Abstract
Purpose of Review
Synovial inflammation is characteristic of inflammatory chronic arthropathies and can cause progressive articular damage, chronic pain, and functional loss. Scientific research has increasingly focused on investigating anti-inflammatory micronutrients present in fruits, vegetables, spices, seeds, tea, and wine. This review aims to examine the anti-inflammatory effect of polyphenols (phytonutrients present in plants) and other micronutrients described in randomized clinical trials conducted in patients with chronic inflammatory arthropathies.
Recent Findings
There is an increasing evidence that differences in micronutrient intake might play an essential role in pathogenesis, therapeutic response, and remission of synovitis. Randomized clinical trials with specific micronutrient- or nutrient-enriched food intake show improvement of symptoms and modulation of both pro- and anti-inflammatory mediators.
Summary
We found convincing evidence of the anti-inflammatory effect of several micronutrients in arthritis symptoms and inflammation. Although in clinical practice nutritional recommendations to patients with chronic joint inflammation are not consistently prescribed, the addition of these nutrients to day-to-day eating habits could potentially change the natural history of inflammatory arthritis. Future research is needed for a consensus on the specific nutritional recommendations for patients with chronic synovial inflammation.
Keywords: Anti-inflammatory, Nutrients, Polyphenols, Clinical trials, Arthritis, Synovitis
Introduction
Rheumatoid arthritis (RA), osteoarthritis (OA), and psoriatic arthritis (PsA) are among the most common chronic joint inflammatory diseases worldwide. Approximately 54.4 million adults are living with doctor-diagnosed arthritis and this remains a leading cause of disability. Chronic synovial inflammation will cause progressive articular damage, chronic pain, functional loss, and reduced quality of life [1]. The origin of chronic synovial inflammation depends on different factors such as genetics, sex, immune and innate response, structural factors, and environmental factors like weight, nutrition, microbiome, and tobacco use that leads to a pro-inflammatory/anti-inflammatory disbalance of local mediators [2]. The vast majority of pro-inflammatory cytokines within the joints (TNF, IL-6, and IL-8 in RA, and TNF, IL-17, and IL-23 in PsA among others) will stimulate the production of degrading enzymes, causing severe cartilage degeneration and bone damage, leading to joint destruction [3, 4]. In OA, although progressive degeneration of cartilage leading to permanent functional joint failure is a critical feature, increasing evidence indicates that low-grade synovial inflammation (synovitis) contributes to OA progression [5]. In addition, a dysregulated oxidative metabolism, which generates high amounts of reactive oxygen species (ROS), together with an insufficient antioxidant system, also plays a role in the pathophysiology of rheumatic diseases [6]. Anti-inflammatory and antioxidant therapeutic options are of great importance in these diseases.
Patients with rheumatic diseases generally trust their rheumatologists for counseling. They frequently seek additional sources of relief and/or treatments with fewer side effects to improve joint symptoms and disease control. They specifically ask about dietary recommendations as many perceive changes in symptoms after consumption or avoidance of specific foods. In our experience working with RA patients, we have noticed that they are very motivated and eager to modify their diet. Our team interviewed different groups of RA patients, and their feedback was assessed to build a diet that incorporates suggested anti-inflammatory ingredients and that was easily adopted by patients. We were able to identify common foods that were incompatible with an anti-inflammatory diet and substituted them appropriately to match patients’ cultural and economic statuses. This diet, which is an omnivorous diet based on a Mediterranean diet (MD), with some modifications, was assessed in a pilot trial with high adherence and feasibility, which highlights the willingness to change habits in this population and the importance in nutrition advice in our rheumatological clinics [7].
Dietary components (i.e., food components or microorganisms within food) may compromise the intestinal barrier and enter the bloodstream, triggering inflammation. However, even though dietary factors were found to be the single leading cause of death and disability according to the 2019 Global Burden of Disease Study [8], and even after an increase of interest in the influence of diet in other inflammatory diseases, such as cardiovascular diseases, by the scientific community, dietary interventions have not been established as a complementary treatment in RA, OA, or PsA. Furthermore, the data on dietary or nutraceutical interventions are extremely limited in PsA, apparent in a recent systematic literature review [9, 10]. In PsA, studies have primarily focused on weight loss, which has been found to be beneficial [11–13].
Diet modifications can directly change the immune response at both the gastrointestinal and systemic levels by providing an extensive range of nutrients and micronutrients. Furthermore, diet shapes the human gut microbiota, which is recognized to have a central role in the modulation of the immune response and arthritis pathogenesis [14]. There are many ways in which the diet may be altered in order to achieve an improvement in diseases. It is known that losing weight or different patterns or diets, like the Mediterranean and plant-based diets (vegetarian or vegan), can play an essential role in the management of rheumatic diseases. Plant-based diets (fruits, vegetables, and whole grains) have shown many anti-inflammatory and antioxidant properties and were shown to be efficacious in RA and OA patients by reducing pain and disability. These diets are rich in vitamins, minerals (selenium, zinc, magnesium), and polyphenols that are present in fruits, vegetables, legumes, and whole grains [14–18]. Obesity is one of the most important modifiable risk factors to improve outcome in PsA and also OA and there is strong evidence recommending dietary weight reduction with a hypocaloric diet in overweight and obese patients with PsA and OA, but diet may have a role beyond weight control [19–21]. However, diet modifications are often challenging to follow as these diets involve a change of lifestyle. In general, it may be more feasible to simply add anti-inflammatory nutrients [14, 15, 22].
This review aims to present the anti-inflammatory and antioxidant properties of some of the micronutrients and their effect in arthritis symptoms and in downstream cytokines and chemokines from randomized clinical trials (Fig. 1, Tables 1, 2, 3, and 4). We will focus on the effects of these micronutrients on clinical scores, as well as the biological effects in patients and animal models. A summary of the effect on inflammatory mediators from in vitro studies can be found in Table 5.
Fig. 1.
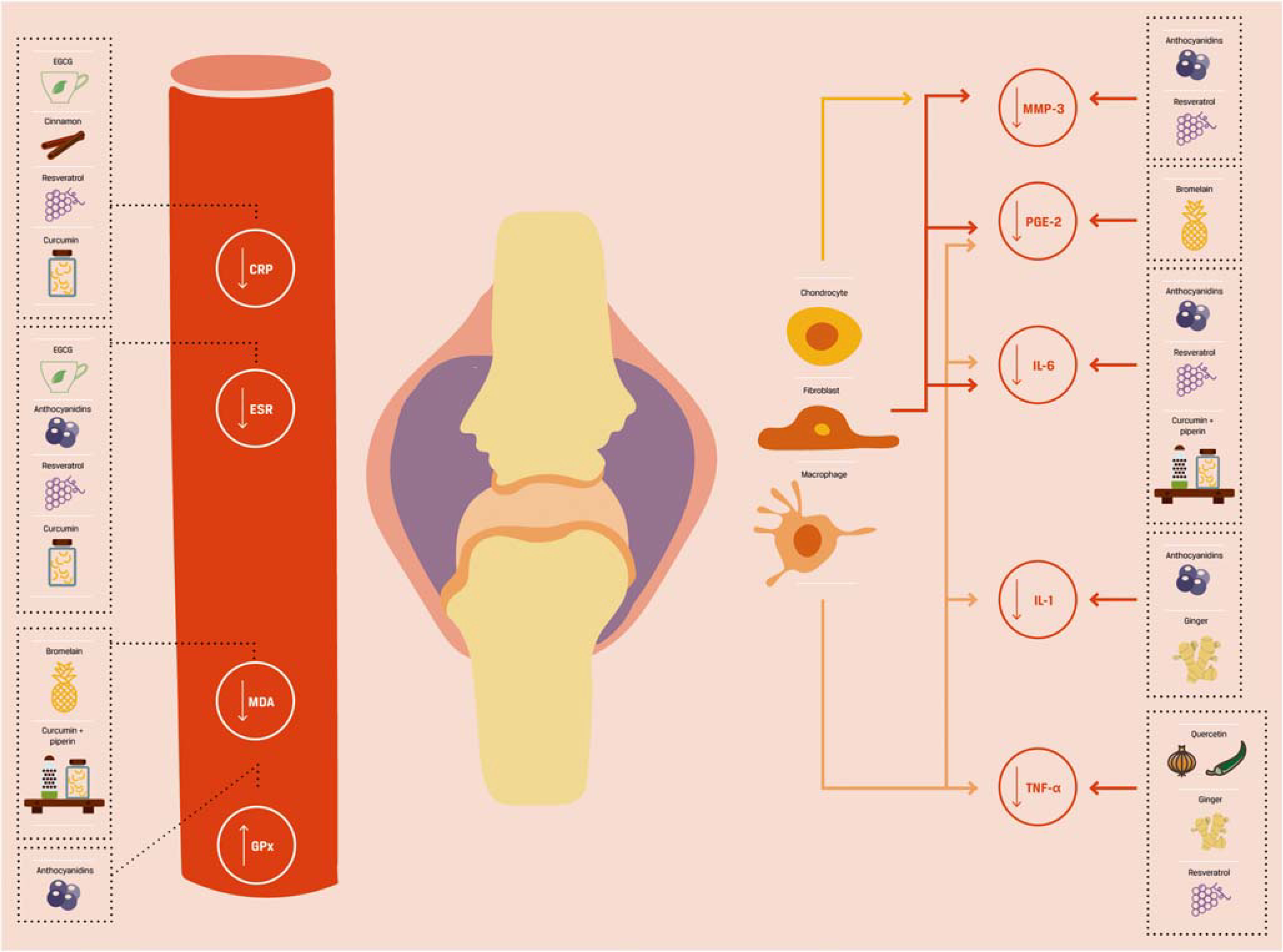
Modulation of pro-inflammatory markers in serum in randomized clinical trials with patients with chronic arthritis
Table 1.
Polyphenols existing in different types of food
| Micronutrients |
Food | ||
|---|---|---|---|
| Polyphenols | Subgroup | ||
| Flavonoids | Anthocyanidins | Cyanidin, delphinidin, malvidin, peonidin, petunidin | Berry fruits (strawberry, raspberry, blackberry, cranberry, blueberry, and açaí berry), black carrot, black soybean, pomegranate, purple corn, purple sweet potato, red cabbage |
| Flavan-3-ols | Catechin, epicatechin, epigallocatechin, epigallocatechin 3-gallate (EGCG) | Apple skin, cacao, carob powder, green tea, other teas, hazelnuts, onions, pecans, plums | |
| Flavanones | Hesperidin | Amaranth, citric fruits (oranges, lemons, mandarins, limes, bergamot), grapefruit, quinoa |
|
| Flavones | Apigenin, luteolin | Amaranth, artichoke, celery, chamomile, kiwi, lettuce, parsley, peppermint, quinoa, spinach, tea (rooibos, green, black, and oolong) | |
| Flavonols | Myricetin, morin, quercetin | Amaranth, chia seeds, cocoa, figs, green tea, quinoa | |
| Isoflavones | Alfalfa, amaranth, legumes (particularly soybean, roasted soybean, soy flour, tofu, miso, and tempeh but also cheakpeas and beans), quinoa, red clover, white clover | ||
| Non-flavonoids | Lignans | Apricots, broccoli, brussel sprouts, outer layers of cereal grains, flaxseed, kale, nuts (cashew nuts), sesame seed | |
| Phenolic acids | Cinnamic acids | Amaranth, cinnamon, quinoa | |
| Gallic acid (benzoic acid) | Amaranth, fruits (as pomegranates, black raspberries, raspberries, strawberries), nuts (as walnuts and almonds), quinoa, seeds |
||
| Vanillic acid (benzoic acid) | Amaranth, berries, Ginkgo biloba leaf extract, honey, quinoa, vanilla | ||
| Polyphenolic amides | Avenanthramides | Oats | |
| Capsaicin | Green and red and chili pepper | ||
| Piperine | Black pepper | ||
| Proteolytic enzymes | Bromelain | Kiwi, papaya, pineapple | |
| Mangiferin | Mango, papaya | ||
| Papain | Papaya | ||
| Others | Curcumin | Turmeric | |
| Ellagic acid | Fruits (pomegranate, guava, berry fruits as black berries, black raspberries, cloudberries, strawberries), nuts (pecans, walnuts) | ||
| Gingerol | Ginger | ||
| Rosmarinic acid | Rosemary | ||
| Ursolic acid | Peel of apples and other fruits and vegetables, as well as in herbs and spices like rosemary, basil and thyme | ||
| Minerals | |||
| Magnesium | Green leafy vegetables (spinach), some legumes (beans and peas), nuts, seeds, and whole grains | ||
| Selenium | Amaranth, bread, eggs, fish, grains, meat, poultry, quinoa, seafood | ||
| Zinc | Beans, fortified breakfast cereals, dairy products, whole grains, nuts, oysters, red meat, seafood (crab and lobster) | ||
Table 2.
