Abstract
The global expansion of coronavirus disease 2019 (COVID-19) caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has emerged as one of the greatest public health challenges and imposes a great threat to human health. Innate immunity plays vital roles in eliminating viruses through initiating type I interferons (IFNs)-dependent antiviral responses and inducing inflammation. Therefore, optimal activation of innate immunity and balanced type I IFN responses and inflammation are beneficial for efficient elimination of invading viruses. However, SARS-CoV-2 manipulates the host’s innate immune system by multiple mechanisms, leading to aberrant type I IFN responses and excessive inflammation. In this review, we will emphasize the recent advances in the understanding of the crosstalk between host innate immunity and SARS-CoV-2 to explain the imbalance between inflammation and type I IFN responses caused by viral infection, and explore potential therapeutic targets for COVID-19.
Keywords: COVID-19, SARS-CoV-2, type I interferons, inflammation, innate immunity
Introduction
The global expansion of coronavirus disease 2019 (COVID-19) caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has emerged as one of the greatest public health challenges and imposes a considerable threat to human health. Similar to SARS-CoV and MERS-CoV infection, SARS-CoV-2 infection frequently induces high levels of proinflammatory cytokines, leukocyte changes, high D-dimer, and increased lactate dehydrogenase levels (1–3). COVID-19 exhibits varied clinical manifestations, ranging from fever, cough, diarrhea, and fatigue to pulmonary edema, septic shock, multiple organ failure, and even death. The multiple symptoms indicate that COVID-19 is a systemic inflammatory disorder rather than a single respiratory disease (2).
Innate immunity functions as the first line of defense for the host to eliminate invading viruses through the initiation of type I IFNs (including IFN-α and IFN-β)-dependent antiviral responses and induction of inflammation (4). As a bridge, it initiates antiviral adaptive immune responses and controls the intensity of adaptive immunity. Therefore, optimal activation of innate immunity and balanced type I IFN responses and inflammation are beneficial for efficient elimination of invading viruses. However, excessive inflammation caused by viral invasion can induce excessive production of inflammatory cytokines and initiate acute respiratory distress syndrome (ARDS), which is associated with the increased risk of death (1, 5–7). In contrast to heightened levels of inflammation, reduced or delayed type I IFN responses accompanied by relatively high viral titers have been observed in COVID-19 patients, particularly in critical patients in intensive care units (ICU) (8, 9). While each cell population of patients with mild COVID-19 displays a coordinated interferon signature, those with severe cases are thought to dampen IFN responses through CD32b (10). Recent evidence from two translational medical researches similarly highlighted the importance of type I IFN. They confirmed that defective IFN signaling stands as the main causes of serious COVID-19 besides advanced age and underlying diseases and may account for nearly 13% of severe cases (11, 12). Imbalance between inflammation and IFN I responses indicates poorer prognosis for COVID-19 patients, and worse disease outcomes. In this review, we will emphasize recent advances in understanding the crosstalk between host innate immunity and SARS-CoV-2, and explain the imbalance between inflammation and IFN I responses to explore potential therapeutic targets for COVID-19.
Entry of SARS-CoV-2
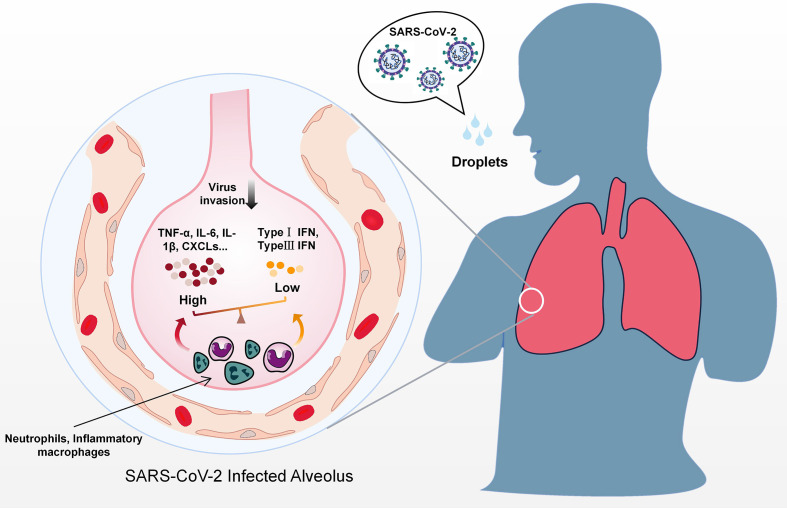
Like many other respiratory coronaviruses, SARS-CoV-2 mainly infects the host via the respiratory tract and is transmitted via respiratory droplets (13) ( Figure 1 ). The SARS-CoV-2 genome is approximately 29.7 kb long and encodes four structural proteins (spike protein S, envelope protein E, membrane protein M, and nucleocapsid protein N), 16 non-structural proteins (NSPs), and 9 accessory proteins (14–16), which share high sequence similarity to their SARS-CoV counterparts.
Figure 1.
Predicted immune dysregulation in the lung during SARS-CoV-2 infection. After contact with droplets containing SARS-CoV-2, host immune responses are activated in the lung. Activation of immune cell subgroups such as inflammatory macrophages and neutrophils results in the secretion of massive amounts of inflammatory cytokines, including TNF-α, IL-6, IL-8, and CXCLs. In contrast to excessive proinflammatory cytokines, IFNs levels are lower during the early phase of infection.
Among all the viral proteins, the S protein initiates the infection process by mediating the attachment of the virus to host cells through angiotensin-converting enzyme 2 (ACE2) (13). ACE2 mainly exists in the intestine, heart, kidneys, and testes, and it is correlated with the distribution of SARS-CoV-2 (17). According to single-cell RNA sequencing (scRNA-Seq), the expression of ACE2 in the lungs was primarily observed in the alveolar type II cells (AT2), which are probably the primary target of SARS-CoV-2 (18). The latest longitudinal study in conjunction with airway and blood samples also indicates that severe inflammation during SARS-CoV-2 infection is mainly driven by cytokines in the lung rather than systemic (19). Even worse, the SARS-CoV-2-induced IFN I responses also evidently increase the cellular ACE2 levels, which may increase susceptibility to infection (20). In addition, SARS-CoV-2-encoded N protein, NSP7b, and NSP8 all participate in viral replication, evolution, and immune evasion during infection (21, 22). Emerging evidence confirms that both structural and nonstructural proteins of SARS-CoV-2 interfere with host innate immune responses and participate in the pathogenesis of COVID-19 (14, 23).
