Abstract
Background:
Despite the association between chronic pain and post-traumatic stress disorder (PTSD), little is known about the longitudinal course of pain and PTSD during cancer treatment.
Objectives:
We examined the prevalence of PTSD and chronic pain at three time periods in veterans with a diagnosis of cancer, and the relationship between the experience of pain and PTSD.
Methods:
Participants (N = 123) with oral-digestive cancers were recruited from the Veterans Healthcare System (age M = 65.31 and SD = 9.13; 98.4% male) and completed face to face interviews at 6, 12, and 18 months post-diagnosis. Measures included the Post-traumatic Stress Disorder Checklist-Stressor-Specific version (PCL-S), Primary care PTSD (PC-PTSD), and the Patient-Reported Outcomes Measurement Information System (PROMIS) Pain Impact Scale.
Results:
About one-third (26.8%) of the sample had chronic pain, defined as elevated pain at two time periods. About one-fifth (20.3%) endorsed symptoms of combat-related PTSD at 6 months, and 22.8% endorsed symptoms of cancer-related PTSD, exceeding a clinical cutoff for older adults (12 months = 21.1%, 18 months = 23.1%). Changes over time were observed for cancer-related PTSD symptom clusters of hyperarousal (F = 3.85 and p = 0.023) and emotional numbing (F = 4.06 and p = 0.018) with a statistically significant quadratic function increasing at 18 months. In logistic regression, individuals with both combat and cancer-related PTSD symptoms at six months had 8.49 times higher odds of experiencing chronic pain (χ2 = 25.91 and p < 0.001; R2 = 0.28).
Conclusions:
Persisting pain may be a concern in veterans with cancer. Individuals who have experienced traumatic events with persisting PTSD symptoms may be at elevated risk for chronic pain. Veterans with PTSD symptoms from both cancer and combat are at the highest risk to experience chronic pain.
Keywords: cancer, chronic pain, oncology, palliative care, post-traumatic stress disorder, trauma
Introduction
Numerous studies document associations between pain and post-traumatic stress disorder (PTSD). For individuals diagnosed with cancer, the relationship between PTSD and pain is particularly complex. Previous trauma, such as combat, is associated with increased pain in cancer patients.1 In a study of individuals with lung cancer undergoing thoracotomy, the pain associated with the procedure served both as a traumatic stressor and predicted symptoms of PTSD.2 Conversely, cancer pain may serve as a reminder of a traumatic stressor and drive further arousal.3 For some traumatic events, pain drives arousal and avoidance.3,4 For individuals with cancer, the potential relationship between pain and PTSD is bidirectional. Cancer pain serves as a particularly vexing potential trigger being an internal stressor, something that cannot be easily avoided.
PTSD symptoms may arise following a traumatic stressor, although most individuals do not develop PTSD following trauma exposure. Life-threatening illnesses such as cancer have been in and out of inclusion criteria as a potentially traumatic event in the Diagnostic and Statistical Manual of Mental Disorders (DSM) (e.g., DSM-III—out, DSM-IV—in, DSM-5—out).5–7 For DSM-5, language for a qualifying traumatic event changed identifying only medical incidents that involved catastrophic events. However, despite attempts at making PTSD criteria more objective, traumatization is a subjective experience. The individual’s perception of threat to life is critical.8 Importantly, previous research finds the perception of cancer as a life-threatening event and the development of cancer-related PTSD independent of cancer stage and severity.9,10
Setting aside changes to DSM inclusion criteria, nearly two decades of research document rates of cancer-related PTSD in cancer survivors from 0% to 35%, often in the range of 10%.11–13 This range is similar to rates of PTSD in persons following other traumatic stressors, although more individuals have symptoms of PTSD that do not meet full diagnostic criteria.14,15 The literature investigating the connection between PTSD and cancer focuses on relatively few types of malignancies. In these studies, PTSD in cancer survivors is associated with prior trauma history.13,16
In this article, we aim to extend the literature on PTSD and pain in adult cancer survivors with two goals. First, we describe the prevalence and longitudinal course of cancer-related PTSD and chronic pain in a sample of veterans following a diagnosis of cancer across varying stages and types. Second, we examine the relationship between PTSD symptoms associated with two potentially traumatic events, a recent stressor—cancer, and an earlier life stressor—combat, and chronic pain.
Methods
Participants
Participants were from the Veterans Cancer Rehabilitation Study (Vet-Cares).17 In this study, individuals were identified from Veterans Health Administration tumor registries in Boston and Houston and screened through the electronic medical record (EMR). Inclusion criteria were clinically confirmed diagnosis of and treatment for oral (head and neck), esophageal, gastric, or colorectal cancer. These cancer types were selected as they are relatively prevalent among veterans and have variable prognoses. Exclusion criteria were not English-speaking, presence of psychotic or dementing disorders, or currently receiving hospice care. Individuals were recruited through letter and phone.