Clinical trials in arthritis (flavonoids; non-flavonoids)
| Flavonoids | |||||
|---|---|---|---|---|---|
| Compound | Control | Duration | Patients | Pathology | Biological-functional outcomes |
| Blueberry juice [23] (50 rnl/day) | Etanercept 50 mg 2 times/week ± blueberry juice | 6 months | 200 ♀♂ < 16 y | J1A | ↓IL1 a and B; ↑ILlRA ↑ACR 20, 50, 70; ↓symptoms; ↓side effects |
| Green tea [24] (500 mg/8 h) | Diclofenac 50 mg/12 h | 4 weeks | 50 ♀♂ 40–75 y | KOA | ↓VAS pain; ↓total WOMAC; ↓WOMAC physical function; less adverse effects |
| Green tea [25] (4 6 cups/day) | Exercise and/or infliximab | 6 months | 120 ♀♂ 60.7 ± 2.53 y | RA | ↓DAS28-ESR; ↓ESR; ↓CRP; ↓DPD; ↓NTX; ↓BSAP |
| Quercetin [26] (500 mg/day) | Placebo | 8 weeks | 50 ♀ 19–70 y | RA | ↓ hs-TNF-α; ↓EMS, morning and after-activity pain; ↓DAS-28; ↓HAQ |
| Quercetin [27] (45 mg) + glucosamine (1200 mg) + chondroitin sulfate (60 mg)/day | Placebo | 4 months | 40 ♀♂ 40–85 y | KOA | ↓urinary CTX-II; ↓serum CPII; ↓VAS; ↓Japanese Orthopaedic Association pain criteria |
| Quercetin [28] (166 mg) + vitamin C (133 mg)/8 h | Lipoic acid (300 mg) or placebo | 4 weeks | 20 ♀♂ 52.1 ± 10.3 y | RA | →TNF-α; →CRP; →IL-1; →IL-6; →VAS |
| Strawberries [29] (50 g/day) | Placebo | 26 weeks | 17 ♀♂ 57±7 y | Knee OA Obese | ↓IL-6; ↓IL-1P; ↓MMP-3 ↓constant pain; ↓intermittent pain; ↓total pain |
| Non-flavonoids | |||||
| Bromelain [30] (500 mg/day) | Diclofenac 100 mg/day | 4–16 weeks | 40 ♀♂ ≥ 50 y | Knee OA (mild-moderate) | ↓LPS-induced PGE2; ↓MDA ↓WOMAC pain; ↓WOMAC stiffness; ↓WOMAC physical function |
| Cinnamon [31] (2 g/day) | Placebo | 8 weeks | 32 ♀ > 18 y | RA | ↓CRP; ↓TNF-α; →FBS; →lipid profile; →liver enzynres; →ESR ↓DAS-28; ↓VAS; ↓TJC; ↓SJC; ↓DBP |
| Curcumin [32] (333 mg/8 h) | Placebo | 12 weeks | 201 ♀♂ 40–70 y | OA | ↓WOMAC pain; ↓WOMAC physical function |
| Curcumin [33] (1,5 g/day) | Ibuprofen 1.2 g/day | 4 weeks | 367 ♀♂ ≥ 50 y | Knee OA | → WOMAC total; → WOMAC pain; →WOMAC function |
| Curcumin [34] (500 mg/8 h) | Diclofenac 50 mg/12 h | 28 days | 139 ♀♂ 38–65 y | Knee OA | →VAS; →KOOS; ↓weight-lowering effect; ↓anti-ulcer effect |
| Curcumin [35] (250–500 mg/12 h) | Placebo | 3 months | 36 ♀♂ 22–55 y | RA | ↓ ESR; ↓ CPR; ↓ RF ↓ VAS; ↓ DAS-28; ↓ ACR response; ↓ adverse effects |
| Curcumin [37] (90 mg/day) | Diclofenac 75 mg/day | 4 weeks | 80 patients | Knee OA | ↓ COX-2 joint fluid |
| Curcumin with piperine [38, 39] (500 mg/8 h + piperine 15 mg/day) | Placebo | 6 weeks | 40 patients 57±9 y | Knee OA (mild-moderate) | ↑SOD; ↑GSH; ↓MDA; ↓IL-4; ↓IL-6; ↓hs-CRP; →TNF-α; →TGF-β; →ESR |
| Ginger [40] (25 mg ginger + 5 mg Echinacea/day) | Placebo | 30 days | 15 ♀♂ 67.2 ± 7.9 y | Knee OA (NSAIDs poor responder) | ↓Lysholm scale score; ↓SF-36; ↓decrease knee circumference |
| Ginger [41] (500 mg/day) | Placebo | 3 months | 120 ♀♂ 50–75 y | Knee OA | ↓TNF-α; ↓IL-β |
| Guava [42] (1 g/day) | Placebo | 3 months | 53 ♀♂ 45–70 y | Knee pain without pain treatment | ↓JKOM pain; ↓JKOM stiffness; ↓VAS |
| Mango [43] (900 mg/day with MTX) | MTX 12.5 mg/w + NSAIDs and/or prednisone (5–10 mg/day) |
6 months | 20 ♀♂ 18–65 y | RA | →ESR ↓DAS28; ↓NSAIDs intake; ↓GI side effects |
| Pomegranate [44] (500 mg/day) | 500 mg/day cellulose | 8 weeks | 55 ♀♂ >40 y | RA | ↓ESR; ↓GPx; →MMP3; → CRP; →MDA levels ↓DAS-28; ↓HAQ score; ↓morning stiffness |
| Resveratrol [45] (1 g/day with their treatment) | Current treatment | 3 months | 100 ♀♂ 44.2 ± 16.4 y | RA | ↓CRP;↓ESR;↓UcOC;↓MMP-3; ↓TNF-α;↓IL-6 ↓DAS28-ESR; ↓SJC-28; ↓TJC- 28 |
Rosmarinic acid [46]
→, no significant difference; y, years old; KOA, knee osteoarthritis; VAS, visual analogue scale; WOMAC, Western Ontario and McMaster Universities Arthritis Index; RA, rheumatoid arthritis; DAS28-ESR, Disease Activity Score-28 for Rheumatoid Arthritis with ESR; ESR, erythrocyte sedimentation rate; CRP, C-reactive protein; DPD, deoxypyridinoline; NTX, amino-terminal telopeptide of type 1 collagen; BSAP, bone alkaline phosphatase; MTX, methotrexate; TNF-α, tumor necrosis factor alpha; EMS, early morning stiffness; DAS-28, Disease Activity Score-28 for Rheumatoid Arthritis; HAQ, health assessment questionnaire; CTX-II, C-telopeptide of type II collagen; CPII, type II procollagen; IL, interleukin; JIA, juvenile idiopathic arthritis; IL1RA, interleukin-1 receptor antagonist; ACR 20, 50, 70, American College of Rheumatology 20, 50, 70; OA, osteoarthritis; MMP-3, Matrix metallopeptidase 3; GPx, glutathione peroxidase; MDA, malondialdehyde; FBS, fasting blood sugar; TJC, tender joints counts; SJC, swollen joints counts; DBS, diastolic blood pressure; UcOC, undercarboxylated osteocalcin; GI, gastrointestinal; OA, osteoarthritis; LPS-induced PGE2, lipopolysaccharide-induced prostaglandin E2; KOOS, Knee Injury and Osteoarthritis Outcome Score; SRP, scaling and root planning; SOD, superoxide dismutase; GSH, glutathione hormone; TGF β, transforming growth factor β; COX-2, cyclooxygenase 2; SF-36, Short Form (36) Health Survey; 6MWT, 6-min walk test; JKOM, Japanese Knee Osteoarthritis Measure
Table 3.
Cytokines, chemokines, and pathways affected by nutrients in animal models of chronic arthritis
| Physiopathology | ||||
|---|---|---|---|---|
| IL-β | Quercetin (5–25-50 mg/day) | CIA mice | Decreases | [47, 48] |
| IL-4 | Quercetin (50 mg/kg/day) | CFA rats | Decreases | [49] |
| IL-6 | Pomegranate (13.6–34 mg/kg/day) | Arthritis mice | Decreases | [50] |
| IL-17 | Quercetin (5–25-50 mg/day) | CIA rats | Decreases | [47, 48] |
| COX-2 | Isorhamnetin (10–20 mg/kg/day) | MIA rats | Decreases | [51] |
| TNF-α | Quercetin (5–25-50 mg/day) | CIA rats | Decreases | [47, 48] |
| Hesperidin (50 mg/kg) | RA rats | Decreases | [52] | |
| Cinnamon (1 –2-4 mg/kg/day) | Arthritis rats | Decreases | [53] | |
| NF-κB | Pomegranate (13.6–34 mg/kg/day) | Arthritis mice | Decreases | [50] |
| iNOS | Isorhamnetin (10–20 mg/kg/day) | MIA rats | Decreases | [51] |
| NO | Hesperidin (160 mg/kg) | RA rats | Decreases | [54] |
| Pomegranate (13.6–34 mg/kg/day) | Arthritis mice | Decreases | [50] | |
| Isorhamnetin (10–20 mg/kg/day) | MIA rats | Decreases | [51] | |
| IFN-γ | Quercetin (50 mg/kg/day) | CFA rats | Decreases | [49] |
| PGE2 | Isorhamnetin (10–20 mg/kg/day) | MIA rats | Decreases | [51] |
| MPO | Quercetin (50 mg/kg/day) | CFA rats | Decreases | [49] |
| MAPK | Pomegranate (13.6–34 mg/kg/day) | CIA mice | Decreases | [50] |
| CTX-II | Spinach (250–500 mg/kg/day) | OA rat | Decreases | [55] |
| Isorhamnetin (10–20 mg/kg/day) | MIA rats | Decreases | [51] | |
| COMP | Spinach (250–500 mg/kg/day) | OA rat | Decreases | [55] |
| Isorhamnetin (10–20 mg/kg/day) | MIA rats | Decreases | [51] | |
| Elastase activity | Hesperidin (50 mg/kg) | RA rats | Decreases | [52] |
| Hesperidin (160 mg/kg) | RA rats | Decreases | [54] | |
| MDA | Hesperidin (50 mg/kg) | RA rats | Decreases | [52] |
| SOD | Hesperidin (160 mg/kg) | RA rats | Increases | [54] |
| Catalase | Hesperidin (160 mg/kg) | RA rats | Increases | [54] |
| GR | Hesperidin (160 mg/kg) | RA rats | Increases | [54] |
| GST | Spinach (250–500 mg/kg/day) | OA rat | Decreases | [55] |
IL, interleukin; CIA, collagen-induced arthritis; CFA, complete Freund’s adjuvant; COX-2, cyclooxygenase 2; MIA, monoiodoacetate-induced arthritis; TNF-α, tumor necrosis factor alpha; RA, rheumatoid arthritis; NF-κB, nuclear factor-kappa beta; iNOS, inducible nitric oxide synthase; NO, nitric oxide; IFN-γ, interferon gamma; PGE2, prostaglandin E2; MPO, myeloperoxidase; MAPK, mitogen-activated protein kinase; CTX-II, C-telopeptide of type II collagen; COMP, cartilage oligomeric matrix protein; OA, osteoarthritis; SOD, superoxide dismutase; GR, glutathione reductase; GST, glutathione S-transferase
Table 4.
Cytokines, chemokines, and pathways affected by nutrients in randomized clinical trials with chronic arthritis
| Physiopathology | ||||
|---|---|---|---|---|
| Cytokine (pathway) | Compound | Model | Action | References |
| hs-CRP | Green tea (4–6 cups/day) | RCT (120 RA, 6 months) | Decreases | [25] |
| Cinnamon (2 g/day) | RCT (32 RA, 8 weeks) | Decreases | [31] | |
| ESR | Green tea (4–6 cups/day) | RCT (120 RA, 6 months) | Decreases | [25] |
| Pomegranate (500 mg/day) | RCT (55 RA, 8 weeks) | Decreases | [44] | |
| IL1-α | Blueberries (50 mL/day) | RCT (200 JIA, 6 months) | Decreases | [23] |
| IL-1β | Blueberries (50 mL/day) | RCT (200 JIA, 6 months) | Decreases | [23] |
| IL-1 RA | Blueberries (50 mL/day) | RCT (200 JIA, 6 months) | Increases | [23] |
| TNF-α | Cinnamon (2 g/day) | RCT (32 RA, 8 weeks) | Decreases | [31] |
| DPD | Green tea (4–6 cups/day) | RCT (120 RA, 6 months) | Decreases | [25] |
| NTX | Green tea (4–6 cups/day) | RCT (120 RA, 6 months) | Decreases | [25] |
| BAP | Green tea (4–6 cups/day) | RCT (120 RA, 6 months) | Decreases | [25] |
| GPx | Pomegranate (500 mg/day) | RCT (55 RA, 8 weeks) | Increases | [44] |
hs-CRP, high-sensitivity C-reactive protein; RCT, randomized clinical trial; RA, rheumatoid arthritis; ESR, erythrocyte sedimentation rate; IL, interleukin; JIA, juvenile idiopathic arthritis; IL-1 RA, interleukin-1 receptor antagonist; TNF-α, tumor necrosis factor alpha; DPD, deoxypyridinoline; NTX, amino-terminal telopeptide of type 1 collagen; BAP, bone alkaline phosphatase; GPx, glutathione peroxidase.
Table 5.