Innate Immune Responses Against SARS-CoV-2
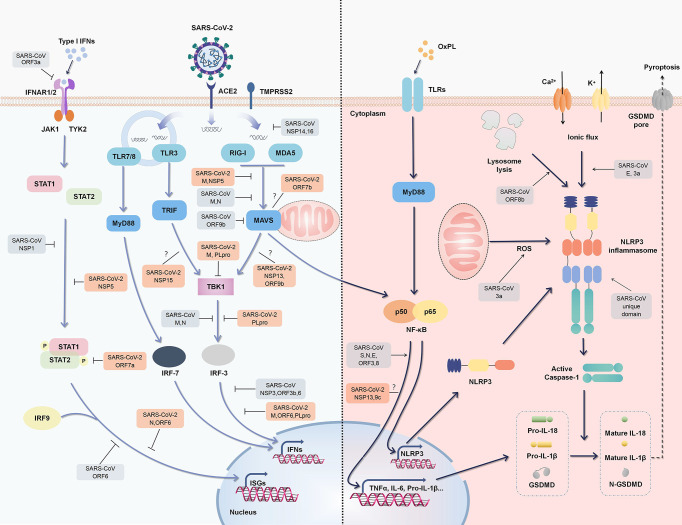
To effectively defend against viruses, host cells initiate antiviral innate immune responses by producing a number of IFNs and other proinflammatory cytokines. IFNs and downstream interferon-stimulated genes (ISGs) play fundamental roles in limiting viral replication (24) ( Figure 2 ). Following invasion, SARS-CoV-2 releases viral RNA and proteins into cells, which are recognized by the host immune system as pathogen associated molecular patterns (PAMPs), thus initiating the secretion of IFNs and antiviral innate immune responses (25). Various pattern recognition receptors (PRRs), including Toll-like receptor 3 (TLR3), TLR7, TLR8, retinoic acid-inducible gene I (RIG-I), and melanoma differentiation-associated protein 5 (MDA5) are candidates for the recognition of SARS-CoV-2, since they are considered to sense viral components; particularly viral RNA (26). Among these PRRs, RIG-I/MDA5 are proven to be the main sensors in MERS-CoV recognition and inflammatory cascade initiation (27). TLR7/8 is also reportedly involved in sensing coronaviruses including SARS-CoV (28).
Figure 2.
Regulation of type I IFN responses and inflammation by SARS-CoV-2 and SARS-CoV. An outline of IFN signaling (left) and major inflammatory signaling (right) is shown, annotated with the known mechanisms by which SARS-CoV activates or suppresses signals (gray). Some SARS-CoV-2 proteins have also been confirmed or speculated to interact with these pathways (orange).
PRR signaling activated by viral components induces nuclear factor (NF)-κB activation, which triggers the expression of a large number of proinflammatory cytokines. Some of the metabolites as oxidized phospholipid (OxPL) are also increased during coronavirus infection, further triggering NF-kB activation via TLR4 (29). NF-κB activation induces the expression of nucleotide-binding oligomerization domain-, leucine-rich repeat- and pyrin domain-containing 3 (NLRP3), pro-IL-1β, and pro-IL-18 (the priming stage of the NLRP3 inflammasome). Subsequently, cellular damage or distress caused by viral infection, leading to the accumulation of reactive oxygen species and ion fluxes (damage associated molecular patterns, DAMPs), results in the activation stage of NLRP3 inflammasome (30). NLRP3 inflammasome activation then mediates the maturation of IL-1β, IL-18, and the propyroptotic factor gasdermin D (GSDMD), and thus induces inflammation-associated cell death known as “pyroptosis” and further amplifies host inflammatory responses (30, 31) ( Figure 2 ).
The activation of multiple PRRs facilitates the establishment of antiviral states by recruiting multiple adaptor proteins (e.g., mitochondrial antiviral signaling protein (MAVS), TANK binding kinase 1 (TBK1), tumor necrosis factor receptor-associated factor (TRAF) 3 and TRAF6) and inducing a variety of cytokines. On this basis, different immune cells are recruited to infection sites and initiate adaptive immune responses (32). Although substantial IFN I responses and optimal inflammation are beneficial for the eradication of invading viruses, an imbalance between type I IFN responses and inflammation induced by different viral proteins can cause numerous detrimental effects ( Figure 1 ). In a SARS-CoV-2 infection hamster model, significant inflammation occurred in distal tissues even while productive SARS-CoV-2 replication was low (33). In the late phase of infection, a large increase in delayed type I IFN responses occurs, which induces the secretion of proinflammatory cytokines such as C-X-C motif chemokine ligand 10 (CXCL10) found in pulmonary autopsy tissues, and recruits and activates inflammatory monocyte-macrophages, resulting in the impairment of T cell responses and lung immunopathology (34, 35). A similar situation was also revealed when single-cell RNA-seq was performed using peripheral blood mononuclear cells obtained from severely affected COVID-19 patients (36). In addition to type I IFNs, type II IFN (IFN-γ) which is mainly secreted by T cells and natural killer cells was also increased in the serum of COVID-19 patients (37). In early SARS-CoV-2 infection increased IFN-γ may contribute to antiviral immunity in multiple ways, including the promotion of antigen presentation, inflammatory cell activation and even by directly stimulating the expression of multiple ISGs (38). As the disease progresses however, the viral infection may destroy T-cells and reduce the production of IFN-γ.
Delayed IFN Responses in SARS-CoV-2 Infection
Like SARS-CoV, SARS-CoV-2 is also highly sensitive to IFNs in vitro (35, 39). In a hamster infection model, intranasal IFN I effectively inhibited SARS-CoV-2 replication and transmission (33). In a phase 2 COVID-19 trial, a combination including IFN-β1b effectively alleviated symptoms and shortened the duration of viral shedding, indicating the critical role of IFNs during SARS-CoV-2 infection (40). Aging and underlying diseases have a potentially negative impact upon IFN production (41–43). Consistent with this, in multiple retrospective cohort studies there have been higher fatalities in elderly patients with severe COVID-19 (2, 3). Similarly, in macaques pulmonary injury was more prominent in an aged group than in a group of young adults (41).