Procedure
Participants completed in-person structured interviews 6(T1), 12(T2), and 18(T3) months following their cancer diagnosis at the medical center or in their home. This analysis focuses on 123 participants with complete data at all three time points. Of the 170 participants who completed T1 interviews, at T3, 14 were deceased, 16 declined interviews, and 17 could not be reached. The Institutional Review Boards of the Boston and Houston (Michael E. DeBakey) Veterans Affairs Medical Centers approved the study. Participants completed informed consent and were compensated for their time.
Measures
Demographics.
Participants reported their age, gender, racial and ethnic identities, and level of education.
Comorbidity index.
Comorbidity ratings used EMR extraction of 22 diagnoses from the Charlson Comorbidity Index, assigning a score of 1 for each illness.
Cancer information.
Information about the cancer diagnosis, stage, and treatment (surgery, chemotherapy, and/or radiation) was obtained from EMR.
Chronic pain.
Following the ICD-11 definition of chronic pain as lasting longer than three months, individuals were classified as having chronic pain if they reported elevated pain (>1 SD above community norms) for two time periods on the Pain Impact scale from the Patient-Reported Outcomes Measurement Information System (PROMIS) 29-item version.18,19 The Pain Impact scale is a reliable and valid measurement of chronic pain in cancer patients,20 considering the extent to which pain interferes with daily activities, work, social activities, and enjoyment of life, rated 1 (not at all) to 5 (very much), in the past seven days, for a possible range of 4–20.
Post-traumatic stress disorder.
Individuals were categorized as having no PTSD symptoms, elevated symptoms of either combat-related or cancer-related PTSD at time 1 or elevated symptoms of both combat-related and cancer-related PTSD at time 1. To assess combat-related PTSD, we asked if, while serving in the military, participants were “in a combat area” defined as “firearms, shelling, and mortars.” Of those who stated yes (39.8%), we administered the 4-item Primary Care PTSD Screen (PC-PTSD21;). A score of ≥3 was used to indicate a potential diagnosis,22 and a score of ≥1 was used to indicate elevated symptoms. This lower score was used because of its high sensitivity and our desire to be inclusive of adults who experience PTSD symptoms.21–23
The Post-traumatic Stress Disorder Check List-Stressor-Specific version (PCL-S)24,25 assessed cancer-related PTSD orienting participants to cancer as a potentially traumatic event (e.g., “Repeated, disturbing memories, thoughts, or images of cancer, your diagnosis, or treatment?”), as similarly adapted for breast cancer survivors.9,26,27 A score of ≥37 was used to indicate a potential diagnosis in older adults,28 while a score of ≥23 (median split) was used to indicate elevated symptoms.
To further investigate the symptom profile of PTSD in individuals with cancer, we created subscores for four clusters29 for each time point: reexperiencing (items B1–B5), effortful avoidance (C1–C2), emotional numbing (C3–C7), and hyperarousal (D1–D5). We standardized cluster subscores by dividing the raw score by the number of items in each cluster, to permit comparisons of symptom severity between clusters. Of note, the DSM-IV version of these instruments was utilized as the data collection occurred when DSM-IV was in use.
Data analysis
Data analyses addressed our two aims. First, to achieve our aim of describing the prevalence and longitudinal course of cancer-related PTSD and chronic pain, we used mean and frequency statistics and compared these over time with repeated-measure ANOVA, evaluating linear and quadratic trends, with post hoc paired t tests. Second, to achieve our aim of examining the relationship between PTSD symptoms from two traumatic events and chronic pain, we used multivariate logistic regression to determine the relationship of PTSD to chronic pain, controlling for demographic variables.
We used logistic regression rather than linear regression as our primary independent variable (PTSD) was categorical and our outcome variable (pain) was dichotomous. We controlled for potential demographic predictors by including those associated with pain at the bivariate level in the logistic regression. We had both categorical (e.g., race) and continuous (e.g., age) predictors, which we dichotomized to permit comparisons across predictors by odds ratios. Analyses were conducted in SPSS.
Results
Participants
Demographic data for participants are presented in Table 1. Participants were mostly male (98.4%). Age ranged from 48 to 88 years (M = 65.63 and SD = 8.80), with about half (48.0%) age 65+. The majority (71.5%) described their race as white or Caucasian (81%) and 28.5% as black or African American, Hispanic, or multiracial. About half (47.2%) had a high school education or less. In terms of health, participants had between 1 and 9 comorbidities at the beginning of treatment. Cancer types were colorectal (57.7%), head and neck (35.0%), and esogastric (7.3%), of AJCC Stage I (27.6%), II (28.5%), III (23.6%), and IV (20.3%), treated by surgery (78.9%), chemotherapy (56.1%), and/or radiation therapy (38.2%). All but 12% of participants were finished with treatment at 18 months post-diagnosis.