Cytokines, chemokines, and pathways affected by nutrients in vitro
| Physiopathology | |||||
|---|---|---|---|---|---|
| Cytokine (pathway) | Compound | Cell type | Action | References | |
| IL1-α | Phytoestrogens (coumestrol + biochanin A) | Chondrocytes | Rat | Decreases | [56, 57] |
| IL-1β | Cyanidin | Chondrocytes | Human | Decreases | [58] |
| Genistein | OA Chondrocytes | Human | Decreases | [59] | |
| Green tea | RA synovium tissue | Human | Inhibits | [60] | |
| Hesperidin | Chondrocytes | Rat | Decreases | [61] | |
| Kaempferitrin | Fibroblast-like synoviocytes | Human | Inhibits | [48] | |
| Phytoestrogens (coumestrol + biochanin A) |
Chondrocytes | Rat | Decreases | [56, 57] | |
| Vanillic acid | OA Chondrocyte | Rat | Decreases | [62] | |
| IL-2 | Phytoestrogens (coumestrol + biochanin A) | Chondrocytes | Rat | Decreases | [56, 57] |
| IL-6 | Cinnamon | Colonic tissues | Rat | Decreases | [63] |
| Cyanidin | OA chondrocytes | Human | Decreases | [58] | |
| Green tea | RA synovium tissue | Human | Decreases | [60] | |
| Green tea | OA chondrocytes | Human | Decreases | [64] | |
| Kaempferitrin | Fibroblast-like synoviocytes | Human | Decreases | [48] | |
| Phytoestrogens (coumestrol + biochanin A) |
Chondrocytes | Rat | Decreases | [56, 57] | |
| Spinach | Vascular endothelial cells | Human | Decreases | [65] | |
| IL-8 | Green tea | RA synovium tissue | Human | Decreases | [60] |
| IL-17 | Curcumin | CD4(+) T cells | Human | Decreases | [66] |
| MCP-1 | Spinach | Vascular endothelial cells | Decreases | [65] | |
| MMPs | Genistein | OA chondrocytes | Human | Decreases | [59] |
| Vanillic acid | OA chondrocyte | Rat | Decreases | [62] | |
| MMP-1 | Kaempferitrin | Fibroblast-like synoviocytes | Human | Decreases | [48] |
| Phytoestrogens (coumestrol + biochanin A) |
Chondrocytes | Rat | Decreases | [56, 57] | |
| Green tea | RA synovium tissue | Human | Decreases | [48] | |
| MMP-2 | Hesperidin | RA fibroblast-like synovial cells | Mice | Decreases | [67] |
| MMP-3 | Hesperidin | RA fibroblast-like synovial cells | Mice | Decreases | [67] |
| Kaempferitrin | Fibroblast-like synoviocytes | Human | Decreases | [48] | |
| Phytoestrogens (coumestrol + biochanin A) |
Chondrocytes | Rat | Decreases | [56, 57] | |
| MMP-9 | Green tea | OA chondrocytes | Human | Decreases | [64] |
| MMP-13 | Cyanidin | OA chondrocytes | Human | Decreases | [58] |
| Hesperidin | OA chondrocytes | Rat | Decreases | [61] | |
| Hesperidin | RA fibroblast-like synovial cells | Mice | Decreases | [67] | |
| Phytoestrogens (coumestrol + biochanin A) |
Chondrocytes | Rat | Decreases | [56, 57] | |
| Phytoestrogens (coumestrol + biochanin A) |
Chondrocytes | Rat | Decreases | [56, 57] | |
| ADAMTS5 | Cyanidin | OA chondrocytes | Human | Decreases | [58] |
| Green tea | RA synovium tissue | Human | Decreases | [60] | |
| Vanillic acid | OA chondrocyte | Rat | Decreases | [62] | |
| COX-2 | Genistein | OA chondrocytes | Human | Decreases | [59] |
| Kaempferitrin | Fibroblast-like synoviocytes | Human | Decreases | [48] | |
| Phytoestrogens (coumestrol + biochanin A) |
Chondrocytes | Rat | Decreases | [56, 57] | |
| Vanillic acid | OA chondrocyte | Rat | Decreases | [62] | |
| Green tea | RA synovium tissue | Human | Decreases | [60] | |
| Hesperidin | OA chondrocytes | Rat | Decreases | [61] | |
| Myricetin (chia seed) | Osteoblastic cell | Human | Inhibits | [68] | |
| Phytoestrogens (coumestrol + biochanin A) |
Chondrocytes | Rat | Decreases | [56, 57] | |
| NF-κB | Anthocyanidin | Collagen-induced arthritis murine model and human PBMC |
Mice-human | Decreases | [69] |
| Cyanidin | OA chondrocytes | Human | Decreases | [58] | |
| Hesperidin | RA fibroblast-like synovial cells | Mice | Decreases | [67] | |
| Phytoestrogens (coumestrol + biochanin A) Spinach |
Chondrocytes Vascular endothelial cells | Rat | Decreases Decreases |
[56, 57] [65] | |
| Vanillic acid | OA chondrocyte | Rat | Decreases | [62] | |
| CD206 | Spinach | Vascular endothelial cells | Human | Decreases | [65] |
| iNOS | Cyanidin | OA chondrocytes | Human | Decreases | [58] |
| Genistein | OA chondrocytes | Human | Decreases | [59] | |
| Hesperidin | OA chondrocytes | Rat | Decreases | [61] | |
| Hesperidin | RA fibroblast-like synovial cells | Mice | Decreases | [67] | |
| Phytoestrogens (coumestrol + biochanin A) |
Chondrocytes | Rat | Decreases | [56, 57] | |
| Spinach | Vascular endothelial cells | Human | Increases | [65] | |
| Vanillic acid | OA chondrocyte | Rat | Decreases | [62] | |
| eNOS | Spinach | Vascular endothelial cells | Human | Increases | [65] |
| NO | Cyanidin | OA chondrocytes | Human | Decreases | [58] |
| Phytoestrogens (coumestrol + biochanin A) |
Chondrocytes | Rat | Decreases | [56, 57] | |
| IFN-γ | Curcumin | CD4(+), CD8(+) T cells, natural killer (NK) and NKT cells | Human | Decreases | [66] |
| Hesperidin | RA fibroblast-like synovial cells | Mice | Decreases | [67] | |
| F4/80 | Hesperidin | OA chondrocytes | Rat | Decreases | [61] |
| Caspase-3 | Cyanidin | OA chondrocytes | Human | Decreases | [58] |
| PGE2 | Phytoestrogens (coumestrol + biochanin A) |
Chondrocytes | Rat | Decreases | [56, 57] |
| Vanillic acid | OA chondrocyte | Rat | Decreases | [62] | |
| VEGFA | Green tea | RA synovium tissue | Human | Decreases | [60] |
| TAK-1 | Vanillic acid | OA chondrocyte | Rat | Decreases | [62] |
| MAPK | Hesperidin | RA fibroblast-like synovial cells | Mice | Inhibits | [67] |
| PI3K/AKT | Anthocyanidin | Collagen-induced arthritis murine model and human PBMC | Human-mice | Decreases | [69] |
| Vanillic acid | OA chondrocyte | Rat | Inhibits | [62] | |
| Th17 cells | Hesperidin | OA chondrocytes | Rat | Increases | [61] |
| Collagen 2a1 | Cyanidin | OA chondrocytes | Human | Increases | [58] |
| Hesperidin | OA chondrocytes | Rat | Increases | [61] | |
| Aggrecan | Cyanidin | OA chondrocytes | Human | Increases | [58] |
| Hesperidin | OA chondrocytes | Rat | Increases | [61] | |
| SOX 9 gene | Green tea | OA chondrocytes | Human | Decreases | [64] |
| BMP-2 | Green tea | OA chondrocytes | Human | Decreases | [64] |
| STC-1 | Hesperidin | OA chondrocytes | Rat | Decreases | [61] |
| MDA | Hesperidin | OA chondrocytes | Rat | Decreases | [61] |
| ROS | Spinach | Vascular endothelial cells | Human | Decreases | [65] |
| VCAM1 | Spinach | Vascular endothelial cells | Human | Decreases | [65] |
| ICAM1 | Phytoestrogens (coumestrol + biochanin A) | Chondrocytes | Rat | Decreases | [56, 57] |
interleukin; RA, rheumatoid arthritis; OA, osteoarthritis; MCP1, monocyte chemoattractant protein-1; MMP, matrix metallopeptidases; ADAMTS5, ADAM metallopeptidase with thrombospondin type 1 motif 5; COX-2, cyclooxygenase 2; TNF-α, tumor necrosis factor alpha; NF-κB, nuclear factor-kappa beta; PBMC, peripheral blood mononuclear cell; CD 206, mannose receptor; iNOS, inducible nitric oxide synthase; eNOS, endothelial nitric oxide synthase; NO, nitric oxide; IFN-γ, interferon gamma; PGE2, prostaglandin E2; VEGFA, vascular endothelial growth factor A; TAK-1, transforming growth factor beta-activated kinase 1; MAPK, mitogen-activated protein kinase; PI3K/AKT, phosphatidylinositol-3-kinase; BMP-2, bone morphogenetic protein-2; STC-1, stanniocalcin; MDA, malondialdehyde; ROS, reactive oxygen species; VCAM 1, vascular cell adhesion molecule 1; ICAM 1, intercellular adhesion molecule 1
Polyphenols
Polyphenols are the biggest group of phytochemicals. They are bioactive compounds produced by plants, biosynthesized from the phenyl propanoid pathway for the phenolic acids or the shikimic acid pathway for gallotannins and analogs. They represent one of the most prevalent natural products in the plant kingdom. Former studies showed that plant-derived polyphenolic compounds were the most potent antioxidant and anti-inflammatory agents among all-natural compounds [16]. Different types of polyphenols can be found in fruits, vegetables, whole grains, legumes, herbs, spices, and plant-based beverages (tea, wine), contributing to their color, flavor, and pharmacological activities [18, 70, 71]. There is an increasing interest in research in this field due to their anti-inflammatory and antioxidant properties and fewer unwanted effects than common drugs to treat arthritis (Table 1). According to their chemical composition, they are classified into two distinctive groups: flavonoids and no flavonoids [70] (Table 1).
Flavonoids
Flavonoids are the primary polyphenol in various fruits and tea and can be classified into 6 subclasses: flavan-3-ols, flavonols, flavones, flavanones, isoflavones, and anthocyanidins [72].
Flavan-3-ols (catechin, gallocatechin, epicatechin, epigallocatechin, epicatechin 3-gallate, epigallocatechin 3-gallate (EGCG)) is one of the flavonoids with more evidence of therapeutic effects and more studies performed. It is found mainly in green tea and several studies support that the majority of beneficial effects of drinking green tea (anti-inflammatory, antioxidative, and immune-modulating) is related to the high content of EGCG, the most biologically active catechin, among others [73]. The administration of EGCG (2–250 mg/kg/day) in aged rats (above 24 months old, versus young, 3 months old), and in adjuvant-induced arthritic (AIA) rats, showed an increase of liver and brain antioxidant enzymes activities (superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GPx), glutathione reductase (GR), and glucose-6-phosphate dehydrogenase), an augmentation of the non-enzymatic antioxidants (tocopherol, ascorbic acid, and glutathione), and an amelioration of malondialdehyde (MDA) levels in all groups. The increase of antioxidant enzyme levels was higher in aged rats than in young rats [74, 75]. Although there is no evidence of an antioxidant effect of flavan-3-ols in humans, randomized clinical trials showed its anti-inflammatory effect. A randomized clinical trial with 50 patients with knee OA (KOA) compared treatment with green tea (500 mg/8 h) versus diclofenac (50 mg/12 h) for 4 weeks, and showed significant clinical improvements (visual analogue scale (VAS) pain, Western Ontario and McMaster Universities Arthritis Index (WOMAC) scores), with fewer adverse effects in the green tea group [24]. In another study, 120 RA patients diagnosed at least 10 years prior to the trial, and not taking any RA treatment during the previous 3 months, were assigned to one of 6 groups accordingly for 6 months: infliximab (3 mg/kg at baseline, at 2 and 6 weeks later, and then every 8 weeks), green tea (4–6 cups/day (60 to 125 mg catechins)), exercise (aerobic exercise for 45–60 min three times per week), infliximab plus green tea, infliximab plus exercise, or green tea plus exercise. Improvement of clinical (Disease Activity Score (DAS28-ESR)) and biochemical parameters (serum C-reactive protein (CRP), erythrocyte sedimentation rate (ESR), and serum levels of bone resorption markers (deoxypyridinoline, amino-terminal telopeptide of type 1 collagen, and bone alkaline phosphatase)) was observed in all groups. In patients subjected to combined treatment modalities, patients treated with green tea plus exercise showed a significant improvement in the above disease activity parameters compared with patients treated with infliximab plus exercise or infliximab plus green tea [25].
Flavonols (kaempferol, myricetin, quercetin) have anti-inflammatory and anti-nociceptive effects [26–28, 76]. In animal models, quercetin (GCQ) induced significant changes in both RA (collagen-induced arthritis (CIA) and complete Freund adjuvant arthritis (CFA)), and OA (monosodium iodoacetate (MIA)), highlighting the importance of conducting trials in patients for validation [47, 49, 51, 77]. Yet, RA studies in humans are contradictory. A randomized, double-blind, placebo-controlled clinical trial with 50 women with RA evaluated the effect of GCQ (500 mg/day) for 8 weeks. Quercetin significantly reduced early morning stiffness, morning and post-activity pain, DAS-28 and health assessment questionnaire (HAQ) scores, and plasma TNF concentration [26]. In contrast, a randomized, placebo-controlled, double-blind clinical trial that was conducted in 20 RA patients comparing GCQ + vitamin C (166 mg/133 mg), lipoic acid (300 mg), and placebo capsules, 3 times a day for 4 weeks, did not find significant changes in pain or a difference in serum levels of TNF, CRP, IL-1, and IL-6 [28]. For OA, we have more evidence of their positive effects. A randomized, double-blind, placebo-controlled study in 40 symptomatic KOA patients assessed the effect of supplementation with oral glucosamine (1200 mg), chondroitin sulfate (60 mg), and GCQ (45 mg) every day for 4 months. All groups showed an amelioration of symptomatic pain (VAS and Japanese Orthopedic Association criteria) and changes in levels of cartilage metabolism biomarkers (urinary CTX-II and serum CPII), but the effect was greater in the GCQ group [27]. A glucosamine (1200 mg)-chondroitin (75–111 mg)-quercetin GCQ (45 mg) supplement was given daily to 46 OA and 22 RA patients for 3 months. Only OA patients showed a significant improvement in pain symptoms, VAS, and daily activities, and also exhibited changes in synovial fluid composition (protein concentration, molecular size of hyaluronic acid, and chondroitin 6-sulfate concentration) [76].
Flavones (apigenin, luteolin, tangeritin) are present in different herbs, tea, green leaves, vegetables, fruit, and cereals. There is a lack of clinical studies with flavones, but the effects of spinach extract (250 and 500 mg/kg/day), which contains different flavones and flavonols, were evaluated in MIA OA rats, which improved histological and radiological scores, and downregulated the oxidative serum marker glutathione S-transferase (GST), and serum cartilage oligomeric matrix protein (COMP) and urinary C-telopeptide of type II collagen (CTX-II), which are markers of cartilage turnover [55]. A CIA mouse model was used to assess the effect of apigenin and showed a decrease of synovial hyperplasia, angiogenesis, and osteoclastogenesis [78].
Flavanones (hesperidin, naringenin) are mainly present on citrus fruits [79]. Hesperidin administration in CIA rats and mice models improved clinical scores, and decreased serum and articular TNF, MDA, glutathione, SOD, CAT, NO, and elastase activity [52, 54, 80]. In rheumatic patients, a randomized, double-blind, placebo-controlled trial studied the effect of hesperidin (3 g/day) for 12 weeks in 19 RA patients, and found clinical improvement (ACR20, VAS) and a decrease of serum CRP [80]. In addition, there is evidence of anti-inflammatory activity of flavanones in other diseases such as diabetes or nonalcoholic fatty disease (NAFD). In a randomized, double-blind, controlled clinical study with 64 diabetic patients, hesperidin (500 mg/day) for 6 weeks significantly decreased systolic blood pressure, mean arterial blood pressure, serum TNF, IL-6, and high sensitivity (hs)-CRP, whereas serum total antioxidant capacity increased in comparison to the baseline values [81]. In another randomized, double-blind, controlled clinical trial, 50 NAFD patients were given either hesperidin 1 g/day or placebo for 3 months. Hesperidin resulted in a significant reduction in serum levels of hs-CRP, TNF, and NF-κB [82].
Isoflavones are part of the phytoestrogen group and are mostly found in legumes, such as soybeans [83]. Although isoflavones are common components of Western diets, there are no randomized clinical trials studying the effect of isoflavones on human arthritis. Genistein, an isoflavone extracted from soybeans, inhibited IL-1β, IL-6, and TNF in the CIA mouse model [84]. The main benefit of soy is attributed to its polyphenols, but isoflavone-free soy protein isolate (SPI) has demonstrated antirheumatic effect, with a decrease of anti-CCP, TNF, MMP3, and MDA in rats with antigen-induced arthritis (AIA) [85].
Anthocyanidins (cyanidin, delphinidin, malvidin) are the main polyphenol of berry fruits and pomegranate. Each fruit has a different combination of polyphenolic compounds, and a different antioxidant capacity [86]. Animal studies in CIA mice, AIA rats, and MIA OA rats showed a protective effect of anthocyanins [50, 69, 87–89]. Two hundred juvenile idiopathic arthritis (JIA) patients were exposed to a combination therapy of etanercept (ETA) and blueberry. They were randomly assigned to 3 different treatment groups for 6 months: ETA (50 mg of etanercept twice weekly), ETABJ (matched etanercept and 50 ml blueberry juice daily), and ETAPJ (matched etanercept and placebo juice). The ETABJ group showed a decrease in disease severity, symptoms, and side effects and reduced serum levels of IL-1 compared to the other two groups [23]. In a randomized, double-blind, cross-over trial with 17 obese patients with radiographic KOA, a strawberry beverage (50 g/day) for 12 weeks significantly decreased serum IL-6, IL-1β, and MMP-3 levels, and reduced constant, intermittent, and total pain in these patients [29]. A randomized, placebo-controlled clinical trial with 55 RA patients showed that the group taking daily pomegranate extract (500 mg/day) for 8 weeks had lower levels of serum ESR, higher levels of glutathione peroxidase (GPx), and a decrease in swollen and tender joints, pain intensity, morning stiffness, and HAQ score, being all statistically significant [44].
