kuipers O.P. Bacteriocins of Lactic Acid Bacteria: Extending the Family. Appl. Microbiol. Biotechnol. 2016;100:2939–2951. doi: 10.1007/s00253-016-7343-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Deegan L.H., Cotter P.D., Hill C., Ross P. Bacteriocins: Biological Tools for Bio-Preservation and Shelf-Life Extension. Int. Dairy, J. 2006;16:1058–1071. doi: 10.1016/j.idairyj.2005.10.026. [DOI] [Google Scholar]
- 40.Fimland G., Johnsen L., Dalhus B., Nissen-Meyer J. Pediocin-like Antimicrobial Peptides (Class IIa Bacteriocins) and Their Immunity Proteins: Biosynthesis, Structure, and Mode of Action. J. Pept. Sci. 2005;11:688–696. doi: 10.1002/psc.699. [DOI] [PubMed] [Google Scholar]
- 41.Nissen-Meyer J., Oppegård C., Rogne P., Haugen H.S., Kristiansen P.E. Structure and Mode-of-Action of the Two-Peptide (Class-IIb) Bacteriocins. Probiotics Antimicrob. Proteins. 2009;2:52–60. doi: 10.1007/s12602-009-9021-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Oppegård C., Rogne P., Emanuelsen L., Kristiansen P.E., Fimland G., Nissen-Meyer J. The Two-Peptide Class II Bacteriocins: Structure, Production, and Mode of Action. J. Mol. Microbiol. Biotechnol. 2007;13:210–219. doi: 10.1159/000104750. [DOI] [PubMed] [Google Scholar]
- 43.Sawa N., Zendo T., Kiyofuji J., Fujita K., Himeno K., Nakayama J., Sonomoto K. Identification and Characterization of Lactocyclicin Q, a Novel Cyclic Bacteriocin Produced by Lactococcus sp. Strain QU 12. Appl. Environ. Microbiol. 2009;75:1552–1558. doi: 10.1128/AEM.02299-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Borrero J., Brede D.A., Skaugen M., Diep D.B., Herranz C., Nes I.F., Cintas L.M., Hernández P.E. Characterization of Garvicin ML, a Novel Circular Bacteriocin Produced by Lactococcus garvieaeDCC43, Isolated from Mallard Ducks (Anas platyrhynchos) Appl. Environ. Microbiol. 2010;77:369–373. doi: 10.1128/AEM.01173-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Gabrielsen C., Brede D.A., Nes I.F., Diep D.B. Circular Bacteriocins: Biosynthesis and Mode of Action. Appl. Environ. Microbiol. 2014;80:6854–6862. doi: 10.1128/AEM.02284-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Iwatani S., Zendo T., Sonomoto K. Class IId or Linear and Non-Pediocin-Like Bacteriocins. Prokaryotic Antimicrob. Peptides. 2011:237–252. doi: 10.1007/978-1-4419-7692-5_13. [DOI] [Google Scholar]
- 47.Nilsen T., Nes I.F., Holo H. Enterolysin A, a Cell Wall-Degrading Bacteriocin from Enterococcus faecalis LMG 2333. Appl. Environ. Microbiol. 2003;69:2975–2984. doi: 10.1128/AEM.69.5.2975-2984.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Sun Z., Wang X., Zhang X., Wu H., Zou Y., Li P., Sun C., Xu W., Liu F., Wang D. Class III bacteriocin Helveticin-M Causes Sublethal Damage on Target Cells through Impairment of Cell Wall and Membrane. J. Ind. Microbiol. Biotechnol. 2018;45:213–227. doi: 10.1007/s10295-018-2008-6. [DOI] [PubMed] [Google Scholar]
- 49.Bauer R., Dicks L. Mode of Action of Lipid II-Targeting Lantibiotics. Int. J. Food Microbiol. 2005;101:201–216. doi: 10.1016/j.ijfoodmicro.2004.11.007. [DOI] [PubMed] [Google Scholar]
- 50.Cotter P.D., Hill C., Ross R.P. Bacteriocins: Developing Innate Immunity for Food. Nat. Rev. Genet. 2005;3:777–788. doi: 10.1038/nrmicro1273. [DOI] [PubMed] [Google Scholar]
- 51.Paiva A.D., Breukink E., Mantovani H.C. Role of Lipid II and Membrane Thickness in the Mechanism of Action of the Lantibiotic Bovicin HC5. Antimicrob. Agents Chemother. 2011;55:5284–5293. doi: 10.1128/AAC.00638-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Diep D.B., Skaugen M., Salehian Z., Holo H., Nes I.F. Common Mechanisms of Target Cell Recognition and Immunity for Class II Bacteriocins. Proc. Natl. Acad. Sci. USA. 2007;104:2384–2389. doi: 10.1073/pnas.0608775104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Nes I.F., Brede D.A., Diep D.B. Chapter 16—Class II Non-Lantibiotic Bacteriocins. In: Kastin A.J., editor. Handbook of Biologically Active Peptides. 2nd ed. Academic Press; Boston, MA, USA: 2013. pp. 85–92. [Google Scholar]