Genetic mutations (TLR3-, TLR7- and IRF7-dependent) and neutralizing auto-antibodies (Abs) that influence type I interferon signaling were shown in severe COVID-19 patients (11, 12, 44). All these findings underscore the importance of IFNs for protection against SARS-CoV-2. During early SARS-CoV-2 infection however, the host type I IFN response can evidently be low and insufficient, which may be partly caused by immune escape mechanisms of viruses. In a longitudinal follow-up study including 32 COVID−19 patients and 16 influenza-associated pneumonia patients who had similar clinical characteristics to the COVID-19 patients, SARS-CoV-2, but not influenza virus, could trigger an untuned immune response that presented as suppressed and delayed IFN responses and a persistent inflammatory response (45). Delayed type I IFN signaling not only provides a key window for viral replication but could induce tissue damage as observed in SARS-CoV infection (35, 46).
Viral Proteins of SARS-CoV-2 Inhibit Type I IFN Secretion
Four non-structural proteins of SARS-COV-2, including NSP13, NSP15, open reading frame (ORF) 7b, and ORF9b, were identified as interactors of host proteins involved in IFN signaling by affinity-purification mass spectrometry (23, 47). At least eight proteins (NSP1, NSP3, NSP12, NSP13, NSP14, ORF3, ORF6, and M) have also been proven to inhibit IFN-β production by gene reporter assays (16). Genetic and clinical data revealed that deletion mutations in NSP1 of SARS-CoV-2 coding region is a variant hotspot that could lead to lower IFN response (48). These studies indicate the existence of a complex regulatory network between SARS-CoV-2 and the host immune system. Viruses tend to downregulate the host immune system by direct disruption of antiviral-associated proteins.
RIG-I activation is dependent on ubiquitination by tripartite motif containing (TRIM)25, of which the interaction could be blocked by NSP5 of SARS-CoV-2 and N protein of SARS-CoV ( Figure 2 ) (26, 49). The M protein of SARS-CoV-2 and SARS-CoV prevents the formation of the TRAF3-TBK1/IKK∈ complex, thus suppressing IFNs production (50, 51). SARS-CoV-2 M protein even affects the formation of the RIG-I–MAVS–TRAF3–TBK1 multi-protein complex and subsequent phosphorylation of IFN regulatory factor 3 (IRF3) (51). As well as steric hindrance, viral components also directly target critical adaptors in antiviral immunity. SARS-CoV-2 ORF6 inhibits IFN signals by preventing IRF3 nuclear translocation (16). As a key protease of SARS-CoV-2 that regulates viral replication and spread, papain-like protease (PLpro, a part of NSP3) attenuates IFN responses via the cleavage of ISG15 from IRF3 (52). SARS-CoV-2 also affects the host through the translational level. NSP1 binds to 40S ribosomal subunits and obstructs host mRNA translation, thus effectively inhibiting RIG-I-mediated IFN responses ( Figure 2 ) (53).
Although a large number of regulatory proteins have been identified via high-throughput screening, their underlying mechanisms with respect to relationships with SARS-CoV-2 are poorly understood. Fortunately, there are great similarities in sequence between SARS-CoV-2 and SARS-CoV, and continued comparison may reveal additional useful information regarding regulation. The N7-methylguanosine (m7G) cap is the defining structural feature of eukaryotic mRNA that distinguishes it from viral RNA. SARS-CoV NSP14 is a novel cap N7-methyltransferase that processes the cap structure of viral RNA to mimic host mRNA and evade recognition (54). ORF9b degrades MAVS, as well as TRAF3 and TRAF6, by usurping poly(C)-binding protein 2 and AID4, a gatekeeper and E3 ligase, thus controlling MAVS expression levels (55). As a crucial adaptor of the antiviral signaling pathway, IRF3 is also a key target of multiple SARS proteins including PLpro, N, ORF3b, and ORF6 protein ( Figure 2 ) (56, 57).
SARS-CoV-2 Proteins Inhibit the JAK-STAT Pathway
As a downstream signaling pathway of IFN, JAK-STAT pathway is crucial for type I IFNs triggered ISGs expression and antiviral responses. During viral infection, responder cells produce and secrete type I IFNs which subsequently bind to their receptor IFNAR (IFN-α/β receptor) to initiate JAK-STAT signaling cascades (58). Both SARS-CoV-2 and SARS-CoV infection affect JAK-STAT signaling, thus inhibiting host antiviral innate immunity independent of the level of IFNs (16, 49, 59–64). Up to now, at least four SARS-CoV-2 proteins (NSP5, ORF7a, N, ORF6) and three SARS-CoV proteins (ORF3a, NSP1, ORF6) have been proved to directly affect the activation of JAK-STAT pathway by multiple mechanisms including mediating STAT1/2 degradation and suppressing their phosphorylation and nuclear translocation (16, 49, 59–64). Resultantly, the expression of ISGs reduced, and antiviral effects get impaired.
Excessive Inflammation Caused by SARS-CoV-2
During SARS-CoV-2 infection, aberrant inflammatory cytokine responses and the induction of a cytokine storm is closely associated with extensive lung damage and disease severity (65). Compared to mild-to-moderate affected patients, in these severe cases higher levels of pro-inflammatory cytokines (including IL-6, IL-10, and TNF-α, etc.) are secreted, and their secretion is correlated with a high serum titer of SARS-CoV-2 and an increased risk of death (66–68). The disrupted secretion of proinflammatory and anti-inflammatory cytokines in severely ill patients results in vascular leakage and fluid accumulation, which is the main cause of ARDS (69). Thus, the seriousness of clinical symptoms may be highly correlated with the inflammatory status of COVID-19 patients.
NF-κB-Associated Inflammation
Abnormal NF-κB activation is vital for the initiation and progression of multiple inflammatory respiratory diseases and ARDS (70, 71). For SARS-CoV, the S, N, and E proteins as well as ORF3 and ORF8 can activate NF-κB signaling, leading to the secretion of proinflammatory cytokines (72–75). During SARS-CoV-2 infection, two non-structural proteins, NSP13 and ORF9c interact with NF-κB signaling proteins (TLE1, TLE3, TLE5, NLRX1, F2RL1, and NDFIP2), suggesting that SARS-CoV-2 can regulate the NF-κB signaling pathway (23). The vasoconstrictor angiotensin II (AngII) is also a key factor involved in the pro-inflammatory responses. As the substrate of ACE2, AngII overexpresses after S protein-induced ACE2 internalization (76). After binding with angiotensin receptor 1 (AT1R), AngII initiates numerous kinase activations that result in subsequent inflammatory factor production (76).