Non-flavonoids
Non-flavonoid polyphenols can be divided in 6 subgroups: phenolic acids (benzoic and cinnamic), polyphenolic amides (avenanthramides (AVA), capsaicinoids, and piperine), stilbenoids (resveratrol (RSV), piceatannol, and pterostilbene), lignans, proteolytic enzymes (mangiferin, papain, and bromelain), among others (Table 1).
Gallic acid (GA) and vanillic acid (VA) are the main benzoic acids. GA is present in fruits, nuts, and seeds [90]. VA is characteristic of vanilla, Ginkgo biloba, berries, and honey [72]. Animal models of inflammatory pain (CFA) showed a significant improvement after VA treatment (suppression of paw skin inflammatory mediators as IL-1β, TNF, IL-33, and NF-κB), and improvement of paw oxidative markers measured using the antioxidant power (FRAP assay), free radical scavenging ability (ABTS assay), GSH levels, and lipid peroxidation (TBARS assay) [91].
Cinnamic acids are the major active ingredient of cinnamon [31]. Cinnamon inhibited TNF, IL-2, IL-4, and IFNγ in acute (carrageenan-induced rat paw edema), subacute (cotton pellet–induced granuloma), and sub-chronic (AIA, adjuvant-induced established polyarthritis) models of inflammation in rats [53, 92]. In a randomized, double-blind, placebo-controlled clinical trial with 32 women with RA, 18 received cinnamon capsules (2 g/day) for 8 weeks. Those in the cinnamon group had a significant decrease in serum levels of CRP and TNF, along with improvements in clinical symptoms (DAS-28, VAS, tender, and swollen joints counts) [31]. In another 16-week double-blind, randomized control trial, cinnamon (3 g/day) was studied in 116 metabolic syndrome patients. Subjects in the cinnamon group had improvements in a number of clinical (waist circumference, body mass index (BMI), blood pressure) and laboratory parameters (fasting blood glucose, glycosylated hemoglobin, cholesterol, and triglycerides), including a decrease in hs-CRP [93].
Polyphenolic amides: There is minimal evidence of positive effects of polyphenolic amides (avenanthramides (AVA), capsaicinoids, and piperine) in rheumatic diseases due to the lack of clinical trials or animal studies in arthritis, although some data suggest they also have anti-inflammatory and antioxidant properties. AVA, found in oats, showed the antioxidant effect of oats AVA (3.12 mg/day) in a randomized, placebo-controlled study in 120 healthy individuals. It increased significantly plasma levels of reduced glutathione, decreased plasma lipid peroxide, and had higher total plasma antioxidant capacity and erythrocyte SOD activity [94, 95]. Capsaicin is the main polyphenol of red and green chili pepper, responsible for its spicy flavor and it is known for its topical properties in improving OA pain [96]. And piperine, found in black pepper and responsible for its pungent taste, has a similar chemical structure as capsaicin and ginger [97]. In vivo animal models and in vitro studies have shown anti-inflammatory, analgesic, and anti-arthritic effects of piperine [98–100]. It improved nociceptive and arthritic symptoms in rat models of carrageenan-induced acute paw pain and arthritis, and inhibited the expression of IL-6, COX-2, PGE2, and MMPs significantly in FLS and human OA chondrocytes [98, 100]. To our knowledge, there are no humans’ studies in arthritis patients with piperine as a unique treatment, but there are trials in combination with other nutrients since piperine increases the oral bioavailability of other polyphenols, like curcuminoids [101].
Stilbenoids are anti-inflammatory compounds found in berries, grapes, red wines, soy, and nuts. Resveratrol (RSV) is the stilbenoid that has received the most attention [102, 103]. A randomized, controlled, clinical trial studied the effect of adding RSV (1 g/day) for 3 months to standard of care (SOC) treatment in 100 patients with RA. The study showed a decrease of serum levels of inflammatory markers (CRP, ESR, undercarboxylated osteocalcin, MMP-3, TNF, and IL-6) in the RSV group, being statistically significant [45]. A double-blind, placebo-controlled, randomized multi-center study involving 110 patients with KOA assessed the effects of meloxicam (15 mg/day) with and without the addition of RSV. RSV 500 mg/day for 3 months significantly improved WOMAC scores compared to placebo [104]. Freund’s AIA and MIA OA in rats also showed the benefits of RSV by decreasing clinical scores (paw thickness measured with caliper and swelling scores), histological scores (synovial hyperplasia, inflammatory cell infiltration, and cartilage degradation), and lowering serum levels of antioxidant enzymes (ROS) and proinflammatory cytokines (TNF-α, IL-1β, IL-6, IL-18, iNOS, and NF-κB) [105–107]. Synovial fluid mononuclear cells from 14 patients with RA and spondyloarthritis were cultured and divided in several groups: no treatment (control group), treatment with RSV alone (25 μM), methotrexate (MTX) (0.5 μg/ml), adalimumab (ADA) (5 μg/ml), RSV + MTX, or RSV + ADA. Compared to the untreated control group, the RSV group reduced MCP-1 production by synovial fluid mononuclear cells significantly, and the reduction was greater when RSV was combined with methotrexate. The effect of resveratrol was greatest in patients with low disease activity (DAS28CRP ≤ 3.2) and in cultures with high lymphocytes count [103].
Lignans are mainly found in sesamin (a bioactive component extracted from sesame) and flaxseeds. In a randomized, triple-blind, placebo-controlled clinical trial in 44 RA patients, with no change in treatment in the last 2 months, sesamin 200 mg/day was compared to placebo for 6 weeks. After the intervention, a significant reduction in the number of tender joints and the severity of pain was observed in the group with sesamin supplementation. Serum levels of hyaluronidase, MMP-3, hs-CRP, TNF, COX-2, and malondialdehyde (MDA) decreased significantly, and total antioxidant capacity (TAC) increased in the sesamin group [108, 109]. Vitex negundo is a medicinal plant used in Chinese medicine that is also rich in lignans. After administration in CIA rats, paw edema and arthritis score decreased. Vitex negundo (10, 20, 40 mg/kg/day) also reduced the infiltration of inflammatory cells and synovial hyperplasia, and attenuated cartilage damage in a dose-dependent manner, and reduced inflammatory cytokines (decreased serum levels of IL-1β, IL-6, IL-8, IL-17A, TNF, MMPs; increased serum level of IL-10; and reduced COX-2, iNOS, and NF-kB in synovial tissues) [110].
Proteolytic enzymes (mangiferin, papain, and bromelain) break down proteins and facilitate gastric and ileal digestion [111]. Mangiferin is the main polyphenol found in mango and papaya. Mango fruit also has other polyphenols, like gallic acid and gallotannins [112]. A randomized study with 20 patients with active RA evaluated the therapeutic effects and the safety of Mangifera indica extract (MIE; 900 mg/day) combined with methotrexate (MTX; 12.5 mg/week) for 6 months on reducing disease activity. Combined treatment significantly improved DAS28, decreased non-steroidal anti-inflammatory drugs (NSAIDs) intake in 100% of the patients, and decreased gastrointestinal side effects in 70% of the patients [43]. In CFA and CIA rat models, a decrease of serum levels of IFN-γ, IL-4, NF-κB, IL-1β, IL-6, and TNF, and tissue MDA, and an increase of serum IL-10 and SOD were observed after mangiferin treatment. The effect of methotrexate was less marked as compared to mangiferin [113, 114]. Carica papaya leaves have papain and abundant ascorbic acid. There is no evidence of its effects in arthritis in humans, but ingesting carica leaves (or however given and in what dose/exposure) led to a significant reduction in paw edema and granulation in animal models of carrageenan-induced paw edema, cotton pellet granuloma, and formaldehyde-induced arthritis rats [115]. Finally, bromelain is a natural mixture of proteolytic enzymes found in pineapple and kiwis that decreases the production of pro-inflammatory cytokines [116]. Bromelain (500 mg/day for 16 weeks) was compared with diclofenac (100 mg/day for 4 weeks) in a randomized clinical trial with 40 patients with mild-moderate KOA. At week 4, the bromelain group had a significant improvement in WOMAC scores, as well as a decrease of serum PGE2 with no increase in side effects. In addition, a significant decrease in serum MDA was observed in the treatment group at 16 weeks [30].
Curcumin, another non-flavonoid polyphenol, is the active component of turmeric, which provides the yellow color and its anti-inflammatory and antioxidant properties [117]. A randomized three-arm, parallel-group, randomized, double-blinded, placebo-controlled clinical trial in 201 OA patients compared curcumin (333 mg curcuminoids, or 350 mg curcuminoids and 150 mg boswellic acid, present in resin of boswellic plant, also rich in polyphenols) taken orally three times a day for 12 weeks versus placebo. Physical performance tests and the WOMAC pain index were better in both curcumin groups than in the placebo group [32]. Another randomized, double-blind, placebo-controlled, three-arm, parallel-group study was conducted to evaluate the efficacy of curcumin (250 mg or 500 mg/12 h) with placebo in 36 RA patients with active disease for 90 days. RA patients who received either dose of curcumin reported statistically significant changes in their clinical symptoms (VAS, DAS28) and in serum ESR, CPR, and RF values, compared to baseline and placebo [35]. Yet, another randomized, double-blind, controlled trial with 65 RA patients did not find any difference in DAS-28, tender joint count (TJC), swollen joint count (SJC) decrease, and serum ESR, between curcumin (120 mg/day) and placebo for 12 weeks [36]. A randomized, double-blind trial compared curcumin plus piperine (500 mg–15 mg/day) versus placebo for 6 weeks in 40 mild-moderate KOA patients, and showed an increase of serum SOD and GSH and a decrease of MDA, but no clinical outcomes were included in that trial [38]. Another randomized, double-blind, placebo-control clinical trial with 40 patients with mild-moderate KOA showed non-significant changes in clinical symptoms (WOMAC score, Lequesne’s pain functional index (LPFI) and visual analogue scale) after treatment with curcumin 1500 mg/day [39]. Some trials compared curcumin effect with NSAIDs. A randomized clinical trial with 367 patients with knee OA with a pain score of 5 or higher had curcumin 1500 mg/day or ibuprofen 1200 mg/day for 1 month. The mean difference of WOMAC total, WOMAC pain, and WOMAC function scores at week 4 of curcumin was noninferior to those for the ibuprofen group, except for the WOMAC stiffness subscale, which showed a trend toward significance, and the number of events of abdominal pain/discomfort was significantly higher in the ibuprofen group [33]. In another randomized, open-label, parallel, clinical study, 139 knee OA patients received either curcumin capsule (500 mg, 3 times per day) or diclofenac (50 mg, twice per day) for 1 month. At days 14 and 28, there was no statistically significant difference between the improvement in pain and KOOS scale between subjects treated with curcumin and those treated with diclofenac. Adverse effects were significantly less in the curcumin group [34]. Another prospective, randomized open, blinded end-point (PROBE design study) compared curcumin (90 mg/day) with diclofenac (75 mg/day) in 80 patients with KOA for 4 weeks. This study showed a decrease of COX-2 secretion by SF monocytes in both groups [37]. In vitro studies showed an anti-inflammatory and immunosuppressive role of curcumin in 34 patients with psoriatic disease (22 with psoriasis and 12 with PsA). Peripheral blood mononuclear cells were pretreated (2 h prior to activation) with curcumin at pharmacological concentrations of 5–10 g/mL. In vitro results showed an inhibition of pro-inflammatory IFN-γ by CD4+, CD8+ T cells, natural killer (NK) and NKT cells, and IL-17 production by CD4+T cells [66].
Other polyphenols such as gingerol, rosmarinic acid (RosA), ellagic acid, and ursolic acid (UA) also have anti-inflammatory properties. Gingerol is the primary polyphenol in ginger, responsible for its pungent taste, and along with shogaol, its analog, has diverse biological activities [118]. A non-randomized study in KOA patients showed improvement in Lysholm Knees score, SF-36, and knee circumference after the intake of 25 mg of ginger and 5 mg of Echinacea for 30 days [40]. In a randomized, double-blind, placebo-controlled 3-month clinical trial, 120 patients with KOA were randomly assigned to the ginger (500 mg/day) or placebo group. A decrease in serum TNF and IL-1β was observed in the ginger group but no clinical outcomes were included in that trial [41]. Animal models of RA and OA (CIA, CFA, and IL-1β-induced OA models in rats) showed a significantly decrease of paw edema and serum levels of IL-1, IL-6, IL-8, MMPs, MCPs, and TNF-α after its administration of ginger and larger effects when compared to ibuprofen, betamethasone, or indomethacin [119–124]. RosA is found in rosemary. A randomized, double-blind study that included 46 participants with KOA treated with RosA for 4 months showed a significant decrease in pain score from baseline compared to placebo [46]. Ellagic acid is present in several fruits and nuts. A double-blind, parallel pilot clinical study was conducted in subjects with knee pain without any history of pain treatment. Participants were randomly assigned to two different groups: 1 g/day of guava leaf extract or placebo for 3 months. Knee pain and stiffness in the guava group were significantly lower measured by VAS and Japanese Knee Osteoarthritis Measure scores [42]. UA is present on the peel of apples and other fruits and vegetables, as well as in herbs and spices [125]. No studies of UR have been carried out in humans. However, US induced an improvement in pain and swelling scores, and a decrease of serum TNF, and IL-1 in Freund’s adjuvant (CFA)–induced arthritic rats [126].
Other Micronutrients
Selenium (Se), zinc (Zn), and magnesium (Mg) are three trace elements for which low levels have been associated with inflammatory and autoimmune diseases [127–136]. Selenium is an essential part of the enzyme glutathione peroxidase (GSH-Px), which protects cells from oxidative damage, and has been shown to have anti-proliferative, anti-inflammatory, and immune-altering effects in diseases like cancer, sepsis, or RA [127]. Zinc is required for controlling essential biological processes that affect normal growth, development, repair, metabolism, and maintenance of cell integrity and functionality, and it is crucial for maintaining homeostasis of immune system, and its deficiency is seen in RA, multiple sclerosis or type 1 diabetes (T1D) [131]. Magnesium is also associated with inflammatory and immune responses in diseases like cancer or T1D [137].
In animal models, a comparison between selenium and celecoxib was conducted in CFA-induced RA rats and selenium supplementation significantly decreased levels of inflammatory cytokines (TNF, IL-1β, IL-6, and MCP-1) and anti-oxidant activity (CAT, GPx1, and COX-2) in joint tissue [138]. In pristane-induced arthritis rats, magnesium deficiency (low-magnesium diet, 0.06 g/kg) correlated with higher levels of arthritis severity score, and synovial tissue expression of IL-6, and, in post-traumatic osteoarthritis rabbits, treatment with magnesium sulfate decreased synovial tissue expression of IL-1β, TNF, and MMP-3 [139, 140].