- 54.Moll G., Hildeng-Hauge H., Nissen-Meyer J., Nes I.F., Konings W.N., Driessen A.J.M. Mechanistic Properties of the Two-Component Bacteriocin lactococcin. G. J. Bacteriol. 1998;180:96–99. doi: 10.1128/JB.180.1.96-99.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Van Belkum M.J., Martin-Visscher L.A., Vederas J.C. Structure and Genetics of Circular Bacteriocins. Trends Microbiol. 2011;19:411–418. doi: 10.1016/j.tim.2011.04.004. [DOI] [PubMed] [Google Scholar]
- 56.Perez R.H., Zendo T., Sonomoto K. Circular and Leaderless Bacteriocins: Biosynthesis, Mode of Action, Applications, and Prospects. Front. Microbiol. 2018;9:2085. doi: 10.3389/fmicb.2018.02085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Simmonds R.S., Pearson L., Kennedy R.C., Tagg J.R. Mode of Action of a Lysostaphin-like Bacteriolytic Agent Produced by Streptococcus zooepidemicus 4881. Appl. Environ. Microbiol. 1996;62:4536–4541. doi: 10.1128/AEM.62.12.4536-4541.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Müller E., Radler F. Caseicin, a Bacteriocin from Lactobacillus casei. Folia Microbiol. 1993;38:441–446. doi: 10.1007/BF02814392. [DOI] [PubMed] [Google Scholar]
- 59.Swe P.M., Cook G.M., Tagg J.R., Jack R.W. Mode of Action of Dysgalacticin: A Large Heat-Labile Bacteriocin. J. Antimicrob. Chemother. 2009;63:679–686. doi: 10.1093/jac/dkn552. [DOI] [PubMed] [Google Scholar]
- 60.Preciado G.M., Michel M.M., Villarreal-Morales S.L., Flores-Gallegos A.C., Aguirre-Joya J., Morlett-Chávez J., Aguilar C.N., Rodríguez-Herrera R. Chapter 16—Bacteriocins and Its Use for Multidrug-Resistant Bacteria Control. In: Kon K., Rai M., editors. Antibiotic Resistance. Academic Press; Cambridge, MA, USA: 2016. pp. 329–349. [Google Scholar]
- 61.De Arauz L.J., Jozala A.F., Mazzola P.G., Penna T.C.V. Nisin Biotechnological Production and Application: A Review. Trends Food Sci. Technol. 2009;20:146–154. doi: 10.1016/j.tifs.2009.01.056. [DOI] [Google Scholar]
- 62.Howell T.H., Fiorellini J.P., Blackburn P., Projan S.J., De La Harpe J., Williams R.C. The Effect of a Mouthrinse Based on Nisin, a Bacteriocin, on Developing Plaque and Gingivitis in Beagle Dogs. J. Clin. Periodontol. 1993;20:335–339. doi: 10.1111/j.1600-051X.1993.tb00369.x. [DOI] [PubMed] [Google Scholar]
- 63.Cunha E., Trovão T., Pinheiro A., Nunes T., Santos R., Da Silva J.M., Braz B.S., Tavares L., Veiga A.S., Oliveira M. Potential of Two Delivery Systems for Nisin Topical Application to Dental Plaque Biofilms in Dogs. BMC Vet. Res. 2018;14:1–10. doi: 10.1186/s12917-018-1692-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Harvey C.E. Periodontal Disease in Dogs. Vet. Clin. N. Am. Small Anim. Pract. 1998;28:1111–1128. doi: 10.1016/S0195-5616(98)50105-2. [DOI] [PubMed] [Google Scholar]
- 65.Oliveira M., Tavares M., Gomes D., Touret T., Braz B.S., Tavares L., Semedo-Lemsaddek T. Virulence Traits and Antibiotic Resistance among Enterococci Isolated from Dogs with Periodontal Disease. Comp. Immunol. Microbiol. Infect. Dis. 2016;46:27–31. doi: 10.1016/j.cimid.2016.04.002. [DOI] [PubMed] [Google Scholar]
- 66.Cunha E., Rebelo S., Carneiro C., Tavares L., Carreira L.M., Oliveira M. A Polymicrobial Biofilm Model for Testing the Antimicrobial Potential of a Nisin-Biogel for Canine Periodontal Disease Control. BMC Vet. Res. 2020;16:1–10. doi: 10.1186/s12917-020-02646-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Iseppi R., Messi P., Anacarso I., Bondi M., Sabia C., Condò C., de Niederhausern S. Antimicrobial Resistance and Virulence Traits in Enterococcus Strains Isolated from Dogs and Cats. New Microbiol. 2015;38:369–378. [PubMed] [Google Scholar]
- 68.Bertelloni F., Salvadori C., Lotti G., Cerri D., Ebani V.V. Antimicrobial Resistance in Enterococcus Strains Isolated from Healthy Domestic Dogs. Acta Microbiol. Immunol. Hung. 2016;64:301–312. doi: 10.1556/030.63.2016.021. [DOI] [PubMed] [Google Scholar]
- 69.Zoonoses. [(accessed on 16 February 2021)]; Available online: https://www.who.int/news-room/fact-sheets/detail/zoonoses.