NLRP3 Inflammasome-Associated Inflammation
Higher numbers of NLRP3 and ASC puncta have been observed in COVID-19 patients, and IL-18 and Caspase-1 p20 levels are correlated with disease severity and clinical outcome (77). Specifically, ion fluxes, protein aggregates, and reactive oxygen species (ROS) are all activators of the NLRP3 inflammasome, which can be induced by viral replication and proliferation (30). For SARS-CoV, the E protein activates the NLRP3 inflammasome by forming an ion channel in host ERGIC/Golgi membranes, and inducing Ca2+ ionic fluxes (78). Similarly, SARS-CoV 3a induces K+ efflux and mitochondrial ROS via ion channel activity (31). ORF8b activates the NLRP3 inflammasome by forming an insoluble complex, as well as direct binding with NLRP3 (79). The E protein, with its unique domain containing three macrodomains (N, M, and C), promotes the production of several cytokines, including IL-1β, TNF-α, IL-6, CXCL10, and C-C motif chemokine ligand (CCL)5, in a NLRP3-dependent manner (78, 80). Such positive feedback loop induced by a pro-inflammatory cytokine eventually results in ARDS, which is a primary contributor to the development of SARS-CoV-induced pulmonary inflammation (30, 81). Moreover, activation of the inflammasome can consequently induce cellular pyroptosis and further aggravate the inflammatory process, leading to extensive tissue inflammation and damage (30). Thus, the NLRP3 inflammasome plays a key role in ARDS and cytokine release syndrome. With the discovery of regulatory proteins for SARS-CoV-2, thorough studies will be conducted to determine the mechanisms governing inflammasome activation.
Potential Therapeutic Targets Based on the Virus-Host Interaction
COVID-19 has rapidly caused a worldwide health crisis, partly because SARS-CoV-2 infects people regardless of age, sex, and race, and no specific antiviral drugs are currently available. Given this situation, effective and specific drugs need to be developed urgently. Innate immune responses in COVID-19 patients can be divided into two phases-early phase (inhibited antiviral innate immunity and low levels of IFNs) and late phase (amplified innate immunity and high levels of IFNs and proinflammatory cytokines), and treatment needs to be administered carefully. Serum IFNs and viral loads need to be detected to determine the suitable timing of treatment administration.
During the early phase, emphasis could be put on inducing IFNs and improving antiviral immunity. In a phase 2 trial for the treatment of SARS-CoV-2 infection, patients receiving SNG001 (nebulized IFN-β1a) exhibited greater improvement and more rapid recovery (82). This phenomenon indicated that local administration of IFN might have different significance from systemic administration. The location, time and duration of IFN exposure may be the key parameters to determine the outcome of viral infection. It also suggested that there may be a substantially greater window (more than 7 days) for beneficial IFN I treatment than initially thought (82). Notably however, in some patients who have neutralizing auto-antibodies against IFNs possibly because of recombination-activating genes (RAG) deficiency, IFNs especially IFN-α administration, is likely to be useless (12). Viral PLpro may cleave proteinaceous post-translational modifications on host proteins as an evasion mechanism against host antiviral immune responses. Given this, GRL-0617 could maintain host IFN responses by targeting SARS-CoV-2 PLpro (52). Similarly, a recent study revealed that famotidine, a histamine receptor-2 blocker, could bind and inhibit the SARS-CoV-2 NSP5 and restore the activation of RIG-I and STAT1 as described above (83). Moreover, in a recent retrospective cohort study, the administration of famotidine in hospitalized COVID-19 patients reduces the risks of severe disease outcomes (84). Currently, the randomized controlled trial of famotidine (NCT04370262) is still underway, whose results are awaited with interest (58).
During the late phase, a primary goal of treatment is to maintain inflammation and antiviral immunity at a moderate level. As mentioned above, the activation of NLRP3 inflammasome during COVID-19 could promote excessive release of inflammatory cytokines. To address this, NLRP3 inflammasome inhibitor (Tranilast) in the treatment of COVID-19 is undergoing clinical trial in China (85). Another potential therapeutic agent is resveratrol which has the ability to suppress NF-κB and inhibit NLRP3 inflammasome activation. Existed studies demonstrated that the administration of resveratrol could inhibit SARS-CoV-2 infection in vitro and ameliorate the pulmonary inflammation and lung injury induced by respiratory viruses in vivo (86, 87). In order to relieve the excessive inflammatory responses, targeted drugs are an extremely important strategy in COVID-19 clinical therapy. Currently, the IL-1 receptor antagonist anakinra, monoclonal antibodies against IFN-γ and IL-6 are all candidates for relieving excess inflammatory responses, and their curative effects in COVID-19 patients were highly anticipated (88, 89).
Conclusion
In this review, we focused on recent advances in interactions between host immune system and SARS-CoV and SARS-CoV-2, with special emphasis on the imbalance of type I IFNs and inflammation caused by viral infection. At present, IFN therapy was suggested as only beneficial at the early stages of SARS-CoV-2 infection, but has little effect on hospitalized patients. Several recent studies have also found that IFN signaling could interfere with lung epithelial repair during recovery from viral infection, thus aggravating the lung injuries (90, 91). In COVID-19, the role, dosage and time of IFN in the treatment are worth further examination. Better understanding of the crosstalk between host and SARS-CoV-2 will help to optimize treatment regimens and explore more potential therapeutic targets for COVID-19.
Author Contributions
JZ drafted the manuscript and figures. CZ and WZ edited the manuscript and figures. All authors contributed to the article and approved the submitted version.
Funding
This work was supported by grants from the National Natural Science Foundation of China (81622030, 31870866, 81861130369, and 81901609). WZ is a Newton Advanced Fellow awarded by the Academy of Medical Sciences (NAF007\1003).