There is a lack of clinical trials assessing trace element effects on inflammation, clinical outcomes, and cytokine modulation in arthritis patients. However, there are some studies with healthy subjects or other inflammatory diseases that evaluate anti-inflammatory and antioxidant effects. For instance, low Se levels are associated with severe sepsis, trauma, and burn injuries and its administration led to significantly reduced mortality and improvement of clinical outcomes (multiorgan failure, acute renal failure, respiratory distress syndrome, and infection) [135]. In randomized, double-blind, placebo-controlled studies, selenium supplementation (200 μg) in women with polycystic ovary syndrome (PCOS) was associated with a decrease in serum hs-CRP and plasma MDA levels [141], and an increase in plasma TAC and GSH levels compared with placebo [142]. Zinc supplementation in hemodialysis patients decreased serum CRP levels [143], and, in elderly people, decreased serum CRP and inflammatory cytokines [130]. A randomized, double-blinded, placebo trial of zinc supplementation (45 mg/day) was performed in 50 healthy elderly subjects for 6 months. Plasma zinc concentrations increased and hs-CRP, IL-6, MCP-1, VCAM-1, and decreased MDA, compared to placebo [130]. Mg deficiency is associated with chronic low-grade inflammation, and/or with pathological conditions for which inflammatory stress is considered a risk factor (metabolic syndrome, obesity, hypertension, diabetes, stroke, coronary heart disease) [144]. A systematic review of 8 randomized controlled trials suggested that serum magnesium levels and magnesium intake from food are inversely correlated with serum CRP levels [145]. In OA and fibromyalgia patients, low magnesium intake and serum levels have also be associated with higher serum CRP levels [137, 146]. Highly dietary daily intake of Mg is associated with better knee cartilage architecture (increase in mean cartilage thickness, cartilage volume at medial tibia, cartilage volume and mean cartilage thickness at central medial femur, and cartilage volume and mean cartilage thickness in the central medial tibiofemoral compartment), lower radiographic KOA and joint space narrow, and lower risk of fracture in patients with knee OA [133, 147–149]. However, in a second, much larger cohort of 5000 people, magnesium intake had no impact on clinical knee OA incidence despite once again being inversely associated with hs-CRP levels [150]. A randomized, placebo-controlled clinical trial with 100 adults with poor sleep quality assessed the effect of 320 mg magnesium/day for 7 weeks. Based on food intake, 58% of the participants were consuming less than the estimated average requirement (EAR) for magnesium. The lower intake of magnesium was associated with a significantly higher BMI and plasma CRP levels. Magnesium supplementation decreased plasma CRP in participants with baseline values > 3.0 mg/L [151].
Nutrients and Microbiome
Diet adjusts the human gut microbiota [14, 152], and several studies demonstrate the shift of microbiome after consuming different foods [153–156]. Bacteria in the gut are not only important in the absorption of certain vitamins and in the synthesis of bile acids, but also have the potential to modify circulating pro- or anti-inflammatory mediators as they are involved in the metabolism of some food-derived metabolites [157, 158]. Of interest, some studies have shown microbiome shifts after the administration of the above micronutrients. Polyphenols present in vegetables, fruits (berries, grape, pomegranate), red wine, green tea, and cocoa were shown to have an effect in gut microbiota shaping, increasing the abundance of beneficial gut microbiome like Bifidobacterium, Lactobacillus, Akkermansia, Faecalibacterium, Prevotella, Pseudoflavonifractor, Oscillibacter, Roseburia, Eubacterium rectale, Bacteroides uniformis, Blautia coccoides, and Eggerthella lenta [reviewed in 159]. In a randomized placebo-controlled study, 29 healthy adults had daily capsules of mixed spices (cinnamon 1 g, oregano 1.5 g, ginger 1.5 g, black pepper 0.85 g, and cayenne pepper 0.15 g) for 14 days. After the intervention, 26 operational taxonomic units of gut bacteria were different between groups. The spice consumption group showed a trend of enrichment in Bacteroidetes, a significant reduction in Firmicutes and an increase of fecal short-chain fatty acid propionate concentration, which is considered an anti-inflammatory metabolite [153]. Another controlled, randomized, five-period, cross-over study with washouts between diet periods was conducted in 18 healthy subjects with 5 different treatments for 3-week diet period: 0 servings/day of almonds (control); 1.5 servings (42 g)/day of whole almonds; 1.5 servings/day of whole, roasted almonds; 1.5 servings/day of roasted, chopped almonds; and 1.5 servings/day of almond butter. Almond consumption induced changes in the microbial composition of gastrointestinal microbiota (increased the relative abundances of Lachnospira, Roseburia, and Dialister). On the other hand, almond processing impacted the relative abundances of bacterial genera (chopped almonds increased Lachnospira, Roseburia, and Oscillospira compared to control, while whole almonds increased Dialister compared to control, and there were no differences between almond butter and control) [154]. Walnut consumption (43 g/day) was evaluated in a randomized, controlled, prospective, cross-over study with 194 healthy adults for 8 weeks. Walnut consumption significantly affected microbiome composition and diversity in this trial. Ruminococcaceae and Bifidobacteria, two normal inhabitants of a healthy human gut, increased significantly while Clostridium sp. decreased significantly during walnut consumption [155]. A single group-design trial in 26 healthy subjects evaluated the effect of consuming 15 mg of inulin-type fructans (ITFs), a type of fermentable dietary fiber, daily for 2 weeks. They had an increased proportion of the Bifidobacterium genus, a decreased level of Clostridiales, and a tendency to decrease Oxalobacteraceae [156]. In animal models, mice were fed with either control or magnesium-deficient diet for 4 days. The group fed with magnesium-deficient diet had a lower gut Bifidobacteria content, and a higher liver and/or intestine TNF and IL-6 levels [160]. Chronic zinc deficiency in a chick model triggered a decrease of gene expression of pro-inflammatory cytokines (IL-1β, IL-6, TNF), a significantly lower phylogenetic diversity, and a significant expansion of β-diversity (what was found in various pathological states such as inflammatory bowel disease, DM, and obesity). Zn deficiency group also had significantly lower levels of Peptostreptococcaceae, and Clostridiales, found in healthy individuals, and higher levels of Enterococcaceae and Enterobacteriaceae, increased in diseases as septic arthritis, irritable bowel syndrome, hepatic encephalopathy, depression, and anorexia [161–163]. Although some species, including Lactobacillus and Bifidobacterium spp., are considered potentially beneficial, more research is needed to explore the complex interaction between nutrients and gut microbiome.
Why Are Dietary Interventions Still Not Mainstream?
While there is greater knowledge of the effects of pharmaceutical therapies for arthritis, there is less knowledge of the effects of many nutrients. And even though there is evidence that physician discussions of diet make a positive influence on patient dietary selections, these conversations are not common in clinical practice. There is a global deficiency of nutrition education in physician training, including the use of nutrients to manage chronic diseases and methods for providing advice to their patients [164, 165]. A recent review highlighted the lack of nutrition knowledge and confidence in counseling among medical students worldwide. Most schools perform less than 25 h of nutrition education over 5 or 6 years of medical training. Trainees often learn about nutrition in biochemistry lectures that are not necessarily relevant to everyday practice [166]. The interest in nutrition among medical students is high. However, it decreases by the time they graduate, since they do not see nutrition substantively incorporated into their curriculum and do not observe clinical mentors incorporating nutritional interventions into their care plans [167]. In fact, a recent study showed that only 36.0% of general practitioners had positive attitudes toward nutrition and nutrition care [168].
This, as well as the data gaps, lack of regulated nutritional supplements with standardized quantities, insufficient or complete lack of insurance coverage for nutritionist or dietician consultations, and lack of dietary programs directed toward arthritis patients result in a lack of confidence in nutrition as a treatment among clinicians. Without adequate nutrition education, it is reasonable to assume that doctors are not able to provide the highest quality care to patients [167].
Conclusions
This review summarizes the current evidence of the effects of polyphenols (second metabolites produced by plants) and other micronutrients present in fruits and vegetables (selenium, zinc, and magnesium) on inflammatory and oxidative response. Although underlying mechanisms of their intracellular effects are now better understood, bioavailability of polyphenols and other micronutrients and their biofunctionality are still unclear. Interpretation of the available data is complicated by the fact that some trials use micronutrient-enriched foods instead of specific micronutrients, as well as the fact that gut microbiota participate in dietary polyphenols metabolism and affect the bioavailability of both polyphenols and their metabolites [169]. Nutrients in turn, can modulate gastrointestinal microbiota and potentially decrease systemic inflammation [170]. Thus, the consequences of observed changes in microbiomes as a result of exposure to micronutrients remain unclear. Since diverse polyphenols have multiple intracellular targets, additional data is required to understand whether or not there are synergistic effects between polyphenols and commonly used medications in patients with rheumatic diseases. Many clinical questions remain unanswered and more longitudinal and randomized trials are needed to understand both short-term and longer-term effects.
Yet, in terms of recommendations for rheumatic patients, polyphenols and micronutrients could be easily added to our patients’ diet for better management of the disease. Although some observational studies suggest micronutrient deficiency in RA patients [128, 129, 134, 171], like selenium, zinc, magnesium, and vitamin D, more studies are needed to verify the deficient nutritional status in those patients and whether patients with deficiencies would benefit more from nutrient supplementation. Some foods rich in several of the anti-inflammatory nutrients (Table 1) would be easy to introduce in their current diet, like fruits, vegetables, spices, and complex food like quinoa, amaranth or chia seeds, which are rich in many different polyphenols. We could also recommend that our patients add a daily cup of green tea, and include different spices like curcumin, ginger, pepper, or cinnamon in their daily cooking. We could also suggest an easy way to have fruits and vegetables in a green morning smoothie with spinach, berries, or enzymatic fruits like papaya, mango, and pineapple. In our experience, smoothies are a good and easy way for the patients to eat more vegetables and fruits.
In conclusion, the most significant studies included in this review support that polyphenols and micronutrients have anti-inflammatory and antioxidant properties that could help to improve patients with chronic inflammatory arthritis. However, clinical trials and subsequent implementation strategies are needed to define how to best integrate dietary interventions into the care of patients with inflammatory arthritis.
Acknowledgments
The authors acknowledge grant support from Krupp Endowed Fund to MBM and MG and NIH AR AR073324 to MG.
Compliance with Ethical Standards
Conflict of Interest MBM has nothing to disclose. AO has consulted for Abbvie, Amgen, BMS, Celgene, Corrona, Janssen, Lilly, Novartis, and Pfizer and received grants from Novartis and Pfizer (to Penn) and Amgen (to Forward) out of the scope of this work. MG has received grants from Novartis and Pfizer out of the scope of this work.
Footnotes
Human and Animal Rights Statements This article does not contain any studies with human or animal subjects performed by any of the authors.