- 70.Cunha E., Freitas F.B., Braz B.S., Da Silva J.M., Tavares L., Veiga A.S., Oliveira M. Polyphasic Validation of a Nisin-Biogel to Control Canine Periodontal Disease. Antibiotics. 2020;9:180. doi: 10.3390/antibiotics9040180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Hamp S.-E., Lindhe J., Löe H. Long Term Effect of Chlorhexidine on Developing Gingivitis in the Beagle Dog. J. Periodontal Res. 1973;8:63–70. doi: 10.1111/j.1600-0765.1973.tb00752.x. [DOI] [PubMed] [Google Scholar]
- 72.Charbonneau D., Snider A. Reduced Chlorhexidine Tooth Stain Coverage by Sequential Administration of Monoperoxyphthalic Acid in the Beagle Dog. J. Dent. Res. 1997;76:1596–1601. doi: 10.1177/00220345970760091201. [DOI] [PubMed] [Google Scholar]
- 73.Damborg P., Top J., Hendrickx A.P.A., Dawson S., Willems R.J.L., Guardabassi L. Dogs Are a Reservoir of Ampicillin-Resistant Enterococcus faecium Lineages Associated with Human Infections. Appl. Environ. Microbiol. 2009;75:2360–2365. doi: 10.1128/AEM.02035-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Furlaneto-Maia L., Ramalho R., Rocha K.R., Furlaneto M.C. Antimicrobial Activity of Enterocins against Listeria SP. and Other Food Spoilage Bacteria. Biotechnol. Lett. 2020;42:797–806. doi: 10.1007/s10529-020-02810-7. [DOI] [PubMed] [Google Scholar]
- 75.Kubašová I., Diep D.B., Ovchinnikov K.V., Lauková A., Strompfová V. Bacteriocin Production and Distribution of Bacteriocin-Encoding Genes in Enterococci from Dogs. Int. J. Antimicrob. Agents. 2020;55:105859. doi: 10.1016/j.ijantimicag.2019.11.016. [DOI] [PubMed] [Google Scholar]
- 76.Taylor J., Hirsh A., Mattick A. The Treatment of Bovine Streptococcal and Staphylococcal Mastitis with Nisin. Vet. Res. 1949;61:197–198. [Google Scholar]
- 77.Broadbent J., Chou Y., Gillies K., Kondo J. Nisin Inhibits Several Gram-Positive, Mastitis-Causing Pathogens. J. Dairy Sci. 1989;72:3342–3345. doi: 10.3168/jds.S0022-0302(89)79496-0. [DOI] [PubMed] [Google Scholar]
- 78.Cao L., Wu J., Xie F., Hu S., Mo Y. Efficacy of Nisin in Treatment of Clinical Mastitis in Lactating Dairy Cows. J. Dairy Sci. 2007;90:3980–3985. doi: 10.3168/jds.2007-0153. [DOI] [PubMed] [Google Scholar]
- 79.Wu J., Hu S., Cao L. Therapeutic Effect of Nisin Z on Subclinical Mastitis in Lactating Cows. Antimicrob. Agents Chemother. 2007;51:3131–3135. doi: 10.1128/AAC.00629-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Feitosa E., Bonassi N.M., Loh W. Vesicle−Micelle Transition in Mixtures of Dioctadecyldimethylammonium Chloride and Bromide with Nonionic and Zwitterionic Surfactants. Langmuir. 2006;22:4512–4517. doi: 10.1021/la052923j. [DOI] [PubMed] [Google Scholar]
- 81.Castelani L., Arcaro J., Braga J., Bosso A., Moura Q., Esposito F., Sauter I., Cortez M., Lincopan N. Short Communication: Activity of Nisin, Lipid Bilayer Fragments and Cationic Nisin-Lipid Nanoparticles against Multi-Drug-Resistant Staphylococcus spp. Isolated from Bovine Mastitis. J. Dairy Sci. 2019;102:678–683. doi: 10.3168/jds.2018-15171. [DOI] [PubMed] [Google Scholar]
- 82.Xue T., Chen X., Shang F. Short Communication: Effects of Lactose and Milk on the Expression of Biofilm-Associated Genes in Staphylococcus Aureus Strains Isolated from a Dairy Cow with Mastitis. J. Dairy Sci. 2014;97:6129–6134. doi: 10.3168/jds.2014-8344. [DOI] [PubMed] [Google Scholar]
- 83.Ceotto-Vigoder H., Marques S.L.S., Santos I.N.S., Alves M.D.B., Barrias E.S., Potter A., Alviano D.S., Bastos M.C.F. Nisin and Lysostaphin Activity against Preformed Biofilm of Staphylococcus aureus Involved in Bovine Mastitis. J. Appl. Microbiol. 2016;121:101–114. doi: 10.1111/jam.13136. [DOI] [PubMed] [Google Scholar]
- 84.Andre C., Pimentel-Filho N.D.J., Costa P.M.D.A., Vanetti M.C.D. Changes in the Composition and Architecture of Staphylococcal Biofilm by Nisin. Braz. J. Microbiol. 2019;50:1083–1090. doi: 10.1007/s42770-019-00135-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.LeBel G., Piché F., Frenette M., Gottschalk M., Grenier D. Antimicrobial Activity of Nisin against the Swine Pathogen Streptococcus suis and Its Synergistic Interaction with Antibiotics. Positron. 2013;50:19–23. doi: 10.1016/j.peptides.2013.09.014. [DOI] [PubMed] [Google Scholar]
- 86.Zhu H., Han L., Ni Y., Yu Z., Wang D., Zhou J., Li B., Zhang W., He K. In vitro and In vivo Antibacterial Effects of Nisin Against Streptococcus suis. Probiotics Antimicrob. Proteins. 2021:1–13. doi: 10.1007/s12602-020-09732-w. [DOI] [PubMed] [Google Scholar]