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
- 1. Wang D, Hu B, Hu C, Zhu F, Liu X, Zhang J, et al. Clinical Characteristics of 138 Hospitalized Patients With 2019 Novel Coronavirus-Infected Pneumonia in Wuhan, China. JAMA (2020) 323(11):1061–9. 10.1001/jama.2020.1585 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Yang X, Yu Y, Xu J, Shu H, Xia J, Liu H, et al. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study. Lancet Respir Med (2020) 8(5):475–81. 10.1016/s2213-2600(20)30079-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Zhou F, Yu T, Du R, Fan G, Liu Y, Liu Z, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet (London England) (2020) 395(10229):1054–62. 10.1016/s0140-6736(20)30566-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Vabret N, Britton GJ, Gruber C, Hegde S, Kim J, Kuksin M, et al. Immunology of COVID-19: Current State of the Science. Immunity (2020) 52(6):910–41. 10.1016/j.immuni.2020.05.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Vardhana SA, Wolchok JD. The many faces of the anti-COVID immune response. J Exp Med (2020) 217(6): e20200678. 10.1084/jem.20200678 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Chen N, Zhou M, Dong X, Qu J, Gong F, Han Y, et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet (London England) (2020) 395(10223):507–13. 10.1016/s0140-6736(20)30211-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Mehta P, McAuley DF, Brown M, Sanchez E, Tattersall RS, Manson JJ, et al. COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet (2020) 395(10229):1033–4. 10.1016/s0140-6736(20)30628-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Blanco-Melo D, Nilsson-Payant BE, Liu WC, Uhl S, Hoagland D, Moller R, et al. Imbalanced Host Response to SARS-CoV-2 Drives Development of COVID-19. Cell (2020) 181(5):1036–45 e9. 10.1016/j.cell.2020.04.026 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Hadjadj J, Yatim N, Barnabei L, Corneau A, Boussier J, Smith N, et al. Impaired type I interferon activity and inflammatory responses in severe COVID-19 patients. Sci (New York NY) (2020) 369(6504):718–24. 10.1126/science.abc6027 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Combes A, Courau T, Kuhn N, Hu K, Ray A, Chen W, et al. Global absence and targeting of protective immune states in severe COVID-19. Nature (2021) 591(7848):124–30. 10.1038/s41586-021-03234-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Zhang Q, Bastard P, Liu Z, Le Pen J, Moncada-Velez M, Chen J, et al. Inborn errors of type I IFN immunity in patients with life-threatening COVID-19. Sci (New York NY) (2020) 370(6515):eabd4570. 10.1126/science.abd4570 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Bastard P, Rosen LB, Zhang Q, Michailidis E, Hoffmann H-H, Zhang Y, et al. Autoantibodies against type I IFNs in patients with life-threatening COVID-19. Sci (New York NY) (2020) 370(6515):eabd4585. 10.1126/science.abd4585 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Tay MZ, Poh CM, Renia L, MacAry PA, Ng LFP. The trinity of COVID-19: immunity, inflammation and intervention. Nat Rev Immunol (2020) 20(6):363–74. 10.1038/s41577-020-0311-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Kim D, Lee JY, Yang JS, Kim JW, Kim VN, Chang H. The Architecture of SARS-CoV-2 Transcriptome. Cell (2020) 181(4):914–21 e10. 10.1016/j.cell.2020.04.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Yin WC, Mao CY, Luan XD, Shen DD, Shen QY, Su HX, et al. Structural basis for inhibition of the RNA-dependent RNA polymerase from SARS-CoV-2 by remdesivir. Science (2020) 368(6498):1499–504. 10.1126/science.abc1560 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Lei X, Dong X, Ma R, Wang W, Xiao X, Tian Z, et al. Activation and evasion of type I interferon responses by SARS-CoV-2. Nat Commun (2020) 11(1):3810. 10.1038/s41467-020-17665-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Li M, Li L, Zhang Y, Wang X. Expression of the SARS-CoV-2 cell receptor gene ACE2 in a wide variety of human tissues. Infect Dis Poverty (2020) 9(1):45. 10.1186/s40249-020-00662-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Zhao Y, Zhao ZX, Wang YJ, Zhou YQ, Ma Y, Zuo W. Single-Cell RNA Expression Profiling of ACE2, the Receptor of SARS-CoV-2. Am J Respir Crit Care Med (2020) 202(5):756–9. 10.1164/rccm.202001-0179LE [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Szabo PA, Dogra P, Gray JI, Wells SB, Connors TJ, Weisberg SP, et al. Longitudinal profiling of respiratory and systemic immune responses reveals myeloid cell-driven lung inflammation in severe COVID-19. Immunity (2021) S1074–7613(21)00117–5. 10.1016/j.immuni.2021.03.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Ziegler CGK, Allon SJ, Nyquist SK, Mbano IM, Miao VN, Tzouanas CN, et al. SARS-CoV-2 Receptor ACE2 Is an Interferon-Stimulated Gene in Human Airway Epithelial Cells and Is Detected in Specific Cell Subsets across Tissues. Cell (2020) 181(5):1016–35 e19. 10.1016/j.cell.2020.04.035 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Mu J, Xu J, Zhang L, Shu T, Wu D, Huang M, et al. SARS-CoV-2-encoded nucleocapsid protein acts as a viral suppressor of RNA interference in cells. Sci China Life Sci (2020) 63(9):1–4. 10.1007/s11427-020-1692-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Fahmi M, Kubota Y, Ito M. Nonstructural proteins NS7b and NS8 are likely to be phylogenetically associated with evolution of 2019-nCoV. Infect Genet Evol (2020) 81:104272. 10.1016/j.meegid.2020.104272 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Gordon DE, Jang GM, Bouhaddou M, Xu J, Obernier K, White KM, et al. A SARS-CoV-2 protein interaction map reveals targets for drug repurposing. Nature (2020) 583(7816):459–68. 10.1038/s41586-020-2286-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Schoggins J. Interferon-Stimulated Genes: What Do They All Do? Annu Rev Virol (2019) 6(1):567–84. 10.1146/annurev-virology-092818-015756 [DOI] [PubMed] [Google Scholar]