References
- 1.Barbour KE, Helmick CG, Boring M, Brady TJ. Vital signs: prevalence of doctor-diagnosed arthritis and arthritis-attributable activity limitation – United States, 2013–2015. Mortal Wkly Rep. 2017;66:246–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Scherer HU, Häupl T, Burmester GR. The etiology of rheumatoid arthritis. J Autoimmun. Elsevier; 2020;102,400. Available from: 10.1016/j.jaut.2019.102400 [DOI] [PubMed] [Google Scholar]
- 3.Brennan FM, McInnes IB. Evidence that cytokines play a role in rheumatoid arthritis. J Clin Invest. 2008;118:3537–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Veale DJ, Fearon U. The pathogenesis of psoriatic arthritis. Lancet [Internet]. Elsevier Ltd.; 2018;391:2273–84. Available from: 10.1016/S0140-6736(18)30830-4 [DOI] [PubMed] [Google Scholar]
- 5.Berenbaum F, Wallace IJ, Lieberman DE, Felson DT. Modern-day environmental factors in the pathogenesis of osteoarthritis. Nat Rev. Rheumatol. Springer US; 2018;14:674–81. [DOI] [PubMed] [Google Scholar]
- 6.Mateen S, Zafar A, Moin S, Khan AQ, Zubair S. Understanding the role of cytokines in the pathogenesis of rheumatoid arthritis. Clin Chim Acta. Elsevier B.V.; 2016. p. 161–71. [DOI] [PubMed] [Google Scholar]
- 7.Bustamante MF, Agustín-Perez M, Cedola F, Coras R, Narasimhan R, Golshan S, et al. Design of an anti-inflammatory diet (ITIS diet) for patients with rheumatoid arthritis. Contemp Clin Trials [Internet]. Elsevier Inc.; 2020;17:100524. Available from: 10.1016/j.conctc.2020.100524 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Cross M, Smith E, Hoy D, Carmona L, Wolfe F, Vos T, et al. The global burden of rheumatoid arthritis: estimates from the Global Burden of Disease 2010 study. Ann Rheum Dis. 2014;73:1316–22. [DOI] [PubMed] [Google Scholar]
- 9.Afshin A, Sur PJ, Fay KA, Cornaby L, Ferrara G, Salama JS, et al. Health effects of dietary risks in 195 countries, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet. 2019;393:1958–72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ford AR, Siegel M, Bagel J, Cordoro KM, Garg A, Gottlieb A, et al. Dietary recommendations for adults with psoriasis or psoriatic arthritis from the medical board of the national psoriasis foundation a systematic review. JAMA Dermatol. American Medical Association. 2018;154:934–50. [DOI] [PubMed] [Google Scholar]
- 11.Mahil SK, McSweeney SM, Kloczko E, McGowan B, Barker JN, Smith CH. Does weight loss reduce the severity and incidence of psoriasis or psoriatic arthritis? A Critically Appraised Topic. Br. J. Dermatol. Blackwell Publishing Ltd.; 2019. p. 946–53. [DOI] [PubMed] [Google Scholar]
- 12.Di Minno MND, Peluso R, Iervolino S, Russolillo A, Lupoli R, Scarpa R. Weight loss and achievement of minimal disease activity in patients with psoriatic arthritis starting treatment with tumour necrosis factor α blockers. Ann Rheum Dis. BMJ Publishing Group. 2014;73:1157–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Klingberg E, Bilberg A, Björkman S, Hedberg M, Jacobsson L, Forsblad-D’Elia H, et al. Weight loss improves disease activity in patients with psoriatic arthritis and obesity: an interventional study. Arthritis Res Ther BioMed Central Ltd; 2019;21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Paolino S, Pacini G, Patanè M, Alessandri E, Cattelan F, Goegan F, et al. Interactions between microbiota, diet/nutrients and immune/ inflammatory response in rheumatic diseases: focus on rheumatoid arthritis. Reumatologia. 2019;57:151–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Alwarith J, Kahleova H, Rembert E, Yonas W, Dort S, Calcagno M, et al. Nutrition interventions in rheumatoid arthritis: the potential use of plant-based diets. A Review. Front Nutr. 2019;6:1–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Khan H, Sureda A, Belwal T, Çetinkaya S, Süntar İ, Tejada S, et al. Polyphenols in the treatment of autoimmune diseases. Autoimmun Rev. 2019;18:647–57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Jiang TA. Health benefits of culinary herbs and spices. J AOAC Int. 2019;102:395–411. [DOI] [PubMed] [Google Scholar]
- 18.Recio MC, Andujar I, Rios JL. Anti-inflammatory agents from plants: progress and potential. Curr Med Chem. 2012;19:2088–103. [DOI] [PubMed] [Google Scholar]
- 19.Martel-Pelletier J, Barr AJ, Cicuttini FM, Conaghan PG, Cooper C, Goldring MB, et al. Osteoarthritis. Natl Rev. 2016;2. [DOI] [PubMed] [Google Scholar]
- 20.Bortoluzzi A, Furini F, Scirè CA. Osteoarthritis and its management - epidemiology, nutritional aspects and environmental factors. Autoimmun Rev. [Internet]. Elsevier B.V; 2018;17:1097–104. Available from: 10.1016/j.autrev.2018.06.002 [DOI] [PubMed] [Google Scholar]
- 21.Ogdie A, Asch DA. Changing health behaviours in rheumatology: an introduction to behavioural economics. Nat Rev Rheumatol Nat Res. 2020;16:53–60. [DOI] [PubMed] [Google Scholar]
- 22.Guerreiro CS, Calado Â, Sousa J, Fonseca JE. Diet, microbiota, and gut permeability-the unknown triad in rheumatoid arthritis. Front Med. 2018;5:1–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Zhong Y, Wang Y, Guo J, Chu H, Gao Y, Pang L. Blueberry improves the therapeutic effect of etanercept on patients with juvenile idiopathic arthritis: phase III study. Tohoku J Exp Med. 2015;237:183–91. [DOI] [PubMed] [Google Scholar]
- 24.Hashempur MH, Sadrneshin S, Mosavat SH, Ashraf A. Green tea (Camellia sinensis) for patients with knee osteoarthritis: a randomized open-label active-controlled clinical trial. Clin Nutr [Internet]. Elsevier Ltd.; 2018;37:85–90. Available from: 10.1016/j.clnu.2016.12.004 [DOI] [PubMed] [Google Scholar]
- 25.Alghadir AH, Gabr SA, Al-Eisa ES. Green tea and exercise interventions as nondrug remedies in geriatric patients with rheumatoid arthritis. J Phys Ther. 2016;28:2820–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Javadi F, Ahmadzadeh A, Eghtesadi S, Aryaeian N, Zabihiyeganeh M, Rahimi Foroushani A, et al. The effect of quercetin on inflammatory factors and clinical symptoms in women with rheumatoid arthritis: a double-blind, randomized controlled trial. J Am Coll Nutr. 2017;36:9–15. [DOI] [PubMed] [Google Scholar]
- 27.Kanzaki N, Saito K, Maeda A, Kitagawa Y, Kiso Y, Watanabe K, et al. Effect of a dietary supplement containing glucosamine hydrochloride, chondroitin sulfate and quercetin glycosides on symptomatic knee osteoarthritis: a randomized, double-blind, placebo-controlled study. J Sci Food Agric. 2012;92:862–9. [DOI] [PubMed] [Google Scholar]
- 28.Bae SC, Jung WJ, Lee EJ, Yu R, Sung MK. Effects of antioxidant supplements intervention on the level of plasma inflammatory molecules and disease severity of rheumatoid arthritis patients. J Am Coll Nutr. 2009;28:56–62. [DOI] [PubMed] [Google Scholar]
- 29.Schell J, Hal Scofield R, Barrett JR, Kurien BT, Betts N, Lyons TJ, et al. Strawberries improve pain and inflammation in obese adults with radiographic evidence of knee osteoarthritis. Nutrients. 2017;9:949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kasemsuk T, Saengpetch N, Sibmooh N. Improved WOMAC score following 16-week treatment with bromelain for knee osteoarthritis. Clin Rheumatol; 2016; Available from: 10.1007/s10067-016-3363-1 [DOI] [PubMed] [Google Scholar]
- 31.Shishehbor F, Rezaeyan Safar M, Rajaei E, Haghighizadeh MH. Cinnamon consumption improves clinical symptoms and inflammatory markers in women with rheumatoid arthritis. J Am Coll Nutr [Internet]. Taylor & Francis; 2018;37:685–90. Available from: 10.1080/07315724.2018.1460733 [DOI] [PubMed] [Google Scholar]
- 32.Haroyan A, Mukuchyan V, Mkrtchyan N, Minasyan N, Gasparyan S, Sargsyan A, et al. Efficacy and safety of curcumin and its combination with boswellic acid in osteoarthritis: a comparative, randomized, double-blind, placebo-controlled study. BMC Complement Altern Med. BMC Complement Altern Med. 2018;18:1–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kuptniratsaikul V, Dajpratham P, Taechaarpornkul W, Buntragulpoontawee M, Lukkanapichonchut P, Chootip C, et al. Efficacy and safety of Curcuma domestica extracts compared with ibuprofen in patients with knee osteoarthritis: a multi-center study. Clin Interv Aging. 2014;9:451–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Shep D, Khanwelkar C, Gade P, Karad S. Safety and efficacy of curcumin versus diclofenac in knee osteoarthritis: a randomized open-label parallel-arm study. Trials Trials. 2019;20:1–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Amalraj A, Varma K, Jacob J, Divya C, Kunnumakkara AB, Stohs SJ, et al. A novel highly bioavailable curcumin formulation improves symptoms and diagnostic indicators in rheumatoid arthritis patients: a randomized, double-blind, placebo-controlled, two-dose, three-arm, and parallel-group study. J Med Food. 2017;20:1022–30. [DOI] [PubMed] [Google Scholar]
- 36.Javadi M, Khadem Haghighian H, Goodarzy S, Abbasi M, Nassiri-Asl M. Effect of curcumin nanomicelle on the clinical symptoms of patients with rheumatoid arthritis: a randomized, double-blind, controlled trial. Int J Rheum Dis. 2019;22:1857–62. [DOI] [PubMed] [Google Scholar]
- 37.Kertia N, Asdie AH, Wasilah Rochmah M. Ability of curcuminoid compared to diclofenac sodium in reducing the secretion of cycloxygenase-2 enzyme by synovial fluid’s monocytes of patients with osteoarthritis. Acta Med Indones. 2012;66:37–9. [PubMed] [Google Scholar]
- 38.Panahi Y, Alishiri GH, Parvin S, Sahebkar A. Mitigation of systemic oxidative stress by curcuminoids in osteoarthritis: results of a randomized controlled trial. J Diet Suppl. 2016;13:209–20. [DOI] [PubMed] [Google Scholar]
- 39.Rahimnia AR, Panahi Y, Alishiri G, Sharafi M, Sahebkar A. Impact of supplementation with curcuminoids on systemic inflammation in patients with knee osteoarthritis: findings from a randomized double-blind placebo-controlled trial. Drug Res. 2014;65:521–5. [DOI] [PubMed] [Google Scholar]
- 40.Rondanelli M, Riva A, Morazzoni P, Allegrini P, Faliva MA, Naso M, et al. The effect and safety of highly standardized Ginger (Zingiber officinale) and Echinacea (Echinacea angustifolia) extract supplementation on inflammation and chronic pain in NSAIDs poor responders. A pilot study in subjects with knee arthrosis. Nat Prod Res [Internet]. Taylor & Francis; 2017;31:1309–13. Available from: 10.1080/14786419.2016.1236097 [DOI] [PubMed] [Google Scholar]
- 41.Mozaffari-Khosravi H, Naderi Z, Dehghan A, Nadjarzadeh A, Fallah HH. Effect of ginger supplementation on proinflammatory cytokines in older patients with osteoarthritis: outcomes of a randomized controlled clinical trial. J Nutr Gerontol Geriatr. 2016;35: 209–18. [DOI] [PubMed] [Google Scholar]
- 42.Kakuo S, Fushimi T, Kawasaki K, Nakamura J, Ota N. Effects of Psidium guajava Linn. leaf extract in Japanese subjects with knee pain: a randomized, double-blind, placebo-controlled, parallel pilot study. Aging Clin Exp Res [Internet]. Springer International Publishing; 2018;0:0. Available from: 10.1007/s40520-018-0953-6 [DOI] [PubMed] [Google Scholar]
- 43.Mantecón AML, Garrido G, Delgado-hernández R, Garridosuárez BB. Combination of Mangifera indica L. extract supplementation plus methotrexate in rheumatoid arthritis patients: a pilot study. Phytother Res. 2014;1172:1163–72. [DOI] [PubMed] [Google Scholar]
- 44.Ghavipour M, Sotoudeh G, Tavakoli E, Mowla K, Hasanzadeh J, Mazloom Z. Pomegranate extract alleviates disease activity and some blood biomarkers of inflammation and oxidative stress in rheumatoid arthritis patients. Eur J Clin Nutr [Internet]. Nature Publishing Group; 2017;71:92–6. Available from: 10.1038/ejcn.2016.151 [DOI] [PubMed] [Google Scholar]
- 45.Khojah HM, Ahmed S, Abdel-Rahman MS, Elhakeim EH. Resveratrol as an effective adjuvant therapy in the management of rheumatoid arthritis: a clinical study. Clin Rheumatol. 2018;37: 2035–42. [DOI] [PubMed] [Google Scholar]
- 46.Connelly AE, Tucker AJ, Tulk H, Catapang M, Chapman L, Sheikh N, et al. High-rosmarinic acid spearmint tea in the management of knee osteoarthritis symptoms. J Med Food. 2014;17: 1361–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Wu J, Zhao FT, Fan KJ, Zhang J, Xu BX, Wang QS, et al. Dihydromyricetin inhibits inflammation of fibroblast-like synoviocytes through regulation of nuclear factor-kB signaling in rats with collagen-induced arthritis. J Pharmacol Exp Ther. 2019;368:218–28. [DOI] [PubMed] [Google Scholar]
- 48.Wang J, Zhao Q. Kaempferitrin inhibits proliferation, induces apoptosis, and ameliorates inflammation in human rheumatoid arthritis fibroblast-like synoviocytes. Phytother Res. 2019;33:1726–35. [DOI] [PubMed] [Google Scholar]
- 49.da Silva Pereira Saccol R, da Silveira KL, Adefegha SA, Manzoni AG, da Silveira LL, Coelho APV, et al. Effect of quercetin on E-NTPDase/E-ADA activities and cytokine secretion of complete Freund adjuvant–induced arthritic rats. Cell Biochem Funct. 2019;37:474–85. [DOI] [PubMed] [Google Scholar]
- 50.Moser A, York KRDM. Consumption of hydrolyzable tannins rich pomegranate extract (POMx) suppresses inflammation and joint damage in rheumatoid arthritis. Bone. 2008;23:1–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Tsai SW, Lin CC, Lin SC, Wang SP, Yang DH. Isorhamnetin ameliorates inflammatory responses and articular cartilage damage in the rats of monosodium iodoacetate-induced osteoarthritis. Immunopharmacol Immunotoxicol [Internet]. Taylor & Francis; 2019;41:504–12. Available from: 10.1080/08923973.2019.1641723 [DOI] [PubMed] [Google Scholar]
- 52.Ahmad S, Alam K, Hossain MM, Fatima M. Anti-arthritogenic and cardioprotective action of hesperidin and daidzein in collagen-induced rheumatoid arthritis. Mol Cell Biochem. Springer US; 2016 [DOI] [PubMed] [Google Scholar]
- 53.Qadir MMF, Bhatti A, Ashraf MU, Sandhu MA, Anjum S, John P. Immunomodulatory and therapeutic role of Cinnamomum verum extracts in collagen-induced arthritic BALB/c mice. Inflammopharmacology. Springer International Publishing. 2018;26:157–70. [DOI] [PubMed] [Google Scholar]
- 54.Umar S, Kumar A, Sajad M, Zargan J. Hesperidin inhibits collagen-induced arthritis possibly through suppression of free radical load and reduction in neutrophil activation and infiltration. Rheumatol Int. 2012; [DOI] [PubMed] [Google Scholar]
- 55.Choudhary D, Kothari P, Tripathi AK, Singh S, Adhikary S, Ahmad N, et al. Spinacia oleracea extract attenuates disease progression and sub-chondral bone changes in monosodium iodoacetate-induced osteoarthritis in rats. BMC Complement Altern Med. 2018;18:1–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Oh JS, Cho IA, Kang KR, You JS, Yu SJ, Lee GJ, et al. Biochanin-A antagonizes the interleukin-1β-induced catabolic inflammation through the modulation of NFκB cellular signaling in primary rat chondrocytes. Biochem Biophys Res Commun. 2016;477:723–30. [DOI] [PubMed] [Google Scholar]