- 87.Campion A., Casey P.G., Field D., Cotter P.D., Hill C., Ross R.P. In vivo activity of Nisin A and Nisin V against Listeria monocytogenes in mice. BMC Microbiol. 2013;13:23. doi: 10.1186/1471-2180-13-23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Lebel G., Vaillancourt K., Frenette M., Gottschalk M., Grenier D. Suicin 90-1330 from a Nonvirulent Strain of Streptococcus suis: A Nisin-Related Lantibiotic Active on Gram-Positive Swine Pathogens. Appl. Environ. Microbiol. 2014;80:5484–5492. doi: 10.1128/AEM.01055-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Vaillancourt K., Lebel G., Frenette M., Fittipaldi N., Gottschalk M., Grenier D. Purification and Characterization of Suicin 65, a Novel Class I Type B Lantibiotic Produced by Streptococcus suis. PLoS ONE. 2015;10:e0145854. doi: 10.1371/journal.pone.0145854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Vaillancourt K., Lebel G., Frenette M., Gottschalk M., Grenier D. Suicin 3908, a New Lantibiotic Produced by a Strain of Streptococcus suis Serotype 2 Isolated from a Healthy Carrier Pig. PLoS ONE. 2015;10:e0117245. doi: 10.1371/journal.pone.0117245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Wang J., Gao Y., Teng K., Zhang J., Sun S., Zhong J. Restoration of Bioactive Lantibiotic Suicin from a Remnantlan Locus of Pathogenic Streptococcus suis Serotype 2. Appl. Environ. Microbiol. 2013;80:1062–1071. doi: 10.1128/AEM.03213-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Bin P., Tang Z., Liu S., Chen S., Xia Y., Liu J., Wu H., Zhu G. Intestinal Microbiota Mediates Enterotoxigenic Escherichia Coli-Induced Diarrhea in Piglets. BMC Vet. Res. 2018;14:1–13. doi: 10.1186/s12917-018-1704-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Liu Y.-Y., Wang Y., Walsh T.R., Yi L.-X., Zhang R., Spencer J., Doi Y., Tian G., Dong B., Huang X., et al. Emergence of PlasMid-mediated Colistin Resistance Mechanism MCR-1 in Animals and Human Beings in China: A Microbiological and Molecular Biological Study. Lancet Infect. Dis. 2016;16:161–168. doi: 10.1016/S1473-3099(15)00424-7. [DOI] [PubMed] [Google Scholar]
- 94.Aguirre L., Vidal A., Seminati C., Tello M., Redondo N., Darwich L., Martín M. Antimicrobial Resistance Profile and Prevalence of Extended-Spectrum Beta-Lactamases (ESBL), AmpC Beta-Lactamases and Colistin Resistance (MCR) Genes in Escherichia Coli from Swine Between 1999 and 2018. Porc. Health Manag. 2020;6:1–6. doi: 10.1186/s40813-020-00146-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Al Atya A.K., Abriouel H., Kempf I., Jouy E., Auclair E., Vachée A., Drider D. Effects of Colistin and Bacteriocins Combinations on the In Vitro Growth of Escherichia coli Strains from Swine Origin. Probiotics Antimicrob. Proteins. 2016;8:183–190. doi: 10.1007/s12602-016-9227-9. [DOI] [PubMed] [Google Scholar]
- 96.Lauková A., Styková E., Kubašová I., Gancarčíková S., Plachá I., Mudroňová D., Kandričáková A., Miltko R., Belzecki G., Valocký I., et al. Enterocin M and its Beneficial Effects in Horses—A Pilot Experiment. Probiotics Antimicrob. Proteins. 2018;10:420–426. doi: 10.1007/s12602-018-9390-2. [DOI] [PubMed] [Google Scholar]
- 97.Lauková A., Chrastinová Ľ., Plachá I., Kandričáková A., Szabóová R., Strompfová V., Chrenková M., Čobanová K., Žitňan R. Beneficial Effect of Lantibiotic Nisin in Rabbit Husbandry. Probiotics Antimicrob. Proteins. 2014;6:41–46. doi: 10.1007/s12602-014-9156-4. [DOI] [PubMed] [Google Scholar]
- 98.Simonová M.P., Chrastinová Ľ., Chrenková M., Formelová Z., Lauková A. Lantibiotic Nisin Applied in Broiler Rabbits and Its Effect on the Growth Performance and Carcass Quality. Probiotics Antimicrob. Proteins. 2019;11:1414–1417. doi: 10.1007/s12602-019-9525-0. [DOI] [PubMed] [Google Scholar]
- 99.Araújo C., Muñoz-Atienza E., Pérez-Sánchez T., Poeta P., Igrejas G., Hernández P.E., Herranz C., Ruiz-Zarzuela I., Cintas L.M., Nisin Z. Production by Lactococcus lactis subsp. Cremoris WA2-67 of Aquatic Origin as a Defense Mechanism to Protect Rainbow Trout (Oncorhynchus mykiss, Walbaum) Against Lactococcus garvieae. Mar. Biotechnol. 2015;17:820–830. doi: 10.1007/s10126-015-9660-x. [DOI] [PubMed] [Google Scholar]
- 100.Baños A., Ariza J.J., Nuñez C., Gil-Martínez L., García-López J.D., Martínez-Bueno M., Valdivia E. Effects of Enterococcus Faecalis UGRA10 and the Enterocin AS-48 against the Fish Pathogen Lactococcus garvieae. Studies In Vitro and In Vivo. Food Microbiol. 2019;77:69–77. doi: 10.1016/j.fm.2018.08.002. [DOI] [PubMed] [Google Scholar]