- 25. Walls AC, Park YJ, Tortorici MA, Wall A, McGuire AT, Veesler D. Structure, Function, and Antigenicity of the SARS-CoV-2 Spike Glycoprotein. Cell (2020) 181(2):281–92 e6. 10.1016/j.cell.2020.02.058 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Hu Y, Li W, Gao T, Cui Y, Jin Y, Li P, et al. The Severe Acute Respiratory Syndrome Coronavirus Nucleocapsid Inhibits Type I Interferon Production by Interfering with TRIM25-Mediated RIG-I Ubiquitination. J Virol (2017) 91(8):e02143–16. 10.1128/JVI.02143-16 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Zhao X, Chu H, Wong B, Chiu M, Wang D, Li C, et al. Activation of C-Type Lectin Receptor and (RIG)-I-Like Receptors Contributes to Proinflammatory Response in Middle East Respiratory Syndrome Coronavirus-Infected Macrophages. J Infect Dis (2020) 221(4):647–59. 10.1093/infdis/jiz483 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Carty M, Guy C, Bowie A. Detection of Viral Infections by Innate Immunity. Biochem Pharmacol (2021) 183:114316. 10.1016/j.bcp.2020.114316 [DOI] [PubMed] [Google Scholar]
- 29. Imai Y, Kuba K, Neely G, Yaghubian-Malhami R, Perkmann T, van Loo G, et al. Identification of oxidative stress and Toll-like receptor 4 signaling as a key pathway of acute lung injury. Cell (2008) 133(2):235–49. 10.1016/j.cell.2008.02.043 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Zhao C, Zhao W. NLRP3 Inflammasome-A Key Player in Antiviral Responses. Front Immunol (2020) 11:211. 10.3389/fimmu.2020.00211 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Chen IY, Moriyama M, Chang MF, Ichinohe T. Severe Acute Respiratory Syndrome Coronavirus Viroporin 3a Activates the NLRP3 Inflammasome. Front Microbiol (2019) 10:50:50. 10.3389/fmicb.2019.00050 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Garcia LF. Immune Response, Inflammation, and the Clinical Spectrum of COVID-19. Front Immunol (2020) 11:1441. 10.3389/fimmu.2020.01441 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Hoagland DA, Møller R, Uhl SA, Oishi K, Frere J, Golynker I, et al. Leveraging the antiviral type-I interferon system as a first line defense against SARS-CoV-2 pathogenicity. Immunity (2021) 54(3):557–70.e5. 10.1016/j.immuni.2021.01.017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Cameron MJ, Ran L, Xu L, Danesh A, Bermejo-Martin JF, Cameron CM, et al. Interferon-mediated immunopathological events are associated with atypical innate and adaptive immune responses in patients with severe acute respiratory syndrome. J Virol (2007) 81(16):8692–706. 10.1128/JVI.00527-07 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Channappanavar R, Fehr AR, Vijay R, Mack M, Zhao J, Meyerholz DK, et al. Dysregulated Type I Interferon and Inflammatory Monocyte-Macrophage Responses Cause Lethal Pneumonia in SARS-CoV-Infected Mice. Cell Host Microbe (2016) 19(2):181–93. 10.1016/j.chom.2016.01.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Lee J, Park S, Jeong H, Ahn J, Choi S, Lee H, et al. Immunophenotyping of COVID-19 and influenza highlights the role of type I interferons in development of severe COVID-19. Sci Immunol (2020) 5(49):eabd1554. 10.1126/sciimmunol.abd1554 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Costela-Ruiz V, Illescas-Montes R, Puerta-Puerta J, Ruiz C, Melguizo-Rodríguez L. SARS-CoV-2 infection: The role of cytokines in COVID-19 disease. Cytokine Growth Factor Rev (2020) 54:62–75. 10.1016/j.cytogfr.2020.06.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Metz P, Dazert E, Ruggieri A, Mazur J, Kaderali L, Kaul A, et al. Identification of type I and type II interferon-induced effectors controlling hepatitis C virus replication. Hepatol (Baltimore Md) (2012) 56(6):2082–93. 10.1002/hep.25908 [DOI] [PubMed] [Google Scholar]
- 39. Mantlo E, Bukreyeva N, Maruyama J, Paessler S, Huang C. Potent Antiviral Activities of Type I Interferons to SARS-CoV-2 Infection. BioRxiv Preprint Server Biol (2020) 2020.04.02.022764. 10.1101/2020.04.02.022764 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Hung IFN, Lung KC, Tso EYK, Liu R, Chung TWH, Chu MY, et al. Triple combination of interferon beta-1b, lopinavir-ritonavir, and ribavirin in the treatment of patients admitted to hospital with COVID-19: an open-label, randomised, phase 2 trial. Lancet (2020) 395(10238):1695–704. 10.1016/s0140-6736(20)31042-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Smits SL, de Lang A, van den Brand JM, Leijten LM, van IWF, Eijkemans MJ, et al. Exacerbated innate host response to SARS-CoV in aged non-human primates. PloS Pathog (2010) 6(2):e1000756. 10.1371/journal.ppat.1000756 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Stout-Delgado HW, Yang X, Walker WE, Tesar BM, Goldstein DR. Aging impairs IFN regulatory factor 7 up-regulation in plasmacytoid dendritic cells during TLR9 activation. J Immunol (2008) 181(10):6747–56. 10.4049/jimmunol.181.10.6747 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Smits SL, van den Brand JM, de Lang A, Leijten LM, van Ijcken WF, van Amerongen G, et al. Distinct severe acute respiratory syndrome coronavirus-induced acute lung injury pathways in two different nonhuman primate species. J Virol (2011) 85(9):4234–45. 10.1128/JVI.02395-10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. van der Made CI, Simons A, Schuurs-Hoeijmakers J, van den Heuvel G, Mantere T, Kersten S, et al. Presence of Genetic Variants Among Young Men With Severe COVID-19. JAMA-J Am Med Assoc (2020) 324(7):663–73. 10.1001/jama.2020.13719 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Galani I, Rovina N, Lampropoulou V, Triantafyllia V, Manioudaki M, Pavlos E, et al. Untuned antiviral immunity in COVID-19 revealed by temporal type I/III interferon patterns and flu comparison. Nat Immunol (2021) 22(1):32–40. 10.1038/s41590-020-00840-x [DOI] [PubMed] [Google Scholar]
- 46. Baas T, Taubenberger JK, Chong PY, Chui P, Katze MG. SARS-CoV virus-host interactions and comparative etiologies of acute respiratory distress syndrome as determined by transcriptional and cytokine profiling of formalin-fixed paraffin-embedded tissues. J Interferon Cytokine Res (2006) 26(5):309–17. 10.1089/jir.2006.26.309 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Stukalov A, Girault V, Grass V, Bergant V, Karayel O, Urban C, et al. Multi-level proteomics reveals host-perturbation strategies of SARS-CoV-2 and SARS-CoV. bioRxiv (2020) 2020:6.17.156455. 10.1101/2020.06.17.156455 [DOI] [Google Scholar]