- 57.You JS, Cho IA, Kang KR, Oh JS, Yu SJ, Lee GJ, et al. Coumestrol counteracts interleukin-1β-Induced catabolic effects by suppressing inflammation in primary rat chondrocytes. Inflammation. 2017;40:79–91. [DOI] [PubMed] [Google Scholar]
- 58.Zhao L, Zou T, Gomez NA, Wang B, Zhu MJ, Du M. Raspberry alleviates obesity-induced inflammation and insulin resistance in skeletal muscle through activation of AMP-activated protein kinase (AMPK) α1. Nutr Diabetes [Internet]. Springer US; 2018;8. Available from: 10.1038/s41387-018-0049-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Liu FC, Wang CC, Lu JW, Lee CH, Chen SC, Ho YJ, et al. Chondroprotective effects of genistein against osteoarthritis induced joint inflammation. Nutrients. 2019;11:1–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Fechtnera S, Singha A. Mukesh Chourasiab SA. Molecular insights into the differences in anti-inflammatory activities of green tea catechins on IL-1β signaling in rheumatoid arthritis synovial fibroblasts. Physiol Behav. 2016;176:139–48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Fan W Effects of hesperidin on H 2 O 2 -treated chondrocytes and cartilage in a rat osteoarthritis model. Med Sci Monit. 2018:9177–86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Huang X, Xi Y, Mao Z, Chu X, Zhang R, Ma X, et al. Vanillic acid attenuates cartilage degeneration by regulating the MAPK and PI3K/AKT/NF-κB pathways. Eur J Pharmacol [Internet]. Elsevier B.V.; 2019;859:172481. Available from: 10.1016/j.ejphar.2019.172481 [DOI] [PubMed] [Google Scholar]
- 63.Hagenlocher Y, Satzinger S, Civelek M, Feilhauer K, Köninger J, Bischoff SC, et al. Cinnamon reduces inflammatory response in intestinal fibroblasts in vitro and in colitis in vivo leading to decreased fibrosis. Mol Nutr Food Res. 2017;61:1–25. [DOI] [PubMed] [Google Scholar]
- 64.Comblain F, Dubuc JE, Lambert C, Sanchez C, Lesponne I, Serisier S, et al. Identification of targets of a new nutritional mixture for osteoarthritis management composed by curcuminoids extract, hydrolyzed collagen and green tea extract. PLoS One. 2016;11:1–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Ishii M, Nakahara T, Araho D, Murakami J, Nishimura M. Glycolipids from spinach suppress LPS-induced vascular inflammation through eNOS and NK-kB signaling. Biomed Pharmacother [Internet]. Elsevier Masson SAS; 2017;91:111–20. Available from: 10.1016/j.biopha.2017.04.052 [DOI] [PubMed] [Google Scholar]
- 66.Skyvalidas D, Mavropoulos A, Tsiogkas S, Dardiotis E, Liaskos C, Mamuris Z, et al. Curcumin mediates attenuation of proinflammatory interferon γ and interleukin 17 cytokine responses in psoriatic disease, strengthening its role as a dietary immunosuppressant. Nutr Res. Elsevier Inc. 2020;75:95–108. [DOI] [PubMed] [Google Scholar]
- 67.Qi W, Lin C, Fan K, Chen Z, Liu L. Chemico-biological interactions hesperidin inhibits synovial cell in fl ammation and macrophage polarization through suppression of the PI3K / AKT pathway in complete Freund’ s adjuvant-induced arthritis in mice. Chem Biol Interact [Internet]. Elsevier; 2019;306:19–28. Available from: 10.1016/j.cbi.2019.04.002 [DOI] [PubMed] [Google Scholar]
- 68.Kuo P-L. Myricetin inhibits the induction of anti-Fas IgM-, tumor necrosis factor- a - and interleukin-1 h -mediated apoptosis by Fas pathway inhibition in human osteoblastic cell line MG-63. Life Sci. 2005;77:2964–76. [DOI] [PubMed] [Google Scholar]
- 69.Min HK, Kim S, Baek S, Woo J, Park J, Cho L, et al. Anthocyanin extracted from black soybean seed coats prevents autoimmune arthritis by suppressing the development of Th17 cells and synthesis of proinflammatory cytokines by such cells, via inhibition of NF- κ B. PLoS One. 2015;1–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Tsao R Chemistry and biochemistry of dietary polyphenols. Nutrients. 2010;2:1231–46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Opara EI, Chohan M. Culinary herbs and spices: their bioactive properties, the contribution of polyphenols and the challenges in deducing their true health benefits. Int J Mol Sci. 2014;15:19183–202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Bhagwat S, Haytowitz DB. USDA Database for the flavonoid content of selected foods release 3.2. 2015; Available from: http://www.ars.usda.gov/nutrientdata
- 73.Wu D, Wang J, Pae M, Meydani SN. Green tea EGCG, T cells, and T cell-mediated autoimmune diseases. Mol Asp Med [Internet]. Elsevier Ltd.; 2012;33:107–18. Available from: 10.1016/j.mam.2011.10.001 [DOI] [PubMed] [Google Scholar]
- 74.Srividhya R, Jyothilakshmi V, Arulmathi K, Senthilkumaran V, Kalaiselvi P. Attenuation of senescence-induced oxidative exacerbations in aged rat brain by (−)-epigallocatechin-3-gallate. Int J Dev Neurosci. 2008;26:217–23. [DOI] [PubMed] [Google Scholar]
- 75.De Almeida GG, De Sá-Nakanishi AB, Wendt MMN, Comar JF, Bersani Amado CA, Bracht A, et al. Green tea extract improves the oxidative state of the liver and brain in rats with adjuvant-induced arthritis. Food Funct. 2015;6:2701–11. [DOI] [PubMed] [Google Scholar]
- 76.Matsuno H, Nakamura H, Katayama K, Hayashi S, Kano S, Yudoh K, et al. Effects of an oral administration of glucosamine-chondroitin-quercetin glucoside on the synovial fluid properties in patients with osteoarthritis and rheumatoid arthritis. Biosci Biotechnol Biochem. 2009;73:288–92. [DOI] [PubMed] [Google Scholar]
- 77.Haleagrahara N, Miranda-Hernandez S, Alim MA, Hayes L, Bird G, Ketheesan N. Therapeutic effect of quercetin in collagen-induced arthritis. Biomed Pharmacother [Internet]. Elsevier Masson SAS; 2017;90:38–46. Available from: 10.1016/j.biopha.2017.03.026 [DOI] [PubMed] [Google Scholar]
- 78.Li Y, Yang B, Yu Bai J, Xia S, Mao M, Li X, et al. The roles of synovial hyperplasia, angiogenesis and osteoclastogenesis in the protective effect of apigenin on collagen-induced arthritis. Int Immunopharmacol [Internet]. Elsevier; 2019;73:362–9. Available from: 10.1016/j.intimp.2019.05.024 [DOI] [PubMed] [Google Scholar]
- 79.Tejada S, Pinya S, Martorell M, Capó X, Tur JA, Pons A, et al. Potential anti-inflammatory effects of hesperidin from the genus citrus. Curr Med Chem. 2018;25:4929–45. [DOI] [PubMed] [Google Scholar]
- 80.Kometani T, Fukuda T, Kakuma T, Kawaguchi K, Tamura W, Kumazawa Y, et al. Effects of α-glucosylhesperidin, a bioactive food material, on collagen-induced arthritis in mice and rheumatoid arthritis in humans. Immunopharmacol Immunotoxicol. 2008;30:117–34. [DOI] [PubMed] [Google Scholar]
- 81.Homayouni F, Ahmadi K. Blood pressure lowering and anti - inflammatory effects of hesperidin in type 2 diabetes; a randomized double - blind controlled clinical trial. Phytother Res. 2018;1–7. [DOI] [PubMed] [Google Scholar]
- 82.Cheraghpour M, Alavian SM, Hekmatdoost A, Hedayati M. Hesperidin improves hepatic steatosis, hepatic enzymes, and metabolic and inflammatory parameters in patients with nonalcoholic fatty liver disease: a randomized, placebo - controlled, double - blind clinical trial. Phytother Res. 2019;1–8. [DOI] [PubMed] [Google Scholar]
- 83.Yu J, Bi X, Yu B, Chen D. Isoflavones: anti-inflammatory benefit and possible caveats. Nutrients. 2016;8:1–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Hu Y, Li J, Qin L, Cheng W, Lai Y, Yue Y, et al. Study in treatment of collagen-induced arthritis in DBA/1 mice model by genistein. Curr Pharm Des. 2016;22:6975–81. [DOI] [PubMed] [Google Scholar]
- 85.El-Ashmawy NE, Khedr EG, Shamloula MM, Kamel MM. Evaluation of the antirheumatic effects of isoflavone-free soy protein isolate and etanercept in rats with adjuvant-induced arthritis. Exp Biol Med. 2019;244:545–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Marhuenda J, Alemán MD, Gironés-vilaplana A, Pérez A, Caravaca G, Figueroa F, et al. In vitro availability of four different berries. 2016;2016. [Google Scholar]
- 87.Dai T, Shi K, Chen G, Shen Y, Pan T. Malvidin attenuates pain and inflammation in rats with osteoarthritis by suppressing NF- j B signaling pathway. Inflamm Res: Springer International Publishing; 2017. [DOI] [PubMed] [Google Scholar]
- 88.Sun YAN, Id O, Article O. Cyanidin-3-glucoside inhibits inflammatory activities in human fibroblast-like synoviocytes and in mice with collagen-induced arthritis. Clin Exp Pharmacol Physiol. 2018;0–1. [DOI] [PubMed] [Google Scholar]
- 89.Jean-Gilles D, Li L, Ma H, Yuan T, Chichester CO III, Seeram NP. Anti-inflammatory effects of polyphenolic-enriched red raspberry extract in an antigen induced arthritis rat model. J Agric Food Chem. 2013;60:5755–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Landete JM. Ellagitannins, ellagic acid and their derived metabolites: a review about source, metabolism, functions and health. Food Res Int [Internet]. Elsevier Ltd.; 2011;44:1150–60. Available from: 10.1016/j.foodres.2011.04.027 [DOI] [Google Scholar]
- 91.Calixto-Campos C, Carvalho TT, Hohmann MSN, Pinho-Ribeiro FA, Fattori V, Manchope MF, et al. Vanillic acid inhibits inflammatory pain by inhibiting neutrophil recruitment, oxidative stress, cytokine production, and NFκB activation in mice. J Nat Prod. 2015;78:1799–808. [DOI] [PubMed] [Google Scholar]
- 92.Rathi B, Bodhankar S, Mohan V, Thakurdesai P. Ameliorative effects of a polyphenolic fraction of Cinnamomum zeylanicum L. bark in animal models of inflammation and arthritis. Sci Pharm. 2013;81:567–89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Gupta Jain S, Puri S, Misra A, Gulati S, Mani K. Effect of oral cinnamon intervention on metabolic profile and body composition of Asian Indians with metabolic syndrome: a randomized double - blind control trial. Lipids Heal Dis. Lipids Health Dis. 2017;16:1–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Sang S, Chu YF. Whole grain oats, more than just a fiber: role of unique phytochemicals. Mol Nutr Food Res. 2017;61:1–31. [DOI] [PubMed] [Google Scholar]
- 95.Liu S, Yang N, Hou ZH, Yao Y, Lu L, Zhou XR, et al. Antioxidant effects of oats avenanthramides on human serum. Agric Sci China. 2011;10:1301–5. [Google Scholar]
- 96.Jones LLL, Jones G. Capsaicin for osteoarthritis pain. Prog Drug Res. 2014;68:277–91. [DOI] [PubMed] [Google Scholar]
- 97.Liu L, Simon SA. Similarities and differences in the currents activated by capsaicin, piperine, and zingerone in rat trigeminal ganglion cells. J Neurophysiol. 1996;76:1858–69. [DOI] [PubMed] [Google Scholar]
- 98.Bang JS, Oh DH, Choi HM, Sur BJ, Lim SJ, Yeon JY, et al. Anti-inflammatory and antiarthritic effects of piperine in human interleukin 1β-stimulated fibroblast-like synoviocytes and in rat arthritis models. Arthritis Res Ther. 2009;11:1–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Umar S, Golam Sarwar AHM, Umar K, Ahmad N, Sajad M, Ahmad S, et al. Piperine ameliorates oxidative stress, inflammation and histological outcome in collagen induced arthritis. Cell Immunol [Internet]. Elsevier Inc.; 2013;284:51–9. Available from: 10.1016/j.cellimm.2013.07.004 [DOI] [PubMed] [Google Scholar]
- 100.Ying X, Chen X, Cheng S, Shen Y, Peng L, Xu H. Piperine inhibits IL-β induced expression of inflammatory mediators in human osteoarthritis chondrocyte. Int Immunopharmacol [Internet]. Elsevier B.V.; 2013;17:293–9. Available from: 10.1016/j.intimp.2013.06.025 [DOI] [PubMed] [Google Scholar]
- 101.Panahi Y, Hosseini MS, Khalili N, Naimi E, Majeed M, Sahebkar A. Antioxidant and anti-inflammatory effects of curcuminoid-piperine combination in subjects with metabolic syndrome: a randomized controlled trial and an updated meta-analysis. Clin Nutr [Internet]. Elsevier; 2015;34:1101–8. Available from: 10.1016/j.clnu.2014.12.019 [DOI] [PubMed] [Google Scholar]
- 102.Dvorakova M, Landa P. Anti-inflammatory activity of natural stilbenoids: a review. Pharmacol Res [Internet]. Elsevier Ltd.; 2017; Available from: 10.1016/j.phrs.2017.08.002 [DOI] [PubMed] [Google Scholar]
- 103.Lomholt S, Mellemkjaer A, Iversen MB, Pedersen SB, Kragstrup TW. Resveratrol displays anti-inflammatory properties in an ex vivo model of immune mediated inflammatory arthritis. BMC Rheumatol; 2018;1–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Hussain SA, Marouf BH, Ali ZS, Ahmmad RS. Efficacy and safety of co-administration of resveratrol with meloxicam in patients with knee osteoarthritis: a pilot interventional study. Clin Interv Aging. 2018;13:1621–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Wang G, Xie X, Yuan L, Qiu J, Duan W, Xu B, et al. Resveratrol ameliorates rheumatoid arthritis via activation of SIRT1-Nrf2 signaling pathway. BioFactors. 2019;1–13. [DOI] [PubMed] [Google Scholar]
- 106.Correâ MG, Pires PR, Ribeiro FV, Pimentel SP, Cirano FR, Napimoga MH, et al. Systemic treatment with resveratrol reduces the progression of experimental periodontitis and arthritis in rats. PLoS One. 2018;13:1–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Wei Y, Jia J, Jin X, Tong W, Tian H. Resveratrol ameliorates inflammatory damage and protects against osteoarthritis in a rat model of osteoarthritis. Mol Med Rep. 2018;17:1493–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Helli B, Shahi MM, Mowla K, Jalali MT, Haghighian HK. A randomized, triple-blind, placebo-controlled clinical trial, evaluating the sesamin supplement effects on proteolytic enzymes, inflammatory markers, and clinical indices in women with rheumatoid arthritis. Phytother Res. 2019;33:2421–8. [DOI] [PubMed] [Google Scholar]
- 109.Helli B, Mowla K, Mohammadshahi M, Jalali MT. Effect of sesamin supplementation on cardiovascular risk factors in women with rheumatoid arthritis. J Am Coll Nutr. 2016;35:300–7. [DOI] [PubMed] [Google Scholar]
- 110.Jing R, Ban Y, Xu W, Nian H, Guo Y, Geng Y, et al. Therapeutic effects of the total lignans from Vitex negundo seeds on collagen-induced arthritis in rats. Phytomedicine. 2019;58:1–10. [DOI] [PubMed] [Google Scholar]
- 111.Kaur L, Rutherfurd SM, Moughan PJ, Drummond L, Boland MJ. Actinidin enhances gastric protein digestion as assessed using an in vitro gastric digestion model. J Agric Food Chem. 2010;58: 5068–73. [DOI] [PubMed] [Google Scholar]
- 112.Kim H, Banerjee N, Barnes RC, Pfent CM, Stephen T. Talcott RHD and SUM-T. Mango polyphenolics reduce inflammation in intestinal colitis— involvement of the miR-126/PI3K/AKT/mTOR axis in vitro and in vivo. Mol Carcinog. 2017;56:197–207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Pal R, Chaudhary MJ, Chandra P, Rajendra T, Kamlesh N, Pant K. Pharmacological and biochemical studies on protective effects of mangiferin and its interaction with nitric oxide (NO) modulators in adjuvant - induced changes in arthritic parameters, inflammatory, and oxidative biomarkers in rats. Inflammopharmacology [Internet]. Springer International Publishing; 2018; Available from: 10.1007/s10787-018-0507-8 [DOI] [PubMed] [Google Scholar]