Table 1.
Demographic Characteristics and Univariate Comparisons
| Total sample demographics | Bivariate comparisons with chronic pain | ||||||
|---|---|---|---|---|---|---|---|
| Variable | Category | n | % | With, % | Without, % | χ2 | p |
| Age | <65 | 64 | 52.0 | 29.7 | 70.3 | 8.80 | 0.003 |
| >65 | 59 | 48.0 | 8.5 | 91.5 | |||
| Education | HS or less | 58 | 47.2 | 31.0 | 69.0 | 0.99 | 0.320 |
| College or more | 65 | 52.8 | 23.1 | 76.9 | |||
| Race | African American, Hispanic, Multiracial | 35 | 28.5 | 40.0 | 60.0 | 4.32 | 0.04 |
| Caucasian (non-Hispanic) | 88 | 71.5 | 21.6 | 78.4 | |||
| Comorbidity | Low | 72 | 58.5 | 29.2 | 70.8 | 0.48 | 0.49 |
| High | 51 | 41.5 | 23.5 | 76.5 | |||
| Cancer stage | Early (I–II) | 69 | 56.1 | 29.0 | 71.0 | 0.37 | 0.54 |
| Advanced (III–IV) | 54 | 43.9 | 24.1 | 75.9 | |||
| Treatment | Surgery | 97 | 78.9 | 28.9 | 71.1 | 0.97 | 0.33 |
| Chemotherapy | 69 | 56.1 | 26.1 | 73.9 | 0.04 | 0.83 | |
| Radiation therapy | 47 | 38.2 | 27.7 | 72.3 | 0.03 | 0.87 | |
| Combat PTSD | Any Current Sx | 25 | 20.3 | 52.0 | 48.0 | 10.13 | 0.001 |
| Cancer PTSD | Elevated Sx 6 months | 64 | 52.0 | 42.2 | 57.8 | 16.03 | <0.001 |
| Combined PTSD | None | 56 | 45.5 | 8.9 | 91.1 | 19.50 | <0.001 |
| Either | 45 | 36.6 | 35.6 | 64.4 | |||
| Both | 22 | 17.9 | 54.5 | 45.6 | |||
PTSD, post-traumatic stress disorder.
Pain
Pain impact scores.
Pain impact scores ranged from 4 to 20 at each time period. At 6 months post-diagnosis, the mean pain impact was M = 8.11 and SD = 0.94; at 12 months M = 8.12 and SD = 4.88; and at 18 months M = 8.68 and SD = 5.33. There was no difference in the mean level of pain impact reported at 6 months versus 12 months (t = 0.05 and p = 0.958) or 12 months versus 18 months (t = 1.24 and p = 0.213).
Elevated and chronic pain compared to community norms.
At 6 months post-diagnosis, 30.1% of the sample (n = 37) had pain that was elevated more than 1 SD above the community norm; at 12 months, 30.9% (n = 38) had elevated pain; and at 18 months, 30.1% (n = 37) had elevated pain. The percentage of the sample with chronic pain—elevated pain at two time periods—was 26.8% (n = 33). In univariate analyses (Table 1), chronic pain was associated with younger age, minority race, and PTSD, but not cancer type, stage, or treatments.
Post-traumatic stress disorder
Combat-related PTSD.
Forty-nine (n = 39.8%) participants stated, while in the service, they were also in combat. Within this sample, 13.8% (n = 17) scored above the cut score of 3 for PTSD, whereas 20.3% (n = 25) had symptoms of combat-related PTSD. In contrast, 19.5% (n = 24) stated they had been in a combat area while in the military, but do not have any current combat-related PTSD symptoms, and 60.2% (n = 74) stated that, while they were in the military, they were not in a combat area.
Cancer-related PTSD.
Cancer-related PTSD scores ranged from 17 to 70 at 6 months, M = 28.49 and SD = 12.90; at 12 months, M = 27.07 and SD = 12.08 (range 17–73); and at 18 months, M = 29.05 and SD = 14.34 (range 17–82). There was no difference in the mean level of PTSD reported at 6 months versus 12 months (t = 1.68 and p = 0.095), but there was a statistically significant increase from 12 months versus 18 months (t = 1.99 and p = 0.049). At 6 months, 22.8% of the sample was above a cut score of 37 for PTSD (12 months = 21.1% and 18 months = 23.1%).