- 48. Lin J-w, Tang C, Wei H-c, Du B, Chen C, Wang M, et al. Genomic monitoring of SARS-CoV-2 uncovers an Nsp1 deletion variant that modulates type I interferon response. Cell Host Microbe (2021) 29(3):489–502.e8. 10.1016/j.chom.2021.01.015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Wu Y, Ma L, Zhuang Z, Cai S, Zhao Z, Zhou L, et al. Main protease of SARS-CoV-2 serves as a bifunctional molecule in restricting type I interferon antiviral signaling. Signal Transduct Targeted Ther (2020) 5(1):221. 10.1038/s41392-020-00332-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Siu KL, Kok KH, Ng MH, Poon VK, Yuen KY, Zheng BJ, et al. Severe acute respiratory syndrome coronavirus M protein inhibits type I interferon production by impeding the formation of TRAF3.TANK.TBK1/IKKepsilon complex. J Biol Chem (2009) 284(24):16202–9. 10.1074/jbc.M109.008227 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Zheng Y, Zhuang M, Han L, Zhang J, Nan M, Zhan P, et al. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) membrane (M) protein inhibits type I and III interferon production by targeting RIG-I/MDA-5 signaling. Signal Transduct Targeted Ther (2020) 5(1):299. 10.1038/s41392-020-00438-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Shin D, Mukherjee R, Grewe D, Bojkova D, Baek K, Bhattacharya A, et al. Papain-like protease regulates SARS-CoV-2 viral spread and innate immunity. Nature (2020) 587(7835):657–62. 10.1038/s41586-020-2601-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Thoms M, Buschauer R, Ameismeier M, Koepke L, Denk T, Hirschenberger M, et al. Structural basis for translational shutdown and immune evasion by the Nsp1 protein of SARS-CoV-2. Science (2020) 369(6508):1249–52. 10.1126/science.abc8665 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Chen Y, Cai H, Pan J, Xiang N, Tien P, Ahola T, et al. Functional screen reveals SARS coronavirus nonstructural protein nsp14 as a novel cap N7 methyltransferase. Proc Natl Acad Sci United States America (2009) 106(9):3484–9. 10.1073/pnas.0808790106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Shi CS, Qi HY, Boularan C, Huang NN, Abu-Asab M, Shelhamer JH, et al. SARS-coronavirus open reading frame-9b suppresses innate immunity by targeting mitochondria and the MAVS/TRAF3/TRAF6 signalosome. J Immunol (2014) 193(6):3080–9. 10.4049/jimmunol.1303196 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Kopecky-Bromberg SA, Martinez-Sobrido L, Frieman M, Baric RA, Palese P. Severe acute respiratory syndrome coronavirus open reading frame (ORF) 3b, ORF 6, and nucleocapsid proteins function as interferon antagonists. J Virol (2007) 81(2):548–57. 10.1128/JVI.01782-06 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Devaraj SG, Wang N, Chen Z, Chen Z, Tseng M, Barretto N, et al. Regulation of IRF-3-dependent innate immunity by the papain-like protease domain of the severe acute respiratory syndrome coronavirus. J Biol Chem (2007) 282(44):32208–21. 10.1074/jbc.M704870200 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Matsuyama T, Kubli S, Yoshinaga S, Pfeffer K, Mak T. An aberrant STAT pathway is central to COVID-19. Cell Death Differ (2020) 27(12):3209–25. 10.1038/s41418-020-00633-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Frieman M, Yount B, Heise M, Kopecky-Bromberg SA, Palese P, Baric RS. Severe acute respiratory syndrome coronavirus ORF6 antagonizes STAT1 function by sequestering nuclear import factors on the rough endoplasmic reticulum/Golgi membrane. J Virol (2007) 81(18):9812–24. 10.1128/JVI.01012-07 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Minakshi R, Padhan K, Rani M, Khan N, Ahmad F, Jameel S. The SARS Coronavirus 3a protein causes endoplasmic reticulum stress and induces ligand-independent downregulation of the type 1 interferon receptor. PloS One (2009) 4(12):e8342. 10.1371/journal.pone.0008342 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Wathelet MG, Orr M, Frieman MB, Baric RS. Severe acute respiratory syndrome coronavirus evades antiviral signaling: role of nsp1 and rational design of an attenuated strain. J Virol (2007) 81(21):11620–33. 10.1128/JVI.00702-07 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Miorin L, Kehrer T, Sanchez-Aparicio M, Zhang K, Cohen P, Patel R, et al. SARS-CoV-2 Orf6 hijacks Nup98 to block STAT nuclear import and antagonize interferon signaling. Proc Natl Acad Sci United States America (2020) 117(45):28344–54. 10.1073/pnas.2016650117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Cao Z, Xia H, Rajsbaum R, Xia X, Wang H, Shi P. Ubiquitination of SARS-CoV-2 ORF7a promotes antagonism of interferon response. Cell Mol Immunol (2021) 18(3):746–8. 10.1038/s41423-020-00603-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Mu J, Fang Y, Yang Q, Shu T, Wang A, Huang M, et al. SARS-CoV-2 N protein antagonizes type I interferon signaling by suppressing phosphorylation and nuclear translocation of STAT1 and STAT2. Cell Discovery (2020) 6:65. 10.1038/s41421-020-00208-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet (London England) (2020) 395(10223):497–506. 10.1016/s0140-6736(20)30183-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Chen G, Wu D, Guo W, Cao Y, Huang D, Wang H, et al. Clinical and immunological features of severe and moderate coronavirus disease 2019. J Clin Invest (2020) 130(5):2620–9. 10.1172/JCI137244 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Gao YM, Xu G, Wang B, Liu BC. Cytokine storm syndrome in coronavirus disease 2019: A narrative review. J Intern Med (2020) 289(2):147–61. 10.1111/joim.13144 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Grein J, Ohmagari N, Shin D, Diaz G, Asperges E, Castagna A, et al. Compassionate Use of Remdesivir for Patients with Severe Covid-19. N Engl J Med (2020) 382(24):2327–36. 10.1056/NEJMoa2007016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Hao D, He L, Qu J, Pan J, Hu B, Zhang J, et al. [A study of pulmonary inflammatory reaction induced by N-protein of SARS-CoV in rat models and effects of glucocorticoids on it]. Zhonghua nei ke za zhi (2005) 44(12):890–3. 10.3760/j.issn:0578-1426.2005.12.004 [DOI] [PubMed] [Google Scholar]