- 114.Tsubaki M, Takeda T, Kino T, Itoh T, Imano M, Tanabe G. Mangiferin suppresses CIA by suppressing the expression of TNF-α, IL-6, IL-1β, and RANKL through inhibiting the activation of NF-κB and ERK1 / 2. Am J Transl Res. 2015;7:1371–81. [PMC free article] [PubMed] [Google Scholar]
- 115.Pandey S, Cabot PJ, Shaw PN, Amitha K. Anti-inflammatory and immunomodulatory properties of Carica papaya. J Immunotoxicol. 2016;13:590–602 6901. [DOI] [PubMed] [Google Scholar]
- 116.Rathnavelu V, Alitheen NB, Sohila S. Potential role of bromelain in clinical and therapeutic applications. Biomed Rep. 2016:283–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Kocaadam B, Şanlier N. Curcumin, an active component of turmeric (Curcuma longa), and its effects on health. Crit Rev Food Sci Nutr. 2017;57:2889–95. [DOI] [PubMed] [Google Scholar]
- 118.Semwal RB, Semwal DK, Combrinck S, Viljoen AM. Gingerols and shogaols: important nutraceutical principles from ginger. Phytochemistry. Elsevier Ltd. 2015;117:554–68. [DOI] [PubMed] [Google Scholar]
- 119.Fouda AMM, Berika MY. Evaluation of the effect of hydroalcoholic extract of Zingiber officinale rhizomes in rat collagen-induced arthritis. Basic Clin Pharmacol Toxicol. 2009;104:262–71. [DOI] [PubMed] [Google Scholar]
- 120.Ramadan G, Al-Kahtani MA, El-Sayed WM. Anti-inflammatory and anti-oxidant properties of Curcuma longa (turmeric) versus Zingiber officinale (ginger) rhizomes in rat adjuvant-induced arthritis. Inflammation. 2011;34:291–301. [DOI] [PubMed] [Google Scholar]
- 121.Ruangsuriya J, Budprom P, Viriyakhasem N, Kongdang P, Chokchaitaweesuk C, Sirikaew N, et al. Suppression of cartilage degradation by zingerone involving the p38 and JNK MAPK signaling pathway. Planta Med. 2017;83:268–76. [DOI] [PubMed] [Google Scholar]
- 122.Villalvilla A, da Silva JA, Largo R, Gualillo O, Vieira PC, Herrero-Beaumont G, et al. 6-Shogaol inhibits chondrocytes’ innate immune responses and cathepsin-K activity. Mol Nutr Food Res. 2014;58:256–66. [DOI] [PubMed] [Google Scholar]
- 123.Kim MK, Chung SW, Kim DH, Kim JM, Lee EK, Kim JY, et al. Modulation of age-related NF-κB activation by dietary zingerone via MAPK pathway. Exp Gerontol [Internet]. Elsevier Inc.; 2010;45:419–26. Available from: 10.1016/j.exger.2010.03.005 [DOI] [PubMed] [Google Scholar]
- 124.Ribel-Madsen S, Bartels EM, Stockmarr A, Borgwardt A, Cornett C, Danneskiold-Samsøe B, et al. A synoviocyte model for osteoarthritis and rheumatoid arthritis: response to ibuprofen, betamethasone, and ginger extract—a cross-sectional in vitro study. Arthritis. 2012;2012:1–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Jäger S, Trojan H, Kopp T, Laszczyk MN, Scheffler A. Pentacyclic triterpene distribution in various plants – rich sources for a new group of multi-potent plant extracts. Molecules. 2016;14:2016–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Ahmad A, Abuzinadah MF, Alkreathy HM, Banaganapalli B, Mujeeb M. Ursolic acid rich Ocimum sanctum L leaf extract loaded nanostructured lipid carriers ameliorate adjuvant induced arthritis in rats by inhibition of COX-1, COX-2, TNF- α and IL-1: Pharmacological and docking studies. PLoS One. 2018;13: e0193451 1–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Yu N, Han F, Lin X, Tang C, Ye J, Cai X. The association between serum selenium levels with rheumatoid arthritis. Biol Trace Elem Res. 2016;172:46–52. [DOI] [PubMed] [Google Scholar]
- 128.Sahebari M, Rezaieyazdi Z, Khodashahi M. Selenium and autoimmune diseases: a review article. Curr Rheumatol Rev. Bentham Science Publishers Ltd. 2018;15:123–34. [DOI] [PubMed] [Google Scholar]
- 129.Sanna A, Firinu D, Zavattari P, Valera P. Zinc status and autoimmunity: a systematic review and meta-analysis. Nutrients. MDPI AG; 2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Bao B, Prasad AS, Beck FWJ, Fitzgerald JT, Snell D, Bao GW, et al. Zinc decreases C-reactive protein, lipid peroxidation, and inflammatory cytokines in elderly subjects: a potential implication of zinc as an atheroprotective agent. Am J Clin Nutr. 2010;91: 1634–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Wu D, Lewis ED, Pae M, Meydani SN. Nutritional modulation of immune function: Analysis of evidence, mechanisms, and clinical relevance. Front Immunol. 2019;10:1–19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Xin L, Yang X, Cai G, Fan D, Xia Q, Liu L, et al. Serum levels of copper and zinc in patients with rheumatoid arthritis: a meta-analysis. Biol Trace Elem Res. Humana Press Inc. 2015;168:1–10. [DOI] [PubMed] [Google Scholar]
- 133.Zeng C, Li H, Wei J, Yang T, Deng ZH, Yang Y, et al. Association between dietary magnesium intake and radiographic knee osteoarthritis. PLoS One. 2015;10:1–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Shahi A, Aslani S, Ataollahi MR, Mahmoudi M. The role of magnesium in different inflammatory diseases. Inflammopharmacology. Birkhauser Verlag AG; 2019. p. 649–61. [DOI] [PubMed] [Google Scholar]
- 135.Duntas LH. Selenium and inflammation: underlying anti-inflammatory mechanisms. Horm Metab Res. 2009;41:443–7. [DOI] [PubMed] [Google Scholar]
- 136.Wang J, Um P, Dickerman BA, Liu J. Zinc, magnesium, selenium and depression: a review of the evidence, potential mechanisms and implications. Nutrients. 2018;10:1–19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Li H, Zeng C, Wei J, Yang T, Gao SG, Luo W, et al. Associations of dietary and serum magnesium with serum high-sensitivity C-reactive protein in early radiographic knee osteoarthritis patients. Mod Rheumatol. 2017;27:669–74. [DOI] [PubMed] [Google Scholar]
- 138.Ren SX, Zhang B, Lin Y, Ma DS, Yan H. Selenium nanoparticles dispersed in phytochemical exert anti-inflammatory activity by modulating catalase, GPx1, and COX-2 gene expression in a rheumatoid arthritis rat model. Med Sci Monit. 2019;25:991–1000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Brenner M, Laragione T, Gulko PS. Short-term low-magnesium diet reduces autoimmune arthritis severity and synovial tissue gene expression. Physiol Genomics. 2017;49:238–42. [DOI] [PubMed] [Google Scholar]
- 140.Chen R, Zhou X, Yin S, Lu Z, Nie J, Zhou W, et al. Study on the protective mechanism of autophagy on cartilage by magnesium sulfate. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2018;32: 1340–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Razavi M, Jamilian M, Kashan ZF, Heidar Z, Mohseni M, Ghandi Y, et al. Selenium supplementation and the effects on reproductive outcomes, biomarkers of inflammation, and oxidative stress in women with polycystic ovary syndrome. Horm Metab Res. 2016;48:185–90. [DOI] [PubMed] [Google Scholar]
- 142.Jamilian M, Mansury S, Bahmani F, Heidar Z, Amirani E, Asemi Z. The effects of probiotic and selenium co-supplementation on parameters of mental health, hormonal profiles, and biomarkers of inflammation and oxidative stress in women with polycystic ovary syndrome 11 Medical and Health Sciences 1103 Clinical Sciences. J Ovarian Res. 2018;11:1–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Rashidi AA, Salehi M, Piroozmand A, Sagheb MM. Effects of zinc supplementation on serum zinc and C-reactive protein concentrations in hemodialysis patients. J Ren Nutr [Internet]. National Kidney Foundation, Inc.; 2009;19:475–8. Available from: 10.1053/j.jrn.2009.04.005 [DOI] [PubMed] [Google Scholar]
- 144.Nielsen FH. Magnesium deficiency and increased inflammation: current perspectives. J Inflamm Res. 2018;11:25–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Mazidi M, Rezaie P, Banach M. Effect of magnesium supplements on serum C-reactive protein: a systematic review and meta-analysis. Arch Med Res. 2018;14:707–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Andretta A, Eliana M, Schieferdecker M, Petterle RR. Relations between serum magnesium and calcium levels and body composition and metabolic parameters in women with fibromyalgia. Adv Rheumatol. 2020;4:1–6. [DOI] [PubMed] [Google Scholar]
- 147.Veronese N, La Tegola L, Caruso MG, Maggi S, Guglielmi G. The association between dietary magnesium intake and magnetic resonance parameters for knee osteoarthritis. Nutrients. 2019;11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Zeng C, Wei J, Li H, Yang T, Zhang FJ, Pan D, et al. Relationship between serum magnesium concentration and radiographic knee osteoarthritis. J Rheumatol. 2015;42:1231–6. [DOI] [PubMed] [Google Scholar]
- 149.Wu Z, Yang J, Liu J, Lian K. The relationship between magnesium and osteoarthritis of knee: a MOOSE guided systematic review and meta-analysis. Medicine (Baltimore). 2019;98:e17774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Konstari S, Sares-Jäske L, Heliövaara M, Rissanen H, Knekt P, Arokoski J, et al. Dietary magnesium intake, serum high sensitivity C-reactive protein and the risk of incident knee osteoarthritis leading to hospitalization—a cohort study of 4,953 Finns. PLoS One. 2019;14:1–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Nielsen FH, Johnson LK, Zeng H. Magnesium supplementation improves indicators of low magnesium status and inflammatory stress in adults older than 51 years with poor quality sleep. Magnes Res. 2010;23:158–68. [DOI] [PubMed] [Google Scholar]
- 152.Mazarakis N, Snibson K, Licciardi PV, Karagiannis TC. The potential use of L-sulforaphane for the treatment of chronic inflammatory diseases: a review of the clinical evidence. Clin Nutr [Internet]. Elsevier Ltd.; 2020;39:664–75. Available from: 10.1016/j.clnu.2019.03.022 [DOI] [PubMed] [Google Scholar]
- 153.Lu QY, Rasmussen AM, Yang J, Lee RP, Huang J, Shao P, et al. Mixed spices at culinary doses have prebiotic effects in healthy adults: a pilot study. Nutrients. 2019;11:1–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Holscher HD, Taylor AM, Swanson KS, Novotny JA, Baer DJ. Almond consumption and processing affects the composition of the gastrointestinal microbiota of healthy adult men and women: a randomized controlled trial. Nutrients. 2018;10:126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Bamberger C, Rossmeier A, Lechner K, Wu L, Waldmann E, Fischer S, et al. A walnut-enriched diet affects gut microbiome in healthy caucasian subjects: a randomized, controlled trial. Nutrients. 2018;10:244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Hiel S, Bindels LB, Pachikian BD, Kalala G, Broers V, Zamariola G, et al. Effects of a diet based on inulin-rich vegetables on gut health and nutritional behaviour in healthy humans. Am J Clin Nutr. Oxford University Press. 2019;109:1683–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Häger J, Bang H, Hagen M, Frech M, Träger P, Sokolova MV, et al. The role of dietary fiber in rheumatoid arthritis patients: a feasibility study. Nutrients. 2019;11:2392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Roager HM, Vogt JK, Kristensen M, Hansen LBS, Ibrügger S, Mærkedahl RB, et al. Whole grain-rich diet reduces body weight and systemic low-grade inflammation without inducing major changes of the gut microbiome: a randomised. Gut. 2019:83–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Yang Q, Liang Q, Balakrishnan B, Belobrajdic DP, Feng Q. Role of dietary nutrients in the modulation of gut. 1–57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Pachikian BD, Neyrinck AM, Deldicque L, De Backer FC, Catry E, Dewulf EM, et al. Changes in intestinal bifidobacteria levels are associated with the inflammatory response in magnesium-deficient mice. J Nutr. 2010;140:509–14. [DOI] [PubMed] [Google Scholar]
- 161.Reed S, Neuman H, Moscovich S, Glahn RP, Koren O, Tako E. Chronic zinc deficiency alters chick gut microbiota composition and function. Nutrients. 2015;7:9768–84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Yow Boris, Stephen McBride Jessica Mowbray, Caughey William, Wong Edbert, Luey Christopher, Siddiqui Ahsan, Alexander Zanazir, Playle Veronica, Askelund Timothy. Enterobacteriaceae native joint septic arthritis [Internet]. Open Forum Infect Dis. 2018. [cited 2020 Jun 5]. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6254232/ [DOI] [PubMed] [Google Scholar]
- 163.Rinninella E, Raoul P, Cintoni M, Franceschi F, Abele G, Miggiano D, et al. What is the healthy gut microbiota composition? A changing ecosystem across age, environment, diet, and diseases. Microorganisms [Internet]. 2019. [cited 2020 Jun 5];7: 14. Available from: www.mdpi.com/journal/microorganisms [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Devries S A global deficiency of nutrition education in physician training: the low hanging fruit in medicine remains on the vine. Lancet Planet Heal [Internet]. The Authors(s). Published by Elsevier Ltd. This is an Open Access article under the CC BYNC-ND 4.0 license; 2019;3:e371–2. Available from: 10.1016/S2542-5196(19)30173-1 [DOI] [PubMed] [Google Scholar]
- 165.Kiraly LN, McClave SA, Neel D, Evans DC, Martindale RG, Hurt RT. Physician nutrition education. Nutr Clin Pr. 2014;29:332–7. [DOI] [PubMed] [Google Scholar]
- 166.Kondro W, Agreement PS, Health O, Plan I, Health L, Networks I. Most doctors lack adequate training to give informed diet advice n. Can Med Assoc J. 2012;184:418–20. [Google Scholar]
- 167.Crowley J, Ball L, Hiddink GJ. Nutrition in medical education: a systematic review. Lancet Planet Heal The Author(s). Published by Elsevier Ltd. This is an Open Access article under the CC BY-NC-ND 4.0 license; 2019;3:e379–89. [DOI] [PubMed] [Google Scholar]
- 168.Dumic A, Miskulin I, Pavlovic N, Cacic Kenjeric D, Orkic Z, Miskulin M. Attitudes toward nutrition care among general practitioners in croatia. J Clin Med. 2018;7:60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Stevens JF and Maier CS Department. The chemistry of gut microbial metabolism of polyphenols [Internet]. Physiol. Behav. 2017. Available from: file:///C:/Users/Carla Carolina/Desktop/Artigos para acrescentar na qualificação/The impact of birth weight on cardiovascular disease risk in the.pdf. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Bailey MA, Holscher HD. Microbiome-mediated effects of the Mediterranean diet on inflammation. Adv Nutr. 2018;9:93–206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Hajian-tilaki K Vitamin D deficiency and rheumatoid arthritis: epidemiological, immunological, clinical and therapeutic aspects behzad. Med J Rheumatol. 2019;30:94–102. [DOI] [PMC free article] [PubMed] [Google Scholar]