Mean cancer-related PTSD cluster scores are presented in Table 2. The overall repeated-measure analysis of change over time was not statistically significant overall or for reexperiencing and avoidance. It was statistically significant for emotional numbing (F = 4.06 and p = 0.018) and hyperarousal (F = 3.85 and p = 0.023). Furthermore, the within-subjects quadratic function for the cluster emotional numbing was statistically significant (F = 7.10 and p = 0.0009), with a statistically significant decrease from time 1 to time 2 (t = 1.99 and p = 0.049) and statistically significant increase from time 2 to time 3 (t = −2.03 and p = 0.045). Similarly, the within-subjects quadratic function for the cluster hyperarousal was significant (F = 8.06 and p = 0.0005), with a statistically significant decrease from time 1 to time 2 (t = 2.04 and p = 0.044) and statistically significant increase from time 2 to time 3 (t = −2.21 and p = 0.029). Changes over time are visualized in Figure 1, using standardized cluster scores to permit comparisons between clusters. The mean standardized cluster score, averaging across time, was highest for hyperarousal (M = 1.82 and SD = 0.79), which was significantly higher than emotional numbing (M = 1.68 and SD = 0.76; t = −3.33 and p = 0.001), reexperiencing (M = 1.55 and SD = 0.71; t = −5.77 and p < 0.001), and avoidance (M = 1.39 and SD = 0.59; t = −8.78 and p < 0.001).
Table 2.
Cancer Related Post-traumatic Stress Disorder Cluster Scores on PCL-C (DSM-IV) at Each Time Point
| Time 1 (6 months) | Time 2 (12 months) | Time 3 (18 months) | ||||||
|---|---|---|---|---|---|---|---|---|
| Cluster | M | SD | M | SD | M | SD | F | p |
| Reexperiencing | 7.83 | 4.07 | 7.49 | 3.88 | 7.93 | 4.36 | 0.55 | 0.58 |
| Active avoidance | 2.72 | 1.42 | 2.71 | 1.55 | 2.90 | 1.67 | 0.42 | 0.67 |
| Emotional numbing | 8.56 | 4.51 | 7.82 | 3.84 | 8.80 | 4.92 | 2.79 | 0.07 |
| Hyperarousal | 9.38 | 4.52 | 8.57 | 4.00 | 9.39 | 5.06 | 2.66 | 0.07 |
DSM-IV, Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition; PCL-C, PTSD checklist-civilian version.
FIG. 1.
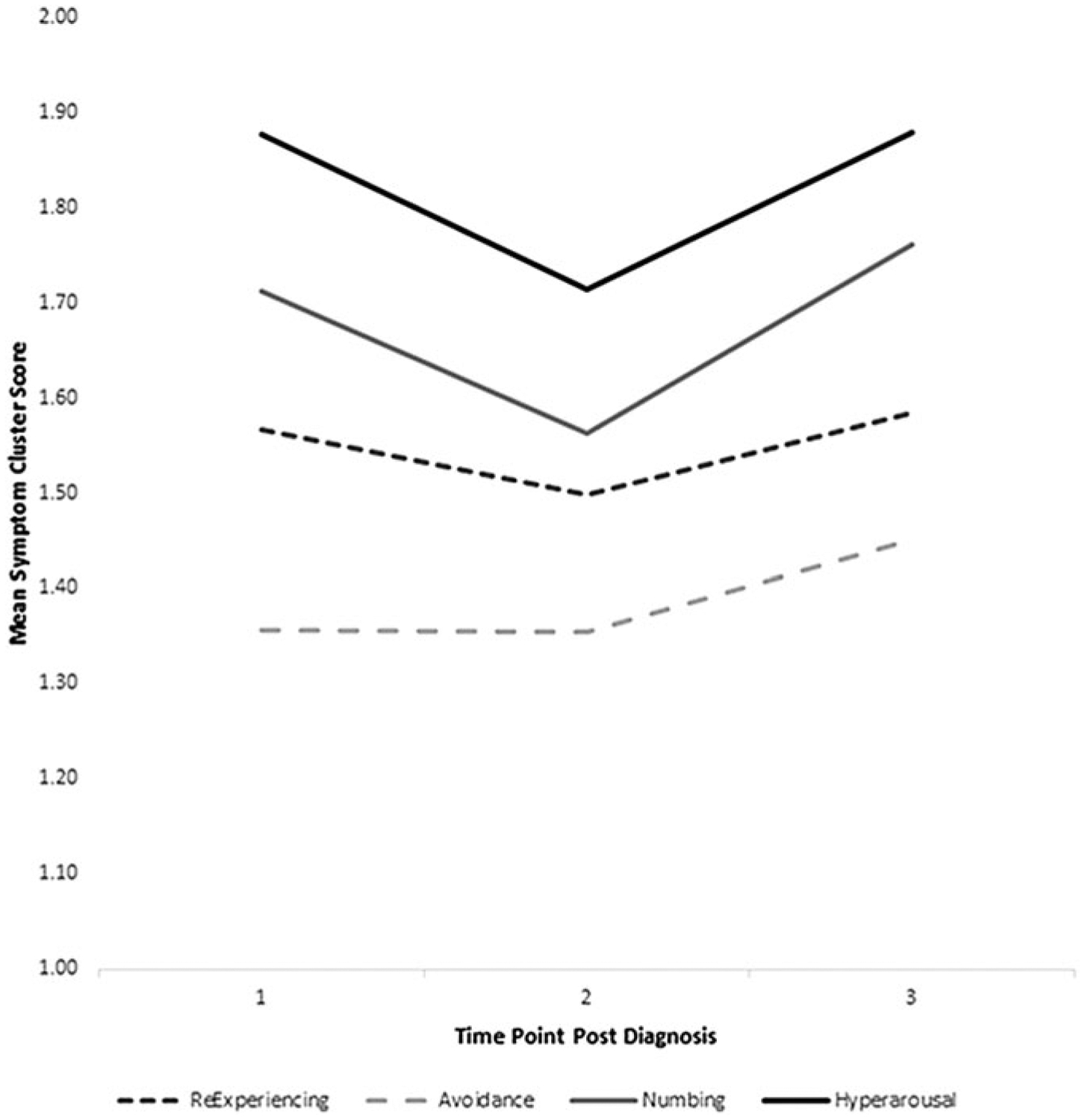
Cancer-related PTSD cluster subscores at 6, 12, and 18 months post-diagnosis. Subscores were standardized by dividing the number of items within each cluster, to permit comparison across clusters. PTSD, post-traumatic stress disorder.
Relationship of PTSD to chronic pain
In univariate comparisons (Table 1), having chronic pain was associated with younger age, racial or ethnic minority status, having combat-related PTSD symptoms, or having cancer-related PTSD symptoms. In multivariate logistic regression (Table 3), only PTSD symptoms were associated with chronic pain. Age and race were not predictive in the multivariate model. Individuals with both combat- and cancer-related PTSD symptoms had 8.49 times higher odds of chronic pain than those with neither combat- nor cancer-related PTSD symptoms. Individuals with either combat- or cancer-related PTSD symptoms have 4.24 times higher odds of having chronic pain (omnibus χ2 = 25.91 and p < 0.001; Nagelkerke R2 = 0.28).