- 101.Sha Y., Wang B., Liu M., Jiang K., Wang L. Interaction between Lactobacillus Pentosus HC-2 and Vibrio Parahaemolyticus E1 in Litopenaeus Vannamei in Vivo and in Vitro. Aquaculture. 2016;465:117–123. doi: 10.1016/j.aquaculture.2016.09.007. [DOI] [Google Scholar]
- 102.Lüders T., Birkemo G.A., Fimland G., Nissen-Meyer J., Nes I.F. Strong Synergy between a Eukaryotic Antimicrobial Peptide and Bacteriocins from Lactic Acid Bacteria. Appl. Environ. Microbiol. 2003;69:1797–1799. doi: 10.1128/AEM.69.3.1797-1799.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Van Der Linden D.S., Short D., Dittmann A., Yu P.-L. Synergistic Effects of Ovine-Derived Cathelicidins and Other Antimicrobials against Escherichia coli O157:H7 and Staphylococcus aureus 1056 MRSA. Biotechnol. Lett. 2009;31:1265–1267. doi: 10.1007/s10529-009-0010-9. [DOI] [PubMed] [Google Scholar]
- 104.Galvin M., Hill C., Ross R.P. Lacticin 3147 Displays Activity in Buffer against Gram-Positive Bacterial Pathogens Which Appear Insensitive in Standard Plate Assays. Lett. Appl. Microbiol. 1999;28:355–358. doi: 10.1046/j.1365-2672.1999.00550.x. [DOI] [PubMed] [Google Scholar]
- 105.McAuliffe O., Hill C., Ross R.P. Inhibition of Listeria monocytogenesin Cottage Cheese Manufactured with a Lacticin 3147-Producing Starter Culture. J. Appl. Microbiol. 1999;86:251–256. doi: 10.1046/j.1365-2672.1999.00663.x. [DOI] [PubMed] [Google Scholar]
- 106.Rea M.C., Clayton E., O’Connor P.M., Shanahan F., Kiely B., Ross R.P., Hill C. Antimicrobial Activity of Lacticin 3147 against Clinical Clostridium Difficile Strains. J. Med Microbiol. 2007;56:940–946. doi: 10.1099/jmm.0.47085-0. [DOI] [PubMed] [Google Scholar]
- 107.Piper C., Draper L.A., Cotter P.D., Ross R.P., Hill C. A Comparison of the Activities of Lacticin 3147 and Nisin against Drug-Resistant Staphylococcus aureus and Enterococcus Species. J. Antimicrob. Chemother. 2009;64:546–551. doi: 10.1093/jac/dkp221. [DOI] [PubMed] [Google Scholar]
- 108.Carroll J., Draper L.A., O’Connor P.M., Coffey A., Hill C., Ross R.P., Cotter P.D., O’Mahony J. Comparison of the Activities of the Lantibiotics Nisin and Lacticin 3147 against Clinically Significant Mycobacteria. Int. J. Antimicrob. Agents. 2010;36:132–136. doi: 10.1016/j.ijantimicag.2010.03.029. [DOI] [PubMed] [Google Scholar]
- 109.Kitching M., Mathur H., Flynn J., Byrne N., Dillon P., Sayers R., Rea M.C., Hill C., Ross R.P. A Live Bio-Therapeutic for Mastitis, Containing Lactococcus lactis DPC3147 with Comparable Efficacy to Antibiotic Treatment. Front. Microbiol. 2019;10:2220. doi: 10.3389/fmicb.2019.02220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Ryan M., Flynn J., Hill C., Ross R., Meaney W. The Natural Food Grade Inhibitor, Lacticin 3147, Reduced the Incidence of Mastitis After Experimental Challenge with Streptococcus dysgalactiae in Nonlactating Dairy Cows. J. Dairy Sci. 1999;82:2625–2631. doi: 10.3168/jds.S0022-0302(99)75519-0. [DOI] [PubMed] [Google Scholar]
- 111.Twomey D., Wheelock A., Flynn J., Meaney W., Hill C., Ross R. Protection against Staphylococcus aureus Mastitis in Dairy Cows Using a Bismuth-Based Teat Seal Containing the Bacteriocin, Lacticin 3147. J. Dairy Sci. 2000;83:1981–1988. doi: 10.3168/jds.S0022-0302(00)75075-2. [DOI] [PubMed] [Google Scholar]
- 112.Kim S., Shin S., Koo H., Youn J.-H., Paik H.-D., Park Y. In Vitro Antimicrobial Effect and in Vivo Preventive and Therapeutic Effects of Partially Purified Lantibiotic Lacticin NK34 against Infection by Staphylococcus Species Isolated from Bovine Mastitis. J. Dairy Sci. 2010;93:3610–3615. doi: 10.3168/jds.2010-3129. [DOI] [PubMed] [Google Scholar]
- 113.Ogunbanwo S., Sanni A., Onilude A. Influence of Bacteriocin in the Control of Escherichia coli Infection of Broiler Chickens in Nigeria. World J. Microbiol. Biotechnol. 2004;20:51–56. doi: 10.1023/B:WIBI.0000013311.43842.74. [DOI] [Google Scholar]
- 114.Wang Q., Cui Y., Wang W., Xu J., Xu L. Production of Two Bacteriocins in Various Growth Conditions Produced by Gram-Positive Bacteria Isolated from Chicken Cecum. Can. J. Microbiol. 2012;58:93–101. doi: 10.1139/w11-108. [DOI] [PubMed] [Google Scholar]
- 115.Stern N.J., Svetoch E.A., Eruslanov B.V., Kovalev Y.N., Volodina L.I., Perelygin V.V., Mitsevich E.V., Mitsevich I.P., Levchuk V.P. Paenibacillus polymyxa Purified Bacteriocin To Control Campylobacter jejuni in Chickens. J. Food Prot. 2005;68:1450–1453. doi: 10.4315/0362-028X-68.7.1450. [DOI] [PubMed] [Google Scholar]