- 70. Galani V, Tatsaki E, Bai M, Kitsoulis P, Lekka M, Nakos G, et al. The role of apoptosis in the pathophysiology of Acute Respiratory Distress Syndrome (ARDS): an up-to-date cell-specific review. Pathol Res Pract (2010) 206(3):145–50. 10.1016/j.prp.2009.12.002 [DOI] [PubMed] [Google Scholar]
- 71. Imanifooladi A, Yazdani S, Nourani M. The role of nuclear factor-kappaB in inflammatory lung disease. Inflammation Allergy Drug Targets (2010) 9(3):197–205. 10.2174/187152810792231904 [DOI] [PubMed] [Google Scholar]
- 72. Dosch S, Mahajan S, Collins A. SARS coronavirus spike protein-induced innate immune response occurs via activation of the NF-kappaB pathway in human monocyte macrophages in vitro. Virus Res (2009) 142:19–27. 10.1016/j.virusres.2009.01.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Liao QJ, Ye LB, Timani KA, Zeng YC, She YL, Ye L, et al. Activation of NF-kappa B by the full-length nucleocapsid protein of the SARS coronavirus. Acta Biochim Et Biophys Sin (2005) 37(9):607–12. 10.1111/j.1745-7270.2005.00082.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. DeDiego ML, Nieto-Torres JL, Regla-Nava JA, Jimenez-Guardeno JM, Fernandez-Delgado R, Fett C, et al. Inhibition of NF-kappaB-mediated inflammation in severe acute respiratory syndrome coronavirus-infected mice increases survival. J Virol (2014) 88(2):913–24. 10.1128/JVI.02576-13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Kanzawa N, Nishigaki K, Hayashi T, Ishii Y, Furukawa S, Niiro A, et al. Augmentation of chemokine production by severe acute respiratory syndrome coronavirus 3a/X1 and 7a/X4 proteins through NF-kappaB activation. FEBS Lett (2006) 580(30):6807–12. 10.1016/j.febslet.2006.11.046 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Higuchi S, Ohtsu H, Suzuki H, Shirai H, Frank GD, Eguchi S. Angiotensin II signal transduction through the AT(I) receptor: novel insig hts into mechanisms and pathophysiology. Clin Sci (2007) 112(7-8):417–28. 10.1042/cs20060342 [DOI] [PubMed] [Google Scholar]
- 77. Rodrigues T, de Sá K, Ishimoto A, Becerra A, Oliveira S, Almeida L, et al. Inflammasomes are activated in response to SARS-CoV-2 infection and are associated with COVID-19 severity in patients. J Exp Med (2021) 218(3):e20201707. 10.1084/jem.20201707 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78. Nieto-Torres JL, Verdia-Baguena C, Jimenez-Guardeno JM, Regla-Nava JA, Castano-Rodriguez C, Fernandez-Delgado R, et al. Severe acute respiratory syndrome coronavirus E protein transports calcium ions and activates the NLRP3 inflammasome. Virology (2015) 485:330–9. 10.1016/j.virol.2015.08.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79. Shi CS, Nabar NR, Huang NN, Kehrl JH. SARS-Coronavirus Open Reading Frame-8b triggers intracellular stress pathways and activates NLRP3 inflammasomes. Cell Death Discovery (2019) 5:12. 10.1038/s41420-019-0181-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80. Chang YS, Ko BH, Ju JC, Chang HH, Huang SH, Lin CW. SARS Unique Domain (SUD) of Severe Acute Respiratory Syndrome Coronavirus Induces NLRP3 Inflammasome-Dependent CXCL10-Mediated Pulmonary Inflammation. Int J Mol Sci (2020) 21(9):3179. 10.3390/ijms21093179 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81. DeDiego ML, Nieto-Torres JL, Jimenez-Guardeno JM, Regla-Nava JA, Castano-Rodriguez C, Fernandez-Delgado R, et al. Coronavirus virulence genes with main focus on SARS-CoV envelope gene. Virus Res (2014) 194:124–37. 10.1016/j.virusres.2014.07.024 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82. Monk P, Marsden R, Tear V, Brookes J, Batten T, Mankowski M, et al. Safety and efficacy of inhaled nebulised interferon beta-1a (SNG001) for treatment of SARS-CoV-2 infection: a randomised, double-blind, placebo-controlled, phase 2 trial. Lancet Respir Med (2020) 9(2):196–206. 10.1016/s2213-2600(20)30511-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83. Wu C, Liu Y, Yang Y, Zhang P, Zhong W, Wang Y, et al. Analysis of therapeutic targets for SARS-CoV-2 and discovery of potential drugs by computational methods. Acta Pharm Sin B (2020) 10(5):766–88. 10.1016/j.apsb.2020.02.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84. Freedberg D, Conigliaro J, Wang T, Tracey K, Callahan M, Abrams J. Famotidine Use Is Associated With Improved Clinical Outcomes in Hospitalized COVID-19 Patients: A Propensity Score Matched Retrospective Cohort Study. Gastroenterology (2020) 159(3):1129–31.e3. 10.1053/j.gastro.2020.05.053 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85. van den Berg D, Te Velde A. Severe COVID-19: NLRP3 Inflammasome Dysregulated. Front Immunol (2020) 11:1580. 10.3389/fimmu.2020.01580 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86. Yang M, Wei J, Huang T, Lei L, Shen C, Lai J, et al. Resveratrol inhibits the replication of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in cultured Vero cells. Phytother Res PTR (2020) 35(3):1127–9. 10.1002/ptr.6916 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87. Filardo S, Di Pietro M, Mastromarino P, Sessa R. Therapeutic potential of resveratrol against emerging respiratory viral infections. Pharmacol Ther (2020) 214:107613. 10.1016/j.pharmthera.2020.107613 [DOI] [PubMed] [Google Scholar]
- 88. van de Veerdonk FL, Netea MG. Blocking IL-1 to prevent respiratory failure in COVID-19. Crit Care (2020) 24(1):445. 10.1186/s13054-020-03166-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89. Wiersinga W, Rhodes A, Cheng A, Peacock S, Prescott H. Pathophysiology, Transmission, Diagnosis, and Treatment of Coronavirus Disease 2019 (COVID-19): A Review. JAMA (2020) 324(8):782–93. 10.1001/jama.2020.12839 [DOI] [PubMed] [Google Scholar]
- 90. Major J, Crotta S, Llorian M, McCabe T, Gad H, Priestnall S, et al. Type I and III interferons disrupt lung epithelial repair during recovery from viral infection. Sci (New York NY) (2020) 369(6504):712–7. 10.1126/science.abc2061 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91. Broggi A, Ghosh S, Sposito B, Spreafico R, Balzarini F, Lo Cascio A, et al. Type III interferons disrupt the lung epithelial barrier upon viral recognition. Sci (New York NY) (2020) 369(6504):706–12. 10.1126/science.abc3545 [DOI] [PMC free article] [PubMed] [Google Scholar]