Table 3.
Association of Demographic and Post-traumatic Stress Disorder Variables with Chronic Pain in Multivariate Analyses
| B | S.E. | Wald | p | Odds ratio | 95% CI | |
|---|---|---|---|---|---|---|
| Younger age | 0.96 | 0.51 | 3.60 | 0.058 | 2.62 | 0.97–7.08 |
| Minority race | 0.51 | 0.51 | 1.01 | 0.316 | 1.67 | 0.61–4.56 |
| Either combat or cancer PTSD Symptoms | 1.44 | 0.59 | 5.94 | 0.015 | 4.24 | 1.33–13.53 |
| Both combat and cancer PTSD symptoms | 2.14 | 0.67 | 10.28 | 0.001 | 8.49 | 2.29–31.38 |
Analysis included only those variables significantly associated with chronic pain in bivariate analyses. Reference group for PTSD analyses is the group with neither combat- nor cancer-related PTSD symptoms.
Discussion
The main findings within this sample are threefold. First, about one-quarter of sample individuals have chronic pain in the period 18 months post-diagnosis. Second, about one-fifth have probable cancer-related PTSD (based on cut-score) and more have elevated PTSD symptoms. Furthermore, these symptoms are relatively stable in the period 18 months post-diagnosis, with hyperarousal and emotional numbing symptoms most elevated with possible reexacerbation at 18 months. Third, PTSD symptoms are associated with the experience of chronic pain, and those with PTSD symptoms associated with combat and cancer are most at risk.
Until recently, most studies of cancer-related PTSD were cross-sectional, but in recent years, a longitudinal literature has emerged examining the course of PTSD in individuals with lung cancer (N = 93, over six months),30 breast cancer (N = 166, over one year),31 blood cancer, post-transplant (N = 102 [with complete data) over one year]32 ovarian cancer (N = 63 [with complete data] over three months), and mixed samples (N = 203 [with complete data] over four years)33 groups. In general, these studies find low rates of diagnosed PTSD (as low as 2.0%) and high rates of PTSD symptoms (as high as 82.5%), and relative stability with modest fluctuations in the short term, with potential improvement in the longer term (e.g., four years). Similar to our findings, PTSD symptomatology is an important concern in the short term for many, and the long term for some.
The complex relationships between PTSD and pain are intriguing, and bring an added dimension to the concept of “total pain.” While integrative treatment approaches for chronic pain and PTSD are reported in populations with noncancer pain, few integrative treatment approaches have been demonstrated for individuals with cancer-related pain.34 Approaches should consider treatment burden and possible community-based and telehealth approaches, as individuals move from active treatment to survivorship and are likely to seek fewer hospital-based visits.