- 116.Daeschel M.A., Klaenhammer T.R. Association of a 13.6-Megadalton Plasmid in Pediococcus pentosaceus with Bacteriocin Activity. Appl. Environ. Microbiol. 1985;50:1538–1541. doi: 10.1128/AEM.50.6.1538-1541.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Piva A., Headon D.R. Pediocin A, a Bacteriocin Produced by Pediococcus pentosaceus FBB61. Microbiology. 1994;140:697–702. doi: 10.1099/00221287-140-4-697. [DOI] [PubMed] [Google Scholar]
- 118.Keyburn A.L., Boyce J.D., Vaz P., Bannam T.L., Ford M.E., Parker D., Di Rubbo A., Rood J.I., Moore R.J. NetB, a New Toxin That Is Associated with Avian Necrotic Enteritis Caused by Clostridium perfringens. PLoS Pathog. 2008;4:e26. doi: 10.1371/journal.ppat.0040026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Grilli E., Messina M.R., Catelli E., Morlacchini M., Piva A. Pediocin A Improves Growth Performance of Broilers Challenged with Clostridium perfringens. Poult. Sci. 2009;88:2152–2158. doi: 10.3382/ps.2009-00160. [DOI] [PubMed] [Google Scholar]
- 120.Józefiak D., Sip A., Rawski M., Rutkowski A., Kaczmarek S., Hojberg O., Jensen B., Engberg R. Dietary Divercin Modifies Gastrointestinal Microbiota and Improves Growth Performance in Broiler Chickens. Br. Poult. Sci. 2011;52:492–499. doi: 10.1080/00071668.2011.602963. [DOI] [PubMed] [Google Scholar]
- 121.Józefiak D., Kierończyk B., Juśkiewicz J., Zduńczyk Z., Rawski M., Długosz J., Sip A., Højberg O. Dietary Nisin Modulates the Gastrointestinal Microbial Ecology and Enhances Growth Performance of the Broiler Chickens. PLoS ONE. 2013;8:e85347. doi: 10.1371/journal.pone.0085347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Kierończyk B., Rawski M., Mikołajczak Z., Świątkiewicz S., Józefiak D. Nisin as a Novel Feed Additive: The Effects on Gut Microbial Modulation and Activity, Histological Parameters, and Growth Performance of Broiler Chickens. Animals. 2020;10:101. doi: 10.3390/ani10010101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Carrasco J.M.D., Casanova N.A., Miyakawa M.E.F. Microbiota, Gut Health and Chicken Productivity: What Is the Connection? Microorganisms. 2019;7:374. doi: 10.3390/microorganisms7100374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Line J.E., Svetoch E.A., Eruslanov B.V., Perelygin V.V., Mitsevich E.V., Mitsevich I.P., Levchuk V.P., Svetoch O.E., Seal B.S., Siragusa G.R., et al. Isolation and Purification of Enterocin E-760 with Broad Antimicrobial Activity against Gram-Positive and Gram-Negative Bacteria. Antimicrob. Agents Chemother. 2007;52:1094–1100. doi: 10.1128/AAC.01569-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Pieterse R., Todorov S.D., Leon M.T.D. Mode of action and In Vitro Susceptibility of Mastitis Pathogens to Macedocin ST91KM and preparation of a teat seal containing the bacteriocin. Braz. J. Microbiol. 2010;41:133–145. doi: 10.1590/S1517-83822010000100020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Dicks L.M.T., Dreyer L., Smith C., Van Staden A.D. A Review: The Fate of Bacteriocins in the Human Gastro-Intestinal Tract: Do They Cross the Gut–Blood Barrier? Front. Microbiol. 2018;9:2297. doi: 10.3389/fmicb.2018.02297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Stern N.J., Svetoch E.A., Eruslanov B.V., Perelygin V.V., Mitsevich E.V., Mitsevich I.P., Pokhilenko V.D., Levchuk V.P., Svetoch O.E., Seal B.S. Isolation of a Lactobacillus salivarius Strain and Purification of Its Bacteriocin, Which Is Inhibitory to Campylobacter jejuni in the Chicken Gastrointestinal System. Antimicrob. Agents Chemother. 2006;50:3111–3116. doi: 10.1128/AAC.00259-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Umu Ö.C., Bäuerl C., Oostindjer M., Pope P.B., Hernández P.E., Pérez-Martínez G., Diep D.B. The Potential of Class II Bacteriocins to Modify Gut Microbiota to Improve Host Health. PLoS ONE. 2016;11:e0164036.:e0164036. doi: 10.1371/journal.pone.0164036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Chaucheyras-Durand F., Durand H. Probiotics in Animal Nutrition and Health. Benef. Microbes. 2010;1:3–9. doi: 10.3920/BM2008.1002. [DOI] [PubMed] [Google Scholar]
- 130.Zhang M., Sun K., Wu Y., Yang Y., Tso P., Wu Z. Interactions between Intestinal Microbiota and Host Immune Response in Inflammatory Bowel Disease. Front. Immunol. 2017;8:942. doi: 10.3389/fimmu.2017.00942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Sharma A., Gilbert J. Microbial Exposure and Human Health. Curr. Opin. Microbiol. 2018;44:79–87. doi: 10.1016/j.mib.2018.08.003. [DOI] [PubMed] [Google Scholar]