For individuals with cancer and chronic pain, providers may consider screening for psychological risk and comorbidity factors, including PTSD. Future research is needed to understand if treatment aimed at improving symptoms of PTSD subsequently improves pain in this population. Interventions that focus on hyperarousal, the cluster most elevated in this sample, and associated with pain in other samples,35,36 may be particularly fruitful.
Several theoretical models postulate the underlying mechanism connecting PTSD and pain such as the shared vulnerability and mutual maintenance models.4 In brief, both shared vulnerability and mutual maintenance models suggest a role for anxiety sensitivity as a predisposing vulnerability, and mutual maintenance elaborates on the idea that pain may trigger reminders of trauma—triggering arousal—which may trigger pain.3 Avoidance behaviors may be a natural response and thought to potentially sustain or exacerbate both PTSD and pain.37 Studies are not consistent on these factors, but certainly point to a complex synergy between PTSD and pain.38 These relationships have been studied primarily when cancer is not the inciting traumatic event, which may have implications on how treatment is addressed.
In our sample, avoidance symptoms were lowest in severity, perhaps because individuals undergoing treatment and/or surveillance of their cancer are to some degree, unable to avoid interaction with the health care system. Further research is needed to understand the relationship between PTSD and pain in cancer, and to create and validate treatment interventions for cancer-related PTSD, as well as measurement of chronic pain as an outcome.
Notable in our study was the increased likelihood of chronic pain when individuals reported both combat-related and cancer-related PTSD symptoms. Previous trauma appeared to make sample individuals more susceptible to development of PTSD with future stressors. For individuals diagnosed with cancer, an assessment of previous trauma and current related PTSD symptoms may help clinicians to identify individuals at risk of chronic pain. In this sample 40% were combat veterans, some of whom had concurrent combat-related PTSD symptoms. Those with symptoms may be at risk, and those without may demonstrate important resiliency.
Limitations
There are numerous limitations to our study. Given the period of data collection, our PTSD screener does not reflect the newer four cluster diagnosis as defined in DSM-5. We assessed PTSD by self-report, and only focused on either combat or cancer as potentially traumatic events. Our study is limited by the use of two different methods to assess PTSD (one for combat and one for cancer), which were self-reported and not validated by a clinician, but both measures demonstrate similar efficacy.21 While we clued participants to trauma “e.g. from cancer,” PTSD symptoms from one trauma may overlap with PTSD symptoms from another. Furthermore, although bivariate analyses did not suggest an association between cancer stage and pain, small sample size limited our ability to consider differences in pain among multiple subgroups or within subgroups of individuals who may have had recurrence or progression of disease.
Lacking a validated method to define chronic pain in a cancer sample, we used a statistical approach of 1 SD elevation at two or more time periods, which needs clinical validation. We did not use a cancer-specific pain measure. Our pain measure may not have fully captured pain associated with specific treatments. Furthermore, as participants were identified through the cancer tumor registry, we did not have a measure of PTSD or pain before diagnosis. In addition, we dichotomized some continuous variables (e.g., age). We find such an approach enhances clinical application by producing more information closer to clinical reasoning for patients with complex comorbidities, but such dichotomization results in a loss of statistical information.
Our study participants were mostly male and recruited from the Veterans Health Administration. Although veteran status is common in the United States with approximately one in two men older than 65 years being veterans,39 and our findings on the prevalence of PTSD and pain are similar to non-veteran samples, the extent to the association between PTSD and pain in the veterans sampled are different from non-veterans is unknown. Our sample included different cancers, providing not only a good representation of a general palliative clinic or oncology practice but also making generalizing the association of pain and PTSD in specific cancer types difficult.
In conclusion, one-fifth of our sample of veterans with cancer experienced cancer-related PTSD, with prominent hyperarousal and emotionally numbing symptoms. PTSD symptoms were associated with chronic pain, and the risk of developing chronic pain increased with traumatic exposures. Further work is needed to replicate these results in other non-veteran samples, but our work deepens the understanding of “total pain.” Clinicians may consider the role of traumatic exposure and PTSD when managing chronic pain in cancer survivors.
Acknowledgments
We thank the members of the Veterans Cancer Rehabilitation Study (Vetcares) Research team. We are indebted to the veterans who have participated in our research studies and allow us to contribute to their health care. Funding for this project was provided by the Department of Veterans Affairs Rehabilitation Research and Development Service #5I01RX000104-02.
Funding Information
This material is the result of work supported with resources and the use of facilities at the Boston VA Medical Center and the Houston VA Health Services Research & Development Center of Excellence (HFP90-020) at the Michael E. DeBakey VA Medical Center.
Footnotes
Author Disclosure Statement
The authors have no conflict of interests relating to this study or this article. Portions of PTSD data from the “time 1” period only were published in the following article: Wachen et al.15
References
- 1.Moye J, June A, Martin LA, et al. : Pain is prevalent and persisting in cancer survivors: Differential factors across age groups. J Geriatr Oncol 2014;5:190–196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Jeantieu M, Gaillat F, Antonini F, et al. : Postoperative pain and subsequent PTSD-related symptoms in patients undergoing lung resection for suspected cancer. J Thorac Oncol 2014;9:362–369. [DOI] [PubMed] [Google Scholar]
- 3.Liedl A, O’Donnell M, Creamer M, et al. : Support for the mutual maintenance of pain and post-traumatic stress disorder symptoms. Psychol Med 2010;40:1215–1223. [DOI] [PubMed] [Google Scholar]
- 4.Asmundson GJ, Coons MJ, Taylor S, Katz J: PTSD and the experience of pain: Research and clinical implications of shared vulnerability and mutual maintenance models. Can J Psychiatry 2002;47:930–937. [DOI] [PubMed] [Google Scholar]
- 5.American Psychiatric Association: Diagnostic and Statistical Manual of Mental Disorders III. Washington, DC: American Psychiatric Association, 1990. [Google Scholar]
- 6.American Psychiatric Association: Diagnostic and Statistical Manual of Mental Disorders (Fourth Edition Text Revision) DSM-IV-TR. Washington, DC: American Psychiatric Association, 2000. [Google Scholar]
- 7.American Psychiatric Association: Diagnostic and Statistical Manual of Mental Disorders, 5th ed. Washington DC: American Psychiatric Association, 2013. [Google Scholar]
- 8.Larsen SE, Berenbaum H: Did the DSM-5 improve the traumatic stressor criterion?: Association of DSM-IV and DSM-5 criterion A with posttraumatic stress disorder symptoms. Psychopathology 2017;50:373–378. [DOI] [PubMed] [Google Scholar]
- 9.Cordova MJ, Andrykowski MA, Kenady DE, et al. : Frequency and correlates of posttraumatic-stress-disorder-like symptoms after treatment for breast cancer. J Consult Clin Psychol 1995;63:981–986. [DOI] [PubMed] [Google Scholar]
- 10.Mulligan EA, Wachen JS, Naik AD, et al. : Cancer as a criterion a traumatic stressor for veterans: Prevalence and correlates. Psychol Trauma 2014;6(Suppl 1):S73–S81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Mundy EA, Blanchard EB, Cirenza E, et al. : Posttraumatic stress disorder in breast cancer patients following autologous bone marrow transplantation or conventional cancer treatments. Behav Res Ther 2000;38:1015–1027. [DOI] [PubMed] [Google Scholar]
- 12.Guglietti CL, Rosen B, Murphy JK, et al. : Prevalence and predictors of posttraumatic stress in women undergoing an ovarian cancer investigation. Psychol Serv 2010;7:266–274. [Google Scholar]
- 13.Cordova MJ, Riba MB, Spiegel D: Post-traumatic stress disorder and cancer. Lancet Psychiatry 2017;4:330–338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Carlson EB, Dalenberg CJ: A conceptual framework for the impact of traumatic experiences. Trauma Violence Abuse 2000;1:4–28. [Google Scholar]
- 15.Wachen JS, Patidar SM, Mulligan EA, et al. : Cancer-related PTSD symptoms in a veteran sample: Association with age, combat PTSD, and quality of life. Psychooncology 2014;23:921–927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Green BL, Krupnick JL, Rowland JH, et al. : Trauma history as a predictor of psychologic symptoms in women with breast cancer. J Clin Oncol 2000;18:1084–1093. [DOI] [PubMed] [Google Scholar]
- 17.Naik AD, Martin LA, Karel M, et al. : Cancer survivor rehabilitation and recovery: Protocol for the Veterans Cancer Rehabilitation Study (Vet-CaRes). BMC Health Serv Res 2013;13:93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.PROMIS Health Organization and the PROMIS Cooperative Group: PROMIS Scoring Manual—Pain Interference. Bethesda MD: National Institutes of Health, 2019. [Google Scholar]
- 19.Treede RD, Rief W, Barke A, et al. : A classification of chronic pain for ICD-11. Pain 2015;156:1003–1007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Amtmann D, Cook KF, Jensen MP, et al. : Development of a PROMIS item bank to measure pain interference. Pain 2010;150:173–182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Prins A, Ouimette P, Kimerling R, et al. : The primary care PTSD screen (PC-PTSD): Development and operating characteristics. Prim Care Psychiatry 2004;9:9–14. [Google Scholar]