- 132.Kubašová I., Strompfová V., Lauková A. Safety Assessment of Commensal Enterococci from Dogs. Folia Microbiol. 2017;62:491–498. doi: 10.1007/s12223-017-0521-z. [DOI] [PubMed] [Google Scholar]
- 133.Strompfová V., Kubašová I., Ščerbová J., Maďari A., Gancarčíková S., Mudroňová D., Miltko R., Belzecki G., Lauková A. Oral Administration of Bacteriocin-Producing and Non-producing Strains of Enterococcus faecium in Dogs. Appl. Microbiol. Biotechnol. 2019;103:4953–4965. doi: 10.1007/s00253-019-09847-3. [DOI] [PubMed] [Google Scholar]
- 134.Zhang J., Deng J., Li Y., Yang Q. The effect of Lactobacillus on the Expression of Porcine β-Defensin-2 in the Digestive Tract of Piglets. Livest. Sci. 2011;138:259–265. doi: 10.1016/j.livsci.2011.01.001. [DOI] [Google Scholar]
- 135.Messaoudi S., Manai M., Kergourlay G., Prévost H., Connil N., Chobert J.-M., Dousset X. Lactobacillus salivarius: Bacteriocin and Probiotic Activity. Food Microbiol. 2013;36:296–304. doi: 10.1016/j.fm.2013.05.010. [DOI] [PubMed] [Google Scholar]
- 136.Corr S.C., Li Y., Riedel C.U., O’Toole P.W., Hill C., Gahan C.G.M. Bacteriocin Production as a Mechanism for the Antiinfective Activity of Lactobacillus salivarius UCC118. Proc. Natl. Acad. Sci. USA. 2007;104:7617–7621. doi: 10.1073/pnas.0700440104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Claesson M.J., Li Y., Leahy S., Canchaya C., Van Pijkeren J.P., Cerdeño-Tárraga A.M., Parkhill J., Flynn S., O’Sullivan G.C., Collins J.K., et al. Multireplicon Genome Architecture of Lactobacillus salivarius. Proc. Natl. Acad. Sci. USA. 2006;103:6718–6723. doi: 10.1073/pnas.0511060103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Riboulet-Bisson E., Sturme M.H.J., Jeffery I.B., O’Donnell M.M., Neville B.A., Forde B.M., Claesson M.J., Harris H., Gardiner G.E., Casey P.G., et al. Effect of Lactobacillus salivarius Bacteriocin Abp118 on the Mouse and Pig Intestinal Microbiota. PLoS ONE. 2012;7:e31113. doi: 10.1371/journal.pone.0031113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Hu J., Ma L., Nie Y., Chen J., Zheng W., Wang X., Xie C., Zheng Z., Wang Z., Yang T., et al. A Microbiota-Derived Bacteriocin Targets the Host to Confer Diarrhea Resistance in Early-Weaned Piglets. Cell Host Microbe. 2018;24:817–832.e8. doi: 10.1016/j.chom.2018.11.006. [DOI] [PubMed] [Google Scholar]
- 140.Da Silva A.B., Back M., Daguer H., Palmeira M., Ploêncio L.A.D.S., Molognoni L., Peripolli V., Bianchi I. Carry-over and Contamination of Veterinary Drugs in Feed Production Lines for Poultry and Pigs. Food Addit. Contam. Part A. 2019;36:740–751. doi: 10.1080/19440049.2019.1585580. [DOI] [PubMed] [Google Scholar]
- 141.Kizerwetter-Swida M., Binek M. Selection of Potentially Probiotic Lactobacillus Strains towards Their Inhibitory Activity against Poultry Enteropathogenic bacteria. Pol. J. Microbiol. 2005;54:287–294. [PubMed] [Google Scholar]
- 142.Timmerman H.M., Veldman A., Elsen E.V.D., Rombouts F.M., Beynen A.C. Mortality and Growth Performance of Broilers Given Drinking Water Supplemented with Chicken-Specific Probiotics. Poult. Sci. 2006;85:1383–1388. doi: 10.1093/ps/85.8.1383. [DOI] [PubMed] [Google Scholar]
- 143.Giannenas I., Tsalie E., Triantafillou E., Hessenberger S., Teichmann K., Mohnl M., Tontis D. Assessment of Probiotics Supplementation via Feed or Water on the Growth Performance, Intestinal Morphology and Microflora of Chickens after Experimental Infection Witheimeria acervulina, Eimeria maxima andeimeria tenella. Avian Pathol. 2014;43:209–216. doi: 10.1080/03079457.2014.899430. [DOI] [PubMed] [Google Scholar]
- 144.Ritzi M.M., Abdelrahman W., Van-Heerden K., Mohnl M., Barrett N.W., Dalloul R.A. Combination of Probiotics and Coccidiosis Vaccine Enhances Protection against an Eimeria Challenge. Vet. Res. 2016;47:1–8. doi: 10.1186/s13567-016-0397-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Pender C., Kim S., Potter T., Ritzi M., Young M., Dalloul R. Effects of in Ovo Supplementation of Probiotics on Performance and Immunocompetence of Broiler Chicks to an Eimeria Challenge. Benef. Microbes. 2016;7:699–705. doi: 10.3920/BM2016.0080. [DOI] [PubMed] [Google Scholar]
- 146.Shin M., Han S., Ji A., Kim K., Lee W. Isolation and Characterization of Bacteriocin-Producing Bacteria from the Gastrointestinal Tract of Broiler Chickens for Probiotic Use. J. Appl. Microbiol. 2008;105:2203–2212. doi: 10.1111/j.1365-2672.2008.03935.x. [DOI] [PubMed] [Google Scholar]
- 147.Sabo S.D.S., Mendes M.A., Araújo E.D.S., Muradian L.B.D.A., Makiyama E.N., Leblanc J.G., Borelli P., Fock R.A., Knöbl T., Oliveira R.P.D.S. Bioprospecting of Probiotics with Antimicrobial Activities against Salmonella Heidelberg and That Produce B-Complex Vitamins as Potential Supplements in Poultry Nutrition. Sci. Rep. 2020;10:1–14. doi: 10.1038/s41598-020-64038-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Hansen J.N., Sandine W.E. Nisin as a Model Food Preservative. Crit. Rev. Food Sci. Nutr. 1994;34:69–93. doi: 10.1080/10408399409527650. [DOI] [PubMed] [Google Scholar]
- 149.Pablo M.A., Gaforio J.J., Gallego A.M., Ortega E., Gã¡lvez A.M., Lã³Pez G.A.D.C. Evaluation of Immunomodulatory Effects of Nisin-Containing Diets on Mice. FEMS Immunol. Med Microbiol. 1999;24:35–42. doi: 10.1111/j.1574-695X.1999.tb01262.x. [DOI] [PubMed] [Google Scholar]
- 150.Małaczewska J., Kaczorek-Łukowska E., Wójcik R., Rękawek W., Siwicki A.K. In Vitro Immunomodulatory Effect of Nisin on Porcine Leucocytes. J. Anim. Physiol. Anim. Nutr. 2019;103:882–893. doi: 10.1111/jpn.13085. [DOI] [PubMed] [Google Scholar]
- 151.Aranha C., Gupta S., Reddy K. Assessment of Cervicovaginal Cytokine Levels Following Exposure to Microbicide Nisin Gel in Rabbits. Cytokine. 2008;43:63–70. doi: 10.1016/j.cyto.2008.04.005. [DOI] [PubMed] [Google Scholar]
- 152.Gupta S.M., Aranha C.C., Mohanty M.C., Reddy K.V.R. Toll-Like Receptors and Cytokines as Surrogate Biomarkers for Evaluating Vaginal Immune Response following Microbicide Administration. Mediat. Inflamm. 2008;2008:1–11. doi: 10.1155/2008/534532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Begde D., Bundale S., Mashitha P., Rudra J., Nashikkar N., Upadhyay A. Immunomodulatory Efficacy of Nisin-A Bacterial Lantibiotic Peptide. J. Pept. Sci. 2011;17:438–444. doi: 10.1002/psc.1341. [DOI] [PubMed] [Google Scholar]
- 154.Kindrachuk J., Jenssen H., Elliott M., Nijnik A., Magrangeas-Janot L., Pasupuleti M., Thorson L., Ma S., Easton D.M., Bains M., et al. Manipulation of Innate Immunity by a Bacterial Secreted Peptide: Lantibiotic Nisin Z Is Selectively Immunomodulatory. Innate Immun. 2012;19:315–327. doi: 10.1177/1753425912461456. [DOI] [PubMed] [Google Scholar]
- 155.Villamil L., Figueras A., Novoa B. Immunomodulatory Effects of Nisin in Turbot (Scophthalmus maximus L.) Fish Shellfish. Immunol. 2003;14:157–169. doi: 10.1006/fsim.2002.0425. [DOI] [PubMed] [Google Scholar]
- 156.Malvisi M., Stuknytė M., Magro G., Minozzi G., Giardini A., De Noni I., Piccinini R. Antibacterial Activity and Immunomodulatory Effects on a Bovine Mammary Epithelial Cell Line Exerted by Nisin A-Producing Lactococcus Lactis Strains. J. Dairy Sci. 2016;99:2288–2296. doi: 10.3168/jds.2015-10161. [DOI] [PubMed] [Google Scholar]
- 157.Jia Z., He M., Wang C., Chen A., Zhang X., Xu J., Fu H., Liu B. Nisin Reduces Uterine Inflammation in Rats by Modulating Concentrations of Pro- and Anti-Inflammatory Cytokines. Am. J. Reprod. Immunol. 2019;81:e13096. doi: 10.1111/aji.13096. [DOI] [PubMed] [Google Scholar]
- 158.Fu T., Yu M., Yan Q., Liu Y.-M. Bacteriocin Isolated from Lactobacillus Rhamnosus L34 Has Antibacterial Effects in a Rabbit Model of Infection After Mandible Fracture Fixation. Med Sci. Monit. 2018;24:8009–8014. doi: 10.12659/MSM.909630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Zhou B., Zhang D. Antibacterial Effects of Bacteriocins Isolated from Lactobacillus rhamnosus (ATCC 53103) In a Rabbit Model of Knee Implant Infection. Exp. Ther. Med. 2018;15:2985–2989. doi: 10.3892/etm.2018.5790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Gong L., He H., Li D., Cao L., Khan T.A., Li Y., Pan L., Yan L., Ding X., Sun Y., et al. A New Isolate of Pediococcus pentosaceus (SL001) with Antibacterial Activity against Fish Pathogens and Potency in Facilitating the Immunity and Growth Performance of Grass Carps. Front. Microbiol. 2019;10:1384. doi: 10.3389/fmicb.2019.01384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Li M.-F., Zhang B.-C., Li J., Sun L. Sil: A Streptococcus iniae Bacteriocin with Dual Role as an Antimicrobial and an Immunomodulator That Inhibits Innate Immune Response and Promotes, S. iniae Infection. PLoS ONE. 2014;9:e96222. doi: 10.1371/journal.pone.0096222. [DOI] [PMC free article] [PubMed] [Google Scholar]