- 22.Freedy JR, Steenkamp MM, Magruder KM, et al. : Post-traumatic stress disorder screening test performance in civilian primary care. Fam Pract 2010;27:615–624. [DOI] [PubMed] [Google Scholar]
- 23.Dirkzwager AJ, Bramsen I, Van der Ploeg HM: The longitudinal course of posttraumatic stress disorder symptoms among aging military veterans. J Nerv Ment Dis 2001;189: 846–853. [DOI] [PubMed] [Google Scholar]
- 24.Weathers FW, Huska JA, Keane TM: PCL-C for DSM-IV. Boston, MA: National Center for PTSD–Behavioral Science Division, 1991. [Google Scholar]
- 25.Weathers FW, Litz BT, Herman DS, et al. : The PTSD checklist (PCL-C): Reliability, validity, and diagnostic utility. In: Annual Convention of the International Society for Traumatic Stress Studies. San Antonio, TX, 1993. [Google Scholar]
- 26.Andrykowski MA, Cordova MJ: Factors associated with PTSD symptoms following treatment for breast cancer: Test of the Andersen model. J Trauma Stress 1998;11:89–203. [DOI] [PubMed] [Google Scholar]
- 27.Andrykowski MA, Cordova MJ, Studts JL, et al. : Post-traumatic stress disorder after treatment for breast cancer: Prevalence of diagnosis and use of the PTSD checklist-civilian version (PCL-C) as a screening instrument. J Consult Clin Psychol 1998;66:86–590. [DOI] [PubMed] [Google Scholar]
- 28.Cook JM, Elhai JD, Arean PA: Psychometric properties of the PTSD Checklist with older primary care patients. J Trauma Stress 2005;18:371–376. [DOI] [PubMed] [Google Scholar]
- 29.King DW, Leskin GA, King LA, Weathers FW: Confirmatory factor analysis of the clinician-administered PTSD scale: Evidence for the dimensionality of posttraumatic stress disorder. Psychol Assess 1998;10:90–96. [Google Scholar]
- 30.Ni J, Feng J, Denehy L, et al. : Symptoms of posttraumatic stress disorder and associated risk factors in patients with lung cancer: A longitudinal observational study. Integr Cancer Ther 2018;17:1195–1203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Voigt V, Neufeld F, Kaste J, et al. : Clinically assessed posttraumatic stress in patients with breast cancer during the first year after diagnosis in the prospective, longitudinal, controlled COGNICARES study. Psychooncology 2017;26:74–80. [DOI] [PubMed] [Google Scholar]
- 32.Esser P, Kuba K, Scherwath A, et al. : Posttraumatic stress disorder symptomatology in the course of allogeneic HSCT: A prospective study. J Cancer Surviv 2017;11:203–210. [DOI] [PubMed] [Google Scholar]
- 33.Chan CMH, Ng CG, Taib NA, et al. : Course and predictors of post-traumatic stress disorder in a cohort of psychologically distressed patients with cancer: A 4-year follow-up study. Cancer 2018;124:406–416. [DOI] [PubMed] [Google Scholar]
- 34.Bosco MA, Gallinati JL, Clark ME: Conceptualizing and treating comorbid chronic pain and PTSD. Pain Res Treat 2013;2013:1–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Lopez-Martinez AE, Ramirez-Maestre C, Esteve R: An examination of the structural link between post-traumatic stress symptoms and chronic pain in the framework of fear-avoidance models. Eur J Pain 2014;18:1129–1138. [DOI] [PubMed] [Google Scholar]
- 36.Feinberg RK, Hu J, Weaver MA, et al. : Stress-related psychological symptoms contribute to axial pain persistence after motor vehicle collision: Path analysis results from a prospective longitudinal study. Pain 2017;158:682–690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Andersen TE, Karstoft KI, Brink O, Elklit A: Paincatastrophizing and fear-avoidance beliefs as mediators between post-traumatic stress symptoms and pain following whiplash injury—A prospective cohort study. Eur J Pain 2016;20:1241–1252. [DOI] [PubMed] [Google Scholar]
- 38.Ravn SL, Hartvigsen J, Hansen M, et al. : Do post-traumatic pain and post-traumatic stress symptomatology mutually maintain each other? A systematic review of cross-lagged studies. Pain 2018;159:2159–2169. [DOI] [PubMed] [Google Scholar]
- 39.Federal Interagency Forum on Aging-Related Statistics: Older Americans 2016: Key Indicators of Well-Being. Federal Interagency Forum on Aging-Related Statistics. Washington, DC: U.S. Government Printing Office. August 2016. https://agingstats.gov/docs/LatestReport/Older-Americans-2016-Key-Indicators-of-WellBeing.pdf (last accessed March 23, 2020). [Google Scholar]


